Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Sustainable and eco-friendly bacterial cellulose production using agro-forest waste for food packaging applications: a review
Tubanur Avcia,
Aslıhan Çalhanb,
Eda Nur Ayar-Sumerc,
Canan Dogan a,
Hamid Kaddamide,
Oguzhan Gunduza and
Roger Narayan
a,
Hamid Kaddamide,
Oguzhan Gunduza and
Roger Narayan *f
*f
aDepartment of Metallurgical and Materials Engineering, Faculty of Technology, Marmara University, Turkey
bDepartment of Chemical Engineering, Faculty of Chemical and Metallurgical Engineering, Yıldız Technical University, Turkey
cDepartment of Food Engineering, Faculty of Chemical and Metallurgical Engineering, Istanbul Technical University, Maslak, 34469 Istanbul, Turkey
dIMED-Lab, Team of Organometallic and Macromolecular Chemistry-Composite Materials, Department of Chemical Sciences, Faculty of Science and Technology, Cadi Ayyad University, Marrakech 40000, Morocco
eSustainable Materials Research Center (SusMat-RC), Mohammed VI Polytechnic University (UM6P), Lot 660, Hay Moulay Rachid, Ben Guerir 43150, Morocco
fJoint Department of Biomedical Engineering, North Carolina State University, Raleigh, USA. E-mail: roger_narayan@ncsu.edu
First published on 30th March 2026
Abstract
Bacterial cellulose (BC) is a biopolymer with several unique properties, including high purity, crystallinity, biocompatibility, biodegradability, and excellent mechanical strength. However, high production costs, mainly due to expensive culture media, limit large-scale industrial use. The European Union Packaging and Packaging Waste Regulation (PPWR) aims for all packaging placed on the EU market to be recyclable in an economically viable manner by the year 2030, which requires compostability for specific categories and limits certain single-use plastic formats. As such, the packaging industry urgently requires truly circular barrier materials. This review explores the potential of agro-forest waste as a sustainable and cost-effective carbon source for BC production, aiming to reduce both cost and environmental impact. Studies have highlighted successful BC production from agricultural and forest waste materials such as tree bark, fruit leaves, waste brewed tea, and ground coffee. Despite promising results, research on producing BC from agro-forest wastes specifically for food packaging remains limited. BC is considered suitable for food packaging due to its ultrafine network, high surface area, and strong mechanical properties. However, there is still a lack of comprehensive studies, particularly those applying life cycle assessment (LCA), to evaluate the circular economy and environmental impact of different BC production methods. By providing insights into BC production from agro-forest waste and its application in food packaging, this review considers the development of sustainable and economically viable production processes. It also describes the untapped potential of agro-forest waste valorization in advancing environmentally friendly biopolymer alternatives for packaging solutions.
Sustainability spotlightBacterial cellulose is a biopolymer that exhibits high purity, crystallinity, biocompatibility, biodegradability, and excellent mechanical strength. This manuscript reviews the potential of agroforestry waste as a sustainable, cost-effective carbon source for BC production to reduce both costs and environmental impact. BC is considered suitable for food packaging due to its ultrafine network, high surface area, and strong mechanical properties. This manuscript describes the untapped potential of agro-forest waste valorisation for advancing environmentally friendly biopolymer alternatives for packaging solutions. This work aligns with UN SDG 12 (Responsible Consumption and Production) by considering sustainable material cycles, SDG 13 (Climate Action) by considering a reduction in fossil-based plastic use, and SDG 2 (Zero Hunger) by considering food preservation practices. |
1. Introduction
Cellulose, a highly abundant organic polymer, is an essential structural component in the primary cell walls of green plants, many types of algae, and oomycetes. In addition to these eukaryotic sources, cellulose is synthesized as an extracellular polysaccharide by specific bacteria. This bacterial version, termed “bacterial cellulose” (BC), is a highly hydrated biofilm that is devoid of lignin, hemicellulose, and pectin, which sets it apart from its plant-derived counterpart.1,2Various bacterial species belonging to the genera Komagataeibacter, Acetobacter, Agrobacterium, Pseudomonas, Sarcina, and Rhizobium are known for their capacity to produce BC.2,3
The intrinsic properties of BC, such as transparency, purity, high water holding capacity, cell proliferation, cell adhesion, biocompatibility, high crystallinity, biodegradability, hydrophilicity, high mechanical strength, high degree of polymerization, nano-porosity, permeability to liquids and gases, flexibility, and moldability, make it an excellent material for many applications. Due to its high crystallinity, mechanical strength, and biocompatibility, BC has attracted significant attention across food, biomedical, and industrial applications.4 These attributes also make BC an ideal candidate for food packaging applications; for example, its biocompatibility and mechanical strength can enhance the durability and safety of packaging materials, while its thermal stability and chemical purity ensure the preservation and protection of food products.2,5
At the same time, the global food packaging sector is undergoing a significant transformation that is driven by sustainability pressures. The packaging industry currently accounts for over 40% of global virgin plastic consumption, and the growing evidence of microplastic accumulation in ecosystems and human tissues has intensified efforts to develop renewable and biodegradable alternatives. In this context, BC has emerged as a promising bio-based material that is capable of addressing both environmental and functional packaging requirements.
Despite its remarkable physicochemical properties, large-scale BC production remains economically constrained due to the high cost of conventional Hestrin–Schramm (HS) medium and relatively low volumetric productivity. To address these limitations, recent research efforts have increasingly focused on two parallel strategies: strain optimization and low-cost substrate valorization.
Recent investigations have significantly expanded the range of efficient BC-producing strains by isolating them directly from biowaste-rich ecological niches. For example, biowaste-soil consortia have been explored as reservoirs for high-yield cellulose-producing bacteria; this approach shows enhanced adaptability to alternative carbon sources and low-cost substrates.6 Similarly, novel Komagataeibacter strains isolated from agro-residual environments exhibit improved metabolic flexibility and cellulose productivity.7 In addition to strain development, cost-effective screening methodologies have been proposed to accelerate the identification of high-performing producers. The use of alternative low-cost media (e.g., gram flour-table sugar formulations) enables rapid and economical strain selection, which reduces dependence on synthetic laboratory media.8 In parallel, agro-industrial waste streams such as papaya peel and fruit pomace have been successfully valorized as fermentation substrates.7,9 Integrating strain improvement, economical screening, and waste substrate utilization facilitates the transition from laboratory-scale optimization toward techno-economically viable BC production systems that are aligned with circular bioeconomy principles.
Agro-forest wastes provide significant economic value on a global scale, offering a promising renewable energy source due to their environmentally friendly nature. Utilizing agro-forest waste is crucial for sustainable bacterial cellulose production. Key sources of agro-forest waste for BC production include agricultural residues, peels, wood residues, and leaf litter. Several agro-industrial wastes with potential utilization in BC production are highlighted in this research, including oranges10 and citrus peels,11 sugarcane juice,12 durian shells,13 wheat straw hydrolysate,14 coffee cherry husks,14 and pineapple residues,15 all of which have demonstrated promising yields across different bacterial strains.16 Rather than representing isolated case studies, these examples collectively demonstrate the versatility of agro-residual biomass as a fermentable carbon source that is capable of significantly reducing medium costs while maintaining comparable cellulose quality.
In addition, cacao mucilage exudates, a byproduct of cocoa fermentation, have been successfully applied for BC production and subsequently valorized for polyhydroxyalkanoate (PHA) synthesis, demonstrating their potential in integrated biopolymer production.17 Similarly, spent Pleurotus mushroom substrate has been converted into cellulose nanofibers, further illustrating the versatility of agro-wastes in generating high-value biomaterials.18
The unit cost of BC produced in standard Hestrin–Schramm medium has been estimated at approximately USD 4–6 kg−1, a level that continues to constrain its large-scale commercialization. By substituting refined sugars with agro-industrial residues such as fruit peels, winery effluents, or soy whey, the medium contribution can be lowered, and overall production costs can be reduced to around USD 2–3 kg−1, while simultaneously adding value to under-utilized biomass streams. Beyond terrestrial residues, marine biomass such as red seaweeds has also been minimally processed into bio-based films for food packaging, broadening the spectrum of renewable feedstocks available for sustainable material development.19 Furthermore, facile synthesis methods have recently demonstrated the high potential of specific marine biomass (e.g., Laminaria hyperborea) to be effectively converted into high-quality cellulose films and fibers via inverted nozzle-pressurized gyration, eliminating the need for cumbersome and toxic traditional dissolution methods.20 The global BC market was valued at roughly USD 0.6 billion in 2023, and is anticipated to expand at a compound annual growth rate of 12–13% through 2030, driven primarily by rising demand for sustainable packaging, biomedical devices, and high-value textiles.21
In this review, the sustainable production of BC from agricultural residues is considered for potential use in environmentally friendly food packaging. BC, synthesized using waste materials such as fruit peels, wheat straw, and sugarcane molasses, not only addresses the issue of waste management but also offers a renewable, biodegradable alternative to plastic packaging. The inherent properties of BC, such as high tensile strength, good barrier qualities, and biocompatibility, make it particularly suitable for food packaging applications. Utilizing agricultural residues as a substrate improves the life cycle environmental performance of BC production by reducing upstream agricultural inputs, minimizing waste disposal impacts, and lowering cumulative energy demand.22 Beyond material performance, the transition of BC-based systems toward commercial food packaging requires alignment with regulatory frameworks governing food contact materials, as well as systematic evaluation of environmental impacts via the Life Cycle Assessment (LCA) approach. In this context, techno-economic feasibility, migration compliance, and identification of LCA hotspots such as fermentation energy demand and downstream processing steps become critical determinants of industrial scalability. Future research should therefore focus not only on optimizing functional performance, such as improving water resistance, flexibility, and mechanical durability to meet industry standards, but also on integrating regulatory readiness and environmental performance validation into process development. This dual focus on waste utilization and sustainable packaging could significantly reduce plastic waste and promote the transition towards a circular economy.
2. Methodology
This review was conducted using a structured narrative approach. Scientific databases, including Web of Science, Scopus, and ScienceDirect, were consulted to identify relevant peer-reviewed articles published primarily between 2015 and 2025. Keywords used in various combinations included: “bacterial cellulose”, “agro-industrial waste”, “agro-forest waste”, “food packaging”, and “life cycle assessment”. A structured literature search was conducted using the Web of Science Core Collection database. The following thematic search strings were applied within the Topic field (Title, Abstract, Keywords):(i) (“Bacterial cellulose” and (“agro-industrial waste” or “agricultural waste” or “agro-forest waste”)),
(ii) (“Bacterial cellulose” and “food packaging”), and.
(iii) (“Bacterial cellulose” and “life cycle assessment”).
The search was limited to peer-reviewed articles and review papers that were published between 2015 and 2025. This approach yielded 127 records related to waste-derived BC production, 409 records addressing food packaging applications, and 54 records focusing on life cycle assessment aspects. Relevant articles were further screened based on thematic alignment with agro-waste valorization, industrial feasibility, and resource circularity assessment.
3. Biochemical and genetic mechanisms of BC biosynthesis
The biosynthesis of bacterial cellulose is a regulated multi-step process comprising intracellular precursor synthesis and extracellular hierarchical assembly. Intracellularly, the synthesis of the direct cellulose precursor from glucose involves four key enzymatic steps. First, glucokinase catalyses the phosphorylation of glucose into glucose-6-phosphate, which is subsequently isomerized to glucose-1-phosphate by phosphoglucomutase. Following this, UDP-glucose pyrophosphorylase (UGPase) synthesizes uridine diphosphoglucose (UDPG), the vital nucleotide sugar precursor for cellulose. Finally, the cellulose synthase enzyme complex polymerizes UDPG into β-1,4-glucan chains.23,24 Genetically, this synthesis is governed by the cellulose synthase (bcs) operon, typically comprising bcsA, bcsB, bcsC, and bcsD genes. For example, the bcsA gene encodes the catalytic subunit responsible for UDP-glucose conversion, while the pathway is stringently regulated by enzymes such as diguanylate cyclase and phosphodiesterase.24,25 Once synthesized, the linear glucan chains are extruded through the bacterial cell envelope as 1.5 nm-wide protofibrils. In the extracellular environment, these protofibrils self-assemble via strong hydrogen bonding into microfibrils, eventually coalescing into 20–100 nm wide crystalline ribbons to form a dense 3D matrix.23 Understanding this biochemical and genetic machinery is an important consideration, as culture conditions and alternative substrates directly affect these metabolic pathways, thereby determining the yield and structural properties of the resulting BC.4. carbon sources derived from agricultural and agro-industrial waste
4.1 Agricultural residues
Agricultural and agro-industrial residues provide a plentiful and economical supply of carbohydrates, making them attractive substrates for BC production. These residues, often including straw, husks, shells, and cobs, are rich in carbohydrates, making them a promising carbon source for BC production. For example, corn stalk hydrolysate, which contains glucose, xylose, mannose, and other compounds, has been used to produce 2.86 g L−1 of BC under optimized conditions. The BC fibrils synthesized by Acetobacter xylinum exhibit a range from 20 to 70 nm in terms of diameter and several micrometers in terms of length.26 Enzymatically processed and glucose-enriched rice husk was used to produce 2.42 g L−1 BC under optimized conditions. The BC fibrils synthesized by Acetobacter xylinum had diameters ranging from 40 to 60 nm.27Wheat straw is a widely available biomass that is often burned after the harvest of wheat grains, contributing significantly to air pollution. By applying acid or enzymatic hydrolysis, this biomass can be transformed into a viable nutrient source for the production of BC. In one study, enriching lime-detoxified wheat straw hydrolysate with nitrogen-rich nutrients led to a BC yield of 8.3 g L−1.28 Another investigation showed that treating wheat straw hydrolysates with calcium hydroxide resulted in a BC yield of 15.4 g L−1, which was 60% higher than yields obtained from media containing glucose, sucrose, and mannitol.29 Additionally, research indicated that enzymatically hydrolyzed wheat straw, subjected to both chemical and thermal pretreatment and with a total sugar concentration of 52.12 g L−1, produced 10.6 g L−1 of BC. Furthermore, using acid-hydrolyzed wheat straw with a total sugar concentration of 43 g L−1 as the fermentation medium yielded 9.7 g L−1 of BC.30
Rice is one of the most widely consumed agricultural crops globally, and its processing generates significant amounts of lignocellulosic waste, particularly rice straw (RS) and rice husk. Rice straw, typically found in the fields after harvest, is considered a low-value material and poses considerable environmental challenges. However, due to its lignocellulosic composition, rice straw holds substantial potential as a resource for producing high-value compounds, especially in the context of food packaging. Utilizing rice straw for such purposes exemplifies the principles of a circular economy, in which agricultural waste is repurposed into valuable products, thereby reducing environmental impact and promoting sustainability. Given that rice is the second most produced crop globally, following wheat, with an annual waste generation of approximately 972 teragrams, the potential for converting rice straw into bio-based packaging materials represents a significant opportunity to address both environmental and economic concerns.31
Photphisutthiphong et al.32 explored the production of BC using organic low-grade rice as a substrate. The research highlights the use of several rice varieties, including Riceberry rice, jasmine rice, red jasmine rice, brown jasmine rice, and Homnil rice, as potential substrates for BC production. The process involves enzymatic hydrolysis of the rice to convert starch into reducing sugars, which are then fermented by the bacterium Komagataeibacter nataicola TISTR 975. The study found that brown jasmine rice yielded the highest amount of bacterial cellulose, with a significant yield of 401.717 g L−1 (on a wet weight basis) after 15 days of incubation. The produced bacterial cellulose was further evaluated for its sensory properties when incorporated into food products, demonstrating high consumer acceptability. This research indicates that low-grade rice, often considered a by-product of the rice industry, can be effectively repurposed as a valuable resource for producing bacterial cellulose, thereby contributing to waste reduction and providing a sustainable alternative for food packaging materials.
Goelzer et al.33 conducted a study on the production and characterization of BC using Acetobacter xylinum ATCC 23769, and processed rice bark as a carbon source. They found that rice bark, an agricultural residue, could be effectively used as a substrate for BC production when combined with glucose. The study involved pre-treating the rice bark enzymatically and then using it in both static fermentation processes and aerated fermentation processes. They observed that the bacterial cellulose produced from rice bark exhibited a unique nanostructure, forming microspheres and nanospheres that were interconnected by cellulose nanofibers. They noted that components present in the rice bark, such as lignins and hemicelluloses, influenced the morphology and crystallinity of the cellulose, which results in the formation of type II cellulose under certain conditions. These findings suggest that the rice bark-derived BC has potential applications in drug delivery and other advanced material fields, demonstrating that agricultural waste can be converted into valuable nanostructured biopolymers.
Sudying et al.34 investigated the production and characterization of BC using rice washing drainage (RWD) as a carbon source, with the bacterium Komagataeibacter nataicola Li1 as the microorganism. The study aimed to explore the feasibility of using RWD, an agro-industrial waste, for BC production, potentially reducing production costs and environmental impact. The research compared BC production in RWD to that in traditional yeast extract (YE) medium under both static culture conditions and agitated culture conditions. The results showed that K. nataicola Li1 produced comparable amounts of BC in static cultures using either RWD or YE medium, with both yielding about 0.20 g L−1 of dry BC. However, in agitated cultures, the YE medium produced significantly more BC than RWD. The BC from RWD had a smaller average particle size (32 nm) compared to that from YE medium (49 nm) but displayed similar structural properties.
4.2 Peels
Recent studies have explored the use of fruit and vegetable peels as a carbon source for BC production, leveraging their high sugar content and bioactive compounds. For instance, peels from citrus fruits, bananas, and other commonly discarded plant materials have been successfully used to produce BC. This approach not only reduces waste but also adds value to these by-products, aligning with circular economy principles. The inedible peels of fruits and vegetables constitute 5–40% of their total weight and are rich in reducing sugars, vitamins, proteins, and acids. Therefore, these waste materials have the potential to be utilized as substrates for the production of value-added products such as bacterial cellulose (BC). Many studies have highlighted the effective utilization of food waste and agricultural residues as cost-efficient substrates for BC production.6 Employing these alternative media not only aids in waste removal from the environment, helping to reduce pollution, but also offers substantial environmental and economic benefits. Additionally, BC produced from these waste materials often displays distinct mechanical and physical characteristics when compared to BC generated from traditional carbon and nitrogen sources. For instance, BC produced from mango peel waste has been reported to possess higher crystallinity than BC produced from other media.35 Additionally, BC synthesized in white grape pomace medium exhibited enhanced water retention capacity and flexibility.36 Studies on banana peel extract have suggested that it can partially replace traditional nitrogen and carbon sources for BC production.37 Moreover, the enzymatic hydrolysis of citrus peels, such as orange peel, has been shown to increase the amount of fermentable sugars available for bacterial use, thereby significantly enhancing BC production.38 These findings indicate that various organic waste materials can serve as efficient substrates for BC production, and integrating waste management with BC production can yield both environmental and economic advantages.5,22,39The production of BC from fruit and vegetable peels represents a promising strategy for integrating waste valorization with biopolymer synthesis. Several studies have demonstrated that fruit peels can serve as efficient alternative carbon sources for BC production. For instance, pineapple peel waste utilized with Komagataeibacter saccharivorans APPK1 resulted in a 43% increase in BC yield through response surface methodology (RSM) and artificial neural network (ANN) optimization approaches.40 Additionally, research utilizing various fruit peels, such as mango and dragon fruit, demonstrated the viability of agricultural wastes as alternative carbon sources, with some isolates yielding high BC concentrations.41 These findings demonstrate the potential of converting agricultural waste into high-value bioproducts such as BC, contributing to lower life cycle impact intensity.
4.3 Wood
Wood waste primarily consists of lignocellulosic biomass, including hemicellulose, cellulose, and lignin. These components make wood waste a rich source of fermentable sugars after appropriate pretreatment. The process of converting these complex carbohydrates into simpler sugars is an important parameter for their use in bacterial cellulose production.Kiziltas et al.42 explored the synthesis of bacterial cellulose using sugars extracted from wood via hot water extraction. The study demonstrated that the extracted sugars could produce a BC yield of 2.8 g L−1, comparable to traditional glucose-based media. The research indicated that utilizing lignocellulosic biomass from wood, a common by-product in the wood and paper industries, could serve as an alternative carbon source for BC production, thereby reducing waste and production costs.
Kadimaliev et al.43 examined the use of residual brewer's yeast biomass combined with bacterial cellulose as a bio-adhesive for producing pressed materials from waste wood. The combination resulted in materials with a tensile strength of 18 MPa and a Young's modulus of 2.3 GPa, which are comparable to those of phenol-formaldehyde resins. This study highlighted the potential of using BC as a sustainable adhesive in the wood industry, offering an eco-friendly alternative to traditional toxic adhesives.
The bacterial cellulose yields obtained from various agricultural waste sources using different carbon and culture sources are provided in Table 1.
| Culture source | Agro-industrial wastes | Sample preparation | Maximum BC productivity | Additional nutrients | Cultivation time (days) | Reference |
|---|---|---|---|---|---|---|
| Komagataeibacter hansenii GA2016 | Citrus peels (lemon, Mandarin, orange, and grapefruit) | Hydrolysis with dilute acid | 2.06–3.92 g L−1 (dry weight) | Hestrin–Schramm medium | 21 | 44 |
| Gluconacetobacter medellinensis | Sugar cane juice and pineapple residues | Juice extraction + filtration (no chemical pretreatment) | 3.24 g L−1 (dry weight) | Hestrin–Schramm medium | 13 | 45 |
| Gluconacetobacter xylinus CH001 | Discarded waste durian shell | Acid hydrolysis | 2.67 g L−1 (dry weight) | Hestrin–Schramm medium | 10 | 13 |
| Acetobacter xylinus ATCC 23770 | Wheat straw | Alkali pretreatment + enzymatic hydrolysis | 8.3 g L−1 (dry weight) | Hestrin–Schramm medium | — | 28 |
| Gluconacetobacter hansenii UAC09 | Coffee cherry husk | Acid hydrolysate | 8.2 g L−1 (dry weight) | Corn steep liquor-10% alcohol-0.5% | 14 | 14 |
| Komagataeibacter rhaeticus | Cashew tree exudates | Not specified (direct use) | 6.0 g L−1 (dry weight) | Hestrin–Schramm medium | 7 | 46 |
| Acetobacter xylinum 0416 MARDI | Extracted date syrup | Dilution (direct use) | 5.8 g L−1 (dry weight) | Hestrin–Schramm medium | 10 | 47 |
| Acetobacter pasteurianus PW1 | Pineapple waste medium (PIWAM) and Pawpaw waste medium (PAWAM) | Juice extraction (direct use) | Piwam: 1 g L−1 (dry weight) | Hestrin–Schramm medium | 15 | 48 |
| PAWAM:3.9 g L−1 (dry weight) | ||||||
| Gluconoacetobacter xylinus BCRC 12334 | Orange peel fluid and orange peel hydrolysate | Enzymatic hydrolysis with cellulase & pectinase | 3.40 g L−1 (dry weight) | Acetate buffer, peptone, and yeast extract | 5 | 38 |
| Gluconacetobacter swingsii | Pineapple peel and sugar cane juice | Direct juice use (no pretreatment | 2.8 g L−1 (dry weight) | Glucose, fructose, and sucrose | 13 | 49 |
| Komagataeibacter hansenii MCM B-967 | Pineapple and watermelon peels | Direct peel extract (no pretreatment) | 125 g L−1 (wet weight) | Sucrose, ammonium sulfate, and cycloheximide | 7 | 50 |
| Kombucha | Apple residues (carbon source) | Direct use (no pretreatment) | 0.21 g L−1 (dry weight) | Hestrin–Schramm medium | 7 | 51 |
| K. hansenii DSM 5602 | Pea pod hydrolysate, mixed-vegetable peel hydrolysate (sole source), celery root peel (supplemented) | Acid hydrolysis | 1.14 g L−1 | Hestrin–Schramm medium | 14 | 52 |
| 1.09 g L−1 | ||||||
| 1.08 g L−1 (dry weight) | ||||||
| K. hansenii UCP1619 | Tomato residue (sole source) | Direct use (no pretreatment) | 8.33 g L−1 (dry weight) | Hestrin–Schramm medium | 14 | 53 |
| K.melomenusus AV436T | Grape pomace hydrolysate (sole source) | Hydrolysis of grape pomace | 1.24 g L−1 | Hestrin–Schramm medium | 4 | 54 |
| 0.98 g L−1 (dry weight) | ||||||
| K.europaeus BCRC 14,148 | Orange extract, papaya extract (carbon source) | Enzymatic hydrolysis with cellulase and pectinase | 3.47 g L−1 | Hestrin–Schramm medium | 7 | 38 |
| 3.48 g L−1 (dry weight) | ||||||
| Achromobacter S33 | Waste peel hydrolysate of apple, citrus, orange, banana, pomegranate, pineapple, mango | Acid hydrolysis | 0.27 g L−1 | Hestrin–Schramm medium | 7 | 35 |
| 0.0018 g L−1 | ||||||
| 0.32 g L−1 | ||||||
| 0.006 g L−1 | ||||||
| 0.38 g L−1 | ||||||
| 1.22 g L−1 (dry weight) | ||||||
| Komagataeibacter sp. CCUG73629 | Corncob (supplemented) | Dilute acid hydrolysis of corncob | 1.6 g L−1 | Hestrin–Schramm medium | 10 | 55 |
| 1.2 g L−1 (dry weight) | ||||||
| K. xylinus | Oil palm empty fruit bunch hydrolysate | Dilute acid hydrolysis of oil palm empty fruit bunch | 2.27 g L−1 (dry weight) | Yeast extract-1% | 8 | 56 |
| Gluconacetobacter xylinus | Pineapple peel waste juice | Juice extraction | 3.8 g L−1 (dry weight) | Sucrose-0.75% ammonium sulfate 0.5% | 14 | 15 |
| K. rhaeticus and K. intermedius | Pear peel and pomace | Direct fermentation (no chemical pretreatment) | 10.94 g L−1 (dry weight) | Glucose-3.5% | 7 | 57 |
| A.xylinum NBRC 13,693 | Apple grape juices Japanese pear orange pineapple | Direct juice use (no pretreatment) | 4.0 g L−1 | Hestrin–Schramm medium | 14 | 58 |
| 1.8 g L−1 | ||||||
| 3.8 g L−1 | ||||||
| 5.9 g L−1 | ||||||
| 4.1 g L−1 (dry weight) | ||||||
| Komagataeibacter nataicola | Banana peel | Boiling and filtration (peel extract) | 0.89 g L−1 (dry weight) | Hestrin–Schramm medium | 9 | 41 |
| Komagataeibacter hansenii | Pineapple and watermelon peels | 30 g L−1 (wet weight) | Hestrin–Schramm medium | 7 | ||
| Komagataeibacter sucrofermentans | Lemon peel | 5.20 g L−1 (dry weight) | Hestrin–Schramm medium | 13 | ||
| Komagataeibacter spp. (Isolate BE073) | Dragon fruit peel | 4.07 g L−1 (dry weight) | Hestrin–Schramm medium | 14 | ||
| Komagataeibacter spp. (Isolate BE052) | Papaya peel | 1.08 g L−1 (dry weight) | Hestrin–Schramm medium | 14 | ||
| Komagataeibacter nataicola TISTR 975 | Organic low-grade rice | Enzymatic hydrolysis with α-amylase & glucoamylase | 401.717 ± 5.383 g L−1 (wet weight) | (NH4)2SO4, alpha-amylase, glucoamylase | 15 | 32 |
| Acetobacter xylinum ATCC 23769 | Rice bark | Rice bark extract | 2.42 g L−1 (dry weight) | Glucose, citric acid | 10 | 33 |
| Komagataeibacter nataicola Li1 | Rice washing drainage | Direct use (no pretreatment | 0.20 g L−1 (dry weight) | Yeast extract, KH2PO4, MgSO4·7H2O, (NH4)2SO4 | 13 | 34 |
| Komagataeibacter rhaeticus QK23 | Agroindustrial asparagus waste | Hydrolysis of asparagus waste | 2.57 g L−1 (dry weight) | Hydrolysed waste | 25 | 59 |
| Novacetimonas sp | Cereal wastes (rice bran, cereal dust) | Fungal solid-state fermentation (SSF) | 1.55 g L−1 (dry weight) | Fungal SSF-treated | 15 | 60 |
| Komagataeibacter sucrofermentans | Restaurant and supermarket wastes | Fermentation broth enriched with organic acids & ethanol | 2.05 gL−1/day (dry weight) | Organic acids, ethanol | Not specified | 61 |
| Gluconacetobacter hansenii | Agro-industrial wastewater (glycerol, vinasse, whey) | Direct use (no pretreatment) | Not specified | Glycerol-based substrate | 28 | 62 |
| Microbial Consortium BA2 | Rice bran | Rice bran extract (direct use) | 4.07 g L−1 (dry weight) | Headspace air | 15 | 63 |
| Komagataeibacter xylinus | Cheese whey | Direct use (no pretreatment) | 7.947 ± 0.618 g L−1 (dry weight) | Ethanol | 6–11 | 64 |
| Komagataeibacter sucrofermentans | Orange juice (food waste) | Direct use (no pretreatment) | 6.4 g L−1 (dry weight) | Ethanol + organic acids | 5 | 61 |
| Komagataeibacter xylinus | Banana peel waste | Direct use (no pretreatment) | 30 g L−1 (wet weight) | None specified | 21 | 65 |
| Acetobacter xylinum | Discarded fruit waste | Soxhlet extraction of neem and sage | Not specified (membranes with high WHC and antimicrobial activity) | Neem and sage extracts (20 wt%) | Not specified | 66 |
| Komagataeibacter xylinus | Fruit processing wastewater | Sequential membrane fractionation | Not specified (improved WHC, tensile strength, crystallinity | Hestrin–Schramm medium + wastewater fractions | Not specified | 67 |
| Microbial consortium | Decayed fruit waste juice | Extraction of decayed fruit juice | 21.719 g L−1 (dry weight) | Glucose (0.75% w/v) | 5 | 68 |
| Komagataeibacter xylinus DSM 2004 | Waste apple pulp | Extraction (glucose & fructose) | 3.38 ± 0.09 g L−1 (dry weight) | Hestrin–Schramm medium | Not specified | 69 |
| Komagataeibacter xylinus DSM 2004 | Stale bread | Dilute acid hydrolysis | 2.07 ± 0.22 g L−1 (dry weight) | Hestrin–Schramm medium | Not specified | 69 |
5. Comparative techno-economic feasibility and industrial scalability
Although numerous studies report promising BC yields from many types of agro-industrial residues, a direct comparison reveals substantial variability in productivity, process complexity, and industrial feasibility. Reported maximum dry weight yields frequently range between 1.0 g L−1 and 15.4 g L; however, when normalized per fermentation time, productivity differences become more evident. For instance, while wheat straw treated with calcium hydroxide achieved a high yield of 15.4 g L−1, it required complex alkaline and enzymatic pretreatments. In contrast, simpler substrates such as orange juice or pineapple waste can yield significant BC (3.8–6.4 g L−1) with minimal processing, offering a more direct route for industrial scale-up.Pretreatment intensity represents a critical techno-economic determinant. Lignocellulosic residues such as wheat straw or rice husk require intensive acid/enzymatic hydrolysis and detoxification, which introduce additional chemical inputs and energy consumption. From a cost perspective, replacing refined sugars in Hestrin–Schramm medium with these residues can reduce production costs from approximately USD 4–6 kg−1 to USD 2–3 kg−1. Specifically, waste-based alternatives have been shown to reduce medium costs by 50% to 98% compared to traditional HS medium.
Fermentation mode further dictates scalability and material quality. Static systems, while simple and producing BC with high crystallinity and mechanical strength, are constrained by oxygen transfer limitations and large surface-area requirements. Agitated fermentation significantly enhances volumetric productivity and dissolved oxygen levels, but often results in BC with lower crystallinity and altered nanofibril organization. Consequently, industrial viability depends on balancing high-throughput production (agitated) with the specific mechanical requirements of food packaging (static).
Downstream processing and purification remain the most energy-intensive stages. Life cycle assessment (LCA) data identifies raw material production (e.g., sodium dihydrogen phosphate) and energy-intensive drying as major environmental “hotspots.” To mitigate these impacts, emerging low-energy manufacturing techniques, such as nozzle-pressurized spinning (NPS), have been proposed to eliminate the need for high voltages and enable high-throughput, continuous production.
Finally, supply chain stability is an important consideration. Seasonally available residues (e.g., rice straw) present logistical challenges related to availability, whereas continuously generated by-products (e.g., cheese whey or brewery effluent) provide more reliable feedstock streams.62 As indicated by the EU's Packaging and Packaging Waste Regulation (PPWR), the transition to these types of circular models is not only an economic necessity but a regulatory requirement to ensure that all packaging is recyclable in an economically viable manner by 2030; compostability is mandated only for certain packaging categories.
6. Fermentation processes
6.1 Static fermentation
Static culture is a straightforward and widely used method for bacterial cellulose production. In this method, cellulose typically forms as a gelatinous film at the air-liquid interface. The medium remains stationary, allowing BC to develop on the surface. However, static culture generally requires long incubation times and large surface areas, which leads to relatively low productivity. As the BC pellicles reach a certain thickness at the air-liquid interface, production slows down due to nutrient depletion and oxygen deficiency at the lower part of the pellicle.70,71Despite its simplicity, static culture has some limitations, including low efficiency and the formation of spontaneous mutants. For example, Komagataeibacter hansenii grown under static conditions has been shown to develop cellulose-negative spontaneous mutants, reducing yield. These issues can sometimes be mitigated by developing cost-effective environments or partially modifying the medium to increase production rates, such as adding ethanol or other supplements to enhance oxygen availability. The production of BC in static culture begins with the formation of island-like particles at the air-liquid interface, which gradually cover the entire surface. Once the pellicle completely covers the surface, growth slows because microorganisms cannot simultaneously access both oxygen and nutrients. When BC production reaches its maximum thickness, the production rate significantly decreases and eventually ceases. This mechanism has been observed in studies using K. xylinus, where pellicle formation was rapid initially but slowed markedly after several days due to the aforementioned oxygen limitation70,71
Several conditions affect BC production in static culture. For instance, while long incubation periods are required, extending this process contributes minimally to BC yield. An adequate supply of both oxygen and nutrients is essential for sustained BC production; otherwise, depletion of these resources leads to reduced output. Additionally, the pH of the medium can drop rapidly within the first few days, which further limits BC production. These factors should be carefully considered as they directly influence the efficiency and quality of BC production in static culture.70,71
The static culture method presents several advantages and disadvantages for bacterial cellulose production. Among its advantages, the simplicity of the method stands out, as it does not require complex equipment; moreover, the method is straightforward to set up and manage. This simplicity makes static culture an accessible and user-friendly option. Additionally, BC produced under static culture conditions generally exhibits stable mechanical properties, crystallinity, and polymerization degree, which enables the production of high-quality BC. From a cost perspective, static culture can be cost-effective when processing conditions are optimized, allowing production costs to be kept low under appropriate circumstances. Furthermore, the static culture method allows for direct oxygen exposure at the air-liquid interface, which facilitates effective oxygen contact with the BC.70–72
Yılmaz and Goksungur 66 evaluated the static fermentation process of waste figs using Komagataeibacter xylinus to obtain BC, which was optimized through RSM by varying parameters such as the initial sugar concentration, initial pH, and temperature. The highest yield of 8.45 g L−1 was obtained at pH 6.05, 62.75 g L−1 sugar, and 30 °C.73
In another study, BC production from Komagataeibacter sucrofermentans was studied using synthetic HS medium and agro-industrial side streams (industrial raisins finishing side stream extract, orange juice, and green tea extract), both individually and in combination. The yield of the non-fortified substrate, a combination of industrial raisin finishing side stream extract, orange juice, and green tea, was 5.9 g L−1, whereas a yield of 19.4 g L−1 was achieved with the industrial raisin finishing side stream extract medium fortified with thiamine, ascorbic, citric, and gallic acids. This increase demonstrates the importance of nutrient fortification and the design of the fermentation matrix in enhancing production from agro-industrial side streams.74
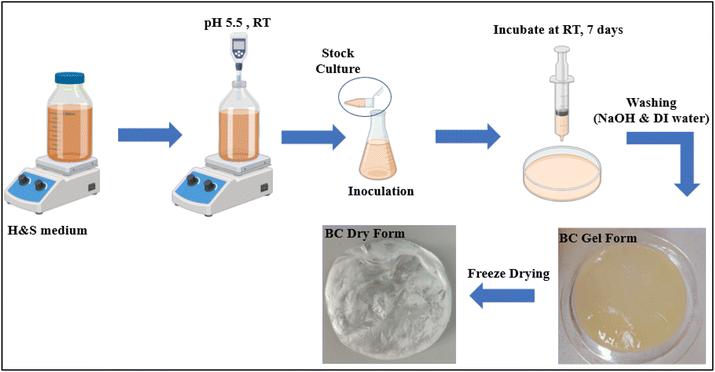
However, there are also several disadvantages associated with the static culture method. The static culture method typically requires long culture periods and large surface areas, which often results in low productivity and can pose challenges for large-scale production. After the pellicle covers the air-liquid interface completely, the production rate significantly decreases and eventually ceases, impacting the overall yield of BC production. Issues such as low yield and the formation of spontaneous mutants can increase production costs and require additional optimization. While static culture is generally suitable for small-scale production, it can present difficulties for large-scale production due to space and time requirements. Additionally, once the pellicle fully covers the interface, oxygen limitations can occur, ultimately halting growth.75,76 The diagram (Fig. 1) illustrates the static fermentation stage, highlighting the key steps associated with the bacterial cellulose production process.
6.2 Agitated fermentation
Agitated culture is a method of bacterial cellulose production in which the fermentation medium is constantly stirred or shaken. This process is different from static culture, in which the medium remains undisturbed. The agitation provides several advantages in BC production, particularly in improving yield and influencing the structural properties of the cellulose.Agitation ensures that the bacteria (e.g., Acetobacter xylinum) receive a continuous supply of oxygen. Since these bacteria are aerobic, oxygen is an important factor for their metabolism and cellulose production. In agitated systems, the dissolved oxygen levels remain high, leading to faster bacterial growth and increased cellulose yield.
The constant movement of the culture medium ensures that nutrients are evenly distributed, preventing localized nutrient exhaustion. This phenomenon helps in maintaining consistent growth conditions for the bacteria throughout the fermentation process. A recent study investigated the production of BC through agitated fermentation;77 they optimized BC production using A. xylinum and pineapple waste as a substrate, demonstrating that agitation at 120 rpm with the addition of microparticles increased the cellulose yield by 70.23% compared to static culture. Cheng et al.78 also explored the use of various additives in agitated conditions, finding that carboxymethylcellulose (CMC) significantly enhanced BC production, providing an output of 8.2 g L−1 compared to 1.3 g L−1 under static conditions. Lazarini et al.79 studied the production of spherical BC (SBC) in agitated cultures, showing that SBC had a larger surface area and improved suitability for drug delivery applications due to the benefits of agitation.
BC produced in an agitated culture typically exhibits a lower crystallinity and a higher water-holding capacity compared to that produced in a static culture. This difference is a result of the constant disruption of the growing cellulose fibers. The BC formed in an agitated culture also tends to have a lower degree of polymerization and mechanical strength compared to the denser, better-structured pellicles formed in static culture.80
Singhsa et al.81 focused on studying how different bacterial strains of Komagataeibacter xylinus and various carbon sources impact the physical properties of BC produced using static and agitated fermentation conditions. This study concluded that bacterial strain, carbon source, and fermentation method (i.e., static or agitated) significantly affect the yield and physical characteristics of bacterial cellulose. Under static conditions, BC formed as membrane-like sheets on the surface, while under agitated conditions, BC particles exhibited spherical or asterisk-like shapes. BC produced under static conditions had a more ordered structure, higher crystallinity, and larger crystallite sizes compared to BC produced under agitated conditions, which showed lower crystallinity and smaller crystallite sizes due to the mechanical stress of agitation. Agitated conditions lead to more porous and less crystalline BC, making it potentially more useful for applications requiring materials with a high surface area (e.g., adsorbents or biocompatible scaffolds).
Agitated culture is a powerful method for enhancing bacterial cellulose production by improving oxygenation and nutrient distribution. It also creates cellulose with unique physical properties that can be tailored for various industrial and biomedical applications. Although agitated cultures generally produce higher yields, they also result in BC with different structural characteristics compared to BC produced using static cultures.
7. Applications of bacterial cellulose as food packaging
In recent years, significant advancements have taken place in food packaging technology. Many of these innovations are associated with the development of intelligent and active packaging systems. Active packaging systems aim to extend the shelf life of products and improve quality by facilitating interaction between packaging components and food products. On the other hand, intelligent packaging utilizes technologies that can monitor, record, track, or transmit information about the product, packaging, or environment. These systems offer a viable alternative to synthetic polymers, both in terms of environmental compatibility and economic benefits. Recent comprehensive reviews indicate that valorizing many types of waste streams into nanocellulose not only resolves critical ecological threats but also yields robust, lightweight materials with tailored crystallinity, which significantly enhance barrier properties, moisture control, and biodegradability in advanced food packaging systems.82 BC has been the subject of various studies aiming to enhance the mechanical performance of packaging and explore its use in active packaging applications. For instance, BC nanocrystals have been employed as reinforcing fillers in hybrid nanocomposites to develop environmentally friendly packaging solutions. The unique structure of BC also facilitates the integration of functional compounds, making it effective in preserving food quality. Recent studies have made significant advancements in antimicrobial and biodegradable food packaging materials. For example, Yordshahi et al.83 prepared an antimicrobial nanofabric for beef wrapping by incorporating lyophilized postbiotics from Lactobacillus plantarum, which proved effective against Listeria monocytogenes. Xie et al.84 found that adding BC to potato peel-based films enhanced the tensile strength of the films, lowered the water vapor permeability of the films, decreased the oxygen permeability of the films, and reduced the moisture content of the films. These films were further improved by incorporating curcumin, which imparted antioxidant properties and functioned as a pH indicator and boric acid tracer, making the films beneficial for food safety and food quality preservation. Similarly, Yang et al.85 developed in situ hybrid films combining BC and chitin nanofibers with curcumin nanoparticles, which offered antioxidant and antimicrobial properties, as well as pH-responsive color changes and sensitivity to boric acid. Zahan et al.86 created biodegradable antimicrobial packaging by treating BC with lauric acid, effectively inhibiting Bacillus subtilis and showing notable biodegradability. Furthermore, Haghighi et al.87 formulated a gelatin-polyvinyl alcohol (GL/PVA) composite film containing BC, leading to a 22% reduction in the water vapor transmission rate and a 14% decrease in water vapor permeability. These innovations in food packaging improve product protection and longevity while meeting the increasing demand for sustainable as well as eco-friendly packaging solutions. Table 2 summarizes the selected organisms and modifications for food packaging applications involving bacterial cellulose.22,88,89| Culture source (waste substrate) | BC form/Composite type | Additives/functional agents | Observed performance/key functional properties | Food product tested | Ref. |
|---|---|---|---|---|---|
| G. sacchari | Biobased ternary film | Thermoplastic starch (TPS), gallic acid (GA) | Good oxygen barrier properties (permeability of 0.91 ± 0.12 cm3 µm m−2 day−1 kPa−1); antibacterial activity against S. aureus (reduction of ∼4.5 log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 CFU per mL); delayed browning and weight loss 10 CFU per mL); delayed browning and weight loss |
Fresh-cut apples | 90 |
| K. xylinus ATCC 700178 | In situ composite film | Zinc oxide (ZnO) nanoparticles | Antimicrobial activity against Gram-negative and gram-positive bacteria; effective against E. coli (3 log reduction) and L. monocytogenes (1–3 log reduction) after 24 h | Not specified | 91 |
| Kombucha (SCOBY) | Double-layered composite | Poly(vinyl alcohol)-chitosan (PVA-CH) nanofibers, prodigiosin (bacterial pigment) | Smooth nanofibers (average diameter 139.63 ± 65.52 nm); moderate hydrophilicity; high porosity; antimicrobial activity against S. aureus and P. aeruginosa | Fresh products (general) | 92 |
| A. xylinum | Antimicrobial edible film | Starch, chitosan | Antimicrobial properties | Not specified (general food packaging alternative) | 93 |
| G. hansenii UCP UCP1619 (corn steep liquor) | Blend membrane | Poly(3-hydroxybutyrate) (PHB), clove essential oil (CLO) | Antimicrobial activity | Not specified (active packaging for food) | 94 |
| G. xylinus ATCC 53582 | Composite biofilm | Yeast (Meyerozyma guilliermondii), carboxymethyl cellulose (CMC), glycerol | High water solubility (42.86%); antimicrobial activity against E. coli (16 mm inhibition zone), P. aeruginosa (10 mm), and S. aureus (15 mm); non-toxic | Oranges, tomatoes | 95 |
| BNC films (unspecified source) | Anti-Listeria film | Lactobacillus sakei postbiotics | Anti-Listeria activity; reduced Listeria counts on meat by 2.3–2.6 log CFU per g over 9 days | Buffalo meat patty | 96 |
| BC (Procured) | Intelligent/active film | TEMPO-oxidized BC, thymol (THY), anthocyanin-rich purple potato extract | UV-blocking ability; pH-sensitive color change; extended shelf life | Shrimp | 97 |
| BC (Procured) | Composite biodegradable film | Curcumin, chitosan (CS) | Higher molecular weight CS improved contact angle, mechanical properties, and barrier performance (lower WVTR and OTR); antioxidant properties | Not specified (general food packaging) | 98 |
| A. xylinum LKN6 (sago liquid waste) | Edible modified film | Carboxymethyl cellulose (CMC), glycerol | Optimal mechanical properties with 1% CMC/1% glycerol: tensile strength = 17.47 MPa, elongation at break = 25.60%, Young's modulus = 6.54 GPa; maintained sausage quality for 6 days | Meat sausage | 99 |
| K. xylinus DSM 2325 (cheese whey permeate) | Nanocrystal conjugates on paper coating | Sakacin-A (bacteriocin) | Active conjugates (75 AU per mg); effective in reducing Listeria population on cheese | Fresh soft cheese (“stracchino”) | 100 |
| K. sucrofermentans DSM 15973 | Nanocrystal composite coating | Pullulan, enzymatically hydrolyzed BC nanocrystals (BCNCs) | High oxygen barrier: OTR of ∼0.7 mL m−2·24 h−1 at 0% RH (orders of magnitude lower than PET) | Not specified (high oxygen barrier coating) | 101 |
| K. xylinus | Antimicrobial nanopaper | Lactobacillus plantarum postbiotics | Antimicrobial activity against L. monocytogenes | Ground meat | 83 |
| K. hansenii CGMCC 3917 | Nanofiber coating | HCl hydrolyzed BC nanofibers (BCNs) | Delayed weight loss, improved firmness, reduced browning index | Fresh-cut apples | 102 |
| Gluconobacter xylinum CCM 3611 | Modified hydrogel film | Guar gum, polyvinyl pyrrolidone–carboxymethyl cellulose (PVP-CMC) | Improved elastic and load-bearing capacity; better barrier and hydrophobic properties; 80% biodegradable in 28 days | Fresh berries | 103 |
| K. xylinus ATCC 53,582 | Nanofibrillated edible film/coating | Pectin, fruit purees (mango, guava) | The addition of puree reduced tensile strength (>90%) but increased elongation (∼13 times); replacing pectin with NFBC made films stronger, stiffer, and improved the water vapor barrier | Not specified (sachets, food wrapping/coating) | 104 |
| G. hansenii ATCC 23769 | Infused film | Essential oils (thyme, clove, etc.) | Strong antimicrobial activity against Cronobacter spp.; thyme oil showed the strongest effect (inhibition zone up to 44.37 mm) | Not specified (general active packaging) | 105 |
| K. xylinus | Oil-infused porous film | Oil infusion | Reduced gas permeability, increased stretchability and transparency; 0% moldy rate on strawberries after 5 days at 23 °C (vs. 100% for PE); degraded in soil in 9 days | Strawberries, tomatoes, pork, shrimp | 106 |
| Mixed bacterial culture | Composite material | Sugarcane bagasse (SCB) | Composite with 70% BC/30% SCB had high tensile strength (46.22 MPa) and flexibility (>100 folds); increased hydrophobicity (WCA = 53.22°) | Not specified (sustainable packaging) | 12 |
| K. xylinus and yeast co-culture | Gamma-irradiated film | Clove and rosehip seed oils | Antioxidant and antimicrobial properties | Fresh cheese curd, beef | 107 |
| Unspecified (likely G. xylinus) | Multi-purpose composite film | Waterborne polyurethane (WPU), sodium alginate (SA), zirconium ions (Zr4+) | High tensile strength (82.8 MPa); high transparency (90.01%); low gas permeability; high biodegradability (85.23% weight loss in 49 days); heat sealable | Banana slices, apple slices, apple juice | 108 |
| Waste-derived bacterial cellulose | BC-thermoplastic starch–gallic acid ternary film | Gallic acid (antioxidant) | Improved mechanical strength, enhanced moisture/water resistance, antioxidant activity, UV protection, biodegradability, and extended shelf life | Fresh-cut apple | 109 |
| Kombucha SCOBY (spent coffee grounds-derived) | PLA–MLO film with 5 wt% BC filler | BC from spent coffee grounds (antioxidant) | Increased tensile strength (31.2 MPa), high modulus (1639 MPa), 37% DPPH radical inhibition, and compostability | — | 110 |
| Kombucha production residues (SCOBY waste) | BC composite film modified with BAC50, glycine, CaCl2, cinnamaldehyde | BAC50, glycine, CaCl2 (crosslinker), cinnamaldehyde | +112% modulus, +120% tensile strength, −29% elongation, strong antimicrobial, −37.6% WVT, −22.4% OTR, +25% thermal stability, biodegradable | — | 111 |
| Kombucha BC coated with ZnO nanolayer | BC–ZnO nanocomposite film | ZnO nanolayer (∼45 nm) | Nearly 100% bactericidal against E. coli and C. albicans, inhibited mould on apples 6 days, reduced pore size (491 → 122 nm) | Apples | 21 |
| Fruit processing wastewater fractions | BC film produced using F#6 (permeate <0.5 kDa) supplement | Wastewater nutrients (no post-additives) | Enhanced tensile strength and Young's modulus, improved thermal stability, and water holding capacity | — | 112 |
In recent years, significant progress has been completed in food packaging technologies, with a focus on environmentally friendly alternatives. With the European Union's strategy to ban single-use plastics by 2030, the need for alternative materials has grown due to the environmental problems associated with conventional plastic packaging, particularly microplastic pollution.
In this context, BC, which is a biodegradable and non-toxic material, stands out as a promising alternative. Thanks to its crystalline structure and dense fiber network, BC offers a strong barrier against external factors such as water vapor and oxygen. For example, reductions in oxygen permeability up to 95% and water vapor permeability up to 46% have been observed in starch-based composite films with BC nanowhisker additives.113 In addition, when BC coatings are applied to plastic surfaces such as PET, oxygen barrier performance can be improved by hundreds of times.114 Thanks to these properties, BC stands out as both a sustainable and functional packaging material.
In addition, BC films, which are enriched with functional compounds such as antimicrobial substances and antioxidants, can also be used in active packaging systems. For example, Yordshahi et al.83 Demonstrated antimicrobial activity against Listeria monocytogenes by integrating Lactobacillus plantarum postbiotics into BC films.83 Similarly, Lin et al.115 enriched BC with chitosan and achieved approximately a 2-log colony-forming unit (CFU)/mL inhibition against pathogens such as E. coli and S. aureus.115 Xie et al.84, on the other hand, modified BC films with curcumin; these films showed antioxidant activity and pH sensitivity.116 Sulistyo et al. extended the shelf life of strawberries up to 14 days with BC bioplastics containing fermented soy milk extracts.117 In addition, Yanti et al.99 enriched BC-based edible films with ginger, garlic, and turmeric extracts, providing the films with antimicrobial and antioxidant properties.118 Recent studies exemplify this shift, moving beyond simple substrate substitution to demonstrate fully integrated ‘waste-to-package’ pipelines that valorize multiple waste streams. For instance, El-Gendi et al.119 utilized enzymatically hydrolyzed prickly pear peels as the sole production medium for BC, which achieved an optimized yield of 6.01 g L−1. The resulting BC membrane was then functionalized by impregnation with another byproduct, pomegranate peel extract. This active film demonstrated significant efficacy in strawberry preservation, extending shelf-life by approximately five days, reducing fruit weight loss by 15%, and lowering the final bacterial count from approximately 7.6 log CFU per g in the control group to 2.45 log CFU per g.119 Adopting a comprehensive zero-waste biorefinery model, Yang et al.120 used distillers' grains as a singular feedstock for multiple components. The hydrolyzed supernatant was fermented to produce BC, while the enzymatic residue was simultaneously converted into a photothermal material (FeDL). The final composite film, incorporating both waste-derived components, exhibited enhanced functionality; it increased antioxidant activity by 175.08% and decreased water vapor permeability by 26.81% compared to the base film. In application, it extended the shelf-life of strawberries by approximately 1.75 times under near-infrared irradiation.120 In a complementary approach, Deng et al.121 focused on valorizing waste as a functional additive. An aqueous phase rich in bioactive compounds was extracted from purple passion fruit peel using hydrothermal carbonization. This extract exhibited potent antimicrobial activity, with inhibition zones of 22.50 mm and 23.28 mm against E. coli and S. aureus, respectively. When incorporated into a chitosan/BC nanofiber film, it dramatically enhanced the antioxidant capacity of the film (e.g., increasing DPPH scavenging activity from 7.04% to 96.84%) and effectively delayed the spoilage of blueberries, with packaged fruit showing signs of shrinkage on day 6 (compared to day 4 for the base film). These examples indicate applications in which agro-industrial residues are not merely low-cost inputs but are integral to creating high-value, active packaging systems, which are directly aligned with the principles of a circular bioeconomy.121
However, the high cost of BC production, its low yield, and long fermentation times are the main problems that limit the widespread use of BC on a commercial scale. The fact that the media used in traditional production methods are expensive and the production time of BC usually takes long periods of time, such as 7–10 days, reduces its cost-effectiveness. In order to overcome these problems, the use of low-cost and sustainable carbon sources such as agricultural waste is being investigated. For example, substrates such as fruit juice pulp and waste sugarcane obtained from the food industry offer economical alternatives in BC production. This approach is supported by a growing body of research demonstrating the feasibility of using many types of agro-industrial residues.122 A recent bibliometric analysis by Infante-Neta et al.21 compiled numerous successful examples, highlighting the versatility of this strategy. For instance, exceptional productivity rates have been achieved using soybean hydrolysate with a Kombucha SCOBY culture, yielding up to 13.83 g per L per day. Other significant yields have been reported from banana peel waste (8.83 g L−1), spruce hydrolysate (8.2 g L−1), and cheese whey (6.9 g L−1), utilizing various microorganisms such as Stenotrophomonas sp. and Komagataeibacter species. The successful use of these varied feedstocks, including general kitchen waste, indicates the potential to significantly lower production costs by valorizing otherwise discarded materials.21
These approaches align with the principles of the circular economy, which aims to enhance ecological sustainability and cost-effectiveness by converting waste into valuable resources. Circular economy strategies in BC production offer considerable advantages, particularly by re-evaluating agro-industrial by-products as alternative sources of carbon and nitrogen. The approach involves optimizing production processes in closed loops while converting organic waste into valuable inputs. In this regard, agricultural wastes such as fruit pulp, corn syrup residues, and vegetable peels have been used as carbon sources, leading not only to significant cost reductions but also to improved waste management and environmental benefits123 It has been frequently reported in the literature that bacteria with high cellulose-producing capacities, such as Gluconacetobacter xylinus, can efficiently produce BC using such low-cost or zero-cost substrates.124 While the cost of conventional HS medium ranges between 1 and 10 USD per liter, studies have shown that this cost can be reduced by 50% to 98% when using waste-based alternatives. For instance, a production medium developed from a symbiotic bacteria-yeast consortium obtained from kombucha culture reduced the cost of HS medium from approximately 1.36 USD per L to 0.10 USD per L, achieving a 92.7% reduction. Similarly, Avcıoğlu et al.125 showed that comparable cost savings were achieved using alternative media.126 Furthermore, studies involving industrial by-products such as molasses (a sugar industry by-product), alcoholic beverage waste, and whey demonstrated that medium costs can be reduced by 98%, 94%, or higher amounts compared to the HS medium.127 Additionally, when post-fermentation residues of agricultural waste are utilized, production costs have been reported to drop by up to 90%.
Also, the optimization of biotechnological processes by genetic engineering (e.g., the use of high-yielding Komagataeibacter strains or acceleration of production by enzymatic means) aims to increase production efficiency. In addition, the integration of nanotechnology can significantly improve both the mechanical strength and gas barrier properties via the addition of additives (e.g., nano-clay, silver nanoparticles, or cellulose nanocrystals) to BC.128
As a result, recent studies have demonstrated not only that circular economy principles can be effectively integrated into BC production through the use of waste-based substrates, leading to a significant cost reduction, but also that BC represents a sustainable and functional alternative to conventional packaging materials, with the potential to meet both the environmental and performance requirements in the food packaging industry.
8. Regulatory compliance and industrial readiness of BC packaging
From a regulatory standpoint, the commercialization of BC-based food packaging must align with established food contact material (FCM) legislation. In the European Union, materials intended for food contact are governed by Regulation (EC) No 1935/2004, which requires that packaging materials do not transfer substances to food in amounts that could endanger human health or compromise food quality. For polymeric materials and composite systems, Regulation (EU) No 10/2011 provides an overall migration limit (OML) of 10 mg dm−2 and establishes specific migration limits (SMLs) for authorized additives. Furthermore, when BC is used in active or intelligent packaging applications, additional compliance with Regulation (EC) No 450/2009 becomes necessary, particularly if functional components are designed to interact with the food environment. Although cellulose as a chemical entity is well established in food-related applications, microbial-derived BC must demonstrate adequate purity, controlled processing, and full migration compliance prior to large-scale industrial use.In the United States, BC-based materials are regulated under the FDA Food Contact Substance (FCS) notification framework. While cellulose is generally recognized as safe (GRAS) for certain applications, microbial cellulose must be evaluated according to its specific conditions of use, including temperature, food type (aqueous, acidic, or fatty), and contact duration. Particular attention is required to ensure the effective removal of residual alkali from purification steps, as well as the absence of fermentation-derived impurities or endotoxins. Compliance with Good Manufacturing Practices (GMP) is also essential. Consequently, successful industrial translation of BC packaging depends not only on material performance but also on standardized migration testing, toxicological evaluation, and comprehensive regulatory documentation.
From a market perspective, commercially available biodegradable polymers (e.g., polylactic acid (PLA) and polyhydroxyalkanoates (PHA)) have already reached industrial-scale production and are widely used in thermoformed trays, films, and molded packaging components. PLA, produced from lactic acid fermentation, is extensively employed in industrially compostable packaging systems and represents a commercially mature technology. PHA materials, also fermentation-derived, offer enhanced biodegradability and, in certain formulations, compatibility with home composting conditions, although they often remain cost-intensive. Compared to these materials, BC exhibits excellent oxygen barrier performance under dry conditions due to its highly crystalline nanofibrillar network. However, its inherent hydrophilicity limits resistance to water vapor unless structural modification or multilayer composite design is applied. In addition, BC production is typically batch-based and relies on surface-mediated or bioreactor fermentation, which presents scale-up and continuous processing challenges not encountered in conventional thermoplastic conversion systems.
Despite promising laboratory results and growing interest in active packaging applications, most BC-based food packaging systems remain at intermediate Technology Readiness Levels (TRL 4–6),129 corresponding to laboratory validation and limited pilot-scale demonstration under the European Commission TRL framework. Several technical barriers continue to hinder advancement toward higher industrial readiness levels, including low volumetric productivity in static cultures, oxygen transfer limitations during scale-up, energy-intensive purification and drying steps, the lack of continuous roll-to-roll film production technologies, and insufficient regulatory validation datasets. In contrast, PLA and PHA packaging systems are widely regarded as fully commercialized technologies (TRL 8–9),130,131 reflecting established industrial manufacturing and operational deployment.
It is important to note that regulatory compliance and industrial readiness are closely interconnected. Progression from laboratory prototypes to commercially viable BC packaging solutions requires not only improved fermentation efficiency and reduced energy consumption but also the generation of robust migration data, toxicological validation, and food-contact compliance documentation. Future research should therefore adopt an integrated approach that simultaneously addresses material optimization, regulatory pathway planning, and process intensification to accelerate the responsible commercialization of BC-based food packaging systems.
9. Challenges and limitations
The production of bacterial cellulose from agro-forest residues offers significant potential as a sustainable approach, yet it faces various challenges and limitations. One primary issue is the chemical composition of these residues, which can vary greatly depending on their source, seasonal changes, and processing methods. This chemical variability can lead to inconsistencies in the quality and yield of BC, making the production process less stable.132,133 Furthermore, environmental parameters such as humidity, pH, and temperature have a direct influence on the size and quality of BC pellicles, which lowers the production efficiency.134 Another limiting factor is the high moisture content typically found in agro-forest residues, which complicates transportation and increases the costs associated with drying and storage. The pretreatment processes required to convert these residues into viable carbon sources for BC production are often complex and costly, involving mechanical, chemical, or biochemical methods. Optimizing these processes is a key consideration for improving efficiency for use in large-scale production. Moreover, maintaining the precise conditions needed for the bacterial cultures to thrive adds to the overall production costs, posing a barrier to commercialization. Finally, improper management of agro-forest residues can have environmental repercussions, since practices such as open burning or inadequate disposal may release harmful pollutants into the atmosphere. These challenges collectively limit the sustainable and economically viable production of BC from agro-forest residues.132–13410. Future directions and LCA for BC production and application
The future of bacterial cellulose production from agro-forest and industrial waste represents an intersection of resource efficiency, bioeconomy, and industrial innovation. This approach not only addresses environmental challenges posed by waste accumulation but also promises to reduce production costs, enhance scalability, and open up new applications for BC in sectors such as biomedical engineering, packaging, and environmental management. To fully appreciate the future potential of this field, it is important to consider the economic, technical, and environmental dimensions of waste-based BC production, including identifying the challenges and pathways for optimization. The production of BC from agro-forest and industrial waste has offered the food-packaging sector several opportunities. Sustainable and biodegradable packaging solutions are in high demand as concerns about plastic pollution worldwide grow. Due to its special qualities, BC has presented a strong substitute for traditional plastics. The production of BC from waste may transform food packaging, which can support the objectives of resource efficiency and recyclability targets.135To fully realize the potential of BC produced from agro-forest and industrial waste for food packaging, several strategies, including technological and industrial strategies, need to be pursued. These strategies should focus on improving production efficiency, enhancing material properties, optimizing waste utilization, and scaling up the technology to meet the growing demand for sustainable packaging.
Life Cycle Assessment (LCA) has become an important methodology for assessing the environmental impacts of product systems from a cradle-to-grave perspective. This approach encompasses all stages of the product lifecycle, including raw material extraction, manufacturing steps, distribution steps, usage, and disposal. By quantifying impacts that include energy consumption, greenhouse gas emissions, and resource depletion, the LCA provides critical insights that guide sustainable development. Particularly, the integration of LCA into early-stage research and development has proven to be a key consideration for guiding innovations towards environmentally conscious solutions. For example, LCA is widely used in evaluating the environmental sustainability of biopolymers such as BC, offering a detailed framework to compare production routes and identify resource-efficient processes.129,136
The production of bacterial cellulose is a compelling case study of the utility of LCA in advancing sustainable materials. BC, a nanostructured biopolymer produced via microbial fermentation, has several applications ranging from biomedicine to food packaging. Studies employing LCA have demonstrated the environmental benefits of utilizing alternative low-cost substrates such as hydrolyzed soybean molasses, diluted molasses, and agricultural residues. Compared to conventional synthetic media, these substrates significantly reduce impacts related to climate change, freshwater eutrophication, and acidification.136–138 For instance, substituting synthetic media for BC production with agro-industrial residues not only lowers environmental burdens but also addresses waste valorization, a key element of the circular economy. These findings indicate the capability of the LCA to integrate environmental considerations into decision-making at the laboratory scale, enabling the selection of optimal production routes for scaling.129,138
LCA also plays an important role in optimizing production processes by identifying critical hotspots that contribute disproportionately to environmental impacts. For BC production, purification steps and energy-intensive drying processes have been identified as important phases requiring optimization.129,136 By modifying inputs, such as replacing chemical reagents or integrating energy-efficient drying techniques, significant reductions in water scarcity impacts and fossil fuel use have been achieved. Beyond the process level, LCA facilitates ecodesign strategies that align product development toward measurable environmental performance indicators. For instance, incorporating LCA during the Technology Readiness Level (TRL) stages allows researchers to anticipate potential environmental impacts as a technology moves from laboratory to industrial scales. This approach ensures that environmental performance remains a priority throughout the innovation cycle.136,138,139
Energy consumption during manufacturing processes is a critical factor determining the overall environmental sustainability of waste-derived biopolymers and cellulose-based packaging. Traditional fiber and film manufacturing methods often rely on high-voltage electric fields and harsh chemicals, leading to high energy demands and a consequent increase in the carbon footprint. To mitigate this cumulative environmental impact, innovative low-energy and high-efficiency manufacturing techniques have emerged, such as nozzle-pressurized spinning (NPS). By combining centrifugal force with gas pressure, NPS completely eliminates the need for high-voltage electricity, significantly reducing energy consumption while enabling the high-throughput continuous manufacturing of cellulose fibers and films from unconventional agricultural wastes, such as cow manure.140 Transitioning to such straightforward, mechanically driven processes aligns the manufacturing stage with circular economy requirements, ensuring that the ecological benefits of using waste feedstocks are not offset by energy-intensive processing.
To further reduce energy demand and improve process efficiency, recent studies have demonstrated the feasibility of off-grid, battery-powered manufacturing systems for cellulose-based materials. Decoupling the production process from a continuous reliance on the power grid allows manufacturers to implement emissions-responsive charging strategies, such as storing energy from renewable sources during off-peak hours when grid carbon intensity is lowest. Furthermore, optimizing the power load in these battery-powered systems ensures that motors operate within their peak efficiency ranges (e.g., 50–100% of rated load), effectively minimizing the energy dissipated as waste heat that is typically observed in mains-powered systems.141 Integrating such highly efficient, decentralized, and low-energy manufacturing methodologies with waste-derived bacterial cellulose will be an important consideration in dramatically reducing the overall Life Cycle Assessment (LCA) impact and production costs of sustainable food packaging.
Emerging advances in LCA methodologies (e.g., attributional and consequential LCA models) further extend the scope of environmental assessments. These models enable scenario-based analyses that examine the implications of alternative inputs, production methods, and end-of-life treatments.137,138 For example, comparative studies have explored the feasibility of integrating carbon-neutral energy sources or developing circular pathways for by-products in bacterial cellulose production. Additionally, probabilistic LCA frameworks can account for uncertainties in data collection, providing more robust and reliable impact assessments. These approaches support the relevance and actionability of the LCA outputs, particularly in sectors such as biopolymer development and renewable energy technologies.129,138
Despite the benefits of LCA, the application of LCA at early research stages faces challenges related to data availability, scalability, and methodological standardization. Many laboratory-scale processes lack comprehensive data on energy use, material flows, and emissions, which complicates the accuracy of an LCA assessment.129,136 Efforts to integrate advanced data collection tools (e.g., process simulation software and real-time monitoring systems) can support more reliable assessments. Future research should also focus on harmonizing LCA methodologies to ensure consistency across studies, particularly for emerging technologies involving biotechnology and nanomaterials.137,139
The LCA of BC production, conducted via a cradle-to-gate approach, reveals several important insights. The production process consumes significant resources, predominantly water (36.1 tons per kg of BC), yet 98% is recoverable post-treatment. Environmental hotspots include raw material production, particularly sodium dihydrogen phosphate, contributing to human toxicity and terrestrial ecotoxicity impacts. Despite the relatively low direct environmental burden associated with BC production, advancements in fermentation efficiency and resource use optimization are important considerations.129 Future efforts can consider eco-efficient culture media and energy reduction strategies, supporting the viability of BC as a green alternative to established processes for nanocellulose production from traditional cellulose sources.
Optimization of the substrate pretreatment and engineering studies in this field is necessary to enhance the ability of BC-producing bacteria to efficiently utilize complex sugars from waste. Process optimization, including fermentation and bioreactor scaling, must be refined to maximize BC yield and reduce costs. Functionalizing BC with antimicrobial agents and reinforcing BC via nanomaterials can improve the performance of the material in food packaging. The standardization of waste feedstocks and quality control systems is important for consistent production, while LCAs will ensure sustainability.
Although significant efforts have been made to develop the industrial production of BC, a deep understanding of the environmental impacts related to these relevant processes is still required. Very few studies were dedicated to LCAs for nanocellulose production in general and BC production in particular. In this regard, two papers have described the implementation of an ecodesign for bacterial cellulose production at a laboratory scale.136 The authors, by means of this approach based on LCA and process modeling, evaluated alternative routes for BC production. Forte et al.129 conducted an investigation on a production process design of BC under static culture conditions, such as wastewater treatment, via a cradle-to-grave approach.129 Their conclusion was that the BC production process was associated with a small contribution related to resource consumption as well as environmental impact on the global life cycle.
LCA is a useful tool for advancing sustainable innovations across industries. By providing a detailed understanding of environmental impacts, LCA informs process optimization, resource efficiency, and product design. In the context of bacterial cellulose production, LCA has demonstrated its potential to reduce environmental burdens while supporting the transition to bio-based and low-impact materials. As LCA methodologies continue to evolve, their integration into research and development processes will be critical for quantifying environmental impact profiles and guiding evidence-based material optimization.129,136,138 Furthermore, public awareness, policy incentives, and collaboration between academia and industry are important for driving innovation and adoption. These steps will allow waste-derived BC to play an important role in sustainable packaging, offering a cost-effective, biodegradable alternative to plastics while promoting circular economy practices.
11. Conclusions
The production of BC through fermentation processes, including static and agitated methods, represents a promising pathway for sustainable material development, particularly in food packaging applications. Static cultures, while simple and capable of producing BC with desirable mechanical properties, face challenges such as low yield and prolonged incubation times, which hinder large-scale production. In contrast, agitated fermentation enhances productivity and yields BC with distinct physical characteristics, although it often results in BC with lower crystallinity and mechanical strength. Advances in optimizing fermentation conditions, including substrate selection and agitation techniques, have significantly improved BC yield and functionality.The use of agro-forest wastes such as agricultural residues, peels, wood residues, and leaf litter as carbon sources for BC production not only reduces environmental impact by repurposing waste but also mitigates the high production costs associated with traditional media. BC derived from these sources demonstrates high crystallinity, mechanical strength, and biocompatibility, making it suitable for a range of applications, including food packaging. Additionally, the enhanced water retention capacity and antimicrobial properties observed in BC produced from fruit and vegetable peels highlight its promise for advanced packaging applications. Moreover, while many types of agro-forest wastes provide valuable fermentable carbon substrates to produce BC, recent advances also indicate the extraction of plant-derived microcrystalline cellulose (MCC) from residues such as durian rind. It is critical to distinguish that this extracted MCC is a final cellulose product rather than a fermentation feedstock; however, it can be effectively utilized as a reinforcing phase in BC-based biocomposites, significantly enhancing the mechanical properties and biodegradability of the final material.142
The potential of BC as a biodegradable alternative to synthetic polymers in food packaging is particularly noteworthy. Its natural structure, combined with its ability to incorporate functional additives such as antimicrobial agents, addresses the increasing demand for eco-friendly packaging solutions. However, consistent BC production from agro-forest residues presents challenges such as substrate variability and process costs. Despite these obstacles, ongoing research into substrate optimization, nanotechnology integration, and bioreactor scaling is an important consideration for overcoming these limitations and fully realizing the potential of BC in industrial applications.
Areas of future research include optimizing production processes, scaling up production, and exploring novel applications, particularly in the food packaging sector. Additional efforts can further optimize fermentation processes, including substrate selection, pretreatment strategies, and agitation methods, to enhance yield and functionality. Integrating these technical improvements with comprehensive LCA will be essential to evaluate environmental performance, compare different BC production routes, and identify the most sustainable and cost-efficient pathways. In addition, studies on the enzymatic degradation and pilot-scale composting of cellulose-based films highlight the importance of integrating end-of-life scenarios into LCA assessments, ensuring that material sustainability is evaluated across the entire lifecycle.143 Addressing regulatory aspects and assessing long-term stability will further support the successful integration of BC-based materials into commercial applications.
Building on these observations, techno-economic studies now estimate that replacing refined sugars in the Hestrin–Schramm medium with the agro-industrial residues mentioned above can reduce factory-gate costs from roughly USD 4–6 kg−1 to USD 2–3 kg−1, an important milestone for commercial feasibility. These cost reductions coincide with, and are incentivized by, the Packaging and Packaging Waste Regulation (PPWR) of the European Union, which aims for all packaging in the EU market to be recyclable in an economically viable manner by 2030, while mandating compostability only for certain specific categories (e.g., tea bags, coffee capsules); furthermore, this regulation progressively restricts specific single-use plastic formats rather than imposing a wholesale ban. BC films derived from waste streams therefore align not only with circular-economy principles but also with evolving legislative drivers that favor biobased and compostable materials.
To capitalize on this alignment, future work should couple process optimization with comprehensive LCA and migration testing under EU food contact regulations. Demonstrating a positive economic and environmental balance sheet alongside robust barrier performance will be an important consideration for industry uptake. Successful efforts involving these activities would position waste-derived BC as a cornerstone material in post-2030 sustainable packaging, biomedical, and high-value textile markets, fulfilling both market demand and regulatory ambitions for a low-carbon, circular economy. Moreover, these trends are reinforced by broader consumer-driven and policy-driven pressures toward sustainable polymers, which are accelerating the transition from fossil-based plastics to biobased alternatives.144
The scarcity of studies on the application of the LCA approach represents an important trend to explore in order to evaluate the circular economy and environmental cost of BC production routes. LCA will be of great importance to investigate:
• A comparative study between the different processes for BC production.
• The experimental parameters and their effect on LCA.
• The carbon source effect.
Author contributions
Tubanur Avci: writing – original draft, investigation, formal analysis, data curation. Aslıhan Çalhan: writing – original draft, investigation, formal analysis. Data curation. Eda Nur Ayar: writing – review & editing. Canan Dogan: methodology, conceptualization, investigation, writing – review & editing, data curation, supervision. Oguzhan Gunduz: methodology, conceptualization, investigation, writing – review & editing, data curation, supervision. Hamid Kaddami: methodology, conceptualization, investigation, writing – review & editing, data curation, supervision. Roger Narayan: writing – review & editing.Conflicts of interest
The authors declare no competing financial or personal interests that could have influenced the work reported in this paper.Data availability
No primary research results and no new data were generated or analysed as part of this review paper.References
- P. K. Gupta, S. S. Raghunath, D. V. Prasanna, P. Venkat, V. Shree, C. Chithananthan, S. Choudhary, K. Surender and K. Geetha, An update on overview of cellulose, its structure and, Cellulose, 2019, 59–80 Search PubMed.
- V. D. Girard, J. Chaussé and P. Vermette, Bacterial cellulose: A comprehensive review, J. Appl. Polym. Sci., 2024, 141(15), e55163 CrossRef CAS.
- H. M. Azeredo, H. Barud, C. S. Farinas, V. M. Vasconcellos and A. M. Claro, Bacterial cellulose as a raw material for food and food packaging applications, Front. Sustainable Food Syst., 2019, 3, 7 CrossRef.
- M. Katyal, R. Singh, R. Mahajan, A. Sharma, R. Gupta, N. K. Aggarwal and A. Yadav, Bacterial cellulose: Nature's greener tool for industries, Biotechnol. Appl. Biochem., 2023, 70(5), 1629–1640 CrossRef CAS PubMed.
- Z. Hussain, W. Sajjad, T. Khan and F. Wahid, Production of bacterial cellulose from industrial wastes: a review, Cellulose, 2019, 26(5), 2895–2911 CrossRef CAS.
- R. Singh, M. Katyal, R. Mahajan, R. Gupta, N. K. Aggarwal and A. Yadav, Utilizing Biowaste-Soil Consortia for Efficient Isolation of Cellulose-Producing Bacteria and Evaluation of Bacterial Cellulose, Biotechnol. Appl. Biochem., 2026 DOI:10.1002/bab.70033.
- R. Singh, M. Katyal, R. Mahajan, R. Gupta, N. K. Aggarwal and A. Yadav, Valorization of Pomace Waste for the Production of Cellulose by Komagataeibacter diospyri RSA4, Biotechnol. Appl. Biochem., 2025, 72(6), 1924–1935 CrossRef CAS PubMed.
- M. Katyal, R. Singh, R. Mahajan, A. Sharma, R. Gupta, N. K. Aggarwal and A. Yadav, A novel cost-effective methodology for the screening of nanocellulose producing micro-organisms, Bioprocess Biosyst. Eng., 2024, 47(9), 1595–1603 CrossRef CAS PubMed.
- M. Katyal, R. Singh, R. Mahajan, A. Sharma, R. Gupta, N. K. Aggarwal and A. Yadav, Valorization of papaya fruit peel waste for the production of nanocellulose by Novacetimonas hansenii BMK-3, Biotechnol. Appl. Biochem., 2025, 72(4), 1069–1078 CrossRef CAS PubMed.
- M. N. Sayan, A. Avci, R. B. Sulutas, A. Calhan, S. Cesur, A. Sahin, O. Gunduz and C. B. Ustundag, Production, characterization, and biocompatibility of polycaprolactone/polylactic acid coaxial nanofiber patches based on bacterial cellulose from orange peels, Emergent Mater., 2025, 8, 1–9 Search PubMed.
- M. Güzel and Ö. Akpınar, Production and characterization of bacterial cellulose from citrus peels, Waste Biomass Valorization, 2019, 10(8), 2165–2175 CrossRef.
- C. J. da Silva Junior, A. D. de Medeiros, A. K. Cavalcanti, J. D. De Amorim, I. J. Durval, Y. D. Cavalcanti, A. Converti, A. F. Costa and L. A. Sarubbo, Towards sustainable packaging using microbial cellulose and sugarcane (Saccharum officinarum L.) bagasse, Materials, 2024, 17(15), 3732 CrossRef CAS PubMed.
- M. T. Luo, C. Zhao, C. Huang, X. F. Chen, Q. L. Huang, G. X. Qi, L. L. Tian, L. Xiong, H. L. Li and X. D. Chen, Efficient using durian shell hydrolysate as low-cost substrate for bacterial cellulose production by Gluconacetobacter xylinus, Indian J. Microbiol., 2017, 57(4), 393–399 CrossRef CAS PubMed.
- M. U. Rani and K. A. Appaiah, Production of bacterial cellulose by Gluconacetobacter hansenii UAC09 using coffee cherry husk, J. Food Sci. Technol., 2013, 50(4), 755–762 CrossRef CAS PubMed.
- B. Anwar, B. Bundjali, Y. Sunarya and I. M. Arcana, Properties of bacterial cellulose and its nanocrystalline obtained from pineapple peel waste juice, Fibers Polym., 2021, 22(5), 1228–1236 CrossRef CAS.
- M. Ul-Islam, M. W. Ullah, S. Khan and J. K. Park, Production of bacterial cellulose from alternative cheap and waste resources: A step for cost reduction with positive environmental aspects, Korean J. Chem. Eng., 2020, 37(6), 925–937 CrossRef CAS.
- M. J. Quintero-Silva, S. J. Suárez-Rodríguez, M. A. Gamboa-Suárez, C. Blanco-Tirado and M. Y. Combariza, Polyhydroxyalkanoates production from cacao fruit liquid residues using a native Bacillus megaterium strain: Preliminary study, J. Polym. Environ., 2024, 32(3), 1289–1303 CrossRef CAS.
- D. M. Panaitescu, M. Oprea, A. N. Frone, B. Trică, I. Popa-Tudor, M. Ghiurea, C. A. Nicolae, A. R. Gabor, G. M. Oprică, C. D. Uşurelu and C. M. Damian, Valorization of spent lignocellulosic substrate of edible mushrooms into cellulose nanofibers for bionanocomposites production, J. Polym. Environ., 2024, 32(12), 6618–6635 CrossRef CAS.
- V. Cebrián-Lloret, A. Martínez-Abad, A. López-Rubio and M. Martínez-Sanz, Sustainable bio-based materials from minimally processed red seaweeds: effect of composition and cell wall structure, J. Polym. Environ., 2023, 31(3), 886–899 CrossRef.
- Y. Dai, D. Sun, S. Sundaram, A. Delbusso, D. O'Rourke, M. Dorris and M. Edirisinghe, Facile synthesis: from Laminaria hyperborea to cellulose films and fibers, Cellulose, 2024, 31(1), 205–216 CrossRef CAS.
- A. A. Infante-Neta, A. P. D'Almeida and T. L. Albuquerque, Bacterial cellulose in food packaging: a bibliometric analysis and review of sustainable innovations and prospects, Processes, 2024, 12(9), 1975 CrossRef CAS.
- L. Urbina, M. Á. Corcuera, N. Gabilondo, A. Eceiza and A. Retegi, A review of bacterial cellulose: sustainable production from agricultural waste and applications in various fields, Cellulose, 2021, 28(13), 8229–8253 CrossRef CAS.
- A. Amr and H. Ibrahim, Bacterial cellulose: biosynthesis and applications, in Next-Generation Textiles, IntechOpen, 2022 Search PubMed.
- S. Mishra, P. K. Singh, R. Pattnaik, S. Kumar, S. K. Ojha, H. Srichandan, P. K. Parhi, R. K. Jyothi and P. K. Sarangi, Biochemistry, synthesis, and applications of bacterial cellulose: a review, Front. Bioeng. Biotechnol., 2022, 10, 780409 CrossRef PubMed.
- M. A. Cruz, O. Flor-Unda, A. Avila, M. D. Garcia and L. Cerda-Mejía, Advances in bacterial cellulose production: a scoping review, Coatings, 2024, 14(11), 1401 CrossRef CAS.
- Z. Cheng, R. Yang, X. Liu, X. Liu and H. Chen, Green synthesis of bacterial cellulose via acetic acid pre-hydrolysis liquor of agricultural corn stalk used as carbon source, Bioresour. Technol., 2017, 234, 8–14 CrossRef CAS PubMed.
- F. D. Goelzer, P. C. Faria-Tischer, J. C. Vitorino, M. R. Sierakowski and C. A. Tischer, Production and characterization of nanospheres of bacterial cellulose from Acetobacter xylinum from processed rice bark, Mater. Sci. Eng., C, 2009, 29(2), 546–551 CrossRef CAS.
- L. Chen, F. Hong, X. X. Yang and S. F. Han, Biotransformation of wheat straw to bacterial cellulose and its mechanism, Bioresour. Technol., 2013, 135, 464–468 CrossRef CAS PubMed.
- F. Hong, Y. X. Zhu, G. Yang and X. X. Yang, Wheat straw acid hydrolysate as a potential cost-effective feedstock for production of bacterial cellulose, J. Chem. Technol. Biotechnol., 2011, 86(5), 675–680 CrossRef CAS.
- W. Al-Abdallah and Y. Dahman, Production of green biocellulose nanofibers by Gluconacetobacter xylinus through utilizing the renewable resources of agriculture residues, Bioprocess Biosyst. Eng., 2013, 36(11), 1735–1743 CrossRef CAS PubMed.
- M. Ramos, E. Laveriano, L. San Sebastián, M. Perez, A. Jiménez, R. M. Lamuela-Raventos, M. C. Garrigós and A. Vallverdú-Queralt, Rice straw as a valuable source of cellulose and polyphenols: Applications in the food industry, Trends Food Sci. Technol., 2023, 131, 14–27 CrossRef CAS.
- Y. Photphisutthiphong and S. Vatanyoopaisarn, The production of bacterial cellulose from organic low-grade rice, Curr. Res. Nutr. Food Sci., 2020, 8(1), 206–216 CrossRef.
- F. D. Goelzer, P. C. Faria-Tischer, J. C. Vitorino, M. R. Sierakowski and C. A. Tischer, Production and characterization of nanospheres of bacterial cellulose from Acetobacter xylinum from processed rice bark, Mater. Sci. Eng., C, 2009, 29(2), 546–551 CrossRef CAS.
- P. Sudying, N. Laingaumnuay and P. Jaturapiree, Production and characterization of bacterial cellulose from rice washing drainage (RWD) by Komagataeibacter nataicola Li1, in Key Eng. Mater, Trans Tech Publications Ltd, 2019, vol. 824, pp. 30–37 Search PubMed.
- M. S. Hasanin, M. Abdelraof, A. H. Hashem and H. El Saied, Sustainable bacterial cellulose production by Achromobacter using mango peel waste, Microb. Cell Fact., 2023, 22(1), 24 CrossRef CAS PubMed.
- L. Ogrizek, J. Lamovšek, F. Čuš, M. Leskovšek and M. Gorjanc, Properties of bacterial cellulose produced using white and red grape bagasse as a nutrient source, Processes, 2021, 9(7), 1088 CrossRef CAS.
- A. B. Adnan, Production of Bacterial Cellulose Using Low-Cost Media, Doctoral dissertation, University of Waikato, 2015.
- C. H. Kuo, C. Y. Huang, C. J. Shieh, H. M. Wang and C. Y. Tseng, Hydrolysis of orange peel with cellulase and pectinase to produce bacterial cellulose using Gluconacetobacter xylinus, Waste Biomass Valorization, 2019, 10(1), 85–93 CrossRef CAS.
- M. Kumar, V. Kumar and S. Saran, Current research and advances in the molecular aspect of bacterial cellulose synthesis with special emphasis on the production of bacterial cellulose from waste and its food applications, Cellulose, 2024, 31(6), 3323–3351 CrossRef CAS.
- A. Patel, P. Patel, M. Parmar and H. Gosai, Employing RSM and ANN-based applications for modelling enhanced bacterial cellulose production from pineapple peel waste using Komagateibacter saccharivorans APPK1, Chem. Eng. J., 2024, 480, 148057 CrossRef CAS.
- P. Singhaboot and P. Kroeksakul, High Performance of Bacterial Strain Isolated from Bio-Extract for Cellulose Production, Pertanika J. Trop. Agric. Sci., 2022, 45(4), 1161–1175 Search PubMed.
- E. E. Kiziltas, A. Kiziltas and D. J. Gardner, Synthesis of bacterial cellulose using hot water extracted wood sugars, Carbohydr. Polym., 2015, 124, 131–138 CrossRef PubMed.
- D. Kadimaliev, E. Kezina, V. Telyatnik, V. Revin, O. Parchaykina and I. Syusin, Residual Brewer’s yeast biomass and bacterial cellulose as an alternative to toxic phenol-formaldehyde binders in production of pressed materials from waste wood, BioResources, 2015, 10(1), 1644–1656 CrossRef.
- M. Güzel and Ö. Akpınar, Production and characterization of bacterial cellulose from citrus peels, Waste Biomass Valorization, 2019, 10(8), 2165–2175 CrossRef.
- I. Algar, S. C. Fernandes, G. Mondragon, C. Castro, C. Garcia-Astrain, N. Gabilondo, A. Retegi and A. Eceiza, Pineapple agroindustrial residues for the production of high value bacterial cellulose with different morphologies, J. Appl. Polym. Sci., 2015, 132, 41237 CrossRef.
- G. Pacheco, C. R. Nogueira, A. B. Meneguin, E. Trovatti, M. C. Silva, R. T. Machado, S. J. Ribeiro, E. C. da Silva Filho and H. D. Barud, Development and characterization of bacterial cellulose produced by cashew tree residues as alternative carbon source, Ind. Crops Prod., 2017, 107, 13–19 CrossRef CAS.
- S. Lotfiman, D. R. Awang Biak, T. B. Ti, S. Kamarudin and S. Nikbin, Influence of date syrup as a carbon source on bacterial cellulose production by Acetobacter xylinum 0416, Adv. Polym. Technol., 2018, 37(4), 1085–1091 CrossRef CAS.
- B. A. Tayo, M. Akıntunde and S. O. Alao, Comparative effect of agrowastes on bacterial cellulose production by Acinetobacter sp. BAN1 and Acetobacter pasteurianus PW1, Turk. J. Agric. Nat. Sci., 2017, 4(2), 145–154 Search PubMed.
- C. Castro, R. Zuluaga, J. L. Putaux, G. Caro, I. Mondragon and P. Gañán, Structural characterization of bacterial cellulose produced by Gluconacetobacter swingsii sp. from Colombian agroindustrial wastes, Carbohydr. Polym., 2011, 84(1), 96–102 CrossRef CAS.
- J. V. Kumbhar, J. M. Rajwade and K. M. Paknikar, Fruit peels support higher yield and superior quality bacterial cellulose production, Appl. Microbiol. Biotechnol., 2015, 99(16), 6677–6691 CrossRef CAS PubMed.
- L. F. Amorim, L. Li, A. P. Gomes, R. Fangueiro and I. C. Gouveia, Sustainable bacterial cellulose production by low cost feedstock: Evaluation of apple and tea by-products as alternative sources of nutrients, Cellulose, 2023, 30(9), 5589–5606 CrossRef CAS.
- G. Bozdağ, O. Pinar, O. Gündüz and D. Kazan, Valorization of pea pod, celery root peel, and mixed-vegetable peel as a feedstock for biocellulose production from Komagataeibacter hansenii DSM 5602, Biomass Convers. Biorefin., 2023, 13(9), 7875–7886 CrossRef.
- Y. F. Cavalcanti, J. D. Amorim, A. D. Medeiros, C. J. da Silva Jr, I. J. Durval, A. F. Costa and L. A. Sarubbo, Microbial cellulose production with tomato (solanum Lycopersicum) residue for industrial applications, Chem. Eng. Trans., 2023, 100, 409–414 Search PubMed.
- S. Gorgieva, U. Jančič, E. Cepec and J. Trček, Production efficiency and properties of bacterial cellulose membranes in a novel grape pomace hydrolysate by Komagataeibacter melomenusus AV436T and Komagataeibacter xylinus LMG 1518, Int. J. Biol. Macromol., 2023, 244, 125368 CrossRef CAS PubMed.
- M. O. Akintunde, B. C. Adebayo-Tayo, M. M. Ishola, A. Zamani and I. S. Horváth, Bacterial cellulose production from agricultural residues by two Komagataeibacter sp. strains, Bioengineered, 2022, 13(4), 10010–10025 CrossRef CAS PubMed.
- Z. A. Zaki, F. Francis and N. A. Zaini, Bacterial cellulose production from oil palm empty fruit bunch (OPEFB) hydrolysate using Komagataeibacter xylinus strain, Biomass Convers. Biorefin., 2023, 13(16), 14629–14640 CrossRef CAS.
- X. Ma, H. Yuan, H. Wang and H. Yu, Coproduction of bacterial cellulose and pear vinegar by fermentation of pear peel and pomace, Bioprocess Biosyst. Eng., 2021, 44(11), 2231–2244 CrossRef CAS PubMed.
- A. Kurosumi, C. Sasaki, Y. Yamashita and Y. Nakamura, Utilization of various fruit juices as carbon source for production of bacterial cellulose by Acetobacter xylinum NBRC 13693, Carbohydr. Polym., 2009, 76(2), 333–335 CrossRef CAS.
- C. Quiñones-Cerna, J. C. Rodríguez-Soto, G. Barraza-Jáuregui, J. Huanes-Carranza, J. A. Cruz-Monzón, W. Ugarte-López, F. Hurtado-Butrón, F. Samanamud-Moreno, D. Haro-Carranza, S. Valdivieso-Moreno and D. Salirrosas-Fernández, Bioconversion of agroindustrial asparagus waste into bacterial cellulose by Komagataeibacter rhaeticus, Sustainability, 2024, 16(2), 736 Search PubMed.
- S. Henry, S. Dhital, H. Sumer and V. Butardo Jr, Solid-state fermentation of cereal waste improves the bioavailability and yield of bacterial cellulose production by a Novacetimonas sp. isolate, Foods, 2024, 13(19), 3052 Search PubMed.
- Z. Liu, F. Siddique, Y. Wei, M. A. Haque, L. Na, X. Yang and C. S. Lin, Efficient production of bacterial cellulose using komagataeibacter sucrofermentans on sustainable feedstocks, ChemSusChem, 2025, 18(6), e202401578 CrossRef CAS PubMed.
- R. Albanez, A. P. Paulinetti, I. M. Augusto, G. Lovato, J. A. Rodrigues and S. M. Ratusznei, Integrated production of bacterial cellulose and biogas from agro-industrial wastewater: technical feasibility of implementing a biorefinery concept, Braz. J. Chem. Eng., 2025, 42(2), 505–516 CrossRef CAS.
- M. A. Páez, M. Casa-Villegas, M. Aldas, M. Luna, D. Cabrera-Valle, O. López, D. Fernández, M. A. Cruz, O. Flor-Unda, M. D. García and L. Cerda-Mejía, Insights into agitated bacterial cellulose production with microbial consortia and agro-industrial wastes, Fermentation, 2024, 10(8), 425 CrossRef.
- F. Lasagni, S. Cassanelli and M. Gullo, How carbon sources drive cellulose synthesis in two Komagataeibacter xylinus strains, Sci. Rep., 2024, 14(1), 20494 Search PubMed.
- J. C. Martinez, L. Davila, R. Zuluaga, A. Ricardo, J. Figueroa, M. G. de Almeida Oliveira and Y. Meriño-Cabrera, Production of bacterial cellulose from banana waste (Musa paradisiaca): A sustainable material for making cardboard paper, Biocatal. Agric. Biotechnol., 2025, 63, 103484 Search PubMed.
- A. Fatima, M. Ul-Islam, S. Yasir, S. Khan, S. Manan, A. Shehzad, M. W. Ahmad, R. Al-Shannaq, S. U. Islam, Y. Abbas and F. Subhan, Ex situ fabrication and bioactivity characterization of Neem and Sage-infused bacterial cellulose membranes for sustainable antimicrobial applications, Int. J. Biol. Macromol., 2025, 287, 138433 Search PubMed.
- C. Mouro, A. Gomes, A. P. Gomes and I. C. Gouveia, Sustainable bacterial cellulose production using low-cost fruit wastewater feedstocks, Nanomaterials, 2025, 15(4), 271 Search PubMed.
- S. Singh, I. Santhosh, S. Selvaraj and S. R. Holla, Decayed fruit waste juice as a sustainable medium for high-yield microbial cellulose production: optimization, characterization and bioactivity studies, Prep. Biochem. Biotechnol., 2025, 55(9), 1166–1179 Search PubMed.
- A. Esmail, M. Morais, U. D. Yilmazer, L. A. Neves and F. Freitas, Bacterial cellulose production through the valorization of waste apple pulp and stale bread, Biomass Convers. Biorefin., 2025, 15(9), 14587–14602 CrossRef CAS.
- N. A. Shavyrkina, E. A. Skiba, A. E. Kazantseva, E. K. Gladysheva, V. V. Budaeva, N. V. Bychin, Y. A. Gismatulina, E. I. Kashcheyeva, G. F. Mironova, A. A. Korchagina and I. N. Pavlov, Static culture combined with aeration in biosynthesis of bacterial cellulose, Polymers, 2021, 13(23), 4241 CrossRef CAS PubMed.
- M. Liu, S. Li, Y. Xie, S. Jia, Y. Hou, Y. Zou and C. Zhong, Enhanced bacterial cellulose production by Gluconacetobacter xylinus via expression of Vitreoscilla hemoglobin and oxygen tension regulation, Appl. Microbiol. Biotechnol., 2018, 102(3), 1155–1165 Search PubMed.
- L. Wang, H. Zheng, W. Wang, K. Deng and H. Tian, Physicochemical properties of bacterial cellulose from a strain of Komagataeibacter intermedius and analytical studies on its application, Int. J. Biol. Macromol., 2024, 283, 137472 Search PubMed.
- M. Yilmaz and Y. Goksungur, Optimization of bacterial cellulose production from waste figs by Komagataeibacter xylinus, Fermentation, 2024, 10(9), 466 CrossRef CAS.
- V. Adamopoulou, A. Bekatorou, V. Brinias, P. Michalopoulou, C. Dimopoulos, J. Zafeiropoulos, T. Petsi and A. A. Koutinas, Optimization of bacterial cellulose production by Komagataeibacter sucrofermentans in synthetic media and agrifood side streams supplemented with organic acids and vitamins, Bioresour. Technol., 2024, 398, 130511 Search PubMed.
- J. T. Hsieh, M. J. Wang, J. T. Lai and H. S. Liu, A novel static cultivation of bacterial cellulose production by intermittent feeding strategy, J. Taiwan Inst. Chem. Eng., 2016, 63, 46–51 Search PubMed.
- J. Chen, F. Hong, H. Zheng, L. Zheng and B. Du, Using static culture method to increase the production of Acetobacter xylinum bacterial cellulose, J. Nat. Fibers, 2024, 21(1), 2288286 Search PubMed.
- C. H. Ch'ng, M. R. Rahman, I. I. Muhamad and D. N. Zaidel, Optimization of Bacterial Cellulose Production from Pineapple Waste using Different Fermentation Method, Chem. Eng. Trans., 2020, 78, 559–564 Search PubMed.
- K. C. Cheng, J. Catchmark and A. Demirci, in Enhanced Production of Bacterial Cellulose Production by Using Biofilm Reactor and its Material Property Analysis, American Society of Agricultural and Biological Engineers, Reno, Nevada, 2009, p. 1 Search PubMed.
- S. C. Lazarini, C. Yamada, T. R. da Nóbrega and W. R. Lustri, Production of sphere-like bacterial cellulose in cultivation media with different carbon sources: a promising sustained release system of rifampicin, Cellulose, 2022, 29(11), 6077–6092 CrossRef CAS.
- K. Watanabe, M. Tabuchi, Y. Morinaga and F. Yoshinaga, Structural features and properties of bacterial cellulose produced in agitated culture, Cellulose, 1998, 5(3), 187–200 CrossRef CAS.
- P. Singhsa, R. Narain and H. Manuspiya, Physical structure variations of bacterial cellulose produced by different Komagataeibacter xylinus strains and carbon sources in static and agitated conditions, Cellulose, 2018, 25(3), 1571–1581 CrossRef CAS.
- M. Ghamari, D. Sun, Y. Dai, C. H. See, H. Yu, M. Edirisinghe and S. Sundaram, Valorization of diverse waste-derived nanocellulose for multifaceted applications: A review, Int. J. Biol. Macromol., 2024, 280, 136130 Search PubMed.
- A. S. Yordshahi, M. Moradi, H. Tajik and R. Molaei, Design and preparation of antimicrobial meat wrapping nanopaper with bacterial cellulose and postbiotics of lactic acid bacteria, Int. J. Food Microbiol., 2020, 321, 108561 Search PubMed.
- Y. Xie, X. Niu, J. Yang, R. Fan, J. Shi, N. Ullah, X. Feng and L. Chen, Active biodegradable films based on the whole potato peel incorporated with bacterial cellulose and curcumin, Int. J. Biol. Macromol., 2020, 150, 480–491 Search PubMed.
- Y. N. Yang, K. Y. Lu, P. Wang, Y. C. Ho, M. L. Tsai and F. L. Mi, Development of bacterial cellulose/chitin multi-nanofibers based smart films containing natural active microspheres and nanoparticles formed in situ, Carbohydr. Polym., 2020, 228, 115370 CrossRef PubMed.
- K. A. Zahan, N. M. Azizul, M. Mustapha, W. Y. Tong, M. S. Rahman and I. S. Sahuri, Application of bacterial cellulose film as a biodegradable and antimicrobial packaging material, Mater. Today: Proc., 2020, 31, 83–88 Search PubMed.
- H. Haghighi, M. Gullo, S. La China, F. Pfeifer, H. W. Siesler, F. Licciardello and A. Pulvirenti, Characterization of bio-nanocomposite films based on gelatin/polyvinyl alcohol blend reinforced with bacterial cellulose nanowhiskers for food packaging applications, Food Hydrocolloids, 2021, 113, 106454 Search PubMed.
- M. Kumar, V. Kumar and S. Saran, Current research and advances in the molecular aspect of bacterial cellulose synthesis with special emphasis on the production of bacterial cellulose from waste and its food applications, Cellulose, 2024, 31(6), 3323–3351 CrossRef CAS.
- R. Yekta, R. Abedi-Firoozjah, S. Azimi Salim, A. Khezerlou and K. Abdolmaleki, Application of cellulose and cellulose derivatives in smart/intelligent bio-based food packaging, Cellulose, 2023, 30(16), 9925–9953 CrossRef CAS.
- T. Almeida, A. Karamysheva, B. F. Valente, J. M. Silva, M. Braz, A. Almeida, A. J. Silvestre, C. Vilela and C. S. Freire, Biobased ternary films of thermoplastic starch, bacterial nanocellulose and gallic acid for active food packaging, Food Hydrocolloids, 2023, 144, 108934 CrossRef CAS.
- F. A. Silva, M. Carvalho, T. B. de Carvalho, M. Gama, F. Poças and P. Teixeira, Antimicrobial activity of in-situ bacterial nanocellulose-zinc oxide composites for food packaging, Food Packag. Shelf Life, 2023, 40, 101201 CrossRef.
- L. F. Amorim, C. Mouro, M. Riool and I. C. Gouveia, Antimicrobial food packaging based on prodigiosin-incorporated double-layered bacterial cellulose and chitosan composites, Polymers, 2022, 14(2), 315 Search PubMed.
- H. Abral, A. B. Pratama, D. Handayani, M. Mahardika, I. Aminah, N. Sandrawati, E. Sugiarti, A. N. Muslimin, S. M. Sapuan and R. A. Ilyas, Antimicrobial edible film prepared from bacterial cellulose nanofibers/starch/chitosan for a food packaging alternative, Int. J. Polym. Sci., 2021, 2021(1), 6641284 Search PubMed.
- R. M. Albuquerque, H. M. Meira, I. D. Silva, C. J. Silva, F. C. Almeida, J. D. Amorim, G. M. Vinhas, A. F. Costa and L. A. Sarubbo, Production of a bacterial cellulose/poly (3-hydroxybutyrate) blend activated with clove essential oil for food packaging, Polym. Polym. Compos., 2021, 29(4), 259–270 Search PubMed.
- O. M. Atta, S. Manan, A. A. Ahmed, M. F. Awad, M. Ul-Islam, F. Subhan, M. W. Ullah and G. Yang, Development and characterization of yeast-incorporated antimicrobial cellulose biofilms for edible food packaging application, Polymers, 2021, 13(14), 2310 Search PubMed.
- Y. Rasouli, M. Moradi, H. Tajik and R. Molaei, Fabrication of anti-Listeria film based on bacterial cellulose and Lactobacillus sakei-derived bioactive metabolites; application in meat packaging, Food Biosci., 2021, 42, 101218 CrossRef CAS.
- Y. Wen, J. Liu, L. Jiang, Z. Zhu, S. He, S. He and W. Shao, Development of intelligent/active food packaging film based on TEMPO-oxidized bacterial cellulose containing thymol and anthocyanin-rich purple potato extract for shelf life extension of shrimp, Food Packag. Shelf Life, 2021, 29, 100709 CrossRef CAS.
- Y. Xu, X. Liu, Q. Jiang, D. Yu, Y. Xu, B. Wang and W. Xia, Development and properties of bacterial cellulose, curcumin, and chitosan composite biodegradable films for active packaging materials, Carbohydr. Polym., 2021, 260, 117778 Search PubMed.
- N. A. Yanti, S. W. Ahmad, L. O. Ramadhan, M. Jamili, T. Walhidayah and J. Mamangkey, Properties and application of edible modified bacterial cellulose film based sago liquid waste as food packaging, Polymers, 2021, 13(20), 3570 Search PubMed.
- M. Rollini, A. Musatti, D. Cavicchioli, D. Bussini, S. Farris, C. Rovera, D. Romano, S. De Benedetti and A. Barbiroli, From cheese whey permeate to Sakacin-A/bacterial cellulose nanocrystal conjugates for antimicrobial food packaging applications: a circular economy case study, Sci. Rep., 2020, 10(1), 21358 Search PubMed.
- C. Rovera, F. Fiori, S. Trabattoni, D. Romano and S. Farris, Enzymatic hydrolysis of bacterial cellulose for the production of nanocrystals for the food packaging industry, Nanomaterials, 2020, 10(4), 735 CrossRef CAS PubMed.
- X. Zhai, D. Lin, W. Li and X. Yang, Improved characterization of nanofibers from bacterial cellulose and its potential application in fresh-cut apples, Int. J. Biol. Macromol., 2020, 149, 178–186 CrossRef CAS PubMed.
- S. Bandyopadhyay, N. Saha, U. V. Brodnjak and P. Sáha, Bacterial cellulose and guar gum based modified PVP-CMC hydrogel films: Characterized for packaging fresh berries, Food Packag. Shelf Life, 2019, 22, 100402 CrossRef.
- R. M. Viana, N. M. Sá, M. O. Barros, B. M. de Fátima and H. M. Azeredo, Nanofibrillated bacterial cellulose and pectin edible films added with fruit purees, Carbohydr. Polym., 2018, 196, 27–32 CrossRef CAS PubMed.
- L. Stasiak-Różańska, A. Berthold-Pluta, T. Aleksandrzak-Piekarczyk, A. Koryszewska-Bagińska and M. Garbowska, Antimicrobial activity against Cronobacter of plant extracts and essential oils in a matrix of bacterial cellulose, Polymers, 2024, 16(16), 2316 CrossRef PubMed.
- G. Chen, K. Wang, P. Chen, D. Cai, Y. Shao, R. Xia, C. Li, H. Wang, F. Ren, X. Cheng and Y. Yu, Fully Biodegradable Packaging Films for Fresh Food Storage Based on Oil-Infused Bacterial Cellulose, Adv. Sci., 2024, 11(23), 2400826 Search PubMed.
- A. Irimia, V. C. Grigoraş and C. M. Popescu, Active cellulose-based food packaging and its use on foodstuff, Polymers, 2024, 16(3), 389 CrossRef CAS PubMed.
- I. T. Liu, C. Y. Li and Y. C. Liao, Sustainable multi-purpose bacterial cellulose composite for food packaging via facile successive infiltration, Carbohydr. Polym. Technol. Appl., 2025, 9, 100703 Search PubMed.
- T. Almeida, A. Karamysheva, B. F. Valente, J. M. Silva, M. Braz, A. Almeida, A. J. Silvestre, C. Vilela and C. S. Freire, Biobased ternary films of thermoplastic starch, bacterial nanocellulose and gallic acid for active food packaging, Food Hydrocolloids, 2023, 144, 108934 Search PubMed.
- Á. Agüero, E. Corral Perianes, S. S. Abarca de las Muelas, D. Lascano, M. D. de la Fuente García-Soto, M. A. Peltzer, R. Balart and M. P. Arrieta, Plasticized mechanical recycled PLA films reinforced with microbial cellulose particles obtained from kombucha fermented in yerba mate waste, Polymers, 2023, 15(2), 285 CrossRef PubMed.
- T. Agha and A. Kati, Development of functional bacterial cellulose composites from Kombucha waste for biodegradable food packaging, Discover Appl. Sci., 2025, 7(8), 840 CrossRef CAS.
- C. Mouro, A. Gomes, A. P. Gomes and I. C. Gouveia, Sustainable bacterial cellulose production using low-cost fruit wastewater feedstocks, Nanomaterials, 2025, 15(4), 271 Search PubMed.
- M. J. Fabra, A. López-Rubio, J. Ambrosio-Martín and J. M. Lagaron, Improving the barrier properties of thermoplastic corn starch-based films containing bacterial cellulose nanowhiskers by means of PHA electrospun coatings of interest in food packaging, Food Hydrocolloids, 2016, 61, 261–268 Search PubMed.
- C. Rovera, F. Fiori, S. Trabattoni, D. Romano and S. Farris, Enzymatic hydrolysis of bacterial cellulose for the production of nanocrystals for the food packaging industry, Nanomaterials, 2020, 10(4), 735 CrossRef CAS PubMed.
- S. P. Lin, L. Hong, C. C. Hsieh, Y. H. Lin, Y. C. Chou, S. P. Santoso, C. W. Hsieh, T. Y. Tsai and K. C. Cheng, In situ modification of foaming bacterial cellulose with chitosan and its application to active food packaging, Int. J. Biol. Macromol., 2024, 279, 135114 CrossRef CAS PubMed.
- X. Ma, Y. Chen, J. Huang, P. Lv, T. Hussain and Q. Wei, In situ formed active and intelligent bacterial cellulose/cotton fiber composite containing curcumin, Cellulose, 2020, 27(16), 9371–9382 CrossRef CAS.
- J. Sulistyo, P. S. Winarno, I. Y. Pratiwi, L. P. Ridfan, K. M. Pranata and R. M. Chick, Preparation of active food packaging and coating material based on bacterial cellulose to increase food safety, J. Teknol. Ind. Pangan, 2023, 34(1), 48–61 CrossRef.
- N. A. Yanti, S. W. Ahmad, L. O. Ramadhan and T. Walhidayah, Antimicrobial and antioxidant activity of bacterial cellulose-based edible film from sago liquid waste incorporated with spices, in IOP Conference Series: Earth and Environmental Science. IOP Publishing, 2023, vol. 1271, 1, p. 012061 Search PubMed.
- H. El-Gendi, A. Salama, E. M. El-Fakharany and A. K. Saleh, Optimization of bacterial cellulose production from prickly pear peels and its ex situ impregnation with fruit byproducts for antimicrobial and strawberry packaging applications, Carbohydr. Polym., 2023, 302, 120383 CrossRef CAS PubMed.
- L. Yang, W. Sheng, Y. Yang, Y. Yang, K. He, G. Jiang and Y. Tian, Bacterial cellulose and photothermal material from biomass-based zero-waste biorefinery functionalized chitosan film for sustainable active food packaging, Food Hydrocolloids, 2025, 162, 110977 CrossRef CAS.
- L. Deng, W. Zheng, Q. Liu, T. Hu, M. Li, X. Wu, Y. Liu, D. Yang, L. Chen, W. Weng and Y. Zhang, Preparation and characterization of chitosan/bacterial cellulose nanofibers-based food packaging films blended with aqueous phase from purple passion fruit peel via hydrothermal carbonization, Int. J. Biol. Macromol., 2025, 301, 140258 CrossRef CAS PubMed.
- L. Quijano, R. Rodrigues, D. Fischer, J. D. Tovar-Castro, A. Payne, L. Navone, Y. Hu, H. Yan, P. Pinmanee, E. Poon and J. Yang, Bacterial cellulose cookbook: A systematic review on sustainable and cost-effective substrates, J. Bioresour. Bioprod., 2024, 9(4), 379–409 CAS.
- L. Quijano, R. Rodrigues, D. Fischer, J. D. Tovar-Castro, A. Payne, L. Navone, Y. Hu, H. Yan, P. Pinmanee, E. Poon and J. Yang, Bacterial cellulose cookbook: A systematic review on sustainable and cost-effective substrates, J. Bioresour. Bioprod., 2024, 9(4), 379–409 CAS.
- T. R. Gagliardi, A. de Farias Nascimento and G. A. Valencia, Kombucha bacterial cellulose: a promising biopolymer for advanced food and nonfood applications, Foods, 2025, 14(5), 738 CrossRef CAS PubMed.
- N. H. Avcioglu, Bacterial cellulose: recent progress in production and industrial applications, World J. Microbiol. Biotechnol., 2022, 38(5), 86 CrossRef CAS PubMed.
- I. C. Nóvak, B. Segat, M. C. Garcia, A. P. Pezzin and A. L. Schneider, Alternative production of bacterial cellulose by Komagataeibacter hansenii and microbial consortium, Polímeros, 2024, 34, e20240021 CrossRef.
- M. V. Parchaykina, E. V. Liyaskina, A. O. Bogatyreva, M. A. Baykov, D. S. Gotina, N. E. Arzhanov, A. I. Netrusov and V. V. Revin, Cost-effective production of bacterial cellulose and tubular materials by cultivating Komagataeibacter sucrofermentans B-11267 on a molasses medium, Polymers, 2025, 17(2), 179 CrossRef CAS PubMed.
- A. D. de Lima, V. F. Caetano and G. M. Vinhas, Development of active biodegradable films based on bacterial cellulose/poly (vinyl alcohol) and antioxidant food complex for application in food packaging, Polym. Bull., 2024, 81(16), 14605–14624 CrossRef CAS.
- A. Forte, F. Dourado, A. Mota, B. Neto, M. Gama and E. C. Ferreira, Life cycle assessment of bacterial cellulose production, Int. J. Life Cycle Assess., 2021, 26(5), 864–878 CrossRef CAS.
- M. Bolla, M. Pettinato, P. F. Ferrari, B. Fabiano and P. Perego, Polyhydroxyalkanoates production from laboratory to industrial scale: A review, Int. J. Biol. Macromol., 2025, 310, 143255 CrossRef CAS PubMed.
- S. Singh, R. Kumari and Z. Kamble, Bridging the Gap Between Laboratory Research and Industrial Implementation of Green Composite Technologies: A Review, Polym. Compos., 2026, 1–30 Search PubMed.
- J. Guo, Y. Zhang, J. Fang, Z. Ma, C. Li, M. Yan, N. Qiao, Y. Liu and M. Bian, Reduction and reuse of forestry and agricultural bio-waste through innovative green utilization approaches: A review, Forests, 2024, 15(8), 1372 CrossRef.
- P. K. Sadh, S. Duhan and J. S. Duhan, Agro-industrial wastes and their utilization using solid state fermentation: a review, Bioresour. Bioprocess., 2018, 5(1), 1 CrossRef.
- P. Deshpande, S. Wankar, S. Mahajan, Y. Patil, J. Rajwade and A. Kulkarni, Bacterial cellulose: Natural biomaterial for medical and environmental applications, J. Nat. Fibers, 2023, 20(2), 2218623 CrossRef.
- P. Cazón and M. Vázquez, Bacterial cellulose as a biodegradable food packaging material: A review, Food Hydrocolloids, 2021, 113, 106530 CrossRef.
- R. D. e Silva, A. I. Santa Brígida, M. de Freitas Rosa, R. M. da Silva Neto, W. A. Spinosa, E. B. de Sá Filho and M. C. de Figueirêdo, An approach for implementing ecodesign at early research stage: A case study of bacterial cellulose production, J. Cleaner Prod., 2020, 269, 122245 CrossRef.
- F. Hong, Y. X. Zhu, G. Yang and X. X. Yang, Wheat straw acid hydrolysate as a potential cost-effective feedstock for production of bacterial cellulose, J. Chem. Technol. Biotechnol., 2011, 86(5), 675–680 CrossRef CAS.
- Y. Huang, C. Zhu, J. Yang, Y. Nie, C. Chen and D. Sun, Recent advances in bacterial cellulose, Cellulose, 2014, 21(1), 1–30 CrossRef CAS.
- H. M. Azeredo, H. Barud, C. S. Farinas, V. M. Vasconcellos and A. M. Claro, Bacterial cellulose as a raw material for food and food packaging applications, Front. Sustainable Food Syst., 2019, 3, 7 CrossRef.
- Y. Dai, D. Sun, D. O'Rourke, S. Velusamy, S. Sundaram and M. Edirisinghe, Harnessing cow manure waste for nanocellulose extraction and sustainable small-structure manufacturing, J. Cleaner Prod., 2025, 509, 145530 CrossRef CAS.
- M. O. Aydogdu, A. Delbusso and M. Edirisinghe, A battery powered approach to pressurised spinning: Introducing the sustainability concept and shaping the future of fibre production methodologies, Appl. Energy, 2025, 397, 126331 CrossRef.
- W. S. Yong, Y. L. Yeu, P. P. Chung and K. H. Soon, Extraction and characterization of microcrystalline cellulose (MCC) from durian rind for biocomposite application, J. Polym. Environ., 2024, 32(12), 6544–6575 CrossRef CAS.
- I. Leppänen, M. Vikman, A. Harlin and H. Orelma, Enzymatic degradation and pilot-scale composting of cellulose-based films with different chemical structures, J. Polym. Environ., 2020, 28(2), 458–470 CrossRef.
- T. A. Hameed, A. Salama and R. A. Nasr, Synthesis of reusable CNF/Ag2Se films as visible-light-driven photocatalysts for photocatalytic degradation of MB, J. Polym. Environ., 2024, 32(9), 4440–4455 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |