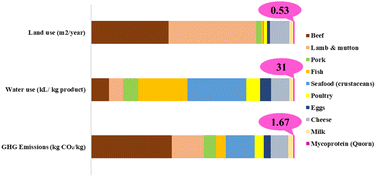Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Fungal factories for feeding the future: mycoproteins as a sustainable protein source at the interface of food, health, and biotechnology innovation
Cristina Firincă†
,
Mariana Constantin†,
Iuliana Răut,
Lucian-Gabriel Zamfir,
Maria-Luiza Jecu,
Mihaela Doni* and
Ana-Maria Gurban *
*
National Research & Development Institute for Chemistry &Petrochemistry – ICECHIM, Biotechnology and Bioanalysis Department, 202 Spl. Independenței, 060021, Bucharest, Romania. E-mail: ana-maria.gurban@icechim.ro; mihaela.doni@icechim.ro
First published on 17th April 2026
Abstract
Mycoproteins are gaining attention as alternative protein sources that have the potential to address the nutritional and environmental issues linked to animal products consumption. Since the discovery of Fusarium venenatum as a viable source of mycoproteins, numerous other fungal strains have been explored, leading to products with improved nutritional profiles. From a nutritional perspective, they provide high-quality, digestible proteins with potential gastrointestinal, metabolic and cardiovascular benefits. Regarding the environmental impact, production methods require significantly less land, water and energy compared with animal-based products, which results in lower greenhouse-gas emissions. Economically, mycoproteins fit well into circular systems by integrating various agro-wastes and industrial by-products into the fermentation process. However, potential allergenicity, limited long-term clinical data, scalable production optimization and consumer acceptance are still important challenges to be taken into consideration. This review investigates both the benefits and drawbacks of conventional technologies, while identifying existing gaps and innovation potential driven by artificial intelligence-assisted process optimization, genetic engineering and potential functional properties.
Sustainability spotlightThe review highlights the benefits and drawbacks of mycoprotein as one of the most promising sustainable alternatives for traditional animal-derived proteins. Its production uses significantly less land, water and energy than animal farming, while avoiding the high greenhouse gas emissions and biodiversity loss associated with meat production. By incorporating renewable energy and secondary agricultural flows as raw materials, the production of mycoproteins supports circular bioeconomy strategies. This paper serves as aid for researchers, stakeholders and consumers alike to better understand the innovative potential of mycoproteins, which strongly allign with the UN's Sustainable Development Goals SDG 2 (Zero Hunger), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action). |
1. Introduction
The growing interest in sustainable and nutritionally balanced foods has drawn greater attention towards mycoproteins, products derived from fungal biomass, as a valuable alternative with applications in biotechnology, circular economy and climate change mitigation efforts.Current demographic statistics indicate that the global population will exceed 8.5 billion people by 2030, and could reach 9.7 billion by 2050.1 The rapid population growth, coupled with the increasing demand for food, has amplified the interest for finding sustainable alternatives to conventional agriculture, as well as provide nutritious, abundant food sources with lower environmental impact and more health benefits. Dietary challenges play an influential role in health, well-being and overall socioeconomic evolution, both in developing countries, where deficiencies and food scarcity tend to be frequent, while in developed countries overconsumption and food waste represent a more prevalent issue.2,3 Furthermore, conventional farming is associated with a number of drawbacks such as high greenhouse gas emissions, extensive land use and water consumption, environment degradation, animal welfare concerns and agrochemicals overuse.4 Meat production has reached over 350 million tons per year globally, while associated greenhouse gas emissions, representing 6% of the total emissions worldwide, reached over 3 Gt CO2e per year in 2021.5,6 For this reason, alternative protein sources have gained considerable attention, deriving from sources that are considered to be more sustainable, such as cell cultures, plants, algae, fungi and even insects.
Alternative protein sources may seem as desirable options for addressing multiple global concerns; however, lab-grown meat or insect-based proteins are still quite controversial and more studies are required to assess the safety, health effects and environmental impact of meat alternatives. As shown in the State of Global Policy Report issued in 2023 by the Good Food Institute Europe,7 governments around the world are investing heavily in research & development and commercialization of alternative protein sources, all in support of climate change mitigation, economic development, food security and public health. Even so, several countries still oppose or stall the production and commercialization of lab-cultured meat and even insect-based products. Examples include the United States, Australia, New Zealand, and European countries such as Italy, France, Hungary, Romania, and Sweden.8
Taking into consideration the limitations regarding public perception and legislation when it comes to alternative proteins, fungal-based proteins, also known as mycoproteins, are desirable for their low production costs, high nutritional value especially in terms of their amino acid profile, resistance to seasonal variations and low environmental impact, being even able to convert waste materials into valuable biomass.9 Their production process involves the fermentation of various substrates through fungal metabolism, requiring less land and water use and generating less greenhouse gas emissions compared with meat products and even plant-based products, rendering them as affordable, more environmentally friendly protein sources.10
Particularly in the last decades, numerous start-ups and projects have emerged, attempting to substitute meat-derived protein in products, with companies such as Quorn, Nature's Fynd, Better Meat Co. or Adamo Foods gaining more recognition.11,12 This trend is also highlighted in the interest of researchers for the subject. An analysis of Google Scholar records on publications from 1990 to 2025 revealed an exponential growth of interest in alternative protein sources starting in 2012, while research specifically focused on mycoproteins has grown considerably, especially from 2022 onward (Fig. 1). This observed trend can be justified by an increase in health awareness and environmental safety consciousness, concentrated especially in developed countries characterized by high food security and income.13 It is important to note that a decline in publications on the particular topic of alternative protein sources was observed starting from 2022. Correlated with the significant growth in publications on mycoprotein, this indicates a transition towards more specialized topics and technologies in the field. In addition, several market and economic factors have also influenced the evolution of the alternative-protein sector. After a period of rapid expansion, global investments in alternative proteins have started to decline, accompanied by an increase in inflationary trends which have affected market dynamics.14 In that sense, while the alternative protein market reaches a point of stagnation, research focuses on more in-depth technological and methodological development.
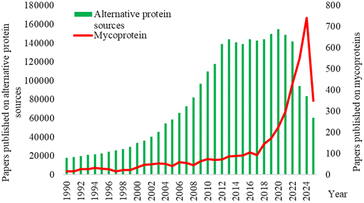 | ||
| Fig. 1 Number of publications on the subject of alternative protein sources (bars) and mycoproteins (line) between 1990–2025. | ||
Mycoproteins open up new opportunities aligned with important global priorities, such as the Sustainable Development Goals (SDGs), and the One Health Initiative by promoting circular economy and holistic ecosystemic practices. More specifically, they can contribute to achieving several SDGs, such as zero hunger, good health and well-being, responsible consumption and production, sustainable cities and communities, sustainable industrialization and innovation, decent work and economic growth, climate action and land conservation.15,16 By strengthening the essential link between environmental integrity and food security, this approach can not only aid in environment protection by reintegrating organic waste into the economic cycle and using less water and energy for production compared to conventional animal-based products,17,18 but it can also contribute to mitigating poverty and food insecurity.19,20 New industrial sectors can be developed as mycoprotein production is further included in research, development and commercialization. Furthermore, numerous organic wastes may be used as substrates, such as seaweed;21 pea processing byproducts;22 date production;23 soybean whey24 or apple pomace25 to be transformed into value-added products.
Since the discovery of Fusarium venenatum as the first strain used and marketed for meat-analogous proteins, numerous other fungal species have been explored, both in the category or micromycetes (filamentous fungi) as well as macromycetes (mushrooms), presenting advantages as well as disadvantages. While micromycetes offer high yield and productivity with less resources, the main risks lie with mycotoxin production and allergic reactions, requiring strict regulation and monitoring.26 On the other hand, macromycetes, particularly edible mushrooms, are more desirable in terms of consumer acceptance, safety, nutritional profile and the presence of medicinal bioactive compounds which may offer additional health benefits.27
To ensure clarity and argumentative coherence of the manuscript, this review follows a structured and integrative approach to guide the reader through key aspects of mycoproteins as an alternative source of sustainable protein. First, an overview of the mycoprotein production process is provided, with a focus on filamentous fungal fermentation technologies and key technological parameters that influence biomass generation and product characteristics. Subsequently, the technological and nutritional contributions of mycoproteins are discussed, highlighting functional properties in food formulations, protein quality and potential health benefits. This review also addresses potential risks, limitations, and safety considerations associated with the consumption and production of mycoproteins, including allergenicity, processing constraints, and consumer perception issues. Particular attention is also paid to the environmental implications of mycoprotein production, along with the current regulatory frameworks governing their approval, labeling, and marketing in different regions. Finally, the review explores future prospects and emerging research directions, highlighting advances in fermentation technologies, strain development and optimization, and innovative applications of mycoproteins within next-generation sustainable food systems.
1.1 Review methodology
This review is based on a structured literature search conducted across Web of Science, PubMed, Scopus, and Google Scholar, covering all relevant publications up to 2025 on alternative proteins and mycoproteins. We included peer-reviewed journal articles published in English, alongside relevant websites from official institution. Studies were selected if they met one or more of the following criteria: (1) described or evaluated mycoprotein production methods, including conventional and emerging approaches; (2) focused on process innovation; (3) reported studies regarding safety and health assessments of fungal-based proteins; (4) advanced technological strategies, in particular, synthetic biology, machine learning and Artificial Intelligence-driven approaches with applicability for mycoproteins; or (5) highlighted challenges and knowledges gaps related to process scale-up or expansion to new strains and by-products, consumer awareness, regulatory implementation and multi-sectorial applicability.2. Mycoprotein production methods
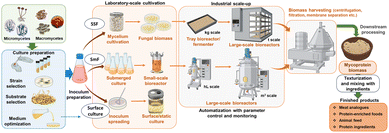
Mycoprotein production is generally based on controlled fungal fermentation processes that transform carbohydrate-rich substrates into high-quality biomass. The principles underlying these processes include selecting a safe, fast-growing filamentous fungus, optimizing the composition of nutrients required for its growth, and maintaining stringent culture media conditions – such as temperature, pH, aeration, and agitation – to obtain consistent growth and metabolite profiles. Successful mycoprotein production is conditioned by efficient harvesting of the fungal biomass and precise heat treatment to lower RNA levels, followed by texturing processes that give the final product its characteristic meat-like structure. Overall, mycoprotein production must primarily consider the safety, scalability, and sustainability of the entire process to ensure a nutritious and reliable alternative source of protein.The main technologies available for mycoprotein production harness the natural fermentative properties of fungi by three approaches, with distinct mechanisms and applications: solid-state fermentation (SSF), submerged fermentation (SmF), and surface culture fermentation (Fig. 2).28
Generally, the first two methods are efficient for large-scale production, while surface culture method is more preferable in laboratory scale settings, as it is more expensive to upscale.26 Although solid-state fermentation (SSF) and submerged fermentation (SmF) represent well-established approaches for fungal cultivation, over the years advances in bioreactor designs, integrated bioprocess engineering, as well as improved control and monitoring of fermentation parameters, have been reported to significantly influence fungal growth, biomass yield, and protein quality. These innovations contribute to improving the efficiency and industrial feasibility of large-scale mycoprotein production, with novel producers emerging on the market of alternative proteins by harnessing advances in microbial biotechnology. Accordingly, the subsequent section will examine the advantages and disadvantages of each method, highlighting their contribution to sustainable food production and emerging biotechnological innovations.
2.1. Solid-state fermentation (SSF)
Solid-state fermentation (SSF) represents a high-potential approach for mycoprotein production, as it allows filamentous fungi to grow on minimally processed agro-industrial residues under low-humidity conditions, mimicking their natural environment.Solid-state fermentation (SSF) is a traditional bioprocess widely used for the production of fermented foods such as miso, tempeh and soy sauce, as well as metabolites such as antibiotics, organic acidsand biosurfactants.29 Recently, SSF has gained new interest for the possibility of using agro-industrial byproducts, with multiple applications in bioremediation, the production of bioactive compounds and biofuels.30 The process requires the cultivation of the selected fungal strains on moist solid substrates, in the absence of free-flowing water. A wide range of materials may be used as carbon and nitrogen sources, such as starchy products, proteinaceous products, lignocellulosic products, and substrates rich in soluble sugars. The moisture content of the medium typically ranges between 30% and 75%, particular values being adjusted to support microbial growth and metabolism. The main advantages of the method are high productivity, reduced production costs, low energy requirements and minimal risk of microbial contamination.28 These advantages stem from the particularities of the process. The limited water requirement and inherent low water activity provide a reduced contamination risk compared with submerged fermentation practices.31 Also, a high inoculum density is often used in SSF processes, which is associated with faster hyphal development, competitive exclusion of potential contaminants and a more efficient utilization of the substrate.31,32 Furthermore, by repurposing wasteful materials, the method has the potential to be inexpensive, depending on the type of byproduct used. For instance, a techno-economic analysis conducted by Cao et al.33 revealed that using almond hull as feedstock for fungal biomass production requires lower costs compared to using glucose or rice straw. However, the authors highlight that raw materials represent a significant variable influencing overall operating costs. Lignocellulosic biomass is an abundant resource, but the main limitation is the complexity of pretreatments certain materials require for their conversion into fermentable sugars.34 Therefore, it is important to identify promising feedstock options for the optimization of costs, production efficiency and substrate valorisation. Additionally, the environmental impact is low, derived from a low production of wastewater and reduced downstream process, with easy recovery of products.29,35 However, the method is difficult to monitor and control on a large scale, the mass and heat transfer within the substrate is not homogenous and can generate uneven growth, along with the long fermentation time required, which pose a challenge for upscaling to biorefineries.36
SSF has gained attention in the biotechnological sector over the years, for the production of a wide range of compounds such as biopesticides, bioactive molecules, antibiotics, biosurfactants and biofuels. The heterogeneity in heat and mass transfer can be addressed through various types of bioreactors, such as static, or dynamic. In terms of scalability, tray bioreactors are considered feasible, easy to maintain, with minimal damage to the mycelium. In this method, the substrate is evenly distributed onto trays stacked in layers, with controlled airflow, humidity and temperature conditions. This setting allows the even and rapid growth of fungal mycelium due to a larger surface area exposed to air compared with a submerged setting.37 Various bioreactor designs, such as rotary disc, rotary drum, static bed, packed-bed, or trickle bed and horizontal tube systems also enhance oxygen availability and uniformize growth conditions throughout the substrates. Several established companies have developed mycelium-based products through SSF. For instance, Ecovative has established important breakthroughs in numerous domains with mycelium-based biomaterials, from packaging, construction materials, to consumer goods such as plant-based meat alternatives. Their process involves growing Pleurotus sp. biomass on trays containing agricultural byproducts, using over 90% less land, less water and generating 33% less CO2eq compared to pork, according to their official information.38 The company leverages patented SSF techniques to control mycelium growth into a highly organized network of hyphae, with enhanced mechanical properties.39 Other companies, such as New Culture, Prime Roots or NovoNutrients produce alternative products from plant-based protein sources, processed through fungal fermentation, with similar sustainability claims as those mentioned by Ecovative.40–42 In recent years, substrate design has become more targeted towards improving functional properties of plant-based proteins, as well as designing nutrient-enhanced biomass from industrial bi-products.43
Few pilot-scale studies describe the optimization of SSF to address these limitations and improve the protein content in fungal-based products. New designs are being developed in order to create more controlled environments that allow the uniform transfer of nutrients and oxygen to the substrate, with potential large-scale implementation. A tray bioreactor configuration, which is already implemented for the industrial production of enzymes, is more preferred for its simple operation, minimal mechanical involvement and efficient growth of the fungal strains as a result of extended surface area.44 Manan and co-authors developed a multi-layer tray bioreactor system, employing the optimization of key parameters to improve fungal growth and metabolism, coupled with real-time monitoring.45 Their findings offer a promising configuration for process control and scale-up potential.
The advantages of this SSF method of obtaining mycoproteins consist of higher biomass yields, improved textural properties and reduced energy requirements compared to submerged fermentation. Furthermore, this method can utilize inexpensive by-products such as cereal bran or fruit pomace, thus increasing sustainability and reducing production costs. However, precise control of parameters such as humidity, aeration and heat transfer is essential to ensure consistently high quality and safety of mycoproteins.
2.2. Submerged fermentation (SmF)
Submerged fermentation (SmF) is the most well-known and widely used method for producing mycoproteins, based on the growth of filamentous fungi in nutrient-rich liquid media under strictly controlled conditions. SmF requires the cultivation of microorganisms in large quantities of liquid medium for the decomposition of the substrate and the production of fungal biomass.The method has the advantages of accessible process control and reproducibility, high yield and suitability for air-lift bioreactors or stirred-tank reactors, allowing for precise control of key parameters such as pH, temperature, dissolved oxygen and agitation, while maintaining constant biomass quality and productivity, making it suitable for industrial-scale applications.46,47 The process allows a better control and monitoring of the physical parameters and final nutrient composition compared with SSF, however, this level of precision also involves a higher energy consumption and higher operating costs.48 Also, there is a risk of increased shear stress at high agitation rates, which may cause damage to the mycelial structure.49
As the benefits eventually outweigh the disadvantages, SmF has been the production method employed by Quorn™ for decades, providing significant experience in optimization of yield and efficiency.50 Several technical innovations have been implemented since the development of this method. While the initial setting was designed as a pilot-scale fermenter, over the years an airlift fermentation process was developed, which optimized oxygen transfer and improved the structure and texture of the fungal biomass, leading to higher yield and consistent quality. However, the main limitation of this process is the requirement for constant, rigorous sterilization in order to prevent the emerging of mutant strains.11 An efficient approach for addressing such issue has been proposed in the form of integrative omics, which enable continuous genetic monitoring of parental strains, identifying genes responsible for mutations and through further genetic engineering, improving stability and preventing mutant emergence.11,51 Pneumatic bioreactors, such as the aforementioned air-lift type, as well as bubble column reactors, represent promising alternatives to stirred tanks, allowing for more careful aeration and reducing shear stress onto the fungal biomass.52 To these structures, a number of improvements were implemented throughout the years, such as the addition of internal draft tubes which improve circulation, as well as computational fluid dynamics modelling, with more complex software and more detailed technologies allow the optimization of mixing, oxygen transfer and growth profiles.53 Modern SmF processes also integrate real-time monitoring of key parameters such as pH, biomass production, glucose consumption and dissolved oxygen, allowing for accurate adjustments throughout the process as well as reduced variability. Additionally, advancements have been made in the customization of bioreactor equipment and mechanisms, to allow a variety of cell preparation methods, expanding beyond simple culture growth towards the production of biopharmaceuticals, biofuels and even the microbial breakdown of pollutants.54
Several recent studies attempt to not only identify the potential of new fungal strain for mycoprotein production, but to also determine cost-effective substrates and optimal conditions for efficient mycoprotein production.55,56 Furthermore, the bioactive compound content in mycoproteins can also be improved. Using seawater fermentation, Yang et al., increased the iron and calcium content in mycoproteins produced with Fusarium venenatum biomass, with a 50% less freshwater waste for production. However, the authors recognize that the study still has limitations when it comes to ensuring a continuous flow culture system, and gaps in long-term consumption effects.57
The SSF and SmF methods can be integrated into sequential or hybrid systems to harness the advantages of each approach, enabling the co-production of valuable secondary metabolites alongside mycoproteins, and enhancing the bioavailability of micronutrients in the resulting biomass.58 Additionally, this combined method allows the valorization of agro-industrial byproducts in order to obtain products with high protein content, improved amino acid profile and high polyphenol content, with results comparable with other plant-based alternatives such as soybean meal.59 The combined use of SSF and SmF can streamline the fermentation process by coupling their complementary steps. In such systems, SSF is often employed as a pretreatment or initial phase to promote enzyme production and partial substrate hydrolysis, generating the required feedstock for SmF, enhancing substrate utilization efficiency and facilitating the bioconversion of complex materials or wastes.60
Although SmF can be energy-intensive due to aeration and mixing requirements, often involving subsequent steps to achieve the desired texture, SmF remains a robust and reliable platform for obtaining safe, high-quality mycoproteins on a large scale.
2.3. Surface-culture fermentation
Surface culture fermentation involves growing filamentous fungi as a thin mycelial layer on the surface of a liquid or semi-solid substrate, representing an intermediate approach between solid-state and submerged systems. While the two methods described above have industrial potential, surface-culture methods are mainly used in laboratory settings for screening, evaluation and optimization strain, due to the considerable labor cost and space that would be required for up-scaling.61 For this reason, attention is often focused on SSF and SmF for large-scale mycoprotein production. A screening of the mycoprotein production potential was demonstrated using several edible fungal strains such as F. venenatum,Aspergillus oryzae, Monascus purpureus, Neurospora intermedia and Rhizopus oryzae cultivated on pea processing byproducts through surface-culture fermentation, with the ulterior purpose for the most efficient strains to be further scaled-up in a bench airlift bioreactor.22 The method can be mainly used to identify the optimal culture conditions for secondary metabolite production62 and biomass accumulation.63 For certain species and target molecules (cordycepin), surface culture can induce the upregulation of specific genes encoding the biosynthesis of desired metabolites, thus increasing their yield, compared with submerged culture.64This method offers a number of advantages such as the need for simpler equipment, lower shear stress on the fungal biomass, and the ability to form naturally structured mycelial mats with desirable textural properties for mycoprotein applications. Although this method of surface culture fermentation reduces energy consumption compared to fully submerged fermentation, it also has limitations, such as slow biomass accumulation, difficulty in maintaining uniform growth conditions, and low scalability. Even so, surface culture fermentation remains a viable and potentially valuable technique for producing structured fungal biomass with minimal processing.
The main operational characteristics, advantages, and challenges associated with the main methods for mycoprotein synthesis are summarized in Table 1.
| Mycoprotein nutritional profile/yield | Substrate | Fungal strains | Advantages | Disadvantages |
|---|---|---|---|---|
| Submerged fermentation (SmF) | ||||
| Protein: 21–31% | • Seaweed21 | • Paradendryphiella salina21 | • Uniform distribution of nutrients26,49 | • High production costs46 |
| Fat: 1.2–4% | • Seaweed waste21 | • Agrocybe aegerita (AAE)65 | • Precise control of parameters26 | • Strict monitoring required for mutations and mycotoxin production29,48 |
| Carbohydrates: 30–75% | • Apple65 | • Pleurotus sapidus65 | • Shorter fermentation time36 | • Strict control requirements for cultivation parameters and sterilization26 |
| Energy: 253 kcal/100 g | • Pomegranate65 | • Lentinula edodes65 | • High biomass homogeneity36 | • Higher risk of shear stress on fungal biomass49 |
| Yield: 250–600 g kg−1 | • Aronia65 | • Wolfiporia cocos65 | • Fully automated process with high reproducibility36 | |
| • Leaf spinach65 | • Stropharia rugosoannulata65 | |||
| • Beet molasses65 | • P. sajorcaju65 | |||
| • P.salmonetramineus65 | ||||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Solid state fermentation (SSF) | ||||
| Protein: 22–47%; | • Stale bread66 | • Neurospora intermedia66 | • Minimum water consumption and wastewater generation26,61 | • Species selectivity for growth substrate, limiting the potential use of other strains26,36 |
| Fat: 4.4%; | • Brewer-spent grain66 | • Rhizopus oryzae66 | • Low energy consumption61 | • Difficulties in automation and quality control36 |
| Carbohydrate: 42% | • Grape bagasse67 | • Agaricus blazei67 | • Small fermenters61 | • Biomass heterogeneity36 |
| Energy: 326 Kcal/100 g | • Auricularia fuscosuccinea67 | • Inexpensive media, substrate as agricultural or industrial waste26,61 | • Heat and mass transfer gradients limit up-scaling26,36 | |
| Yield: 125 g kg−1 (for Pleurotus albidus) | • P. albidus67 | • Process control can be improved with sensors integration26 | ||
| 260 g kg−1 (for Neurospora intermedia) | ||||
| Total amino acids: 7.85% | ||||
| Fiber: 34.35% | ||||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Hybrid fermentation (combining SmF and SSF) | ||||
| Protein: 33% | •Stale bread58 | •N. intermedia58 | • Valorizesa multiple waste streams in one workflow58 | • Added process complexity58 |
| Fat: 26% | • Multiple valuable outputs (ethanol, biomass, secondary metabolites, proteins, enzymes)58 | • Longer processing time58 | ||
| Carbohydrates: 4.63% | • Enhanced bioconversion of complex substrates58 | • Intermediate processing required58 | ||
| Yield: 152 × 103 g kg−1 | • Combined operational precision with resource utilization efficiency58 | |||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Surface culture fermentation (SCF) | ||||
| Yield: −0.26 g−1 of byproduct22 | • Pea by-products;22 | • Aspergillus oryzae22 | • Simple, low-tech method29 | • Low productivity29 |
| Protein: 46.48%68 | • Date extract68 | • F. venenatum68 | • Small-scale optimization29 | • Labour-intensive29 |
| • Waste valorization29 | • High contamination risks29 | |||
| • Limited process control29,49 | ||||
In conclusion, the selection of the appropriate fermentation strategy depends on the production performance of the target strain, substrate availability, desired product characteristics, as well as the economic and environmental constrains of the complete process. Continuous advances in reactor design, bioengineering and monitoring technologies are expected to further enhance the efficiency and scalability of these fermentation processes, along with the broadening of strains and substrate options to be used, supporting the sustainable development of mycoproteins as a well-established alternative protein source. Hereinafter, we will expand on the nutritional benefits of mycoproteins as well as potential health considerations as emerging food products.
3. Nutritional aspects and health considerations
Mycoproteins are fungal-derived protein-rich biomasses, primarily produced from filamentous fungi such as Fusarium venenatum, and have emerged as one of the most innovative and sustainable protein sources in modern food biotechnology.69 Originally conceived in the 1960s as part of research based on single-cell protein production (SCP), mycoproteins have been developed as a strategic response to growing global concerns about food scarcity and protein-energy malnutrition. These early efforts aimed to harness microorganisms as efficient biological factories for nutrient-rich biomass, providing a scalable solution to major nutritional deficits in developing countries.The unique combination of high nutritional value, minimal ecological footprint and functional food properties has led to a growing interest in mycoproteins among consumers, policy makers and researchers alike. The decades-long commercialization of F. venenatum-based products under the brand Quorn has transformed a scientific innovation into a globally recognized food product, demonstrating the industrial and nutritional feasibility of fungal biomass as a high-quality food source, but also the willingness of consumers to adopt mycoprotein-based foods as meat substitutes.70
Given this historical and technological context, a more in-depth analysis of mycoproteins must consider both their nutritional profile and potential health implications. Beyond the issues of their sustainable production and increasing consumer acceptance, the true value of mycoproteins lies in their biochemical composition and physiological effects. In order to fully understand their role as a viable alternative protein source, it is essential to understand in depth the potential health benefits that lie in their macro- and micronutrient profile, as well as any safety considerations.
The following subchapter reviews the basic nutritional aspects of mycoproteins in modern diets, presenting the potential benefits of consuming mycoprotein-based products, along with safety considerations in the case of particular allergies or immunological sensitivities, in order to aid in research prioritization and maximization of the potential mycoproteins hold as sustainable “functional foods”.
3.1. Nutritional benefits of mycoproteins
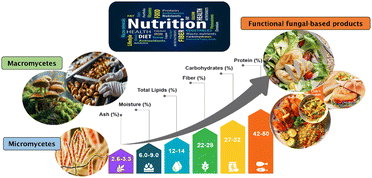
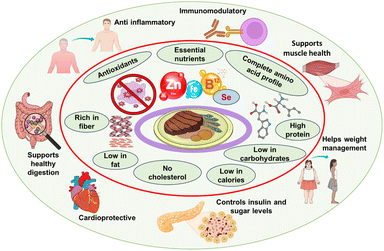
The nutritional profile of mycoproteins positions them between traditional animal- and plant-derived protein sources, combining the advantages of both categories. With a protein content typically exceeding 45% of the biomass dry weight, mycoproteins provide a complete and well-balanced essential amino acid profile, similar to animal proteins. They are particularly rich in lysine and methionine—amino acids often limited in grain-based diets, making them suitable for supporting essential physiological functions, including the maintenance of muscle mass (Fig. 3).71,72In addition, mycoproteins are rich in dietary fibre, mainly β-glucans and chitin, which have been associated with supporting the gastrointestinal health, the modulation of glycemic response and increased satiety. Compared with animal proteins, mycoproteins have an advantageous lipid profile, characterized by a low content of saturated fats and the absence of cholesterol, which makes them particularly suitable for populations at risk of cardiovascular disease.73 Comparative studies have shown that replacing red meat in the diet with mycoproteins can improve lipid metabolism and insulin sensitivity, while maintaining sensory satisfaction, highlighting their potential role in the prevention of chronic diseases.74 Furthermore, recent clinical and epidemiological studies strengthen the argument for the beneficial effects of mycoprotein consumption on improved plasma lipid and glycemic profiles, along with reduced postprandial insulin response,75,76 supporting its role in managing obesity, type 2 diabetes, and cardiovascular disease. These metabolic benefits are attributed to its low glycemic index and fibre-mediated modulation of the gut microbiota, which collectively improve metabolic homeostasis.
Beyond their macronutrient profile (Table 2), mycoproteins contain micronutrients and bioactive compounds that further enhance their functional properties, such as B-complex vitamins (B1, B2, B3, folate), trace elements such as riboflavin, zinc and selenium, and antioxidant compounds capable of mitigating oxidative stress.77
| Content | Fungal based protein | Plant based protein | Animal based protein | ||
|---|---|---|---|---|---|
| Mycoprotein | Soybean | Beef | Chicken | Pork | |
| a PDCAAS – protein digestibility-corrected amino acid score. | |||||
| Essential amino acids (EAAs) (g) | |||||
| Protein (g) | 11.25 | 14.00 | 20.20 | 24.00 | 21.80 |
| Histidine | 0.35 | 0.60 | 0.66 | 0.72 | 0.89 |
| Isoleucine | 0.52 | 1.10 | 0.87 | 1.22 | 1.06 |
| Leucine | 0.86 | 1.80 | 1.53 | 1.82 | 1.83 |
| Lysine | 0.83 | 1.40 | 1.60 | 1.39 | 2.05 |
| Methionine | 0.21 | 0.30 | 0.50 | 0.68 | 0.60 |
| Phenylalanine | 0.49 | 1.10 | 0.76 | 1.17 | 0.91 |
| Tryptophan | 0.16 | 0.30 | 0.22 | 0.19 | 1.03 |
| Threonine | 0.55 | 0.80 | 0.84 | 0.71 | 0.28 |
| Valine | 0.62 | 1.10 | 0.94 | 1.21 | 1.24 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| Micronutrients | |||||
| Iron (mg) | 0.50 | 3.00 | 3.50 | 0.40 | 0.80 |
| Zinc (mg) | 9.00 | 0.90 | 0.40 | 0.70 | 2.10 |
| Sodium (mg) | 5.00 | 1.00 | 43.00 | 60.00 | 63.00 |
| Selenium, µg | Selenium, µg | 5 | 1.00 | 12.00 | 13.00 |
| Vitamin B12 (µg) | Trace | Trace | 0.40 | Trace | Trace |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| Macronutrients | |||||
| Total carbohydrate (g) | 3.00 | 5.10 | 0.06 | Trace | Trace |
| Sugar (g) | 0.50 | 1.64 | Trace | Trace | Trace |
| Fat (g) | 2.90 | 7.30 | 4.30 | 1.10 | 4.00 |
| Saturated fatty acid (SFA) | 0.60 | 0.90 | 1.70 | 0.30 | 1.40 |
| Dietary fiber (g) | 6.00 | 6.10 | 0 | 0 | 0 |
| Digestibility | 0.996 | 1.00 | 1.00 | 1.00 | 1.00 |
| aPDCAAS | |||||
The hybrid nutritional profile makes mycoprotein a compelling alternative for consumers looking for sustainable, health-promoting protein sources without compromising on quality or functionality. Notably, the bioavailability of these constituent essential minerals and vitamins is enhanced by the absence of antinutritional factors that are usually found in certain plant proteins, such as lectins and phytates.81 Emerging studies suggest that the fermentation of these mycoproteins may further enhance their bioavailability, as fungal metabolic activity releases bound nutrients and generates bioactive peptides with potential antioxidant and immunomodulatory properties.82
As such, the combination of these nutrients makes mycoprotein a functional ingredient that not only meets basic dietary requirements, but also contributes to the maintenance of long-term health and disease prevention. All of these health benefits, along with a meat-like texture and versatile sensory properties, have facilitated consumer acceptance, especially among flexitarian and vegetarian populations looking for nutritious yet sustainable protein alternatives.
3.2. Health risks and safety considerations
Advances in engineering and synthetic biology have led to an acceleration of the optimization of fungal strains for improved productivity and nutritional performance.83 From a health and consumer perspective, mycoproteins have gained great interest in addressing contemporary dietary challenges related to excessive consumption of animal proteins. Research and epidemiological evidence consistently associate red and processed meat consumption with significantly increased risks of cardiovascular disease, colorectal cancer, and metabolic disorders.84Advances in the biotechnological production of mycoproteins for texturizing and flavour development through fermentation and extrusion have significantly improved consumer acceptance, allowing mycoprotein-based products to mimic the taste and mouthfeel of poultry, beef, or seafood analogues.85 The high satiety value and favourable lipid profile of mycoproteins make them particularly attractive for weight management and cardio-metabolic health, while their sensory similarity to meat facilitates dietary transitions to plant-based dietary patterns (Fig. 4).3
Although mycoproteins are generally considered safe by major regulatory authorities such as the European Food Safety Authority (EFSA) and the US Food and Drug Administration (FDA), and despite their nutritional benefits, their consumption can be associated with certain health risks for sensitive individuals. The most commonly reported adverse reactions are mainly allergic responses, mild gastrointestinal upset (bloating, flatulence, abdominal discomfort, in rare cases nausea or diarrhoea) and rarely, anaphylaxis.86 These effects are largely due to the chitin and fungal cell wall glycoproteins capable of triggering immune sensitization in susceptible consumers. Furthermore, as mycoprotein products are often extensively processed to achieve desired sensory characteristics, an assessment of the impact of processing on protein digestibility, amino acid oxidation, and potential formation of undesirable compounds is necessary.87 A limited number of cases of allergic reactions have been reported, with symptoms ranging from mild skin irritation or hives to, in very rare cases, anaphylaxis.
Since the introduction of Quorn's microprotein-based products, there have been several complaints from consumers reporting adverse gastrointestinal reactions after ingestion.
Tee et al. showed by cross-reactivity studies that mycoprotein Quorn has several allergenic factors in common with Aspergillus fumigatus, Cladosporium herharum and some with Alternaria alternata, thereby inducing a negative reaction in patients allergic to mold after inhaling or ingesting mycoproteins.88 Screening for specific IgE antibodies to mycoprotein (antigen 1) carried out on 12 workers in the mycoprotein production over a 2 year period showed that only 2 of them had a specific RAST binding more than 2%, but both of them without any symptoms. Other two of the 10 patients were admitted to hospital with vomiting and diarrhoea after ingesting mycoprotein, the skin prick tests and RAST measurements for all three mycoprotein antigens showing a swelling of 2 mm, but none had significantly elevated antibody levels.88
Another example is represented by a case report documented by Hoff et al., of an asthmatic patient with severe hypersensitivity reactions to ingested mycoprotein. In order to identify and characterize the potential responsible allergen, specific IgE antibodies from the patient's serum were used to screen a Fusarium culmorum cDNA expression library.89 The patient was shown to have high sensitization to several known aeroallergens, but, apart from Quorn, not to any other food samples tested. The patient had a history of respiratory sensitivity to mold allergens and subsequent oral ingestion of cross-reacting proteins led to severe food allergic reactions. The study revealed that the 60S acidic ribosomal protein P2 from F. venenatum was likely the cause of the patient's severe hypersensitivity reactions to Quorn mycoprotein, due to its potential cross-reactivity with the allergen Fus c 1 from F. culmorum.
A study conducted by Jacobson86 et al. based on the analysis of self-reports (online questionnaire https://www.quorncomplaints.org) of adverse reactions to Quorn mycoprotein, identified 1752 adverse reactions such as allergies and gastrointestinal symptoms after consuming Quorn products, with another 312 people reporting allergic reactions such as urticaria and anaphylaxis after 4 hours from mycoprotein consumption. The authors reported that of the 45.8% of reactions, 1 was fatal, within 1 hour of exposure, and 188 of the 312 people with allergic and gastrointestinal reactions experienced repeated reactions after repeated consumption of Quorn.86 The study found that Quorn-type mycoprotein-based foods caused gastrointestinal symptoms, including vomiting and diarrhoea, within 8 hours of consumption. Thus, the wide acceptance and distribution of mycoproteins in the food supply should be reconsidered as it was demonstrated that can cause numerous allergic and gastrointestinal reactions, some of them potentially life-threatening.
Beyond their nutritional advantages and health-related aspects, the growing interest in mycoproteins is also closely linked to their low environmental impact compared to conventional protein sources. Therefore, the following section examines the sustainability aspect associated with mycoprotein production systems.
4. Environmental impact and sustainability
According to the planetary boundaries framework, six of the nine boundaries have already been exceeded, in climate change, biosphere integrity, land-system change, freshwater change and biogeochemical flows, highlighting an increasing pressure on the stability and safety of our planet (Stockholm Resilience Centre). Conventional livestock farming is estimated to account for up to 80% of the negative environmental impact of agriculture in the form of greenhouse gas emissions, biodiversity loss, land conversion and intensive agrochemical use. For this reason, it is important to identify and implement food practices and dietary changes with a lower environmental impact and benefits for the consumer. Plant-based alternatives to conventional meat products have increased in popularity and demand in the past decades, being promoted as healthier, cruelty-free and sustainable.90 However, numerous studies indicate that there are still several drawbacks to be taken into consideration. Extensive land use and land conversion required for both livestock feeding as well as plant-based products exerts a significant impact on local biodiversity, affects global biogeochemical cycles and climate patterns such as temperature and precipitation, as well as decreases environmental stability to stressors.91Another crucial aspect of interest regarding plant-based proteins has been genetically engineering crops for the purpose of increasing yield, quality and tolerance to environmental factors. Up to 90% of all genetically modified crops are represented by soy, cotton, maize and canola. Although genetically modified crops bring agricultural, economic and nutritional benefits, the main concerns lie with the transfer of resistant genes from transgenic crops to wild varieties, thus reducing genetic diversity, as well as promoting the evolution of pesticide-resistant weeds, disrupting the soil microbiota and leading to biological diversity in insects and plant species, ultimately affecting ecosystem stability.92 Moreover, monoculture practices are prevalent for staple crops used in plant-based products, which significantly affect biodiversity, soil fertility and structure and may require more pesticides, agrochemicals and water, as well as intensive use of machinery and fossil fuels.93
In the light of these disadvantages, one of the main arguments in favour of mycoproteins is their low environmental impact. The general consensus is that mycoproteins require lower costs and resources for production, while containing essential amino acids, fibres, vitamins, antioxidants and carbohydrates, with a lower carbon and water footprint, as well as significantly less land use than the requirements for animal products.94,95 According to Quorn®, mycoprotein based products produce 80% less GHG emissions, 60% less land use and 65% less water compared with livestock farming, representing even less than other plant-based alternatives such as soybeans and peas (Fig. 5).96 Furthermore, various waste or industrial by-products have been successfully used as substrates for the production of mycoproteins, offering economic and environmental benefits, in the light of a circular economy approach.97–99
Research independent from Quorn®'s own data on the environmental impact of mycoproteins is still scarce and currently, opinions are divided. Firstly, the functional unity for comparison between food products varies, such as kg of product, kg of protein, or kg of protein corrected by digestibility score, thus creating variability throughout studies.100 In that sense, the most significant differences between products analysed by LCA are when mass is replaced by g of proteins as functional unit. Accordingly, using protein digestibility as a complementary unit presented a lower impact on LCA results compared to the other functional units and better accuracy.101 Secondly, methodological variations such as formulation inputs, processing energy, waste management and byproduct valorisation offer different results which influence the conclusions on mycoprotein benefits in comparison with conventional food sources. Various life-cycle analyses (LCA), assessing the sustainability from cradle-to-factory of mycoproteins, reveal contradictory results.
A LCA conducted by Shahid et al. observed the differences in greenhouse gas emissions, land use and freshwater use between plant-based products, mycoprotein-based products and meat products.10 Mycoprotein-based products presented significantly lower values compared with meat and meat-based products; however, completely replacing meat with either mycoproteins or other plant-based alternatives did not significantly reduce freshwater use. Additionally, an important point has been made by Humpenöder et al. regarding potentially replacing meat with microbial protein at large scale.102 Animal by-products not directly used for human consumption, such as pet food, leather products, chemical products and fertilizer would also need to be replaced with synthetic alternatives, which may generate additional GHG emissions and further environmental impacts. Moreover, the production of mycoprotein-based meat alternatives can still present an increased environmental footprint, primarily due to the land and water demands for the raw materials, along with the use of fertilizers used in crop cultivation that serve as substrates. Upcraft et al. analysed the sustainability of producing mycoproteins derived from lignocellulosic residues, in the form of rice straw.72 Results obtained were higher than beef, chicken and tofu in terms of water consumption, and comparable with chicken regarding GHG emissions and freshwater eutrophication. For these effects, the main contributing factors were concluded to be caused straw production, involving the irrigation of rice fields, electricity use and production particularities. Other studies indicate that using glucose, egg white albumin or other by-products, significantly increases the global warming potential of mycoproteins, with effects similar to pork and chicken.103,104
In that sense, although plant-based alternatives present numerous benefits compared with livestock farming, when envisioning a complete shift from traditional practices towards more sustainable, green alternatives, several inputs and outputs must be taken into consideration. Significantly decreasing large-scale production and consumption of animal-based products presents environmental and health benefits, however, balance and optimization of the desired processes and sectors is important for the proper implementation of global sustainable objectives.
Mycoproteins represent promising options for a more sustainable and efficient society, yet research is still needed in order to obtain products with a notably lower impact on the environment, compared with conventional and alternative protein sources. As the alternative protein market continues to evolve, its consolidation depends not only on technological advancement, but also on a uniform regulatory framework governing production, safety assessment, labelling and market authorization, to ensure safety and well-informed choices for the consumers. In this context, the following section touches the current legislative landscape and regulatory considerations, relevant for the commercialization of mycoprotein-based products.
5. Legislative considerations and compliance requirements for mycoprotein-based foods
Meat analogues have to undergo strict food regulations in order to be available on the market. In EU countries, mycoproteins along with other alternative proteins were first regarded as novel foods within the Novel Food Regulation (EC/258/1997), requiring pre-market approval based on risk assessment regarding the safety and nutritional risks of the products.105 The main criteria for food to be considered novel was the absence of consumption to a significant degree within EU member states before 1997. Mycoproteins are not specifically mentioned in this regulation, however they fall within the definition of novel foods, as “foods and food ingredients consisting of, or isolated from microorganisms, fungi or algae” (Regulation (EU) 2015/2283).106 Since then, regulation 2015/2283 replaced EC 298/97 in 2018, offering a centralized EU list of authorized novel foods, and since 2024, the European Commission concluded that mycoproteins derived from the Fusarium venenatum strain A3/5 and produced under the same aerobic fermentation process as Quorn® qualify as traditional food, not requiring novel food authorization due to its long history of consumption before 1997 (ARTICLE 4 REQUEST Regulation (EU) 2015/2283).106Besides commercialization control, labelling is also strictly regulated. Claims such as “high in protein”, “source of fibre” or “source of vitamins and minerals” must be met by specific quantitative thresholds. Health claims must be sustained by generally accepted scientific data and justified by the business operator making these claims. While mycoproteins are not directly mentioned in these regulations, they fall within the definition of “food” accepted within the regulation, meaning any substance or product intended to be ingested by humans (Regulation (EC) No 178/2002;107 Regulation (EC) No 1924/2006).108 It is important to mention that mycoproteins are not included in the EU list of allergens (Regulation (EU) No 1169/2011),109 and including such information on mycoprotein-based product labels is not mandatory.
In the United States, mycoproteins are regulated by the Food and Drug Administration (FDA) under the Generally Recognized as Safe (GRAS) framework since 2002.110 Marlow Foods Ltd, the founder of Quorn™, submitted a notification in 2001 for the recognition of mycoproteins as safe for general food use, allowing the introduction of several mycoprotein based-products into the food supply chain (FDA, 2001).110 Under the Federal Food, Drug, and Cosmetic Act, all food ingredients must meet general requirements for safety, GRAS status being acquired based on generally accepted scientific data and substantial history of consumption.111 The GRAS system functions on a voluntary informing mechanism, in which individuals submit notifications for the approval of foods and food ingredients as GRAS, exempting them from pre-market approval, while having to meet common safety standards.112 Similarly to European law, mycoproteins are not explicitly listed or defined in food labelling law, however they undergo the same requirements as common packaged foods for sustained nutritional and health claims as well as clear ingredient listing (US Code of Federal Regulations, 1977).113 Mycoproteins are not included in the principal list of allergens, however precautionary labelling is encouraged, due to the increased number of allergic reactions reported by consumers over the years, the most severe one being anaphylaxis.86
Currently, mycoproteins products are available in European countries such as the United Kingdom, Denmark, Norway, France, Germany and Switzerland, Asian countries such as Singapore, Thailand and China, as well as the United States and Canada.29 The global market for fungal-based products is expected to grow, with numerous countries potentially introducing novel products on the market and new fungal strains being tested besides Fusarium venenatum.16 Despite growing interest in alternative proteins, there is still a lack of clear guidelines that align across countries, both in terms of consumer safety, as well as fair trade between companies.114
Overall, current regulations continue to evolve, demonstrating both the growing recognition of mycoproteins as safe and valuable food ingredients and the need for continued alignment of standards to support innovation, consumer protection, and global market expansion. As regulatory bodies continue to adapt to the rapid development of the alternative protein sector, new opportunities and demands arise for researchers, producers and policymakers alike. Therefore, the following section will discuss the future perspectives and research directions that further support the development. Scalability and global recognition of mycoprotein-based foods, as well as limitations and gaps in knowledge that still need to be addressed.
6. Future perspectives
Given the continued evolution of research and industrial interest in mycoproteins as a protein source, it is evident that it is important to explore emerging opportunities that could shape their development. Also, consumer expectations and legislative frameworks regulating the use of mycoproteins as a source of protein in foods will likely influence how mycoproteins integrate into global food markets (Fig. 6).Advances in biotechnology, fermentation engineering, and sustainable food systems offer new perspectives for improving production efficiency, nutritional quality, and product diversification.
This section presents the main trends, challenges and perspectives that will define the evolution of mycoprotein-based foods.
6.1. Artificial intelligence-driven innovation
Artificial intelligence (AI) and machine learning (ML) can be used to enable data-driven decision-making, automation and optimization in order to allow advancements in biotechnological sectors both in research & development as well as market. In the alternative protein sector, these technologies can tackle issues regarding safety, consistency and efficiency. Eco-innovative food products have been successfully developed and commercialized with the use of AI, one example being the plant-based alternatives by NotCo™. Yet, its full potential to enhance nutritional functionality, sustainability and consumer relevance is still insufficiently explored.115 In mycoprotein development, AI and ML can reduce costs, minimize risks and advance the development of more sustainable and high-quality products.116As new fungal strains are explored for mycoprotein production, several challenges need to be addressed, such as the selection of safe strains, evaluating their potential in protein production and assessing the optimal growth and development conditions in laboratory or bioreactor settings. In that sense, the operational setpoints can be explored through AI-driven optimization methods, which can enable the development of robust up-scaling with fewer resources and time required.117 ML algorithms trained on genomic and metabolomic data can identify key biomarkers for food safety and improved production. Incorporating selection methods, such as random forests or decision trees, can help identify genes linked to toxin synthesis, as well as metabolic pathways associated with specific enzymatic function, amino acid profiles or probiotic activity.118
Furthermore, AI-based frameworks can define optimal growth and fermentation conditions, while uncovering complex, nonlinear relationships between bioprocess parameters, by integrating data from multi-omics and techno-economic models. Techno-economic assessments (TEAs) can identify attributes that are impactful to capital and operating costs, thus allowing the evaluation of feasibility, scalability and profitability of the production process. However, many of them utilize simulation software's that require economic investments, proper training in order to be used, and exploring the multiple scenarios tailored to process particularities can be time-consuming as well.119 AI/ML tools can be paired with classical optimization models such as Design of Experiments (DOE) and Bayesian Optimization (BO) in order to obtain technologies with higher accuracy, efficiency and flexibility.120 Process control can also become adaptive and autonomous through reinforcement learning (RL), allowing real-time closed-loop control of key parameters in order to maximize product output.121 Additionally, predictions can be achieved through virtual models such as digital twins (DT) that simulate in silico scenarios, thus predicting the effect of changes in parameters or scale, on productivity and composition.122
AI can also be integrated into genetic engineering tools to efficiently improve protein yields while minimizing metabolic burden, reduce the time required for design cycles and identify biological pathways for synthesizing antioxidants and other bioactive compounds responsible for flavor, texture and nutritional enhancement. In this way, the design of more appealing and sustainable products that align with industrial efficiency requirements, consumer health needs and environmental responsibility.123 Although not yet applied directly for mycoprotein production, many of the elements that can be transferable towards its development have been demonstrated, such as ML for Clustered Regularly Interspaces Short Palindromic Repeats (CRISPR) design, accurate genotype to phenotype mapping, autonomous design-build-test-learn (DBTL) cycles and synthetic biology toolkits for enhancing specific traits such as nutritional value and sensory appeal.124–126 The applicability of genetic engineering for mycoprotein development will be treated in more detail in the following subsection.
6.2. Genetically engineering the pathway towards mycoprotein innovation
Synthetic biology and genetic engineering tools can be used to significantly improve the use of microorganisms as cell factories for numerous applications in biotechnology. Both the fermentation process as well as the traits of the strains used can be enhanced and tailored for targeted purposes through genetic manipulation.127 Advancement strategies regarding mycoprotein production generally focus on increasing biomass productivity, improving protein quality and yield, digestibility and bioavailability, securing food safety and enhancing substrate use for new side streams, while minimizing the environmental impact of the fermentation processes.128Conventional strain improvement techniques focus on enhancing microbial metabolic performance through physical and chemical mutagenesis, adaptive evolution, and random recombination by protoplast fusion or genome shuffling, for the production of interspecific or intergeneric hybrids. These practices have been used to improve microbial fitness in terms of biomass production, substrate utilization, adaptation and production yields for various traits and biomolecules of biotechnological importance. However, these methods can be time consuming, laborious, with limited precision and genetic instability.129,130
Synthetic biology tools offer targeted solutions to these limitations by enabling precise and effective editing of genomic sequences in order to alter targeted functions, expression systems or regulatory factors.131 In the context of mycoprotein development, the general consensus recognizes the difficulty of employing certain gene editing procedures for Fusarium venenatum, as a result of the strain's low transformation frequency and low rate of homologous integration.132,133 One of the most widely recognized gene editing platforms, CRISPR/Cas, has been used in other Fusarium strains, such as F. fujikuroi, F. graminearum and F. proliferatum, to enhance the production of gibberellic acid, which plays an important role in plant growth regulation,134 or aurofusarin, a pigment with potential antimicrobial properties.135 Additionally, the polyketide synthase gene (FUM1) responsible for the production of the mycotoxin fumonisin in F. proliferatum was successfully deleted through a modified CRISPR/Cas9 system, with reduced risk of off-target mutation.136
For Fusarium venenatum, the fibrous cell wall rich in β-glucans and chitin control protein bioavailability and digestibility.137 Therefore, the alteration of mycelial structure could influence nutrition and digestive behaviour. For example, through integrated transcriptomics and proteomics techniques, it was observed that mycoprotein yield, extractability and digestibility can be significantly increased in F. venenatum by deleting chitin synthases (Chs) from its genetic code, thus reducing cell wall rigidity.138 Similarly, Hong et al. identified that through knockout of the FvChs2 gene, a significant reduction in the chitin content of the cell wall was also associated with mycelial expansion, decreased ethanol production during fermentation, with a higher carbon conversion efficiency, upregulated nitrogen metabolism and increased protein production.139
Studies regarding direct mycoprotein optimization through genetic engineering mechanisms in the standard F. venenatum strain are still scarce, however the existent studies on other Fusarium strains could lay the foundation for further applications in targeted activation of genes involved in important metabolic pathways such as biosynthesis and resistance, or for the deactivation of genes responsible for mycotoxins and other allergens. Besides the Fusarium genus, other fungal genera are being studied for mycoprotein synthesis, that may also be suited for improvement through genetic engineering and represent a novel opportunity for expanding research and development in this sector.
Aspergillus oryzae is considered a promising candidate, being extensively used in industrial-scale enzyme production and recognized as GRAS status by the FDA and WHO. Aflatoxin production is absent in A. oryzae, and no systemic infections have been reported in healthy individuals, with a limited number of reported allergic reactions.140 Genetic manipulation has been successfully applied to enhance the production of secondary metabolites and proteins through protoplast transformation, multiplexed CRISPR/Cas9 and Agrobacterium-mediated transformation, however the production capacity requires further improvement.141,142 Other possible strains with GRAS status, already integrated in bioprocesses of the food industry, include Aspergillus spp., Rhizopus spp., Neurospora spp., Penicillium spp.,143 which have been subjected to genetic engineering in order to improve enzyme and secondary metabolites production, protein expression or biomass accumulation.144,145
In summary, the integration of genome editing into alternative protein development represents an important step towards higher-quality, safer and more sustainable products. As genome editing and synthetic biology techniques continue to evolve alongside the global interest in alternative protein sources, future research has the potential to accelerate the development of safer, more precisely optimized, higher-quality fungal-based products that contribute to a more sustainable and resource-efficient food system.
6.3 Gaps in knowledge to be addressed
The full potential of fungal-based proteins is still not sufficiently explored and large-scale, long-term studies are necessary to properly assess systemic effects of mycoprotein consumption. Studying more non-model, food-safe fungal strains with potential high yield, nutritional value or health benefits is promising in terms of revolutionizing mycoprotein production.146 For instance, mycoproteins obtained from the fermentation of edible fungi and food-industry by-products were recently discovered to possess antioxidant and anti-inflammatory properties.147 Additionally, there is limited data on the thermal and chemical stability of various bioactive compounds with known antiviral, anti-tumoral and antimicrobial properties, yet it is not known if they may be retained in the final products after industrial processing.148Furthermore, the long-term metabolic and immunologic effects of mycoprotein consumption are not sufficiently studied. A meta-analysis of 9 randomized control trials (178 participants) revealed that mycoprotein consumption reduces total cholesterol, with a significant effect on LDL cholesterol. Notably, the authors highlight the need for larger-scale, randomized trials to support their findings.149 Moreover, the mechanistic understanding of glycaemic control and energy intake regulation exerted by mycoprotein consumption is still quite scarce, and not explored in extensive human trials.150 Besides the high-fibre content of mycoprotein products which may pose several health benefits,79 recent studies indicate that the unique fibre profile of mycoproteins, an insoluble matrix comprised of β-glucan and chitin may be more influential in modulating the digestive and metabolic properties of mycoproteins than its total amount.151 It is important to mention that we could not find clinical trials enrolling immunocompromised individuals or chronically ill patients in the context of potential effects of consuming fungal-based products.
The effects of mycoproteins on intestinal microbiome are also still insufficiently understood. In vitro studies indicate a shift towards β-glucans degrading taxa such as Bacteroides, which in murine models have been shown to improve gut barrier function, reduce gut epithelial inflammation and modulate bile acid profiles.137,152 A higher microbial diversity may be associated with a high-fibre diet and consequently with a lower colorectal cancer risk. A recent study conducted by Farsi et al. compared the effects of mycoprotein consumption with those of red and processed meat on intestinal heath markers and genotoxins production.153 A test period of two weeks revealed an increase in the relative abundance of microbial genera such as Lactobacillus, Akkermansia and Roseburia, which may be linked to improved gut health and metabolic function. Furthermore, an increased consumption of mycoproteins reduced faecal water genotoxicity, a surrogate risk marker for carcinogenicity. However, there is a lack of long-term human studies connecting mycoprotein intake with shifts in gut microbiome composition.
Despite the decades-long history of studies on mycoprotein production, research is still limited regarding the broader bioactive and therapeutic potential of fungal-based products, including the stability and functionality of strain-specific compounds, as well as their long-term physiological, metabolic, immunological and microbiome modulatory effects. In that sense, there is continuous research potential for medical, pharmaceutical and biotechnological applications.
7. Conclusions
The transition of nutrition and cultural trends towards more environmentally-conscious and healthy options plays a significant role in the rising interest for mycoprotein products. As more people move toward vegetarian or flexitarian eating habits, the market for alternative proteins continues to expand. Younger consumers (18–34) tend to be more open to mycoprotein-based foods compared with older generations, according to the Good Food Institute,154 focusing on the perceived health advantages, ethical sourcing, and sustainability benefits. However, acceptance still varies widely by region, driven by differences in culinary traditions, varying levels of familiarity, and regulatory environments. These regional differences underscore the need for more transparent communication between researchers, decision-makers and consumers in order to align regulatory and safety standards, along with improvements in public outreach.As a novel food derived from filamentous fungi, mycoproteins pose particular biochemical traits that may have potential health benefits that are still not sufficiently explored through long-term, broad clinical trials. Furthermore, allergenic or metabolic effects of new strains and substrates require more in depth attention.
Emerging production practices and innovative technologies that integrate synthetic biology or artificial intelligence tools targeted towards optimized fermentation, strain development, and product design can address existing limitations and support the growing role of mycoproteins in future food systems.
In conclusion, mycoproteins represent a promising option as alternative proteins, highlighted by their nutritional profile, sustainable production methods, which can be considered a promising solution to major global challenges—including food security, environmental pressures, and public health.
Author contributions
M. D., A.-M. G. and C. F. conceptualization of the manuscript. M. C., C. F. and A.-M. G. drafted the original manuscript. M. C., I. R, L. G. Z. and M. L. J. data curation, visualization and editing. M. C., C. F. and A.-M. G. writing the original manuscript. I. R., C. F. and A.-M. G. prepared the figures. A.-M. G. and M. D. critically revised the manuscript. All authors read and approved the final manuscript.Conflicts of interest
The authors declare no conflicts of interest, nor competing financial interests.Data availability
No primary data were generated in this study. All information supporting this review are derived from previously published studies, which have been cited in the reference list.Acknowledgements
This work is supported by a grant of the Ministry of Research, Innovation and Digitization, CCCDI – UEFISCDI, through project number PN-IV-P7-7.1-PED-2024-1277-SafeBioChain and within PN 23.06 Core Program—ChemNewDeal, project no PN23.06.01.01-AQUAMAT. Figures created in https://biorender.com/ and Mind the Graph.Notes and references
- T. Hara, in International Handbook of Population Policies, ed. J. F. May and J. A. Goldstone, Springer International Publishing, Cham, 2022, pp. 759–780, DOI:10.1007/978-3-031-02040-7_35.
- T. Gebre, Z. Abraha, A. Zenebe and W. Zeweld, Nutr. Food Sci., 2024, 55, 275–293 CrossRef.
- S. Kumar, G. Kathuria and D. Malhotra, Int. J. Consum. Stud., 2025, 49, e70026 CrossRef.
- I. Chowdhuri and S. C. Pal, Soil Till. Res., 2025, 248, 106442 CrossRef.
- OECD-FAO Agricultural Outlook 2025-2034, OECD | Food and Agriculture Organization of the United Nations, Paris and Rome, 2024.
- United Nations Environment Programme (2024), Emissions Gap Report 2024: No more hot air … please!, 2024, Nairobi, 2024, https://www.unep.org/resources/emissions-gap-report-2024.
- Good Food Institute, Alternative proteins public investment database, Good Food Institute, https://gfi.org/resource/public-investment-database/?_gl=1*19xni5q*_up*MQ..*_ga*MTQ4MjMyMTE1NC4xNzUyMDU2OTMy*_ga_TT1WCK8ETL*czE3NTIwNTY5MjkkbzEkZzEkdDE3NTIwNTY5MjkkajYwJGwwJGgw, (accessed September 2025).
- Italy teams up in Brussels with France and Austria against lab-grown meat, https://www.eunews.it/en/2023/12/11/fumata-nera-sulle-nuove-tecniche-genomiche-a-bruxelles-lollobrigida-fa-squadra-con-austria-e-francia-contro-il-cibo-coltivato/, (accessed September 2025).
- F. R. P. Cedeno, O. J. Olubiyo and S. Ferreira, J. Biol. Eng., 2025, 19, 44 CrossRef CAS.
- M. Shahid, P. Shah, K. Mach, B. Rodgers-Hunt, T. Finnigan, G. Frost, B. Neal and M. Hadjikakou, Future Foods, 2024, 10, 100410 CrossRef CAS.
- R. Gnaim, P. S. Dyer and R. Ledesma-Amaro, Trends Food Sci. Technol., 2025, 159, 104981 CrossRef CAS.
- E. Lurie-Luke, Nat. Commun., 2024, 15, 4425 CrossRef CAS PubMed.
- S. Nájera Espinosa, G. Hadida, A. Jelmar Sietsma, C. Alae-Carew, G. Turner, R. Green, S. Pastorino, R. Picetti and P. Scheelbeek, Nutr. Rev., 2024, 83, e1626–e1646 CrossRef.
- J. Sun, V. Caputo and H. Taylor, Choices, 2025, 40, 1–15 Search PubMed.
- The 17 Goals - 17 Sustainable Development Goals (SDGs), https://sdgs.un.org/goals, (accessed September 2025).
- X. Wang, Y. Pei, J. Wu, X. Zhong, H. Liu and Y. Xue, Compr. Rev. Food Sci. Food Saf., 2025, 24, e70166 CrossRef CAS PubMed.
- X. Llanaj, G. Törős, P. Hajdú, N. Abdalla, H. El-Ramady, A. Kiss, S. Ø. Solberg and J. Prokisch, Foods, 2023, 12, 2671 CrossRef CAS PubMed.
- V. Meyer, E. Y. Basenko, J. P. Benz, G. H. Braus, M. X. Caddick, M. Csukai, R. P. de Vries, D. Endy, J. C. Frisvad, N. Gunde-Cimerman, T. Haarmann, Y. Hadar, K. Hansen, R. I. Johnson, N. P. Keller, N. Kraševec, U. H. Mortensen, R. Perez, A. F. J. Ram, E. Record, P. Ross, V. Shapaval, C. Steiniger, H. van den Brink, J. van Munster, O. Yarden and H. A. B. Wösten, Fungal Biol. Biotechnol., 2020, 7, 5 CrossRef.
- M. L. Chikindas, D. W. Schaffner and S. D. Todorov, Mol. Nutr. Food Res., 2025, 69, e70022 CrossRef CAS.
- J. Lonchamp, K. Stewart, C. D. Munialo, L. Evans, M. Akintoye, S. Gordon, P. S. Clegg, N. Willoughby and S. R. Euston, Food Chem., 2022, 396, 133736 CrossRef CAS.
- C. Landeta-Salgado, P. Cicatiello and M. E. Lienqueo, Algal Res., 2021, 56, 102314 CrossRef.
- P. F. Souza Filho, R. B. Nair, D. Andersson, P. R. Lennartsson and M. J. Taherzadeh, Fungal Biol. Biotechnol., 2018, 5, 5 CrossRef.
- S. F. Seyed Reihani and K. Khosravi-Darani, Appl. Biotechnol. Food Sci. Pol., 2019, 5, 243–352 Search PubMed.
- J. Liang, N. Xu, A.-K. Nedele, M. Rigling, L. Zhu, Y. Zhang, F. Stöppelmann, L. Hannemann, J. Heimbach, R. Kohlus and Y. Zhang, J. Agric. Food Chem., 2023, 71, 9070–9079 CrossRef CAS.
- M. Abbasi-Riyakhuni, S. S. Hashemi, R. S. Alavijeh, S. Mojoodi, A. Shavandi, O. V. Okoro, M. Tabatabaei, M. Aghbashlo, J. F. M. Denayer and K. Karimi, Process Saf. Environ. Prot., 2024, 190, 123–134 CrossRef CAS.
- R. Majumder, S. Miatur, A. Saha and S. Hossain, Sustain. Food Technol., 2024, 2, 81–91 RSC.
- C. s. r. Alicia Gil-Ramírez, in Mushrooms. Cultivation, Antioxidant Properties and Health Benefits, ed. G. Pesti, Nova Science Publishers, 2014, ch. 2, pp. 43–74 Search PubMed.
- Z. Y. Ng, P. E. Kee, R. Abdullah, J. C.-W. Lan, T. C. Ling, J.-J. Jiang, J. W. Lim and K. S. Khoo, Biomass Convers. Biorefin., 2024, 15, 31337–31363 CrossRef.
- R. Khan, F. H. Brishti, B. Arulrajah, Y. M. Goh, M. H. Abd Rahim, R. Karim, S. Hajar-Azhari, S. Kin Kit, F. Anwar and N. Saari, Int. J. Food Sci. Technol., 2023, 59, 522–544 CrossRef.
- M. A. Lizardi-Jiménez and R. Hernández-Martínez, 3 Biotech, 2017, 7, 44 CrossRef.
- S. Borkertas, J. Viskelis, P. Viskelis, P. Streimikyte, U. Gasiunaite and D. Urbonaviciene, Fermentation, 2025, 11, 351 CrossRef CAS.
- M. Perwez and S. Al Asheh, Biotechnol. Rep., 2025, 45, e00873 CrossRef CAS.
- L. Cao, Y. Chen, H. M. E. Mashad, Z. Pan and R. Zhang, Food Bioprocess Technol., 2026, 19, 112 CrossRef CAS.
- E. Voutilainen, V. Pihlajaniemi and T. Parviainen, Bioresour. Technol. Rep., 2021, 14, 100683 CrossRef CAS.
- M. Cebrián and J. Ibarruri, in Current Developments in Biotechnology and Bioengineering, ed. M. J. Taherzadeh, J. A. Ferreira and A. Pandey, Elsevier, 2023, pp. 251–292, DOI:10.1016/B978-0-323-91872-5.00003-X.
- A. Artola, X. Font, J. Moral-Vico and A. Sánchez, Front. Chem. Sci. Eng., 2024, 6, 1463785 CrossRef.
- P. Sentís-Moré, M.-P. Romero-Fabregat, C. Rodríguez-Marca, A.-J. Guerra-Sánchez and N. Ortega-Olivé, Fermentation, 2023, 9, 921 CrossRef.
- MyForest Foodshttps://www.myforestfoods.com/, (accessed September 2025).
- E. Bayer and G. McIntyre, Method of forming a mycological product, US Pat., US2020/0055274-A1, 2020 Search PubMed.
- NovoNutrients, https://www.novonutrients.com/, (accessed September 2025).
- Prime Roots - The Clean Plant Protein, The Clean Deli Meat, https://www.primeroots.com/en-us/pages/about-us, (accessed September 2025).
- New Culture, https://www.newculture.com/, (accessed September 2025).
- X. Feng, K. Ng, S. Ajlouni, P. Zhang and Z. Fang, Food Rev. Int., 2024, 40, 2580–2617 CrossRef CAS.
- P. Sentís-Moré, M.-P. Romero-Fabregat, C. Rodríguez-Marca, A.-J. Guerra-Sánchez and N. Ortega-Olivé, Fermentation, 2023, 9(10), 921 CrossRef.
- M. A. Manan and C. Webb, Am. J. Biochem. Biotechnol., 2024, 20 Search PubMed.
- G. Bakratsas, A. Polydera, O. Nilson, A. V. Chatzikonstantinou, C. Xiros, P. Katapodis and H. Stamatis, Foods, 2023, 12, 2295 CrossRef CAS.
- F. Hashempour-Baltork, K. Khosravi-Darani, H. Hosseini, P. Farshi and S. F. S. Reihani, J. Clean. Prod., 2020, 253, 119958 CrossRef CAS.
- G. M. Mascarin, P. S. Golo, C. de Souza Ribeiro-Silva, E. R. Muniz, A. de Oliveira Franco, N. N. Kobori and É. K. K. Fernandes, Appl. Microbiol. Biotechnol., 2024, 108, 451 CrossRef CAS.
- U. T. Dudekula, K. Doriya and S. K. Devarai, 3 Biotech, 2020, 10, 337 CrossRef.
- Monde Nissin Corporation, Annual Report of Monde Nissin Corporation Pursuant to Section 17 of the Securities Regulation Code, Monde Nissin Corporation, Republic of the Philippines, 2021, https://mondenissin.com/document_group/sec-form-17-a/.
- J. Connell, H. J. Bates, I. Geoghegan, F. Wilson, R. J. Harrison and R. J. Price, Fungal Biol. Biotechnol., 2025, 12, 3 CrossRef CAS PubMed.
- L. Porto de Souza Vandenberghe, A. F. Murawski de Mello, C. Matte Borges Machado, G. Biagini, P. B. Gruening de Mattos, I. Negreiros Piazenski, J. P. Manica Candelario and C. R. Soccol, Syst. Microbiol. Biomanuf., 2025, 5, 15–34 CrossRef CAS.
- F. Cerrone and K. E. O'Connor, Appl. Microbiol. Biotechnol., 2025, 109, 41 CrossRef CAS.
- F. Palladino, P. R. F. Marcelino, A. E. Schlogl, Á. H. M. José, R. de C. L. B. Rodrigues, D. L. Fabrino, I. J. B. Santos and C. A. Rosa, Appl. Sci., 2024, 14, 9346 CrossRef CAS.
- P. V. Devanthi, F. Pratama, I. T. Pramanda, M. D. Bani, A. D. Kadar and K. Kho, J. Fungi, 2024, 10, 555 CrossRef CAS PubMed.
- F. Pratama, R. T. Rahardja, A. R. Rachmadi, W. Q. Salam, K. Kho, A. Adelie and P. V. Devanthi, J. Fungi, 2025, 11, 349 CrossRef CAS.
- B.-H. Yang, T.-C. Lin, T.-W. Lin, S. Lin, I. C. Li, Y.-L. Chen, T.-J. Li and C.-C. Chen, Future Foods, 2024, 10, 100473 CrossRef CAS.
- R. Gmoser, C. Sintca, M. J. Taherzadeh and P. R. Lennartsson, Waste Manage., 2019, 97, 63–70 CrossRef CAS PubMed.
- J. Ibarruri, M. Cebrián and I. Hernández, J. Environ. Manage., 2021, 281, 111901 CrossRef CAS PubMed.
- J. P. López-Gómez and J. Venus, Fermentation, 2021, 7, 76 CrossRef.
- M. I. Ahmad, S. Farooq, Y. Alhamoud, C. Li and H. Zhang, Trends Food Sci. Technol., 2022, 121, 14–29 CrossRef CAS.
- J. R. Virués-Segovia, C. Pinedo, D. Zorrilla, J. Sánchez-Márquez, P. Sánchez, M. C. Ramos, M. de la Cruz, J. Aleu and R. Durán-Patrón, Front. Mar. Sci., 2024, 11, 1386175 CrossRef.
- P. Prakash, S. Namasivayam and S. S. Swetha Sunkar, J. Pharm., Biol. Chem. Sci., 2015, 6, 1251–1255 Search PubMed.
- A. Suparmin, T. Kato, H. Dohra and E. Y. Park, PLoS One, 2017, 12, e0187052 CrossRef.
- J. Ahlborn, A. Stephan, T. Meckel, G. Maheshwari, M. Rühl and H. Zorn, Int. J. Recycl. Org. Waste Agric., 2019, 8, 447–455 CrossRef.
- R. Gmoser, R. Fristedt, K. Larsson, I. Undeland, M. J. Taherzadeh and P. R. Lennartsson, Bioengineered, 2020, 11, 582–598 CrossRef CAS.
- F. Stoffel, W. d. O. Santana, J. G. N. Gregolon, T. B. L. Kist, R. C. Fontana and M. Camassola, Innov. Food Sci. Emerg. Technol., 2019, 58, 102227 CrossRef CAS.
- S. M. Hosseini, K. Khosravi-darani and J. Bioprocess, Biotechnology, 2011, 1, 1–5 Search PubMed.
- S. Tong, W. Chen, R. Hong, M. Chai, Y. Sun, Q. Wang and D. Li, J. Agric. Food Chem., 2024, 72, 604–612 CrossRef CAS PubMed.
- Quorn Nutrition, https://www.quornnutrition.com/mycoprotein/sustainable-nutrition, (accessed September 2025).
- D. C. Sands, C. E. Morris, E. A. Dratz and A. L. Pilgeram, Plant Sci., 2009, 177, 377–389 CrossRef CAS PubMed.
- T. Upcraft, W.-C. Tu, R. Johnson, T. Finnigan, N. Van Hung, J. Hallett and M. Guo, Green Chem., 2021, 23, 5150–5165 RSC.
- A. G. T. Niego, C. Lambert, P. Mortimer, N. Thongklang, S. Rapior, M. Grosse, H. Schrey, E. Charria-Girón, A. Walker, K. D. Hyde and M. Stadler, Fungal Divers., 2023, 121, 95–137 CrossRef CAS.
- D. N. Farsi, J. L. Gallegos, T. J. A. Finnigan, W. Cheung, J. M. Munoz and D. M. Commane, Eur. J. Nutr., 2023, 62, 3349–3359 CrossRef CAS.
- A. Cherta-Murillo, K. Zhou, M. Tashkova, J. Frampton, A. C. Cepas de Oliveira, C. Ho, G. Franco-Becker, E. S. Chambers, A. Dornhorst and G. S. Frost, Nutr. Diabetes, 2025, 15, 23 CrossRef CAS PubMed.
- G. F. Pavis, R. R. Iniesta, H. Roper, H. E. Theobald, E. J. Derbyshire, T. J. A. Finnigan, F. B. Stephens and B. T. Wall, Clin. Nutr., 2024, 43, 649–659 CrossRef CAS PubMed.
- K. Li, K. Qiao, J. Xiong, H. Guo and Y. Zhang, Foods, 2023, 12, 4388 CrossRef CAS PubMed.
- W. R. Aimutis and R. Shirwaiker, Proc. Natl. Acad. Sci. U. S. A, 2024, 121, e2319003121 CrossRef CAS PubMed.
- E. J. Derbyshire and J. Delange, Front. Sustain. Food Syst., 2021, 5, 581682 Search PubMed.
- T. J. A. Finnigan, B. T. Wall, P. J. Wilde, F. B. Stephens, S. L. Taylor and M. R. Freedman, Curr. Dev. Nutr., 2019, 3, nzz021 CrossRef PubMed.
- R. R. Holt, J. P. Munafo, Jr., J. Salmen, C. L. Keen, B. S. Mistry, J. M. Whiteley and H. H. Schmitz, J. Agric. Food Chem., 2024, 72, 2697–2707 CrossRef CAS PubMed.
- I. Giavasis, Curr. Opin. Biotechnol., 2014, 26, 162–173 CrossRef CAS.
- S. M. Mirsalami and M. Mirsalami, Future Foods, 2025, 11, 100601 CrossRef CAS.
- World Health Organization, International Agency for Research on Cancer, PRESS RELEASE N°, IARC Monographs evaluate consumption of red meat and processed meat, Lyon, France, 2015, https://www.iarc.who.int/wp-content/uploads/2018/07/pr240_E.pdf Search PubMed.
- L. C. Juhrich, M. Grosse, J. Mörlein, P. Bergmann, H. Zorn and M. Gand, J. Agric. Food Chem., 2025, 73, 2236–2248 CrossRef CAS.
- M. F. Jacobson, J. DePorter and A. . Allergy, Ann. Allergy Asthma Immunol., 2018, 120, 626–630 CrossRef CAS PubMed.
- D. Günal-Köroğlu, G. Karabulut, G. Ozkan, H. Yılmaz, B. Gültekin-Subaşı and E. Capanoglu, J. Agric. Food Chem., 2025, 73, 7522–7546 CrossRef.
- R. D. Tee, D. J. Gordon, J. A. Welch and A. J. N. Taylor, Clin. Exp. Allergy, 1993, 23, 257–260 CrossRef CAS.
- M. Hoff, R. M. Trüeb, B. K. Ballmer-Weber, S. Vieths and B. Wuethrich, J. Allergy Clin. Immunol., 2003, 111, 1106–1110 CrossRef PubMed.
- A. Ali and P. Bharali, Food Biosci., 2025, 68, 106640 CrossRef.
- X. Liu, P. Zhu, S. Liu, L. Yu, Y. Wang, Z. Du, D. Peng, E. Aksoy, H. Lu and P. Gong, Earth Syst. Dynam., 2024, 15, 817–828 CrossRef.
- M. N. Mohammad Amanullah and S. M. Zahid Hosen, Health Dyn., 2024, 1, 236–243 CrossRef.
- S. Mukhovi and J. Jacobi, Agric. Food Secur., 2022, 11, 19 CrossRef.
- K. R. Shivanna, J. Indian Bot. Soc., 2023, 103, 93–99 Search PubMed.
- J. Sillman, V. Uusitalo, V. Ruuskanen, L. Ojala, H. Kahiluoto, R. Soukka and J. Ahola, Int. J. Life Cycle Assess., 2020, 25, 2190–2203 CrossRef CAS.
- Quorn, Quorn Net Positive Report 2024 – Out Now, 2025, https://www.quorn.us/news/quorn-net-positive-report-2024-out-now.
- R. Aidoo, E. M. Kwofie, P. Adewale, E. Lam and M. Ngadi, Sci. Total Environ., 2024, 912, 169029 CrossRef CAS PubMed.
- L. Fernández-López, P. González-García, A. Fernández-Ríos, R. Aldaco, J. Laso, E. Martínez-Ibáñez, D. Gutiérrez-Fernández, M. M. Pérez-Martínez, V. Marchisio, M. Figueroa, D. B. de Sousa, D. Méndez and M. Margallo, Clean. Circ. Bioecon., 2024, 8, 100079 Search PubMed.
- B. Khoshnevisan, M. Tabatabaei, P. Tsapekos, S. Rafiee, M. Aghbashlo, S. Lindeneg and I. Angelidaki, Renew. Sustain. Energy Rev., 2020, 117, 109493 CrossRef CAS.
- P. F. Souza Filho, D. Andersson, J. A. Ferreira and M. J. Taherzadeh, World J. Microbiol. Biotechnol., 2019, 35, 147 CrossRef.
- U. Sonesson, J. Davis, A. Flysjö, J. Gustavsson and C. Witthöft, J. Clean. Prod., 2017, 140, 470–478 CrossRef.
- F. Humpenöder, B. L. Bodirsky, I. Weindl, H. Lotze-Campen, T. Linder and A. Popp, Nature, 2022, 605, 90–96 CrossRef PubMed.
- D. Y. Lee, E. Mariano, Y. Choi, J. M. Park, D. Han, J. S. Kim, J. W. Park, S. Namkung, Q. Li, X. Li, C. Venter and S. J. Hur, Food Sci. Anim. Resour., 2025, 45, 62–80 CrossRef.
- S. Smetana, A. Mathys, A. Knoch and V. Heinz, Int. J. Life Cycle Assess., 2015, 20, 1254–1267 CrossRef CAS.
- European Commision, Regulation (EC) No 258/97 of the European Parliament and of the Council of 27 January 1997 concerning novel foods and novel food ingredients, 1997, https://eur-lex.europa.eu/eli/reg/1997/258/oj/eng.
- European Commison, Consultation on mycoprotein (Fusarium venenatum). Food Safety, 2024, https://food.ec.europa.eu/document/download/4604819e-c38a-4204-91b2-f79dcbc9195e_en?filename=novel-food_consult-status_mycoprotein-fusarium-venenatum.pdf.
- European Parliament and Council, Regulation (EC) No 178/2002 of the European Parliament and of the Council of 28 January 2002 laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety, 2002, https://eur-lex.europa.eu/eli/reg/2002/178/oj/eng.
- European Parliament and Council, Regulation (EC) No 1924/2006 of the European Parliament and of the Council of 20 December 2006 on nutrition and health claims made on foods, 2006, https://eur-lex.europa.eu/eli/reg/2006/1924/oj/eng.
- E. P. a. Council, Regulation (EU) No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the provision of food information to consumers, amending Regulations (EC) No 1924/2006 and (EC) No 1925/2006 of the European Parliament and of the Council, and repealing Commission Directive 87/250/EEC, Council Directive 90/496/EEC, Commission Directive 1999/10/EC, Directive 2000/13/EC of the European Parliament and of the Council, Commission Directives 2002/67/EC and 2008/5/EC and Commission Regulation (EC) No 608/2004 Text with EEA relevance, 2011, https://eur-lex.europa.eu/eli/reg/2011/1169/oj/eng.
- U.S. Food and Drug Administration, GRN No. 91 Mycoprotein - Use in food in general, excluding meat products, poultry products, and infant formula, Marlow Foods Ltd., 2002, https://hfpappexternal.fda.gov/scripts/fdcc/index.cfm?set=GRASNotices%26id=91.
- U.S. Food and Drug Administration, Generally Recognized as Safe (GRAS), 2023, https://www.fda.gov/food/food-ingredients-packaging/generally-recognized-safe-gras.
- U.S. Food and Drug Administration, How U.S. FDA's GRAS Notification Program Works, 2005, https://www.fda.gov/food/generally-recognized-safe-gras/how-us-fdas-gras-notification-program-works.
- U.S. Code of Federal Regulations, Food labeling, 1977, https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-101.
- Y. Malila, I. O. Owolabi, T. Chotanaphuti, N. Sakdibhornssup, C. T. Elliott, W. Visessanguan, N. Karoonuthaisiri and A. Petchkongkaew, npj Sci. Food, 2024, 8, 53 CrossRef PubMed.
- J. Smanalieva, Bull. Kyrgyz Natl. Agrar. Univ., 2025, 23, 73–83 CrossRef.
- G. Praveen Chakravarthi, V. Rambabu, D. S. V. N. M. Ramamurty, G. Rahul and S. V. G. V. A. Prasad, Int. J. Plant, Anim. Environ. Sci., 2024, 14, 70–80 Search PubMed.
- S. Wainaina and M. J. Taherzadeh, Bioresour. Technol., 2023, 369, 128421 CrossRef CAS PubMed.
- J. Tigrero-Vaca, B. Díaz, G. Gu and J. M. Cevallos-Cevallos, Front. Bioeng. Biotechnol., 2025, 13, 1638957 CrossRef.
- D. Risner, K. A. McDonald, C. Jones and E. S. Spang, Front. Sustain. Food Syst., 2023, 7, 1204307 CrossRef.
- F. Gisperg, R. Klausser, M. Elshazly, J. Kopp, E. P. Brichtová and O. Spadiut, Biotechnol. Bioeng., 2025, 122, 1313–1325 CrossRef CAS PubMed.
- N. Rajasekhar, T. K. Radhakrishnan and S. N. Mohamed, Biotechnol. Bioeng., 2024, 121, 3114–3127 CrossRef CAS.
- M. Helmy, H. Elhalis, M. M. Rashid and K. Selvarajoo, Curr. Opin. Biotechnol., 2024, 87, 103115 CrossRef CAS.
- D. Priyadharshini, I. Muthuvel, S. Saraswathy, P. S. Kavitha and V. Jegadeeswari, Front. Nutr., 2025, 12, 1659511 CrossRef CAS.
- T. James, B. Williamson, P. Tino and N. Wheeler, Bioinformatics, 2025, 41, btaf206 CrossRef CAS PubMed.
- S. Jeon, Y. J. Sohn, H. Lee, J. Y. Park, D. Kim, E. S. Lee and S. J. Park, J. Microbiol., 2025, 63, e2501021 CrossRef CAS PubMed.
- V. Maini Rekdal, C. R. B. van der Luijt, Y. Chen, R. Kakumanu, E. E. K. Baidoo, C. J. Petzold, P. Cruz-Morales and J. D. Keasling, Nat. Commun., 2024, 15, 2099 CrossRef CAS PubMed.
- M. Lübeck and P. S. Lübeck, Microorganisms, 2022, 10, 753 CrossRef PubMed.
- E. D. Muniz, R. T. Montenegro, D. N. da Silva, A. P. D'Almeida, L. R. Gonçalves and T. L. de Albuquerque, Fermentation, 2024, 10, 638 CrossRef CAS.
- T. A. Magocha, H. Zabed, M. Yang, J. Yun, H. Zhang and X. Qi, Bioresour. Technol., 2018, 257, 281–289 CrossRef CAS PubMed.
- S. Salazar-Cerezo, R. P. de Vries and S. Garrigues, J. Fungi, 2023, 9, 834 CrossRef CAS PubMed.
- B. Ma, Y. Li, T. Wang, D. Li and S. Jia, J. Fungi, 2025, 11, 350 CrossRef CAS PubMed.
- S. Tong, K. An, W. Zhou, W. Chen, Y. Sun, Q. Wang and D. Li, J. Fungi, 2022, 8, 169 CrossRef CAS PubMed.
- F. M. Wilson and R. J. Harrison, Fungal Biol. Biotechnol., 2021, 8, 15 CrossRef CAS PubMed.
- T.-Q. Shi, J. Gao, W.-J. Wang, K.-F. Wang, G.-Q. Xu, H. Huang and X.-J. Ji, ACS Synth. Biol., 2019, 8, 445–454 CrossRef CAS PubMed.
- K. R. Westphal, R. D. Wollenberg, F.-A. Herbst, J. L. Sørensen, T. E. Sondergaard and R. Wimmer, Toxins, 2018, 10, 485 CrossRef CAS PubMed.
- M. Ferrara, M. Haidukowski, A. F. Logrieco, J. F. Leslie and G. Mulè, Sci. Rep., 2019, 9, 19836 CrossRef CAS.
- R. Colosimo, F. J. Warren, T. J. A. Finnigan and P. J. Wilde, Food Chem., 2020, 330, 127252 CrossRef CAS.
- X. Wu, Z. Zhou, S. Luo, Y. Wang, M. Yang, Y. Chen, G. Du, J. Chen and X. Liu, Bioresour. Technol., 2025, 436, 133005 CrossRef CAS PubMed.
- R. Hong, S. Tong, M. Chai, W. Chen, X. Liu, Y. Chen and D. Li, J. Agric. Food Chem., 2024, 72, 27274–27283 CrossRef CAS.
- Y. Seidler, G. Rimbach, K. Lüersen, G. Vinderola and I. R. Ipharraguerre, Front. Microbiol., 2024, 15, 1452725 CrossRef CAS.
- F.-J. Jin, S. Hu, B.-T. Wang and L. Jin, Front. Microbiol., 2021, 12, 644404 CrossRef.
- Z. Sun, Y. Wu, S. Long, S. Feng, X. Jia, Y. Hu, M. Ma, J. Liu and B. Zeng, J. Fungi, 2024, 10, 248 CrossRef CAS.
- D. Lee, J. H. Pan, D. Kim, W. Heo, E. C. Shin, Y. J. Kim, Y. Y. Shim, M. J. T. Reaney, S.-G. Ko, S.-B. Hong, H. T. Cho, T. G. Kim, K. Lee and J. K. Kim, Compr. Rev. Food Sci. Food Saf., 2024, 23, e13365 CrossRef CAS.
- Y. Ning, Y. Xu, B. Jiao and X. Lu, Mar. Drugs, 2022, 20, 705 CrossRef CAS PubMed.
- H. Zhu, H. Wang, L. Wang and Z. Zheng, Biotechnol. J., 2024, 19, 2400309 CrossRef CAS.
- B. M. Bartholomai, K. M. Ruwe, J. Thurston, P. Jha, K. Scaife, R. Simon, M. Abdelmoteleb, R. E. Goodman and M. Farhi, Food Chem. Toxicol., 2022, 168, 113342 CrossRef CAS PubMed.
- Z. Ji, W. Ma, P. Liang, X. Wang, S. Zhang, Y. Han and Y. Guo, Int. J. Biol. Macromol., 2024, 281, 136638 CrossRef CAS PubMed.
- K. Wadhwa, N. Kapoor, H. Kaur, E. A. Abu-Seer, M. Tariq, S. Siddiqui, V. K. Yadav, P. Niazi, P. Kumar and S. Alghamdi, Mycobiology, 2024, 52, 335–387 CrossRef.
- M. Shahid, A. Gaines, D. Coyle, R. Alessandrini, T. Finnigan, G. Frost, M. Marklund and B. Neal, Am. J. Clin. Nutr., 2023, 118, 141–150 CrossRef CAS.
- A. Cherta-Murillo and G. S. Frost, Br. J. Nutr., 2022, 127, 1685–1694 CrossRef CAS PubMed.
- M. O. C. Coelho, A. J. Monteyne, M. V. Dunlop, H. C. Harris, D. J. Morrison, F. B. Stephens and B. T. Wall, Nutr. Rev., 2019, 78, 486–497 CrossRef.
- X. Li, Y. Kang, Y. Huang, Y. Xiao, L. Song, S. Lu and Z. Ren, Biomed. Pharmacother., 2021, 137, 111290 CrossRef CAS.
- D. N. Farsi, J. L. Gallegos, G. Koutsidis, A. Nelson, T. J. A. Finnigan, W. Cheung, J. L. Muñoz-Muñoz and D. M. Commane, Eur. J. Nutr., 2023, 62, 1479–1492 CrossRef CAS.
- Good Food Institute, 2024, https://gfi.org/wp-content/uploads/2025/01/Consumer-snapshot-mycoprotein-in-the-US.pdf, (accessed September 2025).
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |