Open Access Article
Open Access ArticleRoles of the volatile and non-volatile fractions of thyme (Thymus vulgaris L.) essential oil in inhibiting Fusarium graminearum growth and trichothecene mycotoxin production
Xiaoxi Qiab,
Zixuan Xua,
Yueqiang Lengc,
Zhao Jina,
Bingcan Chen a and
Jiajia Rao
a and
Jiajia Rao *a
*a
aDepartment of Plant Sciences, North Dakota State University, Fargo, North Dakota 58108, USA. E-mail: jiajia.rao@ndsu.edu; Tel: +1 (701) 231-6277
bSchool of Food Engineering, East University of Heilongjiang, Harbin 150066, Heilongjiang, China
cDepartment of Plant Pathology, North Dakota State University, Fargo, North Dakota 58108, USA
First published on 3rd February 2026
Abstract
This study investigated the antifungal activity, mycotoxin inhibitory effects, and mechanisms of action of whole thyme oil, along with its volatile and non-volatile fractions. Thyme oil was fractionated into relatively volatile and non-volatile components using solvent-assisted flavor evaporation (SAFE) at 50 °C. The compositions of the two fractions were profoundly different. Afterwards, physically stable emulsions were prepared from the two fractions and whole thyme oil, and their effects were evaluated against two chemotypes of Fusarium graminearum including spore germination and mycotoxin production. All emulsions inhibited fungal growth and reduced mycotoxin accumulation to varying degrees. The non-volatile fraction of thyme oil exhibited the strongest suppression of spore germination, validated by the lowest EC50 values (0.54 and 0.74 µg mL−1, respectively) and demonstrated the greatest disruption of cytoplasmic membrane integrity, confirmed by advanced microscopic technologies, and total lipid content analyses. In terms of mycotoxin inhibitory effect, all emulsions significantly reduced deoxynivalenol (DON) levels to values compliant with FDA regulations and suppressed acetylated derivatives to as low as undetectable amounts. A non-detectable level of the acetylated derivatives was achieved by the non-volatile fraction at the EC50 concentration. Tri gene expression analysis in the DON biosynthesis pathway revealed that thyme oil and the non-volatile fraction up-regulated all Tri genes in both isolates, while the volatile fraction down-regulated Tri3 and Tri5 in strain isolate 10-124-1. In summary, the findings highlighted the potential of SAFE fractionation to selectively enrich the non-volatile fraction of Thymus vulgaris L., offering an effective and sustainable strategy for fungal control and mycotoxin reduction in food systems.
Sustainability spotlightDeoxynivalenol (DON), a mycotoxin produced by Fusarium spp. in the field, is frequently detected at high levels in foods. Because food processing is the final barrier against mycotoxin contamination, it is urgent to develop effective intervention strategies. With growing consumer demand for clean-label products free of artificial preservatives, plant extracts and essential oils with antimicrobial or antifungal properties have gained attention. However, the strong flavors of essential oils limit their use as label-friendly antifungal agents. Based on insights from flavor research, essential oils can be fractionated into volatile and non-volatile components using SAFE. We hypothesize that a non-volatile fraction with stronger antifungal or mycotoxin-inhibitory activity could enable lower usage levels and reduce off-flavors, supporting sustainable mycotoxin-control strategies. |
1 Introduction
Fungal spoilage remains a major cause of food waste along the supply chain, reducing quality, safety, and economic value in cereals, fruits, and processed foods. Beyond visible deterioration, many filamentous fungi produce mycotoxins such as aflatoxins, trichothecenes, ochratoxin A, and zearalenone, which are hazardous to human and animal health and result in substantial global economic losses.1–3 Mycotoxins are commonly found in a wide range of agricultural products, including peanuts, grapes and wine, cereals, nuts, dried fruits, coffee, cocoa, spices, oilseeds, fresh fruits, fruit juices, and beer, as well as other food and feed commodities.4 These toxic compounds can be produced by fungi at multiple points throughout the food supply chain (e.g., pre-harvest, harvest, drying, storage, and transportation), thereby posing a risk to consumers through direct intake of contaminated foods or indirectly via animal feed.5 Mycotoxins are generally considered carcinogenic, genotoxic, teratogenic, nephrotoxic, and hepatotoxic. Studies have found that delayed development, stunted growth, liver impairment and even liver cancer in children are linked to exposure to aflatoxins.6 Therefore, the prevention of fungal growth and mycotoxin contamination has become a critical focus in food safety research. Among the fungi identified, Fusarium species have been recognized as major contributors to the deterioration of stored cereal commodities, affecting both their quality and quantity. Apart from the wastage they cause, they also produce secondary metabolites harmful to human health, such as deoxynivalenol (DON) and its acetylated derivatives (e.g., 3-AcDON and 15-AcDON).7 Exposure to DON and its derivatives can lead to symptoms such as diarrhea, dysentery, ataxia, mucosal hemorrhage, and even sudden death. It is also associated with immune dysfunction, gastroenteritis, dermatitis, and hematological disorders, including aleukia (ATA) and aplastic anemia. Deoxynivalenol (DON) induces anorexia and vomiting by disrupting serotonin-regulated neuronal pathways and further exerts toxicity by suppressing protein synthesis through interference with ribosomal subunit binding.8 The economic impact of Fusarium infestations is also substantial, given the post-harvest losses in agricultural products during storage and downstream processes, such as cereal crops, malting and potatoes.9,10The use of essential oils (EOs) has gained increasing attention as promising antifungal agents in food preservation due to their plant-derived and biodegradable properties, and their generally recognized as safe (GRAS) status. Several studies have demonstrated that EOs can inhibit both mycelial growth and spore germination, as well as suppress mycotoxin biosynthesis in contaminated substrates. Peng et al. found that Litsea cubeba EO can the suppress spore germination rate by 87.7% and reduce DON production by more than 20% when applied to the malting process.11 Jiang et al. also studied the chemical composition of hop essential oil and its efficacy against fungal growth and mycotoxin accumulation. They indicated that with humulene and β-myrcene as the main constituents, hop essential oil was able to inhibit fungal growth and prevent DON production.12 Among many plant-derived EOs, thyme oil derived from Thymus vulgaris L., a member of the Lamiaceae family, with thymol, p-cymene, γ-terpinene, and linalool as the major chemical compounds has been widely recognized for its strong antifungal activity towards a board-spectrum range of fungi such as Aspergillus, Penicillium, and Fusarium,13,14 and has a long history of medicinal use.15,16 Puškárová et al. indicated that thyme essential oil vapor can inhibit the mycelium growth of Chaetomium globosum, Aspergillus fumigatus and Penicillium chrysogenum on malt extract agar plates.17 The antifungal mechanism has been linked to disruption of cell membrane integrity, alteration of ergosterol dependent structures, and induction of oxidative stress, ultimately leading to leakage of intracellular components and fungal death, and among the major components, thymol possesses the strongest activity18,19
Therefore, to further investigate the potential synergistic interactions among its minor compounds, we proposed a coarse separation of whole thyme oil into relatively volatile and non-volatile fractions. Our hypothesis is that if the non-volatile fraction exhibits greater antifungal and mycotoxin inhibitory efficacy than the volatile fraction or the whole thyme oil, it could minimize the usage level of EO and the undesirable flavor impact of EO on the foods to which it is applied. As learned from flavor researchers, the fractionation of EO into volatile and non-volatile component mixtures can be achieved using a Solvent-Assisted Flavor Evaporation (SAFE) system. This method uses high vacuum to isolate volatile compounds at low temperatures without oxidizing their chemical components.20 By avoiding extreme heat, it is well suited for the isolation or separation of low-boiling and heat-sensitive compounds. SAFE was originally developed for flavor research and has been widely applied in aroma chemistry and flavor profile analysis.21,22 However, its use for separating volatile components and relatively non-volatile constituents of EO has not yet been explored. This technique shows promise as a valuable tool for elucidating the respective contributions of volatile and non-volatile fractions to the overall antifungal and mycotoxin inhibitory efficacy of thyme oil. Moreover, it may enhance the efficacy of the resulting fractions by increasing the relative concentration of bioactive compounds within each part. Furthermore, if certain volatile chemical constituents are found to have little or no antifungal activity, they could be removed from the thyme oil-based formulation in the future. In addition to their antifungal and mycotoxin inhibitory efficacy, the effects of volatile and non-volatile compounds from thyme oil on the mechanisms involved in mycotoxin biosynthesis, such as the DON pathway, have not yet been investigated. Such knowledge could help expand the use of thyme oil as a natural antifungal agent in foods without causing undesirable flavor impacts, which is a current limitation in the application of EO in food systems.
The overall goals of this study are to evaluate the feasibility of using SAFE technology to fractionate thyme oil based on component volatility and to systematically investigate the antifungal and mycotoxin inhibitory efficacy, as well as the underlying mechanisms. To address the practical stability limitations of plant-derived EO in food applications due to their hydrophobic nature, emulsification technology was employed to encapsulate thyme oil and its fractions, thereby enhancing their stability. The overall objectives of this study were to evaluate the feasibility of applying the SAFE technique to separate thyme oil fractions based on volatility, formulate physically stable oil-in-water emulsions of thyme oil and its fractions, assess their antifungal activity and elucidate the mechanisms of action; and investigate the mycotoxin-inhibitory efficacy and the role of thyme oil and its fractions in regulating key genes (Tri3, Tri4, and Tri5) involved in the mycotoxin biosynthetic pathway of F. graminearum.
2 Materials and methods
2.1 Materials and chemicals
F. graminearum strains (10-124-1 and 10-125-1) with two chemotypes (e.g. 15-AcDON and 3-AcDON) were obtained from the Department of Plant Pathology at North Dakota State University (Fargo, ND, USA). Mycotoxin standards of DON, 3-AcDON and 15-AcDON for HPLC analysis were purchased from Cayman Chemical (Ann Arbor, MI, USA). Thyme essential oil, Tween 80, carboxymethylcellulose sodium salt (CMC), and primers were purchased from Millipore Sigma Co. (St. Louis, MO, USA). Potato dextrose agar (PDA) and potato dextrose broth (PDB) were purchased from Hardy Diagnostics (Sabta Maria, CA, USA). Yeast extract and agar were purchased from Difco Laboratories (Detroit, MI, USA). Mung beans and canola oil were obtained from a local market (Fargo, ND, USA). The RNA extraction kit (T2010S) and DNase I (#M0303S) were purchased from New England Biolabs (Ipswich, MA, USA). SsoAdvanced™ Universal SYBR® Green Supermix was purchased from Bio-Rad (Bio-Rad laboratories, Hercules, CA, USA). All the other chemicals used were of analytical grade and all solutions were prepared using double distilled water (DDW, 18.2 MΩ cm, Barnstead ultrapure water system, Thermo Fisher Scientific, USA).2.2 Characterizing thyme oil and its volatile and non-volatile fractions obtained by solvent-assisted flavor evaporation (SAFE)
2.3 Preparation of thyme oil and its fraction emulsions
Coarse oil-in-water EO emulsions were prepared by blending 5 wt% of thyme oil or its fractions with 5 wt% canola oil, 1 wt% Tween 80, and 89 wt% phosphate buffer (10 mM, pH 7.0) using a high-speed hand blender (M133/128-0, Biospec Products, Inc., ESGC, Switzerland) at speed 6 for 2 min. The resulting coarse emulsions were subsequently processed through a high-pressure homogenizer (LM 20–20 Microfluidizer Processor, Westwood, MA, USA) twice at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 psi to further reduce droplet size.24 Final EO emulsions were stored at 4 °C until use, unless otherwise specified. The concentration of EO in the emulsion systems was 50 mg g−1.
000 psi to further reduce droplet size.24 Final EO emulsions were stored at 4 °C until use, unless otherwise specified. The concentration of EO in the emulsion systems was 50 mg g−1.
2.4 Long-term stability of EO emulsions
To evaluate the physical stability of prepared EO emulsions, the particle size and size distribution of the emulsions prepared in Section 2.3 were monitored over a 63-day period at room temperature. Measurements were performed using a dynamic light scattering instrument (Zetasizer Nano ZEN 3600, Malvern Instruments, Malvern, UK), following the procedure described previously.24 Prior to analysis, all samples were diluted tenfold with phosphate buffer (10 mM, pH 7) to minimize multiple scattering effects. The results are presented as the mean droplet diameter and particle size distribution.2.5 Antifungal activity and mode of action of EO emulsions on spore germination of F. graminearum
 | (1) |
Third-order polynomial regression was used to model the non-linear inhibition rates (R2 > 0.99). The absolute EC50, defined as the concentration of EOs in the formed emulsions required to inhibit 50% of spore germination, was then calculated.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) in a test tube, followed by vigorous shaking for 30 min. One milliliter of the mixture was then centrifuged (Avanti J-26S XPI; Beckman Coulter Inc., Palo Alto, CA, USA) at 4000×g for 10 min, and 0.2 mL of the supernatant was transferred to a boiling water bath to evaporate the solvent to dryness before total lipid content measurement. In terms of total lipid content measurement, five hundred microliters of H2SO4 were added to the lipid containing tube and boiled in a boiling water bath for 10 min. After cooling, 3 mL phosphor–vanillin solution was added, shaken vigorously and then incubated at room temperature for 10 min. The absorbance at 520 nm was utilized to calculate total lipid content from the standard calibration curve using cholesterol as a standard.
1, v/v) in a test tube, followed by vigorous shaking for 30 min. One milliliter of the mixture was then centrifuged (Avanti J-26S XPI; Beckman Coulter Inc., Palo Alto, CA, USA) at 4000×g for 10 min, and 0.2 mL of the supernatant was transferred to a boiling water bath to evaporate the solvent to dryness before total lipid content measurement. In terms of total lipid content measurement, five hundred microliters of H2SO4 were added to the lipid containing tube and boiled in a boiling water bath for 10 min. After cooling, 3 mL phosphor–vanillin solution was added, shaken vigorously and then incubated at room temperature for 10 min. The absorbance at 520 nm was utilized to calculate total lipid content from the standard calibration curve using cholesterol as a standard.2.6 Impact of thyme oil and its fractions on mycotoxin inhibitory efficacy
F. graminearum isolates were cultured in trichothecene biosynthesis induction (TBI) medium to induce mycotoxin production, as previously described.30 In brief, EO emulsions were added to TBI medium at their EC50 concentrations, as determined in Section 2.5.1, while medium without emulsion served as the control. Five hundred microliters of spore suspension (≈106 spores per mL) was inoculated into 50 mL of TBI medium in a flask and incubated at room temperature in the dark with shaking (MaxQ 4000 Digital Orbital Incubator Shaker, Thermo Scientific, Waltham, MA, USA) at 150 rpm for 10 days. After incubation, cultures were filtered through autoclaved Miracloth, and 20 mL of filtrate was extracted with 20 mL ethyl acetate by shaking at 150 rpm for 15 min. A 15 mL portion of the ethyl acetate phase was concentrated using a BUCHI RE121 rotary evaporator (BUCHI, Switzerland) and dried under a gentle nitrogen stream. The resulting residue was re-dissolved in acetonitrile (ACN) prior to high-performance liquid chromatography (HPLC) analysis. Mycotoxin contents of F. graminearum isolates, including DON, 3-AcDON, and 15-AcDON, extracted from TBI medium were quantified using an Agilent 1200 HPLC system (Agilent, Milford, MA, USA) equipped with a SynChropak C18-300 column as described previously (250 × 4.6 mm × 300 Å, HP Genenchem, San Francisco, CA, USA).19 The limits of quantitation (LOQ) and detection (LOD) for DON, 3-AcDON, and 15-AcDON were 0.030/0.090, 0.14/0.41, and 0.23/0.70 µg mL−1, respectively under these conditions.2.7 Impact of thyme oil and its fractions on expression of Tri genes (Tri3, Tri4 and Tri5)
To assess the impact of thyme oil and its fractions on the expression of Tri-cluster genes (Tri3, Tri4, and Tri5) in F. graminearum during the DON biosynthesis pathway, both isolates were cultured in TBI medium containing 0.1 mg mL−1 thyme oil or its fraction emulsions and incubated for 10 days at room temperature with shaking at 150 rpm in the dark. TBI medium without emulsions was used as the control. Mycelia were harvested using sterilized Miracloth, washed three times with sterile DDW, and excess moisture was removed before lyophilization for 72 h.19 Lyophilized mycelia (∼10 mg) were placed in a clean tube and mechanically disrupted using a TissueLyser II (Qiagen, Hilden, Germany) for 1 min at 24 Hz. Total RNA was extracted from each sample using a Monarch® Total RNA Miniprep Kit (New England Biolabs, MA, USA) according to the manufacturer's instructions. RNA quality and quantity were assessed by 1% agarose gel electrophoresis and using a Nanodrop OneC spectrophotometer (Thermo Scientific, Waltham, MA, USA), respectively, and residual DNA was removed by using DNase I (New England Biolabs, MA, USA) treatment following the manufacturer's protocol. Quantitative real-time PCR (qRT-PCR) was conducted following the method of Leng et al. with minor changes.31 A total of 2.8 µg of RNA from each sample was reverse transcribed using a SuperScript® III First-Strand Synthesis System (Invitrogen, Waltham, MA, USA). The resulting cDNA was diluted 10-fold and used as a template for quantitative PCR on a CFX96 Real-Time PCR System (Bio-Rad, Hercules, CA, USA). Amplification data were processed using Bio-Rad CFX Manager software (version 1.6), with threshold cycle (Ct) values automatically determined. Each 20 µL reaction mixture contained 4 µL of diluted cDNA, 10 µL SsoAdvanced™ Universal SYBR® Green Supermix (Bio-Rad), 1 µL of each primer (10 µM), and 4 µL nuclease-free water. Relative expression levels of Tri-cluster genes were calculated using the 2−ΔΔCt method, with elongation factor 1α (EF1α) as the internal reference gene. Primers are listed in SI Table S2. Each cDNA sample was analyzed in a minimum of four replicates.2.8 Statistical analysis
All measurements were performed at least in duplicate using freshly prepared samples, and the results are expressed as mean ± standard deviation. Statistical analysis was conducted using one-way analysis of variance (ANOVA), followed by Fisher's pairwise comparison test to determine significant differences between means at a 95% confidence level (MINITAB 18.0).3 Results and discussion
3.1 Compositions of thyme oil fractions based on volatility
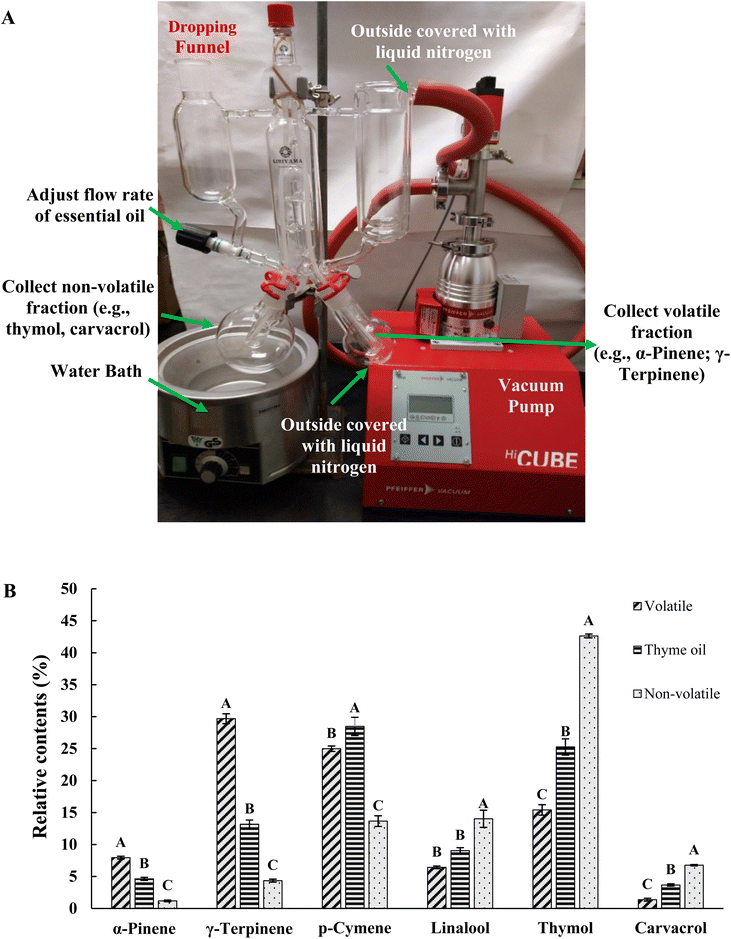
SAFE is a technique used to separate and concentrate volatile compounds by combining solvent extraction with high vacuum (10−3 to 10−4 Pa) and relatively low temperatures (30–50 °C) maintained using water baths.20 It has been used to facilitate the identification and quantification of aroma compounds in various food matrices, such as beer, fish, and ham.32,33 In this study, SAFE was employed as a suitable means to separate the components of thyme oil into two sets of mixtures based on their volatility. Although all major components of thyme oil are volatile, the mixture collected in the re-condensation flask was designated as the volatile fraction, whereas the residue remaining in the receiver was referred to as the non-volatile fraction in this study. In our preliminary experiment (SI Table S1), three different temperatures (30 °C, 40 °C and 50 °C) were applied in the SAFE process. We observed that the relative contents of γ-terpinene and p-cymene, the major low boiling point components of thyme oil, decreased in the non-volatile fraction as the temperature increased. Meanwhile, the relative contents of thymol and carvacrol increased, the major non-volatile compounds in thyme oil, indicating improved separation efficiency. Therefore, subsequent experiments were performed using SAFE at 50 °C to separate thyme oil into two fractions, yielding 20.2 mL of the volatile fraction and 29.1 mL of the non-volatile fraction from a total of 50 mL of thyme oil which indicated a total recovery rate of 98.6%, nearly 100%. The compositions of the two fractions along with thyme oil were analyzed by GC-MS and the results are shown in Table 1. The extraction rate calculated using benzene, 1, 2-dichlorobenzene as the internal standard was 1.04 ± 0.05 (R2 > 0.99), as calculated using the formula below:
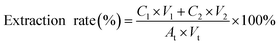 | (2) |
| Components | Boiling point (°C) at (760 mmHg) | Retention time (min) | Thyme oil (%) | Volatile fraction (%) | Non-volatile fraction (%) |
|---|---|---|---|---|---|
| α-Pinene | ∼155 | 8.42 | 4.63 ± 0.06 | 7.94 ± 0.23 | 1.18 ± 0.13 |
| Camphene | ∼159 | 8.80 | 1.45 ± 0.01 | 2.25 ± 0.14 | 0.35 ± 0.03 |
| β-Myrcene | ∼166 | 8.93 | 2.13 ± 0.01 | 4.08 ± 0.07 | 0.57 ± 0.05 |
| Eucalyptol | ∼176 | 9.10 | 2.17 ± 0.03 | 2.74 ± 0.03 | 1.69 ± 0.08 |
| γ-Terpinene | ∼182 | 9.23 | 13.17 ± 0.27 | 29.67 ± 0.77 | 3.35 ± 0.23 |
| p-Cymene | ∼177 | 9.59 | 28.47 ± 0.56 | 24.99 ± 0.42 | 13.66 ± 0.83 |
| Linalool | ∼198 | 12.18 | 9.05 ± 0.08 | 6.42 ± 0.20 | 13.02 ± 1.34 |
| D-Camphor | ∼204 | 13.52 | 2.01 ± 0.04 | 0.67 ± 0.14 | 3.29 ± 0.54 |
| Terpinen-4-ol | ∼212 | 14.32 | 1.83 ± 0.06 | 0.81 ± 0.09 | 3.38 ± 0.04 |
| Caryophyllene | ∼245.3 | 15.39 | 1.49 ± 0.12 | 0.92 ± 0.05 | 2.80 ± 0.04 |
| Isoborneol | ∼212 | 15.94 | 0.92 ± 0.01 | 0.62 ± 0.02 | 1.46 ± 0.01 |
| endo-Borneol | ∼210 | 16.48 | 1.63 ± 0.05 | 0.75 ± 0.09 | 2.79 ± 0.03 |
| Thymol | ∼232 | 24.83 | 25.27 ± 0.02 | 15.41 ± 0.81 | 42.62 ± 0.30 |
| Carvacrol | ∼236 | 30.05 | 3.68 ± 0.11 | 1.36 ± 0.23 | 6.77 ± 0.10 |
| Total | N/A | N/A | 97.90 | 98.53 | 96.93 |
• C1 and C2: relative concentrations of benzene, 1, 2-dichlorobenzene in volatile and non-volatile thyme oil after SAFE extraction;
• V1 and V2: volume of volatile and non-volatile thyme oil after SAFE extraction;
• At: concentration of benzene, 1, 2-dichlorobenzene added to the thyme oil before extraction;
• Vt: total volume of thyme oil subjected to SAFE extraction.
In Table 1, components accounting for more than 95% of thyme oil were listed in order of retention time obtained by GC-MS. Their relative contents (%) in thyme oil, and volatile and non-volatile fractions are presented in Table 1. As shown, the major components of thyme oil, in decreasing order of abundance, were p-cymene, thymol, γ-terpinene, linalool, and carvacrol, which is consistent with previous studies.34 In general, a lower boiling point or retention time of a chemical compound corresponds to higher volatility.35,36 We observed that the first five compounds listed in Table 1 (α-pinene, camphene, β-cymene, eucalyptol, and γ-terpinene) were present at higher relative percentages in the volatile fraction compared to whole thyme oil, and at lower levels in the non-volatile fraction, indicating that these compounds are highly volatile. Interestingly, the relative percentage content of p-cymene in both fractions was lower than that in whole thyme oil, implying a possible loss of p-cymene during the process. This may result from the transformation of p-cymene to other compounds, such as terpenes and thymol.37,38 However, this hypothesis needs further study. Apart from the compounds mentioned above, the rest of the detected components exhibited the opposite trend, meaning that they were more abundant in the non-volatile fractions due to their higher boiling point and hence, lower volatility.
Based on the results in Table 1, we selected six compounds with abundances ≥4.6% to compare their relative content profiles in thyme oil and its two fractions. As shown in Fig. 1B, the levels of all six compounds differed significantly (p < 0.05) between the volatile and non-volatile fractions. These results confirm that the SAFE method we developed for thyme oil is effective, as it successfully separates the two fractions. They also clearly indicate that γ-terpinene and α-pinene are highly volatile compared to other major compounds such as thymol and carvacrol.
3.2 Characterization of EO emulsions
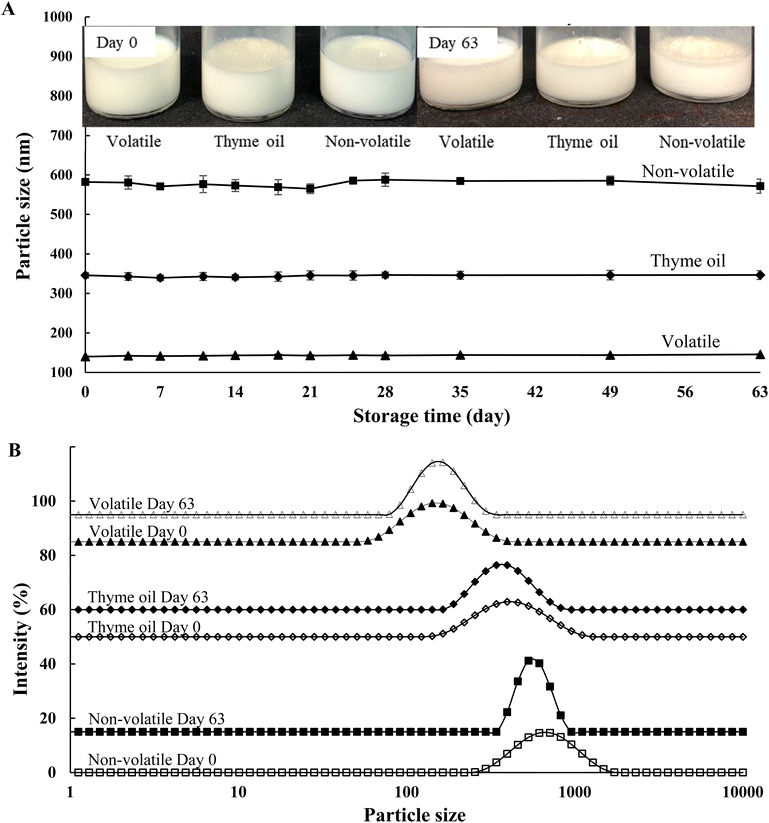
In general, the direct use of EOs as antimicrobial agents has several limitations, including low water solubility and poor stability. To overcome these limitations, oil-in-water emulsion systems were prepared in this study to encapsulate the EOs, as they are simple to formulate and easy to operate.24 The particle size of the freshly prepared emulsions and their physical stability over a 9-week period were monitored by measuring particle size, as shown in Fig. 2. As indicated in Fig. 2A, the particle sizes of the freshly prepared emulsions containing thyme oil, its volatile fraction, and its non-volatile fraction ranged from 100 to 600 nm, which was consistent with the previous study.19 The volatile fraction produced the smallest particles, falling within the typical nanoemulsion range (20–200 nm),39 whereas the non-volatile fraction exhibited the largest particle size. The particle size of an emulsion can be influenced by multiple factors, including the physicochemical properties of the encapsulated bioactive compounds, oil content, the choice of emulsifier, the oil-to-emulsifier ratio, and the homogenization conditions.40 For instance, the particle sizes of emulsions obtained from linalool with the same type of emulsifier but different oil-to-emulsifier ratios were significantly different.41 And a previous study showed that at a lower concentration of thyme oil (2.5% wt), the particle size of the emulsion was smaller than that in the current study.16 Smaller particle sizes were also found where thyme oil and thymol emulsions were formed with chitosan at a 1 : 1 ratio.42 In the current study, the varying factor was the composition of the encapsulated compounds. Although the same set of compounds was present in all three samples, the contents of each constituent differed as shown in the previous section. More specifically, thymol and carvacrol have markedly higher densities (960 kg m−3 and 977 kg m−3, respectively, at 15 °C) compared with α-pinene, p-cymene, γ-terpinene and linalool (840–865 kg m−3 at 15 °C). Therefore, the non-volatile emulsion exhibited considerably larger particle sizes, while thyme oil showed intermediate values. This was consistent with a previous study which demonstrated that lemon oil emulsions prepared with higher-density lemon oil (e.g., 10× concentrated) resulted in larger particle sizes than those prepared with lower-density lemon oils.43Regarding the long-term stability of EO emulsions, all EOs encapsulated in the emulsion-based delivery system retained similar mean particle sizes for an extended period (>2 months) at room temperature, with no evidence of phase separation or oiling-off (Fig. 2A, inserted images). On the other hand, particle size distribution (PSD), an important indicator of droplet homogeneity, was also monitored during storage time. The PSD of the emulsions at day 0 and day 63 is shown in Fig. 2B. As observed, all EO emulsions displayed monomodal distributions with similar widths, even after 63 days of storage. Interestingly, a slightly narrower PSD width was observed for both the volatile and non-volatile emulsions over the storage period, with the effect being more pronounced in the non-volatile emulsion. It could be attributed to Ostwald ripening, a thermodynamically driven process in which smaller droplets lose their encapsulated compounds and redeposit onto larger ones, while extremely large particles gradually break down over time.39 This indicated that although all tested emulsions showed good stability during storage, the non-volatile emulsion likely had relatively lower stability over extended storage compared to the other two.
3.3 Antifungal efficacy of EOs and their fractions in emulsion-based delivery systems
The antifungal efficacy of the three EOs in the emulsion delivery system against F. graminearum isolates 10-124-1 and 10-125-1 based on the spore germination inhibition (SGI) rate is plotted in Fig. S1 (SI Fig. S1). The effective EO concentration in the emulsion required to inhibit 50% of spore germination (EC50) is determined from Fig. S1 and is shown in Table 2.| EO | EC50 (µg mL−1) | |
|---|---|---|
| Isolate 10-124-1 | Isolate 10-125-1 | |
| a Different capitalized letters indicate that the means differ significantly within the same column (p < 0.05). Different small letters indicate that the means of effective concentration differ significantly between two isolates (p < 0.05). | ||
| Volatile | 0.99 ± 0.07 Ab | 1.76 ± 0.12 Aa |
| Thyme oil | 0.67 ± 0.12 Ba | 0.85 ± 0.03 Ba |
| Non-volatile | 0.54 ± 0.01 Ba | 0.74 ± 0.01 Ba |
From Table 2, one can see that the lowest concentrations of EC50 were observed in the non-volatile fraction of thyme oil at 0.54 and 0.74 µg mL−1 for isolates 10-124-1 and 10-15-1, respectively, followed by whole thyme oil. Meanwhile, the amount of volatile EO emulsions required to achieve the same effect was significantly higher (p < 0.05). For instance, the EC50 value of the volatile sample for both tested isolates were 0.99 and 1.76 µg mL−1 which were around twice those of non-volatile samples. However, the EC50 values between non-volatile and thyme oil for each isolate were not significantly different. As shown in Table 1, the volatile fraction was found to be rich in monoterpenes, including α-pinene, p-cymene, and γ-terpinene, whereas the non-volatile fraction was dominated by monoterpenoids (linalool) and phenolic compounds (thymol and carvacrol). The differences in antifungal activity, as measured by SGI, among the three samples were attributed to the higher percentage of phenols, followed by oxygenated terpenoids and terpenes.16 Several studies on the structure–activity relationship (SAR) have explained why different chemical structures exhibit varying antifungal activities. For example, thymol shows much stronger antifungal activity than p-cymene, which differs only by the absence of a hydroxyl group.19 As reported previously, the hydroxyl group in thymol was believed to play a key role in destabilizing the cytoplasmic membrane and reducing the pH gradient by acting as an electron donor, leading to ATP depletion and cell death.44 Moreover, the hydroxyl group was also involved in suppressing the enzymatic activities in cells, such as ATPase, decarboxylase, and protease, and the inactivation of ATPase in cellular respiration that will also cause cell death eventually.45 Cui et al. also studied the SAR of the benzene ring and the antimicrobial activity. They suggested that the benzene ring can participate in π–π stacking interactions or cation–π interactions with target biomolecules, resulting in more potent antifungal activities.46 Therefore, thymol as a phenol compound shows stronger antifungal activity than linalool, which lacks an aromatic ring. In addition to the SAR observed in different fractions, p-cymene may have been partially converted into thymol or carvacrol during the SAFE process, as discussed in Section 3.1, since p-cymene is a precursor and such transformations naturally occur in plants.38 Although this requires further validation, it provides new insights into enhancing the antifungal activity of the resulting fractions by converting p-cymene to thymol during SAFE.
Regarding the two Fusarium isolates, we can clearly see that isolate 10-124-1 was more prone to EOs since the EC50 values of all three treatments were lower than that of isolate 10-125-1. The pattern was more pronounced in the volatile sample, where a significantly higher concentration of EO (p < 0.05) was required to achieve 50% inhibition of isolate 10-125-1 compared to isolate 10-124-1. This is consistent with previous findings that fungal tolerance to bioactive compounds and EOs is highly strain dependent.47,48 Cristani et al. also tested the antibacterial properties of four bioactive compounds commonly found in thyme and oregano (p-cymene, γ-terpinene, thymol and carvacrol) on Gram-positive and Gram-negative microorganisms, respectively. They indicated that thymol is considerably more effective against S. aureus (Gram-positive) than the others, while carvacrol and p-cymene exhibited the strongest inhibitory against E. coli (Gram-negative).49
3.4 Antifungal mode of action
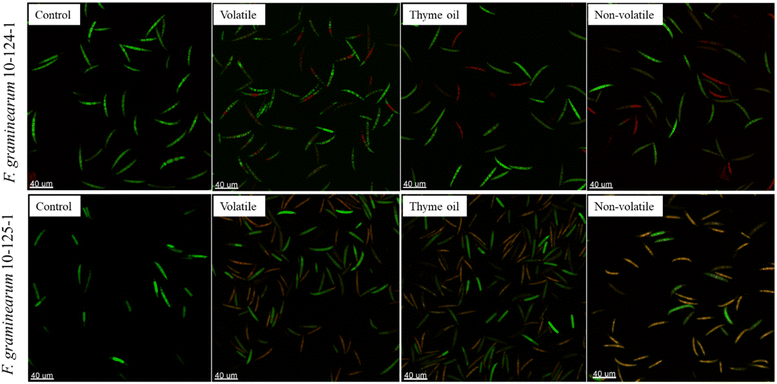
As shown in Fig. 3, spores of both isolates were smooth and plump with 3–4 septa in the control group. The presence of adhesion pads (highlighted in red circles) further indicated spore viability, as adhesion to the host surface is essential for fungal development.51 In contrast, spores exposed to EO emulsions, particularly thyme oil and non-volatile fraction emulsions, displayed pronounced morphological alterations. Noticeable shrinkage, rugged surfaces and partial membrane collapse were observed clearly under ×5000 magnification in both isolates. The shrinkage observed in spores of F. graminearum 10-124-1 was likely a result of substantial cytoplasmic leakage.52 Moreover, multiple pores were found on the surface of F. graminearum 10-125-1 spores in thyme oil and non-volatile fraction emulsion treated samples, and part of the spore structure even collapsed, as indicated by the red arrow in Fig. 3. These observations were consistent with earlier reports demonstrating that thymol, the major active component of thyme oil, can interfere with ergosterol biosynthesis, leading to disruption of the cell membrane and cell wall, which ultimately results in spore and hyphal deformation, collapse, and cell death.53
On the other hand, spores treated with the volatile fraction emulsion also showed some deformation although milder than that observed in spores treated with thyme oil and non-volatile fraction emulsions, which was in accordance with our results from the previous section that the antifungal activity against the two tested Fusarium isolates of the volatile fraction was weaker when compared to thyme oil and the non-volatile fraction.
Consistent with the SEM findings, the control samples contained a large number of viable spores from both F. graminearum isolates, as indicated by the strong green fluorescence signals (Fig. 4). In contrast, EO treated samples of isolate 10-124-1 displayed clear red fluorescence, suggesting compromised cytoplasmic membranes and leakage of cell contents. This damage prevented esterases from converting FDA into a fluorescent signal, while simultaneously allowing PI to penetrate the spores and bind to nucleic acids. Interestingly, in volatile fraction treated samples (Fig. 4), the red signal was localized only to one or more septa rather than the entire spore. This observation implies that thyme oil and its fractions induced irreversible changes in membrane permeability, leading to spore death, although the volatile fraction exerted a relatively weaker effect than the others. In the case of isolate 10-125-1, a different pattern emerged as shown in Fig. 4. The spores exhibited an orange coloration, reflecting simultaneous fluorescence from both FDA and PI. This indicated that membrane damage occurred to an extent sufficient for PI entry but not severe enough to abolish enzymatic activity, as FDA fluorescence was still detectable. These results further highlight that the response of fungal spores to EO treatment is strain dependent.
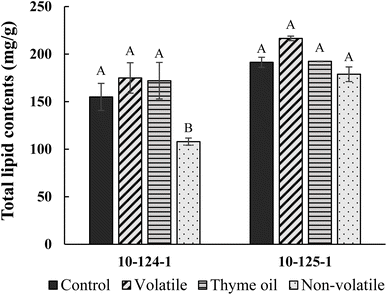
In our study, significant changes in total lipids were only found in the non-volatile fraction treated isolate 10-124-1 sample. On one hand, this was consistent with our previous results indicating that isolate 10-124-1 was less resilient and more prone to the antifungal activity of EOs. Secondly, the significant reduction in total lipid content was likely due to the high thymol concentration in the non-volatile fraction, as thymol is known for the disruption of cell membranes.53 Based on SEM, CLSM and total lipid content results, we gained a clear understanding of the antifungal mode of action of thyme oil and its fractions, which was closely associated with their ability to disrupt cell membrane integrity. Among all the tested EOs, the non-volatile fraction exhibited the strongest antifungal activity, followed by thyme oil and the volatile fraction.
3.5 Mycotoxin inhibitory efficacy and their mode of action
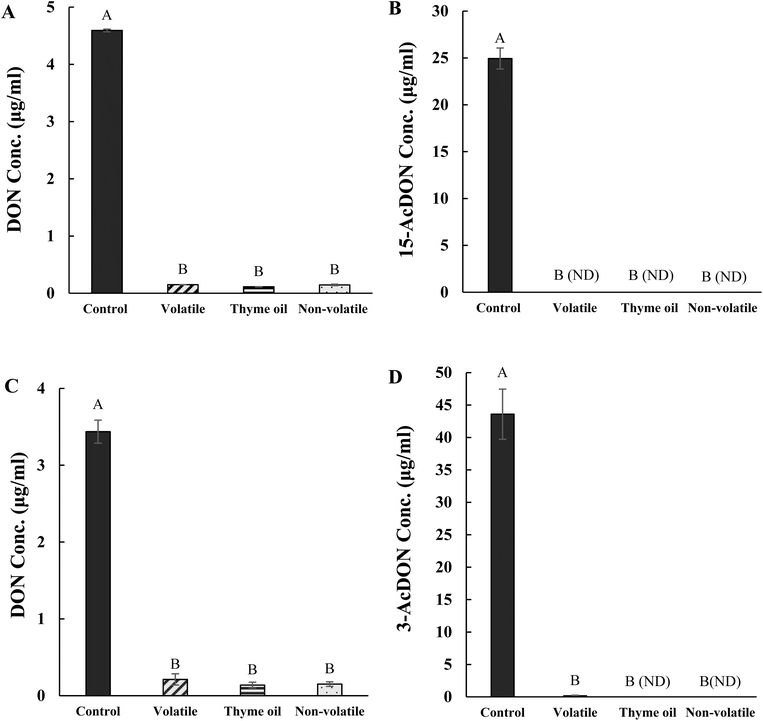
Overall, all EO emulsion treatments significantly (p < 0.05) reduced mycotoxin production in F. graminearum. As shown in Fig. 6A and C, DON levels in both isolates dropped below 1 ppm, which is the FDA advisory limit for finished wheat products.56 Specifically, DON production decreased from 4.5 to 0.11 µg mL−1 in isolate 10-124-1 and from 3.4 to 0.15 µg mL−1 in isolate 10-125-1. A marked reduction in acetylated DON was also observed. In isolate 10-124-1, all emulsions lowered 15-AcDON from approximately 25 µg mL−1 in the control to undetectable (ND) levels (p < 0.05) (Fig. 6B), with a similar outcome for isolate 10-125-1 (Fig. 6D). These findings demonstrate that, at their respective EC50 concentrations, thyme oil and volatile and non-volatile fractions were equally effective in suppressing mycotoxin accumulation.
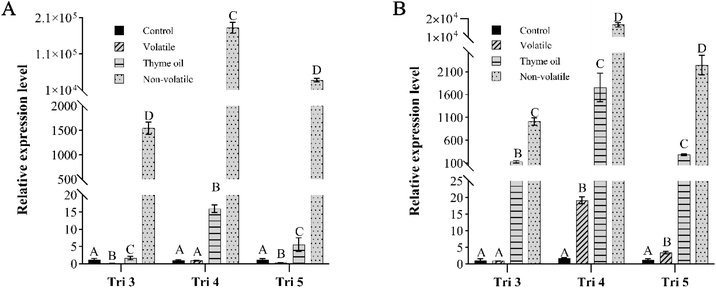
Interesting, significantly (p < 0.05) up-regulated expression levels of all three targeted genes were observed for both isolates when exposed to thyme oil and non-volatile fraction emulsions. Extremely high Tri4 gene expressions were observed in non-volatile fraction emulsion treated samples, especially for isolate 10-124-1 (Fig. 7). In the meantime, the up-regulated expression of Tri5 was also substantial in both isolates yet not as pronounced as that of Tri4. This was in accordance with the previous study where thyme oil emulsion was highly involved in the mycotoxin biosynthesis pathway and affected the expression of Tri4 and Tri5 genes to a great extent.19 According to the results from non-volatile fraction emulsion treated samples, we have good reasons to assume that the up-regulated gene expression was related to the higher concentration of the terpenoids and phenols, such as linalool, thymol and carvacrol in the non-volatile fraction.
For volatile fraction emulsion treatments, gene expression responses varied between isolates. In isolate 10-124-1, the volatile fraction downregulated all three target genes except Tri4 (Fig. 7A). In contrast, isolate 10-125-1 showed upregulation of all genes except Tri3 (Fig. 7B). However, the extent of changes found in volatile fraction emulsion-treated samples was not as pronounced as in the other two treatments. The above results indicated that (1) gene expression responses to specific stimuli were strain dependent; (2) individual genes reacted differently to the same stimulus, with Tri4 showing stronger up-regulation compared to the other two genes; (3) the regulation of Tri-cluster genes was dynamic due to the sequential nature of mycotoxin biosynthesis, and under the current culture conditions, Tri4 and Tri5 genes were regulated to a higher extent.
Previous studies have generally shown that treatment of Fusarium spp. with antifungal agents downregulated Tri gene expression, which was typically associated with reduced mycotoxin production.57 In contrast, our results revealed inconsistent effects of EO emulsions on Tri-cluster gene expression. The only downregulated genes were Tri3 and Tri5 when isolate 10-124-1 was incubated with volatile fraction emulsion. These results indicate that the Tri gene regulation is strain-dependent and up-regulation of certain genes is not necessarily correlated to the increased expression of mycotoxins. Moreover, this discrepancy may be explained by differences in the culture conditions. Most reports of Tri gene downregulation were derived from solid substrates, such as wheat kernels58 rather than liquid medium, which was used in the current study. Moreover, Duan et al. reported that certain fungicides actually enhanced DON biosynthesis and upregulated Tri5 and Tri6 expression in Fusarium grown in TBI liquid medium,59 which was consistent with the results observed for thyme oil and non-volatile fraction emulsion treated samples in the current study. Overall, the mechanism by which EOs regulate mycotoxin biosynthesis genes requires further investigation. Since the volatile fraction had only minor effects on gene regulation, it may be worthwhile to explore whether the elevated gene expression levels could be associated with other aspects of fungal metabolism beyond mycotoxin production.
4 Conclusions
In conclusion, thyme oil was successfully separated into two relative volatile and non-volatile fractions with distinct chemical compositions and properties using the SAFE fractionation method at 50 °C, based on differences in compound volatility. Both fractions, along with the whole thyme oil, were homogenized into oil-in-water emulsions that remained physically stable at ambient temperature for up to 63 days. All of the EO emulsions demonstrated inhibitory effects on fungal growth and mycotoxin production in two chemotypes of F. graminearum, although to varying degrees. Among the tested samples, the non-volatile fraction exhibited the most pronounced suppression of spore germination, largely attributed to its higher content of thymol and carvacrol, followed by the whole thyme oil and then the volatile fraction. SEM and CLSM analyses indicated that the antifungal activity of these EOs was primarily associated with increased permeability of the fungal cytoplasmic membrane. In addition, lipid content measurements further confirmed that the non-volatile fraction had a strong disruptive impact on membrane integrity. With respect to mycotoxin inhibition, all tested emulsions applied at their EC50 concentrations significantly reduced DON levels to within FDA-compliant limits, while also markedly decreasing the acetylated derivatives of DON. Between the two isolates examined, strain 10-125-1 showed greater tolerance to the EOs in both antifungal performance and mycotoxin production. Finally, the effect of EO emulsions on the expression of Fusarium Tri-cluster genes (Tri3, Tri4, and Tri5) involved in the DON biosynthetic pathway was also evaluated. The results showed that thyme oil and the non-volatile fraction significantly (p < 0.05) up-regulated gene expression, particularly Tri4 and Tri5, in both isolates. Conversely, changes induced by the volatile fraction of thyme oil were remarkably small.Altogether, this study demonstrated the feasibility of using the SAFE method to fractionate thyme oil and confirmed that the non-volatile fraction was more effective in reducing spore germination and mycotoxin accumulation in Fusarium strains than the whole EO or volatile fraction. This study provides a new perspective on the application of EOs for controlling fungal growth and mycotoxin accumulation in food systems without significantly affecting the flavor profile of the product using the non-volatile fraction of EOs.
Conflicts of interest
The are no conflicts to declare.Data availability
Data will be available upon request.Supplementary information (SI): figure and table has been cited in manuscript. Fig. S1 cited in Section 3.3. Table S1 cited in Section 3.1 and Table S2 cited in Section 2.7. See DOI: https://doi.org/10.1039/d5fb00909j.
Acknowledgements
This work was supported by the U.S. Wheat and Barley Scab Initiative (FAR0031915) and the United States Department of Agriculture National Institute of Food and Agriculture (FAR0032822). We acknowledge the NDSU Electron Microscopy Center core facility for providing SEM services, supported by the National Science Foundation under Grant No. 0619098.References
- A. Maggiore, A. Afonso, F. Barrucci and G. D. Sanctis, EFSA Supporting Publ., 2020, 17(6), 1–146 Search PubMed.
- S. Karanth, S. Feng, D. Patra and A. K. Pradhan, Front. Microbiol., 2023, 14, 1–17 Search PubMed.
- S. M. Abdelhameed and B. A. Khalifa, BMC Microbiol., 2024, 24(1), 209 Search PubMed.
- S. Agriopoulou, E. Stamatelopoulou and T. Varzakas, Foods, 2020, 9(2), 137 Search PubMed.
- G. Winter and L. Pereg, Eurasian J. Soil Sci., 2019, 70(4), 882–897 Search PubMed.
- C. G. Awuchi, I. O. Amagwula, P. Priya, R. Kumar, U. Yezdani and M. G. Khan, Bull. Environ., Pharmacol. Life Sci., 2020, 9, 149–155 Search PubMed.
- T. Dd Souza, C. Luz, J. Mañes, E. Badiale-Furlong and G. Meca, Food Sci. Technol., 2022, 42, e35821 CrossRef.
- A. Zöngür, J. Plant Dis. Prot., 2020, 127(4), 521–527 Search PubMed.
- Z. Jin, S. Solanki, G. Ameen, T. Gross, R. S. Poudel and P. Borowicz, et al., Mol. Plant-Microbe Interact., 2021, 34(7), 793–802 CrossRef PubMed.
- A. Zöngür, Potato Res., 2025, 68(1), 355–365 Search PubMed.
- Z. Peng, W. Feng, G. Cai, D. Wu and J. Lu, Foods, 2021, 10(12), 3051 Search PubMed.
- H. Jiang, S. Zhong, P. Schwarz, B. Chen and J. Rao, Food Chem., 2023, 400, 134016 Search PubMed.
- L. Shcherbakova, O. D. Mikityuk, L. Arslanova, A. A. Stakheev, D. Erokhin and С. К. Zavriev, et al., Front. Microbiol., 2021, 12, 629426 Search PubMed.
- T. Gao, H. Zhou, W. Zhou, L. Hu, J. Chen and Z. Shi, Molecules, 2016, 21(6), 770 Search PubMed.
- F. Bakkali, S. Averbeck, D. Averbeck and M. Idaomar, Food Chem. Toxicol., 2008, 46(2), 446–475 CrossRef PubMed.
- J. Wan, S. Zhong, P. Schwarz, B. Chen and J. Rao, Food Chem., 2019, 291, 199–206 Search PubMed.
- A. Puškárová, M. Bučková, L. Kraková, D. Pangallo and K. Kozics, Sci. Rep., 2017, 7(1), 8211 CrossRef PubMed.
- R. E. Valle-Gough, B. Y. Samaniego-Gámez, L. Cervantes-Díaz, S. U. Samaniego-Gámez, R. Garruña-Hernández and F. Núñez-Ramírez, et al., Rev. Fac. Agron. Luz, 2021, 38(3), 585–607 Search PubMed.
- X. Qi, S. Zhong, P. Schwarz, B. Chen and J. Rao, Ind. Crops Prod., 2023, 197, 116575 Search PubMed.
- M. T. Liu, J. Zhou, J. K. Qin, Z. Qin, J. Jiang and F. Yu, et al., Foods, 2024, 13(19), 3206 Search PubMed.
- D. Suzuki, Y. Sato, A. Mori and H. Tamura, npj Sci. Food, 2021, 5(1), 16 Search PubMed.
- M. Xiao, F. Zheng, M. Xiao, Q. An, H. Wang and Q. Dai, Flavour Fragrance J., 2021, 37(2), 83–95 CrossRef.
- A. Amanpour, A. S. Sonmezdag, H. Kelebek and S. Selli, Food Chem., 2015, 182, 251–256 CrossRef PubMed.
- J. Wan, Z. Jin, S. Zhong, P. Schwarz, B. Chen and J. Rao, Food Chem., 2020, 312, 126120 CrossRef PubMed.
- J. Wan, S. Zhong, P. Schwarz, B. Chen and J. Rao, Food Control., 2019, 106, 106709 Search PubMed.
- L. Shcherbakova, O. Mikityuk, L. Arslanova, A. Stakheev, D. Erokhin, S. Zavriev and V. Dzhavakhiya, Front. Microbiol., 2021, 12, 629429 CrossRef PubMed.
- M. Rosenberg, N. F. Azevedo and A. Ivask, Sci. Rep., 2019, 9(1), 6483 Search PubMed.
- W. Sun, W. Ma, J. Ji, Y. Wang, B. Bi and J. Zhang, et al., Australas. Plant Pathol., 2025, 1–20 Search PubMed.
- Q. OuYang, X. Duan, L. Li and N. Tao, Front. Microbiol., 2019, 10, 55 CrossRef PubMed.
- X. Kong, A. D. van Diepeningen, T. A. J. van der Lee, C. Waalwijk, J. Xu and J. Xu, et al., Front. Microbiol., 2018, 9, 654 Search PubMed.
- Y. Leng, C. Wu, Z. Liu, T. L. Friesen, J. B. Rasmussen and S. Zhong, Mol. Plant Pathol., 2011, 12(3), 289–298 Search PubMed.
- K. A. Thompson-Witrick, R. L. Rouseff, K. R. Cadawallader, S. E. Duncan, W. N. Eigel and J. M. Tanko, et al., J. Food Sci., 2015, 80(3), C571–C576 Search PubMed.
- X. Liu, X. Du, Y. Yang, H. Wei, F. He and F. Chen, et al., LWT–Food Sci. Technol., 2022, 171, 114152 Search PubMed.
- G. Nieto, Plants, 2020, 9(8), 961 Search PubMed.
- R. Sakthivel, K. Ramesh, R. Purnachandran and P. Mohamed Shameer, Renewable Sustainable Energy Rev., 2018, 82, 2970–2992 Search PubMed.
- D. Singh, D. Sharma, S. L. Soni, S. Sharma and D. Kumari, Fuel, 2019, 253, 60–71 Search PubMed.
- M. Retajczyk and A. Wróblewska, Catalysts, 2019, 9(6), 508 Search PubMed.
- A. J. Poulose and R. Croteau, Arch. Biochem. Biophys., 1978, 187(2), 307–314 Search PubMed.
- D. J. McClements, Soft Matter, 2012, 8(6), 1719–1729 RSC.
- D. J. McClements, Food Emulsions: Principles, Practices, and Techniques, CRC Press, Boca Raton, FL, 2004 Search PubMed.
- M.-J. Tsai, W.-Y. Chang, I.-H. Chiu, I.-L. Lin and P.-C. Wu, Pharmaceutics, 2023, 15(5), 1446 CrossRef PubMed.
- T. Liu and L. Liu, Int. J. Biol. Macromol., 2020, 162, 1509–1515 CrossRef PubMed.
- J. Rao and D. J. McClements, Food Chem., 2012, 134(2), 749–757 CrossRef PubMed.
- A. Ranjbar, A. Ramezanian, S. Shekarforoush, M. Niakousari and S. Eshghi, LWT–Food Sci. Technol., 2022, 161, 113303 CrossRef.
- J. Rao, B. Chen and D. J. McClements, Annu. Rev. Food Sci. Technol., 2019, 10(1), 365–387 CrossRef PubMed.
- Y. Cui, M.-H. Chen, L. Guishan, D. Wen-gui, Q. Li and R. Zou, et al., Chin. J. Org. Chem., 2022, 42(11), 3784 CrossRef.
- M. G. Donadu, Y. Peralta-Ruiz, D. Usai, F. Maggio, J. B. Molina-Hernandez and D. Rizzo, et al., J. Fungi, 2021, 7(5), 383 CrossRef PubMed.
- J. Ruiz-Duran, R. Torres, E. E. Stashenko and C. Ortiz, Antibiotics, 2023, 12(4), 668 CrossRef PubMed.
- M. Cristani, M. D'Arrigo, G. Mandalari, F. Castelli, M. G. Sarpietro and D. Micieli, et al., J. Agric. Food Chem., 2007, 55(15), 6300–6308 CrossRef PubMed.
- C. Li, C. Zhang, X. Chen, H. Cui and L. Lin, Front. Microbiol., 2022, 13, 855905 CrossRef PubMed.
- J. G. Vasselli and B. D. Shaw, Fungal Biol. Rev., 2022, 41, 2–9 CrossRef.
- M. M. G. Yamamoto-Ribeiro, R. Grespan, C. Y. Kohiyama, F. D. Ferreira, S. A. G. Mossini and E. L. Silva, et al., Food Chem., 2013, 141(3), 3147–3152 CrossRef PubMed.
- T. A. Gill, J. Li, M. Saenger and S. R. Scofield, J. Appl. Microbiol., 2016, 121(4), 1103–1116 CrossRef PubMed.
- X. Wang, Z. Wang, Y. Zhao, H. Wang, M. Xiang and Y. Liu, et al., Nat. Prod. Bioprospect., 2023, 13(1), 10 CrossRef PubMed.
- E. A. Elsherbiny, D. H. Dawood and N. A. Safwat, Pestic. Biochem. Physiol., 2021, 171, 104721 CrossRef PubMed.
- FDA, USFDA, Washington, DC, 2000 Search PubMed.
- H. M. Shakam, A. A. Mohamed and M. Z. M. Salem, Arch. Phytopathol. Plant Prot., 2022, 55(8), 951–972 CrossRef.
- J. Shao, Z. Pei, H. Jing, L. Wang, C. Jiang and X. Du, et al., Physiol. Mol. Plant Pathol., 2021, 114, 101635 CrossRef.
- Y. Duan, F. Lu, Z. Zhou, H. Zhao, J. Zhang and Y. Mao, et al., J. Hazard. Mater., 2020, 398, 122908 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |