Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Optimizing drying techniques in euryhaline microalgae to reveal strain-specific phenolic signatures and amplified antioxidant potency
Manpreet Kaur *ab,
Surekha Bhatiac,
Kiran Tewarid and
Urmila Guptae
*ab,
Surekha Bhatiac,
Kiran Tewarid and
Urmila Guptae
aDepartment of Food and Human Nutritional Sciences, University of Manitoba, Winnipeg, R3T 2N2, Canada. E-mail: Manpreet.kaur3@umanitoba.ca; Tel: +1-4313343308
bDepartment of Biochemistry, Punjab Agricultural University, Ludhiana, Punjab 141004, India. E-mail: manibuttar12@gmail.com
cDepartment of Processing and Food Engineering, Punjab Agricultural University, Ludhiana, Punjab 141004, India. E-mail: surekhabhatia@pau.edu
dResearch & Development, Chemical Resources (CHERESO), Panchkula, Haryana, India. E-mail: admin.pkl@chereso.net
eDepartment of Renewable Energy Engineering, Punjab Agricultural University, Ludhiana, Punjab 141004, India. E-mail: urmilphutela@pau.edu
First published on 12th March 2026
Abstract
This research investigated the impact of three distinct drying modalities—freeze-drying (FD), oven drying (OD), and shade drying (SD)—used for desiccating microalgal biomass on phenolic composition and antioxidant properties in a range of euryhaline microalgal strains, including Spirulina subsalsa (B6), Anabaena sp. (B7), and Chlorella sp. (B8), as well as freshwater strains Spirulina NCIM (SN), Chlorella NCIM (CN), and commercially cultivated Spirulina (BR). Irrespective of drying methods, a notably diminished quantity of aggregate polyphenols in both free and bound states was found within the freshwater and commercially produced strains, compared with their euryhaline counterparts. Amongst the euryhaline strains, Anabaena out with significantly higher phenol and flavonoid (p < 0.05) contents under FD and OD, followed by Chlorella and Spirulina. Among the drying methods evaluated, OD demonstrated the best preservation of bound phenols and the highest DPPH and superoxide scavenging activities, which were strongly correlated (r = 0.88 and r = 0.86, respectively) with the former. FD exhibited higher total reducing power and higher free phenolics, showing a robust positive correlation (r = 0.97). Nitric oxide scavenging activities correlated positively with total flavonoids, with high correlation coefficients observed under FD (r = 0.97) and OD (r = 0.95). Furthermore, euryhaline microalgal strains showed significantly higher antioxidant potential than commercial and freshwater strains (p < 0.05). Amongst the euryhaline strains, B7 demonstrated markedly enhanced DPPH scavenging activity, especially under OD (63.06%) attributed to increased bound phenols. These findings provide invaluable insights into the confluence of strain typology and drying methodologies vis-à-vis the bioactive properties of euryhaline microalgae, imparting potential utility across diverse industries.
Sustainability spotlightThis study advances sustainable bioprocessing by identifying eco-efficient drying strategies that preserve the functional integrity of microalgal bioactive compounds, thereby reducing waste and enhancing the value of biomass. By examining both euryhaline and freshwater strains, the work underscores the potential of resilient microalgae as a renewable source of phenolic-rich ingredients for food, nutraceutical, and bioproduct applications. The insights gained support the development of low-energy, high-retention drying pathways that align with circular bioeconomy goals, promote resource-efficient processing, and enable the production of high-quality, nature-derived antioxidants through environmentally responsible practices. |
1 Introduction
Bioactive compounds, encompassing flavonoids, carotenoids, vitamins, and assorted phenolic compounds, are prevalent in significant quantities within vegetables and fruits.1 These phytochemical constituents exhibit robust antiradical and antioxidant properties, effectively mitigating the risk of pathogenic processes instigated by free radicals.2 A recent surge in awareness regarding bioactive constituents and their potential health benefits has led to a growing preference for alternative dietary supplements enriched with functionally potent bioactive compounds. In response to this evolving market trend, food processors are intensifying their focus on products infused with a higher content of bioactive chemicals, diligently striving to optimize their retention throughout the intricacies of food processing.Microalgae, with a specific emphasis on Spirulina and Chlorella, have garnered acclaim as reservoirs of phenolic compounds, an intricate group encompassing phenols and flavonoids.3–5 Polyphenols are esteemed for their robust antioxidant attributes and their prospective advantages to human health. The profusion of these phenolic compounds within microalgal biomass renders them alluring candidates for integration into dietary supplementation regimens and the formulation of functional food products aimed at bolstering overall well-being. Beyond their nutritional appeal, these microalgae are emblematic of a burgeoning interest in harnessing natural and sustainable reservoirs of antioxidants, aligning with the prevailing global focus on sustainable resource utilization. Nevertheless, due to their vulnerability to deterioration and high moisture content, preserving microalgae presents challenges. Consequently, various drying methods and storage systems are employed to prolong the shelf life of microalgae.
The most common approach for commercializing microalgae is to dry the biomass since it improves product durability and stability and makes storage and shipping easy.6 Although drying is widely used for microalgal commercialization, it is an energy-intensive and cost-intensive step. While wet biomass can be processed directly in applications such as biofuels or animal feed to reduce dehydration costs, its high moisture content limits shelf life, transportability, and suitability for nutraceutical and functional food uses. Moreover, wet versus dried processing markedly influences the stability and retention of bioactive compounds, underscoring the importance of optimizing drying strategies for high-value applications.
Microalgal industries use a variety of dryers. Spray drying is the main drying technique utilized, although ovens with forced ventilation are often employed in small microalgal farms.7 However, various methods of drying biomass, including open-air drying, freeze-drying, oven-drying, sun or shade drying, and others, are also utilized.8 Drying methods profoundly influence the preservation of phenols and flavonoids. It is widely known that the drying method and conditions employed can impact the integrity of microalgal biomass, potentially leading to a reduction in its quality. For instance, the process of drying can bring about changes in the characteristics of microalgae, and in the context of the food industry, there is a possibility of diminished nutritional quality as a result of the depletion of bioactive components. Freeze-drying (FD) gently preserves the cellular structure, leading to higher levels of free-form phenolic compounds and flavonoids due to reduced enzymatic activity.9 Shade drying (SD) retains some phenolic compounds under milder conditions. In contrast, oven drying (OD), though effective for bound phenolic compounds, risks the thermal degradation of free forms.10
The antioxidant efficacy of a substance, like microalgae, is intricately shaped by the abundance of polyphenols and their partitioning between free and bound states. In essence, a greater polyphenolic quantity augments antioxidant potential, as more molecules are accessible for quenching free radicals and safeguarding cellular integrity.11 In one of our previous studies, we investigated the polyphenol-rich potential of a salt-tolerant microalga and assessed the influence of various drying strategies on antioxidant properties and drying kinetics.12 Moreover, the interplay between free and bound polyphenol forms modulates the overall antioxidant prowess. A higher proportion of free-form polyphenols provides rapid and strong antioxidant activity by quickly interacting with and neutralizing free radicals. In contrast, bound-form polyphenols are less immediately active. However, they can be gradually released by enzymatic and metabolic processes in the body, thereby offering sustained antioxidant protection over time. Therefore, despite its economic and energetic constraints, drying remains a critical step for ensuring the functional integrity and commercial viability of microalgal bioactive compounds.
The objectives of this study were to comprehensively examine the influence of different drying methods on the phenolic and flavonoid profiles of euryhaline microalgal strains and to elucidate how these compositional changes translate into variations in antioxidant functionality. By integrating biochemical characterization with functional antioxidant assessment, this work aims to establish a mechanistic understanding of drying-induced modifications in microalgal bioactive compounds. The outcomes of this study are intended to provide a rational and application-oriented basis for selecting drying strategies aligned with specific functional objectives, thereby enabling efficient utilization of resources and supporting informed decision-making for technological investment in microalgae-based nutraceutical and functional food applications.
2 Materials and methods
2.1 Materials
The different euryhaline microalgal strains Spirulina subsalsa (B6), Anabaena sp. (B7), and Chlorella sp. (B8) formerly isolated from waterlogged areas of southwest Punjab,13 and two freshwater microalgal strains Spirulina NCIM (S-N) and Chlorella NCIM (C-N) were procured from the Biogas Laboratory, Department of Renewable Energy, College of Agricultural Engineering and Technology, Punjab Agricultural University, Ludhiana, Punjab (India). Spirulina sp. (Spi B) was purchased from Life Green Biotech, Nohar, 335523, Rajasthan, India.2.2 Cultivation of microalgae
These different cultures of microalgae were maintained under optimized conditions in suitable media and pH as per our previous work12 under a luminescence of 54 µ mol photons per m2 per s with a 16–8 hour light–dark photoperiod in 50 L capacity covered fish tanks (Fig. 1). The samples were imaged using a Magnus Icon 528,293 Freedom Model Microscope and identified using the ToupView software program (Olympus, Tokyo, Japan) (Fig. 2). After 30 days, biomass was harvested by sedimentation and filtration as per the process reported in our previous work.14 The collected biomass was further used for drying purposes.2.3 Drying procedure
We utilized various drying techniques, including oven drying (OD), freeze-drying (FD), and shade drying (SD), to desiccate thin layers of microalgal biomass. For OD, we employed a laboratory-type drying oven (Macneill and Magor Ltd, Kolkata, India) operating at a temperature of 60 °C and an air velocity of 1 m s−1. FD was carried out using a laboratory-type freeze dryer (MSW-137, Macro Scientific Works Pvt Ltd, India) with a condenser temperature maintained at −40 °C and a continuous pressure of 10 Pa within the drying chamber. As for SD, we naturally dried samples in the shade, keeping the temperature controlled at 27 ± 2 °C. For each trial (n = 3), 30 g of microalgal paste was evenly spread on a glass Petri dish, maintaining a uniform biomass thickness of 3 mm. The weight of the biomass and the corresponding time were recorded at regular intervals. Drying continued until the sample reached a constant mass, indicating moisture equilibrium.2.4 Analysis of free and bound polyphenols
Dried microalgal biomass weighing 0.5 g was mixed with 8 millilitres of 80% methanol and then subjected to reflux at 80 °C for 10 minutes. Afterwards, the mixture underwent agitation and filtration through Whatman no. 1 filter paper, with the resulting liquid collected and supplemented with an additional 80% methanol to achieve a final volume of 10 millilitres. The remaining residue was subjected to a 90 minute digestion with 4 M sodium hydroxide at a temperature of 25 °C. Subsequently, hydrochloric acid was introduced, and the extraction process was carried out using ethyl acetate for ten iterations. The extracted material was ultimately dried in an oven at 45 °C. The quantification of total phenolic content (TPC) was performed using the Folin–Ciocalteu method, and the results were expressed as milligrams per gram of dry weight (mg g−1 DW).15 To assess the total flavonoid content (TFC), we utilized the aluminium chloride colourimetric method,16 and the results are reported in milligrams per gram of dry weight (mg g−1 DW).2.5 Analysis of different antioxidant activities
The assessment of the hydroxyl radical scavenging assay (HRSA) followed the protocol of Li et al.17 DPPH free radical scavenging activity (DPPHSA) (%) was estimated according to the methodology of Blois.18 Superoxide anion scavenging activity (SARSA) (%) was determined as per the procedure outlined by Marklund and Marklund.19 The nitric oxide radical scavenging assay (NORSA) (%) adhered to the method devised by Marcocci et al.20 Ferrous (Fe2+) ion chelation activity (FICA) (%) was estimated following the method of Minotti and Aust.21 Total reducing power (TRP) was determined by the method of Sreeramulu et al.22 and expressed as milligrams per gram of dry weight (mg g−1 DW). Ferric reducing antioxidant power (FRAP) was quantified using the protocol of Benzie and Devaki23 and expressed as milligrams of Fe2+ per gram of dry weight (mg Fe2+/g DW). Hydrogen peroxide scavenging activity (HPSA) (%) was carried out following the procedure of Borquaye et al.242.6 Statistical analysis
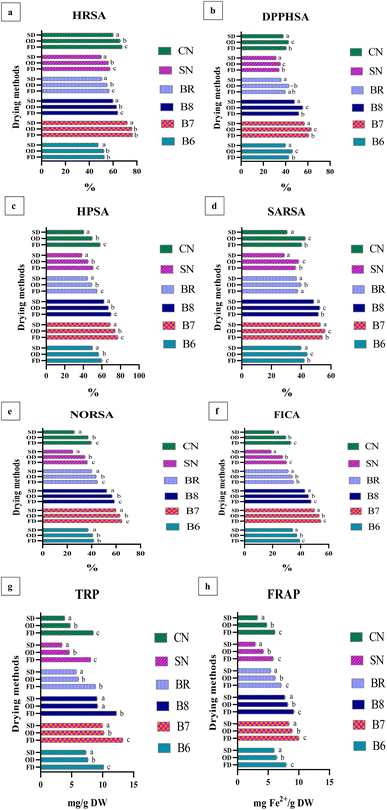
The gathered data underwent statistical analysis using Analysis of Variance (ANOVA) within RStudio version 4.2.0 (RStudio, Inc., Boston, MA). To evaluate differences among the mean values, Tukey's multiple comparison test was employed, with a significance threshold set at p ≤ 0.05. Correlation analysis was performed by calculating Pearson's correlation coefficient. In the figures, distinct letters marked as superscripts indicate statistically significant differences among the microalgal strains during various stages of drying, with a significance level of p ≤ 0.05.3 Results
3.1 Effect of different drying methods on the levels of phenols and flavonoids of the studied microalgae strains
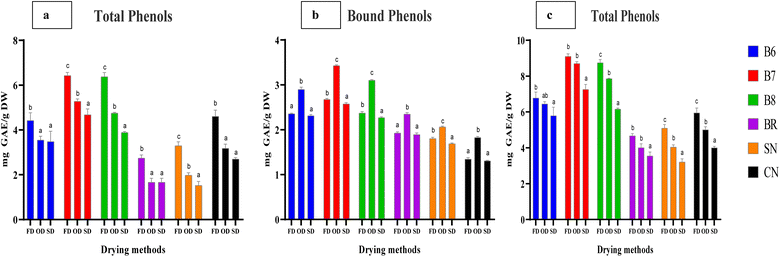
The variations in the levels of phenols and flavonoids in euryhaline Spirulina subsalsa (B6), Anabaena sp. (B7), and Chlorella sp. (B8), commercial Spirulina (BR), and freshwater Spirulina (SN) and Chlorella (CN) treated with different drying methods are presented in Table 1 and Fig. 3, 4, respectively. The levels of phenols and flavonoids were significantly higher in salt-tolerant strains as compared to the freshwater strains (SN and CN) and the commercial strain (BR) (p < 0.05). Under FD, higher levels of free phenols (FP) were observed in strain B7 (6.43 mg gallic acid equivalents (GAE) g−1 DW) (Fig. 3a). On the other hand, euryhaline Spirulina B6 (4.42 mg GAE g−1 DW) showed a significantly high level of FP as compared to commercial Spirulina BR (2.74 mg GAE g−1 DW) and freshwater Spirulina SN (3.29 mg GAE g−1 DW) (p < 0.05) (Fig. 3a). Likewise, euryhaline Chlorella B8 showed a significantly higher FP (6.37 mg GAE g−1 DW) than the freshwater Chlorella (4.61 mg GAE g−1 DW). The levels of bound and total phenols were also recorded significantly higher in salt-tolerant strains B7 (2.68 and 9.10 mg GAE g−1) followed by B8 (2.37 and 8.75 mg GAE g−1) and B6 (2368 and 6.78 mg GAE g−1) as compared to freshwater strains SN (1.80 and 5.10 mg GAE g−1), CN (1.35 and 5.95 mg GAE g−1) and commercial BR (1.93 and 4.67 mg GAE g−1) (p < 0.05) (Table 1 and Fig. 3b, c). Similar trends among different microalgal strains were also observed in phenolic levels under OD and SD.| MS | Freeze drying (FD) | Oven drying (OD) | Shade drying (SD) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| FP | BP | TP | FP | BP | TP | FP | BP | TP | |
| a FD: freeze-drying; OD: oven drying; SD: shade drying; FP: free phenolics; BP: bound phenolics; FF: free flavonoids; BF: bound flavonoids; TP: total phenolics; TF: total flavonoids; TP + TF: total phenolics + total flavonoids. | |||||||||
| B6 | 4.42 ± 0.345b | 2.36 ± 0.01d | 6.78 ± 0.33c | 3.54 ± 0.17b | 2.90 ± 0.04d | 6.44 ± 0.12c | 3.47 ± 0.46c | 2.31 ± 0.02de | 5.79 ± 0.47c |
| B7 | 6.43 ± 0.138c | 2.68 ± 0.01e | 9.10 ± 0.13d | 5.27 ± 0.10d | 3.42 ± 0.01g | 8.70 ± 0.10ef | 4.67 ± 0.26e | 2.57 ± 0.02f | 7.25 ± 0.28e |
| B8 | 6.37 ± 0.183c | 2.37 ± 0.03d | 8.75 ± 0.18d | 4.75 ± 0.03c | 3.10 ± 0.01e | 7.85 ± 0.02d | 3.88 ± 0.04cd | 2.27 ± 0.01d | 6.15 ± 0.06cd |
| BR | 2.74 ± 0.143a | 1.93 ± 0.02c | 4.67 ± 0.12a | 1.65 ± 0.18a | 2.34 ± 0.03c | 4.00 ± 0.21a | 1.65 ± 0.18a | 1.89 ± 0.03c | 3.54 ± 0.21ab |
| SN | 3.29 ± 0.173a | 1.80 ± 0.03b | 5.10 ± 0.20a | 1.98 ± 0.10a | 2.06 ± 0.01b | 4.04 ± 0.12a | 1.52 ± 0.18a | 1.68 ± 0.01b | 3.20 ± 0.18a |
| CN | 4.61 ± 0.279b | 1.34 ± 0.03a | 5.95 ± 0.27b | 3.17 ± 0.18b | 1.82 ± 0.02a | 5.00 ± 0.16b | 2.69 ± 0.06b | 1.30 ± 0.01a | 4.00 ± 0.07b |
| MS | Flavonoids | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| FF | BF | TF | FF | BF | TF | FF | BF | TF | |
| B6 | 0.91 ± 0.014c | 0.47 ± 0.034b | 1.38 ± 0.047b | 0.77 ± 0.006c | 0.54 ± 0.004b | 1.31 ± 0.010b | 0.76 ± 0.004c | 0.47 ± 0.007b | 1.24 ± 0.007b |
| B7 | 1.53 ± 0.012h | 1.10 ± 0.011f | 2.63 ± 0.017h | 1.38 ± 0.008g | 1.20 ± 0.007h | 2.58 ± 0.007f | 1.36 ± 0.008g | 0.99 ± 0.004h | 2.35 ± 0.013g |
| B8 | 1.24 ± 0.012e | 0.88 ± 0.019d | 2.12 ± 0.026e | 1.11 ± 0.005e | 0.91 ± 0.004e | 2.03 ± 0.008e | 1.06 ± 0.010e | 0.78 ± 0.008f | 1.84 ± 0.014e |
| BR | 1.04 ± 0.007d | 0.69 ± 0.019c | 1.73 ± 0.023d | 0.94 ± 0.006d | 0.74 ± 0.005d | 1.68 ± 0.001d | 0.90 ± 0.006d | 0.61 ± 0.009d | 1.52 ± 0.014d |
| SN | 0.73 ± 0.005a | 0.35 ± 0.002a | 1.08 ± 0.007a | 0.67 ± 0.002a | 0.37 ± 0.003a | 1.05 ± 0.003a | 0.66 ± 0.002a | 0.28 ± 0.002a | 0.94 ± 0.001a |
| CN | 0.870.004b | 0.64 ± 0.016c | 1.51 ± 0.016c | 0.72 ± 0.004b | 0.66 ± 0.006c | 1.39 ± 0.010c | 0.71 ± 0.005b | 0.56 ± 0.003c | 1.28 ± 0.005c |
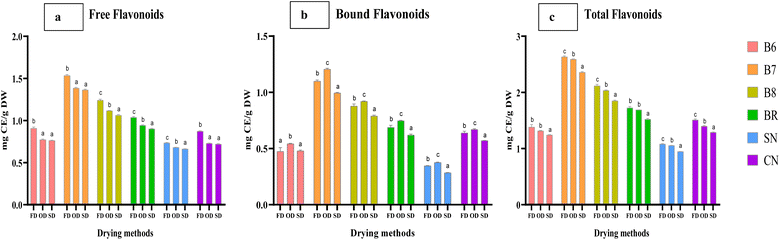
Significant differences were also recorded in phenolic levels between all the drying methods (p < 0.05). Higher levels of FP and TP were observed in freeze-dried strains with an average level of 4.64 and 6.72 mg GAE g−1 DW respectively (p < 0.05), whereas BP levels were significantly higher under OD (2.61 mg GAE g−1 DW) as compared to FD (2.07 mg GAE g−1 DW) and SD (2.00 mg GAE g−1 DW) (p < 0.05) (Fig. 3a–c). Under FD, strains B7 and B8 were observed to have higher levels of FF (1.53 and 1.24 mg CE g−1), BF (1.10 and 0.88 mg CE g−1), and TF (2.63 and 2.12 mg CE g−1) as compared to SN (0.73 mg GAE g−1), and CN (0.87 mg GAE g−1).
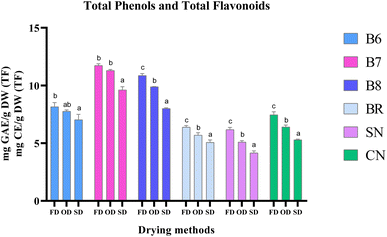
The commercial strain BR had higher levels of FF, BF, and TF (1.04, 0.69, and 1.73 mg CE g−1 respectively) as compared to SN and CN with lower levels than salt-tolerant strains B7 and B8. Similar to phenols, FF and TF levels were significantly higher in FD i.e., average levels of 1.05 and 1.74 mg CE g−1 respectively, whereas BF levels were significantly higher under OD (0.74 mg CE g−1) as compared to FD (0.68 mg CE g−1) and SD (0.62 mg CE g−1) (p < 0.05) (Fig. 4a–c). The levels of TP + TF of the studied strains B6, B7, B8, BR, SN, and CN are depicted in Table 2 and Fig. 5. Under FD, the highest level of TP + TF was observed in salt tolerant strain B7 (11.73 ± 0.152 mg g−1), followed by B8 (10.86 ± 0.16 mg g−1), freshwater Chlorella (CN) (7.46 ± 0.26 mg g−1), euryhaline Spirulina B6 (8.16 ± 0.36 mg g−1), commercial Spirulina (BR) (6.39 ± 0.13 mg g−1) and freshwater SN (6.17 ± 0.2 mg g−1) (p < 0.05). Similar results were also observed under OD with higher levels in euryhaline microalgal strains including Anabaena B7 (11.29 ± 0.1 mg g−1), followed by Chlorella B8 (9.88 ± 0.02 mg g−1) and Spirulina B6 (7.76 ± 0.12 mg g−1), which was significantly higher than those of the corresponding commercial and freshwater microalgal strains (p < 0.05). Further, the antioxidant activities were measured.
| FD | OD | SD | |
|---|---|---|---|
| B6 | 8.16 ± 0.361c | 7.76 ± 0.124d | 7.03 ± 0.466c |
| B7 | 11.73 ± 0.152e | 11.29 ± 0.100g | 9.60 ± 0.285e |
| B8 | 10.86 ± 0.164d | 9.88 ± 0.027e | 8.00 ± 0.065d |
| BR | 6.39 ± 0.134a | 5.69 ± 0.218b | 5.07 ± 0.211b |
| SN | 6.17 ± 0.200a | 5.09 ± 0.123a | 4.15 ± 0.180a |
| CN | 7.46 ± 0.260b | 6.40 ± 0.167c | 5.28 ± 0.072b |
3.2 Antioxidant properties
The antioxidant properties of the studied microalgal strains under freeze-drying (FD), oven-drying (OD), and shade-drying (SD) are presented in Fig. 6 and Table 3. Significant differences (p < 0.05) were observed among the salt-tolerant strains (B7 and B8), freshwater strains (SN and CN), and the commercial strain (BR) across all drying techniques.| Strains | HRSA | DPPHSA | HPSA | SARSA | NORSA | FICA | TRP | FRAP |
|---|---|---|---|---|---|---|---|---|
| a HRSA: hydroxyl radical scavenging activity; DDPHSA: DPPH free radical scavenging activity; HPSA: hydrogen peroxide scavenging activity; SARSA: superoxide anion radical scavenging activity; NORSA: nitric oxide radical scavenging activity; FICA: ferrous ion chelating activity; TRP: total reducing power; FRAP: ferric reducing antioxidant power. | ||||||||
| Freeze drying | ||||||||
| B6 | 52.65 ± 0.20a | 42.73 ± 0.35d | 59.55 ± 1.11d | 42.00 ± 0.20d | 41.64 ± 0.31c | 39.41 ± 0.13d | 10.16 ± 0.03d | 7.91 ± 0.12c |
| B7 | 76.34 ± 0.06h | 60.63 ± 0.37g | 77.04 ± 0.39g | 54.05 ± 0.27f | 64.73 ± 0.17h | 54.35 ± 0.33g | 13.24 ± 0.05f | 9.99 ± 0.05e |
| B8 | 63.93 ± 0.20d | 51.82 ± 0.16e | 69.71 ± 0.48e | 51.46 ± 0.21e | 58.66 ± 0.20e | 47.36 ± 0.28e | 12.23 ± 0.02e | 9.16 ± 0.15d |
| BR | 56.58 ± 0.20b | 39.65 ± 0.25b | 54.73 ± 0.14b | 37.49 ± 0.29b | 44.56 ± 0.37d | 34.67 ± 0.54c | 8.82 ± 0.05c | 7.08 ± 0.05b |
| SN | 57.37 ± 0.09c | 34.09 ± 0.49a | 50.41 ± 0.13a | 36.07 ± 0.43a | 36.44 ± 0.09a | 29.75 ± 0.49a | 8.08 ± 0.01b | 5.80 ± 0.12a |
| CN | 67.55 ± 0.15f | 40.54 ± 0.28c | 58.02 ± 0.36c | 40.17 ± 0.35c | 39.58 ± 0.51b | 32.95 ± 0.49b | 8.49 ± 0.03a | 6.09 ± 0.02a |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||
| Oven drying | ||||||||
| B6 | 52.24 ± 0.20a | 46.10 ± 0.27c | 56.40 ± 0.24c | 43.91 ± 0.29d | 40.76 ± 0.31c | 37.28 ± 0.20d | 7.61 ± 0.05c | 6.36 ± 0.15c |
| B7 | 75.89 ± 0.08h | 63.06 ± 0.31ef | 74.07 ± 0.35f | 55.88 ± 0.23f | 63.07 ± 0.16h | 53.11 ± 0.37g | 10.17 ± 0.06f | 8.89 ± 0.10e |
| B8 | 62.95 ± 0.25d | 55.35 ± 0.37d | 66.83 ± 0.39d | 52.43 ± 0.26e | 56.61 ± 0.35e | 45.52 ± 0.36e | 9.15 ± 0.08d | 8.15 ± 0.10d |
| BR | 54.92 ± 0.33b | 42.93 ± 3.19bc | 48.98 ± 0.44b | 39.69 ± 0.41b | 43.60 ± 0.27d | 33.96 ± 0.62c | 6.07 ± 0.06b | 6.09 ± 0.12c |
| SN | 56.05 ± 0.20c | 35.34 ± 0.18a | 44.94 ± 0.32a | 38.06 ± 0.54a | 34.43 ± 0.45a | 26.97 ± 0.46a | 4.63 ± 0.03a | 4.18 ± 0.10a |
| CN | 65.86 ± 0.35f | 42.57 ± 0.26b | 49.44 ± 0.27b | 42.61 ± 0.35c | 37.05 ± 0.28b | 29.40 ± 0.33b | 4.77 ± 0.03a | 4.75 ± 0.05b |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||
| Shade drying | ||||||||
| B6 | 47.45 ± 0.09a | 39.81 ± 0.48d | 49.76 ± 0.40d | 39.73 ± 0.59d | 36.97 ± 0.51c | 34.32 ± 0.17d | 7.28 ± 0.06d | 5.97 ± 0.05d |
| B7 | 71.89 ± 0.40e | 56.81 ± 0.37g | 68.73 ± 0.48g | 52.80 ± 0.26f | 59.88 ± 0.17h | 49.85 ± 0.28g | 9.97 ± 0.03g | 8.35 ± 0.13f |
| B8 | 59.86 ± 0.47c | 47.97 ± 0.23e | 62.24 ± 0.23e | 48.29 ± 0.28e | 52.33 ± 0.18e | 42.91 ± 0.33e | 9.08 ± 0.05e | 7.71 ± 0.10e |
| BR | 50.35 ± 0.49b | 35.87 ± 0.49b | 44.34 ± 0.48c | 36.81 ± 0.13c | 39.93 ± 0.17d | 30.94 ± 0.62c | 5.74 ± 0.03c | 5.38 ± 0.07c |
| SN | 50.01 ± 0.40b | 31.45 ± 0.20a | 38.35 ± 0.34a | 28.61 ± 0.17a | 24.61 ± 0.15a | 18.90 ± 0.57a | 3.43 ± 0.02a | 2.88 ± 0.13a |
| CN | 59.94 ± 0.40c | 37.90 ± 0.13c | 40.30 ± 0.42b | 30.39 ± 0.43b | 25.75 ± 0.48b | 21.04 ± 0.26b | 3.88 ± 0.02b | 3.22 ± 0.07b |
Overall, salt-tolerant strains, particularly B7 and B8, exhibited superior antioxidant activities across all assays and drying methods. Among the drying techniques, OD promoted higher bound phenolics contributing to DPPHSA, HPSA, and SARSA, while FD preserved free phenolics supporting TRP and FRAP activities. Freshwater and commercial strains consistently displayed lower antioxidant potential. Statistical analyses confirmed significant differences between strains and drying methods (p < 0.05) (Fig. 6a–h).
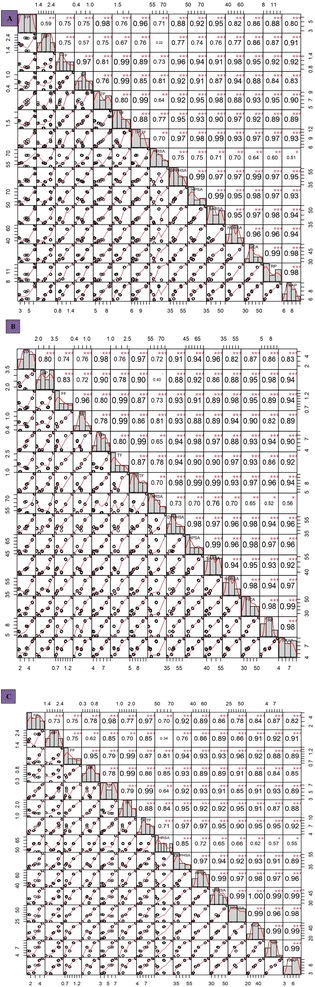
3.3 Correlation between phenols, flavonoids, and antioxidant properties of the studied microalgal strains under diverse drying methods
The impact of different drying methods on free, bound, and total phenols and different antioxidant properties in euryhaline, commercial, and freshwater microalgal strains was assessed using correlation analysis (Fig. 7A–C) in order to study the role of different drying methods on antioxidant components. Under different drying, a positive correlation was observed between the levels of the studied phenols, flavonoids, and antioxidant activities of salt tolerant (B6, B7, and B8), freshwater (SN and CN), and commercial strains (BR) (Fig. 7A–C), indicating an increase in the antioxidant activities with an increase in the levels of phenols and flavonoids or vice versa (p < 0.05). The correlation between phenols, flavonoids, and antioxidant activities was highly significant (p < 0.05).Under FD, more free form is retained, and a strong positive correlation is reported between free forms and total polyphenols. Under FD, a strong positive correlation (r = 0.89) was reported between FF and TP + TF, further contributing to a strong TRP depicted by the correlation (r = 0.92) of FF and TRP (Fig. 7A). On the other hand, TRP and FF showed a correlation of r = 0.89 under OD. While talking about the BP, the positive correlation of r = 0.76 was reported with TP + TF under FD, whereas under OD, as previously reported, the bound form increased, which is also supported by a significant positive correlation of BP with TP + TF under OD (r = 0.90) (Fig. 6B). Likewise, a strong positive correlation was reported between TF and TP + TF under OD (r = 0.86) (Fig. 7B), although it was similar under FD (r = 0.85) (Fig. 7A). High DPPH and SARSA activities were reported under OD, which might be due to more retention of the bound form of polyphenols, also supported by correlation analysis. Under OD, BP and DPPHSA showed a positive correlation of r = 0.88, whereas it is r = 0.77 under FD. Similarly, BF and DPPHSA showed a positive correlation of r = 0.93 and r = 0.92 under OD and FD, respectively. This indicated that BP contributed more to DPPHSA than BF.
Correspondingly, a positive correlation (r = 0.86) was reported between BP and SARSA under OD, whereas this value is r = 0.76 under FD. While talking about flavonoids, a positive correlation of r = 0.89 between TF and SARSA under OD and r = 0.87 under FD was reported. Meanwhile, under SD, it followed the same trend but was comparatively weaker than FD in the case of free form. On the other hand, SD showed a positive correlation of r = 0.85 between BP and TP + TF (Fig. 7C).
3.4 Principal component analysis (PCA)
The scores of the first two principal components for the different drying conditions and biochemical constituents are depicted in Fig. 8. The first principal component accounted for 86.58% of the total variation, while the second principal component explained only 4.72% of the total variation. PC1 exhibited positive correlations with phenols, flavonoids, total phenols, and total flavonoids. It also showed positive associations with various antioxidant activities, including HRSA, DPPHSA, HPSA, SARSA, NORSA, FICA, TRP, and FRAP values. B7, B8, B18, and BS strains demonstrated higher levels of phenols, flavonoids, and antioxidant activities compared to commercial and freshwater samples. Conversely, BR, SN, CN, and B6 were positioned on the left-hand side of the plot, indicating lower levels of phenols, flavonoids, and antioxidant activities compared to the other strains.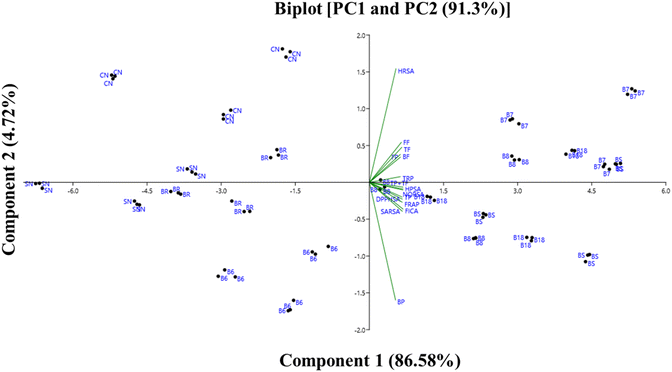 | ||
| Fig. 8 Principal Component Analysis (PCA) of phenols, flavonoids, and anti-oxidant activities of microalgal strains under different drying methods. | ||
4 Discussion
Several drying techniques and storage setups are used to extend microalgae's shelf life. The current research examined how different microalgae strains differed in terms of their phenolic, flavonoid, and antioxidant potential and how different drying techniques affected these factors. Based on the drying technique used, our results showed distinctive variations in the flavonoid and phenolic content as well as antioxidant activities. Our research revealed that as compared to the freshwater strains (S-N and C-N), the marine salt-tolerant strains (B-6, B-7, B-8, B-18, and B-S) had greater levels of phenol, flavonoid content, and antioxidative activities. This finding implies that marine strains have built-in defenses against salt-induced oxidative stress, which results in increased production of phenolic and antioxidant capacity.Additionally, the commercial strain, B-R, showed intermediate values between marine and freshwater strains, exhibiting its potential as a beneficial source of phenolics and antioxidative compounds. Salt-tolerant microalgae make more bioactive compounds to protect themselves from salt stress and strengthen their cells. These adaptations may enhance their polyphenol content compared to freshwater microalgae, which might be supported by their survival in saline environments due to an augmented antioxidant defense system, as reported in our previous study.14 The findings, which showed considerable variations in these parameters based on the drying technique utilized, are in line with those of Kolla et al.25 and Bransica et al.26 Similar to this, Meng et al.27 indicated that the color retention, total flavonoid, total phenolic content, fatty acids, water-soluble polysaccharide content, and antioxidant activity varied significantly in Dendrobium officinale depending on the drying process, with FD showing the most significant results.
With respect to phenolic forms, bound phenolics were more prevalent under OD than FD, while SD yielded comparable levels to FD, as supported by correlation analysis. This suggests that thermal input during oven drying may facilitate the release or formation of bound phenolics, resulting in their higher abundance. OD is often superior for preserving bound polyphenols due to higher temperatures, faster processing, and favorable matrix interaction,28 Similar to this, Lai et al.29 assessed the impact of five drying techniques on the antioxidant capacity and bioactive phytochemicals of navel orange peel, including shade drying (SD), freeze-drying (FD), microwave drying (MD), and hot-air oven drying at 50 and 70 °C, suggesting that OD showed increased bound form, whereas overall phenolics were preserved under FD. Additionally, Ma et al.30 revealed that FD outperformed heat pump drying and hot air drying by retaining a high total phenolic level (29.35 mg g−1) and exhibited the highest antioxidant activity, as evidenced by the highest FRAP (219.57 µmol TE g−1) and ABTS (127.15 µmol TE g−1).
The antioxidant performance of the dried samples appears to be strongly influenced by their free-bound phenolic fraction. The positive association between bound phenolics and DPPH radical scavenging suggests that bound polyphenols may act as more effective hydrogen or electron donors, thereby enhancing radical neutralization capacity. This is in accordance with the previous study where BP extracted from red quinoa exhibited stronger DPPH and ABTS antioxidant activities and had a lower IC50 (10.295 mg mL−1) value in inhibiting α-glucosidase activity.31 This contradicts the results of Mohapatra et al.,32 who discovered that FD treatment resulted in better physical and chemical properties, bioactive retention, and antioxidant potential than those processed using other techniques. On the other hand, higher levels of bound phenolics were also linked to greater SARSA, which was more under OD than FD, also depicted by a correlation of r = 0.86 and r = 0.76, respectively. The more efficient contribution of bound polyphenols to SARSA compared to free-form polyphenols may be associated with their improved interaction, enhanced stability, and greater bioavailability. Our research aligns with Huang et al.33 findings, demonstrating that the bound phenolic fraction extracted from Rosa roxburghii Tratt pomace exhibited superior scavenging activity in in vitro antioxidant assays compared to the free phenolic fraction.
Moreover, higher levels of free phenolics were correlated with increased reducing power as depicted from the correlation analysis under FD (r = 0.92). Free polyphenols, characterized by simpler structures and easy accessibility, react more rapidly with oxidative substances, resulting in greater reducing capacity. In contrast, polyphenols, often bound to other macromolecules, experience limited direct interaction with oxidizing agents. This is in accordance with the previous study, where it was reported that a major portion of longan pulp collected from South China was composed of free phenolics, which contributed substantially to its reducing power.34 Additionally, a previous study by Wani et al.35 reported that the highest FRAP activity was observed in apricot fruits after a storage period compared to the initial fruits. In accordance with our study, higher concentrations of flavonoids were associated with increased NORSA (r = 0.97 under FD and OD, r = 0.95 under SD) and HRSA (r = 0.77, r = 0.78, and r = 0.84 under FD, OD, and SD, respectively). NORSA is exclusive to flavonoids due to their distinct chemical structure and reactivity, which includes hydroxyl and functional groups enabling effective scavenging of these radicals. Conversely, other phenols, despite having antioxidant properties, lack the necessary structural traits for efficient nitric oxide and hydroxyl radical scavenging. Our findings align with a prior study indicating that flavonoids primarily contribute to NORSA and showed an enhanced correlation between total flavonoids and NO scavenging activity, with an R2 value of 0.73.36,37 Additionally, the presence of higher levels of phenolics and flavonoids was linked to increased iron chelating activity, indicating their ability to chelate free iron ions that can otherwise initiate oxidative reactions.
The thorough assessment of both free and bound forms of phenols and flavonoids, in conjunction with their associated antioxidant activity, across different drying techniques like FD, OD, and SD, holds paramount importance for several purposes. Firstly, it plays a pivotal role in the advancement of functional foods by elucidating the influence of diverse drying methods on the content and efficacy of these compounds, which are renowned for their health-promoting attributes, notably antioxidant properties. Secondly, it is instrumental in the realm of nutraceutical production, where phenols and flavonoids frequently serve as pivotal components, by facilitating the development of consistent, high-quality products. Additionally, within pharmaceutical applications, where these compounds are utilized for their potential therapeutic benefits, comprehending the impact of drying methods on their stability and bioavailability is vital for effective drug formulation. Lastly, in a broader industrial context encompassing sectors such as cosmetics and agriculture, optimizing drying techniques for preserving phenols and flavonoids can lead to creating superior-quality products. Moreover, from an industrial perspective, the choice of drying method must also consider cost-effectiveness, scalability, and energy consumption, as OD is generally more energy-efficient and scalable, while FD, although better at preserving free phenolics, is more costly and energy-intensive.
5 Conclusion
Regardless of the drying method, freshwater and commercial strains exhibited significantly lower levels of total phenolic compounds compared to their euryhaline counterparts. Among the euryhaline strains, Anabaena demonstrated the highest phenol and flavonoid content (p < 0.05) under both FD and OD, followed by Chlorella and Spirulina. OD effectively preserves bound phenolics, resulting in heightened DPPH and superoxide scavenging activities, with strong correlations noted. Conversely, FD retains free phenols and flavonoids, displaying robust positive correlations with total reducing powers. Strain B7 stands out with significantly higher phenol and flavonoid content, particularly under FD and OD. Additionally, assessing free and bound phenols and flavonoids with associated antioxidant activity during various drying methods is crucial for enhancing euryhaline microalgal biomass as functional foods, ensuring quality in nutraceuticals, stabilizing pharmaceutical formulations, and improving product quality in industries like cosmetics and agriculture. These insights provide valuable implications for leveraging the natural advantages of euryhaline microalgae in diverse industrial applications.Author contributions
Manpreet Kaur: conceptualization, writing – original draft, methodology, investigation, data curation, and formal analysis; Surekha Bhatia: conceptualization, investigation, writing – review & editing, and supervision; Kiran Tewari: methodology, data curation, writing – review & editing, and validation; Urmila Gupta Phutela: writing – review & editing, and supervision.Conflicts of interest
The authors declare no competing, financial, or personal interests.Abbreviations
| BF | Bound flavonoids |
| BP | Bound phenols |
| DDPHSA | DPPH free radical scavenging activity |
| FD | Freeze-drying |
| FF | Free flavonoids |
| FICA | Ferrous ion chelating activity |
| FP | Free phenols |
| FRAP | Ferric reducing antioxidant power |
| GAE | Gallic acid equivalents |
| HPSA | Hydrogen peroxide scavenging activity |
| HRSA | Hydroxyl radical scavenging activity |
| NORSA | Nitric oxide radical scavenging activity |
| OD | Oven drying |
| SARSA | Superoxide anion radical scavenging activity |
| SD | Shade drying |
| TF | Total flavonoids |
| TP | Total phenols |
| TP + TF | Total phenols + total flavonoids |
| TRP | Total reducing power |
Data availability
All the data used in this study has been presented.Acknowledgements
This study was jointly funded by the Science and Engineering Research Board, Federation of Indian Chambers of Commerce and Industry and Chemical Resources (CHERESO) Panchkula under the Prime Minister Research Fellowship Program. The author(s) express their heartfelt gratitude to Chemical Resources (CHERESO) in Panchkula, Haryana, India, as well as the Science & Engineering Research Board (SERB) and the Federation of Indian Chambers of Commerce and Industry (FICCI) for their invaluable support through the esteemed Prime Minister Research Fellowship Program, which made this project possible.References
- E. J. Rifna, N. N. Misra and M. Dwivedi, Recent advances in extraction technologies for recovery of bioactive compounds derived from fruit and vegetable waste peels: A review, Crit. Rev. Food Sci. Nutr., 2023, 63, 719–752, DOI:10.1080/10408398.2021.1952923.
- A. Dubey, A. Khantal, P. Tomar and A. Padiyar, Review on plants having antioxidant properties, Int. J. Pharmacogn. Life Sci., 2022, 3, 20–26, DOI:10.33545/27072827.2022.v3.i2a.56.
- S. Kapoor, M. Singh, A. Srivastava, M. Chavali, K. Chandrasekhar and P. Verma, Extraction and characterization of microalgae-derived phenolics for pharmaceutical applications: A systematic review, J. Basic Microbiol., 2022, 62, 1044–1063, DOI:10.1002/jobm.202100458.
- J. Zhou, M. Wang, C. Bäuerl, E. Cortés-Macías, J. Calvo-Lerma, M. C. Collado and F. J. Barba, The impact of liquid-pressurized extracts of Spirulina, Chlorella and Phaedactylum tricornutum on in vitro antioxidant, antiinflammatory and bacterial growth effects and gut microbiota modulation, Food Chem., 2023, 401, 134083, DOI:10.1016/j.foodchem.2022.134083.
- M. Kaur, S. Bhatia, U. Gupta, E. Decker and Y. Tak, et al., Microalgal bioactive metabolites as promising implements in nutraceuticals and pharmaceuticals: inspiring therapy for health benefits, Phytochem. Rev., 2023, 22, 903–933, DOI:10.1007/s11101-022-09848-7.
- F. de Farias Neves, M. Demarco and G. Tribuzi, Drying and quality of microalgal powders for human alimentation, Microalgae-from Physiology to Application, 2019, pp. 71–90 Search PubMed.
- F. de Jesús Bonilla-Ahumada, S. Khandual and E. del Carmen Lugo-Cervantes, Microencapsulation of algal biomass (Tetraselmis chuii) by spray-drying using different encapsulation materials for better preservation of beta-carotene and antioxidant compounds, Algal Res., 2018, 36, 229–238, DOI:10.1016/j.algal.2018.10.006.
- K. H. Min, D. H. Kim, M. R. Ki and S. P. Pack, Recent progress in flocculation, dewatering, and drying technologies for microalgae utilization: Scalable and low-cost harvesting process development, Bioresour. Technol., 2022, 344, 126404, DOI:10.1016/j.biortech.2021.126404.
- S. K. Saha, S. Dey and R. Chakraborty, Effect of microwave power on drying kinetics, structure, color, and antioxidant activities of corncob, J. Food Process Eng., 42, e13021, DOI:10.1080/10408398.2021.1952923.
- D. Liu, P. Lopez-Sanchez, M. Martinez-Sanz, E. P. Gilbert and M. J. Gidley, Adsorption isotherm studies on the interaction between polyphenols and apple cell walls: Effects of variety, heating and drying, Food Chem., 2019, 282, 58–66, DOI:10.1016/j.foodchem.2018.12.098.
- M. Rudrapal, G. Rakshit, R. P. Singh, S. Garse, J. Khan and S. Chakraborty, Dietary polyphenols: review on chemistry/sources, bioavailability/metabolism, antioxidant effects, and their role in disease management, Antioxidants, 2024, 13, 429, DOI:10.3390/antiox13040429.
- M. Kaur, S. Bhatia, B. S. Kalsi and U. G. Phutela, Unveiling the biomass conversion potential: study on drying methods' influence on polyphenols and linked antioxidant activities in euryhaline microalgal biomass with AI-predicted drying kinetics, Biomass Convers. Biorefin., 2024, 1–16, DOI:10.1007/s13399-024-05533-1.
- R. A. Dar, Bioprospecting of microalgal isolates from the waterlogged area of Punjab for biogas production, PhD thesis, Punjab Agricultural University, Ludhiana, 2017.
- M. Kaur, S. Bhatia, M. K. Sangha and U. G. Phutela, Crosstalk of AsA/DHA and GSH/GSSG ratios’ role in growth-phase dependent antioxidative defense in euryhaline and freshwater microalgae: explored for the first time, Physiol. Mol. Biol. Plants, 2023, 1–18, DOI:10.1007/s12298-023-01349-x.
- D. Granato, J. S. Santos, L. G. Maciel and D. S. Nunes, Chemical perspective and criticism on selected analytical methods used to estimate the total content of phenolic compounds in food matrices, TrAC, Trends Anal. Chem., 2016, 80, 266–279, DOI:10.1016/J.TRAC.2016.03.010.
- A. M. Shraim, T. A. Ahmed, M. M. Rahman and Y. M. Hijji, Determination of total flavonoid content by aluminum chloride assay: A critical evaluation, LWT--Food Sci. Technol., 2021, 150, 111932, DOI:10.1016/J.LWT.2021.111932.
- H. Li, K. Cheng, C. Wong, K. Fan, F. Chen and Y. Jiang, Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae, Food Chem., 2007, 102, 771–776, DOI:10.1016/j.foodchem.2006.06.022.
- M. S. Blois, Antioxidant Determinations by the use of a stable free radical, Nature, 1958, 181, 1199–1200, DOI:10.1038/1811199a0.
- S. Marklund and G. Marklund, Involvement of the Superoxide Anion Radical in the Autoxidation of Pyrogallol and a Convenient Assay for Superoxide Dismutase, Eur. J. Biochem., 1974, 47, 469–474, DOI:10.1111/J.1432-1033.1974.TB03714.X.
- L. Marcocci, J. J. Maguire, M. T. Droy-Lefaix and L. Packer, The nitric oxide-scavenging properties of Ginkgo biloba extract, Biochem. Biophys. Res. Commun., 1994, 201, 748–755, DOI:10.1006/BBRC.1994.1764.
- G. Minotti and S. D. Aust, An investigation into thee mechanism of citrate-FE2+-dependent lipid peroxidation, Free Radical Biol. Med., 1987, 3, 379–387, DOI:10.1016/0891-5849(87)90016-5.
- D. Sreeramulu, C. Vijaya, K. Reddy and M. Raghunath, Antioxidant activity of commonly consumed cereals, millets, pulses and legumes in India, Indian J. Biochem. Biophys., 2009, 46, 112–115 CAS.
- I. F. F. Benzie and M. Devaki, The ferric reducing/antioxidant power (FRAP) assay for non-enzymatic antioxidant capacity: concepts, procedures, limitations and applications, Meas. Antioxid. Act. Capacity, 2017, 77–106, DOI:10.1002/9781119135388.CH5.
- L. S. Borquaye, M. S. Doetse, S. O. Baah and J. A. Mensah, Anti-inflammatory and anti-oxidant activities of ethanolic extracts of Tamarindus indica L. (Fabaceae), Cogent Chem., 2020, 6, 1743403, DOI:10.1080/23312009.2020.1743403.
- M. C. Kolla, A. Laya, J. P. Bayang and B. B. Koubala, Effect of different drying methods and storage conditions on physical, nutritional, bioactive compounds and antioxidant properties of doum (Hyphaene thebaica) fruits, Heliyon, 2021, 7, e06678, DOI:10.1016/j.heliyon.2021.e06678.
- J. Branisa, K. Jomova, M. Porubska, V. Kollar, M. Simunkova and M. Valko, Effect of drying methods on the content of natural pigments and antioxidant capacity in extracts from medicinal plants: a spectroscopic study, Chem. Pap., 2017, 71, 1993–2002, DOI:10.1007/s11696-017-0193-9.
- Q. Meng, H. Fan, Y. Li and L. Zhang, Effect of drying methods on physico-chemical properties and antioxidant activity of Dendrobium officinale, J. Food Meas. Charact., 2018, 12, 1–10 CrossRef.
- H. Xu, M. Wu, Y. Wang, W. Wei, D. Sun and D. Li, et al., Effect of combined infrared and hot air-drying strategies on the quality of chrysanthemum (Chrysanthemum morifolium ramat.) cakes: Drying behavior, aroma profiles and phenolic compounds, Foods, 2022, 11, 2240, DOI:10.3390/foods11152240.
- C. Lai, Y. Liang, L. Zhang, J. Huang, K. Kaliaperumal, Y. Jiang and J. Zhang, Variations of bioactive phytochemicals and antioxidant capacity of navel orange peel in response to different drying methods, Antioxidants, 2022, 11, 1543, DOI:10.3390/antiox11081543.
- Q. Ma, J. Bi, J. Yi, X. Wu, X. Li and Y. Zhao, Stability of phenolic compounds and drying characteristics of apple peel as affected by three drying treatments, Food Sci. Hum. Wellness, 2021, 10, 174–182, DOI:10.1016/j.fshw.2021.02.006.
- Y. Zhang, B. Bai, Y. Yan, J. Liang and X. Guan, Bound polyphenols from red quinoa prevailed over free polyphenols in reducing postprandial blood glucose rises by inhibiting α-glucosidase activity and starch digestion, Nutrients, 2022, 14, 728, DOI:10.3390/nu14040728.
- P. Mohapatra, A. Ray, S. Jena, S. Nayak and S. Mohanty, Influence of various drying methods on physicochemical characteristics, antioxidant activity and bioactive compounds in Centella asiatica L. leaves: a comparative study, BioTechnologia, 2022, 103, 235–247, DOI:10.5114/2022.118666.
- D. Huang, C. Li, Q. Chen, X. Xie, X. Fu, C. Chen, Q. Huang, Z. Huang and H. Dong, Identification of polyphenols from Rosa roxburghii Tratt pomace and evaluation of in vitro and in vivo antioxidant activity, Food Chem., 2022, 377, 131922, DOI:10.1016/J.FOODCHEM.2021.131922.
- Y. Lin, D. Tang, X. Liu, J. Cheng, X. Wang and D. Guo, et al., Phenolic profile and antioxidant activity of longan pulp of different cultivars from South China, LWT--Food Sci. Technol., 2022, 165, 113698, DOI:10.1016/j.lwt.2022.113698.
- S. M. Wani, F. A. Masoodi, M. Ahmad and S. A. Mir, Processing and storage of apricots: effect on physicochemical and antioxidant properties, J. Food Sci. Technol., 2018, 55, 4505–4514, DOI:10.1007/s13197-018-3381-x.
- M. Ebrahimzadeh, S. F. Nabavi, S. M. Nabavi and F. Pourmorad, Nitric oxide radical scavenging potential of some Elburz medicinal plants, Afr. J. Biotechnol., 2010, 9, 5212–5217 CAS.
- J. Treml and K. Šmejkal, Flavonoids as potent scavengers of hydroxyl radicals, Compr. Rev. Food Sci. Food Saf., 2016, 15, 720–738 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |