Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Optimization of maltodextrin–gum arabic–whey protein systems for freeze-drying microencapsulation of young barley leaf extract
Elham
Azarpazhooh
 *a,
Yeganeh
Sabeghi
*a,
Yeganeh
Sabeghi
 bc,
Masoud Najaf
Najafi
*a,
Shahin
Zomorodi
d,
Soodabeh
Einafshar
a,
Danial
Gandomzadeh
e and
Xin
Rui
bc,
Masoud Najaf
Najafi
*a,
Shahin
Zomorodi
d,
Soodabeh
Einafshar
a,
Danial
Gandomzadeh
e and
Xin
Rui
 f
f
aDepartment of Agricultural Engineering Research Department, Khorasan Razavi Agricultural and Natural Resources Research and Education Center, AREEO, Mashhad, Iran. E-mail: azarpazhooh@gmail.com; mnajafi.mhd@gmail.com
bDepartment of Food Science and Technology, Faculty of Agriculture, Ferdowsi University of Mashhad, Iran
cDepartment of Food Science and Technology, Faculty of Agriculture, Technical and Vocational University (TVU), Iran
dAgricultural Engineering Research Department, West Azerbaijan Agricultural and Natural Resources Research and Education Center, AREEO, Urmia, Iran
eDepartment of Mechanical Engineering, Technical and Vocational University (TVU), Iran
fCollege of Food Science and Technology, Nanjing Agricultural University, 14 1 Weigang Road, Nanjing 211306, China
First published on 28th January 2026
Abstract
Young barley leaves are rich in bioactive compounds but highly prone to oxidation, necessitating stabilization for functional food applications. This study encapsulated barley leaf extract using maltodextrin (MD), whey protein isolate (WPI), and gum arabic (GA), and optimized wall-material ratios through freeze-drying and response surface methodology (RSM). Model fitting confirmed strong statistical performance, with significant quadratic models (p < 0.05), non-significant lack-of-fit, R2 values up to 0.99, and adequate precision above 40, ensuring reliable prediction across the design space. Experimentally, MD-rich formulations achieved the highest microencapsulation yield (∼89%), while encapsulation efficiency (83.67–89.98%) maximized in a ternary MD–WPI–GA blend (67.66![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16.67
16.67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16.67). Moisture content (10.22–11.29%), water activity (0.34–0.40), and particle size (34.50–37.59 µm) indicated good storage stability and structural integrity. Solubility reached 75.29% in MD–GA systems, and the highest glass transition temperature occurred in the ternary blend, reflecting enhanced thermal stability. Bioactive preservation was substantial, with total phenolic content (∼70 mg GAE per g) and antioxidant activity (∼80%) maintained effectively. Overall, MD contributed to high yield, GA to emulsion stability, and WPI to improved film-forming and antioxidant protection. The optimized microcapsules demonstrated strong stability and functional potential for use in nutraceuticals, functional foods, and instant beverage formulations.
16.67). Moisture content (10.22–11.29%), water activity (0.34–0.40), and particle size (34.50–37.59 µm) indicated good storage stability and structural integrity. Solubility reached 75.29% in MD–GA systems, and the highest glass transition temperature occurred in the ternary blend, reflecting enhanced thermal stability. Bioactive preservation was substantial, with total phenolic content (∼70 mg GAE per g) and antioxidant activity (∼80%) maintained effectively. Overall, MD contributed to high yield, GA to emulsion stability, and WPI to improved film-forming and antioxidant protection. The optimized microcapsules demonstrated strong stability and functional potential for use in nutraceuticals, functional foods, and instant beverage formulations.
Sustainability spotlightThis study supports sustainability in food systems by enhancing the stability of young barley leaf extract—an underutilized but nutrient-dense crop—through optimized freeze-drying microencapsulation. By preserving antioxidant and bioactive compounds without relying on synthetic additives or harsh thermal processes, the work enables the development of natural, shelf-stable ingredients for functional foods and nutraceuticals. The approach leverages readily available, food-grade wall materials (maltodextrin, gum arabic, and whey protein isolate), offering a practical and scalable strategy to reduce nutrient loss, extend product shelf life, and minimize food waste. In doing so, it contributes to sustainable innovation in food preservation and the promotion of health-supportive, plant-based products. |
1. Introduction
Young barley leaves (Hordeum vulgare) have gained significant attention for their rich nutritional and bioactive composition, including essential minerals, vitamins (B-complex, C, and E), chlorophyll, β-carotene, and antioxidant enzymes such as superoxide dismutase and catalase.1 These components contribute to their well-documented physiological benefits, including antioxidant protection, immune support, lipid and glucose levels regulation, and modulation of gut microbiota.2,3 Consequently, barley leaf-based powders, beverages, and nutraceutical supplements have become increasingly popular in global health-oriented food markets.4The beneficial effects of young barley leaves are primarily attributed to phenolic acids, flavonoids such as saponarin, polysaccharides, β-glucans, and phytosterols.2,5
Despite these advantages, the direct use of barley leaf extract faces significant limitations due to the instability of its bioactive compounds, which are highly sensitive to environmental factors such as heat, light, oxygen, and pH.3 To overcome these challenges, microencapsulation has emerged as a powerful technique for stabilizing and protecting bioactive compounds during processing and storage.6 This approach involves coating bioactive compounds with polymeric or non-polymeric materials, thereby enhancing their stability, improving sensory quality by masking undesirable flavors, and even increasing food safety through inhibition of microbial growth.6 The effectiveness of encapsulation depends largely on the choice of wall material, which must provide protection, ensure controlled release, and maintain compatibility with the core compound.7
Among the various wall materials, carbohydrates (e.g., maltodextrin (MD), modified starch, cyclodextrins), gums (e.g., gum arabic, agar, carrageenan), lipids (e.g., waxes, beeswax, diacylglycerols), and proteins (e.g., casein, gelatin, whey protein isolate (WPI)) have been extensively studied.8 Maltodextrin and gum arabic (GA) are particularly favored for their solubility, biocompatibility, and film-forming properties,6,9 while WPI has attracted attention for its nutritional value, emulsifying ability, and capacity to stabilize hydrophobic compounds.7,10 Combining these materials often enhances encapsulation efficiency and product stability, addressing the shortcomings of using each material individually.11
Among encapsulation methods, freeze-drying (lyophilization) is especially suitable for heat-sensitive bioactive compounds, as it avoids thermal degradation and preserves functional properties.10,11 For instance, da Silva Júnior et al.12 demonstrated that ciriguela peel extracts had higher quercetin and kaempferol retention in freeze-dried capsules compared with spray-dried ones. Several studies have demonstrated the successful microencapsulation of fruit and herbal extracts using maltodextrin, gum arabic, and protein-based carriers. However, most investigations have focused on single or binary wall systems, with limited systematic exploration of ternary formulations. For instance, Ledari et al.10 encapsulated chlorophyll using maltodextrin and WPI with both freeze-drying (FD) and spray-drying (SD), reporting that FD microcapsules exhibited smaller particle sizes and higher ζ-potential compared to SD. As well, in lipid-based nano-encapsulation, Choudhary et al.13 reported that amorphous freeze-dried nanostructures of linseed-oil carriers, demonstrating high encapsulation efficiencies of 84.32 ± 1.08% for LA and 79.63 ± 1.41% for β-carotene.
Todorović et al.14 encapsulated bilberry anthocyanins with maltodextrin, gum arabic, and their mixture, demonstrating that maltodextrin provided the best protection for anthocyanins during storage. Sirichokworrakit et al.15 compared different encapsulants for Wolffia globosa extract and found that maltodextrin combined with whey protein concentrate achieved the highest encapsulation efficiency and stability. Similarly, Ezhilarasi et al.16 microencapsulated cowa fruit extract using WPI, MD, and their mixture, achieving high recovery of hydroxycitric acid (>85% free and >90% total) in all formulations. Despite these advances, research on ternary combinations, particularly MD–GA–WPI, remain scarce.
To date, no optimized ternary encapsulation system has been specifically developed for young barley leaf extract, despite its high susceptibility to oxidation due to abundant enzymatic antioxidants and chlorophyll. Existing studies have mainly focused on fruit-based or by-product extracts, leaving a clear gap regarding green cereal leaf matrices with complex phytochemical profiles. Therefore, this study aimed to optimize a MD–GA–WPI system for freeze-drying microencapsulation of young barley leaf extract using response surface methodology (RSM). The innovation lied in evaluating the synergistic effects of three biopolymers on encapsulation efficiency, powder characteristics, and retention of antioxidant compounds, enabling the development of stable, bioactive-rich barley leaf powders for functional food and nutraceutical applications. Beyond technological aspects, this work also highlighted sustainability benefits, as effective encapsulation can improve resource efficiency, reduce nutritional losses, and support low-dosage delivery formats for ingredients derived from dedicated cultivation systems such as young barley leave.
2. Material and method
2.1. Materials
Young barley leaves were sourced from the Green Sprout Technology Development Company, located in the Agricultural and Natural Resources Technology and Innovation Village, Khorasan Razavi, Iran (36.30°N, 59.62°E). The crop was cultivated under commercial field conditions during the spring growing season of 2023, with irrigation following standard agronomic practices for young barley production. Leaves were harvested 18–20 days after emergence, corresponding to the early vegetative stage characterized by high chlorophyll and phenolic accumulation. Harvesting was conducted in the early morning to minimize thermal and oxidative stress and to preserve metabolite integrity. Upon arrival, the fresh leaves were washed thoroughly with distilled water to remove impurities, cut into small pieces, and dried using a hot-air dryer (Enex-Co-600; Enex, Koyang, Korea) at 70 °C for 24 h. The dried leaves were then pulverized using a blender (KA-2610; Jworld Tech, Ansan, Korea) for 30 s and passed through a 35-mesh sieve to obtain a uniform powder. The powdered samples were packed in polyethylene bags and stored at −20 °C until further use.Maltodextrin (dextrose equivalent 20), gum arabic, and whey protein isolate were purchased from Sigma-Aldrich Co. (St. Louis, MO, USA). As well, all other chemicals and reagents used in this study were of analytical grade and obtained from Sigma-Aldrich Co. (St. Louis, MO, USA) and Merck Co. (Darmstadt, Germany).
2.2. Methods
Young barley leaf powder (10 g) was extracted using 100 mL of 70% (v/v) ethanol at a solid-to-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v). The mixture was kept in the dark at 25 °C for 48 h under occasional stirring to facilitate the release and solubilization of bioactive compounds. After filtration through Whatman No. 1 paper, the filtrate was concentrated under reduced pressure at 40 °C (Heidolph, Germany) and further dried in a vacuum-oven drying at 0.1 kPa (Memmert VO 400, Frankfurt, Germany) for 48 h. The dried extract was weighed to determine extraction yield and stored at −18 °C until analysis.17
10 (w/v). The mixture was kept in the dark at 25 °C for 48 h under occasional stirring to facilitate the release and solubilization of bioactive compounds. After filtration through Whatman No. 1 paper, the filtrate was concentrated under reduced pressure at 40 °C (Heidolph, Germany) and further dried in a vacuum-oven drying at 0.1 kPa (Memmert VO 400, Frankfurt, Germany) for 48 h. The dried extract was weighed to determine extraction yield and stored at −18 °C until analysis.17
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (w/w). The mixture was homogenized using an Ultra-Turrax homogenizer (IKA T50, Staufen, Germany) at 12
5 (w/w). The mixture was homogenized using an Ultra-Turrax homogenizer (IKA T50, Staufen, Germany) at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min, followed by sonication (Eurosonic 4D, Italy; 300 W, 50 kHz) for 1 min in an ice bath to achieve a uniform and stable solutions. The prepared solutions were then pre-frozen at −70 °C for 19 h to minimize foam formation during drying and to avoid heating of the solution due to the sonication procedure. Freeze-drying was carried out in a laboratory freeze dryer (Operon FDB-550, Seoul, Korea) at −55 °C and 0.15 mmHg for 20 h. Approximately 200 mL of each solution was freeze-dried in stainless steel. After drying, the resulting powders were gently ground with a mortar and pestle, sieved through a 500 µm mesh (ISO 3310-ASTM E11 standard), and packed in airtight polyethylene containers under a nitrogen atmosphere to avoid oxidation. The powders were stored either at −40 °C or in the dark at room temperature (20–22 °C) until further physicochemical analyses.18
000 rpm for 5 min, followed by sonication (Eurosonic 4D, Italy; 300 W, 50 kHz) for 1 min in an ice bath to achieve a uniform and stable solutions. The prepared solutions were then pre-frozen at −70 °C for 19 h to minimize foam formation during drying and to avoid heating of the solution due to the sonication procedure. Freeze-drying was carried out in a laboratory freeze dryer (Operon FDB-550, Seoul, Korea) at −55 °C and 0.15 mmHg for 20 h. Approximately 200 mL of each solution was freeze-dried in stainless steel. After drying, the resulting powders were gently ground with a mortar and pestle, sieved through a 500 µm mesh (ISO 3310-ASTM E11 standard), and packed in airtight polyethylene containers under a nitrogen atmosphere to avoid oxidation. The powders were stored either at −40 °C or in the dark at room temperature (20–22 °C) until further physicochemical analyses.18
| Run | Wall composition (%) | ||
|---|---|---|---|
| Maltodextrin (20 DE) | Whey protein | Arabic gum | |
| 1 | 16.67 | 16.67 | 66.67 |
| 2 | 0 | 0 | 100 |
| 3 | 0 | 100 | 0 |
| 4 | 0 | 100 | 0 |
| 5 | 50 | 50 | 0 |
| 6 | 0 | 50 | 50 |
| 7 | 0 | 0 | 100 |
| 8 | 50 | 0 | 50 |
| 9 | 100 | 0 | 0 |
| 10 | 66.67 | 16.67 | 16.67 |
| 11 | 100 | 0 | 0 |
| 12 | 50 | 50 | 50 |
| 13 | 33.33 | 33.33 | 33.33 |
| 14 | 16.67 | 66.67 | 16.67 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) was added. The suspension was vortexed for 1 min to extract non-encapsulated young barley leaf extract, followed by centrifugation at 3000×g for 2 min at room temperature (Hermle Z383 K, HERMLE Labortechnik, Wehingen, Germany). The supernatant was collected, and young barley leaf extract content was analyzed spectrophotometrically. As well, total young barley leaf extract content was quantified by dissolving an equivalent amount of powder in the same solvent system, followed by extended vortexing and sonication (30 min) to ensure complete rupture of the microcapsules. After centrifugation, the young barley leaf extract concentration in the supernatant was measured under the same analytical conditions. Encapsulation efficiency (%) was then calculated using eqn (1).
1, v/v) was added. The suspension was vortexed for 1 min to extract non-encapsulated young barley leaf extract, followed by centrifugation at 3000×g for 2 min at room temperature (Hermle Z383 K, HERMLE Labortechnik, Wehingen, Germany). The supernatant was collected, and young barley leaf extract content was analyzed spectrophotometrically. As well, total young barley leaf extract content was quantified by dissolving an equivalent amount of powder in the same solvent system, followed by extended vortexing and sonication (30 min) to ensure complete rupture of the microcapsules. After centrifugation, the young barley leaf extract concentration in the supernatant was measured under the same analytical conditions. Encapsulation efficiency (%) was then calculated using eqn (1).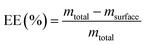 | (1) |
Microencapsulation yield was determined gravimetrically and expressed as the percentage ratio between the weight of the freeze-dried microencapsulated powder (Wfinal) and the theoretical dry weight of the wall materials used in the formulation (Winitial), according to eqn (2).
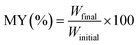 | (2) |
The moisture content of the freeze-dried powders was determined by gravimetric analysis. Approximately 2 g of each powder was weighed and dried in a hot air oven (Memmert, UFB 500, Schwabach, Germany) at 105 ± 1 °C until a constant weight was achieved. Results were expressed as a percentage of weight loss on a dry basis.
Water activity (aw) was measured at 25 °C using a dew-point-based water activity instrument (Pre AquaLab, USA).
Two grams of powder were carefully transferred into a pre-weighed 10-mL graduated cylinder without compacting. The cylinder was tapped three times on the bench to settle the powder, and the final volume was recorded. Bulk density was calculated as the ratio of the sample mass (g) to the occupied volume (cm3) and expressed in g cm−3.
Approximately 1 g of powder was dispersed in 100 mL of distilled water and stirred using a magnetic stirrer (ARE, Velp, Italy) at 400 rpm for 5 min at 25 °C. The suspension was then centrifuged at 3000 rpm for 5 min at 25 °C using a laboratory centrifuge (Hermle Z383 K, HERMLE Labortechnik, Wehingen, Germany).
An aliquot of 20–25 mL of the supernatant was transferred to a pre-weighed glass Petri dish and dried either at 70 °C overnight or at 105 °C for 5 h until constant weight. The solubility index (%) was calculated as the ratio of the dry weight of solids recovered after drying to the initial weight of powder used, according to eqn (3).
 | (3) |
Because freeze-dried microcapsules exhibit irregular, amorphous, and non-spherical shapes, particle size was not determined as a geometric spherical diameter. Instead, particle dimensions were quantified directly from SEM micrographs using ImageJ software. For each formulation, at least 100 particles were randomly selected across multiple fields, and the longest Feret diameter (maximum caliper distance) was used as a standardized descriptor of particle size.23
2.2.8.1. Total phenolic content (TPC). The total phenolic content of the microencapsulated powders was determined using the Folin–Ciocalteu colorimetric method, with slight modifications based on Sharayei et al.24 Briefly, 10 mg of powder was dissolved in methanol and appropriately diluted with distilled water. An aliquot of the extract was mixed with 2.5 mL of Folin–Ciocalteu reagent (previously diluted 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 with distilled water) and allowed to react for 3 min in the dark. Subsequently, 5 mL of sodium carbonate solution (7.5%, w/v) was added, and the mixture was made up to 50 mL with distilled water. The reaction mixture was incubated in the dark at room temperature for 60 min to allow color development (in some cases up to 24 h for complete stabilization). Absorbance was measured at 765 nm using a UV-Vis spectrophotometer (Shimadzu Co., Kyoto, Japan). A calibration curve was prepared using gallic acid standard solutions (Sigma-Aldrich, Darmstadt, Germany), and results were expressed as milligrams of gallic acid equivalents per gram of dry sample (mg GAE per g DW).
10 with distilled water) and allowed to react for 3 min in the dark. Subsequently, 5 mL of sodium carbonate solution (7.5%, w/v) was added, and the mixture was made up to 50 mL with distilled water. The reaction mixture was incubated in the dark at room temperature for 60 min to allow color development (in some cases up to 24 h for complete stabilization). Absorbance was measured at 765 nm using a UV-Vis spectrophotometer (Shimadzu Co., Kyoto, Japan). A calibration curve was prepared using gallic acid standard solutions (Sigma-Aldrich, Darmstadt, Germany), and results were expressed as milligrams of gallic acid equivalents per gram of dry sample (mg GAE per g DW).
2.2.8.2. DPPH radical scavenging activity. The antioxidant capacity of the microcapsules was assessed using the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay. A freshly prepared 0.006% methanolic DPPH solution was used as the radical source. One milliliter of sample solution at different concentrations was mixed with 1 mL of the DPPH solution, vortexed for uniform mixing, and incubated in the dark at room temperature for 1 h. Absorbance was measured at 512 nm against a methanol blank using a UV-Vis spectrophotometer (Shimadzu Co., Kyoto, Japan). The radical scavenging activity (RSA) was calculated as inhibition percentage using eqn (4).
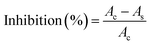 | (4) |
 | (5) |
3. Results and discussion
3.1. Model fitting
Multiple regression models, including linear and quadratic forms, were initially tested for all physicochemical and functional responses to ensure comprehensive model evaluation. The most appropriate model for each response was selected based on statistical significance, lack-of-fit analysis, goodness-of-fit parameters, and predictive ability. As shown in Table 2, the regression models applied to the microencapsulation data demonstrated strong statistical validity. Most responses were best described by quadratic models, with highly significant p-values (p < 0.05), non-significant lack of fit, and high coefficients of determination (R2 up to 0.99). The close agreement between R2, adjusted R2, and predicted R2 (differences <0.2) confirmed that the models accurately captured the variability of each response without overfitting. Adequate precision values—many exceeding 40—indicated strong signal quality and reliable prediction capability within the experimental design space. Additionally, the low CV values reflected excellent repeatability of the measurements. Collectively, these metrics demonstrated that the selected models were robust, precise, and suitable for predicting the physicochemical and functional properties of barley-leaf microcapsules across the tested formulation range.| Features | Model | p-Value | Lack of fit | R 2 | Adj R2 | Pred R2 | Adeq precision | Mean | SD | CV (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Encapsulation efficiency | Quadratic | 0.0072 | 0.19 | 0.74 | 0.65 | 0.49 | 6.57 | 86.42 | 1.14 | 1.32 |
| Microencapsulation yield | Quadratic | <0.0001 | 0.99 | 0.99 | 0.99 | 0.97 | 48.01 | 86.98 | 0.11 | 0.13 |
| Moisture content | Quadratic | 0.0003 | 0.99 | 0.91 | 0.86 | 0.70 | 13.79 | 10.66 | 0.10 | 0.97 |
| Water activity | Quadratic | 0.02 | 0.94 | 0.90 | 0.85 | 0.68 | 12.78 | 0.35 | 0.0076 | 2.13 |
| Bulk density | Quadratic | 0.0088 | 0.95 | 0.84 | 0.78 | 0.56 | 9.36 | 0.21 | 0.01 | 5.98 |
| Solubility index | Linear | <0.0001 | 0.57 | 0.90 | 0.88 | 0.82 | 17.82 | 72.53 | 1.00 | 1.38 |
| Glass transition temperature | Quadratic | 0.03 | 0.13 | 0.62 | 0.46 | 0.03 | 7.95 | 75.47 | 1.00 | 1.33 |
| Particle size distribution | Quadratic | 0.0028 | 0.98 | 0.91 | 0.85 | 0.69 | 11.91 | 35.57 | 0.37 | 1.06 |
| Total phenolic content | Quadratic | <0.0001 | 0.99 | 0.99 | 0.98 | 0.97 | 44.39 | 62.18 | 0.41 | 0.67 |
| DPPH radical scavenging activity | Quadratic | <0.0001 | 0.99 | 0.98 | 0.98 | 0.96 | 42.13 | 76.80 | 0.2 | 0.54 |
3.2. Powder yield of microencapsulation
As shown in Table 3, the microencapsulation yield (MY) ranged from 85.62% to 89.45%. This variation was directly related to the nature and proportion of the encapsulating agents used. When MD alone (100%) was employed, the highest yield of approximately 89% was obtained. This superior performance can be attributed to maltodextrin's excellent solubility, low viscosity, and strong drying properties, which reduce stickiness in the freeze dryer chamber and thus improve powder recovery. In addition, maltodextrin increases the glass transition temperature of the feed and forms a protective layer around droplets, thereby preventing stickiness and enhancing yield. These results are consistent with previous studies by Sarabandi et al.27 and Goula and Adamopoulos,28 who reported similar improvements when maltodextrin was used in the microencapsulation of date juice and orange concentrate, respectively. For WPI alone (100%), yields of around 86.86% were achieved. WPI is known for its excellent emulsifying properties and strong film-forming ability, which reduce stickiness and hygroscopicity, thereby improving yield. The improvement is particularly pronounced at lower concentrations, where proteins migrate to the surface of droplets during drying and form a protein-rich film that prevents adhesion to chamber walls. Similar observations were reported by Wang et al.29 in soy sauce powder and Shi et al.30 in honey powder, where whey proteins increased powder yield by reducing stickiness. However, excessive WPI may reduce yield due to protein denaturation and heat sensitivity during drying. When GA alone (100%) was applied, the yield reached approximately 87.8%, which is higher than WPI but lower than maltodextrin. GA possesses natural emulsifying capacity and good film-forming properties, which contribute to higher recovery.31 However, its higher viscosity compared to maltodextrin can lead to stickier particles and larger droplets during atomization. Nonetheless, increasing the concentration of GA generally led to higher yields, consistent with the findings of Karrar et al.32 who reported improved microencapsulation efficiency when GA was used in higher proportions.| Run | MY (%) | ME (%) | Particle size (µm) | Glass transition temperature (°C) |
|---|---|---|---|---|
| 86.45 | 89.98 | |||
| 1 | 87.80 | 84.47 | 35.59 | 74.24 |
| 2 | 86.86 | 87.05 | 37.59 | 75.81 |
| 3 | 86.86 | 87.05 | 34.83 | 76.53 |
| 4 | 85.91 | 85.63 | 35.01 | 77.52 |
| 5 | 85.75 | 88.82 | 34.98 | 76.32 |
| 6 | 87.80 | 84.47 | 34.82 | 72.27 |
| 7 | 87.45 | 88.94 | 37.59 | 75.89 |
| 8 | 89.45 | 83.67 | 35.49 | 75.75 |
| 9 | 87.05 | 86.13 | 36.31 | 75.29 |
| 10 | 89.00 | 85.81 | 35.21 | 77.98 |
| 11 | 85.91 | 84.63 | 35.31 | 75.29 |
| 12 | 85.82 | 87.13 | 36.00 | 74.55 |
| 13 | 85.62 | 86.17 | 34.72 | 74.42 |
In contrast, mixtures of wall materials produced variable results. The binary blend of MD and WPI (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) resulted in a yield of 85.91%, lower than either component alone. This suggests that the expected synergistic effect did not occur, possibly due to an imbalance between carbohydrate drying behavior and protein thermal sensitivity. Likewise, WPI and GA (50
50) resulted in a yield of 85.91%, lower than either component alone. This suggests that the expected synergistic effect did not occur, possibly due to an imbalance between carbohydrate drying behavior and protein thermal sensitivity. Likewise, WPI and GA (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) yielded 85.75%, offering no clear advantage. The blend of MD and GA (50
50) yielded 85.75%, offering no clear advantage. The blend of MD and GA (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) reached 87.45%, which is comparable to GA alone but still lower than maltodextrin's performance, likely due to increased viscosity from the gum fraction. Ternary formulations, such as equal proportions of MD, WPI, and GA (33
50) reached 87.45%, which is comparable to GA alone but still lower than maltodextrin's performance, likely due to increased viscosity from the gum fraction. Ternary formulations, such as equal proportions of MD, WPI, and GA (33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33
33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33), gave yields around 85.82%, while other three-way blends ranged between 85.6% and 87.05%. Although diversification of wall materials improved emulsion stability, it did not maximize yield. This may be explained by higher viscosity leading to delayed atomization and stickier particles, as reported by Ozdemir et al.33
33), gave yields around 85.82%, while other three-way blends ranged between 85.6% and 87.05%. Although diversification of wall materials improved emulsion stability, it did not maximize yield. This may be explained by higher viscosity leading to delayed atomization and stickier particles, as reported by Ozdemir et al.33
3.3. Microencapsulation efficiency
Encapsulation efficiency (ME) is a critical parameter for improving any encapsulation procedure, regardless of the process or material involved.31 In this study, ME of young leaf extract varied between 83.67% and 89.98% (Table 3), confirming that both the type and ratio of wall materials substantially influenced retention. These variations reflect differences in the polymer matrices formed by each wall material, which display distinct film-forming capacities and retention properties.33 Furthermore, the physicochemical features of the wall and the core—such as emulsifying capacity, interfacial stability, and drying behavior—proved decisive in encapsulating sensitive bioactive compounds.18,34The highest ME (89.98%) was obtained when MD dominated the formulation (MD 67.66%, WPI 16.67%, GA 16.67%). Maltodextrin is widely recognized as a highly soluble, low-viscosity carbohydrate with low sugar content and a colorless solution, properties that facilitate drying, increase the glass transition temperature, and minimize stickiness, thereby forming a stable protective film.35 Our findings suggested that MD performs best when complemented with protein and gum components. When GA alone (100%) was used, ME dropped to 84.47%. GA, a highly branched heteropolysaccharide with covalently bound glycoproteins, is well known for its film-forming and emulsifying abilities across a wide pH range. Its amphiphilic structure allows interactions with both hydrophilic and hydrophobic molecules, which strengthens emulsion stability through steric and thickening effects.36 However, excessive viscosity at high concentrations can produce larger droplets and reduce recovery, explaining the lower efficiency when GA was used alone.
Similarly, MD alone (100%) yielded low efficiency (83.67–85.81%) because, although it enhances drying performance, the absence of emulsifying agents limits interfacial stability. In contrast, WPI alone (100%) achieved a higher ME of 87.05%, outperforming either MD or GA alone. This was attributed to WPI's strong emulsifying activity and its ability to migrate to the droplet surface during atomization, forming a viscoelastic protein-rich film that enhances structural integrity.36 However, the heat sensitivity of proteins may limit their protective role when used as the sole encapsulant.
Mixed formulations revealed clear synergistic effects. A 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 blend of WPI and GA achieved 88.82%, while a 50
50 blend of WPI and GA achieved 88.82%, while a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 blend of MD and GA reached 88.94%, both significantly higher than single-component systems. Consistent with the earlier findings on MY, these results confirmed that GA contributes emulsification and coating capacity, while MD strengthens the protective matrix by reducing stickiness and supporting film formation.37 Similar synergistic effects have been reported: Habib et al.38 observed the highest ME (90.31%) with GA/WPI for astaxanthin, and Rashid et al.39 achieved 85–96% ME for pomegranate peel extract using MD, WPI, and their blends, demonstrating the benefits of combining polysaccharides and proteins. Moreover, electrostatic interactions between WPI and GA are known to reinforce interfacial films, promoting higher encapsulation yields.40,41
50 blend of MD and GA reached 88.94%, both significantly higher than single-component systems. Consistent with the earlier findings on MY, these results confirmed that GA contributes emulsification and coating capacity, while MD strengthens the protective matrix by reducing stickiness and supporting film formation.37 Similar synergistic effects have been reported: Habib et al.38 observed the highest ME (90.31%) with GA/WPI for astaxanthin, and Rashid et al.39 achieved 85–96% ME for pomegranate peel extract using MD, WPI, and their blends, demonstrating the benefits of combining polysaccharides and proteins. Moreover, electrostatic interactions between WPI and GA are known to reinforce interfacial films, promoting higher encapsulation yields.40,41
In line with the previous discussion of MY, the MD + WPI combination (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) yielded only 84.63–85.63%, suggesting weaker synergy compared to formulations containing GA. This supported earlier findings by Barbosa et al.,42 who emphasized that emulsion stability is directly correlated with higher ME, as stable emulsions prevent phase separation and surface deposition. The ternary formulation with equal proportions (33
50) yielded only 84.63–85.63%, suggesting weaker synergy compared to formulations containing GA. This supported earlier findings by Barbosa et al.,42 who emphasized that emulsion stability is directly correlated with higher ME, as stable emulsions prevent phase separation and surface deposition. The ternary formulation with equal proportions (33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33
33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33) produced 87.13%, whereas the blend dominated by maltodextrin (MD 67.66%, WPI 16.67%, GA 16.67%) provided the best result overall. This highlighted a single wall material cannot provide all necessary functionalities, while carefully designed blends exploit the strengths of each component.30
33) produced 87.13%, whereas the blend dominated by maltodextrin (MD 67.66%, WPI 16.67%, GA 16.67%) provided the best result overall. This highlighted a single wall material cannot provide all necessary functionalities, while carefully designed blends exploit the strengths of each component.30
Finally, it is worth noting that emulsion viscosity and rheology play essential roles. As Ozdemir et al.33 suggested, higher emulsion viscosity can enhance ME by reducing internal circulation and accelerating semi-permeable membrane formation. However, excessively high viscosity may decrease volatile retention by exposing encapsulates to high atomization temperatures.
3.4. Moisture content
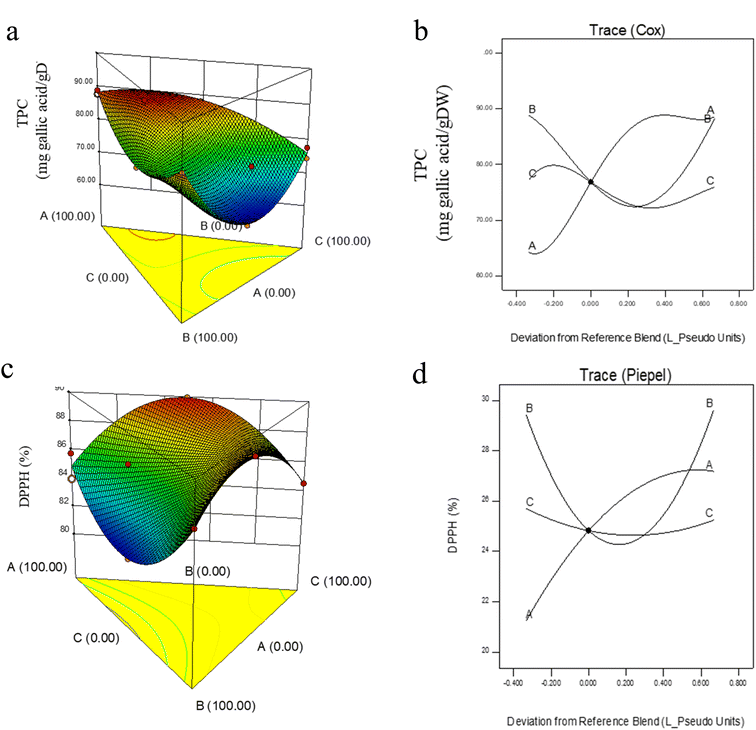
Moisture is a crucial quality parameter of dried powders, as it directly influences drying efficiency, flowability, stickiness, caking tendency, and storage stability, primarily through its effect on glass transition temperature and crystallization behavior.32 As shown in Fig. 1, the moisture content differed significantly among treatments (P < 0.05), with values ranging from 10.22% to 11.29%. Although these levels were slightly higher than the <4% typically recommended for industrial dried powders,32 they remained below the 10% threshold considered microbiologically safe.15 In contrast, the non-encapsulated control sample showed a considerably higher moisture content of approximately 12.8% ± 0.02, indicating its greater hygroscopicity and reduced drying stability when compared with all encapsulated formulations.43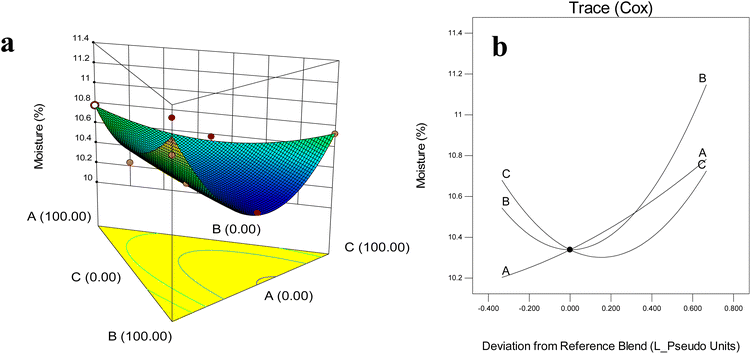 | ||
| Fig. 1 Effect of wall composition on moisture content: (a) response surface diagram, (b) Cox plot. A: MD (DE 20); B: WPI; C: GA. | ||
The type and proportion of wall materials had a decisive effect on the final moisture content, consistent with previous reports.27 Among treatments, the highest moisture was observed in the 100% WPI formulation (11.29%), which can be explained by the strong water-binding affinity of proteins and their rapid film formation during drying, which traps water inside the amorphous matrix.32 Literature similarly reports increased moisture when WPI is present in blends (up to 4.56%) compared to MD-only powders (∼2.55%).15 For example, Sarabandi et al.27 observed higher moisture in sour cherry juice powders prepared with WPI, confirming proteins' water-retention effect. Garlic extract powders encapsulated with WPI based had higher moisture (3.77%) than GA/CH (2.06%), reflecting proteins' stronger affinity for water.43
Formulations rich in GA tended to show higher moisture contents (10.72%), likely due to its branched heteropolysaccharide structure with abundant hydrophilic groups that bind water molecules and hinder their release.44 However, other studies reported contrasting outcomes depending on formulation: Kang et al.45 and Premi and Sharma46 demonstrated that higher GA levels in GA–MD blends often reduced moisture, underscoring the complexity of carrier interactions.
3.5. Water activity
Water activity (aw) is a critical parameter reflecting the amount of free water available in food systems, directly influencing biochemical reactions, microbial growth, and product stability.47 An increase in aw accelerates deterioration reactions, thereby reducing shelf life.47 In this study, the aw of barley leaf powders ranged between 0.34 and 0.40 (Fig. 2). These values indicated acceptable stability, but still fall close to the upper threshold, reinforcing the importance of wall material selection.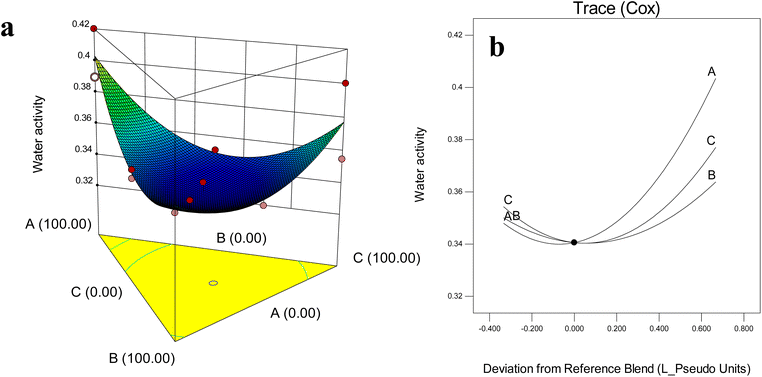 | ||
| Fig. 2 Effect of wall composition on water activity: (a) response surface diagram, (b) Cox plot. A: MD (DE 20); B: WPI; C: GA. | ||
The response surface and Cox plots (Fig. 2a and b) revealed that aw decreased as the proportions of MD, GA, and WPI increased toward the central region of the triangular mixture design. Beyond this central point, however, aw values began to rise again as all three wall materials increased simultaneously. Formulations rich in MD tended to exhibit higher aw compared to those dominated by GA or WPI, highlighting the distinct functional properties of different wall materials.
Protein-based (WPI) carriers generally increase aw due to their strong water-binding capacity, as reported for gurum seed oil and Wolffia globosa extracts.15,32 By contrast, carbohydrate-dominant systems (GA + MD) consistently achieve lower aw values. Likewise, anthocyanin-rich bilberry extracts encapsulated with MD and GA achieved aw ≤ 0.30, enhancing anthocyanin stability during storage.14 Similarly, GA–MD blends have been shown to reduce aw and improve encapsulation stability of blackberry powders.48,49
Other encapsulation studies confirmed these trends, for example, freeze-dried garlic extract encapsulated with WPI/CH or GA/CH achieved exceptionally low aw (<0.20), ensuring strong microbial resistance.43 Encapsulated β-carotene powders prepared with GA or almond gum reached aw values of 0.24–0.26, demonstrating excellent stability.50 In Welsh onion juice powders, aw values remained consistently between 0.27 and 0.37 regardless of GA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MD ratios, all below the 0.40 threshold considered safe.51
MD ratios, all below the 0.40 threshold considered safe.51
The interpretation of moisture content and aw results demonstrated that MD can retained water in a more labile state. Conversely, WPI-containing systems exhibited higher moisture content but lower aw, attributable to the strong water-binding properties of proteins. Therefore, the results emphasized that wall material composition governs not only total water content but also the physical state of water, with ternary MD–GA–WPI.
3.6. Bulk density
Bulk density is a critical property dried powders, with direct implications for transportation, storage, and packaging efficiency. It is strongly influenced by particle size, surface characteristics, and porosity; smoother, more uniform powders generally exhibit higher bulk density due to better packing ability.15 In this study, the bulk density of barley leaf powders varied significantly among treatments (P < 0.05), ranging from 0.189 to 0.276 g cm−3 (Fig. 3a and b).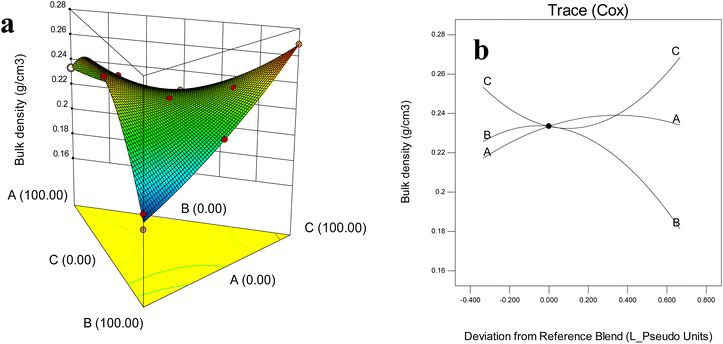 | ||
| Fig. 3 Effect of wall composition on microcapsule bulk density (g cm−3): (a) response surface diagram, (b) Cox plot. A: MD (DE 20); B: WPI; C: GA. | ||
The lowest density was observed in WPI-rich formulations (∼0.18 g cm−3), consistent with studies reporting that protein film formation traps air and increases porosity, creating spongy low-packing powders.32 For instance, WPI-based encapsulation of gurum seed oil (bulk density 0.16–0.26 g cm−3), where PB (WPI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MD, 2
MD, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) systems displayed markedly lower bulk densities compared to carbohydrate-dominant formulations.32 Likewise, W. globosa powders encapsulated with WPI exhibited reduced density (0.15–0.16 g cm−3) compared to MD-based systems (0.32 g cm−3), reinforcing the tendency of proteins to lower packing efficiency.15
1) systems displayed markedly lower bulk densities compared to carbohydrate-dominant formulations.32 Likewise, W. globosa powders encapsulated with WPI exhibited reduced density (0.15–0.16 g cm−3) compared to MD-based systems (0.32 g cm−3), reinforcing the tendency of proteins to lower packing efficiency.15
Powders with higher proportions of GA exhibited higher bulk density compared to those with MD (0.27 g cm−3). This behavior may be explained by highly branched heteropolysaccharide with covalently bound glycoproteins, which leads to improve viscosity and increased powder volume. In contrast, MD—owing to its higher excellent solubility, low viscosity—tends to occupy interstitial spaces more effectively, thereby increasing bulk density but still lower than GA-rich formulations.
3.7. Solubility index
Solubility is a fundamental functional property of dried powders because it determines their ability to be readily reconstituted in aqueous systems, thereby affecting the bioavailability of encapsulated compounds and their practical use in food formulations.44 According to (Fig. 4a and b), the solubility results (65.00–75.29%) showed significant differences among formulations (P < 0.05). The lowest solubility was obtained in powders prepared with the ternary combination of MD (16.67%), WPI; (66.67%), and GA (16.67%). This reduction was likely explained by proteins' high susceptibility to moisture-driven plasticization and thermal denaturation during drying, which promote protein aggregation and formation of dense, compact microstructures, limiting water permeability and delaying hydration during reconstitution.44 As well, this result in lined with Tg and moisture results (refer to Sections 3.4 and 3.8), due to formulations with lower Tg values—often associated with higher moisture—tended to demonstrate reduced solubility, particularly those with high WPI content as previously stated by Breen et al.52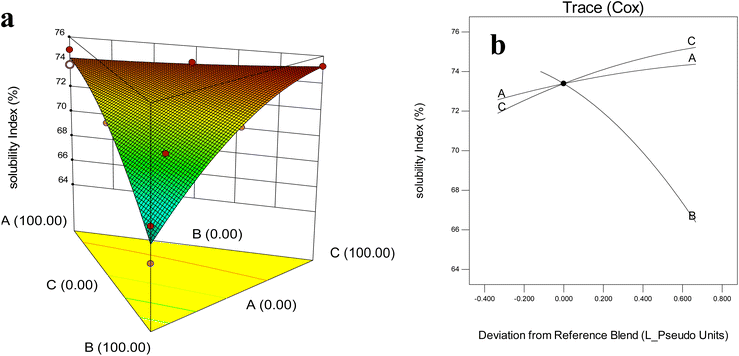 | ||
| Fig. 4 Effect of wall composition on solubility index: (a) response surface diagram, (b) Cox plot. A: MD (DE 20); B: WPI; C: GA. | ||
In contrast, MD–GA blends consistently produced the highest solubility values, attributable to their highly hydrophilic nature, low viscosity, and favorable rehydration properties, making them particularly effective carriers in encapsulation processes.
Fazaeli et al.53 showed that blackberry extract powders encapsulated with 2% MD (DE6) and 6% GA reached solubility levels as high as 87%. More recently, Sirichokworrakit et al.15 demonstrated that encapsulation of Wolffia globosa extract using MD alone achieved solubility up to 92.17%, whereas formulations containing GA and WPI showed reduced solubility (74.90%). Bazaria and Kumar54 similarly reported that MD and GA exhibited superior solubility compared to WPI-based formulations, owing to their higher water affinity and better structural compatibility.
Furthermore, the use of encapsulating agents in combination not only influenced the solubility index but also reduced solubility time, thereby improving wettability and reconstitution properties. For instance, Sirichokworrakit et al.15 observed that MD–GA–WPI blends in encapsulated extracts exhibited shorter solubility times compared to carbohydrate-only systems, a property desirable for instant beverages and functional food products requiring rapid dissolution. Medina-Jaramillo and López-Córdoba51 also reported that the addition of MD and GA improved the solubility of freeze-dried Welsh onion powders from 72% (without carriers) to 88%, highlighting their crucial role in instant food applications. Overall, for barley leaf microcapsules, the combination of MD and GA proved to be the most effective strategy for maximizing solubility, ensuring both technological functionality (instant reconstitution, stability, ease of handling) and nutritional effectiveness (enhanced delivery of bioactive compounds).
3.8. Glass transition temperatures
Differential scanning calorimetry (DSC) thermograms of all microcapsule formulations exhibited a single, clear glass transition step without detectable crystallization or additional exothermic/endothermic peaks, confirming the fully amorphous nature of the wall matrix. The absence of crystallization peaks indicated that no phase separation or ordered structural rearrangement occurred during heating, which is essential for maintaining uniform core entrapment and preventing thermally induced leakage.32T g variations among samples were mechanistically governed by intermolecular chain interactions and plasticization effects. Our results confirmed that wall material composition plays a decisive role in determining Tg. As shown in Table 3, the microcapsules containing maltodextrin (66.67%), WPI (16.67%), and GA (16.67%) exhibited the highest Tg among all formulations tested. This suggested that these capsules maintain structural rigidity longer at ambient temperature, delaying the transition into the rubbery state and enhancing storage stability. The improved thermal resistance was likely attributed to the higher molecular weight and film-forming capacity of GA, combined with the protective glassy nature of MD, which together create a more cohesive wall structure.51
Importantly, the Tg differences observed in our formulations closely correspond to the moisture variations reported in Section 3.4. This relationship was strongly supported by Levine and Slade55 identifying water as the most influential plasticizer in amorphous food systems even small increases in moisture markedly depress the Tg of synthetic and natural polymers by enhancing molecular mobility. As well, no evidence of chemical reactions (e.g., oxidation, degradation, or Maillard-related thermal crosslinking) was detected during scanning, indicating that the polymer carriers remained chemically stable within the tested temperature range. This thermal behavior confirmed that release of encapsulated bioactive compounds above Tg is a diffusion-controlled physical process rather than a reaction-induced rupture or degradation event.32 Comparable results have been reported in previous encapsulation studies. For example, Ozdemir et al.33 demonstrated that basil essential oil microcapsules prepared with GA:WPI and GA:WPI:MD combinations showed significantly higher Tg values (81 °C and 71 °C, respectively) than those produced with WPI:MD alone (58.5 °C), indicating superior resistance to moisture-induced plasticization. Similarly, Karrar et al.32 observed that GA substantially increased Tg compared to MD, due to its higher molar mass and stronger intermolecular interactions.
3.9. Particle size distribution and morphology
The particle size of barley leaf microcapsules, expressed as the volume-weighted mean diameter (D[4, 3]), ranged from 34.50 and 37.59 µm across formulations (Table 3). These values fall within the broad particle size spectrum (20–5000 µm) commonly reported for freeze-dried powders, where ice sublimation and structural collapse during drying typically generate heterogeneous, irregular particles.56 Notably, formulations containing higher proportions of GA (100%) produced the largest particles (37.59 µm), suggesting that the high viscosity and strong emulsifying properties of GA promoted the formation of bulkier and more cohesive particles, a trend that was further reflected in their bulk density behavior. In contrast, samples rich in WPI consistently showed the smallest diameters (34.50–35.01 µm), a finding consistent with their strong film-forming capacity that entraps air, yielding a spongy and porous microcapsule structure.Maltodextrin-dominated systems (100% MD or MD-rich blends) resulted in intermediate sizes (35.21–36.31 µm). This trend indicates that MD, with its's excellent solubility, low viscosity, tended to produce looser particle. Similarly, purple corn anthocyanin powders encapsulated with MD alone displayed small and uniform particles (8–80 µm), whereas MD–GA and MD–WPI systems resulted in broader distributions (10–100 µm) because of increased polydispersity and aggregation.57
Interestingly, ternary blends such as MD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WPI
WPI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA (33.33
GA (33.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33.33
33.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33.33%) and MD
33.33%) and MD![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) WPI
WPI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA (16.67
GA (16.67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 66.67
66.67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16.67%) yielded the smallest average sizes (34.50–34.72 µm), implying a synergistic effect between protein–polysaccharide interactions that restricted particle growth.
16.67%) yielded the smallest average sizes (34.50–34.72 µm), implying a synergistic effect between protein–polysaccharide interactions that restricted particle growth.
Our results aligned with grape seed extract encapsulation trials, where binary WPC–MD and WPC–GA systems (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 or 3
1 or 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) produced smaller particles compared to single carriers, underscoring the stabilizing effect of polysaccharide–protein interactions.58 As well, basil essential oil microcapsules exhibited much smaller sizes (0.47–4.18 µm), with GA and GA–protein systems forming homogeneous distributions, illustrating the influence of emulsion viscosity and stability on final size.33
2) produced smaller particles compared to single carriers, underscoring the stabilizing effect of polysaccharide–protein interactions.58 As well, basil essential oil microcapsules exhibited much smaller sizes (0.47–4.18 µm), with GA and GA–protein systems forming homogeneous distributions, illustrating the influence of emulsion viscosity and stability on final size.33
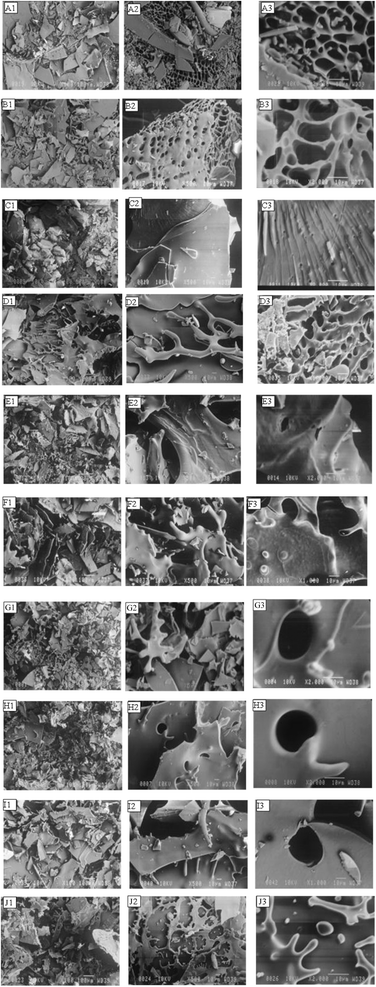
The microstructure of young barley leaf powders, as examined by SEM shown in Fig. 5, clearly demonstrates the powders prepared GA based powders showed deeper surface wrinkles. This effect can be explained by the rapid formation of a proteinaceous shell during the early drying stages, which limits water diffusion across the matrix.59 Increasing GA concentration further enhanced feed viscosity and film thickness, slowing down water evaporation and yielding particles with greater wrinkling.27
SEM images of the prepared microcapsules at different magnifications (50×, 500×, and 1000×) are presented in Fig. 5. The freeze-dried microcapsules lacked defined spherical shapes and instead exhibited irregular, flake-like, and porous morphologies. This is typical of freeze-dried powders, where sublimation of ice crystals under low pressure and temperature leaves behind sponge-like or plate-like aggregates.60 Such irregular glassy structures, formed after milling and sieving, reflect the mechanical stress of processing. These findings were consistent with previous freeze-dried encapsulates of bilberry extract, buriti oil, and onion leaf juice, all of which exhibited broken-glass or crystalline-like morphologies.14,47,51
Notably, wall composition strongly influenced surface features. Microcapsules prepared with MD and GA showed smooth but uneven surfaces, with limited agglomeration and slight surface curvature. By contrast, capsules formed solely with GA or MD were more brittle, displaying deeper surface indentations and wrinkles. Similar trends have been observed in basil essential oil and Wolffia globosa extract microcapsules, where MD produced smoother but more cracked structures, while GA caused irregular, shrunken particles with concave shapes.15,61 WPI, either alone or combined with polysaccharides, consistently reduced shrinkage and surface cracking, producing smoother morphologies due to its greater film-forming ability and flexibility during drying.15,32 These smoother structures were particularly relevant, as they were associated with improved encapsulation efficiency and better retention of bioactive compounds.
Such variability highlights the importance of tailoring encapsulation systems: while MD provides good solubility and smooth surfaces, its high hygroscopicity may lead to stickiness at higher loads.14 GA ensures emulsion stability and smaller particle sizes but introduces greater shrinkage, whereas WPI imparts flexibility and stability to the capsule wall, minimizing cracks and enhancing structural cohesiveness.
3.10. Total phenolic content and antioxidant activity
The total phenolic content (TPC) of the barley leaf microcapsules ranged from 57.11 to 69.97 mg g−1 (Fig. 6a and b). Although the numerical range appears moderate, the differences among formulations were statistically significant (p < 0.05), and the quadratic model demonstrated excellent predictive performance (R2 = 0.99; Pred-R2 = 0.97; Table 2), confirming that wall material composition exerted a meaningful influence on phenolic retention. Among the tested formulations, the microcapsules prepared with a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 blend of MD and GA exhibited the highest phenolic retention, while those containing 100% GA showed the lowest levels. In contrast, the non-encapsulated control sample showed a markedly lower estimated TPC of approximately 50 mg g−1 ± 0.17. This outcome confirmed that wall material composition plays a decisive role in determining the efficiency of phenolic preservation during drying. Previous studies also highlighted that wall–core interactions, the volatility of encapsulated compounds, and the molecular size and flexibility of wall materials strongly affect the diffusion of phenolics through the capsule wall, and thus their stability.15
50 blend of MD and GA exhibited the highest phenolic retention, while those containing 100% GA showed the lowest levels. In contrast, the non-encapsulated control sample showed a markedly lower estimated TPC of approximately 50 mg g−1 ± 0.17. This outcome confirmed that wall material composition plays a decisive role in determining the efficiency of phenolic preservation during drying. Previous studies also highlighted that wall–core interactions, the volatility of encapsulated compounds, and the molecular size and flexibility of wall materials strongly affect the diffusion of phenolics through the capsule wall, and thus their stability.15
The Cox response plots (Fig. 6) demonstrated that TPC initially increased with rising MD concentration up to 33.33%, but declined thereafter. A similar trend was observed in MD–GA combinations, suggesting an optimal carrier ratio for maximizing phenolic retention. These findings were consistent with earlier reports showing that MD with DE 20 provides superior oxygen barrier properties, which reduce oxidative degradation and favor the entrapment of sensitive polyphenols.6,62 This protective capacity is complemented by its high solubility and low viscosity, enabling MD to more effectively occupy interstitial spaces and decrease bulk density, thereby enhancing the structural compactness and stability of the powders.15,61 Comparable observations were made in bilberry and grape seed extract studies, where MD-based carriers provided higher TPC protection than GA alone.14,58
The DPPH radical scavenging activity also differed significantly across formulations (p < 0.05; Fig. 6c and d), supported by strong model statistics (R2 = 0.98; Pred-R2 = 0.96; Table 2). The highest radical scavenging activity was observed in ternary formulations containing MD (66.67%), WPI (16.67%), and GA (16.67%), while the lowest was detected in capsules made of equal proportions of WPI and GA and the control (60% ± 0.22). The MD–WPI blend also produced comparatively low activity, although its values were slightly higher than those of WPI–GA. Cox response plots (Fig. 6d) also indicated that radical scavenging activity increased with MD up to 66.67% and then decreased, while higher proportions of GA and WPI contributed positively within moderate levels. The synergistic effect of MD's protective film-forming capacity, GA's stabilizing and emulsifying properties, and WPI's inherent antioxidant activity through sulfhydryl groups has also been highlighted in studies on beetroot and mulberry polyphenol encapsulation.61,63 As well these results aligned with prior work on mulberry juice and purple corn anthocyanins, where MD–GA systems outperformed single-carrier or protein-dominant systems in stabilizing phenolic compounds during drying.57 Additionally, WPI incorporation was found to enhance antioxidant performance, consistent with reports that moderate protein fractions improve phenolic entrapment and bioactivity.46,58 Generally, these results confirmed that MD provided excellent oxygen resistance and solubility, GA enhanced stability and film strength, and WPI contributed functional antioxidant activity. This structural synergy not only protects phenolic compounds during freeze drying but also improves their release and functional performance upon rehydration, making these formulations particularly promising for use in functional foods and nutraceutical powders.
3.11. Optimization
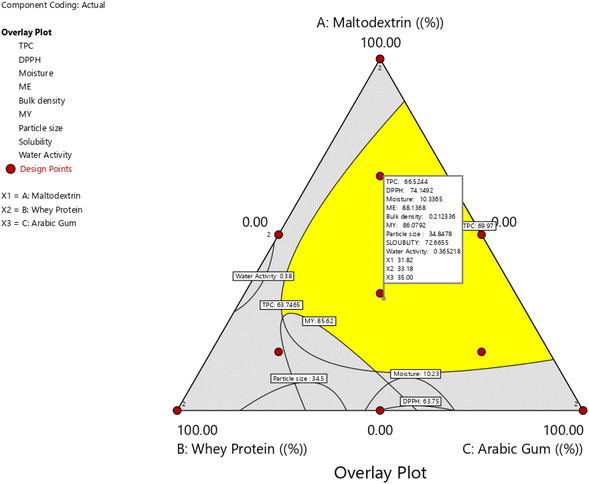
The optimal concentrations of wall materials for the microencapsulation of young barley leaf extract were determined using both numerical and graphical optimization. The graphical optimization results supporting the selection of the optimal wall material ratios are presented in Fig. 7.The primary objectives were to maximize MY and EE, while simultaneously maintaining favorable physicochemical and functional properties. The desirability function identified an optimal wall composition of 31.81% MD, 35.00% GA, and 33.18% WPI.
Although the numerical differences among the tested formulations were moderate, the statistical Quadratic models used for optimization demonstrated strong predictive performance for key responses (MY: R2 = 0.99; EE: R2 = 0.74; moisture content: R2 = 0.91; TPC: R2 = 0.99; DPPH activity: R2 = 0.98; Table 2). These diagnostics indicated that even modest shifts in wall material ratios produced statistically meaningful effects—especially for responses most relevant to microencapsulation stability and efficiency.
To ensure reproducibility and optimization robustness, the predicted optimal formulation was experimentally validated by preparing three fully independent microcapsule production batches (n = 3).
Under the optimized conditions, the microcapsules achieved a production yield of 86.08%, encapsulation efficiency of 88.13%, TPC of 66.52 mg g−1, and radical scavenging activity of 74.15%. Moisture content (10.37%) and bulk density (0.21 g cm−3) also remained within desirable ranges, supporting powder stability.
To ensure robustness, the optimal formulation was experimentally validated. Independent experiments conducted at the predicted optimal ratios produced results that were statistically indistinguishable from model predictions (p > 0.05), as shown in Table 4. This agreement confirmed that the optimization procedure was reliable and that the selected formulation provides a consistent and effective matrix for protecting the bioactive compounds in young barley leaf extract.
| Results | MY (%) | ME (%) | TPC (mg g−1) | Moisture (%) | Bulk density (g cm−3) |
|---|---|---|---|---|---|
| a Values with the same letters within each column (mean ± standard deviation) are not significantly different according to Duncan's multiple range test (p < 0.05). | |||||
| RSM | 86.08a | 88.13a | 66.52a | 10.37a | 0.21a |
| Test | 84.06 ± 0.09a | 0.01a ± 87.5 | 0.24a ± 67.3 | 1.05a ± 10.30 | 1.05a ± 0.23 |
4. Conclusion
This study demonstrated that the choice and proportion of wall materials play a decisive role in determining the physicochemical and functional properties of young barley leaf microcapsules. MD consistently enhanced powder yield, glass transition temperature, and solubility due to its favorable drying behavior, oxygen barrier capacity, and structural compactness. GA, with its strong emulsifying and film-forming capacity, improved emulsion stability and bulk density, though its higher viscosity limited yield at elevated levels. WPI contributed superior emulsification and film formation, while also enhancing antioxidant activity, but its heat sensitivity and strong water-binding capacity tended to increase moisture content and reduce bulk density.Synergistic effects were most evident in blended systems, particularly the ternary formulation dominated by MD with moderate proportions of GA and WPI, which achieved the highest encapsulation efficiency (89.98%) and antioxidant activity (80.39%). Optimization further confirmed that a composition of 31.81% MD, 35.00% GA, and 33.18% WPI provided the most desirable balance of yield, efficiency, phenolic retention, and stability.
From an industrial perspective, the optimized microencapsulated barley leaf powder makes it suitable for incorporation into instant drink mixes, fortified bakery items, smoothies, dairy beverages, capsule supplements, and powdered nutraceutical blends. On the other hand, as young barley leaves require dedicated cultivation rather than utilizing agricultural residues, enhancing their stability and reducing nutrient loss through optimized microencapsulation supports more resource-efficient use of this high-value biomass. Future research should evaluate industrial scalability, long-term storage stability, digestive bioavailability, and integration with more sustainable drying technologies or circular-bioprocessing strategies to further strengthen economic and environmental viability.
Conflicts of interest
There are no conflicts to declare.Data availability
All data generated or analyzed during this study are included in this published article. Also, more information is available from the corresponding author on request.Acknowledgements
We would like to express our sincere gratitude to Green Sprout Technology Development Company, for all the support related to our research.References
- E. Kato, A. Tsuruma, A. Amishima and H. Satoh, Proteinous pancreatic lipase inhibitor is responsible for the antiobesity effect of young barley (Hordeum vulgare L.) leaf extract, Biosci. Biotechnol. Biochem., 2021, 85(8), 1885–1889 CrossRef PubMed.
- M. K. Lemieszek, I. Komaniecka, M. Chojnacki, A. Choma and W. Rzeski, Immunomodulatory properties of polysaccharide-rich young green barley (Hordeum vulgare) extract and its structural characterization, Molecules, 2022, 27(5), 1742 Search PubMed.
- K. J. Gromkowska-Kępka, R. Markiewicz-Żukowska, P. Nowakowski, S. K. Naliwajko, J. Moskwa and A. Puścion-Jakubik, et al., Chemical composition and protective effect of young barley (Hordeum vulgare L.) dietary supplements extracts on UV-Treated human skin fibroblasts in in vitro studies, Antioxidants, 2021, 10(9), 1402 CrossRef PubMed.
- M. Wang, C. Zhang, Y. Xu, M. Ma, T. Yao and Z. Sui, Impact of six extraction methods on molecular composition and antioxidant activity of polysaccharides from young hulless barley leaves, Foods, 2023, 12(18), 3381 CrossRef CAS PubMed.
- M. Kozłowska, M. Ziarno, D. Zaręba and I. Ścibisz, Exploring the possibility of enriching fermented milks with young barley leaves powder preparation, Fermentation, 2023, 9(8), 731 CrossRef.
- K. Sarabandi, S. M. Jafari, A. S. Mahoonak and A. Mohammadi, Application of gum Arabic and maltodextrin for encapsulation of eggplant peel extract as a natural antioxidant and color source, Int. J. Biol. Macromol., 2019, 140, 59–68 CrossRef CAS PubMed.
- C. Drosou and M. Krokida, A comparative study of encapsulation of β-carotene via spray-drying and freeze-drying techniques using pullulan and whey protein isolate as wall material, Foods, 2024, 13(12), 1933 CrossRef CAS PubMed.
- K. Papoutsis, J. B. Golding, Q. Vuong, P. Pristijono, C. E. Stathopoulos and C. J. Scarlett, et al., Encapsulation of citrus by-product extracts by spray-drying and freeze-drying using combinations of maltodextrin with soybean protein and ι-carrageenan, Foods, 2018, 7(7), 115 CrossRef PubMed.
- L. F. Ballesteros, M. J. Ramirez, C. E. Orrego, J. A. Teixeira and S. I. Mussatto, Encapsulation of antioxidant phenolic compounds extracted from spent coffee grounds by freeze-drying and spray-drying using different coating materials, Food Chem., 2017, 237, 623–631 Search PubMed.
- S. A. Ledari, J. M. Milani, S.-A. Shahidi and A. Golkar, Comparative analysis of freeze drying and spray drying methods for encapsulation of chlorophyll with maltodextrin and whey protein isolate, Food Chem.: X, 2024, 21, 101156 Search PubMed.
- L. Pudziuvelyte, M. Marksa, K. Sosnowska, K. Winnicka, R. Morkuniene and J. Bernatoniene, Freeze-drying technique for microencapsulation of Elsholtzia ciliata ethanolic extract using different coating materials, Molecules, 2020, 25(9), 2237 CrossRef CAS PubMed.
- M. E. da Silva Júnior, M. V. R. L. Araújo, A. C. S. Martins, M. dos Santos Lima, F. L. H. Da Silva and A. Converti, et al., Microencapsulation by spray-drying and freeze-drying of extract of phenolic compounds obtained from ciriguela peel, Sci. Rep., 2023, 13(1), 15222 CrossRef PubMed.
- P. Choudhary, S. Dutta, J. Moses and C. Anandharamakrishnan, Recent developments in encapsulation of α-lipoic acid for enhanced bioavailability and stability, Qual. Assur. Saf. Crop Foods, 2023, 15(1), 123–138 CrossRef CAS.
- A. Todorović, L. Šturm, A. Salević-Jelić, S. Lević, I. G. Osojnik Črnivec and I. Prislan, et al., Encapsulation of bilberry extract with maltodextrin and gum arabic by freeze-drying: Formulation, characterisation, and storage stability, Processes, 2022, 10(10), 1991 CrossRef.
- S. Sirichokworrakit, N. Aukkanit and C. Sangsuwon, Optimizing Encapsulation Strategies for Enhanced Antioxidant Stability of Wolffia globosa Extracts: A Comparative Study of Maltodextrin, Gum Arabic, and Whey Protein Concentrate, Appl. Food Res., 2025, 101030 CrossRef CAS.
- P. Ezhilarasi, D. Indrani, B. S. Jena and C. Anandharamakrishnan, Freeze drying technique for microencapsulation of Garcinia fruit extract and its effect on bread quality, J. Food Eng., 2013, 117(4), 513–520 CrossRef CAS.
- S. Einafshar, A. Rohani, Y. Sabeghi, M. H. Tavassoli-Kafrani, R. Farhoosh and D. Gandomzadeh, Unveiling the power of bene (Pistacia atlantica) hull scum: Boosting oxidative stability with methanolic extract and ferrous ions, Food Chem., 2025, 466, 142142 CrossRef CAS PubMed.
- P. Sharayei, A. Rohani, Y. Sabeghi and D. Gandomzadeh, The impact of drying techniques on stabilizing microencapsulated astaxanthin from shrimp shells: A comparative study of spray drying versus freeze drying, J. Food Process Eng., 2024, 47(11), e14755 CrossRef CAS.
- R. González-Ortega, M. Faieta, C. D. Di Mattia, L. Valbonetti and P. Pittia, Microencapsulation of olive leaf extract by freeze-drying: Effect of carrier composition on process efficiency and technological properties of the powders, J. Food Eng., 2020, 285, 110089 CrossRef.
- P. Sharayei, E. Azarpazhooh, S. Einafshar, S. Zomorodi, F. Zare and H. S. Ramaswamy, Optimization of wall materials for astaxanthin powder production from shrimp shell extract using simplex lattice mixture design, J. Food Process. Preserv., 2024, 2024(1), 9794290 Search PubMed.
- E. Azarpazhooh, P. Sharayei, X. Rui, M. Gharibi-Tehrani and H. S. Ramaswamy, Optimization of wall material of freeze-dried high-bioactive microcapsules with yellow onion rejects using simplex centroid mixture design approach based on whey protein isolate, pectin, and sodium caseinate as incorporated variables, Molecules, 2022, 27(23), 8509 CrossRef CAS PubMed.
- E. Azarpazhooh, P. Sharayei, S. Zomorodi and H. S. Ramaswamy, Physicochemical and phytochemical characterization and storage stability of freeze-dried encapsulated pomegranate peel anthocyanin and in vitro evaluation of its antioxidant activity, Food Bioprocess Technol., 2019, 12(2), 199–210 CrossRef CAS.
- S. Einafshar, B. Tajeddin, Y. Sabeghi and D. Gandomzadeh, Optimization of Biodegradable Potato Starch–Nanocellulose Films: Influence of Starch Content, Nanocellulose Concentration, and Plasticizer Type, Starch/Staerke, 2025, e70112 CrossRef CAS.
- P. Sharayei, R. Niazmand, M. S. Jamab, Y. Sabeghi, S. Einafshar and E. Azarpazhooh, et al., Bioactive films from saffron corm and corn husk: A transformative solution for antioxidant and antimicrobial pistachio packaging, LWT-Food Sci. Technol., 2025, 118097 CrossRef CAS.
- Y. Sabeghi, M. Varidi and M. Nooshkam, Bioactive foamulsion gels: a unique structure prepared with gellan gum and Acanthophyllum glandulosum extract, J. Sci. Food Agric., 2024, 104(7), 3853–3864 CrossRef CAS PubMed.
- E. Azarpazhooh, P. Sharayei, Y. Sabeghi, F. Zare, X. Rui and H. S. Ramaswamy, Modified atmosphere packaging of sunflower microgreens (Helianthus annuus) for quality and postharvest shelf-life extension, Sustainable Food Technol., 2026, 15, 2524–2535 Search PubMed.
- K. Sarabandi, S. H. Peighambardoust, A. S. Mahoonak and S. P. Samaei, Effect of carrier types and compositions on the production yield, microstructure and physical characteristics of spray dried sour cherry juice concentrate, J. Food Meas. Char., 2017, 11(4), 1602–1612 CrossRef.
- A. M. Goula and K. G. Adamopoulos, Effect of maltodextrin addition during spray drying of tomato pulp in dehumidified air: II. Powder properties, Dry. Technol., 2008, 26(6), 726–737 Search PubMed.
- W. Wang, Y. Jiang and W. Zhou, Characteristics of soy sauce powders spray-dried using dairy whey proteins and maltodextrins as drying aids, J. Food Eng., 2013, 119(4), 724–730 CrossRef CAS.
- Q. Shi, Z. Fang and B. Bhandari, Effect of addition of whey protein isolate on spray-drying behavior of honey with maltodextrin as a carrier material, Dry. Technol., 2013, 31(13–14), 1681–1692 CrossRef CAS.
- A. A. Mahdi, J. K. Mohammed, W. Al-Ansi, A. D. Ghaleb, Q. A. Al-Maqtari and M. Ma, et al., Microencapsulation of fingered citron extract with gum arabic, modified starch, whey protein, and maltodextrin using spray drying, Int. J. Biol. Macromol., 2020, 152, 1125–1134 CrossRef CAS PubMed.
- E. Karrar, A. A. Mahdi, S. Sheth, I. A. M. Ahmed, M. F. Manzoor and W. Wei, et al., Effect of maltodextrin combination with gum arabic and whey protein isolate on the microencapsulation of gurum seed oil using a spray-drying method, Int. J. Biol. Macromol., 2021, 171, 208–216 CrossRef CAS PubMed.
- N. Ozdemir, A. Bayrak, T. Tat, F. Altay, M. Kiralan and A. Kurt, Microencapsulation of basil essential oil: Utilization of gum arabic/whey protein isolate/maltodextrin combinations for encapsulation efficiency and in vitro release, J. Food Meas. Char., 2021, 15(2), 1865–1876 CrossRef.
- D. Gandomzadeh, M. H. Saeidirad, Y. Sabeghi, A. Rohani, E. Azarpazhooh and Y. Saeidirad, et al., A comprehensive review of drying techniques and quality for saffron, J. Food Meas. Char., 2024, 18(10), 8218–8232 CrossRef.
- P. Sharayei, E. Azarpazhooh, F. Zare and Y. Sabeghi, Analysis of the preservation of the antioxidant, antimicrobial, and sensory capacity of free and microencapsulated shrimp shell extract applied in chicken lunch meat, Translat. Food Sci., 2025, 1(1), vxaf006 Search PubMed.
- S. Meena, W. Prasad, K. Khamrui, S. Mandal and S. Bhat, Preparation of spray-dried curcumin microcapsules using a blend of whey protein with maltodextrin and gum arabica and its in-vitro digestibility evaluation, Food Biosci., 2021, 41, 100990 CrossRef CAS.
- A. Rezvankhah, Z. Emam-Djomeh, M. Safari, M. Salami and G. Askari, Investigating the effects of maltodextrin, gum arabic, and whey protein concentrate on the microencapsulation efficiency and oxidation stability of hemp seed oil, J. Food Process. Preserv., 2022, 46(6), e16554 CAS.
- S. Habib, S. M. Wani, K. Rasool, B. Ashaq, N. Anjum and S. A. Padder, et al., Nanoencapsulation of astaxanthin using gum arabic and whey protein isolate as wall materials: Characterization and invitro release kinetics, Powder Technol., 2025, 458, 120982 CrossRef CAS.
- R. Rashid, S. M. Wani, S. Manzoor, F. Masoodi and A. Altaf, Nanoencapsulation of pomegranate peel extract using maltodextrin and whey protein isolate. Characterisation, release behaviour and antioxidant potential during simulated invitro digestion, Food Biosci., 2022, 50, 102135 CrossRef CAS.
- A. Bassijeh, S. Ansari and S. M. H. Hosseini, Astaxanthin encapsulation in multilayer emulsions stabilized by complex coacervates of whey protein isolate and Persian gum and its use as a natural colorant in a model beverage, Food Res. Int., 2020, 137, 109689 CrossRef CAS PubMed.
- A. M. Cabrales-González, D. M. Núñez-Ramírez, M. A. Martínez-Prado, L. Medina-Torres, W. Rosas-Flores and O. Manero, Microencapsulation of Acidithiobacillus thiooxidans in a biopolymeric matrix of gum arabic and whey protein using complex coacervation and freeze drying, Miner. Eng., 2025, 222, 109164 CrossRef.
- M. Barbosa, C. Borsarelli and A. Mercadante, Light stability of spray-dried bixin encapsulated with different edible polysaccharide preparations, Food Res. Int., 2005, 38(8–9), 989–994 CrossRef CAS.
- L. Tavares, H. L. B. Barros, J. C. P. Vaghetti and C. P. Z. Noreña, Microencapsulation of garlic extract by complex coacervation using whey protein isolate/chitosan and gum arabic/chitosan as wall materials: Influence of anionic biopolymers on the physicochemical and structural properties of microparticles, Food Bioprocess Technol., 2019, 12(12), 2093–2106 CrossRef CAS.
- S. Meena, S. Gote, W. Prasad and K. Khamrui, Storage stability of spray dried curcumin encapsulate prepared using a blend of whey protein, maltodextrin, and gum Arabic, J. Food Process. Preserv., 2021, 45(5), e15472 CAS.
- Y.-R. Kang, Y.-K. Lee, Y. J. Kim and Y. H. Chang, Characterization and storage stability of chlorophylls microencapsulated in different combination of gum Arabic and maltodextrin, Food Chem., 2019, 272, 337–346 CrossRef CAS PubMed.
- M. Premi and H. Sharma, Effect of different combinations of maltodextrin, gum arabic and whey protein concentrate on the encapsulation behavior and oxidative stability of spray dried drumstick (Moringa oleifera) oil, Int. J. Biol. Macromol., 2017, 105, 1232–1240 CrossRef CAS PubMed.
- J. P. de Oliveira, O. P. Almeida, P. H. Campelo, G. Carneiro, LdO. F. Rocha and J. H. Santos, et al., Tailoring the physicochemical properties of freeze-dried buriti oil microparticles by combining inulin and gum Arabic as encapsulation agents, Lwt, 2022, 161, 113372 CrossRef.
- R. V. Tonon, C. Brabet and M. D. Hubinger, Anthocyanin stability and antioxidant activity of spray-dried açai (Euterpe oleracea Mart.) juice produced with different carrier agents, Food Res. Int., 2010, 43(3), 907–914 CrossRef CAS.
- C. C. Ferrari, S. P. Marconi Germer, I. D. Alvim and J. M. de Aguirre, Storage stability of spray-dried blackberry powder produced with maltodextrin or gum arabic, Dry. Technol., 2013, 31(4), 470–478 CrossRef CAS.
- N. Mahfoudhi and S. Hamdi, Kinetic degradation and storage stability of β-carotene encapsulated by freeze-drying using almond gum and gum Arabic as wall materials, J. Food Process. Preserv., 2015, 39(6), 896–906 CrossRef CAS.
- C. Medina-Jaramillo and A. López-Córdoba, Enhancing the Physicochemical, Thermal, and Technological Properties of Freeze-Dried Welsh Onion Leaf Juice: Influence of Maltodextrin and Gum Arabic as Carrier Agents, Polymers, 2025, 17(6), 801 CrossRef CAS PubMed.
- E. Breen, J. Curley, D. Overcashier, C. Hsu and S. Shire, Effect of moisture on the stability of a lyophilized humanized monoclonal antibody formulation, Pharm. Res., 2001, 18(9), 1345–1353 Search PubMed.
- M. Fazaeli, Z. Emam-Djomeh, A. K. Ashtari and M. Omid, Effect of spray drying conditions and feed composition on the physical properties of black mulberry juice powder, Food Bioprod. Process., 2012, 90(4), 667–675 CrossRef CAS.
- B. Bazaria and P. Kumar, Optimization of spray drying parameters for beetroot juice powder using response surface methodology (RSM), J. Saudi Soc. Agric. Sci., 2018, 17(4), 408–415 CrossRef.
- H. Levine and L. Slade, Water as a plasticizer: physico-chemical aspects of low-moisture polymeric systems, Water Sci. Rev., 1988, 3(1), 79–185 Search PubMed.
- N. J. Zuidam and E. Shimoni, Overview of microencapsulates for use in food products or processes and methods to make them, Encapsulation Technologies for Active Food Ingredients and Food Processing, Springer, 2009, pp. 3–29 Search PubMed.
- W. Deng, X. Li, G. Ren, Q. Bu, Y. Ruan and Y. Feng, et al., Stability of purple corn anthocyanin encapsulated by maltodextrin, and its combinations with gum arabic and whey protein isolate, Foods, 2023, 12(12), 2393 CrossRef CAS PubMed.
- K. Yadav, R. K. Bajaj, S. Mandal and B. Mann, Encapsulation of grape seed extract phenolics using whey protein concentrate, maltodextrin and gum arabica blends, J. Food Sci. Technol., 2020, 57(2), 426–434 Search PubMed.
- R. V. Tonon, C. Brabet and M. D. Hubinger, Influence of process conditions on the physicochemical properties of açai (Euterpe oleraceae Mart.) powder produced by spray drying, J. Food Eng., 2008, 88(3), 411–418 Search PubMed.
- C. Luo, Z. Liu, S. Mi and L. Li, Quantitative investigation on the effects of ice crystal size on freeze-drying: The primary drying step, Dry. Technol., 2022, 40(2), 446–458 CrossRef CAS.
- S. Yousefi, M. Kavyanirad, M. Aminifar, W. Weisany and A. Mousavi Khaneghah, Yogurt fortification by microencapsulation of beetroot extract (Beta vulgaris L.) using maltodextrin, gum arabic, and whey protein isolate, Food Sci. Nutr., 2022, 10(6), 1875–1887 CrossRef CAS PubMed.
- S. A. Mahdavi, S. M. Jafari, E. Assadpoor and D. Dehnad, Microencapsulation optimization of natural anthocyanins with maltodextrin, gum Arabic and gelatin, Int. J. Biol. Macromol., 2016, 85, 379–385 CrossRef PubMed.
- B. Bazaria and P. Kumar, Effect of whey protein concentrate as drying aid and drying parameters on physicochemical and functional properties of spray dried beetroot juice concentrate, Food Biosci., 2016, 14, 21–27 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |