 Open Access Article
Open Access ArticleSustainable conversion of waste robusta coffee husk into tea: optimization, properties and diffusion kinetics
Thi-Ngoc-Mai Trana,
Duong Anh Tana,
Nguyen Huu Trungb,
Thi-Hong-No Nguyen c,
Phung Van Trungc,
N. Trung-Tien Dieuc,
Van-Dung Le
c,
Phung Van Trungc,
N. Trung-Tien Dieuc,
Van-Dung Le c,
Dinh Tri Mai*c,
Phuoc-Dat Duongc,
Chi-Hien Dang
c,
Dinh Tri Mai*c,
Phuoc-Dat Duongc,
Chi-Hien Dang c,
Cao-Hien Nguyend,
Nguyen Thi Thanh Tu
c,
Cao-Hien Nguyend,
Nguyen Thi Thanh Tu e and
Thanh-Danh Nguyen
e and
Thanh-Danh Nguyen *c
*c
aInstitute of Applied Sciences, HUTECH University, 475A Dien Bien Phu Street, Thanh My Tay Ward, Ho Chi Minh City, 700000, Vietnam
bIndustrial University of Ho Chi Minh City, Faculty of Chemical Engineering, 12 Nguyen Van Bao, Go Vap District, Ho Chi Minh City, 700000, Vietnam
cInstitute of Advanced Technology, 1B TL29 Street, An Phu Dong Ward, Ho Chi Minh City, 700000, Vietnam. E-mail: maidinhtri@gmail.com; danh5463bd@yahoo.com
dDepartment of Chemical Technology, Ho Chi Minh City University of Industry and Trade, Ho Chi Minh City 700000, Vietnam
eFaculty of Applied Technology, School of Technology, Van Lang University, Ho Chi Minh City, Vietnam. E-mail: tu.ntt@vlu.edu.vn
First published on 16th March 2026
Abstract
This study presents a sustainable approach to upcycle Robusta coffee husk (RCH), a major agro-industrial residue into an instant coffee husk tea product. By combining optimized roasting and aqueous extraction, the natural polyphenols, organic acids, and aromatic precursors of RCH were effectively recovered and stabilized. Response surface methodology guided the selection of mild processing conditions that enhanced antioxidant potential while maintaining sensory quality. Microscopy, FTIR and headspace GC-MS analyses revealed that roasting transformed the dense lignocellulosic matrix into a porous, extractable structure and generated desirable aroma compounds through Maillard and caramelization reactions. The resulting extract was formulated into an instant coffee husk tea powder with balanced flavor, high solubility, and microbial safety, demonstrating its applicability as a natural beverage or functional ingredient. Diffusion-kinetic modeling of phenolic release fitted Fick's second law at 35–45 °C (R2 > 0.96), with diffusion coefficients ranging from 1.73 × 10−9 to 3.19 × 10−9 cm2 s−1. This work highlights the underexplored potential of coffee husk as a sustainable bioresource for food applications, contributing to waste reduction and circular bioeconomy development.
Sustainability spotlightThis study provides a sustainable and scalable valorization pathway for Robusta coffee husk, an abundant agro-industrial residue that is often discarded or burned, contributing to environmental pollution. By integrating response surface-optimized roasting and aqueous extraction, the process efficiently recovers natural antioxidants, polyphenols, and aroma-active compounds using water as a green solvent. The resulting instant tea represents a high-value, shelf-stable product that supports waste reduction, resource recovery, and circular bioeconomy development. Our findings demonstrate how a low-value waste stream can be transformed into a functional food ingredient through mild, energy-efficient treatments that preserve bioactivity and ensure food safety. The approach advances sustainable food technology by reducing reliance on synthetic additives, lowering environmental burden, and promoting full utilization of coffee-processing byproducts in producing countries. |
1. Introduction
Sustainable utilization of agro-industrial byproducts has become an essential strategy in advancing the circular bioeconomy and green food systems.1,2 Robusta coffee (Coffea canephora), widely cultivated in major producing countries such as Brazil, Vietnam, and Indonesia, generates substantial amounts of solid residues during post-harvest processing.3,4 Among these residues, coffee husk is a major byproduct, accounting for approximately 40–45% of the coffee fruit mass, particularly in dry processing systems commonly applied to Robusta coffee.5,6 This lignocellulosic-rich waste is often underutilized and disposed of through open burning or uncontrolled decomposition, leading to greenhouse gas emissions and environmental pollution.7 Therefore, converting Robusta coffee husk (RCH) into value-added food or beverage-related materials represents an environmentally responsible and economically viable approach to reducing agro-industrial waste while creating functional resources for sustainable food innovation.Chemically, the coffee husk contains diverse phytochemicals, including carbohydrates, proteins, lipids, minerals, and abundant secondary metabolites.6,8,9 The husk retains notable levels of chlorogenic acid derivatives, caffeine, tannins, and phenolic acids, in addition to polysaccharides such as cellulose, hemicellulose, and pectin. These constituents confer antioxidant, antimicrobial, and flavor-forming potential, indicating that coffee husk is not merely an agricultural byproduct but also an important reservoir of functional compounds that can be valorized as a low-cost raw material for applications in food and functional products.10,11
Polyphenols from coffee's byproducts have attracted growing attention for their biological activities, including free-radical scavenging, anti-inflammatory, and anti-diabetic effects.12 Several studies have demonstrated that phenolic-rich extracts from RCH can inhibit lipid oxidation and reactive oxygen species, suggesting their potential as natural antioxidants in food preservation and nutraceutical formulations.13,14 Moreover, the recovery of these bioactives from RCH supports the development of natural colorants, flavor enhancers, and functional beverage bases within the food industry.
In recent years, coffee husk has been successfully utilized in various food-related applications. For instance, incorporation of coffee husk extract has improved oxidative stability in meat products and edible oils, functioning as a natural preservative.13,15 It has also been explored as a source of dietary fiber and functional ingredients in bakery formulations, marmalade and cereal bars, enhancing nutritional quality while reducing synthetic additives.16,17 Moreover, pectin and polyphenol fractions from RCH have been employed to develop biodegradable films, fruit coatings, and functional beverages, supporting both food safety and environmental sustainability.18,19
The roasting process plays a decisive role in developing flavor, aroma, and antioxidant properties in coffee-derived materials.20,21 Controlled roasting promotes Maillard and caramelization reactions that generate melanoidins and volatile aromatic compounds while modifying the polyphenolic profile and enhancing sensory acceptability.22,23 However, studies involving roasted coffee husk have so far largely focused on its identification as an adulterant in roasted coffee, emphasizing analytical discrimination and authentication rather than beverage-oriented applications.24 Consequently, in contrast to the extensive body of research on roasted coffee beans and the growing interest in cascara,25 the role of roasting in shaping the chemical composition, sensory attributes, and functional properties of coffee husk when used specifically as a tea material remains insufficiently explored. In particular, systematic investigations into the physicochemical evolution of roasted RCH and how these changes govern aroma development, sensory perception, and bioactivity in coffee husk tea are still lacking.
Moreover, although roasted coffee husk can be further processed via aqueous extraction and drying to produce instant tea, offering a convenient and shelf-stable beverage format, the underlying structure-process-function relationships of roasted RCH tea have yet to be elucidated. In this context, roasting serves as a key processing step that induces substantial physicochemical changes in the original husk material, including alterations in cellular structure, chemical composition, and volatile organic constituents. These structural modifications directly influence the composition of water-soluble compounds released during aqueous extraction, thereby governing the flavor profile, sensory quality, and potential bioactivity of the resulting RCH tea.
In this study, waste RCH was valorized into a functional instant coffee husk tea through an integrated process combining optimized roasting, and aqueous extraction. The specific objective of this work was to systematically evaluate the effect of roasting degree on the physicochemical properties, flavor-related chemical composition, antioxidant activity, and sensory attributes of RCH tea. In particular, the study aimed to elucidate how roasting-induced structural and compositional changes in RCH influence the release of water-soluble compounds during extraction and subsequently govern the quality and functionality of the resulting tea. In addition, response surface methodology was employed to optimize processing conditions in order to maximize antioxidant activity (AA) and diffusion-kinetic modeling for phenolic release of RCH tea was evaluated via Fick's second law. Through this structure-process-function framework, the present work seeks to provide a scientific basis for the rational development of roasted RCH instant tea as a sustainable, value-added beverage derived from coffee-processing byproducts.
2. Materials and methods
2.1. Materials
Dried RCH was obtained as a byproduct from the dry processing of coffee beans at a coffee farm in Thanh My, Lam Dong Province, Vietnam, during the January 2023 harvest season. The material fragments (2–5 mm) comprising the outer skin, dehydrated mucilage layer, and parchment, were collected after hulling. Samples were stored in sealed polyethylene zip bags at ambient temperature in a well-ventilated and dry environment. Prior to use, the RCH was tested for Ochratoxin A contamination according to TCVN 7975:2008, and results confirmed the absence of this mycotoxin.Additional food-grade ingredients used in formulation included Lakanto® sweetener (Saraya Greentek Co., Ltd, Ho Chi Minh City, Vietnam), citric acid (China), and maltodextrin (China, 5–6% moisture content, DE 15–20). All analytical-grade chemicals were sourced from commercial suppliers and used without further purification.
Folin–Ciocalteu reagent (Germany) and sodium carbonate (Na2CO3, 99%, China) were employed for the determination of total polyphenol content, with gallic acid (99%, China) as the calibration standard.
AA assays utilized 2,2-diphenyl-1-picrylhydrazyl (DPPH, Germany) and Trolox (Switzerland) as the reference standard, in combination with sodium acetate trihydrate (C2H3NaO2·3H2O, China), acetic acid (99%, China), 2,4,6-tripyridyl-s-triazine (TPTZ, Germany), and ferric chloride hexahydrate (99%, China), potassium sodium tartrate tetrahydrate (99%, China) and sodium hydroxide (98%, China).
Tannin content was determined using concentrated sulfuric acid (98%, China), potassium permanganate (99%, China), and indigo carmine (99%, China) as the titration indicator.
2.2. Chemical composition of Robusta coffee husk
The chemical composition of the dried RCH was determined following standard analytical protocols.26 For the determination of reducing sugar and total organic acid contents, the RCH was extracted in hot water at 75 °C for 15 min using a solid-to-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 (w/v), and the extract volume was adjusted to 100 mL prior to the analysis.
20 (w/v), and the extract volume was adjusted to 100 mL prior to the analysis.
Total polyphenol content (TPC) and AA were assessed from ethanolic extracts obtained by exhaustive maceration with 90% ethanol, while aqueous extracts prepared at the same solid-to-solvent ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, w/v) were also analyzed for comparison. The parameters evaluated included total organic acids, total ash, total polyphenol content, antioxidant activity, moisture content, protein content, lipid content, and reducing sugar content.
20, w/v) were also analyzed for comparison. The parameters evaluated included total organic acids, total ash, total polyphenol content, antioxidant activity, moisture content, protein content, lipid content, and reducing sugar content.
2.3. Optimization of roasting conditions
The RCH roasting conditions were optimized using response surface methodology (RSM) with a central composite design (CCD) to identify the combination of parameters that maximized the targeted quality attributes. Two independent variables were selected based on preliminary trials: roasting temperature (X1, °C) and roasting time (X2, s).27 The dependent variables (responses) included TPC (Y1), AA (Y2), reducing sugar content (Y3), total organic acid content (Y4), and tannin content (Y5).The experimental domain for each factor was set according to five coded levels (−α, −1, 0, +1, +α), with 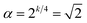 for k = 2 factors. The CCD comprised 13 experimental runs, including five replicates at the center point to provide a reliable estimate of pure experimental error and to enable robust lack-of-fit testing and model adequacy assessment, as commonly recommended in response surface methodology designs.28
for k = 2 factors. The CCD comprised 13 experimental runs, including five replicates at the center point to provide a reliable estimate of pure experimental error and to enable robust lack-of-fit testing and model adequacy assessment, as commonly recommended in response surface methodology designs.28
In the CCD, the roasting temperature was varied over an approximate range of 100 to 180 °C, while the roasting time ranged from approximately 150 to 270 s, as defined based on preliminary experiments (Table S1). The experimental matrix is presented in Table S2.
Analysis of variance (ANOVA) was performed to evaluate model significance, coefficient of determination (R2, adjusted R2, predicted R2), and lack-of-fit. Residual analysis was used to verify the model assumptions of normality and homoscedasticity. Optimization was conducted using a desirability function approach to maximize Y1 and Y2 while controlling Y3, Y4, and Y5 within target ranges suitable for functional and sensory quality. All statistical analyses were performed using Design-Expert software (version 13.0.5.0, Stat-Ease Inc., Minneapolis, USA).
2.4. Optimization of aqueous extraction conditions
The RCH roasted under optimal conditions was used to evaluate extraction. The aqueous extraction process of RCH was further optimized using RSM.29,30 A Box–Behnken design was employed, in which each factor was tested at three coded levels: +1, 0, and −1, corresponding to high, central, and low values, respectively. The factor ranges and coded levels are summarized in Table S3, and the experimental matrix is presented in Table S4.The dependent variables were TPC (Y1), AA (Y2), reducing sugar content (Y3), total organic acid content (Y4), and tannin content (Y5). The independent variables (factors) included solvent-to-husk ratio (X1), extraction temperature (X2), and extraction time (X3). For k = 3 factors, the design required n = 17 runs, including five replicates at the center point, with α = 1. The solvent-to-husk ratio, extraction temperature, and extraction time were investigated over approximate ranges of 10 to 50 (mL g−1), 50 to 90 °C, and 10 to 70 min, respectively (Table S3).
Experimental data were modeled using a second-order polynomial equation, and analysis of variance (ANOVA) was conducted to determine the significance of each factor and their interactions. Optimization was performed using a desirability function to maximize Y1 and Y2 while maintaining Y3, Y4, and Y5 within acceptable functional and sensory limits.
For physicochemical properties of extract, the roasted RCH was extracted at optimal conditions and then frozen at −80 °C for 24 h, followed by freeze-drying at −80 °C for 30 h.
2.5. Physicochemical characterizations
Surface morphology of RCH and particle size of instant coffee husk tea was observed using an optical microscope (Dino-Lite, Taiwan) equipped with the DinoCapture software for image acquisition and surface measurement.Detailed microstructural features were further examined by field-emission scanning electron microscopy (FE-SEM, Hitachi S-4800, Tokyo, Japan). The samples were sputter-coated with a thin layer of gold to enhance conductivity, and images were recorded at an accelerating voltage of 10.0 kV under various magnifications to characterize the surface texture and structural integrity of the raw RCH, roasted RCH and extracted RCH.
Fourier-transform infrared spectroscopy (FTIR) analysis was performed using a Bruker Tensor 27 spectrometer (Bruker Optics, Germany) to identify and characterize the functional groups present in the coffee husk samples. Spectra were recorded in the range of 4000–450 cm−1 with a resolution of 4 cm−1.
2.6. Analysis of volatile organic compounds by headspace GC-MS
The volatile organic compounds (VOCs) released from the RCH samples were analyzed using a gas chromatography-mass spectrometry system (GC-MS QP2020, Shimadzu, Japan) equipped with a static headspace autosampler (HS-20, Shimadzu, Japan) with NIST14 s library version. Approximately 1.00 g of raw or roasted RCH was accurately weighed into a 20 mL headspace vial and hermetically sealed with a PTFE/silicone septum cap. During analysis, the vials were pre-incubated in the autosampler oven at 120 °C for 20 min to allow VOCs to equilibrate in the headspace. The equilibrated vapor phase was automatically injected into the GC inlet under an isothermal transfer line temperature of 150 °C. Chromatographic separation was achieved on an Rxi-5MS capillary column (30 m × 0.25 mm × 0.25 µm; Restek, USA) using helium as the carrier gas at a constant flow rate of 1.88 mL min−1 and a split ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10.
10.
The oven temperature was programmed as follows: initially 30 °C (1 min hold), ramped to 50 °C at 5 °C min−1, then to 180 °C at 10 °C min−1, 280 °C at 15 °C min−1, and finally to 300 °C at 20 °C min−1 (held for 5 min). The separated analytes were ionized in the mass spectrometer at an ion-source temperature of 250 °C, and mass spectra were recorded in electron-impact (EI) mode for compound identification. Compound identification was performed by comparing mass spectra with library data, and only matches with a similarity index (SI, match factor) of ≥ 90% were accepted.
2.7. Formulation ratio optimization for instant coffee husk tea
After obtaining the tea extract from coffee husk, the extract was blended with Lakanto sweetener at concentrations of 7%, 8%, 9%, and 10%, maltodextrin at 10%, 15%, 20%, and 25%, and citric acid at 0.010%, 0.015%, 0.020%, and 0.025%. The blended mixtures were then frozen at −80 °C for 24 h, followed by freeze-drying at −80 °C for 30 h. The resulting instant coffee husk tea powders were subsequently evaluated for sensory quality using a hedonic scoring method. The sensory evaluation was intentionally designed as a formulation-oriented tool to optimize the proportion of ingredients in the instant coffee husk tea. Accordingly, the sensory assessment was limited to a qualitative description of the reconstituted roasted cascara beverage.2.8. Sensory evaluation
The sensory quality of the instant coffee husk tea was evaluated following standard TCVN 3215-79 using a hedonic scoring method.31 In brief, twenty trained panelists (10 females and 10 males; aged 30–45 years) assessed three attributes – color, aroma – taste, and appearance – based on a 5-point scale, where higher scores indicated better sensory quality. Each attribute was assigned a weighting factor: 1.4 for color, 1.4 for aroma–taste, and 1.2 for appearance. Between samples, panelists rinsed their mouths with room temperature drinking water to cleanse the palate and a 3–5 min rest interval was provided between consecutive evaluations to minimize carry-over effects and sensory fatigue. The evaluation standard of sensory was summarized in Table S14. All sensory data were analyzed using one-way analysis of variance (ANOVA). When significant differences were detected (p < 0.05), Duncan's multiple range test was applied as a post hoc procedure to compare mean values.2.9. Release kinetic model of TPC
To investigate the diffusion kinetics of bioactive compounds from instant RCH tea, the release of TPC was modeled according to Fick's second law of diffusion.32 This law describes the temporal change in solute concentration within a diffusing aqueous medium. In this study, the release behavior was examined at different temperatures to evaluate the temperature dependence of the diffusion process. Briefly, instant tea powder (0.5 g) was placed in a dialysis membrane (molecular weight cut-off: 3.5 kDa) and immersed in 120 mL of distilled water. The dialysis was carried out at different temperatures (35, 40, 45, 50, and 55 °C) to monitor the extraction of TPC over time. At 30 minute intervals, aliquots of the dialysate were collected for analysis. The total extraction period lasted for 6 hours.The general form of Fick's second law is expressed as:
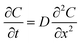 | (1) |
For spherical systems such as instant tea granules, the analytical solution of Fick's second law can be simplified and written in normalized form as eqn (2):33,34
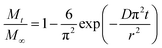 | (2) |
Taking the natural logarithm of both sides of the exponential term in eqn (2), the diffusion equation can be linearized as eqn (3):
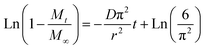 | (3) |
This linearized form allows the estimation of the effective diffusion coefficient (D) from the slope of the regression line obtained by plotting ln (1−Mt M−1∞) versus time (t). This approach provides a quantitative method for describing diffusion-controlled release behavior in instant coffee husk tea powder (with r is estimated 50.0 ± 10.3 µm).
2.10. Data analysis
All experiments were conducted in triplicate, and results were expressed as mean ± standard deviation. Statistical significance was evaluated using one-way analysis of variance (ANOVA), and differences between means were determined using Duncan's multiple range test at a significance level of p < 0.05, performed with Statgraphics Centurion XV software. Duncan's test was selected due to its widespread application in sensory and food science studies, as it provides adequate sensitivity for detecting differences among the changes.3. Results and discussion
The overall research strategy for production of instant coffee husk tea is summarized in Fig. 1. Raw RCH obtained from a Vietnamese coffee farm was characterized for its proximate composition (moisture, protein, lipids, ash, sugars, organic acids) and antioxidant-related parameters (TPC and DPPH activity) to confirm its potential as a functional raw material as shown in Table S5. This provided the baseline for selecting suitable processing windows for roasting and extraction. Roasting conditions were optimized using RSM with a central composite design, where roasting temperature and time were varied to simultaneously modulate total polyphenols, antioxidant activity, reducing sugars, total organic acids, and tannins.35 The RSM design, the selected response variables were chosen to reflect the key determinants of instant coffee husk tea quality and sensory perception. TPC and AA represent the functional value of the beverage, while reducing sugars are closely related to Maillard reaction pathways and aroma development during roasting. Total organic acids contribute to flavor freshness and balance, whereas tannins govern astringency, mouthfeel, and overall sensory acceptance. By simultaneously optimizing these parameters, the RSM approach ensures a balanced improvement of both functional performance and consumer-relevant sensory quality. | ||
| Fig. 1 Overall experimental strategy for valorizing Robusta coffee husk into functional instant coffee husk tea. | ||
The optimal condition was then used to produce roasted RCH for all subsequent experiments. The aqueous extraction of the roasted RCH was optimized using a Box–Behnken design, with solvent-to-husk ratio, extraction temperature, and extraction time as factors.36 This stage aimed to maximize the recovery of phenolic compounds and antioxidant capacity while keeping sugars, acids, and tannins within desirable ranges for beverage quality. The optimized extract was then freeze-dried to obtain a concentrated powder.
To clarify the analytical strategy, different extraction solvents and conditions were deliberately employed to address distinct scientific objectives relevant to RCH tea development. Ethanolic extraction was used to obtain an exhaustive recovery of phenolic compounds, thereby evaluating the maximum polyphenol content and antioxidant potential of RCH. In contrast, aqueous and hot-water extractions were designed to simulate realistic tea-brewing conditions and to characterize the water-soluble components that directly contribute to beverage flavor, sensory perception, and consumer-relevant quality. The dialysis-based extraction approach was further applied to model the release kinetics of polyphenols from instant coffee husk tea under controlled conditions, providing mechanistic insight into diffusion-driven bioactive release rather than compositional extraction alone.
In addition, physicochemical characterization was carried out at each key step (raw, roasted, and extracted RCH) using optical microscopy and SEM to follow morphological changes, FTIR to monitor structural and functional-group transformations, and headspace GC-MS to track the evolution of VOCs responsible for aroma development. These analyses clarified how roasting and extraction alter the lignocellulosic matrix and flavor-active components. Finally, the instant coffee husk teaformulation and functionality were established by blending the optimized extract with maltodextrin, Lakanto sweetener, and citric acid, followed by sensory evaluation to select the most acceptable recipe. The diffusion kinetics of phenolic compounds from the instant tea powder in water were then modeled using Fick's second law to describe diffusion-controlled dissolution behavior at different temperatures.
3.1. Roasting process of robusta coffee husk
In this study, the optimization of RCH roasting was systematically evaluated using response surface methodology. The results demonstrated that roasting temperature and time exerted significant and synergistic effects on total polyphenols, antioxidant activity, reducing sugars, organic acids, and tannins, enabling the identification of an optimal roasting condition at 117 °C for 225 s, which balances functional quality and chemical characteristics. Detailed experimental data, model development, statistical validation, and response surface analyses are provided in the SI (Tables S6–S8 and Fig. S1).To investigate the aroma development and chemical transformation during roasting, the RCH sample obtained under the optimized roasting conditions was subjected to headspace GC-MS analysis, with the raw RCH serving as the control. The headspace GC-MS results revealed marked qualitative and quantitative variations in the VOCs between raw and roasted samples (Table 1). In total, forty VOCs were identified and categorized into six major functional groups and four chemical classes. The compositional shifts clearly indicated that roasting profoundly altered the volatile profile of RCH, primarily driven by Maillard reactions, caramelization, and the thermal degradation of carbohydrates, proteins, and lipids – processes collectively responsible for the formation of the characteristic aroma and flavor complexity of roasted coffee husk.
| No. | RT | Name | Raw RCH (%) | Roasted RCH (%) |
|---|---|---|---|---|
| 1 | 1.117 | Trans-β-ionone-5,6-epoxyde | 49.55 | 44.08 |
| 2 | 1.141 | L-alanine ethylamide![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
33.81 | — |
| 3 | 1.144 | Tetranitromethane | — | 23.85 |
| 4 | 1.181 | Acetaldehyde | 7.67 | 4.72 |
| 5 | 1.25 | Ethanol | 0.69 | 0.46 |
| 6 | 1.33 | Propanesulfonylacetonitrile | 3.66 | — |
| 7 | 1.333 | Isopropenyl acetate | — | 4.41 |
| 8 | 1.422 | Methyl acetate | — | 2.04 |
| 9 | 1.522 | Isobutanal | 0.45 | 0.38 |
| 10 | 1.679 | 3-Methyl-2-butanone | — | 1.58 |
| 11 | 1.786 | Ammonium acetate | — | 0.89 |
| 12 | 2.089 | Bilorin | — | 0.49 |
| 13 | 2.144 | 3-Methylbutanal | 0.57 | 0.8 |
| 14 | 2.223 | 2-Methylbutanal | 0.4 | 1.32 |
| 15 | 2.409 | Acetic acid | 1.12 | 8.05 |
| 16 | 2.56 | 2-Methyl-3-pentanone | — | 0.46 |
| 17 | 2.57 | n-Pentanal | — | 0.22 |
| 18 | 2.805 | Acetol | — | 0.58 |
| 19 | 3.111 | N-methylpyrrole | — | 0.28 |
| 20 | 3.335 | Pyrrole | — | 0.16 |
| 21 | 3.601 | Toluene | — | 0.11 |
| 22 | 3.972 | 3-Hexanone | — | 0.13 |
| 23 | 4.22 | Mesityl oxide | — | 0.09 |
| 24 | 4.276 | n-hexanal | 0.56 | 0.62 |
| 25 | 4.913 | Furfural | — | 1.82 |
| 26 | 6.17 | Heptan-2-one | — | 0.29 |
| 27 | 6.43 | Heptanal | — | 0.12 |
| 28 | 7.647 | Phenylmethanal | — | 0.31 |
| 29 | 7.888 | Methyl 2-furoate | — | 0.12 |
| 30 | 8.223 | 2-Pentylfuran | 0.09 | 0.22 |
| 31 | 8.466 | N-methyl-2-formylpyrrole | — | 0.22 |
| 32 | 8.665 | 2-Formylpyrrole | — | 0.47 |
| 33 | 8.958 | 2-Ethylhexanol | 0.11 | — |
| 34 | 9.234 | Benzeneacetaldehyde | 0.18 | 0.07 |
| 35 | 10.329 | n-nonanal | 0.15 | 0.17 |
| 36 | 12.031 | n-decanal | — | 0.1 |
| 37 | 15.484 | γ-Muurolene | 0.21 | — |
| 38 | 18.371 | Pogostol | — | 0.15 |
| 39 | 20.137 | Caffeine | 0.29 | 0.16 |
| 40 | 23.911 | Butyl stearate | 0.19 | — |
| Unknown compounds | 0.1 | 0.28 | ||
| Total | 100 | 100 | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Category | ||||
| Terpenoid | 49.55 | 44.08 | ||
| Sesquiterpene | 0.21 | 0.64 | ||
| Aromatic compounds | 0.56 | 4.96 | ||
| Others | 49.68 | 50.32 | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Functional group | ||||
| Hydrocarbon | 49.8 | 44.7 | ||
| Alcohol | 0.8 | 0.6 | ||
| Aldehyde | 10.0 | 9.9 | ||
| Acid | 1.1 | 8.1 | ||
| Amine & N-compounds | 37.8 | 26.0 | ||
| Ester | 0.2 | 6.6 | ||
| Others | 0.4 | 4.2 | ||
In the raw RCH, the headspace was dominated by terpenoid hydrocarbons, mainly trans-β-ionone-5,6-epoxyde (49.55%), nearly half of the total VOC content. This compound, derived from carotenoid oxidation, contributes to floral and fruity aroma notes and is commonly reported in unroasted coffee and plant materials.37 Minor components such as L-alanine ethylamide (33.81%) and propanesulfonylacetonitrile (3.66%) indicated the presence of nitrogen-containing precursors associated with amino acid and sulfur metabolism. The relatively high proportion of amine and amide compounds (37.8%) in the unroasted sample supports this observation, suggesting a strong contribution from proteinaceous residues inherent to the coffee husk structure.
After roasting, the volatile composition became markedly diversified, with the total number of detectable compounds increasing from 17 to 39. The terpenoid fraction decreased from 49.55% to 44.08%, while the aromatic and oxygenated compounds (notably aldehydes, acids, and esters) increased substantially. Notably, tetranitromethane emerged as a dominant component in the roasted sample (23.85%), whereas L-alanine ethylamide, which was abundant in the raw material, was no longer detected after roasting. This pronounced compositional shift indicates that thermal treatment induced extensive chemical transformations, likely associated with Maillard and caramelization reactions.
The formation of furfural (1.82%), acetic acid (8.05%), methyl acetate (2.04%), and mesityl oxide (0.09%) evidenced extensive carbohydrate dehydration and esterification reactions, which are characteristic of the roasting process.38 The sharp rise in acidic compounds (from 1.1% to 8.1%) is consistent with the oxidative cleavage of polysaccharide side chains and the decomposition of amino acids, producing volatile carboxylic acids that impart sour or pungent notes.39,40
Simultaneously, the amine and nitrogenous fraction declined significantly, from 37.8% to 26.0%. This reduction is likely attributed to the thermal decomposition of amino precursors through Strecker degradation and subsequent formation of heterocyclic products such as pyrrole, N-methylpyrrole, and 2-formylpyrrole.41 These compounds, newly generated after roasting, are contributed to roasted, nutty, and caramel-like aromas. The emergence of furfural, methyl 2-furoate, and 2-pentylfuran further confirms the transformation of pentose and hexose sugars through pyrolytic fragmentation and cyclization.42 Moreover, the increase in ester content (from 0.2% to 6.6%) reflects secondary esterification between carboxylic acids and alcohols, enhancing the fruity or sweet aroma complexity of roasted RCH.43
The functional-group distribution revealed that hydrocarbons remained predominant (44.7%), but oxygenated volatiles, including acids, esters, and aldehydes, collectively accounted for more than one-third of the roasted VOCs. These shifts indicate progressive oxidation and condensation of unsaturated compounds, leading to more complex aroma-active profiles. The detection of toluene, benzene acetaldehyde, and phenylmethanal points to enhanced aromaticity derived from the degradation of phenolic and lignin constituents during roasting. Therefore, the roasting process induced a distinct transformation from a nitrogen-rich, terpene-dominated volatile profile in raw RCH to an oxygenated, aromatic, and ester-enriched profile in roasted RCH.
Although the relative percentages of VOCs obtained from HS-GC-MS do not directly reflect the sensory intensity or individual aroma contribution of each compound, the pronounced increase in aldehyde-derived compounds (2-methylbutanal, 3-methylbutanal, and phenylacetaldehyde) as well as furfural-related compounds (furfural and 2-pentylfuran) after roasting suggests a plausible association with the aroma attributes perceived during the sensory evaluation of coffee husk.44–46 These results indicated that roasted RCH possesses notable aroma-forming potential and could be developed as a tea product with distinctive flavor characteristics.
3.2. The extraction of roasted coffee husk
The aqueous extraction of roasted RCH was optimized using a Box–Behnken design within the framework of response surface methodology. The results revealed that the solvent-to-husk ratio, extraction temperature, and extraction time exerted significant and synergistic effects on total polyphenols, antioxidant activity, reducing sugars, organic acids, and tannins, allowing the identification of an optimal extraction condition at a solvent-to-husk ratio of 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, extraction temperature of 70 °C, and extraction time of 45 min. Under these conditions, high phenolic recovery and antioxidant capacity were achieved while minimizing thermal degradation. Detailed statistical validation, response surface analyses, and experimental verification are presented in the SI (Tables S10–S12 and Fig. S2–S6).
1, extraction temperature of 70 °C, and extraction time of 45 min. Under these conditions, high phenolic recovery and antioxidant capacity were achieved while minimizing thermal degradation. Detailed statistical validation, response surface analyses, and experimental verification are presented in the SI (Tables S10–S12 and Fig. S2–S6).
Optical microscopy and SEM analyses revealed significant morphological changes of the RCH during the successive processing stages of roasting and aqueous extraction (Fig. 2). The surface of the raw RCH exhibited an intact and compact fibrous matrix typical of lignocellulosic materials.47 The microscopy images showed that the structure appeared rigid, with visible vein-like ridges and an overall glossy brownish-yellow hue characteristic of dried parchment layers. SEM micrographs confirmed a relatively dense microstructure composed of tightly packed fibrils and smooth lamellar surfaces. The fibril bundles were coherent, and no pores or cracks were visible at the sub-micron scale, suggesting that the natural lignin coating effectively cemented polysaccharide domains.
After roasting, distinct changes in color, texture, and microstructure were observed. The optical images (Fig. 3B1 and 3B2) showed a darker, caramelized surface with slight shrinkage and warping, reflecting the progression of Maillard and pyrolytic reactions.22 SEM images (Fig. 3B3 and 3B4) displayed a partially collapsed matrix with the formation of microcracks and irregular voids. The surface roughness increased markedly, and local melting or carbonization regions were discernible. The presence of small globular deposits and amorphous fragments on the surface likely represents polymerized sugars or condensed phenolic residues generated during roasting. Such morphological loosening enhances the porosity and specific surface area of RCH, thereby facilitating subsequent solvent penetration during extraction.
Following aqueous extraction, the post-extraction RCH samples exhibited pronounced structural degradation of the cell-wall surface. Optical micrographs (Fig. 3C1 and C2) revealed a duller brown, more porous appearance, indicating the migration and leaching of water-soluble constituents such as polysaccharides and phenolic compounds under thermal extraction conditions. At the microscale (Fig. 3C3 and C4), SEM images confirmed a distinctly disrupted and highly porous morphology characterized by fiber delamination and exfoliation of the cell-wall layers. The fibrous network became loose with ruptured lamellae, reflecting the hydrolytic cleavage of hydrogen-bonded cellulose–hemicellulose linkages. Several granular residues adhering to the surface were likely insoluble lignin-rich fragments. These morphological transformations demonstrate that aqueous extraction effectively removed soluble matter and partially defibrillated the cell-wall matrix, thereby enhancing its accessibility for subsequent valorization processes.
The sequential morphological evolution, from compact fibrillar integrity to thermally fractured and finally to hydrolyzed porous structures, illustrates the progressive breakdown of the lignocellulosic matrix. Roasting acts as a physicochemical pretreatment that weakens structural bonds through dehydration and lignin softening, whereas hot-water extraction promotes delamination and removal of soluble and low-molecular-weight fractions.48,49 These changes collectively enhance the reactivity and functional potential of coffee-husk residues for downstream applications (e.g., extraction of pectins or phenolics, biosorbent preparation, or biopolymer reinforcement).
Accordingly, SEM analysis revealed that the roasting process not only facilitates the formation of volatile aromatic compounds responsible for the characteristic flavor of coffee husk tea products but also serves as an essential physicochemical pretreatment. This pretreatment promotes cell-wall delamination and facilitates the removal of soluble constituents and low-molecular-weight fractions, ultimately improving extraction efficiency and the accessibility of bioactive compounds.
The FTIR spectra of raw, roasted, and post-extraction RCH samples (Fig. 3) revealed distinct structural transformations induced by roasting and subsequent aqueous extraction. In the raw RCH, the broad band near 3316 cm−1 corresponds to O–H stretching vibrations of polysaccharides and phenolic hydroxyls, while the peaks at 2921 and 2856 cm−1 are attributed to aliphatic C–H stretching. The strong absorption at 1742 cm−1, assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of esterified carboxyl groups in hemicellulose and pectin, together with the dominant band at ∼1025 cm−1 (C–O–C and C–O stretching), reflects the hydrophilic, pectin- and hemicellulose-rich nature of the raw lignocellulosic matrix.
O stretching of esterified carboxyl groups in hemicellulose and pectin, together with the dominant band at ∼1025 cm−1 (C–O–C and C–O stretching), reflects the hydrophilic, pectin- and hemicellulose-rich nature of the raw lignocellulosic matrix.
Roasting markedly altered these spectral features. The attenuation of the O–H band indicates dehydration and disruption of hydrogen bonding, while the reduction of the 1742 cm−1 peak signifies de-esterification and cleavage of acetyl and carboxyl ester groups.50 Enhanced bands at 1629 and 1537 cm−1 represent the formation of aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and conjugated structures. Minor shifts and intensity changes in the fingerprint region (1400–1000 cm−1) further confirm carbohydrate depolymerization and matrix rearrangement during roasting.
C and conjugated structures. Minor shifts and intensity changes in the fingerprint region (1400–1000 cm−1) further confirm carbohydrate depolymerization and matrix rearrangement during roasting.
After hot-water extraction, the FTIR profile was dominated by features of the lignocellulosic backbone. The O–H stretching band persisted, mainly due to adsorbed moisture, whereas the carbonyl peak around 1740 cm−1 was substantially diminished, indicating removal of soluble esterified phenolics and organic acids. The overall decrease in aromatic and carbonyl intensities relative to the roasted sample suggests that low-molecular-weight phenolic and Maillard products were effectively extracted. In contrast, the C–O–C/C–O region (1160–1020 cm−1) remained prominent, confirming that cellulose, hemicellulose, and lignin constitute the residual solid. These results demonstrate that roasting transformed the hydrophilic matrix of RCH into a more aromatic and conjugated structure, while aqueous extraction selectively removed soluble bioactive and thermally generated compounds, leaving behind a condensed, fiber-rich residue.
The physicochemical properties of roasted RCH extract under the optimal conditions are described in Table S13. The reducing sugar content was determined to be 7.13 ± 0.19% g/100 g dw. The total organic acid content (0.85 ± 0.02 g/100 g dw) may be associated with the partial thermal degradation of chlorogenic acids and related phenolic compounds, as commonly reported for roasted coffee byproducts, leading to the formation of volatile and non-volatile acidic compounds that enhance flavor balance.51,52
The water-soluble extract content reached 36.58 ± 2.60 g/100 g dw, exceeding the minimum requirement of 32 g/100 g dw specified in TCVN 5610:2007, confirming good extractability and beverage quality. The TPC of the roasted RCH infusion was 40.31 ± 0.66 mg GAE per g dry weight from water extraction, retaining approximately 83% of the phenolic compounds compared with unroasted RCH, while the AA (518.24 ± 4.83 µmol TE per g dry weight) corresponded to 96% of the initial RCH. The slight decrease reflects the partial thermal degradation of complex polyphenols during roasting and extraction. Meanwhile, tannin content increased to 4.47 ± 0.11 mg g−1 dw−1, likely due to enhanced cell-wall permeability and tannin release during roasting. Caffeine content (9.2 g/100 g dw) was within the range reported for coffee husk (5.4–18 g/100 g dw) and much lower than in aqueous extract of coffee beans (11 – 26 g/100 g dw).53,54
Microbiological evaluation was performed on the RCH extract in accordance with TCVN 7975:2008 and QCVN 8-1:2011/BYT to assess its microbiological safety (Table S13). The results showed that the extract complied with the national safety criteria, with no detectable mold, yeast, or Ochratoxin A (OTA). The absence of OTA is particularly relevant from a food safety perspective, as coffee husk–based materials are commonly associated with OTA contamination during post-harvest handling and storage. The total aerobic microbial count was 1.0 × 105 CFU per g, which is well below the permissible limit of 1.0 × 106 CFU per g. These findings suggest that the combined effects of roasting and the low moisture content of the extract effectively suppressed microbial growth, supporting the safe use of roasted RCH extract as a functional ingredient for instant coffee husk tea formulations.
3.3. Formulation of instant tea from Robusta coffee husk
A sensory optimization study was conducted to identify the most suitable proportions of maltodextrin (carrier), Lakanto (sweetener), and citric acid (acidulant) for the instant coffee husk tea. A five-point hedonic scale was used with 20 untrained panelists to evaluate color, flavor, aroma, and overall acceptability (Fig. 4). All samples were reconstituted in 100 g of hot water (85–90 °C), and results were statistically analyzed at a 95% confidence level (p < 0.05). | ||
| Fig. 4 Effect of maltodextrin concentration (A), Lakanto (B) and citric acid (C) on sensory scores. Different letters indicate significant differences (p < 0.05). Data represent mean ± SD (n = 3). | ||
Increasing the maltodextrin concentration from 10% to 25% improved the recovery yield due to its carrier and drying-aid effects, which reduced stickiness and enhanced powder formation (Fig. 4A). However, formulations containing ≥ 20% maltodextrin showed significantly lower color scores (p < 0.05), reflecting a dilution of the characteristic reddish-brown hue. Based on yield and sensory performance, 15% maltodextrin was selected as the optimal level.
For sweetness optimization, Lakanto concentrations of 7–10% were evaluated while maintaining 15% maltodextrin and 0.015% citric acid. As shown in Fig. 4B, the formulation containing 8% Lakanto exhibited a significantly higher aroma–taste score (4.2/5) compared with the other concentrations (p < 0.05), indicating an optimal balance between sweetness and acidity. Lower concentrations produced overly acidic taste, whereas higher levels masked the natural sour note with excessive sweetness.
Citric acid concentrations from 0.01% to 0.025% were tested under constant 15% maltodextrin and 8% Lakanto. Fig. 4C demonstrates that the addition of 0.015% citric acid produced the highest overall acceptability score (4.35/5), which was significantly different from both lower and higher acid levels (p < 0.05). The final optimized formulation including 15% maltodextrin, 8% Lakanto, and 0.015% citric acid produced a free-flowing, soluble powder with high reconstitution ability, appealing reddish-brown color, and well-balanced sweet–acidic flavor suitable for instant coffee husk tea production.
The data of sensory evaluation for instant RCH tea formed from optimal formulation are shown in Table S15. The results found average scores of 4.55 for color, 4.70 for aroma–taste, and 4.85 for appearance. When applying the weighting factors, the corresponding scores were 6.37, 6.58, and 5.82, yielding a total quality score of 18.77. According to the classification criteria, products with an average unweighted score ≥ 4.8 for each attribute are rated “Good,” while those with scores ≥ 3.8 are rated “Fair”. Based on the obtained results, the instant RCH tea sample met the “Good” quality standard for all evaluated attributes, demonstrating its desirable natural color, distinctive aroma–taste, and clear, uniformly dissolved appearance.
The reconstituted tea exhibited a bright reddish-brown color, clear appearance, complete solubility, and a pleasant aroma with a mild sweet–acidic balance and clean aftertaste. Physicochemical analyses of instant RCH tea showed a moisture content of 1.24 ± 0.23%, pH of 3.763 ± 0.018, and total soluble solids of 9.0 ± 0.5 °Bx at 25 °C. The low moisture level ensures microbial stability and extended shelf life, though airtight, vacuum-sealed packaging is recommended to maintain quality. From a consumer perspective, the high sensory scores indicate that the instant RCH tea exhibits harmonious color and flavor, good ease of use, and suitability as a convenient infusion beverage, particularly for caffeine-sensitive individuals due to its relatively low caffeine content. In addition, the instant RCH tea which is rich in bioactive compounds and derived from an agricultural by-product, has practical significance in enhancing raw material valorization and promoting the development of environmentally sustainable products.
The findings demonstrated strong potential for scalability and industrial implementation. The roasting-aqueous extraction-instant tea powder production process can be readily integrated into existing instant tea processing lines. Coupled with the abundant availability of coffee husk and the use of water as a safe and low-cost solvent, these factors support the practical feasibility of producing roasted RCH instant tea at an industrial scale.
3.4. Release kinetics of phenolic compounds from instant tea in water
During direct dissolution of instant coffee husk tea powders, polyphenol release is strongly influenced by rapid particle disintegration, instantaneous solubilization, and concurrent dissolution of the particle matrix, which can obscure diffusion-controlled behavior. Mechanical breakup of hydrated particles and fast dissolution of matrix-forming components, such as dextrins and polyphenols, may result in an immediate burst release that does not accurately represent molecular diffusion processes. To decouple diffusion-driven release from these physical interferences, a dialysis membrane was employed to physically confine the solid matrix while allowing polyphenol molecules to diffuse freely into the surrounding medium. This strategy minimizes the effects of mechanical disintegration and matrix dissolution, thereby enabling a more reliable assessment of intrinsic diffusion-controlled release kinetics and the influence of temperature on bioactive compound mobility.Accordingly, the release of solutes from instant coffee husk tea powder could be described using Fick's second law of diffusion, which provides a mechanistic framework for modeling time-dependent mass transport from a hydrated solid matrix into an aqueous medium.55 This approach is particularly suitable for systems in which release is governed primarily by molecular diffusion rather than chemical reaction or degradation, where concentration gradients act as the principal driving force for mass transfer, and where particle geometry can be reasonably approximated as spherical or quasi-spherical granules.56–58
In the present system, the instant tea powder consists of a maltodextrin-based carrier encapsulating soluble solids derived from RCH extract. Upon reconstitution, the hydrated carbohydrate network forms a diffusion barrier through which phenolic and antioxidant compounds gradually migrate into the aqueous phase. This modeling approach enables quantitative evaluation of temperature-dependent solute mobility and provides mechanistic insight into the physical processes governing bioactive release from instant RCH tea formulations.46,47 Importantly, the diffusion model employed in this study was not intended to simulate consumer brewing practices, but rather to elucidate the physicochemical role of the maltodextrin-based carrier in protecting phenolic compounds within the instant tea matrix. Upon hydration, the carbohydrate network acts as a diffusion barrier that retards polyphenol mobility, analogous to the confinement of phenolic compounds within plant cell walls or other polysaccharide-rich food matrices.59,60 Within the moderate temperature range investigated, the structural integrity of the matrix is preserved, enabling controlled diffusion and limiting the premature release or degradation of bioactive compounds. In contrast, deviations from Fickian diffusion behavior at elevated temperatures reflect partial structural disruption of the carrier system, indicating a reduction in its protective capacity.
As shown in Fig. 5 and Table 2, the slope obtained from the linear regression of ln (1−Mt M−1∞) versus time corresponds to −Dπ2/r2 and therefore represents the apparent diffusion-controlled release rate of phenolic compounds. A more negative slope indicates a faster release rate, reflecting enhanced molecular mobility of phenolics within the maltodextrin-based matrix. In the temperature range of 35–40 °C, the slopes exhibit relatively small magnitudes and excellent linearity (R2 ≈ 0.99), indicating that phenolic release is predominantly governed by Fickian diffusion through an intact carbohydrate network. The corresponding diffusion coefficients (D ≈ 1.73–1.94 × 10−9 cm2 s−1) suggest strong diffusional restriction, consistent with an effective protective role of the matrix.
 | ||
| Fig. 5 Diffusion kinetics of phenolic compound release from instant Robusta coffee husk tea powder at different temperatures (35–55 °C) based on Fick's second law. | ||
| Temperature (°C) | Slope (−Dπ2/r2) | R2 | D (cm2 s−1) |
|---|---|---|---|
| 35 | −6.8362 × 10−4 | 0.9899 | 1.73 × 10−9 |
| 40 | −7.66815 × 10−4 | 0.9925 | 1.94 × 10−9 |
| 45 | −1.23 × 10−3 | 0.9602 | 3.12 × 10−9 |
| 50 | −9.51355 × 10−4 | 0.9274 | 2.41 × 10−9 |
| 55 | −1.26 × 10−3 | 0.8229 | 3.19 × 10−9 |
Increasing the temperature to 45 °C results in steeper slopes accompanied by a marked increase in D (3.12 × 10−9 cm2 s−1), indicating enhanced solute mobility due to increased flexibility of maltodextrin chains and improved solvent penetration. However, at higher temperatures (50–55 °C), although the apparent slopes and D values further increase, the pronounced reduction in R2 (down to 0.8229) indicates that diffusion is no longer the sole governing mechanism. Under these conditions, partial disruption of the maltodextrin matrix is likely to occur, leading to a combined contribution of diffusion and polysaccharide dissolution. Consequently, the D values obtained at elevated temperatures should be considered empirical indicators of accelerated release rather than true Fickian diffusion coefficients. Overall, the temperature-dependent variations in slope and D demonstrated that the release of phenolic compounds from instant RCH tea powder gradually transitions from diffusion-controlled transport at moderate temperatures to matrix-disruption-dominated behavior at higher temperatures, highlighting the critical role of matrix integrity in regulating phenolic retention and stability.
4. Conclusion
Robusta coffee husk, a major agro-industrial residue, was successfully valorized into a stable, antioxidant-rich instant tea powder through optimized roasting, aqueous extraction, and freeze-drying processes. Controlled roasting at 117 °C for 225 s enhanced phenolic release and aromatic development without excessive degradation, while optimized extraction at 70 °C for 45 min maximized bioactive recovery using water as a green solvent. The combined physicochemical and microstructural analyses confirmed that roasting served as an efficient pretreatment, promoting cell-wall disruption and improving mass transfer during extraction. The instant tea formulation demonstrated desirable sensory quality, solubility, and microbial safety, and its phenolic release followed Fickian diffusion at moderate temperatures, ensuring controlled dissolution in water. From a sustainability perspective, the use of coffee husk contributes to waste reduction and resource valorization without competing with primary food resources. In addition, the use of water as the sole extraction solvent is consistent with green processing principles, reducing the environmental burden associated with the consumption and disposal of organic solvents. Despite these promising results, the present study has certain limitations. In particular, long-term storage stability of the instant coffee husk tea, including potential changes in sensory quality and bioactive compound retention during shelf life, was not evaluated and warrants further investigation in future studies.Author contributions
Thi-Ngoc-Mai Tran, Duong Anh Tan: investigation, formal analysis, validation, software, originate manuscript; Nguyen Huu Trung, Thi-Hong-No Nguyen, Phung Van Trung, N. Trung-Tien Dieu, Van-Dung Le, Phuoc-Dat Duong, Chi-Hien Dang, Cao-Hien Nguyen, Nguyen Thi Thanh Tu: investigation, visualization, validation; Dinh-Tri Mai, Thanh-Danh Nguyen: conceptualization, supervisor, writing – review & editing.Conflicts of interest
The authors of this paper state that they have no competing financial interests or personal relationships that could have influenced the reported work.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5fb00898k.Acknowledgements
This research is funded by Vietnam Academy of Science and Technology under grant number NCXS02.04/24–25.References
- L. F. Rojas, P. Zapata and L. Ruiz-Tirado, Curr. Opin. Green Sustainable Chem., 2022, 34, 100585 CrossRef CAS.
- S. d. O. Silva, A. K. C. Mafra, F. M. Pelissari, L. Rodrigues de Lemos and G. Molina, Microorganisms, 2025, 13, 1789 CrossRef CAS PubMed.
- K. Tsigkou, B. A. Demissie, S. Hashim, P. Ghofrani-Isfahani, R. Thomas, K. F. Mapinga, S. K. Kassahun and I. Angelidaki, Renew. Sustain. Energy Rev., 2025, 210, 115263 CrossRef.
- M. Worku, T. Astatkie and P. Boeckx, Heliyon, 2022, 8(4), e09201 CrossRef CAS PubMed.
- C. Bilen, D. El Chami, V. Mereu, A. Trabucco, S. Marras and D. Spano, Plants, 2022, 12, 102 CrossRef PubMed.
- M. A. Ali and S. Bhowmik, RSC Sustain., 2025, 3, 4410–4425 RSC.
- D. Chavan, S. Arya and S. Kumar, in Advanced Organic Waste Management, Elsevier, 2022, pp. 15–31 Search PubMed.
- A. de Oliveira, T. F. Moreira, B. P. Silva, G. Oliveira, V. M. C. Teixeira, L. S. Watanabe, S. L. Nixdorf, L. E. Leal, L. G. A. Pessoa and F. A. V. Seixas, Food Res. Int., 2024, 178, 113878 CrossRef PubMed.
- M. M. Strieder, V. L. Sanches and M. A. Rostagno, Food Res. Int., 2024, 175, 113690 CrossRef CAS PubMed.
- L. B. Cangussu, J. C. Melo, A. S. Franca and L. S. Oliveira, Foods, 2021, 10, 3125 CrossRef CAS PubMed.
- A. Iriondo-DeHond, N. A. Garcia, B. Fernandez-Gomez, E. Guisantes-Batan, F. V. Escobar, G. P. Blanch, M. I. San Andres, S. Sanchez-Fortun and M. D. del Castillo, Innov. Food Sci. Emerg. Technol., 2019, 51, 194–204 CrossRef CAS.
- S. I. Mussatto, L. F. Ballesteros, S. Martins and J. A. Teixeira, Separ. Purif. Technol., 2011, 83, 173–179 CrossRef.
- A. D. J. de Farias Marques, J. de Lima Tavares, L. M. de Carvalho, T. L. Abreu, D. A. Pereira, M. M. F. Santos, M. S. Madruga, L. L. de Medeiros and T. K. A. Bezerra, Food Chem., 2022, 393, 133451 CrossRef CAS PubMed.
- T. L. Abreu, M. Estévez, L. M. de Carvalho, L. L. de Medeiros, V. C. da Silva Ferreira, B. R. Salu, M. L. V. Oliva, M. S. Madruga and T. K. A. Bezerra, J. Sci. Food Agric., 2024, 104, 1833–1842 CrossRef CAS PubMed.
- E. F. Ribeiro and N. Jorge, Food Sci. Technol., 2017, 37, 5–10 CrossRef.
- D. Gocmen, Y. Sahan, E. Yildiz, M. Coskun and İ. A. Aroufai, J. Food Sci. Technol., 2019, 56, 2979–2988 CrossRef CAS PubMed.
- V.-D. Le, D.-T. Mai, C.-H. Dang, C.-H. Nguyen, H. T. Phat and T.-D. Nguyen, RSC Adv., 2025, 15, 36625–36641 RSC.
- L. H. Reichembach, P. Guerrero, C. L. de Oliveira Petkowicz and K. de la Caba, Carbohydr. Polym., 2024, 334, 122057 CrossRef CAS PubMed.
- G. Divyashri, R. Swathi, T. K. Murthy, M. Anagha, O. Sindhu and B. Sharada, Discover Food, 2024, 4, 181 CrossRef CAS.
- A. Bobková, M. Hudáček, S. Jakabová, Ľ. Belej, M. Capcarová, J. Čurlej, M. Bobko, J. Árvay, I. Jakab and J. Čapla, J. Environ. Sci. Health, Part B, 2020, 55, 495–500 CrossRef PubMed.
- J.-K. Moon, H. S. Yoo and T. Shibamoto, J. Agric. Food Chem., 2009, 57, 5365–5369 CrossRef CAS PubMed.
- L. El Hosry, V. Elias, V. Chamoun, M. Halawi, P. Cayot, A. Nehme and E. Bou-Maroun, Foods, 2025, 14, 1881 CrossRef CAS PubMed.
- H. Wu, P. Lu, Z. Liu, J. Sharifi-Rad and H. A. Suleria, Food Sci. Nutr., 2022, 10, 2408–2425 CrossRef CAS PubMed.
- N. Reis, A. S. Franca and L. S. Oliveira, LWT--Food Sci. Technol., 2013, 50, 715–722 CrossRef CAS.
- V. Sánchez-Martín, M. B. López-Parra, A. Iriondo-DeHond, A. I. Haza, P. Morales and M. D. del Castillo, 2023.
- Q. D. Do, A. E. Angkawijaya, P. L. Tran-Nguyen, L. H. Huynh, F. E. Soetaredjo, S. Ismadji and Y.-H. Ju, J. Food Drug Anal., 2014, 22, 296–302 CrossRef CAS PubMed.
- K.-S. Youn and H.-S. Chung, LWT--Food Sci. Technol., 2012, 46, 305–310 CrossRef CAS.
- G. Coman and G. Bahrim, Ann. Microbiol., 2011, 61, 773–779 CrossRef CAS PubMed.
- G. J. Swamy, A. Sangamithra and V. Chandrasekar, Dyes Pigm., 2014, 111, 64–74 CrossRef CAS.
- R. Ghelich, M. R. Jahannama, H. Abdizadeh, F. S. Torknik and M. R. Vaezi, Composites, Part B, 2019, 166, 527–541 CrossRef CAS.
- P. T. N. Nguyen, C. K. Van, N. A. T. Do, T. C. T. Tran, T. K. T. Dang, T. M. T. Pham, B. L. Tran and N. T. A. Ton, Mater. Today: Proc., 2022, 56, 1461–1467 CAS.
- Y. Deng, S. Zhao, X. Yang, H. Guo, Y. Li, W. Xu, J. Chen, Z. Wang, M. Guo and D. Liu, ACS Sustain. Chem. Eng., 2021, 9, 3702–3709 CrossRef CAS.
- X. Zhuang, Y. Chen, S. Wang, K. Zhong, W. Sui, C. Ma and M. Wu, Foods, 2025, 14, 1345 CrossRef CAS PubMed.
- D. J. Nicolin, D. F. Rossoni and L. M. M. Jorge, J. Food Eng., 2016, 184, 63–68 CrossRef.
- J. Bayuo, M. A. Abukari and K. B. Pelig-Ba, Appl. Water Sci., 2020, 10, 1–12 Search PubMed.
- N. Danbaba, I. Nkama and M. H. Badau, Int. J. Food Sci. Nutr. Eng., 2015, 5, 40–52 Search PubMed.
- A. Gui, F. Ye, J. Xue, S. Wang, P. Liu, X. Wang, J. Teng, L. Feng, J. Xiang and P. Zheng, Foods, 2024, 13, 3968 CrossRef CAS PubMed.
- C. Petisca, T. Pérez-Palacios, A. Farah, O. Pinho and I. M. Ferreira, Food Bioprod. Process., 2013, 91, 233–241 CrossRef CAS.
- O. NOVOtNý, K. CeJpeK and J. VelíšeK, Czech J. Food Sci., 2008, 26, 117–131 Search PubMed.
- M. Ginz, H. H. Balzer, A. G. Bradbury and H. G. Maier, Eur. Food Res. Technol., 2000, 211, 404–410 CrossRef CAS.
- G. P. Rizzi, Food Rev. Int., 2008, 24, 416–435 CrossRef CAS.
- S. Ma, C. Ding, C. Zhou, H. Shi, Y. Bi, H. Zhang and X. Xu, Heliyon, 2024, 10 Search PubMed.
- X. Cao, H. Wu, C. G. Viejo, F. R. Dunshea and H. A. Suleria, Int. J. Food Sci. Technol., 2023, 58, 1007–1027 CrossRef CAS.
- H. Liu, T. Hui, F. Fang, Q. Ma, S. Li, D. Zhang and Z. Wang, Foods, 2021, 10, 2387 CrossRef CAS PubMed.
- S. Chen, D. Wang and Y. Xu, J. Agric. Food Chem., 2013, 61, 9712–9718 CrossRef CAS PubMed.
- C. Chen, W. Zhou, H. Yu, J. Yuan and H. Tian, Molecules, 2020, 25, 4308 CrossRef CAS PubMed.
- F. G. S. Araújo, P. R. C. Marcelino, M. U. do Nascimento, É. P. P. Queiroz, B. D. L. de Araújo, H. R. do Amaral, F. P. D. Lopes, J. C. Moulin and M. P. Oliveira, Cellulose, 2025, 1–13 Search PubMed.
- D. Howe, M. Garcia-Perez, D. Taasevigen, J. Rainbolt, K. Albrecht, H. Li, L. Wei, A. McDonald and M. Wolcott, J. Anal. Appl. Pyrolysis, 2019, 142, 103691 CrossRef.
- Z. Qin, C.-L. Zheng, R. Wang, Y.-L. Chang, A.-H. Zhang, Z.-M. Chen, Y.-C. Ji, Z. Qin, H.-M. Liu and X.-Y. Wen, Int. J. Biol. Macromol., 2025, 305, 141248 CrossRef CAS PubMed.
- T. S. de Souza and H. Y. Kawaguti, Food Bioprocess Technol., 2021, 14, 1446–1477 CrossRef CAS.
- S. Behne, H. Franke, S. Schwarz and D. W. Lachenmeier, Molecules, 2023, 28, 5540 CrossRef CAS PubMed.
- R. Rodrigues, M. B. P. P. Oliveira and R. C. Alves, Cosmetics, 2023, 10, 12 CrossRef CAS.
- E. Olechno, A. Puścion-Jakubik, M. E. Zujko and K. Socha, Foods, 2021, 10, 1208 CrossRef CAS PubMed.
- L. N. Bell, C. R. Wetzel and A. N. Grand, Food Res. Int., 1996, 29, 785–789 CrossRef CAS.
- N. A. Peppas, in Medical Applications of Controlled Release, CRC Press, 2019, pp. 169–188 Search PubMed.
- Y. Deng, W. Wang and D. Liu, Food Bioprocess Technol., 2024, 17, 1017–1029 CrossRef CAS.
- A. J. Krüger and A. T. Tolmay, J. Appl. Polym. Sci., 2002, 84, 806–813 CrossRef.
- P. L. Ritger and N. A. Peppas, J. Contr. Release, 1987, 5, 23–36 CrossRef CAS.
- P. E. Igoumenidis and V. T. Karathanos, J. Food Eng., 2016, 174, 1–7 CrossRef CAS.
- P. E. Igoumenidis, S. V. Iosifidis, E. Lopez-Quiroga, S. Bakalis and V. T. Karathanos, J. Food Sci., 2019, 84, 2859–2867 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |


