Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Ultrasound-enhanced extraction as a green intensification strategy for phenolic release and in vitro gastrointestinal bioaccessibility from longan byproducts
Idris Kaida
Zubairu
 a,
Noppol
Leksawasdi
ab,
Sutee
Wangtueai
a,
Noppol
Leksawasdi
ab,
Sutee
Wangtueai
 ab,
Pinpanit
Boonchuay
c,
Su Lwin
Htike
ab,
Fei
Lao
d,
Juan Manuel
Castagnini
ab,
Pinpanit
Boonchuay
c,
Su Lwin
Htike
ab,
Fei
Lao
d,
Juan Manuel
Castagnini
 e and
Yuthana
Phimolsiripol
e and
Yuthana
Phimolsiripol
 *ab
*ab
aFaculty of Agro-industry, Chiang Mai University, 50100, Thailand. E-mail: yuthana.p@cmu.ac.th
bCenter of Excellence in Agro Bio-Circular-Green Industry, Chiang Mai University, Chiang Mai, 50100 Thailand
cFood Innovation and Packaging Center, Chiang Mai University, 50100, Thailand
dCollege of Food Science and Nutritional Engineering, China Agricultural University, Beijing, 100083, China
eResearch Group in Innovative Technologies for Sustainable Food (ALISOST), Department of Preventive Medicine and Public Health, Food Science, Toxicology and Forensic Medicine, Faculty of Pharmacy and Food Science, Universitat de València, Avda. Vicent Andrés Estellés, s/n, 46100 Burjassot, Valencia, Spain
First published on 20th January 2026
Abstract
Longan byproducts from industry, often discarded as waste, contain bioactive compounds with potential health benefits. In this study, temperature, solvent ratio, and extraction time were optimized using response surface methodology, comparing water bath and ultrasonic probe techniques. The optimized extracts were evaluated for percentage yield, total phenolic content (TPC), total flavonoid content (TFC), and antioxidant capacity. Ultrasonic-assisted extraction (UAE) achieved significantly higher yield (11.23%), as well as higher phenolic content (219.11 mg GAE/100 g DW), and antioxidant activity (DPPH IC50: 3.78) than the water bath method. The optimized extracts were subjected to simulated gastrointestinal conditions. UAE extracts showed superior intestinal retention with more than 120 mg GAE/100 g DW of TPC compared with >20 mg GAE/100 g DW for the water bath extracts. Antioxidant activities were also higher, with UAE extracts retaining >95% activity, whereas water bath extracts showed only 55–75% activity retention. The digested UAE extract was analyzed using high-performance liquid chromatography (HPLC) to evaluate the impact of in vitro digestion on polyphenols. The compounds corilagin, ellagic acid, gallic acid, epicatechin, procyanidin A2, quercetin, and pinostrobin showed significant increases in oral digestion. These findings demonstrate that UAE increased the extraction efficiency of phenolics from longan processing waste and preserved their activity during oral digestion and, to a lesser extent, in the gastric and intestinal stages, as confirmed by HPLC results. Encapsulation to improve compound stability and the evaluation of cell absorption and digestion are recommended to improve the understanding of bioaccessibility. This study supports the valorization of longan processing waste as a sustainable source of nutraceutical ingredients.
Sustainability spotlightThis study introduces ultrasound-assisted extraction (UAE) as an environmentally friendly method that enhances the value of longan processing byproducts—usually thrown away—by turning them into phenolic-rich, bioaccessible extracts with potent antioxidant properties. By greatly decreasing solvent and energy use while improving extraction efficiency, the UAE method demonstrates sustainable progress in food processing. The research promotes a circular bioeconomy through resource recovery and waste reduction, directly supporting UN SDG 12 (responsible consumption and production) and SDG 9 (industry, innovation and infrastructure). Additionally, by encouraging eco-efficient conversion of agricultural waste into nutraceutical ingredients, it contributes to SDG 3 (good health and well-being), emphasizing the environmental and societal advantages of green technologies in the food sector. |
1 Introduction
Longan (Dimocarpus longan Lour.) is a popular fruit in temperate and sub-tropical regions and a significant economic crop in Southeast Asia, with Thailand being the second-largest producer after China. In 2023, 80–90% of Thailand's exports to China were valued at US$474 million. In 2025, Thailand's longan production reached 1.57 million tons, a 10.8% increase from 2024.1 The processing of longan yields a substantial quantity of byproducts, including pericarp and seeds, which account for approximately 30–40% of the fresh fruit by weight.2 These byproducts are highly valued in traditional Chinese medicine for their health-promoting properties, including anticancer, antiaging, and memory-enhancing effects.3 Given their bioactive potential, particularly in extracts, there is increasing interest in using these byproducts to deliver additional functional and health benefits while contributing to sustainability.Extraction of bioactive compounds from longan processing byproducts has gained attention due to their health benefits and various applications, particularly the presence of compounds such as corilagin, ellagic acid, and gallic acid.4,5 These compounds help mitigate oxidative stress and related diseases.6,7 Notably, longan seeds exhibit higher antioxidant activity than lychee seeds (6122 vs. 334 mg GAE/100 g DW) under comparable extraction conditions.8 The growing demand for natural products, coupled with the need to valorize longan processing byproducts, has encouraged research into efficient extraction methods that maximize yield while preserving bioactive compounds.4,6
The stability and bioavailability of bioactive compounds from longan processing byproducts determine their efficacy in functional foods, as the benefits depend on their survival through digestion and absorption. Studies have reported varying stability of bioactive compounds, with some retaining their bioactivity despite exposure to acidic pH, digestive enzymes, and bile salts.9 Additionally, the extraction method can impact the bioaccessibility of bioactive compounds during digestion.10,11 For instance, phenolic compounds in longan peel maintained antioxidant capacity throughout digestion.12 However, ensuring bioavailability remains a challenge, as not all compounds survive digestion or achieve effective concentrations in the bloodstream. Nevertheless, in vitro models suggest a significant proportion remains accessible for uptake, indicating potential in vivo benefits.2,5
Despite advances in extraction techniques, several obstacles remain in achieving optimal extraction conditions. Conventional extraction technologies often yield low recovery13 and compromise compound quality due to thermal degradation of heat-sensitive molecules, oxidation during prolonged processing, non-selective solvent extraction, and limited release of bioactive compounds from rigid plant cell matrices.8 Moreover, studies on the optimization of water bath extraction and ultrasound-assisted extraction (UAE) remain limited. This study applied Response Surface Methodology (RSM) to optimize extraction conditions, aiming to enhance the recovery of bioactive compounds and antioxidants from longan processing byproducts.
This work focuses on optimizing extraction conditions for longan processing byproducts using the UAE and water bath methods via RSM, while assessing various extraction factors. It further examines the relationships among optimal extraction conditions, stability, and gastrointestinal bioaccessibility of bioactive compounds in longan processing byproducts. These findings provide valuable insights into the potential application of longan processing byproducts in functional foods and nutraceuticals, contributing to sustainability and supporting the growing demand for natural, health-promoting ingredients.
2 Materials and methods
2.1 Materials and sample preparation
Longan byproducts (LBP) were used in a mixed form, consisting of pericarps and (seeds and residual pulp) in approximate ratios of 63% and 37%, on a dry weight basis, respectively, as described in Fig. S1, obtained from PM 80 Ltd (Lamphun, Thailand). Chemicals and reagents, including DPPH, TPTZ, ABTS (2,2-diphenyl-1-picrylhydrazyl, 2,4,6-tris(2-pyridyl)-s-triazine, and 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid), respectively), gallic acid, catechin, and Trolox, were all from Sigma (St. Louis, MO, USA). Ethanol (99.8%) was sourced from a liquor distillery in Bangkok. Folin–Ciocalteu reagent, HPLC-grade standards, including procyanidin A2, gallic acid, epicatechin, ellagic acid, corilagin, quercetin, pinocembrin, pinostrobin, and 5-fluorouracil (≥99% purity) were supplied by Merck KGaA (Darmstadt, Germany). Sample preparation followed a modified version of the method described by Tan et al.5 Briefly, the byproducts were milled using an herb grinder (Heavybao, vertical, HLB-JGY-AB, China) and passed through a 60-mesh sieve to produce a fine powder. The powdered samples were transferred into polyethylene bags and sealed until further analysis.2.2 Experimental methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) was adopted from Bai et al.2 and the extraction volume was 30 mL, consisting of equal volumes of food-grade ethanol (99.8% or 95%) and distilled water. The suspension was stirred at 150 rpm at 30 ± 1 °C, centrifuged (Rotina 380R, Hettich Zentrifugen, Tuttlingen, Germany) at 8644 × g for 15 min at 25 °C, and the supernatant was filtered through Whatman No. 1 paper. The solvent was removed under reduced pressure at 40 °C using a rotary evaporator (BUCHI, Rotavapor® R-300, Thailand Ltd), and the resulting crude extract was stored at −20 °C until further analysis. Optimization variables, with coded and actual values, are summarized in Table 1. Regression coefficients were obtained via multiple linear regression fitted to a second-order polynomial model (eqn (1) and (2)), and the desirability function (Minitab® Version 21.1.1, Minitab LLC, Chicago, USA) was applied to determine the optimal extraction conditions.
10) was adopted from Bai et al.2 and the extraction volume was 30 mL, consisting of equal volumes of food-grade ethanol (99.8% or 95%) and distilled water. The suspension was stirred at 150 rpm at 30 ± 1 °C, centrifuged (Rotina 380R, Hettich Zentrifugen, Tuttlingen, Germany) at 8644 × g for 15 min at 25 °C, and the supernatant was filtered through Whatman No. 1 paper. The solvent was removed under reduced pressure at 40 °C using a rotary evaporator (BUCHI, Rotavapor® R-300, Thailand Ltd), and the resulting crude extract was stored at −20 °C until further analysis. Optimization variables, with coded and actual values, are summarized in Table 1. Regression coefficients were obtained via multiple linear regression fitted to a second-order polynomial model (eqn (1) and (2)), and the desirability function (Minitab® Version 21.1.1, Minitab LLC, Chicago, USA) was applied to determine the optimal extraction conditions.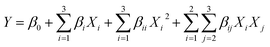 | (1) |
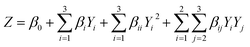 | (2) |
| Extraction methods | Symbol codes | Variables | Units | Levels | ||
|---|---|---|---|---|---|---|
| Low (−1) | Center (0) | High (+1) | ||||
| Water bath method | X 1 | Solvent ratio | % | 60 | 70 | 80 |
| X 2 | Temperature | °C | 40 | 60 | 80 | |
| X 3 | Time | min | 60 | 150 | 240 | |
| UAE method | Y 1 | Solvent ratio | % | 40 | 60 | 80 |
| Y 2 | Temperature | °C | 40 | 60 | 80 | |
| Y 3 | Time | min | 30 | 45 | 60 | |
2.3 Determination of bioactive compounds and antioxidants
 | (3) |
The ABTS radical scavenging activity of the extracts was determined using the method described by Chaiwong et al.16 with minor modifications. Briefly, the ABTS radical cation (ABTS+) was generated, and before analysis, the ABTS+ solution was diluted with deionized water to an absorbance of 0.70 ± 0.02 at 734 nm and equilibrated at 30 °C for 5 min. Absorbance was measured at 734 nm using the microplate reader. Results were expressed as mg Trolox equivalents (TE) per 100 g dry weight (DW).
The FRAP was determined using the method described by Chaiwong et al.16 Absorbance was measured at 595 nm using the microplate reader. A Trolox standard curve (0–100 µg mL−1) was used, and results were expressed as mg Trolox equivalents (TE) per 100 g dry weight (DW).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) methanol to the digested extract. The bioaccessibility was determined using eqn 4
1 (v/v) methanol to the digested extract. The bioaccessibility was determined using eqn 4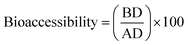 | (4) |
2.4 Identification of compounds by HPLC
Phenolic compounds were quantified by HPLC (1220 Infinity II LC System, Agilent, Santa Clara, CA, USA) using a C18 column (250 × 4.6 mm, 5 µm) at 30 °C with acetonitrile and 0.1% formic acid in water as the mobile phases under gradient elution (1.0 mL min−1, 10–20 µL injection). Detection was performed at 254–280 nm. Samples were diluted in 80% methanol and filtered (0.45 µm). The LOD and LOQ were determined from the standard deviation of the response and the calibration slope;2,5 see the SI.2.5 Statistical analysis
All determinations were performed in triplicate and analyzed using Analysis of Variance (ANOVA) with SPSS software (Version 23.0, Chicago, IL, USA). Duncan's multiple-range test was used to separate means, and differences were considered statistically significant at p < 0.05. Statistical modelling and optimization were conducted using Minitab statistical software (Minitab® Version 21.1.1, Minitab LLC, Chicago, USA).3 Results and discussion
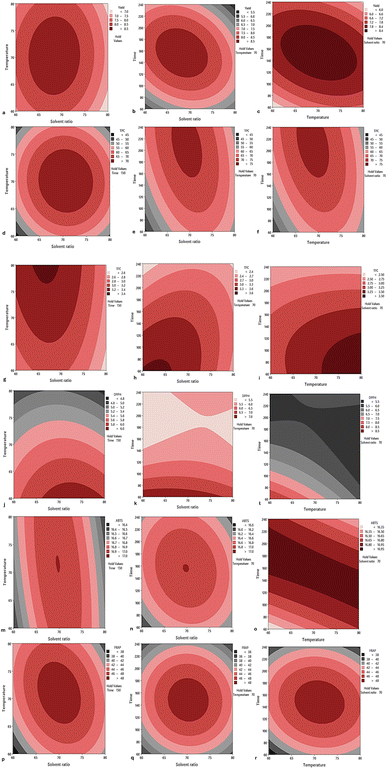
3.1 Experimental responses for the water bath method
The extraction yield ranged from 5.30% to 9.03% (Table 2), indicating that solvent ratio, temperature, and time significantly (p < 0.05) influenced the efficiency of bioactive compound recovery. The highest yield (9.03%) was achieved at 70% solvent, 70 °C, and 150 min, confirming that a combination of moderate temperature and extraction time, with a balanced solvent polarity, is optimal for extracting phenolics and flavonoids. The result was within the range (8.1–15.5%) of extractable yield reported by Sai-Ut et al.,8 for longan seeds. The relatively lower value observed in this study may be due to compound degradation or limited solubilization under extreme conditions. In contrast, the yield was higher than the maximum value (8.5%) reported by Fikry et al.,13 for the ultrasound-assisted extraction of longan seeds, indicating that extreme extraction parameters may cause degradation or reduced efficiency. As shown in Fig. 1a–c, the contour plots illustrate the influence of solvent ratio, temperature, and extraction time on extraction yield, with an optimum region evident at the central points. In contrast, extreme solvent concentrations, prolonged extraction times, or elevated temperatures resulted in reduced yields. The regression equation (Table 4) for percentage yield, derived from the coded factors, indicated a good fit for the linear, quadratic, and interaction models.| Runs | X 1 (solvent ratio, %) | X 2 (temperature, °C) | X 3 (time, min) | Extraction yield (%) | TPC (mg GAE/100 g DW) | TFC (mg CE/100 g DW) | DPPH IC50 | ABTS (mg TE/100 g DW) | FRAP (mg TE/100 g DW) |
|---|---|---|---|---|---|---|---|---|---|
| a GAE: gallic acid equivalent; CE: catechin equivalent; TE: Trolox equivalent; DW: dry weight. | |||||||||
| 1 | 60.00 | 60.00 | 150.00 | 7.49 ± 0.64 | 42.89 ± 0.74 | 2.77 ± 0.07 | 5.82 ± 0.03 | 16.34 ± 0.45 | 35.07 ± 0.06 |
| 2 | 80.00 | 60.00 | 150.00 | 6.35 ± 0.32 | 48.32 ± 0.89 | 2.58 ± 0.03 | 6.19 ± 0.07 | 16.67 ± 0.87 | 41.08 ± 0.77 |
| 3 | 60.00 | 80.00 | 150.00 | 7.56 ± 0.33 | 46.70 ± 0.29 | 3.20 ± 0.05 | 4.64 ± 0.02 | 16.58 ± 0.35 | 42.54 ± 0.46 |
| 4 | 80.00 | 80.00 | 150.00 | 6.65 ± 0.67 | 46.37 ± 0.87 | 2.86 ± 0.12 | 4.63 ± 0.00 | 16.46 ± 0.78 | 39.46 ± 0.28 |
| 5 | 60.00 | 70.00 | 60.00 | 5.35 ± 0.65 | 43.87 ± 0.28 | 3.74 ± 0.11 | 7.37 ± 0.04 | 16.03 ± 0.05 | 35.96 ± 0.67 |
| 6 | 80.00 | 70.00 | 60.00 | 6.13 ± 0.78 | 59.42 ± 0.29 | 2.56 ± 0.34 | 6.78 ± 0.33 | 16.12 ± 0.57 | 36.78 ± 0.75 |
| 7 | 60.00 | 70.00 | 240.00 | 7.03 ± 0.23 | 63.67 ± 0.22 | 2.18 ± 0.17 | 5.20 ± 0.08 | 16.42 ± 0.63 | 36.68 ± 0.88 |
| 8 | 80.00 | 70.00 | 240.00 | 5.30 ± 0.76 | 56.89 ± 0.84 | 2.23 ± 0.19 | 5.77 ± 0.56 | 16.04 ± 0.97 | 35.26 ± 0.64 |
| 9 | 70.00 | 60.00 | 60.00 | 5.61 ± 0.97 | 43.53 ± 0.18 | 3.06 ± 0.23 | 8.47 ± 0.23 | 16.23 ± 0.35 | 36.74 ± 0.45 |
| 10 | 70.00 | 80.00 | 60.00 | 7.08 ± 0.46 | 55.42 ± 0.88 | 3.65 ± 0.09 | 6.09 ± 0.11 | 16.82 ± 0.74 | 39.50 ± 0.63 |
| 11 | 70.00 | 60.00 | 240.00 | 7.20 ± 0.43 | 67.87 ± 0.26 | 2.54 ± 0.32 | 5.10 ± 0.09 | 16.87 ± 0.76 | 38.43 ± 0.67 |
| 12 | 70.00 | 80.00 | 240.00 | 5.78 ± 0.02 | 57.11 ± 0.11 | 2.66 ± 0.37 | 5.89 ± 0.45 | 16.32 ± 0.36 | 36.79 ± 0.98 |
| 13 | 70.00 | 70.00 | 150.00 | 9.03 ± 0.67 | 74.53 ± 0.46 | 3.28 ± 0.06 | 5.79 ± 0.21 | 17.00 ± 0.57 | 47.88 ± 0.57 |
| 14 | 70.00 | 70.00 | 150.00 | 8.76 ± 0.56 | 79.56 ± 0.38 | 3.41 ± 0.02 | 5.32 ± 0.32 | 17.00 ± 0.89 | 51.29 ± 0.88 |
| 15 | 70.00 | 70.00 | 150.00 | 8.69 ± 0.83 | 70.24 ± 0.29 | 3.13 ± 0.36 | 5.89 ± 0.46 | 17.00 ± 0.64 | 48.21 ± 0.44 |
| Adj-R2 | — | — | — | 96.420 | 92.360 | 91.110 | 92.440 | 94.350 | 91.410 |
| p-Value | — | — | — | 0.000 | 0.002 | 0.003 | 0.002 | 0.001 | 0.003 |
TPC ranged from 42.89 to 79.56 mg GAE/100 g DW, with the highest value (79.56 mg GAE/100 g DW) observed at 70% solvent, 70 °C, and 150 min, indicating that moderate extraction conditions promote phenolic recovery. Lower values observed at solvent concentrations of 60% and 80% suggest that both overly polar and non-polar systems reduce solubility and extraction efficiency.12 Similarly, Natungnuy et al.18 reported a TPC of 84.73 mg GAE per g, which was higher than that obtained in this study, likely due to the longer extraction duration (7 days). Mahindrakar and Rathod19 reported that Soxhlet extraction yielded 30.05 mg GAE per g of phenolics at 100 °C after 6 h, whereas batch extraction achieved a markedly higher TPC of 79.87 mg GAE per g at 50 °C within 105 min. Both values were lower than those observed in the present study, possibly due to differences in extraction time, temperature, and the material matrix. Likewise, Cao et al.20 reported a maximum TPC of 27.84 ± 0.33 mg GAE per g DW from pawpaw leaves using RSM-optimized microwave-assisted extraction (14 min, 460 W, 77 °C, 30 mL g−1), which was comparable to the values obtained in this study. Fig. 1d–f further illustrates these effects, with optimum conditions observed near the central points, with temperature–solvent interaction exerting the strongest influence. The regression models (linear, quadratic, and interaction) provided an adequate description of the TPC prediction, as summarized in Table 3.
| Responses | Water bath extraction method |
|---|---|
| Yield | −95.8 + 1.622X1 − 0.01140X12 − 0.00675X22 − 0.000214X32 − 0.000697X1X3 − 0.000803X2X3 |
| TPC | −1570 + 1.100X3 − 0.1436X12 − 0.1434X22 − 0.000549X32 − 0.00620X1X3 − 0.00629X2X3 |
| TFC | −16.70 + 0.459X1 + 0.148X2 − 0.01078X3 − 0.003604X12 − 0.000029X32 + 0.000342X1X3 |
| DPPH | 3.9 + 0.048X2 − 0.1247X3 + 0.000104X32 + 0.000881X2X3 |
| ABTS | −18.29 − 0.004475X12 − 0.000049X32 − 0.001125X1X2 − 0.000131X1X3 − 0.000317X2X3 |
| FRAP | −579.5 − 0.05642X12 − 0.03947X22 − 0.000903X32 − 0.02272X1X2 |
Total flavonoid content ranged from 2.18 to 3.74 mg CE/100 g DW. The highest TFC (3.74 mg CE/100 g DW) was observed at 60% solvent, 70 °C, and 60 min, whereas some runs with higher TPC showed comparatively low TFC values (2.18 mg CE/100 g DW). This indicates that phenolics and flavonoids respond differently to extraction conditions, likely due to structural differences that influence their solubility and stability.14
The contour plots (Fig. 1g–i) highlight an optimum region around the central points, with the solvent–temperature interaction showing the most substantial effect. Prolonged extraction times promoted higher TFC recovery and the recovery of other compounds; however, they increased the risk of degradation.21 Excessively long extraction durations further reduce efficiency, suggesting potential degradation of flavonoids.
For antioxidant activity, DPPH IC50 values ranged widely, with the lowest IC50 (4.63), indicating strong radical-scavenging activity. ABTS values ranged from 16.03 to 17.00 mg TE/100 g DW, which were lower than the value (94.04 mg TE g−1) obtained by Natungnuy et al.,18 for longan seeds. In contrast, FRAP values ranged from 35.07 to 51.29 mg TE/100 g DW, with the highest value (51.29 mg TE/100 g DW) corresponding to the maximum TPC. Antioxidant activity showed a closer association with total phenolic content than with total flavonoid content, suggesting that phenolic compounds were the main contributors to the observed antioxidant capacity.22 These further indicate that natural plant extracts are typically abundant in antioxidants, making them valuable sources of bioactive compounds. This may be due to the broader variety of compounds included in TPC, which exhibit a wider range of antioxidant properties than those represented in TFC.23 As shown in Fig. 1j–l, the contour plots reveal that moderate solvent ratios, around 70%, combined with lower temperatures, favor higher antioxidant activity (lower IC50). In contrast, extreme solvent concentrations and prolonged extraction times increased IC50 values, indicating reduced antioxidant potential, likely due to thermal or oxidative degradation of phenolics.24 Additionally, Fig. 1m–r describes the contour plot interaction effects for ABTS and FRAP, which exhibited different response patterns to the coded variables.
The regression model (Table 3) confirmed highly significant fits for all response parameters (Adj-R2 > 0.91, p < 0.005), indicating that extraction conditions substantially influence both yield and antioxidant activity, as measured by the ABTS and FRAP assays.
These findings highlight that moderate extraction conditions (70% solvent, 70 °C, 150 min) consistently produced higher yields, TPC, and antioxidant activities, while extreme conditions either reduced extraction efficiency or promoted degradation of sensitive compounds, consistent with the findings of Tourabi et al.25 Thus, balanced extraction parameters are critical for achieving both high yield and functional quality of extracts.
The desirability function, as first described by Derringer and Suich's26 approach in Minitab, was employed to determine the optimal extraction conditions by simultaneously considering multiple responses. Responses (% yield, TPC, TFC, DPPH, ABTS, and FRAP) were optimized simultaneously to maximize all responses while minimizing DPPH (antioxidant activity was expressed as IC50, with lower values indicating stronger radical-scavenging activity), targeting practical extraction efficiency. Each response was converted into a dimensionless desirability value (di) ranging from 0 to 1 (undesirable to entirely desirable). These values were aggregated using the geometric mean to calculate the overall desirability of 0.85 for the water bath. The optimization analysis predicted optimal conditions of 69.09% solvent ratio, 72.32 °C, and 149.09 min. Under these conditions, the overall desirability was 0.85, indicating a balance across all responses. The predicted values were experimentally validated, with percentage errors ranging from 0.65 to 7.55%, confirming the adequacy of the optimization model.
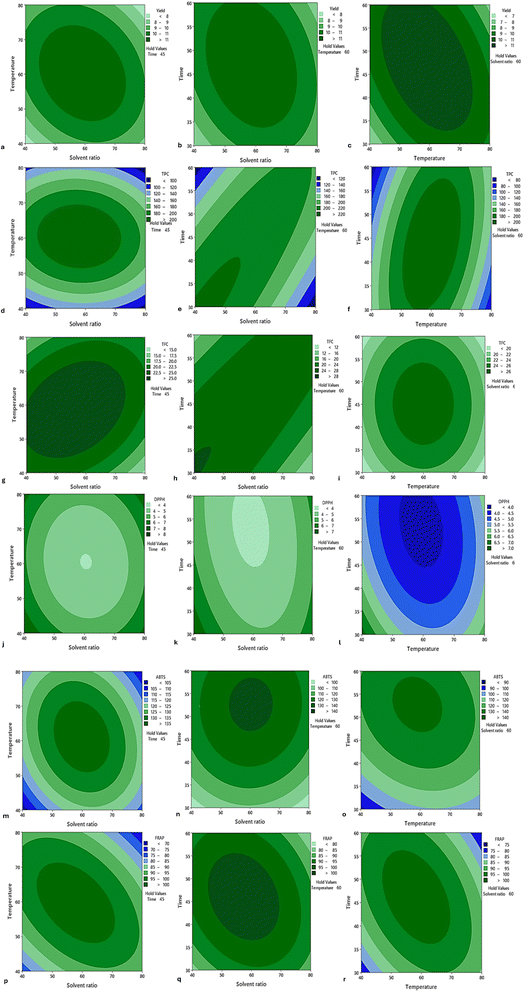
3.2 Experimental responses for the UAE method
Yields ranged from 7.03% to 12.43% (Table 4), which is generally higher than those obtained with water bath extraction. The highest yield (12.43%) was achieved at 60% solvent, 60 °C, and 45 min, indicating that moderate conditions support the highest yield. Similarly, Fikry et al.13 reported that longan seed extraction reached its highest yield at 55% ultrasonic power, 55 °C, and 25 min, which was 2.36 times higher than the minimum yield in their study. The consistent yields above 10% across multiple UAE extraction conditions suggest that it is more effective at breaking cell walls and enhancing solvent penetration than water bath extraction.| Runs | Y 1 (solvent ratio, %) | Y 2 (temperature, °C) | Y 3 (time, min) | Extraction yield (%) | TPC (mg GAE/100 g DW) | TFC (mg CE/100 g DW) | DPPH IC50 | ABTS (mg TE/100 g DW) | FRAP (mg TE/100 g DW) |
|---|---|---|---|---|---|---|---|---|---|
| a Temp.: temperature; GAE: gallic acid equivalent; CE: catechin equivalent; TE: Trolox equivalent; DW: dry weight. | |||||||||
| 1 | 40.00 | 40.00 | 45.00 | 7.40 ± 0.03 | 87.34 ± 0.98 | 21.80 ± 0.48 | 7.85 ± 0.03 | 103.64 ± 0.69 | 73.29 ± 0.86 |
| 2 | 80.00 | 40.00 | 45.00 | 8.43 ± 0.06 | 97.79 ± 0.34 | 14.37 ± 0.07 | 7.21 ± 0.02 | 112.33 ± 1.04 | 91.23 ± 0.57 |
| 3 | 40.00 | 80.00 | 45.00 | 9.36 ± 0.12 | 102.38 ± 0.62 | 16.81 ± 0.37 | 7.48 ± 0.06 | 116.34 ± 0.98 | 92.07 ± 0.76 |
| 4 | 80.00 | 80.00 | 45.00 | 7.03 ± 0.11 | 89.23 ± 0.27 | 20.56 ± 0.38 | 7.87 ± 0.01 | 102.34 ± 0.73 | 72.34 ± 0.92 |
| 5 | 40.00 | 60.00 | 30.00 | 7.52 ± 0.34 | 220.16 ± 0.67 | 28.35 ± 0.18 | 7.97 ± 0.05 | 87.34 ± 0.82 | 85.34 ± 0.84 |
| 6 | 80.00 | 60.00 | 30.00 | 9.05 ± 0.26 | 85.55 ± 0.37 | 11.37 ± 0.85 | 6.57 ± 0.03 | 88.24 ± 0.78 | 86.23 ± 0.47 |
| 7 | 40.00 | 60.00 | 60.00 | 10.11 ± 0.11 | 123.22 ± 0.38 | 15.23 ± 0.95 | 5.67 ± 0.09 | 124.89 ± 0.99 | 93.45 ± 0.73 |
| 8 | 80.00 | 60.00 | 60.00 | 8.67 ± 0.07 | 200.32 ± 0.92 | 25.47 ± 0.02 | 6.32 ± 0.03 | 129.13 ± 0.78 | 77.48 ± 0.85 |
| 9 | 60.00 | 40.00 | 30.00 | 7.21 ± 0.23 | 149.78 ± 0.28 | 18.78 ± 0.05 | 7.43 ± 0.02 | 91.23 ± 1.09 | 76.02 ± 0.58 |
| 10 | 60.00 | 80.00 | 30.00 | 9.84 ± 0.12 | 89.21 ± 0.18 | 18.08 ± 0.73 | 6.07 ± 0.00 | 107.45 ± 0.89 | 87.28 ± 0.48 |
| 11 | 60.00 | 40.00 | 60.00 | 10.23 ± 0.55 | 52.40 ± 0.84 | 18.83 ± 0.37 | 5.34 ± 0.07 | 123.89 ± 1.03 | 95.43 ± 0.94 |
| 12 | 60.00 | 80.00 | 60.00 | 7.32 ± 0.27 | 134.57 ± 0.74 | 17.68 ± 0.29 | 5.62 ± 0.03 | 117.49 ± 1.30 | 73.68 ± 1.03 |
| 13 | 60.00 | 60.00 | 45.00 | 12.11 ± 0.00 | 219.00 ± 0.49 | 29.00 ± 0.02 | 3.45 ± 0.09 | 142.00 ± 1.06 | 103.56 ± 0.87 |
| 14 | 60.00 | 60.00 | 45.00 | 11.23 ± 0.46 | 218.28 ± 0.37 | 25.98 ± 0.37 | 4.12 ± 0.02 | 137.46 ± 0.78 | 99.87 ± 1.07 |
| 15 | 60.00 | 60.00 | 45.00 | 12.43 ± 0.28 | 214.39 ± 0.48 | 27.00 ± 0.54 | 4.38 ± 0.10 | 135.73 ± 0.97 | 105.46 ± 1.05 |
| Adj-R2 | — | — | — | 90.770 | 94.270 | 95.100 | 93.110 | 87.360 | 90.780 |
| p-Value | — | — | — | 0.003 | 0.001 | 0.001 | 0.002 | 0.007 | 0.003 |
In their UAE study of acerola residue, this mechanism yielded a higher recovery of phenolic compounds (approximately 1034 mg GAE/100 g) than conventional extraction (780 mg GAE/100 g), a trend consistent with our observations.27,28 This finding is comparable to those of Zhang et al.,29 who reported TPC values of 22.09–132.47 mg GAE/100 g fresh weight in representative Chinese longan pulps. However, it was lower than the 57.8 ± 0.6 mg GAE per g DW reported by Bai et al.,2 for longan pericarp. The variation may be attributed to the use of coupled technology, combining microwave and ultrasound, in their study, as well as differences in the characteristics of the plant material.
The TFC obtained by the UAE ranged from 11.37 to 29.00 mg CE/100 g DW, with the highest value observed at 60% solvent, 60 °C, and 45 min. This condition correlated with high TPC runs, indicating that flavonoids respond particularly well to UAE extraction. Enhanced recovery of flavonoids and phenolic acid using UAE was reported by Jan and Gavahian27 in mulberry extracts. The suitability of this temperature for maximizing TFC agrees with the findings of Vo et al.,30 who observed that raising the temperature to 60 °C increased TPC and TFC by 1.2 and 1.1-fold, respectively, in passion fruit peels. For antioxidant activity, DPPH IC50 values ranged from 3.45 to 7.97, indicating stronger radical-scavenging capacity under UAE conditions. The best activity (lowest IC50, 3.45) was observed at the center point. Comparable results were reported by Sai-Ut et al.,8 in their optimization of lychee and longan seed extraction. Similarly, Ayele et al.15 obtained IC50 values ranging from 3.5 to 6.4 from Croton macrostachyus root extracts. ABTS values (87.34–142.00 mg TE/100 g DW) and FRAP values (72.34–105.46 mg TE/100 g DW), as presented in Table 4, correlated well with the DPPH results, indicating consistency among different antioxidant assays and confirming the strong radical-scavenging potential of the extracts. Antioxidant assays aligned strongly with TPC and TFC, supporting that phenolics are the main contributors to antioxidant capacity.24
High adjusted R2 values (90.77–95.10) with significant p-values (<0.01) indicate that the model reliably explains the influence of solvent ratio, temperature, and time on extraction outcomes with the UAE method. These findings demonstrate that UAE markedly enhances the extraction efficiency of LBP by enabling higher yields, greater recovery of phenolics and flavonoids, and enhanced antioxidant activity compared to conventional methods.13,31–33 Optimal responses were observed under moderate conditions (60% solvent ratio, 60 °C, 45 min), where both TPC and TFC reached maximum levels and antioxidant capacity was highest. This finding is consistent with those of Fuangchoom et al.,34 who reported similar results using microwave-assisted extraction. In contrast, extreme extraction conditions reduced efficiency or compromised the stability of bioactive compounds.35 Therefore, optimized UAE conditions are essential for maximizing bioactive recovery and preserving the functional integrity of the extracts.
The regression model described by the linear, quadratic, and interactive equations (Table 5) showed strong fits for all responses (Adj-R2 > 0.91, p < 0.005), confirming that extraction conditions significantly influenced yield and antioxidant activity. Moderate conditions (60% solvent, 60 °C, 45 min) gave superior results, while extreme conditions reduced efficiency or caused compound degradation, consistent with previous reports.12,14,19 Therefore, balanced parameters are crucial for maximizing yield and functional quality.
| Responses | Regression equations |
|---|---|
| Yield | −61.47 − 0.004602Y12 − 0.005069Y22 − 0.00554Y32 − 0.002100Y1Y2 − 0.002473Y1Y3 − 0.004617Y2Y3 |
| TPC | −335 − 0.0903Y12 − 0.2173Y22 − 0.1058Y32 + 0.1764Y1Y3 + 0.1190Y2Y3 |
| TFC | 3.5 + 0.119Y3 − 0.00898Y12 − 0.01338Y22 − 0.01615Y32 + 0.00698Y1Y2 + 0.02269Y1Y3 |
| DPPH | 54.98 − 0.4593Y3 + 0.005171Y12 + 0.003877Y22 + 0.002581Y32 + 0.001708Y1Y3 |
| ABTS | −407.1 + 7.90Y3 − 0.04044Y12 − 0.03390Y22 − 0.0659Y32 |
| FRAP | −346.3 − 0.02276Y12 − 0.02907Y22 − 0.03660Y32 − 0.02354Y1Y2 − 0.02751Y2Y3 |
3.3 Validation of the model
The predictive accuracy of the RSM models was validated by the close agreement between predicted and experimental values for both extraction methods (Table 6). The percentage errors across responses were all less than 10%, which is statistically acceptable for model reliability. This confirms that the models provided robust descriptions of extraction performance and can be used with confidence for process optimization.| Responses | Water bath method | UAE method | ||||
|---|---|---|---|---|---|---|
| Predicted values | Experimental values | % error | Predicted values | Experimental values | % error | |
| Yield (%) | 8.83 | 8.21 ± 0.02 | 7.55 | 11.95 | 11.23 ± 0.32 | 6.03 |
| TPC (mg GAE/100 g DW) | 73.80 | 70.86 ± 0.37 | 4.15 | 215.30 | 219.11 ± 1.45 | 1.77 |
| TFC (mg CE/100 g DW) | 3.33 | 3.49 ± 0.85 | 4.59 | 27.21 | 28.12 ± 0.08 | 3.34 |
| DPPH (IC50) | 5.54 | 5.67 ± 0.78 | 2.29 | 3.89 | 3.78 ± 0.05 | 2.83 |
| ABTS (mg TE/100 g DW) | 17.00 | 16.89 ± 0.47 | 0.65 | 140.73 | 141.24 ± 0.27 | 0.36 |
| FRAP (mg TE/100 g DW) | 49.09 | 52.43 ± 0.26 | 6.37 | 102.89 | 101.98 ± 0.13 | 0.88 |
The optimal extraction conditions were determined using the desirability function approach in Minitab software. % yield, TPC, TFC, ABTS, and FRAP were maximized, whereas DPPH was minimized (antioxidant activity was expressed as IC50, where lower values indicate stronger radical scavenging activity) to achieve practical extraction efficiency. Each response was converted into a dimensionless desirability value (di) ranging from 0 (undesirable) to 1 (fully desirable). These values were combined using the geometric mean to calculate an overall desirability of 0.93 for the UAE method. The optimization analysis predicted the optimal conditions to be 59.39%, 59.79 °C, and 47.89 min. Under these conditions, the overall desirability was 0.93, indicating an excellent balance across all responses. The predicted values were experimentally validated, with percentage errors ranging from 0.36 to 6.03%, confirming the adequacy of the UAE optimization model and indicating greater efficiency than the water bath-assisted method. The contour plot in Fig. 2 illustrates the interactive effects of the factors on all measured responses, whereas Table 5 summarizes the regression equations for each response along with their corresponding factors.
A comparative analysis of the two methods revealed that the UAE method outperformed the conventional water bath in extraction yield (%) and bioactive compound content. UAE achieved a higher yield of 11.23%, compared with 8.21% from the water bath-assisted method, a difference likely attributable to the ultrasonic cavitation,31 which promotes solvent penetration and enhances mass transfer of the bioactive compounds.36 Similarly, the UAE yielded TPC and TFC over 3- and 8-fold higher, respectively, highlighting its efficiency in releasing phenolic compounds that are less accessible during conventional thermal extraction.27,33
Antioxidant activity assays further highlighted the advantages of UAE over conventional extraction. Ultrasound-assisted extracts exhibited lower DPPH IC50 values and significantly (p < 0.05) higher ABTS and FRAP activities than those obtained with the water bath method. For instance, FRAP activity was 2 times higher under UAE conditions, reflecting a greater concentration of redox-active metabolites. These findings align with previous reports that UAE not only enhances antioxidant extraction but also preserves thermolabile compounds that may degrade during prolonged heating.35,37
These results suggest that UAE is a greener, more efficient alternative to conventional extraction, yielding higher phenolic levels and stronger antioxidant activity in shorter extraction times with lower solvent use. This demonstrates potential for industrial applications in the production of functional food and nutraceuticals. The validated RSM models further support this applicability by enabling predictive control of process parameters.
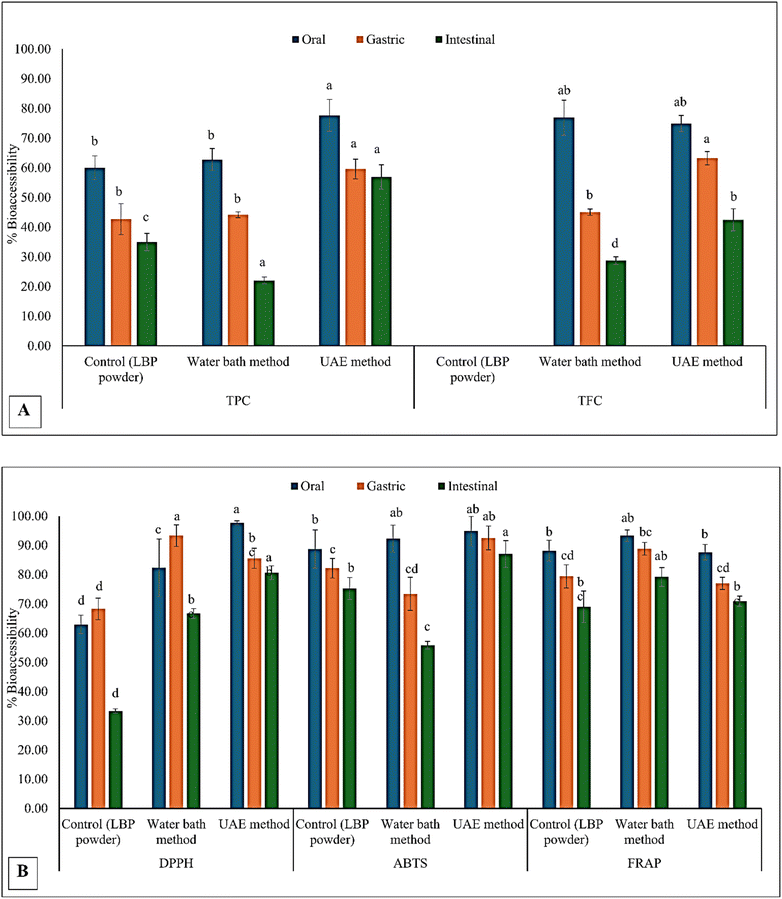
3.4 Stability of compounds and bioactivity under in vitro gastrointestinal conditions
In vitro digestion (Fig. 4) revealed dynamic changes in phenolic content and antioxidant activity across the oral, gastric, and intestinal phases, indicating the influence of digestive conditions on the extract's stability. In general, UAE extracts consistently exhibited the highest bioactive compound and antioxidant activities at all stages, followed by water-bath extracts, while the untreated control remained the lowest. This can be connected to the original contents before digestion, as the values correspond precisely with the results after digestion. Previous studies have suggested that the extraction method influences the bioaccessibility of bioactive compounds during digestion.10,11 The in vitro digested extracts of the samples are presented in Fig. 3.Total phenolic content increased during the gastric phase compared to the oral phase for water bath and control extracts, followed by a significant (p < 0.05) decline in the intestinal phase (Fig. 4). TPC in the UAE extract decreased steadily from the oral (280.80 mg GAE/100 g) to the gastric (236.57 mg GAE/100 g DW) and intestinal phases (153.93 mg GAE/100 g DW), unlike the control and water bath extracts, which increased during the gastric stage. This difference reflects the extraction mechanism: ultrasound disrupts cell walls and liberates free phenolics during processing, leaving fewer bound compounds to be released under acidic gastric conditions38 observed from a study on black chokeberry extract. However, these free phenolics are also more susceptible to acid-induced degradation, accounting for the observed decline in the gastric phase.39 In the intestinal phase, the alkaline pH and bile salts accelerate oxidation and structural transformation. Phenolic acids such as gallic, caffeic, and chlorogenic acids are particularly unstable under these conditions.40 Proanthocyanidins, another polyphenol found in LBP, also undergo rapid degradation under intestinal conditions, which explains the more pronounced TPC reduction in UAE extracts compared to the other treatments.41 Interestingly, total flavonoid content (TFC) increased progressively across the digestion phases, particularly in UAE extracts (from 99.03 to 208.33 mg CE/100 g DW). This suggests that flavonoid glycosides undergo enzymatic or pH-induced hydrolysis, liberating aglycone forms that are more readily detected during intestinal digestion.40,41
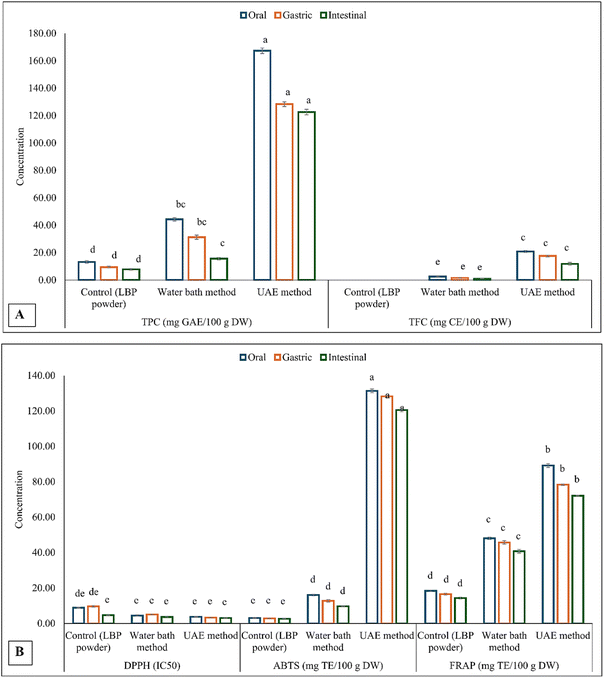 | ||
| Fig. 4 (A) Bioactive compounds and (B) antioxidant activity in vitro digestion. Note: TPC: total phenolic compounds; total flavonoid compounds; GAE: gallic acid equivalent; CE: catechin equivalent. | ||
The antioxidant assays corroborated the phenolic trends, with DPPH radical scavenging (expressed as IC50) generally improving during digestion. UAE extracts exhibited the strongest activity, with an IC50 of 2.43 at the intestinal phase. This could be due to the release of more minor, more active phenolic metabolites during digestion. ABTS and FRAP values increased progressively across the digestive phases, with UAE extracts reaching significantly higher levels (p < 0.05) in the intestinal phase (1313.60 mg TE/100 g DW and 538.44 mg TE/100 g DW, respectively). This trend suggests that the intestinal breakdown of complex phenolics generates metabolites with enhanced radical-scavenging and reducing power, as observed in black and green tea phenolic extracts.42 This agrees with prior reports indicating that in vitro digestion can convert polymeric phenolics into smaller, bioaccessible compounds with higher antioxidant potential.39 The results for pH variations throughout the in vitro digestive process are shown in Table 7.
| Treatment | Digestion | pH | |||||
|---|---|---|---|---|---|---|---|
| 0 min | 10 min | 30 min | 60 min | 90 min | 120 min | ||
| a LBP: longan byproduct; ND: not determined. | |||||||
| Control (LBP powder) | Oral | 6.70 ± 0.00 | 5.92 ± 0.01 | ND | ND | ND | ND |
| Gastric | 1.20 ± 0.00 | ND | 1.34 ± 0.01 | 1.47 ± 0.00 | 1.66 ± 0.01 | 1.63 ± 0.00 | |
| Intestinal | 7.00 ± 0.00 | ND | 7.38 ± 0.02 | 7.34 ± 0.01 | 7.59 ± 0.01 | 7.76 ± 0.02 | |
| Water bath method | Oral | 6.70 ± 0.00 | 5.83 ± 0.02 | ND | ND | ND | ND |
| Gastric | 1.20 ± 0.00 | ND | 1.48 ± 0.02 | 1.52 ± 0.00 | 1.60 ± 0.01 | 1.59 ± 0.00 | |
| Intestinal | 7.00 ± 0.00 | ND | 7.16 ± 0.02 | 7.66 ± 0.02 | 7.51 ± 0.00 | 7.63 ± 0.01 | |
| UAE method | Oral | 6.70 ± 0.00 | 5.92 ± 0.01 | ND | ND | ND | ND |
| Gastric | 1.20 ± 0.00 | ND | 1.43 ± 0.01 | 1.37 ± 0.01 | 1.46 ± 0.00 | 1.54 ± 0.00 | |
| Intestinal | 7.00 ± 0.00 | ND | 7.23 ± 0.02 | 7.35 ± 0.02 | 7.57 ± 0.02 | 7.55 ± 0.02 | |
The superior stability and bioaccessibility of UAE extracts throughout digestion underscore the advantages of ultrasound-assisted extraction in producing phenolic-rich fractions that withstand gastrointestinal conditions. Compared with water bath extracts, the UAE extract not only exhibited higher initial phenolic concentrations but also retained greater activity after intestinal digestion. Similar observations have been reported by Iftikhar et al.,43 for phenolic compounds from rye bran, supporting the robustness of ultrasound-assisted techniques across diverse matrices. These findings collectively suggest that the UAE facilitates the release of phenolic compounds with enhanced resilience to digestive degradation.
The improved stability of UAE extracts may be attributed to multiple factors. First, ultrasound minimizes thermal exposure during processing, reducing the risk of phenolic oxidation or structural alteration compared with conventional heating.44 Second, the mechanical cavitation generated during sonication enhances the solubilization of bound phenolics, thereby increasing their extractability and potential bioaccessibility.45 These advantages suggest that the UAE is a green and effective strategy for extracting fruit byproducts into bioactive-rich ingredients with potential applications in functional foods and nutraceuticals. Fig. 5 shows the bioaccessibility of bioactive compounds and antioxidants.
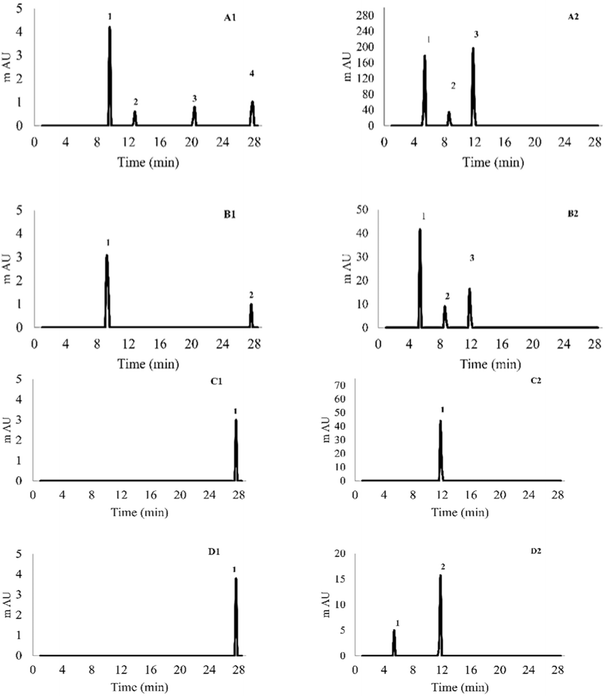
3.5 HPLC analysis of the UAE extract after stimulated digestion
The HPLC chromatogram (Fig. 6) showed that the identified compounds were present in the digested UAE longan byproduct extract under the used conditions. Using established standard curves, the concentrations of these compounds (gallic acid, epicatechin, quercetin, ellagic acid, procyanidin A2, corilagin, and pinostrobin) were quantified at various stages of digestion (oral, gastric and intestinal), including the non-extract sample (Fig. 6A–D) within a retention time from 5 to 28 min and at different absorbances of 254, 270, and 280 nm. Ellagic acid shows the highest peak height across all samples, with a retention time of 11.753–11.776 min, indicating a higher content of the compound than the other available compounds at all stages of digestion. Moreover, quercetin and procyanidin A2 peaks are not present in all digestion stages except the orally digested extract. The intestinal digestion stage has shown only two peaks (ellagic acid and pinostrobin). Zhang et al.29 detected quercetin at 350 nm in longan pulp using a mobile phase of 0.4% acetic acid and acetonitrile with a 45–50 min gradient flow (solution B 35–50%). These were unlike the analytical conditions used in this study, in which quercetin was not detected, likely due to differences in polarity and chromatographic elution strength (Table 8).| Compound | Slope | Intercept | Residual SD | LOD (µg mg−1) | LOQ (µg mg−1) | R 2 |
|---|---|---|---|---|---|---|
| a LOD = limit of detection; LOQ = limit of quantification; SD = standard deviation. | ||||||
| Gallic acid | 13.1 | 377.09 | 332.9 | 84 | 254.09 | 0.9952 |
| Ellagic acid | 0.29 | −3.73 | 14.71 | 169 | 512.82 | 0.9807 |
| Epicatechin | 3.96 | −421.90 | 116.82 | 97 | 295.16 | 0.9935 |
| Corilagin | 8.04 | −465.07 | 144.95 | 60 | 180.29 | 0.9976 |
| Procyanidin A2 | 2.92 | −339.30 | 26.14 | 30 | 89.45 | 0.9994 |
| Quercetin | 21.1 | 62.24 | 63.4 | 9.9 | 30.11 | 0.9997 |
| Pinostrobin | 6.9 | 472.61 | 149.09 | 71 | 216.1 | 0.981 |
Previous investigations have consistently identified ellagic acid, corilagin, and gallic acid as the major phenolic compounds in longan seeds and peels.2,5–7 Notably, Bai et al.2 identified only three phenolics (gallic acid, corilagin, and ellagic acid) within the range of 0.1–10 mg per g dry longan pericarp powder, which corresponds well with the core compounds found in this study, confirming their predominance in longan matrices. Bai et al.2 quantified ellagic acid (17.37 mg kg−1), corilagin (5.25 mg kg−1), and quercetin (3.12 mg kg−1) in purified longan pericarp extracts, while Chindaluang and Sriwattana32 quantified substantially higher levels of corilagin (35.62 mg g−1), ellagic acid (7.02 mg g−1), and gallic acid (16.55 mg g−1) in ultrasound-assisted extracts of longan seed. Similarly, Tang et al.46 reported ellagic acid contents of 0.18 mg g−1 in longan seed, nearly fivefold higher than that in the pericarp (0.83 mg g−1), reinforcing the current finding that ellagic acid is the predominant phenolic compound. However, the observed quantitative differences can be attributed to variations in extraction solvents, chromatographic conditions, plant part analyzed, and the digestion conditions (Table 9).
| Compound name | Oral digested extract | Gastric digested extract | Intestinal digested extract | Non-extract powder |
|---|---|---|---|---|
| a nd and NA means not detected and not available, respectively. Values with different superscripts within a row are significantly different (p < 0.05). | ||||
| Gallic acid | 45.94 ± 2.38 | nd | NA | nd |
| Corilagin | 164.08 ± 1.04a | 138.34 ± 2.58b | NA | NA |
| Ellagic acid | 885.12 ± 0.72a | 119.22 ± 1.15bc | 208.53 ± 1.55b | 70.69 ± 0.61c |
| Epicatechin | 219.78 ± 2.89b | 227.14 ± 2.11a | NA | NA |
| Quercetin | nd | NA | NA | NA |
| Procyanidin A2 | 241.18 ± 7.38 | NA | NA | NA |
| Pinostrobin | nd | nd | nd | nd |
The reduction in phenolic diversity and concentration observed across digestive stages aligns with the findings of Fang et al.,47 who reported that 5-hydroxymethylfurfural (5-HMF) levels in dried longan increased during oral digestion but declined significantly (p < 0.05) after gastric and intestinal digestion. Moreover, 5-HMF can undergo biotransformation into sulfonylmethylfurfural, a genotoxic compound, during in vitro digestion,47 highlighting the complexity of gastrointestinal transformations. Similarly, Bao et al.48 found that although phenolic content and antioxidant activity decreased in digested Tartary buckwheat, the remaining antioxidant potential was still considerable, suggesting that degradation or transformation products retain functional bioactivity. Furthermore, Kessy et al.49 demonstrated that hydrolytic enzymes such as hydropectinase, β-glucosidase, and tannase promote the release and transformation of phenolics in litchi pericarps, processes likely analogous to those occurring in longan digestion.
The persistence of ellagic acid and corilagin throughout digestion may indicate greater structural stability and potential bioaccessibility than those of other phenolics. Corilagin, in particular, exhibits broad pharmacological activities, including antifungal effects against Candida glabrata, antihypertensive action, and cardiovascular protection.46 Additionally, biotransformation during digestion can generate novel metabolites with enhanced or distinct bioactivities,50 implying that gastrointestinal modification of longan phenolics may not solely result in degradation but could also yield beneficial derivatives.
These findings suggest that ellagic acid and corilagin are not only key phenolics in LBP but also among the most stable under simulated digestive conditions. The observed compound-specific variability highlights the importance of gastrointestinal biotransformation in modulating the bioaccessibility and potential bioactivity of longan-derived phenolics.
4 Conclusions
This study optimized the extraction of phenolic-rich compounds from longan industrial byproducts by comparing conventional water-bath-assisted and ultrasound-assisted extraction methods. Response surface methodology using a Box–Behnken design revealed significant differences (p < 0.05), with UAE achieving the highest desirability (D = 0.93). Furthermore, simulated gastrointestinal digestion demonstrated that phenolics from the extract endured the stress and remained bioaccessible, with UAE extracts exhibiting the highest retention of antioxidant activity, suggesting greater potential for in vivo functionality. Although intestinal digestion reduced TPC, the concurrent increase in antioxidant capacity implies functional transformation of polyphenols rather than degradation. Finally, these results uncovered the potential of the UAE method as a green, efficient, and potentially scalable strategy for valorizing longan industrial byproducts into functional food and nutraceutical ingredients. The HPLC profile of the UAE extract showed that compounds were available at the initial stage of digestion, whereas most became unavailable. Future investigations should first focus on encapsulating the phenolic-rich extract to improve targeted bioavailability and clinically validate its health-promoting effects.Author contributions
Idris Kaida Zubairu: investigation, data curation, conceptualization, writing – original draft. Noppol Leksawasdi: writing – review & editing. Sutee Wangtueai: writing – review & editing. Pinpanit Boonchuay: writing – review & editing. Fei Lao: writing – review & editing. Juan Manuel Castagnini: writing – review & editing. Su Lwin Htike: writing – review & editing. Yuthana Phimolsiripol: supervision, conceptualization, writing – review & editing.Conflicts of interest
The authors declare no known competing financial or personal interests that could have influenced the work reported in this article.Data availability
Data is available from the corresponding author upon reasonable request. The cited references in the supplementary information (SI) are provided in the reference list section. Supplementary information: additional figures and detailed statistical analyses supporting the main findings of this study; the proportion of longan raw materials used, contour plots and Pareto charts, response surface regression models, coded coefficients, analysis of variance (ANOVA), regression equations, model summaries, diagnostic tests, and response optimization results for extraction yield, total phenolic content (TPC), total flavonoid content (TFC), and antioxidant activities (DPPH, ABTS, and FRAP); a more comprehensive description of the HPLC method. See DOI: https://doi.org/10.1039/d5fb00879d.Acknowledgements
The authors thank the Center of Excellence in Agro Bio-Circular-Green Industry, Chiang Mai University, for technical support. In addition, the authors are grateful for the financial support from the “Fundamental Fund”, “Reinventing University Program” and the “International Research Fellowship” of Chiang Mai University. The first author is a PhD student in the Division of Food Science and Technology, Faculty of Agro-Industry, Chiang Mai University, under the CMU Presidential Scholarship.References
- T. Sawasdee, Factors behind the increased yield of six Thai fruits in 2025: Market linkages and value enhancement, https://thailand.go.th/useful-information-detail/-6---68-- Search PubMed.
- X. Bai, R. Pan, M. Li, X. Li and H. Zhang, Molecules, 2019, 24, 619 CrossRef PubMed.
- Y. Hu, X. Zhang, D. Li, C. Ma, L. Dong, Y. Luo, X. Hu and F. Chen, Food Chem., 2025, 145985 CrossRef CAS PubMed.
- Z. ke, S. Tan and S. Shi, Anal. Sci., 2023, 39, 1405–1412 CrossRef CAS PubMed.
- S. Tan, Z. Ke, C. Zhou, Y. Luo, X. Ding, G. Luo, W. Li and S. Shi, Molecules, 2023, 28, 2083 CrossRef CAS PubMed.
- K. Rakariyatham, D. Zhou, N. Rakariyatham and F. Shahidi, J. Funct. Foods, 2020, 67, 103846 CrossRef CAS.
- I. K. Zubairu, K. Rakariyatham, S. Bai-Ngew, N. Leksawasdi, J. M. Regenstein, F. Lao, H. Hong, W.-S. Shin, K. J. Alzahrani and Y. Phimolsiripol, Curr. Nutr. Rep., 2025, 14, 28 CrossRef PubMed.
- S. Sai-Ut, P. Kingwascharapong, Md. A. R. Mazumder and S. Rawdkuen, Foods, 2023, 12, 2827 CrossRef CAS PubMed.
- W. Qin, S. Ketnawa and Y. Ogawa, Food Sci. Hum. Wellness, 2022, 11, 669–675 Search PubMed.
- S. Nisa, Y. Bibi, S. Masood, A. Ali, S. Alam, M. Sabir, A. Qayyum, W. Ahmed, S. Alharthi, E. Y. Santali, S. A. Alharthy, W. M. Bawazir and M. N. Almashjary, Molecules, 2022, 27, 7932 Search PubMed.
- M. Rudzińska, A. Grygier, G. Knight and D. Kmiecik, Foods, 2024, 13, 1814 CrossRef PubMed.
- M. Xu, L. Ran, N. Chen, X. Fan, D. Ren and L. Yi, Food Chem., 2019, 297, 124970 CrossRef CAS PubMed.
- M. Fikry, S. Jafari, K. A. Shiekh, I. Kijpatanasilp, S. Khongtongsang, E. Khojah, H. Aljumayi and K. Assatarakul, Ultrason. Sonochem., 2024, 108, 106949 CrossRef CAS PubMed.
- G. S. Seling, R. C. Rivero, C. V. Sisi, V. M. Busch and M. P. Buera, Foods, 2025, 14, 2927 CrossRef CAS PubMed.
- D. T. Ayele, M. L. Akele and A. T. Melese, BMC Chem., 2022, 16, 30 CrossRef CAS PubMed.
- N. Chaiwong, M. Gavahian, P. Chompoorat Tridtitanakiat, P. Therdtatha, C. Moukamnerd, N. Leksawasdi and Y. Phimolsiripol, Innovative Food Sci. Emerging Technol., 2025, 104, 104118 Search PubMed.
- A. Chailangka, N. Autsavapromporn, S. Karnjanapratum, N. Leksawasdi, J. M. Castagnini, F. J. Barba, A. Mousavi Khaneghah and Y. Phimolsiripol, Food Hydrocolloids, 2024, 146, 109288 CrossRef CAS.
- K. Natungnuy, P. P. Chareonsap and S. Poeaim, Int. J. Agric. Technol., 2018, 14, 1505–1514 Search PubMed.
- K. V. Mahindrakar and V. K. Rathod, Chem. Eng. Process., 2020, 149, 107841 Search PubMed.
- D. Cao, X. Qiao, Y. Guo and P. Liu, Food Chem.:X, 2024, 22, 101500 Search PubMed.
- E. Brglez Mojzer, M. Knez Hrnčič, M. Škerget, Ž. Knez and U. Bren, Molecules, 2016, 21, 901 CrossRef PubMed.
- Y. M. Muflihah, G. Gollavelli and Y.-C. Ling, Antioxidants, 2021, 10, 1530 CrossRef CAS PubMed.
- J. A. Domínguez-Avila, Foods, 2023, 12, 4205 CrossRef PubMed.
- S. Aryal, M. K. Baniya, K. Danekhu, P. Kunwar, R. Gurung and N. Koirala, Plants, 2019, 8, 96 Search PubMed.
- M. Tourabi, K. Faiz, R. Ezzouggari, B. Louasté, M. Merzouki, M. Dauelbait, M. Bourhia, K. S. Almaary, F. Siddique, B. Lyoussi and E. Derwich, Bioresources and Bioprocessing, 2025, 12, 24 CrossRef PubMed.
- G. Derringer and R. Suich, J. Qual. Technol., 1980, 12, 214–219 CrossRef.
- K.-C. Jan and M. Gavahian, Food Biosci., 2025, 68, 106686 CrossRef CAS.
- Y. R. R. S. Rezende, J. P. Nogueira, T. O. M. Silva, R. G. C. Barros, C. S. D. Oliveira, G. C. Cunha, N. C. Gualberto, M. Rajan and N. Narain, Food Res. Int., 2021, 140, 109869 CrossRef CAS PubMed.
- R. Zhang, S. A. Khan, Y. Lin, D. Guo, X. Pan, L. Liu, Z. Wei, Y. Zhang, Y. Deng and M. Zhang, Int. J. Food Prop., 2018, 21, 746–759 CrossRef CAS.
- P. T. Vo, H. M. N. Chieng and Q. D. Nguyen, Chemical Engineering Transactions, 2024, 108, 13–18 Search PubMed.
- A. K. Adam, T. Sadhu, I. K. Zubairu, S. S. Nassarawa and M. M. Nasiru, J. Food Compos. Anal., 2025, 148, 108300 CrossRef CAS.
- Y. Chindaluang and S. Sriwattana, CMU J. Nat. Sci., 2014, 13(1), 439–448 Search PubMed.
- L. Shen, S. Pang, M. Zhong, Y. Sun, A. Qayum, Y. Liu, A. Rashid, B. Xu, Q. Liang, H. Ma and X. Ren, Ultrason. Sonochem., 2023, 101, 106646 Search PubMed.
- V. Fuangchoom, N. Narkprasom, S. Jaturonglumlert, J. Varith, Y. Unpaprom and K. Narkprasom, Environ. Qual. Manag., 2024, 34(1) DOI:10.1002/tqem.22245.
- K. Kumar, S. Srivastav and V. S. Sharanagat, Ultrason. Sonochem., 2021, 70, 105325 CrossRef CAS PubMed.
- S. A. Siddiqui, A. Ali Redha, M. Salauddin, I. A. Harahap and H. P. V. Rupasinghe, Crit. Rev. Anal. Chem., 2025, 55, 139–160 Search PubMed.
- X. Lin, J. Chen, G. Xiao, Y. Xu, D. Tang, J. Wu, J. Wen and W. Chen, Food Sci. Biotechnol., 2016, 25, 701–706 CrossRef CAS PubMed.
- S. Lachowicz-Wiśniewska, M. Świeca, I. Kapusta, A. Sip and I. Ochmian, Sci. Rep., 2025, 15, 28805 CrossRef PubMed.
- R. B. Cuvas-Limon, P. Ferreira-Santos, M. Cruz, J. A. Teixeira, R. Belmares and C. Nobre, Antioxidants, 2022, 11, 2479 CrossRef CAS PubMed.
- Y. Tang, W. Liu, J. Zhang, B. Juan, Y. Zhu, L. Zhu, Y. Zhao, M. Daglia, X. Xiao and Y. He, Nutrients, 2025, 17, 2598 Search PubMed.
- J. N. S. Souza, T. Tolosa, B. Teixeira, F. Moura, E. Silva and H. Rogez, Molecules, 2022, 28, 66 CrossRef PubMed.
- G. Annunziata, M. Maisto, C. Schisano, R. Ciampaglia, P. Daliu, V. Narciso, G. C. Tenore and E. Novellino, Nutrients, 2018, 10, 1711 CrossRef PubMed.
- M. Iftikhar, H. Zhang, A. Iftikhar, A. Raza, N. Begum, A. Tahamina, H. Syed, M. Khan and J. Wang, LWT--Food Sci. Technol., 2020, 134, 110243 CrossRef CAS.
- H. V. Annegowda, R. Bhat, L. Min-Tze, A. A. Karim and S. M. Mansor, J. Food Sci. Technol., 2012, 49, 510–514 Search PubMed.
- R. P. Putra, S. I. Aisyah, P. A. Kurniatin and W. Nurcholis, Trop. J. Nat. Prod. Res., 2024, 8, 6552–6557 CrossRef CAS.
- Y.-Y. Tang, X.-M. He, J. Sun, C.-B. Li, L. Li, J.-F. Sheng, M. Xin, Z.-C. Li, F.-J. Zheng, G.-M. Liu, J.-M. Li and D.-N. Ling, Molecules, 2019, 24, 1186 Search PubMed.
- R. Fang, W. Sang, Q. Cheng and S. Liu, Food Sci., 2024, 45, 41–48 CAS.
- T. Bao, Y. Wang, Y. Li, V. Gowd, X. Niu, H. Yang, L. Chen, W. Chen and C. Sun, J. Zhejiang Univ., Sci., B, 2016, 17, 941–951 CrossRef CAS PubMed.
- H. N. E. Kessy, K. Wang, L. Zhao, M. Zhou and Z. Hu, LWT--Food Sci. Technol., 2018, 87, 301–309 CrossRef CAS.
- X. Zhu, H. Wang, J. Sun, B. Yang, X. Duan and Y. Jiang, J. Zhejiang Univ., Sci., B, 2019, 20, 503–512 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |