Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Freezing as a structuring process for protein gels: process–structure relationship, material characterization, and comparison with meats
Israel Jovino Caneschi *a,
Daniel Angelo Longhi
*a,
Daniel Angelo Longhi b,
Charles Windson Isidoro Haminiuk
b,
Charles Windson Isidoro Haminiuk c and
Maria Lucia Masson
c and
Maria Lucia Masson a
a
aPostgraduate Program in Food Engineering, Federal University of Paraná (UFPR), Polytechnic Centre, Curitiba, Paraná CEP 81531-980, Brazil. E-mail: israeljcaneschi@gmail.com
bPostgraduate Program in Food Engineering, Federal University of Paraná (UFPR), Jandaia do Sul Campus, Jandaia do Sul, Paraná CEP 86900-000, Brazil
cPostgraduate Program in Food Engineering, Biotechnology Laboratory, Federal Technological University of Paraná (UTFPR), Curitiba, Paraná CEP 81280-340, Brazil
First published on 11th February 2026
Abstract
The growing demand for alternative proteins arises from population growth and the pursuit of innovative, sustainable products. Protein gels stand out among these alternatives due to their ability to replicate meat-like sensory properties using proteins such as Soy Protein Isolate (SPI). This study evaluated the production of structured SPI gels via freeze structuring. The variables solids content (X1), pH (X2), and freezing temperature (X3) were analysed in relation to the responses hardness (Y1) and cohesiveness (Y2), using a two-level, three-factor full factorial design with a duplicate at the centre point. The results revealed a significant effect of all main effects and they include the interactions solids content × pH and solids content × freezing temperature for Y1, while for Y2, the influential effects were solids content, pH, and the interactions solids content × pH and pH × freezing temperature. The Texture Profile Analysis (TPA) and colour of the gels were comparable to those of sirloin steak, chicken breast, and fish fillet. Gels exhibited acidity between 0.079 and 0.167 g of lactic acid/100 g, pH from 5.54 to 6.54, a water activity (aw) of 0.992 to 0.996, and a moisture content of 63.35% to 69.84%. Treatments S6 (13% solids, pH 5.0, −5 °C), S8 (13% solids, pH 6.0, −5 °C), and S9 (9% solids, pH 5.5, −10 °C) exhibited lamellar structures and a Degree of Texturization (DT) above 1, suggesting unidirectional freezing, confirmed by freezing curves. Overall, freeze structuring proved to be a promising technique for producing structured SPI gels, with optimal parameters being 9% solids, pH 5.5, and −10 °C.
Sustainability spotlightThe demand for affordable and high-quality products increases with global population growth; it can cause environmental impacts and raises ethical concerns. The use of plant proteins and the development of meat analogues represent environmentally friendly alternatives to livestock products. In this context, a protein gel was produced from soy protein isolate, a residual material recovered during legume processing. The process uses an innovative freeze alignment technique that promotes protein rearrangement and organization to achieve meat-like textures. Low freezing rates and limited material requirements enhance reproducibility and contribute to sustainability, in accordance with Sustainable Development Goals 2, 9, and 13. |
1. Introduction
The accelerated growth of the global population, concerns about animal welfare, and the environmental impact caused by livestock production, particularly regarding the exploitation of natural resources such as water and soil and the emission of greenhouse gases,1–4 have influenced consumers to reduce or eliminate meat consumption. Consequently, the demand for alternative and more sustainable protein sources has increased, driving the development of plant-based products.Among the available plant sources, soybean stands out due to its wide availability, low cost, and high protein functionality, presenting properties such as solubility, emulsification, and gelation.5 Preece et al.6 explained that their protein derivatives (soy flour, concentrates, and isolates) are often obtained as co-products from oil extraction and exhibit great potential for the development of textured protein materials.7–9
Meat is a complex raw material composed of aligned myofibrils forming fibres, which are organised into muscles.10 This assembly provides meat with its typical structural anisotropy, a fundamental aspect when attempting to reproduce its texture. To replicate this fibrous morphology in meat analogues, several structuring technologies have been proposed and developed. Among these, the freeze alignment technique stands out for enabling control of the protein orientation during processing. This method is based on the freezing of a binary solution of water and biopolymers, resulting in the formation of plate-shaped ice crystals that promote the alignment and reorganisation of soybean globular proteins, generating a porous and fibrous structure similar to that of muscle tissue. Heat removal must occur unidirectionally to ensure the formation of the desired anisotropic structure.11,12 Despite previous reports on the use of freeze alignment for protein structuring, its application under simplified freezing conditions and using low-complexity formulations remains insufficiently explored.
Given that the performance of the freeze alignment process directly influences product quality, it is essential to parameterise its stages to ensure the desired properties and reproducibility. For this purpose, experimental methodologies allow the simultaneous evaluation of the effects of different factors on parameters of interest. The Full Factorial Design (FFD) is a simple and efficient approach capable of identifying the factors that affect material formation and statistically analysing interactions between multiple parameters within a specific range of values.13,14 In this study, texture was adopted as the response variable, as it represents a key property both for the development of novel food materials and for quality control during production, being one of the main characteristics to be controlled in structured products. According to Di Monaco et al.,15 the study of texture involves the analysis of the mechanical properties of the material, which can be determined by different instrumental methods.
This study investigates the effect of process variables on quality parameters of meat analogue, plant-based gels. The use of formulations composed exclusively of plant protein and a gelling agent, in combination with freezing performed in a single step in a domestic freezer suggests that a cost-effective meat analogue could be obtained. The present work contributes to the United Nations Sustainable Development Goals16 by proposing the use of freeze alignment technology in the production of meat analogues based on soy protein isolate, promoting access to sustainable protein sources and responsible production. Therefore, the aim of this study was to evaluate the effect of solids content, pH, and freezing temperature on the texture of the resulting protein gels. The physicochemical properties of the product were also examined, enabling its characterisation in terms of quality and preservation capacity, as well as the comparison of its texture and colour with those of conventional meats.
2. Materials and methods
2.1. Materials
For the development of this study, Soy Protein Isolate (SPI) donated by the company International Flavors & Fragrances – IFF was used. Its composition on a wet basis was 6.16% moisture, 4.65% ash, 0.19% lipids, 79.13% protein, and 9.87% carbohydrates, along with a solubility in water of 8.88%. Sodium alginate PA (SA) and calcium chloride PA (CaCl2) were acquired from Êxodo Científica, and lactic acid (85%) from Nuclear. Cylindrical moulds made of expanded polystyrene with dimensions of 110 × 68 × 82 mm (height, internal diameter, and external diameter, respectively) were also used.2.2. Methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 ratio (SPI
0.5 ratio (SPI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) SA), along with water, and the pH was adjusted by adding lactic acid. The formulated product was then transferred to expanded polystyrene moulds and subjected to freezing for 36 hours in a freezer (Electrolux PROSDOCIMO H40C). Following freezing, the samples were stabilized with steam in an autoclave (HOENIX brand), operating at 137.29 kPa, cooled to room temperature (20 °C), and subsequently prepared for the relevant analyses.
SA), along with water, and the pH was adjusted by adding lactic acid. The formulated product was then transferred to expanded polystyrene moulds and subjected to freezing for 36 hours in a freezer (Electrolux PROSDOCIMO H40C). Following freezing, the samples were stabilized with steam in an autoclave (HOENIX brand), operating at 137.29 kPa, cooled to room temperature (20 °C), and subsequently prepared for the relevant analyses.
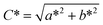 | (1) |
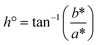 | (2) |
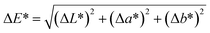 | (3) |
| Standard order | Random order | Solids content (%) | pH (—) | Freezing temperature (°C) | Hardness (N) | Cohesiveness (—) |
|---|---|---|---|---|---|---|
| X1 | X2 | X3 | Y1 | Y2 | ||
| a The results are presented as mean ± standard deviation (SD), based on three replicates. The statistical significance of the terms used to derive mathematical models (presented in Section 3.6) was evaluated using the Student's t-test at a 95% confidence level (α = 0.05). | ||||||
| 1 | 8 | 5 (−1) | 5 (−1) | −15 (−1) | 81.62 ± 2.30 | 0.65 ± 0.01 |
| 2 | 2 | 13 (1) | 5 (−1) | −15 (−1) | 83.76 ± 2.19 | 0.63 ± 0.02 |
| 3 | 3 | 5 (−1) | 6 (1) | −15 (−1) | 50.26 ± 0.52 | 0.70 ± 0.01 |
| 4 | 5 | 13 (1) | 6 (1) | −15 (−1) | 98.45 ± 1.06 | 0.64 ± 0.01 |
| 5 | 9 | 5 (−1) | 5 (−1) | −5 (1) | 74.94 ± 1.93 | 0.62 ± 0.01 |
| 6 | 10 | 13 (1) | 5 (−1) | −5 (1) | 125.42 ± 8.12 | 0.63 ± 0.03 |
| 7 | 1 | 5 (−1) | 6 (1) | −5 (1) | 44.65 ± 2.73 | 0.71 ± 0.04 |
| 8 | 4 | 13 (1) | 6 (1) | −5 (1) | 127.16 ± 3.12 | 0.68 ± 0.01 |
| 9 | 7 | 9 (0) | 5.5 (0) | −10 (0) | 93.06 ± 2.06 | 0.66 ± 0.02 |
| 10 | 6 | 9 (0) | 5.5 (0) | −10 (0) | 91.86 ± 4.88 | 0.67 ± 0.02 |
The responses obtained were determined experimentally, and the first-order equation presented in eqn (4) was fitted to the experimental data to estimate its coefficients.
 | (4) |
Process variables and their levels were established based on preliminary studies and the literature: X1 – solids content (5% and 13%); X2 – pH (5.0 and 6.0); and X3 – freezing temperature (−15 °C and −5 °C). The selection of X1 considered processing feasibility, as higher concentrations hindered homogenization and pH adjustment. For X2, the range sought to align with typical values for red meat and poultry (5.5 to 5.9)25 and Brazilian freshness standards for fish (pH < 7.0), according to national legislation.26 Freezing temperatures (X3) were set to ensure a slow-freezing regime, accounting for the small sample dimensions and similar meat analogue studies.8,17 The centre point was defined at 9%, 5.5, and −10 °C, and the full factorial design comprised 10 randomized treatments to eliminate potential bias.27 Statistical analysis was conducted at a 95% confidence level (p < 0.05), employing linear regression and ANOVA to assess main and secondary effects.
Non-significant interactions were removed, and the model was re-tested. Once the model proved adequate based on R2 and adjusted R2, it was established and presented in this work.
3. Results and discussion
3.1. Colour and texture characteristics in relation to meat standards
The Texture Profile Analysis (TPA) results for the protein gels and the three reference meats are presented in Table 2. According to Jonkers et al.,28 hardness is directly related to a material's resistance to deformation, serving as an indicator of toughness and maturity in meat products. In this study, the treatments resulted in products with hardness ranging from 44.65 N (S7) to 127.16 N (S8), and Tukey's test was applied to compare these values with those of the reference meat products. Among the evaluated meats, the fish fillet showed the lowest hardness (24.04 N), differing significantly from all treatments. Chicken breast (71.41 N) was statistically similar to samples S5 (74.94 N), S1 (81.62 N), and S2 (83.76 N), while beef striploin (54.17 N) was equivalent to S3 (50.26 N) and S7 (44.65 N). These findings support the model generated by the factorial design, which showed that samples with lower solids content, such as S1, S5, S3, and S7, yielded softer textures. In this regard, samples S8 and S6, which were prepared with a higher solidscontent and at a higher freezing temperature, exhibited the highest hardness among the treatments, differentiating them from the evaluated meat products. Furthermore, materials with a high gelling capacity tend to exhibit greater hardness, as is the case with SPI. Chantanuson et al.8 demonstrated in their studies that hardness is influenced by both the solids content and the gelling capacity of the soy protein. They also explained that this capacity is directly related to the solubility of the raw material: the higher the solubility, the greater the gelling capacity. Thus, considering that the solubility obtained for the SPI used in this study was 8.88%, a high value compared to the results obtained by Chantanuson et al.,7 it is believed that the gelling property contributed to the increased hardness of the resulting product.| Standard order | Hardness (N) | Cohesiveness | Springiness | Chewiness (N) |
|---|---|---|---|---|
| a The results are presented as mean ± standard deviation (SD), based on three replicates. Means followed by different letters indicate statistically significant differences (p < 0.05) within the same column for each evaluated parameter. The trials were numbered from 1 to 10, with the combination of factors (solids content (X1) in %; pH (X2); freezing temperature (X3) in °C) for each one being as follows: S1 (5; 5.0; −15), S2 (13; 5.0; −15), S3 (5; 6.0; −15), S4 (13; 6.0; −15), S5 (5; 5.0; −5), S6 (13; 5.0; −5), S7 (5; 6.0; −5), S8 (13; 6.0; −5), S9 (9; 5.5; −10), and S10 (9; 5.5; −10). | ||||
| 1 | 81.62 ± 2.30CD | 0.65 ± 0.01ABC | 0.83 ± 0.00A | 44.02 ± 0.85EF |
| 2 | 83.76 ± 2.19CD | 0.63 ± 0.02C | 0.88 ± 0.03A | 46.76 ± 1.44DE |
| 3 | 50.26 ± 0.52E | 0.70 ± 0.01AB | 0.88 ± 0.03A | 31.35 ± 1.09G |
| 4 | 98.45 ± 1.06B | 0.64 ± 0.01BC | 0.85 ± 0.02A | 53.58 ± 1.71C |
| 5 | 74.94 ± 1.93D | 0.62 ± 0.01CD | 0.84 ± 0.01A | 38.72 ± 1.98F |
| 6 | 125.42 ± 8.12A | 0.63 ± 0.03C | 0.86 ± 0.02A | 67.26 ± 5.88B |
| 7 | 44.65 ± 2.73E | 0.71 ± 0.04A | 0.86 ± 0.06A | 27.38 ± 1.83GH |
| 8 | 127.16 ± 3.12A | 0.68 ± 0.01ABC | 0.90 ± 0.03A | 77.63 ± 0.19A |
| 9 | 93.06 ± 2.06BC | 0.66 ± 0.02ABC | 0.90 ± 0.01A | 55.64 ± 1.38C |
| 10 | 91.86 ± 4.88BC | 0.67 ± 0.02ACB | 0.85 ± 0.02A | 52.34 ± 1.32CD |
| Chicken breast | 71.41 ± 5.80D | 0.24 ± 0.03E | 0.43 ± 0.03C | 7.54 ± 0.57I |
| Fish fillet | 24.04 ± 1.35F | 0.63 ± 0.01C | 0.83 ± 0.04A | 12.54 ± 0.08I |
| Beef sirloin | 54.17 ± 9.22E | 0.55 ± 0.01D | 0.72 ± 0.01B | 21.46 ± 3.39H |
Springiness results showed no significant differences among treatments at the 5% level, with values ranging from 0.83 (S1) to 0.90 (S8). All samples had elasticity values comparable to fish fillet (0.83) but differed significantly from those of striploin (0.72) and chicken breast (0.43). These values suggest that the material tends to break into larger fragments upon compression.29
Cohesiveness reflects the internal bonding of the material and indicates its structural integrity under mechanical stress.30 High cohesiveness values imply stronger intermolecular forces and a more crosslinked structure.31 Among the meat products, chicken breast (0.24) differed significantly from all treatments. Fish fillet (0.63) differed only from S7 (0.71) and S3 (0.70), while striploin (0.55) was statistically similar to S5 (0.62). In general, the samples showed higher cohesiveness than chicken breast and striploin, indicating a highly crosslinked structure.
Chewiness values were higher in all samples compared to fish fillet (12.54 N) and chicken breast (7.54 N), reaching 77.63 N in sample S8. Striploin (21.47 N) was statistically similar to S7 (27.38 N), which had the lowest chewiness among the treatments. Since chewiness reflects the combined effects of hardness, cohesiveness, and elasticity, these results highlight the strong intermolecular interactions and brittle characteristics of the gels. According to Godschalk-Broers; Sala; Scholten,32 chewiness is directly related to the effort required for mastication. Therefore, the high values indicate that the products analysed in this study offer greater resistance to chewing, requiring more effort to consume compared to the reference meat products.
Soy-based analogues produced by Chiang et al.9 using wheat gluten and high-moisture extrusion had hardness values ranging from 45.40 N to 78.61 N, and chewiness values between 34.02 N and 45.32 N. Another study employing soy and rice protein with low-moisture extrusion reported elasticity values between 0.914 and 0.959 and cohesiveness ranging from 0.466 to 0.539.29 These comparisons demonstrate that the textural properties observed in this study are similar to those found in previous research, reinforcing the influence of processing conditions and formulation on the final structure of meat analogues.
The texture results obtained in this study point to opportunities for improving the formulation and the resulting structure. Increasing the SA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) SPI ratio and incorporating oil into the formulation may help enhance texture and bring it closer to that of conventional meat products. According to Nakagawa et al.,4 enhancing protein–water interactions and adding lipids can promote the development of fibrous textures and improve product appearance. These improvements can be achieved using plant-based polysaccharides such as sugars, fibres, and starch.
SPI ratio and incorporating oil into the formulation may help enhance texture and bring it closer to that of conventional meat products. According to Nakagawa et al.,4 enhancing protein–water interactions and adding lipids can promote the development of fibrous textures and improve product appearance. These improvements can be achieved using plant-based polysaccharides such as sugars, fibres, and starch.
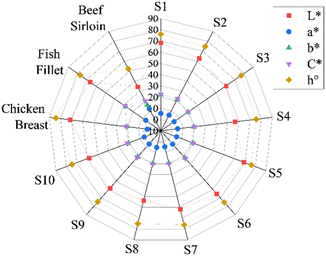
Colour measurements are shown in Fig. 1 and Table S1 in the SI. Mean lightness (L*) values for the treatments ranged from 54.03 (S8) to 68.83 (S5), classifying the samples as light (L* > 50) and indicating higher light reflectance. The a* coordinate (representing the red-green axis) varied between 4.37 (S3) and 5.84 (S4), while the b* coordinate (yellow-blue axis) ranged from 19.18 (S3) to 21.95 (S1), suggesting that the samples displayed colours within the yellow to red spectrum. In a study conducted by Boonarsa et al.,33 soy protein was used as a reference, and their reported values were L* = 74.64, a* = 1.18, and b* = 16.74, also indicating light tones in the yellow-red region. The obtained C* (chroma) ranged from 19.72 (S3) to 22.61 (S1), while h° (hue angle) varied between 74.51° (S10) and 76.34° (S7), indicating a low-intensity colour closer to yellow.19
To assess the similarity or difference between the colour of the samples and that of meat products, the total colour difference (ΔE*) was calculated and is presented in Table 3. Notable colour differences were observed between the grilled beef striploin and the treatments, reflecting both differences in red meat composition and preparation methods. As expected, the ΔE values were high when compared to beef, ranging from 21.31 (S8) to 35.57 (S5). The fish fillet and chicken breast presented lower ΔE values, indicating greater colour similarity with certain samples. Sample S6 exhibited the lowest variation compared to fish (ΔE* = 6.12), while for chicken breast the smallest difference was observed for sample S5 (ΔE* = 3.86).
| Standard order | ΔE* – chicken breast | ΔE* – fish fillet | ΔE* – beef sirloin |
|---|---|---|---|
| a The results are presented as mean ± standard deviation (SD), based on four repetitions. Means followed by different letters within the same column for each evaluated parameter indicate statistically significant differences (p < 0.05). The trials were numbered from 1 to 10, with the combination of factors (solids content (X1) in %; pH (X2); freezing temperature (X3) in °C) for each one being as follows: S1 (5; 5.0; −15), S2 (13; 5.0; −15), S3 (5; 6.0; −15), S4 (13; 6.0; −15), S5 (5; 5.0; −5), S6 (13; 5.0; −5), S7 (5; 6.0; −5), S8 (13; 6.0; −5), S9 (9; 5.5; −10), and S10 (9; 5.5; −10). | |||
| 1 | 4.59 ± 0.21FF | 6.44 ± 0.29F | 35.34 ± 0.34A |
| 2 | 9.15 ± 0.40DE | 7.79 ± 0.33E | 29.88 ± 0.43C |
| 3 | 11.19 ± 1.47C | 11.27 ± 0.58C | 27.56 ± 1.48D |
| 4 | 15.41 ± 1.22B | 13.56 ± 0.90B | 23.39 ± 1.19E |
| 5 | 3.86 ± 0.42F | 8.14 ± 0.34E | 35.57 ± 0.56A |
| 6 | 7.69 ± 0.69E | 6.12 ± 0.23F | 31.84 ± 0.73B |
| 7 | 10.02 ± 0.28CD | 9.99 ± 0.38D | 28.69 ± 0.28CD |
| 8 | 17.61 ± 0.58A | 15.18 ± 0.41A | 21.31 ± 0.52F |
| 9 | 13.72 ± 0.77B | 11.26 ± 0.47C | 25.20 ± 0.61E |
| 10 | 13.73 ± 0.70B | 11.12 ± 0.48CD | 25.21 ± 0.64E |
Colour is a crucial parameter for product acceptance and is directly influenced by composition. Studies by Chiang et al.9 and Lee et al.34 showed that soy-based meat analogues exhibited colour variation when blended with other protein sources, such as wheat gluten and rice protein isolate, respectively. Furthermore, Chantanuson et al.8 employed food colourants, such as Red 102, to achieve a reddish hue similar to raw meat. Thus, soy as a raw material offers broad potential for targeted modifications, due to its naturally light colour between red and yellow, which facilitates approximating the desired colour attributes in meat-like products. Differences in composition between meat products and analogues contribute to the variations observed in this study. Singh et al.,3 in optimising the use of Manila tamarind protein isolate for meat analogue preparation, highlighted that colour differences in comparison with chicken breast were mainly due to compositional differences, since the latter has a higher fat content and lower carbohydrate concentration.
The differences observed in colour parameters (L*, a*, b*) and texture (TPA) between the developed gels and the meats used as references should be interpreted considering the different cooking methods applied. The cooking method, which differs in terms of the heat transfer medium, processing time and applied temperature, directly influences muscle protein transformations and water redistribution within the matrix, thereby affecting the evaluated parameters.35,36 Cooking temperature and time influence the extent of protein denaturation, muscle fibre contraction and water loss,37 impacting colour attributes and the mechanical properties analysed. In addition, grilling, in which heating occurs more rapidly and is concentrated at the surface, promotes greater surface dehydration and intensifies non-enzymatic browning reactions, for example, affecting the final colour of the meat.38 Thus, part of the variations observed in colour and texture results may be attributed not only to the intrinsic characteristics of the evaluated matrices, but also to differences in cooking time, temperature and method. Nevertheless, the results allow the developed gels to be positioned within the range of instrumental properties observed in processed meat products, reinforcing their suitability as meat analogues from a technological perspective.
3.2. Physicochemical characterization of the protein gels
To characterise the obtained product, analyses of titratable acidity, pH, water activity (aw), moisture, and protein content were performed. The samples were labelled according to the standard order established in the full factorial design 23, ranging from S1 to S10. These results are expressed together with the mean comparison analysis using Tukey's test (Table 4).| Standard order | Moisture (%) | aw (—) | Acidity g/100 g lactic acid | pH (—) | Protein (%) |
|---|---|---|---|---|---|
| a The results are expressed as mean ± standard deviation (SD), considering two repetitions for protein content, three for acidity and four for the other parameters. Different letters indicate statistically significant differences (p < 0.05) within the same column for each parameter. The trials were numbered from 1 to 10, with the combination of factors (solids content (X1) in %; pH (X2); freezing temperature (X3) in °C) for each one being as follows: S1 (5; 5.0; −15), S2 (13; 5.0; −15), S3 (5; 6.0; −15), S4 (13; 6.0; −15), S5 (5; 5.0; −5), S6 (13; 5.0; −5), S7 (5; 6.0; −5), S8 (13; 6.0; −5), S9 (9; 5.5; −10), and S10 (9; 5.5; −10). | |||||
| 1 | 69.19 ± 1.73AB | 0.994 ± 0.00AB | 0.16 ± 0.00A | 5.66 ± 0.09C | 25.28 ± 0.02E |
| 2 | 65.61 ± 2.86BCD | 0.992 ± 0.00C | 0.15 ± 0.01A | 5.55 ± 0.01D | 27.78 ± 0.25CD |
| 3 | 69.53 ± 0.73AB | 0.996 ± 0.00A | 0.08 ± 0.00C | 6.54 ± 0.02A | 25.36 ± 0.56E |
| 4 | 64.35 ± 1.64CD | 0.993 ± 0.00BC | 0.10 ± 0.00C | 6.50 ± 0.02A | 29.49 ± 0.02B |
| 5 | 66.79 ± 1.64ABC | 0.995 ± 0.00AB | 0.17 ± 0.01A | 5.67 ± 0.07C | 27.33 ± 0.21D |
| 6 | 63.35 ± 1.85CD | 0.995 ± 0.00AB | 0.16 ± 0.01A | 5.54 ± 0.04D | 30.09 ± 0.19B |
| 7 | 70.74 ± 1.63A | 0.996 ± 0.00A | 0.08 ± 0.01C | 6.50 ± 0.04A | 23.78 ± 0.02F |
| 8 | 61.45 ± 2.46D | 0.996 ± 0.00A | 0.09 ± 0.00C | 6.46 ± 0.02A | 32.92 ± 0.12A |
| 9 | 65.88 ± 1.81BCD | 0.995 ± 0.00AB | 0.12 ± 0.01B | 6.32 ± 0.02B | 28.24 ± 0.05CD |
| 10 | 65.71 ± 1.49BCD | 0.996 ± 0.00A | 0.12 ± 0.01B | 6.33 ± 0.03B | 28.39 ± 0.27C |
The protein gels exhibited protein contents ranging between 23.78% (S7) and 32.92% (S8), with formulations containing lower solids content resulting in lower protein values. However, sample S2 showed a statistically equivalent result to the centre point replicates (S9 and S10), despite having the lowest solids content among them. pH did not demonstrate a significant influence on the protein content, unlike temperature, which impacted the protein levels in formulations with higher solids content (9% and 13%). Under these conditions, samples subjected to lower freezing temperatures (−5 °C and −10 °C) showed higher protein content. This occurs because faster freezing, promoted by lower temperatures, favours the retention of suspended solids. Consequently, during the stabilisation step, there is a greater loss of solids, which affects the final protein content in the material.
The samples displayed titratable acidity ranging between 0.079 and 0.167 g of lactic acid/100 g of the sample. The ANOVA results indicated variation in acidity among the samples, distributed according to the pH adjustment performed during formulation. Therefore, it was expected that the results would be similar for samples adjusted to pH 6.0 (S3, S4, S7, and S8), for those adjusted to pH 5.5 (S9 and S10), and for those adjusted to pH 5.0 (S5, S6, S1, S2), with the highest means observed for samples adjusted to pH 5, where a larger volume of acid was added.
All samples presented an acidic pH, ranging between 5.54 (S6) and 6.54 (S3), with this value being higher than the target pH used in the formulation. Samples adjusted to pH 6.0 resulted in gels with higher pH (6.46–6.54) and lower acidity (0.08–0.10 g of lactic acid/100 g of sample), whereas those adjusted to pH 5.0 showed a lower final pH (5.54–5.67) and higher acidity (0.15–0.17 g of lactic acid/100 g of sample). Since pH indicates the acidity or alkalinity of the medium based on the concentration of hydrogen ions (H+), these results converge with those found for total titratable acidity.
Regarding water activity (aw), the results from the mean comparison test indicated a significant difference among samples, ranging between 0.992 and 0.996. This property is an important quality parameter for food and is related to its stability and safety for consumption. Analysing the degree of water interaction with other components of the food matrix, as well as its availability for reactions and microbial growth, allows for the evaluation of factors that directly influence its deterioration.39 In this regard, foods with a water activity (aw) below 0.85 are considered safe, as they do not favour the activity of pathogenic microorganisms and the production of mycotoxins by fungi.40,41
Finally, moisture content in the samples ranged between 63.35% and 69.84% (S6 and S7, respectively), showing significantly different means at a 5% significance level. Samples with lower solids content exhibited higher moisture, possibly due to a reduced proximity between protein molecules, which favours the formation of protein–water bonds, even with the high availability of free water indicated by the aw values. These results might also be linked to the addition of SA to the formulation, given that carbohydrate polymers, such as alginates derived from seaweed, possess a high water-holding capacity, promoting water incorporation and reducing syneresis.5 Soy protein has been used in various studies as a reference product. Boonarsa et al.33 used freezing as a texturising technology for meat analogues based on silkworm pupae, and for samples containing only soy, the moisture content was 69.59%. Chiang et al.,9 when using high-moisture extrusion to obtain analogue products, reported lower results of 58.01%, which was also less than that of cooked chicken used for comparison in their study.
The results obtained for acidity, water activity (aw) and moisture content are typical of highly perishable products and must be carefully considered when selecting packaging systems and storage conditions. The adoption of preservation strategies widely employed in the food industry, such as refrigeration combined with appropriate packaging systems, including modified atmosphere packaging, has been reported as effective in reducing microbiological and oxidative spoilage in high-aw products, thereby contributing to shelf-life extension without compromising product characteristics.42 Physical processes aimed at partially reducing water activity, such as controlled dehydration, have also been applied as an alternative to limit the availability of free water and improve physicochemical stability during storage.43 Additionally, adjustments to formulation and packaging systems, including the incorporation of compounds with antioxidant and antimicrobial activity directly into the food matrix or into active packaging systems, are described as complementary and effective strategies to retard oxidative reactions and microbial growth in highly perishable foods.44,45 Overall, the combined application of these approaches indicates that, despite the high perishability, there are viable and sustainable technological pathways for the practical application and commercialisation of the developed gels, while maintaining physicochemical properties, protein content and pH comparable to those of conventional meat products.
3.3. Structure assessment
Structural characterisation was performed using analyses of both the moist sample, through digital photography, and the freeze-dried sample, using a magnifying lens and scanning electron microscopy (SEM). Based on the SEM images (Fig. 2), it was possible to examine the microstructure of the material, which revealed a porous morphology in all samples. This was a result of ice crystal formation during the freezing process. However, detailed analysis of this microstructure did not reveal any clear porosity pattern that would suggest specific directional crystal growth or significant differences between samples.Images obtained with the stereomicroscope, shown in Fig. 3, demonstrated differences in the structures resulting from the various treatments. A porous appearance was again observed across all samples. Some treatments, such as S1, S2, S3, S4, and S5, resulted in smaller openings, whereas samples such as S6, S7, S8, and S9 exhibited slightly larger openings. The absence of lamellar structures may be explained by the cutting direction and magnification used. Nonetheless, there was a trend towards the formation of larger openings in treatments conducted at higher temperatures (−5 °C and −10 °C).
Images of the analogues, taken from longitudinal sections, allowed visualisation of the structure on a macroscopic scale (Fig. 4), essential for observing the lamellar morphology. Upon examining the images, it was evident that the structure differed between treatments, ranging from lamellar to porous forms. Samples S1 and S3 were predominantly porous. This outcome was associated with the formation of smaller ice crystals due to the lower processing temperature (−15 °C), combined with the lower solids content (5%). Samples S6, S8, and S9 exhibited predominantly lamellar structures, contrasting with the samples S6 and S8 which were prepared using 13% solids at −5 °C, while S9 was prepared under the central point conditions (9% solids and −10 °C). The combination of these processing parameters proved promising for the formation of the desired fibrous structure, showing better performance compared to the other treatments.
Samples S6, S8, and S9 showed the most promising results, taking into account the aim of the study. Although the macroscopic structure confirmed the formation of lamellar regions in these treatments, the images obtained using SEM and a stereoscope did not allow a definitive conclusion regarding the unidirectional orientation of ice crystal growth. This result stems from the orientation of the cut, made perpendicular to the growth plane of the leaves. Additionally, the high magnification used reduced the field of view, affecting the perception of the parallel arrangement of the layers.
3.4. Cutting force and degree of texturization (DT)
Samples S6, S8, and S9 were assessed for shear force in the vertical (FV) and transverse (FL) directions relative to the fibre orientation (Table S2 in the SI). The recorded ranges were: S6 (3.18–1.61 N), S8 (5.30–2.80 N), and S9 (5.71–2.12 N) in the vertical direction, and S6 (2.26–0.94 N), S8 (4.69–1.65 N), and S9 (4.79–1.12 N) in the transverse direction. These results indicated that sample S8 exhibited the highest resistance to shear, which is consistent with its hardness values, where it also recorded the highest result among the evaluated samples.The ratio between the shear forces in the two directions was calculated to be 1.65 (S6), 1.67 (S8), and 1.45 (S9). According to Chen et al.,23 this ratio serves as an indicator of the DT, with values greater than 1 suggesting the presence of fibrous structure formation. This is due to the higher resistance observed when fibres are aligned in parallel, compared to the perpendicular orientation. Thus, the results confirm the fibrous formation and anisotropic behaviour of the protein gels. Similar findings were reported by Chiang et al.,9 Fang et al.46 and Singh et al.,3 showing degrees of texturization comparable to those observed in this study.
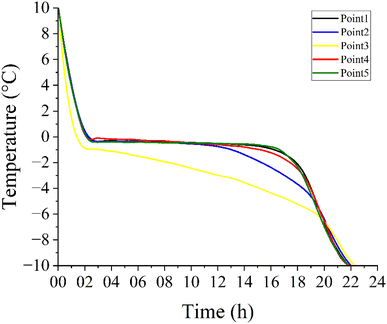
3.5. Freezing curve
The samples were frozen in cylindrical polystyrene moulds, insulated at the base and laterally. Five different points were monitored for temperature during the freezing process of the centre point samples (9% solids, pH 5.5 and temperature of −10 °C). The temperature profiles are shown in Fig. 5, which also identifies the respective locations of the sensors within the cylinder.The freezing curves differed between Point 3 and the other measured points. This variation is attributed to the sample remaining static during freezing, which led to phase separation between the dispersed and continuous phases. The dispersed phase settled at the bottom of the cylinder, reaching Point 2, while the continuous phase was represented by Point 3. At this position, a marked depression in the freezing point and a wider ice crystal growth range (between −0.8 °C and −6.7 °C) were observed, indicating solute dissolution in water due to the colligative properties of solutions.22,47
Points 1, 2, 4, and 5 displayed similar behaviour, representing the freezing of the lower region of the sample where the dispersed solids accumulated. The separation between water and the solute initiated nucleation and ice crystal formation at freezing points close to that of pure water, ranging from 0 °C to −2 °C.8,47,48 It was observed that heat loss predominantly occurred in the vertical direction, from bottom to top. Laterally, the temperature gradient was minimal, characterising unidirectional freezing of the material. Despite the insulating effect of the upper water layer, this freezing pattern was maintained.
3.6. Experimental design
| Y1 = 87.12 + 22.91X1 − 5.65X2 + 7.26X3 + 19.52X1X2 + 20.67X1X3 | (5) |
| Y2 = 0.659 − 0.014X1 + 0.025X2 − 0.012X1X2 + 0.021X2X3 | (6) |
| Effect | Value | Standard error | t-Value | Prob > |t| | |
|---|---|---|---|---|---|
| (Intercept) | 87.119 | 1.6898 | 51.5556 | 8.47 × 10−7 | |
| X1 | 45.8258 | 22.913 | 1.8893 | 12.1280 | 0.0003 |
| X2 | −11.3025 | −5.651 | 1.8893 | −2.9913 | 0.0403 |
| X3 | 14.5208 | 7.260 | 1.8893 | 3.8430 | 0.0184 |
| X1X2 | 19.5225 | 9.761 | 1.8893 | 5.1667 | 0.0067 |
| X1X3 | 20.6658 | 10.333 | 1.8893 | 5.4693 | 0.0054 |
| R2 | 0.983 | ||||
| Adj. R2 | 0.961 |
| Effect | Value | Standard error | t-Value | Prob > |t| | |
|---|---|---|---|---|---|
| (Intercept) | 0.659 | 0.0028 | 232.0052 | 1.77 × 10−7 | |
| X1 | −0.029 | −0.014 | 0.0032 | −4.4861 | 0.0207 |
| X2 | 0.051 | 0.025 | 0.0032 | 7.9942 | 0.0041 |
| X3 | 0.001 | 0.001 | 0.0032 | 0.1700 | 0.8759 |
| X1X2 | −0.024 | −0.012 | 0.0032 | −3.7256 | 0.0337 |
| X1X3 | 0.014 | 0.007 | 0.0032 | 2.1900 | 0.1163 |
| X2X3 | 0.021 | 0.011 | 0.0032 | 3.3523 | 0.0440 |
| R2 | 0.974 | ||||
| Adj. R2 | 0.923 |
Based on the analysis of Table 5, considering a p-value lower than α (0.05), it was observed that the constant term (indicated in the table as the “intercept”), the main effects X1, X2 and X3, as well as the interaction terms X1X2 and X1X3, exhibited significant coefficients and were statistically different from zero. In other words, these effects are associated with the response variable hardness (Y1), as described by using eqn (5).
The same analytical procedure was applied to the response variable cohesiveness (Y2), whose coefficients are presented in Table 6. In this case, the significant coefficients differed from those observed for Y1. For Y2, the constant term (intercept), the main effects X1 and X2, and the interaction terms X1X2 and X2X3 were significant at the 5% probability level (p < 0.05), being associated with the model described by using eqn (6). In contrast to the model obtained for hardness (Y1), the coefficients X3 and X1X3 were not significant and were therefore removed from the equation.
The Student's t-test can also be analysed using the t-critical value. The effects of the Y1 and Y2 models, respectively, were represented in the Pareto diagram (Fig. 6), in which they were ordered based on the degree of importance from the most significant to the least significant, as explained by Adio et al.49
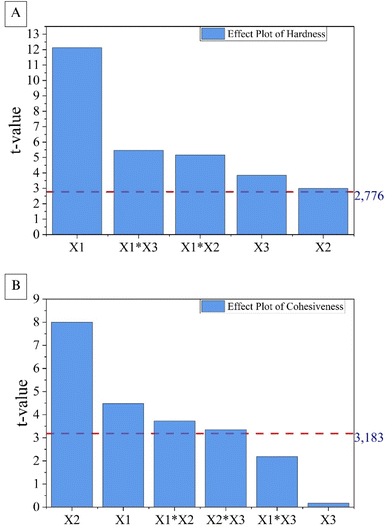 | ||
| Fig. 6 Pareto chart showing the standardised effects from factorial analysis of (A) hardness (Y1) and (B) cohesiveness (Y2), considering solids content (X1), pH (X2) and freezing temperature (X3). | ||
In the graph, the critical line is represented by the dashed horizontal line in red. Effects that exceed the t-critical line are considered significant, while those that are within its limit were not significant for the model. Thus, the interpretations agree, indicating that the five effects shown in the diagram for hardness (Y1) were significant, while X3 and X1X3 were not significant for cohesiveness (Y2).
In order to check the adequacy of the model, the R2 and R2aj. values were analysed, according to Nowalid et al.50 and these values indicate how close the data are to the regression line. According to Karazhiyan et al.51 models present good experimental fits for a minimum R2 of 0.8. Rahmani et al.52 indicated that higher values close to one for R2 and the proximity between this value and that of R2aj. suggest the high capacity of the fitted models. Thus, the equations generated for hardness (Y1) and cohesiveness (Y2) are well adjusted to the experimental data, as shown in Tables 5 and 6, indicating that the model has a good ability to describe the properties studied.
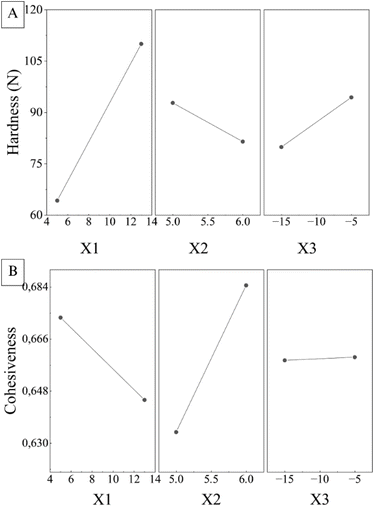 | ||
| Fig. 7 Main effect plots showing the influence of solids content (X1), pH (X2) and freezing temperature (X3) on (A) hardness (Y1) and (B) cohesiveness (Y2). | ||
The results show that X1, X2 and X3 have a significant influence on hardness (Y1). In particular, X1 and X3 positively affect Y1, so that an increase in the solids content or freezing temperature results in a higher hardness of the material. On the other hand, pH shows the opposite behaviour, indicating that when the formulation has a high pH, hardness tends to decrease.
When evaluating these effects on cohesiveness (Y2), this property is predominantly affected by pH, with higher values when the formulation has a higher pH. The solids content also has an influence, although inversely and with less impact compared to pH. Freezing temperature, on the other hand, has a minimal influence on the Y2 response.
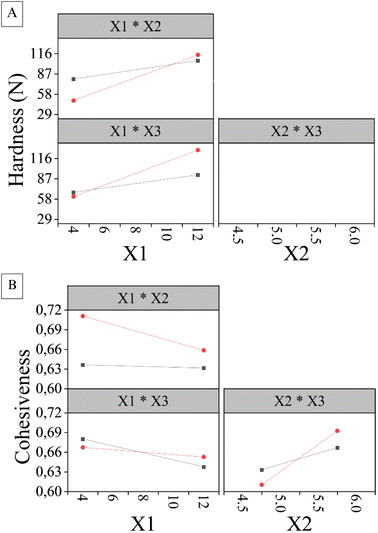 | ||
| Fig. 8 Interaction effect plots showing the combined influence of solids content (X1), pH (X2) and freezing temperature (X3) on (A) hardness (Y1) and (B) cohesiveness (Y2). | ||
The graphs only show the interactions that showed statistical significance in the previous analysis. Thus, the X2X3 interaction is shown exclusively for cohesiveness (Y2), since it was removed for hardness (Y1) because it was not significant and impacted the main effects. According to Altayb et al.,14 steep slopes suggest interaction between the factors analysed, which can be seen in Fig. 8. Both the X2X3 interaction for Y2 and the X1X2 and X1X3 interactions for Y1 and Y2 are represented by lines with a clear slope, indicating interaction between the respective factors.
Based on Fig. 8, we can observe that hardness (Y1), influenced by the X1X2 and X1X3 interactions, reached higher values at their respective high levels. In contrast, cohesiveness (Y2) showed the opposite behavior, being lower at these same levels. Additionally, it was found that high levels of the X2X3 interaction resulted in an increase in cohesiveness (Y2) in the product.
| Levels|parameters | −0.5 | 0.5 |
|---|---|---|
| a Solids content (X1), pH (X2), freezing temperature (X3), hardness (Y1), cohesiveness (Y2), theoretical value (T) and experimental value (E). | ||
| X1 (%) | 7 | 11 |
| X2 | 5.25 | 5.75 |
| X3 (°C) | −12.5 | −7.5 |
| Properties | T | E | T | E |
|---|---|---|---|---|
| Y1 (N) | 79.88 | 93.92 | 104.40 | 121.87 |
| Y2 | 0.66 | 0.73 | 0.67 | 0.75 |
4. Conclusions
Based on the full factorial design, it was observed that hardness (Y1) was predominantly influenced by solids content (X1), while cohesiveness (Y2) was most significantly impacted by pH (X2). The material demonstrated high potential for replicating the colour and texture of conventional meats, presenting values aligned with the literature and minimal colour differences compared to chicken breast and fish fillets. The freezing front suggests unidirectional crystal growth, while the DT indicated anisotropic behavior in samples S6 (13; 5.0; −5), S8 (13; 6.0; −5), and S9 (9; 5.5; −10), consistent with data described for protein gels. The centre point parameters (S9) yielded the best results regarding the formed structure and textural properties, indicating that the proposed methodology is promising for obtaining gels with meat-like properties. This study establishes a solid foundation in the field of alternative proteins, opening opportunities for further refinement through sensory studies, as well as the optimization of aspects related to the preservation and food safety of the developed products.Author contributions
Israel Jovino Caneschi was responsible for the original draft preparation, data curation, formal analysis, investigation, methodology development, conceptualisation, and technical formatting. Daniel Angelo Longhi contributed to the formal analysis and data curation, and participated in the review and editing of the manuscript. Charles Windson Isidoro Haminiuk was responsible for data acquisition and language revision. Maria Lucia Masson contributed to data curation, methodology, and supervision, participated in the review and editing of the manuscript, and was responsible for funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fb00871a.Acknowledgements
This study was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES) – Finance Code 001. Professor ML Masson thanks the Brazilian National Council for Scientific and Technological Development (CNPq – Grant 309504/2022-8). We also extend our gratitude to Embrapa – Empresa Brasileira de Pesquisa Agropecuária – Embrapa Florestas Unit, the Multidisciplinary Centre for Materials Characterisation (CMCM) – Federal Technological University of Paraná (UTFPR), and the Wood Anatomy Laboratory, Department of Forest Engineering – Federal University of Paraná (UFPR) for the infrastructure support.References
- U. Fresán, M. A. Mejia, W. J. Craig, K. Jaceldo-Siegl and J. Sabaté, Sustainability, 2019, 11, 3231 Search PubMed.
- H. Aiking, Trends Food Sci. Technol., 2011, 22, 112–120 Search PubMed.
- A. Singh and N. Sit, Int. J. Gastron. Food Sci., 2025, 39, 101095 Search PubMed.
- K. Nakagawa, R. Chantanuson, P. Boonarsa, N. Seephua and S. Siriamornpun, Food Chem.: X, 2024, 22, 101402 Search PubMed.
- L. Sha and Y. L. Xiong, Trends Food Sci. Technol., 2020, 102, 51–61 Search PubMed.
- K. E. Preece, N. Hooshyar and N. J. Zuidam, Innovative Food Sci. Emerging Technol., 2017, 43, 163–172 Search PubMed.
- R. Chantanuson, S. Nagamine, T. Kobayashi and K. Nakagawa, J. Food Eng., 2024, 362, 111779 Search PubMed.
- R. Chantanuson, S. Nagamine, T. Kobayashi and K. Nakagawa, Food Struct., 2022, 32, 100258 Search PubMed.
- J. H. Chiang, S. M. Loveday, A. K. Hardacre and M. E. Parker, Food Struct., 2019, 19, 100102 CrossRef.
- G. Giménez-Ribes, M. Oostendorp, A. J. van der Goot, E. van der Linden and M. Habibi, Food Hydrocolloids, 2024, 149, 109509 CrossRef.
- O. K. Ozturk and B. R. Hamaker, Future Foods, 2023, 8, 100248 Search PubMed.
- O. Yuliarti, T. J. Kiat Kovis and N. J. Yi, J. Food Eng., 2021, 288, 110138 Search PubMed.
- N. S. A. Mohd Sharif, M. F. Jamaluddin and N. Zainol, Mater. Today: Proc., 2021, 46, 1763–1769 CAS.
- H. N. Altayb, B. Kouidhi, O. A. S. Baothman, J. A. Abdulhakim, L. Ayed, M. Hager and K. Chaieb, J. Water Process Eng., 2021, 44, 102429 Search PubMed.
- R. Di Monaco, S. Cavella and P. Masi, J. Texture Stud., 2008, 39, 129–149 Search PubMed.
- United Nations, The 17 Goals, https://sdgs.un.org/, accessed 21 October 2025 Search PubMed.
- M. L. Masson and H. A. Fugmann, Bol. Cent. Pesqui. Process. Aliment., 1998, 16, 189–202 Search PubMed.
- Association of Official Analytical Chemists (AOAC), Official Methods of Analysis of the Association of Official Analytical Chemists, Virgínia (USA), 16th edn, 1995 Search PubMed.
- P. B. Pathare, U. L. Opara and F. A.-J. Al-Said, Food Bioprocess Technol., 2013, 6, 36–60 Search PubMed.
- C. E. Wagner, L. Levine, S. R. Saunders, R. Bergman, X. Guo and G. M. Ganjyal, Food Res. Int., 2024, 192, 114760 Search PubMed.
- O. Yuliarti, K. H. Mei, Z. Kam Xue Ting and K. Y. Yi, Food Hydrocolloids, 2019, 89, 216–223 Search PubMed.
- W. Jie, L. Lite and D. Yang, J. Food Eng., 2003, 60, 481–484 Search PubMed.
- F. L. Chen, Y. M. Wei, B. Zhang and A. O. Ojokoh, J. Food Eng., 2010, 96, 208–213 Search PubMed.
- OriginPro, version 2025, Northampton, MA, USA, 2025 Search PubMed.
- S. Damodaran and K. L. Parkin, Química de alimentos de Fennema, Artmed Editora Ltda, 5th edn, 2019 Search PubMed.
- P. e A. (MAPA) Ministério da Agricultura, Instrução Normativa no. 21 de 31 de maio de 2017, Brazil, 2017 Search PubMed.
- B. Gültekin Subaşı, B. Vahapoğlu, E. Capanoglu and M. A. Mohammadifar, Crit. Rev. Food Sci. Nutr., 2021, 62, 6682–6697 Search PubMed.
- N. Jonkers, J. A. W. van Dommelen and M. G. D. Geers, Mech. Time-Depend. Mater., 2022, 26, 323–346 Search PubMed.
- J.-S. Lee, H. Oh, I. Choi, C. S. Yoon and J. Han, LWT, 2022, 157, 113056 CrossRef CAS.
- A. J. Rosenthal and P. Thompson, J. Texture Stud., 2021, 52, 294–302 Search PubMed.
- Y. Fan, S. Zheng, P. K. Annamalai, B. Bhandari and S. Prakash, Sustainable Food Technol., 2024, 2, 826–836 RSC.
- L. Godschalk-Broers, G. Sala and E. Scholten, Foods, 2022, 11, 2227 CrossRef CAS PubMed.
- P. Boonarsa, K. Nakagawa, K. Banlue and S. Siriamornpun, Food Chem.: X, 2025, 27, 102370 CAS.
- J.-S. Lee, I. Choi and J. Han, Food Res. Int., 2022, 161, 111840 Search PubMed.
- D. Barbanti and M. Pasquini, LWT–Food Sci. Technol., 2005, 38, 895–901 Search PubMed.
- J. Kim, Y. Zhang, Y. Han, Y. Lee, C. Kang, H. Jung and B.-J. Gu, Food Sci. Preserv., 2025, 32, 246–259 CrossRef.
- B. I. Zielbauer, J. Franz, B. Viezens and T. A. Vilgis, Food Biophys., 2016, 11, 34–42 Search PubMed.
- J. Valentim, C. Afonso, R. Gomes, A. Gomes-Bispo, J. A. M. Prates, N. M. Bandarra and C. Cardoso, Heliyon, 2024, 10, e27171 CrossRef CAS PubMed.
- E. P. Ribeiro and E. A. G. Seravalli, in Química de alimentos, Editora Blucher, 2nd edn, 2007, pp. 96–121 Search PubMed.
- L. R. Beuchat, E. Komitopoulou, H. Beckers, R. P. Betts, F. Bourdichon, S. Fanning, H. M. Joosten and B. H. Ter Kuile, J. Food Prot., 2013, 76, 150–172 Search PubMed.
- M. H. Taylor and M.-J. Zhu, Trends Food Sci. Technol., 2021, 116, 802–814 CrossRef CAS.
- U. Habiba, A. Bajpai, Z. Shafi, V. K. Pandey and R. Singh, J. Stored Prod. Res., 2025, 112, 102657 Search PubMed.
- D. Alp and Ö. Bulantekin, Eur. Food Res. Technol., 2021, 247, 1333–1343 Search PubMed.
- E. Zang, L. Jiang, H. Cui, X. Li, Y. Yan, Q. Liu, Z. Chen and M. Li, Food Rev. Int., 2023, 39, 5132–5163 CrossRef CAS.
- A. Kumar, S. Das, S. Ali, S. G. Jaiswal, A. Rabbani, S. M. E. Rahman, R. Chelliah, D.-H. Oh, S. Liu and S. Wei, Food Biosci., 2025, 74, 107864 CrossRef CAS.
- Y. Fang, B. Zhang and Y. Wei, J. Food Eng., 2014, 121, 32–38 CrossRef CAS.
- A. L. de Oliveira, Refrigeração e cadeia do frio para alimentos, Universidade de São Paulo, Faculdade de Zootecnia e Engenharia de Alimentos, Pirassununga, 2020 Search PubMed.
- S. Damodaran, K. L. Parkin and O. R. Fennema, Química de alimentos de Fennema, Artmed, 2010 Search PubMed.
- S. O. Adio, M. H. Omar, M. Asif and T. A. Saleh, Process Saf. Environ. Prot., 2017, 107, 518–527 CrossRef CAS.
- W. F. W. M. Nowalid, H. A. Hamid and S. H. Giwa, Food Chem.: Mol. Sci., 2024, 8, 100196 CAS.
- H. Karazhiyan, S. M. A. Razavi and G. O. Phillips, Food Hydrocolloids, 2011, 25, 915–920 Search PubMed.
- R. Rahmani and R. Firouzi, Int. J. Commun. Network. Distr. Syst., 2021, 26, 1 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |