Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Sustainable delivery of encapsulated probiotics and their survival in fruit-based beverages and under simulated gastrointestinal conditions
Zahida
Naseem
*a,
Sajad Mohd
Wani
 *a,
Sajad Ahmad
Mir
b and
Naseh
Nisar
a
*a,
Sajad Ahmad
Mir
b and
Naseh
Nisar
a
aDivision of Food Science and Technology, Sher-e-Kashmir University of Agricultural Sciences and Technology of Kashmir, Jammu and Kashmir 190025, India. E-mail: wanisajad82@gmail.com; zahidanaseem83@gmail.com; Tel: +91 9858445878 Tel: +91 8825045657
bDepartment of Life Sciences (Microbiology & Food Science and Technology), GITAM (Deemed to be University), Visakhapatnam, 530045, India
First published on 5th January 2026
Abstract
Fruit and vegetable-based beverages are excellent non-dairy carriers for probiotics. However, maintaining probiotic viability in beverages while preserving product quality remains a significant challenge. The present research addressed this gap by evaluating the impact of encapsulation using gum arabic, soy protein, and their combination as wall materials for enhancing probiotic stability in apple and apple-carrot blend beverages. The effect of encapsulated probiotics on the physicochemical and sensory attributes of beverages was investigated. Furthermore, the viability of probiotics in the beverages and under simulated gastrointestinal tract conditions was evaluated. The results revealed that viability of encapsulated probiotics was retained at a level of 8.38 to 9.52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log CFU mL−1 in the beverages and 7.33 to 8.51
log CFU mL−1 in the beverages and 7.33 to 8.51![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log CFU mL−1 in the simulated GIT. Also, the incorporation of encapsulated probiotics did not significantly affect the physicochemical and sensory attributes of the beverages except for colour. The microencapsulated probiotics containing a mixture of gum arabic and soy protein as the wall material provided better protection against low pH environments to probiotics in apple and apple-carrot blend beverages compared to those containing gum arabic and soy protein alone. This study demonstrates that encapsulation using gum arabic and soy protein as wall materials is an effective strategy for improving probiotic viability in fruit and vegetable-based beverages without compromising product quality.
log CFU mL−1 in the simulated GIT. Also, the incorporation of encapsulated probiotics did not significantly affect the physicochemical and sensory attributes of the beverages except for colour. The microencapsulated probiotics containing a mixture of gum arabic and soy protein as the wall material provided better protection against low pH environments to probiotics in apple and apple-carrot blend beverages compared to those containing gum arabic and soy protein alone. This study demonstrates that encapsulation using gum arabic and soy protein as wall materials is an effective strategy for improving probiotic viability in fruit and vegetable-based beverages without compromising product quality.
Sustainability spotlightThis study supports sustainability by developing fruit and vegetable-based functional beverages that contain encapsulated probiotics. It promotes minimal processing and adds value to seasonal fruits by developing fruit-based beverages, which are frequently made from locally accessible agricultural commodities. In line with recent efforts to valorise plant matrices as sustainable substrates and carriers for probiotics, this study employs apple and carrot beverages as plant-based vehicles for encapsulated Lactobacillus strains. The encapsulating materials are food-grade, biodegradable, and eco-friendly, and there is less need for artificial stabilizers and preservatives. Furthermore, increasing probiotic survivability supports the global trend toward clean-label functional foods and improves nutritional and health advantages. This study demonstrates how scientific improvements to basic fruit drinks can promote environmental sustainability and consumer health, fostering a more conscientious and forward-thinking food system. |
1. Introduction
Probiotics are beneficial microbes that have therapeutic effects on the organisms ingesting them.1 Probiotics help prevent diarrhea, reduce stomach discomfort, reduce inflammation, improve lactose utilization, lower blood ammonia levels, control intestinal infections and boost immunity.2 To experience the health benefits of probiotics, a daily intake of at least 106–107 CFU mL−1 is advised. The probiotication of beverages can boost their market value due to the presence of beneficial bacteria. The market for plant-based products has been expanding more quickly in recent years, and there is a growing need for probiotics to be added to fruit and vegetable juices. Recent studies have also highlighted the use of fruit-derived by-products and plant matrices as functional substrates and carriers for probiotics, demonstrating both good viability and added nutritional or prebiotic value.3 Consumer preferences have shifted toward healthier options as a result of growing understanding of the allergenicity and high amounts of lipids and cholesterol in milk.4 Additionally, companies have expanded into dairy-free and vegan-based drinks due to vegetarian, animal, and environmental advocacy and lactose sensitivity. Therefore, it is necessary to use fruits and vegetables in probiotic drinks in order to satisfy the demand for vegan and dairy-free beverages. Fruit and vegetable-based drinks are also well-liked by people of all ages.5 They can provide favourable conditions for the growth and survival of probiotic microbes due to their high nutritional value. Hence, non-dairy probiotic products would be an alternative way to benefit from the functional effects of probiotic strains.6 However, it is challenging to maintain viability of probiotics in fruit and vegetable juices due to the adverse effects of the low pH environment (<pH 4.0).7 Most probiotic microorganisms rapidly lose their viability in the absence of protection. The loss of probiotic viability impairs the functionality because a sufficient number of viable bacteria (>7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log CFU g−1 of product) is essential to provide health benefits when the probiotic product is consumed.8 The viability of probiotics in fruit-based juice is affected by storage temperature, oxygen saturation and fibre content. Numerous strategies have been used to improve the ability of probiotics to survive in low pH environments and microencapsulation can be used as a potential method to ensure such a high concentration.9
log CFU g−1 of product) is essential to provide health benefits when the probiotic product is consumed.8 The viability of probiotics in fruit-based juice is affected by storage temperature, oxygen saturation and fibre content. Numerous strategies have been used to improve the ability of probiotics to survive in low pH environments and microencapsulation can be used as a potential method to ensure such a high concentration.9
Encapsulation reduces cell death and damage by keeping the cells inside a membrane.10 Microencapsulation has gained a lot of interest as a method to protect probiotic cells in food and against storage conditions. It is needed to enhance the viability of probiotic cells in intestinal juice and during food processing so that the probiotics reach the intestinal content in appropriate quantities (107 CFU mL−1)11 and exert a favourable effect on the host.12 Encapsulating materials such as proteins or polysaccharides shields the probiotics from environmental stresses including moisture, gas exchange, solute migration, UV radiation, heat, etc. For successful probiotic microencapsulation, the compatibility of each element such as the type of microbe, the method of microencapsulation, and the coating material is crucial.13
Apple juice is known as a functional food due to its health promoting properties in addition to its nutritional value. It contains a number of nutrients and minerals, including boron, which may help maintain strong bones. Apple juice consumption increases the acetylcholine levels in the brain, which may improve memory. It is known to provide many health benefits and regular consumption of apple juice is linked to a lower risk of diabetes, asthma, cardiovascular related diseases, and cancer as well.14 Also, carrot juice is a rich source of beta-carotene, vitamin A, and minerals such as calcium and potassium. It is easier to digest than raw or cooked carrots. Moreover, carrot juice is well-known for its therapeutic benefits because it works effectively to treat urogenital disorders.15
Beverages prepared from apple and carrot will be incredibly nutritious, pleasant, and refreshing and also provide vitamins, minerals, antioxidants, and vital flavonoids. Juice blending is one of the best methods to improve aroma, taste and the nutritional quality of the juice. It can improve the vitamin and mineral content depending on the kind and quality of fruits and vegetables used.
In line with recent efforts to valorise plant matrices as sustainable substrates and carriers for probiotics,16 this study employs apple and carrot as plant-based vehicles to produce a functional beverage incorporated with microencapsulated probiotics to investigate the viability of probiotics in functional beverages and under simulated gastrointestinal conditions.
2. Materials and methods
2.1. Chemicals and reagents
Analytical grade chemicals and reagents were used in the research. Hexane, acetone, ethanol, methanol, toluene, petroleum ether, gallic acid, HCl, sodium thiosulfate, Folin–Ciocalteu (FC) reagent, potassium oxalate, 2,2-diphenyl-1-picrylhydrazyl (DPPH), lead acetate, sodium chloride, sodium carbonate and lysozyme were procured from HiMedia (Mumbai, India).2.2. Procurement of raw materials
Fresh and mature apples and carrots were purchased from a local market in Shalimar, Srinagar, India. The obtained apples and carrots were thoroughly cleaned and washed.2.3. Preparation of probiotic fruit and vegetable-based beverages
Preparation of beverages from apple (100%) and apple (50%) blended with carrot (50%) as per FSSAI specifications was carried out at the Food Processing Training Centre, Division of Food Science & Technology, SKUAST-K, Shalimar, Srinagar, India. The previously prepared encapsulated probiotic powder in which five probiotic strains of Lactobacills spp. viz L. acidophillus, L. bulgaricus, L. casei, L. plantarum and L. rhamnosus were encapsulated in gum arabic, soy protein isolate, and a combination of the two using the freeze drying technique (Naseem et al., 2025,17) was added to each beverage. 15 treatment combinations such as T1, T2, T3, T4, T5, T6, T7, T8, T9, T10, T11, T12, T13, T14 and T15 were formed (Table 1).| Treatments | Apple/apple-carrot blend beverage | Wall material |
|---|---|---|
| T0 | Beverages without probiotics | Probiotic strains encapsulated in gum arabic (G) |
| T1 | Beverages containing GLa | |
| T2 | Beverages containing GLb | |
| T3 | Beverages containing GLc | |
| T4 | Beverages containing GLp | |
| T5 | Beverages containing GLr | |
| T6 | Beverages containing SLa | Probiotic strains encapsulated in soy protein isolate (S) |
| T7 | Beverages containing SLb | |
| T8 | Beverages containing SLc | |
| T9 | Beverages containing SLp | |
| T10 | Beverages containing SLr | |
| T11 | Beverages containing SGLa | Probiotic strains encapsulated in a combination of gum arabic and soy protein isolate (SG) |
| T12 | Beverages containing SGLb | |
| T13 | Beverages containing SGLc | |
| T14 | Beverages containing SGLp | |
| T15 | Beverages containing SGLr |
2.4. Physiochemical properties
The titratable acidity (as percent malic acid) was determined by titrating the sample (10 mL) with 0.1 N NaOH using phenolphthalein as an indicator. The results were calculated using the following equation:
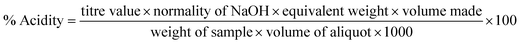 | (1) |
The pH value was determined using a pH meter (Thermo Scientific Orion Star A111, USA) at 25 °C.
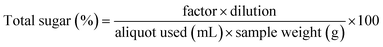 | (2) |
Reducing sugar was estimated by the Lane and Eynon method described by Rangana.18
As per Brand-Williams et al.,20 the antioxidant activity in terms of DPPH radical scavenging activity of the sample was assessed with a minor modification. About 3.9 mL of DPPH solution (6 × 10−5 mol L−1) and 0.1 mL of sample were mixed. The resulting mixture was left undisturbed in the dark for half an hour and optical density was recorded at 517 nm using a spectrophotometer. The values were expressed as % inhibition using the following formula:
 | (3) |
| Total carotenoid (mg per 100 mL) = (ΔA/€L) × MW × D × (V/G) | (4) |
Total anthocyanin content was estimated according to the modified method of Naseem et al.22 About 50 mL of ethanolic HCl (85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) was added to 10 mL. The mixture was transferred to a volumetric flask and brought to a final volume of 250 mL and refrigerated overnight at 4 °C. The spectrophotometer was used to check the absorbance of the aliquot at 535 nm after 2 mL of it was once again diluted to 100 mL with ethanolic HCl.
15) was added to 10 mL. The mixture was transferred to a volumetric flask and brought to a final volume of 250 mL and refrigerated overnight at 4 °C. The spectrophotometer was used to check the absorbance of the aliquot at 535 nm after 2 mL of it was once again diluted to 100 mL with ethanolic HCl.
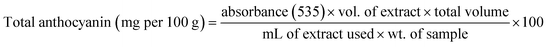 | (5) |
Turbidity was determined nephelometrically using a turbidity meter (Aquasol AP-TB-01). The results were expressed as nephelometric turbidity units (NTUs).
The survival of encapsulated probiotics in gastrointestinal simulation was assessed as described previously by Krasaekoopt & Watcharapoka,24 with minor alterations. The simulated gastric solution (0.08 M HCl with 0.2% NaCl and 0.3% pepsin, pH 1.55) was combined with 10 mL of beverage and incubated for one hour. The intestinal solution (0.05 M KH2PO4 with 0.6% bile salts and 0.1% pancreatin, pH 7.4) was then added and incubated for 2 hours. Following incubation, 1 mL of the solution combination was combined with PBS, serially diluted, and plated onto MRS agar in order to determine the log CFU mL−1 and ascertain the number of live bacteria.
3. Results and discussion
3.1. TSS, titratable acidity and pH
Total soluble solids (TSS) and titratable acidity were maintained at 15 °B and 0.30% respectively as per FSSAI specifications. The total soluble solid refers to the total amount of solids dissolved in a solution, mostly sugars such as sucrose, fructose and glucose, and fruit acids. The TSS of control and probiotic fortified apple beverages and apple-carrot blend beverages was 15 °B. The addition of probiotics encapsulated in different wall materials, viz gum arabic, soy protein and the combination of the two, did not affect the TSS of the apple and apple-carrot blend beverages (Table 2). This is because the encapsulation matrix isolates the probiotics from the juice, preventing immediate interaction between the probiotics and the juice's sugars or other soluble solids.25 Ding & Shah26 also showed similar results for TSS while analysing orange and apple juices with free and microencapsulated probiotic bacteria.| Treatment product | T0 | T1 | T2 | T3 | T4 | T5 | T6 | T7 | T8 | T9 | T10 | T11 | T12 | T13 | T14 | T15 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Values are expressed as mean ± standard deviation. b Values with different superscripts (capital letters) (within columns) differ significantly (p < 0.05). | ||||||||||||||||
| TSS (°B) | ||||||||||||||||
| Apple beverage | 15.00 ± 0.02aA | 15.01 ± 0.05aA | 15.01 ± 0.03aA | 15.01 ± 0.04aA | 15.02 ± 0.07aA | 15.02 ± 0.03aA | 15.01 ± 0.06aA | 15.02 ± 0.01aA | 15.02 ± 0.09aA | 15.01 ± 0.03aA | 15.01 ± 0.08aA | 15.02 ± 0.05aA | 15.02 ± 0.03aA | 15.01 ± 0.04aA | 15.01 ± 0.05aA | 15.02 ± 0.02aA |
| Apple-carrot blend | 15.00 ± 0.15aA | 15.01 ± 0.19aA | 15.02 ± 0.13aA | 15.01 ± 0.14aA | 15.02 ± 0.17aA | 15.02 ± 0.11aA | 15.01 ± 0.12aA | 15.01 ± 0.13aA | 15.02 ± 0.16aA | 15.00 ± 0.14aA | 15.00 ± 0.17aA | 15.01 ± 0.19aA | 15.00 ± 0.16aA | 15.03 ± 0.04aA | 15.00 ± 0.05aA | 15.02 ± 0.08aA |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Acidity (%) | ||||||||||||||||
| Apple beverage | 0.30 ± 0.01aA | 0.30 ± 0.03aA | 0.30 ± 0.05aA | 0.30 ± 0.06aA | 0.30 ± 0.02aA | 0.30 ± 0.01aA | 0.30 ± 0.07aA | 0.30 ± 0.03aA | 0.30 ± 0.07aA | 0.30 ± 0.01aA | 0.30 ± 0.01aA | 0.30 ± 0.02aA | 0.30 ± 0.06aA | 0.30 ± 0.04aA | 0.30 ± 0.01aA | 0.30 ± 0.05aA |
| Apple-carrot blend | 0.30 ± 0.03aA | 0.30 ± 0.03aA | 0.30 ± 0.05aA | 0.30 ± 0.01aA | 0.30 ± 0.02aA | 0.30 ± 0.04aA | 0.30 ± 0.07aA | 0.30 ± 0.03aA | 0.30 ± 0.07aA | 0.30 ± 0.01aA | 0.30 ± 0.04aA | 0.30 ± 0.02aA | 0.30 ± 0.06aA | 0.30 ± 0.04aA | 0.30 ± 0.02aA | 0.30 ± 0.05aA |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| pH | ||||||||||||||||
| Apple beverage | 3.27 ± 0.05aA | 3.27 ± 0.02aA | 3.27 ± 0.04aA | 3.27 ± 0.07aA | 3.27 ± 0.03aA | 3.27 ± 0.06aA | 3.27 ± 0.04aA | 3.27 ± 0.08aA | 3.27 ± 0.11aA | 3.27 ± 0.09aA | 3.27 ± 0.03aA | 3.27 ± 0.07aA | 3.27 ± 0.10aA | 3.27 ± 0.05aA | 3.27 ± 0.06aA | 3.27 ± 0.04aA |
| Apple-carrot blend | 3.21 ± 0.03aA | 3.21 ± 0.07aA | 3.21 ± 0.02aA | 3.21 ± 0.06aA | 3.21 ± 0.08aA | 3.21 ± 0.03aA | 3.21 ± 0.02aA | 3.21 ± 0.05aA | 3.21 ± 0.09aA | 3.21 ± 0.07aA | 3.21 ± 0.02aA | 3.21 ± 0.04aA | 3.21 ± 0.07aA | 3.21 ± 0.01aA | 3.21 ± 0.05aA | 3.21 ± 0.08aA |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Total sugar (%) | ||||||||||||||||
| Apple beverage | 13.79 ± 0.02aB | 13.79 ± 0.04aB | 13.79 ± 0.03aB | 13.79 ± 0.05aB | 13.79 ± 0.07aB | 13.79 ± 0.05aB | 13.79 ± 0.09aB | 13.79 ± 0.06aB | 13.79 ± 0.03aB | 13.79 ± 0.06aB | 13.79 ± 0.09aB | 13.79 ± 0.01aB | 13.79 ± 0.07aB | 13.79 ± 0.04aB | 13.79 ± 0.08aB | 13.79 ± 0.03aB |
| Apple-carrot blend | 13.31 ± 0.11aA | 13.31 ± 0.13aA | 13.31 ± 0.15aA | 13.31 ± 0.13aA | 13.31 ± 0.16aA | 13.31 ± 0.17aA | 13.31 ± 0.15aA | 13.31 ± 0.18aA | 13.31 ± 0.13aA | 13.31 ± 0.17aA | 13.31 ± 0.13aA | 13.31 ± 0.16aA | 13.31 ± 0.15aA | 13.31 ± 0.19aA | 13.31 ± 0.14aA | 13.31 ± 0.18aA |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Reducing sugar (%) | ||||||||||||||||
| Apple beverage | 6.15 ± 0.03aB | 6.15 ± 0.04aB | 6.15 ± 0.06aB | 6.15 ± 0.08aB | 6.15 ± 0.09aB | 6.15 ± 0.05aB | 6.15 ± 0.07aB | 6.15 ± 0.04aB | 6.15 ± 0.01aB | 6.15 ± 0.05aB | 6.15 ± 0.02aB | 6.15 ± 0.06aB | 6.15 ± 0.08aB | 6.15 ± 0.04aB | 6.15 ± 0.05aB | 6.15 ± 0.10aB |
| Apple-carrot blend | 5.81 ± 0.10aA | 5.81 ± 0.11aA | 5.81 ± 0.12aA | 5.81 ± 0.15aA | 5.81 ± 0.17aA | 5.81 ± 0.14aA | 5.81 ± 0.18aA | 5.81 ± 0.13aA | 5.81 ± 0.12aA | 5.81 ± 0.15aA | 5.81 ± 0.14aA | 5.81 ± 0.17aA | 5.81 ± 0.12aA | 5.81 ± 0.13aA | 5.81 ± 0.18aA | 5.81 ± 0.15aA |
Titratable acidity quantifies the total acid concentration in a sample and is usually expressed as a percentage of the dominant organic acid.27 In this study, it was measured as a percentage of malic acid, as apples and carrots28 contain a high proportion of malic acid. The titratable acidity of apple beverages and apple-carrot blend beverages with and without addition of encapsulated probiotic powder was 0.30% (Table 2). A non-significant (p ≤ 0.05) difference was found among all the treatments of the two beverages. The probiotics encapsulated in different wall materials viz gum arabic, soy protein and the combination of the two, did not have a significant effect on the titratable acidity of the apple beverages and apple-carrot blend beverages. This indicates that wall materials used for encapsulation of probiotics do not contribute to the overall acidity of the beverage. Similar results were observed by Nwaokoro & Akanbi.29 They found that adding hydrocolloids such as xanthan gum and carboxymethyl cellulose had no effect on the titratable acidity value of tomato and carrot juice.
pH refers to the negative logarithm of the dissolved hydrogen ion (H+) concentration and measures the acidity or basicity of a solution. The pH of the control apple beverage and control apple-carrot blend beverage was 3.27 and 3.21 respectively (Table 2). The pH varied significantly (p ≥ 0.05) between the two beverages while a non-significant (p ≤ 0.05) difference in pH was observed in different treatments of the two beverages. The different acid content and the diluting impact of mixing the more acidic apple juice with the less acidic carrot juice could be the cause of the pH variation between the two beverages. The addition of encapsulated probiotic powder did not affect the pH of the beverages, as microencapsulation involves embedding probiotics in a protective coating. This barrier typically delays direct interaction between the probiotics and the beverages, resulting in a minimal immediate impact on pH upon addition. Yousefi et al.30 also reported no change in pH on day 0 of apple juice containing encapsulated probiotics. The findings were consistent with those of Bonaccorso et al.23 who found that the pH of orange juice containing both free and encapsulated L. rhamnosus GG remained stable during storage.
3.2. Total sugars and reducing sugars
Total sugars include reducing sugars and non-reducing sugars. It encompasses both naturally occurring sugars, like fructose and glucose, and any added sugars. In fruits and vegetables, sugars contribute to their flavor, making them palatable and desirable for consumption. The main sugars present in apples and carrots are sucrose, fructose, and glucose.31,32 The values of total sugar content are depicted in Table 2. The total sugar content of apple and apple-carrot blend beverages with encapsulated probiotics was 13.79% and 13.31% respectively. The total sugar content of apple beverages was significantly (p ≤ 0.05) higher than that of the carrot blend beverages. This might be ascribed to the higher concentrations of simple sugars like sucrose, fructose, and glucose in apples. These sugars are the primary contributors to the total sugar content in apple-based beverages. The addition of encapsulated probiotics did not alter the total sugar content of the two beverages. The outcomes of the present study are in line with those by Mostafa et al.33 in their study of production of a novel probiotic date juice with anti-proliferative activity against Hep-2 cancer cells.The reducing sugar content of apple and apple-carrot blend beverages with encapsulated probiotic powder varied significantly (p ≤ 0.05) and was 6.15% and 5.81% respectively (Table 2). The encapsulating material did not contribute to the reducing sugar content of the beverages. Higher reducing sugar was found in apple beverages and lower reducing sugar was found in the apple-carrot blend beverages. The findings are in line with the results of ref. 33.
3.3. Total phenolic content and antioxidant activity
Fruits and vegetables contain a mixture of phenols which contribute to their astringency, bitterness, odor, color, flavor, and oxidative stability. Phenolic compounds are a diverse group of compounds with varying stability. These compounds have distinct structures and are directly impacted by temperature, light, oxygen, and pH.34 The total phenolic content (TPC) is a critical measure of the antioxidant capacity of fruit and vegetable beverages. In this study, the TPC of apple and apple-carrot blend beverages was analysed and the results are depicted in Table 3. The TPC of the control apple beverage was 351.50 mgGAE/100 mL which is significantly (p ≤ 0.05) different from that of the control apple-carrot blend beverage (272.71 mgGAE/100 mL). The TPC of different apple beverages with encapsulated probiotic powder varied non-significantly (p ≥ 0.05) between 351.50 and 351.54 mgGAE/100 mL. Also, a non-significant (p ≥ 0.05) trend in TPC was noted in different treatments of apple-carrot blend beverages with added probiotic powder (272.70 to 272.73 mgGAE/100 mL). The higher total phenolic content observed in apple beverages compared to apple-carrot blend beverages could be attributed to the inherent differences in phenolic compound concentrations between apples and carrots. The same was found by Grubišić et al.35 while analysing the impact of wheatgrass juice on the nutritional value of orange, lemon, carrot, beet, and apple juice. However, the consistency between treatments showed that TPC was not changed by the addition of encapsulated probiotic powder.| Treatment product | T0 | T1 | T2 | T3 | T4 | T5 | T6 | T7 | T8 | T9 | T10 | T11 | T12 | T13 | T14 | T15 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Values are expressed as mean ± standard deviation. b Values with different superscripts (capital letters) (within columns) differ significantly (p < 0.05). | ||||||||||||||||
| Total phenolic content (mgGAE per 100 mL) | ||||||||||||||||
| Apple beverage | 351.50 ± 0.31aB | 351.51 ± 0.36aB | 351.53 ± 0.32aB | 351.51 ± 0.34aB | 351.51 ± 0.33aB | 351.52 ± 0.35aB | 351.52 ± 0.37aB | 351.53 ± 0.39aB | 351.53 ± 0.38aB | 351.54 ± 0.41aB | 351.51 ± 0.40aB | 351.53 ± 0.46aB | 351.52 ± 0.45aB | 351.50 ± 0.49aB | 351.52 ± 0.43aB | 351.54 ± 0.42aB |
| Apple-carrot blend | 272.71 ± 0.15aA | 272.73 ± 0.16aA | 272.70 ± 0.13aA | 272.72 ± 0.12aA | 272.72 ± 0.14aA | 272.72 ± 0.11aA | 272.71 ± 0.20aA | 272.71 ± 0.17aA | 272.70 ± 0.22aA | 272.71 ± 0.19aA | 272.71 ± 0.21aA | 272.71 ± 0.23aA | 272.71 ± 0.18aA | 272.72 ± 0.22aA | 272.70 ± 0.25aA | 272.73 ± 0.26aA |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Antioxidant activity (% DPPH inhibition) | ||||||||||||||||
| Apple beverage | 64.76 ± 0.12aB | 64.76 ± 0.14aB | 64.76 ± 0.13aB | 64.76 ± 0.23aB | 64.76 ± 0.25aB | 64.76 ± 0.16aB | 64.76 ± 0.17aB | 64.76 ± 0.26aB | 64.76 ± 0.21aB | 64.76 ± 0.18aB | 64.76 ± 0.24aB | 64.76 ± 0.11aB | 64.76 ± 0.19aB | 64.76 ± 0.29aB | 64.76 ± 0.27aB | 64.76 ± 0.28aB |
| Apple-carrot blend | 54.15 ± 0.05aA | 54.14 ± 0.09aA | 54.15 ± 0.02aA | 54.14 ± 0.11aA | 54.15 ± 0.15aA | 54.15 ± 0.08aA | 54.13 ± 0.12aA | 54.15 ± 0.07aA | 54.14 ± 0.13aA | 54.15 ± 0.16aA | 54.15 ± 0.10aA | 54.14 ± 0.30aA | 54.15 ± 0.29aA | 54.15 ± 0.18aA | 54.13 ± 0.22aA | 54.15 ± 0.20aA |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Total carotenoids (mg per 100 mL) | ||||||||||||||||
| Apple beverage | 3.33 ± 0.11aA | 3.34 ± 0.13aA | 3.36 ± 0.12aA | 3.39 ± 0.14aA | 3.33 ± 0.19aA | 3.36 ± 0.15aA | 3.34 ± 0.18aA | 3.35 ± 0.17aA | 3.39 ± 0.21aA | 3.35 ± 0.19aA | 3.37 ± 0.23aA | 3.38 ± 0.25aA | 3.39 ± 0.16aA | 3.36 ± 0.20aA | 3.35 ± 0.10aA | 3.39 ± 0.24aA |
| Apple-carrot blend | 28.88 ± 0.41aB | 28.89 ± 0.43aB | 28.89 ± 0.48aB | 28.87 ± 0.42aB | 28.89 ± 0.46aB | 28.89 ± 0.45aB | 28.87 ± 0.47aB | 28.89 ± 0.49aB | 28.89 ± 0.50aB | 28.88 ± 0.55aB | 28.89 ± 0.46aB | 28.86 ± 0.52aB | 28.89 ± 0.51aB | 28.87 ± 0.62aB | 28.89 ± 0.60aB | 28.87 ± 0.59aB |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Anthocyanin (mg per 100 mL) | ||||||||||||||||
| Apple beverage | 23.01 ± 0.20aB | 23.01 ± 0.19aB | 23.01 ± 0.11aB | 23.01 ± 0.13aB | 23.01 ± 0.15aB | 23.01 ± 0.25aB | 23.01 ± 0.14aB | 23.01 ± 0.17aB | 23.01 ± 0.12aB | 23.01 ± 0.18aB | 23.01 ± 0.16aB | 23.01 ± 0.21aB | 23.01 ± 0.23aB | 23.01 ± 0.22aB | 23.01 ± 0.19aB | 23.01 ± 0.24aB |
| Apple-carrot blend | 15.70 ± 0.01aA | 15.71 ± 0.03aA | 15.71 ± 0.02aA | 15.72 ± 0.08aA | 15.71 ± 0.12aA | 15.70 ± 0.06aA | 15.71 ± 0.05aA | 15.71 ± 0.09aA | 15.72 ± 0.04aA | 15.71 ± 0.07aA | 15.70 ± 0.10aA | 15.71 ± 0.01aA | 15.71 ± 0.12aA | 15.72 ± 0.13aA | 15.71 ± 0.09aA | 15.72 ± 0.03aA |
Plant polyphenols are among the most potent antioxidants because of their capacity to scavenge free radicals and deactivate other pro-oxidants. The primary components responsible for the antioxidant action of fruit juices are phenolic chemicals.36 The antioxidant activity of control beverages, apple beverages and apple-carrot blend beverages incorporated with probiotics is shown in Table 3. The significantly (p ≤ 0.05) highest antioxidant activity was observed in the apple beverage (64.76%) and the lowest was in the apple-carrot blend beverage (54.15%). The antioxidant activity of apple beverages with encapsulated probiotics was 64.76% across all treatments while it varied non-significantly (p ≥ 0.05) from 54.13 to 54.15% in all the treatments of apple-carrot blend beverages with added probiotic powder. The difference in antioxidant activity between the apple beverage and apple-carrot blend beverage can be attributed to the higher phenolic content and ascorbic acid in apples as compared to the carotenoid-dominated antioxidant profile of carrots.37 Since total phenol and flavonoid contents are linked to antioxidant activity that scavenges radicals, juices with higher levels of these compounds demonstrated higher antioxidant activity. Also, the wall material used for the encapsulation of probiotics did not affect the antioxidant activity of the two beverages. The outcomes are in agreement with the results of Gao & Rupasinghe,38 who revealed that the blend with the maximum apple-to-carrot ratio (90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) exhibited the maximum antioxidant activity, while the blend with the lowest apple-to-carrot ratio (60
10) exhibited the maximum antioxidant activity, while the blend with the lowest apple-to-carrot ratio (60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40) demonstrated the minimum antioxidant activity. Similarly, Suzuki et al.3 observed that probiotics in red pitaya pulp preserved key bioactive compounds (betacyanins) and resulted in acceptable sensory profiles.
40) demonstrated the minimum antioxidant activity. Similarly, Suzuki et al.3 observed that probiotics in red pitaya pulp preserved key bioactive compounds (betacyanins) and resulted in acceptable sensory profiles.
3.4. Total carotenoids and anthocyanins
Carotenoids are isoprenoid pigments synthesized by plants, contributing to the colour and nutritional value of fruits and vegetables. Several carotenoids such as lutein, β-carotene, zeaxanthin, violaxanthin, β-cryptoxanthin, and neoxanthin are present in apples39 and carrots are rich in β-carotene and α-carotene. Carotenoids are associated with improved immunological function and a lower risk of deteriorating diseases such as cancer, heart disease, age-related muscular disintegration, and cataract development. The data on total carotenoids are presented in Table 3. The carotenoid content of the control apple beverage was recorded at 3.33 mg/100 mL, significantly (p ≤ 0.05) lower than that of the control apple-carrot blend beverage (28.88 mg/100 mL). The carotenoid content of different apple beverages with added probiotic powder varied non-significantly (p ≥ 0.05) between 3.33 and 3.39 mg/100 mL. Also, a non-significant (p ≥ 0.05) difference from 28.87 to 28.89 mg/100 mL was observed in the apple-carrot blend beverages with encapsulated probiotics. The higher carotenoid concentration of the apple-carrot blend beverage could be attributed to the higher carotenoid levels naturally found in carrots compared to apples. An analogous trend was also observed by Bunea et al.40 while analysing anthocyanins, carotenoids and antioxidant activity of coloured commercially available juices. The addition of encapsulated probiotic powder did not have any significant effect on the carotenoid concentration of the two beverages.Anthocyanins are antioxidants with analgesic, neuroprotective and anti-inflammatory properties. Their stability and color are significantly influenced by oxygen, pH, temperature, light and interactions with other chemicals including metal ions.41 The anthocyanin content between the two beverages varied significantly (p ≤ 0.05) and is shown in Table 3. The highest anthocyanin content (23.01 mg/100 mL) was found in the apple beverage and the lowest was found in the apple-carrot blend beverage (10.70 mg/100 mL). However, no significant (p ≥ 0.05) difference was found in different treatments of apple beverage (23.01 mg/100 mL) and apple-carrot blend beverage (10.70 to 10.72 mg/100 mL) containing encapsulated probiotics. The observed difference in anthocyanin content between apple and apple-carrot blend beverages might be attributed to the inherent anthocyanin concentration in the respective raw materials. Apples contain a higher concentration of anthocyanins, whereas orange-colored carrots are naturally low in anthocyanins, as their pigmentation is mainly due to carotenoids rather than anthocyanins. The results are consistent with those by Yoo et al.42 who reported that orange coloured carrots have low anthocyanin content. Furthermore, the incorporation of encapsulated probiotic powder did not significantly alter the anthocyanin concentration of the two beverages. Similar results were reported by Dobson et al.43 who observed no change in the initial anthocyanin content of fruit-based beverages.
3.5. Color (L* a* b*)
Color is one of the vital quality parameters of fruits and vegetables and is a critical factor affecting consumer acceptance of the product. The L* value ranges from 0 (black) to 100 (white), the positive and negative a* value implies redness and greenness and the b* value measures blueness to yellowness of the sample. Data in Table 4 represent the L* a* b* values of apple and apple-carrot blend beverages (controls and enriched with encapsulated probiotics). The L* value of the control apple beverage was 63.84 and the L* value of the control apple-carrot blend beverage was 56.12. The significantly (p ≤ 0.05) higher L* value of the apple beverage could be attributed to its lighter colour. The L* value of the apple beverage with encapsulated probiotics decreased significantly (p ≤ 0.05) between 63.31 and 61.13. Also, the L* value of the apple-carrot blend beverage with encapsulated probiotics decreased significantly (p ≤ 0.05) between 56.12 and 53.96. The decrease in the L* value by the addition of encapsulated probiotics in apple and apple-carrot blend beverages might be ascribed to the improved turbidity caused by the wall materials viz gum arabic, soy protein and the blend of the two, used for the encapsulation of probiotics. The findings of the present study are in direct agreement with the results of Aghajanzadeh et al.44 who reported a decrease in the L* value of watermelon juice by the addition of xanthan gum.| Treatment product | T0 | T1 | T2 | T3 | T4 | T5 | T6 | T7 | T8 | T9 | T10 | T11 | T12 | T13 | T14 | T15 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Values are expressed as mean ± standard deviation. b Values with different superscripts (capital letters) (within columns) differ significantly (p < 0.05). | ||||||||||||||||
| L* value | ||||||||||||||||
| Apple beverage | 63.84 ± 0.01dB | 63.31 ± 0.05dB | 63.29 ± 0.09dB | 63.22 ± 0.06dB | 63.19 ± 0.02dB | 63.28 ± 0.07dB | 61.52 ± 0.02aB | 61.42 ± 0.04bB | 61.29 ± 0.08bB | 61.13 ± 0.03bB | 61.25 ± 0.06bB | 62.81 ± 0.08cB | 62.83 ± 0.11cB | 62.79 ± 0.10dB | 62.73 ± 0.07cB | 62.81 ± 0.04cB |
| Apple-carrot blend | 56.12 ± 0.05dA | 56.07 ± 0.06dA | 56.01 ± 0.08dA | 55.96 ± 0.07cA | 55.84 ± 0.09cA | 56.06 ± 0.10cA | 54.27 ± 0.11bA | 54.16 ± 0.09bA | 54.29 ± 0.03bA | 54.00 ± 0.08bA | 53.96 ± 0.04aA | 54.87 ± 0.06aA | 54.91 ± 0.09aA | 54.62 ± 0.10bA | 54.61 ± 0.07bA | 54.79 ± 0.11bA |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| a* value | ||||||||||||||||
| Apple beverage | 7.71 ± 0.11aA | 7.57 ± 0.13aA | 7.81 ± 0.15aA | 7.63 ± 0.17aA | 7.74 ± 0.12aA | 7.69 ± 0.14aA | 7.35 ± 0.16aA | 7.21 ± 0.18aA | 7.17 ± 0.19aA | 7.28 ± 0.15aA | 7.42 ± 0.16aA | 7.57 ± 0.14bA | 7.28 ± 0.17aA | 7.68 ± 0.13aA | 7.75 ± 0.15aA | 7.67 ± 0.12aA |
| Apple-carrot blend | 16.76 ± 0.04aB | 16.81 ± 0.07aB | 16.95 ± 0.08aB | 17.16 ± 0.09bB | 17.43 ± 0.06bB | 17.63 ± 0.05bB | 18.19 ± 0.07bB | 18.08 ± 0.10cB | 18.27 ± 0.08cB | 18.41 ± 0.07cB | 18.61 ± 0.10cB | 17.69 ± 0.06cB | 17.91 ± 0.08cB | 17.67 ± 0.04bB | 17.51 ± 0.07bB | 17.87 ± 0.10bB |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| b* value | ||||||||||||||||
| Apple beverage | 19.01 ± 0.04bA | 19.09 ± 0.06bA | 19.16 ± 0.02bA | 18.19 ± 0.07aA | 18.63 ± 0.08aA | 18.90 ± 0.04aA | 20.09 ± 0.03cA | 20.18 ± 0.05cA | 20.32 ± 0.07cA | 20.28 ± 0.09cA | 20.19 ± 0.06cA | 19.54 ± 0.10bA | 19.63 ± 0.05bA | 19.65 ± 0.02cA | 19.78 ± 0.04bA | 19.66 ± 0.08bA |
| Apple-carrot blend | 35.52 ± 0.02aB | 35.67 ± 0.04aB | 35.91 ± 0.06aB | 36.11 ± 0.05bB | 36.23 ± 0.07bB | 36.37 ± 0.03bB | 37.45 ± 0.06bB | 37.59 ± 0.08bB | 37.41 ± 0.09cB | 37.53 ± 0.07cB | 37.49 ± 0.04cB | 36.56 ± 0.05bB | 36.79 ± 0.09bB | 36.63 ± 0.07bB | 37.01 ± 0.05cB | 37.03 ± 0.09cB |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Browning index (420 nm) | ||||||||||||||||
| Apple beverage | 0.014 ± 0.01aA | 0.019 ± 0.04aA | 0.019 ± 0.07aA | 0.018 ± 0.02aA | 0.017 ± 0.03aA | 0.019 ± 0.10aA | 0.053 ± 0.07cA | 0.059 ± 0.07cA | 0.052 ± 0.08cA | 0.058 ± 0.07cdA | 0.059 ± 0.01dA | 0.041 ± 0.02bA | 0.047 ± 0.04bA | 0.45 ± 0.05bA | 0.042 ± 0.05bA | 0.043 ± 0.02bA |
| Apple-carrot blend | 0.027 ± 0.17aB | 0.032 ± 0.12aB | 0.039 ± 0.13aB | 0.035 ± 0.18aB | 0.037 ± 0.14aB | 0.034 ± 0.01aB | 0.101 ± 0.13cB | 0.109 ± 0.09dB | 0.104 ± 0.10cdB | 0.101 ± 0.11cB | 0.108 ± 0.15dB | 0.066 ± 0.09bB | 0.064 ± 0.11bB | 0.069 ± 0.17bB | 0.067 ± 0.07bB | 0.068 ± 0.08bB |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||||||||
| Turbidity (NTU*) | ||||||||||||||||
| Apple beverage | 150 ± 1.03aA | 156 ± 1.05aA | 153 ± 1.07aA | 151 ± 1.09aA | 157 ± 1.07aA | 155 ± 1.09aA | 275 ± 2.13cA | 270 ± 2.14cA | 279 ± 2.16cA | 277 ± 2.15cA | 275 ± 2.16cA | 233 ± 2.09bA | 230 ± 2.12bA | 236 ± 2.11bA | 239 ± 2.11bA | 235 ± 2.12bA |
| Apple-carrot blend | 228 ± 1.11aB | 236 ± 1.22aB | 231 ± 1.21aB | 232 ± 1.23aB | 235 ± 1.14aB | 238 ± 1.17aB | 435 ± 2.07cB | 437 ± 2.03cB | 431 ± 2.08cB | 439 ± 2.07cB | 438 ± 2.28cB | 305 ± 1.15bB | 302 ± 2.01bB | 302 ± 2.25bB | 304 ± 2.13bB | 304 ± 2.06bB |
The a* value of the control apple beverage and control apple-carrot blend beverage was 7.71 and 16.76 respectively (Table 4). A higher a* value was found in apple-carrot blend beverages and a lower a* value was in apple beverages. The higher a* value of the apple-carrot blend beverage revealed a significantly (p ≤ 0.05) higher redness in carrot. The a* value of different apple beverages with encapsulated probiotics ranged between 7.17 and 7.81. The a* value of different apple-carrot blend beverages with encapsulated probiotics ranged between 16.81 and 18.19. The apple-carrot blend beverages have a higher a* value as compared to apple beverages, which might be due to the high carotenoid concentration in carrots. The increase in the a* value in apple and apple-carrot blend beverages with probiotics might be attributed to the wall material that decreases the lightness of the two beverages.
The b* chromaticity coordinate represents the yellow-blue attribute of the two beverages, with positive values indicating yellowness and negative values signifying blueness. A significant (p ≤ 0.05) difference in the b* value was seen between the two beverages (Table 4). The b* value of the control apple beverage was 19.01 and that of the control apple-carrot blend beverage was 35.52. The b* value of apple beverages with encapsulated probiotics increased and was between 18.19 and 20.32. Also, the b* value of apple-carrot blend beverages containing encapsulated probiotics ranged between 35.67 and 37.59. The higher b* value was found in the beverages that contain probiotics encapsulated in soy protein isolate as compared to the beverages containing probiotics encapsulated in gum arabic and the combination of the two wall materials. This could be due to the dark colour of soy protein. Similar results for a* and b* values were also reported by Ajlouni & Bhoi,45 while examining the survivability of both free and encapsulated probiotics in fruit juices throughout refrigeration and in vitro gastrointestinal digestion.
3.6. Browning index and turbidity
The browning index (BI) is a measure of enzymatic browning brought about by the interaction of phenolic compounds with enzymes that produce brown color and is quantified through absorbance and reflects intensity of the pigment. This index is essential for assessing quality, shelf life, and consumer preference of food products. The browning index of control apple and control apple-carrot blend beverages varies significantly (p ≤ 0.05) and was 0.014 and 0.027 respectively (Table 4). The BI of different apple beverages with encapsulated probiotics was in the range of 0.017 to 0.059 and the BI of apple-carrot blend beverages with encapsulated probiotics ranged between 0.032 and 0.109. The higher browning index was found in beverages with probiotics entrapped in the soy protein isolate followed by beverages that contain probiotics encapsulated in the mixture of gum arabic and soy protein. The lowest BI was found in the beverages that contain probiotics encapsulated in the gum arabic. The higher BI of the beverages might be attributed to the effect of wall materials on the beverage. The addition of encapsulated probiotic powder decreases the lightness of beverages and therefore increases the darkness of the beverages. The outcomes of the present study are consistent with those reported by Yousefi et al.,46 who investigated how carrier type and spray drying affected the physicochemical characteristics of powdered and reconstituted pomegranate juice (Punica granatum L.).Turbidity is used to assess the degree of light dispersed by colloidal and suspended polysaccharides like starch, pectin and hemicellulose components. It can show the extent of browning under certain conditions. The turbidity of the control apple beverage and control apple-carrot blend beverage varies significantly (p ≤ 0.05) and was found to be 150 and 228 respectively (Table 4). The highest turbidity was found in the control apple-carrot beverage and lowest was in the control apple beverage. The initial turbidity might be due to the colloidal particles like pectin and partially dissolved insoluble substances suspended in the beverages.42 The turbidity of different apple beverages containing encapsulated probiotics displayed a significant (p ≤ 0.05) difference and ranged between 151 and 279. Also, the turbidity of different apple-carrot beverages with encapsulated probiotics varies significantly (p ≤ 0.05) between 231 and 439. The addition of probiotics encapsulated in different wall materials showed a significant effect on turbidity of both the beverages. The increase in turbidity with the addition of probiotic powder could be due to the electrostatic force between the cloud particles suspended in the beverage and molecules of the wall materials. Consequently, electrostatic repulsion among the negatively charged particles and wall material molecules may result from the addition of encapsulated probiotic powder to the beverage. As a result, it is possible to prevent the molecules in the beverage from aggregating, maintaining the particle suspension and increasing turbidity and cloud stability. The same results were found by Tuan Azlan et al.,47 while analysing the impact of adding gum arabic to roselle juice on physicochemical and organoleptic properties.
3.7. Viability of encapsulated probiotics
Viability is a key factor in defining the efficacy and quality of functional products, especially those containing probiotics. It is imperative to confirm a high probiotic survival rate throughout production and storage of the product, in order to maintain consumer confidence in the probiotic product.48 The viability of microencapsulated probiotics in the apple beverages and apple-carrot blend beverages is shown in Fig. 1. The initial viable count of the five probiotic strains encapsulated in the three different wall materials ranged between 8.38 and 9.52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log CFU mL−1. However, these viable counts showed no significant (p ≥ 0.05) decline (<1
log CFU mL−1. However, these viable counts showed no significant (p ≥ 0.05) decline (<1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log CFU mL−1) in both the apple beverage and apple-carrot blend beverages. The encapsulated probiotics did not lose their viability in the beverages. This is likely due to the protective effect of encapsulation, which creates a favourable atmosphere for the sensitive probiotic bacteria and acts as a physical barrier against acidic conditions of the beverage. Similar results were observed by Ding & Shah,26 while analysing existence of free and encapsulated probiotic bacteria in orange and apple juices. Ajlouni & Bhoi45 also reported <1
log CFU mL−1) in both the apple beverage and apple-carrot blend beverages. The encapsulated probiotics did not lose their viability in the beverages. This is likely due to the protective effect of encapsulation, which creates a favourable atmosphere for the sensitive probiotic bacteria and acts as a physical barrier against acidic conditions of the beverage. Similar results were observed by Ding & Shah,26 while analysing existence of free and encapsulated probiotic bacteria in orange and apple juices. Ajlouni & Bhoi45 also reported <1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log CFU mL−1 reduction in viable counts on day 1 of storage in apple juice and tomato juice supplemented with encapsulated probiotics.
log CFU mL−1 reduction in viable counts on day 1 of storage in apple juice and tomato juice supplemented with encapsulated probiotics.
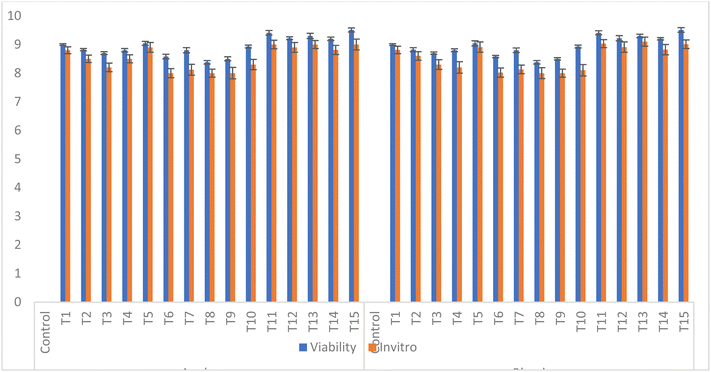 | ||
| Fig. 1 Viability of probiotics in apple and apple-carrot blend beverages and under simulated gastrointestinal conditions. | ||
These findings are consistent with other studies using plant-based systems as probiotic carriers, where high survival rates have been reported during storage and simulated digestion, including cashew by-products in fermented milk and fruit matrices49 and red pitaya pulp.3
Viability study of probiotics under simulated gastrointestinal conditions is a key component to determine the capability of probiotic strains to survive and colonize in the human digestive system. The gastric and intestinal digestion of apple beverages and apple-carrot blend beverages was conducted and the results are shown in Fig. 1. From the figure, a significant (p ≤ 0.05) decrease in the probiotic count from 7.33 to 8.49 and from 7.35 to 8.51 log CFU mL−1 was observed in most treatments after the in vitro digestion of apple beverages and apple-carrot blend beverages containing encapsulated probiotics. The highest reading for simulated gastric conditions (7.96 to 8.51) was observed in treatments T1, T5, T11, T13 and T15. The maximum probiotic survival rate during exposure to gastrointestinal conditions might be attributed to the acid resistance of probiotic strains and type and concentrations of encapsulating materials in the matrix. The results highlight how gum arabic and soy protein work together to enhance probiotic effectiveness in low-acid environments, acting as efficient carriers for the passage of live probiotic cells from the stomach into the lower intestines. The same was found by Nazir et al.,50 while examining the behaviour and survival of probiotic bacteria in a non-dairy matrix and under in vitro GIT conditions that are enclosed by internal gelation.
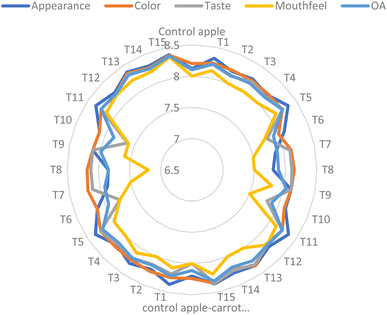
3.8. Sensory evaluation
The sensory properties of apple beverages and carrot blend beverages with and without encapsulated probiotics are illustrated in Fig. 2. The acceptability of the beverages was evaluated based on their appearance, colour, taste, mouthfeel and overall acceptability characteristics on a 9-point hedonic scale. The findings demonstrated that all samples had mean hedonic ratings of above 7.20 for every attribute, falling within the acceptable range.The score for appearance of the control apple and control apple-carrot blend beverage presented a significant (p ≤ 0.05) difference. The appearance of different apple beverages and apple-carrot blend beverages containing probiotics encapsulated in gum arabic, soy protein and their combination ranged between 7.80 and 8.36 and 7.84 and 8.38 respectively. The highest score of appearance (8.24 to 8.35) was observed in the beverages containing probiotics encapsulated in the combination of gum arabic and soy protein followed by beverages containing probiotics encapsulated in gum arabic. The appearance score was the least (7.80 to 8.14) for beverages containing probiotics encapsulated in soy protein.
For the colour attribute, the apple beverages and apple-carrot blend beverages with or without encapsulated probiotics attained a non-significant (p ≥ 0.05) acceptability score. The different apple-carrot blend beverages showed the highest score (8.00 to 8.35) for colour as compared to apple beverages (8.10 to 8.37). The highest score for colour may be attributed to the carrot percent in the blend beverage that led to improved color. Moselhy et al.51 also reported a higher score for colour in juices containing aloe vera gel and probiotics. However, the wall material did not have much effect on the colour of beverages.
For the taste attribute, all the beverages (apple and apple-carrot blend) with and without encapsulated probiotics acquired a similar score. This could be attributed to the taste of fruit used for the preparation of the beverage. The encapsulated probiotics did not have an effect on the taste of beverages. The lack of a noticeable taste change in beverages containing encapsulated probiotics is likely due to the inherently tasteless nature of encapsulation materials like gum arabic and soy protein.52 Similar results were found by Tuan Azlan et al.47 They claimed that during the sensory assessment, the taste was not considerably changed by the addition of gum arabic (Acacia senegal) to roselle juice.
Mouthfeel is an essential sensory characteristic. The data of mouthfeel indicated that all the apple and apple-carrot blend beverages have acceptable scores between 7.20 and 8.24. However, the highest score (8.12 to 8.24) was for the beverage fortified with probiotics encapsulated in the combination of soy protein and gum arabic followed by the beverages fortified with the probiotics encapsulated in gum arabic. The use of gum arabic alone and the combination of gum arabic and soy protein as wall materials contributed to a smooth and uniform mouthfeel of the beverage, thus preventing phase separation or sedimentation. The lowest score (7.20 to 7.80) for mouthfeel was observed in the beverages with probiotics encapsulated in soy protein.
The overall acceptability (OA) attribute of all the beverages (apple and apple-carrot blend beverages) with or without encapsulated probiotics ranged between 7.84 and 8.36. The higher OA score (8.22 to 8.36) was observed for the apple-carrot blend beverages with probiotics encapsulated in the mixture of gum arabic and soy protein isolate. Therefore, the findings showed that addition of encapsulated probiotics has the least impact on the organoleptic properties of the beverage, thus supporting the feasibility of plant-based matrices as carriers for probiotics without compromising nutritional or sensory quality.
4. Conclusion
Fruit and vegetable-based beverages are recognized as a suitable carrier for probiotics due to their nutritional value and consumer acceptability. Beverages fortified with encapsulated probiotics have gained attention due to their potential health benefits. This study investigates the effect of encapsulated probiotics on the physicochemical and sensory attributes of apple and apple-carrot blend beverages. Also, the viability of probiotics was assessed in both beverage matrices and under simulated gastrointestinal conditions. The findings showed that encapsulation greatly increased probiotic viability while maintaining the sensory appeal of beverages. Physicochemical measures such as total soluble solids, pH, titratable acidity, total sugars, antioxidant activity, and total phenolic content showed slight changes. Overall, the results demonstrate the efficacy of encapsulation as a viable method for safeguarding probiotics and improving their stability in beverages prepared from fruits and vegetables.Ethical statement
Ethical permission was not required for this study. The authors confirm that appropriate protocols for protecting the rights and privacy of all participants were utilized during the execution of the research. Prior informed consent was obtained from all participants, and no personal data was disclosed without prior knowledge and consent.Author contributions
Zahida Naseem: writing – original draft, investigation, formal analysis, data curation. Sajad Mohd Wani: supervision, conceptualization, validation. Sajad Ahmad Mir: writing – review & editing, data curation. Naseh Nisar: writing – review & editing.Conflicts of interest
The authors declare no conflict of interest.Data availability
All the data generated and analyzed during this study are included in the manuscript.References
- F. D. Mojikon, M. E. Kasimin, A. M. Molujin, J. A. Gansau and R. Jawan, Probiotication of nutritious fruit and vegetable juices: an alternative to dairy-based probiotic functional products, Nutrients, 2022, 14(17), 3457 Search PubMed.
- M. Afzaal, F. Saeed, M. Saeed, A. Ahmed, H. Ateeq, M. T. Nadeem and T. Tufail, Survival and stability of free and encapsulated probiotic bacteria under simulated gastrointestinal conditions and in pasteurized grape juice, J. Food Process. Preserv., 2020, 44(3), e14346 Search PubMed.
- J. Y. Suzuki, M. E. Herkenhoff, O. Brödel, A. C. C. Cucick, M. Frohme and S. M. I. Saad, Exploring the potential of red pitaya pulp (Hylocererus sp.) as a plant-based matrix for probiotic delivery and effects on betacyanin content and flavoromics, Food Res. Int., 2024, 192 Search PubMed.
- A. Bevilacqua, D. Campaniello, M. R. Corbo, L. Maddalena and M. Sinigaglia, Suitability of Bifidobacterium spp. and Lactobacillus plantarum as probiotics intended for fruit juices containing citrus extract, J. Food Sci., 2013, 78(11), M1764–M1771 Search PubMed.
- A. S. M. Al-Muslhi and H. L. Ali, Assessing the impact of pomegranate juice on liver tissue in male rats exposed to a commercial energy drink, J. Glob. Innov. Agric. Sci., 2024, 12, 417–426 CrossRef.
- G. M. Da Costa, J. V. C. Silva, J. D. Mingotti, C. E. Barao and S. J. Klososki, Effect of ascorbic acid or oligofructose supplementation on L. paracasei viability, physicochemical characteristics and acceptance of probiotic orange juice, LWT-Food Sci. Technol., 2017, 75, 195–201 Search PubMed.
- N. P. Shah, W. K. Ding, M. J. Fallourd and G. Leyer, Improving the stability of probiotic bacteria in model fruit juices using vitamins and antioxidants, J. Food Sci., 2010, 75(5), M278–M282 Search PubMed.
- D. Ying, S. Schwander, R. Weerakkody, L. Sanguansri and C. Gantenbein-Demarchi, Microencapsulated Lactobacillus rhamnosus GG in whey protein and resistant starch matrices: Probiotic survival in fruit juice, J. Funct. Foods, 2013, 5(1), 98–105 Search PubMed.
- E. F. Craparo, S. E. Drago, G. Giammona and G. Cavallaro, Production of polymeric micro-and nanostructures with tunable properties as pharmaceutical delivery systems, Polymer, 2020, 200, 122596 CrossRef CAS.
- M. K. Morsy, O. M. Morsy, M. A. Abdelmonem and R. Elsabagh, Anthocyanin-colored microencapsulation effects on survival rate of Lactobacillus rhamnosus GG, color stability, and sensory parameters in strawberry nectar model, Food Bioprocess Technol., 2022, 15(2), 352–367 Search PubMed.
- S. Moumita, K. Goderska, E. M. Johnson, B. Das and D. Indira, Evaluation of the viability of free and encapsulated lactic acid bacteria, LWT-Food Sci. Technol., 2017, 77, 460–467 CrossRef CAS.
- R. Karimi, A. M. Mortazavian and A. G. Da Cruz, Viability of probiotic microorganisms in cheese during production and storage: A review, Dairy Sci. Technol., 2011, 91(3), 283–308 Search PubMed.
- M. Safeer Abbas, M. Afzaal, F. Saeed, A. Asghar, L. Jianfeng, A. Ahmad and M. A. Shah, Probiotic viability as affected by encapsulation materials: Recent updates and perspectives, Int. J. Food Prop., 2023, 26(1), 1324–1350 Search PubMed.
- A. B. Oyenihi, Z. A. Belay, A. Mditshwa and O. J. Caleb, The potentials of apple bioactive constituents for chronic disease prevention, J. Food Sci., 2022, 87, 2291–2309 CrossRef CAS PubMed.
- A. Shakeel, M. Saeed, H. K. W. Aslam, N. Naheed and M. Shoaib, Optimization of colour and cloud stability of carrot juice by using hydrocolloids and antioxidant, J. Glob. Innov. Agric. Soc. Sci., 2015, 3, 84–89 CrossRef.
- M. E. Herkenhoff, I. U. D. De Medeiros, L. H. G. Garutti, M. K. Salgaço, K. Sivieri and S. M. I. Saad, Cashew by-product as a functional substrate for the development of probiotic fermented milk, Foods, 2023, 12(18), 3383 Search PubMed.
- Z. Naseem, S. A. Mir, S. M. Wani, S. A. Ganai and N. Nazir, Investigating gum arabic and soy protein isolate as wall material for encapsulation of five strains of Lactobacillus, Int. J. Biol. Macromol., 2025, 140083 Search PubMed.
- S. Ranganna, Handbook of Analysis and Quality Control for Fruit and Vegetable Products, Tata McGraw-Hill Education, 1986 Search PubMed.
- I. M. Bhat, S. M. Wani, S. A. Mir and Z. Naseem, Effect of microwave-assisted vacuum and hot air oven drying, Food Prod., Process. Nutr., 2023, 5(1), 26 Search PubMed.
- W. Brand-Williams, M. Cuvelier and C. Berset, Use of a free radical method to evaluate antioxidant activity, LWT-Food Sci. Technol., 1995, 28(1), 25–30 Search PubMed.
- T. Assad, Z. Naseem, S. M. Wani, A. Sultana and I. Bashir, Impact of ultrasound assisted pretreatment, Ultrason. Sonochem., 2025, 112, 107194 Search PubMed.
- Z. Naseem, A. Gull, S. A. Wani, F. A. Masoodi and T. A. Ganaie, Changes in anthocyanin, total phenolic, antioxidant activity, Postharvest Biol. Technol., 2017, 5(2), 79–88 Search PubMed.
- A. Bonaccorso, N. Russo, A. Romeo, C. Carbone and M. A. Grimaudo, Coating Lacticaseibacillus rhamnosus G, AAPS PharmSciTech, 2021, 22(1), 1–14 Search PubMed.
- W. Krasaekoopt and S. Watcharapoka, Effect of prebiotics on microencapsulated probiotics in fruit juice, LWT-Food Sci. Technol., 2014, 57, 761–776 CrossRef CAS.
- E. Ephrem, A. Najjar, C. Charcosset and H. Greige-Gerges, Encapsulation of natural active compound, J. Funct. Foods, 2018, 48, 65–84 CrossRef CAS.
- W. K. Ding and N. P. Shah, Survival of free and microencapsulated probiotic bacteria, Int. Food Res. J., 2008, 15(2), 219–232 Search PubMed.
- C. Tyl and G. D. Sadler, pH and titratable acidity, Food Anal., 2017, 389–406 Search PubMed.
- E. Yusuf, K. Tkacz, I. P. Turkiewicz, A. Wojdyło and P. Nowicka, Analysis of chemical compounds in carrots, Eur. Food Res. Technol., 2021, 247, 3053–3062 CrossRef CAS.
- O. G. Nwaokoro and C. T. Akanbi, Effect of hydrocolloids on tomato-carrot juice blend, J. Nutr. Health Food Sci., 2015, 3(1), 1–10 Search PubMed.
- M. Yousefi, E. Khanniri, N. Khorshidian, S. Sohrabvandi and A. M. Mortazavian, Development of probiotic apple juice, Appl. Food Biotechnol., 2023, 10(3), 205–213 CAS.
- E. Aprea, M. Charles, I. Endrizzi, M. L. Corollaro and E. Betta, Sweet taste in apple, Sci. Rep., 2017, 7(1), 44950 CrossRef CAS PubMed.
- T. M. Gocan, D. N. Măniuţiu, I. Andreica, S. Balcău, V. Lazăr and I. Bogdan, Sugar content of carrot roots influenced by sowing period, J. Hortic. Biotechnol., 2013, 17(1), 66–69 Search PubMed.
- H. S. Mostafa, M. R. Ali and R. M. Mohamed, Production of probiotic date juice with anti-proliferative activity, Food Sci. Technol., 2020, 41(1), 105–115 Search PubMed.
- F. D. Krumreich, A. P. A. Corrêa, J. C. Nachtigal, G. L. Buss and J. K. Rutz, Stabilization of guava nectar with hydrocolloids, Polímeros, 2018, 28(1), 53–60 CrossRef.
- S. Grubišić, M. Kristić, M. Lisjak, K. Mišković Špoljarić and S. Petrović, Effect of wheatgrass juice on nutritional quality of mixed juices, Foods, 2022, 11(3), 445 CrossRef PubMed.
- B. M. Lepaus, A. K. P. O. Santos, A. F. Spaviero, P. S. Daud and J. F. B. de São José, Thermosonication of orange-carrot juice blend, Molecules, 2023, 28(5), 2196 CrossRef CAS PubMed.
- G. Giomaro, A. Karioti, A. R. Bilia, A. Bucchini and L. Giamperi, Polyphenols and antioxidant activity in apple, Chem. Cent. J., 2014, 8, 1–10 Search PubMed.
- J. Gao and H. V. Rupasinghe, Quality of ultrasound-treated apple-carrot juice blends, Food Nutr. Sci., 2012, 3(2), 212–218 Search PubMed.
- H. Wang, Y. Tian, Y. Li, J. Wei and F. Ma, Carotenoids and gene expression in apple, J. Agric. Food Chem., 2023, 71(41), 15121–15131 CrossRef CAS PubMed.
- A. Bunea, S. Andrei, C. El-Mahdi, A. Cucerdean and F. D. Bora, Anthocyanins, carotenoids and antioxidant activity of juices, Stud. Univ. Babes-Bolyai, Chem., 2019, 64(2), 111–126 Search PubMed.
- M. Abid, S. Jabbar, T. Wu, M. M. Hashim and B. Hu, Sonication enhances quality of apple juice, Ultrason. Sonochem., 2014, 21(1), 93–97 CrossRef CAS PubMed.
- K. S. Yoo, H. Bang, L. Pike, B. S. Patil and E. J. Lee, Comparing carotene, anthocyanins, and terpenoid in carrot lines, Hortic., Environ. Biotechnol., 2020, 61, 385–393 CrossRef CAS.
- G. Dobson, G. J. McDougall, D. Stewart, M. Á. Cubero and R. O. Karjalainen, Stability of black currant anthocyanins during storage, J. Food Sci., 2017, 82(1), 44–52 CrossRef CAS PubMed.
- S. Aghajanzadeh, A. M. Ziaiifar and M. Kashaninejad, Influence of processing on watermelon juice, LWT-Food Sci. Technol., 2017, 85, 66–74 CrossRef CAS.
- S. Ajlouni and Y. Bhoi, Survival of encapsulated vs free probiotics in fruit juices, J. Dairy Res. Technol., 2024, 6(1), 018–028 Search PubMed.
- S. Yousefi, Z. Emam-Djomeh and S. M. Mousavi, Spray drying effects on pomegranate juice, J. Food Sci. Technol., 2011, 48, 677–684 CrossRef PubMed.
- T. Tuan Azlan, Y. Hamzah and H. Abd Majid, Effect of gum arabic on roselle juice, Food Res., 2020, 4(2), 449–458 Search PubMed.
- M. K. Tripathi and S. K. Giri, Probiotic functional foods: survival of probiotics, J. Funct. Foods, 2014, 9, 225–241 CrossRef CAS.
- M. E. Herkenhoff, I. U. D. de Medeiros, L. H. G. Garutti, M. K. Salgaço, K. Sivieri and S. M. I. Saad, Prebiotic and probiotic potential of fermented milk with cashew (Anacardium occidentale) by-products evaluated in microbiome model, Food Biosci., 2024, 61, 104669 CrossRef CAS.
- S. Nazir, M. Afzaal, F. Saeed, A. Ahmad and H. Ateeq, Survivability of encapsulated probiotics in non-dairy matrix, PLoS One, 2024, 19(6), e0303091 CrossRef CAS PubMed.
- S. N. Moselhy, A. A. Al-Nashwi, E. Raya-Álvarez, F. O. Abu Zaid and H. S. T. Shalaby, Properties of aloe vera probiotic juices and antidiabetic effects, Front. Nutr., 2024, 11, 1328548 CrossRef PubMed.
- A. Cittadini, P. E. S. Munekata, M. Pateiro, M. V. Sarriés and R. Domínguez, Encapsulation techniques to increase lipid stability, Food Lipids, 2022, 413–459 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |