Open Access Article
Open Access ArticleMicroplastic release from food processing to the environment: contamination pathways, health implications, and sustainability perspectives
Md Sabir
Ahmed Mondol
a,
Mehvish
Ayoub
b,
Ubaida
Akbar
c,
Kshirod Kumar
Dash
 d,
Aamir Hussain
Dar
d,
Aamir Hussain
Dar
 *e and
Urba Shafiq
Sidiqi
e
*e and
Urba Shafiq
Sidiqi
e
aDepartment of Agricultural Biochemistry, Bidhan Chandra Krishi Viswavidyalaya, Mohanpur, Nadia, West Bengal, India
bDepartment of Food Science & Technology, University of Kashmir, Srinagar, India
cDepartment of Food Technology and Nutrition, Lovely Professional University, Phagwara, Punjab, India
dDepartment of Food Engineering and Technology, Ghani Khan Choudhury Institute of Engineering and Technology Malda, West Bengal, India
eDepartment of Food Technology, Islamic University of Science & Technology, Kashmir, India. E-mail: Kshirod12@gmail.com; daraamirft@gmail.com
First published on 19th March 2026
Abstract
Microplastics have rapidly emerged as a key food safety and environmental concern, however their connection with food processing, packaging and human health have not been supported yet with substantial study reported in the literature. Defined as synthetic polymer particles under 5 mm, MPs originate both from intentional manufacturing and from the degradation of larger plastics. This review provides an updated and interdisciplinary perspective by examining the pathways through which MPs migrate from processing equipment, packaging materials, and storage environments into foods, and how these particles subsequently disperse into terrestrial, aquatic, and atmospheric systems. Human exposure to MPs is discussed, primarily through the ingestion of contaminated foods and beverages alongside inhalation and dermal routes. We further summarize available evidence on the toxicological effects of MPs, including oxidative stress, inflammation, and metabolic disruption, and their potential role as the carriers of hazardous chemicals. Advances in detection methods, such as spectroscopic and chromatographic approaches, are critically evaluated, with an emphasis on the current lack of standardized detection protocols. Finally, the review highlights knowledge gaps, regulatory needs, and innovative mitigation strategies, including sustainable alternatives to plastics. By linking food processing, contamination pathways, health implications, and detection strategies, this review offers a comprehensive outlook designed to inform future research, regulatory frameworks, and industrial practices.
Sustainability spotlightThis review highlights the urgent need to integrate sustainability principles into food processing and packaging systems to mitigate microplastic pollution, a critical yet often overlooked sustainability challenge. By tracing the pathways of microplastics from food contact materials and industrial processes to environmental and human exposure routes, the current study highlights the coherence between food safety, ecological integrity, and public health. This review highlights how transitioning toward biodegradable alternatives, circular economy models, and improved waste management strategies can substantially reduce the production of microplastics across the food chain. Moreover, it paves way for research opportunities in green materials, green processing technologies and regulatory harmonization in line with sustainability development goals. Through its holistic synthesis of contamination mechanisms, toxicological insights, and mitigation strategies, this study advances the sustainable transformation of food systems, providing a scientific basis for collaboration among policy, industry, and research toward a plastic-resilient future. |
1. Introduction
The numerous benefits of plastics, including their low-cost manufacturing, resistance to degradation, waterproof nature, light weight, and high durability, have resulted in their widespread applications across various industries.1,2 It is therefore envisaged that the worldwide consumption of plastics would increase considerably, from 464 Mt in 2020 to an estimated 884 Mt in 2050, with an approximate accumulative amount of over 4725 Mt of waste plastics generated from 2000 to 2050.3 Notwithstanding the advancements in recycling technologies and waste disposal measures, only 21% of global plastic wastes are currently being recycled or incinerated, and the majority of them ends up piling up in terrestrial and marine ecosystems.4,5 Plastics, which were obsolete until the 1950s, acquired immense industrial significance later and are today irreplaceable in different applications such as packaging and building applications, electronics, and medical applications due to their remarkable mechanical strength, thermal resistance, and low costs.6 The widespread presence of these materials in diverse ecosystems, combined with improper disposal practices, has led to the substantial release of microplastics, synthetic polymer particles smaller than 5 mm in diameter.7 MPs originate as primary microplastics, which include intentionally manufactured microscale particles and micro-sized fragments formed during the use of consumer and industrial products, and secondary microplastics, which result from the fragmentation of larger plastic debris.8 From 1950 to 2015, an estimated 6.3 billion tons of plastic materials were produced globally, of which a mere 9% was subjected to recycling processes, 12% was incinerated, and the remaining 79% was disposed of in landfills or directly released into the environment.9 Currently, over 380 million tons of plastics is being produced worldwide annually, and a substantial amount of these is earmarked for single-use packaging purposes. Upon degradation, the plastics disperse as microplastics and still smaller nanoplastics within the immediate environment. These are found in seawater, rivers, soils, particulate matter within the atmosphere, sea salts, potable waters, seafood, and an array of edibles, including the digestive tracts of marine lifeforms.8 As a consequence of its minuscule nature and far-reaching distribution, microplastics stand a high chance of being ingested or inhaled by humans during routine eating, drinking, and breathing.10 Recent analyses indicate the smallest particles have the capacity to cross biological membrane barriers, infiltrate cellular structures, interfere with cellular activities, and evoke inflammatory reactions.8 However, the extent to which the prevalent microplastics in human tissues will give rise to substantive consequences on health is not clearly known. This continuing ambiguity can be attributed to ethical constraints associated with sampling methodologies, technological impediments in microplastic detection, and the scarcity of suitable long-term experiments on human subjects.8,9 The health risks associated with microplastics depend mainly on the size of the particles, their chemical nature, the duration of exposure and the biological nature of the tissue that is exposed.11,12 The fact that microplastics can adversely affect cellular structures, tissues, and whole organ systems in vitro under highly controlled laboratory conditions, and thus with a high degree of credibility, raises grave concerns over human health issues. Since there is growing evidence of the occurrence of MP in the food system, the purpose of this review is to.• Systematically examine the primary and secondary pathways through which microplastics originate, migrate, and accumulate in food processing, packaging, distribution, and storage systems.
• Evaluate human exposure routes and associated toxicological implications, with emphasis on ingestion, inhalation, and dermal contact linked specifically to food-processing and food-contact environments.
• Assess current analytical techniques and emerging sustainable strategies to detect, monitor, and mitigate microplastics across the food industry, while identifying gaps that require future research.
2. Sources of microplastic contamination in food processing
2.1. Primary sources of microplastics in food processing
Microplastics could potentially contaminate food products at various stages in the processing environment, especially when food ingredients come into contact with equipment, surfaces, or process water that is contaminated with microplastics (Fig. 1 and Table 1).13,14 The use of plastic in food processing is widespread due to its flexibility, ease of cleaning, chemical resistance, and cost-effectiveness; however, plastics are prone to degradation due to mechanical, thermal, and chemical stress. To ensure that the food production process is clearly understood and the role of microplastics in food products is properly illustrated, the addition of microplastics to food products is described below in five major stages of food processing: raw material processing, washing, mixing and grinding, heat processing, and filling and sealing.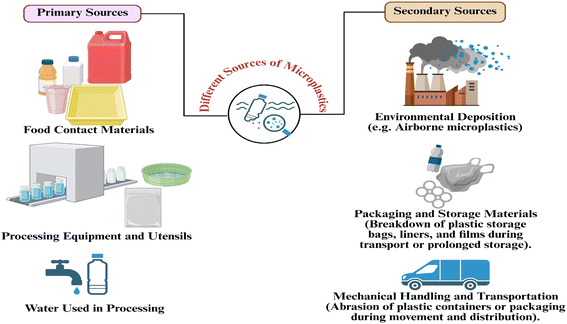 | ||
| Fig. 1 Primary (within processing lines) and secondary (after packaging and distribution) sources of microplastics in food processing and handling. | ||
| Processing stage | How microplastics enter | Typical polymer types | Source of microplastic release | Examples of food products affected | References |
|---|---|---|---|---|---|
| Raw material handling (harvesting, unloading, sorting) | Abrasion from crates, bins, mesh bags; environmental deposition; soil and water adherence | PE, PP, PA, PES | Transport crates, packaging sacks, mesh bags, contaminated soil/water | Fruits, vegetables, grains, nuts, seafood | 12–15,18,20,21,25–27 |
| Washing and cleaning | Release from hoses, tanks, PVC pipes, gaskets, pump seals; contaminated wash water | PVC, PP, PE | Water hoses, cleaning tanks, pipe linings, rubber/plastic seals | Fresh produce, fish, meat, spices | |
| Cutting, peeling, grinding, slicing | Wear from cutting boards, blades with polymer coatings, HDPE surfaces; conveyor belt friction | HDPE, LDPE, PP | Cutting boards, conveyor belts, polymer-coated blades | Meat cuts, vegetables, cheese, spices, flour | |
| Mixing, blending, kneading | Shedding from mixers, paddles, polymer-coated shafts, lining materials | PP, PE, PA | Mixer bowls, paddles, internal linings, rotating shafts | Dough, batter, sauces, dairy mixtures | |
| Thermal processing (boiling, frying, blanching, pasteurizing) | Heat-induced degradation of plastic utensils, non-stick coatings, polymer containers | PTFE, PP, PE | Non-stick cookware, polymer ladles, food-grade plastics exposed to heat | Ready-to-eat meals, soups, bakery fillings | |
| Filtration, sieving, straining | Release from nylon or polyester mesh filters, cloth filters, sieves | PA (nylon), PES, PP | Strainers, filter cloths, nylon mesh | Tea, coffee, juices, milk products | |
| Packaging (primary and secondary) | Migration from films, trays, bottles, caps, laminates; tearing and sealing processes | PET, PP, PS, PVC | Plastic films, multilayer laminates, PET bottles, PP caps, PS trays | Bottled water, snacks, meat, bakery items | |
| Filling and sealing | Friction at sealing jaws, cap torque application, wearing of liners and gaskets | PP, PET, PVC | Sealers, capping machines, gaskets, liners | Beverages, oils, sauces, spreads | |
| Cold chain and storage | Film cracking, tray deformation, freezer brittleness; abrasion during stacking | PS, PE, PP | Freezer trays, PE wraps, cold storage packaging | Frozen foods, dairy, seafood | |
| Transport and distribution | Vibration-induced abrasion of packaging materials and pallet films | PE, PP, PET | Pallet stretch wrap, secondary packaging films | All packaged products | |
| Retail display and handling | Repeated opening, surface rubbing, environmental deposition, film wear | PE, PP, PES | Wrapper films, retail trays, display packaging | Fresh produce, bakery, meat | |
| Consumer food preparation | Heating in plastic containers; microwave-induced wear; cutting on plastic boards | PP, PE, PS | Microwave containers, plastic bowls, cutting boards | Ready meals, leftovers, reheated foods |
2.2. Secondary sources of microplastics
Secondary contamination takes place after the food has exited the production line and is primarily linked with the decay of packaging containers during storage, transportation, distribution and retailing processes. Whereas primary sources are the result of direct interaction of food and processing equipment, secondary sources involve the release of microplastics due to physical loads on the packaged food, temperature variation, vibration, or long durations of storage. Here, packaging performs quite differently compared to the on-line packaging steps outlined in Section 2.1.5. On-line packaging occurs in the processing space and it entails direct contact with the equipment, so it is a primary source. Conversely, the degradation of packaging materials that occurs once the processing is finished comes out as a secondary source since it does not occur on the production line. Packaging materials such as expanded polystyrene, plastic-lined packaging, bottle caps, and closure systems have the potential to leach microplastics. The degradation of these plastics is increased when they are subjected to heat, friction, pressure, or storage conditions, and it is easy for microplastic particles to migrate into ready-to-eat and stored foods.23,24 Indicatively, packaging materials of fast food and plastic-lined paper wrappers and trays have been found to accumulate and liberate microplastics, particularly when subjected to high temperatures or when stored for longer durations.16 Another source of secondary contamination is through environmental deposition. Microplastics that are present in indoor airways, especially microfibers in clothing, plastic dust and parts of ventilation systems, will tend to deposit on food surfaces. In certain cases, where air filtration is low or storage facilities are not optimal, it is believed that the likelihood of microplastics being present in the surrounding air increases substantially. Further release of microplastics may also occur due to improper handling or through contact of the surfaces. During food transportation and distribution, mechanical stress, temperature fluctuations, and continuous handling of packaging materials can accelerate their degradation, leading to the release and transfer of microplastics from the packaging into the surrounding environment or the food itself. As an example, plastic packages used for rice, dried spices and dehydrated food may fracture during long-distance transportation, releasing even more plastic particles into these foods.11,162.3. Influence of the food type
The extent of microplastic pollution depends on the type of food, and the marine foods are more susceptible to microplastic contamination. Especially small fish consumed as whole poses serious health risks, due to the accumulation of the microplastics in the gastrointestinal tract of seafoods.28 Bivalves and filter-feeding organisms are particularly vulnerable owing to the feeding modality of these organisms, which tends to encourage the ingestion of plankton-sized pellets of plastic. Various commercially available food items, such as fish, sea salt, honey, beer, and bottled water, have been reported to contain detectable levels of MPs.7,29,30 Contamination of foods can arise not only from exposure to the environment but also from contact with processing lines and packaging material surfaces.31 In addition, inhalation of MPs and dust airborne during food packaging and processing is also a growing exposure pathway.29 Furthermore, microplastics are made of diverse polymeric materials, produced at microscale level due to the degradation of macroplastics. In seawaters, MPs are similar to plankton and often provide surfaces for the attachment of microbes. This micro-ecosystem is known as the plastisphere and may harbour bacteria of potentially pathogenic species of Vibrio, which also introduces another dimension of health risk.323. Pathways of microplastic contamination from food processing to the environment
3.1. Direct pathway
Some of the most prominent direct sources of microplastic pollution are the processing and packaging of food. During these stages, microplastics may get released into food products through the direct contact at the primary packaging stage, particularly when the package is subjected to thermal treatment or mechanical damage. Important sources are plastic belts, storage containers, covering films, and machine parts.8 Processes that are highly thermal, e.g., sterilizing or microwaving in plastic containers, are likely to accelerate the movement of microplastic particles and additives, e.g., bisphenol A (BPA) and phthalates, to foods.10Flour mills, spice mills, coffee mills and cereal mills are also sources of airborne microplastics. These sectors employ plastic-lined hoppers, storage bins and pneumatic transfer systems, which continuously vibrate as the materials pass through the equipment. This liberates small particles of polymer that are mixed with the dust produced during milling and grinding.16,40 The fibres of the workers' clothing and the personal protective gear are also synthetic and release fibres into the air when the workers move around the processing plants. Over time, microplastics accumulate in ventilation ducts and are released into the outdoor atmosphere at a minimal rate.6 Processing plants that do not perform well with respect to air filtration or dust removal become point sources of microplastics that add to the atmospheric load and are deposited on soil and in water bodies.
3.2. Indirect pathways
Another major possible source of microplastic contamination is the transportation of food commodities packaged in plastic wrappings or containers. Long-distance delivery of goods causes changes in temperature and mechanical strains that lead to tearing of the wrapping material, ultimately causing microplastic dust to be deposited on the surfaces of edible goods or in the immediate surrounding environment.4 Furthermore, emissions of transport fuel and tire wear from transport vehicles contribute to microplastic buildup on the roadway and soil ecosystems.Deposition is a complex process that takes place in open markets, cold storage units, and distribution centers where foods are packed without sealed protective packaging. Airborne fibers of polyethylene, polypropylene, and polyester are found in the atmospheres of urban areas and are deposited on both packaged and unpackaged foods during unloading, sorting, and marketing. The deposited materials are then removed through waste streams during cleaning or become attached to packaging materials, which degrade into microplastics upon disposal.11,12,14,15 Environmental deposition of microplastics is a complex transport process, and the atmosphere is the major vector through which microplastics are transported from food-processing environments to the natural environment.
4. Environmental impacts of microplastics from food processing
4.1. Aquatic ecosystems
Being the byproduct of food packaging materials and processing, microplastics are a recent pollutant that has been detected in oceans and freshwater systems. Microplastics are produced by abrasion, degradation, and photolysis, and are released into sewage systems and distributed by rivers, ponds, coastal areas, and aquaculture systems, where they are suspended or deposited in the bottom sediments. Due to their small size and ability to persist, the microplastics are absorbed by plankton, fish, and bottom-living organisms, thereby affecting their feeding, nutrient, and physiological processes.19 In addition, microplastics serve as channels for the transportation of pesticides, heavy metals and pathogens, which are subsequently released as contaminants via predator–prey relationships in the aquatic food web.4.2. Terrestrial ecosystems
The terrestrial ecosystem encompasses much lesser area than the aquatic ecosystem, that may serve as sources and sinks of food packaging and degradation of plastics resulting in the leaching out of microplastics. The most common pathways by which microplastics enter terrestrial ecosystems are littering, landfill leachates, land application of sewage sludge and manure, and atmospheric deposition.33 Microplastics can persist in soil for up to several decades. The movement of microplastics into deeper layers of soil is determined by the size of the particles and other physicochemical characteristics. It has been suggested that the presence of microplastics in soil can impact the soil structure, water-holding capacity, and soil fertility, thereby affecting the growth and productivity of plants. Earthworms and nematodes absorb microplastics as soil organisms, which can affect their reproduction, burrowing behaviour, and nutrient cycling in the soil.33,50,51 Moreover, microplastics can also affect microbial communities, reduce the rate of decomposition, and potentially contribute to the spread of pollutants, thereby threatening the health of the soil and its sustainability in agriculture.A second cause of concern centers on the absorption of micro- and nano-plastic particles by crop root hairs, resulting in the introduction of such particles into the food system and human diet.33 The long term exposure to the microplastic contamination may pave way for depletion of soil fertility, affecting sustainable agriculture as well as human health. Efficient management requires strict adherence to regulations controlling the correct disposal of plastics in landfills, new studies into effective technologies for product encapsulation, and enhanced attention to the study of soil-microplastic interactions.
4.3. Ecosystem disruption
Microplastics originating from food preparation and packaging have been found to have substantial ecological significance because of their interaction with various species and assemblages of the ecosystem.11,15,53 The ability of microplastics to migrate, persist, and accumulate in high amounts leads to their extensive distribution in the ecosystem. Microplastics are non-biodegradable, unlike other pollutants, which makes their continuous accumulation and impact on the ecosystem more severe. The impact of microplastics on the ecosystem is caused by the disruption of microbial communities. Microplastics provide habitats for opportunistic and pathogenic microbes, which develop and thrive in these new environments, while simultaneously reducing the amount of beneficial microbes.51,53 This can lead to the disruption of critical ecological processes, such as the decomposition of organic matter, dinitrogen fixation, and carbon sequestration. In aquatic ecosystems, this can significantly affect water quality and the concentration of dissolved oxygen, while in terrestrial ecosystems, it can affect soil quality, agricultural productivity, and vegetation.49,50 A second mechanism is the ingestion of microplastics by a wide variety of species, such as zooplankton, birds, mammals, and invertebrates. The ingestion of microplastic particles causes physiological harm and decreases reproductive rates and population numbers, with the population-level impact being considerably more adverse in keystone populations.45,47–49 Decreased population numbers have a negative impact on trophic cascades and cause top-down effects due to decreased plankton in major water layers or decreased plant pollination in terrestrial ecosystems. In addition, microplastics have direct habitat-related effects. The presence of microplastics in terrestrial and aquatic sediments changes chemical and physical properties, which have negative effects on benthic organisms, while the accumulation of microplastics in coral reef habitats, whether hard or soft, reduces light transmission and negatively affects reef structure.47,48 In terrestrial ecosystems, microplastics change soil properties and decrease water infiltration, with secondary effects on plant survival and organism activity, thereby affecting soil function.50 These large-scale effects represent significant threats to ecosystem services that are critical to human health, including water filtration, nutrient cycling, and climate regulation.33 To counteract these effects, it is necessary to decrease plastic inputs simultaneously with habitat restoration in disturbed regions.5. Health Implications of microplastic release from food processing
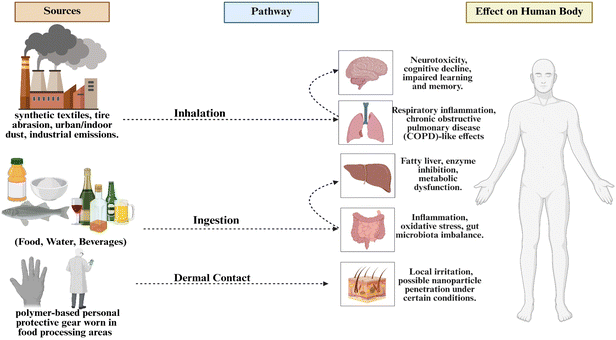
Food contact materials that give rise to microplastics differ depending on the polymer type and the specific use of the polymer in the processing and packaging systems. Polyethylene terephthalate is widely used in bottles, trays, and beverage containers, while polypropylene is used in caps, closures, and hot-fill products. Polyethylene, in both high-density and low-density forms, is extensively utilized in films, liners, and flexible packaging, whereas polyvinyl chloride is employed in tubing, gaskets, and certain conveyor components. These uses are directly related to the category of materials listed in Table 1, which are most closely related to food-related exposure to microplastics. Exposure of humans to microplastics (MPs) is primarily via three major routes: ingestion, inhalation, and dermal exposure.54 Ingestion is considered the dominant pathway and involves the consumption of MPs via foods and beverages. Microplastics (MPs) have been found to abound within a very large spectrum of edible products, including seafood, mussels, fruits and vegetables, meat, cereals and legumes, sea salts, sugars, and drinks.55 The major polymers that represent the MPs within food and beverages include polypropylene (PP), polyethylene terepthalate (PET), PVC, polyethlyene including LDPE and HDPE respectively.55 Additionally, another pathway through which MPs can be breathed in is significantly narrowed down to indoor and urban areas. Microplastics can enter the air through various emission processes, including textile and rubber tyre breakdown, building exterior emissions, and resuspension of aerosols after their initial deposition.56 Airborne microplastics primarily comprise man-made fibers and particulate matter emitted from PP, acrylics, polyester, polyamide (PA), and PE.25,57 Although attracting less attention, dermal exposure is another significant pathway; this can arise through contact with MPs contained within personal care and cosmetic products, and within households through a portion of the ambient dust, and upon contact with manmade textile fibers. Cosmetics that contain fibers and microbeads, along with particles that shed from clothes, represent a significant pathway of exposure to PE, PP, PET, and nylon through the dermis.54In summary, there are three major routes through which MPs can enter the human body: ingestion, inhalation, and dermal contact. As discussed above, ingestion of MPs can occur from foods and drinks contaminated by MPs. Inhalation of MPs can happen through airborne particles originating from synthetic textiles, urban dust, and rubber tyres.58 Dermal exposure to MPs that can be present in cosmetic products, textiles, or dust may occur through wounds, sweat glands, or hair follicles.59,60 These routes will be further explained in the following sections.
5.1. Ingestion
Major human exposure to microplastics is through ingestion.57 This exposure pathway is confirmed by the fact that microplastics are found in human feces; the most common polymeric substances are polyethylene terephthalate, polyamide, and polypropylene.61,62 Collectively, these studies indicate the probability that microplastics penetrate the human body through polluted food and beverages. Besides the gastrointestinal tract, microplastics have also been detected in the human placenta,63 human breast milk,64 human blood,65 infant formula,66 and some oral products.67 The prevalence of microplastics in different biological samples indicates that there might be systemic exposure following the translocation of microplastics through the gastrointestinal barrier. However, the ultimate fate of microplastics within the human gastrointestinal system remains largely unresolved in existing research, highlighting the need for further studies on how these particles are processed in the GI tract. Exposure occurs through the ingestion of food and beverages containing microplastics, as well as through mucociliary uptake following inhalation.68 As indicated, microplastics have been detected in some food and drink items, including seafood, table salt, sugar, honey, bottled water, and beer. Table salt is typically contaminated during seawater evaporation, crystallization, and milling, with polyethylene, polypropylene, and polystyrene fragments and fibers being the most prevalent. In bivalves, microplastics are derived from uptake and depuration tank water, with polyethylene, polypropylene, and nylon fibers being the most abundant. Contamination of sugar may occur during refining, crystallization, milling, and packaging in polypropylene- or polyethylene-lined bags, and bottled water contamination may mostly be associated with scratching of the PET and polypropylene bottle surfaces and caps. Polyethylene and polypropylene pieces can also be present in cereals and flour, with silo linings, conveyor belts, and milling equipment being the source of contamination. Cox et al. (2019)69 estimated that an average American diet might expose an individual to approximately 39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to 52
000 to 52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 microplastic particles annually. Eighteen thousand particles were estimated to be consumed by Europeans every year through the consumption of bivalves alone.28 Table salt research points out that there are different amounts of microplastic consumption across geographical areas, including Europe and China.7,55 Such estimates highlight the nutritional relevance of dietary intake and demonstrate that even simple food groups and condiments can significantly influence overall microplastic exposure.
000 microplastic particles annually. Eighteen thousand particles were estimated to be consumed by Europeans every year through the consumption of bivalves alone.28 Table salt research points out that there are different amounts of microplastic consumption across geographical areas, including Europe and China.7,55 Such estimates highlight the nutritional relevance of dietary intake and demonstrate that even simple food groups and condiments can significantly influence overall microplastic exposure.
The behaviour of microplastics in the gastrointestinal tract after ingestion is dependent on the size, surface chemistry, and shape, as well as their ability to interact with mucus and epithelial tissues. The paracellular route of transit of microplastics may seem to be unlikely due to the tight junctions between intestinal epithelial cells which have pore sizes of approximately 1.5 nm and cannot allow normal microplastic particles to pass through them.70 Rather, the absorption of microplastics tends to occur via specialised immune apparatus in the gut. The microplastics may enter the digestive tract via a cellular process called endocytosis by the microfold cells of Peyer's patche.71 Experimental murine model studies also strengthen the idea of the involvement of immune cells in microplastic processing. Peritoneal macrophages have been shown to phagocytose polymethyl methacrylate and polystyrene particles of 1 µm, 5 µm, and 12 µm, with uptake varying according to particle size. This demonstrates that phagocytic cells recognize and internalize particles in a size-dependent manner.72 In the physiological environment, dissolvable particles may circumvent the intestinal mucus layer. Their mobility in mucus is increased as they develop a protein corona; this happens when proteins on the intestinal surface stick to the particle surface and change their hydrophobicity, charge, and biological interactions.73 This study demonstrates that oral exposure may influence the potential of particles to be taken up by epithelial or immune cells, and may influence the retention or clearance of particles in the gastrointestinal tract. There are also other interaction modes of microplastics with the gastrointestinal components (digestive enzymes, bile salts, lipids, and food components) within the gastrointestinal environment. Such interactions impact on the characteristics of microplastics, aggregation, or bioavailability. Excretion of larger particles, which are not absorbed, is normally through feces, as shown in the analysis of human fecal samples.61 However, smaller particles in the micro- and submicron-size ranges are more likely to interact with immune tissues or translocate into the systemic circulation, as shown in the recent studies on microplastics in human blood, placenta, and breast milk.63–65 All these observations suggest that ingestion is the most common and important route of human exposure to microplastics. The types of polymers found in the gastrointestinal tract are similar to the polymers found in food packaging materials, food processing equipment, cooking utensils, and storage containers, thus highlighting the role of the food chain in microplastic uptake. Further studies are required to understand the implications of microplastic ingestion in humans.
5.2. Inhalation
High-friction food processing activities, including spice and grain grinding, dough and sauce processing, and packaging processes, including film cutting, sealing, and high-speed form-fill-seal processing (also explained in Section 2.1.3 and 2.1.5), are sources of high inhalation exposures of airborne microplastics (MPs) in food processing plants (Fig. 2). Moreover, micro-exposure to synthetic fibers from personal protective equipment (PPE) and textiles takes place during handling.57 The respiratory tract provides a substantial alveolar surface of roughly 150 m2, having a thin epithelial barrier (less than 1 mm), enabling deep penetration of small MPs (less than 5 mm in diameter). These particles may result in macrophage phagocytosis and inflammation through the ROS (Reactive oxygen Species) and may result in systemic translocation. The daily exposure of the microplastics varied significantly during different industrial activities.74,755.3. Dermal exposure
Direct interaction with wet, semi-liquid, or contaminated food-processing materials that contain microplastics (MPs) is the major cause of dermal exposure in food-processing settings. Another source of exposure is through abrasion and shedding of polymer-based personal protective gear, including disposable gloves, aprons, sleeve covers, hairnets, and protective clothes worn in food-processing areas (Fig. 2). The another route through microplastics may get embedded into skin includes equipment cleaning. Cutting, sealing and high-speed packaging processes generate fine plastic dust that gets on the hands and forearms of the operatives. Wet processes such as rinsing seafood, washing meat, and processing vegetables promote the adhesion of MP to moist surfaces of the body through micro-abrasions, hair follicles or sweat ducts. The microplastics incorporation through dermal route is not so substantial than through ingestion and inhalation routes, there is a reason to believe that nanoscale particles (less than 100 nm) may penetrate the Stratum corneum barrier under the influence of moisture, repeated contact and chemical cleaners, which would either result in localized inflammation or serve as an additive carrier.59,60,765.4. Biomonitoring of microplastics in humans
Biomonitoring is used to determine exposure to environmental contaminants by measuring the chemical constituents themselves, their transformation products or their metabolites within biological media such as blood, stool, urine or tissue materials.77 The biomonitoring encompasses the various routes of exposure (foods, cosmetics and personal care products, water and air) to the various routes of exposure (dermal adsorption, ingestion and inhalation).78 Biomonitoring provides the direct evidence about the presence of specific chemicals within the human body.79 One of the key limitations regarding the information about the quantum of uptake of MPs by human population is the availability of limited data to validate the exposure of human population to the microplastics and study their effect on the human cellular mass.80 One of the significant shortcomings within the measurement of human uptake of MPs is sparse data to answer exposure of inhabitants, including workers, dispersion, and dynamics of MPs into the tissue of the organic mass of the living being, and the potential cocktail effect MPs can cause with other co-present contaminants.81 Regardless of human beings incessantly faced by humongous amounts of contaminants, including MPs, various biological barricades are installed to protect human beings against the entry of foreign and potentially harmful entities.82Clearance mechanisms such as sneezing, coughing, mucociliary escalator, macrophage-mediated phagocytosis, as well as clearance through the lymphatic transport system, are induced to prevent particles from penetrating biological barriers, reaching target cells or organs, or leading to bioaccumulation.83,84 To date, no study has reported categorical findings for the biokinetics of human microplastics. As such, their uptake, distribution, retention, and excretion are not accurately known, unlike the highly studied nanoparticles. Polymer nanoparticles have been extensively studied for use in both diagnostic applications and drug delivery applications because of their high stability.85,86 Polymeric nanoparticles not only cross biological barriers but are also transported by certain organs into the bloodstream for systemic distribution. Consequently, nanoparticles access secondary organs as well as the central nervous and immune systems.87 The diameter of nanoparticles significantly determines their deposition within the extrathoracic and intrathoracic parts of the respiratory tract.82 Apart from particle size, additional factors such as material characteristics, shape, surface functionalization, and structural characteristics together affect nanoparticle biodistribution.81 Similarly, the size of the microplastics dictates the uptake by the digestive tract,88 the respiratory system,89 as well as the integumentary system itself.90 Larger particulates tend to be eliminated from the organism by faecal excretion,29 but the smaller particulates could go into systemic circulation and cause physiologic effects such as endocrine disruption or elevated obesity.81 Microplastics have been found in numerous biological matrices, such as blood, urine, feces, placenta, and breast milk.
5.5. Toxicological effects of microplastics
Microplastics (MPs) induce toxicological responses by multiple mechanisms that are governed by their physical and chemical properties, such as size, morphology, surface charge, additives, and environmental interactions.91,92 They not only exhibit inherent toxicity but also act as vectors for other contaminants, such as heavy metals and hydrophobic organic compounds (HOCs), and thus allow their entry into biological compartments.93,94 The toxicity of MPs is typically evaluated across multiple biological levels, ranging from whole organisms to subcellular structures, using diverse experimental models comprising both in vitro and in vivo testing.91 Multiple physicochemical parameters define the toxicity profile of MPs. Small particles exhibit an enhanced capability for cell internalization and induce robust oxidative stress responses.95 Irregularly structured particles induce larger volumes of mechanical injuries relative to their sphere-structured counterparts.96 Cellular uptake depends greatly on the surface-charge zeta potential of the particle, leading to the efficient internalization of positive-charge MPs.97 Weathering processes as well as environmental aging modify the shape of the surface, the crystalline nature, and the chemical reactivity, thereby enhancing their biological reactivity potential.93 Moreover, microplastics often contain additives that can leach under the physicochemical conditions of the human body, potentially contributing to their increased toxicity.91,93 High surface areas of MPs enable the adsorption of co-contaminants such as HOCs (Hydrophobic Organic Compounds) as well as heavy metals, potentially as a consequence of an increase in their cumulative toxicity.19,94 When broken down into nanoplastics, these materials can create a biomolecular corona that can modify their bioavailability, persistence, and toxicity, which is then modulated by environmental aging.92 At the cellular scale, MPs have been found to cause cytotoxic effects through oxidative stress, membrane disruption, and DNA damage. Such processes heavily rely on particle size, concentration, charge, exposure duration, and additive types.88,98 Human cell lines, including gastrointestinal, airway, and immune cells, respond to MPs with varying levels of sensitivity, with some demonstrating immune dysregulation depending on the surface modifications of the MPs.88,99 MPs have been found to suppress growth and impair the antioxidant system of microalgae.100,101 Advanced developments using organoid technology provide better physiologically relevant systems to determine MP toxicity. The self organized 3D tissue cultured stem cells derived from forebrain, intestinal and liver have shown substantial organ impairement when exposed to MPs.102,103 MPs have been found to infiltrate through cellular layers, cause apoptosis, trigger inflammatory processes, and impair lipid metabolism through these models. Importantly, co-exposure of MPs with other chemicals, including bisphenol A (BPA), results in combined hepatotoxic effects, affirming the role of MPs as chemical transport agents.102 Animal models also confirm the potential toxicity of MPs, with many of their organ systems impaired. MPs contribute to metabolic impairment, including disrupted lipid digestion, liver injury with oxidative damage, enzyme inhibition, and gut microbiota dysbiosis in both fish and rodent models.104,105 Immune-mediated toxicity consists of inflammatory cytokine expression augmentation (e.g., IL-1β and TNF-α) and disruption of mucosal immunity after exposure to MPs.106,107 Neurotoxicity is attributed to as MPs block acetylcholinesterase (AchE) activity, disruption in blood–brain barrier integrity, impaired learning and memory in animal models.108,109 MPs have adverse effects on reproductive health in terms of lowered oocyte and sperm quality, disruption of the sexual ratio, and impaired embryo growth.86,110 It was proposed that these adverse effects could be monitored using immunoglobulin A (IgA) as a biomarker.110 Epidemiologic findings of human contact with MPs are infrequent but disturbing. Professionals working in the plastic industry have been susceptible to exposure to MPs, resulting in the chronic respiratory ailments.111,112 Patients with inflammatory bowel disease exhibit increased fecal concentration of MPs as compared to the healthy controls, and this was correlated with disease progression.62 Cirrhotic liver tissue and arterial thrombi in human liver were found to contain MPs, which can be indicative of a relationship with liver and cardiovascular pathology.113,114 It can be concluded from the studies reported, that there is growing concern about the public health problems attributed to exposure to MPs as well as about the future research in this area, whereas authentic cause can be validated only after the substantial reasearch is conducted in this field.6. Detection and monitoring of microplastics in food processing and the environment
Different analytical methods are used to analyze microplastics in food samples and are optimized to achieve this goal (Table 2). The main techniques used are visual analysis, microscopy, and spectroscopic analysis.16 In visual analysis, the first step, which is very important for larger microplastics (larger than 1 mm), is done manually based on criteria such as particle size, shape, and colour. Microscopic analysis, which includes optical microscopy, stereomicroscopy, and scanning electron microscopy (SEM), allows microplastics to be visualized and provides information on morphology and surface characteristics.16 Besides morphological analysis, chemical and molecular analyses are needed. Fourier transform infrared (FTIR) spectroscopy and Raman spectroscopy are mainly used for this purpose, making it possible to specifically analyze polymeric materials rather than non-polymeric materials in food samples. By combining visual, microscopic, and spectroscopic analyses, a more detailed analysis of microplastics can be made, including both morphological and chemical characteristics.115 Pyrolysis gas chromatography-mass spectrometry (Py-GC/MS) is one of the most widely used analytical techniques for polymeric materials. This technique involves the thermal decomposition of microplastics into smaller pieces, which are then separated and analyzed by GC-MS. Py-GC/MS provides information on the chemical composition of microplastics, making it possible to identify and measure microplastics in food samples.16 These methods have been widely used in various studies to assess microplastic contamination in food samples, with a focus on packaging-based sources. For example, Xu et al. (2019)116 employed FTIR microscopy to analyze microplastic contamination in bottled water, finding physical abrasion and leaching to be the main contamination sources. Identical research employed Raman spectroscopy and pyrolysis-gas chromatography-mass spectrometry (Py-GC-MS) to determine microplastics in seafood, and the study revealed that the level of contamination is higher as the packaging materials degrade. In another study, thermal extraction and desorption GC-MS were used by Singh and Kumar (2024)117 as a method of analyzing microplastics in different foodstuffs; this study also clarified the significance of thermal degradation during food processing and storage. The multi-analytical method of visual analysis, FTIR spectroscopy and scanning electron microscopy (SEM) analysis of microplastics in canned foods is gaining support due to the evidence of contamination associated with the mechanical force of the canning process.116,118 According to Sewwandi et al. (2023),14 based on FTIR and Raman spectroscopy studies, the migration of microplastics from bottle caps and closures into beverages is significant, underscoring the need for improved packaging designs. Despite the significant advances in the study of microplastics in the food chain, certain issues remain regarding inconsistencies in the concentration, type, and size of the microplastics.13 Future research must aim to enhance the sensitivity and rate of analysis and establish cost-efficient solutions that can be easily practiced in the food sector. The analytical methods to be adopted in applied studies may be determined by the food matrix being studied, but particular attention should focus on fatty and protein-rich foods. Oils, meat, dairy products, nuts, seeds, emulsified foods, and other foods that consist of complex samples might demand complex sample preparation procedures to minimize interference by the lipid and protein food matrix components. This interference is most severe in oils, meat, dairy products, nuts, seeds, and emulsified foods, as these foods require complex preprocessing steps to counter lipid and protein interference. Digestion by protease or lipase enzymes can allow more effective removal of proteins and lipids while maintaining the integrity of polymers that may otherwise be broken down by severe chemical oxidants. Digestion with hydrogen peroxide is more suitable for carbohydrate-containing samples, which have to be carefully processed to prevent the destruction of polymers. For lipid-rich samples, solvent extraction with ethanol or hexane can simplify the sample matrix prior to filtration, making the preparation process easier. During microscopic and spectroscopic analysis of particulate substances, especially spices, tea, coffee, flour, and cereals, density separation is required to eliminate interference of organic matter or mineral particulates. It is possible to isolate buoyant microplastics using buoyancy solutions that include sodium chloride, zinc chloride, or sodium iodide to allow the separation of particles. By applying these sample preparation methods, complex matrices may not provide desirable results in optical clarity and spectroscopy analysis, thus pyrolysis GC/MS and thermal extraction methods could be used as alternative methods in samples, where particle counting and visualization are not viable. All these factors underscore the paramount role of sample preparation for the successful analysis of microplastics in food. It is also crucial to select analytical tools that are compatible with the food matrix under consideration and its characteristics.| Analytical method | Information obtained | Strengths | Limitations/challenges | Best suited food matrices |
|---|---|---|---|---|
| Visual inspection/light microscopy | Size, color, shape | Fast; inexpensive; preliminary sorting | Not reliable for confirmation; cannot identify polymers | Large visible plastics; low-fat solids; coarse foods |
| Stereo zoom microscopy/SEM | Detailed morphology, surface characteristics | High resolution; suitable for morphology | Requires clean, debris-free samples; SEM costly | Powders (spices, tea), cereals, seafood tissues |
| FTIR spectroscopy (ATR-FTIR, µFTIR) | Polymer identification | Widely used; reliable chemical fingerprinting | Particles must be > 20 µm; organic residues interfere | Bottled water, salt, sugar, dried foods |
| Raman spectroscopy | High-resolution polymer identification | Detects submicron particles; strong chemical specificity | Fluorescence interference; sensitive to pigments | Seafood, beverages, ready-to-eat meals, spices |
| Pyrolysis–GC–MS | Polymer composition (mass-based) | Works well for complex matrices; no visual sorting needed | No particle counts; destructive method | High-fat foods, oils, meat emulsions, dairy |
| Thermal extraction and desorption GC-MS | Polymer mass, additives | Suitable for contaminated samples; high sensitivity | Cannot size/count particles | Packaged foods, processed foods, bakery items |
| Enzymatic digestion (pretreatment) | Removes proteins/fats | Gentle; preserves particle integrity | Time-consuming; expensive | Meat, seafood, dairy, infant formula |
| Oxidative digestion (H2O2) | Removes organic matter | Useful for plant-based foods | May damage sensitive polymers | Fruits, vegetables, cereals, spices |
7. Emerging sustainable strategies for reducing plastic use in the food industry
The concept of sustainability has gained significant importance in contemporary food technology, particularly in connection with microplastic contamination, environmental destruction, and inefficient resource usage. The present research emphasis is to reduce the utilization of conventional fossil-derived plastics and promote the utilization of the more environmentally friendly materials, processes, and packaging technologies in the food industry. The synergistic push of scientific advancement and government policy, and the growing demand for environmentally safe and sustainable food packaging technologies, drives the agenda of innovation. The innovation agenda focuses on the development and evaluation of bio-based and biodegradable polymers for food packaging applications.119–122 Bio-based and biodegradable polymers such as polylactic acid, polyhydroxyalkanoates, starch-based films, cellulose-based composites, and chitosan-based coatings have become popular due to their biodegradability and bio-based nature. There is empirical evidence to show that these materials can be designed to possess mechanical, barrier, and thermal properties similar to those of some fossil fuel-based plastics.120–125 Despite these improvements, there remain issues with regard to the rate of processing, water resistance, and the ability to withstand high temperatures in the food processing industry. Current research aims to enhance functional properties through compounding, nanocomposite reinforcement, and surface modification. In addition to these developments, current research and development in sustainable food processing technologies aim to decrease plastic usage in food processing plants.119,120,122 This involves redesigning components traditionally manufactured from polymeric materials by replacing them with reusable high-grade stainless-steel surfaces and enhancing wear-resistant coatings, thereby reducing particle flaking. Food processing companies are also pursuing developments in cleaning-in-place technology and closed-loop water management systems to reduce the discharge of plastic particles into wastewater. Contemporaneous research and development in modular packaging systems, refillable packaging designs, and bulk-dispensing packaging configurations aim to decrease single-use plastics in foodservice and retail packaging.126 Recycling and the circular economy are major goals of sustainability projects. Technological developments in recycling, such as chemical recycling and depolymerization, are expected to make it possible to recycle mixed or contaminated plastics back into reusable monomers or feedstocks. These results are not possible with traditional mechanical recycling, which is limited in its ability to recycle multilayer packaging materials, colored polymers, and food-contaminated packaging. The Easily Recyclable Mono-Material Packaging and Smart Labeling project is a strategic measure to enhance food packaging material sustainability (advancing the sorting efficiency). Estimation of microplastic emissions of alternative materials is one of the main research fields. Although biodegradable and compostable plastics are suggested as environmentally friendly alternatives to plastics, there are emerging indications that these plastics may degrade into nanoparticles under some degradation conditions.120–122,126 More studies are needed to better predict the environmental destiny of these plastics, with emphasis on the processes of degradation and fragmentation and how this affects soil and water. This is an area that needs immediate research focus to ensure the transition to sustainable packaging does not lead to new sources of particulate pollution. Sustainability laws in the food industry have been on the rise, including the outlawing of single-use plastics, recycling, compostability, and biodegradability guidelines, which are already in place in most nations. All these sustainability requirements demand that the food industry and the research fraternity collaborate to develop safe, functional, and sustainable materials.Generally, strategies employed to enhance the sustainability of plastics in food processing and packaging adopt a holistic strategy entailing the search for alternatives and innovations, a circular economy strategy, and creating an enabling environment. More research is required to refine these methods and ensure that the developed sustainable food systems contribute to a reduction in microplastic pollution.
8. Conclusion
Microplastic pollution in food webs is a critical issue for food safety, health, and the sustainability of ecosystems in the long term. The present review shows that microplastics can contaminate food throughout the food chain, which includes the primary processing stage, processing equipment abrasion, packaging material degradation, storage, transportation and handling. Primary and secondary sources are analyzed, and it is revealed that the extent of microplastic transfer depends on the nature of the polymer, the nature of the food-contact material, and the amount of mechanical, thermal, and chemical forces applied during food processing. Ingestion is found to be the main route to exposure, and seafood, bottled water, salt, sugar, breakfast cereals, and ready-to-eat foods are found to be important sources. Although inhalation and dermal exposure are significant routes to exposure, their relative contribution is lower but still important. Biomonitoring studies have confirmed the presence of microplastics in the human biological system, and current research on gastrointestinal interactions, immune cell uptake, and cellular responses suggests a need for further toxicological studies. Current analytical difficulties remain, especially in complex food samples in which lipids, proteins, and particulates cause interference in microplastic separation and analysis. The use of modern pretreatment methods together with sophisticated spectroscopic and thermogravimetric/thermal analytical instruments is necessary to ensure the accuracy and comparability of the results for a wide range of food samples. The growing emphasis on sustainability is becoming increasingly evident in strategies developed to mitigate microplastic pollution across the agri-food chain. Bio-based and biodegradable polymers, single-material recyclable packaging systems, process technologies, and circular economy solutions are promising approaches to reduce plastic use and, consequently, limit microplastic transfer to food. However, it is crucial to assess the environmental fate of new materials to prevent pollution in other industries. In conclusion, a holistic approach to interventions that are developed to counteract microplastic exposure and food quality issues is necessary. With the development of the food industry, technologies that can reduce microplastics in food processing and packaging systems are expected to play a crucial role in ensuring food safety.Author contributions
Md Sabir Ahmad Mondol; wrote the whole manuscript, Mehvish Ayoub; revised the manuscript, Ubaida Akbar; formal analysis, Kshirod Kumar Dash; project administration, Aamir Hussain Dar; conceptualization, Urba Shafiq Sidiqi; software.Conflicts of interest
The authors declare no competing interests.Data availability
No new data were generated or analysed as part of this review.Acknowledgements
No funding was received for this study.References
- R. Geyer. Production, use, and fate of synthetic polymers. Plastic Waste and Recycling, Elsevier; 2020, pp. 13–32, DOI:10.1016/B978-0-12-817880-5.00002-5.
- A. Max, V. Martín-Vélez, J. Navarro, A. Borrell, T. Montalvo and O. Garcia-Garin, Characterization of plastic ingestion in urban gull chicks and its implications for their use as pollution sentinels in coastal cities, Mar. Pollut. Bull., 2025, 211, 117409, DOI:10.1016/j.marpolbul.2024.117409.
- M. Dokl, A. Copot, D. Krajnc, Y. V. Fan, A. Vujanović and K. B. Aviso, et al., Global projections of plastic use, end-of-life fate and potential changes in consumption, reduction, recycling and replacement with bioplastics to 2050, Sustain. Prod. Consum., 2024, 51, 498–518, DOI:10.1016/j.spc.2024.09.025.
- C. Campanale, C. Massarelli, I. Savino, V. Locaputo and V. F. Uricchio, A Detailed Review Study on Potential Effects of Microplastics and Additives of Concern on Human Health, Int. J. Environ. Res. Public Health, 2020, 17, 1212, DOI:10.3390/ijerph17041212.
- A. Brown, Planetary health digest, Lancet Planet Health, 2019, 3, e378, DOI:10.1007/s10584-019.
- M. Lee, H. Kim, H.-S. Ryu, J. Moon, N. A. Khant and C. Yu, et al., Review on invasion of microplastic in our ecosystem and implications, Sci. Prog., 2022, 105 DOI:10.1177/00368504221140766.
- D. Yang, H. Shi, L. Li, J. Li, K. Jabeen and P. Kolandhasamy, Microplastic Pollution in Table Salts from China, Environ. Sci. Technol., 2015, 49, 13622–13627, DOI:10.1021/acs.est.5b03163.
- D. Barceló, Y. Picó and A. H. Alfarhan, Microplastics: Detection in human samples, cell line studies, and health impacts, Environ. Toxicol. Pharmacol., 2023, 101, 104204, DOI:10.1016/j.etap.2023.104204.
- S. Aslam, A. Khurram, R. Hussain, A. Qadir and S. R. Ahmad, Sources, distribution, and incipient threats of polymeric microplastic released from food storage plastic materials, Environ. Monit. Assess., 2023, 195, 638, DOI:10.1007/s10661-023-11242-5.
- F. Haque and C. Fan, Fate and Impacts of Microplastics in the Environment: Hydrosphere, Pedosphere, and Atmosphere, Environments, 2023, 10, 70, DOI:10.3390/environments10050070.
- C. G. Eze, C. E. Nwankwo, S. Dey, S. Sundaramurthy and E. S. Okeke, Food chain microplastics contamination and impact on human health: a review, Environ. Chem. Lett., 2024, 22, 1889–1927, DOI:10.1007/s10311-024-01734-2.
- M. Saeedi, How microplastics interact with food chain: a short overview of fate and impacts, J. Food Sci. Technol., 2024, 61, 403–413, DOI:10.1007/s13197-023-05720-4.
- J.-H. Kwon, J.-W. Kim, T. D. Pham, A. Tarafdar, S. Hong and S.-H. Chun, et al., Microplastics in Food: A Review on Analytical Methods and Challenges, Int. J. Environ. Res. Public Health, 2020, 17, 6710, DOI:10.3390/ijerph17186710.
- M. Sewwandi, H. Wijesekara, A. U. Rajapaksha, S. Soysa and M. Vithanage, Microplastics and plastics-associated contaminants in food and beverages; Global trends, concentrations, and human exposure, Environ. Pollut., 2023, 317, 120747, DOI:10.1016/j.envpol.2022.120747.
- P. Sharma, Microplastic Contamination in Food Processing: Role of Packaging Materials, Food Sci. Eng., 2024, 271–287, DOI:10.37256/fse.5220244519.
- P. Sharma, P. Sharma and K. Abhishek, Sampling, separation, and characterization methodology for quantification of microplastic from the environment, J. Hazard. Mater. Adv., 2024, 14, 100416, DOI:10.1016/j.hazadv.2024.100416.
- P. Makhdoumi, A. A. Amin, H. Karimi, M. Pirsaheb, H. Kim and H. Hossini, Occurrence of microplastic particles in the most popular Iranian bottled mineral water brands and an assessment of human exposure, J. Water Process Eng., 2021, 39, 101708, DOI:10.1016/j.jwpe.2020.101708.
- J. A. Conesa and N. Ortuño, Reuse of Water Contaminated by Microplastics, the Effectiveness of Filtration Processes: A Review, Energies (Basel), 2022, 15, 2432, DOI:10.3390/en15072432.
- Y. Xiang, L. Jiang, Y. Zhou, Z. Luo, D. Zhi and J. Yang, et al., Microplastics and environmental pollutants: Key interaction and toxicology in aquatic and soil environments, J. Hazard. Mater., 2022, 422, 126843, DOI:10.1016/j.jhazmat.2021.126843.
- Ł. Zbucki and E. Plażuk, MECHANICAL GRINDING OF SPICES IN GRINDERS WITH POLYMERIC BURRS AND TRANSFER OF MICROPLASTICS TO FOOD, Health Probl. Civiliz., 2024, 18, 339–353, DOI:10.5114/hpc.2024.135399.
- L. Fian, L. M. Schmidlechner, U. Felt, T. Hofmann, M. P. White and S. Pahl, Microplastics in food and drink: perceptions of the risks, challenges, and solutions among individuals in the ‘farm-to-fork’ food chain, J. Risk Res., 2024, 27, 986–1009, DOI:10.1080/13669877.2024.2431900.
- A. Dąbrowska, M. Mielańczuk and M. Syczewski, The Raman spectroscopy and SEM/EDS investigation of the primary sources of microplastics from cosmetics available in Poland, Chemosphere, 2022, 308, 136407, DOI:10.1016/j.chemosphere.2022.136407.
- C.-L. Bai, L.-Y. Liu, J.-L. Guo, L.-X. Zeng and Y. Guo, Microplastics in take-out food: Are we over taking it?, Environ. Res., 2022, 215, 114390, DOI:10.1016/j.envres.2022.114390.
- X. Wu, P. Liu, H. Shi, H. Wang, H. Huang and Y. Shi, et al., Photo aging and fragmentation of polypropylene food packaging materials in artificial seawater, Water Res., 2021, 188, 116456, DOI:10.1016/j.watres.2020.116456.
- J. Gasperi, S. L. Wright, R. Dris, F. Collard, C. Mandin and M. Guerrouache, et al., Microplastics in air: Are we breathing it in?, Curr. Opin. Environ. Sci. Health, 2018, 1, 1–5, DOI:10.1016/j.coesh.2017.10.002.
- R. Geyer, J. R. Jambeck and K. L. Law, Production, use, and fate of all plastics ever made, Sci. Adv., 2017, 3 DOI:10.1126/sciadv.1700782.
- S. K. Nabil, N. Mustari and M. S. Khan. Sources and Health Impacts of Chemical Contaminants in Foods. Techniques to Measure Food Safety and Quality, Springer International Publishing, Cham, 2021, pp. 31–68, DOI:10.1007/978-3-030-68636-9_3.
- L. Van Cauwenberghe and C. R. Janssen, Microplastics in bivalves cultured for human consumption, Environ. Pollut., 2014, 193, 65–70, DOI:10.1016/j.envpol.2014.06.010.
- M. Smith, D. C. Love, C. M. Rochman and R. A. Neff, Microplastics in Seafood and the Implications for Human Health, Curr. Environ. Health Rep., 2018, 5, 375–386, DOI:10.1007/s40572-018-0206-z.
- K. Toner and S. R. Midway, Historic fish samples from the Southeast USA lack microplastics, Sci. Total Environ., 2021, 776, 145923, DOI:10.1016/j.scitotenv.2021.145923.
- G. Liebezeit and E. Liebezeit, Synthetic particles as contaminants in German beers, Food Addit. Contam.,:Part A, 2014, 31, 1574–1578, DOI:10.1080/19440049.2014.945099.
- E. R. Zettler, T. J. Mincer and L. A. Amaral-Zettler, Life in the “Plastisphere”: Microbial Communities on Plastic Marine Debris, Environ. Sci. Technol., 2013, 47, 7137–7146, DOI:10.1021/es401288x.
- F. A. Bello, A. B. Folorunsho, R. W. Chia, J.-Y. Lee and S. A. Fasusi, Microplastics in agricultural soils: sources, impacts on soil organisms, plants, and humans, Environ. Monit. Assess., 2025, 197, 448, DOI:10.1007/s10661-025-13874-1.
- R. Jakhar, S. K. Sandwal, I. Ali and K. Styszko, Plastic Waste to Microplastic Pollution and Its Impacts: A Comprehensive Review on Delhi, India, Appl. Sci., 2025, 16, 61, DOI:10.3390/app16010061.
- H. Hettiarachchi and J. N. Meegoda, Microplastic Pollution Prevention: The Need for Robust Policy Interventions to Close the Loopholes in Current Waste Management Practices, Int. J. Environ. Res. Public Health, 2023, 20, 6434, DOI:10.3390/ijerph20146434.
- N. Nabi, I. Ahmad, A. Amin, M. A. Rather, I. Ahmed and Y. A. Hajam, et al., Understanding the sources, fate and effects of microplastics in aquatic environments with a focus on risk profiling in aquaculture systems, Rev Aquac, 2024, 16, 1947–1980, DOI:10.1111/raq.12941.
- O. H. Fred-Ahmadu, F. O. Ahmadu, A. E. Adedapo, I. Oghenovo, O. T. Ogunmodede and N. U. Benson, Microplastics and chemical contamination in aquaculture ecosystems: The role of climate change and implications for food safety—a review, Environ. Sci. Eur., 2024, 36, 181, DOI:10.1186/s12302-024-00995-6.
- H. Yadav, M. R. H. Khan, M. Quadir, K. A. Rusch, P. P. Mondal and M. Orr, et al., Cutting Boards: An Overlooked Source of Microplastics in Human Food?, Environ. Sci. Technol., 2023, 57, 8225–8235, DOI:10.1021/acs.est.3c00924.
- J. B. Kasper, L. A. Parker, S. Postema, E. M. Höppener, A. H. Leighton and A. M. D. Finnegan, et al., Losses and emissions in polypropylene recycling from household packaging waste, Waste Manage., 2025, 191, 230–241, DOI:10.1016/j.wasman.2024.11.029.
- S. Swinnerton, J. Su and C. S. J. Tsai, The emission and physicochemical properties of airborne microplastics and nanoplastics generated during the mechanical recycling of plastic via shredding, Sci. Rep., 2024, 14, 24755, DOI:10.1038/s41598-024-73775-0.
- C. Dessì, E. D. Okoffo, J. W. O'Brien, M. Gallen, S. Samanipour and S. Kaserzon, et al., Plastics contamination of store-bought rice, J. Hazard. Mater., 2021, 416, 125778, DOI:10.1016/j.jhazmat.2021.125778.
- M. Kosuth, S. A. Mason and E. V. Wattenberg, Anthropogenic contamination of tap water, beer, and sea salt, PLoS One, 2018, 13, e0194970, DOI:10.1371/journal.pone.0194970.
- R.-S. Yu and S. Singh, Microplastic Pollution: Threats and Impacts on Global Marine Ecosystems, Sustainability, 2023, 15, 13252, DOI:10.3390/su151713252.
- R. Mercogliano, C. G. Avio, F. Regoli, A. Anastasio, G. Colavita and S. Santonicola, Occurrence of Microplastics in Commercial Seafood under the Perspective of the Human Food Chain. A Review, J. Agric. Food Chem., 2020, 68, 5296–5301, DOI:10.1021/acs.jafc.0c01209.
- L. Boone, N. Préat, T. T. Nhu, F. Fiordelisi, V. Guillard and M. Blanckaert, et al., Environmental performance of plastic food packaging: Life cycle assessment extended with costs on marine ecosystem services, Sci. Total Environ., 2023, 894, 164781, DOI:10.1016/j.scitotenv.2023.164781.
- J. Lehel and S. Murphy. Microplastics in the Food Chain: Food Safety and Environmental Aspects, 2021, pp. 1–49, DOI:10.1007/398_2021_77.
- I. M. Jaikumar, M. Tomson, A. Meyyazhagan, B. Balamuralikrishnan, R. Baskaran and M. Pappuswamy, et al., A comprehensive review of microplastic pollution in freshwater and marine environments, Green Anal. Chem., 2025, 12, 100202, DOI:10.1016/j.greeac.2024.100202.
- A. K. Mishra, J. Singh and P. P. Mishra, Microplastics in freshwater ecosystem: A serious threat for freshwater environment, Int. J. Environ. Sci. Technol., 2023, 20, 9189–9204, DOI:10.1007/s13762-022-04489-x.
- H. T. Nair and S. Perumal, Trophic Transfer and Accumulation of Microplastics in Freshwater Ecosystem: Risk to Food Security and Human Health, Int. J. Ecol., 2022, 2022, 1–11, DOI:10.1155/2022/1234078.
- S. Gao, X. Mu, W. Li, Y. Wen, Z. Ma and K. Liu, et al., Invisible threats in soil: Microplastic pollution and its effects on soil health and plant growth, Environ. Geochem. Health, 2025, 47, 158, DOI:10.1007/s10653-025-02464-2.
- H. Yu, Y. Zhang, W. Tan and Z. Zhang, Microplastics as an Emerging Environmental Pollutant in Agricultural Soils: Effects on Ecosystems and Human Health, Front. Environ. Sci., 2022, 10 DOI:10.3389/fenvs.2022.855292.
- Z. Xiong, Y. Zhang, X. Chen, A. Sha, W. Xiao and Y. Luo, et al., Soil Microplastic Pollution and Microbial Breeding Techniques for Green Degradation: A Review, Microorganisms, 2024, 12, 1147, DOI:10.3390/microorganisms12061147.
- T. T. Kim, T. T. Bao, N. N. Trinh, L. T. K. Thoa, N. K. Phung and N. T. Bay, The Impact of Plastic on the Ecological Environment: The Pathway of Microplastics and the Control of Microplastic Pollution, Water, Air, Soil Pollut., 2025, 236, 684, DOI:10.1007/s11270-025-08311-5.
- J. C. Prata, J. P. da Costa, I. Lopes, A. C. Duarte and T. Rocha-Santos, Environmental exposure to microplastics: An overview on possible human health effects, Sci. Total Environ., 2020, 702, 134455, DOI:10.1016/j.scitotenv.2019.134455.
- A. Karami, A. Golieskardi, C. Keong Choo, V. Larat, T. S. Galloway and B. Salamatinia, The presence of microplastics in commercial salts from different countries, Sci. Rep., 2017, 7, 46173, DOI:10.1038/srep46173.
- C. J. Weber, C. Opp, J. A. Prume, M. Koch and P. Chifflard, Meso- and microplastic distribution and spatial connections to metal contaminations in highly cultivated and urbanised floodplain soilscapes – a case study from the Nidda River (Germany), Microplast. Nanoplast., 2022, 2, 25, DOI:10.1186/s43591-022-00044-0.
- R. Lehner, C. Weder, A. Petri-Fink and B. Rothen-Rutishauser, Emergence of Nanoplastic in the Environment and Possible Impact on Human Health, Environ. Sci. Technol., 2019, 53, 1748–1765, DOI:10.1021/acs.est.8b05512.
- J. C. Prata, Airborne microplastics: Consequences to human health?, Environ. Pollut., 2018, 234, 115–126, DOI:10.1016/j.envpol.2017.11.043.
- M. Revel, A. Châtel and C. Mouneyrac, Micro(nano)plastics: A threat to human health?, Curr. Opin. Environ. Sci. Health, 2018, 1, 17–23, DOI:10.1016/j.coesh.2017.10.003.
- M. Schneider, F. Stracke, S. Hansen and U. F. Schaefer, Nanoparticles and their interactions with the dermal barrier, Dermatoendocrinol, 2009, 1, 197–206, DOI:10.4161/derm.1.4.9501.
- P. Schwabl, S. Köppel, P. Königshofer, T. Bucsics, M. Trauner and T. Reiberger, et al., Detection of Various Microplastics in Human Stool, Ann. Intern. Med., 2019, 171, 453–457, DOI:10.7326/M19-0618.
- Z. Yan, Y. Liu, T. Zhang, F. Zhang, H. Ren and Y. Zhang, Analysis of Microplastics in Human Feces Reveals a Correlation between Fecal Microplastics and Inflammatory Bowel Disease Status, Environ. Sci. Technol., 2022, 56, 414–421, DOI:10.1021/acs.est.1c03924.
- L. Zhu, J. Zhu, R. Zuo, Q. Xu, Y. Qian and L. AN, Identification of microplastics in human placenta using laser direct infrared spectroscopy, Sci. Total Environ., 2023, 856, 159060, DOI:10.1016/j.scitotenv.2022.159060.
- A. Ragusa, V. Notarstefano, A. Svelato, A. Belloni, G. Gioacchini and C. Blondeel, et al., Raman Microspectroscopy Detection and Characterisation of Microplastics in Human Breastmilk, Polymers, 2022, 14, 2700, DOI:10.3390/polym14132700.
- H. A. Leslie, M. J. M. van Velzen, S. H. Brandsma, A. D. Vethaak, J. J. Garcia-Vallejo and M. H. Lamoree, Discovery and quantification of plastic particle pollution in human blood, Environ. Int., 2022, 163, 107199, DOI:10.1016/j.envint.2022.107199.
- K. Kadac-Czapska, P. Jutrzenka Trzebiatowska, M. Mazurkiewicz, P. Kowalczyk, E. Knez and M. Behrendt, et al., Isolation and identification of microplastics in infant formulas – A potential health risk for children, Food Chem., 2024, 440, 138246, DOI:10.1016/j.foodchem.2023.138246.
- G. B. Protyusha, K. B, R. S. Robin, N. A, T. R. Ineyathendral and S. S. Shivani, et al., Microplastics in oral healthcare products (OHPs) and their environmental health risks and mitigation measures, Environ. Pollut., 2024, 343, 123118, DOI:10.1016/j.envpol.2023.123118.
- S. Y. Salim, G. G. Kaplan and K. L. Madsen, Air pollution effects on the gut microbiota, Gut Microbes, 2014, 5, 215–219, DOI:10.4161/gmic.27251.
- K. D. Cox, G. A. Covernton, H. L. Davies, J. F. Dower, F. Juanes and S. E. Dudas, Human Consumption of Microplastics, Environ. Sci. Technol., 2019, 53, 7068–7074, DOI:10.1021/acs.est.9b01517.
- M. S.-L. Yee, L.-W. Hii, C. K. Looi, W.-M. Lim, S.-F. Wong and Y.-Y. Kok, et al., Impact of Microplastics and Nanoplastics on Human Health, Nanomaterials, 2021, 11, 496, DOI:10.3390/nano11020496.
- M. Bergmann, L. Gutow and M. Klages. Marine Anthropogenic Litter. 2015 Search PubMed.
- K. E. Carr, S. H. Smyth, M. T. McCullough, J. F. Morris and S. M. Moyes, Morphological aspects of interactions between microparticles and mammalian cells: intestinal uptake and onward movement, Prog. Histochem. Cytochem., 2012, 46, 185–252, DOI:10.1016/j.proghi.2011.11.001.
- J. J. Powell, V. Thoree and L. C. Pele, Dietary microparticles and their impact on tolerance and immune responsiveness of the gastrointestinal tract, Br. J. Nutr., 2007, 98, S59–S63, DOI:10.1017/S0007114507832922.
- C.-D. Dong, C.-W. Chen, Y.-C. Chen, H.-H. Chen, J.-S. Lee and C.-H. Lin, Polystyrene microplastic particles: In vitro pulmonary toxicity assessment, J. Hazard. Mater., 2020, 385, 121575, DOI:10.1016/j.jhazmat.2019.121575.
- A. Vianello, R. L. Jensen, L. Liu and J. Vollertsen, Simulating human exposure to indoor airborne microplastics using a Breathing Thermal Manikin, Sci. Rep., 2019, 9, 8670, DOI:10.1038/s41598-019-45054-w.
- R. Alvarez-Román, A. Naik, Y. N. Kalia, R. H. Guy and H. Fessi, Skin penetration and distribution of polymeric nanoparticles, J. Controlled Release, 2004, 99, 53–62, DOI:10.1016/j.jconrel.2004.06.015.
- D. A. Haines, G. Saravanabhavan, K. Werry and C. Khoury, An overview of human biomonitoring of environmental chemicals in the Canadian Health Measures Survey: 2007–2019, Int. J. Hyg. Environ. Health, 2017, 220, 13–28, DOI:10.1016/j.ijheh.2016.08.002.
- H. Louro, M. Heinälä, J. Bessems, J. Buekers, T. Vermeire and M. Woutersen, et al., Human biomonitoring in health risk assessment in Europe: Current practices and recommendations for the future, Int. J. Hyg. Environ. Health, 2019, 222, 727–737, DOI:10.1016/j.ijheh.2019.05.009.
- E. Govarts, L. Gilles, L. Rodriguez Martin, T. Santonen, P. Apel and P. Alvito, et al., Harmonized human biomonitoring in European children, teenagers and adults: EU-wide exposure data of 11 chemical substance groups from the HBM4EU Aligned Studies (2014–2021), Int. J. Hyg. Environ. Health, 2023, 249, 114119, DOI:10.1016/j.ijheh.2023.114119.
- G. Kutralam-Muniasamy, V. C. Shruti, F. Pérez-Guevara and P. D. Roy, Microplastic diagnostics in humans: “The 3Ps” Progress, problems, and prospects, Sci. Total Environ., 2023, 856, 159164, DOI:10.1016/j.scitotenv.2022.159164.
- K. Kannan and K. Vimalkumar, A Review of Human Exposure to Microplastics and Insights Into Microplastics as Obesogens, Front. Endocrinol., 2021, 12 DOI:10.3389/fendo.2021.724989.
- F. Alexis, E. Pridgen, L. K. Molnar and O. C. Farokhzad, Factors Affecting the Clearance and Biodistribution of Polymeric Nanoparticles, Mol. Pharm., 2008, 5, 505–515, DOI:10.1021/mp800051m.
- X. M. Bustamante-Marin and L. E. Ostrowski, Cilia and Mucociliary Clearance, Cold Spring Harbor Perspect. Biol., 2017, 9, a028241, DOI:10.1101/cshperspect.a028241.
- M. Geiser, Update on Macrophage Clearance of Inhaled Micro- and Nanoparticles, J. Aerosol Med. Pulm. Drug Delivery, 2010, 23, 207–217, DOI:10.1089/jamp.2009.0797.
- B. Kinkead, A. A. Ali, J.-C. Boyer and B. D. Gates, Optically Active Nanoparticle Coated Polystyrene Spheres, MRS Proc., 2013 DOI:10.1557/opl.2013.606.
- Y. Liu, W. Cheng, H. Xin, R. Liu, Q. Wang and W. Cai, et al., Nanoparticles advanced from preclinical studies to clinical trials for lung cancer therapy, Cancer Nanotechnol., 2023, 14, 28, DOI:10.1186/s12645-023-00174-x.
- M. Geiser and W. G. Kreyling, Deposition and biokinetics of inhaled nanoparticles, Part. Fibre Toxicol., 2010, 7, 2, DOI:10.1186/1743-8977-7-2.
- A. Banerjee and W. L. Shelver, Micro- and nanoplastic induced cellular toxicity in mammals: A review, Sci. Total Environ., 2021, 755, 142518, DOI:10.1016/j.scitotenv.2020.142518.
- L. F. Amato-Lourenço, L. dos Santos Galvão, L. A. de Weger, P. S. Hiemstra, M. G. Vijver and T. Mauad, An emerging class of air pollutants: Potential effects of microplastics to respiratory human health?, Sci. Total Environ., 2020, 749, 141676, DOI:10.1016/j.scitotenv.2020.141676.
- E. A. Sykes, Q. Dai, K. M. Tsoi, D. M. Hwang and W. C. W. Chan, Nanoparticle exposure in animals can be visualized in the skin and analysed via skin biopsy, Nat. Commun., 2014, 5, 3796, DOI:10.1038/ncomms4796.
- J.-L. Xu, X. Lin, J. J. Wang and A. A. Gowen, A review of potential human health impacts of micro- and nanoplastics exposure, Sci. Total Environ., 2022, 851, 158111, DOI:10.1016/j.scitotenv.2022.158111.
- J. Wen, H. Sun, Z. Liu, X. Zhu, Z. Qin and E. Song, et al., Aging Processes Dramatically Alter the Protein Corona Constitution, Cellular Internalization, and Cytotoxicity of Polystyrene Nanoplastics, Environ. Sci. Technol. Lett., 2022, 9, 962–968, DOI:10.1021/acs.estlett.2c00650.
- X. Guo and J. Wang, The chemical behaviors of microplastics in marine environment: A review, Mar. Pollut. Bull., 2019, 142, 1–14, DOI:10.1016/j.marpolbul.2019.03.019.
- J. Ren, N. Andrikopoulos, K. Velonia, H. Tang, R. Cai and F. Ding, et al., Chemical and Biophysical Signatures of the Protein Corona in Nanomedicine, J. Am. Chem. Soc., 2022, 144, 9184–9205, DOI:10.1021/jacs.2c02277.
- W. Wang, J. Ge and X. Yu, Bioavailability and toxicity of microplastics to fish species: A review, Ecotoxicol. Environ. Saf., 2020, 189, 109913, DOI:10.1016/j.ecoenv.2019.109913.
- D. Choi, J. Bang, T. Kim, Y. Oh, Y. Hwang and J. Hong, In vitro chemical and physical toxicities of polystyrene microfragments in human-derived cells, J. Hazard. Mater., 2020, 400, 123308, DOI:10.1016/j.jhazmat.2020.123308.
- S. Jeon, J. Clavadetscher, D.-K. Lee, S. V. Chankeshwara, M. Bradley and W.-S. Cho, Surface Charge-Dependent Cellular Uptake of Polystyrene Nanoparticles, Nanomaterials, 2018, 8, 1028, DOI:10.3390/nano8121028.
- L. Rubio, I. Barguilla, J. Domenech, R. Marcos and A. Hernández, Biological effects, including oxidative stress and genotoxic damage, of polystyrene nanoparticles in different human hematopoietic cell lines, J. Hazard. Mater., 2020, 398, 122900, DOI:10.1016/j.jhazmat.2020.122900.
- A.-K. Fuchs, T. Syrovets, K. A. Haas, C. Loos, A. Musyanovych and V. Mailänder, et al., Carboxyl- and amino-functionalized polystyrene nanoparticles differentially affect the polarization profile of M1 and M2 macrophage subsets, Biomaterials, 2016, 85, 78–87, DOI:10.1016/j.biomaterials.2016.01.064.
- Z. Yan, L. Xu, W. Zhang, G. Yang, Z. Zhao and Y. Wang, et al., Comparative toxic effects of microplastics and nanoplastics on Chlamydomonas reinhardtii: Growth inhibition, oxidative stress, and cell morphology, J. Water Process Eng., 2021, 43, 102291, DOI:10.1016/j.jwpe.2021.102291.
- S. Ye, M. Rao, W. Xiao, J. Zhou and M. Li, The relative size of microalgal cells and microplastics determines the toxicity of microplastics to microalgae, Process Saf. Environ. Prot., 2023, 169, 860–868, DOI:10.1016/j.psep.2022.11.077.
- W. Cheng, Y. Zhou, Y. Xie, Y. Li, R. Zhou and H. Wang, et al., Combined effect of polystyrene microplastics and bisphenol A on the human embryonic stem cells-derived liver organoids: The hepatotoxicity and lipid accumulation, Sci. Total Environ., 2023, 854, 158585, DOI:10.1016/j.scitotenv.2022.158585.
- W. Cheng, X. Li, Y. Zhou, H. Yu, Y. Xie and H. Guo, et al., Polystyrene microplastics induce hepatotoxicity and disrupt lipid metabolism in the liver organoids, Sci. Total Environ., 2022, 806, 150328, DOI:10.1016/j.scitotenv.2021.150328.
- H.-M. Kang, E. Byeon, H. Jeong, M.-S. Kim, Q. Chen and J.-S. Lee, Different effects of nano- and microplastics on oxidative status and gut microbiota in the marine medaka Oryzias melastigma, J. Hazard. Mater., 2021, 405, 124207, DOI:10.1016/j.jhazmat.2020.124207.
- Y. Deng, Y. Zhang, R. Qiao, M. M. Bonilla, X. Yang and H. Ren, et al., Evidence that microplastics aggravate the toxicity of organophosphorus flame retardants in mice (Mus musculus), J. Hazard. Mater., 2018, 357, 348–354, DOI:10.1016/j.jhazmat.2018.06.017.
- Y. Yuan, M. S. Sepúlveda, B. Bi, Y. Huang, L. Kong and H. Yan, et al., Acute polyethylene microplastic (PE-MPs) exposure activates the intestinal mucosal immune network pathway in adult zebrafish (Danio rerio), Chemosphere, 2023, 311, 137048, DOI:10.1016/j.chemosphere.2022.137048.
- Y. Liu, R. Guo, S. Zhang, Y. Sun and F. Wang, Uptake and translocation of nano/microplastics by rice seedlings: Evidence from a hydroponic experiment, J. Hazard. Mater., 2022, 421, 126700, DOI:10.1016/j.jhazmat.2021.126700.
- M. Prüst, J. Meijer and R. H. S. Westerink, The plastic brain: neurotoxicity of micro- and nanoplastics, Part. Fibre Toxicol., 2020, 17, 24, DOI:10.1186/s12989-020-00358-y.
- H. Jin, C. Yang, C. Jiang, L. Li, M. Pan and D. Li, et al., Evaluation of Neurotoxicity in BALB/c Mice following Chronic Exposure to Polystyrene Microplastics, Environ. Health Perspect., 2022, 130 DOI:10.1289/EHP10255.
- E.-J. Park, J.-S. Han, E.-J. Park, E. Seong, G.-H. Lee and D.-W. Kim, et al., Repeated-oral dose toxicity of polyethylene microplastics and the possible implications on reproduction and development of the next generation, Toxicol. Lett., 2020, 324, 75–85, DOI:10.1016/j.toxlet.2020.01.008.
- A. M. Kremer, T. M. Pal, J. S. Boleij, J. P. Schouten and B. Rijcken, Airway hyperresponsiveness, prevalence of chronic respiratory symptoms, and lung function in workers exposed to irritants, Occup. Environ. Med., 1994, 51, 3–13, DOI:10.1136/oem.51.1.3.
- W. L. ESCHENBACHER, K. KREISS, M. D. LOUGHEED, G. S. PRANSKY, B. DAY and R. M. CASTELLAN, Nylon Flock–Associated Interstitial Lung Disease, Am. J. Respir. Crit. Care Med., 1999, 159, 2003–2008, DOI:10.1164/ajrccm.159.6.9808002.
- T. Horvatits, M. Tamminga, B. Liu, M. Sebode, A. Carambia and L. Fischer, et al., Microplastics detected in cirrhotic liver tissue, EBioMedicine, 2022, 82, 104147, DOI:10.1016/j.ebiom.2022.104147.
- D. Wu, Y. Feng, R. Wang, J. Jiang, Q. Guan and X. Yang, et al., Pigment microparticles and microplastics found in human thrombi based on Raman spectral evidence, J. Adv. Res., 2023, 49, 141–150, DOI:10.1016/j.jare.2022.09.004.
- C. N. Andoh, F. Attiogbe, N. O. Bonsu Ackerson, M. Antwi and K. Adu-Boahen, Fourier Transform Infrared Spectroscopy: An analytical technique for microplastic identification and quantification, Infrared Phys. Technol., 2024, 136, 105070, DOI:10.1016/j.infrared.2023.105070.
- J.-L. Xu, K. V. Thomas, Z. Luo and A. A. Gowen, FTIR and Raman imaging for microplastics analysis: State of the art, challenges and prospects, TrAC, Trends Anal. Chem., 2019, 119, 115629, DOI:10.1016/j.trac.2019.115629.
- B. Singh and A. Kumar, Advances in microplastics detection: A comprehensive review of methodologies and their effectiveness, TrAC, Trends Anal. Chem., 2024, 170, 117440, DOI:10.1016/j.trac.2023.117440.
- A. Sridhar, D. Kannan, A. Kapoor and S. Prabhakar, Extraction and detection methods of microplastics in food and marine systems: A critical review, Chemosphere, 2022, 286, 131653, DOI:10.1016/j.chemosphere.2021.131653.
- M. A. Mohsin and A. H. Abd zaid, Microplastic Pollution in the Environment: A Chemical Engineering Perspective on Sources, Fate, and Mitigation Strategies, Polymers, 2025, 18, 29, DOI:10.3390/polym18010029.
- Z. Piao and Y. Yao, The role of biodegradable plastics in the global plastic future, Nat. Rev. Clean Technol., 2026 DOI:10.1038/s44359-025-00142-1.
- Z. Zhu, W. Liu, S. Ye and L. Batista, Packaging design for the circular economy: A systematic review, Sustain. Prod. Consum., 2022, 32, 817–832, DOI:10.1016/j.spc.2022.06.005.
- M. M. S. Chung, Y. Bao, B. Y. Zhang, T. M. Le and J.-Y. Huang, Life Cycle Assessment on Environmental Sustainability of Food Processing, Annu. Rev. Food Sci. Technol., 2022, 13, 217–237, DOI:10.1146/annurev-food-062420-014630.
- R. Patel, R. Trivedi, M. Raj and L. Raj, A brief review of polymeric blends based on natural polymers and synthetic thermoplastics polymers, Chem. Pap., 2024, 78, 665–697, DOI:10.1007/s11696-023-03139-x.
- J. R. A. Pires, V. G. L. Souza, P. Fuciños, L. Pastrana and A. L. Fernando, Methodologies to Assess the Biodegradability of Bio-Based Polymers—Current Knowledge and Existing Gaps, Polymers, 2022, 14, 1359, DOI:10.3390/polym14071359.
- E. Balla and D. N. Bikiaris, Editorial Outline: Overview of Recent Developments in Biobased and Biodegradable Polymers, Polymers, 2025, 17, 3264, DOI:10.3390/polym17243264.
- D. Moshood T, G. Nawanir, F. Mahmud, F. Mohamad, M. Hanafiah Ahmad and A. AbdulGhani, A Literature Review on Sustainability of Bio-Based and Biodegradable Plastics: Challenges and Opportunities, Energy Eng., 2022, 119, 1611–1647, DOI:10.32604/ee.2022.019028.
| This journal is © The Royal Society of Chemistry 2026 |