 Open Access Article
Open Access ArticleFunctional food formulation by incorporating the encapsulated extract of Phlogacanthus thyrsiflorus flower
Indrani Chetiaa,
Rituraj Chakraborty b,
Rupak Mukhopadhyay
b,
Rupak Mukhopadhyay b and
Laxmikant S. Badwaik
b and
Laxmikant S. Badwaik *a
*a
aDepartment of Food Engineering and Technology, School of Engineering, Tezpur University, Napaam 784028, Assam, India. E-mail: laxmikantbadwaik@gmail.com; Fax: +91-03712-267005; Tel: +91-9706368117
bDepartment of Molecular Biology and Biotechnology, School of Sciences, Tezpur University, Napaam 784028, Assam, India
First published on 24th March 2026
Abstract
Phlogacanthus thyrsiflorus flower (locally known as Nongmangkha), an underutilized medicinal flower, is a rich source of polyphenols with reported antioxidant, hypoglycemic, and hepatoprotective activities. However, its direct incorporation into food systems is constrained by sensory interference, thermal degradation of polyphenols, and limited bioavailability. This study presents a technological approach for delivering phenolics from this flower through encapsulation and their incorporation into watermelon gummies (containing 0.5–2% encapsulated flower extract) and ash gourd ready-to-serve (RTS) beverages (containing 1% encapsulated flower extract), with appropriate controls. The flower extract was obtained via ultrasound–microwave-assisted extraction (250 W, 15 min and 700 W, 5 min) and encapsulated through ionic gelation (3% sodium alginate and 5% CaCl2). The total phenolic content in the gummies increased (0.078 to 1.35 mg GAE per g), accompanied by an improved flavonoid content and antioxidant activity, without altering certain parameters such as moisture, titratable acidity (0.05–0.06%), pH (4.2–4.6), and total soluble solids (∼72 °Brix) in the gummies. Texture analysis revealed formulation-dependent changes in hardness and chewiness, while 1.5% incorporation of the encapsulated flower extract achieved optimal sensory acceptability. RTS beverages also demonstrated a significantly elevated phenolic content and antioxidant radical-scavenging capacity. The cell viability study of the flower extract exhibited minimal cytotoxicity in the HEK-293 cell line at a concentration of 12.5 µg mL−1. This study demonstrates a scalable approach for integrating encapsulated edible flower bioactives into functional food formulations. However, their in vivo bioavailability and storage stability warrant further investigation.
Sustainability spotlightA sustainable approach for preserving the bioactive compounds in edible flowers is incorporating the flower extract into food formulations, which can provide enhanced nutrient availability and various health benefits. The innovative aspect of this research lies in the encapsulation of the Nongmangkha (Phlogacanthus thyrsiflorus) flower extract and its utilization for the development of functional food products with enhanced stability and bioactive retention. This approach addresses key challenges such as low stability, poor bioavailability, and potential sensory issues of direct plant extract incorporation, offering a novel solution for fortifying foods such as gummies and beverages with edible flowers. |
1 Introduction
The growing interest in functional food products has paved the way for the development of innovative food products with the aim to provide health benefits beyond basic nutritional attributes. Consumers generally seek functional foods with enhanced sensory attributes such as aroma, flavor, texture, and visual appeal while also resembling traditional foods in the market.1 Functional foods are natural or processed foods that contain biologically active compounds. When they are consumed in nontoxic or adequate amounts, they provide clinically proven health benefits. Health promotion, disease risk reduction (e.g., chronic or viral conditions etc.), and management of their symptoms can be clinically evidenced using biomarkers.2 Functional foods bridge the gap between traditional or general food systems and nutrition by delivering nutritional and bioactive compounds beyond basic nutritional requirements, mitigating diseases, promoting health and reducing health care costs.One of the promising approaches involves incorporating extracts rich in bioactive compounds for the development of food formulations that can provide better nutrients and various health benefits.3 In contrast, direct use of phytochemicals encounters various challenges, as these substances are sensitive and unstable when exposed to some environmental factors such as heat, light, and oxygen, and they also have limited bioavailability, which may affect the color and taste of the developed food.4 In this case, encapsulation of flower extracts is an effective approach to address these limitations prior to their addition to food formulations. Encapsulation is a technique that encloses a core, i.e., bioactive compounds, within a wall material, enhancing their stability and controlled release while maintaining the properties of the extract. This technique is beneficial for the development of food products with a better shelf life, higher nutritional value, and better sensory attributes.
Phlogacanthus thyrsiflorus flower, also known as Nongmangkha, is a brick-red flower belong to the Acanthaceae family and is an important medicinal plant that blooms from February to April. These flowers were reported to deliver beneficial health effects on hyperlipidemia, as shown by their antioxidant and radical scavenging activities, and possessed hypoglycemic and hypolipidemic properties. These flowers were believed to cure pox, prevent skin diseases such as sores and scabies, have anti-allergic effects, treat wounds and tumors, act as a blood purifier, and treat kidney stones and liver disorders. They contain steroids, terpenoids, flavonoids, phenols, etc.5
Functional food products are described as offering additional health benefits beyond their conventional nutritional value.6 Importantly, during the development of functional food products, they must be standardized to ensure the safety of administering bioactive compounds as a health optimization tool. Indeed, it is crucial to set up a standardized process for the development of functional food products to guarantee the safe delivery of bioactive compounds to support their improvement of health.2 The demand for functional food products is increasing, which might be due to rising health care costs, the steady increase in life expectancy, interest by the elderly in the improvement of life quality, etc. Edible flowers can be termed as functional foods due to their nutritional properties, antioxidant activity, antimicrobial activity, color, flavor, mood and stress reduction capability, etc. Gummy candy is highly favored by all age groups. Gummy candies are produced using concentrated sugar solutions, gelling agents, and other components.7 Gummies are generally low in required nutritional value and may lead to obesity, hyperglycemia, tooth problems, etc.
The innovative aspect of this research lies in the utilization of an encapsulated flower extract for the development of functional food products. In this study, an encapsulated phytochemical extract from Phlogacanthus thyrsiflorus flower was incorporated into gummies and ready-to-serve (RTS) beverages and their physicochemical properties and sensory characteristics were explored. Also, the cytotoxicity of the flower extract was evaluated in this study.
2 Materials and methods
2.1. Materials
Flowers were collected from Sivasagar, Assam, India. Watermelon and ash gourd (https://www.google.com/search?sca_esv=1c8765e962fa8ef3&rlz=1C1YTUH_enIN1005IN1006&q=ash+gourd&spell=1&sa=X&ved=2ahUKEwiiucfqrZaMAxWiyjgGHajSA_UQkeECKAB6BAgIEAE) were collected from a local market in Tezpur, Assam, India. All the materials or ingredients used for the preparation of the gummies and RTS beverage including pectin, agar–agar, and citric acid were of food grade. All chemicals utilized for experimental analysis were analytical grade.2.2. Collection of flowers
Flowers were harvested during their full bloom season in the morning. The flowers were procured in the morning (6 am to 10 am) and transported to the laboratory on the same day and kept in refrigerator at 4 °C. The collected flowers were then cleaned, trimmed, and washed with distilled water before drying. Furthermore, the flowers were dried in the shade with blowing air (fan) and the dried flowers were ground, sieved and stored in an air-tight container under refrigerated conditions for further analysis.2.3. Preparation of flower extract
The flower extract was prepared using an ultrasound-pretreated microwave-assisted extraction technique, as standardized in our previous study.8 Briefly, 1 g of shade-dried flower powder was mixed with 10 mL of 80% ethanol (solid-to-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, w/v). The mixture was subjected to ultrasound pre-treatment at 250 W for 15 min to enhance cell wall disruption and facilitate solvent penetration. Subsequently, microwave-assisted extraction was performed at 700 W for 5 min to improve the recovery of bioactive compounds. The resulting extract was filtered through Whatman filter paper, and the solvent was removed using a rotary evaporator (ROTEVA-8703, Equitron, India) at 40 °C under reduced pressure. The dried extract was stored at 4 °C until further analysis and encapsulation.
10, w/v). The mixture was subjected to ultrasound pre-treatment at 250 W for 15 min to enhance cell wall disruption and facilitate solvent penetration. Subsequently, microwave-assisted extraction was performed at 700 W for 5 min to improve the recovery of bioactive compounds. The resulting extract was filtered through Whatman filter paper, and the solvent was removed using a rotary evaporator (ROTEVA-8703, Equitron, India) at 40 °C under reduced pressure. The dried extract was stored at 4 °C until further analysis and encapsulation.
2.4. Cytotoxicity assay
The crude flower extract was diluted to 1 mg mL−1 with Milli-Q water and filtered using a 0.2 µm nylon filter. In this assay, HEK 293 cells (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 per well) were seeded into a 96-well plate. The cells were treated with extract concentrations ranging from 12.5 to 200 µg mL−1 for 24 h. After incubation, MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) (5 mg mL−1) was added and incubated for 3 h. Post-incubation, the medium was removed carefully, and MTT dissolving solution was added. The absorbance was measured at a wavelength of 590 nm using a UV-vis spectrophotometer.
000 per well) were seeded into a 96-well plate. The cells were treated with extract concentrations ranging from 12.5 to 200 µg mL−1 for 24 h. After incubation, MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) (5 mg mL−1) was added and incubated for 3 h. Post-incubation, the medium was removed carefully, and MTT dissolving solution was added. The absorbance was measured at a wavelength of 590 nm using a UV-vis spectrophotometer.
2.5. Preparation of encapsulated flower extract
The flower extract obtained through ultrasound-pretreated microwave-assisted extraction was subsequently used for encapsulation. Encapsulation was carried out using the ionic gelation technique, following the method described by Patel et al.9 and Chetia et al.10 A 3% sodium alginate solution was prepared, and 3% of the dried flower extract (based on alginate weight) was dispersed uniformly in the alginate solution under continuous stirring to ensure homogeneity. Separately, a 5% calcium chloride solution was prepared at room temperature as the cross-linking medium. Encapsulation was performed using an encapsulator (Model B-390, Büchi, Switzerland), where the alginate–extract mixture was extruded dropwise into the calcium chloride solution under mild stirring to facilitate bead formation. The formed beads were allowed to cure in the calcium chloride solution for 30 min to ensure complete ionic crosslinking. Subsequently, the encapsulated flower extract were collected, washed gently with distilled water to remove excess calcium ions, and air-dried on glass Petri plates under ambient conditions. The dried encapsulated flower extract were packed in zip-lock pouches, placed in airtight containers, and stored at 4 °C until further use.2.6. Gummy preparation
The composition of the gummies was finalized by performing various trials to process the gummy development. Initially, watermelon juice (50 mL) was taken and a small amount of water was also added to mix well with the other ingredients. Then juice sugar (52.75%) was added and heated while stirring until it dissolved properly. Pectin (2.53%), agar–agar (2.11%) and citric acid (0.41%) were also added later to the mixture.11 The whole mixture was mixed properly until it reached to TSS 72 °Brix. Except for the control (C) set of gummies, 0.5%, 1%, 1.5% and 2% encapsulated flower extract were added to the gummies and coded as G1, G2, G3 and G4, respectively. The gummy mixture was poured into moulds immediately and kept in a refrigerator for setting properly. After the formation of gummies, they were kept in an air-tight container for further analysis (Fig. 1).2.7. RTS beverage preparation
In the case of the RTS beverage, ash gourd juice (50%) was added to a pre-prepared sugar syrup solution. The sugar syrup solution was prepared by dissolving sugar (11.5%) and citric acid (0.22%) in water (38.29%) with continuous stirring. The whole mixture was heated until it reached TSS 13 °Brix and homogenized with crude phytochemical extract (CERTS), and separately another set was prepared where encapsulates were added (BRTS). After that, the RTS beverage was filled in hot sterilized glass bottles and pasteurized at 90 °C for 25 min. 3 different sets of beverages were prepared including the control (CRTS), with 1% encapsulates (BRTS) and with 0.5% of flower extract (CERTS). They were kept in a refrigerator for further analysis.2.8. Proximate analysis
The freshly prepared 5 sets of gummies, which were coded as C, G1, G2, G3 and G4, were analysed for moisture, total protein, total fiber and ash by adopting the AOAC12 method. Moisture content was determined using a drying oven (NDO-710W, Eyela) at 105 °C. Ash was analyzed using a muffle furnace. Total fiber was analysed employing a Fibro plus (FES06) apparatus (Pelican Equipment, Chennai, India).2.9. Color analysis
The color parameters of all the gummies and RTS beverages were analyzed using a Hunter colorimeter (Hunter Lab, Reston, Virginia, USA). The color readings of L*, a* and b* parameters were recorded in triplicate for the gummies (G1, G2, G3 and G4) and RTS beverages (CRTS, BRTS and CERTS).2.10. Texture profile analysis (TPA)
The textural properties of the gummies were determined by employing a texture analyser (TA-XT plus, Stable Micro System, UK) using a 0.5 R probe and a load cell of 30 kg. The texture parameters such as hardness, adhesiveness, cohesiveness and chewiness were analyzed for the developed gummies (C, G1, G2, G3 and G4).2.11. Titratable acidity
The titratable acidity of the gummies and RTS beverage was determined by adopting a volumetric method, where 1 g of gummy was dissolved in 9 mL of Milli-Q water. In case of the RTS beverage, it was also mixed with Milli-Q water.13 After that, a few drops of phenolphthalein were mixed in the solution. This homogenized solution was titrated with sodium hydroxide (0.1 N) and continued until a purple color was observed. The titratable acidity (%) was determined using eqn (1).
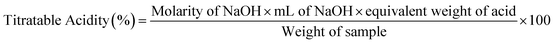 | (1) |
2.12. TSS and pH
Total soluble solids were determined in the gummies and RTS beverage. In a hand-held Atago refractometer, a 2 mm-thick slice sample (in the case of the gummies) was placed in the visor for the measurement.14 In the case of the RTS beverage, a few drops of beverage were placed in the visor of the refractometer and readings taken.pH was measured using a pH meter, which was equipped with a glass combined electrode. The gummies were cut into thin slices and mixed with hot water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 w/v) at 27 °C and homogenized.13 In the case of the RTS beverage, its pH was checked directly using the pH meter.
10 w/v) at 27 °C and homogenized.13 In the case of the RTS beverage, its pH was checked directly using the pH meter.
2.13. Water activity
The water activity of the gummies was measured using a water activity meter (Aqualab 4TE Decagon Devices Inc. Washington, USA) at 25 °C with 2 g of sample.132.14. Preparation of gummy extract for TPC, TFC and DPPH radical scavenging activity
The extraction of the gummies was done by adopting the method of Mohd et al.13 with slight modifications, using 5 g of gummy dissolved in 50 mL of distilled water. It was left stirring on a magnetic stirrer until it was dissolved well. After that, it was centrifuged at 6000 rpm for 15 min and the supernatant was filtered through Whatman no 1 filter paper. The extract was kept under refrigerated conditions for further analysis.2.15. Total phenolic content
The phenolic content of the gummies and RTS beverage was determined using the Folin–Ciocalteu reagent (FCR) method.15 Then, 0.2 mL of the gummy extract and the same amount of RTS beverage were added to separate test tubes. Each was mixed with 0.5 mL of FCR (diluted with water 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10), and after 5 minutes 2 mL of sodium carbonate (20%) was added. The whole mixture was incubated for 1 h. The absorbance was measured at 650 nm using a UV-vis spectrophotometer. Gallic acid was used as a standard for the calculation of the calibration curve and the results were represented as mg gallic acid equivalent per gram (gummies) or mL (RTS beverage).
10), and after 5 minutes 2 mL of sodium carbonate (20%) was added. The whole mixture was incubated for 1 h. The absorbance was measured at 650 nm using a UV-vis spectrophotometer. Gallic acid was used as a standard for the calculation of the calibration curve and the results were represented as mg gallic acid equivalent per gram (gummies) or mL (RTS beverage).
2.16. Total flavonoid content
The total flavonoid content in the gummies and RTS beverage was determined by following the method of Panhekar et al.15 1 mL of the extract was taken in a test tube and 4 mL of distilled water was added into it. After that, 0.3 mL of sodium nitrite (5%) was added to it, and after 5 min, 0.3 mL of aluminium chloride (AlCl3) (10%) was added. The whole mixture was shaken and kept for 5 min, and after that 2 mL of 1 M sodium hydroxide was added to the mixture and finally it was diluted to 10 mL using distilled water and mixed well. The absorbance of the whole mixture was recorded at 510 nm and the results were expressed in terms of quercetin equivalent (mg QE per g).2.17. DPPH radical scavenging activity
The DPPH radical scavenging activity of the gummies and RTS beverage was measured by adopting the procedure of Tundis et al.,16 with slight modifications. 0.5 mL of the extract was taken and mixed with 2.5 mL of DPPH solution (0.1 mM DPPH). This mixture was incubated for 30 min in the dark at room temperature, and its absorbance was recorded at 517 nm using a spectrophotometer. DPPH radical scavenging activity was calculated using eqn (2), as follows:
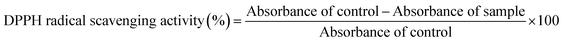 | (2) |
2.18. Sensory evaluation
The developed gummies were subjected to sensory evaluation by approximately 30 semi-trained panellists (male and female; age group 25–35 years) from the Department of Food Engineering and Technology, Tezpur University. Panellists were selected based on their regular consumption of similar confectionery products and the absence of known food allergies related to the ingredients used in this study. Prior to evaluation, the purpose of the study and the evaluation procedure were explained to the participants.Samples were coded with random numbers and presented in a randomized order to minimize bias. Sensory evaluation was conducted using a nine-point hedonic scale, where 1 represented “dislike extremely” and 9 represented “like extremely”. The panellists evaluated the samples for appearance, color, taste, mouthfeel, aftertaste, texture, and overall acceptability.
2.19. Statistical analysis
The data were analysed using analysis of variance (ANOVA) at a 95% level of significance (p < 0.05) using IBM SPSS Statistics v.20 (SPSS, Chicago, IL, USA). All experiments for the gummies and RTS beverage were carried out in triplicate and the results are expressed as mean ± standard deviation. ANOVA was performed to determine significant differences among treatments. Duncan's multiple-range test (DMRT) was applied as a post hoc test to identify pairwise differences between means at a significance level of p < 0.05.3 Results and discussion
3.1. Cytotoxicity of flower extract to HEK-293 cells
To determine the cell viability, HEK-293 cells were treated with various concentrations (12.5 to 200 µg mL−1) of the flower extract for 24 h. The extract exhibited appreciable cell viability (95.19% ± 4.72%) at 12.5 µg mL−1. Furthermore at 25 µg mL−1, the cell viability was 87.93% ± 1.11% (Table 1). Thus, this study indicates that at low doses, the extract exhibits minimal cytotoxicity to HEK-293 cells.| Concentration of extract (µg mL−1) | Cell viability (%) |
|---|---|
| Superscripts a–f in the ANOVA table denote significant differences (p < 0.05) determined by Duncan's multiple range test among means within rows. | |
| 0 (Control) | 100.00 ± 0.00a |
| 12.5 | 95.19 ± 4.72b |
| 25 | 87.93 ± 1.11c |
| 50 | 79.53 ± 5.36d |
| 100 | 70.33 ± 4.17e |
| 200 | 63.44 ± 2.37f |
3.2. Composition of gummies
The addition of encapsulated flower extract showed a significant enhancement in the nutritional and functional properties of the gummies compared to the control gummy. The moisture (wet basis), ash, total fiber and total protein contents of the gummies were analyzed, and the data are presented in Table 2. It was observed that the moisture content of all the gummies exhibited no significant differences (p > 0.05). Thus, it can be assumed that the incorporation of encapsulated flower extract did not considerably alter the water retention capacity of the product. This consistency is desirable for maintaining its textural properties and shelf stability. The lowest moisture content was observed in the G1 gummy, i.e. 35.16%, and the highest was found in the G3 gummy (37.18%). Burey et al.17 found a moisture content of 55.08% in gummies containing sucralose, which was used as the substitute sugar, and 29.44% in gummies when brown sugar was utilized. Teixeira-Lemos et al.18 found the moisture content of 18.2 (g water per g sample) in an orange and honey gummy and 20.82 (g water per g sample) in a mixed berries gummy. It can be understood that the moisture content of gummies varies based on the composition of their ingredients. The gummies were found to have an intermediate moisture content level due to their composition of high sugars along with other hygroscopic substances.| Parameters | Control | G1 | G2 | G3 | G4 |
|---|---|---|---|---|---|
| a All data are the mean ± SD of three replicates. Mean followed by different letters in the same row differs significantly (p ≤ 0.05).b G1 = 0.5% encapsulates added, G2 = 1% encapsulates added, G3 = 1.5% encapsulates added and G4 = 2% encapsulates added. | |||||
| Moisture (% wet basis) | 36.24 ± 0.94a | 37.26 ± 1.83 a | 37.97 ± 1.32 a | 37.85 ± 0.69 a | 37.04 ± 1.73a |
| Water activity | 0.74 ± 0.00 a | 0.78 ± 0.00b | 0.78 ± 0.00b | 0.79 ± 0.03b | 0.83 ± 0.03c |
| Ash (%) | 0.15 ± 0.05a | 0.68 ± 0.5b | 0.77 ± 0.12b | 0.77 ± 0.08b | 0.79 ± 0.07b |
| Crude fiber (%) | 0.26 ± 0.04a | 0.37 ± 0.04b | 0.37 ± 0.07b | 0.53 ± 0.03c | 0.57 ± 0.02c |
| Protein (%) | 0.74 ± 0.06a | 1.08 ± 0.09b | 1.38 ± 0.16c | 1.20 ± 0.2bc | 1.01 ± 0.17bc |
| Titratable acidity (%) | 0.05 ± 0.03a | 0.05 ± 0.09 a | 0.06 ± 0.02 a | 0.06 ± 0.05 a | 0.05 ± 0.05 a |
| pH | 4.33 ± 0.25 a | 4.3 ± 0.2 a | 4.6 ± 0.15 a | 4.2 ± 0.25 a | 4.3 ± 0.25 a |
| Total soluble solids (°Brix) | 72.17 ± 0.29 a | 71.83 ± 0.29 a | 72 ± 0.0 a | 72.17 ± 0.29 a | 72.17 ± 0.29 a |
| TPC (mg GAE per g) | 0.078 ± 0.01a | 0.089 ± 0.009a | 1.14 ± 0.056b | 1.20 ± 0.031b | 1.35 ± 0.020c |
| TFC (mg QE per g) | 0.015 ± 0.002a | 0.03 ± 0.004b | 0.041 ± 0.007bc | 0.048 ± 0.003 cd | 0.053 ± 0.03d |
| DPPH activity (%) | 12.016 ± 1.32a | 12.92 ± 0.33 ab | 13.83 ± 0.27b | 15.91 ± 0.23c | 16.35 ± 0.40c |
The ash content increased significantly in all the functional gummies compared to the control gummy (0.1%). The encapsulate-incorporated gummies were found to have an ash content ranging from 0.66% (G4) to 0.76% (G1). This enhancement is likely due to the mineral content of the encapsulated phytochemical matrix and the overall encapsulate composition, especially sodium alginate, demonstrating the nutritional enrichment of the gummies. The lowest ash content (0.1%) was recorded for the control, whereas G1 exhibited the highest value (0.76%). Viswanathan et al.19 compared the ash content of sodium alginate from various brown seaweeds, and reported that the highest ash content of 23.01% was observed in Padina gymnospora. Others such as Chnoospora implexa and Lobophora variegata showed ash contents of 21.53% and 12.78%, respectively. Given that the encapsulated flower extract were prepared using sodium alginate polymer, it can be seen that sodium alginate also increased the ash content in the gummies.
The total fiber content also exhibited a gradual increase from the control (0.29%) to the encapsulate-incorporated gummies. G4 (0.45%) showed a comparable fiber level to G3. The enhanced fiber levels are beneficial for functional food claims, as dietary fiber contributes to improved gut health. The crude fiber content showed a notable increase from the control (0.26) to G4 (0.45).
The control had the lowest protein content (0.79%), while G3 recorded the highest value (1.32%). A slight decline in protein content was observed in G3 and G4 compared to G2, suggesting that the treatment process might affect the protein stability or interaction with other components, which is also possibly due to the variability in ingredient dispersion at lower encapsulate concentrations.
3.3. Water activity
The water activity was also in the range from 0.67aw (C) to 0.78 aw (G3). The water activity showed a noticeable increase in the bead-containing samples compared to the control. G1 (0.75), G2 (0.77), and G3 (0.78) exhibited significantly higher values (p < 0.05). This increase might be attributed to the moisture-binding capacity of the encapsulates. The slightly elevated water activity of the gummies compared to the control gummy is consistent with their increased bead concentration, which can influence their microbial stability and shelf life. The water activity showed a significant variation across the groups. The increase in water activity from G1 to G4 suggests a possible correlation with the addition of encapsulates. Generally, the moisture content of gummies is intermediate, and they are high in sugars and other hygroscopic substances, resulting in low water activity.3.4. Titratable acidity, pH and TSS
There was no significant difference (p > 0.05) in titratable acidity (TA) and pH among the gummy formulations (Table 2). The TA values ranged between 0.05 and 0.06%, while the pH values varied from 4.2 to 4.6, indicating that the incorporation of the encapsulated flower extract up to 2% did not significantly alter the overall acidity of the gummies. This stability may be attributed to the sodium alginate–calcium chloride ionic gelation system, which likely restricted direct interaction between the encapsulated phenolic compounds and the gummy matrix. In high-sugar gel systems, organic acids such as citric acid primarily govern the total acidity; therefore, the addition of encapsulated phytochemicals at low concentrations does not substantially influence the formulation. All the samples retained the same TSS value of 72 °Brix, confirming that the inclusion of the encapsulates did not alter the sugar content or sweetness levels of the gummies. The total soluble solids (°Brix) showed no significant differences among the gummies, with values ranging from 71.83 to 72.17 °Brix.Furthermore, our earlier findings demonstrated that the encapsulation of a flower extract using ionic gelation (3% sodium alginate and 5% CaCl2) effectively protected the phenolic compounds, as evidenced by an in vitro release study showing controlled release.10 The encapsulation matrix provided protection against acidic degradation under simulated gastric conditions and prolonged the phenolic availability during the intestinal phase.
3.5. Total phenolic content (TPC) and total flavonoid content (TFC)
A significant increase in total phenolic content was observed from the control (0.078 mg GAE per g) to G4 (1.35 mg GAE per g) (Table 2). The progressive increase in TPC with an increasing amount of encapsulates highlights the potential enhancement of phenolic compounds in the gummies. This trend highlights the contribution of encapsulated phytochemicals to the functional properties of the gummies.The total flavonoid content also increased significantly among the gummies. The control exhibited the lowest TFC (0.015 mg QE per g), while G4 recorded the highest value (0.053 mg QE per g). This trend aligns with the observed increase in phenolic content, reinforcing the contribution of flavonoids to the antioxidant profile of the treated samples and indicating that a higher amount of encapsulates is necessary to achieve significant flavonoid enrichment.
Phenolic enrichment in functional food products has been widely reported when plant extracts are incorporated.20 An earlier study found that jelly formulated with anthocyanin-loaded microcapsules exhibited enhanced physicochemical and functional properties compared to the control sample.21 The substantial increase in TPC confirms that the alginate-based encapsulation system successfully protected and incorporated bioactive compounds without significant degradation during heating and gel setting.
3.6. DPPH radical scavenging activity (%)
The antioxidant activity, measured as DPPH radical scavenging activity, was observed to increase significantly with an increase in encapsulate concentration in the gummies from 12.016% (control) to 16.35% (G4) (Table 2). These results demonstrate the ability of encapsulates to deliver bioactive compounds or phenolic compounds with antioxidant properties into the food matrix effectively. The correlation among TPC, TFC, and DPPH activity further underscores the role of phenolic and flavonoid compounds in enhancing antioxidant property. The binding properties of sodium alginate may aid in the retention or activation of phenolic and flavonoid compounds, as reported by Huang et al.22 The progressive increase in TPC and TFC, along with improved DPPH radical scavenging activity, reflects the potential of sodium alginate to enhance antioxidant property.233.7. Color
The color parameters (L*, a*, and b*) of the gummies revealed significant differences among them (Table 3). The lightness (L*) values decreased from 46.44 (control) to 43.88 (G4), indicating a slight darkening of the gummies. The redness (a*) values increased significantly in the gummies, with values ranging from 16.91 (control) to 18.68 (G3), though no significant differences were observed among the gummies incorporated with encapsulates. Yellowness (b*) showed a significant increase from 11.91 (control) to 13.73 (G3), suggesting enhanced chromaticity in the treated samples. The observed changes in color parameters could be attributed to the Maillard reaction and pigment incorporation resulting from the addition of colored encapsulates. The slight darkening and increased chromaticity observed are consistent with the findings reported by Kim et al.,24 who attributed these changes to the Maillard reaction and pigment transformation during processing or incorporation of encapsulates.| Sample | L* | a* | b* |
|---|---|---|---|
| a All data are the mean ± SD of three replicates. Mean followed by different letters in the same column differs significantly (p ≤ 0.05).b G1 = 0.5% encapsulates added, G2 = 1% encapsulates added, G3 = 1.5% encapsulates added and G4 = 2% encapsulates added. | |||
| Control (C) | 46.74 ± 0.87c | 16.91 ± 1.28a | 11.91 ± 1.02a |
| G1 | 45.82 ± 0.48bc | 19.01 ± 0.70b | 12.68 ± 0.15 ab |
| G2 | 44.91 ± 0.24 ab | 19.74 ± 0.17b | 13.40 ± 0.27bc |
| G3 | 44.87 ± 0.11 ab | 19.89 ± 0.072b | 13.70 ± 0.08c |
| G4 | 43.88 ± 1.08a | 18.68 ± 0.88b | 13.73 ± 0.35c |
3.8. Texture profile analysis
Texture is an essential parameter to analyze for gummies or any other confectionary items. Hardness, springiness, cohesiveness, gumminess and chewiness were measured for all the gummy formulations and the results are shown in Table 4. The hardness values varied significantly (p < 0.05) across the gummies. The control gummy exhibited a hardness of 36.21 g, which was higher than that of G3 (30.28 g) but lower than that of G1 (40.07 g). G4 had a value of 38.51 g, indicating that the addition of encapsulates affected its firmness. G2 showed intermediate hardness (34.84 g), suggesting a moderate effect of the treatment. Pyrovolou et al.25 reported that the hardness of commercial jelly gum was 43.1 (N), which is close to our results. Springiness decreased significantly from the G1 (0.98) to G4 (0.56) gummies, with the control showing a value of 0.93. G1 exhibited a relatively higher springiness, indicating potential variations in elasticity due to structural modifications from the encapsulates. Springiness and cohesiveness showed a declining trend with increasing encapsulate incorporation, suggesting reduced elastic recovery and internal bonding strength. The incorporation of encapsulates may disrupt the matrix continuity and reduce its elastic resilience. Cohesiveness helps to determine the strength, the breakable range of inner bonds that hold the structure of gummy, and deformation.26 The cohesiveness varied across the samples, with the highest value observed in G1 (0.08) and the lowest in G4 (0.029). The control group displayed a moderate cohesiveness of 0.04. Gumminess and chewiness followed similar trends, as these parameters are dependent on both hardness and cohesiveness. Chewiness and gumminess are closely related to the hardness of a gummy. The firmer the structure of the gummy, the harder it becomes, resulting in increased chewiness and gumminess.27 The gumminess values followed a similar trend, with the control recording the highest gumminess (11.39) and G3 the lowest (0.69). Chewiness decreased significantly (p < 0.05) compared to the control (10.59) in all the gummies.| Sample | Hardness (N) | Springiness | Cohesiveness | Gumminess (N) | Chewiness (N) |
|---|---|---|---|---|---|
| a All data are the mean ± SD of three replicates. Mean followed by different letters in the same row differs significantly (p ≤ 0.05).b G1 = 0.5% encapsulates added, G2 = 1% encapsulates added, G3 = 1.5% encapsulates added and G4 = 2% encapsulates added. | |||||
| Control (C) | 36.21 ± 0.44c | 0.931 ± 0.05c | 0.315 ± 0.04c | 11.39 ± 1.34c | 10.59 ± 1.18c |
| G1 | 40.07 ± 0.32e | 0.975 ± 0.05c | 0.083 ± 0.01b | 3.34 ± 0.48b | 3.27 ± 0.63b |
| G2 | 34.84 ± 1.16b | 0.800 ± 0.07b | 0.037 ± 0.01a | 1.31 ± 0.24a | 1.04 ± 0.23a |
| G3 | 30.28 ± 0.54a | 0.706 ± 0.05b | 0.030 ± 0.01a | 0.98 ± 0.24a | 0.69 ± 0.17a |
| G4 | 38.51 ± 0.62d | 0.560 ± 0.09a | 0.029 ± 0.01a | 1.62 ± 0.14a | 0.90 ± 0.11a |
3.9. Sensory evaluation
Sensory evaluation is a scientific field that measures, examines, and interprets how people react to or accept certain food by their sense organs.28 Sensory evaluation of food applies experimental design principles and statistical analysis. Sensory analysis of the gummies was done within 24 h of their preparation. As mentioned earlier, a 9 point hedonic scale was used with 1 being extremely disliked, 2 signified slightly disliked, 3 moderately disliked, 4 slightly disliked, 5 neither liked nor disliked, 6 slightly liked, 7 moderately liked, 8 highly liked and 9 extremely liked.27 All the gummies were evaluated by parameters including appearance, color, taste, mouth feel, after taste, texture and overall acceptability with the help of semi-trained panellists and the results are shown in Fig. 2. The responses from the panellists presented more insights and inferences about the gummies. In many ways, our perception of products is largely shaped by visual cues, especially color perception.29 Since color significantly affects consumer appetite, it plays a crucial role in the overall palatability of food.30 According to Fig. 2, it can be observed that the appearance that was most appreciated by the panellists was the control gummy, and after that G3. In the case of color, control gummy C was also highly accepted, followed by gummy G3. Similarly, taste was also highly appreciated by control gummy C, followed by gummy G3. Thus, the addition of encapsulates affected the taste of the gummies. Mouth feel was found mostly accepted in the case of control gummy C, followed by G1 and G3. The after taste was found highly appreciated both in control gummy C and G3. Texture was highly accepted in control gummy C, followed by G2. The addition of encapsulates increased the hardness of the gummies, which might not be liked by the sensory panellists. Overall acceptability was observed for the G3 and control gummies. Thus, it can be assumed that along with the control gummy, the G3 gummy showed the best consumer acceptability.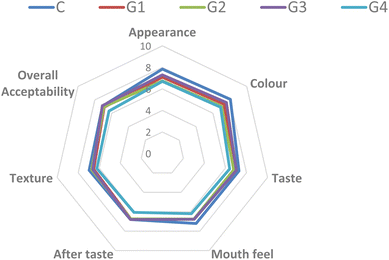 | ||
| Fig. 2 Sensory score of gummies. G1 = 0.5% encapsulates added, G2 = 1% encapsulates added, G3 = 1.5% encapsulates added and G4 = 2% encapsulates added. | ||
3.10. Properties of RTS beverage
The results of the study on the ash gourd ready-to-serve (RTS) beverage formulations are presented in Table 5, which include physicochemical properties and color analysis of CRTS, CERTS, and BRTS. These parameters play a crucial role in determining the quality, stability, and consumer acceptability of RTS beverages. The physicochemical characteristics of the RTS formulations indicate significant variations among CRTS, CERTS, and BRTS in terms of titratable acidity, pH, TPC, TFC, and DPPH radical scavenging activity.| Sl no. | Parameters | CRTS | CERTS | BRTS |
|---|---|---|---|---|
| a All data are the mean ± SD of three replicates. Mean followed by different letters in the same row differs significantly (p ≤ 0.05). | ||||
| 1 | Titratable acidity (%) | 0.27 ± 0.02a | 0.31 ± 0.08a | 0.27 ± 0.09b |
| 2 | pH | 3.37 ± 1.84a | 3.7 ± 1.47a | 3.23 ± 0.82a |
| 3 | TPC (mg mL−1) | 0.41 ± 0.07a | 3.81 ± 0.15c | 1.19 ± 0.17b |
| 4 | TFC (mg mL−1) | 0.07 ± 0.005a | 0.64 ± 0.008c | 0.49 ± 0.004b |
| 5 | DPPH activity (%) | 37.61 ± 1.85a | 57.17 ± 1.48c | 51.43 ± 3.2b |
| 6 | L* | 31.57 ± 2.45a | 27.05 ± 3.52c | 29.06 ± 2.85b |
| 7 | a* | −0.42 ± 0.03a | 0.9 ± 0.07c | −0.27 ± 0.02b |
| 8 | b* | 2.86 ± 0.24a | 3.98 ± 1.18b | 8.19 ± 2.09c |
3.11. Total phenolic content, total flavonoid content and DPPH radical scavenging activity
The TPC of CRTS, CERTS and BRTS was estimated and it was observed (Table 5) that the addition of both encapsulates and crude significantly (p < 0.05) increased the TPC, TFC and DPPH radical scavenging activity. The phenolic content of CERTS (3.81 mg GAE per mL) was significantly higher than that of BRTS (1.19 mg GAE per mL) and CRTS (0.41 mg mL−1). Similarly, the flavonoid content followed the same trend (Table 5), with CERTS (0.64 mg QE per mL) exhibiting the highest TFC, followed by BRTS (0.49 mg QE per mL) and CRTS (0.07 mg QE per mL). Phenolic and flavonoid compounds contribute to antioxidant properties and have been linked to several health benefits, including reduced oxidative stress and improved cardiovascular health.20 The higher phenolic and flavonoid contents in CERTS could be attributed to the presence of certain bioactive compounds present in the crude extract and in the encapsulates. The antioxidant activity (Table 5), as measured by DPPH radical scavenging percentage, was the highest in CERTS (57.17%), followed by BRTS (51.43%) and CRTS (37.61%). The high antioxidant activity of CERTS may be directly related to its higher phenolic and flavonoid contents. The high antioxidant activity of CERTS suggests that it may offer greater protection against oxidative stress, which is beneficial for health-conscious consumers. BRTS, with moderate antioxidant activity, remains a good alternative, whereas CRTS, with the lowest antioxidant capacity, is likely less effective in delivering health benefits. This suggests that CERTS might have better health-promoting properties, making it a more potent functional beverage.3.12. Titratable acidity and pH
The acidity levels varied slightly, with CERTS showing (Table 5) the highest titratable acidity (0.31%), followed by CRTS (0.27%) and BRTS (0.27%). The pH values were also within a narrow range (3.23–3.7) (Table 5), with CERTS exhibiting the highest pH (3.7), suggesting its relatively lower acidity compared to CRTS and BRTS. The pH and acidity levels significantly affect the microbial stability and sensory properties of beverages.31 A higher acidity level typically enhances the shelf life and contributes to a tart flavor, which can be desirable in fruit-based beverages. CERTS had slightly higher acidity, making it more stable and tangier.3.13. Color analysis
Color is an essential quality attribute of RTS beverages, influencing consumer preference and perception. CRTS exhibited the highest lightness value (31.57), while CERTS had the lowest (27.05) (Table 5). The incorporation of the flower extract significantly affected the color attributes (L*, a*, and b*) of the ash gourd RTS beverages compared to the control sample. A decrease in L* value indicates reduced lightness, which can be attributed to the natural pigments and phenolic compounds present in the flower extract. Plant-derived phenolics and flavonoids are known to contribute to darker tones due to their inherent chromophoric structures and potential oxidation reactions in aqueous systems. BRTS exhibited an intermediate L* value of 29.06. Lower L* values suggest a darker appearance, which may be due to the presence of more intense pigments from the crude extract of flowers. The lower L* value for CERTS suggests a more intense color, potentially due to a higher concentration of anthocyanins or carotenoids, which also contribute to its superior antioxidant properties. An earlier study reported similar reductions in lightness in fruit- and herb-enriched functional beverages due to the addition of phenolic-rich extracts.20 The a* values showed that CRTS (−0.42) and BRTS (−0.27) were more towards the green spectrum, whereas CERTS (0.9) had a slight red hue. The positive a* value for CERTS suggests that it contains higher anthocyanin or carotenoid pigments, which could also be linked to its higher phenolic content. The b* values revealed significant differences in the yellow-blue color range. BRTS (8.19) exhibited the highest b* value, indicating a more intense yellowish appearance, whereas CRTS had the lowest (2.86) and CERTS was in between (3.98). The variations in b* values suggest differences in ingredient composition, effect of processing conditions on the encapsulates and crude flower extract, and also pigment stability. The encapsulated extract exhibited comparatively moderate color changes relative to the incorporation of the crude extract. This may be attributed to the protective alginate matrix, which can limit immediate pigment dispersion and reduce direct interaction with the beverage environment.4 Conclusions
The flower extract showed minimal cytotoxicity at 12.5 µg mL−1 in HEK-293 cells. While this observation is highly encouraging, future studies using appropriate animal models are required to ascertain the food safety parameters of this extract. In the case of gummies, notable enhancements were observed in ash content, crude fiber, protein, and antioxidant activity, along with significant increases in TPC, TFC and DPPH radical scavenging activity. However, variations in textural parameters such as hardness, springiness, and chewiness indicate that encapsulate incorporation influenced the structural properties of the gummies. In RTS beverages, the beverage with 0.5 % of crude flower extract (CERTS) exhibited higher phenolic and flavonoid contents and consequently stronger antioxidant activity than CRTS and BRTS. Future research should focus on optimizing the formulation parameters, evaluating storage stability, and investigating in vitro and in vivo bioavailability to substantiate the health-promoting claims and support the commercial scalability of these functional food products.Ethical statement
This study does not involve any human or animal testing.Author contributions
Indrani Chetia: data curation, formal analysis, investigation; and writing – original draft; Rituraj Chakraborty: methodology and resources for carrying out cytotoxicity assay of the flower extract. Rupak Mukhopadhyay: methodology and resources to carrying out cytotoxicity assay of the flower extract; and Laxmikant S. Badwaik: conceptualization, project administration, resources, supervision, and writing – review & editing.Conflicts of interest
The authors have declared that there are no conflicts of interest in this work.Data availability
All data are presented in this article.Acknowledgements
The financial support received by Ms Indrani Chetia from Tezpur University, Napaam, Assam, India, under Research and Innovation Grant (No. DoRD/RIG/10-73/1544-A; dated 21.12.2020) for carrying out this work is duly acknowledged. Authors are thankful for the support received from Tezpur University, DST-FIST and UGC-SAP for carrying out this work.References
- M. T. Baker, P. Lu, J. A. Parrella and H. R. Leggette, Consumer acceptance toward functional foods: A scoping review, Int. J. Environ. Res. Public Health, 2022, 19, 1217 CrossRef PubMed.
- D. Martirosyan, H. Kanya and C. Nadalet, Can functional foods reduce the risk of disease? Advancement of functional food definition and steps to create functional food products, Funct. Foods Health Dis., 2021, 11, 213–221 Search PubMed.
- J. Xiao and W. Bai, Bioactive phytochemicals, Crit. Rev. Food Sci. Nutr., 2019, 59, 827–829 CrossRef PubMed.
- M. Hao, X. Tan, K. Liu and N. Xin, Nanoencapsulation of nutraceuticals: enhancing stability and bioavailability in functional foods, Front. Nutr., 2025, 12, 1746176 Search PubMed.
- S. Ponneganti, U. S. Murty, C. Bagul, R. M. Borkar and P. Radhakrishnan, Phyto-metabolomics of Phlogacanthus thyrsiformis by using LC-ESI-QTOF-MS/MS and GC/QTOF-MS: Evaluation of antioxidant and enzyme inhibition potential of extracts, Food Res. Int., 2022, 161, 111874 Search PubMed.
- M. Fekete, A. Lehoczki, A. Kryczyk-Poprawa, V. Zábó, J. T. Varga, M. Bálint and P. Varga, Functional foods in modern nutrition science: mechanisms, evidence, and public health implications, Nutrients, 2025, 17, 2153 Search PubMed.
- M. Tarahi, S. Hedayati, M. R. Kianiani, S. Tahmouzi, S. Ebrahimi and M. Niakousari, Current innovations in the development of functional gummy candies, Foods, 2023, 13, 45 Search PubMed.
- I. Chetia, A. J. Das and L. S. Badwaik, Extraction of phenolic compounds from edible flowers Phlogacanthus thyrsiflorus using ultrasound-microwave-assisted extraction and its screening by HPLC, J. Food Process Eng., 2024, 47, e14616 CrossRef CAS.
- N. Patel, D. Lalwani, S. Gollmer, E. Injeti, Y. Sari and J. Nesamony, Development and evaluation of a calcium alginate based oral ceftriaxone sodium formulation, Prog. Biomater., 2016, 5, 117–133 Search PubMed.
- I. Chetia, T. Tongbram, S. P. G. Ponnam and L. S. Badwaik, Alginate-based encapsulation of edible flower extract: Process optimization, characterization and in vitro release kinetics, Int. J. Biol. Macromol., 2025, 148877 Search PubMed.
- M. Krajewska, P. Findura, D. Andrejko, J. Mazur, M. Prístavka and A. Nawrocka, Effect of watermelon pomace on physicochemical, textural, and antioxidant properties of vegan jellies, Sci. Rep., 2025, 15, 34251 Search PubMed.
- AOAC, Official Methods of Analysis, Association of Official Analytical Chemists, Washington, DC, 2000 Search PubMed.
- N. S. Mohd Isa, H. Y. Cheok, A. Z. Mohsin and N. M. Mohd Maidin, The Effect of Different Citric Acid Concentrations on Physicochemical and Antioxidant Properties of Red Pitaya Peel Gummy Candies, Trop. J. Nat. Prod. Res., 2024, 8(12), 9654–9663 Search PubMed.
- P. Delgado and S. Bañón, Determining the minimum drying time of gummy confections based on their mechanical properties, CyTA–Journal of Food, 2015, 13, 329–335 Search PubMed.
- D. Panhekar, T. Sawant and D. P. Gogle, Phytochemicals Extraction, Separation and Analysis, Global Education Limited, 2019 Search PubMed.
- R. Tundis, M. Marrelli, F. Conforti, M. C. Tenuta, M. Bonesi, F. Menichini and M. R. Loizzo, Trifolium pratense and T. repens (Leguminosae): Edible flower extracts as functional ingredients, Foods, 2015, 4, 338–348 CrossRef CAS PubMed.
- P. Burey, B. R. Bhandari, R. P. G. Rutgers, P. J. Halley and P. J. Torley, Confectionery gels: A review on formulation, rheological and structural aspects, Int. J. Food Prop., 2009, 12, 176–210 CrossRef CAS.
- E. Teixeira-Lemos, A. R. Almeida, B. Vouga, C. Morais, I. Correia, P. Pereira and R. P. Guiné, Development and characterization of healthy gummy jellies containing natural fruits, Open Agric., 2021, 6, 466–478 Search PubMed.
- S. Viswanathan and T. Nallamuthu, Extraction of sodium alginate from selected seaweeds and their physiochemical and biochemical properties, Extraction, 2014, 3(4), 10998–11002 Search PubMed.
- F. Shahidi and P. Ambigaipalan, Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects – A review, J. Funct. Foods, 2015, 18, 820–897 Search PubMed.
- L. M. Devi and L. S. Badwaik, Development of functional coconut water jelly by incorporating anthocyanin microcapsules and its characterization, J. Food Meas. Charact., 2025, 19, 1981–1991 CrossRef.
- Y. Huang, L. Zhao and S. Liu, Role of sodium alginate in preserving bioactive compounds during food processing, J. Agric. Food Chem., 2019, 67, 542–550 Search PubMed.
- H. Zhou, J. Liang, J. Xiao, Y. Liu, L. Sun and Y. Wang, Sodium alginate encapsulation enhances phenolic retention and antioxidant capacity in functional foods, Int. J. Biol. Macromol., 2020, 164, 2887–2895 Search PubMed.
- H. Kim, J. Park and K. Lee, Effects of thermal processing on color and antioxidant properties of food products, Food Qual. Prefer., 2019, 27, 112–119 Search PubMed.
- K. Pyrovolou, E. C. Panopoulou, C. Tsogka, A. Sklavou, E. Gogou, I. F. Strati and A. Batrinou, Development of Calcium Alginate Hydrogels with Chlorella, Carob (Cetatonia siliqua L.) and Encapsulated Probiotics in Edible Jelly-Gums with Enhanced Bioactivity, Gels, 2025, 12, 1 Search PubMed.
- E. Bölük, D. S. Atik, S. Kolaylı, A. Ş. Demirci and I. Palabiyik, Delivery of phenolics and caffeic acid phenethyl ester by propolis resin: Chewing gum system, Food Biosci., 2021, 41, 101090 Search PubMed.
- M. M. Mahat, A. S. M. Sabere, M. A. Nawawi, H. H. Hamzah, M. A. F. M. Jamil, N. C. Roslan and M. F. Safian, The sensory evaluation and mechanical properties of functional gummy in the Malaysian market, Preprints, 2020 DOI:10.20944/preprints202010.0213.v1.
- P. B. Lohar, P. M. Shrivastav and S. S. Kulavoor, Development of fiber rich gummies, IJARIIT, 2020, 6, 52–55 Search PubMed.
- R. Spencer, A. Primbetova, A. N. Koppes, R. A. Koppes, H. Fenniri and N. Annabi, Electroconductive gelatin-methacryloyl-PEDOT:PSS composite hydrogels: Design, synthesis, and properties, ACS Biomater. Sci. Eng., 2018, 4, 1558–1567 Search PubMed.
- A. Patras, Stability and colour evaluation of red cabbage waste hydroethanolic extract in presence of different food additives or ingredients, Food Chem., 2019, 275, 539–548 CrossRef CAS.
- D. Saha, J. Shinde and S. Mishra, Effect of pH and acidity on microbial stability of fruit-based beverages, J. Food Sci. Technol., 2020, 57, 921–930 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |

