Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Characterization of Ziziphus jujuba seeds and optimization of waffle formulations via D-optimal mixture design
Aman
Jyoti
a,
Mahendra
Gunjal
 a,
Prasad
Rasane
a,
Prasad
Rasane
 a,
Supriya Singh
Gaur
a,
Sezai
Ercisli
bc and
Jyoti
Singh
a,
Supriya Singh
Gaur
a,
Sezai
Ercisli
bc and
Jyoti
Singh
 *a
*a
aDepartment of Food Technology and Nutrition, Lovely Professional University, Phagwara, Punjab 144411, India. E-mail: jyotisingh9377@gmail.com; Tel: +91-8146637739
bDepartment of Horticulture, Faculty of Agriculture, Ataturk University, 25240 Erzurum, Turkiye
cHGF Agro, ATA Teknokent, Erzurum, Turkiye
First published on 13th March 2026
Abstract
This study aimed to increase the nutritional and bioactive composition of waffles by incorporating Ziziphus jujuba seed flour without affecting their sensory quality parameters. Preliminary testing identified the nutritional composition of the seeds, followed by FTIR and SEM characterization. Using D-optimal design, 24 multigrain waffle formulations with varying proportions of jowar, bajra, wheat, and seed flour were developed. The optimal formulation containing jowar (25%), bajra (25%), wheat (40%), and jujuba seed (10%) flour achieved high nutritional and sensory scores. The formulation had a flavonoid content of 48.17 ± 0.01 mg QE per g, phenolic content of 32.39 ± 0.04 mg GAE per g, and antioxidant activity (DPPH) of 25.43 ± 0.01%. The overall acceptability score was 8.24 ± 0.02. Further analyses of proximate composition, shelf life, and antioxidant activity were conducted using two packaging materials (metallic polyester and low-density polyethylene) at three temperatures (8 °C, 25 °C, and 37 °C). The metallic polyester retained the bioactive components during the selected storage period. The results suggest that Ziziphus jujuba seed flour significantly enhances the nutritional value of waffles while maintaining sensory qualities, indicating commercial potential.
Sustainability spotlightThis study highlights a sustainable approach to food innovation by valorizing Ziziphus jujuba seeds, an underutilized by-product of fruit processing, into value-added functional ingredients. Through D-optimal mixture design, the research optimizes waffle formulations that not only enhance nutritional quality but also minimize food waste and promote resource efficiency. By integrating food waste valorization with product development, the work contributes to sustainable food system goals and supports circular economy principles within the food industry. |
1 Introduction
Waffles are a popular breakfast option worldwide owing to their flavor and versatility. Nevertheless, there is an increasing need for healthier options that satisfy customers who care about their health. This has led to interest in improving the nutritional profiles of traditional foods without compromising their taste.1Ziziphus jujuba, commonly known as jujube or Chinese date, is a deciduous species in the family Rhamnaceae. It is native to China, where it has been cultivated for more than 4000 years for both nutritional and medicinal purposes. Over time, Ziziphus jujuba spread to other regions of Asia, the Middle East, Europe, and parts of Africa, adapting well to diverse climatic conditions. The many health advantages of jujube seeds are attributed to their rich nutritional qualities, which include high concentrations of flavonoids, phenolic compounds, and antioxidants.2 The seeds of Ziziphus jujuba are valuable resources with a wide range of uses. Studies have shown that these seeds are rich in fatty acids such as oleic and linoleic acids, which possess anti-inflammatory and antioxidant properties.3 They can be used as natural nutritional supplements to delay aging, treat insomnia, and improve learning and memory. Mature seeds of wild jujube have positive effects on central nervous system disorders.4Ziziphus jujuba seeds, typically discarded as waste, can be repurposed to produce functional beverages such as herbal tea or coffee, which may improve sleep and cognitive function through their sedative and neuroprotective effects.2The novelty of this study lies in the use of Ziziphus jujuba seed flour, an underutilized fruit by-product, as a functional ingredient in waffle formulations. The incorporation of this seed flour into bakery products has not been previously explored. This study is the first to demonstrate that jujube seed flour can be effectively used to enhance the nutritional value and antioxidant activity of waffles while maintaining acceptable sensory characteristics.
2 Materials and methodology
2.1. Collection of raw material
Ziziphus jujuba fruits were purchased from a local market in Ludhiana, Punjab, India. The fruits were thoroughly cleaned to remove any unnecessary material, including dirt and debris. The seeds were then separated after the peel and pulp were carefully removed. To improve their flavor and aroma qualities, the seeds were dried in a tray drier for 12 hours at 50 ± 1 °C, following the procedure outlined by Kanbargi et al.5 After drying, the seeds were ground with a commercial mixer grinder (Bajaj Electrical and Electronics Pvt. Ltd, Rex 500, Punjab, India) to obtain a fine powder. The resulting seed powder and other powder ingredients were sieved through a 75 µm British sieve, as shown in SI Fig. 1. The Ziziphus jujuba seed powder was stored at 4 °C and sealed in zip-lock laminate packaging materials (Well-worth Packers Pvt. Ltd, Delhi, India) until analysis and processing.2.2. Experimental design
The experimental design for the development of multigrain waffles incorporated with Ziziphus jujuba seed flour was established using a D-optimal mixture design. This statistical approach was selected for its efficiency in evaluating multiple formulation components and their interactions while minimizing the number of experimental runs. Four ingredients, including jowar flour, bajra flour, wheat flour, and Ziziphus jujuba seed flour, were used as independent variables within predefined ranges. To ensure a light texture, bajra flour, jowar flour, wheat flour, and corn starch were sieved to remove lumps. Modifications were made using the preparation method described by Antonic et al.6 to optimize the waffles. The proportions of jowar and bajra flours ranged from 25–30%, wheat flour from 40–50%, and seed flour from 0–10%. Based on these constraints, the D-optimal mixture design generated a total of 24 experimental formulations. The complete composition and detailed description of all waffle formulations are presented in Table 3, which lists the exact percentage of each ingredient used in every treatment. This table provides a clear and comprehensive overview of the experimental samples, ensuring transparency and reproducibility of the formulation design. The waffles were cooked for 15 to 20 minutes, until they were crisp and golden brown (SI Fig. 1). Each formulation was prepared according to standardized processing conditions and evaluated for biochemical, antioxidant, and sensory responses. The data obtained from these formulations were used to identify the optimal combination based on overall desirability and performance criteria.2.3. Nutritional composition analysis
The proximate composition of the samples was assessed by analyzing their protein content,7,8 fat content,9 crude fiber content,10 moisture content,11 ash content,10 and carbohydrate content.122.4. Biochemical and antioxidant activity determination
To detect the biochemical compounds and antioxidant activities in a sample, 500 mg of the sample was weighed and subsequently blended with 100 mL of 80% ethanol. Following centrifugation of the extract at 6000×g for 15 min, the ethanol-based supernatant was stored in glass vials at −20 °C before quantification.13 The total phenolic content of the samples was determined according to a previously described method.14 To determine the total flavonoid content of the samples, the colorimetric method was used as described by Orsavová et al.15 The ascorbic acid content of each sample was calculated by the method of Singh et al.14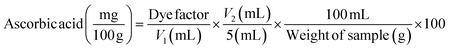 | (1) |
The radical scavenging potential of each sample was evaluated by the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay, following the procedure outlined by Rajaei et al.16 Briefly, ethanoic extracts (0.1 mL) were mixed with a DPPH solution (2.9 mL), and the absorbance of the resulting solution was measured at 517 nm using a UV-vis spectrophotometer (LB-925, Labcare, Mumbai, India) after a 30 min incubation period. The percentage of inhibition of DPPH radical scavenging activity was determined by eqn (2):
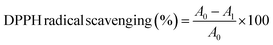 | (2) |
2.5. Determination of antinutrients
The tannin content and phytic acid content estimation procedure followed the methods described by Nwakife et al.,10 and the saponin content was calculated according to Arjmand et al.172.6. Techno-functional properties
Water absorption and solubility indices were determined through the methods of Budiman et al.18 The oil absorption capacity was calculated via the procedure described by Kakar et al.,19 the swelling power and solubility were calculated by the procedure described by Timothy and Abigail,20 the dispersibility of the sample was calculated through the procedure described by Singh et al.,14 the emulsion capacity and stability of emulsions were determined by the method described by Geng et al.,21 the foaming capacity and stability, bulk density, and tapped density were calculated by the method described by Aro et al.,22 and the Hausner ratio and Carr's index were analyzed via the method described by Singh et al.142.7. Characterization of Ziziphus jujuba seeds
2.8. Sensory evaluation
Sensory evaluation was carried out for the different concentrations of the sample in comparison to the control through a questionnaire with a hedonic scale for each parameter, as described by Ahmed et al.24 The organoleptic analysis of the prepared waffles was carried out by 20 semitrained panellists (10 females and 10 males) from Lovely Professional University, Phagwara. A 9-point hedonic scale ranging from the lowest 1 (dislike extremely) to the highest 9 (like extremely) was used. The evaluation was performed on the basis of appearance, texture, smell, taste, and overall acceptability parameters.24,252.9. Principal component analysis (PCA)
To determine the interrelationship between the antioxidant activity (TPC, TFC, and DPPH) and sensory parameters (appearance & colour, texture, flavour & sweetness, and overall acceptability) of the optimised jujuba-based waffles, principal component analysis (PCA) was performed through OriginPro.26,272.10. Microbial growth determination
The microbial growth analysis of the prepared product followed the methods described by Siddique et al.28 and Asadi et al.29 A 1 g sample was aseptically mixed with 9 mL of sterile distilled water and homogenized. Serial dilutions were prepared, and 1 mL from each dilution was plated. Nutrient agar medium was used for total plate count determination, potato dextrose agar was used for yeast and mold counts, and MacConkey agar was used for coliform count determination. All the plates were incubated at different temperatures for total plate counts and coliform counts (37 °C for 24–48 hours) or for yeast and mold (25–30 °C for 24–48 hours). Colonies were subsequently counted on plates with 30–300 colonies, and the results are expressed as colony-forming units per gram (cfu g−1).2.11. Shelf-life study
The shelf-life of the Ziziphus jujuba seed-based waffles was evaluated under controlled storage conditions to assess the effect of temperature and packaging material on product stability. Freshly prepared waffles were packed separately in low-density polyethylene (LDPE) and metallic polyester (MP) packaging materials and sealed to prevent external contamination. The packaged samples were stored at three temperatures: 0 °C, 25 °C, and 37 °C. At predetermined storage intervals, the samples were analyzed for changes in biochemical parameters and microbial quality to monitor quality deterioration over time.2.12. Data analysis
The data collected from the design experiments were subjected to ANOVA for each experiment, and all the experiments were carried out in triplicate. The data were statistically analyzed via a Tukey post hoc test (P < 0.05), and the statistical software SPSS V. 22 (SPSS Inc., Chicago, IL, USA) was used to analyze the experimental data. The collected data are presented as the mean ± standard deviation, taking into account the indicated significant (>0.05) p values. Moreover, to study the impact of various parameters on treatments generated by Design Expert, the data were analysed using OriginPro for Principal Component Analysis.3 Results and discussion
3.1. Nutritional and antioxidant properties of Ziziphus jujuba seeds
The moisture content of the Ziziphus jujuba seed flour was determined, and the estimated value was 2.66 ± 0.25% (Table 1). On the other hand, Ziziphus lotus seeds have a moisture content of 6.05%.30 The reported moisture content was higher than that of the present study. The estimated value denotes the equilibrium moisture in the environment. The dried seed powder had a reduced moisture content because of its larger surface area, reduced particle size, and removal of free water present in flour particles. The varying values of moisture content indicate that seeds of Ziziphus jujuba typically have a low moisture content, which is advantageous for both storage and consumption. The rich nutritional composition and possible therapeutic effects of these seeds make them suitable for a variety of uses, such as nutritional and mineral supplements, and their low moisture content contributes to their suitability.| Nutritional and functional properties | Ziziphus jujuba seed composition |
|---|---|
| a The obtained results are represented as mean ± standard deviation values (n = 3). | |
| Moisture content (%) | 2.66 ± 0.25 |
| Ash content (%) | 3.38 ± 0.28 |
| Carbohydrate content (%) | 49.18 ± 0.08 |
| Protein content (%) | 30.98 ± 0.21 |
| Fat content (%) | 3.26 ± 0.11 |
| Fiber content (%) | 10.54 ± 0.38 |
| Ascorbic acid (mg per 100 g) | 46.91 ± 0.87 |
| Total phenolic content (mg GAE per g) | 73.66 ± 0.40 |
| Total flavonoid content (mg QE per g) | 118.61 ± 0.63 |
| DPPH activity (%) | 58.09 ± 0.61 |
| Tannins (%) | 7.06 ± 0.12 |
| Phytates (mg per 100 g) | 290.34 ± 0.23 |
| Saponins (mg per 100 g) | 6.04 ± 0.28 |
The ash content of the moisture-free Ziziphus jujuba seed flour was analyzed, and the value obtained was 3.38 ± 0.28% (Table 1). Ziziphus jujuba seeds contain an ash concentration of 7.2%, according to research by Fukalova et al.,31 whereas Yerima & Adamu9 reported that the ash percentage for Ziziphus jujuba seeds collected from different locations in Nigeria was 2.79%, and as observed by Adilah et al.,32 it was 3.62%. The ash content analysis of the above mentioned studies was in agreement with the estimated value of ash content obtained in this study. The findings of these studies on the ash content of jujuba seed flour are relatively similar to our findings.
The fat content of the jujuba seed flour was estimated, and the value obtained from the triplicate analysis was 3.26 ± 0.11% (Table 1). Studies conducted by Fukalova et al.31 support this statement, and the determined fat content was 8.45%. Another study conducted by Snoussi et al.33 reported fat contents ranging from 20.13% to 24.57%, which was significantly different from the results reported. Furthermore, the location and method of cultivation of Ziziphus jujuba seeds might affect their fatty acid profile, resulting in differences in fat content among various production zones. Overall, the fat content of Ziziphus jujuba seeds makes them an excellent source of necessary fatty acids with certain nutritional advantages.
Fiber content estimation was performed, and the estimated value was 10.54 ± 0.38% (Table 1). A study on Ziziphus mauritiana seeds reported a fiber content of 11.04 ± 0.88% in the samples.9 This study supports the fiber estimation of our findings and is in agreement with the compositional value. These results highlight the significant amount of fiber found in Ziziphus seeds, highlighting their nutritional value and possible use as industrial raw materials and in food formulations.
The protein content of the jujuba seed flour was determined by the Kjeldahl method, and the estimated value was 30.98 ± 0.21% (Table 1). Additionally, a study on Ziziphus mauritiana seeds reported a crude protein content of 36.10 mg per 100 g of sample.9 Another study conducted by Fukalova et al.31 reported crude protein contents of 11.56% and 19.11% in Ziziphus lotus seeds, which were significantly different from the results reported. The difference was due to the species' genotype, growth, and environmental conditions, such as rainfall, day duration, and light intensity. All of these results indicate the high amount of protein found in Ziziphus jujuba seeds, which makes them an excellent source of this important nutrient that may be used for a variety of purposes, including as a supplement to diets.
The carbohydrate content was obtained by subtracting the proximal values of moisture, ash, protein, fat, and fiber from 100. The carbohydrate content of the jujuba seed flour obtained was 49.18 ± 0.08% (Table 1). Yerima & Adamu9 reported that the carbohydrate content of Ziziphus jujuba seeds collected from different locations in Nigeria was 21.26 ± 0.63 mg/100 g. Ziziphus oenopolia seeds contain high carbohydrate contents of 42.96%,34 whereas the carbohydrate content is 35.25% in Ziziphus spina Christi seeds.35 These estimated values are in disagreement with our findings concerning the carbohydrate composition of jujuba seed flour, which may be due to differences in species and genotype. The carbohydrate content indicates the starch and sugar contents, such as those of amylose and amylopectin. It acts as an energy-providing source to meet the desired dietary requirements.
The total phenolic content of the jujuba seed flour was analyzed to determine the quantitative composition of the phenolic components responsible for its antioxidative properties. The total phenolic content estimated in this study was approximately 73.66 ± 0.40 mg GAE per g (Table 1). Phenolic substances are bioactive metabolites found in fruits and vegetables that influence both the functional properties, including anticancer, antioxidant, and antidiabetic effects, and the sensory qualities, such as taste, flavor, and color, of food materials.36Ziziphus spina-christi seeds presented a total phenolic content ranging from 4.84 to 49.58 mg g−1.37 The difference in the total phenolic content was due to the distinct genotypes, types of extraction methods, extraction times, extraction temperatures, and concentrations of the sample seed extracts. In comparison, our findings revealed a greater total phenolic content in the ethanolic seed extract, which indicates that jujuba seed flour is composed of components responsible for antioxidant activity, which is a key factor for the pharmacological importance of incorporating jujuba flour into the daily diet.
The total flavonoid content of the jujuba seed flour was analyzed to determine the quantitative composition of the phenolic components responsible for its antioxidative properties. The total flavonoid content estimated in this study was approximately 118.61 ± 0.63 mg QE per g (Table 1). Ziziphus lotus L. seeds had flavonoid contents of 69.19 ± 0.10 mg g−1 in the ethanolic extract and 53.13 ± 0.55 mg g−1 in the methanolic extract.37 Our findings revealed a relatively high total flavonoid content in the ethanolic seed extract, which indicates that jujuba seed flour is composed of components responsible for antioxidant activity, which is a key factor for the pharmacological importance of incorporating jujuba flour into the daily diet.
The ascorbic acid content of the jujuba seeds was found to be 46.913 ± 0.87 mg per 100 g (Table 1). Z. jujuba was analyzed for ascorbic acid content, and the value was approximately 4.752 mg of vitamin C per 1 g of seed.38 This study was consistent with the values obtained in our study. The ascorbic acid content of the Ziziphus mauritiana seeds of the Dil-Bahar variety was the highest at 51.69 ± 0.05 mg per 100 g. These findings contrast with our results, possibly because of genotypic changes or differences in geographic factors.39 The estimated ascorbic acid content indicates that jujuba seeds possess potent antioxidant capacity, capable of neutralizing free radicals.
The DPPH activity of the jujuba seeds was analyzed for their antioxidant potential, and the % inhibition was 58.09 ± 0.61% (Table 1). The antioxidant potential of DPPH was 4.53 µg mL−1 according to Akanda & Hasan.40 A DPPH assay of Z. jujuba seed-derived protein hydrolysate revealed that the obtained value was approximately 8.36%.5 These findings contrast with our study, in which the highest antioxidant potential was achieved due to higher phenolic and flavonoid contents.
3.2. Antinutritional properties of Ziziphus jujuba seeds
The tannin content of the jujuba seed flour was analyzed, and the value obtained was 7.06% (Table 1). The tannin content in Ziziphus seeds varies among different species. Research on Ziziphus lotus L. seeds revealed that the methanol extract had a relatively high tannin content of 9.12 ± 1.07 mg g−1, whereas the ethanol and aqueous extracts had tannin contents of 4.97 ± 0.95 mg g−1 and 1.88 ± 0.47 mg g−1, respectively.41 This comparison demonstrates differences in tannin concentrations across species and extraction techniques, highlighting the importance of selecting an appropriate extraction solvent in influencing tannin results. Tannins are known to inhibit the activities of enzymes such as trypsin, amylase, and lipase, and interfere with the absorption of dietary iron. However, high contents may lead to calcium and iron deficiencies in the body and often cause osteoporosis and anemia. Tannins in jujuba seeds are beneficial because of their antioxidant, anti-inflammatory, and antimicrobial properties, which help neutralize harmful free radicals, reduce inflammation, and protect against bacterial infections.42 Additionally, tannins support digestive health by improving gut integrity and protecting against diarrhea. These findings highlight the presence of tannins in Ziziphus seeds, suggesting their potential health benefits and supporting their traditional medicinal uses; however, tannins also add nutritional value when consumed in moderation, enhancing overall health benefits.43 This balance between beneficial effects and potential drawbacks further emphasizes the importance of dosage and processing techniques when incorporating tannin-rich foods into the diet.The concentration of phytate used for Ziziphus jujuba seeds was 290.34 mg per 100 g (Table 1). Research on Ziziphus lotus L. seeds revealed a high content of phytates (8764.63 ± 0.16 mg per 100 g).10 The concentration of phytate in the seeds was greater than that in the pulp. The negative effect of the presence of phytate on mineral uptake is of major concern. Phytate is reported to interact with carbohydrates, which limits their bioavailability and digestion. Phytic acid is a well-known antinutrient that can bind to essential minerals such as iron, zinc, and calcium, inhibiting their absorption in the digestive system. High levels of phytic acid in foods can reduce the bioavailability of these vital minerals, potentially leading to nutrient deficiencies in individuals who rely heavily on seed flour as a dietary staple.
The saponin concentration of Ziziphus jujuba seeds was 6.04 mg/100 g (Table 1). Research on Ziziphus jujuba seeds revealed high saponin contents ranging from 52.10 to 75.20%.44 Differences in saponin content were observed due to differences in genotype, type of extraction method, time, temperature of extraction, and concentration of the sample seed extract. Furthermore, research on Ziziphus jujuba Mill. fruit identified saponins such as Ziziphus saponin III, jujuboside B, and jujuboside A, indicating that a diverse range of saponins are present in different parts of the plant. Saponins extracted from Ziziphus jujuba seeds have been found to possess antioxidant properties, increasing resistance to oxidative damage and increasing antioxidant enzyme activities. Additionally, saponins from Ziziphus jujuba seeds have shown hypoglycemic effects by inhibiting digestive enzymes, improving glucose consumption, and activating key proteins involved in energy metabolism and insulin resistance, thus potentially assisting in the management of hyperglycemia and related symptoms. Furthermore, saponins from Ziziphus seeds have been reported to exhibit anti-inflammatory, antiulcer, and anticancer activities, highlighting their diverse pharmacological potential.
3.3. Techno-functional properties
The water absorption capacity of the jujuba seed flour was examined, and the value was 134.66 ± 0.74% (Table 2). A higher water absorption capacity indicates that flour can entrap larger amounts of water molecules, which is favorable for preparing slurries from instant mixture formulations.45 The water absorption index (WAI) of the dried powder of jujuba seed flour was analyzed, and the WAI was 3.65 ± 0.02 g g−1 (Table 2). The WAI indicates that flour contains hydrophilic components such as starch and protein, which are responsible for swelling and gelatinizing properties.46 These findings indicate that jujuba seed flour has good starch gelatinization properties, which are important for rehydration and cooking in instant mixture formulations. The oil absorption capacity (OAC) of the powder was analyzed, and the value was 2.39 ± 0.80 g g−1 (Table 2). This property indicates that the sensory characteristics of flour particles increase the OAC, providing an enhanced mouth feel and palatability. The reason behind the high oil absorption capacity was found to be that amylose can freely disperse in an aqueous solution.14 The highest swelling power was observed because of the increased molecular mobility in the amorphous region, resulting in weaker binding forces. The lowest solubility was found because of the lower amylose and higher amylopectin contents. Dispersibility was determined for the powder particles of jujuba seed flour and was 65.46 ± 0.39% (Table 2). The higher dispersibility ratio implies that flour particles have a high rehydration ratio, indicating that they reconstitute well for the preparation of batter. It also correlates with the protein content of flour particles. The dispersibility value was greater because of the higher protein content of jujuba seed flour.47| Techno-functional properties | Jujuba seed flour composition |
|---|---|
| a The obtained results are represented as mean ± standard deviation values (n = 3). | |
| Water absorption capacity (%) | 134.66 ± 0.74 |
| Water absorption index (g g−1) | 3.65 ± 0.02 |
| Oil absorption capacity (g g−1) | 2.39 ± 0.80 |
| Swelling power (g g−1) | 16.95 ± 0.39 |
| Solubility (%) | 7.66 ± 0.34 |
| Dispersibility (%) | 65.46 ± 0.39 |
| Emulsion capacity (%) | 70.36 ± 0.61 |
| Stability of emulsions (%) | 54.75 ± 0.41 |
| Foam capacity (%) | 73.66 ± 0.95 |
| Bulk density (g ml−1) | 0.780 ± 0.04 |
| Tapped density (g ml−1) | 0.895 ± 0.02 |
| Hausner ratio | 1.375 ± 0.03 |
| Carr's index | 29.73 ± 0.10 |
The emulsion capacity and stability of the Jujuba seed flour samples were analyzed, and the values were 70.36 ± 0.61 and 54.75 ± 0.41%, respectively (Table 2). High emulsion activity was correlated with increased total protein content, and the two major protein fractions, albumin and globulin, are responsible for emulsion formation. These two protein fractions can migrate through the oil–water interface, resulting in emulsification.48 The foam capacity and stability of the jujuba seed flour were 73.66 ± 0.95 and 57.30 ± 0.53%, respectively (Table 2). The foam capacity and stability indicate that the protein present in jujuba seed flour can strongly interact with aqueous solutions. This leads to strong interactions at the air–water interface, and the protein structure enables cohesive foam film formation with increased strength and stability. At 10 min, it showed the highest stability; as time increased, the bubble collapsed, and the foam stability decreased.14
The bulk density of the jujuba seed flour was analyzed, and the value obtained was 0.780 ± 0.04 g mL−1 (Table 2). The bulk density indicates the matrix composition of flour and its flow properties during packaging. In contrast, a lower bulk density results in better mixing and cooking properties in the preparation of instant-mixed food formulations because of their loose-flowing properties.49 The tap density of the jujuba seed flour was analyzed, and the estimated value was 0.895 ± 0.02 g mL−1 (Table 2). This property shows that jujuba seed flour particles have better compressibility and density due to their finer particle size and lower aerated density.50 The Hausner ratio and Carr's index of the jujuba seed flour were found to be 1.375 ± 0.036 and 29.73 ± 0.10, respectively (Table 2). These results indicate that jujube seed flour has moderate flow characteristics and compressibility, both of which are essential for a variety of uses, including food preparation.51
3.4. Characterization of Ziziphus jujuba seeds
Using scanning electron microscopy (SEM), we observed the morphology of the sample. The SEM images reveal that the structures are predominantly spherical in shape. An example of such a structure is shown in Fig. 1. The SEM parameters for this image are a scale bar representing 10 µm, a magnification of 500×, an accelerating voltage of 20.0 kV, and a working distance of 10 mm. These settings were chosen to optimize the resolution and clarity of the spherical structures. A high accelerating voltage of 20.0 kV was utilized to ensure sufficient electron penetration and detailed imaging of the sample surface. The chosen working distance of 10 mm provided an optimal depth of field, allowing for clear visualization of the spherical morphology across the sample. The magnification level of ×500 allowed to observe the microscale features of the structures effectively, with the scale bar indicating that the observed structures were within the 10 µm range.
A study on a Ziziphus jujube waste seed-derived biomass adsorbent (ZYB) by Fakhar et al.52 presented SEM images, which revealed a porous and uneven surface morphology with a range of particle sizes. The surface of ZYB featured pores and cracks, which suggested that an adequate surface area might be used to adsorb Cd2+ ions from aqueous solutions. The irregularities and pores present at the surface enhance the texture and increase the crispiness of the waffles, as studied by Kelley et al.53
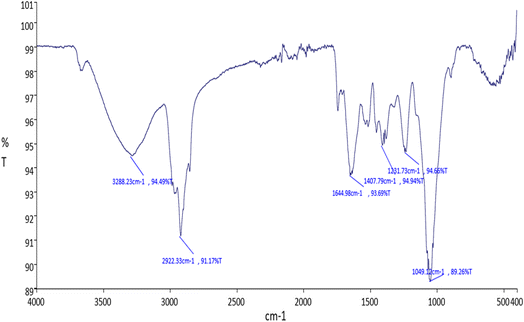
The data point at 2922.33 cm−1 with 91.17% transmittance on the FTIR graph indicated moderate absorption, likely due to the C–H stretching vibrations of alkanes. The transmittance value indicated that the absorption by this functional group was noticeable but not very strong, suggesting a moderate presence of methylene (CH2) or methyl (CH3) groups in the sample.
The data point at 1644.98 cm−1 with 93.69% transmittance on the FTIR spectrum showed weak to moderate absorption, likely due to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations in alkenes or C
C stretching vibrations in alkenes or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations in amides. The transmittance value indicated that the absorption by this functional group was relatively weak, suggesting a low to moderate presence of the respective functional group in the sample.
O stretching vibrations in amides. The transmittance value indicated that the absorption by this functional group was relatively weak, suggesting a low to moderate presence of the respective functional group in the sample.
The data point at 1407.79 cm−1 with 94.94% transmittance on the FTIR graph suggested weak absorption, likely due to C–H bending (deformation) vibrations in alkanes, bending vibrations in methyl (CH3) and methylene (CH2) groups, or possibly C–O stretching in phenols and some carboxylic acids. The high transmittance value indicated that the absorption by this functional group was relatively weak, suggesting a low presence of the respective functional group in the sample.
The data point at 1231.73 cm−1 with 94.66% transmittance on the FTIR graph demonstrated weak absorption, likely due to C–N stretching vibrations in amines; C–O stretching vibrations in alcohols, ethers, carboxylic acids, or esters; or C–H bending vibrations in aromatic compounds. The high transmittance value indicated that the absorption by this functional group was relatively weak, suggesting a low presence of the respective functional group in the sample.
The data point at 1049.12 cm−1 with 89.26% transmittance on the FTIR graph represents moderate absorption, likely due to C–O stretching vibrations in alcohols, ethers, and esters or Si–O stretching vibrations in silicates and silicones. The transmittance value indicated that the absorption by this functional group was noticeable but not very strong, suggesting a moderate presence of the respective functional group in the sample. Similarly, Ziziphus oenopolia seed extract was analyzed via FTIR, confirming the presence of various compounds, such as alcohols, alkanes, and carboxylic acids, indicating potential pharmacological activities (Fig. 2).
3.5. Model fitting and adequacy of experimental data
The optimization of jujube seed flour-based waffles was performed after all the interactions between variables and responses were analyzed. All four components, namely jowar flour, bajra flour, wheat flour, and jujuba seed flour, were set within a particular range, as shown in Table 3, and accordingly, 24 treatments were suggested by the D-optimal design. The responses, such as total phenolic content, total flavonoid content, DPPH, appearance and color, texture, flavor and sweetness, and overall acceptability, were considered when finalizing the optimized proportions for the preparation of the waffles (Tables 3 and 4).| Run | Jowar flour (%) | Bajra flour (%) | Wheat flour (%) | Jujuba seed powder (%) | Total flavonoid content (mg QE per g) | Total phenolic content (mg GAE per g) | DPPH (%) |
|---|---|---|---|---|---|---|---|
| a Data are expressed as mean ± standard deviation (n = 3), and values with different lowercase letters in the same column are significantly different (Tukey, p < 05). | |||||||
| W1 | 30.00 | 25.00 | 45.00 | 0.00 | 33.22 ± 0.36j | 15.70 ± 0.06k | 5.06 ± 0.06n |
| W2 | 27.79 | 25.00 | 43.70 | 3.51 | 38.39 ± 0.14f | 17.75 ± 0.24h | 12.60 ± 0.24h |
| W3 | 30.00 | 30.00 | 40.00 | 0.00 | 33.2 ± 0.03j | 15.62 ± 0.08k | 3.16 ± 0.01o |
| W4 | 25.00 | 25.00 | 42.55 | 7.45 | 44.18 ± 0.02c | 24.44 ± 0.02c | 21.26 ± 0.09b |
| W5 | 27.08 | 26.45 | 45.42 | 1.05 | 34.52 ± 0.08i | 18.04 ± 0.04g,h | 8.55 ± 0.09k |
| W6 | 27.79 | 25.00 | 43.70 | 3.51 | 38.39 ± 0.04f,g | 17.76 ± 0.04h | 12.58 ± 0.03h |
| W7 | 25.00 | 27.37 | 47.63 | 0.00 | 32.76 ± 0.04j | 17.41 ± 0.08i | 8.16 ± 0.02k |
| W8 | 25.00 | 28.35 | 43.26 | 3.39 | 37.89 ± 0.04g | 18.13 ± 0.03g | 13.30 ± 0.05g |
| W9 | 25.00 | 30.00 | 45.00 | 0.00 | 32.65 ± 0.02j | 14.80 ± 0.08l | 7.12 ± 0.02m |
| W10 | 27.30 | 25.01 | 40.00 | 7.69 | 44.76 ± 0.01b | 25.71 ± 0.01b | 19.82 ± 0.02c |
| W11 | 25.00 | 27.59 | 40.00 | 7.41 | 44.12 ± 0.08b,c | 25.44 ± 0.02b | 20.12 ± 0.02c |
| W12 | 25.00 | 28.35 | 43.26 | 3.39 | 37.89 ± 0.02g | 18.09 ± 0.02g,h | 13.29 ± 0.02g |
| W13 | 27.90 | 28.81 | 43.29 | 0.00 | 32.82 ± 0.01j | 18.03 ± 0.01g,h | 5.27 ± 0.01n |
| W14 | 25.00 | 25.00 | 50.00 | 0.00 | 32.75 ± 0.01j | 14.64 ± 0.02l | 9.10 ± 0.02j |
| W15 | 25.00 | 25.00 | 40.00 | 10.00 | 48.17 ± 0.01a | 32.39 ± 0.04a | 25.43 ± 0.01a |
| W16 | 27.85 | 28.71 | 40.00 | 3.44 | 38.22 ± 0.08f,g | 18.67 ± 0.02e,f | 11.01 ± 0.02i |
| W17 | 25.00 | 25.00 | 47.54 | 2.46 | 36.45 ± 0.08h | 15.63 ± 0.08k | 13.10 ± 0.01g |
| W18 | 29.61 | 25.00 | 40.00 | 5.39 | 41.38 ± 0.08d | 20.03 ± 0.01d | 14.25 ± 0.02f |
| W19 | 25.00 | 25.00 | 44.95 | 5.05 | 40.57 ± 0.08e | 19.07 ± 0.08e | 17.32 ± 0.02d |
| W20 | 30.00 | 30.00 | 40.00 | 0.00 | 33.11 ± 0.04j | 15.64 ± 0.01k | 3.17 ± 0.02o |
| W21 | 27.85 | 28.71 | 40.00 | 3.44 | 38.20 ± 0.01f,g | 18.67 ± 0.08f | 11.02 ± 0.01i |
| W22 | 30.00 | 26.50 | 41.55 | 1.95 | 36.10 ± 0.01h | 17.95 ± 0.01g,h | 7.68 ± 0.02l |
| W23 | 30.00 | 25.00 | 45.00 | 0.00 | 33.20 ± 0.02j | 15.70 ± 0.03k | 5.12 ± 0.04n |
| W24 | 25.00 | 30.00 | 40.00 | 5.00 | 40.50 ± 0.01e | 16.98 ± 0.04j | 15.33 ± 0.02e |
Table 1 presents the experimental data on the quality characteristics of the jujuba seed waffles, including the total flavonoid content (mg GAE per g), total phenolic content (mg GAE per g), antioxidant activity (DPPH (%), appearance and color, texture, flavor, and sweetness, and overall acceptability. These characteristics were analyzed via analysis of variance (ANOVA), coefficient of determination (R2), standard deviation, mean, and coefficient of variation (C.V. %) with Design Expert software, version 13. Special quartic model equations were employed to evaluate the strength of the experimental data. Eqn (3)–(9) illustrate the model equations for the total flavonoid content (mg GAE per g), total phenolic content (mg GAE per g), DPPH (%), appearance and color, texture, flavor and sweetness, and overall acceptability of the jujuba seed waffles (Fig. 4).
| TFC = 33.52 × A + 32.67 × B + 32.75 × C + 48.15 × D + 0.2256 × AB + 0.4775 × AC − 0.1263 × AD − 0.1125 × BC + 0.1629 × BD − 0.1235 × CD − 4.39 × ABC − 1.67 × ABD + 0.2817 × ACD − 1.08 × BCD | (3) |
| TPC = 10.43 × A − 6.60 × B + 14.64 × C + 32.39 × D + 54.86 × AB + 12.66 × AC − 9.08 × AD + 43.15 × BC + 16.34 × BD − 18.12 × CD + 0.5674 × ABC − 0.0073 × ABD − 0.0969 × ACD + 0.2283 × BCD | (4) |
| DPPH = 1.11 × A + 5.56 × B + 9.11 × C + 25.42 × D − 0.6797 × AB − 0.0826 × AC + 0.0780 × AD − 0.7675 × BC − 0.7201 × BD − 0.0871 × CD + 0.1457 × ABC + 1.11 × ABD + 0.0924 × ACD + 0.6270 × BCD | (5) |
| Appearance and colour = 5.92 × A + 9.80 × B + 6.12 × C + 8.58 × D − 10.03 × AB − 2.72 × AC + 3.62 × AD − 9.48 × BC − 2.88 × BD + 4.87 × CD + 5.43 × ABC − 3.14 × ABD + 1.91 × ACD + 9.26 × BCD | (6) |
| Run | Jowar flour (%) | Bajra flour (%) | Wheat flour (%) | Jujuba seed powder (%) | Appearance and colour | Texture | Flavour and sweetness | Overall acceptability |
|---|---|---|---|---|---|---|---|---|
| a Data are expressed as mean ± standard deviation (n = 3), and values with different lowercase letters in the same column are significantly different (Tukey, p < 05). | ||||||||
| W1 | 30.00 | 25.00 | 45.00 | 0.00 | 5.63 ± 0.11b,c | 5.83 ± 0.28h,i | 5.19 ± 0.06j,k | 5.62 ± 0.11h,i,j |
| W2 | 27.79 | 25.00 | 43.70 | 3.51 | 7.97 ± 0.26c,d | 7.04 ± 0.17f | 7.82 ± 0.04d | 7.30 ± 0.08e,g |
| W3 | 30.00 | 30.00 | 40.00 | 0.00 | 5.15 ± 0.01g | 5.21 ± 0.04i | 5.89 ± 0.05h | 5.87 ± 0.02h,i,j |
| W4 | 25.00 | 25.00 | 42.55 | 7.45 | 8.83 ± 0.20a,b | 8.85 ± 0.03a | 8.04 ± 0.01c,d | 8.44 ± 0.02a |
| W5 | 27.08 | 26.45 | 45.42 | 1.05 | 5.50 ± 0.03f,g | 5.13 ± 0.08i | 5.22 ± 0.02j,k | 5.77 ± 0.02i,j |
| W6 | 27.79 | 25.00 | 43.70 | 3.51 | 7.67 ± 0.02c,d | 7.50 ± 0.04e,f | 7.26 ± 0.03f | 7.31 ± 0.08e,f |
| W7 | 25.00 | 27.37 | 47.63 | 0.00 | 5.60 ± 0.06b,c | 5.45 ± 0.03h,i | 5.32 ± 0.08j,k | 5.48 ± 0.008l |
| W8 | 25.00 | 28.35 | 43.26 | 3.39 | 7.59 ± 0.01c,d | 7.86 ± 0.02c,d,e | 7.41 ± 0.03e | 7.52 ± 0.08c,d |
| W9 | 25.00 | 30.00 | 45.00 | 0.00 | 5.33 ± 0.01f,g | 5.40 ± 0.02h,i | 5.12 ± 0.04k,l | 5.91 ± 0.03h |
| W10 | 27.30 | 25.01 | 40.00 | 7.69 | 8.45 ± 0.03a,b | 8.15 ± 0.01b,c,d | 8.27 ± 0.04g | 8.30 ± 0.01a |
| W11 | 25.00 | 27.59 | 40.00 | 7.41 | 8.17 ± 0.01a,b,c | 8.24 ± 0.02b,c | 7.98 ± 0.02c,d | 8.40 ± 0.02a |
| W12 | 25.00 | 28.35 | 43.26 | 3.39 | 7.95 ± 0.08b,c | 7.70 ± 0.04d,e,f | 8.04 ± 0.01c,d | 7.50 ± 0.02c |
| W13 | 27.90 | 28.81 | 43.29 | 0.00 | 5.39 ± 0.04f,g | 5.35 ± 0.12h,i | 5.15 ± 0.04j,k,l | 5.19 ± 0.04l |
| W14 | 25.00 | 25.00 | 50.00 | 0.00 | 5.95 ± 0.01e,f | 5.74 ± 0.12h | 5.52 ± 0.01i | 5.68 ± 0.08j,k |
| W15 | 25.00 | 25.00 | 40.00 | 10.00 | 8.74 ± 0.02a | 8.62 ± 0.02a,b | 8.67 ± 0.01a | 8.24 ± 0.02a,b |
| W16 | 27.85 | 28.71 | 40.00 | 3.44 | 7.11 ± 0.03d | 7.29 ± 0.01f | 7.05 ± 0.01f | 7.33 ± 0.01d,e |
| W17 | 25.00 | 25.00 | 47.54 | 2.46 | 7.90 ± 0.01b,c | 7.71 ± 0.03d,e,f | 8.02 ± 0.04c,d | 7.10 ± 0.08f |
| W18 | 29.61 | 25.00 | 40.00 | 5.39 | 8.34 ± 0.01a,b | 8.55 ± 0.04a,b | 8.25 ± 0.01b | 8.39 ± 0.01a |
| W19 | 25.00 | 25.00 | 44.95 | 5.05 | 8.36 ± 0.04a,b | 8.57 ± 0.02a,b | 8.27 ± 0.08b | 8.07 ± 0.08b |
| W20 | 30.00 | 30.00 | 40.00 | 0.00 | 5.50 ± 0.01f,g | 5.72 ± 0.28h | 4.99 ± 0.02l | 5.9 ± 0.04h,i |
| W21 | 27.85 | 28.71 | 40.00 | 3.44 | 7.18 ± 0.08d | 7.34 ± 0.04e,f | 7.50 ± 0.01e | 7.33 ± 0.02c,d,e |
| W22 | 30.00 | 26.50 | 41.55 | 1.95 | 6.20 ± 0.01e | 6.36 ± 0.08g | 6.63 ± 0.08g | 6.77 ± 0.01g |
| W23 | 30.00 | 25.00 | 45.00 | 0.00 | 5.13 ± 0.28f,g | 5.82 ± 0.08h | 5.31 ± 0.02i,j | 5.63 ± 0.01j,k |
| W24 | 25.00 | 30.00 | 40.00 | 5.00 | 8.53 ± 0.01a,b | 8.68 ± 0.01a,b | 8.15 ± 0.01b,c | 8.48 ± 0.02a |
| Texture = 9.08 × A + 10.37 × B + 5.84 × C + 8.46 × D − 16.91 × AB − 6.71 × AC − 1.30 × AD − 9.79 × BC − 3.02 × BD + 5.99 × CD + 7.01 × ABC + 5.51 × ABD − 13.44 × ACD + 5.68 × BCD | (7) |
| Flavour and sweetness = 8.27 × A + 9.06 × B + 5.86 × C + 8.33 × D − 12.76 × AB − 7.42 × AC − 0.1397 × AD − 8.57 × BC − 2.27 × BD + 4.89 × CD + 8.02 × ABC + 8.32 × ABD + 4.91 × ACD + 14.65 × BCD | (8) |
| Overall acceptability = 8.66 × A + 8.57 × B + 5.67 × C + 8.25 × D − 10.92 × AB − 6.17 × AC − 0.2089 × AD − 4.84 × BC + 0.3179 × BD + 4.40 × CD − 1.72 × ABC − 0.6618 × ABD − 0.5864 × ACD − 0.0392 × BCD | (9) |
3.6. Constraints
Jowar and bajra flour were kept in the range of 25% as the minimum range and 30% as the maximum range, with upper and lower weights of 1 and an importance of 3. Wheat flour was in the range of 40% as a minimum and 50% as the maximum range, with upper and lower weights of 1 and an importance of 3. Jujuba seed powder was in the range of 0% as the minimum and 10% as the maximum range, with upper and lower weights of 1 and an importance of 3. The total flavonoid content was in the range of 32.65%, which was the minimum, and 48.17%, which was the maximum range, with an upper and lower weight of 1 and an importance of 3. The total phenolic content was in the range of 14.64%, which was the minimum, and 32.39%, which was the maximum range, with an upper and lower weight of 1 and an importance of 3. DPPH was maximized by 3.16% as the minimum and 25.43% as the maximum range, with an upper and lower weight of 1 and an importance of 5. The appearance and color ranged from 5.13% as the minimum value and 8.83% as the maximum value, with upper and lower weights of 1 and an importance of 5. The texture was optimized within a range of 5.13% to 8.85%, with upper and lower weights set to 1 and an importance level of 4. Flavor and sweetness were maximized by 4.99% as the minimum and 8.67% as the maximum range, with upper and lower weights of 1 and an importance of 4. The overall acceptability ranged from 5.19% as the minimum value and 8.48% as the maximum value, with upper and lower weights of 1 and an importance of 3, as shown in Table 2.3.7. Predicted vs. observed values
Table 5 compares the predicted and observed values for various parameters related to a study on a particular substance, most likely a food product. Table 5 clearly shows that the observed and predicted values significantly differed (P < 0.05). In Table 5, the predicted values refer to the responses estimated by a statistical or mathematical model developed during the study, rather than values obtained directly from experimental measurements. These predicted values are generated to describe how the studied parameters are expected to behave under specific experimental conditions. Typically, predicted values are obtained through modelling approaches such as Response Surface Methodology (RSM), regression analysis, or other optimization techniques.| Responses | Predicted values | Observed values |
|---|---|---|
| a Data are expressed as mean ± standard deviation (n = 3), and values with different lowercase letters in the same column are significantly different (Tukey, P < 0.05). | ||
| Total flavonoid content (mg QE per g) | 48.150b | 48.18 ± 0.01a |
| Total phenolic content (mg GAE per g) | 32.39b | 32.40 ± 0.02a |
| DPPH (%) | 25.421b | 25.45 ± 0.08a |
| Appearance and colour | 8.577b | 8.76 ± 0.04a |
| Texture | 8.457b | 8.60 ± 0.02a |
| Flavour and sweetness | 8.335b | 8.69 ± 0.04a |
| Overall acceptability | 8.248b | 8.25 ± 0.010a |
According to the best combination suggested by the D-optimal design (Table 3, Fig. 3 and 4), sample W15 (jowar flour = 25 g, bajra flour = 25 g, wheat flour = 40 g and jujuba seed powder = 10 g) was the most preferable and had high desirability (SI Fig. 2), a sensory value of 8.24 ± 0.02 and an antioxidant DPPH value of 25.43 ± 0.01% (Tables 3 and 4). This sample was further chosen for proximate analysis and shelf-life analysis.
3.8. Principal component analysis (PCA)
This study used PCA to describe treatments, analyze the relationships between traits, and effectively discriminate treatments. PCA is a statistical technique that is widely used for a variety of traits in similar applications. It is a very effective way to group highly correlated variables and is used to extract significant information from high-dimensional data to low-dimensional data. The first principal component (PC1) accounted for 88.53% of the total variance, while the second principal component (PC2) explained 8.91%, together capturing 97.44% of the total variability, indicating that the PCA model effectively summarized the multivariate relationships among treatments and measured traits. The PCA biplot illustrates the relationships between experimental treatments and measured traits based on the first two principal components. Treatments positioned close to each other exhibit similar multivariate characteristics, while greater distances indicate dissimilarity. The vectors for total phenolic content (TPC), total flavonoid content (TFC), and antioxidant activity (DPPH) showed strong loadings on the first two components, indicating their major contribution to sample discrimination. Treatments W4, W10, and W11 were positioned closer to these vectors, suggesting a stronger association with antioxidant-related traits. Although W15 was oriented in the same general direction, its greater distance from W4 indicates that it does not cluster with W4 and exhibits a distinct overall response profile. In coordinate-1, W4, W10, W11 and W15 were clustered where the vectors for total phenolic content (TPC), total flavanoid content (TFC) and antioxidant activity (DPPH) are shown, ensuring a strong bond between them (W10, W11 and W15) as shown in Fig. 5.3.9. Optimized product proximate composition
The moisture content of the Ziziphus jujuba seed-based waffles was determined, and the estimated value was 10.76 ± 0.15% (SI Table 3). A study conducted by Chen et al.54 described a method for increasing the moisture content of cooked pancakes by 6 to 12%. The moisture content analysis of the above-mentioned studies was in agreement with the estimated value of moisture content obtained in this study. The estimated value denotes the equilibrium moisture in the environment. The higher the moisture content, the greater the chance of bacterial and microbial growth, which can lead to a decrease in the shelf-life period of the waffles.The ash content of the moisture-free Ziziphus jujuba seed-based waffles was analyzed, and the value obtained was 4.26 ± 0.16% (SI Table 3). Research conducted by Rani et al.55 on pancakes incorporated with field pea flour and besan revealed ash contents ranging from 1.57 to 3.07%. The ash content analysis of the abovementioned studies was in disagreement with the estimated value of ash content obtained in this study. The findings of these studies on the ash content of jujube seed flour-based waffles are relatively high compared with our findings. This proves that a greater mineral content is present in the sample.
The carbohydrate content was obtained by subtracting the proximal values of moisture, ash, protein, fat, and fiber from 100. The percentage of carbohydrates obtained from the jujube seed flour-based waffles was 16.81 ± 0.12% (SI Table 3). A study conducted by Kennedy et al.56 revealed the presence of 20 to 50% carbohydrates in pancakes. These estimated values were in agreement with our findings concerning the % carbohydrate composition of jujuba seed flour. The carbohydrate content indicates the starch and sugar contents, such as those of amylose and amylopectin. It acts as an energy-providing source to meet the desired dietary requirements.
The protein content of the jujuba seed-based waffles was determined via the Kjeldahl method, and the estimated value was 39.34 ± 0.31% (SI Table 3). Additionally, a study on waffles containing pea protein, chicory root syrup, apple fiber, finger millet, pearl millet, resistant starch, and sacred lotus stamen revealed an increase in protein content.57 This finding is in agreement with the results indicating that this incorporation significantly increased the protein content of the waffles.
The fat content of the jujuba seed-based waffles was estimated, and the value obtained after triplicate analysis was 10.34 ± 0.09% (SI Table 3). Studies conducted by Huber & Schoenlechner58 reported the use of a mixture of margarine and Raftilin in waffle crisps at a ratio of 23.33–25.98%. This is in disagreement with the results due to the use of different fats, which might affect their fatty acid profiles, resulting in differences in fat content. Overall, the fat content of waffles is due to the milk and butter used, which makes them excellent sources of necessary fatty acids with certain nutritional advantages.
Fiber content estimation was performed, and the estimated value was 18.49 ± 0.11% (SI Table 3). A study on waffles by Ioniţă–Mîndrican et al.59 revealed the presence of 5 g of fiber per serving. These studies do not support the fiber estimation of our findings and disagree with the compositional value. The results highlighted a significant increase in the amount of fiber of waffles due to millet and seed powder incorporation, highlighting their nutritional value and possible use as industrial raw materials and in food formulations.
3.10. Shelf-life study of the optimized product
| Storage days | Temperature (°C) | Packaging materials | Total flavonoid content (mg QE per g) | Total phenolic content (mg GAE per g) | DPPH activity (%) | Moisture content (%) | Total plate count (×102 CFU g)−1 | Total yeast and mold count (×102 CFU g)−1 |
|---|---|---|---|---|---|---|---|---|
| a Data are expressed as mean ± standard deviation (n = 3), and values with different lowercase letters in the same column are significantly different (Tukey, p < 0.05); abbreviations: LDPE, Low-Density Poly Ethylene; MP, Metallic Polyester. | ||||||||
| 0 | 8 | LDPE | 31.45 ± 0.33a,b | 15.60 ± 0.06a | 5.01 ± 0.23a | 2.47 ± 0.17b | 0.01 ± 0.23b | 0.04 ± 0.19b |
| 0 | 8 | MP | 31.44 ± 0.45a,b | 15.70 ± 0.54a | 5.02 ± 0.32a | 2.45 ± 0.49b | 0.02 ± 0.54b | 0.03 ± 0.32b |
| 0 | 25 | LDPE | 31.56 ± 0.34a,b | 15.60 ± 0.09a,b | 5.03 ± 0.25a | 2.47 ± 0.13b | 0.01 ± 0.32a,b | 0.04 ± 0.23a,b |
| 0 | 25 | MP | 31.55 ± 0.47a,b | 15.50 ± 0.57a,b | 5.01 ± 0.35a | 2.44 ± 0.53b | 0.03 ± 0.57a,b | 0.02 ± 0.37a,b |
| 0 | 37 | LDPE | 31.43 ± 0.32a | 15.60 ± 0.12a | 5.02 ± 0.29a,b | 2.47 ± 0.12a | 0.01 ± 0.33a | 0.04 ± 0.28a |
| 0 | 37 | MP | 31.44 ± 0.49a | 15.50 ± 0.59a | 5.03 ± 0.38a,b | 2.46 ± 0.56a | 0.04 ± 0.59a | 0.05 ± 0.43a |
| 4 | 8 | LDPE | 30.67 ± 0.36a,b | 15.20 ± 0.15b | 5.00 ± 0.32a | 2.47 ± 0.13b | 0.12 ± 0.39b | 0.06 ± 0.26b |
| 4 | 8 | MP | 30.66 ± 0.53a | 15.30 ± 0.63a,b | 4.90 ± 0.42b | 2.48 ± 0.59b | 0.11 ± 0.62b | 0.04 ± 0.48b |
| 4 | 25 | LDPE | 25.67 ± 0.39a,b | 13.47 ± 0.19a,b | 4.97 ± 0.36a,b | 2.78 ± 0.15b | 0.28 ± 0.42a | 0.13 ± 0.25b |
| 4 | 25 | MP | 25.65 ± 0.57a,b | 13.46 ± 0.67a,b | 4.98 ± 0.46a,b | 2.77 ± 0.63b | 0.27 ± 0.66a | 0.12 ± 0.52b |
| 4 | 37 | LDPE | 20.34 ± 0.42a | 10.97 ± 0.22b | 4.78 ± 0.39b | 3.23 ± 0.22a | 0.45 ± 0.45a | 0.24 ± 0.27a |
| 4 | 37 | MP | 20.33 ± 0.59a | 10.96 ± 0.69b | 4.79 ± 0.48b | 3.24 ± 0.67a | 0.43 ± 0.69a | 0.25 ± 0.56a |
| 8 | 8 | LDPE | 29.23 ± 0.45a | 14.93 ± 0.26b | 4.95 ± 0.41a,b | 2.68 ± 0.25b | 0.20 ± 0.51b | 0.10 ± 0.29b |
| 8 | 8 | MP | 29.22 ± 0.61a | 14.94 ± 0.73b | 4.94 ± 0.51a,b | 2.67 ± 0.71b | 0.10 ± 0.72b | 0.2 ± 0.53b |
| 8 | 25 | LDPE | 22.37 ± 0.47a,b | 10.35 ± 0.29a,b | 4.88 ± 0.38a,b | 2.96 ± 0.31b | 0.47 ± 0.55a | 0.28 ± 0.32b |
| 8 | 25 | MP | 22.38 ± 0.65a,b | 10.36 ± 0.77a,b | 4.87 ± 0.55a,b | 2.95 ± 0.74b | 0.46 ± 0.75a | 0.27 ± 0.57b |
| 8 | 37 | LDPE | 17.76 ± 0.51a | 8.63 ± 0.32b | 4.63 ± 0.42b | 3.92 ± 0.34a | 0.83 ± 0.59a | 0.52 ± 0.36a |
| 8 | 37 | MP | 17.78 ± 0.68a | 8.64 ± 0.79b | 4.62 ± 0.59b | 3.93 ± 0.77a | 0.84 ± 0.78a | 0.53 ± 0.54a |
| 12 | 8 | LDPE | 19.32 ± 0.53b | 13.78 ± 0.35b | 4.9 ± 0.45b | 2.96 ± 0.37b | 0.29 ± 0.61b | 0.14 ± 0.39b |
| 12 | 8 | MP | 19.33 ± 0.66b | 13.77 ± 0.82b | 4.8 ± 0.62b | 2.94 ± 0.78b | 0.28 ± 0.81b | 0.15 ± 0.59b |
| 12 | 25 | LDPE | 25.53 ± 0.55a | 9.63 ± 0.31a,b | 4.76 ± 0.49b | 3.15 ± 0.42b | 0.98 ± 0.64a | 0.36 ± 0.37b |
| 12 | 25 | MP | 25.55 ± 0.69a,b | 9.62 ± 0.85a,b | 4.77 ± 0.65b | 3.14 ± 0.83b | 0.97 ± 0.84a | 0.35 ± 0.63b |
| 12 | 37 | LDPE | 13.45 ± 0.57a | 7.45 ± 0.34b | 4.57 ± 0.47b | 4.01 ± 0.45a | 1.57 ± 0.68a | 0.78 ± 0.41a |
| 12 | 37 | MP | 13.44 ± 0.72a | 7.44 ± 0.87a | 4.56 ± 0.68b | 4.04 ± 0.87a | 1.56 ± 0.86a | 0.77 ± 0.67a |
The total yeast and mold counts followed a similar trend to that of the total plate count. Initially, the yeast and mold counts were low (0.02–0.05) and increased over time. By 12 days, the counts reached 0.77–0.78 at 37 °C. The data revealed a clear increase in yeast and mold growth with increasing temperatures and extended storage periods.61 The differences between packaging materials are that LDPE and MP provide similar levels of protection, with LDPE slightly outperforming MP under some conditions. Both materials effectively manage moisture content and microbial growth, but higher temperatures and longer storage durations lead to increased moisture and microbial proliferation.62 In summary, the results of this study indicate that the shelf-life of Ziziphus jujuba seed-based waffles is significantly influenced by the storage temperature and duration. Higher temperatures and extended storage times result in increased moisture content and microbial growth. While both LDPE and MP packaging materials are effective, LDPE shows a marginal advantage in controlling these parameters under certain conditions.
Additionally, the phenolic content tended to decrease over the storage period, with a gradual decline.63 Initially, the phenolic content was relatively stable across all the samples at 15.5–15.7 mg GAE per g, as shown in Table 6. However, the phenolic content decreases over time, especially at relatively high temperatures. By day 12, the phenolic content was significantly lower at 37 °C (7.44–7.45 mg GAE per g). At lower temperatures (8 °C), the decline is less severe, with values ranging from approximately 13.77–13.78 mg GAE per g. The stability of phenolic compounds decreases with time and temperature.64 Both packaging materials exhibit comparable effectiveness in retaining phenolic content.
Initially, the DPPH activity was approximately 5.01–5.03%. Over time, there is a slight reduction, with a more noticeable decline at higher temperatures. By day 12, the DPPH activity decreased to 4.56–4.57% at 37 °C, whereas at 8 °C, it remained relatively high (4.8–4.9%). Similar results were observed, in which the DPPH activity decreased with increasing temperature and time for the food products.65 LDPE and MP both maintained similar levels of antioxidant activity. A shelf-life study revealed that storage temperature and duration significantly impact the biochemical and antioxidant properties of Ziziphus jujuba seed-based waffles. Higher temperatures and longer storage durations lead to greater degradation of the flavonoid content, phenolic content, and antioxidant activity. Both LDPE and MP packaging materials provide comparable protection, with LDPE offering a slight advantage under certain conditions. Maintaining lower storage temperatures can help preserve the quality of waffles over time.66
LDPE (low-density polyethylene) is more effective than multilayered polyethylene (MP) for packaging, especially in preserving the quality of products such as waffles. LDPE offers superior moisture resistance, which is crucial for maintaining the texture and freshness of food items. Its flexibility also ensures an airtight seal, reducing the risk of contamination and spoilage.
4 Conclusion
The current study aimed to increase waffle nutritional value by incorporating Ziziphus jujuba seed flour while maintaining its sensory quality parameters. The optimal formulation of 25% jowar flour, 25% bajra flour, 40% wheat flour, and 10% jujuba seed flour achieved high nutritional and sensory scores. This formulation had a flavonoid content of 48.17 ± 0.01 mg QE per g, a phenolic content of 32.39 ± 0.04 mg GAE per g, and an antioxidant activity (DPPH) of 25.43 ± 0.01%. The sensory scores included 8.74 ± 0.02 for appearance, 8.62 ± 0.02 for texture, 8.67 ± 0.01 for flavor, and 8.24 ± 0.02 for overall acceptability. Further analysis of proximate composition and shelf-life stability via microbial studies and biochemical assays was conducted. Compared with metallic polyester (MP), low-density polyethylene (LDPE) has superior results in preserving the antioxidant activity, flavonoid content, and overall freshness of waffles, making it the preferred choice for extending shelf-life. The incorporation of Ziziphus jujuba seed flour significantly enhanced nutritional qualities, particularly flavonoid and phenolic contents, without compromising sensory attributes. This study demonstrates the potential for nutritionally enriched, sensory-pleasing waffle products, offering health benefits and market growth in the functional food industry.Ethical approval
This study involved sensory evaluation conducted with semi-trained panellists. The sensory evaluation process for the beverage was approved by the Lovely Professional University Ethical Committee (reference number LPU/CA/025/28/06/14135). All semi-trained participants provided explicit informed consent and were fully apprised of the study nature and their right to withdraw at any stage. No additional regulations were required, affirming our commitment to ethical standards.Author contributions
A. J., and M. G.: investigation, writing-original draft; visualization; S. E. and S. S. G.: supervision, validation, review and editing; P. R. and J. S.: supervision, validation, reviewing and editing, software, conceptualization.Conflicts of interest
The authors declare that there are no conflicts of interest regarding the publication of this article.Data availability
The data will be made available upon request.Supplementary information (SI): information and raw data of ANOVA and regression analysis of waffles, constraints for selecting the best formulation and proximate composition of optimized waffles. See DOI: https://doi.org/10.1039/d5fb00822k.
Acknowledgements
The authors gratefully acknowledge the Central Instrumentation Facility of Lovely Professional University, Phagwara, Punjab, India for providing access to analytical instruments and technical support essential for conducting the analyses required in this research.References
- I. Oliveira, B. Marinho, U. Szymanowska, M. Karas and A. Vilela, Chemical and sensory properties of waffles supplemented with almond skins, Molecules, 2023, 28(15), 5674, DOI:10.3390/molecules28155674.
- W. Cai, H. Zhuang, X. Wang, X. Fu, S. Chen and L. Yao, et al., Functional nutrients and jujube-based processed products in Ziziphus jujuba, Molecules, 2024, 29(14), 3437, DOI:10.3390/molecules29143437.
- R. Laoufi, K. Boudjema, S. Lefkir, N. Benhabyles, O. Bouchenak and K. Yahiaoui, Valorization and biological effect of Ziziphus jujuba Mill. vegetable oil, Nutr. Santé, 2023, 12(1), 38–47, DOI:10.30952/ns.12.1.5.
- Y. Hua, X. X. Xu, S. Guo, H. Xie, H. Yan and X. F. Ma, et al., Wild jujube (Ziziphus jujuba var. spinosa): A review of its phytonutrients, health benefits, metabolism, and applications, J. Agric. Food Chem., 2022, 70(26), 7871–7878, DOI:10.1021/acs.jafc.2c01905.
- K. D. Kanbargi, S. K. Sonawane and S. S. Arya, Functional and antioxidant activity of Ziziphus jujube seed protein hydrolysates, J. Food Meas. Char., 2015, 10(2), 226–235, DOI:10.1007/s11694-015-9297-5.
- B. Antonic, D. Dordevic, S. Jancikova, D. Holeckova, B. Tremlova and P. Kulawik, Effect of grape seed flour on the antioxidant profile, textural and sensory properties of waffles, Processes, 2021, 9(1), 131, DOI:10.3390/pr9010131.
- M. A. Kahfi, A. N. Sutisna, H. Ainia and A. R. Cecep, Using Design Expert D-optimal for formula optimization of a functional drink enriched with moringa leaf extract (Moringa oleifera), IOP Conf. Ser. Earth Environ. Sci., 2021, 759(1), 012002, DOI:10.1088/1755-1315/759/1/012002.
- M. C. Chang and Y. J. Choo, Effects of whey protein, leucine, and vitamin D supplementation in patients with sarcopenia: A systematic review and meta-analysis, Nutrients, 2023, 15(3), 521, DOI:10.3390/nu15030521.
- B. I. Yerima and H. M. Adamu, Proximate chemical analysis of nutritive contents of jujube (Ziziphus mauritiana) seeds, Int. J. Phys. Sci., 2011, 6(36), 8079–8082, DOI:10.5897/ijps09.024.
- C. N. Nwakife, Y. A. Iyaka and M. Musa, Proximate, elemental and anti-nutritional compositions of the pulp and seeds of Ziziphus lotus L. from Minna Metropolis and its environs, KASU J Chem Sci., 2021, 1(2), 57–67 Search PubMed.
- A. Hussain, S. Laaraj, T. Kausar, A. Tikent, H. Azzouzi and S. Kauser, et al., Food application of orange seed powder through incorporation in wheat flour to boost vitamin and mineral profiles of formulated biscuits, Int. J. Food Sci., 2023, 2023(1), 6654250, DOI:10.1155/2023/6654250.
- A. K. Bhardwaj, N. K. Kashyap, S. K. Bera, M. Hait and H. Dewangan, Proximate composition and mineral content analysis of Curcuma caesia rhizome, Biochem. Syst. Ecol., 2023, 109, 104661, DOI:10.1016/j.bse.2023.104661.
- X. Xue, A. Zhao, Y. Wang, H. Ren, J. Du, D. Li and Y. Li, Composition and content of phenolic acids and flavonoids among the different varieties, development stages, and tissues of Chinese jujube (Ziziphus jujuba Mill.), PLoS One, 2021, 16(10), e0254058, DOI:10.1371/journal.pone.0254058.
- J. Singh, P. Rasane, S. Kaur and V. Nanda, Comparative analysis of antioxidant potential and techno-functional properties of selected corn silk varieties at different developmental stages, J. Food Meas. Char., 2022, 16(4), 2685–2698, DOI:10.1007/s11694-022-01382-6.
- J. Orsavová, T. Juríková, R. Bednaříková and J. Mlček, Total phenolic and total flavonoid content, individual phenolic compounds and antioxidant activity in sweet rowanberry cultivars, Antioxidants, 2023, 12(4), 913, DOI:10.3390/antiox12040913.
- A. Rajaei, D. Salarbashi, N. Asrari, B. S. F. Bazzaz, S. M. Aboutorabzade and R. Shaddel, Antioxidant, antimicrobial, and cytotoxic activities of extracts from the seed and pulp of Ziziphus jujuba grown in Iran, Food Sci. Nutr., 2020, 9(2), 682–691, DOI:10.1002/fsn3.2031.
- S. Arjmand, N. Mollakhalili-Meybodi, F. Akrami Mohajeri, F. Madadizadeh and E. Khalili Sadrabad, Quinoa dough fermentation by Saccharomyces cerevisiae and lactic acid bacteria: Changes in saponin, phytic acid content, and antioxidant capacity, Food Sci. Nutr., 2023, 11(12), 7594–7604, DOI:10.1002/fsn3.3679.
- A. Budiman, A. Rusdin and D. L. Aulifa, Current techniques of water solubility improvement for antioxidant compounds and their correlation with its activity: Molecular pharmaceutics, Antioxidants, 2023, 12(2), 378, DOI:10.3390/antiox12020378.
- A. Kakar, T. F. Miano, A. H. Soomro, A. Yar, S. A. Memon and B. Khan, Oil and water absorption capacity of wheat, rice and gram flour powders, Int. J. Ecosyst. Ecol. Sci., 2022, 12(2), 585–594, DOI:10.31407/ijees12.232.
- B. O. Timothy and O. O. Abigail, Temperature impact on swelling power and solubility of ogi powder produced from four indigenous maize varieties, Ann.:Food Sci. Technol., 2017, 18(1), 21–28 Search PubMed.
- S. Geng, Y. Wang and B. Liu, Fabrication, characterization and application of Pickering emulsion gels stabilized by defatted grape seed powder, Food Chem.:X, 2024, 22, 101476, DOI:10.1016/j.fochx.2024.101476.
- N. Aro, D. Ercili-Cura, M. Andberg, P. Silventoinen, M. Lille and W. Hosia, et al., Production of bovine beta-lactoglobulin and hen egg ovalbumin by Trichoderma reesei using precision fermentation technology and testing of their techno-functional properties, Food Res. Int., 2023, 163, 112131, DOI:10.1016/j.foodres.2022.112131.
- A. Nawaz, I. Khalifa, N. Walayat, J. M. Lorenzo, S. Irshad and Abdullah, et al., Whole fish powder snacks: Evaluation of structural, textural, pasting, and water distribution properties, Sustainability, 2021, 13(11), 6010, DOI:10.3390/su13116010.
- T. Ahmed, A. A. Sabuz, A. Mohaldar, H. S. Fardows, B. S. Inbaraj and M. Sharma, et al., Development of novel whey-mango based mixed beverage: Effect of storage on physicochemical, microbiological, and sensory analysis, Foods, 2023, 12(2), 237, DOI:10.3390/foods12020237.
- Y. Zarroug, J. Sriti, D. Sfayhi, B. Slimi, W. Allouch and K. Zayani, et al., Effect of addition of Tunisian Ziziphus lotus L. fruits on nutritional and sensory qualities of cookies, Ital. J. Food Sci., 2021, 33(4), 84–97, DOI:10.15586/ijfs.v33i4.2095.
- M. Gunjal, A. Khalangre, J. Singh, S. Kaur, S. Ercisli, E. Macit and P. Rasane, Process optimization for microgreens-based dairy (lassi) beverage: Consumer acceptability, bioactive composition, and storage life using kinetic modelling, J. Stored Prod. Res., 2025, 112, 102596, DOI:10.1016/j.jspr.2025.102596.
- G. Sharma, D. Banik, C. M. Mehta, E. Nishihara, K. Inubushi and S. Sudo, et al., A comparative study of fresh and residual biochar effects on wheat growth and yield metrics, Eurasian J. Soil Sci., 2025, 14(2), 158–177, DOI:10.18393/ejss.1647314.
- A. Siddique, A. Shafique, I. Younas, A. A. Abbasi, U. Ali and T. A. Mughal, et al., Antibacterial activity of three indigenous plants against pathogenic bacteria, Kashmir J. Sci., 2023, 2(1), 60–69 CrossRef.
- S. Z. Asadi, M. A. Khan and S. Zaidi, A study on the shelf life of cookies incorporated with sapota and beetroot leaf powders, J. Food Sci. Technol., 2022, 59(10), 3848–3856, DOI:10.1007/s13197-022-05408-1.
- M. Chouaibi, N. Mahfoudhi, L. Rezig, F. Donsì, G. Ferrari and S. Hamdi, Nutritional composition of Ziziphus lotus L. seeds, J. Sci. Food Agric., 2011, 92(6), 1171–1177, DOI:10.1002/jsfa.4659.
- T. F. Fukalova, M. D. García-Martínez, P. E. Ciudad and M. D. R. Jiménez, The effects of low-input (wild and organic farming) conditions on the nutritional profile of Ziziphus jujuba Mill. fruits from the Valencian Mediterranean, Sustainability, 2023, 15(19), 14587, DOI:10.3390/su151914587.
- H. N. Adilah, M. I. Saleh, N. D. A. Az-Zahra, E. Cho and E. Sinaga, Total phenolic and total flavonoid content, antioxidant activity, and nutritional profile of Ziziphus mauritiana fruit juice, Int. J. Biol. Phys. Chem. Stud., 2023, 5(1), 1–8, DOI:10.32996/ijbpcs.2023.5.1.1.
- A. Snoussi, H. B. H. Koubaier, M. Chouaibi and N. Bouzouita, Comparative study of the fatty acids and tocopherol profiles, physical properties, and antioxidant activities of Ziziphus lotus L. seed oils based on the geographical origin, Chem. Afr., 2021, 5(1), 69–78, DOI:10.1007/s42250-021-00294-0.
- H. N. Murthy, K. S. Joseph, A. Madiwal, D. R. Gerald, M. Badiger, L. Kolkar and R. Hiremath, Physicochemical characteristics of the seed and seed oil of the potentially medicinal plant Ziziphus oenopolia, J. Diet. Suppl., 2017, 14(6), 691–696, DOI:10.1080/19390211.2017.1304485.
- I. A. Amoo and O. A. Jokotagba, Comparative analysis of proximate, minerals, and functional properties of Tamarindus indica pulp and Ziziphus spina-christi fruit and seed, Greener J. Agric. Sci., 2012, 2(1), 21–25, DOI:10.15580/gjas.2013.3.11-020.
- M. A. Halim, M. A. Wazed, S. Al Obaid, M. J. Ansari, A. Tahosin, M. T. Rahman, F. Noor, N. R. Mozumder and A. A. Khatun, Effect of storage on physicochemical properties, bioactive compounds and sensory attributes of drinks powder enriched with pumpkin (cucurbita moschata L.), J. Agric. Food Res., 2024, 18, 101337, DOI:10.1016/j.jafr.2024.101337.
- C. Rais, A. Driouch, C. Slimani, A. Bessi, M. Balouiri and L. E. Ghadraoui, et al., Antimicrobial and antioxidant activity of pulp extracts from three populations of Ziziphus lotus L, Nutr. Food Sci., 2019, 49(6), 1014–1022, DOI:10.1108/nfs-08-2018-0232.
- A. Markovski and L. Velkoska-Markovska, Content of vitamin C in the fruits of jujube (Ziziphus jujuba Mill.) varieties and their progenies, Agric. For., 2016, 62(2), 217–225, DOI:10.17707/agricultforest.62.2.19.
- A. Naz, N. Raza, M. Sharif, K. Razzaq, S. Ullah and M. I. Afzal, et al., Performance evaluation of different grown ber (Ziziphus mauritiana) cultivars under low-temperature storage, Int. J. Food Prop., 2023, 26(1), 1736–1748, DOI:10.1080/10942912.2023.2230377.
- M. K. M. Akanda and A. H. M. N. Hasan, Characterization of pharmacological properties of methanolic seed and stem bark extracts of Ziziphus mauritiana (BAU Kul) using in vitro and in vivo animal (Swiss albino male mice) model, Clin. Phytosci., 2021, 7(1), 8, DOI:10.1186/s40816-020-00246-0.
- R. Chaimae, B. Meryem, S. Chaimae, E. O. Bouchamma, E. Hamza and E. Laila, et al., Antimicrobial and radical scavenging activities of Moroccan Ziziphus lotus L. seeds, J. Phytopharm., 2019, 8(4), 155–160, DOI:10.31254/phyto.2019.8402.
- I. E. Sallam, A. Abdelwareth, H. Attia, R. K. Aziz, M. N. Homsi, M. von Bergen and M. A. Farag, Effect of gut microbiota biotransformation on dietary tannins and human health implications, Microorganisms, 2021, 9(5), 965, DOI:10.3390/microorganisms9050965.
- P. W. P. Gomes, E. R. S. Gomes, A. R. Carvalho, H. Mannochio-Russo, T. F. Leão and J. D. E. Reis, et al., Characterization of a tetrameric proanthocyanidin in Stryphnodendron pulcherrimum and an overview on potential health benefits of condensed tannins via interaction with gut microbiota, J. Funct. Foods, 2023, 109, 105801, DOI:10.1016/j.jff.2023.105801.
- S. J. Liu, Y. P. Lv, Z. S. Tang, Y. Zhang, H. B. Xu and D. B. Zhang, et al., Ziziphus jujuba Mill., a plant used as medicinal food: A review of its phytochemistry, pharmacology, quality control, and future research, Phytochem. Rev., 2020, 20(3), 507–541, DOI:10.1007/s11101-020-09709-1.
- R. P. Panjaitan, D. Kartikawati and W. D. Ilminingtyas, The use of pregelatinized sweetcorn (Zea mays saccharata Sturt.) in the production of instant corn bakwan flour mix, IOP Conf. Ser. Earth Environ. Sci., 2023, 1177(1), 012037, DOI:10.1088/1755-1315/1177/1/012037.
- R. Kaur and K. Prasad, Characterization and classification of selective Indian chickpea cultivars based on physical and hydration properties, using image and multivariate analysis, Legume Sci., 2023, 5(3), e168, DOI:10.1002/leg3.168.
- B. Koç and T. Yörükoğlu, The effect of carrier agent addition on the physicochemical properties and sorption behavior of freeze-dried jujube, Turk. J. Agric. Food Sci. Technol., 2023, 11(1), 18–28, DOI:10.24925/turjaf.v11i1.18-28.5403.
- K. Chiodza and N. J. Goosen, Emulsion formation during enzymatic protein hydrolysis and its effect on protein recovery and molecular weight distribution of protein hydrolysates from sardine (Sardina pilchardus) byproducts, Biomass Convers. Biorefin., 2023, 14(19), 24069–24080, DOI:10.1007/s13399-023-04438-9.
- H. Wang, A. Yue, X. Zhang, J. Tang, X. He and X. Luo, et al., Bulk density response and experimental study of pulsed neutron-gamma density logging, Front. Earth Sci., 2022, 10, 803775, DOI:10.3389/feart.2022.803775.
- J. Song, J. Liu, K. Wang, L. Gao, X. Wang, J. Peng and N. Wang, Effect of jujube powder addition on the aroma profile of quinoa snacks (QS), Food Sci. Nutr., 2024, 12(7), 4810–4818, DOI:10.1002/fsn3.4128.
- R. Singh, S. Singh and D. C. Saxena, Studies on standardization of alcohol-aided starch extraction process from Chenopodium album and its characterization, J. Food Meas. Char., 2021, 15(6), 5379–5391, DOI:10.1007/s11694-021-01105-3.
- N. Fakhar, S. A. Khan, W. A. Siddiqi and T. A. Khan, Ziziphus jujube waste-derived biomass as a cost-effective adsorbent for the sequestration of Cd2+ from aqueous solution: Isotherm and kinetics studies, Environ. Nanotechnol. Monit. Manag., 2021, 16, 100570, DOI:10.1016/j.enmm.2021.100570.
- K. Kelley, A. M. Raczkowski, O. Klykov, P. Jaroenlak, D. Bobe and M. Kopylov, et al., Waffle method: A general and flexible approach for improving throughput in FIB-milling, Nat. Commun., 2022, 13(1), 1857, DOI:10.1038/s41467-022-29501-3.
- C. Chen, M. Zhang, W. Liu and Z. Lin, Baking characteristic improvement and starch retrogradation inhibition of Chinese pancakes by hydrocolloids, J. Food Process. Preserv., 2022, 46(5), e16529, DOI:10.1111/jfpp.16529.
- A. Rani, P. Devi, U. C. Jha, K. D. Sharma, K. H. M. Siddique and H. Nayyar, Developing climate-resilient chickpea involving physiological and molecular approaches with a focus on temperature and drought stresses, Front. Plant Sci., 2020, 10, 478034, DOI:10.3389/fpls.2019.01759.
- S. Kennedy, L. Ryan and M. Clegg, The effect of a breakfast containing multiple functional ingredients on glucose response, insulin response, and cognitive performance in adolescents, Proc. Nutr. Soc., 2016, 75(OCE3), E204, DOI:10.1017/s0029665116002196.
- A. S. Burlyaeva, Y. U. Pronina, Z. H. Nabiyeva and A. I. Samadun, The study of the influence of plant additives on increasing the food and biological value of soft waffles, J. Almaty Technol. Univ., 2023 DOI:10.48184/2304-568X-2023-1-55-62.
- R. Huber and R. Schoenlechner, Waffle production: Influence of batter ingredients on sticking of waffles at baking plates—Part II: Effect of fat, leavening agent, and water, Food Sci. Nutr., 2016, 5(3), 513–520, DOI:10.1002/fsn3.425.
- C. B. Ioniţă-Mîndrican, K. Ziani, M. Mititelu, E. Oprea, S. M. Neacşu and E. Moroşan, et al., Therapeutic benefits and dietary restrictions of fiber intake: A state-of-the-art review, Nutrients, 2022, 14(13), 2641, DOI:10.3390/nu14132641.
- O. Panza, V. Lacivita, C. Palermo, A. Conte and M. A. Del Nobile, Food by-products to extend shelf life: The case of cod sticks breaded with dried olive paste, Foods, 2020, 9(12), 1902, DOI:10.3390/foods9121902.
- E. Ezaka, O. Nchedo, E. Ugbo, A. Adediran and O. Ayanda, Effects of environmental factors on the growth and proliferation of yeasts, Niger. J. Biotechnol., 2021, 38(1), 166–178, DOI:10.4314/njb.v38i1.20.
- P. V. Rodrigues, D. M. Vieira, P. C. Martins, V. G. Martins, M. C. R. Castro and A. V. Machado, Evaluation of active LDPE films for packaging of fresh orange juice, Polymers, 2022, 15(1), 50, DOI:10.3390/polym15010050.
- D. Šuput, L. Pezo, B. Lončar, S. Popović, A. T. Horecki and T. Daničić, et al., The influence of biopolymer coating based on pumpkin oil cake activated with Mentha piperita essential oil on the quality and shelf-life of grape, Coatings, 2023, 13(2), 299, DOI:10.3390/coatings13020299.
- M. Mrázková, D. Sumczynski and J. Orsavová, Influence of storage conditions on stability of phenolic compounds and antioxidant activity values in nutraceutical mixtures with edible flowers as new dietary supplements, Antioxidants, 2023, 12(4), 962, DOI:10.3390/antiox12040962.
- L. Angeli, S. Imperiale, Y. Ding, M. Scampicchio and K. Morozova, A novel stoichio-kinetic model for the DPPH assay: The importance of the side reaction and application to complex mixtures, Antioxidants, 2021, 10(7), 1019, DOI:10.3390/antiox10071019.
- K. Saarniit, H. Lang, R. Kuldjärv, O. Laaksonen and S. Rosenvald, The stability of phenolic compounds in fruit, berry, and vegetable purees based on accelerated shelf-life testing methodology, Foods, 2023, 12(9), 1777, DOI:10.3390/foods12091777.
| This journal is © The Royal Society of Chemistry 2026 |