Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Recovering phytochemicals from a brewery by-product: a sustainable reuse proposal using a lactic acid-based deep eutectic solvent
Klycia Fidélis Cerqueira e Silva *a,
Paula Virginia de Almeida Pontesb,
Patrícia Tonon de Souzab,
Monique Martins Strieder
*a,
Paula Virginia de Almeida Pontesb,
Patrícia Tonon de Souzab,
Monique Martins Strieder c,
Eduardo Augusto Caldas Batista
c,
Eduardo Augusto Caldas Batista b and
Miriam Dupas Hubinger
b and
Miriam Dupas Hubinger *a
*a
aLaboratory of Process Engineering (LEP), Department of Food Engineering and Technology (DETA), School of Food Engineering, University of Campinas (UNICAMP), 13083-862, Campinas, São Paulo, Brazil. E-mail: klycia.fidelis@gmail.com; mhub@unicamp.br; Tel: +55(19)3521-4036
bLaboratory of Extraction, Applied Thermodynamics and Equilibrium (EXTRAE), Department of Food Engineering and Technology (DETA), School of Food Engineering, University of Campinas (UNICAMP), 13083-862, Campinas, São Paulo, Brazil
cMultidisciplinary Laboratory of Food and Health (LabMAS), School of Applied Sciences (FCA), University of Campinas (UNICAMP), Rua Pedro Zaccaria, 1300, Limeira, 13484-350, São Paulo, Brazil
First published on 26th March 2026
Abstract
Hot trub (HT), a nutrient-rich by-product of beer production, remains underutilized despite its potential for resource recovery. This study explores the valorization of HT, producing two phytochemical-rich extracts with potential functional applications and a protein-rich solid residue that can be repurposed as a food ingredient. The extraction of phytochemicals from HT, particularly xanthohumol (XN), was evaluated through a sequential extraction process employing a deep eutectic solvent (DES) composed of choline chloride (ChCl) and lactic acid (Lac) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio, followed by ethanol-based solvent extraction. The influence of temperature and DES-to-HT ratios was evaluated, as defined by the experimental design. DESs selectively extracted XN, reaching a maximum yield of 36 µg g−1 at 50 °C with 8 g of DES and 4 g of HT. However, sequential extraction with ethanol on DES-treated solid residues at 50 °C and equal DES-to-HT mass ratios yielded a higher XN extraction (∼114 µg g−1), emphasizing the use of DES as an effective pretreatment. The temperature influenced the extraction of other phytochemicals, including desmethylxanthohumol and phenolic acids, contributing to enriched antioxidant activity. Structural analysis of HT after extraction revealed modifications, such as shifts and loss of functional chemical groups induced by DESa, improving phytochemical accessibility for the ethanol extraction step. The analysis of the proposed process using the Path2Green metric demonstrated a strong alignment with the principles of green chemistry and the circular bioeconomy.
2 molar ratio, followed by ethanol-based solvent extraction. The influence of temperature and DES-to-HT ratios was evaluated, as defined by the experimental design. DESs selectively extracted XN, reaching a maximum yield of 36 µg g−1 at 50 °C with 8 g of DES and 4 g of HT. However, sequential extraction with ethanol on DES-treated solid residues at 50 °C and equal DES-to-HT mass ratios yielded a higher XN extraction (∼114 µg g−1), emphasizing the use of DES as an effective pretreatment. The temperature influenced the extraction of other phytochemicals, including desmethylxanthohumol and phenolic acids, contributing to enriched antioxidant activity. Structural analysis of HT after extraction revealed modifications, such as shifts and loss of functional chemical groups induced by DESa, improving phytochemical accessibility for the ethanol extraction step. The analysis of the proposed process using the Path2Green metric demonstrated a strong alignment with the principles of green chemistry and the circular bioeconomy.
Sustainability spotlightThis article demonstrates how beer residue can be valorized as a whole, serving as an alternative source of economically important phytochemicals, such as bitter acids that are primarily responsible for the bitter taste of beer, as well as indirectly acting as a raw material for plant-based proteins. To achieve this goal, we have developed an extraction process utilizing green solvents (ethanol and deep eutectic solvents), which enables the targeted extraction of phytochemicals and eliminates the need for high-cost processes that can fractionate compounds. Our work contributes to the implementation of the UN Sustainable Development Goals, particularly SDG 12 and SDG 15, by promoting efficient methods to reduce waste and minimize environmental impact through recycling for the production of food ingredients. |
1. Introduction
Beer is the most consumed alcoholic beverage in the world, with approximately 189 million kiloliters produced in 2022.1 Consequently, tons of food by-products, such as brewer's spent grains, hot trub (HT), spent hops, and brewer's spent yeast, were produced along with beer manufacturing steps per year. These by-products present significant nutritional properties, offering an excellent source of protein, fiber, and phytochemicals with therapeutic properties.2 Among brewing by-products, HT has been underutilized as a food source for human consumption and is typically used to produce animal feed or discarded as fertilizer. HT is a by-product generated during the wort boiling stage of beer production, formed by the combination of spent hops and the agglomeration of high-molecular-weight proteins with a wide range of phytochemicals. After proteins, phytochemicals are the most abundant components in HT and have garnered increasing interest because they can serve as active compounds for creating healthier and cosmetic products.3,4 The phytochemical market was valued at $1.6 billion in 2021 and is expected to increase at a compound annual growth rate of 7.4% from 2022 to 2030, demonstrating the economic potential of the HT use approach.5XN and bitter acids (Fig. 1) are the most commercially important phytochemicals found in HT due to their anticarcinogenic, antioxidant, anti-aging, and flavor properties.6,7 Contrasting bitter acids, XN has low water solubility, improving the cost-effectiveness of extraction in the hops industry. They have used solvents recognized as safe (GRAS) for XN extraction via a combination of supercritical CO2 extraction and adsorption techniques.8 When entering the XN extraction, it is important to acknowledge the main challenge: how to avoid the co-extraction of bitter acids, as reported by Silva et al.4 The co-extraction of a broad spectrum of phytochemicals is not essentially a big issue, but when aiming to produce an isolated commercial product, the use of a selective extraction medium can significantly reduce separation and purification costs. In this context, the use of Deep Eutectic Solvents (DESs) can be a strategy to enhance XN content in a selective way. Unlike other solvents, DESs are not pure substances but rather mixtures of compounds with tailored properties that can serve diverse purposes.9,10 Introduced by Abbott et al.,11 such systems are prepared by mixing a hydrogen bond donor (HBD) and a hydrogen bond acceptor (HBA), with chloride being the most commonly used HBA due to its low price, biocompatibility, low volatility, ease of preparation, non-flammability, miscibility with water, and high solvation.12–16
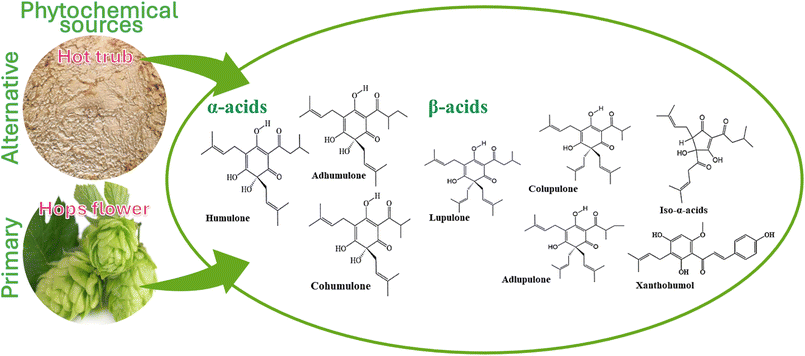 | ||
| Fig. 1 The scheme illustrates the resources of bitter acids and XN in relation to their molecular structure. | ||
Furthermore, DESs are designed to be recyclable and reusable, representing a commitment to sustainability in biorefinery processes via reducing toxic solvent consumption.17 Owing to these advantages, DES was used for XN extraction from spent hops and hop flowers, respectively, via combinations of alcohol-based18 and lactic acid-based DES (Lac-based DES),19 with the latter being the most reported approach.20 Extracts obtained using Lac-based DES can be directly applied in various fields without solvent removal, due to their low cytotoxicity. Specifically, these DESs exhibit the lowest cytotoxicity among chloride-based DESs containing organic acids when evaluated against HT-29, Caco-2, MCF-7, and MRC-5 cell lines, with cytotoxic effects shown to be concentration-dependent.21 Regarding their applications, DESs composed of ChCl and organic acids have demonstrated antimicrobial activity, due to the low pH of the system, which disrupts cell membranes and denatures membrane-associated proteins, leading to microbial death.22
Therefore, HT extracts obtained via the DES route can enhance product functionality while protecting against microbial activity. The novelty of this study lies in evaluating Lac-based DES extraction as a strategy to minimize the co-extraction of bitter acids during XN recovery, beyond the use of HT as an alternative source of hop-derived products. To this end, an experimental design approach was employed, as it allows the investigation of multiple variables with a limited number of experiments and the assessment of interaction effects on the response.23 Thus, as a first step, the effects of the DES ratio and temperature were examined to evaluate XN extraction and the co-extraction of other phytochemicals from wet HT. The second step consisted of recovering the remaining phytochemicals via ethanol extraction to maximize resource utilization. Along with evaluating the chemical profile and antioxidant activity of the extracts from both steps, the study also investigated how the extraction process modified the residual solids. Finally, the sustainability of the process was assessed using the Path2Green metric proposed by Souza Mesquita et al.24
2. Materials and methods
2.1. Materials and chemicals
The HT (corresponding to Pilsen beer formulated with Magnum, Tettnanger, and Hallertau tradition hops) was donated by the Cevada Pura brewery located in Piracicaba/São Paulo (Brazil). The HT material was stored at −22 °C and thawed as required for each assay. Its composition on a dry basis was 86.83 ± 0.03% of moisture, 18.2 ± 0.6% of total protein; 5.3 ± 0.3% of total fat; 1.65 ± 0.03% of ash; 74.85% of total carbohydrate (calculated by differences), with a particle size (D[43]) of 132.4 ± 6.7 µm measured via the laser diffraction method (Mastersizer 2000, Malvern, United Kingdom). It is necessary to highlight that HT was used in its wet form, as received by the brewing industry, throughout this study, from raw material characterization to the extraction processes. The characterization of ChCl (≥98%, Sigma-Aldrich, USA) showed a water content of 1.61 ± 0.38% (by the Karl Fischer method). The DES mixture in the ratio of 1 part ChCl to 2 parts Lac (prepared according to Section 2.2) had a density of 1.15 ± 0.15 g cm−3 and a viscosity of 0.095 ± 0.002 mPa s.2.2. DES preparation
According to Zhang et al.,25 the DES was prepared by mixing 50 g of ChCl with 100 g of Lac, and then stirring under magnetic agitation at 60 °C until a clear liquid was obtained. After preparation, the solvent was stored at room temperature for 24 h to ensure the formation of a translucent, solid-free liquid.2.3. Extraction process of polyphenol and bitter acids
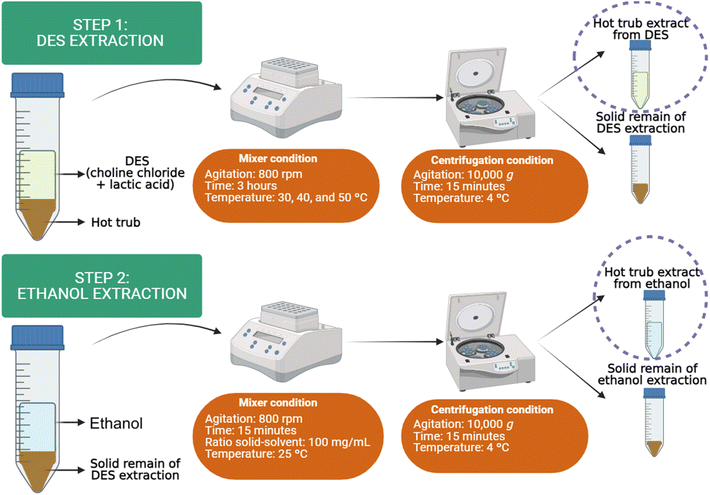
The extraction process was carried out by utilizing two successive extraction steps on the wet form of HT. The first extraction (step 1) involved applying DES, while the subsequent process (step 2) used ethanol to extract any remaining phytochemicals into the solid residue. It is important to emphasize that the second extraction was performed to maximize resource utilization and to determine how much XN remains in the solid residue after the DES extraction step. For step 1, an experimental design was applied in the first extraction to optimize the extraction parameters, considering the DES-to-HT mass (grams) and temperature (°C). A thermos-shaker (KASVI, K80-200 model, China) was used for the extraction process, with conditioning for 3 h at 800 rpm, as described by de Almeida Pontes et al.26| Independent variables | Levels | |||||
|---|---|---|---|---|---|---|
| −1.41 | −1 | 0 | +1 | +1.41 | ||
| Temperature (°C) | X1 | 25.9 | 30.0 | 40.0 | 50.0 | 54.1 |
| DES to HT mass ratio (g) | X2 | (3.18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
(4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
(6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
(8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
(8.82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
After step 1, the DES extracts were analyzed for XN content (Y1) and antioxidant capacity (Y2), and the experimental design responses were obtained. The solid residue remaining from step 1 led to step 2. Curvatures were investigated using a first-order model (eqn (1)) to explain the extraction pattern.
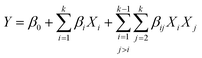 | (1) |
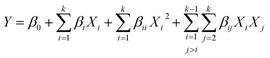 | (2) |
For both equations, i ≠ j; k is the number of independent variables; Y is the dependent variable (response), Xi and Xj are the independent variables (extraction parameters), and β0, βi, and βij are the regression coefficients for intercept, linear, and interaction parameters, respectively. For eqn (2), the quadratic parameters are represented by βii. It is noteworthy that if significant curvature is detected, axial points will be included to adjust the quadratic model's parameters.
The extraction protocol was the same as that employed with ethanol: in a thermo-shaker (KASVI, K80-200 model, China) under the fixed conditions of time (15 min), temperature (25 °C), solid-to-solvent ratio (100 mg mL−1), and agitation speed (800 rpm). Extracts and solid residues were stored in a freezer (−21 °C) until the subsequent analysis. The solid residues from all extraction steps were subjected to freeze-drying (LS 3000, Terroni Equipamentos Científicos, Brazil) at −40 °C and 100 µmHg for 48 h. After this, the samples were stored in desiccators.
2.4. Extract characterization
The mass detector was operated in the positive mode, ranging from 130 to 600 m/z (mass/charge ratio), under fixed conditions: source temperature of 100 °C, desolvation temperature of 300 °C, desolvation gas flow of 600 L h−1, cone gas flow of 60 L h−1, capillary voltage of 4 kV, extractor voltage of 5 V, RF lens of 0.7 V, and cone voltage ranging from 10 to 100 V.
2.5. Thermo-shaking versus magnetic mixing
A comparative analysis was proposed between thermo-shaking and magnetic mixing, focusing on evaluating the impact of these two mixing strategies on phytochemical yields and on the characterization of solid residues. This evaluation was conducted as a preliminary test to understand pattern extraction across different equipment, providing a foundation for future studies on the operational aspects of scale-up. Magnetic mixing (model Hei-Tec, Heidolph, Germany) was performed under the same operating conditions as thermo-shaking (as described in Sections 2.3 and 2.3.2), including temperature, processing time, DES-to-HT mass ratio, and ethanolic solution at 70%. For magnetic mixing, the DES extraction was conducted at 30 °C with agitation at 300 rpm for 3 h. Subsequently, the second step involved mixing a 70% (v/v) ethanolic solution with the solid residue at 25 °C for 15 min, maintaining agitation at 300 rpm. All the solid residues were dried via lyophilization operation for evaluation in an attempt to track microstructure alteration, while the extract was stored under refrigerator (−10 °C).2.6. Extraction process sustainability evaluation by Path2Green
The Path2Green metric was used to verify the sustainability score of the proposed extractive process, focusing on its effects on environmental, social, and economic aspects. This analysis evaluates biomass, transport, pretreatment, solvent, scaling, purification, yield, post-treatment, energy, application, repurposing, and waste management for the extraction process.2.7. Statistical analysis
In step 1, the experimental design, analysis of variance (ANOVA), and RSM were evaluated using TIBCO Statistica Software Inc., version 14 (2020). The results of step 2 were statistically analyzed using one-way ANOVA in Minitab® 16.0 (USA). Mean analysis was performed using Tukey's procedure at a p-value ≤0.05.3. Results and discussion
3.1. Polyphenol compound identification
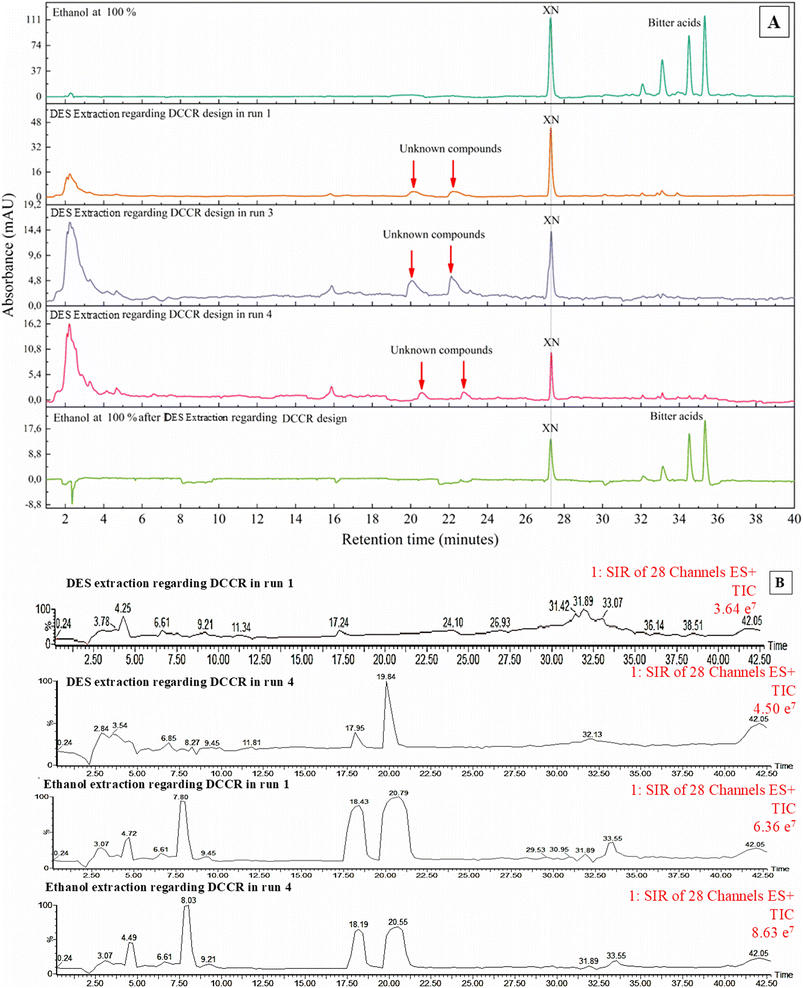
To evaluate the effect of DES, an ethanol extract (100% v/v) was used as a control to identify compounds present in the HT by chromatography (Fig. 3A). In this extract, the target compounds, XN and bitter acids, showed significant peaks. However, the DES extracts showed no peaks corresponding to bitter acids, suggesting a pronounced effect of extraction selectivity. In the chromatogram (Fig. 3A), two unidentified peaks were observed between 20 and 24 min. To address this, a phytochemical identification strategy was implemented using an UHPLC-PDA/MS-ESI, which served as a scanning method to determine the mass-to-charge ratio (m/z). To identify the unknown compounds, an extensive inventory of molecules commonly found in hops and beer products was compiled based on the studies of Wannenmacher et al.,29 and Bravi et al.30 Table S1 presents the twenty-nine molecular masses used, including the respective names, precise relative molecular masses (m/z; M+H]+), and corresponding molecular formulae. The runs 01 (−1; −1), 10 (0, 0 – central point), and 04 (+1, +1) of experimental design for the two extraction steps (DES and ethanol) were conducted for this examination.In general, the extraction patterns of phytochemicals were mainly influenced by temperature and solvent, as indicated by the examination of total ion current chromatograms in Fig. S1–S6. The traces of tyrosol, quercetin/morin, and coumaric acid derivatives were only noticed in the DES extraction at the minimum point. This result suggests that DESs modified the raw material during treatments at temperatures above 30 °C, potentially leading to its degradation. On the other hand, increasing the temperature facilitated the extraction of ferulic and sinapic acids in both extraction steps. The concentrations of both compounds increased by approximately 11% when operated at 50 °C with DES as the solvent (the maximum point) compared to the treatment at 40 °C (the central point). Furthermore, the temperature also promoted increased extraction of chlorogenic acid in DES environments.
DES extracts obtained in this work also comprised phytochemicals with molecular masses corresponding to caffeic acid, formononetin, biochanin A/glycitein, apigenin/genistein, naringenin, myricetin, isocohumulone, and desmethylxanthohumol, along with XN and bitter acid traces that had already been identified. Moreover, traces of daidzein, kaempferol/luteolin, epicatechin/catechin, hesperetin, postlupulone, and naringin were also detected in the DES extracts, as shown in Fig. S1, S2, and S3. The subsequent ethanolic extract presented daidzein, apigenin/genistein, naringenin, biochanin A/glycitein, epicatechin/catechin, quercetin/morin, hesperetin, gallocatechin, myricetin, isocohumolune, desmethylxanthohumol, and chlorogenic acid. These compounds exhibited poor solubility in DES and remained in the raw material after the first extraction stage. Notably, gallocatechin was the only phytochemical found exclusively in ethanol extracts. Traces of postlupulone, naringin, epicatechin/catechin, formononetin, and kaempferol/luteolin were detected in ethanolic extracts. Based on the reported phytochemical profiles, DES extracts contained higher levels of phenolic acids than ethanolic treatments. Moreover, unlike studies that employ single organic solvents (e.g., methanol, ethanol, or chloroform), DESs were able to extract compounds not previously reported for HT, such as desmethylxanthohumol and chlorogenic acid.3,31 This compositional difference conferred superior antioxidant capacity to the DES extracts, as discussed in the following sections.
Furthermore, desmethylxanthohumol was identified as a previously unreported compound in DES extracts, as shown in Fig. 3, probably due to its higher solubility in aqueous media. The enhanced extraction performance of DES is related to its physicochemical properties, particularly its capacity to form hydrogen-bonding networks that promote interactions with more hydrophilic phenolic compounds.19,32 Grudniewska and Popłoński18 also reported the presence of an unknown compound in DES extracts of spent hops, later identified as desmethylxanthohumol. In addition, it showed that temperature affected its concentration, resulting in an approximately 30% increase, as indicated by the total ion current chromatograms in Fig. S1 and S2.
3.2. Experimental design of polyphenol extraction via DES
The DES extractions were selective for XN, as the chromatograms of these extracts did not show the bitter acid peaks observed in the ethanolic extract (Fig. 3A and B). The results of the experimental design of the DES extraction step are shown in Table 2. The XN range varied from 5.27 (run 07) to 36.23 (run 04) µg g−1 of dried HT (Y1), and the antioxidant capacity from 14.42 (run 05) to 18.91 (run 04) mmol Trolox eq. kg−1 of dried HT (Y2). Both models (Y1 and Y2) indicated that curvature was not statistically significant (p ≥ 0.05). Therefore, first-order models without axial points are enough to describe the experimental data. The models constructed by a multiple regression analysis of the experimental data are shown below (eqn (3) and (4)):| Y1 = 20.57 + 4.21X1 + 10.79X2 | (3) |
| Y2 = 15.88 + 1.34X1 + 0.90X1X2 | (4) |
| Trials | Independent variables | Responses | Relative deviationc (%) | |||||
|---|---|---|---|---|---|---|---|---|
| Temperature (°C) | DES mass ratio(g) | XN (Y1) | FRAP (Y2) | XN | FRAP | |||
| µg XN g−1 of dried HTa | mmol Trolox eq. kg−1 of dried HTa | |||||||
a Mass of HT in dried weight.b HT mass is equal for all trials.c  |
||||||||
| 01 | 30.0 | (−1) | 4.00 | (−1) | 6.2 | 15.2 | 9.7 | 1.9 |
| 02 | 30.0 | (−1) | 8.00 | (+1) | 25.9 | 14.4 | 4.9 | 5.5 |
| 03 | 50.0 | (+1) | 4.00 | (−1) | 12.7 | 16.0 | 10.0 | 1.8 |
| 04 | 50.0 | (+1) | 8.00 | (+1) | 36.2 | 18.9 | 1.8 | 4.2 |
| 05 | 25.9 | (−1.41) | 6.00 | (0) | 23.8 | 14.4 | — | — |
| 06 | 54.1 | (+1.41) | 6.00 | (0) | 22.6 | 16.5 | — | — |
| 07 | 40.0 | (0) | 3.18 | (−1.41) | 5.3 | 15.2 | — | — |
| 08 | 40.0 | (0) | 8.82 | (+1.41) | 34.8 | 14.8 | — | — |
| 09 | 40.0 | (0) | 6.00 | (0) | 18.9 | 15.2 | 8.9 | 4.9 |
| 10 | 40.0 | (0) | 6.00 | (0) | 20.4 | 15.9 | 1.0 | 0.1 |
| 11 | 40.0 | (0) | 6.00 | (0) | 23.7 | 15.6 | 13.2 | 1.8 |
| Mean | 7.2 | 2.9 | ||||||
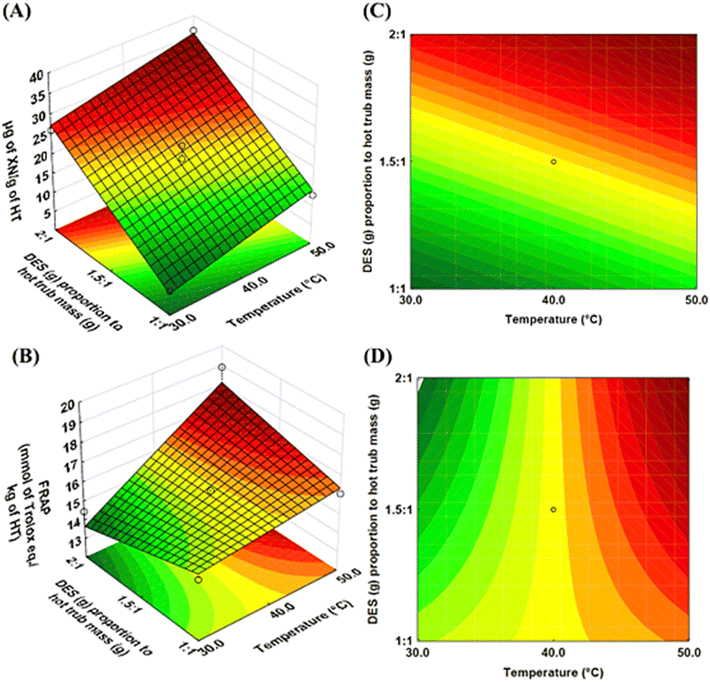
The results of ANOVA and the F-test with a significance of 0.05 are shown in Table S2. The good fit of the models to the experimental data is confirmed by the low average relative deviations of 7.20% and 2.87%, and the high R2 values of 0.97 and 0.84 for Y1 and Y2, respectively. Models were statistically significant, MQReg/MQRe > Ftable, and did not exhibit a lack of fit because MQLF/MQPE < Ftable, and the p-value for the lack of fit was greater than 0.05. The response surfaces and contour curves were constructed from these first-order polynomial models, as shown in Fig. 4.
Fig. 4 shows a direct correlation between temperature (X1) and both responses, XN content (Y1) and antioxidant capacity (Y2), i.e., increasing temperature increases both responses. The positive signs of the linear coefficients for variable X1 in both models (eqn (3) and (4)) indicate a positive effect of this variable on both responses. The increase in temperature promotes a decrease in solvent viscosity and solvent surface tension during the extraction of phytochemicals, allowing the improvement of solvent permeation in the sample matrices and resulting in a higher transfer rate.33 Additionally, chemical interactions, such as hydrogen bonds, van der Waals forces, and electrostatic interactions, are the main factors that retain phytochemicals within the biomass matrix. As the temperature increases, these chemical interactions weaken, which enhances the extraction of the desired compounds from the biomass matrix into the solvents.32 In the literature, the same pattern was observed by Grudniewska and Popłoński18 working with XN extraction from spent hops using four different DES mixtures. The authors reported that temperatures between 40 and 60 °C increased the XN content. However, above 60 °C, the XN content started to decrease, indicating that using higher temperatures can lead to its degradation.18
Regarding the DES-to-HT mass ratio (X2), it also yielded a positive linear coefficient for XN content (Y2), as shown in Fig. 4 and eqn (3). The findings suggest that XN extraction is enhanced with increasing DES proportion. Conversely, the water content adversely affects the XN extraction, which in this work ranged from 30 to 44% (w/w) (data not shown), from the minimum (−1) to the maximum (+1) point of the experimental design. It is worth mentioning that the modulation of water content occurred indirectly; for instance, it was increased or even decreased by the growth of the DES ratio. This approach was chosen because the DES mixture naturally absorbs water from its environment, making the method more practical by eliminating the need for additional pre-treatments of raw materials, such as oven drying or freeze-drying, thereby reducing overall processing time and costs.34
The water ratio in the DES mixture plays a key role as it significantly influences the solvent's physicochemical properties, such as viscosity, conductivity, and polarity, which in turn affect its extraction capacity.35,36 In our approach, the water content used is low compared to other studies in the literature, where it usually ranges from 50 to 80%.37,38 This means that XN did not dissolve well in a high-polarity environment. A similar pattern was observed in our previous study on XN extraction from HT using hydroalcoholic mixtures as solvents, where high-polarity treatments (with water content above 45% in ethanol) were less effective, or even ineffective.4 In this sense, Metaj et al.39 explored several organic solvents and DES mixtures in order to understand the relationship between the hydrophobicity of bitter acids and XN from hop cones and the characteristic properties of these solvents. Their findings indicate that XN dissolves more effectively in low-polarity, low-viscosity environments, such as diethyl ether and ChCl: phenol. Therefore, to maximize XN extraction from HT using the DES approach, it may be necessary to adjust polarity and other physicochemical properties, such as viscosity.
On the other hand, the antioxidant capacity values did not show a significant effect of increasing DES proportion alone. However, when both DES proportion and temperature were increased simultaneously, antioxidant capacity increased significantly, as shown in Fig. 3B. This effect occurred because temperature alters chemical interactions and intensifies the interface between the solvent and the solute, thereby enhancing mass transfer from biomass to DES.39 Additionally, increased temperature reduces DES viscosity, which, combined with a higher solvent proportion, can improve solute diffusion and enhance the mobility of a wide range of phenolic compounds and other phytochemicals within the solvent 25. Consequently, this leads to a higher rate of polyphenol transfer. Similar results were reported by Athanasiadis et al.,40 who observed that temperature and DES proportion modulated the extraction of total phenolic compounds from peppermint. Additionally, as seen in RSM (Fig. 4), the increase in antioxidant capacity did not correlate with the pattern of increased XN content.
Based on the results, we can infer that according to the CCRD, the optimized conditions were a temperature of 50 °C (X1; +1) and 2 parts of DES to 1 part of HT ratio mass (X2; +1). Under these conditions, the highest yield of XN and significant antioxidant capacity were achieved. Furthermore, the predicted model demonstrated a strong ability to accurately describe the experimental data, with low relative deviations (XN at 1.8 and antioxidant capacity at 4.2) under the optimized conditions (+1; +1).
3.3. Ethanol-based subsequent extraction from experimental design and ethanol concentration scan
The subsequent ethanol extraction recovered the remaining polyphenols and bitter acids that DES had not extracted. Table 3 displays the composition of XN and bitter acids, as well as the antioxidant capacity evaluated by the FRAP method. The ethanol step yielded a higher XN content than that obtained via DES extraction, ranging from 69.1 ± 2.8 to 113.9 ± 1.1 µg g−1 of dried HT. The highest XN content achieved in this step was correlated with the previous DES extractions at lower DES mass levels (−1), particularly in runs 1, 3, and 7. This result was expected, as the first extraction step using DES yielded a low extraction efficiency under the extraction conditions of runs 1, 3, and 7 (Fig. 4 and Table 2). This result indicated that minimizing XN transfer to the solvent during the DES step was essential for the compound to remain in the solid residue, which can then be effectively extracted during the ethanol step. A low XN solubility in DES mixtures (1 ChCl: 2 glycerol; 1 ChCl: 2 propylene glycol; 1 ChCl: 2 ethylene glycol; 1 ChCl: 2 Lac) was also noted by Grudniewska and Popłoński.18 They reported that organic solvents extracted almost 6 times more XN than DES mixtures, attributing this result to their poor solubility in polar systems.| Trials | Cohumulone (µg g−1 of dried HTb) | N-+Adhumulone (µg g−1 of dried HTb) | Colupulone (µg g−1 of dried HTb) | N-+Adlupulone (µg g−1 of dried HTb) | Xanthohumol (µg g−1 of dried HTb) | FRAP (mmol Trolox eq. kg−1 of dried HTb) |
|---|---|---|---|---|---|---|
| a CCRD – central composite rotatable design; HT – hot trub; FRAP – ferric reducing antioxidant power.b Mass of HT in dried weight. Different letters in the same column indicate statistical distinctions (p-value ≤0.05). Mean (n = 3) ± standard deviation of three independent experiments. | ||||||
| 01 | 82.3 ± 1.0 | 329.9 ± 4.3 | 723.8 ± 14.6 | 1012.5 ± 12.7 | 107.0 ± 0.7b | 10.3 ± 0.2bcde |
| 02 | 63.9 ± 2.4 | 327.9 ± 3.8 | 556.2 ± 14.0 | 822.4 ± 19.2 | 84.4 ± 0.6d | 9.4 ± 0.2e |
| 03 | — | — | — | — | 113.9 ± 1.1a | 12.1 ± 0.1a |
| 04 | — | — | — | — | 70.1 ± 0.5e | 9.7 ± 0.2de |
| 05 | 48.2 ± 1.5 | 219.4 ± 2.7 | 534.5 ± 6.4 | 802.1 ± 11.1 | 92.4 ± 1.5c | 10.8 ± 0.1b |
| 06 | — | — | — | — | 92.9 ± 0.8c | 10.6 ± 0.5bc |
| 07 | — | — | — | — | 107.2 ± 0.4b | 10.7 ± 0.3b |
| 08 | — | — | — | — | 69.1 ± 2.8e | 9.7 ± 0.1cde |
| 09 | — | — | — | — | 84.7 ± 2.5d | 10.3 ± 0.2bcde |
| 10 | — | — | — | — | 84.1 ± 1.9d | 10.4 ± 0.6bcd |
| 11 | — | — | — | — | 84.3 ± 3.0d | 10.3 ± 0.5bcde |
Regarding antioxidant capacity, the ethanol extracts showed a 60% reduction compared to DES extraction, suggesting that this solvent has greater extraction efficiency for a wide range of phytochemicals. Furthermore, these findings suggest that residual phytochemicals in the biomass matrix were minimal across all conditions tested in the DES step. In the studies conducted by Chagnoleau et al.41 (2 betaine: 3 citric acid, 1 ChCl: 1 malonic acid, 1 betaine hydrochloride: 10 ethylene glycol) and Lee et al.42 (2 glycerol: 1 betaine; 3 glycerol: 1 betaine; 2 glycerol: 1 D-sorbitol; 3 glycerol: 1 D-sorbitol; 3 glycerol: 1 xylose; 3 glycerol: 1 glucose; 3 glycerol: 1 fructose; 1 glycerol: 1 urea), it was reported that DES mixtures produced extracts with significantly higher antioxidant capacity than traditional solvents, like ethanol and methanol.
Despite the strong antioxidant capacity of DESs, this study focused on identifying the method that maximizes XN extraction. Therefore, based on the high XN content extracted in step 2, run 1 (−1; −1) was selected as the best result. Additionally, the extracts obtained with ethanol presented a high content of bitter acids, which are the majority. The concentrations varied from 48.2 ± 1.5 to 1012.5 ± 12.7 µg g−1 of dried HT, corresponding to cohumulone and n+adlupulone. The bitter acids were detected only in ethanol extracts obtained from biomass treated with DES at 30 °C or lower. The presence of bitter acids in DES extracts was observed by Macchioni et al.,19 who applied Lac-based mixtures at room temperature to dried hops cones. On the other hand, Grudniewska and Popłoński18 did not report the presence of bitter acids in their study, which used a 60 °C extraction temperature. Therefore, higher temperatures (>30 °C) used in the DES step may have promoted the thermal degradation of bitter acids. Thus, DESs associated with temperature can be used to modulate the phytochemical composition of the extract.
Once the condition for DES extraction was determined for the point minimum of XN extraction (−1; −1), the approach of reducing ethanol concentration was applied, seeking to evaluate how the phytochemicals are affected by the increase in polarity in step 2. The ethanol concentrations ranged from 40 to 100% (v/v), with n-+adlupulone and cohumulone exhibiting the highest and the lowest concentrations, 1225.4 ± 2 µg g−1 of dried HT and 23.5 ± 2.2 µg g−1 of dried HT, respectively, as shown in Table 3. The maximum concentration of XN was achieved at 70% (v/v) ethanol, with a value of 108 ± 1 µg g−1 of dried HT, indicating no negative effect on increasing polarity by around 30%. However, this increase in polarity significantly affects antioxidant capacity, suggesting that the major phytochemicals responsible for it were not soluble in a 70% ethanolic solution.
3.4. Effect of the mixing strategy on extraction yield and solid residue
The mixing strategy was investigated to assess the reproducibility and extraction efficiency of two different mixing methods (magnetic mixing and thermo-shaking). For this purpose, the minimum conditions for the DES (−1; −1) and the ethanolic extraction at 70% (v/v) were utilized. The extract produced by thermo-shaking was significantly higher (p-value ≤0.05) in terms of XN content and bitter acid composition when compared to that obtained via magnetic mixers involving both extraction steps, as displayed in Table 4. Regarding antioxidant capacity, no significant differences were observed between the two mixing strategies from the DES step. However, an 8.5% decline in antioxidant capacity was observed for the magnetic mixer compared with ethanolic treatments.| EtOH%b | Cohumulone (µg g−1 of dried HT) | N-+Adhumulone (µg g−1 of dried HT) | Colupulone (µg g−1 of dried HT) | N-+Adlupulone (µg g−1 of dried HT) | Xanthohumol (µg g−1 of dried HT) | FRAP (mmol Trolox eq. kg−1 of dried HT) |
|---|---|---|---|---|---|---|
| a Mass of HT in dried weight; HT – hot trub; FRAP – ferric reducing antioxidant power.b EtOH% – ethanol concentration% (v/v). Different letters in the same column indicate statistical distinctions (p-value ≤0.05). Mean (n = 3) ± standard deviation of three independent experiments. | ||||||
| 40 | 23.5 ± 2.2c | 56.8 ± 5.1d | 39.7 ± 4.1d | 28.1 ± 3.6d | 34.4 ± 2.6d | 2.8 ± 0.3d |
| 55 | 91.9 ± 6.9b | 279.6 ± 7.1c | 557.8 ± 10c | 682.3 ± 16.9c | 88 ± 3c | 3.9 ± 0.3c |
| 70 | 125.5 ± 4.2a | 371.7 ± 9.1a | 893 ± 12.7a | 1225.4 ± 23a | 108 ± 1a | 4.3 ± 0.1b |
| 100 | 96.3 ± 10.7b | 309.6 ± 5.2b | 671 ± 17.7b | 956.5 ± 24.3b | 103.8 ± 2.4b | 10.3 ± 0.2a |
| Thermo shaking (extraction 1) | — | — | — | — | 11.3 ± 0.4a | 9.8 ± 0.2a |
| Magnetic mixer (extraction 1) | — | — | — | — | 9.6 ± 0.9b | 9.8 ± 0.7a |
| Thermo shaking (extraction 2) | 125.5 ± 4.2a | 371.7 ± 9.1a | 893 ± 12.7a | 1225.4 ± 23a | 108 ± 1.2a | 4.3 ± 0.1b |
| Magnetic mixer (extraction 2) | 86.8 ± 10.1b | 343.7 ± 30.3b | 624.7 ± 66.3b | 851.1 ± 93.4b | 91.2 ± 7.8b | 4.7 ± 0.3a |
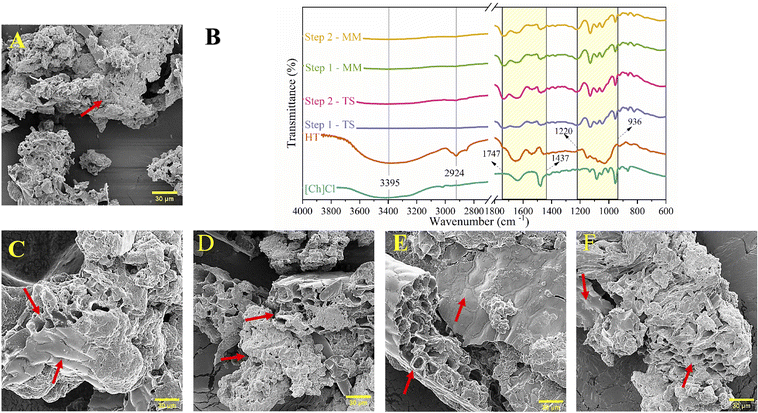
The impact of two mixer types on a solid raw material was also evaluated using SEM and FTIR, as presented in Fig. 5. The dried HT used as a control sample (Fig. 5A) displayed a particulate microstructure and a rough surface, with only small pieces of spent hops. After the extraction processes, the solid residues appeared more like pellets than particles, with an internal microstructure resembling a heap of cylindrical holes. In addition, the DES extractions (Fig. 5C and E) resulted in a smoother surface than the ethanolic treatments, indicating that the solvents remained in the raw material.
No differences in the shape of the microstructure of the solid residue were observed between the two mixing processes, indicating that neither promoted maceration of the raw material during extraction. It is worth noting that the solid residue exhibited distinct characteristics when compared between the DES and ethanolic extractions. Ethanolic treatments produced a more porous, fragmented structure than DES-treated samples, suggesting better dissolution of phytochemicals by the solvent. Ethanol extraction is not typically recognized for promoting expressive fragmentation during the process. However, DES solvents can induce biomass fragmentation. Huang et al.43 highlighted the efficacy of DES in drawing out phytochemicals (crocins) by plant cell rupture, leading to fragmentation. They did not observe this damage when they performed the extraction in 50% ethanol. De Almeida Pontes et al.26 also reported the expressive damage caused by DESs on olive leaves, and the same pattern was not noted for the extraction samples with ethanol. Thus, the fragmentation observed in the ethanol treatments can be attributed to the previous extraction with DESs, which was carried over to the subsequent steps.
Regarding the FTIR findings (Fig. 5B), the solid residue was compared to the spectra of HT and ChCl to detect any modifications. The HT spectrum exhibited the presence of vibration bands at around 1584, 1490, 1182, 1133 (deep valley), and 940 cm−1, corresponding to C–N stretching, N1H bending, and N2H bending; C–O–C stretching; C–O stretching, and CO2H bending on the plane, respectively,44,45 while the ChCl spectrum displayed a band near 1480 cm−1, referring to a specific vibrational mode of the CH2 group bending in an angular deformation as a scissor.26 After the extraction steps, significant modifications were observed in the solid residues, particularly in shifts and the disappearance of bands associated with characteristic functional groups of the raw material. Additionally, no differences were observed in the FTIR spectral profiles among the samples.
The most expressive alterations in solid residues were related to the alcohol and hydroxyl (–OH) functional groups, initially located at 1130 (C–O stretching) and 3395 cm−1 (OH– stretching).46 In both situations, the extraction process resulted in a decrease in intensity and the disappearance of bands, leading to the partial or complete removal of compounds containing these functional groups. Furthermore, the DES was the primary cause of biomass modification, as the subsequent ethanol extraction did not show significant changes in the FTIR spectra. The disappearance of the band at 3395 cm−1, which corresponds to intermolecular hydrogen bonds, clearly indicates that DES has caused some structural changes.
Another indication of these structural changes in solid residues was the appearance of a new band at 1700 cm−1, revealing the presence of the amide I functional group. Moreover, a decrease in intensity was observed in peaks associated with nitrogenous compounds in the spectral range from 1437 to 1747 cm−1. The appearance of the amide I signal suggests that the bonds connecting proteins to phytochemicals were cleaved during DES extraction. It is worth noting that the extraction process involving DESs was conducted at a controlled temperature not exceeding 30 °C. Then, any observed effects can be attributed exclusively to the properties inherent to the DES used in the experiment.
Therefore, the study of the mixing strategy showed that the two approaches did not exhibit significant differences in the microstructure and functional group profiles of the solid residues. The XN content and antioxidant capacity were affected by less than 10%, indicating good reproducibility. Moreover, the solid residue fragmentation and disappearance of functional groups corresponding to alcohol demonstrate that phytochemicals were effectively extracted from biomass. This protein-rich solid residue can be repurposed as a food and pharmaceutical ingredient.
3.5. Sustainability evaluation by Path2Green
The evaluation of XN, bitter acids, and other phenolic compounds extracted from hot trub using the sequential extraction process proposed in this study, assessed through the Path2Green metric, provides a comprehensive perspective on its sustainability (Table S3). In summary, the process received a positive score of +0.592 (from −1 to 1), as indicated by the pictogram in Fig. 6, indicating alignment with green chemistry and circular bioeconomy principles.The use of HT as a raw material strongly contributes to sustainability (Principle 1: biomass choice) by valorizing an agro-industrial by-product and enabling the production of new high-added-value products. This valorization aligns well with circular economy strategies and received the highest score in the Path2Green assessment. Moreover, the use of non-treatment for subsequent extraction reduces the energy required for the process, earning a +1 in Principle 3. The process achieved exhaustive recovery of phytochemicals using solvents of renewable origin, highlighting a positive contribution to overall sustainability. Although the DES is inherently a higher-cost solvent, it enabled enhanced extraction selectivity, which may be advantageous for specific applications of XN. In addition, the DES based on ChCl and La combined with XN can be directly applied in several formulations and applications, such as active packaging materials.47 In contrast, the use of ethanol as an extraction solvent is more complex, as it is volatile and must be removed from the environment before the phytochemicals are applied. However, ethanol recovery systems, such as vacuum rotary evaporation, can be employed to minimize industrial waste and reduce expenses associated with solvent consumption, which typically represents one of the highest cost contributors in economic assessments of extractive processes.48 The score of each principle also reveals areas that merit improvement to enhance its overall sustainability. The process was carried out in batch mode, which makes it challenging to scale up. Additionally, it generated approximately 13% biomass residue. These points could be improved in the process to further enhance its sustainability, including adopting a process for converting residual biomass into a protein ingredient.
4. Conclusion
The DES based on a mixture of ChCl and Lac as a single solvent proved highly efficient for the recovery of very hydrophilic compounds in HT, such as chlorogenic acid and desmethylxanthohumol, although it was not the most effective solvent for the target compound (XN). Despite this limitation, the DES should not be considered unsuitable, as it plays a significant role in irreversibly modifying interactions and bonds between macromolecules and micromolecules. This pattern thereby enabled the extraction of a wide range of phytochemicals. Then, using DES as a pretreatment to increase selectivity during subsequent extraction is very useful, and different solvent-to-raw-material ratios and temperatures prompt distinct responses in phytochemical composition. It is important to emphasize that the DES chosen achieved a goal in avoiding the coextraction of bitter acids and XN. From a sustainability perspective, the proposed two-step extraction approach aligns with green chemistry principles and circular bioeconomy concepts by reducing the use of conventional organic solvents. Our work contributes to SDG 12 (Responsible Consumption and Production) and SDG 15 (Life on Land), by promoting efficient methods to reduce waste and minimize environmental impact by recycling brewery by-products into food ingredients. The favorable Path2Green assessment further confirms the environmental advantages of integrating DESs into brewery by-product processing chains. The solid residue obtained after DES pretreatment is an underexplored resource with preserved protein content, highlighting HT as a promising source of plant-based proteins. Further research is required to investigate protein extraction strategies and their technological functionality, and evaluate the economic feasibility of industrial implementation for HT valorization in hop-derived product manufacturing.Author contributions
Klycia Fidélis Cerqueira e Silva: writing – original draft, conceptualization, methodology, and investigation. Paula Virginia de Almeida Pontes: methodology and writing – original draft. Patrícia Tonon de Souza: writing – review & editing and investigation. Monique Martins Strieder: formal analysis and writing – review & editing. Eduardo Augusto Caldas Batista: writing – review & editing and supervision. Miriam Hubinger: writing – review & editing, supervision, and funding acquisition.Conflicts of interest
The authors declare no competing interests.Data availability
All data supporting the findings of this study are included in the article and supplementary information (SI). No additional data are available. Raw data can be obtained from the corresponding author upon request. The final, accepted version of the manuscript will be uploaded to the REDU platform (https://redu.unicamp.br/), an institutional repository of the University of Campinas (UNICAMP).Supplementary information is available. See DOI: https://doi.org/10.1039/d5fb00820d.
Acknowledgements
The authors are grateful to FAPESP (2019/27354-3; 2014/21252-0; 2021/12264-9), CAPES with Finance Code 001, and CNPq (428644/2018-0; 309022/2021-5; 426913/2018-3) for the financial and fellowship support.References
- BarthHaas, Report 2021/2022, Nuremberg, 2023.
- A. Karlović, A. Jurić, N. Ćorić, K. Habschied, V. Krstanović and K. Mastanjević, Fermentation, 2020, 6, 82 CrossRef.
- F. Senna Ferreira Costa, T. Roquete Amparo, J. Brandão Seibert, B. M. Silveira, R. Gomes da Silva, D. Inocêncio Pereira, R. Gontijo Garcia Barbosa, O. D. H. dos Santos, G. C. Brandão, L. F. de Medeiros Teixeira, P. Melo de Abreu Vieira and G. H. Bianco de Souza, Waste Biomass Valorization, 2021, 12, 2037–2047 CrossRef CAS.
- K. F. C. e. Silva, G. Feltre, F. S. Zandonadi, R. S. Rabelo, A. Sussulini and M. D. Hubinger, J. Sci. Food Agric., 2024, 104, 5381–5390 CrossRef CAS PubMed.
- I. Martín, C. López, J. García-González and S. Mateo, J. Environ. Manage., 2024, 367, 121969 CrossRef PubMed.
- R. Durello, L. Silva and S. Bogusz Jr, Quim. Nova, 2019, 42, 900–919 CAS.
- J. F. Stevens, in Natural Products for Cancer Chemoprevention: Single Compounds and Combinations, Springer International Publishing, Cham, 2020, pp. 320–350 Search PubMed.
- M. Biendl, Acta Hortic., 2013, 131–140 CrossRef.
- M. A. R. Martins, S. P. Pinho and J. A. P. Coutinho, J. Solution Chem., 2019, 48, 962–982 CrossRef CAS.
- D. O. Abranches and J. A. P. Coutinho, Annu. Rev. Chem. Biomol. Eng., 2023, 14, 141–163 CrossRef CAS PubMed.
- A. P. Abbott, G. Capper, D. L. Davies, R. K. Rasheed and V. Tambyrajah, Chem. Commun., 2003, 70–71 RSC.
- A. P. Abbott, D. Boothby, G. Capper, D. L. Davies and R. K. Rasheed, J. Am. Chem. Soc., 2004, 126, 9142–9147 CrossRef CAS PubMed.
- E. A. Crespo, J. M. L. Costa, A. M. Palma, B. Soares, M. C. Martín, J. J. Segovia, P. J. Carvalho and J. A. P. Coutinho, Fluid Phase Equilib., 2019, 500, 112249 CrossRef CAS.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Chem. Rev., 2014, 114, 11060–11082 CrossRef CAS PubMed.
- Md. A. Alam, G. Muhammad, M. N. Khan, M. Mofijur, Y. Lv, W. Xiong and J. Xu, J. Clean. Prod., 2021, 309, 127445 CrossRef CAS.
- M. H. Zainal-Abidin, M. Hayyan, A. Hayyan and N. S. Jayakumar, Anal. Chim. Acta, 2017, 979, 1–23 CrossRef CAS PubMed.
- W. Volpato Maroldi, I. de Andrade Arruda Fernandes, B. Demczuk Junior, A. Cristina Pedro, G. Maria Maciel and C. Windson Isidoro Haminiuk, Bioresour. Technol., 2024, 413, 131447 CrossRef CAS PubMed.
- A. Grudniewska and J. Popłoński, Sep. Purif. Technol., 2020, 250, 117196 CrossRef CAS.
- V. Macchioni, K. Carbone, A. Cataldo, R. Fraschini and S. Bellucci, Sep. Purif. Technol., 2021, 264, 118039 CrossRef CAS.
- F. S. Bragagnolo, M. M. Strieder, R. S. Pizani, L. M. de Souza Mesquita, M. González-Miquel and M. A. Rostagno, TrAC, Trends Anal. Chem., 2024, 175, 117726 CrossRef CAS.
- B. M. Popović, N. Gligorijević, S. Aranđelović, A. C. Macedo, T. Jurić, D. Uka, K. Mocko-Blažek and A. T. Serra, RSC Adv., 2023, 13, 3520–3527 RSC.
- A. Mišan and M. Pojić, Adv. Bot. Res., 2021, 97, 333–359 CrossRef.
- M. A. Bezerra, R. E. Santelli, E. P. Oliveira, L. S. Villar and L. A. Escaleira, Talanta, 2008, 76, 965–977 CrossRef CAS PubMed.
- L. M. de Souza Mesquita, L. S. Contieri, F. A. e Silva, R. H. Bagini, F. S. Bragagnolo, M. M. Strieder, F. H. B. Sosa, N. Schaeffer, M. G. Freire, S. P. M. Ventura, J. A. P. Coutinho and M. A. Rostagno, Green Chem., 2024, 26, 10087–10106 RSC.
- W. H. Zhang, M. N. Chen, Y. Hao, X. Jiang, X. L. Zhou and Z. H. Zhang, J. Mol. Liq., 2019, 278, 124–129 CrossRef CAS.
- P. V. de Almeida Pontes, I. Ayumi Shiwaku, G. J. Maximo and E. A. Caldas Batista, Food Chem., 2021, 352, 129346 CrossRef CAS PubMed.
- J. De Keukeleire, G. Ooms, A. Heyerick, I. Roldan-Ruiz, E. Van Bockstaele and D. De Keukeleire, J. Agric. Food Chem., 2003, 51, 4436–4441 CrossRef CAS PubMed.
- I. F. F. Benzie and J. J. Strain, Anal. Biochem., 1996, 239, 70–76 CrossRef CAS PubMed.
- J. Wannenmacher, M. Gastl and T. Becker, Compr. Rev. Food Sci. Food Saf., 2018, 17, 953–988 CrossRef CAS PubMed.
- E. Bravi, G. De Francesco, V. Sileoni, G. Perretti, F. Galgano and O. Marconi, Antioxidants, 2021, 10, 165 CrossRef CAS PubMed.
- P. Scognamiglio, V. Carlucci, N. Benedetto, L. Milella, R. Teta, G. Esposito and V. Costantino, Food Chem., 2026, 505, 147977 CrossRef CAS PubMed.
- Q. Yu, F. Wang, Y. Jian, V. M. Chernyshev, Y. Zhang, Z. Wang, Z. Yuan and X. Chen, J. Mol. Liq., 2022, 363, 119848 CrossRef CAS.
- J. Dai and R. J. Mumper, Molecules, 2010, 15, 7313–7352 CrossRef CAS PubMed.
- S. O. Tebbi, N. Debbache-Benaida, N. Kadri, R. Kadi and S. Zaidi, Sustainable Chem. Pharm., 2023, 31, 100933 CrossRef CAS.
- X.-J. Zhang, Z.-T. Liu, X.-Q. Chen, T.-T. Zhang and Y. Zhang, Food Chem., 2023, 422, 136224 CrossRef CAS PubMed.
- M. Vilková, J. Płotka-Wasylka and V. Andruch, J. Mol. Liq., 2020, 304, 112747 CrossRef.
- S. Taweekayujan, S. Somngam and T. Pinnarat, Heliyon, 2023, 9, e17942 CrossRef CAS PubMed.
- A. Bermúdez-Oria, A. Fernández-Prior, M. Luisa Castejón, G. Rodríguez-Gutiérrez and J. Fernández-Bolaños, Food Chem., 2023, 419, 136073 CrossRef PubMed.
- I. Metaj, D. Hajdini, K. Gliha, I. J. Košir, M. Ocvirk, M. Kolar and J. Cerar, Plants, 2023, 12, 2890 CrossRef CAS PubMed.
- V. Athanasiadis, D. Palaiogiannis, S. Grigorakis, E. Bozinou, S. I. Lalas and D. P. Makris, J. Appl. Res. Med. Aromat. Plants, 2023, 33, 100456 CAS.
- J.-B. Chagnoleau, A. M. Ferreira, J. A. P. Coutinho, X. Fernandez, S. Azoulay and N. Papaiconomou, Food Chem., 2023, 401, 133992 CrossRef CAS PubMed.
- S. Y. Lee, Y. N. Liang, D. C. Stuckey and X. Hu, Sep. Purif. Technol., 2023, 317, 123677 CrossRef CAS.
- H. Huang, Y. Zhu, X. Fu, Y. Zou, Q. Li and Z. Luo, Food Chem., 2022, 380, 132216 CrossRef CAS PubMed.
- A. Barth, Biochim. Biophys. Acta, Bioenerg., 2007, 1767, 1073–1101 CrossRef CAS PubMed.
- J. Coates, in Encyclopedia of Analytical Chemistry, John Wiley & Sons, Ltd, 2006 Search PubMed.
- R. N. Oliveira, M. C. Mancini, F. C. S. de Oliveira, T. M. Passos, B. Quilty, R. M. da S. M. Thiré and G. B. McGuinness, Matéria, 2016, 21, 767–779 CAS.
- E. Jakubowska, M. Gierszewska, J. Nowaczyk and E. Olewnik-Kruszkowska, Carbohydr. Polym., 2021, 255, 117527 CrossRef CAS PubMed.
- M. M. Strieder, M. I. L. Neves, G. L. Zabot, E. K. Silva and M. A. A. Meireles, Sep. Purif. Technol., 2021, 258, 117978 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |