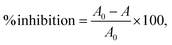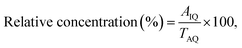Open Access Article
Open Access ArticleEnzyme-assisted extraction and characterization of red roselle calyx (Hibiscus sabdariffa L.) bioactives and their application in ice cream matrices
A. Priyankaa,
B. Nila Shireena,
S. Ganga Kishoreb,
Sudha Paramadhas*a,
R. Rahul c,
Madhuresh Dwivedi
c,
Madhuresh Dwivedi *b,
T. Pandiarajana,
R. Divyabharathia and
Nongmaithem Seema Chanub
*b,
T. Pandiarajana,
R. Divyabharathia and
Nongmaithem Seema Chanub
aDepartment of Food Process Engineering, Agricultural Engineering College & Research Institute, Tamil Nadu Agricultural University, Coimbatore-641 003, Tamil Nadu, India. E-mail: sudha.p@tnau.ac.in
bDepartment of Food Process Engineering, National Institute of Technology, Rourkela-769 008, Odisha, India. E-mail: madhureshd@gmail.com
cDepartment of Food Technology, Dhanalakshmi Srinivasan College of Engineering, Coimbatore-641 105, Tamil Nadu, India
First published on 25th February 2026
Abstract
Roselle calyx (Hibiscus sabdariffa Linn.) is widely recognized for its rich phenolic and anthocyanin contents, which contribute to its antioxidant properties, colour, and health-promoting benefits. However, conventional extraction methods often result in low yields and compound degradation. Therefore, this study explored enzyme-assisted extraction using amylase, pectinase, and cellulase individually to enhance the recovery of bioactive compounds from roselle calyces under varying enzyme concentrations and temperatures. The optimized condition obtained from response surface methodology revealed that cellulase (10%, 60 °C) produced the highest total phenolic (55.34 mg GAE/100 g) and flavonoid (193.05 mg CE/100 g) contents, pectinase (15%, 50 °C) yielded the highest anthocyanin content (127.79 mg/100 g), and amylase (3%, 60 °C) achieved the highest antioxidant activity (39.14%). The incorporation of enzyme-optimized roselle extracts into ice cream formulations improved the colour intensity, texture, and overall sensory appeal. Cellulase provided the most vibrant colour and dense structure, resulting in a smoother and more aerated product with balanced sweetness and a moderately light texture. HPLC analysis of cellulase-assisted roselle calyx extract showed peaks corresponding to quercetin, with a high relative concentration of 52.87%, indicating the efficient enzymatic release of this key flavonoid without any structural degradation. FTIR spectroscopic analysis confirmed the retention of key phenolic and carbohydrate-based functional groups. SEM analysis revealed that fine roselle fragments were uniformly embedded within the ice cream matrix, supporting a smoother texture and reduced ice crystal formation. Overall, enzyme-assisted extraction emerges as a sustainable and effective approach for producing roselle-based natural colourants.
Sustainability spotlightThis study highlights the sustainable potential of enzyme-assisted extraction (EAE) as an eco-friendly and efficient method for obtaining natural colorants and bioactive compounds from roselle (Hibiscus sabdariffa Linn.) calyces. By employing enzymes such as amylase, pectinase, and cellulase, the process significantly enhanced the extraction of anthocyanins, phenolics, and flavonoids, thereby improving the antioxidant activity and pigment yield. The optimized conditions, particularly with cellulase at 10% concentration and 60 °C, yielded the highest levels of total phenolic and flavonoid contents while maintaining compound stability. The enzyme-infused roselle bio-extract was further incorporated into ice cream formulations, resulting in improved colour intensity, texture, and sensory properties. Advanced characterization techniques such as FTIR spectroscopy and SEM confirmed the presence of polyphenols, flavonoids, and proteinaceous compounds, as well as enhanced microstructural properties that contributed to better texture and water retention. Overall, this study demonstrates that enzyme-assisted extraction is a sustainable and effective alternative to conventional methods for producing natural, stable, and functional food colorants, supporting their potential application in the food and nutraceutical industries as substitutes for synthetic additives. |
1 Introduction
The food industry across the world witnessed a rising shift in consumer needs towards natural and organic ingredients due to increasing health concerns and environmental hazards caused by synthetic colorants.1,2 Synthetic dyes like tartrazine, allura red, and sunset yellow, though stable and effective, have been reported to induce allergic reactions, hyperactivity, and carcinogenic effects.3 Consequently, the demand for plant-based bio-colorants witnessed active acceleration with increased focus on extraction efficiency, stability, and compatibility in food matrices.4 Among the plant-based alternatives, Hibiscus sabdariffa Linn., commonly known as roselle, gained significant attention due to its vibrant red calyces, which are rich in phenols.5 Anthocyanins not only impart color but also provide antioxidant, antimicrobial, and antihypertensive properties, making them ideal functional ingredients in food and nutraceutical formulations.6Roselle is widely cultivated in the tropical regions of Africa, Asia, and the Caribbean. The colour of the calyces determines the bioactive components present in them, especially their flavonoid contents.7,8 The calyces of Hibiscus sabdariffa (roselle) are complex matrices composed of not only basic nutrients (such as modest amounts of protein, fibre, carbohydrates, minerals, and organic acids) but also a rich suite of bioactive phytochemicals. Among the phenolic-type phytochemicals, calyces are especially notable for their high content of anthocyanin pigments, which confer the characteristic deep red colour of red-calyx varieties. In addition, a variety of other phenolic acids (e.g., chlorogenic, neochlorogenic, caffeic, gallic, and protocatechuic acids) and flavonoids (e.g., rutin, quercetin derivatives, and other glycosides) have been identified. Roselle calyces also contain organic acids (citric, malic, and hibiscus acid), some vitamins (notably ascorbic acid), and diet-relevant constituents such as dietary fibers. Importantly, many of these bioactive polyphenols are not freely extractable but are present in a “bound” form, often interacting with the plant cell wall matrix (polysaccharides, fibers, proteins, and other macromolecules) or forming complexes that affect their solubility, extractability, and stability. The actual yield and composition of bioactive compounds from calyces depend heavily on the extraction method, the solvent used, and the processing conditions, whether the aim is to recover pigments (anthocyanins), soluble phenolics, or more tightly associated bound phenolics. This bound fraction is crucial: it represents a reservoir of bioactive potential that can be released (or stabilized) depending on processing and contributes to the overall functional value of roselle calyces for antioxidant, anti-hyperglycemic, and other health-related effects.5,6,8,9
The principal anthocyanins, delphinidin-3-sambubioside and cyanidin-3-sambubioside, are found in the calyces that are responsible for the deep red color.9 Moreover, the calyces are rich in organic acids, flavonoids, ascorbic acid, and polysaccharides, contributing to corresponding sensory and therapeutic qualities.10 Despite their high pigment content, the large-scale utilization of roselle calyces as bio-colorants is limited by challenges in extraction efficiency and pigment stability during processing and storage.11 Anthocyanins are prone to thermal degradation, oxidative reactions, and enzymatic browning, which can result in potential color loss, specifically during conventional drying and solvent extraction.12 These bioactive compounds exist in bound forms, either conjugated to cell wall components or trapped within the matrix, limiting their extractability using conventional solvent methods. This rationale supports the use of enzyme-assisted extraction, as specific enzymes (e.g., cellulases and pectinases) can hydrolyse the cell wall or matrix components, releasing these bound bioactives more efficiently.13–15
Conventional solvent-based anthocyanin extraction methods, such as maceration and reflux, are energy-demanding and time-consuming, and may cause degradation of thermolabile compounds.16 These barriers have shifted the research focus to explore eco-friendly and efficient alternatives for pigment recovery from plant matrices. Traditional methods of anthocyanin extraction, mostly solvent-based techniques using ethanol,17 methanol,18 or acidified water,19 have been widely applied for retaining anthocyanins from roselle. These methods are often time-consuming, require high solvent usage, and may cause partial degradation of thermolabile pigments.20,21 Recent advancements in green extraction technologies such as ultrasound-assisted,22 microwave-assisted,23 and enzyme-assisted extraction24 have shown improved pigment recovery and stability with lower environmental impacts. Among these, enzyme-assisted extraction (EAE) has emerged as a promising technique due to its eco-friendly25 approach in specificity and the ability to disrupt plant cell walls for enhanced release of intracellular pigments.26 Enzymes like pectinase,13 cellulase,14 and hemicellulase15 can synergistically act and hydrolyse the polysaccharides of the calyces, which may lead to a higher pigment yield.13
Furthermore, EAE is considered a sustainable alternative due to its lower energy consumption,27 absence of toxic residues28 and the ability to maintain anthocyanin stability.29 The efficiency of EAE is influenced by extraction parameters including enzyme concentration, temperature, pH, and incubation time.30 Anthocyanins derived from roselle have proven applications in various food systems such as beverages, yoghurts, bakery products, and confectionery items, improving both visual appeal and antioxidant potential.31 However, the stability during storage and thermal processing is a critical concern.11 To overcome these challenges, the enzyme-assisted extraction technique could significantly enhance the pigment yield, stability, and functionality while maintaining product safety and sustainability.5 Therefore, the present study aims to evaluate the effect of enzyme-assisted extraction parameters on the recovery of bio-colorants from dried roselle (Hibiscus sabdariffa Linn.) calyces and their application in food systems. The research specifically focuses on optimizing the enzyme type, concentration, and process temperature to achieve the maximum pigment yield and stability, thereby contributing to the development of natural, safe, and sustainable colorant alternatives for the food industry.
2 Materials and methods
2.1. Chemicals, reagents, and equipment
All the reagents and solvents used were of analytical grade. The chemicals employed for extraction and analytical assays included hydrochloric acid (HCl), Folin–Ciocalteu reagent, sodium carbonate (20% Na2CO3), 95% methanol, pyrocatechol (stock solution), aluminium chloride (10% AlCl3), sodium hydroxide (1 M NaOH), sodium nitrate (5% NaNO2), and 2,2-diphenyl-1-picrylhydrazyl (DPPH). Enzymes such as amylase, cellulase, and pectinase were procured in powder form from HiMedia, Mumbai, India. The instruments used for extraction and analysis included a UV-visible spectrophotometer (UV-1800, Shimadzu, Japan), a digital pH meter, a Lovibond tintometer (Lovibond, LC100, The Tintometer Ltd, UK), a high-performance liquid chromatograph (HPLC; LC-6AD, Shimadzu, Japan), a color extraction unit, and an analytical balance.2.2. Preparation of the roselle bio-extract from the calyx portion
Fresh roselle (Hibiscus sabdariffa L.) calyces at 35 days post-bloom (Wong et al., 2003) were harvested from a farm in Pothur village (13.1602° N, 80.1512° E), near Chennai, Tamil Nadu, India. The calyces were manually separated from the buds using a hand tool, thoroughly washed to remove adhering soil and impurities, and tray-dried at 60 °C until the moisture content decreased from 86% to 11%.32 Approximately 5 kg of fresh calyces yielded about 1.5 kg of dried material after 48 h of drying. The tray-drying process took almost 2 days to completely remove the moisture content present in the calyces. The initial moisture content of the sample was 86%, and it was reduced to 11%.2.3. Experimental design for enzyme-assisted extraction
The experimental plan was developed by using varying combinations of independent variables: individual enzyme type (amylase, cellulase, and pectinase), enzyme concentration (1%, 5%, and 10% w/v), and temperature (50 °C, 60 °C, and 70 °C). Consequently, the dependent variables chosen were total phenolic content (TPC), total flavonoid content (TFC), total anthocyanin content (TAC), antioxidant activity (AOX), pH, and color parameters (L*, a*, b*, and hue angle h°) for analyzing the qualitative properties preserved across all experiments. The final best condition was obtained from the dependent variables of trial combinations. The experiments were carried out under ambient laboratory conditions. For each run, 500 g of pre-dried roselle calyces were mixed with distilled water in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (w/v) ratio in a borosilicate extraction vessel. This mixture was then heated in a thermostatically controlled water bath at temperatures of 50 °C, 60 °C, and 70 °C. Enzyme solutions with different concentrations as per the design matrix were prepared simultaneously. The enzyme solutions were equilibrated to their respective temperatures before mixing, ensuring uniform reaction conditions during bio-colorant extraction. Prepared enzyme solutions were then introduced to the vessels containing roselle calyces and a water mixture. The extraction was allowed to proceed under controlled independent variable conditions until completion. After extraction, the mixture was filtered through Whatman No. 1 filter paper, and the filtrates were collected for subsequent analyses.
2 (w/v) ratio in a borosilicate extraction vessel. This mixture was then heated in a thermostatically controlled water bath at temperatures of 50 °C, 60 °C, and 70 °C. Enzyme solutions with different concentrations as per the design matrix were prepared simultaneously. The enzyme solutions were equilibrated to their respective temperatures before mixing, ensuring uniform reaction conditions during bio-colorant extraction. Prepared enzyme solutions were then introduced to the vessels containing roselle calyces and a water mixture. The extraction was allowed to proceed under controlled independent variable conditions until completion. After extraction, the mixture was filtered through Whatman No. 1 filter paper, and the filtrates were collected for subsequent analyses.
2.4. Antioxidant content (AOX) (scavenging activity of DPPH (2,2-diphenyl-1-picrylhydrazyl))
The EAE extract's AOX was assessed based on its free radical scavenging effect using the DPPH (2,2-diphenyl-1-picrylhydrazyl) assay, as per the procedure reported by Koley et al.33 An aliquot of each sample extract (1 mL) was mixed with 4mL of 0.0634 mM DPPH solution prepared in 95% methanol. The mixture was stirred and then vortexed. The extract was kept in a UV-visible spectrophotometer (UV-1800, Shimadzu, Japan), and the absorbance was calculated at 520 nm. After 15 minutes, the absorbance value was noted. The inhibition percentage of DPPH in the extract was calculated using the following formula:where A0 is the initial absorbance at 520 nm and A is the extract absorbance at 520 nm. Methanol (95%) was used as a blank.
2.5. Total phenol content
The TPC of the sample extract after the enzyme-assisted extraction was given spectrophotometrically using the Folin–Ciocalteu method with some modifications. First, 0.5 mL of the reagent (Folin–Ciocalteu) and 3.4 mL of distilled water were added to give 0.1 mL of each of the extracts.34 The combination was thoroughly blended and set aside for 10 minutes. Furthermore, 20% sodium carbonate was added to 1 mL of the blend and left for 30 minutes. Using a UV-visible spectrophotometer (UV-1800, Shimadzu, Japan), the absorbance was then noted at 750 nm. The results were expressed as mg GAE/100 g.2.6. Total flavonoids
Total flavonoids of roselle bio-extract were estimated using the aluminium chloride technique developed by Koley et al.33 Then, 4 mL of distilled water was added into a 10 mL volumetric flask, followed by the addition of 0.3 mL of 10% AlCl3·6H2O, 0.3 mL of 5% NaNO2 and an extract of 1 mL of the sample together. The mixed solution was left undisturbed at room temperature for 6 minutes. Following this, 2 mL of 1 N NaOH was added, and distilled water was used for dilution. The absorbance value of the solution was estimated at 510 nm against a blank solution, and the results were given in the form of catechin equivalent (mg CE/100 g).2.7. Colour
The hue angle (h°) of the roselle bio-extracts was determined using a Handheld tintometer (Lovibond, LC100, The Tintometer Ltd, UK). Before determining the colour of the bio-extracts, the instrument should be calibrated first. The liquid extract was placed in a cuvette, and the Lovibond meter, which has a lens and a slider at the back, was opened and placed on the liquid sample to detect the hue angle (h°) value. It also detects the L*, a*, and b* values.352.8. Total anthocyanins
The total anthocyanin content of roselle calyx extracts and fractions was determined using the pH differential method described in a previous work,36 with minor modifications. The extracts and fractions were prepared at a concentration of 25 mg mL−1 in distilled water. All analyses were performed in triplicate. The total anthocyanin content was quantified using a cyanidin-3-glucoside calibration curve and expressed as mg cyanidin-3-glucoside equivalents per 100 g of sample.2.9. Statistical analysis
The data were analysed using the Design Expert statistical software (ver. 13.0.5.0; Stat-Ease Inc., Minneapolis, MN, USA). The Central Composite Design (CCD) was applied to evaluate the effects of the independent variables of extraction on the response variables. Analysis of variance (ANOVA) was performed to determine the statistical significance of the models developed. The mean values and standard deviations are presented. The significance of the data was verified, and the differences among the mean values were determined at a confidence level of p < 0.05.2.10. Preparation of roselle-flavoured ice creams
Four ice cream formulations were developed: a control (without adding extract) and three enzyme-infused variants prepared utilizing amylase (ICA), cellulase (ICC), and pectinase (ICP) roselle extracts. To prepare 1 kg of mix, 50% milk and 23.5% cream were heated to 40 °C, followed by the addition of 25 mL of roselle extract, 15% sucrose, and 0.1% stabilizer. Subsequently, 1.3% starch, 5% milk solid-not-fat (MSNF), and 0.08% emulsifier were introduced. This mixture was then heated to 70 °C for 30 min, homogenized, cooled to 4 °C, and finally frozen at −4 °C using an ice cream maker. The same procedure was followed for the control sample also.372.11. Sensory attributes
The sensory attributes were assessed based on the generic descriptive method. The evaluation was performed by a semi-trained panel (n = 10, aged 22–25 years) using a 9-point hedonic scale to assess appearance, colour, flavour, texture, and overall acceptability. The samples were coded and presented under hygienic conditions, with water for palate cleansing.2.12. HPLC analysis
Quercetin in the plant extract was quantified using an HPLC system equipped with a C18 column (150 mm × 4.6 mm, 5 µm) and a UV detector. The mobile phase consisted of a methanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water ratio (such as 80
water ratio (such as 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, v/v, with 0.1% formic acid), operated in the isocratic mode at a flow rate of 1.0 mL min−1. The injection volume was 10 µL, and detection was carried out at 372 nm. Quantification was performed using an external calibration curve prepared with standard quercetin solutions.38
20, v/v, with 0.1% formic acid), operated in the isocratic mode at a flow rate of 1.0 mL min−1. The injection volume was 10 µL, and detection was carried out at 372 nm. Quantification was performed using an external calibration curve prepared with standard quercetin solutions.38where AIQ denotes the area of the individual quercetin-assigned peak and TAQ denotes the total area of quercetin-assigned peaks.
2.13. FTIR spectroscopic analysis
The roselle extract was analysed by an infrared beam using an FTIR spectrometer (Alpha E, Vertex 70, Bruker Optics, Inc., Billerica, MA, Germany). Measurements were carried out at 25 °C within the wavenumber range of 400–4000 cm−1, using a spectral resolution of 4 cm−1. The analysis was carried out following the methodology described by Montoya-Escobar et al.392.14. SEM analysis
The morphological characterization of the freeze-dried roselle extract-infused ice cream was performed using a Scanning Electron Microscope (SEM) (JEOL JSM-6360, Japan) at two different magnifications (100× and 250×). Gold-sputter-coated samples were mounted on aluminium stubs within the specimen chamber, and the images were captured at an accelerating voltage of 8 kV.403 Results and discussions
3.1. Effect of amylase on the extraction of roselle calyx at different enzyme concentrations and temperatures
The effect of amylase concentration and extraction temperature on the recovery of bioactive compounds from roselle (Hibiscus sabdariffa L.) calyces was studied. The experimental values are summarised in Table 1, and the 3D surface plots obtained are illustrated in Fig. 1. The results indicate that both parameters significantly influenced the total phenolic content (TPC), total anthocyanin content (TAC), total flavonoid content (TFC), antioxidant activity, and color properties of the extracts. The TPC ranged from 19.62 to 36.15 mg GAE/100 g, with the highest yield observed at 2.9% enzyme concentration and 60 °C, and the lowest at 10% enzyme concentration and 45.9 °C. Moderate enzyme concentrations at an optimum temperature of around 60 °C enhanced the release of phenolic compounds. This can be attributed to amylase-mediated hydrolysis of starch and cell wall polysaccharides, which improves solvent accessibility and facilitates the diffusion of bound phenolics.41,42 At higher temperatures (≥70 °C), TPC decreased, probably due to the thermal degradation of phenolics and enzyme denaturation, as similarly reported by Chongwilaikasem et al.36 and Lai et al.43 The TAC ranged between 75.61 and 115.89 mg/100 g, with the maximum value obtained at 17.1% enzyme concentration and 60 °C. The enhanced release of anthocyanins under these conditions suggests that amylase effectively disrupted polysaccharide matrices that encapsulate pigments, promoting the liberation of vacuolar anthocyanins.44 However, when the temperature exceeded the enzyme's thermal tolerance, anthocyanins degraded to colorless chalcones or brown degradation products, consistent with the findings by Bangi45 and Ahmed et al.46 The antioxidant activity, which ranged from 13.52% to 50.23%, correlated positively with both phenolic and anthocyanin contents. The highest antioxidant activity occurred at 10% enzyme concentration and 60 °C, indicating that optimal enzyme action enhances the release of redox-active phytochemicals. Phenolic compounds and anthocyanins contribute hydrogen atoms and electrons that neutralize free radicals, explaining this direct relationship.43,47 Lower activities under suboptimal conditions may result from incomplete hydrolysis and reduced release of antioxidant molecules.| Enzyme concentration (%) | Temperature (°C) | TPC (mg GAE/100 g) | TAC (mg/100 g) | Antioxidant (%) | TFC (mg CE/100 g) | Hue angle | pH |
|---|---|---|---|---|---|---|---|
| Amylase | |||||||
| 5.0 | 50.0 | 25.55 ± 0.23 | 93.50 ± 0.87 | 27.37 ± 0.03 | 126.17 ± 0.65 | 17.19 ± 1.08 | 2.40 ± 0.81 |
| 15.0 | 50.0 | 27.00 ± 1.45 | 85.86 ± 0.56 | 24.99 ± 1.65 | 119.98 ± 0.24 | 11.58 ± 0.96 | 2.45 ± 0.76 |
| 5.0 | 70.0 | 30.07 ± 2.10 | 80.39 ± 0.13 | 33.57 ± 0.74 | 110.51 ± 0.42 | 15.61 ± 0.47 | 2.23 ± 0.93 |
| 15.0 | 70.0 | 25.44 ± 0.54 | 115.17 ± 0.41 | 47.45 ± 0.28 | 137.32 ± 1.34 | 11.83 ± 1.23 | 2.69 ± 1.02 |
| 2.9 | 60.0 | 36.15 ± 0.68 | 97.03 ± 0.97 | 40.67 ± 0.47 | 150.68 ± 0.02 | 17.65 ± 0.74 | 2.30 ± 0.58 |
| 17.1 | 60.0 | 33.84 ± 1.12 | 115.89 ± 0.48 | 47.33 ± 1.05 | 166.89 ± 0.84 | 11.15 ± 0.16 | 2.74 ± 1.04 |
| 10.0 | 45.9 | 19.62 ± 0.16 | 75.61 ± 0.19 | 13.52 ± 1.64 | 88.00 ± 0.38 | 14.17 ± 0.49 | 2.37 ± 2.15 |
| 10.0 | 74.1 | 20.37 ± 3.01 | 81.28 ± 0.04 | 33.79 ± 0.24 | 88.39 ± 1.07 | 13.16 ± 0.53 | 2.40 ± 0.22 |
| 10.0 | 60.0 | 30.00 ± 0.65 | 100.75 ± 0.24 | 50.23 ± 0.33 | 160.00 ± 0.01 | 12.39 ± 0.08 | 1.29 ± 0.15 |
| 10.0 | 60.0 | 30.00 ± 0.65 | 100.75 ± 0.24 | 50.23 ± 0.33 | 160.00 ± 0.01 | 12.39 ± 0.08 | 1.29 ± 0.15 |
| 10.0 | 60.0 | 30.00 ± 0.65 | 100.75 ± 0.24 | 50.23 ± 0.33 | 160.00 ± 0.01 | 12.39 ± 0.08 | 1.29 ± 0.15 |
| 10.0 | 60.0 | 30.00 ± 0.65 | 100.75 ± 0.24 | 50.23 ± 0.33 | 160.00 ± 0.01 | 12.39 ± 0.08 | 1.29 ± 0.15 |
| 10.0 | 60.0 | 30.00 ± 0.65 | 100.75 ± 0.24 | 50.23 ± 0.33 | 160.00 ± 0.01 | 12.39 ± 0.08 | 1.29 ± 0.15 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Pectinase | |||||||
| 5.0 | 50.0 | 6.65 ± 0.38 | 109.62 ± 0.22 | 20.33 ± 0.17 | 121.45 ± 0.06 | 17.58 ± 0.11 | 2.35 ± 0.24 |
| 15.0 | 50.0 | 20.10 ± 0.41 | 127.85 ± 0.16 | 19.00 ± 0.94 | 101.38 ± 0.33 | 20.24 ± 0.77 | 2.45 ± 0.45 |
| 5.0 | 70.0 | 42.45 ± 1.74 | 104.20 ± 0.98 | 19.69 ± 1.25 | 128.73 ± 0.41 | 16.15 ± 0.60 | 2.53 ± 0.71 |
| 15.0 | 70.0 | 22.76 ± 0.36 | 99.36 ± 0.71 | 26.32 ± 0.87 | 135.96 ± 0.28 | 16.79 ± 0.14 | 2.41 ± 0.59 |
| 2.9 | 60.0 | 23.59 ± 0.21 | 118.35 ± 0.47 | 16.25 ± 0.04 | 152.70 ± 0.44 | 13.46 ± 1.05 | 2.43 ± 0.70 |
| 17.1 | 60.0 | 19.98 ± 0.65 | 126.49 ± 0.01 | 21.26 ± 1.10 | 144.53 ± 0.39 | 16.34 ± 2.33 | 2.42 ± 1.35 |
| 10.0 | 45.9 | 11.42 ± 0.14 | 110.47 ± 1.06 | 21.38 ± 0.49 | 81.34 ± 0.17 | 23.19 ± 0.41 | 2.44 ± 0.24 |
| 10.0 | 74.1 | 36.87 ± 0.23 | 86.46 ± 1.48 | 26.81 ± 0.32 | 110.94 ± 0.09 | 18.96 ± 0.82 | 2.55 ± 0.39 |
| 10.0 | 60.0 | 51.00 ± 0.08 | 122.71 ± 0.12 | 39.30 ± 0.04 | 170.00 ± 0.20 | 13.08 ± 0.33 | 2.03 ± 0.01 |
| 10.0 | 60.0 | 51.00 ± 0.08 | 122.71 ± 0.12 | 39.30 ± 0.04 | 170.00 ± 0.20 | 13.08 ± 0.33 | 2.03 ± 0.01 |
| 10.0 | 60.0 | 51.00 ± 0.08 | 122.71 ± 0.12 | 39.30 ± 0.04 | 170.00 ± 0.20 | 13.08 ± 0.33 | 2.03 ± 0.01 |
| 10.0 | 60.0 | 51.00 ± 0.08 | 122.71 ± 0.12 | 39.30 ± 0.04 | 170.00 ± 0.20 | 13.08 ± 0.33 | 2.03 ± 0.01 |
| 10.0 | 60.0 | 51.00 ± 0.08 | 122.71 ± 0.12 | 39.30 ± 0.04 | 170.00 ± 0.20 | 13.08 ± 0.33 | 2.03 ± 0.01 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Cellulase | |||||||
| 5.0 | 50.0 | 17.51 ± 0.38 | 82.19 ± 0.63 | 28.98 ± 0.26 | 113.44 ± 0.33 | 20.56 ± 0.80 | 2.39 ± 0.12 |
| 15.0 | 50.0 | 22.37 ± 1.25 | 117.25 ± 0.71 | 11.88 ± 0.31 | 105.43 ± 0.23 | 22.69 ± 0.08 | 2.33 ± 0.35 |
| 5.0 | 70.0 | 52.37 ± 2.01 | 99.27 ± 0.87 | 20.15 ± 2.10 | 135.80 ± 0.60 | 21.86 ± 0.83 | 2.28 ± 0.47 |
| 15.0 | 70.0 | 14.00 ± 0.94 | 112.22 ± 0.33 | 32.35 ± 0.91 | 90.63 ± 0.43 | 19.14 ± 0.24 | 2.83 ± 0.21 |
| 2.9 | 60.0 | 40.36 ± 0.23 | 99.01 ± 0.28 | 27.12 ± 0.46 | 113.08 ± 0.28 | 25.37 ± 0.32 | 2.30 ± 0.63 |
| 17.1 | 60.0 | 16.53 ± 0.10 | 127.26 ± 0.74 | 23.06 ± 0.57 | 75.47 ± 0.37 | 23.21 ± 0.45 | 2.59 ± 0.81 |
| 10.0 | 45.9 | 15.66 ± 0.64 | 83.55 ± 0.96 | 18.46 ± 0.14 | 128.71 ± 0.51 | 19.61 ± 0.28 | 2.35 ± 0.94 |
| 10.0 | 74.1 | 34.66 ± 0.47 | 88.09 ± 0.55 | 32.23 ± 0.01 | 133.05 ± 0.21 | 17.92 ± 0.73 | 2.63 ± 0.57 |
| 10.0 | 60.0 | 56.00 ± 0.05 | 101.11 ± 0.30 | 32.23 ± 0.01 | 195.00 ± 0.11 | 27.22 ± 0.34 | 2.90 ± 0.02 |
| 10.0 | 60.0 | 56.00 ± 0.05 | 101.11 ± 0.30 | 32.23 ± 0.01 | 195.00 ± 0.11 | 27.22 ± 0.34 | 2.90 ± 0.02 |
| 10.0 | 60.0 | 56.00 ± 0.05 | 101.11 ± 0.30 | 32.23 ± 0.01 | 195.00 ± 0.11 | 27.22 ± 0.34 | 2.90 ± 0.02 |
| 10.0 | 60.0 | 56.00 ± 0.05 | 101.11 ± 0.30 | 32.23 ± 0.01 | 195.00 ± 0.11 | 27.22 ± 0.34 | 2.90 ± 0.02 |
| 10.0 | 60.0 | 56.00 ± 0.05 | 101.11 ± 0.30 | 32.23 ± 0.01 | 195.00 ± 0.11 | 27.22 ± 0.34 | 2.90 ± 0.02 |
The TFC ranged from 88.00 to 166.89 mg CE/100 g, peaking at 17.1% enzyme concentration and 60 °C. The trend mirrors those of TAC and TPC, implying that flavonoids (many of which occur as glycosides) are efficiently extracted following enzymatic cleavage of glycosidic linkages and structural carbohydrates.19,41 The enzymatic mechanism involves amylase catalyzing the hydrolysis of α-1,4-glycosidic bonds in starch and hemicellulase, leading to cell wall loosening and enhanced mass transfer of secondary metabolites into the solvent phase. Colour characteristics (hue angle) and pH values further supported the biochemical trends. Samples with high anthocyanin content exhibited lower hue angles (11–12), indicating a deeper red hue, while extracts maintained acidic pH (1.29–2.74), favoring the stability of anthocyanins in their flavylium cation form.48 The slight pH variations may reflect the release of organic acids and breakdown of pectic substances during enzymatic action. Overall, the results confirm that amylase-assisted extraction at moderate temperatures (60 °C) and enzyme concentrations (10–17%) maximizes the extraction of phenolics, anthocyanins, and flavonoids from roselle calyces, leading to a higher antioxidant potential and a richer colour intensity. The synergistic effect arises from both enzymatic hydrolysis of carbohydrate matrices and thermal facilitation of mass transfer, provided that the temperature remains below degradation thresholds. Similar optimal ranges and mechanisms have been reported in recent green extraction studies of roselle.36,41,42 This supports the potential of enzyme-assisted extraction as an eco-friendly and efficient alternative to conventional solvent extraction for enhancing the functional and sensory qualities of roselle products.
| Source | df | TPC (mg GAE/100 g) | TAC (mg/100 g) | Antioxidant (%) | TFC (mg CE/100 g) | Hue angle | pH |
|---|---|---|---|---|---|---|---|
| a F-values mentioned in the table. **P is significant at 1% level (P ≤ 0.01), *P is significant at 5% level (P ≤ 0.05). NS – non-significant; CV – coefficient of variation. | |||||||
| Amylase | |||||||
| Model | 5 | 405.95** | 222.22** | 2499.22** | 1631.66** | 1095.03** | 3252.34** |
| Enzyme concentration (%) (A) | 1 | 39.85** | 217.34** | 380.19** | 2015.89** | 2240.51** | 599.82** |
| Temperature (°C) (B) | 1 | 15.50** | 44.02** | 2855.19** | 5.29NS | 966.17** | 5.91NS |
| A × A | 1 | 301.59** | 42.09** | 506.21** | 21.75* | 2090.80** | 768.42** |
| B × B | 1 | 1400.03** | 490.19* | 8692.57** | 2617.27** | 2917.32** | 734.02** |
| A × B | 1 | 70.88** | 270.11** | 459.41** | 2315.58** | 850.53** | 157.30** |
| Pure error | 4 | — | — | — | — | — | — |
| Total | 10 | — | — | — | — | — | — |
| CV% | — | 0.226 | 0.197 | 0.292 | 0.315 | 0.170 | 0.421 |
| R2 | — | 0.9966 | 0.9937 | 0.994 | 0.9999 | 0.9999 | 0.9996 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Pectinase | |||||||
| Model | 5 | 5161.04** | 5026.91** | 2796.50** | 1830.53** | 452.32** | 615.11** |
| Enzyme concentration (%) (A) | 1 | 121.93** | 1064.50** | 248.85** | 742.04** | 117.87** | 0.79NS |
| Temperature (°C) (B) | 1 | 5250.64** | 906.45** | 334.49** | 870.46** | 255.84** | 59.50** |
| A × A | 1 | 1238.92** | 3.49NS | 568.83** | 119.78** | 84.26** | 1386.55** |
| B × B | 1 | 495.78** | 4146.87** | 5249.11** | 5283.91** | 1857.31** | 1940.49** |
| A × B | 1 | 2080.64** | 1827.31** | 205.58** | 1858.72** | 17.70* | 65.94** |
| Pure error | 4 | — | — | — | — | — | — |
| Total | 10 | — | — | — | — | — | — |
| CV% | — | 0.363 | 0.269 | 0.277 | 0.316 | 0.240 | 0.013 |
| R2 | — | 0.9997 | 0.9997 | 0.9995 | 0.9999 | 0.9969 | 0.9977 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Cellulase | |||||||
| Model | 5 | 1077.35** | 79.03** | 51.46** | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 053.01** 053.01** |
174.41** | 1205.04** |
| Enzyme concentration (%) (A) | 1 | 683.38** | 203.50** | 6.69NS | 2920.15** | 12.75** | 707.50** |
| Temperature (°C) (B) | 1 | 879.48** | 8.97NS | 57.20** | 48.45** | 9.73* | 539.45** |
| A × A | 1 | 662.93** | 68.21** | 53.65** | 6885.03** | 15.77** | 2570.77** |
| B × B | 1 | 679.33** | 68.31** | 50.32** | 1505.32** | 101.95** | 2092.40** |
| A × B | 1 | 3733.07** | 25.71** | 101.45** | 712.67** | 34.47** | 649.85** |
| Pure error | 4 | — | — | — | — | — | — |
| Total | 10 | — | — | — | — | — | — |
| CV% | — | 0.140 | 0.218 | 0.145 | 0.695 | 0.413 | 0.011 |
| R2 | — | 0.9999 | 0.9826 | 0.9735 | 0.9999 | 0.9920 | 0.9988 |
The 3D response surface and contour plots (Fig. 1a) revealed a clear saddle-shaped curvature, showing that maximum TPC occurred at intermediate enzyme concentrations and around 60 °C. The surface slope became flatter beyond this point, confirming the existence of an optimum region. TPC decreased due to insufficient substrate hydrolysis at low enzyme concentrations and high temperatures, while at high enzyme concentrations and temperatures above 70 °C, enzyme inactivation and phenolic oxidation became dominant. The pronounced curvature in the contour map illustrates the strong interaction between both variables, consistent with the significant A × B term in the model. Amylase hydrolyzes α-1,4-glycosidic linkages in starch and hemicellulose, loosening the cell wall matrix and facilitating solvent penetration and release of bound phenolics.36,41 The optimal phenolic recovery at 60 °C supports the findings by Chenxin et al.44 and Lai et al.,43 who reported similar temperature optima for enzyme-assisted extractions. In summary, the contour and response surface analyses confirm that moderate temperatures (60 °C) and enzyme concentrations (2.9–10%) provide optimal conditions for maximizing the TPC. These parameters achieve a balance between enhanced enzymatic hydrolysis and phenolic stability, reinforcing that controlled amylase-assisted extraction is an effective green technique for improving the phenolic yield from roselle calyces.
The strong quadratic terms for both enzyme concentration (A2) and temperature (B2), as well as their highly significant interaction (A × B), underscore the nonlinear and interdependent nature of these variables in modulating the antioxidant yield. This synergy suggests that the enzyme efficacy is intricately linked to temperature-dependent conformational dynamics, which regulate catalytic turnover and substrate accessibility.19,45 The enhanced antioxidant extraction achieved here aligns with green extraction trends advocating enzymatic pretreatment combined with optimized thermal conditions to maximize the yield while minimizing the solvent and energy usage.41,48 This enzymatic approach significantly increases the antioxidant recovery compared to conventional extraction methods. It confirms the potential of amylase-assisted green extraction in valorizing roselle calyces as functional food ingredients rich in polyphenols and anthocyanins.42,46 The optimal parameters defined here can inform scalable extraction protocols aimed at producing roselle extracts with enhanced bioactivity for applications in nutraceutical, pharmaceutical, and food industries.
The significant temperature and enzyme interaction (A × B) observed here emphasizes that the optimal flavonoid extraction depends on achieving a balance between sufficient enzymatic hydrolysis and the thermal stability of both enzyme and flavonoids. Similar synergistic effects have been observed in enzyme-assisted and ultrasound-assisted extractions of roselle, where moderate conditions maximized TFC and antioxidant activity due to enhanced enzymatic catalysis and reduced matrix resistance.44,47 The near-perfect model fit (R2 = 0.9999) underscores the robustness and reproducibility of the optimization process, validating response surface methodology as a powerful tool for process refinement.19 These findings demonstrate that amylase-assisted extraction at controlled temperatures offers an efficient green extraction route for maximizing the flavonoid yield in roselle calyces. The process supports the sustainable valorization of this high-antioxidant botanical source for application in nutraceuticals and functional foods.41,48
The significant quadratic (A2, B2) and interaction (A × B) terms confirm the complex interdependence between enzymatic hydrolysis and temperature on pigment integrity. Higher temperatures (>70 °C) or enzyme excess may lead to partial pigment oxidation and browning due to Maillard-type reactions or phenolic condensation, contributing to higher hue values and duller coloration.36,47 The highly consistent model fit (R2 = 0.9999) indicates that the variations in hue angle are predominantly governed by the controlled process variables rather than experimental error. These findings support earlier reports that optimized enzymatic and thermal processing of roselle enhances the pigment extraction efficiency and visual quality of anthocyanin-rich products.45,48 The current results further emphasize that a balanced combination of amylase activity and moderate temperature can maximize both the pigment yield and the color intensity, which are the key parameters for the development of natural colorants and high-value roselle-based beverages or functional food formulations.19,41
The significant quadratic and interaction effects (B2 and A × B) underscore the nonlinear dependency of pH on both enzyme activity and thermal energy. This suggests that acid generation and stability result from the balance between enzymatic hydrolysis, phenolic liberation, and thermally driven chemical transformations. The previous studies on enzyme-assisted roselle extraction similarly reported decreases in pH linked to enhanced organic acid release and anthocyanin hydrolysis, improving pigment solubility and colour intensity.46,48 The strong model predictability (R2 = 0.9996) confirms that pH variation is highly controllable through process parameter optimization. Overall, these results demonstrate that moderate amylase concentration (10%) and controlled temperature (60 °C) effectively reduce the extract pH by promoting acid release without compromising biochemical stability. This pH modulation enhances anthocyanin stability and extraction efficiency, reinforcing enzymatic treatment as a valuable step in green processing of roselle for application in natural colorants and functional beverages.19,41
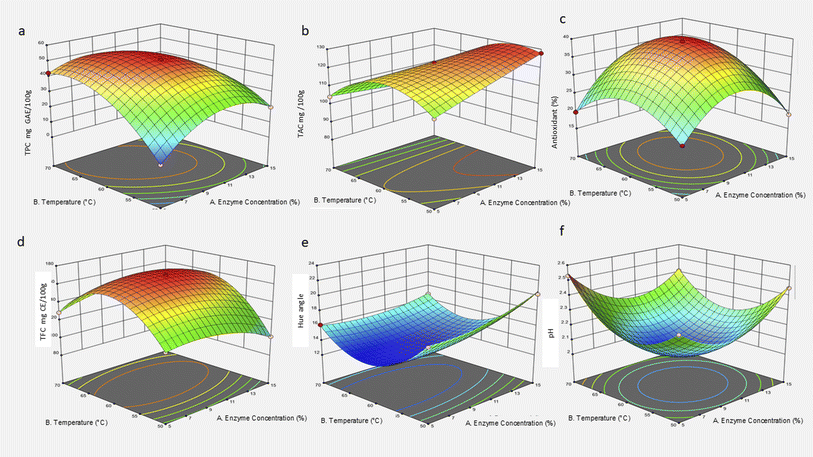
3.2. Effect of pectinase on the extraction of roselle calyx at different enzyme concentrations and temperatures
Pectinase-assisted extraction significantly enhanced the recovery of bioactive compounds from Hibiscus sabdariffa calyces, with both enzyme concentration and temperature exerting distinct influences on the total phenolic content (TPC), total anthocyanin content (TAC), antioxidant activity, total flavonoid content (TFC), hue angle, and pH. The results are summarized in Table 1, and the 3D surface plots obtained are illustrated in Fig. 2. The response patterns indicate that the moderate enzyme concentration (10%) combined with a temperature of 60 °C yielded optimal extraction performance across all parameters achieving TPC = 51.00 mg GAE/100 g, TAC = 122.71 mg/100 g, antioxidant activity = 39.30%, and TFC = 170.00 mg CE/100 g, accompanied by a low hue angle (13.08°) indicative of intense red pigmentation and a pH of 2.03, suggesting strong acidity and pigment stability. Pectinase hydrolyzes α-1,4-glycosidic linkages in pectin, a major structural polysaccharide in roselle cell walls, effectively degrading the middle lamella and cell wall matrix. This facilitates solvent penetration and the release of phenolic compounds, anthocyanins, and flavonoids from vacuolar and cell-bound compartments.41,42 Enhanced phenolic liberation under moderate thermal conditions can be attributed to improved enzyme kinetics and diffusion rates without denaturation, while excessive heating (>70 °C) or enzyme overload likely reduces efficiency due to pectinase inactivation or phenolic oxidation.36,43The strong correlation among TPC, TFC, and antioxidant activity reflects the co-extraction of phenolic and flavonoid compounds that contribute synergistically to radical scavenging capacity.45,46 The optimal extraction point (10%, 60 °C) also corresponded to the lowest hue angle, signifying high anthocyanin content and more saturated red coloration. Anthocyanins, primarily delphinidin and cyanidin derivatives, exhibit greater chromatic intensity under acidic conditions (pH 2–3), consistent with the measured pH decrease at optimal extraction.44,47 The acidity probably stems from organic acid release (hibiscus acid and citric acid) facilitated by the enzymatic breakdown of pectic substances.48 The combined influence of enzyme concentration and temperature revealed a synergistic effect where controlled enzymatic hydrolysis, coupled with moderate heat, maximized both extraction efficiency and pigment stability. Excessive enzyme activity or temperature appeared to diminish yields, potentially from anthocyanin degradation, polymerization, or phenolic condensation reactions.19 These findings align with recent green-extraction optimizations reporting that pectinase-assisted treatments at 55–65 °C achieve superior polyphenol and pigment recovery while minimizing solvent and energy input.13,41 Pectinase at 10% and 60 °C proved to be the most effective condition for maximizing phenolic and flavonoid extraction while enhancing the antioxidant potential and maintaining pigment stability. This supports the integration of pectinase-assisted green extraction as a sustainable, efficient method for valorizing roselle calyces into high-value natural colorants and functional ingredients for application in food products, nutraceuticals, and beverages.
The 3D response surface plot (Fig. 2a) revealed a dome-like curve with the TPC peak centred around 60 °C and 10% enzyme concentration. This visualized the interaction effects, confirming that the optimal phenolic recovery occurs only within a narrow process window. The significant quadratic and interaction terms support this finding and align with previous optimization studies using response surface methodology.19,46 In conclusion, enzymatic-assisted extraction using pectinase at 60 °C and 10% concentration offers a green and efficient method to maximize the TPC from roselle calyces. This balance prevents degradation while enhancing bioactive release, supporting its potential applications in clean-label functional foods and nutraceutical formulations.36,48
The highest TAC was obtained at 15% enzyme concentration and 50 °C, indicating that a relatively high enzyme load under mild thermal conditions maximized anthocyanin release while minimizing thermal degradation. This aligns with the response surface analysis, where the 3D plot (Fig. 2b) typically displayed a peak TAC within this zone and a steady decline at higher temperatures. The significant A × B interaction confirms that the combined effect of enzyme and temperature is non-additive, and that optimal anthocyanin extraction requires fine-tuning of both parameters.19,43 In conclusion, effective extraction of anthocyanins from roselle using pectinase depends on maintaining moderate temperatures (around 50–60 °C) to avoid thermal breakdown while applying sufficient enzyme (10–15%) to disrupt cellular matrices. These findings support enzyme-assisted extraction as a green, efficient alternative for preserving anthocyanin integrity in food and nutraceutical products.36,48
The 3D response surface (Fig. 2c) would illustrate a peak centered near 10% enzyme concentration and 60 °C, forming a curved optimum region. This contour reflects the significant interaction (A × B) observed in the statistical model, where enzyme and temperature synergistically influence antioxidant release. These findings are consistent with prior studies that emphasize controlled enzymatic treatment and mild thermal conditions for maximizing bioactive functionality in plant extracts.36,43 In summary, pectinase-assisted extraction under optimal conditions significantly enhances the antioxidant activity of roselle calyx extract by improving the release and preservation of phenolic antioxidants. This supports its potential use in functional foods and nutraceutical formulations targeting natural antioxidant delivery.
The plateau (Fig. 2d) in flavonoid content at enzyme concentrations above 10% suggests enzyme saturation due to limited available substrate sites or possible enzyme inhibition due to the presence of excessive enzymes.43 Furthermore, the significant enzyme–temperature interaction indicates that neither factor alone optimizes extraction; rather, their synergistic balance is critical.19 These results demonstrate that moderate enzyme concentration and temperature maximize flavonoid recovery from roselle calyx via enhanced enzymatic cell wall disruption while preserving the compound stability. This supports the application of enzyme-assisted extraction as an effective, green method for obtaining flavonoid-rich extracts with potential health-promoting antioxidant properties suitable for food and nutraceutical industries.36,48
The significant interaction (Fig. 2e) between enzyme concentration and temperature underscores that optimal color extraction is not governed by a single factor but rather by the synergistic balance between enzymatic activity and thermal stability. This balance is critical in maintaining the vibrant color quality essential for consumer acceptance and application in functional foods, where visual appeal correlates with perceived freshness and antioxidant potential.36,48 In conclusion, controlling both the enzyme concentration and temperature during pectinase-assisted extraction is crucial for maximizing the pigment yield and stability, thereby preserving the desirable red hue of roselle calyx extracts. These findings support the application of enzyme-assisted extraction as a green, efficient method to enhance not only bioactive compound recovery but also the sensory quality of roselle-derived products.
The interaction effect (Fig. 2f) indicates that certain combinations of enzyme concentration and temperature can synergistically influence pH, potentially through modulating enzyme efficiency and substrate accessibility, which in turn affects the release of acidic compounds.42 The maintenance of a low pH is crucial for anthocyanin stability and overall extract quality, as acidic environments preserve pigment colour and antioxidant properties.43,46 In conclusion, the pectinase concentration plays a more dominant role than temperature in modulating the pH of roselle calyx extract, with enzymatic hydrolysis contributing to the maintenance of the acidic conditions favourable for pigment stability and bioactive compound preservation. These findings reinforce the importance of optimizing enzyme dosage alongside temperature to control extract acidity and quality during enzymatic extraction processes.
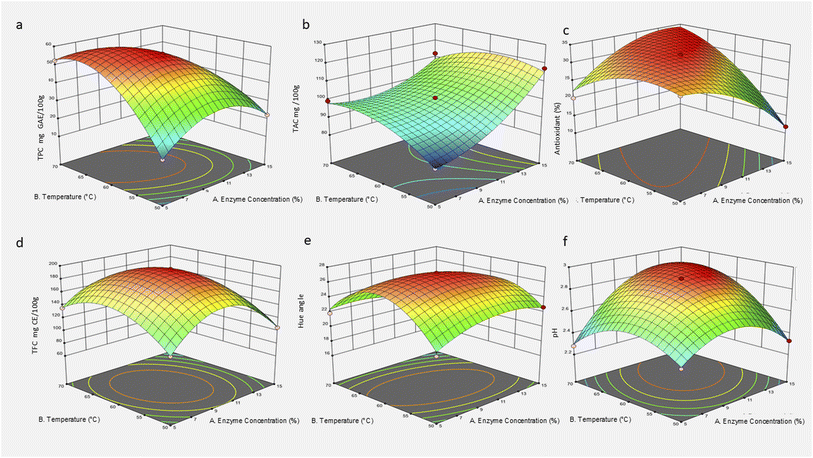
3.3. Effect of cellulase on the extraction of roselle calyx at different enzyme concentrations and temperatures
The enzyme extraction of roselle calyx bioactives using cellulase showed pronounced effects of both the enzyme concentration and the temperature on the quality and yield of the extracts. These parameters influenced the total phenolic content (TPC), total anthocyanin content (TAC), antioxidant capacity, total flavonoid content (TFC), hue angle, and pH, reflecting their critical roles in optimizing extraction, and the results are summarized in Table 1 and the obtained 3D surface plots are illustrated in Fig. 3. TPC increased significantly with temperature, peaking at 56.00 mg/100 g at 10% enzyme concentration and 60 °C. This enhancement is primarily due to cellulase's ability to hydrolyze the cellulose components of the plant cell wall, thereby disrupting the structural barriers and facilitating the release of bound phenolic compounds into the solvent.41 The rigid cellulase matrix in roselle calyx limits the availability of phenolics; cellulase enzymatic action breaks down β-1,4-glycosidic bonds in cellulose, loosening the matrix and increasing phenolic diffusion.51 However, TPC declined at higher enzyme concentrations (15%) and temperature (70 °C), which may be explained by enzyme denaturation, substrate inhibition, or thermal degradation of sensitive phenolics, suggesting that an optimal window for extraction exists.42TAC reached a maximum of 127.26 mg/100 g at 17.1% enzyme and 60 °C. The release of anthocyanins, water-soluble pigments responsible for roselle's characteristic red color, is similarly enhanced by cellulase-mediated cell wall disruption, which facilitates their liberation from vacuoles and cell compartments. However, anthocyanins are known to be thermolabile and susceptible to degradation under extreme temperature or prolonged enzymatic treatment, explaining why increases in enzyme concentration and temperature beyond optimal conditions did not yield further TAC improvement.46 Additionally, pH plays a role in anthocyanin stability, with acidic conditions (pH 2.28–2.90 in extracts) favouring pigment preservation.41 Antioxidant activity, measured as the percentage of radical scavenging capacity, peaked around 32% at enzyme concentrations of 10–15% and temperatures of 60–70 °C. This correlates with the enhanced release of phenolics and anthocyanins, which contribute significantly to antioxidant potential. The initial increase indicates efficient enzymatic breakdown of the cell wall matrix, releasing antioxidant molecules, while declines or plateaus at higher temperatures or enzyme levels suggest the degradation of antioxidants or enzyme inactivation.42,43 TFC exhibited the highest value of 195 mg CE/100 g at 10% enzyme and 60 °C. Flavonoids, a subclass of phenolics, are structurally embedded within the plant cell wall matrix. Enzymatic hydrolysis by cellulase exposes and liberates these molecules. However, increased enzyme concentration beyond 10% led to reduced flavonoid yields, possibly due to enzymatic degradation or complexation reactions at higher substrate or enzyme loads.45
The hue angle ranged from approximately 17.92 to 27.22, with higher values at a moderate enzyme concentration and temperature. This parameter reflects the stability and quality of anthocyanin pigments in the extract. Increased hue angle suggests more vivid red coloration, while lower values may indicate pigment degradation or shifts toward brownish hues due to anthocyanin breakdown under harsh conditions.36 Optimal cellulase treatment preserves pigment integrity by balancing cell wall disruption and minimal pigment degradation. The extract pH remained acidic, ranging between 2.28 and 2.90, consistent with the natural acidic environment of roselle calyx. The slight increase in pH at higher enzyme concentrations and temperatures may be related to the release of organic acids and enzymatic hydrolysis products. Acidic pH is beneficial for anthocyanin stability, preventing their degradation during extraction.41 The mechanism underlying these observations is rooted in cellulase's enzymatic hydrolysis of cellulose, the main structural polysaccharide in the plant cell wall. By cleaving β-1,4-glycosidic linkages, cellulase disrupts the rigid cellulose framework, enhancing cell wall permeability and allowing easier diffusion of bioactive compounds such as phenolics, anthocyanins, and flavonoids into the extraction medium. This cell wall deconstruction increases the extractability and yield of target compounds.45,46
Temperature is a critical factor influencing enzyme activity and compound stability. At moderate temperatures (60 °C), cellulase exhibits optimal catalytic efficiency, maximizing cell wall breakdown without significant thermal denaturation or degradation of heat-sensitive compounds. However, temperatures above this threshold may denature the enzyme or degrade phenolic and pigment molecules, reducing the overall extract quality. Similarly, the enzyme concentration affects the availability of active sites for substrate hydrolysis. Low to moderate enzyme concentrations improve the extraction yield by increasing the hydrolysis rates, while excessive enzyme amounts can lead to substrate inhibition, enzyme aggregation, or nonspecific degradation of bioactives, resulting in diminished returns.42 In summary, cellulase-assisted extraction of roselle calyx bioactives is highly dependent on the fine-tuning of enzyme concentration and extraction temperature. Optimal conditions, particularly around 10% enzyme concentration and 60 °C, maximize the release and stability of phenolics, anthocyanins, flavonoids, and antioxidant capacity while preserving pigment color and pH. These findings highlight the importance of balancing enzymatic hydrolysis and thermal stability to enhance the functional quality of roselle extracts for application in food, nutraceutical, and pharmaceutical products.
The significant quadratic and interaction terms (p < 0.01) in the model suggest that the effects of enzyme concentration and temperature are non-linear and interdependent. For example, higher enzyme concentrations at elevated temperatures yielded lower TPC, probably due to substrate saturation and thermal instability of phenolics.43,45 These findings underscore the importance of balancing enzyme load and thermal conditions for efficient extraction. The response surface model showed excellent fit (R2 = 0.9999; CV = 0.140) (Table 2), confirming the reliability of the results. The model further supports that cellulase-assisted extraction is highly tunable and efficient under optimized conditions. In conclusion, a moderate enzyme concentration (10%) combined with a controlled temperature (60 °C) yields the highest TPC from roselle calyx. These conditions maximize phenolic release while minimizing degradation, offering a practical and sustainable approach for producing antioxidant-rich extracts for application in functional foods or nutraceuticals.
A relatively high TAC (117.25 mg/100 g) was also recorded at 15% enzyme concentration and 50 °C, demonstrating that even at lower temperatures, a high enzyme loading can sufficiently degrade plant tissue to release anthocyanins while minimizing the thermal degradation. This suggests that optimal anthocyanin recovery is not solely temperature-dependent but requires a well-balanced enzyme-to-substrate interaction.36 The significant A × B interaction reflects the synergistic role of enzyme concentration and temperature. For example, high enzyme concentrations are more effective at moderate temperatures but may not yield proportional improvements at elevated temperatures due to the possible anthocyanin breakdown or enzyme inactivation. This behaviour aligns with prior observations in enzyme-assisted extraction systems from other plant sources, where anthocyanin stability limited extraction efficiency at higher processing temperatures.44,45 In conclusion, cellulase-assisted extraction significantly improves anthocyanin yield from roselle calyces when applied under optimized conditions. A temperature of 60 °C and an enzyme concentration of 15–17.1% were most effective, achieving high pigment recovery while preserving anthocyanin stability. These findings are important for the development of clean-label natural colorants and antioxidant-rich functional ingredients derived from roselle.
The significant A × B interaction (Fig. 3c) indicates that the enzyme concentration modifies how temperature affects the antioxidant activity. For instance, higher enzyme levels may require elevated temperatures to achieve optimal antioxidant release, but excessive heat may also degrade some thermolabile compounds. The presence of a strong quadratic effect further suggests that both under- and over-processing can limit the antioxidant yield, reinforcing the need for a precisely controlled extraction environment.36,45 In conclusion, the antioxidant activity of roselle extract can be maximized using cellulase under optimized conditions, specifically at 15% enzyme concentration and 70 °C. While higher enzyme concentrations and temperatures generally enhance antioxidant recovery, a balanced interaction between these factors is critical to avoid compound degradation and ensure functional potency of the extract.
The strong quadratic effects (A2 and B2) indicate that both under- and over-processing (low or high enzyme levels and temperatures) can reduce the flavonoid yield. This aligns with previous reports where enzymatic overactivity or thermal stress degraded sensitive phenolic structures.43,44 Additionally, the interaction effect (A × B) suggests that temperature modulates the enzyme's efficiency, where higher temperatures can boost enzyme activity to a point, but beyond optimal levels, the degradation of heat-sensitive flavonoids probably offsets the benefits. The plateau in TFC values at 195.00 mg CE/100 g across multiple replicates at 10% and 60 °C indicates a saturation point beyond which no additional release occurs, possibly due to extraction equilibrium.36,45 In conclusion, cellulase-assisted extraction significantly enhances flavonoid recovery from roselle calyces when the enzyme concentration and temperature are optimized. The best conditions observed were 10% cellulase at 60 °C, where the maximum TFC was consistently achieved. These findings are crucial for designing efficient, enzyme-based extraction protocols for natural antioxidants for application in functional foods and nutraceuticals.
The significant interaction between the temperature and the enzyme concentration (A × B) suggests that the temperature amplifies the enzymatic impact on colour. At optimal moderate enzyme levels (10%) and elevated temperatures (60 °C), the hue angle peaked, potentially due to maximized anthocyanin release and stabilization. In contrast, either too low or too high enzyme levels, combined with suboptimal temperatures, resulted in hue reduction, likely linked to pigment oxidation or polymerization.36 In conclusion, the hue angle of cellulase-treated roselle extract is significantly affected by extraction conditions. A moderate enzyme concentration (10%) combined with elevated temperatures (60 °C) preserves the most vivid coloration by optimizing the anthocyanin extraction while minimizing degradation. These findings are crucial for industrial applications aiming to retain the visual appeal and functional colour properties of roselle-based products.
3.4. Comparative analysis of amylase, cellulase, and pectinase under optimized conditions
The enzyme-assisted extraction of roselle calyx bioactives reveals that the nature of the enzyme and extraction conditions critically influence the yield and profile of phenolic compounds, anthocyanins, antioxidant activity, flavonoids, color attributes, and pH. The comparative analysis results are listed (Table 3). Cellulase, applied at 10% concentration and 60 °C, yielded the highest total phenolic content (55.34 mg GAE/100 g) and total flavonoid content (193.05 mg CE/100 g). Cellulase hydrolyzes β-1,4-glycosidic bonds in cellulose, a major structural polysaccharide in plant cell walls. By breaking down cellulase microfibrils, cellulase disrupts the cell wall matrix by facilitating the release of intracellular and cell wall-bound phenolic and flavonoid compounds.41,42 This enzymatic degradation increases cell wall porosity and extractability, which is consistent with the enhanced phenolic recovery reported in green extraction methods.36 The increase in flavonoids, a subclass of phenolics often bound to polysaccharides, further supports cellulase's role in liberating complexed bioactives. Pectinase at 15% and 50 °C was most effective in extracting anthocyanins (127.79 mg/100 g), pigments responsible for the characteristic red hue of roselle. Pectinase catalyzes the breakdown of pectin, a polysaccharide present in the middle lamella of plant cells, which acts as a cementing agent holding cells together.44 The degradation of pectin loosens cell adhesion and enhances pigment release from vacuoles, which are the primary storage sites of anthocyanins. This mechanism explains the superior anthocyanin content and the lower hue angle observed with pectinase treatment, indicating a more vivid red coloration.45,48 Additionally, pectinase treatment can improve the juice yield and clarity, beneficial for industrial processing.42| Enzymes | Enzyme concentration (%) | Temperature (°C) | TPC (mg GAE/100 g) | TAC (mg/100 g) | Antioxidant (%) | TFC (mg CE/100 g) | Hue angle | pH |
|---|---|---|---|---|---|---|---|---|
| a TPC – total phenolic content (mg GAE/100 g); TAC – total anthocyanin content (mg/100 g); TFC – total flavonoid content (mg CE/100 g); AOX – antioxidant activity (%); h° – hue angle (°); results are expressed as mean ± SD (n = 3). | ||||||||
| Amylase | 3 | 60 | 35.09 ± 1.20 | 99.65 ± 0.46 | 39.14 ± 0.11 | 151.88 ± 0.03 | 17.93 ± 1.16 | 2.37 ± 0.13 |
| Pectinase | 15 | 50 | 33.35 ± 0.34 | 127.79 ± 0.48 | 28.95 ± 0.41 | 133.07 ± 0.88 | 17.32 ± 1.02 | 2.24 ± 0.38 |
| Cellulase | 10 | 60 | 55.34 ± 0.17 | 102.36 ± 0.52 | 32.36 ± 0.07 | 193.05 ± 0.32 | 27.08 ± 0.53 | 2.91 ± 0.29 |
Amylase, used at 3% concentration and 60 °C, led to the highest antioxidant activity (39.14%). Amylase hydrolyzes starch molecules into simpler sugars such as maltose and glucose, which may contribute to increased antioxidant potential either by releasing antioxidant-bound polysaccharides or through the formation of Maillard reaction products during processing, known for antioxidant properties.43,46 Although amylase showed lower phenolic and flavonoid extraction than cellulase, its enhancement of antioxidant capacity suggests a complementary mechanism where starch breakdown supports the availability or stability of antioxidant compounds. The pH values observed in cellulase-treated extracts were higher (around 2.9), which could influence anthocyanin stability and color expression, as anthocyanins are pH-sensitive pigments.19 Pectinase-treated extracts exhibited lower pH values, potentially preserving anthocyanin stability and enhancing colour intensity. In summary, in the differential enzymatic actions, cellulase breaks down cellulose, pectinase degrades pectin, and amylase hydrolyzes starch, and each facilitates the liberation of specific bioactive compounds from the roselle calyx matrix. This specificity underscores the importance of enzyme selection based on target compounds and desired extract qualities.41 Integrating these enzymes sequentially or simultaneously could synergistically improve the extraction efficiency, maximizing the yield and functional properties of Roselle extracts for applications in food, nutraceutical, and pharmaceutical products.
3.5. Comparative analysis of amylase, cellulase, and pectinase under optimized conditions infused in ice creams
The integration of amylase, cellulase, and pectinase enzymes into roselle-infused ice creams led to distinct differences in colour intensity, texture, structure, and air incorporation, which are visually represented (Table 4). These changes are attributed to the enzymatic breakdown of complex macromolecules (starch, cellulase, and pectin), which significantly influenced the physicochemical and sensory characteristics of the final product. The amylase-treated ice cream exhibited a light pink hue with a porous, aerated surface. Amylase acts on starches present in dairy and plant-derived ingredients by hydrolyzing α-1,4-glycosidic bonds, converting polysaccharides into maltose and glucose. This reaction not only increases the sweetness of the base but also reduces the viscosity, leading to improved aeration and overrun during churning.53 The visible fine air bubbles (Table 4) suggest enhanced emulsification due to reduced starch thickness, allowing better dispersion of fat and water phases. The degradation of starch reduces its water-holding capacity, enabling better freezing and a smoother melt-down profile. The increased sugar concentration due to amylolysis also acts as a cryoprotectant, influencing freezing point depression, which contributes to the softer texture observed.54 However, excessive amylase activity may lead to overly thin consistency, which is why optimization is crucial.The cellulase-infused sample showed a deep red, dense ice cream matrix with minimal air incorporation. This enzyme hydrolyzes β-1,4-glycosidic bonds in cellulase, producing glucose or cellobiose from plant-based fibers in the roselle calyces. The breakdown of cell wall components facilitates the release of intracellular pigments, including anthocyanins, which account for the pronounced red coloration.55 This cellulolytic action improves colorant bioavailability and may enhance antioxidant retention. Additionally, the liberated soluble fibers increase water-binding and viscosity, contributing to a firmer, gel-like consistency with minimal overrun.37 The limited presence of air bubbles suggests the reduced ability of the mixture to entrap air during freezing, probably due to increased viscosity, which restricts the formation of a lighter structure. The mechanism here reflects dual benefits: improved colour extraction from roselle tissues and structural enhancement due to fiber solubilization. However, excessive viscosity may lead to a heavy mouthfeel, which should be balanced for consumer acceptability.
The pectinase-treated ice creams exhibited a uniform pink tone and a visibly smoother surface, with evenly distributed micro-bubbles indicating excellent aeration and emulsification. Pectinase catalyzes the hydrolysis of α-1,4-glycosidic bonds in pectic substances, particularly polygalacturonic acid chains found in plant cell walls. This depolymerization facilitates the release of water-soluble anthocyanins and flavonoids, enhancing color uniformity.56 The degradation of high-molecular-weight pectins lowers viscosity, improves flowability, and enhances the interaction between air, fat, and water during churning. The result is a creamier texture, better aeration, and a refined mouthfeel.53 However, excessive pectin degradation can reduce the body and stability of the ice cream, though under this optimized condition, a balanced structure was maintained. Additionally, the enhanced diffusion of bioactive compounds improves the product's antioxidant profile and visual appeal, both essential for consumer preference and health-oriented formulations.54
These findings are consistent with previous studies highlighting the benefits of enzyme-assisted extraction and modification in functional food systems. Gengatharan56 and Bandara & Mahendran55 showed that pectinase and cellulase, respectively, improve pigment extraction and product texture. Similarly, Homayouni Rad et al.53 confirmed that enzyme-treated ice cream formulations using plant extracts yield better sensory and nutritional outcomes. The application of food-grade enzymes (amylase, cellulase, and pectinase) in roselle-colored ice creams not only improved the aesthetic quality (colour and uniformity), but also affected physical properties such as texture, viscosity, and aeration. The underlying biochemical mechanisms of each enzyme significantly influence the matrix behaviour, making them valuable tools in the formulation of clean-label, functional ice creams. Optimal enzyme use allows the tailoring of product attributes based on consumer preferences and nutritional goals. Overall, enzymatic infusion improved the colour extraction, texture, and visual appeal of roselle bio-colorant ice creams, with each enzyme offering distinct advantages. Cellulase provided the most vibrant colour and dense structure, with a smoother and more aerated product, which contributed to a balanced sweetness and moderately light texture.
| Sample | L* | a* | b* | WI | Hue (°) |
|---|---|---|---|---|---|
| a L* – lightness; a* – red-green value; b* – yellow-blue value; WI – whiteness index; hue (°) – colour tone. Results are expressed as mean ± SD (n = 3). Different superscript letters within a column indicate significant differences (p < 0.05). | |||||
| Control | 72.50a ± 0.30 | 1.10a ± 0.10 | 5.30a ± 0.20 | 89.00a ± 0.40 | 87.80a ± 0.50 |
| ICA | 71.70a ± 0.40 | 0.07a ± 0.10 | 2.80a ± 0.20 | 78.20a ± 0.45 | 73.30a ± 0.40 |
| ICC | 93.00b ± 0.20 | 0.67a ± 0.10 | 0.50b ± 0.10 | 60.50b ± 0.35 | 62.10a ± 0.30 |
| ICP | 82.20a ± 0.30 | 2.20a ± 0.10 | 0.43b ± 0.20 | 82.50b ± 0.40 | 29.90a ± 0.40 |
| Sample | Control | ICA | ICC | ICP |
|---|---|---|---|---|
| a ICA – amylase-infused ice cream; ICC – cellulase-infused ice cream; ICP – pectinase-infused ice cream. Results are expressed as mean ± SD (n = 3). | ||||
| Appearance | 6.9 ± 0.30 | 7.2 ± 0.42 | 8.0 ± 0.47 | 7.1 ± 0.30 |
| Flavor | 7.1 ± 0.30 | 7.0 ± 0.00 | 7.5 ± 0.00 | 7.0 ± 0.00 |
| Texture | 7.4 ± 0.52 | 8.1 ± 0.52 | 8.2 ± 0.42 | 7.8 ± 0.42 |
| Creamy | 7.1 ± 0.30 | 7.0 ± 0.00 | 8.0 ± 0.42 | 7.0 ± 0.00 |
| Sweetness | 8.0 ± 0.30 | 7.0 ± 0.00 | 7.0 ± 0.00 | 7.6 ± 0.50 |
| Milky | 8.4 ± 0.52 | 7.6 ± 0.50 | 7.4 ± 0.33 | 7.2 ± 0.47 |
| Overall acceptability | 7.8 ± 0.42 | 7.4 ± 0.50 | 8.0 ± 0.00 | 7.4 ± 0.00 |
Pectinase-treated ice creams (ICP) showed a strong red colour (a* = 2.20) with the lowest hue angle (29.90°), indicating the optimal anthocyanin release. This aligns with the enzyme's ability to degrade pectic polysaccharides, loosening plant cell structures and enhancing pigment diffusion.56 ICP also maintained high lightness (L* = 82.20) and moderate WI (82.50), resulting in a uniformly coloured and creamy appearance. Sensory attributes such as texture (7.8), appearance (7.1), and sweetness (7.6) were favorable, though slightly lower creaminess (7.0) and milky perception (7.2) suggested that excessive pectin breakdown may have reduced stabilizing effects within the matrix. These findings demonstrate that cellulase had the most pronounced effect on overall quality, improving both pigment extraction and sensory texture. Amylase enhanced sweetness naturally but contributed least to colour development. Pectinase was most effective in enhancing red pigmentation, but slightly compromised the body. These enzyme-specific effects on starch, cellulase, and pectin structures directly influenced anthocyanin release, viscosity, texture, and flavour perception, supporting previous research on enzyme-assisted functional ice creams.37,53–56
3.6. Spectroscopic and chromatographic analysis
| Peak | Retention time (min) | Area | Height | Relative concentration (%) | Assignment |
|---|---|---|---|---|---|
| 1 | 4.001 | 64![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 556 556![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
53![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 330 330 |
52.87 | Quercetin |
| 2 | 4.019 | 57![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 554 554![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 320 320 |
16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 833 833 |
47.13 | Quercetin standard |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching (amide I) and C
O stretching (amide I) and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching in aromatic rings, which is characteristic of phenolic acids and residual proteinaceous components. This aligns with previous reports that enzymatic extraction can liberate phenolics and other associated bioactive compounds.43,44 The absorption features in the region of 1200–1000 cm−1 correspond to the C–O and C–O–C stretching vibrations, typical of carbohydrates, glycosides, and polysaccharide structures present in roselle. The weak band in the lower wavenumber region around 420.28 cm−1 represents skeletal vibrations of complex biomolecular matrices or mineral-associated signals commonly found in plant-based extracts.45 Overall, the FTIR spectrum confirms that cellulase-assisted extraction of roselle calyces retains a diverse range of functional groups associated with phenolic, proteinaceous, and carbohydrate-based compounds. This molecular diversity supports the enhanced bioactivity of roselle extracts obtained through enzymatic treatment, as documented in related studies.19,47
C stretching in aromatic rings, which is characteristic of phenolic acids and residual proteinaceous components. This aligns with previous reports that enzymatic extraction can liberate phenolics and other associated bioactive compounds.43,44 The absorption features in the region of 1200–1000 cm−1 correspond to the C–O and C–O–C stretching vibrations, typical of carbohydrates, glycosides, and polysaccharide structures present in roselle. The weak band in the lower wavenumber region around 420.28 cm−1 represents skeletal vibrations of complex biomolecular matrices or mineral-associated signals commonly found in plant-based extracts.45 Overall, the FTIR spectrum confirms that cellulase-assisted extraction of roselle calyces retains a diverse range of functional groups associated with phenolic, proteinaceous, and carbohydrate-based compounds. This molecular diversity supports the enhanced bioactivity of roselle extracts obtained through enzymatic treatment, as documented in related studies.19,47
| Wavenumber (cm−1) | Assigned functional group | Possible compound class |
|---|---|---|
| 420.28 | Aromatic skeletal vibration/ring deformation | Aromatic phenolic rings, anthocyanidin core structures, mineral–organic complexes |
| 1633.00 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (amide I) stretching and C O (amide I) stretching and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic stretching C aromatic stretching |
Protein amide groups, hydroxycinnamic acids, anthocyanins, conjugated phenolic structures |
| 2163.55 | Weak C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N or C N or C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C-related overtone/combination band C-related overtone/combination band |
Trace nitrogenous residues, minor plant secondary metabolites, or instrumental artifacts |
| 3303.30 | O–H and N–H stretching (broad, H-bonded) | Flavonoids, anthocyanins (glycosylated OH), polysaccharide hydroxyls, proteinaceous NH groups |
4 Conclusion
This study demonstrated that enzyme-assisted extraction using amylase, pectinase, and cellulase significantly enhances the recovery of bioactive compounds from Hibiscus sabdariffa L. calyces compared to conventional techniques. Among the enzymes, cellulase produced the highest total phenolic and flavonoid yields, pectinase maximized anthocyanin recovery and colour intensity, while amylase enhanced antioxidant activity. Incorporating enzyme-treated roselle extracts into ice cream formulations improved the colour uniformity, texture, and sensory acceptability, demonstrating practical potential for developing functional and visually appealing food products. The spectroscopic and chromatographic analysis confirmed the preservation of key compounds such as quercetin, validating the efficiency of the optimized enzymatic process. Overall, the study highlights enzyme-assisted extraction as an eco-friendly, scalable, and energy-efficient approach for producing natural colorants and antioxidant-rich ingredients from roselle. The future research should focus on scaling up extraction for industrial applications, exploring synergistic enzyme combinations and assessing the stability and bioavailability of extracted compounds during product processing and storage. Additionally, integrating advanced green technologies such as ultrasound or microwave-assisted enzymatic extraction could further enhance the yield and sustainability, paving the way for broader use of roselle in functional foods, nutraceuticals, and natural product formulations.Consent for publication
All authors agreed on the publication of this research work.Author contributions
Priyanka A: conceptualization, methodology, writing – original draft. Nila Shireen B: validation, writing – original draft, formal analysis. Ganga Kishore: investigation, validation, writing – original draft, formal analysis. Sudha Paramadhas: conceptualization, investigation, supervision, writing – original draft, formal analysis. S. R. Rahul: writing – original draft, validation, formal analysis. Madhuresh Dwivedi: methodology, writing – original draft, review and editing, formal analysis. Pandiarajan T: supervision. Divyabharathi R: validation, investigation, formal analysis. Nongmaithem Seema Chanu: formal analysis.Conflicts of interest
The authors declare no competing interest.Abbreviations
| Hibiscus sabdariffa L. | Hibiscus sabdariffa Linn. |
| HCl | Hydrochloric acid |
| HPLC | High-performance liquid chromatography |
| FTIR | Fourier transform infrared |
| SEM | Scanning electron microscope |
| EAE | Enzyme-assisted extraction |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| TPC | Total phenol content |
| TFC | Total flavonoid content |
| TAC | Total anthocyanin content |
| AOX | Antioxidant activity |
| CE | Catechin equivalent |
| MSNF | Milk solid-not-fat |
| ICA | Ice cream with amylase-treated extract |
| ICC | Ice cream with cellulase-treated extract |
| ICP | Ice cream with pectinase-treated extract |
| cyn-3-sam | Cyanidin-3-sambubioside |
| dp-3-glu | Delphinidin-3-glucoside |
| cyn-3-glu | Cyanidin-3-glucoside |
| CCD | Central composite design |
| ANOVA | Analysis of variance |
| FWHM | Full width at half maximum |
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Acknowledgements
Funded by the ICAR-AICRP-PHET scheme.References
- J. Golijan and B. Dimitrijević, Acta Agric. Serb., 2018, 23, 125–140 Search PubMed.
- L. T. Raynolds, World Dev., 2004, 32, 725–743 CrossRef.
- R. Banc, L. Filip, A. Cozma-Petruţ, D. Ciobârcă and D. Miere, Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca. Food Sci. Technol., 2024, 81, 1–17 CAS.
- R. Bocker and E. K. Silva, Food Chem.:X, 2022, 15, 100398 CAS.
- N. Li, J. E. Simon and Q. Wu, Food Chem., 2024, 461, 140782 CrossRef CAS PubMed.
- H. A. Sindi, L. J. Marshall and M. R. A. Morgan, Food Chem., 2014, 164, 23–29 Search PubMed.
- T. Estiasih, J. E. Witoyo, G. K. M. Runtung, D. D. P. Moelyono, A. R. Murini, W. Setyaningsih and K. Ahmadi, CyTA–J. Food, 2024, 22, 2356847 CrossRef.
- A. A. Abou-Arab, F. M. Abu-Salem and E. A. Abou-Arab, Am. J. Sci., 2011, 7, 445–456 Search PubMed.
- P. J. Tsai, J. McIntosh, P. Pearce, B. Camden and B. R. Jordan, Food Res. Int., 2002, 35, 351–356 Search PubMed.
- N. Mahadevan, S. Shivali and P. K. Pradeep Kamboj, Nat. Product. Radiance, 2009, 8, 77–83 Search PubMed.
- M. Cissé, P. Bohuon, F. Sambe, C. Kane, M. Sakho and M. Dornier, J. Food Eng., 2012, 109, 16–21 Search PubMed.
- M. Dyrby, N. Westergaard and H. Stapelfeldt, Food Chem., 2001, 72, 431–437 Search PubMed.
- M. Mardiah, N. Novidahlia, M. Khoirunnisa, H. Hanafi and A. Aminullah, J. Teknol. Ind. Has. Pertan., 2021, 26, 65–71 Search PubMed.
- N. Stanek-Wandzel, A. Krzyszowska, M. Zarębska, K. Gębura, T. Wasilewski, Z. Hordyjewicz-Baran and M. Tomaka, Int. J. Mol. Sci., 2024, 25, 13538 Search PubMed.
- M. Spagnuolo, C. Crecchio, M. D. R. Pizzigallo and P. Ruggiero, Bioresour. Technol., 1997, 60, 215–222 Search PubMed.
- F. Chemat, N. Rombaut, A. G. Sicaire, A. Meullemiestre, A. S. Fabiano-Tixier and M. Abert-Vian, Ultrason. Sonochem., 2017, 34, 540–560 Search PubMed.
- A. Escobar-Ortiz, E. Castaño-Tostado, N. E. Rocha-Guzmán, J. A. Gallegos-Infante and R. Reynoso-Camacho, J. Sci. Food Agric., 2021, 101, 110–119 CrossRef CAS PubMed.
- M. M. K. Chowdhury, S. Chowdhury, L. Bari and S. Yeasmin, Dhaka Univ. J. Biol. Sci., 2024, 33, 139–147 CrossRef.
- K. N. Yulianda Setyawan and K. Kartini, J. Appl. Biol. Biotechnol., 2023, 11, 91–97 Search PubMed.
- A. A. Harahap, K. Letare S and N. Hendrianie, AIP Conf. Proc., 2023, 2667, 020003 Search PubMed.
- A. N. Nunes, A. Borges, A. A. Matias, M. R. Bronze and J. Oliveira, Molecules, 2022, 27, 368 Search PubMed.
- G. Linares and M. L. Rojas, Front. Nutr., 2022, 9, 891462 Search PubMed.
- D. L. Nonglait and J. S. Gokhale, Food Bioprocess Technol., 2024, 17, 1681–1705 CrossRef.
- I. Benucci, C. Lombardelli, C. Mazzocchi and M. Esti, Compr. Rev. Food Sci. Food Saf., 2022, 21, 2715–2737 CrossRef PubMed.
- J. J. Liu, M. A. A. Gasmalla, P. Li and R. Yang, Innovative Food Sci. Emerging Technol., 2016, 35, 184–193 CrossRef CAS.
- K. Rafińska, O. Wrona, A. Krakowska-Sieprawska, J. Walczak-Skierska, A. Kiełbasa, Z. Rafiński and B. Buszewski, Ind. Crops Prod., 2022, 187, 115424 CrossRef.
- D. Alavarsa-Cascales, M. J. Aliaño-González, M. Palma, G. F. Barbero and C. Carrera, Agronomy, 2022, 12, 2327 Search PubMed.
- M. José Aliaño González, C. Carrera, G. F. Barbero and M. Palma, Food Chem.:X, 2022, 13, 100192 Search PubMed.
- H. T. Ngoc Nhon, N. T. Diem My, V. N. Tuong Vi, P. T. Kim Lien, N. T. Thao Minh, L. N. Doan Duy, L. T. Hong Anh, D. T. Anh Dao and J. Agric, Food Res., 2022, 10, 100408 Search PubMed.
- P. R. Amulya and R. ul Islam, Food Chem.:X, 2023, 18, 100643 CAS.
- K. Duangmal, B. Saicheua and S. Sueeprasan, Proceedings of the AIC, 2004, pp. 155–158 Search PubMed.
- M. V. de Paiva, M. Z. Sanches, L. G. Chies, A. L. Vieira, J. F. Nicoleti and L. S. Sakanaka, Res. Soc. Dev., 2021, 10, e12910212224 Search PubMed.
- T. K. Koley, C. Kaur, S. Nagal, S. Walia and S. Jaggi, Arabian J. Chem., 2016, 9, S1044–S1052 Search PubMed.
- L. Jakobek and P. Matić, Croat. J. Food Sci. Technol., 2021, 13, 176–183 CrossRef.
- R. E. Wrolstad, R. W. Durst and J. Lee, Trends Food Sci. Technol., 2005, 16, 423–428 Search PubMed.
- N. Chongwilaikasem, P. Sithisarn, P. Rojsanga and P. Sithisarn, J. Food Sci., 2024, 89, 8819–8835 Search PubMed.
- T. M. Singo and D. Beswa, Int. J. Food Prop., 2019, 22, 42–53 CrossRef CAS.
- A. Al-Rifai, A. Aqel, A. Awaad and Z. A. ALOthman, Commun. Soil Sci. Plant Anal., 2015, 46, 1411–1418 CrossRef CAS.
- N. Montoya-Escobar, D. Ospina-Acero, J. A. Velásquez-Cock, C. Gómez-Hoyos, A. Serpa Guerra, P. F. Gañan Rojo, L. A. Vélez Acosta, C. Castro Herazo and R. Zuluaga Gallego, Polymers, 2022, 14, 5199 Search PubMed.
- U. Pithanthanakul, S. Vatanyoopaisarn, B. Thumthanaruk, C. Puttanlek, D. Uttapap, B. Kietthanakorn and V. Rungsardthong, Flavour Fragrance J., 2021, 36, 365–373 CrossRef CAS.
- L. A. Cira-Chávez, L. E. Gassós-Ortega, J. Nuñez-Vega, G. A. Camelo-Méndez, R. Cañedo-Urias, A. K. Blanco-Rios and M. I. Estrada-Alvarado, Appl. Food Res., 2025, 5, 101149 CrossRef.
- M. Mardiah, M. A. Fulazzaky, S. I. Rahmawati and M. Fulazzaky, Next Res., 2025, 2(3), 100536 CrossRef.
- Y.-J. Lai, Y.-C. Chiang, Y.-S. Jhan, T.-Y. Song and M.-C. Cheng, Foods, 2024, 13, 2172 CrossRef CAS PubMed.
- L. U. Chenxin, L. I. Yangxinhui, Y. Jinyi, G. Longyao and Z. Zhen, Food Mach, 2025, 40, 32–42 Search PubMed.
- U. K. M. Bangi, Sains Malays., 2023, 52, 3147–3162 CrossRef.
- T. Ahmed, M. R. Rana, M. A. Hossain, S. Ullah and M. Suzauddula, Biomass Convers. Biorefin., 2024, 14, 28985–28999 Search PubMed.
- N. N. Aneke, W. I. Okonkwo, S. L. Ezeoha, G. I. Okafor and C. N. Anyanwu, Res. Innov. Food Sci. Technol., 2023, 11, 437–450 Search PubMed.
- F. D'Heureux-Calix and N. Badrie, Food Serv. Technol., 2004, 4, 141–148 Search PubMed.
- A. Rani, Y. Arfat, R. S. Aziz, L. Ali, H. Ahmed, S. Asim, M. Rashid and C. H. Hocart, Environ. Technol. Innov., 2021, 23, 101620 CrossRef CAS.
- F. Weber and L. R. Larsen, Food Res. Int., 2017, 100, 354–365 CrossRef CAS PubMed.
- H.-K. Tran, T.-T.-H. Nguyen, T. T. Huynh, N. M. T. Vo, H. O. Le, D.-H. Truong, H.-C. Nguyen and C. J. Barrow, Int. J. Food Sci. Technol., 2025, 60(1) DOI:10.1093/ijfood/vvaf057.
- L. Wang, Y. Wu, Y. Liu and Z. Wu, Molecules, 2017, 22, 1648 CrossRef PubMed.
- A. Homayouni Rad, F. Karbalaei, M. A. Torbati, M. Moslemi, F. Shahraz, R. Babadi and M. Javadi, J. Food Sci. Technol., 2022, 59, 735–744 CrossRef CAS PubMed.
- C. Malalis, M. Barquilla, V. Zabala, K. Casas, M. Maye and A. J. O. Y. Ramayan, J. Food Nutr. Res., 2024, 63(4) DOI:10.26596/wefgm627.
- K. Bandara and T. Mahendran, J. Technol. Value Addit., 2020, 2, 97–109 Search PubMed.
- A. Gengatharan, G. Dykes and W. S. Choo, J. Food Sci. Technol., 2021, 58, 1401–1410 CrossRef CAS PubMed.
- Y. Zhao, J. Xu, Y. Zheng, Q. Li, Y. Huang, M. Zong, H. Wu and N. Li, Molecules, 2024, 29, 478 CrossRef CAS PubMed.
- S. Wang, Z. Hua, T. Wang, G. Yu and Y. Sun, Foods, 2025, 14, 292 Search PubMed.
- W. Sorndech, C. Auranwiwat, T. Ploypetchara, S. Butseekhot and J. Agric, Food Res., 2024, 18, 101310 CAS.
- T. Xiao, M. Sun, S. Cao, J. Hao, H. Rao, D. Zhao, Y. Dong and X. Xu, Food Chem.:X, 2024, 22, 101269 Search PubMed.
- J. I. Goldstein, D. E. Newbury, P. Echlin, D. C. Joy, C. E. Lyman, E. Lifshin, L. Sawyer and J. R. Michael, Scanning Electron Microscopy and X-Ray Microanalysis, Springer US, Boston, MA, 2003 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |