 Open Access Article
Open Access ArticleOptimizing UV-C irradiation for microbial safety and quality of Kinnow fruit juice
Vimal Challana *a,
Ankita Naykodia,
Shivanand S. Shirkole
*a,
Ankita Naykodia,
Shivanand S. Shirkole ab and
Akshaya K. Sahoo*a
ab and
Akshaya K. Sahoo*a
aDepartment of Food Engineering and Technology, Institute of Chemical Technology Mumbai, ICT-IOC Campus, Bhubaneswar, 751013, India. E-mail: ak.sahoo@iocb.ictmumbai.edu.in; vimalchallana@gmail.com
bDepartment of Food Technology, School of Engineering and Technology, D. Y. Patil Agriculture and Technical University, Talsande, Kolhapur, 416112, India
First published on 22nd April 2026
Abstract
The study analyzed UV-C irradiation as a non-thermal preservation method for Kinnow fruit juice (KFJ), aiming to reduce microbial contamination while preserving vitamin C content. By eliminating the need for thermal processing and chemical preservatives, the approach promotes energy-efficient and environmentally sustainable juice preservation. Using Response Surface Methodology (RSM), optimal UV exposure (25 minutes at 5 cm) predicted 34.056% vitamin C loss and 3.539 and 3.485 log reductions of AM and YM, respectively. The ANN model demonstrated superior performance over RSM, indicating that the ANN gives more accurate and reliable predictions. First-order kinetic modeling (R2 = 0.983) shows better fittings than the Weibull model. Shelf-life studies revealed that KFJ stored at 4 °C in glass containers retained higher vitamin C and microbial stability, extending product shelf-life by nearly 6 days. These findings suggest that UV-C processing, in combination with proper packaging and storage conditions, holds the potential for juice preservation at the commercial level.
Sustainability spotlightThis research advances sustainable food preservation by optimizing UV-C irradiation as a non-thermal method for enhancing the microbial safety and quality of Kinnow fruit juice. By reducing microbial load and maintaining vitamin C content without the use of heat or chemical preservatives, the process offers an energy-efficient and environmentally friendly alternative to conventional thermal pasteurization. The integration of modeling tools such as RSM and ANN ensures process precision, while shelf-life evaluation under refrigerated storage demonstrates extended product stability by nearly six days, effectively reducing post-harvest and processing losses. Through improved resource efficiency, waste reduction, and adoption of low-energy technologies, this study contributes to SDG 2 (Zero Hunger) by promoting food availability through longer shelf life, SDG 9 (Industry, Innovation, and Infrastructure) by applying innovative non-thermal and data-driven processing approaches, and SDG 12 (Responsible Consumption and Production) by encouraging sustainable food manufacturing practices that minimize environmental impact. |
1 Introduction
Kinnow is a cross between ‘King’ (Citrus nobilis) and ‘Willow Leaf’ mandarin (Citrus deliciosa) and belongs to the Rutaceae family and is extensively cultivated in India and Pakistan. According to the Department of Agriculture & Farmers Welfare,1 citrus fruit production in India reached 14.81 million metric tonnes, with cultivation over 1.095 million hectares. Major citrus fruits grown in India include Kinnow, mosambi, orange, lemon, lime, grapefruit, tangerine, and galgal, mainly suitable for Southeast Asian agro-climatic conditions. In India, its production is mainly in regions such as Punjab, Rajasthan, Himachal Pradesh, Jammu & Kashmir, Haryana, and Uttarakhand. Kinnow—a hybrid mandarin variety which is extensively cultivated in Punjab, with annual yields reaching 1.2 million metric tonnes. Kinnow is recognized by its medium-sized, oblate fruits with smooth, glossy orange skin and deep orange flesh.2,3 The fruit is highly cherished for its pleasant flavor, vibrant color and therapeutic properties. Kinnow fruit consists of components such as flavedo, albedo, seeds and juice sacs (present in segments).Kinnow is a seasonal fruit harvested from mid-December to mid-March, with peak yield in January and February. Kinnow fruit grows best at temperatures of 10–35 °C and with an annual rainfall of 500–1200 mm, moderate humidity levels (60–70%), and 6–8 hours of daily sunlight. However, the abundance of fruit during this period results in significant post-harvest losses if not managed properly. Due to its high water content, Kinnow is highly susceptible to microbial attacks.4 Harvest and post-harvest losses of Kinnow account for 25–30%.5
Various processing techniques, such as ultrafiltration, can be employed to minimize the losses and extend its availability.4 Kinnow juice is a primary processed product from a ripened fruit rich in vitamin C, folate, provitamin A, and other nutrients, such as asparagine, arginine, aspartic acid, proline and glutamine-like amino acids. It also provides essential minerals, meeting the body's calcium, magnesium, phosphorus, manganese, iron, zinc, and copper requirements. Moreover, the juice is abundant in bioactive components like phenols, flavones, flavonoids, and limonoids, along with carotenoids, which can contribute to its health benefits.6 The juice from the Kinnow fruit can be extracted using hand-reaming and mechanical juice extractors. The extracted Kinnow fruit juice (KFJ) can be consumed in its natural or raw state and processed for extended shelf-life and numerous applications. KFJ is easily prone to microbial spoilage, which can limit its market value due to its limited shelf life. For enhancing the safety and stability of KFJ, it is crucial to eliminate both microbes and inherent enzymes. The presence of inherent enzymes can result in loss of color, flavor, texture and nutrients in Kinnow.
Microbial growth and enzymatic activity play an important role in the spoilage of fruit juices, mainly due to aerobic mesophiles (AM) and yeasts and molds (YM). Both conventional and emerging preservation techniques have been assessed to address this issue. Studies have reported the use of ohmic heating, pasteurization7 and combinations involving preservatives,8,9 ultrafiltration,4 and ozonation10,11 in retaining the juice quality. While thermal treatments can effectively inactivate spoilage organisms, they often come at the cost of reduced nutritional and sensory attributes. In contrast, non-thermal treatments preserve these qualities, which makes them a preferable alternative for juice stabilization. In the era of novel non-thermal preservation techniques, ultraviolet (UV) irradiation stands out as an effective and promising method for preservation. UV treatment provides microbial safety and helps in retaining nutritional and sensory attributes, making it a low-energy, cost-effective alternative to conventional thermal methods without using preservatives.12 Very limited literature is available on traditional heat treatment and non-thermal techniques like UV processing for KFJ.
The mechanism of microbial inactivation is associated with UV light through multiple pathways. DNA damage is the primary cause behind microbial cell inactivation, while lipid peroxidation and protein denaturation contribute to the same. UV-C radiation induces thymine–thymine dimer formation, which can alter the bioactive properties of microbial cells. In addition, UV-C generates cyclobutane-pyrimidine dimers with cytotoxic effects and 6–4 photoproducts, which are highly lethal and mutagenic. Reactive oxygen species produced during UV treatment can lead to lipid oxidation, disrupting the lipid bilayer by forming pores and disintegrating the membrane. The treatment induces oxidative changes in amino acids, resulting in conformational and compositional changes that inhibit critical enzymes and disrupt cellular functions.13 Fig. 1 illustrates the mechanism behind microbial inactivation using UV light.
This study evaluates the impact of UV and thermal treatments on the quality characteristics of KFJ during storage. The juice was packaged in glass (GS) and polyethylene terephthalate (PET) and stored at two different temperatures (4 °C and room temperature). Key parameters, like ascorbic acid and microbial load, were evaluated to assess the quality degradation over storage time.
2 Materials and methods
2.1 Kinnow juice preparation
Fresh and ripe Kinnow fruit was bought from the local market in Bhubaneswar, Odisha, India. The fruits underwent cleaning and sorting, before storing the fruit at 4 °C until further analysis. The fruit peel was removed using a sanitized stainless-steel knife. The albedo portion was removed manually to separate the edible segments before juice extraction. The KFJ was extracted using a multifunctional food processor (Bajaj Electricals Limited, Model: FX11 600 Watts food processor). After extraction, the juice underwent filtration thrice using three-layered cheesecloth to separate juice from larger pulp particles and seeds.2.2 Description of the ultraviolet treatment system
The UV chamber was designed to provide uniform exposure to UV rays. The system consists of a cuboid closed chamber constructed from 20 mm white acrylic sheets, chosen for their durability, ease of cleaning, and high visibility to detect stains or spilled juice, which can aid in sterilization and contamination prevention. The chamber dimensions are 48 cm in length, 15 cm in breadth, and 24 cm in height (Fig. 2). A UV-C lamp (GHO436T5L, Arklite Specialty Lamps Pvt. Ltd, India) of 48 W power was installed inside the chamber. The distance between the juice and the lamp can be regulated through a movable base, ensuring consistent UV exposure to the KFJ. To maintain sterile conditions, the chamber was sanitized with 80% ethanol before any experimental run. The operation of the lamp was controlled manually using a switch and stopwatch to ensure accurate treatment durations.2.3 Experimental design and optimization
A face-centered central composite design optimized UV treatment conditions for maximum microbial reduction and minimizing vitamin C loss. The distance between the lamp and juice (Y1) and treatment time (Y2) were selected as the independent variables. In contrast, dependent variables assessed during the experiment design were total plate count (log reduction), yeast and mold (log reduction), and percentage loss of vitamin C. Independent parameters were evaluated at three distinct levels: distance at 3, 5, and 7 cm and treatment time at 15, 20, and 25 min. UV intensity was maintained constant throughout the experiment due to operational limitations of the UV system used; therefore, it was not included as an independent variable in the optimization study. The independent parameter limits were determined through preliminary trials. The experimental design and data analysis used Design-Expert software (version 13, Stat-Ease Inc., USA). 13 experimental runs were obtained, including 8 factorial runs, which represent different combinations of independent parameters, and 5 replicates at the center point to assess experimental error and ensure the reproducibility of the results.2.4 Thermal treatment
Thermal treatment (TT) was conducted using a water bath (Meta Lab Scientific Industries Ltd, Mumbai, India). The KFJ sample was transferred to sterile stainless-steel containers covered with lids. The containers were placed in a preheated water bath at 90 °C. The KFJ was held at 90 °C for 2 minutes after the temperature reached 90 °C. After pasteurization, the juice was immediately cooled to room temperature by immersion in an ice bath. The juice was transferred to GS and PET bottles in a sterile environment.2.5 Enumeration of microbial population
The AM and YM microbial populations in the KFJ were enumerated using the serial dilution-pour plate method. Fresh, UV and TT-treated KFJ samples were diluted with sterilized saline water (0.85% w/v sodium chloride in distilled water).14 The prepared dilutions were transferred as 1 mL of diluted inoculum to a sterile Petri plate, and 10–12 mL of agar was added. The Petri plate was rotated gently to ensure uniform mixing before the agar solidified. A second layer of 8–10 mL agar was poured to complete the plating process. Nutrient agar (HIMEDIA® M001-500 G) was used for AM, while potato dextrose agar (HIMEDIA® M096-500 G) was used for YM. The Petri plates were incubated for 24–36 hours at 37 °C for AM, while YM were incubated at 30 °C for 3–5 days. AM and YM can be expressed as colony-forming units per milliliter (CFU mL−1). The reduction in microbial load can be calculated in the form of log cycles using the following equation:
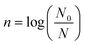 | (i) |
2.6 Vitamin C
Vitamin C was determined spectrophotometrically using 2,6-dichlorophenolindophenol at 518 nm, in accordance with Challana et al.15 The ascorbic acid content was expressed as milligrams per 100 mL of juice (mg AA/100 mL).2.7 Artificial neural network
The number of neurons in the hidden layer was determined through a trial-and-error approach to optimize network performance. The number of neurons varied from 1 to 20. A small number of neurons could result in underfitting, whereas excessive neurons can cause overfitting and prolong the training time. Before splitting the dataset into training (70%), validation (15%), and testing (15%) subsets, the datasets underwent randomization to ensure a representative distribution. The available dataset consists of 13 observations per variable, constrained by experimental data availability. To address these constraints and strengthen the model validation, a k-fold cross-validation strategy was employed. The strategy ensured that each data point contributed to training and testing, which can help overcome the effects of limited datasets and enhance the model's reliability and prediction accuracy. A comparative analysis was conducted between the ANN and RSM models to assess better fitting, predictive capability, and accuracy.15,16
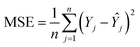 | (ii) |
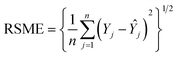 | (iii) |
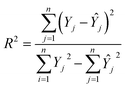 | (iv) |
2.8 Curve fitting for microbial inactivation
Microbial inactivation during UV treatment was described using first-order kinetics and the Weibull model.
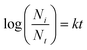 | (v) |
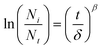 | (vi) |
β is the deciding factor for the Weibull distribution curve.
• β < 1 represents upward concavity, indicating microbial resistance and adaptation to UV stress over time.
• β > 1 represents downward concavity, suggesting accelerated microbial inactivation and increasing susceptibility with prolonged UV treatment.
• β = 1 represents a linear curve and reflects a constant microbial inactivation rate where the microbial inactivation is independent of exposure duration.
2.9 Shelf-life estimation
PET and GS bottles were the two packaging materials for storing and processing. A storage analysis was performed for fresh (JF), UV, and TT samples (JU and JT), with a primary focus on ensuring the microbiological safety of KFJ. The KFJ samples were unacceptable if the microbial count exceeded 6 log counts.19 Fresh samples (untreated juice), UV-treated samples, and TT samples were analyzed after 48 hours to evaluate the effect of different treatments. Fresh samples refer to untreated juice with no processing applied. UV-treated samples were those exposed to the optimized UV treatment conditions determined in this study. TT samples were processed at 90 °C for 2 minutes prior to storage. The shelf-life was determined by evaluating the critical parameters such as AM, YM, and vitamin C.3 Results and discussion
3.1 Effect of independent parameters on responses
The effect of independent variables and their interactions on dependent variable observations is provided in Table 1. The fresh KFJ has an AM count of 4.38 log CFU mL−1, a YM count of 5.12 log CFU mL−1, and a vitamin C concentration of 35.65 ± 2.75 mg of ascorbic acid/100 mL.| Run | Factor 1 | Factor 2 | Response 1 | Response 2 | Response 3 |
|---|---|---|---|---|---|
| Distance [cm] | Time [minute] | Aerobic mesophiles [log reduction] | Yeast and mold [log reduction] | Vitamin C [% loss] | |
| A | B | Y1 | Y2 | Y3 | |
| 1 | 7 | 15 | 2.279 | 2.159 | 21.842 |
| 2 | 5 | 20 | 3.169 | 2.987 | 32.554 |
| 3 | 7 | 25 | 2.371 | 3.415 | 25.127 |
| 4 | 5 | 20 | 3.315 | 3.214 | 29.895 |
| 5 | 5 | 15 | 2.314 | 2.359 | 29.639 |
| 6 | 5 | 20 | 3.478 | 3.150 | 30.588 |
| 7 | 7 | 20 | 2.314 | 2.987 | 27.856 |
| 8 | 3 | 15 | 3.059 | 2.538 | 38.430 |
| 9 | 5 | 25 | 3.812 | 3.536 | 34.295 |
| 10 | 5 | 20 | 3.515 | 3.090 | 27.845 |
| 11 | 3 | 20 | 3.840 | 3.369 | 43.799 |
| 12 | 3 | 25 | 3.989 | 3.599 | 50.235 |
| 13 | 5 | 20 | 3.418 | 2.895 | 31.114 |
| Factors | Aerobic mesophiles [log reduction] | Yeast and mold [log reduction] | Vitamin C [% loss] | |||
|---|---|---|---|---|---|---|
| (Y1) | (Y2) | (Y3) | ||||
| Coeff | p-Value | Coeff | p-Value | Coeff | p-Value | |
| a Superscript a represents a p-value less than 0.05 (p < 0.05), whereas superscript b represents a p-value greater than or equal to 0.1 (p ≥ 0.1). | ||||||
| Intercept | 3.356 | 3.085 | 30.880 | |||
| A | −0.654a | <0.001a | −0.158a | 0.009a | −9.607a | <0.001a |
| B | 0.420a | 0.005a | 0.582a | <0.001a | 3.291a | 0.065a |
| AB | −0.210b | 0.145b | 0.049b | 0.393b | −2.13b | 0.083b |
| A2 | −0.223b | 0.190b | 0.048b | 0.483b | 3.745a | 0.021a |
| B2 | −0.237b | 0.167b | −0.183a | 0.025a | −0.115b | 0.930b |
| Lack of fit | Not significant | Not significant | Not significant | |||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| ANOVA of the model | ||||||
| F-values | 13.09 | 39.90 | 30.61 | |||
| p-value(model) | 0.002 | <0.0001 | 0.001 | |||
| p-value(LOF) | 0.052 | 0.817 | 0.238 | |||
| R2 | 0.903 | 0.966 | 0.956 | |||
Eqn (vii) gives the correlation between the AM log reduction and independent parameters
| AM (log reduction) = +3.36 − 0.654 A + 0.42B − 0.21AB − 0.223 A2 − 0.237B2 | (vii) |
From the above equation, the negative coefficient of distance suggests that distance and reduction in AM are inversely proportional to each other, which means a decrease in distance between the UV lamp and KJF resulted in higher AM count reduction (Fig. 4A and B). Similarly, the positive coefficient of time suggests that exposure time is directly linked with a reduction in AM count. The trends align with the findings reported by Kim et al.,20 where reducing the sample's distance from the UV lamp and increasing the exposure time resulted in improved microbial reduction in lettuce. The trend could be ascribed to an increase in the distance, resulting in a lower intensity of UV, which diminished the antimicrobial effect, while prolonged treatment enhances the antimicrobial impact. The decrease in effectiveness with longer distances can be explained by the “inverse square law,” which states that doubling the distance resulted in a four-fold decrease in irradiance.21,22 As UV intensity diminishes with distance, prolonged treatment is required to achieve the same effect as at a closer distance.
The correlation between YM reduction and independent parameters is given below:
| YM (log reduction) = +3.09 − 0.158A + 0.582B + 0.049AB + 0.048A2 − 0.183B2 | (viii) |
From the above equation, the negative coefficient indicates that reducing the gap between the UV lamp and KFJ enhances YM reduction, demonstrating an inverse relationship as illustrated in Fig. 4(C and D). On the other hand, the positive coefficient of time suggested that longer exposure leads to a higher YM reduction. The negative coefficient of B2 indicates that while increasing treatment time improves the YM reduction, the effect decreases after a certain time, indicating a plateau or decline in effectiveness with longer exposure.
The reduction in YM is less pronounced than AM, indicating that UV treatment is more effective against AM, which can be attributed to the reduced pyrimidine nucleosides (cytosine and thymine) in the YM genetic material, along with the thicker cell wall of the YM in comparison to AM.23,24 These factors contribute to the higher resistance of yeast cells to UV treatment. Similar findings were reported by Torkamani and Niakousari25 in UV-treated orange juice, where bacteria were more affected than YM. This was likely due to their smaller size, simpler cell wall composition, and higher pyrimidine content, resulting in bacterial DNA being more prone to UV-induced damage.
| % Vitamin C loss = +30.88 − 9.61A + 3.29B − 2.13AB + 3.75A2 − 0.116B2 | (vi) |
The perturbation plot (Fig. 4(F)) highlights that distance was found to have a greater influence on vitamin C degradation than exposure time. The plot clearly illustrates that increasing the distance between the UV lamp and the sample reduces the vitamin C loss as the effectiveness of UV reduces with an increase in the gap. On the other hand, longer treatment time was directly linked with higher vitamin C degradation. The positive coefficient of A2 indicates an inverted parabolic response, which means that the loss of vitamin C initially increases with an increase in distance, but beyond a certain limit, the loss of vitamin C decreases.
Spectrophotometric analysis of KFJ (Fig. 5) revealed maximum absorption peaks mainly in the UV region (200–400 nm), with minimal absorption beyond 400 nm in the visible spectrum, which signifies that most of the light is transmitted in the visible range. This suggests that vitamin C degradation is more rapid in the UV region, with minimal impact from visible light. Similar findings were observed by de Souza et al.,26 who reported a decrease of nearly 23% in vitamin C content in citrus juice after UV-C treatment. The degradation could be attributed to the absorbance of ascorbic acid near 260 nm, which overlaps with the UV-C region, thereby accelerating its degradation. As vitamin C is very sensitive to light and temperature,27 prolonged exposure to UV accelerates its degradation when the distance between the juice and the minimum distance to the higher intensity of UV light results in more significant vitamin C loss.
The results show that distance is important in microbial inactivation and vitamin C retention. The increase in distance not only resulted in lower microbial inactivation but also helped in better retention of vitamin C, whereas the increase in treatment time improved the microbial inactivation and diminished the vitamin C concentration. These responses indicate an inverse relation between nutrient retention and microbial inactivation. Torkamani and Niakousari25 also reported that enhancing UV-C treatment resulted in microbial reduction but also caused loss of vitamin C. These findings highlight the inverse relationship between microbial load and nutrient retention.
The relative importance (r) values were assigned to prioritize the outcomes; r = 5 was set to minimize the vitamin C loss and r = 3 was set to maximize AM reduction. The overall desirability value (D) was used to evaluate the fitness of each solution. A higher D value means closer alignment with the desired objectives. The solution yielding the highest D value was selected. The optimized conditions were 5 cm distance between juice and the UV lamp and 25 minutes of exposure time with a D value of 0.826. The predicted responses were 34.056% vitamin C loss, 3.539 log reduction of AM, and 3.485 log reduction of YM. Hakguder Taze et al.28 reported that the UV-treated orange juice at an intensity of 1.32 mW cm−2 for 20 minutes resulted in a YM reduction of 1.76 log count at a dose of 108.42 mJ cm−2.
The experimental values for the log reduction of AM and YM were 3.311 ± 0.186 (93.56%) and 3.220 ± 0.170 (92.37%), respectively. Similarly, vitamin C loss was observed at 30.874 ± 0.657%, with an accuracy of 90.66%. Validation of responses under optimized conditions is presented in Table 3. These results confirm the reliability of the predictive models used for process optimization. Minor deviations between predicted and experimental values were within acceptable limits for practical applications.
| Optimized process | Predicteda | Experimentalb | Accuracy (%) |
|---|---|---|---|
| a Predicted value of dependent parameters with the help of quadratic equations.b Data are the average of 6 experiments under the expected optimum conditions of the independent variables. | |||
| Aerobic mesophiles [log reduction] | 3.539 | 3.311 ± 0.186 | 93.56 |
| Yeast and mold [log reduction] | 3.485 | 3.219 ± 0.168 | 92.37 |
| Vitamin C [% loss] | 34.056 | 30.874 ± 0.657 | 90.66 |
3.2 Artificial neural network
 | ||
| Fig. 6 Comparison of ANN performance based on the least RMSE values for AM, YM (log reduction), and vitamin C loss with different hidden neuron numbers. | ||
Fig. 7A–C illustrates the ANN model's training progress, including MSE across training, validation and test data sets over the 7 epochs for AM, 20 for YM, and 13 for vitamin C loss. The best validation performance was observed with MSE values of 0.0202 (at epoch 1) for AM, 0.0199 (at epoch 16) for YM, and 7.431 (at epoch 7) for vitamin C loss. An overview of the training process for all models is illustrated in Fig. 7D–F. The ANN-predicted correlation coefficients (R) for the training, validation and testing phases were 0.9838, 0.8166, and 0.9897 for AM; 0.9668, 0.9631, and 0.9736 for YM; and 0.9108, 0.9880, and 0.9951 for vitamin C loss, respectively (Fig. 7G–I).
| Parameters | AM (log reduction) | YM (log reduction) | % Vitamin C loss | |||
|---|---|---|---|---|---|---|
| RSM | ANN | RSM | ANN | RSM | ANN | |
| MSE | 0.035 | 0.012 | 0.006 | 0.010 | 2.400 | 1.470 |
| RMSE | 0.187 | 0.110 | 0.079 | 0.101 | 1.549 | 1.210 |
| R2 | 0.903 | 0.967 | 0.966 | 0.946 | 0.956 | 0.973 |
Although the RSM model showed a marginal advantage in predicting YM (log reduction), it had a lower MSE (0.006) and RMSE (0.079). In addition, the ANN model retained a strong R2 value (0.946 vs. 0.966), which signifies that the ANN is still a reliable approach. It can be concluded that the ANN model demonstrated superior performance in handling complex, non-linear associations30 and offers enhanced predictive accuracy for microbial and quality parameters and RSM is limited to second-order polynomial approximations.
3.3 Microbial inactivation modeling for ultraviolet treatment
The microbial inactivation modeling for UV treatment was performed using first-order kinetics and the Weibull distribution model. Table 5 shows that the first-order kinetics model (R2 = 0.983) fits better than the Weibull distribution model (R2 = 0.979). The inactivation rate constant (k) for first-order kinetics was determined to be 0.108, suggesting a steady microbial inactivation over time. In the Weibull distribution model, α (the scale parameter) was 14.14 and β (the shape parameter) was 1.403. β > 1 predicts downward concavity, suggesting accelerated microbial inactivation and increasing susceptibility with prolonged UV exposure. This suggests that the microorganisms become more vulnerable with time when exposed to UV due to cumulative damage induced by UV light. Both models effectively describe the inactivation of microbes, but the Weibull model provides an extra edge in varying resistance patterns.| Parameters | First-order kinetics | Weibull distribution model |
|---|---|---|
| R2 | 0.983 | 0.979 |
| Equation | y = −kx | y = −(x/α)β |
| Parameters | k = 0.108 | α = 14.14 |
| β = 1.403 |
3.4 Shelf-life
The AM and YM counts of untreated KFJ were 4.29 and 5.12 log CFU mL−1, respectively. In the JF (control) sample, AM exceeded 6.0 log CFU mL−1 after 8 days (at 4 °C) and >2 days (at 25 °C), while YM crossed the threshold level after 2 days (at 4 °C) and <2 days (at 25 °C) during storage. Under refrigeration, UV-treated juice samples exhibited a shelf life of >18 days in PET and <20 days in glass bottles, while TT extended the shelf life to nearly 24 days in both PET and glass bottles. In comparison, at room temperature, UV-treated juice demonstrated reduced microbial stability, lasting only >6 days in glass bottles. TT juice demonstrated comparatively higher stability, with a shelf life of >8 days in PET and <8 days in glass bottles for AM. The thermally treated juice showed higher stability for YM. The KFJ lasts for nearly 20 days (at 4 °C) and up to 8 days (at 25 °C) while stored in glass bottles. In contrast, UV-treated KFJ has lower stability, whereas juice has nearly 16- and 8-day stability under refrigeration and room temperature storage in b. The findings demonstrated that storage at ambient temperature compromises treatment efficacy, which could be attributed to enhanced proliferation of yeast under these conditions in contrast to refrigerated storage.33
The juice samples in glass bottles have lower microbial counts than those in PET bottles, ascribed to GS's excellent oxygen and moisture barrier properties.34 The reduced water vapor and transmission rate of glass effectively retards microbial growth by minimizing the external contamination, which is essential for microbial growth. In addition to that, UV treatment has been shown to extend the shelf-life of fruit juices like starfruit,35 watermelon,36 orange,37 and apple and grape juices.38 The effect becomes more pronounced when the juice is stored under refrigerated conditions, suppressing microbial metabolism.39 Thus, the combination of glass packaging and UV treatment, along with optimal storage conditions, plays an important role in enhancing the shelf life of KFJ.
The juice samples stored in glass bottles exhibited higher AA values, which could result from their superior barrier characteristics. This protective effect hindered oxygen permeation and minimized the conversion of ascorbic acid to dehydroascorbic acid. In contrast, juice stored in PET bottles had lower concentrations of vitamin C due to the limited barrier properties of PET against moisture and gases, which allowed oxygen migration into the product, thereby promoting the oxidative degradation of vitamin C.34,40
Conclusion
The study optimized UV irradiation as a non-thermal treatment to lower the vitamin C loss and maximize AM's log reduction. The optimal conditions chosen were 25 minutes of UV exposure at a distance of 5 cm between juice and the UV lamp, which resulted in a predicted vitamin C loss of 34.06%, 3.54, and 3.49 log reduction of AM and YM, respectively. Experimental validation showed accuracies of 90.66% for vitamin C loss, 92.37% for YM reduction, and 93.56% for AM reduction. The ANN model (2-15-1 for AM and 2-13-1 for vitamin C loss) has better predictive ability, fitting, and accuracy than RSM. The Weibull distribution model provided a superior fit (R2 = 0.979) compared to the first-order kinetics model (R2 = 0.983). The shelf-life study indicated that KFJ stored at 4 °C in glass bottles exhibited enhanced microbial and vitamin C stability compared to room temperature in PET bottles. Notably, the shelf-life of the JU-GS sample was improved by nearly six days compared to the untreated sample.However, some limitations should be acknowledged. Microbial analysis was limited to aerobic mesophiles and yeast and mold counts, and further evaluation of specific foodborne pathogens such as E. coli, Salmonella, and L. monocytogenes would provide a more comprehensive assessment of microbial safety. In addition, shelf-life assessment mainly considered microbial stability and vitamin C retention, while inclusion of other quality attributes such as phenolic compounds, flavonoids, antioxidant activity, and sensory characteristics could provide a broader understanding of the impact of UV treatment on juice quality.
Although the present work was conducted using a laboratory scale batch UV system, the optimized treatment conditions provide a useful basis for the development of pilot scale or continuous flow UV processing systems. Future studies should therefore focus on evaluating additional quality parameters, assessing pathogen inactivation, optimizing reactor design for continuous processing, and examining the industrial feasibility of UV treatment for commercial juice processing.
Author contributions
Vimal Challana: conceptualization, methodology, investigation, formal analysis, software, writing – original draft. Ankita Naykodi: writing – review & editing, supervision. Shivanand S. Shirkole: software, formal analysis, methodology, writing – review & editing. Akshaya K. Sahoo: conceptualization, methodology, supervision, project administration, validation, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Abbreviations
| KFJ | Kinnow fruit juice |
| AM | Aerobic mesophiles |
| YM | Yeast and molds |
| UV | Ultraviolet |
| GS | Glass |
| PET | Polyethylene terephthalate |
| CFU mL−1 | Colony-forming units per milliliter |
| JU | UV-treated juice |
| JT | Thermally treated juice |
| TT | Thermal treatment |
| AA | Ascorbic acid |
Data availability
The datasets used, analyzed, and generated during this study are included in the article.Acknowledgements
This research received no specific grant from any funding agency. The authors acknowledge the support and guidance provided by Jeet Pankaj Mehta, Prashant Mahesh Singh, Shubhada Gad, Nikita Sanjay Bhatkar and Keshram Dulait from Institute of Chemical Technology, Mumbai, ICT-IOC Campus, Bhubaneswar, India (751013).References
- Department of Agriculture & Farmers Welfare, Agricultural Statistics at a Glance 2022, 2023 Search PubMed.
- M. K. Mahawar, K. Jalgaonkar, B. Bibwe, B. Bhushan, V. S. Meena and R. K. Sonkar, Post-harvest processing and valorization of Kinnow mandarin (Citrus reticulate L.): A review, J. Food Sci. Technol., 2020, 57, 799–815 CrossRef CAS PubMed.
- R. Kumar, Kinnow: Punjab's king of fruit-A review, Just agriculture, 2020, 1, 294–298 Search PubMed.
- S. A. Ilame and S. V. Singh, Physico-chemical properties of ultrafiltered kinnow (mandarin) fruit juice, J. Food Sci. Technol., 2018, 55, 2189–2196 CrossRef CAS PubMed.
- S. V. Singh, R. K. Jain and A. K. Gupta, Adsorptive Reduction of Naringin from Kinnow Mandarin Juice with Non-ionic Macroporous Adsorbent Resin, Indian Chem. Eng., 2016, 58(2), 136–156 CrossRef CAS.
- S. S. Purewal and K. S. and Sandhu, Nutritional Profile and Health Benefits of Kinnow: An Updated Review, Int. J. Fruit Sci., 2020, 20, S1385–S1405 CrossRef.
- A. Bhattacharjee and S. Chakraborty, Design of a batch Ohmic heater and evaluating the influence of different treatment conditions on quality attributes of kinnow (Citrus nobilis × Citrus deliciosa) juice, Innovative Food Sci. Emerging Technol., 2022, 82, 103186 CrossRef CAS.
- R. Bhardwaj and S. Mukherjee, Effects of fruit juice blending ratios on kinnow juice preservation at ambient storage condition, Afr. J. Food Sci., 2011, 5, 281–286 CAS.
- R. Bhardwaj and U. Nandal, Effect of storage temperature on physico-chemical and sensory evaluation of kinnow mandarin juice blends, J. Food Process. Technol., 2014, 5, 1–4 Search PubMed.
- A. Iqbal, N. Muhammad, A. Ammara, A. Kashif, A. N. Muhammad, S. Muhammad, M. Faiqa and A. and Siddeeg, The impact of ozonation on the physicochemical properties, antioxidant potential and shelf life of Kinnow (Citrus Reticulata Blanco) juice, Int. J. Food Prop., 2022, 25, 2551–2560 CrossRef CAS.
- A. Iqbal, N. Muhammad, A. Ammara, Q. T. Mahmood, K. Waseem, M. Faiqa, R. Salim-Ur, R. Abdul, Z. K. Muhammad, A. Naushad, N. Asad and M. Ahmed, Quality evaluation of ozone-processed Kinnow (Citrus reticulata Blanco) juice at ambient temperature, Int. J. Food Prop., 2023, 26, 2420–2432 CrossRef CAS.
- V. Yemmireddy, A. Adhikari and J. Moreira, Effect of ultraviolet light treatment on microbiological safety and quality of fresh produce: An overview, Front. Nutr., 2022, 9, 871243 CrossRef PubMed.
- Y. Kebbi, A. I. Muhammad, A. S. Sant'Ana, L. do Prado-Silva, D. Liu and T. Ding, Recent advances on the application of UV-LED technology for microbial inactivation: Progress and mechanism, Compr. Rev. Food Sci. Food Saf., 2020, 19, 3501–3527 CrossRef PubMed.
- S. Chakraborty, P. S. Rao and H. N. Mishra, Effect of combined high pressure–temperature treatments on color and nutritional quality attributes of pineapple (Ananas comosus L.) puree, Innovative Food Sci. Emerging Technol., 2015, 28, 10–21 CrossRef CAS.
- V. Challana, A. M. Kaimal, S. Shirkole and A. K. Sahoo, Comparative analysis and investigation of ultrasonication on juice yield and bioactive compounds of kinnow fruit using RSM and ANN models, Sci. Rep., 2025, 15, 9859 CrossRef CAS PubMed.
- B. S. Kalsi, S. Sandhya, A. M. Shafiq and G. K. and Sidhu, Comparison of ANN and ANFIS modeling for predicting drying kinetics of Stevia rebaudiana leaves in a hot-air dryer and characterization of dried powder, Int. J. Food Prop., 2023, 26, 3356–3375 CrossRef CAS.
- R. Zalpouri, M. Singh, P. Kaur, S. Singh, S. Kumar and A. Kaur, Mathematical and artificial neural network modelling for refractance window drying kinetics of coriander (Coriandrum sativum L.) followed by the determination of energy consumption, mass transfer parameters and quality, Biomass Convers. Biorefin., 2025, 15, 967–983 CrossRef.
- M. A. J. S. van Boekel, On the use of the Weibull model to describe thermal inactivation of microbial vegetative cells, Int. J. Food Microbiol., 2002, 74, 139–159 CrossRef PubMed.
- S. Pipliya, S. Kumar and P. P. Srivastav, Impact of cold plasma and thermal treatment on the storage stability and shelf-life of pineapple juice: A comprehensive postharvest quality assessment, Food Phys., 2024, 1, 100025 CrossRef.
- Y.-H. Kim, S.-G. Jeong, K.-H. Back, K.-H. Park, M.-S. Chung and D.-H. Kang, Effect of various conditions on inactivation of Escherichia coli O157: H7, Salmonella Typhimurium, and Listeria monocytogenes in fresh-cut lettuce using ultraviolet radiation, Int. J. Food Microbiol., 2013, 166, 349–355 CrossRef CAS PubMed.
- S. Simmons, C. Dale, J. Holt, K. Velasquez and M. Stibich, in Ultraviolet Light in Human Health, Diseases and Environment, ed. S. I. Ahmad, Springer International Publishing, Cham, 2017, pp. 255–266, DOI:10.1007/978-3-319-56017-5_21.
- H. Nakashima, A. Utsunomiya, J. Takahashi, N. Fujii and T. Okuno, Hazard of ultraviolet radiation emitted in gas metal arc welding of mild steel, J. Occup. Health, 2016, 58, 452–459 CrossRef CAS PubMed.
- R. Shamsudin, N. M. Adzahan, Y. P. Yee and A. Mansor, Effect of repetitive ultraviolet irradiation on the physico-chemical properties and microbial stability of pineapple juice, Innovative Food Sci. Emerging Technol., 2014, 23, 114–120 CrossRef CAS.
- R. Shamsudin, N. Mohd Adzahan, Y. Pui Yee and A. Mansor, Effect of repetitive ultraviolet irradiation on the physico-chemical properties and microbial stability of pineapple juice, Innovative Food Sci. Emerging Technol., 2014, 23, 114–120 CrossRef CAS.
- A. Torkamani and M. Niakousari, Impact of UV-C light on orange juice quality and shelf life, Int. Food Res. J., 2011, 18, 1265 CAS.
- V. R. de Souza, V. Popović, S. Bissonnette, I. Ros, L. Mats, L. Duizer, K. Warriner and T. Koutchma, Quality changes in cold pressed juices after processing by high hydrostatic pressure, ultraviolet-c light and thermal treatment at commercial regimes, Innovative Food Sci. Emerging Technol., 2020, 64, 102398 CrossRef CAS.
- S. Sablani, L. Opara and K. Al-Balushi, Influence of bruising and storage temperature on vitamin C content of tomato fruit, J. Food, Agric. Environ., 2006, 4, 54 CAS.
- B. Hakguder Taze, S. Unluturk, S. Buzrul and H. Alpas, The impact of UV-C irradiation on spoilage microorganisms and colour of orange juice, J. Food Sci. Technol., 2015, 52, 1000–1007 CrossRef CAS PubMed.
- R. Zalpouri, M. Singh, P. Kaur, A. Kaur, K. K. Gaikwad and A. Singh, Drying Kinetics, Physicochemical and Thermal Analysis of Onion Puree Dried Using a Refractance Window Dryer, Processes, 2023, 11(3), 700 CrossRef CAS.
- B. S. Kalsi, S. Singh, M. S. Alam and G. K. Sidhu, Comparison of ANN and ANFIS modeling for predicting drying kinetics of Stevia rebaudiana leaves in a hot-air dryer and characterization of dried powder, Int. J. Food Prop., 2023, 26, 3356–3375 CrossRef CAS.
- S. Pipliya, S. Kumar and P. P. Srivastav, Modeling of the inactivation kinetics of aerobic mesophiles and yeasts and molds natural microbiota in nonthermal plasma-treated pineapple (Ananas comosus) juice, J. Food Sci., 2023, 88, 3905–3919 CrossRef CAS PubMed.
- A. C. Polydera, N. G. Stoforos and P. S. Taoukis, Comparative shelf life study and vitamin C loss kinetics in pasteurised and high pressure processed reconstituted orange juice, J. Food Eng., 2003, 60, 21–29 CrossRef.
- S. C. Andrés, L. Giannuzzi and N. E. Zaritzky, The effect of temperature on microbial growth in apple cubes packed in film and preserved by use of orange juice, Int. J. Food Sci. Technol., 2004, 39, 927–933 CrossRef.
- S. B. M. Jaime, R. M. V. Alves and P. F. J. Bócoli, Moisture and oxygen barrier properties of glass, PET and HDPE bottles for pharmaceutical products, J. Drug Delivery Sci. Technol., 2022, 71, 103330 CAS.
- R. Bhat, S. B. Ameran, H. C. Voon, A. A. Karim and L. M. Tze, Quality attributes of starfruit (Averrhoa carambola L.) juice treated with ultraviolet radiation, Food Chem., 2011, 127, 641–644 CrossRef CAS PubMed.
- M. Feng, K. Ghafoor, B. Seo, K. Yang and J. Park, Effects of ultraviolet-C treatment in Teflon®-coil on microbial populations and physico-chemical characteristics of watermelon juice, Innovative Food Sci. Emerging Technol., 2013, 19, 133–139 CrossRef CAS.
- Ç. U. Pala and A. K. Toklucu, Microbial, physicochemical and sensory properties of UV-C processed orange juice and its microbial stability during refrigerated storage, LWT–Food Sci. Technol., 2013, 50, 426–431 CrossRef.
- A. Müller, L. Noack, R. Greiner, M. R. Stahl and C. Posten, Effect of UV-C and UV-B treatment on polyphenol oxidase activity and shelf life of apple and grape juices, Innovative Food Sci. Emerging Technol., 2014, 26, 498–504 CrossRef.
- L. S. Briones, J. E. Reyes, G. E. Tabilo-Munizaga and M. O. Pérez-Won, Microbial shelf-life extension of chilled Coho salmon (Oncorhynchus kisutch) and abalone (Haliotis rufescens) by high hydrostatic pressure treatment, Food Control, 2010, 21, 1530–1535 CrossRef.
- A. Jacob, I. P. Sudagar, R. Pandiselvam, P. Rajkumar and M. Rajavel, Effect of packaging materials and storage temperature on the physicochemical and microbial properties of ultrasonicated mature coconut water during storage, Food Control, 2023, 149, 109693 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |







