Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Harnessing non-thermal techniques for sustainable protein extraction from blue foods and their potential applications in circular food systems
Nikashini
Thirugnanam†
 ab,
Monisha
Chandran†
a,
Pathare Ashutosh
Dattatrya
ab and
Loganathan
Manickam
*a
ab,
Monisha
Chandran†
a,
Pathare Ashutosh
Dattatrya
ab and
Loganathan
Manickam
*a
aNational Institute of Food Technology, Entrepreneurship and Management, Thanjavur (NIFTEM-T), Thanjavur, India. E-mail: logu@iifpt.edu.in
bSustainable Food Automation, Faculty of Sustainable Design Engineering, University of Prince Edward Island, Charlottetown, Prince Edward Island, Canada
First published on 17th March 2026
Abstract
Blue foods contain a diverse group of aquatic resources and are essential owing to their nutritional profile, including proteins, fatty acids and key micronutrients, for maintaining human health. Numerous chemical and biological elements exist in marine habitats, which make the ocean a perfect source of high-value compounds with several applications. Marine sources provide distinctive proteins and polysaccharides, along with micronutrients and bioactive compounds. Modern developments in non-thermal technologies have facilitated environmentally friendly, green and sustainable methods for the effective extraction of high-value proteins from marine resources, which support environmental sustainable goals. This review focused on the advantages of novel non-thermal treatments in the extraction of protein from blue foods. It evaluates new processing methods that reduce waste and enhance protein extraction efficiency to produce functional products. The analysis identified the obstacles encountered during the production of marine proteins. The main goal is to reduce environmental effects using unused marine biomass through energy-efficient methods, which also generate less waste. Non-thermal processing methods enhance marine protein functionality and convert waste materials into food-grade products to support circular bioeconomy models. These methods essentially benefit sustainable food systems by minimizing pollution and optimizing resource management. Sustainable marine protein extraction technologies support global biodiversity preservation and the development of climate-resistant food innovations.
Sustainability spotlightThis review advances sustainable food systems by exploring non-thermal, energy-efficient technologies for protein extraction, highlighting their role as a green technology as they do not have any negative impacts on the environment compared with thermal processing methods. Techniques such as ultrasound, pulsed electric field, high-pressure processing, and enzyme-assisted extraction help in minimizing waste generation and reducing chemical usage while improving yield and quality. By valorising marine biomass and underutilized aquatic species, this work supports the transformation of low-value by-products into high-value food ingredients, strengthening the foundation of a circular bioeconomy. The sustainable extraction of marine proteins also fosters biodiversity preservation and responsible resource management. This study aligns directly with the United Nations Sustainable Development Goals (SDGs), particularly SDG 12 (responsible consumption and production), SDG 13 (climate action), and SDG 14 (life below water), by promoting low-impact processing technologies that reduce environmental burdens. Furthermore, the proposed framework will serve as a basis for the exploration of the potential of integrating these eco-innovative technologies into industrial-scale applications, thereby reducing the dependence on land-based proteins and mitigating the greenhouse gas emissions associated with conventional processing. The implementation of such sustainable extraction methods can contribute to food and nutritional security, promote circularity in marine-based industries, and establish a model for green innovation within the global food sector. Overall, this review highlights how non-thermal technologies can serve as transformative tools to achieve climate-resilient, waste-free, and resource-efficient food production systems. |
1. Introduction
A prevalent perspective suggests that the production of aquatic sustenance, termed blue foods, obtained from marine and freshwater environments through capture or cultivation, must increase in the upcoming decades to meet the growing nutritional demands driven by population growth.1 The blue food protein sector has gained increased focus within the food sector due to its abundance of high-quality ingredients that boast exceptional nutritional value and beneficial properties for health. Blue foods encompass both wild and cultivated items from marine and freshwater environments. They have recently gained the attention of scientists, nutritionists, and private and public organizations. This interest stems from their potential to play a role in alleviating global hunger and ensuring nutritional security for humanity.2 On a global scale “blue foods” encompassing fish, invertebrates, and algae, obtained from marine ecosystems play a vital role in ensuring the food and financial stability of billions of individuals. As they serve as pivotal sources of proteins, fatty acids, and micronutrients, these blue foods are indispensable in addressing challenges related to malnutrition and diseases.3 The global consumption of blue foods has been steadily increasing, with per capita intake reaching approximately 20.2 kg in 2020. Blue foods contribute around 17% of the total animal protein supply worldwide, with their share exceeding 50% in several Asian and African countries. In many regions, blue foods are not only more accessible but also more affordable than other animal-sourced proteins.4 With the expansion of aquaculture, there is a corresponding need for aquafeeds that are both environmentally sustainable and cost-effective. This demand is projected to rise to 87 million tonnes by the year 2025.5 Consequently, their production is on the rise, making them one of the most traded commodities in the world. They also provide a range of job opportunities, life expectancy, and income globally.4Ironically, although certain marine resources face over-exploitation or inefficient utilization for human sustenance, other abundant marine food sources remain largely untapped or are utilized primarily for producing feed for aquaculture or terrestrial animals. In general, there is a substantial loss, typically exceeding 90% of nutritional value, such as proteins, at each trophic level in the food chain. Therefore, utilizing unexploited or underexploited blue foods directly for human consumption presents an ideal approach to optimize the use of our natural global resources and simultaneously address the challenge of feeding a growing population with nutritious food.6,7 The challenges of a growing global population and increasing demand for protein sources highlight the utilization of marine proteins, which present a promising avenue for sustainable food production. Beyond their nutritional contributions, blue foods are increasingly recognized within global sustainability frameworks, particularly under the United Nations 2030 Agenda for Sustainable Development. Blue food systems directly contribute to Sustainable Development Goals (SDGs) 2 (Zero Hunger), 12 (Responsible Consumption and Production), 13 (Climate Action), and 14 (Life Below Water). Recent evidence suggests that the strategic integration of blue foods into national dietary policies can simultaneously improve nutritional outcomes, reduce greenhouse gas emissions, and enhance livelihood resilience across coastal and low-income regions.8 The Food and Agriculture Organization has further emphasized “blue transformation” as a pathway to reorient aquatic food systems toward sustainability, equity, and circular resource use.9 Despite increasing production, blue food systems are characterized by significant inefficiencies, particularly during processing stages, where 30–70% of biomass may become underutilized by-products, such as heads, bones, skins, viscera, and shells. Conventional linear processing models often divert these residues toward low-value feed or landfill streams, resulting in nutrient losses and environmental burdens. Transitioning toward a circular bioeconomy framework, where protein, lipids, minerals, and bioactive compounds are sequentially recovered and valorised, can significantly reduce landfill burden, minimize nutrient loss, and improve overall resource efficiency.5,10 The valorization of fish processing waste not only enhances economic returns but also contributes to climate mitigation through reduced waste decomposition emissions.
Several studies published in the past decade have concentrated on the beneficial applications of different aquatic species and their by-products (mostly scales, bones, and skins) in collagen production. These are high-collagen materials derived from aquatic species. The primary sources of collagen in aquatic species (freshwater, deep-sea, and so forth) and processing circumstances (acid-aided, pepsin-aided, and so forth) were the primary sources of variation in aquatic species collagen.11 The proteins from blue foods play a vital role as a functional and nutritional property in the food chain. The protein influences characteristics such as water absorption, oil absorption, foaming, emulsion, solubility, and texture of food.12 Numerous food protein qualities can be improved or changed using non-thermal technology. Protein coagulation, aggregation, or gelation can be modulated by these shifts created from non-thermal effects, which enable varying degrees of unfolding and denaturation of the protein structure.13 Marine-derived proteins are broadly classified into myofibrillar, sarcoplasmic, and stromal proteins, each contributing distinctly to the nutritional and techno-functional properties of blue foods. Myofibrillar proteins, primarily myosin and actin, play a crucial role in gel formation, water-holding capacity, and textural attributes, while sarcoplasmic proteins influence solubility and emulsification behaviour. The functional performance of these proteins is governed by their structural organization, including primary amino acid sequences, secondary folding patterns, and higher-order conformations. Processing parameters such as pH, ionic strength, temperature, and mechanical stress can significantly alter protein structure through denaturation, aggregation, or unfolding, thereby influencing extraction efficiency and end-use functionality.13,14 Therefore, maintaining structural integrity during extraction is essential to preserve both nutritional quality and techno-functional characteristics. Recent studies have emphasized that advanced and non-conventional extraction technologies can enhance protein recovery while minimizing excessive structural degradation, thereby improving functional performance in food applications.15 In addition to the functionality of intact proteins, controlled hydrolysis of marine proteins leads to the generation of bioactive peptides with enhanced biological activities. These peptides are produced through specific enzymatic cleavage of parent proteins and have been reported to exhibit antioxidant, antihypertensive, antimicrobial, and immunomodulatory properties. The production of such peptides further expands the utilization potential of blue biomass within the food, pharmaceutical, and nutraceutical sectors.14
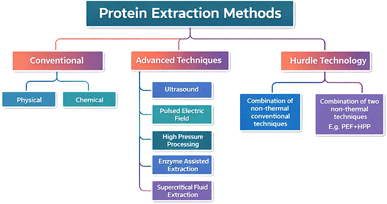
Several approaches have been developed to modify food proteins to improve their functional characteristics. These techniques include chemical methods (such as controlled enzymolysis and chemical modification), physical methods (such as ultrasound, high-pressure treatment, pulsed electric field, and solvent extraction), and hurdle techniques.16–18 Conventional protein extraction techniques, such as strong acid or alkali solubilization, although effective, often require high chemical inputs, extensive water usage, and subsequent neutralization steps that increase the environmental load. In contrast, non-thermal technologies, including ultrasound, pulsed electric field (PEF), high-pressure processing (HPP), and enzyme-assisted extraction, offer promising green alternatives. These approaches enhance cell disruption efficiency while operating at lower temperatures, thereby reducing energy demand and preserving protein functionality.14,15 Moreover, enzyme-based and physical extraction methods minimize hazardous effluent generation and align more closely with sustainable processing principles. These technologies, therefore, represent critical enablers in transitioning from linear extraction models to environmentally responsible protein recovery systems.
Although several studies have investigated protein extraction from marine organisms, previous studies predominantly focused on yield optimization and functional characterization, with limited integration of sustainability metrics, circular bioeconomy frameworks, and residue valorization strategies. Furthermore, the comparative evaluation of conventional versus non-thermal extraction technologies from an environmental perspective remains underdeveloped.14,19 There is a pressing need to critically examine how advanced extraction technologies can contribute to circular food systems while maintaining techno-functional performance. Therefore, this review aims to critically evaluate conventional and non-thermal protein extraction techniques from blue foods; analyse their sustainability implications, including energy demand and environmental footprint; explore circular bioeconomy integration through residue valorization pathways; and assess future opportunities within sustainable food systems.
2. Protein sources from blue foods
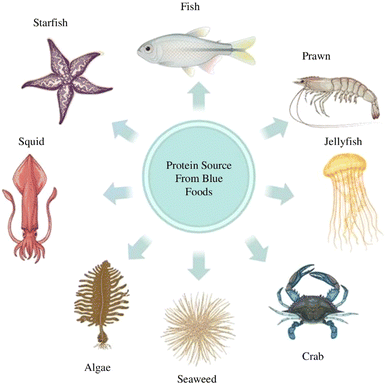
The vitality of marine organisms as originators of distinct proteins, fats, micronutrients, and bioactive substances continues to increase. Compounds obtained from marine organisms have emerged as a rich reservoir for exploring new peptides although they remain relatively obscure due to the challenges associated with their retrieval. Marine peptides, constituting 8% of all bioactive marine compounds, play a significant role. A notable proportion of novel compounds has been identified from marine invertebrates, marine algae, and marine vertebrates (Fig. 1) in this section.2.1. Algae-based sources for protein compounds
Most green and red macroalgae species have considerably higher protein content than terrestrial plant protein sources, such as soybeans, cereals, and nuts.20 In addition to their nutritional properties, proteins and peptides derived from marine algae have significant value because of their pharmaceutical, nutraceutical, and cosmeceutical properties, such as antioxidant, immune modulatory, anti-hypertensive, and hepato-protective properties.21 Brown seaweed has a low protein content (3–15% of dry weight), while green and red seaweed have modest and high protein contents (9–26% and 47% of dry weight, respectively). A few examples are Undaria pinnatifida, Ulva pertusa, Ulva lactuca, Porphyra yezoensis, Palmaria palmata, Laminaria digitata, Ulva rotundata, Ulva armoricana, Ulva rigida, Chondrus crispus, and Ulva ohndi.22,23Microalgae act as an important source of bioactive molecules. They are rich in carbohydrates, lipids, proteins, and minerals. Among microalgal species, some of the prominent classes are cyanophyceae (blue-green algae), bacillariophyceae (comprising diatoms), chrysophyceae (encompassing golden algae) and chlorophyceae (green algae).24 Compared with macroalgae, microalgae have an abundant number of proteins, which comprise around 70% of the biomass dry weight in some notable species.25 Out of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 species, only a few species of microalgae are considerably safe for human consumption, such as Spirulina or Arthrospira platensis, Chlorella or Chlorella vulgaris, Dunaliella, Nostoc, and Aphanizomenon.26 Microalgae act as an exceptionally prominent source of proteins for human consumption since the amino acid profile almost matches that of the human body.
000 species, only a few species of microalgae are considerably safe for human consumption, such as Spirulina or Arthrospira platensis, Chlorella or Chlorella vulgaris, Dunaliella, Nostoc, and Aphanizomenon.26 Microalgae act as an exceptionally prominent source of proteins for human consumption since the amino acid profile almost matches that of the human body.
2.2. Marine animal sources for protein compounds
Marine animal sources include vertebrates and invertebrates, like sea urchins, sea anemones, starfish, cuttlefish, crabs, prawns, squid, octopus, and jellyfish, and different variants of fish, viz. carp, tilapia, marine eel fish, amur sturgeon, red drum fish, big eye snapper and giant croaker.27–29 Nowadays, marine sources are scrutinized as one of the safest sources of protein. They also aid in reducing the risk of diseases like BSE (Bovine Spongiform Encephalopathy), TSE (Transmissible Spongiform Encephalopathy), and FMD (Foot and Mouth Disease), which may be caused by the consumption of land-based animal-derived protein. These marine animals have high amounts of collagen, which assists with the valorization of fish waste.27Aromatic (tryptophan, tyrosine, and phenylalanine) and nucleophilic amino acids with sulphur side chains (cysteine and methionine) are more efficient in terms of the bioactivity of fish hydrolysates because of their ability to easily donate hydrogen atoms and interact with free radicals.30 The most remarkable bioactivities of fish-based hydrolysates are determined by their amino acid composition as well as their structural and conformational properties.31 Taurine, a conditionally essential amino acid, is particularly intriguing. Taurine's potential has been tailored for use as a therapeutic agent against congestive heart failure and a variety of other conditions, including blood pressure reduction, improved cardiac performance, and blood cholesterol reduction.30
Fish proteins are rich in amino acids and peptides, making them highly digestible, especially in balanced proportions. The digestibility of amino acids and proteins is critical to the bioavailability and therapeutic efficacy of fish products.32 Protein hydrolysis can produce bioactive peptides, particularly those derived from various fish species. The protein in oily and white fish breaks down into polypeptides, amino acids, and peptides. Bioactive chemicals are so named because most have bioactive properties. Bioactive chemicals are additional nutritional ingredients found in foods that occur naturally in small amounts. These compounds are beneficial to human health.33
They can be transported through intestinal enterocytes before reaching the circulation, where they have beneficial biological effects. Peptides can deliver a wide range of biotechnological products with enhanced bioactivity, including antibacterial, antioxidant, and antihypertensive properties.34 Lysine and methionine are two essential amino acids found in large amounts in fish proteins. Angiotensin I converting enzyme (ACE) is a type of inhibitory peptide found in fish, originally in sardine flesh. Similarly, fatty fish have around 20% protein, but their water (62–70%) and oil levels are much higher (10–18%).35
3. Extraction of protein from blue foods
3.1. Conventional techniques for protein extraction
Conventional protein extraction methods include the pH shift process and solvent extraction. Acid and alkali solubilization methods are used for protein extraction under the pH shift process (Table 1). However, the most common extraction technique is the conventional technique discussed in the subsequent section (Fig. 2).| Sources | Method of extraction | Extraction conditions | Yield (%) | Reference |
|---|---|---|---|---|
| Algae | ||||
| Microalgae | ||||
| Aglaothamnion uruguayense | Alkali extraction | Addition of 1 mL of 0.1 N NaOH to the centrifuged pellet with occasional shaking | 22.38 | 36 |
| Hillea sp. | Alkali extraction | Addition of 1 mL of 0.1 N NaOH to the centrifuged pellet with occasional shaking | 25.04 | 36 |
| Amphidinium carterae | Alkali extraction | Addition of 1 mL of 0.1 N NaOH to the centrifuged pellet with occasional shaking | 24.06 | 36 |
| Haematococcus pluvialis | Ultrasonication | Frequency-20 kHz, 5 s cycles with 15 s of resting time for 30 min | 8.5 ± 0.0 | 37 |
| Porphyridium cruentum | Ultrasonication | Frequency-20 kHz, 5 s cycles with 15 s of resting time for 30 min | 67.0 ± 0.9 | 37 |
| Arthrospira platensis | High pressure homogenization (HPH) | 2700 bar pressure in two passes | 78.0 ± 2.8 | 37 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Macroalgae | ||||
| Ulva ohnoi | PEF coupled with a mechanical press | 200 pulses with a field strength of 1 kV cm−1, repetition rate and pulse duration 3 Hz and 50 µs, and combined with mechanical pressing at 107![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 682 N m−2 682 N m−2 |
14.94 | 23 |
| Ulva armoricana | Enzyme-assisted extraction | 6% endoprotease enzyme, soaked at 50 °C for 3 h | 41.40 | 38 |
| Chondracanthus chamissoi | Enzyme-assisted extraction | Cellulase enzyme, pH 4.5, for 12 h at 50 °C, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 enzyme-solvent ratio 10 enzyme-solvent ratio |
36.10 | 39 |
| Gracilaria sp. | Ultrasound-assisted alkaline extraction | Suspension of the sample in 10% (v/v) NaOH, sonication for 2 h, further filtration (0.45 µm), and dialysis (2 kDa) is done | 86 | 40 |
| Ascophyllum nodosum | Sequential acid-alkaline extraction | 0.4 M HCl for 1 h at 4 °C and 0.4 M NaOH for 1 h at 4 °C | 59.75 | 41 |
| Fucus vesiculosus | HPP | 600 MPa for 40 min, followed by filtration | 23.7 | 42 |
| Chlorella vulgaris | PEF with bead milling | 14 pulses with a field strength of 20.6 kV cm−1 and tip speed of 8 m s−1 | ∼30 protein release | 43 |
| Neochloris oleoabundans | PEF with bead milling | 14 pulses with a field strength of 19.7 kV cm−1 and tip speed of 8 m s−1 | ∼50 protein release | 43 |
| Ulva sp. | Enzyme-assisted PEF extraction | Enzyme pretreatment with Cellulase Onzuka R-10, 30 pulses with the field strength of 1 kV cm−1, pulse duration, and repetition rate is 30 µs and 5 Hz | 19.6 ± 0.33 | 44 |
| Gracilaria verrucosa | Enzymatic extraction | Agarase + cellulase, 2 h and 14 h incubation | 63%–2 h | 45 |
| 21%–14 h | ||||
| Chondrus crispus | Enzymatic extraction | Carrageenase + cellulase, 2 h and 14 h incubation | 32%–2 h and 14 h | 45 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Marine animals | ||||
| Yellowfin Tuna | Acid extraction | 0.5 M acetic acid in a sample-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 for 48 h at 4 °C 10 for 48 h at 4 °C |
1.07 | 46 |
| Thunnus albacares | ||||
| Grass carp | Acid extraction | 0.5 M acetic acid in a sample-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 for 72 h at 4 °C 10 for 72 h at 4 °C |
90 | 47 |
| Ctenopharyngodon idella | ||||
| Catla | Acid extraction | 0.5 M acetic acid in a sample-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 for 24 h at 4 °C 10 for 24 h at 4 °C |
63 | 48 |
| Catla catla | ||||
| Rohu | Acid extraction | 0.5 M acetic acid in a sample-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 for 24 h at 4 °C 10 for 24 h at 4 °C |
46 | 48 |
| Labeo rohita | ||||
| Small spotted catshark | Acid extraction | 0.5 M acetic acid at 25 °C for 24 h | 61.24 | 49 |
| Scyliorhinus canicula | ||||
| Thornback ray | Pepsin-aided acid extraction | 0.5 M acetic acid in a sample solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, followed by the addition of 5 g of pepsin per g, T = 4 °C for 18 h 10, followed by the addition of 5 g of pepsin per g, T = 4 °C for 18 h |
30.16 | 50 |
| Raja clavata | ||||
| Red drum fish | Pepsin-aided acid extraction | 15 L of 1 M acetic acid, 0.5% pepsin, t = 8 h | 4.32 ± 0.30 | 29 |
| Sciaenops ocellatus | ||||
| Mackerel | Isoelectric solubilization precipitation | 0.2 M NaOH in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 sample-solvent ratio 10 sample-solvent ratio |
48.08 ± 0.04 | 51 |
| Starfish | A combination of ultrasound and high-shear mechanical homogenization | 0.1 M NaOH in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 sample solvent ratio, 4000 rpm for 2.5 min, 70% amplitude for 10 min 10 sample solvent ratio, 4000 rpm for 2.5 min, 70% amplitude for 10 min |
3.37 ± 0.10 | 52 |
| Golden carp | Ultrasound-assisted acid extraction | Acid pretreatment in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 sample solvent ratio, 20 Hz frequency, 80% amplitude for 30 min 10 sample solvent ratio, 20 Hz frequency, 80% amplitude for 30 min |
81.53 | 53 |
| Probarbus jullieni | ||||
| Clown featherback | Ultrasound-assisted extraction | 750 W, f = 20 kHz, 20, 40, 60, and 80% amplitude with different time intervals (10, 20, and 30 min) | 27.18 to 57.35 | 54 |
| Chitala ornata | ||||
| Jellyfish | Acid extraction | 0.5 M acetic acid (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 w/v) 1000 w/v) |
65.20 ± 1.12 | 55 |
| Acromitus hardenbergi | ||||
According to Pal and Suresh (2016), acid extraction can be conducted under high temperatures using 6 M hydrochloric acid for 18–48 hours to get a resultant collagen from the fish.59 Another study states that the fish is minced and homogenized with cold water for 1 minute; then, the pH is adjusted to 2.5 using 2 M HCl. Subsequently, the mixture is kept at 4 °C, and it is centrifuged to obtain the soluble proteins. The yield of collagen is maximum (90%) when the Baltic cod is treated with lactic acid or acetic acid when compared with HCl and citric acid, which are around 18 and 60%, respectively.27 The protein extraction of seaweed (Ascophyllum nodosum) was achieved by dissolving the seaweed in distilled water and incubating it at 4 °C for 16 h. Further, the mixture is centrifuged to obtain pellets, which are treated with HCl of different molarity in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 solid-to-solvent ratio. Later, the mixture was stirred and centrifuged to obtain the proteins.
15 solid-to-solvent ratio. Later, the mixture was stirred and centrifuged to obtain the proteins.
The interaction behaviour of collagen molecules was studied, where the aggregated state of collagen molecules and the acid concentration are interrelated. When the concentration of acid is increased, there is a better recovery of proteins. The rheological behaviours also depend on each other. The viscosity of collagen tends to decrease with increasing acid concentration, resulting in the collagen's increased ability to flow.60 Protein extracted using this method can be potentially used in commercial applications.
Several studies have shown that the recovery of protein and solubilization is high at pH values above.11 This clearly shows that the recovery of protein and the solubilization of protein mainly depend on the pH value. If the pH value is higher, the consistency of the muscle is lower, so the phospholipid membranes are separated, leaving the protein during the first centrifugation, which increases the solubility of the protein.63
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 solid–liquid ratio, yielding 19.94%. The defatted sample leads to high-quality fish protein. Since proteins like insoluble collagen have hydrophobic and disulfide bonds, they rarely dissolve in an aqueous medium. Using high-reactive solvents, like hydrogen peroxide, ethylene dichloride, and sodium hypochlorite, the amino acid profile may alter, resulting in the formation of toxic peptides and choline derivatives.65
1 solid–liquid ratio, yielding 19.94%. The defatted sample leads to high-quality fish protein. Since proteins like insoluble collagen have hydrophobic and disulfide bonds, they rarely dissolve in an aqueous medium. Using high-reactive solvents, like hydrogen peroxide, ethylene dichloride, and sodium hypochlorite, the amino acid profile may alter, resulting in the formation of toxic peptides and choline derivatives.65
A concept called deep eutectic solvent (DES) extraction is applied in the extraction of proteins, where two solvents are used together, a hydrogen bond donor and a hydrogen bond acceptor, respectively.60 According to Bai (2017), the extraction yield was maximum when the extraction was performed using choline chloride and oxalic acid compared to six different methods.66 In further studies, it was found that the extraction yield of protein is higher when the amount of oxalic acid is increased and the choline chloride is kept constant. It can be used as a sustainable extraction of collagen.60
3.2. Advanced techniques for protein extraction
The sustainable alternate protein from blue foods can be extracted using advanced and eco-friendly techniques, like enzyme-assisted, sonication, pulsed electric field, and high pressure processing (Table 1). The influence of advanced techniques on protein extraction efficiency is discussed in the following subsections.For instance, it was demonstrated that enzyme-assisted extraction methods successfully extract bioactive compounds from seaweeds and microalgae used in the nutraceutical industry.68 Similarly, the extraction and identification of proteins from the red seaweed Palmaria palmata was accomplished, which resulted in a better yield and improved functional properties.69 Enzyme-assisted methods have been used to extract materials from fish and fish by-products. Wang (2020) developed antioxidant-rich protein hydrolysates from cobia liver through enzymatic hydrolysis.70 The successful extraction of proteins and fish oil from Baltic herring was demonstrated by employing proteolytic enzymes.71 Proteins were extracted from fish backbones using a combination of steam explosion methods and enzymatic hydrolysis. The extracted amount of fish backbone proteins increased through the combined use of steam explosion and enzymatic hydrolysis.71,72
Tran (2023) demonstrated that Basa fish skin collagen extraction through enzymatic hydrolysis maintained the bioactivity and structural integrity of collagen peptides. Appropriate enzyme choice alongside processing duration and conditions determines both product quantity and protein quality.73 The enzyme-assisted extraction method presents multiple benefits over traditional solvent-based techniques by speeding up processing times and reducing harmful chemical use while enhancing environmental sustainability. Enzyme-assisted extraction has become a leading method for maximizing blue food resources and satisfying the rising demand for functional and bioactive protein ingredients.
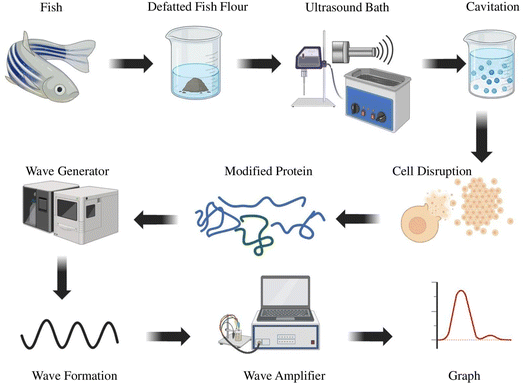
Ultrasonic wave vibration creates cavitation. The dissipation of energy spreads throughout the medium by developing and disintegrating vapor bubbles. Cavitation additionally increases the kinetic energy of the particles within the treated substance (Fig. 4). As a result of the ultrasonic energy's shock effect during the extraction process, high temperature offers enough energy for a reaction.74 Numerous intra- and inter-molecular covalent cross-links involving lysine and hydroxylysine residues, ester bonds, and other interactions with saccharides must be broken during collagen extraction. Mass transportation in wet processes is improved by ultrasonication.
Among all the proteins, collagen is the most abundant structural protein, which accounts for about 25–30% and is found in the animal body. Collagen consists of a large molecule that has a triple helical structure with each polypeptide chain mostly containing repeating units of glycine, proline, and hydroxyproline.52 Conventional methods are prolonged processes, and collagen residue tissue will be more; in addition, a high amount of insoluble collagens will be left behind.52 The ultrasonication technique is non-destructive and non-invasive; the good quality of protein can be obtained without interrupting the molecular structure of collagen.75 To extract collagen from various marine sources, such as clown featherback, golden carp, sea bass, soft-shelled turtle, and grass carp calipash, the ultrasonication method is used.
Moreover, ultrasonication reduces the size of collagen particles despite increasing their pH and salt-induced solubility.76 Even though ultrasonication is a favourable and propitious technology for enhancing the quality and amount of resulting collagen, the amplitudes and extraction times need to be carefully regulated. The use of ultrasonication in collagen extraction can shorten extraction times and improve the quality and quantity of collagen recovered. Low-intensity ultrasound alters the state of bacteria and promotes their development by causing repairable damage and stable cavitation.77 Furthermore, water molecules may be broken down more easily by ultrasound, which can potentially generate free radicals and damage bacterial DNA and enzymes.78
Even though ultrasonication has many advantages, prolonged use of the technology results in a cavitation effect, which increases the temperature, shear force, and pressure of the medium and breaks hydrogen bonds and van der Waals interactions in polypeptide chains, and this denaturizes proteins and enzymes.51
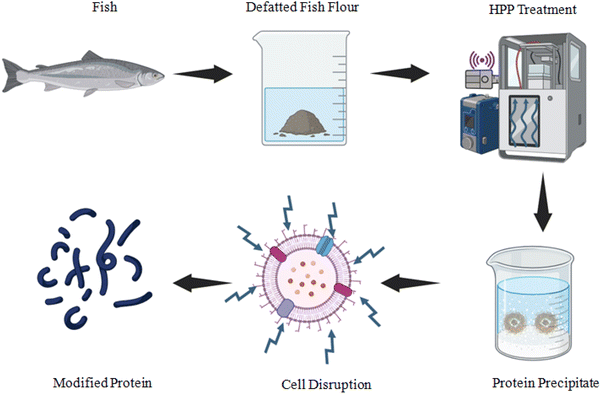
HPP can break down the cell walls of both Gram-positive and Gram-negative bacteria to facilitate a greater amount of protein yield through extraction. The interaction of side-to-side chains through covalent and non-covalent bonding promotes aggregation and gelation when HPP is introduced to myosin. Gels are subjected to pressure treatments that encourage the formation of non-covalent linkages.13 The development of a uniform and smooth gel structure contributed to the growth of the myosin matrix, which was significantly enhanced by the use of 300 MPa. Additionally, modifications to the structure of myofibrillar proteins may have an impact on the quality of meat products and the textural characteristics and properties of gels.84 HPP enhances gelling properties in marine foods.
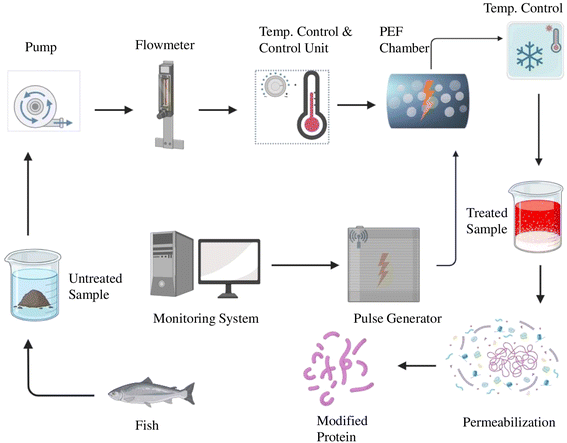
Buchmann (2019) determined the interrelationship between the strength of the electric field and the concentration of protein extracted from Chlorella vulgaris subjected to an electric field strength of 20 kV cm−1.87 As an instance from the study, the protein concentration was 0.55 ± 0.01 g L−1 under an electric field strength of 10 kV cm−1, and this was increased to 0.80 ± 0.04 g L−1 in 20 kV cm−1, indicating an increase in the average protein extraction with increasing electric field strength. Some studies have shown that PEF is not feasible for the extraction of selective proteins alone because it does not completely disintegrate the cell wall.43 Polikovsky (2016) demonstrated the extraction of selective proteins using PEF, wherein the yield of specific proteins was higher.21 By allowing the extraction of intact cytosolic proteins, there was a notable increase in the protein extraction of C. vulgaris and Nannochloropsis salina.88
3.2.6.1. PEF and enzymatic extraction. In the combination of PEF and enzymatic extraction, PEF could reduce the foaming properties and enhance the emulsifying properties and the solubility of the protein from the abalone viscera, resulting in a promising yield. A study showed that the yield of combined treatment is higher than that of pure enzymatic extraction.90
3.2.6.2. PEF and mechanical press. In this extraction process, PEF is coupled with a mechanical press, where 200 pulses with a field strength of 1 kV cm−1 were applied to Ulva ohndi. The duration and repetition of pulses were 50 µs and 3 Hz, respectively. This was done concurrently with mechanical pressing at 107
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 682 N m−2. Around a 14.94% yield of protein was obtained through this extraction.23
682 N m−2. Around a 14.94% yield of protein was obtained through this extraction.23
3.2.6.3. PEF and hydraulic pressure. According to Polikovsky (2016), the biomass of Ulva sp. was submerged in deionized water and 75 pulses with a field strength of 2.964 kV cm−1 were applied to the solution with a pulse repetition rate of 0.5 Hz and 5.70 µs of pulse duration. The extraction was combined with hydraulic pressing for 5 min under a pressure of 450 N cm−2. The total protein concentration of the extract after treatment is 59.13 ± 3.82 µg mL−1, which is higher than that of the untreated sample.21 The advanced non-thermal approach leads to optimistic nutritional and functional characteristics (Table 2) that can be utilized for enhanced protein extraction from blue foods. Hybrid techniques pave a new direction for researchers to obtain synergistic effects in blue foods.
| S. no. | Treatments | Sources | Functional property | Reference |
|---|---|---|---|---|
| a WHC-water holding capacity, WAC-water absorption capacity, ES-emulsion stability, EA-emulsifying activity, RP-rheological property, FC-foaming capacity, FS-foaming stability, OAC-oil absorption capacity, FBC-fat binding capacity. | ||||
| 1 | Ultrasound-assisted extraction | Spirulina platensis | WAC, OAC, EA, ES, FC. FS | 91 |
| 2 | Osmotic shock + ultrasound | Chlorella vulgaris | WAC, OAC, EA, ES, FC, FS | 92 |
| 3 | Isoelectric precipitation | Loligo vulgaris | FC, FS, ES, FBC, WHC | 93 |
| 4 | HPP | Callinectes sapidus | WBC | 94 |
| 5 | Ultrasound-assisted extraction | Dosidicus gigas | FC, FS | 95 |
| 6 | PEF | Haliotis discus hannai Ino | FC, FS, EA, ES | 90 |
| 7 | HPP | Blue crab | Gelation property | 13 |
| 8 | Ultrasound-assisted extraction | Dasyatis zugei | Viscosity, RP, WHC, ES | 75 |
| 9 | Ultrasound-assisted extraction | Tuna | WHC, ES, EA, viscosity | 76 |
4. Factors affecting proteins from blue foods and their mitigations
4.1. Impurities of protein purity
Even though algae are rich in protein, they also tend to gather toxic heavy metals, like mercury, lead, arsenic, and cadmium. The presence of lead and mercury is under legal limits for safe human consumption, whereas the presence of arsenic and cadmium is above legal limits. There is a chance of protein contamination with heavy metals when extracted from algae. There is an acceptable level of heavy metal in the case of microalgae.864.2. Food allergenicity as a barrier to protein consumption
Protein consumption faces significant obstacles because food allergies trigger immune responses that can be either IgE-mediated or non-IgE-mediated. The release of histamine through IgE-mediated allergies produces symptoms spanning the skin and the respiratory, digestive, and circulatory systems.96,97 Food allergies affect 2.5% of people worldwide, while fish is one of the main allergenic sources. The WHO/IUIS has recognized 12 fish proteins as allergens, including parvalbumin, along with tropomyosin and aldolase A. People who develop fish allergies typically have symptoms that last throughout their lives although the severity changes over time. The fish species cod, salmon, and mackerel contain parvalbumin, which serves as a primary allergen. Protein structures undergo changes due to processing methods, like drying, heating, and smoking, which can influence their potential to cause allergic reactions.98The effects of processing methods on allergenicity depend on the specific protein matrix structure, alongside detection techniques and antibody specificity. Experimental findings from ELISA and western blotting indicate that thermal processing alters IgE protein binding patterns. IgE binding to heated whiff parvalbumin vanishes rapidly during digestion but remains in some cases after extended heating periods.99,100 Allergenicity varies based on demographic factors, such as age, gender, region, and ethnicity, showing increased prevalence in Asian populations as well as in females and adults.
4.3. Economic perspectives on commercial usage
The commercialization of microalgae to extract proteins was found to be a challenge.101 The cost of microalgae production varies depending on the upstream process and downstream processes. Some studies have shown that microalgae can be cultivated in wastewater, but this results in contamination of the produce, and it cannot be used in foods.102 To obtain better growth of microalgae, photobioreactors can be used, but the operating costs for this reactor are high. The development of some robust processes is needed for the commercialization of microalgae.1034.4. Environmental sustainability
To enhance transparency in the environmental sustainability discussion, the carbon footprint considerations presented in this review follow a cradle-to-gate system boundary. This includes raw material acquisition from blue food sources, pre-treatment processes, non-thermal extraction stages, and subsequent drying operations while excluding downstream processes such as packaging, distribution, retail handling, consumer use, and end-of-life disposal. This boundary definition aligns with standardized environmental assessment approaches used in food system evaluations.104 The functional unit was defined as 1 kg of isolated protein produced, ensuring comparability with food-system-level sustainability metrics reported for blue foods.2,8Carbon emission estimations were primarily based on reported energy consumption associated with non-thermal technologies, including ultrasound, pulsed electric field, and high-pressure processing. Emissions were conceptually calculated by multiplying energy demand (kWh) by the corresponding carbon intensity factors of electricity, which is consistent with the carbon footprint frameworks used in food basket and resource assessment studies.104 Assumptions include average industrial electricity consumption, partial water reuse during extraction, and solvent recovery efficiencies where applicable. Capital equipment manufacturing and transportation emissions were excluded due to limited reporting across extraction studies. By explicitly defining system boundaries, functional units, and calculation approaches, the environmental discussion is aligned with broader sustainability assessments of blue foods and global food systems, thereby improving clarity, transparency, and methodological consistency.2,8
5. Applications of proteins from blue foods
The aquatic ecosystem is still an unexploited reservoir of many nutritional and functional components. The marine protein (collagen) plays a vital role in pharmaceutical, food, cosmetic and other domains. The food-based application of protein from blue foods is highlighted in this review.5.1. Taylor-made foods/functional foods from blue foods
Algal proteins are incorporated into foods due to their nutritional and sensorial properties and to overcome the challenges associated with the low dispersibility of proteins. The algal proteins are used in the formulation of yogurt with A. plantensis and pasta with C. vulgaris and A. maxima.105,106 The microalgal proteins exhibit foaming, emulsifying, and stabilizing properties, which show their advantages over plant proteins.103Some studies have shown that bread can be enriched with fish proteins through different formulations. The acceptability rate was also high, and this showed that there was not much difference in the sensorial properties of bread.107
5.2. Nutraceuticals from aquatic organisms
Natural bioactive compounds with therapeutic benefits, called nutraceuticals, have become more popular as synthetic drug substitutes because disease rates continue to rise. Shrimp processing generates 40–50% biomass waste, which is considered valuable among aquatic sources. Shrimp waste contains high levels of astaxanthin (C40H52O4), which acts as a powerful antioxidant while giving shrimp meat its red color and making up 65–98% of its total carotenoid content.108 Each year, India generates over 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tonnes of shrimp waste, which represents a significant potential for resource recovery.109
000 tonnes of shrimp waste, which represents a significant potential for resource recovery.109
Feed microalgae-derived β-carotene or zeaxanthin undergoes oxidative conversion to create astaxanthin, which holds substantial commercial importance in both food and pharmaceutical sectors.110 The process of extracting astaxanthin from crustacean waste achieves sustainable waste management goals while meeting increasing market needs for natural dietary supplements. Product consistency and global market needs present major challenges to scaling shrimp-waste-based nutraceutical production.
5.3. Valorization of biowaste from marine sources
Generally, macroalgae are used as a feasible feedstock for biomass. The challenging one is the fractionation of biomass; some sustainable processes and methods are needed to upscale it. Without fractionation, the algae become waste. To reduce this, PEF coupled with mechanical pressing is applied to the macroalgae (U. ohnoi) for the extraction of a wholesome amount of proteins.23The optimization of low-value foods and underutilized foods is necessary to avoid wastage. Underutilized fish can be used to extract proteins. Through this, the underutilized fish are converted into valuable ones. Further, fish proteins can be incorporated into the foods.111
5.4. Sustainable animal feed and aquafeed
Blue food-derived protein hydrolysates, which are typically by-products, present nutritious and sustainable feed options for animals and aquaculture that yield better results. Protein hydrolysates contain abundant bioavailable amino acids and bioactive peptides that support animal growth performance and immunity while improving nutrient absorption. The process of enzyme-assisted hydrolysis produces fish protein hydrolysates from sardine by-products and marine wastes, which deliver high-quality feed supplements with minimal environmental impact.67 These protein compounds exhibit functional advantages through their antioxidant and antimicrobial properties, which enhance animal well-being and lessen antibiotic dependence in feed.65Aquaculture systems can use fish protein hydrolysates to replace traditional fishmeal either partially or completely, which helps reduce the exploitation of wild fish stocks. For example, microalgal proteins extracted through non-thermal methods, such as pulsed electric fields or enzymatic hydrolysis, show great potential for use in aquafeeds.103 Alternative protein sources have exhibited excellent digestibility and palatability while offering a balanced profile of amino acids.
The residual biomass from blue food production, including seaweed residues and microalgal cell walls, can be transformed into feed components or bio-stimulants for use in integrated aquaculture systems.86 Methods for transforming algal residues with high structural carbohydrate and protein content into viable dietary components for fish and shrimp following proper processing were examined. Nutrient sources from these residues also support gut health and maintain microbial balance among aquatic organisms.
5.5. Potential medical applications from marine sources
Proteins, peptides, and amino acids obtained from marine sources possess enormous potential for advanced biomedical research, providing insight into the functioning of both healthy and diseased human bodies. Moreover, they provide the potential to create highly targeted and effective medications to treat a range of illnesses. For example, consider collagen taken from some marine mammals. Tissue-like structures in 3D bioprinting are often built on collagen, a structural protein essential for many body tissues. These collagen scaffolds are very useful for tissue engineering applications, such as blood vessels, skin, and bone regeneration.112Furthermore, collagen is an invaluable medical excipient due to its exceptional qualities, which include low antigenicity and biocompatibility. It has several uses, including drug transport capsules, bone tissue regeneration fillers, and deionized membranes in surgery. When introduced into the body, these membranes function well with host cells and tissues, reducing negative immune reactions.113 Collagen is biodegradable and naturally absorbed by the body.114
5.6. Innovative food products
Marine proteins have become popular in the food industry due to their high nutritional value. Collagen from marine animals is used to make gelatin, a versatile ingredient.115 Gelatin is a major component of many delicious desserts, such as fudges, puddings, marshmallows, and salads. Algal proteins, rich in amino acids and readily digestible, have been incorporated into innovative food products.116 These proteins are incorporated into vegan meat substitutes, protein-rich snacks, and dairy alternatives. Proteins derived from edible microalgae, like Isochrysis galbana and Chlorella vulgaris, show potential as food supplements and additives. Phycobiliproteins from Porphyridium cruentum (red algae) and Synechococcus spp. (blue-green algae) are utilized as natural food colorants. These specialized protein groups are integrated into various food products, including dairy, chewing gums, jellies, and ice sherbaths, to serve as colouring agents.1145.7. Residue valorization and circular bioeconomy
Protein extraction from blue biomass generates significant residual fractions that are rich in lipids, minerals, collagen, chitin, and other bioactive compounds. These residues should not be considered waste because they possess high biological and commercial value. The effective utilization of these fractions plays a vital role in strengthening the circular bioeconomy model and improving the sustainability of the blue food supply chain.5 The lipid fraction remaining after protein extraction may contain long-chain omega-3 fatty acids, such as EPA and DHA, which are widely used in nutraceutical and functional food formulations. Tuna processing side streams are successfully utilized for the recovery of proteins, lipids, and minerals using integrated biorefinery approaches, thereby reducing environmental burden and enhancing economic return.117 Similarly, invasive marine species, such as blue crab (Callinectes sapidus), are valorised into high-value nutritional ingredients and bioactive compounds, converting ecological challenges into sustainable opportunities.118,119Collagen and gelatin extracted from fish skin, bones, and connective tissues have wide applications in the food, pharmaceutical, and biomedical industries due to their functional and structural properties. Mineral-rich residues, particularly from bivalve shells and crustacean exoskeletons, are important sources of calcium carbonate and chitin. Valorization of shell waste has gained attention within circular blue bioeconomy strategies for applications in agriculture, environmental remediation, and material science.120 Moreover, the remaining biomass after sequential extraction can be utilized as an animal feed ingredient or converted into bioenergy through anaerobic digestion, thereby reducing landfill accumulation and greenhouse gas emissions. The integration of protein extraction into a multi-product biorefinery framework enables the cascading utilization of marine biomass and supports sustainable, circular food systems.121
6. Practical and industrial challenges in the application of isolated blue food proteins
Despite the promising functional and nutritional attributes of isolated proteins derived from blue foods, several practical and industrial challenges remain that may limit their large-scale implementation. One major limitation involves variability in raw material composition, which can significantly influence protein yield, purity, and techno-functional performance.103,122 Seasonal variation, species diversity, and environmental conditions affect protein structure and extractability, thereby complicating process standardization. Furthermore, purification and isolation technologies often require multiple processing steps, including membrane filtration, precipitation, and chromatographic separation, which increase operational costs and energy demand.123 Although non-thermal techniques improve extraction efficiency, scale-up feasibility and equipment investment remain critical constraints.In addition, structural modifications occurring during extraction and drying may alter solubility, emulsifying capacity, and gelation behaviour, potentially limiting functional performance in complex food matrices.124,125 Sensory challenges, including off-flavors, color instability, and marine-associated aromas, can also affect consumer acceptance, particularly in mainstream food applications.126 Regulatory considerations, allergenicity assessments, and labelling requirements further complicate commercialization pathways for novel protein ingredients. Therefore, although isolated blue food proteins demonstrate considerable potential for sustainable food system transformation, addressing techno-economic feasibility, sensory optimization, and regulatory compliance is essential for successful real-world adoption.
7. Conclusion, limitations, and future perspectives
This review explored innovative techniques for protein extraction from blue foods based on the concept of non-thermal techniques, which are a safe, green approach, with less treatment time, and can enhance the extraction efficiency of protein of supreme quality. Proteins extracted from marine resources can be effectively utilized for commercial applications, particularly in nutraceuticals and functional bioactive products. There is a projected substantial market for food products, supplements, and natural health products containing marine bioactives due to several possible health benefits. Despite providing a comprehensive overview of non-thermal techniques for sustainable protein extraction from blue foods, this review is limited by the availability of large-scale industrial data and standardized environmental assessment metrics across studies. Variations in reported extraction efficiencies, functional properties, and processing conditions restrict direct quantitative comparisons. Additionally, techno-economic feasibility and long-term storage stability remain underexplored in the current literature, highlighting the need for pilot-scale validation and integrated life cycle assessments in future research. This review directs future research and development on modified proteins, or bioactive compounds in foods for prominence, as these components have significant nutritional and functional benefits in addressing various health concerns. In the future, the commercialization process will gain advantages by applying biorefinery approaches that aim to create cost-effective and environmentally friendly extraction methods for generating bioactive compounds with well-measured beneficial effects. Innovative non-thermal technologies have arisen as green approaches, promising, safe, and efficient methods for retrieving high-quality nutritional components, specifically protein. These modified proteins have optimistic structural and functional properties that can be commercially utilized as functional ingredients.Author contributions
Nikashini Thirugnanam – conceptualization, design of methodology, data acquisition, data curation, writing original draft, review, and editing. Monisha Chandran – conceptualization, design of methods, writing original draft, review, and editing, Pathare Ashutosh Dattatrya – conceptualization, design of methods, data acquisition, data curation, writing original draft. Loganathan Manickam – conceptualization, design of methods, review and editing, supervision, project administration.Conflicts of interest
The authors declare that there is no conflict of interest.Abbreviations
| 3D | Three Dimensional |
| ACS | Acid Soluble Collagen |
| BSE | Bovine Spongiform Encephalopathy |
| DES | Deep Eutectic Solvent |
| DNA | Deoxyribonucleic Acid |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| FMD | Foot and Mouth Disease |
| FPH | Fish Protein Hydrolysates |
| HPP | High-Pressure Processing |
| HVAC | High Value Compounds |
| IUIS | International Union of Immunological Societies |
| PEF | Pulsed Electric Field |
| SCF | Super Critical Fluid |
| TSE | Transmissible Spongiform Encephalopathy |
| WHO | World Health Organisation |
Data availability
Data sharing does not apply to this article as no new data were created or analyzed in this study. This study is based entirely on published literature that is appropriately cited and available in the public domain.References
- R. L. Naylor, A. Kishore, U. R. Sumaila, I. Issifu, B. P. Hunter and B. Belton, et al., Blue food demand across geographic and temporal scales, Nat. Commun., 2021, 12(1), 5413 CrossRef CAS PubMed.
- M. S. Bank, C. M. Duarte and C. Sonne, Intergovernmental Panel on Blue Foods in Support of Sustainable Development and Nutritional Security, Environ. Sci. Technol., 2022, 56, 5302–5305 CrossRef CAS PubMed.
- J. R. Watson, C. M. Spillman, L. R. Little, A. J. Hobday and P. S. Levin, Enhancing the resilience of blue foods to climate shocks using insurance, ICES J. Mar. Sci., 2023, 2457–2469 CrossRef.
- J. Atalah and P. Sanchez-Jerez, On the wrong track: Sustainable and low-emission blue food diets to mitigate climate change, Front. Sustain. Food Syst., 2022, 6, 994840 CrossRef.
- S. M. Colombo, K. Roy, J. Mraz, A. H. L. Wan, S. J. Davies and S. M. Tibbetts, et al., Towards achieving circularity and sustainability in feeds for farmed blue foods, Rev. Aquac., 2023, 15(3), 1115–1141 CrossRef.
- C. V. Schmidt and O. G. Mouritsen, Cephalopods as Challenging and Promising Blue Foods: Structure, Taste, and Culinary Highlights and Applications, Foods, 2022, 11, 2559 CrossRef CAS PubMed.
- C. Monisha and M. Loganathan, Impact of drying methods on the physicochemical properties and nutritional composition of defatted black soldier fly (Hermetia illucens) pre-pupae flour, J. Food Process. Preserv., 2022, 46(1), e16184 CAS.
- B. I. Crona, E. Wassénius, M. Jonell, J. Z. Koehn, R. Short and M. Tigchelaar, et al., Four ways blue foods can help achieve food system ambitions across nations, Nature, 2023, 616(7955), 104–112 CrossRef CAS PubMed.
- R. Sharma, D. F. Reguaera, C. Fuentevilla, V. Agostini and M. Barange, Aquatic food systems for blue transformation: a vision for FAO, in Transformation of Agri-Food Systems, Springer, 2024, pp. 193–204 Search PubMed.
- J. A. J. Verreth, K. Roy and G. M. Turchini, Circular bio-economy in aquaculture, Rev. Aquac., 2023, 15(3), 944–946 CrossRef.
- Y. Zou, P. Xu, P. Li, P. Cai, M. Zhang and Z. Sun, et al., Effect of ultrasound pre-treatment on the characterization and properties of collagen extracted from soft-shelled turtle (Pelodiscus sinensis), LWT, 2017, 72–81 CrossRef CAS.
- S. Momen, F. Alavi and M. Aider, Alkali-mediated treatments for extraction and functional modification of proteins: Critical and application review, Trends Food Sci. Technol., 2021, 110, 778–797 CrossRef CAS.
- G. Velazquez, M. G. Méndez-Montealvo, J. Welti-Chanes, J. A. Ramírez and M. A. Martínez-Maldonado, Effect of high pressure processing and heat treatment on the gelation properties of blue crab meat proteins, LWT, 2021, 146, 111389 CrossRef CAS.
- S. Fan, Y. Yin, Q. Liu, X. Yang, D. Pan and Z. Wu, et al., Blue food proteins: Novel extraction technologies, properties, bioactivities and applications in foods, Curr. Res. Food Sci., 2024, 9, 100878 CrossRef CAS PubMed.
- C. da S. Carvalho, G. X. Ojoli, M. G. Paco, N. A. Bonetti, S. C. de Pinho and J. T. do P. Silva, et al., The Role of Nonconventional Technologies in the Extraction Enhancement and Technofunctionality of Alternative Proteins from Sustainable Sources, Foods, 2025, 14(21), 3612 CrossRef CAS PubMed.
- Z. X. Tang, R. F. Ying and L. E. Shi, Physicochemical and functional characteristics of proteins treated by a pH-shift process: a review, Int. J. Food Sci. Technol., 2021, 56, 515–529 CrossRef.
- Z. Yu, Y. Gao, X. Jia, S. Cui, L. Ma and D. Zheng, et al., Recent advance in high-intensity ultrasound modification of blue food protein: Mechanisms, functional properties and structural alterations, Trends Food Sci. Technol., 2024, 143, 104271 CrossRef CAS.
- M. Chandran, S. Vignesh, L. Manickam Professor, S. Anandakumar and L. Manickam, Utilization of black soldier fly (Hermetia illucens) prepupae flour in development of bio-packaging film, Pharma Innov., 2021, 335(11), 335–340 Search PubMed.
- I. Jain, R. Kaur, A. Kumar, M. Paul and N. Singh, Emerging protein sources and novel extraction techniques: a systematic review on sustainable approaches, Int. J. Food Sci. Technol., 2024, 59(10), 6797–6820 CrossRef CAS.
- P. A. Harnedy and R. J. FitzGerald, Extraction of protein from the macroalga Palmaria palmata, LWT, 2013, 51(1), 375–382 CrossRef CAS.
- M. Polikovsky, F. Fernand, M. Sack, W. Frey, G. Müller and A. Golberg, Towards marine biorefineries: Selective proteins extractions from marine macroalgae Ulva with pulsed electric fields, Innov. Food Sci. Emerg. Technol., 2016, 37, 194–200 CrossRef CAS.
- J. Fleurence, Seaweed proteins, in Proteins in Food Processing, Elsevier, 2004, pp. 197–213 Search PubMed.
- M. S. Prabhu, K. Levkov, Y. D. Livney, A. Israel and A. Golberg, High-Voltage Pulsed Electric Field Preprocessing Enhances Extraction of Starch, Proteins, and Ash from Marine Macroalgae Ulva ohnoi, ACS Sustain. Chem. Eng., 2019, 7(20), 17453–17463 CrossRef CAS.
- Y. Wang, S. Tibbetts and P. McGinn, Plankton untuk makanan manusia.pdf, Foods, 2021, 10(3002), 1–18 Search PubMed.
- M. P. Caporgno and A. Mathys, Trends in Microalgae Incorporation Into Innovative Food Products With Potential Health Benefits, Front. Nutr., 2018, 5, 1–10 Search PubMed.
- F. J. Barba, N. Grimi and E. Vorobiev, New Approaches for the Use of Non-conventional Cell Disruption Technologies to Extract Potential Food Additives and Nutraceuticals from Microalgae, Food Eng. Rev., 2015, 7, 45–62 CrossRef CAS.
- M. Ahmed, A. K. Verma and R. Patel, Collagen extraction and recent biological activities of collagen peptides derived from sea-food waste: A review, Sustain. Chem. Pharm., 2020, 18, 100315 CrossRef.
- S. Benjakul, Y. Thiansilakul, W. Visessanguan, S. Roytrakul, H. Kishimura and T. Prodpran, et al., Extraction and characterisation of pepsin-solubilised collagens from the skin of bigeye snapper (Priacanthus tayenus and Priacanthus macracanthus), J. Sci. Food Agric., 2010, 90(1), 132–138 CrossRef CAS PubMed.
- S. Chen, H. Chen, Q. Xie, B. Hong, J. Chen and F. Hua, et al., Rapid isolation of high purity pepsin-soluble type I collagen from scales of red drum fish (Sciaenops ocellatus), Food Hydrocoll., 2016, 52, 468–477 CrossRef CAS.
- D. N. Kundam, I. O. Acham and A. T. Girgih, Bioactive Compounds in Fish and Their Health Benefits, Asian Food Sci. J., 2018, 4(4), 1–14 CrossRef.
- S. Heffernan, L. Giblin and N. O’Brien, Assessment of the biological activity of fish muscle protein hydrolysates using in vitro model systems, Food Chem., 2021, 359, 129852 CrossRef CAS PubMed.
- S. Balami, A. Sharma and R. Karn, Significance Of Nutritional Value Of Fish For Human Health, Malays. J. Halal Res., 2019, 2(2), 32–34 CrossRef.
- M. Aucoin, K. Cooley, C. Knee, H. Fritz, L. G. Balneaves and R. Breau, et al., Fish-Derived Omega-3 Fatty Acids and Prostate Cancer: A Systematic Review, Integr. Cancer Ther., 2017, 16, 32–62 CrossRef CAS PubMed.
- R. Ma, M. Mhr, S. Mk, C. Sh and S. Mm, Snakehead Fish (hanna striata) and Its Biochemical Properties for Therapeutics and Health Benefits, J. Biotechnol. Biomed. Eng., 2020, 1, 1 Search PubMed.
- C. G. Awuchi, C. N. Chukwu, A. O. Iyiola, S. Noreen, S. Morya and A. O. Adeleye, et al., Bioactive Compounds and Therapeutics from Fish: Revisiting Their Suitability in Functional Foods to Enhance Human Wellbeing, BioMed Res. Int., 2022, 1, 3661866 CrossRef PubMed.
- E. Barbarino and S. O. Lourenço, An evaluation of methods for extraction and quantification of protein from marine macro- and microalgae, J. Appl. Phycol., 2005, 17(5), 447–460 CrossRef CAS.
- C. Safi, A. V. Ursu, C. Laroche, B. Zebib, O. Merah and P. Y. Pontalier, et al., Aqueous extraction of proteins from microalgae: Effect of different cell disruption methods, Algal Res., 2014, 3(1), 61–65 CrossRef.
- K. Hardouin, G. Bedoux, A. S. Burlot, C. Donnay-Moreno, J. P. Bergé and P. Nyvall-Collén, et al., Enzyme-assisted extraction (EAE) for the production of antiviral and antioxidant extracts from the green seaweed Ulva armoricana (Ulvales, Ulvophyceae), Algal Res., 2016, 16, 233–239 CrossRef.
- V. Vásquez, R. Martínez and C. Bernal, Enzyme-assisted extraction of proteins from the seaweeds Macrocystis pyrifera and Chondracanthus chamissoi: characterization of the extracts and their bioactive potential, J. Appl. Phycol., 2019, 31(3), 1999–2010 CrossRef.
- M. Kazir, Y. Abuhassira, A. Robin, O. Nahor, J. Luo and A. Israel, et al., Extraction of proteins from two marine macroalgae, Ulva sp. and Gracilaria sp., for food application, and evaluating digestibility, amino acid composition and antioxidant properties of the protein concentrates, Food Hydrocoll., 2019, 87, 194–203 CrossRef CAS.
- V. Ummat, M. Garcia-Vaquero, M. M. Poojary, M. N. Lund, C. O'Donnell and Z. Zhang, et al., Green extraction of proteins, umami and other free amino acids from brown macroalgae Ascophyllum nodosum and Fucus vesiculosus, J. Appl. Phycol., 2021, 33(6), 4083–4091 CrossRef CAS PubMed.
- J. O’ Connor, S. Meaney, G. A. Williams and M. Hayes, Extraction of protein from four different seaweeds using three different physical pre-treatment strategies, Molecules, 2020, 25(8), 2005 CrossRef PubMed.
- G. P. ’t Lam, P. R. Postma, D. A. Fernandes, R. A. H. Timmermans, M. H. Vermuë and M. J. Barbosa, et al., Pulsed Electric Field for protein release of the microalgae Chlorella vulgaris and Neochloris oleoabundans, Algal Res., 2017, 24, 181–187 CrossRef.
- E. Steinbruch, J. Wise, K. Levkov, A. Chemodanov, Á. Israel and Y. D. Livney, et al., Enzymatic cell wall degradation combined with pulsed electric fields increases yields of water-soluble-protein extraction from the green marine macroalga Ulva sp, Innov. Food Sci. Emerg. Technol., 2023, 84, 103231 CrossRef CAS.
- J. Fleurencel, L. Massiani, O. Guyader and S. Mabeau, Use of enzymatic cell wall degradation for improvement of protein extraction from Chondrus crispus, Gracilaria verrucosa and Pabmaria palmata, J. Appl. Phycol., 1995, 7(4), 393–397 CrossRef.
- O. Kaewdang, S. Benjakul, T. Kaewmanee and H. Kishimura, Characteristics of collagens from the swim bladders of yellowfin tuna (Thunnus albacares), Food Chem., 2014, 155, 264–270 CrossRef CAS PubMed.
- D. Liu, G. Wei, T. Li, J. Hu, N. Lu and J. M. Regenstein, et al., Effects of alkaline pretreatments and acid extraction conditions on the acid-soluble collagen from grass carp (Ctenopharyngodon idella) skin, Food Chem., 2015, 172, 836–843 CrossRef CAS PubMed.
- G. K. Pal, T. Nidheesh and P. V. Suresh, Comparative study on characteristics and in vitro fibril formation ability of acid and pepsin soluble collagen from the skin of catla (Catla catla) and rohu (Labeo rohita), Food Res. Int., 2015, 76, 804–812 CrossRef CAS PubMed.
- M. Blanco, J. A. Vázquez, R. I. Pérez-Martín and C. G. Sotelo, Collagen extraction optimization from the skin of the small-spotted catshark (S. Canicula) by response surface methodology, Mar. Drugs, 2019, 17(1), 40 CrossRef CAS.
- I. Lassoued, M. Jridi, R. Nasri, A. Dammak, M. Hajji and M. Nasri, et al., Characteristics and functional properties of gelatin from thornback ray skin obtained by pepsin-aided process in comparison with commercial halal bovine gelatin, Food Hydrocoll., 2014, 41, 309–318 CrossRef CAS.
- C. Álvarez, P. Lélu, S. Lynch and B. K. Tiwari, Optimised protein recovery from mackerel whole fish by using sequential acid/alkaline isoelectric solubilization precipitation (ISP) extraction assisted by ultrasound, LWT, 2017, 88, 210–216 CrossRef.
- N. K. Vate, I. Undeland and M. Abdollahi, Resource efficient collagen extraction from common starfish with the aid of high shear mechanical homogenization and ultrasound, Food Chem., 2022, 393, 133426 CrossRef CAS PubMed.
- A. M. M. Ali, H. Kishimura and S. Benjakul, Extraction efficiency and characteristics of acid and pepsin soluble collagens from the skin of golden carp (Probarbus Jullieni) as affected by ultrasonication, Process Biochem., 2018, 66, 237–244 CrossRef CAS.
- T. Petcharat, S. Benjakul, S. Karnjanapratum and S. Nalinanon, Ultrasound-assisted extraction of collagen from clown featherback (Chitala ornata) skin: yield and molecular characteristics, J. Sci. Food Agric., 2021, 101(2), 648–658 CrossRef CAS PubMed.
- N. M. H. Khong, F. M. Yusoff, B. Jamilah, M. Basri, I. Maznah and K. W. Chan, et al., Improved collagen extraction from jellyfish (Acromitus hardenbergi) with increased physical-induced solubilization processes, Food Chem., 2018, 251, 41–50 CrossRef CAS PubMed.
- S. Jiang, J. Ding, J. Andrade, T. M. Rababah, A. Almajwal and M. M. Abulmeaty, et al., Modifying the physicochemical properties of pea protein by pH-shifting and ultrasound combined treatments, Ultrason. Sonochem., 2017, 38, 835–842 CrossRef CAS PubMed.
- J. Jiang, Q. Wang and Y. L. Xiong, A pH shift approach to the improvement of interfacial properties of plant seed proteins, Curr. Opin. Food Sci., 2018, 19, 50–56 CrossRef.
- T. R. L. Senadheera, D. Dave and F. Shahidi, Sea cucumber derived type i collagen: A comprehensive review, Mar. Drugs, 2020, 18(9), 471 CrossRef CAS PubMed.
- G. K. Pal and P. V. Suresh, Sustainable valorisation of seafood by-products: Recovery of collagen and development of collagen-based novel functional food ingredients, Innov. Food Sci. Emerg. Technol., 2016, 37, 201–215 CrossRef CAS.
- H. Jafari, A. Lista, M. M. Siekapen, P. Ghaffari-Bohlouli, L. Nie and H. Alimoradi, et al., Fish collagen: Extraction, characterization, and applications for biomaterials engineering, Polymers, 2020, 12, 1–37 CrossRef PubMed.
- P. Vareltzis and I. Undeland, Removal of lipids and diarrhetic shellfish poisoning toxins from blue mussels (Mytilus edulis) during acid and alkaline isolation of proteins, J. Agric. Food Chem., 2008, 56(10), 3675–3681 CrossRef CAS PubMed.
- S. Rawdkuen, S. Sai-Ut, S. Khamsorn, M. Chaijan and S. Benjakul, Biochemical and gelling properties of tilapia surimi and protein recovered using an acid-alkaline process, Food Chem., 2009, 112(1), 112–119 CrossRef CAS.
- M. Abdollahi and I. Undeland, Physicochemical and gel-forming properties of protein isolated from salmon, cod and herring by-products using the pH-shift method, LWT, 2019, 101, 678–684 CrossRef CAS.
- S. U. Kadam, B. K. Tiwari and C. P. O'Donnell, Application of novel extraction technologies for bioactives from marine algae. Vol. 61, J. Agric. Food Chem., 2013, 4667–4675 CrossRef CAS PubMed.
- A. C. Kumoro, D. H. Wardhani, T. D. Kusworo, M. Djaeni, T. C. Ping and Y. Ma'rifat Fajar Azis, Fish protein concentrate for human consumption: A review of its preparation by solvent extraction methods and potential for food applications, Ann. Agric. Sci., 2022, 67, 42–59 CrossRef.
- C. Bai, Q. Wei and X. Ren, Selective Extraction of Collagen Peptides with High Purity from Cod Skins by Deep Eutectic Solvents, ACS Sustain. Chem. Eng., 2017, 5(8), 7220–7227 CrossRef CAS.
- M. S. Benhabiles, N. Abdi, N. Drouiche, H. Lounici, A. Pauss and M. F. A. Goosen, et al., Fish protein hydrolysate production from sardine solid waste by crude pepsin enzymatic hydrolysis in a bioreactor coupled to an ultrafiltration unit, Mater. Sci. Eng., C, 2012, 32(4), 922–928 CrossRef CAS.
- K. K. A. Sanjeewa, K. H. I. N. M. Herath, Y. S. Kim, J. Y. Jin and S. K. Kim, Trends in Analytical Chemistry Enzyme-assisted extraction of bioactive compounds from seaweeds and microalgae, Trends Anal. Chem., 2023, 167, 117266 CrossRef CAS.
- A. Naseri, G. S. Marinho, S. L. Holdt, J. M. Bartela and C. Jacobsen, Enzyme-assisted extraction and characterization of protein from red seaweed Palmaria palmata, Algal Res., 2020, 47, 101849 CrossRef.
- Y. H. Wang, K. C. hung, C. L. Lee and K. W. Cheng, Enzyme-Assisted Aqueous Extraction of Cobia Liver Oil and Protein Hydrolysates with antioxidant activity, Catalysts, 2020, 10(11), 1323 CrossRef CAS.
- H. Clupea, E. Aitta, A. Marsol-vall, A. Damerau and B. Yang, Enzyme-Assisted Extraction of Fish Oil from Whole Fish and by-Products of Baltic Herring (Clupea harengus membras), Foods, 2021, 10(8), 1811 CrossRef PubMed.
- Y. Dong, W. Yan and X. D. Zhang, Steam Explosion-Assisted Extraction of Protein from Fish Backbones and Effect of Enzymatic Hydrolysis on the Extracts, Foods, 2021, 10(8), 1942 CrossRef CAS.
- Q. T. Tran, T. V. Nguyen, H. V. Nguyen, T. T. Dang, T. V. Phan and C. T. Nguyen, Enzyme-assisted extraction and properties of collagen from Basa fish (Pangasius bocourti) skin Enzyme-assisted extraction and properties of collagen from Basa fish (Pangasius bocourti) skin, IOP Conf. Ser. Earth Environ. Sci., 2023, 1278(1), 012008 CrossRef.
- W. C. Lu, C. S. Chiu, Y. J. Chan, A. T. Mulio and P. H. Li, Characterization and biological properties of marine by-product collagen through ultrasound-assisted extraction, Aquac. Rep., 2023, 29, 101514 CrossRef.
- M. I. Shaik, N. F. Asrul Effendi and N. M. Sarbon, Functional properties of sharpnose stingray (Dasyatis zugei) skin collagen by ultrasonication extraction as influenced by organic and inorganic acids, Biocatal. Agric. Biotechnol., 2021, 35, 102103 CrossRef CAS.
- S. Pezeshk, M. Rezaei and M. Abdollahi, Impact of ultrasound on extractability of native collagen from tuna by-product and its ultrastructure and physicochemical attributes, Ultrason. Sonochem., 2022, 89, 106129 CrossRef CAS PubMed.
- G. Huang, S. Chen, C. Dai, L. Sun, W. Sun and Y. Tang, et al., Effects of ultrasound on microbial growth and enzyme activity, Ultrason. Sonochem., 2017, 37, 144–149 CrossRef CAS PubMed.
- T. Wu, X. Yu, A. Hu, L. Zhang, Y. Jin and M. Abid, Ultrasonic disruption of yeast cells: Underlying mechanism and effects of processing parameters, Innov. Food Sci. Emerg. Technol., 2015, 28, 59–65 CrossRef CAS.
- Y. Cao, T. Xia, G. Zhou and X. Xu, The mechanism of high pressure-induced gels of rabbit myosin, Innov. Food Sci. Emerg. Technol., 2012, 16, 41–46 CrossRef CAS.
- H. W. Huang, S. J. Wu, J. K. Lu, Y. T. Shyu and C. Y. Wang, Current status and future trends of high-pressure processing in food industry, Food Control, 2017, 72, 1–8 CrossRef.
- L. Chen, L. Ma, M. Zhou, Y. Liu and Y. Zhang, Effects of pressure on gelatinization of collagen and properties of extracted gelatins, Food Hydrocoll., 2014, 36, 316–322 CrossRef CAS.
- Z. Zhang, Y. Yang, P. Zhou, X. Zhang and J. Wang, Effects of high pressure modification on conformation and gelation properties of myofibrillar protein, Food Chem., 2017, 217, 678–686 Search PubMed.
- A. Aertsen, F. Meersman, M. E. G. Hendrickx, R. F. Vogel and C. W. Michiels, Biotechnology under high pressure: applications and implications, Trends Biotechnol., 2009, 27, 434–441 Search PubMed.
- X. Chen, P. J. Li, T. Nishiumi, H. Takumi, A. Suzuki and C. G. Chen, Effects of High-Pressure Processing on the Cooking Loss and Gel Strength of Chicken Breast Actomyosin Containing Sodium Alginate, Food Bioprocess Technol., 2014, 7(12), 3608–3617 Search PubMed.
- M. Cermeño, T. Kleekayai, M. Amigo-Benavent, P. Harnedy-Rothwell and R. J. FitzGerald, Current knowledge on the extraction, purification, identification, and validation of bioactive peptides from seaweed, Electrophoresis, 2020, 41, 1694–1717 CrossRef PubMed.
- S. Bleakley and M. Hayes, Algal proteins: Extraction, application, and challenges concerning production, Foods, 2017, 6(5), 1–34 Search PubMed.
- L. Buchmann, I. Brändle, I. Haberkorn, M. Hiestand and A. Mathys, Pulsed electric field based cyclic protein extraction of microalgae towards closed-loop biorefinery concepts, Bioresour. Technol., 2019, 291, 121870 CrossRef CAS PubMed.
- M. Coustets, N. Al-Karablieh, C. Thomsen and J. Teissié, Flow process for electroextraction of total proteins from microalgae, J. Membr. Biol., 2013, 751–760 Search PubMed.
- T. Ahmad, F. A. Masoodi, S. A. Rather, S. M. Wani and A. Gull, Supercritical Fluid Extraction: A Review, J. Biol. Chem. Chron., 2019, 5(1), 114–122 Search PubMed.
- M. Li, J. Lin, J. Chen and T. Fang, Pulsed Electric Field-Assisted Enzymatic Extraction of Protein from Abalone (Haliotis Discus Hannai Ino) Viscera, J. Food Process. Eng., 2016, 39(6), 702–710 CrossRef CAS.
- A. Yücetepe, Ö. Saroğlu and B. Özçelik, Response surface optimization of ultrasound-assisted protein extraction from Spirulina platensis: investigation of the effect of extraction conditions on techno-functional properties of protein concentrates, J. Food Sci. Technol., 2019, 56(7), 3282–3292 CrossRef PubMed.
- A. Yucetepe, A combination of osmotic shock and ultrasound pre-treatments and the use of enzyme for extraction of proteins from Chlorella vulgaris microalgae: Optimization of extraction conditions by RSM, J. Food Meas. Charact., 2022, 16(2), 1516–1527 Search PubMed.
- B. E. Abdelmalek, J. Gómez-Estaca, A. Sila, O. Martinez-Alvarez, M. C. Gómez-Guillén and S. Chaabouni-Ellouz, et al., Characteristics and functional properties of gelatin extracted from squid (Loligo vulgaris) skin, LWT, 2016, 65, 924–931 CrossRef CAS.
- M. A. Martínez, G. Velazquez, D. Cando, R. Núñez-Flores, A. J. Borderías and H. M. Moreno, Effects of high pressure processing on protein fractions of blue crab (Callinectes sapidus) meat, Innov. Food Sci. Emerg. Technol., 2017, 41, 323–329 CrossRef.
- I. Arredondo-Parada, W. Torres-Arreola, G. M. Suárez-Jiménez, J. C. Ramírez-Suárez, J. E. Juárez-Onofre and F. Rodríguez-Félix, et al., Effect of ultrasound on physicochemical and foaming properties of a protein concentrate from giant squid (Dosidicus gigas) mantle, LWT, 2020, 121, 108954 CrossRef CAS.
- S. G. O. Johansson, T. Bieber, R. Dahl, P. S. Friedmann, B. Q. Lanier and R. F. Lockey, et al., Revised nomenclature for allergy for global use: Report of the Nomenclature Review Committee of the World Allergy Organization, October 2003, J. Allergy Clin. Immunol., 2004, 113(5), 832–836 CrossRef CAS PubMed.
- X. Jiang and Q. Rao, Effect of processing on fish protein antigenicity and allergenicity, Foods, 2021, 10, 969 Search PubMed.
- B. Abraha, H. Admassu, A. Mahmud, N. Tsighe, X. W. Shui and Y. Fang, Effect of processing methods on nutritional and physico-chemical composition of fish: a review, MOJ. Food Process. Technol., 2018, 6(4), 376–382 Search PubMed.
- U. Griesmeier, M. Bublin, C. Radauer, S. Vázquez-Cortés, Y. Ma and M. Fernández-Rivas, et al., Physicochemical properties and thermal stability of Lep w 1, the major allergen of whiff, Mol. Nutr. Food Res., 2010, 54(6), 861–869 Search PubMed.
- B. Keshavarz, X. Jiang, Y. H. P. Hsieh and Q. Rao, Matrix effect on food allergen detection – A case study of fish parvalbumin, Food Chem., 2019, 274, 526–534 CrossRef CAS PubMed.
- Y. S. Chung, J. W. Lee and C. H. Chung, Molecular challenges in microalgae towards cost-effective production of quality biodiesel, Renew. Sustain. Energy Rev., 2017, 74, 139–144 CrossRef CAS.
- L. Gouveia, S. Graça, C. Sousa, L. Ambrosano, B. Ribeiro and E. P. Botrel, et al., Microalgae biomass production using wastewater: Treatment and costs. Scale-up considerations, Algal Res., 2016, 16, 167–176 CrossRef.
- M. L. Amorim, J. Soares, J. S. Coimbra, R. dos, M. De O. Leite, L. F. T. Albino and M. A. Martins, Microalgae proteins: production, separation, isolation, quantification, and application in food and feed, Crit. Rev. Food Sci. Nutr., 2021, 61, 1976–2002 CrossRef CAS PubMed.
- A. Mohammadi, S. Javadi, H. Yousefi, H. Pouraram and T. O. Randhir, A Framework for Assessing Food Baskets Based on Water and Carbon Footprints, Water, 2024, 16(9), 1196 CrossRef.
- M. Barkallah, M. Dammak, I. Louati, F. Hentati, B. Hadrich and T. Mechichi, et al., Effect of Spirulina platensis fortification on physicochemical, textural, antioxidant and sensory properties of yogurt during fermentation and storage, LWT, 2017, 84, 323–330 CrossRef CAS.
- M. Fradique, A. P. Batista, M. C. Nunes, L. Gouveia, N. M. Bandarra and A. Raymundo, Incorporation of Chlorella vulgaris and Spirulina maxima biomass in pasta products. Part 1: Preparation and evaluation, J. Sci. Food Agric., 2010, 90(10), 1656–1664 CrossRef CAS PubMed.
- S. Khan, A. Rehman, H. Shah, R. M. Aadil, A. Ali and Q. Shehzad, et al., Fish Protein and Its Derivatives: The Novel Applications, Bioactivities, and Their Functional Significance in Food Products, Food Rev. Int., 2020, 38(8), 1607–1634 CrossRef.
- K. Prameela, K. Venkatesh, S. B. Immandi, A. P. K. Kasturi, C. Rama Krishna and C. Murali Mohan, Next generation nutraceutical from shrimp waste: The convergence of applications with extraction methods, Food Chem., 2017, 237, 121–132 CrossRef CAS PubMed.
- N. M. Sachindra and N. S. Mahendrakar, Process optimization for extraction of carotenoids from shrimp waste with vegetable oils, Bioresour. Technol., 2005, 96(10), 1195–1200 CrossRef CAS PubMed.
- K. Yamamoto, K. Y. Hara, T. Morita, A. Nishimura, D. Sasaki and J. Ishii, et al., Enhancement of astaxanthin production in Xanthophyllomyces dendrorhous by efficient method for the complete deletion of genes, Microb. Cell Fact., 2016, 15(1), 155 CrossRef PubMed.
- A. R. Shaviklo, Development of fish protein powder as an ingredient for food applications: a review, J. Food Sci. Technol., 2015, 52, 648–661 CrossRef CAS PubMed.
- W. C. Lu, C. S. Chiu, Y. J. Chan, A. T. Mulio and P. H. Li, Characterization and biological properties of marine by-product collagen through ultrasound-assisted extraction, Aquac. Rep., 2023, 29, 101514 CrossRef.
- S. Jacobsson, E. Alirol and M. Unemo, In vitro activity of the ketolide cethromycin in multidrug-resistant clinical Neisseria gonorrhoeae isolates and international reference strains, J. Chemother., 2019, 31(5), 246–251 CrossRef CAS PubMed.
- W. Zhang, I. D. Boateng and J. Xu, Novel marine proteins as a global protein supply and human nutrition: Extraction, bioactivities, potential applications, safety assessment, and deodorization technologies, Trends Food Sci. Technol., 2024, 143, 104283 CrossRef CAS.
- V. Gudipati, Fish Gelatin: A Versatile Ingredient for the Food and Pharmaceutical Industries, Marine Proteins and Peptides: Biological Activities and Applications, 2013, pp. 271–295 Search PubMed.
- K. Samarakoon and Y. J. Jeon, Bio-functionalities of proteins derived from marine algae - A review, Food Res. Int., 2012, 48(2), 948–960 Search PubMed.
- A. Sasidharan, T. Rustad and G. M. Cusimano, Tuna sidestream valorization: a circular blue bioeconomy approach, Environ. Sci. Pollut. Res., 2024, 31(53), 62230–62248 CrossRef PubMed.
- R. Arena, G. Renda, G. Ottaviani Aalmo, F. Debeaufort, C. M. Messina and A. Santulli, Valorization of the invasive blue crabs (Callinectes sapidus) in the Mediterranean: nutritional value, bioactive compounds and sustainable by-products utilization, Mar. Drugs, 2024, 22(9), 430 CrossRef CAS PubMed.
- E. Tamburini, The blue treasure: comprehensive biorefinery of blue crab (Callinectes sapidus), Foods, 2024, 13(13), 2018 CrossRef CAS PubMed.
- D. Summa, M. Lanzoni, G. Castaldelli, E. A. Fano and E. Tamburini, Trends and opportunities of bivalve shells' waste valorization in a prospect of circular blue bioeconomy, Resources, 2022, 11(5), 48 CrossRef.
- M. Bürck, C. Fratelli, M. Campos Assumpção de Amarante and A. R. C. Braga, Unveiling the potential of Spirulina biomass—A Glimpse into the future circular economy using green and blue ingredients, Biomass, 2024, 4(3), 704–719 Search PubMed.
- P. Patel, Y. L. J. Pang, W. J. Choi and A. Wong, Protein extraction and isolation from legumes and algae: an industry primer, Food Bioprocess Technol., 2025, 18(10), 8380–8408 Search PubMed.
- M. Du, Z. Hou, L. Liu, Y. Xuan, X. Chen and L. Fan, et al., 1Progress, applications, challenges and prospects of protein purification technology, Front. Bioeng. Biotechnol., 2022, 10, 1028691 CrossRef PubMed.
- Y. Fu, N. Zhang and Y. Zhang, Proteins from blue foods to meet the demand in the food sector, Trends Food Sci. Technol., 2024, 153, 104722 CrossRef CAS.
- S. Minić, N. Gligorijević, L. Veličković and M. Nikolić, Narrative review of the current and future perspectives of phycobiliproteins' applications in the food industry: From natural colors to alternative proteins, Int. J. Mol. Sci., 2024, 25(13), 7187 Search PubMed.
- R. A. Williams, Opportunities and challenges for the introduction of new food proteins, Annu. Rev. Food Sci. Technol., 2021, 12(1), 75–91 CrossRef PubMed.
Footnote |
| † Equally contributed. |
| This journal is © The Royal Society of Chemistry 2026 |