Supercritical fluid extraction of black cardamom (Amomum subulatum) oleoresin: a sustainable approach for enhanced yield, phytochemical composition, and antioxidant potential
Received
30th October 2025
, Accepted 5th January 2026
First published on 6th January 2026
Abstract
Oleoresin extraction from black cardamom is highly valuable due to its rich composition of bioactive compounds, yet it remains underexplored, with no industrial-scale or systematic studies, leaving a critical gap in the valorisation of this underutilised spice. This study addresses the need for an advanced technique by investigating the potential of supercritical fluid extraction (SFE) to efficiently extract high-quality oleoresin. The primary aim is to optimise the SFE process parameters by systematically varying pressure (15–40 MPa), temperature (30–50 °C), and CO2 flow rate (3–50 g min−1), with ethanol as the cosolvent. The highest oleoresin yield (8.80% ± 0.12%) was achieved at 30 MPa, 50 °C, and 50 g min−1, with increased energy efficiency (2.93 ± 0.03 g kW−1 h−1) and a favourable solvent-to-feed ratio (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). GC-MS profiling revealed superior retention of aroma-active volatiles (1,8-cineole, terpineol, and α-terpinyl acetate) under SFE, whereas GC-FID analysis revealed a nutritionally beneficial fatty acid profile dominated by oleic and linoleic acids. FTIR analysis confirmed the presence of esters, phenolics, and terpenoids. The extracts obtained under optimal conditions presented the highest total phenolic content (90.61 ± 2.25 mg GAE per g), total flavonoid content (124.96 ± 0.24 mg QE per g), and antioxidant activity (DPPH-88.63% ± 0.02%, ABTS-63.28% ± 0.15%, and FRAP-79.02 ± 0.24 µmol Fe2+ per g), significantly outperforming those obtained under all other conditions (p < 0.05). The residual ethanol concentration was within regulatory limits (<12.53 mg kg−1), confirming food-grade applicability. The total volatile and non-volatile contents, colorimetric attributes, and extraction efficiency were significantly influenced by pressure. This green extraction approach offers a sustainable method to increase the bioactive and sensory value of black cardamom oleoresin for food, nutraceutical, and cosmetic applications.
1). GC-MS profiling revealed superior retention of aroma-active volatiles (1,8-cineole, terpineol, and α-terpinyl acetate) under SFE, whereas GC-FID analysis revealed a nutritionally beneficial fatty acid profile dominated by oleic and linoleic acids. FTIR analysis confirmed the presence of esters, phenolics, and terpenoids. The extracts obtained under optimal conditions presented the highest total phenolic content (90.61 ± 2.25 mg GAE per g), total flavonoid content (124.96 ± 0.24 mg QE per g), and antioxidant activity (DPPH-88.63% ± 0.02%, ABTS-63.28% ± 0.15%, and FRAP-79.02 ± 0.24 µmol Fe2+ per g), significantly outperforming those obtained under all other conditions (p < 0.05). The residual ethanol concentration was within regulatory limits (<12.53 mg kg−1), confirming food-grade applicability. The total volatile and non-volatile contents, colorimetric attributes, and extraction efficiency were significantly influenced by pressure. This green extraction approach offers a sustainable method to increase the bioactive and sensory value of black cardamom oleoresin for food, nutraceutical, and cosmetic applications.
Sustainability spotlight
The valorization of underutilized black cardamom via SFE supports greener and more energy-efficient food processing technologies. The conventional methods of solvent extraction are energy-intensive, generate chemical waste, and cause the deterioration of heat-sensitive bioactives. This study illustrates that SFE enhanced yield, reduced the use of organic solvents, minimized thermal degradation, and enabled full recovery of the solvent used in the process, promoting circular resource usage. Improving extraction efficiency and maintaining food-grade purity will help attain sustainability in industrial practices and develop high-value products across the food, nutraceutical, and cosmetic industries. The work is related to UN SDGs 9 (Industry, Innovation, and Infrastructure), 12 (Responsible Consumption and Production), and 13 (Climate Action) by improving eco-friendly extraction technologies for natural bioresources.
|
1 Introduction
Spices have played a vital role throughout human history, serving as key ingredients in culinary, medicinal, food, and pharmaceutical applications. Derivatives of various plant parts, such as seeds, bark, roots, and leaves, are highly valued for their distinctive flavours, aromas, and bioactive properties.1 They are rich in essential oils, phenolic compounds, and flavonoids, enhance taste, impart fragrance, and offer potential health benefits. In addition to their culinary appeal, spices exhibit antimicrobial, antioxidant, and anti-inflammatory properties, making them valuable in food preservation and pharmaceutical applications.2 Among the different spices, black cardamom (Amomum subulatum) holds greater significance. It is distinct from green cardamom (Elletaria cardamom), a ‘queen of spices’ with a different flavour. Black cardamom is a perennial herbaceous plant belonging to the Zingiberaceae family.3 It is native to the Eastern Himalayas and cultivated extensively in regions such as India, Nepal, and Bhutan. Known for its smoky, earthy aroma and robust flavour, black cardamom holds a significant place in culinary traditions, particularly in South Asian and Middle Eastern cuisines. In addition to its culinary uses, black cardamom has been traditionally valued in herbal medicine because of its rich composition of bioactive compounds and essential oils.4
The use of extracted oleoresin from black cardamom is essential in various food and pharmaceutical applications. Essential oils, which primarily consist of volatile compounds, are typically obtained through steam or hydrodistillation. In contrast, oleoresins are extracted via food-grade solvents, yielding concentrated, semi-solid spice extracts containing both volatile and non-volatile components that together contribute to the overall flavour. These extracts capture the full spectrum of flavour and aroma compounds, offering a more consistent and shelf-stable alternative to whole or ground spices.5 The efficiency and quality of oleoresin extraction largely depend on the method used.6 Traditional techniques, such as cold extraction, Soxhlet extraction, and hydrodistillation, are widely used but have notable limitations. Cold extraction, performed at low temperatures, helps to retain heat-sensitive bioactive compounds but results in lower yields. Soxhlet extraction, which uses continuous solvent reflux, improves yield but exposes oleoresin to prolonged heat, risking degradation of volatile compounds. These conventional methods often suffer from limitations, such as solvent residues, thermal degradation, and lower extraction yields.7 These drawbacks raise sustainability concerns related to energy consumption, solvent waste, and environmental impact, necessitating the development of greener extraction alternatives.
In response to these limitations, advanced techniques, such as supercritical fluid extraction (SFE), microwave-assisted extraction (MAE), pulsed electric field (PEF), and ultrasound-assisted extraction (UAE), are reasonable alternatives. Among these, SFE is an advanced and eco-friendly technique that efficiently extracts temperature-sensitive compounds using supercritical CO2 as the solvent.7 Under controlled high-pressure and high-temperature conditions, CO2 has both liquid and gas-like properties, allowing it to efficiently penetrate the plant matrix and dissolve bioactive compounds. SFE offers significant advantages over conventional methods, including higher selectivity, minimal thermal degradation, and solvent-free extraction, making it particularly suitable for food and pharmaceutical applications.8 However, the widespread adoption of SFE remains limited due to the high cost of equipment and the need for process optimization to achieve maximum yield and bioactive compound retention. Conventional techniques, such as Soxhlet extraction, are still regarded as standard methods for evaluating the effectiveness of newly developed approaches. However, non-conventional methods are increasingly gaining attention because of their eco-friendliness, faster processing times, and enhanced extraction efficiency.9 Although SFE has been successfully applied to various spices such as black pepper and turmeric,10,11 its application to black cardamom remains underutilised and relatively unexplored, especially in the context of value-added and industrial applications. This study addresses this gap by performing a systematic evaluation of SFE alongside multiple conventional methods for black cardamom oleoresin. This research also evaluated extraction performance on the basis of yield, bioactive compound profile, antioxidant activity, and functional properties, providing insights into the efficiency and industrial relevance of SFE for black cardamom.
Therefore, this study aims to optimise SFE process conditions for improving the yield of black cardamom oleoresin by systematically evaluating the effects of pressure and temperature, while keeping the CO2 flow rate, extraction time, and co-solvent flow rate constant. The oleoresin yield obtained from optimised SFE was compared with that from various conventional extraction methods. This research provides insights into extraction efficiency, sustainability metrics, and product quality. Furthermore, GC-MS profiling is conducted on all high-yielding oleoresins to analyse and compare their volatile and semi-volatile bioactive compound profiles. The highest-yielding oleoresin obtained from SFE is further evaluated for residual solvent content, physicochemical properties, fatty acid composition, and functional attributes to determine its potential for use in food and pharmaceutical formulations.
2 Materials and methods
2.1 Materials
Authentic black cardamom (Amomum subulatum) pods were obtained from the Indian Cardamom Research Institute, Sikkim, India. Food-grade ethanol (99% pure), n-hexane, acetone, Folin-Ciocalteu's reagent, gallic acid, DPPH, ABTS, and FRAP were purchased from Sigma-Aldrich, Bangalore, India.
2.2 Methods
2.2.1 Sample preparation.
Black cardamom samples with a moisture content of 8.5% ± 0.3% (w.b) were pulverized and sieved to obtain a uniform particle size of <1000 µm using a sieving machine (Vibratory Sieve Shaker, AS 200 Basic, Jayant Scientific India, Mumbai, India). Each trial was carried out with 100 g of sieved black cardamom. Ethanol was selected as the solvent across all methods because of its GRAS (Generally Recognized as Safe) status, food safety, and lower toxicity compared to hexane, ensuring suitability for food applications.
2.2.2 SFE extraction procedure.
Black cardamom oleoresin was extracted using SFE as described by Abdullah et al. (2022).12 The prepared sample was wrapped in a muslin cloth to ensure consistent flow and prevent channelling and loaded into the laboratory-scale SFE (SFE 500, Thar Technologies, USA). A schematic diagram of the SFE extractor with major components is shown in Fig. S1. CO2 (99.99% purity) was used as the primary extraction solvent. Ethanol was used as a co-solvent at 1 mL min−1 to increase the solubility of moderately polar compounds, such as flavonoids and phenolics, which have limited solubility in pure CO2.13 The selected low concentration was optimized to balance selectivity while avoiding significant solvent residues. A six-zone temperature controller and automated backpressure regulator controlled the conditions inside the extractor. The extraction time was fixed at 60 min, as preliminary trials indicated no significant improvement in yields beyond this duration. Therefore, 60 min was considered the optimal extraction time for this study.
2.2.3 Single-factor optimization of SFE parameters on oleoresin yield.
To optimize the SFE extraction conditions, a single-factor experimental design was conducted by systematically varying individual process parameters while keeping others constant. The experimental parameters are presented in Table 1. Temperature and CO2 flow rate (g min−1) variations were conducted independently at different pressures to evaluate their interactive effects on extraction efficiency.
Table 1 Single-factor experimental design for SFE optimization
| Parameters |
Level 1 |
Level 2 |
Level 3 |
Level 4 |
Level 5 |
Level 6 |
| Pressure (MPa) |
15 |
20 |
25 |
30 |
35 |
40 |
| CO2 flow rate (g min−1) |
30, 40, 50 |
30, 40, 50 |
30, 40, 50 |
30, 40, 50 |
30, 40, 50 |
30, 40, 50 |
| Temperature (°C) |
30, 40, 50 |
30, 40, 50 |
30, 40, 50 |
30, 40, 50 |
30, 40, 50 |
30, 40, 50 |
2.2.4 Extraction kinetics of oleoresin yield.
To evaluate extraction kinetics, periodic sampling was performed at 10 min intervals throughout a 60 min extraction period. Each collected fraction was immediately subjected to rotary evaporation (IKA RV 10, Bangalore, India) under reduced pressure (≤40 °C) to remove residual ethanol and moisture prior to determining the yields. Final yields were calculated on the basis of oleoresin weight after complete solvent removal. After a sufficient volume of oleoresin was obtained, the extract was stored in amber bottles at 4 °C for further analysis.
2.2.5 Energy and solvent utilization efficiency of SFE.
The sustainability and operational performance of the supercritical CO2 (SCCO2) extraction process were assessed by analysing the energy demand and solvent usage of the system. Key operational variables such as extraction time, CO2 flow rate, and the power requirements of major equipment (compressor, heater, and pump) were monitored. These parameters were used to quantify process efficiency indicators related to energy consumption and solvent.
2.2.5.1 Energy consumption, solvent-to-feed ratio, and process efficiency analysis.
The performance of the supercritical CO2 (SCCO2) extraction process was quantitatively assessed by analysing energy requirements and solvent usage under different operational parameters, including CO2 flow rate, extraction time, and system components' power ratings. Energy consumption (kWh), solvent-to-feed ratio (kg CO2 per kg raw material), energy efficiency (g oil kW−1 h−1) and energy consumption per kilogram of CO2 were calculated to assess the overall process sustainability and performance.
2.2.5.2 CO2 solvent-to-feed ratio.
The solvent-to-feed ratio was determined by calculating the total mass of CO2 consumed per kilogram of the raw material. The CO2 mass was calculated based on the formulas (1) and (2).| |  | (1) |
| |  | (2) |
This metric indicates the volume of solvent required to extract oleoresin from 1 kg of feedstock under each condition.
2.2.5.3 Energy consumption estimation.
Total energy consumption was estimated by summing the individual power inputs of the major components of the SFE apparatus, including the CO2 pump, heaters (two zones), chiller, and control system, which are shown in Table 2. The power ratings represent the manufacturer-specified nominal values for each component. Actual power consumption during operation may vary depending on operational load and efficiency. These nominal values were used to establish relative energy consumption patterns across different extraction conditions within the laboratory-scale system. The total energy consumption (kWh) for each experimental condition was calculated using eqn (3).| | | Energy consumption (kWh) = ∑(Pi × t) | (3) |
Here, Pi is the power rating (kW) of each component and t is the extraction time (h).
Table 2 Estimated power rating and energy consumption of laboratory-scale supercritical CO2 extraction system components
| Component |
Power rate |
Energy consumption |
| CO2 pump |
∼1.5 kW |
1.5 kWh |
| Heater (2 zones) |
∼0.5–1.0 kW each |
1.0–2.0 kWh |
| Chiller |
∼0.5 kW |
0.5 kWh |
| Control system |
∼0.1 kW |
3.0–4.0 kWh |
2.2.5.4 Energy efficiency.
Energy efficiency and consumption per kilogram of CO2 were calculated based on eqn (4) and (5):| | 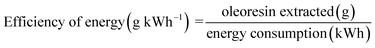 | (4) |
| | 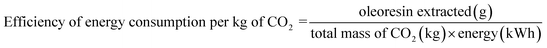 | (5) |
2.2.5.5 CO2-specific extraction efficiency per unit solvent.
| |
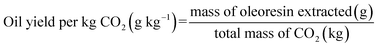
| (6) |
2.2.6 Comparison with other methods on oleoresin yield.
To understand the effectiveness of SFE and to show general performance differences, black cardamom oleoresin was also extracted using hydrodistillation, Soxhlet extraction, cold extraction, and ultrasound-assisted solvent extraction (UASE). Hydrodistillation was performed using a Clevenger-type apparatus with 100 g of the powdered sample and 1.5 L of distilled water (solid-to-liquid ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15, w/v), heated at 0.8–1.0 kW to maintain a temperature of 95–100 °C for 3–4 h.4 Soxhlet extraction was carried out using 500 mL of ethanol (1
15, w/v), heated at 0.8–1.0 kW to maintain a temperature of 95–100 °C for 3–4 h.4 Soxhlet extraction was carried out using 500 mL of ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, w/v), with the heating mantle operated at 350–400 W to maintain the temperature at 60–80 °C for 6 h (12–14 cycles h−1).14 Cold extraction involved soaking 100 g of the sample in 200 mL of ethanol (1
5, w/v), with the heating mantle operated at 350–400 W to maintain the temperature at 60–80 °C for 6 h (12–14 cycles h−1).14 Cold extraction involved soaking 100 g of the sample in 200 mL of ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, w/v) at 25 °C for 1 h with mild magnetic stirring at 150 rpm, without external heating.15 For UASE, the sample was mixed with 300 mL of ethanol (1
2, w/v) at 25 °C for 1 h with mild magnetic stirring at 150 rpm, without external heating.15 For UASE, the sample was mixed with 300 mL of ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, w/v) and subjected to ultrasonic treatment at 250 W power and 40% amplitude, operating at 50 Hz for 20–30 min while maintaining the temperature between 30 and 50 °C.16 All extracts were subsequently filtered, and the solvents were removed using a rotary evaporator (RV 10 digital V, IKA, India) under reduced pressure at ≤40 °C. These conventional methods were not intended for direct comparison with SFE, as they differ significantly in extraction time, temperature, and solvent-feed ratios, and were included to provide a practical benchmark for relative extraction performance.
3, w/v) and subjected to ultrasonic treatment at 250 W power and 40% amplitude, operating at 50 Hz for 20–30 min while maintaining the temperature between 30 and 50 °C.16 All extracts were subsequently filtered, and the solvents were removed using a rotary evaporator (RV 10 digital V, IKA, India) under reduced pressure at ≤40 °C. These conventional methods were not intended for direct comparison with SFE, as they differ significantly in extraction time, temperature, and solvent-feed ratios, and were included to provide a practical benchmark for relative extraction performance.
2.3 Analytical techniques
2.3.1 Gas chromatography-mass spectrometry (GC-MS) analysis.
GC-MS analysis was performed using a Shimadzu GC-MS-QP2010 SE with an HP5-MS column. Helium was used as the carrier gas (1 mL min−1). The oven temperature was programmed from 50 °C to 250 °C at 4 °C min−1. The injector and detector were set at 210 °C and 230 °C, respectively, with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 split ratio and 70 eV ionization energy. Compound identification was performed by matching mass spectra with the NIST and Wiley spectral libraries, using a minimum similarity index threshold of ≥80%. Retention indices (RI) were calculated using a homologous series of n-alkanes (C8–C30) analyzed under identical chromatographic conditions and were further confirmed by comparison with reported literature values from established databases.17
10 split ratio and 70 eV ionization energy. Compound identification was performed by matching mass spectra with the NIST and Wiley spectral libraries, using a minimum similarity index threshold of ≥80%. Retention indices (RI) were calculated using a homologous series of n-alkanes (C8–C30) analyzed under identical chromatographic conditions and were further confirmed by comparison with reported literature values from established databases.17
2.3.2 Residual solvent analysis by headspace gas chromatography (HS-GC).
Residual solvent analysis was carried out using headspace gas chromatography (HS-GC) on an Agilent 7890B GC-FID system with a G1888 headspace sampler. For each analysis, 20 mg of oleoresin was placed in a 20 mL headspace vial, and 1 mL of hexane was added as the diluent. The vial was sealed and equilibrated at 80 °C for 30 minutes. Headspace vapors were injected onto a DB-624 column (30 m × 0.32 mm, 1.8 µm) using helium as the carrier gas at 1.0 mL min−1. The oven temperature was programmed from 40 °C (5 min hold) to 200 °C at 10 °C min−1. Solvents were identified by comparing retention times with those of ethanol as standards and quantified using external calibration.
2.3.3 GC-FID.
Fatty acid profiling of black cardamom oleoresin was conducted using gas chromatography coupled with a flame ionization detector (GC-FID) (GC-17, Shimadzu, Japan), following the protocol described by Subtain et al. (2024).18 A DB-WAX (30 m, 0.25 mm) column was used, with nitrogen gas as the mobile phase at a flow rate of 30 mL min−1. The initial oven temperature was set at 140 °C for 5 min, and then gradually increased to 240 °C at a rate of 4 °C min−1. After maintaining 240 °C for 30 min, the temperature was further increased to 250 °C before sample injection. The analysed samples were compared with FAME (Fatty Acid Methy Ester – fatty acid standards), and calculated retention indices (RI) using homologous series of n-alkane standards (C8–C24) under identical chromatographic conditions. The calculated RIs were compared with literature values from the NIST Chemistry WebBook for polar columns (DB-WAX equivalent), with acceptance criteria of ±10 RI units, and the detected fatty acids were quantified as percentages.
2.3.4 FTIR-ATR measurement of black cardamom oleoresin.
The spectra of the extracted oleoresin were measured using attenuated total reflectance (FTIR-ATR, Bruker Vertex 70v, USA) within the wavenumber range of 400 to 4000 cm−1.
2.4 Quality attributes
2.4.1 Total volatile and non-volatile contents.
The total volatile and non-volatile contents of oleoresins were determined using a modified protocol based on Subtain et al. (2024),18 with modifications. A total of 1 g of each independent replicate was taken from the homogenized batch oleoresin sample and placed in a hot air oven (LabTech, LDO-150 N, Gyeonggi-do, Korea) at 100 °C for 45 min. After heating, the sample was reweighed, and the weight loss was recorded as the total volatile content, while the remaining weight represented the total non-volatile content. Both values are expressed as percentages.
2.4.2 Color measurement.
The surface color characteristics of oleoresin were evaluated using a colorimeter (ColorQuest XE, Hunter Lab, Virginia, USA). Measurements were performed in triplicate to ensure accuracy, and the results of L* (lightness), a* (red-green axis), b* (yellow-blue axis), and dE values were expressed in triplicate.
2.5 Phytochemical analysis
2.5.1 Total phenol content (TPC).
The total phenolic content (TPC) was determined using the Folin–Ciocalteu method with slight modifications, as outlined by Madhusankha et al. (2023).14 In brief, oleoresin was dissolved in methanol to a concentration of 1 mg mL−1. From this, 200 µL of the solution was mixed with 1 mL of 10% Folin–Ciocalteu reagent, followed by the addition of 3 mL of 75% sodium carbonate (Na2CO3). The mixture was then incubated at 20 °C for 30 min. Absorbance was measured at 765 nm using a spectrophotometer. TPC was quantified using a gallic acid calibration curve, as shown in Fig. S3A, and TPC was expressed in mg GAE per g.
2.5.2 Total flavonoid content (TFC).
Initially, 1 mL of oleoresin (diluted in methanol at 1 mg mL−1) was mixed with 4 mL of deionized water and 0.3 mL of 5% sodium nitrite (NaNO3). After 5 min, 0.3 mL of 10% aluminium chloride hexahydrate (AlCl3·6H2O) and 2 mL of 1 M sodium hydroxide (NaOH) were added, following the method described by Madhusankha et al. (2023).14 The absorbance was measured at 510 nm using a spectrophotometer. TFC was quantified using a quercetin calibration curve, as shown in Fig. S3B, and expressed in mg QE per g.
2.6 Antioxidant properties
Antioxidant activity of the oleoresins was assessed using three different assays, following the procedure outlined by Madhusankha et al. (2023),14 with minor modifications. To mitigate potential precipitation issues typical in antioxidant assays involving oleoresin, all samples were first dissolved in methanol and vortexed thoroughly to ensure homogeneity prior to reagent addition.
2.6.1 DPPH assay.
DPPH radical scavenging activity was evaluated by preparing a 0.1 mM methanolic solution of 2,2-diphenyl-1-picrylhydrazyl (DPPH), and 900 µL of this solution was mixed with 100 µL of a 1 mg mL−1 sample. The mixture was then vortexed and incubated in the dark for 30 min. After incubation, the absorbance was measured at 517 nm. The antioxidant potential was calculated by the following eqn (7):| |  | (7) |
2.6.2 ABTS assay.
The ABTS assay involved preparing a radical solution by dissolving 7.4 mM ABTS in water and reacting it with 2.6 mM potassium persulfate. This mixture was kept in the dark for 12–16 h to facilitate radical formation. The resulting ABTS+ solution was then diluted with methanol to obtain an absorbance of 0.700 at 734 nm. For the test, 100 µL of the sample was combined with 900 µL of the ABTS+ solution and incubated at 20 °C in the dark for 30 min. The absorbance was measured at 734 nm and calculated using eqn (8):| |  | (8) |
2.6.3 Ferric reducing antioxidant power (FRAP) assay.
The FRAP reagent was freshly prepared before each experiment by mixing 300 mM acetate buffer (pH 3.6), 10 mM 2,4,6-tris-(2-pyridyl)-s-triazine (TPTZ) in 40 mM HCl, and 20 mM FeCl3 in a 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) ratio. For the assay, 90 µL of the sample (1 mg mL−1) was mixed with 270 µL of methanol and 2.7 mL of FRAP reagent, followed by incubation at 37 °C for 30 min. The absorbance was measured at 595 nm, and the antioxidant capacity was quantified using a Trolox calibration curve quantified using a Trolox standard curve (Fig S3C), with values expressed as µM Fe+2 per g.
1 (v/v) ratio. For the assay, 90 µL of the sample (1 mg mL−1) was mixed with 270 µL of methanol and 2.7 mL of FRAP reagent, followed by incubation at 37 °C for 30 min. The absorbance was measured at 595 nm, and the antioxidant capacity was quantified using a Trolox calibration curve quantified using a Trolox standard curve (Fig S3C), with values expressed as µM Fe+2 per g.
2.7 Statistical analysis
All experiments were conducted as three independent extractions (n = 3), with each sample analyzed in triplicate (technical replicates). Values are presented as means ± standard deviations (SD). Statistical differences among groups were assessed using one-way analysis of variance (ANOVA), followed by Duncan's multiple-range test at a significance level of p < 0.05. Statistical analyses were performed using SPSS software (IBM SPSS Statistics, Version 27, Chicago, IL, USA). Different superscript letters indicate significant differences between means (p < 0.05).
3 Results and discussion
3.1 Single-factor optimization of SFE parameters and other extraction methods on oleoresin yield
The extraction yield of black cardamom oleoresin using SFE was influenced by single-factor experimental design on pressure, temperature, and CO2 flow rate as described in the methods section and shown in Table 3. The pressure, which is crucial for solute solubility and SCCO2 density, was optimized first, followed by temperature and CO2 flow rate. With increasing pressure from 15 to 40 MPa, oleoresin at 30 MPa, 50 g min−1, and 50 °C, SFE yielded 8.80% ± 0.12% with superior selectivity for aroma-active monoterpenes, which aligns with findings of Nagavekar and Singhal (2018)10 on pepper oleoresin extraction. The yield varied between 1.80% ± 0.03% and 8.80% ± 0.12%, with the highest yield occurring at 30 MPa, 50 g min−1 CO2 flow rate, and 50 °C, and the lowest (1.80% ± 0.03%) at 40 MPa, 30 g min−1 CO2 flow rate, and 30 °C. This variation is attributed to the optimal balance at 30 MPa, while excessive pressure at 40 MPa may reduce efficiency by decreasing CO2 diffusivity.
Table 3 Effect of extraction conditions on pressure, CO2 flow rate, and temperature on oleoresin yield, and energy efficiencya
| Pressure (MPa) |
CO2 flow rate (g min−1) |
Temperatures (°C) |
Yield (%) |
Amount of oil extracted per kg of CO2 (g kg−1) |
Energy efficiency (g kW−1 h−1) |
Efficiency of energy consumption per kg of CO2 (g kg−1 kWh−1) |
|
Values are expressed as means ± SD (n = 3 independent experiments). Different superscript letters within the same column indicate statistically significant differences (p < 0.05).
|
| 15 |
30 |
30 |
1.98 ± 0.17i |
1.10 ± 0.02 fg |
0.83 ± 0.01k |
2.64 ± 0.02k |
| 15 |
40 |
40 |
2.01 ± 0.17i |
0.84 ± 0.02gh |
0.74 ± 0.02l |
2.26 ± 0.01n |
| 15 |
50 |
50 |
2.50 ± 0.04hi |
0.83 ± 0.01gh |
0.83 ± 0.02k |
2.50 ± 0.01l |
| 20 |
30 |
30 |
2.50 ± 0.05hi |
1.39 ± 0.01f |
1.04 ± 0.02j |
3.33 ± 0.02i |
| 20 |
40 |
40 |
2.20 ± 0.17hi |
0.92 ± 0.02gh |
0.81 ± 0.02k |
2.50 ± 0.02l |
| 20 |
50 |
50 |
3.20 ± 0.17g |
1.07 ± 0.02e |
1.07 ± 0.01ij |
3.20 ± 0.02j |
| 25 |
30 |
30 |
3.00 ± 0.15 fg |
1.67 ± 0.01e |
1.25 ± 0.01g |
4.00 ± 0.01g |
| 25 |
40 |
40 |
3.00 ± 0.10 fg |
1.25 ± 0.02 fg |
1.11 ± 0.02j |
3.46 ± 0.01h |
| 25 |
50 |
50 |
3.50 ± 0.20f |
1.17 ± 0.01 fg |
1.17 ± 0.01h |
3.50 ± 0.01h |
| 30 |
30 |
30 |
8.20 ± 0.10c |
4.56 ± 0.02a |
3.42 ± 0.03a |
10.93 ± 0.03a |
| 30 |
40 |
40 |
8.50 ± 0.05b |
3.54 ± 0.03b |
3.15 ± 0.03b |
9.62 ± 0.03b |
| 30 |
50 |
50 |
8.80 ± 0.12a |
2.93 ± 0.02c |
2.93 ± 0.03c |
8.80 ± 0.03c |
| 35 |
30 |
30 |
3.80 ± 0.02d |
2.11 ± 0.02d |
1.58 ± 0.02d |
5.06 ± 0.02d |
| 35 |
40 |
40 |
4.10 ± 0.05e |
1.71 ± 0.01e |
1.52 ± 0.01e |
4.61 ± 0.01e |
| 35 |
50 |
50 |
4.20 ± 0.02e |
1.40 ± 0.02ef |
1.40 ± 0.01f |
4.20 ± 0.01f |
| 40 |
30 |
30 |
1.80 ± 0.03j |
1.00 ± 0.01gh |
0.75 ± 0.02l |
2.40 ± 0.01m |
| 40 |
40 |
40 |
1.90 ± 0.02i |
0.79 ± 0.03h |
0.70 ± 0.02m |
2.14 ± 0.02o |
| 40 |
50 |
50 |
2.10 ± 0.10hi |
0.70 ± 0.02i |
0.70 ± 0.01m |
2.10 ± 0.01o |
Pressure is a critical parameter in SFE, and directly influences the CO2 density and solvating power. Increased pressure generally enhances solubility and mass transfer.19 At lower pressures (15–25 MPa), the extraction yield was relatively low (1.98% ± 0.17% to 3.50% ± 0.20%), which is consistent with the findings of Nagavekar and Singhal (2018).10 At 30 MPa, the yield improved significantly to 4.20% ± 0.02%, achieving the maximum yield (8.80% ± 0.12%), indicating an optimal pressure for oleoresin extraction, similar to findings of Soldan et al. (2021)20 for capsicum oleoresin. The increased solvent strength at higher densities facilitated compound dissolution and diffusion, which favored the recovery of both volatile and non-volatile constituents. Nagavekar and Singhal (2019)11 reported increased oil yields up to 30 MPa, beyond which matrix compaction occurred, restricting solvent penetration and reducing mass transfer. At 40 MPa, reduced CO2 diffusivity and matrix compaction reduced the mass transfer, lowering extraction efficiency. Excessive pressure above 30–35 MPa reduces matrix permeability and solute transport due to higher viscosity and diffusivity, a similar trend also observed by Soldan et al. (2021).20 Prolonged exposure to high pressure may degrade phenolic compounds and flavonoids, negatively impacting their antioxidant properties. Thus, 30 MPa is optimal for maximizing yield and bioactive retention.
Temperature has a dual effect on SFE, influencing its solubility and SCCO2 density. At constant pressure, increased temperature enhances the solute vapor pressure and extraction efficiency, although excessively high temperatures reduce the solvent power. The yield consistently increased with temperature (30 °C to 50 °C) across all pressures, which is consistent with the findings of Nagavekar and Singhal (2018)10 and is likely due to improved mass transfer and CO2 vapor pressure. In this study, the highest yield was obtained at 50 °C, indicating an optimal balance of solvating capacity and solute volatility, in agreement with the findings of Martinez-Correa et al. (2017),17 who reported that 60–65 °C was optimal for the extraction of curcuminoid-rich oleoresin.
3.2 Extraction kinetics of oleoresin yield
The extraction kinetics of BCO under SCCO2 at various pressures (15–40 MPa), flow rates (30, 40, and 50 g min−1) and temperatures (30, 40, and 50 °C), are shown in Fig. 1. Across all pressure conditions, the yield increased progressively with time and increased toward the end of the 60 min extraction period, indicating near-equilibrium solute recovery. A consistent enhancement in extraction yield was observed with increasing CO2 flow rate and temperature, which was attributed to improved solubility and faster mass transfer of solutes under elevated thermodynamic conditions. The extraction followed a biphasic kinetic profile with an initial rapid solubility-driven phase followed by a diffusion-limited phase.13
 |
| | Fig. 1 Extraction curve of SFE versus time and yield at (A) 15 MPa, (B) 20 MPa, (C) 25 MPa, (D) 30 MPa, (E) 35 MPa, and (F) 40 MPa at CO2 flow rates of 30, 40, 50 g min−1 and temperatures of 30, 40, and 50 °C. | |
3.3 Energy and solvent utilization efficiency of SFE
Fig. 2A shows the direct relationship between the CO2 flow rate and total solvent consumption. The influence of the solvent flow rate on extraction yield was evident at CO2 flow rates of 30, 40, and 50 g min−1, with the corresponding solvent-to-feed (S![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) F) ratios of 1.8
F) ratios of 1.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 2.4
1, 2.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 3
1, and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively. Higher CO2 flow rates (up to 50 g min−1, S
1, respectively. Higher CO2 flow rates (up to 50 g min−1, S![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) F 3
F 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) accelerated the extraction kinetics but reduced the extraction efficiency per unit CO2 due to the oversupply of solvent. The energy consumption in the SFE system is calculated based on the estimated power rating, as shown in Table 2. The energy consumption is associated with different pressures as shown in Fig. 2B, which are overlaid on a color-coded energy gradient. The increase in pressure reached >3.0 kWh at 40 MPa. Notably, the highest yield was achieved at 30 MPa with moderate energy input (∼2.75–2.85 kWh), indicating that this is the most energy-efficient condition. Beyond this pressure, increased energy did not result in increased extraction yields, reflecting a decrease in extraction efficiency. These values correspond to the energy efficiency, as shown in Table 3. These values represent estimated energy use at the laboratory scale, which is calculated from the equipment's power rating and extraction conditions, which helps in comparing different SFE settings under controlled conditions. However, it is important to note that the energy consumption estimates presented in this study are based on laboratory-scale equipment power ratings and do not reflect industrial-scale energy demand. The values serve as comparative indicators among experimental conditions rather than absolute energy requirements for commercial implementation. Future work should incorporate real-time energy monitoring systems, comprehensive life cycle assessment (LCA) modeling, and pilot-scale validation to accurately assess the industrial feasibility and environmental impact of SFE for black cardamom oleoresin extraction.
1) accelerated the extraction kinetics but reduced the extraction efficiency per unit CO2 due to the oversupply of solvent. The energy consumption in the SFE system is calculated based on the estimated power rating, as shown in Table 2. The energy consumption is associated with different pressures as shown in Fig. 2B, which are overlaid on a color-coded energy gradient. The increase in pressure reached >3.0 kWh at 40 MPa. Notably, the highest yield was achieved at 30 MPa with moderate energy input (∼2.75–2.85 kWh), indicating that this is the most energy-efficient condition. Beyond this pressure, increased energy did not result in increased extraction yields, reflecting a decrease in extraction efficiency. These values correspond to the energy efficiency, as shown in Table 3. These values represent estimated energy use at the laboratory scale, which is calculated from the equipment's power rating and extraction conditions, which helps in comparing different SFE settings under controlled conditions. However, it is important to note that the energy consumption estimates presented in this study are based on laboratory-scale equipment power ratings and do not reflect industrial-scale energy demand. The values serve as comparative indicators among experimental conditions rather than absolute energy requirements for commercial implementation. Future work should incorporate real-time energy monitoring systems, comprehensive life cycle assessment (LCA) modeling, and pilot-scale validation to accurately assess the industrial feasibility and environmental impact of SFE for black cardamom oleoresin extraction.
 |
| | Fig. 2 (A) Total consumption of CO2 (kg) and (B) energy consumption at different extraction pressures; the color gradient indicates the energy consumed. | |
3.4 Comparison with other extraction methods
The extraction efficiency of SFE was evaluated against other extraction methods to assess its relative performance in recovering black cardamom oleoresin. SFE achieved the highest oleoresin yield, significantly outperforming Soxhlet extraction (5.43% ± 0.18%), UASE (2.47% ± 0.09%), cold extraction (2.54% ± 0.11%), and hydrodistillation (2.07% ± 0.08%). The superior performance of SFE can be attributed to the unique mass-transfer properties of supercritical CO2, which exhibits gas-like diffusivity and liquid-like density, enabling superior penetration into the plant matrix compared to conventional solvents.12 The substantial difference between SFE and UASE aligns with Chemat et al. (2017),16 who reported 40–60% lower yields with ultrasound-assisted methods due to limited penetration depth and potential degradation under intense cavitation conditions. The low yield from cold extraction (2.54% ± 0.11%) reflects the fundamental limitations of room-temperature extraction, as noted by Kulal et al. (2025), who reported 50–70% lower yields due to reduced solubility at ambient temperatures and insufficient mass transfer. Hydrodistillation produced the lowest yield (2.07%), which is expected as this method is designed primarily for volatile oil extraction rather than comprehensive oleoresin recovery.12 SFE proved to be a superior technique for black cardamom oleoresin extraction, delivering higher yields, better retention of bioactive compounds, and improved product quality compared to other methods. This advantage arises from the tunable solvating power of supercritical CO2, which enhances the recovery of thermolabile constituents while minimizing degradation. In contrast, conventional techniques suffer from heat-induced losses and lower solvent efficiency. The variation in extraction yield is attributed to changes in solvent density, solubility, and mass transfer effects. However, it is important to recognize that traditional methods vary significantly in extraction time and solvent-to-feed ratios, making direct comparisons with SFE inherently unequal and potentially misleading. Hence, the comparisons are indicative of relative performance trends rather than exact equivalence under uniform conditions.
3.5 Analytical techniques
3.5.1 GC-MS and HS-GC analysis.
The evaluation of various extraction techniques, such as SFE, Soxhlet extraction, UASE, cold extraction, and hydrodistillation, revealed notable differences in the qualitative profiles of the extracted oleoresin as analyzed by GC-MS (Fig. 3 and Table 4). This study focused on major volatile components and highlighted the efficiency and selectivity of each method toward aroma-active compounds. Among them, SFE showed the highest efficiency in isolating volatiles such as 1,8-cineole (64.99% ± 2.13%), terpineol (16.06% ± 0.82%), and α-terpinyl acetate 5.61% ± 0.28%), demonstrating strong selectivity for monoterpenes and aroma-active compounds. These findings are consistent with those of Abdullah et al. (2021),21 who reported high concentrations of similar volatiles in cardamom oils. These bioactive compounds are known for their potent antioxidant, antimicrobial and anti-inflammatory properties. Soxhlet extraction, known for its deep solvent penetration, results in the recovery of a broad range of volatiles and non-volatile compounds, including 1,8-cineole and α-terpinyl acetate, confirming its effectiveness for accessing polar and semi-polar constituents. UASE selectively extracts lipophilic, non-volatile compounds like ethyl isoallocholate, sitosterol, and nerolidol, aided by ultrasound-induced cell disruption.15 Cold extraction preserved heat-sensitive volatiles, such as 1,8-cineole and α-terpinyl acetate, which are ideal for retaining oxygenated monoterpenes. Hydrodistillation yields lower levels of major volatile compounds due to thermal degradation, but it still enables the detection of other volatiles, indicating the presence of essential oils. However, the primary focus remains on the major volatile constituents. Similar trends were observed by Morsy (2015),22 who reported significant levels of α-terpinyl acetate and 1,8-cineole in hydrodistilled green cardamom oil. In summary, each method offers unique selectivity, guiding extraction choices on the basis of application needs in the food, pharmaceutical, and cosmetic industries.
 |
| | Fig. 3 Chromatogram of the GC-MS analysis of black cardamom oleoresin extracted using (A) SFE, (B) Soxhlet extraction, (C) UASE, (D) cold extraction, and (E) hydrodistillation. | |
Table 4 Compounds identified through GC-MS analysisa
| RT |
Compound |
RI* |
RI** |
SFE |
Soxhlet extraction |
UASE |
Cold extraction |
Hydro-distillation |
|
ND-not detected; NA-not available; RI-retention indices; RI*-experimental; RI**-literature; SFE-supercritical fluid extraction; UASE-ultrasound assisted solvent extraction. Values are expressed as means ± SD (n = 3 independent experiments). Different superscript letters within the same row indicate statistically significant differences (p < 0.05).
|
| 13.478 |
Bornyl acetate |
975 |
961 |
4.44 ± 0.21b |
6.96 ± 0.34a |
ND |
17.85 ± 0.82c |
17.11 ± 0.76c |
| 15.154 |
1,8-Cineole |
1031 |
1030 |
64.99 ± 2.13a |
27.33 ± 1.45b |
0.37 ± 0.02d |
10.30 ± 0.58c |
8.31 ± 0.44c |
| 17.753 |
Nerolidol |
1177 |
1175 |
ND |
2.07 ± 0.11b |
8.17 ± 0.41a |
ND |
8.93 ± 0.52a |
| 18.340 |
Terpinene |
1189 |
1190 |
4.82 ± 0.23c |
4.10 ± 0.19c |
0.06 ± 0.01d |
24.18 ± 1.12a |
25.34 ± 1.34a |
| 22.946 |
α-Terpinyl acetate |
1285 |
1287 |
5.61 ± 0.28c |
13.32 ± 0.67b |
0.83 ± 0.04d |
15.42 ± 0.73 ab |
7.40 ± 0.41c |
| 29.394 |
L-Terpineol |
1349 |
1367 |
16.06 ± 0.82a |
ND |
ND |
12.94 ± 0.65b |
18.02 ± 0.91a |
| 36.447 |
6-Epi-Shyobunol |
1495 |
1561 |
2.96 ± 0.14c |
12.38 ± 0.62a |
ND |
6.38 ± 0.31b |
ND |
| 39.447 |
Triethyl citrate |
1563 |
1589 |
1.11 ± 0.06c |
13.12 ± 0.68a |
ND |
8.91 ± 0.45b |
ND |
| 49.307 |
Cisvaccenic acid |
1591 |
1659 |
ND |
8.91 ± 0.43a |
ND |
ND |
ND |
| 49.389 |
Pregna |
1794 |
1792 |
ND |
11.81 ± 0.59a |
ND |
ND |
8.17 ± 0.42b |
| 41.860 |
Sabinene |
1968 |
2105 |
ND |
ND |
1.60 ± 0.08c |
4.03 ± 0.21b |
6.73 ± 0.35a |
| 64.932 |
Ethyl iso allocholate |
2103 |
NA |
ND |
ND |
73.73 ± 3.42a |
ND |
ND |
| 66.118 |
2,3-Dihydroxy propyl elaidate |
2187 |
NA |
ND |
ND |
3.98 ± 0.19a |
ND |
ND |
| 68.441 |
Sitosterol |
2245 |
3187 |
ND |
ND |
8.37 ± 0.41a |
ND |
ND |
| 69.303 |
3-Pinane-benzyl sulfone |
2312 |
NA |
ND |
ND |
2.90 ± 0.14a |
ND |
ND |
The residual ethanol content in the oleoresin extracted using SFE was 12.53 mg kg−1, as determined by headspace gas chromatography (HS-GC) analysis (Fig. S2). This value is well below the ICH Q3C guideline limit for ethanol as a Class 3 solvent (5000 mg kg−1), indicating efficient solvent removal during processing. In addition, the residual ethanol level complies with multiple regulatory standards, including the FDA limit for food-grade products (≤2%, equivalent to 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg kg−1) and Codex Alimentarius recommendations for spice extracts, which typically require residual solvent levels below 50 mg kg−1 for direct food use. Similar studies on Cinnamomum burmannii oleoresins have reported substantially higher ethanol residues, ranging from 2.27% to 11.98%, which were attributed to inefficient solvent evaporation or inadequate vacuum control during post-extraction processing.23 These findings highlight the importance of optimized post-extraction handling, particularly effective vacuum evaporation and temperature control, in minimizing residual solvent levels. Overall, the very low residual ethanol content achieved in this study confirms the food-grade quality and regulatory compliance of oleoresin, making it suitable for food, pharmaceutical, and nutraceutical applications without the need for additional solvent removal steps.
000 mg kg−1) and Codex Alimentarius recommendations for spice extracts, which typically require residual solvent levels below 50 mg kg−1 for direct food use. Similar studies on Cinnamomum burmannii oleoresins have reported substantially higher ethanol residues, ranging from 2.27% to 11.98%, which were attributed to inefficient solvent evaporation or inadequate vacuum control during post-extraction processing.23 These findings highlight the importance of optimized post-extraction handling, particularly effective vacuum evaporation and temperature control, in minimizing residual solvent levels. Overall, the very low residual ethanol content achieved in this study confirms the food-grade quality and regulatory compliance of oleoresin, making it suitable for food, pharmaceutical, and nutraceutical applications without the need for additional solvent removal steps.
3.5.2 GC-FID.
The GC-FID analysis of high-yielding black cardamom oleoresin extracted at 30 MPa, 50 g min−1 CO2 flow rate, and 50 °C revealed a diverse fatty acid profile, including SFAs, MUFAs, and PUFAs, contributing to its physicochemical and bioactive properties. The chromatograms are shown in Table S1 and Fig. 4. Oleic acid (C18:1n-9) was predominant (38.29 ± 1.35 g/100 g) and linoleic acid (C18:2n-6) (6.83 ± 0.28 g/100 g) contributed to a high unsaturated fatty acid content. The predominance of oleic acid and linoleic acids enhances the nutraceutical potential of oleoresin, contributing to cardiovascular benefits and anti-inflammatory effects. Additionally, the high MUFA content, particularly oleic acid, imparts oxidative stability, making the extract suitable for developing functional foods with an extended shelf-life. Among the SFAs, palmitic (4.03 ± 0.15 g/100g) and stearic (1.10 ± 0.06 g/100 g) acids were present in moderate amounts, supporting structural integrity and emulsification. The PUFA fraction is mainly composed of linoleic (6.83 ± 0.28 g/100 g) and alpha-linolenic (0.96 ± 0.05 g/100 g) acids, whereas smaller amounts of eicosatrienoic (0.73 ± 0.04g/100 g) and other acids were detected below the quantification levels. These essential fatty acids (EFAs) are known for their anti-inflammatory and cardioprotective benefits. The omega-6 to omega-3 ratio highlights the potential of oleoresin for functional foods. Trace trans fatty acids, including trans-9 elaidic acid (0.27 ± 0.02 g/100 g), indicate minimal lipid isomerization under controlled extraction conditions. Compared with ginger oleoresin,17 black cardamom oleoresin has a higher MUFA concentration, which supports lipid metabolism.
 |
| | Fig. 4 GC-FID chromatogram of the fatty acid composition of the SFE-extracted oleoresin at 30 MPa, 50 g min−1 CO2 flow rate, and 50 °C. | |
3.5.3 FTIR-ATR analysis of black cardamom oleoresin.
FTIR-ATR analysis of black cardamom oleoresin revealed key functional groups, reflecting its complex chemical composition (Fig. 5). The peaks at 2913 cm−1, 2843 cm−1, and 2999 cm−1 correspond to C–H stretching vibrations of alkanes, which are common in essential oils like terpenes and hydrocarbons. A similar peak at 2934 cm−1 in ginger oleoresin also indicates C–H bond stretching.13 The broadness of these peaks suggests the presence of hydroxyl (–OH) groups, indicating the presence of phenolic or alcohol compounds. The peaks at 2334 cm−1, 2099 cm−1, and 1991 cm−1 likely correspond to C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C or C
C or C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching vibrations, related to volatile compounds. A prominent peak at 1719 cm−1 represents C
N stretching vibrations, related to volatile compounds. A prominent peak at 1719 cm−1 represents C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations, indicating that carbonyl compounds, such as esters, ketones, or aldehydes, contribute to the aroma and antioxidant properties.11 The peaks at 1447 cm−1 and 1334 cm−1 reflect the C
O stretching vibrations, indicating that carbonyl compounds, such as esters, ketones, or aldehydes, contribute to the aroma and antioxidant properties.11 The peaks at 1447 cm−1 and 1334 cm−1 reflect the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching in aromatic rings, confirming the presence of flavonoids and terpenoids. The peak at 1015 cm−1 corresponds to C–O stretching, typical of ethers, esters, and alcohols, suggesting that oxygenated compounds like phenolic ethers, enhance flavor properties.24 Overall, FTIR confirmed the functional groups associated with the bioactive compounds, supporting the antioxidant and aromatic properties of black cardamom oleoresin and its potential for use in food applications.
C stretching in aromatic rings, confirming the presence of flavonoids and terpenoids. The peak at 1015 cm−1 corresponds to C–O stretching, typical of ethers, esters, and alcohols, suggesting that oxygenated compounds like phenolic ethers, enhance flavor properties.24 Overall, FTIR confirmed the functional groups associated with the bioactive compounds, supporting the antioxidant and aromatic properties of black cardamom oleoresin and its potential for use in food applications.
 |
| | Fig. 5 FTIR-ATR analysis of the SFE-extracted black cardamom oleoresin at 30 MPa. | |
3.6 Quality attributes of the SFE-extracted oleoresin
3.6.1 Effects of pressure on the yield, volatile and non-volatile contents, and color of black cardamom oleoresin.
The highest yield at each pressure was consistently achieved at 50 °C and 50 g min−1 CO2 flow rate, the conditions used for all subsequent quality analyses. The extraction pressure had a significant effect on the yield, total volatile (TV) and non-volatile (NV) fractions, and color of black cardamom oleoresin (p < 0.05), as shown in Table 5. The yield of oleoresin increased with pressure up to 30 MPa, reaching a maximum of 8.8%, indicating that higher pressures enhance extraction efficiency up to a certain threshold, likely due to increased solubility of oleoresin in SCCO2.25 The lowest yield was observed at 15 MPa (1.2%), indicating that lower pressure conditions were insufficient for efficient oleoresin extraction. The significant drop in yield above 30 MPa suggests possible degradation or excessive compaction of the matrix, reducing the diffusion of extractable compounds.
Table 5 Effects of pressure on oleoresin yield, total volatiles, non-volatiles, and color parameters during the SFE of black cardamoma
| Pressure (MPa) |
Yield (%) |
TV% |
NV% |
Color |
|
L* |
a* |
b* |
dE |
|
TV – total volatile content; NV – total non-volatile content; L* – lightness (0 = black and 100 = white); a* – red-green axis (positive = red and negative = green); b* – yellow-blue axis (positive = yellow and negative = blue); and dE – total color difference. The values are expressed as the means ± standard deviations (n = 3). Mean values with different superscript letters within the same column indicate statistically significant differences (p < 0.05).
|
| 15 |
2.50% |
28.03 ± 0.15a |
71.96 ± 0.15e |
56.16 ± 0.33a |
−1.31 ± 0.18 ab |
2.98 ± 0.05b |
37.80 ± 0.33e |
| 20 |
3.20% |
10.07 ± 0.11c |
89.93 ± 0.11c |
47.13 ± 0.63b |
−1.40 ± 0.37b |
2.17 ± 0.34b |
46.85 ± 0.63d |
| 25 |
3.50% |
5.90 ± 0.10d |
94.10 ± 0.10b |
40.75 ± 0.69d |
−1.13 ± 0.42 ab |
7.25 ± 0.90a |
53.35 ± 0.67b |
| 30 |
8.80% |
4.03 ± 0.57e |
95.97 ± 0.05a |
38.74 ± 0.43e |
−0.38 ± 0.38a |
1.42 ± 0.34b |
55.27 ± 0.44a |
| 35 |
4.20% |
6.10 ± 0.17d |
93.90 ± 0.17b |
39.02 ± 0.16e |
−0.91 ± 0.44 ab |
2.71 ± 1.15b |
54.96 ± 0.14a |
| 40 |
2.10% |
18.10 ± 0.36b |
81.90 ± 0.36d |
45.31 ± 0.49c |
−1.12 ± 0.24 ab |
1.84 ± 0.04b |
48.67 ± 0.49c |
3.6.2 Total volatile and non-volatile fractions.
The total volatile (TV) and non-volatile (NV) components exhibited contrasting trends. Prior to the TV measurement, the samples were subjected to rotary evaporation and vacuum drying to remove ethanol and moisture. The loss in mass of oleoresin upon drying is expressed as TV on a moisture-free basis. The highest TV content was recorded at 15 MPa (28.03% ± 0.15%), indicating that the content of essential oil was significantly greater than at the other pressures (p < 0.05). The NV content was 71.96% ± 0.15%, but it was relatively low compared with the other conditions. Moreover, with increasing SFE pressure, the extracted TV and NV contents are inversely proportional to each other. This suggests that lower pressure (15 MPa) promotes the extraction of volatile compounds, whereas high pressure (30 MPa) favors the extraction of non-volatile compounds. Additionally, excessively high pressure (>30 MPa) can suppress the desorption of volatile compounds from the matrix, limiting their extraction. This phenomenon aligns with the principle that SCCO2 effectively extracts heavier and less volatile compounds at higher pressures.25 Similarly, a study by Subtain et al. (2024)17 reported that the TV and NV in ginger oleoresin extracted at 30 MPa were 1.96% ± 0.11% and 98.04% ± 0.11%, respectively.
3.6.3 Color properties.
The color (L*, a*, b*) was significantly influenced by pressure (p < 0.05), reinforcing the importance of selecting appropriate extraction parameters to balance the yield, composition, and color stability. The lightness (L*) values ranged from 38.74 to 56.16, with the highest value at 15 MPa (56.16 ± 0.33), indicating a lighter-colored extract. At this lower pressure, mainly volatile compounds were extracted, while fewer pigments were recovered, resulting in a pale oleoresin. As pressure increased to 30 MPa, the L* value decreased to 38.74 ± 0.43, indicating a darker extract due to enhanced extraction of pigments, such as carotenoids, chlorophyll derivatives, and phenolic compounds. This shows that moderate pressures improve the extraction of colored, non-volatile compounds. The redness (a*) values remained relatively stable across the pressure range, suggesting minimal pressure-induced variation in red chromophores. Yellowness (b*) values were the highest at 15 MPa (−1.31 ± 0.18) and decreased at 30 MPa (1.42 ± 0.34), indicating a relative reduction in yellow carotenoid pigments at moderate pressures, likely due to differential solubility and competitive extraction of other pigment classes under supercritical conditions. The total color difference (dE) varied significantly with pressure (p < 0.05), with the lowest dE observed at 15 MPa (55.27 ± 0.44), reflecting minimal deviation in color attributes. At higher pressures (≥35 MPa), increased extraction of non-volatile resinous fractions and possible oxidative or pressure-induced degradation of labile chromophoric compounds may contribute to color stabilization or slight lightening, while also enhancing the recovery of volatile compounds at the expense of non-volatile retention and overall yield. Collectively, these findings indicate that 30 MPa represents an optimal extraction pressure for maximizing oleoresin yield and non-volatile content while maintaining a stable, green-yellowish color profile suitable for functional and industrial applications.13
3.7 Phytochemical analysis of SFE black cardamom oleoresin
3.7.1 Total phenolic content (TPC) and total flavonoid content (TFC).
As shown in Fig. 6A, TPC and TFC of black cardamom oleoresin extracted using SFE were significantly dependent on pressure, with both reaching their highest values at 30 MPa. The maximum TPC (90.61 ± 2.25 mg GAE per g) and TFC (124.96 ± 0.24 mg QE per g) were recorded at this pressure, whereas lower pressures (15–20 MPa) and excessive pressure (40 MPa) led to a decline in bioactive compound recovery. This trend aligns with previous studies by Abdullah et al. (2022),12 which demonstrated that increasing pressure enhances polyphenol and flavonoid solubility and mass transfer efficiency up to an optimal level, beyond which excessive matrix compression restricts solute diffusion. The decline at 40 MPa may be attributed to reduced CO2 diffusivity and decreased matrix permeability due to high-pressure-induced densification, a phenomenon observed in other SCCO2 extractions of cardamom and similar spices.10
 |
| | Fig. 6 (A) Total phenol and total flavonoid contents and (B) antioxidant assay of the SFE-extracted black cardamom oleoresin. | |
Flavonoids, which are more hydrophobic than polyphenols, exhibit improved solubility in SCCO2 at moderate pressures, allowing for enhanced extraction efficiency. However, at 40 MPa, the TFC significantly decreased to 92.31 ± 0.13 mg QE per g, likely due to solvent strength and density of CO2 increasing with pressure, potentially altering the selectivity toward different compound classes. This observation is consistent with findings from Subtain et al. (2024)17 on ginger oleoresin extracted at 30 MPa and 50 °C, where flavonoid retention was maximized under similar conditions. Statistical analysis confirmed significant differences (p < 0.05) in both TPC and TFC across pressure levels, further reinforcing 30 MPa as the optimal pressure for the extraction of polyphenol and flavonoid-rich oleoresin.
3.7.2 Antioxidant activity (DPPH, FRAP, and ABTS).
The antioxidant activity of black cardamom oleoresin, as assessed through DPPH, FRAP, and ABTS assays, significantly influenced the pressure (p < 0.05) as shown in Fig. 6B. Increasing the pressure from 15 MPa to 40 MPa increased all the measured bioactive compounds, with maximum values at 30 MPa, which was correlated with the highest TPC and TFC. The observed antioxidant activity correlates with elevated phenolic compounds and oxygenated monoterpenes such as 1,8-cineole, α-terpinyl acetate, and terpineol. These compounds are known for their free radical-scavenging potential, as the highest DPPH (88.63% ± 0.02%), FRAP (79.02 ± 0.24 µmol Fe2+ per g), and ABTS inhibition (63.28% ± 0.15%) observed at 30 MPa indicate a synergistic effect between polyphenols and volatile antioxidants. These results align with previous research on polyphenol-rich extracts, where higher phenolic and flavonoid contents led to improved antioxidant properties.26 Nagavekar and Singhal (2018)10 reported an increase in antioxidant activity when the pressure increased from 10 MPa to 30 MPa. However, at 40 MPa, all antioxidant activities decreased significantly, suggesting degradation or reduced solubility of bioactive compounds at high pressures, consistent with studies on SCCO2 extraction from black pepper and black cumin.10,12 The decline at 35–40 MPa may result from pressure-induced matrix densification, limiting antioxidant release.27 These findings support the optimization of SFE pressure to increase antioxidant activity and bioactive isolation.28 Overall, 30 MPa was confirmed to be optimal for extracting phenolics, flavonoids, and antioxidants from black cardamom oleoresin.
4 Conclusion
This study confirms that SFE is an efficient and sustainable method for obtaining high-quality black cardamom oleoresin. By modulating the CO2 pressure, optimal conditions were identified, resulting in the highest oleoresin output and enhanced retention of bioactive compounds. Among the other methods, SFE outperforms other multiconventional methods in preserving key aroma-active compounds and ensuring higher yields of non-volatile contents, total phenolics, flavonoids, and antioxidant activity. The optimized extracts retained a richer profile of volatile and bioactive compounds, including 1,8-cineole, α-terpinyl acetate, and terpineol. Additionally, SFE produced oleoresins with a favorable fatty acid profile and minimal solvent residues, reinforcing its eco-friendly and efficient nature. The oil recovery efficiency per kilogram of CO2 supports the sustainable and scalable nature of this process. While this study demonstrated the superior extraction performance and phytochemical profile achieved through SFE, it was limited to laboratory-scale observations. Industrial-scale implementation of SFE for black cardamom will require further studies on cost analysis, energy modeling, scalability of equipment, and techno-economic feasibility. These aspects are essential for validating the practical viability of SFE in commercial processing of spices.
Author contributions
SA: conceptualization, methodology, validation, formal analysis, investigation, data curation, writing – original draft, software; PG: conceptualization, supervision, resources, writing – review & editing; and KLRG: resources, review & editing. All the authors have reviewed and edited the manuscript.
Conflicts of interest
The author declares that they have no competing interests.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
All data supporting the findings of this study are available within the article and its supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5fb00781j.
Acknowledgements
The authors gratefully acknowledge the SRM Central Instrumentation Facility (SCIF), Nanotechnology Research Centre (NRC), Material Characterization Facility, and SRMIST for providing instrumentation facilities. We also extend our sincere thanks to the ICAR-Central Institute of Fisheries Technology for permitting the use of facilities for the supercritical fluid extraction. During the preparation of this work, the author(s) used QuillBot v23.9.4 to assist with paraphrasing and improving the clarity and readability of the manuscript. Following the use of this tool, the author(s) thoroughly reviewed, revised, and edited the content to ensure accuracy, originality, and compliance with academic standards. The author(s) take full responsibility for the content of the publication.
References
- E. Győri, A. Varga and I. Fábián, J. Supercrit. Fluids, 2019, 148, 16–23 CrossRef.
- L. T. Danh, N. D. A. Triet and J. Zhao, J. Supercrit. Fluids, 2012, 70, 27–34 CrossRef CAS.
- P. Mande and N. Sekar, J. Indian Chem. Soc., 2021, 98, 100201 CrossRef CAS.
- J. Wang, Y. Li and Q. Lu, Ind. Crops Prod., 2021, 168, 113580 CrossRef CAS.
- M. Sethunga, K. K. D. S. Ranaweera and K. D. P. P. Gunathilake, J. Food Bioprocess Eng., 2022, 5(2), 151–167 Search PubMed.
- P. P. Said, O. P. Arya and R. C. Pradhan, J. Food Process. Eng., 2015, 38(2), 107–114 CrossRef CAS.
- R. K. Gaikwad, I. H. Mondal and K. K. Dash, J. Agric. Food Res., 2024, 19, 101546 Search PubMed.
- S. D. Manjare and K. Dhingra, Mater. Sci. Energy Technol., 2019, 2, 463–484 Search PubMed.
- G. L. Teixeira, S. M. Ghazani and M. L. Corazza, J. Supercrit. Fluids, 2018, 133, 122–132 CrossRef CAS.
- N. Nagavekar and R. S. Singhal, J. Food Process. Eng., 2018, 41(1), e12670 CrossRef.
- N. Nagavekar and R. S. Singhal, Ind. Crops Prod., 2019, 134, 134–145 CrossRef CAS.
- A. N. Abdullah, W. Tian and S. Zengliu, Front. Nutr., 2022, 9, 1024820 CrossRef PubMed.
- E. Gil-Martín, T. Forbes-Hernández and A. Romero, Food Chem., 2022, 378, 131918 CrossRef PubMed.
- G. D. M. Madhusankha, L. F. Siow and Y. Y. Thoo, Food Biosci., 2023, 56, 103171 Search PubMed.
- R. Kulal, B. B. Borse and M. S. Peddha, J. Food Meas. Char., 2025, 18, 100219 CrossRef.
- F. Chemat, N. Rombaut and A. G. Sicaire, Ultrason. Sonochem., 2017, 34, 540–560 Search PubMed.
- S. Abishek and P. Gurumoorthi, Food Biophys., 2024, 19, 1095–1113 CrossRef.
- M. Subtain, I. Pasha and A. Rakha, J. Food Meas. Char., 2024, 18(6), 4611–4619 CrossRef.
- U. K. Vijayan, S. Varakumar and R. S. Singhal, LWT, 2019, 116, 108564 CrossRef CAS.
- A. C. F. Soldan, S. Arvelos and É. O. Watanabe, J. Clean. Prod., 2021, 297, 126593 Search PubMed.
- A. Abdullah, A. Algburi and A. Asghar, J. Food Sci. Technol., 2021, 58, 3183–3191 CrossRef CAS PubMed.
- N. F. Morsy, Ind. Crops Prod., 2015, 65, 287–292 CrossRef CAS.
- L. U. Khasanah, P. Prasetyawan and R. Utami, IOP Conf. Ser. Mater. Sci. Eng., 2017, 193(1), 012021 Search PubMed.
- Q. Zhang, Y. Chen and F. Geng, Molecules, 2023, 28(20), 7075 Search PubMed.
- M. J. H. Akanda, M. Z. I. Sarker and S. Ferdosh, Molecules, 2012, 17, 1764–1794 CrossRef CAS PubMed.
- A. Rauf, Z. Ahmad and D. Frmanowicz, Front. Chem., 2024, 12, 1463755 CrossRef CAS.
- H. A. Martinez-Correa, J. T. Paula and A. C. A. Kayano, J. Supercrit. Fluids, 2017, 119, 122–129 Search PubMed.
- F. Herzyk, D. Piłakowska-Pietras and M. Korzeniowska, Foods, 2024, 13, 1713 CrossRef CAS PubMed.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *a and
Kumar
Lekshmi R. G.
b
*a and
Kumar
Lekshmi R. G.
b
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). GC-MS profiling revealed superior retention of aroma-active volatiles (1,8-cineole, terpineol, and α-terpinyl acetate) under SFE, whereas GC-FID analysis revealed a nutritionally beneficial fatty acid profile dominated by oleic and linoleic acids. FTIR analysis confirmed the presence of esters, phenolics, and terpenoids. The extracts obtained under optimal conditions presented the highest total phenolic content (90.61 ± 2.25 mg GAE per g), total flavonoid content (124.96 ± 0.24 mg QE per g), and antioxidant activity (DPPH-88.63% ± 0.02%, ABTS-63.28% ± 0.15%, and FRAP-79.02 ± 0.24 µmol Fe2+ per g), significantly outperforming those obtained under all other conditions (p < 0.05). The residual ethanol concentration was within regulatory limits (<12.53 mg kg−1), confirming food-grade applicability. The total volatile and non-volatile contents, colorimetric attributes, and extraction efficiency were significantly influenced by pressure. This green extraction approach offers a sustainable method to increase the bioactive and sensory value of black cardamom oleoresin for food, nutraceutical, and cosmetic applications.
1). GC-MS profiling revealed superior retention of aroma-active volatiles (1,8-cineole, terpineol, and α-terpinyl acetate) under SFE, whereas GC-FID analysis revealed a nutritionally beneficial fatty acid profile dominated by oleic and linoleic acids. FTIR analysis confirmed the presence of esters, phenolics, and terpenoids. The extracts obtained under optimal conditions presented the highest total phenolic content (90.61 ± 2.25 mg GAE per g), total flavonoid content (124.96 ± 0.24 mg QE per g), and antioxidant activity (DPPH-88.63% ± 0.02%, ABTS-63.28% ± 0.15%, and FRAP-79.02 ± 0.24 µmol Fe2+ per g), significantly outperforming those obtained under all other conditions (p < 0.05). The residual ethanol concentration was within regulatory limits (<12.53 mg kg−1), confirming food-grade applicability. The total volatile and non-volatile contents, colorimetric attributes, and extraction efficiency were significantly influenced by pressure. This green extraction approach offers a sustainable method to increase the bioactive and sensory value of black cardamom oleoresin for food, nutraceutical, and cosmetic applications.

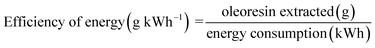
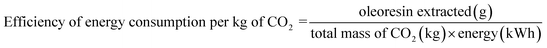
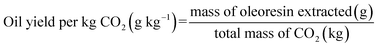
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15, w/v), heated at 0.8–1.0 kW to maintain a temperature of 95–100 °C for 3–4 h.4 Soxhlet extraction was carried out using 500 mL of ethanol (1
15, w/v), heated at 0.8–1.0 kW to maintain a temperature of 95–100 °C for 3–4 h.4 Soxhlet extraction was carried out using 500 mL of ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, w/v), with the heating mantle operated at 350–400 W to maintain the temperature at 60–80 °C for 6 h (12–14 cycles h−1).14 Cold extraction involved soaking 100 g of the sample in 200 mL of ethanol (1
5, w/v), with the heating mantle operated at 350–400 W to maintain the temperature at 60–80 °C for 6 h (12–14 cycles h−1).14 Cold extraction involved soaking 100 g of the sample in 200 mL of ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, w/v) at 25 °C for 1 h with mild magnetic stirring at 150 rpm, without external heating.15 For UASE, the sample was mixed with 300 mL of ethanol (1
2, w/v) at 25 °C for 1 h with mild magnetic stirring at 150 rpm, without external heating.15 For UASE, the sample was mixed with 300 mL of ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, w/v) and subjected to ultrasonic treatment at 250 W power and 40% amplitude, operating at 50 Hz for 20–30 min while maintaining the temperature between 30 and 50 °C.16 All extracts were subsequently filtered, and the solvents were removed using a rotary evaporator (RV 10 digital V, IKA, India) under reduced pressure at ≤40 °C. These conventional methods were not intended for direct comparison with SFE, as they differ significantly in extraction time, temperature, and solvent-feed ratios, and were included to provide a practical benchmark for relative extraction performance.
3, w/v) and subjected to ultrasonic treatment at 250 W power and 40% amplitude, operating at 50 Hz for 20–30 min while maintaining the temperature between 30 and 50 °C.16 All extracts were subsequently filtered, and the solvents were removed using a rotary evaporator (RV 10 digital V, IKA, India) under reduced pressure at ≤40 °C. These conventional methods were not intended for direct comparison with SFE, as they differ significantly in extraction time, temperature, and solvent-feed ratios, and were included to provide a practical benchmark for relative extraction performance.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 split ratio and 70 eV ionization energy. Compound identification was performed by matching mass spectra with the NIST and Wiley spectral libraries, using a minimum similarity index threshold of ≥80%. Retention indices (RI) were calculated using a homologous series of n-alkanes (C8–C30) analyzed under identical chromatographic conditions and were further confirmed by comparison with reported literature values from established databases.17
10 split ratio and 70 eV ionization energy. Compound identification was performed by matching mass spectra with the NIST and Wiley spectral libraries, using a minimum similarity index threshold of ≥80%. Retention indices (RI) were calculated using a homologous series of n-alkanes (C8–C30) analyzed under identical chromatographic conditions and were further confirmed by comparison with reported literature values from established databases.17


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) ratio. For the assay, 90 µL of the sample (1 mg mL−1) was mixed with 270 µL of methanol and 2.7 mL of FRAP reagent, followed by incubation at 37 °C for 30 min. The absorbance was measured at 595 nm, and the antioxidant capacity was quantified using a Trolox calibration curve quantified using a Trolox standard curve (Fig S3C), with values expressed as µM Fe+2 per g.
1 (v/v) ratio. For the assay, 90 µL of the sample (1 mg mL−1) was mixed with 270 µL of methanol and 2.7 mL of FRAP reagent, followed by incubation at 37 °C for 30 min. The absorbance was measured at 595 nm, and the antioxidant capacity was quantified using a Trolox calibration curve quantified using a Trolox standard curve (Fig S3C), with values expressed as µM Fe+2 per g.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) F) ratios of 1.8
F) ratios of 1.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 2.4
1, 2.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 3
1, and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively. Higher CO2 flow rates (up to 50 g min−1, S
1, respectively. Higher CO2 flow rates (up to 50 g min−1, S![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) F 3
F 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) accelerated the extraction kinetics but reduced the extraction efficiency per unit CO2 due to the oversupply of solvent. The energy consumption in the SFE system is calculated based on the estimated power rating, as shown in Table 2. The energy consumption is associated with different pressures as shown in Fig. 2B, which are overlaid on a color-coded energy gradient. The increase in pressure reached >3.0 kWh at 40 MPa. Notably, the highest yield was achieved at 30 MPa with moderate energy input (∼2.75–2.85 kWh), indicating that this is the most energy-efficient condition. Beyond this pressure, increased energy did not result in increased extraction yields, reflecting a decrease in extraction efficiency. These values correspond to the energy efficiency, as shown in Table 3. These values represent estimated energy use at the laboratory scale, which is calculated from the equipment's power rating and extraction conditions, which helps in comparing different SFE settings under controlled conditions. However, it is important to note that the energy consumption estimates presented in this study are based on laboratory-scale equipment power ratings and do not reflect industrial-scale energy demand. The values serve as comparative indicators among experimental conditions rather than absolute energy requirements for commercial implementation. Future work should incorporate real-time energy monitoring systems, comprehensive life cycle assessment (LCA) modeling, and pilot-scale validation to accurately assess the industrial feasibility and environmental impact of SFE for black cardamom oleoresin extraction.
1) accelerated the extraction kinetics but reduced the extraction efficiency per unit CO2 due to the oversupply of solvent. The energy consumption in the SFE system is calculated based on the estimated power rating, as shown in Table 2. The energy consumption is associated with different pressures as shown in Fig. 2B, which are overlaid on a color-coded energy gradient. The increase in pressure reached >3.0 kWh at 40 MPa. Notably, the highest yield was achieved at 30 MPa with moderate energy input (∼2.75–2.85 kWh), indicating that this is the most energy-efficient condition. Beyond this pressure, increased energy did not result in increased extraction yields, reflecting a decrease in extraction efficiency. These values correspond to the energy efficiency, as shown in Table 3. These values represent estimated energy use at the laboratory scale, which is calculated from the equipment's power rating and extraction conditions, which helps in comparing different SFE settings under controlled conditions. However, it is important to note that the energy consumption estimates presented in this study are based on laboratory-scale equipment power ratings and do not reflect industrial-scale energy demand. The values serve as comparative indicators among experimental conditions rather than absolute energy requirements for commercial implementation. Future work should incorporate real-time energy monitoring systems, comprehensive life cycle assessment (LCA) modeling, and pilot-scale validation to accurately assess the industrial feasibility and environmental impact of SFE for black cardamom oleoresin extraction.


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg kg−1) and Codex Alimentarius recommendations for spice extracts, which typically require residual solvent levels below 50 mg kg−1 for direct food use. Similar studies on Cinnamomum burmannii oleoresins have reported substantially higher ethanol residues, ranging from 2.27% to 11.98%, which were attributed to inefficient solvent evaporation or inadequate vacuum control during post-extraction processing.23 These findings highlight the importance of optimized post-extraction handling, particularly effective vacuum evaporation and temperature control, in minimizing residual solvent levels. Overall, the very low residual ethanol content achieved in this study confirms the food-grade quality and regulatory compliance of oleoresin, making it suitable for food, pharmaceutical, and nutraceutical applications without the need for additional solvent removal steps.
000 mg kg−1) and Codex Alimentarius recommendations for spice extracts, which typically require residual solvent levels below 50 mg kg−1 for direct food use. Similar studies on Cinnamomum burmannii oleoresins have reported substantially higher ethanol residues, ranging from 2.27% to 11.98%, which were attributed to inefficient solvent evaporation or inadequate vacuum control during post-extraction processing.23 These findings highlight the importance of optimized post-extraction handling, particularly effective vacuum evaporation and temperature control, in minimizing residual solvent levels. Overall, the very low residual ethanol content achieved in this study confirms the food-grade quality and regulatory compliance of oleoresin, making it suitable for food, pharmaceutical, and nutraceutical applications without the need for additional solvent removal steps.![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C or C
C or C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching vibrations, related to volatile compounds. A prominent peak at 1719 cm−1 represents C
N stretching vibrations, related to volatile compounds. A prominent peak at 1719 cm−1 represents C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations, indicating that carbonyl compounds, such as esters, ketones, or aldehydes, contribute to the aroma and antioxidant properties.11 The peaks at 1447 cm−1 and 1334 cm−1 reflect the C
O stretching vibrations, indicating that carbonyl compounds, such as esters, ketones, or aldehydes, contribute to the aroma and antioxidant properties.11 The peaks at 1447 cm−1 and 1334 cm−1 reflect the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching in aromatic rings, confirming the presence of flavonoids and terpenoids. The peak at 1015 cm−1 corresponds to C–O stretching, typical of ethers, esters, and alcohols, suggesting that oxygenated compounds like phenolic ethers, enhance flavor properties.24 Overall, FTIR confirmed the functional groups associated with the bioactive compounds, supporting the antioxidant and aromatic properties of black cardamom oleoresin and its potential for use in food applications.
C stretching in aromatic rings, confirming the presence of flavonoids and terpenoids. The peak at 1015 cm−1 corresponds to C–O stretching, typical of ethers, esters, and alcohols, suggesting that oxygenated compounds like phenolic ethers, enhance flavor properties.24 Overall, FTIR confirmed the functional groups associated with the bioactive compounds, supporting the antioxidant and aromatic properties of black cardamom oleoresin and its potential for use in food applications.




