Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Characterization of roll-pulps: a functional snack from co-products of apple juice production
R. Nunes da Silva *ab,
B. Maurício
*ab,
B. Maurício bc,
G. Rodrigues
bc,
G. Rodrigues c,
B. Antunes
c,
B. Antunes d,
J. M. Vieira
d,
J. M. Vieira ef,
I. R. Alvesg,
M. J. Alegriag,
R. M. Rodrigues
ef,
I. R. Alvesg,
M. J. Alegriag,
R. M. Rodrigues ef,
A. Fernandesb,
I. Fernandes
ef,
A. Fernandesb,
I. Fernandes ab,
A. Vicente
ab,
A. Vicente ef,
N. Mateus
ef,
N. Mateus ab and
V. de Freitas
ab and
V. de Freitas ab
ab
aUniversity of Porto, Faculty of Sciences, Rua do Campo Alegre 689, 4169-007 Porto, Portugal. E-mail: raq.silva@fc.up.pt
bLAQV-REQUIMTE, Chemistry and Biochemistry Department, Faculty of Sciences, University of Porto, Rua do Campo Alegre 689, 4169-007 Porto, Portugal
cFaculty of Engineering, University of Porto, s/n, R. Dr. Roberto Frias, 4200-465 Porto, Portugal
dNOVA School of Science and Technology, Largo da Torre, 2829-516 Caparica, Portugal
eCenter of Biological Engineering, University of Minho, Campus de Gualtar, 4710-057 Braga, Portugal
fLABBELS – Associate Laboratory, Braga/Guimarães, Portugal
gSUMOL COMPAL Marcas S.A, Rua Dr. António João Eusébio, N° 24, 2790-179, Carnaxide, Portugal
First published on 14th April 2026
Abstract
This study explores the development of innovative fruit-based snacks, specifically roll-pulps, derived from apple juice production co-products, aiming to promote healthier dietary habits and contributing to food waste reduction. Fruit intake remains below recommendations, highlighting the need for convenient, nutrient-rich products. Apple co-products were processed into roll-pulps, and their polyphenols and antioxidant stability were analyzed. Total polyphenol content of apple pulp measured was 5.94 mg g−1 fresh weight (FW), with 80% recovery post-processing into roll-pulps. HPLC analysis indicated that key polyphenols such as chlorogenic acid, epicatechin, phloretin, and phloretin xylosyl-glucoside, remained largely stable. Procyanidins content decreased after dehydrations whilst rutin showed an increase in concentration. The total polyphenol content of the pulp, as determined by HPLC, was 0.70 mg g−1 FW with 66% recovery after processing. DPPH and FRAP assays showed high antioxidant retention (84% and 90%), confirming bioactive stability at 70 °C. The incorporation of additives, including agave syrup and maltodextrin, modulated textural and mechanical properties, influencing hardness, chewiness, flexibility, and water activity. These results show the feasibility of converting apple juice co-products into sustainable, health-promoting snacks, supporting circular economy efforts to reduce food waste.
Sustainability spotlightApple pomace, a major by-product of apple juice production, is often underutilized and contributes to food waste, representing a missed opportunity for sustainable resource use. This study valorises apple pomace by developing and characterizing fruit roll-pulps enriched in polyphenols and antioxidants, demonstrating their stability and potential as functional, health-promoting snacks. By converting processing residues into value-added products, the research contributes to waste reduction, promotes circular economy strategies, and supports sustainable food production. These findings align with the UN Sustainable Development Goals 12 (Responsible Consumption and Production) and 2 (Zero Hunger), offering a practical approach to enhance food system efficiency while fostering nutritious and sustainable food options. |
1. Introduction
World apple production in 2024 reached 83.98 million metric tons according to the most recent data from the United States Department of Agriculture, placing fresh apples in the top 3 of global fruit production, just behind watermelon and bananas.1 The fresh apple market is estimated to be around 101.04 billion USD in 2024.2 In Europe, apples are the second-most popular fruit for consumption, with an average consumption of 15.4 kg per capita. In 2024, the European Union produced 10.21 million metric tons with Poland accounting for the majority of this contribution, followed by Italy and France.3 In Portugal, 343 thousand metric tons of apple were harvested, 319 thousand of those for fresh consumption and the other 24 for processing into various products.4 Whole fresh apples for consumption account for 70–75% of the total world production while 25 to 30% is processed into other products such as jams, juice, wine, dried products and others.5Apple-based juices and other food items are a staple food to many people worldwide; however, the co-products generated during their transformation are often overlooked. The general processing of apples that leads to juice production is comprised of a few steps such as apple preparation, milling or crushing, pressing, clarification, filtration, pasteurization, and eventual concentration.6 The most important co-product of apple processing is apple pomace, which represents approximately 25% of the fresh weight of apple that is left over from the processing of apples for juice production. Apple pulp, obtained from edible pure is another possible co-product.7
On a global scale, annually, it is estimated that approximately 4 million metric tons of apple pomace are generated as co-products of the industrial processing of apples.8 According to the United Nations Environment Program, 1.05 billion tons of food went to waste in 2022.9 Some is retrieved as animal feed, however this material is not suitable for such because of its extremely low protein content and high sugar levels.10 Consequently, this leaves a considerable portion of this undervalued resource to be often disposed inadequately in landfills and composting sites.11 With its high water, sugar and organic content, apple pomace may start to decompose and undergo fermentation, leading to pollution and, following the growth of microbial flora, decreasing soil nitrogen availability, thereby affecting its C/N ratio.12
Considering these environmental changes, apple pomace should be viewed as a valuable resource rather than mere waste, and efforts can be redirected towards sustainability and recycling practices that prioritize both environmental conservation and economic prosperity.13 In fact, outside the food industry, extensive research indicates the potential for valorization of apple pomace in the production of biofuels, bioethanol, biochar, biogas, biopolymers, and composites, as well as environmental applications, such as textile dye removal and absorption of heavy metals.12,14,15
Commonly, apple pomace consists mainly of leftover flesh and peel (95%), and, in a lesser amount, seeds (2–4%), stems (1%) and calyx.16 The approximate composition of dry apple pomace contains a diverse array of essential nutrients such as dietary fiber including insoluble carbohydrates (cellulose, hemicellulose and lignin), sugars, proteins, and vitamins, as well as bioactive substances such as phenolic compounds and prebiotic oligosaccharides.17 Moreover, studies have demonstrated that the bioactives present in this co-product can offer various health benefits, such as prevention of cardiovascular diseases, constipation and hypertension, lowering plasma and liver cholesterol and triglycerides, and contributing to the removal of certain harmful substances in the human body, due to its antioxidant properties.18,19
Additionally, the general chemical composition of apple pomace is approximately 46.8% carbon, 43.6% oxygen, 6.4% hydrogen, 0.6% nitrogen, and 0.3% sulphur, on a dry basis. Various studies have concluded that apple pomace can contain around 70 to 85% moisture, 7 to 44% cellulose, 4 to 24% hemicellulose, 15 to 23% lignin, 9 to 23% fiber, 3 to 7% protein, 9 to 20% pectin and 0.5 to 1.6% ash.20–22
Polyphenols are plant secondary metabolites, characterized by the presence of one or more aromatic rings and two or more hydroxyl groups. In plants, polyphenols play an essential role, serving as antioxidants and defense mechanisms against pathogens.23 In human nutrition, they have gained considerable attention due to their anti-inflammatory, anti-mutagenic and immunomodulatory properties, contributing significantly to the biological activity of apple pomace.18
However, the concentration and composition of polyphenolic compounds present in apples vary depending on factors such as the cultivar, stage of ripeness, the part of fruit, agricultural practices (such as pesticide use), growing conditions (including soil composition and climate), post-harvest handling, and processing methods, such as cooking or juicing.24
Several studies have detected varying quantities of (+)-catechin, (−)-epicatechin, rutin, o-coumaric acid, p-coumaric acid, quinic acid, gallic acid, caffeic acid, chlorogenic acid, procyanidins, phloridzin, 3-hydroxyphloridzin, phloretin-2-O-glucoside, phloretin-2′-xyloglucoside and quercetin glycosides such as quercetin-3-O-galactoside, quercetin-3-O-rhamnoside, quercetin-3-glucoside, quercetin-3-xyloside and quercetin-3-arabinoside.10,12,14,15 In apple purees, chlorogenic acid is the most abundant polyphenol with other polyphenols identified in high concentrations including (−)-epicatechin, procyanidin dimers, and some lower quantities of quercetin glycosides.16 Furthermore, different parts of the apple contribute differently to this profile, with higher concentrations of quercetin glycosides in the peel and phenolic acids in the flesh.23
As a result, apple pomace is a rich source of natural antioxidants, such as phloridzin, catechins, procyanidins, caffeic acid, phloretin glycosides, quercetin glycosides, chlorogenic acid and many other phenolic components that contain antioxidant capacities.18 Antioxidants inhibit or delay undesired oxidation reactions by neutralizing reactive oxygen species and free radicals, preventing oxidative stress related to various diseases such as high blood pressure, cancer and neurodegenerative disorders.24
A fruit roll-up, also known as fruit leather, is a type of snack, industrial or homemade, generally made with fruit puree. The process involves mashing fruit into a smooth mixture, often adding sweeteners, flavor enhancers and dyeing agents, spreading it thinly on a surface, and then drying it. Once dried, the resulting product is pliable and is cut and rolled into various shapes and sizes.25
Furthermore, fruit roll-ups are popular as a snack because of their practical, portable, relatively non-perishable attributes offering a fruit-based, sweet treat. At their simplest, fruit roll-ups made from pureed fruit with no added ingredients preserve much of the nutritional profile from the original fruit, making them a healthy snack option. However, many commercially produced fruit roll-ups, often marketed as a healthier alternative to candy, include added sugars, artificial color and flavor additives, and preservatives, which diminish their nutritional benefits.26
The healthy snacks market is a growing industry, with a market value of 86.01 billion US dollars in 2022.27 Various pomaces have been tested and introduced into various snack options to create an alternative healthy version of them, using agro-waste. A fruit roll-up is a type of fruit snack that is commonly based on fruit puree and juices. Nevertheless, fruit roll-ups made from juice production pomace or pulp have not yet been developed and are not available on the market.
Fruits like apple, pineapple, mango, grapes, and strawberries can be used to produce these roll-ups. Apple is the most used due to its high pectin content which provides the desired malleable texture.28 Fruit puree is usually the main ingredient for fruit leather production, as it is first poured and thinly spread on a flat surfaced tray.29 Next, these are dried/dehydrated at a particular temperature to achieve a final moisture content achieved is around 15–20%. Different drying methods can be applied, such as, hot air drying, solar drying, cabinet drying, freeze drying and microwave oven drying. After drying the roll-ups are pulled from the surface, rolled, and properly packed. Different drying methods and additives that can be added to the purees will result in variations in the quality of the produced roll-up. These snacks generally last a long time and do not require refrigeration.29
This study addresses the underutilization of apple juice processing co-products, particularly apple pulp, which represents a significant fraction of processed fruit mass and is often economically undervalued or inadequately managed. Although rich in polyphenols and dietary compounds, this material is rarely transformed into high-value food products due to technological challenges, especially the risk of bioactive degradation during thermal processing. The research therefore tackles the dual challenge of waste valorisation and preservation of thermolabile functional compounds.
The aim of this study is to upcycle apple co-product originated from the industrial processing of Golden Delicious apples by developing a novel, sustainable, and practical roll-up style snack. Additional ingredients, such as natural sweeteners, were included in the formulations to enhance appealing organoleptic properties: texture, flavor, and appearance. The study offers a practical framework for fruit processors to convert co-products into marketable, shelf-stable snacks, improving resource efficiency and economic return. The detailed evaluation of polyphenol stability, antioxidant retention, water activity, and texture provide industry-relevant data for formulation, process optimization, and product positioning within the growing functional snack sector. By integrating circular economy principles with technological validation, the research delivers both sustainability value and applied guidance for industrial implementation. To the best of our knowledge, while apple puree has been repurposed for this kind of snack, apple pomace has only been used in the formulation of cookies, bread, muffins, jams, and so on and apple pulp is even less used.30,31 Thus, this is an innovative way to upcycle this valuable resource.
In this study, the co-product used, more accurately described as pulp, consists of the remaining fraction of puree with peel remaining after juice extraction. Accordingly, these snacks have been named roll-pulps, highlighting their unique feature of being made entirely, or predominantly, from pulp derived from industrial apple juice production.
2. Materials and methods
2.1. Reagents
All reagents and solvents used were of analytical grade. Water was obtained from a Milli-Q® Direct Water Purification System. Sodium carbonate and reference standards including those of chlorogenic acid, (+)-catechin and quercetin-3-O-glucoside were obtained from Sigma-Aldrich (Sigma Aldrich, St. Louis, MO, USA). Acetonitrile (99.8%), formic acid (99%) and methanol (99.8%) were purchased from ChemLab (ChemLab, Zedelgem, Belgium). Folin–Ciocalteu reagent was purchased from LabChem (ACP Chemicals Inc., Zelienople, PA, USA). Agave syrup (OrigensBio, Portugal), carob syrup (SweetLife, Switzerland), molasses and pectin (Condi-Alimentar, Portugal) were purchased at a local supermarket.2.2. Processing and sampling
The apple (Golden Delicious) puree and apple pulp samples were obtained as co-products of juice production (Fig. 1).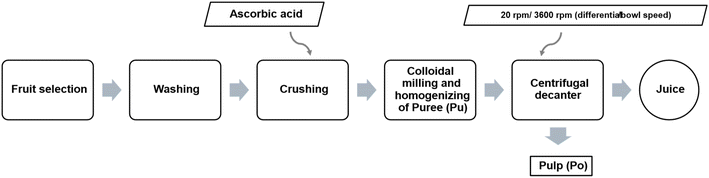 | ||
| Fig. 1 Partial flowchart illustrating the industrial production of apple juice, by SUMOL+COMPAL, S. A. Pulp (Po) is a co-product and puree (Pu) as an intermediary.32,33 | ||
Pulp (Po) and puree (Pu) were selected as the study aimed to focus specifically on co-products generated during juice production, which represent underutilized side streams with high valorization potential. This approach is aligned with the needs of our industrial partners, who generate significant amounts of these materials and are interested in exploring sustainable applications. Pulp was chosen as the main innovative matrix, as fruit roll-type products have traditionally been developed using juice and puree. Puree was included as an intermediary and comparative matrix, functioning as a reference condition to validate the feasibility of the formulation approach.
2.3. Unbounded phenolic compounds extraction from apple pulp
5 g of apple pulp was extracted with 50 mL of MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O
H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HCOOH (89
HCOOH (89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and ultraturraxed at 9500 min−1 for 10 min (T25-Ultra-turrax, IKA-Labortechnik, Staufen, Germany) while the samples remained in an ice bath. The mixture was centrifuged at 4 °C for 10 min at 13
1) and ultraturraxed at 9500 min−1 for 10 min (T25-Ultra-turrax, IKA-Labortechnik, Staufen, Germany) while the samples remained in an ice bath. The mixture was centrifuged at 4 °C for 10 min at 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm (Dynamica Velocity 14 Refrigerated Centrifuge, Dynamic Scientific Ltd; Livingston,UK). The supernatant was reserved, and the pellet was re-extracted using the same procedure. The resulting supernatants were combined and filtered through a vacuum filtration system with paper filter. This filtrated product was evaporated on a rotary evaporator under vacuum (Rotavapor R-114, Buchi, Flawil, Switzerland). The residue was resuspended in MeOH
000 rpm (Dynamica Velocity 14 Refrigerated Centrifuge, Dynamic Scientific Ltd; Livingston,UK). The supernatant was reserved, and the pellet was re-extracted using the same procedure. The resulting supernatants were combined and filtered through a vacuum filtration system with paper filter. This filtrated product was evaporated on a rotary evaporator under vacuum (Rotavapor R-114, Buchi, Flawil, Switzerland). The residue was resuspended in MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O Milli Q (50
H2O Milli Q (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) and refrigerated at −18 °C. Extraction was performed in triplicate.
50) and refrigerated at −18 °C. Extraction was performed in triplicate.
2.4. Optimized procedure of the production of the four prototypes under study
For optimizing the conditions of roll-pulp, a series of steps were carried out. For clarity, the detailed procedure is provided in the Supporting Information (Section 1).To produce optimized prototypes, five grams of apple pulp per roll-pulp were weighed, with an addition of 5% (in weight) of agave syrup and/or 5% of maltodextrin. The mixture was thinly spread out (around 1 mm thickness) on a baking sheet in a rectangular shape. These roll-pulps were then dried using a dehydrator (Clatronic Drier DR3525, Clatronic, Kempen, Deutschland) at a 70 °C setting until no longer wet to touch (Table 1).
| Designation | Main component | Additives |
|---|---|---|
| Po100D | 100% apple pulp | No additives |
| Po100DAg | Apple pulp | Agave (5%) |
| Po100DM | Apple pulp | Maltodextrin (5%) |
| Po100DAgM | Apple pulp | Agave (5%) and maltodextrin (5%) |
2.5. Unbounded phenolic compounds extraction from apple roll-pulp
The first step was the rehydration of the roll-pulp sample in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (pulp
1 (pulp![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water) to achieve an equivalent moisture content to the pulp. The extraction of unbounded phenolic compounds (polyphenols not bound to other compounds or cellular structures) was performed in triplicate, as described previously at 2.3.
water) to achieve an equivalent moisture content to the pulp. The extraction of unbounded phenolic compounds (polyphenols not bound to other compounds or cellular structures) was performed in triplicate, as described previously at 2.3.
2.6. Quantification of the total phenolic content in apple pulp and roll-pulp extracts by Folin–Ciocalteu colorimetric assay
3.75 µL of each extract was mixed with 18.75 µL of Folin–Ciocalteu phenol reagent and 125 µL of distilled water in each well. The mixture was shaken for 30 s, and 75 µL of 20% Na2CO3 and 152.5 µL of water were added for a final volume of 375 µL. The mixtures were shaken again and left at room temperature in the dark for 30 minutes. The mixtures were analyzed using absorbance at 750 nm in a plate reader (Biotek, Berthold Technologies, Bad Wildbad, Germany). A chlorogenic acid calibration curve was performed, with results expressed as equivalents of chlorogenic acid (CAE) per gram of fresh weight (FW).2.7. Quantification of phenolic content in apple pulp and roll-pulp extracts by HPLC-DAD-ESI-MS/MS
The phenolic content of each extract was analyzed through High performance liquid chromatography. A reverse phase C18 column (Agilent Poroshell 120 EC-C18, 250 × 4.6 mm, i.d.: 2.7 µm) was used at 25 °C. The solvents used were H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HCOOH (99.5
HCOOH (99.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, v/v) (A) and CH3CN
0.5, v/v) (A) and CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HCOOH (99.5
HCOOH (99.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, v/v) (B), at a flow rate of 0.4 mL min−1 (0–10 min: 88–83% A, 10– 13 min: 83–80% A, 13–50 min: 80–75% A, 50–55 min: 75–65% A, 55–61 min: 65–0% A, 61–70 min: 0% A, 70–73 min: 0–88% A, 73–85 min: 88% A). These solvents were previously filtered. Detection was made at 280 nm. Mass spectrometry analysis was performed using Teixeira et al.'s33 method.
0.5, v/v) (B), at a flow rate of 0.4 mL min−1 (0–10 min: 88–83% A, 10– 13 min: 83–80% A, 13–50 min: 80–75% A, 50–55 min: 75–65% A, 55–61 min: 65–0% A, 61–70 min: 0% A, 70–73 min: 0–88% A, 73–85 min: 88% A). These solvents were previously filtered. Detection was made at 280 nm. Mass spectrometry analysis was performed using Teixeira et al.'s33 method.
2.8. Evaluation of antioxidant capacity in apple pulp and roll-pulp extracts
The DPPH reagent was prepared at a concentration of 60 µM in methanol. 270 µL of this reagent was added to each well with 30 µL of previously prepared phenolic extract. For 20 minutes, the decrease in absorbance was measured at 515 nm, every 5 minutes at 25 °C. The calibration curve was done with a Trolox solution, and the results were expressed as µM Trolox equivalents per gram of fresh weight.34The FRAP reagent was prepared with a solution consisting of 10 vol of 300 mM acetate buffer, pH 3.6, 1 vol of 10 mM TPTZ in 40 mM HCl and 1 vol of 20 mM FeCl3·6H2O. This solution was then diluted to one-third with acetate buffer, and 270 µL were added to each well, with 30 µL of extract. The absorbance at 593 nm was measured at 0 and 4 minutes, at 37 °C. A Trolox calibration curve was used, and the results are expressed as µM Trolox equivalents per gram of fresh weight.35
2.9. Mechanical analysis
Texture profile analysis (TPA) tests were carried out on a TA HD Plus Texture Analyzer (Stable Micro Systems. UK) with an aluminum plate probe with a diameter of 70 mm. The actuation force was 0.05 N, and the compression rate was 80% at a speed of 1 mm s−1. Penetration tests were conducted using the same equipment with a 2 mm diameter needle probe. The probe penetrated 5 mm into the sample at a speed of 1 mm s−1.Then, the apple roll-ups were then unrolled and sliced into uniform strips (20 mm-wide, 60 mm-long), which were mounted between tensile grips. The initial grip separation and the crosshead speed were set at 60 mm and 1.0 mm s−1, respectively. The tensile strength (force/initial cross-sectional area) and the elongation at break were computed directly from the strength curves vs. elongation curves.
For both mechanical analyses, each roll-pulp sample was tested in triplicate at 21 °C. For data analysis, the Texture Exponent version 6.1.1.0 software by Stable Microsystems (Surrey, UK) was used.
2.10. Water activity
Water activity (aW) measurements were made using an Aqualab 3 analyzer (Decagon Devices, USA). 5 mL of the samples were placed in the sample container and aW was continually measured until reaching the equilibrium. The determinations were performed in triplicate at 25 °C.2.11. Statistical analysis
Values are expressed as arithmetic mean ± standard deviation. Experiments were performed in triplicate for each experimental condition to ensure the reproducibility of the results. The mean values and standard deviation were evaluated using one-way analysis (ANOVA) at Graph Pad Prism 9.3. Results and discussion
3.1. Apple roll-pulp production
According to the SI, regarding roll-pulp formulation testing, four prototypes were selected for further texture analysis: the pulp-only prototype (Po100D), pulp with agave (Po100DAg), pulp with maltodextrin (Po100DM), and pulp with both additives (Po100DAgM). For polyphenol analysis only Po100D was evaluated to decrease variability between samples. For detailed information on the formulations and processing steps used to obtain and characterize these prototypes, please refer to the SI (Fig. S1 SI at Section 1 and Tables S1–S3).3.2. Unbounded phenolic compounds stability
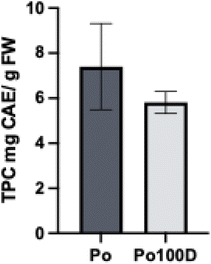
Various studies have investigated the most efficient methods for extracting polyphenols, comparing different solvent systems and conditions. Alcohol–water mixtures have shown higher polyphenol yields, with increased alcohol content improving the extraction process.36–38 Methanol, especially in 80% aqueous solutions, has consistently been identified as the most effective solvent due to its higher polarity and its ability to reduce polyphenol oxidation by inhibiting polyphenol oxidase activity.39–41 The addition of small amounts of acid, such as formic or hydrochloric acid, further enhances extraction efficiency by preventing oxidation, hydrolyzing cellulose and pectin to release bound polyphenols, and stabilizing the extracts.12,41,42 In this study, acidified methanol–water mixtures provided high recoveries of phenolic compounds when combined with an efficient extraction technique with an ultra-turrax.The Folin–Ciocalteu method was used to calculate the total phenolic content (TPC) of the pulp and roll-pulp samples. The roll-pulp TPC content was of 5.9 ± 0.5 mg CAE per g FW, with a slight loss of total polyphenol content being detected compared to the pulp content (7.4 ± 1.1 mg CAE per g FW), although not significantly different (p > 0.05) (Fig. 2). Recovery percentage was estimated as percentage of polyphenols retained after dehydration.
The reduction of phenolic content from pulp to roll-pulp, may be associated with multiple physical and chemical changes, as these polyphenols are considered heat-labile compounds,43 and various studies have detected decreases in polyphenol content of various fruit roll-ups.44,45 Increases in drying temperatures lead to higher degradation of polyphenols and as the temperatures applied to produce the roll-pulps were considerably low (70 °C), minimal polyphenol degradation was expected. Their thermal degradation depends on the pre-treatment, solvent type, pH and extraction method.46 The recovery of TPC in the roll-pulp was of 80%. Torres et al.,45 found that apple leather had a TPC recovery of around 70%, whilst Nizamlioglu et al.47 had a recovery over 99% in an oven dried apple roll-up. In other fruit leathers, TPC presented recovery percentages of 70 to 87%.44 In apple cubes that were dried using different methods, most of the samples had under 60% of its original phenolic content.48
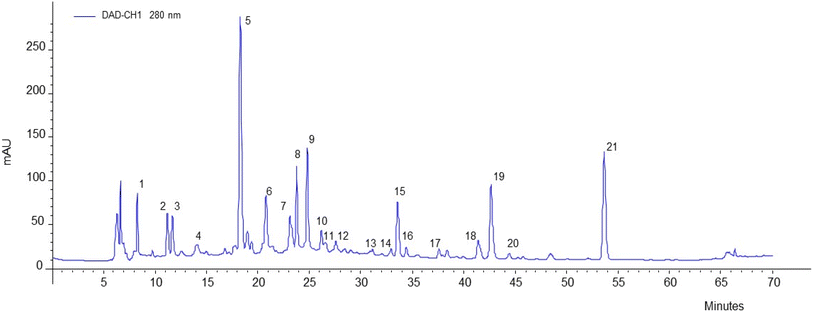
Considering that the optimized formulation was the one containing 100% apple pulp, a comparison of phenolic content and profile between this ingredient and the roll-pulp was made by HPLC-DAD and HPLC-ESI-MS analysis. Twenty-one compounds (20 polyphenols and quinic acid) were tentatively identified in both samples with tentative identification present in SI (Table A5). The chromatogram (Fig. 3) shows that the resolution (elution) of phenolic compounds agrees with the structural class with first elution of polar phenolic acids and epicatechins, followed by procyanidins, and lastly by quercetin glycosides.
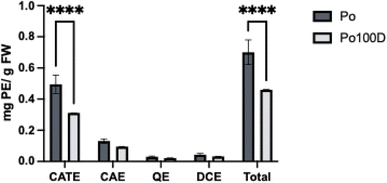
These 21 compounds were organized into four groups, where each compound's concentration within the same group was calculated using the respective calibration curve according to its maximum absorbance wavelength. The chlorogenic acid equivalents (CAE) group, with 3 polyphenolic compounds, was represented by a chlorogenic acid calibration curve at 326 nm, the catechin equivalents (CATE) group with 10 compounds, represent by a catechin calibration curve at 280 nm, the quercetin equivalents (QE) with 6 compounds represented by a quercetin-3-O-Glc calibration curve at 353 nm and the last group of dihydrochalcone equivalents (DCE), represented by a phloretin calibration curve at 285 nm. The roll-pulp polyphenol content is present in Fig. 4, in a direct comparison with its main ingredient, apple pulp. The concentration of each polyphenol in the pulp and roll-pulp is present in supplementary information (Table A6).
The total polyphenol concentration in the roll-pulp was 0.460 ± 0.002 mg E PP per g FW, a decrease of 0.24 mg E PP per g FW when compared to the pulp (0.70 ± 0.08 mg E PP per g FW), corresponding to a recovery of 66% in total polyphenols. Recovery of the quercetin equivalents was 71%, 73% for the chlorogenic acid equivalents, 63% for the catechins and 74% for dihydrochalcones. The differences between the total polyphenol content in roll-pulp and pulp are statistically significant.
The most abundant polyphenols in the roll-pulp were (−)-epicatechin (0.09 mg g−1 FW), chlorogenic acid (0.08 mg g−1 FW) and procyanidin dimer (0.04 mg g−1 FW), remaining the same as the predominant ones in the pulp. The major phenolics identified in apple pomace extracts in other studies were also chlorogenic acid, epicatechin, and quercetin derivatives, whilst catechins, procyanidin dimers and phlorizin were also found in high concentrations.49,50 Other studies have also concluded that among the phenolic compounds present in apple pulp, flavonols and phenolic acids are the most abundant groups, especially the derivatives of quercetin, and chlorogenic acid, respectively.51,52 Even though these studies pointed to high concentrations of quercetin derivates, the present study's lower concentration is due to the presence of quercetin derivatives in the bound fraction of polyphenols, which were not extracted in this work.10
Some flavonols such as rutin and various quercetin derivates, were a group of compounds whose concentration showed significant variations between them. These results confirmed a significantly higher concentration of the galactoside derivative, managing to present an increase of 2 to 5 times the concentration of other quercetin derivatives. Other studies also concluded the presence of this quercetin-3-O-galactoside as the most prominent quercetin derivative in apple.10,41 There can be a wide variety of results when comparing with values present in the literature, fitting into a large range of possible values. These discrepancies are explained by the many different factors that must be considered when analyzing a sample's polyphenolic content. The solvent used, sample–solvent ratio, extraction time and method have a significant impact on the measured concentration of polyphenols.41,42 Methanol–water mixtures are more commonly used, with 80% methanol being the preferred choice for extracting polyphenols in apple peel and flesh, as this higher methanol concentration results in a greater yield of polyphenols.53 When acidified methanol is used for extraction, which is the case in the present study, the content of monomeric polyphenols such as (+)-catechin, (−)-epicatechin, and quercetin increases due to hydrolysis of oligomeric and polymeric proanthocyanidins in an acidic environment.41 Since polyphenols are thermolabile and can therefore be degraded, the temperature in question needs to be considered, as well as pH, considering it could directly affect the structure of some polyphenolic compounds. Other factors such as apple variety, climatic and soil conditions, agrotechnical conditions, maturation degree, storage conditions, processing method and sample preparation will also bring variety to phenolic content.36 Salazar-Orbea's study evaluated how different processing techniques and conditions, influence the phenolic profile and they concluded that the intensity of the processing technique will lead to the mechanical disruption of fruit tissue which overall improves phenols extractability and therefore higher polyphenol content.54
As previously mentioned, polyphenols will undergo a degradation process once high temperatures are applied, so a slight decrease in polyphenol content was to be likely. Table 2 represents the recovery percentage of each compound in the roll-pulp, with values in red (over 100%) representing increases in concentration.
| Identification (peak #) | Recovery % | Identification (peak #) | Recovery % |
|---|---|---|---|
| Quinic acid (1) | 102.83 | Double charged procyanidin pentamer (12) | 54.73 |
| Hydroxytyrosol hexose (2) | 63.45 | Rutin (13) | 548.75 |
| nd (3) | 123.70 | Procyanidin dímer (14) | 29.15 |
| Procyanidin dímer (4) | 75.32 | Quercetin-3-O-galactoside (15) | 61.61 |
| Chlorogenic acid (5) | 73.39 | nd (16) | 77.85 |
| Procyanidin dímer (6) | 65.98 | Quercetin-3-O-pentoside (17) | 51.70 |
| Chlorogenic acid derivative (7) | 61.30 | Quercetin-3-O-pentoside (18) | 25.70 |
| (−)-Epicatechin (8) | 54.25 | Phloretin xylosyl-glucoside (19) | 67.35 |
| nd (9) | 73.30 | Quercetin-3-O-rhamnoside (20) | 330.45 |
| Procyanidin tetramer (10) | 55.40 | Phloretin (21) | 76.83 |
| Procyanidin trimer (11) | 38.28 |
Higher losses were identified for procyanidins and their derivatives, epicatechin and quercetin-3-O-pentoside. Some procyanidin derivatives showed as low as 29% recovery when compared to the concentration in pulp, with an average recovery of 53%. In contrast epicatechin showed 54% recovery and quercetin-3-O-pentoside an average of 39%. As flavonols and flavan-3-ols are more heat sensitive, the degradation of both quercetin glycosides and epicatechin is expected even at low heating temperatures, such as the 70 °C used for the roll-pulp production.46 In dehydrated apple slices, Suna et al.55 also reported significant losses of epicatechin, chlorogenic acid and quercetin glycosides after drying at 70 °C. In another apple chip production,56 although quercetin concentrations remained similar, or even showed slight increases after dehydration, the concentrations of epicatechin and procyanidins were significantly affected, having decreased to almost half of their original concentration in fresh apples, which are very similar findings to this roll-pulp production. Phenolic compounds expressed as catechin equivalents remained the predominant one, but also presented the highest degradation, as only 63% recovery was obtained.
It is important to note that a few compounds had a sharp increase in their concentration after dehydration, such as quinic acid, quercetin 3-O-rhamoside and rutin. Tan et al.,57 also revealed that the rutin concentration was significantly increased after drying in tomato samples, and in apple crisps production, Ertekin Filiz et al.,58 had detected no rutin in fresh apple samples, which was also the case in this apple pulp, but after drying at temperatures of 65–75 °C, rutin concentrations increased. Other studies have also revealed a general increase in polyphenol concentration after heat exposure, which may be related to the release of bound polyphenols from cells walls and other matrix components as well as possible hydrolysis of complex phenolics.43
The recovery of TPC was 66%, a value significantly lower than that obtained for the Folin method (80%). This finding could support the assumption that other non-polyphenol compounds are reducing the FC reagent, causing an overestimation of the TPC. The FC method is a non-specific spectrophotometric assay which can be affected by sugars, proteins, amino acids and other reducing agents (e.g. ascorbic acid) present in these matrixes.59,60 For instance, thermal degradation for most polysaccharides usually only starts at around 100 °C,61 therefore their presence will remain after roll-pulp production and still be detected by the FC reagent, leading to a smaller decrease in TPC when compared to the total polyphenol concentration detected in HPLC, which is based solely on the sum of the 21 detectable polyphenols.
3.3. Roll-pulp antioxidant stability
The DPPH assay is based on the scavenging of the DPPH radical by antioxidants,62 whilst the FRAP assay is an electron transfer-based method that measures the reduction of ferric ion (Fe3+) ligand complex to ferrous (Fe2+) complex by antioxidants in an acidic environment, pH of 3.6, to provide iron solubility and facilitate electron transfer.63 The results are presented in Fig. 5.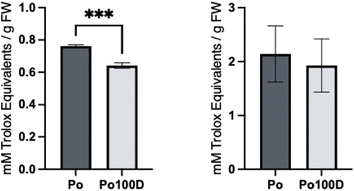 | ||
| Fig. 5 Antioxidant capacity measured by DPPH (left) and FRAP (right) assay with recovery %, in pulp (Po) and roll-pulp (Po100D). *** indicates significant differences (p < 0.001). | ||
Recovery percentage was estimated as percentage of antioxidant capacity retained after dehydration. There were significant differences (p < 0.05) between the antioxidant capacity of the pulp and roll-pulp with a recovery of 84% for DPPH and 90% for FRAP with the latter of which not presenting significant differences.
It is important to note that various studies showed contradictory findings, with some43 presenting decreases in this antioxidant capacity, others pointed to increases.47 These increases in antioxidant capacity can be caused by the heat processing, by enhancing antioxidant properties of naturally occurring compounds or by the formation of new compounds.64 These new compounds could be products from the Maillard reaction, such as melanoidins and reductones that are formed in the early stages of the reaction and exhibit strong antioxidant capacity, as well as other polyphenols hydrolysis products.45 Decreases in antioxidant capacity, such as the case observed for this study, are related to the degradation of polyphenols and other compounds with antioxidant properties.64 Considering the TPC of the roll-pulp samples decreased, this degradation of polyphenols is therefore affecting the overall antioxidant capacity of the product. Comparing some of the studies in which the antioxidant capacity suffered a decrease, the recovery percentages found were significantly smaller than those in the study with 58%45 for apple leather, and 40% for dehydrated apples.43
The high recovery found points towards a product with antioxidant stability. The processing method applied is being able to maintain these properties which are crucial for the roll-pulp to preserve its health benefits. If even higher recoveries were to be necessary, these could be maximized with the use of different drying methods, varying temperatures and time of cooking to avoid possible degradations.45
3.4. Texture profile analysis
Texture Profile Analysis (TPA) is a double compression test commonly applied for determining the textural properties of various food products, analyzing multiple textural parameters in a single experiment.65 The hardness (peak force obtained during the first compression), chewiness, gumminess, springiness (how well the roll-up will physically spring back after its deformation), resilience (how much a food product will “fight” to regain its original shape and size) and cohesiveness (how well the product can resist a second deformation in relation to its resistance under the first deformation)65 were all measured with values present below in Table 3.| Batch | Hardness (g) | Chewiness | Gumminess | Springiness | Cohesiveness | Resilience |
|---|---|---|---|---|---|---|
| Po100D | 4635.97 ± 1010.36a | 4233.05 ± 1094.66a | 4574.72 ± 1219.39a | 0.93 ± 0.04a | 0.98 ± 0.16a | 0.38 ± 0.06a |
| Po100DAg | 2899.05 ± 997.94b | 2551.61 ± 1315.10b | 2562.68 ± 1197.54b | 0.98 ± 0.07a | 0.89 ± 0.26a | 0.32 ± 0.05b |
| Po100DM | 2672.10 ± 777.55b | 2574.31 ± 1082.88b | 2744.81 ± 1184.32b | 0.94 ± 0.02a | 1.00 ± 0.22a | 0.31 ± 0.07b |
| Po100DAgM | 2155.27 ± 523.80b | 1809.70 ± 555.07b | 1943.61 ± 577.90b | 0.93 ± 0.02a | 0.90 ± 0.16a | 0.24 ± 0.03c |
There were significant differences (p < 0.05) between the control roll-pulp Po100D and all three other samples tested, regarding hardness, chewiness, and gumminess. A similar decrease in hardness was detected with the addition of agave syrup and maltodextrin. The addition of sugar syrups has been found to also lower the hardness force in fruit leathers.66 Considering maltodextrins, which are saccharide polymers of glucose units,67 their behavior in the pulp mixture could potentially mimic a common sugar, and therefore the maltodextrin addition also caused a decrease in hardness.
Whilst gumminess defines the energy required to disintegrate food for swallowing, chewiness defines the length of time needed to masticate it.68 The addition of agave syrup has lowered the chewiness and gumminess of the roll-ups, with Huang et al.66 also reporting a lower chewiness in pear roll-ups when adding sugar-based syrups. Maltodextrin is commonly added to provide the chewy desirable texture of fruit roll-ups;69 the fact that its addiction decreased chewiness and gumminess indicates a slight contradiction. However, dextrin has also been found to reduce chewiness in date-tamarind leathers.70
The fracturability and adhesiveness were also measured, however, high standard deviations made it difficult to draw a conclusion with significance. The respective values are present in supporting information (Table A7). Even if not significant, there was a decrease in fracturability with the addition of both agave and maltodextrin. Adhesiveness is an important parameter for fruit roll-ups since it is directly related to their stickiness.71 Even though not significant, a decrease in both agave and maltodextrin addition were detected, and according to literature, sugar addition usually increases adhesiveness. Sugars can attract and retain moisture and considering their glass transition temperature is lower than the drying temperature applied, this leads to an increase in stickiness of the product.71
There were no significant differences (p > 0.05) between samples for springiness and cohesiveness. About springiness, a decrease was expected for samples with agave and maltodextrin, as sugars are known to decrease this parameter in fruit leather.66 An average value for all roll-pulp samples was 0.945, indicating a high value of springiness, with other fruit leathers in literature presenting ranges around 0.700 to 0.900.66,72 Cohesiveness presented a high average of 0.947, indicating an extremely cohesive product, with some other studies found in literature, have produced roll-ups with cohesiveness values below 0.800,66 and some even averaging closer to 0.600.73 However, contrast to what was verified in this work, Valenzuela et al. have found that maltodextrin addition to apple roll-ups decrease cohesion levels, as well as springiness and resilience.74 Regarding resilience, significant differences were observed when no additives were added and when both were present. These results suggest that a combination of both the syrup and maltodextrin can contribute to a decrease in resilience of the roll-pulps.
After the TPA studies on the roll-pulps, the rolls were unrolled, and their sheets were analyzed in terms of tensile strength and elongation at break (Table 4).
| Batch | Tensile strength (MPa) | Elongation at break (%) |
|---|---|---|
| a Different letters show significant differences (p < 0.05). | ||
| Po100D | 1.67 ± 0.18a | 15.48 ± 2.06a |
| Po100DAg | 0.96 ± 0.13b | 17.41 ± 2.95a |
| Po100DM | 1.75 ± 0.23a | 16.85 ± 1.77a |
| Po100DAgM | 0.72 ± 0.10c | 13.43 ± 0.65b |
The only significant differences (p < 0.05) for elongation at break, were observed between the agave+maltodextrin (Po100DAgM) sample and the others. Elongation at break is the material's ability to stretch or deform before it breaks expressed as the percentage increase in the material's original length at the point of failure.75 The percentages obtained were quite low for all samples, consistently below 20%, with another study reporting around 19% elongation at break for their apple roll-up.76 The addition of agave and maltodextrin, with sugars being plasticizers that reduce intermolecular forces within the material's matrix,77 led to a small increase of the elongation at break percentage, making the product more stretchable. However, the combination of agave and maltodextrin decreased this percentage, as their interaction is reducing the overall flexibility of the matrix. For instance, maltodextrin could alter the dispersion or solubility of sugar, affecting the overall mechanical properties. Adding both agave and maltodextrin increases the total solids content in the roll-pulp Po100DAgM, which may lead to a denser and less flexible matrix, counteracting the individual plasticizing effects of sugar and maltodextrin.
Samples with agave (Po100DAg) and agave+maltodextrin (Po100DAgM), showed significant differences (p < 0.05) in tensile strength with the other two samples. Even if not statistically significant, there was a very slight increase in tensile strength with maltodextrin addition. However, higher sugar contents are also expected to increase tensile strength, as opposed to what was observed in these roll-pulps for agave addition (Po100DAg). It is possible that by adding sugar made with a syrup, and therefore containing water, it is decreasing the tensile strength of the product.
3.5. Water activity
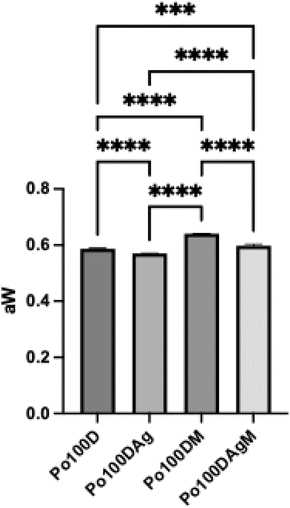
To assess the stability of the samples under different conditions, water activity (aw) was measured and compared, as can be seen in Fig. 6. Water activity is defined as the ratio of the water vapor pressure in the sample to the vapor pressure of pure water at the same temperature, reflecting the availability of water for chemical reactions and microbial growth.78All samples presented significant differences in aw (p < 0.05). A decrease of aw with the addition of sugars and maltodextrin was to be expected due to the previously mentioned hygroscopic nature of these compounds. However, this was only detected for agave syrup, with maltodextrin showing an increase in aw. The roll-pulps produced with maltodextrin could have potentially been less dried than the control ones, which could have led to this unforeseen rise in aw, as well as the possibility of a weaker interaction with water leading to more available water molecules. The aw values obtained for the roll-pulps, from 0.57 to 0.64, allow them to maintain microbial stability providing safe storage at room temperature, preventing growth of pathogenic microorganism and of non-pathogenic fungi and yeasts.79
Some inconsistent results were obtained for aw and mechanical properties (Section 3.2). These results could be influenced by the lack of homogenous spreading in replicas, which may lead to different drying rates, with some samples possibly presenting higher moisture content. The lack of a consistent rolling of the samples also contributed to these discrepancies as some roll-pulps had a more compact rolling, leading to higher values. Another possible factor for these results could be the drying rate of samples with additives. These samples may have required a longer drying time due to the added components, which could have introduced differences when compared to the control samples, whose drying times are better established based on prior experience with multiple batches. Nonetheless, since all samples were subjected to the same drying process, it is still possible to draw conclusions regarding their water retention capacity and the influence of each additive.
4. Conclusions
Within the food processing sector, increasing attention has been given to the valorization of agri-food by-products. In this context, the present study demonstrates that apple pulp, an underutilized co-product of juice processing, can be successfully transformed into value-added fruit roll snacks with promising functional properties. In this study, apple pulp was used as a raw material for the production of fruit roll snacks, and its physicochemical and functional properties were evaluated. Apple pulp contained a diverse profile of polyphenolic compounds with measurable antioxidant capacity.The produced roll-pulps showed good polyphenol retention following dehydration, which was carried out at relatively low temperatures. Most compounds maintained satisfactory recovery percentages, although procyanidins exhibited higher degradation levels. In contrast, rutin concentration increased, possibly due to the release of cell wall-bound forms during processing. Antioxidant capacity showed limited reduction after dehydration, as confirmed by two independent analytical assays.
Mechanical analysis revealed significant differences between formulations with and without agave syrup and maltodextrin. The addition of these ingredients significantly affected most texture-related parameters measured. Water activity values remained within ranges commonly associated with microbiological stability at room temperature. However, additional studies, including extended shelf-life assessments, are required to further evaluate product stability over time.
Overall, this work highlights the feasibility of upcycling apple processing by-products into nutritionally valuable, stable, and consumer-relevant snack products, contributing to more sustainable and circular food systems.
Conflicts of interest
There are no conflicts to declare.Data availability
The datasets generated and/or analysed during this study, including polyphenol and antioxidant profiles, extraction conditions, and texture analysis results, are included in the article and its SIsupplementary information (SI). Additional data supporting the findings of this study are available from the corresponding author upon reasonable request. Supplementary information is available. See DOI: https://doi.org/10.1039/d5fb00762c.Acknowledgements
This work received financial support from FCT/MCTES (UIDP/50006/2020 DOI 10.54499/UIDP/50006/2020) through national funds. Project Agenda VIIAFOOD – Plataforma de Valorizacao, Industrializacao e Inovacao comercial para o AgroAlimentar (no C644929456-00000040), funded by the PRR – Recovery and Resilience Plan and by the European NextGeneration EU Funds. This work received financial support from the PT national funds (FCT/MECI, Fundação para a Ciência e Tecnologia and Ministério da Educação, Ciência e Inovação) through the project UID/50006/2025 DOI 10.54499/UID/50006/2025 -Laboratório Associado para a Química Verde – Tecnologias e Processos Limpos. AF acknowledges FCT through research contract (https://doi.org/10.54499/CEECIND/00029/2018/CP1545/CT0010). IF acknowledges her assistant professor contract CEECINST/00064/2021.References
- USDA, Production – Apples, https://fas.usda.gov/data/production/commodity/0574000, (accessed 9 April 2024).
- Market Size of Apple Industry, https://www.marketgrowthreports.com/market-reports/apple-market-112927, (accessed 11 April 2024).
- W. G. Huis, EU Apple Production Per Country 2024, 2024 Search PubMed.
- European Comission, The apple market in the EU: Vol. 1: production, areas and yields, https://agriculture.ec.europa.eu/document/download/bd71aca7-dd45-4ab0-8dc1-f27321084916_en?filename=apples-production_en.pdf, (accessed 11 April 2024).
- F. Lyu, S. Luiz, D. Azeredo, S. Ajlouni and S. Ranadheera, Processes, 2020, 8, 319 CrossRef CAS.
- H. P. V. Rupasinghe and H. Thilakarathna, in Handbook of Functional Beverages and Human Health, ed. F. Shahidi and C. Alasalvar, 2016, pp. 93–106 Search PubMed.
- S. Vidović, A. Tepić Horecki, J. Vladić, Z. Šumić, A. Gavarić and A. Vakula, in Valorization of Fruit Processing By-Products, ed. F. P. B. Galanakis, Academic Press, 2020, pp. 17–42 Search PubMed.
- P. A. R. Fernandes, C. Le Bourvellec, C. M. G. C. Renard, F. M. Nunes, R. Bastos, E. Coelho, D. F. Wessel, M. A. Coimbra and S. M. Cardoso, Food Chem., 2019, 294, 9–18 CrossRef CAS PubMed.
- Food Waste Index Report 2024: Think Eat Save: Tracking Progress to Halve Global Food Waste, 2024 Search PubMed.
- W. Li, R. Yang, D. Ying, J. Yu, L. Sanguansri and M. A. Augustin, Ind. Crops Prod., 2020, 147, 112250 CrossRef CAS.
- S. Pathania and N. Kaur, Bioact. Carbohydr. Diet. Fibre, 2022, 27, 100295 CrossRef CAS.
- P. A. R. Fernandes, C. Le Bourvellec, C. M. G. C. Renard, F. M. Nunes, R. Bastos, E. Coelho, D. F. Wessel, M. A. Coimbra and S. M. Cardoso, Food Chem., 2019, 294, 9–18 CrossRef CAS PubMed.
- V. Sharma, M.-L. Tsai, P. Nargotra, C.-W. Chen, C.-H. Kuo, P.-P. Sun and C.-D. Dong, Catalysts, 2022, 12, 1373 CrossRef CAS.
- I. Mourtzinos and A. Goula, Natural Food Colorants, in NAtural Food Additives Ingredientes and Flavourings, Academic Press, 2019, pp. 23–44 Search PubMed.
- M. C. Soares, E. T. Ribeiro, E. M. Kuskoski, L. Gonzaga, A. Lima, J. Filho and R. Fett, Semina:Cienc. Agrar., 2008, 29, 339–348 CAS.
- M. Alongi, L. Manzocco, C. Comuzzi, A. Gorassini, G. Verardo, A. Rossi, M. Anese and M. C. Nicoli, Int. J. Food Sci. Technol., 2023, 58, 3189–3200 CrossRef CAS.
- A. Septembre-Malaterre, F. Remize and P. Poucheret, Food Res. Int., 2018, 104, 86–99 CrossRef CAS PubMed.
- F. Lyu, S. Luiz, D. Azeredo, S. Ajlouni and S. Ranadheera, Processes, 2020, 8, 319 CrossRef CAS.
- S. Vidović, A. Tepić Horecki, J. Vladić, Z. Šumić, A. Gavarić and A. Vakula, in Valorization of Fruit Processing By-Products, ed. F. P. B. Galanakis, Academic Press, 2020, pp. 17–42 Search PubMed.
- J. M. Costa, L. C. Ampese, H. D. D. Ziero, W. G. Sganzerla and T. Forster-Carneiro, J. Environ. Chem. Eng., 2022, 10, 108358 CrossRef CAS.
- S. Bhushan, K. Kalia, M. Sharma, B. Singh and P. Ahuja, Crit. Rev. Biotechnol., 2008, 28, 285–296 CrossRef CAS PubMed.
- N. O'Shea, A. Ktenioudaki, T. Smyth, P. Mcloughlin, L. Doran, M. Auty, E. Arendt and E. Gallagher, J. Food Eng., 2015, 153, 89–95 CrossRef.
- J. Kschonsek, T. Wolfram, A. Stöckl and V. Böhm, Antioxidants, 2018, 7, 20 CrossRef PubMed.
- J. Rumpf, R. Burger and M. Schulze, Int. J. Biol. Macromol., 2023, 233, 123470 CrossRef CAS PubMed.
- G. S. Dhillon, S. Kaur and S. K. Brar, Renewable Sustainable Energy Rev., 2013, 27, 789–805 CrossRef.
- L. M. Diamante, X. Bai and J. Busch, Int. J. Food Sci., 2014, 2014, 139890 Search PubMed.
- Healthy Snacks Industry Prospective, https://www.zionmarketresearch.com/report/healthy-snacks-market#:%7E:text=The%20global%20healthy%20snacks%20market, between 2023 and 2030, (accessed 8 April 2024).
- R. da Silva Simão, J. O. de Moraes, B. A. M. Carciofi and J. B. Laurindo, Food Eng. Rev., 2020, 12, 68–82 CrossRef.
- L. Silva and A. Miano Pastor, J. Food Process Eng., 2026, 49, e70311 CrossRef.
- P. Sobczak, R. Nadulski, Z. Kobus and K. Zawiślak, Sustainability, 2022, 14, 5470 CrossRef CAS.
- E. Gołębiewska, M. Kalinowska and G. Yildiz, Materials, 2022, 15, 1788 CrossRef PubMed.
- P. A. R. Fernandes, S. S. Ferreira, R. Bastos, I. Ferreira, M. T. Cruz, A. Pinto, E. Coelho, C. P. Passos, M. A. Coimbra, S. M. Cardoso and D. F. Wessel, Antioxidants, 2019, 8, 189 CrossRef CAS PubMed.
- J. Teixeira, A. Ribeiro, R. D. Simões, M. Alegria, N. Mateus, V. de Freitas, R. Perez and S. Soares, Plants, 2023, 12, 3397 CrossRef CAS PubMed.
- K. Mishra, H. Ojha and N. K. Chaudhury, Food Chem., 2012, 130, 1036–1043 CrossRef CAS.
- M. do S. M. Rufino, R. E. Alves, E. S. de Brito, J. Pérez-Jiménez, F. Saura-Calixto and J. Mancini-Filho, Food Chem., 2010, 121, 996–1002 CrossRef CAS.
- L. Pollini, L. Cossignani, C. Juan and J. Mañes, Molecules, 2021, 26, 4272 CrossRef CAS PubMed.
- V. Santarelli, L. Neri, G. Sacchetti, C. D. Di Mattia, D. Mastrocola and P. Pittia, Food Chem., 2020, 308, 125570 CrossRef CAS PubMed.
- G. Sacchetti, E. Cocci, G. Pinnavaia, D. Mastrocola and M. Dalla Rosa, Int. J. Food Sci. Technol., 2008, 43, 797–804 CrossRef CAS.
- A. Petkovska, V. Gjamovski and M. Stefova, Maced. J. Chem. Chem. Eng., 2016, 35, 29 CrossRef CAS.
- L. C. da Silva, M. C. Souza, B. R. Sumere, L. G. S. Silva, D. T. da Cunha, G. F. Barbero, R. M. N. Bezerra and M. A. Rostagno, Food Chem., 2020, 318, 126450 CrossRef CAS PubMed.
- L. Jakobek, M. Boc and A. R. Barron, Food Anal. Methods, 2015, 8, 2612–2625 CrossRef.
- L. A. Orozco-Flores, E. Salas, B. Rocha-Gutiérrez, M. D. R. Peralta-Pérez, G. González-Sánchez and L. Ballinas-Casarrubias, ACS Omega, 2024, 9, 196–203 CrossRef CAS PubMed.
- M. Lutz, J. Hernández and C. Henríquez, CyTA – Journal of Food, 2015, 13, 541–547 CAS.
- A. Concha-Meyer, V. D'Ignoti, B. Sáez, R. Diaz and C. Torres, J. Food Sci., 2016, 81, C569–C577 CrossRef CAS PubMed.
- C. A. Torres, L. A. Romero and R. I. Diaz, LWT–Food Sci. Technol., 2015, 62, 996–1003 CrossRef CAS.
- A. Antony and M. Farid, Appl. Sci., 2022, 12, 2107 CrossRef CAS.
- N. M. Nizamlioglu, S. Yasar and Y. Bulut, LWT–Food Sci. Technol., 2022, 153, 112420 CrossRef CAS.
- C. H. Chong, C. L. Law, A. Figiel, A. Wojdyło and M. Oziembłowski, Food Chem., 2013, 141, 3889–3896 CrossRef CAS PubMed.
- L. C. da Silva, J. Viganó, L. M. de Souza Mesquita, A. L. B. Dias, M. C. de Souza, V. L. Sanches, J. O. Chaves, R. S. Pizani, L. S. Contieri and M. A. Rostagno, Food Chem. X, 2021, 12, 100133 CrossRef CAS PubMed.
- S. Rana, A. Rana, A. Gulati and S. Bhushan, Food Anal. Methods, 2013, 7, 1424–1432 CrossRef.
- D. Gumul, R. Ziobro, J. Korus and M. Kruczek, Antioxidants, 2021, 10, 807 CrossRef CAS PubMed.
- A. Schieber, P. Hilt, P. Streker, H.-U. Endreß, C. Rentschler and R. Carle, Innovative Food Sci. Emerging Technol., 2003, 4, 99–107 CrossRef CAS.
- L. Jakobek, M. Boc and A. R. Barron, Food Anal. Methods, 2015, 8, 2612–2625 CrossRef.
- G. L. Salazar-Orbea, R. García-Villalba, M. J. Bernal, A. Hernández, F. A. Tomás-Barberán and L. M. Sánchez-Siles, Food Chem., 2023, 401, 134099 CrossRef CAS PubMed.
- S. Suna, C. Tamer, B. Incedayi, G. Ozcan Sinir and Ö. Çopur, Indian J. Tradit. Know., 2014, 13, 47–55 Search PubMed.
- T. Tarko, A. Duda-Chodak, P. Sroka, P. Satora and T. Tuszynki, J. Food Process. Preserv., 2010, 34, 728–742 CAS.
- S. Tan, Z. Ke, D. Chai, Y. Miao, K. Luo and W. Li, Food Chem., 2021, 338, 128062 CrossRef CAS PubMed.
- B. Ertekin Filiz and A. C. Seydim, J. Food Biochem., 2018, 42, e12676 CrossRef.
- M. Samara, A. Nasser and U. Mingelgrin, Am. J. Anal. Chem., 2022, 13, 476–493 CrossRef CAS.
- M. Pérez, I. Dominguez-López and R. M. Lamuela-Raventós, J. Agric. Food Chem., 2023, 71, 17543–17553 CrossRef PubMed.
- V. Soldi, Polysaccharides: Structural diversity and functional versatility, 2004, 395–409 Search PubMed.
- S. Škrovánková, L. Mišurcová and L. Machů, in Advances in Food and Nutrition Research, ed. N. R. Henry, Academic Press, 2012, vol. 67, pp. 75–139 Search PubMed.
- Y. Zhong and F. Shahidi, in Handbook of Antioxidants for Food Preservation, ed. F. P. Shahidi, Woodhead Publishing, 2015, pp. 287–333 Search PubMed.
- A. Vega-Gálvez, K. Ah-Hen, M. Chacana, J. Vergara, J. Martínez-Monzó, P. García-Segovia, R. Lemus-Mondaca and K. Di Scala, Food Chem., 2012, 132, 51–59 CrossRef PubMed.
- M. Johnson, Overview of Texture Profile Analysis, https://texturetechnologies.com/resources/texture-profile-analysis, (accessed 5 December 2024).
- X. Huang and F. Hsieh, J. Food Sci., 2006, 70, E177–E186 Search PubMed.
- P. Dokic, J. Jakovljevic and Lj. Dokic-Baucal, Colloids Surf., A, 1998, 141, 435–440 CrossRef CAS.
- L. S. Young, in Woodhead Publishing Series in Food Science, Technology and Nutrition, ed. E. Cauvain, Woodhead Publishing, 2012, pp. 562–579 Search PubMed.
- J. N. BeMiller, Carbohydrates, ed. E. Caballero, Academic Press, Oxford, 2003, pp. 1773–1775 Search PubMed.
- K. Al-Hinai, N. Guizani, V. Singh, M. Rahman and L. Alsubhi, Food Sci. Technol. Res., 2013, 49, 531–538 CrossRef.
- T. Mendes da Silva, P. Cristiana, G. Giacalone and N. Giuggioli, Ital. J. Food Sci., 2019, 31, 743–746 Search PubMed.
- Y. Bed
![[i with combining dot above]](https://www.rsc.org/images/entities/char_0069_0307.gif) r and M. Karaoglu, Int. J. Gastron. Food Sci., 2022, 30, 100613 CrossRef.
r and M. Karaoglu, Int. J. Gastron. Food Sci., 2022, 30, 100613 CrossRef. - M. Momchilova, G. Zsivanovits, Il. Milkova-Tomova, D. Buhalova and P. Dojkova, Bulg. Chem. Commun., 2016, 48, 428–434 Search PubMed.
- C. Valenzuela and J. Aguilera, J. Food Eng., 2015, 149, 51–60 CrossRef.
- K. de Naoum, Elongation at Break: What It Is and How It Works, https://www.xometry.com/resources/materials/elongation-at-break/ (accessed 5 December 2024).
- C. Valenzuela and J. Aguilera, Drying Technol., 2013, 31, 1951–1959 CrossRef CAS.
- P. Veiga-Santos, L. M. Oliveira, M. P. Cereda and A. R. P. Scamparini, Food Chem., 2007, 103, 255–262 CrossRef CAS.
- M. Mathlouthi, Food Control, 2001, 12, 409–417 CrossRef.
- N. A. Quintero Ruiz, S. M. Demarchi, J. F. Massolo, L. M. Rodoni and S. A. Giner, LWT–Food Sci. Technol., 2012, 47, 485–492 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |