Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Sustainable valorization of peach palm (Bactris gasipaes Kunth) peel flour to improve nutritional properties and carotenoid bioaccessibility in gluten-free cookies
Jhonathan Vinícius Menezes Silva *a,
Allien Monique Rosa Machado
*a,
Allien Monique Rosa Machado ab,
Andressa Mara Baseggio
ab,
Andressa Mara Baseggio ad,
Renan Campos Chisté
ad,
Renan Campos Chisté c and
Lilian Regina Barros Mariutti
c and
Lilian Regina Barros Mariutti a
a
aDepartamento de Ciência de Alimentos e Nutrição, Faculdade de Engenharia de Alimentos (FEA), Universidade Estadual de Campinas (UNICAMP), Campinas, São Paulo 13083-862, Brazil. E-mail: jhonathanvinicius27@gmail.com
bFood and Nutrition Graduate Program, Federal University of State of Rio de Janeiro (UNIRIO) – Av. Pasteur 296, Urca, Rio de Janeiro – Rio de Janeiro, 22290-250, Brazil
cFaculty of Pharmacy, Universidade Federal de Minas Gerais (UFMG), Belo Horizonte, Minas Gerais 31270-901, Brazil
dFaculdade de Ciências Aplicadas, Universidade Estadual de Campinas (UNICAMP), Limeira, SP 13484-350, Brazil
First published on 9th February 2026
Abstract
The growing demand for gluten-free bakery goods has increased interest in ingredients that improve physicochemical properties and nutritional value. Agro-industrial by-products are promising due to their health-related composition. Peach palm peel, a highly discarded Amazonian by-product, is rich in carotenoids; however, its application in food products remains limited. This study formulated gluten-free cookies by replacing rice flour with peach palm peel flour (PF) at 10 and 20%, and evaluated their physical, nutritional, and carotenoid composition, as well as in vitro bioaccessibility. Substitution with 20% PF increased dietary fiber, fat, and protein contents, and resulted in cookies with larger diameter and greater hardness. Scanning electron microscopy analysis revealed denser matrices in PF-added formulations. Seventeen carotenoids were quantified in PF and cookies, with all-E-β-carotene and all-E-lutein being predominant. Carotenoid bioaccessibility remained similar between 10% and 20% PF, and its modulation was influenced by lipid and dietary fiber contents, as well as by changes in the carbohydrate-rich food matrix resulting from PF substitution. Overall, PF incorporation enhanced the nutritional quality and modified relevant physical characteristics of gluten-free cookies while delivering substantial amounts of bioaccessible provitamin A carotenoids. These findings highlight PF as a functional and sustainable ingredient and an alternative strategy to valorize Amazonian fruit by-products.
Sustainability statementThis research contributes to the sustainable valorization of peach palm (Bactris gasipaes Kunth) peel, an abundant agroindustrial by-product from the Amazon region, by incorporating it into gluten-free cookies as a source of fiber and provitamin A carotenoids. The approach promotes circular economy principles by transforming food processing residues into value-added ingredients, reducing waste generation, and encouraging the efficient use of natural resources. Furthermore, it strengthens local bioeconomies by providing new opportunities for regional supply chains linked to Amazonian biodiversity. The work directly supports the United Nations Sustainable Development Goals (SDGs), particularly SDG 2 (Zero Hunger) by improving the nutritional quality of foods, SDG 9 (Industry, Innovation and Infrastructure) by fostering sustainable food innovations, SDG 12 (Responsible Consumption and Production) through waste reduction and resource efficiency, and SDG 13 (Climate Action) by enhancing sustainable production practices and minimizing environmental impacts. |
1 Introduction
Cookies are baked products, usually served as treats, made with sugar, shortening (i.e., butter), and flour, shaped into small and crispy products that are highly appreciated by children and adults.1 Traditionally, they are made with wheat flour, which naturally contains gluten, a viscoelastic protein complex composed mainly of gliadins and glutenins that confers structure, elasticity, and desirable rheological properties to dough systems. According to the Codex Alimentarius, gluten refers specifically to the proteins in wheat and related cereals that are insoluble in water and play a central role in dough development.2 However, the increasing prevalence of celiac disease and gluten sensitivity worldwide has intensified the demand for safe dietary alternatives, leading to a growing need for gluten-free bakery products that combine adequate technological quality with improved nutritional value.3–5Rice flour is the most commonly used flour in the development of gluten-free products. However, its low protein and fiber contents may impair some physicochemical properties of the final products, thereby reducing their acceptability. To address technological limitations and improve nutritional value, blends of rice flour with other gluten-free flours have been investigated for the development of new formulations.6,7 Among the alternatives, flours obtained from pulses (such as pea and chickpea), cereals (corn), and pseudocereals (buckwheat and quinoa) have been evaluated.8–10 In addition, the use of flours derived from agro-industrial by-products has emerged as a trend in the formulation of gluten-free products, representing sustainable and health promoting ingredients capable of modifying diverse characteristics of the final product.11,12
Peach palm (Bactris gasipaes Kunth.) is an Amazonian fruit with nutritional value and color, with shades varying from green, red, orange, white, and yellow.13,14 Peach palm peels are the main edible by-product generated during fruit processing and represent a rich source of lipids and dietary fiber, making them suitable for new applications as a functional ingredient.11,15,16 In addition, this by-product is rich in carotenoids with characteristic colors, with orange hues mainly attributed to β-carotene (provitamin A) and yellow hues to lutein, which is associated with eye health.16,17 Taken together, the use of peach palm peel flour can improve the functional properties of products, enriching them with bioactive compounds that can consequently contribute to increasing the bioaccessibility and bioavailability of carotenoids.
On the other hand, the food matrix plays a fundamental role in the bioaccessibility of bioactive compounds. Bioaccessibility refers to the fraction of food components effectively digested by enzymes and become accessible for absorption.18 Lipid-rich foods may improve the efficiency of carotenoid bioaccessibility and uptake due to increased micelle formation. Moreover, this property depends on the food matrix, carotenoid type and source, dietary fiber and the fatty acid profile of the food.19
Several studies indicate that peach palm peels are an excellent source of carotenoids.15,20 However, a knowledge gap remains regarding their potential application in food matrices for functional purposes, such as improving carotenoid bioaccessibility. Therefore, this study aimed to develop carotenoid rich gluten-free cookies formulated with different concentrations of yellow peach palm peel flour (PF). Specifically, we sought to characterize the chemical composition and carotenoid profile of the formulations, assess their physicochemical and textural properties, and determine the in vitro bioaccessibility of carotenoids to evaluate their functional potential.
2 Materials and methods
2.1 Raw materials
Peach palm peels were purchased at a local fair in Castanhal, Pará, Brazil (latitude: 1°17′50″ S; longitude: 47°55′20″ W), and access to this species was registered in the Brazilian National System for the Management of Genetic Heritage and Associated Traditional Knowledge (SisGen) under the code AA233C1. Whole fruits were sanitized in a sodium hypochlorite solution (100 mg L−1 of free chlorine) for 15 min, followed by rinsing with potable water, and then cooked in boiling water for 30 min. The pulp was manually separated from the peel, and the peels were dried in a forced-air circulation oven (Marconi MA035/5-PB) at 60 °C for 48 h. The dried peach palm peels were ground using a domestic food processor until a visually homogeneous flour was obtained; the flour was then vacuum-packed and stored at 5 °C in the dark until cookie preparation. Rice flour, powdered milk, sugar, salt, butter, baking powder, eggs, and whole milk were purchased from local markets in Campinas, SP, Brazil.2.2 Preparation of gluten-free cookies
Cookies were prepared according to the formulations described in Table 1, adapted from an original procedure described by Licona-Aguilar et al.21 To produce the cookie dough, all the dry ingredients (rice flour, peach palm peel flour, powdered milk, sugar, salt, and baking powder) were homogenized in a plastic bowl. Subsequently, butter, eggs, and whole milk were added and manually mixed with the dry ingredients using a silicone spatula. Portions of 30 g were weighed, and the cookies were shaped into a round form. The cookies were then baked in an electric oven (Rational, São Paulo, SP) at 180 °C for 15 min. The formulations were produced in duplicate (n = 2).| Ingredients | Formulations | ||
|---|---|---|---|
| Control | 10% | 20% | |
| Rice flour (g) | 200 | 180 | 160 |
| PF (g) | — | 20 | 40 |
| Powder milk (g) | 35 | 35 | 35 |
| Sugar (g) | 6 | 6 | 6 |
| Salt (g) | 3 | 3 | 3 |
| Butter (g) | 50 | 50 | 50 |
| Baking powder (g) | 1.5 | 1.5 | 1.5 |
| Egg white (g) | 45 | 45 | 45 |
| Whole milk (mL) | 88 | 88 | 88 |
2.3 Proximate composition
Moisture and ash contents were determined according to the AOAC procedure.22 The lipid content was determined by the Bligh and Dyer method. Protein content was determined by the Kjeldahl method, by converting the total nitrogen content using a conversion factor of 6.25. Total dietary fiber was determined using the enzymatic method,23 and carbohydrate content was calculated by difference. All the results were expressed as g/100 g, on a dry basis (d.b). All analyses were performed in triplicate (n = 3).2.4 Water activity
The water activity (aw) was measured using an Aqualab Series 3 TE (Decagon Devices®, Pullman Inc., WA 99163) at 25 °C. Five replicates were performed for each sample (n = 5).2.5 Color
The color of the three cookie formulations was measured according to the method previously reported by Ribeiro et al.,24 using diffuse illumination/0° viewing geometry (specular component included) and D65 as the light source. Color coordinates were obtained in the CIELAB system, where L* (lightness, L* = 0 is black and L* = 100 is white) and the chromatic coordinates a* (−a* = green and +a* = red) and b* (−b* = blue and +b* = yellow) were used to calculate chroma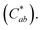 The total color difference was calculated employing the following equation:
The total color difference was calculated employing the following equation:
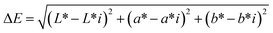 | (1) |
2.6 Texture analysis
The textural properties of the cookies were determined using a TA-XT2 texture analyzer (Stable Micro Systems, Godalming, UK) equipped with an HDP/BS probe for hardness and a Crisp Fracture Rig (HDP/CFS) probe for fracturability. Test parameters were defined as follows: a pre-test speed of 2.0 mm s−1, test speed of 2.0 mm s−1, and post-test speed of 10 mm s−1. Texture Expert software was used for data collection and processing, and measurements were conducted using three cookies per formulation (n = 3).2.7 Scanning electron microscopy (SEM)
The structure of the cookies was analyzed by scanning electron microscopy (SEM) using a LEO Electron Microscopy system (Oxford Instruments). Both surface and cross-section microstructures were evaluated. For cross-section analysis, the samples were previously fractured using liquid nitrogen, mounted on aluminum stubs with double-sided carbon adhesive tape, aided by nickel carbon paste, and sputter-coated with gold using a sputter coater (EMITECH K450, Kent, UK). The samples were examined at magnifications of 45×, 100×, and 500× for both surface and cross-sections, at an accelerating voltage of 10 kV.2.8 Carotenoid extraction
Carotenoid extraction was carried out according to the procedure described by Silva et al.25 Briefly, approximately 1 g of PF and control, 10% PF, and 20% PF cookies were macerated using a pestle and mortar with acetone and celite until exhaustive extraction. The acetone extracts were partitioned with petroleum ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl ether (1
ethyl ether (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, v/v) and washed with distilled water to remove acetone. Subsequently, the carotenoid extracts were saponified overnight (16 h) with KOH
2, v/v) and washed with distilled water to remove acetone. Subsequently, the carotenoid extracts were saponified overnight (16 h) with KOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (30
MeOH (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70, v/v) and again partitioned with petroleum ether
70, v/v) and again partitioned with petroleum ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl ether (1
ethyl ether (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, v/v), followed by washing with distilled water until complete removal of the alkali (pH ≈ 7). The extracts were evaporated using a rotary evaporator (T < 38 °C) and solubilized in petroleum ether. An aliquot of each extract was dried under a nitrogen flow and stored at −18 °C. Analyses were performed in triplicate (n = 3).
2, v/v), followed by washing with distilled water until complete removal of the alkali (pH ≈ 7). The extracts were evaporated using a rotary evaporator (T < 38 °C) and solubilized in petroleum ether. An aliquot of each extract was dried under a nitrogen flow and stored at −18 °C. Analyses were performed in triplicate (n = 3).
2.9 In vitro digestion and bioaccessibility of carotenoids
The in vitro digestion of the cookies was carried out according to the protocol proposed by INFOGEST 2.0,26 and the in vitro bioaccessibility of carotenoids was determined according to the adaptation for lipophilic compounds proposed by Rodrigues et al.27 Briefly, 5 g ± 0.2 g of previously crushed cookies were weighed. In the simulation of the oral phase, simulated salivary fluid (SSF), calcium chloride (0.3 M), and amylase were added, and the mixture was incubated for 2 min at 37 °C. In the simulation of the gastric phase, simulated gastric fluid (SGF), CaCl2 (0.3 M), and pepsin and gastric lipase were added to the bolus. After that, the pH was adjusted to 3.0 ± 0.2 with NaCl (0.1 M), and the mixture was incubated for 2 h at 37 °C. In the next step, simulated intestinal fluid (SIF), CaCl2 (0.3 M), pancreatin, and bile solution were added to the chyme. The pH was adjusted to 7.0 ± 0.2 with NaOH (0.1 M), and the mixtures were incubated for 2 h at 37 °C.After obtaining the simulated intestinal chyle, the material was centrifuged to separate the digesta and obtain the micellar fraction containing the bioaccessible carotenoid fraction. For this, the material was centrifuged at 4 °C, 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g for 15 min (Dupont Sorvall Rc5C and Sorvall rotor) and the supernatant was transferred to another centrifuge tube. Carotenoids were extracted from the micellar fraction using ethyl ether and NaCl solution (10%). Extraction was carried out by centrifugation at 4 °C (20
000 g for 15 min (Dupont Sorvall Rc5C and Sorvall rotor) and the supernatant was transferred to another centrifuge tube. Carotenoids were extracted from the micellar fraction using ethyl ether and NaCl solution (10%). Extraction was carried out by centrifugation at 4 °C (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g, 5 min) and repeated until the supernatant became completely colorless. The extracts obtained in each step were transferred to a separatory funnel containing distilled water and ethyl ether, after which the aqueous phase was discarded. The organic phase was then saponified with a 10% KOH methanolic solution (30
000 g, 5 min) and repeated until the supernatant became completely colorless. The extracts obtained in each step were transferred to a separatory funnel containing distilled water and ethyl ether, after which the aqueous phase was discarded. The organic phase was then saponified with a 10% KOH methanolic solution (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70, v/v) for 16 h (overnight) and subsequently partitioned in a separatory funnel with petroleum ether and ethyl ether (1
70, v/v) for 16 h (overnight) and subsequently partitioned in a separatory funnel with petroleum ether and ethyl ether (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) in a 1
1, v/v) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 extract ratio. Finally, the extract was washed with distilled water until complete removal of KOH and concentrated in a rotary evaporator (T < 38 °C). The bioaccessible carotenoid extract was dried under a nitrogen atmosphere and stored at −18 °C until the moment of HPLC-DAD analysis. Analyses were performed in triplicate (n = 3).
1 extract ratio. Finally, the extract was washed with distilled water until complete removal of KOH and concentrated in a rotary evaporator (T < 38 °C). The bioaccessible carotenoid extract was dried under a nitrogen atmosphere and stored at −18 °C until the moment of HPLC-DAD analysis. Analyses were performed in triplicate (n = 3).
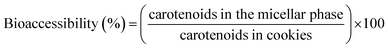
The bioaccessibility of carotenoids was determined using the equation below, which correlates the concentration of carotenoids in the micellar phase with the concentration of carotenoids in the cookie formulations:
 | (2) |
2.10 Identification and quantification of carotenoids by HPLC-DAD
The dry carotenoid extracts were solubilized in MeOH/methyl tert-butyl ether (MTBE) 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (v/v), filtered (0.22 µm), and injected into an Agilent HPLC (model 1200 Series, Agilent) equipped with a diode array detector (model G1315D, Agilent). The carotenoids were separated on a C30 YMC column (5 µm, 4.6 × 250 mm, Waters, USA) at 29 °C, flow rate of 0.9 mL min−1, with a linear gradient of MeOH/MTBE in the ratio of 95
10 (v/v), filtered (0.22 µm), and injected into an Agilent HPLC (model 1200 Series, Agilent) equipped with a diode array detector (model G1315D, Agilent). The carotenoids were separated on a C30 YMC column (5 µm, 4.6 × 250 mm, Waters, USA) at 29 °C, flow rate of 0.9 mL min−1, with a linear gradient of MeOH/MTBE in the ratio of 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 to 70
5 to 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 in 30 min, followed by 50
30 in 30 min, followed by 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 in 50 min to 95
50 in 50 min to 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 in 75 min, and maintaining these conditions for 7 min. Chromatograms were processed at 286, 347, and 450 nm.13,20 Carotenoids were identified based on the elution order on the C30 column, UV-vis [λmax], spectral fine structure (%III/II), cis peak intensity (%AB/AII), and comparison with literature data. Quantification was performed by external standardization using a nine-point calibration curve of lutein (0.72–183.8 µg mL−1), with LOD (2.00 µg mL−1) and LOQ (6.05 µg mL−1). Carotenoid composition analyses were performed in triplicate (n = 3).
5 in 75 min, and maintaining these conditions for 7 min. Chromatograms were processed at 286, 347, and 450 nm.13,20 Carotenoids were identified based on the elution order on the C30 column, UV-vis [λmax], spectral fine structure (%III/II), cis peak intensity (%AB/AII), and comparison with literature data. Quantification was performed by external standardization using a nine-point calibration curve of lutein (0.72–183.8 µg mL−1), with LOD (2.00 µg mL−1) and LOQ (6.05 µg mL−1). Carotenoid composition analyses were performed in triplicate (n = 3).
2.11 Pro-vitamin A activity
Vitamin A activity was calculated as retinol activity equivalents (RAE) according to the conversion factors described by NAS-ION,28 which establish that 12 µg of all-trans β-carotene corresponds to 1 µg RAE. For other provitamin A carotenoids with 50% vitamin A activity, a conversion factor of 24 µg was applied. All calculations were performed in triplicate (n = 3), and the results were expressed as µg RAE/g.2.12 Data analysis
The results were analyzed using one-way analysis of variance (ANOVA). Differences among the samples were evaluated using Tukey's test (p ≤ 0.05). Principal component analysis (PCA) was applied to explore correlations associated with PF addition in the cookie formulations. All analyses were performed using OriginLab software (version 2024b).3. Results and discussion
3.1 Proximate composition
Regarding the proximate composition, PF has proven to be a source of proteins (10.4 g/100 g), lipids (19.6 g/100 g), dietary fiber (33.4 g/100 g), and carbohydrates (34.5 g/100 g). The protein and lipid contents in this study were higher than those reported by Martínez-Girón et al.,29 who evaluated peel flours from the yellow and red varieties of peach palm cultivated in Colombia. Overall, PF was shown to be an excellent source of nutrients, with levels comparable to those observed in commonly used agro-industrial by-product flours, such as rice bran, extruded-expelled soybean bran, and millet, applied in gluten-free pasta formulations.30 In addition, PF exhibited higher nutrient contents than flours derived from other fruit and vegetable by-products, such as artichoke and banana, used in the production of gluten-free bakery products, except for ash and fiber contents.31,32 However, the high fiber content of PF paves the way for the exploration of this by-product flour as an alternative source of dietary fiber through its application in new food products. This approach may contribute to the WHO/FAO and EFSA recommendations, which indicate an average daily fiber intake of 25 g of fiber per adult.33The results of the proximate composition of the cookies (Table 2) showed that, among the cookie formulations, lipid and protein contents increased, while the carbohydrate content decreased in 10% and 20%-PF formulations compared to the control. Dietary fiber content increased proportionally with PF addition, making these formulations high-fiber products suitable for individuals with celiac disease. Table 2 shows that the control cookie, with 5 g of fiber per 100 g, is already considered a source of dietary fiber. With PF inclusion, the cookies reached the classification of “high in fiber” according to Brazilian legislation.34 These results are promising, as dietary fiber consumption is associated with several health benefits, including hypoglycemic activity, cholesterol regulation, modulation of the gut microbiota, and immunomodulatory effects.35 The higher ash content observed in the 10% PF formulation compared to the control cookie suggests a high mineral content in the peach palm peel.
| Parameter | Control | 10%-PF | 20%-PF |
|---|---|---|---|
| a Mean ± SD values in the same line with different superscript letters are significantly different (p < 0.05). The results are expressed on a dry basis. Carbohydrates are calculated by difference. | |||
| Ash (g/100 g) | 1.90 ± 0.08a | 1.85 ± 0.09b | 2.12 ± 0.03a |
| Lipid (g/100 g) | 17.8 ± 2.20b | 20.0 ± 0.54ab | 21.8 ± 0.32a |
| Protein (g/100 g) | 10.6 ± 0.55b | 11.3 ± 0.52ab | 11.8 ± 0.09a |
| Total dietary fiber (g/100 g) | 5.39 ± 0.56c | 7.00 ± 0.93c | 10.61 ± 1.13b |
| Carbohydrates (g/100 g) | 64.2 ± 2.60a | 59.6 ± 1.03b | 53.5 ± 0.36c |
| Total energy value (kcal/100 g) | 466.3 ± 0.07a | 464.8 ± 2.35a | 458.5 ± 1.47a |
3.2 Physical, textural, and color characteristics
Table 3 presents the physical properties of the cookies. The partial replacement of rice flour with peach palm peel flour (PF) significantly altered the color parameters of the cookies. As expected, there was a progressive increase in yellow (a*) and red (b*) components with increasing PF levels, while the L* value, which indicates lightness, decreased in PF-added cookies. These changes were attributed to the high carotenoid content of PF, especially the yellow-orange and red-orange pigments β-carotene and lutein.36,37 Color is considered a fundamental attribute of the sensory quality of gluten-free cookies, strongly influencing consumer preference.38 In addition to the positive visual impact, the color of carotenoid rich ingredients has been associated with healthier food perceptions. Bechoff et al.39 reported that yellowish dough-like preparations made with carotenoid-enriched cassava were associated by consumers with attributes such as “good for eyesight,” resulting in higher acceptability compared to products made with traditional white cassava.| Parameters | Control | 10%-PF | 20%-PF |
|---|---|---|---|
| a Mean ± SD values in the same line with different superscript letters are significantly different (p < 0.05). L* = lightness; a* = red-green coordinate; b* = yellow-blue coordinate; PF = peach palm peel flour. | |||
| Weight (g) | 25.5 ± 0.02b | 26.0 ± 0.02a | 25.2 ± 0.01c |
| Diameter (cm) | 5.40 ± 0.36b | 5.73 ± 0.15b | 6.53 ± 0.25a |
| Water activity | 0.90 ± 0.02a | 0.90 ± 0.02a | 0.87 ± 0.03a |
| Thickness (cm) | 1.72 ± 0.03a | 1.54 ± 0.03a | 1.68 ± 0.42a |
| Hardness (N) | 35.4 ± 2.30b | 38.4 ± 2.18ab | 41.2 ± 0.99a |
| Fracturability (mm) | 7.72 ± 0.43a | 7.29 ± 0.29a | 6.63 ± 0.71a |
| L* | 81.3 ± 0.37a | 73.5 ± 0.38b | 66.4 ± 0.38c |
| a* | 5.70 ± 0.06c | 9.33 ± 0.49b | 11.8 ± 0.11a |
| b* | 38.6 ± 0.52b | 42.6 ± 1.30a | 44.3 ± 0.32a |
| C* | 39.1 ± 0.52c | 43.6 ± 1.36b | 45.9 ± 0.29a |
| ΔE | — | 9.49 ± 0.76b | 17.0 ± 0.13a |
The total color difference (ΔE) was 9.4 and 17 for the 10% and 20% PF formulations, respectively. As reported in ref. 40 and the perceptibility thresholds established in the CIELAB color space, ΔE values between 2 and 10 are perceptible at a glance, while values between 11 and 49 indicate clearly distinguishable colors. The ΔE values reported in this study, therefore, indicate color differences that are readily perceptible to the human eye, demonstrating that PF incorporation resulted in pronounced visual changes that became more intense with increasing PF concentration. These findings are consistent with the high carotenoid content of the ingredient.
PF addition not only altered the color of the cookies but also introduced complementary compounds into the formulation, enabling chemical interactions within the food matrix that affected physical properties. Specifically, cookie hardness increased by 8.5% and 16.4% in formulations containing 10% and 20% PF, respectively, compared to the control, indicating a progressive reinforcement of the cookie structure. Hardness modifications may be attributed to the fiber fraction of PF, which accounts for approximately 10–25% of the total carbohydrate content.29 Dietary fibers can restrict water availability within the dough matrix due to their water-binding capacity, thereby increasing structural rigidity during baking. In gluten-free systems, this behavior may partially mimic some structural functions of the gluten network, contributing to increased hardness.41
In contrast, fracturability values did not differ significantly among formulations (p > 0.05). Nevertheless, from a mechanistic perspective, the higher lipid content of PF-based cookies may have influenced the cookie structure by enhancing tenderness and plasticity, which are well established functions of fats in cookie manufacturing.42 Additionally, the increase in unsaturated fatty acids promoted by PF replacement may be related to the higher diameter observed in 20% PF cookies, as unsaturated fatty acids reduce the melting point of fats, resulting in greater spread during baking.43
Water activity (aw) is a critical parameter in cookie production, as it controls the texture,44 and directly affects microbiological stability and shelf life.45,46 As shown in Table 3, the incorporation of peach palm peel flour (PF) did not significantly influence aw values (p > 0.05), which ranged from 0.874 to 0.909. The lack of variation in water activity (aw) among the control formulation and those containing 10% and 20% PF may be explained by the type of dietary fiber present in peach palm peel flour. In general, dietary fibers promote water retention through binding interactions, and water strongly associated with fiber matrices reduces the fraction of free water in the system, which may lead to lower aw values.47,48 However, this mechanism is strongly modulated by fiber type, being more pronounced in the presence of soluble fibers, whereas approximately 80% of the fibers present in PF are insoluble.49
On the other hand, these values are higher than those reported for other gluten-free cookie formulations (0.24–0.48), indicating a greater availability of water in the system.10,32,50 The high aw values observed for all formulations suggest a potential risk of microbiological instability, highlighting the limitations of the formulations proposed in this study and emphasizing the need for future formulation adjustments to obtain PF enriched cookies with improved safety for consumption.
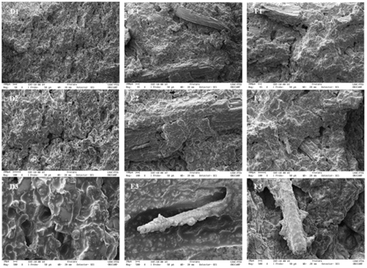
3.3 Scanning electron microscopy
The impact of PF on the microstructure of the cookies was assessed by scanning electron microscopy (SEM). The images revealed that the control cookies exhibited more regular surfaces (Fig. 1; A1, A2, and A3), which may be attributed to the higher levels of gelatinized starch in this formulation. Gelatinization causes the granules to melt, altering their structure into a stretched form, which enhances the chewiness of the cookies.51 Moreover, punctual round starch granules can be observed on the surface of all cookies, especially small and medium-sized granules in PF-based cookies (Fig. 1, B3 and C3). This observation is consistent with the results obtained by Indrianingsih et al.52 for cookies containing mixtures of wheat and sorghum flour with mangosteen peels in different percentages.In the cross-sectional view, continuous open starch structures were observed, indicating the entrapment of gas bubbles during baking (Fig. 2, D3, E3, and F3). Furthermore, an attached fiber structure was observed exclusively in PF cookies (Fig. 2, E3 and F3), disrupting the continuity of the open starch matrix. The presence of fibers may also reduce starch gelatinization and restrict its susceptibility to enzymatic hydrolysis during digestion. A similar SEM observation was reported by Ng et al.53 for cookies formulated with Pleurotus sajor-caju powder, a dietary fiber source, which was associated with a low glycemic response.
3.4 Carotenoid composition
Fig. 3 presents the chromatogram with the carotenoid profile of PF and cookies formulated with rice flour and PF, while Table 4 shows the elution order of the compounds, as well as their spectral data. Seventeen carotenoids were tentatively identified based on spectral data and information available in the literature. The major carotenoids identified in PF were all-E-lutein (peak 3) and all-E-β-carotene (peak 10). The same profile was observed by Silva et al.,16 who evaluated the impact of cooking peach palm fruits on the carotenoid profile of this fruit peel flour. On the other hand, Martínez-Girón et al.29 did not find lutein in yellow peach palm peel flour. This behavior may be associated with several factors, such as climatic seasons and fruit growth in different regions, which can influence the composition of carotenoids in different matrices.54–56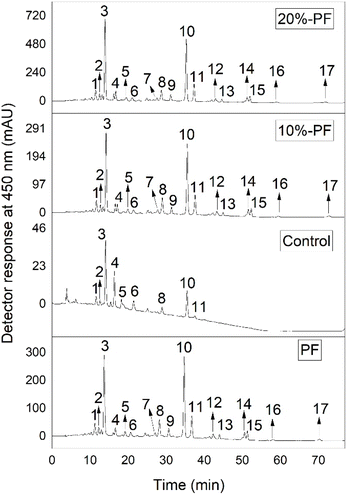 | ||
| Fig. 3 HPLC-DAD chromatogram recorded at 450 nm of carotenoids from yellow peach palm peel flour and control, 10%-PF, and 20%-PF cookies. Carotenoid identification according to Tables 4 and 5. | ||
| Peaka | Carotenoidb | RTc (min) | lmáxd (nm) | %III/II | %AB/AII |
|---|---|---|---|---|---|
| a Numbered according to the chromatogram in Fig. 4.b Tentative identification based on UV-vis spectra, retention time on the C30 column, and published data (Chisté et al., 2021; Menezes Silva et al., 2023).c Retention time on the C30 column.d Linear gradient of methanol/MTBE. n.c: not calculated. | |||||
| 1 | 15Z-Lutein | 11.3 | 330, 418, 449, 470 | 32 | 57 |
| 2 | 13Z-Lutein | 12.3 | 330, 410, 440, 465 | 43 | 38 |
| 3 | (All-E)-lutein | 13.7 | 421, 445, 474 | 61 | 0 |
| 4 | (All-E)-zeaxanthin | 16.6 | 424, 449, 476 | 33 | 0 |
| 5 | (9Z)-Lutein | 17.1 | 330,415, 440, 469 | 62 | 7 |
| 6 | (9Z)-Lutein (isomer 2) | 19.2 | 329, 415, 440, 469 | 61 | 7 |
| 7 | 15Z-β-Carotene | 27.7 | 335, 424, 447, 474 | n.c | n.c |
| 8 | 13Z-β-Carotene | 28.1 | 338, 420, 447, 460 | 15 | 45 |
| 9 | 9Z,13Z-β-Carotene | 30.6 | 422, 445, 473 | 57 | 0 |
| 10 | (All-E)-β-carotene | 34.6 | 425, 450, 477 | 25 | 0 |
| 11 | 9Z-β-Carotene | 36.7 | 416, 445, 473 | 66 | 9 |
| 12 | Di-Z-δ-carotene | 42.6 | 351, 428, 452, 481 | 48 | 12 |
| 13 | (All-E)-δ-carotene | 43.9 | 430, 454, 483 | 50 | 0 |
| 14 | 9Z-γ-Carotene | 50.4 | 358, 438, 458, 490 | 63 | n.c |
| 15 | All-E-γ-carotene | 51.3 | 436, 457, 491 | 50 | 0 |
| 16 | 9Z-Lycopene | 57.9 | 360, 440, 466, 498 | 73 | 12 |
| 17 | All-E-lycopene | 70.2 | 444, 470, 502 | 75 | 0 |
Regarding the cookie's carotenoid profile, nine compounds were identified in the control formulation; the main ones were all-E-lutein (peak 3), all-E-zeaxanthin (peak 4), and all-E-β-carotene (peak 10). These carotenoids may be associated with some of the ingredients used in the formulation, such as eggs, butter, and milk.57–59 On the other hand, the carotenoid profile of the cookies 10%-PF and 20%-PF presented 17 compounds, the same ones found in PF.
Furthermore, the addition of PF promoted an increase in the intensity of all-E-lutein and all-E-β-carotene peaks (Fig. 3). These carotenoids have important biological activities in the human body, such as the formation of the macula lutea,60 slowing the progression of age-related macular degeneration,61 protecting the skin against sunlight, reducing the risk of cancer, and provitamin A activity.62
In addition to lutein and β-carotene, Z-isomers of these carotenoids were also identified, such as the carotenoids 9Z-, 13Z-, and 15Z-lutein (peaks 5, 6, 2, and 1, respectively) and 9Z-, 9Z, 13Z-, 13Z-, and 15Z-β-carotene (peaks 11, 9, 8, and 7, respectively). Peaks 12 and 13 were identified as di-Z-δ-carotene and (all-E)-δ-carotene. These carotenoids are less common in nature; however, previous studies with the pulp and peel of peach palm also reported these compounds.13,16 The carotenoids all-E-γ-carotene (peak 15) and all-E-lycopene (peak 17) were also identified in the cookies 10%-PF and 20%-PF, both of which were previously detected by Martínez-Girón et al.29 in yellow peach palm peel flour. Furthermore, the respective Z-isomers of the carotenoids mentioned above, 9Z-γ-carotene and 9Z-lycopene, were also identified (peaks 14 and 16, respectively) in cookies with PF.
Table 5 presents the carotenoid content (µg g−1) and the vitamin A activity (µg RAE/g) in PF and the gluten-free cookies. Carotenoid content of PF was 264.1 µg g−1, a value higher than those reported by Silva et al.16 and Martínez-Girón et al.,29 who observed levels between 75.0 and 129 µg g−1 in yellow peach palm peel flour. This difference may be associated with climatic factors and growing regions, as highlighted previously. The (all-E)-lutein content was 73.2 µg g−1, a value close to that observed in green leafy vegetables, considered excellent sources of this carotenoid.63,64 The all-E-β-carotene content was 85.4 µg g−1, contributing to approximately 72% of the provitamin A activity of PF (7.1 µg RAE/g).
| Peaka | Carotenoid | Carotenoid contents (µg g−1, dry weight) | |||
|---|---|---|---|---|---|
| PF | Control | 10%-PF | 20%-PF | ||
| a Carotenoids were quantified as β-carotene equivalents. Means with different lowercase letters within a line were significantly different (p < 0.05). nd = not detected. | |||||
| 1 | 15Z-Lutein | 11.1 ± 0.30a | 0.18 ± 0.08d | 0.80 ± 0.03c | 1.79 ± 0.05b |
| 2 | 13Z-Lutein | 6.37 ± 0.11a | 0.10 ± 0.04d | 0.46 ± 0.22c | 1.00 ± 0.03b |
| 3 | (All-E)-lutein | 73.2 ± 1.03a | 1.12 ± 0.07d | 5.82 ± 0.11c | 13.6 ± 0.14b |
| 4 | Zeaxanthin | 6.15 ± 0.19a | 0.58 ± 0.01b | 0.50 ± 0.03b | 0.75 ± 0.01b |
| 5 | 9Z-Lutein | 3.46 ± 0.14a | 0.15 ± 0.08d | 0.48 ± 0.01c | 1.08 ± 0.04b |
| 6 | 9Z-Lutein (isomer 2) | 4.96 ± 0.18a | 0.24 ± 0.06c | 0.38 ± 0.10c | 0.90 ± 0.03b |
| 7 | 15Z-β-Carotene | 3.80 ± 0.14a | nd | 0.34 ± 0.07b | 0.54 ± 0.02b |
| 8 | 13Z-β-Carotene | 19.0 ± 0.16a | 0.17 ± 0.06d | 1.51 ± 0.06c | 2.38 ± 0.03b |
| 9 | 9Z, 13Z-β-carotene | 9.59 ± 0.91a | nd | 0.53 ± 0.01b | 1.11 ± 0.05b |
| 10 | (All-E)-β-carotene | 85.4 ± 4.22a | 0.54 ± 0.01d | 6.24 ± 0.02c | 12.4 ± 0.11b |
| 11 | 9Z-β-Carotene | 18.8 ± 0.19a | 0.07 ± 0.01d | 1.89 ± 0.02c | 3.51 ± 0.10b |
| 12 | Di-Z-δ-carotene | 1.60 ± 0.02a | nd | 0.30 ± 0.02c | 1.02 ± 0.56b |
| 13 | (All-E)-δ-carotene | 3.40 ± 0.05a | nd | 0.52 ± 0.01b | 0.54 ± 0.01b |
| 14 | 9Z-γ-Carotene | 4.94 ± 2.46a | nd | 0.43 ± 0.02b | 0.69 ± 0.02b |
| 15 | All-E-γ-carotene | 7.47 ± 0.35a | nd | 0.59 ± 0.02b | 1.02 ± 0.01b |
| 16 | 9Z-Lycopene | 2.03 ± 0.03a | nd | 0.17 ± 0.01b | 0.16 ± 0.01b |
| 17 | (All-E)-Lycopene | 2.13 ± 0.19a | nd | 0.21 ± 0.01b | 0.35 ± 0.01b |
| Total carotenoids | 264.1 ± 1.56a | 3.18 ± 0.08d | 21.2 ± 0.20c | 42.9 ± 0.38b | |
| Vitamin A equivalent (µg RAE/g) | 9.77 ± 0.27a | 0.06 ± 0.01d | 0.74 ± 0.01c | 1.43 ± 0.01b | |
Regarding the carotenoid content of the gluten-free cookies, a significant increase in the final concentration—from 3.73 to 21.2 and 42.9 µg g−1, for 10% and 20% PF addition, respectively—was observed. Furthermore, significant increases in individual all-E-lutein content were observed with the addition of PF (0.14, 5.82, and 13.6 µg g−1) and its respective Z isomers, 15Z-lutein, 13Z-lutein, and 9Z-lutein. The same behavior was observed for all-E-β-carotene (0.67, 6.24, and 12.4 µg g−1) and the 13Z-β-carotene and 9Z-β-carotene isomers. The provitamin A activity also increased with the addition of PF, going from 0.06 µg RAE/g in the control formulation to 0.74 and 1.43 µg RAE/g in cookies with 10% and 20% PF, respectively. Considering the consumption of up to three cookies and the portion size in Table 3, the provitamin A content increased to 1.43 µg RAE/g in cookies formulated with 20% PF. Considering the consumption of three cookies (portion size shown in Table 3), this amount corresponds to approximately 27% and 18% of the RDA values for vitamin A, as proposed by National Institutes of Health (NIH),65 for children aged 4–8 years and 9–13 years, respectively.
3.5 Bioaccessibility of lutein e β-carotene
Fig. 4 and Table 6 present the bioaccessibility, in percentage (%), and the bioaccessible content (µg g−1) of lutein and β-carotene in gluten-free cookies, respectively, after simulated in vitro digestion.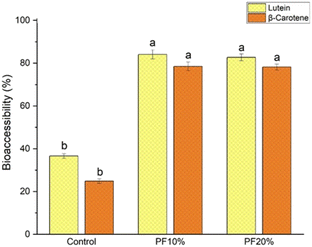 | ||
| Fig. 4 Bioaccessibility of lutein and β-carotene in cookies formulated with rice flour: control (100% rice flour) and with the addition of PF (10%-PF and 20%-PF). | ||
| Carotenoid | Bioaccessible carotenoid content (µg g−1) | ||
|---|---|---|---|
| Control | 10%-PF | 20%-PF | |
| a Different letters on the same line indicate significant differences (p < 0.05). | |||
| (All-E)-lutein | 0.41 ± 0.02c | 4.90 ± 0.14b | 11.2 ± 1.18a |
| (All-E)-β-carotene | 0.13 ± 0.01c | 4.89 ± 0.04b | 9.73 ± 2.21a |
The control cookie showed bioaccessibility of 36.6% for (all-E)-lutein (0.41 µg g−1) and 24.8% for (all-E)-β-carotene (0.13 µg g−1), respectively. Furthermore, the addition of PF promoted a positive and significant impact (p < 0.05) on the bioaccessibility of these two carotenoids in the cookies with 10%-PF and 20%-PF. The bioaccessible contents (Table 6) of lutein were 4.90 and 11.2 µg g−1, and those of β-carotene were, 4.89 and 9.73 µg g−1 in the 10%-PF and 20%-PF cookies, respectively. Although there was an increase in the bioaccessible content of the carotenoids, the bioaccessibility of these two carotenoids remained stable in the cookie formulations with PF. A similar behavior was reported in ref. 66 who incorporated lutein in cupcakes (2, 3, and 6 mg of lutein per serving) and found bioaccessibility of this xanthophyll around 60%.
There are some limiting factors in the bioaccessibility of carotenoids, such as release from the food matrix, formation of micelles, and interactions with other components in the matrix.67 Some hypotheses can be raised concerning the stability of the in vitro bioaccessibility of lutein and β-carotene in 10%-PF and 20%-PF cookies, including the effect of lipids. Table 2 shows that these two cookie formulations have the same amount of lipids, and this could be a limiting factor in the ability to form micelles. Lipids stimulate bile secretion, which promotes lipid emulsification, and stimulates the secretion of pancreatic lipase, which in turn hydrolyzes lipids into smaller molecules that together with bile salts, form mixed micelles.68 However, some studies suggest that at certain concentration levels, lipids can reduce the micellarization of carotenoids in some food systems.69,70
In this context, another possible hypothesis for the stability of bioaccessibility values is the interference of fibers in 20%-PF, which contained 51% more fiber than 10%-PF. Insoluble fibers can form fiber–carotenoid complexes that tend to hinder the release of carotenoids from the food matrix. Furthermore, soluble fibers can promote an increase in the viscosity of the food bolus, and this can be a key factor in reducing lipid emulsification.71,72
3.6 Principal component analysis (PCA)
Fig. 5 presents the PCA biplot graph aimed at helping understand the direct association between the addition of PF and the analyzed parameters in gluten-free cookies. The first principal component (PC1) and the second principal component (PC2) explained 84.41% and 15.59% of the variance, respectively, which provided a robust representation of the data since the sum of explained variances was 100%. The control cookie formulation was more associated with thickness (r = −0.529, PC2) and fracturability (r = −0.133, PC2), whereas the 10%-PF formulation showed higher association with protein (r = 0.479, PC2), lutein bioaccessibility (r = 0.273, PC2), and color parameters (b* = 0.123, C* = 0.103, PC2). The 20%-PF formulation was closer to weight (r = 0.513, PC2), β-carotene (r = 0.263, PC2), and provitamin A (r = 0.012, PC2).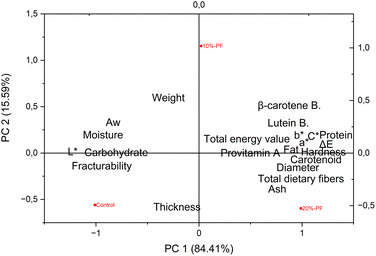 | ||
| Fig. 5 Biplot graph of the Principal Component Analysis (PCA) presenting correlations (100% of the variance) among the evaluated parameters of the control, 10%-PF and 20%-PF cookies. | ||
In general, the right side of the PCA biplot is strongly associated with nutritional characteristics, including carotenoid content and bioaccessibility, while the left side, where the control sample is located, is more related to physicochemical properties, such as fracturing, water activity (aw), and thickness. These results are in accordance with previous analyses and indicate that the addition of peach palm peel flour (PF) positively impacts the nutritional composition and carotenoid content of cookies. Furthermore, the graph suggests that this replacement may have reduced parameters such as thickness and fracturability, since the control sample moved to the opposite region in the PCA biplot. It is worth highlighting that 10%-PF represents an intermediate formulation, showing characteristics of both cookies (control and 20%-PF), which reinforces the observed trend. The PCA biplot allowed us to visualize a significant differentiation between all cookie formulations, in addition to identifying trends and possible adjustments that can be explored in future studies on the incorporation of peach palm peel flour into bakery products.
4 Conclusions
The present study demonstrated that yellow peach palm peel flour (PF) can be seen as a promising alternative as a functional ingredient, rich in carotenoids and fiber, for use in the formulation of gluten-free cookies based on rice flour. The addition of PF can be a strategy to improve the nutritional quality of cookies, in addition to contributing to the incorporation and bioaccessibility of lutein and β-carotene, compounds associated with provitamin A activity and eye health. The PCA indicated that adjustments in the formulation can be planned to optimize the nutritional and technological characteristics of cookies. Furthermore, the use of PF, a by-product of an Amazonian fruit, represents a promising approach for the full use of fruits from the Amazon region to stimulate a circular bioeconomy.Some aspects remain to be explored, particularly sensory evaluation, to better understand consumer acceptance. Future studies should address starch digestibility, antioxidant activity, glycemic response, and the development of vegan formulations by replacing animal-derived ingredients. In addition, economic feasibility, food safety aspects, and the potential presence of contaminants such as mycotoxins should be evaluated to support large-scale application and contributions to food security. Packaging strategies and shelf-life stability should also be investigated to further improve the technological and functional performance of the cookies.
Author contributions
Jhonathan Vinícius Menezes Silva: conceptualization, methodology, investigation, formal analysis, data curation, writing – original draft. Allien Monique Rosa Machado: methodology, investigation, formal analysis, data curation, writing – original draft. Andressa Mara Baseggio: methodology, investigation, formal analysis, data curation, writing – original draft. Renan Campos Chisté: funding acquisition, resources, methodology, investigation, formal analysis, data curation, writing – review & editing. Lilian Regina Barros Mariutti: funding acquisition, resources, methodology, investigation, formal analysis, data curation, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
Data can be obtained by request to the authors.Acknowledgements
Fundação de Amparo à Pesquisa do Estado de São Paulo – FAPESP (grant number 2022/09998-3 and process EMU 09/51580-1), Fundação Amazônia de Amparo a Estudos e Pesquisas – FAPESPA (Process 2023/557510, TO 037/2023) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq (Process n° 140834/2023-0) are acknowledged.References
- E. Ervina, Int. J. Gastron. Food Sci., 2023, 33, 100796 CrossRef.
- Joint Fao/Who Food Standards Programme Codex Alimentarius Commission Twenty-Second Session Geneva, 23–28 June 1997 Report Of The 1 Wentieth Session Of The Codex Committee On Nutrition And Foods For Special Dietary Uses.
- J. Xu, Y. Zhang, W. Wang and Y. Li, Trends Food Sci. Technol., 2020, 103, 200–213 CrossRef CAS.
- D. Singla, T. Malik, A. Singh, S. Thakur and P. Kumar, Food Chem. Adv., 2024, 4, 100627 CrossRef.
- R. Fernandes Almeida, L. A. Borges, D. Anacleto, M. S. N. de Souza, L. B. M. da Silva, Y. M. Monroy, E. A. C. Batista and A. P. O. Machado, Food and Humanity., 2025, 4, 100549 CrossRef.
- G. Vici, L. Belli, M. Biondi and V. Polzonetti, Clinical Nutrition, 2016, 35(6), 1236–1241 CrossRef CAS PubMed.
- H. B. Itagi, K. J. D. Sartagoda, N. Gupta, V. Pratap, P. Roy, R. N. Tiozon, A. Regina and N. Sreenivasulu, LWT--Food Sci. Technol., 2023, 186, 115245 CrossRef CAS.
- E. A. Özer, J. Food Process. Preserv., 2022, 46, e16302 Search PubMed.
- L. Schmelter, H. Rohm and S. Struck, Future Foods, 2021, 4, 100038 Search PubMed.
- S. Kumari, B. Singh and A. Kaur, Food Chem. Adv., 2023, 3, 100557 CrossRef.
- J. Martínez-Girón, C. Osorio and L. E. Ordoñez-Santos, Food Sci. Technol. Int., 2022, 28, 535–544 Search PubMed.
- M. Di Cairano, F. Galgano, R. Tolve, M. C. Caruso and N. Condelli, Trends Food Sci. Tech., 2018, 81, 203–212 CrossRef CAS.
- R. C. Chisté, E. L. N. Costa, S. F. Monteiro and A. Z. Mercadante, J. Food Compos. Anal., 2021, 99, 103873 CrossRef.
- S. D. Soares, O. V. Dos Santos, F. D. C. A. D. Nascimento, R. da and S. Pena, Inter. J. Food Prop., 2022, 25(1), 2146–2165 CrossRef CAS.
- K. A. Noronha Matos, D. Praia Lima, A. P. Pereira Barbosa, A. Zerlotti Mercadante and R. Campos Chisté, Food Chem., 2019, 272, 216–221 CrossRef CAS PubMed.
- J. V. M. SilvaG. P. MacêdoJ. S. MouraR. C. Chisté, in Ciências Agrárias: o avanço da ciência no Brasil, Editora Científica Digital, 2022, vol. 5, pp. 214–233 Search PubMed.
- S. Mitra, A. Rauf, A. M. Tareq, S. Jahan, T. Bin Emran, T. G. Shahriar, K. Dhama, F. A. Alhumaydhi, A. S. M. Aljohani, M. Rebezov, M. S. Uddin, P. Jeandet, Z. A. Shah, M. A. Shariati and K. R. Rengasamy, Food Chem. Toxicol., 2021, 154, 112328 CrossRef CAS PubMed.
- D. B. Rodrigues, M. C. Marques, A. Hacke, P. S. Loubet Filho, C. B. B. Cazarin and L. R. B. Mariutti, Current Research in Food Science, 2022, 5, 228–233 CrossRef CAS PubMed.
- X. Yan, J. Huang, L. Huang, C. Luo, Z. Li, P. Xu, K. Tan, K. L. Cheong and K. Tan, LWT--Food Sci. Technol., 2024, 200, 116171 Search PubMed.
- J. V. Menezes Silva, A. Silva Santos, G. Araujo Pereira and R. Campos Chisté, Heliyon, 2023, 9(4), e14933 CrossRef CAS PubMed.
- I. Licona-Aguilar, J. A. Lois-Correa, A. M. Torres-Huerta, M. A. Domínguez-Crespo, V. Urdapilleta-Inchaurregui, A. E. Rodríguez-Salazar and S. B. Brachetti-Sibaja, LWT--Food Sci. Technol., 2023, 184, 115061 CrossRef.
- AOAC, Official Methods of Analysis, Association of Official Analytical Chemists, Gaithersburg, USA, 2000, vol. 17th Search PubMed.
- AOAC, AOAC Official Methods of Analysis, Association of Official Analytical Chemists. , Gaithersburg, USA, 18th edn, 2012 Search PubMed.
- G. Silva Ribeiro, M. K. Conceição Monteiro, J. Rodrigues do Carmo, R. da Silva Pena and R. Campos Chisté, Heliyon, 2021, 7(5), e07062 CrossRef CAS PubMed.
- J. V. M. Silva, I. S. Amorim, D. S. Amorim, C. A. Camargo and L. R. B. Mariutti, Carotenoid Anal. Methods Protoc. Food Sci., 2025, 1, 25–43 Search PubMed.
- A. Brodkorb, L. Egger, M. Alminger, P. Alvito, R. Assunção, S. Ballance, T. Bohn, C. Bourlieu-Lacanal, R. Boutrou, F. Carrière, A. Clemente, M. Corredig, D. Dupont, C. Dufour, C. Edwards, M. Golding, S. Karakaya, B. Kirkhus, S. Le Feunteun, U. Lesmes, A. Macierzanka, A. R. Mackie, C. Martins, S. Marze, D. J. McClements, O. Ménard, M. Minekus, R. Portmann, C. N. Santos, I. Souchon, R. P. Singh, G. E. Vegarud, M. S. J. Wickham, W. Weitschies and I. Recio, Nat. Protoc., 2019, 14, 991–1014 CrossRef CAS PubMed.
- D. B. Rodrigues, L. R. B. Mariutti and A. Z. Mercadante, Food Funct., 2016, 7, 4992–5001 Search PubMed.
- Institute of Medicine, Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc, National Academies Press, Washington, D.C., 2001 Search PubMed.
- J. Martínez-Girón, C. Cafarella, F. Rigano, D. Giuffrida, L. Mondello, Y. Baena, C. Osorio and L. E. Ordóñez-Santos, ACS Omega, 2024, 9, 28738–28753 CrossRef PubMed.
- K. F. Irigoytia, C. E. Genevois and M. F. de Escalada Pla, Future Foods, 2025, 11, 100515 Search PubMed.
- I. Proetto, F. Pesce, E. Arena, A. Grasso, L. Parafati, B. Fallico and R. Palmeri, Int. J. Gastron. Food Sci., 2024, 38, 101015 CrossRef.
- P. Pomasa, S. Sai-Ut, S. Sinthusamran and C. Techavuthiporn, J. Agric. Food Res. DOI:10.1016/j.jafr.2025.101744.
- World Health Organization, WHO Updates Guidelines On Fats And Carbohydrates, 2023 Search PubMed.
- ANVISA, Resolução RDC no 54, de 12 de novembro de 2012: Regulamento técnico sobre informação nutricional complementar, Brazil, 2012.
- W. Tang, X. Lin, N. Walayat, J. Liu and P. Zhao, Crit. Rev. Food Sci. Nutr., 2024, 64(22), 7895–7915 Search PubMed.
- H. E. Khoo, K. N. Prasad, K. W. Kong, Y. Jiang and A. Ismail, Molecules, 2011, 16(2), 1710–1738 CrossRef CAS PubMed.
- A. J. Meléndez-Martínez, G. Britton, I. M. Vicario and F. J. Heredia, Food Chem., 2007, 101, 1145–1150 Search PubMed.
- V. D. Capriles, E. Valéria de Aguiar, F. Garcia dos Santos, M. E. A. Fernández, B. G. de Melo, B. L. Tagliapietra, M. Scarton, M. T. P. S. Clerici and A. C. Conti, Food Res. Inter., 2023, 173(2), 113389 Search PubMed.
- A. Bechoff, U. Chijioke, A. Westby and K. I. Tomlins, PLoS One, 2018, 13(9), e0203421 Search PubMed.
- A. Nemś, J. Miedzianka and A. Kita, J. Sci. Food Agric., 2022, 102, 4629–4639 CrossRef PubMed.
- S. Rout, R. K. Gupta, H. B. Jadhav, P. P. Srivastav and U. Annapure, in Development of Gluten-Free Pasta, Elsevier, 2024, pp. 157–178 Search PubMed.
- M. Goubgou, L. T. Songré-Ouattara, F. Bationo, H. Lingani-Sawadogo, Y. Traoré and A. Savadogo, Food Prod. Proc. Nutr., 2021, 3, 26 CrossRef PubMed.
- A. Devi and B. S. Khatkar, J. Food Sci. Technol., 2018, 55, 321–330 CrossRef CAS PubMed.
- D. J. McClements, in Future Foods, Springer International Publishing, Cham, 2019, pp. 27–60 Search PubMed.
- M. Bawa, A. Dzigbor, V. A. Dela Gobe, G. F. Opoku, A. A. T. Barima and A. K. L. Donkor, J. Food Process. Preserv., 2023, 2023, 6762289 Search PubMed.
- S. Z. Asadi, M. A. Khan and S. Zaidi, J. Food Sci. Technol., 2022, 59, 3848–3856 Search PubMed.
- P. M. G. Ishida and C. J. Steel, J. Food Sci. Technol., 2015, 34, 746–754 CrossRef.
- M. B. Różańska, J. Zembrzuska, P. Rychlewski, M. Kidoń, Ł. Masewicz, S. Mildner-Szkudlarz and H. M. Baranowska, Food Chem., 2025, 491, 145194 CrossRef PubMed.
- J. Martínez-Girón, X. Rodríguez-Rodríguez, L. X. Pinzón-Zárate and L. E. Ordóñez-Santos, Corpoica Ciencia y Tecnologia Agropecuaria, 2017, 18, 599–613 Search PubMed.
- K. Selvaraju, M. Mathur, S. SSM, J. K. Sahu, P. Hariprasad and A. Malik, Algal Res., 2024, 82, 103683 Search PubMed.
- M. Thakur, A. K. Rai and S. P. Singh, ACS Omega, 2023, 8, 25799–25807 CrossRef CAS PubMed.
- A. W. Indrianingsih, Y. Khasanah, C. Darsih, H. P. Hastuti, A. E. Suryani, M. Hastuti, D. Ni’maturrohmah, U. Laila, E. Noviana, E. Rahayu and T. Wiyono, Bioact. Carbohydr. Diet. Fibre, 2025, 33, 100467 CrossRef CAS.
- S. H. Ng, S. D. Robert, W. A. N. Wan Ahmad and W. R. Wan Ishak, Food Chem., 2017, 227, 358–368 Search PubMed.
- J. Stoń-Egiert, M. Ostrowska and R. Majchrowski, Oceanologia, 2024, 66, 405–423 Search PubMed.
- J. Singh, R. Metrani, G. K. Jayaprakasha, K. M. Crosby, J. L. Jifon, S. Ravishankar, P. Brierley, D. L. Leskovar, T. A. Turini, J. Schultheis, T. Coolong, W. Guan and B. S. Patil, J. Food Compos. Anal., 2022, 106, 104306 Search PubMed.
- Q. Lu, X. Huang, S. Lv and S. Pan, Food Chem., 2017, 232, 788–798 CrossRef CAS PubMed.
- D. Zurak, Z. Svečnjak, G. Kiš, V. Pirgozliev, D. Grbeša and K. Kljak, Poult. Sci., 2025, 104(3), 104843 Search PubMed.
- T. F. O'Callaghan, H. Faulkner, S. McAuliffe, M. G. O'Sullivan, D. Hennessy, P. Dillon, K. N. Kilcawley, C. Stanton and R. P. Ross, J. Dairy Sci., 2016, 99, 9441–9460 Search PubMed.
- T. Mitani, K. Kobayashi, K. Ueda and S. Kondo, Anim. Sci. J., 2021, 92(1), e13570 Search PubMed.
- B. Li, F. yen Chang, Z. Wan, N. A. Giauque, E. K. Addo and P. S. Bernstein, Exp. Eye Res., 2024, 247, 110043 CrossRef CAS PubMed.
- S. Sabour-pickett, S. Beatty, E. Connolly, J. Loughman, J. Stack, A. Howard, R. Klein, B. E. Klein, S. M. Meuer, C. E. Myers, K. O. Akuffo and J. M. Nolan, Retina, 2014, 34(9), 1757–1766 CrossRef CAS PubMed.
- D. B. Rodriguez-Amaya, in Global Perspectives on Astaxanthin: From Industrial Production to Food, Health, and Pharmaceutical Applications, Elsevier, 2021, pp. 693–717 Search PubMed.
- J. N. Eriksen, A. Y. Luu, L. O. Dragsted and E. Arrigoni, Food Chem., 2016, 203, 23–27 Search PubMed.
- D. Šamec, B. Urlić and B. Salopek-Sondi, Crit. Rev. Food Sci. Nutr., 2019, 59(15), 2411–2422 CrossRef PubMed.
- National Institutes of Health, Vitamin A – Fact Sheet for Health Professionals, 2022 Search PubMed.
- A. A. O. Xavier, I. Carvajal-Lérida, J. Garrido-Fernández and A. Pérez-Gálvez, J. Food Compos. Anal., 2018, 68, 60–64 Search PubMed.
- A. A. O. Xavier and A. Z. Mercadante, Current Opinion in Food Science, 2019, 26, 1–8 CrossRef.
- E. Fernández-García, I. Carvajal-Lérida, M. Jarén-Galán, J. Garrido-Fernández, A. Pérez-Gálvez and D. Hornero-Méndez, Food Res. Inter., 2012, 46(2), 438–450 Search PubMed.
- F. C. Petry and A. Z. Mercadante, Food Funct., 2019, 10, 8250–8262 RSC.
- Y. Tan, Z. Zhang, H. Zhou, H. Xiao and D. J. McClements, Food Funct., 2020, 11, 7126–7137 Search PubMed.
- C. Holland, P. Ryden, C. H. Edwards and M. M. L. Grundy, Foods, 2020, 9(2), 201 CrossRef CAS PubMed.
- A. M. B. Priyadarshani, Crit. Rev. Food Sci. Nutr., 2017, 57, 1710–1717 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |