Open Access Article
Open Access ArticleTolerance of freeze-dried autochthonous Lacticaseibacillus paracasei to food-related stress factors using by-product-based media
Marina E. Navarro†
ab,
Natalia S. Brizuela† *ab,
Liliana C. Semorilea,
Bárbara M. Bravo-Ferradaab and
Emma E. Tymczyszyn
*ab,
Liliana C. Semorilea,
Bárbara M. Bravo-Ferradaab and
Emma E. Tymczyszyn ab
ab
aLaboratorio de Microbiología Molecular, Instituto de Microbiología Básica y Aplicada (IMBA), Departamento de Ciencia y Tecnología, Universidad Nacional de Quilmes, Bernal, Argentina. E-mail: nsbrizuela@gmail.com
bConsejo Nacional de Investigaciones Científcas y Técnicas (CONICET), Godoy Cruz 2290, Argentina
First published on 30th January 2026
Abstract
The production of dried cultures of lactic acid bacteria (LAB) at low cost and in a sustainable form is relevant for the elaboration of fermented foods and beverages. Lacticaseibacillus paracasei UNQLpc10 was grown in whey permeate and beer lees (WP-BL), then freeze-dried, and its tolerance to different stress factors, low pH, salt, ethanol and lysozyme, was studied. UNQLpc10 grown in this sustainable medium was dried either with or without the same medium and maltodextrin (MD). Cell survival was assessed after freeze-drying and following inoculation under stress conditions. The recovery of viable cells was influenced by the rehydration medium, with rich nutrient media such as MRS and, to a lesser extent, WP-BL, proving necessary for effective cell repair and maintenance of viability under harsh conditions. In contrast, rehydration with physiological solution resulted in high susceptibility and a significant loss of viability. UNQLpc10 can be cultivated in by-product-based media, frozen at −20 °C in the same medium supplemented with buffer and MD (without centrifugation), freeze-dried, and later rehydrated in the same medium, demonstrating good tolerance to stress. This study highlights the potential of combining local LAB strains with agro-industrial by-products and outlines future directions, including evaluation under combined stressors and in real food matrices.
Sustainability spotlightThis study promotes sustainable biotechnology by developing a low-cost process for producing lactic acid bacteria using food industry by-products such as whey permeate and beer lees as culture media. In this formulation, whey permeate fully replaces the primary fermentable carbon source of commercial medium (MRS) and beer lees partially replace the conventional nitrogen source, substantially reducing the reliance on refined medium components. This approach valorizes agro-industrial waste, reducing environmental impact and production costs associated with conventional media. Furthermore, a simplified freeze-drying protocol at −20 °C without centrifugation minimizes energy and resource use, facilitating industrial scalability. The obtained dried cultures retain functionality under stress conditions, supporting their application in fermented foods. This work contributes to sustainable food production and aligns with the United Nations Sustainable Development Goals (SDG 9: Industry, Innovation and Infrastructure; SDG 12: Responsible Consumption and Production; and SDG 13: Climate Action). |
1. Introduction
Lacticaseibacillus paracasei is a member of the normal human and animal gut microbiota and is extensively used in the food industry as a starter culture of dairy products or as a probiotic. Strains of this species have also been isolated from a variety of fermented artisanal products such as fermented milk, cheese, sourdough bread, and fermented vegetables.1 Lcb. paracasei UNQLpc10 strain was isolated from wine in a previous work. Analysis of the UNQLpc10 genome showed the possibility of its use for several applications in the food industry due to its potential abilities to confer different characteristics of texture, flavour and pleasant aroma to fermented foods, as well as the presence of genes of tolerance to different stress factors and other relevant enzymatic activities.2Scaling up the production of lactic acid bacteria (LAB) biomass for use as starter cultures involves establishing processes that are not only suitable for industrial application but also cost-efficient and environmentally responsible. To achieve high biomass yields at an industrial level, it is crucial to develop affordable cultivation media.
Because LAB, particularly Lactobacillus species, possess limited biosynthetic capabilities, they are considered nutritionally demanding organisms.3 To support their growth, various natural substrates especially residues and by-products from the food industry have been explored as alternative culture bases.4–10 Similar valorisation strategies using plant-derived by-products have demonstrated the capacity to enhance LAB growth, prebiotic activity, and microbial resilience, as shown in fermented milk supplemented with cashew by-products.11 Nevertheless, these materials often lack essential nutrients and therefore must be enriched to enable adequate microbial proliferation. Common enrichment agents include protein compounds like yeast extract and peptone, which, despite their effectiveness, significantly raise production costs, representing approximately 30% of the total expenses.12,13
Previous studies showed that the biomass of UNQLpc10 can be obtained by cultivating them in a medium based on whey permeate (WP) supplemented with salts, Tween 80, and yeast extract (YE) obtained by sonication treatment of beer lees (dead yeast from beer production), with similar viability to that obtained in MRS (commercial medium).7
For industrial purposes, cultures must be tolerant to several stress factors, including tolerance to hydric stress during the preservation process, such as dehydration and re-hydration. These cultures are introduced in different food matrices as starters, and according to the fermentative process, LAB must be tolerant to different matrix conditions, such as, low pH, high ethanol content, high salt concentration, presence of lysozyme, etc.14–16
The growth conditions, temperature, nutrient availability, presence of sub-lethal stress factor, etc., may change the physiological state of cells, as well as the membrane lipid composition and expression of several proteins related to resistant to stress.17 Particularly, the growth in an alternative medium formulated with by-products must not only produce high biomass levels, but also must maintain their technological properties, for example, the ability to ferment under different stress factors present in food and beverages matrices.
With this background and the results previously obtained in our laboratory, the aim of this work was the optimization of freeze-drying cultures of Lcb. paracasei UNQLpc10 grown in a whey permeate based medium and yeast extract from beer lees, as a research alternative that provides an experimental framework with the potential for extrapolation to the industrial field. Cultures were freeze-dried with or without the growth medium in the presence of maltodextrin as the cryoprotectant. Survival after the freeze-drying process and subsequent rehydration under different conditions (MRS, by-product media and physiological solution) was also evaluated. Finally, bacterial resistance to different environmental stress conditions, such as high ethanol concentration, low pH, salinity and the presence of lysozyme, was analyzed for freeze-dried cultures.
2. Materials and methods
2.1 Strain information
Lacticaseibacillus paracasei UNQLpc10 (complete genome GenBank Accession code CP029686.1) was isolated from a Patagonian Pinot noir wine vintage 2014 [2].Sovereign Rights Over Natural Resources: the indigenous LAB strain mentioned above is a genetic resource belonging to the Province of Rio Negro (UNQLpc10) and was deposited in the strain collection of the Laboratorio de Microbiologia Molecular (IMBA-DCyT-UNQ-CIC) and registered with the Environment and Climate Change Secretariat—Province of Rio Negro Government. Its preservation follows the protocols of the Convention on Biological Diversity and the Nagoya Protocol, which safeguard the ownership and use of genetic resources from the regions where they were isolated (RESOL-2024-626-E-GDERNE-SAYCC#SGG, NO-2024-00628737-GDERNE-SAYCC#SGG).
2.2 Strain growth
The strain was grown in food by-product-based medium using whey permeate (WP) and yeast extract from beer lees (BL). WP obtained by drying deproteinized sweet whey contained approximately 80% w/w lactose, 6% w/w of ashes, and 3% w/w proteins, kindly supplied by Arla Foods Ingredients S.A. (Buenos Aires, Argentina). BL was obtained according to Brizuela et al.,7 Low hopping BL, kindly donated by Bierlife S.A–Sarandí–Buenos Aires –Argentina, were maintained frozen at −20 °C until use. The BL were thawed and the suspension was sonicated (ultrasonic cleaner-BioBase) for 2 h. Then, samples were sterilized in an autoclave, for 15 min at 121 °C to inactivate all enzymes and microorganism present in the sample. Then, the samples were centrifuged at 5000 g for 10 min at 4 °C and the obtained supernatant was dehydrated by freeze-drying.WP and BL were used at 5% and 1% w/v, respectively, in distilled water (WP-BL). WP-BL was supplemented with a mixture of salts as proposed by Cerdeira et al. 2025.8 Cultures were inoculated at 1% v/v of culture in stationary state (∼1 × 107 CFU mL−1) and incubated at 28 °C for 48 h under aerobic conditions to reach the stationary phase (∼5 × 109 CFU mL−1) with an OD600 of 2 (growth profile was previously published by Brizuela et al., 2025). MRS broth was used as the positive control. Cell viability was determined by bacterial colony count on MRS agar plates, incubated at 28 °C for 48 h. The data obtained were expressed as log CFU mL−1.
2.3 Freeze-drying conditions
Bacterial cells grown in the supplemented WP-BL were treated in three different ways: (A) cultures were centrifuged and then resuspended in the same volume of maltodextrin (MD) 20% w/v; (B) cultures were centrifuged and then resuspended in the same volume of physiological solution (control); (C) cultures in the stationary phase (without centrifugation) were supplemented with potassium phosphate buffer 1 M pH 7 (to neutralize the pH)10 and MD stock solution (20% w/v) in a proportion of culture:buffer:MD of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1:1, where the final buffer concentration was 0.25 M and MD 5% w/v. Condition B (PS) was only used as a control for initial freeze-drying survival and not used for subsequent stress assays because of its very low survival.
1:1, where the final buffer concentration was 0.25 M and MD 5% w/v. Condition B (PS) was only used as a control for initial freeze-drying survival and not used for subsequent stress assays because of its very low survival.
Aliquots of 1 mL of cell suspension were frozen for 48 h in glass vials at −20 °C. Samples were then dehydrated in a freeze-dryer equipment (BIOBASE freeze-dryer system/BK-FD10P, Shandong, China) for 48 h (condenser temperature: −55 °C; chamber pressure: 0.06 mbar). To assess cell survival immediately after freeze-drying, the samples were rehydrated and plate counts were performed.
2.4 Storage conditions
Freeze-dried powders were stored for 30 days at 4 °C, under vacuum conditions, in sealed packaging containing silica gel and protected from light. The samples were vacuum-packed with a Foodsaver® OSTER tumble dryer (model V204).2.5 Cellular rehydration and effect on incubation under stress conditions
Freeze-dried cultures were rehydrated in three different media: (a) MRS, (b) WP-BL and (c) physiological solution (PS) (as control). Cells were rehydrated in the same volume used prior to drying (1 mL). The rehydrated cultures were incubated at 21 °C for 24 h, a period considered sufficient for cell damage repair while preventing growth, according to Santos et al.18 and Brizuela et al.6 Cultures were then inoculated under stress conditions (pH: 3, ethanol 14% v/v, NaCl 8% w/v or lysozyme 25 mg mL−1) and later, their survival was evaluated. These stresses were evaluated in the presence or absence of nutrients. For evaluating stress in a rich medium, UNQLpc10 was inoculated in 10 mL of MRS broth with 14% v/v ethanol, MRS broth pH 3, MRS broth with 8%w/v NaCl and MRS broth with 25 mg L−1 of lysozyme. For evaluating stress in a poor medium, UNQLpc10 was inoculated in 10 mL of PS containing the same stressing conditions as in the MRS medium. The pH was adjusted to 3.0 by adding 1 M HCl, and the medium was then sterilized. For the ethanol stress condition, absolute ethanol was added after sterilization of the medium; therefore, the media (MRS or PS) were prepared with a reduced water content to account for ethanol addition. NaCl and lysozyme stock solution were sterilized by filtration and added after sterilization medium.The tolerance to stress conditions was evaluated by measuring the viability of cultures after 48 h of incubation, and aliquots of samples were plated on MRS agar in order to compare the cell survival under different conditions. Tolerance was defined as the absence of significant differences in viability relative to fresh cultures in MRS, while decreased viability was interpreted as susceptibility to the applied stress. These simplified model stress conditions were selected to isolate the effect of individual stressors, before testing combinations or real food systems in future studies.
2.6 Reproducibility of results and statistical analysis
All experiments were carried out using three independent bacterial cultures. The relative differences were reproducible, independent of the culture used. Analysis of variance (ANOVA) was carried out using the statistical program STATISTIX 8 Software (Analytical Software, Tallahassee, FL, USA). Means were compared by the Tukey or Dunnet test for multiple comparisons and the difference was considered significant when p < 0.05. Hierarchical clustering heat maps of rehydration conditions and stress medium were assayed by the web tool ClustVis (https://biit.cs.ut.ee/clustvis/).3. Results
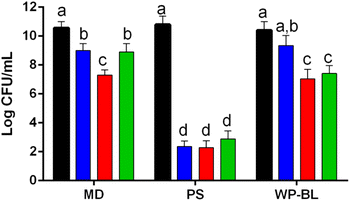
The growth of UNQLpc10 in the WP-BL media was compared with that in MRS broth, and no significant differences were observed between both cultures. The initial inoculum was 1 × 107 CFU mL−1 and reached 5 × 109 after 48 h of incubation. These results show that supplemented WP-BL demonstrates good performance in the production of cell biomass. Then, the freeze-drying of UNQLpc10 (grown in WP-BL) was studied, with or without growth media, in the presence of MD as the cryoprotectant.Fig. 1 shows the cell survival of samples that were freeze-dried (FD) under different conditions: maltodextrin (20% w/v, final pH 6.5), PS, WP-BL + phosphate buffer and MD (final pH: 7.0). Each sample was also rehydrated under three conditions: MRS, WP-BL and PS (as mentioned in the Materials and methods section).
Cultures resuspended in physiological solution showed a higher mortality rate, with a decay of 8 log units after freeze drying. In contrast, cultures resuspended in maltodextrin or freeze-dried in the culture medium (without centrifugation) showed a decrease from 1 to 3.3 log units in cell viability, showing better values when cultures were rehydrated in MRS, and worse values when rehydrated with physiological solution. Based on these results, samples resuspended in physiological solution were not used for subsequent stress assays due to their low survival rate.
When dried lactic acid bacteria are incorporated into food matrices, they often encounter specific inhibitory factors, such as acidity, salt, ethanol, and lysozyme, which strongly affect their survival and activity. In fermented sausages and cheeses, freeze-dried LAB must rapidly adapt to high salt concentrations that impose osmotic stress and reduce membrane integrity, while progressive acidification lowers intracellular pH and challenges enzyme function. In wine-related and sourdough matrices, dried LAB can be exposed to ethanol, which disrupts cell membranes and denatures proteins, particularly during rehydration when cells are the most vulnerable.19 Also, in dairy products, LAB may also encounter lysozyme, an antimicrobial enzyme intentionally added to control spoilage organisms, which compromises the bacterial cell wall by hydrolyzing peptidoglycan.20 These conditions create a highly selective environment, where LAB must survive and remain functional within such food matrices.
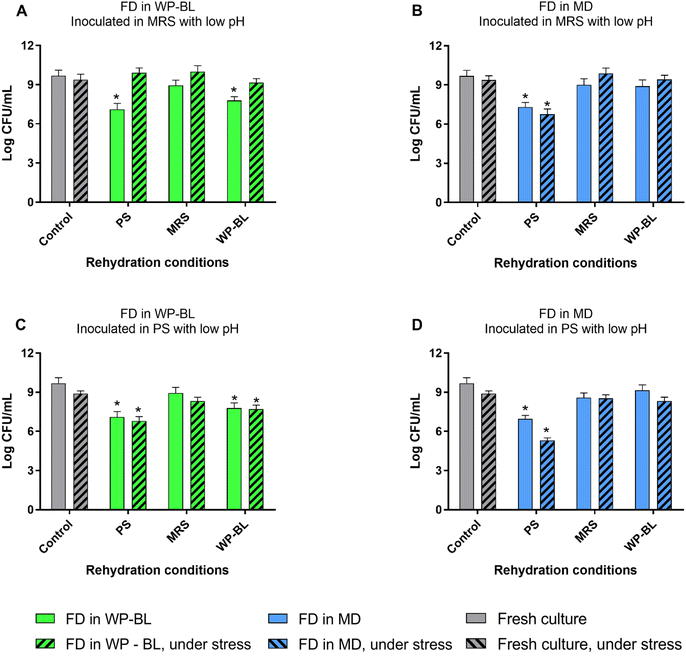
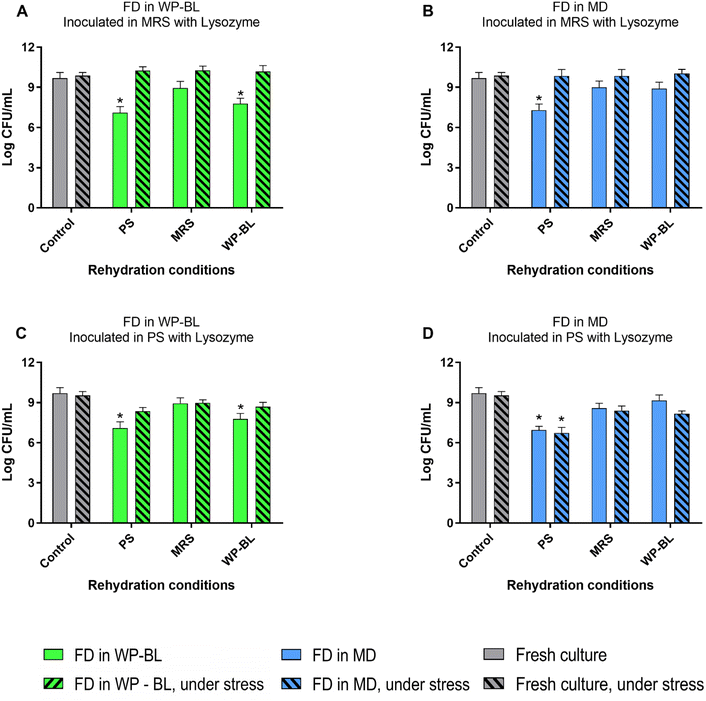
To evaluate the behaviour of freeze-dried UNQLpc10 under stress conditions, cultures obtained as shown in Fig. 1 were inoculated in MRS broth (medium rich in nutrients) or PS (medium poor in nutrients) with different types of stress, and the viability was measured after 48 h of incubation.
Fresh culture of UNQLpc10 in MRS showed to be tolerant to acid stress, as inoculation in MRS or PS with pH 3 did not show significant differences with respect to control (Fig. 2). When freeze-dried UNQLpc10 was inoculated into MRS at pH 3, comparable growth to the control was observed across all conditions, regardless of the rehydration medium (Fig. 2A and B). However, when dried cells were inoculated in PS pH 3, only cultures previously rehydrated in MRS were tolerant to acid (no significant difference respect to control), for both cultures FD in WP-BL or FD in MD.
Cells rehydrated in PS showed loss of viability under all conditions, but were less susceptible to acid when they were dried in WP-BL. However, cells rehydrated in WP-BL were more susceptible to acid when they were previously dried in WP-BL growth medium and were tolerant when dried in MD.
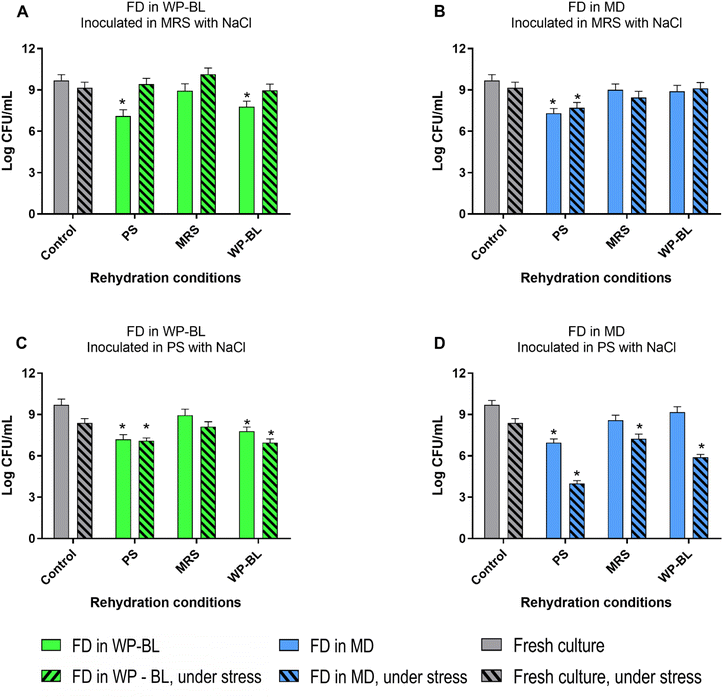
Fig. 3 shows the survival of the UNQLpc10 inoculated in two media supplemented with 8% w/v NaCl. The fresh culture of UNQLpc10 grown in MRS exhibited tolerance to salt stress in both MRS and PS media. When dried UNQLpc10 cells were rehydrated in MRS or WP-BL, similar survival levels to the fresh control were observed for cultures inoculated in MRS with NaCl. However, rehydration in PS resulted in different outcomes depending on whether the cells had been freeze-dried in WP-BL or MD. In the latter case, rehydrated cells showed increased susceptibility to salt stress, whereas drying in WP-BL improved cell viability after 48 h of incubation.
On the other hand, when rehydrated cells were inoculated into PS supplemented with NaCl, cultures freeze-dried in MD showed a significant reduction in viability across all three-rehydration media, with the most pronounced decrease observed in PS, where viability dropped by 3 log units. In contrast, cultures freeze-dried in WP-BL and rehydrated in MRS exhibited the highest tolerance to stress, with no significant differences compared to the fresh control.
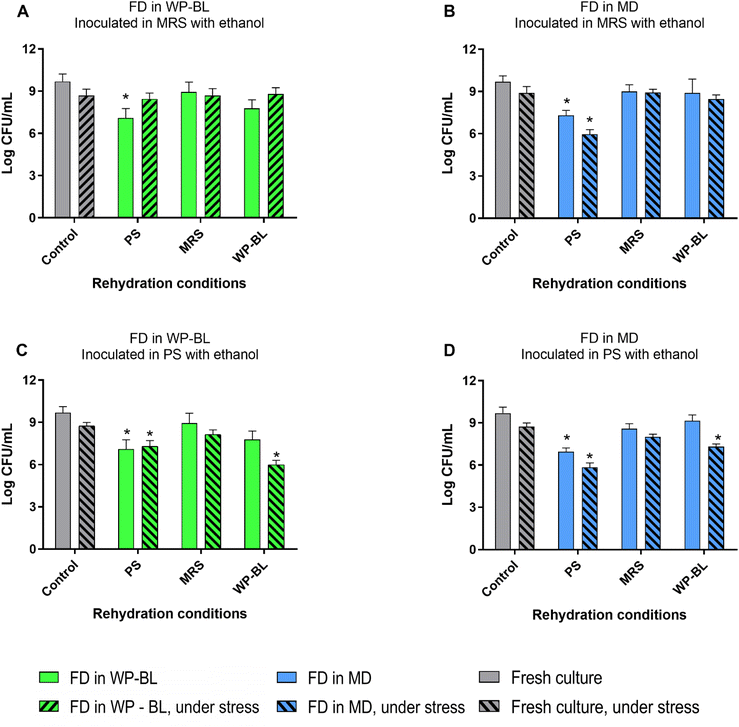
Fig. 4 shows the survival of the UNQLpc10 inoculated in two media with ethanol 14% v/v. Fresh culture of UNQLpc10 in MRS showed to be tolerant to inoculation in medium with ethanol 14% v/v, for both MRS and PS medium. When dried UNQLpc10 cells were rehydrated in MRS or WP-BL, their behaviour was similar to that of the fresh control. However, rehydration in PS led to different outcomes, particularly for cultures that had been freeze-dried in WP-BL or MD. In the latter case, the rehydrated cells were susceptible to ethanol, whereas those dried in WP-BL exhibited greater stress tolerance. In contrast, cells dried in MD may lack such protective effects, leading to lower recovery, as observed in Fig. 3. On the other hand, when rehydrated cells were inoculated in PS with ethanol, only cultures rehydrated in MRS were able to tolerate stress, without significant difference to fresh control, indicating that only under these conditions, the cells were able to repair the damage.
Lysozyme is a lytic enzyme used as GRAS food preservative in several countries; in Argentina, the use of 25 mg L−1 in milk is allowed. For this reason, we evaluated the cell survival of UNQLpc10 in MRS medium or physiological solution supplemented with lysozyme at a concentration of 25 mg L−1 (Fig. 5). Both fresh and freeze-dried UNQLpc10 cultures exhibited tolerance to lysozyme in MRS and PS media. Only the culture freeze-dried with MD and rehydrated in PS showed reduced viability both before and after lysozyme inoculation in PS; however, the presence of lysozyme itself did not result in cell death.
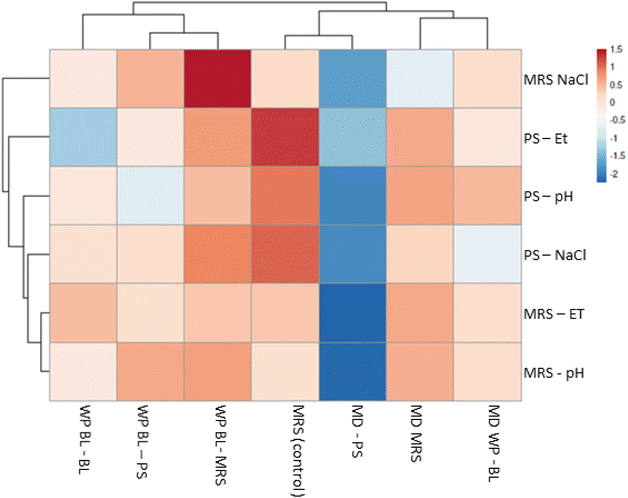
Finally, to summarize the effects of rehydration and inoculation conditions under different stress factors, a multivariate analysis was performed using data from low pH, salt, and ethanol treatments (Fig. 6). Lysozyme was excluded from the analysis, as UNQLpc10 did not show susceptibility to this compound under the conditions tested in the present study, but the effect of lysozyme in combination with other stress factor present in the food matrix could be considered in future studies. The ClustVis heat map clearly showed that the fresh culture (MRS) was able to tolerate ethanol, salt, and acid stress in both media. In contrast, cultures freeze-dried in MD and rehydrated in PS (MD–PS) exhibited a marked decrease in viability across all stress conditions.
For cultures freeze-dried in MD, the choice of rehydration medium prior to stress exposure appeared to be a critical factor. In some cases, rehydration with WP-BL yielded good viability results and could represent a cost-effective strategy. In contrast, freeze-drying in WP-BL did not result in major differences among the rehydration media tested. Under this condition, the presence of residual growth medium and added phosphate buffer during rehydration may have contributed to pre-adapting UNQLpc10 cells to adverse conditions.
Regarding the inoculation conditions, low pH and ethanol stress in MRS appeared to be better tolerated by dried cells (except for those freeze-dried in MD and rehydrated in PS). For salt stress, cultures freeze-dried in WP-BL showed the best performance. However, under ethanol and low pH stress in PS, freeze-drying in MD (followed by rehydration in MRS or WP-BL) resulted in slightly better cell viability than cultures dried in WP-BL.
4. Discussion
The commercialization of LAB starter cultures produced at low cost using by-product-based media and with proven efficacy in fermented foods also depends on the preservation technologies employed, which are essential to ensure long-term culture stability in terms of viability and functionality.21 Ideally, these cultures should be available in a dehydrated form with demonstrated activity for direct inoculation into the fermentation process.Freeze-drying is the most commonly used preservation method for LAB; however, it can cause cellular damage and death due to reduced water activity.22–24 Therefore, optimizing dehydration and rehydration conditions is crucial to maintain the technological properties of the dried cultures. The success of the preservation process is influenced by several factors, including the physiological state of the microorganisms (determined by the growth medium and culture conditions), the selection of protective agents, and the storage conditions.10,23–25 UNQLpc10 has shown resistance under wine-related stress conditions, and its whole-genome analysis revealed the presence of several genes associated with stress adaptation.2
Regarding protective agents, various compounds such as polymers, sugars, and amino acids are commonly added prior to the freeze-drying process to minimize cellular stress and enhance the tolerance of LAB to dehydration.23,26–28 In this study, maltodextrin was selected as the protective agent for freeze-drying, as it has demonstrated strong cryoprotective effects in several lactic acid bacteria.6,29–31 Maltodextrin offers several practical advantages, including high solubility in aqueous solutions, low viscosity, and low cost.32 Its protective effect was evident in the strain under study, where cell survival was up to six orders of magnitude higher (∼109 CFU mL−1) compared to the control condition using physiological solution (Fig. 1). While maltodextrin has been widely reported as a cryoprotectant, our contribution shows its effectiveness in combination with culture growth in by-product media and a simplified FD protocol (direct culture frozen at −20 °C and without cells, centrifugation for washing and concentration) applied to an autochthonous strain with technological potential.
Survival after FD is significantly higher when the cryoprotective medium is neutralized.10 Some authors have reported the correlation between the decrease in pH and the decrease in bacterial survival after a freeze-drying treatment. In the study by Hua et al. 2009,33 an oenological strain of the O. oeni species was tested and it was observed that the acid stress of the cryoprotective medium produced a decrease in cell viability after the freeze-drying treatment and a change in the concentration of membrane fatty acids. In the study by Schoug et al. 2008,34 their results showed a negative effect on the freeze-drying tolerance of Lb. coryniformis Si3 following growth under acidic conditions, as well as after acid stress applied in the late exponential phase. Therefore, the pH of the suspension medium should be adjusted to fall within the range of 5.5 to 7.0. In this study, we demonstrated that cultures can be FD without the need for a centrifugation step. This approach may reduce both the cost and preparation time of cultures intended for dehydration. In addition, freezing was performed at −20 °C, a condition that is more feasible for industrial-scale applications than freezing at −80 °C. Under these conditions, maltodextrin and phosphate buffer were added to the WP-BL culture.
A good recovery of cell viability was observed when cultures were rehydrated and plated on a rich medium such as MRS, but, a slight difference in survival was noted depending on the rehydration medium used (Fig. 1).
The damage and subsequent recovery of microorganisms following the freeze-drying process is a complex phenomenon that requires further investigation to fully understand the underlying cellular restoration mechanisms. Across all stress conditions evaluated (low pH, salt, ethanol, and lysozyme), a consistent pattern emerged underscoring the central role of the rehydration medium in post-freeze-drying performance. Based on the results presented here, it can be inferred that certain rehydration conditions—such as the presence of sugar-rich media and room temperature—favour the recovery of a high number of viable cells after inoculation in different types of stress, in contrast to results obtained for cultures rehydrated with PS (Fig. 2–5). However, cells dried in WP-BL were less susceptible to stress factors when rehydrated in PS. This may be explained by the fact that WP-BL includes components of the growth medium, which could facilitate the repair of cell damage upon rehydration, compared to cells dried only in the presence of MD. In addition to supplying nutrients, WP-BL may provide functional compounds such as complex carbohydrates and other bioactive constituents derived from dairy and brewery byproducts, which can facilitate membrane repair and physiological recovery during rehydration. This functional contribution may explain the intermediate protective effect of WP-BL compared to MRS, particularly under stress conditions, supporting its potential use as a sustainable rehydration medium. The protective role of cereal and brewery by-products in enhancing LAB recovery after stress was also demonstrated for brewer's spent grain, which improved the post-stress viability of probiotic strains in fermented milk.35 It is possible that incubation in a nutrient-rich medium promotes the complete restoration of cellular macro-molecules, particularly the cytoplasmatic membrane.18 The recovery of membrane barrier properties may enhance the cell's ability to survive in hostile environments, such as low pH, high concentrations of salt, ethanol and the presence of lysozyme.19 These findings highlight that rehydration represents a critical and often underestimated step that should be standardized and explicitly provided by starter culture suppliers, and that rehydration in water or physiological solution alone should be avoided, especially prior to inoculation into harsh food matrices. Importantly, these effects reflect differences in cell survival rather than growth, as rehydration conditions were designed to allow cellular repair while preventing proliferation. The functional contribution of food-industry residues to microbial physiology has also been demonstrated in probiotic dairy systems, where cashew by-products improved microbial performance and fermentative potential.36 Further studies are required to corroborate cell membrane repair during rehydration.
Lysozyme is a lytic enzyme used as a GRAS food preservative in countries such as the USA, Mexico37 and Argentina (Argentine Food Code- Chapter VIII), and European Union and it is considered GRAS [30]. The results obtained in this work are consistent with those reported for Lcb. casei Shirota38 and by variousLpb. plantarum strains,15,39 where LAB are tolerant to this compound. In this work, we corroborate that that freeze-dried UNQLpc10 was not affected by the presence of lysozyme and could be added in different fermentation processes, such as cheese production, without significant differences with the fresh culture.
In food systems, pH is a key factor influencing the growth of most microorganisms. For this reason, acidity has long been used as an effective strategy to control pathogenic microbes in various food matrices.40 Lactic acid bacteria exhibit valuable technological properties when used as starter cultures in the food industry, including their ability to lower the pH of the product and thereby create an acidic environment that imposes stress on undesirable microorganisms. In this group of foods, we can find drinks such as boza, fruit juices, and dairy products such as yogurts, kimchi, cheeses and wine, among others.41–45 In wine and other alcoholic beverages, LAB produce some physicochemical transformations, called malolactic fermentation (MLF), which reduces wine acidity and improves the aromatic characteristics of the product. In these fermented beverages, the main stress factor for LAB is the presence of ethanol.46 For this reason, the tolerance to ethanol stress is crucial for dried malolactic starter cultures. In a previous work, we evaluated the performance of this strain when cultured in MRS medium and inoculated into wine-like medium at pH 3.6, containing 10% and 14% ethanol.2 In forthcoming experiments, the same parameters will be assessed for the UNQLpc10 grown in the proposed alternative medium, incorporating freeze-drying and rehydration steps, to determine whether its performance is comparable to that observed under traditional commercial culture conditions.
In the case of salt stress, the increase in the osmotic pressure (decrease in water activity) of food matrices is among one of the most used methods to preserve food products. Osmotic stress and acid stress represent the main stress faced by LAB during the fermentation of many food products such as cheeses, sauerkraut, or kimchi.47–50 Although in this study we evaluated selected stress conditions, including the presence of lysozyme, low pH, and increased salinity, future research could incorporate additional factors to more closely simulate specific food matrices. For example, a cheese-like medium with combined stress conditions involving acidification, salt gradients, and ripening temperatures would provide a more realistic assessment of bacterial performance and technological robustness under actual cheese production and maturation conditions.
Rehydration must ensure that bacteria are able to repair cellular damage and reach optimal physiological conditions, especially before being inoculated into food or beverage matrices containing some stress factors. This is particularly important when microorganisms must survive adverse environmental conditions and express their physiological and technological potential. As demonstrated by Lee et al.,28 rehydration is a critical step in the successful recovery of freeze-dried microorganisms used as starter cultures. In a previous work, it was demonstrated that the rehydration of a strain of Lpb. plantarum in a rich medium (such as MRS) was necessary for survival and malolactic activity in wine.6 The costs of MRS warrant the search for nutritious, sustainable and low-cost supplements that improve the physiological conditions of dehydrated crops and optimize the required conditions (time, temperature, type of nutrient, and osmolarity), such as the formulated WP-BL medium in this work.
From an industrial perspective, Fig. 6 shows rehydration with PS or water should be avoided. Instead, specific supplements and rehydration conditions should be selected and provided by commercial starter culture suppliers for each particular food or beverage fermentation process. Although we tested individual stressors, real food matrices often involve combinations. This limitation will be addressed in follow-up studies by evaluating additive and synergistic effects of multiple stressors. Further experiments are needed to identify which components of the MRS or WP-BL media are critical for cell damage repair, as well as to elucidate the kinetics of this process in order to determine the optimal rehydration time for different LAB strains according to their intended applications.
5 Conclusions
The optimization of drying/dehydration and rehydration processes for lactic acid bacteria used as starter cultures in food and beverage fermentations is essential to enable their practical, efficient application in the industry. Additionally, sustainable and cost-effective production methods are required. Certain food industry by-products represent promising nutrient sources, and their potential use adds value to environmentally problematic waste due to their high biochemical oxygen demand.Our research group has been working for several years on the preservation and formulation of starter cultures for food fermentations. In previous studies, our laboratory optimized various aspects of the freeze-drying process, including the use of different cryoprotectants, freezing temperatures, and storage conditions.
In this study, UNQLpc10 was grown in whey permeate and supplemented with yeast extract derived from beer lees, salts, and Tween 80, showing good survival rates after freeze-drying in the presence of maltodextrin as a cryoprotectant. Furthermore, it was demonstrated that the UNQLpc10 culture can be dehydrated without prior centrifugation or pellet washing, by neutralizing the final pH through the addition of potassium phosphate. This approach reduces both preparation time and equipment costs.
On the other hand, although the evaluated storage period is limited, the viability results obtained after one month provide a reliable indication of the bacteria's initial behaviour under controlled storage conditions.
Finally, the tolerance of LAB (grown in economical media, dried and stored) to inoculation in media with different types of stress was analysed, thus evaluating its potential use in the fermented food industry. This work lays the foundations for obtaining sustainable and economically competitive nutritional media, which are optimal for increasing biomass production, as well as for the rehydration of freeze-dried cultures of LAB. Also, the replacement of conventional culture media for the growth of LAB, prepared at low cost, easy to use and proven fermentative efficiency in foods, appears as a valuable strategic tool to provide greater added value to by-products of the food industry. As a first step, this study validates the feasibility of producing freeze-dried autochthonous LAB in by-product-based media. Future work will extend this approach to multiple strains, combined stress factors, and real food matrices in order to further strengthen its industrial applicability.
Author contributions
Formal analysis and investigation: N. S. B., M. E. N., B. B. F and E. E. T.; methodology: M. E. N., N. S. B., B. B. F.; project administration: L. S. and E. T.; writing – original draft: B. B. F. and E. T.; writing – review & editing: M. E. N., N. S. B., B. B. F., and E. T.Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article, including viability obtained in all conditions assayed, are available at the institutional CONICET repository under the name Navarro, Marina Edith; Tymczyszyn, Emma Elizabeth; Bravo Ferrada, Barbara Mercedes; Brizuela, Natalia Soledad; (2025): Tolerancia a factores de estrés del crecimiento de UNQLpc10 liofilizado en un medio basado en subproductos. Consejo Nacional de Investigaciones Científicas y Técnicas at (dataset): http://hdl.handle.net/11336/267702Acknowledgements
N. S. B., B. M. B–F. and E. E. T. are members of the Research Career of the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), M.E.N is a fellow of CONICET (PhD student), L. S. is a member of the Research Career of the CIC-PBA.Notes and references
- S. S. Chiang and T. M. Pan, Beneficial effects of Lactobacillus paracasei subsp. paracasei NTU 101 and its fermented products, Appl. Microbiol. Biotechnol., 2011, 933(93), 903–916 Search PubMed.
- N. G. Iglesias, M. E. Navarro, N. S. Brizuela, D. Valdés La Hens, L. C. Semorile, E. E. Tymczyszyn and B. M. Bravo Ferrada, Analysis of the Genome Architecture of Lacticaseibacillus paracasei UNQLpc 10, a Strain with Oenological Potential as a Malolactic Starter, Fermentation, 2022, 8, 726 Search PubMed.
- S. A. Hayek and S. A. Ibrahim, Current Limitations and Challenges with Lactic Acid Bacteria: A Review, Food Nutr. Sci., 2013, 04, 73–87 CAS.
- A. Ahmad, F. Banat and H. Taher, A review on the lactic acid fermentation from low-cost renewable materials: Recent developments and challenges, Environ. Technol. Innov., 2020, 20, 101138 Search PubMed.
- J. A. Vázquez, A. I. Durán, A. Menduíña, M. Nogueira, A. M. Gomes, J. Antunes, A. C. Freitas, E. Dagá, P. Dagá and J. Valcarcel, Bioconversion of Fish Discards through the Production of Lactic Acid Bacteria and Metabolites: Sustainable Application of Fish Peptones in Nutritive Fermentation Media, Foods, 2020, 9(9), 1239 CrossRef PubMed.
- N. S. Brizuela, M. Arnez-Arancibia, L. Semorile, B. M. Bravo-Ferrada and E. E. Tymczyszyn, Whey permeate as a substrate for the production of freeze-dried Lactiplantibacillus plantarum to be used as a malolactic starter culture, World J. Microbiol. Biotechnol., 2021, 377(37), 1–12 Search PubMed.
- N. S. Brizuela, M. E. Navarro, G. Rivas, G. Gómez, C. Pérez, L. Semorile, E. E. Tymczyszyn and B. Bravo-Ferrada, Optimization of the Treatment of Beer Lees for Their Use in Sustainable Biomass Production of Lactic Acid Bacteria, Appl. Microbiol., 2025, 5(2), 51 Search PubMed.
- V. Cerdeira, B. M. Bravo-Ferrada, L. Semorile and E. Tymczyszyn, Design of a low-cost culture medium based in whey permeate for biomass production of enological Lactobacillus plantarum strains, Biotechnol. Prog., 2019, 35, e2791 Search PubMed.
- V. Cerdeira, N. S. Brizuela, B. M. Bravo-Ferrada, D. Valdés La Hens, A. Caballero, L. C. Semorile and E. Tymczyszyn, Use of Apple pomace as substrate for production of Lactiplantibacillus plantarum as malolactic starter cultures, Fermentation, 2021, 7, 244 CrossRef CAS.
- M. E. Navarro, N. S. Brizuela, N. E. Flores, M. Morales, L. C. Semorile, D. Valdes La Hens, A. C. Caballero, B. M. Bravo-Ferrada and E. E. Tymczyszyn, Preservation of Malolactic Starters of Lactiplantibacillus plantarum Strains Obtained by Solid-State Fermentation on Apple Pomace, Beverages, 2024, 10, 52 CrossRef CAS.
- M. E. Herkenhoff, I. U. D. de Medeiros, L. H. G. Garutti, M. K. Salgaço, K. Sivieri and S. M. I. Saad, Prebiotic and probiotic potential of fermented milk with cashew (Anacardium occidentale) by-products evaluated in microbiome model, Food Biosci., 2024, 61, 104669 CrossRef CAS.
- G. Bustos, A. B. Moldes, J. M. Cruz and J. M. Domínguez, Evaluation of Vinification Lees as a General Medium for Lactobacillus Strains, J. Agric. Food Chem., 2004, 52, 5233–5239 CrossRef CAS PubMed.
- J. Pejin, M. Radosavljević, S. Kocić-Tanackov, R. Marković, A. Djukić-Vuković and L. Mojović, Use of spent brewer's yeast in L-(+) lactic acid fermentation, J. Inst. Brew., 2019, 125, 357–363 CrossRef CAS.
- A. Reale, T. Di Renzo, F. Rossi, T. Zotta, L. Iacumin, M. Preziuso, E. Parente, E. Sorrentino and R. Coppola, Tolerance of Lactobacillus casei,Lactobacillus paracasei and Lactobacillus rhamnosus strains to stress factors encountered in food processing and in the gastro-intestinal tract, LWT--Food Sci. Technol., 2015, 60, 721–728 CrossRef CAS.
- B. M. Bravo-Ferrada, A. Hollmann, L. Delfederico, D. Valdés La Hens, A. Caballero and L. Semorile, Patagonian red wines: selection of Lactobacillus plantarum isolates as potential starter cultures for malolactic fermentation, World J. Microbiol. Biotechnol., 2013, 29, 1537–1549 CrossRef CAS PubMed.
- B. M. Bravo-Ferrada, A. Hollmann, N. S. Brizuela, D. V. La Hens, E. E. Tymczyszyn and L. Semorile, Growth and consumption of l-malic acid in wine-like medium by acclimated and non-acclimated cultures of Patagonian Oenococcus oeni strains, Folia Microbiol., 2016, 61(5), 365–373 CrossRef CAS PubMed.
- X. Gao, J. Kong, H. Zhu, B. Mao, S. Cui and J. Zhao, Lactobacillus,Bifidobacterium and Lactococcus response to environmental stress: Mechanisms and application of cross-protection to improve resistance against freeze-drying, J. Appl. Microbiol., 2022, 132, 802–821 CrossRef CAS PubMed.
- M. I. Santos, C. Araujo-Andrade, E. Esparza-Ibarra, E. Tymczyszyn and A. Gómez-Zavaglia, Galacto-oligosaccharides and lactulose as protectants against desiccation of Lactobacillus delbrueckii subsp. bulcaricus, Biotechnol. Prog., 2014, 30, 1231–1238 CrossRef CAS PubMed.
- B. Sionek, A. Szydłowska, M. Trząskowska and D. Kołożyn-Krajewska, The Impact of Physicochemical Conditions on Lactic Acid Bacteria Survival in Food Products, Ferment, 2024, 10, 298 CrossRef CAS.
- N. Khorshidian, E. Khanniri, M. R. Koushki, S. Sohrabvandi and M. Yousefi, An Overview of Antimicrobial Activity of Lysozyme and Its Functionality in Cheese, Front. Nutr., 2022, 9, 833618 CrossRef PubMed.
- A. S. Carvalho, J. Silva, P. Ho, P. Teixeira, F. X. Malcata and P. Gibbs, Relevant factors for the preparation of freeze-dried lactic acid bacteria, Int. Dairy J., 2004, 14, 835–847 CrossRef CAS.
- S. Ambros, F. Hofer and U. Kulozik, Protective effect of sugars on storage stability of microwave freeze-dried and freeze-dried Lactobacillus paracasei F19, J. Appl. Microbiol., 2018, 125, 1128–1136 CrossRef CAS PubMed.
- B. M. Bravo-Ferrada, N. S. Brizuela, E. Gerbino, A. Gómez-Zavaglia, L. Semorile and E. E. Tymczyszyn, Effect of protective agents and previous acclimation on ethanol resistance of frozen and freeze-dried Lactobacillus plantarum strains, Cryobiology, 2015, 71, 522–528 CrossRef CAS PubMed.
- B. M. Bravo-Ferrada, S. Gonçalves, L. Semorile, N. C. Santos, N. S. Brizuela, E. Tymczyszyn and A. Hollmann, Cell surface damage and morphological changes in Oenococcus oeni after freeze-drying and incubation in synthetic wine, Cryobiology, 2018, 85, 15–21 Search PubMed.
- C. Santivarangkna, B. Higl and P. Foerst, Protection mechanisms of sugars during different stages of preparation process of dried lactic acid starter cultures, Food Microbiol., 2008, 25, 429–441 CrossRef CAS PubMed.
- R. Cárcoba and A. Rodríguez, Influence of cryoprotectants on the viability and acidifying activity of frozen and freeze-dried cells of the novel starter strain Lactococcus lactis ssp. lactis CECT 5180, Eur. Food Res. Technol., 2000, 211, 433–437 CrossRef.
- A. S. Carvalho, J. Silva, P. Ho, P. Teixeira, F. X. Malcata and P. Gibbs, Survival of freeze-dried Lactobacillus plantarum and Lactobacillus rhamnosus during storage in the presence of protectants, Biotechnol. Lett., 2002, 24, 1587–1591 CrossRef CAS.
- S. B. Lee, D. H. Kim and H. D. Park, Effects of protectant and rehydration conditions on the survival rate and malolactic fermentation efficiency of freeze-dried Lactobacillus plantarum JH287, Appl. Microbiol. Biotechnol., 2016, 100, 7853–7863 CrossRef CAS PubMed.
- N. Sosa, E. Gerbino, M. A. Golowczyc, C. Schebor, A. Gómez-Zavaglia and E. E. Tymczyszyn, Effect of Galacto-Oligosaccharides: Maltodextrin Matrices on the Recovery of Lactobacillus plantarum after Spray-Drying, Front. Microbiol., 2016, 7, 584 Search PubMed.
- V. Sanchez, R. Baeza, M. V. Galmarini, M. C. Zamora and J. Chirife, Freeze-Drying Encapsulation of Red Wine Polyphenols in an Amorphous Matrix of Maltodextrin, Food Bioprocess Technol., 2013, 6, 1350–1354 Search PubMed.
- A. Sohail, M. S. Turner, A. Coombes and B. Bhandari, The Viability of Lactobacillus rhamnosus GG and Lactobacillus acidophilus NCFM Following Double Encapsulation in Alginate and Maltodextrin, Food Bioprocess Technol., 2013, 6, 2763–2769 Search PubMed.
- A. Madene, M. Jacquot, J. Scher and S. Desobry, Flavour encapsulation and controlled release – a review, Int. J. Food Sci. Technol., 2006, 41, 1–21 CrossRef CAS.
- L. Hua, Z. WenYing, W. Hua, L. ZhongChao and W. AiLian, Influence of culture pH on freeze-drying viability of Oenococcus oeni and its relationship with fatty acid composition, Food Bioprod. Process., 2009, 87(1), 56–61 CrossRef.
- Å. Schoug, J. Fischer, H. J. Heipieper, J. Schnürer and S. Håkansson, Impact of fermentation pH and temperature on freeze-drying survival and membrane lipid composition of Lactobacillus coryniformis Si3, J. Ind. Microbiol. Biotechnol., 2008, 35, 175–181 CrossRef PubMed.
- C. Battistini, M. E. Herkenhoff, M. de S. Leite, A. D. S. Vieira, R. Bedani and S. M. I. Saad, Brewer's Spent Grain Enhanced the Recovery of Potential Probiotic Strains in Fermented Milk After Exposure to In Vitro-Simulated Gastrointestinal Conditions, Probiotics Antimicrob. Proteins, 2023, 15, 326–337 CrossRef CAS PubMed.
- M. E. Herkenhoff, I. U. D. de Medeiros, L. H. G. Garutti, M. K. Salgaço, K. Sivieri and S. M. I. Saad, Cashew By-Product as a Functional Substrate for the Development of Probiotic Fermented Milk, Foods, 2023, 12(18), 3383 CrossRef CAS PubMed.
- P. M. Davidson, F. J. Critzer and T. Matthew Taylor, Naturally occurring antimicrobials for minimally processed foods, Annu. Rev. Food Sci. Technol., 2013, 4, 163–190 CrossRef CAS PubMed.
- E. Rodríguez, J. L. Arqués, R. Rodríguez, Á. Peirotén, J. M. Landete and M. Medina, Antimicrobial properties of probiotic strains isolated from breast-fed infants, J. Funct.Foods, 2012, 4, 542–551 CrossRef.
- M. Zago, M. E. Fornasari, D. Carminati, P. Burns, V. Suàrez, G. Vinderola, J. Reinheimer and G. Giraffa, Characterization and probiotic potential of Lactobacillus plantarum strains isolated from cheeses, Food Microbiol., 2011, 28, 1033–1040 Search PubMed.
- S. A. Ibrahim, R. D. Ayivi, T. Zimmerman, S. A. Siddiqui, A. B. Altemimi, H. Fidan, T. Esatbeyoglu and R. V. Bakhshayesh, Lactic Acid Bacteria as Antimicrobial Agents: Food Safety and Microbial Food Spoilage Prevention, Foods, 2021, 10, 3131 CrossRef CAS PubMed.
- A. Botes, S. D. Todorov, J. W. von Mollendorff, A. Botha and L. M. T. Dicks, Identification of lactic acid bacteria and yeast from boza, Process Biochem., 2007, 42, 267–270 CrossRef CAS.
- M. Naeem, M. Ilyas, S. Haider, S. Baig and M. Saleem, Isolation characterization and identification of lactic acid bacteria from fruit juices and their efficacy against antibiotics, Pakistan J. Bot., 2012, 44(8), 323–328 CAS.
- J. M. Park, J. H. Shin, D. W. Lee, J. C. Song, H. J. Suh, U. J. Chang and J. M. Kim, Identification of the lactic acid bacteria in kimchi according to initial and over-ripened fermentation using PCR and 16S rRNA gene sequence analysis, Food Sci. Biotechnol., 2010, 19, 541–546 CrossRef CAS.
- L. González, H. Sandoval, N. Sacristán, J. M. Castro, J. M. Fresno and M. E. Tornadijo, Identification of lactic acid bacteria isolated from Genestoso cheese throughout ripening and study of their antimicrobial activity, Food Control, 2007, 18, 716–722 CrossRef.
- R. Medina, M. Katz, S. Gonzalez and G. Oliver, Characterization of the Lactic Acid Bacteria in Ewe's Milk and Cheese from Northwest Argentina, J. Food Prot., 2001, 64, 559–563 CrossRef CAS PubMed.
- K. M. Sumby, J. Niimi, A. L. Betteridge and V. Jiranek, Ethanol-tolerant lactic acid bacteria strains as a basis for efficient malolactic fermentation in wine: evaluation of experimentally evolved lactic acid bacteria and winery isolates, Aust. J. Grape Wine Res., 2019, 25, 404–413 Search PubMed.
- T. Guinee and P. F. Fox, Cheese: Chemistry, Physics and Microbiology, Vol. 2, General Aspects, 3rd edn, 2004 Search PubMed.
- L. Mayorga-Reyes, P. Bustamante-Camilo, A. Gutiérrez-Nava, E. Barranco-Florido and A. Azaola-Espinosa, Growth, survival and adaptation of Bifidobacterium infantis to acidic conditions, Rev. Mex. Ing. Quím., 2009, 8, 259–264 CAS.
- D. I. Serrazanetti, D. Gottardi, C. Montanari and A. Gianotti, Dynamic stresses of lactic acid bacteria associated to fermentation processes, in Lactic Acid Bacteria-R & D for Food, Health and Livestock Purposes, IntechOpen, 2013 Search PubMed.
- S. Koch, G. Oberson, E. Eugster-Meier, L. Meile and C. Lacroix, Osmotic stress induced by salt increases cell yield, autolytic activity, and survival of lyophilization of Lactobacillus delbrueckii subsp. lactis, Int. J. Food Microbiol., 2007, 117, 36–42 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |