Open Access Article
Open Access ArticleStatic biological aging as a sustainable method for low-alcohol sherry wines: impact on composition and typicity
Lourdes
Vega-Espinar
a,
Raquel
Muñoz-Castells
 b,
Victor
Palacios
a,
Juan
Moreno
b and
Cristina
Lasanta
b,
Victor
Palacios
a,
Juan
Moreno
b and
Cristina
Lasanta
 *a
*a
aDepartment of Chemical Engineering and Food Technology, University of Cádiz, Agrifood Campus of International Excellence (ceiA3), Wine and Agri-Food Research Institute (IVAGRO), Puerto Real, Cádiz 11510, Spain. E-mail: cristina.lasanta@uca.es; lourdes.espinar@uca.es
bDepartment of Agricultural Chemistry, Edaphology and Microbiology, Marie Curie (C3) and Severo Ochoa (C6) Buildings, Agrifood Campus of International Excellence (ceiA3), University of Córdoba, Ctra. N-IV-A, km396, Cordoba, 14014, Spain. E-mail: b52mucar@uco.es; qe1movij@uco.es
First published on 29th December 2025
Abstract
This study evaluated static biological aging as a sustainable alternative to conventional dynamic aging for producing low-alcohol sherry wines. We investigated the feasibility of using a reduced alcohol strength, addressing market trends for lower-alcohol beverages while assessing the impact on chemical composition and sensory typicity. Wines from two Jerez wineries were statically aged for 12 months, comparing a low-alcohol test group (13–14% v/v) against a traditional-alcohol control (15–15.5% v/v). Results demonstrated that static aging at lower alcohol content significantly enhanced flor yeast metabolism. This was evidenced by the accelerated consumption of key substrates (ethanol, glycerol, volatile acidity) and amplified production of key biological markers, such as acetaldehyde and acetoin, compared to the control wines. Sensory analysis validated these chemical findings, confirming that the reduced-alcohol wines met all quality parameters and exhibited an enhanced characteristic aroma. Results suggest that specific outcomes may vary depending on the distinct microclimatic conditions and characteristic yeast strains inherent to each winery, warranting further research to optimize operational conditions. We conclude that static biological aging at reduced strength is a viable strategy for producing lower-alcohol Fino-style wines that align with consumer trends and offer substantial economic and sustainability benefits, including lower fortification costs and reduced greenhouse gas emissions (GHG), supporting more sustainable enological practices.
Sustainability spotlightThe use of static biological aging to elaborate low-alcohol sherry wines represents an important innovation in sustainable winemaking, shifting the paradigm from an energy intensive process to one of inherent efficiency. The core sustainability advantage of this method lies in its direct challenge to conventional, resource-heavy practices. By demonstrating that high-quality biological aging is achievable at a lower alcohol level (13-14% v/v), this process reduces the need for fortification. This reduction is not merely an economic saving; it is a significant environmental victory. It slashes the associated energy consumption and Greenhouse Gas (GHG) emissions typically required to produce, transport, and integrate the fortifying alcohol. This method effectively decouples quality wine production from a high carbon footprint. This process innovation contributes directly to the United Nations Sustainable Development Goals (SDGs). By championing resource efficiency (less alcohol, less energy) and demonstrably lowering emissions, it is a clear and actionable example of goal 12 (responsible consumption and production) and goal 13 (climate action). It provides a viable pathway for the wine industry to reduce its environmental impact while simultaneously aligning with modern consumer trends. |
1. Introduction
Biological aging is an oenological practice traditionally established in southern Spain, where regions such as Jerez, Sanlúcar de Barrameda, Montilla-Moriles, Condado de Huelva, Málaga, and Lebrija have refined this unique method for centuries. Similar biological aging processes are also used in other global wine regions, such as for “Vin jaune” in France,1 Szamorodni in Hungary2 and Vernaccia di Oristano in Italy.3Biological aging is a microbiological process based on the spontaneous growth of film-forming yeasts, known as the ‘veil of flor’, which develop on the wine's surface.4,5 The development and maintenance of this yeast biofilm require specific environmental conditions of temperature and humidity.6 According to various studies, the yeasts that make up the veil of flor tend to belong mostly to the species Saccharomyces cerevisiae,6 including four main races: beticus, cheresiensis, motuliensis and rouxii (also known as Zygosaccharomyces rouxii).7–9 However, nowadays modern molecular techniques have made it possible to identify other species of yeast non-Saccharomyces.10–12 Veil of flor yeasts perform an aerobic metabolism characterized primarily by the consumption of oxygen, ethanol, and glycerol, although other substrates, such as acetic acid, lactic acid, and ethyl acetate, may also be metabolized.5,13 This metabolic activity generates key volatile compounds, including acetaldehyde, higher alcohols, acetoin, and 2,3-butanediol,14,15 which confer the unique sensory characteristics typical of these wines, marked by pungent notes, nutty aromas, and yeast-derived flavours.16,17
Wines produced under the Protected Designations of Origin (PDO) ‘Jerez-Xérès-Sherry’ and ‘Manzanilla-Sanlúcar’, specifically Fino and Manzanilla, are obtained from the Palomino Fino grape variety. Following alcoholic fermentation, the base wine is fortified with wine alcohol to reach 15–15.5% v/v before starting the biological aging stage. The wine is then aged in 600 litre American oak (Quercus alba) casks, which are intentionally filled to five-sixths of their capacity to facilitate the development of the veil of flor.18
The most representative aging method in the Jerez area is the dynamic ‘solera y criaderas’ system, which involves the periodic extraction (‘saca’) of a wine fraction from the ‘solera’ scale (containing the oldest wine) and its replacement (‘rocío’) with wine from the immediately superior ‘criadera’ (1st criadera). This process is repeated successively through the younger ‘criaderas’ (2nd, 3rd, etc.), with the system being fed by the base wine (‘sobretabla’). The dynamic nature of this system ensures a continuous blending of different vintages, conferring stability and sensory homogeneity over time. Moreover, the periodic ‘rocíos’ sustain the veil of flor's vitality by supplying nutrients and dissolved oxygen, which are essential for the yeasts' aerobic metabolism. Wineries in Jerez typically perform 3 to 4 ‘sacas’ and ‘rocíos’ annually to maintain the requisite conditions for biological aging.19 A less widespread alternative, though one gaining recent prominence, is the static biological aging system (or ‘añada’). In this method, each cask is filled with wine from a single harvest and remains sealed throughout maturation, without the periodic ‘sacas’ and ‘rocíos’. The absence of replenishment with young wine limits the nutrient and oxygen supply available to the yeasts.20 This results in a distinct evolution of the veil of flor and a greater expression of the specific vintage conditions, yielding a more heterogeneous and complex wine profile.21
In response to growing global demand for lower-alcohol beverages, driven by health and sustainability trends, the wine sector has initiated a scientific and regulatory re-evaluation concerning biologically aged wines.22–24 The objective is to produce these wines at alcohol levels below the traditional standard. In this regard, the European Commission recently modified the ‘liqueur wine’ category specifications, which includes Fino and Manzanilla wines.25,26 This revision permits a potential reduction in their alcohol content to 14% v/v, subject to approval by the respective PDOs and inclusion in their product specifications. However, it is necessary to verify that this reduction does not significantly alter the essential physicochemical or sensory characteristics of these wines. These modifications aim to reduce dependence on external wine alcohol addition, moving towards a more ecological and sustainable process by lowering energy consumption, greenhouse gas (GHG) emissions,27 and the associated water footprint. However, ethanol metabolism by flor veil yeasts and the reduction in alcohol content could weaken the wine's natural microbiological barrier. This may favour the proliferation of undesirable microorganisms, such as lactic acid bacteria (LAB), acetic acid bacteria (AAB), or Brettanomyces spp.28 that could increase volatile acidity to the detriment of its sensory quality.29
This work represents one of the first industrial-scale studies on the effects of reduced alcohol content in static biological aging within the ‘Jerez-Xérès-Sherry’ and ‘Manzanilla de Sanlúcar’ PDOs. The primary objective is to evaluate the technical feasibility of this process in two representative wineries, assessing its sustainability, environmental impact, and oenological influence on flor veil yeast metabolism, volatile compound evolution, and sensory profile.
2. Materials and methods
2.1 Experimental
All experiments were conducted in two wineries, “winery A” and “winery B”, located in Jerez de la Frontera (Cádiz, Spain), belonging to the Jerez-Xèrés-Sherry Protected Designation of Origin (PDO). In each winery, four 600 L capacity barrels were selected, each containing 500 L of Palomino Fino variety wine. The selected wines had a medium aging of 3 years of biological aging to produce ‘Fino’ type wines. Two of them were left as controls for traditional biological aging, starting with an alcoholic strength between 15.5 and 16% v/v, and two others started with an alcoholic strength around 15% v/v to study the evolution of biological aging with a lower alcohol content. All of them underwent static aging under flor veil to study their evolution over a 12 month period. In winery A, temperature ranged from 15.36 °C in winter to 23.11 °C in summer and relative humidity from 69.67 to 73.91%, and in winery B, the seasonal range was more pronounced, with temperatures varying from 14.03 °C in winter to 26.93 °C in summer, and relative humidity ranging from 65.96% to 81.85%.Three samples were collected from each of the selected barrels: at the start, at six months, and at the end of the study period. In each case, measurement of physicochemical parameters (alcohol content, total and volatile acidity, pH, sulphur dioxide, glycerol, and abs 420 nm), determination of major and minor volatile compounds, and a sensory analysis were carried out in triplicate.
2.2 Standard oenological parameters
Standard oenological parameters (ethanol content, total and volatile acidity, pH, and sulphur dioxide) were analysed according to the protocols established by the International Organisation of Vine and Wine (OIV, 2025).30 The Total Polyphenol Index (TPI) was calculated from the absorbance at 280 nm, and the yellow colour was estimated from the absorbance measurement at 420 nm. Measurements were performed using a UV-VIS spectrophotometer (Genesys™ 10, Thermo Fisher Scientific).2.3 Volatile compounds
Major volatile compounds and polyols were determined by Gas Chromatography-Flame Ionization Detection (GC-FID). The analysis was performed on an Agilent GC-FID system (Agilent Technologies, Santa Clara, CA, USA) equipped with a CP-WAX 57 CB capillary column (60 m × 0.25 mm × 0.4 µm film thickness).For sample preparation, a 10 mL aliquot of wine was combined with 1 mL of the internal standard (4-methyl-2-pentanol, 1.018 g L−1) and 0.2 g of CaCO3. The mixture was briefly sonicated (30 s) before centrifugation (5000 rpm for 10 min at 2 °C). 0.7 µL of the supernatant was injected into the GC inlet using the split mode at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 ratio. Quantification relied on external calibration curves, which were generated using standard solutions prepared from certified analytical-grade compounds (Merck and Sigma-Aldrich Chemicals).
30 ratio. Quantification relied on external calibration curves, which were generated using standard solutions prepared from certified analytical-grade compounds (Merck and Sigma-Aldrich Chemicals).
Minor volatile compounds were analysed using Stir Bar Sorptive Extraction-Thermal Desorption-Gas Chromatography-Mass Spectrometry (SBSE-TD-GC-MS). The instrumental setup utilized an Agilent 7890A GC coupled to an Agilent 5975C MS (Agilent, Palo Alto, CA, USA) with a Gerstel Multi-Purpose Sampler.
For sample preparation and analysis, 1 mL aliquot of wine was mixed with 0.1 mL of hexyl butyrate (0.4116 g L−1 in ethanol) as the internal standard, and 8.9 mL of a buffered solution (12% (v/v) ethanol, pH 3.5) to a final volume of 10 mL. PDMS-coated Twister stir bars were then added and extracted for 120 minutes at 1200 rpm and 20 °C.
The stir bars were subsequently thermally desorbed, and analytes were separated on an HP-5MS capillary column (60 m × 0.25 mm × 0.25 µm). The GC oven temperature started at 50 °C (2 min), and was ramped at 4 °C min−1 to 190 °C (10 min hold). The MS acquired data in electron impact mode (70 eV, 35–550 Da). Identification was achieved through comparison with NIST08 and Wiley7 libraries and pure standards. Triplicate analysis and quantification were performed using the calibration table method outlined by Palenzuela et al. (2023).31
2.4 Sensory analysis
Sensory evaluations were conducted by a trained tasting panel from the OECCA Foundation, the entity responsible for the tasting and certification of wines under the PDOs Jerez-Xeres-Sherry and Manzanilla de Sanlúcar. The panel is ISO 17025 accredited and consists of 22 members (equally distributed by gender). Each wine sample was tasted in three independent sessions by groups of seven panelists per session. The wines were tasted blind, strictly following the standard evaluation protocols for these PDOs.In each session, 30 mL of each wine sample was served at room temperature (20 °C). The samples were presented in standardized tasting glasses (ISO 3591:1977),32 and were coded with random numbers to ensure the uniformity and anonymity of the analysis. Participants evaluated various attributes across three distinct phases: visual, olfactory, and gustatory. Participation was entirely voluntary, and all panellists were fully informed of the study's objectives. All results were collected and processed anonymously to comply with current data protection regulations.
A total of 12 attributes were evaluated: three in the visual phase (color, viscous appearance, and visual defects), six in the olfactory phase (biological aging, oxidative aging, raisining notes, ethyl acetate, TCA, and other olfactory defects), and three in the gustatory phase (sweetness, body, and gustatory defects). All attributes were assessed using a seven-point scale, where 1 represents absence and 7 indicates maximum intensity for each trained parameter. These are the standard attributes determined by the tasting panel to assess whether the wines comply with the specifications established in the PDO's book of requirements.
2.5 Statistical analysis
Data obtained from physicochemical parameters and major and minor volatile analyses were subjected to statistical evaluation using Prism GraphPad 10 (GraphPad Software, San Diego, CA, USA). ANOVA was performed, followed by Bonferroni's least significant difference, and differences were considered statistically significant at p < 0.05. Multivariate statistical techniques, including principal component analysis (PCA) with orthogonal rotation (varimax) as the factor extraction method, were employed. PCA was applied to both major and minor volatile compound datasets using IBM SPSS Statistics (International Business Machines Corporation, Armonk, New York, United States).3. Results and discussion
3.1 Oenological parameters
Tables 1 and 2 present the values of the main oenological parameters quantified during static biological aging (t = 0, 6, and 12 months) in test barrels aged at <15% v/v and control barrels at >15% v/v from the A and B wineries. Regarding ethanol content, one of the main substrates consumed by flor yeast, the control barrels in both A and B wineries show a decrease of approximately 3% of total content after one year, whereas the test barrels exhibit a reduction of 10% (A) and 5% (B). This consumption was significantly pronounced during the first six months (from May to November). This indicates that yeast metabolism of ethanol in a static biological aging system is enhanced when the alcoholic strength is below 15% v/v. In static aging systems, the omission of “sacas and rocíos” eliminates the periodic supply of micronutrients and oxygen, a condition that can limit the proliferation of flor yeast. Under these circumstances, a lower alcoholic strength may improve the growth conditions for the yeast by reducing stress, thereby promoting its aerobic metabolism and cellular development. The significant difference in ethanol consumption, with the A test barrels showing a rate double that of the B test barrels, could be due to differences in the predominant yeast strains in each solera system5 and other factors such as a higher or lower presence of nitrogenous micronutrients as reported by some authors.33| Winery A | 0 control | 0 test | p-value | 6 control | 6 test | p-value | 12 control | 12 test | p-value |
|---|---|---|---|---|---|---|---|---|---|
| pH | 3.20 ± 0.00 | 3.24 ± 0.01 | 0.2612 | 3.20 ± 0.01 | 3.26 ± 0.04 | 0.0924 | 3.20 ± 0.02 | 3.26 ± 0.01 | 0.0924 |
| Total acidity (g L−1) | 5.28 ± 0.03 | 4.80 ± 0.04 | 0.0014 | 4.79 ± 0.01 | 3.88 ± 0.04 | <0.0001 | 5.03 ± 0.01 | 3.77 ± 0.11 | <0.0001 |
| Volatile acidity (g L−1) | 0.21 ± 0.01 | 0.21 ± 0.04 | ns | 0.19 ± 0.01 | 0.14 ± 0.04 | 0.0347 | 0.17 ± 0.02 | 0.11 ± 0.01 | 0.0149 |
| Ethanol (% v/v) | 15.40 ± 0.07 | 14.73 ± 0.25 | 0.0105 | 15.18 ± 0.04 | 13.65 ± 0.07 | 0.0001 | 14.93 ± 0.04 | 13.10 ± 0.07 | <0.0001 |
| Abs 420 nm | 0.18 ± 0.00 | 0.17 ± 0.01 | ns | 0.20 ± 0.01 | 0.18 ± 0.00 | 0.4503 | 0.18 ± 0.00 | 0.17 ± 0.01 | ns |
| Absorbance 280 nm (TPI) | 13.55 ± 0.07 | 13.65 ± 0.21 | ns | 13.55 ± 0.21 | 13.70 ± 0.00 | ns | 13.60 ± 0.14 | 13.70 ± 0.14 | ns |
| Glycerol (g L−1) | 1.89 ± 0.12 | 1.46 ± 0.14 | 0.0268 | 1.08 ± 0.07 | 0.41 ± 0.01 | 0.0025 | 0.94 ± 0.01 | 0.37 ± 0.00 | 0.0061 |
| Winery B | 0 control | 0 test | p-value | 6 control | 6 test | p-value | 12 control | 12 test | p-value |
|---|---|---|---|---|---|---|---|---|---|
| pH | 3.11 ± 0.03 | 3.10 ± 0.03 | ns | 3.10 ± 0.03 | 3.14 ± 0.05 | ns | 3.08 ± 0.04 | 3.12 ± 0.00 | ns |
| Total acidity (g L−1) | 4.66 ± 0.04 | 4.53 ± 0.21 | ns | 4.51 ± 0.07 | 4.50 ± 0.13 | 0.6480 | 4.63 ± 0.05 | 4.34 ± 0.11 | 0.6480 |
| Volatile acidity (g L−1) | 0.21 ± 0.02 | 0.28 ± 0.21 | 0.2736 | 0.18 ± 0.01 | 0.4 ± 0.14 | 0.0010 | 0.19 ± 0.01 | 0.30 ± 0.05 | 0.0398 |
| Ethanol (% v/v) | 15.93 ± 0.11 | 14.98 ± 0.11 | 0.0037 | 15.69 ± 0.01 | 14.50 ± 0.28 | 0.0011 | 15.41 ± 0.02 | 14.15 ± 0.08 | 0.0008 |
| Abs 420 nm | 0.22 ± 0.002 | 0.22 ± 0.00 | ns | 0.21 ± 0.00 | 0.21 ± 0.00 | ns | 0.22 ± 0.002 | 0.21 ± 0.01 | ns |
| Absorbance 280 nm (TPI) | 14.30 ± 0.04 | 14.41 ± 0.11 | ns | 14.31 ± 0.01 | 14.35 ± 0.08 | ns | 14.23 ± 0.04 | 14.40 ± 0.09 | 0.4475 |
| Glycerol (g L−1) | 1.23 ± 0.16 | 0.52 ± 0.01 | 0.0017 | 1.39 ± 0.10 | 0.41 ± 0.02 | 0.0003 | 1.61 ± 0.04 | 0.40 ± 0.02 | <0.0001 |
Concerning total acidity, divergent trends were observed between wineries A and B. Whereas no differences in total acidity were detected between the test and control barrels at any time point in winery B, the test barrels in winery A exhibited significantly lower total acidity than the control barrels. Likewise, in the case of A total acidity decreased significantly over time unlike in B, where values remained constant. This is likely due to a greater precipitation of tartaric salts in winery A, suggesting that the control and test wines from this winery may have higher concentrations of tartaric acid and/or potassium than those from winery B. Additionally, the A wines (both control and test) had a higher average pH of 3.2 compared to the B wines (3.1). A pH around 3.2 favors the formation and precipitation of potassium bitartrate which leads to a reduction in total acidity.34
Volatile acidity is a key parameter for monitoring the progress of biological aging. Acetic acid is metabolized by the flor yeasts during this process35–38 through its incorporation into the fatty acid metabolic pathway. However, acetic acid is also one of the main products generated by certain microorganisms, such as lactic and acetic acid bacteria.39,40 At certain levels acetic acid can affect yeast growth and viability (above 0.5 g L−1).41 As shown in Tables 1 and 2, volatile acidity in A was significantly reduced (p < 0.05) over time in both control and test barrels. This decrease was much more pronounced in the test barrels (around 48%) compared to the control wine (19%), similar to the greater ethanol consumption observed in the test barrels during the warmer first stage (May–November). This again corroborates that yeast metabolism is more accentuated in barrels with lower alcoholic strength. The behavior in B was different: while the control's volatile acidity slightly decreased, the test barrel's volatile acidity initially surged by 47% (from 0.28 to 0.4 g L−1) in the first six months before settling again at 0.3 g L−1.
This initial increase could be due to some microorganisms, such as lactic or acetic acid bacteria, which thrive during the warmer summer months when flor yeast growth is inhibited6 causing acetic acid production to exceed consumption, leading to accumulation at tolerable levels. Therefore, in a static aging system, while a lower alcoholic strength favors flor yeast growth, it may also promote the development of other microorganisms due to their better adaptation to high temperatures during the first period. The presence of specific substrates, such as malic or gluconic acid, can promote the growth of these microorganisms, making their control necessary in static biological aging.42 Subsequently, when conditions become more favorable for flor yeast development (November–May), acetic acid consumption surpasses production, leading to an overall decrease in volatile acidity in winery B. Thus, the static biological aging process itself demonstrated the capacity to purify the medium and control the acetic acid levels in the wine.
Glycerol is another key substrate consumed by flor yeast. It has been used as an indicator to establish the time or degree of biological aging.43 As can be seen (Tables 1 and 2), glycerol levels at time zero were relatively low, indicating that the wines had undergone a significant period of biological aging prior to the experiment. In both A and B wineries, glycerol values were significantly lower (p < 0.05) in the test barrels compared to the controls. A similar trend was observed for its consumption over time: a decrease of nearly 75% was observed in the A test barrels (vs. 50% in controls), while in the B barrel, the reduction was 25% (vs. a 31% increase in control). Thus, although winery B exhibited lower glycerol levels than winery A at time zero, its intermediate and final concentrations were higher, particularly in the control barrels. As with ethanol, glycerol consumption was greater during the first stage of the trial (May–November). This again demonstrates that a lower alcoholic strength improves the metabolism of flor yeast. This may lead to a marked increase in dryness or a lack of sweetness and viscosity on wine, a characteristic feature of Fino sherry wines.2
Absorbance at 420 nm measures the intensity of yellow tones in wine, which increases as oxygen exposure causes phenolic compounds to polymerize into brown pigments, serving as a primary indicator of the wine's oxidation level. In biological aging, the development of the veil of flor protects the wine from oxidation by consuming dissolved oxygen, making a stable absorbance at 420 nm a key indicator of a healthy and successful aging process. In this case, the values of absorbance remained stable over time and showed no significant differences between the control and test barrels for either A or B wineries. This indicates that in all cases, including the barrels with lower alcoholic strength (<15%), the flor layer developed well, with active oxygen consumption from both the barrel headspace and the wine itself, minimizing the oxidation reactions of polyphenolic compounds.2
3.2 Major volatile compounds and polyols
Tables 3 and 4 show the average concentrations of the major volatile compounds in the test and control sherry barrels from both wineries.| Winery A | CAS | 0 control | 0 test | p-value | 6 control | 6 test | p-value | 120 control | 120 test | p-value |
|---|---|---|---|---|---|---|---|---|---|---|
| Acetaldehyde | 75-07-0 | 378.05 ± 12.57 | 392.76 ± 4.53 | ns | 318.38 ± 8.14 | 543.4 ± 13.14 | <0.0001 | 323.06 ± 14.54 | 707.03 ± 14.79 | <0.0001 |
| Ethyl acetate | 141-78-06 | 63.74 ± 0.83 | 61.0 ± 0.75 | ns | 64.58 ± 1.30 | 50.12 ± 0.64 | 0.0005 | 62.82 ± 0.73 | 46.65 ± 2.57 | 0.003 |
| Diethyl acetal | 105-57-57 | 20.54 ± 0.75 | 18.69 ± 0.88 | 0.3569 | 4.82 ± 0.67 | 7.06 ± 0.10 | 0.1624 | 3.94 ± 0.23 | 9.93 ± 0.23 | 0.0010 |
| Methanol | 67-56-1 | 110.90 ± 3.00 | 104.51 ± 3.30 | ns | 79.05 ± 3.86 | 77.20 ± 9.62 | ns | 50.94 ± 2.80 | 81.54 ± 3.7 | 0.0129 |
| 1-Propanol | 71-23-8 | 57.52 ± 0.68 | 56.24 ± 0.65 | ns | 49.68 ± 2.91 | 53.79 ± 1.79 | 0.5198 | 52.11 ± 0.80 | 52.02 ± 0 | ns |
| Isobutanol | 78-83-1 | 51.62 ± 0.83 | 58.11 ± 0.29 | 0.0027 | 43.94 ± 1.48 | 56.72 ± 0.55 | <0.0001 | 44.91 ± 0.51 | 58.92 ± 0.51 | <0.0001 |
| 2-Methyl-1-butanol | 137-32-6 | 38.93 ± 1.59 | 41.16 ± 0.76 | 0.8733 | 37.66 ± 1.33 | 41.45 ± 0.55 | 0.1106 | 37.70 ± 0.05 | 42.53 ± 0.54 | 0.0347 |
| 3-Methyl-1-butanol | 123-51-3 | 199.93 ± 12.57 | 208.91 ± 4.53 | 0.8062 | 232.81 ± 8.14 | 256.60 ± 13.14 | 0.0108 | 239.22 ± 14.54 | 264.28 ± 14.79 | 0.0082 |
| Acetoin | 513-86-0 | 54.67 ± 5.65 | 101.11 ± 6.40 | 0.0393 | 52.76 ± 6.69 | 194.39 ± 12.81 | <0.0001 | 46.36 ± 1.24 | 268.17 ± 15.75 | <0.0001 |
| Ethyl lactate | 97-64-3 | 505.20 ± 34.16 | 361.13 ± 15.85 | 0.0040 | 343.62 ± 9.02 | 138.99 ± 8.41 | 0.0006 | 339.09 ± 22.46 | 115.26 ± 8.31 | 0.0003 |
| 2,3-Butanediol (levo) | 24347-58-8 | 1041.6 ± 111.23 | 965.06 ± 78.35 | ns | 646.29 ± 46.36 | 772.25 ± 53.97 | ns | 647.17 ± 34.25 | 786.60 ± 63.86 | ns |
| 2,3-Butanediol (meso) | 5341-95-7 | 412.22 ± 42.78 | 452.54 ± 37.12 | ns | 240.14 ± 14.64 | 353.61 ± 25.60 | ns | 237.66 ± 12.88 | 388.60 ± 19.86 | ns |
| Diethyl succinate | 123-25-1 | 40.06 ± 5.32 | 39.04 ± 3.73 | ns | 19.14 ± 0.72 | 17.53 ± 0.90 | ns | 18.99 ± 1.06 | 18.10 ± 0.96 | ns |
| 2-Phenyl-ethanol | 60-12-8 | 50.23 ± 4.05 | 49.95 ± 3.12 | ns | 33.38 ± 2.95 | 40.60 ± 2.54 | 0.9249 | 31.63 ± 2.84 | 42.48 ± 3.18 | 0.2054 |
| Winery B | CAS | 0 control | 0 test | p-value | 6 control | 6 test | p-value | 12 control | 12 test | p-value |
|---|---|---|---|---|---|---|---|---|---|---|
| Acetaldehyde | 75-07-0 | 187.63 ± 2.14 | 245.69 ± 245.69 | 0.0091 | 179.26 ± 2.60 | 202.86 ± 10.39 | 0.5619 | 220.88 ± 5.42 | 274.23 ± 15.59 | 0.0143 |
| Ethyl acetate | 141-78-06 | 49.86 ± 1.80 | 34.70 ± 0.64 | 0.1828 | 45.09 ± 4.41 | 77.41 ± 4.52 | 0.0042 | 51.64 ± 4.83 | 75.83 ± 6.56 | 0.0197 |
| Diethyl acetal | 105-57-57 | 13.63 ± 0.84 | 13.26 ± 1.22 | ns | 4.01 ± 0.12 | 0.94 ± 0.95 | 0.1223 | 3.40 ± 0.05 | 3.67 ± 0.80 | ns |
| Methanol | 67-56-1 | 106.5 ± 6.51 | 117.61 ± 10.74 | <0.0001 | 78.63 ± 9.76 | 85.73 ± 4.75 | ns | 93.66 ± 3.98 | 103.02 ± 6.64 | ns |
| 1-Propanol | 71-23-8 | 50.00 ± 0.34 | 48.04 ± 0.96 | ns | 44.34 ± 0.41 | 40.92 ± 2.54 | 0.4232 | 43.46 ± 0.57 | 38.91 ± 0.71 | 0.1305 |
| Isobutanol | 78-83-1 | 92.31 ± 0.83 | 97.86 ± 0.91 | 0.0120 | 77.84 ± 1.11 | 80.44 ± 1.20 | 0.40 | 77.82 ± 0.45 | 79.57 ± 0.63 | ns |
| 2-Methyl-1-butanol | 137-32-6 | 67.42 ± 0.95 | 70.37 ± 1.82 | ns | 64.76 ± 2.01 | 65.07 ± 1.25 | ns | 64.9 ± 0.53 | 64.13 ± 2.10 | ns |
| 3-Methyl-1-butanol | 123-51-3 | 293.7 ± 0.67 | 300.22 ± 2.15 | ns | 341.57 ± 6.22 | 339.88 ± 5.17 | ns | 341.04 ± 2.88 | 337.36 ± 1.74 | ns |
| Acetoin | 513-86-0 | 46.56 ± 2.82 | 76.43 ± 7.12 | 0.0151 | 75.12 ± 3.34 | 91.92 ± 5.67 | 0.2317 | 89.31 ± 4.66 | 96.10 ± 5.23 | ns |
| Ethyl lactate | 97-64-3 | 99.14 ± 3.65 | 36.99 ± 2.63 | <0.0001 | 74.80 ± 3.71 | 19.14 ± 0.22 | <0.0001 | 75.88 ± 2.95 | 18.10 ± 0.64 | <0.0001 |
| 2,3-Butanediol (levo) | 24347-58-8 | 798.54 ± 70.57 | 978.51 ± 126.97 | 0.5273 | 529.33 ± 15.50 | 582.13 ± 48.80 | ns | 752.60 ± 27.30 | 625.11 ± 44.87 | ns |
| 2,3-Butanediol (meso) | 5341-95-7 | 327.57 ± 26.40 | 412.09 ± 48.85 | 0.2974 | 218.45 ± 15.36 | 255.45 ± 23.06 | ns | 295.44 ± 9.26 | 257.51 ± 19.61 | ns |
| Diethyl succinate | 123-25-1 | 53.36 ± 4.42 | 68.52 ± 6.08 | 0.0502 | 28.32 ± 1.41 | 20.28 ± 0.86 | 0.7081 | 27.94 ± 1.01 | 20.60 ± 1.53 | 0.9517 |
| 2-Phenyl-ethanol | 60-12-8 | 68.37 ± 2.47 | 77.77 ± 5.77 | 0.6589 | 52.10 ± 3.14 | 52.78 ± 3.44 | ns | 55.28 ± 2.92 | 53.18 ± 3.52 | ns |
Acetoin is another characteristic compound of biological aging, imparting a bitter almond aroma. It is formed through the acyloin condensation of two acetaldehyde molecules.48 As shown in Tables 3 and 4, like acetaldehyde, acetoin concentrations were higher in the test barrels than in the control barrels at both wineries. However, the evolution of this compound over time differed between the wineries. In the A control barrels, acetoin levels remained relatively constant with a slight decrease, and A test barrels showed a significant increase, reaching concentrations of 268 mg L−1, nearly six times that of the control. A correlation between acetoin and acetaldehyde levels was observed in winery A with a linear regression coefficient (r2) of 0.97, which aligns with previous studies.49 In winery B, acetoin increased over time in both control and test barrels, showing no correlation with acetaldehyde levels.50,51
Conversely, 2,3-butanediol, which is produced by yeast via the reduction of acetoin, showed no significant differences between the test and control barrels. Regarding its evolution, 2,3-butanediol levels remained constant in the A test barrel. In all other barrels, its concentration decreased during the initial stage and then stabilized.
Diethyl acetal is another major acetal found in biologically aged wines, contributing balsamic and woody notes.52,53 It is formed through the chemical reaction of ethanol and acetaldehyde, and also via the metabolism of flor yeast. Similar to 2,3-butanediol, no significant differences in diethyl acetal concentration were found between the control and test barrels in either winery, with a very similar evolution in all cases.
Regarding ethyl acetate, significant differences were observed between the test and control barrels in both wineries at 6 and 12 months. The behavior of this compound differed between the locations. In winery A, the test barrels exhibited lower levels of ethyl acetate than the control barrels. Conversely, in winery B, the test barrels showed higher levels. The ethyl acetate concentration in the A test barrels decreased over time, with an overall reduction of 23% after 12 months. In contrast, the B test barrels showed a significant increase of 123% within the first 6 months. This behavior is linked to the wine's volatile acidity. In winery A, the consumption of acetic acid by the yeast led to a decrease in ethyl acetate. In winery B, however, the production of acetic acid exceeded its consumption during the initial months, resulting in higher ethyl acetate formation.
Diethyl succinate, associated with aging on lees,56 showed no significant differences between treatments and followed a pattern of initial decrease followed by stabilization, similar to 2,3-butanediol and diethyl acetal.
Isobutanol and 2-phenylethanol are produced by flor yeast from the amino acids valine and phenylalanine, respectively, via the keto acid pathway.58 Their production is reportedly related to cell growth and the concentration of dissolved oxygen.59 In winery A, isobutanol levels were higher in the test barrels than in the control ones, which could suggest greater yeast cell growth in the lower-alcohol environment. In winery B, however, no significant differences were observed. Isobutanol concentrations tended to decrease during the first six months and then stabilize, a pattern also seen with diethyl acetal, 2,3-butanediol, and diethyl succinate. The same was observed for 2-phenylethanol in both wineries (Tables 3 and 4).
Finally, the isoamyl alcohols 2-methyl-1-butanol and 3-methyl-1-butanol are produced by flor yeast from the amino acid precursors isoleucine and leucine, respectively.50 No significant differences in 2-methyl-1-butanol were observed between the control and test barrels at either winery, and its concentration remained constant throughout the experiment.
However, for 3-methyl-1-butanol, the results varied. In winery A, the test barrels had significantly higher concentrations than the control barrels at 6 and 12 months and increased during the first six months before stabilizing. In winery B, no significant differences were found between the test and control groups, but both showed a similar pattern of an initial increase followed by stabilization.
| PC1 | PC2 | PC3 | |
|---|---|---|---|
| Alcohol (%) | 0.292 | 0.105 | 0.889 |
| Volatile acidity | 0.603 | −0.039 | 0.218 |
| Total acidity | −0.162 | 0.033 | 0.956 |
| pH | −0.905 | −0.045 | −0.345 |
| Abs 420 nm | 0.924 | −0.087 | 0.219 |
| Glycerol | −0.249 | 0.264 | 0.790 |
| Acetaldehyde | −0.706 | 0.119 | −0.682 |
| Ethyl acetate | −0.097 | −0.563 | 0.246 |
| Diethyl acetal | −0.438 | 0.841 | 0.238 |
| Methanol | 0.320 | 0.801 | 0.166 |
| 1-Propanol | −0.857 | 0.434 | 0.112 |
| Isobutanol | 0.904 | 0.398 | −0.061 |
| 2-Methyl-1-butanol | 0.977 | 0.176 | 0.012 |
| 3-Methyl-1-butanol | 0.922 | −0.236 | −0.211 |
| Acetoin | −0.265 | 0.078 | −0.936 |
| Ethyl lactate | −0.840 | 0.053 | 0.524 |
| 2,3-Butanediol (levo) | −0.366 | 0.881 | 0.119 |
| 2,3-Butanediol (meso) | −0.379 | 0.876 | −0.172 |
| Diethyl succinate | 0.305 | 0.846 | 0.339 |
| 2-Phenyl-ethanol | 0.696 | 0.706 | 0.098 |
| Variances% | 40 | 27 | 19 |
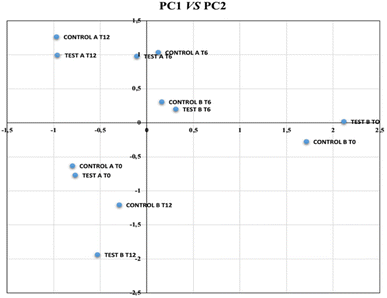
PC1 (40% variance), the “winery factor”, effectively discriminated between the A and B wineries, regardless of the barrel type (test or control). PC1 was negatively correlated (factor loading > 0.6) with pH, acetaldehyde, 1-propanol, and ethyl lactate, and positively correlated with volatile acidity, isobutanol, 2-methyl-1-butanol, 3-methyl-1-butanol, and 2-phenylethanol. As shown in Fig. 1, B samples scored positively on PC1, while A samples scored negatively. This indicates that the specific operating conditions of each winery (e.g., temperature, dominant flor yeast strain, flor surface area) are the primary drivers of variance, allowing for a clear distinction between the wines from each location. This finding aligns with previous studies showing that the dominant yeast strain in a winery is a determining factor in the aromatic profile of biologically aged wines.12,45,60
 | ||
| Fig. 1 Biplot (PC1 vs. PC2) from the PCA of the physicochemical parameters and major volatile compounds. | ||
PC2 (27% variance), the “time factor”, was positively correlated with variables that decreased during aging, particularly in the initial months. This factor only discriminated the samples at t = 0 (positive scores) from those at later time points (t = 6 and t = 12 months), which had lower scores (Fig. 1). Therefore, PC2 is independent of both winery conditions and barrel type.
PC3 (19% variance), the “aging factor”, was positively correlated with substrates (alcohol, glycerol) and total acidity, and negatively with key metabolic products (acetaldehyde, acetoin). This “aging factor” successfully discriminated between the test and control barrels, in both wineries.
As seen in Fig. 2, test barrels (negative scores) and control (positive scores) provide a measure of the metabolic intensity in each group. In winery A, the test barrels at 6 and 12 months showed a much more pronounced metabolic activity (PC3 scores between −1.5 and −2.18) compared to the control barrels (PC3 score of +0.7). Furthermore, the PC3 scores in the test barrels continued to decrease over time, a trend not observed in the controls. In winery B, the difference in PC3 scores between test and control barrels was much smaller. The aging evolution was more favourable in the control barrels; a metabolic setback was observed in the test barrels after 6 months, likely due to the increase in volatile acidity (Table 2).
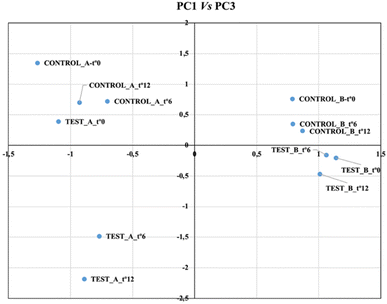 | ||
| Fig. 2 Biplot (PC1 vs. PC3) from the PCA of the physicochemical parameters and major volatile compounds. | ||
Overall, the yeast metabolism during biological aging in the lower-alcohol wines was significantly more pronounced during the warmer months (May to November) compared to the cooler period (November to May). This trend was observed in both wineries, although it was less marked in B.
3.3 Minor volatile compounds
A total of 37 minor volatile compounds were identified and quantified in test (<15% v/v alcohol) and control (>15% v/v alcohol) barrels from wineries A and B at 0, 6, and 12 months. The detailed composition of minor volatile compounds is summarised in Tables S1 and S2 (SI). In winery A, the total concentration of minor volatiles increased significantly over the first 6 months (56–79%) before returning to near-initial levels. In contrast, winery B exhibited a continuous decrease throughout the aging period, although its concentrations remained consistently higher than those in A. This suggests that the evolution of minor aromatic compounds is primarily governed by the specific conditions of each winery (temperature, dominant flor yeast strain, flor surface area, …). As expected, the behavior of most volatile families followed the same trend of the total aroma sum within their respective wineries, for both test and control wines. Nonetheless, significant differences between test and control barrels were noted for specific aromatic families, likely due to the metabolic activity of flor yeasts or other factors.In winery A, acetate concentrations increased significantly during the first six months, particularly in the test barrels. Subsequently, levels decreased, more notably in the control barrels, during the second aging period (autumn to spring). This suggests that the dominant yeast strain in A modulates its synthesis/hydrolysis activity in response to environmental conditions and that aging at a lower alcoholic strength favors synthesis over hydrolysis. In winery B, although test barrels also maintained higher acetate levels than controls, concentrations significantly decreased over time, especially within the first six months in the control barrels. This indicates that the predominant yeast strain in winery B exhibits greater hydrolytic activity compared to the strain in winery A. Furthermore, the metabolic activity in the B test barrels may have been inhibited by an increase in volatile acidity and potential bacterial growth. Consequently, the final acetate concentrations were higher in A than in B, particularly in the test barrels.
The two wineries displayed divergent trends. In A, ethyl ester concentrations doubled within the first 6 months before decreasing slightly by month 12. Conversely, in B, a continuous decline was observed throughout the aging period. While these findings reaffirm the influence of specific winery conditions, a key distinction from acetates emerged: test barrels contained lower concentrations of ethyl esters than control barrels. This suggests that the higher ethanol content in the control wines, which promotes esterification, was a more dominant factor than the higher yeast metabolic activity observed in the test wines. Specifically, ethyl 2-methylbutanoate, ethyl 3-methylbutanoate and ethyl octanoate showed significant differences between test and control conditions.
β-Damascenone was the only norisoprenoid identified. It has a very low perception threshold (0.05 µg L−1) and imparts fruity, floral, and honey-like aromas.69 Its temporal behavior varied depending on the winery. In winery A, its concentration progressively increased to 9 µg L−1 in the test barrels and 16 µg L−1 in the control barrels, with no significant differences at any time point. In contrast, at winery B, a slight increase occurred at 6 months, followed by a decrease to very low levels. Significant differences between the test and control barrels were only observed at the 6 month mark. Some studies have shown that β-damascenone can slightly increase during the initial years of aging due to the enzymatic action of flor yeasts, which hydrolyse norisoprenoid glycosides.67 However, its concentration tends to decrease over longer aging periods due to oxidation and possible adsorption by the flor velum.68
Terpenes were found in very low levels, as is typical for biologically aged wines. The most significant terpenes identified were limonene and (E)-methyldihydrojasmonate. Their evolution over time differed by winery. In winery A, limonene concentration increased steadily, reaching up to 66 µg L−1. In B, however, limonene appeared only briefly at 6 months before disappearing. In contrast, (E)-methyl dihydrojasmonate was initially present in both wineries but became undetectable within a year. The presence of limonene in biologically aged wines has been previously reported.24,70 This phenomenon has been linked to mites, such as Carpoglyphus lactis, found on barrel corks.70,71 These mites can fall into the wine during sampling, releasing terpenes that impart characteristic citrus and floral notes.70
| PC1 | PC2 | |
|---|---|---|
| Acetates | 0.425 | 0.429 |
| Ethyl esters | 0.908 | 0.144 |
| Higher alcohols | 0.876 | −0.010 |
| Lactones | 0.945 | 0.123 |
| Carbonyl compounds | 0.056 | 0.619 |
| Volatile phenols | 0.958 | −0.003 |
| Terpenes and derivates | −0.573 | 0.692 |
| Norisoprenoids | −0.060 | 0.935 |
| Variance (date)% | 49 | 14 |
PC1 (49% variance), the “aroma factor”, was positively correlated (factor loading > 0.6) with the main groups of minor aroma compounds: ethyl esters, higher alcohols, lactones, and volatile phenols. Consistent with numerous studies, most of these compounds tend to decrease during biological aging and are significantly influenced by flor yeast. The “aroma factor” successfully discriminated between the barrels based on aging time and winery (Fig. 3).
However, it did not distinguish between test and control barrels, indicating that aging at a lower alcohol concentration did not generally affect these minor aroma compounds. The only exception was the acetate family, which showed significant differences between test and control barrels, but its loading in the PCA was split between PC1 and PC2. The “aroma factor” score was consistently higher in winery B at all sampling times and its score decreased over time, whereas in A, it increased slightly at 6 months before decreasing at 12 months, showing little overall change. This different behavior may be attributed to the dominant yeast strains in each winery, which can either promote the synthesis or the hydrolysis and transformation of these compounds. This effect is evidenced by the temporal evolution of the cumulative concentrations of ethyl esters, higher alcohols, lactones, and volatile phenols (Fig. 4), which reveals markedly different trends during the first six months. Whereas winery A showed an approximate increase of 60–80% in these compounds, indicating a greater synthetic capacity of flor yeasts, winery B exhibited a 40% decrease, reflecting the prevalence of hydrolytic and transformation activities.
 | ||
| Fig. 4 Temporal evolution of the cumulative concentrations of volatile compounds in control and test barrels from wineries A and B during biological aging. | ||
PC2 (14% variance), the “barrel factor”, was positively correlated (factor loading > 0.6) with carbonyls (furfural), terpenes (limonene and (E)-methyl dihydrojasmonate), and the volatile phenol 4-ethylguaiacol. These compounds are associated with oak wood, either directly or indirectly (e.g., via mites), and can be released into the wine during aging. The “barrel factor” score tended to increase over time in the A barrels but decrease in the B barrels. In general, test barrels showed a slightly lower “barrel factor” score compared to control barrels, except at t = 0 in B. This difference may be due to the higher alcohol content in the control barrels, which could enhance the extraction of these compounds from the wood.
3.4 Sensory analysis
Fig. 5 and 6 present the results obtained for the principal parameters evaluated in the tasting panel for the two wineries throughout the entire study period. It should be noted that, to simplify, the attributes ‘visual defects,’ ‘olfactory defects,’ and ‘gustatory defects' are not shown, since no defects were identified in any of the samples. Furthermore, the attribute ‘viscous appearance’ was omitted since all samples consistently exhibited the minimum score, as expected for wines of this category. | ||
| Fig. 5 Mean intensity values for sensory attributes in biologically aged wines from winery A, comparing traditional (control) and lower (test) ethanol content over 12 months (t = 0, 6, 12). | ||
 | ||
| Fig. 6 Mean intensity values for sensory attributes in biologically aged wines from winery B, comparing traditional (control) and lower (test) ethanol content over 12 months (t = 0, 6, 12). | ||
The ‘biological aging’ attribute registered the highest mean scores and the largest variance across both wineries and wine types, which is specific for these wines. In winery A, the values for this parameter remained virtually constant throughout the study in the control wines; however, in the test wines, we can observe an increase over time and a decrease in alcoholic strength, which is in concordance with the data obtained for acetaldehyde content and the previously mentioned intensification of the flor yeast metabolism resulting in a significantly higher score in the low-alcohol wines at the end of the study. In winery B, no substantial increase was observed with time, although the final score at the end of the study was also significantly higher for the wines with lower alcoholic strength. The remaining parameters in the olfactory assessment (oxidative aging and raisining attributes) registered scores very close to one in all cases, indicating their absence or presence at levels near the detection threshold. This finding aligns with the typical aromatic profile for this wine type and with the characteristics stipulated in its product specifications. The parameters ethyl acetate and TCA also received scores of 1 (denoting absence) or values closely approximating it.
In the gustatory assessment, all evaluated wines, regardless of alcohol content and winery, received very low scores for body. This is characteristic of these wines, which typically exhibit a very light body, likely attributed to glycerol consumption by the yeast. Furthermore, the sweetness parameter was rated at the minimum score (absence) for all samples, consistent with their classification as dry wines.
The color remained nearly constant in the first cellar, averaging values near 2.5, which is characteristic of a straw-yellow hue. In winery B, a minor evolution was noted potentially attributable to seasonal flor velum activity but no differences were observed between the control and test casks. In this winery, values were closer to 3, corresponding to a pale golden color. This chromatic range is typical for this wine category.
Finally, it should be noted that all wines evaluated throughout the study period met the required sensory specifications outlined in the regulations for the Jerez-Xérès-Sherry PDO.
3.5 Sustainability assessment
This section evaluates the potential economic, energy, and environmental impacts of reducing the alcohol degree of biologically aged sherry wines from 15% to 14% (v/v). The primary benefits stem from the reduced input of wine alcohol (ethanol) required for the initial fortification and subsequent adjustments during the aging process. For this analysis, a simplified assumption of a cumulative 1% (v/v) reduction in total ethanol addition throughout the entire aging stage was adopted. Table 7 lists the parameters used for this approximation.| Parameter | Value used | Reference |
|---|---|---|
| Ethanol (95%) required per 1000 L of wine | 5.3 L | Calculated directly from proportion (1% v/v × 500 L/95%) |
| Average market price of 95% ethanol | 7 € per L | MAPA (2024)72 |
| Energy consumption for ethanol distillation | 6.5 kW h L−1 | Soleymani Angili et al. (2021)73 |
| GHG emissions for ethanol production | 2.3 kg CO2 eq per L | Therasme et al. (2021)74 |
| Density of 95% ethanol | 0.81 kg L−1 | Standard physical property |
| Road transport emission factor | 0.09 kg CO2 eq per t per km | EEA (2022)75 |
| Bulk tanker capacity (liquid food transport) | 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 L 000 L |
Typical european road tanker specification |
| Average tanker freight cost | 2.0 € per km | Estimated for specialised food-grade bulk transport (Spain) |
| Regulatory framework for sherry fortification | — | Consejo regulador DO Jerez-Xérès-Sherry (2023)76 |
Based on proportional calculations, lowering the alcohol content by one percentage point saves approximately 5.3 L of 95% ethanol per 500 L barrel. This saving translates directly into economic, energy, and emission benefits. Considering an average market price of 7 € per L, this reduction yields a direct economic saving of approximately 37 € per barrel. Furthermore, applying an energy intensity factor of 6.5 kW h L−1 and an emission factor of 2.3 kg CO2 eq per L—validated against published bioethanol Life Cycle Assessment (LCA) studies and the Ecoinvent v3.10 database77 – this 5.3 L reduction corresponds to savings of 37.34 kW h and 12 kg CO2 eq per barrel, respectively.
To illustrate the potential regional impact, these per-barrel savings were extrapolated based on an estimated average annual production of 5000 barrels (2.5 million L) from the participating wineries. The aggregated calculations are presented in Table 8. This extrapolation yields a total annual saving of 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 325 L of ethanol, 184
325 L of ethanol, 184![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 275 €, 171 MW h, and approximately 60.5 t CO2 eq in each winery. The analysis also incorporated the avoided transport of ethanol from Tomelloso (the primary supply region) to Jerez, a distance of 540 km. This contributes an additional saving of approximately 1 t CO2 eq and 1000 € in freight costs, equivalent to 0.94 fewer tanker trips. While transport-related impacts represent less than 2% of the total GHG savings, their inclusion provides a more complete life-cycle perspective.
275 €, 171 MW h, and approximately 60.5 t CO2 eq in each winery. The analysis also incorporated the avoided transport of ethanol from Tomelloso (the primary supply region) to Jerez, a distance of 540 km. This contributes an additional saving of approximately 1 t CO2 eq and 1000 € in freight costs, equivalent to 0.94 fewer tanker trips. While transport-related impacts represent less than 2% of the total GHG savings, their inclusion provides a more complete life-cycle perspective.
| Parameter | Unit | Per barrel | Total (5000 barrels) |
|---|---|---|---|
| Ethanol saved (95%) | L | 5.27 | 26.325 |
| Economic saving (ethanol) | € | 36.9 | 184.275 |
| Energy saved (ethanol production) | kW h | 34.3 | 171.113 |
| GHG emissions avoided (ethanol production) | kg CO2eq | 12.1 | 60.548 |
| Mass of ethanol avoided | t | 0.00427 | 21.32 |
| Transport distance | km | 540 | 540 |
| GHG emissions avoided (transport) | kg CO2eq | 0.21 | 1.036 |
| Transport cost avoided | € | 0.20 | 1.015 |
Equivalent tanker trips avoided (28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 L) 000 L) |
— | 0.00019 | 0.94 |
In summary, for an illustrative annual production of 5000 barrels, reducing the alcohol degree by 1% (v/v) avoids the use of over 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 L of 95% wine ethanol, generates savings of approximately 184
000 L of 95% wine ethanol, generates savings of approximately 184![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 €, and prevents a total of ∼61 t CO2 eq (including production and transport logistics). These findings demonstrate that even a marginal reduction in fortification strength can yield significant economic, energy, and environmental benefits, contributing to the sherry industry's alignment with European climate-neutrality and energy-efficiency objectives.
000 €, and prevents a total of ∼61 t CO2 eq (including production and transport logistics). These findings demonstrate that even a marginal reduction in fortification strength can yield significant economic, energy, and environmental benefits, contributing to the sherry industry's alignment with European climate-neutrality and energy-efficiency objectives.
4. Conclusions
Static biological aging of sherry wines with an alcoholic strength below 15% v/v proves to be a viable and effective strategy, resulting in a significant enhancement of flor yeast metabolism. This was clearly evidenced by the accelerated consumption of primary substrates, including ethanol, glycerol, and volatile acidity. This metabolic enhancement is, however, strongly modulated by the specific ecological and climatic conditions of the winery. The process is favored by milder, more stable temperatures and exhibits slight seasonal variations. In this context, while a rapid decrease in alcohol content during warmer months might, in some cases, favor the growth of competing microorganisms, the static biological aging process itself demonstrated an intrinsic capacity to purify the medium and control acetic acid levels. In addition, the flor veil consistently maintained its protective capacity against oxidation across all scenarios, as evidenced by stable color over time, independent of the alcohol content.The intensified metabolic activity translates into an enhancement of typicity. Key chemical markers of biological aging, especially acetaldehyde and acetoin, reached significantly higher concentrations in the reduced-alcohol wines. Concurrently, the minor volatile profile shifted, characterized by higher concentrations of acetate esters and lower levels of ethyl esters and furfural. These chemical findings were validated by sensory analysis. The reduced-alcohol wines not only met all quality parameters but also received significantly higher scores for the primary ‘biological aging aroma’ attribute, which is characterized by notes of nuts and apple, typical of this wine style.
Finally, a life-cycle assessment (LCA) quantified significant sustainability and economic advantages, revealing that even a minor reduction in the final alcohol content can lead to considerable savings in costs, energy consumption, and carbon dioxide equivalent emissions for a given volume of wine. Therefore, static biological aging at a reduced strength represents a sustainable method for producing lower-alcohol sherry wines that successfully retain and even amplify their characteristic Fino profile. However, its successful implementation is contingent upon rigorous microclimatic control to mitigate the increased microbial competition. Thus, further research is necessary to determine the optimal operational conditions and to examine specific aspects, such as how this reduction in alcoholic strength affects the flor veil microbiota, in order to fully understand and optimize the process.
Author contributions
Conceptualization, CL and JM; methodology, CL and JM; software, LV-E and VP; formal analysis, RM-C and LV-E; investigation, CL and LV-E; resources, CL and JM; writing—original draft preparation, LV-E, VP and CL; writing—review and editing, CL and VP; visualization, LV-E and VP; supervision, CL and JM; funding acquisition: CL and JM. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included within the article and/or in supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5fb00733j.Acknowledgements
This research was funded by JUNTA ANDALUCIA (SPAIN) through the projects: Program: Specialized Transfer Actions Universities-CEIS-RIS3-FEDER. Singular Project AGROMIS-ceiA3, SUB-SECTOR: TRADITIONAL WINES OF ANDALUCIA. Call 2020: Grants for R+D+i projects in the field of Innovation Ecosystems of the International Centers of Excellence (PY2020 ECOSIS INNOV CEIS). This research was also funded by JUNTA ANDALUCIA (SPAIN) through the Project JA-CAPDR: Ayudas a la creación y el funcionamiento de grupos operativos de la Asociación Europea de Innovación (AEI) en materia de productividad y sostenibilidad agrícolas. Línea 1: Operación 16.1.2. Ayudas al funcionamiento de los grupos operativos de la AEI en materia de productividad y sostenibilidad agrícola. Grant number GOPG-CO-23-0007 INNOFINO. “Implementación de prácticas innovadoras para reducción de grado alcohólico vinos tipo Fino Andaluces, preservando la calidad del vino”. The authors gratefully acknowledge the support of González Byass, Williams Humbert, and Yuste wineries for their invaluable collaboration, specifically for the provision of wine samples and essential technical support required for this research. Special thanks are extended to the Regulatory Council of the PDOs Jerez-Xérès-Sherry and Manzanilla de Sanlúcar for their administrative facilitation and their collaboration in the dissemination of the research findings. This work was also made possible with the essential support provided by the Campus de Excelencia Internacional Agroalimentario (ceiA3).References
- C. Charpentier, A. Colin, A. Alais and J. L. Legras, Antonie van Leeuwenhoek, 2009, 95, 263–273 CrossRef PubMed.
- D. Avdanina and A. Zghun, Fermentation, 2022, 8, 381 CrossRef CAS.
- G. Petretto, P. P. Urgeghe, R. Cabizza, A. Del Caro and A. Del Caro, Eur. Food Res. Technol., 2023, 249, 1887–1897 CrossRef CAS.
- J. L. Legras, J. Moreno-Garcia, S. Zara, G. Zara, T. Garcia-Martinez, J. C. Mauricio, I. Mannazzu, A. L. Coi, M. B. Zeidan, S. Dequin, J. Moreno and M. Budroni, Front. Microbiol., 2016, 7, 191049 Search PubMed.
- H. Alexandre, Int. J. Food Microbiol., 2013, 167, 269–275 CrossRef CAS PubMed.
- I. Cañas, E. G. Navia-Osorio, C. Porras-Amores and F. R. Mazarrón, Innovative Food Sci. Emerging Technol., 2022, 82, 103164 CrossRef.
- G. Cordero-Bueso, M. Ruiz-Muñoz, M. González-Moreno, S. Chirino, M. del C. Bernal-Grande and J. M. Cantoral, Fermentation, 2018, 4, 19 CrossRef.
- B. Esteve-Zarzoso, M. J. Peris-Torán, E. Garcıa-Maiquez, F. Uruburu and A. Querol, Appl. Environ. Microbiol., 2001, 67, 2056–2061 CrossRef CAS PubMed.
- S. Zara, A. T. Bakalinsky, G. Zara, G. Pirino, M. A. Demontis and M. Budroni, Appl. Environ. Microbiol., 2005, 71, 2934–2939 CrossRef CAS PubMed.
- M. Ruiz-Muñoz, G. Cordero-Bueso, F. Benítez-Trujillo, S. Martínez, F. Pérez and J. M. Cantoral, Food Microbiol., 2020, 92, 103553 CrossRef.
- M. Ruiz-Muñoz, M. Hernández-Fernández, G. Cordero-Bueso, S. Martínez-Verdugo, F. Pérez and J. M. Cantoral, Fermentation, 2022, 8, 456 CrossRef.
- M. Marin-Menguiano, S. Romero-Sanchez, R. R. Barrales and J. I. Ibeas, Int. J. Food Microbiol., 2017, 244, 67–73 CrossRef CAS PubMed.
- P. Martínez, A. C. Codón, L. Pérez and T. Benítez, Yeast, 1995, 11, 1399–1411 CrossRef PubMed.
- A. G. Cordente, C. D. Curtin, C. Varela and I. S. Pretorius, Appl. Microbiol. Biotechnol., 2012, 96, 601–618 CrossRef CAS.
- A. Blandino, I. Caro and D. Cantero, Biotechnol. Lett., 1997, 19, 651–654 CrossRef CAS.
- J. A. Regodón Mateos, F. Pérez-Nevado and M. Ramírez Fernández, Enzyme Microb. Technol., 2006, 40, 151–157 CrossRef.
- M. V. García-Moreno, M. M. Sánchez-Guillén, M. J. Delgado-González, E. Durán-Guerrero, M. C. Rodríguez-Dodero, C. García-Barroso and D. A. Guillén-Sánchez, LWT, 2021, 140, 110706 Search PubMed.
- M. Ruiz-Muñoz, G. Cordero-Bueso, S. Martínez, F. Pérez, D. Hughes-Herrera, I. Izquierdo-Bueno and J. M. Cantoral, BIO Web Conf., 2019, 15, 02018 Search PubMed.
- T. M. Berlanga, R. Peinado, C. Millán, J. C. Mauricio and J. M. Ortega, J. Agric. Food Chem., 2004, 52, 2577–2581 CrossRef CAS PubMed.
- M. J. Valcárcel-Muñoz, M. Guerrero-Chanivet, M. D. C. Rodríguez-Dodero, M. d. V. García-Moreno and D. A. Guillén-Sánchez, Molecules, 2022, 27, 365 CrossRef PubMed.
- J. Moreno-García, R. M. Raposo and J. Moreno, Food Res. Int., 2013, 54, 285–292 CrossRef.
- C. Lasanta, R. Muñoz-Castells, J. Gómez and J. Moreno, BIO Web Conf., 2023, 56, 02032 CrossRef.
- C. Lasanta, J. Moreno, R. Muñoz-Castells, R. Arnedo, P. Medina, M. Villa and J. Gómez, BIO Web Conf., 2023, 68, 02007 CrossRef.
- R. Muñoz-Castells, L. Vega-Espinar, J. Carlos García-García, T. Alcalá-Jiménez, J. Moreno-García, C. Lasanta and J. Moreno, Fermentation, 2025, 11, 575 Search PubMed.
- European Comisión Implementan Regulation (EU) 2025/1713 of 29 July 2025 (OJEU L 1713 of 20 August 2025) approving the amendment of the specifications of the Jerez-Xérès-Sherry PDO, Accessed 10 October 2025.
- European Comisión Implementan Regulation (EU) 2025/1714 of 29 July 2025 (OJEU L 1713 of 20 August 2025) approving the amendment of the specifications of the manzanilla de sanlucar PDO, Accessed 21 October 2025.
- L. Pinto da Silva and J. C. G. Esteves da Silva, Clean. Circ. Bioeconomy, 2022, 2, 100021 Search PubMed.
- J. M. Sánchez-Ruiz, M. Á. Pozo-Bayón and M. V Moreno-Arribas, OENO One, 2022, 56, 355–369 Search PubMed.
- V. Palacios, A. Roldán, A. Jiménez-Cantizano and A. Amores-Arrocha, PLoS One, 2018, 13, e0208330 Search PubMed.
- Organisation Internationale de la Vigne et du Vin (OIV), Compendium of International Methods of Analysis of Wines and Musts, OIV, Paris, 2025, Accessed 7 October 2025 Search PubMed.
- M. d. V. Palenzuela, N. López de Lerma, F. Sánchez-Suárez, R. Martínez-García, R. A. Peinado and A. Rosal, Appl. Sci., 2023, 13, 8001 Search PubMed.
- International Organization for Standardization, ISO 3591:1977 — Sensory Analysis — Apparatus — Wine-Tasting Glass, 1977, https://www.iso.org/standard/9009.html Search PubMed.
- P. Sancho-Galán, A. Amores-Arrocha, A. Jiménez-Cantizano and V. Palacios, Molecules, 2019, 24, 1763 Search PubMed.
- J. Moreno, J. Moreno-García, B. López-Muñoz, J. C. Mauricio and T. García-Martínez, Food Chem., 2016, 213, 90–97 Search PubMed.
- E. M. De la ossa, I. Caro, M. Bonat, L. PéREZ and B. Domecq, Am. J. Enol. Vitic., 1987, 38, 321–325 Search PubMed.
- E. M. De La Ossa, L. Pérez and I. Caro, Am. J. Enol. Vitic., 1987, 38, 293–297 Search PubMed.
- P. Martínez, L. Pérez Rodríguez and T. Benítez, Am. J. Enol. Vitic., 1997, 48, 55–62 Search PubMed.
- P. Martínez, L. P. Rodríguez and T. Benítez, Am. J. Enol. Vitic., 1997, 48, 160–168 Search PubMed.
- G. S. Drysdale and G. H. Fleet, Am. J. Enol. Vitic., 1988, 39, 143–154 CrossRef CAS.
- E. J. Bartowsky, Lett. Appl. Microbiol., 2009, 48, 149–156 CrossRef CAS PubMed.
- I. Magyar, D. Nyitrai-Sárdy, A. Leskó, A. Pomázi and M. Kállay, Int. J. Food Microbiol., 2014, 178, 1–6 CrossRef CAS PubMed.
- A. M. Roldán, F. Sánchez-García, L. Pérez-Rodríguez and V. M. Palacios, Foods, 2021, 10, 1–22 CrossRef PubMed.
- J. Moreno-García, R. M. Raposo and J. Moreno, Food Res. Int., 2013, 54, 285–292 CrossRef.
- J. A. Moreno, L. Zea, L. Moyano and M. Medina, Food Control, 2005, 16, 333–338 CrossRef CAS.
- M. L. Morales, M. Ochoa, M. Valdivia, C. Ubeda, S. Romero-Sanchez, J. I. Ibeas and E. Valero, Food Res. Int., 2020, 128, 108771 CrossRef CAS PubMed.
- L. Zea, M. P. Serratosa, J. Mérida and L. Moyano, Compr. Rev. Food Sci. Food Saf., 2015, 14, 681–693 CrossRef CAS.
- M. Del Carmen Plata, J. C. Mauricio, C. Millán and J. M. Ortega, J. Ferment. Bioeng., 1998, 85, 369–374 CrossRef CAS.
- P. Romano and G. Suzzi, Appl. Environ. Microbiol., 1996, 62, 309–315 CrossRef CAS PubMed.
- T. M. Berlanga, R. Peinado, C. Millán, J. C. Mauricio and J. M. Ortega, J. Agric. Food Chem., 2004, 52, 2577–2581 CrossRef CAS PubMed.
- P. Martínez, M. J. Valcárcel, L. Pérez and T. Benítez, Am. J. Enol. Vitic., 1998, 49, 240–250 CrossRef.
- P. Martínez, L. Pérez Rodríguez and T. Bénitez, Syst. Appl. Microbiol.l, 1997, 20, 154–157 Search PubMed.
- X. K. Zhang, P. T. Liu, X. W. Zheng, Z. F. Li, J. P. Sun, J. S. Fan, D. Q. Ye, D. M. Li, H. Q. Wang, Q. Q. Yu and Z. Y. Ding, Molecules, 2024, 29, 4279 CrossRef CAS PubMed.
- A. Genovese, N. Caporaso and L. Moio, Appl. Sci., 2021, 11, 7767 Search PubMed.
- C. Virdis, K. Sumby, E. Bartowsky and V. Jiranek, Front. Microbiol., 2021, 11, 612118 Search PubMed.
- P. Sancho-Galán, A. Amores-Arrocha, V. Palacios and A. Jiménez-Cantizano, Agronomy, 2020, 10, 205 CrossRef.
- C. Ubeda, I. Kania-Zelada, R. del Barrio-Galán, M. Medel-Marabolí, M. Gil and Á. Peña-Neira, Food Res. Int., 2019, 119, 554–563 CrossRef CAS PubMed.
- D. Muñoz, R. A. Peinado, M. Medina and J. Moreno, Eur. Food Res. Technol., 2006, 222, 629–635 Search PubMed.
- G. H. Fleet, Wine Microbiology and Biotechnology - Google Libros, 1993 Search PubMed.
- A. Mallouchos, M. Komaitis, A. Koutinas and M. Kanellaki, J. Agric. Food Chem., 2002, 50, 3840–3848 Search PubMed.
- J. I. Ibeas, I. Lozano, F. Perdigones and J. Jimenez, Am. J. Enol. Vitic., 1997, 48, 75–79 CrossRef.
- J. J. Moreno, C. Millán, J. M. Ortega and M. Medina, J. Ind. Microbiol., 1991, 7, 181–189 Search PubMed.
- H. Hata, S. Shimizu and H. Yamada, Agric. Biol. Chem., 1987, 51, 3011–3016 CAS.
- L. Zea, J. Moreno, J. M. Ortega, J. C. Mauricio and M. Medina, Biotechnol. Lett., 1995, 17, 1351–1356 Search PubMed.
- L. Moyano, L. Zea, J. Moreno and M. Medina, J. Agric. Food Chem., 2002, 50, 7356–7361 CrossRef CAS PubMed.
- L. J. Pérez-Prieto, J. M. López-Roca, A. Martínez-Cutillas, F. Pardo-Mínguez and E. Gómez-Plaza, J. Agric. Food Chem., 2003, 51, 5444–5449 Search PubMed.
- M. Cano-López, F. Pardo-Minguez, J. M. López-Roca and E. Gómez-Plaza, Am. J. Enol. Vitic., 2006, 57, 325–331 CrossRef.
- L. Zea, L. Moyano, J. A. Moreno and M. Medina, J. Sci. Food Agric., 2007, 87, 2319–2326 CrossRef CAS.
- R. A. Peinado, J. Moreno, M. Medina and J. C. Mauricio, Biotechnol. Lett., 2004, 26, 757–762 CrossRef CAS PubMed.
- P. Winterhalter and R. Rouseff, ACS Symposium Series, American Chemical Society, Washington, DC, 2001 Search PubMed.
- J. Marín, R. Ocete, M. Pedroza, A. Zalacain, C. de Miguel, M. A. López and M. R. Salinas, J. Food Compos. Anal., 2009, 22, 745–750 CrossRef.
- J. Carbonero-Pacheco, M. D. Rey, J. Moreno-García, J. Moreno, T. García-Martínez and J. C. Mauricio, Food Microbiol., 2023, 116, 12 CrossRef PubMed.
- Ministry of Agriculture, Fisheries and Food (MAPA), Report on the prices of wine alcohol and other distilled products, Government of Spain, Madrid, 2024, https://www.mapa.gob.es, Accessed 7 October 2025.
- T. Soleymani Angili, K. Grzesik, A. Rödl and M. Kaltschmitt, Energies, 2021, 14, 2–18 Search PubMed.
- O. Therasme, T. A. Volk, M. H. Eisenbies, T. E. Amidon and M. O. Fortier, Biotechnol. Biofuels, 2021, 14, 1–15 Search PubMed.
- European Environment Agency (EEA), EMEP/EEA Air Pollutant Emission Inventory Guidebook 2022: Road transport, Publications Office of the European Union, Luxembourg, 2022, https://www.eea.europa.eu/publications/emep-eea-guidebook-2022, Accessed 16 October 2025.
- Web of Consejo Regulador de la Denominación de Origen Jerez-Xérès-Sherry, Sherry Wines, https://www.sherry.wine/sherry-region/consejo-regulador, accessed 15 January 2026 Search PubMed.
- Ecoinvent, ecoinvent v3.10, https://ecoinvent.org/ecoinvent-v3-10/, Accessed 7 October 2025.
| This journal is © The Royal Society of Chemistry 2026 |