Open Access Article
Open Access ArticleEffects of high-pressure processing and conventional thermal processing on enzyme activities, antioxidant properties, and volatile metabolic profiling of “Phulae” pineapple purée
Supapich Chalumputa,
Prinya Wongsaab,
Phunsiri Suthilukab,
Matchima Naradisornab,
Daisuke Hamanakac and
Sutthiwal Setha *ab
*ab
aSchool of Agro-Industry, Mae Fah Luang University, Chiang Rai 57100, Thailand. E-mail: sutthiwal.set@mfu.ac.th
bResearch Group of Postharvest Technology, Mae Fah Luang University, Chiang Rai 57100, Thailand
cFaculty of Agriculture, Kagoshima University, 1-21-24 Korimoto, Kagoshima 890-0065, Japan
First published on 5th February 2026
Abstract
This study investigated the effects of high-pressure processing (HPP) at 400 and 600 MPa (10 min, 25 °C) compared with conventional thermal processing (CTP; 10 min, 80 °C) on the quality of “Phulae” pineapple purée (Ananas comosus L. cv. Phulae). Untreated purée served as the control. HPP effectively preserved key quality parameters of the purée, including total soluble solids (13.5 °Brix) and color (L* 46.6–48.0; b* 21.3–22.0), with only slight deviations from the untreated samples (L* 49.4; b* 24.1). The vitamin C content was higher in the HPP-treated purée (216.9 mg kg−1) than in the CTP-treated purée (167.0 mg kg−1). The antioxidant capacity was enhanced in the HPP-treated purée, particularly with HPP at 400 MPa, with a total phenolic content of 73.8 g gallic acid equivalents/kg fresh weight (FW), 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activity of 2.3 mmol Trolox equivalents (TE)/kg FW, and ferric reducing antioxidant power (FRAP) of 4.7 mmol ascorbic acid equivalents/kg FW. 2,2′-Azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) radical scavenging activity was the highest in purée treated with HPP at 600 MPa (4.6 g Trolox equivalent antioxidant capacity/kg FW). HPP also preserved enzyme-related activities, including bromelain activity (3441.7–3733.3 casein digestion units/L) and hyaluronidase (HA) inhibition (78.8–79.9%). Moreover, nitric oxide (NO) radical inhibition was significantly reduced in CTP-treated purée, while HPP-treated samples retained higher inhibition levels (47.0 mmol TE per kg FW). Volatile compound analysis identified esters and terpenes, including their various subgroups, as the dominant contributors to the aroma profile. Based on principal component analysis, principal components 1 and 2 explained 60.4% and 25.3% of the total variance, respectively, accounting for a combined 85.7% of the variability in the dataset. HPP-treated purée clustered closely with the untreated purée, indicating a preserved aroma profile. These findings highlight that HPP is a promising non-thermal technique for maintaining the nutritional, functional, and sensory qualities, especially aroma, of fruit-based products.
Sustainability spotlightHigh-pressure processing (HPP) is a sustainable, non-thermal alternative to conventional heat treatment for preserving “Phulae” pineapple puree. HPP retained vitamin C, phenolics, antioxidant activity, enzyme function, and natural aroma while reducing nutrient loss and energy consumption. By extending shelf life and minimizing the need for chemical preservatives, HPP supports cleaner production and reduces food waste, enhancing the value of local fruit resources. These outcomes contribute to SDG 2 (Zero Hunger) by improving food quality and nutrition, SDG 12 (Responsible Consumption and Production) through energy-efficient, low-waste processing, and SDG 13 (Climate Action) by promoting lower-carbon food technologies. |
1 Introduction
Pineapple (Ananas comosus L. Merr) is a tropical fruit noted for its sweet and sour flavor. It is an important commercial fruit that comes in a variety of cultivars and types1 from throughout the world. Pineapple contains abundant vitamin C, dietary fiber, bromelain, carotenoid, and phenolic compounds, which provide it with notable antioxidant activity and the ability to reduce the risk of health problems or diseases.2 The “Phulae” pineapple is a geographical indication (GI) product recognized for its sweet, crispy, and aromatic qualities, originates from the Nang-Lae Subdistrict, Chiang Rai province of Thailand and is classified under the Queen pineapple group.3 “Phulae” pineapples are harvested at peak ripeness and are primarily consumed fresh, with some being transported to local and export markets. Processing pineapples into purée adds value by extending shelf life and reducing postharvest losses. The global fruit purée market was valued at approximately USD 5.09 billion in 2024 and is projected to grow at a Compound Annual Growth Rate (CAGR) of 6.7% through 2030, driven by increasing demand for natural and minimally processed ingredients in beverages, bakery products, and infant foods.4 However, CTP can adversely affect sensory quality and degrade nutritional bioactive compounds. In response to consumer preference for clean-label, minimally processed fruit products with high nutritional value, non-thermal technologies such as pulsed ultraviolet light, irradiation, cold plasma, ultrasonication, and ozone treatment have gained increasing attention.5 Among these technologies, HPP is suitable for pineapple purée. Despite its higher cost, HPP ensures microbial safety while better preserving nutritional quality and the unique sensory attributes of “Phulae” pineapple. HPP is a non-thermal pasteurization method that applies high isostatic pressures (100–600 MPa) to inactivate spoilage and pathogenic microorganisms without the need for intense heat.6 Unlike CTP, HPP minimizes nutrient loss and quality deterioration and does not require additional energy for heat generation, making it a more energy-efficient preservation method.7 It has become increasingly popular as consumers seek minimally processed, safe, and wholesome food alternatives.8 In fruit-based products, sensory quality, particularly aroma, is a critical determinant of consumer acceptance. Pineapple aroma is primarily governed by a complex mixture of volatile compounds, including esters, lactones, terpenes, aldehydes, and sulfur-containing compounds, which collectively contribute to its characteristic fruity and sweet flavor profile.9 These volatile compounds are highly sensitive to processing conditions, especially thermal treatments, which can induce volatilization, degradation, or chemical transformation, leading to aroma loss or the formation of off-flavors. Consequently, preserving the native aromatic profile of pineapple during processing remains a significant technological challenge.10 Because HPP does not involve high temperatures, it preserves heat-sensitive nutrients such as vitamins, minerals, and enzymes, and ensures the retention of antioxidants and the integrity of essential fatty acids and bioactive compounds. Additionally, HPP can enhance the bioavailability of certain nutrients, making them more easily absorbed by the body. Previous studies have demonstrated the effectiveness of HPP in retaining bioactive compounds and antioxidant properties in kiwi berry juice, orange juice-milk, and strawberry purée.11–13 A study in pineapple juice treated with HPP at 500 MPa for 10 min retained its original color, antioxidant activity, bioactive compounds, and volatile compounds.14 Such findings highlight the potential of using HPP to process pineapple purée while retaining its nutritional and sensory quality. Overall, the nutritional benefits and functional potential of HPP are of great interest to consumers, researchers, and food manufacturers, as this non-thermal technology holds strong potential for preserving health-promoting compounds while ensuring food safety and quality.The objective of this study was to evaluate the impact of HPP at two pressures (400 and 600 MPa) and CTP on the quality, enzyme activities, antioxidant properties, and volatile metabolic profiling of “Phulae” pineapple purée. Comprehensive analyses were conducted to evaluate changes in bioactive compounds (such as vitamin C and total polyphenols), antioxidant activity (including 2,2-diphenyl-1-picrylhydrazyl [DPPH], 2,2′-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid [ABTS], and nitric oxide [NO] radical scavenging assays; the ferric reducing antioxidant power [FRAP] assay; and the oxygen radical absorbance capacity [ORAC] assay), enzyme activities (bromelain and hyaluronidase [HA]), and volatile compound profiles. These findings may help guide the selection of optimal processing technologies for maintaining both nutritional and sensory qualities in fruit-based products.
2 Materials and methods
2.1 Materials
“Phulae” pineapples were harvested at full maturity (i.e., with total soluble solids [TSS] of 13–15 °Brix) from the same orchard in Nang-Lae District, Chiang Rai Province, Thailand (19.95°N, 99.87°E, 390 m above sea level).2.2 Chemicals
All chemicals and reagents used in this study were of analytical grade, unless otherwise specified. Standards and solvents, including ascorbic acid, the Folin–Ciocalteu phenol reagent, gallic acid, DPPH, Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), and 2,4,6-tris(2-pyridyl)-s-triazine (TPTZ), among others, were purchased from Sigma-Aldrich (St. Louis, MO, USA). Methanol, glacial acetic acid, and other solvents were purchased from RCI Labscan (Bangkok, Thailand). Distilled water was used throughout the experiments. All other chemicals were obtained from KemAus and QReC and used without further purification.2.3 “Phulae” pineapple purée preparation
Fresh “Phulae” pineapples were washed with tap water, manually peeled, and cut into small pieces. To minimize enzymatic browning, the fruits were kept chilled (4 ± 2 °C) and processed rapidly. The sliced fruit was blended at 38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 30 s (Viva Collection HR2088/91, Philips, China). The purée (80 mL) was immediately packed into 100 mL screw-cap PET spouted pouches (8 × 12 cm) for further experimentation.
000 rpm for 30 s (Viva Collection HR2088/91, Philips, China). The purée (80 mL) was immediately packed into 100 mL screw-cap PET spouted pouches (8 × 12 cm) for further experimentation.
2.4 HPP
Eighty milliliters of purée was packed into a 100 mL screw-cap polyethylene terephthalate (PET) plastic spouted pouch (8 × 12 cm) and treated at 400 or 600 MPa for 10 min in a HPP apparatus (Bao Tou KeFa High Pressure Technology Co., Ltd, Baotou, China) with a capacity of 3.0 L at room temperature (25 °C). The purée were subjected to each HPP treatment. The fluid that transmitted pressure in this investigation was reverse osmosis water; the time spent applying or releasing pressure was not included in the treatment duration. The treated “Phulae” pineapple purée was analyzed immediately, with remaining samples stored at −20 °C until use.2.5 CTP
There were two controls in this study: untreated purée and purée subjected to CTP at 80 ± 5 °C for 10 min. This condition was established based on our previous research and a preliminary study, which identified them as optimal for achieving consistent microbial inactivation and enzymatic stability.2 The purée was prepared for CTP. First, the purée was heated to 80 °C and held at that temperature for 10 min, then immediately cooled in an ice-water bath to stop the reaction. Subsequently, 80 mL of purée was packed into a 100 mL screw-cap PET plastic spouted pouch (8 × 12 cm). The treated “Phulae” pineapple purée was analyzed immediately, with the remaining samples stored at −20 °C until use.2.6 Determination of physicochemical properties
2.7 Determination of bioactive compounds
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×g and 4 °C for 15 min. The supernatant was filtered through Whatman no. 4 filter paper. Four milliliters of the extracted sample was combined with 2 mL of 0.02% indophenol, 4 mL of 2% thiourea, and 2 mL of 2% 2, 4-dinitrophenylhydrazine solution; this mixture was incubated at 37 °C for 3 h. Next, 1 mL of 85% H2SO4 was added, and the absorbance at 540 nm was measured using a microplate reader (Multiskan Go, Thermo Fisher Scientific, Tokyo, Japan). The data are presented as mg ascorbic/kg extract.
000×g and 4 °C for 15 min. The supernatant was filtered through Whatman no. 4 filter paper. Four milliliters of the extracted sample was combined with 2 mL of 0.02% indophenol, 4 mL of 2% thiourea, and 2 mL of 2% 2, 4-dinitrophenylhydrazine solution; this mixture was incubated at 37 °C for 3 h. Next, 1 mL of 85% H2SO4 was added, and the absorbance at 540 nm was measured using a microplate reader (Multiskan Go, Thermo Fisher Scientific, Tokyo, Japan). The data are presented as mg ascorbic/kg extract.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v/v). The mixture was incubated at 37 °C for 30 min. Then, the absorbance at 595 nm was measured using a microplate reader (Multiskan Go, Thermo Fisher Scientific, Tokyo, Japan). Ferrous sulfate equivalent was used as a standard, and distilled water served as the blank.
1 (v/v/v). The mixture was incubated at 37 °C for 30 min. Then, the absorbance at 595 nm was measured using a microplate reader (Multiskan Go, Thermo Fisher Scientific, Tokyo, Japan). Ferrous sulfate equivalent was used as a standard, and distilled water served as the blank.ORAC is expressed as mmol TE per kg sample.
2.8 NO radical scavenging assay
The NO radical scavenging activity was analyzed as published22 previously with some modifications. In brief, NO radicals are generated from the interaction between oxygen in the sample and aqueous sodium nitroprusside (SNP) and quantified via the Griess–Illosvoy reaction, which measures nitrite ions (NO2−). First, 800 µL of 10 mM SNP and 40 µL of diluted purée were mixed and incubated at room temperature for 150 min. Then, 200 µL of the solution was transferred to a fresh tube, and 400 µL of 0.33% sulfanilamide in 20% glacial acetic acid was added; the mixture was incubated at room temperature for 5 min. Next, 400 µL of 0.1% N-(1-naphthyl) ethylene-diamine dihydrochloride was added, and the mixture was incubated at room temperature for 30 min. The absorbance at 540 nm was measured with a microplate reader (Multiskan Go, Thermo Fisher Scientific, Tokyo, Japan). The standard curve was generated from Trolox (0.2–0.5 mg mL−1). The results are presented as mmol TE per kg FW.2.9 Determination of HA inhibition
HA inhibition was measured as described in previous publications.23,24 One hundred microliters of HA (type IV-S, derived from bovine testes; Sigma-Aldrich) was diluted in buffer to a final concentration of 5 mg mL−1 and incubated at 37 °C. Subsequently, 200 µL of compound 48/80 (Sigma-Aldrich) was added to the buffer at a final concentration of 0.5 mg mL−1 and incubated at 37 °C for 20 min. Freeze-dried samples were reconstituted in distilled water, vortexed, and centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min; added to the reaction mixture; and incubated at 37 °C for 20 min. Hyaluronic acid sodium salt (extracted from rooster comb; Wako, Osaka, Japan) was added to a final concentration of 0.4 mg mL−1, and the mixture was incubated at 37 °C for 40 min. To terminate the reaction, 200 µL of 0.4 M NaOH and 200 µL of borate buffer (pH 8.0) were added, and the mixture was boiled in water for 3 min before being rapidly cooled on ice. Subsequently, 3000 µL of p-dimethylaminobenzaldehyde (Wako) solution was introduced, followed by incubation at 37 °C for 20 min. The absorbance at 585 nm was measured using a SpectraMax mini plate reader equipped with SoftMax Pro 7.2 software (Molecular Devices San Jose, California, United States), and HA inhibition (%) was calculated.
000 rpm for 10 min; added to the reaction mixture; and incubated at 37 °C for 20 min. Hyaluronic acid sodium salt (extracted from rooster comb; Wako, Osaka, Japan) was added to a final concentration of 0.4 mg mL−1, and the mixture was incubated at 37 °C for 40 min. To terminate the reaction, 200 µL of 0.4 M NaOH and 200 µL of borate buffer (pH 8.0) were added, and the mixture was boiled in water for 3 min before being rapidly cooled on ice. Subsequently, 3000 µL of p-dimethylaminobenzaldehyde (Wako) solution was introduced, followed by incubation at 37 °C for 20 min. The absorbance at 585 nm was measured using a SpectraMax mini plate reader equipped with SoftMax Pro 7.2 software (Molecular Devices San Jose, California, United States), and HA inhibition (%) was calculated.
2.10 Determination of enzyme activity
Bromelain activity was determined by quantifying the low-molecular-weight digestion products, which are soluble in trichloroacetic acid (TCA), resulting from its proteolytic action. The assay was performed based on a previous publication.25 It was conducted using 5 mL of 0.75% casein prepared in anhydrous disodium phosphate buffer (50 mM, pH 7, with the pH adjusted by gradually adding 0.1 N HCl). The solution was preincubated at 37 °C for 10 min before introducing the enzyme, which had been diluted to a final volume of 1 mL using an activating buffer (20.7 mM L-cysteine in 6 mM disodium ethylenediaminetetraacetic acid, pH 7). The proteolytic reaction was terminated after 10 min by adding 5 mL of 30% (w/v) TCA, and the mixture was incubated at 37 °C for 30 min. Then, the solution was cooled to room temperature and filtered twice using Whatman no. 42 filter paper. The absorbance of the resulting filtrate was measured at 280 nm using an ultraviolet-visible (UV-Vis) spectrophotometer (U-2900, Hitachi High-Technologies Corporation, Tokyo, Japan). The enzymatic activity was quantified based on a standard calibration curve of absorbance versus the tyrosine concentration; it is presented as casein digestion units (CDU) per liter.2.11 Determination of volatile compounds
Headspace solid phase microextraction (SPME) combined with gas chromatography-mass spectrometry (GC-MS) was performed as described previously,14 with slight modifications, to extract and analyze the volatile compounds. Five grams of pineapple purée was added to 20 mL headspace glass vials. The volatile compounds present in the headspace were absorbed to an SPME fiber (Supelco, Bellefonte, PA, USA) coated with 50/30 µm divinylbenzene/carboxen/polydimethylsiloxane (DVB/CAR/PDMS) for 30 min at 40 °C. The gases were extracted and then thermally desorbed in the injection inlet of a GC-MS system (7890A/5975C, Agilent Technologies Co., Ltd, Santa Clara, CA, USA) for 6 min at 270 °C. The samples were injected in splitless mode. A HP-5MS capillary (30 m × 0.25 mm × 0.25 µm; Agilent Technologies) was used to separate volatile chemicals. The carrier gas was helium, with a constant flow rate of 1.0 mL min−1. The oven temperature was initially set at 40 °C for 2 min, increased to 220 °C at 5 °C min−1, increased to 260 °C at 10 °C min−1, and then maintained at 260 °C for 2 min. The mass spectra were obtained using electron impact ionization mode, with the ion source operating at 230 °C. ChemStation software (Agilent Technologies) was used for peak area integration and to visualize chromatograms. n-Alkanes (C8–C20) obtained from Sigma–Aldrich, Germany were also run. The volatile components were tentatively identified by comparing the mass spectra of the samples with the retention index (RI), authentic references, and the data system library (NIST 20). The RI was calculated from the following formula:where tR, tRz, and tR(z + 1) are the elution time of the unknown compound, the n-alkane that eluted just before the unknown compound, and the n-alkane that eluted just after the unknown compound, respectively; and z is the number of carbon atoms in pre-eluted hydrocarbon RI.
2.12 Statistical analysis
Data are presented as the mean ± standard deviation (SD) of three independent batches, with each analysis performed in triplicate. The data were also subjected to analysis of variance (ANOVA) followed by Duncan's multiple range tests using IBM SPSS Statistics 20.0 (IBM Corp., Armonk, NY, USA). P < 0.05 was considered to indicate a statistically significant difference. The principal component analysis (PCA) was calculated for volatile composition. The concentration data were log-transformed and Pareto-scaled before being analyzed with MetaboAnalyst 6.0 (https://www.metaboanalyst.ca, accessed on August 12, 2024).3 Results and discussion
3.1 Effect of HPP on physicochemical properties
Untreated “Phulae” pineapple purée had a pH of 3.96 ± 0.01, TSS of 13.54 ± 0.01 °Brix, and TA of 0.44%, consistent with data from the literature.26 These baseline values were used to assess the impact of HPP and CTP on “Phulae” pineapple purée. The TSS of the untreated control and HPP-treated purées did not differ significantly (P > 0.05) (Table 1). Moreover, there was no significant difference between HPP treatment at 400 and 600 MPa. These findings suggest that HPP may not significantly impact soluble solids, similar to the findings from passion fruit purée.27 However, TSS and TA increased significantly (P < 0.05) in the CTP-treated purées compared to the untreated purées (Table 1). The increase in TSS during CTP was primarily attributed to the dehydration of the purée. The thermal energy applied during CTP likely facilitated moisture evaporation, thereby concentrating the soluble solids.28 This physical concentration effect is a dominant factor, although it may be partially supplemented by biochemical reactions, such as the hydrolysis of complex polysaccharides into soluble sugars during heating.29 This explanation is further supported by a comparison with mango smoothies, where TSS and pH remained stable under HPP. The stability observed in the mango study highlights that without dehydration, the intrinsic chemical changes in such fruit matrices are insufficient to significantly alter TSS, confirming that the elevation observed in this study is a specific result of the dehydration effect inherent to the CTP process.30 The pH decreased significantly (P < 0.05) in the CTP-treated and HPP-treated purées compared with the untreated control purées. Finally, the TA tended to increase in all treated purées compared with the untreated control purée. This increase could be due to the disruption of cellular membranes by heat and/or high pressure, leading to the release of organic acids from within the cells, which in turn increases the acid concentration and slightly decreases the pH.31| Treatments | pH | TSS (°Brix) | TA | L* | a* | b* |
|---|---|---|---|---|---|---|
| a Different letters in the same column indicate a significant difference (p < 0.05) among treatments. CTP, conventional thermal processing at 80 °C for 10 min; HPP-400, high-pressure processing at 400 MPa for 10 min; HPP-600, high-pressure processing at 600 MPa for 10 min; TA, total acidity; TSS, total soluble solids; L*, lightness; a*, redness; b*, yellowness. | ||||||
| Untreated | 3.96 ± 0.01a | 13.54 ± 0.11b | 0.44 ± 0.02c | 49.41 ± 0.39a | −2.41 ± 0.23a | 24.11 ± 1.10a |
| CTP | 3.94 ± 0.01b | 14.36 ± 0.23a | 0.51 ± 0.03a | 45.08 ± 0.78d | −2.34 ± 0.21a | 19.69 ± 0.89c |
| HPP 400 | 3.92 ± 0.01c | 13.51 ± 0.12b | 0.48 ± 0.01b | 46.63 ± 0.81c | −2.40 ± 0.21a | 22.04 ± 0.87b |
| HPP 600 | 3.92 ± 0.01c | 13.61 ± 0.14b | 0.46 ± 0.00b | 48.02 ± 0.83b | −2.71 ± 0.15b | 21.27 ± 0.90b |
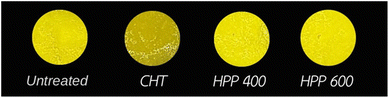
Regarding the color parameters, treatment with HPP, especially at 600 MPa, minimized color changes in the pineapple purée compared with CTP (Fig. 1). HPP provided better preservation of the lightness (L*) and yellowness (b*) values, indicating less color alteration. This effect may be partly attributed to the high sugar content of pineapple purée. During thermal processing, sugars promote non-enzymatic browning via caramelization and Maillard reactions. Furthermore, in non-thermal processes like HPP, high sugar concentrations can exert a protective effect on browning-related enzymes.32 Our finding is consistent with the results observed for strawberry and blackberry purées,33 where HPP also resulted in minimal color deviations.34 This result is also similar to a previous study that showed that HPP retained the original color of mango smoothies.30 On the other hand, CTP caused more pronounced color shifts in the purée, particularly a reduction in b* values, signifying a shift toward a more yellow hue. This was likely due to the alteration of pigments by heat and the precipitation of insoluble particles.2
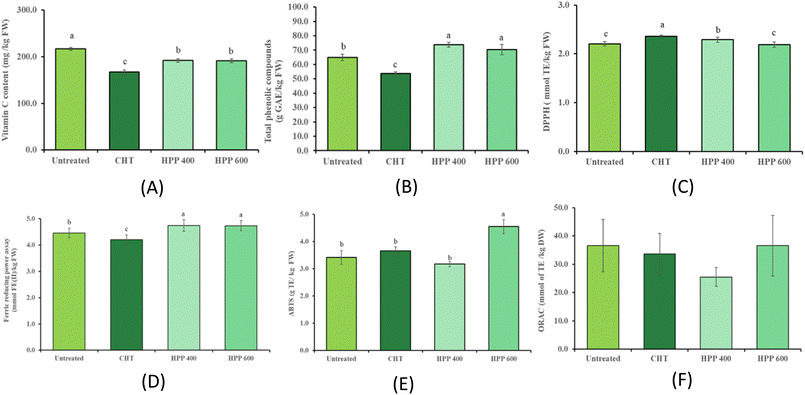
3.2 Effect of HPP on bioactive compounds
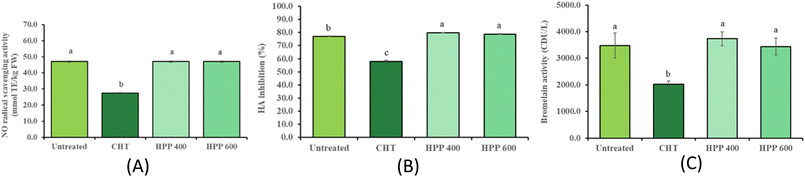
3.3 Effect of HPP on NO radical scavenging activity
The NO radical scavenging activity did not differ significantly (P > 0.05) between the untreated purée (47.0 mmol TE per kg FW) and the HPP-treated purée (47.0 mmol TE per kg FW), indicating that HPP preserves the compounds responsible for NO scavenging. In contrast, CTP-treated purée showed the lowest activity (27.4 mmol TE per kg FW; Fig. 3A), possibly due to the heat-induced degradation of bioactive compounds. These assays are commonly used as preliminary indicators linked to inflammatory pathways; however, they are based on in vitro chemical measurements and do not directly reflect physiological anti-inflammatory effects in vivo. This decline is likely attributed to the thermal degradation of heat-sensitive bioactive compounds, such as bromelain and phenolic antioxidants, which possess the functional groups necessary for electron or hydrogen atom transfer to NO radicals.41 Consistently, previous studies have demonstrated that HPP retains the stability of phenolic acids and vitamin C, which are key contributors to the NO scavenging capacity in fruit matrices.14 The lack of significant change in NO radical scavenging activity for HPP supports the notion that HPP provides a gentler processing mechanism compared with heat treatment, and thus it may preserve these anti-inflammatory compounds.3.4 Effect of HPP on HA inhibition
The untreated purée showed 77.1% HA inhibition. Following CTP treatment, this activity was significantly (P < 0.05) reduced to 58.0% (Fig. 3B). This substantial decline in inhibitory potential is largely attributed to the thermal sensitivity of bromelain and other bioactive proteases, where heat-induced unfolding of the protein structure disrupts the active sites responsible for HA interaction.42 In contrast, HPP at 400 and 600 MPa preserved HA inhibition effectively (78.8–79.9%), showing no significant degradation compared to the untreated. Bromelain is known for its ability to inhibit HA activity, which plays a role in its anti-inflammatory and anti-metastatic effects.43 Heat can denature bromelain, significantly reducing its bioactivity,42 whereas HPP has been reported to preserve its molecular conformation and enzymatic function.44 These findings are consistent with previous studies emphasizing the advantages of HPP over heat treatments in retaining enzyme activity and biofunctional properties in fruits.45,46 They suggest that HPP, a non-thermal preservation technique, better retains the functional integrity of bioactive compounds responsible for HA inhibition, likely due to minimal structural damage to proteins and enzymes.3.5 Effect of HPP on enzyme activity
Bromelain activity in untreated purée was 3483.3 CDU/L, serving as the baseline for this study. Following CTP treatment, activity declined significantly (P < 0.05) to 2025 CDU/L, representing a loss of nearly 40% compared to the control (Fig. 3C). This reduction is primarily attributed to the thermal denaturation of bromelain's protein structure, where high temperatures disrupt the hydrogen bonds and hydrophobic interactions necessary for retaining its active site conformation.44 In contrast, HPP-treated samples (3441.7–3733.3 CDU/L) showed no significant difference from the untreated samples, highlighting HPP's ability to preserve enzymatic function. The stability of bromelain under high pressure can be explained by the fact that HPP primarily affects large molecular weight components through volume contraction but often leaves low-energy covalent bonds intact, thus retaining the enzyme's catalytic integrity.44 One study indicated that HPP treatment can significantly enhance the in vivo fibrinolytic activity of fruit-derived bromelain, thereby increasing the functional value and market potential of pineapple-based products.47 When interpreted alongside HA inhibition data, it becomes evident that the retention of bromelain activity under HPP conditions directly contributes to the preservation of pineapple's bioactive potential.3.6 Effect of HPP on volatile compounds
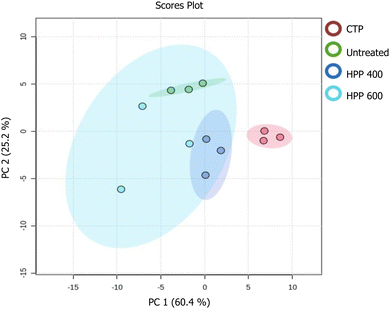
Principal component analysis (PCA) was performed to examine the effects of treatments on the volatile compound composition. As illustrated in the score plot (Fig. 4) and the corresponding biplot (Fig. 5), the first two principal components (PC1 and PC2) accounted for 60.4% and 25.2% of the total variance, respectively. The samples were categorized based on their volatile profiles. The PCA revealed significant differences between the treatments, indicating that the processing methods impacted the aroma profile of the pineapple purée (Table 2). A total of 18 volatile compounds were detected in the pineapple purée, predominantly esters and terpenes. These findings are consistent with a previous study that reported esters are the most common volatile compounds in pineapple.9 The untreated purée (green circles) exhibited the highest concentration of methyl propionate (35.2%), a compound responsible for sweet, rum-like, fruity odors. This is consistent with consumer preferences for fresh pineapple flavor.32 In contrast, the CTP-treated purée (red circles) showed a marked reduction in methyl propionate (4.5%) and a substantial increase in methyl 2-methylbutanoate (59.0%). The latter ester has an ethereal, fruity, and pineapple-like aroma. The change in volatile compounds indicates that although CTP can boost some pleasant fruity aromas, it may also reduce others, which could affect the overall flavor profile of the pineapple purée.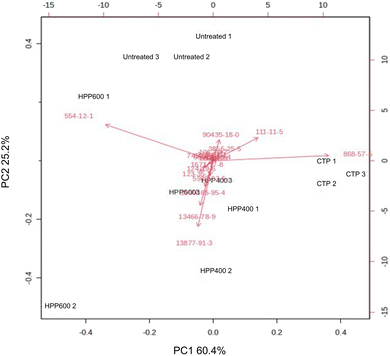 | ||
| Fig. 5 The biplot obtained from principal component analysis (PCA), with 18 metabolites labeled 1–18. See Table 2 for the identity of each metabolite. CTP, conventional thermal processing at 80 °C for 10 min; HPP-400, high-pressure processing at 400 MPa for 10 min; HPP-600, high-pressure processing at 600 MPa for 10 min. | ||
| No. | Compounds | Odor description | Classification | CAS no. | RI | Peak area (%) | |||
|---|---|---|---|---|---|---|---|---|---|
| Untreated | CTP | HPP 400 MPa | HPP 600 MPa | ||||||
| a CAS no., chemical abstracts service number; CTP, conventional thermal processing; HPP, high-pressure processing; RI, retention index. | |||||||||
| 1 | Methyl propionate | Sweet, rum-like, fruity | Ester | 554-12-1 | 611 | 35.24 | 4.54 | 19.06 | 36.40 |
| 2 | Isovaleric acid | Sweaty, cheesy, rancid | Carboxylic acid | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 435-18-0 435-18-0 |
651 | 3.31 | 1.58 | 0.85 | 0.10 |
| 3 | Butanoic acid, 2-methyl-, methyl ester | Ethereal, fruity, tutti-frutti, pineapple-like | Ester | 868-57-5 | 689 | 36.97 | 59.02 | 45.43 | 23.88 |
| 4 | Butanoic acid, 2-methyl-, ethyl ester | Fruity, juicy, tropical | Ester | 7452-79-1 | 856 | 1.92 | 0.03 | 2.08 | 0.80 |
| 5 | Hexanoic acid, methyl ester | Fruity, pineapple-like | Ester | 106-70-7 | 931 | 0.43 | — | — | — |
| 6 | Beta-myrcene | Earthy, spicy, citrusy, piney, evergreen | Monoterpene | 123-35-3 | 998 | 0.47 | — | 0.75 | 0.75 |
| 7 | 1-Pentanone, 1-(4-methylphenyl) | Raspberry-like | Ketone | 1671-77-8 | 1033 | 0.04 | — | — | — |
| 8 | D-limonene | Citrusy | Monoterpene | 5989-27-5 | 1036 | 0.53 | 0.66 | 0.93 | 0.85 |
| 9 | 3-Carene | Sweet, pungent | Monoterpene | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 466-78-9 466-78-9 |
1055 | 4.48 | 6.84 | 8.66 | 9.23 |
| 10 | Beta-ocimene | Floral, green, herbal | Monoterpene | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 877-91-3 877-91-3 |
1056 | 8.78 | 14.35 | 19.29 | 18.80 |
| 11 | Alpha-phellandrene | Pepper, mint, and fruity | Monoterpene | 99-83-2 | 1067 | 0.03 | — | — | — |
| 12 | Nonanal | Rose-orange | Aldehyde | 124-19-6 | 1112 | — | — | 0.56 | 0.63 |
| 13 | Octanoic acid, methyl ester | Fruity, orange-like, pineapple-like | Ester | 111-11-5 | 1123 | 6.30 | 9.93 | — | — |
| 14 | Octanoic acid, ethyl ester | Fruity, orange-like, pineapple-like | Ester | 106-32-1 | 1132 | 0.13 | — | — | — |
| 15 | Isoledene | Woody, musky | Terpene | 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 910-36-4 910-36-4 |
1224 | 0.12 | — | — | — |
| 16 | Decanoic acid, methyl ester | Fruity, orange-like | Ester | 110-42-9 | 1233 | 0.04 | — | — | — |
| 17 | Copaene | Woody, spicy | Terpene | 3856-25-5 | 1289 | 1.13 | 1.19 | — | — |
| 18 | Cis-muurola-3,5-diene | Woody, earthy, herbal | Terpene | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 365-95-4 365-95-4 |
1297 | 0.03 | 1.85 | 1.64 | 3.06 |
The purée subjected to HPP at 400 MPa (dark blue circles) showed intermediate values for both methyl propionate (19.1%) and methyl 2-methylbutanoate (45.4%), which suggests that HPP at 400 MPa at least partially preserves the pineapple purée aroma profile. In contrast, the purée subjected to HPP at 600 MPa (light blue circles) showed a higher concentration of methyl 2-methyl butanoate (23.9%) but a lower concentration of methyl propionate (36.4%) compared with untreated purée. This indicates that while high pressure (600 MPa) may enhance some fruity aromas, it may not fully retain the original volatile character of the untreated pineapple purée. The HPP treatments also led to the release of monoterpenes, such as β-myrcene and limonene (Table 2), which are associated with citrusy and piney aromas. These compounds were present only in the HPP-treated purée, suggesting that HPP might contribute to a more complex and diverse aroma profile. The biplot reveals a clear separation between treatments based on their volatile profiles. The HPP-treated samples were characterized by a high abundance of fresh-fruity esters, such as methyl octanoate, methyl 2-methylbutanoate, and methyl propionate, which were positioned far from the origin and clustered near the HPP group along PC1. These compounds acted as the primary drivers differentiating HPP samples from the CTP group. “Phulae” pineapple contains around 30 volatile chemicals, including esters, terpenes, aldehydes, ketones, and hydrocarbons. Esters such as hexanoic acid methyl ester, butanoic acid 2-methylmethyl ester, and octanoic acid methyl ester are key components contributing to the pineapple flavor.48 The observed changes in the specific volatile compounds likely contribute to the sensory differences between pineapple purée subjected to the different processing treatments. For example, while HPP can preserve or enhance certain fruity and citrusy aromas, CTP may result in the reduction of desirable volatiles, thus altering the overall aroma profile. Further research into consumer perception of these aroma changes would be valuable to optimize processing techniques to achieve an ideal balance between aroma preservation and shelf-life extension.
4 Conclusion
In conclusion, HPP offers significant advantages over CTP in preserving the overall quality and nutritional integrity of “Phulae” pineapple purée. As a non-thermal technology, HPP more effectively retained key physicochemical properties, including pH, total soluble solids, and color. It also demonstrated superior retention of sensitive nutritional and functional components. Specifically, HPP increased the vitamin C content by approximately 22–25% and bromelain activity by 80–95% compared with CTP, highlighting its effectiveness in preserving thermolabile nutrients and enzymes. Furthermore, antioxidant capacity assessed through FRAP, DPPH, and ABTS radical scavenging assays was significantly improved in HPP-treated purée, especially at higher pressure levels. HPP also helped preserve volatile aromatic compounds, particularly esters and terpenes, which are essential for the fresh and fruity aroma profile of pineapple. HPP-treated purée showed higher in vitro chemical activities, including a 54% increase in NO radical scavenging and a 30% increase in HA inhibition.The results indicate that HPP is a technical alternative to thermal processing for “Phulae” pineapple purée as it retains the fruit's characteristic bioactive profile and aromatic compounds. These findings provide a basis for the implementation of high-pressure technologies in the production of minimally processed fruit products where retaining natural nutritional value is prioritized. Therefore, further studies using cellular or animal models are recommended to substantiate definitive biological claims, along with investigations into shelf-life stability and long-term bioactivity retention.
Author contributions
Supapich Chalumput: methodology, formal analysis, data curation, writing – original draft, writing – review & editing; Prinya Wongsa, Phunsiri Suthiluk, Daisuke Hamanaka, and Matchima Naradisorn: conceptualization, investigation, project administration, resources, supervision; Sutthiwal Setha: conceptualization, methodology, formal analysis, investigation, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that the data supporting the findings of this study are available within the article.Acknowledgements
This research was also supported by Mae Fah Luang University under the National Science, Research, and Innovation Fund (NSRF) 2023 [grant 662A04020]. The authors would like to thank Mae Fah Luang University, Chiang Rai, Thailand, and Kagoshima University, Japan, for providing research facilities. The authors thank Professor Satoru Kondo for his kind advice on manuscript preparation and language editing support.References
- P. Esquivel, J. Usaga, R. Schweiggert, C. B. Steingass and V. M. Jiménez, ACS Food Sci. Technol., 2022, 2(3), 455–473, DOI:10.1021/acsfoodscitech.1c00414.
- P. Suthiluk, N. Chuensombat, S. Setha and M. Naradisorn, Front. Sustain. Food Syst., 2023, 7, 979943, DOI:10.3389/fsufs.2023.979943.
- M. Naradisorn, K. Yuenyongsaen and S. Setha, Int. J. Trop. Subtrop. Hortic., 2022, 77(5), 1–8, DOI:10.17660/th2022/021.
- Grand View Research. Fruit puree market size, share & trends analysis report by product (tropical, citrus, berries), 2022 [cited 2025 Dec 19]. Available from: https://www.grandviewresearch.com/industry-analysis/fruit-puree-market.
- K. Pravallika and S. Chakraborty, Appl. Food Res., 2022, 2(2), 100229, DOI:10.1016/j.afres.2022.100229.
- Y.-Y. Lai, J.-H. Chen, Y.-C. Liu, Y.-T. Hsiao and C.-Y. Wang, J. Food Sci. Technol., 2022, 59(3), 990–1000, DOI:10.1007/s13197-021-05103-7.
- P. Himashree and R. Mahendran, Sustainable Food Technol., 2025, 3(3), 714–724, 10.1039/D5FB00042D.
- R. K. Tadapaneni, H. Daryaei, K. Krishnamurthy, I. Edirisinghe and B. M. Burton-Freeman, J. Agric. Food Chem., 2014, 62(18), 3877–3885, DOI:10.1021/jf404400q.
- L. Y. Zheng, G. M. Sun, Y. G. Liu, L. L. Lv, W. X. Yang, W. F. Zhao and C. B. Wei, Int. J. Mol. Sci., 2012, 13(6), 7383–7392, DOI:10.3390/ijms13067383.
- C. B. Steingass, R. Carle and H. G. Schmarr, Anal. Bioanal. Chem., 2015, 407(9), 2591–2608, DOI:10.1007/s00216-015-8474-z.
- L. Chen, T. Wang, Y. Sui, M. Gong, M. Li, X. Du and S. Zhang, Food Chem. X, 2025, 27, 102354, DOI:10.1016/j.fochx.2025.102354.
- F. J. Barba, C. Cortés, M. J. Esteve and A. Frígola, Food Bioprocess Technol., 2012, 5, 2222–2232, DOI:10.1007/s11947-011-0570-2.
- K. Marszałek, Ł. Wo´zniak, S. Sk ˛apska and M. Mitek, J. Food Sci. Technol., 2017, 54, 832–841, DOI:10.1007/s13197-017-2529-4.
- W. Wu, G. Xiao, Y. Yu, Y. Xu, J. Wu, J. Peng and L. Li, Food Control, 2021, 130, 108293, DOI:10.1016/j.foodcont.2021.108293.
- L. Kowitcharoen, C. Wongs-Aree, S. Setha, R. Komkhuntod, Kondo and V. Srilaong, J. Agric. Nat. Resour., 2018, 52(2), 146–154, DOI:10.1016/j.anres.2018.06.003.
- X. Cao, X. Bi, W. Huang, J. Wu, X. Hu and X. Liao, Innov. Food Sci. Emerg. Technol., 2012, 16, 181–190, DOI:10.1016/j.ifset.2012.05.008.
- M. K. Roy, M. Koide, T. P. Rao, T. Okubo, Y. Ogasawara and L. R. Juneja, Int. J. Nutr. Food Sci., 2010, 61(2), 109–124, DOI:10.3109/09637480903292601.
- L. G. Malta, R. H. Liu and N. K. Van Alfen, Encyclopedia of Agriculture and Food Systems, Academic Press, 2014, pp 305–314, DOI:10.1016/B978-0-444-52512-3.00058-9.
- R. S. Samakradhamrongthai, Food Appl. Biosci. J., 2020, 8(2), 53–67 Search PubMed . https://li01.tci-thaijo.org/index.php/fabjournal/article/view/240061.
- R. L. Prior, H. Hoang, L. Gu, X. Wu, M. Bacchiocca, L. Howard, M. Hampsch-Woodill, D. Huang, B. Ou and R. Jacob, J. Agric. Food Chem., 2003, 51(11), 3273–3279, DOI:10.1021/jf0262256.
- Y. Shibata, K. Ohara, K. Matsumoto, T. Hasegawa and M. Akimoto, J. Nutr. Sci. Vitaminol., 2021, 67(3), 201–209, DOI:10.3177/jnsv.67.201.
- S. T. Win and S. Setha, Horticulturae, 2022, 8(6), 555, DOI:10.3390/horticulturae8060555.
- H. Samejima and B.-J. Park, J. Agric. Rural Dev., 2019, 63(1), 12–17, DOI:10.11248/jsta.63.12.
- J. R. Zhou, Y. Kanda, A. Tanaka, H. Manabe, T. Nohara and K. Yokomizo, J. Agric. Food Chem., 2016, 64(2), 403–408, DOI:10.1021/acs.jafc.5b05320.
- R. V. Devakate, V. V. Patil, S. S. Waje and B. N. Thorat, Sep. Purif. Technol., 2009, 64(3), 259–264, DOI:10.1016/j.seppur.2008.09.012.
- A. Kongsuwan, P. Suthiluk, T. Theppakorn, V. Srilaong and S. Setha, J. Agric. Food Res., 2009 DOI:10.1016/J.FOODCHEM.2010.01.037.
- H. Niu, L. Yuan, H. Zhou, Y. Yun, J. Li, J. Tian, K. Zhong and L. Zhou, Foods, 2022, 11(5), 632, DOI:10.3390/foods11050632.
- V. Kumar, D. Kohli, B. Naik, A. Ratore, A. K. Gupta, J. M. Khan, M. Irfan, M. S. Preet, N. Chatterjee and S. Rustagi, J. King Saud Univ. Sci., 2023, 35(7), 102819, DOI:10.1016/j.jksus.2023.102819.
- B. Yuan, M.-G. C. Danao, M. Lu, S. A. Weier, J. E. Stratton and C. L. Weller, Innov. Food Sci. Emerg. Technol., 2018, 47, 241–248, DOI:10.1016/j.ifset.2018.03.006.
- X. Bi, Z. Zhou, T. Qin, X. Wang, Y. Ma, Y. Xing and Z. Che, RSC Adv., 2020, 10(52), 31333–31341, 10.1039/D0RA05181K.
- A. Gómez-Maqueo, J. Welti-Chanes and M. P. Cano, Food Res. Int., 2020, 130, 108909, DOI:10.1016/j.foodres.2019.108909.
- S. Chakraborty, P. S. Rao and H. N. Mishra, Food Bioprocess Technol., 2016, 9(5), 768–791, DOI:10.1007/s11947-015-1663-0.
- M. C. Razola-Díaz, S. Volpe, A. M. Gómez-Caravaca, E. Torrieri and V. Verardo, LWT–Food Sci. Technol., 2024, 206, 0023–6438, DOI:10.1016/j.lwt.2024.116612.
- A. Patras, N. P. Brunton, S. Da Pieve and F. Butler, Innov. Food Sci. Emerg. Technol., 2009, 10(3), 308–313, DOI:10.1016/j.ifset.2008.12.004.
- I. Odriozola-Serrano, R. Soliva-Fortuny and O. Martin-Belloso, J. Sci. Food Agric., 2008, 88, 2606–2614, DOI:10.1002/jsfa.3368.
- F. J. Barba, M. J. Esteve and A. Frigola, Food Res. Int., 2013, 50(2), 545–549, DOI:10.1016/j.foodres.2011.02.038.
- O. M. Atrooz, Int. J. Food Sci. Technol., 2008, 43(3), 490–494, DOI:10.1111/j.1365-2621.2006.01478.x.
- A. Cilla, M. J. Rodrigo, B. De Ancos, C. Sánchez-Moreno, M. P. Cano, L. Zacarías, R. Barberá and A. Alegría, Food Funct., 2020, 11(10), 8951–8962, 10.1039/D0FO02048F.
- C. Pérez-Lamela, I. Franco and E. Falqué, Molecules, 2021, 26(17), 5265, DOI:10.3390/molecules26175265.
- F. J. Barba, L. R. B. Mariutti, N. Bragagnolo, A. Z. Mercadante, G. V. Barbosa-Cánovas and V. Orlien, Trends Food Sci. Technol., 2017, 67, 195–206, DOI:10.1016/j.tifs.2017.07.006.
- S. Chakraborty, P. S. Rao and H. N. Mishra, Food Bioprocess Technol., 2015, 8(1), 10–23, DOI:10.1016/j.ifset.2015.01.004.
- K. Chobotova, A. B. Vernallis and F. A. Majid, Cancer Lett., 2010, 290(2), 148–156, DOI:10.1016/j.canlet.2009.08.001.
- R. Pavan, S. Jain and A. Shraddha, Int. J. Biotechnol. Res., 2012, 976203, DOI:10.1155/2012/976203.
- Y. A. Bernardo, D. K. A. do Rosario and C. A. Conte-Junior, Foods, 2023, 12(3), 476, DOI:10.3390/foods12030476M.
- D. Knorr, V. Heinz and R. Buckow, Biochim. Biophys. Acta, 2006, 1764(3), 619–631, DOI:10.1016/j.bbapap.2006.01.017.
- N. K. Rastogi, K. S. M. S. Raghavarao, V. M. Balasubramaniam, K. Niranjan and D. Knorr, Crit. Rev. Food Sci. Nutr., 2007, 47(1), 69–112, DOI:10.1080/10408390600626420.
- N.-N. Zhi, K. Zong, X.-Y. Jia, L. Wang and J. Liang, J. Food Process. Eng., 2019, 42(6), e13146, DOI:10.1111/jfpe.13146.
- S. L. Thu, H. L. M. Tam, W. Tongdeesoontorn and P. Suthiluk, Curr. J. Appl. Sci. Technol., 2017, 17(2), 162–171 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |