 Open Access Article
Open Access ArticleSustainable production of a bioinoculant and lactic acid from a native lactic acid strain Pediococcus acidilactici A40: the role of nitrogen sources
Luis Fernando Mejía-Avellaneda ab,
Elizabeth Céspedes-Gutiérrez
ab,
Elizabeth Céspedes-Gutiérrez b,
Diego Francisco Cortés-Rojas
b,
Diego Francisco Cortés-Rojas b and
Leyanis Mesa
b and
Leyanis Mesa *b
*b
aInstituto de Ciencia y Tecnología de Alimentos (ICTA), Facultad de Ciencias Agrarias, Universidad Nacional de Colombia, Sede Bogotá. Bogotá, D.C., Colombia
bCorporación Colombiana de Investigación Agropecuaria – AGROSAVIA, Departamento de Bioproductos, Km 14 Bogotá-Mosquera, Mosquera, Cundinamarca, Colombia. E-mail: leyanis.mesa@gmail.com
First published on 20th January 2026
Abstract
The development of cost-effective and sustainable microbial inoculants is essential to promote environmentally friendly agricultural practices and circular bioeconomy strategies. In this study, a native strain of Pediococcus acidilactici A40 was cultivated using low-cost organic nitrogen sources to produce viable biomass intended for application as a silage bioinoculant, while simultaneously generating lactic acid (LA) as a value-added co-product that can be further processed as an ingredient for food technology purposes. An experimental mixture design (MD) was applied to evaluate yeast extract (YE), corn steep liquor (CSL), and hydrolysed soybean protein (HSP) as nitrogen sources. The influence of these sources on cell viability and LA concentration was assessed, and a cost–benefit analysis (CBA) was conducted to determine the most economically viable fermentation medium. The results showed that the partial substitution of YE (40%) with CSL (60%) maintained high cell viability (>11 log CFU g−1 dry biomass (DB)) and acceptable LA concentrations. This combination achieved a 22.85% reduction in medium cost and outperformed the YE-only medium in terms of overall economic efficiency. Desirability analysis indicated that the CSL–YE combination provided an optimal balance between performance and cost. This study contributes to the valorisation of native microbial resources and agro-industrial byproducts, aligning with the principles of bioeconomy. By addressing the need to explore alternative nitrogen sources in biotechnological processes—a key strategy to enhance process sustainability—the proposed dual-purpose bioprocess supports the development of affordable, locally produced bioinputs and bioproducts for agricultural and industrial sectors, offering a practical alternative to reduce reliance on imported inputs and to strengthen circularity in production chains.
Sustainability spotlightA native strain of Pediococcus acidilactici A40 was used to develop a dual-purpose fermentation process that enables the simultaneous production of a bioinoculant and lactic acid. The partial replacement of yeast extract with corn steep liquor—an agro-industrial byproduct—reduces production costs and environmental impact while valorizing regional microbial biodiversity. This strategy fosters local bioinput and bioproduct manufacturing and contributes to circular bioeconomy models. The innovation aligns with key United Nations Sustainable Development Goals (SDGs), particularly SDG 2 (Zero Hunger), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action), offering a scalable and sustainable alternative for bioprocessing in agri-food and bio-based industries. |
Introduction
The transition to sustainable food systems has become a global strategic priority, driving the use and commercial production of bioinputs and bioproducts. In this context, the development of more efficient, diversified and sustainable bioprocesses plays a key role, as it aligns with the bioeconomy policies adopted by many countries. The feedstuff and foodstuff sectors are seeking ingredients and inputs obtained through sustainable processes that utilize native biodiversity resources within circular systems.1,2Lactic acid bacteria (LAB), microorganisms considered GRAS, are widely used as starters, probiotics and bioinoculants, and their bioprocesses yield numerous metabolites with applications in the food, pharmaceutical, and agricultural industries. LAB has been established as a natural technology for preserving food and fodder through fermentation, including the silage process, which is essential for conserving plant biomass for animal feed. The addition of LAB-based inoculants in silos significantly improves the aerobic stability, nutritional value and microbiological safety of forage, reducing the proliferation of pathogenic microorganisms and the need for chemical additives.3,4
To enhance the sustainability and economic viability of LAB-based bioprocesses, it is essential to recover and capitalise on all potential coproducts derived from fermentation in complex growth media. Among these metabolites, lactic acid (LA) is one of the most promising platform compounds in the current bioeconomy because of its efficacy as a natural preservative, acidulant, and antimicrobial agent.5 It is used in the food and beverage sector as a preservative and pH-adjusting agent, and in the pharmaceutical, cosmetic, and chemical industries as a solvent and starting material for the production of lactate esters. However, a limiting factor for LA fermentations is the high cost of the media components, which influences the total cost of the process.6,7 The integrated production of a viable LAB-based bioinoculant and LA within a single fermentation process represents an innovative strategy that improves technical feasibility and enhances bioprocess sustainability.8–10 This diversified approach to bioprocessing maximizes the value of LAB by promoting multi-product production schemes that enhance resource efficiency and support sustainable process integration.
In the development of bioinoculants, it is essential that the selected microorganisms be comprehensively characterized with respect to their genetic, morphological, and functional traits and that their safety for human, animal, and plant health be established. Key characteristics include a rapid and efficient conversion of water-soluble carbohydrates into lactic acid, enabling a fast pH decline and early stabilization of the ensiling environment, thereby limiting the growth of undesirable microorganisms. In addition, effective silage inoculants require high tolerance to environmental stresses such as low pH, anaerobic conditions, and fluctuating temperatures, as well as strong competitiveness against the epiphytic microbiota naturally present on forages. In this context, Pediococcus acidilactici is a Gram-positive LAB belonging to the Lactobacillaceae family, widely recognized for its probiotic and antimicrobial properties, as well as its industrial applications.11–16 It can grow on various substrates and produce LA under suitable conditions.17,18 Also, Pediococcus acidilactici integrates stress tolerance mechanisms commonly observed in LAB with unique genomic and metabolic features, conferring good adaptability and resilience under adverse environmental conditions.19 The strain P. acidilactici A40, originally isolated from oat forage,20 has been identified as a promising silage bioinoculant owing to its capacity for rapid LA production and its antimicrobial properties, which enhance fermentation quality while inhibiting spoilage microorganisms. These attributes are in agreement with previous findings reported for this strain.3,4 Moreover, P. acidilactici A40 exhibits potential for broader applications as a bioinoculant, including its use as a plant biocontrol agent and as a plant growth promoter.21,22 To ensure efficacy in agricultural settings, these microorganisms require specialized formulation processes, typically as solid products, that preserve viability during storage, facilitate handling, and allow uniform application upon dilution. Therefore, the potential of native P. acidilactici strains utilized on regionally available forages represents a sustainable alternative tailored to local conditions and resources, reducing dependence on imported bioinputs and aligning with the principles of the bioeconomy.
A highly effective strategy for reducing fermentation media costs involves the valorisation of agro-industrial residues as alternative substrates, thereby promoting a circular economy framework. Residues such as sugarcane bagasse, fruit hulls, dairy by-products, molasses, by-products from fish, chicken, brewer's spent yeast, and wheat bran have been proven to be viable sources of carbon and nitrogen for LAB growth.17,23–26 This approach not only reduces these costs, but also mitigates the environmental impact associated with waste disposal, thereby strengthening the overall sustainability of the process. Carbon sources from agro-industrial residues have been widely documented as substrates for LAB fermentation.6,17,18,23,27–29 Sugars derived from lignocellulosic and agri-food residues are among the most extensively studied.30 However, nitrogen sources—although present in smaller proportions in the fermentation medium—have not been studied as intensively as carbon sources, even though they significantly influence production costs, particularly when the carbon source is already derived from residual materials.31–33
Despite the extensive research addressing the use of agro-industrial residues as alternative carbon sources in LAB fermentations, there is a lack of systematic studies evaluating low-cost alternative nitrogen sources in bioprocesses aimed at the simultaneous production of highly viable LAB biomass and LA. The objective of this study was to evaluate the technical feasibility and economic sustainability of totally or partially replacing standard yeast extract (YE) with alternative nitrogen sources—such as urea, corn steep liquor (CSL) and hydrolysed soy protein (HSP)—in the fermentation of the native strain Pediococcus acidilactici A40, with the aim of optimizing the simultaneous production of a bioinoculant for silage preservation and LA as a co-product.
Materials and methods
Fermentation study and raw materials preliminary evaluation
Pediococcus acidilactici A40 has been previously characterized at both genomic and phenotypic levels. The complete genome sequence of strain A40 has been reported, revealing no genes associated with virulence or known pathogenic determinants, and confirming its taxonomic affiliation within a LAB species with a long history of safe use20,21 In addition, phenotypic evaluations described in previous studies have demonstrated that strain A40 exhibits beneficial biological activities without evidence of pathogenic behaviour, supporting its suitability for applied fermentation and bioprocessing systems.
Reagents and raw materials
Molasses used in this study was obtained from Riopaila-Castilla (Zarzal, Valle del Cauca, Colombia) and contained 52.1% total sugars. The nitrogen sources employed included urea (Cimpa S.A.S., Colombia), yeast extract (YE) (Oxoid™, United Kingdom), corn steep liquor (CSL) (Sigma-Aldrich™, United States), and hydrolysed soy protein (HSP) (Tecnas S.A., Colombia).Inoculum preparation and medium composition
The Pediococcus acidilactici A40 strain was reactivated in 50 mL of MRS broth and incubated for 8 h at 37 °C ± 2 °C and 200 rpm in a shaking incubator (LabTech LSI-1005R, South Korea). A subsequent transfer was made to the same volume of MRS broth and incubated for 16 h under the same conditions. Fermentation was carried out in 250 mL Erlenmeyer flasks at a 5% (v/v) inoculum size, a 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 C
2 C![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N source ratio, and a 3
N source ratio, and a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 medium-to-flask volumetric ratio, at 37 °C and 200 rpm for 24 h in a shaking incubator. The fermentation media were formulated with 100 g L−1 of total sugars from molasses (192 g L−1 of sugarcane molasses), and the nitrogen sources were added according to the proportions and concentrations specified in each of the experimental designs. pH was initially adjusted at 7.0 ± 0.02 with 0.02 M NaH2PO4–Na2HPO4·7H2O (Merck™, United States) buffer, with 1 N NaOH addition. The seal of the Erlenmeyer flasks was made with cotton gauze swabs fixed with a stretchable plastic film to favour microaerophilic conditions. Alternative nitrogen sources were tested to partially or fully replace the amount of YE in the medium. First, urea—a low-cost nitrogen source—was evaluated. Subsequently, two organic sources—CSL and HSP—were assessed as supplementary nitrogen sources to YE.
5 medium-to-flask volumetric ratio, at 37 °C and 200 rpm for 24 h in a shaking incubator. The fermentation media were formulated with 100 g L−1 of total sugars from molasses (192 g L−1 of sugarcane molasses), and the nitrogen sources were added according to the proportions and concentrations specified in each of the experimental designs. pH was initially adjusted at 7.0 ± 0.02 with 0.02 M NaH2PO4–Na2HPO4·7H2O (Merck™, United States) buffer, with 1 N NaOH addition. The seal of the Erlenmeyer flasks was made with cotton gauze swabs fixed with a stretchable plastic film to favour microaerophilic conditions. Alternative nitrogen sources were tested to partially or fully replace the amount of YE in the medium. First, urea—a low-cost nitrogen source—was evaluated. Subsequently, two organic sources—CSL and HSP—were assessed as supplementary nitrogen sources to YE.
Effect of adding urea to the medium
The impact of urea and YE in the culture medium was evaluated. For both nitrogen sources, concentrations of 20 g L−1 and 40 g L−1 were used as the minimum and maximum levels, respectively. A full factorial 22 experimental design was applied to assess the individual and combined effects of these variables on bacterial biomass cell concentration and LA concentration. The design included 8 experiments (one replicate per condition). The randomized design matrix for these experiments is presented in Table 1.| Raw material costs | USD kg−1 |
|---|---|
| YE | 15 |
| CSL | 9 |
| HSP | 6 |
| Molasses | 0.3 |
Effect of adding organic nitrogen sources to the medium
Based on the results obtained from the preliminary factorial screening, urea was excluded from further optimization, and subsequent experiments focused on organic nitrogen sources. The effects of incorporating three organic nitrogen sources into the culture medium were evaluated: YE, CSL, and HSP. An experimental mixture design (MD), specifically a simplex centroid design based on Scheffé models, was employed to evaluate the combined effects of the three organic nitrogen sources. The total concentration of nitrogen sources was kept constant at 40 g L−1, and the experimental factors were expressed as relative proportions of each component within the mixture. The design included 10 experiments, with 3 centre points, as shown in Fig. 1. The randomized experimental matrix is shown in Table 2.| Trials | Variables | Response parameters | ||
|---|---|---|---|---|
| X1(YE concentration, g L−1) | X2 (urea concentration, g L−1) | Cell concentration (log cells per g DB) | LA concentration (g L−1) | |
| 1 | 1 (40) | 1 (40) | 9.13 ± 0.41 | 1.91 ± 0.057 |
| 2 | 1 (40) | −1 (20) | 12.08 ± 0.092 | 16.09 ± 1.74 |
| 3 | −1 (20) | 1 (40) | 10.53 ± 0.622 | 1.85 ± 0.021 |
| 4 | −1 (20) | −1 (20) | 10.83 ± 0.191 | 10.94 ± 0.149 |
The concentration of sugarcane molasses was kept constant across all experiments, and culture conditions followed those previously described. The response variables were cell viability on a dry weight basis (log CFU g−1 DB) and LA concentration (g L−1). A special cubic Scheffé model was fitted for the analysis (see eqn (1)).
 | (1) |
Prior to model fitting, an exploratory data analysis was conducted for each response to evaluate distributional properties and variance behavior. Normality (Shapiro–Wilk) and homoscedasticity (Breusch–Pagan) diagnostics indicated deviations from classical linear model assumptions for some responses. Therefore, a robust inference framework was adopted to ensure valid statistical conclusions without relying on strict normality or constant-variance assumptions. Model parameters and adjusted R2 were estimated by ordinary least squares (OLS), and inference relied on robust “sandwich” standard errors chosen automatically. The variance–covariance matrix for inference was selected in a data-driven manner: HC3 when heteroskedasticity was indicated (Breusch–Pagan), Newey–West when autocorrelation was detected (Breusch–Godfrey/Durbin–Watson), and otherwise HC2. Statistical term significance was assessed via term-wise Wald F tests; p-values were multiplicity-adjusted using the Holm method within each response (to control FWER), and the classical overall model F was reported for context. Model adequacy was examined with studentized residuals, standard diagnostics and standard graphical checks. To visualize uncertainty, one-dimensional profile plots with Bonferroni simultaneous bands were produced. The validity of the model assumptions was verified at a 5% significance level. For the desirability analysis, the function proposed by Derringer and Suich (eqn (2)) was applied, aiming to maximize both response variables,34
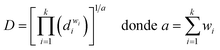 | (2) |
| C = 0.0225 + 0.6 × YE + 0.36 × CSL + 0.24 × HSP | (3) |
The weights assigned to the response variables for calculating global desirability were defined as w1 = 3, w2 = 3, and w3 = 1, corresponding to biomass viability, LA concentration and medium cost, respectively, in order to prioritize technological variables. The individual desirability values for each response variable were maximized according to the following function (eqn (4)):
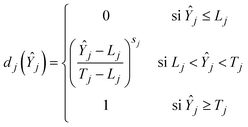 | (4) |
Analysis methods
Determination of cell concentration on a dry basis
At the end of the 24 h fermentation, triplicate samples of the fermentation broth were analysed for cell concentration using a modified Neubauer counting chamber (Brand™ GmbH + Co, Germany).38 A volume of 10 µL from each selected dilution was loaded into each chamber section, and two sets of ten randomly selected type #3 squares were counted. Decimal dilutions were prepared in 0.9% saline solution, and counts were performed with a microscope (Olympus™ BX53, Germany) under a 40× objective lens. Eqn (5) was used to calculate cell concentration:
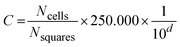 | (5) |
Determination of viability on a dry basis
After 24 h of fermentation, viability was determined from triplicate samples of the fermentation broth using the spread plate method with decimal dilutions in 0.9% saline solution on MRS agar.39 Plates were incubated anaerobically (AnaeroGen™, Oxoid™, United Kingdom) at 37 °C for 48 h (Isotherm® Forced Convection Lab Incubator IFA-54-9, Esco™ Lifesciences Group, Singapore) and colonies were counted as CFU mL−1. As well as with the cell concentration, viability was converted into CFU g−1 biomass on a dry basis using the corresponding values of DB.Determination of LA concentration
A separate set of duplicate samples of the fermentation broth were centrifuged at 8000×g for 10 min (Sorvall™ Biofuge Primo™ R, Thermo Scientific™, United States). The resulting cell-free supernatant was collected and filter-sterilized using 0.22 µm pore size polyethersulfone (PES) filters (Millex™ GP, Millipore™, United States) for LA concentration analysis. Quantification was performed by HPLC with refractive index detection (Waters IR 2414, Waters Acquity™, United States) using a Biorad HPX-87H HPLC column (BioRad™, United States). The mobile phase consisted of 5 mM H2SO4, with an oven temperature of 65 °C, a detector temperature of 40 °C, and a flow rate of 0.6 mL min−1. Analyses were conducted on a UPLC Waters Acquity™ system (Waters™, United States).Formulation of the viable cell biomass for inoculant prototype
For the final bioinoculant formulation, the fermentation broth was centrifuged at 4500 rpm at 25 °C (Rotina™ 420 Hettich 4701-01, Andreas Hettich™ GmbH, Germany) to separate the biomass from the cell-free supernatant. The recovered biomass was mixed with an excipient at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 biomass-to-excipient ratio, granulated, and dried in a fluidized bed dryer (Glatt™ Uni-glatt 8512, Germany) at 35 °C to achieve a final moisture content below 8%. The biomasses obtained from the fermentation broth using CSL and YE as nitrogen sources were used to formulate solid prototypes, which were stored at 25 °C for 90 days. Microbial viability was assessed over time to evaluate the effect of the nitrogen source on prototype stability.
2 biomass-to-excipient ratio, granulated, and dried in a fluidized bed dryer (Glatt™ Uni-glatt 8512, Germany) at 35 °C to achieve a final moisture content below 8%. The biomasses obtained from the fermentation broth using CSL and YE as nitrogen sources were used to formulate solid prototypes, which were stored at 25 °C for 90 days. Microbial viability was assessed over time to evaluate the effect of the nitrogen source on prototype stability.
Cost–benefit analysis (CBA)
A preliminary CBA was conducted based on raw material costs and estimated revenues from the bioinoculant and LA. Although this evaluation does not account for capital or operational expenditures, it offers an initial indication of the economic feasibility of the process. The costs of raw materials considered for CBA are shown in Table 1. The estimated market prices for the bioinoculant and LA were 90 USD kg−1 and 2 USD kg−1, respectively. Eqn (6) was used to calculate the cost–benefit index (CB). Product prices were based on market data, adopting the most conservative available values.
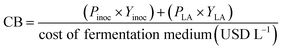 | (6) |
Results and discussion
The production of bioinputs and bioproducts must fulfill the requirement of economic competitiveness. Among the main factors influencing total production costs, the fermentation medium represents a significant component. In this study, although molasses had already been selected as a cost-effective carbon source,40 the high proportion of YE used still contributed substantially to the overall medium cost. Therefore, a strategy was developed to totally or partially replace YE. The analysis began with the evaluation of urea, a low-cost non-protein nitrogen source commonly used in industrial fermentation processes. Nevertheless, its use resulted in decreased cell concentration and LA production. Consequently, the study proceeded with the evaluation of organic nitrogen sources in combination with YE—specifically CSL and HSP—which led to improved performance compared to urea.Evaluation of urea as a N source
Urea has been utilised as a nitrogen source in bioprocesses, offering a cost-effective and widely available alternative to conventional inorganic compounds. Its combination with YE has been shown to enhance LA production by Lactobacillus delbrueckii NCIM 2025, demonstrating the potential of mixed nitrogen strategies to optimize nutrient utilization and process performance.5,41 The results of biomass cell concentration and LA concentration are presented in Table 2.Experiment 2 yielded the highest LA concentration (16.09 g L−1) and the highest cell viability. In contrast, experiments 1 and 3, both of which involved high levels of urea, showed a marked decrease in LA concentration. Cell concentration of the biomass in experiment 1 remained relatively low, where the urea level was the highest, reaching only log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 cells per g DB. This result suggests an inhibitory effect of urea on the growth of the strain. The resulting models for cell concentration (Y1) and LA concentration (Y2) obtained from the statistical analyses are shown in eqn (7) and (8):
9 cells per g DB. This result suggests an inhibitory effect of urea on the growth of the strain. The resulting models for cell concentration (Y1) and LA concentration (Y2) obtained from the statistical analyses are shown in eqn (7) and (8):
| Y1 = 10.64 − 0.03X1 − 0.81X2 − 0.67X1X2 | (7) |
| Y2 = 7.70 − 1.31X1 − 5.82X2 − 1.27X1X2 | (8) |
According to Fig. 2 and eqn (7) and (8), urea had a statistically significant effect on both response variables, with a negative coefficient. This indicates that, within the tested concentration range, urea significantly reduced LA production to very low levels (1.85 g L−1) and decreased cell concentration from 12 to 9.13 log cells per g DB. The inhibitory effect of urea on LA production by Lactobacillus casei has been previously reported.42 Despite these results, certain non-protein nitrogen sources such as ammonium sulphate and urea can partially replace costly YE without significantly reducing LA production when used in combination with organic sources or supplemented with B vitamins.43 Regression and ANOVA analyses are presented in SI 1 (SI 1). The use of YE led to a significant increase in both response variables. Other protein nitrogen sources, such as meat extract and peptone, are also relevant, as they not only supply nitrogen, but also provide essential vitamins that support cell growth and metabolic activity. In particular, the relevance of YE during the initial stages of fermentation is attributed to its high content of amino acids, peptides, and B-complex vitamins, which stimulate microbial proliferation and enhance LA production.44,45
 | ||
| Fig. 2 Pareto graph of cell concentration (A) and LA concentration (B) in the 22 full factorial experiment. | ||
The obtained results indicate that the use of urea as a partial replacement for YE as a nitrogen source in the fermentation medium must be carefully evaluated. High urea concentrations negatively affected LA production and inhibited cell proliferation to some extent. The most favourable outcome was achieved with high YE concentration (40 g L−1) and low urea concentration (20 g L−1), resulting in both high cell concentration and LA production. Accordingly, urea levels should be carefully optimized. A balanced combination of protein and non-protein nitrogen sources may enhance process efficiency while reducing production costs.
In the present study, bacterial biomass is one of the target products for recovery, intended for use as a bioinoculant for silage preservation. Therefore, maintaining high viability is essential to ensure its biological activity during subsequent formulation steps. When urea is included in the fermentation medium, a considerable increase in total biomass is observed (data not shown), which comprises both LAB biomass and residual medium components. This increase, however, negatively impacts biomass viability. Consequently, downstream formulation processes and the overall effectiveness of the bioinoculant may be compromised. Typically, Pediococcus acidilactici is not expected to possess urease activity, in contrast to some foodstuff and feedstuff pathogens that do; this enzymatic activity can be detrimental for the ensiling process.46,47 Therefore, the effectiveness of urea depends on its concentration and the overall nutritional balance of the medium. While it can contribute non-protein nitrogen, it cannot fully replace the nutrients provided by organic sources.48 For this reason, we decided not to continue with the optimization of partial YE substitution by urea.
Following the evaluation of urea as a partial substitute for YE in the diversified fermentation of Pediococcus acidilactici A40, other organic nitrogen sources—such as CSL and HSP—were subsequently assessed. These alternatives were selected due to their lower cost and their proven effectiveness in supporting microbial growth across various fermentation processes.49,50
Results of the use of organic nitrogen sources
An MD was used to evaluate the effects of different organic nitrogen sources. This type of design is commonly applied to assess the composition of culture media components, such as sugar sources, residues, and other nutrient mixtures.51–53 Table 3 presents the results for each experimental combination. The replicate at the central point exhibited low variability for both response variables (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10.85 ± 0.04 CFU g−1 DB; 17.34 ± 0.23 g L−1 LA). The highest LA concentrations were observed in treatments containing YE, supporting the findings previously discussed. In contrast, the combinations that included HSP showed the lowest performance in terms of LA production and viable biomass. A possible explanation for this preliminary observation is that HSP is not a significant source of B-complex vitamins, unlike YE and CSL.54 Additionally, variability in the production processes of HSP may affect the presence of components that promote cell growth and LA formation.55
10.85 ± 0.04 CFU g−1 DB; 17.34 ± 0.23 g L−1 LA). The highest LA concentrations were observed in treatments containing YE, supporting the findings previously discussed. In contrast, the combinations that included HSP showed the lowest performance in terms of LA production and viable biomass. A possible explanation for this preliminary observation is that HSP is not a significant source of B-complex vitamins, unlike YE and CSL.54 Additionally, variability in the production processes of HSP may affect the presence of components that promote cell growth and LA formation.55| Cell viability, log CFU g−1 DB = 11.830X1 + 11.587X2 + 10.716X3 + 1.028X2X3 − 11.616X1X2X3 | (9) |
| Treatment | Variables | Response parameters | |||
|---|---|---|---|---|---|
| X1 (YE) | X2 (CSL) | X3 (HSP) | Cell viability (log CFU g−1 DB) | LA concentrationa (g L−1) | |
| a Results of the chromatograms of LA are presented in SI 2. | |||||
| 1 | 1 (40) | 0 | 0 | 11.83 | 18.32 |
| 2 | 0 | 1 (40) | 0 | 11.59 | 11.82 |
| 3 | 0 | 0 | 1 (40) | 10.72 | 6.85 |
| 4 | 0.5 (20) | 0.5 (20) | 0 | 11.62 | 18.15 |
| 5 | 0.5 (20) | 0 | 0.5 (20) | 11.53 | 13.69 |
| 6 | 0 | 0.5 (20) | 0.5 (20) | 11.14 | 8.76 |
| 7 | 0.33 (13.2) | 0.33 (13.2) | 0.33 (13.2) | 10.88 | 17.60 |
| 8 | 0.33 (13.2) | 0.33 (13.2) | 0.33 (13.2) | 10.88 | 17.14 |
| 9 | 0.33 (13.2) | 0.33 (13.2) | 0.33 (13.2) | 10.80 | 17.29 |
| 10 | 0.25 (10) | 0.38 (15.2) | 0.38 (15.2) | 10.74 | 15.43 |
The fitted Scheffé mixture model (eqn (9)) describing biomass viability exhibited excellent statistical performance and predictive reliability. The coefficient of determination (R2 = 0.99) and adjusted R2 (0.99) indicate that nearly all the experimental variability in biomass viability was explained by the model, with no evidence of overfitting. Model precision was further supported by a low root mean square error (RMSE = 0.053 log CFU g−1 DB) and a small residual standard deviation (σ = 0.0978), confirming minimal unexplained variability within the experimental domain. Information criteria values (AIC = −14.19; BIC = −11.77) further indicate that the selected model achieves an optimal balance between goodness of fit and parsimony.
The regression model for dry-based cell viability (eqn (9)) indicates that all components had individually statistically significant linear effects, with YE exerting the highest influence (see Table 4). Nonetheless, its effect was only marginally greater, as the linear coefficients for all components were very similar. Only the binary interaction between CSP and HSP was significant, and the ternary interaction effect is negative and of high magnitude in the model. This indicates that the synergy of the three components may cause a significant decrease in viability.
| Cell viability, log UFC g−1 DB | |||||
|---|---|---|---|---|---|
| Variables | Estimate | Std.err | t-value | F-wald | P holm |
| YE | 11.83 | 0.039 | 299.48 | 8.21 × 10−8 | 5.75 × 10−7* |
| CSL | 11.59 | 0.039 | 293.328 | 8.74 × 10−8 | 5.75 × 10−7* |
| HSP | 10.71 | 0.039 | 271.278 | 1.1 × 10−7 | 5.75 × 10−7* |
| YE:CSL | −0.36 | 0.193 | −1.891 | 0.155 | 0.31 |
| YE:HSP | 1.03 | 0.193 | 5.312 | 0.013 | 0.0391* |
| CSL:HSP | −0.011 | 0.193 | −0.061 | 0.955 | 0.955 |
| YE:CSL:HSP | −11.62 | 1.624 | −7.142 | 0.0056 | 0.0226* |
| R2 | 0.99 | ||||
| R2 adjust | 0.99 | ||||
Fig. 3A shows that cell viability increases with increasing proportions of YE, reaching a maximum of approximately log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11.8 CFU g−1 DB near the upper vertex. The lowest values are observed in the centre and toward the lower right vertex of the diagram, corresponding to regions where HSP is predominant. The difference between the minimum and maximum values is approximately log
11.8 CFU g−1 DB near the upper vertex. The lowest values are observed in the centre and toward the lower right vertex of the diagram, corresponding to regions where HSP is predominant. The difference between the minimum and maximum values is approximately log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 CFU g−1 DB. The effect plot along the Cox direction for the fitted cell viability model in Fig. 4 shows that YE is the most statistically significant positive component of the mixture in terms of cell viability, while HSP exerts a statistically significant negative effect, continuously decreasing cell viability as its proportion in the mixture increases, until reaching a value near log
1 CFU g−1 DB. The effect plot along the Cox direction for the fitted cell viability model in Fig. 4 shows that YE is the most statistically significant positive component of the mixture in terms of cell viability, while HSP exerts a statistically significant negative effect, continuously decreasing cell viability as its proportion in the mixture increases, until reaching a value near log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10.75 CFU g−1 DB, which is considered acceptable for formulation. This suggests that HSP is not highly unfavourable for viable biomass formation, as cell viability remains above log
10.75 CFU g−1 DB, which is considered acceptable for formulation. This suggests that HSP is not highly unfavourable for viable biomass formation, as cell viability remains above log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 CFU g−1 DB, although YE yields superior results. The use of HSP with an 85.14% degree of hydrolysis as a nitrogen source resulted in satisfactory viability (log
10 CFU g−1 DB, although YE yields superior results. The use of HSP with an 85.14% degree of hydrolysis as a nitrogen source resulted in satisfactory viability (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.45 CFU mL−1), comparable to that achieved with the standard microbiological medium (MRS) (log
9.45 CFU mL−1), comparable to that achieved with the standard microbiological medium (MRS) (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.73 CFU mL−1) for Lactobacillus plantarum Dad 13 in a complex medium also containing coconut milk, soybean sprout extract, and tomato extract.56 CSL also supports the growth of LAB, as mixtures near the lower left vertex display viability values exceeding log
9.73 CFU mL−1) for Lactobacillus plantarum Dad 13 in a complex medium also containing coconut milk, soybean sprout extract, and tomato extract.56 CSL also supports the growth of LAB, as mixtures near the lower left vertex display viability values exceeding log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 CFU g−1 DB. Another study involving batch fermentation of Lactococcus lactis subsp. lactis var. diacetylactis (SLT 6) using 12 g L−1 CSL supplemented with 2 g L−1 YE reported a final cell concentration of log
11 CFU g−1 DB. Another study involving batch fermentation of Lactococcus lactis subsp. lactis var. diacetylactis (SLT 6) using 12 g L−1 CSL supplemented with 2 g L−1 YE reported a final cell concentration of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.97 CFU mL−1.57 Similarly, CSL was found to significantly enhance biomass production in batch fermentation of Lactobacillus casei KH-1.58
9.97 CFU mL−1.57 Similarly, CSL was found to significantly enhance biomass production in batch fermentation of Lactobacillus casei KH-1.58
 | ||
| Fig. 3 Effect of nitrogen sources on (A.) cell viability (log CFU g−1 DB) and (B.) LA concentration (g L−1). | ||
Biomass with high viability levels (>log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 CFU g−1 DB) ensures that, during the formulation operation, cell viability remains above log
10 CFU g−1 DB) ensures that, during the formulation operation, cell viability remains above log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 CFU g−1 formulated product. This guarantees the delivery of a sufficient number of viable cells upon application to dominate the fermentation process in the silage. Such high concentrations facilitate achieving the recommended effective inoculation dose (≥105–106 CFU of LAB per gram of forage). As a result, a rapid and efficient LA fermentation is established, which is essential for abundant LA production, a swift pH drop, and the inhibition of undesirable microorganisms. These effects enhance aerobic stability and help preserve the nutritional value of the silage.59,60
10 CFU g−1 formulated product. This guarantees the delivery of a sufficient number of viable cells upon application to dominate the fermentation process in the silage. Such high concentrations facilitate achieving the recommended effective inoculation dose (≥105–106 CFU of LAB per gram of forage). As a result, a rapid and efficient LA fermentation is established, which is essential for abundant LA production, a swift pH drop, and the inhibition of undesirable microorganisms. These effects enhance aerobic stability and help preserve the nutritional value of the silage.59,60
Statistical analysis of the MD for LA concentration showed that all linear terms in the model (eqn (10)) were statistically significant (p < 0.05) (see Table 5), along with the ternary interaction, all of them with positive terms. These results indicated that YE was the most influential independent variable, followed by CSL and HSP (YE > CSL > HSP). Fig. 3B shows a region where the maximum LA concentration is found adjacent to high proportions of YE and lower proportions of CSL, while the lowest concentrations are located near the HSP vertex. The effect plot in the Cox direction shown in Fig. 5 indicates that higher proportions of YE promote LA concentration, whereas higher proportions of HSP have the opposite effect, and CSL appears to have a detrimental effect when its proportion increases. This finding aligns with the well-established role of YE as a rich nutrient source, containing free amino acids, peptides, B-complex vitamins, and other essential growth factors that support LAB metabolism and LA synthesis.61 However, the relatively strong performance of the alternatives containing CSL suggests its potential as a partial substitute for YE. This is particularly relevant in the context of cost reduction, as it may allow for a significant decrease in medium costs without compromising cell viability or LA production. The response surface illustrated in Fig. 3B supports this interpretation, showing high LA concentrations near the upper vertex (YE) and notably competitive values along the CSL axis, while lower concentrations are associated with regions where HSP is present in higher proportions.
| LA, g L−1 = 18.332X1 + 11.817X2 + 6.845X3 + 8.667X1X2X3 | (10) |
| Lactic acid, g L−1 | |||||
|---|---|---|---|---|---|
| Variables | Estimate | Std.err | t-value | F-wald | P holm |
| YE | 18.33 | 0.571 | 32.075 | 1028.82 | 0.0005* |
| CSL | 11.82 | 0.572 | 20.673 | 427.368 | 0.0015* |
| HSP | 6.84 | 0.572 | 11.976 | 143.413 | 0.0063* |
| YE:CSL | 12.39 | 2.799 | 4.429 | 19.614 | 0.0642 |
| YE:HSP | 4.52 | 2.799 | 1.614 | 2.606 | 0.41 |
| CSL:HSP | −2.56 | 2.786 | −0.921 | 0.848 | 0.425 |
| YE:CSL:HSP | 88.66 | 13.31 | 6.661 | 44.372 | 0.0276* |
| R2 | 99.99% | ||||
| R2 adjust | 99.99% | ||||
The fitted Scheffé mixture model describing LA concentration (eqn (10)) demonstrated strong statistical performance and high explanatory power. Both the coefficient of determination (R2 = 0.99) and the adjusted R2 (0.99) indicate that the model explains nearly all the experimental variability in LA production, confirming the relevance of the selected mixture terms and the absence of overfitting.
The model exhibited a RMSE of 0.208 g L−1 and a residual standard deviation (σ) of 0.3812 g L−1, which are low relative to the observed range of LA concentrations. These values indicate good predictive accuracy, although they are higher than those obtained for biomass viability, reflecting the intrinsically higher biological and analytical variability associated with metabolite production compared to cell viability.
Information criteria analysis yielded AIC and BIC values of 13.05 and 15.47, respectively. While these absolute values are not directly interpretable in isolation, they indicate that the selected model provides an adequate balance between goodness of fit and model complexity when compared with alternative formulations. The higher corrected Akaike Information Criterion (AICc = 157.05) reflects the limited sample size typical of mixture designs and the greater sensitivity of metabolite-related responses to experimental variability.
The validation graphics for Models 9 and 10 are presented in SI 3 (SI 3). In the model (eqn (10)), the effect of YE was nearly three times greater than that of HSP, confirming its superior suitability for achieving higher LA concentrations. However, the results also indicate that replacing YE with alternative nitrogen sources tends to reduce LA production to some extent. Previous studies provide a nuanced view of these alternatives. For instance, CSL has been reported to have no significant impact on LA production by Lactobacillus casei KH-1, regardless of its association with cellular growth.58 In contrast, other findings suggest that LA production by Lactobacillus SMI8 increases proportionally with the concentration of CSL used,54 highlighting the strain-specific nature of these responses. Similarly, while HSP has been considered a satisfactory nitrogen source for LAB growth, its efficacy in promoting LA synthesis depends strongly on its degree of hydrolysis. For example, in the fermentation process using Lactobacillus amylovorus NRRL B-4542, peptides with an average molecular weight of ∼700 Da at 3% concentration were found to be optimal for LA production.62 This suggests that not only the nitrogen source but also the degree of protein hydrolysis is directly related to its effectiveness as a nitrogen source in LA production.
In a previously published study, CSL emerged as the most effective alternative to YE, offering both strong technical performance and economic viability. This low-cost byproduct of wet corn milling supplies key nutrients, including amino acids, vitamins, and minerals, which can partially sustain LA production, with productivities comparable to or even exceeding those achieved with YE.63 In contrast, although HSP is less expensive than both YE and CSL and can be nutritionally adequate when properly processed, it showed inferior performance in this study. This may be attributed to the compositional variability of HSP and the possible lack of essential nutrients required by the Pediococcus acidilactici A40 strain.
Although all three nitrogen sources supply branched-chain amino acids (BCAAs), the relative balance of amino acids and peptide sizes differs substantially, which can influence metabolic regulation in LAB. Essential amino acids and BCAAs such as valine have been implicated in the modulation of glycolytic flux and cofactor balances in LAB, potentially diverting carbon away from lactate formation under conditions of imbalance or excess of certain peptides or amino acids. These metabolic adjustments may preserve cellular viability (growth and maintenance) even when LA production is reduced due to altered enzymatic activity or redox balance under specific nitrogen profiles.64 In this context, CSL and HSP may support biomass viability but result in lower LA yields compared with YE because of differences in amino acid proportions and their availability to central metabolic pathways.
These findings reinforce the strategic importance of identifying cost-effective nitrogen sources that maintain high LA yields without compromising LAB viability. While YE remains the most effective component in terms of fermentative performance, the partial substitution with CSL presents a viable alternative for reducing medium costs. However, achieving consistent outcomes with HSP may require further optimization of its physicochemical properties, particularly regarding peptide size distribution and degree of hydrolysis. In this context, a more detailed evaluation of cell viability and LA production becomes essential to assess the overall suitability of these alternative nitrogen sources within an industrial fermentation framework.
Recently, various residual nitrogen sources have been evaluated as suitable alternatives for LAB-based bioprocesses. These include dairy by-products, such as whey, which provides peptides and amino acids and can enhance yields when supplemented with YE or peptone;6 agricultural and food industry residues, including date palm by-products, barley sprouts, and liquid potato waste;7,65 and animal-derived materials, such as chicken feather hydrolysates66 and fish-processing residues67 (e.g., tuna heads, self-hydrolyzed viscera, and stingray viscera). All these sources are rich in amino acids and can effectively support the growth of LAB. Although some may yield lower productivity compared to conventional media, their use represents a cost-effective and environmentally sustainable alternative for LA fermentation.67 Insect-processing residues have also been explored as a nitrogen source for producing LAB biomass without the addition of an external carbon source, representing a novel and innovative approach that has shown promising results and opens further possibilities for application.68
Selection of the optimal condition maximizing response parameters and minimizing cost: application of the desirability function
According to the MD, the highest cell viability and LA concentration were achieved at the highest levels of YE. However, the primary objective of this study was to develop a cost-efficient production process of a bioinoculant and LA. Since LA is the main coproduct generated by P. acidilactici A40 during bioinoculant production and given its importance for downstream recovery and applications, as well as the need to produce LA as a food preservation bioproduct at a competitive cost, a desirability function was applied. This approach enabled the identification of an optimal medium that balances maximizing viable cell biomass and LA concentration while minimizing fermentation medium costs.It is evident that efforts to reduce costs by limiting the use of YE result in LA concentrations that are not the highest observed. Therefore, it is essential to achieve a compromise between minimizing costs and maintaining cell viability and LA concentration within acceptable ranges. To address this, a desirability analysis using the Derringer and Suich function (eqn (6)) was conducted within the experimental design space. The resulting surface plot, along with the contours for global desirability at the base, is presented in Fig. 6. It shows that the most desirable mixtures are located in the regions along the lateral borders of the diagram, closer to YE, with proportions above 40%. In contrast, the use of HSP at proportions greater than 30% is not recommended based on the observed performance in the plot. The combined use of CSL and YE, or HSP and YE, results in higher desirability values compared to the combination of all three components of the mixture simultaneously.
 | ||
| Fig. 6 Global desirability of the experiment maximizing cell viability and LA concentration while minimizing culture medium cost. | ||
Therefore, the strategy was to identify the highest desirability values within the feasible region, prioritizing combinations with lower YE proportions while still remaining within the high-desirability zone. This approach aligns with the study's objective of reducing fermentation medium costs. To determine the most suitable composition, an analysis of the global desirability behaviour across the proportions of each component was conducted, as shown in Fig. 7. Additionally, representative points with varying component proportions were selected based on their global desirability values, as presented in SI 4 (SI 4).
 | ||
| Fig. 7 Desirability profile (individual for each variable and global) along the proportions of YE, CSL and HSP. | ||
The desirability profile indicates that higher global desirability values, ranging from 0.60 to 0.80, were generally associated with YE and CSL proportions between 40% and 60%. Within this high-desirability range, LA concentrations reached up to 16.81 g L−1. Cell viability remained consistently above log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 CFU g−1 DB (considered suitable for the formulation operations) across the proportions of YE and CSL, while the inclusion of HSP decreased it. Additionally, the cost of the culture medium ranged from 0.48 to 0.57 USD L−1 in the most suitable desirability combinations.
11 CFU g−1 DB (considered suitable for the formulation operations) across the proportions of YE and CSL, while the inclusion of HSP decreased it. Additionally, the cost of the culture medium ranged from 0.48 to 0.57 USD L−1 in the most suitable desirability combinations.
It is worth noting that, in most of the most desirable combinations, CSL or HSP were present in minimal proportions. From a practical perspective, it would be expected that the synergistic effects of ternary mixtures outperform binary ones in balancing the desired responses. However, in this case, the latter achieved comparable or even superior response values to the ternary mixtures at reduced costs, which, theoretically, is not ideal.
The selection of an alternative nitrogen source to partially replace YE within the region of highest desirability is particularly relevant, as these conditions offer balanced performance by maintaining adequate LA concentrations that support downstream process efficiency, while moderately reducing medium costs. Considering the region of highest global desirability rather than a single optimum point, and with the aim of reducing medium costs, the mixture comprising 40% YE and 60% CSL was selected as a representative condition within this optimal region to experimentally validate the partial replacement of YE in the fermentation medium (see SI 4). In the validation experiment, a biomass viability of 12.23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log CFU g−1 DB was obtained, corresponding to approximately 10–11 g of viable biomass per liter of fermentation medium, together with a LA concentration of 14.54 g L−1. The corresponding values predicted by the fitted Scheffé mixture model were 11.68
log CFU g−1 DB was obtained, corresponding to approximately 10–11 g of viable biomass per liter of fermentation medium, together with a LA concentration of 14.54 g L−1. The corresponding values predicted by the fitted Scheffé mixture model were 11.68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log CFU g−1 DB and 14.42 g L−1, respectively, resulting in relative prediction errors of 4.7% for biomass viability and 0.8% for LA concentration. The close agreement between experimental and predicted values (<10%) for both responses confirms the robustness and predictive capability of the mixture model and supports its suitability for medium optimization purposes.
log CFU g−1 DB and 14.42 g L−1, respectively, resulting in relative prediction errors of 4.7% for biomass viability and 0.8% for LA concentration. The close agreement between experimental and predicted values (<10%) for both responses confirms the robustness and predictive capability of the mixture model and supports its suitability for medium optimization purposes.
Preliminary evaluation of biomass formulation
The fermentation medium composition and the process conditions can have a direct impact on bacterial cell resistance. Factors such as pH, osmotic pressure, temperature, and salinity are known to promote the production and accumulation of compounds that enhance cellular resistance.69,70 Pediococcus sp. has been reported to undergo changes in membrane fatty acid composition depending on growth conditions.71 Since drying and formulation operations can compromise cell viability, having cells with greater resistance is particularly important in the development of bioinoculants. LAB viability is critical for a successful establishment in silage and for preventing contamination by moulds and yeasts through rapid pH reduction. During formulation, protectants and conditioners are added to shield cell membranes and structures from stressors such as temperature, pH, and osmotic pressure.72 In this context, it was important to assess whether changes in the fermentation medium—specifically the partial replacement of YE with CSL—would affect the viability and storage stability of the formulated prototype.The combination of YE and CSL in the fermentation medium had no effect on the microbial viability of the prototype, during storage for up to 90 days. The initial viability of the formulated prototype was 3.24 × 1011 CFU g−1 for the medium containing only YE as the nitrogen source and 1.86 × 1011 CFU g−1 for the medium containing 40% YE and 60% CSL. After 90 days of storage at 25 °C, viability decreased to 7.50 × 107 CFU g−1 and 5.73 × 107 CFU g−1, respectively. These results indicate that, under the evaluated conditions, the nitrogen source composition of the fermentation medium did not substantially affect the survival of strain A40 over the 90-day storage period. The fluidized bed drying operation must be optimized, as the technological properties of LAB may be compromised by exposure to drying conditions in such systems. The selection of protective agents helps overcome the challenges associated with maintaining LAB viability during and after drying. These viability losses have already been reported when spray drying is applied to LAB cultures.70 Nevertheless, the results suggest that further improvement and optimization of the overall formulation scheme are still necessary.
Cost–benefit analysis
CBA evaluates the effects of implemented measures on costs and benefits in monetary terms and serves as an important source of information for companies in their decision-making processes from the early stages of research. The most significant cost in industrial microbial processes is associated with the production phase, particularly the culture medium, followed by purification, formulation, packaging, safety testing, and transportation.73 Hence, the use of agricultural byproducts and residual materials has been proposed as a viable and sustainable strategy to reduce the cost of the culture medium.The benefits are influenced by various consumer-related factors, which vary depending on the society to which the consumers belong. Consumer decisions depend not only on their willingness to pay, but also on broader social demands, such as the fulfilment of the Sustainable Development Goals (SDGs). In this sense, understanding market trends becomes essential for accurately assessing the potential benefits of a given biotechnological process.
The silage bioinoculant sector is experiencing sustained growth due to its ability to improve forage quality, stability and preservation—key aspects in animal nutrition. In 2024, the global market for silage inoculants and additives was valued at USD 1.2 billion, and it is projected to reach USD 2.0 billion by 2033, with a compound annual growth rate (CAGR) of 6.5% between 2026 and 2033. This growth is driven by an increasing global demand for efficient animal feed, along with a rising preference for sustainable, biologically based solutions. In particular, LAB-based inoculants are gaining prominence in regions with intensive livestock production, such as North America, Europe, and the Asia–Pacific region.74
The LA market, on the other hand, was valued at USD 511.11 million in 2024 and is projected to reach USD 624.54 million by 2029, with a CAGR of 4.09%. This growth is driven by the rising demand for food-grade acidulants, the widespread use of LA as an internationally approved preservative, and the role of LA as a key precursor of polylactic acid (PLA), a compostable bioplastic that is gaining increasing commercial and environmental relevance.75
Fig. 8 illustrates the net value derived from the CB analysis (eqn (6)). The results reveal that employing a combination of YE and CSL as nitrogen sources yields a higher economic return (2.34) compared to using a fermentation medium with YE alone (1.8). The combination of 40% YE and 60% CSL reduced the culture medium cost to 0.486 USD L−1, representing a 22.85% decrease relative to the YE-only medium.
Although this combination of nitrogen sources led to a slight reduction in LA concentration, the overall economic benefit was greater, as demonstrated by the CB index. This finding is particularly relevant considering that LA concentrations were only marginally affected, while viable biomass levels—crucial for bioinoculant functionality—remained stable and even showed a slight increase in the medium containing the combined nitrogen sources. A similar trend was reported in the development of a process to produce prodigiosin from Serratia marcescens, where modifications to the culture medium resulted in an increase in the saleable value compared to commercial production, as demonstrated through a CBA.76
In this analysis, the selling price of the bioinoculant was estimated at 90 USD kg−1. In the Colombian market, the price of bioinoculants, such as Magniva® (Lallemand, Canada), ranges from approximately 50 to 80 USD per 100 g bag, with variable availability. The considered cost of the bioinoculant significantly surpasses that of LA, which was estimated at 2 USD kg−1. In the market, the price of LA fluctuates between 2 and 15 USD L−1 depending on its purity and final application. The lower price used for LA in this study was based on a final concentration of 40–50 g L−1 after the separation process, with a purity of 50–60%, corresponding to applications in the food, pharmaceutical, or industrial-grade sectors. Consequently, the bioinoculant represents the major contributor to the overall process value due to its higher unit price. Importantly, Pediococcus acidilactici A40 is a native strain, and the bioprocess was specifically designed to enable the dual production of viable biomass and LA; therefore, preserving cell integrity is essential. Previous studies have demonstrated that stress factors, such as acidic conditions, can impair cell viability due to acid stress.77 While nutrient availability plays a key role in microbial performance, environmental stress factors such as acidification and osmotic pressure also influence the viability of Pediococcus acidilactici.19,78,79 This species is known to activate protective responses that help maintain intracellular homeostasis under adverse conditions.78 Consistent with these mechanisms, the results showed that strain P. acidilactici A40 was able to maintain high viability (>log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 109 CFU mL−1 when cultivated as free cells) under uncontrolled pH conditions and without LA removal. Notably, in all fermentations, the final pH reached values close to 4, yet the strain demonstrated a remarkable capacity to tolerate the combined effects of acid and osmotic stress.
109 CFU mL−1 when cultivated as free cells) under uncontrolled pH conditions and without LA removal. Notably, in all fermentations, the final pH reached values close to 4, yet the strain demonstrated a remarkable capacity to tolerate the combined effects of acid and osmotic stress.
In this context, several strategies—such as cell immobilization24 or metabolic engineering5,80,81—have been proposed to increase LA yields and, consequently, improve the cost–benefit ratio of fermentation processes. However, these approaches could not be implemented in this study, as the process was intentionally designed to recover both viable biomass and coproducts. Moreover, recombinant microbial bioinputs may have adverse effects on customer preferences, especially in the agricultural, cosmetic and food industries.73 Additionally, regulatory restrictions in many countries regarding the use of genetically modified strains in bioinput production further limit the applicability of such strategies.
In this regard, future scale-up studies should evaluate bioprocess strategies to increase LA titers, such as fed-batch operation,66,82 extractive fermentation using resins for LA recovery,83 and in situ LA removal via membrane-based bioprocesses.84–89 The recovery of organic acids from fermentation broths is widely recognized as a major technical and economic challenge, particularly under dilute product conditions. Downstream processing can account for more than 50% of total process costs due to the high water content and complexity of fermentation media, as well as the limitations of both conventional and emerging separation technologies.90 Nevertheless, downstream technologies for LA recovery have advanced to enable recovery from lower LA concentrations. For example, ion-exchange technology has reported recovery efficiencies above 80% at LA concentrations of approximately 10 g L−,91 facilitating processing at lower titers. Similarly, a separate five-step lactic acid recovery process applied to broths with ≈3 g L−1, associated with cultured-meat production, reported a net recovery cost of US$0.71 per kg of 88% LA and a simple payback period of 7.5 years.92
Amid the growing global demand for both products, the initial CBA analysis—focused on the fermentation medium cost, one of the main factors influencing variable costs—provides insights into the future economic feasibility of dual production processes for bioinputs and bioproducts. The results obtained from the CBA at this early research stage show values close to 2, indicating a margin that allows for the inclusion of additional parameters in subsequent stages. In particular, further CBAs in more advanced research phases could integrate additional factors from the techno-economic analysis that have an effect on the overall assessment.
Capital expenditures (CAPEX) are largely determined by the choice of process equipment and the installed capacity, while operating expenses (OPEX) encompass utilities and labour. The CBA may also be affected by fluctuations in the prices of the fermentation medium and final products, which should be examined through sensitivity analysis. Although the present analysis focuses on fermentation medium costs and product pricing, future studies should expand the economic scope to include CAPEX, OPEX, sensitivity analysis, and net present value (NPV) to provide a more comprehensive techno-economic evaluation. The demonstrated profitability of simultaneous production of a bioinoculant and LA using a low-cost fermentation medium underscores the strategic relevance of this approach for meeting growing market demand while promoting process sustainability.
Conclusions
This study demonstrates that partially replacing YE with CSL in the fermentation medium (40% YE and 60% CSL) enabled the co-production of highly viable bacterial biomass (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12.23 CFU g−1 DB), with approximately 20% lower LA concentration relative to the maximum observed when using only YE. This reduction remained within the high-desirability region and was offset by improved economics. This outcome is critical for developing a dual-purpose bioprocess that yields both a silage bioinoculant and LA for food applications. A CBA confirmed that the YE–CSL combination provided a more favourable economic return than YE alone: the medium cost was reduced by 22.85%, and overall economic performance improved. Within the explored design space—and using a desirability function that weighted technological variables (viability and LA) threefold relative to cost (w_viability = w_LA = 3; w_cost = 1)—the YE–CSL blend emerged as the most suitable combination. During 90 days of storage, no differential effect attributable to the nitrogen source was detected, although formulation (drying/protectants) still requires optimisation. Urea showed inhibitory effects in the tested range, and HSP underperformed relative to YE and CSL.
12.23 CFU g−1 DB), with approximately 20% lower LA concentration relative to the maximum observed when using only YE. This reduction remained within the high-desirability region and was offset by improved economics. This outcome is critical for developing a dual-purpose bioprocess that yields both a silage bioinoculant and LA for food applications. A CBA confirmed that the YE–CSL combination provided a more favourable economic return than YE alone: the medium cost was reduced by 22.85%, and overall economic performance improved. Within the explored design space—and using a desirability function that weighted technological variables (viability and LA) threefold relative to cost (w_viability = w_LA = 3; w_cost = 1)—the YE–CSL blend emerged as the most suitable combination. During 90 days of storage, no differential effect attributable to the nitrogen source was detected, although formulation (drying/protectants) still requires optimisation. Urea showed inhibitory effects in the tested range, and HSP underperformed relative to YE and CSL.
Beyond its technical and economic relevance, this approach advances bioeconomy principles in two key aspects: it valorises a native microbial resource, Pediococcus acidilactici A40, and integrates the production of bacterial biomass for a bioinoculant and LA within a single process. Notably, this co-production was achieved by partially replacing YE with CSL, an agro-industrial residue used here as an alternative organic nitrogen source, thereby improving process sustainability and cost-effectiveness. This integrated strategy enhances resource efficiency, potentially reduces reliance on imported inputs where CSL is locally available, and contributes to sustainable, circular bioprocesses tailored to local needs. Although further work is needed to optimise formulation conditions and improve long-term biomass stability, these findings provide a strong basis for developing cost-competitive bioinputs and bioproducts for the local market.
Author contributions
LFMA: conceptualization, methodology, software, formal analysis, investigation, data curation, writing – original draft, writing – review & editing, visualization; ECG: investigation; DFCR: investigation; LM: conceptualization, methodology, formal analysis, data curation, writing – original draft, writing – review & editing, visualization, supervision, project administration. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declared that this work was performed without any commercial or financial relationships, and thus there is no conflict of interest.List of abbreviations
| LAB | Lactic acid bacteria |
| GRAS | Generally recognized as safe |
| LA | Lactic acid |
| YE | Yeast extract |
| CSL | Corn steep liquor |
| HSP | Hydrolysed soy protein |
| MRS | De Man, Rogosa, Sharpe |
| MD | Mixture design |
| OLS | Ordinary least squares |
| DB | Dry biomass |
| BCAA | Branched-chain amino acid |
| CBA | Cost–benefit analysis |
| CB | Cost-benefit index |
Data availability
All data regarding this research work are available through the manuscript.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fb00684h.
References
- T. H. Nguyen, X. Wang, D. Utomo, E. Gage and B. Xu, Cleaner and Circular Bioeconomy, 2025, 11 DOI:10.1016/j.clcb.2025.100145.
- M. Rajković, D. Popović-Minić, D. Milinčić and M. Zdravković, Zast. Mater., 2020, 61, 229–250 Search PubMed.
- H. Alhaag, X. Yuan, A. Mala, J. Bai and T. Shao, Appl. Sci., 2019, 9, 1247 CrossRef CAS.
- A. Sifeeldein, X. Yuan, Z. Dong, J. Li and T. Shao, Kafkas Univ Vet Fak Derg, 2018, 24(3), 371–378 Search PubMed.
- E. Abedi and S. M. B. Hashemi, Heliyon, 2020, 6, e04974 Search PubMed.
- G. Juodeikiene, D. Zadeike, E. Bartkiene and D. Klupsaite, LWT–Food Sci. Technol., 2016, 72, 399–406 Search PubMed.
- G. Juodeikiene, D. Klupsaite, D. Zadeike, D. Cizeikiene, I. Vidziunaite, E. Bartkiene and D. Cernauskas, Int. J. Food Sci. Technol., 2016, 51, 2682–2691 CrossRef CAS.
- J. A. Mora-Villalobos, J. Montero-Zamora, N. Barboza, C. Rojas-Garbanzo, J. Usaga, M. Redondo-Solano, L. Schroedter, A. Olszewska-Widdrat and J. P. López-Gómez, Fermentation, 2020, 6, 1–21 CrossRef.
- Z. Y. Zhang, B. Jin and J. M. Kelly, World J. Microbiol. Biotechnol., 2007, 23, 229–236 Search PubMed.
- L. F. Mejía-Avellaneda, H. Suárez, H. Jiménez and L. Mesa, Crit. Rev. Biotechnol., 2022, 42, 1028–1044 Search PubMed.
- J. I. I. Fugaban, J. E. Vazquez Bucheli, Y. J. Park, D. H. Suh, E. S. Jung, B. D. G. de M. Franco, I. V. Ivanova, W. H. Holzapfel and S. D. Todorov, J. Appl. Microbiol., 2022, 132, 311–330 Search PubMed.
- J. Barbosa, S. Borges and P. Teixeira, Int. J. Food Sci. Technol., 2015, 50, 1151–1157 CrossRef CAS.
- M. C. W. Porto, T. M. Kuniyoshi, P. O. S. Azevedo, M. Vitolo and R. P. S. Oliveira, Biotechnol. Adv., 2017, 35(3), 361–374 Search PubMed.
- L. Biliavska, Y. Pankivska, O. Povnitsa and S. Zagorodnya, Medicina, 2019, 55, DOI:10.3390/medicina55090519.
- Y. Qi, L. Huang, Y. Zeng, W. Li, D. Zhou, J. Xie, J. Xie, Q. Tu, D. Deng and J. Yin, Front. Microbiol., 2021, 12, 762467 CrossRef PubMed.
- B. F. Carvalho, G. F. C. Sales, R. F. Schwan and C. L. S. Ávila, J. Appl. Microbiol., 2021, 130, 341–355 Search PubMed.
- H. Katepogu, Y. J. Wee, K. Anu Appaiah, S. V. Chinni, S. C. B. Gopinath, A. Syed, M. Verma and V. R. Lebaka, Biomass Convers. Biorefin., 2025, 15, 22347–22357 CrossRef CAS.
- Z. Zhang, Y. Li, J. Zhang, N. Peng, Y. Liang and S. Zhao, Microorganisms, 2020, 8, 1–9 Search PubMed.
- T. Aziz, M. Naveed, M. A. Shabbir, J. Naseeb, A. Sarwar, L. Zhao, Z. Yang, C. Haiying, L. Lin, A. Shami and F. Al-Asmari, Sci. Rep., 2025, 15, 6050 Search PubMed.
- C. Vargas, D. Bautista, H. Jimenez, M. Soto-Suarez, S. Restrepo, C. Gonzalez and P. Zuluaga, Microbiol. Resour. Announce., 2023, 12, e00530 Search PubMed.
- C. Vargas, L. Botero, E. Rodriguez, L. Dávila, C. Racedo, C. Barrera, A. J. Bernal, H. Jiménez, P. Zuluaga and C. González, Eur. J. Plant Pathol., 2025, 172, 7–22 CrossRef CAS.
- C. Vargas, H. R. Jiménez, C. G. Almario and A. G. Almario, J. Appl. Microbiol., 2023, 134(2), lxac053 Search PubMed.
- Y. Sun, Z. Xu, Y. Zheng, J. Zhou and Z. Xiu, Process Biochem., 2019, 81, 132–138 Search PubMed.
- A. Thakur, P. S. Panesar and M. S. Saini, Waste Biomass Valorization, 2019, 10, 1119–1129 Search PubMed.
- H. Xu, D. Li, X. Jiang, Q. Pei, Z. Li, P. Madjirebaye, M. Xie, T. Xiong and Z. Liu, Foods, 2025, 14(2), 150 Search PubMed.
- J. R. Ngouénam, P. M. Kaktcham, C. H. Momo Kenfack, E. M. Foko Kouam and F. Z. Ngoufack, Int. J. Food Sci., 2021, 2021, 1742018 Search PubMed.
- J. Campos, J. Bao and G. Lidén, J. Biotechnol., 2021, 335, 1–8 Search PubMed.
- B. Savithra Krishna, N. K. Saibaba, S. Sai Nikhilesh Gantala, B. Tarun, G. Sarva Sai Nikhilesh, N. K. Saibaba V and R. Gopinadh, Int. J. Biotechnol. Res., 2018, 1, 42–54 Search PubMed.
- P. S. Panesar and S. Kaur, Int. J. Food Sci. Technol., 2015, 50, 2143–2151 Search PubMed.
- J. R. Xavier, I. Nallamuthu and O. P. Chauhan, Sustainable Food Technol., 2024, 2, 741–749 Search PubMed.
- M. Altaf, B. J. Naveena and G. Reddy, Bioresour. Technol., 2007, 98, 498–503 CrossRef CAS PubMed.
- L. F. Coelho, C. J. B. De Lima, M. P. Bernardo and J. Contiero, Appl. Biochem. Biotechnol., 2011, 164, 1160–1171 CrossRef CAS PubMed.
- F. Hernández-Rosas, J. D. Castilla-Marroquín, J. M. Loeza-Corte, M. A. Lizardi-Jiménez and R. Hernández-Martínez, Rev. Mex. Ing. Quim., 2021, 20, Bio2429 Search PubMed.
- G. C. Derringer, Rubber Chem. Technol., 1988, 61, 377–421 CAS.
- G. Derringer and R. Suich, J. Qual. Technol., 1980, 12, 214–219 CrossRef.
- R Core Team, The R Foundation for Statistical Computing, 2024, https://www.R-project.org/ Search PubMed.
- RStudio Team, Posit Software, PBC, 2025, https://posit.co/products/open-source/rstudio/ Search PubMed.
- N. J. Krüger, C. Buhler, A. N. Iwobi, I. Huber, L. Ellerbroek, B. Appel and K. Stingl, PLoS One, 2014, 9, e88108 Search PubMed.
- J. C. De Man, M. Rogosa, M. E. Sharpe and J. oppl, Bact, 1960, 23, 130–135 Search PubMed.
- L. F. Mejía-Avellaneda, F. Romero-Perdomo, V. Chavarro-Anzola and L. Mesa, Circ. Econ. Sustain., 2025, 5, 4731–4753 CrossRef.
- S. M. Bhatt and S. K. Srivastava, Food Biotechnol., 2008, 22, 115–139 Search PubMed.
- N. Nancib, A. Nancib, A. Boudjelal, C. Benslimane, F. Blanchard and J. Boudrant, Bioresour. Technol., 2001, 78, 149–153 Search PubMed.
- A. Nancib, N. Nancib, D. Meziane-Cherif, A. Boubendir, M. Fick and J. Boudrant, Bioresour. Technol., 2005, 96, 63–67 Search PubMed.
- M. Hujanen and Y.-Y. Linko, Appl. Microbiol. Biotechnol., 1996, 45, 307–313 Search PubMed.
- Y. H. Lim, H. L. Foo, T. C. Loh, R. Mohamad and R. A. Rahim, Molecules, 2020, 25, 779 CrossRef CAS PubMed.
- D. Mora and S. Arioli, PLoS Pathog., 2014, 10, e1004472 Search PubMed.
- S. Arioli, G. Della Scala, A. Martinović, L. Scaglioni, S. Mazzini, F. Volonté, M. B. Pedersen and D. Mora, Microbiol. Spectr., 2022, 10, e02760–21 Search PubMed.
- N. M. Salatein, R. K. Hassan, R. Adel, S. Desouky, N. M. Salatein, R. A. Ibrahim, S. E. Desouky, E. F. El-Belely, M. A. Abdel-Rahman and I. Fahim, Results Eng., 2025, 105161 Search PubMed.
- L. Yu, T. Lei, X. Ren, X. Pei and Y. Feng, Biochem. Eng. J., 2008, 39, 496–502 CrossRef CAS.
- Z. Bai, Z. Gao, J. Sun, B. Wu and B. He, Bioresour. Technol., 2016, 207, 346–352 Search PubMed.
- Y. Lamas, A. C. de Armas, Y. Albernas and E. González, Cent. Azúcar, 2023, 50, e1035 Search PubMed.
- P. da Silva Delabona, C. Sanchez Farinas, D. J. da Silva Lima and J. G. Da Cruz Pradella, Bioresour. Technol., 2013, 132, 401–405 CrossRef CAS PubMed.
- H. Yin, Z. Chen, Z. Gu and Y. Han, LWT–Food Sci. Technol., 2009, 42, 327–331 Search PubMed.
- C. J. Bolner De Lima, L. Fontes Coelho, K. C. Blanco and J. Contiero, Afr. J. Biotechnol., 2009, 8, 5842–5846 Search PubMed.
- L. Djemal, J. von Hagen, H. Kolmar and V. Deparis, Biotechnol. Prog., 2021, 37, e3147 CrossRef CAS PubMed.
- T. Utami, E. N. Kusuma, R. Satiti, E. S. Rahayu and M. N. Cahyanto, Int. Food Res. J., 2019, 26, 117–122 CAS.
- M. Hamdi, S. Hamza, N. Mtimet, N. Hmida, C. Cornelius, S. Zgouli, A. Mahjoub and P. Thonart, Bioprocess Eng., 2000, 22, 23–27 Search PubMed.
- S. Kim, Y. Lee, M. Kim and S. Kim, J. Biosci. Bioeng., 2003, 96, 134–140 CrossRef.
- X. Guo, D. Xu, F. Li, J. Bai and R. Su, Microb. Biotechnol., 2023, 16, 67–87 Search PubMed.
- C. O. Okoye, Y. Wang, L. Gao, Y. Wu, X. Li, J. Sun and J. Jiang, Microbiol. Res., 2023, 266, 127212 Search PubMed.
- Z. Tao, H. Yuan, M. Liu, Q. Liu, S. Zhang, H. Liu, Y. Jiang, D. Huang and T. Wang, J. Microbiol. Biotechnol., 2023, 33, 151–166 CrossRef CAS PubMed.
- C. M. Hsieh, F.-C. Yang and E. L. Iannotti, Process Biochem., 1999, 34, 173–179 Search PubMed.
- X. Li, W. Xu, J. Yang, H. Zhao, H. Xin and Y. Zhang, Anim. Nutr., 2016, 2, 345–350 CrossRef PubMed.
- Y. Zhou, X. Zhang, Y. Wang and H. Liu, Fermentation, 2024, 10(4), 179 Search PubMed.
- Z. Zhang, Y. Li, J. Zhang, N. Peng, Y. Liang and S. Zhao, Microorganisms, 2020, 8, 1–9 Search PubMed.
- L. Paulova, J. Chmelik, B. Branska, P. Patakova, M. Drahokoupil and K. Melzoch, Braz. Arch. Biol. Technol., 2020 DOI:10.1590/1678-4324-2020190151.
- P. J. Yeboah, S. A. Ibrahim and A. Krastanov, Food Sci. Appl. Biotechnol., 2023, 2023, 215–240 Search PubMed.
- A. Vilas-Franquesa, C. Lakemond and M. Mishyna, Bioresour. Technol., 2024, 413, 131540 CrossRef CAS PubMed.
- P. B. Conrad, D. P. Miller, P. R. Cielenski and J. J. De Pablo, Cryobiology, 2000, 41, 17–24 Search PubMed.
- M. T. C. Moreira, E. Martins, Í. T. Perrone, R. de Freitas, L. S. Queiroz and A. F. de Carvalho, Compr. Rev. Food Sci. Food Saf., 2021, 20, 3267–3283 Search PubMed.
- B. A. Annous, M. F. Kozempel and M. J. Kurantz, Appl. Environ. Microbiol., 1999, 65, 2857–2862 Search PubMed.
- I. Coulibaly, R. Dubois-Dauphin, S. Danthine, L. Majad, T. Mejoub, J. Destain, F. Béra, J.-P. Wathelet and P. Thonart, Biotechnol. Agron. Soc. Environ., 2011, 15, 287–299 Search PubMed.
- K. Fukuda and H. Kono, in Microbial Exopolysaccharides as Novel and Significant Biomaterials, A. Kumar, K. V. Sajna and S. Sharma, Springer Nature, Switzerland, 1st edn, 2021, pp. 303–339 Search PubMed.
- Verified Market Research, Global Business Solutions Global Silage Inoculant and Additive Market, 2025 Search PubMed.
- Mordor Intelligence, Tamaño del mercado de ácido láctico y análisis de participación tendencias de crecimiento y pronósticos (2024-2029), 2023 Search PubMed.
- T. Paul, A. Mondal, T. K. Bandyopadhyay and B. Bhunia, Biomass Convers. Biorefin., 2024, 14, 4091–4110 Search PubMed.
- K. Papadimitriou, Á. Alegría, P. A. Bron, M. de Angelis, M. Gobbetti, M. Kleerebezem, J. A. Lemos, D. M. Linares, P. Ross, C. Stanton, F. Turroni, D. van Sinderen, P. Varmanen, M. Ventura, M. Zúñiga, E. Tsakalidou and J. Kok, Microbiol. Mol. Biol. Rev., 2016, 80, 837–890 CrossRef CAS PubMed.
- A. S. Derunets, A. I. Selimzyanova, S. V. Rykov, A. E. Kuznetsov and O. V. Berezina, World J. Microbiol. Biotechnol., 2024, 40, 126 CrossRef CAS PubMed.
- M. Othman, A. B. Ariff, H. Wasoh, M. R. Kapri and M. Halim, AMB Express, 2017, 7(1), 215 CrossRef PubMed.
- L. Liu, D. Yang, Z. Zhang, T. Liu, G. Hu, M. He, S. Zhao and N. Peng, Appl. Environ. Microbiol., 2021, 87, 1–13 Search PubMed.
- X. Yi, P. Zhang, J. Sun, Y. Tu, Q. Gao, J. Zhang and J. Bao, J. Biotechnol., 2016, 217, 112–121 CrossRef CAS PubMed.
- J. Campos, L. G. Tejada, J. Bao and G. Lidén, Process Biochem., 2023, 125, 162–170 CrossRef CAS.
- M. Othman, A. B. Ariff, M. R. Kapri, L. Rios-Solis and M. Halim, Front. Microbiol., 2018, 9, 2554 CrossRef PubMed.
- J. Sikder, S. Chakraborty, P. Pal, E. Drioli and C. Bhattacharjee, Biochem. Eng. J., 2012, 69, 130–137 CrossRef CAS.
- M. Boonmee, O. Cotano, S. Amnuaypanich and N. Grisadanurak, Arabian J. Sci. Eng., 2016, 41, 2067–2075 Search PubMed.
- M. Othman, A. B. Ariff, L. Rios-Solis and M. Halim, Front. Microbiol., 2017, 8 DOI:10.3389/fmicb.2017.02285.
- M. Othman, A. B. Ariff, H. Wasoh, M. R. Kapri and M. Halim, AMB Express, 2017, 7(1), 215 CrossRef PubMed.
- N. Phanthumchinda, S. Thitiprasert, S. Tanasupawat, S. Assabumrungrat and N. Thongchul, Process Biochem., 2018, 68, 205–213 CrossRef CAS.
- M. Othman, A. B. Ariff, M. R. Kapri, L. Rios-Solis and M. Halim, Front. Microbiol., 2018, 9, 2554 CrossRef PubMed.
- A. A. Kiss, J. P. Lange, B. Schuur, D. W. F. Brilman, A. G. J. van der Ham and S. R. A. Kersten, Biomass Bioenergy, 2016, 95, 296–309 CrossRef CAS.
- X. Vecino, M. Reig, C. Valderrama and J. L. Cortina, Water, 2021, 13(11), 1572 CrossRef CAS.
- J. Wimble, R. Ashizawa and E. W. Swartz, Biotechnol. Prog., 2025, e70094 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




