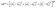Open Access Article
Open Access ArticleSustainable shelf-life extension of date palm–bael–jujube beverages through HTST pasteurization and protective packaging
Pitchaya Tuntiteeraboona,
Saeid Jafaria,
Sochannet Chheng ab,
Khursheed Ahmad Shiekha,
Isaya Kijpatanasilpac and
Kitipong Assatarakul
ab,
Khursheed Ahmad Shiekha,
Isaya Kijpatanasilpac and
Kitipong Assatarakul *a
*a
aDepartment of Food Technology, Faculty of Science, Chulalongkorn University, Bangkok, Thailand 10330. E-mail: Kitipong.A@chula.ac.th; Tel: +66-2218-5328
bDepartment of Food Chemical Engineering, Kampong Speu Institute of Technology, Kampong Speu, 050601, Cambodia
cDepartment of Food Science, Purdue University, West Lafayette, IN 47907, USA
First published on 20th March 2026
Abstract
The impacts of packaging (transparent glass bottle vs. amber-colored glass bottle) and pasteurization (LTLT vs. HTST) as viable options for preserving ready-to-drink beverages made from date palm extract mixed with bael fruit and jujube (date palm extract 70%: bael fruit juice 15%: jujube juice 15%) were investigated in this study. Physico–chemical properties and microbiological counts were also investigated during storage at 4 °C. Pasteurization and packaging type significantly affected the preservation of bioactives and antioxidant activity of products during cold storage (p ≤ 0.05). At the end of the storage period (9 weeks), the HTST-pasteurized samples in amber-colored glass bottles exhibited the highest total phenolic compound (586.33 mg GAE/100 mL), total carotenoid content (3.25 mg β-CE/100 mL), and antioxidant activity (DPPH: 763.54 mM Trolox/100 mL; FRAP: 786.21 mM Trolox/100 mL). During storage, total soluble solids (ranging from 6.6 to 7.2 °Brix) showed no significant changes (p > 0.05). HTST pasteurization caused the least color change (ΔE) in amber-colored glass bottles during storage. No microbial, yeast, or mold counts were detected in HTST-pasteurized samples stored in amber-colored glass bottles until the 6th and 7th weeks of storage, respectively. Based on maximum antioxidant activity retention and microbiological safety, the shelf life of HTST-pasteurized samples in amber-colored glass bottles was calculated to be 7 weeks. Finally, the use of HTST pasteurization together with amber-colored glass packaging may represent a practical approach for improving the preservation of ready-to-drink beverages.
Sustainability spotlightThis study presents a sustainable approach for extending the shelf life of ready-to-drink fruit-based beverages by combining high-temperature short-time (HTST) pasteurization with amber-colored glass packaging. This method effectively preserves bioactive compounds, including phenolics and carotenoids, and maintains antioxidant activity, while ensuring microbial safety during storage. HTST pasteurization has been shown to require approximately 30% less energy than low-temperature long-time (LTLT) methods due to shorter processing times and rapid heat transfer, contributing to improved energy efficiency in food processing operations (Stoforos, 2016). By extending shelf life, this approach reduces food spoilage and waste, which can account for 20–30% of post-production losses in fruit-based beverages (FAO, 2019), thereby supporting more efficient resource utilization. Amber glass packaging further enhances sustainability: it is highly recyclable with material recovery rates exceeding 90% in well-established glass recycling systems (Ahlstrand, 2025), and its durability reduces dependence on single-use PET plastics. Life cycle assessment studies indicate that PET bottles typically generate 70–85% higher greenhouse gas emissions per functional unit compared to reusable glass packaging systems when reuse rates are adequately high (Issifu & Sumaila, 2025). Overall, this strategy promotes both product quality and environmental sustainability in the beverage industry by combining energy-efficient processing, waste minimization, and environmentally responsible packaging. |
1 Introduction
The date palm (Phoenix dactylifera L.) is a Middle Eastern native that thrives in dry regions. It contains carbohydrates, dietary fibers, vitamins (C, B1, B2, and A), and minerals (calcium, iron, potassium, and zinc). Date palm phytochemicals are categorized into three categories: phenolic, carotenoid, and tannins.1 Instead of using compression methods, enzymatic extraction using pectinase causes the fruit's characteristics to increase the release of bioactive substances such as pigments and antioxidants from plant tissues.2Researchers have expected the growth of unique beverage recipes as consumption preferences have switched to the selection of healthy and helpful beverages. A drink made from date palm extract, bael fruit, and jujube, all of which are high in antioxidants and vitamins, is a viable example.3,4 However, even when refrigerated at 4 °C, these beverages have a short shelf life of about 2 weeks.
As a result, it is vital to utilize a preservation method that can increase the product's shelf life. Pasteurization in combination with consumer-friendly packaging that preserves the quality of ready-to-drink (RTD) beverage goods is a viable alternative. The packaging of a product has a significant impact on its shelf life.5,6 Choosing the correct packaging in combination with pasteurization not only extends the shelf life of the food, but also protects it against moisture, microbiological contamination, light, air, and different gases during handling, shipping, and storage.7,8
Despite the importance of both thermal processing and packaging in sustainable beverage preservation, limited research has investigated complementary effects of pasteurization and protective packaging on the quality and stability of RTD functional beverages, particularly those containing multiple fruit sources.
Therefore, this study evaluated the influence of pasteurization conditions (low-temperature long-time (LTLT) and high-temperature short-time (HTST)) and glass bottle color (transparent vs. amber) on the physicochemical characteristics, bioactive retention, antioxidant activity, and microbiological quality of a date palm–based RTD beverage mixed with bael fruit and jujube during refrigerated storage. The findings are expected to provide insights into energy-efficient and sustainable preservation strategies for functional beverages with extended shelf life.
2 Materials and methods
2.1. Sample preparation
The fresh date palms (Bahi variety) at the rutab stage were supplied from Kanchanaburi province in Thailand. Dried bael fruit and jujube were also obtained from Yaowarat Old Market in Bangkok, Thailand. All samples were delivered to Department of Food Technology, Chulalongkorn University. The date palms were dried in a hot air oven (Memmert, DO 6062, Germany) at 60 °C for 48 hours until moisture content was reduced to <10%, and then vacuum-packed in aluminum-laminated foil bags, and kept at −20 °C for subsequent use.For date palm juice extraction, dried date palms were rehydrated in distilled water at a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (w/v) and subjected to enzymatic treatment with 0.1% pectinase (Sigma-Aldrich, USA) at 45 °C for 2 h to enhance juice yield and bioactive compound extraction. The mixture was then filtered through cheesecloth to obtain the juice. Date palm concentrate (33.33% w/v) was prepared by dissolving the concentrated extract in distilled water to achieve the desired concentration.
3 (w/v) and subjected to enzymatic treatment with 0.1% pectinase (Sigma-Aldrich, USA) at 45 °C for 2 h to enhance juice yield and bioactive compound extraction. The mixture was then filtered through cheesecloth to obtain the juice. Date palm concentrate (33.33% w/v) was prepared by dissolving the concentrated extract in distilled water to achieve the desired concentration.
Bael fruit juice and jujube juice were obtained separately by boiling dried bael fruit and dried jujube, respectively, in distilled water at a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v) for 5 min, followed by filtration through cheesecloth. The final ready-to-drink beverage was formulated by mixing date palm extract (70% v/v), bael fruit juice (15% v/v), and jujube juice (15% v/v).
10 (w/v) for 5 min, followed by filtration through cheesecloth. The final ready-to-drink beverage was formulated by mixing date palm extract (70% v/v), bael fruit juice (15% v/v), and jujube juice (15% v/v).
2.2. Preparation of the ready-to-drink beverage from date palm extract
Based on our prior experiment, the optimum extraction conditions as well as the best ready-to-drink beverage from date palm extract were used (data not shown). The best ready-to-drink beverage was date palm juice extract (70% v/v) mixed with beal fruit juice (15% v/v) and jujube juice (15% v/v). Date palm juice was prepared from date palm concentrate at a concentration of 33.33% (w/v), while bael fruit juice and jujube juice were obtained by boiling dried bael fruit and dried jujube, respectively, for 5 min at a ratio of fruit to water of 10% (w/v).2.3. Thermal processing and packaging of the ready-to-drink beverage from date palm extract during storage at 4 °C
The thermal treatments were applied according to established pasteurization protocols. For HTST pasteurization, the beverage samples were rapidly heated to 72 °C and held at this temperature for 20 s using a steam-jacketed pasteurizer to ensure uniform heat distribution, as described in ref. 9. For LTLT pasteurization, the samples were gradually heated to 63 °C and maintained at this temperature for 30 min in a thermostatically controlled water bath. Following heat treatment, the samples were immediately subjected to rapid cooling in an ice bath to quickly reduce the temperature to 4 °C within 5 min, thereby preventing overprocessing and preserving bioactive compounds. Pre-sterilized (autoclaved at 121 °C for 15 min) transparent and amber-colored glass bottles (250 mL capacity) were aseptically filled with both HTST and LTLT-treated beverages and sealed immediately with sterilized screw caps under a laminar flow hood to minimize post-process contamination. As a control, unpasteurized samples were handled under the same aseptic conditions without thermal treatment. All samples were stored at 4 °C for up to 9 weeks, with weekly assessments of physicochemical parameters to monitor product quality and safety during storage.2.4. Determination of physical and chemical properties of the ready-to-drink beverage from date palm extract during storage at 4 °C
Color was determined by using a chroma meter (Monica Minolta CR-400, Japan) and CIE color system and L*, a* and b* values were adjusted by the machine before every sample measurement. L* represents the lightness value ranging from 0 (dark) to 100 (white), a* represents redness (+a*) and greenness (−a*) and b* represents yellowness (+b*) and blueness (−b*). The color difference (ΔE*) was measured by using the following equation:where subscript 1 is the initial color value and subscript 2 is the color value measured at a specific time point.
Total soluble solid content (°Brix) was measured by using a digital refractometer (HI96801, Hanna, USA) at 0–85 °Brix. The pH was measured by using a pH meter (Mettler toledo, Switzerland). The determination of total acid content (% citric acid) was conducted by the titration method according to ref. 10.
Total phenolic compound (TPC) was determined by using the Folin–Ciocalteu reagent as described in ref. 11 with some modifications. In brief, the sample (100 µL) was mixed with distilled water (7 mL) and Folin–Ciocalteu reagent (500 µL) and left at room temperature for 3–5 min. Then, sodium carbonate (400 µL) was added to the solution and left for another 30 min in a dark place. The absorption was measured with a UV-visible spectrophotometer (GENE-SYSTM 20 Visible, Thermo Fisher Scientific, USA) at a wavelength of 765 nm and the TPC was calculated using the absorption obtained compared with the gallic acid solution standard curve and the results were represented as mg gallic acid equivalent/liter (mg GAE/100 mL).
Total carotenoid content was determined using a spectrophotometric method as described in ref. 12, with slight modifications. Briefly, an aliquot of the sample was extracted with an appropriate solvent system (e.g., acetone: hexane, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 v/v) and centrifuged at 5000×g for 10 min to remove particulates. The clear supernatant was collected, and the absorbance was measured at 450 nm using a UV-visible spectrophotometer (GENE-SYSTM 20 Visible, Thermo Fisher Scientific, USA). A standard curve was prepared using known concentrations of β-carotene dissolved in the same solvent system. Total carotenoid content was calculated from the standard curve and expressed as milligrams of β-carotene equivalents per 100 mL of the sample (mg β-CE/100 mL).
6 v/v) and centrifuged at 5000×g for 10 min to remove particulates. The clear supernatant was collected, and the absorbance was measured at 450 nm using a UV-visible spectrophotometer (GENE-SYSTM 20 Visible, Thermo Fisher Scientific, USA). A standard curve was prepared using known concentrations of β-carotene dissolved in the same solvent system. Total carotenoid content was calculated from the standard curve and expressed as milligrams of β-carotene equivalents per 100 mL of the sample (mg β-CE/100 mL).
The antioxidant activity was obtained by the 2,2-diphenly-1-picrylhydrazyl (DPPH) method as described previously in ref. 13. The sample (250 µL) was mixed with DPPH solution (4.75 mL) and set in the dark at room temperature for 15 min. The absorbance of the samples was measured at a wavelength of 515 nm using methanol as a blank. Then, the absorption value of the DPPH solution (Ainitial) was subtracted from the absorbance of the sample (Afinal). The calculation of the DPPH was based on the Adifferance values and the standard curve of Trolox and expressed as mM Trolox/100 mL.
The antioxidant activity was determined by ferric reducing antioxidant power (FRAP) assay as described in ref. 14 with slight modifications. The sample (50 µL) was mixed with FRAP solution (950 µL) and left at room temperature for 4 min. The absorbance of the sample was obtained at a wavelength of 593 nm, using distilled water as the blank. The concentration of FRAP was determined based on the absorbance difference of the sample and the FRAP solution as well as the standard curve of Trolox and expressed as mM Trolox/100 mL.
All analyses were performed in triplicate. Standard curves were prepared using gallic acid (0–100 mg/L, R2 > 0.99) for TPC, β-carotene (0–10 µg/mL, R2 > 0.99) for TCC, and Trolox (0–500 µM, R2 > 0.99) for DPPH and FRAP assays. Method validation included recovery studies (95–105%) and precision assessment (RSD < 5%).
2.5. Determination of total plate count and yeast and mold count of the ready-to-drink beverage from date palm extract during storage at 4 °C
For microbiological analyses, the samples were aseptically transferred to sterile containers, and serial dilutions were prepared using 0.1% peptone water. For total plate count, 1 mL of appropriate dilutions was pipetted into sterile Petri dishes, followed by the addition of 15–20 mL of molten Plate Count Agar (PCA; Merck, Germany) cooled to 45 °C. The plates were gently swirled for even distribution and allowed to solidify. After solidification, plates were incubated at 35 ± 2 °C for 48 hours. For yeast and mold enumeration, 0.1 mL of appropriate dilutions was spread onto the surface of pre-poured PDA (Merck, Germany) plates acidified with 10% tartaric acid to inhibit bacterial growth. Plates were incubated at 25 ± 2 °C for 3–5 days. Colonies were counted and the results expressed as log CFU/mL.Coliform counts were determined according to the method described in ref. 15 and expressed as the most probable number per milliliter (MPN/mL) of sample. The three-tube MPN technique was employed using Lauryl Tryptose Broth (LTB; Merck, Germany) for presumptive coliform detection. For each dilution (100, 10−1, and 10−2), three tubes containing 10 mL of LTB with inverted Durham tubes were inoculated with 1 mL of the appropriate sample dilution. Tubes showing gas production after incubation at 35 ± 0.5 °C for 24–48 hours were considered presumptive positive. Confirmatory tests were then performed by transferring a loopful from positive LTB tubes into Brilliant Green Lactose Bile (BGLB) broth (Merck, Germany) and incubating at 35 ± 0.5 °C for an additional 24–48 hours. Tubes exhibiting gas formation were recorded as confirmed positive for coliforms, and the results were calculated using standard MPN tables.
Shelf life was determined based on the earliest occurrence of either (1) microbiological exceedance of regulatory limits, or (2) significant reduction in antioxidant activity compromising functional quality. According to the Ministry of Public Health, Thailand, the microbiological limits for pasteurized fruit beverages are: total bacteria count ≤ 4 log CFU/mL, yeast and mold count ≤ 2 log CFU/mL, and coliforms < 3 MPN/mL. For antioxidant activity, a 50% reduction from initial values was considered the threshold for significant quality loss, as this represents the point at which the functional claim of the beverage would be compromised.
2.6. Statistical analysis
The experimental design followed a completely randomized design (CRD) with three replicates per treatment. A two-factor analysis of variance (ANOVA) was employed to evaluate the effects of pasteurization (control, LTLT, and HTST) and packaging type (transparent glass vs. amber-colored glass) during storage. Data were analyzed using the Statistical Package for the Social Sciences (SPSS, Version 22.0; IBM Corp., Armonk, NY, USA). When significant differences were observed among treatments (p ≤ 0.05), mean comparisons were performed using Tukey's Honestly Significant Difference (HSD) test at a 95% confidence level. All results are presented as mean ± standard deviation (SD).3 Results and discussion
3.1. Effects of thermal processing and packaging on physicochemical properties of the ready-to-drink beverage from date palm extract during storage at 4 °C
During the 9 weeks of storage, the total acid content of the samples, particularly control samples, tended to increase (Table 1). The samples had an initial total acid level of 0.10 to 0.11% citric acid (p > 0.05). At the end of the storage period, the total acid level of control samples in transparent and amber-colored glass bottles was 0.38 and 0.39%, respectively. At the end of storage (9 weeks), the total acid level of pasteurized samples in transparent and amber-colored glass bottles ranged from 0.11 to 0.13%. Pasteurization had an impact on the product storage phase since it kills the bacteria that cause food degradation while also preserving it.9| Storage (week) | Treatments | |||||
|---|---|---|---|---|---|---|
| Transparent glass bottles | Amber-colored glass bottles | |||||
| Control | LTLT | HTST | Control | LTLT | HTST | |
| a High temperature-short time (HTST, temperature 72 °C, 20 s), low temperature-long time (LTLT, temperature 63 °C, 30 min) and “ns” indicates no significant (p > 0.05) difference in each row. Control: (date palm extract (70%): bael fruit juice (15%): jujube juice (15%)). Data are presented as mean ± standard deviation; different letters (a and b) refer to the significant differences (p ≤ 0.05) in each row. | ||||||
| 0ns | 0.11 ± 0.01 | 0.10 ± 0.01 | 0.10 ± 0.01 | 0.11 ± 0.01 | 0.10 ± 0.01 | 0.10 ± 0.01 |
| 1 | 0.11 ± 0.01a | 0.10 ± 0.01 | 0.12 ± 0.01a | 0.09 ± 0.01b | 0.10 ± 0.01 | 0.11 ± 0.01a |
| 2 | 0.18 ± 0.01a | 0.11 ± 0.01b | 0.11 ± 0.01b | 0.18 ± 0.01a | 0.11 ± 0.01b | 0.11 ± 0.01b |
| 3 | 0.27 ± 0.01a | 0.10 ± 0.01d | 0.13 ± 0.01c | 0.20 ± 0.01b | 0.10 ± 0.01d | 0.13 ± 0.01c |
| 4 | 0.34 ± 0.01a | 0.11 ± 0.01c | 0.12 ± 0.01c | 0.25 ± 0.01b | 0.11 ± 0.01c | 0.12 ± 0.01c |
| 5 | 0.32 ± 0.01a | 0.12 ± 0.01de | 0.14 ± 0.01c | 0.26 ± 0.01b | 0.11 ± 0.01e | 0.13 ± 0.01 cd |
| 6 | 0.39 ± 0.01a | 0.10 ± 0.01d | 0.13 ± 0.02c | 0.31 ± 0.01b | 0.11 ± 0.01 cd | 0.11 ± 0.01 cd |
| 7 | 0.36 ± 0.01a | 0.11 ± 0.01c | 0.11 ± 0.01c | 0.30 ± 0.01b | 0.10 ± 0.01c | 0.12 ± 0.01c |
| 8 | 0.37 ± 0.01a | 0.10 ± 0.01c | 0.14 ± 0.01b | 0.36 ± 0.01a | 0.08 ± 0.01d | 0.11 ± 0.01c |
| 9 | 0.38 ± 0.01a | 0.12 ± 0.03b | 0.13 ± 0.01b | 0.39 ± 0.01a | 0.11 ± 0.01b | 0.13 ± 0.01b |
Table 2 shows that the initial pH value ranged from 4.35 to 4.37, which was not significantly different (p > 0.05). Control samples in clear and amber-colored glass bottles had pH values of 3.81 and 3.82, respectively, which were lower than those of the other samples. Throughout the storage period, there were no significant differences in total soluble solids (p > 0.05). During storage, the total soluble solids in the samples tends to decrease slightly, which is consistent with the pH value. Table 3 shows that total soluble solids ranged from 6.9 to 7.2 °Brix. These findings suggest that total soluble solids are closely associated with total acid content and pH. The observed decrease in pH is attributed to the accumulation of acids resulting from the metabolic activity of lactic acid bacteria, which utilize sugars for growth and fermentation, consequently reducing the total soluble solids.16
| Storage (week) | Treatments | |||||
|---|---|---|---|---|---|---|
| Transparent glass bottles | Amber-colored glass bottles | |||||
| Control | LTLT | HTST | Control | LTLT | HTST | |
| a High temperature-short time (HTST, temperature 72 °C, 20 s), low temperature-long time (LTLT, temperature 63 °C, 30 min) and “ns” indicates no significant (p > 0.05) difference in each row. Control: (date palm extract (70%): bael fruit juice (15%): jujube juice (15%)). Data are presented as mean ± standard deviation; different letters (a–b) refer to the significant differences (p ≤ 0.05) in each row. | ||||||
| 0ns | 4.37 ± 0.01 | 4.35 ± 0.01 | 4.36 ± 0.01 | 4.37 ± 0.01 | 4.35 ± 0.01 | 4.36 ± 0.01 |
| 1 | 4.27 ± 0.02b | 4.35 ± 0.01a | 4.36 ± 0.02a | 4.26 ± 0.02b | 4.35 ± 0.01a | 4.36 ± 0.02a |
| 2 | 3.87 ± 0.02b | 4.38 ± 0.01a | 4.36 ± 0.01a | 3.96 ± 0.01b | 4.35 ± 0.01a | 4.36 ± 0.01a |
| 3 | 3.85 ± 0.01b | 4.32 ± 0.01a | 4.35 ± 0.01a | 3.86 ± 0.02b | 4.35 ± 0.02a | 4.35 ± 0.01a |
| 4 | 3.84 ± 0.01b | 4.32 ± 0.02a | 4.35 ± 0.02a | 3.86 ± 0.02b | 4.36 ± 0.02a | 4.35 ± 0.01a |
| 5 | 3.83 ± 0.01b | 4.32 ± 0.02a | 4.35 ± 0.01a | 3.84 ± 0.02b | 4.36 ± 0.04a | 4.35 ± 0.03a |
| 6 | 3.80 ± 0.01b | 4.32 ± 0.01a | 4.32 ± 0.04a | 3.84 ± 0.03b | 4.36 ± 0.04a | 4.35 ± 0.03a |
| 7 | 3.81 ± 0.02b | 4.30 ± 0.02a | 4.32 ± 0.01a | 3.83 ± 0.02b | 4.34 ± 0.03a | 4.35 ± 0.01a |
| 8 | 3.80 ± 0.01b | 4.30 ± 0.08a | 4.31 ± 0.03a | 3.83 ± 0.05b | 4.34 ± 0.02a | 4.34 ± 0.05a |
| 9 | 3.81 ± 0.03b | 4.30 ± 0.02a | 4.31 ± 0.01a | 3.82 ± 0.05b | 4.32 ± 0.03a | 4.34 ± 0.01a |
| Storage (week)ns | Treatments | |||||
|---|---|---|---|---|---|---|
| Transparent glass bottles | Amber-colored glass bottles | |||||
| Control | LTLT | HTST | Control | LTLT | HTST | |
| a High temperature-short time (HTST, temperature 72 °C, 20 s), low temperature-long time (LTLT, temperature 63 °C, 30 min) and “ns” indicates no significant (p > 0.05) difference in each row. Control: (date palm extract (70%): bael fruit juice (15%): jujube juice (15%)). | ||||||
| 0 | 7.2 ± 0.06 | 6.9 ± 0.06 | 6.9 ± 0.06 | 7.2 ± 0.06 | 6.9 ± 0.06 | 6.9 ± 0.06 |
| 1 | 7.1 ± 0.10 | 6.8 ± 0.06 | 6.9 ± 0.01 | 7.2 ± 0.06 | 6.9 ± 0.06 | 6.9 ± 0.01 |
| 2 | 7.1 ± 0.06 | 6.8 ± 0.06 | 6.8 ± 0.06 | 7.2 ± 0.12 | 6.9 ± 0.06 | 6.9 ± 0.10 |
| 3 | 7.0 ± 0.06 | 6.8 ± 0.06 | 6.8 ± 0.06 | 7.1 ± 0.10 | 6.9 ± 0.06 | 6.9 ± 0.10 |
| 4 | 7.0 ± 0.10 | 6.8 ± 0.06 | 6.8 ± 0.01 | 7.1 ± 0.06 | 6.9 ± 0.12 | 6.9 ± 0.06 |
| 5 | 6.9 ± 0.01 | 6.8 ± 0.06 | 6.8 ± 0.01 | 7.1 ± 0.10 | 6.9 ± 0.10 | 6.9 ± 0.01 |
| 6 | 6.9 ± 0.06 | 6.8 ± 0.06 | 6.8 ± 0.06 | 7.0 ± 0.12 | 6.8 ± 0.17 | 6.9 ± 0.10 |
| 7 | 6.7 ± 0.06 | 6.8 ± 0.10 | 6.8 ± 0.06 | 6.8 ± 0.38 | 6.8 ± 0.20 | 6.9 ± 0.12 |
| 8 | 6.6 ± 0.06 | 6.8 ± 0.01 | 6.8 ± 0.06 | 6.7 ± 0.15 | 6.8 ± 0.17 | 6.9 ± 0.10 |
| 9 | 6.6 ± 0.06 | 6.8 ± 0.06 | 6.8 ± 0.01 | 6.7 ± 0.15 | 6.8 ± 0.10 | 6.8 ± 0.17 |
In terms of color values, the L* and b* values showed decreasing trends during storage. The L* value was 52.14 to 58.61, while the b* value was 67.52 to 70.59 (Table 4). Carotenoids are phytochemicals that are sensitive to oxygen, heat, and other environmental factors. As a result of the reaction of polyphenol oxidase (PPO) or the browning process associated with this enzyme, the b* value that shows yellowness is lowered during storage, while the a* value that indicates redness is increased. As shown in our results, pasteurized samples (HTST) contained in amber-colored glass bottles had less color change, resulting in samples having less color changes than samples from the LTLT. The color change (ΔE*) in HTST samples contained in clear and amber-colored glass bottles ranged from 1.16 to 6.66 and 0.65 to 4.81, respectively. However, the color change (ΔE*) among LTLT samples in clear and amber-colored glass bottles ranged respectively from 2.90 to 8.57 and 0.62 to 5.71 during storage. Amber-colored glass bottles provide superior protection against light-induced degradation of ready-to-drink beverages, such as date palm extract, by effectively blocking ultraviolet (UV) and visible light wavelengths below 450 nm. This shielding prevents photooxidation, thereby preserving the color, antioxidant activity, and overall quality of the product.17
| Treatments | ||||||
|---|---|---|---|---|---|---|
| Transparent glass bottles | Amber-colored glass bottles | |||||
| Control | LTLT | HTST | Control | LTLT | HTST | |
| a High temperature-short time (HTST, temperature 72 °C, 20 s), low temperature-long time (LTLT, temperature 63 °C, 30 min) and “ns” indicates no significant (p > 0.05) difference in each row. Control: (date palm extract (70%): quince juice (15%): jujube juice (15%)). Data are presented as mean ± standard deviation. Different letters (a–e) mean the significant differences (p ≤ 0.05) in each row. | ||||||
| L* | ||||||
| 0 | 58.61 ± 1.51a | 52.14 ± 1.99c | 55.14 ± 2.99b | 58.61 ± 1.51a | 52.14 ± 1.99c | 55.14 ± 2.99b |
| 1 | 56.21 ± 2.01a | 51.21 ± 0.01b | 54.21 ± 3.01a | 57.71 ± 0.51a | 51.56 ± 1.57b | 54.87 ± 2.67a |
| 2 | 55.71 ± 1.51a | 50.92 ± 0.04d | 53.71 ± 0.51bcd | 57.21 ± 0.01a | 51.06 ± 1.07 cd | 54.67 ± 1.55abc |
| 3 | 55.21 ± 11.01b | 50.21 ± 0.01c | 53.65 ± 0.47b | 57.02 ± 0.18a | 50.56 ± 0.57c | 54.72 ± 0.01b |
| 4 | 54.71 ± 0.51a | 50.71 ± 0.51c | 52.71 ± 0.49bc | 57.36 ± 0.64a | 51.68 ± 2.46bc | 53.00 ± 0.11bc |
| 5 | 53.71 ± 0.49b | 49.93 ± 0.05d | 51.77 ± 0.55c | 56.16 ± 1.05a | 49.93 ± 0.06d | 53.90 ± 0.01b |
| 6 | 53.03 ± 0.18c | 48.88 ± 0.10e | 50.93 ± 0.39d | 55.93 ± 0.28a | 48.93 ± 0.06e | 54.55 ± 0.34b |
| 7 | 51.03 ± 0.18b | 46.84 ± 0.06c | 53.71 ± 1.49a | 54.93 ± 0.72a | 48.01 ± 0.02c | 52.65 ± 0.25a |
| 8 | 50.23 ± 0.02c | 45.51 ± 0.39e | 49.68 ± 0.36c | 53.88 ± 0.67a | 47.51 ± 0.48d | 51.70 ± 0.30b |
| 9 | 48.73 ± 0.52c | 44.77 ± 0.35e | 48.73 ± 0.09c | 52.88 ± 0.33a | 46.73 ± 0.29d | 50.58 ± 0.18b |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| a*ns | ||||||
| 0 | 28.66 ± 0.02 | 29.46 ± 0.01 | 29.70 ± 0.01 | 28.66 ± 0.02 | 29.46 ± 0.01 | 29.70 ± 0.01 |
| 1 | 29.14 ± 20.50 | 29.54 ± 0.01 | 29.74 ± 0.02 | 29.16 ± 0.52 | 29.62 ± 0.01 | 29.72 ± 0.01 |
| 2 | 29.16 ± 0.48 | 30.29 ± 0.15 | 29.86 ± 0.02 | 29.67 ± 0.03 | 29.67 ± 0.02 | 29.92 ± 0.05 |
| 3 | 29.22 ± 0.50 | 30.46 ± 0.02 | 29.97 ± 0.02 | 29.70 ± 0.01 | 29.68 ± 0.01 | 29.73 ± 0.04 |
| 4 | 29.32 ± 0.01 | 30.45 ± 0.01 | 30.27 ± 0.03 | 29.16 ± 0.52 | 29.56 ± 0.04 | 30.10 ± 0.01 |
| 5 | 29.52 ± 0.16 | 30.96 ± 0.52 | 30.33 ± 0.01 | 29.74 ± 0.02 | 29.71 ± 0.01 | 30.03 ± 0.02 |
| 6 | 29.65 ± 0.04 | 30.99 ± 0.49 | 29.78 ± 0.04 | 29.82 ± 0.01 | 29.72 ± 0.02 | 29.97 ± 0.02 |
| 7 | 30.15 ± 0.47 | 31.05 ± 0.44 | 30.15 ± 0.01 | 29.84 ± 0.01 | 29.79 ± 0.05 | 30.14 ± 0.01 |
| 8 | 30.25 ± 0.36 | 31.47 ± 0.05 | 29.88 ± 0.06 | 29.94 ± 0.01 | 29.89 ± 0.04 | 30.17 ± 0.02 |
| 9 | 30.75 ± 0.14 | 32.57 ± 0.95 | 30.06 ± 0.14 | 30.02 ± 0.01 | 29.96 ± 0.03 | 30.27 ± 0.28 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| b* | ||||||
| 0 | 70.59 ± 0.02a | 67.52 ± 1.98b | 69.98 ± 0.54a | 70.59 ± 0.02a | 67.52 ± 1.98b | 69.98 ± 0.54a |
| 1 | 70.56 ± 0.01a | 67.50 ± 0.01c | 69.46 ± 0.02b | 69.86 ± 0.08b | 67.44 ± 2.04c | 69.46 ± 0.52b |
| 2 | 70.52 ± 0.01a | 67.49 ± 0.01bc | 69.40 ± 0.05a | 69.84 ± 0.09a | 67.32 ± 2.01c | 69.26 ± 0.52a |
| 3 | 70.51 ± 0.01a | 67.29 ± 0.06c | 68.97 ± 0.47bc | 69.82 ± 0.06a | 67.30 ± 2.01c | 69.31 ± 0.47a |
| 4 | 70.46 ± 0.01a | 67.39 ± 0.01c | 68.75 ± 0.55b | 69.85 ± 0.09a | 67.46 ± 0.02c | 68.86 ± 0.02b |
| 5 | 70.33 ± 0.02a | 67.16 ± 0.03c | 68.74 ± 0.54b | 69.77 ± 0. 0a | 67.21 ± 2.00bc | 68.91 ± 0.03b |
| 6 | 70.27 ± 0.02a | 67.04 ± 0.01c | 68.66 ± 0.46b | 69.22 ± 0.52a | 67.11 ± 1.20bc | 69.19 ± 0.45a |
| 7 | 70.15 ± 0.04a | 66.97 ± 0.02b | 68.81 ± 0.61a | 69.13 ± 0.43a | 66.81 ± 0.90b | 68.82 ± 0.02a |
| 8 | 70.11 ± 0.02a | 66.86 ± 0.02c | 68.62 ± 0.42b | 69.08 ± 0.38a | 66.77 ± 0.86c | 68.77 ± 0.03b |
| 9 | 70.03 ± 0.02a | 66.29 ± 0.09c | 68.22 ± 0.20b | 69.05 ± 0.35a | 66.66 ± 0.75c | 68.67 ± 0.07b |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| ΔE | ||||||
| 0 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 1 | 2.48 ± 0.59 | 2.90 ± 0.63a | 1.16 ± 0.21c | 1.64 ± 0.42bc | 0.62 ± 0.40c | 0.65 ± 0.14c |
| 2 | 2.98 ± 0.09a | 3.06 ± 0.81a | 2.57 ± 1.50b | 2.21 ± 0.97b | 1.17 ± 0.86c | 1.64 ± 0.41c |
| 3 | 3.49 ± 0.40a | 3.46 ± 0.97a | 2.78 ± 1.72b | 2.40 ± 1.14b | 1.68 ± 1.34c | 3.06 ± 0.42a |
| 4 | 3.96 ± 0.98a | 3.52 ± 0. 9b | 3.78 ± 2.59a | 2.50 ± 1.00c | 2.07 ± 0.16c | 3.16 ± 2.15b |
| 5 | 5.01 ± 1.92a | 3.85 ± 0.72b | 4.00 ± 3.31b | 3.26 ± 1.92c | 2.35 ± 1.93d | 3.19 ± 1.32c |
| 6 | 5.69 ± 1.64a | 4.47 ± 1.00b | 4.41 ± 3.52b | 3.34 ± 1.64c | 3.48 ± 1.79c | 3.43 ± 0.59c |
| 7 | 7.74 ± 1.74a | 6.07 ± 1.39b | 4.72 ± 1.64c | 4.14 ± 0.85 cd | 4.48 ± 1.65c | 3.53 ± 2.45d |
| 8 | 8.54 ± 1.52a | 7.30 ± 1.26b | 5.64 ± 2.77c | 5.14 ± 0.87c | 5.02 ± 2.11c | 3.89 ± 3.07d |
| 9 | 10.12 ± 0.92a | 8.57 ± 1.40b | 6.66 ± 3.14c | 6.10 ± 1.81 cd | 5.71 ± 1.43d | 4.81 ± 3.15e |
3.2. Effects of thermal processing and packaging on bioactive compounds and antioxidant activityof the ready-to-drink beverage from date palm extract during storage at 4 °C
Throughout the storage period, the total amount of phenolic compounds (TPC) in all samples decreased, which could be due to oxidation and non-enzymatic reactions, which can be accelerated by factors like oxygen exposure, light, and prolonged storage. TPC was reduced by 49.87 percent over the same period from its starting value in the samples. This was in line with the findings in ref. 18 which reported the storage quality of non-thermally pasteurized and microfiltered pineapple juice at various temperatures (4, 27, and 37 °C). The results indicated that TPC of the juice significantly decreased as storage time and temperature increased (p ≤ 0.05), with the highest retention observed at 4 °C. This suggests that lower storage temperatures help preserve the phytochemical properties of the juice. Our findings also revealed that ready-to-drink date palm juice (HTST) packed in amber-colored glass bottles could maintain the highest level of TPC during storage. In line with our results ref. 19, demonstrated that blueberry extract preserved in glass bottles at different temperatures (−20, 6, 23 and 35 °C) for 60 days showed a mild decrease in the TPC value especially at −20 °C (Table 5).| Storage (week) | Treatments | |||||
|---|---|---|---|---|---|---|
| Transparent glass bottles | Amber-colored glass bottles | |||||
| Control | LTLT | HTST | Control | LTLT | HTST | |
| a TPC: total phenolic compound (mg GAE/100 mL = mg gallic acid equivalent/100 mL), TCC: total carotenoid content (mg β-CE/100 mL = mg β-carotene equivalent/100 mL), DPPH: 2,2-diphenly-1-picrylhydrazyl (mM trolox/100 mL) and FRAP: ferric reducing antioxidant power (mM trolox/100 mL). High temperature-short time (HTST, temperature 72 °C, 20 s), low temperature-long time (LTLT, temperature 63 °C, 30 min) and “ns” indicates no significant (p > 0.05) difference in each row. Control: (date palm extract (70%): bael fruit juice (15%): jujube juice (15%)). Data are presented as mean ± standard deviation; different letters (a–e) mean the significant differences (p ≤ 0.05) in each row. | ||||||
| TPC | ||||||
| 0 | 1174.67 ± 10.41a | 1151.33 ± 7.64b | 1169.67 ± 7.64a | 1174.67 ± 10.41a | 1151.33 ± 7.64b | 1169.67 ± 7.64a |
| 1 | 1066.33 ± 7.64cd | 1038.00 ± 18.03d | 1073.00 ± 13.23bc | 1103.00 ± 15.00a | 1124.67 ± 10.41a | 1126.33 ± 7.64a |
| 2 | 963.00 ± 5.00b | 946.33 ± 7.64b | 969.67 ± 10.41b | 1056.33 ± 15.28a | 1079.67 ± 12.58a | 1081.33 ± 10.41a |
| 3 | 874.67 ± 7.64b | 853.00 ± 5.00b | 891.33 ± 10.41b | 1006.33 ± 7.64a | 1024.67 ± 7.64a | 1033.00 ± 10.00a |
| 4 | 781.33 ± 7.64cd | 768.00 ± 13.23d | 804.67 ± 10.41c | 956.33 ± 7.64b | 983.00 ± 10.00a | 984.67 ± 7.64a |
| 5 | 688.00 ± 10.00bc | 668.00 ± 5.00c | 706.33 ± 7.64b | 861.33 ± 5.77a | 881.33 ± 12.58a | 883.00 ± 10.00a |
| 6 | 603.00 ± 5.00cd | 591.33 ± 7.64d | 618.00 ± 13.23c | 766.33 ± 7.64b | 793.00 ± 10.00a | 803.00 ± 5.00a |
| 7 | 561.33 ± 5.77cd | 556.33 ± 7.64d | 586.33 ± 10.41c | 671.33 ± 12.58b | 691.33 ± 10.41a | 701.33 ± 7.64a |
| 8 | 496.33 ± 7.64bc | 473.00 ± 13.23c | 516.33 ± 12.58b | 618.00 ± 13.23a | 628.00 ± 10.00a | 638.00 ± 13.23a |
| 9 | 381.33 ± 17.56c | 384.67 ± 12.58bc | 419.67 ± 12.58b | 556.33 ± 7.64a | 574.67 ± 15.28a | 586.33 ± 15.28a |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| TCC | ||||||
| 0 | 7.22 ± 0.07a | 7.11 ± 0.01b | 7.18 ± 0.01a | 7.22 ± 0.07a | 7.11 ± 0.01b | 7.18 ± 0.01a |
| 1 | 6.05 ± 0.02b | 6.17 ± 0.07b | 6.13 ± 0.02b | 6.96 ± 0.05a | 6.98 ± 0.17a | 7.06 ± 0.04a |
| 2 | 5.50 ± 0.01c | 5.59 ± 0.03c | 5.65 ± 0.06bc | 5.78 ± 0.01a | 5.82 ± 0.04a | 5.97 ± 0.09a |
| 3 | 4.52 ± 0.01e | 4.62 ± 0.02d | 4.72 ± 0.03c | 5.25 ± 0.02b | 5.29 ± 0.04b | 5.38 ± 0.06a |
| 4 | 3.57 ± 0.01d | 3.61 ± 0.02d | 3.64 ± 0.01d | 4.86 ± 0.03c | 4.94 ± 0.03b | 5.03 ± 0.05a |
| 5 | 2.78 ± 0.07c | 2.81 ± 0.05c | 2.88 ± 0.02c | 4.31 ± 0.05b | 4.41 ± 0.01a | 4.53 ± 0.05a |
| 6 | 2.58 ± 0.04c | 2.63 ± 0.05c | 2.67 ± 0.01c | 3.72 ± 0.01b | 3.79 ± 0.05a | 3.82 ± 0.02a |
| 7 | 1.40 ± 0.01d | 1.45 ± 0.02d | 1.57 ± 0.04c | 3.62 ± 0.01b | 3.69 ± 0.05 ab | 3.71 ± 0.03a |
| 8 | 0.90 ± 0.03c | 0.95 ± 0.02bc | 1.01 ± 0.05b | 3.55 ± 0.02a | 3.58 ± 0.01a | 3.60 ± 0.03a |
| 9 | 0.45 ± 0.02d | 0.50 ± 0.02 cd | 0.54 ± 0.01c | 3.16 ± 0.03b | 3.21 ± 0.05 ab | 3.25 ± 0.03a |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| DPPH | ||||||
| 0 | 1448.96 ± 9.55a | 1421.88 ± 6.25b | 1436.46 ± 9.55a | 1448.96 ± 9.55a | 1421.88 ± 6.25b | 1436.46 ± 9.55a |
| 1 | 1317.71 ± 26.02b | 1305.21 ± 15.73b | 1321.88 ± 16.54b | 1369.79 ± 13.01a | 1371.88 ± 12.50a | 1382.29 ± 15.73a |
| 2 | 1180.21 ± 15.73b | 1194.79 ± 13.01b | 1213.33 ± 31.81b | 1311.46 ± 26.02a | 1309.38 ± 12.50a | 1321.88 ± 12.50a |
| 3 | 1128.13 ± 22.53b | 1094.79 ± 26.02b | 1138.54 ± 15.73b | 1240.63 ± 12.50a | 1263.54 ± 15.73a | 1288.54 ± 9.55a |
| 4 | 1059.38 ± 28.64b | 1023.96 ± 21.95b | 1076.04 ± 19.09b | 1184.38 ± 12.50a | 1196.88 ± 12.50a | 1207.29 ± 26.02a |
| 5 | 1001.04 ± 23.66b | 934.38 ± 12.50c | 1023.96 ± 15.73b | 1121.88 ± 12.50a | 1134.38 ± 12.50a | 1144.79 ± 26.02a |
| 6 | 873.96 ± 26.02b | 869.79 ± 15.73b | 886.46 ± 9.55b | 1059.38 ± 12.50a | 1071.88 ± 12.50a | 1082.29 ± 26.02a |
| 7 | 757.29 ± 19.09b | 748.96 ± 20.09b | 761.46 ± 13.01b | 936.46 ± 20.09a | 944.79 ± 19.09a | 961.46 ± 13.01a |
| 8 | 694.79 ± 19.09b | 684.38 ± 16.54b | 711.46 ± 7.22b | 809.38 ± 16.54a | 819.79 ± 19.09a | 836.46 ± 7.22a |
| 9 | 638.54 ± 13.01b | 640.63 ± 25.00b | 646.88 ± 6.25b | 744.79 ± 19.09a | 751.04 ± 36.62a | 763.54 ± 13.01a |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| FRAP | ||||||
| 0 | 1430.26 ± 14.43a | 1419.55 ± 4.12b | 1427.88 ± 10.31a | 1430.26 ± 14.43a | 1419.55 ± 4.12b | 1427.88 ± 10.31a |
| 1 | 1323.12 ± 17.62c | 1345.74 ± 4.12bc | 1369.55 ± 8.99 ab | 1381.45 ± 5.46a | 1385.02 ± 8.99a | 1393.36 ± 12.88a |
| 2 | 1257.64 ± 7.14c | 1287.40 ± 12.54b | 1294.55 ± 8.99b | 1283.83 ± 8.99b | 1323.12 ± 12.54a | 1325.50 ± 3.57a |
| 3 | 1150.50 ± 7.14d | 1183.83 ± 5.46c | 1196.93 ± 3.57b | 1186.21 ± 7.14c | 1212.40 ± 7.43b | 1248.12 ± 14.87a |
| 4 | 943.36 ± 3.57d | 962.40 ± 14.43 cd | 986.21 ± 6.19bc | 981.45 ± 10.91bc | 1000.50 ± 10.71b | 1024.31 ± 5.46a |
| 5 | 857.64 ± 6.19d | 860.02 ± 10.31d | 912.40 ± 5.46c | 933.83 ± 17.62bc | 954.07 ± 10.71b | 967.17 ± 14.87a |
| 6 | 819.55 ± 5.46c | 825.50 ± 9.45c | 862.40 ± 17.98b | 855.26 ± 5.46b | 861.21 ± 9.45b | 896.93 ± 9.45a |
| 7 | 723.12 ± 10.91d | 729.07 ± 6.19d | 758.83 ± 14.43c | 819.55 ± 5.46b | 825.50 ± 9.45b | 857.64 ± 7.14a |
| 8 | 677.88 ± 7.43c | 688.60 ± 8.99c | 699.31 ± 5.46c | 796.93 ± 7.14b | 812.40 ± 5.46a | 827.88 ± 11.48a |
| 9 | 588.60 ± 10.91c | 594.55 ± 14.43c | 613.60 ± 8.99c | 754.07 ± 3.57b | 761.21 ± 14.29a | 786.21 ± 3.57a |
The degradation of phenolic compounds during storage can be attributed to multiple mechanisms, including enzymatic oxidation by polyphenol oxidase (PPO), non-enzymatic oxidation, and polymerization reactions. PPO catalyzes the oxidation of phenolic compounds to quinones, which subsequently polymerize to form brown pigments, leading to both color changes and reduced antioxidant capacity.20 The higher retention of TPC in HTST-treated samples compared to LTLT-treated samples can be explained by the rapid inactivation of PPO during HTST processing, as the short exposure time at high temperature effectively denatures enzymes while minimizing thermal degradation of heat-sensitive phenolic compounds.21
Carotenoids are naturally occurring pigments found in various fruits and vegetables, imparting red, orange, yellow, and green hues. These compounds are not only responsible for the vibrant colors of many plant-based foods but also offer significant health benefits. Among them, β-carotene is the most prevalent provitamin A carotenoid, which the human body can convert into vitamin A, essential for vision, immune function, and skin health.22 It was found that the total β-carotene content (TCC) values of the samples tended in the same direction as TPC did during storage. However, pasteurized (HTST) samples-packaged in amber-colored glass bottles contained a higher TCC value (3.25 mg β-CE/100 mL) at the end of the storage (Table 5). The average reduction of TCC was approximately 54.74% from the initial value. The study in ref. 23 investigated the effects of refrigerated storage on the stability of bioactive compounds in orange juice. The researchers observed that after 40 days of storage at 4 °C, TCC of pasteurized orange juice decreased by 16.90%. This finding underscores the impact of storage conditions on the retention of carotenoids in fruit juices, highlighting the need for optimized preservation methods to maintain their nutritional quality.23 In the context of ready-to-drink beverages derived from date palm extract, TCC plays a crucial role in determining the nutritional value and overall quality of the product.
Carotenoid degradation occurs primarily through photo-oxidation and auto-oxidation mechanisms. Carotenoids are highly susceptible to isomerization and oxidation when exposed to light, particularly UV radiation, which generates singlet oxygen and free radicals that attack the conjugated double-bond system.22 Amber-colored glass bottles provide protection by filtering light wavelengths below 450 nm, including UV and blue light, which are most damaging to carotenoids. This explains the significantly higher TCC retention (3.25 mg β-CE/100 mL) in amber glass-packaged samples compared to transparent glass-packaged samples (0.54 mg β-CE/100 mL) at week 9. The reduction pattern of bioactive compounds observed in our study is consistent with findings from other fruit beverage studies ref. 23 reported a 16.9% decrease in total carotenoid content of pasteurized orange juice after 40 days of storage at 4 °C, while we observed a 54.7% reduction over 63 days (9 weeks), which is comparable when normalized to time (approximately 0.42% per day vs. 0.87% per day). The higher degradation rate in our product may be attributed to the presence of multiple fruit sources with different carotenoid profiles and potentially different stability characteristics.
Ref. 18 reported that pineapple juice stored at 4 °C retained significantly higher TPC compared to storage at 27 °C or 37 °C, emphasizing the critical role of refrigeration in preserving bioactive compounds. Our results reinforce these findings by demonstrating that packaging with light protection provides an additional preservation benefit beyond temperature control alone.
It was found that the antioxidant activity by DPPH and FRAP assays of all samples tended to decrease throughout the cold storage period. The initial DPPH and FRAP values of the samples ranged respectively from 1421.88 to 1448.96 mM trolox/100 mL and 1419.55 to 1430.26 mM trolox/100 mL and reached 638.54 and 588.60 for DPPH and FRAP, respectively. The DPPH and FRAP decrease during the storage were 46.85% and 44.94% from the initial antioxidant activity, respectively (Table 5). The highest DPPH and FRAP values were found in pasteurization (HTST) samples maintained in amber-colored glass bottles, with values of 763.54 and 786.21 mM trolox/100 mL, respectively (Table 5).
Ref. 24 reported that thermal processing of mango juice led to an increase in antioxidant activity compared to untreated control samples. This enhancement is likely associated with the inactivation of polyphenol oxidase (PPO), an enzyme responsible for catalyzing the oxidation of polyphenolic compounds. By inhibiting PPO activity, thermal treatment reduces the degradation of phenolic compounds, thereby preserving or even enhancing the antioxidant capacity of the juice. Similar mechanisms may explain the observations in the present study, where controlled heat processing contributed to the retention of bioactive compounds and the improvement of antioxidant properties in the ready-to-drink beverage from date palm juice ref. 25 also discovered that the type of packaging could impact the number of antioxidants and their effects during preservation.
3.3. Effects of thermal processing and packaging on microbiological quality of the ready-to-drink beverage from date palm extract during storage at 4 °C
Thermal processing is a widely employed method in food preservation that utilizes heat to destroy harmful microorganisms and inactivate enzymes responsible for food spoilage. This technique extends the shelf life of food products, ensuring safety and quality during storage and distribution.26Packaging plays a crucial role in maintaining the quality and extending the shelf life of food products. The choice of packaging material affects factors such as oxygen permeability, moisture resistance, and protection from light and contaminants. Glass bottles are particularly effective in preserving food quality due to their excellent gas and moisture barrier properties, as well as their inertness to chemical and biological interactions.8
Incorporating both thermal processing and appropriate packaging materials can significantly enhance food preservation, benefiting both consumers and businesses by ensuring food safety and reducing waste.
The most prevalent and crucial aspect in food products is the degradation process induced by microbes, as it directly impacts the shelf life of food and is an indicator of consumer safety. As a result, the number of microorganisms in food products should not be excessive. Food deterioration induced by microorganisms is caused by the growth and increase in the quantity of microbes, which causes food to change in various ways, including putrid odors and color changes. Therefore, microbial spoilage is often found in fresh foods such as vegetables, fruits, fresh milk and meat, and spoilage is caused by three main types of microorganisms: bacteria, yeast and mold.27–29
The total microbial count of control samples in both amber-colored and clear glass bottles was found to be higher than that of pasteurized (LTLT and HTST) samples. Until 5 weeks after storage, all pasteurized (LTLT and HTST) samples revealed no signs of bacterial contamination (Table 6). At the end of storage, the total microbial content in the control group grew from 1.39 to 7.06 log CFU/mL. According to a statement from the Ministry of Public Health, total microbial content in foods must not exceed 4 log CFU/mL, which was the case for our pasteurization samples.
| Storage (week) | Treatments | |||||
|---|---|---|---|---|---|---|
| Transparent glass bottles | Amber-colored glass bottles | |||||
| Control | LTLT | HTST | Control | LTLT | HTST | |
| a High temperature-short time (HTST, temperature 72 °C, 20 s) and low temperature-long time (LTLT, temperature 63 °C, 30 min). Control: (date palm extract (70%): bael fruit juice (15%): jujube juice (15%)). Data are presented as mean ± standard deviation. ND: not detected. | ||||||
| Total microbial count | ||||||
| 0 | 1.39 ± 0.12 | ND | ND | 1.39 ± 0.12 | ND | ND |
| 1 | 2.39 ± 0.12 | ND | ND | 2.09 ± 0.12 | ND | ND |
| 2 | 3.39 ± 0.12 | ND | ND | 3.11 ± 0.10 | ND | ND |
| 3 | 4.02 ± 0.09 | ND | ND | 3.57 ± 0.04 | ND | ND |
| 4 | 4.52 ± 0.74 | ND | ND | 4.02 ± 0.09 | ND | ND |
| 5 | 5.27 ± 0.02 | ND | ND | 4.29 ± 0.08 | ND | ND |
| 6 | 5.41 ± 0.02 | 1.10 ± 0.14 | 1.00 ± 0.01 | 5.06 ± 0.08 | 1.02 ± 0.03 | ND |
| 7 | 6.04 ± 0.06 | 2.10 ± 0.14 | 1.81 ± 0.05 | 5.35 ± 0.07 | 1.93 ± 0.04 | 1.65 ± 0.07 |
| 8 | 6.22 ± 0.06 | 3.21 ± 0.13 | 2.80 ± 0.14 | 5.98 ± 0.03 | 2.74 ± 0.06 | 2.24 ± 0.34 |
| 9 | 7.06 ± 0.08 | 4.22 ± 0.06 | 3.22 ± 0.06 | 6.18 ± 0.10 | 3.98 ± 0.03 | 2.98 ± 0.03 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Yeast and mold count | ||||||
| 0 | 2.40 ± 0.13 | ND | ND | 2.40 ± 0.13 | ND | ND |
| 1 | 2.95 ± 0.07 | ND | ND | 2.69 ± 0.13 | ND | ND |
| 2 | 3.28 ± 0.03 | ND | ND | 2.99 ± 0.12 | ND | ND |
| 3 | 3.80 ± 0.14 | ND | ND | 3.28 ± 0.03 | ND | ND |
| 4 | 4.16 ± 0.06 | ND | ND | 3.71 ± 0.15 | ND | ND |
| 5 | 4.24 ± 0.09 | ND | ND | 4.17 ± 0.12 | ND | ND |
| 6 | 4.63 ± 0.21 | 1.00 ± 0.01 | ND | 4.31 ± 0.08 | ND | ND |
| 7 | 5.04 ± 0.06 | 1.85 ± 0.21 | 1.50 ± 0.28 | 4.80 ± 0.14 | 1.15 ± 0.21 | 1.00 ± 0.01 |
| 8 | 5.22 ± 0.06 | 2.45 ± 0.21 | 2.24 ± 0.34 | 5.02 ± 0.03 | 2.19 ± 0.16 | 2.09 ± 0.12 |
| 9 | 6.04 ± 0.06 | 3.35 ± 0.07 | 3.04 ± 0.06 | 5.16 ± 0.02 | 2.80 ± 0.14 | 2.57 ± 0.12 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Coliform (MPN/mL) | ||||||
| 0 | <3 | <3 | <3 | <3 | <3 | <3 |
| 1 | <3 | <3 | <3 | <3 | <3 | <3 |
| 2 | <3 | <3 | <3 | <3 | <3 | <3 |
| 3 | <3 | <3 | <3 | <3 | <3 | <3 |
| 4 | <3 | <3 | <3 | <3 | <3 | <3 |
| 5 | <3 | <3 | <3 | <3 | <3 | <3 |
| 6 | <3 | <3 | <3 | <3 | <3 | <3 |
| 7 | <3 | <3 | <3 | <3 | <3 | <3 |
| 8 | <3 | <3 | <3 | <3 | <3 | <3 |
| 9 | <3 | <3 | <3 | <3 | <3 | <3 |
Yeast and mold content in control samples stored in amber and transparent glass bottles increased from 2.40 to 5.16 and 6.04 log CFU/mL, respectively, during storage. Pasteurized (LTLT) samples packed in transparent glass bottles started showing yeast and mold growth in week 6 (1.00 log CFU/mL), while pasteurized (LTLT and HTST) samples packed in amber glass bottles started growing yeast and mold in week 7 (1.15 log CFU/mL) (Table 6). The observation that amber-colored glass bottles delayed microbial growth compared to transparent glass bottles can be explained by the light-filtering properties of amber glass. Amber glass effectively blocks ultraviolet (UV) and visible light wavelengths below 450 nm, which are known to promote microbial growth through several mechanisms. First, light exposure can increase the temperature of the product through radiative heating, creating more favorable conditions for microbial proliferation. Second, photo-oxidation reactions generate reactive oxygen species that can stress and potentially weaken microbial cells, but paradoxically, some microorganisms may adapt and become more resilient.17 Third, light-induced degradation of antimicrobial compounds naturally present in the beverage (such as phenolic compounds) may reduce the product's intrinsic antimicrobial activity over time. The delayed onset of yeast and mold growth in amber glass-packaged samples (week 7) compared to transparent glass-packaged samples (week 6) suggests that light protection helps maintain the beverage's natural antimicrobial barriers for a longer period. This finding aligns with ref. 30, which reported that light-protective packaging significantly reduced fungal spoilage in fresh produce.
Pasteurization is an effective method for reducing microbial contamination in fruit juices. For instance, the Ministry of Public Health stipulates that yeast and mold counts in fruit juices should not exceed 2 log CFU/mL. In line with this, a study in ref. 31 demonstrated that pasteurization of Passiflora setacea pulp led to a significant reduction in yeast and mold counts, achieving levels below the recommended threshold. Similarly ref. 32, reported that pasteurized pomegranate juice stored at both 5 °C and 25 °C exhibited lower microbial growth compared to unpasteurized controls, highlighting the efficacy of pasteurization in enhancing the microbiological safety of fruit juices during storage. These findings underscore the importance of pasteurization in ensuring the microbiological quality of fruit juices, aligning with both regulatory standards and scientific evidence.
During storage, coliforms were not detected in any of the samples (Table 6), indicating that the production processes were conducted under hygienic and appropriate conditions.33 This finding highlights the effectiveness of good manufacturing practices in ensuring microbiological safety. Moreover, extending the shelf life of food products requires careful consideration of packaging selection, as packaging is a critical factor in controlling the growth of bacteria, yeast, and mold during storage. Proper packaging can create barriers against microbial contamination, oxygen, and moisture, thereby maintaining product quality and safety over time. Compared to pomegranate juice studies,32 which showed microbial stability for 4–5 weeks at 5 °C, our combination of HTST pasteurization and amber glass packaging extended microbial stability to 7 weeks, representing a 40–75% improvement.
Based on yeast and mold counts and measurements of antioxidant activity, the ready-to-drink beverage prepared from date palm extract and subjected to HTST pasteurization, when stored in amber-colored glass bottles, exhibited the longest shelf life of 7 weeks at 4 °C. Applying these criteria, yeast and mold counts in HTST-pasteurized samples in amber glass remained below 2 log CFU/mL until week 7 (1.00 log CFU/mL at week 7), exceeding the limit only at week 8, while antioxidant activity (DPPH) decreased by 46.85% from the initial values by week 9, approaching but not exceeding the 50% threshold until that time. Therefore, week 7 represents the conservative shelf-life endpoint based on the most limiting factor (yeast and mold count). This condition outperformed other treatment combinations, with control samples exceeding microbiological limits by week 3, indicating that the combination of HTST pasteurization and amber glass packaging extended shelf life by approximately four weeks. These results highlight the effectiveness of integrating optimized thermal treatment and protective packaging to preserve both microbiological quality and bioactive properties, maximizing shelf life and functional quality in fruit-based beverages.
While the present study demonstrates the efficacy of thermal pasteurization and protective packaging, it is important to acknowledge that alternative non-thermal preservation technologies are emerging as potentially more energy-efficient options for beverage preservation. UV treatment, particularly in the germicidal wavelength range of 254–280 nm, has shown promise for inactivating microorganisms in fruit juices without the thermal degradation associated with pasteurization.34 Studies on UV-treated apple juice and orange juice have demonstrated significant microbial reduction while maintaining higher levels of heat-sensitive bioactive compounds compared to thermal pasteurization.35 However, UV treatment efficacy can be limited by juice turbidity and color, which reduce light penetration—a challenge that may be relevant for the dark-colored date palm beverage in our study.
Other non-thermal technologies including high-pressure processing (HPP), pulsed electric fields (PEF), and cold plasma have also been investigated for fruit juice preservation. These technologies offer the advantage of reduced energy consumption and better retention of fresh-like qualities, but require higher capital investment and may have regulatory approval limitations in some regions.36
Aseptic packaging with improved surface disinfection represents another sustainable approach, combining sterile filling with advanced packaging materials that incorporate oxygen scavengers or antimicrobial compounds.37 Future research could explore combinations of mild thermal treatment with these emerging technologies to further optimize the sustainability-performance balance for date palm-based beverages.
4 Conclusions
This study demonstrated that both the pasteurization method and packaging type significantly influenced the physicochemical properties, antioxidant activity, and microbial stability of ready-to-drink beverages formulated from date palm extract, bael juice, and jujube juice during storage at 4 °C. HTST pasteurization combined with amber-colored glass bottles effectively preserved bioactive compounds, including total phenolic compound (586.33 mg GAE/100 mL), total carotenoid content (3.25 mg β-CE/100 mL), and antioxidant activities (DPPH: 763.54 mM trolox/100 mL; FRAP: 786.21 mM trolox/100 mL), while maintaining microbial safety throughout storage. The use of durable, reusable, amber-colored glass bottles, coupled with efficient HTST pasteurization, not only extended shelf life to seven weeks but also supports sustainability by reducing reliance on single-use packaging and minimizing food waste. Overall, these results indicate that the application of optimized thermal processing alongside sustainable packaging practices may contribute to maintaining product quality while supporting environmentally responsible production of ready-to-drink fruit-based beverages.Author contributions
Pitchaya Tuntiteeraboon: investigation, formal analysis, data curation and writing – original draft. Sochannet Chheng: data curation and writing – original draft. Saeid Jafari: data curation and writing – original draft. Isaya Kijpatanasilp: data curation and writing – original draft. Kitipong Assatarakul: conceptualization, data curation, funding acquisition, project administration, supervision, writing – original draft and writing – review & editing.Conflicts of interest
The authors declare no conflicts of interest, financial or personal, that could have influenced the research presented in this paper.Data availability
The data in the current study are available from the corresponding author upon reasonable request.Acknowledgements
This research project was financially supported by the 90th Anniversary of Chulalongkorn University Scholarship under the Ratchadapisek Somphot Endowment Fund, the Graduate School, Chulalongkorn University and the Department of Food Technology, Faculty of Science, Chulalongkorn University. Gratitude is extended to the Second Century Fund (C2F) at Chulalongkorn University for providing postdoctoral fellowships to Dr Saeid Jafari and Dr Sochannet Chheng.References
- S. Maqsood, O. Adiamo, M. Ahmad and P. Mudgil, Food Chem., 2020, 308, 125522 CrossRef CAS PubMed.
- L. Boateng, W. Quarpong, A. Ohemeng, M. Asante and M. Steiner-Asiedu, Food Sci. Nutr., 2019, 7, 302–311 CrossRef CAS PubMed.
- J. Chen, X. Liu, Z. Li, A. Qi, P. Yao, Z. Zhou, T. T. Dong and K. W. Tsim, J. Evidence-Based Complementary Altern. Med., 2017, 2017, 3019568 CrossRef PubMed.
- S. V. Singh, R. Singh, A. Singh, S. Thangalakshmi, B. P. Kaur, M. G. Kamble, A. Tarafdar and A. Upadhyay, J. Food Process. Preserv., 2022, 46, e16315 CAS.
- T. Fadiji, M. Rashvand, M. O. Daramola and S. A. Iwarere, Processes, 2023, 11, 2 Search PubMed.
- D. Mudgal, E. Pagone and K. Salonitis, J. Cleaner Prod., 2024, 481, 144133 CrossRef.
- L. O. Onyeka, A. A. Adesiyun, K. H. Keddy, A. Manqele, E. Madoroba and P. N. Thompson, Food Control, 2021, 123, 107746 CrossRef CAS.
- I. Franco and E. Falqué, Glass packaging, in Reference Module in Food Science, Elsevier, 2016 Search PubMed.
- B. A. O. Sanchez, S. M. C. Celestino, M. B. de Abreu Gloria, I. C. Celestino, M. I. O. Lozada, S. D. A. Júnior, E. R. de Alencar and L. de Oliveira, Food Chem.:X, 2020, 6, 100084 CAS.
- AOAC, Official Methods of Analysis of AOAC International, 17th edn, Association of Official Analytical Chemists, Gaithersburg, MD, USA, 2000 Search PubMed.
- S. Chheng, M. Fikry, S. Jafari, S. K. Mehta, D. K. Mishra and K. Assatarakul, ES Food Agrofor., 2025, 19, 1434 CAS.
- A. Biswas, J. Sahoo and M. Chatli, Food Sci. Technol., 2011, 44, 1809–1813 CAS.
- S. Baliyan, R. Mukherjee, A. Priyadarshini, A. Vibhuti, A. Gupta, R. P. Pandey and C.-M. Chang, Molecules, 2022, 27, 1326 CrossRef CAS PubMed.
- S. Chheng, M. Fikry, S. Jafari, D. K. Mishra and K. Assatarakul, J. Stored Prod. Res., 2025, 112, 102660 CrossRef CAS.
- P. Bird, B. Bastin, N. Klass, E. Crowley, J. Agin, D. Goins, H. Bakken, C. Lingle and A. Schumacher, J. AOAC Int., 2020, 103, 513–522 CrossRef PubMed.
- J. Yeom, D. J. Yim, S. Ma and Y.-H. Lim, Microorganisms, 2021, 10, 48 CrossRef PubMed.
- M. H. Hasnul Hadi, P. J. Ker, V. A. Thiviyanathan, S. G. H. Tang, Y. S. Leong, H. J. Lee, M. A. Hannan, M. Z. Jamaludin and M. A. Mahdi, Sensors, 2021, 21, 6866 CrossRef CAS PubMed.
- A. Laorko, S. Tongchitpakdee and W. Youravong, J. Food Eng., 2013, 116, 554–561 CrossRef CAS.
- A. Srivastava, C. C. Akoh, W. Yi, J. Fischer and G. Krewer, J. Agric. Food Chem., 2007, 55, 2705–2713 CrossRef CAS PubMed.
- C. G. Vazquez-Lima, M. Quintana-Quirino, A. L. Bravo, R. López-Simeon and H. Nájera, Food Sci. Nutr., 2025, 13, e71175 CrossRef CAS PubMed.
- A. Amanina, S. Rosnah, M. Noranizan and S. Alifdalino, Food Res., 2019, 3, 362–372 CrossRef PubMed.
- P. Crupi, M. F. Faienza, M. Y. Naeem, F. Corbo, M. L. Clodoveo and M. Muraglia, Antioxidants, 2023, 12, 1069 CrossRef CAS PubMed.
- L. Plaza, C. Sánchez-Moreno, P. Elez-Martínez, B. De Ancos, O. Martín-Belloso and M. P. Cano, Eur. Food Res. Technol., 2006, 223, 487–493 CrossRef CAS.
- A. Amanina, S. Rosnah, M. Noranizan and S. J. F. R. Alifdalino, Food Res., 2019, 3(4), 362–372 CrossRef PubMed.
- M. V. Geraldi, C. B. B. Cazarin, F. L. Dias-Audibert, G. A. Pereira, G. G. Carvalho, D. Y. Kabuki, R. R. Catharino, G. M. Pastore, J. H. Behrens and M. J. L. Cristianini, LWT, 2021, 139, 110548 CrossRef CAS.
- N. Maurya, A. Thapa and D. Nino, J. Food Nutr. Res., 2025, 8, 1–7 Search PubMed.
- S. Jafari, N. Rungroj, R. W. Worobo and K. Assatarakul, Int. J. Food Microbiol., 2021, 358, 109404 CrossRef CAS PubMed.
- Y. Mu, J. Huang, R. Zhou, F. Mao, Q. Pan, S. Chen, Z. Lu, L. Du and F. Xie, Food Res. Int., 2022, 155, 111062 CrossRef CAS PubMed.
- O. Alegbeleye, O. A. Odeyemi, M. Strateva and D. Stratev, Appl. Food Res., 2022, 2, 100122 CrossRef CAS.
- A. López-Gómez, M. Ros-Chumillas, A. Navarro-Martínez, M. Barón, L. Navarro-Segura, A. Taboada-Rodríguez, F. Marín-Iniesta and G. B. Martínez-Hernández, Foods, 2021, 10, 1196 CrossRef PubMed.
- B. A. O. Sanchez, S. M. C. Celestino, M. B. de Abreu Gloria, I. C. Celestino, M. I. O. Lozada, S. D. A. Júnior, E. R. de Alencar and L. d. L. de Oliveira, Food Chem.:X, 2020, 6, 100084 CAS.
- S. Vegara, N. Martí, P. Mena, D. Saura and M. Valero, LWT--Food Sci. Technol., 2013, 54, 592–596 CrossRef CAS.
- A. López-Gómez, M. Ros-Chumillas, A. Navarro-Martínez, M. Barón, L. Navarro-Segura, A. Taboada-Rodríguez, F. Marín-Iniesta and G. B. J. F. Martínez-Hernández, Foods, 2021, 10, 1196 CrossRef PubMed.
- T. Koutchma, in Validation of Food Preservation Processes Based on Novel Technologies, Elsevier, 2022, pp. 113–167 Search PubMed.
- S. Jafari, N. Rungroj, R. W. Worobo and K. Assatarakul, Int. J. Food Microbiol., 2021, 358, 109404 CrossRef CAS PubMed.
- B. T. Abelbaeviсh, M. Zamzagul, M. B. Zhaksylykovna, A. L. Zhanabayevna, T. Sholpan, Y. Yerkin and B. A. Sarsenbayevna, Caspian J. Environ. Sci., 2024, 22, 513–520 Search PubMed.
- S. Kamarudin, M. Rayung, F. Abu, S. Ahmad, F. Fadil, A. Karim, M. Norizan, N. Sarifuddin, M. Desa and M. Basri, Polymers, 2022, 14(1), 174 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |