Open Access Article
Open Access ArticleThe state-of-the-art on chitosan: historical perspectives and non-thermal modification technologies for sustainable packaging films
Susmita Bora†
and
Tabli Ghosh†
 *
*
Department of Food Engineering and Technology, School of Engineering, Tezpur university, Assam 784028, India. E-mail: tablighosh1@gmail.com
First published on 4th May 2026
Abstract
Chitosan has garnered significant attention in the field of polymer science over the decades due to its associated characteristics, such as biodegradability, antimicrobial properties, and film-forming abilities. With increasing concerns, extensive research has been conducted to enhance the functional properties of chitosan using various modification techniques. Recently, the modification of chitosan-based materials using non-thermal technologies has drawn attention due to their ability to improve mechanical strength, barrier properties, and bioactivity without compromising environmental sustainability. This review discusses the historical discovery and development of chitin and chitosan and further emphasizes their growing significance in biomaterials science. A key focus of this study is to apply non-thermal modification technologies, such as cold plasma, irradiation, high pressure processing, ultrasonication, pulsed electric fields, and electrospinning, for the modification of chitosan-based materials for their application in active and intelligent packaging systems and shelf-life extensions, thereby aligning with the principles of green chemistry. Non-thermal modification technologies significantly enhance the structural and functional properties of chitosan films, making them viable alternatives to conventional plastic packaging. The mechanical properties, antimicrobial efficacy, oxygen barrier properties, and thermal stability of chitosan are among its key benefits, and the proposed material has been successfully used as a food packaging material for preserving fresh produce and increasing the shelf life of several types of food products. This approach not only supports the transition towards a circular economy but also contributes to a significant reduction in plastic packaging.
Sustainability spotlightChitosan, a deacetylated derivative of chitin, has evolved from a waste-derived biopolymer into a promising material for sustainable packaging. Initially extracted from crustacean shells, it is now widely explored in films due to its biodegradability and antimicrobial properties. This review outlines the historical development of chitosan in materials science and highlights the recent advancements in non-thermal modification techniques, such as ultrasound, cold plasma, pulsed electric fields, and high-pressure processing. These methods improve the solubility, film-forming ability, and barrier properties without affecting its natural functionality. This study also integrates sustainability metrics, including life cycle assessment and carbon footprint reduction, linking them with structural and performance characteristics for smart, active, and future-ready food packaging systems. |
Introduction
In recent years, fossil-derived polymers have posed significant environmental challenges because they are non-biodegradable in nature, which in turn increases the carbon footprint and toxic pollutants.1 Additionally, various chemicals are used for the production of synthetic polymers, and some of them are considered carcinogenic and harmful to the environment. Therefore, there is a growing shift towards the use of natural polymers, which are biodegradable and eco-friendly.2 According to the Institute of Bioplastics and Biocomposites (2016), global bioplastic production grew from 1.6 million tons in 2013 to 2.0 million tons in 2015, indicating a steady increase in the adoption of biodegradable and biobased plastics. In 2019, the global market for biodegradable plastics was valued at about USD 4.65 billion, and it was projected to grow by the end of 2025 at a compound annual growth rate of 17.04%, reaching a market value of up to 12.06 million. Biodegradable plastics such as PLA (polylactic acid) and PHA (polyhydroxyalkanoates) account for over 55.5% of the total global bioplastic production.3 Bioplastic production is dominated by Asia, which contributes to 63.1%, followed by North America (13.5%), Europe (13.0%), and South America (10.0%). While designing packaging, one of the key attributes of sustainable packaging is biodegradability, ensuring that materials break down naturally without causing any environmental hazards. Biodegradability is not only a functional requirement but also a crucial environmental necessity. Biodegradable polymers break down 60% faster than conventional plastics, which can take over 1000 years to degrade in the environment. Biodegradable plastics can fully decompose within 180 days.4 Non-biodegradability contributes to landfills, plastic waste, and pollution, causing an ecological imbalance. As a result, the use of natural polymers or biopolymers (substances derived inherently from living matter) has gained more attention due to their associated advantages and increasing consumer awareness regarding the use of synthetic polymers.2Among the available biopolymers, chitin is considered the second most abundant biopolymer after cellulose, as first described by Henri Braconnot in 1811. This naturally occurring biopolymer is found in ordered macrofibrils within the exoskeletons of mollusks and crustaceans and in the cell walls of fungi; it is reported that 70% of chitin comes from marine species.5 However, ‘chitin’ was formally named in 1823 by Odier, who isolated it from the cuticles of insects. During the 19th and 20th centuries, researchers recognised its structural similarity to cellulose. Chitosan, a deacetylated form of chitin, was first described by Charles Rouget in 1859. Despite these early discoveries, the application of chitin and chitosan remained limited until the mid-20th century. By the late 20th century, chitin and chitosan had gained much popularity due to their non-toxicity, biodegradability, and biocompatibility.
Chitosan, a naturally derived polysaccharide, has gained significant attention across various industries due to its non-toxicity, biocompatibility, excellent film-forming properties, and cost-effectiveness.6 Chitosan is the N-deacetylation product of chitin and is a heteropolysaccharide composed of β-1 → 4-linked glucosamine and N-acetylglucosamine units, and it is the second most abundant biopolymer after cellulose.7 Chitosan is widely used in the medicine, food, cosmetics, biomedical and packaging industries due to its biodegradability, antimicrobial, and other bioactive functions.6 However, the use of chitosan is limited due to its low solubility, high crystallinity, and limited functional groups for any chemical reaction.8 Its limited solubility in neutral and alkaline solutions poses a significant challenge for broader industrial applications. The rigid crystalline structure of native chitosan prevents it from dissolving in aqueous solvents at pH > 6.5, making functionalization and processing crucial. Moreover, chitosan is insoluble in most organic solvents, restricting its application for various purposes. Therefore, the modification of chitosan has become essential in enhancing its functional properties to expand its applications.9 The primary methods for chitosan modification include chemical modification, where chemical modification introduces functional groups to improve solubility, reactivity, and bioactivity; however, these methods have several drawbacks. Conventional chemical modifications require harsh reagents that generate toxic by-products, and harsh chemical modification alters chitosan's biocompatibility, degrades its molecular structure, and limits its applications in various sensitive fields, such as pharmaceuticals and food packaging. Additionally, it often requires high temperature and prolonged processing times, making the process energy intensive and costly, leading to low yields.10 Hence, to overcome these limitations, eco-friendly and sustainable modifications of biopolymers using non-thermal technologies, such as cold plasma, high pressure processing, irradiation, pulse electric field, ultrasonication, and microwaves, have gained significant attention.11 Social consciousness as a catalyst has been increasing dramatically as the public concern related to plastic pollution, climate change, and an imbalance in the ecosystems has heightened its demand for environmentally friendly alternative solutions and a shift towards greener technologies.12 These modifications of the natural biopolymer chitosan using non-thermal technologies in contrast to conventional modifications allow for controlled modification without compromising its structural integrity, enhancing functional properties without any degradation or harmful by-products. Additionally, these techniques do not rely on heat, but the use of mechanical-, electrical-, and radiation-based energy to alter and modify their structure and functionality is being explored.13 Non-thermal technology often allows for targeted modifications, e.g. improved solubility, antimicrobial activity, and biocompatibility without any structural breakdown. Chitosan modification using non-thermal technology has been explored for a wide range of applications, such as plasma treatment, which introduces functional groups onto chitosan's surface, thereby improving its properties, such as hydrophilicity, adhesion and biocompatibility.14 In ultrasonication-based non-thermal technology, ultrasonication time and heat treatment influence the structure and properties of chitosan/graphene oxide tailored films. Hence, sonication improved the UV and visible light barrier properties of the chitosan/graphene oxide films, making them effective for packaging applications.15 By adopting these methods, industries can reduce their ecological footprint and provide a greener, eco-friendly alternative to conventional modification methods, thereby supporting sustainability, safety, and environmental responsibility.
Based on the above discussion, this review critically discusses recent advances in the non-thermal modification of chitosan, highlighting its impact on the structural, physicochemical, and functional properties relevant to sustainable packaging films. Furthermore, key challenges, knowledge gaps, and future research directions are discussed to support the development of next-generation chitosan-based packaging materials.
History of chitin and chitosan
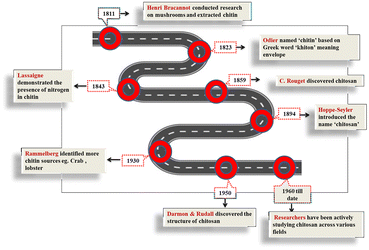
Polymers are large molecules composed of repeating structural units called monomers and play a crucial role in various scientific and technological fields. Based on their origin, polymers can be classified as synthetic and natural.16 Among natural polymers, the discovery of chitin over two centuries ago marked the beginning of a scientific journey into one of nature's most abundant and versatile biopolymers. Trailing only in prevalence after ‘cellulose’, chitin is considered the second most abundant polysaccharide. Over the past 220 years, the study of chitin has progressively evolved from scientific curiosity towards being one of the fundamental elements of biomaterials science, as illustrated in Fig. 1, which traces the historical development of chitin and chitosan from their discovery to the current period.In 1811, Henri Braconnot, a French chemist, isolated a material from fungi using water, alcohol, and dilute alkali, and named the insoluble residue “fongine” or “fungine”. This insoluble residue was later identified as chitin, making Braconnot's discovery the first recorded identification of a polysaccharide from fungi. Braconnot's discovery made a remarkable footprint, highlighting his pioneering contribution towards biochemistry, after the discovery of the most abundant polymer, “cellulose,” about thirty years earlier.17 Further, August Odier, a Swiss physician, in 1823, during his experiment with cockchafer betel, found a distinct substance, hornlike material, and named it “Khiton, Chitine,” which means ‘envelop’ in Greek. Another scientist, Opperman, in 1832, envisaged that chitin was extracted from insects; substances similar to chitin can also be found in the structure of insects.18 Therefore, another conflict arose between scientists regarding chitin produced by arthropods and cellulose produced by plants. In 1859, French physiologist Charles Rouget discovered that heating chitin made it soluble in organic solvents, leading to the discovery of its derivative, chitosan. As reported by Crini, the era between 1894 and 1930 led to another significant controversy about the presence of chitin and cellulose in the cell walls of fungi and their solubility in organic solvents.17 In 1930, Rammelberg identified more sources of chitin apart from insects and fungi and highlighted that it can be extracted from the exoskeletons of marine arthropods such as crabs, shrimps, and lobsters. Later that year, he made a remarkable contribution by identifying chitin as a polysaccharide of glucosamine. Using polarised radiation spectra, in 1950, Darmon and Rudall identified the structures of chitin, chitosan, and cellulose.19 From 1930 to 1950, chitin and chitosan gained much attention in terms of their extraction and isolation processes, where the first book was published in 1951, which included research conducted by various scientists on chitosan. Therefore, in the late 1960s, different polymeric forms of chitin were identified.17 Since 1960, numerous researchers have been actively studying chitosan across various fields. This ongoing research has explored its applications in areas such as biomedicine, agriculture, food technology, and water treatment. The versatility of chitosan, due to its biocompatibility, biodegradability, and non-toxicity, has made it a subject of extensive scientific interest and innovation.20
With the advancement of technology and urbanization, concerns have been increasing in materials science regarding the use of sustainable processing methods for the extraction of biopolymers, such as chitosan.21 The discovery of non-thermal technologies for the extraction of chitosan modification emerged as a response to the need for gentle and more sustainable processing methods, eventually maintaining the biopolymer's delicate structure and bioactivity in comparison to other thermal technologies that can degrade the structure of the biopolymer along with its integral mechanical properties. This era began in the late 20th and early 21st centuries, as materials science and environmental concerns drove the search for alternatives to traditional thermal technologies.
With due course of time, researchers have analysed the use of non-thermal technologies such as cold plasma, irradiation, high-pressure processing (HPP), and ozone treatment, which can effectively alter chitosan's surface properties, molecular weight, functional groups, copolymerization, hydrophilicity, and others, avoiding the potential risk of its degradation associated with heat and other parameters, which makes this biopolymer invaluable in the area of polymer science with its diverse application.22
Chitin and chitosan: structural and chemical properties
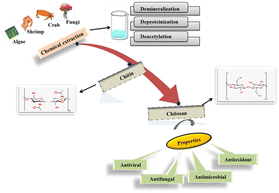
Biopolymers are the units of monomers derived from biological sources. Their molecular structure can be linear, branched or cross-linked, thereby influencing their properties and applications. Biopolymers are broadly classified into three types depending on their monomeric units: (i) polynucleotides: these biopolymers are composed of repeating monomeric units, e.g., DNA and RNA. (ii) polypeptides: these consist of short polymers of amino acids. (iii) polysaccharides: these consist of linearly bonded polymeric sugar (saccharide) units linked together, e.g., cellulose, chitosan, lignin, hemicellulose and glycogen. Polysaccharide-based polymer membranes have gained significant attention in food packaging due to their non-toxicity, biodegradability and excellent film-forming abilities. These characteristics make them a potent candidate for use as a sustainable alternative to plastic-based packaging, thereby focusing on sustainable development goals.23The last decades can be considered the golden era for the most abundant biopolymer chitin (CT), which is present in nature and its N-deacetylated form chitosan (CS), due to its extensive and diverse applications in various sectors.24 The second most abundant biopolymer present after cellulose, viz. ‘Chitin’, can be defined as the abundant mucopolysaccharide present in nature, which is hard and inelastic and is made up of nitrogenous compounds.5 Further, unlike any other polysaccharide, such as glycogen or starch, being considered a ‘storage polysaccharide’, chitin is considered a ‘structural polysaccharide’.25 In this regard, an overview of chitosan sources, extraction, and modification techniques is illustrated in Table 1. Compared to conventional chemical routes, emerging non-thermal and green modification techniques, such as microwaves, ultrasonication, cold plasma, and pulsed electric fields, offer reduced processing times, lower chemical consumption, and improved functional efficiency. These methods also minimize environmental burden and preserve the intrinsic structure of chitosan, thereby enhancing its bioactivity and performance. However, challenges related to process scalability, equipment costs, and uniformity of treatment still limit large-scale industrial adoption. Addressing these limitations is essential for the broader implementation of sustainable chitosan processing technologies.
| Sl. No. | Parameters | Specification and details | References |
|---|---|---|---|
| 1 | Sources of chitosan | • Exoskeletons of crustaceans: lobsters, shrimps, krill, and crayfish. | 31 |
| • Mollusks: octopus, clams, oysters, squids, and snails. | |||
| • Algae: diatoms, brown algae, and green algae. | |||
| • Insects: houseflies, silkworms, cockroaches, spiders, beetles.etc. | |||
| 2 | Chitin content (%) | • Crustaceans: 20–30% | 31 |
| • Shrimp cuticles: 30–40% | |||
| • Crab cuticles: 15–30% | |||
| • Fungal cell wall: 2–44% | |||
| 3 | Extraction techniques (chemical extraction) | • Demineralization: using acids, such as HCl and HNO3; temperature: 25 °C–100 °C; and time: 30 min–12 h. | 31,32 |
| • Deproteinization: using alkalis, such as NaOH, KOH, Na2SO3, and Na2CO3; temperature: 25 °C–100 °C; and time: 30 min–72 h. | |||
| • Deacetylation: using alkalis, such as NaOH/KOH (30–65%); temperature: 80 °C–150 °C; and time: 1–8 h. | |||
| 4 | Structural properties | • Degree of deacetylation (more than 50%) | 33 |
| • Crystallinity | |||
| • Molecular weight | |||
| • Solubility | |||
| 5 | Modification techniques | • Chemical modification: Acylation, esterification, etherification, quaternization, carboxylation and graft copolymerization. | 34 |
| • Non-thermal modification: cold plasma, irradiation, pulsed electric fields, supercritical fluid extraction, microwave, high pressure processing, electrospinning and ultrasonication. | |||
| 6 | Applications | • Drug delivery, food industry, agriculture, biomedical and pharmaceuticals, packaging, textile industry, paper industry, and wastewater treatment. | 31 |
This natural polymer is the principal component in the exoskeleton of arthropods, as the arthropods use chitin in their cuticle to form exoskeletons and microstructural fibrils.26 It is also found in crustaceans, such as crabs, lobsters, shrimps, mollusks, and insects, as well as in the cell walls of fungi. Chitin is composed of N-acetylglucosamine units linked by β-(1 → 4) glycosidic bonds due to its higher degree of crystallinity and strong intermolecular hydrogen bonding; chitin is insoluble in water and in most other organic solvents.27 Chitin usually contains 6–7% of nitrogen compounds. There are three polymeric forms of chitin: α, β and γ; in α-chitin, the chains are arranged in an anti-parallel fashion; because of this anti-parallel nature of α-chitin, it enhances the intermolecular hydrogen bonding, making the structure more compact and stable; because of this strong hydrogen bonding, chitin has higher crystallinity, which eventually decreases its solubility in water and most of the organic solvents, thereby making it durable and rigid.28 The β-form is generally found in mollusks and is aligned parallelly; however, the γ-nature of chitin is arranged so that two strands align parallel to each other and one is arranged anti-parallel. Chitin, derived from sources like crustaceans, fungi, and algae, as presented in Fig. 2, undergoes a chemical extraction process involving demineralization, deproteinization, and deacetylation to produce ‘chitosan’. When chitin is deacetylated, its N-acetyl groups are partially removed, which converts N-acetyl-glucosamine units into D-glucosamine units; this derivative is called chitosan.29 This structural transformation enables its solubility and bioactivity to be applied in diverse applications. This biopolymer is a valuable candidate due to its antimicrobial, antifungal, and antioxidant properties, contributing to its widespread use in various fields. The degree of deacetylation (DD) in chitosan is represented as the proportion of D-glucosamine units relative to N-acetyl-D-glucosamine units in the polymer. A higher degree of deacetylation of chitin indicates greater removal of the acetyl group, increasing the number of free amino (–NH2) groups in the molecule. In simple terms, chitosan can be defined as chitin with a degree of deacetylation (DD) value of 50% or higher, thereby making it soluble in acidic solutions. When chitosan is dissolved in an acidic solution, its free amino (–NH2) groups become protonated (–NH3+) in an acidic environment, signifying its unique characteristic as the only naturally occurring cationic (positively charged) polymer. Due to this cationic nature, chitosan gives it distinct and functional properties.30 However, chitosan is more reactive than chitin because of the presence of functional groups such as primary hydroxyl (–OH) at carbon-6 (C6), secondary hydroxyl (–OH) at carbon-3 (C3), and amino (–NH2) group at carbon 2 (C2), where the presence of these groups makes chitosan highly diverse and versatile for chemical modifications. Basically, the free amino site (-NH2) in chitosan is considered highly reactive, allowing the molecule to be functionalized via acetylation, grafting, crosslinking and carboxylation. Thus, functionalizing chitosan tailors its properties for specialized industrial uses, making it a valuable material across multiple domains.24 Precisely, because of the flexible nature of chitosan, its mechanical, physical, and chemical properties can be modified accordingly, thereby enhancing its biodegradability, biocompatibility, and strength. This adaptability has made chitosan suitable for myriad applications in diverse fields, including agriculture, biomedical engineering, pharmaceuticals, drug delivery, food packaging, and wastewater treatment.5
Modification of chitosan using non-thermal technologies
Technological advancements with minimal effects, maintaining quality and safety, have drawn a lot of interest.35 As stated, the modification of chitosan using non-thermal technologies, such as plasma treatment, irradiation, high-pressure processing, pulse electric field, and ultrasonication, alters its functional and physiochemical properties without using excessive heat, maintaining structural integrity, making it a versatile component and expanding the application of chitosan in various industries by aligning with all the principles of green chemistry.Decontamination is a crucial step for food products, such as nuts, due to microbial resistance, particularly from spore forming bacteria. Akhavan-Mahdavi et al. 2023 in their study found that when plasma treatment was given to chitosan-based coated pistachios (0.5 and 1.5% w/v), the treatment extended the shelf life and minimized aflatoxin and peroxide levels. Further, the plasma treatment of 120 s along with chitosan coating of concentration 1.5% preserved the hardness and color of pistachios during storage. This combined effect of edible coating along with plasma treatment can be a cutting-edge technology for the preservation of fresh products, ensuring microbial safety along with high-quality standards for commercial application and exports.45 A two-step modification was applied to zein films, involving an initial enhancement with chitosan, followed by cold plasma technology to improve their functional properties. It was also observed that by the combination of treatment, the elongation at break increased from 1.13% to 4.13%, and after the application of 60 s of plasma treatment, the composite film showed higher tensile strength and a better water vapour barrier (Fig. 3). ATR-FTIR (Attenuated Total Reflectance-Fourier Transform Infrared) analysis revealed a molecular interaction between zein and chitosan after plasma treatment. As a non-thermal, chemical-free process, it maintains the film's biodegradability while boosting its suitability for food packaging.46 With the nexus of consumer preferences, replacing synthetic materials with sustainable alternatives has reached its peak. Cold plasma treatment was applied to a chitosan-based packaging film along with cinnamon oil. The study revealed that cold plasma treatment enhances the surface roughness, wettability, and antimicrobial activity of chitosan-cinnamon oil films by generating reactive species that damage bacterial cell walls and facilitate controlled oil release. Additionally, plasma-induced crosslinking improves the oxygen barrier properties. Thus, cold plasma-treated chitosan–cinnamon oil films show strong potential for active food packaging, supporting the circular economy.47 Pankaj et al. 2017 in their study observed the effect of cold plasma treatment on antimicrobial chitosan films containing thymol as an active ingredient. Plasma treatment significantly increased surface roughness due to etching effects by improving film adhesion. In terms of thermal properties, there were no significant changes, no evidence of thermal decomposition, vaporization or volatile loss after plasma exposure and the thymol diffusion rate increased after the treatment. Plasma-treated chitosan–thymol films offer better antimicrobial efficiency and control the release of active ingredients.48 Atomic force microscopy (AFM), surface free energy, and Fourier transform infrared spectroscopy (FTIR) analyses revealed that cold plasma treatment (optimal at 75 W) increased the surface roughness and introduced polar functional groups, thereby improving chitosan coating adhesion onto PLA films. The plasma-treated films exhibited enhanced water resistance and improved mechanical strength, both of which were directly correlated with the increased chitosan deposition level at this power. Furthermore, the surface-modified films exhibited strong antimicrobial activity against Staphylococcus aureus. In contrast, higher discharge powers (≥100 W) adversely affected film performance due to polymer degradation.49 A drastic increase in water repellence was observed with the contact angle reaching 121° when ultrafast plasma modification was applied to chitosan/nanocellulose biocomposite films while maintaining the mechanical strength and barrier properties of the films. Further, X-ray photoelectron spectroscopy (XPS) showed irreversible fluorine-related bonds inducing surface fluorination. This modification remained stable for over 31 days under storage conditions. It was observed that the contact angle, atomic composition, and mechanical properties remained unchanged over time. This cold plasma treatment makes it a promising, eco-friendly solution for water-resistant food packaging applications.50 This non-thermal technology strengthens adhesion, modifies surface chemistry, and extends the shelf life without compromising its biodegradability. Cold plasma-treated films offer a sustainable alternative to conventional packaging, aligning with global efforts towards green and smart packaging solutions.
 | ||
| Fig. 3 Development of the composite film using cold plasma for packaging applications. Reproduced from ref. 46 with permission from Elsevier, Chen et al., Industrial Crops and Products, 2019, 129, 318–326. Copyright, 2019. | ||
Biopolymer zein and chitosan were tailored with synthetic polymer PVA and pre-treated using a pulsed electric field at different pulse energies. This study underscores the unique potential of the pulsed electric field as a sustainable and pre-treatment method for the modification of biodegradable films, thereby improving their mechanical and thermal properties without the use of any type of chemical cross-linkers. It was evidenced that the pulsed electric field strengths (0.9–3.5 kV cm−1), pulse frequencies (50–300 Hz) and specific energies (80–650 kJ kg−1) positively influenced the tensile properties and water stability of the tailored films by enhancing their stability and durability; pulsed electric field also influenced the film morphology from amorphous to a semi-crystalline structure. By reducing the reliance on chemical cross linkers and additives, the pulsed electric field supports the development of environmentally friendly materials.72 Molecular weight (MW) significantly affects the film forming ability of chitosan owing to its application. Chitosan with a higher molecular weight often limits its application due to its viscosity and film forming abilities. Traditional degradation methods, such as enzyme hydrolysis and acid hydrolysis, tend to be time consuming and environmentally harmful. Pulsed electric fields have emerged as a non-thermal technique for chitosan degradation, offering an environmentally friendly alternative. A pulsed electric field was applied up to a strength of 25 kV cm−1; with the increasing use of a pulsed electric field, the molecular weight of chitosan was lowered, making it suitable for applications requiring a low viscosity solution for drug delivery or film formation. Lower molecular weight chitosan produced by a pulsed electric field shows enhanced bioactivity, including antioxidant and antimicrobial properties.73 Pulsed electric field provides a sustainable and controlled condition for the reconstruction of biopolymers, where studies dictate that it is a powerful tool for modification of protein-polysaccharide (zein/chitosan) interactions, enhancing colloidal dispersion properties and promoting bioconjugation. Its ability to modify structural integrity and molecular interactions makes it a promising alternative to conventional processing techniques, fostering advancements in biomaterials science and sustainable packaging industries. Treating zein-chitosan/polyvinyl alcohol film with pulse electric fields induced conformational changes in the structure of the biopolymeric films. It was observed that a low intensity pulse electric field enhanced the stability of the edible film against enzyme degradation, and no toxicity was found. Pulse electric field-modified edible film can be a potent technology for value addition in edible food packaging.74 Reinforcing microcrystalline cellulose with starch/chitosan and applying a moderate electric field to the biopolymeric film improved the physiological properties of the film. The composite polysaccharide-based film showed excellent thermal properties, mechanical strength and barrier properties. Surface morphology can be assessed by scanning electron microscopy (SEM) and X-ray diffraction (XRD), which revealed that different concentrations of microcrystalline cellulose along with a moderate electric field are responsible for changes in the properties of the films. It was observed that the application of a moderate electric field was sufficiently efficient in decreasing water vapour permeability in chitosan-based films. Therefore, this reinforced biopolymeric film along with a moderate electric field can be a potent application for changing and modifying the properties of the film.75 The use of pulse electric fields in the application for the modification of biopolymers presents a promising technique for enhancing material properties, such as conductivity, mechanical strength, and surface modifications. This method enables precise control over the polymer structure and facilitates applications in diverse fields.
An edible coating of chitosan/tea polyphenol along with high pressure processing as a synergistic effect has been applied to shrimp to increase its antioxidant and antimicrobial properties. This combined treatment of 400 MPa of high pressure along with chitosan/tea polyphenol coating inhibited microbial growth, delayed spoilage, and showed better oxidative stability and the shrimp retained hardness and colour stability during the entire storage period. This method can be an effective way to extend the shelf life of shrimp while maintaining its quality.79 The study performed by Martillanes et al. 2021 focuses on evaluating the effectiveness of high-pressure processing combined with chitosan-based films in reducing L. monocytogenes contamination on sliced dry-cured Liberian ham. The combination of high-pressure processing with nisin and oryzanol-enriched chitosan film resulted in the greatest reduction of L. monocytogenes, achieving a 6 log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cfu per g decrease. This hurdle approach is effective, making it a promising strategy for improving food safety in dry-cured meats.80 High hydrostatic pressure and nano-TiO2 can also be applied in chitosan/polyvinyl alcohol-based films to enhance surface properties. It was observed that high hydrostatic pressure enhanced the molecular interactions between chitosan and alcohol films. This combined treatment improved the mechanical and barrier properties of the film, leading to better durability and stability, with higher antimicrobial properties. The applied hydrostatic pressure in the range of 200–400 MPa maintains the integrity of the film, which suggests resistance to degradation or dissolution and reduces the migration of TiO2 nanoparticles from the films, making them a potent candidate for sustainable and functional food packaging.81 The use of high pressure processing and antioxidant-enriched gelatin-chitosan edible films with oregano and rosemary extract improved the chemical stability (preventing oxidation) and microbial safety of cold-smoked sardines (Sardina pilchardus), making them long lasting. When the sardine muscles were coated with edible films, the phenol content and antioxidant power increased. This effect was even stronger when high pressure was applied likely because the pressure helped transfer antioxidant compounds from the film to the fish. This combined effect was more effective at reducing microbial counts, particularly sulphide-reducing bacteria; no luminescent bacteria or Enterobacteriaceae were detected in any samples after the treatment.82 This can be an effective way to enhance the shelf life of fresh produce at a low cost without any energy consumption. High pressure processing is a promising sustainable technology that enhances the functional, preservation, and modification of biopolymers while aligning with environmental goals. Its ability to reduce energy efficiency makes it an eco-friendly solution for various industries, such as food, medicine and biomedical applications.
cfu per g decrease. This hurdle approach is effective, making it a promising strategy for improving food safety in dry-cured meats.80 High hydrostatic pressure and nano-TiO2 can also be applied in chitosan/polyvinyl alcohol-based films to enhance surface properties. It was observed that high hydrostatic pressure enhanced the molecular interactions between chitosan and alcohol films. This combined treatment improved the mechanical and barrier properties of the film, leading to better durability and stability, with higher antimicrobial properties. The applied hydrostatic pressure in the range of 200–400 MPa maintains the integrity of the film, which suggests resistance to degradation or dissolution and reduces the migration of TiO2 nanoparticles from the films, making them a potent candidate for sustainable and functional food packaging.81 The use of high pressure processing and antioxidant-enriched gelatin-chitosan edible films with oregano and rosemary extract improved the chemical stability (preventing oxidation) and microbial safety of cold-smoked sardines (Sardina pilchardus), making them long lasting. When the sardine muscles were coated with edible films, the phenol content and antioxidant power increased. This effect was even stronger when high pressure was applied likely because the pressure helped transfer antioxidant compounds from the film to the fish. This combined effect was more effective at reducing microbial counts, particularly sulphide-reducing bacteria; no luminescent bacteria or Enterobacteriaceae were detected in any samples after the treatment.82 This can be an effective way to enhance the shelf life of fresh produce at a low cost without any energy consumption. High pressure processing is a promising sustainable technology that enhances the functional, preservation, and modification of biopolymers while aligning with environmental goals. Its ability to reduce energy efficiency makes it an eco-friendly solution for various industries, such as food, medicine and biomedical applications.
| Sl. No. | Material | Non-thermal technologies | Outcomes | References |
|---|---|---|---|---|
| 1. | • Chitosan/wampee seed essential oil | • Cold plasma treatment | • Provides synergistic effects due to the combined use of the edible coating of chitosan along with cold plasma treatment on golden pompano fillets. | 93 |
| • The treated fillets showed the lowest bacterial count and peroxide values and inhibited lipid lysis with enhanced antimicrobial activity, thereby extending the shelf life throughout the storage period. | ||||
| 2. | • Chitosan/cellulose nanocrystals | • Cold plasma treatment (low temperature generated) | • Improved packaging film property through surface modification, such as improved water repellence, significantly increasing the contact angle while boosting mechanical strength and reducing water vapour transmission. | 50 |
| • The treated packaging film showed the highest stability during the 31 days storage period. | ||||
| 3. | • Chitosan | • Pulsed electric fields/ozone treatment | • Combined effects resulted in a lower molecular weight of chitosan (Mw < 2500 Da) and lower crystallinity. | 94 |
| 4. | • Chitosan/grape | • Gamma-irradiation | • Used as an edible coating on chicken breast meat. The combined effects of irradiation (2.5 kGy) and chitosan/GSE coating enhanced the antimicrobial activity. | 61 |
| • All the coated samples showed lower TBA (thiobarbituric acid) and pH values, and the sensory quality was retained during the entire 21 days storage period. | ||||
| 5. | • Chitosan/essential oil/silver nanoparticles | • Gamma-irradiation | • Provides a synergistic effect due to the combined use of irradiation and edible coating on fresh strawberries. | 55 |
| • The composite film exhibited strong antimicrobial activity against pathogenic bacteria. | ||||
| • Weight loss, firmness and decay (%) were retained during the 12 days storage period. | ||||
| 6. | • Quinoa protein/chitosan/transglutaminase (TG) | • Ultrasonication | • The thermal stability of the composite film increased after ultrasonication treatment. | 95 |
| • The treatment enhanced the structural and mechanical properties of the composite films. | ||||
| 7 | • Chitosan/Sargassum pallidum polysaccharides | • Ultrasonication | • Ultrasonication enhanced the transparency, elongation, and tensile strength of the film along with the antioxidant activity, which has also increased. | 96 |
| • The treated films extended the shelf life of the strawberry, inhibiting deterioration at room temperature (25 °C) during the 7 days storage period. | ||||
| 8 | • Chitosan/Mandarin essential oil | • High pressure processing (HPP)/pulsed light processing | • The combination of edible coating along with HPP on green beans resulted in a significant reduction of L. innocua, thereby increasing the shelf life. | 97 |
| • The treatment also retained the firmness of green beans. | ||||
| 9 | • Chitosan/starch/microcrystalline cellulose | • Pulsed electric fields | • The treated composite film presented good thermal and mechanical properties, generating more rigid films. | 75 |
| 10 | • Chitosan/polyvinyl alcohol/essential oils | • Electrospinning | • Antioxidant activity showed the highest value for the composite film. | 98 |
| • The bilayer lamination of the composite film increased the thermal resistance with an increase in antimicrobial activity. | ||||
| 11 | • Chitosan | • High hydrostatic pressure (HHP) | • The shelf life of rainbow trout fillets increased due to the combined effect of chitosan-based coatings and HHP. | 99 |
| • The combined treatment also resulted in a protective effect both chemically and microbiologically. | ||||
| 12 | • Chitosan/cinnamon oil | • Cold plasma treatment | • The strength and the antioxidant property of the films increase after the treatment. | 47 |
| • Cold plasma-induced composite film showed higher antimicrobial activity along with improved oxygen barrier properties. |
Further, electrospinning is a versatile fabrication method that is used to create nanofibrous materials from biopolymers. It works on the principle of applying high voltage to a polymer solution and causing the material to form ultrafine fibres. The resultant electrospun fibres possess unique properties, such as high surface area and porosity with enhanced mechanical properties, making them useful in a wide range of applications.87 Fat dissolving oral films (FDOFs) were developed using chitosan and pullulan through electrospinning technology. These edible films are precisely designed to dissolve quickly in the mouth, making drug delivery more efficient. Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), and thermal analysis revealed a reduction in crystallinity due to electrospinning, which made the film more amorphous through dissolution. With the addition of chitosan, the thermal stability also increased; the water solubility test showed that the composite film could dissolve within 60 s in water. This composite-modified film using electrospinning can be a promising solution and value-added edible coating.88 PVA/chitosan blend films were prepared using electrospinning under specific conditions at 21 kV and a 15 cm distance. The study reveals that the films have tiny fibre-like structures, which show profound antibacterial properties. These fibres have a large surface area, making them more effective against E. coli. Additionally, the special surface texture of electrospun fibres hinders the cell wall of bacteria by disrupting their adhesion to the biopolymeric film, thereby decreasing the growth of the bacteria and projecting its antibacterial activity.89 Overall, the combination of electrospinning and chitosan-based films/edible coating can be used as a potent candidate for an antimicrobial packaging solution for perishable food products.
Additionally, the allotrope of oxygen is ozone (O3) with a higher oxidizing power. Ozone is a strong oxidizing agent that reacts with organic molecules by introducing functional groups, such as hydroxyl, carbonyl and carboxyl groups. Ozone treatment is considered safe for use in food processing, as approved by the Food and Drug Administration (FDA). Ozone treatment alters the surface chemistry of biopolymer nanofibers or films by introducing oxygen-containing functional groups. This enhances the hydrophilicity, wettability, mechanical and barrier properties of the film.90 Additionally, PHA is a class of biodegradable biopolymers produced by microorganisms, and they possess excellent biodegradability and biocompatibility but generally lack strong antimicrobial properties; to enhance their antimicrobial properties, they are tailored with other antimicrobial agents, such as chitosan and chitooligosaccharides, using ozone treatment. This combined treatment enhances the antibacterial and biodegradable properties. Researchers claim that the antibacterial effects are particularly strong against Gram-negative bacteria due to their negatively charged cell walls, which attract the positively charged chitosan molecules, and ozone treatment introduces functional groups onto the composite film, thereby improving reactivity.91 In this study, lignocellulose films were blended with 2% chitosan to develop edible films. Short ozone treatment enhanced the strength and hydrophobicity, while prolonged treatment of 120 min improved the film uniformity, swelling and antibacterial effects. Chitosan incorporation further strengthens the film and increases moisture absorption, making it particularly effective for fruit preservation and antimicrobial food wraps. This ozone-treated composite food coating wrap can be a potent source for extending the shelf life and minimizing post-harvest losses.92 Across different non-thermal technologies, such as microwave processing, electrospinning, ozone treatment, irradiation, ultrasonication, and cold plasma, the improvement in chitosan-based films can be largely attributed to molecular-level interactions. These techniques modify the surface chemistry and microstructure of chitosan by introducing or activating functional groups, thereby promoting hydrogen bonding, electrostatic interactions, and physical entanglement between chitosan chains and incorporated additives or reinforcing agents. These interactions enhance interfacial adhesion, uniform dispersion, and controlled release behaviour, ultimately leading to improved mechanical strength, barrier performance, and functional properties of the modified films.44,56
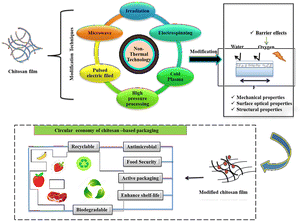
Non-thermal technologies enhance the functionality of chitosan without degrading its structure, as illustrated in Fig. 4. These methods improve its biodegradability, making it eco-friendlier and more suitable for sustainable applications. Further, enhanced biodegradation supports waste reduction and aligns with circular economy principles by promoting resource efficiency. This approach enables the development of biodegradable materials for the agriculture and food packaging industries, thereby reducing the environmental impact.
Circular economy and life cycle assessment of chitosan-based packaging
The circular economy approach to the non-thermal modifications of chitosan focuses on sustainable processing methods that minimize energy consumption and waste generation. By utilizing non-thermal modification techniques, such as cold plasma, irradiation, pulsed electric field, high pressure processing, and electrospinning, chitosan can be tailored for various applications in food, biomedical, and bioplastics while maintaining its biodegradability. These eco-friendly techniques align with the circular economy principles, thereby extending the shelf-life, minimizing post-harvest losses, reducing reliance on harsh chemicals, and potentially utilizing waste from marine and agricultural sources.Life-cycle assessment (LCA) of chitosan-based films modified using non-thermal techniques evaluates their environmental impact from raw material sourcing to end-of-life disposal. In a study, the LCA of cellulose nanofibril (CNF)-reinforced chitosan composite films evaluates the environmental impact across their production, usage and disposal phases. This assessment considers raw material extraction (e.g. chitosan from seafood waste and CNF from plant fibres), energy consumption in non-thermal modification techniques and the environmental footprint of film fabrication processes, like casting or extrusion. The results indicate that CNF-reinforced chitosan films have a lower carbon footprint, enhanced mechanical properties and improved biodegradability, supporting circular economy principles. The end-of-life scenario, such as composting or recycling, further enhances their sustainability, making them a viable eco-friendly alternative to synthetic plastic packaging.100 LCA of chitosan-based films confirms their eco-friendly potential, thereby lowering environmental impact and biodegradability compared to conventional plastics, which supports sustainable packaging solutions.
Challenges to industrial scale-up and cost barriers
Despite significant advances in non-thermal modification technologies and the demonstrated potential of chitosan-based films for sustainable packaging, the translation of these laboratory-scale innovations into industrial-scale production remains challenging. Critical barriers related to process scalability, economic feasibility, energy demand, and equipment costs continue to limit large-scale adoption, necessitating a focused discussion on industrial implementation constraints.Chitosan requires additional chemical or physical modifications often to improve mechanical strength, barrier performance, and pH stability, further increasing production costs and complicating food-contact regulatory approval. Intrinsic limitations such as moisture sensitivity (starch-based films) and pH-dependent solubility of chitosan restrict industrial applicability, and although oxygen barrier improvements of 40–60% have been reported at the laboratory scale, these are difficult to reproduce under industrial processing conditions. Moreover, fewer than 20% of studies address scalability, cost reduction, and regulatory compliance, highlighting a persistent gap between academic research and industrial translation. Nevertheless, commercial examples including Mater-Bi® and fungal chitosan-based coatings demonstrate that industrial adoption is feasible when economic, scalability, and regulatory challenges are addressed concurrently.44 Among polysaccharides, chitosan has attracted considerable interest for edible coatings of pre-processed fish products due to its biodegradability, film-forming ability, and inherent antimicrobial activity. However, the large-scale industrial implementation of chitosan-based coatings remains challenging because of the variability in raw material quality, pH-dependent solubility, and difficulties in achieving uniform coating thickness under high-throughput processing conditions. To enhance mechanical strength, barrier properties, and stability, chitosan often requires chemical, physical, or composite modifications, which can increase formulation complexity, production costs, and regulatory burdens. Additionally, the incorporation of active compounds may affect coating reproducibility and storage stability. Addressing these scale-up and modification challenges is essential for the successful industrial adoption of chitosan-based edible coatings in fishery products.101 Overall, the chitosan-based coatings show strong potential for food preservation; their industrial adoption is limited by scale-up challenges related to raw material variability, pH-dependent solubility, and formulation sensitivity. Moreover, modification strategies required to improve mechanical and barrier properties increase costs and regulatory complexity. Overcoming these issues through cost-effective processing, scalable modifications, and standardized protocols is essential for successful commercial implementation.
Conclusions
The biopolymer chitosan has recently drawn researchers' attention due to its excellent antimicrobial and antioxidant properties and film forming abilities. It is developed from naturally available sources with historical significance as a highly promising material for sustainable food packaging. Moreover, its non-toxicity and biocompatibility features provide an eco-friendly alternative to conventional synthetic plastic materials. Owing to these attractive properties, chitosan has been used in a number of fields, such as medicine, agriculture, biomedical, food packaging, wastewater treatment, biosensors, and textile industries. However, the need to improve chitosan's mechanical properties, water resistance, and oxygen barrier properties has driven research into various modification techniques. Among these, non-thermal technologies have emerged as potential candidates for enhancing chitosan's functional and structural properties without compromising its bioactivity, thereby aligning with the principles of ‘green chemistry’. The current paper reviewed applications of chitosan-based films modified using non-thermal technologies, such as cold plasma, irradiation, pulsed electric field, ultrasonication, high pressure processing, microwave, electrospinning and ozone treatment. These advancements not only enhance the structural and barrier characteristics of chitosan films but also expand their application in active and intelligent packaging systems. The non-thermal modification of chitosan plays a pivotal role in sustainable development, enhancing its properties in energy efficiency, environmental friendliness, and economic viability. These modifications enable the widespread adoption of biodegradable high-performance chitosan-based films, contributing to waste reduction, food preservation and reduction in fossil fuel-based plastic usage. With increasing market demand, industries seek greener alternatives for packaging; non-thermal modification can be a resilient pathway for achieving global sustainable development goals, thereby maintaining material efficiency and functionality.According to researchers, future studies should focus on optimizing non-thermal modification strategies for industrial-scale applications and cost-effectiveness, and on developing hybrid biopolymer composites to enhance performance and meet the growing demand for sustainable materials in additive manufacturing. Modified chitosan shows great potential as a bio-ink for 3D printing applications in food packaging, biomedical scaffolds and tissue engineering. With continued innovation, chitosan holds immense promise as a next-generation material for eco-friendly and functional food packaging systems.
Future outlook and industrial implications
Non-thermal modification technologies (e.g., cold plasma, ultrasonication, irradiation, microwave processing, and electrospinning) offer scalable and energy-efficient routes for enhancing the functionality of chitosan-based films, with reduced thermal degradation and improved retention of bioactive additives, making them highly attractive for industrial food packaging applications. Modified chitosan films with tailored surface properties and enhanced barrier and antimicrobial performance can significantly reduce post-harvest losses and extend the shelf life of perishable foods, supporting the circular economy and sustainability goals by replacing petroleum-based plastics. However, comprehensive investigations into scale-up feasibility, cost-benefit analysis, and regulatory compliance are required to facilitate the industrial adoption of these non-thermal technologies. Environmental impact assessments, including life cycle analysis (LCA) and evaluation of surface wettability under real storage conditions, are essential to quantitatively assess sustainability benefits and to optimise material design. Recent advances in surface wettability and sustainable coating technologies highlight the critical role of surface functionality in governing the barrier performance of packaging materials.102Future studies should also prioritise the development of standardised processing protocols, long-term performance evaluation, and real-world application trials, supported by strong industry-academia collaborations to bridge the gap between laboratory-scale innovations and commercial implementation.
Author contributions
Susmita Bora: original draft writing, review and editing. Tabli Ghosh: conceptualization, supervision, validation, final review and editing.Conflicts of interest
The authors declare no conflicts of interest.Data availability
No datasets were generated or analysed in the study.Acknowledgements
The authors are thankful to the Sustainable Food Lab at the Department of Food Engineering and Technology, School of Engineering, Tezpur University, Assam, India, for the support to carry out the work.References
- L. G. Pinaeva and A. S. Noskov, Biodegradable biopolymers: Real impact on environmental pollution, Sci. Total Environ., 2024, 947, 174445, DOI:10.1016/j.scitotenv.2024.174445.
- M. Vajpayee, M. Singh and L. Ledwani, Non-thermal plasma treatment of cellulosic biopolymer to enhance its surface properties for various applications: A review, Mater. Today Proc., 2021, 43, 3250–3255, DOI:10.1016/j.matpr.2021.01.905.
- S. Shaikh, M. Yaqoob and P. Aggarwal, An overview of biodegradable packaging in the food industry, Curr. Res. Food Sci., 2021, 4, 503–520, DOI:10.1016/j.crfs.2021.07.005.
- S. K. Verma, A. Prasad and V. Katiyar, State-of-the-art review on sustainable biodegradable polymers with a market overview for sustainable packaging, Mater. Today Sustain., 2024, 26, 100776, DOI:10.1016/j.mtsust.2024.100776.
- S. Islam, M. R. Bhuiyan and M. N. Islam, Chitin and chitosan: Structure, properties and applications in biomedical engineering, J. Polym. Environ., 2017, 25, 854–866, DOI:10.1007/s10924-016-0865-5.
- N. Oladzadabbasabadi, A. M. Nafchi, F. Ariffin, M. J. O. Wijekoon, A. A. Al-Hassan, M. A. Dheyab and M. Ghasemlou, Recent advances in extraction, modification, and application of chitosan in packaging industry, Carbohydr. Polym., 2022, 277, 118876, DOI:10.1016/j.carbpol.2021.118876.
- Y. Luo and Q. Wang, Recent development of chitosan-based polyelectrolyte complexes with natural polysaccharides for drug delivery, Int. J. Biol. Macromol., 2014, 64, 353–367, DOI:10.1016/j.ijbiomac.2013.12.017.
- M. Ul-Islam, K. F. Alabbosh, S. Manan, S. Khan, F. Ahmad and M. W. Ullah, Chitosan-based nanostructured biomaterials: Synthesis, properties, and biomedical applications, Adv. Ind. Eng. Polym. Res., 2024, 7, 79–99, DOI:10.1016/j.aiepr.2023.07.002.
- H. Luo, D. Liang, Q. Liu, L. Niu, K. Temirlan and W. Li, Electron beam irradiation combined with cold plasma modification of chitosan to enhance physicochemical and functional properties, Carbohydr. Polym., 2025, 354, 123308, DOI:10.1016/j.carbpol.2025.123308.
- J. M. Wasikiewicz and S. G. Yeates, Green molecular weight degradation of chitosan using microwave irradiation, Polym. Degrad. Stab., 2013, 98, 863–867, DOI:10.1016/j.polymdegradstab.2012.12.028.
- H. Liu, J. Bao, Y. Du, X. Zhou and J. F. Kennedy, Effect of ultrasonic treatment on the biophysicochemical properties of chitosan, Carbohydr. Polym., 2006, 64, 553–559, DOI:10.1016/j.carbpol.2005.11.007.
- G. A. Sutil, K. S. Andrade, E. A. Rebelatto and M. Lanza, Effects of incorporation of pure or multicomponent active agents in biopolymers for food packaging using supercritical CO2, Trends Food Sci. Technol., 2022, 120, 349–362, DOI:10.1016/j.tifs.2022.01.025.
- D. Das, P. S. Panesar, C. S. Saini and J. F. Kennedy, Improvement in properties of edible film through non-thermal treatments and nanocomposite materials: A review, Food Packag. Shelf Life, 2022, 32, 100843, DOI:10.1016/j.fpsl.2022.100843.
- Y. Li, C. Wu, Y. Bai, S. Liu, C. Yuan, T. Ding and Y. Hu, Effect of glow discharge plasma on surface modification of chitosan film, Int. J. Biol. Macromol., 2019, 138, 340–348, DOI:10.1016/j.ijbiomac.2019.07.039.
- L. F. Han, C. P. Tan, R. M. Zawawi and N. H. ZA, Effect of sonication time and heat treatment on the structural and physical properties of chitosan/graphene oxide nanocomposite films, Food Packag. Shelf Life, 2021, 28, 100663, DOI:10.1016/j.fpsl.2021.100663.
- V. Siracusa, P. Rocculi, S. Romani and M. Dalla Rosa, Biodegradable polymers for food packaging: A review, Trends Food Sci. Technol., 2008, 19, 634–643, DOI:10.1016/j.tifs.2008.07.003.
- G. Crini, Historical review on chitin and chitosan biopolymers, Environ. Chem. Lett., 2019, 17, 1623–1643, DOI:10.1007/s10311-019-00901-0.
- M. H. Periayah, A. S. Halim and A. Z. M. Saad, Chitosan: A promising marine polysaccharide for biomedical research, Pharmacogn. Rev., 2016, 10, 39, DOI:10.4103/0973-7847.176545.
- M. Rinaudo and A. Domard, “Chitin and chitosan: Solution properties of chitosan, In Chitin and Chitosan, London, UK: Elsevier, 1989, pp. 71–86 Search PubMed.
- H. Yadav, R. Malviya and N. Kaushik, Chitosan in biomedicine: A comprehensive review of recent developments, Carbohydr. Polym. Technol. Appl., 2024, 8, 100551, DOI:10.1016/j.carpta.2024.100551.
- R. Paneru, S. H. Ki, P. Lamichhane, L. N. Nguyen, B. C. Adhikari, I. J. Jeong and E. H. Choi, et al., Enhancement of antibacterial and wettability performances of polyvinyl alcohol/chitosan film using non-thermal atmospheric pressure plasma, Appl. Surf. Sci., 2020, 532, 147339, DOI:10.1016/j.apsusc.2020.147339.
- R. N. Pereira and A. A. Vicente, Environmental impact of novel thermal and non-thermal technologies in food processing, Food Res. Int., 2010, 43, 1936–1943, DOI:10.1016/j.foodres.2009.09.013.
- P. Cazón, G. Velazquez, J. A. Ramírez and M. Vázquez, Polysaccharide-based films and coatings for food packaging: A review, Food Hydrocolloids, 2017, 68, 136–148, DOI:10.1016/j.foodhyd.2016.09.009.
- S. M. Joseph, S. Krishnamoorthy, R. Paranthaman, J. A. Moses and C. Anandharamakrishnan, A review on source-specific chemistry, functionality, and applications of chitin and chitosan, Carbohydr. Polym. Technol. Appl., 2021, 2, 100036, DOI:10.1016/j.carpta.2021.100036.
- A. Kumar and K. Y. J. Zhang, Human chitinases: Structure, function, and inhibitor discovery, In Targeting Chitin-Containing Organisms, 2019, pp. 221–251, DOI:10.1007/978-981-13-7318-3_11.
- H. Fabritius, C. Sachs, D. Raabe, S. Nikolov, M. Friák and J. Neugebauer, Chitin in the exoskeletons of Arthropoda: From ancient design to novel materials science, In Chitin: Formation and Diagenesis, Springer Netherlands, Dordrecht, 2010, pp. 35–60, DOI:10.1007/978-90-481-9684-5_2.
- L. Shamshina, O. Zavgorodnya and R. D. Rogers, Advances in processing chitin as a promising biomaterial from ionic liquids, In Application of Ionic Liquids in Biotechnology, 2018, pp. 177–198, DOI:10.1007/10_2018_63.
- P. Sikorski, R. Hori and M. Wada, Revisit of α-chitin crystal structure using high resolution X-ray diffraction data, Biomacromolecules, 2009, 10, 1100–1105, DOI:10.1021/bm801251e.
- M. Kozma, B. Acharya and R. Bissessur, Chitin, chitosan, and nanochitin: Extraction, synthesis, and applications, Polymers, 2022, 14, 3989, DOI:10.3390/polym14193989.
- M. Rinaudo, Chitin and chitosan: Properties and applications, Prog. Polym. Sci., 2006, 31, 603–632, DOI:10.1016/j.progpolymsci.2006.06.001.
- A. Pellis, G. M. Guebitz and G. S. Nyanhongo, Chitosan: Sources, processing and modification techniques, Gels, 2022, 8, 393, DOI:10.3390/gels8070393.
- H. El Knidri, R. Belaabed, A. Addaou, A. Laajeb and A. Lahsini, Extraction, chemical modification and characterization of chitin and chitosan, Int. J. Biol. Macromol., 2018, 120, 1181–1189, DOI:10.1016/j.ijbiomac.2018.08.139.
- M. Pakizeh, A. Moradi and T. Ghassemi, Chemical extraction and modification of chitin and chitosan from shrimp shells, Eur. Polym. J., 2021, 159, 110709, DOI:10.1016/j.eurpolymj.2021.110709.
- J. Wang and S. Zhuang, Chitosan-based materials: Preparation, modification and application, J. Clean. Prod., 2022, 355, 131825, DOI:10.1016/j.jclepro.2022.131825.
- P. Putnik, Ž. Kresoja, T. Bosiljkov, A. R. Jambrak, F. J. Barba, J. M. Lorenzo and D. B. Kovačević, et al., Life cycle assessment of manufacturing cellulose nanofibril-reinforced chitosan composite films for packaging applications, Int. J. Life Cycle Assess., 2022, 27, 380–394, DOI:10.1016/j.foodchem.2018.11.131.
- S. K. Pankaj, Z. Wan and K. M. Keener, Effects of cold plasma on food quality: A review, Foods, 2018, 7, 4, DOI:10.3390/foods7010004.
- S. K. Pankaj, C. Bueno-Ferrer, N. N. Misra, V. Milosavljević, C. P. O'Donnell, P. Bourke and P. J. Cullen, Applications of cold plasma technology in food packaging, Trends Food Sci. Technol., 2014, 35, 5–17, DOI:10.1016/j.tifs.2013.10.009.
- B. A. Niemira, Cold plasma decontamination of foods, Annu. Rev. Food Sci. Technol., 2012, 3, 125–142, DOI:10.1146/annurev-food-022811-101132.
- S. P. Bangar, S. Suri, P. Nayi and Y. Phimolsiripol, Cold plasma for microbial safety: Principle, mechanism, and factors responsible, J. Food Process. Preserv., 2022, 46, e16850, DOI:10.1111/jfpp.16850.
- A. R. Ganesan, U. Tiwari, P. N. Ezhilarasi and G. Rajauria, Application of cold plasma on food matrices: A review on current and future prospects, J. Food Process. Preserv., 2021, 45, e15070, DOI:10.1111/jfpp.15070.
- S. Dong, P. Guo, Y. Chen, G. Y. Chen, H. Ji and Ye. Ran, et al., Surface modification via atmospheric cold plasma (ACP): Improved functional properties and characterization of zein film, Ind. Crops Prod., 2018, 115, 124–133, DOI:10.1016/j.indcrop.2018.01.080.
- N. Bahrami, D. Bayliss, G. Chope, S. Penson, T. Perehinec and I. D. Fisk, Cold plasma: A new technology to modify wheat flour functionality, Food Chem., 2016, 202, 247–253, DOI:10.1016/j.foodchem.2016.01.113.
- S. Beikzadeh, M. Ghorbani, N. Shahbazi, F. Izadi, Z. Pilevar and A. M. Mortazavian, The effects of novel thermal and nonthermal technologies on the properties of edible food packaging, Food Eng. Rev., 2020, 12, 333–345, DOI:10.1007/s12393-020-09227-y.
- P. M. Junaid, A. Saikumar, R. U. Islam, M. K. Patel, D. H. U. Eranda, M. Islam and L. S. Badwaik, Polysaccharide-based biopolymers: Exploring film fabrication techniques, molecular interactions, and their potential food applications, Qual. Assur. Saf. Crop Foods, 2025, 17, 257–283, DOI:10.15586/qas.v17i4.1581.
- S. Akhavan-Mahdavi, M. Mirzazadeh, Z. Alam and S. Solaimanimehr, The effect of chitosan coating combined with cold plasma on the quality and safety of pistachio during storage, Food Nutr. Sci., 2023, 11, 4296–4307, DOI:10.1002/fsn3.3355.
- G. Chen, S. Dong, S. Zhao, S. Li and Y. Chen, Improving functional properties of zein film via compositing with chitosan and cold plasma treatment, Ind. Crops Prod., 2019, 129, 318–326, DOI:10.1016/j.indcrop.2018.11.072.
- M. Kongboonkird, P. Chuesiang, V. Ryu and U. Siripatrawan, Enhancing functional properties of active chitosan film incorporated with cinnamon oil nanoemulsion using argon dielectric barrier discharge cold plasma treatment, Food Hydrocolloids, 2024, 156, 110171, DOI:10.1016/j.foodhyd.2024.110171.
- S. K. Pankaj, C. Bueno-Ferrer, N. N. Misra, L. O'Neill, P. Bourke and P. J. Cullen, Effects of cold plasma on surface, thermal and antimicrobial release properties of chitosan film, J. Renew. Mater., 2017, 5, 14–20, DOI:10.7569/JRM.2016.634105.
- G. Y. Chen, T. L. Yang, Y. H. Wang, S. H. Li and Y. Chen, “Properties enhancement of antimicrobial chitosan-deposited polylactic acid films via cold plasma treatment,”, J. Food Health, 2023, 5, 11, DOI:10.53388/FH2023011.
- A. Oberlintner, A. Vesel, K. Naumoska, B. Likozar and U. Novak, Permanent hydrophobic coating of chitosan/cellulose nanocrystals composite film by cold plasma processing, Appl. Surf. Sci., 2022, 597, 153562, DOI:10.1016/j.apsusc.2022.153562.
- J. O. Abu, K. G. Duodu and A. Minnaar, Effect of γ-irradiation on some physicochemical and thermal properties of cowpea (Vigna unguiculata L. Walp) starch, Food Chem., 2006, 95, 386–393, DOI:10.1016/j.foodchem.2005.01.008.
- C. Fischbach, J. Tessmar, A. Lucke, E. Schnell, G. Schmeer, T. Blunk and A. Göpferich, Does UV irradiation affect polymer properties relevant to tissue engineering?, Surf. Sci., 2001, 491, 333–345 CrossRef CAS.
- A. Sionkowska, M. Wiśniewski, J. Skopińska, C. J. Kennedy and T. J. Wess, Molecular interactions in collagen and chitosan blends, Biomaterials, 2004, 25, 795–801, DOI:10.1016/S0142-9612(03)00595-7.
- A. Sionkowska, H. Kaczmarek, M. Wiśniewski, J. Skopińska, S. Lazare and V. Tokarev, The influence of UV irradiation on the surface of chitosan films, Surf. Sci., 2006, 600, 3775–3779, DOI:10.1016/j.susc.2006.01.090.
- S. Shankar, D. Khodaei and M. Lacroix, Effect of chitosan/essential oils/silver nanoparticles composite films packaging and gamma irradiation on shelf life of strawberries, Food Hydrocolloids, 2021, 117, 106750, DOI:10.1016/j.foodhyd.2021.106750.
- D. H. U. Eranda, M. Chaijan, W. Panpipat, S. Karnjanapratum, M. A. Cerqueira and R. Castro-Muñoz, Gelatin–chitosan interactions in edible films and coatings doped with plant extracts for biopreservation of fresh tuna fish products: A review, Int. J. Biol. Macromol., 2024, 280, 135661, DOI:10.1016/j.ijbiomac.2024.135661.
- R. Severino, G. Ferrari, K. D. Vu, F. Donsì, S. Salmieri and M. Lacroix, Antimicrobial effects of modified chitosan based coating containing nanoemulsion of essential oils, modified atmosphere packaging and gamma irradiation against Escherichia coli O157:H7 and Salmonella Typhimurium on green beans, Food Control, 2015, 50, 215–222, DOI:10.1016/j.foodcont.2014.08.029.
- J. George, Siddaramaiah “High performance edible nanocomposite films containing bacterial cellulose nanocrystals, Carbohydr. Polym., 2012, 87, 2031–2037, DOI:10.1016/j.carbpol.2011.10.019.
- M. Salari, M. S. Khiabani, R. R. Mokarram, B. Ghanbarzadeh and H. S. Kafil, Use of gamma irradiation technology for modification of bacterial cellulose nanocrystals/chitosan nanocomposite film, Carbohydr. Polym., 2021, 253, 117144, DOI:10.1016/j.carbpol.2020.117144.
- M. A. García, L. Pérez, N. de la Paz, J. González, M. Rapado and A. Casariego, Effect of molecular weight reduction by gamma irradiation on chitosan film properties, Mater. Sci. Eng., C, 2015, 55, 174–180, DOI:10.1016/j.msec.2015.05.009.
- P. Hassanzadeh, H. Tajik, S. M. R. Rohani, M. Moradi, M. Hashemi and J. Aliakbarlu, Effect of functional chitosan coating and gamma irradiation on the shelf-life of chicken meat during refrigerated storage, Radiat. Phys. Chem., 2017, 141, 103–109, DOI:10.1016/j.radphyschem.2017.06.014.
- D. J. McClements, Advances in the application of ultrasound in food analysis and processing, Trends Food Sci. Technol., 1995, 6, 293–299, DOI:10.1016/S0924-2244(00)89139-6.
- I. Majid, G. A. Nayik and V. Nanda, Ultrasonication and food technology: A review, Cogent Food Agric., 2015, 1, 1071022, DOI:10.1080/23311932.2015.1071022.
- T. Mi, X. Zhang, P. Liu, W. Gao, J. Li, N. Xu, C. Yuan and B. Cui, Ultrasonication effects on physicochemical properties of biopolymer-based films: A comprehensive review, Crit. Rev. Food Sci. Nutr., 2023, 63, 5044–5062, DOI:10.1080/10408398.2021.2012420.
- Y. Wang, Y. Feng, X. Wang, C. Ji, Z. Xiao and Y. Luo, Ultrasonication-based preparation of raw chitin nanofiber and evaluation of its reinforcement effect on chitosan film for functionalization with curcumin, Food Hydrocolloids, 2024, 155, 110193, DOI:10.1016/j.foodhyd.2024.110193.
- M. Thirupathi Vasuki, V. Kadirvel and G. Pejavara Narayana, Smart packaging—An overview of concepts and applications in various food industries, Food Bioeng., 2023, 2, 25–41, DOI:10.1002/fbe2.12038.
- J. Ding, R. Zhang, S. Ahmed, Y. Liu and W. Qin, Effect of sonication duration in the performance of polyvinyl alcohol/chitosan bilayer films and their effect on strawberry preservation, Molecules, 2019, 24, 1408, DOI:10.3390/molecules24071408.
- L. Wang, J. Ding, Y. Fang, X. Pan, F. Fan, P. Li and Q. Hu, Effect of ultrasonic power on properties of edible composite films based on rice protein hydrolysates and chitosan, Ultrason. Sonochem., 2020, 65, 105049, DOI:10.1016/j.ultsonch.2020.105049.
- W. Qu, T. Guo, X. Zhang, Y. Jin, B. Wang, H. Wahia and H. Ma, Preparation of tuna skin collagen-chitosan composite film improved by sweep frequency pulsed ultrasound technology, Ultrason. Sonochem., 2022, 82, 105880, DOI:10.1016/j.ultsonch.2021.105880.
- R. N. Arshad, Z. Abdul-Malek, U. Roobab, M. A. Munir, A. Naderipour, M. I. Qureshi and R. M. Aadil, et al., Pulsed electric field: A potential alternative towards a sustainable food processing, Trends Food Sci. Technol., 2021, 111, 43–54, DOI:10.1016/j.tifs.2021.02.041.
- L. M. Castro, E. M. Alexandre, J. A. Saraiva and M. Pintado, Starch extraction and modification by pulsed electric fields, Food Rev. Int., 2023, 39, 2161–2182, DOI:10.1080/87559129.2021.1945620.
- S. G. Giteru, M. A. Ali and I. Oey, Optimisation of pulsed electric fields processing parameters for developing biodegradable films using zein, chitosan and poly(vinyl alcohol), Innov. Food Sci. Emerg. Technol., 2020, 60, 102287, DOI:10.1016/j.ifset.2020.102287.
- W. B. Luo, Z. Han, X. A. Zeng, S. J. Yu and J. F. Kennedy, Study on the degradation of chitosan by pulsed electric fields treatment, Innov. Food Sci. Emerg. Technol., 2010, 11, 587–591, DOI:10.1016/j.ifset.2010.04.002.
- S. G. Giteru, A. Ali and I. Oey, Understanding the relationship between rheological characteristics of pulsed electric fields treated chitosan-zein-poly(vinyl alcohol)-polyethylene glycol composite dispersions and the structure-function of their resulting thin-films, Food Hydrocoll., 2021, 113, 106452, DOI:10.1016/j.foodhyd.2020.106452.
- C. C. Coelho, M. A. Cerqueira, R. N. Pereira, L. M. Pastrana and O. Freitas-Silva, et al., Effect of moderate electric fields in the properties of starch and chitosan films reinforced with microcrystalline cellulose, Carbohydr. Polym., 2017, 174, 1181–1191, DOI:10.1016/j.carbpol.2017.07.007.
- D. F. Farkas, A short history of research and development efforts leading to the commercialization of high-pressure processing of food, in High Pressure Processing of Food: Principles, Technology and Applications, Springer New York, New York, NY, 2016, pp. 19–36, DOI:10.1007/978-1-4939-3234-4_2.
- L. M. Júnior, M. Cristianini, M. Padula and C. A. R. Anjos, Effect of high-pressure processing on characteristics of flexible packaging for foods and beverages, Food Res. Int., 2019, 119, 920–930, DOI:10.1016/j.foodres.2018.10.078.
- M. Hoque, C. McDonagh, B. K. Tiwari, J. P. Kerry and S. Pathania, Effect of high-pressure processing on the packaging properties of biopolymer-based films: A review, Polymers, 2022, 14, 3009, DOI:10.3390/polym14153009.
- L. Chen, D. Jiao, B. Zhou, C. Zhu, J. Liu, D. Zhang and H. Liu, Shrimp (Penaeus monodon) preservation by using chitosan and tea polyphenol coating combined with high-pressure processing, Food Nutr. Sci., 2022, 10, 3395–3404, DOI:10.1002/fsn3.2939.
- S. Martillanes, J. Rocha-Pimienta, J. Llera-Oyola, M. V. Gil, M. C. Ayuso-Yuste, J. García-Parra and J. Delgado-Adámez, Control of Listeria monocytogenes in sliced dry-cured Iberian ham by high pressure processing in combination with an eco-friendly packaging based on chitosan, nisin and phytochemicals from rice bran, Food Control, 2021, 124, 107933, DOI:10.1016/j.foodcont.2021.107933.
- Z. Lian, Y. Zhang and Y. Zhao, Nano-TiO2 particles and high hydrostatic pressure treatment for improving functionality of polyvinyl alcohol and chitosan composite films and nano-TiO2 migration from film matrix in food simulants, Innov. Food Sci. Emerg. Technol., 2016, 33, 145–153, DOI:10.1016/j.ifset.2015.10.008.
- J. Gómez-Estaca, P. Montero, B. Giménez and M. C. Gómez-Guillén, Effect of functional edible films and high pressure processing on microbial and oxidative spoilage in cold-smoked sardine (Sardina pilchardus), Food Chem., 2007, 105, 511–520, DOI:10.1016/j.foodchem.2007.04.006.
- S. Chandrasekaran, S. Ramanathan and T. Basak, Microwave food processing—A review, Food Res. Int., 2013, 52, 243–261, DOI:10.1016/j.foodres.2013.02.033.
- X. Chen, J. Yang, M. Shen, Yi. Chen, Q. Yu and J. Xie, Structure, function and advance application of microwave-treated polysaccharide: A review, Trends Food Sci. Technol., 2022, 123, 198–209, DOI:10.1016/j.tifs.2022.03.016.
- X. Huang, F. Xie and X. Xiong, Surface-modified microcrystalline cellulose for reinforcement of chitosan film, Carbohydr. Polym., 2018, 201, 367–373, DOI:10.1016/j.carbpol.2018.08.085.
- C. Petit, S. Reynaud and J. Desbrières, Amphiphilic derivatives of chitosan using microwave irradiation: Toward an eco-friendly process to chitosan derivatives, Carbohydr. Polym., 2015, 116, 26–33, DOI:10.1016/j.carbpol.2014.04.083.
- J. D. Schiffman and C. L. Schauer, A review: electrospinning of biopolymer nanofibers and their applications, Polym. Rev., 2008, 48, 317–352, DOI:10.1080/15583720802022182.
- Z. Y. Qin, X. W. Jia, Q. Liu, B. H. Kong and H. Wang, Fast dissolving oral films for drug delivery prepared from chitosan/pullulan electrospinning nanofibers, Int. J. Biol. Macromol., 2019, 137, 224–231, DOI:10.1016/j.ijbiomac.2019.06.22.
- S. Nokhasteh, A. M. Molavi, M. Khorsand-Ghayeni and A. Sadeghi-Avalshahr, Preparation of PVA/chitosan samples by electrospinning and film casting methods and evaluating the effect of surface morphology on their antibacterial behavior, Mater. Res. Express, 2019, 7, 015401, DOI:10.1088/2053-1591/ab572c.
- E. Mercan, Gelatine-based biopolymer film produced from ozone-treated film-forming solutions containing whey protein concentrate: Effects on physical, mechanical, and thermal characteristics, Appl. Sci., 2024, 14, 2176, DOI:10.3390/app14052176.
- S. G. Hu, C. H. Jou and M. C. Yang, Antibacterial and biodegradable properties of polyhydroxyalkanoates grafted with chitosan and chitooligosaccharides via ozone treatment, J. Appl. Polym. Sci., 2003, 88, 2797–2803, DOI:10.1002/app.12055.
- S. S. Ebrahimi, Y. Hamzeh, A. Ashori, M. Roohani, N. Marlin and G. Spigno, Ozone-activated lignocellulose films blended with chitosan for edible film production, Int. J. Biol. Macromol., 2024, 270, 132285, DOI:10.1016/j.ijbiomac.2024.132285.
- S. Wang, Z. Liu, M. Zhao, C. Gao, J. Wang, C. Li and D. Zhou, et al., Chitosan-wampee seed essential oil composite film combined with cold plasma for refrigerated storage with modified atmosphere packaging: A promising technology for quality preservation of golden pompano fillets, Int. J. Biol. Macromol., 2023, 224, 1266–1275, DOI:10.1016/j.ijbiomac.2022.10.212.
- G. Dan, Z. H. Zhang, X. A. Zeng, Z. Han, W. B. Luo, C. Tang and S. Y. Quek, Synergetic effects of pulsed electric field and ozone treatments on the degradation of high molecular weight chitosan, Int. J. Food Eng., 2014, 10, 775–784, DOI:10.1515/ijfe-2014-0100.
- A. Vera, C. Tapia and L. Abugoch, Effect of high-intensity ultrasound treatment in combination with transglutaminase and nanoparticles on structural, mechanical, and physicochemical properties of quinoa proteins/chitosan edible films, Int. J. Biol. Macromol., 2020, 144, 536–543, DOI:10.1016/j.ijbiomac.2019.12.120.
- D. Yuan, H. Meng, Q. Huang, C. Li and X. Fu, Preparation and characterization of chitosan-based edible active films incorporated with Sargassum pallidum polysaccharides by ultrasound treatment, Int. J. Biol. Macromol., 2021, 183, 473–480, DOI:10.1016/j.jfoodeng.2024.112440.
- F. Donsì, E. Marchese, P. Maresca, G. Pataro, K. D. Vu and S. Salmieri, et al., Green beans preservation by combination of a modified chitosan-based coating containing nanoemulsion of mandarin essential oil with high pressure or pulsed light processing, Postharvest Biol. Technol., 2015, 106, 21–32, DOI:10.1016/j.postharvbio.2015.02.006.
- S. Ebrahimzadeh, M. R. Bari, H. Hamishehkar, H. S. Kafil and L. T. Lim, Essential oils-loaded electrospun chitosan–poly(vinyl alcohol) nonwovens laminated on chitosan film as bilayer bioactive edible films, LWT--Food Sci. Technol., 2021, 144, 111217, DOI:10.1016/j.lwt.2021.111217.
- A. Günlü, S. Sipahioğlu and H. Alpas, The effect of chitosan-based edible film and high hydrostatic pressure process on the microbiological and chemical quality of rainbow trout (Oncorhynchus mykiss Walbaum) fillets during cold storage (4 ± 1 °C), High Press. Res., 2014, 34, 110–121, DOI:10.1080/08957959.2013.836643.
- P. G. Ponnusamy and S. Mani, Life cycle assessment of manufacturing cellulose nanofibril-reinforced chitosan composite films for packaging applications, Int. J. Life Cycle Assess., 2022, 27, 380–394, DOI:10.1007/s11367-022-02035-y.
- D. H. U. Eranda, M. Chaijan, I. Uysal-Unalan, W. Panpipat, A. S. Naik and A. L. Dib, et al., Biopreservation of pre-processed fresh fish by bio-based coatings: A single strategy with multiple benefits towards waste prevention, Food Biosci., 2024, 58, 103696, DOI:10.1016/j.fbio.2024.103696.
- D. H. U. Eranda, M. Chaijan and R. Castro-Muñoz, Current advances in surface wettability in food packaging materials: Strategies, methods and future trends, J. Food Eng., 2025, 391, 112440, DOI:10.1016/j.jfoodeng.2024.112440.
Footnote |
| † The authors have contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |