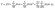Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Valorization of rambutan peel (Nephelium lappaceum L.) byproducts through pulsed electric field-assisted green extraction of pectin optimized by response surface methodology
Luqman Agung Wicaksono ab,
Ganes Aurora Santosoa,
Muhammad Alfid Kurniantoab,
Anugerah Dany Priyanto
ab,
Ganes Aurora Santosoa,
Muhammad Alfid Kurniantoab,
Anugerah Dany Priyanto ac,
Teeradate Kongpichitchoke
ac,
Teeradate Kongpichitchoke d and
Muhammad Rosyid Wardianto
d and
Muhammad Rosyid Wardianto *a
*a
aDepartment of Food Technology, Faculty of Engineering and Sciences, Universitas Pembangunan Nasional Veteran Jawa Timur, Surabaya, East Java, Indonesia. E-mail: rosyid_wardianto.tp@upnjatim.ac.id
bResearch Group of Emerging Technologies for Food Processing, Universitas Pembangunan Nasional Veteran Jawa Timur, Indonesia
cDepartment of Tropical Agriculture and International Cooperation, International College, National Pingtung University of Science and Technology, Pingtung, Taiwan
dBritish Academic Center, Bangkok, Thailand
First published on 29th January 2026
Abstract
The global demand for sustainable food ingredients has driven interest in fruit by-product valorization into high-value compounds. Rambutan (Nephelium lappaceum L.) peel, an underutilised fruit by-product, contains abundant pectin, which is suitable for various industrial applications such as gelling and emulsifying agents. This study explored a green extraction strategy combining pulsed electric field (PEF) pretreatment with optimization by response surface methodology (RSM) to recover high-quality pectin while minimising thermal degradation and processing time. Three identified key factors influencing extraction performance, such as electric field strength, extraction time, and solid-to-solvent ratio, were optimized using a Central Composite Design (CCD). The optimum condition (20 kV cm−1, 12.69 min, 30 mL g−1) produced pectin with 4.37% yield, 8.76% methoxyl content, 75.72% galacturonic acid, and 65.66% degree of esterification, classifying it as high methoxyl pectin. Structural characterisation via Fourier Transform Infrared Spectroscopy confirmed functional groups consistent with commercial standards, while Scanning Electron Microscopy revealed extensive cell wall disruption induced by PEF. This approach not only enhances extraction efficiency but also provides an eco-friendly valorisation pathway for tropical fruit waste, contributing to circular economy practices in the food industry.
Sustainability spotlightThis study advances sustainability by valorizing rambutan peel, an underutilized agro-waste, through pulsed electric field (PEF)-assisted extraction of pectin. The method reduces energy use and chemical inputs while enhancing efficiency, aligning with SDG 12 (Responsible Consumption and Production) and SDG 13 (Climate Action). |
1 Introduction
Rambutan (Nephelium lappaceum L.) is a red, round tropical fruit with soft spines on its skin, which is widely cultivated in Southeast Asia, particularly Indonesia, and has lots of varieties, such as the Binjai cultivar, which has medium-thick skin.1 National statistics show consistently high yields—884![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 702 tons, 855
702 tons, 855![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 162 tons, and 845
162 tons, and 845![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 107 tons from 2021 to 2023—indicating its abundant availability.2 Rambutan skin, also known as rambutan peels, are a notable source of several important food-grade compounds, including pectin. Pectin is a heteropolysaccharide naturally present in the primary cell wall and middle lamella of most plants, comprising about 70% galacturonic acid monomers, which may be acetylated or methyl-esterified.3 In food sector, it serves as an emulsifier, film-former, and viscosity enhancer, commonly used in products such as jams, jellies, and fruit juices.4 It also holds potential as a biodegradable food packaging material.5 Commercial pectin is typically extracted via conventional acid hydrolysis (pH 1.5–3, 75–100 °C), a method that poses environmental concerns.3 In contrast, greener extraction methods aim to reduce extraction time, lower chemical use, and minimize energy consumption while preserving extract quality and are increasingly needed.6 Sustainable techniques include Supercritical Fluid Extraction (SFE), Ultrasound-Assisted Extraction (UAE), Microwave-Assisted Extraction (MAE), Enzyme-Assisted Extraction (EAE), and Pulsed Electric Field (PEF), to name a few, are being applied across food processing.3 Each employs distinct mechanisms—SFE operates under supercritical conditions;7 UAE generates cavitation to disrupt cells;8,9 MAE uses microwave-induced frictional heating;10 EAE applies specific enzymes for selective release; and PEF enhances intracellular diffusion through cell membrane permeabilization.11 Among these, PEF offers notable advantages, including reduced energy use, shorter processing times, and operation at relatively low temperatures.12
107 tons from 2021 to 2023—indicating its abundant availability.2 Rambutan skin, also known as rambutan peels, are a notable source of several important food-grade compounds, including pectin. Pectin is a heteropolysaccharide naturally present in the primary cell wall and middle lamella of most plants, comprising about 70% galacturonic acid monomers, which may be acetylated or methyl-esterified.3 In food sector, it serves as an emulsifier, film-former, and viscosity enhancer, commonly used in products such as jams, jellies, and fruit juices.4 It also holds potential as a biodegradable food packaging material.5 Commercial pectin is typically extracted via conventional acid hydrolysis (pH 1.5–3, 75–100 °C), a method that poses environmental concerns.3 In contrast, greener extraction methods aim to reduce extraction time, lower chemical use, and minimize energy consumption while preserving extract quality and are increasingly needed.6 Sustainable techniques include Supercritical Fluid Extraction (SFE), Ultrasound-Assisted Extraction (UAE), Microwave-Assisted Extraction (MAE), Enzyme-Assisted Extraction (EAE), and Pulsed Electric Field (PEF), to name a few, are being applied across food processing.3 Each employs distinct mechanisms—SFE operates under supercritical conditions;7 UAE generates cavitation to disrupt cells;8,9 MAE uses microwave-induced frictional heating;10 EAE applies specific enzymes for selective release; and PEF enhances intracellular diffusion through cell membrane permeabilization.11 Among these, PEF offers notable advantages, including reduced energy use, shorter processing times, and operation at relatively low temperatures.12
Optimizing electric field strength, extraction time, and solvent ratio is critical in Pulsed Electric Field (PEF)-assisted pectin extraction, as these parameters directly influence the yield and functional properties of the extracted pectin. Increasing electric field strength generally improves yield; however, excessive intensity can damage and degrade essential components.12 Extraction time is inversely related to field strength—higher intensities reduce the duration required.1 Solvent properties such as solubility, conductivity, and polarity also play key roles, as higher conductivity enhances membrane electroporation.13 Previous studies have explored electric field-assisted pectin extraction in citrus peels. For example, De Oliveira et al., (2015) used a Moderate Electric Field (MEF) of 100 V, which produced a lower yield (6.70%) compared to conventional extraction (10.90%).14 In contrast, Du et al., (2024) demonstrated that High Intensity Pulsed Electric Field (HIPEF) at 30 kV cm−1 as a pre-treatment yielded more pectin (18.81%) than conventional methods (17.34%).15 A quite similar study with more concern on extraction time was also carried out in (ref. 1), which showed that pectin extraction is most efficient at 20 kV cm−1 with just 10 minutes of processing, yielding 3.48% and proving that a short, targeted process can match the effectiveness of optimized solvent ratios. However, no studies have yet applied PEF to Binjai rambutan peel, presenting an opportunity for innovation and new contributions in this area. This research was conducted in two stages: a preliminary study and an optimization study. The preliminary stage uses the One Factor at a Time (OFAT) approach to determine operational limits for each factor.1 Optimization is performed using Response Surface Methodology (RSM) with a Central Composite Design (CCD) in Design Expert 13 software. RSM was selected due to its efficiency in significantly reducing the number of experiments compared to simple factorial designs.16 CCD is particularly advantageous for exploratory studies, as its axial points allow for broader prediction of optimal conditions.1,17 The optimization focuses on three main factors—PEF electric field strength (kV cm−1), extraction time (minutes), and solid-to-solvent ratio (w/v)—with responses in terms of yield (%), equivalent weight (mg), methoxyl content (%), galacturonic acid content (%), and degree of esterification (%).18 This study aims to determine the optimal voltage, extraction time, and solid-to-solvent ratio for PEF-assisted pectin extraction from Binjai rambutan peel using RSM, while evaluating their effects on yield, equivalent weight, methoxyl content, galacturonic acid, degree of esterification, proximate composition, functional groups, and structural integrity of the optimized product.
2 Materials and methods
2.1 Materials and chemicals
Fresh rambutan (Nephelium lappaceum L., Binjai cultivar) peels were collected from a local market in Sidoarjo, East Java, Indonesia. The chemicals used in this study included citric acid (C6H8O7, 0.1 M, technical grade; Bohr chemical), hydrochloric acid (HCl, 0.1 N and 37%, analytical grade; SAP chemical), ethanol (96%, technical grade), sodium chloride (NaCl, analytical grade; SAP chemical), sodium hydroxide (NaOH, analytical grade; SAP chemical), and phenolphthalein (PP, indicator; SAP chemical). Distilled water was used throughout the experiments.2.2 Equipment
The instruments used included a food dehydrator, a household blender (Panasonic), an 80-mesh sieve, and an analytical balance (Sartorius, precision 0.0001 g). Additional equipment comprised a magnetic stirrer hot plate, a water bath shaker (Daihan scientific), and a lab-scale pulsed electric field (PEF) system operating at 0–20 kV cm−1, 8.197 kHz frequency, 66 µs pulse width, and 50 rpm stirring speed. A centrifuge (Corona, 12-place rotor) with 15 mL tubes, a 400-mesh filter cloth, pH indicator strips (Merck), and an oven dryer were also used. Pectin characterization was performed using a Fourier Transform Infrared Spectrophotometer (FTIR, IRTracer-100, Shimadzu, Japan) and a Scanning Electron Microscope (SEM, SU3500, Hitachi, Japan).2.3 Preparation of Rambutan peel flour
Rambutan peel flour was prepared following the methods of Laiya et al., (2020) with modifications.19 Rambutan peels were washed under running water, cut into small pieces, and dried at 70 °C for 8 h in a food dehydrator. The dried peels were ground into powder, sieved through an 80-mesh screen, and stored in sealed polyethylene bags with silica gel until use.2.4 Extraction of pectin
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) and allowed to stand for 24 h. The precipitate was washed with ethanol until colorless and dried at 45 °C for 5 h. The dried extract was collected as pectin. This method was adapted from De Oliveira et al., (2015) and Du et al., (2024) with modifications.14,15
1, v/v) and allowed to stand for 24 h. The precipitate was washed with ethanol until colorless and dried at 45 °C for 5 h. The dried extract was collected as pectin. This method was adapted from De Oliveira et al., (2015) and Du et al., (2024) with modifications.14,15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, w/v). The mixture was heated at 80 °C for 2 h, centrifuged at 3500 rpm for 20 min, and the supernatant was precipitated with ethanol (1
30, w/v). The mixture was heated at 80 °C for 2 h, centrifuged at 3500 rpm for 20 min, and the supernatant was precipitated with ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) for 24 h. The precipitate was washed with ethanol, dried at 45 °C for 5 h, and collected as pectin.
1, v/v) for 24 h. The precipitate was washed with ethanol, dried at 45 °C for 5 h, and collected as pectin.2.5 Optimization study
A two-stage optimization strategy was applied. Preliminary trials used a One Factor at a Time (OFAT) approach to establish operational ranges for PEF voltage (5–20 kV cm−1), extraction time (5–20 min), and solid-to-solvent ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20–1
20–1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50, w/v). Optimization was then performed using Response Surface Methodology (RSM) with a Central Composite Design (CCD), generating 20 experimental runs. The optimization process was modeled using a second-order response surface methodology (RSM) equation.20 The general form of the model is:
50, w/v). Optimization was then performed using Response Surface Methodology (RSM) with a Central Composite Design (CCD), generating 20 experimental runs. The optimization process was modeled using a second-order response surface methodology (RSM) equation.20 The general form of the model is:where Y represents the predicted response, β0 is the constant term, βi denotes the linear coefficients, βii represents the quadratic coefficients, and βij denotes the interaction coefficients. The independent variables are expressed as xi and xj. This quadratic polynomial model allows simultaneous evaluation of linear, quadratic, and interactive effects of the process parameters on the response, thereby providing a robust framework for optimization.
2.6 Evaluation of pectin properties
Pectin yield, moisture content, and ash content were determined using AOAC21,46 methods. Equivalent weight, methoxyl content, galacturonic acid, and degree of esterification were analyzed following established protocols.1,22 Functional groups were identified using FTIR (IRTracer-100 Shimadzu), while surface morphology was examined with SEM (SU3500 Hitachi).2.7 Statistical analysis
The experimental design and optimization were conducted using Design Expert version 13 (Stat-Ease Inc., Minneapolis, USA). The statistical software was employed to develop the Response Surface Methodology (RSM) model, perform model fitting, and evaluate the significance of linear, quadratic, and interaction effects through Analysis of Variance (ANOVA). Model adequacy was validated by Analysis of Variance (ANOVA), with the following criteria: (i) a significant regression model (p < 0.05), (ii) non-significant lack of fit (p > 0.05), (iii) adjusted and predicted R2 values differing by <0.2, and (iv) adequate precision >4. Predicted optimal conditions were verified by comparison with experimental results within a 95% confidence interval.3 Results and discussion
3.1 Optimization of pectin extraction from rambutan peel
As depicted in Fig. 1, the experimental design generated 20 treatment runs, with pectin yield ranging from 1.90% to 4.30%. The lowest yield (1.90%) was observed at 6.59 kV cm−1 for 10 minutes with a solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25, whereas the highest yield (4.30%) occurred at 20 kV cm−1 for 15 minutes with a 1
25, whereas the highest yield (4.30%) occurred at 20 kV cm−1 for 15 minutes with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
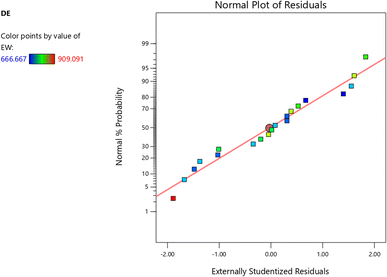
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 solvent ratio. This confirmed that stronger electric fields and balanced extraction times enhance cell wall permeabilization and release of pectic substances, while excessively low or prolonged conditions reduced efficiency. Equivalent weight values ranged between 666.67 and 909.09 mg, with the highest found at lower field intensities (10 kV cm−1, 5 minutes, 1
30 solvent ratio. This confirmed that stronger electric fields and balanced extraction times enhance cell wall permeabilization and release of pectic substances, while excessively low or prolonged conditions reduced efficiency. Equivalent weight values ranged between 666.67 and 909.09 mg, with the highest found at lower field intensities (10 kV cm−1, 5 minutes, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 ratio), indicating higher molecular stability of pectin under milder extraction conditions. Galacturonic acid content, a marker of pectin purity, varied from 51.04% to 78.32%. All galacturonic acid results found in this study were well above the minimum international standard of 35%,23 with the highest content recorded under conditions of 20 kV cm−1 for 15 minutes at a 1
20 ratio), indicating higher molecular stability of pectin under milder extraction conditions. Galacturonic acid content, a marker of pectin purity, varied from 51.04% to 78.32%. All galacturonic acid results found in this study were well above the minimum international standard of 35%,23 with the highest content recorded under conditions of 20 kV cm−1 for 15 minutes at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 ratio. The degree of esterification ranged from 61.54% to 66.29%, consistently classifying the pectin as high-methoxyl pectin (HMP, >50%), which is known for its superior gelling ability in conventional jam and jelly formulations.
30 ratio. The degree of esterification ranged from 61.54% to 66.29%, consistently classifying the pectin as high-methoxyl pectin (HMP, >50%), which is known for its superior gelling ability in conventional jam and jelly formulations.
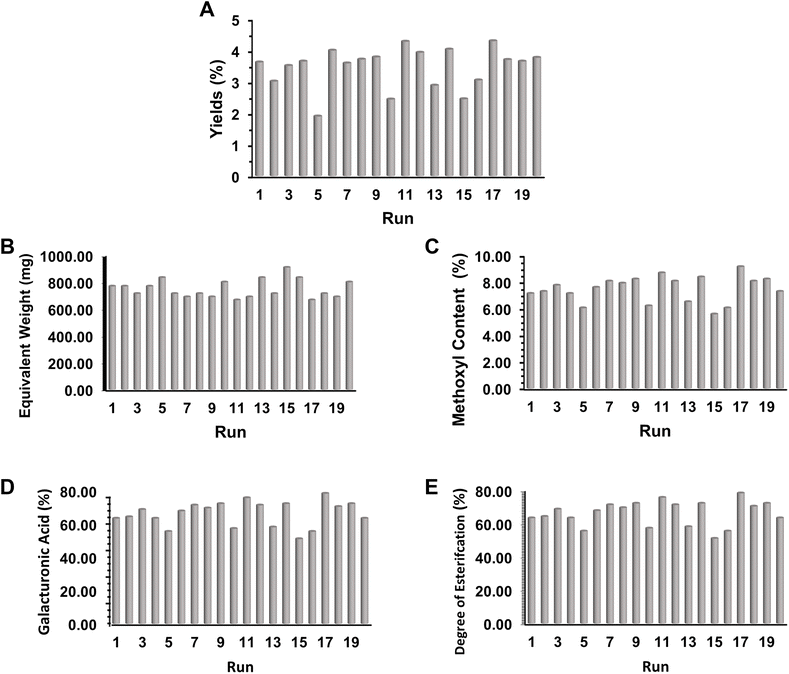 | ||
| Fig. 1 Histograms of (A) pectin yield test results, (B) equivalent weight, (C) methoxyl content, (D) galacturonic acid content and (E) degree of esterification. | ||
Overall, the RSM model demonstrated that the combined effects of high electric field intensity, moderate extraction time, and an optimal solid-to-solvent ratio significantly enhanced both pectin yield and quality. The optimized condition—20 kV cm−1, 15 minutes, and a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 ratio—yielded pectin with high purity (GalA > 75%), favorable methoxyl content (9%), and strong gelling properties (DE > 65%). These findings highlighted the potential of rambutan peel as a sustainable source of functional pectin, aligning with the broader goals of valorizing agro-industrial by-products for food technology applications. The optimization analysis indicated that the maximum response was obtained at an electric field strength and solid-to-solvent ratio of 20 kV cm−1 and 1
30 ratio—yielded pectin with high purity (GalA > 75%), favorable methoxyl content (9%), and strong gelling properties (DE > 65%). These findings highlighted the potential of rambutan peel as a sustainable source of functional pectin, aligning with the broader goals of valorizing agro-industrial by-products for food technology applications. The optimization analysis indicated that the maximum response was obtained at an electric field strength and solid-to-solvent ratio of 20 kV cm−1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 (w/v), corresponding to the upper boundary of the investigated experimental range. This suggests that a stationary point was not fully captured within the studied domain, and the true optimum may lie beyond the current experimental limits.
30 (w/v), corresponding to the upper boundary of the investigated experimental range. This suggests that a stationary point was not fully captured within the studied domain, and the true optimum may lie beyond the current experimental limits.
3.2 Optimized condition of pectin yield
The analysis of pectin yield demonstrated that the quadratic regression model provided the best fit for describing the response behavior. As summarized in Table 1, the quadratic model showed a significant effect on yield (p < 0.0001), a non-significant lack of fit (p = 0.4278), and the lowest PRESS value of 0.4906, confirming its predictive accuracy compared to linear, 2FI, or cubic models. Normality of residuals was confirmed, as illustrated in Fig. 2. ANOVA results confirmed that the main effects of electric field strength (A), extraction time (B), and solid-to-solvent ratio (C), along with their quadratic terms (A2, B2, and C2), significantly influenced pectin yield (p ≤ 0.05). In contrast, interaction terms (AB, AC, and BC) were not significant. Model adequacy was further supported by the high coefficient of determination (R2 = 0.9870), a small gap between adjusted R2 (0.9752) and predicted R2 (0.9375), and a high adequate precision value (34.15), which exceeds the threshold of 4, confirming a strong signal-to-noise ratio.20 The resulting quadratic regression equation describing the yield response was:| Y = 3.66 + 0.6661X1 + 0.1962X2 + 0.0838X3 − 0.0363X1X2 + 0.0577X1X3 + 0.0201X2X3 − 0.2211X12 − 0.1342X22 + 0.0653X32 |
| Source | Yields | EW | MC | KGalA | DE | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Quadratic | Cubic | Quadratic | Cubic | Quadratic | Cubic | Quadratic | Cubic | Quadratic | Cubic | |
| a EW: equivalent weight; MC: methoxyl content; KGalA: galacturonic acid content; DE: degree of esterification. | ||||||||||
| Sequential p-value | <0.0001 | 0.7305 | <0.0001 | 0.3665 | <0.0001 | 0.3163 | <0.0001 | 0.3919 | <0.0001 | 0.4574 |
| Lack of Fit p-value | 0.4278 | 0.1361 | 0.4955 | 0.5662 | 0.4948 | 0.8221 | 0.5843 | 0.942 | 0.3763 | 0.2249 |
| R2 | 0.987 | 0.9903 | 0.979 | 0.9888 | 0.9816 | 0.9908 | 0.9824 | 0.9903 | 0.9616 | 0.9774 |
| Adjusted R2 | 0.9752 | 0.9692 | 0.9601 | 0.9644 | 0.9651 | 0.9708 | 0.9666 | 0.9693 | 0.9271 | 0.9283 |
| Predicted R2 | 0.9375 | 0.1636 | 0.9044 | 0.8114 | 0.9143 | 0.9643 | 0.9247 | 0.9836 | 0.8026 | −0.406 |
| PRESS | 0.4906 | 6.57 | 8325.9 | 16421.61 | 1.54 | 0.6427 | 80.43 | 17.52 | 6.47 | 46.05 |
| Adequate precision value | 34.15 | 25.51 | 24.93 | 25.49 | 18.84 | |||||
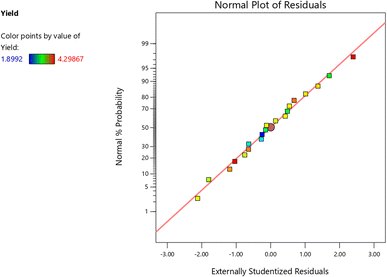
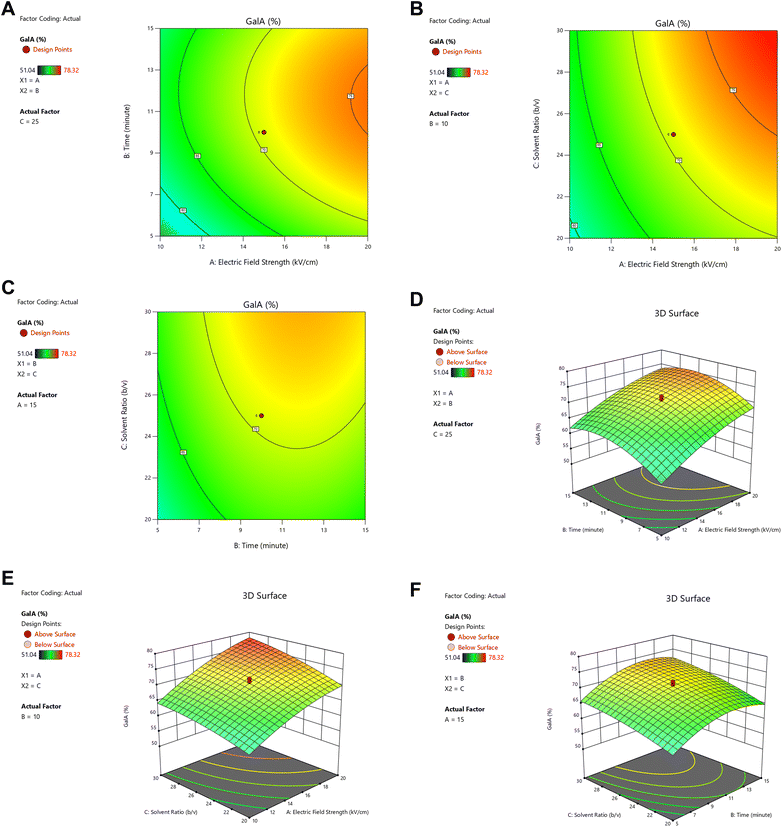
Contour and surface plots (Fig. 3) revealed that increases in electric field strength, extraction time, and solid-to-solvent ratio positively affected pectin yield. Stronger electric fields facilitated cell wall disintegration, enhancing solvent penetration and pectin release, which was consistent with previous findings on PEF-assisted extraction.15 Extended extraction time and higher solvent ratios further promoted protopectin hydrolysis and solubilization, although excessive treatment risked yield decline due to structural degradation.24 Overall, the combined analysis demonstrated that yield improvement primarily resulted from independent factor effects rather than factor interactions. The quadratic model not only fulfilled statistical assumptions but also aligned with previous studies on pectin optimization from tropical fruit peels,25 supporting its suitability for predicting and optimizing extraction conditions in sustainable food processing.
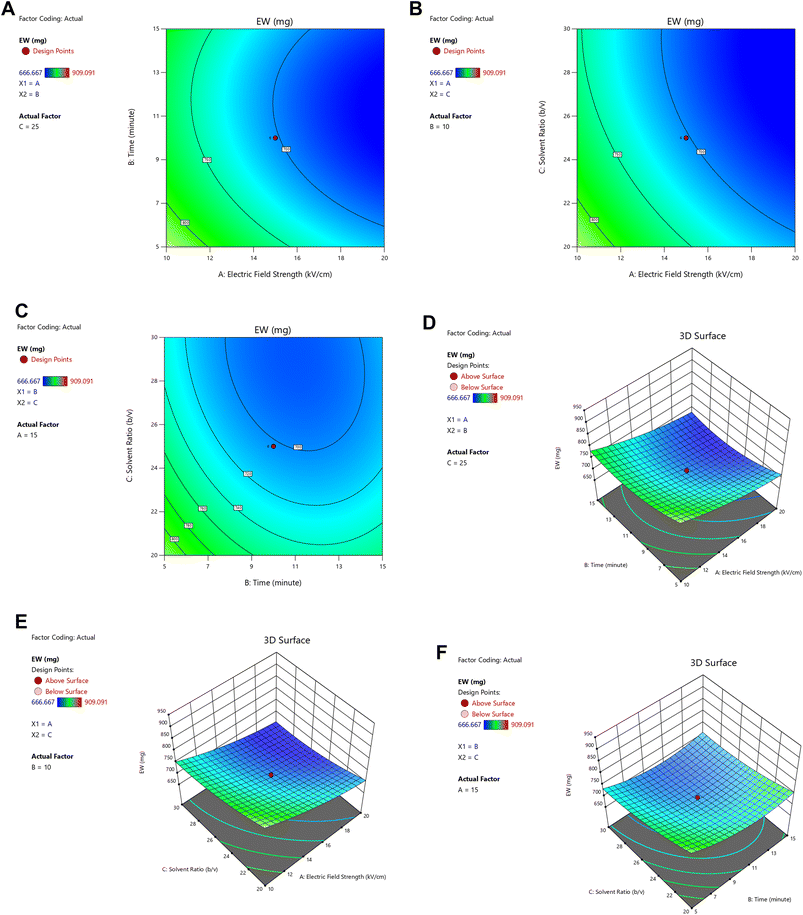
3.3 Optimized condition of equivalent weight
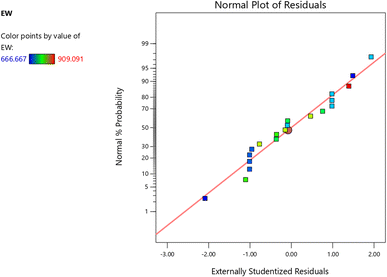
RSM analysis for pectin equivalent weight (EW) (Table 1) identified the quadratic model as most suitable, showing a significant effect on yield (p < 0.0001), non-significant lack of fit (p = 0.4955), high adjusted R2 (0.9601), close predicted R2 (0.9044), and lowest PRESS (8325.90), confirming superior predictive accuracy over other models.26 Normality of residuals was confirmed through the normal probability plot (Fig. 4), where points aligned closely with the reference line, indicating that the data met the normal distribution assumptions.27,28 Subsequent ANOVA showed that electric field strength, extraction time, and solid-to-solvent ratio significantly affected EW (p ≤ 0.05), while interactions were non-significant.20The model adequacy was validated by the high R2 value (0.9790), the minimal difference between adjusted R2 and predicted R2 (0.0557 < 0.2), and an adequate precision value of 25.51. The regression equation derived from the quadratic model was:
| Y = 702.12 − 55.07X1 − 21.40X2 − 32.55X3 + 3.57X1X2 + 5.75X1X3 + 9.62X2X3 + 16.00X12 + 34.13X22 + 18.53X32 |
The positive constant (702.12) indicated that in the absence of treatment factors, the baseline EW of pectin was within the acceptable range (600–800 mg) defined by IPPA,23 whilst the negative coefficients for main factors suggested that higher electric field strength, longer extraction time, and greater solvent ratio decreased EW, reflecting pectin degradation into smaller fragments and free galacturonic acid units, consistent with previous findings.29 Conversely, positive coefficients for quadratic and interaction terms indicated that moderate factor levels could stabilize or enhance EW. Contour and 3D surface plots (Fig. 5) further illustrated these relationships. Increasing electric field intensity promoted cell wall disruption and solvent penetration, which facilitated de-esterification and lowered EW.15 Similarly, higher solvent ratios increased solubilization but promoted de-esterification into pectic acid, reducing EW.1 Prolonged extraction time progressively reduced equivalent weight (EW) through hydrolysis and impurity release, yielding smaller molecular weight fractions and underscoring the trade-off between extraction efficiency and structural preservation.30 Overall, the quadratic model reliably described the response of equivalent weight, confirming that stronger electric fields, extended extraction times, and higher solvent ratios led to significant reductions in EW. These findings align with established mechanisms of pectin degradation under electro-permeabilization and acid hydrolysis and provide a robust statistical basis for optimizing extraction conditions to maintain EW within functional ranges for gel formation in sustainable food applications.
3.4 Optimized condition of methoxyl content
The response surface methodology (RSM) analysis for methoxyl content of pectin identified the quadratic model as the most suitable representation of the data. Fit summary results (Table 1) showed that both linear (p < 0.0001) and quadratic (p < 0.0001) models were significant, but the quadratic model was preferred due to its non-significant lack of fit (p = 0.4948), higher adjusted R2 (0.9651), and closer agreement with predicted R2 (0.9143), compared to other models.PRESS statistics confirmed this selection, as the quadratic model exhibited a low prediction error (1.54), supporting its superior predictive accuracy.20 Normality of residuals was verified by the normal probability plot (Fig. 6), where residuals closely followed the reference line, indicating that the assumption of normal distribution was met.27,28
ANOVA results further demonstrated that the main factor significantly influenced methoxyl content (p ≤ 0.05), while their interaction terms were not significant. Quadratic effects of electric field strength and extraction time were also significant, whereas the solvent ratio was not. Model adequacy was confirmed by the high R2 value (0.9816), minimal difference between adjusted and predicted R2 (0.0508 < 0.2), and adequate precision (24.93 > 4), reflecting robustness and reliability. The regression equation derived from the quadratic model was:
| Y = 8.03 + 0.7898X1 + 0.394X2 + 0.4174X3 − 0.0581X1X2 + 0.1356X1X3 + 0.0969X2X3 − 0.2425X12 − 0.5165X22 − 0.0781X32 |
The positive constant (8.03) indicated that in the absence of treatment factors, methoxyl content was within the functional range for pectin, confirming its baseline gel-forming capacity.31 Positive coefficients of main factors (X1, X2, and X3) suggested that increases in electric field strength, extraction time, and solvent ratio enhanced methoxyl content, while negative quadratic terms indicated that excessive factor levels could reduce the response. Contour and surface plots (Fig. 7) illustrated these relationships. Stronger electric fields disrupted cell walls, accelerating methanol release, which provided methyl groups for esterification with galacturonic acid, thereby increasing methoxylation.14 Higher solvent ratios improved pectin solubilization and promoted methyl ester formation, consistent with previous studies.1 Prolonged extraction times also increased esterification reactions, yielding higher methoxyl content, as reported by Lestari.31 The response surfaces generally formed plateau-like curves, indicating gradual increases rather than sharp optima and suggesting balanced interactions among the factors.20,30 Overall, the quadratic model adequately described the methoxyl content response, confirming that moderate increases in electric field strength, solvent ratio, and extraction time significantly enhanced methoxylation of pectin. These findings highlight the potential of pulsed electric field-assisted extraction in improving functional properties of pectin for sustainable food application.
3.5 Optimized condition of galacturonic acid
The analysis of galacturonic acid content in extracted pectin demonstrated that the quadratic model was the most appropriate for representing the response, as indicated by its significant sequential p-value (<0.0001), non-significant lack of fit (p = 0.5843), high adjusted R2 (0.9666), and close agreement with predicted R2 (0.9247). Although the cubic model showed the lowest PRESS value (Table 1), it was aliased and therefore unsuitable. The quadratic model fulfilled the statistical requirements with adequate precision (25.49), confirming its predictive reliability.20,32 Normality of residuals was confirmed using the normal probability plot (Fig. 8), where the residuals aligned with the reference line, indicating that they followed a normal distribution. ANOVA further revealed that electric field strength (PEF), extraction time, and solvent ratio, along with their quadratic terms, significantly influenced galacturonic acid levels (p ≤ 0.05), while their interactions were not significant. The positive regression coefficients of the main factors indicated that increasing electric field strength, extraction duration, and solvent ratio enhanced galacturonic acid release, consistent with previous findings that higher PEF intensities disrupt cell walls and facilitate pectin solubilization.15,33,34 Similarly, higher solvent ratios improved contact with pectin polymers, promoting bond cleavage,1 while prolonged extraction allowed more extensive hydrolysis of homogalacturonan chains.31 Conversely, negative quadratic coefficients suggested diminishing returns at excessive levels of these factors, consistent with the findings of Nieminen.29 Visualization through contour and 3D surface plots (Fig. 9) confirmed these trends, showing a plateau-like response surface where galacturonic acid content increased with treatment factors but reached a curvature indicating stabilization at higher levels. This response pattern suggests that optimization of PEF-assisted extraction lies in balancing electric field intensity, extraction time, and solvent ratio to maximize galacturonic acid purity, aligning with IPPA standards of >35% galacturonic acid for high-quality pectin.473.6 Optimized condition of esterification degree
The degree of esterification (DE) is a critical parameter influencing the gelling functionality of pectin.35,36 Model fitting (Table 1) through Design Expert 13 indicated that the quadratic model best represented the DE response, supported by its significant sequential p-value (<0.0001), non-significant lack of fit (p = 0.3763), high adjusted R2 (0.9271), close agreement with predicted R2 (0.8026), and the lowest PRESS value (6.47). These metrics confirm its predictive reliability, aligning with the recommendation that models with minimal PRESS and non-significant lack of fit are most appropriate.20 Residual normality analysis (Fig. 10) further confirmed the adequacy of this model, as data points closely followed the normal distribution line, suggesting no deviation from statistical assumptions.37 ANOVA results demonstrated that electric field strength, extraction time, and solid-to-solvent ratio, along with their quadratic terms, significantly influenced DE (p ≤ 0.05). Moreover, the interactions also showed significant effects, indicating synergistic contributions of the extraction factors. The regression model (Y = 64.53 + 0.8632X1 + 0.7023X2 + 0.3038X3 − 0.3213X1X2 + 0.3434X1X3 + 0.4531X2X3 − 0.4107X12 − 0.6277X22 + 0.3158X32) confirmed that most coefficients were positive, meaning that higher electric field strength, longer extraction time, and greater solvent ratios enhanced esterification. Negative quadratic coefficients suggested diminishing returns at excessive levels, consistent with the findings in ref. 29. Adequate precision (18.84) exceeded the threshold of 4, confirming strong signal-to-noise performance.32 As portrayed by Fig. 11, response surface and contour plots revealed that DE increased with rising electric field strength and extraction duration, reflecting improved cell wall disruption and methanol-mediated esterification.38 Similarly, higher solvent ratios facilitated methanol release and esterification.1 The plateau-like 3D surfaces suggested that while DE improved with treatment intensification, further increases approached stabilization rather than decline, likely because experimental conditions remained within the optimal range before de-esterification dominated.36 Overall, these findings demonstrate that pulsed electric field-assisted extraction effectively enhances pectin esterification, yielding DE values consistent with high-quality functional pectin standards.3.7 Response optimization using RSM-CCD
Optimization of pectin extraction from rambutan peel using pulsed electric field (PEF) and citric acid solvent was conducted through RSM-CCD with the aid of statistical software (Design Exper t 13), considering electric field strength, extraction time, and solvent ratio as the main factors. The optimization aimed to maximize yield, methoxyl content, galacturonic acid, and degree of esterification (DE), while maintaining equivalent weight within the acceptable IPPA range (600–800 mg). Among these, methoxyl content and DE were assigned the highest importance, as both parameters determine the functional gelling properties and classification of pectin.35,39 The software generated 36 possible solutions, with the optimal point determined based on desirability values, which range from 0 to 1 and indicate the suitability of variable combinations for achieving target responses. As shown in Table 2, the highest desirability value (0.995) was achieved at an electric field strength of 20 kV cm−1, extraction time of 12.7 minutes, and solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 (w/v), yielding predicted values of 4.4% for yield, 660.3 mg for equivalent weight, 9.1% for methoxyl content, 78.5% for galacturonic acid, and 66.2% for DE. These results confirm the system's recommendation that optimal extraction conditions combine high electric field intensity and adequate solvent ratio within moderate extraction times, consistent with previous findings that desirability values close to 1 represent the most reliable optimization outputs.
30 (w/v), yielding predicted values of 4.4% for yield, 660.3 mg for equivalent weight, 9.1% for methoxyl content, 78.5% for galacturonic acid, and 66.2% for DE. These results confirm the system's recommendation that optimal extraction conditions combine high electric field intensity and adequate solvent ratio within moderate extraction times, consistent with previous findings that desirability values close to 1 represent the most reliable optimization outputs.
| EFS | ET | SR | Y | EW | MC | Gal. | DE | Des. |
|---|---|---|---|---|---|---|---|---|
| a EFS: electric field strength (kV cm−1); ET: extraction time (min); SR: solvent ratio (w/v); Y: yield (%); EW: equivalent weight (mg); MC: methoxyl content (%); Gal: galacturonic acid content (%); DE: degree of esterification (%); Des: desirability. | ||||||||
| 20.0 | 12.7 | 30.0 | 4.4 | 660.3 | 9.1 | 78.5 | 66.2 | 0.995 |
| 20.0 | 12.8 | 30.0 | 4.4 | 661.1 | 9.1 | 78.5 | 66.2 | 0.994 |
| 20.0 | 12.6 | 29.9 | 4.4 | 659.6 | 9.1 | 78.5 | 66.2 | 0.993 |
| 19.9 | 12.0 | 30.0 | 4.4 | 657.2 | 9.1 | 78.6 | 66.2 | 0.992 |
| 20.0 | 11.4 | 30.0 | 4.4 | 655.2 | 9.1 | 78.6 | 66.1 | 0.990 |
| 19.0 | 12.8 | 30.0 | 4.3 | 664.3 | 9.1 | 77.8 | 66.2 | 0.981 |
| 20.0 | 13.5 | 29.3 | 4.3 | 663.6 | 9.0 | 77.9 | 66.0 | 0.972 |
3.8 Verification of optimum extraction conditions
Verification of the optimum extraction conditions predicted by Design Expert 13 was conducted by comparing wet-lab results with the predicted values. All experimental outcomes fell within the 95% prediction interval (PI), confirming that the model was accurate and reliable for application under real conditions. Table 3 shows the pectin properties obtained from the optimum condition using PEF (20 kV cm−1, 12.7 min, solvent ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30), which were 4.4% yield, 677.83 equivalent weight, 8.9% methoxyl content, 76.49% galacturonic acid, and 66.05% degree of esterification. As shown in Table 4, conventional extraction (HCl 0.1 N, 80 °C, 2 h) provided higher yield than PEF-extracted pectin, which demonstrated lower yield (4.4% vs. 7.35%) but superior quality in terms of methoxyl content, galacturonic acid, and degree of esterification. The relatively lower yield in this study is likely due to the moderate electric field strength (20 kV cm−1), as previous studies on citrus peel using higher intensities (30 kV cm−1) achieved higher yields,15 whereas lower intensities resulted in reduced yields.14 Equivalent weight under PEF (677.83 mg) was lower than conventional extraction(748.97 mg), suggesting that PEF facilitated greater contact between pectin and the acid solvent through cell disintegration, leading to partial hydrolysis and reduced equivalent weight.34,40 Conversely, methoxyl content under PEF (8.9%) exceeded conventional extraction (6.17%). This categorizes the extracted pectin as High Methoxyl Pectin (HMP), consistent with the International Pectin Producers Association (IPPA) classification.23 Similarly, galacturonic acid content was higher under PEF (76.49%) than conventional extraction (58.51%), reflecting greater purity and compliance with IPPA standards (>35%).15 The elevated galacturonic acid content also aligns with findings that HIPEF extraction produces pectins richer in galacturonic acid and lower in neutral sugars compared to conventional methods.15 Degree of esterification followed the same trend, with PEF pectin reaching 66.05% versus 59.83% under conventional extraction, further confirming its classification as HMP.35,36 PEF treatment exerts a dual effect on pectin. While partial depolymerization may occur, as reflected by reduced equivalent weight, electroporation primarily enhances mass transfer and preserves native esterified carboxyl groups. Consequently, the high methoxyl content and degree of esterification are attributed to improved release and retention of naturally esterified pectin rather than PEF-induced de-esterification.
30), which were 4.4% yield, 677.83 equivalent weight, 8.9% methoxyl content, 76.49% galacturonic acid, and 66.05% degree of esterification. As shown in Table 4, conventional extraction (HCl 0.1 N, 80 °C, 2 h) provided higher yield than PEF-extracted pectin, which demonstrated lower yield (4.4% vs. 7.35%) but superior quality in terms of methoxyl content, galacturonic acid, and degree of esterification. The relatively lower yield in this study is likely due to the moderate electric field strength (20 kV cm−1), as previous studies on citrus peel using higher intensities (30 kV cm−1) achieved higher yields,15 whereas lower intensities resulted in reduced yields.14 Equivalent weight under PEF (677.83 mg) was lower than conventional extraction(748.97 mg), suggesting that PEF facilitated greater contact between pectin and the acid solvent through cell disintegration, leading to partial hydrolysis and reduced equivalent weight.34,40 Conversely, methoxyl content under PEF (8.9%) exceeded conventional extraction (6.17%). This categorizes the extracted pectin as High Methoxyl Pectin (HMP), consistent with the International Pectin Producers Association (IPPA) classification.23 Similarly, galacturonic acid content was higher under PEF (76.49%) than conventional extraction (58.51%), reflecting greater purity and compliance with IPPA standards (>35%).15 The elevated galacturonic acid content also aligns with findings that HIPEF extraction produces pectins richer in galacturonic acid and lower in neutral sugars compared to conventional methods.15 Degree of esterification followed the same trend, with PEF pectin reaching 66.05% versus 59.83% under conventional extraction, further confirming its classification as HMP.35,36 PEF treatment exerts a dual effect on pectin. While partial depolymerization may occur, as reflected by reduced equivalent weight, electroporation primarily enhances mass transfer and preserves native esterified carboxyl groups. Consequently, the high methoxyl content and degree of esterification are attributed to improved release and retention of naturally esterified pectin rather than PEF-induced de-esterification.
| Response | Prediction mean | 95% PI low | Actual data | 95% PI high |
|---|---|---|---|---|
| a EW: equivalent weight (mg); MC: methoxyl content (%); Gal: galacturonic acid content (%); DE: degree of esterification (%). | ||||
| Yield (%) | 43.711 | 41.528 | 44.000 | 45.893 |
| EW (mg) | 660.242 | 631.058 | 677.830 | 689.427 |
| MC (%) | 91.407 | 87.486 | 89.000 | 95.329 |
| Gal (%) | 775.177 | 755.613 | 764.900 | 814.742 |
| DE (%) | 662.118 | 654.468 | 660.500 | 669.769 |
| Characteristic | Optimum pectin (PEF) | Conventional pectin |
|---|---|---|
| Yield (%) | 4.40 ± 0.09 | 7.35 ± 0.02 |
| Equivalent weight (mg) | 677.83 ± 0.19 | 748.97 ± 0.14 |
| Methoxyl content (%) | 8.90 ± 0.13 | 6.17 ± 0.03 |
| Galacturonic acid (%) | 76.49 ± 0.72 | 58.51 ± 0.22 |
| Degree of esterification (%) | 66.05 ± 0.33 | 59.83 ± 0.14 |
Overall, while yield under PEF extraction was slightly lower than conventional extraction, the higher methoxyl content, galacturonic acid concentration, and degree of esterification demonstrate that PEF not only produces functionally superior pectin but also yields material of higher purity, underscoring its potential as a sustainable extraction technology.
3.9 Physicochemical and structural characteristics
In this study, the physicochemical and structural characteristics of the optimum pectin obtained from rambutan peel using Pulsed Electric Field (PEF) extraction were compared with those extracted conventionally. The moisture content of PEF-extracted pectin (9.78%) was slightly higher than that of the conventional method (9.51%), which may be attributed to the lower temperature and shorter duration applied during PEF treatment. Higher temperatures and longer extraction times, as in conventional methods, can shorten pectin chains and reduce the amount of water entrapped within the polymer.41,42 Despite this difference, the moisture content of both samples complied with the IPPA standard, which sets the maximum limit at 12%. The ash content of PEF-extracted pectin (4.98%) was nearly twice that of conventional pectin (2.59%), but remained within the acceptable IPPA threshold (<10%). The higher ash content in PEF samples likely reflects greater mineral release from disrupted cell walls, where divalent ions such as magnesium and calcium are displaced during acid hydrolysis.The relationship between moisture and ash content in PEF-extracted pectin is rooted in the intensity of cell wall disintegration. The higher ash content (4.98% in PEF vs. 2.59% in conventional extraction) is attributed to the enhanced release of minerals (such as calcium and magnesium) from the cell walls due to electroporation. However, the slightly higher moisture (9.78%) is linked to the lower temperatures and shorter processing times of PEF. High temperatures in conventional methods tend to shorten pectin chains and reduce the structure's ability to entrap water. Thus, PEF creates a more porous and disrupted structure (increasing ash via mineral release) while preserving the polymer's integrity at lower temperatures (increasing moisture via water entrapment). This finding aligns with reports that increasing solvent intensity during extraction promotes mineral solubilization, thereby elevating ash content.19
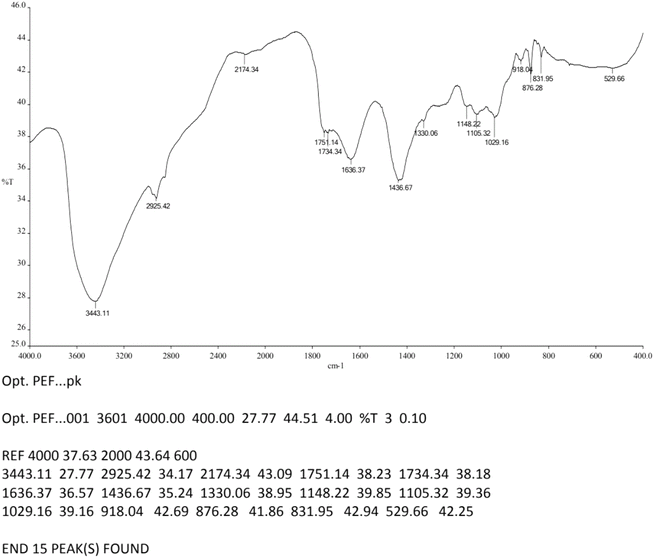
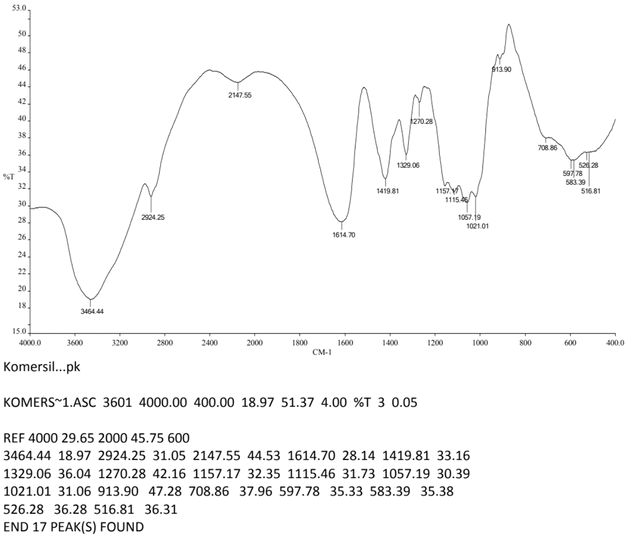
Functional group analysis using FTIR confirmed that the PEF-extracted pectin exhibited characteristic absorption peaks corresponding to hydroxyl (–OH), methyl (–CH3), carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), and ether (–O–) groups (Fig. 12), consistent with the spectral signatures of commercial citrus pectin (Fig. 13) and literature reports.43,44 These peaks validate the structural integrity of the extracted pectin and its classification as a high-quality polysaccharide.
O), and ether (–O–) groups (Fig. 12), consistent with the spectral signatures of commercial citrus pectin (Fig. 13) and literature reports.43,44 These peaks validate the structural integrity of the extracted pectin and its classification as a high-quality polysaccharide.
It is confirmed by the result that, the peak range for carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) groups at 1636.37–1751.14 cm−1 is critical. Specifically, the vibration near 1735 cm−1 signifies esterified carboxyl groups. This spectral evidence directly supports the high degree of esterification (66.05%) found in the chemical analysis. When it comes to the structural integrity, the presence of the hydroxyl (–OH) peak at 3443.11 cm−1 and ether (–O–) bonds at 1148.22 cm−1 confirms that the PEF treatment, despite its intensity, did not destroy the fundamental polygalacturonic acid backbone of the pectin.
O) groups at 1636.37–1751.14 cm−1 is critical. Specifically, the vibration near 1735 cm−1 signifies esterified carboxyl groups. This spectral evidence directly supports the high degree of esterification (66.05%) found in the chemical analysis. When it comes to the structural integrity, the presence of the hydroxyl (–OH) peak at 3443.11 cm−1 and ether (–O–) bonds at 1148.22 cm−1 confirms that the PEF treatment, despite its intensity, did not destroy the fundamental polygalacturonic acid backbone of the pectin.
| No. | Wavenumber (cm−1) | Functional group | |
|---|---|---|---|
| Rambutan peel pectin | Commercial pectin | ||
| 1 | 3443.11 | 3464.44 | –OH |
| 2 | 2925.42 | 2924.25 | CH |
| 3 | 1636.37–1751.14 | 1614.7 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
| 4 | 1436.67 | 1419.81 | –CH3 |
| 5 | 1148.22 | 1157.17 | –O– |
Finally, SEM micrographs demonstrated that PEF treatment caused visible disruption of the rambutan peel cell walls, with cracks and pores clearly evident compared to the intact structure of untreated samples (Fig. 14). This morphological alteration highlights the electroporation effect of PEF, where pulsed electric fields create transmembrane potential differences that weaken cell wall integrity and facilitate mass transfer.45 Such structural damage corroborates the enhanced release of intracellular pectin observed in this study. Taken together, these results indicate that while PEF extraction yields pectin with slightly higher moisture and ash contents, it preserves essential structural features and facilitates improved cell disruption, thereby producing functionally and structurally comparable pectin compared to that obtained by conventional methods, with potential for higher efficiency and sustainability.
4 Conclusion
Based on this study, three main conclusions can be drawn. First, electric field strength, extraction time, and solid-to-solvent ratio significantly influenced pectin extraction from rambutan peel. Preliminary OFAT trials established effective working ranges (10–20 kV cm−1, 5–15 min, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20–1
20–1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 w/v), which were subsequently refined using RSM. Second, the optimal conditions predicted by design-expert (20 kV cm−1, 12.69 min, 1
30 w/v), which were subsequently refined using RSM. Second, the optimal conditions predicted by design-expert (20 kV cm−1, 12.69 min, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 w/v) yielded pectin with favorable physicochemical characteristics, including a yield of 4.4%, a equivalent weight of 677.83 mg, a methoxyl content of 8.9%, a galacturonic acid content of 76.49%, and a degree of esterification of 66.05%, classifying it as high-methoxyl pectin. Third, compositional analysis confirmed acceptable moisture (9.78%) and ash content (4.98%), while FTIR spectra verified characteristic functional groups consistent with commercial pectin. SEM observations further demonstrated substantial cell wall disruption following PEF treatment, confirming effective electroporation-driven mass transfer enhancement.
30 w/v) yielded pectin with favorable physicochemical characteristics, including a yield of 4.4%, a equivalent weight of 677.83 mg, a methoxyl content of 8.9%, a galacturonic acid content of 76.49%, and a degree of esterification of 66.05%, classifying it as high-methoxyl pectin. Third, compositional analysis confirmed acceptable moisture (9.78%) and ash content (4.98%), while FTIR spectra verified characteristic functional groups consistent with commercial pectin. SEM observations further demonstrated substantial cell wall disruption following PEF treatment, confirming effective electroporation-driven mass transfer enhancement.
Although the PEF-assisted method produced a lower yield than conventional extraction, it offered superior pectin quality and clear sustainability advantages. Future challenges lie in process scale-up, economic feasibility, and standardization. Successful industrial translation will require improved yield–energy balance, consistent feedstock management, regulatory compliance, and validation of functional performance across food, packaging, and biomedical applications, supported by comprehensive life-cycle assessments. While this study successfully optimized PEF-assisted pectin extraction in terms of yield and some of the key parameters, comprehensive physicochemical characterization was beyond its scope and will be addressed in ongoing and future studies. A limitation of this study is that the optimal conditions for electric field strength (20 kV cm−1) and solid-to-solvent ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 w/v) were identified at the upper boundary of the experimental design. Consequently, the reported value represented an optimum within the investigated range. Future studies should extend the experimental domains to determine the global optimum.
30 w/v) were identified at the upper boundary of the experimental design. Consequently, the reported value represented an optimum within the investigated range. Future studies should extend the experimental domains to determine the global optimum.
Author contributions
Luqman Agung Wicaksono: conceptualization, project administration, writing –original draft, and supervision. Ganes Aurora Santoso: investigation, conceptualization, data curation, formal analysis, and writing – original draft. Anugerah Dany Priyanto: project administration, funding acquisition, conceptualization, formal analysis, and supervision. Muhammad Alfid Kurnianto: project administration. Teeradate Kongpichitchoke: project administration. Muhammad Rosyid Wardianto: formal analysis, writing – original draft, and writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The article includes the data that were utilized for the study.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fb00658a.
Acknowledgements
The authors would like to acknowledge LPPM UPN “Veteran” Jawa Timur for providing support under the scheme of international research collaboration.References
- G. A. Santoso, L. A. Wicaksono, M. A. Kurnianto, H. Munarko, A. Y. T. Putra and A. D. Priyanto, Pectin extraction from Nephelium lappaceum (Binjai variety) peel using pulsed electric field, Konversi, 2025, 14(1), 7–11, DOI:10.20527/k.v14i1.22031.
- BPS, “[BPS] Badan Pusat Statistik. (2024). Produksi Tanaman Buah-Buahan 2021-2023. Diakses Tanggal 22 Maret 2024.Pdf,” 2024 Search PubMed.
- M. C. N. Picot-Allain, B. Ramasawmy and M. N. Emmambux, Extraction, Characterisation, and Application of Pectin from Tropical and Sub-Tropical Fruits: A Review, Food Rev. Int., 2022, 38(3), 282–312, DOI:10.1080/87559129.2020.1733008.
- L. N. Gerschenson, E. N. Fissore, A. M. Rojas, A. M. Idrovo Encalada, E. F. Zukowski and R. A. Higuera Coelho, Pectins obtained by ultrasound from agroindustrial by-products, Food Hydrocoll., 2021, 118, 106799, DOI:10.1016/j.foodhyd.2021.106799.
- R. Sakooei-Vayghan, S. H. Peighambardoust, J. Hesari and D. Peressini, Effects of osmotic dehydration (with and without sonication) and pectin-based coating pretreatments on functional properties and color of hot-air dried apricot cubes, Food Chem., 2020, 311, 125978, DOI:10.1016/j.foodchem.2019.125978.
- T. Belwal, et al., Recent advances in scaling-up of non-conventional extraction techniques: Learning from successes and failures, TrAC, Trends Anal. Chem., 2020, 127, 115895, DOI:10.1016/j.trac.2020.115895.
- S. P. Ishwarya and P. Nisha, Headway in Supercritical Extraction of Fragrances and Colors, Elsevier, 2020, DOI:10.1016/b978-0-08-100596-5.22677-2.
- S. Bachtler and H. J. Bart, Increase the yield of bioactive compounds from elder bark and annatto seeds using ultrasound and microwave assisted extraction technologies, Food Bioprod. Process., 2021, 125, 1–13, DOI:10.1016/j.fbp.2020.10.009.
- M. Vinatoru, T. J. Mason and I. Calinescu, Ultrasonically assisted extraction (UAE) and microwave assisted extraction (MAE) of functional compounds from plant materials, TrAC, Trends Anal. Chem., 2017, 97, 159–178, DOI:10.1016/j.trac.2017.09.002.
- P. Karbuz and N. Tugrul, Microwave and ultrasound assisted extraction of pectin from various fruits peel, J. Food Sci. Technol., 2021, 58(2), 641–650, DOI:10.1007/s13197-020-04578-0.
- G. Ntourtoglou et al., “Pulsed Electric Field Extraction of α and β-Acids From Pellets of Humulus lupulus (Hop),” 2020. DOI:10.3389/fbioe.2020.00297.
- O. Parniakov, E. Roselló-Soto, F. J. Barba, N. Grimi, N. Lebov+ka and E. Vorobiev, New approaches for the effective valorization of papaya seeds: Extraction of proteins, phenolic compounds, carbohydrates, and isothiocyanates assisted by pulsed electric energy, Food Res. Int., 2015, 77, 711–717, DOI:10.1016/j.foodres.2015.03.031.
- I. Efthymiopoulos, et al., Influence of solvent selection and extraction temperature on yield and composition of lipids extracted from spent coffee grounds, Ind. Crops Prod., 2018, 119, 49–56, DOI:10.1016/j.indcrop.2018.04.008.
- C. F. De Oliveira, D. Giordani, P. D. Gurak, F. Cladera-Olivera and L. D. F. Marczak, Extraction of pectin from passion fruit peel using moderate electric field and conventional heating extraction methods, Innov. Food Sci. Emerg. Technol., 2015, 29, 201–208, DOI:10.1016/j.ifset.2015.02.005.
- Y. Du, et al., High-intensity pulsed electric field-assisted acidic extraction of pectin from citrus peel: Physicochemical characteristics and emulsifying properties, Food Hydrocoll., 2024, 146, 109291, DOI:10.1016/j.foodhyd.2023.109291.
- K. A. Mohamad Said and M. A. Mohamed Amin, Overview on the Response Surface Methodology (RSM) in Extraction Processes, J. Appl. Sci. Process Eng., 2016, 2(1), 8–17, DOI:10.33736/jaspe.161.2015.
- R. H. Myers, D. C. Montgomery, and C. M. Anderson-Cook, Response Surface Methodology: Process and Product Optimization Using Designed Experiments, edn. 3rd, John Wiley & Sons, Inc., Hoboken, New Jersey, 2009, DOI:10.3109/9781439822463.187.
- M. Sharma, et al., Optimization of ultrasonication assisted extraction of Aegle marmelos fruit shell nano polysaccharide and evaluation of photocatalytic dye reduction and edible coating for fresh-cut fruits, Food Chem.:X, 2024, 24, 101895, DOI:10.1016/j.fochx.2024.101895.
- Y. Laiya, M. Lasindrang, and Z. Antuli, “Physicochemical Characteristics of Pectin from Rambutan Skin Waste Nephelium Lappaceum Linn,” 2020 Search PubMed.
- S. J. M. Breig and K. J. K. Luti, Response surface methodology: A review on its applications and challenges in microbial cultures, Mater. Today Proc., 2021, 42, 2277–2284, DOI:10.1016/j.matpr.2020.12.316.
- S. Anderson, S. Aldana, M. Beggs, J. Birkey, A. Conquest, R. Conway, T. Hemminger, J. Herrick, C. Hurley, C. Ionita, J. Longbind, S. McMaignal, A. Milu, T. Mitchell, K. Nanke, A. Perez, M Phelps, J. Reitz, A. Salazar and S. Zerr, Determination of Fat, Moisture, and Protein in Meat and Meat Products by Using the FOSS FoodScan Near-Infrared Spectrophotometer with FOSS Artificial Neural Network Calibration Model and Associated Database: Collaborative Study, J. AOAC Int., 2007, 90, 1073–1083 CrossRef CAS.
- T. Lindriati, M. Belgis, M. Fauzi, Q. E. Juwita and S. Sakinah, Ultrasound Assisted Extraction and Characterization of Pectin from Red Dragon Fruit (Hylocereus Polyrhizus) Peels, J. appl. agricultural sci. technol., 2023, 7(2), 91–99, DOI:10.55043/jaast.v7i2.76.
- IPPA, Pectin in Organic Food Products, Int. Pectin Prod. Assoc., 2020, 2020 Search PubMed.
- M. Y. Sayah, et al., Yield, esterification degree and molecular weight evaluation of pectins isolated from orange and grapefruit peels under different conditions, PLoS One, 2016, 11(9), 1–16, DOI:10.1371/journal.pone.0161751.
- E. Girma and M. T. Worku, Extraction and Characterization of Pectin From Selected Fruit Peel Waste, Int. J. Sci. Res. Publ., 2016, 6(2), 447–454 Search PubMed , [Online]. Available: www.ijsrp.org.
- T. Estiasih, K. Ahmadi, E. Ginting and D. Kurniawati, Yield Optimization of Lecithin Extraction of Anjasmoro Variety Soybean Oil by Water Degumming, J. Teknol. dan Ind. Pangan, 2013, 24(1), 97–104, DOI:10.6066/jtip.2013.24.1.97.
- Y. Yakubu, Z. Q. Aliyu, A. Usman and O. Evans, Split-plot Central Composite Experimental Design Method for Optimization of Cake Height to Achieve desired Texture, Niger. J. Basic Appl. Sci., 2021, 28(1), 30–39, DOI:10.4314/njbas.v28i1.5.
- M. J. Anderson and S. . Kraber, Improving Your DOE - Analysis with Response Transformations, J. Plast. Film Sheeting, 2022, 15–20, DOI:10.1177/87560879211069688.
- P. Nieminen, Application of Standardized Regression Coefficient in Meta-Analysis, BioMedInformatics, 2022, 2(3), 434–458, DOI:10.3390/biomedinformatics2030028.
- S. K. Behera, H. Meena, S. Chakraborty and B. C. Meikap, Application of response surface methodology (RSM) for optimization of leaching parameters for ash reduction from low-grade coal, Int. J. Min. Sci. Technol., 2018, 28(4), 621–629, DOI:10.1016/j.ijmst.2018.04.014.
- A. P. Lestari, J. Jariyah and D. F. Rosida, CHARACTERIZATION OF PECTIN FROM EXTRACTION FROM SOYBEAN HULLS (Glycine max. L) USING MICROWAVE ASSISTED EXTRACTION METHOD (Study of Electric Power and Extraction Time), Universitas Pembangunan Nasional “Veteran” Jawa Timur, 2023 Search PubMed , [Online]. Available:https://repository.upnjatim.ac.id/13803/9/17033010030.-cover.pdf.
- Y. Variyana, R. S. C. Muchammad and M. Mahfud, Box-behnken design for the optimization using solvent-free microwave gravity extraction of garlic oil from Allium sativum L, IOP Conf. Ser. Mater. Sci. Eng., 2019, 673, 012005, DOI:10.1088/1757-899X/673/1/012005.
- J. N. Sauceda-Gálvez, M. Tió-Coma, M. Martinez-Garcia, M. M. Hernández-Herrero, R. Gervilla and A. X. Roig-Sagués, Effect of single and combined UV-C and ultra-high pressure homogenisation treatments on inactivation of Alicyclobacillus acidoterrestris spores in apple juice, Innov. Food Sci. Emerg. Technol., 2020, 60, 102299, DOI:10.1016/j.ifset.2020.102299.
- M. Gavahian, Y. H. Chu and S. Sastry, Extraction from Food and Natural Products by Moderate Electric Field: Mechanisms, Benefits, and Potential Industrial Applications, Compr. Rev. Food Sci. Food Saf., 2018, 17(4), 1040–1052, DOI:10.1111/1541-4337.12362.
- R. K. Shaha, Y. N. A. P. Punichelvana and A. Afandi, Optimized Extraction Condition and Characterization of Pectin from Kaffir Lime (Citrus hystrix), Res. J. Agric. For. Sci., 2013, 1(2), 1–11 Search PubMed.
- Y. S. Duwee, P. L. Kiew and W. M. Yeoh, Multi-objective optimization of pectin extraction from orange peel via response surface methodology: yield and degree of esterification, J. Food Meas. Charact., 2022, 16(2), 1710–1724, DOI:10.1007/s11694-022-01305-5.
- L. González-Ceballos, et al., A simple one-pot determination of both total phenolic content and antioxidant activity of honey by polymer chemosensors, Food Chem., 2021, 342, 128300, DOI:10.1016/j.foodchem.2020.128300.
- D. Redondo, M. E. Venturini, E. Luengo, J. Raso and E. Arias, Pulsed electric fields as a green technology for the extraction of bioactive compounds from thinned peach by-products, Innov. Food Sci. Emerg. Technol., 2018, 45, 335–343, DOI:10.1016/j.ifset.2017.12.004.
- R. Ciriminna, et al., Controlling the Degree of Esterification of Citrus Pectin for Demanding Applications by Selection of the Source, ACS Omega, 2017, 2(11), 7991–7995, DOI:10.1021/acsomega.7b01109.
- M. Kazemi, F. Khodaiyan, M. Labbafi, S. Saeid Hosseini and M. Hojjati, Pistachio green hull pectin: Optimization of microwave-assisted extraction and evaluation of its physicochemical, structural and functional properties, Food Chem., 2019, 271, 663–672, DOI:10.1016/j.foodchem.2018.07.212.
- J. Chen, et al., Extraction temperature is a decisive factor for the properties of pectin, Food Hydrocoll., 2021, 112, 106160, DOI:10.1016/j.foodhyd.2020.106160.
- D. Silsia, M. Febreini and L. Susanti, Rendemen and characteristics of pektins red dragon fruit leather (Hylocereus costaricensis) with the difference in extraction method and time, J. Agroindustri, 2021, 11(2), 120–132, DOI:10.31186/j.agroindustri.11.2.120-132.
- D. L. Pavia, G. M. Lampman, G. S. Kriz, and J. R. Vyvyan, in Introduction to Spectroscopy, Department of Chemistry Western Washington University, Belingham, Washington, edn. 4th Fourt., no. 1, 2009, DOI:10.3917/popu.p1977.32n1.0034.
- A. Kozioł, K. Środa-Pomianek, A. Górniak, A. Wikiera, K. Cyprych and M. Malik, Structural Determination of Pectins by Spectroscopy Methods, Coatings, 2022, 12, 546, DOI:10.3390/coatings12040546.
- R. N. Arshad, et al., Electrical systems for pulsed electric field applications in the food industry: An engineering perspective, Trends Food Sci. Technol., 2020, 104, 1–13, DOI:10.1016/j.tifs.2020.07.008.
- M. A. Sotelo-Turban, et al., Dietary supplementation of conjugated linoleic acid in adult Creole goats: Effects on weight gain, carcass yield and meat quality, Emir. J. Food Agric., 2025, 37, 1–9 CrossRef.
- C. G. Nadar, A. Arora and Y. Shastri, Sustainability challenges and opportunities in pectin extraction from fruit waste, ACS Eng. Au, 2022, 2(2), 61–74 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |