Open Access Article
Open Access ArticleColored avocado (Persea americana Mill.) seed extract using a natural deep eutectic solvent for potential sustainable food coloring applications: towards a circular bioeconomy
Lourdes M. Orejuela-Escobar *abcde,
Carolina Tituaña
*abcde,
Carolina Tituaña fg,
Arleth Gualle
fg,
Arleth Gualle ah,
Andrea C. Landázuri
ah,
Andrea C. Landázuri abcd,
Edgar J. Guerrón-Orejuela
abcd,
Edgar J. Guerrón-Orejuela ei,
Emilia Moralesj,
Verónica Castañeda
ei,
Emilia Moralesj,
Verónica Castañeda j and
Andrés Caicedo
j and
Andrés Caicedo cjkl
cjkl
aUniversidad San Francisco de Quito USFQ, Colegio de Ciencias e Ingenierías, Applied Circular Engineering & Simulation Group (GICAS), Chemical Engineering Department, Diego de Robles y Vía Interoceánica, P.O. Box 17-0901, Quito, Ecuador. E-mail: lorejuela@emerito.usfq.edu.ec
bUniversidad San Francisco de Quito USFQ, Instituto de Investigaciones Biológicas y Ambientales (Biósfera), Diego de Robles y Vía Interoceánica, P.O. Box 17-0901, Quito, Ecuador
cUniversidad San Francisco de Quito USFQ, Instituto de Investigaciones en Biomedicina iBioMed, Diego de Robles y Vía Interoceánica, P.O. Box 17-0901, Quito, Ecuador
dUniversidad San Francisco de Quito USFQ, Institute for Energy and Materials, Diego de Robles y Vía Interoceánica, P.O. Box 17-0901, Quito, Ecuador
eGeocircular Consulting Group, Tampa, FL 33617, USA
fDepartamento de Ingeniería en Biotecnología, Universidad San Francisco de Quito USFQ, Diego de Robles y Vía Interoceánica, P.O. Box 17-0901, Quito, Ecuador
gInstituto Superior Tecnológico 17 de Julio, Hacienda San José Road, Yachay Polygon, Urcuquí, Ecuador
hChimie des Interactions Plasma-Surface (ChIPS), Research Institute for Materials Science and Engineering, University of Mons, 7000 Mons, Belgium
iSchool of Geosciences, University of South Florida, Tampa, FL 33620, USA
jUniversidad San Francisco de Quito USFQ, Colegio de Ciencias de la Salud, Escuela de Medicina, Quito, Ecuador
kMito-Act Research Consortium, Quito, Ecuador
lSistemas Médicos SIME, Universidad San Francisco de Quito USFQ, Quito, Ecuador
First published on 27th February 2026
Abstract
Avocado (Persea americana Mill.) production and consumption are rapidly increasing worldwide, with the global yield expected to reach 12 million tons by 2030. This growth generates substantial amounts of agro-industrial residues, mainly seeds and peels, which can pose environmental issues but also represent a rich source of bioactive compounds. Valorizing these byproducts through cascade biorefineries and green processing methods supports the sustainable production of high value-added bioproducts and fosters a circular bioeconomy. This study explores the extraction of natural colorants from avocado seeds as potential eco-friendly substitutes for synthetic orange-reddish food colorants. Colored avocado seed extracts (CASEs) from Fuerte and Hass cultivars were produced using hot distilled water, 70% ethanol (reflux), and glycerol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sodium acetate (Gly
sodium acetate (Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc, 3
NaOAc, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) natural deep eutectic solvent (NADES). Prior to extraction, the seed paste was exposed to air for 35 min to enable polyphenol oxidase-mediated color development. Aqueous and ethanolic extractions yielded dried powders, and the Gly
1) natural deep eutectic solvent (NADES). Prior to extraction, the seed paste was exposed to air for 35 min to enable polyphenol oxidase-mediated color development. Aqueous and ethanolic extractions yielded dried powders, and the Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES produced a stable, ready-to-use liquid CASE/NADES with high recoveries (88.6% Hass and 88.2% Fuerte), calculated as the mass of the final liquid system relative to the initial dry seed mass. CASEs showed maximum absorbance at 480 nm and tartrazine-equivalent quantification. The CASE/Gly
1) NADES produced a stable, ready-to-use liquid CASE/NADES with high recoveries (88.6% Hass and 88.2% Fuerte), calculated as the mass of the final liquid system relative to the initial dry seed mass. CASEs showed maximum absorbance at 480 nm and tartrazine-equivalent quantification. The CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES formulation exhibited pH-dependent color stability across pH 3–7 and maintained chromatic integrity under varied storage and light conditions. Antioxidant capacity, expressed in µmol copper reducing equivalents (CRE) per mL, was the highest in ethanolic extracts (3980.33 for the Fuerte variety and 1980.33 for the Hass variety), followed by NADES extracts with 934.61 and 736.56 CRE per mL, respectively. The CASE/(Gly
NaOAc NADES formulation exhibited pH-dependent color stability across pH 3–7 and maintained chromatic integrity under varied storage and light conditions. Antioxidant capacity, expressed in µmol copper reducing equivalents (CRE) per mL, was the highest in ethanolic extracts (3980.33 for the Fuerte variety and 1980.33 for the Hass variety), followed by NADES extracts with 934.61 and 736.56 CRE per mL, respectively. The CASE/(Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc 3
NaOAc 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) system exhibited color intensification under basic conditions and remained stable under both light and dark storage conditions. Total phenolic content (TPC) reached 203.45 mg GAE/100 g fresh weight for the Hass variety and 171.92 mg GAE/100 g fresh weight for the Fuerte variety when extracted with the Gly
1) system exhibited color intensification under basic conditions and remained stable under both light and dark storage conditions. Total phenolic content (TPC) reached 203.45 mg GAE/100 g fresh weight for the Hass variety and 171.92 mg GAE/100 g fresh weight for the Fuerte variety when extracted with the Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES. Cytotoxicity assays (MTT) on human skin fibroblasts confirmed that the CASE/(Gly
1) NADES. Cytotoxicity assays (MTT) on human skin fibroblasts confirmed that the CASE/(Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc) 3
NaOAc) 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NADES systems were non-toxic and potentially suitable as natural food-coloring alternatives. The green valorization of avocado seeds into functional colorants offers a promising route for waste reduction, sustainable bioproduct development and new industrial value chains, supporting the advancement of a resilient circular bioeconomy.
1 NADES systems were non-toxic and potentially suitable as natural food-coloring alternatives. The green valorization of avocado seeds into functional colorants offers a promising route for waste reduction, sustainable bioproduct development and new industrial value chains, supporting the advancement of a resilient circular bioeconomy.
Sustainability spotlightThe food industry contributes up to a third of annual greenhouse gas (GHG) emissions worldwide, with food loss and waste responsible for approximately half of these emissions. The valorization of food and agro-industrial bioresidues and by-products such as avocado seeds and peels, as well as residues from other fruits and vegetables, within integrated biorefineries can significantly reduce GHG emissions and foster circular engineering, circular bioeconomy and sustainability. Colored avocado seed extract (CASE) represents a promising natural alternative to synthetic colorants in the food industry, offering additional antimicrobial properties that enhance the safety and functionality of food products, including functional foods and nutraceuticals. Its incorporation can also extend product shelf life and help minimize food waste. Moreover, natural colorants are increasingly used in hypoallergenic foods for children and specialized dietary products. The development and manufacturing of plant-based colorant alternatives are fully aligned with the United Nations Sustainable Development Goals (SDGs) 2, 3, 8, 9, 12 and 15. |
1 Introduction
Avocado (Persea americana Mill.) is widely consumed in Ecuador and globally. Its cultivation has emerged as an important alternative crop, supporting agricultural diversification and strengthening rural economies. In Ecuador, the main cultivated varieties in the country are “Fuerte”, which is primarily consumed domestically, and “Hass”, which is mainly dedicated to exportation.1 The international demand for avocado has steadily grown over the past five years, which is attributable, in part, to its recognized bioactive properties. Several studies have highlighted the antioxidant and anti-inflammatory activities of avocado, supporting its potential role in disease prevention and management as well as its relevance as an ingredient in functional foods, nutraceuticals and pharmaceuticals.2The use of natural colorants in food processing is an ancestral practice. In recent years, interest in natural colorants with health-promoting properties has grown significantly among consumers. The non-biodegradable and carcinogenic risks associated with synthetic colorants have spurred a global shift toward natural alternatives across the food, textile, pharmaceutical and cosmetic industries. In the food sector, natural colorants not only enhance the aesthetic quality of foods and beverages but also increase consumer acceptance and demand.3 There is a pressing need for new, stable, safe and functional sources of natural colorants. Biomass, including agro-industrial and food processing residues, represents a renewable source of natural compounds, such as colorants and other high value-added chemicals and materials. The industrial production of avocado oil, guacamole and sauces has increased substantially in recent years, generating significant quantities of residues and by-products, primarily peels and seeds. In 2019, approximately 2 million tons of these residues were produced worldwide. If not sustainably managed, such residues remain underutilized and can negatively affect the environment.4 Second-generation biorefineries in cascade promote zero-waste technologies, circular bioeconomy and sustainability, aligning with the United Nations 2030 agenda.5
Avocado seed (AS) represents approximately 16% of the total fruit weight and contains valuable compounds, such as antioxidants (polyphenols, pigments, acetogenins, fatty acids, and lipids) and biopolymers, such as starch, cellulose, hemicellulose, and lignin (Table 1). These characteristics make the seed an excellent candidate for the valorization of agro-industrial by-products through integrated biorefinery approaches, starting with the extraction of phytochemicals from avocado seeds, followed by the extraction of proteins and starch, and the subsequent fractionation of cell wall components to obtain cellulose, hemicellulose and lignin.6,7 The main polyphenols in AS are procyanidin B1, while no anthocyanins have been detected,2,8 as shown in Table 1.
| Chemical compound | Hass variety | Fuerte variety | Observations | References |
|---|---|---|---|---|
| Total phenolic content | 9510.00–57300.00 (µg per g dry weight) varying values by source | 59![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200.00 (µg per g dry weight) varying values by source 200.00 (µg per g dry weight) varying values by source |
Quantified as gallic acid equivalents | Bangar et al., 2022 Tremocoldi et al., 2018 |
| Catechin | 3.64 (µg per g dry weight) | 8.13 (µg per g dry weight) | Present in peels of both varieties. Important for antioxidant activity in seed extracts | Bangar et al., 2022 Tremocoldi et al., 2018 |
| Epicatechin | 10.27 (µg per g dry weight) | 11.06 (µg per g dry weight) | Found in higher concentrations in both varieties | Bangar et al., 2022 Tremocoldi et al., 2018 |
| Procyanidin B1 | 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 380.00 (µg per g dry weight) 380.00 (µg per g dry weight) |
28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 340.00 (µg per g dry weight) 340.00 (µg per g dry weight) |
Major procyanidin found | Bangar et al., 2022 Tremocoldi et al., 2018 |
| Trans-5-O-caffeoylquinic acid | 1630.00 (µg per g dry weight) | 5740.00 (µg per g dry weight) | Representative phenolic acid | Bangar et al., 2022 Tremocoldi et al., 2018 |
| Anthocyanins | Not reported in seeds | Not reported in seeds | Avocado seeds generally lack anthocyanin pigments | Bangar et al., 2022 Tremocoldi et al., 2018 |
| Pigments/colorants | Lutein (low), zeaxanthin (low), chlorophylls (low), carotenoids (high), perseorangin (high) | — | Perseorangin is unique to avocado seeds but is an unspecified variety in the Hatzakis et al. study | Bangar et al., 2022 Hatzakis et al., 2019 |
| Starch | 63.70% (ripe) 58.7% (overripe) | ∼60–65% | Major polysaccharide (>90% of carbohydrate fraction) | Dávila et al., 2017 |
| Cellulose | 14.72% (ripe) 16.36% (overripe) | 15–17% | Not quantified per variety | Dávila et al., 2017 |
| Hemicellulose | 49.75% (ripe) 34.15% (overripe) | 30–50% | Minor polysaccharide (fiber) | Dávila et al., 2017 |
| Lignin | 9.82% (ripe, insoluble) 15.25% (overripe) | ∼10–15% | <10% (fiber) | Dávila et al., 2017 |
| Other | Rich in acetogenins, fatty acids, lipids | — | Oleic acid/linoleic/palmitic acids dominate the lipid profile, with no explicit comparison between Fuerte and Hass | Bangar et al., 2022 |
Different solvents have been employed for the extraction of natural colorants. In the food industry, organic solvents are most commonly used; however, these solvents are volatile, inflammable and pose health hazards. When ingested, inhaled above permissible limits or in direct contact with the skin, they may cause irritation.9 In contrast, alcohol–water mixtures are considered more environmentally favorable than pure alcohols.10 Wang et al. (2010) studied the phytochemical composition, antioxidant capacity and total phenolic content (TPC) of various avocado strains and cultivars using acetone/water/acetic acid solvent systems, finding a strong correlation between antioxidant capacity, phenolic content and procyanidins.11 Similarly, Dabas et al. (2011) used methanol to obtain an extract whose antioxidant activity was dependent on extraction time and polyoxidase enzyme activity, suggesting its potential as a food colorant.12 The extraction of valuable bioactive compounds from agro-industrial wastes, including avocado peels and seeds, using methanol has been widely reported.13 Similarly, Suarez et al. (2016) reported higher yields and greater colorant concentrations when extracting a natural colorant from fresh AS (Hass variety) using ethanol as a solvent.14
Bioactive phytochemical extraction from AS was optimized using microwave-assisted extraction with acetone and ethanol. Acetone is more effective than ethanol in extracting bioactive compounds with higher antioxidant activity, demonstrating that solvent polarity significantly influences the solubility of phenolic compounds.15 Hue et al. (2021) compared maceration, percolation, hot extraction, and Soxhlet extraction of AS and found that Soxhlet extraction yielded the highest TPC, the strongest antioxidant activities, and no acute oral toxicity.16 Shi Ong et al. (2022) evaluated an organic solvent-free method using pressurized hot water extraction (PHWE), and they reported that the extract exhibited antioxidant activity and inhibited oxidative stress-induced metabolomic changes in endothelial cells.17 More recently, Kupnik et al. (2023) compared ultrasound-assisted extraction (UAE), Soxhlet extraction, and supercritical fluid extraction (SFE) using water, ethanol, and scCO2 – ethanol as solvents, respectively, and found that Soxhlet extraction yielded the highest antioxidant activity.18
Sustainable and greener solvent alternatives are urgently needed. Deep eutectic solvents (DESs) and their subclass, natural deep eutectic solvents (NADESs), have emerged as promising green options for conventional organic solvents for the extraction of natural products and the valorization of agro-food residues. Their advantages include eco-friendliness, low toxicity, biodegradability, and tunable physicochemical properties (hydrophilic, hydrophobic, and therapeutic).19,20 DESs are typically composed of Brønsted or Lewis acids and bases, containing various cations and/or anions. These mixtures become liquids at room temperature due to their eutectic behavior and are considered “green solvents” because of their low toxicity and vapor pressure compared with traditional molecular solvents.21 NADES are formed from two or more natural compounds that yield a liquid phase at room temperature and are generally based on hydrogen bond acceptors and donors. NADESs also exhibit a strong thermal stabilizing ability attributed to extensive hydrogen bonding interactions between solutes and solvent molecules. For instance, Dai et al. (2014) demonstrated that the high viscosity of sugar-based NADESs with minimal water content ensures stable molecular interactions with the colorant carthamin from safflower, thereby enhancing the stability of the phenolic compounds.22 Among NADESs, glycerol-based eutectic mixtures have proven particularly efficient for extracting phenolic compounds from various agro-residues and by-products. Mouratoglou et al. (2016) investigated glycerol-based eutectic mixtures, such as glycerol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) choline chloride (Gly
choline chloride (Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ChCl, 3
ChCl, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), glycerol
1), glycerol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sodium acetate (Gly
sodium acetate (Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc, 3
NaOAc, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), and glycerol
1), and glycerol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sodium tartrate
sodium tartrate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water (Gly
water (Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaT
NaT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) W, 5
W, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) and found that Gly
4) and found that Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ChCl (3
ChCl (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and Gly
1) and Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) exhibited the highest extraction yields.23
1) exhibited the highest extraction yields.23
From a safety perspective, glycerol is a well-known food additive (E 422) widely used in the food industry and is considered safe.24 Sodium acetate, commonly used as a food preservative and flavoring agent, has also been shown to be safe at low concentrations according to cytotoxicity and genotoxicity studies.25 Both glycerol and sodium acetate have already been used in diverse industrial processes. Therefore, synthesizing NADESs from Gly and NaOAc represents a suitable strategy for producing safe and effective solvents for the extraction of natural colorants, which could serve as ready-to-use food coloring agents, offering environmental and health benefits over traditional solvents.
A major challenge in the use of CASE as a food agent is its instability at high temperatures. This limitation can be addressed by employing NADESs. Del Castillo-Llamosas et al. (2023)26 evaluated the extraction of phenolic and carbohydrate compounds from AS using ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Gly (1
Gly (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) DES, 50% ethanol and 80% ethanol as solvents, demonstrating that ChCl
1) DES, 50% ethanol and 80% ethanol as solvents, demonstrating that ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Gly DES is a more efficient solvent. More recently, Grisales-Mejía et al. (2025)27 synthesized ten binary NADES mixtures at a 1
Gly DES is a more efficient solvent. More recently, Grisales-Mejía et al. (2025)27 synthesized ten binary NADES mixtures at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 M ratio using avocado Hass peels and seeds. They employed ChCl and betaine (bet) as HBAs, and glycerol, fructose, glucose, lactic acid, and citric acid as HBDs, adding 10% water to facilitate NADES formation. The authors concluded that ChCl
1 M ratio using avocado Hass peels and seeds. They employed ChCl and betaine (bet) as HBAs, and glycerol, fructose, glucose, lactic acid, and citric acid as HBDs, adding 10% water to facilitate NADES formation. The authors concluded that ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) fructose and ChCl
fructose and ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) lactic acid exhibited the highest extraction efficiencies. Thus, DESs and NADESs can be considered non-toxic, eco-friendly and cost-effective extraction media, representing one of the most promising green alternatives to conventional organic solvents for recovering valuable bioactive phytochemicals.
lactic acid exhibited the highest extraction efficiencies. Thus, DESs and NADESs can be considered non-toxic, eco-friendly and cost-effective extraction media, representing one of the most promising green alternatives to conventional organic solvents for recovering valuable bioactive phytochemicals.
Avocado seeds are increasingly incorporated into various commercial products. For instance, several functional foods have been enriched with nano-sized avocado seed powder as an antioxidant ingredient, including yogurt, fortified breads, and diverse beverages and baked products.28–30 In parallel, the demand for natural food colorants is increasing among both consumers and manufacturers. This shift is largely driven by growing concerns about synthetic colorants, which are typically petroleum-derived and linked to health and environmental effects such as carcinogenicity, allergic reactions, and hyperactivity in children.31 Recently, the United States Food and Drug Administration (FDA) prohibited the use of the synthetic dye red no. 3 due to its association with cancer.32 Consequently, there is a pressing need for natural alternatives capable of replacing synthetic red hues.
The use of human primary skin fibroblasts is essential for this research because these cells closely reproduce the physiological conditions and responses of their in vivo counterparts.33,34 Such models are particularly suitable for reliable cytotoxicity evaluations, whereas immortalized cell lines often present altered behaviors and phenotypes that fail to reflect normal cellular mechanisms.35,36 This level of physiological sensitivity is crucial when assessing the safety of natural extracts, including novel NADES-based formulations, whose biocompatibility must be rigorously verified. Applying this approach is fundamental not only for advancing potential applications of these extracts but also for meeting regulatory requirements that demand robust and physiologically relevant toxicological data.
This study reports the sustainable extraction of non-toxic colored avocado seed extracts (CASEs) with low cytotoxicity using three solvents: distilled water, 70% ethanol EtOH and a natural deep eutectic solvent (Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc 3
NaOAc 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NADES) composed of glycerol and sodium acetate in a 3
1 NADES) composed of glycerol and sodium acetate in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio. The extraction processes were evaluated in terms of yield, color concentration, extraction kinetics, stability, antioxidant activity, and the effect of pH. Additionally, representative extracts were assessed to determine the cytotoxicity of the resulting CASE/Gly
1 molar ratio. The extraction processes were evaluated in terms of yield, color concentration, extraction kinetics, stability, antioxidant activity, and the effect of pH. Additionally, representative extracts were assessed to determine the cytotoxicity of the resulting CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc 3
NaOAc 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NADES system. The results demonstrate a simple, practical green extraction approach for producing an antioxidant, ready-to-use, and non-toxic CASE/Gly
1 NADES system. The results demonstrate a simple, practical green extraction approach for producing an antioxidant, ready-to-use, and non-toxic CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc 3
NaOAc 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NADES system that could potentially serve as a natural substitute for synthetic pigments and dyes.
1 NADES system that could potentially serve as a natural substitute for synthetic pigments and dyes.
2 Materials and methods
2.1 Materials and reagents
Avocados (Hass and Fuerte varieties) sourced from Guayllabamba, Pichincha Province, Ecuador, were used in the experiments. All reagents employed in the extract preparation were of food-grade. Glycerol (Loba Chemie, India), ethanol (Labsupply, Ecuador), sodium acetate, and choline chloride were obtained from Becton, Dickinson and Company (New Jersey, USA). For the antioxidant activity assays, a commercial OxiSelect™ TAC kit was purchased from Cell Biolabs (San Diego, USA). To determine total polyphenol content, Folin–Ciocalteu reagent was acquired from Scharlau (Barcelona, Spain), and sodium carbonate and gallic acid were supplied by Loba Chemie (India).2.2 Avocado seed selection, Gly/NaOAc 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 1 NADES synthesis and extraction preparation
1 NADES synthesis and extraction preparation
Avocado fruits were selected using a non-probabilistic approach method, prioritizing samples with comparable maturity and similar morphological characteristics. Seed preparation followed the protocol described by Dabas et al. (2011)10,12 to obtain avocado seed paste. Nine fruits per variety were washed, seeds removed, and cut into pieces approximately 0.5 × 0.5 mm in size. The seed pieces were blended with 0.7 volumes of distilled water until a homogeneous paste was obtained. This paste was spread to a thickness of ∼1 cm on a chopping board and left to stand for 35 min, enabling polyphenol oxidase (PPO) enzyme activity. PPOs, present in bacteria, mammals, and plants, are responsible for enzymatic browning and yellow-red pigment formation in fruits and vegetables.
CASE extraction was carried out using distilled water, 70% ethanol (EtOH), and Gly/NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES as solvents. For aqueous and ethanolic extractions, protocols combining elements from Suárez et al. (2016)14 and Kupnik et al. (2023)18 – selected for their high yields and practicality – were employed. For the aqueous extraction, 12.5 g of seed paste were mixed with 250 mL of distilled water in a 500 mL round-bottom flask and heated at 70 °C for 120 min (hot water extraction).For the ethanolic extraction, 12.5 g of seed paste was mixed with 250 mL of 70% ethanol in a 500 mL round-bottom flask equipped with a reflux condenser and heated at 70 °C for 120 min. The resulting extract was centrifuged (1000 rpm, 10 min), the supernatant was decanted, concentrated under reduced pressure using a rotary evaporator, and subsequently dried at 40 °C for 12 h, yielding a CASE powder, which was then weighed. The yield of the aqueous and ethanolic extractions was determined by the ratio between the weight of the CASE powder and the weight of the avocado seed used. The dried colored powder was redissolved in 20% ethanol at a fixed concentration of 2 mg mL−1, followed by centrifugation at 1000 rpm for 10 min. This standardized concentration was used for all aqueous and ethanolic extracts to enable direct comparison of color intensity at identical solute concentrations and optical path lengths. After confirming homogeneity, the visible spectrum was recorded, and the maximum absorbance peak was measured at 480 nm.
1) NADES as solvents. For aqueous and ethanolic extractions, protocols combining elements from Suárez et al. (2016)14 and Kupnik et al. (2023)18 – selected for their high yields and practicality – were employed. For the aqueous extraction, 12.5 g of seed paste were mixed with 250 mL of distilled water in a 500 mL round-bottom flask and heated at 70 °C for 120 min (hot water extraction).For the ethanolic extraction, 12.5 g of seed paste was mixed with 250 mL of 70% ethanol in a 500 mL round-bottom flask equipped with a reflux condenser and heated at 70 °C for 120 min. The resulting extract was centrifuged (1000 rpm, 10 min), the supernatant was decanted, concentrated under reduced pressure using a rotary evaporator, and subsequently dried at 40 °C for 12 h, yielding a CASE powder, which was then weighed. The yield of the aqueous and ethanolic extractions was determined by the ratio between the weight of the CASE powder and the weight of the avocado seed used. The dried colored powder was redissolved in 20% ethanol at a fixed concentration of 2 mg mL−1, followed by centrifugation at 1000 rpm for 10 min. This standardized concentration was used for all aqueous and ethanolic extracts to enable direct comparison of color intensity at identical solute concentrations and optical path lengths. After confirming homogeneity, the visible spectrum was recorded, and the maximum absorbance peak was measured at 480 nm.
Gly/NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES was prepared based on the method developed by Mouratoglou et al. (2016)23 with modifications to account for the strong stabilization capacity of NADES. Gly and NaOAc were mixed in a 3
1) NADES was prepared based on the method developed by Mouratoglou et al. (2016)23 with modifications to account for the strong stabilization capacity of NADES. Gly and NaOAc were mixed in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio and heated at 70 °C, with constant stirring (700 rpm) for 45 min until a clear liquid was formed. The resulting NADES was stored in a sealed glass container and protected from light at room temperature until use. For extraction, NADES was diluted to 90% (v/v) with water to facilitate handling.
1 molar ratio and heated at 70 °C, with constant stirring (700 rpm) for 45 min until a clear liquid was formed. The resulting NADES was stored in a sealed glass container and protected from light at room temperature until use. For extraction, NADES was diluted to 90% (v/v) with water to facilitate handling.
Colored compounds from AS were extracted using Gly/NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES via a percolation method due to the non-volatile nature of the solvent. Extraction conditions (temperature and duration) were identical to those used for the ethanol and aqueous protocols. Briefly, 12.5 g of avocado seed paste was mixed with 250 mL of Gly/NaOAc (3
1) NADES via a percolation method due to the non-volatile nature of the solvent. Extraction conditions (temperature and duration) were identical to those used for the ethanol and aqueous protocols. Briefly, 12.5 g of avocado seed paste was mixed with 250 mL of Gly/NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES in a round-bottom flask and heated in a water bath, 70–80 °C, 120 min. The resulting mixture was vacuum filtered and centrifuged at 1000 rpm for 10 min. The supernatant was collected for yield determination.
1) NADES in a round-bottom flask and heated in a water bath, 70–80 °C, 120 min. The resulting mixture was vacuum filtered and centrifuged at 1000 rpm for 10 min. The supernatant was collected for yield determination.
Since the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES system is a ready-to-use liquid extract, it was directly employed as the stock extract solution without drying or concentration. The extraction yield was calculated as the mass of the recovered CASE/NADES fraction in the supernatant divided by the initial mass of the avocado seed paste. Because NADES is non-volatile, both the extract and the solid residue cannot be dried without altering their chemical composition. As a result, yield determination based on the dry mass or insoluble residue difference was not experimentally feasible. Therefore, the extraction yield for the NADES system was determined gravimetrically based on the liquid-phase CASE fraction. Extraction efficiencies among solvents were compared primarily using calibrated CASE concentration (ppm) and gravimetric yield trends rather than absolute dry-mass equivalence.
1) NADES system is a ready-to-use liquid extract, it was directly employed as the stock extract solution without drying or concentration. The extraction yield was calculated as the mass of the recovered CASE/NADES fraction in the supernatant divided by the initial mass of the avocado seed paste. Because NADES is non-volatile, both the extract and the solid residue cannot be dried without altering their chemical composition. As a result, yield determination based on the dry mass or insoluble residue difference was not experimentally feasible. Therefore, the extraction yield for the NADES system was determined gravimetrically based on the liquid-phase CASE fraction. Extraction efficiencies among solvents were compared primarily using calibrated CASE concentration (ppm) and gravimetric yield trends rather than absolute dry-mass equivalence.
For spectrophotometric analysis, an aliquot of the supernatant was diluted with distilled water (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v; extract
1, v/v; extract![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water) to reduce viscosity and improve accuracy. The diluted extract was centrifuged at 1000 rpm for 10 min and scanned across the visible spectrum, with maximum absorbance recorded at 480 nm. All extractions were performed in triplicate.
water) to reduce viscosity and improve accuracy. The diluted extract was centrifuged at 1000 rpm for 10 min and scanned across the visible spectrum, with maximum absorbance recorded at 480 nm. All extractions were performed in triplicate.
The CASE concentration in the NADES extract was quantified using a tartrazine calibration curve (100–500 ppm), with a maximum absorbance at 480 nm. For aqueous and ethanolic extracts, the dried CASE fraction was reconstituted to 2 mg mL−1, allowing direct interpolation of the dilution correction. In contrast, the NADES extract required a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dilution prior to UV-vis analysis, and its concentration was obtained by interpolation of the dilution-corrected absorbance. The resulting CASE concentrations (ppm) were used to compare the extraction efficiency among the solvent systems.
1 dilution prior to UV-vis analysis, and its concentration was obtained by interpolation of the dilution-corrected absorbance. The resulting CASE concentrations (ppm) were used to compare the extraction efficiency among the solvent systems.
2.3 Determination of polyphenol content
The total polyphenol content was determined using CASE powders obtained from the two avocado seed varieties (Hass and Fuerte). The CASEs were diluted in distilled water at different concentrations prior to analysis. For each measurement, 100 µL of the diluted extract was mixed with 500 µL of 0.2 N Folin–Ciocalteu reagent. After 5 minutes, 400 µL of sodium carbonate solution was added, and the mixture was incubated for 2 h at room temperature. The absorbance was then recorded at 760 nm using a UV-vis spectrophotometer. A calibration curve was prepared with gallic acid solutions ranging from 0.25 to 2 mM. All measurements for both standards and samples were performed in triplicate.2.4 Total antioxidant capacity
The total antioxidant capacity was determined using a colorimetric assay with the “OxiSelect TM TAC” kit (Cell Biolabs, USA) following the manufacturer's instructions. Prior to analysis, the CASE samples were centrifuged and stored at 4 °C for 24 h. Absorbance was measured at 490 nm, and a calibration curve was constructed using uric acid standards ranging from 0.06 to 1.43 mM.2.5 Kinetics, pH and stability
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio solely to reduce viscosity before UV-vis analysis. Absorbance was then recorded at 480 nm using a UV-vis spectrophotometer.
1 ratio solely to reduce viscosity before UV-vis analysis. Absorbance was then recorded at 480 nm using a UV-vis spectrophotometer.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 volume ratio (CASE
1 volume ratio (CASE![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PBS, v/v). Samples were gently mixed and centrifuged at 5000 rpm for 10 min at room temperature, and their absorbance spectra were recorded at 480 nm. The normalized absorbance value, defined as A0, was used as the reference for subsequent pH-dependent comparisons.
PBS, v/v). Samples were gently mixed and centrifuged at 5000 rpm for 10 min at room temperature, and their absorbance spectra were recorded at 480 nm. The normalized absorbance value, defined as A0, was used as the reference for subsequent pH-dependent comparisons.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (CASE
1 ratio (CASE![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PBS, v/v) and adjusting according to each sample's DF. The tubes were sealed and stored under varying temperatures (4 °C and 25 °C) and light conditions (light/dark). Absorbance was measured daily over 15 days and expressed as A/A0, where A0 denotes the day-0 absorbance of the normalized sample.
PBS, v/v) and adjusting according to each sample's DF. The tubes were sealed and stored under varying temperatures (4 °C and 25 °C) and light conditions (light/dark). Absorbance was measured daily over 15 days and expressed as A/A0, where A0 denotes the day-0 absorbance of the normalized sample.2.6 Cytotoxicity assessments
The cytotoxicity of aqueous and ethanolic CASEs was evaluated for both avocado varieties, while the CASE/Gly/NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES system was tested only for the Hass variety, given its global commercial predominance. Different concentrations of CASEs (5, 0.5, 0.05, 0.005, and 0.0005 mg mL−1) were prepared in Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% fetal calf serum (FCS), glutamine, and 1% penicillin–streptomycin (Gibco, Cascade Biologics, Portland, OR, USA), following a procedure previously described by our research group.36
1) NADES system was tested only for the Hass variety, given its global commercial predominance. Different concentrations of CASEs (5, 0.5, 0.05, 0.005, and 0.0005 mg mL−1) were prepared in Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% fetal calf serum (FCS), glutamine, and 1% penicillin–streptomycin (Gibco, Cascade Biologics, Portland, OR, USA), following a procedure previously described by our research group.36
Primary human fibroblasts were isolated from a skin biopsy of a middle-aged female donor and obtained with informed consent at Los Andes University (Chile) and kindly provided by Dr Maroun Khoury. The effects of the treatments were assessed after 24 and 48 h of incubation using the MTT assay. For this assay, fibroblasts (1250 cells per well) were seeded in 96-well plates containing 100 µL DMEM (10% FCS) and cultured for 24 h at 37 °C and 5% CO2. After incubation, the medium was replaced with 100 µL of each extract solution, and control wells were prepared for each extract type. The controls contained only DMEM (10% FCS). At the end of each incubation period, the medium was replaced with 100 µL of an MTT solution (500 µg mL−1 in DMEM supplemented with 1% FCS; Invitrogen, Waltham, Massachusetts, United States) and incubated for an additional 2 h under the same conditions. The MTT solution was then removed, and 50 µL of DMSO (Santa Cruz Biotechnology, Dallas, TX, United States) was added to each well to solubilize the formed crystals. Absorbance was measured immediately at 570 nm using an Epoch™ microplate spectrophotometer. All assays were performed in quintuplicate.
2.7 Evaluation of CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) NaOAc NADES system as a natural colorant in yogurt
NaOAc NADES system as a natural colorant in yogurt
Preliminary experiments were carried out to assess the potential application of avocado seed extract (CASE), formulated in glycerol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sodium acetate natural deep eutectic solvent, referred to as the CASE/Gly
sodium acetate natural deep eutectic solvent, referred to as the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system, as a ready-to-use natural colorant in dairy products. Three commercial brands of natural yogurt were selected and evaluated independently. For each sample, 1 mL of this CASE/NADES system was added to 100 mL of yogurt and mixed gently until a homogeneous color distribution was achieved. Untreated yogurt from each brand served as the control for evaluating color variation following the CASE system incorporation.
NaOAc NADES system, as a ready-to-use natural colorant in dairy products. Three commercial brands of natural yogurt were selected and evaluated independently. For each sample, 1 mL of this CASE/NADES system was added to 100 mL of yogurt and mixed gently until a homogeneous color distribution was achieved. Untreated yogurt from each brand served as the control for evaluating color variation following the CASE system incorporation.
Prior to UV-vis analysis, all yogurt samples (treated and controls) were diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (v/v) with distilled water to reduce turbidity and limit light scattering. The diluted suspensions were clarified by centrifugation at 6000 rpm for 10 min, and the resulting clear supernatant was used for spectrophotometric measurements.
10 (v/v) with distilled water to reduce turbidity and limit light scattering. The diluted suspensions were clarified by centrifugation at 6000 rpm for 10 min, and the resulting clear supernatant was used for spectrophotometric measurements.
Visible absorption spectra were recorded between 400 and 700 nm using a Thermo Scientific UV-vis spectrophotometer to determine the wavelength of maximum absorbance (λmax) and evaluate the color shift induced by the addition of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system. Measurements were conducted at room temperature in quartz cuvettes using appropriate blanks.
NaOAc NADES system. Measurements were conducted at room temperature in quartz cuvettes using appropriate blanks.
The density of the yogurt samples was determined gravimetrically by weighing a known volume (10 mL) of the sample before and after addition of the extract, with the results expressed in g mL−1. The pH was measured with a calibrated pH meter (buffers pH 4.00 and 7.00). All measurements were performed in triplicate, and the results are reported as the mean ± standard deviation.
2.8 Statistical analysis
All quantitative variables (yield, absorbance, concentration, and antioxidant capacity) were evaluated using a two–factor experimental design. The fixed factors were solvent (distilled water, 70% ethanol, and Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc 3
NaOAc 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NADES) and avocado seed variety (Hass and Fuerte), with three replicates per treatment. Residuals were tested for normality using the Anderson–Darling method and for homogeneity of variances using Levene's test. As both assumptions were met, the data were subjected to two-way analysis of variance (ANOVA, α = 0.05). When significant differences were detected, Tukey's HSD test (95% confidence) was applied for pairwise comparisons. In the figures, uppercase letters denote statistically different solvent groups, while lowercase letters denote differences between varieties.
1 NADES) and avocado seed variety (Hass and Fuerte), with three replicates per treatment. Residuals were tested for normality using the Anderson–Darling method and for homogeneity of variances using Levene's test. As both assumptions were met, the data were subjected to two-way analysis of variance (ANOVA, α = 0.05). When significant differences were detected, Tukey's HSD test (95% confidence) was applied for pairwise comparisons. In the figures, uppercase letters denote statistically different solvent groups, while lowercase letters denote differences between varieties.
For the stability analysis, absorbance values were averaged from three replicates and normalized to the initial value (A0) to calculate A/A0 over storage time. Since the data did not satisfy the assumptions of normality and homoscedasticity, differences among solvents, storage temperatures (4 °C and 25 °C), and light/dark conditions were assessed using the Kruskal–Wallis test (α = 0.05).
The relationship between absorbance at 480 nm and antioxidant capacity was explored using a matrix plot in Minitab, distinguishing samples by solvent and variety. This analysis was exploratory and descriptive, without regression modeling or correlation testing.
All statistical analyses were performed using Minitab® 19.1 (Minitab, LLC, State College, PA, USA).
3 Results and discussion
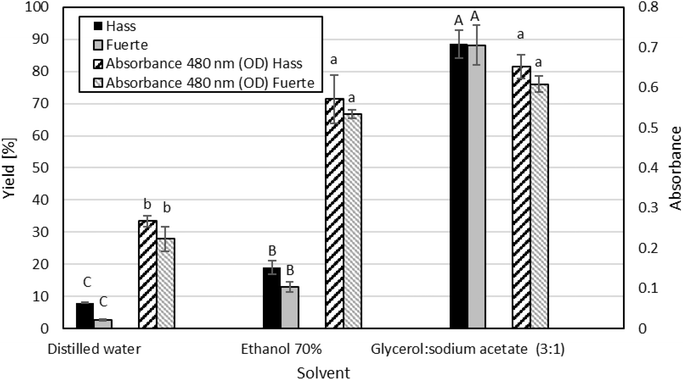
3.1 Yields of the extractions
Fig. 1 shows the visual appearance of the CASE samples prepared using the three selected solvent systems. The aqueous CASEs exhibited a transparent orange color, while the ethanolic CASE displayed a more intense orange hue. In contrast, the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES system showed vivid reddish-orange coloration.
1) NADES system showed vivid reddish-orange coloration.
The analysis of variance revealed a significant effect of solvent type on CASE yield (p < 0.05), whereas no statistical differences were observed between varieties. Tukey's multiple comparison test (95% confidence) identified three statistically distinct groups: Group A, percolation extraction using Gly/NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES, which produced the highest yields (88.6% for Hass; 88.2% for Fuerte); Group B, reflux extraction with 70% ethanol (19.0% Hass; 13.0% Fuerte); and Group C, hot aqueous extraction with distilled water at 70 °C (8.0% Hass; 2.6% Fuerte) (Fig. 2).
1) NADES, which produced the highest yields (88.6% for Hass; 88.2% for Fuerte); Group B, reflux extraction with 70% ethanol (19.0% Hass; 13.0% Fuerte); and Group C, hot aqueous extraction with distilled water at 70 °C (8.0% Hass; 2.6% Fuerte) (Fig. 2).
Using distilled water, the yield for Hass was lower than that reported by Kupnik et al. (2023),18 who obtained 12.0% by employing the UAE. For ethanol-based extraction, the present results were similar to those of Suárez et al. (2016),14 who reported 20.5% for the avocado Hass variety using Soxhlet extraction with absolute ethanol. Kupnik et al. (2023)18 also obtained 9.88% with Soxhlet extraction (20 h, 40 °C), indicating that elevated temperatures combined with shorter residence times can enhance extraction efficiency. In contrast, Munthe et al. (2023)37 reported a much higher yield for the avocado Hass variety (39.58%) using reflux extraction with 70% ethanol for 15 h at 40 °C, compared with the current values (19.0% for Hass and 13% for Fuerte). These findings suggest that longer extraction times improve efficiency; however, extended processes also entail greater energy consumption, thereby reducing environmental sustainability.
The high yield obtained with Gly/NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES (Group A) can be attributed to the high NADES/CASE concentration in the newly formed CASE/Gly
1) NADES (Group A) can be attributed to the high NADES/CASE concentration in the newly formed CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc system, which eliminates drying or concentration requirements. This system could be used directly as a ready-to-use product, given the low acute toxicity of its components: glycerol ingestion must exceed 125 mg kg−1 h−1 to cause adverse effects, while sodium acetate has no established daily intake.24 Thus, this system exhibits strong potential as an alternative food coloring agent.
NaOAc system, which eliminates drying or concentration requirements. This system could be used directly as a ready-to-use product, given the low acute toxicity of its components: glycerol ingestion must exceed 125 mg kg−1 h−1 to cause adverse effects, while sodium acetate has no established daily intake.24 Thus, this system exhibits strong potential as an alternative food coloring agent.
Absorbance at 480 nm, used exclusively as a qualitative indicator of color intensity, also showed significant differences among solvents. Tukey's test classified the samples into two groups: Group a, comprising Gly/NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES and reflux extraction with 70% ethanol, which exhibited the highest absorbance values (0.534–0.652); and Group b, distilled water, which showed the lowest values (0.223–0.267) (Fig. 2).
1) NADES and reflux extraction with 70% ethanol, which exhibited the highest absorbance values (0.534–0.652); and Group b, distilled water, which showed the lowest values (0.223–0.267) (Fig. 2).
It is important to note that the absorbance values shown in Fig. 2 reflect the color intensity corresponding to the working concentrations used for each extraction system (2 mg mL−1 for aqueous and ethanolic extracts, and a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dilution for NADES extracts). Since these concentrations are not equivalent, the absorbance values are qualitative and not intended for a quantitative comparison among solvents. Extraction efficiency was instead assessed based on the gravimetric yield and the calibrated CASE concentration (ppm) rather than on absorbance measurements.
1 dilution for NADES extracts). Since these concentrations are not equivalent, the absorbance values are qualitative and not intended for a quantitative comparison among solvents. Extraction efficiency was instead assessed based on the gravimetric yield and the calibrated CASE concentration (ppm) rather than on absorbance measurements.
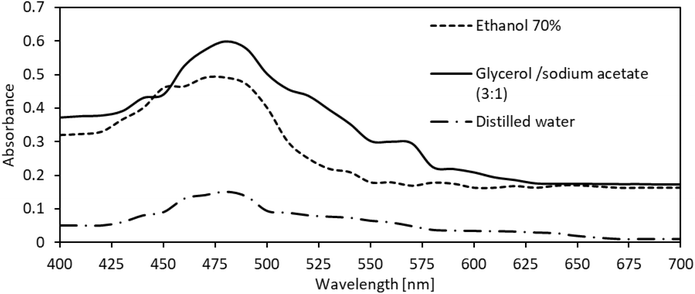
3.2 Concentration of reddish-orange CASEs
The color concentration of the CASEs was determined using UV-vis spectrophotometry. The maximum absorption wavelength (λmax = 480 nm) was first identified from the full spectral scans of the samples. Quantitative analysis was conducted at 480 nm using a tartrazine calibration curve (Fig. 3), and the results were expressed as tartrazine equivalents (ppm), consistent with previous reports.10 All concentration values were adjusted for the corresponding dilution factors applied to each extraction system. Although absorbance at λmax indicates relative color intensity, calibrated concentration values, rather than raw absorbance, were used for quantitative comparisons among solvents and varieties.However, the resulting CASE concentrations varied considerably. In this study, extraction with 70% ethanol yielded concentrations of 2210 ppm for the Hass avocado variety and 1310 ppm for the Fuerte variety. Potential interferences in the absorbance-based quantification of CASEs mainly depend on the linear relationship between concentration and absorbance. In complex mixtures, such as plant extracts, overlapping absorption bands or molecular interactions may occur, introducing additional measurement errors.38Dabas et al. (2011)12 used methanol and a sonication-assisted method to obtain a CASE exhibiting a maximum absorbance of 0.4 at λmax = 480 nm (24 °C), corresponding to the blue-green region of the visible spectrum and appearing orange in color. Similarly, Suárez et al. (2016)14 employed absolute ethanol for extraction from Hass avocado seeds, reporting an absorbance of 0.415 at λmax = 480 nm, with a reddish-orange appearance. In our study, the maximum absorbance peak likewise occurred at 480 nm for all CASEs extracted with the three solvents tested. The absorbance bands were broader and slightly variable, reflecting the complex polyphenolic composition of the extracts, depending on the solvent properties. This spectral variation typically spanning 400–500 nm, corresponds to red-orange hues.39 Notably, the CASE obtained using Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES exhibited a higher absorbance (0.460) for the Hass variety, exceeding the values reported for aqueous and ethanolic CASEs (Fig. 4). Statistical analysis confirmed significant differences between avocado varieties (p < 0.001) and among solvents (p < 0.001). The CASEs from the Hass variety presented higher concentrations than those from the Fuerte variety, while Gly
1) NADES exhibited a higher absorbance (0.460) for the Hass variety, exceeding the values reported for aqueous and ethanolic CASEs (Fig. 4). Statistical analysis confirmed significant differences between avocado varieties (p < 0.001) and among solvents (p < 0.001). The CASEs from the Hass variety presented higher concentrations than those from the Fuerte variety, while Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 significantly) differed from 70% ethanol and distilled water, which were not significantly different from each other (p > 0.05).
1 significantly) differed from 70% ethanol and distilled water, which were not significantly different from each other (p > 0.05).
Because absorbance is directly proportional to solute concentration, a higher absorbance value of 0.460 indicates a greater color concentration in the Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES extracts. This enhanced concentration performance may be attributed to several solvent characteristics: a slightly basic pH8,9 derived from sodium acetate, a viscosity reduced by 58.03% relative to pure glycerol,33 and a favorable polarity-solubility balance that enables the dissolution of both polar and non-polar compounds—an uncommon feature among traditional solvents. Moreover, exposure of the avocado seed material to air prior to extraction was found to be a crucial factor enhancing color concentrations, as evidenced by the kinetic studies discussed later.
NaOAc NADES extracts. This enhanced concentration performance may be attributed to several solvent characteristics: a slightly basic pH8,9 derived from sodium acetate, a viscosity reduced by 58.03% relative to pure glycerol,33 and a favorable polarity-solubility balance that enables the dissolution of both polar and non-polar compounds—an uncommon feature among traditional solvents. Moreover, exposure of the avocado seed material to air prior to extraction was found to be a crucial factor enhancing color concentrations, as evidenced by the kinetic studies discussed later.
Despite these promising results, limited research on CASE extraction with NADES hinders the full identification of factors affecting concentration variability. Recent studies have evaluated extraction efficiency not only through process parameters (e.g., solvent type, temperature, and time) but also by antioxidant activity, often reported as IC50 values (2025).16 However, IC50 values depend on the extract's chemical composition and do not fully capture its overall bioactivity. Although IC50 reflects radical scavenging capacity, other antioxidant mechanisms, such as metal chelation or enzyme interaction, are not represented.36
Although the isolation of pure or semi-pure pigments from CASEs was beyond the scope of this work, it is noteworthy that Hatzakis et al. (2019)40 isolated a pigment from CASE and elucidated its chemical structure using preparative HPLC. The compound was identified as a glycosylated benzotropone derivative with the molecular formula of C29H30O14 as the most abundant yellow-orange solid.
3.3 Kinetics, pH and stability
In avocado seeds, the activity of PPO specifically contributes to the formation of the characteristic reddish-orange coloration that develops upon oxidation in the presence of oxygen. Hatzakis et al. (2019)40 subsequently isolated and identified the orange pigment in CASE extracted using conventional solvents. Previous studies reported a 35 minute air exposure time of the paste for color quantification without promoting over-oxidation or secondary degradation.12,41 However, the mechanism of color formation using NADES has not been fully elucidated.It is important to emphasize that the kinetic profiles discussed herein correspond exclusively to the air exposure time of the avocado seed paste prior to extraction, reflecting PPO-driven color formation rather than solvent extraction kinetics.
The kinetic analysis in our study (Fig. 5) shows that the intensity of the orange coloration increases with longer air exposure of avocado seed paste, consistent with the findings of Dabas et al. (2011)12 and Hatzakis et al.40 (2019). The kinetic profiles varied depending on the solvent used. The CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES system displayed a markedly faster initial increase in absorbance upon air exposure, particularly within the first 2–5 minutes, followed by a continuous and sustained evolution over time. In contrast, the ethanolic system exhibited a slower initial rise in absorbance and more gradual progression throughout the experimental duration. These observations suggest that the NADES medium facilitates more rapid early-stage color development compared to the ethanolic solvent (Fig. 5, Hass variety shown as representative).
1) NADES system displayed a markedly faster initial increase in absorbance upon air exposure, particularly within the first 2–5 minutes, followed by a continuous and sustained evolution over time. In contrast, the ethanolic system exhibited a slower initial rise in absorbance and more gradual progression throughout the experimental duration. These observations suggest that the NADES medium facilitates more rapid early-stage color development compared to the ethanolic solvent (Fig. 5, Hass variety shown as representative).
 | ||
Fig. 5 Kinetics (absorbance versus air exposure time) of the Hass variety using the Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3 NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES, ethanol, and water. 1) NADES, ethanol, and water. | ||
The extension of the lag-phase in an enzyme-catalyzed reaction is generally attributed to delayed polymerization events rather than direct inhibition of enzymatic activity. Mouratoglou et al. (2016)23 reported that eutectic solvents, including Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES, achieved polyphenol extraction efficiencies comparable to those of methanol, which is typically used as a reference solvent in such studies. The solubilization capacity of an NADES depends strongly on its composition. The Gly
1) NADES, achieved polyphenol extraction efficiencies comparable to those of methanol, which is typically used as a reference solvent in such studies. The solubilization capacity of an NADES depends strongly on its composition. The Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES exhibits strong hydrogen bonding and van der Waals interactions, forming a stable solvent with polarity comparable to pure glycerol. This polarity enhances solubility and selectivity, as confirmed by the FTIR analysis reported by Tarigan et al. (2025).42 It is essential to clarify that this experiment evaluates the reaction kinetics of PPO during the air exposure of the avocado seed paste, prior to the extraction step. Consequently, the observed differences in absorbance profiles cannot be ascribed to solvent-induced variations in PPO catalytic activity, as no solvent is present during the air incubation period. The faster initial increase in absorbance observed in samples subsequently extracted with Gly
1) NADES exhibits strong hydrogen bonding and van der Waals interactions, forming a stable solvent with polarity comparable to pure glycerol. This polarity enhances solubility and selectivity, as confirmed by the FTIR analysis reported by Tarigan et al. (2025).42 It is essential to clarify that this experiment evaluates the reaction kinetics of PPO during the air exposure of the avocado seed paste, prior to the extraction step. Consequently, the observed differences in absorbance profiles cannot be ascribed to solvent-induced variations in PPO catalytic activity, as no solvent is present during the air incubation period. The faster initial increase in absorbance observed in samples subsequently extracted with Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES reflects differences in the stabilization and solubilization of oxidation-derived chromophoric intermediates during extraction rather than changes in PPO reaction kinetics. In contrast, ethanolic extracts exhibited a more gradual increase in absorbance likely due to variations in solvent–solute interactions and polarity influencing pigment solubilization after air exposure. Therefore, the apparent absence of a lag phase in NADES-extracted samples should be interpreted as a solvent-related post-oxidation effect rather than an intrinsic modification of PPO-mediated reaction kinetics.
1) NADES reflects differences in the stabilization and solubilization of oxidation-derived chromophoric intermediates during extraction rather than changes in PPO reaction kinetics. In contrast, ethanolic extracts exhibited a more gradual increase in absorbance likely due to variations in solvent–solute interactions and polarity influencing pigment solubilization after air exposure. Therefore, the apparent absence of a lag phase in NADES-extracted samples should be interpreted as a solvent-related post-oxidation effect rather than an intrinsic modification of PPO-mediated reaction kinetics.
Increasing the pH from 2 to 10 caused a progressive rise in absorbance at 480 nm. Visually, the CASE exhibited a pale yellow-orange hue at acidic pH = 2–6, a bright orange-pink hue at neutral pH = 7–8, and a deep reddish-orange hue at alkaline pH = 9–11. Thus, higher pH values increased the concentration of the chromophoric species, in agreement with Dabas et al. (2011).12 The gradual absorbance increase from pH 2 to 8 (from approximately 0.145 to 0.289 for the 70% ethanol and from approximately 0.103 to 0.213 for the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES system) may result from hydroxyl ions promoting the formation of species with enhanced electronic conjugation. Bond cleavage, ring opening or dissociation events could explain the sharp absorbance rise observed in aqueous CASEs at pH 8, compared to the ethanolic system (70%), where absorbance increased from 0.289 to 0.81. Dabas et al. (2011)12 reported a similar trend. At pH 2, the absorbance was about 0.1 at 480 nm, increasing gradually to 0.6 at pH 9. Beyond this point, the increase was more sharply thereafter, reaching 2.2 at pH 11. The increase in absorbance as a function of pH was irreversible under aerobic conditions. Lower absorbance values were recorded for the CASE/Gly
1) NADES system) may result from hydroxyl ions promoting the formation of species with enhanced electronic conjugation. Bond cleavage, ring opening or dissociation events could explain the sharp absorbance rise observed in aqueous CASEs at pH 8, compared to the ethanolic system (70%), where absorbance increased from 0.289 to 0.81. Dabas et al. (2011)12 reported a similar trend. At pH 2, the absorbance was about 0.1 at 480 nm, increasing gradually to 0.6 at pH 9. Beyond this point, the increase was more sharply thereafter, reaching 2.2 at pH 11. The increase in absorbance as a function of pH was irreversible under aerobic conditions. Lower absorbance values were recorded for the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system likely due to the buffering effect of NaOAc, which moderated pH and diluted the extract until convergence of absorbance values. In addition, the high viscosity of the NADES may have reduced molecular mobility, slowing the reaction kinetics of some species whose stabilization is favored at higher pH values.19 High viscosity can hinder substrate diffusion and molecular interactions; therefore, small amounts of water or buffering agents may be added to partially reduce viscosity and improve mass transfer without altering the NADES structure.43,44 Furthermore, solvent–solute interactions may protect chromophoric compounds from bond rupture or dissociation, limiting absorbance increases compared with aqueous and ethanolic systems.19
NaOAc NADES system likely due to the buffering effect of NaOAc, which moderated pH and diluted the extract until convergence of absorbance values. In addition, the high viscosity of the NADES may have reduced molecular mobility, slowing the reaction kinetics of some species whose stabilization is favored at higher pH values.19 High viscosity can hinder substrate diffusion and molecular interactions; therefore, small amounts of water or buffering agents may be added to partially reduce viscosity and improve mass transfer without altering the NADES structure.43,44 Furthermore, solvent–solute interactions may protect chromophoric compounds from bond rupture or dissociation, limiting absorbance increases compared with aqueous and ethanolic systems.19
These results and their corresponding kinetic profiles support the hypothesis that the coloration of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system is not related to anthocyanins.2,8 Anthocyanins typically show orange-red tones at pH ≤ 3, shift to bluish hues at pH 6–7 due to deprotonation, and degrade at higher pH.45 Although anthocyanins are widely used natural pigments to impart orange-red tones in food applications, their stability is poor at elevated pH.
NaOAc NADES system is not related to anthocyanins.2,8 Anthocyanins typically show orange-red tones at pH ≤ 3, shift to bluish hues at pH 6–7 due to deprotonation, and degrade at higher pH.45 Although anthocyanins are widely used natural pigments to impart orange-red tones in food applications, their stability is poor at elevated pH.
In contrast, the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system appears to be a promising alternative because it remains stable throughout the typical pH range of food products (pH 3–7). Dabas et al.12 reported that adding CASE to milk (pH between 6.6 and 6.8) produced a pinkish hue. Similarly, in our preliminary trials, the addition of the CASE/Gly
NaOAc NADES system appears to be a promising alternative because it remains stable throughout the typical pH range of food products (pH 3–7). Dabas et al.12 reported that adding CASE to milk (pH between 6.6 and 6.8) produced a pinkish hue. Similarly, in our preliminary trials, the addition of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system to yogurt (pH 4.4–4.6) yielded an orange hue, while its addition to milk resulted in a pinkish coloration. These results confirm that pH is a critical parameter influencing the final color and must be carefully controlled for food applications. Generally, incorporation of the CASE/Gly
NaOAc NADES system to yogurt (pH 4.4–4.6) yielded an orange hue, while its addition to milk resulted in a pinkish coloration. These results confirm that pH is a critical parameter influencing the final color and must be carefully controlled for food applications. Generally, incorporation of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system into foods near neutral pH results in hues shifting toward red rather than orange. Moreover, lower concentrations of CASE are needed at higher pH, as color intensity increases under these conditions.
NaOAc NADES system into foods near neutral pH results in hues shifting toward red rather than orange. Moreover, lower concentrations of CASE are needed at higher pH, as color intensity increases under these conditions.
CASEs maintained color stability for 15 days under both light and dark conditions, regardless of temperature or solvent, except for the extract prepared with distilled water, which lost color stability before the 15 days when exposed to light at room temperature.
Among all solvent systems evaluated, the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES exhibited the greatest stability over time and under various storage conditions (Fig. 6). Water content was identified as a major factor influencing stability. Dai et al. (2013)19 associated the high stability of extracts obtained using eutectic solvents with their elevated viscosity (limiting molecular mobility and promoting stable interactions), presence of sugar components (facilitating hydrogen bonding), and pH maintenance capacity.
1) NADES exhibited the greatest stability over time and under various storage conditions (Fig. 6). Water content was identified as a major factor influencing stability. Dai et al. (2013)19 associated the high stability of extracts obtained using eutectic solvents with their elevated viscosity (limiting molecular mobility and promoting stable interactions), presence of sugar components (facilitating hydrogen bonding), and pH maintenance capacity.
These findings indicate that the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system formulations have strong potential for application in food products stored at both room temperature and under refrigeration, irrespective of light exposure, except when distilled water is the solvent. Stability analysis revealed that solvent type was the main factor affecting A/A0 values (Kruskal–Wallis, H = 48.23; df = 2; p < 0.001), while storage temperature had a significant but less pronounced effect (H = 8.95; df = 1; p = 0.003). No significant differences were found between light and dark storage conditions (H = 0.00; df = 1; p = 0.968), confirming the suitability of this system under varying illuminations. An increase in the absorbance ratio A/A0 above 1 may indicate the formation of degradation products that absorb at or near the same wavelength, leading to transient absorbance changes. The instability of colorants in aqueous systems is often attributed to oxygen-mediated and pH-dependent chemical reactions. Many natural pigments or colorants containing conjugated double bonds are prone to oxidation and structural rearrangements in aqueous environments, altering their electronic structure and chromophores, which in turn result in color shifts or fading over time.46
NaOAc NADES system formulations have strong potential for application in food products stored at both room temperature and under refrigeration, irrespective of light exposure, except when distilled water is the solvent. Stability analysis revealed that solvent type was the main factor affecting A/A0 values (Kruskal–Wallis, H = 48.23; df = 2; p < 0.001), while storage temperature had a significant but less pronounced effect (H = 8.95; df = 1; p = 0.003). No significant differences were found between light and dark storage conditions (H = 0.00; df = 1; p = 0.968), confirming the suitability of this system under varying illuminations. An increase in the absorbance ratio A/A0 above 1 may indicate the formation of degradation products that absorb at or near the same wavelength, leading to transient absorbance changes. The instability of colorants in aqueous systems is often attributed to oxygen-mediated and pH-dependent chemical reactions. Many natural pigments or colorants containing conjugated double bonds are prone to oxidation and structural rearrangements in aqueous environments, altering their electronic structure and chromophores, which in turn result in color shifts or fading over time.46
For potential commercial formulations, extraction systems with reduced water content, such as Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) containing 10% water, are recommended. Future studies should explore microemulsion systems to improve dispersion in aqueous environments and investigate encapsulation strategies to enhance the stability of avocado-derived chromophoric compounds under diverse storage conditions. Further stability evaluations over longer durations considering wider variables, including pH range, higher temperatures, and varying water contents, are also recommended.
1) containing 10% water, are recommended. Future studies should explore microemulsion systems to improve dispersion in aqueous environments and investigate encapsulation strategies to enhance the stability of avocado-derived chromophoric compounds under diverse storage conditions. Further stability evaluations over longer durations considering wider variables, including pH range, higher temperatures, and varying water contents, are also recommended.
3.4 Determination of total polyphenol content (TPC)
In this study, the highest polyphenol content in the CASE samples was observed for the Hass variety, followed by the Fuerte variety, when Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was used as the extraction solvent. The corresponding values were 203.45 and 171.92 mg GAE/100 g fresh weight (FW), respectively, calculated based on the final sample resuspensions at a concentration of 2 mg mL−1, and were significantly higher than those obtained using the other solvents evaluated (Fig. 7). A similar trend was reported previously, where avocado peel extracts obtained with a eutectic solvent composed of choline chloride
1) was used as the extraction solvent. The corresponding values were 203.45 and 171.92 mg GAE/100 g fresh weight (FW), respectively, calculated based on the final sample resuspensions at a concentration of 2 mg mL−1, and were significantly higher than those obtained using the other solvents evaluated (Fig. 7). A similar trend was reported previously, where avocado peel extracts obtained with a eutectic solvent composed of choline chloride![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) lactic acid (2
lactic acid (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) exhibited a high polyphenol content of 92.03 ± 2.11 mg GAE per g, while extraction with 96% ethanol yielded a significantly lower value of 25.27 ± 0.92 mg GAE per g.47
1) exhibited a high polyphenol content of 92.03 ± 2.11 mg GAE per g, while extraction with 96% ethanol yielded a significantly lower value of 25.27 ± 0.92 mg GAE per g.47
The highest polyphenol concentrations in the CASE samples were thus achieved using Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES. This solvent can dissolve and extract a wide range of natural compounds, including anthocyanins, xanthones, curcumins, and various phenolic constituents. Owing to their tunable polarity profiles, certain synthetic compounds can also be solubilized.24 Therefore, solvent selection is a critical factor governing the efficiency of phenolic compound extraction. Parameters such as eutectic composition, pH, and viscosity strongly influence extraction performance. In a study focused on recovering phenolics from AS, several NADESs—based on choline chloride, betaine, fructose, glucose, lactic acid, and glycerol—were systematically evaluated. Among the different extraction techniques tested, pressurized liquid extraction using choline chloride
1) NADES. This solvent can dissolve and extract a wide range of natural compounds, including anthocyanins, xanthones, curcumins, and various phenolic constituents. Owing to their tunable polarity profiles, certain synthetic compounds can also be solubilized.24 Therefore, solvent selection is a critical factor governing the efficiency of phenolic compound extraction. Parameters such as eutectic composition, pH, and viscosity strongly influence extraction performance. In a study focused on recovering phenolics from AS, several NADESs—based on choline chloride, betaine, fructose, glucose, lactic acid, and glycerol—were systematically evaluated. Among the different extraction techniques tested, pressurized liquid extraction using choline chloride![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) fructose and choline chloride
fructose and choline chloride![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) lactic acid provided the highest total phenolic content (TPC) values.27
lactic acid provided the highest total phenolic content (TPC) values.27
3.5 Antioxidant activity
In our study, the highest antioxidant activity for a CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) system sample was 3980.33 µmol copper per mL reducing equivalents for Fuerte extract, and 1980.33 µmol copper per mL reducing equivalents for Hass variety, as determined by the TAC assay (Fig. 8). However, it should be noted that TROLOX displays a faster increase in antioxidant concentration than uric acid, and the ORAC assay is generally considered more sensitive than the TAC assay. Dabas et al. (2013)48 reported an antioxidant activity of 2012.1 ± 300 TE per mg for CASE samples extracted with methanol using the ORAC assay. Similarly, Segovia et al. (2016)49 applied the same assay and obtained a value of 350 mg TE per g dry weight from polyphenols extracted from avocado seeds.
1) system sample was 3980.33 µmol copper per mL reducing equivalents for Fuerte extract, and 1980.33 µmol copper per mL reducing equivalents for Hass variety, as determined by the TAC assay (Fig. 8). However, it should be noted that TROLOX displays a faster increase in antioxidant concentration than uric acid, and the ORAC assay is generally considered more sensitive than the TAC assay. Dabas et al. (2013)48 reported an antioxidant activity of 2012.1 ± 300 TE per mg for CASE samples extracted with methanol using the ORAC assay. Similarly, Segovia et al. (2016)49 applied the same assay and obtained a value of 350 mg TE per g dry weight from polyphenols extracted from avocado seeds.
In our study, both the avocado variety and the extraction solvent significantly influenced the antioxidant capacity (Kruskal–Wallis, p < 0.05) (Fig. 8). The Fuerte variety exhibited higher antioxidant activity than the Hass variety (p = 0.001). Similarly, extracts obtained using NADES showed significantly greater antioxidant capacity than those produced with ethanol and distilled water (p < 0.001), with no significant differences between the latter two solvents. In contrast, Dabas et al. (2013)48 reported that the AS Hass variety had a higher phenolic content than the AS Fuerte variety (51.6 ± 1.6 vs. 31.5 ± 2.2 mg GAE per g). The same research group observed that colored extracts contained 22.5% less phenolic content than non-colored extracts, supporting the notion that polyphenols largely contribute to the functional properties of avocado seeds.47
The relationship between absorbance at 480 nm and antioxidant capacity was not strictly linear but was significantly affected by both solvent and avocado seed variety (Fig. 9). In aqueous extracts, relatively high absorbance values were associated with low antioxidant activity, suggesting that color intensity alone does not reliably reflect bioactivity. In contrast, extracts obtained using Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES displayed the highest antioxidant capacity for both varieties, even when absorbance values were comparable to or lower than those of ethanol extracts. These findings indicate that eutectic solvents enhance polyphenol extraction efficiency and contribute to preserving antioxidant functionality.
1) NADES displayed the highest antioxidant capacity for both varieties, even when absorbance values were comparable to or lower than those of ethanol extracts. These findings indicate that eutectic solvents enhance polyphenol extraction efficiency and contribute to preserving antioxidant functionality.
Mouratoglou et al. (2016)23 reported similar trends and observed that eutectic solvents yielded extracts with higher phenolic content than those obtained using ethanol and water.
3.6 Cytotoxicity
Both glycerol and sodium acetate are generally regarded as non-toxic when used at low concentrations. However, it remains essential to evaluate the toxicity of Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES synthesized in this study, along with its potential effects when incorporated into food matrices, to ensure product safety. In this study, the cytotoxicity of representative extracts was assessed.
1) NADES synthesized in this study, along with its potential effects when incorporated into food matrices, to ensure product safety. In this study, the cytotoxicity of representative extracts was assessed.
For the CASE sample from the Fuerte variety, concentrations ranging from 0.0005 to 5 mg mL−1 were tested. No significant differences in cell viability were observed among the concentrations in either avocado variety when compared to the control after 24 h and 48 h of exposures (Fig. 10).
 | ||
| Fig. 10 Cytotoxicity of the Persea americana (Fuerte variety) seed extracts in human dermal fibroblasts evaluated by the MTT assay. | ||
The cells were treated with aqueous or ethanolic extracts at concentrations of 5, 0.5, 0.05, 0.005, and 0.0005 mg mL−1 for 24 h and 48 h. Data are presented as fold changes in absorbance relative to the untreated control (CTL). The results indicate that neither aqueous nor ethanolic extracts produced significant cytotoxic effects under the experimental conditions tested.
For the CASE sample from the Hass variety (Fig. 11), aqueous extracts exhibited non-cytotoxic effects after 24 h and 48 h of exposure, while ethanolic extracts were non-cytotoxic only at 24 h. After 48 h of exposure, ethanolic extracts induced cytotoxicity at concentrations above 0.5 mg mL−1, leading to a modest reduction in cell viability. This cytotoxicity observed at 48 h may be attributed to bioactive phytochemicals (polyphenols and carotenoids) that promote apoptosis and necrosis via oxidative stress, disruption of cell division, and cellular damage, particularly at higher extract concentrations. These responses are consistent with cell death processes and reduced cell proliferation. In the case of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system, no cytotoxic effects were detected, and cell viability remained comparable to that of the control.
NaOAc NADES system, no cytotoxic effects were detected, and cell viability remained comparable to that of the control.
Previous in vitro and in vivo studies have reported multiple health-promoting properties of CASE, including antimicrobial, anticancer, anti-obesity, antioxidant, antidiabetic, and anti-inflammatory activities. Regarding toxicity, Ozolua et al. (2009)50 evaluated aqueous extracts of the AS administered orally to rats at doses of 2–10 g kg−1 for two weeks. No behavioral changes, such as depression, squirming, diarrhea, or aggression, were observed. Similarly, sub-chronic toxicity testing at a dose of 2.5 g kg−1 for 28 days did not result in significant alterations in hematological parameters and plasma biomarkers of hepatotoxicity remained within normal limits. Nevertheless, the authors emphasized the need for further studies to confirm safety in humans. The green extraction methodology proposed in this work represents a promising approach for the safe incorporation of CASE into functional food formulations.
3.7 Potential of Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) NAOAc NADES/CASE inclusion in functional foods: benefits and challenges
NAOAc NADES/CASE inclusion in functional foods: benefits and challenges
Color plays a critical role in consumers' perception and experience of foods, influencing not only visual appeal but also expectations of freshness, flavors and sensory quality, including perceived sweetness and sourness.51 In fruits and fruit-based products, consumer preferences are particularly linked to the intended mode of consumption. Approximately 65% of health-conscious consumers report being highly sensitive to the color and nutritional value of fruits, believing that fresh fruits provide natural nutrients and antioxidants that promote health and help prevent diseases. In comparison, 58% of consumers identify safety and taste as the primary drives of purchase decisions,52 highlighting the strong influence of color on food acceptance and selection.
Color also serves as a key predictor of taste expectations. For instance, red is often associated with ripeness and sweetness, yellow and green with sourness, white with saltiness, and black or brown with bitterness. These associations extend beyond the food itself to external factors, such as packaging, labeling, and serving plates, all of which can modulate consumer perception and desirability.
Cultural factors profoundly shape the significance of color in relation to taste, health perception, and consumer preferences, resulting in considerable variations across food traditions and regions worldwide. Individual characteristics, including gender, age, and body mass index, influence how color is perceived as an indicator of flavor and quality. Overall, color acts as a multisensory cue directing judgements about flavor, freshness, ripeness, nutritional value, and overall food quality, even prior to tasting.53
In the food industry, colorants, including dyes and pigments, are extensively employed to enhance the visual appeal of both solid and liquid products. Typical applications include dairy items, batters, processed meats, fruits, and canned or emulsified foods, such as sauces, juices, and soups. These colorants may be synthetic or naturally derived, with selection governed by stability, regulatory approval, and desired visual effects. For example, annatto (E160b) is commonly used in dairy products to impart a yellow-orange hue to cheese, butter and margarine; carotenoids also find frequent application.54 Synthetic dyes such as tartrazine (FD and C yellow no. 5), sunset yellow FCF (yellow no. 6) and Allura red AC (red no. 40) are widely included in flavored yogurts, ice creams, puddings and desserts. Betaine, a pigment obtained from beetroot, provides pink-to-reddish tones in dairy desserts but exhibits limited stability due to heat and light sensitivity.
Other synthetic food colorants, such as brilliant blue FCF (blue 1), fast green FCF (green 3), and erythrosine (red 3), are routinely incorporated into icings, decorative gels, and batters. However, synthetic colorants are subject to growing health concerns linked to potential allergenic, mutagenic or carcinogenic effects. Notably, the U.S. Food and Drug Administration (FDA) has banned erythrosine (red 3) owing to evidence of mutagenic and carcinogenic risks, heightening the demand for safe natural alternatives. The increasing consumer preference for clean-label products has reinforced the market shift for natural colorants sourced primarily from plants and agricultural by-products. Although natural alternatives improve consumer perceptions and align with sustainability objectives, they generally exhibit lower stability than synthetic colorants and remain under continuous safety and regulatory assessment.
Regarding the practical applications of glycerol-based NADES, successful extractions have been reported at temperatures of up to 135 °C for up to 45 min.55 However, the thermal stability of the specific CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system – defined as the onset temperature of decomposition determined by thermal gravimetric analysis (TGA) and differential scanning calorimetry (DSC) – has not yet been evaluated. This parameter is crucial if this eutectic system is to be considered for incorporation in food formulations, particularly in thermally processed or baked products. Since the CASE/Gly
NaOAc NADES system – defined as the onset temperature of decomposition determined by thermal gravimetric analysis (TGA) and differential scanning calorimetry (DSC) – has not yet been evaluated. This parameter is crucial if this eutectic system is to be considered for incorporation in food formulations, particularly in thermally processed or baked products. Since the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system has not undergone drying, lyophilization, purification or further processing, a more comprehensive toxicological assessment is required before it can be proposed as a ready-to-use food ingredient. Moreover, the replacement of Red 3 (erythrosine) with this system would only be feasible if the dye or pigment responsible for the reddish/orange coloration is properly isolated, purified, characterized, and confirmed to be safe for consumption.
NaOAc NADES system has not undergone drying, lyophilization, purification or further processing, a more comprehensive toxicological assessment is required before it can be proposed as a ready-to-use food ingredient. Moreover, the replacement of Red 3 (erythrosine) with this system would only be feasible if the dye or pigment responsible for the reddish/orange coloration is properly isolated, purified, characterized, and confirmed to be safe for consumption.
In this study, preliminary tests were conducted by incorporating the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system into three commercial brands of natural yogurt to assess its potential as a natural colorant. A noticeable change was observed following the addition of the extract (data are not shown), which shifted from whitish to yellowish-orange, suggesting that CASEs could serve as a potential natural colorant for dairy products. UV-vis spectroscopic analysis (400–700 nm) revealed distinct absorption peaks corresponding to each treated sample, confirming pigment incorporation and color enhancement. Control yogurts without extract did not exhibit significant absorption peaks in this region. Visually, all three yogurts exhibited a comparable orange hue after the addition of the CASE/Gly
NaOAc NADES system into three commercial brands of natural yogurt to assess its potential as a natural colorant. A noticeable change was observed following the addition of the extract (data are not shown), which shifted from whitish to yellowish-orange, suggesting that CASEs could serve as a potential natural colorant for dairy products. UV-vis spectroscopic analysis (400–700 nm) revealed distinct absorption peaks corresponding to each treated sample, confirming pigment incorporation and color enhancement. Control yogurts without extract did not exhibit significant absorption peaks in this region. Visually, all three yogurts exhibited a comparable orange hue after the addition of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system.
NaOAc NADES system.
Dabas et al. (2013) reported that methanolic extracts of avocado seeds imparted a pale orange coloration to milk, an effect attributed to the acidic pH of yogurt (4.25).48 In the present study, the density of yogurt increased slightly from 1.040 ± 0.002 to 1.050 ± 0.002 g mL−1 likely due to the relatively higher density of the ready-to-use CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system formulation. The pH of all samples remained stable, with no statistically significant differences between control and treated yogurts (p > 0.05), and within the acceptable range for this type of product.
NaOAc NADES system formulation. The pH of all samples remained stable, with no statistically significant differences between control and treated yogurts (p > 0.05), and within the acceptable range for this type of product.
Overall, these results demonstrate that the incorporation of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system into yogurt effectively modifies the visual characteristics of the product without adversely affecting its physical parameters, supporting its potential application as a natural colorant for functional foods. Future research should include sensory and consumer preference studies (flavor, aroma, and overall acceptance), comprehensive colorimetric analyses, and evaluations of the thermal and light photostability of the CASE/Gly
NaOAc NADES system into yogurt effectively modifies the visual characteristics of the product without adversely affecting its physical parameters, supporting its potential application as a natural colorant for functional foods. Future research should include sensory and consumer preference studies (flavor, aroma, and overall acceptance), comprehensive colorimetric analyses, and evaluations of the thermal and light photostability of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system formulations under extended storage conditions.
NaOAc NADES system formulations under extended storage conditions.
3.8 Market potential
Avocado production is projected to reach 12 million tons by 2030, driven by the fruit's well-documented health benefits and versatile industrial applications that sustain strong consumer demand.56 According to the OECD-FAO Agricultural Outlook 2021–2030,57 avocados are expected to become the second most traded tropical fruit worldwide, following bananas. The United States and Europe will remain the leading importers, accounting for approximately 40% and 31% of global imports, respectively. Meanwhile, demand in China is increasing steadily, with Mexico, Perú, Chile, Kenya and Tanzania emerging as its main suppliers.The market potential for avocado residue-based biorefinery products is substantial and rapidly expanding, driven by both the growing global consumption of avocados and the transition toward sustainable circular bioeconomy models. By-products from avocado processing can be valorized into a diverse range of high-value products and materials suitable for various sectors. In the food and beverage industry, these include bioactive additives such as antioxidants, polyphenols and dietary fibers, which can be used as natural colorants, food stabilizers and nutritional enhancers in soups, beverages, baked goods, and snacks. Functional derivatives from avocado, such as avocado proteins, supplements or emulsifiers, are used in processed foods and meat-based products.58
Beyond the food sector, AS residues and by-products offer opportunities in pharmaceuticals and nutraceuticals. Their bioactive compounds and extracts with antioxidant, anti-inflammatory, and antimicrobial properties support the development of therapeutic products, dietary supplements, drug delivery systems and tissue engineering materials. The cosmeceutical and personal care industries likewise benefit from AS-derived cosmetic ingredients, functional lipids, oils, and extracts that exhibit antioxidative and hydrating functions, contributing to skincare, haircare, and anti-aging formulations. Furthermore, recent advances show that AS extracts can serve as precursors for advanced nanomaterials, such as carbon dots and nanoparticles with promising applications in functional foods, targeted drug delivery, and environmental remediation.58 The efficient utilization of these bioresources, combined with increasing consumer demand for natural ingredients and the global shift toward sustainability, is accelerating the adoption of avocado-derived materials across multiple industries.
The avocado biorefinery market is expected to reach $105.8 billion by 2032, supported by a robust compound annual growth rate (CAGR) reflecting an expansion of avocado-based products and materials.59 Growth is particularly notable in regions with regulatory frameworks that encourage sustainable production and the use of bio-based ingredients. Among these opportunities, the deep orange-reddish colorant extracted from AS (CASE) shows strong market potential as a novel natural colorant, aligned with global trends favoring clean-label and eco-friendly food additives. However, its successful commercialization depends on process optimization and the technological readiness level (TRL) of NADES-assisted extraction at scale, alongside comprehensive toxicological and safety evaluations and customer acceptance.
3.9 Scalability
The scalability of AS extraction using NADESs appears highly promising for industrial applications, supported by both technical feasibility and environmental benefits. Key factors influencing scale-up include extraction efficiency, process optimization and solid-to-solvent ratio. Current studies indicate that NADESs frequently outperform conventional organic solvents in terms of extraction yield and the retention of bioactive properties in CASEs. Optimization of critical parameters, such as temperature, residence time, and water content, can be efficiently achieved using statistical experimental designs, including Box–Behnken models. Several reports demonstrate that scalable extraction at moderate temperatures (approximately 70 °C), lower water contents (around 10%), and short residence times (≤2 h) is feasible while maintaining high product quality. Moreover, solid-to-solvent ratios and mixing strategies can be tailored for both batch and continuous flow configurations.The environmental performance of this process – driven by its safety, eco-friendliness, and solvent recyclability – combined with the valorization of avocado processing bioresidues and by-products, supports its environmental and economic viability. The use of inexpensive, non-toxic and biodegradable NADESs establishes a sustainable extraction platform for recovering bioactive phytochemicals from ASs and other tropical fruit by-products.60 NADESs can typically be recycled within the extraction cycle, enabling large-scale implementation while minimizing solvent consumption and operational costs. From a biorefinery perspective, this approach is straightforward, readily adaptable, and facilitates the recovery of high-value CASEs with potential applications as natural food colorants. Additionally, the remaining starch-rich solids can undergo enzymatic hydrolysis, integrating seamlessly into cascade biorefinery schemes and maximizing AS biomass valorization.
Nonetheless, several limitations persist. The high intrinsic viscosity of NADESs can impede mass transfer during large-scale operations. These limitations can be alleviated by modulating the water content or employing process intensification techniques, such as mechanical agitation, ultrasound, or microwave-assisted extraction. Although the direct application of NADES/CASE systems appears promising, further toxicological and safety evaluations are necessary to comply with current regulatory frameworks. Moreover, if pigment recovery from the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system as a powder is desired, scalable separation methods, such as antisolvent precipitation or membrane filtration, should be developed.
NaOAc NADES system as a powder is desired, scalable separation methods, such as antisolvent precipitation or membrane filtration, should be developed.
In terms of industrial feasibility, successful upscaling requires extensive laboratory and pilot-scale validation as well as compliance with regulatory standards and market acceptance. In summary, the use of food grade, non-toxic constituents in the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES systems demonstrates high scalability and significant application potential in the food, nutraceutical and cosmeceutical industries. The technology combines high extraction efficiency, operational adaptability, economic sustainability and environmental safety, positioning it as a viable strategy for the large-scale valorization of AS by-products.
NaOAc NADES systems demonstrates high scalability and significant application potential in the food, nutraceutical and cosmeceutical industries. The technology combines high extraction efficiency, operational adaptability, economic sustainability and environmental safety, positioning it as a viable strategy for the large-scale valorization of AS by-products.
3.10 Waste management, environmental impact, circular economy gaps, and sustainable development
The global avocado industry is experiencing rapid expansion, resulting in the generation of a significant amount of bioresidues and by-products, mainly seeds and peels. The seed alone accounts for approximately 16% of the total fruit mass, generating substantial quantities of agro-industrial bioresidues worldwide. The disposal of these bulky residues poses significant environmental challenges. Unmanaged organic waste decomposes in landfills, releasing methane and contributing to greenhouse gas emissions, while the accumulation of seeds and peels can cause local pollution, unpleasant odors, and pest proliferation. Furthermore, avocado cultivation is linked to deforestation from land clearing, soil degradation caused by intensive monoculture practices, water scarcity due to high irrigation demands, and biodiversity loss associated with the extensive use of agrochemicals.61These environmental concerns underscore the urgency of developing sustainable strategies for managing avocado residues to prevent ecological degradation, minimize reliance on fossil-based resources and enhance the economic value of agro-industrial by-products. The valorization of these residues aligns with circular bioeconomy principles and fosters sustainability throughout the value chain.
The conversion of avocado bioresidues (seeds and peels) into value-added products through integrated cascade biorefineries provides an eco-friendly pathway to close nutrient and material loops. This approach transforms what would otherwise be solid-waste streams (∼35–40% of fresh fruit mass) into high-value coproducts, thereby strengthening circularity within the avocado supply chain. Moreover, this valorization pathway contributes to several United Nations Sustainable Development Goals (SDGs):
• SDG 2 – zero hunger: the development of new value chains can improve the productivity and competitiveness of the avocado processing industry while enhancing micronutrient availability for vulnerable populations.
• SDG 3 – good health and well-being: bioactive compounds extracted from ASs (e.g. antioxidants and antimicrobials) support the prevention of diet-related non-communicable diseases.
• SDG 8 – decent work and economic growth: the establishment of biorefineries fosters local employment by creating opportunities for processors, smallholder farmers and operators in rural regions, both near cultivation areas and adjacent processing facilities.
• SDG 9 – industry innovation and infrastructure: scalable biorefinery platforms promote technological advancement and modernization across the agro-industrial sector.
• SDG 12 – responsible consumption and production: the cascade use of pulp, peel and seeds enables the reintegration of over 90% of the original biomass into productive cycles, significantly reducing waste generation.
• SDG 15 – life on land: sustainable residue management practices help mitigate soil pollution, preserve biodiversity, and reduce the ecological footprint of avocado production.
Through these strategies, the avocado industry can transition towards a more sustainable and circular model, promoting environmental protection while delivering socioeconomic benefits.
4 Conclusions
In this study, the potential of CASEs obtained from Fuerte and Hass varieties was evaluated using three solvents: distilled water, 70% ethanol, and a glycerol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sodium acetate natural deep eutectic solvent (Gly
sodium acetate natural deep eutectic solvent (Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc 3
NaOAc 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NADES). The starting material for extraction consisted of a homogeneous avocado seed paste, which was exposed to air for 35 min to promote the action of polyphenol oxidase (PPO) enzymes responsible for producing brown and yellow-red pigments in fruits and vegetables. The Gly
1 NADES). The starting material for extraction consisted of a homogeneous avocado seed paste, which was exposed to air for 35 min to promote the action of polyphenol oxidase (PPO) enzymes responsible for producing brown and yellow-red pigments in fruits and vegetables. The Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) NADES was synthesized using a simple heating method. This solvent system resulted in significantly higher extraction yields than those previously reported. Percolation extraction using NADES produced a CASE/Gly
1) NADES was synthesized using a simple heating method. This solvent system resulted in significantly higher extraction yields than those previously reported. Percolation extraction using NADES produced a CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc (3
NaOAc (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) system as a viscous reddish-orange liquid, achieving yields of 88.6% for the Hass variety and 88.2% for the Fuerte variety. In contrast, aqueous and ethanolic extractions yielded lower values, consistent with previous reports.
1) system as a viscous reddish-orange liquid, achieving yields of 88.6% for the Hass variety and 88.2% for the Fuerte variety. In contrast, aqueous and ethanolic extractions yielded lower values, consistent with previous reports.
The color intensity of the CASEs, evaluated via absorbance measurements, varied considerably across the 400–500 nm spectral range, aligning with reddish-orange hues, as previously reported. Although the underlying mechanism of color formation remains unresolved, both kinetic and pH stability analyses indicate that the coloration in the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system does not originate from anthocyanins, which are unstable at elevated pH levels and have not been detected in avocado seed extracts. Notably, the CASE/Gly
NaOAc NADES system does not originate from anthocyanins, which are unstable at elevated pH levels and have not been detected in avocado seed extracts. Notably, the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system demonstrates stability throughout the pH range characteristic of food products (pH 3–7), positioning it as a promising alternative for food industry applications.
NaOAc NADES system demonstrates stability throughout the pH range characteristic of food products (pH 3–7), positioning it as a promising alternative for food industry applications.
The highest total phenolic content (TPC) was observed in the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system of the Hass variety, followed by that of the Fuerte variety. The Gly
NaOAc NADES system of the Hass variety, followed by that of the Fuerte variety. The Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES, used in this study effectively dissolved and extracted a broad array of natural compounds, particularly polyphenolics, confirming previous observations for other NADES. Analogously, the CASE/Gly
NaOAc NADES, used in this study effectively dissolved and extracted a broad array of natural compounds, particularly polyphenolics, confirming previous observations for other NADES. Analogously, the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system yielded the highest antioxidant activities, with the Fuerte variety exhibiting superior antioxidant capacity. These findings suggest that eutectic solvents enhance polyphenol extraction efficiency and contribute to preserving antioxidant functionality.
NaOAc NADES system yielded the highest antioxidant activities, with the Fuerte variety exhibiting superior antioxidant capacity. These findings suggest that eutectic solvents enhance polyphenol extraction efficiency and contribute to preserving antioxidant functionality.
Regarding cytotoxicity, the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc NADES system from the Hass variety showed no detectable cytotoxic effects and did not significantly reduce cell viability. Similarly, all CASEs from both varieties were non-cytotoxic at concentrations below 5 mg mL−1 after 24 h of exposure. Most samples remained non-cytotoxic after 48 h, with the exception of the ethanolic extract of the Hass variety. Overall, no substantial cytotoxic differences were observed between the two avocado varieties.
NaOAc NADES system from the Hass variety showed no detectable cytotoxic effects and did not significantly reduce cell viability. Similarly, all CASEs from both varieties were non-cytotoxic at concentrations below 5 mg mL−1 after 24 h of exposure. Most samples remained non-cytotoxic after 48 h, with the exception of the ethanolic extract of the Hass variety. Overall, no substantial cytotoxic differences were observed between the two avocado varieties.
Further toxicological evaluation is recommended to confirm the safety of these extracts for food and cosmetic applications. Overall, CASEs represent a sustainable, non-toxic, and eco-friendly alternative to synthetic dyes, contributing to the circular bioeconomy and supporting sustainable development goals. Future studies should include both qualitative and quantitative characterization of the chemical composition of the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc system, together with sensory analyses to assess its performance in final products, such as yogurt, a non-alcoholic beverage, and other consumer goods. In addition, a comprehensive regulatory assessment is required to ensure compliance with standards for certified food color additives. Finally, process optimization, scale-up strategies, and performance validation are essential steps before the CASE/Gly
NaOAc system, together with sensory analyses to assess its performance in final products, such as yogurt, a non-alcoholic beverage, and other consumer goods. In addition, a comprehensive regulatory assessment is required to ensure compliance with standards for certified food color additives. Finally, process optimization, scale-up strategies, and performance validation are essential steps before the CASE/Gly![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaOAc system can be considered for industrial application as a natural food colorant.
NaOAc system can be considered for industrial application as a natural food colorant.
Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.Data availability
All data are in the manuscript.Acknowledgements
We wish to extend our heartfelt appreciation to the laboratories of Plant Biotechnology, Animal Biotechnology, and Agricultural Biotechnology, and the Chemical Engineering Department at Universidad San Francisco de Quito, and to their respective directors and personnel. We thank them for providing the essential equipment and reagents required to conduct this research. Verónica Castañeda thanks the ANID-Subdirección de Capital Humano/Doctorado Nacional for their support and FONDECYT 11190998; Proyecto Centro Basal ANID IMPACT:FB210024. During the preparation of this work, the author(s) used Perplexity AI in order to revise grammar and scientific style. After using this tool/service, the author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the published article.References
- J. J. Á. Flores, H. V. Cevallos, V. J. G. Montealegre and H. C. Romero, Análisis de la producción de aguacate en el Ecuador y su exportación a mercados internacionales en el periodo 2008 al 2018, Rev. Metrop. Cienc. Apl., 2021, 164–175 Search PubMed , Available from: https://remca.umet.edu.ec/index.php/REMCA/article/view/424.
- M. A. Tremocoldi, P. L. Rosalen, M. Franchin, A. P. Massarioli, C. Denny and É. R. Daiuto, et al., Exploration of avocado by-products as natural sources of bioactive compounds, PLoS One, 2018, 13(2), 1–12 CrossRef PubMed.
- A. Gürses, M. Açıkyıldız, K. Güneş and E. Şahin, Natural dyes and pigments in food and beverages, in Renewable Dyes and Pigments [Internet], Elsevier, 2024, pp. 49–76, Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780443152139000132 Search PubMed.
- D. A. S. Wibisono, C.-Y. Saw, T.-Y. Wu and C.-F. Chau, Advancing Industrial Food Byproduct Management: Strategies, Technologies, and Progress in Waste Reduction, Processes, 2025, 13(1), 84 CrossRef CAS , Available from: https://www.mdpi.com/2227-9717/13/1/84.
- L. M. Orejuela-Escobar, A. C. Landázuri and B. Goodell, Second generation biorefining in Ecuador: Circular bioeconomy, zero waste technology, environment and sustainable development: The nexus, J. Bioresour. Bioprod., 2021, 1–25 Search PubMed.
- B. Rodríguez-Martínez, A. Romaní, G. Eibes, G. Garrote, B. Gullón and P. G. del Río, Potential and prospects for utilization of avocado by-products in integrated biorefineries, Bioresour. Technol., 2022, 364, 128034 CrossRef PubMed , Available from: https://linkinghub.elsevier.com/retrieve/pii/S0960852422013670.
- J. A. Dávila, M. Rosenberg, E. Castro and C. A. Cardona, A model biorefinery for avocado (Persea americana mill.) processing, Bioresour. Technol., 2017, 243, 17–29 CrossRef PubMed.
- S. P. Bangar, K. Dunno, S. B. Dhull, A. Kumar Siroha, S. Changan and S. Maqsood, et al., Avocado seed discoveries: Chemical composition, biological properties, and industrial food applications, Food Chem.:X, 2022, 16, 100507 CAS , Available from: https://linkinghub.elsevier.com/retrieve/pii/S2590157522003054.
- D. R. Joshi and N. Adhikari, An Overview on Common Organic Solvents and Their Toxicity, J. Pharm. Res. Int., 2019, 28(3), 1–18 Search PubMed.
- C. Capello, U. Fischer and K. Hungerbühler, What is a green solvent? A comprehensive framework for the environmental assessment of solvents, Green Chem, 2007, 9(9), 927 RSC , Available from: https://xlink.rsc.org/?DOI=b617536h.
- W. Wang, T. R. Bostic and L. Gu, Antioxidant capacities, procyanidins and pigments in avocados of different strains and cultivars, Food Chem., 2010, 122(4), 1193–1198 CrossRef CAS , Available from: https://linkinghub.elsevier.com/retrieve/pii/S0308814610004127.
- D. Dabas, R. J. Elias, J. D. Lambert and G. R. Ziegler, A Colored Avocado Seed Extract as a Potential Natural Colorant, J. Food Sci., 2011, 76(9), C1335–C134 CrossRef CAS PubMed , Available from: https://ift.onlinelibrary.wiley.com/doi/10.1111/j.1750-3841.2011.02415.x.
- G. R. Velderrain-Rodríguez, J. Quero, J. Osada, O. Martín-Belloso and M. J. Rodríguez-Yoldi, Phenolic-Rich Extracts from Avocado Fruit Residues as Functional Food Ingredients with Antioxidant and Antiproliferative Properties, Biomolecules, 2021, 11(7), 977 CrossRef PubMed , Available from: https://www.mdpi.com/2218-273X/11/7/977.
- D. Suárez, O. Marín, S. N. Dueñas, A. J. Ortiz, A. E. Puentes, A. Mejía, R. M. Suárez, S. Sierra and D. C. Zuluaga, Evaluation of the extraction conditions of a natural dye from avocado seeds, Agron. Colomb., 2016, 34(1Supl), S241–S243 Search PubMed.
- R. G. Araújo, R. M. Rodriguez-Jasso, H. A. Ruiz, M. Govea-Salas, M. E. Pintado and C. N. Aguilar, Process optimization of microwave-assisted extraction of bioactive molecules from avocado seeds, Ind. Crops Prod., 2020, 154, 112623 CrossRef , Available from: https://linkinghub.elsevier.com/retrieve/pii/S0926669020305392.
- H. T. Hue, N. M. Thai and P. T. A. Dao, Survey on different polyphenol extraction methods from avocado seed ( Persea americana Mill) and assay on their antioxidant and acute toxicity activities, Vietnam J. Chem., 2021, 59(6), 870–876 CrossRef CAS , Available from: https://onlinelibrary.wiley.com/doi/10.1002/vjch.202100060.
- E. S. Ong, J. Low, J. C. W. Tan, S. Y. Foo and C. H. Leo, Valorization of avocado seeds with antioxidant capacity using pressurized hot water extraction, Sci. Rep., 2022, 12(1), 13036 CrossRef CAS PubMed , Available from: https://www.nature.com/articles/s41598-022-17326-5.
- K. Kupnik, M. Primožič, V. Kokol, Ž. Knez and M. Leitgeb, Enzymatic, Antioxidant, and Antimicrobial Activities of Bioactive Compounds from Avocado (Persea americana L.) Seeds, Plants, 2023, 12(5), 1201 CrossRef CAS PubMed , Available from: https://www.mdpi.com/2223-7747/12/5/1201.
- Y. Dai, J. van Spronsen, G. J. Witkamp, R. Verpoorte and Y. H. Choi, Natural deep eutectic solvents as new potential media for green technology, Anal. Chim. Acta, 2013, 766, 61–68 CrossRef CAS PubMed.
- R. F. González-Laredo, V. I. Sayago-Monreal, M. R. Moreno-Jiménez, N. E. Rocha-Guzmán, J. A. Gallegos-Infante and L. F. Landeros-Macías, et al., Natural deep eutectic solvents as an emerging technology for the valorisation of natural products and agro-food residues: a review, Int. J. Food Sci. Technol., 2023, 58(12), 6660–6673 CrossRef , Available from: https://academic.oup.com/ijfst/article/58/12/6660/7807005.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Deep Eutectic Solvents (DESs) and Their Applications, Chem. Rev., 2014, 114(21), 11060–11082, DOI:10.1021/cr300162p.
- Y. Dai, R. Verpoorte and Y. H. Choi, Natural deep eutectic solvents providing enhanced stability of natural colorants from safflower ( Carthamus tinctorius ), Food Chem., 2014, 159, 116–121, DOI:10.1016/j.foodchem.2014.02.155.
- E. Mouratoglou, V. Malliou and D. P. Makris, Novel Glycerol-Based Natural Eutectic Mixtures and Their Efficiency in the Ultrasound-Assisted Extraction of Antioxidant Polyphenols from Agri-Food Waste Biomass, Waste Biomass Valorization, 2016, 7(6), 1377–1387 CrossRef CAS.
- A. Mortensen, F. Aguilar, R. Crebelli, A. Di Domenico, B. Dusemund and M. J. Frutos, et al., Re-evaluation of glycerol (E 422) as a food additive, EFSA J., 2017, 15(3), 1–64, DOI:10.2903/j.efsa.2017.4720.
- H. Mohammadzadeh-Aghdash, Y. Sohrabi, A. Mohammadi, D. Shanehbandi, P. Dehghan and J. Ezzati Nazhad Dolatabadi, Safety assessment of sodium acetate, sodium diacetate and potassium sorbate food additives, Food Chem., 2018, 257, 211–215 CrossRef CAS PubMed , Available from: https://linkinghub.elsevier.com/retrieve/pii/S0308814618304370.
- A. Del-Castillo-Llamosas, F. Rodríguez-Rebelo, B. Rodríguez-Martínez, A. Mallo-Fraga, P. G. Del-Río and B. Gullón, Valorization of Avocado Seed Wastes for Antioxidant Phenolics and Carbohydrates Recovery Using Deep Eutectic Solvents (DES), Antioxidants, 2023, 12(6), 1156 CrossRef CAS PubMed , Available from: https://www.mdpi.com/2076-3921/12/6/1156.
- J. F. Grisales-Mejía, V. Cedeño-Fierro, J. P. Ortega, H. G. Torres-Castañeda, M. M. Andrade-Mahecha and H. A. Martínez-Correa, et al., Advanced NADES-based extraction processes for the recovery of phenolic compounds from Hass avocado residues: A sustainable valorization strategy, Sep. Purif. Technol., 2024, 351, 128104 CrossRef , Available from: https://linkinghub.elsevier.com/retrieve/pii/S1383586624018434.
- S. Elsayed and H. Elsayed, Boosting the nutritional value of stirred yogurt by adding nano-sized avocado seed powder and Lactobacillus acidophilus, Egypt. J. Chem., 2024, 13–25 Search PubMed , Available from: https://ejchem.journals.ekb.eg/article_336817.html.
- E. Viola, C. Buzzanca, I. Tinebra, L. Settanni, V. Farina and R. Gaglio, et al., A Functional End-Use of Avocado (cv. Hass) Waste through Traditional Semolina Sourdough Bread Production, Foods, 2023, 12(20), 3743 CrossRef CAS PubMed , Available from: https://www.mdpi.com/2304-8158/12/20/3743.
- C. O. Nyakang’i, E. Marete, R. Ebere and J. M. Arimi, Physicochemical Properties of Avocado Seed Extract Model Beverages and Baked Products Incorporated with Avocado Seed Powder, Int. J. Food Sci., 2023, 1–9 Search PubMed , Available from: https://www.hindawi.com/journals/ijfs/2023/6860806/.
- C. Novais, A. K. Molina, R. M. V. Abreu, C. Santo-Buelga, I. C. F. R. Ferreira and C. Pereira, et al., Natural Food Colorants and Preservatives: A Review, a Demand, and a Challenge, J. Agric. Food Chem., 2022, 70(9), 2789–2805, DOI:10.1021/acs.jafc.1c07533.
- T. Turner, Ohio State Scientist Unlocks Natural Food Color Alternatives to Red No. 3 [Internet], Ohio State News, 2025, Available from: https://news.osu.edu/ohio-state-scientist-unlocks-natural-food-color-alternatives-to-red-no-3/ Search PubMed.
- M. Scanu, L. Mancuso and G. Cao, Evaluation of the use of human Mesenchymal Stem Cells for acute toxicity tests, Toxicol. In Vitro, 2011, 25(8), 1989–1995 CrossRef CAS PubMed , Available from: https://linkinghub.elsevier.com/retrieve/pii/S0887233311001962.
- H. Peng, X. Zhang, Y. Wei, W. Liu, S. Li and G. Yu, et al., Cytotoxicity of Silver Nanoparticles in Human Embryonic Stem Cell-Derived Fibroblasts and an L-929 Cell Line, J. Nanomater., 2012, 2012, 1–12, DOI:10.1155/2012/160145.
- C. Pan, C. Kumar, S. Bohl, U. Klingmueller and M. Mann, Comparative Proteomic Phenotyping of Cell Lines and Primary Cells to Assess Preservation of Cell Type-specific Functions, Mol. Cell. Proteomics, 2009, 8(3), 443–450 CrossRef CAS PubMed , Available from: https://linkinghub.elsevier.com/retrieve/pii/S1535947620306095.
- A. C. Landázuri, A. Gualle, V. Castañeda, E. Morales, A. Caicedo and L. M. Orejuela-Escobar, Moringa oleifera Lam. leaf powder antioxidant activity and cytotoxicity in human primary fibroblasts, Nat. Prod. Res., 2020, 1–6 Search PubMed.
- S. W. N. Munthe, R. Riskianto, D. Juvi and J. Novia, Antioxidant, Total Phenolic, and Total Flavonoid of 70% Ethanol Extract of Avocado Seeds (Persea americana Mill.), Pharmacogn. J., 2023, 15(4), 599–605 CrossRef CAS , Available from: https://phcogj.com/article/2087.
- A. Gitelson and A. Solovchenko, Non-invasive quantification of foliar pigments: Possibilities and limitations of reflectance- and absorbance-based approaches, J. Photochem. Photobiol., B, 2018, 178, 537–544 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/abs/pii/S1011134417311594?via%3Dihub.
- S. Gajewska, J. Wróblewska, S. Agnieszka and B. Michalkiewicz, Avocado Seed Waste as a Green Catalyst for the Sustainable Oxidation of Limonene with Molecular Oxygen, Sustainability, 2025, 17(9), 3923 CrossRef CAS , Available from: https://www.mdpi.com/2071-1050/17/9/3923.
- E. Hatzakis, E. P. Mazzola, R. M. Shegog, G. R. Ziegler and J. D. Lambert, Perseorangin: A natural pigment from avocado (Persea americana) seed, Food Chem., 2019, 293, 15–22 CrossRef CAS PubMed , Available from: https://linkinghub.elsevier.com/retrieve/pii/S0308814619307307.
- A. A. Arlene, K. A. Prima, L. Utama and S. A. Anggraini, The Preliminary Study of the Dye Extraction from the Avocado Seed Using Ultrasonic Assisted Extraction, Procedia Chem., 2015, 16, 334–340 CrossRef CAS , Available from: https://linkinghub.elsevier.com/retrieve/pii/S1876619615002090.
- I. L. Tarigan, Green Extraction of Bioactive Compounds from Peronema canescens Jack. Using Sodium Acetate/Glycerol-Based Natural Deep Eutectic Solvents as a Source of Natural Antioxidants, J. Kartika Kim, 2025, 8(1), 60–70 Search PubMed , Available from: https://jkk.unjani.ac.id/index.php/jkk/article/view/864.
- M. E. Khlupova, O. V. Morozova and I. S. Vasil’eva, et al., Polymerization of (+)-Catechin in a Deep Eutectic Solvent Using a Fungal Laccase: Physicochemical Properties of the Products and Inhibition of α-Glucosidase, Appl. Biochem. Microbiol., 2021, 57, 712–718, DOI:10.1134/S0003683821060065.
- D. S. Freitas, A. Ribeiro, A. Cavaco-Paulo and C. Silva, The Versatility of NADES Across Applications, Molecules, 2025, 30(19), 3862 CrossRef CAS PubMed , Available from: https://www.mdpi.com/1420-3049/30/19/3862.
- Ib U. Kalthum, I. Idayu Muha and S. R. Mohd, The Effect of pH on Color Behavior of Brassica oleracea Anthocyanin, J. Appl. Sci., 2011, 11(13), 2406–2410 CrossRef , Available from: https://www.scialert.net/abstract/?doi=jas.2011.2406.2410.
- Z. Chen, J. Zheng and L. A. Zhou, A universal strategy for real-time discrimination and evaluation of food freshness using natural colorants as indicators: from detection to antioxidant and antibacterial properties, npj Sci. Food, 2025, 9(213), 1–16 Search PubMed , Available from: https://www.nature.com/articles/s41538-025-00568-8#Abs1.
- B. Rodríguez-Martínez, P. Ferreira-Santos, I. M. Alfonso, S. Martínez, Z. Genisheva and B. Gullón, Deep Eutectic Solvents as a Green Tool for the Extraction of Bioactive Phenolic Compounds from Avocado Peels, Molecules, 2022, 27(19), 6646 CrossRef PubMed , Available from: https://www.mdpi.com/1420-3049/27/19/6646.
- D. Dabas, R. Shegog, G. Ziegler and J. Lambert, Avocado (Persea americana) Seed as a Source of Bioactive Phytochemicals, Curr. Pharm. Des., 2013, 19(34), 6133–6140 CrossRef CAS PubMed.
- F. J. Segovia, J. J. Corral-Pérez and M. P. Almajano, Avocado seed: Modeling extraction of bioactive compounds, Ind. Crops Prod., 2016, 85, 213–220 CrossRef CAS , Available from: https://linkinghub.elsevier.com/retrieve/pii/S0926669016301364.
- R. Ozolua, O. Anaka, S. Okpo and S. Idogun, Acute and sub-acute toxicological assessment of the aqueous seed extract of Persea americana Mill (Lauraceae) in rats, Afr. J. Tradit., Complementary Altern. Med., 2009, 6(4), 573–578 Search PubMed.
- U. Hoppu, S. Puputti, H. Aisala, O. Laaksonen and M. Sandell, Individual Differences in the Perception of Color Solutions, Foods, 2018, 7(9), 154 CrossRef PubMed , Available from: https://www.mdpi.com/2304-8158/7/9/154.
- Y. Wang, L. Liu and Y. Wei, The Influence of Consumption Purpose on Consumer Preferences for Fruit Attributes: The Moderating Effect of Color Perception, Foods, 2025, 14(11), 1902 CrossRef CAS PubMed , Available from: https://www.mdpi.com/2304-8158/14/11/1902.
- Hunterlab, Effect Color Has on Food Perception, Flavor and Quality [Internet], Hunterlab, 2023, Available from: https://www.hunterlab.com/blog/effect-color-has-on-food-perception-flavor-and-quality/ Search PubMed.
- M. M. Silva, F. H. Reboredo and F. C. Lidon, Food Colour Additives: A Synoptical Overview on Their Chemical Properties, Applications in Food Products, and Health Side Effects, Foods, 2022, 11(3), 379 CrossRef CAS PubMed , Available from: https://www.mdpi.com/2304-8158/11/3/379.
- E. Bozinou, D. Palaiogiannis, V. Athanasiadis, A. Chatzilazarou, S. I. Lalas and D. P. Makris, Glycerol-Based Deep Eutectic Solvents for Simultaneous Organosolv Treatment/Extraction: High-Performance Recovery of Antioxidant Polyphenols from Onion Solid Wastes, Sustainability, 2022, 14(23), 15715 CrossRef CAS , Available from: https://www.mdpi.com/2071-1050/14/23/15715.
- CBI, The European Market Potential for Avocados [Internet], Government of Netherlands, 2024, Available from: https://www.cbi.eu/market-information/fresh-fruit-vegetables/avocados/market-potential Search PubMed.
- OECD-FAO, OECD-FAO Agricultural Outlook 2021-2030 [Internet], OECD, 2021, (OECD-FAO Agricultural Outlook), Available from: https://www.oecd.org/en/publications/oecd-fao-agricultural-outlook-2021-2030_19428846-en.html Search PubMed.
- A. Mora-Sandí, A. Ramírez-González, L. Castillo-Henríquez, M. Lopretti-Correa and J. R. Vega-Baudrit, Persea Americana Agro-Industrial Waste Biorefinery for Sustainable High-Value-Added Products, Polymers, 2021, 13(11), 1727 CrossRef PubMed , Available from: https://www.mdpi.com/2073-4360/13/11/1727.
- Market Insight Digest, Biorefinery Applications Market Size 2025 Overview, Manufacturers, Types, Applications, Share, Growth Rate and Forecast 2032, Mark. Insight Dig., 2025 Search PubMed , Available from: https://www.linkedin.com/pulse/biorefinery-applications-market-size-2025-overview-k0aee/.
- N. Otero-Guzman and R. Andrade-Pizarro, Bioactive compounds from tropical fruit by-products: Extraction, characterization and therapeutic potential, J. Agric. Food Res., 2025, 21, 101983 CAS , Available from: https://linkinghub.elsevier.com/retrieve/pii/S2666154325003540.
- A. Denvir, E. Y. Arima, A. González-Rodríguez and K. R. Young, Ecological and human dimensions of avocado expansion in México: Towards supply-chain sustainability, Ambio, 2022, 51(1), 152–166, DOI:10.1007/s13280-021-01538-6.
| This journal is © The Royal Society of Chemistry 2026 |