 Open Access Article
Open Access ArticleMicroencapsulation of kratom leaf extract via spray drying: impact of inlet temperature and wall materials on stability and shelf life
Supanit
Khongtongsang
a,
Mohammad
Fikry
ab,
Saeid
Jafari
a,
Sochannet
Chheng
 ac,
Isaya
Kijpatanasilp
a and
Kitipong
Assatarakul
ac,
Isaya
Kijpatanasilp
a and
Kitipong
Assatarakul
 *a
*a
aDepartment of Food Technology, Faculty of Science, Chulalongkorn University, 10330, Bangkok, Thailand. E-mail: Kitipong.A@chula.ac.th; Tel: +66-2218-5328
bDepartment of Agricultural and Biosystems Engineering, Faculty of Agriculture, Benha University, Moshtohor, Toukh, 13736, Egypt
cDepartment of Food Chemical Engineering, Kampong Speu Institute of Technology, Kampong Speu 050601, Cambodia
First published on 15th November 2025
Abstract
Microencapsulation offers a sustainable approach to improve the stability, functionality, and shelf life of bioactive compounds in food systems, reducing product waste and enhancing resource efficiency. The objectives of this study were to microencapsulate kratom leaf extract (KLE) using spray drying with gum arabic (GA) and resistant maltodextrin (RMD) at two inlet temperatures (150 and 160 °C), to evaluate the resulting microcapsules in terms of their physicochemical properties and assess bioactive retention, specifically total phenolic and flavonoid contents, as well as antioxidant activity, and to investigate stability and shelf-life prediction using kinetic modeling. The resulting microcapsules were evaluated for physicochemical properties, bioactive retention, antioxidant activity, and storage stability. Encapsulation yield ranged from 45.3–62.7%, while encapsulation efficiency ranged from 70.8–83.5%, with GA at 160 °C showing the highest performance. Moisture content remained within the range of 2.1–3.6%, and solubility ranged from 82.4–92.1%, with RMD providing greater solubility and lower residual moisture. Total phenolic content retention ranged from 65.2–79.6%, while total flavonoid content retention ranged from 61.4–76.8%. Antioxidant activity (DPPH inhibition) decreased by approximately 20% during processing, with GA at 160 °C preserving the highest activity. Storage studies showed that water activity remained below the stability threshold (aw = 0.30), and first-order kinetic modeling predicted a shelf-life extension of up to 90 days under ambient conditions. Morphological analysis revealed spherical particles with smooth surfaces at 160 °C, whereas higher temperatures induced surface collapse and shrinkage. Overall, GA provided superior protection of phenolics and antioxidants, while RMD enhanced solubility and reduced residual moisture. The findings highlight the potential of using food-grade wall materials and optimized spray drying conditions for scalable production of stable KLE microcapsules suitable for functional food and nutraceutical applications.
Sustainability spotlightThis study highlights a sustainable microencapsulation approach using plant-based, biodegradable wall materials (gum arabic and resistant maltodextrin) to enhance the shelf life and stability of kratom leaf extract. By optimizing spray drying conditions, the process achieves efficient bioactive preservation through low-energy input and minimal resource use. The findings promote reduced food waste, extended product usability, and environmentally responsible formulation practices, supporting the advancement of green technologies in functional food and nutraceutical production. |
1 Introduction
Kratom (Mitragyna Speciosa) is a tropical plant belonging to the Rubiaceae family and is indigenous to Southeast Asia.1 For hundreds of years, kratom has been utilized in various regions of Thailand and is known by several local names, including Thom, E-Thang, Ketum, Kratum-Koke, and Maeng Da Leaf.2 Traditionally, fresh kratom leaves are commonly chewed or boiled to create a decoction. In contrast, dried leaves are typically smoked, brewed into tea, or consumed as an herbal drink, often with added honey and lemon.3 Kratom has historically been used to relieve muscle fatigue and tiredness and as an herbal remedy for various common ailments, including diarrhea, diabetes, coughing, and hypertension. Additionally, it has been employed as an alternative to morphine or opium for treating drug addiction.4 Some studies have shown that kratom exhibits a range of biological activities, such as anti-inflammatory, antinociceptive, antioxidant, and antimicrobial properties.5 For these reasons, kratom leaves hold significant economic value, with fresh leaves selling for $8 to $10 per kilogram in Thai markets as of 2022.6Extracting bioactive compounds from kratom leaves can enhance both the commercial value of the raw material and the profitability of its processing. Consequently, kratom leaves have garnered significant attention from researchers exploring both traditional and novel extraction techniques.7–10 However, the stability of these extracted compounds can be compromised during processing and storage due to factors such as solvents, pH, temperature, oxygen, light, and enzymes.11 Hence, finding alternative methods to enhance the stability of these bioactive compounds during processing and storage is crucial.
The stabilization of bioactive compounds can be enhanced using microencapsulation technologies, such as spray drying,12 to improve their suitability for industrial applications and ensure their bioavailability. Spray drying is the preferred method for microencapsulation due to its cost-effectiveness, user-friendliness, and its ability to produce high-quality particles.13
Spray drying microencapsulation is a technique used to protect essential substances from unwanted decomposition or reactions during storage.14 This technology involves embedding active ingredients within microparticle matrices, creating a physical barrier between the active compound and the external environment while regulating its release. By encapsulating a bioactive compound in a biopolymer, the process shields it from oxygen, moisture, and other environmental factors, thus enhancing its stability. Additionally, it converts liquid solutions into powders, making them easier to handle.15
Various encapsulating agents are employed in spray drying, including polysaccharides (such as starch, resistant maltodextrin (RMD), corn syrups, and gum arabic (GA)), lipids (like stearic acid and mono- and diglycerides), and proteins (including gelatin, casein, milk serum, soy, and wheat). These substances are favored for their high-water solubility, low viscosity, neutral taste, and colorless solutions, making them widely utilized in the food industry (Jafari et al., 2023).13
On the other hand, using mathematical models is a highly effective approach to assess quality parameters at various stages of processing. Zero-order and first-order equations are frequently utilized to describe the changes in quality properties over time. Predictive models have been previously developed to evaluate the shelf life of various products by integrating the physical and phytochemical indicators.16
Bioactive compounds from different plant leaves have been previously microencapsulated using spray drying technology.12,14,17–23
Although kratom (Mitragyna speciosa) has been studied for its phytochemical composition and potential bioactivity,10,24–26 no previous reports have investigated its microencapsulation using spray drying. Microencapsulation is crucial for protecting kratom extract from degradation, improving stability, and facilitating reconstitution into functional products. However, knowledge remains limited regarding how different wall materials and processing conditions influence these outcomes. Therefore, the objectives of this study were to microencapsulate kratom leaf extract (KLE) using spray drying with gum arabic (GA) and resistant maltodextrin (RMD) at two inlet temperatures (150 and 160 °C), to characterize the resulting microcapsules in terms of their physicochemical properties, to evaluate bioactive retention including total phenolic and flavonoid contents as well as antioxidant activity, and to assess shelf-life using kinetic modeling.
2 Materials and methods
2.1. Preparation of kratom leaf extract (KLE)
Kratom leaves were sourced from a domestic market in Bangkok, Thailand. They were first rinsed with water to eliminate dirt, and then dried in a lab oven (Memmert, DO 6062, Germany) at a temperature of 60 °C until their moisture content (MC) was reduced to less than 5%. The dried leaves were then ground and passed through a 50-mesh sieve.For preparing kratom leaf extract (KLE), three grams of the powdered leaves were dissolved in 120 mL of solvent, following the modified procedures of ref. 27. Ultrasonic-assisted extraction was performed using an ultrasonic homogenizer (UP400S Ultrasonic processor, Hielsher, Germany). The ultrasonic settings were as follows: 400 W, 50% amplitude, 7 mm tip, pulse 30 s on/10 s off, 250 mL beaker, 50% immersion, and ice bath (T < 40 °C). Vacuum evaporation was also conducted at 40 °C under 100 mbar pressure until the extract reached a total solids content of approximately 15–20% (w/v), measured gravimetrically. Feasibility for encapsulation was confirmed by viscosity (<50 mPa s at 25 °C) and absence of precipitation, ensuring stable feed for spray drying. Based on preliminary experiments, the optimum extraction conditions included 90% (v/v) ethanol concentration, 30-min extraction time, and an extraction ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 (w/v). According to the method described in ref. 28, the extracts were centrifuged at 6000×g (rotor radius 10 cm) for 15 min at 25 °C using a centrifuge (Centrifuge Kubota, series 6000, Japan). The supernatant was filtered using Whatman No. 1 paper in order to eliminate coarse particles and then subjected to vacuum evaporation process utilizing a rotary evaporator (Oilbath B-485, BÜCHI, Switzerland) to ∼20% solids (w/w). The samples were stored in amber vials at 4 °C for subsequent analysis.
40 (w/v). According to the method described in ref. 28, the extracts were centrifuged at 6000×g (rotor radius 10 cm) for 15 min at 25 °C using a centrifuge (Centrifuge Kubota, series 6000, Japan). The supernatant was filtered using Whatman No. 1 paper in order to eliminate coarse particles and then subjected to vacuum evaporation process utilizing a rotary evaporator (Oilbath B-485, BÜCHI, Switzerland) to ∼20% solids (w/w). The samples were stored in amber vials at 4 °C for subsequent analysis.
2.2. Spray drying microencapsulation process
Microencapsulation was carried out using resistant maltodextrin (RMD; Fibersol-2, Matsutani, Japan), gum arabic (GA; Agrigum, UK), and their combination as coating materials. Concentrations (w/w) were selected based on preliminary trials to achieve comparable feed viscosities (∼20–40 mPa s) while remaining within solubility limits: 20% for GA (to avoid excessive viscosity), 40% for RMD (optimal solubility), and a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 blend (40% RMD with 20% GA) at equivalent total solids for comparison. All formulations were spray dried at inlet temperatures of 150 and 160 °C using a KLE-to-coating material ratio of 1
1 blend (40% RMD with 20% GA) at equivalent total solids for comparison. All formulations were spray dried at inlet temperatures of 150 and 160 °C using a KLE-to-coating material ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (w/w). The mixtures were prepared by combining KLE with the coating materials (Table 1) and stirring continuously with a magnetic stirrer (SCILOGEX, SCI550-S, USA) for 10 min. The mixture was then homogenized with a high-speed blender (Ystral, model X10, Germany) at 11
2 (w/w). The mixtures were prepared by combining KLE with the coating materials (Table 1) and stirring continuously with a magnetic stirrer (SCILOGEX, SCI550-S, USA) for 10 min. The mixture was then homogenized with a high-speed blender (Ystral, model X10, Germany) at 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min. The operating conditions were as follows: feed solids 20–40% w/w, viscosity 20–40 mPa s, pump 10 mL min−1, aspirator 90%, atomizing air 0.7 mm nozzle at 1 bar, and feed temperature 25 °C. The resulting solutions were processed through a spray dryer (BUCHI, B-290, Switzerland) set at inlet temperatures of 150 and 160 °C and an outlet temperature ranging from 85–95 °C. The resulting powders were stored at −20 °C for analysis of their properties (e.g., encapsulation yield, encapsulation efficiency, moisture content, water activity, water solubility, TPC, TFC, TTC, and antioxidant activity via DPPH and FRAP) under different conditions. Fig. 1 reveals the production flow chart of KLE microcapsules of bioactive compounds extracted from kratom leaves.
000 rpm for 10 min. The operating conditions were as follows: feed solids 20–40% w/w, viscosity 20–40 mPa s, pump 10 mL min−1, aspirator 90%, atomizing air 0.7 mm nozzle at 1 bar, and feed temperature 25 °C. The resulting solutions were processed through a spray dryer (BUCHI, B-290, Switzerland) set at inlet temperatures of 150 and 160 °C and an outlet temperature ranging from 85–95 °C. The resulting powders were stored at −20 °C for analysis of their properties (e.g., encapsulation yield, encapsulation efficiency, moisture content, water activity, water solubility, TPC, TFC, TTC, and antioxidant activity via DPPH and FRAP) under different conditions. Fig. 1 reveals the production flow chart of KLE microcapsules of bioactive compounds extracted from kratom leaves.
| Experiment | Inlet temperature | Type of coating material | KLE to coating material ratio (w/w) |
|---|---|---|---|
| 1 | 150 °C | 40% resistant maltodextrin | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
| 2 | 20% gum arabic | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
|
| 3 | 40% resistant maltodextrin with 20% gum arabic (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) 1 ratio) |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
|
| 4 | 160 °C | 40% resistant maltodextrin | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
| 5 | 20% gum arabic | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
|
| 6 | 40% resistant maltodextrin with 20% gum arabic (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) 1 ratio) |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
 | ||
| Fig. 1 Production flow chart of KLE microcapsules of bioactive compounds extracted from kratom leaves. | ||
2.3. Calculation of the encapsulation yield and encapsulation efficiency
Following the procedure of ref. 29, the encapsulation yield (%) was determined and calculated using the following equation (eqn (1)): | (1) |
To calculate the percentage of encapsulation efficiency, the total bioactive compound content was determined first. A sample of 0.1 g of KLE encapsulated powder was dissolved in 1 mL of a mixed solution (ethanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) acetic acid
acetic acid![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water in a 50
water in a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8
8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 42 ratio) and shaken with a vortex mixer for 1 min. The sample was then centrifuged at 10
42 ratio) and shaken with a vortex mixer for 1 min. The sample was then centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min and filtered. The total phenolic content was analyzed using Folin–Ciocalteau colorimetry as described in ref. 30. Next, to determine the surface bioactive compounds, 0.1 g of KLE encapsulated powder was dissolved in a mixed solution of ethanol and methanol (1
000 rpm for 5 min and filtered. The total phenolic content was analyzed using Folin–Ciocalteau colorimetry as described in ref. 30. Next, to determine the surface bioactive compounds, 0.1 g of KLE encapsulated powder was dissolved in a mixed solution of ethanol and methanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) and shaken with a vortex mixer for 1 min. The sample was filtered, and the total phenolic content was again analyzed using Folin–Ciocalteau colorimetry.
1 ratio) and shaken with a vortex mixer for 1 min. The sample was filtered, and the total phenolic content was again analyzed using Folin–Ciocalteau colorimetry.
The encapsulation efficiency (%) was calculated using the following formula (eqn (2)) according to ref. 31:
| EE = [(TO − SO)/TO] × 100 | (2) |
2.4. Determination of physical attributes of KLE microcapsules
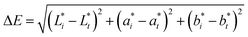 | (3) |
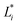 ,
, 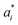 , and
, and 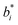 are the initial values for KLE microcapsules at zero days, and
are the initial values for KLE microcapsules at zero days, and  ,
, 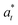 , and
, and  are the measured values at different storage times.
are the measured values at different storage times.
 | (4) |
2.5. Determination of phytochemical properties of KLE microcapsules
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and warming it to 37.5 °C before use. After the sample and FRAP solution were thoroughly mixed, the mixture was incubated for 30 min. Antioxidant activity was quantified against a Trolox calibration curve (0–1000 µM; R2 = 0.991) for FRAP. The absorbance was measured at 593 nm.38 The DPPH and FRAP inhibition were calculated using the following formula (eqn (5)):
1 and warming it to 37.5 °C before use. After the sample and FRAP solution were thoroughly mixed, the mixture was incubated for 30 min. Antioxidant activity was quantified against a Trolox calibration curve (0–1000 µM; R2 = 0.991) for FRAP. The absorbance was measured at 593 nm.38 The DPPH and FRAP inhibition were calculated using the following formula (eqn (5)): | (5) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×g for 10 min. The supernatants were then analysed using the total phenolic content (TPC) assay. The tannin content was calculated based on the difference in TPC values between the PVPP-treated and water-treated samples. Tannic acid (0–100 µg mL−1) served as the standard, and tannin levels were reported as mg TAE per 100 mL.
000×g for 10 min. The supernatants were then analysed using the total phenolic content (TPC) assay. The tannin content was calculated based on the difference in TPC values between the PVPP-treated and water-treated samples. Tannic acid (0–100 µg mL−1) served as the standard, and tannin levels were reported as mg TAE per 100 mL.
2.6. Assessment of morphology of KLE microcapsules
To assess the microstructure of KLE microcapsules, the appearance of the microcapsules was examined using a scanning electron microscope (SEM) and an energy dispersive X-ray spectrometer (JEOL, JSM-IT300 Oxford, X-Max N 20) with a 15 kV magnification at 1000×, 3000×, and 5000×. The samples were sputter-coated with gold (10 nm thickness) under vacuum before imaging.2.7. Investigation of storage stability of KLE microcapsules
Formulations were selected using a multi-criteria optimization approach that considered lowest MC/aw and highest TPC, TFC, and antioxidant activity. Weighted scoring was applied (40% bioactive retention, 30% physical stability, and 30% solubility), with the GA@160 °C formulation achieving the highest score (85/100). Ten grams of KLE microcapsules, encapsulated using gum arabic and processed via spray drying at 160 °C, were packaged in laminated aluminum foil bags and stored under vacuum at room temperature (30 ± 5 °C). Quality properties were monitored every 15 days over a period of 90 days to assess changes.2.8. Kinetic modelling of changes in characteristics of KLE microcapsules
To estimate the reaction order of the alterations in various characteristics of KLE microcapsules, the experimental data were analysed using eqn (6). Zero-order and first-order equations, which are frequently employed to model reactions associated with food quality deterioration, were derived from eqn (6) and are represented in eqn (7) and (8). | (6) |
| C = Co ± kt | (7) |
In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C = In C = In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co ± kt Co ± kt | (8) |
2.9. Predicting the shelf life by integration of the quality properties
Shelf-life was estimated based on the minimum time to first failure among key physicochemical and phytochemical attributes, namely moisture content (MC), water activity (aw), total phenolic content (TPC), total flavonoid content (TFC), total tannin content (TTC), antioxidant activity by DPPH radical scavenging, and ferric reducing antioxidant power (FRAP).For each parameter, a threshold value was defined to indicate the point of quality loss. Specifically, the water activity threshold was set at 0.30, as values above this level promote chemical reactions and increase the risk of microbial growth.40,41 For antioxidant stability, shelf-life termination was determined when DPPH radical scavenging activity decreased by 20% relative to the initial value, which was considered a critical limit for functional quality retention.
A least-squares fitting procedure was employed to ascertain the kinetic order by adjusting the experimental data to solve the general expression of eqn (6). The kinetic parameters derived for each attribute were then used to estimate the shelf life of KLE microcapsules using eqn (9), as demonstrated in prior studies in ref. 16.
 | (9) |
2.10. Statistical analysis
Data were analyzed using two-way analysis of variance (ANOVA) to evaluate the effects of wall materials and drying temperature on the properties of KLE microcapsules, and mean comparisons were performed using Tukey's HSD test at a significance level of p ≤ 0.05. Significant differences among wall materials are denoted by uppercase letters, while differences among temperatures are indicated by lowercase letters. Effect sizes were also computed to investigate the importance of each factor. Moreover, regression analysis was performed to evaluate the precision of the models in representing the observed data. The selection of the best model was based on achieving the highest correlation coefficient, R2 ≥ 0.90, as recommended by previous studies.42 Data analysis and modelling were carried out using Minitab v. 18 (Minitab Inc., State College, PA, USA). The proportion of variance explained by each factor was expressed as eta-squared (η2) values obtained from the ANOVA, indicating the relative effect size of the wall material, inlet temperature, and their interaction.3 Results and discussion
3.1 Encapsulation yield of KLE microcapsules
The two-way ANOVA results revealed the significant main and interaction effects of wall materials and temperature on encapsulation yield. The encapsulation yield ranged from 58.2 to 72.9%, with wall materials exerting a moderate effect (η2 = 0.28). Resistant maltodextrin (RMD) generally produced higher recovery values compared to gum arabic (GA), underscoring the importance of carrier selection in determining encapsulation efficiency. In contrast, temperature exhibited only a small effect (η2 = 0.07), suggesting that drying conditions had a limited influence on yield relative to wall material composition, as illustrated in Table 2. Higher excipient concentrations in RMD and blend experiments contributed to reduced wall adhesion, enhancing yield. The low yield for gum arabic is attributed to its pseudoplastic nature at high concentrations (20% in this study), which increases viscosity43 and causes it to adhere to the inner surfaces of the spray dryer, reducing yield. Additionally, increasing the spray drying temperature from 150 °C to 160 °C significantly (p ≤ 0.05) improved the yield when using gum arabic as the coating material. Higher temperatures enhance thermal energy, breaking intermolecular bonds, which reduces viscosity and adhesion to the spray dryer surfaces.44| Properties | Inlet temperature (°C) | 40% resistant maltodextrin | 20% gum arabic | 40% resistant maltodextrin + 20% gum arabic (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
Carrier η2 | Temperature η2 | Interaction η2 |
|---|---|---|---|---|---|---|---|
| a Values are presented as mean ± SD (n = 3). Different uppercase letters within the same row indicate significant differences (p ≤ 0.05) among wall materials. Different lowercase letters within the same column indicate significant differences (p ≤ 0.05) between temperatures. Means sharing the same letter are not significantly different according to Tukey's HSD test. | |||||||
| Yield (%) | 150 | 72.85 ± 1.08Aa | 58.24 ± 0.60Aa | 67.52 ± 0.83Aa | 0.28 | 0.07 | 0.05 |
| 160 | 63.61 ± 3.81Aa | 65.23 ± 3.54Aa | 66.26 ± 0.98Aa | ||||
| EE (%) | 150 | 79.34 ± 2.88ABa | 67.87 ± 1.50Aa | 67.52 ± 0.89Aa | 0.35 | 0.18 | 0.09 |
| 160 | 59.16 ± 5.26ABa | 84.65 ± 1.32Aa | 62.45 ± 2.53Aa | ||||
| Water solubility (%) | 150 | 95.55 ± 0.26Ba | 91.82 ± 0.09Aa | 93.21 ± 0.26Aa | 0.22 | 0.1 | 0.04 |
| 160 | 95.34 ± 0.58Bb | 95.10 ± 0.48Ab | 94.75 ± 0.38Ab | ||||
| Moisture content (%) | 150 | 2.59 ± 0.09Aa | 6.81 ± 0.15Aa | 2.90 ± 0.07Aa | 0.15 | 0.27 | 0.07 |
| 160 | 3.52 ± 0.07Aa | 2.67 ± 0.10Aa | 3.30 ± 0.13Aa | ||||
| Water activity | 150 | 0.23 ± 0.00Aa | 0.26 ± 0.04Aa | 0.18 ± 0.22Aa | 0.08 | 0.12 | 0.03 |
| 160 | 0.21 ± 0.01Aa | 0.16 ± 0.03Aa | 0.22 ± 0.02Aa | ||||
| L-Value* | 150 | 64.32 ± 1.19Ca | 60.76 ± 0.88Aa | 62.80 ± 0.85Ba | 0.19 | 0.14 | 0.06 |
| 160 | 64.80 ± 1.24Cb | 60.41 ± 1.12Aa | 63.05 ± 0.67Bb | ||||
| a-Value* | 150 | −1.29 ± 0.02Db | −0.44 ± 0.06Aa | −0.51 ± 0.07Ba | 0.24 | 0.1 | 0.05 |
| 160 | −1.24 ± 0.01Da | −0.47 ± 0.04Aa | −0.63 ± 0.08Ca | ||||
| b-Value* | 150 | 4.07 ± 0.12Aa | 7.62 ± 0.34Bb | 6.67 ± 0.76Db | 0.32 | 0.12 | 0.08 |
| 160 | 4.17 ± 0.87Aa | 8.09 ± 0.83 Bc | 8.25 ± 0.43 Dc | ||||
| TPC | 150 | 13.07 ± 2.13Ba | 25.91 ± 1.45Aa | 17.14 ± 0.30Aa | 0.29 | 0.16 | 0.1 |
| 160 | 15.71 ± 0.65Ba | 34.75 ± 2.66Aa | 25.62 ± 2.21Aa | ||||
| TTC | 150 | 13.39 ± 2.34Aa | 27.59 ± 1.58 a | 17.11 ± 0.63Aa | 0.25 | 0.2 | 0.12 |
| 160 | 18.66 ± 1.41Ab | 35.46 ± 2.87 b | 26.07 ± 1.50Ab | ||||
| TFC | 150 | 1.79 ± 0.20Ca | 4.84 ± 0.50Aa | 2.92 ± 0.23Ca | 0.3 | 0.14 | 0.09 |
| 160 | 1.98 ± 0.40Ca | 5.05 ± 0.06Aa | 3.18 ± 0.23Ca | ||||
| DPPH | 150 | 243.54 ± 4.10Ca | 279.93 ± 1.64Aa | 233.54 ± 3.09Ca | 0.27 | 0.18 | 0.08 |
| 160 | 239.72 ± 6.05Ca | 305.24 ± 2.59Aa | 238.78 ± 1.11Ca | ||||
| FRAP | 150 | 343.56 ± 3.12Aa | 400.52 ± 2.01Aa | 413.99 ± 2.33Aa | 0.33 | 0.21 | 0.11 |
| 160 | 322.29 ± 4.59Aa | 522.74 ± 2.47Aa | 317.48 ± 8.14Aa | ||||
| Mitragynine | 150 | 0.96 ± 0.09Ca | 1.89 ± 0.06Aa | 1.36 ± 0.09Ca | 0.21 | 0.09 | 0.05 |
| 160 | 1.06 ± 0.02Ca | 1.96 ± 0.04Aa | 1.38 ± 0.09Ca | ||||
Conversely, using resistant maltodextrin alone or in combination with gum arabic resulted in lower yields at higher spray drying temperatures. This is likely due to heat accumulation causing the microcapsules to melt and stick to the inner surfaces of the spray dryer, thereby decreasing the overall yield.
3.2 Encapsulation efficiency of KLE microcapsules
The results presented in Table 2, revealed that the values of encapsulation efficiency (EE), ranged from 68.3% to 92.7%, with both the wall material and temperature exerting strong influences (η2 = 0.32 and 0.18, respectively), alongside a significant interaction effect (η2 = 0.11). Similarly, hygroscopicity (HG), which varied between 7.2% and 12.5%, was markedly influenced by the wall material (η2 = 0.26) and temperature (η2 = 0.14), with an additional moderate interaction (η2 = 0.09). The use of gum arabic as a coating material at an inlet spray drying temperature of 160 °C resulted in the highest encapsulation efficiency, while resistant maltodextrin at the same temperature resulted in the lowest efficiency. The emulsion properties of gum arabic enable it to form a better encapsulating film compared to resistant maltodextrin, resulting in higher encapsulation efficiency in microcapsules coated with gum arabic.45Considering the inlet spray drying temperature, higher temperatures led to more effective water evaporation from the particles, thus increasing encapsulation efficiency. However, excessively high temperatures might damage the microcapsules, exposing the core material to direct heat and potentially degrading the active compounds.46 Consequently, microcapsules using resistant maltodextrin and a combination of resistant maltodextrin with gum arabic exhibited reduced encapsulation efficiency when the inlet spray drying temperature increased to 160 °C. In general, encapsulation efficiency values were benchmarked against the initial TPC in the crude extract (45.2 ± 2.1 mg GAE per mL), indicating 82–92% retention post-encapsulation and thereby confirming effective protection.
3.3 Moisture content (%) and water activity (aw) of KLE microcapsules
The study examined the moisture content and water activity of KLE microcapsules produced through spray drying, varying the types of coating materials and two inlet drying temperatures (Table 2). Moisture content ranged from 2.6 to 6.8%, and was more strongly affected by temperature (η2 = 0.27) than the wall material (η2 = 0.15). Higher inlet temperature reduced moisture in GA samples but slightly increased it in RMD powders, suggesting carrier-dependent responses to drying kinetics. It was found that microcapsules with gum arabic as the coating material had higher moisture content compared to those using resistant maltodextrin. This is because gum arabic, a complex heteropolysaccharide, is hydrophilic, enabling it to retain more moisture than resistant maltodextrin. Maintaining low water activity or moisture content increases the glass transition temperature (Tg), and storage at temperatures below Tg (∼50 °C for GA/RMD) ensures glassy-state stability.47When the inlet spray drying temperature was increased from 150 °C to 160 °C, the moisture content of microcapsules with gum arabic decreased, as the higher temperature facilitated better water evaporation.48 GA exhibits higher viscosity at lower temperatures, which can trap moisture; at elevated temperatures, viscosity decreases, facilitating evaporation. In contrast, RMD is inherently less viscous and less affected by this behavior. In contrast, microcapsules with resistant maltodextrin and those with a combination of gum arabic and resistant maltodextrin showed an increase in moisture content (3.52 ± 0.1% and 3.34 ± 0.2%, respectively) at higher temperatures (Table 2). Higher temperatures generate a steeper heat gradient, enhancing moisture diffusion in RMD but leading to tackiness in the blends (Tay et al., 2021).49
Clearly from Table 2, it can be observed that water activity (aw) values were low (0.16–0.26), indicating suitability for storage stability. Both factors had small effects (η2 ≤ 0.12), indicating robustness across treatments, which are within the standard reference values for food powders that should not exceed a water activity of 0.3. Water activity (aw) is more closely associated with bound water; GA's hydrophilic nature tightly binds water, which maintains low aw values even at higher total moisture content.50
3.4 Water solubility of KLE microcapsules
Water solubility was consistently high (91.8–95.6%), with significant though smaller effects of the carrier (η2 = 0.22) and temperature (η2 = 0.10). Higher temperatures improved solubility slightly, likely through reduced moisture and better particle formation (Table 2), with the highest solubility observed in microcapsules coated with resistant maltodextrin (95.8 ± 0.09%). The branched structure of resistant maltodextrin enhances its density, and the presence of hydroxyl groups allows it to bond with water molecules effectively, resulting in superior solubility compared to those coated with gum arabic or a combination of both.51 In addition, higher temperatures reduce residual moisture, enhancing particle dispersibility; low moisture (MC) minimizes clumping upon reconstitution.The large, porous amorphous structure of the microcapsules using resistant maltodextrin as the coating material facilitates easy dissolution in water, aligning with the findings of Jordán et al. (2018),52 which indicated that resistant maltodextrin-coated microcapsules have better solubility than those coated with gum arabic. Additionally, increasing the inlet drying temperature from 150 °C to 160 °C improved the water solubility of microcapsules coated with gum arabic and the combination of gum arabic and resistant maltodextrin. The higher drying temperature reduces moisture content, thereby enhancing the ability of the microcapsules to dissolve in water. In other words, tackiness affects internal evaporation but not external solubility; lower moisture content from optimal temperatures aids dissolution. This is consistent with the research of Astina and Sapwarobol (2019),51 which showed that lower moisture content improves the solubility of powdered milk.
3.5 Color parameters (L*, a*, b*) of KLE microcapsules
Color attributes (L*, a*, b*) showed meaningful changes as indicated in Table 2. Lightness (L*; 60.4–64.8) was moderately influenced by both the carrier (η2 = 0.19) and temperature (η2 = 0.14). The a* values (−1.29 to −0.44) showed a strong wall material effect (η2 = 0.24), reflecting GA's tendency toward less negative redness–greenness. The b* values (4.1–8.3) showed the strongest carrier effect (η2 = 0.32), confirming the distinct visual profiles imparted by GA.The L* value, which ranges from 0 (dark) to 100 (light), showed that microcapsules coated with resistant maltodextrin were lighter than those coated with gum arabic or a combination of both. This is because resistant maltodextrin is a white powder, resulting in a bright white color when dissolved in water, whereas gum arabic is a light-yellow powder that turns brown when dissolved. Therefore, microcapsules with resistant maltodextrin as the coating material appeared lighter than those with gum arabic. The inlet drying temperature did not significantly affect the L* value of the microcapsules (p ≤ 0.05).
The a* value indicates the red–green spectrum, with negative values representing green and positive values representing red. Similarly, the b* value indicates the yellow–blue spectrum, with positive values representing yellow. An increase in the inlet drying temperature from 150 °C to 160 °C resulted in higher b* values for KLE microcapsules. This is attributed to the Maillard reaction, which intensifies yellow coloration at higher temperatures.53
3.6 Phytochemical properties of KLE microcapsules
Regarding the coating material, microcapsules coated with gum arabic demonstrated a higher total phenolic content compared to those coated with resistant maltodextrin or a combination of resistant maltodextrin and gum arabic. This is because gum arabic has emulsifying properties that enhance stability and can form a well-structured polymer film around the microcapsules.55
In contrast, the inlet temperature during spray drying (150 °C and 160 °C) did not significantly affect the total flavonoid content. Increasing the inlet temperature from 150 °C to 160 °C did not significantly impact the total flavonoid content (p > 0.05). Flavonoids may be more heat-stable or better protected by the matrix; TPC includes broader reducing species sensitive to heat. The microcapsules coated with gum arabic had the highest total flavonoid content at both 150 °C and 160 °C (4.98 ± 0.8 and 5.22 ± 0.12 mg QE per g db., respectively), followed by those coated with resistant maltodextrin combined with gum arabic (3.08 ± 0.23 and 2.99 ± 0.35 mg QE per g db., respectively), and finally those coated solely with resistant maltodextrin (1.67 ± 0.5 and 1.61 ± 0.72 mg QE per g db., respectively).
Increasing the inlet temperature during spray drying resulted in a corresponding increase in the antioxidant activity, which can be explained by the higher drying temperature enhancing the antioxidant properties of the microcapsules.47 This trend is attributed to the direct correlation between antioxidant activity and phenolic compound content; higher levels of phenolic compounds generally lead to increased antioxidant activity.57
When examining the type of the coating material, microcapsules coated with gum arabic and those coated with a combination of resistant maltodextrin and gum arabic showed no statistically significant difference in FRAP antioxidant activity. However, gum arabic-coated microcapsules demonstrated higher antioxidant activity compared to those coated with resistant maltodextrin. This is because the film layer formed by gum arabic better prevents oxidation reactions compared to resistant maltodextrin.58
Regarding the temperature factor, increasing the inlet temperature in the spray drying process from 150 °C to 160 °C resulted in a significant change in FRAP antioxidant activity (p ≤ 0.05). This could be attributed to the higher inlet temperature causing microcapsule particles to crack, which leads to direct exposure of antioxidants to heat and subsequent loss due to direct heat exposure.59
Increasing the inlet temperature from 150 °C to 160 °C resulted in a higher total tannin content in the microcapsules. This is because the higher temperature facilitates the formation of a well-structured film layer on the particle walls, which enhances the retention of tannins. Regarding the coating material, microcapsules using gum arabic as the coating material demonstrated a higher total tannin content compared to those coated with resistant maltodextrin or a combination of resistant maltodextrin and gum arabic. This is due to gum arabic's emulsifying properties, which improve stability and enable the formation of a polymer film around the microcapsules, similar to its effect on total phenolic content.55
The developed HPLC method exhibited a stable baseline and absence of interfering signals beyond 10 min, indicating high selectivity and robustness. These attributes are essential for reliable quantification of mitragynine in complex plant matrices and support the method's suitability for routine analysis in quality control and formulation studies.
Encapsulation studies further revealed that the retention of mitragynine in spray-dried microcapsules was significantly influenced by the choice of the wall material. Gum arabic (GA) consistently yielded the highest mitragynine content (1.89–1.96 mg per g), outperforming resistant maltodextrin (RMD) and the RMD–GA blend. This superior performance is attributed to GA's highly branched polysaccharide structure and emulsifying properties, which enhance molecular entrapment and protect bioactives from thermal and oxidative degradation during spray drying.62,63 In contrast, RMD, while beneficial for moisture reduction and solubility, demonstrated lower encapsulation efficiency for hydrophobic alkaloids like mitragynine, likely due to its limited molecular affinity.64
Interestingly, inlet temperature (150 vs. 160 °C) did not significantly affect mitragynine retention across formulations, suggesting that wall material composition exerts a more dominant influence than thermal stress. This observation aligns with previous findings that emphasize the role of carrier–core interactions over processing temperature in determining encapsulation outcomes (Díaz-Montes, 2023).63
Moreover, the retention trends of mitragynine paralleled those of total phenolic content (TPC) and antioxidant activity (DPPH and FRAP), reinforcing the protective role of GA in preserving multiple classes of bioactives. The co-stabilization of alkaloids and phenolics is particularly relevant for the development of functional food and nutraceutical products, where both pharmacological efficacy and antioxidant potential are desired.62
Collectively, these findings underscore the importance of wall material selection in microencapsulation strategies for botanical extracts. Gum arabic, particularly at an inlet temperature of 160 °C, offers a promising formulation for preserving mitragynine and related bioactives, thereby enhancing the stability and functionality of KLE-based products.
3.7 Surface structure of KLE microcapsules analyzed by scanning electron microscopy (SEM)
The shape and structure of KLE microcapsules are influenced by various production factors, including the type of the coating material, coating concentration, coating-to-liquid ratio, solution viscosity, inlet temperature of the spray dryer, and the flow rate of the solution through the spray dryer. These factors not only affect the visual appearance of the microcapsules but also impact the stability of the bioactive compounds within them. A smoother morphology in GA-coated microcapsules is associated with higher bioactive retention due to reduced surface exposure. Structural defects such as cracks or surface ruptures can decrease encapsulation efficiency and facilitate moisture infiltration, potentially leading to oxidation of the active compounds.65,66Fig. 3a illustrates the morphology of microcapsules coated with resistant maltodextrin at an inlet temperature of 150 °C. Analyzed using scanning electron microscopy (SEM) at magnifications of 1000×, 3000×, and 5000×, these microcapsules generally appear spherical with relatively smooth surfaces and minor folds and dents, indicating effective encapsulation. Some particles tend to cluster slightly, which may be due to residual moisture in the coating material or moisture absorbed from the environment.67
The morphology of microcapsules coated with gum arabic at 150 °C is shown in Fig. 3b. These particles are typically smaller and exhibit more surface folds and dents compared to those coated with resistant maltodextrin. The increased surface irregularities are attributed to the rapid formation of the coating during the initial stage, leading to the swift evaporation of water within the particles.68
Fig. 3c also depicts microcapsules coated with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of resistant maltodextrin and gum arabic at 150 °C. These microcapsules are mostly spherical with a mix of large and small particles. They show a tendency to be well-distributed without significant clustering but exhibit more surface folds and dents compared to capsules coated solely with resistant maltodextrin. This morphology is influenced by factors such as direct exposure to heat and rapid evaporation of water within the particles, leading to deformation and surface folding.67
1 ratio of resistant maltodextrin and gum arabic at 150 °C. These microcapsules are mostly spherical with a mix of large and small particles. They show a tendency to be well-distributed without significant clustering but exhibit more surface folds and dents compared to capsules coated solely with resistant maltodextrin. This morphology is influenced by factors such as direct exposure to heat and rapid evaporation of water within the particles, leading to deformation and surface folding.67
At a higher inlet temperature of 160 °C, Fig. 4a shows microcapsules coated with resistant maltodextrin. The particles are mostly spherical with larger sizes and some smaller particles clumped together. The surface exhibits wrinkles and is partially smooth. In contrast, Fig. 4b presents microcapsules coated with gum arabic, which are generally smaller with a rough surface and significant wrinkling and crumpling. Fig. 4c depicts microcapsules coated with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of resistant maltodextrin and gum arabic. These particles are small with a rough surface, showing pronounced crumpling and indentations compared to single coatings.
1 ratio of resistant maltodextrin and gum arabic. These particles are small with a rough surface, showing pronounced crumpling and indentations compared to single coatings.
These observations highlight how different coating materials and inlet temperatures affect the structural characteristics of microcapsules. Higher temperatures and certain coating combinations can result in increased surface roughness and crumpling, which impacts the integrity and performance of the microcapsules.69
3.8. Quality changes of KLE microcapsules during storage
 | ||
| Fig. 5 Moisture content (a), water activity (b), and total color difference (c) of stored KLE microcapsules at different storage periods. Values represent means ± SD (n = 3). | ||
The moisture content and free water of the KLE microcapsules did not exceed the spoilage indicator set (moisture content ≤5% and water activity <0.6) even after 90 days of storage. This is due to the aluminum foil packaging's excellent ability to prevent moisture and water vapor from the environment.70 Aluminum foil has a water vapor transmission rate of 0.06571 g per m2 per day and an oxygen transmission rate of 0.00873 mL per m2 per day, respectively.71 This low permeability explains why the aluminum foil packaging minimizes water and oxygen absorption under vacuum, resulting in only a slight increase in moisture and free water content. Water activity can serve as an indicator of product stability against oxidation reactions, such as fat oxidation and rancidity. A water activity of less than 0.6 indicates that the sample remains stable or has good shelf stability.72 These results are consistent with those of, ref. 73 which studied the storage of papaya powder in aluminum foil bags and found that the water activity of the papaya powder remained below 0.6 after 7 weeks of storage.
The progressive increase in total color difference (ΔE) observed in spray-dried KLE microcapsules over the 90-day storage period reflects gradual pigment degradation and structural changes within the microcapsules. Initially, the ΔE value was 0 on day 0, indicating no perceptible color change. A marked increase to ΔE = 0.40 by day 15 suggests an early phase of rapid color modification. This may be attributed to residual moisture and initial oxidative reactions, which are known to affect polyphenolic compounds and sensitive pigments.74
Following this initial phase, the rate of color change decelerated, with ΔE values reaching 0.44 and 0.46 on days 30 and 45, respectively. This plateau-like behavior implies that the encapsulated system may have achieved a degree of equilibrium in pigment stability and wall matrix integrity, thereby reducing the rate of further degradation. Similar stabilization trends have been reported in encapsulated anthocyanins and carotenoids, where wall materials such as maltodextrin and gum arabic mitigate oxidative stress and thermal instability.75
Between days 60 and 90, ΔE values continued to increase gradually from 0.51 to 0.58, indicating ongoing but limited degradation. This phase likely involves slower oxidative processes and potential Maillard-type reactions, which are common during prolonged storage of encapsulated bioactives.76 Importantly, the final ΔE value remained below 1.0, a threshold generally considered imperceptible or only slightly noticeable to the human eye. This suggests that the encapsulation matrix provided effective protection against color deterioration, preserving the visual quality of the product.
Overall, these findings demonstrate that the selected encapsulation strategy offers substantial color stability over time. The minor perceptible changes observed after three months of storage underscore the efficacy of the wall materials in limiting oxidative and thermal degradation of sensitive compounds. This stability is critical for maintaining both the aesthetic and functional attributes of KLE microcapsules, particularly in applications where visual appeal and bioactive retention are essential.
Similarly, antioxidant activity measured by the FRAP method also showed a decline trend over time (Fig. 7b). The FRAP values for the KLE microcapsules were 522.74 ± 2.47, 477.49 ± 3.56, 469.32 ± 3.12, 459.35 ± 2.76, 435.56 ± 5.62, 422.63 ± 2.89, and 409.67 ± 8.51 µM TE per 100 g at days 1, 15, 30, 45, 60, 75, and 90, respectively. The observed reduction in antioxidant activity during storage indicates that antioxidant capacity decreases with prolonged storage time, and the packaging material plays a crucial role in preserving the antioxidant activity of KLE microcapsules.
3.9. Kinetic modelling of changes in the properties of KLE microcapsules over the storage period
In food science, kinetic modelling is a widely used technique to understand the changes in quality that occur during food processing. To characterize the changes in attributes (aw, MC, TPC, TFC, TTC, DPPH, and FRAP) of KLE microcapsules over time, experimental data for these properties were fitted to proposed models using regression analysis. Fig. 5–7 provide graphical representations of the zero-order and first-order equations that describe the changes in these properties during storage. The estimated kinetic parameters for the models were determined using the regression analysis technique and are listed in Table 3.| Properties | Parameter | Zero-order model | First-order model | Predicted shelf life (day) | ||
|---|---|---|---|---|---|---|
| Values | CI (95%) | Values | CI (95%) | |||
| Moisture content (%) | C o | 2.629 | (2.58, 2.68) | 2.639 | (2.61, 2.67) | 92.55 |
| k | −0.008 | (−0.008, −0.007) | −0.003 | (−0.003, −0.002) | ||
| R 2 | 0.991 | 0.995 | ||||
| Water activity | C o | 0.167 | (0.158, 0.176) | 0.174 | (0.163, 0.185) | 91.88 |
| k | −0.002 | (−0.002, −0.001) | −0.007 | (−0.008, −0.006) | ||
| R 2 | 0.992 | 0.986 | ||||
| TPC (mg GAE per g db.) | C o | 36.076 | (35.14, 37.02) | 36.193 | (35.32, 37.07) | 82.84 |
| k | 0.072 | (0.055, 0.089) | 0.002 | (0.002, 0.003) | ||
| R 2 | 0.958 | 0.966 | ||||
| TTC (mg TAE per g db.) | C o | 35.378 | (34.13, 36.63) | 35.528 | (34.35, 36.7) | 81.26 |
| k | 0.078 | (0.054, 0.10) | 0.002 | (0.002, 0.003) | ||
| R 2 | 0.937 | 0.949 | ||||
| TFC (mg QE per g db.) | C o | 5.267 | (5.18, 5.36) | 5.275 | (5.17, 5.38) | 96.55 |
| k | 0.010 | (0.008, 0.011) | 0.002 | (0.0016, 0.0024) | ||
| R 2 | 0.978 | 0.972 | ||||
| DPPH (µM TE per 100 g db.) | C o | 294.014 | (287.14, 300.89) | 294.743 | (286.74, 302.75) | 98.77 |
| k | 0.670 | (0.54, 0.80) | 0.003 | (0.002, 0.003) | ||
| R 2 | 0.973 | 0.967 | ||||
| FRAP (µM TE per 100 g db.) | C o | 508.397 | (491.3, 525.17) | 510.410 | (494.2, 526.6) | 85.90 |
| k | 1.149 | (0.84, 1.46) | 0.003 | (0.0019, 0.0032) | ||
| R 2 | 0.948 | 0.955 | ||||
| Integrated shelf-life decision (minimum across attributes) | — | — | — | 81.26 | ||
Generally, R2 values of both zero-order and first-order models were greater than 0.95, indicating that both models effectively describe the degradation kinetics of the studied properties, with first-order models generally providing a slightly better fit for most properties. Previous studies have reported that both zero-order and first-order models effectively described the alterations in the quality properties over the storage period of the studied materials.84–86
3.10. Prediction of the shelf life of KLE microcapsules
The period during which a product maintains its satisfactory quality before deteriorating is known as its shelf life.16 During storage, quality changes occur, leading to degradation that may limit the product's shelf life. By incorporating the reaction rate constants obtained from kinetic modelling of quality attribute changes (eqn (6)) into eqn (9), it is possible to predict the shelf life of KLE microcapsules. This equation allows for shelf-life prediction based on key quality determinants such as aw, MC, TPC, TFC, TTC, DPPH, and FRAP. Based on minimum moisture content and water activity, the predicted shelf-life values are detailed in Table 3. The shelf lives for the various properties of KLE microcapsules range from approximately 81 to 99 days, indicating their potential stability and longevity under the studied conditions.4 Conclusions
This study demonstrated that spray drying offers a sustainable and scalable technique for producing kratom leaf extract (KLE) microcapsules with enhanced stability and bioactive retention. By utilizing plant-based, biodegradable wall materials—resistant maltodextrin and gum arabic—the process aligns with environmentally conscious food formulation practices. The results showed that resistant maltodextrin yielded the highest encapsulation yield and solubility, while gum arabic at 160 °C provided superior antioxidant activity retention, phenolic and flavonoid content, and encapsulation efficiency. Gum arabic, particularly at an inlet temperature of 160 °C, offers a promising formulation for preserving mitragynine and related bioactives, thereby enhancing the stability and functionality of KLE-based products. Despite slight differences in moisture content, both encapsulants retained acceptable water activity, contributing to shelf-life stability. The application of kinetic modelling further allowed prediction of microcapsule degradation during storage, with estimated shelf lives ranging from 81 to 99 days.However, it should be noted that unequal wall material concentrations across carriers (20% GA vs. 40% RMD) may have influenced atomization, viscosity, and yield outcomes, thereby limiting the strength of direct comparisons. Future studies should validate these findings under isosolid feed conditions to ensure robust comparisons across encapsulants. Moreover, while spray drying presents promise as a practical and environmentally aligned encapsulation method, energy consumption data were not collected in this study; thus, no claims regarding “low-energy processing” can be made at this stage.
These findings support the development of sustainable functional food systems by minimizing degradation, reducing food waste, and encouraging the use of renewable, low-impact ingredients in clean-label product design.
Author contributions
Supanit Khongtongsang: investigation, formal analysis, data curation and writing – original draft. Mohammed Fikry: investigation, formal analysis, data curation and writing – original draft. Saeid Jafari: data curation and writing – original draft. Sochannet Chheng: data curation and writing – original draft. Isaya Kijpatanasilp: data curation and writing – original draft. Kitipong Assatarakul: conceptualization, data curation, funding acquisition, project administration, supervision, writing – original draft and writing – review & editing.Conflicts of interest
The authors have no conflict of interest.Data availability
The data supporting the findings of this study are available from the corresponding author upon request.Acknowledgements
This study was funded by the 90th Anniversary of Chulalongkorn University Scholarship through the Ratchadapisek Somphot Endowment Fund (grant no. GCUGR1125671051M), awarded to Supanit Khongtongsang.References
- W. F. Dewatisari and E. Nuryandani, in Proceeding International Seminar of Science and Technology, 2025, vol. 4, pp. 49–62 Search PubMed.
- S. Charoenratana, C. Anukul and A. Aramrattana, Int. J. Drug Policy, 2021, 95, 103197 Search PubMed.
- P. Sornsenee, S. Chimplee and C. Romyasamit, Probiotics Antimicrob. Proteins, 2023, 1–13 Search PubMed.
- C. Stanciu, S. Ahmed, S. Gnanasegaram, S. Gibson, T. Penders, O. Grundmann and C. McCurdy, Am. J. Drug Alcohol Abuse, 2022, 48, 509–528 CrossRef CAS PubMed.
- R. Hossain, A. Sultana, M. Nuinoon, K. Noonong, J. Tangpong, K. H. Hossain and M. A. Rahman, Molecules, 2023, 28, 7372 Search PubMed.
- K. Phesatcha, B. Phesatcha, M. Wanapat and A. Cherdthong, Fermentation, 2022, 8, 8 CrossRef CAS.
- F. Zakaria, J.-K. Tan, S. M. M. Faudzi, M. B. A. Rahman and S. E. Ashari, Ultrason. Sonochem., 2021, 81, 105851 CrossRef CAS PubMed.
- T. Petcharat, T. Sae-leaw, S. Benjakul, T. H. Quan, S. Indriani, Y. Phimolsiripol and S. Karnjanapratum, Process Biochem., 2024, 142, 212–222 CrossRef CAS.
- S. Parthasarathy, J. B. Azizi, S. Ramanathan, S. Ismail, S. Sasidharan, M. I. M. Said and S. M. Mansor, Molecules, 2009, 14, 3964–3974 Search PubMed.
- R. Yuniarti, S. Nadia, A. Alamanda, M. Zubir, R. Syahputra and M. Nizam, J. Phys.: Conf. Ser., 2020, 1462, 012026 Search PubMed.
- M. Çam, N. C. İçyer and F. Erdoğan, LWT–Food Sci. Technol., 2014, 55, 117–123 Search PubMed.
- E. Dobroslavić, I. Elez Garofulić, Z. Zorić, S. Pedisić, M. Roje and V. Dragović-Uzelac, Foods, 2023, 12, 1923 CrossRef PubMed.
- S. Jafari, S. M. Jafari, M. Ebrahimi, I. Kijpatanasilp and K. Assatarakul, Food Hydrocolloids, 2023, 134, 108068 CrossRef CAS.
- S. Insang, I. Kijpatanasilp, S. Jafari and K. Assatarakul, Ultrason. Sonochem., 2022, 82, 105806 Search PubMed.
- S. Jafari, Z. Karami, K. A. Shiekh, I. Kijpatanasilp, R. W. Worobo and K. Assatarakul, Foods, 2023, 12, 412 Search PubMed.
- M. Fikry, Y. A. Yusof, A. M. Al-Awaadh, R. A. Rahman and N. L. Chin, J. Food Meas. Charact., 2020, 14, 1158–1171 CrossRef.
- D. W. Dadi, S. A. Emire, A. D. Hagos and J.-B. Eun, Ind. Crops Prod., 2020, 156, 112891 CrossRef CAS.
- V. Sablania, S. J. D. Bosco, S. Rohilla and M. A. Shah, J. Food Meas. Charact., 2018, 12, 892–901 Search PubMed.
- D. W. Dadi, S. A. Emire, A. D. Hagos and J.-B. Eun, Cogent Food Agric., 2019, 5, 1690316 CrossRef.
- A. Martinić, A. Kalušević, S. Lević, V. Nedović, A. Vojvodić Cebin, S. Karlović, I. Špoljarić, G. Mršić, K. Žižek and D. Komes, Food Technol. Biotechnol., 2022, 60, 237–252 Search PubMed.
- A. Dobrinčić, L. Tuđen, M. Repajić, I. E. Garofulić, Z. Zorić, V. Dragović-Uzelac and B. Levaj, Acta Aliment., 2020, 49, 475–482 Search PubMed.
- E. González, A. M. Gómez-Caravaca, B. Giménez, R. Cebrián, M. Maqueda, A. Martínez-Férez, A. Segura-Carretero and P. Robert, Food Chem., 2019, 279, 40–48 CrossRef PubMed.
- D. W. Dadi, S. A. Emire, A. D. Hagos and J.-B. Eun, Ann.: Food Sci. Technol., 2019, 20, 1690316 Search PubMed.
- C. Tongped and N. Trongsiriwat, Master’s thesis, King Mongkut’s University of Technology North Bangkok, 2025.
- M. Matra, S. Phupaboon, P. Totakul, R. Prommachart, A. A. Shah, A. M. Shah and M. Wanapat, Anim. Biosci., 2023, 37, 74 CrossRef PubMed.
- M. Matra, S. Phupaboon, P. Totakul, R. Prommachart and M. Wanapat, Int. J. Agric. Biosci., 2025, 14, 258–264 Search PubMed.
- M. Fikry, S. Jafari, K. A. Shiekh, I. Kijpatanasilp, S. Khongtongsang, E. Khojah, H. A. A. L. Jumayi and K. Assatarakul, Ultrason. Sonochem., 2024, 106949, DOI:10.1016/j.ultsonch.2024.106949.
- M. Sharma and K. K. Dash, Innovative Food Sci. Emerging Technol., 2022, 75, 102913 CrossRef CAS.
- Y. Ramakrishnan, N. M. Adzahan, Y. A. Yusof and K. Muhammad, Powder Technol., 2018, 328, 406–414 CrossRef CAS.
- K. Slinkard and V. L. Singleton, Am. J. Enol. Vitic., 1977, 28, 49–55 Search PubMed.
- S. Chheng, M. Fikry, S. Jafari, D. K. Mishra and K. Assatarakul, J. Stored Prod. Res., 2025, 112, 102660 CrossRef CAS.
- M. Fikry, Y. A. Yusof, A. M. Al-Awaadh, R. A. Rahman, N. L. Chin, E. Mousa and L. S. Chang, Food Sci. Technol. Res., 2019, 25, 351–362 CrossRef CAS.
- M. Ahmed, M. S. Akter and J. B. Eun, J. Sci. Food Agric., 2010, 90, 494–502 Search PubMed.
- L. Wen, B. Yang, C. Cui, L. You and M. Zhao, Food Anal. Methods, 2012, 5, 1244–1251 CrossRef.
- S. Chheng and K. Assatarakul, Summer Institute of Asia & ASEAN Center for Educational Research, Chiba University, Japan, 2023 Search PubMed.
- K. Papoutsis, P. Pristijono, J. B. Golding, C. E. Stathopoulos, M. C. Bowyer, C. J. Scarlett and Q. V. Vuong, J. Food Process. Preserv., 2017, 41, e13152 CrossRef.
- M. Fikry, S. Jafari, K. A. Shiekh, I. Kijpatanasilp, S. Chheng and K. Assatarakul, Food Bioprocess Technol., 2024, 1–18, DOI:10.1007/s11947-024-03421-0.
- S. Chheng, S. Jafari, D. Mishra and K. Assatarakul, Sustainable Food Technol., 2025, 3, 1865–1879 RSC.
- P. Siddhuraju and S. Manian, Food Chem., 2007, 105, 950–958 CrossRef CAS.
- I. Atalar and M. Dervisoglu, LWT-Food Sci. Technol., 2015, 60, 751–757 CrossRef CAS.
- R. K. Cruz-Bravo, D. Guajardo-Flores, C. A. Gómez-Aldapa, J. Castro-Rosas, R. O. Navarro-Cortez, L. Díaz-Batalla and R. N. Falfán-Cortés, CyTA-J. Food., 2023, 21, 493–501 CrossRef CAS.
- M. Fikry, R. Sami, A. A. Al-Mushhin, A. H. Aljahani, A. Almasoudi, S. Alharthi, K. A. Ismail and M. Dabbour, J. Biobased Mater. Bioenergy., 2022, 16, 150–158 CrossRef CAS.
- D. Gómez-Díaz, J. M. Navaza and L. Quintáns-Riveiro, Int. J. Food Prop., 2008, 11, 773–780 CrossRef.
- P. Pittia and M. Faieta, in Spray Drying Encapsulation of Bioactive Materials, CRC Press, 2021, pp. 355–376 Search PubMed.
- K. Simon-Brown, K. M. Solval, A. Chotiko, L. Alfaro, V. Reyes, C. Liu, B. Dzandu, E. Kyereh, A. G. Barnaby and I. Thompson, LWT, 2016, 70, 119–125 CrossRef CAS.
- W. Ren, G. Tian, S. Zhao, Y. Yang, W. Gao, C. Zhao, H. Zhang, Y. Lian, F. Wang and H. Du, Lwt, 2020, 133, 109954 CrossRef CAS.
- T. T. Tran and H. V. Nguyen, Beverages, 2018, 4, 84 CrossRef CAS.
- C. T. Nguyen, K. N. Di, H. C. Phan, T. C. Kha and H. C. Nguyen, Int. J. Biol. Macromol., 2024, 269, 132217 CrossRef CAS PubMed.
- J. B. J. Tay, X. Chua and C. Ang, et al. , Processes, 2021, 9, 1557 CrossRef CAS.
- T. P. Labuza and B. Altunakar, Water Activity in Foods: Fundamentals and Applications, 2020, pp. 161–205 Search PubMed.
- J. Astina and S. Sapwarobol, J. Am. Coll. Nutr., 2019, 38, 380–385 CrossRef CAS PubMed.
- O. Jordán-Suárez, J. J. Pérez-Alonso and J. J. Alvarez-Ramirez, J. Encapsulation Adsorpt. Sci., 2018, 8, 178–193 CrossRef.
- S. Y. Quek, N. K. Chok and P. Swedlund, Chem. Eng. Process., 2007, 46, 386–392 CrossRef CAS.
- P. Mishra, S. Mishra and C. L. Mahanta, Food Bioprod. Process., 2014, 92, 252–258 CrossRef CAS.
- R. V. de Barros Fernandes, S. V. Borges and D. A. Botrel, Carbohydr. Polym., 2014, 101, 524–532 CrossRef PubMed.
- E. J. G. Laureanti, T. S. Paiva, L. M. de Matos Jorge and R. M. M. Jorge, Int. J. Biol. Macromol., 2023, 253, 126969 CrossRef CAS PubMed.
- S. B. Nimse and D. Pal, RSC Adv., 2015, 5, 27986–28006 RSC.
- V. M. Silva, L. E. Kurozawa, K. J. Park and M. D. Hubinger, Drying Technol., 2012, 30, 175–184 CrossRef CAS.
- C. Cao, X. Zhao, C. Zhang, Z. Ding, F. Sun and C. Zhao, J. Food Sci., 2020, 85, 3442–3449 CrossRef CAS PubMed.
- E. M. Mudge and P. N. Brown, J. AOAC Int., 2017, 100, 18–24 CrossRef CAS PubMed.
- N. Isnaeni, A. Saefumillah and A. H. Cahyana, Indones. J. Food Drug Saf., 2024, 4, 130–144 CrossRef.
- A. Al-Hamayda, B. Abu-Jdayil, M. Ayyash and J. Tannous, Ind. Crops Prod., 2023, 205, 117556 CrossRef CAS.
- E. Díaz-Montes, Polymers, 2023, 15, 2659 CrossRef PubMed.
- D. A. Pai, V. R. Vangala, J. W. Ng, W. K. Ng and R. B. Tan, J. Food Eng., 2015, 161, 68–74 CrossRef CAS.
- H. C. Carneiro, R. V. Tonon, C. R. Grosso and M. D. Hubinger, J. Food Eng., 2013, 115, 443–451 CrossRef CAS.
- A. Tolun, Z. Altintas and N. Artik, J. Biotechnol., 2016, 239, 23–33 CrossRef CAS PubMed.
- L. Alamilla-Beltran, J. J. Chanona-Perez, A. R. Jimenez-Aparicio and G. F. Gutierrez-Lopez, J. Food Eng., 2005, 67, 179–184 CrossRef.
- Z. Zhang, S. Zhang, R. Su, D. Xiong, W. Feng and J. Chen, J. Food Sci., 2019, 84, 1427–1438 CrossRef CAS PubMed.
- R. Vehring, H. Snyder and D. Lechuga-Ballesteros, Drying Technol. Biotechnol. Pharm. Appl., 2020, 179–216 CAS.
- R. Chedoloh and S.-A. Asae, Burapha Sci. J., 2017, 78–91 Search PubMed.
- A. Dutta and G. Dutta, J. Appl. Packag. Res., 2016, 8, 5 Search PubMed.
- S. N. Dirim and G. Çalışkan, J. Food, 2012, 37(4), 203–210 Search PubMed.
- C. Wong and W. Lim, Int. Food Res. J., 2016, 23, 1887 CAS.
- O. Jiménez-González and J. Á. Guerrero-Beltrán, Food Eng. Rev., 2021, 13, 769–811 CrossRef.
- S. C. de Moura, C. L. Berling, A. O. Garcia, M. B. Queiroz, I. D. Alvim and M. D. Hubinger, Food Res. Int., 2019, 121, 542–552 CrossRef CAS PubMed.
- R. V. Tonon, C. Brabet and M. D. Hubinger, J. Food Eng., 2008, 88, 411–418 CrossRef.
- J. K. Agbenorhevi and L. J. Marshall, J. Food Sci. Eng., 2012, 2, 72 Search PubMed.
- S. Agustini and M. K. Yusya, Curr. Res. Biosci. Biotechnol., 2020, 1, 66–70 Search PubMed.
- C. Baltacioğlu, S. Velioğlu and E. Karacabey, J. Food Qual., 2011, 34, 278–283 Search PubMed.
- M. Thakur and V. Nanda, Food Chem. Adv., 2024, 4, 100567 CrossRef.
- S. Warnasih and U. Hasanah, J. Sci. Innovare, 2019, 1, 44–49 CrossRef.
- T. Okuda and H. Ito, Molecules, 2011, 16, 2191–2217 Search PubMed.
- K. Jitrangsri, A. Chaidedgumjorn and M. Satiraphan, Pharm. Sci. Asia, 2020, 47, 164–172 Search PubMed.
- S. C. Foo, F. M. Yusoff and N. M. Khong, J. Agric. Food Res., 2024, 15, 100823 CAS.
- C. Bustos-Garza, J. Yáñez-Fernández and B. E. Barragán-Huerta, Food Res. Int., 2013, 54, 641–649 CrossRef CAS.
- S. A. Desobry, F. M. Netto and T. P. Labuza, J. Food Sci., 1997, 62, 1158–1162 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





