Open Access Article
Open Access ArticleExploring the effect of dehulling and malting on the antioxidant properties, phenolic composition, and metabolite profile of Bambara groundnut (Vigna subterranea) flour obtained from three varieties
Mpho Edward Mashau *,
Thakhani Takalani,
Shonisani Eugenia Ramashia,
Nakisani Babra Moyo
*,
Thakhani Takalani,
Shonisani Eugenia Ramashia,
Nakisani Babra Moyo and
Oluwaseun Peter Bamidele
and
Oluwaseun Peter Bamidele
Department of Food Science and Technology, Faculty of Science, Engineering and Agriculture, University of Venda, Thohoyandou 0950, South Africa. E-mail: Mpho.mashau@univen.ac.za
First published on 11th May 2026
Abstract
Bambara groundnut (Vigna subterranea) is an under-exploited legume with significant nutritional potential and is known for its adaptability to challenging environmental conditions. This study evaluated the effects of dehulling and malting on the antioxidant properties, and the phenolic and metabolite profiles of Bambara groundnut (BGN) flours derived from three distinct varieties (cream, brown, and red). The antioxidant properties and phenolic profiles of BGN flour were determined using standard methods. On the other hand, the metabolomic profile of BGN flour was determined using liquid chromatography-quadrupole time-of-flight mass spectrometry (LC-qTOF-MS). Malting significantly enhanced the total phenolic content (TPC), with malted brown and red varieties having a higher value of 0.43 mg GAE/g. Furthermore, the total flavonoid content (TFC) of malted BGN flours was higher, with values ranging from 0.27 (malted cream) to 0.49 mg QE/g (malted red). Malting also increased the antioxidant activity (DPPH and FRAP) of BGN flour. On the other hand, dehulling decreased (p < 0.05) the TPC and TFC of BGN flour, and the values ranged between 0.23 (control cream) to 0.15 mg GAE/g (dehulled cream) and TFC from 0.26 (control brown) to 0.23 mg QE/g (dehulled brown) and 0.32 (control red) to 0.26 mg GAE/g (dehulled red variety). Moreover, dehulling decreased the DPPH and FRAP of the BGN flour. Both processing methods increased (p < 0.05) the levels of protocatechuic and syringic acids in the red BGN variety. Malting also increased (p < 0.05) the vanillic and ferulic acid contents of the red BGN variety. Both processing methods decreased most phenolic acids in the BGN flour samples. Dehulling and malting showed some notable differences in the metabolic profile of BGN, particularly for the red variety, while the other two varieties showed only minor differences. Dehulling was observed to increase oxylipins in red BGN, with much lower levels detected in malted and whole samples.
Sustainability spotlightBambara groundnut (Vigna subterranea) is a climate-smart crop, an under-utilised legume that is rich in nutrients and bioactive compounds. Its metabolites are related to direct and indirect sustainability wins across several SDGs, from food security and nutrition to climate resilience and sustainable livelihoods. The high protein content and micronutrients in Bambara groundnut enhance dietary quality, which is related to SDG 2 – Zero Hunger. Meanwhile, the high fibre content, antioxidants, and bioactive peptides support metabolic health and decrease the risk of diseases, aligning with SDG 3 – Good Health and Well-being. Drought tolerance and low-input growth reduce vulnerability and emissions from synthetic fertilizers, aligning with SDG 13 – Climate Action. |
1 Introduction
In addition to being an affordable dietary source of protein, legumes have attracted increasing interest in recent years owing to their beneficial effects on human health.1 The frequent consumption of legumes has been reported to help manage several diseases, such as type 2 diabetes, cancer, and cardiovascular diseases, and these benefits are associated with the presence of phytochemicals.2,3 Given that processing methods can significantly alter the composition of secondary metabolites in legumes and other plant-based foods, it is crucial to explore their effects. Rooted in Africa, the Bambara groundnut (Vigna subterranea (L.) Verdc.) is a dioecious legume known for its nutritional value and adaptability, ranking alongside peanuts and cowpeas.4,5 Bambara groundnut (BGN) is a rich source of protein, fibre, fat, carbohydrates, and indispensable amino acids such as methionine and lysine, and is regarded as a complete food.6 It can withstand poor soils, low rainfall, and harsh climates.7 The ability of BGN to sustain harsh environmental conditions and fix atmospheric nitrogen makes it suitable for sustainable agriculture,8 making it well-suited for regions prone to food scarcity and climate change in low-income countries.6The BGN grains have different colours and shapes, such as cream, brown, red, white, and black. The red and cream/black eye have big kernels and higher yield, but the red variety is harvested very late due to its late maturity compared to other BGN varieties. The cream/no-eye variety has tiny pods, while cream/brown eye has a medium kernel and has a good yield. Brown BGN has moderate- to big-sized kernels, and its colour may be light and dark brown.9 The black BGN is a very thin, moderate-sized kernel that is mainly one-seeded and matures earlier than the other BGN varieties. Seed coat colours show extensive diversity, including cream and red (26% each), black-cream and cream-purple (19.33% each), and black (9.33%).10,11 The phenotypic diversity in BGN is expressed across multiple characteristics, including colour, shape, texture, flexibility, growth habit, pilosity, and hardness in both aerial organs and underground pods.12 This extensive phenotypic variation provides the foundation for understanding how different colour variants – including brown, red, cream, and black types – may relate to underlying genetic differences.
Bambara groundnut is neglected and underutilised in Southern Africa. Traditionally, BGN is consumed by boiling freshly harvested grains and eaten as a relish with maize-meal porridge.13,14 Nonetheless, BGN has recently become a subject of interest because it is regarded as a complete food product. An area that scientists have not investigated is the biochemical composition of the different varieties of BGN using state-of-the-art equipment, including liquid chromatography-tandem mass spectrometry (LC-MS/MS). Metabolomics, the study of small molecules called metabolites within an organism's cells, effectively addresses this issue. By using sophisticated analytical methods like gas chromatography-tandem mass spectrometry (GC-MS/MS), LC-MS/MS, and nuclear magnetic resonance (NMR) spectroscopy, scientists can achieve the goal of metabolomics, which is to identify and measure bioactive substances and nutritional metabolites present in food, as well as changes caused by food processing methods.15,16
Food processing methods, such as malting, enhance the digestibility, taste, and concentration of legumes, including the bioavailability of their bioactive compounds.17 Malting produces three important enzymes, such as protease, amylase, and phytase, each providing distinct functions in the alteration of legume grain. During malting, protease hydrolyses proteins into amino acids and smaller peptides, providing a nitrogen source for the developing seedling.18 Amylase hydrolyses starches into fermentable sugars, and phytase releases phosphorus and other nutrients by breaking down phytate compounds.19 Pal et al.20 reported reduced quercetin, catechin, and gallic acid levels in germinated lentils. In contrast, malting increased the levels of volatile compounds in lentils at different times.21 Processing BGN can also help reduce antinutrients such as tannins, phytates, and trypsin inhibitors, while increasing polyphenols and oligosaccharides.22 Mudau and Adebo23 observed an increased total phenolic and flavonoid content, while the ABTS and FRAP of malted mixed BGN flour decreased.
Although some studies have evaluated the impact of processing methods, including dehulling24 and malting,25,26 on the metabolite composition of BGN, a substantial gap remains in understanding how these methods influence the overall metabolome, particularly across different BGN varieties. This represents a critical knowledge gap, especially considering the growing interest in BGN as a nutraceutical. Adetokunboh et al.25 determined the influence of malting on BGN specialty malts and syrups by focusing on primary metabolites (amino acids, fatty acids, and sugars) and volatile compounds. A study by Oladimeji and Adebo26 mainly focused on volatile compounds in malted BNG flour, whereas Adedayo et al.24 examined the impact of dehulling on the metabolite profiles of different varieties of BGN flour, providing insights into the phytochemical differences attributed to this processing technique. Despite these valuable contributions, subtle differences can easily be missed using conventional approaches for analysing mass spectrometry data. Recent advances in computational metabolomics, particularly molecular networking, allow visualisation and comparison of the entire metabolome of samples, thereby facilitating a holistic exploration of metabolic variations attributed to different food processing methods.
To the best of the knowledge of the authors, this is the first study that used ultra-high-performance liquid chromatography quadrupole time-of-flight mass spectrometry (UHPLC-qTOF-MS) to evaluate the metabolite composition of processed BGN flour from three varieties (cream, brown, and red) followed by visualisation through molecular networking. This study determined the effects of dehulling and malting on the antioxidant properties, phenolic composition, and metabolomic profile of three distinctive varieties of BGN. Apart from the study's data contributing to the body of knowledge, particularly on the functionality of processed BGN flours, it will also promote the use of processed BGN flour in the food sector, thereby achieving food security in middle-income countries. Furthermore, this study provides scientific information on how dehulling and malting affect the health-promoting compounds of BGN.
2 Materials and methods
2.1. Raw materials and chemicals
A total of 10 kg of mixed BGN grains was obtained from a supermarket (Thohoyandou, South Africa). All analytical-grade chemicals and reagents were obtained from Microsep (Johannesburg, Gauteng, South Africa). The width of the cream, brown, and red BGN grains was 9.54, 9.66, and 9.98 mm, respectively. The length of the BGN grains was: 12.02 mm (cream), 12.16 mm (brown), and 13.58 mm (red). The width and the length of 100 randomly selected BGN grains were measured by micrometre reading to 0.001 mm. The weight of the BGN grains was measured. The 1000 BGN grain mass was 576 g for cream, 582 g for brown, and 754 g for red, and the BGN grains were randomly selected, and the weight was measured.2.2. Preparation of processed Bambara groundnut flours
Bambara groundnut grains were sorted by colour (cream, brown, and red), washed to remove impurities, and soaked at room temperature for 20 h to facilitate dehulling. After discarding the soaking water, the grains were manually dehulled by removing the seed coat. A conventional oven drier was used to dry the dehulled grains for 14 h at 70 °C to obtain a moisture content of ≤ 12%.27 The dehulled grains were milled (Perten-3303 Laboratory Mill, USA), sifted using a 355 µm sieve to fine flour, and labelled as dehulled BGN flour.Bambara groundnut grains were steeped in water (5 L bucket) for 20 h at 30 ± 2 °C to facilitate malting. After draining, four trays were used to germinate BGN grains at 28 °C for 120 h,28 with water sprayed twice daily to maintain a suitable moisture content. The germinated grains were oven-dried using a conventional dryer at 70 °C for 14 h, ground into flour using an ultra-centrifugal mill (ZM 200; Retch, Germany), sifted using a 355 µm sieve to fine flour, and marked as malted BGN flour. Untreated whole BGN grains were similarly milled, sifted using a 355 µm sieve to fine flour, and designated as control BGN flour. All flours (whole, dehulled, and malted) were stored in polyethylene bags at 4 °C until further analysis.
2.3. Total phenolic content (TPC) determination
To prepare the extract, the BGN flour sample (50 g) was suspended in 500 mL of methanol. The extract was shaken for 2 h and then centrifuged (Rotina 380 R-Labotec Ecotherm (Pty) Ltd, Midrand, South Africa) for 20 min at 3000 rpm. The extract was collected, filtered using Whatman no. 1 paper, and kept at an environment of 4 °C until needed.The TPC of the BGN flour was examined as per the modified Folin-Ciocalteu method.29 Briefly, 200 mg of the flour sample was suspended in 1.5 mL of Folin Ciocalteu reagent (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 diluted with distilled water) and allowed to rest for 5 min at room temperature. Subsequently, 2.5 mL of a 7% sodium bicarbonate solution was added. The supernatant was vortexed thoroughly for one minute and centrifuged at 5000 rpm for 10 min. The absorbance of BGN flour was recorded at a wavelength of 765 nm using a UV spectrophotometer (VIS-7220N, Beijing Rayleigh Analytical Instrument Co., Ltd, China). The same spectrophotometer was used to evaluate the total flavonoid content, DPPH, and FRAP of the BGN flour. Gallic acid was used to generate a standard curve. The results were expressed as mg gallic acid equivalent per gram of dry sample.
10 diluted with distilled water) and allowed to rest for 5 min at room temperature. Subsequently, 2.5 mL of a 7% sodium bicarbonate solution was added. The supernatant was vortexed thoroughly for one minute and centrifuged at 5000 rpm for 10 min. The absorbance of BGN flour was recorded at a wavelength of 765 nm using a UV spectrophotometer (VIS-7220N, Beijing Rayleigh Analytical Instrument Co., Ltd, China). The same spectrophotometer was used to evaluate the total flavonoid content, DPPH, and FRAP of the BGN flour. Gallic acid was used to generate a standard curve. The results were expressed as mg gallic acid equivalent per gram of dry sample.
2.4. Total flavonoid content (TFC) determination
The TFC of the BGN flour samples was examined using the aluminium chloride colorimetric assay according to Mudau and Adebo,23 with some modifications. A BGN flour extract (10 µL) was suspended in 30 µL of methanol, 30 µL of AlCl3 solution, 100 µL of sodium hydroxide solution, and 10 mL of a volumetric flask was prepared by adding distilled water. The mixture was vortexed for one minute and allowed to rest for 30 min in a dark environment. The absorbance of the BGN flour samples was recorded at 450 nm using an ultraviolet spectrophotometer. Quercetin was used to generate a standard curve, and TFC was expressed as mg quercetin equivalent on a dry matter basis (mg QE/g).2.5. Analysis of DPPH (2,2-diphenyl-1-pycryl-hydroxyl) free-radical-scavenging activity
With minor modifications, the method explained by Ramatsetse et al.30 was employed to evaluate the DPPH assay of the BGN flour samples. The DPPH solution was prepared by dissolving 1.465 mg in 50 mL of methanol. BGN flour extract (2 mL) was suspended in methanol. Following this, 1 mL of DPPH solution was added, and the mixture was adjusted to 25 mL with methanol. The supernatant was properly shaken at ambient temperature (25 ± 2 °C) before being incubated for 30 min in a dark room. The absorbance was recorded at 570 nm using an ultraviolet spectrophotometer, and the results were reported as the percentage of DPPH.2.6. Ferric-reducing antioxidant power (FRAP) determination
The FRAP of the BGN flour samples was examined as explained by Mudau and Adebo23 with minor modifications. Briefly, 100 µL BGN flour was suspended in 300 mL distilled water in a test tube. Subsequently, 3 mL of FRAP reagent was added to 250 µL of BGN flour. The mixture was vortexed and placed in a water bath for 20 min at ambient temperature. An ultraviolet spectrophotometer was employed to record the absorbance of the sample at 700 nm, and the results were reported as mg GAE/g.2.7. Determination of polyphenols
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 rpm for 20 min. Nylon filters (0.22 µm) were used to filter the flour samples into HPLC vials and stored at 4 °C for further analysis. LC-MS-grade methanol, acetonitrile, and water were acquired from Microsep (Midrand, South Africa).
800 rpm for 20 min. Nylon filters (0.22 µm) were used to filter the flour samples into HPLC vials and stored at 4 °C for further analysis. LC-MS-grade methanol, acetonitrile, and water were acquired from Microsep (Midrand, South Africa).2.8. Feature-based molecular networking and metabolite annotation
Molecular networking was performed using the GNPS platform (https://gnps.ucsd.edu, accessed July 28, 2025) and its online workflow (https://ccms-ucsd.github.io/GNPSDocumentation/). Raw data from the Shimadzu LCMS-9030 q-TOF was first converted to the open-source mzML format. mzML files were pre-processed using MSDial 4.9 to obtain an mgf file and a quantification table needed for feature-based molecular networking. MS2 fragment ions within ±17 Da of the precursor m/z were filtered during uploading. The spectra were further refined by selecting the top four fragment ions in a ±50 Da window, with mass tolerances for both the precursor and fragment ions set at 0.02 Da. Visualisation of the molecular network was performed using Cytoscape. Accurate mass and MS2 fragmentation data were used to deduce empirical formulas, facilitating the annotation of matched and selected unmatched metabolite nodes. These identifications were cross-checked against dereplication databases such as KNApSAck.2.9. Statistical analysis and experimental design
All the experiments were replicated three times unless otherwise stated and results were reported as mean values. The data obtained were analysed using SPSS software (version 27.0; SPSS Inc., Chicago, Illinois, USA). Analyses were performed through analysis of variance (ANOVA) at 95% confidence and accepted at the level of significance of p < 0.05, using Duncan's multiple range test to determine significant differences among means.3 Results and discussion
3.1. Polyphenols and antioxidant activity of dehulled and malted Bambara groundnut flours
Table 1 presents the polyphenol content and antioxidant activity of processed (dehulling and malting) BGN varieties (cream, brown, and red). Dehulling decreased the TPC and TFC of BGN flours (all varieties), with data ranging from 0.23 (control cream) to 0.15 mg GAE/g (dehulled cream) and from 0.26 (control brown) to 0.23 mg QE/g (dehulled brown) and 0.32 (control red) to 0.26 mg GAE/g (dehulled red variety). The seed coat or outer layers of legumes contain higher levels of polyphenols, with higher amounts in dark varieties.32 Shitta et al.33 reported that roughly 80–90% of the total phenolic compounds in legume grains are present in the seed coats, making dehulling an effective method of removing these compounds. Furthermore, the hulls (seed coats) of BGN are rich sources of flavonoids and tannins, with red and brown varieties having higher values.34 Previous studies reported that dehulling of legumes decreases TPC by 45%, TFC by 48.6%, and tannin by 64%, respectively.35,36 Flavonoids are the standard components of legumes that can promote health. Inevitably, dehulling legumes removes notable levels of polyphenols. Therefore, the decreased TPC and TFC of dehulled BGN flours could be interrelated to the disintegration of polyphenol compounds due to seed coat removal.37 Similarly, Guajardo-Flores et al.38 observed reduced TPC of dehulled black beans, whereas Lakshmipathy et al.39 reported decreased TFC of dehulled grass peas. Nonetheless, dehulling did not affect the TPC of faba beans.40| Samples | TPC (mg GAE/g) | TFC (mg QE/g) | FRAP (mg GAE/g) | DPPH (%) |
|---|---|---|---|---|
| a Values represent the mean ± SD from three replicates; values (n = 3) within a column for the same variety with different superscripts are significantly different (p < 0.05). | ||||
| Cream | ||||
| Control | 0.23 ± 0.04b | 0.21 ± 0.02b | 0.26 ± 0.03b | 17.79 ± 1.89b |
| Dehulled | 0.15 ± 0.01c | 0.15 ± 0.01c | 0.21 ± 0.01c | 13.85 ± 2.13c |
| Malted | 0.30 ± 0.01a | 0.27 ± 0.02a | 0.43 ± 0.02a | 24.04 ± 1.85a |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Brown | ||||
| Control | 0.26 ± 0.02b | 0.26 ± 0.05b | 0.26 ± 0.04b | 21.26 ± 3.29b |
| Dehulled | 0.23 ± 0.02c | 0.23 ± 0.02c | 0.22 ± 0.04c | 14.87 ± 0.41c |
| Malted | 0.43 ± 0.03a | 0.38 ± 0.02a | 0.31 ± 0.02a | 30.08 ± 6.24a |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Red | ||||
| Control | 0.32 ± 0.01b | 0.35 ± 0.05b | 0.23 ± 0.03b | 25.71 ± 2.71b |
| Dehulled | 0.26 ± 0.04c | 0.26 ± 0.04c | 0.15 ± 0.00c | 17.46 ± 0.82c |
| Malted | 0.43 ± 0.02a | 0.49 ± 0.02a | 0.33 ± 0.02a | 34.92 ± 0.74a |
Nonetheless, other processing steps, such as soaking and drying (after dehulling), might also have contributed to the decrease in TPC and TFC of the dehulled BGN flours. These phenolic compounds might have migrated into the soaked water, and the drying treatment might also have degraded them.41 The soaking step usually allows phenolic compounds to leach from the grains into the water, creating another route for their loss.42 Higher drying temperature leads to reduced TPC, demonstrating that heat-sensitive phenolic compounds were degraded or compounds like tannins were polymerised, thereby decreasing their extractability and solubility.43 These thermal effects work alongside the mechanical hull's removal to create compound losses that extend beyond dehulling alone. The mechanisms of dehulling, soaking, and drying help explain that the low levels of phenolic acids in BGN flour could be associated with dehulling before milling.44
The TPC of the malted BGN flour increased, with the malted brown and red varieties having the same values (0.43 mg GAE/g). Compared to controls 0.23 (cream), 0.26 (brown), and 0.32 mg GAE/g (red), the malted BGN TPC results were higher, with malted cream having 0.30 mg GAE/g, malted brown and red having 0.43 mg GAE/g. The enhanced TPC of BGN flours could be ascribed to malting, which hydrolysed the cell wall with the help of enzymes and released bound phenolic compounds.45 The increased TPC might also be related to the hydrolysis of condensed procyanidins during malting.46 Moreover, the enhanced TPC of malted BGN flours could be ascribed to the integration of polyphenols by enzymes during malting and non-enzymatic reactions during kilning, which induces endogenous antioxidants.47 Enzyme synthesis occurs during malting via the phenylpropanoid pathway, shikimic acid, and degradation of the cell wall of polysaccharides, thereby releasing polyphenols that were bound earlier inside the cell wall.48 This is important because phenolic acids like p-coumaric and ferulic acids are bound to non-starch polysaccharides in the cell wall of the grain via relationships, including ester and ether bonds. Furthermore, the action of enzymes, such as esterases, that degrade these bonds releases bound phenolic acids.45 Similarly, increased TPC of the germinated brown BGN variety was reported by Chinma et al.49 Soaking of BGN grains might have also played a role in decreasing the TPC since condensed tannins were solubilised during water penetration and disruption of cellular structures.23 This structural disintegration may have allowed the liberation of polyphenols from the cell walls of the BGN grain tissue.50
The TFC of malted BGN flours was greater than that of dehulled, and data varied from 0.27 (malted cream variety) to 0.49 mg QE/g (malted red variety). This rise in TFC during malting could be ascribed to the synthesis of flavonoid compounds by chalcone isomerase. The metabolic modifications that occur in the BGN grain during malting and produce flavonoids might also be related to the rise in TFC. Moreover, the increased TFC of malted BGN flours may be ascribed to the release of glycones from conjugated glycosides that are activated by the induction of enzymes or by the modification or production of flavonoids.51 Similarly, Mudau and Adebo23 reported an increased TFC in malted mixed BGN flour.
Dehulling significantly decreased the antioxidant activity of BGN flours, with data for FRAP varying from 0.26 and 0.23 mg GAE/g (control cream, brown, and red varieties) to 0.21, 0.22, and 0.15 mg GAE/g (dehulled cream, brown, and red varieties). The same trend was noted for DPPH, with values varying from 17.79, 21.26, and 25.71% for control cream, brown, and red varieties to 13.85, 14.87, and 17.46% for dehulled cream, brown, and red varieties, respectively. The decreased antioxidant activity might be linked to the removal of the BGN hull. High levels of polyphenols in the hulls of BGN are associated with antioxidant activity. The phenolic compounds available in seed coats are closely related to their antioxidant capacities. Guajardo-Flores et al.38 indicated that removing the hull (seed coat) with high polyphenol content reduces antioxidant capacity. Similarly, Amanipour et al.52 reported lower values of FRAP (53.6 mg/TE100g) and DPPH (110.8 mg/TE100g) of dehulled grey pea flour when compared to the whole (control) grey pea flour (151.5 and 194.9 mg/TE100g).
Malting enhanced the DPPH and FRAP of BGN flours, and the data varied from 0.26 (control cream variety) to 0.43 mg GAE/g (malted cream) and from 17.79 (control cream variety) to 34.92% (malted red variety), respectively (Table 1). Enzyme activity during malting enhances the production of polyphenols from hulls to cotyledons,52 which might be the cause of the increase in the antioxidant activity of the BGN varieties. Furthermore, enzyme activity might have resulted in an array of natural processes, such as enzymatic hydrolysis, breakdown of cell walls, and depolymerisation into phenolic monomers, thereby increasing the antioxidant capacity of the malted BGN flour.53 The higher antioxidant activity of malted BGN flours demonstrates the capacity of polyphenols to modulate reactive radicals.54 The availability of antioxidants in malted BGN flours is ideal because they might reduce oxidative stress and diseases like cardiovascular disease, cancer, and diabetes mellitus. Nonetheless, the antioxidant activity differs depending on the response system. This could be associated with the production of intramolecular hydrogen bonds between functional groups, and thus, might vary in other systems.55 Similarly, Mao et al.56 reported an increased antioxidant activity in germinated chickpea flour.
3.2. Phenolic acids in processed Bambara groundnut flours
Table 2 lists the phenolic profiles in the processed BGN flour. The profiled phenolic compounds included vanillic, protocatechuic, m-coumaric, syringic, p-coumaric, ferulic, and caffeic acids. The control samples demonstrated varietal differences in phenolic content. The cream BGN flour had the highest protocatechuic acid content (839.85 µg g−1), followed by brown (821.94 µg g−1) and, red (450.09 µg g−1). Brown BGN flour is rich in ferulic acid: 96.98 µg g−1 and caffeic acid: 14.53 µg g−1. Most phenolics were lower in the red BGN flour than in the brown BGN flour, except for p-coumaric acid. These differences are probably due to genetic differences in the varieties and environmental influences on seed coat composition.57 The high content of protocatechuic acid gives cream and brown BGN varieties the potential to act as functional ingredients because of their claimed benefits, including anti-inflammatory and anti-carcinogenic properties.58| Samples | Vanillic acid | Protocatechuic acid | M-Coumaric | Syringic acid | P-coumaric | Ferulic acid | Caffeic acid |
|---|---|---|---|---|---|---|---|
| a Values represent the mean ± SD from three replicates; values within a column for the same variety with different letters are significantly different (p < 0.05). ND = Not detected. | |||||||
| Cream | |||||||
| Control | 31.81 ± 0.30a | 839.85 ± 18.67a | 4.10 ± 0.17a | 70.28 ± 5.79a | 31.64 ± 2.60a | 55.31 ± 2.29a | 7.34 ± 5.92a |
| Dehulled | 7.82 ± 0.47c | 697.88 ± 16.56c | 3.89 ± 0.14b | 13.76 ± 2.26c | 8.29 ± 2.55b | 52.45 ± 9.01a | ND |
| Malted | 21.80 ± 2.43b | 823.64 ± 18.86b | 3.92 ± 0.10b | 48.20 ± 3.02b | 10.52 ± 0.42b | 53.38 ± 1.90a | ND |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Brown | |||||||
| Control | 37.11 ± 0.91a | 821.94 ± 15.98a | 4.23 ± 0.40a | 49.33 ± 12.40a | 22.39 ± 1.98a | 96.98 ± 2.68a | 14.53 ± 0.24b |
| Dehulled | 19.26 ± 3.03a | 709.85 ± 16.33b | 3.88 ± 0.01b | 43.86 ± 13.31 ab | 10.78 ± 3.03b | 38.93 ± 2.86c | ND |
| Malted | 16.26 ± 0.37a | 814.85 ± 16.03a | 3.91 ± 0.01b | 33.73 ± 11.94c | 8.40 ± 1.29b | 67.93 ± 2.44b | 0.15 ± 0.21a |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Red | |||||||
| Control | 13.71 ± 0.08a | 450.09 ± 21.29b | 4.06 ± 0.17a | 8.17 ± 0.83c | 18.85 ± 1.25a | 44.64 ± 2.62b | ND |
| Dehulled | 12.05 ± 0.22c | 748.84 ± 22.47a | 3.90 ± 0.01b | 42.70 ± 0.30a | 9.40 ± 1.13b | 46.70 ± 1.22b | ND |
| Malted | 12.51 ± 1.40b | 746.29 ± 24.47a | 3.92 ± 0.02b | 25.42 ± 0.39b | 8.90 ± 0.61b | 76.38 ± 3.80a | ND |
Across all BGN flour varieties, dehulling consistently reduced the levels of most of the phenolic acids. Significant variations (p < 0.05) were noted in vanillic, protocatechuic, m-coumaric, and p-coumaric acids in cream and brown BGN flour and in vanillic and p-coumaric acids in the red BGN variety. For cream BGN flour, vanillic acid dropped from 31.81 (control) to 7.82 µg g−1 (dehulled), and protocatechuic acid decreased from 839.85 to 697 µg g−1. Ferulic acid decreased from 96.98 to 38.93 µg g−1 in brown BGN flour, and p-coumaric acid decreased from 18.85 to 9.40 µg g−1 in red BGN flour. This decrease was likely related to the amount of phenolic acids in the hilum (seed coat), so dehulling removes significant amounts of these compounds. Wang et al.59 demonstrated that the seed coat contains the highest level of individual-bound phenolic acids, followed by the bran and the cotyledon. This irregular distribution means that removing the seed coat via dehulling resulted in distinctive losses of phenolic acids compared to the other components of the grains. Phenolic compounds are commonly present in the outer portions of seeds as they act as protective antioxidants against environmental stressors.60 Similar data were reported on other legumes, such as cowpeas and soybeans, which showed a TPC loss of almost 50% due to dehulling.61,62
Remarkably, in the red variety, protocatechuic acid increased post-dehulling (from 450.0 to 748.84 µg g−1). This was likely due to varietal differences in the distribution of phenolics or the unmasking of bound phenolics within the endosperm during processing.63 Furthermore, milling and other processes involved in dehulling might have broken down cell walls that bind protocatechuic acid and disrupted protein-phenolic complexes, releasing it in unbound form that is more easily extracted and measured. Protocatechuic acid appears to be available in the whole red BGN seed, meaning it is retained in the final dehulled flour, unlike those polyphenols that are concentrated in the hull. The structural and biochemical properties should be examined more closely to understand these phenomena. Caffeic acid was not detected (ND) in dehulled samples of all varieties, indicating that this compound was either confined to the seed coat or was extremely vulnerable to processing conditions. These findings agree with those of previous studies that have documented the absence of some hydroxycinnamic acids in dehulled grains.64
Malting showed mixed results for phenolic compounds (where the impacts differed by phenolic compounds and variety). In cream BGN flour, vanillic acid dropped from a concentration of 31.8 (control) to 21.80 µg g−1 (malted), while protocatechuic acid remained almost unchanged (823.64 to 839.85 µg g−1). In brown BGN flour, ferulic acid was reduced from 96.98 (control) to 67.93 µg g−1 (malted), although it still exceeded the values in the dehulled samples (38.93 µg g−1).
The decreased ferulic and vanillic acids during malting might be related to phenolic acid esterases that disintegrated these compounds.65 In red BGN flour, considerable increases in ferulic acid to 76.38 (malted) from 44.64 µg g−1 (control) were observed, and syringic acid also increased from 8.17 to 25.42 µg g−1. The rise in some phenolics, such as ferulic and syringic acids, in red BGN flour might be related to the activation of some natural enzymes like phenylalanine ammonia-lyase, which is associated with the biosynthesis of phenolic acid during germination.66
Other researchers have suggested that malting processes also increase the bound phenolics released from cell wall structures, thereby increasing their extractability.67,68 The loss of some phenolics, such as vanillic acid, in cream and brown varieties of BGN might arise from leaching during soaking or oxidative decomposition during germination, as was noted in malted sorghum and barley.69 Furthermore, malting might have activated different cell wall-degrading enzymes, such as cellulases, hemicellulases, and esterases, which completely disintegrated the structural components that bind phenolic acids. In malted samples, caffeic acid levels were either reduced to undetectable levels, with trace amounts of 0.15 µg g−1 in brown BGN flour. This finding indicates that malted caffeic acid is extremely vulnerable to breakdown during malting, which could be due to the activity of polyphenol oxidases or even thermal drying.51
3.3. Metabolic profiling of Bambara groundnut flour extracts and molecular networking
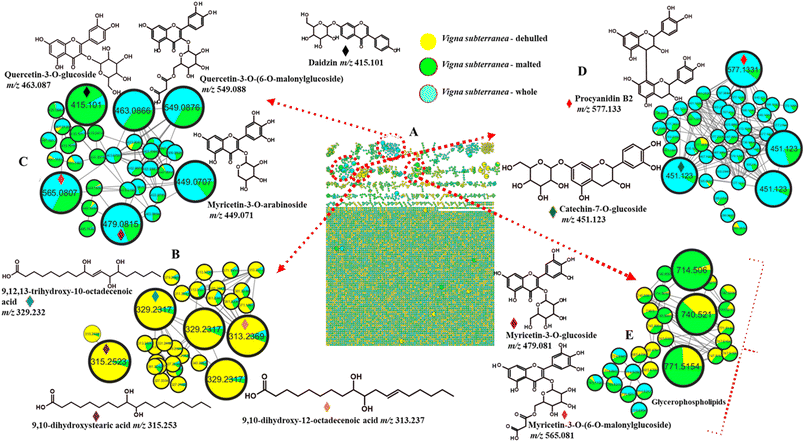
The compounds that can serve as markers associated with the effect of dehulling and malting in the present study are listed in Table 3. The results revealed that while the red BGN variety exhibited noticeable differences in its chemical composition between dehulled and malted samples (Fig. 1), the cream (Fig. 2) and brown (Fig. 3) varieties displayed relatively similar metabolite profiles under both treatments compared to the control BGN flours. Among the observed metabolic differences, dehulled red BGN flour (Fig. 1B) exhibited a high abundance of oxylipins at m/z 313.237, 315.252, and 329.232 [M–H]−, tentatively identified as 9,10-dihydroxy-12-octadecenoic acid, 9,10-dihydroxystearic acid, and 9,12,13-trihydroxy-10-octadecenoic acid. Although these compounds were also detected in whole and malted samples, their levels were lower based on peak area. All oxylipins showed a fragment ion at m/z 171, which is assumed to be characteristic of (–OOC(CH2)7CH–OH), resulting from fragmentation of the C–C bond between carbons 9 and 10 of these oxylipins.70–72 The presence of oxylipins in dehulled red BGN flour might be associated with the removal of the seed coat, which exposed the cotyledons to atmospheric oxygen.73 This exposure might have initiated the oxidation of polyunsaturated fatty acids, especially linolelaidic and elaidic acids that are abundant in BGN flour.74 Furthermore, dehulling physically disrupted cellular structures, and milling processes might have released natural enzymes that interact with their fatty acid substrates, thereby increasing oxylipin production. Thus, dehulled and milled legume flours tend to have higher amounts of aldehydes and other oxylipin compounds than the whole flour.73 The removal of the seed coat is particularly important because it provides some protection against oxidative processes, and its removal makes the cotyledon more vulnerable to enzymatic and non-enzymatic oxidation.44| BGN variety | tR (min) | Compound name | Molecular ion mass | Molecular ion formula | Fragment ions | Control | Dehulled | Malted | |
|---|---|---|---|---|---|---|---|---|---|
| a + = present; – = absent, BGN = Bambara groundnut. | |||||||||
| 1 | Red | 9.06 | Quercetin-3-O-glucoside | 463.087 | C21H20O12 | 301, 300, 151 | + | – | + |
| 2 | 8.80 | Quercetin-3-O-rutinoside | 609.144 | C27H30O16 | 463, 301, 300, 151 | + | – | + | |
| 3 | 8.40 | Myricetin-3-O-glucoside | 479.081 | C21H20O13 | 317, 316 | + | – | + | |
| 4 | 5.47 | Procyanidin B2 | 577.133 | C30H26O12 | 407, 289, 125 | + | – | + | |
| 5 | 5.83 | Procyanidin C1 | 865.196 | C45H38O18 | 695, 577, 451, 407, 289 | + | – | + | |
| 6 | 3.83 | Catechin-7-O-glucoside | 451.123 | C21H24O11 | 289 | + | – | + | |
| 7 | 8.50 | Myricetin-3-O-arabinoside | 449.070 | C20H18O12 | 316, 271 | + | – | + | |
| 8 | 9.24 | Quercetin-3-O-(6-O-malonylglucoside) | 549.087 | C24H22O15 | 301, 300 | + | – | + | |
| 9 | 8.12 | Daidzin | 415.101 | C20H20O9 | 253, 252 | + | – | + | |
| 10 | 8.54 | Myricetin-3-O-(6-O-malonylglucoside) | 565.081 | C24H22O16 | 317, 316 | + | – | + | |
| 11 | 24.62 | 1-Palmitoyl-2-oleoyl-sn-glycero-3-phosphate | 673.479 | C37H71O8P | 409, 391, 281, 255, 152 | – | + | + | |
| 12 | 24.85 | 1-Hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphoethanolamine | 714.506 | C39H74O8P | 458, 402, 279, 255 | – | + | + | |
| 13 | 25.59 | 1-(9Z-octadecenoyl)-2-(9Z,12Z-octadecadienoyl)-glycero-3-phospho-(1′-sn-glycerol) | 771.515 | C42H77O10P | 509, 507, 491, 281, 279, 152 | – | + | + | |
| – | + | + | |||||||
| 14 | 25.91 | PE 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1_18 1_18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
740.521 | C41H76NO8P | 478, 476, 281, 279 | – | + | + | |
| 7 | 14.00 | 9,12,13-Trihydroxy-10-octadecenoic acid | 329.232 | C18H34O11 | 311, 189, 171, 139 | + | + | + | |
| 8 | 17.27 | 9,10-Dihydroxy-12-octadecenoic acid | 313.237 | C18H34O4 | 295, 277, 201, 171, 127 | + | + | + | |
| 9 | 18.14 | 9,10-Dihydroxystearic acid | 315.252 | C18H36O4 | 313, 297, 171, 141, 127 | + | + | + | |
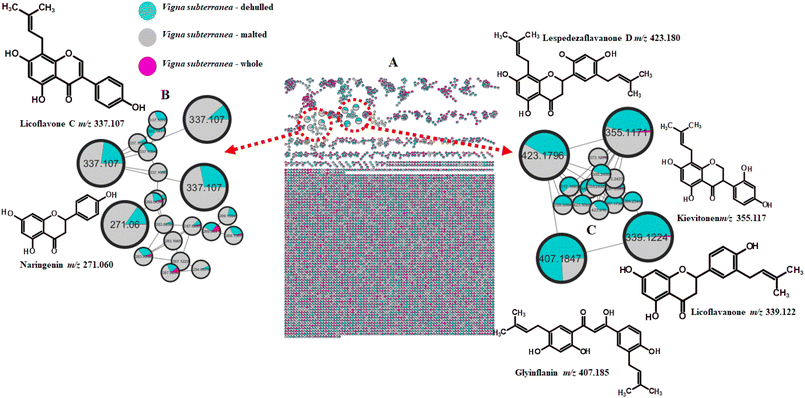
| 10 | Cream | 17.69 | Glyinflanin A | 407.185 | C25H28O5 | 229, 177 | – | + | + |
| 11 | 14.29 | Licoflavanone | 339.122 | C20H20O5 | 151, 132, 107 | + | + | + | |
| 12 | 14.78 | Kievitone | 355.117 | C20H20O6 | 193, 161 | + | + | + | |
| 13 | 18.01 | Lespedezaflavanone D | 423.179 | C25H28O6 | 193, 151, 149 | – | + | + | |
| 15 | 8.46 | Naringenin | 271.060 | C15H12O5 | 119 | – | + | + | |
| 16 | 18.65 | Licoflavone C | 337.107 | C15H12O5 | 268, 265, 240 | – | + | + | |
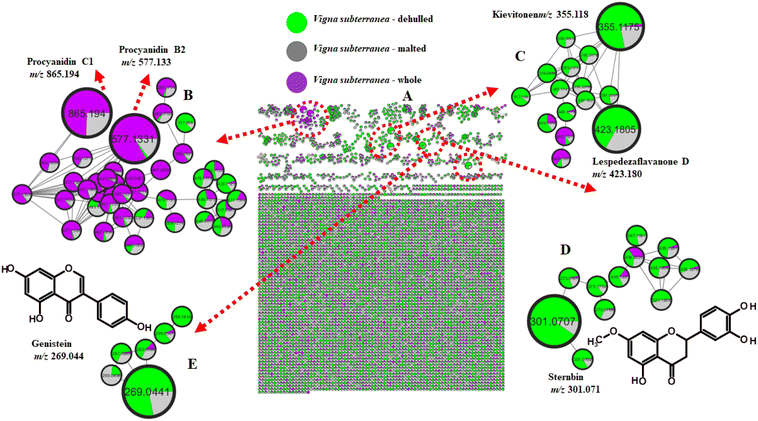
| 10 | Brown | 9.80 | Sternbin | 301.071 | C16H14O6 | 163, 137,109 | – | + | + |
| 11 | 11.10 | Genisten | 269.044 | C15H10O5 | 133 | – | + | + | |
The presence of oxylipins in malted red BGN flour might be related to enzymes such as lipases and lipooxygenases (LOX) that become activated, which are significant for the synthesis of oxylipins. The oxidation of polyunsaturated fatty acids occurs in the presence of these enzymes, resulting in the synthesis of aldehydes and other volatile compounds through complex reactions.75 Furthermore, malting involves breaking down the physical structure of seeds such that lipids become vulnerable to oxidation, producing oxylipins. The degradation of endosperm during malting exposes fat globules (oleosins) to hydrolysis and subsequent oxidation.76 Although there has been little research conducted on the oxylipin content in legume malting, research conducted on lentils has provided direct evidence of the effect of malting on oxylipins in legume flours. Malting has been found to increase the level of trans-2-nonenal, a product of free fatty acid oxidation, in all the varieties of lentils studied.77 Trans-2-nonenal is a type of oxylipin that plays a role in food flavour and aroma. An increase in this oxylipin in all the varieties of lentils studied indicates that the enzymatic processes activated in controlled germination do influence the oxylipins in the malted legume flour. Different legumes, such as soybeans, chickpeas, black beans, and lupin, have been found to contain oxylipins, both in the seeds and sprouts.78,79
Dehulled and malted flour samples from red BGN also showed the presence of glycerophospholipids at m/z 673.479, 714.506, 740.521 and 771.515 [M–H]− (Table 3) which could be the source of oxylipins observed in these samples. A compound at m/z 673.479 tentatively identified as 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphate was characterized by the presence of peaks at m/z 281 and 255 showing the presence of oleic and palmitic acid moieties, respectively.1 A 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphoethanolamine (m/z 714.506) and 1-(9Z-octadecenoyl)-2-(9Z,12Z-octadecadienoyl)-glycero-3-phospho-(1′-sn-glycerol) (m/z 771.515) as depicted in Fig. 1, showed similar fragmentation patterns where peaks responsible for fatty acid residues were observed.
The presence of glycerophospholipids in malted BGN flour might be associated with the activities of phospholipases and cell-wall degrading enzymes that disintegrate cellular structures and the membrane system, where these metabolites are typically bound. During germination, phospholipases are actively expressed and catalyse the hydrolysis of membrane glycerophospholipids, leading to membrane restructuring and lipid mobilisation.80,81 Additionally, hydrolytic enzyme activity, such as that of lipases and other enzymes responsible for the degradation of the cell wall, assists in the release of lipids, including glycerophospholipids.82 Dehulling removes the seed coat (hull), which acts as a protective barrier and may physically restrict access to intracellular components, thereby enhancing the extractability of lipids and associated compounds.20 Consequently, the removal of outer layers reduced structural barriers and facilitated the release and detection of membrane-bound phospholipids.
The red BGN flour (control and malted) also exhibited the presence of two different flavonoid classes, flavonols and flavan-3-ols, which were not observed in dehulled samples, suggesting that these flavonoids were concentrated on the seed coat. The presence of flavonols in malted red BGN flour might be associated with the activation of enzymes that hydrolyse quercetin glycosides, yielding free quercetin as the final product. This process might have been promoted by glycosyl hydrolases, which break down glycosides such as glycosylated flavonoids.83 For flavan-3-ols, the mechanisms are equally specific, but various pathways are involved. Enzyme synthesis during malting might have increased catechin content since free catechin is directly released from catechin glucosides, which are naturally available in the grain. The level of these catechin glucosides substantially rises during the malting process, contributing to substrate-releasing enzymes.84
Under the flavonol cluster, quercetin glycosides at m/z 463.086 [M–H]−, as shown in Fig. 1C and 609.144 [M–H]− (Fig. S1), were characterised by the presence of intense peaks at m/z 301 and 300 (Table 3), corresponding to the quercetin aglycone and its radical, respectively.57 Myricetin-3-O-arabinoside (m/z 449.070) and myricetin-3-O-glucoside (m/z 479.081) were also observed in samples of whole and malted red BGN flour (Fig. 1C), mainly identified by fragment ions at m/z 317 and 316, indicating the loss of a hexose sugar. Myricetin and quercetin glucosides acylated to malonic acid were also tentatively identified with the diagnostic peaks showing the presence of the aglycones in whole and malted red BGN at m/z 549.087 and 565.081, respectively. An isoflavone, daidzin (m/z 415.101 [M–H]−), was also detected in whole and malted BGN samples, and it was characterized by an intense peak at m/z 253, representative of the daidzin aglycone.85 A previous study demonstrated that a specific pre-treatment condition can achieve the highest quercetin-3-O-glucoside content, showing significant differences compared to other processing methods while maintaining levels similar to the control sample.41 In malted red BGN flour, glycosidase enzymes became active and might have converted quercetin and kaempferol glycosides into their aglycone forms, increasing the levels of free flavonoids.86 Catechin-7-O-glucoside (m/z 451.123), procyanidin B2 (m/z 577.133) (Fig. 1D), and C1 (m/z 865.196) (Fig. S1) under the subclass of flavan-3-ols revealed an intense fragment ion at m/z 289, which is characteristic of a catechin moiety.87 Fragment ions at m/z 407 for the procyanidin dimer and trimer have been reported to be a product of the retro-Diels-Alder (RDA) reaction.88
Nevertheless, procyanidin was not available in dehulled and malted BGN flour, as shown in Table 3. The absence of procyanidin in dehulled red BGN flour might be ascribed to the removal of the seed coat, which serves as a protective layer for the cotyledons and contains high amounts of phenolic compounds.52 Previous research on lentils demonstrated that catechins, procyanidins, flavanols, and flavones were abundant in the seed coat, while cotyledons only provided a small amount of phenolic compounds, such as cinnamic and benzoic acid.89 The absence of procyanidin in malted red BGN (Table 3) might be linked to polyphenol oxidase, which condensed procyanidins to form other molecules with a higher degree of polymerisation.90 Furthermore, the enzymes' activity during malting might have hydrolysed numerous components, such as phenolic compounds,52 which can degrade procyanidins, making them less available in their natural form. Another important mechanism is the loss of procyanidins due to leaching during soaking of BGN grains. Although procyanidins possess remarkable antioxidant activity, these polyphenols may decrease the bioavailability of certain nutrients and inhibit the activity of trypsin and amylase, thereby interfering with digestion.91,92 Highly pigmented legumes have been reported to contain higher phenolic content due to the presence of flavonoids, anthocyanins, and condensed tannins in their seed coat.93 Therefore, dehulling may help to reduce the levels of antinutrients, including procyanidins (condensed tannins). However, other beneficial phytochemicals might also be lost.
Several prenylflavonoids (Fig. 2 and 3C) were detected in dehulled and malted cream and brown BGN samples, all characterized by their RDA cleavage products. Licoflavanone (m/z 339.122), kievitone (m/z 355.117), lespedezaflavanone D (m/z 423.179), and licoflavone C (m/z 337.107) were putatively identified based on diagnostic RDA ions according to previous reports.94–96 While other phenolic compounds are mainly concentrated in the hull, prenylflavonoids are distributed differently within the structure of the legume seed, enabling some of them to be retained in the cotyledon part of the legume seed during the flour production process. Therefore, the presence of prenylflavonoids in dehulled legume flour means that there is retention of certain bioactive compounds during the dehulling process. The malting process seems to enhance the bioavailability and amounts of these compounds, which might be mediated by cell wall degradation and activation of dormant biosynthetic enzymes present in native BGN flour.77 This change also makes malted legume flours richer in prenylflavonoids compared to their control counterparts. Prenylflavonoids are widely distributed in legumes, as they have been reported in fermented mung bean95 and wild soybean.97
Naringenin and genistein, detected in malted and dehulled brown BGN samples, were similarly confirmed via RDA cleavage. Glyinflanin A (m/z 407.185 [M–H]−), a benzenoid, was also characterized by its RDA-derived ions (m/z 229, 177).98 The key to understanding why these compounds exist in dehulled legume flour lies in the fact that the legume plant, such as BGN, contains three unique enzymes that are exclusive to the legume plant. The enzymes include chalcone reductase, type II chalcone isomerase, and isoflavone synthase. The enzymes work in a series of steps, converting the compound chalcone and the intermediate naringenin into a series of isoflavones. This enables the legume plant to produce isoflavones, which are unique to the legume plant. Therefore, dehulled legume flour is a natural source of naringenin and its derived compound genistein.
The hydrolysis carried out by enzymes during the malting process might have effectively converted the accumulated and dormant forms of these flavonoids to their active free forms, thereby justifying the presence of these active forms of flavonoids, such as naringenin and genistein, in malted brown BGN flour compared to raw BGN flour.77 This enzymatic activity during malting increases the availability and concentration of these compounds in the final flour product. Flavan-3-ols were abundant in whole and malted brown BGN (Fig. 3B), confirming their concentration in the coloured seed coat, consistent with the red variety.
Although several compounds were identified as potential markers of the treatments applied in this study, additional compounds that may serve the same role remained unidentified, showing the effects of malting and dehulling on the secondary metabolite profile of BGN flour. This observation highlights the challenges associated with deconvoluting MS/MS data based solely on spectral information in metabolomic studies. Based on the above results, it can be said that the processing methods used in this study have positive effects on the metabolites of flour from BGN varieties. Oxylipins that were detected in abundance in dehulled red BGN flour and not in other samples showed the effects of dehulling on the metabolites. This implies that the seed coat might be chelating these metabolites, making them less detectable in whole and malted BGN.
4 Conclusions
This study examined the effect of processing methods (dehulling and malting) on polyphenol content, antioxidant activity, and phenolic and metabolomic profiles of three BGN varieties. Dehulling reduced the polyphenol content, antioxidant activity, and phenolic profiles of all varieties of BGN. Malting increased the antioxidant properties of all the BGN varieties. In terms of BGN variety, red BGN flour had higher TPC and TFC than cream and brown varieties. Dehulling and malting increased the protocatechuic and syringic acid contents of the red BGN variety. Several prenylflavonoids were markers associated with dehulled and malted cream and brown BGN flours. On the other hand, flavonoids (flavonols and flavan-3-ols) were seen to be abundant in whole and malted red BGN samples. Also, new compounds (oxylipins) were abundant in the dehulled red BGN variety in the analysed BGN flour. The presence of secondary metabolites such as flavonoids in processed BGN flours shows that they can be utilised as functional components in food systems. Nonetheless, the results of this study show that the red BGN variety is the most beneficial for further cultivation compared to the cream and brown varieties. Future studies should focus on determining the metabolomic profiles and volatile compounds of other BGN varieties, such as black, purple, and red.Author contributions
Conceptualization, M. EM.; N. B. M. and O. P. B.; methodology, M. E. M.; N. B. M. and O. P. B.; validation, M. E. M., T. T. and S. E. R.; formal analysis, M. E. M. and N. B. M; data curation, M. E. M., O. P. B., T. T. and S. E. R.; supervision, T. T. and S. E. R.; writing-original draft preparation, M. E. M and N. B. M.; writing—review and editing, M. E. M., O. P. B., T. T.; N. B. M. and S. E. R.; funding acquisition, S. E. R. and M. E. M. Project administration, O. P. B. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The article and supplementary information (SI) contain all data necessary to support the findings, statements, and conclusions. Supplementary information is available. See DOI: https://doi.org/10.1039/d5fb00611b.Acknowledgements
This project was supported by the Agricultural Research Council: “Human, Research and Innovation Capacity Development Initiative (HRICDI). An Initiative of the Department of Science and Innovation (DSI), Managed by the Agricultural Research Council (ARC), Title: Utilisation of traditional processing methods (fermentation and malting) to improve the nutritional value of cereal grains. Univen Cost center E601.References
- R. M. Ibrahim, P. M. Abdel-Baki, O. G. Mohamed, A. A. Al-Karmalawy, A. Tripathi and R. A. El-Shiekh, Metabolites profiling, in-vitro and molecular docking studies of five legume seeds for Alzheimer's disease, Sci. Rep., 2023, 14, 19637 CrossRef PubMed.
- D. J. Jenkins, C. W. Kendall, L. S. Augustin, S. Mitchell, S. Sahye-Pudaruth, S. B. Mejia, L. Chiavaroli, A. Mirrahimi, C. Ireland, B. Bashyam and E. Vidgen, Effect of legumes as part of a low glycemic index diet on glycemic control and cardiovascular risk factors in type 2 diabetes mellitus: a randomized controlled trial, Arch. Intern. Med., 2012, 172, 1653–1660 CrossRef CAS PubMed.
- D. Chinnapun and N. Sakorn, Structural characterization and antioxidant and anti-inflammatory activities of new chemical constituent from the seeds of Bambara groundnut (Vigna subterranea (L.) Verdc.)., CyTA-J. Food., 2022, 20, 93–101 CrossRef CAS.
- F. M. Oloyede, D. S. Ola and E. A. Iwalewa, E.A. Biodiversity conservation of the neglected and underutilized Nigerian horticultural crops, Acta Hortic. Regiotecturae., 2023, 26, 64–72 CrossRef.
- S. S. Sobowale, O. P. Bamidele and J. A. Adebo, Physicochemical, functional, and antinutritional properties of fermented Bambara groundnut and sorghum flours at different times, Food Chem. Adv, 2024, 4, 100729 CrossRef.
- K. E. Ramatsetse, S. E. Ramashia and M. E. Mashau, A review on health benefits, antimicrobial and antioxidant properties of Bambara groundnut (Vigna subterranean), Int. J. Food Prop., 2023, 26, 91–107 CrossRef CAS.
- A. Bhartiya, J. P. Aditya and L. Kant, Nutritional and remedial potential of an underutilized food legume horsegram (Macrotyloma uniflorum): A review, J. Anim. Plant Sci., 2015, 25, 908–920 CAS.
- A. Soumare, A. G. Diedhiou and A. Kane, Bambara groundnut: a neglected and underutilized climate-resilient crop with great potential to alleviate food insecurity in sub-Saharan Africa, J. Crop Improv., 2022, 36, 747–767 CrossRef.
- Department of Agriculture, Forestry & Fisheries. Production Guideline for Bambara Groundnuts: Pretoria, South Africa, Published by Directorate Agricultural Information Services & Department of Agriculture, Forestry & Fisheries, 2011, pp. 1–10, URL https://www.nda.agri.za/docs/Brochures/ProguideBambara.pdf. accessed 01 March, 2026 Search PubMed.
- M. Khan, M. Y. Rafii, S. I. Ramlee, M. Jusoh and M. Al-Mamun, Bambara groundnut (Vigna subterranea L. Verdc): A crop for the new millennium, its genetic diversity, and improvements to mitigate future food and nutritional challenges, Sustainability, 2021, 13, 5530 CrossRef CAS.
- M. Sari, S. Ilyas, M. Rahmad-Suhartanto and A. Qadir, Pre-harvest sprouting on high-level seed dormancy of Bambara groundnut (Vigna subterranea) landraces, Biodiversitas, 2021, 22, 5617–5623 Search PubMed.
- B. S. Bonny, D. Seka, K. Adjoumani, K. G. Koffi, L. C. Kouonon and R. S. Sié, Evaluation of the diversity in qualitative traits of Bambara groundnut germplasm (Vigna subterranea (L.) Verdc.) of Côte d'Ivoire, Afr. J. Biotechnol., 2019, 18, 23–36 CrossRef.
- S. A. Oyeyinka, S. Singh, P. O. Adebola, A. S. Gerrano and E. O. Amonsou, Physicochemical properties of starches with variable amylose contents extracted from Bambara groundnut genotypes, Carbohydr. Polym., 2015, 133, 171–178 CrossRef CAS PubMed.
- K. P. Baviskar, D. V. Jain, S. D. Pingale, S. S. Wagh, S. P. Gangurde, S. A. Shardul, A. R. Dahale and K. S. Jain, A review on hyphenated techniques in analytical chemistry, Curr. Anal. Chem., 2022, 18, 956–976 CrossRef CAS.
- C. Li and J. A. Yergey, Continuous flow liquid secondary ion mass spectrometric characterization of phospholipid molecular species, J. Mass Spectrom., 1997, 32, 314–322 CrossRef CAS.
- M. M. Pedrosa, E. Guillamón and C. Arribas, Autoclaved and extruded legumes as a source of bioactive phytochemicals: A review, Foods, 2021, 10, 379 CrossRef CAS PubMed.
- M. Bahmani, C. O'Lone, A. Juhász, M. G. Nye-Wood, H. Dunn, I. B. Edwards and M. L. Colgrave, Application of mass spectrometry-based proteomics to barley research, J. Agric. Food Chem., 2021, 69, 8591–8609 CrossRef CAS PubMed.
- E. Neylon, E. K. Arendt, K. M. Lynch, E. Zannini, P. Bazzoli, T. Monin and A. W. Sahin, Rootlets, a malting by-product with great potential, Fermentation, 2020, 6, 117 CrossRef CAS.
- R. S. Pal, A. Bhartiya, P. Yadav, L. Kant, K. K. Mishra, J. P. Aditya and A. Pattanayak, A. Effect of dehulling, germination and cooking on nutrients, anti-nutrients, fatty acid composition and antioxidant properties in lentil (Lens culinaris), J. Food Sci. Technol., 2017, 54, 909–920 CrossRef CAS PubMed.
- M. Nafula, D. Ongeng, D. Alowo, L. Nabatanzi and S. Zziwa, Effect of cereal based traditional malting technology on nutritional quality of iron-rich bean flour, Afr. Crop Sci. J., 2022, 30, 85–107 CrossRef.
- X. L. Tan, S. Azam-Ali, E. V. Goh, M. Mustafa, H. H. Chai, W. K. Ho, S. Mayes, T. Mabhaudhi, S. Azam-Ali and F. Massawe, F. (). Bambara groundnut: An underutilized leguminous crop for global food security and nutrition, Front. Nutr., 2020, 7, 601496 CrossRef PubMed.
- M. Mudau and O. A. Adebo, Effect of traditional and novel processing technologies on the thermo-pasting, microstructural, nutritional, and antioxidant properties of finger millet and Bambara groundnut flours, Int. J. Food Sci. Technol., 2025, 60, vvae037 CrossRef CAS.
- B. C. Adedayo, T. A. Anyasi, M. J. Taylor, F. Rautenbauch, M. Le Roes-Hill and V. A. Jideani, Phytochemical composition and antioxidant properties of methanolic extracts of whole and dehulled Bambara groundnut (Vigna subterranea) seeds, Sci. Rep., 2021, 11, 14116 CrossRef CAS PubMed.
- A. H. Adetokunboh, A. O. Obilana and V. A. Jideani, Physicochemical characteristics of Bambara groundnut speciality malts and extract, Molecules, 2022, 27, 4332 CrossRef CAS PubMed.
- B. M. Oladimeji and O. A. Adebo, Properties and metabolite profiling of Bambara groundnut flour as affected by different food processing conditions, Appl. Food Res., 2024, 4, 100389 CrossRef CAS.
- D. Yahaya, O. A. Seidu, C. H. Tiesaah and M. B. Iddrisu, The role of soaking, steaming, and dehulling on the nutritional quality of Bambara groundnuts (Vigna subterranea (L) Verdc.), Front. Sustain. Food Syst., 2022, 6, 887311 CrossRef.
- M. A. Akpapunam, S. O. Igbedioh and I. Aremo, I. Effect of malting time on chemical composition and functional properties of soybean and Bambara groundnut flours, Int. J. Food Sci. Nutr., 1996, 47, 27–33 CrossRef CAS PubMed.
- L. M. Mahloko, H. Silungwe, M. E. Mashau and T. E. Kgatla, Bioactive compounds, antioxidant activity and physical characteristics of wheat-prickly pear and banana biscuits, Heliyon, 2019, 5, e02479 CrossRef CAS PubMed.
- K. E. Ramatsete, S. E. Ramashia and M. E. Mashau, Effect of partial mutton meat substitution with Bambara groundnut (Vigna subterranea (L.) Verdc.) flour on physicochemical properties, lipid oxidation, and sensory acceptability of low-fat patties, Food Sci. Nutr., 2024, 12, 4019–4037 CrossRef PubMed.
- B. Moyo, N. T. Tavengwa and N. E. Madala, Diverse chemical modifications of the chlorogenic acid composition of Viscum combreticola Engl: A premise for the state of readiness against excessive sunlight exposure, J. Photochem. Photobiol B., 2022, 233, 112501 CrossRef CAS PubMed.
- A. Singh and S. Sharma, S. Bioactive components and functional properties of biologically activated cereal grains: a bibliographic review, Crit. Rev. Food Sci. Nutr., 2017, 57, 3051–3071 CrossRef CAS PubMed.
- N. S. Shitta, A. Chukwudi Edemodu, W. Gebreselassie Abtew, and A. Abebe Tesfaye, A. A Review on the cooking attributes of African yam bean (Sphenostylis stenocarpa). Legumes Research - Volume 2, ed. J. C. Jimenez-Lopez and A. Clemente, InTechOpen, 2021, Croatia Search PubMed.
- T. Harris, V. Jideani, M. Le Roes-Hill and M. Flavonoids, and tannin composition of Bambara groundnut (Vigna subterranea) of Mpumalanga, South Africa, Heliyon, 2018, 4, e00833 CrossRef PubMed.
- A. Nelom, B. B. Otchon, D. Souina and N. M. Antoine, Variability and dehulling effect on seed antinutrients and antioxidant activity of Cowpea (Vigna unguiculata L. Walp.) genotypes grown in two agroecological zones of Chad, EAS J. Biotechnol. Genet., 2023, 5, 1–9 CrossRef.
- A. M. Nakitto, J. H. Muyonga and D. Nakimbugwe, Effects of combined traditional processing methods on the nutritional quality of beans, Food Sci. Nutr., 2015, 3, 233–241 CrossRef CAS PubMed.
- J. N. C. Okafor, V. A. Jideani, M. Meyer and M. Le Roes-Hill, Bioactive components in Bambara groundnut (Vigna subterraenea (L.) Verdc) as a potential source of nutraceutical ingredients, Heliyon, 2022, 8, e09024 CrossRef CAS PubMed.
- D. Guajardo-flores, E. P´erez-carrillo, I. Romo-lopez, L. E. Ramírez-valdez and J. A. Guti´errezuribe, Effect of dehulling and germination on physicochemical and pasting properties of black beans (Phaseolus vulgaris L.), Cereal Chem., 2017, 94, 98–103 CrossRef CAS.
- K. Lakshmipathy, B. Buvaneswaran, A. Ashish Rawson and D. V. Chidanand, Effect of dehulling and germination on the functional properties of grass pea (Lathyrus sativus) flour, Food Chem., 2024, 449, 139265 CrossRef CAS PubMed.
- I. Rajhia, M. Mondher Boulaaba, B. Baccouric, F. Rajhi, J. Hammamia, F. Barhoumia, G. Flaminie and H. Mhadhbi, Assessment of dehulling effect on volatiles, phenolic compounds and antioxidant activities of faba bean seeds and flours, South Afr. J. Bot., 2022, 147, 741–753 CrossRef.
- A. Mejía-Terán, C. M. Blanco-Lizarazo, J. E. Leiva Mateus, I. Sotelo-Díaz, D. Mejía Terán, E. Geffroy and E. Pretreatments, particle size on the glycemic index and rheological and functional food properties of bean flours, Int. J. Food Sci., 2024, 6336837 Search PubMed.
- M. H. Kamani, J. Liu, S. M. Fitzsimons, M. A. Fenelon and E. G. Murphy, Determining the influence of fava bean pre-processing on extractability and functional quality of protein isolates, Food Chem: X, 2024, 21, 101200 CAS.
- K. Ben Saıd, A. Hedhili, S. Bellagha, H. Gliguem and M. Dufrechou, M. The potential of combining faba bean (Vicia faba L.) and pea pod (Pisum sativum L.) flours to enhance the nutritional qualities of food products, Foods, 2025, 14, 2167 CrossRef PubMed.
- M. Consumi, G. Tamasi, C. Bonechi, M. Andreassi, G. Leone, A. Magnani and C. Rossi, Effect of flaking and precooking procedures on antioxidant potential of selected ancient cereal and legume flours, Foods, 2022, 11, 1592 CrossRef CAS PubMed.
- J. X. Perales-Sánchez, C. Reyes-Moreno, M. A. Gómez-Favela, J. Milán-Carrillo, E. O. Cuevas- Rodríguez, A. Valdez-Ortiz, R. Gutiérrez-Dorado and R. Plant, Foods Human Nutr, 2014, 69, 196–202 CrossRef PubMed.
- O. P. Ibidapo, F. O. Henshaw, T. A. Shittu and W. A. Afolabi, Bioactive components of malted millet (Pennisetum glaucum), Soy residue “okara” and wheat flour and their antioxidant properties, Int. J. Food Prop., 2019, 22, 1886–1898 CrossRef CAS.
- J. Wannenmacher, M. Gastl and T. Becker, T. Phenolic substances in beer: Structural diversity, reactive potential and relevance for brewing process and beer quality, Compr. Rev. Food Sci. Food Saf., 2018, 17, 953–988 CrossRef CAS PubMed.
- P. J. Cáceres, C. Martínez-Villaluenga, L. Amigo and J. Frias, J. Maximising the phytochemical content and antioxidant activity of Ecuadorian brown rice sprouts through optimal germination conditions, Food Chem., 2014, 152, 407–414 CrossRef PubMed.
- C. E. Chinma, J. O. Abu, B. N. Asikwe, T. Sunday and O. A. Adebo, Effect of germination on the physicochemical, nutritional, functional, thermal properties and in vitro digestibility of Bambara groundnut flours, LWT- Food Sci. Technol., 2021, 140, 110749 CrossRef CAS.
- M. Emkani, B. Oliete and R. Saurel, R. Effect of lactic acid fermentation on legume protein properties, a review, Fermentation, 2022, 8, 244 CrossRef CAS.
- R. Sharma, M. Bhandari, S. Sharma and R. Bhardwaj, Compositional, structural, and functional characteristics of millets as modified by bioprocessing techniques: A review, J. Food Process. Preserv., 2022, 46, e16885 CAS.
- A. Amanipour, Y. Samaei, O. Böök, Y. Granfeldt and C. E. Lazarte, Impact of dehulling, germination, and fermentation on the bioactive and functional properties of grey pea flour, Front. Nutr., 2024, 11, 1478399 CrossRef PubMed.
- Y. O. Kewuyemi, C. E. Chinma, H. Kesa, O. Alabi, E. O. Amonsou and A. O. Adebo, Rheological, thermal and physicochemical properties of bioprocessed flour from cowpea, sorghum and orange fleshed sweet potato, J. Food Process Eng., 2024, 47, e14777 CrossRef CAS.
- S. Boudjou, B. D. Oomah, F. Zaidi and F. Hosseinian, Phenolics content and antioxidant and anti-inflammatory activities of legume fractions, Food Chem., 2013, 138, 1543–1550 CrossRef CAS PubMed.
- J. Chen, J. Yang, L. Ma, J. Li, N. Shahzad and C. K. Kim, C.K. Structure-antioxidant activity relationship of methoxy, phenolic hydroxyl, and carboxylic acid groups of phenolic acids, Sci. Rep., 2020, 10, 2611 CrossRef CAS PubMed.
- H. Mao, S. Yuan, Q. Li, X. Zhao, X. Zhang, H. Liu, M. Yu and M. Wang, Influence of germination on the bioactivity, structural, functional and volatile characteristics of different chickpea flours, Food Chem. X, 2024, 21, 101195 CrossRef CAS PubMed.
- V. Nyau, S. Prakash, J. Rodrigues and J. Farrant, Identification of nutraceutical phenolic compounds in Bambara groundnuts (Vigna subterranea L. Verdc) by HPLC-PDA-ESI-MS, Braz. J. Appl. Sci. Technol., 2014, 6, 77–85 CrossRef.
- M. B. S. Al-Shuhaib and J. M. Al, Shuhaib Assessing Therapeutic Value and Side Effects of key botanical compounds for optimized medical treatments, Chem. Biodiversity, 2025, 22, 02401754 CrossRef PubMed.
- K. Y. Khan, B. Ali, G. Li, B. Iqbal, N. R. Siddiqui, S. Jabbar, I. Ali, A. M. Hassan, D. N. Binjawhar, U. K. Abdel-Hameed, S. Fahad and D. Du, Distribution of nutrients, bioactive compounds, and antioxidant properties of grain-based milling fractions of Glycine max L, CyTA – J. Food., 2024, 22, 2290831 CrossRef.
- S. Pratyusha. Phenolic Compounds in the Plant Development and Defense. Plant Stress Physiology: Perspectives in Agriculture, ed. M. Hasanuzzaman, and K. Nahar, InTechOpen, Croatia, 2022, pp. 1–17 Search PubMed.
- N. S. Affrifah, R. D. Phillips and F. K. S. Cowpeas, Nutritional profile, processing methods and products—A review, Legume Sci., 2022, 4, e131 CrossRef CAS.
- D. Kathuria, A. K. Dhiman, S. Attri and M. Kumar, Effect of processing method on quality characteristics of harit soybean (Glycine max): in vitro protein digestibility, HPLC, FTIR analysis, Nutr. Food Sci, 2022, 52, 684–697 CrossRef.
- B. A. Acosta-Estrada, J. A. Gutiérrez-Uribe and S. O. Serna-Saldivar, Bound phenolics in foods: a review, Food Chem., 2014, 152, 46–55 CrossRef CAS PubMed.
- Y. Salinas-Moreno, P. Scott and A. Domínguez-López, Total phenolic acids and hydroxycinnamates in the grain tissues of brown midrib (bm) maize mutants, Cereal Res. Comm., 2025, 53, 969–978 CrossRef CAS.
- K. Vingrys, M. Mathai, J. F. Ashton, L. Stojanovska, T. Vasiljevic, A. J. McAinch and O. N. Donkor, The effect of malting on phenolic compounds and radical scavenging activity in grains and breakfast cereals, J. Food Sci., 2022, 87, 4188–4202 CrossRef CAS PubMed.
- J. O. Ampofo and M. Ngadi, Stimulation of the phenylpropanoid pathway and antioxidant capacities by biotic and abiotic elicitation strategies in common bean (Phaseolus vulgaris) sprouts, Process Biochem., 2021, 100, 98–106 CrossRef CAS.
- S. Olivares-Galván, M. L. Marina and M. C. García, Extraction of valuable compounds from brewing residues: Malt rootlets, spent hops, and spent yeast, Trends Food Sci. Technol., 2022, 127, 181–197 CrossRef.
- H. Rani and R. D. Bhardwaj, Quality attributes for barley malt: The backbone of beer, J. Food Sci., 2021, 86, 3322–3340 CrossRef CAS PubMed.
- M. H. Dicko, H. Gruppen, A. S. Traore, W. J. H. Van Berkel and A. G. J. Voragen AGJ, Evaluation of the effect of germination on phenolic compounds and antioxidant activities in sorghum varieties, J. Agric. Food Chem., 2005, 53, 2581–2588 CrossRef CAS PubMed.
- N. P. Nørskov, M. S. Hedemann, P. K. Theil and K. E. Knudsen, Oxylipins discriminate between whole grain wheat and wheat aleurone intake: a metabolomics study on pig plasma, Metabolomics, 2013, 9, 464–479 CrossRef.
- V. D. Garcia, M. Fronza, R. Von Borowski, G. Alves-Silva, A. R. Zimmer, T. Ruaro, S. C. Gnoatto, A. Dallegrave and R. M. Silveira, First report of chemical composition and cytotoxicity evaluation of Foraminispora rugosa basidiomata from Brazil, Bot. Stud., 2022, 63, 33 CrossRef CAS PubMed.
- M. A. Acquavia, J. J. Benítez, G. Bianco, M. A. Crescenzi, J. Hierrezuelo, M. Grifé-Ruiz, D. Romero, S. Guzmán-Puyol and J. A. Heredia-Guerrero, Incorporation of bioactive compounds from avocado by-products to ethyl cellulose-reinforced paper for food packagig applications, Food Chem., 2023, 429, 136906 CrossRef CAS PubMed.
- A. Badjona, R. Bradshaw, C. Millman, M. Howarth, B. N. Dubey and B. N. Faba, bean flavor effects from processing to consumer acceptability, Foods, 2023, 12, 2237 CrossRef CAS PubMed.
- M. E. Mashau, T. Takalani, O. P. Bamidele and S. E. Ramashia, Elucidation of nutritional quality, antinutrients, and protein digestibility of dehulled and malted flours produced from three varieties of Bambara groundnut (Vigna subterranean), Foods, 2025, 14, 2450 CrossRef CAS PubMed.
- A. Czubaszek, M. Gertchen, A. Gasiński, J. Miedzianka and J. Kawa-Rygielska, Nutritional quality rye bread with the addition of selected malts from beans, Molecules, 2025, 30, 1006 CrossRef CAS PubMed.
- E. Opiela, A. Czubaszek, A. Gasiński, J. Miedzianka and J. Kawa-Rygielska, Malted soybeans as a substrate for plant-based beverages—Analysis of nutritional properties, antioxidant activity, and volatiles, Molecules, 2025, 30, 3845 CrossRef CAS PubMed.
- A. Gasiński, J. Kawa-Rygielska and J. Malting, A method for modifying volatile composition of black, brown and green lentil seeds, PLoS One, 2023, 18, e0290616 CrossRef PubMed.
- J. Arora, B. Kanthaliya, A. Joshi, M. Meena, S. Meena, M. H. Siddiqui, S. A. Alamri and H. P. Devkota, Evaluation of total isoflavones in chickpea (Cicer arietinum L.) sprouts germinated under precursors (p-coumaric acid and L-phenylalanine) Supplementation, Plants, 2023, 12, 2823 CrossRef CAS PubMed.
- A. Galanty, P. Zagrodzki, M. Miret and P. Paśko, Chickpea and lupin sprouts, stimulated by different LED lights, as novel examples of isoflavones-rich functional food, and their impact on breast and prostate cells, Molecules, 2022, 27, 9030 CrossRef CAS PubMed.
- W. Wang, S. Ryu and X. Wang, The functions of phospholipases and their hydrolysis products in plant growth, development and stress responses, Prog. Lipid Res., 2022, 86, 101158 CrossRef PubMed.
- C. May, R. Preisig-Müller, M. Höhne, P. Gnau and H. Kindl, A phospholipase A2 is transiently synthesized during seed germination and localized to lipid bodies, Biochim. Biophys., 2023, 1393, 267–276 CrossRef PubMed.
- I. A. Graham, I.A. Seed storage oil mobilization, Annu. Rev. Plant Biol., 2008, 59, 115–142 CrossRef CAS PubMed.
- M. A. Cipollone, A. G. Abraham, A. R. Fontana and V. A. Tironi, Autochthonous fermentation as a means to improve the bioaccessibility and antioxidant activity of proteins and phenolic compounds of yellow pea flour, Foods, 2024, 659, 13 Search PubMed.
- D. O. Carvalho, A. F. Curto and L. F. Guido, Determination of phenolic content in different barley varieties and corresponding malts by liquid chromatography-diode array detection-electrospray ionization tandem mass spectrometry, Antioxidants, 2015, 4, 563–576 CrossRef CAS PubMed.
- C. Fang, X. Wan, H. Tan and C. Jiang, Identification of isoflavonoids in several kudzu samples by high-performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry, J. Chromatogr. Sci., 2006, 44, 57–63 CAS.
- G. Bonassi and V. Lavelli, Hydration and fortification of common bean (Phaseolus vulgaris L.) with grape skin phenolics—Effects of ultrasound application and heating, Antioxidants, 2024, 13, 615 CrossRef CAS PubMed.
- D. Escobar-Avello, J. Lozano-Castellón, C. Mardones, A. J. Pérez, V. Saéz, S. Riquelme, D. von Baer and A. Vallverdú-Queralt, Phenolic profile of grape canes: Novel compounds identified by lc-esi-ltq-orbitrap-ms, Molecules, 2019, 24, 3763 CrossRef CAS PubMed.
- M. D. Rush, E. A. Rue, A. Wong, P. Kowalski, J. A. Glinski and R. B. van Breemen, Rapid determination of procyanidins using MALDI-ToF/ToF mass spectrometry, J. Agric. Food Chem., 2018, 66, 11355–11361 CrossRef CAS PubMed.
- D. Pathiraje, J. Carlin, T. J. Der, J. P. Wanasundara and P. J. Shand, Generating multi-functional pulse ingredients for processed meat products—Scientific evaluation of infrared-treated lentils, Foods, 2023, 12, 1722 CrossRef CAS PubMed.
- E. Borges-Martínez, T. Gallardo-Velázquez, A. Cardador-Martínez, D. D. Moguel-Concha, G. Osorio-Revilla, J. C. Ruiz-Ruiz and C. J. Martínez, Phenolic compounds profile and antioxidant activity of pea (Pisum sativum L.) and black bean (Phaseolus vulgaris L.) sprouts, Food Sci. Technol., 2021, v42, e45920 Search PubMed.
- V. Samukha, F. Fantasma, G. D’Urso, E. Colarusso, A. Schettino, N. Marigliano, M. G. Chini, G. Saviano, V. De Felice, G. Lauro and F. Maione, Chemical profiling of polar lipids and the polyphenolic fraction of commercial Italian phaseolus seeds by UHPLC-HRMS and biological evaluation, Biomolecules, 2024, 20(14), 1336 CrossRef PubMed.
- R. Goncalves, N. Mateus, I. Pianet, M. Laguerre and V. de Freitas, Mechanisms of tannin-induced trypsin inhibition: a molecular approach, Langmuir, 2011, 27, 13122–13129 CrossRef CAS PubMed.
- C. Chávez-Mendoza and E. Sánchez, Bioactive compounds from Mexican varieties of the common bean (Phaseolus vulgaris): Implications for health, Molecules, 2017, 22, 1360 CrossRef PubMed.
- M. Alsafran and D. M. Kasote, HPLC-QTOF-MS-based chemical Profiling and antioxidant activity of Qatari propolis, Int. J. Food Prop., 2026, 29, 2602276 CrossRef.
- S. Lang, L. Liu, Z. Li, S. Liu, J. Liang, L. Lu and L. Wang, Untargeted metabolomics reveals phenolic compound dynamics during mung bean fermentation, Food Chem: X., 2025, 31, 103189 CAS.
- X. Zhang, W. Yao, Y. Tang, J. Ye, T. Niu, L. Yang, R. Wang and Z. Wang, Coupling the isopentenol utilization pathway and prenyltransferase for the biosynthesis of licoflavanone in recombinant Escherichia coli, J. Agric. Food Chem., 2024, 72, 15832–15840 CrossRef CAS PubMed.
- T. Xu, M. Yang, Y. Li, X. Chen, Q. Wang, W. Deng, X. Pang, K. Yu, B. Jiang, S. Guan and D. A. Guo, An integrated exact mass spectrometric strategy for comprehensive and rapid characterization of phenolic compounds in licorice, Rapid Commun. Mass Spectrom. Rapid Communications in Mass Spectrometry, 2013, 27, 2297–2309 CrossRef CAS PubMed.
- N. Bisht, A. Gupta, P. Awasthi, A. Goel, D. Chandran, N. Sharma and N. Singh, Development of a rapid LC-MS/MS method for the simultaneous quantification of various flavonoids, isoflavonoids, and phytohormones extracted from Medicago truncatula leaves, J. Liq. Chromatogr. Relat. Technol., 2021, 44, 776–787 CrossRef CAS.
- Y. J. Li, J. Chen, Y. Li, Q. Li, Y. F. Zheng, Y. Fu and P. Li, Screening and characterization of natural antioxidants in four Glycyrrhiza species by liquid chromatography coupled with electrospray ionization quadrupole time-of-flight tandem mass spectrometry, J. Chromatogr. A., 2011, 1218, 8181–8191 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |