Open Access Article
Open Access ArticlePotential of utilizing plant stem waste as an excellent source of dietary fiber for novel food applications
P. D. N. Boteju a,
C. M. Senanayake
a,
C. M. Senanayake b and
K. G. L. R. Jayathunge
b and
K. G. L. R. Jayathunge *ab
*ab
aFaculty of Graduate Studies, University of Sri Jayewardenepura, Nugegoda 10250, Sri Lanka. E-mail: lasanthi@sjp.ac.lk
bDepartment of Biosystems Technology, Faculty of Technology, University of Sri Jayewardenepura, Pitipana, Homagama, Sri Lanka
First published on 11th March 2026
Abstract
Substantial volumes of plant stem residues are formed annually as residues of agriculture and byproducts of food processing. Yet, their potential as a rich dietary fiber source has remained largely unexplored. These lignocellulosic biomasses are naturally rich in structural polysaccharides that could serve as functional ingredients to improve the nutritional and technological quality of food products. However, over the past years, the utilization of plant stem waste for dietary fiber valorization has received minimal scientific attention, leaving a significant research gap in sustainable food interventions. This review aims to highlight the potential of plant stem waste as a valuable source of dietary fiber. It underscores that the functional properties of stem-derived fibers are primarily influenced by lignification, fiber composition, and cell wall structure rather than by total fiber content alone. The application of green extraction technologies is shown to enhance fiber extractability, solubility, and functional attributes. Furthermore, by integrating principles of reuse, recyclability, and value addition within a circular economy framework, the review presents innovative strategies for developing novel food applications that enhance nutritional quality, improve functional performance, and contribute to environmental and economic sustainability in agri-food systems.
Sustainability spotlightThis review highlights the potential of valorizing plant stem waste as a rich source of dietary fiber, contributing to sustainable food innovation. Stems, often discarded during processing, contain valuable cellulose, hemicellulose, and pectins that can be efficiently recovered using green extraction technologies such as ultrasound-, microwave-, and subcritical water–assisted methods. Incorporating stem-derived fibers into bakery, dairy alternatives, beverages, and functional foods not only enhances nutritional value but also supports circular economy principles by reducing agro-industrial waste. This approach is going to fill the existing gap in utilizing underexplored biomass for sustainable, fiber-enriched food applications while directly contributing to Sustainable Development Goals (SDGs) 2, 3, and 12—Zero Hunger, Good Health and Well-being, and Responsible Consumption and Production. |
1. Introduction
Plant-based agri-food waste refers to waste generated along the agri-food chain from production to consumption. This waste includes leaves, straws, seed pods, stalks, bagasse, or husks. Although these are usually discarded, they are edible and also contain important nutritional components, including carbohydrates, proteins, dietary fibre, phytochemicals, and antioxidants. According to the Food and Agriculture Organization (FAO) of the United Nations, nearly one-third of global food production, equivalent to approximately 1.3 billion tons, is wasted annually.1 Plant-based agri-food waste can be categorized into two primary types: field residues and process residues. Field residues refer to the organic materials that remain in agricultural fields after crops have been harvested. In contrast, process residues are the byproducts generated during the processing of crops into value-added products, which persist even after the primary agricultural output has been transformed into a final product.2 As agri-food industries generate enormous amounts of waste, this presents an important research area aimed at minimizing and managing them efficiently to support the zero waste and circular economy concepts.3 Although numerous reviews have broadly examined the valorization of agri-food waste and the recovery of dietary fiber, many take a generalized stance that combines various by-products such as peels, pomace, husks, and bran, or focus on non-food uses like biofuels, biomaterials, and energy generation. In contrast, plant stem wastes, including stems, stalks, cores, and pseudostems, are relatively underexplored as a separate category, despite their distinct lignocellulosic structure and consistently high fiber content. The structural and compositional characteristics of stems differ fundamentally from other plant residues, directly influencing fiber extractability, hydration behavior, fermentability, and food application potential. Among various types of agri food waste, plant stem waste is highly abundant and present in both field and process residues. In traditional crop production, it is a frequent practice to discard the stems of many crops in the field, using them as low-value animal fodder and biomass. However, these plant stem wastes represent an unexplored source of dietary fibers with beneficial bioactive compounds. According to recent studies, banana pseudostem flour exhibits a remarkably high total dietary fiber (TDF) content, ranging from approximately 60% to 67% on a dry-weight (dw) basis.4 Similarly, sugarcane bagasse is composed largely of lignocellulosic fiber and reported to contain 79.5% (dw) TDF.5 Broccoli stalk fractions isolated as fibre-rich ingredients have produced TDF concentrates in the range of 61–69% (dw), demonstrating that cruciferous stems are also promising fibre sources.6 These quantitative examples show that stem residues are not marginal waste streams but concentrated repositories of functional dietary fibre. Building on this evidence, the present review adopts a stem-centric and functionality driven perspective, focusing exclusively on plant stem wastes as dietary fiber sources for food applications. Rather than merely cataloguing fiber-rich residues, this review integrates botanical origin, fiber chemistry, extraction feasibility, and techno-functional performance to critically evaluate the suitability of different stem types for value-added food use. By linking intrinsic structural features, such as cellulose crystallinity, hemicellulose content, and lignin distribution, to functional outcomes including water-holding capacity, oil-holding capacity, swelling behavior, and, this review enables differentiation between high-potential stem wastes and those more appropriate for non-food valorization pathways. Dietary fiber has great potential as a functional food ingredient due to its well-established physiological advantages as well as its techno-functional qualities, which include improving texture, water-binding, and stability. Short-chain fatty acids (SCFAs) like butyrate, propionate, and acetate are produced in the colon by fermentable fibers and support the integrity of the intestinal barrier, lower inflammation, and improve gastrointestinal motility.7 Researchers are looking for new uses for agri-food waste and converting it into value-added products. Studies on dietary fiber significantly add to the economic value of food waste, highlighting the significance of fiber in improving the health of the population. The potential reuse of agricultural waste improves the economic efficiency of agriculture. Importantly, high dietary fiber content alone does not necessarily translate into optimal nutritional and technological performance, as trade-offs often exist between fiber composition, extractability, fermentability, and functional quality. Additionally, the scalability and sustainability of extraction technologies remain key challenges in translating laboratory findings into industrial applications. Therefore, beyond summarizing existing literature, this review critically examines trade-offs, identifies the most promising stem waste streams for food use, and highlights current challenges and future opportunities in the food-grade valorization of plant stem fibers.2. Plant stem waste
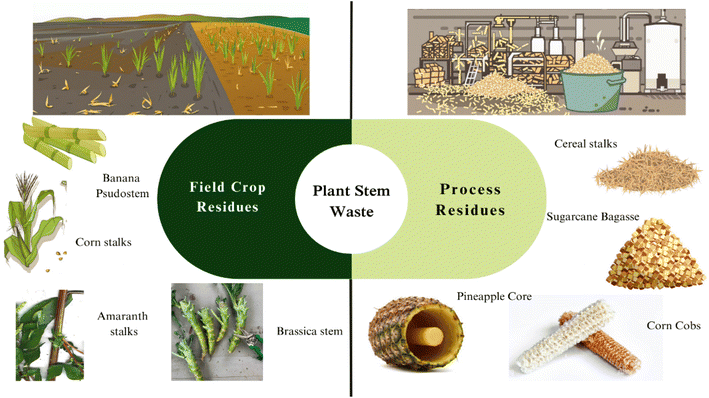
The plant stem is a crucial axial organ consisting of several specialized tissues arranged to provide structural support and functional competence. The epidermis is the outermost layer, a continuous barrier to protects the underlying tissues from desiccation and physical damage. It also contains stomata, which facilitate passageways for gas exchange and transpiration. Beneath the epidermis lies the cortex, made up of parenchyma cells that function in storage and repair, and collenchyma cells that offer flexible support at the new growth areas of elongating stems. The innermost region of the stem contains vascular tissue organized as bundles or continuous cylinders. The xylem transports the water and dissolved minerals taken up from the soil via the roots to the aerial part of the plant, and the phloem transports organic nutrients with a specific emphasis on photosynthates to all parts of the plant.8 In addition to serving as conduits for assimilated nutrients, mineral and water transportation stems also act as storage sites for carbohydrates, secondary metabolites and water. Furthermore, they provide mechanical support and optimize photosynthesis by allowing plants to elevate leaves toward sunlight.9Plant stem waste primarily occurs in agricultural and food processing industries as a byproduct of crop harvesting and food production. The main types of plant stem waste are depicted in Fig. 1. This waste is generated from various sources, including crop residues left in fields after harvest, trimmings from fruit and vegetable processing, and discarded stems from ornamental plant production. A recent study outlined that approximately 1.28 billion tonnes per year of maize stems are discarded globally.10 Similarly, global sugarcane production hovers around 1.87 billion tonnes annually, generating roughly 130 kg of dry bagasse per tonne of cane. That translates to about 243 million tonnes of bagasse per year.11 Furthermore, the preservation of food quality necessitates the intentional removal of plant stems and fibrous components in the food processing industry. Refined and processed foods often purposely remove fibrous components, leaving a mound of fibrous agricultural waste that typically includes stems.12 For instance, only a fraction of sugarcane stems is processed for juice extraction, while the remaining fibrous residue (bagasse) is often underutilized or burned, leading to environmental burdens. This waste stems not from the absence of nutritional value but from processing inefficiencies, market limitations, and the tough, lignified texture of many stems, which hinders direct consumption.13 However, these plant stem wastes represent a hidden source of dietary fiber and contain both soluble and insoluble dietary fibers, which can be effectively utilized as an excellent source of dietary fiber for novel food applications. This review aims to explore the often-overlooked components of agri-food waste, specifically focusing on the identification of potential hidden fibrous stems, stalks, cores, and cobs. By addressing this gap in research, we aimed to highlight the value of these materials and their potential applications in the food industry, contributing to a more sustainable approach in waste management and resource utilization.
3. Dietary fibers in plant stems/stalks
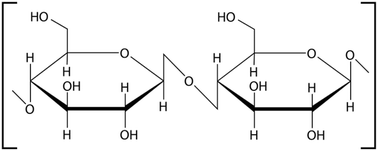
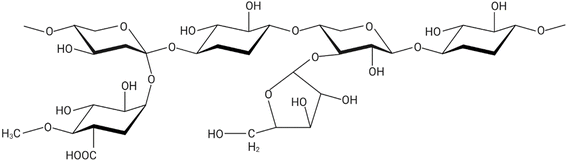
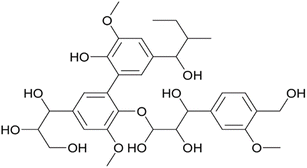
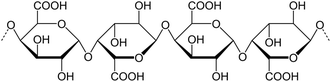
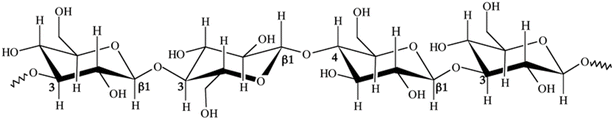
Dietary fiber (DF) extracted from plant stems and stalks primarily consists of complex polysaccharides that are resistant to digestion in the human gastrointestinal tract. Depending on the water solubility, dietary fibers are categorized as soluble dietary fiber (SDF) and insoluble dietary fiber (IDF).14 Soluble fiber types include pectin, beta-glucans, gums and mucilage, whereas insoluble dietary fiber types include cellulose, hemicellulose, and lignin. Plant stem waste is comprised of various fibers, each present in differing proportions, that play a significant role in modulating digestive health and influencing metabolic processes.15 Stems and stalks of agricultural plants, traditionally considered lignocellulosic waste, are now gaining attention as sustainable sources of dietary fiber due to their abundance and rich fiber profiles.163.1 Insoluble fibers
3.2 Soluble fibers
4. Potential sources of dietary fibers in agricultural food stem waste
Stems, stalks, cores and cobs are examples of axial plant residues, which are an unexplored class of lignocellulosic biomass produced by agricultural practices. Despite having a high dietary fiber content, especially insoluble components like cellulose and hemicellulose, these structures, which are oriented along the central axis of the plant, are frequently discarded after harvesting and processing. In the following context, some of the vital research findings relevant to dietary fiber from axial plant residues like stems, stalks, cores and cobs are comprehensively covered.4.1 Sugarcane stem bagasse (SCB)
Sugarcane bagasse is a by-product of the sugar manufacturing process, sugarcane treacle and sugarcane jaggery industries, which is frequently burned, causing substantial air pollution and posing serious environmental challenges. This fibrous bagasse contains a significant amount of dietary fiber, which can be effectively utilized to produce dietary fibre-enriched food products, nutraceuticals, and pharmaceuticals.13 A study reported that SCB contains 79.5% TDF (dw), consisting mainly of 75.7% (IDF) and 3.8% of SDF.39 According to Sangnark and Noomhorm,40 the chemical constituents of bagasse are variable with cellulose, hemicellulose and lignin fibers ranging from 55–58% (dw), 26–32%, and 19–22%, respectively. Similarly, another study found that the chemical composition of sugarcane bagasse included 37.2% (dw) cellulose, 22.1% hemicellulose, and 26.5% lignin.41 The peel of sugarcane is rich in cellulose, hemicellulose, and lignin. Removing the peel results in a significant loss of these fibers. Consequently, a study reported that sugarcane bagasse with the peel yields a higher amount than bagasse without the peel. The fiber content in sugarcane bagasse with the peel is 12.43% (dw), compared to 8.61% in bagasse without the peel. This demonstrates that the peel itself contains a substantial amount of dietary fiber. Thus, the removal of the peel diminishes the overall fiber content of the sugarcane bagasse.424.2 Brassica stem
Cruciferous crops, or Brassicas, are grown worldwide as temperate crops, including cabbage, cauliflower and broccoli. Brassica crop residues and byproducts have been produced in significant quantities along the supply chain as a result of the recent global expansion of cruciferous crop production. In 2022, the FAO reported that the global production of cruciferous crops, including cabbage, reached a total of 72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 604 kt.43 Broccoli production and consumption have increased in the last decade because of changes in population lifestyles, leading to a high adherence to healthy diets. The waste from the broccoli industry has become an environmental problem for the agri-food industries. In this regard, only 15% of the total plant biomass is constituted by the edible and a large quantity of by-products, especially the stalk, is generated after harvesting.6 However, the Brassica stems are rich in dietary fiber and other health-beneficial components, including dietary fiber, protein, vitamins, minerals, and significant bioactive compounds44
604 kt.43 Broccoli production and consumption have increased in the last decade because of changes in population lifestyles, leading to a high adherence to healthy diets. The waste from the broccoli industry has become an environmental problem for the agri-food industries. In this regard, only 15% of the total plant biomass is constituted by the edible and a large quantity of by-products, especially the stalk, is generated after harvesting.6 However, the Brassica stems are rich in dietary fiber and other health-beneficial components, including dietary fiber, protein, vitamins, minerals, and significant bioactive compounds44
According to a prior study, broccoli stems have about 38% (dw) total dietary fiber, of which 3.2% (dw) is soluble dietary fiber and 34.9% (dw) is insoluble.6 The same study found that broccoli stem fiber contains cellulose 18.5% (dw) and hemicellulose 24.1% (dw) of dry weight.6 Cauliflower stem dietary fiber is predominantly composed of insoluble dietary fiber (IDF), with a lesser proportion of soluble dietary fiber (SDF), reflecting its rich cell wall matrix. The IDF fraction is primarily composed of cellulose, hemicellulose, and lignin. Cellulose constitutes the structural backbone of the fiber, forming crystalline microfibrils that confer rigidity and mechanical strength. Hemicellulose, although present in lower amounts, contributes to the flexibility and water-binding properties of the fiber. Quantitatively, Eyupoglu et al.,45 reported that dried cauliflower stem contained approximately 36.5% (dw) TDF, of which IDF comprised 87.7% (dw) and SDF 12.3% (dw). The cellulose content was notably high at 58.5% (dw) of the fiber, followed by hemicellulose at 24.2% (dw) and lignin at 17.3% (dw). The cabbage stalk often discarded during minimal processing, is a rich source of dietary fiber and minerals, making it a promising ingredient for food valorization. According to Brito et al.,46 cabbage stalk flour obtained after drying and milling contained 42.67% (dw) of TDF. The fiber was predominantly insoluble, with IDF accounting for approximately 37.81% (dw) of the mass in a 100-gram sample, while SDF constituted about 4.86% (dw).
4.3 Corn cobs and stalk
Corn (Zea mays L.) ranks as one of the three major cereal crops globally, leading production with approximately 1162 million tonnes (Mt) of grain harvested in 2020.47 The cultivation of corn not only yields grain but also generates significant agricultural residues, such as corn stalks and cobs. For every 1 kg of dry corn grain harvested, around 0.15 kg of cobs is produced,48 resulting in an estimated 174.3 Mt of corn cobs in 2020 alone.49 Corncobs are exceptionally fibrous and contain high levels of total dietary fiber (TDF). Recent studies reported that dried cob meal contains approximately 89–93% (dw) of total dietary fiber.6 Compositionally, corncobs are rich in hemicellulose and cellulose, typically comprising 33–43% (dw) cellulose, 26–36% (dw) hemicellulose, and 17–21% (dw) lignin.50 For instance, one analysis indicated that corn cob fiber consists of 38.5% (dw) cellulose, 41.9% (dw) hemicellulose, and 6.7% (dw) lignin.51 Additionally, corncobs contain both free and bound phenolic compounds, as well as soluble dietary fiber and β-glucans.52 Ground corncobs exhibit a high total dietary fiber content, ranging from 89.9% (dw) to 93.2% (dw) on a dry weight basis, with soluble dietary fiber making up 0.8% (dw) to 2.2% (dw). Their composition also includes 43–46% (dw) of hemicellulose, 35–39% (dw) of cellulose and 3.2–6.4% (dw) of lignin.53 However, it is important to note that the nutritional composition of corn stover, which includes cobs and stalks, can vary significantly based on factors such as variety, environmental conditions, and processing methods.544.4 Sorghum stalk
Sorghum stalk residues, particularly from sweet sorghum and grain sorghum, are rich in lignocellulosic components and represent a significant source of dietary fiber. Qiu et al.55 investigated the dietary fiber content of different sorghum fractions, found that total dietary fiber (TDF) and insoluble dietary fiber (IDF) were highest in sorghum stalk bagasse, followed by whole stalk biomass, and lowest in bran. Their study also showed that the bagasse, being cellulose-rich, contributed predominantly to the IDF fraction, while branched hemicellulose was the primary contributor to soluble dietary fiber (SDF). Hu et al.56 reported that sweet sorghum bagasse—the fibrous material remaining after syrup extraction-contains approximately 37.7% (dw) cellulose, 28.1% (dw) hemicellulose, and 21.5% (dw) lignin. Grain sorghum straw has also shown comparable or higher cellulose content, as shown by Andrade Alves et al.,57 recording 49.4% (dw) cellulose, 19.2% (dw) hemicellulose, and 30.4% (dw) lignin in Brazilian sorghum straw. Saeed et al.58 found that Sudanese sorghum bagasse contained 43.0% (dw) cellulose, 21.1% (dw) hemicellulose, and 15.2% (dw) lignin. These values are generally consistent with previously reported ranges for sweet sorghum, which include 27–45% (dw) of cellulose, 25–27% (dw) of hemicellulose and 11–25% (dw) of lignin.594.5 Pineapple core/stem
Pineapple fruit yields two major fibrous by-products: the core (inner cylinder) and the stem (plant stalk left after harvest), and both are rich in plant cell-wall materials. Recent analyses show that the pineapple core is dominated by insoluble fibers with only a small soluble fraction.60 Shiau et al.61 measured the dietary fiber in pineapple cores and found a total dietary fiber (TDF) content of 53.6% (dw), consisting of 51.1% (dw) of insoluble dietary fiber (IDF) and 2.45% (dw) of soluble dietary fiber. The lignocellulose composition of pineapple core has been reported as cellulose 14.2% (dw), hemicellulose 36.06% (dw) and lignin 10.05% (dw).62 In contrast, the whole dehydrated core press-cake after juice extraction is less fibre-rich, and a study found that its TDF is 17.70%, with IDF 16.17% and SDF 1.53%.63 Pineapple stem, the lower stalk or trunk of the pineapple plant left after fruit harvest, is even more lignocellulosic than the core. It contains large amounts of structural polysaccharides, often coexisting with high starch content. Recent studies show that whole pineapple plant stems fiber contains 46.15% of hemicellulose, 31.86% cellulose and 18.6% lignin.64 After pineapple juice extraction, the remaining dried stem press-cake is also fiber-rich. Campos et al.65 reported the stem press-cake had 44% IDF, implying very high insoluble fiber content, and both core and stem were strongly insoluble as they contain significantly low SDF content.4.6 Water hyacinth stem
Water hyacinth (Eichhornia crassipes) stem represents an underutilized lignocellulosic biomass with significant potential as a novel dietary fiber ingredient. The cell wall composition of water hyacinth stems is primarily characterized by cellulose and hemicellulose, with a low lignin content.66 Enyi et al. (2020)67 demonstrated that the water hyacinth stem is the most fibrous part of the plant, and total dietary fiber content was around 58% by fresh weight (FW). Another study indicated that the dry biomass of water hyacinth stem consists of approximately 24.5% of cellulose and about 34.1% of hemicellulose.68 Another analysis found higher levels of cellulose, with around 67.5% of cellulose and 22.3% of hemicellulose in the isolated fiber fraction.69 In general, water hyacinth stems have a high cellulose and hemicellulose content with a lower amount of lignin content.70 Chonsakorn et al.71 also indicated that the stem fiber comprises approximately 72.17% cell wall material, with significant amounts of cellulose (50.38%) and hemicellulose (19.54%), while containing a relatively low lignin content (2.25%). Moreover, another study also demonstrated that hyacinth stem lignin content is around 4–8% of dry stem mass.72 This composition indicates a predominance of insoluble dietary fiber components, with cellulose contributing to structural integrity and bulking capacity, and hemicellulose providing both soluble and insoluble fractions that could influence water-holding and viscosity-related functional properties. The low lignin content suggests that water hyacinth stem fiber may be less rigid compared to lignin-rich sources.714.7 Banana pseudostem
Banana plants are tall herbs belonging to the Musa genus of the family Musaceae. This major commercial fruit crop is predominantly grown in subtropical and tropical regions. Bananas and plantains are the fourth-largest staple crop in the world and are essential to maintaining food and nutritional security for over 400 million people worldwide.73 Approximately 60% of the banana biomass is wasted after harvest and post-processing, accounting for 114.08 million metric tonnes globally.74 However, after harvesting, the resultant agri-biowaste is usually burned or released into the surroundings.75 For every ton of banana fruit harvested, approximately 3 tonnes of pseudostem are discarded.76 However, the discarded biomass is an abundant source of phosphorus, calcium, magnesium, and potassium. The effect and quantity of these phytocompounds found in the pseudostem vary according to age, variety, and genotype. Furthermore, it contains a variety of nutrients, including fiber, potassium, and vitamin B6, which can support detoxification and body weight management.77 Multiple studies have demonstrated that the banana pseudo-stems contain substantial amounts of both insoluble and soluble dietary fiber, along with associated bioactive components. Bhaskar et al.78 reported that the pseudostem of Musa sp. var. elakki bale contains approximately 28.8% total dietary fiber (TDF), with 27.4% being insoluble dietary fiber (IDF) and 1.4% soluble dietary fiber (SDF). Subagyo et al.76 found that pseudostem sheaths contain 60–85% lignocellulosic material, with cellulose constituting around 50% of dry weight, accompanied by substantial hemicellulose, lignin, pectin, and ash. In a subsequent study on Musa sp. cv. Nanjangud Rasa Bale, Ramu et al.79 reported a total dietary fiber (TDF) content of 61%, predominantly consisting of insoluble dietary fiber (IDF), with cellulose levels surpassing those of hemicellulose and lignin.4.8 Amaranth stem
Amaranth (Amaranthus spp.) is a fast-growing, drought-resistant plant widely cultivated across the world for its edible leaves and protein-rich grains. While most attention has historically focused on amaranth seeds and leaves due to their high nutritional value, the plant generates a substantial volume of stem biomass that is often discarded as agricultural waste. The amaranth stem, which comprises both fibrous outer tissues and a softer inner core, has largely been underutilized despite its significant structural and biochemical complexity. Recent studies reported that amaranth stalks are highly lignocellulosic and rich in dietary fiber, predominantly in the insoluble fraction. For example, Emam et al. analyzed red amaranth (Amaranthus cruentus) stems and found that the outer bark contains 43.6% cellulose, 17.6% hemicellulose and 20.3% lignin (dry weight), while the central pith contains 37.5% cellulose, 12.1% hemicellulose and 9.4% lignin.80 Likewise, Bamisaye et al.81 reported green amaranth (Amaranthus hybridus) stem waste with roughly 11.1% cellulose, 4.2% hemicellulose and 12.3% lignin. These figures indicate that amaranth stems can contain a very high proportion of insoluble dietary fiber. In 2020, a detailed survey of 17 genotypes of stem amaranth (Amaranthus lividus) reported mean dietary fiber values around 78.9 µg per g fresh weight (FW) with significant genotype variation ranging from 62 to 96 µg per g FW.82 Meanwhile, an investigation of Amaranthus hypochondriacus genotypes found dietary fiber in the range of 7.3 to 10.3 g/100 g FW across eleven genotypes, averaging around 8.7 g/100 g FW.83 In animal-feed studies, diets incorporating stem and leaf powder of A. hypochondriacus were reported to contain approximately 6.2% to 9.6% total dietary fiber, reinforcing that stem waste contributes materially to fiber content.84 Taken together, these findings suggest that amaranth stalk residues – often an agricultural waste could be valorized as a novel fiber-rich ingredient. The dominant cellulose/hemicellulose content should confer classic fiber benefits, including gut motility, controlling glycemic index and cholesterol reduction, making stem-derived fiber potentially useful in food formulations or nutraceuticals. Comparative ranking of plant stem waste for food applicability, extractability and fermentability is shown in Table 1.| Food applicability | Plant stem waste source | TDF range (% dry weight) | General extractability | Dominant extractable fiber fractions | Fermentability potential | Key reference |
|---|---|---|---|---|---|---|
| Low–moderate (applicable with proper delignification and IDF to SDF conversion) | Corn stalk | 89–93% | Low to moderate | IDF-dominant limited SDF | Low fermentability | 50, 51 and 53 |
| Requires strong alkaline and physicochemical pretreatment to disrupt lignin–carbohydrate complexes | High in lignin, cellulose and hemicellulose (glucurono-arabinoxylans) | High cellulose crystallinity and lignin limit microbial accessibility | ||||
| Sugarcane stem bagasse | 75–80% | Low | Strongly IDF dominant and negligible native SDF | Low fermentability due to high lignin and low soluble polysaccharides | 40 and 41 | |
| Highly lignified matrix, extractability improves only after delignification | High in lignin, cellulose, xylans | |||||
| Sorghum stalk | 35–50% | Low extractability | IDF dominant | Low fermentability | 56–58 | |
| High lignin content and phenolic cross-linking restrict extraction | High proportion of lignin | High lignin and phenolic cross-linking restrict extraction | ||||
| High | Banana pseudostem | 60–67% | Moderate to high | IDF-dominant with moderate SDF fractions | Moderate fermentability | 76, 78 and 79 |
| Cell walls are less rigid, alkaline, ultrasound and enzyme assistance is effective to extract | Cellulose, hemicellulose, pectic polysaccharides | The presence of hemicellulose and pectin supports partial fermentation | ||||
| Brassica stem | 36–45% | Moderate extractability | Balanced IDF/SDF | Moderate fermentability | 14, 44 and 53 | |
| Lower lignification than cereal stalks, pectin extractable | Cellulose, hemicellulose, pectin | Lower lignification than cereal stalks, pectin extractable | ||||
| Pineapple core/stem | 45–54% | High extractability | SDF enriched | High fermentability | 61–63 | |
| Relatively loose matrix, pectin readily extractable | High pectin content | Relatively loose matrix, pectin readily extractable | ||||
| Water hyacinth stem | 50–58% | High extractability | Mixed IDF/SDF | Moderate to high fermentability | 67–69 | |
| Low lignin content, easily disrupted by mild chemical and hydrothermal treatments | Cellulose, hemicellulose, pectin | Pectin and amorphous hemicellulose enhance fermentability |
As shown in Table 1, stems with high total dietary fiber (TDF) content and high lignification, such as sugarcane bagasse, corn cobs, and sorghum stalks, have low extractability and fermentability, making them less suitable for food applications. In contrast, stems with moderate TDF content and lower lignification, such as banana pseudostems, pineapple stems/cores, and water hyacinth stems, improve their extractability and hydration properties. This makes them more suitable for producing food-grade dietary fiber. Among these options, banana pseudostem stands out as particularly promising due to its abundance, minimal processing requirements, and versatility in food formulations.
5. Plant stem waste valorization
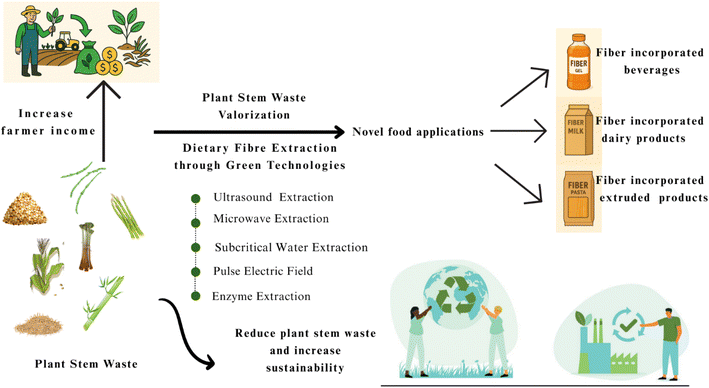
Valorization refers to the process of converting waste into value-added products, closing the loop of resource use in agriculture. The importance of utilizing discarded plant stems is progressively acknowledged as a pivotal part of sustainable farming and circular economic visions. This waste valorization approach aims to reduce waste while enhancing resource productivity, thereby contributing to ecological sustainability and economic resilience.85 As previously mentioned, agricultural food waste remains a significant issue for the food and agriculture sectors. To solve this issue, a viable and sustainable valorization program must be established. Plant stems, stalks, cores, and cobs comprise a significant portion of the various types of agricultural waste.86 These byproducts present excellent opportunities for creative recycling and repurposing initiatives due to their abundance and high dietary fibre content.87 A study indicated that sugarcane bagasse has been used for dietary fiber extraction as a sustainable valorization approach and developed fiber-fortified bakery products to improve texture and nutritional value.88,89 Moreover, a research study on the extraction of dietary fibre from banana pseudostem has demonstrated excellent functional and nutritional properties, including water retention and prebiotic potential, making it suitable for use in fibre-fortified food formulations.73 Also, broccoli stems, which are often thrown away, are full of dietary fiber and glucosinolates, which are good for health as they fight cancer and free radicals.6 The potential for recycling plant stem residues can help farmers diversify income streams, reduce on-farm waste, and improve overall resource efficiency. This also helps the environment by reducing agri-waste and making food systems more resilient.89 A variety of valorization methods have been established to extract dietary fibers from different plant sources, as documented in the literature. However, not all plants are suitable for these extraction techniques. The methods are tailored to accommodate the specific fibrous structures present in the cobs, cores, stalks, and stems of each plant.90 By considering the unique structural properties of the source material, each method is thoughtfully designed to enhance both the yield and quality of dietary fibers.91 Liquid-to-solid ratio, contact time, and temperature can influence the yield of dietary fiber.92 Several studies have argued the effect of treatment methods on the structure of dietary fibers in various ways. For example, alkali and acid-mediated extraction can damage the molecular structure of dietary fiber, while enzymatic extraction techniques may result in incomplete extraction.93 Alternatively, combined extraction methods that utilize both enzymatic and solvent techniques can be effective for extracting dietary fiber. Apart from that, the modified wet-milling method has been recommended for improving extractability.94 This method is cost-effective, produces high-purity fiber, and requires fewer chemicals and less water than other commonly used methods. According to findings, the wet-milling technique yields dietary fiber with a purity of 50% to 90%.95 On the other hand, pulsed electric fields, ultrasound, microwaves, high hydrostatic pressure, and ionizing radiation are examples of novel green extraction technologies that each have unique benefits and drawbacks. These innovative, sustainable valorization methods enable high-quality extraction that is repeatable, manageable, and less harmful to the environment.96 For instance, Sun et al.97 extracted insoluble dietary fiber from soybean residues using a combination technique of ultrasonic-assisted alkali extraction, yielding 744 mg of insoluble fiber per gram of raw soybean. Recently, Wen et al.98 explored the effects of ultrasonic-microwave-assisted extraction on soluble dietary fiber from coffee waste and compared this method to traditional solvent extraction techniques and similarly reported that ultrasonic-microwave-assisted extraction produced more soluble dietary fiber yield. Begum and Deka99 effectively extracted dietary fiber from culinary banana bracts using ultrasonic-assisted extraction. The yields for soluble, insoluble, and total dietary fibers were reported at 4.65%, 78.7% g, and 83.9% respectively. In a separate study, the pulsed electric field technique was utilized for the extraction of cellulose from the fibers of the Mendong plant. The cellulose extracted using this method exhibited a superior crystalline index relative to that obtained through alkali extraction.100 This technique enhanced the crystallinity of the extracted cellulose by 83–86%. Food-processing industries worldwide are actively seeking new ways to source dietary fiber from lesser-known plant resources, aiming to incorporate it as a value-added healthy ingredient.93 Despite the recognition of plant stem waste as a valuable resource, there has been a notable lack of comprehensive research focused on the various dietary fiber extraction techniques that could be utilized for its valorization. This research is crucial as it offers a sustainable alternative to the common practice of incinerating plant stems in agricultural fields, which poses environmental concerns. Exploring innovative methods for extracting dietary fiber not only has the potential to reduce waste and promote sustainability but also opens up avenues for developing new, healthy food products and industrial applications. By delving deeper into these techniques, we can better harness the nutritional and functional benefits of plant fibers, highlighting their significance in both food science and waste management.101 A summary of plant stem waste valorization is illustrated in Fig. 7.6. Potential green extraction techniques for plant stem dietary fiber extraction
The utilization of green extraction techniques for isolating dietary fiber from plant stems offers significant advantages over conventional methods in terms of environmental sustainability, efficiency, and product quality. Traditional extraction methods, such as acid or alkaline hydrolysis and solvent-based techniques, often involve the use of hazardous chemicals, extended processing times, and high energy consumption, which may lead to the degradation of bioactive components and diminished functional properties of the extracted fiber.102 In contrast, green extraction technologies, including ultrasound-assisted extraction (UAE), microwave-assisted extraction (MAE), enzyme-assisted extraction (EAE), and subcritical water extraction, enable higher extraction yields with reduced solvent usage, shorter processing times, and lower energy requirements. These methods effectively preserve the physicochemical and functional properties of dietary fibers, such as water-holding capacity, oil-binding capacity, and fermentability. They accomplish this by effectively removing lignin from highly lignified plant stems and facilitating the conversion of insoluble dietary fibers (IDF) to soluble dietary fibers (SDF). Such properties are critical for their application as functional food ingredients.103 Furthermore, green extraction technologies contribute to circular economy principles by enabling the valorization of underutilized agricultural residues like plant stems, thereby reducing agro-industrial waste and environmental burden. Given the growing consumer demand for clean-label and eco-friendly products, integrating green extraction approaches is essential for producing high-quality, sustainable dietary fiber ingredients from plant stem biomass.104 Potential novel green technologies for plant stem dietary fiber extraction are shown in Table 2.| Green extraction technology | Plant source | Key observations | References |
|---|---|---|---|
| Ultrasound extraction | Garlic stem residue | The IDF became more porous, while the WHC increased from 7.38 to 9.72 g g−1, resulting in improved texture and functionality | 123 |
| Ultrasound-assisted alkaline extraction | Rice straw | The IDF yield increased from 29% to approximately 37%, while ultrasound enhanced both porosity and water affinity | 124 |
| Banana pseudostem | Achieved approximately 99.5% pure cellulose yield, consisting of insoluble fiber. The extracted fiber exhibited increased crystallinity of about 68% and enhanced thermal stability | 109 | |
| Ethanol/hot water sequential extraction | Broccoli stalk | TDF yield – 68.9%, IDF yield around 54%, and SDF is approximately 14.7%. WHC varies from 3.9 to 8.2 g g−1, while OHC is approximately 4 g g−1; these properties show excellent swelling and emulsification capabilities | 6 |
| Microwave-assisted alkaline extraction | Sugarcane bagasse | Hemicellulose yield increased from 62.59% to 82.75%. Enhanced porosity and surface area were confirmed by SEM and BET analysis, with reduced NaOH usage. High molecular weight hemicellulose was obtained | 125 |
| Microwave-assisted alkaline hydrolysis | Sugarcane bagasse/tops | Microwaving at 600 W for 5 minutes during NaOH treatment significantly enhanced lignin removal compared to conventional alkaline extraction | 126 |
| Enzymatic extraction (EAE) | Corn cob | An increase in soluble dietary fiber (SDF) from insoluble dietary fiber (IDF) also resulted in enhanced total phenolic content and improved antioxidant activity | 127 |
| Alkaline pretreatment and enzymatic hydrolysis | Chinese cabbage waste | Alcohol-insoluble fiber (AIF) from enzymatically hydrolyzed Chinese cabbage waste significantly reduced glucose diffusion and bile acid binding compared to commercial fibers, highlighting its potential as a sustainable functional dietary fiber with hypoglycemic and hypolipidemic benefits | 128 |
| Subcritical water extraction (SWE) | Rice straw | Cellulose content: 86.2%; water index: 80.1; yield: 46.2%; crystallinity: 67%; improved thermostability. Supercritical water extraction (SWE) at 180 °C produced purer, whiter, and more crystalline cellulose compared to 160 °C, making it a green alternative for high-quality fiber extraction | 129 |
| Subcritical (hot-compressed) water extraction | Defatted corn hull | At a temperature of 150 °C for 60 minutes, the yield of the dry fraction (DF) is 33.0%, which includes both insoluble and soluble components. Increasing the temperature to 180 °C increases the yield to approximately 56.9%. This observation indicates that elevated temperatures correlate positively with DF yield | 130 |
| Pulsed electric field (PEF) assisted alkali extraction | Banana pseudostem | The TDF is approximately 62.5%, showing moderate enrichment of SDF. Additionally, PEF enhances swelling capacity and improves the retention of bioactive compounds | 131 |
6.1 Subcritical water extraction (SWE)
Subcritical water extraction (SWE) uses hot pressurized water (100–374 °C, under enough pressure to keep it liquid) to hydrolyze plant material. At high temperatures, the dielectric constant of water decreases, and it acts like a mild solvent for polysaccharides.105 SWE can break down cell walls and solubilize hemicellulose and pectin without added chemicals. Notably, SWE generates no toxic solvent waste and has fast heat transfer. Yan et al.105 examined how subcritical water extraction methods influence the physicochemical and functional properties of soluble dietary fibers extracted from wheat bran. In this study, subcritical water extraction (SWE), SWE in aqueous citric acid (pH 5.0) (SWEC), and ultrasound-assisted SWEC (USWEC) were used to extract soluble dietary fiber (SDF) from wheat bran. Results revealed that SWE-based methods significantly influenced the physicochemical, functional, and biological properties of the SDF. The USWEC treatment demonstrated a superior soluble dietary fiber (SDF) yield of 46.3%, accompanied by a high carbohydrate content of 82.9%. Additionally, this treatment results in a reduced weight-average molecular weight of 65.2 kDa and a particle size of 1.17 µm, which contribute to a looser and more porous surface structure. In other work, subcritical extraction of citrus peels successfully isolated pectin, cellulose and hemicellulose from Citrus junos residue. Modifying SWE by adding a weak acid and ultrasound can further increase soluble fiber yield and antioxidant activity.106 Important advantages of using this type of extraction are high extraction efficiency, short processing time, and positive environmental impact, without the residual organic solvents. The high temperatures and pressures required for the process necessitate the use of specialized equipment, specifically high-pressure reactors, which can incur high costs. Furthermore, thermal treatment may cause the degradation of certain fibers or provoke Maillard reactions in the presence of sugars. Additionally, the harsh conditions associated with subcritical water extraction (SWE) can compromise pectin quality by reducing its methoxylation levels if not carefully regulated.Subcritical water extraction (SWE) for fiber extraction generally operates at technology readiness levels (TRL) 4–6 and is not yet commonplace in industrial applications. The technology necessitates high-pressure vessels, heat exchangers, and pumps, which are both expensive and complex. Additionally, operating costs remain high due to significant energy requirements needed to heat water to near-critical temperatures, while the process throughput is constrained by heat transfer rates. Although water is the only solvent used, eliminating costs associated with organic solvents, the overall energy costs are substantial, leading to high energy consumption in SWE.107
6.2 Ultrasound-assisted extraction (UAE)
Ultrasound employs high-frequency sound waves (usually 20–40 kHz) that propagate through the solvent, causing rapid compression/expansion cycles and acoustic cavitation. Cavitation bubbles form and collapse violently, generating microjets that shear cell walls and improve mass transfer. This mechanical effect disrupts plant matrices, releasing intracellular contents. UAE can thus dramatically shorten the extraction time and reduce solvent needs. Moreover, ultrasound treatment can convert insoluble fiber into more soluble forms by breaking down cellulose/hemicellulose complexes. A recent study demonstrated that combining ultrasound with subcritical water on wheat bran produced an SDF fraction with a small particle size (1.17 µm) and a high yield (46.3%).105 Ultrasound is also widely used as a pretreatment for agri food waste before chemical or enzymatic extraction, leading to higher fiber release. For instance, Rawat & Ghosh108 optimized UAE of dietary fiber from bhimkol (Musa balbisiana) peel, an underutilized banana cultivar waste. Using a central composite design, they varied ultrasound time (20–60 min), solvent-to-solid ratio (30–70 mL g−1) and temperature (40–80 °C). The highest total dietary fiber yield reached 49.58 ± 0.88% under optimal conditions 60 min, 30 mL g−1, 40 °C. The extracted fiber was characterized for physicochemical, functional, and thermal properties, showing enhanced water- and oil-holding capacity, glucose absorption, and thermal stability compared to control fiber samples. A recent study on cellulose extraction from banana pseudostem waste demonstrated the efficacy of ultrasound-assisted extraction (UAE) on plant-stem waste. Utilizing a focused ultrasound probe at 200 W and 26 kHz, combined with maintaining a temperature of 40 °C, an alkaline treatment with sodium hydroxide (NaOH) achieved up to 99.5% cellulose, featuring a high crystallinity of 67.9%. This process resulted in improved thermal stability, exceeding that of commercial pine and eucalyptus fibers. Scanning electron microscopy (SEM) and thermogravimetric analysis (TGA) data corroborated the significant role of ultrasound in enhancing both fiber morphology and purity during recovery.109 A study on pulse hulls (lentil, pea) reported that ultrasound treatment led to a 6% increase in soluble dietary fiber yield compared to acid hydrolysis alone. However, the insoluble dietary fiber fraction was significantly reduced, shifting the total fiber composition toward more soluble forms. This effect is attributed to enhanced solvent penetration and cell disruption from cavitation, facilitating partial release or solubilization of the polysaccharide component.110UAE has advantages over conventional heating methods, including lower energy consumption, shorter operating time, reduced solvent volumes, and increased yields.109 Cavitation effects can be uneven in large batches, making scale-up challenging. Power ultrasound can also generate radicals that slightly degrade some compounds. Energy input per unit volume can be high if processing thick slurries. In practice, the UAE is best for slurries or liquids like juices and suspensions rather than dry fibers.111
In practice, ultrasound-assisted extraction (UAE) for stem biomass is currently at a pilot scale, with a technology readiness level (TRL) of approximately 4 to 6. Capital costs are moderate while operating costs remain low, as the process requires only electricity and minimal amounts of solvent or water. UAE typically involves the use of a wet slurry of biomass. The energy consumption is relatively low due to the efficient cavitation process; in fact, sonication is recognized for its lower energy requirements when compared to heating methods. UAE has advantages over conventional heating methods, including lower energy consumption, shorter operating time, reduced solvent volumes, and increased yields.109
6.3 Microwave-assisted extraction (MAE)
Microwave frequencies range from 300 MHz to 300 GHz and belong to non-ionizing radiation.112 In the food matrix, microwaves produce heat due to their interactions with the cellular compounds of a polar nature. The produced heat causes the ionic conduction and rotation of dipoles.109,112 Gan et al.113 evaluated the structural and functional modifications of grapefruit peel SDF extracted using MAE alone and in combination with sodium hydroxide, enzymatic hydrolysis, and ultrasound. Pure microwave treatment (PMT) increased SDF yield from 3.62% to 7.94%. While modified MAE treatments yielded even more. SDF yield 17.19% with microwave-sodium hydroxide treatment, 9.13% with microwave enzymatic hydrolysis treatment, and 8.35% with microwave-ultrasound treatment. MAE helped convert insoluble fiber (IDF) to SDF, likely by rupturing the cell wall due to intracellular steam pressure and rapid heating. Importantly, all MAE-modified fibers exhibited enhanced molecular weight, surface porosity, and crystallinity. Among them microwave-ultrasonic treated SDF demonstrated the most pronounced improvements in functional properties, including the highest water-holding capacity (11.39 g g−1), oil-holding capacity (25.01 g g−1), cholesterol adsorption capacity (26.48 mg g−1), glucose adsorption capacity (24.42 mg g−1), and nitrite ion adsorption capacity (219.43 µg g−1 at pH 2). Scanning electron microscopy (SEM) revealed that microwave-treated samples had more porous, looser, and multi-layered structures. Additionally, microwave-ultrasonic-treated SDF exhibited superior thermal stability and a broader monosaccharide profile compared to other treatments and the highest average molecular weight (614.1 kDa), indicating more effective solubilization of polysaccharides. XRD and FT-IR analyses showed that combined microwave treatments increased crystallinity and altered glycosidic bonds. The advantages of microwave-assisted extraction over conventional methods are the reduced use of applied solvents, increased yields, shorter process time, and a reduction in energy consumption.109 Microwave heating is characterized by the uneven distribution of thermal energy, which can result in the formation of hot spots. These hot spots may lead to localized overheating or the caramelization of sugars. Furthermore, non-polar solvent systems lack microwave activity, resulting in poor heating efficiency under microwave irradiation.Microwave-assisted extraction demonstrates a TRL of approximately 5–7 for dietary fiber recovery, with most applications remaining at laboratory or pilot scale. The technology requires specialized microwave reactors, resulting in moderate capital costs, while operating expenses are dominated by electricity usage. MAE offers relatively low overall energy consumption due to rapid volumetric heating and shortened processing times; however, energy efficiency can decline during scale-up because of non-uniform heating. MAE is most effective for wet or moisture-rich biomass, as water enhances microwave absorption, whereas dry biomass often requires added moisture to achieve efficient heating.114
6.4 Enzyme-assisted extraction (EAE)
Enzyme-assisted extraction methods utilize lower extraction temperatures and higher pH values and are therefore considered a greener alternative to conventional methods. There are two explanations for the enzyme-assisted extraction of polysaccharides: (i) the enzymes disrupt the cell walls and membranes, and desirable compounds are thus liberated, and (ii) enzymes partially degrade polysaccharides.115 Enzymes which are commonly used for the extraction of soluble dietary fibers are cellulase, hemicellulose, protease, xylanase, pectinase, and glucanase. Since combinations of enzymes may achieve higher yields, blends of different enzymes, customized for particular cell wall type disruptions, can be bought.116For example, combined use of cellulase with xylanase and pectinase enhances disruption of cellulose, hemicellulose pectin networks, leading to greater release of soluble dietary fiber and improved modification of insoluble fractions, as demonstrated in rice stalks,117 potato residue,118 cocoa bean shells,119 and sorghum stalks.120 Even though EAE show promising improvements in fibre quality and functional properties, quantitative dietary fibre yield is variable due to partial degradation of structural polysaccharides, processing conditions and substrate variability.
Often, enzymatic extraction is coupled with mild heating or assisted by ultrasound or microwave techniques to enhance efficiency. For instance, Buljeta et al.15 used cellulase together with microwave heating on apple pomace to obtain soluble fiber. The net effect of EAE is usually to increase the yield of soluble dietary fiber and improve its hydration properties, because partial hydrolysis produces lower-MW fibers.
EAE is characterized by its gentle and highly specific nature, allowing for the enhancement of both yield and purity of desired fiber fractions under mild conditions. Furthermore, EAE is considered environmentally friendly, as it utilizes biodegradable reagents.
EAE for dietary fiber has reached an early stage of industrial readiness, typically situated at TRL 4 to TRL 6, as it moves from pilot-scale validation toward full commercial integration. From an economic perspective, EAE is increasingly feasible when integrated into a circular bioeconomy model where it valorizes agricultural by-products, such as plant stem waste, into high-value functional fiber fractions. While the high cost of commercial enzymes remains a primary economic hurdle compared to conventional methods like acid and alkaline-assisted extractions, this is often offset by the superior quality, purity, and functional properties of the extracted fiber, which leads to a premium market price. Energy considerations further support EAE adoption, as it operates under mild, non-thermal conditions like 40 to 60 °C. This significantly reduces the total energy footprint compared to high-temperature or high-pressure conventional extractions, which often require intensive heating. Moreover, as green extraction technology EAE eliminates toxic chemical solvents, supporting global decarbonization goals.115
6.5 Pulsed electric field assisted-extraction (PEF)
Pulsed electric fields use short bursts of high-voltage pulses (micro- to milliseconds) to induce electroporation in plant cells. The electric pulses create temporary pores in cell membranes, greatly enhancing solvent penetration and release of intracellular compounds.15 Because PEF is non-thermal and rapid, it can improve the extraction of polysaccharides without significant heating. For instance, Fan et al.121 applied pulsed electric field (PEF) treatment to orange peel and found that the yield of soluble dietary fiber (SDF) more than doubled compared to a control sample. The maximum SDF content was 0.238 g per gram of dry peel with PEF treatment, while it was approximately 0.11 g g−1 without PEF. Moreover, this study indicated that the optimal parameters of PEF assisted extraction SDF was temperature of 45 °C with the electric field intensity of 6.0 kV cm−1, pulses number of 30, and time of 20 min and SDF treated with PEF showed the higher water solubility, water-holding and oil-holding capacity, swelling capacity, emulsifying activity, emulsion stability, foam stability and higher binding capacity for Pb2+, As3+, Cu2+, and resulted from the higher viscosity due to PEF treatment. Compared with the untreated orange peel, the SDF obtained with PEF exhibited stronger antioxidant activities, which was due to its smaller molecular weight (189 vs. 512 kDa). In addition, scanning electron micrograph images demonstrated that the surface of PEF-SDF was rough and collapsed.121 In another example, wheat bran exposed to an induced electric field (500 V, 40 min) increased its soluble fiber content from 7.69% to 12.02%. PEF is a non-thermal process that preserves heat-sensitive fibers. It is highly efficient, significantly increasing yield, and can be applied at moderate temperatures. Additionally, it enhances diffusion and extraction kinetics through cell disintegration.115,116 Pulsed electric field technology exhibits a TRL of 5–6 for fiber extraction. High-voltage equipment is quite expensive and demands substantial energy consumption when operated at a large scale. Moreover, excessive or prolonged pulsing can lead to the degradation of certain polysaccharides, making it crucial to optimize the operating conditions. In practice, pulsed electric fields (PEF) are most effective with liquid or paste matrices. Consequently, pre-processing solid materials like dry stalk fibers may require hydration.1226.6 Technical and economic comparison of potential green extraction technologies for dietary fiber extraction
Across these green extraction technologies, capital cost, operational expenditure, energy demand, scalability, and technology readiness levels (TRLs) vary considerably when specifically assessed for dietary fiber extraction from plant stem biomass. Capital investment is generally lowest for microwave-assisted extraction (MAE),132 which relies on relatively simple microwave generators and pressure-resistant vessels, and moderate for ultrasound-assisted extraction (UAE),133 as industrial ultrasonic reactors are commercially available and comparatively affordable. Consequently, both MAE and UAE have reached pilot-scale validation for dietary fiber extraction (TRL 5–6),134 with several studies demonstrating reproducible yields and functional improvements at kilogram-scale processing. Enzyme-assisted extraction (EAE) requires only conventional stirred tanks and mild processing conditions, resulting in low capital expenditure.135 However, its operational costs are high, primarily due to enzyme procurement, limited reusability, and longer processing times. For plant stem dietary fiber extraction, EAE remains largely confined to laboratory and early pilot demonstrations (TRL 4–5), as industrial-scale deployment is constrained by high enzyme cost and batch variability.In contrast, pulsed electric field (PEF) and subcritical water extraction (SWE) exhibit the highest capital requirements, owing to the need for high-voltage pulse generators and high-pressure, high-temperature reactors, respectively. From a readiness standpoint, PEF is the most industrially mature technology, with established continuous-flow systems already commercialized in the food industry. When adapted as a pretreatment or intensification step for dietary fiber extraction from stems, PEF can be reasonably assigned a TRL of 7–8, reflecting near-industrial readiness. Conversely, SWE remains at pilot scale (TRL 4–5) for dietary fiber extraction, as its high energy demand, pressure constraints, and risk of polysaccharide degradation have so far limited large-scale food-grade implementation.136
With respect to operational costs, UAE, MAE, and PEF are economically favorable, as they primarily consume electricity and water and avoid expensive chemical reagents. These methods also offer cost-saving advantages through reduced extraction time and lower solvent consumption, improving process throughput. PEF, in particular, has demonstrated lower net energy consumption compared with conventional thermal treatments, due to its non-thermal, short-pulse operation. In contrast, SWE incurs high operating costs because of continuous heating to subcritical temperatures, while EAE remains cost-intensive due to enzyme usage despite its low energy input.133
From an energy-demand perspective, UAE, MAE, and PEF can be classified as low-energy technologies, as they rely on efficient energy transfer mechanisms like acoustic cavitation, volumetric microwave heating, and short electrical pulses. EAE exhibits moderate energy demand, mainly for controlled heating and agitation, whereas SWE is clearly energy-intensive, requiring sustained thermal input at elevated pressures. When evaluating TRL alongside economic and energy criteria, PEF currently demonstrates the highest level of industrial readiness. In contrast, UAE and MAE emerge as the most balanced options for near-term scale-up, owing to their moderate costs, low energy requirements, and TRL 5–6 maturity. While EAE and SWE are environmentally appealing, they are not yet as industrially prepared for large-scale dietary fiber extraction from plant stems and need further techno-economic optimization before they can be commercialized.137
7. Functional properties of plant stem dietary fiber
Plant stems and cores are often discarded during harvesting and food processing and are emerging as valuable sources of dietary fiber with promising functional and bioactive compounds. Dietary fibers isolated from plant stems generally exhibit high water-holding capacity (WHC), reflecting their utility in increasing the moisture and bulk of food matrices. WHC arises from adsorption of water by hydrophilic polysaccharide chains and the entrapment of water within porous fiber networks. For instance, a study indicate that sugarcane bagasse fiber retains about 1.87 g water per g fiber.138 Banana pseudostem fibers typically exhibit WHC values in the range of 3.9–7.2 g g−1, attributed to their porous structure and high accessible polysaccharide content.139 Isolated broccoli stalk fiber fractions have moderate WHC (3.9–8.2 g g−1), consistent with balanced cellulose and pectic compositions that support water adsorption and swelling.6 In contrast, cellulose-rich fractions from sorghum stalks demonstrate exceptionally high WHC (22.8–35.3 g g−1), reflecting extensive fibrillation and high surface area after processing.108 These differences arise from variations in cellulose and hemicellulose, lignin, and pectin contents that determine the number of hydrophilic sites and network porosity.However, research studies show that fiber different extraction methods have the potential to increase the water-holding capacity. For example, alkaline-treated sugarcane bagasse fiber retained about 3.02 g water per g fiber compared to 1.87 g g−1 in untreated bagasse,140 indicating that fiber treatments can nearly double WHC. Still, plant stem fibers typically hold several times their weight in water, contributing to texture and satiety benefits in food applications.
Oil-holding capacity (OHC) measures the ability to absorb lipids by fiber, and it varies with fiber surface's hydrophobicity and porosity. A study on the isolated fiber fraction of banana pseudostems revealed a high oil holding capacity of approximately 9.3 g of oil per gram of fiber.141 However, the fiber extraction method also affects the oil holding capacity, and there is a significant difference between raw and processed or treated fiber fractions. For instance, alkaline-treated sugarcane bagasse fiber showed moderate OHC. In one study, raw sugarcane fiber bound 4.83 mL oil per g, while the alkaline-treated form bound 2.67 mL g−1.140 Moreover, according to Zhou et al.,142 fermentation of corn cob dietary fiber using Aspergillus niger significantly enhances its OHC. Specifically, the OHC of unfermented corn cob soluble dietary fiber was reported as 5.10 g g−1, while after fermentation, the OHC increased markedly to 6.97 g g−1. In general, fibers with greater surface area and hydrophobic sites absorb more oil. High OHC is technologically useful for bakery products because it can improve mouthfeel and act as a fat replacer.143
Glucose adsorption capacity (GAC) is a functional property of dietary fiber that describes its ability to bind free glucose molecules from a surrounding solution. It is typically measured as the amount of glucose adsorbed per gram of fiber under controlled conditions, often mimicking the human gastrointestinal environment (e.g., pH 6–7, 37 °C).144 However, glucose adsorption capacity values depend on fiber structure. Many insoluble fibers bind only small amounts, whereas some highly porous fibers bind better. For example, a study of banana pseudostem dietary fiber reported a GAC of 82.75 mg glucose per g fiber, significantly higher than that of purified cellulose, which had 64.21 mg g−1 of GAC. This indicates that banana stem fiber can adsorb free glucose effectively. Additionally, extraction techniques influence the glucose adsorption capacity (GAC), promoting a more porous structure. A study on the fermentation of corn cob soluble dietary fiber (SDF) by A. niger demonstrated a significant enhancement in GAC, achieving a value of 2635.57 µmol g−1, which represents an increase of 23.78% compared to the unfermented corn cob SDF, which measured 2129.18 µmol g−1.142 High GAC is desirable for lowering the glycemic impact of foods, as it slows the diffusion and uptake of sugars.
The solubility index and swelling power of dietary fiber affect its hydration behavior. These properties are typically determined by the composition of fiber, including pectin content, degree of polymerization and particle size. As an illustration, banana pseudostem fiber showed a swelling capacity of about 15.8 mL water per g fiber and a water solubility of 13.2%, reflecting moderate swelling and low solubility.141 Fibers with higher pectin content demonstrate significantly increased solubility. For instance, pineapple core powders show remarkable water solubility, quantified at approximately 60–70%.145 One study revealed that sugarcane bagasse has a relatively low swelling power of about 0.35%, which is due to its dense and lignified structure that limits water absorption. However, after treating the bagasse with a 0.1 N sodium hydroxide (NaOH) solution, the swelling power increased significantly to 0.99%. This indicates that mild alkaline treatment effectively disrupts the lignin–carbohydrate complex, making cellulose and hemicellulose more accessible for water interaction and hydration.5 Plant stem fibers are not just leftovers from food production, and they are remarkable sources of dietary benefits that enhance day-to-day meals and contribute positively to human health.
8. Health benefits of plant stem dietary fiber
In this context, we examine recent findings on stem-derived fibers, especially from broccoli, cauliflower, banana, pineapple, and sugarcane, focusing on their prebiotic effects, functional-food applications, interactions with minerals/phytochemicals, and impacts on antioxidant and metabolic health.8.1 Prebiotic effects and gut microbiota modulation
Stem fibers function as non-digestible substrates for colonic microbiota, leading to the production of short-chain fatty acids (SCFAs), including acetate, propionate, and butyrate. This fermentation process selectively promotes the growth of beneficial microbial populations such as Bifidobacterium, Lactobacillus, Faecalibacterium, and Roseburia. These SCFAs are crucial for maintaining gut health and have been linked to various positive physiological effects, including anti-inflammatory properties and improved intestinal barrier function.146 In a human intervention trial, Chong et al.147 found that green banana pseudostem powder increased seven beneficial species, including Faecalibacterium prausnitzii, Bifidobacterium longum, B. bifidum, B. adolescentis, Ruminococcus inulinivorans, etc., while pineapple stem fiber powder enriched six species like Bacteroides ovatus, B. intestinalis, R. inulinivorans. Similarly, the levels of short-chain fatty acids (SCFAs) in participants were significantly enhanced. Both supplements improved SCFA production, metabolism and bowel function. Such findings underscore the importance of dietary fibers in influencing gut microbiota and their metabolic byproducts.147In vitro fermentations further confirm these effects, and a study reported that pineapple stem boosted faecal growth of Lactobacilli and Bifidobacteria and yielded high acetate, propionate and butyrate levels.148 Campos et al.149 demonstrated that pineapple by-product flours promote overall gut fermentation and release phenolics into the lumen, thereby creating an antioxidant environment. Broccoli stalk fiber fractions are known to possess prebiotic properties. Núñez-Gómez et al.6 isolated insoluble fiber from broccoli stalks, which is particularly rich in uronic acids. Following a 48-hour fermentation with human faecal samples, their study found that this isolated fiber produced a significantly higher total yield of short-chain fatty acids (SCFAs) compared to freeze-dried whole broccoli stalks. This was attributed to the greater presence of uronic acid, the main component of pectin and polyphenol content in the insoluble broccoli stalk fiber fractions.6
Sugarcane-derived dietary fibers have been shown to modulate the gut microbiota and enhance the production of beneficial microbial metabolites. Gamage et al.146 utilized an in vitro gut fermentation model to compare sugarcane stem fiber with other dietary supplements. The study revealed that sugarcane fibre induced specific increases in short-chain fatty acids (SCFAs), which were positively correlated with the proliferation of bacteria that produce SCFAs. In a complementary in vivo study, Pelpolage et al.148 administered a soluble fraction of sugarcane bagasse fiber (BSF) to rats and observed a significant elevation in cecal acetate and total SCFA concentrations compared to controls. BSF supplementation also promoted the abundance of health-associated bacterial genera such as Bifidobacterium, Blautia, Akkermansia, and Roseburia. Remarkably, the SCFA profile in BSF-fed rats was comparable to that observed with commercial xylo-oligosaccharide supplementation. Furthermore, BSF intake enhanced markers of gut barrier integrity, including elevated levels of mucin and immunoglobulin A (IgA), indicating improved intestinal health.148 Recent research findings underscore a notable trend in commercial fiber products. For example, an in vitro study demonstrated that the combination of a dried sugarcane-stem fiber supplement with wheat dextrin and psyllium significantly increased levels of beneficial short-chain fatty acids, specifically acetate, propionate, and butyrate. This suggests a compelling synergy among these fibers, highlighting their potential to effectively enhance gut health.146
SCFAs, mainly acetate, propionate, and butyrate, are produced by microbial fermentation and are known to be crucial for maintaining host health. These metabolites serve as energy sources and signaling molecules that strengthen the intestinal barrier and control immunity. By activating G-protein-coupled receptors (GPR41, GPR43, and GPR109A) on immune cells, SCFAs function as essential signaling molecules that enhance anti-inflammatory mediators, suppress pro-inflammatory cytokines, and promote regulatory T cell differentiation. In the gastrointestinal tract, butyrate serves as the main energy source for colonocytes and strengthens epithelial integrity by upregulating tight junction proteins, stimulating mucus secretion, and inducing antimicrobial peptide production, collectively reducing gut permeability and pathogen translocation. Additionally, SCFAs decrease luminal pH and promote mucus production, which inhibits pathogens and lessens inflammation. As a result, SCFAs connect fiber consumption to immunological and metabolic advantages by lowering inflammation and maintaining intestinal barrier function.
Moreover, SCFAs regulate insulin metabolism through several pathways: propionate stimulates intestinal gluconeogenesis and enhances insulin sensitivity, acetate modulates the release of incretin hormones such as GLP-1 and PYY, and butyrate improves pancreatic β-cell function while also supporting insulin sensitivity, lipid metabolism, and even distant tissues epigenetic regulation.150 Major SCFAs acetate and propionate affect glucose and lipid metabolism in part by activating the sympathetic nervous system (SNS). The exact mechanisms are complex and encompass interactions with particular cellular receptors, hormone release, and the gut-brain axis.151 When it comes to systemic metabolic regulations, acetate reduces food intake by modulating hypothalamic neurons after crossing the blood–brain barrier. SCFAs support metabolic homeostasis throughout the body by promoting mitochondrial function, energy expenditure, and thermogenesis through the activation of the AMPK and PPAR pathways. SCFAs are the biochemical bridge between dietary fibers from plant stems and systemic health. They not only fuel colonocytes but also train the immune system, seal the gut barrier, fine-tune hormone release, and reprogram metabolism toward efficiency and resilience. Therefore, stem-derived fibers that produce abundant SCFAs can be positioned as powerful functional food ingredients.152 A schematic diagram of the prebiotic effect of stem-derived dietary fiber and SCFAs production is illustrated in Fig. 8.
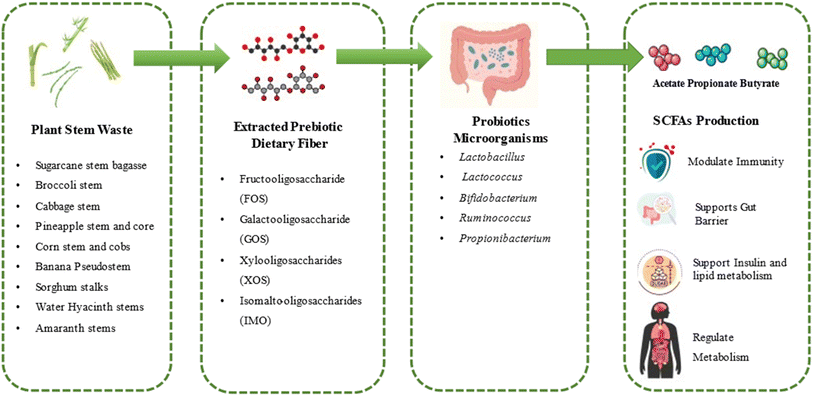 | ||
| Fig. 8 Schematic diagram of the prebiotic effect of stem-derived dietary fiber and SCFAs production. | ||
8.2 Bioavailability and phytochemical interactions
One of the major health advantages linked to dietary fibers derived from plant stems is increased nutrient bioavailability. Minerals such as calcium, magnesium, and iron can be bound by soluble fractions of these fibers, especially pectin and hemicelluloses, in the gastrointestinal tract. This can delay the precipitation of these minerals and allow for a gradual release, which enhances absorption in the small intestine. To mitigate deficiencies commonly associated with plant-based diets, for instance, dietary fiber derived from sugarcane bagasse has been shown to improve intestinal uptake of iron through its chelating and slow-release properties. Similar to this, broccoli stems, which are high in soluble fibers and pectic polysaccharides, have shown that they can increase calcium bioavailability by preserving mineral solubility at intestinal pH levels. Beyond minerals, stem fibers also modulate the bio accessibility of polyphenols and other phytonutrients by delaying gastric emptying and extending the residence time of digesta in the upper gastrointestinal tract.153 A study revealed that native banana pseudo-stem flour (Musa spp.) contains elevated levels of total dietary fiber, polyphenols, and flavonoids, surpassing those found in its tender core, and concurrently demonstrates stronger antioxidant and radical-scavenging activity.154 However, the fiber matrix can modulate their release and absorption. For example, broccoli stalk fibers contain 100 mg/100 g of glucosinolates and polyphenols 75 mg/100 g by dry weight.6 D. Campos et al.149 investigated the behavior of phenolic compounds in pineapple flours, noting that these compounds are predominantly bound within cell walls and can be liberated during fermentation. The study demonstrated that the digestion of pineapple fiber facilitates a substantial release of phenolic compounds into the colon, thereby augmenting local antioxidant capacity. It outlined that stem fibers may serve as effective carriers for antioxidants. Moreover, interactions with specific phytochemicals are notable in Brassica crops. A study on isolated dietary fiber fractions from broccoli and cauliflower stems reported the ability to trap glucosinolates and flavonoids.6 Insoluble broccoli fiber is rich in uronic acid and has a higher concentration of glucosinolates. When fermented by gut bacteria, these glucosinolates can be transformed into isothiocyanates, which possess anticancer properties. Although the stem fiber does not create new compounds, its structural matrix can affect the rate and site of release for these bioactive substances within the gut. Conversely, dietary fiber has the potential to chelate minerals, thereby decreasing their absorption within the small intestine. The research regarding broccoli suggests that fiber may influence the bioavailability of minerals and phenolic compounds, as these substances are bound to the fiber matrix. Consequently, certain minerals such as calcium and iron and phytochemicals present in the stems may remain partially shielded until they reach colonic fermentation.168.3 Lowering the glycemic index (GI) of foods
Incorporating plant stem fibers into foods typically lowers their glycemic index and attenuates post-meal glucose spikes. The high fiber content slows starch hydrolysis and delays glucose absorption. For example, experimental diets containing banana pseudostem were found to have a low GI in animal studies.155 Bhaskar et al.78 reported that banana pseudostem dietary fiber has low glycemic index. More broadly, fibers increase the viscosity of the intestinal contents and can form gel-like matrices, thereby blunting the rate of sugar diffusion. Studies incorporating banana pseudostem flour into staples like noodles and biscuits reveal a notable increase in total dietary fiber and a corresponding drop in GI. For example, fiber-enriched noodles made with banana pseudostem showed an in vitro glycemic index (GI) reduction from the medium range 50–75 to the low-GI category, along with an increase in fiber content from approximately 3.3% to 4.7%.156 Sugarcane bagasse, traditionally a waste product from sugar processing, is rich in insoluble fiber (cellulose, hemicellulose, lignin) and has been successfully leveraged in baked goods. Fortified biscuits and noodles enriched with bagasse demonstrated higher fiber content and predicted lower GI responses, suggesting suitability for diabetic diets.39,1579. Key trade – offs in utilization plant stem waste as dietary fiber source for food applications
Plant stem wastes exhibit wide variability in dietary fiber composition and physicochemical behavior, giving rise to multiple interrelated trade-offs that ultimately determine their suitability for food applications. While high fiber yield is often emphasized, functional performance in foods and physiological relevance depend on a careful balance between fiber composition, extractability, hydration behavior, fermentability, and processing-induced changes. A primary trade-off exists between total dietary fiber (TDF) content and fermentability. Several stem residues, such as corn stalks, corn cobs, sugarcane bagasse, and sorghum stalks, contain exceptionally high TDF levels as discussed earlier. However, this fiber is predominantly composed insoluble dietary fiber embedded within lignin-rich cell walls. Lignin, which may account for 10–30% of stem biomass, is non-fermentable and entirely insoluble, conferring bulking capacity and structural rigidity but substantially limiting microbial fermentation and short-chain fatty acid (SCFA) production in the colon.158 For example, sugarcane bagasse and corn stalks have very high TDF values that frequently reflect a largely insoluble, low-fermentability fiber matrix. Highly lignified stems offer mechanical stability and thermal resistance, but require harsh processing to achieve food-grade functionality.159,160In contrast, stems with moderately high fiber content but lower lignin levels, such as banana pseudostem,73 water hyacinth,67 and Brassica stalks,6 exhibit mixed and balanced polysaccharide profiles comprising cellulose, hemicellulose, and pectic substances. These mixed matrices offer a more favorable balance between bulking effects and fermentability, supporting both water retention and microbial utilization, despite slightly lower overall TDF levels.
A second critical trade-off concerns extraction yield versus functional quality. Importantly, this trade off not solely depend on the botanical origin and structural characteristics of the stem waste, but is strongly influenced by depend on extraction method. Aggressive extraction methods, including strong acids, alkali and high-intensity physical treatments, can achieve very high fiber recovery and may yield nearly pure cellulose. However, such fibers are highly crystalline, entirely insoluble, and exhibit limited swelling, viscosity, and fermentability.115 In contrast, milder extraction approaches that preserve hemicellulose and pectin fractions, although resulting in lower mass yield, retain hydrophilic and gel-forming components that significantly enhance water-holding capacity, fermentability, and physiological functionality.161 Thus, maximizing yield often occurs at the expense of functional performance relevant to food applications. The balance between insoluble dietary fiber (IDF) and soluble dietary fiber (SDF) further governs hydration behavior and fermentation kinetics. Insoluble-dominant fibers form porous structures with high water-holding capacity and strong bulking effects but ferment slowly, whereas SDF-rich fibers contribute viscosity, gel formation, and rapid microbial fermentation. For example, sugarcane stem bagasse and corn cob fibers are overwhelmingly insoluble and function primarily as bulking agents, while Brassica stems and banana pseudostem contain sufficient soluble fractions to support both hydration and prebiotic activity.161
Beyond composition, particle size reduction introduces an additional trade-off between technological performance and physiological functionality. While fine milling improves dispersibility and mouthfeel in food systems, excessively small particles may reduce bulking effects and diminish stimulation of intestinal motility. Evidence suggests that coarse insoluble fibers are more effective at increasing faecal bulk and modulating gastrointestinal transit than finely ground fibers, even when chemical composition is similar. Thus, particle size optimization must balance processability with physiological efficacy. However, all these factors can be varied according to the cultivar type and maturity stage, which also affect fiber extractability and functionality.162
When these trade-offs are critically evaluated, banana pseudostem, Brassica stems and water hyacinth emerge as the most promising stem wastes for multifunctional food-grade dietary fiber applications. Pineapple stem fiber represents a moderately promising alternative, particularly suited for bulking and texture-modifying roles, owing to its high insoluble fiber content and moderate hydration capacity. However, its limited soluble fiber fraction restricts fermentability and prebiotic functionality unless further modification is applied. In contrast, highly lignified residues such as sugarcane bagasse, corn stalks and sorghum, are primarily suited for bulking applications rather than multifunctional dietary fiber use.
10. Applications of plant stem dietary fiber in the food industry
Significant amounts of industrial waste and by-products generated during food processing remain underutilized, contributing to considerable environmental pollution. Specific food products can serve as valuable sources of dietary fiber, which not only aids in reducing pollution but also enhances nutritional value.93 While earlier sections of this review have identified important trade-offs between fiber composition, extraction severity, and functional performance, existing food application studies demonstrate that a wide range of stem-derived fibers can be successfully integrated into food matrices when formulation level, particle size, and processing conditions are appropriately optimized.Researchers are currently incorporating fibers derived from plant stems into foods and supplements to enhance their nutritional value. One notable application of these stem-derived dietary fibers is in the production of fiber-fortified baked goods. This approach aims to improve the nutritional quality of various food products. For instance, sugarcane bagasse fiber has been added to bakery and pasta products. Confocal microscopy studies show that adding ∼5–8% bagasse fiber (w/w) to chapatti (Indian flatbread) or noodles is feasible: chapattis with 8% SCB displayed integrated fiber networks and maintained microbial safety.126 Similarly, banana pseudostem flour has been tested in bread: incorporation up to 10% (g/100 g flour) raises fiber content but can alter loaf volume and color.163 Corn cob fiber powder, derived from corn cob residues, has been investigated for its incorporation into a variety of food products. Sweet corn cob flour (SCCF) has been used as a partial substitute for rice flour in gluten-free muffins, resulting in products with improved volume, texture, and sensory properties. A study revealed that up to 20% substitution with SCCF enhanced the total dietary fiber content of muffins while maintaining consumer acceptability.164 Nevertheless, higher substitution levels negatively affected the dough rheology and bread texture, emphasizing the importance of balance between functionality and sensory quality. While most research has focused on corn cob applications, studies on the use of corn stalk or stem fiber in food products remain limited. However, due to the similar lignocellulosic composition of corn stalks, primarily composed of cellulose, hemicellulose, and lignin, these materials also present a promising source of insoluble dietary fiber. Further investigations into stalk fiber incorporation could potentially expand the scope of functional food development from corn agricultural residues.
In addition, many researchers have fortified fiber supplements and powders with stem dietary fiber. Munir et al. (2024) studied green banana pseudostem powder and pineapple stem fiber powder as nutrition supplements. Such supplements can deliver resistant starch and fiber with minimal dietary change.165 Likewise, commercial products (e.g., inulin powders, resistant starch) are analogous to stem fiber supplements. As evidence of functionality, adding banana pseudostem fiber to meat formulations increased water-holding capacity and antioxidant stability,166 suggesting its use as a texturizer and natural antioxidant. In traditional Southeast Asian confectionery, corn cob powder (CCP) has been employed to enrich fiber content. For example, Kumar et al.167 incorporated CCP into Kuih Kaswi, a local dessert, at varying levels between 5% and 25%. The study reported that formulations with up to 15% CCP maintained favorable physicochemical properties and sensory acceptability. The inclusion of CCP contributed to a significant increase in crude fiber content, demonstrating its potential as a functional additive in traditional food matrices. However, there is limited research that has been conducted on plant stem-derived fiber and its incorporation into food applications. Despite the rich potential that lies within the fibers extracted from plant stems, research exploring their incorporation into food applications remains scarce. This limited investigation creates an opportunity for further academic inquiry into the potential benefits and functional properties of these fibers within the realm of food science and nutrition. Applications of plant stem dietary fiber in the food industry are summarized in Table 3.
| Plant source | Waste type | Food application | Reference |
|---|---|---|---|
| Sugarcane | Stem bagasse | Sugarcane stem bagasse (SB) dietary fiber incorporated into noodles. In this research, SB was incorporated into the development of noodles at three different ratios 5%, 10% and 15%. Total dietary fibre of noodles significantly increased from 3.39% to 13.85% with 15% SB incorporation | 39 |
| Sugarcane stem bagasse dietary fiber incorporated into bread dough. The substitution of dietary fiber from sugarcane bagasse varied from 0 to 15 g/100 g of wheat flour mass | 40 | ||
| Dietary cookies made with wheat flour and sugarcane bagasse fiber had good texture, color and mouthfeel; consumer panels rated bagasse-enriched cookies and biscuits highly | 88 | ||
| Banana | Pseudostem | Banana pseudostem substitution in wheat flour biscuits. 10% banana-pseudostem flour showed much higher ash, protein, fiber and antioxidant phenolics and had comparable taste and texture to regular biscuits | 168 |
| Functional juice developed from banana pseudostem by enzymatically converting sap from banana trunk/stalk, and found rich in nondigestible oligosaccharides and rare sugars. This juice leverages the high cellulosic content of the banana pseudostem to yield prebiotic fiber and low-cal sweeteners in a drink | 169 | ||
| Fiber enrichment in noodles using banana pseudo-stem (Musa paradisiaca) tender core (BPP) fibrous powder. BPP was incorporated at different levels, 5, 10 and 15% in which 5% was found to be the best based on cooking characteristics and sensory evaluation | 156 | ||
| Pineapple | Stem | Prebiotic fiber supplement significantly increased gut Bifidobacteria and SCFA production, improving bowel regularity and reducing constipation symptoms. Thus, pineapple fiber powders act as prebiotic fiber supplements | 147 |
| Sweet corn | Stem and cobs | Sweet corn cobs fibrous flour incorporated gluten-free muffins – study revealed that treatment: 25% of corn cob flour and 75% wheat flour with a specific proportion of sweet corn cob flour, received the highest ratings from the sensory evaluation | 164 |
| Bamboo | Shoots | Incorporating bamboo shoot fiber into gluten-free/low-carb bread. Bamboo fiber was introduced to the bread recipe in proportions ranging from 0 to 10%. The results showed that adding from 4 to 6% bamboo fiber to the control bread resulted in a slight rise of around 3% in volume. The incorporation of the fiber had the greatest impact on the textural parameters. Bamboo fiber incorporated bread had very low carbohydrate content (15.3%), it was also characterized by a high fiber content (12.2%) and a low caloric value (176.7 kcal/100 g) | 170 |
| Incorporating bamboo shoots dietary fiber to milk pudding (cooked milk gel) increased the product's yield stress, viscosity and hardness (up to an optimum fiber level), producing a firmer, more elastic pudding | 171 | ||
| Bamboo shoots fortified yoghurt – yoghurt with 1.0% bamboo shoot powder was found to be the best among the treatments with appearance, texture, aroma, taste and overall acceptability | 172 | ||
| Reduced-salt, phosphate-free Bologna sausage was made with 2.5–5.0% added bamboo fiber. The fiber-filled sausages had higher emulsion stability and firmer bite, developing a denser protein network | 173 | ||
| Amaranth | Stems | Incorporating amaranth as a bread-enriching ingredient. The incorporation of amaranth flour into bread and related bakery formulations significantly enhances their functional and biological value. Experimental results indicate improvements in the nutritional content, antioxidant capacity, and digestibility of the final products. The use of amaranth flour has a positive influence on technological properties, such as dough stability, moisture retention, and shelf-life extension | 174 |
11. Challenges and opportunities
Many plant stem wastes are high in dietary fiber, but turning them into food-grade ingredients faces various technological, biological, and knowledge-based challenges beyond just composition. A significant challenge arises from the structural recalcitrance of lignocellulosic matrices, which hinders effective fiber modification and limits the conversion of insoluble fractions into physiologically relevant, fermentable forms without extensive processing. Although this recalcitrance provides stability during food processing, it complicates the creation of multifunctional fibers with predictable physiological outcomes.From a processing perspective, scalability and process integration remain critical bottlenecks. Although green extraction technologies such as UAE, MAE, SWE, and PEF have demonstrated promising improvements in fiber accessibility and functionality at laboratory scale, their industrial readiness, energy efficiency, and economic feasibility for large-volume stem waste streams remain insufficiently validated. The absence of standardized performance benchmarks and techno-economic assessments further hinders industrial adoption.175
Another key challenge with stem-derived feedstocks is their inherent variability due to seasonal availability, botanical diversity, agronomic practices, and processing methods. This heterogeneity in fiber composition complicates ingredient standardization and consistent performance in food formulations, requiring advanced characterization, blending strategies, or adaptive processing for reproducible functionality.176 Biological evidence gaps remain significant. The current understanding of stem-derived dietary fibers mainly relies on compositional analysis and in vitro studies, while in vivo data on fermentability, dose–response, gastrointestinal tolerance, and long-term effects are limited. This lack of physiological validation hinders regulatory confidence and slows the transition from promising raw materials to validated functional ingredients.
These challenges also present notable opportunities for innovation. Targeted approaches for structural modification, such as controlled delignification, selective depolymerization, and the conversion of insoluble dietary fiber (IDF) to soluble dietary fiber (SDF), offer paths to customize fiber functionality without causing excessive degradation. Hybrid processing strategies that combine mechanical, enzymatic, and mild physicochemical treatments may help preserve the complexity of functional polysaccharides while enhancing their fermentability. Additionally, advancements in microbiome science and structure–function modeling create new chances to design application-specific stem-derived fibers that align with specific physiological outcomes. Collectively, continued advancements in green processing technologies, along with a better understanding of fiber–microbiota interactions and standardized evaluation frameworks, can facilitate the development of high-value, food-grade dietary fiber ingredients from plant stem wastes. This supports sustainable food systems and the objectives of a circular bioeconomy.
12. Conclusion
Plant stem waste represents a major yet underexploited fraction of agri-food biomass with significant potential as a dietary fiber source for novel food applications. This review highlights the potential of plant stem waste, an underutilized by-product of the agri-food sector, as a source of dietary fiber for new food applications. It emphasizes that the suitability of stem wastes for food depends not only on total fiber content but also on the interplay of anatomical structure, fiber composition, extractability, functional performance, and technological feasibility.Stems with lower lignification and higher parenchymatous tissue content, particularly banana pseudostem, pineapple stem and core, and water hyacinth stem, emerge as the most promising candidates for food-grade dietary fiber production due to their balanced soluble and insoluble fiber profiles, superior extractability, and favorable hydration and fermentability characteristics. Among these, banana pseudostem stands out owing to its abundance, relatively mild processing requirements, and demonstrated applicability in food formulations. In contrast, highly lignified stem wastes such as sugarcane bagasse, corn cobs, and sorghum stalks, despite their high fiber content, present significant barriers to food use and are more suitably directed toward non-food or hybrid valorization pathways. Moderately lignified stems, including Brassica and amaranth stems, offer selective opportunities for food applications but require targeted processing and formulation strategies. Also, this review highlights that green extraction methods such as UAE, MAE and EAE particularly well suited to low and moderately lignified stems, enabling improved fiber recovery while preserving functional properties. In contrast, highly lignified stem residues such as sugarcane bagasse, corn cobs, and sorghum stalks exhibit limited direct suitability for food applications due to poor extractability and reduced fermentability. However, these materials need not be excluded from food systems; instead, they may be strategically valorized through targeted delignification and controlled conversion of insoluble dietary fiber into soluble fractions using chemical, enzymatic, or physicochemical treatments. When coupled with appropriate green extraction technologies, particularly UAE, MAE, and EAE, such approaches may enhance fiber recovery and functional performance. Nevertheless, their practical implementation will depend on process efficiency, cost-effectiveness, and regulatory compliance, highlighting the need for further techno-economic and safety evaluations.
Author contributions
Writing, P. D. N. Boteju, review and editing K. G. L. R. J. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare that they have no conflicts of interest.Data availability
No datasets were generated or analyzed during the current study.Acknowledgements
This research was funded by the Research Council, University of Sri Jayewardenepura, Sri Lanka, grant number RC/URG/FOT/2024/68.References
- FAO, Definitional Framework of Food Loss - Save Food: Global Initiative on Food Loss and Waste Reduction, Defin Framew Food Loss - Work Pap, 2014, vol. 18, pp. 1–10, Available from: http://www.fao.org/fileadmin/user_upload/save-food/PDF/FLW_Definition_and_Scope_2014.pdf.
- S. Sarkar, M. Skalicky, A. Hossain and M. Brestic, Management of crop residues for improving input use efficiency and agricultural sustainability, Sustainability, 2020, 12(23), 9808, DOI:10.3390/su12239808.
- E. Imbert, Food waste valorization options: Opportunities from the bioeconomy, Open Agric., 2017, 2(1), 195–201, DOI:10.1515/opag-2017-0020.
- F. Zou, C. Tan, B. Zhang and W. Wu, The valorization of banana by-products: Nutritional composition, bioactivities, applications, and future development, Foods, 2022, 11(20), 3170, DOI:10.3390/foods11203170.
- S. Paroha, V. P. Srivastava and N. Chaturvedi, Sugarcane bagasse as dietary fibre, Indian J. Pure Appl. Biosci., 2020, 8(6), 590–597, DOI:10.18782/2582-2845.8442.
- V. Núñez-Gómez, R. González-Barrio, N. Baenas, D. A. Moreno and M. J. Periago, Dietary-fibre-rich fractions isolated from broccoli stalks as a potential functional ingredient with phenolic compounds and glucosinolates, Int. J. Mol. Sci., 2022, 23(21), 13309, DOI:10.3390/ijms232113309.
- N. A. Sagar, S. Pareek, S. Sharma, E. M. Yahia and M. G. Lobo, Fruit and vegetable waste: Bioactive compounds, their extraction, and possible utilization, Compr. Rev. Food Sci. Food Saf., 2018, 17(3), 512–531, DOI:10.1111/1541-4337.12330.
- F. H. Schweingruber and A. Börner, The Plant Stem, Cham, Springer, 2018, pp. 1–15, DOI:10.1007/978-3-319-73524-5.
- T. Speck and I. Burgert, Plant stems: Functional design and mechanics, Annu. Rev. Mater. Res., 2011, 41, 169–193, DOI:10.1146/annurev-matsci-062910-100425.
- G. W. Sileshi, E. Barrios, J. Lehmann and F. N. Tubiello, An organic matter database (OMD): Consolidating global residue data from agriculture, fisheries, forestry and related industries, Earth Syst. Sci. Data, 2025, 17(2), 369–391, DOI:10.5194/essd-17-369-2025.
- E. O. Ajala, J. O. Ighalo, M. A. Ajala, A. G. Adeniyi and A. M. Ayanshola, Sugarcane bagasse: a biomass sufficiently applied for improving global energy, environment and economic sustainability, Bioresour. Bioprocess, 2021, 8(1), 1–9, DOI:10.1186/s40643-021-00440-z.
- A. M. Shah, H. Zhang, M. Shahid and H. Ghazal, The vital roles of agricultural crop residues and agro-industrial by-products to support sustainable livestock productivity in subtropical regions, Animals, 2025, 15(8), 1184, DOI:10.3390/ani15081184.
- C. Hiranobe, A. S. Gomes, F. Friol and G. Tolosa, Sugarcane bagasse: Challenges and opportunities for waste recycling, Clean Technol., 2024, 6(2), 662–699, DOI:10.3390/cleantechnol6020035.
- S. Hussain, I. Jõudu and R. Bhat, Dietary fiber from underutilized plant resources—A positive approach for valorization of fruit and vegetable wastes, Sustainability, 2020, 12(13), 5401, DOI:10.3390/su12135401.
- I. Buljeta, D. Šubarić, J. Babic and A. Pichler, Extraction of dietary fibers from plant-based industry waste: A comprehensive review, Appl. Sci., 2023, 13(16), 9309, DOI:10.3390/app13169309.
- V. Núñez-Gómez, R. González-Barrio and M. J. Periago, Interaction between dietary fibre and bioactive compounds in plant by-products: Impact on bioaccessibility and bioavailability, Antioxidants, 2023, 12(4), 976, DOI:10.3390/antiox12040976.
- H. Seddiqi, E. Oliaei, H. Honarkar, J. Jin, L. C. Geonzon and R. G. Bacabac, et al., Cellulose and its derivatives: towards biomedical applications, Cellulose, 2021, 28, 1893–1931, DOI:10.1007/s10570-020-03674-w.
- J. Liu, H. Sixta, Y. Ogawa, M. Hummel, M. Sztucki and Y. Nishiyama, et al., Multiscale structure of cellulose microfibrils in regenerated cellulose fibers, Carbohydr. Polym., 2023, 324, 121512, DOI:10.1016/j.carbpol.2023.121512.
- Y. Zeng, M. E. Himmel and S. Y. Ding, Visualizing chemical functionality in plant cell walls, Biotechnol. Biofuels, 2017, 10(1), 286, DOI:10.1186/s13068-017-0953-3.
- H. Carlsen and A. M. Pajari, Dietary fiber – a scoping review for Nordic Nutrition Recommendations 2023, Food Nutr. Res., 2023, 67, 9979, DOI:10.29219/fnr.v67.9979.
- E. Marinho, Cellulose: A comprehensive review of its properties and applications, Sustainable Chem. Environ., 2025, 11, 100283, DOI:10.1016/j.scenv.2025.100283.
- M. Akhlaghi, The role of dietary fibers in regulating appetite, an overview of mechanisms and weight consequences, Crit. Rev. Food Sci. Nutr., 2024, 64(10), 3139–3150, DOI:10.1080/10408398.2022.2130160.
- P. Portincasa, L. Bonfrate, M. Vacca and M. De Angelis, Gut microbiota and short chain fatty acids: Implications in glucose homeostasis, Int. J. Mol. Sci., 2022, 23(3), 1105, DOI:10.3390/ijms23031105.
- B. M. Bohrer, M. Izadifar and S. Barbut, Structural and functional properties of modified cellulose ingredients and their application in reduced-fat meat batters, Meat Sci., 2022, 195, 109011, DOI:10.1016/j.meatsci.2022.109011.
- D. Lavanya, P. Kulkarni, M. Dixit, P. K. Raavi and L. N. V. Krishna, Sources of cellulose and their applications: A review, Int. J. Drug Formulation Res., 2011, 2, 19–38 Search PubMed.
- H. V. Scheller and P. Ulvskov, Hemicelluloses, Annu. Rev. Plant Biol., 2010, 61, 263–289, DOI:10.1146/annurev-arplant-042809-112315.
- W. Xiong, L. Devkota, B. M. Flanagan and Z. Gu, Plant cell wall composition modulates the gut microbiota and metabolites in in-vitro fermentation, Carbohydr. Polym., 2023, 316, 121074, DOI:10.1016/j.carbpol.2023.121074.
- J. J. Liao, N. H. Abd Latif, D. Trache and N. Brosse, Current advancement on the isolation, characterization and application of lignin, Int. J. Biol. Macromol., 2020, 162, 1676–1693, DOI:10.1016/j.ijbiomac.2020.06.168.
- V. K. Ponnusamy, D. D. Nguyen, J. Dharmaraja, S. Shobana, R. Banu and G. Saratale, et al., A review on lignin structure, pretreatments, fermentation reactions and biorefinery potential, Bioresour. Technol., 2018, 271, 462–472, DOI:10.1016/j.biortech.2018.09.070.
- D. Mohnen, Pectin structure and biosynthesis, Curr. Opin. Plant Biol., 2008, 11, 266–277, DOI:10.1016/j.pbi.2008.03.006.
- C. Polesca, R. Sousa, M. Dias and J. Coimbra, Extraction of pectin from passion fruit peel, Food Eng. Rev., 2020, 12, 1–13, DOI:10.1007/s12393-020-09254-9.
- M. Drahansky, Liveness detection in biometrics, in Advanced Biometric Technologies, ed. Chetty G. and Yang J., IntechOpen, London, 2011, pp. 173–198, DOI:10.5772/17205.
- M. Marić, A. Ninčević Grassino, Z. Zhu, F. J. Barba, M. Brnčić and S. Rimac Brnčić, An overview of the traditional and innovative approaches for pectin extraction from plant food wastes and by-products: Ultrasound-, microwaves-, and enzyme-assisted extraction, Trends Food Sci. Technol., 2018, 76, 28–37, DOI:10.1016/j.tifs.2018.03.022.
- H. U. Yoo, M. J. Ko and M. S. Chung, Hydrolysis of beta-glucan in oat flour during subcritical-water extraction, Food Chem., 2019, 308, 125670, DOI:10.1016/j.foodchem.2019.125670.
- J. Bai, Y. Ren, M. Fan, H. Qian, L. Wang and G. Wu, et al., Physiological functionalities and mechanisms of β-glucans, Trends Food Sci. Technol., 2019, 88, 98–107, DOI:10.1016/j.tifs.2019.03.023.
- G. Maheshwari, S. Sowrirajan and B. Joseph, Extraction and isolation of β-glucan from grain sources: A review, J. Food Sci., 2017, 82, 1223–1233, DOI:10.1111/1750-3841.13765.
- M. M. Tosif, A. Najda, A. Bains, R. Kaushik, S. B. Dhull and P. Chawla, et al., A comprehensive review on plant-derived mucilage: Characterization, functional properties, applications, and its utilization for nanocarrier fabrication, Polymers, 2021, 13(7), 1066, DOI:10.3390/polym13071066.
- M. S. Amiri, V. Mohammadzadeh, M. E. Taghavizadeh Yazdi, M. Barani, A. Rahdar and G. Kyzas, Plant-based gums and mucilages applications in pharmacology and nanomedicine: A review, Molecules, 2021, 26(6), 1770, DOI:10.3390/molecules26061770.
- K. Q. Lau, M. R. Sabran, S. R. Shafie and S. W. Chan, Incorporation of sugarcane bagasse in the development of high dietary fibre noodles, Int. J. Food Sci. Technol., 2022, 57(7), 3371–3381, DOI:10.1111/ijfs.15796.
- A. Sangnark and A. Noomhorm, Effect of dietary fiber from sugarcane bagasse and sucrose ester on dough and bread properties, LWT-Food Sci. Technol., 2004, 37, 697–704, DOI:10.1016/j.lwt.2004.02.015.
- S. Imman, P. Khongchamnan, N. Laosiripojana, T. Kreetachat, C. Sakulthaew and C. Chokejaroenrat, et al., Fractionation and characterization of lignin from sugarcane bagasse using a sulfuric acid catalyzed solvothermal process, RSC Adv., 2021, 11, 26773–26784, 10.1039/d1ra03237b.
- M. Vijerathna, I. Wijesekara, R. Perera, A. Maralanda, M. Jayasinghe and I. Wickramasinghe, Physico-chemical characterization of cookies supplemented with sugarcane bagasse fibres, Vidyodaya J. Sci., 2019, 22, 29–39, DOI:10.4038/vjs.v22i1.6062.
- T. Shinali, Y. Zhang, M. Altaf, A. Nsabiyeze, Z. Han and S. Shi, et al., The valorization of wastes and byproducts from cruciferous vegetables: A review on the potential utilization of cabbage, cauliflower, and broccoli byproducts, Foods, 2024, 13(8), 1163, DOI:10.3390/foods13081163.
- A. Mansour, L. Shekib, N. Elshimy and M. Sharara, Biological evaluation of raw and steamed broccoli and cauliflower as sources of dietary fibers, J. Food Dairy Sci., 2017, 8, 185–190, DOI:10.21608/jfds.2017.38199.
- S. Eyupoglu, Characterization of a Novel Cellulosic Fiber from Brassica oleracea Var. Italic Stem, J. Nat. Fibers, 2022, 19(14), 7480–7490, DOI:10.1080/15440478.2021.1950098.
- T. B. N. Brito, A. P. A. Pereira, G. M. Pastore, R. F. A. Moreira, M. S. L. Ferreira and A. E. C. Fai, Chemical composition and physicochemical characterization for cabbage and pineapple by-products flour valorization, Lwt, 2020, 124, 109028, DOI:10.1016/j.lwt.2020.109028.
- D. R. Joshi, H. Sieverding, H. Xu, H. Kwon, M. Wang and S. Clay, et al., A global meta-analysis of cover crop response on soil carbon storage within a corn production system, Agron. J., 2023, 115, 1–15, DOI:10.1002/agj2.21340.
- Y. Zhang, A. Ghaly and B. X. Li, Physical properties of corn residues, Am. J. Biochem. Biotechnol., 2012, 8, 44–53, DOI:10.3844/ajbbsp.2012.44.53.
- H. H. Bae, J. Y. Ha, Y. S. Go, J. H. Son, B. Y. Son and J. H. Kim, et al., High phytosterol levels in corn cobs point to their sustainable use as a nutritional source, Appl. Biol. Chem., 2022, 65(1), 2–12, DOI:10.1186/s13765-022-00736-4.
- P. Gandam, M. Chinta, A. Gandham, N. Prashanth, S. Konakanchi and A. Bhavanam, et al., A new insight into the composition and physical characteristics of corncob—substantiating its potential for tailored biorefinery objectives, Fermentation, 2022, 8(12), 704, DOI:10.3390/fermentation8120704.
- O. I. Oni, F. M. Olajuyigbe and C. O. Fatokun, Effective substrate loading for saccharification of corn cob and concurrent production of lignocellulolytic enzymes by Fusarium oxysporum and Sporothrix carnis, Curr. Biotechnol., 2019, 8(2), 109–115, DOI:10.2174/2211550108666191008154658.
- G. Castorina, C. Cappa, N. Negrini, F. Criscuoli, M. C. Casiraghi and A. Marti, et al., Characterization and nutritional valorization of agricultural waste corncobs from Italian maize landraces through the growth of medicinal mushrooms, Sci. Rep., 2023, 13(1), 1–17, DOI:10.1038/s41598-023-48252-9.
- J. Aniola, J. Gawecki, J. Czarnocinska and G. Galiński, Corncobs as a source of dietary fiber, Pol. J. Food Nutr. Sci., 2009, 59, 247–252 CAS.
- A. Nasir and N. Kamaruddin, Assessing the nutritional composition of sweet corn (Zea mays L. var. saccharata) stover and kernel corn (Zea mays L. var. indentata) stover for ruminant feed, J. Asian Sci. Res., 2023, 13, 136–148, DOI:10.55493/5003.v13i3.4907.
- S. Qiu, M. P. Yadav and L. Yin, Characterization and functionalities study of hemicellulose and cellulose components isolated from sorghum bran, bagasse and biomass, Food Chem., 2017, 230, 225–233, DOI:10.1016/j.foodchem.2017.03.028.
- W. Hu, L. Zhou and J. H. Chen, Conversion sweet sorghum biomass to produce value-added products, Biotechnol. Biofuels Bioprod., 2022, 15(1), 1–11, DOI:10.1186/s13068-022-02170-6.
- J. A. Andrade Alves, M. D. Lisboa dos Santos, C. C. Morais, J. L. Ramirez Ascheri, R. Signini and D. M. dos Santos, et al., Sorghum straw: Pulping and bleaching process optimization and synthesis of cellulose acetate, Int. J. Biol. Macromol., 2019, 135, 877–886, DOI:10.1016/j.ijbiomac.2019.06.123.
- H. Saeed, Y. Liu, L. Lucia and H. Chen, Evaluation of Sudanese sorghum and bagasse as a pulp and paper feedstock, BioResources, 2017, 12, 5212–5222, DOI:10.15376/biores.12.3.5212-5222.
- N. B. Appiah-Nkansah, J. Li, W. Rooney and D. Wang, A review of sweet sorghum as a viable renewable bioenergy crop and its techno-economic analysis, Renewable Energy, 2019, 143, 1121–1132, DOI:10.1016/j.renene.2019.05.066.
- A. Ibrahim, M. Hussin and A. S. Norhartini, Dietary fibre powder from pineapple by-product as a potential functional food ingredient, Acta Hortic., 2011, 902, 565–568, DOI:10.17660/ActaHortic.2011.902.75.
- S. Y. Shiau, M. Y. Wu and Y. L. Liu, The effect of pineapple core fiber on dough rheology and the quality of mantou, J. Food Drug Anal., 2015, 23(3), 493–500, DOI:10.1016/j.jfda.2014.10.010.
- E. Mardawati, A. T. Hartono, B. Nurhadi and H. N. Fitriana, Xylitol production from pineapple cores (Ananas comosus (L.) Merr) by enzymatic and acid hydrolysis using microorganisms Debaryomyces hansenii and Candida tropicalis, Fermentation, 2022, 8(12), 694, DOI:10.3390/fermentation8120694.
- D. I. Santos, C. F. Martins, R. A. Amaral, L. Brito, J. A. Saraiva and A. A. Vicente, et al., Pineapple (Ananas comosus L.) by-products valorization: Novel bio ingredients for functional foods, Molecules, 2021, 26, 3216, DOI:10.3390/molecules26113216.
- P. H. Chu, M. A. Jenol, L. Y. Phang, M. F. Ibrahim and S. Prasongsuk, Starch extracted from pineapple ( Ananas comosus ) plant stem as a source for amino acids production, Chem. Biol. Technol. Agric., 2021, 1–16, DOI:10.1186/s40538-021-00227-6.
- D. Campos, T. Ribeiro, J. Teixeira, L. Pastrana and M. Pintado, Integral valorization of pineapple (Ananas comosus L.) by-products through a green chemistry approach towards added value ingredients, Foods, 2020, 9, 60–75, DOI:10.3390/foods9010060.
- E. Auma, O. Peter, K. Ndiba and P. Gikuma, Characterization of water hyacinth ( E . crassipes ) from Lake Victoria and ruminal slaughterhouse waste as co - substrates in biogas production, SN Appl. Sci., 2019, 1(8), 1–10, DOI:10.1007/s42452-019-0871-z.
- C. N. Enyi, A. A. Uwakwe and M. O. Wegwu, Nutritional potentials of water hyacinth (Eichhornia crassipes), Direct Res. J. Pharm. Biol. Chem. Sci., 2020, 5(2), 14–18, DOI:10.26765/DRJPHET18652788.
- T. Ruan, R. Zeng, X. Y. Yin, S. X. Zhang and Z. H. Yang, Water hyacinth (Eichhornia crassipes) biomass as a biofuel feedstock by enzymatic hydrolysis, BioResources, 2016, 11, 2372–2380, DOI:10.15376/biores.11.1.2372-2380.
- S. Rezania, H. Alizadeh, J. Cho, N. Darajeh, J. Park and B. Hashemi, et al., Changes in composition and structure of water hyacinth based on various pretreatment methods, BioResources, 2019, 14, 6088–6099 CAS.
- S. Tanpichai, S. Biswas, S. Witayakran and H. Yano, Water hyacinth: a sustainable lignin-poor cellulose source for the production of cellulose nanofibers, ACS Sustainable Chem. Eng., 2019, 7, 1–12, DOI:10.1021/acssuschemeng.9b04095.
- S. Chonsakorn, S. Srivorradatpaisan and R. Mongkholrattanasit, Effects of different extraction methods on some properties of water hyacinth fiber, J. Nat. Fibers, 2019, 16(7), 1015–1025, DOI:10.1080/15440478.2018.1448316.
- A. Bayu, D. Nandiyanto, R. Ragadhita and S. Nur, Progress in the utilization of water hyacinth as effective biomass material, Environ. Dev. Sustainability, 2024, 26, 24521–24568, DOI:10.1007/s10668-023-03655-6.
- G. S. Pillai, S. Morya, W. Khalid, M. Z. Khalid, R. S. Almalki and A. Siddeeg, Banana Pseudostem: An Undiscovered Fiber Enriched Sustainable Functional Food, J. Nat. Fibers, 2024, 21(1), 1–15, DOI:10.1080/15440478.2024.2304004.
- S. Alzate Acevedo, C. Á. J. Díaz, E. Flórez-López and C. D. Grande-Tovar, Recovery of Banana Waste-Loss from Production and Processing: A Contribution to a Circular Economy, Molecules, 2021, 26, 5296 CrossRef PubMed.
- L. Al-Mqbali and M. Hossain, Cytotoxic and antimicrobial potential of different varieties of ripe banana used traditionally to treat ulcers, Toxicol Rep, 2019, 6, xxx, DOI:10.1016/j.toxrep.2019.10.003.
- A. Subagyo and A. Chafidz, Banana pseudo-stem fiber: preparation, characteristics, and applications, in Lignocellulosic Fibers and Wood Handbook, IntechOpen, London, 2018, pp. 1–16, DOI:10.5772/intechopen.82204.
- S. Balda, A. Sharma, N. Capalash and P. Sharma, Banana fibre: a natural and sustainable bioresource for eco-friendly applications, Clean Technol. Environ. Policy, 2021, 23, 1–9, DOI:10.1007/s10098-021-02041-y.
- J. Bhaskar, M. Shobha, K. Sambaiah and V. Salimath, Beneficial effects of banana (Musa sp. var. elakki bale) flower and pseudostem on hyperglycemia and advanced glycation end-products (AGEs) in streptozotocin-induced diabetic rats, J. Physiol. Biochem., 2011, 67, 415–425, DOI:10.1007/s13105-011-0091-5.
- R. Ramu, P. S. Shirahatti, K. R. Anilakumar, S. Nayakavadi, F. Zameer, B. L. Dhananjaya and M. N. Nagendra Prasad, Assessment of nutritional quality and global antioxidant response of banana (Musa sp. cv. Nanjangud Rasa Bale) pseudostem and flower, Pharmacogn. Res., 2017, 9(suppl. 1), S74–S83, DOI:10.4103/pr.pr_67_17.
- T. M. Emam, A. E. M. Hosni, A. Ismail, R. G. El-Kinany, M. Hewidy, H. S. Saudy, M. M. A. Omar, M. T. S. Ibrahim, S. Sui and S. M. El-Sayed, Physiological and molecular responses of red amaranth (Amaranthus cruentus L.) and green amaranth (Amaranthus hypochondriacus L.) to salt stress, J. Soil Sci. Plant Nutr., 2025, 25, 171–182, DOI:10.1007/s42729-024-02125-w.
- A. Bamisaye, I. Rapheal, K. A. Adegoke, I. Adegoke, M. P. Bamidele and Y. Alli, et al., Amaranthus hybridus waste solid biofuel: comparative and machine learning studies, RSC Adv., 2024, 14, 11541–11556, 10.1039/d3ra08378k.
- U. Sarker, S. Oba and M. A. Daramy, Nutrients minerals antioxidant pigments and phytochemicals, and antioxidant capacity of the leaves of stem amaranth, Sci. Rep., 2020, 1–9, DOI:10.1038/s41598-020-60252-7.
- U. Sarker, Nutritional and bioactive constituents and scavenging capacity of radicals in Amaranthus hypochondriacus, Sci. Rep., 2020,(0123456789), 1–10, DOI:10.1038/s41598-020-71714-3.
- Q. Sun, Y. Yang, H. Chen, S. Zhou, S. Zhao and W. Chen, Effect of dietary supplementation with different proportions of Amaranthus hypochondriacus stem and leaf powder on intestinal digestive enzyme activities, volatile fatty acids and microbiota of broiler chickens., Fermentation, 2024, 10, 1–14, DOI:10.3390/fermentation10100511.
- S. Maqsood, W. Khalid, P. Kumar, I. E. Benmebarek, I. Faiz Ul Rasool and M. Trif, et al., Valorization of plant-based agro-industrial waste and by-product for the production of polysaccharide: Towards a more circular economy, Appl. Food Res., 2025, 5, 100954, DOI:10.1016/j.afres.2025.100954.
- S. Bairagi, W. Ali and D. Bhattacharyya, Valorization of agricultural wastes: An approach to impart environmental friendliness, in Handbook of Biomass Valorization for Industrial Applications, Wiley, Scrivener Publishing, Hoboken, NJ, 2022, pp. 369–393, DOI:10.1002/9781119818816.ch16.
- A. A. Morales-Tapia, C. A. Ortiz-Sánchez, D. Ojeda Juarez and J. A. Del Ángel-Zumaya, Application of sugarcane bagasse as potentially functional ingredient in cookies formulation: A study on physicochemical, sensory, and texture profile properties, Rev. Mex. Ing. Quim., 2023, 22, 238–249, DOI:10.24275/rmiq/Alim238.
- A. Licona-Aguilar, J. A. Lois-Correa, A. Torres-Huerta, M. A. Domínguez-Crespo, V. Urdapilleta-Inchaurregui, A. Rodriguez and B. Brachetti, Production of dietary cookies based on wheat-sugarcane bagasse: Determination of textural, proximal, sensory, physical and microbial parameters, LWT, 2023, 184, 115061, DOI:10.1016/j.lwt.2023.115061.
- J. D. D. M. Ufitikirezi, M. Filip, M. Ghorbani, T. Zoubek, P. Olsan and R. Bumbalek, et al., Agricultural Waste Valorization: Exploring Environmentally Friendly Approaches to Bioenergy Conversion, Sustainability, 2024, 16, 3617, DOI:10.3390/su16093617.
- J. M. Fuentes-Alventosa, G. Rodríguez-Gutiérrez, S. Jaramillo-Carmona, A. Espejo, R. Rodríguez Arcos and J. Fernandez-Bolanos, et al., Effect of extraction method on chemical composition and functional characteristics of high dietary fibre powder obtained from asparagus by-products, Food Chem., 2009, 113, 665–671, DOI:10.1016/j.foodchem.2008.07.075.
- F. Figuerola, M. Hurtado, A. Estévez, I. Chiffelle and F. Asenjo, Fiber concentrate from apple pomace and citrus peel as potential fiber sources for food enrichment, Food Chem., 2005, 91, 395–401, DOI:10.1016/j.foodchem.2004.04.036.
- M. Al-Farsi and C. Lee, Optimization of phenolics and dietary fibre extraction from date seeds, Food Chem., 2008, 108, 977–985, DOI:10.1016/j.foodchem.2007.12.009.
- S. Plakantonaki, I. Roussis, D. Bilalis and G. Priniotakis, Dietary fiber from plant-based food wastes: a comprehensive approach to cereal, fruit, and vegetable waste valorization, Processes, 2023, 11, 1580, DOI:10.3390/pr11051580.
- C. Bitwell, S. S. Indra, C. Luke and K. Kakoma, A review of modern and conventional extraction techniques and their applications for extracting phytochemicals from plants, Sci. Afr., 2023, 19, e01585–e01596, DOI:10.1016/j.sciaf.2023.e01585.
- P. Saini, A. Sinha and K. Prasad, Wet refining: a novel approach for modification of wheat bran fiber, Innovative Food Sci. Emerging Technol., 2023, 90, 103508, DOI:10.1016/j.ifset.2023.103508.
- F. Chemat, N. Rombaut, A. Meullemiestre, M. Turk, S. Perino and A.-S. Fabiano-Tixier, et al., Review of green food processing techniques: Preservation, transformation, and extraction, Innovative Food Sci. Emerging Technol., 2017, 41, 1–12, DOI:10.1016/j.ifset.2017.04.016.
- J. Sun, Z. Zhang, F. Xiao, W. Quanzeng and J. Zonghui, Ultrasound-assisted alkali extraction of insoluble dietary fiber from soybean residues, IOP Conf. Ser.: Mater. Sci. Eng., 2018, 392, 052005, DOI:10.1088/1757-899X/392/5/052005.
- L. Wen, Z. Zhang, M. Zhao, R. Senthamaraikannan, R. B. Padamati and D. W. Sun, et al., Green extraction of soluble dietary fibre from coffee silverskin: impact of ultrasound/microwave-assisted extraction, Int. J. Food Sci. Technol., 2020, 55(5), 2242–2250 CrossRef CAS.
- Y. Begum and S. Deka, Effect of processing on structural, thermal, and physicochemical properties of dietary fiber of culinary banana bracts, J. Food Process. Preserv., 2019, 43, e14256, DOI:10.1111/jfpp.14256.
- H. Suryanto, A. Fikri, A. Ayu, U. Yanuhar and S. Sukardi, Pulsed electric field assisted extraction of cellulose from Mendong fiber (Fimbristylis globulosa) and its characterization, J. Nat. Fibers, 2018, 15, 406–415, DOI:10.1080/15440478.2017.1330722.
- T. Tufail, H. B. U. Ain, F. Saeed, M. Nasir, S. Basharat and Mahwish, et al., A retrospective on the innovative sustainable valorization of cereal bran in the context of circular bioeconomy innovations, Sustainability, 2022, 14, 14597, DOI:10.3390/su142114597.
- F. Chemat, M. Vian, A. Fabiano-Tixier, J. Strube, L. Uhlenbrock and V. Gunjević, et al., Green extraction of natural products: origins, current status, and future challenges, TrAC, Trends Anal. Chem., 2019, 118, 248–263, DOI:10.1016/j.trac.2019.05.037.
- C. Cannavacciuolo, S. Pagliari, R. Celano, L. Campone and L. Rastrelli, Critical analysis of green extraction techniques used for botanicals: Trends, priorities, and optimization strategies—A review, TrAC, Trends Anal. Chem., 2024, 173, 117627, DOI:10.1016/j.trac.2024.117627.
- C. Samanez, M. Huamán Carrión, W. Calsina-Ponce, G. Cruz, D. Huamaní and D. Cabel-Moscoso, et al., Technological innovations and circular economy in the valorization of agri-food by-products: advances, challenges and perspectives, Foods, 2025, 14, 1950, DOI:10.3390/foods14111950.
- J. K. Yan, L. X. Wu, W. D. Cai, G. S. Xiao and Y. Duan, Subcritical water extraction-based methods affect the physicochemical and functional properties of soluble dietary fibers from wheat bran, Food Chem., 2019, 298, 124987, DOI:10.1016/j.foodchem.2019.124987.
- M. Tanaka, A. Takamizu, M. Hoshino, M. Sasaki and M. Goto, Extraction of dietary fiber from Citrus junos peel with subcritical water, Food Bioprod. Process., 2012, 90, 1–12, DOI:10.1016/j.fbp.2011.03.005.
- X. Liang and Q. Fan, Application of sub-critical water extraction in pharmaceutical industry, J. Mater. Sci. Chem. Eng., 2013, 01, 1–6, DOI:10.4236/msce.2013.15001.
- L. Rawat and T. Ghosh, Optimization of ultrasonic-assisted extraction of dietary fiber from bhimkol (Musa balbisiana) peel using central composite design: physicochemical, functional, and thermal properties, Sustainable Food Technol., 2024, 3, 204–214, 10.1039/D4FB00230J.
- A. N. Ardila, E. Arriola-Villaseñor, E. E. V. González, H. E. G. Guerrero, J. A. Hernández-Maldonado, E. Gutiérrez-Pineda and C. C. Villa, Enhanced cellulose extraction from banana pseudostem waste: a comparative analysis using chemical methods assisted by conventional and focused ultrasound, Polymers, 2024, 16, 2785, DOI:10.3390/polym16192785.
- E. Kaya, N. Tuncel and N. Tuncel, The effect of ultrasound on some properties of pulse hulls, J. Food Sci. Technol., 2017, 54, 1–10, DOI:10.1007/s13197-017-2714-5.
- T. Lippert, J. Bandelin, F. Schlederer, J. Drewes and K. Koch, Impact of ultrasound-induced cavitation on the fluid dynamics of water and sewage sludge in ultrasonic flatbed reactors, Ultrason. Sonochem., 2019, 55, 1–14, DOI:10.1016/j.ultsonch.2019.01.024.
- H. Wang, J. Ding and N.-Q. Ren, Recent advances in microwave-assisted extraction of trace organic pollutants from food and environmental samples, TrAC, Trends Anal. Chem., 2015, 75, 197–208, DOI:10.1016/j.trac.2015.05.005.
- J. Gan, Z. Huang, Y. Qiang, P. Guanyi, Y. Chen, X. Jian, N. Shao and X. Mingyoung, Microwave assisted extraction with three modifications on structural and functional properties of soluble dietary fibers from grapefruit peel, Food Hydrocolloids, 2019, 101, 197–207, DOI:10.1016/j.foodhyd.2019.105549.
- A. González Pereira, L. Cruz, L. Cassani, F. N. Rivo, C. Lourenço-Lopes and V. de Freitas, et al., Comparative study of microwave-assisted extraction and ultrasound-assisted extraction techniques (MAE vs. UAE) for the optimized production of enriched extracts in phenolic compounds of Camellia japonica var Eugenia de Montijo, Eng. Proc., 2023, 37, 14615, DOI:10.3390/ECP2023-14615.
- S. Berktaş and M. Çam, Effects of acid, alkaline and enzymatic extraction methods on functional, structural and antioxidant properties of dietary fiber fractions from quince (Cydonia oblonga Miller), Food Chem., 2024, 464, 141596, DOI:10.1016/j.foodchem.2024.141596.
- L. Gerschenson, A. M. Rojas and E. N. Fissore, Conventional and emerging extraction technologies, in Dietary Fiber: Properties, Recovery and Applications, Academic Press, 2019, pp. 199–245, DOI:10.1016/B978-0-12-816495-2.00007-1.
- J. Shaikh, S. Chakraborty, A. Odaneth and U. Annapure, A sequential approach of alkali enzymatic extraction of dietary fiber from rice bran: Effects on structural, thermal, crystalline properties, and food application, Food Res. Int., 2024, 193, 114847, DOI:10.1016/j.foodres.2024.114847.
- Q. Ma, Z. Ma, W. Wang, J. Mu, Y. Liu, J. Wang, L. Stipkovits, S. Hui, G. Wu and J. Sun, The effects of enzymatic modification on the functional ingredient - Dietary fiber extracted from potato residue, LWT, 2021, 153, 112511, DOI:10.1016/j.lwt.2021.112511.
- V. Disca, Y. Jaouhari, F. Carrà, M. Martoccia, F. Travaglia, M. Locatelli, M. Bordiga and M. Arlorio, Effect of carbohydrase treatment on the dietary fibers and bioactive compounds of cocoa bean shells (CBSs), Foods, 2024, 13, 2545, DOI:10.3390/foods13162545.
- N. U. Sruthi, P. S. Rao, S. J. Bennett and R. R. Bhattarai, Formulation of a synergistic enzyme cocktail for controlled degradation of sorghum grain pericarp, Foods, 2023, 12(2), 306, DOI:10.3390/foods12020306.
- R. Fan, L. Wang, J. Fan, W. Sun and H. Dong, The pulsed electric field assisted-extraction enhanced the yield and the physicochemical properties of soluble dietary fiber from orange peel, Front. Nutr., 2022, 9, 925642, DOI:10.3389/fnut.2022.925642.
- G. Pataro and G. Ferrari, Limitations of pulsed electric field utilization in food industry, in Pulsed Electric Fields Technology for the Food Industry, Academic Press, 1st edn, 2020, pp. 283–310, DOI:10.1016/B978-0-12-816402-0.00013-6.
- L. Huang, X. Ding, Y. Zhao, Y. Li and H. Ma, Modification of insoluble dietary fiber from garlic straw with ultrasonic treatment, J. Food Process. Preserv., 2017, 42, e13399, DOI:10.1111/jfpp.13399.
- P. A. Freitas, C. Gonzalez-Martinez and A. Chiralt, Applying ultrasound-assisted processing to obtain cellulose fibres from rice straw to be used as reinforcing agents, Innovative Food Sci. Emerging Technol., 2022, 76, 102932, DOI:10.1016/j.ifset.2022.102932.
- J. Xu, Y. Xiao, J. Zhang, Z. Shang, Z. Tian and X. Zhu, et al., Microwave expansion pretreatment for enhancing microwave-assisted alkaline extraction of hemicellulose from bagasse, Biomass Convers. Biorefin., 2022, 14, 1–13, DOI:10.1007/s13399-022-03220-7.
- D. I. L. Gil-López, J. A. Lois-Correa, M. E. S. Pardo, M. A. Domínguez-Crespo, A. Torres-Huerta, A. Rodriguez and V. N. Orta-Guzmán, Data supporting the production of dietary fibers from sugarcane bagasse and sugarcane tops using microwave-assisted alkaline treatments, Data Brief, 2019, 24, 104026, DOI:10.1016/j.dib.2019.104026.
- N. H. Hoang, H. Do, T. Dang, M. N. Ton, T. T. T. Tran and V. Le, Fiber-enriched biscuits prepared with enzyme-treated corncob powder: nutritional composition, physical properties, and sensory acceptability, J. Food Process. Preserv., 2022, 46, e16784, DOI:10.1111/jfpp.16784.
- S. Park and K. Yoon, Enzymatic production of soluble dietary fiber from the cellulose fraction of Chinese cabbage waste and potential use as a functional food source, Food Sci. Biotechnol., 2015, 24, 529–535, DOI:10.1007/s10068-015-0069-0.
- P. A. V. Freitas, L. G. Santana, C. González-Martínez and A. Chiralt, Combining subcritical water extraction and bleaching with hydrogen peroxide to obtain cellulose fibres from rice straw, Carbohydr. Polym. Technol. Appl., 2024, 7, 1–12, DOI:10.2139/ssrn.4707778.
- L. Wang, H. M. Liu, A. Xie, C. Y. Zhu and G. Y. Qin, Dietary fiber extraction from defatted corn hull by hot-compressed water, Pol. J. Food Nutr. Sci., 2017, 68(2), 133–140, DOI:10.1515/pjfns-2017-0015.
- M. Razavi and M. Tahmasebi, Methods in the extraction of dietary fiber from banana pseudostem, in Dietary Fiber: Sources, Extraction, and Applications, Springer, Cham, 2025, pp. 1–23, DOI:10.1007/978-1-0716-4490-4_1.
- M. Usman, M. Nakagawa and S. Cheng, Emerging trends in green extraction techniques for bioactive natural products, Processes, 2023, 11, 3444, DOI:10.3390/pr11123444.
- V. Disca, Y. Jaouhari, F. Carrà, M. Martoccia, F. Travaglia, M. Locatelli, M. Bordiga and M. Arlorio, Effect of carbohydrase treatment on the dietary fibers and bioactive compounds of cocoa bean shells (CBSs), Foods, 2024, 13(16), 2545, DOI:10.3390/foods13162545.
- E. Díaz-de-Cerio and E. Trigueros, Evaluating the sustainability of emerging extraction technologies for valorization of food waste: microwave, ultrasound, enzyme-assisted, and supercritical fluid extraction, Agriculture, 2025, 15, 2100, DOI:10.3390/agriculture15192100.
- S. Das, S. S. Nadar and V. K. Rathod, Integrated strategies for enzyme assisted extraction of bioactive molecules: a review, Int. J. Biol. Macromol., 2021, 191, 899–917, DOI:10.1016/j.ijbiomac.2021.09.060.
- M. M. J. Anwar, Advances in green technologies for bioactive extraction and valorization of agro-waste in food and nutraceutical industries, Haya, 2025, 10, 184–195, DOI:10.36348/sjls.2025.v10i05.005.
- G. Landi, M. Benedetti, E. Eslami, G. Ferrari and G. Pataro, Cost, energy efficiency, and environmental impact analysis of orange juice pasteurization: comparing pulsed electric fields with traditional thermal treatment, in Proceedings of the 2024 IEEE EEEIC/ICPSEurope, 2024, vol. 17, pp. 1–6, DOI:10.1109/EEEIC/ICPSEurope61470.2024.10751382.
- G. S. M. Allam, A. M. A. El-Aal, M. K. S. Morsi and E. A. S. E. Mohamed, Producing dietary fibers from sugarcane bagasse using various chemical treatments and evaluation of their physicochemical, structural, and functional properties, Appl. Sci. Eng. Prog., 2024, 17(3), 7381, DOI:10.14416/j.asep.2024.06.002.
- L.-H. Ho, A. S. Maizatul, R. A. Zaizuliana and A. Fazilah, Physicochemical, functional, pasting and thermal properties of resistant starch type III from banana (Musa acuminata × balbisiana cv. Awak) pseudostem, Food Res., 2025, 9(2), 222–232, DOI:10.26656/fr.2017.9(2).273.
- H. W. Kim, D. Setyabrata, Y. J. Lee and Y. Kim, Efficacy of alkali-treated sugarcane fiber for improving physicochemical and textural properties of meat emulsions with different fat levels, Korean J. Food Sci. Anim. Resour., 2018, 38(2), 315–324, DOI:10.5851/kosfa.2018.38.2.315.
- P. Bhattacharyya, M. Das, A. Saikia and K. K. Dash, Physicochemical and functional properties of dietary fibres extracted from the pseudostem of Musa balbisiana Colla, Ann. Phytomed., 2024, 13(1), 931–940, DOI:10.54085/ap.2024.13.1.100.
- Y. Zhou, Q. Sun, C. Teng, M. Zhou, G. Fan and P. Qu, Preparation and improvement of physicochemical and functional properties of dietary fiber from corn cob fermented by Aspergillus niger, J. Microbiol. Biotechnol., 2024, 34(2), 330–339 CrossRef CAS PubMed.
- R. Nordin, M. I. Mazlan, A. Z. Mohsin and I. N. Madzuki, Characterization of dietary fibers from corn cobs as a potential functional ingredient in muffin, MAB J., 2025, 54(1), 64–75, DOI:10.55230/mabjournal.v54i1.3176.
- X. Meng, C. Wu, H. Liu, Q. Tang and X. Nie, Dietary fibers fractionated from gardenia (Gardenia jasminoides Ellis) husk: structure and in vitro hypoglycemic effect, J. Sci. Food Agric., 2021, 101(9), 3723–3731, DOI:10.1002/jsfa.11003.
- H. Norhayati, S. Mw and A. Najjah, Characterization of Three Different Types of Pineapple Waste Powder, Food Nutr. Curr. Res., 2020, 3(1), 271–279 Search PubMed . Available from: https://www.scitcentral.com/.
- H. K. A. H. Gamage, S. G. Tetu, R. W. W. Chong, D. Bucio-Noble, C. P. Rosewarne, L. Kautto, M. S. Ball, M. P. Molloy, N. H. Packer and I. T. Paulsen, Fiber supplements derived from sugarcane stem, wheat dextrin and psyllium husk have different in vitro effects on the human gut microbiota, Front. Microbiol., 2018, 9, 1618, DOI:10.3389/fmicb.2018.01618.
- C. Chong, M. Liew, W. Ooi, H. Jamil, A. Lim, S. Hooi, C. Tay and G. Tan, Effect of green banana and pineapple fibre powder consumption on host gut microbiome, Front. Nutr., 2024, 11, 1437645, DOI:10.3389/fnut.2024.1437645.
- S. W. Pelpolage, Y. Goto, R. Nagata, N. Fukuma, T. Furuta, M. Mizu, K. H. Han and M. Fukushima, Colonic fermentation of water soluble fiber fraction extracted from sugarcane (Saccharum officinarum L.) bagasse in murine models, Food Chem., 2019, 292, 336–345, DOI:10.1016/j.foodchem.2019.04.063.
- D. A. Campos, E. R. Coscueta, A. A. Vilas-Boas, S. Silva, J. A. Teixeira and L. M. Pastrana, et al., Impact of functional flours from pineapple by-products on human intestinal microbiota, J. Funct. Foods, 2020, 67, 103830, DOI:10.1016/j.jff.2020.103830.
- Y. P. Silva, A. Bernardi and R. L. Frozza, The role of short-chain fatty acids from gut microbiota in gut-brain communication, Front. Endocrinol., 2020, 11, 25, DOI:10.3389/fendo.2020.00025.
- I. Kimura, D. Inoue, T. Maeda, T. Hara, A. Ichimura and S. Miyauchi, et al., Short-chain fatty acids and ketones directly regulate sympathetic nervous system via G protein-coupled receptor 41 (GPR41), Proc. Natl. Acad. Sci. U. S. A., 2011, 108(19), 8030–8035, DOI:10.1073/pnas.1016088108.
- S. Facchin, L. Bertin, E. Bonazzi, G. Lorenzon, C. De Barba, B. Barberio, F. Zingone, D. Maniero, M. Scarpa and C. Ruffolo, et al., Short-chain fatty acids and human health: from metabolic pathways to current therapeutic implications, Life, 2024, 14, 559, DOI:10.3390/life14050559.
- L. Yoon and K. Michels, Characterizing the effects of calcium and prebiotic fiber on human gut microbiota composition and function using a randomized crossover design—a feasibility study, Nutrients, 2021, 13, 1937, DOI:10.3390/nu13061937.
- A. H. T. Hefnawy, A. E. Awad, K. M. Wahdan and M. A. Doheim, Chemical and functional properties of the native banana (Musa) pseudo-stem, Zagazig J. Agric. Res., 2022, 49(6), 841–852, DOI:10.21608/zjar.2022.278247.
- E. B. Giuntini, F. A. H. Sardá and E. W. de Menezes, The effects of soluble dietary fibers on glycemic response: an overview and future perspectives, Foods, 2022, 11, 3934, DOI:10.3390/foods11233934.
- A. Karthiayani, M. Ganga and C. Kumar, Fiber enrichment in noodles using banana pseudo-stem (Musa paradisiaca), Asian J. Dairy Food Res., 2021, 1–13, DOI:10.18805/ajdfr.DR-1571.
- K. Ojha and A. Verma, Physico-chemical parameters and sensory characteristics of biscuits incorporated with sugarcane bagasse powder, Pharma Innovation, 2023, 12, 1491–1493, DOI:10.5281/zenodo.8221994.
- H. D. Holscher, Dietary fiber and prebiotics and the gastrointestinal microbiota, Gut Microbes, 2017, 8(2), 172–184, DOI:10.1080/19490976.2017.1290756.
- M. Józó, R. Várdai, A. Bartos, J. Móczó and B. Pukánszky, Preparation of biocomposites with natural reinforcements: the effect of native starch and sugarcane bagasse fibers, Molecules, 2022, 27, 6423, DOI:10.3390/molecules27196423.
- T. B. Shabangu, L. Z. Linganiso, M. Tshwafo and S. Motloung, Comparison of fibre from maize stalk domains and sugar cane bagasse, Drev. Vysk., 2019, 64(4), 601–612 CAS.
- I. Buljeta, D. Šubarić, J. Babic and A. Pichler, Extraction of dietary fibers from plant-based industry waste: a comprehensive review, Appl. Sci., 2023,(16), 9309, DOI:10.3390/app13169309.
- F. Lyu, M. Thomas, W. H. Hendriks and A. F. B. Poel, Size reduction in feed technology and methods for determining, expressing and predicting particle size: A review, Anim. Feed Sci. Technol., 2019, 261, 114347, DOI:10.1016/j.anifeedsci.2019.114347.
- L. H. Ho, N. A. Abdul Aziz, R. Bhat and B. Azahari, Storage studies of bread prepared by incorporation of the banana pseudo-stem flour and the composite breads containing hydrocolloids, CyTA – J. Food, 2014,(2), 141–149, DOI:10.1080/19476337.2013.806597.
- T. Lau, T. Clayton, N. Harbourne, J. Rodriguez-Garcia and M. J. Oruna-Concha, Sweet corn cob as a functional ingredient in bakery products, Food Chem.: X, 2022, 13, 100180, DOI:10.1016/j.fochx.2021.100180.
- H. Munir, H. Alam, M. T. Nadeem and R. S. Almalki, Green banana resistant starch: a promising potential as functional ingredient against certain maladies, Food Sci. Nutr., 2024, 12(6), 1–14, DOI:10.1002/fsn3.4063.
- D. E. Carballo, I. Caro, C. Gallego and A. R. González, Banana pseudo-stem increases the water-holding capacity of minced pork batter and the oxidative stability of pork patties, Foods, 2021,(9), 2173, DOI:10.3390/foods10092173.
- N. E. Kumar and F. Ahmad, Effect incorporation of corn cob powder on the physicochemical properties and sensory acceptability of Kuih Kaswi, J. Biochem. Microbiol. Biotechnol., 2024, 12(SP1), 25–26, DOI:10.54987/jobimb.v12iSP1.929.
- R. Chakraborty, S. Sabruna, R. Roy, S. Majumdar and S. Roy, Banana pseudostem substitution in wheat flour biscuits enriches the nutritional and antioxidative properties with considerable acceptability, SN Appl. Sci., 2021, 3(1), 1–12, DOI:10.1007/s42452-020-03988-1.
- K. S. Kaaviya, R. Yuvaraj, S. Keerthika, H. Karuppasamy, R. Brimapureeswaran and U. Antony, Development of ready to drink banana pseudostem beverage incorporated with pineapple and grape extract, Asian J. Dairy Food Res., 2024, 44(4), 1–8, DOI:10.18805/ajdfr.DR-2063.
- M. Wójcik, A. Matwijczuk, L. Ślusarczyk and R. Różyło, The use of bamboo fiber in the production of high-fiber, low-carbohydrate, and gluten-free bread: physicochemical and spectroscopic properties, Processes, 2025, 83, 1–13, DOI:10.3390/pr13010083.
- J. Zheng, J. Wu, Y. Dai and J. Kan, Influence of bamboo shoot dietary fiber on the rheological and textural properties of milk pudding, LWT, 2017, 84, 1–12, DOI:10.1016/j.lwt.2017.05.051.
- V. Liani and A. K. Dhiman, Sensory parameters of bamboo shoot powder fortified yoghurt, Int. J. Curr. Microbiol. Appl. Sci., 2020,(4), 6–11, DOI:10.20546/ijcmas.2020.904.002.
- I. M. C. Magalhães, C. D. S. Paglarini, V. A. S. Vidal and M. A. R. Pollonio, Bamboo fiber improves the functional properties of reduced salt and phosphate free Bologna sausage, J. Food Process. Preserv., 2020, 44(12), e14929, DOI:10.1111/jfpp.14929.
- N. Derkanosova, A. Stakhurlova, I. Pshenichnaya and I. Ponomarev, Amaranth as a bread enriching ingredient, Foods Raw Mater., 2020, 8(2), 223–231, DOI:10.21603/2308-4057-2020-2-223-231.
- T. Belwal, F. Chemat, R. Venskutonis, G. Cravotto, D. Jaiswal, I. Bhatt, H. Devkota and Z. Luo, Recent advances in scaling-up of non-conventional extraction techniques: learning from successes and failures, TrAC, Trends Anal. Chem., 2020, 12, 115895, DOI:10.1016/j.trac.2020.115895.
- A. Desye, D. Teferi, M. Geremew and D. Kassa, Biorefinery and valorization strategies for sugarcane bagasse: integrating food, health, economic, and industrial applications, Food Sci. Nutr., 2025,(12), 1–12, DOI:10.1002/fsn3.71262.
| This journal is © The Royal Society of Chemistry 2026 |