Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Comparative effects of pretreatments and their combinations on uncooked and microwave-assisted cooked lentil (Lens culinaris) flours: effect on technofunctional, antinutritional, bioactive, and structural properties
Kathika Dasab,
Shubham Mandliya *bc and
Pradyuman Kumara
*bc and
Pradyuman Kumara
aDepartment of Food Engineering and Technology, Sant Longowal Institute of Engineering and Technology, Longowal, Punjab, India
bAgricultural and Food Engineering Department, Indian Institute of Technology Kharagpur, Kharagpur, West Bengal, India. E-mail: shubham.mandliya15@gmail.com; s.mandliya@reading.ac.uk
cDepartment of Food and Nutritional Sciences, University of Reading, Reading, England, UK
First published on 21st April 2026
Abstract
This study focused on the nutritional, technofunctional, bioactive, and microstructural properties of lentils obtained with different pretreatments and processing methods, including soaking, germination, dehulling, microwave-assisted cooking (MAC), and their combinations, to produce value-added lentil flour with improved characteristics. The combination of dehulling and germination resulted in a significant increase in protein content (p < 0.05), with the highest value of 30.91%. Similarly, crude fiber content increased due to the combined effects of germination and MAC (3.62%). In contrast, fat content showed a decreasing trend. Dehulling resulted in maximum reduction of tannin content (85.57%), phytic acid was most effectively reduced by germination (73.14%), and the trypsin inhibitor was mainly reduced by MAC (93.81%). Antioxidant activity was highest in the untreated sample (35.37%) and decreased further after subsequent pretreatments; a similar reduction was observed for TPC. Soaking dehulled lentil flour resulted in the highest peak and final viscosities (1366 cP; 1800 cP). MAC can improve both the technofunctional and nutritional properties of lentil flour, and a combination approach can enhance lentil value and diversify its use.
Sustainability spotlightWith the rising demand for sustainable and clean-label food solutions, lentils offer a promising foundation for developing climate-smart, plant-based ingredients. This research offers a sustainable enhancement of lentil flour utilizing low-input pre-treatments such as dehulling, soaking, and germination, with microwave-assisted cooking, an energy-efficient processing technology. With these techniques, this study resulted lentil flours with reduced antinutritional factors, and enhanced functional properties, improving both nutritional value and processing versatility. This work aligns well with several SDGs (2: zero hunger, 3: good health and well-being, 7: affordable and clean energy, 12: responsible consumption and production, and 13: climate action). |
1. Introduction
Lentils (Lens culinaris) are increasingly recognized internationally as a sustainable source of protein owing to their minimal environmental impact, relatively modest water needs, and capacity to enhance soil health.1 They rank among the most economical vegetarian protein sources, and their escalating application in plant-based and functional food products reflects a rising demand for sustainable, transparent, and nutrient-dense ingredients. However, integrating lentils into plant-based foods often poses challenges, including longer cooking times, which can diminish their nutritional value and require greater energy consumption. Moreover, lentils contain significant levels of antinutrients, which reduce their bioavailability and thereby limit their utilization.2 Effectively reducing these antinutritional components while retaining nutritional quality remains a vital obstacle in lentil processing.To address these challenges, various traditional and thermal treatments used in industry, such as soaking, germination, dehulling, and cooking, may be utilized. Soaking is a simple, industry-aligned first step that hydrates lentils, leaches water-soluble antinutrients into soaked water, and reduces cooking time. Studies on red and green lentils reported reductions in phytic acid and trypsin of 14.5–43.8% and 58.2–80.1%, respectively, following soaking and drying.3 Germination effectively reduces antinutrients (phytates, tannins, etc.) by activating endogenous phytases and proteases, while improving vitamin and antioxidant profiles and often enhancing technofunctional properties (hydration and emulsification).4 Dehulling removes the seed coat, thereby decreasing phytic acid, condensed tannins, and polyphenols, thus increasing bioavailability despite a reduction in polyphenols or minerals.5
Dehulling, soaking, and germination are widely used household- and industry-scale processing methods. After pretreatments, cooking is done either traditionally using thermal methods (most commonly used in households and industries) or using alternative thermal technologies. Traditional cooking significantly reduces antinutritional factors in legumes and plant-based foods.6 However, this method might be energy-intensive, require a longer duration, lead to higher leaching losses, degrade heat-sensitive qualities, and decrease levels or bioavailability of some bioactive compounds.7 These limitations have prompted the exploration of alternative thermal technologies, notably microwave technology.
Microwave processing has opened a range of opportunities for the food industry, offering time and energy savings and less degradation in the nutritional quality of foods without compromising safety.8 Microwave heating relies on the specific characteristics of the material, including electrical conductivity, moisture content, and dielectric properties, to directly heat the product by generating heat within it.7,8 The heating effect, therefore, arises from friction between molecules, which occurs through ionic conduction and dipolar rotation, thereby absorbing electrical energy from the electromagnetic field.9 In contrast to conventional boiling, which leads to significant nutrient loss, microwave cooking expedites cooking and preserves the optimal nutritional content of lentils.10 A comparison of microwave, pressure, and conventional cooking of lentil and other legume pastes found that microwave cooking yielded the highest soluble solids, improved protein digestibility, and beneficial changes in protein and bioactive release, highlighting the potential of microwave processing to enhance nutrient accessibility in legumes.11 In a separate investigation of rice, microwave heating produced results comparable to those of traditional cooking with shorter time and less water usage.12 Contemporary research indicates that microwave-assisted cooking (MAC) facilitates rapid volumetric heating, thereby supporting various applications including blanching, drying, sterilization, and the functional modification of food matrices. This method effectively preserves color, flavor, and vitamins compared with cooking/boiling.7,8,13–15 Moreover, microwave-induced structural modifications in proteins, starches, and lipids can enhance the technofunctional properties of processed foods. These benefits position MAC as a thermally based method that aligns with sustainability considerations and offers nutritional advantages compared to traditional cooking techniques.
Existing research has investigated pretreatment strategies, including dehulling, germination, and thermal processing, to improve the palatability and nutrient bioavailability of pulses and their effects on compositional and functional attributes.4,6 Although soaking, germination, dehulling, and cooking have each been shown to reduce antinutritional factors and improve the nutritional quality of lentils and related legumes, most studies evaluate these treatments independently or under conventional heating conditions. Consequently, comparative and systematic assessments of combined pretreatments, particularly regarding their technofunctional behavior, antinutrient reduction, and microstructural transformations, remain limited, and research integrating these pretreatments with MAC is notably scarce. Given their feasibility and scalability within pulse-ingredient processing, these pretreatments were selected to address this gap. Therefore, the present study investigates the effect of dehulling, soaking, and germination, applied individually and in combination using a general full factorial design, on the technofunctional, bioactive, antinutritional, and microstructural properties of lentil flours under both raw and microwave-cooked conditions.
2. Materials and methods
2.1. Materials
Red lentils (Lens culinaris) of the PL8 variety, cultivated in Punjab (India), were obtained from the Longowal village market within 2 months of harvest, with a thousand-kernel weight of 41.5 ± 3.45 g. After being cleansed and freed from dust and other foreign matter, the seeds were stored in zip-lock bags, sealed, and kept at room temperature until further use. All the reagents and chemicals used were of analytical reagent grade.2.2. Preparation of lentil flour
The lentil seeds were pretreated with dehulling, soaking, and germination, individually and in combination (soaking + dehulling and germination + dehulling), followed by drying and milling for all samples. Initially, a batch of seeds (200 g) was cleaned and rinsed and then soaked at ambient temperature (22–25 °C) for 12 h with a seed mass-to-water volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
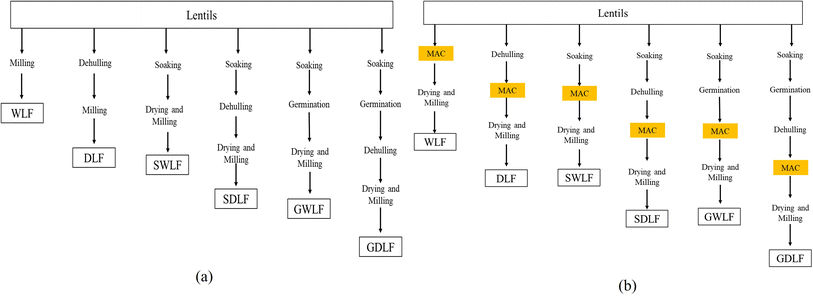
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (w/v). After soaking, the water was drained, and half of the seeds were retained as soaked samples. In contrast, the remaining half was germinated for 40 h at 27 °C in an environmental chamber (MSW-125, Micro Scientific Works Pvt. Ltd, New Delhi, India), resulting in germinated samples. Germination was carried out at 92% relative humidity under dark conditions. The soaked seeds were wrapped in a clean cotton cloth, placed on a stainless-steel tray, and transferred into the environmental chamber for germination. After soaking and germination, half of the seeds were manually dehulled with hands to avoid cotyledon damage and the other half were left with their hulls, yielding whole and dehulled samples. All six samples after the desired pretreatments were dried in a hot-air oven (IG-50HAO, iGene Labserve Pvt. Ltd, New Delhi, India) at 45 ± 2 °C until a final moisture content of 10% was achieved. Moisture content was monitored using the AOAC oven-drying method (AOAC 925.10); drying time (12–14 h) was varied until the final moisture content was reached.16 The drying process was followed by milling (Supermix, Sujata Appliances, Mittal Electronics, New Delhi, India), sieving through a 150 µm sieve to obtain a standardized sieve-classified fraction with an upper particle size limit of 150 µm, and storing the powder in airtight pouches. The particle size distribution analysis was not performed, and thus, the actual particle size of the flour is not known, which is a limitation of this study. The samples were denoted as WLF (whole lentil flour), DLF (dehulled lentil flour), SWLF (soaked whole lentil flour), SDLF (soaked dehulled lentil flour), GWLF (germinated whole lentil flour), and GDLF (germinated dehulled lentil flour) (Fig. 1a).
4 (w/v). After soaking, the water was drained, and half of the seeds were retained as soaked samples. In contrast, the remaining half was germinated for 40 h at 27 °C in an environmental chamber (MSW-125, Micro Scientific Works Pvt. Ltd, New Delhi, India), resulting in germinated samples. Germination was carried out at 92% relative humidity under dark conditions. The soaked seeds were wrapped in a clean cotton cloth, placed on a stainless-steel tray, and transferred into the environmental chamber for germination. After soaking and germination, half of the seeds were manually dehulled with hands to avoid cotyledon damage and the other half were left with their hulls, yielding whole and dehulled samples. All six samples after the desired pretreatments were dried in a hot-air oven (IG-50HAO, iGene Labserve Pvt. Ltd, New Delhi, India) at 45 ± 2 °C until a final moisture content of 10% was achieved. Moisture content was monitored using the AOAC oven-drying method (AOAC 925.10); drying time (12–14 h) was varied until the final moisture content was reached.16 The drying process was followed by milling (Supermix, Sujata Appliances, Mittal Electronics, New Delhi, India), sieving through a 150 µm sieve to obtain a standardized sieve-classified fraction with an upper particle size limit of 150 µm, and storing the powder in airtight pouches. The particle size distribution analysis was not performed, and thus, the actual particle size of the flour is not known, which is a limitation of this study. The samples were denoted as WLF (whole lentil flour), DLF (dehulled lentil flour), SWLF (soaked whole lentil flour), SDLF (soaked dehulled lentil flour), GWLF (germinated whole lentil flour), and GDLF (germinated dehulled lentil flour) (Fig. 1a).
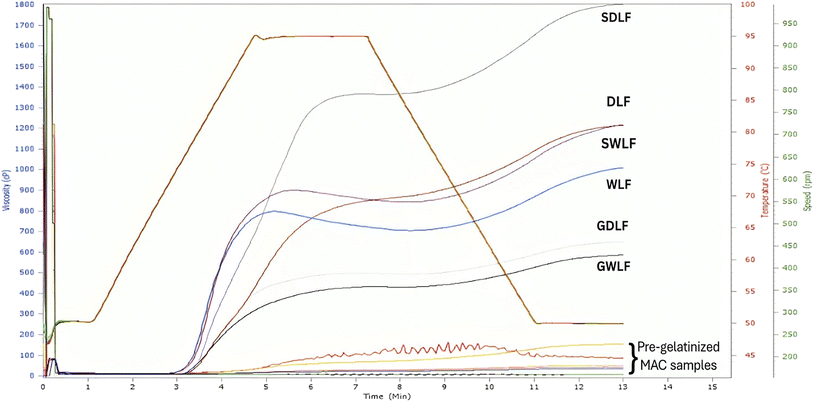
Another set of six samples was prepared in the same way, with an additional MAC step. A detailed step-by-step process is shown in Fig. 1b. Fifty grams of each sample (seeds) were transferred into a 500 mL microwave-safe glass beaker, with an internal diameter of 87 mm and a height of 127 mm, and supplemented with distilled water (at room temperature) at a seed mass-to-water volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (w/v). The cooking process was performed at 800 W in a microwave oven (MS23K3513AK/T, Samsung, Malaysia), with the beaker kept open and not stirred until cooking was complete. The cooking endpoint was identified by the same trained operator throughout the study, using a standardized method.17 Accurately, 10 grains were tested every minute by pressing them between the fingers, and the endpoint was defined as the point at which no hard core remained after 30 s of cooling. The cooking time was 13 minutes. However, this method relies on sensory endpoint assessment and was not validated using instrumental methods (e.g., texture analysis), which is a limitation. After treatment, the cooking liquor was discarded, and the respective samples were cooled, dried, ground, sieved, and stored for further analysis.
4 (w/v). The cooking process was performed at 800 W in a microwave oven (MS23K3513AK/T, Samsung, Malaysia), with the beaker kept open and not stirred until cooking was complete. The cooking endpoint was identified by the same trained operator throughout the study, using a standardized method.17 Accurately, 10 grains were tested every minute by pressing them between the fingers, and the endpoint was defined as the point at which no hard core remained after 30 s of cooling. The cooking time was 13 minutes. However, this method relies on sensory endpoint assessment and was not validated using instrumental methods (e.g., texture analysis), which is a limitation. After treatment, the cooking liquor was discarded, and the respective samples were cooled, dried, ground, sieved, and stored for further analysis.
2.3. Proximate composition
The moisture content of the lentil flour samples was determined by oven drying in accordance with AOAC 925.10. Nitrogen content was analyzed utilizing the Kjeldahl method (AOAC 920.87), and protein content was computed by multiplying the nitrogen value by a conversion factor of 6.25. Ash content was also measured using a muffle furnace according to the procedures outlined by AOAC (AOAC 923.03).16 Crude fat content was estimated by Soxhlet extraction, as specified in the AOAC guidelines.18 Finally, the carbohydrate (CHO, %) content was calculated as the difference: carbohydrate% = [100 − moisture% − protein% − fat% − ash% − crude fiber%].192.4. Technofunctional properties
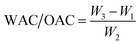 | (1) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v water
1 v/v water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) oil), prepared by blending distilled water with refined rice bran oil. The resulting mixture was homogenized using a homogenizer (Model MT-30K, Moxcare, India) at 10
oil), prepared by blending distilled water with refined rice bran oil. The resulting mixture was homogenized using a homogenizer (Model MT-30K, Moxcare, India) at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for one min; subsequently centrifugation was carried out at 6000 rpm (4025 × g) for 30 min using a refrigerated centrifuge. All the measurements were taken immediately after centrifugation. The volume of the emulsified layer was measured to facilitate the calculation of EC (%), defined as the height of the emulsified layer divided by the total height (eqn (2)).
000 rpm for one min; subsequently centrifugation was carried out at 6000 rpm (4025 × g) for 30 min using a refrigerated centrifuge. All the measurements were taken immediately after centrifugation. The volume of the emulsified layer was measured to facilitate the calculation of EC (%), defined as the height of the emulsified layer divided by the total height (eqn (2)).
 | (2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 rpm for two min at ambient temperature (25 ± 2 °C). The height of the resulting foam was measured immediately by pouring the sample into a graduated cylinder and visually estimating its volume. FC was expressed as the percentage increase in volume due to foam formation, as defined in eqn (3).
500 rpm for two min at ambient temperature (25 ± 2 °C). The height of the resulting foam was measured immediately by pouring the sample into a graduated cylinder and visually estimating its volume. FC was expressed as the percentage increase in volume due to foam formation, as defined in eqn (3).
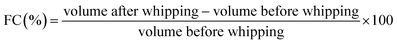 | (3) |
2.5. Antinutrients and bioactives
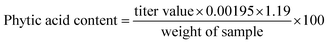 | (4) |
The antioxidant activity (AOX) of lentil samples was measured using the DPPH assay, as described by Pathiraja et al., with minor modifications.26 Briefly, 24 mg of DPPH were dissolved in 100 mL of methanol to create a methanol stock solution, which was then diluted with methanol. An aliquot of 100 µL of each sample was mixed with 3 mL of DPPH stock solution, shaken well, and incubated in the dark for 30 min. A blank was then prepared without extract and measured at 517 nm. The % DPPH inhibition was calculated using eqn (5).
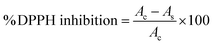 | (5) |
2.6. Pasting properties
The pasting profile was determined with a Rapid Visco Analyzer (RVA) (TecMaster, Warriewood, Australia) as described by Vishwakarma et al.25 A designated heating and cooling cycle was programmed, involving a 3 g sample with distilled water added to attain the appropriate moisture content for viscosity profile generation. The experimental procedure involved initially holding the samples at 50 °C for 1 min, then heating to 95 °C at 12 °C min−1, and holding at 95 °C for 2.5 min. Subsequently, the samples were cooled from 95 °C to 45 °C at 12 °C min−1, followed by a 2-min holding period at 45 °C. All the samples were subject to the same procedure.2.7. Morphological analysis
The morphological characteristics of lentil flour samples were analyzed using scanning electron microscopy (SEM) (JSM-7610F Plus, JEOL, Japan) with a secondary electron (SE) detector to examine the microstructural changes under high vacuum conditions induced by different pretreatments in both raw and MAC samples. Each sample was sputter-coated with a thin layer of gold (360 Å thickness) under vacuum at room temperature before image analysis.27 A 10 kV power was used at 850× and 1000× magnifications.2.8. Statistical analysis
The experiment employed a full factorial design to assess the effects of cooking methods (2: raw and microwave-assisted) and pretreatments (5: soaking, germination, dehulling, soaking + dehulling, and germination + dehulling) on proximate, technofunctional, antinutrient, and bioactive properties. Two control samples were included – one for raw and another for MAC without any pretreatment. All experimental runs were conducted in triplicate, with the analyzed properties expressed as mean ± standard deviation across biological replicates. An analysis of variance (ANOVA) was performed to determine the statistical significance of the effects of cooking, pretreatment, or their interaction on the measured responses. The mean comparisons were performed using Tukey's HSD test to delineate differences among factor levels at the 0.05 significance level (α). All statistical analyses were conducted using OriginPro 2021 (OriginLab Corporation, Northampton, MA, USA).3. Results and discussion
3.1. Proximate composition
The proximate composition of the lentil flour samples, as presented in Table 1, reflects their nutritional attributes. The absolute protein content in raw and MAC-treated samples ranged from 26.68% to 30.91% and from 25.88% to 27.98%, respectively, demonstrating a statistically significant difference (p < 0.05). The protein content of lentils increased by both soaking and germination, which may be associated with enzyme synthesis, amino acid release, and breakdown of protein molecules into simpler peptides. Furthermore, the breakdown and utilization of carbohydrates and lipids as energy sources during germination may result in a relative increase in protein concentration when expressed on a dry-weight basis; however, a significant decrease in absolute protein content was observed after MAC, which may be attributed to the leaching out of nitrogenous compounds and soluble proteins into the cooking water.28 Additionally, prolonged heat exposure can lead to protein denaturation and aggregation, which may reduce protein content.29 Although protein losses in the cooking water or changes in protein solubility were not directly quantified in the present study, these mechanisms are widely reported and are likely contributors to the observed reduction. It was observed that dehulling, germination, and their combined treatment had the most significant impact on raw lentil flour samples, resulting in absolute protein percentages of 28.97%, 29.02%, and 30.91%, respectively.| Pretreatment | Moisture (%) | Protein (%) | Fat (%) | Crude fiber (%) | Ash (%) | Carbohydrate (%) | |
|---|---|---|---|---|---|---|---|
| a Data are presented as mean ± standard deviation. Means sharing different superscript letters within the same column differ significantly (p < 0.05) as per two-way ANOVA followed by the post hoc Tukey test. WLF – whole lentil flour, DLF – dehulled lentil flour, SWLF – soaked whole lentil flour, SDLF – soaked & dehulled lentil flour, GWLF – germinated whole lentil flour, and GDLF – germinated & dehulled lentil flour. | |||||||
| Raw | WLF | 8.33 ± 0.08fg | 26.68 ± 0.18efg | 1.38 ± 0.07ab | 1.67 ± 0.04e | 3.61 ± 0.18a | 58.33 ± 0.68ab |
| DLF | 8.49 ± 0.05efg | 28.75 ± 0.14bc | 1.52 ± 0.06a | 1.05 ± 0.27g | 3.43 ± 0.10a | 56.76 ± 0.32abc | |
| SWLF | 8.24 ± 0.03g | 27.13 ± 0.17def | 1.34 ± 0.05ab | 1.71 ± 0.05e | 3.57 ± 0.12a | 58.01 ± 0.31abc | |
| SDLF | 8.27 ± 0.05g | 28.97 ± 0.25b | 1.47 ± 0.07a | 1.13 ± 0.13fg | 3.39 ± 0.34a | 56.77 ± 0.91abc | |
| GWLF | 9.59 ± 0.13b | 29.02 ± 0.32b | 1.29 ± 0.22abc | 3.84 ± 0.33b | 3.21 ± 0.04a | 53.41 ± 1.07de | |
| GDLF | 10.04 ± 0.22a | 30.91 ± 0.19a | 1.40 ± 0.04a | 2.96 ± 0.04c | 3.18 ± 0.16a | 51.51 ± 0.89e | |
| MAC | WLF | 8.61 ± 0.07def | 25.90 ± 0.27g | 0.96 ± 0.21cd | 2.39 ± 0.20d | 3.35 ± 0.08a | 58.79 ± 0.67a |
| DLF | 8.81 ± 0.09d | 27.58 ± 0.35de | 1.03 ± 0.05bcd | 1.47 ± 0.12efg | 3.21 ± 0.30a | 57.90 ± 0.81abc | |
| SWLF | 8.69 ± 0.04de | 25.88 ± 0.27g | 0.89 ± 0.24d | 2.43 ± 0.07d | 3.21 ± 0.07a | 58.90 ± 1.17a | |
| SDLF | 8.64 ± 0.12de | 27.61 ± 0.46de | 0.97 ± 0.03cd | 1.51 ± 0.05ef | 3.17 ± 0.14a | 58.71 ± 0.51a | |
| GWLF | 9.19 ± 0.14c | 26.55 ± 0.55fg | 0.74 ± 0.05d | 4.32 ± 0.13a | 3.15 ± 0.04a | 56.05 ± 1.14bc | |
| GDLF | 8.82 ± 0.10d | 27.98 ± 0.23cd | 0.82 ± 0.04d | 3.61 ± 0.05b | 3.11 ± 0.18a | 55.66 ± 1.03cd | |
Fat content was significantly influenced by cooking treatment (MAC) and pretreatment. MAC samples exhibited lower fat levels (0.74–1.03%) than their raw counterparts (1.29–1.52%), indicating a stronger effect of MAC. Among raw samples, dehulled flours (DLF, SDLF, and GDLF) had higher fat content compared to whole lentil flours (Table 1). The observed rise in fat content subsequent to dehulling in raw flours presumably indicates a compositional redistribution following hull removal, given that lipids are predominantly accumulated within the cotyledon fraction. Germination showed a tendency toward reduced fat levels, which may indicate lipid utilization during metabolic processes; however, these differences were relatively minor compared with those observed with MAC. MAC decreased fat content by a larger margin, which may be associated with the diffusion of lipid into water during cooking.
The crude fiber content in raw and MAC samples ranged from 1.05% to 3.84% and 1.51% to 4.32%, respectively. The seed coat contains the majority of this fiber; dehulling lentils reduces fiber content in both raw and MAC samples, thereby increasing protein content.30 Both the raw and MAC lentil samples exhibited a significant (p < 0.05) increase in crude fiber following germination compared to their non-dehulled counterparts. Since crude fiber primarily comprises cellulose, lignin, and hemicelluloses, the observed increase in its content may be associated with the accumulation of dry matter as the seed undergoes germination.31 MAC samples had higher crude fiber levels than their raw counterparts. This phenomenon may be attributed to the formation of protein-fiber complexes that develop during MAC.32 The GWLF of MAC yielded the highest crude fiber content, which may be ascribed to the combined effects of fiber accumulation during germination and structural modifications induced by MAC. Furthermore, the presence of the seed coat, which was not removed in these samples, likely contributed to the increased crude fiber content.
Both raw and MAC-treated lentil samples showed a non-significant (p > 0.05) decrease in ash content. Among the treated samples, a slight reduction in ash content was observed for SWLF following MAC. This slight decrease may be attributed to the leaching of mineral components into the cooking medium during processing, a trend consistently reported in earlier studies on thermally processed legumes.4
There is a notable absolute decrease in the total carbohydrate content in raw samples following pretreatments, as shown in Table 1. The most significant reduction (p < 0.05) was observed in the germinated samples (GWLF and GDLF) for both raw and MAC samples. During germination, seeds activate enzymes that break down complex carbohydrates (starch) into simpler sugars and, during respiration, glucose is metabolized into carbon dioxide and water, leading to a loss of carbohydrate mass from the seed and an overall reduction in total carbohydrates.33 A similar downward trend was observed in MAC-treated samples. Nevertheless, an overall relative increase in total carbohydrate content was observed following MAC. This apparent increase does not indicate a gain or enrichment attributable to cooking; rather, it reflects reductions in other components (protein, fat, and ash) and leaching during MAC.
3.2. Technofunctional properties
| Pretreatment | WAC (g water per g flour) | OAC (g oil per g flour) | EC (%) | FC (%) | |
|---|---|---|---|---|---|
| a Data are presented as mean ± standard deviation. Means sharing different superscript letters within the same column differ significantly (p < 0.05) as per two-way ANOVA followed by the post hoc Tukey test. WLF – whole lentil flour, DLF – dehulled lentil flour, SWLF – soaked whole lentil flour, SDLF – soaked & dehulled lentil flour, GWLF – germinated whole lentil flour, and GDLF – germinated & dehulled lentil flour. WHC – water holding capacity, OHC – oil holding capacity, EC – emulsifying capacity, and FC – foaming capacity. | |||||
| Raw | WLF | 0.88 ± 0.08e | 0.93 ± 0.08h | 44.45 ± 0.93k | 15.06 ± 0.21f |
| DLF | 1.02 ± 0.07de | 1.31 ± 0.11g | 72.33 ± 0.69f | 17.36 ± 0.35d | |
| SWLF | 0.91 ± 0.04e | 1.12 ± 0.03h | 52.08 ± 0.92j | 15.78 ± 0.19e | |
| SDLF | 1.17 ± 0.08d | 1.33 ± 0.04g | 78.04 ± 0.90de | 18.41 ± 0.82c | |
| GWLF | 1.12 ± 0.23de | 1.81 ± 0.09e | 64.78 ± 1.12g | 19.55 ± 0.35b | |
| GDLF | 1.26 ± 0.05d | 2.13 ± 0.05d | 88.3 ± 1.24b | 21.37 ± 0.39a | |
| MAC | WLF | 1.78 ± 0.04c | 1.53 ± 0.04f | 57.88 ± 1.17i | 10.33 ± 0.61i |
| DLF | 1.82 ± 0.05c | 1.93 ± 0.03e | 78.95 ± 0.79d | 10.51 ± 0.79i | |
| SWLF | 1.92 ± 0.04c | 2.43 ± 0.04c | 61.38 ± 1.15h | 10.86 ± 1.15hi | |
| SDLF | 2.23 ± 0.08b | 2.53 ± 0.02bc | 83.66 ± 0.91c | 11.25 ± 0.91h | |
| GWLF | 2.42 ± 0.04b | 2.68 ± 0.04b | 75.43 ± 1.07e | 11.83 ± 1.07gh | |
| GDLF | 2.92 ± 0.07a | 2.91 ± 0.05a | 94.11 ± 1.11a | 12.22 ± 1.11g | |
A consistent pattern was observed in the MAC flour samples after germination, with WAC increasing by 35.96–60.44% and OAC by 50.77–75.16% compared to their non-germinated MAC counterparts. Lentils, when soaked, absorb water, leading to softening of the seed coat and cotyledon, which in turn increases their WAC.35 Microwave treatment may trap more water molecules by unfolding proteins. Moreover, microwave energy penetrates deeply, generating rapid internal heating that causes the food matrix to expand. This volumetric expansion may result in the formation of micro-pores and cracks within the structure, increasing the surface area and the availability of binding sites for water and oil molecules.36 This is also evident in the later sections describing morphological changes (Fig. 3ii). Moreover, it was observed that soaked and germinated samples responded more strongly to MAC. During MAC, proteins may denature upon heating, and exposed hydrophobic sites may increase lipid interactions, resulting in an overall increase in OAC (36.12–116.96%) compared to their raw samples.37
Unlike other technofunctional properties, MAC treatments decreased FC, consistent with the findings of Mahalaxmi et al.43 Although proteins are generally regarded as foam stabilizers, prolonged thermal unfolding and subsequent aggregation during MAC compromise their interfacial film-forming ability, thereby decreasing foamability.41 It is evident that MAC exerts a significant (p < 0.05) influence on FC, compared with pretreatments, indicating that MAC diminishes the sensitivity of FC to prior pretreatments. This loss of significance can be attributed to the thermal sensitivity of foaming properties, as extensive protein denaturation and aggregation during MAC likely dominate interfacial behavior, consequently reducing the relative impact of individual pretreatments and resulting in an overall decrease in FC.
3.3. Antinutritional factors
| Pretreatment | Phytic acid (mg per g dw) | Tannin (mg CE per g dw) | Trypsin inhibitor activity (TIU per mg dw) | TPC (mg GAE per g) | DPPH inhibition (%) | |
|---|---|---|---|---|---|---|
| a Data are presented as mean ± standard deviation. Means sharing different superscript letters within the same column differ significantly (p < 0.05) as per two-way ANOVA followed by the post hoc Tukey test. WLF – whole lentil flour, DLF – dehulled lentil flour, SWLF – soaked whole lentil flour, SDLF – soaked & dehulled lentil flour, GWLF – germinated whole lentil flour, and GDLF – germinated & dehulled lentil flour. dw – dry weight, CE – catechin equivalent, TIU – trypsin inhibitor unit, GAE – gallic acid equivalent, and DPPH – 2,2-diphenyl-1-picrylhydrazyl. | ||||||
| Raw | WLF | 10.8 ± 0.23a | 7.53 ± 0.29a | 4.69 ± 0.27a | 7.87 ± 0.29a | 35.37 ± 1.79a |
| DLF | 5.5 ± 0.21c | 1.26 ± 0.08ef | 3.17 ± 0.11b | 3.87 ± 0.16c | 28.82 ± 1.12bc | |
| SWLF | 8.1 ± 0.47b | 4.42 ± 0.16b | 2.02 ± 0.10d | 6.12 ± 0.11b | 31.94 ± 1.09b | |
| SDLF | 4.2 ± 0.18d | 0.83 ± 0.12fg | 1.29 ± 0.07e | 3.71 ± 0.14c | 22.99 ± 1.15d | |
| GWLF | 2.9 ± 0.25e | 2.98 ± 0.18d | 1.13 ± 0.05ef | 2.25 ± 0.16de | 27.15 ± 02.08c | |
| GDLF | 1.6 ± 0.09fg | 0.43 ± 0.07gh | 0.84 ± 0.09f | 1.97 ± 0.09e | 16.96 ± 1.11ef | |
| MAC | WLF | 4.2 ± 0.42d | 3.67 ± 0.14c | 0.29 ± 0.05g | 3.62 ± 0.12c | 19.69 ± 2.06de |
| DLF | 2.3 ± 0.72ef | 0.60 ± 0.17gh | 0.21 ± 0.08g | 1.87 ± 0.18e | 14.95 ± 1.07fgh | |
| SWLF | 2.7 ± 0.49e | 2.82 ± 0.11d | 0.17 ± 0.04c | 2.62 ± 0.14d | 16.72 ± 1.11efg | |
| SDLF | 1.2 ± 0.27g | 0.36 ± 0.13h | 0.12 ± 0.07c | 1.12 ± 0.08f | 12.78 ± 1.46gh | |
| GWLF | 1.0 ± 0.17g | 1.57 ± 0.16e | 0.11 ± 0.04g | 0.92 ± 0.09fg | 13.38 ± 0.94fgh | |
| GDLF | 0.6 ± 0.06g | 0.19 ± 0.09h | 0.06 ± 0.03g | 0.55 ± 0.07g | 11.75 ± 0.56h | |
Overall, GDLF (germination followed by dehulling) reduced phytic acid by approximately 85%. This may be attributed to the dehulling process, followed by germination, which involves enzymatic breakdown and activation of phytase, thereby resulting in a significant reduction in this antinutrient. MAC alone reduced phytic acid by 61.11%, and when combined with pretreatments, it may further reduce it to 94.44% relative to the control raw sample (WLF). This may arise from the heat-sensitive nature of phytic acid; moreover, as a water-soluble component, it facilitates the latching process during cooking.46 Therefore, in addition to germination, its combination with MAC may be advantageous for substantial reduction.
MAC reduces tannin content by approximately 51.26%. When seeds are heated, especially during cooking, the high temperature may break down tannin–protein and tannin–polysaccharide complexes, releasing tannins, particularly hydrolysable types, which can degrade directly under heat and make them more susceptible to removal.50 Several studies reported that microwave heating is better than conventional cooking methods for reducing tannins.51,52 The highest tannin reduction was observed in GDLF, followed by MAC samples, with 97.47% reduction. However, such extensive removal may affect sensory quality and the retention of beneficial polyphenols. Tannins contribute to characteristic flavor attributes, such as astringency, and also possess antioxidant activity. Therefore, while their reduction improves mineral bioavailability and protein digestibility, it may also result in partial loss of health-promoting phenolic compounds, as reflected by decreases in total phenolic content and antioxidant activity (Table 3).
3.4. Total phenolic content (TPC) and antioxidant activity (AOX)
Lentils are known for their rich bioactive compounds (phenolic content and antioxidants), so analyzing the effects of pretreatments, with and without MAC, on these compounds is crucial. TPC and DPPH inhibition activity was highest in the WLF (raw) sample; these values decreased after dehulling, suggesting that the lentil seed coat may also contain TPC and antioxidants (Table 3). Soaking and germination significantly (p < 0.05) decreased the AOX of raw lentil flours. The percentage reduction in DPPH inhibition activity was around 9.69–20.22% on soaking and 23.24–52.07% after germination. A similar trend was observed for TPC, with a positive correlation.54 After the MAC, AOX and TPC significantly (p < 0.05) decreased by 30.71–48.12% and 51.67–66.76%, respectively. As described by Thepthanee et al., considerable levels of phenolic content were detected in soaking, steaming, and boiling water as a result of leaching.55 Additionally, it has been reported that this reduction results from the mobilization of stored phenolics due to enzyme activation, particularly polyphenol oxidase, during germination.56 Conversely, Sharma and Sahni reported that lucerne germination improved AOX despite a decline in TPC, indicating that changes in AOX during germination are not solely dependent on phenolic concentration.57 Dehulling also significantly reduced DPPH inhibition activity and TPC in both raw and MAC lentil flours, as most phenolic compounds in legumes are present in their seed coat.58 The control sample (WLF-raw) exhibited the highest TPC and the most potent DPPH radical scavenging activity. MAC treatment resulted in a substantial decrease in TPC and AOX across all pretreatments. GDLF following MAC showed the lowest phenolic level (0.55 mg GAE per g dw), which was significantly lower than that of raw WLF. This consistent downward trend suggests that thermal processing exerts a strong influence on phenolic retention, potentially through mechanisms of degradation or leaching, as evidenced in Table 3.3.5. Pasting profile
The pasting properties of a food relate to changes that occur when it is heated up in the presence of water. Such changes influence the digestibility, texture, and, ultimately, the use of the food product. MAC samples exhibited only a negligible increase in viscosity during pasting analysis, unlike raw samples, which showed fully developed curves, as inferred from RVA pasting behavior. This is likely related to prior thermal processing/microwave cooking, which gelatinized the starch and altered its native structure, thereby reducing further viscosity development during this hydrothermal process (Fig. 2). Heating processes such as cooking induce structural modifications in starch, commonly referred to as gelatinization. These modifications result in the release of amylose, the separation of amylopectin double helices, and changes in the starch granule structure. Increased amylose leaching leads to the formation of viscous substances.59,60 The peak viscosity measures the thickening power of the starch.61 The SDLF-raw sample has the highest peak viscosity of 1366 cP, followed by the DLF-raw sample, with a peak viscosity of 900 cP (Table 4). In lentils, the combination of soaking, which promotes water penetration, and dehulling, which removes the highly fibrous outer layer, may enhance starch availability and swelling, thereby contributing to the observed higher peak viscosity. Dehulling reduces the non-starch components of lentils, increasing the overall starch concentration by weight and thereby improving pasting properties. The study found that α-amylase enzyme, which breaks down starch molecules into simpler units, is responsible for the decrease in peak viscosity.28 A similar reduction in peak viscosity was observed for GWLF. However, higher peak viscosities indicate greater starch swelling and thickening power, which is favorable in foods such as porridges, soups, and sauces that require rapid starch viscosity development. The control sample (WLF) has the highest breakdown viscosity, indicating the least resilience to heat and shear stress during cooking. This measurement reflects the stability of the starch. According to Awoyale et al., the final viscosity determines the paste's ability to form a viscous paste after preparation, cooking, and cooling and its resistance to shear forces during stirring.62 The SDLF sample (1800 cP) has the final viscosity, followed by DLF (1216 cP) and SWLF (1214 cP), indicating a stronger paste structure after cooking and cooling with improved gel-forming ability. This suggests a synergistic influence of both soaking and dehulling on starch gelatinization and pasting behavior.| Pretreatment | Peak viscosity (cP) | Trough viscosity (cP) | Breakdown viscosity (cP) | Final viscosity (cP) | Setback viscosity (cP) | Peak time (min) | Pasting temperature (°C) | |
|---|---|---|---|---|---|---|---|---|
| a Data are presented as mean ± standard deviation. Means sharing different superscript letters within the same column differ significantly (p < 0.05) as per two-way ANOVA followed by the post hoc Tukey test. WLF – whole lentil flour, DLF – dehulled lentil flour, SWLF – soaked whole lentil flour, SDLF – soaked & dehulled lentil flour, GWLF – germinated whole lentil flour, and GDLF – germinated & dehulled lentil flour. nd: not defined (no pasting temperature observed as they were already gelatinized). | ||||||||
| Raw | WLF | 798 ± 24c | 705 ± 17d | 93 ± 3.34a | 1009 ± 39c | 304 ± 9.22d | 5.13 ± 0.12a | 76.65 ± 4.21a |
| DLF | 900 ± 12b | 844 ± 21b | 56 ± 1.05c | 1216 ± 24b | 372 ± 7.31c | 5.6 ± 0.10a | 77.45 ± 3.11a | |
| SWLF | 840 ± 30c | 770 ± 16c | 70 ± 2.17b | 1214 ± 21b | 444 ± 11b | 7 ± 0.66a | 79.85 ± 0.93a | |
| SDLF | 1366 ± 47a | 1299 ± 39a | 67 ± 1.03b | 1800 ± 50a | 501 ± 13a | 7 ± 0.20a | 79.05 ± 0.89a | |
| GWLF | 430 ± 12e | 410 ± 19f | 20 ± 0.83e | 586 ± 14d | 176 ± 5.2e | 7 ± 0.32a | 79.9 ± 0.79a | |
| GDLF | 497 ± 16d | 485 ± 13e | 12 ± 0.79f | 648 ± 18d | 163 ± 8.2e | 6.8 ± 0.41a | 80.75 ± 1.68a | |
| MAC | WLF | 22 ± 4.55g | 20 ± 3.70h | 2 ± 0.32g | 35 ± 5.66f | 15 ± 7.22g | 6.86 ± 2.23a | nd |
| DLF | 27 ± 6.2g | 25 ± 5.61h | 2 ± 0.44g | 41 ± 5.98f | 16 ± 4.34g | 6.53 ± 1.98a | nd | |
| SWLF | 27 ± 9.21g | 25 ± 2.66h | 2 ± 0.27g | 48 ± 6.34f | 23 ± 5.20g | 6.73 ± 2.43a | nd | |
| SDLF | 67 ± 7.11fg | 59 ± 8.17gh | 8 ± 0.67fg | 153 ± 9.87e | 94 ± 11f | 7 ± 3.11a | nd | |
| GWLF | 28 ± 6.34g | 26 ± 7.32h | 2 ± 0.29g | 49 ± 4.76f | 23 ± 7.6g | 6.67 ± 1.87a | nd | |
| GDLF | 114 ± 14f | 81 ± 11g | 33 ± 7.32d | 86 ± 3.56f | 5 ± 1.76g | 6.73 ± 3.43a | nd | |
3.6. Morphology
Analyzing flour morphology is essential for understanding the internal physical structure and surface characteristics of flour particles, which directly influence their technofunctional, processing, and nutritional properties. Fig. 3i illustrates a uniform microstructure across all raw samples. Smooth-surfaced starch granules were clearly detected, with shapes ranging from ovoid to spherical; they measure 22–30 µm in length and 10–17 µm in width. According to Aguilera et al., the visible, irregular, and dispersed particles above or near the starch granules are either protein bodies or portions of the protein matrix that were broken up during milling; they may also contain fiber components.63 On the other hand, a slight break in the starch granule surface and a gradual loss of the continuous matrix, which included protein bodies, occurred during soaking (Fig. 3i(C and D)) and germination (Fig. 3i(E and F)). The results agreed with those of Frias et al., who demonstrated physicochemical changes in raw and germinated lentil starch and its components.64 Dehulling, on the other hand, showed no notable changes in the microstructure. Fig. 3ii presents images captured with SEM of lentil samples that have undergone dehulling, soaking, and germination and were subjected to MAC. The raw and the cooked samples have different microstructures. These granules are larger than those observed in the raw samples, with rugged, irregular surfaces and some broken starch granules. This phenomenon may be attributed to the vaporization of water from the granules, leading to puffing after MAC. The presence of voids and fractured surfaces in MAC samples indicates localized expansion and structural rupture, likely associated with rapid internal heating during MAC. Even a mixture of both proteins and starch could provide a homogeneous network of starch granules cross-linked by proteins. Denaturation of proteins during MAC disrupts the individual protein bodies. The disrupted, porous microstructure of MAC flours is consistent with the higher WAC values, as greater surface roughness and porosity may contribute to increased water entrapment. Similarly, the absence of whole starch granules and the fusion of the matrix are consistent with the reduced pasting values of MAC samples, which showed low viscosity likely influenced by prior starch gelatinization.3.7. Limitations
This study was conducted on a single lentil cultivar under controlled conditions; therefore, it may not fully reflect the variability observed across cultivars. Additionally, the research was performed at a laboratory scale, necessitating scale-up for commercial applications. The cooking time for MAC was measured manually using a finger-press test performed by a single trained operator, following the standardized method. Although operator consistency was maintained, the absence of instrumental validation (texture analysis) represents a limitation. Furthermore, milling was followed by sieving through a 150 µm sieve. However, no particle size distribution was performed; therefore, homogeneity of particle size within the fraction was not identified. Future investigations should incorporate multiple cultivars, particle size distribution, an objective cooking endpoint using texture analysis, and pilot-scale processing to validate and extend these findings.4. Conclusions
This study demonstrates that pretreatments (dehulling, soaking, germination, soaking + dehulling, and germination + dehulling), either independently or in conjunction with MAC, significantly diminish antinutrients and modulate the technofunctional properties of lentil flour. An increase in protein content was observed following pretreatments; however, this was primarily due to dry matter loss. Conversely, fat content declined slightly across these treatments. The pretreatment methods, particularly when combined with MAC, markedly reduced antinutrients; nonetheless, TPC and AOX were also diminished following these pretreatments. Pretreatments, such as soaking and dehulling, improved the pasting properties of raw lentil flours. The MAC process, combined with pretreatments, improved technofunctional characteristics while preserving important macronutrients; however, a loss of thermosensitive- and hydro-sensitive constituents was evident. These findings support the potential for further research into lentil flours processed as functional food ingredients. Future investigations, including sensory evaluation, mineral bioavailability assessment, and in vivo validation, would deepen the contextual understanding of these results and facilitate the translation of processing methods into valuable and widely acceptable food products.Author contributions
Kathika Das: conceptualization, methodology, formal analysis, data curation, investigation, visualization, writing – original draft. Shubham Mandliya: formal analysis, data curation, investigation, writing – original draft, writing – review and editing. Pradyuman Kumar: conceptualization, methodology, visualization, resources, supervision, writing – review and editing.Conflicts of interest
The authors declare no conflict of interest with respect to the work described in the manuscript.Data availability
The data will be made available from the corresponding author upon reasonable request.Acknowledgements
The authors acknowledge the facilities provided by the Department of Food Engineering and Technology, Sant Longowal Institute of Engineering and Technology, and Food Chemistry and Technology Laboratory, Indian Institute of Technology Kharagpur. The corresponding author also acknowledges the facilities provided by the Department of Food and Nutritional Sciences, University of Reading, to write and review the article.References
- D. Gupta, R. H. R. Dadu, P. Sambasivam, I. Bar, M. Singh and N. Beera, et al., Toward climate-resilient lentils: challenges and opportunities, in Genomic Designing of Climate-Smart Pulse Crops, ed. C. Kole, Springer, Cham, 2019, pp. 165–234, DOI:10.1007/978-3-319-96932-9_4.
- S. Ram, S. Narwal, O. P. Gupta, V. Pandey and G. P. Singh, Anti-nutritional factors and bioavailability: approaches, challenges, and opportunities, in Wheat and Barley Grain Biofortification, ed. O. P. Gupta, V. Pandey, S. Narwal, P. Sharma, S. Ram and G. P. Singh, Woodhead Publishing, 2020, pp. 101–128, DOI:10.1016/B978-0-12-818444-8.00004-3.
- B. G. Mazı, D. Yıldız and I. Barutçu Mazı, Influence of different soaking and drying treatments on anti-nutritional composition and technological characteristics of red and green lentil (Lens culinaris Medik.) flour, J. Food Meas. Charact., 2023, 17(4), 3625–3643, DOI:10.1007/s11694-023-01906-8.
- P. Thakur, K. Kumar, N. Ahmed, D. Chauhan, Q. U. E. H. Rizvi and S. Jan, et al., Effect of soaking and germination treatments on nutritional, anti-nutritional, and bioactive properties of amaranth (Amaranthus hypochondriacus L.), quinoa (Chenopodium quinoa L.), and buckwheat (Fagopyrum esculentum L.), Curr. Res. Food Sci., 2021, 4, 917–925, DOI:10.1016/j.crfs.2021.11.019.
- Y. M. Choi, H. Yoon, M. J. Shin, S. Lee, J. Yi and Y. A. Jeon, et al., Nutrient levels, bioactive metabolite contents, and antioxidant capacities of faba beans as affected by dehulling, Foods, 2023, 12(22), 4063, DOI:10.3390/foods12224063.
- S. Santos, C. B. Silva, L. Valente, M. P. Gruber and M. W. Vasconcelos, The effect of sprouting in lentil (Lens culinaris) nutritional and microbiological profile, Foods, 2020, 9(4), 400, DOI:10.3390/foods9040400.
- S. Mandliya, J. Majumdar, S. Misra, M. Pattnaik and H. N. Mishra, Evaluation of dry microwave and hot water blanching on physicochemical, textural, technofunctional and organoleptic properties of Indian gooseberry (Phyllanthus emblica), J. Food Meas. Charact., 2023, 17(3), 2881–2891, DOI:10.1007/s11694-023-01833-8.
- R. Pande, S. Tyagi, S. Vishwakarma and S. Mandliya, Dry microwave and hot water blanching of underutilized star fruit (Averrhoa carambola): A comparative assessment of quality attributes and sensory evaluation using chemometric approach, Food Phys., 2025, 2, 100056, DOI:10.1016/j.foodp.2025.100056.
- M. Pattnaik, S. Mandliya and H. N. Mishra, Applications of microwaves in dairy industries, in Microwave Processing of Foods: Challenges, Advances and Prospects, ed. A. Pratap Singh, F. Erdogdu, S. Wang and H. S. Ramaswamy, Food Engineering Series, Springer International Publishing, Cham, 2024, pp. 551–564, DOI:10.1007/978-3-031-51613-9_27.
- T. H. Hefnawy, Effect of processing methods on nutritional composition and antinutritional factors in lentils (Lens culinaris), Ann. Agric. Sci., 2011, 56(2), 57–61, DOI:10.1016/j.aoas.2011.07.001.
- M. Gallego, M. Arnal, J. M. Barat and P. Talens, Effect of cooking on protein digestion and antioxidant activity of different legume pastes, Foods, 2020, 10(1), 47, DOI:10.3390/foods10010047.
- L. Chin, N. Therdthai and W. Ratphitagsanti, Effect of microwave cooking on quality of riceberry rice (Oryza sativa L.), J. Food Qual., 2020, 2020(1), 4350274, DOI:10.1155/2020/4350274.
- L. Chin, N. Therdthai and W. Ratphitagsanti, Effect of conventional and microwave cooking conditions on quality and antioxidant activity of Chinese kale (Brassica alboglabra), Appl. Food Res., 2022, 2(1), 100079, DOI:10.1016/j.afres.2022.100079.
- S. Lisciani, A. Aguzzi, P. Gabrielli, E. Camilli, L. Gambelli and L. Marletta, et al., Effects of household cooking on mineral composition and retention in widespread Italian vegetables, Nutrients, 2025, 17(3), 423, DOI:10.3390/nu17030423.
- W. Ramadhan, S. Purwaningsih and S. Nafisah, Effects of microwave cooking on the physicochemical and sensory properties of seaweed-based analogue rice, Int. J. Food Sci. Technol., 2024, 59(3), 1770–1780, DOI:10.1111/ijfs.16930.
- AOAC, Official Methods of Analysis, ed. W. Horwitz, Association of Official Agricultural Chemists, Washington, DC, 13th edn, 1980, pp. 56–132 Search PubMed.
- C. G. Dalbhagat and H. N. Mishra, Effects of extrusion process conditions on system parameters; physicochemical properties and cooking characteristics of extruded fortified rice kernels, J. Cereal Sci., 2019, 89, 102782, DOI:10.1016/j.jcs.2019.05.016.
- AOAC, Official Methods of Analysis of AOAC International, AOAC International, Gaithersburg, MD, 18th edn, 2005 Search PubMed.
- S. Mandliya and H. N. Mishra, Effect of different drying methods on quality attributes and microstructure of mycelium (Pleurotus eryngii), Biol. Life Sci. Forum, 2021, 6(1), 10, DOI:10.3390/foods2021-10950.
- S. Mandliya, A. Pratap-Singh, S. Vishwakarma, C. G. Dalbhagat and H. N. Mishra, Incorporation of mycelium (Pleurotus eryngii) in pea protein based low moisture meat analogue: Effect on its physicochemical, rehydration and structural properties, Foods, 2022, 11(16), 2476, DOI:10.3390/foods11162476.
- C. Badia-Olmos, J. Sánchez-García, L. Laguna, E. Zúñiga, C. M. Haros and A. M. Andrés, et al., Flours from fermented lentil and quinoa grains as ingredients with new techno-functional properties, Food Res. Int., 2024, 177, 113915, DOI:10.1016/j.foodres.2023.113915.
- D. S. Gupta, A. Dutta, V. S. Sharanagat, J. Kumar, A. Kumar and V. Kumar, et al., Effect of growing environments on the minerals and proximate composition of urdbeans (Vigna mungo L. Hepper), J. Food Compos. Anal., 2022, 114, 104746, DOI:10.1016/j.jfca.2022.104746.
- C. E. Palacios, A. Nagai, P. Torres, J. A. Rodrigues and A. Salatino, Contents of tannins of cultivars of sorghum cultivated in Brazil, as determined by four quantification methods, Food Chem., 2021, 337, 127970, DOI:10.1016/j.foodchem.2020.127970.
- A. I. Martinez-Gonzalez, Á. G. Díaz-Sánchez, L. A. de la Rosa, I. Bustos-Jaimes and E. Alvarez-Parrilla, A novel approach to trypsin inhibition by flavonoids, J. Food Bioact., 2021, 14, 102–113, DOI:10.31665/jfb.2021.14272.
- S. Vishwakarma, S. Mandliya, C. G. Dalbhagat, J. Majumdar and H. N. Mishra, Effect of marjoram leaf powder addition on nutritional, rheological, textural, structural, and sensorial properties of extruded rice noodles, Foods, 2023, 12(5), 1099, DOI:10.3390/foods12051099.
- D. Pathiraja, J. P. Wanasundara, F. M. Elessawy, R. W. Purves, A. Vandenberg and P. J. Shand, Water-soluble phenolic compounds and their putative antioxidant activities in the seed coats from different lentil (Lens culinaris) genotypes, Food Chem., 2023, 407, 135145, DOI:10.1016/j.foodchem.2022.135145.
- D. Pathiraja, J. P. Wanasundara, F. M. Elessawy, R. W. Purves, A. Vandenberg and P. J. Shand, Water-soluble phenolic compounds and their putative antioxidant activities in the seed coats from different lentil (Lens culinaris) genotypes, Food Chem., 2023, 407, 135145, DOI:10.1111/jfpe.14124.
- A. Ghumman, A. Kaur and N. Singh, Impact of germination on flour, protein and starch characteristics of lentil (Lens culinari) and horsegram (Macrotyloma uniflorum L.) lines, LWT--Food Sci. Technol., 2016, 65, 137–144, DOI:10.1016/j.lwt.2015.07.075.
- A. Ghumman, A. Kaur and N. Singh, Impact of germination on flour, protein and starch characteristics of lentil (Lens culinaris) and horsegram (Macrotyloma uniflorum L.) lines, LWT--Food Sci. Technol., 2016, 65, 137–144, DOI:10.1021/jf303739n.
- H. Sharma, N. Ramawat and C. Gupta, Nutritive content of lentil, Journal of Nutritional Health & Food Engineering, 2022, 12(1), 27–32, DOI:10.15406/jnhfe.2022.12.00351.
- S. Hussain, T. Liu, N. Iqbal, M. Brestic, T. Pang and M. Mumtaz, et al., Effects of lignin, cellulose, hemicellulose, sucrose and monosaccharide carbohydrates on soybean physical stem strength and yield in intercropping, Photochem. Photobiol. Sci., 2020, 19(4), 462–472, 10.1039/c9pp00369j.
- T. Messou, K. M. Adrien, G. K. Cyrile and T. Kablan, Effect of cooking time on the nutritional and anti-nutritional properties of red and black beans of Phaseolus lunatus (L.) consumed in south and east of Côte d'Ivoire, GSC Biol. Pharm. Sci., 2023, 22(1), 269–281, DOI:10.30574/gscbps.2023.22.1.0459.
- M. Thakur, P. Sharma, A. Anand, V. K. Pandita, A. Bhatia and S. Pushkar, Raffinose and hexose sugar content during germination are related to infrared thermal fingerprints of primed onion (Allium cepa L.) seeds, Front. Plant Sci., 2020, 11, 579037, DOI:10.3389/fpls.2020.579037.
- H. Bobade, S. Sharma and B. Singh, Effect of ingredient interactions on techno-functional properties, in Food Formulation: Novel Ingredients and Processing Techniques, ed. S. Pathania and B. K. Tiwari, CRC Press, 2021, pp. 71–99, DOI:10.1002/9781119614760.ch5.
- J. Munthali, S. G. Nkhata, K. Masamba, T. Mguntha, R. Fungo and R. Chirwa, Soaking beans for 12 h reduces split percent and cooking time regardless of type of water used for cooking, Heliyon, 2022, 8(9), e10561, DOI:10.1016/j.heliyon.2022.e10561.
- W. Sui, S. Wang, Y. Chen, X. Li, X. Zhuang and X. Yan, et al., Insights into the structural and nutritional variations in soluble dietary fibers in fruits and vegetables influenced by food processing techniques, Foods, 2025, 14(11), 1861, DOI:10.3390/foods14111861.
- S. Ashraf, S. M. G. Saeed, S. A. Sayeed and R. Ali, Impact of microwave treatment on the technofunctionality of cereals and legumes, Int. J. Agric. Biol., 2012, 14(3), 365–370 CAS.
- H. Nawaz, R. Waheed, M. Nawaz and D. Shahwar, Physical and chemical modifications in starch structure and reactivity, in Chemical Properties of Starch, ed. M. Emeje, IntechOpen, 2020, DOI:10.5772/intechopen.88870.
- G. Scott and J. M. Awika, Effect of protein–starch interactions on starch retrogradation and implications for food product quality, Compr. Rev. Food Sci. Food Saf., 2023, 22(3), 2081–2111, DOI:10.1111/1541-4337.13141.
- D. Donmez, L. Pinho, B. Patel, P. Desam and O. H. Campanella, Characterization of starch–water interactions and their effects on two key functional properties: starch gelatinization and retrogradation, Curr. Opin. Food Sci., 2021, 39, 103–109, DOI:10.1016/j.cofs.2020.12.018.
- Z. Wang, X. Liu, H. Luo, B. Peng, X. Sun and Y. Liu, et al., Foaming properties and foam structure of produced liquid in alkali/surfactant/polymer flooding production, J. Energy Resour. Technol., 2021, 143(10), 103005, DOI:10.1115/1.4050498.
- Y. Di, X. Li, X. Chang, R. Gu, X. Duan, F. Liu, X. Liu and Y. Wang, Impact of germination on structural, functional properties and in vitro protein digestibility of sesame (Sesamum indicum L.) protein, LWT, 2022, 15(154), 112651, DOI:10.1016/j.lwt.2021.112651.
- S. Mahalaxmi, P. Himashree, B. Malini and C. K. Sunil, Effect of microwave treatment on the structural and technofunctional properties of proteins in lentil flour, Food Chem. Adv., 2022, 1, 100147, DOI:10.1016/j.focha.2022.100147.
- R. K. Gupta, S. S. Gangoliya and N. K. Singh, Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains, J. Food Sci. Technol., 2015, 52, 676–684, DOI:10.1007/s13197-013-0978-y.
- R. S. Pal, A. Bhartiya, P. Yadav, L. Kant, K. K. Mishra and J. P. Aditya, et al., Effect of dehulling, germination and cooking on nutrients, anti-nutrients, fatty acid composition and antioxidant properties in lentil (Lens culinaris), J. Food Sci. Technol., 2017, 54, 909–920, DOI:10.1007/s13197-016-2351-4.
- S. Sarkhel and A. Roy, Phytic acid and its reduction in pulse matrix: structure–function relationship owing to bioavailability enhancement of micronutrients, J. Food Process Eng., 2022, 45(5), e14030, DOI:10.1111/jfpe.14030.
- A. Thakur, V. Sharma and A. Thakur, An overview of anti-nutritional factors in food, Int. J. Chem. Stud., 2019, 7(1), 2472–2479 CAS.
- M. Mirali, R. W. Purves and A. Vandenberg, Profiling the phenolic compounds of the four major seed coat types and their relation to color genes in lentil, J. Nat. Prod., 2017, 80(5), 1310–1317, DOI:10.1021/acs.jnatprod.6b00872.
- S. Khandelwal, S. A. Udipi and P. Ghugre, Polyphenols and tannins in Indian pulses: effect of soaking, germination and pressure cooking, Food Res. Int., 2010, 43(2), 526–530, DOI:10.1016/j.foodres.2009.09.036.
- S. Molino, A. Lerma-Aguilera, L. G. Gómez-Mascaraque, J. Á. Rufián-Henares and M. P. Francino, Evaluation of tannin-delivery approaches for gut microbiota modulation: comparison of pectin-based microcapsules and unencapsulated extracts, J. Agric. Food Chem., 2023, 71(38), 13988–13999, DOI:10.1021/acs.jafc.3c02949.
- M. A. U. El-Geddawy, M. A. Sorour, S. H. Abou-El-Hawa and E. M. M. Taha, Effect of domestic processing and microwave heating on phenolic compounds and tannins in some oil seeds, SVU Int. J. Agric. Sci., 2019, 1(2), 23–32, DOI:10.21608/svuijas.2019.67094.
- R. A. Habiba, Changes in anti-nutrients, protein solubility, digestibility, and HCl-extractability of ash and phosphorus in vegetable peas as affected by cooking methods, Food Chem., 2002, 77(2), 187–192, DOI:10.1016/S0308-8146(01)00335-1.
- B. Fraser, Structure, function, and inhibition of type 2 transmembrane serine proteases, Doctoral dissertation, University of British Columbia, Vancouver, 2024, http://hdl.handle.net/2429/88963.
- H. S. Gujral, M. Angurala, P. Sharma and J. Singh, Phenolic content and antioxidant activity of germinated and cooked pulses, Int. J. Food Prop., 2011, 14(6), 1366–1374, DOI:10.1080/10942911003672167.
- C. Thepthanee, H. Li, H. Wei, C. Prakitchaiwattana and S. Siriamornpun, Effect of soaking, germination, and roasting on phenolic composition, antioxidant activities, and fatty acid profile of sunflower (Helianthus annuus L.) seeds, Horticulturae, 2024, 10(4), 387, DOI:10.3390/horticulturae10040387.
- M. S. Alkaltham, M. Musa Özcan, N. Uslu, A. M. Salamatullah and K. Hayat, Changes in antioxidant activity, phenolic compounds, fatty acids, and mineral contents of raw, germinated, and boiled lentil seeds, J. Food Sci., 2022, 87(4), 1639–1649, DOI:10.1111/1750-3841.16099.
- S. Sharma and P. Sahni, Dynamics of germination behaviour, protein secondary structure, technofunctional properties, antinutrients, antioxidant capacity and mineral elements in germinated dhaincha, Food Technol. Biotechnol., 2021, 59(2), 238–250, DOI:10.17113/ftb.59.02.21.6922.
- B. Singh, J. P. Singh, A. Kaur and N. Singh, Phenolic composition and antioxidant potential of grain legume seeds: a review, Food Res. Int., 2017, 101, 1–16, DOI:10.1016/j.foodres.2017.09.026.
- S. Wang and L. Copeland, Molecular disassembly of starch granules during gelatinization and its effect on starch digestibility: a review, Food Funct., 2013, 4(11), 1564–1580, 10.1039/c3fo60258c.
- X. Wei, Y. Gong and X. Hu, Effects of starch, protein structure change on quality of raw and cooked frozen oat roll, LWT--Food Sci. Technol., 2022, 167, 113839, DOI:10.1016/j.lwt.2022.113839.
- M. Vandana, S. Vishwakarma, S. Mandliya, H. N. Mishra and Y. Bhosale, Incorporation of microcrystalline cellulose into extruded rice analogue: effects on the physicochemical, technofunctional, textural, cooking and structural properties, in vitro digestibility and estimated glycaemic index, Sustainable Food Technol., 2026, 4, 1509–1524, 10.1039/d5fb00272a.
- W. Awoyale, A. A. Oyebanji and E. A. Irondi, Technofunctional and pasting properties of succinylated cocoyam (Xanthosoma sagittifolium) starch: an insight into its potential industrial applications, Food Environ. Saf., 2024, 23(3), 171–185, DOI:10.4316/fens.2024.014.
- Y. Aguilera, R. M. Esteban, V. Benítez, E. Molla and M. A. Martín-Cabrejas, Starch, technofunctional properties, and microstructural characteristics in chickpea and lentil as affected by thermal processing, J. Agric. Food Chem., 2009, 57(22), 10682–10688, DOI:10.1021/jf902042r.
- J. Frias, J. Fornal, S. G. Ring and C. Vidal-Valverde, Effect of germination on physico-chemical properties of lentil starch and its components, LWT--Food Sci. Technol., 1998, 31(3), 228–236, DOI:10.1006/fstl.1997.0340.
| This journal is © The Royal Society of Chemistry 2026 |