Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
An assessment of spruce wood as a replacement for peat in the production of whisky: chemical and sensory analysis of new make spirits
Kacper P. Krakowiak a,
Barry Harrisonb,
Irene Baxterb,
Nicholle G. A. Bell
a,
Barry Harrisonb,
Irene Baxterb,
Nicholle G. A. Bell c,
Sam Fergussonc,
Clayton Magill
c,
Sam Fergussonc,
Clayton Magill d,
Ruaraidh D. McIntosh
d,
Ruaraidh D. McIntosh *a and
David Ellis
*a and
David Ellis *a
*a
aInstitute of Chemical Sciences, School of Engineering and Physical Sciences, Heriot-Watt University, Edinburgh, EH14 4AS, UK. E-mail: kk67@hw.ac.uk; r.mcintosh@hw.ac.uk; d.ellis@hw.ac.uk; Tel: +44 01314518039 Tel: +44 01314518038
bScotch Whisky Research Institute, Riccarton, Edinburgh, EH14 4AP, UK
cEastCHEM School of Chemistry, University of Edinburgh, David Brewster Rd, Edinburgh, EH9 3FJ, UK
dThe Lyell Centre, Heriot-Watt University, Edinburgh, EH14 4BA, UK
First published on 10th April 2026
Abstract
Thermal decomposition of spruce wood and peat was investigated and compared using thermogravimetric analysis and pyrolysis-enabled gas chromatography mass spectrometry (GC–MS). Afterwards, new make spirit samples were produced using both materials as sources of smoke, with the resulting spirits analysed using a combination of NMR, FT-ICR-MS and sensory analysis. Thermal degradation studies revealed that while spruce wood and peat both feature similar compositional biopolymers, spruce contains relatively more (poly)carbohydrates and less lignin than peat. Additionally, while both materials showed dominance of guaiacyl (G) units in the lignin structure, the abundance of G units was more extreme in spruce wood. Desirable phenolic species were produced upon pyrolysis of both peat and spruce wood, however, greater abundance of them was produced by peat. These observations were reflected in our results of spirit analysis, which showed similar levels of some phenolics, in particular cresols, but greater amount of methoxyphenolic species in peated new make spirit. Sensory analysis revealed that spruce smoked new make spirit could be described as “lightly smoked” with no unpleasant off notes detected by the sensory panel, which aligns with our instrumental findings. For a better comparison of the balance of smoke aromas, the intensities of wood and peat smoked new make spirit would need to be matched. These results do suggest that some softwoods may be used to provide a smoky aroma with similarities to that traditionally associated with peat.
Sustainability spotlightIn this work we show how a sustainable alternative to peat may be used in the production of Scotch Whisky. Peat is a non-renewable resource which plays an important role in absorbing and storing carbon dioxide. Unfortunately, commercial extraction has led to degradation of peatlands, which not only negatively impacts their ability to act as carbon sinks, but also leads to destruction of natural habitat of a variety of wildlife. Due to need to preserve and restore peat its current users, including the Scotch Whisky industry, must look towards minimising peat use and exploring other, more sustainable, options. This work contributes towards the UN's Sustainable Development Goals 12 (responsible production and consumption), 13 (climatic action), and 15 (life on land). |
1. Introduction
Scotch Whisky is the most prominent alcoholic drink produced in Scotland, enjoyed and recognised worldwide for its sensory quality and unique flavour. Some of the most distinct types of Scotch are those made using peat smoke to augment the spirit's aroma and flavour profile.1,2 These characteristics are developed as a result of the uptake of specific compounds (often referred to as congeners) by the malt, which are released upon thermal degradation of peat, during the malting process.3,4 Peat itself is a type of soil made up of slowly decaying organic matter, with its primary components being large biopolymers – cellulose, hemicellulose and lignin.5,6 A large number of smaller molecules have also been detected within peat.7 As a result of its chemical complexity and its variance in composition, depending on depth of the sample studied, its geographical origin and several other factors, peat has been referred to as one of the most complex mixtures on Earth.8,9For the purpose of whisky production, peat is sourced from independent suppliers by third-party maltsters from peatlands across Scotland. The exact geographical origin differs between distilleries and the amount used annually has been estimated to be approximately 8000 tonnes. The primary concern in terms of flavour imparted by the smoke is the nature of the molecules released upon thermal degradation, which is related to the constituent biomass. Several studies have been performed on thermal decomposition of individual biopolymers, as well as on bulk peat biomass, providing an insight into which compounds may be present in peat smoke.10–13 These findings, combined with studies on whisky congeners show clearly that phenolic species, originating from lignin and other polyaromatics, together with carbohydrate decomposition products can be most closely connected to the smoky characteristics.
Unfortunately, peat is a limited resource owing to its slow accumulation rate, estimated to be approximately 1 mm per year.5,14 This, combined with continuous historical exploitation of peatlands for fuel, agriculture, construction and horticulture has led to increasing scarcity of this natural resource alongside a concerted drive to limit its use, potentially impacting on the whisky industry.15,16 Several programmes aiming to reduce the usage of peat have been recently launched, and research into making the peating process more efficient is already underway.17,18 Another possible solution to reducing peat usage in whisky production while also allowing preservation of the smoky characteristics, would be to augment peat with an alternative material that is capable of producing similar peat thermal decomposition congeners, in comparable quantities when burned. Such an alternative should logically be composed of similar biopolymers as peat. Among the potential candidates are varieties of wood, that are already used for smoking other alcoholic beverages (e.g. beer) for which they were found to produce flavour compounds comparable to those produced from peated malt.19,20 But, the production processes for whisky and beer differ significantly. Therefore, any attempt to directly translate research about beer to whisky would be misplaced, and highlights a need for further, targeted investigation. Another matter is whether the approach of substituting peat use with another material would be acceptable. According to the Scotch Whisky technical file, the spirit must remain the expected organoleptic properties associated with Scotch Whisky to be considered as such. Peated whisky would be expected to have burnt, smoky and/or medicinal-type aromas. Our study aims to investigate whether that is possible and to provide information for any future discussion the topic.
Both wood and peat are composed of cellulose, hemicellulose and lignin, and the primary difference between them is how much of each biopolymer contributes to their overall composition. As was mentioned previously, there is marked variation between peat from different sources and similarly pronounced differences are present in wood depending on what part of the plant it originates from (e.g. heartwood versus sapwood), taxonomy and biogeographical origin.21,22 One of the most important distinctions is between softwoods (gymnosperms) and hardwoods (angiosperms), the former being identified by presence of naked seeds and needlelike or scalelike leaves, while the latter produces seeds within fruiting bodies. In terms of chemical composition, an important distinguishing feature of these two types of wood is that gymnospermous taxa tend to have a higher lignin content, while angiosperms contain more pentosan hemicellulose.23 The composition of lignin itself differs between softwoods and hardwoods, with angiosperms being proportionally richer in syringyl (S) units as compared to guaiacyl (G) units, and the opposite is the case for softwoods.24 These compositional differences are expected to create differences in the congeners released upon thermal degradation. Since softwoods contain more abundant G unit building blocks, corresponding gymnosperms would be expected to produce smoke containing higher concentrations of desirable phenolic congeners. A representative softwood taxa commonly found in Scotland is spruce. Therefore, this material was chosen to be investigated in this study due to the above reasoning, and it's known high phenolic content.25
Because of the complex composition of smoke and its precursors, we employ multiple complementary analytical techniques to study the materials and their thermal degradation products. Thermogravimetric analysis (TGA) is perhaps the simplest method in concept but nonetheless provides useful information by measuring changes in mass of the sample with increasing temperature under oxygen-free atmosphere. As a result, it is possible to establish how much of the sample degrades across various temperature ranges, which in turn is correlated to their composition. TGA is a commonly applied technique for analysis of thermal behaviour of materials, ranging from biomatter to synthetic polymers.26,27 This technique does not, however, provide any information on the molecular composition of released compounds, which is ultimately the most important factor in whisky production. In order to address this, we made use of pyrolysis coupled to gas chromatography and mass spectrometry (pyrolysis-enabled GC–MS or pyGC–MS). Using this method, it is possible to identify particular compounds released during pyrolysis. It also allows for precise temperature control, which has been previously shown to be crucial for control over thermal decomposition processes.28,29 Some of the limitations of pyGCMS include difficulty in obtaining quantitative results and a somewhat limited scope of compounds that can be detected in a single experiment, though when considering flavour, it tends to be the volatile, GC amenable, compounds we are interested in.30
Another method used was Nuclear Magnetic Resonance (NMR) spectroscopy, which has been employed previously for analysis of whisky and new make spirits, giving a quick overview of the chemical composition of the sample.31–33 While it allows for detection and potential quantitation of a large variety of compounds in one experiment, it suffers from limited sensitivity and issues involving signal overlap, especially for complex mixture analysis. To address these issues, Fourier Transform Ion Cyclotron Resonance Mass Spectrometry (FT-ICR-MS) was employed, a highly sensitive non-targeted technique, able to detect thousands of compounds in whisky and new make spirit samples.34,35 The biggest limitation of the technique is its inability to determine molecular structure without using additional instruments and extensive amount of work. As a result of the large amount of data involved and the aforementioned limitations the results obtained are most often interpreted using statistical methods, including multivariate analysis, which reveals underlying trends in the data, even if precise identification of compounds is not practical. Both NMR and FT-ICR-MS are also helpful in the comparison of samples, which is the goal of our work. Ultimately, the most important factor is whether the spirit produced has the desired sensory characteristics, which is why we have also employed sensory analysis, allowing for correlation of chemical data obtained using instrumental analysis with the sensory characteristics potential consumers would experience.36 Through the combination of the aforementioned techniques, we aimed to test whether spruce wood is a suitable replacement for peat in whisky production, both in terms of chemical composition of the spirit and its sensory properties. The primary aspects investigated were whether substituting peat with spruce would affect the “smoky”, “medicinal” and “burnt” sensory notes and if any unexpected or unpleasant off notes would be introduced. However, it is important to note that the understanding of the relationship between the chemical profile of the spirit and its sensory perception remains underdeveloped and as a result, attempts to predict one based purely on the other are challenging. Using a range of techniques, we can identify and quantify a wide range of compounds but the extent to which these compounds interact with one another in the sensory experience is challenging to assess and leads to discrepancies between the sensory characteristics experienced by consumers. This gap in the current understanding highlights the need to combine both sensory evaluation and chemical analysis to make a more accurate assessment of any changes we make to the process through the impact it has on both the composition and perceived flavour profile of the resulting spirit.
2. Experimental
2.1 Samples
Four solid samples were analysed by TGA and PyGCMS, including two peat samples from different locations (Islay – PI, Aberdeenshire – PA, surface vegetation dominated by heather, surface peat, 0–5 cm depth in both cases) and two spruce (Picea stichensis) wood branch samples with (SB) and without bark (SNB). Peat samples were ground up into a homogenous powder using a ceramic mortar and pestle. Dust from wood samples was obtained using a 120 grit sanding paper. 0.5 mg of each sample was used for pyGCMS and 40–100 mg for TGA (see SI for exact weights used for pyGCMS and TGA). Eight distillate samples produced on a laboratory scale were analysed using NMR (full details on new make spirit preparation are available in the SI): peated low wines (PLW), three peated distillation fractions (PNMS), spruce smoked low wines (SLW) and three spruce smoked spirit distillation fractions (SNMS). Low wines result from the first distillation of the fermented wash, the spirit distillation fractions result from the second distillation. The peated samples were prepared using Islay peat and the distillation cut points used were: three consecutive 100 mL spirit distillation fractions. Five spirit samples were analysed using FT-ICR-MS: peated new make spirit, spruce-smoked low wines and three spruce-smoked distillation fractions. For the sensory analysis, Islay peat smoked malt and spruce wood (with bark) smoked malt were used to produce respectively the peated and the spruce smoked new make spirits. Islay peat and spruce with bark were chosen based on TGA and PyGCMS results. Duplicate samples of peated NMS (PNMS1, PNMS2) and spruce smoked NMS (SNMS1, SNMS2) were produced.2.2 TGA
TG analysis was performed using an STA PT 1600 Linseis analyser with the following configuration: 100 cm3 min−1 nitrogen flow, temperature range 20 °C–800 °C with a heating ramp of 10 °C per minute, dwell time of 10 minutes and cooling rate of 10 °C per minute.2.3 PyGC–MS
Pyrolysis was achieved through a multi-shot pyrolyser – Frontier Lab EGA PY-3030D with peripheral auto-shot sampler (Frontier Lab AS-2020E) coupled to a split/splitless inlet GC (Thermo Scientific Trace 1300; split 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 250) and single quadrupole MS (Thermo Scientific ISQ) in electron impact ionisation (70 eV) mode. The pyrolyser was operated in single shot mode with temperature set to 550 °C. The GC was outfitted with a 30 m TG-5SilMS (0.25 mm inner diameter; 0.25 µm film thickness) capillary column with helium as carrier gas at 1 mL min−1 flow rate. Inlet, transfer line and source temperatures were set to 300 °C. Initial oven temperature was set to 70 °C, with heating rate of 10 °C for 17 minutes, followed by heating rate of 40 °C for 2 minutes, reaching a final temperature of 320 °C held for 3 minutes. Ionisation occurred at 70 eV, and ionised fragments were identified through the Chromeleon Chromatography Data System (CDS; version 7.2.10) via comparison with authentic standards when available in conjunction with retention index, relative elution order, and spectrometric analysis of pyrogram fragmentation spectra against the NIST 2017 mass spectral library and literature.12,37–40
250) and single quadrupole MS (Thermo Scientific ISQ) in electron impact ionisation (70 eV) mode. The pyrolyser was operated in single shot mode with temperature set to 550 °C. The GC was outfitted with a 30 m TG-5SilMS (0.25 mm inner diameter; 0.25 µm film thickness) capillary column with helium as carrier gas at 1 mL min−1 flow rate. Inlet, transfer line and source temperatures were set to 300 °C. Initial oven temperature was set to 70 °C, with heating rate of 10 °C for 17 minutes, followed by heating rate of 40 °C for 2 minutes, reaching a final temperature of 320 °C held for 3 minutes. Ionisation occurred at 70 eV, and ionised fragments were identified through the Chromeleon Chromatography Data System (CDS; version 7.2.10) via comparison with authentic standards when available in conjunction with retention index, relative elution order, and spectrometric analysis of pyrogram fragmentation spectra against the NIST 2017 mass spectral library and literature.12,37–40
2.4 NMR spectroscopy
NMR samples were prepared by adding acetic acid/sodium acetate buffer solution containing sodium trimethylsilylpropanesulfonate (DSS) (0.1 mL) to the sample (0.5 mL) to give a total volume of approximately 0.6 mL. The buffer was prepared following methodology of Kew et al. by mixing sodium acetate-d3 (0.0277 g, 0.204 mmol), acetic acid-d4 (0.0820 g, 1.28 mmol), DSS (0.0131 g, 0.0600 mmol) and filling up to mark with D2O in a 10 mL volumetric flask.31 The final concentration of the DSS standard was 1.00 mM in a standard 5 mm NMR tube and the pH of the solution was 3.95. Selected congeners were quantified through comparison of integration values of NMR signals, compared to the signal of the DSS standard of known concentration. ABV (alcohol by volume, measured as mL of ethanol in 100 mL of beverage) values were determined using previously established NMR methodology and compared with values obtained using a portable alcohol meter (Snap 50 Portable Alcohol Meter for Distillates, Anton-Paar; Graz, Austria).41All NMR experiments were performed on a 600 MHz Bruker AVIII spectrometer equipped with a 5 mm TCI cryoprobe with z gradients at 300 K at the School of Chemistry, University of Edinburgh. NMR data obtained was processed using TopSpin and Microsoft Excel software. Spectra were acquired using a multiple solvent suppression sequence optimised for whisky analysis used previously and described in detail by Kew et al.33 The assignment of signals was done by comparing with online databases, literature and spectra of pure compounds.31,42–46
2.5 FT-ICR-MS
MS samples were prepared by adding 300 µL of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of LC–MS grade water/methanol (Fisher) to 30 µL of sample. Triplicates were prepared for each sample. Each sample was prepared immediately prior to introduction into the electrospray ionisation (ESI) source. Mass spectra were acquired on a Bruker SolariX 12 Tesla FT-ICR mass spectrometer with an ESI source in negative mode. This has been widely reported as the preferred ionisation method for whisky samples.47 The flow rate was set to 120 µL h; nebuliser gas flow was set to 1.8 bar; drying gas flow was 6 L min−1 at 180 °C. Ions were accumulated over 0.3 s with a time of flight between the quadrupole and the ICR cell set to 0.6 ms. 4 MW FIDs were acquired for 200 total scans sampling between 98.29 m/z and 1000 m/z.
1 mixture of LC–MS grade water/methanol (Fisher) to 30 µL of sample. Triplicates were prepared for each sample. Each sample was prepared immediately prior to introduction into the electrospray ionisation (ESI) source. Mass spectra were acquired on a Bruker SolariX 12 Tesla FT-ICR mass spectrometer with an ESI source in negative mode. This has been widely reported as the preferred ionisation method for whisky samples.47 The flow rate was set to 120 µL h; nebuliser gas flow was set to 1.8 bar; drying gas flow was 6 L min−1 at 180 °C. Ions were accumulated over 0.3 s with a time of flight between the quadrupole and the ICR cell set to 0.6 ms. 4 MW FIDs were acquired for 200 total scans sampling between 98.29 m/z and 1000 m/z.
Spectra were acquired in a randomised order and the ion source was flushed with water/methanol between samples. Blank solvent spectra were acquired using the same water/methanol mixture at the start and end of the session as well as after every third sample. Details on spectral processing can be found in the SI.
2.6 Sensory analysis
All evaluations were carried out by the expert sensory panel at the Scotch Whisky Research Institute (SWRI). For this study, fourteen panellists (SWRI employees, over 18 years of age, mixed gender) were involved, all of whom have training and expertise in the evaluation of Scotch Whisky. The three consecutive 100 mL distillation fractions for each sample run were composited to provide adequate volume for sensory analysis. Samples were diluted to 20% ABV using distilled water on the day of testing and 20 mL samples presented in blind-coded tulip-shaped nosing glasses covered with watch glasses to trap the volatile aromas. Quantitative Descriptive Profiling (QDP) conducted in accordance with ISO 13299![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2016 was used to assess the aroma characteristics of the samples using SWRI's peated NMS vocabulary as detailed in Table 1. Panellists nosed the samples and scored the intensity of each of these attributes using a 0–3 continuous line scale marked at 0.5 intervals. Panellists were also given the opportunity to comment on any additional or unusual aromas detected in the samples.
2016 was used to assess the aroma characteristics of the samples using SWRI's peated NMS vocabulary as detailed in Table 1. Panellists nosed the samples and scored the intensity of each of these attributes using a 0–3 continuous line scale marked at 0.5 intervals. Panellists were also given the opportunity to comment on any additional or unusual aromas detected in the samples.
| Attribute | Defintion |
|---|---|
| Overall peat intensity | Impression of the overall ‘peatiness’ of the sample |
| Burnt | Tar, char, ash |
| Smoky | Burning wood, smoked foods, barbeque. Bonfire |
| Medicinal | Antiseptic, TCP, carbolic soap, first aid kit |
| Feinty | Leathery, tobacco, sweaty |
| Cereal | Malted barley, digestive biscuits, popcorn, mash, wort, malt extract |
| Green/Grassy | Leafy, herbal, green vegetables |
| Floral | Fresh flowers, perfumed |
| Fruity/Estery | Apples, pears, bananas, tropical fruit, citrus, artificial fruit flavourings |
| Solventy | Paint thinners, nail varnish remover |
| Soapy | Unperfumed soap |
| Sweet | Vanilla, honey, toffee |
| Nutty | Coconut, almonds, nuts, toasted |
| Oily | Oils, fats, waxy |
| Sour | Vinegary, cheesy, vomit |
| Stale | Earthy, musty, metallic |
Sensory testing was conducted in the Sensory Laboratory at SWRI under standard conditions (balanced sample presentation order; tests conducted under red lighting) as per SWRI's operating procedure for sensory analysis.
Mean scores for each attribute were calculated and analysis of variance (ANOVA) used to determine statistically significant (p < 0.05) differences between the samples followed by a Tukey's post hoc test to determine sample groupings for any significant result. Compusense® cloud based sensory software (Compusense Inc., Guelph, ON, Canada) was used for statistical analysis of the sensory data.
3. Results and discussion
3.1 TGA of peat and spruce wood
Thermal gravimetric analysis of two samples of peat from different locations and two different spruce wood samples – one with bark and one without – was performed to establish the range of temperature in which these materials decompose. The two peat samples show similar behaviour in terms of mass loss over time with the largest difference observed in the size of initial mass decrease, which is particularly pronounced once the rate of mass loss is plotted against temperature (Fig. 1). The peak observed at 100 °C corresponds to evaporation of water and other volatile compounds and the difference observed between samples is most likely related to moisture content in the peat samples. Following a short induction period (plateau), a steady decrease in mass was observed in both samples as the temperature was increased above 200 °C, slowing down at approximately 500 °C and reaching completion and then a constant mass above 700 °C.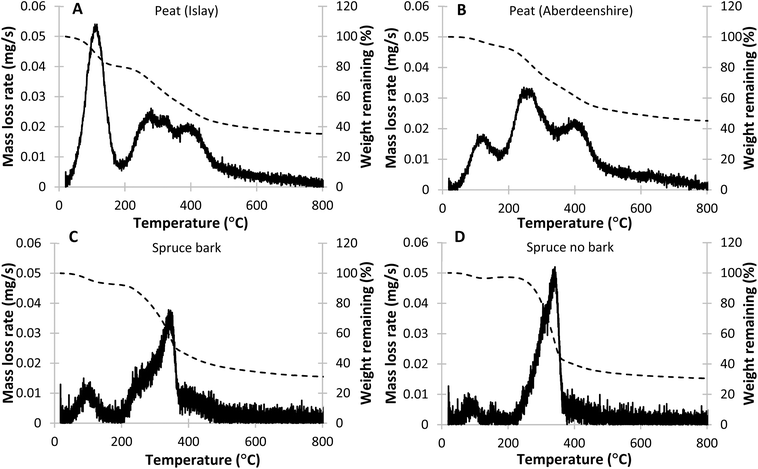 | ||
| Fig. 1 TGA mass loss rate against temperature plots for Islay peat (A), Aberdeenshire peat (B), Spruce with (C) and without bark (D). | ||
Plotting the rate of mass loss reveals additional detail. Three maxima can be observed in the Islay peat sample (excluding the one at 100 °C) at 280 °C, 325 °C, 400 °C, corresponding to decomposition of hemicellulose, cellulose and lignin respectively.24,26,48 In the Aberdeenshire peat sample, the hemicellulose and cellulose peaks appear to coalesce at a maximum temperature of 260 °C, while the lignin peak appears similar to that observed in Islay peat, with a maximum at 400 °C. Once the contribution from water is removed, the total mass lost in the observed range originating from cellulose and hemicellulose is 28% of dry mass for Islay peat and 26% for Aberdeenshire peat (Table 1). While it is difficult to distinguish between hemicellulose and cellulose contributions due to proximity of the maxima it can be deduced that contribution of hemicellulose is higher in Aberdeenshire peat, compared to the Islay peat samples, as the highest mass loss rate is observed at a lower temperature. Lignin decomposition appears to occur in two distinct stages, where the first corresponds to a quick loss of mass with a maximum mass loss rate at 400 °C, followed by a slow decrease in mass from 500 °C to approximately 700 °C. The weight lost corresponding to decomposition of lignin in Islay peat is 28% of dry mass and 24% in Aberdeenshire peat. These results show that while there are certainly differences between the composition of biopolymers and their contribution to overall mass of the material, our peat samples are broadly similar to each other in terms of thermal behaviour.
The thermal decomposition profiles of spruce wood with and without bark appear very different to that observed for peat (Fig. 1). The initial mass loss corresponding to water and volatiles evaporation is much lower, indicating less moisture content in the material. The majority of the mass loss occurs rapidly above 200 °C, slowing down significantly at approximately 350 °C, with decomposition seemingly complete at approximately 700 °C. The sample containing bark appears broadly similar, however analysis of the mass loss rate graph reveals some differences, compared to the sample without bark. While the spruce with no bark appears to only have one mass loss rate maximum at 340 °C, the sample containing bark has a noticeable “hump” at 250 °C, showing that contribution from hemicellulose to the overall mass of the sample is comparatively more significant. Additionally, while the peak corresponding to degradation of lignin is not as distinct as in the peat samples, some contribution from lignin decomposition can be noted. The mass loss rate corresponding to lignin decomposition appears larger in the spruce bark sample than in the no-bark sample, however it is possible that the additional contribution originates from suberin decomposition, which occurs in bark and shows similar thermal decomposition temperature range as lignin.49 Overall, the combined contribution of cellulose and hemicellulose decomposition is 57% of dry mass in the sample without bark and 48% in the sample with bark, while the portion of mass lost above 370 °C, corresponding to polyphenolic species and suberin, is 11% of dry mass in the former sample and 18% in the latter (Table 2). These values reflect larger contributions from polycarbohydrates and lower from lignin in wood, compared to peat, which is likely to have an effect on released compounds and therefore the final flavour profile. The results obtained in this study agree with what has been found previously for spruce wood at a smaller scale (5 mg).50 This shows that while the same biopolymers are present in both peat and wood, their distribution is very different, which likely results in differences in composition of the smoke.
| Sample | Mass lost (% of dry mass) | ||||
|---|---|---|---|---|---|
| Hemicellulose | Cellulose | Lignin | Tail | Sum | |
| PI | 17.0 | 11.3 | 19.2 | 9.1 | 56.7 |
| PA | 26.3 | 16.5 | 7.9 | 50.7 | |
| SNB | 57.2 | 7.1 | 4.2 | 68.6 | |
| SB | 18.8 | 29.7 | 11.1 | 7.2 | 66.8 |
3.2 PyGC–MS of peat and spruce wood
Use of PyGCMS enabled identification of a large number of peat pyrolysates (97 for Islay peat, 119 for Aberdeenshire peat) and spruce wood pyrolysates (108 for sample with bark, 79 for sample with no bark). For the full list of identified compounds refer to the SI. Several groups of compounds, including short and long chain alkanes, alkenes as well as their various aliphatic derivatives, namely organic acids, alcohols and ketones were detected. Of particular interest to our research are aromatic species, anhydrous sugars and furans. These compounds originate primarily from decomposition of lignin, cellulose and hemicellulose, and often have low sensory thresholds, thus contributing significantly to aroma perception of the smoke and smoked beverages.51 For this research, the compounds of interest were divided into four groups. Three of these groups primarily arise from the degradation of lignin: aromatic species without hydroxy or methoxy side groups; phenolic species without methoxy side groups and methoxyphenolic species. The final of the four groups are the anhydrous sugars and furan derivatives which primarily originate from decomposition of cellulose and hemicellulose.Methoxyphenolic compounds and those originating from thermal degradation of carbohydrates form the largest groups of compounds identified, with 32 and 34 identified compounds, respectively. Looking at the distribution of pyrolysates in spruce wood and peat (Fig. 2), it is clear that a significant portion of compounds is found only in peat. That is especially true for compounds originating from decomposition of cellulose and hemicellulose, for which 19 molecules were found only in peat, while 10 can be found in both peat and wood pyrolysis products. For all other aromatic species, including phenolic and methoxyphenolic species, nearly half of identified compounds can be found in at least one peat sample and at least one wood sample pyrolysates. These include important congeners, which have been previously connected to smoky aroma in peated whisky, such as guaiacol, 4-ethylguaiacol, p-cresol, 4-vinylguaiacol, 4-ethyl-2-methylphenol.52
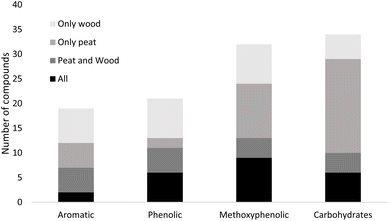 | ||
| Fig. 2 Number of compounds in selected categories found only in wood, only in peat, in at least one sample of both wood and peat and in all samples. | ||
The divergence in terms of aromatic composition is most likely the result of differences in the structure of lignin in peat and spruce wood. One parameter which may be used to measure this is the ratio of S and G units, related to ratio of pyrolysis products with one (G derivatives) or two (S derivatives) methoxy side groups. Here, we measured the ratios using either all identified products or only specific ones (syringol, 4-methylsyringol, 4-vinylsyringol and trans-4-propenylsyringol for S units, guaiacol, 4-methylguaiacol, 4-vinylguaiacol, trans-isoeugenol for G units) as carried out previously by Nunes et al. for Eucalyptus-derived lignin.53 Both methods gave comparable results (Fig. 2) with S/G values obtained using selected compounds being 0.04–0.07 higher for all samples, except for PI, which was 0.08 lower. Measured S/G ratio for each sample was below 1.0 signifying that guaiacyl units are dominant in the lignin structure, however, values for peat were between 0.60 and 0.72, while those for spruce wood measured between 0.07 and 0.13. This shows that while all materials tested contain lignin rich in guaiacyl units, spruce wood shows an even greater dominance of these, compared to syringyl units, as would be expected for softwood. Interestingly, the S/G ratio of samples with and without bark are nearly the same, showing that presence of bark doesn't have a significant impact. S/G ratio observed for peats from different locations showed a difference of approximately 20%, which could be of practical significance, but any specific conclusions are difficult to make with only two geographically distinct samples (Fig. 3).
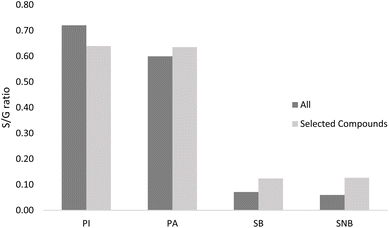 | ||
| Fig. 3 S/G ratios of analysed samples based on all detected compounds and on a selection of compounds. PI – Islay peat; PA – Aberdeenshire peat; SB – spruce with bark; ANB – spruce without bark. | ||
3.3 Sensory analysis
The mean panel scores for each sample per sensory attribute are shown in Table S10 and also as flavour profiles in Fig. 4. Statistically significant differences between the samples were noted for four attributes: overall peat intensity, burnt, smoky and meaty aromas. The peated new make spirit samples were assigned higher scores in the first three of these categories, while spruce smoked new make spirit had a stronger “meaty” aroma. These results suggest that the spruce smoked new make spirits contained less phenolic compounds, which are associated with peaty notes. However, many simpler phenolics such as cresols, phenol and ethylphenols are usually associated with the “medicinal” characteristic, which was similar in intensity in all samples. On the other hand, guaiacol and its derivatives are strongly connected to the smoky characteristic and thus may be expected in lower concentration. These results show that some aspects of the smoky aroma of peated spirit can be replicated using another material as a source of smoke, though the exact sensory profile will likely differ in terms of intensity of specific notes, owing to potential differences in chemical composition. | ||
| Fig. 4 Mean panel scores plotted as flavour profiles (* indicates a statistically significant difference between samples). | ||
Importantly, there were no additional panellist comments regarding the aroma of the samples and thus no off notes were detected in the new make spirits produced using malt smoked with spruce wood. This observation aligns well with instrumental analysis, which revealed no unexpected compounds in the thermal decomposition products, which could produce unwanted aromas or flavours. This shows that such techniques can be used successfully to assess whether the produced spirit will contain compounds responsible for potentially undesirable sensory notes even though using instrumental methods to predict sensory qualities is challenging.
While there were some differences between the replicates for both the peated and spruce smoked samples, in general, the peated NMS samples were more burnt, smokier and more intensely peaty than the spruce wood smoked samples. That said, the spruce smoked samples still produced a distinct smoky sensory experience, without using peat. The sensory panellists did not report any unusual aromas or off-notes present in the spruce wood smoked samples and none of the other characteristics showed statistically significant differences between spruce smoked and peated samples.
3.4 NMR analysis of new make spirits
Analysis of peated new make spirit has been covered in a previous publication, therefore the discussion here will focus on comparison between peated and spruce smoked samples.32 Additionally, the behaviour and composition of different distillation fractions will be discussed. Three distillate fractions of peated and spruce smoked new make spirits were analysed, together with low wines. Fraction 2 was selected to be the main focus of the study, with differences between fractions noted when relevant. All frequencies given in the text refer to this second fraction, though it should be noted that slight variation was observed between samples due to differences in ethanol concentration. For the purpose of the analysis the NMR spectrum was divided into three regions: low frequency region (0–3 ppm), middle frequency region (3–6 ppm) and high frequency/aromatic region (above 6 ppm). Most signals in the spruce-smoked and peated new make spirit spectra were the same or very similar (Fig. 5). Some signals in the spectrum originate from the DSS standard used, namely that at 0 ppm, used for calibration of spectra and rough quantitation of selected compounds, this standard also generates multiplets at 0.59 ppm, 1.77 ppm, 2.01 ppm and 2.82 ppm.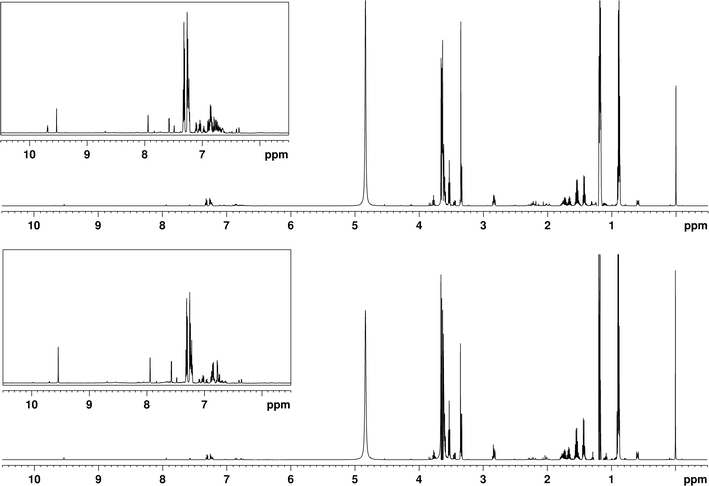 | ||
| Fig. 5 1H NMR spectra of peated (bottom) and spruce smoked (top) new make spirits (distillation fraction 2), recorded using a 600 MHz instrument. | ||
The low frequency region consists primarily of signals originating from alkyl groups of alcohol and carbonyls and does not differ significantly between samples using different smoke sources for the most part, though some changes were observed. One such case is the doublet at 1.31 ppm, which displays a much larger intensity in the spruce-smoked samples, compared to peated ones. This difference is particularly pronounced in the early distillate fraction, where the signal is most intense. The signal could correspond to either 1,1-diethoxyethane CH3 or lactic acid CH3, with the former generally being found in matured whisky, while the latter can be a product of fermentation where lactic acid bacteria are present.54,55 That said, similar behaviour can be observed for the quartet at 4.12 ppm, which reinforces the notion that they both originate from lactic acid. Similar differences in intensity were also observed for acetate signal at 2.06 ppm and acetaldehyde doublet at 2.23 ppm. This shows that concentrations of some compounds other than aromatic species are also affected by choice of smoke source, in particular those of acids, esters and aldehydes. On the other hand, some groups of compounds are largely unaffected, e.g., alcohols. The most prominent difference between fractions was observed for signals of higher alcohols, often referred to as fusel alcohols or fusel oils (Fig. 6), which include 3-methyl-1-butanol (1.43 ppm quartet, 1.66 ppm nonet, 3.60 ppm triplet), n-propanol (1.54 ppm sextet, 3.53 ppm triplet) and isobutanol (1.73 pm nonet, 3.34 ppm doublet). Comparing intensities of the signals clearly shows that as the distillation progresses the concentration of aforementioned alcohols decreases. First fraction showed concentrations 2–3 times higher than the main distillate, while the signals in the last fraction were in some cases 100 times less intense. This confirms that amount of fusel alcohols can be adjusted by modifying the distillation procedure and also shows that this process could be monitored using in-line or at-line NMR, possibly even at low field strengths given large intensity of signals of interest.
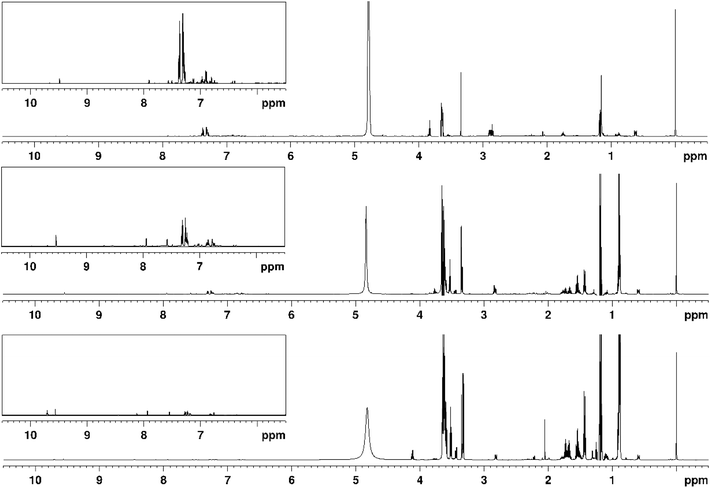 | ||
| Fig. 6 1H NMR spectra of peated spirit distillation fractions 1, 2 and 3 (from bottom to top), recorded at 600 MHz. | ||
Signals corresponding to alkyl side groups of aromatic species can also be noted, including those of o-cresol (2.18 ppm), p-cresol (2.26 ppm), 2-phenylethanol (2.83 ppm) and 2-phenylethyl acetate (2.93 ppm). These compounds do not differ significantly between samples in terms of signal intensity and no clear trend is visible between them. Interestingly, the sample with lowest ethanol concentration (last distillation fraction) shows a clear separation between DSS and 2-phenylethanol signals, which reveals a triplet at 2.86 ppm, confirming presence of the compound. Overall, the low frequency region of the spectrum provides much information on the composition of the spirit. The presence of specific signals and their intensities may be correlated with changes that occur during the distillation process and therefore could be used in monitoring. The impact of the source of smoke on this region appears to be minimal with only a small selection of signals being related to congeners originating from smoke.
The middle frequency region shows a limited number of signals and almost no differences between spruce smoked and peated samples, with the exception of two groups of overlapping multiplets at 3.47 ppm and 3.78 ppm. In both cases the signals are relatively stronger in the spruce-smoked samples, with the difference being particularly visible in the first fraction. This region of the spectrum consists of signals corresponding to protons in proximity of electronegative atoms, such as oxygen. This includes methoxy groups, ethers and esters. A possible assignment for the triplet at 3.78 ppm is phenylethanol, however no corresponding change in signal intensity was observed in the aromatic region, meaning that the signal likely originates from a similar, currently unidentified compound. Opposite behaviour was observed for the methoxy singlet of guaiacol at 3.82 ppm, potentially signifying decreased concentration of methoxyphenols in spruce smoked spirit, which could explain the weaker smoky characteristic noted by the sensory panel. Other signals in the range under consideration, show small variations in intensity between samples, however the differences are not nearly as large as those discussed earlier and most likely arise as a result of variation in spirit preparation processes, rather than alteration of the smoke source. Possible exceptions to this are signals corresponding to hemiacetals originating from acetaldehyde, which show higher concentration in spruce-smoked samples, this correlating well with the observed increased concentration of acetaldehyde. Strong signals of higher alcohols described previously can be observed here, following the same pattern in terms of signal intensity between different distillation fractions. Additionally, the singlet indicating presence of methanol was identified at 3.35 ppm. The intensity of the signal and thus the concentration does not appear to change significantly between distillation fractions, which is somewhat unexpected given the behaviour of other alcohols.
In the aromatic region we find the greatest differences between samples. The most striking feature of the region is how nearly all signals increase in intensity with distillation progress. Taking the phenylethanol signals as examples, their concentration in middle fraction is approximately eight times higher than in the first fraction, while the concentration in the third fraction is twelve times higher. Similar observations can be made for other phenolics in the region, which include o-cresol, p-cresol and phenol. This clearly shows the impact of distillation on the concentration of phenolic species, regardless of their origin (as phenylethanol is a fermentation product, rather than a smoke component). It also shows how choosing the cutoff point of the distillation can help manage abundance and thus the flavour of the spirit. Not all signals in the region follow this trend however, with the prime example being furfural. Corresponding signals at 6.77 ppm, 7.57 ppm, 7.94 ppm and 9.54 ppm all show highest intensity in the second distillation fraction, approximately two times larger than either of the other fractions. This in turn highlights that modifying the distillation procedure impacts different groups of compounds in different ways, implying that one cannot optimise the process for all groups of compounds at the same time.
When it comes to comparison between new make spirits produced using peat and spruce wood the signal distribution and intensities are quite similar for the first and last fraction irrespective of the smoke source, but the main fraction displays interesting changes. Overall, the intensity of observable signals of aromatic species is larger in spruce smoked samples, corresponding to increased concentration of cresols, phenol and phenylethanol. Additionally, some signals not present in peated new make spirit can be found in the spruce smoked sample, including those at 6.80 ppm and 6.90 ppm. Extensive overlap in the region makes identification of these peaks particularly difficult. These observations may appear to be contradictory to what was noted by the sensory panel, given general association of phenolic compounds with peatiness, however the majority of the phenolics observed in the NMR spectra would be expected to affect the medicinal characteristic, which was given similar scores by the panel. Additionally, no significant off notes were produced, which correlates well to lack of signals in unexpected regions of the spectrum, which was the case for some other alternative materials tested previously.32 Another difference of note is the potentially impactful lack of particular signals in the spruce-smoked sample, such as those at 8.00 ppm, 8.05 ppm and 8.15 ppm, likely corresponding to heteroaromatic species. These could be potentially undesirable at high concentrations due to their unpleasant aroma. That said, the compounds have been found in peated new make spirit and thus their absence could have an impact on the flavour and aroma of the spirit. Finally, as noted before the signal corresponding to acetaldehyde is much stronger in spruce smoked samples.
3.5 FT-ICR-MS of new make spirits
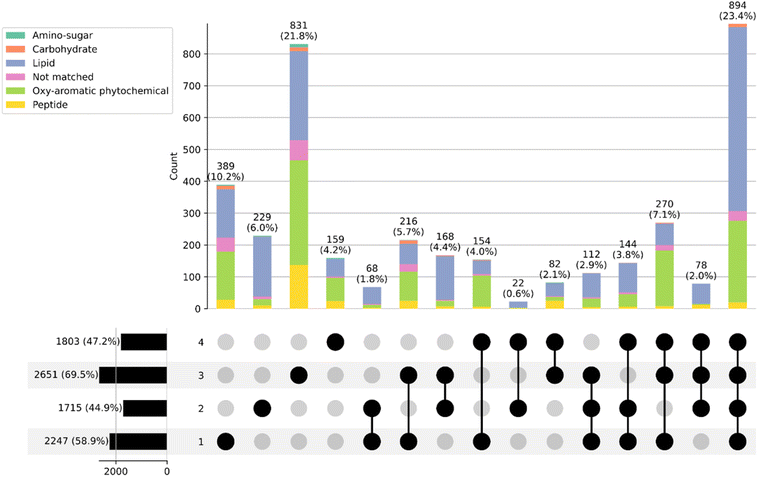
The number of assigned compounds and associated metrics such as aromaticity index (AI), mean CHONS and compound class for each samples examined are listed in Table S1. In terms of average number of assigned molecular formulae, spruce smoked NMS second fraction had the highest (2651), while fraction 1 had the lowest (1715), demonstrating the complexity of the main distillation fraction. Considering sulfur, the low wines had a much higher amount of S per formula than any of the spirit distillation fractions. The low wines also held a percentage of sulfur-containing compounds that is almost double that of other samples. This matches what is expected as sulphurous compounds are generally undesirable.56 AI increases in the later distillation fractions, signifying increased presence of aromatic species, which correlates well with what was found using NMR. There is an increase in mean N in the NMS compared to other samples and this is reflected in the % CHON compounds. Examining the compound class % breakdown, the increase in amino sugar-like and peptide-like compounds in these samples explains this increase in N. The lower AI for fraction 1 is demonstrated by the lower % lignin or tannin-like compounds (21%) in this sample compared to the rest (34–38%).To compare the similarity in compound make-up of each fraction, UpSet analysis was conducted with the resulting plot show in Fig. 7.
There are 894 molecular formulae found in all four fractions, showing almost half of each sample share the same compounds, which can be assigned to lipid-like and aromatic-like. This is interesting as it does show that there is a base level of compounds that are present after the first distillation and remain present in any cut in the second distillation. Examining the unique compounds, the NMS contains the most unique (831), followed by the low wines (389), and these are made of almost identical splits of lipid and aromatic like compounds, however with more peptide like compounds in the NMS. The unique compounds of fraction 2 stick out, while there are only 229 of them, they are almost all lipid-like.
Comparing peated and spruce NMS, the profiles are different, with the main differences summarised in Table 3. Both spirits contain similar numbers of assigned formulae but peated NMS has higher S content and lower, N, AI and subsequently lower amino sugars, peptides and tannin/lignan like compounds than the spruce spirit, with sugars and lipids being higher. This, together with the NMR results, shows that the phenolic species in spruce smoked new make spirit are more abundant in terms of concentration and number of compounds than peated NMS. It is therefore surprising that the sensory analysis showed weaker peaty characteristics. The difference in intensity of smoky notes most likely arises as a result of lower concentration of guaiacol species and shows the number of phenolics or their summed concentration may not be the most accurate criteria to evaluate contribution to peaty character.
| NMS | Assigned formula | Mean S | Mean N | AI | Lipids% | Sugars % | Amino sugars % | Peptides % | Tannin/lignins % |
|---|---|---|---|---|---|---|---|---|---|
| Peated | 2700 | 0.15 | 0.20 | 0.07 | 60.89 | 3.15 | 0.33 | 5.19 | 23.00 |
| Spruce | 2651 | 0.08 | 0.55 | 0.12 | 49.19 | 1.51 | 0.53 | 9.06 | 34.36 |
4. Conclusions
Thermal degradation behaviour of spruce and peat was analysed, along with the chemical composition and sensory characteristics of new make spirit fractions produced using smoke from these materials. TGA and pyGCMS results showed that both materials, while made primarily of the same natural polymers, differ in terms of their distribution, which results in differences in the variety and concentration of species released upon thermal degradation. In particular, greater abundance of methoxyphenolic species was produced from peat, which is likely related to greater contribution of syringyl units to its lignin structure. While all samples tested showed dominance of guaiacyl compounds in the thermal decomposition products (and thus their dominance in the lignin), the difference in abundance of the different building blocks was more pronounced in the wood samples. The differences between different peat samples and spruce wood with and without bark were generally minimal, though spruce wood with bark showed presence of additional compounds, compared to the sample without bark. Despite the differences, many of the desired compounds, including guaiacols, cresols and other species connected to smoky aromas and flavours were identified both in peat and spruce wood, suggesting that the latter may be a suitable candidate for replacing/supplementing use of peat.This was further confirmed by the results of NMR analysis of new make spirits, which showed little difference between the spectra of the peated and spruce smoked samples, both in terms of compounds identified and their concentrations, including phenols. Some differences were noted, however, one of most significant being the decreased concentration of guaiacol in the spruce smoked sample, suggesting that the spirit may contain less methoxyphenolic species and thus have weaker “smoky” aroma and flavour, while notes connected to other phenolic species would likely be comparable. Similar observations were made using FT-ICR-MS, which surprisingly revealed greater abundance of aromatic species in the spruce smoked new make spirit, though the exact nature of the compounds could not be established. Analysis of different new make spirit fractions also revealed that as the distillation progresses the concentration of phenolic species increases, while that of higher alcohols decreases. This confirms that by adjusting the distillation parameters the composition of the final spirit can be influenced, which could make up for the differences related to using an alternative smoke source. Any such changes should be made with caution, as other aspects of the spirit are likely to be affected, as shown by how different groups of compounds display varying behaviour with distillation progress.
The analytical results align well with what was found by the sensory panel, which identified burnt, smoky and medicinal characteristics in the spruce smoked new make spirit, though the first two were not as intense as the peated sample. Nonetheless, it appears possible to replicate aspects of the smokiness of peated spirit with an alternative material with no undesirable off notes. The results do not preclude the potential inclusion of spruce wood smoked malt in the mash bill to impart gentle smoky flavours to NMS, reducing the amount of peat required, but this would require further investigation regarding whether changing smoking and distillation conditions could improve the results or whether mixing peat and wood could produce even better results. As such, future work in the area would likely be focused on addressing these questions, in addition to identifying other potential candidates for peat substitution.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: additional experimental details and data for FT-ICR-MS, TGA, pyGC-MS, NMR as well as details on new make spirit preparation. See DOI: https://doi.org/10.1039/d5fb00560d.Acknowledgements
The authors would like to thank the Carnegie Trust for Universities of Scotland for funding as well as Lorna Murray, Juraj Bella and Andrew Hall for maintaining the NMR facility at The University of Edinburgh.References
- B. Guthrie, J. Beauchamp, A. Buettner, S. Toth and M. Qian, Sex, Smoke, and Spirits the Role of Chemistry, American Chemical Society, Washington, DC, 2019 Search PubMed.
- Islay.com, Islay Whisky Making, Distilling on Islay, Peat, https://islay.com/islay-whisky-making/peat/, 2022.
- T. A. Bringhurst and J. Brosnan, in Whisky, Elsevier Ltd, 2nd edn, 2015, ch. 6, pp. 49–122 Search PubMed.
- K. Y. M. Lee, A. Paterson, J. R. Piggott and G. D. Richardson, J. Inst. Brew., 2001, 107, 287–313 CrossRef.
- T. J. Kelly, I. T. Lawson and L. E. S. Cole, in Encyclopedia of Geochemistry: A Comprehensive Reference Source on the Chemistry of the Earth, ed. W. M. White, Springer International Publishing, Cham, 2018, pp. 1197–1200, DOI:10.1007/978-3-319-39312-4_187.
- H. Biester, K. H. Knorr, J. Schellekens, A. Basler and Y. M. Hermanns, Biogeosciences, 2014, 11, 2691–2707 CrossRef CAS.
- G. Vinci, P. Mazzei, M. Drosos, C. Zaccone and A. Piccolo, Chem. Biol. Technol. Agric., 2020, 7, 1–11 Search PubMed.
- A. J. Simpson, D. J. McNally and M. J. Simpson, Prog. Nucl. Magn. Reson. Spectrosc., 2011, 58, 97–175 CrossRef CAS.
- B. M. Harrison and F. G. Priest, J. Agric. Food Chem., 2009, 57, 2385–2391 CrossRef CAS.
- M. M. Nassar and G. D. M. Mackay, Wood Fiber Sci., 1984, 16, 441–453 CAS.
- H. Yang, R. Yan, H. Chen, C. Zheng, D. H. Lee and D. T. Liang, Energy Fuels, 2006, 20, 388–393 CrossRef CAS.
- J. Schellekens, P. Buurman and T. W. Kuyper, Soil Biol. Biochem., 2012, 53, 32–42 CrossRef CAS.
- K. Krakowiak, R. D. McIntosh and D. Ellis, Sustainable Food Technol., 2024, 2, 92–103 RSC.
- Y. Arlen-Pouliot and N. Bhiry, Holocene, 2005, 15, 408–419 CrossRef.
- M. R. Turetsky and V. L. St. Louis, in Boreal Peatland Ecosystems, ed. R. K. Wieder and D. H. Vitt, Springer Berlin Heidelberg, Berlin, Heidelberg, 2006, pp. 359–379, DOI:10.1007/978-3-540-31913-9_16.
- The Scottish Government, Environment and Forestry Directorate, Ending the Sale of Peat in Scotland: Analysis of Consultation Responses, 2023.
- IUCN UK, Peatland Programme, https://www.iucn-uk-peatlandprogramme.org/, 2022.
- Department for Environment, Food and Rural Affairs, Sale of Horticultural Peat to Be Banned in Move to Protect England's Precious Peatlands, 2022.
- R. Jung, M. Karabín, L. Jelínek and P. Dostálek, Eur. Food Res. Technol., 2023, 249, 33–45 CrossRef CAS.
- R. Prado, M. Gastl and T. Becker, Compr. Rev. Food Sci. Food Saf., 2021, 20, 4816–4840 CrossRef CAS.
- R. C. Pettersen, in The Chemistry of Solid Wood, ed. R. M. Rowell, ACS, Washington D.C., 1984, vol. 20, ch. 2, pp. 57–126 Search PubMed.
- E. Sjöström, Wood Chemistry : Fundamentals and Applications, Academic Press, New York, 1981 Search PubMed.
- J. A. Maga, 6000 Broken Sound Parkway NW, Suite 300, Smoke in Food Processing, CRC Press, 1st edn, 1988 Search PubMed.
- M. Asmadi, H. Kawamoto and S. Saka, J. Anal. Appl. Pyrolysis, 2011, 92, 417–425 CrossRef CAS.
- A. Alshehri, C. Dunn, C. Freeman, S. Hugron, T. G. Jones and L. Rochefort, Front. Environ. Sci., 2020, 8, 48 CrossRef.
- D. K. Shen and S. Gu, Bioresour. Technol., 2009, 100, 6496–6504 CrossRef CAS PubMed.
- H. M. Ng, N. M. Saidi, F. S. Omar, K. Ramesh, S. Ramesh and S. Bashir, in Encyclopedia of Polymer Science and Technology, 2018, pp. 1–29, DOI:10.1002/0471440264.pst667.
- R. K. Sharma and M. R. Hajaligol, J. Anal. Appl. Pyrolysis, 2003, 66, 123–144 CrossRef CAS.
- F.-X. Collard and J. Blin, Renew. Sustain. Energy Rev., 2014, 38, 594–608 CrossRef CAS.
- AMCTB No. 85 and Analytical Methods Committee, Anal. Methods, 2018, 1, 5463–5467 Search PubMed.
- W. Kew, I. Goodall and D. Uhrin, Food Chem., 2019, 298, 8 CrossRef PubMed.
- K. P. Krakowiak, I. Baxter, B. Harrison, N. Pitts, S. Fergusson, N. G. A. Bell, D. Ellis and R. D. McIntosh, Sustain. Food Technol., 2024, 2, 1747–1756 RSC.
- W. Kew, N. G. A. Bell, I. Goodall and D. Uhrín, Magn. Reson. Chem., 2017, 55, 785–796 CrossRef CAS.
- W. Kew, I. Goodall, D. Clarke and D. Uhrín, J. Am. Soc. Mass Spectrom., 2017, 28, 200–213 CrossRef CAS.
- J. S. Garcia, B. G. Vaz, Y. E. Corilo, C. F. Ramires, S. A. Saraiva, G. B. Sanvido, E. M. Schmidt, D. R. J. Maia, R. G. Cosso, J. J. Zacca and M. N. Eberlin, Food Res. Int., 2013, 51, 98–106 CrossRef CAS.
- F. Jack, in Whisky and Other Spirits, ed. I. Russell, G. G. Stewart and J. Kellershohn, Academic Press, 3rd edn, 2022, pp. 321–333, DOI:10.1016/B978-0-12-822076-4.00012-7.
- S. C. Moldoveanu, in Pyrolysis of Organic Molecules, ed. S. C. Moldoveanu, Elsevier, 2rd edn, 2019, DOI:10.1016/B978-0-444-64000-0.00004-4, ch. 4, pp. 207–278.
- S. C. Moldoveanu, in Analytical Pyrolysis of Natural Organic Polymers, ed. S. C. Moldoveanu, Elsevier, 2rd edn, 2021, vol. 20, ch. 6, pp. 283–309 Search PubMed.
- P. J. Linstrom and W. G. Mallard, J. Chem. Eng. Data, 2001, 46, 1059–1063 CrossRef CAS.
- S. C. Moldoveanu, in Analytical Pyrolysis of Natural Organic Polymers, ed. S. C. Moldoveanu, Elsevier, 2rd edn, 2021, vol. 20, ch. 4, pp. 111–269 Search PubMed.
- K. Krakowiak, R. D. McIntosh and D. Ellis, Measurement of Beverage ABV by Benchtop NMR Spectroscopy, Oxford Instruments, 2023 Search PubMed.
- ChemicalBook, Chemical Book, https://www.chemicalbook.com/, 2022.
- I, John Wiley & Sons, https://spectrabase.com/, 2022.
- T. Y. T. Saito, K. Hayamizu, M. Yanagisawa, O. Yamamoto, Spectral Database for Organic Compounds SDBS, https://sdbs.db.aist.go.jp/, 2022.
- D. S. Wishart, A. Guo, E. Oler, F. Wang, A. Anjum, H. Peters, R. Dizon, Z. Sayeeda, S. Tian, B. L. Lee, M. Berjanskii, R. Mah, M. Yamamoto, J. Jovel, C. Torres-Calzada, M. Hiebert-Giesbrecht, V. W. Lui, D. Varshavi, D. Varshavi, D. Allen, D. Arndt, N. Khetarpal, A. Sivakumaran, K. Harford, S. Sanford, K. Yee, X. Cao, Z. Budinski, J. Liigand, L. Zhang, J. Zheng, R. Mandal, N. Karu, M. Dambrova, H. B. Schiöth, R. Greiner and V. Gautam, Nucleic Acids Res., 2022, 50, D622–D631 CrossRef CAS PubMed.
- H. E. Gottlieb, V. Kotlyar and A. Nudelman, J. Org. Chem., 1997, 62, 7512–7515 CrossRef CAS PubMed.
- C. Roullier-Gall, J. Signoret, D. Hemmler, M. A. Witting, B. Kanawati, B. Schäfer, R. D. Gougeon and P. Schmitt-Kopplin, Front. Chem., 2018, 6, 29 CrossRef.
- C. Di Blasi, Prog. Energy Combust. Sci., 2008, 34, 47–90 CrossRef CAS.
- F. Ghonjizade-Samani, L. Haurie, R. Malet and V. Realinho, Materials, 2023, 16, 3829 CrossRef CAS PubMed.
- H. Flity, M. Abdo, L. Terrei, Z. Acem, R. Mehaddi, P. Lardet and G. Parent, Fire Saf. J., 2023, 141, 103979 CrossRef.
- K. Y. M. Lee, A. Paterson, J. R. Piggott and G. D. Richardson, J. Inst. Brew., 2000, 106, 287–294 CrossRef CAS.
- H. H. Jeleń, M. Majcher and A. Szwengiel, Food Sci. Technol., 2019, 107, 56–63 Search PubMed.
- C. A. Nunes, C. F. Lima, L. C. A. Barbosa, J. L. Colodette, A. F. G. Gouveia and F. O. Silvério, Bioresour. Technol., 2010, 101, 4056–4061 CrossRef CAS PubMed.
- J. Conner, in Whisky, Elsevier Ltd, 2nd edn, 2015, ch. 11, pp. 199–220, DOI:10.1016/B978-0-12-401735-1.00011-8.
- N. R. Wilson, The Effect of Lactic Acid Bacteria on Congener Composition and Sensory Characteristics of Scotch Malt Whisky, Heriot-Watt University, 2010 Search PubMed.
- A. Wanikawa and T. Sugimoto, Molecules, 2022, 27, 1672 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |