Optimization of pre-drying concentration and type of foaming agents to produce shelf-stable black mulberry juice powder using the foam mat drying technique
Received
2nd September 2025
, Accepted 26th January 2026
First published on 13th February 2026
Abstract
In today's world, consumers increasingly demand shelf-stable products with longer shelf life and high levels of natural bioactive compounds. Mulberry (Morus nigra L.) juice powder is a cheap alternative at the industrial scale for the development of instant products with significant bioactive composition. This study explores the application of underutilized mulberry fruit juice through the foam mat drying process using plant-based and dairy-based alternatives. The carrier type and concentration of the foaming agents were optimized for the production of high-quality foam mat dried black mulberry juice powder. A whey protein isolate (WPI), soy protein isolate (SPI) and WPI:maltodextrin blend (WPI:M) were used for foam mat drying at concentrations of 2–10%. The effect of these carriers on the physicochemical properties, bioactive retention and subsequent powder functionality was determined. The results revealed that WPI-treated mulberry powder (8–10%) had higher solubility (68–71%), lower moisture content and improved flow properties. By contrast, lower concentrations of WPI-treated mulberry powder (2–4%) contributed to the preservation of antioxidant activity (65.3%), phenolics (180.6 mg GAE/100 g) and flavonoids (363.6 mg QE/100 g). For the blend of WPI:M, a 10% concentration offered a balanced formulation which had good solubility (64.9%) and anthocyanin stability (192.1 mg/100 g). FTIR spectroscopy confirmed the presence of phenols, proteins and anthocyanins in both T2 (WPI) and T6 (SPI). The WPI (T2)-treated powder demonstrated superior morphology and mineral distribution, along with stronger encapsulation, as compared to SPI (T6). Overall, powder quality was strongly dependent on the carrier type and concentration. WPI offered the most favorable functional attributes, SPI served as an effective plant-based alternative, and the WPI:M blend provided enhanced stability of the bioactive components. These insights allow formulation strategies to be tailored to specific industrial and nutritional requirements for the production of mulberry juice powder.
Sustainability spotlight
Consumers in today's world demand shelf-stable products with extended shelf life while retaining natural bioactive composition. Given that fruit has a short shelf-life, mulberry juice powder is an inexpensive, industrial-scale alternative for the development of instant products that are rich in natural bioactive components. This study explores the industrial-scale development of this cheap but underutilized mulberry fruit juice through the sustainable foam mat drying process using plant- and dairy-based alternatives.
|
1 Introduction
Mulberry (Morus spp.) has gained increasing scientific recognition due to its rich composition of bioactive compounds, including anthocyanins, flavonoids, and ascorbic acid.1,2 These phytochemicals contribute to a wide range of health-promoting effects, which include antioxidant, anti-inflammatory, antidiabetic, hepatoprotective, and neuroprotective effects. Black mulberry (Morus nigra L.) is particularly notable for its high level of cyanidin-3-O-rutinoside; an anthocyanin compound responsible for intense pigmentation and strong free radical-scavenging properties, thus serving as a promising nutraceutical ingredient for the development of functional foods.3,4 The phytochemical and health-promoting components of mulberry find use in a variety of products, such as syrups, wines, teas, squash, jams, jellies, yogurts, vinegar, biscuits and cosmetic products.5 Mulberry juice and beverages are new to the consumer market and have attained popularity because of the attractive color, pleasant taste and great antioxidant properties. Nevertheless, mulberry juice is highly perishable, and its heat-sensitive nutrients are prone to decay. During processing and storage, anthocyanins, vitamin C, and other phenolic compounds are easily degraded by oxidation, enzymatic activity and microorganisms. These problems reduce its distribution and shelf life significantly and limit its industrial use, despite its nutritional value. Thus, to enhance stability, reduce transportation expenses, and expand the scope of its use in processed food manufacturing, the conversion of the mulberry juice into a shelf-stable powder has proven to be an efficient approach.6–8
Several drying technologies, such as spray drying, freeze drying, vacuum drying, and hot-air drying, are commonly applied for producing fruit juice powders, yet they have notable limitations. Spray-drying is a cost-effective and widely used method, but it can cause degradation of heat-sensitive pigments. Moreover, high levels of carrier agents are required to counteract stickiness due to the high sugar content of the mulberry juice.9 Similarly, hot-air or cabinet drying can lead to prolonged thermal exposure, resulting in pigment darkening, reduced solubility, and lower-quality powders.10 On the other hand, freeze drying is an effective way to preserve such heat-sensitive pigments, but it is expensive, energy-intensive, and cannot be used in large-scale commercial production.11 These limitations have accelerated interest in alternative technologies capable of preserving sensitive bioactive compounds. Novel drying techniques offer several advantages, including reduced thermal degradation, improved rehydration ratio, lower risk of microbial contamination, decreased bulk density and weight, and minimized oxidative damage, thereby preserving the texture, color, nutritional quality, and bioactive constituents of fruit juice powders.12 Foam-mat drying has emerged as an effective technique for processing liquid and semi-liquid fruit products, particularly those containing heat-sensitive components. In this method, foaming agents are used to whip the juice into a foam-like layer, and then dried at low temperatures in thin layers.13,14 This method is, however, dependent on the stability and expansion of the foam, which is determined by the nature of the foaming agent as well as the concentration of pre-drying solids.
Foaming agents commonly employed in the drying of fruit juices include proteins such as whey protein and egg albumin, as well as polysaccharides such as maltodextrin and carboxymethyl cellulose (CMC). These agents are selected based on the drying method employed and the desired physicochemical and functional properties of the product.15 Polyphenol compounds can easily interact with proteins to form complexes that may affect the structural properties, functional behavior, and nutritional properties of these components. Key factors governing these interactions include processing temperature, pH, protein source and concentration, as well as the chemical nature and structural features of the phenolic compounds involved.16 Plant-based foaming proteins, such as SPI, are highly effective in forming viscoelastic foams, which are more stable and offer better protection to the antioxidant compounds, whereas high concentrations of polysaccharides, such as gum arabic, although less expensive, reduce the solubility of antioxidant compounds by increasing the viscosity.17 Similarly, CMC helps to increase foam stability and preserve heat-sensitive nutrients, including anthocyanins, but, in large quantities, has adverse effects on reconstitution characteristics and mouthfeel. Lipid-based agents such as glycerol monostearates enhance foam structure but may reduce solubility at higher concentrations.18,19 Therefore, foaming agents are important as they improve the drying kinetics, foam stability, and bioactive retention, but their concentration, type, and interaction with processing environments must be carefully optimized to balance functional performance with nutritional quality and sensory acceptability. Thus, the current research focuses on the concentration and the choice of foaming agents to produce a high-quality, shelf-stable black mulberry juice powder using the foam-mat drying method. This approach aims to maximize the use of the underutilized mulberry resources and enable the development of useful and antioxidant fruit powders for industrial purposes.
2 Experimental
2.1 Materials
Mulberry fruits were gathered directly from mulberry plants at LPU, Phagwara. Whey protein isolate and soya protein isolate were purchased from Asitis Nutrition (Gratus Life Science Pvt. Ltd, Bengaluru, India), and maltodextrin was obtained from LOBA-CHEMIE Pvt. Ltd, Mumbai, Maharashtra.
2.2 Foam preparation
The fruits were cleaned properly and its juice was extracted by squeezing using a muslin cloth (pore size: 2 mm), and kept in deep freezer (−18 °C) for further research. The mulberry juice was mixed with variable concentrations of whey protein isolate (2–10 g/100 mL), soya protein isolate (2–10 g/100 mL), and maltodextrin (2–10 g/100 mL). Further, the formulations were freshly whipped using a hand blender (Primesky, milk frother, wattage: 1200.00), according to the method of Khatri and team (2024).20 The samples were then subjected to foaming and physicochemical property analysis.
2.3 Drying method
Mulberry juice mixed with different concentration of foaming agents was spread in a 5 mm thick layer on borosilicate glass Petri dishes (120 × 200 mm). It was then dried in a hot-air oven (Bio Techno Lab, Mumbai, Maharashtra) at 60 °C with an air flow of 2 m s−1. After drying, the material was gently scraped off, ground into a fine powder using a mortar and pestle, and sealed in 0.5 mm thick plastic pouches covered with laminated wraps. The powder was then stored in a deep freezer (Blue Star CHF300, Mumbai) at −10 °C for later use. Fig. 1 represents the flowchart and unit operations involved in the development of foam mat dried mulberry juice powder.
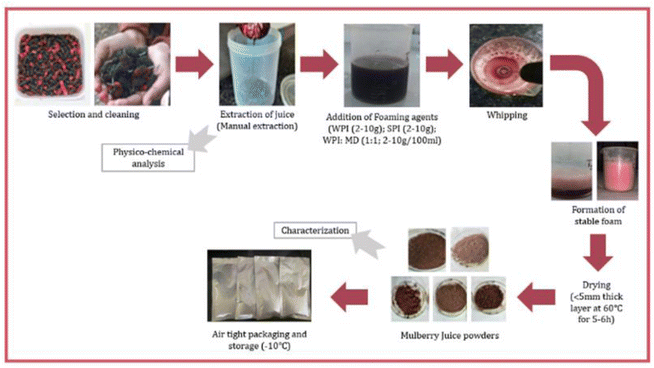 |
| | Fig. 1 Process flowchart for the development of the foam mat dried mulberry juice powder. | |
2.4 Foaming properties
2.4.1 Foam expansion. Foam expansion (FE) was measured using a modified version of the method outlined by Kandasamy et al. (2012).21 Mulberry juice was whipped for 5 minutes with a handheld Primesky milk frother (4.5 W), which is used for foaming beverages. The increase in foam volume was calculated by comparing the volume before (V0) and after whipping (V1).
| Foam expansion = [V1 − V0/V0] × 100 |
2.4.2 Foam stability. The foam stability (FS) was determined as previously described by Marinova et al. (2021).22 The variation in foam volume was evaluated after resting for 2 h at room temperature, with the foam stability reported as the final foam volume in percentage of its initial foam volume.
2.4.3 Foam density. Foam density was measured according to the procedure described by Falade et al. (2003).23 The foam (50 mL) was transferred into a pre-weighed glass beaker using a spatula. The mass of foam was then recorded, and foam density was calculated and expressed as g cm−3.
| Foam density = (mass of the foam)/(volume of the foam) |
2.5 Physicochemical characteristics
2.5.1 Yield. The powder yield was calculated by weighing the dried fruit juice sample using the formula:
| Powder yield (%) = W2/W1 × 100 |
where W2 is the weight of the dried juice powder and W1 is the initial weight of the fresh juice used.
2.5.2 Bulk density (BD, kg m−3). Bulk density is a significant parameter in the measurement of flowability, packing capacity, and utilization efficiency of a powder in various formulations. These properties have a significant impact on storage, transportation, and reconstitution behavior. In the current study, 1 gram of dried mulberry juice foam mat powder was loosely filled in a 10 mL graduated cylinder without exerting pressure on the sample or compacting the sample. The powder volume was measured, and the bulk density was calculated using the formula:
| BD (kg m−3) = (mass of powder in the cylinder (g))/(volume occupied (mL)) |
2.5.3 Tapped density (TD, kg m−3). To determine tapped density, the same graduated cylinder used for bulk density was dropped from a consistent height of 10 cm multiple times (100 taps) until no volume change occurred. The final volume was recorded, and the tapped density was calculated as:
| TD (kg m−3) = (mass of the powder in the cylinder (g) tapped 100 times)/(volume occupied (mL)) |
2.5.4 Carr's index (CI, %). Carr's index is used to measure the compressibility of the powders and indicates their behavior when flowing. It is calculated from the bulk and tapped density values:
| % CI = (tapped density − bulk density)/(tapped density) × 100 |
2.5.5 Hausner ratio (HR). The Hausner ratio is an important parameter utilized to quantify powder cohesiveness and flowability. It is calculated as the ratio of bulk density to tapped density:
| Hausner ratio (HR) = TD/BD |
2.5.6 Color. Color measurement of mulberry powder was carried out using a HunterLab colorimeter (Konica Minolta CR 410), calibrated with the standard white reference tile included with the apparatus. The sample was placed in a glass Petri plate beneath the lens in order to measure its color. The lens's nose cone was positioned in a manner such that the sample's surface was exactly perpendicular to the light source, and readings were noted.
2.5.7 Total soluble solids. The total soluble solid (TSS; °Bx) content of the mulberry fruit powder was determined, as described by Ranganna (2009).24 Mulberry powder (2 g) was taken and suspended in 20 mL of distilled water. The sample was mixed well to ensure complete dissolution. The combined mixture was then filtered over Whatman filter paper to remove any undissolved residues. TSS content was measured by analyzing a few drops of the filtrate placed onto a digital refractometer, and the mean value of three replicates was determined.
2.5.8 Ash. A known weight of the sample was placed in a pre-weighed crucible. The sample was first ignited over a flame until it was charred and no fumes were evolved. After that, the charred crucibles were burned at 550 °C for 5 hours via a muffle furnace to ensure organic material was completely burned out. Following incineration, the crucibles were allowed to cool in a desiccator to avoid adsorption of moisture. The ash content was determined and expressed in terms of percent ash:
| Ash% = (weight of ash)/(weight of sample) × 100 |
2.5.9 Water activity. Water activity (aw) is the unbound free water content of a product and a key parameter of its chemical and microbial stability. Low aw values are most often found to be associated with good shelf life and product stability. Water activity of foam mat dried mulberry juice powder in this research was measured using a calibrated water activity meter (NOVASINA LabSwift-aw). The powder sample was evenly distributed across the sample holder to ensure uniform exposure to the water activity meter and prevent gaps and surface irregularities that would invalidate the readings. The sample holder was placed in the measuring chamber of the instrument and allowed to equilibrate. Each sample was scanned three times, and the mean aw value was recorded.
2.5.10 pH. The sample mulberry powder (2 g) was taken in a beaker, 20 mL of distilled water was added, and the mixture was homogenized by stirring thoroughly. The pH was measured with a calibrated pH meter by dipping an electrode within the sample mixture, and was allowed to stabilize prior to noting the value. This procedure was repeated for three replicates in order to determine the accuracy of the results.
2.5.11 Titratable acidity. The titratable acidity of mulberry powder was measured using the method of Ranganna (2009).24 The powder (2 g) was dissolved in 100 mL of distilled water, and the solution was well mixed to ensure optical clarity. A 25 mL aliquot was taken from the mixture, and 2–3 drops of phenolphthalein indicator were added. Titration was carried out with 0.1 N sodium hydroxide (NaOH), which was taken in a burette. The titration was continued until a persistent pink color appeared and lasted for 30 seconds, which showed the endpoint. The acidity percentage was calculated using the formula:
| Acidity% = (titre value × normality of alkali × equivalent weight of acid × volume made up)/(weight of sample for estimation × aliquot taken for estimation × 1000) × 100 |
2.5.12 Ascorbic acid. The titrimetric method was performed using 2,6-dichlorophenol-indophenol dye to measure the ascorbic acid content in mulberry fruit. This was diluted (100 mL) into a known volume of 3% metaphosphoric acid solution. A dilution of the extract was titrated against a 2,6-dichlorophenol-indophenol standardized dye solution until a faint pink color wasobserved. To standardize the dye solution, it was titrated into a standard solution of ascorbic acid that had 0.1 mg of L-ascorbic acid per milliliter of 3% HPO3 solution, and dye factor was calculated. The ascorbic acid content of the mulberry fruit sample was then determined and expressed as milligrams/100 grams of the sample.24
| Dye factor = 0.5/(titre value) × 100 |
| Ascorbic acid (mg/100 g) = (titre value × dye factor × volume made up)/(aliquot of extract taken × weight/volume of the sample) × 100 |
2.5.13 Total anthocyanin content. The total anthocyanin content (TAC) was measured in compliance with the method reported by Shehata et al. (2020).25 For this, 1 g of mulberry powder was mixed with 5 mL of 95% ethanolic HCl, then stored at 4 °C overnight. Filtration of the mixture was done using Whatman No. 1 filter paper. The obtained extract was kept in the dark for 2 hours before obtaining the optical density at 535 nm with a visible-light spectrophotometer (HVGPS System, Ahmedabad).
| Total optical density = (optical density value at 535 nm × volume made for color estimation × total volume)/(mL of aliquot taken for estimation × weight of the sample) × 100 |
| Total anthocyanin content (mg/100 g) = (total optical density)/98.2 × 100 |
2.6 Functional properties
2.6.1 Hygroscopicity. The moisture absorption capacity was determined as the moisture absorbed by 1 gram of mulberry juice powder. The powder was placed in a sealed container with a saturated sodium carbonate (Na2CO3) solution to keep the humidity steady. After a certain time, the weight gain was recorded to calculate the moisture absorbed, using the method described by Cai and Corke (2000).26
2.6.2 Solubility. Mulberry juice powder (0.5 g) was dispersed in 10 mL of distilled water and then centrifuged at 3000 rpm (REMI R-8C BL, ILE Company, Chennai). The precipitate was dried in a hot-air oven (Bio Techno Lab, Mumbai) at 105 ± 5 °C for 5 h. The solubility was calculated from the mass of the dried residue, as per the method described by Hogekamp and H. Schubert (2003).27
2.6.3 Total phenolic content. The total phenol content of mulberry juice powder was measured in accordance with the Folin–Ciocalteu method.28 Two grams of powder was homogenized with 80% ethanol and centrifuged at 1200 rpm for 20 min. The supernatant was transferred to glass tubes. To 1 mL of extract, Folin–Ciocalteu reagent (0.5 mL) was added. After 3 minutes, 2 mL of 20% sodium carbonate (Na2CO3) was added with thorough mixing. The test tubes were placed into a boiling water bath for 1 minute, then cooled. The absorbance at 650 nm of the final blue-colored material was determined by measuring against a water blank with a spectrophotometer.The calibration curve was created using gallic acid as a standard. A stock solution of gallic acid was prepared by adding 0.5 g to 100 mL of distilled water. Aliquots (0–10 mL) of stock solution were then transferred to volumetric flask and diluted with distilled water. From each, 1 mL was transferred to a separate 100 mL volumetric flask, and 60 mL of water was added, followed by Folin–Ciocalteu reagent (5 mL) and 20% Na2CO3 (15 mL) in order. The volume was made up to 100 mL with distilled water. After 2 hours, the absorbance was measured at 650 nm and used for the creation of the standard curve. The total phenol content in the mulberry sample was determined from the curve and expressed as mg GAE/100 g of sample.
2.6.4 Total flavonoid content. The total flavonoid content of the mulberry juice powder was analyzed using a visible-light spectrophotometer (HVGPS System, Ahmedabad) by the colorimetric method.25 Stock rutin solution was prepared from rutin (≥94% purity), and stock quercetin solution was prepared from quercetin (≥95% purity), both initially dissolved in 80% methanol to obtain 1 mg mL−1 solution. Different concentrations (100–500 µL) of standards were diluted 1 mL distilled water and 1 mL of 2% aluminum chloride and prolonged during dark for one hour. The absorbance was measured at 415 nm. TFC was calculated from the standard curve and expressed as mg RE/100 g and mg QUE/100 g.
2.6.5 Antioxidant activity. The antioxidant activity of mulberry juice powder was evaluated using DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging method.29 A 0.5 g of powder was mixed with 3 mL of methanol before filtration through Whatman No. 1 filter paper. DPPH solution (3.9 mL) was added to the filtrate, after which the mixture was kept for 30 min in the dark. The absorbance measurements at 515 nm were performed using the HVGPS System (Ahmedabad) visible-light spectrophotometer, with a methanolic DPPH solution as a blank solution.
| Antioxidant activity = (optical density of blank − optical density of sample)/(optical density of blank) × 100 |
2.6.6 Oil absorption capacity. To study the oil absorption properties, 0.5 g of mulberry powder was added to 10 mL of soybean oil (specific gravity 0.09092). The solution was mixed for 2 min using a vortex mixer (Remi CM 101, Praxor Instruments and Scientific Co., Chennai), then centrifuged at 3000 rpm for 30 min (REMI R-8C BL, ILE Company, Chennai). The filtrate was then filtered through Whatman No. 1 filter paper. The oil absorption capacity was measured and expressed in terms of oil retained by the powder on a gram basis, using the method explained by Egbuonu (2015).30
| OAC (g g−1) = (final weight (g) − initial weight (g))/(initial weight (g)) × 100 |
2.6.7 Water absorption capacity. To assess water absorption capacity, 0.5 g of mulberry juice powder was dispersed in 10 mL of distilled water and vortexed for 2 minutes using a vortex mixer (Remi CM 101, Praxor Instruments and Scientific Co., Chennai). The mixture was then centrifuged at 3000 rpm for 30 min using a centrifuge (REMI R-8C BL, ILE Company, Chennai). The supernatant was then filtered through Whatman No. 1 filter paper. The water absorption capacity was calculated and reported as grams of water retained per gram of powder.30
| WAC (g g−1) = (final weight (g) − initial weight (g))/(initial weight (g)) × 100 |
2.7 FTIR
Qualitative analysis of the mulberry powder samples was conducted using an FTIR analyzer (PerkinElmer) equipped with Diamond ATR and a KBr beam splitter. 5 mg of potassium bromide (KBr) was mixed with 5 mg of each sample. The FTIR spectra were obtained in the range 400 to 4000 cm−1.
2.8 FE-SEM
The shape and structure of the dried mulberry juice powder foam mat were examined using FE-SEM. To make the samples electrically conductive, a thin layer of gold was applied by sputter coating. The microscopic examination was carried out at magnifications of 2000–4000×, 4000–6000×, 6000–8000× and 8000–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×.
000×.
2.9 Statistical analysis
Triplicate observations of the parameters of dried mulberry powder were run, and the mean and standard deviation values were calculated in Excel. The significance among treatments was evaluated using Minitab Statistical Software (version 19.1); the statistically significant differences were assessed using one-way ANOVA with a critical difference of p < 0.05.
3 Results and discussion
3.1 Foaming properties of foam mat dried mulberry juice powder
3.1.1 Foam expansion. In this study, the foaming properties of foam mat-dried mulberry juice powder were evaluated using whey protein isolate (T1–T5), soy protein isolate (T6–T10), and whey protein isolate–maltodextrin blends (T11–T15) as foaming agents. The foam expansion (volume increase incurred on air incorporation) appeared highly differentiated across foaming agents. Whey protein samples foamed significantly better as concentration increased, with foam expansion increasing from 50% (T1) to 150% (T2), and 300% (T3), and reaching a maximum of 400% (T4), before decreasing slightly to 380% (T5). This trend showed the surface-active superiority of whey proteins with respect to their superior air incorporation and foam volume.31 Notably, the low pH and the presence of anthocyanins in mulberry juice lead to unfolding of the whey proteins, increasing their interfacial activity and enhancing the solubility. It has been demonstrated that acidic fruit matrices enhance WPI denaturation and hydrophobic group exposure, thus enhancing foaming activity.32 Mulberry sugars and pectic substances also synergistically stabilize adsorbed whey protein, which contributes to the high expansion.In contrast, the foam expansion values of the soy protein samples varied in a more moderate way from 30% in T6 to 80% in T7, 140% in T8 to 200% in T9, with a slight decrease to 180% in T10. This behavior is consistent with the moderate foaming capacity of soy protein reported by Farid et al. (2023), which was attributed to its globular structure, with formation of foams optimized at maximum concentration.33 Additionally, hydrophobic and hydrogen-bond interactions enable polyphenols in mulberry juice, including quercetin and anthocyanins, to interact with soy protein, resulting in SPI–polyphenol complexes.34 This may slightly inhibit protein flexibility at the air–water interface, thereby reducing the foam expansion compared to WPI. Another study conducted by Wu et al. (2024) also found that variations in the structure and functionality of SPI–anthocyanin complexes, depending on the anthocyanin type/concentration, can affect the foaming or interfacial behavior.35 The foam expansion with T11 to T15 ranged from 40 to 160% in the whey protein isolate:maltodextrin blend samples. The results indicated that maltodextrin increased the system viscosity and enabled air-bubble retention, while it failed to confer foam volume enhancement beyond that achieved with whey protein alone, similar to previous observations by Chandrakar et al. (2024).36 Maltodextrin can react with soluble sugars and organic acids in mulberry juice, and increase viscosity to enhance bubble retention but decrease the mobility of WPI at the interface. This might have led to lower foam expansion than achieved using pure WPI formulations.37
The interaction behavior of polyphenols with soy protein isolate (SPI) and whey protein isolate (WPI) differs due to fundamental variations in protein structure, conformational flexibility, and response to acidic, polyphenol-rich environments. SPI mainly consists of globular storage proteins, which possess relatively rigid tertiary structures with defined hydrophobic domains.38 In the presence of mulberry polyphenols, including anthocyanins, SPI readily forms non-covalent complexes through hydrogen bonding and hydrophobic interactions between phenolic hydroxyl groups and aromatic amino acid residues.39 These interactions result in the formation of SPI–polyphenol complexes, which increase molecular rigidity and limit protein unfolding at the air–water interface. In contrast, WPI exhibits greater conformational adaptability and surface activity. Under the acidic conditions typical of fruit juices, whey proteins undergo partial denaturation, which enhances the exposure of hydrophobic groups and increases their affinity for the air–water interface.40 Instead, polyphenols, along with endogenous sugars and pectic substances present in mulberry juice, can synergistically stabilize the unfolded whey protein layer at the interface, reinforcing foam structure without impairing protein mobility.41
3.1.2 Foam stability. The foam stability of the whey protein isolate (WPI) samples ranged from 50 to 53% (T1–T4), with a slight decrease to 51% at higher protein concentrations (T5). This observation is consistent with the study by Parmar et al. (2020), which showed that enhanced stability arises from whey proteins forming viscoelastic films that are resistant to bubble collapse.31 Sugars and organic acids present in mulberry juice probably contributed to increasing the WPI film elasticity through protein unfolding and the development of hydrogen bonds with the protein matrix that stabilize natural foams. The anthocyanins may have acted by enhancing the viscosity and interlocking with the network of proteins, which slows down the process of liquid drainage.42 Moderate foam stability (T6–T9: 48%, T9: 52%) was reported for the SPI samples, consistent with the observation of Farid et al. (2023), wherein soy proteins make slightly less elastic films than whey proteins and hence foam stability is somewhat lower.33 Phenolic compounds may bind soy protein in SPI–mulberry systems, and this increases the rigidity of the interfacial layer, but decreases the film elasticity, which accounts for intermediate foam stability.Foam stability of 45.00% (T12) to 52.31% (T13) in whey protein isolate–maltodextrin blends varied depending on the balance of maltodextrin as a viscosity-enhancing agent and whey protein as a film-forming agent. Hajiaghaei and Sharifi (2022) reported that while maltodextrin increased viscosity and slowed liquid drainage from foam, excessive maltodextrin destabilized the foam.43 Maltodextrin prevents pigment–protein interactions in mulberry juice by binding free water, which prevents over-hydration of the protein film and positively affects stability. Nevertheless, excessive MD may limit the flexibility of the WPI and decrease film strength at the interface. Similarly, Ozcelik and Kulozik (2023) evaluated the effect of maltodextrin concentration on the stability of anthocyanins and noted that maltodextrin is effective in protecting anthocyanin stability.44 This suggests that maltodextrin can stabilize anthocyanins, ascorbic acid, and color, among other quality parameters in fruit juices.
3.1.3 Foam density. Protein concentration was found to be a key parameter affecting drying efficiency in a foam-mat drying process and was indicated to influence foam density, which decreased with increasing protein concentration. However, WPI samples showed a dramatic drop in foam density, from 0.57 kg m−3 (T1) to 0.15 kg m−3 (T4), suggesting that significantly lighter foams were generated at higher protein concentrations. The observation of lower foam densities and elevated drying rates at higher WPI concentrations in foam mat dried papaya pulp parallels the results of this work.31 The decrease in foam density can be associated with the reaction of WPI with mulberry organic acids, which enhances the partial denaturation and film expansion that entraps more air and decreases density.45Soy protein isolate samples exhibited a declining foam density from 0.66 kg m−3 for T6 to 0.26 kg m−3 for T9, showing that SPI had a moderate ability to reduce density as concentration increased. Higher SPI concentrations in foam-mat dried strawberry powder enhanced foam structure and reduced density, as SPI acts to form cohesive protein networks that entrap air effectively.33 Foam densities from 0.29 kg m−3 (T15) to 0.64 kg m−3 (T11) were measured with the WPI–maltodextrin (MD) blend samples, which demonstrated that the combination of WPI and MD can reduce foam density. The conjugates between WPI and MD reported by Chandrakar et al. (2024) improved foam stability and decreased density, attributed to the increased viscosity by MD and the WPI's ability to foam, rendering them appropriate for efficient applications involving drying.36 The MD in mulberry matrices competes with sugars in binding water, raising the viscosity and enhancing bubble retention but reducing protein spreading at the interface, explaining the moderate foam density reduction compared to pure WPI formulations.46
3.2 Physicochemical parameters of foam mat dried mulberry juice powder
3.2.1 Yield. In the present study, powder yields varied between 8.13 ± 0.23% and 20.5 ± 0.5%, with WPI treatments (T1–T5) yielding from 10.1 ± 0.28% to 20.5 ± 0.5%, SPI treatments (T6–T10) from 8.13 ± 0.23% to 19.8 ± 0.1% and whey protein isolate:maltodextrin blend treatments (T11–T15) of 10.4 ± 0.05%. The results are in agreement with those reported by Shishir et al. (2016), wherein the powder yield of spray-dried pink guava increased with optimized drying and formulation parameters, showing the critical effect of carrier concentration and type on the final product recovery.47 Table 1 lists various physicochemical parameters of the foam mat dried mulberry juice powder.
Table 1 Physicochemical characteristics of foam mat dried mulberry juice powdera
| Treatments |
Yield (g/100 g) |
L* |
a* |
b* |
Water activity |
TSS (°Bx) |
pH |
Titratable acidity (g/100 g) |
Ash (g/100 g) |
The results are presented as the mean of triplicates ± standard deviation (SD). WPI: whey protein isolate; SPI: soya protein isolate; WPI:M: whey protein isolate:maltodextrin (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), [T1 = 2 g/100 mL WPI, T2 = 4 g/100 mL WPI, T3 = 6 g/100 mL WPI, T4 = 8 g/100 mL WPI, T5 = 10 g/100 mL WPI, T6 = 2 g/100 mL SPI, T7 = 4 g/100 mL SPI, T8 = 6 g/100 mL SPI, T9 = 8 g/100 mL SPI, T10 = 10 g/100 mL SPI, WPI:M T11 = 2 g/100 mL WPI:M, T12 = 4 g/100 mL WPI:M, T13 = 6 g/100 mL WPI:M, T14 = 8 g/100 mL WPI:M, and T15 = 10 g/100 mL WPI:M]. 1), [T1 = 2 g/100 mL WPI, T2 = 4 g/100 mL WPI, T3 = 6 g/100 mL WPI, T4 = 8 g/100 mL WPI, T5 = 10 g/100 mL WPI, T6 = 2 g/100 mL SPI, T7 = 4 g/100 mL SPI, T8 = 6 g/100 mL SPI, T9 = 8 g/100 mL SPI, T10 = 10 g/100 mL SPI, WPI:M T11 = 2 g/100 mL WPI:M, T12 = 4 g/100 mL WPI:M, T13 = 6 g/100 mL WPI:M, T14 = 8 g/100 mL WPI:M, and T15 = 10 g/100 mL WPI:M]. |
| T1 |
10.12 ± 0.28fg |
32.45 ± 0.06c |
2.27 ± 0.03d |
1.5 ± 0.01d |
0.337 ± 0.005b |
12.10 ± 0.0d |
4.5 ± 0.5d |
1.913 ± 0.01a |
4.16 ± 0.28c |
| T2 |
13 ± 0.11def |
38.7 ± 0.81b |
3.30 ± 0.15c |
3.20 ± 0.34c |
0.326 ± 0.005bc |
12.60 ± 0.11d |
4.62 ± 0.01d |
1.523 ± 0.04abc |
5.16 ± 0.02bc |
| T3 |
14.92 ± 0.05cd |
37.8 ± 0.16b |
4.51 ± 0.20ba |
4.39 ± 0.22b |
0.278 ± 0.002de |
13 ± 0.05cd |
4.97 ± 0.01cd |
1.286 ± 0.01cdef |
6.33 ± 0.28ab |
| T4 |
17.84 ± 0.1abc |
38.3 ± 0.41b |
3.95 ± 0.07b |
3.57 ± 0.03c |
0.269 ± 0.001fg |
13.78 ± 0.11cd |
5.06 ± 0.17c |
0.966 ± 0.05fg |
6.66 ± 0.3a |
| T5 |
20.56 ± 0.5a |
42.4 ± 0.15a |
4.65 ± 0.09a |
5.13 ± 0.04a |
0.242 ± 0.005h |
18.65 ± 0.0a |
5.26 ± 0.17bc |
0.833 ± 0.05g |
6.83 ± 0.28a |
| T6 |
8.13 ± 0.23g |
34.5 ± 0.23d |
3.25 ± 0.02c |
2.02 ± 0.04c |
0.335 ± 0.001b |
11.27 ± 0.11e |
4.95 ± 0.01cd |
1.918 ± 0.05abcd |
4.33 ± 0.28c |
| T7 |
12 ± 0.6def |
37.59 ± 0.34c |
3.37 ± 0.05c |
2.6 ± 0.26c |
0.321 ± 0.005bc |
13.12 ± 0.05cd |
5.2 ± 0.005bc |
1.285 ± 0.05bcde |
4.83 ± 0.30c |
| T8 |
14.15 ± 0.1de |
39.53 ± 0.04b |
4.34 ± 0.04a |
4.2 ± 0.05a |
0.309 ± 0.002cd |
14 ± 0.02cd |
5.45 ± 0.005ab |
1.289 ± 0.11def |
4.93 ± 0.30c |
| T9 |
16.90 ± 0.2abc |
38.39 ± 0.18a |
3.93 ± 0.03b |
3.3 ± 0.05b |
0.290 ± 0.009de |
14.31 ± 0.11cd |
5.6 ± 0.11a |
0.960 ± 0.02fg |
5.20 ± 0.11bc |
| T10 |
19.80 ± 0.1ab |
39.55 ± 0.28a |
3.70 ± 0.26b |
3.91 ± 0.20b |
0.271 ± 0.018ef |
15.80 ± 0.11bc |
5.8 ± 0.11ab |
0.642 ± 0.05g |
6.16 ± 0.26abc |
| T11 |
10.47 ± 0.05fg |
33.54 ± 0.31e |
3.17 ± 0.03d |
1.11 ± 0.01d |
0.387 ± 0.005a |
11.76 ± 0.17e |
4.66 ± 0.02d |
1.763 ± 0.02ab |
4.50 ± 0.06c |
| T12 |
12.22 ± 0.20def |
36.41 ± 0.24d |
4.00 ± 0.06b |
3.40 ± 0.03c |
0.325 ± 0.002bc |
12.90 ± 0.17d |
4.88 ± 0.02cd |
1.283 ± 0.06cdef |
5.50 ± 0.5c |
| T13 |
14.55 ± 0.1cd |
35.54 ± 0.25c |
3.61 ± 0.16b |
3.45 ± 0.17c |
0.313 ± 0.002cd |
13.66 ± 0.02cd |
5.39 ± 0.01bc |
0.933 ± 0.05fg |
5.56 ± 0.57abc |
| T14 |
15.41 ± 0.05bcd |
37.47 ± 0.41b |
3.49 ± 0.08c |
3.09 ± 0.05c |
0.268 ± 0.005fg |
16.62 ± 0.11ab |
5.41 ± 0.01abc |
0.912 ± 0.05fg |
5.93 ± 0.40abc |
| T15 |
18.11 ± 0.1abc |
42.29 ± 0.44a |
5.01 ± 0.17a |
6.49 ± 0.02a |
0.267 ± 0.009g |
17.38 ± 0.17ab |
5.6 ± 0.001a |
0.640 ± 0.005g |
6.10 ± 0.17abc |
3.2.2 Bulk density. The treated samples showed decrease bulk density values that decreased from 0.71 ± 0.02 to 0.52 ± 0.09 g cm−3 as the WPI concentration increased; the SPI treated (T6–T10) samples ranged from 0.7 ± 0.011 to 0.52 ± 0.001 g cm−3, and the whey protein isolate:maltodextrin blends (T11–T15) ranged from 0.67 ± 0.020 to 0.55 ± 0.001 g cm−3. The results obtained in this study are in accordance with those observed by Aisyah et al. (2024), where they recorded a correlation of decreasing bulk density with increasing foaming agent concentration, indicating improvement in porosity and foam structure of the powdered products.48
3.2.3 Tapped density. Across all treatments, the tapped density of the foam mat-dried mulberry powder decreased with increasing foaming agent concentration: WPI (0.90 ± 0.03 to 0.75 ± 0.009 g cm−3), SPI (0.87 ± 0.002 to 0.73 ± 0.011 g cm−3) and WPI:M (0.86 ± 0.004 to 0.72 ± 0.009 g cm−3). The reduction is largely attributed to the greater foaming ability at greater concentrations, leading to formation of more stable air bubbles in the matrix during drying, thereby increasing the porosity and decreasing the particle packing density. Therefore, the structure of the powder is more open, with a lower mass of bulk material per unit volume. This behaviour is in agreement with the study by Khatri et al. (2023), where an increase in the amount of foaming agent reduced the tapped density.20
3.2.4 Carr's index. According to standard classifications, Carr's Index (CI) values below 15% are considered to have excellent fluidity, values from 15% to 20% are defined as good fluidity, values from 20 to 35% are average fluidity, values from 35 to 45% are poor fluidity, and values greater than 45% are considered very poor fluidity. The study indicates that foam mat dried mulberry powders prepared with the foaming agents WPI, SPI and WPI:M at varying concentrations, have an increasing CI trend. The CI for whey was between 17.40 ± 0.5% and 27.70 ± 0.1%, 20.0 ± 0.2% and 30.6 ± 0.1% in the case of soy, and 19.20 ± 0.02% and 30 ± 0.37% for (WPI:M). These results are consistent with the findings of Jakubczyk et al. (2011), who reported that increasing the foaming agent concentration can alter the powder flow properties. Moderate increases in foaming agent concentration may reduce the Carr Index (CI) and improve flowability, but excessive concentrations lead to a deterioration in the flow properties.49
3.2.5 Hausner ratio. The Hausner ratio (HR) is frequently used to evaluate the flowability of foam mat dried powders. Values in the range of 1.0–1.10 are indicative of free-flowing powders, whereas values above 1.25 represent poor flowability. For the present study, the HR values varied between 1.21 and 1.42 for different foaming agents and concentrations. The HR values for the treatments with WPI ranged between 1.21 and 1.36, with SPI between 1.22 and 1.42 and the WPI:M combination between 1.40 and 1.42, suggesting more cohesive, less free-flowing powders at a higher foaming concentration. The results agree with the findings of Jakubczyk et al. (2011), who found an increase in HR with increasing levels of foaming agent and carrier (maltodextrin) present in apple puree powders, also indicating that the addition of carrier and foam incorporation inhibits powder flowability.49
3.2.6 Color. Increasing the concentration of the foaming agent significantly enhanced the color retention in the foam mat dried mulberry juice powder. For treatments T1–T5, where whey protein was used as the foaming agent, the L* (lightness) values ranged from 32.45 ± 0.06 to 42.40 ± 0.15, a* (red-green) values from 2.27 ± 0.03 to 4.65 ± 0.09, and b* (yellow-blue) values from 1.50 ± 0.01 to 5.13 ± 0.04. Formulations T6–T10, prepared using soy-based foaming systems, exhibited L* values between 34.50 ± 0.23 and 39.55 ± 0.28, a* values from 3.25 ± 0.02 to 4.34 ± 0.04, and b* values ranging from 2.02 ± 0.04 to 4.20 ± 0.05, indicating moderate color preservation compared to whey-based formulations. In contrast, treatments T11–T15, incorporating a WPI–maltodextrin blend, demonstrated superior color retention. Among these, T15 showed the highest values for the color parameters (L* = 42.29 ± 0.44, a* = 5.01 ± 0.17, b* = 6.49 ± 0.02), highlighting the synergistic effect of protein-carrier combinations in stabilizing pigments during foam-mat drying.The improvement in color properties with the increase in foaming-agent concentration can be explained by the increase in the stability of the foam, the decrease in thermal exposure, and the formation of protective matrices around the heat- and oxygen-sensitive pigments. This trend aligns with the findings of Belal et al. (2023), who indicated the optimum color stability of foam-mat-dried tomato powder at higher concentrations of foaming agents.50 Similarly, Chaipoot et al. (2024) reported enhanced color retention in honey-bee-brood protein powder dried using foam-mat technique, as the concentrations of carboxymethyl cellulose and glycerol monostearate increased.51 All these findings confirm that the type, concentration, and compositional synergy of the foaming agents are crucial factors in defining the visual characteristics of foam-mat-dried products.
3.2.7 Total soluble solids. The TSS concentration of the foam-mat-dried mulberry juice powders increases progressively with foaming-agent content, especially in formulations with whey protein isolate (WPI). The TSS values increased with WPI concentration up to 12.1 °Bx (T1) to 18.6 °Bx (T5), and the same trend was observed to a slightly smaller degree with powders prepared using the soy protein isolate (SPI) and WPI–maltodextrin mixtures. Such an increase in TSS has been attributed to the addition of solid foaming agents introduced into the system, and less residual moisture left after drying, resulting in an increased concentration of soluble solids in the powder bed. The same trend was reported for papaya and guava pulp drying, for which an increase in TSS was observed with increasing concentration of foaming agents in the foam-mat dried powder.52
3.2.8 Ash content. The high foaming-agent concentration increased the ash content of whey protein isolate-based treatments (T1–T5), varying from 4.16 ± 0.28 to 6.80 ± 0.28 g/100 g. Similarly, values of 4.30 ± 0.28 to 6.10 ± 0.26 g/100 g were found for soy protein isolate (T6–T10). Ash contents of the blend of whey protein isolate:maltodextrin (T11–T15) ranged from 4.50 ± 0.06 to 6.10 ± 0.17 g/100 g. The increased amount of ash may be a result of the gradual addition of foaming agents in the formulation, due to the decrease in the moisture content during drying, which concentrates the inorganic constituents in the final powder. These results are consistent with the findings of Khatri (2024), who reported that ash content in foam-mat-dried black mulberry juice powder increased with the concentration of foaming agent.20
3.2.9 Water activity. The addition of foaming agents led to a gradual decrease in the water activity (aw) of the foam mat-dried mulberry juice powders of all formulations. In powders prepared using whey protein isolate (WPI), the water activity was noted as 0.337 ± 0.005 at 2% foaming agent concentration, which reduced to 0.242 ± 0.005 at 10% foaming agent concentration. Likewise, there was a decrease from 0.335 ± 0.001 to 0.271 ± 0.009 between soy protein isolate (SPI) and powder made with WPI and maltodextrin over the same concentration range. The measured reduction in the water activity can be attributed to the greater foaming-agent concentration, which contributes to the greater foaming stability and the greater solid content of the product, enhancing the efficiency of removing water in the drying process and minimizing the free-water content in the juice powder.53 Similar results were reported by Ahmad et al. (2024), where the water activity of foam-mat-dried red-flesh pitaya puree decreased significantly on increasing the concentration of egg albumin and whey protein isolate (5 to 30%). These results demonstrate that increasing the foaming-agent concentration is a viable approach for water activity reduction and extending the shelf-life of fruit powders.54
3.2.10 pH. The pH of the dried foam mat mulberry powder increased significantly with an increase in foaming agent concentration from 4.5 ± 0.5 for 2% to 5.26 ± 0.17 at 6% and to 5.8 ± 0.11 at 10%. It was observed that the acidity decreased as the concentration of the foaming agent increased. These results are in agreement with other studies, which reported that pH increases with increased levels of foaming agent in dried fruit powders.52,55
3.2.11 Titratable acidity. The titratable acidity (TA) of foam-mat-dried mulberry powder varied significantly with an increase in the concentration of the foaming agent in all the formulations. The TA values in whey protein treatments (T1–T5) decreased from 1.90 ± 0.01 to 0.80 ± 0.05 g/100 g, and the TA values in soy protein treatments (T6–T10) decreased from 1.92 ± 0.05 to 0.64 ± 0.05 g/100 g. Correspondingly, the T11 to T15 constituted whey protein isolate–maltodextrin blend led to a reduction in TA of 1.76 ± 0.02 to 0.64 ± 0.005 g/100 g with the increase in foaming agent concentration. Such reductions in TA can be attributed to the higher proportion of non-acidic solids as the foaming agents dilute the relative concentration of organic acids, and the interactions between the foaming agents and fruit acids may cause acid neutralization during drying. Similar results were reported in several other foam-mat drying studies. According to Hossain et al. (2021), the concentration of foaming and stabilizing agents added in foam-dried tomato powder led to considerable changes in the acidity through the neutralizing action of the foaming agents as well as enhanced moisture elimination during the drying process.55
3.2.12 Ascorbic acid. The ascorbic acid content of the mulberry juice powder obtained by foam mat drying varied between 10.0 ± 0.7 mg/100 g to 28.16 ± 0.28 mg/100 g. Within the group of whey-based formulations, the values were significantly increased with increasing concentration of foaming agent from a low concentration (12.2 ± 0.46 mg/100 g) to a high concentration (28.16 ± 0.28 mg/100 g), suggesting improved retention of ascorbic acid at increased foaming agent concentrations. The content of ascorbic acid for the soy protein isolate also increased from 10.0 ± 0.7 to 22.56 ± 0.76 mg/100 g and in the WPI:M combination from 10.83 ± 0.7 to 24.3 ± 0.8 mg/100 g. The results clearly show that better preservation of ascorbic acid was achieved using higher concentrations of foaming agents. The foaming matrix forms a protective structure that minimizes exposure of the ceramics to direct heat and oxidation during drying. This aligns with the findings of Khatri et al. (2024), where ascorbic acid values of 5.96 ± 0.004 to 26.65 ± 0.006 mg/100 g were recorded for different treatments.56 These studies demonstrate that foam mat drying, together with the optimal type and level of foaming agent, can retain heat-sensitive bioactive compounds.
3.2.13 Total anthocyanin content. An increasing trend in anthocyanin content in the foam mat dried mulberry juice powders was found with increasing foaming agent concentration. The different treatments yielded anthocyanin values between 96.1 ± 0.28 and 202.16 ± 0.28 mg/100 g, with the highest retention occurring at the highest foaming agent concentrations. In other words, more foaming agent may be needed to preserve anthocyanin after drying, presumably because it shields the anthocyanin from direct contact with heat and oxygen. The total anthocyanin content (TAC) was found to range from 87.09 to 129.53 mg/100 g for the treatments, showing that an increase in foaming agent concentration increases anthocyanin content.56,57 This result is in agreement with the results of Ozcelik (2019), in which maltodextrin-based foams were used to dry raspberry puree, presenting a structured network with larger spacing between pores.58
3.3 Functional properties of foam mat dried mulberry juice powder
3.3.1 Hygroscopicity. Increasing concentrations of foaming agents significantly decrease the hygroscopicity of the foam mat dried mulberry powder. The hygroscopicity values decreased specifically from 28.36 ± 0.47% at 2% foaming agent to 21.70 ± 0.60% at 10%, for WPI; from 29.4 ± 0.4% to 22.30 ± 0.43% with SPI; and from 28.83 ± 0.47% to 21.53 ± 0.7% with WPI:M. Higher concentrations of foaming agents may have a negative impact on the moisture absorptivity of the dried powder. These results align with the work of Hamad et al. (2024), who reported a reduction in hygroscopicity at higher concentrations of foaming agents in foam mat dried red dragon fruit powder.59 The improved foam stability and lower moisture content at higher foaming agent concentrations were attributed to the lower moisture absorption of the powder at higher concentrations.
3.3.2 Solubility. In all treatments, the solubility of the foam mat dried mulberry powder increased significantly with increasing foaming-agent concentration. The solubility of the powders obtained with whey protein isolate (WPI) increased from 50.5 ± 0.5% to 70.7 ± 0.75%, with soy protein isolate (SPI) from 48.7 ± 0.66% to 64.7 ± 0.62%, and with the WPI–maltodextrin blend from 51.8 ± 0.2% to 64.9 ± 0.83%. Such improvement in solubility is often associated with changes in structure and in better dispersion properties induced by higher levels of foaming agent. These results correlate with the findings of Rosda and Rosida (2024). The solubility of Dayak onion powder derived from the foam mat drying process increased with an increase in the concentration of egg white and maltodextrin from 42.33 to 48.17%.60 This observation is consistent with the findings of Khatri et al. (2024), who reported that increasing the concentration of foaming agents enhanced the solubility of the foam-mat dried powder.20 Table 2 presents various functional characteristics of foam mat dried mulberry juice powder.
Table 2 Functional characteristics of foam mat dried mulberry juice powdera
| Treatment |
Solubility (g/100 mL) |
Hygroscopicity (%) |
Bulk density (g cm−3) |
Tapped density (g cm−3) |
Carr's index (%) |
Hausner ratio |
Oil absorption capacity (g per g DW) |
Water absorption capacity (g per g DW) |
WPI: whey protein isolate; SPI: soya protein isolate; WPI:M: whey protein isolate:maltodextrin (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), [T1 = 2 g/100 mL WPI, T2 = 4 g/100 mL WPI, T3 = 6 g/100 mL WPI, T4 = 8 g/100 mL WPI, T5 = 10 g/100 mL WPI, T6 = 2 g/100 mL SPI, T7 = 4 g/100 mL SPI, T8 = 6 g/100 mL SPI, T9 = 8 g/100 mL SPI, T10 = 10 g/100 mL SPI, WPI:M, T11 = 2 g/100 mL WPI:M, T12 = 4 g/100 mL WPI:M, T13 = 6 g/100 mL WPI:M, T14 = 8 g/100 mL WPI:M, and T15 = 10 g/100 mL WPI:M]. 1), [T1 = 2 g/100 mL WPI, T2 = 4 g/100 mL WPI, T3 = 6 g/100 mL WPI, T4 = 8 g/100 mL WPI, T5 = 10 g/100 mL WPI, T6 = 2 g/100 mL SPI, T7 = 4 g/100 mL SPI, T8 = 6 g/100 mL SPI, T9 = 8 g/100 mL SPI, T10 = 10 g/100 mL SPI, WPI:M, T11 = 2 g/100 mL WPI:M, T12 = 4 g/100 mL WPI:M, T13 = 6 g/100 mL WPI:M, T14 = 8 g/100 mL WPI:M, and T15 = 10 g/100 mL WPI:M]. |
| T1 |
50.53 ± 0.5c |
28.36 ± 0.47a |
0.71 ± 0.02a |
0.86 ± 0.002ab |
17.40 ± 0.5e |
1.21 ± 0.005d |
0.04 ± 0.005g |
0.38 ± 0.01fg |
| T2 |
54 ± 0.3c |
27.16 ± 0.20ab |
0.67 ± 0.02ab |
0.83 ± 0.04bc |
19.20 ± 0.2d |
1.23 ± 0.005cd |
0.06 ± 0.007fg |
0.44 ± 0.01defg |
| T3 |
62.06 ± 0.3b |
25.46 ± 0.41bc |
0.66 ± 0.04ab |
0.85 ± 0.011bc |
22.30 ± 0.1cd |
1.27 ± 0.01bc |
0.10 ± 0.005cd |
0.54 ± 0.005cd |
| T4 |
68.13 ± 0.32a |
24.36 ± 0.47cd |
0.52 ± 0.09c |
0.72 ± 0.009c |
27.70 ± 0.1ab |
1.36 ± 0.01abc |
0.12 ± 0.005bc |
0.60 ± 0.004bc |
| T5 |
70.76 ± 0.75a |
21.70 ± 0.60e |
0.56 ± 0.017bc |
0.75 ± 0.009c |
25.36 ± 0.1bc |
1.33 ± 0.01abc |
0.13 ± 0.01bc |
0.62 ± 0.003ab |
| T6 |
48.73 ± 0.66c |
29.43 ± 0.40a |
0.70 ± 0.009a |
0.86 ± 0.004ab |
20 ± 0.2d |
1.22 ± 0.02cd |
0.06 ± 0.005fg |
0.32 ± 0.005gh |
| T7 |
52.16 ± 0.20c |
27 ± 0.40ab |
0.71 ± 0.011a |
0.90 ± 0.03a |
21.33 ± 0.3cd |
1.28 ± 0.01bc |
0.07 ± 0.005ef |
0.40 ± 0.01efg |
| T8 |
60.60 ± 0.87bc |
25.53 ± 0.50bc |
0.66 ± 0.011ab |
0.86 ± 0.002ab |
23.2 ± 0.2cd |
1.29 ± 0.05bc |
0.08 ± 0.0005de |
0.48 ± 0.003de |
| T9 |
62.36 ± 0.32 |
24.36 ± 0.40cd |
0.52 ± 0.001c |
0.73 ± 0.011c |
25 ± 0.3bc |
1.39 ± 0.02abc |
0.11 ± 0.01cd |
0.52 ± 0.004cd |
| T10 |
64.70 ± 0.62b |
22.30 ± 0.43de |
0.71 ± 0.009c |
0.75 ± 0.08c |
30.63 ± 0.1a |
1.42 ± 0.005a |
0.17 ± 0.01ab |
0.64 ± 0.01a |
| T11 |
51.80 ± 0.2c |
28.83 ± 0.47a |
0.67 ± 0.020ab |
0.83 ± 0.06bc |
19.20 ± 0.02d |
1.22 ± 0.01cd |
0.063 ± 0.005fg |
0.24 ± 0.002h |
| T12 |
55.70 ± 0.43c |
28.03 ± 0.15a |
0.58 ± 0.011b |
0.82 ± 0.09bc |
28.70 ± 0.04cd |
1.40 ± 0.01ab |
0.086 ± 0.005de |
0.38 ± 0.002fg |
| T13 |
58.60 ± 0.55bc |
25 ± 0.35bc |
0.62 ± 0.009b |
0.87 ± 0.002a |
28 ± 0.31ab |
1.41 ± 0.03ab |
0.11 ± 0.01bcd |
0.46 ± 0.005def |
| T14 |
62.36 ± 0.40b |
23 ± 0.40de |
0.62 ± 0.002b |
0.82 ± 0.001a |
22.23 ± 0.35ab |
1.42 ± 0.01a |
0.16 ± 0.005ab |
0.62 ± 0.004ab |
| T15 |
64.93 ± 0.83b |
21.53 ± 0.7de |
0.55 ± 0.001c |
0.79 ± 0.001c |
30 ± 0.37a |
1.42 ± 0.005a |
0.18 ± 0.05a |
0.68 ± 0.002a |
3.3.3 Total phenolic content. Foam mat dried mulberry powder showed a quite noticeable decrease in TPC at higher levels of foaming agent in all treatments. For the mulberry samples treated with WPI, the phenol content decreased gradually from 180.63 ± 0.56 mg GAE/100 g at the lowest foaming concentration to 94.7 ± 0.52 mg GAE/100 g at the highest foaming concentration. Similarly, SPI lowered the phenolic content from 158.4 ± 0.45 to 69.83 ± 0.45 mg GAE/100 g, and WPI:M reduced the phenolic content from 161.78 ± 0.36 to 73.93 ± 0.40 mg GAE/100 g. Decreasing phenolic compound content with increasing foaming agent concentration is observed, and these data suggest that this trend is a dilution effect, since the foaming agents themselves do not contain phenols. Furthermore, the phenolic content is reduced by thermal degradation and enzymatic activity during the drying process itself. These results are in agreement with previous studies, which reported a significant reduction in phenolic content with increasing levels of foaming agents in foam mat-dried fruit powders.61,62
3.3.4 Total flavonoid content. The total flavonoid content of foam-mat-dried mulberry juice powder ranged from 254.76 ± 0.60 to 363.6 ± 0.53 mg QE/100 g (21.0 ± 0.4 to 57.97 ± 0.65 mg RE/100 g), and the highest levels were achieved with compositions based on the whey protein isolate (331.33 ± 0.36 to 363.6 ± 0.53 mg QE/100 g; 34.67 ± 0.49 to 57.97 ± 0.65 mg RE/100 g). A decrease in flavonoid content with an increase in foaming agent concentration was observed, which may be due to dilution of bioactive compounds because the flavonoids are native to the mulberry juice and the foaming agents do not contribute to this content. Similarly, the presence of these bioactive components have been reported previously by Khatri et al. (2024) using different foaming agents.20 In addition, it was reported that air bubbles in the foam matrix can preserve flavonoids during drying via partial heat shielding.57
3.3.5 Antioxidant activity. A decreasing trend was observed for the antioxidant activity (%) of foam mat dried mulberry juice powders with increasing foaming agent concentrations. WPI values were in the range of 65.53 ± 0.83% to 32.2 ± 0.41%; SPI value was between 55.6 ± 0.80% and 32.7 ± 0.40% and WPI:M values were 64.66 ± 0.57% to 43.23 ± 0.25%. These are consistent with the trend reported by Wang et al. (2020) and Khatri et al. (2024), who showed that elevated foaming agent levels correlated with decreased antioxidant activity.29,56 An increase in foaming agent concentration expands the air–liquid interfacial area. As a result, the antioxidant concentration at the interface becomes diluted, reducing the activity.63
3.3.6 Oil absorption capacity. Oil absorption capacity (OAC) provides insight into the powder's interaction with lipid systems, which influences flavor retention, fat binding, and integration into the composite food products. Although the mulberry juice dried powder is low in fat, it can be utilized as a functional ingredient for lipid-containing food products, such as confectionery, bakery, ice cream, and emulsified systems.64,65 OAC is influenced by the presence of hydrophobic groups, protein-based foaming agents, and the microstructural changes induced by foam-mat drying. Powders with higher OAC often exhibit improved flavor retention, enhanced mouthfeel, and better integration into fat-containing matrices. Foam-mat drying with foaming agents such as whey protein isolate (WPI), soy protein isolate (SPI), and WPI–maltodextrin blends significantly enhanced oil absorption capacity, increasing from 0.04 ± 0.005 to 0.13 ± 0.01 g per g DW for (T1–T5), 0.06 ± 0.005 to 0.17 ± 0.01 g per g DW for (T6–T10), and 0.063 ± 0.005 to 0.18 ± 0.05 g per g DW for (T11–T15) as the concentration increased. This improvement is attributed to protein denaturation during foam-mat drying, which exposes hydrophobic groups that enhance oil binding.66 Similarly, Khatri et al. (2024) reported that increasing the foaming-agent concentration in foam-mat dried powders improves OAC due to a greater exposure of hydrophobic sites and enhanced oil entrapment capacity.20
3.3.7 Water absorption capacity. WAC is a key indicator of the powder's ability to bind and retain water, and it directly influences the rehydration properties, dispersibility, and solubility of the powder in aqueous food systems such as instant drinks and mixes. WAC helps determine the effect of drying conditions and foaming agents on the powder's reconstitution behavior.67,68 Higher WAC values are associated with powders that rapidly dissolve, rehydrate efficiently, and integrate well into aqueous food systems. The water absorption capacity (WAC) of the samples ranged from 0.24 ± 0.002 g per g DW to 0.68 ± 0.002 g per g DW, showing a progressive increase in WAC with increasing concentration. The lowest WAC was observed at 0.24 g per g DW, while the highest was recorded at 0.68 g per g DW. This significant enhancement in WAC can be attributed to the greater availability of hydrophilic components at higher concentrations. The incorporation of carbohydrates, such as locust bean gum in protein-based systems, led to improved water-holding capacity and functional performance, demonstrating the synergistic effect of hydrocolloids on matrix hydration properties.69 This conclusion aligns with the study by Khatri et al. (2024), in which the increase in concentration of foaming agent increased the water absorption capacity.20
3.4 FTIR characterization of foam mat dried mulberry juice powder
The FTIR spectrum of WPI-based foam mat dried mulberry fruit juice powder (2 g/100 mL; T2) exhibited several characteristic absorption peaks attributed to different functional groups. The spectrum includes medium-intensity peaks attributed to O–H stretching vibrations of alcohol and phenol groups, at 3279.92 cm−1. C–H stretching and bending vibrations that are characteristic of methyl and methylene groups correspond to peaks at 2923.68 cm−1 and 1403.26 cm−1, respectively. A strong absorption band at 1626.90 cm−1 was assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching, characteristic of carbonyl compounds and phenols. Additionally, bands at 1241.24 cm−1 and 1002.84 cm−1 (C–H or C–O bending/stretching) confirmed the presence of proteinaceous compounds. In addition, a strong peak at 1051.47 cm−1 and weak bending vibrations at 865.98 cm−1 support the presence of methyl and methylene compounds. Weak peaks at 817.06 cm−1 and 777.32 cm−1 indicate aromatic structures such as benzene and monosubstituted benzene rings. Also, a weak peak at 585.65 cm−1 corresponds to P–O bending vibrations which indicates the presence of anthocyanin compounds (Fig. 2). Further, sulfonic acid or sulfonate groups were indicated by a weak peak at 516.14 cm−1 from the S–O bending vibrations. These observations are consistent with the findings of previous studies on several foam mat dried powders.20,36,70
O stretching, characteristic of carbonyl compounds and phenols. Additionally, bands at 1241.24 cm−1 and 1002.84 cm−1 (C–H or C–O bending/stretching) confirmed the presence of proteinaceous compounds. In addition, a strong peak at 1051.47 cm−1 and weak bending vibrations at 865.98 cm−1 support the presence of methyl and methylene compounds. Weak peaks at 817.06 cm−1 and 777.32 cm−1 indicate aromatic structures such as benzene and monosubstituted benzene rings. Also, a weak peak at 585.65 cm−1 corresponds to P–O bending vibrations which indicates the presence of anthocyanin compounds (Fig. 2). Further, sulfonic acid or sulfonate groups were indicated by a weak peak at 516.14 cm−1 from the S–O bending vibrations. These observations are consistent with the findings of previous studies on several foam mat dried powders.20,36,70
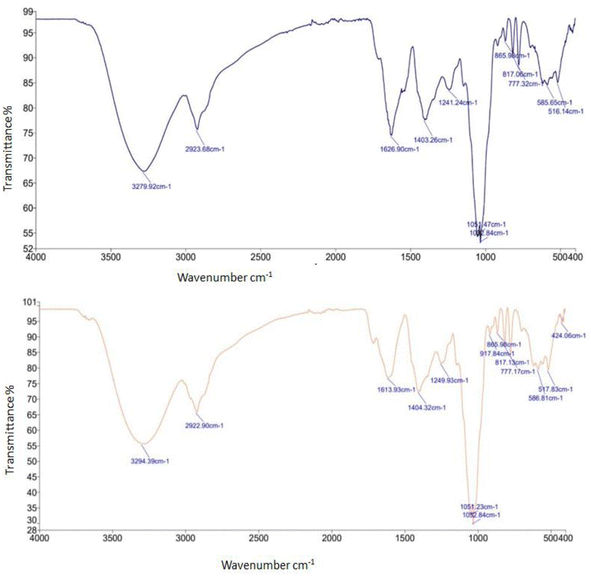 |
| | Fig. 2 FTIR spectrum (top and bottom) of the mulberry fruit juice powder treated with 10 g/100 mL of WPI (T2) and 10 g/100 mL of SPI (T6), respectively. | |
The FTIR spectrum of SPI-based foam mat dried mulberry fruit juice powder (T6) shows several clear absorption peaks (Fig. 2) that are attributed to functional groups present in the powder. O–H stretching (the presence of alcohols and phenolic compounds) was confirmed by a medium intensity peak at 3294.39 cm−1. At 2922.90 cm−1 , the C–H stretching of methyl and methylene groups was observed, and the bending vibration of these groups was observed at 1404.32 cm−1. A peak at 1613.93 cm−1 is an indicative of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of carbonyl compounds and phenols. Medium peaks at 1249.93 cm−1 are attributed to C–H bending of proteins. Peaks at 1051.23 cm−1 and 1025.84 cm−1 are observed with lower transmittance, which are attributed to C–H or C–O bending/stretching in the phenolic compounds. The peaks at 865.96, 817.13 and 777.17 cm−1 are attributed to the presence of carbohydrates and benzene derivatives. The peaks at 586.81 cm−1 and 517.83 cm−1 are linked to P
O stretching of carbonyl compounds and phenols. Medium peaks at 1249.93 cm−1 are attributed to C–H bending of proteins. Peaks at 1051.23 cm−1 and 1025.84 cm−1 are observed with lower transmittance, which are attributed to C–H or C–O bending/stretching in the phenolic compounds. The peaks at 865.96, 817.13 and 777.17 cm−1 are attributed to the presence of carbohydrates and benzene derivatives. The peaks at 586.81 cm−1 and 517.83 cm−1 are linked to P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and S
O and S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds related to anthocyanins and sulfonic acid/sulfate groups, respectively. Also, a peak at 424.06 cm−1 is indicative of C
O bonds related to anthocyanins and sulfonic acid/sulfate groups, respectively. Also, a peak at 424.06 cm−1 is indicative of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bending in aromatic compounds. This observation aligns with previous studies on foam mat dried powder.20,71 The presence of phenolics, sugars, proteins and anthocyanin-related groups was evidenced by the appearance of characteristic absorption bands in both T2 and T6 powders corresponding to O–H, C–H, C
C bending in aromatic compounds. This observation aligns with previous studies on foam mat dried powder.20,71 The presence of phenolics, sugars, proteins and anthocyanin-related groups was evidenced by the appearance of characteristic absorption bands in both T2 and T6 powders corresponding to O–H, C–H, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, C–N/C–O, and aromatic compounds. However, notable differences were observed in the intensity of these peaks, which indicated the presence of compositional and structural differences between the two treatments. T6 exhibited an O–H stretch at 3294 cm−1 relative to 3279 cm−1 in T2, indicating stronger hydrogen bonding or higher retention of phenolic compounds. The enhanced OH bonding and the phenolic-related bands in T6 indicated stable SPI–polyphenol complexation, which can stabilize the molecular interaction and the reduced foaming activity of the SPI-based system. In contrast, T2 displayed less restrictive, looser interactions due to the unfolding of whey proteins with acid and the stabilization of the protein by fruit sugars and pectic materials as a synergistic effect. These differences are also consistent with the foaming attributes and functionality of WPI-formulations.
O, C–N/C–O, and aromatic compounds. However, notable differences were observed in the intensity of these peaks, which indicated the presence of compositional and structural differences between the two treatments. T6 exhibited an O–H stretch at 3294 cm−1 relative to 3279 cm−1 in T2, indicating stronger hydrogen bonding or higher retention of phenolic compounds. The enhanced OH bonding and the phenolic-related bands in T6 indicated stable SPI–polyphenol complexation, which can stabilize the molecular interaction and the reduced foaming activity of the SPI-based system. In contrast, T2 displayed less restrictive, looser interactions due to the unfolding of whey proteins with acid and the stabilization of the protein by fruit sugars and pectic materials as a synergistic effect. These differences are also consistent with the foaming attributes and functionality of WPI-formulations.
3.5 Field-emission scanning electron microscopy
The surface morphology and microstructural characteristics of foam mat dried mulberry fruit juice powder produced with 2% whey protein isolate (Fig. 3) and soy protein isolate (Fig. 4) were studied using field-emission scanning electron microscopy (FE-SEM). At lower magnification (50× and 100×), both samples showed loosely agglomerated, irregular particles that could have come from structural collapse and mechanical disruption during drying or scraping. Analysis of the powder surface with SEM showed that the soy protein-treated powder displayed a denser and more continuous surface with less debris, while the whey protein-treated powder was more fragmented with visible voids in the surface. At magnifications in the intermediate range (500× to 1000×), both samples showed the formation of flake-like structures and films that coated the sample, which indicates the encapsulation of mulberry juice solids into the protein matrix. The variations of porosity on the surfaces of the whey sample were more compact, with smoother regions, which also indicated a stronger matrix formation. By comparison, the soy-protein-treated powder had angular and aggregated particles with a rougher texture. Distinct morphological features were observed in higher magnification images (5000× to 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00×). The sample treated with whey protein had lamellar and fibrous-like networks with compact surfaces that supported efficient encapsulation. The structural heterogeneity was characterized by more porous and granular textures in a soy protein-based powder. The elemental composition of both powders was further confirmed by energy dispersive spectroscopy (EDS) analysis (Fig. 5). For the whey protein isolate (WPI) treated foam mat dried (FMD) sample (T2), the elements found to be present at greatest weight percent were carbon (51.46 wt%, 59.64 at%) and oxygen (36.49 wt%, 31.75 at%), with nitrogen (6.82 wt%, 6.78 at%) and trace amounts of sodium, magnesium, potassium, calcium and copper. Similarly, in the SPI-FMD mulberry powder sample (T6), carbon (51.88 wt%, 59.30 at%) and oxygen (35.72 wt%, 30.65 at%) were the dominant elements, followed by nitrogen (9.25 wt%, 9.06 at%). Trace amounts of magnesium, potassium, and copper were also detected at lower concentrations.
00×). The sample treated with whey protein had lamellar and fibrous-like networks with compact surfaces that supported efficient encapsulation. The structural heterogeneity was characterized by more porous and granular textures in a soy protein-based powder. The elemental composition of both powders was further confirmed by energy dispersive spectroscopy (EDS) analysis (Fig. 5). For the whey protein isolate (WPI) treated foam mat dried (FMD) sample (T2), the elements found to be present at greatest weight percent were carbon (51.46 wt%, 59.64 at%) and oxygen (36.49 wt%, 31.75 at%), with nitrogen (6.82 wt%, 6.78 at%) and trace amounts of sodium, magnesium, potassium, calcium and copper. Similarly, in the SPI-FMD mulberry powder sample (T6), carbon (51.88 wt%, 59.30 at%) and oxygen (35.72 wt%, 30.65 at%) were the dominant elements, followed by nitrogen (9.25 wt%, 9.06 at%). Trace amounts of magnesium, potassium, and copper were also detected at lower concentrations.
 |
| | Fig. 3 FE-SEM micrograph of the foam mat dried mulberry powder WPI-2% (T2) [(a) 50×; (b) 100×; (c) 1000×; (d) 5000×; (e) 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×]. 000×]. | |
 |
| | Fig. 4 FE-SEM micrograph of the foam mat dried mulberry powder SPI-2% (T6) [(a) 50×; (b) 100×; (c) 1000×; (d) 5000×; (e) 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×]. 000×]. | |
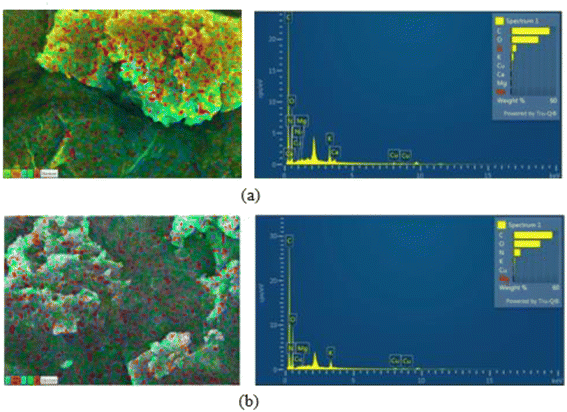 |
| | Fig. 5 EDS mapping and the spectrum of (a) FMD-T2 and (b) FMD-T6. | |
4 Conclusions
Foam mat drying was demonstrated to be an effective approach for the production of stable mulberry juice powder, while preserving bioactive constituents and their functional properties. WPI-treated mulberry powder demonstrated 68–71% solubility at 8–10% concentration, retaining 65.3% antioxidant activity at concentrations <4% and 180.6 mg GAE/100 g of phenolic compounds. Comparative analysis displayed that SPI provided satisfactory functionality as a plant-based alternative, but with lower solubility. In particular, the whey–maltodextrin composite (WPI:M) system achieved balanced physicochemical functionality (64.9% solubility) and phytochemical preservation (192.1 mg/100 g anthocyanins), which suggests that protein and carbohydrate components interact synergistically during the drying process. These findings provide a scientific rationale for carrier selection in industrial applications, with WPI recommended for maximizing functional properties, SPI as a viable plant-based alternative, and WPI:M for nutraceutical applications (bioactive retention). Although this study clearly established the role of carrier type and concentration in determining the physicochemical attributes and functional performance of foam-mat dried mulberry juice powder, additional research is possible to enhance the knowledge of bioactive stability. Future studies can focus on the profiling of individual phenolic constituents and anthocyanins. This specific characterization will enable precise identification and quantification of key bioactive compounds, which will further help in understanding their stability and possible degradation mechanism during foam-mat drying.
Author contributions
Farzeen – writing-original draft, data curation, formal analysis, investigation, Hamid – conceptualization, supervision, writing-review and editing, Navjot Kaur – writing-review and editing, Poonam Choudhary and Sandeep Mann – supervision, writing-review and editing.
Conflicts of interest
The authors have declared no conflicts of interest for this article.
Data availability
The data that support the findings are included in this manuscript.
Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fb00549c.
Acknowledgements
The first author would like to acknowledge the facilities and support provided by the Lovely Professional University, Phagwara-144411, Punjab (India), for conducting this research.
References
- S. Ercisli and E. Orhan, Food Chem., 2007, 103, 1380–1384 CrossRef CAS.
- Q.-Z. Du, J. Zheng and Y. Xu, J. Food Compos. Anal., 2008, 21, 390–395 Search PubMed.
- D. Bhattacharjya, A. Sadat, P. Dam, D. F. Buccini, R. Mondal, T. Biswas, K. Biswas, H. Sarkar, A. Bhuimali and A. Kati, Curr. Opin. Food Sci., 2021, 40, 121–135 Search PubMed.
- T. Chen, F.-F. Shuang, Q.-Y. Fu, Y.-X. Ju, C.-M. Zong, W.-G. Zhao, D.-Y. Zhang, X.-H. Yao and F.-L. Cao, Molecules, 2022, 27, 2688 CrossRef CAS PubMed.
- B. K. Singhal, M. A. Khan, A. Dhar, F. M. Baqual and B. B. Bindroo, J. Fruit Ornamental Plant Res., 2010, 18, 83–99 Search PubMed.
- I. Kim and J. Lee, Antioxidants, 2020, 9, 242 CrossRef CAS PubMed.
- L. Ma, G. Xiao, J. Wu, J. Wen, Z. Bu and D. Tang, in Asian Berries, CRC Press, 2020, pp. 21–39 Search PubMed.
- D. Bhattacharjya, A. Sadat, P. Dam, D. F. Buccini, R. Mondal, T. Biswas, K. Biswas, H. Sarkar, A. Bhuimali and A. Kati, Curr. Opin. Food Sci., 2021, 40, 121–135 Search PubMed.
- J. Barbosa and P. Teixeira, Food Rev. Int., 2017, 33, 335–358 Search PubMed.
- F. Elmas, E. Varhan and M. Koç, J. Food Meas. Char., 2019, 13, 70–86 CrossRef.
- H. Aryaee, P. Ariaii, D. Zare, S. Mirdamadi and S. N. Raeisi, J. Food Process. Preserv., 2023, 1, 5597647 Search PubMed.
- A. Michalska, A. Wojdyło, K. Lech, G. P. Łysiak and A. Figiel, Food Chem., 2016, 207, 223–232 CrossRef CAS PubMed.
- T. S. Franco, C. A. Perussello, L. N. Ellendersen and M. L. Masson, LWT--Food Sci. Technol., 2016, 66, 503–513 Search PubMed.
- Z. Hardy and V. A. Jideani, Crit. Rev. Food Sci. Nutr., 2017, 57, 2560–2572 Search PubMed.
- R. Gao, L. Xue, Y. Zhang, Y. Liu, L. Shen and X. Zheng, LWT, 2022, 154, 112811 CrossRef CAS.
- T. Ozdal, E. Capanoglu and F. Altay, Food Res. Int., 2013, 51, 954–970 CrossRef CAS.
- C. Leyva-Porras, M. Z. Saavedra-Leos, L. A. López-Martinez, V. Espinosa-Solis, Y. Terán-Figueroa, A. Toxqui-Terán and I. Compeán-Martínez, Molecules, 2021, 26, 5466 Search PubMed.
- J. Huang, X. Feng, Q. Wang, D. Liu, S. Zhang and L. Chu, Int. J. Biol. Macromol., 2024, 254, 128039 Search PubMed.
- I. Shabir, A. H. Dar, K. K. Dash, S. Srivastava, V. K. Pandey, S. Manzoor, S. Manzoor and I. Bashir, J. Agric. Food Res., 2023, 14, 100853 CAS.
- B. Khatri and A. K. Jaiswal, LWT, 2024, 205, 116512 CrossRef CAS.
- P. Kandasamy, N. Varadharaju and S. Kalemullah, Food Sci. Qual. Manag., 2012, 9, 17–27 Search PubMed.
- K. G. Marinova, E. S. Basheva, B. Nenova, M. Temelska, A. Y. Mirarefi, B. Campbell and I. B. Ivanov, Food Hydrocolloids, 2009, 23, 1864–1876 CrossRef CAS.
- K. O. Falade, K. I. Adeyanju and P. I. Uzo-Peters, Eur. Food Res. Technol., 2003, 217, 486–491 Search PubMed.
- S. Ranganna, Handbook of Analysis and Quality Control for Fruit and Vegetable Products, Tata McGraw-Hill Education, 1986 Search PubMed.
- W. A. Shehata, M. S. Akhtar and T. Alam, Trends Appl. Sci. Res., 2020, 15, 179–186 Search PubMed.
- Y.-Z. Cai and H. Corke, J. Food Sci., 2000, 65, 1248–1252 CrossRef CAS.
- S. Hogekamp and H. Schubert, Food Sci. Technol. Int., 2003, 9, 223–235 CrossRef.
- V. L. Singleton and J. A. Rossi, Am. J. Enol. Vitic., 1965, 16, 144–158 CrossRef CAS.
- R. Wang, Y. Zhao, L. Zhu, Z. Fang and Q. Shi, J. Food Meas. Char., 2020, 14, 1201–1212 CrossRef.
- A. C. C. Egbuonu, Res. J. Environ. Toxicol., 2015, 9, 160 CrossRef.
- A. R. Parmar, P. R. Davara, N. U. Joshi, P. J. Rathod and D. K. Antala, Int. J. Curr. Microbiol. Appl. Sci., 2020, 9, 669–681 CrossRef CAS.
- N. Li and A. L. Girard, Food Hydrocolloids, 2023, 134, 108100 CrossRef CAS.
- E. Farid, S. Mounir, H. Siliha, S. El-Nemr and E. Talaat, J. Food Meas. Char., 2023, 17, 3815–3826 CrossRef.
- L. Ao, P. Liu, A. Wu, J. Zhao and X. Hu, Foods, 2021, 10, 2813 CrossRef CAS PubMed.
- H. Wu, B. Li, M. Corradini, L. Zhong, J. Zhou and X. Liu, Food Prod., Process. Nutr., 2024, 6, 93 CrossRef CAS.
- N. Chandrakar, S. Padhi, S. S. Saraugi, R. Sehrawat, A. Singh and W. Routray, Chem. Eng. Res. Des., 2024, 209, 367–379 CrossRef CAS.
- P. Kubbutat, U. Kulozik and J. Dombrowski, Foods, 2021, 10, 1163 CrossRef CAS PubMed.
- S. Damodaran, J. Food Sci., 2005, 70, R54–R66 CrossRef CAS.
- H. Wu, B. Li, M. Corradini, L. Zhong, J. Zhou and X. Liu, Food Prod., Process. Nutr., 2024, 6, 93 CrossRef CAS.
- E. A. Foegeding, J. P. Davis, D. Doucet and M. K. McGuffey, Trends Food Sci. Technol., 2002, 13, 151–159 CrossRef CAS.
- L. Jakobek, Food Chem., 2015, 175, 556–567 CrossRef CAS PubMed.
- X. Zhao, C. Li and F. Xue, Future Foods, 2023, 7, 100211 CrossRef CAS.
- M. Hajiaghaei and A. Sharifi, J. Food Qual., 2022, 2022, 7499994 CrossRef.
- M. Ozcelik and U. Kulozik, Foods, 2023, 12, 1673 Search PubMed.
- S. T. Sullivan, S. A. Khan and A. S. Eissa, Whey Processing, Functionality and Health Benefits, 2008, pp. 99–132 Search PubMed.
- P. Kubbutat, L. Leitão and U. Kulozik, Foods, 2021, 10, 1876 CrossRef CAS PubMed.
- M. R. Islam Shishir, F. S. Taip, N. A. Aziz, R. A. Talib and M. S. Hossain Sarker, Food Sci. Biotechnol., 2016, 25, 461–468 CrossRef CAS PubMed.
- T. N. A. A. T. Ahmad, N. Yusof, N. Z. R. Anwar and T. F. I. C. K. Jusoh, J. Trop. Resour. Sustain. Sci., 2024, 12, 19–27 Search PubMed.
- E. Jakubczyka, E. Gondeka, K. Tamborb, E. Jakubczyk, E. Gondek and K. Tambor, in ICEF 11 International Congress on Engineering and Food, International Association of Engineering and Food Athens, 2011 Search PubMed.
- M. Belal, M. A. Hossain, S. Mitra and W. Zzaman, J. Microbiol., Biotechnol. Food Sci., 2023, 12, e4741 CAS.
- S. Chaipoot, R. Phongphisutthinant, P. Wiriyacharee, G. Kanthakat, W. Wongwatcharayothin, C. Somjai, K. Danmek and B. Chuttong, Foods, 2024, 13, 2265 CrossRef CAS PubMed.
- C. Thakur, A. Kumar Verma and P. Chaudhary, Pharma Innovation, 2021, 10, 78–82 CrossRef.
- M. R. I. Shishir and W. Chen, Trends Food Sci. Technol., 2017, 65, 49–67 CrossRef CAS.
- T. N. A. T. Ahmad, N. Yusof, N. Z. R. Anwar and T. F. I. C. K. Jusoh, J. Trop. Resour. Sustain. Sci., 2024, 12, 19–27 Search PubMed.
- M. A. Hossain, S. Mitra, M. Belal and W. Zzaman, Food Res., 2021, 5, 291–297 Search PubMed.
- B. Khatri, Hamid, R. Shams, K. K. Dash, A. M. Shaikh and K. Béla, Discover Food, 2024, 4, 166 CrossRef.
- A. Motevali, S. Minaei and M. H. Khoshtagaza, Energy Convers. Manage., 2011, 52, 1192–1199 CrossRef.
- M. Ozcelik, A. Heigl, U. Kulozik and S. Ambros, Innovative Food Sci. Emerging Technol., 2019, 56, 102183 CrossRef CAS.
- A. Hamad, A. H. Mulyadi, H. Yulianti, D. Hartanti and M. Naveed, J. Appl. Sci. Eng. Technol. Educ., 2024, 6, 143–154 CrossRef.
- S. Rosda and R. Rosida, Asian Journal of Applied Research for Community Development and Empowerment, 2024, 8, 247–257 Search PubMed.
- M. Ali, Y. Durrani and M. Ayub, Sarhad J. Agric., 2016, 32, 80–88 CrossRef.
- R. Wang, Y. Zhao, L. Zhu, Z. Fang and Q. Shi, J. Food Meas. Char., 2020, 14, 1201–1212 CrossRef.
- M. Costa, S. Losada-Barreiro, F. Paiva-Martins and C. Bravo-Diaz, Molecules, 2021, 26, 6058 CrossRef CAS PubMed.
- S. Sheikh, F. Siddique, K. Ameer, R. S. Ahmad, A. Hameed, A. Ebad, I. A. Mohamed Ahmed and S. Shibli, Food Sci. Nutr., 2023, 11, 204–215 Search PubMed.
- L. M. Resende and A. S. Franca, in Flour and Breads and their Fortification in Health and Disease Prevention (Second Edition), ed. V. R. Preedy and R. R. Watson, Academic Press, 2019, pp. 387–401 Search PubMed.
- S. Phongthai, Food and Applied Bioscience Journal, 2023, 11, 26–40 Search PubMed.
- M. Z. Islam, M. I. Jahan, K. Monalisa, R. Rana and M. M. Hoque, LWT, 2024, 200, 116158 Search PubMed.
- R. V Tonon, C. Brabet and M. D. Hubinger, J. Food Eng., 2008, 88, 411–418 CrossRef.
- B. Başyiğit, G. Altun, M. Yücetepe, A. Karaaslan and M. Karaaslan, Int. J. Biol. Macromol., 2023, 231, 123352 CrossRef PubMed.
- Z. Ma, J. Cheng, S. Jiao and P. Jing, Int. J. Food Sci. Technol., 2022, 57, 2267–2276 CrossRef CAS.
- A. S. Brar, P. Kaur, G. Kaur, J. Subramanian, D. Kumar and A. Singh, Int. J. Fruit Sci., 2020, 20, S1495–S1518 CrossRef.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *a,
Navjot Kaura,
Poonam Choudharyb and
Sandeep Mannb
*a,
Navjot Kaura,
Poonam Choudharyb and
Sandeep Mannb
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×.
000×.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), [T1 = 2 g/100 mL WPI, T2 = 4 g/100 mL WPI, T3 = 6 g/100 mL WPI, T4 = 8 g/100 mL WPI, T5 = 10 g/100 mL WPI, T6 = 2 g/100 mL SPI, T7 = 4 g/100 mL SPI, T8 = 6 g/100 mL SPI, T9 = 8 g/100 mL SPI, T10 = 10 g/100 mL SPI, WPI:M T11 = 2 g/100 mL WPI:M, T12 = 4 g/100 mL WPI:M, T13 = 6 g/100 mL WPI:M, T14 = 8 g/100 mL WPI:M, and T15 = 10 g/100 mL WPI:M].
1), [T1 = 2 g/100 mL WPI, T2 = 4 g/100 mL WPI, T3 = 6 g/100 mL WPI, T4 = 8 g/100 mL WPI, T5 = 10 g/100 mL WPI, T6 = 2 g/100 mL SPI, T7 = 4 g/100 mL SPI, T8 = 6 g/100 mL SPI, T9 = 8 g/100 mL SPI, T10 = 10 g/100 mL SPI, WPI:M T11 = 2 g/100 mL WPI:M, T12 = 4 g/100 mL WPI:M, T13 = 6 g/100 mL WPI:M, T14 = 8 g/100 mL WPI:M, and T15 = 10 g/100 mL WPI:M].![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), [T1 = 2 g/100 mL WPI, T2 = 4 g/100 mL WPI, T3 = 6 g/100 mL WPI, T4 = 8 g/100 mL WPI, T5 = 10 g/100 mL WPI, T6 = 2 g/100 mL SPI, T7 = 4 g/100 mL SPI, T8 = 6 g/100 mL SPI, T9 = 8 g/100 mL SPI, T10 = 10 g/100 mL SPI, WPI:M, T11 = 2 g/100 mL WPI:M, T12 = 4 g/100 mL WPI:M, T13 = 6 g/100 mL WPI:M, T14 = 8 g/100 mL WPI:M, and T15 = 10 g/100 mL WPI:M].
1), [T1 = 2 g/100 mL WPI, T2 = 4 g/100 mL WPI, T3 = 6 g/100 mL WPI, T4 = 8 g/100 mL WPI, T5 = 10 g/100 mL WPI, T6 = 2 g/100 mL SPI, T7 = 4 g/100 mL SPI, T8 = 6 g/100 mL SPI, T9 = 8 g/100 mL SPI, T10 = 10 g/100 mL SPI, WPI:M, T11 = 2 g/100 mL WPI:M, T12 = 4 g/100 mL WPI:M, T13 = 6 g/100 mL WPI:M, T14 = 8 g/100 mL WPI:M, and T15 = 10 g/100 mL WPI:M].![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching, characteristic of carbonyl compounds and phenols. Additionally, bands at 1241.24 cm−1 and 1002.84 cm−1 (C–H or C–O bending/stretching) confirmed the presence of proteinaceous compounds. In addition, a strong peak at 1051.47 cm−1 and weak bending vibrations at 865.98 cm−1 support the presence of methyl and methylene compounds. Weak peaks at 817.06 cm−1 and 777.32 cm−1 indicate aromatic structures such as benzene and monosubstituted benzene rings. Also, a weak peak at 585.65 cm−1 corresponds to P–O bending vibrations which indicates the presence of anthocyanin compounds (Fig. 2). Further, sulfonic acid or sulfonate groups were indicated by a weak peak at 516.14 cm−1 from the S–O bending vibrations. These observations are consistent with the findings of previous studies on several foam mat dried powders.20,36,70
O stretching, characteristic of carbonyl compounds and phenols. Additionally, bands at 1241.24 cm−1 and 1002.84 cm−1 (C–H or C–O bending/stretching) confirmed the presence of proteinaceous compounds. In addition, a strong peak at 1051.47 cm−1 and weak bending vibrations at 865.98 cm−1 support the presence of methyl and methylene compounds. Weak peaks at 817.06 cm−1 and 777.32 cm−1 indicate aromatic structures such as benzene and monosubstituted benzene rings. Also, a weak peak at 585.65 cm−1 corresponds to P–O bending vibrations which indicates the presence of anthocyanin compounds (Fig. 2). Further, sulfonic acid or sulfonate groups were indicated by a weak peak at 516.14 cm−1 from the S–O bending vibrations. These observations are consistent with the findings of previous studies on several foam mat dried powders.20,36,70

![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of carbonyl compounds and phenols. Medium peaks at 1249.93 cm−1 are attributed to C–H bending of proteins. Peaks at 1051.23 cm−1 and 1025.84 cm−1 are observed with lower transmittance, which are attributed to C–H or C–O bending/stretching in the phenolic compounds. The peaks at 865.96, 817.13 and 777.17 cm−1 are attributed to the presence of carbohydrates and benzene derivatives. The peaks at 586.81 cm−1 and 517.83 cm−1 are linked to P
O stretching of carbonyl compounds and phenols. Medium peaks at 1249.93 cm−1 are attributed to C–H bending of proteins. Peaks at 1051.23 cm−1 and 1025.84 cm−1 are observed with lower transmittance, which are attributed to C–H or C–O bending/stretching in the phenolic compounds. The peaks at 865.96, 817.13 and 777.17 cm−1 are attributed to the presence of carbohydrates and benzene derivatives. The peaks at 586.81 cm−1 and 517.83 cm−1 are linked to P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and S
O and S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds related to anthocyanins and sulfonic acid/sulfate groups, respectively. Also, a peak at 424.06 cm−1 is indicative of C
O bonds related to anthocyanins and sulfonic acid/sulfate groups, respectively. Also, a peak at 424.06 cm−1 is indicative of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bending in aromatic compounds. This observation aligns with previous studies on foam mat dried powder.20,71 The presence of phenolics, sugars, proteins and anthocyanin-related groups was evidenced by the appearance of characteristic absorption bands in both T2 and T6 powders corresponding to O–H, C–H, C
C bending in aromatic compounds. This observation aligns with previous studies on foam mat dried powder.20,71 The presence of phenolics, sugars, proteins and anthocyanin-related groups was evidenced by the appearance of characteristic absorption bands in both T2 and T6 powders corresponding to O–H, C–H, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, C–N/C–O, and aromatic compounds. However, notable differences were observed in the intensity of these peaks, which indicated the presence of compositional and structural differences between the two treatments. T6 exhibited an O–H stretch at 3294 cm−1 relative to 3279 cm−1 in T2, indicating stronger hydrogen bonding or higher retention of phenolic compounds. The enhanced OH bonding and the phenolic-related bands in T6 indicated stable SPI–polyphenol complexation, which can stabilize the molecular interaction and the reduced foaming activity of the SPI-based system. In contrast, T2 displayed less restrictive, looser interactions due to the unfolding of whey proteins with acid and the stabilization of the protein by fruit sugars and pectic materials as a synergistic effect. These differences are also consistent with the foaming attributes and functionality of WPI-formulations.
O, C–N/C–O, and aromatic compounds. However, notable differences were observed in the intensity of these peaks, which indicated the presence of compositional and structural differences between the two treatments. T6 exhibited an O–H stretch at 3294 cm−1 relative to 3279 cm−1 in T2, indicating stronger hydrogen bonding or higher retention of phenolic compounds. The enhanced OH bonding and the phenolic-related bands in T6 indicated stable SPI–polyphenol complexation, which can stabilize the molecular interaction and the reduced foaming activity of the SPI-based system. In contrast, T2 displayed less restrictive, looser interactions due to the unfolding of whey proteins with acid and the stabilization of the protein by fruit sugars and pectic materials as a synergistic effect. These differences are also consistent with the foaming attributes and functionality of WPI-formulations.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00×). The sample treated with whey protein had lamellar and fibrous-like networks with compact surfaces that supported efficient encapsulation. The structural heterogeneity was characterized by more porous and granular textures in a soy protein-based powder. The elemental composition of both powders was further confirmed by energy dispersive spectroscopy (EDS) analysis (Fig. 5). For the whey protein isolate (WPI) treated foam mat dried (FMD) sample (T2), the elements found to be present at greatest weight percent were carbon (51.46 wt%, 59.64 at%) and oxygen (36.49 wt%, 31.75 at%), with nitrogen (6.82 wt%, 6.78 at%) and trace amounts of sodium, magnesium, potassium, calcium and copper. Similarly, in the SPI-FMD mulberry powder sample (T6), carbon (51.88 wt%, 59.30 at%) and oxygen (35.72 wt%, 30.65 at%) were the dominant elements, followed by nitrogen (9.25 wt%, 9.06 at%). Trace amounts of magnesium, potassium, and copper were also detected at lower concentrations.
00×). The sample treated with whey protein had lamellar and fibrous-like networks with compact surfaces that supported efficient encapsulation. The structural heterogeneity was characterized by more porous and granular textures in a soy protein-based powder. The elemental composition of both powders was further confirmed by energy dispersive spectroscopy (EDS) analysis (Fig. 5). For the whey protein isolate (WPI) treated foam mat dried (FMD) sample (T2), the elements found to be present at greatest weight percent were carbon (51.46 wt%, 59.64 at%) and oxygen (36.49 wt%, 31.75 at%), with nitrogen (6.82 wt%, 6.78 at%) and trace amounts of sodium, magnesium, potassium, calcium and copper. Similarly, in the SPI-FMD mulberry powder sample (T6), carbon (51.88 wt%, 59.30 at%) and oxygen (35.72 wt%, 30.65 at%) were the dominant elements, followed by nitrogen (9.25 wt%, 9.06 at%). Trace amounts of magnesium, potassium, and copper were also detected at lower concentrations.

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×].
000×].
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×].
000×].

