Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Recovery of phytochemical compounds from the aqueous extract remaining after enzymatic extraction of sesame oil using cellulase and pepsin
Zeinab Zaheri Abdehvanda,
Mohammad Javaherian *a,
Maryam Kolahi*b and
Mohammad Sabaeiancd
*a,
Maryam Kolahi*b and
Mohammad Sabaeiancd
aDepartment of Chemistry, Faculty of Science, Shahid Chamran University of Ahvaz, Ahvaz, Iran. E-mail: m.kolahi@scu.ac.ir
bDepartment of Biology, Faculty of Science, Shahid Chamran University of Ahvaz, Ahvaz, Iran. E-mail: m.javaherian@scu.ac.ir
cDepartment of Physics, Faculty of Science, Shahid Chamran University of Ahvaz, Ahvaz, Iran
dCentre for Research on Laser and Plasma, Shahid Chamran University of Ahvaz, Iran
First published on 24th December 2025
Abstract
Sesame (Sesamum indicum L.) is rich in phytochemicals like phenolic compounds, lignans, and vitamins. Enzymatic extraction enhances the oil yield, yet its aqueous by-product is often discarded. The oil yields were 18.35% for the control, 25.86% for cellulase, and 21.83% for pepsin, with cellulase achieving the highest yield. This study evaluated the nutritional value of that extract using pepsin and cellulase. Total phenols in pepsin- and cellulase-treated samples were 52.9 and 37.3 mg/100 g (as gallic acid) and flavonoids were 29.65 and 12.24 mg/100 g (as quercetin), higher than the control (p < 0.05). Antioxidant capacities were 76.89%, 65.59%, and 60.77% for cellulase, pepsin, and control (p < 0.05), respectively. Enzyme treatment enhanced phenolic release and antioxidant activity. Carbohydrate and protein levels increased significantly, peaking in pepsin-treated samples (0.93 g/100 g and 88.84 mg/100 g, respectively). Pepsin hydrolyzes proteins and releases carbohydrates. Vitamin and pigment contents varied according to enzymes and reaction conditions. This study showed that sesame oil's enzymatic aqueous extract contains valuable bioactive compounds for use in food, pharmaceutical, and cosmetic industries.
Sustainability spotlightLarge amounts of aqueous by-products are generated during enzymatic oil extraction and are commonly discarded, creating waste and environmental burdens. This study demonstrates that the aqueous extract remaining after sesame oil extraction is not a residue but a valuable source of phenolics, vitamins, proteins, and antioxidants. By applying cellulase and pepsin, we enhanced the recovery of bioactive compounds that can be reused as functional ingredients in food, pharmaceutical, and cosmetic products. This sustainable approach reduces agro-industrial waste, supports resource efficiency, and adds economic value to sesame processing. This work directly aligns with the UN Sustainable Development Goals: **SDG 12 (Responsible Consumption and Production)** and **SDG 3 (Good Health and Well-being)** by promoting healthier products and circular bioeconomy practices. |
1. Introduction
Sesame (Sesamum indicum L.) is an annual herbaceous plant in the Pedaliaceae family, which is cultivated in tropical and subtropical regions. Due to its high oxidation resistance, it is known as the “queen of oilseeds”.1 Sesame seeds are recognized as healthy and nutritious in the Middle East and contain 83 to 90 percent unsaturated fatty acids. They are rich in minerals such as iron, phosphorus, calcium, magnesium, and copper. Sesame seeds and oil contain various phytochemicals, including phenolic acids, flavonoids, phytosterols, tocopherols, and lignans like sesamin, sesamol, and sesamolin.2 Sesame seeds also contain about 20 to 25 percent carbohydrates and proteins.3 Extraction is the first and most important step in isolating bioactive compounds from plant sources. The plant cell wall comprises complex polysaccharides such as cellulose, hemicellulose, lignin, and pectin, which significantly hinder access to phytochemicals and other valuable compounds.4In recent years, despite the availability of various methods for oil extraction from oilseeds, aqueous enzymatic extraction has emerged as a novel, safe, and environmentally friendly technique that can serve as an effective alternative to conventional methods. In this approach, water is used as the extraction medium, and enzymes are employed to break down cell walls and release oil.5 Enzyme-assisted aqueous extraction (EAE) is an innovative and environmentally friendly method that uses enzymes and water to simultaneously extract high-quality oil and protein from oilseeds. Optimizing the water-to-solid ratio in this process not only improves enzyme performance but also leads to the production of a valuable by-product called protein hydrolysates, which are small, soluble peptide fragments. These hydrolysates possess desirable functional properties such as solubility, emulsifying ability, and foaming capacity, making them highly useful in food, pharmaceutical, cosmetic, and animal feed industries.6 Enzymatic extraction is based on the hydrolysis of plant cell walls under optimized conditions. In this process, cell wall components interact with the enzyme's active site, causing a conformational change in the enzyme, which leads to effective interaction with the cell wall. This interaction results in the degradation of the cell structure and the easy release of bioactive compounds.7 In addition to oil and protein, phytochemical compounds such as phenols and flavonoids are also bound to the polysaccharides of the cell wall and are less frequently transferred to the solvent in traditional extraction methods. The use of enzymes in the extraction process breaks these bonds and significantly enhances the extraction of bioactive compounds.8 Enzymatic extraction is a novel and effective method to increase the yield and improve sesame oil quality; however, some bioactive and nutritious seed components, due to water solubility, transfer to the aqueous phase and are often discarded. Unlike previous studies which focused primarily on protein content,9 this study focuses on the identification and quantification of phytochemicals, vitamins, and pigments in the aqueous extract, providing baseline data on the bioactive and nutritional compounds present in this usually discarded by-product.
Enzyme-assisted aqueous extraction (EAE) is an innovative and environmentally friendly method that enables the simultaneous extraction of high-quality oil and protein from oilseeds using enzymes and water. In this process, optimizing the water-to-solid ratio not only enhances enzyme performance but also results in the production of valuable by-products—protein hydrolysates—with desirable functional properties such as solubility, emulsifying ability, and foaming capacity, making them suitable for use in food, pharmaceutical, and animal feed industries.6,9 This study aims to identify and quantify phytochemicals, vitamins, and pigments in the aqueous extract remaining after enzymatic extraction of sesame oil. Unlike previous studies that focused primarily on protein content, this study provides a comprehensive profile of bioactive and nutritional compounds present in these by-products and generates baseline information for exploring their potential for value-added utilization in future applications.
2. Materials and methods
Sesame (Sesamum indicum L.) seeds of a native Iranian variety were obtained in December 2022 from the Behbahan Agricultural Center (latitude: 30° 35′ 45.24″ N, longitude: 50° 14′ 30.12″ E). The seeds were manually cleaned to remove impurities. Their initial moisture content was 6%, as measured using a digital moisture analyzer (DBS60-3, Germany). All chemicals used in this study were procured from Merck (Germany) and Sigma. The pepsin enzyme (250 U mg−1), derived from porcine gastric mucosa, was purchased from Merck (Germany), and it exhibits optimal activity at pH 2–4 and in a temperature range of 37–42 °C. The cellulase enzyme (5 KU g−1), extracted from Pichia pastoris and supplied by Bioluence (Iran), functions optimally at pH 7–10 and temperatures between 40 and 60 °C.2.1. Enzyme-assisted aqueous extraction
The sesame seeds were ground and then passed through an 80-mesh sieve. Then, 10 g of the ground seeds were mixed with distilled water at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio in a 250 mL Erlenmeyer flask, resulting in a slurry-like mixture that allowed effective mixing and enzyme–substrate interaction. The pH was adjusted using 0.1 M NaOH or HCl (pH 2 for pepsin and pH 7 for cellulase). Enzymes were added at their optimal concentrations (2% w/v for pepsin and 3% w/v for cellulase), which were determined based on preliminary experiments aimed at optimizing the extraction yield and enzyme efficiency. A control sample without enzyme addition was prepared under the same conditions. All treatments, including controls, were performed in triplicate to ensure reproducibility. Hydrolysis was performed in a shaker incubator (Fanaveran Sahand Azar, Iran) at 40 °C and 150 rpm for 6 h. After incubation, the mixture was centrifuged at 10
2 ratio in a 250 mL Erlenmeyer flask, resulting in a slurry-like mixture that allowed effective mixing and enzyme–substrate interaction. The pH was adjusted using 0.1 M NaOH or HCl (pH 2 for pepsin and pH 7 for cellulase). Enzymes were added at their optimal concentrations (2% w/v for pepsin and 3% w/v for cellulase), which were determined based on preliminary experiments aimed at optimizing the extraction yield and enzyme efficiency. A control sample without enzyme addition was prepared under the same conditions. All treatments, including controls, were performed in triplicate to ensure reproducibility. Hydrolysis was performed in a shaker incubator (Fanaveran Sahand Azar, Iran) at 40 °C and 150 rpm for 6 h. After incubation, the mixture was centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 15 min (HS18500, Iran). The aqueous phase was collected using a micropipette. To deactivate enzyme activity, the extract was heated in an oven (UNB400, Germany) at 90 °C for 15 min.8 The aqueous phase was then centrifuged at 8000 rpm for 10 min to ensure clarity and remove suspended particles and subsequently collected for the analysis of phytochemicals, vitamins, pigments, and antioxidant capacity.
000 rpm for 15 min (HS18500, Iran). The aqueous phase was collected using a micropipette. To deactivate enzyme activity, the extract was heated in an oven (UNB400, Germany) at 90 °C for 15 min.8 The aqueous phase was then centrifuged at 8000 rpm for 10 min to ensure clarity and remove suspended particles and subsequently collected for the analysis of phytochemicals, vitamins, pigments, and antioxidant capacity.
2.2. Determination of the oil yield
The oil yield was determined using the equation below.
 | (1) |
2.3. Determination of total phenol and flavonol contents
Total phenolic content was determined using a modified Folin–Ciocalteu method. To 200 µL of the extract, 1.5 mL of 10% Folin reagent was added. After 6 min, 1.5 mL of 7.5% sodium carbonate solution was added, and the mixture was kept in the dark for 30 min. The development of a dark blue color indicated the presence of phenolics, and absorbance was measured at 760 nm using a V730 spectrophotometer (Japan). Phenolic content was expressed as mg of gallic acid per 100 g of fresh weight (FW).11 As Sánchez-Rangel et al.12 noted, reducing compounds in the extract can interfere with TPC measurements; however, no pre-treatment or correction was performed in this study, and therefore the results should be interpreted with caution.12The flavonol content was determined using the aluminum chloride colorimetric method. To 2 mL of the extract, 2 mL of aluminum chloride (20 mg mL−1) and 6 mL of sodium acetate (50 mg mL−1) were added and mixed. The absorbance was read at 440 nm after 2.5 h. The flavonol content was calculated using a quercetin calibration curve and expressed as mg quercetin per 100 g of sample.13
2.4. Determination of antioxidant activity and hydrogen peroxide (H2O2) content
The antioxidant activity (AA) was evaluated by the ability to scavenge the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radicals. A volume of 0.3 mL of each extract was mixed with 2.7 mL of DPPH radical solution (6 × 10−5 mol L−1). The mixture was thoroughly shaken and kept in the dark for 60 min until the absorbance stabilized. The reduction of DPPH radicals was monitored by measuring the decrease in absorbance at 517 nm, and the DPPH scavenging effect was calculated as the percentage of DPPH discoloration using the following equation:11| Antioxidant activity% = (DPPH absorption − sample absorption/DPPH absorption) × 100 | (2) |
For the determination of hydrogen peroxide (H2O2), 1 mL of sesame extract was mixed with 5 mL of trichloroacetic acid (TCA) solution (0.1 g in 100 mL distilled water), 2 mL of potassium iodide (KI) solution (16.6 g in 100 mL distilled water), and 1 mL of potassium phosphate buffer (10 mM, pH = 7.0). The reaction resulted in the formation of a colored product proportional to the H2O2 concentration in the sample. The absorbance of this product was measured using a spectrophotometer at a wavelength of 390 nm.14
2.5. Determination of vitamin C, vitamin B12 and folic acid (B9) content
Vitamin C content was determined by a slightly modified method of Hernández et al.15 using 0.1% dichlorophenolindophenol dye. 2 mL of the extract was mixed with 25 mL of distilled water, shaken for 60 min, filtered, and treated with 5% orthophosphoric acid. The volume of dye required to change the solution' color from colorless to onion-skin color was recorded.15Vitamin B12 content was determined using the method of Kumudha & Sarada (2015) with slight modifications. For this purpose, 4 mL of extract was mixed with 24 mL of acetate buffer (pH = 4) and 1 mL of sodium cyanide solution (0.25 g in 25 mL water). Then, 0.25 g of α-amylase was added, and the mixture was incubated at 42 °C for 30 min. After adjusting the pH to 4.8, the mixture was heated at 98 °C for an additional 30 min. The mixture was then centrifuged at 4000 rpm for 15 min at 4 °C, and the resulting supernatant was used to measure vitamin B12 using the MONOBIND kit.16
Folic acid content was determined using the method of Xiang et al. with slight modifications. For this purpose, 5 mL of the extract was homogenized with 10 mL of phosphate buffer containing 1% ascorbic acid. The resulting mixture was heated in a 100 °C water bath for 10 min and then immediately cooled on ice for 10 min. Afterwards, the sample was centrifuged at 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min at 4 °C. The resulting clear supernatant was used to measure the folic acid content using the MONOBIND kit.17
000 rpm for 10 min at 4 °C. The resulting clear supernatant was used to measure the folic acid content using the MONOBIND kit.17
2.6. Determination of pigment content
To measure photosynthetic pigments, the sample's absorbance was recorded at 470, 646, and 663 nm using a UV-vis spectrophotometer. The levels of chlorophyll a, chlorophyll b, total chlorophyll, and carotenoids were calculated (eqn (3)–(6)):18| Chl a (µg mL−1) = (12.25A663) − (2.79A646) | (3) |
| Chl b (µg mL−1) = (21.50A646) − (5.1A663) | (4) |
| Chl T (µg mL−1) = Chl a + Chl b | (5) |
| Carotenoid (µg mL−1) = (1000A470 − 1.82Chl a − 85.02Chl b)/198 | (6) |
The total anthocyanin content (TAC) in the extracts was determined spectrophotometrically. Briefly, 2 mL of extract was mixed with 10 mL of acidic methanol containing 1 mL of hydrochloric acid. After centrifugation, the absorbance of the clear supernatant was measured at 550 nm, and the anthocyanin content was calculated according to eqn (7).
| A = εbc | (7) |
2.7. Determination of carbohydrate and protein content
Carbohydrate content was determined by mixing 1 mL of extract (diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 with water) with 0.5 mL of 5% phenol and 2.5 mL of 98% sulfuric acid. After 10 min, the samples were vortexed for 30 s and incubated at room temperature for 20 minutes. A color change indicated the presence of sugars, and absorbance was measured at 490 nm. The carbohydrate content was quantified based on a glucose standard calibration curve and reported as mg of glucose per 100 g of sample.20
100 with water) with 0.5 mL of 5% phenol and 2.5 mL of 98% sulfuric acid. After 10 min, the samples were vortexed for 30 s and incubated at room temperature for 20 minutes. A color change indicated the presence of sugars, and absorbance was measured at 490 nm. The carbohydrate content was quantified based on a glucose standard calibration curve and reported as mg of glucose per 100 g of sample.20
The aqueous extract was freeze-dried (FD-5005-BT, Iran), and its protein content was subsequently determined.6 For protein determination, the ABC reagent was first prepared:
Reagent A: 2 g sodium hydroxide and 10 g sodium carbonate in 100 mL distilled water.
Reagent B: 1 g copper sulfate in 25 mL distilled water.
Reagent C: 4.91 g sodium potassium tartrate tetrahydrate in 100 mL distilled water.
Preparation of ABC reagent: 15 mL of reagent A, 0.75 mL of reagent B, and 0.75 mL of reagent C were mixed.
Then, 1 g of the freeze-dried extract powder was mixed with 0.1 M potassium phosphate buffer at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 ratio and centrifuged. 5 mL of the ABC solution was added to 1 mL of the supernatant and incubated at room temperature for 15 min. Next, 3 mL of 2 N Folin reagent was added to each sample, and after 30 min of incubation, the absorbance was measured at 660 nm. The protein content was determined using a bovine serum albumin (BSA) standard calibration curve and expressed as mg of BSA per 100 g of sample.21
10 ratio and centrifuged. 5 mL of the ABC solution was added to 1 mL of the supernatant and incubated at room temperature for 15 min. Next, 3 mL of 2 N Folin reagent was added to each sample, and after 30 min of incubation, the absorbance was measured at 660 nm. The protein content was determined using a bovine serum albumin (BSA) standard calibration curve and expressed as mg of BSA per 100 g of sample.21
2.8. Data analysis
Data analysis was performed using SPSS version 20.0 (SPSS Inc., Chicago, IL, USA). The results are presented as mean ± standard deviation. Each experiment was conducted in triplicate to ensure accuracy. Due to the small sample size and to avoid assumptions of normality, statistical comparisons were performed using the non-parametric Kruskal–Wallis test. A significance level of p <0.05 was applied for all analyses.3. Results and discussion
3.1. Evaluation of the oil yield using different extraction methods
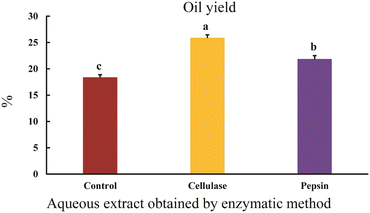
The results indicated that enzymatic extraction using cellulase and pepsin significantly increased the oil yield compared to the control. The oil yields were 18.35% for the control, 25.86% for cellulase, and 21.83% for pepsin, with cellulase achieving the highest yield (p < 0.05) (Fig. 1). The addition of enzymes such as cellulase, protease, and pectinase during aqueous extraction enhances the oil yield by breaking down the cell wall and hydrolyzing structural polysaccharides and lipoprotein bodies.6 Temperature and pH also influence the activity of cellulase and pepsin, playing an important role in extraction efficiency. Additionally, pepsin facilitates oil release by breaking down oleosomes, the lipid structures that store oil in plant cells. These findings highlight the important role of enzymes in optimizing sesame oil extraction.83.2. Evaluation of phenolic content in aqueous sesame extracts from different extraction methods
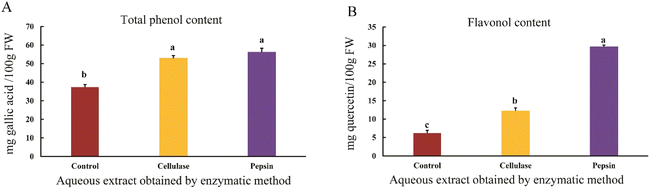
The results showed that the total phenolic and flavonol contents of the aqueous sesame extract increased when using pepsin and cellulase enzymes compared to the control sample (p < 0.05) (Fig. 2A and B). The plant cell wall is composed of cellulose, hemicellulose, pectin, and proteins, and phenolic compounds are typically bound to polysaccharides within this structure. Enzymes facilitate the release of phenolic compounds by degrading the cell wall structure and cleaving bonds, thereby enhancing the presence of bioactive compounds.4 The use of proteolytic enzymes can increase the phenolic content compared to solvent extraction.22 Additionally, Neutrase is more effective than pectinase in disrupting the cell wall and releasing phenolic compounds. In general, proteases, cellulases, and pectinases, through the hydrolysis of polysaccharides and lipid bodies, significantly improve the extraction of phenolic and protein compounds. Studies have shown that the release of phenolic compounds by proteases such as Alcalase and protamex was higher than that of Kemzyme; however, another carbohydrase, Viscozyme, performed better in releasing phenolic compounds. This difference depends on the nature and spectrum of enzyme activity. Pepsin, due to its proteolytic property and ability to hydrolyze protein matrices, provides better release of phenolics bound to proteins.4 Ghandahari Yazdi et al.23 demonstrated that enzymatic treatments increase the yield of phenolic compound extraction. Moreover, the use of enzyme combinations enhances the release of these compounds. In addition to the type of enzyme, factors such as particle size, enzyme concentration, sample-to-solvent ratio, and reaction time also significantly impact polyphenol extraction.23 Reducing the particle size results in more effective disruption of the cell wall, facilitating the release of phenolic compounds. Smaller particles also have a higher surface area-to-volume ratio, which increases the contact area between the enzymes, solvent, and substrate particles. In this study, it appears that grinding the sesame seeds and passing them through a sieve resulted in uniform and fine particle size, which led to effective disruption of the cell wall.24 Despite the increased release of phenolic compounds, the TPC measured by the Folin–Ciocalteu method may be influenced by reducing compounds present in the extract. As reported by Sánchez-Rangel et al.,12 compounds such as ascorbic acid can elevate the measured TPC. In this study, no corrections were applied to eliminate these interferences; therefore, the results should be interpreted with caution.123.3. Evaluation of antioxidant activity and H2O2 content in aqueous sesame extracts from different extraction methods
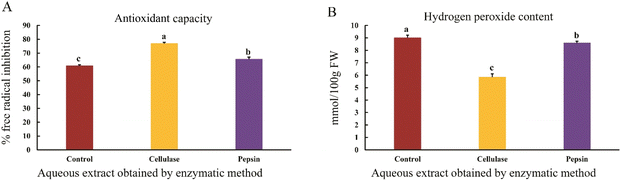
The results showed that the antioxidant capacity of the aqueous sesame extract increased when using cellulase and pepsin enzymes compared to the control sample (p < 0.05) (Fig. 3A). Antioxidants are compounds that prevent damage caused by reactive oxygen species. Enzymatic extraction facilitates the release of phenolic compounds bound to the cell wall, increasing the antioxidant capacity. This process is influenced by factors such as temperature, time, enzyme concentration, and the sample-to-solvent ratio.25 There is a direct relationship between the amount of phenolic compounds and the ability to scavenge free radicals; as the amount of phenolic compounds increases, the antioxidant activity significantly improves. Studies have shown that enzymatic extraction using a combination of cellulase, hemicellulase, pectinase, and papain from orange peel has a greater effect on antioxidant capacity compared to ultrasonic and solvent extraction methods.26 Additionally, enzymatic extraction leads to an increase in phenolic compounds, and at higher concentrations the increased number of hydroxyl groups enhances the potential for donating hydrogen to free radicals, thereby increasing the inhibition power.27The results showed that the H2O2 content of the aqueous sesame extract decreased when using pepsin and cellulase enzymes compared to the control sample. The lowest hydrogen peroxide content was observed in the extract obtained using cellulase (p < 0.05) (Fig. 3B). The findings indicate that phenolic compounds play a significant role as antioxidants by inhibiting hydrogen peroxide and scavenging free radicals, thereby enhancing the antioxidant capacity.28 The water-to-sample ratio also affects the stability of extracted compounds; increasing this ratio elevates dissolved oxygen, which may promote oxidation of certain compounds. Papain enzyme, by hydrolyzing proteins, enhances antioxidant activity and reduces hydrogen peroxide content in the extract.29 It appears that a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water-to-sample ratio reduces oxidation by lowering dissolved oxygen. A strong correlation was observed between antioxidant activity and total phenolic content (DPPH, R2 = 0.75; H2O2, R2 = 0.71) in wild vegetable extracts from Nepal. Phenolic compounds act as hydrogen or electron donors, and their hydroxyl groups directly contribute to antioxidant activity, resulting in the scavenging of free radicals and the reduction of oxidative species such as H2O2.30 Enzymatic extraction increases both phenolic content and the antioxidant capacity of the extract.23 Moreover, several studies have reported similar trends in various plant extracts, and although correlation analysis was not performed in this study, a similar pattern appears to exist between phenolic content, antioxidant capacity, and H2O2 levels.
1 water-to-sample ratio reduces oxidation by lowering dissolved oxygen. A strong correlation was observed between antioxidant activity and total phenolic content (DPPH, R2 = 0.75; H2O2, R2 = 0.71) in wild vegetable extracts from Nepal. Phenolic compounds act as hydrogen or electron donors, and their hydroxyl groups directly contribute to antioxidant activity, resulting in the scavenging of free radicals and the reduction of oxidative species such as H2O2.30 Enzymatic extraction increases both phenolic content and the antioxidant capacity of the extract.23 Moreover, several studies have reported similar trends in various plant extracts, and although correlation analysis was not performed in this study, a similar pattern appears to exist between phenolic content, antioxidant capacity, and H2O2 levels.
3.4. Evaluation of vitamin content in aqueous sesame extracts from different extraction methods
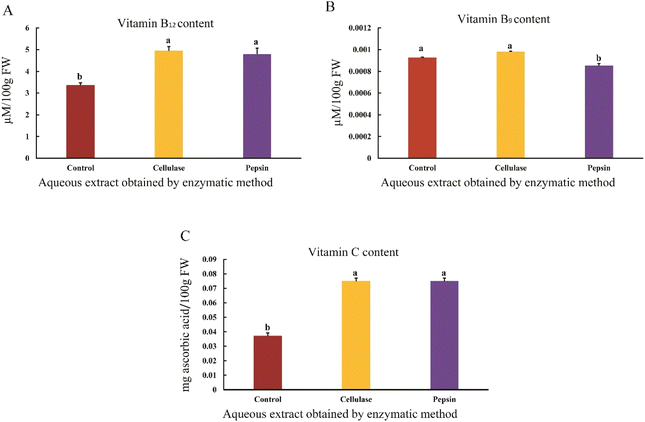
The results showed that vitamin B12 content of the aqueous sesame extract increased when using pepsin and cellulase enzymes compared to the control sample (p < 0.05) (Fig. 4A). Recent studies have shown that B vitamins act as strong antioxidants and are capable of deactivating reactive oxygen species.31 Vitamin B12 is significantly affected by oxidizing and reducing agents and is degraded when exposed to reactive oxygen species, such as hydroxyl radicals.32 Additionally, high temperatures can lead to the degradation of B12, which is related to the removal of the base part, changes in the lower ligand sugar part, and structural alterations. It seems that lower temperatures in this study prevented the degradation of vitamin B12.33 Enzymatic extraction with proteases like pepsin and papain can break peptide bonds and release vitamin B12 from protein complexes.34 Vitamin B12 binds to dietary proteins, and these complexes are hydrolyzed by pancreatic proteases in the body.35 Furthermore, cellulase is used to break down cellulose and improve access to other nutrients, and it appears to increase the amount of vitamin B12.36The results showed that the folic acid content of the aqueous sesame extract increased when using cellulase and pepsin enzymes compared to the control sample. No significant difference was found between the folic acid content in the extracts obtained using cellulase and pepsin enzymes (p < 0.05) (Fig. 4B). Folic acid is the oxidized form and monoglutamate of vitamin B9, which exists in plants in both stored and free forms. Processes such as oxidation and enzymatic conversion transform this vitamin into active folate forms.32 Degradation of the cell wall and pectin leads to the release of folate into the intercellular space. High temperatures during storage can oxidize folate and reduce its amount. In contrast, enzymatic extraction with cellulase increases the folate content, as cellulase helps break down the cell wall and facilitates access to folate.37 Furthermore, the folate extraction process involves the release of folate from the cellular matrix and the conversion of polyglutamate forms to monoglutamate and diglutamate forms. Enzymatic extraction releases the folate-bound forms. To extract folate from cereal seeds, protease and alpha-amylase enzymes were used, but prolonged incubation can reduce folate content due to oxidation.38
The results showed that the vitamin C content of the aqueous sesame extract increased when using pepsin and cellulase enzymes compared to the control sample (p < 0.05) (Fig. 4C). Vitamin C, as a water-soluble compound, diffuses into the aqueous environment during processes such as soaking.39 Studies have shown that enzymatic extraction of date juice using cellulase and pectinase enzymes leads to cell wall degradation and the release of vitamin C. Various factors, such as enzyme concentration, water-to-substrate ratio, time, and temperature, significantly influence the ascorbic acid content. Higher enzyme concentrations improve access to vitamin C, while elevated temperatures and higher water ratios over prolonged periods result in reduced ascorbic acid levels.40 Ascorbic acid is most stable within a pH range of 2–4.41 It appears that in pepsin-assisted extraction, due to the use of an acidic pH (2), a temperature of 40 °C, and a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 sample-to-solvent ratio, the ascorbic acid (vitamin C) content was preserved and increased.
2 sample-to-solvent ratio, the ascorbic acid (vitamin C) content was preserved and increased.
3.5. Evaluation of pigment content in aqueous sesame extracts from different extraction methods
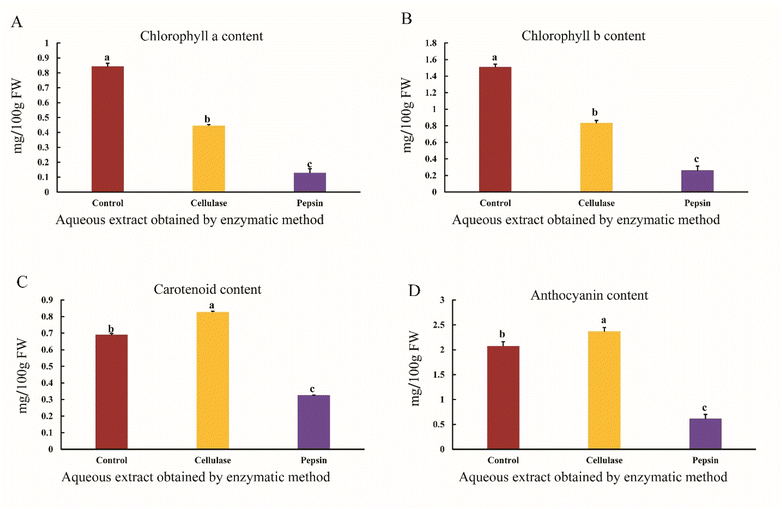
Chlorophyll a and b contents of the aqueous sesame extract were measured in control and enzyme-treated samples (Fig. 5A and B). Although sesame seeds are mainly valued for their lipids and phenolics, small amounts of chlorophyll and its derivatives can also be detected in ethanolic extracts. These pigments likely come from residual green tissues, immature seeds, or seed coat fragments remaining after harvesting and cleaning. Ethanol, as a semi-polar solvent, can dissolve both phenolics and certain chlorophyll-type compounds. Thus, chlorophyll in sesame seed extracts may result from the co-extraction of these minor pigments. The results showed that chlorophyll a and chlorophyll b contents of the aqueous sesame extract decreased when using cellulase and pepsin enzymes compared to the control sample (p < 0.05). Extraction is recognized as a key step in isolating bioactive compounds from plants. Parameters such as enzyme concentration, temperature, time, and pH significantly affect the extraction process. Changes in any of these parameters can impact the total chlorophyll content.42 Previous studies have shown that increasing enzyme concentration, extraction time, and temperature lead to a decrease in total chlorophyll content. Chlorophyll is a fat-soluble pigment, and its amount decreases in aqueous extracts. The degradation rate of chlorophyll a and b increases with decreasing pH, with chlorophyll a degrading faster than chlorophyll b, and overall the total chlorophyll content significantly decreases under acidic conditions.43 The cellulase enzyme, by degrading the cell wall and breaking down the cellulose matrix, releases the stored chlorophyll. However, if the cellulase concentration is too high, it can lead to the degradation of sensitive compounds such as chlorophyll.44 As shown in Fig. 5A and B, it appears that pepsin-assisted extraction, due to the acidic pH (2), and cellulase-assisted extraction, due to the higher enzyme concentration (3% w/v), may result in a decrease in chlorophyll a and b contents compared to the control in the aqueous sesame extract.The results showed that the carotenoid content of the aqueous sesame extract increased when using cellulase enzymes compared to the control sample, while extraction with the pepsin enzyme resulted in a decrease in carotenoid content compared to the control sample (p < 0.05) (Fig. 5C). Studies have shown that enzymes such as cellulase and pectinase can increase carotenoid levels by breaking down the cell wall. In the present study, the use of cellulase resulted in higher carotenoid extraction, whereas pepsin had a lower effect and likely caused a decrease in carotenoid content compared to the control, probably due to the acidic extraction conditions.45 Cellulase releases carotenoids trapped in the plant matrix by breaking down the cellulose in the cell wall. Studies have shown that using cellulase significantly increases carotenoid extraction. Optimal conditions, such as neutral pH and mild temperatures, help prevent carotenoid degradation.36 In contrast, acidic environments cause isomerization or oxidative degradation of carotenoid structures, reducing carotenoid stability.46 As shown in Fig. 5C, cellulase-assisted extraction, due to a temperature of 40 °C and neutral pH (7), helped preserve carotenoids in the aqueous sesame extract compared to the control, whereas pepsin-assisted extraction, due to the acidic pH (2), likely caused oxidation of carotenoids and a decrease in their content compared to the control.
The results showed that the anthocyanin content of the aqueous sesame extract did not differ significantly when using the cellulase enzyme compared to the control sample, and extraction with the pepsin enzyme resulted in a decrease in anthocyanin content compared to the control sample (p < 0.05) (Fig. 5D). Anthocyanins are natural, water-soluble, safe, and non-toxic pigments that, with their antioxidant properties, play a role in eliminating free radicals and improving human health. These compounds dissolve well in water and organic solvents such as ethanol, methanol, and acetone. Enzymes, by gently breaking down cell walls and intercellular structures, release anthocyanins into the extraction medium.47 Cellulase aids in the breakdown of potato cell walls, while papain helps by hydrolyzing cellular proteins, contributing to the release and preventing oxidation of anthocyanins. Studies have shown that the highest yield of anthocyanins has been reported at 40 °C, and higher temperatures result in a decrease. Moreover, increasing the water-to-sample ratio increases dissolved oxygen, leading to oxidation and a reduction in anthocyanins.29 As shown in Fig. 5D, in this study, the use of low temperature (40 °C) and a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 sample-to-water ratio, along with cell wall degradation by cellulase, helped increase anthocyanin content. However, under similar conditions with pepsin, the acidic pH (2) likely led to the degradation of these compounds. Moreover, recent studies have reported that colored sesame seeds (brown, black, and yellow) contain anthocyanins, which play a significant role in their antioxidant capacity.48,49 In the present study, the sesame seeds used had a dark-colored seed coat, which could explain the presence of anthocyanins detected in the extract.
2 sample-to-water ratio, along with cell wall degradation by cellulase, helped increase anthocyanin content. However, under similar conditions with pepsin, the acidic pH (2) likely led to the degradation of these compounds. Moreover, recent studies have reported that colored sesame seeds (brown, black, and yellow) contain anthocyanins, which play a significant role in their antioxidant capacity.48,49 In the present study, the sesame seeds used had a dark-colored seed coat, which could explain the presence of anthocyanins detected in the extract.
3.6. Evaluation of carbohydrate and protein content in aqueous sesame extracts from different extraction methods
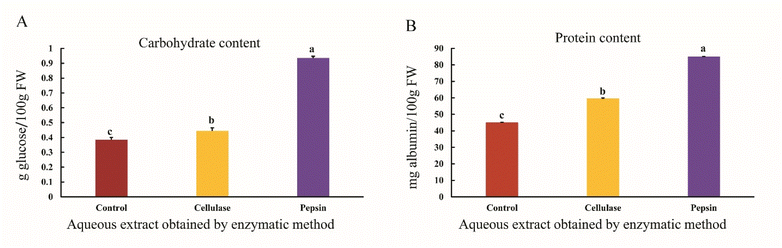
The results showed that the carbohydrate content of the aqueous sesame extract increased when using pepsin and cellulase enzymes compared to the control sample (p < 0.05) (Fig. 6A). The plant cell wall is a strong structure composed of cellulose, proteins, and polysaccharide–peptide complexes, which creates a physical barrier that prevents the release of bioactive compounds. Protease enzymes break down the amide bonds in proteins, while cellulase enzymes release carbohydrates by breaking the β-1,4 bonds in polysaccharides.50 Enzymatic extraction is an environmentally friendly and effective method for increasing the yield of carbohydrate extraction. Studies have shown that various factors, such as extraction time, temperature, and pH, affect the extraction yield, with the pH plays a determining role in the process. Increasing the pH within a specific range leads to a higher carbohydrate content in blueberries, whereas beyond this range, the yield decreases.51 Proteins can hinder the release of carbohydrates; therefore, the use of protease enzymes, through the hydrolysis of proteins, increases the carbohydrate extraction yield.52 It appears that extraction with the enzyme pepsin, through protein hydrolysis, facilitates the release of carbohydrates and increases their yield, whereas cellulase, by degrading the polysaccharides in the cell wall, has a lesser effect on the carbohydrate yield, which depends on the pH of the environment and the type of enzyme activity.The results showed that the protein content of the aqueous sesame extract increased when using pepsin and cellulase enzymes compared to the control sample (p < 0.05) (Fig. 6B). Hydrolysis time significantly affects nutritional parameters such as essential amino acid index, biological value, and protein efficiency ratio. In one study, increasing the enzymatic hydrolysis time from 0 to 180 min resulted in 27% protein degradation with the pepsin enzyme.53 Enzymatic extraction is primarily influenced by pH and temperature, and under optimal conditions, it leads to higher protein recovery.54 The enzyme-assisted aqueous extraction (EAE) process disrupts the cell structure and hydrolyzes macromolecular compounds such as lipoproteins and polysaccharides, resulting in the release of oil and protein. These compounds are separated due to differences in density and their combined properties. Proteases improve the extraction of oil and protein by breaking down proteins into smaller molecules.9 Protease enzymes, under optimal conditions, are capable of producing hydrolyzed proteins, while cellulase enzymes do not significantly affect the amount of proteins.55 As shown in Fig. 6B, pepsin-assisted extraction led to an increase in protein content in the aqueous sesame extract compared to the control, whereas cellulase had no significant effect on protein amount. These results indicate that protein hydrolysis by proteases plays a key role in the release and recovery of proteins. The higher carbohydrate content observed in the pepsin-assisted extract compared to the cellulase extract can be attributed to the specific actions of the enzymes. Proteases are capable of hydrolyzing over 50% of the matrix proteins and releasing glucose-based oligomers, indicating that proteinaceous barriers can limit the solubilization of carbohydrates.56 One of the main obstacles faced by cellulase enzymes during lignocellulose hydrolysis is their limited access to the bulk of the cellulose.57 Thus, the presence of proteins acts as a physical barrier, preventing enzymes such as cellulase from efficiently hydrolyzing cellulose. These findings underscore the importance of protein hydrolysis in improving carbohydrate extraction from plant matrices.
4. Conclusion
The results showed that the remaining aqueous extract after sesame oil extraction using pepsin and cellulase enzymes had higher levels of carbohydrates, protein, vitamins B12 and C, folic acid, phenolic compounds, and flavonoids and high antioxidant capacity compared to the control. The extract obtained with pepsin exhibited the greatest increase in protein and carbohydrate content, highlighting its potential application in the food industry due to improved nutritional value, texture, and product stability. Pigment content varied; chlorophyll a and b levels decreased, while carotenoid and anthocyanin contents increased in the extract obtained with cellulase and decreased in the extract obtained with pepsin. This difference may be due to the acidic pH during extraction with pepsin. Rather than being discarded, this nutrient- and bioactive-rich extract can serve as a valuable ingredient in the food, pharmaceutical, and cosmetic industries. Moreover, the economic feasibility of the extraction process was not assessed in this study and should be considered in future research to evaluate its industrial applicability. This study can serve as a basis for industrial applications and the development of new products using this extract.Conflicts of interest
The authors declare that there are no conflict of interest and that they are responsible for the content and writing of the article.Data availability
All data supporting the findings of this study are included within the article. No supplementary information (SI) is provided.Acknowledgements
This work was funded by a grant from the Shahid Chamran University of Ahvaz Research Council.References
- R. Batool, T. Batool, F. U. Nisa, F. Khalid and Z. Asghar, Ethnobotanical, Pharmacological, and Therapeutic Importance of Sesame Seeds Along with Their Role in the Biogenic Synthesis of Important Chemicals, in Therapeutic and Pharmacological Applications of Ethnobotany, IGI Global, 2024, pp. 241–273, DOI:10.4018/979-8-3693-1986-4.ch009.
- M. Li, J. Luo, M. A. Nawaz, R. Stockmann, R. Buckow and C. Barrow, et al., Phytochemistry, Bioaccessibility, and Bioactivities of Sesame Seeds: An Overview, Food Rev. Int., 2024, 40(1), 309–335, DOI:10.1080/87559129.2023.2168280.
- M. H. H. Khodaparast, M. B. H. Najafi, A. H. E. Rad and N. Divandari, Process optimization of sesame milk according to Iranian preferences, IFSTA, 2006 DOI:10.22067/ifstrj.v2i1.222.
- J. Azmir, I. S. M. Zaidul, M. M. Rahman, K. M. Sharif, A. Mohamed and F. Sahena, et al., Techniques for extraction of bioactive compounds from plant materials: a review, J. Food Eng., 2013, 117(4), 426–436, DOI:10.1016/j.jfoodeng.2013.01.014.
- M. Balvardi, K. Rezaei, J. A. Mendiola and E. Ibáñez, Optimization of the Aqueous Enzymatic Extraction of Oil from Iranian Wild Almond, J. Am. Oil Chem. Soc., 2015, 92(7), 985–992, DOI:10.1007/s11746-015-2671-y.
- S. Latif and F. Anwar, Aqueous enzymatic sesame oil and protein extraction, Food Chemi., 2011, 125(2), 679–684, DOI:10.1016/j.foodchem.2010.09.064.
- F. Shahidi, Bailey's Industrial Oil and Fat Products, Industrial and Nonedible Products from Oils and Fats, John Wiley & Sons, 2005, vol. 6 Search PubMed.
- Z. Z. Abdehvand, M. Javaherian, M. Kolahi and M. Sabaeian, Evaluation of the quality and phytochemical composition of sesame oil extracted using cellulase and pepsin enzymes, Appl. Food Res., 2025, 5(2), 101046, DOI:10.1016/j.afres.2025.101046.
- C. Liu, L. Hao, F. Chen and C. Yang, Study on extraction of peanut protein and oil bodies by aqueous enzymatic extraction and characterization of protein, J. Chem., 2020, 2020(1), 5148967, DOI:10.1155/2020/5148967.
- J. Piseskul, U. Suttisansanee, C. Chupeerach, C. Khemthong, S. Thangsiri and P. Temviriyanukul, et al., Optimization of enzyme-assisted mechanical extraction process of Hodgsonia heteroclita oilseeds and physical, chemical, and nutritional properties of the oils, Foods, 2023, 12(2), 292, DOI:10.3390/foods12020292.
- R. Esmaeilzadeh Kenari, F. Mohsenzadeh and Z. R. Amiri, Antioxidant activity and total phenolic compounds of Dezful sesame cake extracts obtained by classical and ultrasound-assisted extraction methods, Food Sci. Nutr., 2014, 2(4), 426–435, DOI:10.1002/fsn3.118.
- J. C. Sánchez-Rangel, J. Benavides, J. B. Heredia, L. Cisneros-Zevallos and D. A. Jacobo-Velázquez, The Folin–Ciocalteu assay revisited: improvement of its specificity for total phenolic content determination, Anal. Methods, 2013, 5(21), 5990–5999, 10.1039/C3AY41125G.
- A. Kumaran and R. J. Karunakaran, In vitro antioxidant activities of methanol extracts of five Phyllanthus species from India, LWT--Food Sci. Technol., 2007, 40(2), 344–352, DOI:10.1016/j.foodchem.2005.09.079.
- Z. Fartoosi, F. Heidarizadeh, M. Kolahi and A. Goldson-Barnaby, Phytochemical and nutritional analysis of Daucus carota L. (carrots) exposed to various processing conditions, Appl. Food Res., 2025, 101414, DOI:10.1016/j.afres.2025.101414.
- Y. Hernández, M. G. Lobo and M. González, Determination of vitamin C in tropical fruits: a comparative evaluation of methods, Food Chem., 2006, 96(4), 654–664, DOI:10.1016/j.foodchem.2005.04.012.
- A. Kumudha and R. Sarada, Effect of different extraction methods on vitamin B12 from blue green algae, Spirulina platensis, Pharm. Anal. Acta, 2015, 6(337), 10–4172, DOI:10.4172/2153-2435.1000337.
- N. Xiang, J. Hu, T. Wen, M. A. Brennan, C. S. Brennan and X. Guo, Effects of temperature stress on the accumulation of ascorbic acid and folates in sweet corn (Zea mays L.) seedlings, J. Sci. Food Agric., 2020, 100(4), 1694–1701, DOI:10.1002/jsfa.10184.
- O. Youssef, N. B. Youssef, Z. Mokhtar and F. Guido, Influence of olive storage period on volatile compounds and oil quality of two Tunisian cultivars of Olea europea, Chemlali and Chetoui, Int. J. Food Sci. Technol., 2011, 46(6), 1245–1252, DOI:10.1111/j.1365-2621.2011.02621.x.
- Z. Fartoosi, M. Kolahi, F. Heidarizadeh and A. Goldson-Barnaby, The impact of thermal and non-thermal processing on the phytochemical characteristics and nutritional value of Daucus carota (carrots), Appl. Food Res., 2025, 5(1), 100732, DOI:10.1016/j.afres.2025.100732.
- P. C. Quero-Jiménez, O. N. Montenegro, R. Sosa, D. L. Pérez, A. Santana Rodríguez and R. Ramos Méndez, et al., Total carbohydrates concentration evaluation in products of microbial origin, Afinidad, 2019, 76(587), 195–203 Search PubMed.
- T. Tanaka, Y. Abo, S. Hamano, Y. Fujishima and Y. Kaneo, Intracellular disposition of arabinogalactan and asialofetuin in HepG2 cells, J. Drug Delivery Sci. Technol., 2013, 23(5), 435–438, DOI:10.1016/S1773-2247(13)50062-1.
- S. Latif and F. Anwar, Effect of Aqueous Enzymatic Processes on Sunflower Oil Quality, J. Am. Oil Chem. Soc., 2009, 86(4), 393–400, DOI:10.1007/s11746-009-1357-8.
- A. P. Ghandahari Yazdi, M. Barzegar, M. A. Sahari and H. Ahmadi Gavlighi, Optimization of the enzyme-assisted aqueous extraction of phenolic compounds from pistachio green hull, Food Sci. Nutr., 2019, 7(1), 356–366, DOI:10.1002/fsn3.900.
- M. Pinelo, B. Zornoza and A. S. Meyer, Selective release of phenols from apple skin: mass transfer kinetics during solvent and enzyme-assisted extraction, Sep. Purif. Technol., 2008, 63(3), 620–627, DOI:10.1016/j.seppur.2008.07.007.
- M. R. Islam, M. M. Kamal, M. R. Kabir, M. M. Hasan, A. R. Haque and S. K. Hasan, Phenolic compounds and antioxidants activity of banana peel extracts: testing and optimization of enzyme-assisted conditions, Meas. Food, 2023, 10, 100085, DOI:10.1016/j.meafoo.2023.100085.
- Z. Wang, X. Mei, X. Chen, S. Rao, T. Ju and J. Li, et al., Extraction and recovery of bioactive soluble phenolic compounds from brocade orange (Citrus sinensis) peels: effect of different extraction methods thereon, LWT--Food Sci. Technol., 2023, 173, 114337, DOI:10.1016/j.lwt.2022.114337.
- S. L. Rodríguez De Luna, R. E. Ramírez-Garza and S. O. Serna Saldívar, Environmentally Friendly Methods for Flavonoid Extraction from Plant Material: Impact of Their Operating Conditions on Yield and Antioxidant Properties, Sci. World J., 2020, 1–38, DOI:10.1155/2020/6792069.
- Z. Sroka and W. Cisowski, Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids, Food Chem. Toxicol., 2003, 41(6), 753–758 CrossRef CAS PubMed.
- F. Wang, S. Zhang, G. Deng, K. Xu, H. Xu and J. Liu, Extracting total anthocyanin from purple sweet potato using an effective ultrasound-assisted compound enzymatic extraction technology, Molecules, 2022, 27(14), 4344, DOI:10.3390/molecules27144344.
- S. Aryal, M. K. Baniya, K. Danekhu, P. Kunwar, R. Gurung and N. Koirala, Total phenolic content, flavonoid content and antioxidant potential of wild vegetables from Western Nepal, Plants, 2019, 8(4), 96, DOI:10.3390/plants8040096.
- N. Abdiani, M. Kolahi, M. Javaheriyan and M. Sabaeian, Effect of storage conditions on nutritional value, oil content, and oil composition of sesame seeds, J. Agric. Food Res., 2024, 16, 101117, DOI:10.1016/j.jafr.2024.101117.
- Y. M. Hemery, L. Fontan, A. Laillou, V. Jallier, R. Moench-Pfanner and S. Avallone, et al., Influence of storage conditions and packaging of fortified wheat flour on microbial load and stability of folate and vitamin B12, Food Chemistry: X, 2020, 5, 100076, DOI:10.1016/j.fochx.2019.100076.
- F. Watanabe, Y. Yabuta, Y. Tanioka and T. Bito, Biologically Active Vitamin B12 Compounds in Foods for Preventing Deficiency among Vegetarians and Elderly Subjects, J. Agric. Food Chem., 2013, 61(28), 6769–6775, DOI:10.1021/jf401545z.
- E. Campos-Gimnez, P. Fontannaz, M. J. Trisconi, T. Kilinc, C. Gimenez and P. Andrieux, Determination of vitamin B12 in food products by liquid chromatography/UV detection with immunoaffinity extraction: single-laboratory validation, J. AOAC Int., 2008, 91(4), 786–793, DOI:10.1093/jaoac/91.4.786.
- Ž. Temova Rakuša, R. Roškar, N. Hickey and S. Geremia, Vitamin B12 in foods, food supplements, and medicines—a review of its role and properties with a focus on its stability, Molecules, 2022, 28(1), 240, DOI:10.3390/molecules28010240.
- S. Natarajan, S. A. Yadav and S. P. Manoharan, Influence of macerating enzyme-cellulase on the extraction of valuable compounds: carotenoid and camptothecin, Int. J. Res. Pharm. Sci., 2018, 9, 115–120 CrossRef CAS.
- F. Coffigniez, M. Rychlik, C. Sanier, C. Mestres, L. Striegel and P. Bohuon, et al., Localization and modeling of reaction and diffusion to explain folate behavior during soaking of cowpea, J. Food Eng., 2019, 253, 49–58, DOI:10.1016/j.jfoodeng.2019.02.012.
- S. Cho, Y. Choi, J. Lee and R. R. Eitenmiller, Optimization of Enzyme Extractions for Total Folate in Cereals Using Response Surface Methodology, J. Agric. Food Chem., 2010, 58(19), 10781–10786, DOI:10.1021/jf102751w.
- E. Burguieres, P. McCue, Y. I. Kwon and K. Shetty, Effect of vitamin C and folic acid on seed vigour response and phenolic-linked antioxidant activity, Bioresour. Technol., 2007, 98(7), 1393–1404, DOI:10.1016/j.biortech.2006.05.046.
- S. Mohanty, S. Mishra and R. C. Pradhan, Optimisation of enzymatic extraction and characterization of palm (Borassus flabellifer) juice, Journal of Food Measurement and Characterization, 2018, 12(4), 2644–2656, DOI:10.1007/s11694-018-9882-5.
- M. C. Giannakourou and P. S. Taoukis, Effect of alternative preservation steps and storage on vitamin C stability in fruit and vegetable products: critical review and kinetic modelling approaches, Foods, 2021, 10(11), 2630, DOI:10.3390/foods10112630.
- G. Özkan and S. E. Bilek, Enzyme-assisted extraction of stabilized chlorophyll from spinach, Food Chem., 2015, 176, 152–157, DOI:10.1016/j.foodchem.2014.12.059.
- N. Koca, F. Karadeniz and H. S. Burdurlu, Effect of pH on chlorophyll degradation and colour loss in blanched green peas, Food Chem., 2007, 100(2), 609–615 CrossRef CAS.
- C. Mazzocchi, I. Benucci, C. Lombardelli and M. Esti, Enzyme-assisted extraction for the recovery of food-grade chlorophyll-based green colorant, Foods, 2023, 12(18), 3440, DOI:10.3390/foods12183440.
- D. Neagu, L. F. Leopold, P. Thonart, J. Destain and C. Socaciu, Enzyme-assisted extraction of carotenoids and phenolic derivatives from tomatoes, Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca, Anim. Sci. Biotechnol., 2014, 71(1) DOI:10.15835/buasvmcn-asb:71:1:10093.
- C. Pénicaud, N. Achir, C. Dhuique-Mayer, M. Dornier and P. Bohuon, Degradation of β-carotene during fruit and vegetable processing or storage: reaction mechanisms and kinetic aspects: a review, Fruits, 2011, 66(6), 417–440, DOI:10.1051/fruits/2011058.
- X. Li, F. Zhu and Z. Zeng, Effects of different extraction methods on antioxidant properties of blueberry anthocyanins, Open Chem., 2021, 19(1), 138–148, DOI:10.1515/chem-2020-0052.
- S. S. K. Dossou, F. Xu, J. You, R. Zhou, D. Li and L. Wang, Widely targeted metabolome profiling of different colored sesame (Sesamum indicum L.) seeds provides new insight into their antioxidant activities, Food Res. Int., 2022, 151, 110850, DOI:10.1016/j.foodres.2021.110850.
- M. Kouighat, A. Nabloussi, A. Adiba, M. E. Fechtali and H. Hanine, First study of improved nutritional properties and antioxidant activity in novel sesame mutant lines as compared to their wild-types, Plants, 2022, 11(9), 1099, DOI:10.3390/plants11091099.
- A. Łubek-Nguyen, W. Ziemichód and M. Olech, Application of enzyme-assisted extraction for the recovery of natural bioactive compounds for nutraceutical and pharmaceutical applications, Appl. Sci., 2022, 12(7), 3232, DOI:10.3390/app12073232.
- J. Zhang, S. Jia, Y. Liu, S. Wu and J. Ran, Optimization of enzyme-assisted extraction of the Lycium barbarum polysaccharides using response surface methodology, Carbohydr. Polym., 2011, 86(2), 1089–1092, DOI:10.1016/j.carbpol.2011.06.027.
- A. Al Loman and L. K. Ju, Enzyme-based processing of soybean carbohydrate: recent developments and future prospects, Enzyme Microb. Technol., 2017, 106, 35–47, DOI:10.1016/j.enzmictec.2017.06.013.
- T. Amza, A. Balla, F. Tounkara, L. Man and H. M. Zhou, Effect of hydrolysis time on nutritional, functional and antioxidant properties of protein hydrolysates prepared from gingerbread plum (Neocarya macrophylla) seeds, Int. Food Res. J., 2013, 20(5), 2081 Search PubMed.
- X. Guan and H. Yao, Optimization of Viscozyme L-assisted extraction of oat bran protein using response surface methodology, Food Chem., 2008, 106(1), 345–351, DOI:10.1016/j.foodchem.2007.05.041.
- A. Rosenthal, D. L. Pyle, K. Niranjan, S. Gilmour and L. Trinca, Combined effect of operational variables and enzyme activity on aqueous enzymatic extraction of oil and protein from soybean, Enzyme Microb. Technol., 2001, 28(6), 499–509, DOI:10.1016/S0141-0229(00)00351-3.
- C. B. Faulds, S. Collins, J. A. Robertson, J. Treimo, V. G. Eijsink and S. W. Hinz, et al., Protease-induced solubilisation of carbohydrates from brewers' spent grain, J. Cereal Sci., 2009, 50(3), 332–336, DOI:10.1016/j.jcs.2009.01.004.
- V. Arantes and J. N. Saddler, Cellulose accessibility limits the effectiveness of minimum cellulase loading on the efficient hydrolysis of pretreated lignocellulosic substrates, Biotechnol. Biofuels, 2011, 4(1), 3, DOI:10.1186/1754-6834-4-3.
| This journal is © The Royal Society of Chemistry 2026 |