Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Unravelling the impact of high-intensity ultrasound on the water mobility of meat products by LF-NMR
Davi
dos Santos e Santos
abc,
Yago Alves de Aguiar
Bernardo
 *abcde and
Carlos Adam
Conte-Junior
*abcde and
Carlos Adam
Conte-Junior
 *abcdfgh
*abcdfgh
aCenter for Food Analysis (NAL), Technological Development Support Laboratory (LADETEC), Federal University of Rio de Janeiro (UFRJ), Cidade Universitária, Rio de Janeiro, RJ 21941-598, Brazil. E-mail: yagoaabernardo@ufrrj.br; Tel: +55 21-98418-7126
bLaboratory of Advanced Analysis in Biochemistry and Molecular Biology (LAABBM), Department of Biochemistry, Federal University of Rio de Janeiro (UFRJ), Cidade Universitária, Rio de Janeiro, RJ 21941-909, Brazil. E-mail: conte@iq.ufrj.br; Tel: +55 21-3938-7824
cAnalytical and Molecular Laboratorial Center (CLAn), Institute of Chemistry (IQ), Federal University of Rio de Janeiro (UFRJ), Cidade Universitária, Rio de Janeiro, RJ 21941-909, Brazil
dDepartment of Food Technology, Federal Rural University of Rio de Janeiro (UFRRJ), Seropédica, RJ 23890-000, Brazil
eGraduate Program in Food Science (PPGCAL), Institute of Chemistry (IQ), Federal University of Rio de Janeiro (UFRJ), Cidade Universitária, Rio de Janeiro, RJ 21941-909, Brazil
fGraduate Program in Veterinary Hygiene (PPGHV), Faculty of Veterinary Medicine, Fluminense Federal University (UFF), Vital Brazil Filho, Niterói, RJ 24220-000, Brazil
gGraduate Program in Chemistry (PGQu), Institute of Chemistry (IQ), Federal University of Rio de Janeiro (UFRJ), Cidade Universitária, Rio de Janeiro, RJ 21941-909, Brazil
hGraduate Program in Biochemistry (PPGBq), Institute of Chemistry (IQ), Federal University of Rio de Janeiro (UFRJ), Cidade Universitária, Rio de Janeiro, RJ 21941-909, Brazil
First published on 21st January 2026
Abstract
High-intensity ultrasound (HIUS) is a green, cost-effective, and safe non-thermal technology that has been widely studied for the processing of meat and meat products, including brining, freezing, thawing, and cooking. It's based on acoustic waves applied through a liquid medium that generates cavitation bubbles, which implode, causing thermal, mechanical, and chemical damage to the food matrix. Given that the muscle is composed mainly of water, it's expected that HIUS can modify the water distribution in meat. This review aims to evaluate the effects of high-intensity ultrasound (HIUS) on water mobility in meat products using low-field NMR (LF-NMR) technology. To this end, articles that applied HIUS directly to the meat matrix and used LF-NMR technology to assess changes in water distribution and mobility were considered. Following a systematic search in the Scopus, Embase, and Web of Science databases, 57 articles were selected after screening, and 7 were added manually, totaling 64 articles. The results were divided based on the process evaluated in each article, which includes the application of HIUS. Thus, the following processes were evaluated: curing, freezing, thawing, heating, fermentation, bacterial inactivation, and quality improvement (HIUS only). The results demonstrated that the effects of HIUS on water retention depend mainly on operational parameters, such as time, power, and frequency, and can also be influenced by the nature of the matrix, the equipment, the process itself, and the application method. Furthermore, the possibilities of applying HIUS to meat products and their challenges were also discussed.
Sustainability spotlightThe water mobility and distribution in meat products is a good indicator of structural changes in these products. Low-field nuclear magnetic resonance (LF-NMR) is a cheap and fast technique often utilized for this. HIUS is an environmentally friendly, low-cost and safe non-thermal technology, which causes several physicochemical changes in meat tissue, which can affect the water mobility and its distribution in these products. The aim of this review is to understand the effects of high-intensity ultrasound (HIUS) on the water mobility of meat products. This study can contribute to the formation of a more environment-friendly food industry, as both techniques (LF-NMR and HIUS) are recognized as green technologies. This work is aligned with the Sustainable Development Goals 7 (affordable and clean energy) and 9 (industry, innovation and infrastructure). |
1 Introduction
The growing world population is driving the demand for high-quality meat.1,2 Meat is fundamental to human nutrition, as it is a rich source of energy and essential nutrients such as proteins, zinc, iron, and vitamin B12.1 However, its high nutritional value combined with its high water content makes the product highly perishable.3 Given this scenario, the meat industry has been using several traditional methods for food preservation, such as heating, freezing, and drying, to provide high-quality, safe, and durable products.3 On the other hand, these methods cause nutritional and sensory deterioration, do not ensure the complete inactivation of foodborne pathogens, are costly, energy-intensive, and have negative environmental impacts.4,5 Therefore, low-cost and environmentally friendly processes that ensure microbial safety, quality, and shelf-life, and maintain the nutritional and sensory attributes of meat and meat products are desirable.6 In this scenario, non-thermal preservation technologies (NTTs), such as ultraviolet type C (UV-C), gamma radiation, high hydrostatic pressure (HPP), alternative chemical compounds (ACC), and high-intensity ultrasound (HIUS), are effective alternatives for meat processing, capable of maintaining or improving the quality and shelf-life of meat and meat products.7 Among these methods, HIUS has attracted attention for its results on meat tenderization and for the possibilities of application in different meat processes, such as curing, freezing, and thawing.8,9Ultrasound is an environmentally friendly, economical, and safe NTT for food processing based on acoustic waves greater than 20 kHz. Ultrasound can be classified as low-intensity (<100 kHz; < 1 W cm−2) and high-intensity (20–500 kHz; >1 W cm−2) ultrasound, and can be applied directly using a probe or by immersing the product in an ultrasonic bath.10,11 Whereas the bath system is more cost-effective, as it can process several samples per batch, the probe system is more potent due to its smaller application area and direct contact with the sample, in addition to having lower acoustic energy losses, as it directly contacts the sample.11 Furthermore, the propagation of the ultrasonic waves is irregular in the bath system due to their reflection in the bath walls. In contrast, in the probe system, the release of the metal that constitutes the probe can occur, contaminating the treated sample.12 Another important aspect of the HIUS application is the application mode, which can be continuous or pulsed. The first one, although it is better in terms of microbiological inactivation, causes an increase in temperature.12 The HIUS, when it is applied in a liquid medium, causes the cavitation phenomenon, generating bubbles that expand and contract continuously until they reach the critical volume and implode. The implosion of the cavitation bubbles causes the formation of micro-jets, free radicals, reactive oxygen species (ROS), and hot spots.7 The increase in pressure and temperature due to the implosion of the cavitation bubbles breaks down the water molecules, generating H+ and OH−, thus promoting chemical reactions and modifying other molecules. Furthermore, the bubbles attract each other, generating microstreaming that can improve the diffusion of reactants, enhance mass and heat transfer, and cause damage to solid surfaces.13 Thus, HIUS processing can cause significant structural changes in meat proteins, affecting important quality parameters, including water holding capacity (WHC), water distribution within the meat structure, juiciness, tenderness, and general appearance.5
Therefore, HIUS can cause changes on both microscopic and macroscopic scales when applied to biological tissues and may have positive effects on meat processing, such as accelerating and improving curing, drying, and tenderization.14,15 However, it is worth noting that the effects of HIUS application on the physicochemical properties of the meat products are often contradictory, depending on the intensity, time, frequency, application system, and the matrix itself.5,14 Some of the adverse effects of the cavitation induced by the HIUS include thermal damage and macromolecule depolymerization, even in the absence of bubble implosion, in addition to the oxidative stress resulting from the free radicals (ROS) and the local overheating, thus impairing the product quality.14 From this, it can be seen that the effects of the HIUS application on the physicochemical properties of meat and meat products are, to some extent, difficult to control, which is a significant challenge for this technology.
Given that muscle tissue is composed mostly of water, primarily within the myofibrils, the structural changes induced by HIUS in the muscle structure result in alterations in water mobility and distribution.16 In fact, as reviewed by Kang et al.,5 the HIUS application is capable of improving the gel-forming ability and the solubility of the proteins, and, consequently, the WHC of the meat, when operational parameters such as intensity, frequency, and time are correctly chosen. Hence, the changes in the physicochemical properties of the meat induced by the ultrasound application were the premise of the work by Stadnik et al.,17 who evaluated the effects of HIUS application on beef during ageing by evaluating its WHC, water mobility, and distribution. Additionally, considering the complex structure of the muscle, which is composed mainly of proteins and connective tissue, water, a small molecule, can accumulate at various levels within it. Therefore, evaluating the mobility and distribution of the water in meat can provide important information about its structure.
The Low-Field Nuclear Magnetic Resonance (LF-NMR) technology is an effective tool for this purpose. The LF-NMR technology is a cost-effective, rapid, and non-invasive technique widely used for determining the distribution and mobility of water in foods.18,19 It is based on the absorption of resonance radiofrequency pulses by protons aligned with an external magnetic field, B0.20 The absorption of this energy causes the protons to rotate in a plane perpendicular to B0. When the pulses cease, relaxation occurs, involving two phenomena. (I) The protons return to rotate in their original direction, aligned with B0. This is the longitudinal (or spin–lattice) relaxation, and its characteristic constant time is T1. This is the result of the energy change between the protons and their surroundings (lattice). (II) Randomization (or the loss of phase coherence) of the protons occurs in the plane perpendicular to B0. This is the transversal (or spin–spin) relaxation, and is governed by the constant time T2. The proton interactions cause differences in local magnetic fields, leading to different T2 values.21 According to the diffusive and chemical exchange model, the water molecules diffuse to the biopolymer surface and exchange with the biopolymer protons, thus providing information about the morphological state of the biopolymers.21 Therefore, under this model, the proton populations can be classified based on their transversal relaxation time, T2, as follows. (i) T2b, with the lowest relaxation time and the lowest relative population, P2b, assigned to protein protons and water closely associated with proteins. This proton population is commonly recognized as “bound water”.21 (ii) T21, with intermediate relaxation time and the highest relative population, P21, relative to water entrapped by intermolecular interactions, such as hydrogen bond and steric effects, and related to sarcomere lengths, juiciness, and taste. This proton population, in meat products is often called “immobilized water”.22,23 (iii) T22, with the highest relaxation time, and relative population P22, assigned to water weakly associated with the matrix, between fiber bundles, in the sarcoplasmic area which can be easily lost and utilized by microorganisms. This water population is often recognized as “free water”.24,25 Therefore, LF-NMR can provide several information about the meat morphology. However, this technology has some limitations, including low sensitivity and data that are difficult to process and interpret. Besides this, the algorithm used to process the data and the sample processing can affect the results, and there are still controversies about the relationship between water mobility and its distribution in meat and meat products.16,20,21 Furthermore, the T2 relaxation patterns can vary depending on several factors such as the species, muscle type, slaughter procedure, pH, and temperature.16
Thus, this systematic review aims to discuss the changes induced by HIUS on water mobility and distribution in meat and meat products, as well as the main mechanisms described in the literature. To this end, the effects of HIUS applied alone and in combination with other processes are evaluated, as well as its synergistic effects on water mobility using LF-NMR technology. Furthermore, the relationships between water mobility and other quality parameters, as well as the challenges and gaps in the literature regarding the HIUS application, are also discussed.
2 Materials and methods
2.1 Systematic search
The systematic search was completed in June 2024 in the following databases: Web of Science, Scopus, and Embase. The search string was obtained through the PICO (Population, Intervention, Comparison, and Outcomes) methodology. The search aimed to answer the following question: what is the effect of ultrasound on water mobility in meat products? For this, all studies that applied the HIUS directly to meat products (in natura or derivatives), alone or combined with other preservation technologies, were considered.Thus, the search terms were: “pork”, “meat”, “chicken”, “fish”, “beef” (population), “ultrasound” (intervention), “moisture”, “water”, “distribution”, “compartmentalization”, “migration”, and “mobility” (context). The final search string was obtained by the combination of these blocks using the appropriate Boolean operators: [(“moisture” OR “water”) AND (“distribution” OR “compartmentalization” OR “migration” OR “mobility”) AND (“pork” OR “meat” OR “chicken” OR “fish” OR “beef”) AND “ultrasound”].
2.2 Eligibility criteria
The articles included in this review were selected following the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) methodology.26 The removal of duplicated articles and the further screening processes were carried out using the StArt© (State of the Art through Systematic Review) software. Only articles written in English were included in the review, with no restriction on the publication date. The inclusion and exclusion criteria are listed in Table 1.| Inclusion criteria | Exclusion criteria |
|---|---|
| a HIUS: high-intensity ultrasound, LF-NMR: low-field nuclear magnetic resonance. | |
| Use of LF-NMR as an approach to evaluate water mobility | Review articles; meta-analysis; book chapters; annals; conference papers, abstracts, theses and dissertations |
| Application of HIUS as processing technology | |
| Meat and meat products as population | Articles not written in English |
2.3 Risk of bias
This work may have a potential source of bias, including language, the chosen databases, inclusion and exclusion criteria, the period of the systematic search, and the impact of missing data.3 Results and discussion
3.1 Search results
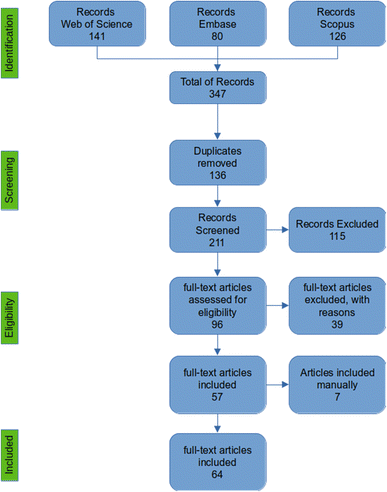
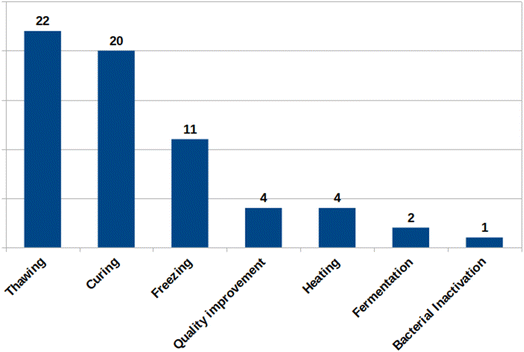
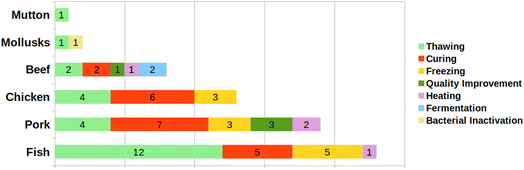
The search returned 347 articles (Web of Science: 141; Embase: 80; and Scopus: 126), of which 136 were duplicates. From the remaining 211 articles, 115 were excluded after reading titles and abstracts, and 39 were excluded after full reading because they did not fit with the eligibility criteria, totaling 57 articles included in the review (Fig. 1). Furthermore, seven papers were added manually. The reviewed articles are organized in Table 2 according to the application of HIUS in each work: curing (20), freezing (11), thawing (22), quality improvement (4), bacterial inactivation (1), fermentation (2), and heating (4) (Fig. 2). The following matrices were analyzed: fish (23), pork (19), chicken (13), beef (8), mollusks (2), and mutton (1). The meat matrices by treatment are presented in Fig. 3. The findings were expressed based on the LF-NMR results of each article, and the fundamental aspects of the processes were compiled, considering aspects such as the equipment (probe or bath), the variables assessed, and the groups studied, using the same labels as the authors.| Processing | Matrix/product | Parameters US (W; W mL−1; W cm−2; W L−1; Hz; kHz; min; h) | Process | Findings | References |
|---|---|---|---|---|---|
| a CAR-UT: HIUS treatment and 1% salt and 0.3% carrageenan; CCCP: collagen peptide solution from chicken cartilage; SR: salt replacer; CUD-UT: HIUS treatment and 1% salt and 0.3% curdlan; EUT: SAEW thawing; EVO: extra virgin olive; FFUT: HIUS thawing with fixed frequency at 28 kHz; GUA: HIUS in 1% chitosan-grafted-chlorogenic acid solution; HIUS: high-intensity ultrasound; MUT: microwave combined with HIUS thawing; PA: potassium alginate; P2b: bound water population; P21: immobilized water population; P22: free water population; SAEW: slightly acidic electrolyzed water; SEU: HIUS-assisted brine in different frequency modes (mono-, dual- and tri-frequency) in sequential mode; SFUT: HIUS-assisted thawing with a sweep frequency of 28 ± 2 kHz; SIU: HIUS-assisted brine in different frequency modes (mono-, dual- and tri-frequency) in simultaneous mode; T2b: transversal relaxation time constant of bound water; T21: transversal relaxation time constant of immobilized water; T22: transversal relaxation time constant of free water; UA: HIUS in distilled water; UAF: HIUS-assisted freezing; UCP: HIUS treatment and later immersion of the sample in collagen peptide solution from chicken cartilage (CCCP); UDC: HIUS-assisted dry curing; UDW: HIUS treatment and immersion of the sample in deionized water for 55 min, 4 °C; UET: HIUS-assisted enzyme; UF: HIUS-assisted immersion freezing; UIF: HIUS-assisted immersion freezing; UIT: ultrasonics combined far infrared thawing/HIUS-assisted thawing; UMH: HIUS pretreatment and further marination in L-histidine (1.5 g L−1) solution for 55 min; UMT: HIUS combined with microwave thawing; US: HIUS treatment; US-BP: HIUS treatment combined with inoculation of P. acidilactici BP2; USC: HIUS combined with sodium bicarbonate-assisted curing; UST: HIUS-assisted saline thawing; USV: HIUS-assisted sous-vide cooking; UT: HIUS thawing/HIUS-assisted thawing/HIUS-assisted immersion thawing/HIUS treatment and 1% salt (NaCl)/HIUS-treated samples; UVT: HIUS-assisted vacuum thawing; UWT: HIUS water thawing; W: water; WUT: HIUS-assisted water thawing. | |||||
| Curing | Reduced-salt pork batter (Mesoglutaeus) | 40 kHz; 192 W; 0–60 min | • 25 g NaCl, 0 g NaHCO3; 0 min | • Increase in the content of P21 and decrease of P22 on addition of sodium bicarbonate and increase of HIUS time | Kang et al.25 |
| • 20 g NaCl and 2 g NaHCO3; 0 min | |||||
| • 20 g NaCl and 2 g NaHCO3; 30 min | |||||
| • 20 g NaCl and 2 g NaHCO3; 60 min | • T21 and T22 values decreased with addition of sodium bicarbonate and increased with increase of HIUS time | ||||
| • The prepared batter was vacuum packaged and then sonicated using an ultrasonic machine | |||||
| Chicken meat breast batter (Pectoralis major) | 40 kHz; 300 W; 10, 20, 30 and 40 min | • Meat batters with varying salt content % (w/w): 1.0%, 1.5% and 2.0% | • The 20 and 40 min treatments did not change T2 constant times significantly, for any salt level | Li et al.87 | |
| • Ultrasonic bath | • The samples treated with HIUS for 20 min had a higher proportion of immobilized water and lower proportion of free water | ||||
| Pork (M. semitendinosus) | 20 kHz; 9.0 and 54.9 W cm−2; 120 min | • The HIUS treatment was performed using an ultrasonic probe in 1000 mL of 50 g L−1 brine solution (NaCl or salt replacer(SR)) | • Decrease in T21 and increase in T22 values after 120 min of brine for NaCl and SR | Ojha et al.34 | |
| • Decrease of P21 with the increase of ultrasonic power | |||||
| • Lower water retention than control | |||||
| • Greater water exudation with increasing ultrasonic intensity | |||||
| Beef (longissimus dorsi) | 20 kHz; 150, 200, 250 and 300 W; 30 and 120 min | • HIUS-assisted brine | • The 150 and 200 W treatments did not significantly affect T21 values | Kang et al.30 | |
| • Ultrasonic probe | • The treatments of 250 and 300 W led to an increase in T21 values | ||||
| • Treatments lasting more than 120 min showed higher T21 values | |||||
| • Brine (w/v): sucrose (1.5%), sodium tripolyphosphate (0.16%), sodium pyrophosphate (0.16%) and sodium hexametaphosphate (0.8%) | • P21 increased with increase of power and treatment time | ||||
| Chicken breast (pectoralis major) | 20 kHz; 200 W; 15.6 W cm−2; 5 min | • Ultrasonic probe (12 mm diameter) | • The HIUS treatment improved water retention in treated samples (UDW and UCP) | Zou et al.68 | |
| • UDW: HIUS treatment and immersion of the sample in deionized water for 55 min, 4 °C | |||||
| • UCP: HIUS treatment and later immersion of the sample in collagen peptide solution from chicken cartilage (CCCP) for 55 min, 4 °C | |||||
| • 40 mm × 30 mm × 10 mm pieces weighing about 20 ± 1 g | • UCP samples had the highest P21 values for the same storage time | ||||
| Chicken breast (pectoralis major) | 20 kHz; 300 W; 50 min (5 s on/5 s off) | • USC: HIUS combined with sodium bicarbonate-assisted curing (solution with 6% salt and 2% sodium bicarbonate) | • Increase in time constants of bound water | Xiong et al.29 | |
| • (5 cm × 4 cm × 3 cm), 50 g ± 2 g pieces | |||||
| • 6-mm ultrasonic probe (2 cm away from the meat) | • Decrease of T21 and T22 values | ||||
| Pork (m. longissimus thoracis et lumborum) | 20 kHz; 4.2, 11.0 and 19.0 W cm−2; 10, 25 and 40 min | • Cylindrical samples (35ϕ × 4.2 mm), weighing 4.2 ± 0.5 g | • The treatment does not affect the bound and free water populations significantly | McDonnell et al.31 | |
| • Ultrasonic probe | |||||
| • 65 mL brine solution (5.7% (w/w) NaCl) | • The 19 W cm−2 treatments increased T21 values | ||||
| Chicken breast (pectoralis major) | 40 kHz; 300 W; 10, 20, 40 and 80 min | • HIUS treatment (UT) (80 min, only) | • UT and UET treatments shortening T2b values | Cao et al.32 | |
| • HIUS-assisted enzyme (UET-10, 20, 40 and 80 min) | |||||
| • UET samples were injected with 1/10 of their weights with papain 45 U g−1 | • UT and UET-10, 20 and 40 samples showed increase of P21 and decrease of P22 values | ||||
| • Ultrasonic bath at 30 °C | • UET treatment decreased T22 values compared to enzyme treatments without HIUS | ||||
| • 30 mm × 30 mm × 20 mm pieces weighing 9.00 ± 0.50 g | • HIUS treatments less than 20 min longer showed positive effects on WHC | ||||
| Surimi (Nemipterus virgatus) | 40 kHz; 150 W; 20 min | • The HIUS was applied with and without curdlan gum 0.4% (w/w) | • HIUS and curdlan alone and HIUS-combined curdlan showed higher immobilized content and the lowest free water content | Zhang et al.88 | |
| Chicken meat paste (pectoralis major) | 20 kHz; 300 W; 1 min, 3 times (2 s on/4 s off) | • Sonicated samples: UT: 1% salt (NaCl); CAR-UT: 1% salt and 0.3% carrageenan; CUD-UT: 1% salt and 0.3% curdlan | • HIUS-treated samples showed decreased P2b, without significant difference for P21 and P22 compared to control | Zhao et al.89 | |
| • CAR-UT showed lower P2b than the CAR group | |||||
| • Ultrasonic probe | • The CUD-UT group showed higher T21 and T22 values compared to the CUD group | ||||
| Chicken breast (pectoralis major) | 40 kHz; 300 W; 60 min; 7.84 W cm−2 | • The samples were marinated with 2% complex phosphate at 4 °C for 30 min and then sonicated | • Decrease of T2b | Tong et al.90 | |
| • Increase of T21 and P21 | |||||
| • T22 and P22 disappeared compared to deionized water curing | |||||
| Sea bass (Lateolabrax japonicus) | 20 kHz; 600 W; 10 min | • HIUS in distilled water (UA); HIUS in 1% chitosan-grafted-chlorogenic acid solution (GUA) | • GUA samples had higher P21 and lower P22 values | Yang et al.91 | |
| Silver carp surimi (Hypophthalmichthys molitrix) | 120, 150, 180, 210 and 240 W; 10, 15, 20 and 30 min | • 150 g samples sealed in a 160 × 240 × 0.2 mm polyethylene bag | • Low-salt (0–0.5%) samples treated by HIUS, had lower P22 and higher P21 values than control | Gao et al.92 | |
| • Ultrasonic bath varying salt contents | • HIUS treatment decreased T22 values in any salt level | ||||
| Beef (m. semitendinosus) | 20 kHz; 300 W; 15.6 W cm−2; 5 min | • UDW: HIUS pretreatment and further marination in deionized water for 55 min | • HIUS-treated samples had lower T21 values | Shi et al.93 | |
| • UMH: HIUS pretreatment and further marination in L-histidine (1.5 g L−1) solution for 55 min | |||||
| • 50 mm × 20 mm × 10 mm samples, weighing about 30 ± 1 g | • UDW and UMH samples had higher P21 and P22 than control | ||||
| • Ultrasonic probe | |||||
| Pork loin (longissimus) | 25 kHz; 320 W; 30 min; 10 s on/5 s off | • HIUS combined with glycerol-mediated curing in different concentrations | • Decrease of T21 and T22 values | Gu et al.94 | |
| • 50 × 40 × 10 mm3 pieces (50 ± 1 g) | |||||
| • Ultrasonic probe | • Conversion of immobilized water into bound water and decrease of P22 | ||||
| Snakehead fish (Channa argus) | 40 kHz; 216 W; 10 min | • HIUS-assisted impregnation with a cryoprotectant solution | • Lower moisture loss | Zheng et al.95 | |
| • Ultrasonic bath | |||||
| Reduced-sodium bacon (pork belly) | 20 kHz; 300 and 800 W; 30 and 60 min; 3 s on/3 s off | • HIUS-assisted dry curing (UDC) | • The (300 W/60 min) and (800 W/30 min) treatments increased T21 values | Pan et al.96 | |
| • 120 × 90 × 30 mm samples weighing 200 ± 20 g | |||||
| • Ultrasonic probe | |||||
| Pork (longissimus dorsi) | 20, 40, and 60 kHz; 20/40 kHz, 20/60 | • HIUS-assisted brine in different frequency modes (mono-, dual-) | • Samples treated by HIUS had higher T2b and T21 values, but lower T22 than control samples | Guo et al.97 | |
| kHz, and 40/60 kHz; | And tri-frequency) in sequential (SEU) and simultaneous modes (SIU) | ||||
| 20 + 40 kHz, 20 + 60 kHz, and 40 + 60 kHz; 50 W L−1. 20 + 40 kHz, 20 + 60 kHz, 40 + 60 kHz, and 20 + 40 + 60 kHz; 100 W L−1 | • Ultrasonic bath | ||||
| Yellowfin tuna (Thunnus albacares) | 20 kHz; 540 W; 30 min | • HIUS-assisted salting | • Samples treated with HIUS showed better water retention than the control group | He et al.98 | |
| • 3 × 3 × 1 cm3 samples weighing 11 ± 1 g | |||||
| • Ultrasonic bath | |||||
| Pork (longissimus dorsi) | 20 + 40 kHz; 400 W; 5 min | • Dual-frequency HIUS combined with L-lysine treatment | • Higher P2b and P21 values, and lower P22 values | Xu et al.99 | |
| • 10 cm × 4 cm × 2 cm samples weighing 50 g ± 2 g | |||||
| Freezing | Prepared ground pork (boneless pork leg) | 20 kHz; 80 W L−1; 10 s on/15 s off | • HIUS-assisted immersion freezing (UIF) | • The samples treated with HIUS showed the smallest changes in the immobilized water population and the lowest P22 values after 90 days of storage among treated samples | Wu et al.100 |
| • The sample was treated using an ultrasonic probe in a constant temperature tank, filled with a mixture of 95% ethanol/5% fluoride | • T21 values of UIF samples had minor changes | ||||
| • The treatment was interrupted when the center temperature reached −18 °C | • The T2b values showed lower increase between the days 30 and 90 of storage. No significant changes for P2b values | ||||
| Chicken breast (pectoralis major) | 30 kHz; 125, 165, 205, and 245 W; 8 min (30 s on/30 s off) | • HIUS-assisted immersion freezing (UF) | • There were no significant differences in T2b and P2b values | Zhang et al.41 | |
| • Ultrasonic bath | |||||
| • 7-cm-long pieces with 110 ± 6 g | • UF-165 treatment had lower water mobility and T21 values | ||||
| • Temperature kept at −25 ± 0.5 °C, using 95% ethanol/5% fluoride (v/v) as the coolant with a continuous flow of 1.5 L min−1 | |||||
| • The HIUS was applied when the central temperature of samples reached 0 °C | • UF-245 treatment led to a decrease in P21 and increase in P22 values | ||||
| Common carp (Cyprinus carpio) | 30 kHz; 175 W; 9 min (30 s on/30 s off) | • 5-cm-long pieces with 210 ± 15 g | • Lower T2b,T21 and T22 values, during storage than air freezing and immersion freezing methods | Sun et al.101 | |
| • HIUS-assisted immersion freezing (UIF) | |||||
| • Temperature: −25 ± 0.5 °C, 95% ethanol/5% fluoride as the coolant | |||||
| • The treatment was performed until the core temperature reached −18 °C | |||||
| • −18 ± 1 °C for 180 days storage after treatments | |||||
| Porcine (longissimus lumborum) | 30 kHz; 120, 180, 240, and 300 W; 8 min (30 s on/30 s off) | • HIUS-assisted immersion freezing (UIF) | • The UIF-180 treatment had lower T21 and T22 values | Zhang et al.40 | |
| • Ultrasonic bath | • UIF-180 showed lower water mobility | ||||
| • 95% ethanol/5% fluoride coolant with continuous flows of 1.5 L min−1 | |||||
| • 30-mm-thick slices weighed 120 ± 2 g | • The UIF-300 samples had high T21 values | ||||
| Porcine (longissimus lumborum) | 30 kHz; 180 W; 8 min (30 s on/30 s off) | • HIUS-assisted immersion freezing (UIF) performed as described by Zhang et al.40 | • Lowest T2b values after day 30 | Zhang et al.102 | |
| • Low water mobility | |||||
| • Lower T21 values | |||||
| • 3-cm-thick chops (120 ± 2 g) | • Higher P21 values | ||||
| • The samples were stored at −18 ± 1 °C for 0, 30, 60, 90, 120, 150, and 180 days after treatments | • Minor changes in free water | ||||
| Common carp (Cyprinus carpio) | 30 kHz; 125, 150, 175, 200, and 225 W; 30 s on/30 s off; 9 min | • HIUS-assisted immersion freezing (UIF) | • The UIF-175 samples had the lowest T21 value | Sun et al.103 | |
| • 95% ethanol as the coolant | |||||
| • 5-cm chops (210 ± 15 g) | |||||
| • When the central temperature of samples reached approximately −18 °C, they were stored at −18 ± 1 °C for 24 h | • T21 value decreased at low ultrasonic power and increased at higher power levels | ||||
| • Ultrasonic bath | |||||
| Chicken breast (pectoralis major) | 30 kHz; 125, 165, 205 and 245 W; 8 min | • UF: HIUS-assisted immersion freezing | • UF-165 group had lower T21 than IF group | Zhang et al.104 | |
| • 7 cm shops weighing about 110 ± 6 g | |||||
| • HIUS treatment was started when the center temperature of the samples reached 0 °C | |||||
| • The process was stopped when the center temperature of the samples reached −18 ± 0.5 °C | • UF-165-treated samples had higher P21 and lower P22 than IF-treated samples | ||||
| • Ultrasonic bath | |||||
| Yellow croaker (Pseudosciaena crocea) | 25 kHz; 200 W; 30 s on/45 s off | • HIUS-assisted freezing (UIF) | • The UIF process reduced the loss of immobilized water and the increase of free water | Li et al.73 | |
| • The process was stopped when the center temperature of samples reached −18 °C | |||||
| • After that, the samples were subjected to 5 freeze–thaw cycles | |||||
| • Ultrasonic bath | |||||
| Yellow croaker (Pseudosciaena crocea) | 28 kHz; 160 W, 175 W and 190 W; 0.800 W L−1, 0.875 W L−1 and 0.950 W L−1 | • The samples were subjected to vacuum immersion with water (W) and potassium alginate solution (PA) and then to HIUS-assisted freezing (UAF) | • PA-UAF-175 treatment showed the highest T21 and P21 values | Cheng et al.105 | |
| • The process was stopped when the central temperature of the samples reached −18 °C | |||||
| • 10 cm × 5 cm × 1 cm samples | • Samples treated at 175 W showed better moisture results, while treatments at 190 W had higher free water contents | ||||
| • Ultrasonic bath | |||||
| Chicken breast (pectoralis major) | 165 W; 8 min; 30 s on 30 s off | • HIUS-assisted immersion freezing (UF) | • Small increase of T2 values | Zhang et al.106 | |
| • 7.0 ± 0.2-cm samples weighing 104 ± 5 g | |||||
| • The process was stopped when the temperature of the sample geometric centre was −18 ± 0.5 °C | • P21 values higher than air freezing and immersion freezing processes | ||||
| • Ultrasonic bath | |||||
| Common carp (Cyprinus carpio) | 30 kHz; 125, 150, 175, 200 and 225 W; 9 min; 30 s on/30 s off | • HIUS-assisted freezing at different power levels (UF) | • T21 reached the minimum values at 175 W and the maximum at 225 W among HIUS-treated samples | Sun et al.107 | |
| • 5-cm samples of 210 ± 15 g | |||||
| • Ultrasonic bath | • 175 W had the lowest increase of T22 | ||||
| Thawing | Mirror carp (Cyprinus carpio L.) | 200 W | • HIUS-assisted saline thawing (UST) and HIUS thawing (UT) | • The samples with 0.10% salt content showed the smallest increase in T21 value | Li et al.42 |
| • The UST treatment was carried out using varying salt contents (0.05%, 0.10% and 0.20%) | |||||
| • Treatments with HIUS until the center temperature of the sample reached 4 °C | |||||
| Common carp (Cyprinus carpio) | 30 kHz; 100, 300, and 500 W | • HIUS-assisted thawing (UT) | • The UT-300 group had the lowest T21 values among thawed samples, while the UT-500 group had higher T21 values | Sun et al.43 | |
| • The samples were frozen until the core temperature reached −18 °C | |||||
| • 5-cm-long pieces and 4-cm-thick | |||||
| • The treatment was stopped when the core temperature of the sample reached 4 °C | • UT-300 showed the lowest increase in T22 values | ||||
| • Ultrasonic bath with distilled water (20 ± 0.5 °C) | |||||
| Red seabream (Pagrus major) | 40 kHz; 200 W | • HIUS-assisted thawing (UT) and HIUS-assisted vacuum thawing (UVT) | • The UT and UVT treatments showed lower conversion of immobilized water to free water and higher immobilized water content | Cai et al.108 | |
| • Ultrasonic bath | |||||
| • Temperature: 10 °C | |||||
| • 8 cm long × 4 cm wide pieces, weighed approximately 100 g | |||||
| Prepared ground pork | 20 kHz; 20 s on/25 s; 90, 120 and 150 W L−1 | • HIUS-assisted thawing (UIT) | • The UIT-120 samples had the lowest T21 and T22 values; and the highest P21 value and the lowest P22 value | Wu et al.109 | |
| • Ultrasonic probe | |||||
| • The process was stopped when the center temperature reached 4 °C | • The UIT-150 samples showed increased T21 and T22 values compared to other HIUS-treated samples | ||||
| • The samples were thawed after 30 days of storage | |||||
| Frozen beef (sirloin) | 22, 33 and 40 kHz; 22/33, 22/40, and 33/40 kHz; 22/33/40 kHz; 100 W L−1 | • The samples were thawed using HIUS in different frequency modes (mono-, di- and tri-) | • The samples treated with 22 and 22/33 kHz modes had lower T21 than control | Wu et al.110 | |
| • 10 × 10 × 1 cm (about 150 g) | |||||
| • Ultrasonic bath | • The 22 kHz treatment had higher P2b, P21 and P22 than control | ||||
| Chicken breast (pectoralis major) | 40 kHz; 200 W | • HIUS-assisted thawing with water (WUT) | • P22 of the EUT group was not significantly different from the control | Kong et al.111 | |
| • Slightly acidic electrolyzed water (SAEW) thawing (EUT) | |||||
| • 3 × 3 × 3 cm3 (64 ± 5 g) samples | |||||
| • The process was stopped when the center temperature reached 0 °C | |||||
| • Ultrasonic bath | |||||
| Cuttlefish | 53 kHz; 200 W | • HIUS water thawing (UWT) | • UWT samples had lower T21 and T22 values than control | Lv & Xie74 | |
| • Samples weighing about 1100 ± 50 g | |||||
| • The process was stopped when the center temperature of samples reached 4 °C | |||||
| Chicken breast (pectoralis major) | 40 kHz; 200 W | • WUT: HIUS-assisted water thawing | • T21 values for EUT-treated samples was not significantly different from the control group | Kong et al.112 | |
| • EUT: HIUS-assisted SAEW thawing | |||||
| • 3 cm × 3 cm × 3 cm samples weighing about 64 ± 5 g | • P21 values of EUT and WUT groups was not significantly different from the control group | ||||
| • Ultrasonic bath | |||||
| Red drum (Sciaenops ocellatus) | 40 kHz; 400 W | • HIUS-assisted thawing (UT) | • UT, UMT and UIT samples had higher P21 values than fresh samples, and UMT was the highest among them | Cai et al.113 | |
| • HIUS combined with microwave thawing (UMT) | |||||
| • Ultrasonics combined far infrared thawing (UIT) | • UT samples had highest T2 values and greater conversion of bound water into immobilized water | ||||
| • 30 g samples | |||||
| • Ultrasonic bath | |||||
| Yellow croaker (Pseudosciaena crocea) | 28 kHz, 40 kHz and 28/40 kHz; 200 W | • HIUS-assisted thawing in different frequency modes (mono and dual) | • The samples treated with dual-frequency mode treatment had the highest T2b and T21 values and WHC | Cheng et al.114 | |
| • The process was stopped when the center temperature of samples reached 4 °C | |||||
| • Ultrasonic bath | |||||
| Pompano (Trachinotus ovatus) | 200 W; 33 min | • HIUS-assisted thawing | • Free water content was not significantly affected | Lan et al.115 | |
| • The process was stopped when the temperature of samples reached 0 °C | |||||
| • Ultrasonic bath | |||||
| Largemouth bass (Micropterus salmoides) | 40 kHz; 200 W | • Vacuum combined with HIUS thawing | • Samples thawed with HIUS had T2b and T21 values higher than fresh samples | Cai et al.116 | |
| • The process was stopped when the temperature of samples reached 0 °C | • Decrease of T22 | ||||
| • 3 × 3 × 2 cm3 samples weighing about 20 g | |||||
| • Ultrasonic bath | • Decrease of P21 and increase of P22 | ||||
| Chicken breast (pectoralis major) | 30 kHz; 200, 300, 400, and 500 W; 30 s on/30 s off | • HIUS-assisted immersion thawing (UT) | • UT-300 samples had the lowest T2b, T21 and T22 values | Zhang et al.44 | |
| • 7-cm long chops weighing 106 ± 3 g | • UT-300 samples had the highest P2b and P21 values and the lowest P22 values | ||||
| • The process was stopped when the geometric center temperature of the samples reached 4 ± 0.5 °C | • Among the HIUS-treated samples, UT-500 group had the highest T2b, T21 and T22 values | ||||
| • Ultrasonic bath | • UT-500 treatment decreased P21 and increased P22 values | ||||
| Large yellow croaker (Pseudosciaena crocea) | 40 kHz; 200, 240, 280, and 320 W | • HIUS-assisted thawing | • T2b values decreased at 240 W and increased at 320 W | Chu et al.117 | |
| • The process was stopped at 0 °C | • The 240 and 280 W groups had the lowest P22 values among treated samples | ||||
| • Ultrasonic bath | • 320 W group had the highest P22 value | ||||
| Tuna (Thunnini) | 40 kHz; 11.63 W cm−2; (280 W; 6, 12, 18, and 24 min); (160, 280, and 400 W; 12 min) | • HIUS-assisted thawing | • Increase of T2b compared to the control group | Ma et al.118 | |
| • T21 and T22 values increased with treatment time | |||||
| • T21 increased with power levels | |||||
| • Ultrasonic bath | • The 280 W; 12 min treatment had the highest T2b and P21 values | ||||
| • P21 decreased at 400 W | |||||
| Small yellow croaker (Larimichthys polyactis) | 26, 28 and 30 kHz; 60 W L−1 | • HIUS thawing with a fixed frequency at 28 kHz (FFUT) | • T2b values of the FFUT group increased, but was not significantly changed in the SFUT group, when compared to the control group | Wang et al.119 | |
| • HIUS-assisted thawing with a sweep frequency of 28 ± 2 kHz (SFUT) | |||||
| • 16 ± 0.5 cm samples weighing 30 ± 0.5 g | • SFUT samples showed higher P2b and P21 and lower P22 values than the FFUT group | ||||
| • Ultrasonic bath | |||||
| Large yellow croaker (Larimichthys crocea) | 20 kHz; 20/28 kHz; 20/28/40 kHz; 0.9 W L−1 | • HIUS-assisted thawing in different frequency modes (mono-, di- and tri-) | • P2b, P21 and P22 values of samples treated with multiple frequencies were not significantly different from fresh samples | Bian et al.120 | |
| • 30 ± 5 cm samples weighing 500 ± 20 g | |||||
| • The process was stopped when the central temperature of samples reached 4 ± 1 °C | • For the mono-frequency group, P2b and P21 values were lower than those of fresh samples, but P22 values were higher | ||||
| • Ultrasonic bath | |||||
| Chicken breasts (pectoralis major) | 30 kHz; 200, 300, 400, and 500 W; 30 s on/30 s off | • HIUS-assisted thawing at different power levels | • Samples treated at 300 W had the lowest T21 and the highest P21 values | Zhang et al.45 | |
| • 7-cm-long chops (106 ± 3 g) | |||||
| • The process was stopped when the central temperature of the samples reached 4.0 ± 0.5 °C | • 500 W treatments increased T21 and decreased P21 values | ||||
| • Ultrasonic bath | |||||
| Pork (longissimus lumborum) | 500 W; 20 min | • HIUS-assisted thawing | • Increase of T21, T22 and P22, and decrease of P21 compared to fresh samples | Wang et al.121 | |
| • Samples weighing 100 ± 0.1 g | |||||
| • Ultrasonic bath | |||||
| Yellowtail (Seriola quinqueradiata) | 40 kHz; 200 W | • HIUS-assisted thawing (UT) and microwave combined with HIUS thawing (MUT) | • Decrease of P22 in samples treated with HIUS | Shen et al.72 | |
| • 250 ± 30 g samples | |||||
| • The process was stopped when the central temperature reached 0 °C | • MUT samples had the best water retention | ||||
| • Ultrasonic bath | |||||
| Pork (longissimus dorsi) | 500 W; 45 min | • HIUS-assisted thawing | • Lower T21 values | Wang et al.122 | |
| • 100 ± 0.1 g samples | |||||
| • Ultrasonic bath | |||||
| Pork, beef and mutton (tenderloin) | 45 kHz; 300 W; 9–10 min | • HIUS-assisted thawing | • Highest values of P2b and P21 | Gan et al.123 | |
| • 3 × 3 × 3 cm samples | |||||
| • Ultrasonic bath | |||||
| Quality improvement | Pork loin | 20 kHz; 200, 400 and 600 W; 15, 30 and 45 min | • Ultrasonic bath in ice varying the power and temperature | • When the time of treatment was 15 min and 30 min, the group of 600 W showed higher P21 and lower P22 values | Luo et al.124 |
| • 9 cm × 6 cm × 3 cm samples weighting about 110 g | • For 45 min treatment, the 600 W group showed the lowest P21 values and the higher P22 values | ||||
| Beef (M. semimembranosus) | 45 kHz; 2 W cm−2; 2 min | • Ultrasonic bath with cold water | • The sonicated samples showed higher T21 values after 48, 72 and 96 h of ageing | Stadnik et al.17 | |
| • sonicated samples had lower T22 values compared to control samples at 24 and 72 h post mortem | |||||
| • The sonicated samples showed better WHC values | |||||
| Unsmoked bacon | 20 kHz; 250, 500 and 750 W; 60 min | • HIUS treatment was applied in dry-cured (3% NaCl and 0.01% NaNO2 (w/w)) bacon samples before ageing | • HIUS-treatment increased P21 and decreased P22 of samples, improving their WHC | Zhang et al.125 | |
| • Ultrasonic bath | |||||
| Frankfurter-type sausages (pork (lean and back-fat)) | 25 kHz; 240 W; 15, 20, 25, 30, and 35 min | • Ultrasonic bath | • 25 min-treatment increased P21 and decreased P22 | Zhang et al.126 | |
| Heating | Silver carp surimi (Hypophthalmichthys molitrix) | 40 kHz; 270 W; 50 min | • HIUS-assisted water bath heated at 40 °C with addition of extra virgin olive (EVO) oil, the samples then were heated in a water-bath at 90 °C for 30 min | • Sonicated samples with less than 3% (w/w) EVO showed increase of T2b and P2b and decrease of P21 | Lu et al.50 |
| • Samples with more than 3% of oil treated by HIUS showed decreased P2b and increased P21 | |||||
| • Sonicated samples had lower P22 | |||||
| Defective Jinhua ham (Biceps femoris) | 25 kHz; 300 and 1000 W; 6 h | • 300 W/40 °C; 300 W/50 °C; 1000 W/40 °C; 1000 W/50 °C treatments | • HIUS-treated samples had increased T2b, T21 and T22 | Zhou et al.54 | |
| • P2b decreased with increase of HIUS power; for the same ultrasonic power, temperature did not significantly affect P2b | |||||
| • Ultrasonic bath | • HIUS-treated samples had lower P21 and higher P22 than control | ||||
| • Increase of T2b, T21 and T22 | |||||
| Beef (anterior tendon) | 28 kHz; 60 W; 37 min | • HIUS-assisted sous-vide cooking (USV) at different cooking times | • Highest proportion of immobilized water in USV samples cooked for 60 and 80 min | Wang et al.51 | |
| • 8 cm × 6 cm × 3 cm samples weighing 150 ± 5 g | |||||
| • The samples were cooked at 71 °C | |||||
| • Ultrasonic bath | |||||
| Pork meatballs (hind legs) | 20 kHz; 150, 300, 450, 600, and 750 W; 10 min | • HIUS-assisted cooking | • Samples cooked at 450 W had the highest P21 and the lowest P22 values | Zhao et al.52 | |
| • 2.5-cm-diameter samples weighing about 30 g | |||||
| • The samples were cooked until the central temperatures reached 75 °C | |||||
| • Ultrasonic bath | |||||
| Bacterial inactivation | Oyster (Crassostrea gigas) | 20 kHz; 300 W; 7.5 W mL−1; 12.5 min | • The samples were inoculated with Vibrio parahaemolyticus and placed in a beaker with 0.85% NaCl solution and sonicated using an ultrasonic probe with 10 mm of diameter | • Small decrease in the content of immobilized water after 5 days of cold storage | Ma et al.65 |
| • After the treatment, the samples were stored at 4 °C for 5 days | |||||
| Fermentation | Beef (Semitendinosus) | 30 kHz; 300 W; 30 min | • US: HIUS treatment | • HIUS-treated samples had higher P2b values | Hu et al.58 |
| • US-BP: HIUS treatment combined with inoculation of P. acidilactici BP2 | • US and US-BP delayed values of time constants T2b, T21 and T22 compared to control | ||||
| • (2 cm × 2 cm × 10 cm) strips | • HIUS treated samples showed increase of P21 and decrease of P22 | ||||
| Beef (m. semitendinosus) | 25, 33 and 45 kHz; 65 W; 30 min | • The samples were pre-treated with ultrasound before curing | • Uncultured pre-treated samples at 25 and 45 kHz had lower P21 decrease | Ojha et al.59 | |
| • 10 cm length 10 cm, 4 cm width and 0.2 cm thickness slices | • Cultured samples pre-treated at 25 kHz and 33 kHz had lower P21 decrease | ||||
3.2 Cure
Traditional curing methods have some challenges, such as (i) the high salt content in meat, needing a posterior desalting process, (ii) possible cross-contamination due to manipulation and handling, and (iii) textural damage.5 The HIUS technology may be applied in the curing process for the improvement of the mass transfer of salt to meat, thus reducing the cure time and resulting in a product with less salt than traditional methods.5,14 Additionally, HIUS is suggested to reduce microbial load and improve or maintain product texture when applied as a brining-assisted technology, thereby avoiding the limitations of traditional curing methods.11,15Recent studies reported that ultrasound-assisted curing improved the WHC of meat products (Table 2). The mechanism used to explain these results involves the swelling of myofibrils and the extraction of soluble proteins, caused by the increased mass transfer of brine in meat induced by HIUS.15 The salt ions can bind to myofibrillar proteins, causing electrostatic repulsion and increasing the space between them.27 The enhanced gaps allow the exposition of protein side chains that can bind to water molecules.28 LF-NMR measurements carried out by Xiong et al.29 found that HIUS-assisted cure of chicken breast decreased T21 values, indicating that the immobilized water is more tightly bound to the matrix and its conversion to free water is impaired; meanwhile, Kang et al.30 reported that at high ultrasonic powers (250 W or 300 W) and treatments above 120 min, T21 values increased in beef longissimus dorsi. This is in accordance with the study by McDonnell et al.,31 which did not observe a significant difference in bound and free water constant times in HIUS-treated samples but found that 19 W cm−2 treatments increased T21 values.
Cao et al.32 found that the ultrasound-assisted enzymatic treatments improved the WHC of samples compared to the treatments without HIUS. They explained that the improvements may be the result of the action of enzymes and the cavitation phenomenon caused by ultrasound on myofibrils, which improved their capacity to retain water. However, treatments longer than 40 min had negative effects on WHC. This can be explained by the ultrasound-induced rupture of muscle cells during prolonged treatments.33 On the other hand, Ojha et al.34 found an increase in the free water population and a decrease in immobilized water in ultrasound-treated samples. Meanwhile, their results can be attributed to the increased salt content in meat, which leads to structural changes in meat proteins and osmotic dehydration. The HIUS technology showed suitable results in the curing process, increasing the WHC of the samples, when operational parameters such as power, time, and salt concentration are appropriately chosen. Nevertheless, as pointed out by Kang et al.,5 the changes in meat proteins, in HIUS-assisted curing, also depend on the type and geometry of the equipment and of the sample and the distance between the sample and the transducer; therefore, these factors still need further studies for a better understanding of the effects of HIUS on water mobility in the curing process.
Considering that studies applying HIUS with high time and power have shown contradictory results, it is reasonable to consider that, in addition to the experimental parameters of HIUS, external factors such as the matrix, the type, and the concentration of the salt used can be determining factors for WHC results. This is based on the fact that different salts have different kinetics and possibly different behaviors under the HIUS effects. However, the studies analyzed here vary in several fundamental aspects, such as the type of meat and muscle, operational conditions, and the method of application. Thus, studies evaluating the effects of these variables individually and their potential interactions are necessary.
3.3 Freezing
Freezing is a traditional method for food preservation that renders the components necessary for microbiological metabolism unusable, thereby slowing down the spoilage process. Therefore, frozen foods are recognized for having improved shelf-life.35 However, the ice crystals formed during the freezing process cause physical damage to the food structure, leading to quality losses.36 In meat products, this phenomenon increases the exudation of the water of meat during thawing.37 Thus, rapid and cost-effective freezing processes, which maintain as many as possible of the original characteristics of the matrix, are desirable. Immersion freezing is a freezing method in which the packaged food is immersed in a cryogen or a fluid refrigerant. The advantages of this method include its short time, uniform temperature, and non-contact between the food and the liquid medium.38 The combined use of immersion-freezing and HIUS has been studied because ultrasound technology increases the freezing rate and generates the smallest ice crystals, thus decreasing the physical damage caused to food when thawing.37Several authors have demonstrated that ultrasound-assisted immersion freezing, under appropriate conditions, improves the water retention of meat samples (Table 2). These results are confirmed by WHC and LF-NMR measurements. HIUS causes the formation of small, evenly distributed ice crystals through the cavitation effect. The cavitation bubbles formed by sonication act as ice nuclei, thus increasing the number of ice nuclei. Moreover, ice crystals broken when hit by ultrasonic waves lead to shortening of the ice crystals.39 Notwithstanding, Zhang et al.40 and Zhang et al.41 found that ultrasonic treatments, under some conditions (30 kHz; 300 W; 8 min; porcine longissimus muscles, and 30 kHz; 245 W; 8 min; chicken breast, respectively), increased water mobility and decreased the WHC of samples. The authors explained that these results are due to the high powers of ultrasound, which destroy the protein structure, thereby reducing the ability of proteins to retain water. Therefore, despite the promising application of HIUS as a freezing-assisted technology, there is a need to optimize parameters, such as power and time, to enhance the cavitation effects positively.
Most of the studies evaluated in this review assessed the effects of HIUS on the thawing of various samples, using application times ranging from 8 to 9 minutes. Overall, a trend can be observed suggesting that high power levels decrease water retention in the samples. However, this hypothesis disregards other parameters such as the application mode, equipment, and matrix used. Thus, it is worth highlighting that the application mode (continuous or pulsed) and the equipment (bath or probe) can affect the results due to ultrasonic efficiency, homogeneity, and temperature control, meaning that even favorable time and power conditions may not lead to the expected result.
3.4 Thawing
Thawing is the opposite of freezing and is the processing step during which the major loss of sensory and nutritional qualities, as well as damage to the muscle tissue of meat, occurs. Although slow thawing times are better for meat products, HIUS can achieve good results when correctly applied in the thawing process.5 Several authors have found that ultrasound-assisted thawing improves the retention of water in meat products (Table 2). The principal explanation for these results is that the HIUS reduces protein damage, thereby delaying drip loss.14 Li et al.42 reported that the use of salt at a 0.10% concentration helps to avoid osmotic dehydration in mirror carp (Cyprinus carpio L.), as shown by the lower increase in T21 values among the analyzed samples. Meanwhile, Sun et al.43 found that the HIUS treatment at 500 W disrupted the muscle cell structure through ultrasonic waves, as indicated by the increase in T21 values, suggesting that the muscle's capacity to bind water was impaired and the interactions between water and proteins were weakened. Their results are in accordance with the studies of Zhang et al.44 and Zhang et al.45 (Table 2), which present similar trends for T21 values in chicken breast muscle thawed using HIUS, and that samples with decreased T21 values had higher P21 values, and vice versa. They explained that, under appropriate conditions, the HIUS can slow down the degradation of muscle tissue, strengthening the interactions between water and meat proteins, and increasing the WHC of the meat. As explained by Leygonie et al.,46 the decrease in thawing time may be related to the reduction of exudation of water, caused by the reabsorption of water by the fiber cells after the melting of the ice crystals in the extracellular space. This depends on the relationship between the amount of water available and the capacity of fiber muscles to absorb this water.HIUS-assisted thawing is a promising approach that can promote higher thawing rates, decreasing the damage suffered by meat proteins during thawing. These beneficial effects can be observed by LF-NMR as a decrease in T21 values and/or an increase in P21 values, indicating less damaged myofibrils. Nevertheless, the application of HIUS in the thawing process still presents some challenges, as pointed out by Soltani Firouz et al.47 and Kang et al.,5 such as the heat generated by cavitation bubbles induced by HIUS can overheat the surface of the matrix while the inner layer is less heated. This can cause sensory and nutritional losses of the meat, requiring optimized parameters for its application in meat thawing.
Thus, it is possible to assume that the power and application time of HIUS are determining factors in this process. Furthermore, another factor that cannot be overlooked is the sample format, which, if chosen appropriately, can reduce the application time and the effects of surface overheating. Furthermore, as discussed earlier, the equipment and application method, which can have important effects on temperature and efficiency, should be considered. Additionally, the frequency can also affect the results, as depicted in Table 2.
3.5 Heating
Heat has been widely used in food processes such as pasteurization, sterilization, drying, evaporation, and cooking.48 However, the food is exposed to high temperatures for a long time to reach the required quality and safety parameters, promoting chemical reactions responsible for color changes, and loss of odor, taste, and nutrients during and after the thermal processing.49 Therefore, processes that decrease the damage caused to myofibrillar proteins are desirable, because their functional properties are directly related to the quality of meat products.5 Thus, HIUS-assisted heating is a worthy alternative, since HIUS can improve the heat transfer, reducing the processing time and the damage caused to the muscle tissue, resulting in faster cooking, higher water retention, and lower cooking losses.14In our review, four studies about heating processing assisted by HIUS were assessed (Table 2). In the studies of Lu et al.,50 Wang et al.,51 and Zhao et al.,52 samples treated with HIUS showed an increase in P21 values under appropriate conditions (Table 2). They explained that heat treatment assisted by HIUS can solubilize and partially denature the myofibrillar proteins, resulting in the formation of a complex gel structure that retains water. This is in accordance with the work of Saleem & Ahmad,53 which found that HIUS facilitated the denaturation and aggregation of myofibrillar proteins in chicken breast (pectoralis major), forming a more homogeneous network of gels. On the other hand, the HIUS-assisted heat treatment of Jinhua ham (25 kHz; 300 and 1000 W; 6 h) increased the amount of free water and decreased the immobilized water content, possibly due to the denaturation of proteins and shortening of myofibrils, which caused the exudation of water into the extra myofibrillar space.54 The application of HIUS in thermal processing can improve the water retention in meat and meat products, when operational parameters are properly chosen. Moreover, the limited number of studies and the significant variations in time (50 min–6 h) and power (60–1000 W) make it challenging to comprehend their effects on water mobility in these matrices.
3.6 Fermentation
Fermented meat and meat products have an extended shelf life, distinct flavor, color, and texture, and their consumption has increased over the last few years.55 The fermentation process is made by microorganisms or enzymes and requires specific conditions of temperature and humidity to occur.56 The fermentation can be carried out by the natural microbiota present in the raw material, or by the use of a starter culture such as lactic acid bacteria (LAB) and coagulase-negative Staphylococci (CNS).57 During the fermentation process with LAB, protein degradation in meat is caused by enzymes of the meat and produced by the bacteria, improving several parameters such as texture, flavor, and nutritional composition.55,56Hu et al.58 and Ojha et al.59 studied the production of beef jerky by the fermentation of beef (semitendinosus) with P. acidilactici BP2 and L. sakei, respectively, using HIUS as pre-treatment. In the work of Hu et al.58 fermented samples treated with HIUS and inoculated with LAB had the lowest T21 and T22 values, but did not have significantly different water populations. They explained that the improvement in water retention is a result of the synergic effect of cavitation caused by HIUS and the hydrolysis of muscle proteins caused by LAB. Ojha et al.59 observed that the pre-treated samples, under appropriate conditions (Table 2), had fewer changes in the immobilized water population. The effects of HIUS and inoculation of a starter culture on T2 values can be explained by the possible increase in substrate availability for fermentative bacteria caused by HIUS-induced cavitation on meat proteins.47 In this context, despite the lack of studies on water migration of HIUS-treated fermented meat products, we can hypothesize that HIUS and starter cultures present an additive or synergistic effect in promoting water retention; however further studies are needed to assess the extent of this combined effect on the physicochemical, functional, and sensory properties and propose the appropriate conditions for using these technologies.
3.7 Bacterial inactivation
Meat and fish products are highly susceptible to bacterial spoilage due to their composition, nutritional value, higher water activity, and high moisture content, which have negative consequences for the economy and public health.60,61 Traditional food preservation methods, such as heating and the use of chemical compounds, present several issues, including high energy costs, incomplete inactivation of microorganisms, the formation of harmful compounds that affect human health, higher acidity and salt content, and final products with poor nutritional and sensory attributes.62,63 Thus, the use of HIUS technology for bacterial inactivation is a viable alternative to conventional methods, as it can inactivate microorganisms and clear surfaces through the cavitation effect, without impairing the nutritional and textural properties of the meat matrix; however, the degree of decontamination depends on the nature of the microorganism.7,64 Ma et al.65 compared the HIUS with heat treatment in the decontamination of oysters (Crassostrea gigas) inoculated with Vibrio parahaemolyticus. Their results showed that the sonicated samples had lower loss of immobilized water than thermal treatment after five days of storage. The results can be explained by the difference in protein denaturation induced by the different treatments. They explained that heat caused the denaturation and shrinkage of myofibrillar proteins, allowing water to be extruded between them. Nevertheless, the cavitation effect produced by HIUS can modify the structure of proteins, increasing the protein–water interactions and, thus, the WHC of the meat matrix.5 Therefore, HIUS seems to be a promising alternative to achieve a safe meat product with improved technological properties. On the other hand, the literature lacks data on water mobility regarding the inactivation of foodborne pathogens in red meat matrices.3.8 Quality parameters
Quality parameters such as pH, WHC, color, tenderness, juiciness, flavor, and appearance affect the overall quality and consumer's acceptance of meat and meat products.66 Thus, the application of HIUS on meat processing has been intensively studied due to its promising results without harming the quality of the final product.62 According to the results previously discussed, HIUS, when applied alone, shows good results on water retention in different meat and meat products. The main explanation for this is the implosion of cavitation bubbles, causing the disruption of protein aggregates in the myofibrils, decreasing particle size and increasing the superficial area, improving the protein–water interactions and WHC.67There is no clear relationship between texture parameters and water mobility and distribution. Kang et al.25 found that the samples with higher P21 values had higher hardness in pork meat batters of mesoglutaeus, in contrast with the study of Zou et al.,68 which showed lower hardness in samples with the highest P21 in chicken breast meat. Ojha et al.34 stated that samples of pork (m. semitendinosus) with lower WHC had lower Warner–Bratzler shear force (WBSF) values, indicating a higher tenderization in samples with lower P21 and T21 values. On the other hand, Zhao et al.52 reported a negative relationship between P21 values and the hardness of pork meatballs. In fact, this discordance has already been discussed by Pearce et al.,16 who suggested that this divergence may be a result of differences in intramuscular fat or glycogen contents of meat. Moreover, it's worth mentioning that Tasoniero et al.69 found a correlation between hardness and T2b, T21, and T22 relaxation times only in wooden-breast chicken meat, but not in normal breast muscle.
Furthermore, the relationship between color and LF-NMR measurements remains unclear. Kang et al.25 demonstrated an increase in yellowness (b*) and a decrease in redness (a*) values, accompanied by an increase in ultrasonic time (i.e., an increase in P21); however, the same parameter did not significantly affect lightness (L*) values in pork meat batters. Nevertheless, Zhao et al.52 found that the samples of pork meatballs with the highest P21 values showed both decreased L*, a*, and b* values, and Cao et al.32 showed that chicken breast samples with increased P21 had increased L* and b* values, and decreased a* values. Thus, it is not possible to state that the changes in the color of meat are direct results of changes in water mobility and distribution, because the color of meat and meat products is affected by several factors, such as pH, age, muscle type, species, pre- and postmortem conditions, and myoglobin contents.70,71
The results for the changes in pH are discrepant. Zhang et al.40 and Sun et al.43 found no significant differences among samples with different T21 values, whereas Shen et al.72 reported that samples with higher P21 values had higher pH levels, and vice versa. This is in accordance with Pearce et al.,16 who said that the reason for this is unknown.
Regarding the protein and lipid oxidation, the results are diverse. Li et al.73 did not observe significant differences in protein oxidation among samples with different P21 values; however, lipid oxidation was lower in samples with higher P21 values. In contrast, the work of Lv & Xie,74 observed significantly different protein oxidation results in samples with similar P21 values, but samples with higher P21 values presented a higher degree of lipid oxidation. The effect of protein oxidation on water mobility is dependent on the degree of oxidation, which can increase the swelling of the myofilaments, enhancing the WHC, or increase the number of cross-linked structures between proteins, impairing the ability of myofibrils to swell, decreasing the WHC.75 These contradictory results indicate that the relationship between the results provided by LF-NMR and important quality parameters of meat, such as pH, color, and texture, needs to be better understood. Since the studies differ greatly from one another, our ability to formulate hypotheses becomes limited. However, it is still reasonable to assume that the type of meat (e.g., beef, chicken, pork, or fish), the type of muscle fiber (e.g., type I or type II), and the post-mortem time have a significant impact on the results. This is important because such factors are closely related to glycogen and fat content, pH, and also to the amount of myoglobin and, consequently, the meat's response to oxidative stress caused by the HIUS application. The effects of these parameters on the water distribution and mobility were discussed by Pearce et al.16 Also, a detailed discussion of the relationship between muscle fiber type and meat quality parameters can be found in the article by Picard & Gagaoua.76 Thus, it is reasonable to assume that the type of meat and muscle fiber may have caused such discrepancies in the results after the application of HIUS.
4 Challenges and perspectives
The HIUS is an emergent technology with great potential for meat and meat product processing. The studies assessed in this review generally showed promising results. Nevertheless, given their diverse nature in terms of equipment, operational parameters, and samples, it's not possible to determine the causes of the differences among them. This was considered by Soltani Firouz et al.47 as a challenge to the industrial application of HIUS. Moreover, it's worth mentioning that no article included in this review applies HIUS to alternative meat products. This indicates the scarcity of this type of work. However, it is worth mentioning that HIUS can be applied in the processing of edible insects and plant proteins, for example, increasing their solubility, extraction, and modifying the texture when applied appropriately, as reviewed by Sengar et al.77 and Ojha et al.78 Meat alternatives are meat-free products that try to emulate the sensory and nutritional attributes of conventional meat, and include plant-based, legumes, edible mushrooms, lab-grown meat, and edible insects.79–81 For example, plant proteins are constituted mainly by globular proteins, which makes the structure of plant-based products different from traditional meat; moreover, the presence of carbohydrate polymers also affects the WHC of these products.82,83 Also, given that the effects of HIUS can vary depending on the amino acid composition,14 it is expected that HIUS will yield different results in food products containing different species of edible insects. Thus, the effects of HIUS on this matrix type and, consequently, on parameters such as WHC, water distribution, juiciness, and texture still need to be understood. Furthermore, based on these results, it is possible to say that the standardization of parameters and the inconsistency of results are the primary challenges for implementing HIUS on an industrial scale. This is because the limited knowledge of the effects of HIUS on different matrices reduces the predictability and reproducibility of the results. Furthermore, as some reviews point out, HIUS faces additional challenges, including low energy efficiency, limited microbiological inactivation, limited information regarding food safety, and regulatory issues.47,84,85 Additionally, as noted by Monteiro et al.,86 misinformation on the part of consumers can also be a barrier to the market. Another aspect that should be considered is that, although HIUS may be potentially advantageous and safer when compared to traditional food preservation methods, it may not be so in relation to other non-thermal methods such as HPP and UV-C, for example, as discussed by Yaya-González et al.85 Finally, it is worth highlighting that HIUS equipment can also be highly noisy, potentially posing an occupational hazard. Therefore, there is still a long way to go in optimizing and scaling HIUS for industrial use.5 Conclusion
Our review assessed water mobility in meat products undergoing HIUS-assisted processes, such as curing, freezing, and thawing, and discussed the main mechanisms and discrepancies in the results. From the results discussed, it was possible to note that the effects of HIUS application on water retention in meat products are contradictory and closely related to the extent of damage caused to muscle tissue. Thus, T2 values measured by LF-NMR serve as an indicator of the structural changes undergone by the meat, such as an increase in the space between myofibrils. It was observed that, under optimized power, temperature, and frequency conditions, HIUS can alter the hydrophobicity of proteins, thereby increasing protein–water interactions and favoring the formation of thinner and more homogeneous gels that retain more water. However, outside of these conditions, at high times and power levels, for example, HIUS can favor the formation of hydrophobic protein–protein interactions, expel water, and even destroy muscle structure, decreasing WHC. The possibilities of applying HIUS to alternative meat products were also discussed. Although studies have applied HIUS in the processing of alternative proteins, information on its applications in hybrid products, such as beef patties, is lacking. Finally, the implementation of these technologies (LF-NMR and HIUS) still faces several challenges. For LF-NMR, the difficulty in interpreting and processing the data hinders the technique's widespread use. As for HIUS, the lack of standardization, low energy efficiency, and lack of data regarding the safety of treated foods hinder the scaling up of this technology to an industrial scale.Author contributions
Davi S. Santos: conceptualization, methodology, investigation, writing – original draft; Yago A. A. Bernardo: conceptualization, methodology, writing – review & editing, supervision; Carlos A. Conte-Junior: resources, writing – review & editing, supervision, funding acquisition.Conflicts of interest
The authors declare that they have no conflict of interest.Data availability
There were no generated data.Acknowledgements
The authors are thankful for the financial support provided by the Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) Brazil – grant numbers [E-26/200.891/2021 and E-26/200.060/2024], and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) – grant numbers [313119/2020-1 and 139777/2024-5].References
- H. C. J. Godfray, P. Aveyard, T. Garnett, J. W. Hall, T. J. Key and J. Lorimer, et al., Meat consumption, health, and the environment, Science, 2018, 361(6399), eaam5324 CrossRef PubMed.
- T. Lu, B. A. Abdalla Gibril, J. Xu and X. Xiong, Unraveling the Genetic Foundations of Broiler Meat Quality: Advancements in Research and Their Impact, Genes, 2024, 15(6), 746 Search PubMed.
- U. Ur Rahman, A. Sahar, A. Ishaq, R. M. Aadil, T. Zahoor and M. H. Ahmad, Advanced meat preservation methods: A mini review, J. Food Saf., 2018, 38(4), e12467 CrossRef.
- H. B. Jadhav, U. S. Annapure and R. R. Deshmukh, Non-thermal Technologies for Food Processing, Front. Nutr., 2021, 8, 657090 CrossRef PubMed.
- D. Kang, W. Zhang, J. M. Lorenzo and X. Chen, Structural and functional modification of food proteins by high power ultrasound and its application in meat processing, Crit. Rev. Food Sci. Nutr., 2021, 61(11), 1914–1933 CrossRef CAS PubMed.
- L. C. S. Lima, A. P. A. A. Salim, I. M. M. Trezze, M. S. Ferreira, M. L. G. Monteiro and F. J. Vasconcellos-Junior, et al., High-intensity ultrasound improves color and oxidative stability of beef from grain-fed and pasture-fed Nellore cattle, Meat Sci., 2023, 206, 109324 CrossRef CAS PubMed.
- D. K. A. Rosario, B. L. Rodrigues, P. C. Bernardes and C. A. Conte-Junior, Principles and applications of non-thermal technologies and alternative chemical compounds in meat and fish, Crit. Rev. Food Sci. Nutr., 2021, 61(7), 1163–1183 CrossRef CAS PubMed.
- Y. A. D. A. Bernardo, D. K. A. Do Rosario and C. A. Conte-Junior, Principles, Application, and Gaps of High-Intensity Ultrasound and High-Pressure Processing to Improve Meat Texture, Foods, 2023, 12(3), 476 CrossRef CAS PubMed.
- A. D. Alarcon-Rojo, L. M. Carrillo-Lopez, R. Reyes-Villagrana, M. Huerta-Jiménez and I. A. Garcia-Galicia, Ultrasound and meat quality: A review, Ultrason. Sonochem., 2019, 55, 369–382 CrossRef CAS PubMed.
- R. O. Caraveo-Suarez, I. A. Garcia-Galicia, E. Santellano-Estrada, L. M. Carrillo-Lopez, M. Huerta-Jimenez and A. D. Alarcon-Rojo, Integrated multivariate analysis as a tool to evaluate effects of ultrasound on beef quality, J. Food Process Eng., 2023, 46(6), e14112 CrossRef CAS.
- L. P. Marques, Y. A. A. Bernardo and C. A. Conte-Junior, Applications of high-intensity ultrasound on shrimp: Potential, constraints, and prospects in the extraction and retrieval of bioactive compounds, safety, and quality, J. Food Sci., 2024, 89(6), 3148–3166 CrossRef CAS PubMed.
- E. Beitia, E. Gkogka, P. Chanos, C. Hertel, V. Heinz and V. Valdramidis, et al., Microbial decontamination assisted by ultrasound-based processing technologies in food and model systems: A review, Compr. Rev. Food Sci. Food Saf., 2023, 22(4), 2802–2849 CrossRef PubMed.
- Y. Tao and D. W. Sun, Enhancement of Food Processes by Ultrasound: A Review, Crit. Rev. Food Sci. Nutr., 2015, 55(4), 570–594 CrossRef PubMed.
- A. D. Alarcon-Rojo, H. Janacua, J. C. Rodriguez, L. Paniwnyk and T. J. Mason, Power ultrasound in meat processing, Meat Sci., 2015, 107, 86–93 CrossRef CAS PubMed.
- Y. A. A. Bernardo, D. K. A. Do Rosario, L. P. Marques, B. D. Da Silva, M. L. G. Monteiro and C. A. Conte Junior, High-intensity ultrasound processing to improve the quality of semitendinosus steak from Bos indicus cattle, Sustainable Food Technol., 2025, 3(2), 482–491 RSC.
- K. L. Pearce, K. Rosenvold, H. J. Andersen and D. L. Hopkins, Water distribution and mobility in meat during the conversion of muscle to meat and ageing and the impacts on fresh meat quality attributes — A review, Meat Sci., 2011, 89(2), 111–124 CrossRef PubMed.
- J. Stadnik, Z. J. Dolatowski and H. M. Baranowska, Effect of ultrasound treatment on water holding properties and microstructure of beef (m. semimembranosus) during ageing, LWT–Food Sci. Technol., 2008, 41(10), 2151–2158 CrossRef CAS.
- C. D. S. Carneiro, E. T. Mársico, R. D. O. R. Ribeiro, C. A. Conte Júnior, T. S. Álvares and E. F. O. De Jesus, Quality Attributes in Shrimp Treated with Polyphosphate after Thawing and Cooking: A Study Using Physicochemical Analytical Methods and Low-F ield1 H NMR, J. Food Process Eng., 2013, 36(4), 492–499 CrossRef CAS.
- U. Erikson, I. B. Standal, I. G. Aursand, E. Veliyulin and M. Aursand, Use of NMR in fish processing optimization: a review of recent progress, Magn. Reson. Chem., 2012, 50(7), 471–480 CrossRef CAS PubMed.
- M. C. Ezeanaka, J. Nsor-Atindana and M. Zhang, Online Low-field Nuclear Magnetic Resonance (LF-NMR) and Magnetic Resonance Imaging (MRI) for Food Quality Optimization in Food Processing, Food Bioprocess Technol., 2019, 12(9), 1435–1451 Search PubMed.
- J. Yao, Y. Chen, S. Tian, X. Zhu, T. Li and Y. Fu, Investigating Morphology of Food Systems and Water-biopolymer Interactions in Food Using 1H NMR Relaxometry, Food Biophys., 2022, 17(2), 150–164 CrossRef.
- C. D. S. Carneiro, E. T. Mársico, R. D. O. R. Ribeiro, C. A. Conte-Júnior, S. B. Mano and C. J. C. Augusto, et al., Low-Field Nuclear Magnetic Resonance (LF NMR 1H) to assess the mobility of water during storage of salted fish (Sardinella brasiliensis), J. Food Eng., 2016, 169, 321–325 Search PubMed.
- J. Mitchell, L. F. Gladden, T. C. Chandrasekera and E. J. Fordham, Low-field permanent magnets for industrial process and quality control, Prog. Nucl. Magn. Reson. Spectrosc., 2014, 76, 1–60 CrossRef CAS PubMed.
- I. Sánchez-Alonso, P. Moreno and M. Careche, Low field nuclear magnetic resonance (LF-NMR) relaxometry in hake (Merluccius merluccius, L.) muscle after different freezing and storage conditions, Food Chem., 2014, 153, 250–257 CrossRef PubMed.
- Z. L. Kang, X. Y. Shang, Y. P. Li and H. J. Ma, Effect of ultrasound-assisted sodium bicarbonate treatment on gel characteristics and water migration of reduced-salt pork batters, Ultrason. Sonochem., 2022, 89, 106150 CrossRef CAS PubMed.
- D. Moher, A. Liberati, J. Tetzlaff and D. G. Altman, Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement, Int. J. Surg., 2010, 8(5), 336–341 CrossRef PubMed.
- Y. A. A. Bernardo, M. L. G. Monteiro, L. P. Marques and C. A. Conte-Junior, Modeling high-intensity ultrasound to enhance shrimp (Penaeus vannamei) brining without compromising crucial purchase attributes, J. Food Process Eng., 2024, 47(6), e14661 CrossRef CAS.
- A. Visy, G. Jónás, D. Szakos, Z. Horváth-Mezőfi, K. I. Hidas and A. Barkó, et al., Evaluation of ultrasound and microbubbles effect on pork meat during brining process, Ultrason. Sonochem., 2021, 75, 105589 Search PubMed.
- G. Xiong, X. Fu, D. Pan, J. Qi, X. Xu and X. Jiang, Influence of ultrasound-assisted sodium bicarbonate marination on the curing efficiency of chicken breast meat, Ultrason. Sonochem., 2020, 60, 104808 CrossRef CAS PubMed.
- K. D. cheng, G. X. qin, Ge Q. feng, Z. G. hong and Z. W. gang, Effects of ultrasound on the beef structure and water distribution during curing through protein degradation and modification, Ultrason. Sonochem., 2017, 38, 317–325 Search PubMed.
- C. K. McDonnell, P. Allen, C. Morin and J. G. Lyng, The effect of ultrasonic salting on protein and water–protein interactions in meat, Food Chem., 2014, 147, 245–251 CrossRef CAS PubMed.
- C. Cao, Z. Xiao, H. Tong, X. Tao, D. Gu and Y. Wu, et al., Effect of ultrasound-assisted enzyme treatment on the quality of chicken breast meat, Food Bioprod. Process., 2021, 125, 193–203 CrossRef CAS.
- H. Chang, Q. Wang, C. Tang and G. Zhou, Effects of Ultrasound Treatment on Connective Tissue Collagen and Meat Quality of Beef Semitendinosus Muscle, J. Food Qual., 2015, 38(4), 256–267 CrossRef CAS.
- K. S. Ojha, D. F. Keenan, A. Bright, J. P. Kerry and B. K. Tiwari, Ultrasound assisted diffusion of sodium salt replacer and effect on physicochemical properties of pork meat, Int. J. Food Sci. Technol., 2016, 51(1), 37–45 CrossRef CAS.
- Y. H. B. Kim, J. Seo, R. Kemp and A. Stuart, Effect of postmortem aging and fast-freezing on meat quality of various lamb cuts under prolonged frozen storage and repeated freezing/thawing conditions, Meat Muscle Biol., 2024, 8(1), 17604 CAS . Available from: https://www.iastatedigitalpress.com/mmb/article/id/17604/.
- C. Bilbao-Sainz, A. J. G. Sinrod, L. Dao, G. Takeoka, T. Williams and D. Wood, et al., Preservation of grape tomato by isochoric freezing, Food Res. Int., 2021, 143, 110228 CrossRef CAS PubMed.
- C. James, G. Purnell and S. J. James, A Review of Novel and Innovative Food Freezing Technologies, Food Bioprocess Technol., 2015, 8(8), 1616–1634 CrossRef CAS.
- D. Liang, F. Lin, G. Yang, X. Yue, Q. Zhang and Z. Zhang, et al., Advantages of immersion freezing for quality preservation of litchi fruit during frozen storage, LWT–Food Sci. Technol., 2015, 60(2), 948–956 CrossRef CAS.
- L. Zheng and D. W. Sun, Innovative applications of power ultrasound during food freezing processes—a review, Trends Food Sci. Technol., 2006, 17(1), 16–23 CrossRef CAS.
- M. Zhang, N. Haili, Q. Chen, X. Xia and B. Kong, Influence of ultrasound-assisted immersion freezing on the freezing rate and quality of porcine longissimus muscles, Meat Sci., 2018, 136, 1–8 CrossRef CAS PubMed.
- C. Zhang, X. Li, H. Wang, X. Xia and B. Kong, Ultrasound-assisted immersion freezing reduces the structure and gel property deterioration of myofibrillar protein from chicken breast, Ultrason. Sonochem., 2020, 67, 105137 CrossRef CAS PubMed.
- F. Li, B. Wang, B. Kong, X. Xia and Y. Bao, Impact of Ultrasound-assisted Saline Thawing on the Technological Properties of mirror carp (Cyprinus carpio L.), Ultrason. Sonochem., 2022, 86, 106014 CrossRef CAS PubMed.
- Q. Sun, B. Kong, S. Liu, O. Zheng and C. Zhang, Ultrasound-assisted thawing accelerates the thawing of common carp (Cyprinus carpio) and improves its muscle quality, LWT–Food Sci. Technol., 2021, 141, 111080 CrossRef CAS.
- C. Zhang, H. Liu, X. Xia, F. Sun and B. Kong, Effect of ultrasound-assisted immersion thawing on emulsifying and gelling properties of chicken myofibrillar protein, LWT–Food Sci. Technol., 2021, 142, 111016 CrossRef CAS.
- C. Zhang, Q. Sun, Q. Chen, Q. Liu and B. Kong, Effectiveness of ultrasound-assisted immersion thawing on the thawing rate and physicochemical properties of chicken breast muscle, J. Food Sci., 2021, 86(5), 1692–1703 Search PubMed.
- C. Leygonie, T. J. Britz and L. C. Hoffman, Impact of freezing and thawing on the quality of meat: Review, Meat Sci., 2012, 91(2), 93–98 CrossRef PubMed.
- M. Soltani Firouz, H. Sardari, P. Alikhani Chamgordani and M. Behjati, Power ultrasound in the meat industry (freezing, cooking and fermentation): Mechanisms, advances and challenges, Ultrason. Sonochem., 2022, 86, 106027 CrossRef CAS PubMed.
- R. N. Pereira and A. A. Vicente, Environmental impact of novel thermal and non-thermal technologies in food processing, Food Res. Int., 2010, 43(7), 1936–1943 CrossRef.
- B. Ling, J. Tang, F. Kong, E. J. Mitcham and S. Wang, Kinetics of Food Quality Changes During Thermal Processing: a Review, Food Bioprocess Technol., 2015, 8(2), 343–358 CrossRef CAS.
- Y. Lu, Y. Zhu, T. Ye, Y. Nie, S. Jiang and L. Lin, et al., Physicochemical properties and microstructure of composite surimi gels: The effects of ultrasonic treatment and olive oil concentration, Ultrason. Sonochem., 2022, 88, 106065 CrossRef CAS PubMed.
- H. Wang, Z. Gao, X. Guo, S. Gao, D. Wu and Z. Liu, et al., Changes in Textural Quality and Water Retention of Spiced Beef under Ultrasound-Assisted Sous-Vide Cooking and Its Possible Mechanisms, Foods, 2022, 11(15), 2251 CrossRef CAS PubMed.
- X. Zhao, X. Sun, B. Lai, R. Liu, M. Wu and Q. Ge, et al., Effects of ultrasound-assisted cooking on the physicochemical properties and microstructure of pork meatballs, Meat Sci., 2024, 208, 109382 CrossRef CAS PubMed.
- R. Saleem and R. Ahmad, Effect of ultrasonication on secondary structure and heat induced gelation of chicken myofibrils, J. Food Sci. Technol., 2016, 53(8), 3340–3348 Search PubMed.
- C. Y. Zhou, Q. Xia, J. He, Y. Y. Sun, Y. L. Dang and C. R. Ou, et al., Improvement of ultrasound-assisted thermal treatment on organoleptic quality, rheological behavior and flavor of defective dry-cured ham, Food Biosci., 2021, 43, 101310 CrossRef CAS.
- J. Mao, X. Wang, H. Chen, Z. Zhao, D. Liu and Y. Zhang, et al., The Contribution of Microorganisms to the Quality and Flavor Formation of Chinese Traditional Fermented Meat and Fish Products, Foods, 2024, 13(4), 608 CrossRef CAS PubMed.
- Z. Wang, Z. Wang, L. Ji, J. Zhang, Z. Zhao and R. Zhang, et al., A Review: Microbial Diversity and Function of Fermented Meat Products in China, Front. Microbiol., 2021, 12, 645435 CrossRef PubMed.
- K. S. Ojha, J. P. Kerry, G. Duffy, T. Beresford and B. K. Tiwari, Technological advances for enhancing quality and safety of fermented meat products, Trends Food Sci. Technol., 2015, 44(1), 105–116 CrossRef CAS.
- Y. Hu, Z. Dong, R. Wen, B. Kong, W. Yu and J. Wang, et al., Combination of ultrasound treatment and starter culture for improving the quality of beef jerky, Meat Sci., 2023, 204, 109240 Search PubMed.
- K. S. Ojha, J. P. Kerry and B. K. Tiwari, Investigating the influence of ultrasound pre-treatment on drying kinetics and moisture migration measurement in Lactobacillus sakei cultured and uncultured beef jerky, LWT–Food Sci. Technol., 2017, 81, 42–49 CrossRef CAS.
- A. D. J. Cortés-Sánchez, M. E. Jaramillo-Flores, M. Díaz-Ramírez, L. D. Espinosa-Chaurand and E. Torres-Ochoa, Biopreservation and the Safety of Fish and Fish Products, the Case of Lactic Acid Bacteria: A Basic Perspective, Fishes, 2024, 9(8), 303 CrossRef.
- J. Chen, J. Zhang, N. Wang, B. Xiao, X. Sun and J. Li, et al., Critical review and recent advances of emerging real-time and non-destructive strategies for meat spoilage monitoring, Food Chem., 2024, 445, 138755 CrossRef CAS PubMed.
- W. Khalid, A. Maggiolino, J. Kour, M. S. Arshad, N. Aslam and M. F. Afzal, et al., Dynamic alterations in protein, sensory, chemical, and oxidative properties occurring in meat during thermal and non-thermal processing techniques: A comprehensive review, Front. Nutr., 2023, 9, 1057457 CrossRef PubMed.
- T. Aymerich, P. A. Picouet and J. M. Monfort, Decontamination technologies for meat products, Meat Sci., 2008, 78(1–2), 114–129 CrossRef CAS PubMed.
- Y. Alves De Aguiar Bernardo, D. Kaic Alves Do Rosario and C. Adam Conte-Junior, Ultrasound on Milk Decontamination: Potential and Limitations Against Foodborne Pathogens and Spoilage Bacteria, Food Rev. Int., 2023, 39(1), 320–333 CrossRef.
- J. Ma, L. Meng, S. Wang, J. Li and X. Mao, Inactivation of Vibrio parahaemolyticus and retardation of quality loss in oyster (Crassostrea gigas) by ultrasound processing during storage, Food Res. Int., 2023, 168, 112722 CrossRef CAS PubMed.
- G. A. Ferreira, A. G. Barro, D. K. Terto, E. B. Bosso, É. R. Dos Santos and N. N. Ogawa, et al., Sensory quality of beef with different ultimate pH values – A Brazilian perspective, Meat Sci., 2024, 209, 109415 CrossRef CAS PubMed.
- A. Amiri, P. Sharifian and N. Soltanizadeh, Application of ultrasound treatment for improving the physicochemical, functional and rheological properties of myofibrillar proteins, Int. J. Biol. Macromol., 2018, 111, 139–147 CrossRef CAS PubMed.
- Y. Zou, L. Li, J. Yang, B. Yang, J. Ma and D. Wang, et al., Effect of ultrasound assisted collagen peptide of chicken cartilage on storage quality of chicken breast meat, Ultrason. Sonochem., 2022, 89, 106154 CrossRef CAS PubMed.
- G. Tasoniero, H. C. Bertram, J. F. Young, A. Dalle Zotte and E. Puolanne, Relationship between hardness and myowater properties in Wooden Breast affected chicken meat: A nuclear magnetic resonance study, LWT–Food Sci. Technol., 2017, 86, 20–24 CrossRef CAS.
- R. A. Mancini and M. C. Hunt, Current research in meat color, Meat Sci., 2005, 71(1), 100–121 CrossRef CAS PubMed.
- U. S. Geletu, M. A. Usmael, Y. Y. Mummed, and A. M. Ibrahim, Quality of Cattle Meat and Its Compositional Constituents, ed. S. Nandi, Veterinary Medicine International, 2021, pp. 1–9 Search PubMed.
- X. Shen, J. Gu, X. Jiang, Q. Liang, D. Wang and S. Zhang, et al., Effect of different thawing methods on the quality of yellowtail (Seriola quinqueradiata) fillets, Int. J. Food Sci. Technol., 2024, 59(8), 5389–5397 CrossRef CAS.
- H. Li, L. Wang, J. Wang, X. Li, J. Li and F. Cui, et al., Effects of ultrasound–assisted freezing on the quality of large yellow croaker (Pseudosciaena crocea) subjected to multiple freeze–thaw cycles, Food Chem., 2023, 404, 134530 CrossRef CAS PubMed.
- Y. Lv and J. Xie, Quality of Cuttlefish as Affected by Different Thawing Methods, Int. J. Food Prop., 2022, 25(1), 33–52 CrossRef CAS.
- R. Domínguez, M. Pateiro, P. E. S. Munekata, W. Zhang, P. Garcia-Oliveira and M. Carpena, et al., Protein Oxidation in Muscle Foods: A Comprehensive Review, Antioxidants, 2021, 11(1), 60 CrossRef PubMed.
- B. Picard and M. Gagaoua, Muscle Fiber Properties in Cattle and Their Relationships with Meat Qualities: An Overview, J. Agric. Food Chem., 2020, 68(22), 6021–6039 CrossRef CAS PubMed.
- A. S. Sengar, N. Thirunavookarasu, P. Choudhary, M. Naik, A. Surekha and C. K. Sunil, et al., Application of power ultrasound for plant protein extraction, modification and allergen reduction – A review, Appl. Food Res., 2022, 2(2), 100219 CrossRef CAS.
- S. Ojha, S. Bußler, M. Psarianos, G. Rossi and O. K. Schlüter, Edible insect processing pathways and implementation of emerging technologies, J. Insect Food Feed, 2021, 7(5), 877–900 Search PubMed.
- M. Starowicz, K. Kubara Poznar and H. Zieliński, What are the main sensory attributes that determine the acceptance of meat alternatives?, Curr. Opin. Food Sci., 2022, 48, 100924 CrossRef CAS.
- Y. A. A. Bernardo and C. A. Conte-Junior, Oxidative stability in edible insects: Where is the knowledge frontier?, Trends Food Sci. Technol., 2024, 148, 104518 CrossRef CAS.
- B. D. Da Silva and C. A. Conte-Junior, Perspectives on cultured meat in countries with economies dependent on animal production: A review of potential challenges and opportunities, Trends Food Sci. Technol., 2024, 149, 104551 CrossRef CAS.
- S. Y. J. Sim, A. Srv, J. H. Chiang and C. J. Henry, Plant Proteins for Future Foods: A Roadmap, Foods, 2021, 10(8), 1967 CrossRef CAS PubMed.
- R. Moss, J. LeBlanc, M. Gorman, C. Ritchie, L. Duizer and M. B. McSweeney, A Prospective Review of the Sensory Properties of Plant-Based Dairy and Meat Alternatives with a Focus on Texture, Foods, 2023, 12(8), 1709 CrossRef CAS PubMed.
- M. Z. Mahmoud, M. A. Fagiry, R. Davidson and W. K. Abdelbasset, The benefits, drawbacks, and potential future challenges of the most commonly used ultrasound-based hurdle combinations technologies in food preservation, J. Radiat. Res. Appl. Sci., 2022, 15(1), 206–212 Search PubMed.
- A. Yaya-González, J. Laika and Y. Peralta-Ruiz, Emerging nonthermal technologies for food safety: trends, limitations, and future research, Curr. Opin. Chem. Eng., 2025, 49, 101178 CrossRef.
- M. L. G. Monteiro, R. Deliza, E. T. Mársico, M. De Alcantara, I. P. L. De Castro and C. A. Conte-Junior, What Do Consumers Think About Foods Processed by Ultraviolet Radiation and Ultrasound?, Foods, 2022, 11(3), 434 CrossRef PubMed.
- K. Li, Z. L. Kang, Y. F. Zou, X. L. Xu and G. H. Zhou, Effect of ultrasound treatment on functional properties of reduced-salt chicken breast meat batter, J. Food Sci. Technol., 2015, 52(5), 2622–2633 CrossRef CAS PubMed.
- C. Zhang, M. Lu, C. Ai, H. Cao, J. Xiao and M. Imran, et al., Ultrasonic treatment combined with curdlan improves the gelation properties of low-salt Nemipterus virgatus surimi, Int. J. Biol. Macromol., 2023, 248, 125899 CrossRef CAS PubMed.
- X. Zhao, C. Zhou, X. Xu, X. Zeng and T. Xing, Ultrasound combined with carrageenan and curdlan addition improved the gelation properties of low-salt chicken meat paste, LWT–Food Sci. Technol., 2022, 172, 114230 CrossRef CAS.
- H. Tong, C. Cao, Y. Du, Y. Liu and W. Huang, Ultrasonic-assisted phosphate curing: a novel approach to improve curing rate and chicken meat quality, Int. J. Food Sci. Technol., 2022, 57(5), 2906–2917 Search PubMed.
- X. Yang, W. Lan and J. Xie, Ultrasound assisted treatment improves the preservation performance of chitosan-grafted-chlorogenic acid on refrigerated sea bass (Lateolabrax japonicus) fillets, J. Sci. Food Agric., 2023, 103(2), 900–907 Search PubMed.
- X. Gao, J. You, T. Yin, S. Xiong and R. Liu, Simultaneous effect of high intensity ultrasound power, time, and salt contents on gelling properties of silver carp surimi, Food Chem., 2023, 403, 134478 CrossRef CAS PubMed.
- H. Shi, I. Ali Khan, R. Zhang, Y. Zou, W. Xu and D. Wang, Evaluation of ultrasound-assisted L-histidine marination on beef M. semitendinosus: Insight into meat quality and actomyosin properties, Ultrason. Sonochem., 2022, 85, 105987 CrossRef CAS PubMed.
- S. Gu, Q. Zhu, Y. Zhou, J. Wan, L. Liu and Y. Zhou, et al., Effect of Ultrasound Combined with Glycerol-Mediated Low-Sodium Curing on the Quality and Protein Structure of Pork Tenderloin, Foods, 2022, 11(23), 3798 CrossRef CAS PubMed.
- J. Zheng, J. Qu, H. Peng, W. Chen and A. Hu, Application of ultrasound-assisted cryoprotectant impregnation for improving the storage quality of snakehead fish fillets, Food Sci. Technol. Int., 2023, 29(6), 641–649 CrossRef CAS PubMed.
- Q. Pan, G. Yang, Y. Wang, X. Wang, Y. Zhou and P. Li, et al., Application of ultrasound-assisted and tumbling dry-curing techniques for reduced-sodium bacon, J. Food Process. Preserv., 2020, 44(8), e14607 CAS.
- L. Guo, X. Xu, X. Zhang, Z. Chen, R. He and H. Ma, Application of simultaneous ultrasonic curing on pork (Longissimus dorsi): Mass transport of NaCl, physical characteristics, and microstructure, Ultrason. Sonochem., 2023, 92, 106267 CrossRef CAS PubMed.
- Y. He, Z. Zhao, Y. Wu, Z. Lu, C. Zhao and J. Xiao, et al., Effects of Quality Enhancement of Frozen Tuna Fillets Using Ultrasound-Assisted Salting: Physicochemical Properties, Histology, and Proteomics, Foods, 2024, 13(4), 525 CrossRef CAS PubMed.
- S. Xu, X. Guo, C. Fu, J. Wang, X. Meng and T. Hui, et al., Effect of ultrasound-assisted L-lysine treatment on pork meat quality and myofibrillar protein properties during postmortem aging, J. Food Sci., 2024, 89(7), 4162–4177 CrossRef CAS PubMed.
- Z. Wu, W. Ma, Z. Xian, Q. Liu, A. Hui and W. Zhang, The impact of quick-freezing methods on the quality, moisture distribution and microstructure of prepared ground pork during storage duration, Ultrason. Sonochem., 2021, 78, 105707 CrossRef CAS PubMed.
- Q. Sun, F. Sun, X. Xia, H. Xu and B. Kong, The comparison of ultrasound-assisted immersion freezing, air freezing and immersion freezing on the muscle quality and physicochemical properties of common carp (Cyprinus carpio) during freezing storage, Ultrason. Sonochem., 2019, 51, 281–291 CrossRef CAS PubMed.
- M. Zhang, X. Xia, Q. Liu, Q. Chen and B. Kong, Changes in microstructure, quality and water distribution of porcine longissimus muscles subjected to ultrasound-assisted immersion freezing during frozen storage, Meat Sci., 2019, 151, 24–32 CrossRef PubMed.
- Q. Sun, X. Zhao, C. Zhang, X. Xia, F. Sun and B. Kong, Ultrasound-assisted immersion freezing accelerates the freezing process and improves the quality of common carp (Cyprinus carpio) at different power levels, LWT–Food Sci. Technol., 2019, 108, 106–112 CrossRef CAS.
- C. Zhang, Q. Sun, Q. Chen, B. Kong and X. Diao, Effects of ultrasound-assisted immersion freezing on the muscle quality and physicochemical properties of chicken breast, Int. J. Refrig., 2020, 117, 247–255 CrossRef CAS.
- H. Cheng, C. Bian, H. Yu, J. Mei and J. Xie, Effect of ultrasound-assisted freezing combined with potassium alginate on the quality attributes and myofibril structure of large yellow croaker (Pseudosciaena crocea), LWT–Food Sci. Technol., 2022, 167, 113869 CrossRef CAS.
- C. Zhang, Y. Li, X. Xia, Q. Liu, Q. Chen and B. Kong, Changes in muscle quality and physicochemical characteristics of chicken breast subjected to ultrasound-assisted immersion freezing during long-term frozen storage, Int. J. Refrig., 2022, 142, 10–18 CrossRef CAS.
- Q. Sun, C. Zhang, Q. Li, X. Xia and B. Kong, Changes in functional properties of common carp (Cyprinus carpio) myofibrillar protein as affected by ultrasound-assisted freezing, J. Food Sci., 2020, 85(9), 2879–2888 CrossRef CAS PubMed.
- L. Cai, M. Cao, A. Cao, J. Regenstein, J. Li and R. Guan, Ultrasound or microwave vacuum thawing of red seabream (Pagrus major) fillets, Ultrason. Sonochem., 2018, 47, 122–132 CrossRef CAS PubMed.
- Z. Wu, W. Ma, S. J. Xue, A. Zhou, Q. Liu and A. Hui, et al., Ultrasound-assisted immersion thawing of prepared ground pork: Effects on thawing time, product quality, water distribution and microstructure, LWT–Food Sci. Technol., 2022, 163, 113599 CrossRef CAS.
- B. Wu, C. Qiu, Y. Guo, C. Zhang, X. Guo and Y. Bouhile, et al., Ultrasonic-assisted flowing water thawing of frozen beef with different frequency modes: Effects on thawing efficiency, quality characteristics and microstructure, Food Res. Int., 2022, 157, 111484 CrossRef PubMed.
- D. Kong, C. Quan, Q. Xi, R. Han, S. Koseki and P. Li, et al., Study on the quality and myofibrillar protein structure of chicken breasts during thawing of ultrasound-assisted slightly acidic electrolyzed water (SAEW), Ultrason. Sonochem., 2022, 88, 106105 CrossRef CAS PubMed.
- D. Kong, C. Quan, Q. Xi, R. Han, P. Li and Q. Du, et al., Effects of ultrasound-assisted slightly acidic electrolyzed water thawing on myofibrillar protein conformation and gel properties of chicken breasts, Food Chem., 2023, 404, 134738 CrossRef CAS PubMed.
- L. Cai, W. Zhang, A. Cao, M. Cao and J. Li, Effects of ultrasonics combined with far infrared or microwave thawing on protein denaturation and moisture migration of Sciaenops ocellatus (red drum), Ultrason. Sonochem., 2019, 55, 96–104 Search PubMed.
- H. Cheng, C. Bian, Y. Chu, J. Mei and J. Xie, Effects of Dual-Frequency Ultrasound-Assisted Thawing Technology on Thawing Rate, Quality Properties, and Microstructure of Large Yellow Croaker (Pseudosciaena crocea), Foods, 2022, 11(2), 226 Search PubMed.
- W. Lan, Y. Zhao, T. Gong, J. Mei and J. Xie, Effects of different thawing methods on the physicochemical changes, water migration and protein characteristic of frozen pompano (Trachinotus ovatus), J. Food Biochem., 2021, 45(8), e13826 Search PubMed.
- L. Cai, J. Wan, X. Li and J. Li, Effects of different thawing methods on physicochemical properties and structure of largemouth bass (Micropterus salmoides), J. Food Sci., 2020, 85(3), 582–591 Search PubMed.
- Y. Chu, M. Tan, C. Bian and J. Xie, Effect of ultrasonic thawing on the physicochemical properties, freshness, and protein-related properties of frozen large yellow croaker (Pseudosciaena crocea), J. Food Sci., 2022, 87(1), 52–67 Search PubMed.
- Y. Ma, X. Li, P. Sun and J. Li, Effect of ultrasonic thawing on gel properties of tuna myofibrillar proteins, J. Food Process. Preserv., 2022, 46(2), e16268 CAS.
- Y. Y. Wang, J. K. Yan, Y. Ding, M. T. Rashid and H. Ma, Effect of sweep frequency ultrasound and fixed frequency ultrasound thawing on gelling properties of myofibrillar protein from quick-frozen small yellow croaker and its possible mechanisms, LWT–Food Sci. Technol., 2021, 150, 111922 Search PubMed.
- C. Bian, H. Cheng, H. Yu, J. Mei and J. Xie, Effect of multi-frequency ultrasound assisted thawing on the quality of large yellow croaker (Larimichthys crocea), Ultrason. Sonochem., 2022, 82, 105907 CrossRef CAS PubMed.
- B. Wang, X. Bai, X. Du, N. Pan, S. Shi and X. Xia, Comparison of Effects from Ultrasound Thawing, Vacuum Thawing and Microwave Thawing on the Quality Properties and Oxidation of Porcine Longissimus Lumborum, Foods, 2022, 11(9), 1368 CrossRef PubMed.
- B. Wang, X. Du, B. Kong, Q. Liu, F. Li and N. Pan, et al., Effect of ultrasound thawing, vacuum thawing, and microwave thawing on gelling properties of protein from porcine longissimus dorsi, Ultrason. Sonochem., 2020, 64, 104860 CrossRef CAS PubMed.
- S. Gan, M. Zhang, A. S. Mujumdar and Q. Jiang, Effects of different thawing methods on quality of unfrozen meats, Int. J. Refrig., 2022, 134, 168–175 CrossRef CAS.
- M. Luo, K. Shan, M. Zhang, W. Ke, D. Zhao and Y. Nian, et al., Application of ultrasound treatment for improving the quality of infant meat puree, Ultrason. Sonochem., 2021, 80, 105831 CrossRef CAS PubMed.
- J. Zhang, W. Zhang, C. Ma and J. Cai, Evaluation of ultrasound-assisted process as an approach for improving the overall quality of unsmoked bacon, Ultrason. Sonochem., 2023, 98, 106490 CrossRef CAS PubMed.
- F. Zhang, H. Zhao, C. Cao, B. Kong, X. Xia and Q. Liu, Application of temperature-controlled ultrasound treatment and its potential to reduce phosphate content in frankfurter-type sausages by 50%, Ultrason. Sonochem., 2021, 71, 105379 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |