 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Identifying mechanistic differences between co-fed CO2 hydrogenation and reactive CO2 capture using Ru and Pd dual function materials
Chae Jeong-Potter†
 *,
Neha Mehra
*,
Neha Mehra ,
Carrie A. Farberow
,
Carrie A. Farberow and
Daniel A. Ruddy
and
Daniel A. Ruddy *
*
National Laboratory of the Rockies, Catalytic Carbon Transformation and Scale-Up Center, Golden, Colorado 80401, USA. E-mail: dan.ruddy@nlr.gov
First published on 30th January 2026
Abstract
Dual function materials (DFMs) enable reactive carbon capture (RCkeC), an intensified approach to carbon dioxide capture and utilization for cost and energy input reductions. Yet, there is a fundamental lack of understanding of mechanisms around CO2 adsorption and subsequent conversion on these materials, hindering further development. Herein, we investigated several supported alkaline metal oxides for their CO2 adsorption characteristics to find that Na/Al2O3 had the highest CO2 adsorption capacity, accompanied by a variety of CO2 adsorption geometries as identified by in situ DRIFTS and computational modeling. The addition of catalytic metals (Ru, Pd) increased the adsorption capacity of Na/Al2O3 without altering binding modes. In the subsequent reactive desorption step, acetate and formate intermediates were observed. Notably, this mechanistic investigation identified that the formation of acetate species was unique to RCC on a DFM, as these species were not observed in co-fed hydrogenation over the DFM or RCC over a Na-free catalyst.
Broader contextReactive carbon capture (RCC) seeks to address an overarching challenge in CO2 utilization approaches: typical carbon capture and utilization (CCU) technologies require large amounts of energy for the multiple steps of purification, compression, and transportation of CO2 after capture for conversion at a remote facility. To reduce the energy demand and break through current limitations of CCU, the RCC process instead employs dual function materials (DFMs) to enable capture and conversion in a single reactor. While recent literature has focused on the discovery of new DFM formulations and novel RCC processes, this study seeks to provide a comprehensive understanding of the key interactions between CO2 and DFM components, and how reactive desorption mechanisms are affected. Our findings reveal that RCC catalytic mechanisms cannot be directly extrapolated from traditional co-fed hydrogenation mechanisms, providing an important learning on how the scientific community should approach the design of next generation DFMs with improved performance. |
Introduction
The use of waste CO2 as a carbon feedstock to produce fuels and chemicals through CO2 recycling (i.e. capture and utilization, CCU)1 holds great potential as a means of chemical storage to combat intermittency of renewables, a pathway commonly dubbed “power-to-X”.2–4 However, typical CCU technologies require large amounts of energy for the multiple steps of purification, compression, and transportation of CO2 after capture for conversion at a remote facility.5 To reduce the energy demand and break through current limitations of CCU, multi-functional materials that allow capture and conversion to occur in a single reactor through a process dubbed reactive carbon capture (RCC) have been the topic of recent research.One such catalytic technology platform employs dual function materials (DFMs), which are composed of sorbents and catalysts co-dispersed on the same high surface area carrier. The sorbent enables selective capture of CO2 from a gas stream (including dilute CO2 concentrations) and the catalyst component subsequently converts the adsorbed CO2 upon introduction of a reactive gas (typically H2) in situ.6–8 The product from the most established DFM, comprised of Ru and/or Ni with an alkaline sorbent, is methane via the CO2 methanation reaction.9,10 While renewable methane would be an excellent transition fuel that can leverage currently available infrastructure, it is economically noncompetitive relative to inexpensive fossil methane (averaging $2.19 per MMBTU in 2024 in the US11). These challenges motivate the design and investigation of DFMs that enable CO2 capture and conversion to more valuable and more useful C1 products, like CO or methanol (average price of methanol was $35.66 per MMBTU in 202412). These products can be used in the chemical industry or upgraded to high energy density synthetic fuels for heavy duty vehicles, marine, and aviation.
A survey of DFM formulations reveals a variety of sorbent-catalyst combinations that can perform RCC to methane, CO, and/or methanol.8,13,14 In stark contrast to fundamental catalysis science and mechanistic studies of conventional co-fed CO2 hydrogenation reactions (which include the Sabatier reaction, CO2 + 4H2 → CH4; reverse water gas shift, CO2 + H2 → CO; and methanol synthesis, CO2 + 3H2 → CH3OH),15–20 there is a lack of depth in the fundamental understanding of how CO2 binds to DFMs, and importantly, how the subsequent mechanisms during reactive desorption are affected. Furthermore, recent explorations of novel DFMs to achieve more valuable C1 products like MeOH have shown that co-fed CO2 hydrogenation activity does not necessarily directly translate to RCC performance, suggesting that there are important mechanistic differences between the two modes of operation.21 Such differences were also suggested by Moreno Bravo et al., when studying Ru DFMs composed of rare earth oxides.22 While possible mechanisms over DFMs have been proposed,23–25 a systematic study is needed to understand: (1) the role of the alkali sorbent site in CO2 binding, (2) the subsequent effect of catalyst metal identity on the resulting transformation of the adsorbed CO2, and (3) the differences in adsorbed intermediates between the RCC and co-fed hydrogenation mechanisms.
Herein, we first characterized several Al2O3-supported sorbents for their capacity and CO2 binding geometries. We then characterized Ru and Pd DFMs with the same sorbent component (motivated by the breadth of RCC and co-fed CO2 hydrogenation literature around these two catalytic metals) to determine the effect of the metal catalyst on CO2 capacity and binding, followed by operando DRIFTS characterization of observable intermediates during simulated reactive desorption. Observed CO2 binding geometries and resulting intermediates were further verified through molecular modeling. Comparisons between co-fed hydrogenation over the Ru DFM and RCC on Na-free Ru/Al2O3 identified that the formation of acetate surface species was unique to RCC on the DFM.
Experimental
Material synthesis
Supported sorbents were prepared by introducing alkali (Na, K) and alkaline earth (Mg, Ca) metal oxides via incipient wetness impregnation of γ-Al2O3 (Puralox TH100/150, Sasol). Aqueous solutions of Na2CO3, K2CO3, Ca(NO3)2·4H2O, and Mg(NO3)2·6H2O salts (Sigma Aldrich) were used for impregnation onto γ-Al2O3 powder to achieve a metal![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al2O3 molar ratio of 0.202. These salts were chosen to be representative of synthesis routes used by many previous RCC reports.8,21,25–27 The impregnated powders were dried at 120 °C overnight (>12 h) in static air. Decomposition of the carbonate or nitrate precursors was performed using calcination at 400 °C for 4 h before characterization.
Al2O3 molar ratio of 0.202. These salts were chosen to be representative of synthesis routes used by many previous RCC reports.8,21,25–27 The impregnated powders were dried at 120 °C overnight (>12 h) in static air. Decomposition of the carbonate or nitrate precursors was performed using calcination at 400 °C for 4 h before characterization.
Ru and Pd DFMs were prepared by modifying Na/Al2O3 (prepared as described above) through incipient wetness impregnation using aqueous solutions of Ru(NO)(NO3)3 (Thermo Scientific Chemicals) and Pd(NO3)2·2H2O (Sigma Aldrich) with a catalytic metal to sorbent metal molar ratio of 0.02 (equivalent to ca. 2 wt% Ru or Pd). The resulting DFMs were then dried at 50 °C overnight in static air. The catalytic metals were activated through a pre-reduction at 300 °C for 4 h in H2 before characterization.
A non-DFM Ru/Al2O3 catalyst was prepared in the same manner by utilizing an aqueous solution of Ru(NO)(NO3)3 for impregnation of γ-Al2O3. The sample was dried at 120 °C overnight in static air and activated through a pre-reduction at 300 °C for 4 h in H2.
Characterization
CO2 and CO chemisorption were measured using an Autosorb-1C gas sorption analyzer (Quantachrome Instruments). Approximately 100–150 mg of material was diluted with 1 g of quartz chips in a u-shaped analysis tube. For CO2 chemisorption, sorbent materials were calcined at 400 °C in N2 for 8 h, and DFMs were reduced at 300 °C in H2 for 8 h. The sample was then evacuated for 8 h prior to analysis. CO2 chemisorption was measured at 100, 200, and 300 °C between 80 and 760 Torr in 50 Torr increments. The site density of strong CO2 chemisorption was determined by the difference of the total and weak isotherms, extrapolated to zero pressure. For CO chemisorption, DFMs were reduced at 300 °C in H2 for 4 h, followed by evacuation for 8 h prior to analysis. CO adsorption isotherms were measured at 40 °C between 80 and 760 Torr in 50 Torr increments. The site density of total CO uptake from the combined isotherm, extrapolated to zero pressure, was used to determine the average particle size of Ru and Pd. A metal (M) to CO binding stoichiometry, M:CO, of 1.5 was used to represent the known distribution of atop (M–CO, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and bridged (M–CO–M, 2
1) and bridged (M–CO–M, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) CO binding on these metals. The chemisorption data has an estimated error of ±4.1% based on the reproducibility error of the calibration standard provided by Quantachrome.
1) CO binding on these metals. The chemisorption data has an estimated error of ±4.1% based on the reproducibility error of the calibration standard provided by Quantachrome.
DRIFTS analysis
In situ and operando diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) experiments were performed using a Thermo Scientific Nicolet iS50R FT-IR spectrometer equipped with a Harrick Praying Mantis high temperature reaction chamber attachment with CaF2 windows. The DRIFTS spectra and backgrounds were collected at a resolution of 4 cm−1 with 128 scans. The samples were loaded into the sample holder on top of a small bed of quartz chips and pre-treated in situ prior to analysis. Al2O3-supported sorbents were calcined under flowing He for 4 h at 400 °C. DFMs and Al2O3-supported catalysts were reduced under flowing 50% H2/He for 4 h at 300 °C. Backgrounds were taken at 300 °C in He and 50% H2/He to be subtracted from the adsorption and reactive desorption steps, respectively. Flowrates were kept at 40 sccm and ramp rates at 5 °C min−1, unless otherwise specified.CO2 adsorption was conducted at 300 °C with 5% CO2/He for 30 min, with spectra collected in 5 min intervals. Spectra were also collected in 5 min intervals during a 1 h purge step under flowing He following the adsorption step. For operando analysis, reactive desorption was performed on DFMs and Al2O3 by switching to 50% H2/He for 1 h, with spectra again being collected in 5 min intervals. Effluent gas of the DRIFTS cell was analyzed using a downstream on-line FT-IR (600 HSC from California Analytical Instruments) to monitor product formation.
Operando co-fed hydrogenation was conducted in the DRIFTS cell at 300 °C with a gas mixture of 10% CO2, 40% H2, balance He, with a background spectrum collected in 40% H2/He. Spectra were collected after a 2-h hold at reaction conditions, after which CO2 flow was stopped. Spectra were then collected at 5 min intervals to monitor the evolution of surface species over 1 h. Effluent gas of the DRIFTS cell was again analyzed using a downstream on-line FT-IR to monitor product formation.
Computational methods
Density functional theory (DFT) calculations were implemented using the vienna ab initio simulation package (VASP).28–30 The generalized gradient approximation (GGA) through the Perdew–Burke–Ernzerhof (PBE) functional was used to approximate the exchange–correlation energy.31,32 Electron–ion interactions were captured by projector augmented-wave (PAW) potentials, with valence wavefunctions expanded in a plane-wave basis set with an energy cutoff of 500 eV.33 Gaussian smearing with a smearing width of 0.01 eV was employed. Spin-polarized calculations were carried out for systems involving radical adsorbates. Electronic relaxation was converged to a tolerance of 1 × 10−6 eV and the norm of force convergence was set to 0.03 eV Å−1. Atomistic construction of hydrated γ-Al2O3 surfaces denoted as Al2O3(110)2H2O and Al2O3(100)2H2O and Na-modified alumina are described in the SI.The adsorption energy (ΔEads,i) for adsorbate (i) is defined as
| ΔEads,i = Ei/slab − Eslab,r − Ei(g) | (1) |
Vibrational modes and harmonic frequencies of adsorbed species were obtained using perturbation theory without symmetry.35 Scaling factors were not applied because the calculated scaling value for CO2 was near unity (Table S8). Atomic simulations environment (ASE) was used to visualize and confirm the vibrational modes.36 Frequencies for carbonate (CO3*) and bicarbonate (HCO3*) species were computed by relaxing CO2Os and CO2OsHs atoms, where ‘s’ denotes surface atoms. Only adsorbate atoms were relaxed in vibrational calculations of CH3COO and HCOO; all substrate atoms were fixed. Dispersion corrections to energies were not applied as this analysis focuses on strongly chemisorbed species likely to persist at 300 °C for which binding is dictated by electrostatic interactions with the surface.
Born effective charges and the displacement vectors are used to determine the intensities (I) of infrared active vibrational modes. Intensities are calculated as
 | (2) |
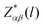 is the Born effective charge tensor of the l-th atom.37
is the Born effective charge tensor of the l-th atom.37
Results and discussion
Adsorption characteristics of Ru and Pd-based DFMs
Most DFMs employ group 1 alkali (G1-Alk) or group 2 alkaline earth (G2-Alk) metal oxides as their main sorbent component, which when dispersed on a high surface area oxide support (like Al2O3)38,39 offers an intermediate CO2 binding strength that retains CO2 on the surface during purge and heating steps of RCC processes (e.g., 200–400 °C) and is also reactive upon the introduction of H2. Prior literature teaches that adsorption capacity, strength, and the subsequent product, can vary dramatically depending on the identity of the sorbent metal.21,40 We began our investigation by comparing the adsorption characteristics of Al2O3-supported G1-Alk and G2-Alk metals, standardized by a Alk![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al2O3 molar ratio of 0.202, equivalent to ca. 10% CaO/Al2O3, a standard weight loading used in recent DFM reports (elemental analysis reported in Table S1). Chemisorption analysis with the in situ DRIFTS spectra for this series of sorbents (Fig. S1) revealed Na/Al2O3 has the highest gravimetric adsorption capacity, the greatest efficiency in terms of molar adsorption capacity, and a diversity of CO2 binding modes (Fig. S2). As previous RCC reports also commonly chose Na as the sorbent material,27,41–44 Na/Al2O3 was selected in this work as the most interesting candidate for a deeper spectroscopic study.
Al2O3 molar ratio of 0.202, equivalent to ca. 10% CaO/Al2O3, a standard weight loading used in recent DFM reports (elemental analysis reported in Table S1). Chemisorption analysis with the in situ DRIFTS spectra for this series of sorbents (Fig. S1) revealed Na/Al2O3 has the highest gravimetric adsorption capacity, the greatest efficiency in terms of molar adsorption capacity, and a diversity of CO2 binding modes (Fig. S2). As previous RCC reports also commonly chose Na as the sorbent material,27,41–44 Na/Al2O3 was selected in this work as the most interesting candidate for a deeper spectroscopic study.
It is noted that there is currently no clear agreement in DFM literature on the exact structure and phase of alumina-supported Na species; however, we are able to speculate that the majority of Na sites are well dispersed based on our chemisorption and DRIFTS data. First, following analysis by Keturakis et al., we believe to be at just below a monolayer of Na. Keturakis et al. ascribed 5% “Na2O” as the weight loading for monolayer formation on 200 m2 g−1 γ-Al2O3, resulting in a Na density of 5.11 Na atoms per nm2.39 Our DFMs contain 2.7 wt% Na (3.6% “Na2O”) on 142 m2 g−1 γ-Al2O3, resulting in 4.8 Na atoms per nm2. These materials are below the expected monolayer coverage, which would favor high dispersion of Na species after activation. We consider that Na is likely to exist in a combination of oxide or hydroxide forms (NaOx, NaAlOx, NaOH) and form surface carbonates and/or bicarbonates upon CO2 adsorption. Stoichiometrically, this would result in molar capacities (molCO2 molNa−1) ranging from 1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Na
1 Na![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2 binding, as in NaHCO3 or NaAlCO3(OH)2 formation) to 0.5 (2
CO2 binding, as in NaHCO3 or NaAlCO3(OH)2 formation) to 0.5 (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Na
1 Na![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2, as in Na2CO3 formation) in a perfectly dispersed material. Comparatively, Na/Al2O3 exhibited a strong chemisorption capacity of 0.22 molCO2 molNa−1 at 100 °C which we equate to between 22 and 44% dispersion, indicating that there may a portion of clustered, inaccessible Na sites or the existence of lower binding strength sites that do not strongly bind CO2 at 100 °C. Realistically, our DRIFTS data (Fig. S2c) at 300 °C showed a mixture of polydentate (Na:CO2 > 2) carbonates, conceptually favored by larger Na (or Na-O-Al) clusters, as well as bridged bidentate (Na:CO2 = 2) and chelating bidentate (Na:CO2 = 1) carbonates, favored by smaller particles. Thus, Na speciation is likely a mixture of well-dispersed oxides and hydroxides with some portion that is either inaccessible in clusters or binds CO2 too weakly.
CO2, as in Na2CO3 formation) in a perfectly dispersed material. Comparatively, Na/Al2O3 exhibited a strong chemisorption capacity of 0.22 molCO2 molNa−1 at 100 °C which we equate to between 22 and 44% dispersion, indicating that there may a portion of clustered, inaccessible Na sites or the existence of lower binding strength sites that do not strongly bind CO2 at 100 °C. Realistically, our DRIFTS data (Fig. S2c) at 300 °C showed a mixture of polydentate (Na:CO2 > 2) carbonates, conceptually favored by larger Na (or Na-O-Al) clusters, as well as bridged bidentate (Na:CO2 = 2) and chelating bidentate (Na:CO2 = 1) carbonates, favored by smaller particles. Thus, Na speciation is likely a mixture of well-dispersed oxides and hydroxides with some portion that is either inaccessible in clusters or binds CO2 too weakly.
Na/Al2O3 was modified with two representative CO2 hydrogenation catalysts, Ru and Pd. Ru is commonly used in DFM formulations to achieve methanation, showing excellent redox behavior (i.e., rapid reduction following oxidation) and long-term stability.26,27 Pd is a versatile metal that catalyzes reverse water gas shift (RWGS) and methanol formation from CO2, and we employ it here to study CO formation over a DFM.45–47 The catalysts were introduced via serial incipient wetness impregnation, targeting a molar ratio of catalyst![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sorbent metal of 0.1, resulting in ca. 2 wt% target loading (analyzed to be 2.3% Ru and 2.5% Pd). After reduction at 300 °C, CO chemisorption at 40 °C was employed to determine the average particle sizes. As expected, the low wt loadings resulted in small average particle sizes of 0.70 and 1.1 nm for Ru+Na/Al2O3 and Pd+Na/Al2O3, respectively. We note that DFMs for CO and MeOH synthesis based on Cu are exciting new additions to the RCC field,21 but these are less well understood compared to Ru- and Pd-based DFMs, and thus, are not the most suitable catalysts for the goals of this study.
sorbent metal of 0.1, resulting in ca. 2 wt% target loading (analyzed to be 2.3% Ru and 2.5% Pd). After reduction at 300 °C, CO chemisorption at 40 °C was employed to determine the average particle sizes. As expected, the low wt loadings resulted in small average particle sizes of 0.70 and 1.1 nm for Ru+Na/Al2O3 and Pd+Na/Al2O3, respectively. We note that DFMs for CO and MeOH synthesis based on Cu are exciting new additions to the RCC field,21 but these are less well understood compared to Ru- and Pd-based DFMs, and thus, are not the most suitable catalysts for the goals of this study.
The strongly chemisorbed CO2 on the resulting DFMs (termed, Ru+Na/Al2O3 and Pd+Na/Al2O3) were quantified over a range of temperatures (Fig. 1A). At all adsorption temperatures, the DFMs displayed enhanced adsorption capacity relative to the metal-free Na/Al2O3 (Fig. 1B). The Ru-DFM exhibited a greater increase in strong CO2 chemisorption which was attributed to two factors: (i) the reported behavior of Ru to promote the decomposition of Na2CO3 precursor salts during DFM pretreatment, activating more Na adsorption sites,25,48 and (ii) additional CO2 adsorption on Ru/RuOx during the capture step. Considering these two factors for the Pd-DFM, the more modest increase in CO2 adsorption capacity versus Na/Al2O3 may be caused by a combination of less effective participation of Pd to activate Na sites during pretreatmentrelative to Ru and a limited CO2 adsorption on Pd/PdOx surface species, which has been described to be nondissociative and weak.49,50
Characterization of CO2 binding geometries on Ru- and Pd-DFMs was performed through in situ DRIFTS. The resulting spectra after pre-treatment, CO2 exposure, and He purge are displayed in Fig. 2 with the spectrum after CO2 adsorption on Na/Al2O3 included for comparison. The broad absorbances observed were deconvoluted to make precise peak assignments, associate them to adsorption geometries (Table 1), and detect any perturbations resulting from the presence of a catalytic metal. We note that exact assignment between chelating and bridged bidentate carbonate geometries to the peaks fit at 1618/1311 cm−1 and 1669/1378 cm−1 on the Na/Al2O3 – which we will refer to as CO3*(B-I) and CO3*(B-II), respectively – is difficult as metrics for determining the strength of binding were found to be contradictory. For instance, CO3*(B-II), located at higher wavenumbers and showing the greatest decrease in intensity after an inert purge (Fig. S2), would be expected to correspond to a weaker bound bidentate geometry. Yet, the Δν3 splitting of the CO3* (B-II) species – a heuristic that can be used to determine the strength of surface carbonate binding – is lower at 291 cm−1 compared to 307 cm−1 of the CO3*(B-I) species, indicating a marginally stronger bound species. It is more conservative to conclude that these bidentate geometries are quite similar in binding strength and refrain from making exact assignments based on this dataset. Peaks at 1554 and 1340 cm−1 correlated to a polydentate geometry (CO3*(P)), and a small shoulder at 1755 cm−1 corresponded to Na2CO3 nanoparticles.39
| Surface species | Na/Al2O3 | Ru+Na/Al2O3 | Pd+Na/Al2O3 |
|---|---|---|---|
| CO3* (B-I) | 1618 cm−1 | 1614 cm−1 | 1614 cm−1 |
| 1311 cm−1 | 1320 cm−1 | 1305 cm−1 | |
| (Δν3 = 307 cm−1) | (Δν3 = 294 cm−1) | (Δν3 = 309 cm−1) | |
| CO3* (B-II) | 1669 cm−1 | 1659 cm−1 | 1657 cm−1 |
| 1378 cm−1 | 1371 cm−1 | 1367 cm−1 | |
| (Δν3 = 291 cm−1) | (Δν3 = 288 cm−1) | (Δν3 = 290 cm−1) | |
| CO3* (P) | 1554 cm−1 | 1559 cm−1 | 1549 cm−1 |
| 1340 cm−1 | 1349 cm−1 | 1330 cm−1 | |
| (Δν3 = 214 cm−1) | (Δν3 = 211 cm−1) | (Δν3 = 219 cm−1) | |
| Carbonyls | — | 1990 cm−1 | 2045 cm−1 |
| 1859 cm−1 | 1957 cm−1 | ||
| 1799 cm−1 |
Further understanding of potential CO2 binding geometries on Na/Al2O3 was obtained through DFT calculations. Na/Al2O3 models were constructed by adding a Na2O molecule to γ-Al2O3 surfaces (Al2O3(110)2H2O and Al2O3(100)2H2O, Fig. S4–S6, Table S2). We note that simulations of CO2 bound to Al2O3(110)2H2O and Al2O3(100)2H2O showed reasonable agreement between the computed IR spectra of various CO2 binding geometries and the observed spectra on γ-Al2O3 (Fig. S7 and Table S3). In the most stable configurations, Na2O on Al2O3(110)2H2O and Al2O3(100)2H2O transformed to Na2O(H) through migration of a H* from a surface Al-hydroxyl group (Fig. S8). Simulated IR spectra of unique CO2 adsorption geometries, varying in CO2 coordination to surface Na, Al, or to both Na and Al surface atoms on Na/Al2O3 models (Table S4), showed that various bicarbonate, bidentate, and polydentate species were well-aligned with the experimental spectrum (Fig. S9).
The spectra of both DFMs show broad absorbances at 1700–1500 cm−1 and 1400–1300 cm−1, similar to Na/Al2O3, with minor perturbations to the exact peak locations. On Ru+Na/Al2O3, the symmetric vibration of the CO3*(B-I) species (lower wavenumber peak) shifted, resulting in a Δν3 splitting value of 294 cm−1, compared to 307 cm−1 on the Na/Al2O3, indicating a marginally stronger carbonate-surface binding strength. CO3*(B-II) peaks both shifted to lower wavenumbers, with a Δν3 value of 288 cm−1 (291 cm−1 on Na/Al2O3). The CO3*(P) shifted to higher wavenumbers, yielding a similar Δν3 value of 211 cm−1 for Ru-DFM compared to 214 cm−1 for Na/Al2O3. The Ru-DFM also displayed strong interactions between Ru and carbonyl species, presumably formed from CO2 dissociation during the adsorption step, as evidenced by the complex feature between 2000 and 1750 cm−1. We attributed peaks observed at 1990 and 1799 cm−1 to a mixture of linear, di-, and bridged carbonyls on Ru.51 The additional peak at 1859 cm−1 could be attributed to a Ru carbonyl that is further stabilized by the presence of Na species. Such phenomenon has been reported with other noble metals (e.g., Au, Pd), where the presence of Na+ (and other alkali metal cations) induced red-shifted IR peaks of carbonyls associated with the noble metal site.52 We further confirmed this assignment using CO DRIFTS at 300 °C on Ru+Na/Al2O3 and Na-free Ru/Al2O3 (Fig. S10), which revealed a peak near 1850 cm−1 only on the Ru+Na/Al2O3 material.
For Pd+Na/Al2O3, no major perturbations were detected for the CO3*(B-I) peak locations (1614/1305 cm−1versus 1618/1311 cm−1 on Na/Al2O3), preserving a Δν3 value of 309 cm−1. A greater shift was observed on the CO3*(B-II) species to lower wavenumbers as also observed on the Ru-DFM. The CO3*(P) peaks shifted to lower wavenumbers as well; however, neither shifts associated with the two geometries resulted in significant differences to the splitting values. The Pd component of the DFM also exhibited interactions with in situ generated CO, as detected by peaks at 2045 and 1957 cm−1.53 Based on the low peak intensities, however, these Pd-(CO)x species are much less abundant than for Ru+Na/Al2O3, consistent with prior reports of low CO2 and CO binding energy on Pd.49,50
Reactive desorption over DFMs and sorbent-free catalysts
The subsequent transformation or decomposition of the surface carbonates during a simulated reactive desorption step (i.e., hydrogenation) of an RCC cycle were monitored using operando DRIFTS. Following the adsorption and purge steps as outlined in the previous section, 50% H2/He was introduced to the DRIFTS reactor at ambient pressure. Upon the introduction of H2 to the CO2-laden Ru+Na DFM (Fig. 3A), there was rapid consumption of the Ru-carbonyls, CO3*(B-II), and CO3*(B-I) species, correlated with a sharp rise in the signal for gas-phase CH4 in the reactor effluent (Fig. 3B). The carbonyl and carbonate species were almost fully consumed by the end of H2 exposure (1 h), and the Ru+Na DFM maintained selective production of CH4 without any CO observed. A peak at 1460 cm−1 also rapidly formed upon the introduction of H2, presumably associated with an intermediate species. While this species was largely consumed, some small fraction persisted at the end of H2 exposure, along with peaks at ∼1575 and ∼1350 cm−1. Based on literature reports for traditional CO2 methanation over a Ru/Al2O3 catalyst (i.e., not a DFM and not operated with a cyclic CO2–H2 feed), an Al2O3-associated formate (HCOO*) intermediate would be expected, exhibiting peaks at 1593, 1392, and 1375 cm−1.15 The observed peaks here do not align with these expected wavenumber positions, and with CO2 binding dominated at Na sites as indicated by the data in Fig. S2, we do not expect an HCOO*-Al intermediate to form under these reaction conditions. Instead, it may be possible that a formate is associated with the Na sites. Previous reports assign Na-formate with peaks at 1620–1567 and 1366–1377 cm−1, which would lie under the large peaks observed in our spectra, and are also in the range for polydentate surface carbonates noted above.54 Thus, an exact assignment for a Na-formate species is difficult. Further identification of the observed intermediate(s), including those associated with 1460 cm−1, was explored with computational modeling (vide infra). As mentioned, the peaks at 1575, 1350, and 1460 cm−1 persist at low intensities even after 1 h of reactive desorption, indicating that the Na sites that are occupied by the assocciated intermediates are not available for CO2 adsorption in subsequent cycles. This is in line with the larger DFM literature, which often show a decrease in CO2 adsorption capacity in the first few cycles. It is worth noting, however, that extended aging studies of Ru+Na DFMs have been well reported, showing no decrease in methanation activity over 50+ cycles under “clean CO2” adsorption conditions (i.e., no contaminants such as H2O and O2 present).27 We thus believe that the residual build up of intermediates observed here would neither cause cycle-to-cycle deactivation in the studied conditions nor hinder the mechanistic characterization presented in this work. | ||
| Fig. 3 DRIFTS spectra of reactive desorption following CO2 adsorption and inert purge (as seen in Fig. 2) on (A) Ru+Na/Al2O3 and (C) Pd+Na/Al2O3 with accompanying downstream product analysis, (B) and (D), respectively. Dark gray spectra were taken at the end of a 30 min He purge following the CO2 adsorption step; light gray spectra were taken following exposure to H2 in 5 min intervals; and blue spectra were taken after 1 h of continuous H2 exposure. All steps were carried out at 300 °C. | ||
The simulated reactive desorption step progressed similarly on Pd+Na/Al2O3, with the introduction of H2, there was a sharp and selective release of CO (Fig. 3C and D), correlated with the initial rapid consumption of each surface carbonate species. However, even after 1 h of H2 exposure, a large portion of the observed peak intensity for surface carbonates remained. It is expected that the kinetics for CO production are slow at this low temperature of 300 °C, since thermodynamics dictate that the RWGS reaction is generally operated above 400 °C. With the persistence of the carbonate species, it is difficult to clearly identify specific intermediate structures that may lie under those peaks. However, a broad peak around 1460 cm−1 appeared rapidly with the introduction of H2, indicative of a surface intermediate similar to that formed on the Ru+Na DFM. It has been proposed that the mechanism of CO2 hydrogenation on Pd/Al2O3 progresses through a CO* intermediate (derived from other intermediates such as formate47,53 or carboxyl45). Here, we observed that adsorption of CO on Pd is not favored (as evidenced by the low intensity of peaks between 2100 and 1900 cm−1), likely resulting in desorption of gaseous CO (the major product) rather than further hydrogenation of CO* to CH3OH or CH4.
On both DFMs, the peak at 1460 cm−1 may correlate best with a surface acetate intermediate (CH3COO*), reported to form by reaction between hydroxyl groups (OH*) and carbonyls (CO*), hydrogenation of CO* (to form H–C–O* intermediates), or between CO* and methyl groups (CH3*).55–57 Interestingly, acetate intermediates are more often associated with CO methanation or Fischer–Tropsch rather than CO2 methanation, which we also confirmed by comparing DRIFTS spectra during co-fed CO2 hydrogenation and CO hydrogenation over non-DFM Ru/Al2O3 (Fig. S11); importantly, a 1460 cm−1 peak was only observed during CO hydrogenation. To further confirm the assignment of an acetate species, DFT calculations were performed to compute infrared active bands of acetate and formate adsorbed on Na-free (Fig. S12, Table S5) and Na-modified hydrated alumina surfaces (Fig. S13, Table S6). Stable adsorbed acetate and formate species on Na/Al2O3(110)2H2O are shown in Fig. 4A–F. Acetates coordinated at Na, both Na and Al, and Al (Fig. 4A–C, respectively), all displayed vibrational signatures between 1400 and 1500 cm−1, corresponding well to the observed 1460 cm−1 peak. Among the computed species, Al-coordinated CH3COO* was the most stable (−1.99 eV, Table S6) and formed chelating bidentate motifs, exhibiting major peaks at 1530 and 1462 cm−1 associated with υas(OCO) vibration and υs(OCO) stretch mixed with CH3 bending, respectively. The bridged acetates coordinated to two surface Na sites and that coordinated to a Na and Al site exhibited peaks at 1448 and 1430 cm−1, respectively, indicating that there is likely a mixture of CH3COO* geometries and site coordination. In contrast, HCOO*—regardless of coordination and geometry—did not display bands in the 1400–1500 cm−1 range (Fig. 4D–F), further corroborating that the observed 1460 cm−1 peak in the DRIFTS spectrum can be assigned to a surface acetate. Formates did, however, exhibit peaks at ca. 1600 and ca. 1350 cm−1, corresponding well to the 1575 cm−1 and 1350 cm−1 envelopes observed in DRIFTS. HCOO* geometries also had adsorption energies similar to CH3COO* (Table S6), indicating that they would be stable on the DFM surface and suggesting that they are likely present in the observed DRIFTS spectra. It is worth mentioning that CO3*(P) species were also assigned to similar wavenumbers (1559/1349 cm−1); however, we conclude that their contributions are likely minor compared to that of the hydrogenated intermediates, given that their calculated adsorption energies are notably higher (−0.99 eV versus −1.99 eV, Table S4). In summary, experimentally observed envelopes at 1575, 1460, and 1350 cm−1 during reactive desorption correlated well with predicted IR active modes of adsorbed CH3COO* and HCOO*, verifying the formation and presence of these key intermediates during RCC.
As evidenced in the DRIFTS spectra, the Na sorbent sites in these DFMs dominate the interactions with CO2. To understand differences between surface species and reactive intermediates with and without Na sorbent sites, RCC on a Na-free Ru/Al2O3 catalyst was probed with DRIFTS (Fig. 5). After CO2 exposure (green spectrum, Fig. 5A), the DRIFTS spectrum exhibited a sharp peak at 2042 cm−1, which is assigned to linear atop CO* species on Ru.58 Notably, the peak associated with Na-stabilized Ru carbonyl species (1859 cm−1, Fig. 2B) was not observed, further supporting our initial assignment of this peak to the presence of Na species. Further, the complex envelope between 1650 and 1200 cm−1 was distinct in shape compared to that on the Ru+Na DFM, and indicated various Al2O3-carbonate structures: (i) a bicarbonate species (HCO3*) designated by peaks at 1648, 1439, and 1228 cm−1, (ii) a monodentate species indicated by a peak at 1520 cm−1, and (iii) a formate species, likely formed by interaction between bicarbonate species and residual H* on the catalyst surface persisting after pre-reduction, designated by peaks at 1590, 1392, and 1375 cm−1, all in accordance with reported peak positions for these surface species. After a He purge (dark gray spectrum, Fig. 5A), the intensity of all peaks decreased; in particular, the peaks associated with the bicarbonate species were no longer detectable, indicating complete desorption of this surface species that is critical to RCC performance. The remaining peaks correspond to formate and monodentate species.
 | ||
| Fig. 5 DRIFTS spectra of Ru/Al2O3 (A) after 30 min of CO2 exposure followed by a 1 h purge with He and (B) reactive desorption in H2 at 300 °C. | ||
Upon exposure to H2 (Fig. 5B), the carbonyl 2042 cm−1 peak rapidly decreased, and in its place, peaks at 2040, 2006, and 1970 cm−1 appeared. While there was a significant decrease in the intensities of these new peaks, 2040 and 1970 cm−1 features persisted even after 1 h of H2 exposure. According to a previous report by Wang et al., these peaks can be associated with geminal di-carbonyl species on low coordination Ru sites and are designated as a spectator species during CO2 methanation.15,59 Thus, the decrease in the peak intensities detected here can be described as a simple desorption of the carbonyl species, rather than consumption for conversion to CH4. The remaining features, associated with formate and monodentate carbonate species, were consumed almost to completion during the H2 exposure.
The geometries and activity of adsorbed COx species are considerably altered by the presence of Na sites. On the Ru+Na DFM, Na dominates as the primary adsorption site, offering strongly bound (but chemically reactive) bidentate and polydentate carbonate structures, compared to weakly bound bicarbonate and monodentate geometries on Al2O3 that desorb before reactive desorption begins. The presence of Na also impacts the distribution of carbonyl species, largely evidenced by a peak at ca. 1850 cm−1 that only appears on the Ru+Na DFM; notably, all carbonyls on the Ru+Na DFM are reactive and produce CH4, compared to the persistence of an unreactive geminal di-carbonyl on the Na-free Ru/Al2O3. Lastly, acetate emerges as a key intermediate in the presence of Na, compared to formates on a Na-free Ru/Al2O3, likely as a result of the different coverage and distribution of carbonyls on the two surfaces.
Ultimately, we are able to provide insight on the mobility of surface species in RCC. It is debated whether RCC mechanisms are driven by CO2 desorption/re-adsorption, CO3* migration, or H* migration. We hypothesize that migration of a variety of surface species may occur under these conditions. We observed that the formation of reactive hydrogenated intermediates (e.g., acetate, formate) is rapid and associated with the Na sorption site, likely driven by the migration of H* from Ru active sites to surface carbonates on the Na site. As proposed in co-fed CO2 hydrogenation, formates likely undergo hydrogenation to CO*, which migrate to Ru sites for further hydrogenation to CH4. Additionally, CO* and CHx* form acetates that can occupy diverse Na and Al site types and form a relatively more stable intermediate. These reported surface phenomena are coupled with no CO2 slip (i.e., no detection of gas phase CO2), further reducing the probability that CO2 is desorbed into the gas phase to be readsorbed to the surface for reaction (i.e., CO2 desorption/re-adsorption mechanism).
The migration of intermediates could also conceptually coincide with the migration of active phases (i.e., Ru and Na species). Based on the fact that water is formed during the CO2 hydrogenation reaction (co-fed or RCC), one would presume that Na species hydrate and dehydrate through the course of the reaction. The Na–O–Na dimer (as depicted in Fig. S8) may disperse to two adjacent NaOH species when hydrated and migrate to similar alumina surface sites. This migration would theoretically depend on the binding energy of NaOH to alumina and proximity of other alumina sites and may be aided by surface hydration, where Na+ moves from one bound water site to another with protons moving in the other direction. As this concept pertains to acetate/formate surface migration, similarly, one must consider binding energy, proximity of Na binding sites for migration, and species moving in the opposite direction for charge balance. Mechanistically, Na migration and acetate/formate migration are completely independent of each other. Na sites may be strongly bound to the alumina surface or may move with acetate/formate, and one would presume that protons are the charge compensating species moving in the other direction. Yet, regardless of whether the intermediates traverse in the bound state or in the ionic state, the ultimate result is that they must traverse close to a Ru site to be further reacted.
Proposed mechanistic differences between reactive desorption and co-fed hydrogenation over Ru+Na/Al2O3
To investigate potential differences in the reaction mechanisms of CO2 methanation during RCC cycles compared to co-fed hydrogenation, a mixed stream of CO2 and H2 was fed over Ru+Na/Al2O3 in the DRIFTS reactor cell. The DRIFTS spectra after 2 h of reaction at 300 °C and a CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2 ratio of 1
H2 ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 are displayed with the black solid line in Fig. 6A, and the corresponding effluent gas analysis is presented in the blue region of Fig. 6B. Several differences in surface species populations were notable in the spectrum after 2 h of co-fed reaction compared to RCC reactive desorption. In the co-fed condition, the carbonate envelopes were centered at 1604 and 1340 cm−1 (compared to 1650 and 1350 cm−1), indicating a different distribution of carbonate species during co-fed hydrogenation. Also, a set of triplet peaks between 3000 and 2600 cm−1 corresponded well with the C–H bond of a Na-formate species (NaOOCH).54 These triplet peaks were not observed in the previous reactive desorption experiment (comparison in Fig. S14), and coupled with the shift in the carbonate envelopes, formates appear to be abundant. The carbonyl region was also distinct with higher intensity peaks in the lower wavenumber region (1850–1750 cm−1), indicating a higher surface population of bridged carbonyls on Ru.
4 are displayed with the black solid line in Fig. 6A, and the corresponding effluent gas analysis is presented in the blue region of Fig. 6B. Several differences in surface species populations were notable in the spectrum after 2 h of co-fed reaction compared to RCC reactive desorption. In the co-fed condition, the carbonate envelopes were centered at 1604 and 1340 cm−1 (compared to 1650 and 1350 cm−1), indicating a different distribution of carbonate species during co-fed hydrogenation. Also, a set of triplet peaks between 3000 and 2600 cm−1 corresponded well with the C–H bond of a Na-formate species (NaOOCH).54 These triplet peaks were not observed in the previous reactive desorption experiment (comparison in Fig. S14), and coupled with the shift in the carbonate envelopes, formates appear to be abundant. The carbonyl region was also distinct with higher intensity peaks in the lower wavenumber region (1850–1750 cm−1), indicating a higher surface population of bridged carbonyls on Ru.
A particularly clear difference was the absence of a peak at 1460 cm−1, indicating that the corresponding acetate intermediate observed during reactive desorption was not formed/stabilized during co-fed hydrogenation. Importantly, the different surface speciation led to a different product distribution (Fig. 6B). During co-fed hydrogenation (blue region), a mix of CO and CH4 was detected, in stark contrast to the high selectivity to CH4 observed during RCC reactive desorption. After 2 h of co-fed CO2 and H2, the flow of CO2 was stopped, and the DRIFTS spectra and product evolution were continuously monitored under flowing H2 (Fig. 6A, gray spectra, Fig. 6B, yellow region). Upon the change in reactant gas flow, there was significant consumption of the surface species and a slight increase in intensity at 1460 cm−1 (zoom on region in Fig. S15), accompanied by a rapid decrease in the signals for CO2 and CO, and rapid, selective formation of CH4, mirroring the evolution of products during RCC reactive desorption.
The evolution of surface species during co-fed hydrogenation and reactive desorption over a Ru+Na/Al2O3 is summarized in Fig. 7. In co-fed CO2 methanation over Ru+Na/Al2O3, the Na component dominates interactions with CO2 (i.e., not Al2O3 as observed on Na-free Ru/Al2O3), wherein the CO2 was found to adsorb as surface carbonates (CO3*). The formation of HCOO* was also observed during the co-fed reaction, presumably as CO3* are hydrogenated by Ru-activated H*, following hypotheses that Al–HCO3* species are hydrogenated to HCOO*.15 A high surface coverage of Ru–CO* groups was observed, mostly in the linear and bridged configurations, and there was a notable absence of unreactive gem-dicarbonyls over this DFM.
 | ||
| Fig. 7 Schematic of surface species evolution in co-fed hydrogenation and reactive carbon capture over a Ru+Na/Al2O3 DFM. | ||
Under RCC conditions, CO2 also preferentially adsorbed onto Na sites as CO3* species. Additionally, it adsorbed as CO* on Ru sites, though in lower abundance compared to co-fed hydrogenation. Once H2 was introduced, CO* and some CO3* species were rapidly consumed. It is likely that the CO3* species were hydrogenated via H* from Ru, much like in co-fed hydrogenation, to form a formate intermediate. However, a distinct peak at 1460 cm−1 indicates the formation of an acetate intermediate, assigned through computational modeling and CO methanation experiments. This is likely formed between CHx* and CO* species as reported for CO methanation and based on our reaction conditions.55 The acetate species is consumed, albeit slowly, and forms methane with high selectivity. Falbo et al. reported that acetates on Ru/Al2O3 started decomposing at 250 °C, supplying CO* to the surface.55 Furthermore, Na-acetate has been reported to decompose to form methane in biomass hydrotreating applications.60 Thus, it is likely that the surface Na-acetate formed under RCC conditions also decomposes to form methane, given that the reactive desorption step is carried out at 300 °C. We propose that the formation of this acetate species contributes to the high methane selectivity in reactive desorption: in the co-fed environment, there is a constant population of CO* occupying the Ru sites which must also activate H2. To activate H2, some CO must desorb, resulting in the observed CO in the product stream, and thus, a lower CH4 selectivity. However, in RCC reactive desorption, the CO* on Ru is quickly reacted or drawn away to form acetate species, liberating Ru sites for H2 activation. The acetate species then serve as a temporary reservoir of CO* species for hydrogenation when more hydrogenating sites are available, accompanied by liberating an equivalent of CH4 when they decompose. The net result is no desorption of CO and high methane selectivity.
Conclusions
This work provides a comprehensive understanding of the key interactions between CO2 and DFM components – sorbent and catalytic metal – and how reactive desorption mechanisms proceed over a Ru+Na DFM. We first investigated the CO2 adsorption characteristics on several Al2O3-supported group 1 and group 2 metal oxides, which showed that Na/Al2O3 maintains the highest gravimetric and molar adsorption capacity over a range of flue gas temperatures and adsorbed CO2 in diverse geometries (bidentate, polydentate, bicarbonate). Catalytic metals (Ru, Pd) introduced to Na/Al2O3 generally increased adsorption capacity, with the greatest increase on Ru, and without major perturbation of adsorbed CO2 binding geometries on Na sites. In subsequent reactive desorption on the DFMs, Ru+Na/Al2O3 selectively produced CH4 while Pd+Na/Al2O3 selectivity produced CO, indicating tunability of product selectivity subject to catalytic metal. Through operando DRIFTS and computational modeling, formate and acetate species were identified as likely intermediates on both DFMs. Comparison of RCC over a Na-free Ru/Al2O3 showed that Na plays a key role in stabilizing carbonates and altering the distribution of carbonyls. Notably, (i) carbonates species bind stronger on Ru+Na/Al2O3, (ii) the acetate intermediate was only detected on Ru+Na/Al2O3, and (iii) unreactive geminal di-carbonyls were only formed on Ru/Al2O3. Finally, a bound acetate intermediate does not persist under co-fed conditions over Ru+Na/Al2O3, accompanied by lower selectivity to CH4 when compared to RCC. We posit that the acetate species during RCC draw CO* species away from Ru sites during RCC, which are required for hydrogen activation, leading to selective CH4 formation. In summary, we propose that the characteristics and relative populations of adsorbed COx species are affected by (1) the presence of strongly basic sites in DFMs when compared to sorbent-free catalysts, and (2) the step-wise introduction of CO2 and H2 compared to co-fed CO2 and H2. The unique synergy between the DFM materials properties and RCC process operation thus results in a reaction mechanism and catalytic performance that cannot be directly extrapolated from traditional co-fed hydrogenation literature.Conflicts of interest
There are no conflicts to declare.Note added after first publication
This article replaces the version published on 30th January 2026, to add the Acknowledgement section omitted from the PDF version during the production process.Data availability
The following data are included in the supplementary information (SI): metal content, CO2 chemisorption data, and DRIFTS analysis of adsorbed CO2 for sorbents; structural models of slabs and adsorbed CO2 used in computational analyses; computed vibrational spectra for adsorbed CO2 and related species; additional in situ DRIFTS data on DFMs with relevant time-dependent spectra; surface energies of various slab terminations. See DOI: https://doi.org/10.1039/d5ey00320b. All computational files have been made freely available at: C. Jeong-Potter, N. Mehra, C. A. Farberow and D. A. Ruddy, Identifying mechanistic differences between co-fed CO2 hydrogenation and reactive CO2 capture using Ru and Pd dual function materials, Materials Cloud Archive, 2026, 32, https://doi.org/10.24435/materialscloud:nr-g4.Acknowledgements
This work was authored by the National Laboratory of the Rockies (NLR) for the U.S. Department of Energy (DOE) under Contract No. DE-AC36-08GO28308. This work was supported by the Laboratory Directed Research and Development (LDRD) Program at NLR. A portion of this research was performed using computational resources sponsored by the U.S. Department of Energy's Office of Critical Minerals and Energy Innovation and located at NLR. The views expressed in the article do not necessarily represent the views of the DOE or the U.S. Government. The U.S. Government retains and the publisher, by accepting the article for publication, acknowledges that the U.S. Government retains a nonexclusive, paid-up, irrevocable, worldwide license to publish or reproduce the published form of this work, or allow others to do so, for U.S. Government purposes. The authors thank Dr W. Wilson McNeary (NLR) for his assistance with elemental analysis.References
- IPCC, in Special Report: Global Warming of 1.5 °C, 2018.
- S. Stucki, A. Schuler and M. Constantinescu, Int. J. Hydrogen Energy, 1995, 20, 653–663 Search PubMed.
- C. Graves, S. D. Ebbesen, M. Mogensen and K. S. Lackner, Renewable Sustainable Energy Rev., 2011, 15, 1–23 CrossRef CAS.
- F. V. Vázquez, J. Koponen, V. Ruuskanen, C. Bajamundi, A. Kosonen, P. Simell, J. Ahola, C. Frilund, J. Elfving, M. Reinikainen, N. Heikkinen, J. Kauppinen and P. Piermartini, J. CO2 Util., 2018, 28, 235–246 CrossRef.
- M. Fasihi, O. Efimova and C. Breyer, J. Cleaner Prod., 2019, 224, 957–980 Search PubMed.
- M. S. Duyar, M. A. Arellano and R. J. Farrauto, Appl. Catal., B, 2015, 168, 370–376 Search PubMed.
- M. S. Duyar, S. Wang, M. A. Arellano-treviño and R. J. Farrauto, J. CO2 Util., 2016, 15, 65–71 Search PubMed.
- I. S. Omodolor, H. O. Otor, J. A. Andonegui, B. J. Allen and A. C. Alba-Rubio, Ind. Eng. Chem. Res., 2020, 59, 17612–17631 Search PubMed.
- S. Wang, R. J. Farrauto, S. Karp, J. H. Jeon and E. T. Schrunk, J. CO2 Util., 2018, 27, 390–397 Search PubMed.
- M. A. Arellano-treviño, N. Kanani, C. W. Jeong-potter and R. J. Farrauto, Chem. Eng. J., 2019, 375, 121953 Search PubMed.
- U.S. Energy Information Administration, Henry Hub Natural Gas Spot Price, https://www.eia.gov/dnav/ng/hist/rngwhhdm.htm, (accessed 16 July 2025).
- Methanex, Historical Methanol Posted Price, https://www.methanex.com/our-products/about-methanol/pricing/, (accessed 16 July 2025).
- P. Melo Bravo and D. P. Debecker, Waste Disposal Sustainable Energy, 2019, 1, 53–65 CrossRef.
- S. B. Gharamaleki, T. R. Reina and M. S. Duyar, Prog. Energy, 2025, 7, 012001 CrossRef.
- X. Wang, Y. Hong, H. Shi and J. Szanyi, J. Catal., 2016, 343, 185–195 Search PubMed.
- M. A. Nolen, S. A. Tacey, S. Kwon and C. A. Farberow, Appl. Surf. Sci., 2023, 637, 157873 CrossRef CAS.
- M. A. Abir, R. E. Phillips, J. Z. M. Harrah and M. R. Ball, Catal. Sci. Technol., 2024, 14, 4506–4521 Search PubMed.
- C. Zhang, Z. Xiao, X. Guo, X. Tan, J. Gu, J. Li, G. Li, J.-J. Zou and D. Wang, Chem. Eng. J., 2025, 519, 165414 Search PubMed.
- R. Ye, J. Ding, T. R. Reina, M. S. Duyar, H. Li, W. Luo, R. Zhang, M. Fan, G. Feng, J. Sun and J. Liu, Nat. Synth, 2025, 4, 288–302 CrossRef CAS.
- A. J. Al Abdulghani, S. Ganguly, R. H. Hagmann, Z. Sun, M. Alvear, L. O. Mark, E. Nikolla, Y. J. Pagán-Torres and I. Hermans, J. Am. Chem. Soc., 2025, 147, 27438–27448 Search PubMed.
- C. Jeong-Potter, M. A. Arellano-Treviño, W. W. McNeary, A. J. Hill, D. A. Ruddy and A. T. To, EES. Catal., 2024, 2, 253–261 Search PubMed.
- L. Moreno Bravo, F. C. Meunier and J. Kopyscinski, Appl. Catal., B, 2025, 361, 124591 Search PubMed.
- L. Proaño, E. Tello, M. A. Arellano-Trevino, S. Wang, R. J. Farrauto and M. Cobo, Appl. Surf. Sci., 2019, 479, 25–30 Search PubMed.
- L. Proaño, M. A. Arellano-Treviño, R. J. Farrauto, M. Figueredo, C. Jeong-Potter and M. Cobo, Appl. Surf. Sci., 2020, 533, 147469 Search PubMed.
- S. Sapru, K. D. Hart, C. Zhou, G. Liccardo, J. Oh, M. J. Hollobaugh, J. Osio-Norgaard, A. Majumdar, B. D. Chandler and M. Cargnello, ACS Nano, 2025, 19, 2484–2496 Search PubMed.
- M. A. Arellano-Treviño, Z. He, M. C. Libby and R. J. Farrauto, J. CO2 Util., 2019, 31, 143–151 Search PubMed.
- C. Jeong-Potter, A. Porta, R. Matarrese, C. G. Visconti, L. Lietti and R. Farrauto, Appl. Catal., B, 2022, 310, 121294 Search PubMed.
- G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 49, 14251–14269 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 Search PubMed.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 Search PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 Search PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1997, 78, 1396 Search PubMed.
- P. E. Blöchl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef PubMed.
- K. Momma and F. Izumi, J. Appl. Crystallogr., 2008, 41, 653–658 CrossRef CAS.
- M. Gajdoš, K. Hummer, G. Kresse, J. Furthmüller and F. Bechstedt, Phys. Rev. B: Condens. Matter Mater. Phys., 2006, 73, 045112 Search PubMed.
- A. Hjorth Larsen, J. Jørgen Mortensen, J. Blomqvist, I. E. Castelli, R. Christensen, M. Dułak, J. Friis, M. N. Groves, B. Hammer, C. Hargus, E. D. Hermes, P. C. Jennings, P. Bjerre Jensen, J. Kermode, J. R. Kitchin, E. Leonhard Kolsbjerg, J. Kubal, K. Kaasbjerg, S. Lysgaard, J. Bergmann Maronsson, T. Maxson, T. Olsen, L. Pastewka, A. Peterson, C. Rostgaard, J. Schiøtz, O. Schütt, M. Strange, K. S. Thygesen, T. Vegge, L. Vilhelmsen, M. Walter, Z. Zeng and K. W. Jacobsen, J. Phys.: Condens. Matter, 2017, 29, 273002 Search PubMed.
- D. Karhánek, Self-Assembled Monolayers Studied by Density-Functional Theory, University of Vienna, 2010 Search PubMed.
- P. Gruene, A. G. Belova, T. M. Yegulalp, R. J. Farrauto and M. J. Castaldi, Ind. Eng. Chem. Res., 2011, 50, 4042–4049 Search PubMed.
- C. J. Keturakis, F. Ni, M. Spicer, M. G. Beaver, H. S. Caram and I. E. Wachs, ChemSusChem, 2014, 7, 3459–3466 CrossRef CAS PubMed.
- A. Porta, R. Matarrese, C. G. Visconti, L. Castoldi and L. Lietti, Ind. Eng. Chem. Res., 2021, 60, 6706–6718 Search PubMed.
- A. Bermejo-López, B. Pereda-Ayo, J. A. González-Marcos and J. R. González-Velasco, J. CO2 Util., 2019, 34, 576–587 CrossRef.
- L. Li, S. Miyazaki, S. Yasumura, K. W. Ting, T. Toyao, Z. Maeno and K. Shimizu, ACS Catal., 2022, 12, 2639–2650 Search PubMed.
- T. Sasayama, F. Kosaka, Y. Liu, T. Yamaguchi, S.-Y. Chen, T. Mochizuki, A. Urakawa and K. Kuramoto, J. CO2 Util., 2022, 60, 102049 Search PubMed.
- L. C. Wirner, F. Kosaka, T. Sasayama, Y. Liu, A. Urakawa and K. Kuramoto, Chem. Eng. J., 2023, 470, 144227 Search PubMed.
- N. C. Nelson, M.-T. Nguyen, V.-A. Glezakou, R. Rousseau and J. Szanyi, Nat. Catal., 2019, 2, 916–924 Search PubMed.
- X. Jiang, X. Nie, X. Guo, C. Song and J. G. Chen, Chem. Rev., 2020, 120, 7984–8034 Search PubMed.
- X.-G. Wang, L.-Y. Qiao, L.-Y. Xu, Y.-Y. Zeng, Z.-S. Huang, S.-S. Zong, J.-X. Wang, Z.-F. Zhou and Y.-G. Yao, Chem. Eng. J., 2024, 500, 156937 Search PubMed.
- C. Jeong-Potter, M. Abdallah, S. Kota and R. Farrauto, Ind. Eng. Chem. Res., 2022, 61, 10474–10482 Search PubMed.
- F. Solymosi and A. Barko, J. Catal., 1986, 101, 458–472 Search PubMed.
- M. Bowker, P. Stone, R. Bennett and N. Perkins, Surf. Sci., 2002, 497, 155–165 Search PubMed.
- S. Y. Chin, C. T. Williams and M. D. Amiridis, J. Phys. Chem. B, 2006, 110, 871–882 Search PubMed.
- L.-Y. Gan and Y.-J. Zhao, J. Chem. Phys., 2010, 133, 094703 CrossRef PubMed.
- X. Wang, H. Shi, J. H. Kwak and J. Szanyi, ACS Catal., 2015, 5, 6337–6349 Search PubMed.
- K. Ito and H. J. Bernstein, Can. J. Chem., 1956, 34, 170–178 Search PubMed.
- L. Falbo, C. G. Visconti, L. Lietti and J. Szanyi, Appl. Catal., B, 2019, 256, 117791 Search PubMed.
- A. de Klerk, Chapter 4: Fischer Tropsch Synthesis in Fischer–Tropsch Refining, 2011 Search PubMed.
- B. H. Davis, Catal. Today, 2009, 141, 25–33 CrossRef CAS.
- B. T. Loveless, C. Buda, M. Neurock and E. Iglesia, J. Am. Chem. Soc., 2013, 135, 6107–6121 CrossRef CAS PubMed.
- S. Xu, S. Chansai, S. Xu, C. E. Stere, Y. Jiao, S. Yang, C. Hardacre and X. Fan, ACS Catal., 2020, 10, 12828–12840 CrossRef CAS.
- J. A. Onwudili and P. T. Williams, Green Chem., 2010, 12, 2214 Search PubMed.
Footnote |
| † Present address: Department of Biomedical and Chemical Engineering, Syracuse University, Syracuse, New York, 13244, USA. E-mail: cjeongpo@syr.edu |
| This journal is © The Royal Society of Chemistry 2026 |




