Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Experimental assessment of granular activated carbon's effect on horizontal gene transfer
Hamed Rasouli Sadabad *ad,
James S. G. Dooley
*ad,
James S. G. Dooley b,
Heather M. Colemanc,
Barry O'Haganb,
William J. Snelling
b,
Heather M. Colemanc,
Barry O'Haganb,
William J. Snelling b and
Joerg Arnscheidt*a
b and
Joerg Arnscheidt*a
aSchool of Geography and Environmental Sciences, Ulster University, BT52 1SA, UK. E-mail: h.rasoulisadabad@ulster.ac.uk; j.arnscheidt@ulster.ac.uk
bSchool of Biomedical Sciences, Ulster University, BT52 1SA, UK
cSchool of Pharmacy and Pharmaceutical Sciences, Ulster University, BT52 1SA, UK
dBelfast School of Architecture and the Built Environment, Ulster University, BT15 1ED, UK
First published on 12th March 2026
Abstract
With the total number of annual deaths related to antimicrobial resistance estimated to reach ten million globally by 2050, it is essential to evaluate the impact of every process such as adsorption on horizontal gene transfer. Activated carbon is the most frequently applied adsorbent for elimination of a broad range of chemicals like antibiotics from water. The current study assessed the influence of three types of granular activated carbons (GACs) on the transfer frequency of vancomycin resistance genes from donor to recipient Enterococcus faecalis strains. The bacterial attachment on the GAC surfaces was evaluated using scanning electron microscope (SEM) imagery, and a method was developed to correlate bacterial spatial proximity and cell-to-cell contacts on the GAC surface with gene transfer frequency in SEM micrographs. The results indicated that regardless of different surface properties, all studied GACs supported bacterial attachment and facilitated horizontal gene transfer. For example, loading 0.5 g of Filtrasorb-400 into 5 mL growth media increased the gene transfer frequency by more than 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 times, from 3.33 × 10−9 (in the bulk) to 9.55 × 10−5. The response patterns of gene transfer frequency between bacteria and GAC dose were similar across GAC types but varied in amplitude. The normalized values of cell-to-cell contacts on the number of bacteria on a unit surface area of activated carbon showed a high correlation with its observed gene transfer frequency. Findings of this research emphasized the importance of considering adsorbents' (e.g. activated carbon) contribution in stimulation of horizontal gene transfer, along with operational parameters in designing water treatment processes.
700 times, from 3.33 × 10−9 (in the bulk) to 9.55 × 10−5. The response patterns of gene transfer frequency between bacteria and GAC dose were similar across GAC types but varied in amplitude. The normalized values of cell-to-cell contacts on the number of bacteria on a unit surface area of activated carbon showed a high correlation with its observed gene transfer frequency. Findings of this research emphasized the importance of considering adsorbents' (e.g. activated carbon) contribution in stimulation of horizontal gene transfer, along with operational parameters in designing water treatment processes.
Water impactGranular activated carbon is widely used in drinking water treatment due to its unique characteristics. However, this study shows it can strongly promote the transfer of antibiotic resistance genes between bacteria during water treatment processes. Findings of this research emphasize the need to consider horizontal gene transfer, alongside operational parameters in designing adsorption process for water treatment systems and contaminant removal. |
1. Introduction
The World Health Organization has identified antimicrobial resistance and its spread as one of the top threats to global public health.1 Emergence and dissemination of antimicrobial resistance have raised global attention in various sectors, especially in water treatment processes, purification plants, storage, and distribution systems due to their close relation to public biosafety.2 It is estimated that 4.95 million deaths occurred in 2019 globally due to antimicrobial resistance, and without a significant strategy to stop the current trend, it is likely to approach ten million annual deaths by 2050,3,4 highlighting the importance of investigating horizontal gene transfer in all processes related to public health. Importantly, conjugative transfer, as the predominant mechanism for dissemination of antibiotic resistance genes in environment strictly depends on direct physical contact between donor and recipient cells as plasmids and other mobile genetic elements are transferred through cell-to-cell interaction.2,5–7 Therefore any process like adsorption that enhance bacterial aggregation, surface attachment, or spatial proximity may substantially increase the probability of effective plasmid transfer and allows for the rapid dissemination of antibiotic-resistant bacteria in the environment.8Adsorption, as a stand-alone process or in combination with other processes, is widely applied in water treatment due to its versatility in removing a wide variety of compounds including pharmaceuticals, chemicals and trace metal ions even at extremely low concentrations i.e. μg L−1 or ng L−1.9–11 Activated carbon as the most frequently applied adsorbent is an ideal choice for elimination of a broad range of chemicals like antibiotics in various sectors including pharmaceutical industries, water and wastewater treatment plants,12 and medical applications like gastrointestinal decontamination.13–18 Activated carbon consists of significant roughness and a huge surface area compared with other adsorbents, turning it into a desirable substance for many microorganisms, such as bacteria, some of which can easily attach to its surface and form a biofilm.2,19 These characteristics of activated carbon have benefited different water treatment processes in biodegradation or removal of various contaminants via biological activated carbon.2,19,20 However, the high density of biomass and the possibly high nutrient content on biological activated carbon provide an ideal environment for microbial growth.2 Thus, it is a medium that can concentrate antibiotic resistance genes on its surface, which is likely to facilitate the horizontal gene transfer and dissemination of antibiotic-resistant bacteria after activated carbon filter treatment.2
Since activated carbon filtration is frequently used in drinking water purification plants with an upward trend,21 its contribution to horizontal gene transfer may affect public drinking water safety. This is important to consider because bacterial leakage (bio-leakage) from activated carbon filters has been reported by various studies, and the presence of bacteria (including pathogens) has been detected in filter effluent, even if the filter is integrated into a disinfection unit.21,22 Yu et al. (2019) have reported that leakage of Enterococcus faecalis (E. faecalis) from activated carbon filters into the effluent ranged 8–18% depending on ambient liquid's ionic strength values.21 This phenomenon is likely to involve the deposition and migration of bacteria on activated carbon, as has also been observed on other porous solid materials such as quartz sand, soil, and glass beads.21,23–25
Bio-leakage from filters in water treatment represents a potential source of human exposure to fecal bacteria, compounded by antibiotic resistance and in 2012, unsafe drinking water, along with a lack of sanitation and personal hygiene, was reported as the main cause of 88% of diarrheal diseases and 1.7 million deaths globally.21
Other than water treatment-related applications, activated carbon in edible and medical grades is utilized for elimination of compounds, controlling the overdose poisoning and removing gut microbiota-associated problems.13,14 Coincidence of the targeted contaminants in the gut microbiota besides the present bacteria may lead to facilitating horizontal gene transfer in presence of activated carbon.13,14,26–28
With the significantly increasing number of annual deaths due to antimicrobial resistance,3,4 it is crucial to evaluate the impact of any process on horizontal gene transfer. Currently, there is a notable research gap on understanding the direct contribution of activated carbon on facilitating horizontal gene transfer and majority of the related research has been conducted on macro-assessments, e.g. measuring the frequency of antibiotic resistance genes (ARG) before and after treatment processes rather than focusing on the fine details. The novel aspect of this study is to experimentally evaluate the potential stimulation of horizontal gene transfer by granular activated carbon (GAC), with particular attention on bacterial attachment conditions, aggregations and spatial densities on its surface, as an attempt to tighten the knowledge gap in the micro-scale assessment of this phenomenon. To conduct the study, various doses of different activated carbons were introduced into the mixture of donor and recipient E. faecalis strains, and their gene transfer frequencies were determined and analyzed. After confirming the contribution of activated carbon in enhancing gene transfer frequency, the bacterial attachments on its surface were examined using scanning electron microscopy (SEM), both qualitatively and quantitatively. During this study, reliable and replicable methods were developed to detach bacteria from the GAC surface (for gene transfer studies) and to fix the cells on GAC surfaces for SEM analysis with minimized defects on them.
2. Materials and methods
2.1. Chemicals
Tryptic soy agar (TSA), tryptic soy broth (TSB), and phosphate-buffered saline (PBS) were obtained from Thermo Scientific (UK) and prepared as described in their manual and autoclaved (121 °C, 15 min) before use. Chemicals, including methanol, ethanol, and acetone were purchased from Fisher Chemical (UK). Deionized water was used to prepare all aqueous solutions (PURELAB Option, ELGA).2.2. Granular activated carbon (GAC) types
Three types of GAC were selected based on the differences in their attributes and adsorption performance according to the authors' previous research, which comprehensively studied the characteristics of various GACs and their performances in the removal of antibiotics from aqueous environments.29 GACs were kindly supplied by their manufacturers upon request (Filtrasorb-400 by Chemviron, Belgium, Norit-PK1-3, and Norit-RB4W by Cabot Norit, Netherlands). The selected GACs were different in their relevant material properties like surface morphology, specific surface area, pore characteristics, point of zero charge pH (pHpzc), and functional surface groups. Filtrasorb-400 possesses higher mesopore volume, mesopore width, and specific surface area than the other two GACs. Norit-RB4W has intermediate values for these parameters, while Norit-PK1-3 has the lowest values.29 After rinsing in deionized water and oven drying at 105 ± 5 °C for 72 h, GACs were stored in a desiccator until the time of use.2.3. Bacterial strains
In this study, two strains of E. faecalis were used as the donor (MF06036Van) and the recipient (MW01105Rif) to assess horizontal gene transfer by conjugation. The donor strain was susceptible to rifampicin, but resistant to vancomycin due to containing vancomycin resistance genes (vanA). In contrast, the recipient strain was susceptible to vancomycin and resistant to rifampicin. The different resistance profiles of the strains enabled tracking the transfer of the vanA gene from donors to recipients and generating transconjugants, as donors were resistant against vancomycin, recipients were resistant against rifampicin and transconjugants (as product of horizontal gene transfer) were resistant to both vancomycin and rifampicin.It is reported that rifampicin-resistant strains of E. faecalis are able to receive vancomycin-resistance genes via pheromone-inducible conjugation in aqueous environments and can thus become multi-resistant.30,31 Vancomycin-resistant enterococci (VRE) have been considered a serious threat by the US Department of Health and Human Services,32 and infections associated with them cause a high mortality rate and longer hospital stays, measured by the number of days spent in the hospital which leads to higher healthcare costs.33 It has been reported that substantial numbers of enterococci isolates from clinical patients were resistant to vancomycin (approximately 30% in the US and 20% in Europe and Africa) in 2019.34 These organisms are able to survive in dry hospital environments for more than seven days,35 and can be extensively transmitted by proximate surfaces.36
During the project, the strains were kept in 50% glycerol in 2.0 mL cryogenic tubes (Thermo Scientific, UK), at −80 ± 2 °C for the duration of the experiment. Before conducting the experiments, the minimum inhibitory concentration (MIC) of each strain was determined. The regrowth of overnight bacterial cultures in TSB at 37.0 ± 0.5 °C was used to initiate conjugation assays. For each experiment, 5 mL of autoclaved TSB was inoculated with a specific strain and left in an incubator (37.0 ± 0.5 °C) for overnight growth (18–24 h). Then, 1.0 mL of the overnight growth was diluted 10 times by autoclaved TSB (9.0 mL) in a universal tube and incubated (37.0 ± 0.5 °C) for 240 ± 15 min to achieve a bacterial population in exponential growth phase. Then, the concentration of cells in the culture was adjusted to approximately 1.0 × 109 CFU mL−1, corresponding to an optical density of 0.12 ± 0.01 at 600 nm (OD600). This protocol was the starting point for all experiments.
2.4. Viable colony counting method for determining the concentration of bacteria
Viable colony counting was used in this study to evaluate the concentration of cells in different conditions by the heterotrophic plate counting (HPC) method. To carry out the measurement, 100 μL of the sample was serially ten-fold diluted. For each dilution step, 100 μL of bacterial suspension was transferred into 900 μL of PBS in Eppendorf tubes. Counting involved 100 μL subsamples of suitable dilutions spotted (5 spots per each disk, 20 μL per spot) onto standard petri dish plates (100 mm × 15 mm) containing suitable agar media (e.g. antibiotic containing TSA). Then the plates were incubated at 37.0 ± 0.5 °C for approximately 24 h. Fig. S1 illustrates the schematic protocol of this method for counting viable colonies.Only results from plates with colony counts between 30 and 300 were considered in calculations.37 The concentration of bacteria in the liquid media was calculated using the attained number of viable colonies on the plate via eqn (1), as follows.
| Bacteria concentration, CFU mL−1 = (number of viable colonies (CFU) × dilution factor)/(volume of spotted liquid (mL)) | (1) |
2.5. Effect of activated carbon on conjugation
Conjugation studies were carried out in the presence of different doses of three dissimilar types of GAC to assess its possible contribution to horizontal gene transfer between MF06036Van (donor) and MW01105Rif (recipient) strains, schematically shown in Fig. S2.To carry out conjugation assays, 1.0 mL of overnight culture of each strain was added to 9.0 mL of TSB and incubated for 4 h at 37.0 ± 0.5 °C to obtain regrowth and fresh cultures. Then 9.0 mL of the regrowth of MW01105Rif (recipients) and 1.0 mL of MF06036Van (donors) were added to a universal tube and vortexed to obtain mixed strains. The previous research in the School of Biomedical Sciences of Ulster University revealed that the highest efficiency of vanA gene transfer from MF06036Van (donor) to MW01105Rif (recipient) via conjugation occurs when the volumetric ratio of the recipient strain to the donor is adjusted on 9 to 1.38,39 The aliquots of 0.5 mL of the mixed cultures were added to 4.5 mL of TSB in universal tubes, vortexed, and then the defined doses of activated carbon samples were added to them in separate universal tubes. For each type of activated carbon, these doses were considered: 0.1, 0.25, 0.50, 0.75, 1.0, 1.5 and 2.0 g per 5 mL of liquid medium. The selected dosage range was planned to systematically evaluate dose-dependent effect of GACs on gene transfer frequency in various scenarios covering low to high loadings of activated carbon in aqueous environment. Loading more than 2.0 g GAC per 5 mL media would leave some parts of adsorbent granules untouched by the liquid media. This range enabled identification of the full rise-and-fall pattern and detection of peak value for transfer frequency. A control sample containing no activated carbon was also considered in each experiment. The prepared samples were shaken at 100 rpm in the incubator for 24 h at 37.0 ± 0.5 °C. After incubation, the bacteria were detached from the activated carbon surface. For this purpose, solid particles were separated from the liquid phase and immersed in 15 mL of 1% Tween-80 in PBS solution and vortexed at 2500 rpm for 4 min.
Detaching bacteria from activated carbon was developed based on previous reports in the literature and optimized to separate enterococci from solid surfaces into to the liquid phase with the lowest possible harm to the viable organisms.40,41 In addition, the impacts of 1.0% Tween-80 in PBS solution as well as vortexing at 2500 rpm on MF06036Van (donor), MW01105Rif (recipient) and transconjugants were assessed to ensure that they had no significant effect on the number of their viable-colony-forming units.
Then 100 μL of the supernatant was used for determining the concentrations of donor, recipient, and transconjugant bacteria by viable colony counting on four different groups of plates as below:
- TSA plate containing 10 μg mL−1 vancomycin and 100 μg mL−1 rifampicin: to determine the number of transconjugants.
- TSA plate containing 10 μg mL−1 vancomycin: to determine the number of MF06036Van (donor) plus transconjugants.
- TSA plate containing 100 μg mL−1 rifampicin: to determine the number of MW01105Rif (recipient) plus transconjugants.
- TSA plate containing no antibiotic: to check if viability of the strains if there is no growth on the other plate.
Then, the results of colony counting on each plate were converted to a viable cell content of activated carbon particles (CFU g−1) by eqn (2).
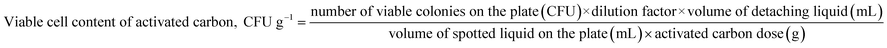 | (2) |
| Gene Transfer Frequency = Transconjugant content of activated carbon, CFU g−1/Recipients content of activated carbon, CFU g−1 | (3) |
2.6. Sample preparation for scanning electron microscopy (SEM)
To approach a profound understanding of the contribution of GAC in horizontal gene transfer, GAC granules containing bacteria were sampled and analyzed by SEM. Sample preparation for SEM analysis was carried out in several steps, as follows (Fig. S3).- Preparing activated carbon particles containing bacteria on their surface: similar to the conjugation study, 1.0 mL aliquots of an overnight culture of MF06036Van (donor) and MW01105Rif (recipient) strains were added to 9.0 mL of TSB, individually and incubated for 4 h at 37.0 ± 0.5 °C to obtain regrowth and fresh cultures. Subsequently, 9.0 mL of the regrowth of MW01105Rif and 1.0 mL of MF06036Van were added to a universal tube and vortexed to obtain a mix of the strains. Then, an aliquot of 0.5 mL of the mixed culture was added to 4.5 mL of TSB in universal tubes, vortexed, and added to defined doses of activated carbon samples in separate universal tubes. A control sample containing no activated carbon was produced. Prepared samples were shaken at 100 rpm in an incubator for 24 h at 37.0 ± 0.5 °C. After incubation, activated carbon particles were separated from the liquid phase. Since the GAC particles were in granular shape, they were separated from their suspensions via laboratory tweezers.
- Fixing bacteria on activated carbon surface: according to the scanning electron microscope (SEM) working mechanism, the samples must be free from moisture and covered by a super-thin electrically conductive coating.42–44 To avoid washing bacteria off from activated carbon particles' surface as well as their possible deformation during dehydration, they must be fixed. To fix bacteria, activated carbon granules were immersed in 4.0% glutaraldehyde solution in PBS for 24 h at 4 °C. Then, the particles were immersed in PBS for 5 minutes at ambient temperature to be washed.44
- Dehydration: activated carbon particles with fixed bacteria were dehydrated by serial immersion in 0, 25, 50, 75, and 100% ethanol solutions in PBS. The particles were immersed for 15 min in each solution. The outcome of 100% ethanol was immersed in acetone for 15 min to remove the remaining ethanol from solid particles' structure and left to be air dried for 15 min at ambient temperature.
- Covering the samples with a conductive layer: since bacteria and the products of their reaction with glutaraldehyde are not conductive they were covered by a gold sputter coating before the SEM analysis to avoid the creation of negative charges and electrostatic repulsion. The samples were kept in aseptic, dust-free, and tightly closed boxes until to be analysed by SEM.
2.7. Statistical analysis, image processing, and plotting graphs
All tests were carried out in triplicate within three independent experiments, and the statistical analyses of results were conducted via GraphPad Prism 10.0. The normal distribution of samples was analyzed by the Shapiro–Wilk test. Since the resulting data were not normally distributed and there were several groups of samples in each experiment (e.g., 10 series of samples in the effect of GAC dose study), the Kruskal–Wallis was used as a non-parametric test.45,46 The significance threshold was set to P ≤ 0.05. In the cases of significant differences, Dunn's test was carried out as a post hoc pair-wise test. For quantitative analysis of SEM micrographs and to study whether the micrographs, taken from various granules (as one group) and several points of a random granule (as another group) are significant, Mann–Whitney U test was utilized.The graphs were plotted by GraphPad Prism 10.0. SEM micrographs were processed by ImageJ, as an open source Java-based image processing program. The schematics are prepared using BioRender-Scientific Image and Illustration Software.
3. Results
3.1. Effect of activated carbon dosage on conjugation
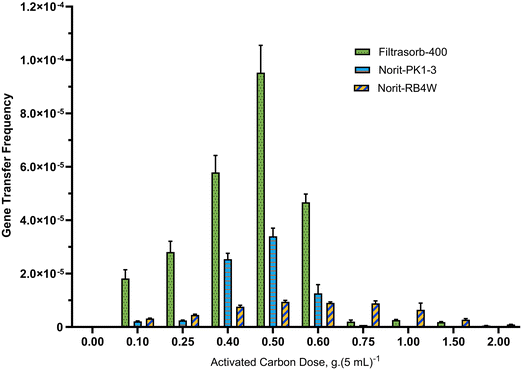
Gene transfer frequencies of multiple samples containing doses in the range 0.1 to 2.0 g of the three tested GACs were evaluated and the obtained results are shown in Fig. 1. The control in the graphs contained no activated carbon. All tested GACs augmented the conjugation process and led to a significant increase in gene transfer frequency between the studied strains (Fig. 1(a)–(c)), and the observed change in the magnitude of gene transfer frequency was associated with the dosage of activated carbon.As shown in Fig. 1, loading 0.1 g Filtrasorb-400 into 5 mL medium augmented the frequency of vancomycin-resistant genes from the donor to the recipient strain by more than 4350 times (i.e., from 3.33 × 10−9 in the control sample to 1.45 × 10−5). Further increase of the GAC dosage resulted in a remarkable ascent with the peak value of 9.55 × 10−5 for 0.5 g of activated carbon in 5 mL of media, which exceeded the result for the control sample by factor 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700. A sharp decline in gene transfer frequency was observed for a further augmentation of the GAC dosage from 9.55 × 10−5 in 0.5 g GAC per 5 mL to 1.7 × 10−6 in 0.75 g GAC per 5 mL of media without significant changes in samples containing larger GAC doses.
700. A sharp decline in gene transfer frequency was observed for a further augmentation of the GAC dosage from 9.55 × 10−5 in 0.5 g GAC per 5 mL to 1.7 × 10−6 in 0.75 g GAC per 5 mL of media without significant changes in samples containing larger GAC doses.
A similar pattern with a rise and fall in horizontal gene transfer frequency as dosage increased was observed for Norit-PK1-3, even though the domains of influence were slightly different. Exposing 0.1 g Norit-PK1-3 to 5 mL media increased the dissemination of vancomycin-resistant gene by factor 637 to 2.12 × 10−6 compared to the control sample (3.33 × 10−9), illustrated by Fig. 1(b).
A significant change in conjugation was also observed for adding Norit-RB4W to the media, but the observed changes were more gradual with increasing of GAC dosage. Addition of 0.1 g Norit-RB4W into 5 mL medium increased gene transfer frequency from 3.33 × 10−9 (in control sample) to 3.16 × 10−6; an upward trend in values of gene transfer frequency was observed up to 0.5 g GAC in 5 mL approaching 9.45 × 10−6, as the peak value with an insignificant difference for dosage 0.75 g GAC in 5 mL, followed by a gradual downward trend to 9.39 × 10−7 in 2.0 g GAC in 5 mL (Fig. 1(c)).
For comparison between tested GAC types, Fig. 2 shows all observed gene transfer frequencies of conjugation between the donor and recipient strains in a combined graph. It illustrates that the magnitude of the observed impact on gene transfer frequency varied substantially between GAC types, with a maximum for Filtrasorb-400, followed by Norit-PK1-3, and the minimum for Norit-RB4W. Yet overall, the observed pattern was the same, i.e. there was an inclining trend by increasing activated carbon dosage up to a peak value at 0.5 g GAC per 5 mL, and a downward trend for a further increase of the GAC dosage.
The evaluation of GAC surface properties in detail in the authors' previous study recognized that studied GACs are substantially different in their morphology, pore characteristics, and the quantity of functional groups on their surface.29 The size of the donor and recipient bacteria in this study i.e. E. faecalis is approximately 500 to 1000 nm,47,48 which is far larger than micropores, mesopores, and the majority of macropores, inhibiting these bacteria from diffusing into pores of the activated carbon structure. As a result, the attachment of donor and recipient bacteria is confined to the external surface of activated carbon and possibly some parts of super-large macropores, as documented in the literature.48–50 In this regard, these dissimilar surface properties may affect bacterial attachment or distribution on their surface, contributing to the observed differences in the dissemination of vancomycin resistance genes between the studied strains. Therefore, their role warranted the demand for further investigations by SEM.
3.2. SEM study on bacteria on activated carbon
GAC surface morphology in assays with bacteria was studied by SEM, as a determining factor in interactions between microorganisms and the adsorbent. These assessments were carried out in three categories to shed light on the results, shown in Fig. 1 and 2 by SEM, as follows.(i) assess the condition of bacteria, attached to the GAC surface.
(ii) qualitative analysis of the relation of GAC dosage and the spatial distribution of bacteria on the adsorbent surface.
(iii) quantify spatial distribution of bacteria on the GAC surface and investigate the potential association with gene transfer frequency.
 | ||
| Fig. 3 SEM micrographs of E. faecalis with 24k magnification on GACs surface; (a): Filtrasorb-400, (b): GAC Norit-PK1-3, (c): Norit-RB4W. | ||
All micrographs confirm that bacteria adhered to all types of topographic features of the uneven GAC surface including voids, flat surfaces and edges, even though the distribution density was not homogenous. Moreover, as shown in all micrographs, the bacteria attached to the activated carbon surface were probably viable, since some cells appeared to be actively dividing. It is consistent with the documented results in the literature that activated carbon provides neutral conditions for bacteria to attach and stay adhered to it due to ionic interactions and a wide variety of weak but effective chemical bonds such as hydrogen bonds, van der Waals, etc.11,51,52
Fig. 4 presents a higher magnification with evidence of close contact between cells as well as between bacteria and activated carbon by providing micrographs with 50k and 100k magnification. These higher magnifications enabled closer observation of interfaces of cell–cell and cell-GAC surface contacts.
Most of the GAC attached bacteria occurred in clusters on the activated carbon surface of organisms rather than as single cells with even distribution, according to Fig. 3. A clustered distribution may be formed either in the initial moments of adsorption or by the multiplication of microorganisms. If such a cluster contained a mixture of donors and recipients, which is highly probable considering the numbers of cells in the liquid media, it would facilitate the conjugation via direct cell to cell contact on the GAC surface, resulting in higher gene transfer frequency between them.
The micrographs in Fig. 3 confirm that the bacterial distribution patterns on all GAC types bear evidence of spatial proximity between bacterial cells as an essential prerequisite for horizontal gene transfer between the donor and recipient bacteria through the pheromone-assisted conjugation process regardless of differences in carbon surface morphology. Activated carbon, and bacteria and their extracellular polymeric substances contain different functional chemical groups on their surface, likely to enable attachment of bacteria to the activated carbon surface. Ionic interactions and various chemical bonds play an important role in keeping the bacteria attached to the adsorbent.20,21,48,53 It leads to substantial increase of gene transfer frequency as shown in graphs in Fig. 1.
Micrographs related to the samples containing 0.1 g (Fig. 5(a) and (b)) and 0.5 g (Fig. 5(c) and (d)) of GAC exhibited a noticeably high spatial density of bacteria on the adsorbent surface. In contrast, micrographs of samples containing 1.0 g of activated carbon (Fig. 5(e) and (f)) illustrated comparably lower accumulation of bacteria, adhered on the surface.
This observation is explained by considering that the bacterial concentration was identical across all three series of samples (containing 0.1, 0.5, and 1.0 g GAC). At 0.1 g GAC, the available surface area was smaller than the bacterial demand for attachment, leading to complete surface occupation by attached bacteria. A similar condition persisted at 0.5 g GAC, where bacteria continued to fully occupy the available surface. However, when the dosage increased to 1.0 g GAC, the available surface area exceeded the number of bacteria, resulting in lower spatial density compared with the 0.1 and 0.5 g samples.
These results are likely to comply with the values of gene transfer frequency in Fig. 1 for samples containing 0.5 g and 1.0 g of activated carbon. In other words, the noteworthy lower accumulation and proximity of bacteria on the adsorbent surface for the samples containing 1.0 g of GAC provided less opportunities for them to contact each other, leading to less conjugal transfer of gene from donors to recipients, compared with the samples containing 0.1 g and 0.5 g of GAC in the media.
The micrographs of the particles taken from samples containing 0.1 g (Fig. 5(a) and (b)) and 0.5 g (Fig. 5(c) and (d)) of activated carbon did not reveal notable differences in bacterial aggregation on the adsorbent surface. However, the gene transfer frequency of the latter exceeded that of the sample containing 0.1 g by factor 6.5 (Fig. 1). Since the concentration of bacteria cells remained identical across the three treatments with different doses, these observations could be explained by the general effect of activated carbon in facilitating the spread of antibiotic resistance via conjugation.
In other words, by introducing activated carbon to the media, noticeable amounts of bacteria attach to its surface and benefit from its advantages of facilitating horizontal gene transfer. By increasing the relative dose of activated carbon in the range between 0.1 to 0.5 g, the total available surface area for bacteria to adhere to will have increased, and as spatial proximity (and thus the number of cell to cell contacts per unit area) has remained virtually unchanged, the total gene transfer frequency has risen. In this dosage range, the available surface area of activated carbon determined the achieved gene transfer frequency, and the spatial proximity of cells on the adsorbent surface was relatively high.
GAC dosage of 1.0 g further increased the available GAC surface area but reduced the spatial proximity of bacteria cells on the adsorbent surface, leading to a lower probability of conjugation. In this dosage range, availability of GAC surface area far exceeded the area required by bacteria cells seeking attachment and thus decreased the chance of attached donor and recipient microorganisms to contact each other and transfer vancomycin-resistant genes via conjugation (as illustrated in Fig. 5(e) and (f) for samples containing 1.0 g GAC).
Since presence of higher numbers of bacteria per unit surface area of the adsorbent increased the probability of effective contact between the donor and recipient cells, the variation of the gene transfer frequency in treatments with different GAC dosages was compared with the number of bacteria per unit adsorbent surface area (spatial density) as well as the number of cell to cell contacts between them, quantitatively. For this purpose, several granules of activated carbon were selected from three samples containing 0.1, 0.5, and 1.0 g of Filtrasorb-400 and were prepared for SEM analysis, as described in section 2.6. Then, micrographs with the magnification of 10k were randomly taken from twelve granules on the stand (Fig. 6(b)) as well as evenly distributed twelve points on a single random particle (Fig. 6(a)).
The number of bacteria and the number of contacts between them were counted in each micrograph, covering the same surface area, i.e. approximately 7.61 × 10−10 m2. These values were graphed along with the gene transfer frequency of the sample, which the granules were taken from (Fig. 8 and 9). Fig. S4(a) and (b) show examples of counting numbers of bacteria and contacts between them in SEM micrographs, respectively.
To assess whether the recorded micrographs can represent whole samples, the number of bacteria and contacts between cells on various GAC granules (as one group) were compared with the results for different points on a single random granule (as another group) as shown in Fig. 7(a) and (b). For this purpose, Mann Whitney U test was carried out. It was hypothesized that significant difference between these two groups reveals non-uniformity of samples, so the points of which the micrographs are taken cannot represent all granules. Otherwise (non-significant outcome of the statistical analysis), the randomly selected points are roughly similar and can appropriately represent the all GAC granules in the samples.
 | ||
| Fig. 7 The comparison of attached-cell numbers (a), and contacts between cells (b), on various GAC granules and different points of a random granule. | ||
According to the results of the statistical analysis and as shown in Fig. 7(a) and (b), the number of cells and the number of contacts between them on several GAC granules' surfaces were not significantly different from various points of the random GAC granule. This result confirmed that the micrographs taken, represent all GAC granules in the media for the quantitative analysis.
Fig. 8 illustrates the results of the quantitative assessment of SEM micrographs of various samples containing different doses of activated carbon.
According to Fig. 8, the variation in the number of bacteria per unit area (spatial density) and the contacts between them per unit area follow a similar trend as gene transfer frequency versus activated carbon dosage.
To attain a normalized parameter that represents the spatial proximity of bacteria on the adsorbent surface, the ratio between the numbers of cell-to-cell contacts to the total number of attached bacteria in each micrograph was calculated. Fig. 9 compares the trend of this ratio with gene transfer frequency for each sample for the GAC dosage range investigated.
According to Fig. 8 and 9 and in conformity with qualitative comparison, the quantitative analysis reveals that with a low dosage of activated carbon (0.1 g per 5 mL) in the medium, the bacteria would have used all available space suitable for their surface attachment with the highest possible spatial proximity of cells. This spatial proximity was kept approximately constant (with a slight insignificant increase) at a higher activated carbon dosage in the media of 0.5 g per 5 mL, associated with a sharp increase in gene transfer frequency to achieve its peak value as the available surface for bacterial attachment increased fivefold in Fig. 1(a). However, since the initial bacteria concentration was identical in all treatments, further increase in the adsorbent dosage (to 1.0 g per 5 mL) and thus increase in GAC surface area reduced the normalised value of the number of cell-contacts to the number of bacteria per unit area on GAC surface, which was associated with a remarkable drop in the horizontal gene transfer frequency.
4. Discussion
The experimental results showed a substantial increase in gene transfer frequency by introducing activated carbon to the growth media (as shown in Fig. 1 and 2). In addition, the normalized number of cell-to-cell contacts based on the spatial distribution of the bacteria on the GAC surface in SEM micrographs revealed a direct correlation with the measured gene transfer frequency (Fig. 9). To get a broader comprehension of these findings, the mechanism of the conjugation between E. faecalis strains as well as their interactions with the studied GACs were discussed.4.1. The promotion of conjugation between E. faecalis strains in presence of GAC
Conjugation between two E. faecalis strains is a pheromone-inducible process of genetic materials transfer. Regardless of many Gram-negative bacteria, E. faecalis benefits from a specialized signaling system to initiate “on-demand” conjugation process instead of relying on a pilus for mating pair formation which is constitutively on for gene transfer.54–56 This mechanism promotes the transfer of conjugative plasmids that often carry important traits like antibiotic resistance genes (vancomycin-resistance genes in this study) and involves four key steps i.e. pheromone sensing, donor activation, cell-to-cell aggregation and DNA transfer.54,57 In brief, it initiates with excreting a small peptide pheromone by recipient cells as their tendency to receive the conjugative plasmid, which is recognized by donor cells carrying the plasmid and intensifies a series of gene expression and activates the gene transfer system in their structure. Subsequently, the donor cell generates a surface protein called aggregation substance, causing donor and recipient cells to clump together and enabling them for direct cell-to-cell contact and efficient transfer of the conjugative plasmid. Once the mating aggregates are formed, the conjugative plasmid is transferred in a single strand of DNA form from the donor to the recipient cell via a gate, created between two cells. Once inside the recipient, this single strand is replicated to restore the double-stranded plasmid.54,56,57The observed correlation of the normalized cell-to-cell contacts value on the spatial density of bacteria on GAC surface with gene transfer frequency (Fig. 8 and 9), seems to be closely related to the critical role of contacts between mating cells in this type of conjugation mechanism, as the GACs facilitates creation of aggregates on its surface and surrounding, leading to more contacts between cells and resultantly, more efficient plasmid transfer from the donor to the recipient E. faecalis cells.
4.2. Interactions between E. faecalis and GACs
SEM micrographs in Fig. 3 to 5 showed the studied strains cells on granular activated carbons' surface. Activated carbon is a porous material and contains a far larger surface area compared with a wide variety of other solid materials.9,17 Pores in activated carbon structure are categorized into three main groups according to their average diameter i.e. less than 2 nm for micropores, 2–50 nm for mesopores, and more than 50 nm for macropores.48,58,59 As discussed in section 3.1, the size of the donor and recipient E. faecalis bacteria in this study is approximately 500 to 1000 nm,47,48 which is far larger than micropores, mesopores, and the majority of macropores, inhibiting these microbes from diffusing into pores of the activated carbon structure. As a result, the attachment of donor and recipient bacteria is confined to the external surface of activated carbon and possibly some parts of super-large macropores and the observed contribution of studied activated carbons with horizontal gene transfer (Fig. 1 and 2) seems to be related to interactions between E. faecalis bacteria and the surface of the studied activated carbons.Activated carbon's surface interactions with bacteria, e.g. enterococci are primarily affected by surface charges. These interactions start with ionic repulsion or attraction within aqueous environments, followed by other bonds.9,48,49,60,61 E. faecalis bacteria carry slightly negative charges in the aqueous environment and the activated carbon granules possess various negative and positive sites, distributed on their surface, depending on their isoelectric point (pHiep) values.62 The pHiep values of activated carbon (Table 1) describes its surface charge, as the solid surface carries positive charges in pH values lower than pHiep and negative charges in pH values above it.52,62
| Adsorbent name | Isoelectric point (pHiep) |
|---|---|
| Filtrasorb-400 | 7.8 ± 0.2 |
| Norit-PK1-3 | 8.1 ± 0.2 |
| Norit-RB4W | 7.9 ± 0.3 |
Regarding Table 1, GACs in this study carry slightly positive charges in TSB, according to their pHiep values and the pH value of the inoculated TSB media (i.e. pH = 7.5 ± 0.3). The measured zeta potentials of E. faecalis were −12.02 ± 3 mV and −13.84 ± 3 mV for the donor (MF06036Van) and recipient (MW01105Rif) strains, respectively. Therefore, bacteria in the media seem to confront no serious constraint in approaching the activated carbon's surface, because the ionic interactions are likely to be attraction, at least in the initial moment of interaction between bacterial cell and carbon surface.
Another important group of interactions between bacteria and activated carbon that come into effect once they approach the activated carbon's surface are Lifshitz–van der Waals forces as well as the hydrophobicity of the contacting surfaces.61,63 These bonds are capable of keeping bacteria strongly attached to the adsorbent surface, even if the ionic interactions are repulsion and remove waterborne microorganisms such as like E. faecalis from drinking water.21,64 These forces along with bi-polar and hydrogen bonds are determinants in the adhesion and mostly passive movement of bacterial cells on the activated carbon surface, leading to the facilitation of horizontal gene.61,63
The composition and properties of the microorganisms' surface may change considerably in different conditions. These alterations may cause different forces to control the interactions of bacteria with activated carbon. Excretion of pheromone by recipient E. faecalis cells and aggregation proteins by the donors (for conjugation and gene transfer purposes)54,57,65 and generation of extracellular polymeric substances (EPS, for strengthening their adhesion to solid surfaces) are two examples of these alterations.9,63,65,66 As a result, there is a wide range of functional groups and negative or positively charged sites on bacterial assemblage generating interactions with the activated carbon surface and the ambient solutions. In real-scale activated carbon filtration these interactions take place instantly, and deposition, attachment, adhesion, and detachment occur simultaneously, since the system is dynamic.53
As mentioned in section 4.1, aggregation of bacteria and creation of a close contact between mating cells plays crucial participation in E. faecalis conjugation process. It appears to be associated with facilitation of the aggregation proteins performance in clumping mating recipient and donor cells via interactions formed between the functional groups on their structure and GACs surface. In other words, activated carbon promotes conjugation processes quantity and efficiency by providing a favourable surface for E. faecalis cells to attach on its surface and remain there on one hand (Fig. 3), and holding the excreted pheromones and aggregation proteins close to its surface on the other hand, which ends in firmer aggregation and more efficient gene transfer process. This interpretation complies well with the observed difference in various GACs' ability in boosting horizontal gene transfer (Fig. 2), as they possess substantially dissimilar quantity of functional groups on their surface (Table 2).
| Functionality, mmol g−1 | Filtrasorb-400 | Norit-PK1-3 | Norit-RB4W |
|---|---|---|---|
| Total basicity | 0.541 | 0.179 | 0.146 |
| Total acidity | 0.213 | 0.049 | 0.041 |
| Carboxylic | 0.005 | 0.001 | 0.003 |
| Phenolic | 0.175 | 0.022 | 0.020 |
| Lactonic | 0.033 | 0.026 | 0.018 |
According to Table 2, Filtrasorb-400 contains the highest number of functional surface groups, and Norit-PK1-3 contains slightly more basic, acidic, phenolic, and lactonic groups than Norit-RB4W. These results are correlated to the level of measured gene transfer frequency, shown in Fig. 2, as these groups are capable of generating hydrogen bonds and van der Waals forces between cell walls, pheromone, aggregation proteins and EPS polymeric structures. Overall, it is likely that the observed difference in gene transfer frequency between the GAC types in this study, is possibly associated with the difference in the number of their accessible surface sites where interactions with bacteria result in net attraction, thus keeping the bacteria cells attached to the GAC surface. Further targeted tests such as functional group blocking experiments and surface modification operations are required to support this interpretation.
5. Limitations of research
The experiments were conducted in tryptic soy broth (TSB) in a laboratory-scale setup to ensure attaining measurable gene transfer frequency values under controlled conditions and applicable time duration to identify conjugation. Furthermore, this study included three types of granular activated carbon (GAC) while there are numerous types of activated carbon as well as other adsorbents, utilized in water and wastewater treatment processes. Regardless of these limitations, the outcome of this research is intended to develop a base for further studies to be conducted in wastewater or drinking water media in real-scale experiments and other adsorbents.6. Conclusions
The effect of three different types of activated carbon on horizontal gene transfer of vancomycin-resistance genes between two strains of E. faecalis was investigated. The outcome of the studies revealed that:- All GAC types in this study significantly facilitated the proliferation of vancomycin resistance genes compared to control samples (with no GAC), regardless of the differences in their surface properties. The GAC dose response patterns of horizontal gene transfer between bacteria were very similar but varied strongly in amplitude.
- Activated carbon provided a favourable substrate for E. faecalis strains to attach on its surface, confirmed by SEM studies.
- Image processing of SEM micrographs and the developed quantitative model validated the direct association of the observed gene transfer frequency with the normalized value of cell-to-cell contacts on the number of bacteria on GAC surface.
- Among the studied GACs, Filtrasorb-400 showed the highest boost to the gene transfer frequency. This adsorbent exhibited substantially higher quantities of mesopore volume and specific surface area and represented the best performance in removal and retaining antibiotic molecules from aqueous environment according to authors' previous studies. This is one of the most significant findings of this study and should be carefully considered by water treatment process designers and engineers in cases with higher bacterial loads. In other words, the potential of GAC to facilitate horizontal gene transfer through its surface properties and microbial interactions, must be evaluated alongside its capacity to adsorb and retain antibiotics and other micro-pollutants from water. However, further targeted tests such as functional group blocking experiments are required to confirm specific relation between these properties and the magnitude of their influence on gene transfer frequency, individually and synergistically.
According to the findings of this study a testable hypotheses is proposed as the activated carbon with more functional surface groups content and higher pore volume (leading to better removal efficiency) is likely to impose more selection pressure on spread of antibiotic resistance. In addition, an actionable recommendation for treatment process engineers is to evaluate the potential of activated carbon to facilitate horizontal gene transfer by considering its surface characteristics and associated microbial interactions, alongside its capacity to remove antibiotics and other contaminants. Such an integrated evaluation would enable identification of optimal operational conditions that achieve maximum contaminant removal while minimizing the increment of horizontal gene transfer.
Author contributions
Hamed Rasouli Sadabad: conceptualization, methodology, investigation, formal analysis, writing – original draft, writing – review & editing James S. G. Dooley: conceptualization, methodology, writing – review & editing Heather M. Coleman: conceptualization, methodology, writing – review & editing Barry O'Hagan: methodology, investigation William J. Snelling: conceptualization, Joerg Arnscheidt: supervision, conceptualization, methodology, writing – original draft, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of study are included within the article and its supplementary material (SI). The materials used and experimental protocols developed in this research are described in detail in the Materials and methods section, with corresponding schematics provided in the SI to ensure reproducibility.Supplementary information is available. See DOI: https://doi.org/10.1039/d5ew01295c.
Acknowledgements
The authors are grateful to Ulster University Doctoral College and the Department for the Economy (DfE) for funding this research through a Vice Chancellor's Research Scholarship for Hamed Rasouli Sadabad.References
- WHO, Antimicrobial Resistance, 2023, Available from: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance.
- Q. Tan, W. Li, J. Zhang, W. Zhou, J. Chen and Y. Li, et al., Presence, dissemination and removal of antibiotic resistant bacteria and antibiotic resistance genes in urban drinking water system: A review, Front. Environ. Sci. Eng., 2019, 13(3), 36, DOI:10.1007/s11783-019-1120-9.
- C. J. L. Murray, K. S. Ikuta, F. Sharara, L. Swetschinski, G. Robles Aguilar and A. Gray, et al., Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis, Lancet, 2022, 399(10325), 629–655, DOI:10.1016/S0140-6736(21)02724-0.
- I. Sanseverino, A. N. Cuenca, R. Loos, D. Marinov and T. Lettieri, State of the Art on the Contribution of Water to Antimicrobial Resistance, 2018, Dec. Report.
- M. C. Haug, S. A. Tanner, C. Lacroix, L. Meile and M. J. A. Stevens, Construction and characterization of Enterococcus faecalis CG110/gfp/pRE25*, a tool for monitoring horizontal gene transfer in complex microbial ecosystems, FEMS Microbiol. Lett., 2010, 313(2), 111–119, DOI:10.1111/j.1574-6968.2010.02131.x.
- Q. Jiang, M. Feng, C. Ye and X. Yu, Effects and relevant mechanisms of non-antibiotic factors on the horizontal transfer of antibiotic resistance genes in water environments: A review, Sci. Total Environ., 2022, 806, 150568, DOI:10.1016/j.scitotenv.2021.150568.
- Y. Yang, X. Yang, H. Zhou, Y. Niu, J. Li and X. Fu, et al., Bisphenols Promote the Pheromone-Responsive Plasmid-Mediated Conjugative Transfer of Antibiotic Resistance Genes in Enterococcus faecalis, Environ. Sci. Technol., 2022, 56(24), 17653–17662, DOI:10.1021/acs.est.2c05349.
- C. Kristich, L. Rice and C. Arias, Enterococcal Infection—Treatment and Antibiotic Resistance, in Enterococci: From Commensals to Leading Causes of Drug Resistant Infection, ed. M. Gilmore, D. Clewell and Y. Ike, Massachusetts Eye and Ear Infirmary, Boston, 2014, [cited 2023 Dec 20], Available from: https://www.ncbi.nlm.nih.gov/books/NBK190420/ Search PubMed.
- J. Yuan, E. Passeport and R. Hofmann, Understanding adsorption and biodegradation in granular activated carbon for drinking water treatment: A critical review, Water Res., 2022, 210, 118026, DOI:10.1016/j.watres.2021.118026.
- K. Slipko, D. Reif, H. Schaar, E. Saracevic, A. Klinger and L. Wallmann, et al., Advanced wastewater treatment with ozonation and granular activated carbon filtration: Inactivation of antibiotic resistance targets in a long-term pilot study, J. Hazard. Mater., 2022, 438, 129396, DOI:10.1016/j.jhazmat.2022.129396.
- M. J. Luján-Facundo, M. I. Iborra-Clar, J. A. Mendoza-Roca and M. I. Alcaina-Miranda, Pharmaceutical compounds removal by adsorption with commercial and reused carbon coming from a drinking water treatment plant, J. Cleaner Prod., 2019, 238, 117866, DOI:10.1016/j.jclepro.2019.117866.
- L. W. Huang, S. Chen, Z. Tian and J. Wei, Thermal desorption characteristics of the adsorbate in activated carbon based on a two-dimensional heat and mass transfer model, Appl. Therm. Eng., 2022, 214, 118775, DOI:10.1016/j.applthermaleng.2022.118775.
- L. C. G. Hoegberg, G. Shepherd, D. M. Wood, J. Johnson, R. S. Hoffman and E. M. Caravati, et al., Systematic review on the use of activated charcoal for gastrointestinal decontamination following acute oral overdose, Clin. Toxicol, 2021, 59(12), 1196–1227, DOI:10.1080/15563650.2021.1961144.
- M. Wang, G. Bera, K. Mitra, T. L. Wade, A. H. Knap and T. D. Phillips, Tight sorption of arsenic, cadmium, mercury, and lead by edible activated carbon and acid-processed montmorillonite clay, Environ. Sci. Pollut. Res., 2021, 28(6), 6758–6770, DOI:10.1007/s11356-020-10973-z.
- M. M. Heimesaat, N. Schabbel, L. Q. Langfeld, N. W. Shayya, S. Mousavi and S. Bereswill, Prophylactic Oral Application of Activated Charcoal Mitigates Acute Campylobacteriosis in Human Gut Microbiota-Associated IL-10−/− Mice, Biomolecules, 2024, 14(2), 141, DOI:10.3390/biom14020141.
- K. Y. Foo and B. H. Hameed, Insights into the modeling of adsorption isotherm systems, Chem. Eng. J., 2010, 156(1), 2–10, DOI:10.1016/j.cej.2009.09.013.
- V. Rakić, V. Rac, M. Krmar, O. Otman and A. Auroux, The adsorption of pharmaceutically active compounds from aqueous solutions onto activated carbons, J. Hazard. Mater., 2015, 282, 141–149, DOI:10.1016/j.jhazmat.2014.04.062.
- D. Mangla, S. A. Annu and S. Ikram, Critical review on adsorptive removal of antibiotics: Present situation, challenges and future perspective, J. Hazard. Mater., 2022, 425, 127946, DOI:10.1016/j.jhazmat.2021.127946.
- X. Shi, R. Pereira, S. L. Uzma, B. Poursat and D. Quinn, et al., Microbial stratification and DOM removal in drinking water biofilters: Implications for enhanced performance, Water Res., 2024, 262, 122053, DOI:10.1016/j.watres.2024.122053.
- S. M. Korotta-Gamage and A. Sathasivan, A review: Potential and challenges of biologically activated carbon to remove natural organic matter in drinking water purification process, Chemosphere, 2017, 167, 120–138, DOI:10.1016/j.chemosphere.2016.09.097.
- J. Yu, D. Zhang, W. Ren and B. Liu, Transport of Enterococcus faecalis in granular activated carbon column: Potential energy, migration, and release, Colloids Surf., B, 2019, 183, 110415, DOI:10.1016/j.colsurfb.2019.110415.
- X. Wang, Y. Mao, S. Tang, H. Yang and Y. F. Xie, Disinfection byproducts in drinking water and regulatory compliance: A critical review, Front. Environ. Sci. Eng., 2015, 9(1), 3–15, DOI:10.1007/s11783-014-0734-1.
- M. B. Salerno, M. Flamm, B. E. Logan and D. Velegol, Transport of Rodlike Colloids through Packed Beds, Environ. Sci. Technol., 2006, 40(20), 6336–6340, DOI:10.1021/es0614565.
- P. Devasia, K. A. Natarajan, D. N. Sathyanarayana and G. R. Rao, Surface Chemistry of Thiobacillus ferrooxidans Relevant to Adhesion on Mineral Surfaces, Appl. Environ. Microbiol., 1993, 59(12), 4051–4055, DOI:10.1128/aem.59.12.4051-4055.1993.
- P. Han, X. Shen, H. Yang, H. Kim and M. Tong, Influence of nutrient conditions on the transport of bacteria in saturated porous media, Colloids Surf., B, 2013, 102, 752–758, DOI:10.1016/j.colsurfb.2012.08.053.
- E. Burchacka, M. Łukaszewicz and M. Kułażyński, Determination of mechanisms of action of active carbons as a feed additive, Bioorg. Chem., 2019, 93, 102804, DOI:10.1016/j.bioorg.2019.02.029.
- S. Bereswill, S. Mousavi, D. Weschka and M. M. Heimesaat, Disease-Alleviating Effects of Peroral Activated Charcoal Treatment in Acute Murine Campylobacteriosis, Microorganisms, 2021, 9(7), 1424, DOI:10.3390/microorganisms9071424.
- E. Burchacka, K. Pstrowska, E. Beran, H. Fałtynowicz, K. Chojnacka and M. Kułażyński, Antibacterial Agents Adsorbed on Active Carbon: A New Approach for S. aureus and E. coli Pathogen Elimination, Pathogens, 2021, 10(8), 1066, DOI:10.3390/pathogens10081066.
- H. Rasouli Sadabad, H. M. Coleman, J. S. G. Dooley, W. J. Snelling, B. O'Hagan and A. Y. Ganin, et al., Desorption of Antibiotics from Granular Activated Carbon during Water Treatment by Adsorption, Environ. Processes, 2024, 11(4), 64, DOI:10.1007/s40710-024-00740-4.
- T. O. Olanrewaju, M. McCarron, J. S. G. Dooley and J. Arnscheidt, Transfer of antibiotic resistance genes between Enterococcus faecalis strains in filter feeding zooplankton Daphnia magna and Daphnia pulex, Sci. Total Environ., 2019, 659, 1168–1175, DOI:10.1016/j.scitotenv.2018.12.314.
- A. Cartwright, J. Arnscheidt, M. Conwell, J. S. G. Dooley, C. McGonigle and P. J. Naughton, Effects of freshwater sponge Ephydatia fluviatilis on conjugative transfer of antimicrobial resistance in Enterococcus faecalis strains in aquatic environments, Lett. Appl. Microbiol., 2020, 71(1), 39–45, DOI:10.1111/lam.13310.
- CDC, US Department of Health and Human Services, Vancomycin-resistant Enterococci (VRE) in Healthcare Settings - Antibiotic resistance threats in the United States, 2019, [cited 2023 Dec 20], Available from: https://www.cdc.gov/hai/organisms/vre/vre.html.
- M. Serra-Burriel, M. Keys, C. Campillo-Artero, A. Agodi, M. Barchitta and A. Gikas, et al., Impact of multi-drug resistant bacteria on economic and clinical outcomes of healthcare-associated infections in adults: Systematic review and meta-analysis, PLoS One, 2020, 15(1), e0227139, DOI:10.1371/journal.pone.0227139.
- M. Fukushige, L. S. Syue, K. Morikawa, W. L. Lin, N. Y. Lee and P. L. Chen, et al., Trend in healthcare-associated infections due to vancomycin-resistant Enterococcus at a hospital in the era of COVID-19: More than hand hygiene is needed, J. Microbiol., Immunol. Infect., 2022, 55(6), 1211–1218, DOI:10.1016/j.jmii.2022.08.003.
- A. Hanczvikkel and Á. Tóth, Quantitative study about the role of environmental conditions in the survival capability of multidrug-resistant bacteria, J. Infect. Public Health, 2018, 11(6), 801–806, DOI:10.1016/j.jiph.2018.05.001.
- C. Vaziri and C. Darkoh, Environmental contamination contributing to the spread of vancomycin-resistant enterococci in nursing homes, Lancet Healthy Longev., 2023, 4(11), e587–e588, DOI:10.1016/S2666-7568(23)00197-6.
- APHA, Standard Methods for the Examination of Water and Wastewater, ed. E. W. Rice, L. Baird and A. D. Eaton, American Public Health Association, Washington, DC, 23rd edn, 2017, vol. 10 Search PubMed.
- T. O. Olanrewaju, The Emergence and Transfer of Antibiotic Resistance in Aquatic Environments: Effects of Plankton Interactions, PhD thesis, Ulster University, 2020 Search PubMed.
- M. Conwell, Transfer of antibiotic resistances between enterococci in biofilm, PhD thesis, Ulster University, 2018 Search PubMed.
- M. Kim, D. Gutiérrez-Cacciabue, A. Schriewer, V. B. Rajal and S. Wuertz, Evaluation of detachment methods for the enumeration of Bacteroides fragilis in sediments via propidium monoazide quantitative PCR, in comparison with Enterococcus faecalis and Escherichia coli, J. Appl. Microbiol., 2014, 117(5), 1513–1522, DOI:10.1111/jam.12630.
- K. Huang, Y. Tian, D. Salvi, M. Karwe and N. Nitin, Influence of Exposure Time, Shear Stress, and Surfactants on Detachment of Escherichia coli O157:H7 from Fresh Lettuce Leaf Surfaces During Washing Process, Food Bioprocess Technol., 2018, 11(3), 621–633, DOI:10.1007/s11947-017-2038-5.
- J. A. Quek, S. M. Lam, J. C. Sin and A. R. Mohamed, Visible light responsive flower-like ZnO in photocatalytic antibacterial mechanism towards Enterococcus faecalis and Micrococcus luteus, J. Photochem. Photobiol., B, 2018, 187, 66–75, DOI:10.1016/j.jphotobiol.2018.07.030.
- M. A. Kim, V. Rosa and K. S. Min, Characterization of Enterococcus faecalis in different culture conditions, Sci. Rep., 2020, 10(1), 21867, DOI:10.1038/s41598-020-78998-5.
- A. Kumar Oli, R. Sungar, N. Shivshetty, R. Hosamani and K. Chandrakanth Revansiddappa, A Study of Scanning Electron Microscope of Vancomycin Resistant Enterococcus faecalis from Clinical Isolates, Adv. Microbiol., 2012, 02(02), 93–97, DOI:10.4236/aim.2012.22012.
- E. Ostertagová, O. Ostertag and J. Kováč, Methodology and Application of the Kruskal-Wallis Test, Appl. Mech. Mater., 2014, 611, 115–120, DOI:10.4028/www.scientific.net/AMM.611.115.
- Y. Dodge, Kruskal-Wallis Test, in The Concise Encyclopedia of Statistics, Springer New York, New York, NY, 2008, [cited 2024 Nov 5], pp. 288–290, Available from: DOI:10.1007/978-0-387-32833-1_216.
- Q. Yuan, C. Zhou, J. Xie, D. Zhang, L. Zheng and Y. Li, et al., Subgingival Microbes, in Atlas of Oral Microbiology, Elsevier, 2015, pp. 67–93, DOI:10.1016/B978-0-12-802234-4.00004-5.
- H. Kim, S. Park, C. Lee, S. Kim and K. Kim, Bacterial attachment to iron-impregnated granular activated carbon, Colloids Surf., B, 2009, 74(1), 196–201, DOI:10.1016/j.colsurfb.2009.07.018.
- X. Bai, X. Ma, F. Xu, J. Li, H. Zhang and X. Xiao, The drinking water treatment process as a potential source of affecting the bacterial antibiotic resistance, Sci. Total Environ., 2015, 533, 24–31, DOI:10.1016/j.scitotenv.2015.06.082.
- P. Servais, G. Billen and P. Bouillot, Biological Colonization of Granular Activated Carbon Filters in Drinking-Water Treatment, J. Environ. Eng., 1994, 120(4), 888–899, DOI:10.1061/(ASCE)0733-9372(1994)120:4(888).
- H. Yankovych, I. Melnyk and M. Václavíková, Understanding of mechanisms of organohalogens removal onto mesoporous granular activated carbon with acid-base properties, Microporous Mesoporous Mater., 2021, 317, 110974, DOI:10.1016/j.micromeso.2021.110974.
- A. Rao, A. Kumar, R. Dhodapkar and S. Pal, Adsorption of five emerging contaminants on activated carbon from aqueous medium: kinetic characteristics and computational modeling for plausible mechanism, Environ. Sci. Pollut. Res., 2021, 28(17), 21347–21358, DOI:10.1007/s11356-020-12014-1.
- C. C. Wu, N. G. Love and T. M. Olson, Bacterial transmission and colonization in activated carbon block (ACB) point-of-use (PoU) filters, Environ. Sci., 2021, 7(6), 1114–1124, 10.1039/D0EW00982B.
- X. Yang, Y. Niu, Y. Yang, H. Zhou, J. Li and X. Fu, et al., Pheromone effect of estradiol regulates the conjugative transfer of pCF10 carrying antibiotic resistance genes, J. Hazard. Mater., 2023, 451, 131087, DOI:10.1016/j.jhazmat.2023.131087.
- D. B. Clewell, Tales of conjugation and sex pheromones, Mobile Genet. Elem., 2011, 1(1), 38–54, DOI:10.4161/mge.1.1.15409.
- D. B. Clewell and M. V. Francia, Conjugation in Gram-Positive Bacteria, in Plasmid Biology, ASM Press, Washington, DC, USA, 2014, pp. 227–256, DOI:10.1128/9781555817732.ch10.
- R. A. Moore and C. E. Carr, Snyder and Champness Molecular Genetics of Bacteria, Emerg Infect Dis., 5th edn, 2022, vol. 28(1), pp. 266–266, DOI:10.3201/eid2801.211628.
- T. J. Bandosz, Activated carbon surfaces in environmental remediation, ed. J. Teresa, Elsevier, Bandosz, 2006 Search PubMed.
- Z. Lu, W. Sun, C. Li, W. Cao, Z. Jing and S. Li, et al., Effect of granular activated carbon pore-size distribution on biological activated carbon filter performance, Water Res., 2020, 177, 115768, DOI:10.1016/j.watres.2020.115768.
- J. Yu, D. Zhang, W. Ren and B. Liu, Transport of Enterococcus faecalis in granular activated carbon column: Potential energy, migration, and release, Colloids Surf., B, 2019, 183, 110415, DOI:10.1016/j.colsurfb.2019.110415.
- H. J. Busscher, R. J. B. Dijkstra, D. E. Langworthy, D. I. Collias, D. W. Bjorkquist and M. D. Mitchell, et al., Interaction forces between waterborne bacteria and activated carbon particles, J. Colloid Interface Sci., 2008, 322(1), 351–357, DOI:10.1016/j.jcis.2008.03.018.
- H. N. Kim, S. A. Bradford and S. L. Walker, Escherichia coli O157:H7 Transport in Saturated Porous Media: Role of Solution Chemistry and Surface Macromolecules, Environ. Sci. Technol., 2009, 43(12), 4340–4347, DOI:10.1021/es8026055.
- R. Bos, H. C. van der Mei and H. J. Busscher, Physico-chemistry of initial microbial adhesive interactions – its mechanisms and methods for study, FEMS Microbiol. Rev., 1999, 23(2), 179–230, DOI:10.1111/j.1574-6976.1999.tb00396.x.
- M. Park, S. Wu, I. J. Lopez, J. Y. Chang, T. Karanfil and S. A. Snyder, Adsorption of perfluoroalkyl substances (PFAS) in groundwater by granular activated carbons: Roles of hydrophobicity of PFAS and carbon characteristics, Water Res., 2020, 170, 115364, DOI:10.1016/j.watres.2019.115364.
- Z. Li, T. Li, X. Xing, Z. Bi, P. Qi and C. Hu, et al., Inhibiting the increase of antibiotic resistance genes during drinking water distribution by superior microbial interface using Fe modified granular activated carbon, J. Cleaner Prod., 2022, 335, 130225, DOI:10.1016/j.jclepro.2021.130225.
- W. Zhao, J. You, S. Yin, H. Yang, S. He and L. Feng, et al., Extracellular polymeric substances—antibiotics interaction in activated sludge: A review, Environ. Sci. Ecotechnology, 2023, 13, 100212, DOI:10.1016/j.ese.2022.100212.
| This journal is © The Royal Society of Chemistry 2026 |