Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Exploration of how the molecular structure surrounding the β-dicarbonyl group impacts formation of brominated haloacetic acids
Polly Grundy *a,
Peter Jarvis
*a,
Peter Jarvis a,
John Fawella,
Pablo Campo
a,
John Fawella,
Pablo Campo a,
Emma Goslan
a,
Emma Goslan a,
John Haley
a,
John Haley b and
Bruce Jefferson
b and
Bruce Jefferson a
a
aCranfield University, College Road, Cranfield, Bedfordshire, MK43 0AL, UK. E-mail: polly.grundy@cranfield.ac.uk
bUK Water Industry Research Ltd. (UKWIR), 1st Floor, 3 More London Riverside, London SE1 2RE, UK
First published on 8th May 2026
Abstract
Disinfection by-products (DBPs) are formed during the chlorination of drinking water and present a risk to human health, with brominated DBPs identified as more toxic than their chlorinated analogues. The most widely regulated DBPs are trihalomethanes (THMs), but increasingly focus is on haloacetic acids (HAAs), including consideration of all nine brominated and chlorinated HAAs for regulation (HAA9). Recently, aliphatic carbonyl compounds have been recognised as important DBP precursors and to favour bromine incorporation. This research specifically focuses on smaller aliphatic carbonyl precursors, enabling identification of the molecular-level features that are most significant for increasing the risk of forming the more toxic brominated DBPs and for reducing compliance with potential HAA9 regulations. This showed that β-keto acids present a particular problem due to their high propensity for bromination and Br-HAA formation. However, in the context of HAA9 standards, the neutral ester compounds were equally significant due to formation of the mixed chlorinated and brominated bromochloroacetic acid, which is within the currently unregulated HAAs. The enhanced understanding of Br-DBP formation from such hydrophilic organic precursors is important as such compounds are poorly removed by standard coagulation water treatment and are formed during oxidative treatment processes. Consequently, source waters containing these compounds and bromide are at significant risk of forming unacceptable levels of HAA9, a problem which becomes increasingly urgent as pressure on resources forces reliance on poorer quality drinking water sources containing higher levels of bromide. Further, the work shows that current monitoring approaches are inadequate when we need to consider HAA9.
Water impactThis study explored the interaction between chlorine, bromide and aliphatic carbonyl compounds during the disinfection of drinking water. Understanding the molecular level influences on brominated haloacetic acid (HAA) formation from chlorination of such hydrophilic organic compounds is vital for identifying waters at risk of non-compliance with proposed HAA9 regulation, and for directing treatment processes to minimise potential DBP-derived toxicity. |
1 Introduction
Disinfection by-products (DBPs) such as trihalomethanes (THMs) and haloacetic acids (HAAs) are formed during the disinfection of drinking water with chlorine, with the more toxic brominated analogues forming when bromide is present.1 Although THMs and up to five HAAs (HAA5) are widely regulated in countries such as EU, US and Scotland,2–4 the larger brominated HAAs are currently excluded and limits for all nine chlorinated and brominated HAAs (HAA9) are not yet in place. However, it is anticipated that future regulations will be extended to include HAA9, with this being actively considered in the UK and the US.5,6In light of this potential regulatory change, it is prudent to re-examine the role of precursor characteristics on the formation of HAA9 and particularly how inclusion of the currently unregulated HAA4 changes thinking on precursor management. This remains challenging as dissolved organic matter (DOM) as a precursor for DBPs is a complex and poorly characterised heterogeneous mix of compounds from both natural and anthropogenic sources. It is known to include both aromatic and aliphatic compounds with a range of molecular weights and hydrophilicities.7 Existing research into DBP precursors has identified that in real world water sources THMs tend to form at higher levels from the hydrophobic, aromatic fraction of organic matter, whereas HAAs form equally from both the hydrophobic and the more hydrophilic, aliphatic fraction.8 This tendency has enabled treatment enhancements to deliver increasing levels of compliance with THM regulations as the hydrophobic fraction is more readily identified, e.g. by UV absorbance, and is also more readily removed during standard coagulation processes.9 However, as new legislation directs towards also controlling the HAAs, including the brominated unregulated HAA4, it is posited that in the future equal prominence will need to be given to the hydrophilic fraction of organic matter as a precursor for DBPs. For some lowland surface water sources this will present a particular challenge as these often have higher levels of hydrophilic organic matter and bromide and have historically received less treatment optimisation for dissolved organic carbon (DOC) removal due to their lower formation of the regulated THMs.10,11 Additionally, the presence of ions including metal cations and halide anions are known to promote HAA formation and increase bromination of DBPs, with this of lesser import for THMs.12,13 As such, hydrophilic compounds represent a potentially important and currently overlooked source of DBP derived toxicity and may constrain compliance with proposed HAA9 regulation.
Aliphatic carbonyls are hydrophilic organic compounds which are likely to be present in raw waters and can also be created during treatment (Table 1). Although they may be removed by biological activity,14 the likelihood remains that some will be present in treated water at the point of chlorination, and they are thought to account for some of the observed higher formation of HAAs from hydrophilic organic matter.15 For example, small organic acids have been investigated in drinking water treatment plants and found to be higher during winter and after ozonation/chlorination.16 However, levels of aliphatic carbonyls in source waters, including the important HAA precursor group of β-dicarbonyls, are currently uncertain with such small, polar organic compounds difficult to analyse and their occurrence in DOC poorly characterised. Consequently, they demand further attention.
| Type | Source of carbonyls | Examples | Ref. |
|---|---|---|---|
| Natural – allochthonous | Natural processes creating and utilising carbonyls from both plants and animals | Organic acids synthesised in plants e.g. maleic acid in legumes, acetoacetates in fruit – citric acid ‘Krebs’ cycle | 17–19 |
| Metabolites in urine include ketones and acetoacetates – fatty acid metabolism | |||
| Breakdown of particulates including from soil run off or atmospheric dry deposition | Wildfires form carbonyls which are deposited in water or washed in from soil It has been observed that dry deposition contains more hydrophilic organic matter. Diacids including malonic acid detected in precipitation up to 77 μg L−1 | 20–24 | |
| Natural – autochthonous | Breakdown of humic and fulvic acids by light. Could be during reservoir storage or water transport by canal | Photolysis of humic shown to give dicarbonyls and low molecular weight organic acids e.g. malonic acid demonstrated in raw waters from reservoirs and surface sources. Increasing organic acids, and decreasing UV absorbance, observed with light exposure. Temperature and seasonal impact expected | 25–28 |
| Microbial degradation of organic matter to smaller soluble microbial products (SMPs) | SMPs include products from natural biodegradation processes which are known to generate smaller, less aromatic organic matter and DBP precursors | 29, 30 | |
| Anthropogenic – allochthonous | Contaminants in water from human activity i.e. human induced OM (HiOM). Could be point source or run-off | Industrial discharges including foodstuffs (coffee, fruit, bread contain acetoacetates), acetylacetone used in paint, β-dicarbonyls used for synthetic chemistry, chelating agents | 14, 31–34 |
| Wastewater effluent known to include carbonyls and carboxyls e.g. direct and indirect water reuse projects | |||
| Anthropogenic – autochthonous | Breakdown of larger organic matter in water source during treatment | Oxidative treatments (e.g. ozonation, advanced oxidation) break down larger organic matter to smaller, oxidised aliphatic compounds. Carbonyls, including malonic acid, are known by-products of ozonation | 35–39 |
| Cleaning or maintenance procedures can temporarily increase DBP precursors | Use of ozone to reduce microfiltration fouling; use of peracetic acid to reduce mussels | 14, 40, 41 | |
| Pre-treatments to increase efficiency of main removal technology | UVC pre-irradiation, pre-ozonation, pre-chlorination (or multiple ‘booster’ chlorinations) are all oxidative and can increase smaller, aliphatic compounds e.g. by-products of pre-ozonation include β-keto acids | 42–45 |
In addition to understanding more about the organic matter involved, levels of bromide will be a key factor due to the enhanced toxicity of the brominated DBPs and the move towards including brominated HAAs in an HAA9 standard. Bromide levels of 0–1 mg L−1 have been reported in fresh waters,46 and the problem will be compounded as the challenges of population increase and climate change force reliance on poorer quality source waters.47 Further, bromide is recalcitrant to removal by most water treatment processes, so high bromide is not readily corrected.48
When investigating this DBP risk from hydrophilic organic matter, model compound studies have been utilised to demonstrate that aliphatic carbonyls, and specifically β-dicarbonyls (1,3-dicarbonyls), are particularly prolific sources of HAAs.49 Furthermore, some aliphatic compounds have been shown to more readily incorporate bromide into the final DBPs than aromatic compounds.50 However, although the importance of the β-dicarbonyl group has been established, understanding of the molecular level influences on the DBP formation potential of this key group is missing. Previous laboratory studies of individual model compounds under chlorination conditions have tended to overlook aliphatic compounds, with far more focus on humic and fulvic acids and their breakdown products, or the degradation of micropollutants. Further, the attention has historically been on the formation of the regulated THMs, often omitting some or all of the HAAs; also model compound laboratory studies rarely include bromide.50,51 Similarly, while kinetic or molecular structural models have been used to investigate the formation of Br-DBPs during chloramination,52,53 there has been little research investigating such models for chlorination of aliphatic compounds or including both chlorine and bromide in competition during the formation of HAAs. Such models would need to establish the transformation pathways and intermediates for Br-HAA formation and their rate constants, and it is intended that this research provides a framework for deciding which compounds are of heightened interest for such studies.
Therefore, in this study model compounds were identified which allowed the influence of the molecular functional groups surrounding the β-dicarbonyl to be investigated, demonstrating where these increased bromine incorporation into HAAs. The compounds were selected based on existing knowledge of the key influences on DBP formation, which include that β-dicarbonyls have a high propensity for electrophilic substitution at the α-carbon and will follow this pathway over that of nucleophilic reactions under the conditions of drinking water chlorination.49,54 Bromination of dissociated aromatic phenolates has been identified as significantly faster than for the equivalent non-dissociated phenols,55 and so both acidic and neutral β-dicarbonyls were included to assess the impact of dissociation on bromine incorporation. Similarly, longer chain aliphatic dicarbonyls have been shown to form more THMs than shorter chain compounds,50 warranting further investigation of these compounds as a risk for both THM and HAA formation. In addition, pre-halogenated compounds were included to provide information on the differing behaviour of halogenated intermediates and 13C labelled diethyl malonate used to prove the hypothesis that the α-carbon is retained in HAAs formed (Table 2).
This study is unique in focusing on the aliphatic carbonyl precursors for DBPs and including both bromide and excess chlorine, as would be seen in real water disinfection. By performing these experiments at two concentrations, the molecular structures with the highest proclivity to incorporate bromide are demonstrated. The key role of the ratio between bromide and compound was further explored through stepwise changes in concentrations for three aliphatic compounds, selected based on the presence of molecular features that were significant for both THM and HAA formation. The data was then analysed through the comparative lens of the regulated HAAs and the unregulated HAA4 to ascertain how the adopting of or transferring to proposed HAA9 regulation changes the way we need to think about DBP monitoring and management. Additionally, this approach allows propositions for the most relevant compound types for Br-HAA formation and will inform future targeted research when developing kinetic models and demonstrating molecular formation pathways, providing additional insight into how the presence of hydrophilic organic matter can increase the toxicity of chlorinated drinking water.
2 Materials and method
2.1 Model compounds
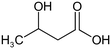
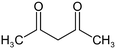
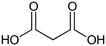
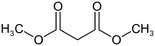
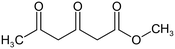
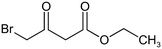
The following aliphatic carbonyl model compounds, divided into groups by molecular feature, were selected for this research: no β-ketone (3-hydroxybutanoic acid, methyl acetate, 3-aminobutanoic acid, acetone); β-ketone only (acetylacetone, 2,6-dimethyl-3,5-heptanedione); 3-carbon β-keto acids (malonamic acid, malonic acid); 3-carbon β-keto ester (methyl malonate, diethyl malonate; diethyl malonate-2-13C, dimethyl malonate); >3-carbon β-keto acid (3-oxobutanoic acid, 3-oxohexanedioic acid, 3-oxopentanedioic acid); >3-carbon β-keto ester (tert-butyl acetoacetate, ethyl acetoacetate, methyl acetoacetate, methyl 3,5-dioxohexanoate, dimethyl 3-oxopentanedioate); singly halogenated β-dicarbonyls (methyl 2-bromoacetate, diethyl bromomalonate, ethyl malonate chloride, ethyl 4-bromooacetoacetate, ethyl 4-chloroacetoacetate) and triply halogenated carbonyls (ethyl-4,4,4-trichloroacetoacetate, hexachloroacetone), with further detail in Table S1.2.2 Reagents and chemicals
Ammonium chloride (reagent grade), N,N-diethyl-1,4-phenylenediamine (DPD, Hach Lange™ free chlorine reagent powder pillows), formic acid (LCMS grade), methanol (HPLC and LCMS grade), methyl tert-butyl ether (MTBE, extra pure), potassium bromide (extra pure), potassium phosphate monobasic (extra pure), sodium hydroxide (1 M, NIST standard), sodium hypochlorite (technical ≈14%), sodium phosphate dibasic (reagent grade), sodium sulphate (anhydrous, extra pure), sulfuric acid (concentrated, reagent grade) and model compounds (other than the exceptions detailed below) were purchased from Fisher Scientific (Pittsburgh, USA).2,3-Dibromopropionic acid (2,3-DBPA), standard solutions of 4-bromofluorobenzene (4-BFB, 2000 μg ml−1 in methanol) and trihalomethanes (THM4, 2000 μg ml−1 in methanol) and model compounds 3-oxobutanoic acid (as lithium salt), 3-aminobutanoic acid, ethyl 4,4,4-trichloroacetoacetate, ethyl 4-bromoacetoacetate and 3-oxopentanedioic acid were purchased from Merck (Darmstadt, Germany). Diethyl malonate-2-13C and 3-oxohexanedioic acid were purchased from LGC Standards (London, UK). Methyl 3,5-dioxohexanoate was purchased from Key Organics (Camelford, UK). Malonamic acid was purchased from Apollo Scientific (Cheshire, UK). Haloacetic acids (HAA9, 1000 μg ml−1 in MTBE) standard solution was purchased from Restek (Buckinghamshire, UK).
THM4 comprises chloroform (trichloromethane, TCM), bromodichloromethane (BDCM), dibromochloromethane (DBCM) and bromoform (tribromomethane, TBM).
HAA9 comprises monochloroacetic acid (MCAA), monobromoacetic acid (MBAA), dichloroacetic acid (DCAA), dibromoacetic acid (DBAA) and trichloroacetic acid (TCAA), which are within HAA5, and also bromochloroacetic acid (BCAA), bromodichloroacetic acid (BDCAA), dibromochloroacetic acid (DBCAA) and tribromoacetic acid (TBAA), which are the currently unregulated HAA4.
Ultrapure (UP) water with resistivity 18.2 MΩ cm was provided by an Elga Purelab Ultra Genetic system.
2.3 Chlorination conditions
Sample solutions in ultrapure water were prepared in triplicate at concentrations of 1.5 μM and 15 μM, with chlorine added at a 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio of chlorine to organic compound and bromide at 5 μM (400 μg L−1) to represent high bromide waters. Two concentrations were chosen to demonstrate the influence of the ratio of bromide to compound on the bromine incorporation into the DBPs formed, whilst still allowing for excess chlorine to be present as would be encountered during disinfection procedures. The solutions were buffered at pH 7 with 5 mM phosphate, ensuring consistent conditions to enable key molecular features to be isolated, within the normal pH range of water treatment of pH 6 to pH 8. The chlorination solution was prepared from ≈14% sodium hypochlorite and standardised before use to establish the exact concentration. Standardisation was performed using a Hach Pocket Colorimeter II and N,N-diethyl-1,4-phenylenediamine (DPD) following a method adapted from Standard Method 4500-Cl G for drinking water.56 Experiments were maintained at 20 °C in the dark for 24 hours prior to quenching with 100 mg L−1 ammonium chloride (for HAAs) or 100 mg L−1 sodium sulphite (for THMs), in line with US EPA procedures. Glassware was soaked in ≈1 mM chlorine solution for at least 24 hours prior to chlorination experiments to reduce any risk of organic matter contamination.
1 molar ratio of chlorine to organic compound and bromide at 5 μM (400 μg L−1) to represent high bromide waters. Two concentrations were chosen to demonstrate the influence of the ratio of bromide to compound on the bromine incorporation into the DBPs formed, whilst still allowing for excess chlorine to be present as would be encountered during disinfection procedures. The solutions were buffered at pH 7 with 5 mM phosphate, ensuring consistent conditions to enable key molecular features to be isolated, within the normal pH range of water treatment of pH 6 to pH 8. The chlorination solution was prepared from ≈14% sodium hypochlorite and standardised before use to establish the exact concentration. Standardisation was performed using a Hach Pocket Colorimeter II and N,N-diethyl-1,4-phenylenediamine (DPD) following a method adapted from Standard Method 4500-Cl G for drinking water.56 Experiments were maintained at 20 °C in the dark for 24 hours prior to quenching with 100 mg L−1 ammonium chloride (for HAAs) or 100 mg L−1 sodium sulphite (for THMs), in line with US EPA procedures. Glassware was soaked in ≈1 mM chlorine solution for at least 24 hours prior to chlorination experiments to reduce any risk of organic matter contamination.
2.4 Targeted compounds for changing concentrations
Three model compounds (malonic acid, 3-oxobutanoic acid and 3-oxopentanedioic acid) were selected for further investigation of the influence of changing the ratio of bromide to compound. These represent compounds with 3, 4 and 5 carbons, and a high propensity to form either HAAs (malonic acid), THMs (3-oxobutanoic acid) or both (3-oxopentanedioic acid). These compounds also demonstrated a high tendency to incorporate bromide during the initial experiments, thus giving increased information on high Br-DBP formation as well as differing mechanisms for formation of THM and HAAs. Each of these targeted compounds was prepared at eight different concentrations, between 1 and 8 μM, with bromide maintained at 5 μM. Additionally, eight solutions maintaining model compound concentration at 2 μM and varying levels of bromide from 0 to 10 μM were prepared. All experiments were repeated in triplicate, a 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 chlorination ratio was maintained, and other chlorination conditions were as previously detailed.
1 chlorination ratio was maintained, and other chlorination conditions were as previously detailed.
2.5 DBP analysis
Analysis for nine HAAs was performed using the liquid chromatography tandem mass spectrometry (LC–MS/MS) procedure developed and validated by Grundy et al.57 In brief, samples were adjusted to pH 2 and analysed by direct injection using reverse phase chromatography and negative electrospray ionisation tandem mass spectrometry. For the analysis of 13C-HAAs the mass transitions were adapted as required and semi-quantitative results obtained from comparison of 13C-HAA responses with the equivalent unlabelled HAA responses. Analysis for four THMs used a procedure based on EPA 551.1 which entailed liquid–liquid extraction into MTBE and analysis by gas chromatography electron capture detection (GC-ECD). Further details are provided in section S1.2.6 Interpretation
Instrument responses were quantified against a calibration curve using an internal standard (2,3-DBPA for HAAs and 4-BFB for THMs) to give results as μg L−1. These were used to calculate the following metrics for results comparison:Formation potential – FP – μM DBP per mM model compound or μg DBP per mg carbon.
Bromine substitution factor – BSF – moles of bromine per mole of halogen present in DBPs formed (0–1).
van der Waals volume – V(vdW) – steric factor calculated to represent the size of the group(s) adjacent to the β-dicarbonyl moiety, using the calculations described by Zhao et al. (2003)58 as detailed in Table S1.
3 Results and discussion
3.1 DBPFP from aliphatic carbonyls
The substantial influence of molecular structure on type and levels of DBPs formed from aliphatic carbonyl precursor compounds is demonstrated and the pre-eminence of the β-dicarbonyl group is confirmed (Fig. 1). For example, where no β-ketone was present, minimal DBP formation occurred (median 0.14, range 0–3 DBPFP μM mM−1 from 3-hydroxybutanoic acid, methyl acetate, 3-aminobutanoic acid and acetone), but for all compounds with β-ketone, significant DBP formation occurred (median 926, range 221–2118 μM mM−1). The importance of electrophilic substitution at the α-carbon of β-ketones had previously been established,49 but this trial extended learning by determining how the molecular environment of this key group affected yield and crucially bromine incorporation into DBPs formed. Where only a β-ketone functional group was present the formation was predominantly THMs (average 404 μM mM−1 THMFP and only 44 μM mM−1 HAAFP from acetylacetone and 2,6-dimethyl-3,5-heptanedione). However, when molecular functionality adjoined the β-ketone then HAA formation dominated, with average 501, 654 and 1182 μM mM−1 of HAAFP from the 3 carbon β-keto acid, 3 carbon β-keto ester and >3 carbon β-keto ester groups respectively, and an average of <1, <1 and 40 μM mM−1 THMFP respectively. Notably, the >3 carbon β-keto acid group (3-oxobutanoic acid, 3-oxohenanedioic acid and 3-oxopentanedioic acid) had mixed formation of both groups of DBPs, mostly as THMs (average 686 μM mM−1) but also, in the case of 3-oxopentanedioic acid, HAAs (1079 μM mM−1 HAAFP for this compound, average 62 μM mM−1 for the other compounds). Where the precursor was already singly halogenated, HAAs remained the major outcome (average 983 μM mM−1 HAAFP and 29 μM mM−1 THMFP from methyl bromoacetate, diethyl bromomalonate, ethyl malonate chloride, ethyl 4-bromoacetoacetate and ethyl-4-chloroacetoacetate). However, when the precursor was triply halogenated, both THMs and tri-halogenated HAAs were formed (average THMFP 767 μM mM−1 and HAAFP 970 μM mM−1 from ethyl-4,4,4-trichloroacetoacetate and hexachloroacetone). Consequently, although hydrophilic aliphatic carbonyls can form both THMs and HAAs, THMFP is a poor proxy for HAAFP from these compounds. This finding is reflected in the poor correlation between THMs and HAAs in waters with low UV absorbance59 and the ineffectiveness of using THM levels to model HAA4 in real waters.60This pattern in HAAFP and THMFP was broadly retained when the compound concentration was adjusted to 1.5 μM and molar ratio bromide to compound moved from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 to 10
3 to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (Fig. 1c and d). The exceptions were 3-oxobutanoic acid and diethyl malonate which had a +160% THMFP and −58% HAAFP change respectively, and ethyl-4,4,4-trichloroacetoacetate which had a +50% THMFP and −64% HAAFP change respectively. This is posited to be due to the faster kinetics of bromination which promotes bromine incorporation on the terminal carbon for 3-oxobutanoic acid, increasing Br-THMFP. This highlights how kinetic factors may drive bromination over chlorination and consequently increase toxicity of chlorinated waters, with further research required to develop suitable kinetic models for aliphatic dicarbonyl compounds.61 Also, the steric restrictions of larger bromine on multiple halogenation at a sterically hindered α-carbon in diethyl malonate, reducing HAAFP, with both steric and kinetic factors affecting ethyl-4,4,4-trichloroacetoacetate (van der Waals volumes of the groups adjacent to the β-dicarbonyl calculated as 101 Å3 for diethyl malonate and 121 Å3 for ethyl-4,4,4-trichloroacetoacetate, but only 39 Å3 for 3-oxobutanoic acid, Table S1).
3 (Fig. 1c and d). The exceptions were 3-oxobutanoic acid and diethyl malonate which had a +160% THMFP and −58% HAAFP change respectively, and ethyl-4,4,4-trichloroacetoacetate which had a +50% THMFP and −64% HAAFP change respectively. This is posited to be due to the faster kinetics of bromination which promotes bromine incorporation on the terminal carbon for 3-oxobutanoic acid, increasing Br-THMFP. This highlights how kinetic factors may drive bromination over chlorination and consequently increase toxicity of chlorinated waters, with further research required to develop suitable kinetic models for aliphatic dicarbonyl compounds.61 Also, the steric restrictions of larger bromine on multiple halogenation at a sterically hindered α-carbon in diethyl malonate, reducing HAAFP, with both steric and kinetic factors affecting ethyl-4,4,4-trichloroacetoacetate (van der Waals volumes of the groups adjacent to the β-dicarbonyl calculated as 101 Å3 for diethyl malonate and 121 Å3 for ethyl-4,4,4-trichloroacetoacetate, but only 39 Å3 for 3-oxobutanoic acid, Table S1).
As expected, a notable change across the groups is observed in relation to the increase in proportion of Br-DBPs formed as the ratio of bromide to carbon increases. Consequently, the proportion of THMs which are brominated was an average of 24% at 15 μM compound while at 1.5 μM it was 94% (excluding pre-halogenated compounds). For HAAs, the equivalent proportions are 14% and 58%. This is significant for proposed HAA9 regulation, as the unregulated brominated HAA4 increase as a proportion of HAAs formed, from 13% to 37%, implying at higher ratio of organic matter to bromide, measurement of HAA5 will underestimate the levels of HAA9 and THM bromination will not translate to HAAs.
Within the HAAs formed, there was almost exclusive formation of di-halogenated HAAs (DXAAs), with this extending across the groups studied other than those already triply halogenated. DXAAs constitute 100%, 100%, 76%, 98%, 61% of HAAs from 3 carbon β-keto acids, 3 carbon β-keto esters, >3 carbon β-keto acids, 3 carbon β-keto esters and singly halogenated compounds respectively, but only 19% of the triply halogenated precursors. This is in line with existing research where hydrophobic precursors align with higher tri-halogenated HAAs (TXAAs) and hydrophilic precursors align more with DXAAs.62 The findings support a proposed discriminatory mechanism for DXAAs such as a 6-membered hydrogen bonded intermediate formed during decarboxylation (proposed in our previous work50 and detailed in Fig. S14). Confirmation is provided in lower formation from malonamic acid (201 μM mM−1) compared to malonic acid (814 μM mM−1), which is proposed to reflect the weaker hydrogen bonding of the amino group, as well as the slower reaction from the non-dissociated imine of malonamic acid when compared to the dissociated enolate of malonic acid. These factors relate to the relative electronegativity of oxygen and nitrogen (3.44 and 3.04 on the Pauling scale) whereby the more electronegative oxygen stabilises negative charge of the enolate and with two lone pairs of electrons forms hydrogen bonds more effectively. It is further proposed that such a concerted mechanism could be influenced by the pH at chlorination, with both acidic and basic conditions potentially promoting DXAA formation (Fig. S14).
Further insight into the influence of formation pathways on variability in overall HAAFP comes from comparisons between compounds within a group. To illustrate, malonic acid and its esters methyl and dimethyl malonate have demonstrably different formation patterns (DBPFP 816, 209 and 929 μM mM−1 respectively, as DXAAs). The results support the hypothesis that HAA outcome depends on the balance between the competing pathways for electrophilic substitution, decarboxylation and ester hydrolysis. Electrophilic substitution will be faster in the acid but slower than decarboxylation in the single ester, and of no consequence where decarboxylation is not possible in the double ester, represented visually in Fig. S9. In a further example, t-butyl acetoacetate is observed to form THMs (THMFP 19 and 67 μM mM−1 at 15 and 1.5 μM compound respectively) whereas THMs were not detected from the equivalent methyl and ethyl esters. This is proposed to be due to the influence of the ester group on ester hydrolysis, with t-butyl more readily hydrolysed due to the stability conferred on the carbocation intermediate. The product of this hydrolysis, 3-oxobutanoic acid, was known to form extensive THMs (THMFP 286 and 745 μM mM−1 at 15 and 1.5 μM compound respectively) and so this is proposed as a minor pathway only with HAAFP only slightly reduced in the t-butyl ester (Fig. S11). Consequently, these model compounds act as probe compounds for elucidating DBP formation patterns as well as demonstrating that small differences in molecular structure can significantly alter the DBP outcome. Note that these small differences have implications for models which predict formation of HAAs from the carbon content of the water and demonstrate potential underlying reasons for why universal models remain elusive, with commonly used indicators such as organic carbon and UV absorbance unlikely to reliably predict formation from aliphatic β-dicarbonyl HAA precursors.63 Improvements to increase accuracy of predictions would involve inclusion of HAA speciation and bromide, but ultimately this work suggests that measurement of HAA9 is required to reliably understand the risk from HAAs, with THMs and HAA5 failing to adequately represent the risk to consumers in waters containing bromide (section S3.3). Advancements in the analytical methods available to test HAA9 makes such analysis more accessible.57
3.2 Molecular-level influences on speciation
The acidity of groups adjacent to the β-dicarbonyls, and therefore extent of dissociation of the enol, is identified as the key molecular feature which differentiates between a tendency for full bromination or retention of a degree of chlorination. Comparison of the total result set from the experiments at 15 μM to those at 1.5 μM (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and 10
3 and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio bromide to model compound respectively) reveals, as expected, that relative brominated THM and HAA formation expands with higher ratio of bromide to compound, to the detriment of chlorinated THMs and HAAs. Consequently, the average BSF increases from 0.17 to 0.52 for THMs and from 0.15 to 0.44 for HAAs (Fig. 2a and b). The formation behaviour is linked to the molecular functionalities, with acidic compounds (represented by circles) favouring bromination where the ratio of bromide to compound increases, but esters (represented by triangles) less so. This proposed effect is demonstrated by comparing the pKa of the compounds against the BSF of the DBPs formed, with a correlation of R2 = 0.65 and R2 = 0.82 observed at 15 μM and 1.5 μM model compound respectively (section S3.2.6 Fig. S15a, for non-pre-halogenated compounds). The acidity of the compounds as represented by pKa is proposed to influence bromination as enolates increase the speed of the halogenation by electrophilic substitution when compared to enols, an effect observed when comparing phenol and phenolate compounds.55,64 This kinetic advantage promotes formation of the kinetic product which is brominated, over the thermodynamic product which is chlorinated. pKa values represent the stability of the conjugate base in the acid–base equilibrium of the compound and so are linked to the reducibility and will include the ability of the compound to stabilise the negative charge through resonance and inductive effects, which also promote enolate formation and are proposed to stabilise anionic leaving groups during cleavage. This study therefore provides confirmation of the potential for association between pKa and bromination of the DBPs formed when bromide and chlorine are present concurrently.65
3 ratio bromide to model compound respectively) reveals, as expected, that relative brominated THM and HAA formation expands with higher ratio of bromide to compound, to the detriment of chlorinated THMs and HAAs. Consequently, the average BSF increases from 0.17 to 0.52 for THMs and from 0.15 to 0.44 for HAAs (Fig. 2a and b). The formation behaviour is linked to the molecular functionalities, with acidic compounds (represented by circles) favouring bromination where the ratio of bromide to compound increases, but esters (represented by triangles) less so. This proposed effect is demonstrated by comparing the pKa of the compounds against the BSF of the DBPs formed, with a correlation of R2 = 0.65 and R2 = 0.82 observed at 15 μM and 1.5 μM model compound respectively (section S3.2.6 Fig. S15a, for non-pre-halogenated compounds). The acidity of the compounds as represented by pKa is proposed to influence bromination as enolates increase the speed of the halogenation by electrophilic substitution when compared to enols, an effect observed when comparing phenol and phenolate compounds.55,64 This kinetic advantage promotes formation of the kinetic product which is brominated, over the thermodynamic product which is chlorinated. pKa values represent the stability of the conjugate base in the acid–base equilibrium of the compound and so are linked to the reducibility and will include the ability of the compound to stabilise the negative charge through resonance and inductive effects, which also promote enolate formation and are proposed to stabilise anionic leaving groups during cleavage. This study therefore provides confirmation of the potential for association between pKa and bromination of the DBPs formed when bromide and chlorine are present concurrently.65
 | ||
Fig. 2 Comparison at different ratios of bromide to model compound (Br![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MC) for a) THM BSF b) HAA BSF c) Cl-THMs d) Br-THMs e) HAA5 f) unregulated HAA4. MC) for a) THM BSF b) HAA BSF c) Cl-THMs d) Br-THMs e) HAA5 f) unregulated HAA4. | ||
The variation in BSF is observed to a lesser extent for the pre-brominated model compounds, suggesting that steric influences may be significant in limiting multiple bromination at the α carbon. Interestingly, the tendency for retaining a degree of chlorination in HAAs was not observed in research by Chowdhury,66 which reported that where a high level of 2% seawater intrusion was modelled, bromoform increased to ≈90% of THMs and TBAA to ≈60% and DBAA ≈20% of HAAs. The high BSF of DBPs formed from this groundwater source (≈0.88) infers that precursor organic matter was more acidic, thus favouring full bromination.
While a clear shift from chlorinated to brominated THMs with increasing ratio of bromide to compound is observed (Fig. 2c and d and excepting the tri-chlorinated compounds), this picture is less distinct when considering the regulated and unregulated HAAs (Fig. 2e and f). Here, the increasing ratio of bromide to compound reduces the formation potential of the regulated HAA5 for all compounds except malonic acid, including those compounds already tri-chlorinated. However, there is not such a direct link to increased formation potential of the unregulated HAA4, despite all compounds within this group being brominated. For the ester precursor compounds and those singly chlorinated, increasing the ratio of bromide to compound does increase unregulated HAA4FP, but acid compounds lie closer to or below the parity line, as do the brominated and tri-chlorinated compounds. This reflects the presence of fully brominated and chlorinated HAAs in the HAA5 group, but mixed brominated and chlorinated HAAs in the unregulated HAA4. Consequently, although the acidic β-dicarbonyl compounds favour bromination, it is the non-dissociated esters which have increased significance for formation of the unregulated HAA4 by forming unregulated BCAA at the expense of the regulated DBAA. To illustrate, at 1.5 μM compound and 5 μM bromide, the ester groups of compounds formed an average of 826 μM mM−1 (range 195–1950) HAAs of which 41% was as the unregulated HAA4 but the acidic groups of compounds formed 432 μM mM−1 (range 53–889) of HAAs of which only 24% was as the unregulated HAAs. Real-world HAA9 data from UK water sources determined that 38% of unregulated HAA4 formed was as BCAA, suggesting precursors for this species may be significant for identifying where formation of HAA5 differs from HAA9 even where high bromide is present.60
This enhanced bromine incorporation of β-keto acids is posited to be due to a combination of both kinetic and steric factors. The faster reactions of the dissociated acids are proposed to favour electrophilic substitution with the more reactive bromine, thus giving more fully brominated DBPs. This is consistent with findings from phenolic compounds, where bromination is markedly faster than chlorination for the dissociated phenolate anion than the non-dissociated phenol,64 although further work is required to demonstrate this effect in β-dicarbonyl compounds and in particular how competition between the formation of the kinetically favoured brominated product and the thermodynamically favoured chlorinated product is influenced by molecular structure and chlorination conditions. In addition to kinetic effects, the steric effect of the bromine (atomic radius 1.85 Å compared to chlorine 1.76 Å) at the ester site results in slower hydrolysis because of the sterically hindered tetrahedral intermediate as well as the hindrance of the ester group itself on bromination at the α carbon.67 The proposal that steric effects influence the bromine incorporation of DBPs formed from aliphatic carbonyls was explored by calculating the van der Waals volume of the groups either side of the key β-dicarbonyl group and comparing to the BSF of the DBPs formed (Table S1 and section S3.2.6 Fig. S15b, for non-pre-halogenated compounds). The size of the groups influences the extent of chlorination over bromination in DBPs formed whilst not appearing to influence overall DBP yield, likely due to the pre-eminence of the tendency of β-dicarbonyls for halogenation. This coarse correlation (R2 = 0.66 at 15 μM and R2 = 0.44 at 1.5 μM model compound) is proposed to occur because bromination is hindered by larger groups adjacent to the α-carbon and so chlorination increases, with a commensurate decrease in BSF. Further work is required to demonstrate these proposals, but this understanding would provide insight into why HAA5 and HAA9 formation is not well corelated for some aliphatic dicarbonyls.
The impact of the ester group is significant when considering potential precursor organic matter in drinking water, particularly when identifying effective treatments for high bromide and hydrophilic organic matter sources such as lowland waters.10 Firstly, there is the potential that the more acidic groups may be more readily removed by charged based treatment options, such as coagulation or ion exchange.68 Secondly, the sources of such precursor compounds may be different. For example, plants such as legumes tend to form acids whereas metabolic biological processes form esters; similarly anthropogenic sources form acids during oxidative treatments whereas esters are more likely found in industrial discharges (Table 1). Notably, due to the high formation of the unregulated HAA4 from both acid and ester compounds, the extent of dissociation does not distinguish the precursors which form HAA5 from those which form HAA9.
Interestingly, unlike shorter chain acids where THMs were not formed, the longer chain β-keto-acids (3-oxobutanoic acid, 3-oxohexanoic acid and 3-oxopentanedioic acid) formed THMs at an average of 734 μM mM−1, while retaining the high BSFs observed with shorter chain equivalents (average BSF 0.76 at 1.5 μM compound). HAAFP was variable at 79, 16 and 862 μM mM−1 respectively. The shift to THM formation is congruent with the relative stability of the carbanion formed by decarboxylation, which enables further halogenation and THM formation. Without this decarboxylation step, the equivalent esters follow an alternate pathway to DXAA formation via electrophilic substitution, cleavage and hydrolysis (illustrated in section S3.2). Therefore, longer chain aliphatic carbonyls, such as may represent less degraded organic matter, can provide a route for the formation of THMs from hydrophilic, low UV absorbance waters. Further, it is proposed that such compounds can explain why in some waters there is limited predictive ability of hydrophilicity of organic matter for DBP formation.69
The study therefore demonstrates that β-dicarbonyl compounds are significant for both currently regulated THMs and HAAs and the brominated HAA4 proposed for regulation, demonstrating that hydrophilic organic matter cannot be overlooked as DBP precursors when bromide is present. However, as minor changes in molecular structure influence which groups of DBPs are formed, and the speciation within them, differentiation between THM and HAA5 and HAA9 precursors will be difficult using existing monitoring techniques. This difficulty has been observed in the poor ability of models to predict DBP formation from dissolved organic carbon (DOC) in waters containing organic matter of low aromaticity and UV absorbance.59 Similarly, in real waters, poor correlation has been observed between DBPFP and specific UV absorbance at 254 nm (SUVA254, the wavelength which is used to indicate aromaticity) in the low molecular weight fraction of water and, further, the bromination of DBPs varies with SUVA254 and is higher in waters of low SUVA254.70,71 Therefore the findings in this work support that future models for HAA9 would be improved by ensuring that aromatic and aliphatic (UV absorbing or not) organic matter is modelled differently and the precursors for the DXAAs and TXAAs are treated as separate groups. Such models must include the levels of bromide at the point of chlorination given the strong tendency for bromine incorporation of some aliphatic compounds (e.g. an average BSF of 0.79 for β-keto acids) and the demonstrated link between ratio of bromide to precursor and risk to consumer (section S3.3).72 Ultimately, however, this work indicates there may be limited existing measures which reliably predict HAA9 formation other than HAA9 itself.
3.3 Changing ratio to bromide
The importance of the ratio between bromide and compound, and by extension chlorine, is highlighted by the findings from the additional experiments on the β-keto acids malonic acid, 3-oxobutanoic acid and 3-oxopentanedioic acid at changing concentrations of compound and bromide (Fig. 3 with detail Fig. S1).For HAAs, where bromide remains fixed and compound concentration increases (Fig. 3a–c), a clear increase in HAAs is expected and observed, where HAAs are formed. Interestingly, at the lowest concentration of compound, where the ratio of bromide to chlorine is at its highest, it is the regulated DBAA which is dominant, with this shifting to unregulated BCAA then regulated DCAA as the compound concentration increases. To illustrate, for malonic acid, at 1 μM the major HAA formed is DBAA at 76% of the total HAAs, at 3 μM the main HAA is BCAA at 45% and by 6 μM it is DCAA at 43% of total HAAs (Fig. S1). Similarly, at fixed compound and increasing bromide, formation shifts from entirely Cl-HAAs to unregulated Br-HAAs then regulated Br-HAAs, with both regulated and unregulated Br-HAAs present at the highest bromide concentration (malonic acid 80% and 3-oxopentanedioic acid 52% DBAA at 10 μM bromide). Consequently, it is predicted that unregulated Br-HAAs will remain a significant fraction of HAAs formed at high, as well as low, ratio of bromide to compound with this behaviour very compound specific, hence regulatory control encompassing all brominated HAAs i.e. HAA9 is advisable.
For THMs the formation patterns are clear, with an increasing ratio of bromide to compound moving THM formation (where formed) from chloroform to bromoform via the singly and doubly brominated BDCM and DBCM. To illustrate, for 3-oxopentanedioic acid molar THM formation moved from maximum 100% chloroform with no bromide to 41% BDCM at 3.75 μM bromide, 48% DBCM at 6.25 μM bromide and 63% bromoform at 10 μM bromide, with a similar pattern observed for 3-oxobutanoic acid. Interestingly, for 3-oxobutanoic acid, bromide also increases the molar formation of THMs (from 0.33 μM with no bromide to 1.89 μM at 10 μM bromide), but this is not observed for 3-oxopentanedioic acid (1.62 μM with no bromide to 1.69 μM at 10 μM bromide). Such differentiation is proposed due to the enhanced electrophilic substitution capability of bromine resulting in halogenation at the terminal carbon for 3-oxobutanoic acid (section S3.2). This has significance for procedures such as blending of source waters, as an increase of bromide even without increasing organic matter can lead to a significant increase in THMs.73
The findings reported here have ramifications for water companies aspiring to meet both existing and proposed HAA9 regulation. Notably, with the risk of non-compliance with these mass-based targets is exacerbated by the larger mass of bromine than chlorine and the finding that both molar formation and bromine incorporation increase with bromide in some compounds. For instance, both 3-oxobutanoic acid and malonic acid show an approximate 4-fold increase in molar DBP formation potential as bromide levels increase from none to 10 μM (800 μg L−1), despite compound concentrations remaining the same (2 μM or 208 μg L−1 and 204 μg L−1 respectively). This is in contrast to previous research on hydrophobic organic matter precursors (humic acid), where the molar formation remained similar at increasing bromide74 and consequently, divergence between formation behaviour of aliphatic dicarbonyls and the more widely studied aromatic humic compounds is demonstrated. Given the higher molecular weight of bromine, this FP increase has an even bigger impact on the mass concentrations, with the DBPs increasing from 70 to 375 μg L−1 for malonic acid and from 56 to 443 μg L−1 from 3-oxobutanoic.
However, when the changes in molar FP and bromine incorporation are interpreted to differentiate between HAA5 and HAA9 precursors, it is noted that it is the ratio of bromide to compound which most influences the formation of the unregulated HAAs. Importantly, the maximum divergence is not observed at maximum bromide, but rather where the unregulated BCAA is optimised. To illustrate, for malonic acid the maximum HAA4 formation of 129 μg L−1 is at 3.75 μM bromide, with this dropping to 61 μg L−1 at 10 μM bromide. Consequently, water sources which may contain more hydrophilic organic matter should be prioritised for monitoring prior to regulatory change as neither THM nor HAA5 is a suitable proxy for HAA9 and will not reliably represent the risk from formation of the unregulated HAA4 (SI 3.3).
3.4 Future work
The findings presented here demonstrate the potential importance of hydrophilic β-dicarbonyl compounds on the formation of both the regulated and unregulated Br-HAAs and indicate the need for new monitoring parameters and wider adoption of standards for HAA9.60 These compounds have received less attention in terms of the key influences on their DBP formation, in part because of their lower formation of THMs but also due to the challenges in their measurement. Consequently, future work should focus on developing appropriate analytical methods for these polar compounds with a specific focus on β-dicarbonyls, as well as improving the existing models for predicting HAA9 formation. Improved analytical methods which quantify small chain β-keto acids and β-keto esters will allow identification of precursor sources and inform on precursor prevalence, as well as enabling more detailed kinetics studies for HAA9 formation from hydrophilic organic matter. To illustrate, by measuring aliphatic carbonyl precursor degradation under chlorination conditions and identifying the intermediates formed, transformation pathways and rate constants can be established for modelling DBP formation behaviour from this under-studied compound group. The ability to isolate by β-keto acid or ester compounds would also be highly informative, but may be challenging. Predictive models may be improved by separating out the factors which influence DXAA and TXAA formation, by allowing for different formation pathways from aliphatic and aromatic (high UV absorbing) precursors, and crucially by including bromide. Significantly, given the historical focus on THMs and hydrophobic organic precursor types, an improved dataset of HAA9 and water quality parameters and chlorination conditions will be required to develop these more effective models. Future research into the key precursor organic matter types for formation of the more toxic brominated HAAs is particularly pertinent given the predicted increases in bromide concentrations in source waters.46Moreover, the data presented here represents a significant resource for the development and testing of predictive models for formation of HAAs from hydrophilic model compounds, including their propensity for bromine incorporation, with both factors previously receiving reduced attention. It is proposed that factors such as the electronegativity of functional groups around the carbonyl, the pKa and the steric restrictions on halogenation at the α-carbon are all potentially significant factors to include in such predictive models.75,76 This work has proposed formation of a 6-membered intermediate which provides a discriminatory pathway for the formation of DXAAs from β-dicarbonyls, a route which will become increasingly important as regulation of HAAs is increased. Such a mechanism allows for promotion of HAA formation by chelating cations such as metal ions and allows for the pH at chlorination to influence DXAA formation. This would explain real world observations on the effect of metals and pH on HAA formation,77–79 with the stabilised β-dicarbonyl intermediate promoting DXAA formation and ester hydrolysis (Fig. S14). Notably, metals have also been observed to increase bromination in DBPs and so understanding this mechanism may be particularly significant for formation of the unregulated HAA4 or increasing the toxicity of the chlorinated water.13 Consequently, further work on the impact of pH and metal ions on Br-HAA formation, especially those present in treated water and distribution systems such as Fe2+ and Cu2+, would be beneficial and may provide an explanation of the conditions and source types where DXAA formation differs from that of TXAAs and THMs (as has been observed in real waters62).
It is also proposed that future work should investigate the implications of current and proposed treatment technologies on the formation and/or removal of aliphatic carbonyls as precursors for HAA9. Notably, processes which preferentially remove hydrophobic organics with minimal impact on bromide (for example coagulation or granulated activated carbon) should be carefully considered as these have the potential to increase the risk to consumers in high bromide water sources by increasing the ratio of bromide to hydrophilic organic compounds such as those studied here.74 Similarly, technologies with potential for formation of aliphatic carbonyls during treatment, such as advanced oxidation processes or ozonation, should measure the HAAFP of treated waters to assess whether risk is increased. Alternative treatments which could be investigated include those which have potential to degrade small organic compounds, such as biologically active carbon, or membrane technology including nanofiltration which could remove even small organic compounds. In particular, this work would suggest the high potential of anion exchange treatments for high HAA9 forming water sources due to the ability to remove both bromide and small, negatively-charged organics such as β-keto acids.80
4 Conclusions
New insight into the formation of brominated DBPs from the key compound class of aliphatic β-dicarbonyls is provided, revealing that dissociated β-keto acids demonstrate a strong propensity for full bromination in DBPs formed. This is attributed to the faster kinetics of bromination by electrophilic substitution induced by the dissociated enolate. Contrastingly, similar ester compounds, which are not dissociated at the pH of water treatment, were shown to maintain a degree of chlorination during DBP formation, even as ratio of bromide to chlorine increased. Both groups had a consistently high proportion of HAAs formed as DXAAs. This differing behaviour has importance when thinking about the proposed regulation of HAAs, as the β-keto acid class of compounds ultimately tend to DBAA and the regulated HAA5 as ratios of bromide to compound increase, but the equivalent esters do not. These preferentially form the unregulated BCAA, therefore providing a previously unidentified route by which regulated HAA formation is differentiated from unregulated. Further, the study identifies that aliphatic precursors for THMs and HAAs are generally different. THM formation is promoted where there is no functionality adjacent to a β-ketone group or where decarboxylation can promote halogenated terminal carbons, with these molecular features precluding HAA formation, meaning THMs do not provide an indicator of HAAs from aliphatic carbonyls. However, although the findings expand understanding of the formation routes for DBPs from aliphatic dicarbonyl compounds, they also highlight the current paucity of knowledge around the makeup of hydrophilic organic matter in water, and particularly the levels at the point of chlorination. It is proposed that future research should focus on developing the techniques required to better understand the nature and sources of hydrophilic organic matter and in particular the prevalence and type of β-dicarbonyl compounds. Such information must be paired with an understanding of the concentrations of bromide in treated drinking water sources at the point of chlorination, with both factors and the ratio between them predicted to influence Br-HAA formation potential. This becomes increasingly important as the regulatory environment moves towards inclusion of brominated HAAs, with the challenge compounded by the likelihood of increasing bromide levels in source waters.Author contributions
Polly Grundy: visualisation, investigation, methodology, formal analysis, data curation, writing – original draft, and writing – review & editing. Emma Goslan: conceptualisation, supervision, funding acquisition, writing – review & editing. Peter Jarvis; conceptualisation, supervision, funding acquisition, writing – review & editing. Bruce Jefferson: conceptualisation, supervision, funding acquisition, writing – review & editing. Pablo Campo: writing – review & editing. John Fawell: writing – review & editing. John Haley: supervision, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The processed data supporting this article have been included as part of the supplementary information (SI). See DOI: https://doi.org/10.1039/d5ew01262g.Further data for this article, including additional results and processed data, are available at https://doi.org/10.57996/cran.ceres-2745.
References cited in the SI are listed as ref. 81–105 in the article reference list.
Acknowledgements
The work was supported by UK Water Industry Research Ltd. (UKWIR) [TX/05/A/204] and Engineering and Physical Sciences Research Council (EPSRC) through their funding of the Water Infrastructure and Resilience (WIRe) Centre for Doctoral Training [EP/S023666/1]. MarvinSketch was used for drawing, displaying and characterising chemical structures and reactions, MarvinSketch 24.1.2, Chemaxon (https://www.chemaxon.com). Icons made by Freepik from https://www.flaticon.com.References
- M. J. Plewa, J. E. Simmons, S. D. Richardson and E. D. Wagner, Mammalian cell cytotoxicity and genotoxicity of the haloacetic acids, a major class of drinking water disinfection by-products, Environ. Mol. Mutagen., 2010, 51, 871–878 CrossRef CAS PubMed.
- European Commission, Directive (EU) 2020/2184 of the European Parliament and of the Council of 16 December 2020 on the quality of water intended for human consumption (recast), 2020 Search PubMed.
- DWQR, Drinking Water Quality in Scotland, Drinking Water Quality Regulator (DWQR), Scotland, 2024 Search PubMed.
- US EPA, Six-Year Review 3 Technical Support Document for Disinfectants/Disinfection Byproducts Rules, US EPA, 2016 Search PubMed.
- DWI, Drinking water quality standards in England and Wales: Advisory group recommendations, Drinking Water Inspectorate, 2024 Search PubMed.
- US EPA, Fourth Unregulated Contaminant Monitoring Rule, https://www.epa.gov/dwucmr/fourth-unregulated-contaminant-monitoring-rule, (accessed 11 September 2023).
- W. Chen and H.-Q. Yu, Advances in the characterization and monitoring of natural organic matter using spectroscopic approaches, Water Res., 2021, 190, 116759 CrossRef CAS PubMed.
- K. Dejaeger, J. Criquet, M. Vanoppen, C. Vignal, G. Billon and E. R. Cornelissen, Identification of disinfection by-product precursors by natural organic matter fractionation: a review, Environ. Chem. Lett., 2022, 20, 3861–3882 CrossRef CAS.
- M. Valenti-Quiroga, M. J. Farré and P. Roccaro, Upgrading water treatment trains to comply with the disinfection by-products standards introduced by the Directive (EU) 2020/2184, Curr. Opin. Environ. Sci. Health., 2024, 39, 100547 CrossRef.
- I. Carra, J. Fernandez Lozano, S. Johannesen, M. Godart-Brown, E. H. Goslan, P. Jarvis and S. Judd, Sorptive removal of disinfection by-product precursors from UK lowland surface waters: Impact of molecular weight and bromide, Sci. Total Environ., 2021, 754, 142152 CrossRef CAS PubMed.
- A. Knap-Bałdyga and M. Żubrowska-Sudoł, Natural Organic Matter Removal in Surface Water Treatment via Coagulation—Current Issues, Potential Solutions, and New Findings, Sustainability, 2023, 15, 13853 CrossRef.
- X. Liu, Z. Chen, L. Wang and J. Shen, Effects of metal ions on THMs and HAAs formation during tannic acid chlorination, Chem. Eng. J., 2012, 211–212, 179–185 CAS.
- J. Hu, Z. Qiang, H. Dong and J. Qu, Enhanced formation of bromate and brominated disinfection byproducts during chlorination of bromide-containing waters under catalysis of copper corrosion products, Water Res., 2016, 98, 302–308 CrossRef CAS PubMed.
- E. L. Marron, C. Prasse, J. V. Buren and D. L. Sedlak, Formation and Fate of Carbonyls in Potable Water Reuse Systems, Environ. Sci. Technol., 2020, 54, 10895–10903 CrossRef CAS PubMed.
- A. Li, X. Zhao, R. Mao, H. Liu and J. Qu, Characterization of dissolved organic matter from surface waters with low to high dissolved organic carbon and the related disinfection byproduct formation potential, J. Hazard. Mater., 2014, 271, 228–235 CrossRef CAS PubMed.
- B. Jurado-Sánchez, E. Ballesteros and M. Gallego, Occurrence of carboxylic acids in different steps of two drinking-water treatment plants using different disinfectants, Water Res., 2014, 51, 186–197 CrossRef PubMed.
- A. P. Deshmukh, C. Pacheco, M. B. Hay and S. C. B. Myneni, Structural environments of carboxyl groups in natural organic molecules from terrestrial systems. Part 2: 2D NMR spectroscopy, Geochim. Cosmochim. Acta, 2007, 71, 3533–3544 CrossRef CAS.
- J. Huang, A. M. Yeung, R. M. Bergenstal, K. Castorino, E. Cengiz, K. Dhatariya, I. Niu, J. L. Sherr, G. E. Umpierrez and D. C. Klonoff, Update on Measuring Ketones, J. Diabetes Sci. Technol., 2024, 18, 714–726 CrossRef CAS PubMed.
- A. U. Igamberdiev and A. T. Eprintsev, Organic Acids: The Pools of Fixed Carbon Involved in Redox Regulation and Energy Balance in Higher Plants, Front. Plant Sci., 2016, 7, 1042 Search PubMed.
- H. Chen, H. Uzun, A. T. Chow and T. Karanfil, Low water treatability efficiency of wildfire-induced dissolved organic matter and disinfection by-product precursors, Water Res., 2020, 184, 116111 CrossRef CAS PubMed.
- J. He, F. Wang, T. Zhao, S. Liu and W. Chu, Characterization of dissolved organic matter derived from atmospheric dry deposition and its DBP formation, Water Res., 2020, 171, 115368 CrossRef CAS PubMed.
- K. Hickenbottom, K. Pagilla and D. Hanigan, Wildfire impact on disinfection byproduct precursor loading in mountain streams and rivers, Water Res., 2023, 244, 120474 CrossRef CAS PubMed.
- K. Kawamura, E. Tachibana, K. Okuzawa, S. G. Aggarwal, Y. Kanaya and Z. F. Wang, High abundances of water-soluble dicarboxylic acids, ketocarboxylic acids and α-dicarbonyls in the mountaintop aerosols over the North China Plain during wheat burning season, Atmos. Chem. Phys., 2013, 13, 8285–8302 CrossRef.
- R. Sempére and K. Kawamura, Comparative distributions of dicarboxylic acids and related polar compounds in snow, rain and aerosols from urban atmosphere, Atmos. Environ., 1994, 28, 449–459 CrossRef.
- S. Bertilsson and L. J. Stefan, Photochemically produced carboxylic acids as substrates for freshwater bacterioplankton, Limnol. Oceanogr., 1998, 43, 885–895 CrossRef CAS.
- A. T. Chow, D. M. Leech, T. H. Boyer and P. C. Singer, Impact of Simulated Solar Irradiation on Disinfection Byproduct Precursors, Environ. Sci. Technol., 2008, 42, 5586–5593 CrossRef CAS PubMed.
- N. Corin, P. Backlund and M. Kulovaara, Degradation products formed during UV-irradiation of humic waters, Chemosphere, 1996, 33, 245–255 CrossRef CAS.
- S. Bertilsson and L. J. Tranvik, Photochemical transformation of dissolved organic matter in lakes, Limnol. Oceanogr., 2000, 45, 753–762 CrossRef CAS.
- J.-L. Liu and X.-Y. Li, Removal of soluble microbial products as the precursors of disinfection by-products in drinking water supplies, Environ. Technol., 2015, 36, 722–731 CrossRef CAS PubMed.
- H. L. Tang, Y.-C. Chen, J. M. Regan and Y. F. Xie, Disinfection by-product formation potentials in wastewater effluents and their reductions in a wastewater treatment plant, J. Environ. Monit., 2012, 14, 1515–1522 RSC.
- Y.-H. Chuang, A. Szczuka and W. A. Mitch, Comparison of Toxicity-Weighted Disinfection Byproduct Concentrations in Potable Reuse Waters and Conventional Drinking Waters as a New Approach to Assessing the Quality of Advanced Treatment Train Waters, Environ. Sci. Technol., 2019, 53, 3729–3738 CrossRef CAS PubMed.
- X. Huang, Z. Zhang, X. Ren, P. Gu, K. Yang and H. Miao, Nontargeted identification of disinfection by-product precursors from soluble microbial products in municipal secondary effluent, J. Cleaner Prod., 2023, 430, 139662 CrossRef CAS.
- A. T. Chow, Natural organic matter under human-influenced environments: Implications for future environmental quality research, J. Environ. Qual., 2021, 50, 1347–1350 CrossRef CAS PubMed.
- X. Liu, R. Liu, B. Zhu, T. Ruan and G. Jiang, Characterization of Carbonyl Disinfection By-Products During Ozonation, Chlorination, and Chloramination of Dissolved Organic Matters, Environ. Sci. Technol., 2020, 54, 2218–2227 CrossRef CAS PubMed.
- J. Houska, T. Manasfi, I. Gebhardt and U. von Gunten, Ozonation of lake water and wastewater: Identification of carbonous and nitrogenous carbonyl-containing oxidation byproducts by non-target screening, Water Res., 2023, 232, 119484 CrossRef CAS PubMed.
- S. J. Khan and H. S. Weinberg, Characterisation of carbonyl byproducts of drinking water ozonation, Water Sci. Technol.: Water Supply, 2007, 7, 95–100 CAS.
- T. Manasfi, J. Houska, I. Gebhardt and U. von Gunten, Formation of carbonyl compounds during ozonation of lake water and wastewater: Development of a non-target screening method and quantification of target compounds, Water Res., 2023, 237, 119751 CrossRef CAS PubMed.
- X. Zhong, C. Cui and S. Yu, The determination and fate of disinfection by-products from ozonation-chlorination of fulvic acid, Environ. Sci. Pollut. Res., 2017, 24, 6472–6480 CrossRef CAS PubMed.
- J. Molnar, J. Agbaba, B. Dalmacija, S. Rončević, M. Prica and A. Tubić, Influence of pH and ozone dose on the content and structure of haloacetic acid precursors in groundwater, Environ. Sci. Pollut. Res., 2012, 19, 3079–3086 CrossRef CAS PubMed.
- S. Mondal, E. Mackey and R. Hofmann, Peracetic acid to reduce disinfection by-product formation in drinking water, Environ. Sci.: Water Res. Technol., 2024, 10, 1499–1505 RSC.
- D. Palma, A. Khaled, M. Sleiman, G. Voyard and C. Richard, Effect of UVC pre-irradiation on the Suwannee river Natural Organic Matter (SRNOM) photooxidant properties, Water Res., 2021, 202, 117395 CrossRef CAS PubMed.
- X. Cheng, H. Dong and Z. Qiang, Formation and transformation of pre-chlorination-formed disinfection byproducts in drinking water treatment process, Sci. Total Environ., 2023, 904, 166241 CrossRef CAS PubMed.
- Y. Zhang, N. Zhang, P. Zhao and Z. Niu, Characteristics of molecular weight distribution of dissolved organic matter in bromide-containing water and disinfection by-product formation properties during treatment processes, J. Environ. Sci., 2018, 65, 179–189 CrossRef CAS PubMed.
- S. Zhu, H. Zheng, H. Sun, J. Liu, X. Ma, X. Li, Q. Li and A. M. Dietrich, Insights for booster chlorination strategy based on DBPs control in a large-scale water supply system, Sci. Total Environ., 2022, 833, 155001 CrossRef CAS PubMed.
- S. D. Richardson, A. D. Thruston, T. V. Caughran, P. H. Chen, T. W. Collette, T. L. Floyd, K. M. Schenck, B. W. Lykins, G. Sun and G. Majetich, Identification of New Ozone Disinfection Byproducts in Drinking Water, Environ. Sci. Technol., 1999, 33, 3368–3377 CrossRef CAS.
- A. C. Leri, O. Hettithanthri, S. Bolan, T. Zhang, J. Unrine, S. Myneni, D. R. Nachman, H. T. Tran, A. J. Phillips, D. Hou, Y. Wang, M. Vithanage, L. P. Padhye, T. Jasemi Zad, A. Heitz, K. H. M. Siddique, H. Wang, J. Rinklebe, M. B. Kirkham and N. Bolan, Bromine contamination and risk management in terrestrial and aquatic ecosystems, J. Hazard. Mater., 2024, 469, 133881 CrossRef CAS PubMed.
- C. Kolb, M. Pozzi, C. Samaras and J. M. VanBriesen, Climate Change Impacts on Bromide, Trihalomethane Formation, and Health Risks at Coastal Groundwater Utilities, ASCE-ASME J. Risk Uncertain. Eng. Syst., 2017, 3, 04017006 CrossRef.
- S. Chowdhury, T. M. F. Koyappathody and T. Karanfil, Removal of halides from drinking water: technological achievements in the past ten years and research needs, Environ. Sci. Pollut. Res., 2022, 29, 55514–55527 CrossRef CAS PubMed.
- E. R. V. Dickenson, R. S. Summers, J.-P. Croué and H. Gallard, Haloacetic acid and Trihalomethane Formation from the Chlorination and Bromination of Aliphatic β-Dicarbonyl Acid Model Compounds, Environ. Sci. Technol., 2008, 42, 3226–3233 CrossRef CAS PubMed.
- P. Grundy, P. Jarvis, J. Fawell, E. Goslan, J. Haley and B. Jefferson, Using model compounds to show how a change in thinking is required for regulation of brominated haloacetic acids in drinking water, J. Hazard. Mater., 2026, 505, 141496 CrossRef CAS PubMed.
- T. Bond, E. H. Goslan, S. A. Parsons and B. Jefferson, A critical review of trihalomethane and haloacetic acid formation from natural organic matter surrogates, Environ. Technol. Rev., 2012, 1, 93–113 CrossRef CAS.
- S. E. Duirk and R. L. Valentine, Modeling dichloroacetic acid formation from the reaction of monochloramine with natural organic matter, Water Res., 2006, 40, 2667–2674 CrossRef CAS PubMed.
- H. Zhai, X. Zhang, X. Zhu, J. Liu and M. Ji, Formation of Brominated Disinfection Byproducts during Chloramination of Drinking Water: New Polar Species and Overall Kinetics, Environ. Sci. Technol., 2014, 48, 2579–2588 CrossRef CAS PubMed.
- A. L. B. Forster, S. L. Wiskur and S. D. Richardson, Formation of Eight Classes of DBPs from Chlorine, Chloramine, and Ozone: Mechanisms and Formation Pathways, Environ. Sci. Technol., 2025, 59, 15594–15611 CrossRef CAS PubMed.
- J. Criquet, E. M. Rodriguez, S. Allard, S. Wellauer, E. Salhi, C. A. Joll and U. von Gunten, Reaction of bromine and chlorine with phenolic compounds and natural organic matter extracts – Electrophilic aromatic substitution and oxidation, Water Res., 2015, 85, 476–486 CrossRef CAS PubMed.
- W. Lipps, T. Baxter and E. Braun-Howland, in Standard Methods for the Examination of Water and Wastewater, American Public Health Association, 2017, vol. 24 Search PubMed.
- P. L. Grundy, P. R. Jarvis, B. Jefferson, J. Fawell, J. A. Haley and E. H. Goslan, A validated reverse-phase LC-MS/MS method for the analysis of haloacetic acids in drinking water: supporting the transition from HAA5 to HAA9, H2Open J., 2024, 7, 272–285 CrossRef.
- Y. Zhao, M. Abraham and A. Zissimos, Fast Calculation of van der Waals Volume as a Sum of Atomic and Bond Contributions and Its Application to Drug Compounds, J. Org. Chem., 2003, 68, 7368–7373 CrossRef CAS PubMed.
- G. Hua, D. A. Reckhow and I. Abusallout, Correlation between SUVA and DBP formation during chlorination and chloramination of NOM fractions from different sources, Chemosphere, 2015, 130, 82–89 CrossRef CAS PubMed.
- P. Grundy, P. Jarvis, J. Fawell, E. Goslan, J. Haley and B. Jefferson, Evaluation of the implications and efficacy of proposed changes to haloacetic acid drinking water regulation, Sci. Total Environ., 2026, 1028, 181741 CrossRef CAS PubMed.
- X. Zhu and X. Zhang, Modeling the formation of TOCl, TOBr and TOI during chlor(am)ination of drinking water, Water Res., 2016, 96, 166–176 CrossRef CAS PubMed.
- H. C. Hong, F. Q. Huang, F. Y. Wang, L. X. Ding, H. J. Lin and Y. Liang, Properties of sediment NOM collected from a drinking water reservoir in South China, and its association with THMs and HAAs formation, J. Hydrol., 2013, 476, 274–279 CrossRef CAS.
- A. Lacroix, T. Goli, D. Hanigan and K. Pagilla, Formation, speciation, and temporal variability of DBPs in drinking water distribution systems in the context of ASR operations and extended storage periods, Chemosphere, 2024, 364, 143154 CrossRef CAS PubMed.
- M. B. Heeb, J. Criquet, S. G. Zimmermann-Steffens and U. von Gunten, Oxidative treatment of bromide-containing waters: Formation of bromine and its reactions with inorganic and organic compounds — A critical review, Water Res., 2014, 48, 15–42 CrossRef CAS PubMed.
- P. Grundy, PhD, Cranfield University, 2025 Search PubMed.
- S. Chowdhury, Effects of seawater intrusion on the formation of disinfection byproducts in drinking water, Sci. Total Environ., 2022, 827, 154398 CrossRef CAS PubMed.
- S. W. Krasner, C. F. T. Lee, R. Chinn, S. Hartono, H. Weinberg, S. D. Richardson, J. G. Pressman, T. F. Speth, R. J. Miltner and J. E. Simmons, in Proceedings of 2008 AWWA Water Quality Technology Conference, AWWA, Denver, CO., 2008, pp. 1692–1708 Search PubMed.
- B. Bolto, D. Dixon, R. Eldridge, S. King and K. Linge, Removal of natural organic matter by ion exchange, Water Res., 2002, 36, 5057–5065 CrossRef CAS PubMed.
- D. M. Golea, A. Upton, P. Jarvis, G. Moore, S. Sutherland, S. A. Parsons and S. J. Judd, THM and HAA formation from NOM in raw and treated surface waters, Water Res., 2017, 112, 226–235 CrossRef CAS PubMed.
- M. S. Ersan, C. Liu, G. Amy and T. Karanfil, The interplay between natural organic matter and bromide on bromine substitution, Sci. Total Environ., 2019, 646, 1172–1181 CrossRef CAS PubMed.
- N. Ates, M. Kitis and U. Yetis, Formation of chlorination by-products in waters with low SUVA—correlations with SUVA and differential UV spectroscopy, Water Res., 2007, 41, 4139–4148 CrossRef CAS PubMed.
- Y. Yang, Y. Komaki, S. Y. Kimura, H.-Y. Hu, E. D. Wagner, B. J. Mariñas and M. J. Plewa, Toxic Impact of Bromide and Iodide on Drinking Water Disinfected with Chlorine or Chloramines, Environ. Sci. Technol., 2014, 48, 12362–12369 CrossRef CAS PubMed.
- T. X. H. Le, Y. Xiang, C. Shang, C. Lin and K. Wang, Tracing disinfection byproducts during shock chlorination of a seawater reverse osmosis (RO) desalination pilot and the blended and chlorinated potable water produced, Desalination, 2025, 609, 118861 CrossRef CAS.
- P. A. Neale and F. D. L. Leusch, Assessing the role of different dissolved organic carbon and bromide concentrations for disinfection by-product formation using chemical analysis and bioanalysis, Environ. Sci. Pollut. Res., 2019, 26, 17100–17109 CrossRef CAS PubMed.
- T. Bond and N. Graham, Predicting chloroform production from organic precursors, Water Res., 2017, 124, 167–176 CrossRef CAS PubMed.
- J. A. Cordero, K. He, K. Janya, S. Echigo and S. Itoh, Predicting formation of haloacetic acids by chlorination of organic compounds using machine-learning-assisted quantitative structure-activity relationships, J. Hazard. Mater., 2021, 408, 124466 CrossRef CAS PubMed.
- H. Zhang and S. A. Andrews, Catalysis of copper corrosion products on chlorine decay and HAA formation in simulated distribution systems, Water Res., 2012, 46, 2665–2673 CrossRef CAS PubMed.
- Y. Zhao, H. Yang, S. Liu, S. Tang, X. Wang and Y. F. Xie, Effects of metal ions on disinfection byproduct formation during chlorination of natural organic matter and surrogates, Chemosphere, 2016, 144, 1074–1082 CrossRef CAS PubMed.
- K. M. Boenisch-Oakes, R. S. Summers and S. V. Masters, Disinfection Byproducts in Direct Potable Reuse and Conventional Drinking Waters: Influence of pH, Cu2+, and Water Age under Distribution System and Premise Plumbing Conditions, Environ. Sci. Technol., 2025, 59, 22168–22179 CrossRef CAS PubMed.
- P. A. Neale, S. J. Khan, J. Li, D. M. O'Carroll and F. D. L. Leusch, Assessing the ability of advanced pre-treatment to reduce disinfection by-product formation and finished water toxicity, Water Res., 2025, 282, 123755 CrossRef CAS PubMed.
- Y. Zhao, M. Abraham and A. Zissimos, Fast Calculation of van der Waals Volume as a Sum of Atomic and Bond Contributions and Its Application to Drug Compounds, J. Org. Chem., 2003, 68, 7368–7373 CrossRef CAS PubMed.
- W. Lipps, T. Baxter and E. Braun-Howland, in Standard Methods for the Examination of Water and Wastewater, American Public Health Association, 2017, vol. 24 Search PubMed.
- P. L. Grundy, P. R. Jarvis, B. Jefferson, J. Fawell, J. A. Haley and E. H. Goslan, A validated reverse-phase LC-MS/MS method for the analysis of haloacetic acids in drinking water: supporting the transition from HAA5 to HAA9, H2Open J., 2024, 7, 272–285 CrossRef.
- A. Hodgeson and D. J. Munch, EPA METHOD 551.1: Determination of Chlorinated Disinfection Byproducts, Chlorinated Solvents, and Halogenated Pesticides/Herbicides in Drinking Water by Liquid-Liquid Extraction and Gas Chromatography with Electron-Capture Detection, Revision 1.0., 1995.
- E. R. V. Dickenson, R. S. Summers, J.-P. Croué and H. Gallard, Haloacetic acid and Trihalomethane Formation from the Chlorination and Bromination of Aliphatic β-Dicarbonyl Acid Model Compounds, Environ. Sci. Technol., 2008, 42, 3226–3233 CrossRef CAS PubMed.
- K. Kumar and D. W. Margerum, Kinetics and mechanism of general-acid-assisted oxidation of bromide by hypochlorite and hypochlorous acid, Inorg. Chem., 1987, 26, 2706–2711 CrossRef CAS.
- M. B. Heeb, J. Criquet, S. G. Zimmermann-Steffens and U. von Gunten, Oxidative treatment of bromide-containing waters: Formation of bromine and its reactions with inorganic and organic compounds — A critical review, Water Res., 2014, 48, 15–42 CrossRef CAS PubMed.
- J. Criquet, E. M. Rodriguez, S. Allard, S. Wellauer, E. Salhi, C. A. Joll and U. von Gunten, Reaction of bromine and chlorine with phenolic compounds and natural organic matter extracts – Electrophilic aromatic substitution and oxidation, Water Res., 2015, 85, 476–486 CrossRef CAS PubMed.
- U. Pinkernell and U. von Gunten, Bromate Minimization during Ozonation: Mechanistic Considerations, Environ. Sci. Technol., 2001, 35, 2525–2531 CrossRef CAS PubMed.
- Y. Lee and U. von Gunten, Transformation of 17α-Ethinylestradiol during Water Chlorination: Effects of Bromide on Kinetics, Products, and Transformation Pathways, Environ. Sci. Technol., 2009, 43, 480–487 CrossRef CAS PubMed.
- A. Bondi, van der Waals Volumes and Radii, J. Phys. Chem., 1964, 68, 441–451 CrossRef CAS.
- M. Deborde and U. von Gunten, Reactions of chlorine with inorganic and organic compounds during water treatment—Kinetics and mechanisms: A critical review, Water Res., 2008, 42, 13–51 CrossRef CAS PubMed.
- J. Li, J. Jiang, T. Manasfi and U. von Gunten, Chlorination and bromination of olefins: Kinetic and mechanistic aspects, Water Res., 2020, 187, 116424 CrossRef CAS PubMed.
- P. Grundy, P. Jarvis, J. Fawell, E. Goslan, J. Haley and B. Jefferson, Using model compounds to show how a change in thinking is required for regulation of brominated haloacetic acids in drinking water, J. Hazard. Mater., 2026, 505, 141496 CrossRef CAS PubMed.
- M. Deborde, S. Rabouan, H. Gallard and B. Legube, Aqueous Chlorination Kinetics of Some Endocrine Disruptors, Environ. Sci. Technol., 2004, 38, 5577–5583 CrossRef CAS PubMed.
- J. L. Acero, F. J. Benitez, F. J. Real, G. Roldan and E. Rodriguez, Chlorination and bromination kinetics of emerging contaminants in aqueous systems, Chem. Eng. J., 2013, 219, 43–50 CrossRef CAS.
- M. R. Rose, S. S. Lau, C. Prasse and J. D. Sivey, Exotic Electrophiles in Chlorinated and Chloraminated Water: When Conventional Kinetic Models and Reaction Pathways Fall Short, Environ. Sci. Technol. Lett., 2020, 7, 360–370 CrossRef CAS.
- T. Prasert, F. Kurisu and P. Phungsai, Characterizing the precursors of byproducts formed by chlorine and chlorine dioxide disinfection using unknown screening analysis with Orbitrap mass spectrometry, Sci. Total Environ., 2025, 959, 178296 CrossRef CAS PubMed.
- Y. Lu, J.-K. Liang, H.-Y. Wang, C. Wang, Z.-M. Song, Q. Hu and Q.-Y. Wu, Novel chlorinated disinfection byproducts from tannic acid: nontargeted identification, formation pathways, and computationally predicted toxicity, J. Hazard. Mater., 2022, 425, 127827 CrossRef CAS PubMed.
- H. Zhai and X. Zhang, Formation and Decomposition of New and Unknown Polar Brominated Disinfection Byproducts during Chlorination, Environ. Sci. Technol., 2011, 45, 2194–2201 CrossRef CAS PubMed.
- R. Liu, C. Tian, C. Hu, Z. Qi, H. Liu and J. Qu, Effects of bromide on the formation and transformation of disinfection by-products during chlorination and chloramination, Sci. Total Environ., 2018, 625, 252–261 CrossRef CAS PubMed.
- J. Jiang, J. Han and X. Zhang, Nonhalogenated Aromatic DBPs in Drinking Water Chlorination: A Gap between NOM and Halogenated Aromatic DBPs, Environ. Sci. Technol., 2020, 54, 1646–1656 CrossRef CAS PubMed.
- C. Tian, R. Liu, H. Liu and J. Qu, Disinfection by-products formation and precursors transformation during chlorination and chloramination of highly-polluted source water: Significance of ammonia, Water Res., 2013, 47, 5901–5910 CrossRef CAS PubMed.
- WHO, in Guidelines for drinking-water quality, Fourth edition incorporating the first and second addenda, World Health Organization, 2022, vol. 8 Search PubMed.
- S. Bull and R. Bevan, Toxicity Assessment of Brominated Haloacetic Acids, Tara Consulting, 2024, DOI:10.57996/cran.ceres-2756.
| This journal is © The Royal Society of Chemistry 2026 |