Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Advanced treatment of facultative lagoon effluent: microfiltration and nanofiltration for water reuse
Christopher B.
Yazzie
a,
Lauren
Vasquez
b,
Jack
Welchert
a,
Doorae
Lee
 a,
Maya L.
Begay
c,
Matthew L.
Begay
c,
Shazia Tabassum
Hakim
c,
Charles
Gerba
d and
Vasiliki
Karanikola
a,
Maya L.
Begay
c,
Matthew L.
Begay
c,
Shazia Tabassum
Hakim
c,
Charles
Gerba
d and
Vasiliki
Karanikola
 *a
*a
aChemical and Environmental Engineering Department, University of Arizona, Tucson, Arizona 85721, USA. E-mail: vkaranik@arizona.edu; Tel: +1 (520) 621 5881
bBiosystems Engineering Department, University of Arizona, Tucson, Arizona 85721, USA
cSchool of STEM, Diné College, Tuba City, Arizona 86045, USA
dDepartment of Environmental Science, University of Arizona, Tucson, Arizona 85721, USA
First published on 12th January 2026
Abstract
This study assessed the effectiveness of microfiltration (MF) as pretreatment combined with nanofiltration (NF) for tertiary treatment of secondary wastewater effluent (SWE) from a facultative lagoon system. The SWE was found to have turbidity levels which require pretreatment, and microfiltration was chosen in this study for its ability to remove particles to 1 micron, its low-cost and the ease of operation and maintenance. Three commercially available NF membranes (NF90, NF245, and NF270) were evaluated using a bench-scale system operated in recycle mode, in which a fixed batch volume of SWE obtained from Tuba City, Arizona, was continuously recirculated through each membrane module. The NF90 membrane demonstrated the highest rejection of organic and inorganic compounds and consistently maintained a 96% reduction in total dissolved solids throughout 7 days of operation. In contrast, the NF245 and NF270 membranes showed decreasing performance over time due to fouling. All membranes successfully removed more than 95% of contaminants of emerging concern. Characterization of the membranes indicated changes in hydrophobicity and surface charge for NF245 and NF270 after SWE filtering, while the properties of the NF90 membrane surface remained relatively unchanged. The treated effluents from NF245 and NF270 met Arizona's Class A+ reclaimed water standards for non-potable reuse. This study illustrates the potential of MF–NF treatment to upgrade facultative lagoon effluents to satisfy water reuse standards and to offer a sustainable solution for small communities experiencing water scarcity.
Water impactThis study addresses the challenge of freshwater scarcity by developing an innovative treatment process for the reuse of wastewater from facultative lagoons. The results show that water reuse of facultative lagoons can significantly enhance the availability of freshwater in arid regions, which have historically relied on groundwater for irrigation, and contribute to the advancement of water reuse technologies. |
Introduction
Water scarcity is a critical global issue, and the increasing demand for sustainable freshwater has prompted societies to seek alternative water sources.1 In arid regions, such as the southwestern United States, persistent drought conditions and over-extraction of groundwater have depleted aquifers, causing severe consequences, such as land subsidence in the Central Valley, CA, and parts of southern and central Arizona.2,3 To address these challenges, water resource managers have initiated the use of reclaimed wastewater to augment freshwater resources.4,5 Wastewater reuse provides an opportunity to meet non-potable water demands, particularly for agricultural irrigation, industrial processes, and landscape maintenance, offering a sustainable solution to mitigate the stress on the limited freshwater resources.6,7The use of reclaimed water is governed by strict guidelines to ensure public health and environmental protection. Worldwide, governmental agencies such as the United States Environmental Protection Agency (U.S. EPA), European Union (EU), China Ministry of Ecology and Environment, and World Health Organization (WHO) have established standards and guidelines for various water reuse applications.8–14 For non-potable water reuse applications, these standards typically focus on reducing contaminants, such as pathogens, nutrients, and inorganic pollutants, to levels that pose minimal risks to environmental and human health.15–18 A class of water contaminants, called contaminants of emerging concern (CECs), may have the potential to harm human health and the environment at concentrations into the picograms per liter (pg L−1), and can persist in the environment for extended periods, earning some the designation of “forever chemicals”.19–22 An example of these contaminants are per- and polyfluoroalkyl substances (PFAS), benzotriazole, carbamazepine, 1,4 dioxane and other pharmaceuticals and pesticides. Due to these regulatory frameworks, many wastewater treatment facilities, particularly lagoon-style systems in rural communities, face significant challenges in meeting reuse standards.23 Approximately one-quarter of U.S. Wastewater Treatment Plants (WWTPs) (approximately 4500) primarily serve small communities and operate lagoon-style facultative wastewater treatment systems.24 In a facultative lagoon system, the top, middle, and bottom zones are aerobic, facultative, and anaerobic zones, respectively.25,26 Facultative lagoon systems provide cost-effective secondary treatment, but the incorporation of tertiary treatment is essential to upgrade these effluents to meet reuse criteria, particularly for irrigation and other non-potable uses.
Several advanced tertiary water treatment technologies are available for improving wastewater effluents to meet water reuse standards.27,28 Tertiary treatment options can incorporate biological, chemical, and physical technologies. Tertiary biological treatments may include additional polishing ponds or membrane biological reactors (MBR) to reduce nutrients further.29 Although polishing ponds require low economic investment in operation and maintenance, they require additional land requirements and are susceptible to seasonal temperature variations that can reduce their efficiency.30,31 Tertiary chemical treatments include coagulation/flocculation, chemical precipitation, ozonation, chlorination, advanced oxidation, and ion exchange. These processes effectively remove contaminants but have drawbacks, such as high operational costs, complex operation, chemical storage and handling requirements, and sludge disposal issues.32,33 Tertiary physical treatment includes sedimentation, flotation, centrifugation, and filtration. Physical treatment methods are generally less effective at removing dissolved substances.34 The use of activated carbon is effective for charged ion contaminants and organics but requires periodic backwashing, regeneration or replacement.35,36 Filtration offers a wide range of contaminant removal methods based on particle and solute characteristics. Although filters can become clogged or fouled, effective mitigation techniques are available, and operation and maintenance are relatively inexpensive and straightforward.37,38
Filtration techniques have been employed throughout history, initially utilizing natural materials, such as fabric and clay vessels. Advancements in filtration technology have incorporated synthetic materials, such as in membrane filtration. Membrane filtration technologies have been established as reliable water treatment methods because of their capacity to remove pathogens, molecules, and individual ions.39–45 Nanofiltration (NF) membranes, in particular, offer a promising solution for achieving high levels of contaminant removal while maintaining moderate energetic requirements compared to reverse osmosis (RO).6 When NF was used to treat secondary wastewater effluent (SWE), the water product complied with the U.S. EPA, WHO, and other water reuse guidelines, and even removed CECs, which are not currently regulated.46–52 NF-treated SWE has been used for irrigation and industrial water in many areas of the world.7,50,53 Studies have reported membrane fouling and a reduction in water flux when NF and RO are used to treat SWE.50,54 One solution to the fouling challenge is the use of microfiltration (MF) as a pretreatment step to mitigate the fouling of NF membranes when treating SWE. In previous studies, when microfiltration (MF) was used for the treatment of SWE, it reduced the total organic carbon (TOC), biological oxygen demand (BOD), and total suspended solids (TSS); however, there was no reduction in the total dissolved solids (TDS).6,53,55,56
To date, there are limited peer-reviewed research studies evaluating the performance, fouling behavior, and water reuse potential of MF pretreated facultative lagoon secondary wastewater effluent. While prior studies have explored nanofiltration (NF) treatment of secondary effluent from urban, full-scale wastewater treatment plants, these systems differ significantly from lagoon-based treatment commonly used in remote and rural areas. Our work addresses this gap by demonstrating MF/NF treatment for lagoon effluent, providing a detailed analysis of water quality and potential irrigation applications. There are no comparable studies of water reuse of secondary effluent derived from facultative lagoons. The MF–NF configuration offers the potential to reduce operational challenges by enhancing the MF pretreatment stage and minimizing fouling during the NF stage. Additionally, this approach could provide an economical and scalable solution for small communities seeking to comply with water reuse standards. Investigating the feasibility and performance of this combined process is critical for addressing the gaps in the current body of knowledge and advancing the application of reclaimed water for sustainable use.
The objective of this study was to evaluate the effectiveness of MF pretreatment combined with NF membranes for tertiary treatment of facultative lagoon SWE. Batch mode bench scale experiments were conducted using real SWE to assess the performance of three commercially available membranes (NF90, NF245, and NF270). Analytical methods, including liquid chromatography-mass spectrometry (LC-MS), inductively coupled plasma mass spectrometry (ICP-MS), ion chromatography (IC), and total organic carbon (TOC) analysis, have been employed to characterize permeate water quality. The findings were compared with established water reuse guidelines to identify potential beneficial applications. By demonstrating the feasibility of the MF–NF process, this study contributes to the development of cost-effective strategies for upgrading facultative lagoon effluents to meet reuse standards, thereby enhancing water security and supporting environmental sustainability in underserved communities.
Materials and methods
Membrane materials
This study used six commercially available membranes: three MF filter cartridges and three NF membranes. Microfiltration (MF) was selected as a pretreatment for three principal reasons. First, MF systems exhibit operational simplicity and low maintenance requirements, which is critical for deployment in rural and remote contexts where technical capacity and resources may be limited; MF cartridge replacement can be performed with basic tools. Second, MF effectively removes suspended solids (>10 μm) as well as fine particulates (0.5–5 μm) thus mitigating fouling propensity and enhancing operational stability downstream of the NF. Finally, MF offers a cost-efficient alternative to chemical pretreatment, with readily available off-the-shelf components (e.g., filters, hoses, and fittings). The suitability of MF as a pretreatment to pressure-driven membrane processes has been reported in the literature for NF and RO applications.57,58 The MF filters selected in this study are porous polypropylene and had particle removal ratings of 10, 5, and 0.5 μm (McMaster Carr). The MF filters were selected based on preliminary pre-filtration tests, which helped to develop system operation protocol and water quality analysis techniques. MF filters were intended to remove larger particles to minimize fouling of the NF membrane surfaces. The three polyamide-based NF membranes are commonly referred to as NF90, NF245, and NF270 (Dow Filmtec). The nominal pore sizes of the NF membranes are considered to be less than 1 nm but generally exhibit a large geometric standard deviation.41 Prior to experimentation, all six membranes were soaked in DI water for 24 hours to ensure adequate hydration.59 The NF membranes were subsequently placed in custom-made membrane housings and operated with DI water (Milli-Q, Millipore Sigma) at 100 psi to facilitate adequate membrane compaction, which required approximately 24 hours. Compaction was assumed when permeate production stabilized, and results were verified using membrane performance data from previous studies.59 The baseline flux was measured after the 24 hour compaction period, and before the introduction of the SWE.Wastewater sampling and characterization
The Tuba City WWTP is located in Kerley Valley, Arizona, within the boundaries of the Navajo Nation, and serves approximately 9000 people. The facility operates a facultative treatment system with multiple lagoons and receives and releases approximately 2082 m3 per day (0.55 million gallons daily). The receiving waterway is Moenkopi Wash, an ephemeral stream that flows into the Little Colorado River 15 miles downstream. SWE was collected from the WWTP discharge point in April 2024. Approximately 22 L of SWE was collected in plastic containers, and the temperature and pH were recorded. The SWE (22 L) was then immediately transported to the University of Arizona, Tucson, Arizona, and filtration experiments commenced within 6 hours of sampling. Separately, 6 L of SWE was collected in 1 L plastic containers, and using a 2% nitric acid solution, the pH was adjusted to 2 for preservation of the sample to limit microbial growth, suppress precipitation of metal species, and stabilize trace inorganic analytes. This preserved low-pH SWE sample was stored at 4 °C until analytical chemistry analyses were performed.Analytical chemistry measurements
A Fisher Scientific Acumet AB150 meter was utilized to determine the pH and temperature of the SWE and treated wastewater samples. The electrical conductivity (EC) was measured using an Oakton PH 600 pH meter and is expressed throughout the results as total dissolved solids (TDS). Sample preparation for inorganic metal analysis consisted of first diluting the water sample 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 with 2% nitric acid solution and analyzing with inductively coupled plasma mass spectrometry (ICP-MS, Elan DRC-II), using U.S. EPA Method 6020B.60 Sample preparation for cation and anion analysis consisted of first diluting the water sample 10
1 with 2% nitric acid solution and analyzing with inductively coupled plasma mass spectrometry (ICP-MS, Elan DRC-II), using U.S. EPA Method 6020B.60 Sample preparation for cation and anion analysis consisted of first diluting the water sample 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 with DI water and quantifying it by ion chromatography (IC, Thermo Scientific Dionex ICS 5000) using U.S. EPA Method 300.1.61 CEC's were analyzed using Liquid Chromatography-Mass Spectrometry (LC-MS, Agilent 6490 LC/QQQ/MS) with a direct water injection method (U.S. EPA Method 900.1).62,63 Samples for CECE measurement were filtered through 0.2 μm polyethylene sulfonate (PES) filters, subsequently mixed with a mixture of isotopically labeled standard (internal standard) to correct for matrix effects. QA/QC routines were performed for each sample run, as outlined in the literature.64,65 The total organic carbon (TOC) concentrations in the feed and permeate were diluted 4
1 with DI water and quantifying it by ion chromatography (IC, Thermo Scientific Dionex ICS 5000) using U.S. EPA Method 300.1.61 CEC's were analyzed using Liquid Chromatography-Mass Spectrometry (LC-MS, Agilent 6490 LC/QQQ/MS) with a direct water injection method (U.S. EPA Method 900.1).62,63 Samples for CECE measurement were filtered through 0.2 μm polyethylene sulfonate (PES) filters, subsequently mixed with a mixture of isotopically labeled standard (internal standard) to correct for matrix effects. QA/QC routines were performed for each sample run, as outlined in the literature.64,65 The total organic carbon (TOC) concentrations in the feed and permeate were diluted 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 with DI, and then HCl was used to bring the sample to pH 4. The samples were then quantified by TOC analysis (TOC, Shimadzu TOC-L) U.S. EPA Method 9060A.66 Turbidity in the feed and permeate was determined by colorimetric analysis (UV-vis, Thermo Scientific Evolution 201) at 254 nm using U.S. EPA Method 900.67 A list of all the water quality characteristics that were quantified in this study using various analytical methods can be found at Table S4.
1 with DI, and then HCl was used to bring the sample to pH 4. The samples were then quantified by TOC analysis (TOC, Shimadzu TOC-L) U.S. EPA Method 9060A.66 Turbidity in the feed and permeate was determined by colorimetric analysis (UV-vis, Thermo Scientific Evolution 201) at 254 nm using U.S. EPA Method 900.67 A list of all the water quality characteristics that were quantified in this study using various analytical methods can be found at Table S4.
Microbiological analysis
Determination of Escherichia coli numbers was determined by the most probable numbers method (MPN) using the IDEXX Colilert system with Quantitray/200.68 Calculation of MPN/100 mL was done by consulting the MPN tables provided with the system. At specified time points throughout the experiment, water samples were collected from both the untreated SWE and the permeate produced by each NF membrane. The samples were promptly cooled to 4 °C and examined within 24 hours after collection. For examination, one mL of each dilution of water samples was spread over the surface of the respective nutrient agar plates using sterile glass beads, and incubated at 37 °C for 24–48 hours. After incubation, bacterial colony-forming units (CFUs) were counted using a colony counter, and bacterial concentrations were calculated based on the dilution factors.Analytical profile index (API) bacterial analysis was performed to identify the bacterial species present in the samples. The API 20E® identification system (bioMérieux, Inc.) was used according to the manufacturer's protocol.69 Isolated colonies were selected from the agar plates and suspended in sterile saline to make a bacterial suspension with turbidity equivalent to 0.5 MacFarland's index. The bacterial suspension was then transferred to API 20E test strip® capsules using sterile Pasteur pipettes and incubated at 37 °C for 18–24 hours. The results were recorded based on color changes in the wells, and biochemical profiles were interpreted using the APIweb® identification software to identify the bacterial species.
Membrane filtration experiments
Prefiltration of the SWE was conducted using a centrifugal pump at a flow rate of 1.5 L min−1 through three MF prefilters arranged in series, individually having a nominal pore size of 10 μm, 5 μm, and 0.5 μm, respectively (Fig. 1). Approximately 55 mL of water was collected from each prefilter. Temperature, pH, and conductivity measurements were taken immediately, and the samples were stored at 4 °C until further analysis. After pre-filtration, approximately 17 L was transferred to the feed tank of the bench-scale NF unit, as shown in Fig. 1. The solution in the feed tank was maintained at 20 °C during filtration.The NF bench scale unit included a feed tank with a capacity of 20 L. The NF system was operated continuously for seven days (168 hours), recycling the permeate and retentate streams back into the feed tank. Operating the system in recycling mode serves several purposes: it maintains relatively constant concentrations of solutes and contaminants, simulates the operational behavior of a full-scale system, and facilitates the study of fouling behavior through the accumulation of solutes.
Permeate sampling was conducted throughout the seven days of operation. Concurrent with the permeate flux measurements, EC and pH were also recorded. The NF system was operated continuously, and at each sampling event, the time required to generate 50 mL of permeate from the membrane modules was measured and documented over the seven-day operational period. Collected samples were then stored at 4 °C until further analytical chemistry measurements were performed.
A 240 volt centrifugal pump (Hydra-Cell, Minneapolis, MN) pressurizes the feed stream into three custom-made membrane housing modules arranged in a parallel configuration. Each membrane housing features internal dimensions of 25 × 45 mm. The flow rate across each membrane working area was 0.5 L min−1 with a calculated cross-flow velocity of 10.2 m s−1. Using the crossflow velocity and dimensions of the membrane housing, the Reynolds number was calculated as >3000, indicating a turbulent flow regime on the feed stream side of the membrane. Operating the system in a turbulent flow regime on the feed stream side prevents fouling because the fluid is well-mixed as it crosses and interacts with the membrane.70
Calculations
The permeate production during the SWE filtration experiments using different NF membranes was measured, and the permeate flux was calculated using the following equation: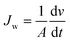 | (1) |
Rejection was calculated using the concentrations of solutes analyzed in both the feed and permeate of the filtration experiments.
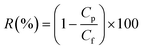 | (2) |
Membrane characterization
Two membrane characterization methods were used to assess the changes in membrane properties and to understand the effects of SWE filtration on the membranes. The first membrane analysis involved streaming zeta potential measurements (Anton-Parr SurPASS 3, Austria) using a flat sheet gap analyzer unit attachment to determine the differences in the electrical charge characteristics between the new and used NF membranes. The objective was to quantify the charge in the zeta potential region, which can indicate membrane fouling or other alterations in the membrane properties. All measurements used 1 mM HCl and NaOH buffers to adjust the pH of the solution. The pH was initially set to 10, and the average of four zeta potential measurements was recorded. This process was repeated at each pH interval, as the pH was incrementally decreased to approximately pH 4. All zeta potential measurements were conducted at 25 °C.The second membrane analysis involved contact-angle measurements (Kruss Drop Shape Analyzer, DSA25, Hamburg) to compare the new and used membranes. DI water was used as the droplet solution to obtain the average of four contact angle measurements. The objective was to determine changes in hydrophobicity, which could indicate membrane fouling or alterations in the membrane's surface properties. All the contact angle measurements were conducted at 25 °C.
Results and discussion
Water quality regulations
Water reuse practices are governed by guidelines and regulations designed to protect human and environmental health. This study compiled water reuse regulations and guidelines from the U.S. EPA and the State of Arizona, which are presented in Tables S1–S3. CECs, which consist of pharmaceuticals, organics, and other manufactured chemicals, are shown in Table S1; trace metals are shown in Table S2; and inorganic anions and cations, TOC, and TDS are shown in Table S3. The allowable concentrations of the various analytes depend on the specific application of reclaimed water.Water quality characterization
The SWE collected from the Tuba City WWTP was analyzed within 72 hours of sampling to ensure compliance with U.S. EPA water testing standards. Table 1 presents the analytes detected in the SWE and their corresponding concentrations in the permeate after treatment using three different NF membranes (NF90, NF245, and NF270). Reviewing Tables S1–S3 and 2 revealed that the electrical conductivity (EC), chloride (Cl−), turbidity, fecal coliform (E. coli), and nitrogen levels in the untreated SWE (Table 1) exceeded water reuse standards. Therefore, tertiary treatment is necessary before initiating any water reuse applications.| Analyte/parameter | Untreated SWE | Permeate of membrane treatment | ||
|---|---|---|---|---|
| NF90 | NF245 | NF270 | ||
| Note: ND (not detected) is used if no analytes were found in the sample or the concentration was below the detection limit for the analytical instrument. The limits of quantification (LOQ) for CEC are detailed in Appendix Table S5. The relative standard deviation (RSD%) derived from the mean of multiple measurements of all CEC was less than 15%. | ||||
| pH | 9.6 | 9.02 | 8.59 | 8.86 |
| E. coli [cfu per 100 mL] | 2.84 × 108 | 632 | 0 | 0 |
| EC (TDS) [μS cm−1] | 662 | 22.29 | 322.33 | 300.43 |
| Turbidity [NTU] | 113.5 | ND | ND | 0.05 |
| TOC [mg L−1] | 24 | ND | 0.03 | 0.26 |
| Na+ [mg L−1] | 105.6 | ND | 56.22 | 53.21 |
| NH4+ [mg-N L−1] | 5.4 | 0.24 | ND | 0.58 |
| K+ [mg L−1] | 26.7 | ND | 9.73 | 9.21 |
| Mg2+ [mg L−1] | 5.6 | ND | 1.51 | 1.56 |
| Ca2+ [mg L−1] | 26.7 | 0.14 | 7.31 | 6.87 |
| F− [mg L−1] | 0.04 | 0.05 | 0.10 | 0.11 |
| Cl− [mg L−1] | 104.5 | 5.76 | 82.12 | 73.06 |
| NO2− [mg-N L−1] | 5.5 | 0.52 | 1.02 | 1.13 |
| SO42− [mg L−1] | 58.4 | 0.33 | 0.99 | ND |
| Br− [mg L−1] | 1.3 | ND | ND | ND |
| NO3− [mg-N L−1] | 0.7 | 0.02 | 0.08 | 0.14 |
| Be [μg L−1] | 1.9 | 0.16 | 0.04 | 0.13 |
| Al [μg L−1] | 39.8 | ND | ND | ND |
| V [μg L−1] | 4.2 | 0.37 | 0.80 | 0.65 |
| Cr [μg L−1] | 0.2 | ND | 0.03 | ND |
| Mn [μg L−1] | 12.8 | 1.20 | ND | ND |
| Co [μg L−1] | 0.5 | 0.03 | ND | 0.02 |
| Cu [μg L−1] | 4.9 | ND | ND | ND |
| As [μg L−1] | 2.7 | 0.02 | ND | ND |
| Se [μg L−1] | 0.007 | ND | ND | ND |
| Mo [μg L−1] | 2.8 | 0.28 | ND | 0.46 |
| Ag [μg L−1] | 0.2 | 0.21 | 0.04 | 0.09 |
| Cd [μg L−1] | 0.04 | ND | ND | ND |
| Sn [μg L−1] | 0.08 | ND | ND | ND |
| Sb [μg L−1] | 2 | 0.10 | 0.19 | 0.02 |
| Ba [μg L−1] | 17.3 | ND | 5.09 | 4.23 |
| Tl [μg L−1] | 0.6 | 0.06 | 0.10 | 0.09 |
| Pb [μg L−1] | 2.7 | ND | 1.81 | 0.77 |
| U [μg L−1] | 0.95 | 0.05 | 0.14 | 0.15 |
| Benzotriazole [ng L−1] | 1366.9 | 271.1 | 1280.6 | 1131.1 |
| Caffeine [ng L−1] | 357.2 | 14.1 | 114.7 | 79.2 |
| Carbamazepine [ng L−1] | 50.3 | ND | 11.3 | 8.4 |
| Gemfibrozil [ng L−1] | 53.9 | ND | ND | ND |
| Sucralose [ng L−1] | 31898.9 | 420.2 | 2106.0 | 2802.9 |
| Sulfamethoxazole [ng L−1] | 90.7 | ND | ND | ND |
| Class | Direct reuse purposes | Turbidity | Fecal coliform | Nitrogen removal |
|---|---|---|---|---|
| Note: NA = not applicable. BOD5 and TSS are other water quality measurements with no specified limit for Arizona recycled water standards. All classes minimally require secondary treatment (class C), with class B requiring additional disinfection from class C, and classes B+, A, and A+ requiring additional nitrogen removal from class B. | ||||
| A+ | All direct reuse purposes listed | 24 h avg ≤2 NTU, with max ≤5 NTU | Non-detectable in 4 out of the last 7 daily samples | 5-Sample geometric mean of total nitrogen is <10 mg L−1 |
| Single sample max of 23 cfu per 100 mL | ||||
| A | Irrigation of food crops, recreational impoundments, residential, school, and open landscape irrigation, toilet and urinal flushing, fire protection systems, spray irrigation of an orchard or vineyard, commercial closed-loop air conditioning system, vehicle and equipment washing, and snowmaking. Class direct B and C purposes | 24 h avg ≤2 NTU, with max ≤5 NTU | Non-detectable in 4 out of the last 7 daily samples | NA |
| Single sample max of 23 cfu per 100 mL | ||||
| B+ | Any B & C direct reuse purposes | NA | ≤200 cfu per 100 mL in 4 out of the last 7 daily samples | 5-Sample geometric mean of total nitrogen is <10 mg L−1 |
| Single sample max of 800 cfu per 100 mL | ||||
| B | Surface irrigation of an orchard or vineyard, golf course irrigation, restricted access landscape irrigation, landscape impoundment, dust control, soil compaction and similar construction activities, pasture for milking animals, livestock watering (dairy animals), concrete and cement mixing, materials washing and sieving, and street cleaning | NA | ≤200 cfu per 100 mL in 4 out of the last 7 daily samples | NA |
| Single sample max of 800 cfu per 100 mL | ||||
| C | Pasture for non-dairy animals, livestock watering (non-dairy animals), irrigation of sod farms, irrigation of fiber, seed, forage, and similar crops, and silviculture | NA | ≤1000 cfu per 100 mL in 4 out of the last 7 daily samples | NA |
| Single sample max of 4000 cfu per 100 mL | ||||
The EC in the SWE was measured at 662 μS cm−1, which imposes slight to moderate restrictions on its use for irrigation due to the sensitivity of some crops to high salinity. Elevated EC levels can cause soil salinization and osmotic stress, which may result in reduced plant growth.71 Chloride was recorded at 104.5 mg L−1, exceeding the U.S. EPA water reuse regulations for reclaimed water. Chloride ions are small, highly mobile, and commonly found in natural waters because of the dissolution of natural salts such as sodium chloride and potassium chloride, as well as in wastewater due to the use of chlorination products for post-treatment disinfection. Although chloride is an essential micronutrient in plants, at high concentrations, it can lead to toxicity symptoms in leaves and inhibit nutrient uptake in roots. Moreover, long-term accumulation of chloride in the soil can restrict plant growth.72
Pretreatment of SWE
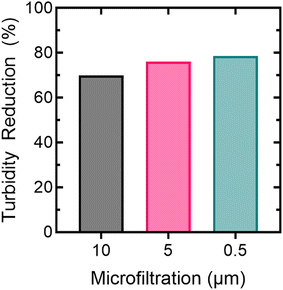
The turbidity of the collected SWE was measured at 113.5 NTU, and it contained suspended solids that were visually observable. Suspended solids found in the SWE can potentially foul membranes, leading to reduced water production and reduce contaminant rejection.73 To address membrane fouling caused by suspended solids, a pretreatment stage was implemented using a series of polypropylene MF modules with pore sizes of 10, 5, and 0.5 μm. While MF is not designed to remove TDS, as measured by EC, it is expected to reduce suspended solids that would otherwise foul the NF membranes. Fig. 2 illustrates the turbidity reduction achieved using each of the three MF filters.The 10, 5, and 0.5 μm MF filters reduced the turbidity by 68%, 75%, and 78%, respectively. As expected, no reduction in conductivity was observed, suggesting that the MF modules did not reduce the concentrations of metals, inorganics, or trace organics, all of which were dissolved constituents contributing to the measured EC of the water.74,75 The prefiltration effluent had a turbidity of 24.4 NTU and was immediately transferred to the 20 L NF bench-scale feed tank, as shown in Fig. 1.
NF permeate production
The NF system was operated continuously, and at each sampling event, the time required to generate 50 mL of permeate from the membrane modules was measured and documented over the seven-day operational period. The permeate flux results for each membrane during the experiment are shown in Fig. 3. | ||
| Fig. 3 Water flux comparison of NF90, NF245, and NF270 membranes during SWE treatment, which consisted of feed pressure of 100 psi, feed flow rate of 0.5 L min−1, and temperature of 20 °C. | ||
Baseline membrane permeate fluxes were determined using DI feed, and were calculated (eqn (1)) as 12.3, 83.4, and 79.5 L m−2 h−1 for the NF90, NF245, and NF270 membranes, respectively. After one hour into the experiment, the permeate flux decreased by approximately 50% for both NF245 and NF270, while NF90 showed only a slight decline. Over the first 72 hours, the permeate fluxes of the NF245 and NF270 membranes continued to decrease before stabilizing, whereas the NF90 stabilized after 48 hours. After one week (168 hours), the final water flux measurements were 9.8, 13.3, and 14.8 L m−2 h−1 for the NF90, NF245, and NF270 membranes, respectively.
After seven days of operation, permeate flux decreased by 20%, 84%, and 85% for the NF90, NF245, and NF270 membranes, respectively. These reduction trends align with previous studies that utilized RO to treat SWE.50,54 All membranes were operated in parallel under the same conditions, including an applied pressure of 100 psi, and feed flow rates to ensure turbulent flow (Reynolds number >3000) within the cell membranes. The observed flux decrease reflects the intrinsic membrane properties rather than operational variability. In practical applications, such as pilot-scale or full-scale membrane operations, maintaining constant transmembrane pressure is standard practice.
NF90, which has a comparatively tight polyamide active layer and lower permeability, exhibited the lowest flux decline. The NF245 and NF270 displayed substantially higher fluxes due to their looser polyamide active layer and higher permeability. Consequently, the NF245 and NF270 displayed more rapid fouling than the NF90 under the same operational pressure.
The overall decline in flux across all membranes can be attributed to residual suspended solids smaller than 0.5 μm passing through the MF pretreatment step. Despite implementing preventive measures, such as ensuring turbulent flow, foulant deposition still occurred, resulting in measurable membrane fouling. Incorporating membrane rinsing strategies in future work could prove beneficial; typically, rinsing is performed when the flux decreases by 20%.76 In our case, the NF245 and NF270 membranes could have benefited from rinsing after the first hour. Further analysis of the membrane surface was conducted to examine the changes in membrane surface hydrophobicity and surface charge.
Membrane characterization
Hydrophobicity describes the tendency of a membrane to repel water. This property is important because it can influence the fouling resistance of a membrane and, to some extent, the solute selectivity.77 Pre- and post-experimental contact angle measurements of the three membranes were conducted using a goniometer (Fig. 4A). Following exposure to SWE, the NF245 and NF270 membranes exhibited increased water contact angles relative to their pristine condition, indicating increased surface hydrophobicity. The membranes also operated at higher permeate fluxes than the NF90. This increase in hydrophobicity was likely due to membrane fouling caused by the adsorption and/or deposition of hydrophobic substances onto the membrane surface. Fouling layers composed of natural organic matter, proteins, and microbial extracellular polymeric substances can accumulate on membrane surfaces, thereby increasing hydrophobicity.78,79 These foulants tend to be hydrophobic, and their deposition increases the overall hydrophobicity of the membrane surface.80A decrease in permeate flux was observed for both the NF245 and NF270 membranes, indicating that the increase in hydrophobicity caused by fouling adversely affected membrane performance. In contrast, the NF90 membrane did not exhibit an increase in hydrophobicity compared to the pristine NF90 measurement; instead, a slight reduction of the water contact angle was observed. Initially, the NF90 membrane exhibited a higher baseline hydrophobicity than the NF245 and NF270 membranes. This greater initial hydrophobicity may have influenced the interaction between the membrane and foulants. A more hydrophobic membrane may reduce the adsorption of hydrophilic foulants, potentially mitigating fouling; however, hydrophobic membranes can exhibit increased adhesion of hydrophobic foulants due to hydrophobic–hydrophobic interactions.81
To further evaluate the effects of SWE on the membrane surface, zeta potential measurements were conducted before and after the experiments, with results presented in Fig. 4B–D. The NF245 and NF 270 membranes (Fig. 4C and D) both showed significant decreases in zeta potential measurements following exposure to SWE, indicating a reduction in negative surface charge. This reduction in surface charge is likely due to the adsorption of foulants onto the membrane surfaces, which can alter or neutralize the original negative charge of the membranes.81 The fouling layer may change the surface properties of the membrane, thereby affecting the electrostatic interactions between the membrane and solutes.82 A less negatively charged membrane surface may have reduced electrostatic repulsion against negatively charged foulants, facilitating further foulant deposition and increasing the fouling effects on the membrane.82
Foulants smaller than 0.5 μm, which can pass through the MF pretreatment, may include colloidal particles as small as 1 nm, composed of silt and biological tissues.83 These particles can accumulate on the membrane surface to form a fouling layer that alters the surface charge and hydrophobicity of the membrane.81,82 Changes in membrane surface properties due to fouling can lead to decreased permeate flux, as observed for the NF245 and NF270 membranes.
In contrast, NF90 (Fig. 4B) showed minimal changes in zeta potential across the tested pH range. The polyamide structure of NF90 is known to be similar to that of an RO membrane, characterized by a denser matrix due to its higher crosslinking density than the looser NF245 and NF270 membranes. This tight structure may limit the penetration of foulants into the membrane and the adsorption of foulants onto the membrane surface.84 The combination of a higher initial contact angle (indicating greater hydrophobicity) and a more negative zeta potential may enhance the fouling resistance of the NF90 membrane. A more negative surface charge can repel negatively charged foulants owing to electrostatic repulsion, while an increased hydrophobicity may diminish the adhesion of hydrophilic foulants. These synergistic effects can help sustain membrane performance by minimizing fouling-related flux decline. Furthermore, the lower permeate flux of the NF90 membrane may diminish the impact of concentration polarization, potentially leading to a decrease in membrane fouling.85
At constant operating pressure, changes to the membrane surface properties can significantly impact the permeate flux. Increased hydrophobicity may reduce water permeability because water molecules have less affinity for hydrophobic surfaces, leading to decreased flux. Similarly, a reduction in the negative surface charge can alter the selectivity and permeability of the membrane by affecting the electrostatic interactions with charged species in the feed water.86 Therefore, the decline in flux observed in the NF245 and NF270 membranes can be attributed to both physical blockage of pores by foulants and the changes in surface properties that hinder water transport. These factors may explain the lower reduction in water flux observed for the NF90 membrane than for the NF245 and NF270 membranes. The NF90 membrane exhibited greater resistance to fouling by maintaining its surface charge and hydrophobicity, resulting in a more stable permeate flux over time.
EC permeate analysis
To assess the permeate quality of each membrane, we monitored the EC as an indicator of TDS rejection (Fig. 5). The feed SWE initially exhibited a conductivity of 662 μS cm−1. Within the first hour, the NF90 membrane achieved a 96.6% reduction in EC and maintained this level of performance throughout the experiment, demonstrating its effectiveness for long-term EC removal. In contrast, the NF245 and NF270 membranes achieved EC reductions of 51.3% and 54.6%, respectively, after one hour. Over time, the NF245 and NF270 membranes exhibited increasing EC levels in the permeate, eventually stabilizing at 428 and 405 μS cm−1, respectively.The NF90 membrane effectively reduced permeate EC while maintaining consistent permeate production. In contrast, the NF245 and NF270 membranes experienced slight increases in permeate EC accompanied by significant decreases in permeate production. The gradual increase in permeate EC of the NF245 and NF270 membranes can be attributed to membrane fouling, which not only increases flow resistance, thus a decreased permeate flux, but may also lead to an increase in the effective pore size, as described in the literature.87,88 The increased contact angle indicates enhanced hydrophobicity, which may limit water's interaction with the membrane surface, negatively impacting water transport across the membrane. Additionally, fouling layers can physically obstruct pores and generate additional flow resistance, further diminishing permeate flux.81 The reduction in the negative zeta potential may alter the interactions between the membrane surface and charged solutes. Consequently, negatively charged solutes experience decreased electrostatic repulsion from the less negatively charged membrane surface, facilitating their transport through the membrane.82 It is important to compare all NF permeate flux data to illustrate how fouling impacts membrane rejection performance and to emphasize operational trade-offs which are important for real-world applications.
Collectively, these findings suggest that membrane fouling not only impacts the flux, but also compromises the rejection performance of the membranes, highlighting the need for comprehensive fouling mitigation strategies. Implementing effective cleaning protocols and optimizing operating conditions could help maintain membrane performance over the long term.89,90 These results underscore the importance of developing a cleaning protocol, particularly for the NF245 and NF270 membranes, to enhance their long-term performance.
NF permeate characteristics
Fig. 6 illustrates the rejection rates for each membrane, specifically highlighting two analytes from the untreated SWE that exceeded the water reuse standard. Additionally, Fig. 6 presents all CECs detected in the untreated SWE, along with their respective rejection rates for each membrane permeate.In all NF membrane permeate samples, turbidity and TOC levels were undetectable by the analytical chemistry methods employed in this study. The NF245 and NF270 membranes achieved Cl− removal percentages of 20% and 30%, respectively, while the NF90 membrane exceeded 95% removal. By analyzing the levels of Cl− and EC in the NF permeate against the water reuse standards in Table S3, we confirm that all three NF membranes have successfully reduced Cl− and EC to levels that comply with the regulations for using permeate in irrigation.
It is anticipated that multivalent ions will be removed more effectively than monovalent ions, as NF membranes utilize steric and Donnan charge exclusion mechanisms for ion rejection.41 Overall, the NF90 membrane demonstrated superior ion and molecule rejection compared to the NF245 and NF270 membranes, which is attributed to its smaller nominal pore size.91 These findings emphasize the importance of selecting membranes based on specific treatment objectives, especially when the removal of monovalent ions or smaller molecules is required.
Investigation of contaminants of emerging concern
We investigated the presence of 24 CECs, also known as trace organics, in the untreated SWE obtained from TC WWTP. Table 1 lists the analytes detected in untreated SWE. Benzotriazole is a corrosion inhibitor that is commonly found in industrial chemicals and dish-cleaning detergents and is among the identified CECs. Benzotriazole is persistent in the environment and has the potential to inhibit beneficial bacteria in soil biomes and wastewater treatment processes as well as induce estrogenic effects on marine life.92,93Carbamazepine, gemfibrozil, and sulfamethoxazole are pharmaceutical compounds used to treat various human ailments and can enter wastewater through human excretion or from the improper disposal of unused medications. Sulfamethoxazole, an antibiotic, contributes to the development of antibiotic-resistant bacteria in the environment. In addition, its presence in irrigation water may disrupt beneficial soil microbiomes, thereby affecting nutrient cycling and plant health.94 Chemicals that do not adequately degrade during wastewater treatment may remain in the effluent, and their presence in released SWE can potentially harm the environment and human health.95
Caffeine and sucralose are food additives known to pass through the human body and are not fully degraded by wastewater treatment processes because of their stable and chlorinated molecular structures.96,97 Research has shown that sucralose can serve as an indicator compound in wastewater effluent and in advanced treated SWE due to its resistance to degradation in conventional wastewater treatment systems. Additionally, it persists in the environment even after the effluent is released to the environment and has been linked to the inhibition of cyanobacterial growth and DNA damage to freshwater fish.98,99
Fig. 6 presents the rejection results for each membrane with respect to the CEC analytes identified in the untreated SWE. The NF90 membrane removed >95% of all CEC analytes, except for benzotriazole. As indicated in Table S6, benzotriazole has the lowest molecular weight among the CEC analytes at 119.2 g mol−1, and its pKa suggests it carries a positive charge at the experimental pH of 9.2. Both factors likely contributed to lower rejection of benzotriazole for all membranes tested, as lower molecular weight correlates with smaller molecular size, and a positive charge may lead to attraction towards the negatively charged membranes (Fig. 4).41 The NF245 and NF270 membranes displayed similar rejection trends for the other CEC analytes.
Caffeine has a molecular weight of 194.19 g mol−1, which is slightly larger than benzotriazole, and is hydrophilic, facilitating its dissolution in water, and may influence its transport through the NF245 and NF270 membranes. This characteristic could explain the rejection of caffeine by the NF245 and NF270 membranes at 66% and 75%, respectively. Carbamazepine is negatively charged and hydrophobic, both of which would limit its transport through the NF245 and NF270 membranes. However, its molecular weight of 236.27 g mol−1, which is near the molecular weight cutoff for NF245, and its shape may contribute to the observed rejection of carbamazepine of 77% and 81% for NF245 and NF270, respectively. Gemfibrozil and sulfamethoxazole have similar molecular weights of 250 and 253 g mol−1, respectively, and are positively charged at the experimental pH. Gemfibrozil and sulfamethoxazole compounds were also hydrophobic and exhibited 100% rejection rates, as their concentrations in the membrane permeate streams were below the detection limits of the LC-MS analytical method used in this study. Sucralose showed approximately 88% rejection for all three membranes. Sucralose is an interesting molecule to study because it is the largest CEC analyte detected in SWE, with a molecular weight of 397 g mol−1, and is hydrophilic and uncharged. Despite its relatively large molecular weight, the neutral charge of sucrose may facilitate its transport through membranes. A compilation of relevant information on CEC analytes is provided in the SI section (Tables S5 and S6).
Bacterial removal performance
Samples of untreated SWE and permeate from the NF membranes were analyzed for Escherichia coli using the MPN method (Table 1). The untreated SWE exhibited an MPN of 2.84 × 108 CFU per 100 mL, exceeding typical WWTP effluent concentrations.100 The elevated microbial count in the untreated SWE can be attributed to various factors, including insufficient retention time due to hydraulic or organic overloading, winter microbial dormancy, and spring sediment resuspension, as the sample was collected in April with an average temperature of 12.8 °C (55 °F).101–104After four hours of experimental operation, samples of the permeates from all three NF membranes were collected. The results indicated no detection of E. coli in 100 mL of the permeate from the NF245 and NF270 membranes, while the NF90 membrane permeate showed an MPN of 632 CFU per 100 mL. The extended duration required to gather approximately 50 mL of NF90 permeate (approximately 4 hours) at ambient temperature (∼25 °C) may have facilitated bacterial growth. In contrast, permeate collection for the NF245 and NF270 membranes took approximately 15 min and was immediately refrigerated at 4 °C, which likely explains the absence of detectable bacteria in those samples.
These findings suggest that the NF245 and NF270 membranes are effective at removing E. coli to less than 100 CFU per 100 mL under our experimental conditions. The presence of E. coli in the NF90 permeate may be attributed to post-filtration bacterial growth rather than membrane inefficiency. This highlights the importance of prompt sample handling and refrigeration to prevent bacterial proliferation during sample collection and analysis. Furthermore, the initial high microbial load in the SWE reinforces the need for tertiary treatment to remove pathogenic organisms before reuse applications.
Acceptable uses of NF-treated SWE
Advanced wastewater treatment allows for both potable and non-potable applications. Potable reuse can be either direct or indirect, with indirect reuse incorporating an environmental buffer. Currently, only a few locations, such as Texas, Namibia, and Singapore, are implementing direct potable reuse (DPR).105–107 However, future projections suggest an increased adoption as advanced wastewater treatment continues to improve. Non-potable applications of treated wastewater include irrigation, industrial processes, environmental and recreational uses, municipal applications, groundwater recharge, and construction activities.13 The value of treated wastewater increases with the extent of treatment; therefore, its intended use must be evaluated against a cost–benefit analysis, ensuring that treatment costs do not exceed the derived benefits.108–110 To mitigate the unintended adverse consequences of wastewater reuse, governmental bodies, and regional authorities have established regulatory guidelines that govern the implementation of wastewater reuse practices. As global water scarcity intensifies due to population growth and climate change, the reuse of treated wastewater has become increasingly vital for sustainable water resource management.Regulatory frameworks for wastewater reuse aim to protect human health and the environment by addressing potential hazards. Pathogens that survive wastewater treatment pose significant risks, and residual chemicals and nutrient contaminants can also have detrimental effects. CECs can persist through conventional wastewater treatment processes, and their long-term impacts remain largely unknown. Public perception plays a crucial role in the success of wastewater reuse programs, and negative incidents resulting from mismanagement can undermine confidence in water use. Therefore, stringent oversight, transparent communication, and robust safety measures are critical for fostering public trust and ensuring the responsible implementation of wastewater reuse initiatives.
Arizona, USA, has a population of 7.36 million people, with the majority (5 million, 68% of the state population) residing in metropolitan Phoenix. Approximately 41% of the state's water supply comes from groundwater, while 36% is sourced from the Colorado River via the 270-mile Central Arizona Project canal (CAP).111 Arizona's urban centers and agricultural regions have implemented wastewater reclamation for irrigation and groundwater recharge to supplement water availability.112 To ensure the safe reuse of treated wastewater, the Arizona Department of Environmental Quality (ADEQ) has established a classification system that defines permissible uses based on water quality criteria. Table 2 outlines the Arizona grading system and its guidelines for the non-potable reuse of reclaimed wastewater.
Evaluating the potential applications of NF-treated SWE requires consideration of both the Arizona classification system (Table 2) and the U.S. EPA guidelines (Tables S1–S3). By comparing the water quality results from Table 1 with these regulatory frameworks, Table 3 presents the classification and corresponding application of NF-treated SWE.
| Advanced water treatment (type of membrane) | U.S. EPA water reuse standards compliance | Arizona water reuse standards compliance | Arizona water reuse grade |
|---|---|---|---|
| A “+” symbol indicates that permeate produced from the NF membrane satisfies water reuse requirements. | |||
| NF90 | + | + | Grade B+ |
| NF245 | + | + | Grade A+ |
| NF270 | + | + | Grade A+ |
Bench-scale experiments in this study demonstrated that the NF245 and NF270 membranes produced class A+ reclaimed water. Further tests may reveal A+ class reclaimed water quality for NF90 membrane permeate. These classifications allow for various non-potable uses, including irrigation, landscaping, watering livestock, toilet flushing, commercial closed-loop air-conditioning systems, vehicle and equipment washing, and snowmaking. However, CECs are currently unregulated in water reuse applications. Detection of CECs requires specialized analytical methods, such as LC-MS, which may not be readily available in all locations. Furthermore, due to the extensive variety of CECs, ongoing research aims to identify indicator compounds that can serve as proxies for membrane filtration efficiency in CEC removal.114 Establishing reliable indicators can improve monitoring capabilities and support the integration of advanced treatment technologies, ensuring the safety and sustainability of reclaimed water for various applications.
Conclusion
The results of this study demonstrate the effectiveness of NF membrane technology in treating SWE to a quality suitable for reuse. MF, as a pretreatment method, effectively removed suspended particles, measured as turbidity. The subsequent application of NF membranes significantly reduced contaminants in SWE, making the permeate suitable for irrigation purposes. This study provides the foundation for developing a framework for the reuse of SWE for irrigation, which can contribute to the conservation of freshwater resources. By integrating NF membranes into advanced wastewater treatment processes, communities can enhance water sustainability and resilience to water scarcity. Further research is necessary to optimize the NF process and enhance the long-term performance of membranes.These findings have significant implications for wastewater management. NF membranes offer an effective solution for removing CECs that have not been eliminated or degraded by conventional wastewater treatment methods. This study highlights the potential of NF membranes as an advanced water treatment technology capable of conserving freshwater resources and enhancing the quality of treated effluents for irrigation purposes. In conclusion, NF membrane technology is a promising solution for treating SWE. Utilizing NF membranes can provide an environmentally friendly approach to water resource management while reducing the risk of agricultural and environmental contamination. The implementation of NF membranes in SWE treatment could be instrumental in addressing global water challenges by facilitating the safe reuse of treated wastewater, thus alleviating the pressure on freshwater supplies and contributing to regional agricultural economies.
Author contributions
C. B. Y.: conceptualization, data curation, formal analysis, methodology, visualization, validation, writing – original draft, writing, investigation. L. V.: investigation. J. W.: data curation, formal analysis, validation, methodology, writing – review and editing. D. L.: data curation, formal analysis, validation, methodology, writing – review and editing. Maya L. B.: investigation. Matthew L. B.: investigation. S. T. H.: conceptualization, writing – review and editing, supervision, project administration, funding acquisition. C. G.: conceptualization, writing – review and editing, funding acquisition. V. K.: conceptualization, writing – review and editing, supervision, project administration, funding acquisition.Conflicts of interest
The authors declare no conflicts of interest.Data availability
Data for this article, including flux, and rejection data are available at the KORES lab BOX repository.Data supporting the results are provided in the supplementary information (SI) and additional data (e.g., processed datasets, metadata) can be made available from the corresponding author upon request.
Supplementary information: the SI includes tables with detailed water quality analysis and relevant data for analytes. See DOI: https://doi.org/10.1039/d5ew01220a.
Acknowledgements
This study was funded by a grant from the United States Department of Agriculture, National Institute of Food and Agriculture-USDA Award number #2021-38424-33465 and a grant by the National Science Foundation, INFEWS: Indigenous Food, Energy, and Water Security and Sovereignty Award Number DGE #1735173. Supplemental funding for C. B. Y. was provided by the Alfred P. Sloan Foundation's Indigenous Graduate Partnership Fellowship.Notes and references
- M. M. Mekonnen and A. Y. Hoekstra, Sci. Adv., 2016, 2(2) DOI:10.1126/sciadv.1500323.
- G. Herrera-García, P. Ezquerro, R. Tomás, M. Béjar-Pizarro, J. López-Vinielles, M. Rossi, R. M. Mateos, D. Carreón-Freyre, J. Lambert, P. Teatini, E. Cabral-Cano, G. Erkens, D. Galloway, W.-C. Hung, N. Kakar, M. Sneed, L. Tosi, H. Wang and S. Ye, Science, 2021, 371(6524), 34–36 Search PubMed.
- Y. Wada, L. P. H. van Beek, C. M. van Kempen, J. W. T. M. Reckman, S. Vasak and M. F. P. Bierkens, Geophys. Res. Lett., 2010, 37, L20402 Search PubMed.
- Z. H. Wang, R. von Gnechten, D. A. Sampson and D. D. White, Sustainability, 2019, 11(13), 3537 CrossRef.
- P. Sardana, A. Javernick-Will and S. M. Cook, ACS ES&T Water, 2025, 5, 3–19 Search PubMed.
- K. Czuba, A. Bastrzyk, A. Rogowska, K. Janiak, K. Pacyna, N. Kossińska, M. Kita, P. Chrobot and D. Podstawczyk, Sci. Total Environ., 2021, 791, 148266 CrossRef CAS PubMed.
- A. Yasar, E. Can Dogan, H. S. Ayberk and C. Aydiner, Clean, 2022, 50, 2200280 Search PubMed.
- Ministry of Water and Electricity, Executive Regulations of Law of Treated Sewage Water and its Reuse Thereof, Kingdom of Saudi Arabia, 2015.
- Ministry of Environment Forest & Climate Change, Guidelines for Treated Wastewater Reuse, Central Pollution Control Board, Government of India, 2017.
- Ministry of Ecology and Environment of China, GB/T 18921-2002: Standards for the Reuse of Urban Recycling Water - Water Quality Standard for Urban Miscellaneous Water Consumption, People's Republic of China, 2002.
- Ministry of Land Infrastructure and Transport (MLIT), Guidelines for the Reuse of Treated Wastewater, Government of Japan, 2018.
- National Water Quality Management Strategy, Australian Guidelines for Water Recycling: Managing Health and Environmental Risks (Phase 1), Government of Australia, 2006.
- U.S. Environmental Protection Agency, Guidelines for Water Reuse EPA/600/R-12/618, 2012.
- European Union, Regulation (EU) 2020/741 of the European Parliament and of the Council of 25 May 2020 on minimum requirements for water reuse, 2020.
- S. D. Richardson and T. A. Ternes, Anal. Chem., 2014, 86, 2813–2848 Search PubMed.
- D. W. Kolpin, E. T. Furlong, M. T. Meyer, E. M. Thurman, S. D. Zaugg, L. B. Barber and H. T. Buxton, Environ. Sci. Technol., 2002, 36, 1202–1211 Search PubMed.
- M. Petrovic, M. Gros and D. Barcelo, J. Chromatogr. A, 2006, 1124, 68–81 Search PubMed.
- I. Ferrer and E. M. Thurman, J. Chromatogr. A, 2012, 1259, 148–157 CrossRef CAS PubMed.
- S. A. Snyder, Ozone: Sci. Eng., 2008, 30, 65–69 Search PubMed.
- S. Sauvé and M. Desrosiers, Chem. Cent. J., 2014, 8, 15 Search PubMed.
- K. Fent, A. Weston and D. Caminada, Aquat. Toxicol., 2006, 76, 122–159 Search PubMed.
- Y. Tang, Y. Zhong, H. Li, Y. Huang, X. Guo, F. Yang and Y. Wu, Water Environ. Res., 2020, 92, 1811–1817 Search PubMed.
- M. Schulhof, The Universe of Lagoons: An analysis of state and tribal lagoon wastewater treatment systems and socioeconomic, environmental justice, and compliance patterns in small, rural communities in the United States, https://www.epa.gov/system/files/documents/2022-06/universe-lagoons-report-2022.pdf, (accessed 3 September 2024).
- U.S. Environmental Protection Agency, Guidelines for Water Reuse, 2004, http://www.epa.gov.owm/ Search PubMed.
- S. R. Qasim, Wastewater Treatment Plants, Routledge, 2017 Search PubMed.
- G. Olsson and B. Newell, Wastewater treatment systems, IWA publishing, 1999 Search PubMed.
- D. Bixio, C. Thoeye, J. De Koning, D. Joksimovic, D. Savic, T. Wintgens and T. Melin, Desalination, 2006, 187, 89–101 Search PubMed.
- M. Jaramillo and I. Restrepo, Sustainability, 2017, 9, 1734 CrossRef.
- M. Prado, L. Borea, A. Cesaro, H. Liu, V. Naddeo, V. Belgiorno and F. Ballesteros, Int. Biodeterior. Biodegrad., 2017, 119, 577–586 CrossRef CAS.
- P. Verlicchi, M. Al Aukidy, A. Galletti, E. Zambello, G. Zanni and L. Masotti, J. Hydrol., 2012, 432–433, 127–136 CrossRef CAS.
- M. von Sperling and J. G. B. de Andrada, Water Sci. Technol., 2006, 54, 199–205 CrossRef CAS PubMed.
- S. F. Ahmed, M. Mofijur, S. Nuzhat, A. T. Chowdhury, N. Rafa, Md. A. Uddin, A. Inayat, T. M. I. Mahlia, H. C. Ong, W. Y. Chia and P. L. Show, J. Hazard. Mater., 2021, 416, 125912 Search PubMed.
- V. K. Gupta, I. Ali, T. A. Saleh, A. Nayak and S. Agarwal, RSC Adv., 2012, 2, 6380 RSC.
- D. P. Zagklis and G. Bampos, Processes, 2022, 10, 2304 CrossRef CAS.
- K. A. Thompson, K. K. Shimabuku, J. P. Kearns, D. R. U. Knappe, R. S. Summers and S. M. Cook, Environ. Sci. Technol., 2016, 50, 11253–11262 Search PubMed.
- E. M. Verdugo, M. Gifford, C. Glover, A. A. Cuthbertson, R. A. Trenholm, S. Y. Kimura, H. K. Liberatore, S. D. Richardson, B. D. Stanford, R. S. Summers and E. R. V. Dickenson, Water Res.: X, 2020, 9, 100068 CAS.
- M. E. Ersahin, H. Ozgun, R. K. Dereli, I. Ozturk, K. Roest and J. B. van Lier, Bioresour. Technol., 2012, 122, 196–206 CrossRef CAS PubMed.
- S. Hube, M. Eskafi, K. F. Hrafnkelsdóttir, B. Bjarnadóttir, M. Á. Bjarnadóttir, S. Axelsdóttir and B. Wu, Sci. Total Environ., 2020, 710, 136375 Search PubMed.
- R. J. Petersen, J. Membr. Sci., 1993, 83, 81–150 Search PubMed.
- N. G. Voros, Z. B. Maroulis and D. Marinos-Kouris, Desalination, 1996, 104, 141–154 CrossRef CAS.
- J. Schaep, B. Van der Bruggen, C. Vandecasteele and D. Wilms, Sep. Purif. Technol., 1998, 14, 155–162 Search PubMed.
- B. A. M. Al-Rashdi, D. J. Johnson and N. Hilal, Desalination, 2013, 315, 2–17 CrossRef CAS.
- N. Hilal, H. Al-Zoubi, N. A. Darwish, A. W. Mohamma and M. A. Arabi, Desalination, 2004, 170, 281–308 CrossRef CAS.
- M. Mohamed, M. Tagliabue and A. Tiraferri, Membranes, 2024, 14, 107 CrossRef CAS PubMed.
- S. Lu, A. McGaughey, S. Im, Y. Liu, X. Wang, A. Leininger, D. Jassby, E. Hoek and Z. J. Ren, Water Res., 2024, 265, 122306 CrossRef CAS PubMed.
- A. Egea-Corbacho, S. Gutiérrez Ruiz and J. M. Quiroga Alonso, J. Cleaner Prod., 2019, 214, 514–523 CrossRef CAS.
- M. C. Hacıfazlıoğlu, H. R. Tomasini, L. Bertin, T. Pek and N. Kabay, Desalination, 2019, 466, 89–96 CrossRef.
- S. M. Mrayed, P. Sanciolo, L. Zou and G. Leslie, Desalination, 2011, 274, 144–149 Search PubMed.
- J. L. Acero, F. J. Benitez, A. I. Leal, F. J. Real and F. Teva, J. Hazard. Mater., 2010, 177, 390–398 Search PubMed.
- E. C. Dogan, A. Yasar, U. Sen and C. Aydiner, Urban Water J., 2016, 13, 553–568 CrossRef CAS.
- J. L. Acero, F. Javier Benítez, F. J. Real and E. Rodriguez, Desalin. Water Treat., 2016, 57, 11685–11698 Search PubMed.
- M. Abbaszadegan, A. Alum, M. Kitajima, T. Fujioka, Y. Matsui, D. Sano and H. Katayama, Water, 2025, 17, 789 CrossRef CAS.
- M. Al-Shammiri, A. Al-Saffar, S. Bohamad and M. Ahmed, Desalination, 2005, 185, 213–225 CrossRef CAS.
- C. Jarusutthirak and G. Amy, Water Sci. Technol., 2001, 43, 225–232 CrossRef CAS PubMed.
- M. Monnot, B. Nguyen, F. Zaviska, G. Lesage and M. Héran, Desalin. Water Treat., 2017, 95, 18–33 Search PubMed.
- L. Crane, H. Ray, F. Perreault and T. H. Boyer, ACS ES&T Water, 2023, 3, 1835–1846 Search PubMed.
- F. Fatima, S. Fatima, H. Du and R. R. Kommalapati, Separations, 2024, 11, 115 Search PubMed.
- S. Lee and C. Lee, Sep. Sci. Technol., 2006, 41, 1–23 Search PubMed.
- C. B. Yazzie, C. Elias and V. Karanikola, Environ. Sci.: Water Res. Technol., 2024, 10, 2075–2086 Search PubMed.
- U.S. Environmental Protection Agency, Method 6020B (SW-846): Inductively coupled plasma-mass spectrometry, 2014.
- D. P. Hautman and D. J. Munch, Method 300.1 Determination of inorganic anions in drinking water by ion chromatography, U.S. EPA, 1997.
- J. Zou, B. Yao, S. Yan and W. Song, Environ. Pollut., 2022, 303, 119112 CrossRef CAS PubMed.
- K. T. Ng, H. Rapp-Wright, M. Egli, A. Hartmann, J. C. Steele, J. E. Sosa-Hernández, E. M. Melchor-Martínez, M. Jacobs, B. White, F. Regan, R. Parra-Saldivar, L. Couchman, R. U. Halden and L. P. Barron, J. Hazard. Mater., 2020, 398, 122933 Search PubMed.
- T. Anumol, S. Wu, M. Marques dos Santos, K. D. Daniels and S. A. Snyder, Environ. Sci.: Water Res. Technol., 2015, 1, 632–643 RSC.
- V. Martínez, D. Lee, I. Alyami, H. Zimila, F. Bautista, A. Fuentes, M. J. López, G. Valencia, D. Quanrud, R. G. Arnold and A. E. Sáez, Environ. Pollut., 2025, 367, 125622 CrossRef PubMed.
- U.S. Environmental Protection Agency, Method 9060A: Total Organic Carbon, 2004.
- J. O'Dell, Method 180.1 Determination of turbidity by nephelometry, US Environmental Protection Agency, 1993.
- IDEXX, Quanti-Tray/2000 inset and most probable number (MPN) table, 2013.
- bioMerieuz Inc, Analytical Profile Index (API) identification system, 2002.
- H. Choi, K. Zhang, D. D. Dionysiou, D. B. Oerther and G. A. Sorial, J. Membr. Sci., 2005, 248, 189–199 Search PubMed.
- S. R. Grattan and C. M. Grieve, Sci. Hortic., 1998, 78, 127–157 CrossRef.
- S. R. Corsi, L. A. De Cicco, M. A. Lutz and R. M. Hirsch, Sci. Total Environ., 2015, 508, 488–497 Search PubMed.
- A. I. Schäfer, A. G. Fane and T. D. Waite, Desalination, 2000, 131, 215–224 Search PubMed.
- F. Fatima, H. Du and R. R. Kommalapati, Water, 2021, 13, 1905 Search PubMed.
- R. H. Davis, in Current Trends and Future Developments on (Bio-) Membranes, Elsevier, 2019, pp. 29–67 Search PubMed.
- A. Al-Amoudi and R. W. Lovitt, J. Membr. Sci., 2007, 303, 4–28 CrossRef CAS.
- G. Hurwitz, G. R. Guillen and E. M. V. Hoek, J. Membr. Sci., 2010, 349, 349–357 CrossRef CAS.
- A. R. D. Verliefde, E. R. Cornelissen, S. G. J. Heijman, E. M. V. Hoek, G. L. Amy, B. Van der Bruggen and J. C. van Dijk, Environ. Sci. Technol., 2009, 43, 2400–2406 Search PubMed.
- S. Lee, S. Kim, J. Cho and E. M. V. Hoek, Desalination, 2007, 202, 377–384 Search PubMed.
- H.-C. Flemming, Appl. Microbiol. Biotechnol., 2002, 59, 629–640 Search PubMed.
- E. M. Vrijenhoek, S. Hong and M. Elimelech, J. Membr. Sci., 2001, 188, 115–128 Search PubMed.
- A. E. Childress and M. Elimelech, Environ. Sci. Technol., 2000, 34, 3710–3716 Search PubMed.
- C. Y. Tang, T. H. Chong and A. G. Fane, Adv. Colloid Interface Sci., 2011, 164, 126–143 CrossRef CAS PubMed.
- R. J. Petersen, J. Membr. Sci., 1993, 83, 81–150 Search PubMed.
- O. Peer-Haim, I. Shefer, P. Singh, O. Nir and R. Epsztein, Environ. Sci. Technol. Lett., 2023, 10(4), 363–371 Search PubMed.
- A. E. Childress and M. Elimelech, J. Membr. Sci., 1996, 119, 253–268 CrossRef CAS.
- G. Kang and Y. Cao, J. Membr. Sci., 2014, 463, 145–165 Search PubMed.
- B. Van der Bruggen and C. Vandecasteele, Environ. Pollut., 2003, 122, 435–445 Search PubMed.
- Q. Li and M. Elimelech, Environ. Sci. Technol., 2004, 38, 4683–4693 CrossRef CAS PubMed.
- A. Al-Amoudi and R. W. Lovitt, J. Membr. Sci., 2007, 303, 4–28 Search PubMed.
- R. Wang and S. Lin, J. Membr. Sci., 2021, 620, 118809 Search PubMed.
- A. G. Asimakopoulos, L. Wang, N. S. Thomaidis and K. Kannan, Environ. Int., 2013, 59, 274–281 Search PubMed.
- M. D. Alotaibi, A. J. McKinley, B. M. Patterson and A. Y. Reeder, Water, Air, Soil Pollut., 2015, 226, 226 CrossRef.
- P. Grenni, V. Ancona and A. Barra Caracciolo, Microchem. J., 2018, 136, 25–39 Search PubMed.
- K. Kümmerer, Chemosphere, 2009, 75, 417–434 CrossRef PubMed.
- A. Henderson, B. Ng, S. Landeweer, N. Quinete and P. Gardinali, Bull. Environ. Contam. Toxicol., 2020, 105, 351–357 CrossRef CAS PubMed.
- L. Soh, K. A. Connors, B. W. Brooks and J. Zimmerman, Environ. Sci. Technol., 2011, 45, 1363–1369 CrossRef CAS PubMed.
- G. Heredia-García, L. M. Gómez-Oliván, J. M. Orozco-Hernández, M. Luja-Mondragón, H. Islas-Flores, N. SanJuan-Reyes, M. Galar-Martínez, S. García-Medina and O. Dublán-García, Sci. Total Environ., 2019, 692, 411–421 CrossRef PubMed.
- A. G. Westmoreland, T. B. Schafer, K. E. Breland, A. R. Beard and T. Z. Osborne, Environ. Monit. Assess., 2024, 196, 451 Search PubMed.
- G. Tchobanoglus, F. Burton and H. D. Stensel, J. - Am. Water Works Assoc., 2003, 95, 201 Search PubMed.
- R. C. Leitão, A. C. Van Haandel, G. Zeeman and G. Lettinga, Bioresour. Technol., 2006, 97, 1105–1118 CrossRef PubMed.
- A. W. Mayo and T. Noike, Water Res., 1996, 30, 447–455 Search PubMed.
- K. Smyth, R. Vendramelli, D. Dankewich and Q. Yuan, J. Environ. Manage., 2018, 214, 224–231 CrossRef CAS PubMed.
- H. Ryu, Y. Addor, N. E. Brinkman, M. W. Ware, L. Boczek, J. Hoelle, J. H. Mistry, S. P. Keely and E. N. Villegas, Water, 2021, 13, 1452 Search PubMed.
- D. Ghernaout, N. Elboughdiri and A. Alghamdi, OAlib, 2019, 06, 1–10 Search PubMed.
- Direct Potable Reuse Water|Nuvonic, https://www.nuvonicuv.com/direct-potable-reuse-water/, (accessed 19 April 2025).
- Countries that recycle wastewater into drinking water - Université de Montpellier, https://www.umontpellier.fr/en/articles/ces-pays-qui-recyclent-les-eaux-usees-en-eau-potable, (accessed 19 April 2025).
- Q. K. Tran, K. A. Schwabe and D. Jassby, Environ. Sci. Technol., 2016, 50, 9390–9399 CrossRef CAS PubMed.
- N. Voulvoulis, Curr. Opin. Environ. Sci. Health, 2018, 2, 32–45 CrossRef.
- A. A. Keller, Y. Su and D. Jassby, ACS ES&T Eng., 2022, 2, 273–291 Search PubMed.
- The future of water in Arizona|ASU News, https://news.asu.edu/20221115-arizona-impact-future-water-arizona?utm_source=chatgpt.com, (accessed 28 January 2025).
- J. Cusimano, J. E. McLain, S. Eden and C. M. Rock, Agricultural Use of Recycled Water for Crop Production in Arizona, Arizona Cooperative Extension, June 2015 Search PubMed.
- Arizona Administrative Code|Article 3 - RECLAIMED WATER QUALITY STANDARDS |Casetext, https://casetext.com/regulation/arizona-administrative-code/title-18-environmental-quality/chapter-11-department-of-environmental-quality-water-quality-standards/article-3-reclaimed-water-quality-standards, (accessed 15 September 2024).
- P. Verlicchi, V. Grillini, E. Lacasa, E. Archer, P. Krzeminski, A. I. Gomes, V. J. P. Vilar, M. A. Rodrigo, J. Gäbler and L. Schäfer, Sci. Total Environ., 2023, 873, 162359 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |