 Open Access Article
Open Access ArticleFull-scale hybrid membrane aerated biofilm reactor (MABR) upgrade enhances the removal of organic micropollutants
Narasimman
Lakshminarasimman†
 *a,
Sondus
Jamal
a,
Leslie
Bragg
b,
Mark
Servos
b and
Wayne
Parker
*a
*a,
Sondus
Jamal
a,
Leslie
Bragg
b,
Mark
Servos
b and
Wayne
Parker
*a
aDepartment of Civil and Environmental Engineering, University of Waterloo, Waterloo, ON, Canada. E-mail: Narasimman.lakshminarasimman@stantec.com; wjparker@uwaterloo.ca
bDepartment of Biology, University of Waterloo, Waterloo, ON, Canada
First published on 2nd March 2026
Abstract
Hybrid membrane aerated biofilm reactors (MABRs) integrate a range of redox conditions with increased biomass residence times in biofilms to create conditions that are expected to enhance the removal of organic micropollutants (OMPs). However, the removal of OMPs with MABRs remains unexplored. This study monitored the behavior of 20 OMPs over multiple seasons, both before and after the upgrade of a full-scale conventional activated sludge (CAS) facility to a hybrid MABR. More than half of the target compounds (13 out of 20) were present at lower concentrations in the effluent after MABR addition when compared to pre-upgrade operation. Plantwide removal efficiencies of highly removable compounds (>75%) (acetaminophen, ibuprofen, naproxen, triclosan, triclocarban, and norfluoxetine) and the recalcitrant carbamazepine were not impacted by the addition of the MABR process. However, the removal of three endocrine disruptors (bisphenol A, estrone, and estradiol) improved from a moderate removal (25 to 75%) to a high removal. Further, removal of an additional seven pharmaceuticals (gemfibrozil, sulfamethoxazole, trimethoprim, diclofenac, atorvastatin, and its ortho-hydroxy metabolites) improved from poor (<25%) to high or moderate removal with the hybrid MABR/CAS configuration. After the MABR upgrade, it was found that six OMPs exhibited higher removal rates under warm weather conditions (19.3 ± 1.6 °C) compared to cold weather conditions (13 ± 1.2 °C). Mass balance analysis on the MABR alone revealed a broad range of compound-specific responses, such as complete biotransformation (acetaminophen), partial removal (naproxen), and potential transformation from conjugates (sulfamethoxazole, carbamazepine). Overall, the three-year monitoring of the full-scale facility before and after the upgrade revealed that upgrading the CAS to a hybrid MABR configuration can enhance the removal of some OMPs that are poorly removed by the CAS process alone.
Water impactMABRs are increasingly adopted to intensify nitrogen removal under reduced energy and technology footprint. However, their potential for removing harmful organic micropollutants remains poorly understood. This study demonstrated that upgrading a full-scale conventional activated sludge process with a hybrid MABR can enhance the removal of pharmaceuticals, personal care product ingredients, and endocrine disruptors. |
1. Introduction
Organic micropollutants (OMPs) are a broad class of anthropogenic contaminants that occur at ng L−1 to μg L−1 concentrations in natural waters due to their everyday use as pharmaceuticals, personal care product ingredients (PPCP), natural and synthetic hormones, surfactants, and biocides.1 Although their concentrations are typically low, the presence of OMPs is concerning because they are often biologically active with persistent, bioaccumulative, toxic, and endocrine-disrupting characteristics.2–5 Water resource recovery facilities (WRRFs) can be conveyors of these contaminants in the environment, as they are not normally designed to treat them.6,7 Commonly adopted treatment technologies, such as conventional activated sludge (CAS), have been found to only remove some OMPs, with the extent of removal dependent on their chemical structure and treatment operating conditions.8 Toxicological surveys in wastewater effluent-impacted streams have shown that WRRF upgrades to improve nitrification can also reduce the presence of some OMPs and decrease the incidence of adverse reproductive effects in fish.9,10 Hence, there is an interest in assessing whether emerging nutrient removal technologies can further reduce the discharge of OMPs to watersheds and protect aquatic ecosystems.Prior studies have shown that advanced biological wastewater treatment processes can enhance OMP removal when compared to the CAS configuration. Biological nutrient removal (BNR) configurations that include multiple redox zones have been found to enhance the biotransformation of some OMPs in the anoxic and anaerobic zones11–13 and also reduce effluent toxicity when compared to CAS.14 This is because of the recirculation of activated sludge between different redox zones that enables facultative heterotrophic organisms and their diverse enzyme pools with broad specificity to metabolize OMPs even under low dissolved oxygen (DO) conditions.15 Other treatment designs, such as membrane bioreactors (MBR) characterized by high mixed liquor suspended solids (MLSS) concentration and longer solids retention time (SRT), have also shown higher removal of OMPs due to the growth of slow-growing organisms.16,17 Similarly, biofilm based processes such as the moving bed biofilm reactor (MBBR) and integrated fixed film activated sludge system (IFAS) have been reported to achieve higher removals and more rapid biotransformation of OMPs when compared to suspended growth systems.18–20 The longer biomass retention time in the biofilms and the stratified microbial communities, along with substrate and redox gradients, have been identified as possible reasons for improved removal of OMPs in biofilm-based treatment processes.21 Overall, advanced biological treatment designs favor the growth of a more diverse microbial community that eventually improves the removal of a broader class of OMPs when compared to CAS systems.
The membrane aerated biofilm reactor (MABR) process integrates the beneficial features of several advanced secondary treatment technologies. MABRs support biofilm growth on an oxygen-permeable membrane that can result in a stratification of redox zones associated with the establishment of an oxygen gradient in the biofilm. The presence of biofilms can also increase the overall SRT to support the growth of diverse, slow-growing microorganisms. The potential for enhanced OMP degradation in a stratified biofilm could be further enhanced by immersing the biofilm in an anoxic suspended growth basin (hybrid design) that would further support the removal of pollutants through metabolic pathways associated with the presence of multiple redox conditions.22 Mei et al. (2020) observed a stepwise degradation of nitroaniline in a stratified MABR where the parent compound was reduced to form phenylenediamine in the anaerobic layer of the biofilm that was further oxidized in the aerobic layer. The nitrate that was formed as a secondary pollutant was denitrified in the anoxic layer.23 Thus, hybrid MABR systems could potentially be advantageous in enhancing the removal of a wide array of OMPs through a combination of oxidizing and reducing conditions.
Some bench-scale studies have investigated the impact of MABR operational conditions on the fate and removal of OMPs. Çelik et al. (2018)24 and Taşkan et al. (2019)25 investigated the removal of oxytetracycline and tetracycline, respectively, and found that their flux into the biofilm increased with hydraulic retention time (HRT) (7.5 to 18 hours) and O2 pressure in the membrane (0.14 to 0.41 bar). In addition, increased biofilm thickness (0.33 to 1.02 mm) in a lab-scale biofilm-only MABR system improved the removal of multiple OMPs.26 The enhanced removal with thicker biofilms was attributed to the combined activity of both nitrifiers and heterotrophs.27 Overall, the literature indicates that MABR operational conditions and biofilm thickness could impact OMP removal.
While these smaller-scale studies provide a basic understanding of the role of operating conditions on OMP removal, they were often conducted with a synthetic wastewater that did not contain additional sources of carbon,28 and with elevated OMP concentrations.24,25 It is anticipated that these conditions may lead to responses that are not representative of real wastewater systems where OMP concentrations are dilute, and alternative, more readily biodegradable substrates are available to support biomass growth. It is considered critical to evaluate OMP removal in real wastewater with a complex substrate profile and representative concentrations to understand the true performance of MABRs. In addition, prior studies have focused on biofilm-only systems that did not have a bulk phase suspended sludge and had small membrane surface areas (10−3 to 10−1 m2). No study has so far evaluated the fate of OMP in full-scale hybrid MABRs that have an anoxic suspended growth phase. Further, OMP removal in MABRs has also not been investigated over the range of conditions (i.e. temperature and flow) that can change with season. Hence, the objective of this study was to evaluate the performance of a full-scale hybrid MABR system in removing an array of OMPs over a range of seasonal conditions that are typical of North America. This was achieved by monitoring the target contaminants in a full-scale WRRF before and after an MABR upgrade over a three year period. The data obtained from the multi-year sampling campaign were used to construct contaminant mass balances to elucidate the fate mechanisms and individual treatment processes responsible for OMP removal. The findings provided much-needed insights into the efficacy of MABR, a popular technology for treatment process intensification, against these harmful contaminants.
2. Methodology
2.1 Sampling plan
The wastewater plant in the study was originally a CAS facility (Fig. 1a) with an average rated flow of 9320 m3 d−1. The plant was later upgraded to include a hybrid MABR process (Fig. 1b) with four parallel anoxic tanks and a return active sludge (RAS) recycle. The MABR tanks housed 36 ZeeLung cassettes (Veolia Water Technologies, Oakville, Canada), arranged as rows of nine in four parallel tanks amounting to a total surface area of 69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 120 m2. The suspended sludge SRT in the plant was 5.4 ± 2.1 days and 5.1 ± 1.0 days before and after the upgrade, respectively. The daily average aeration tank temperature varied seasonally between 11 °C and 22 °C. Before upgrade, the plant was capable of seasonal nitrification with effluent ammonia spike as high as 20 mg N per L during cold weather conditions.29 MABR addition enhanced the performance to year long nitrification (<10 mg N per L) with twice the total nitrogen removal as the pre-upgrade performance (40% to 80%).29 A detailed description of the plant operations and its long-term BOD and N removal, and electricity consumption before and after upgrade can be found in Lakshminarasimman and Parker (2025).29
120 m2. The suspended sludge SRT in the plant was 5.4 ± 2.1 days and 5.1 ± 1.0 days before and after the upgrade, respectively. The daily average aeration tank temperature varied seasonally between 11 °C and 22 °C. Before upgrade, the plant was capable of seasonal nitrification with effluent ammonia spike as high as 20 mg N per L during cold weather conditions.29 MABR addition enhanced the performance to year long nitrification (<10 mg N per L) with twice the total nitrogen removal as the pre-upgrade performance (40% to 80%).29 A detailed description of the plant operations and its long-term BOD and N removal, and electricity consumption before and after upgrade can be found in Lakshminarasimman and Parker (2025).29
The full-scale monitoring in this study spanned across three years to capture the OMP removal with and without MABRs. Before the upgrade, composite samples (24 equally weighted hourly samples) of the plant influent and effluent (Fig. 1a) were collected using refrigerated autosamplers (Hach, London, ON, Canada). Samples were collected on three consecutive days during select months of 2020 (September to December), 2021 (January) and 2022 (February to August). Sampling days were designated as either warm or cold depending on whether the aeration tank temperature was greater or less than 15 °C. After the upgrade, an additional sampling point was included for the MABR effluent (Fig. 1b) to facilitate quantification of OMP removal in the MABR and aeration tank separately. Post-upgrade samples were collected on three consecutive days each month between Sept. 2022 and Sept. 2023. The samples were transported in coolers containing ice packs (<2 °C) to the university lab within a few hours of collection. As soon as the samples were received in the lab, they were preserved with 1 g L−1 sodium azide and 50 mg L−1 ascorbic acid and filtered using a Cytiva 1 μm pore size glass fiber filter (VWR, Mississauga, Canada). The filtered and preserved samples were extracted within 72 hours.
The sampling campaign included monitoring of the plant operations to support the subsequent assembly of contaminant mass balances. Data on wastewater and solids stream flows and aeration tank temperature were obtained from online sensors at the plant. The total suspended solids (TSS) concentration in the composite samples of the raw influent and MABR effluent and grab samples of the aeration tank mixed liquor suspended solids (MLSS) and the solids wasting stream were measured during every sampling event following standard methods 2540 D (Baird and Bridgewater, 2017). The key plant operational data collected for the full study period is shown in Table S1 in SI.
2.2 Reagents and chemicals
The target list of 20 OMPs and their physico-chemical properties are shown in Table 1. These chemicals were selected because: i) they are known to occur in a variety of watersheds;7,30,31 ii) some of them are known to elicit effects in aquatic life;32–34 and iii) they represent a wide range of usage and chemical functionalities. The samples were collected in pre-cleaned 1 L amber glass bottles (Systems Plus, Baden, ON, Canada) and were disposed after single use. All chemicals employed to prepare stock solutions and establish standard curves were purchased from Sigma-Aldrich, USA. Deuterated standards used for the quantitation of each of the target chemicals were purchased from TRC Chemicals, Canada, and CDN Isotopes, Canada. Both the regular and deuterated standards were dissolved in methanol to prepare a 1 g L−1 stock solution, which was stored in amber glass vials in a −20 °C freezer. All the solvents used in the extraction and instrumental analysis were of HPLC grade and were purchased from Fisher Scientific, Canada, while ultrapure water was from a lab-scale MilliQ water purification system (MilliporeSigma, Oakville, Canada).| Organic micropollutant | Category | Chemical formula | pKaa | Sorption distribution coefficient Kdb in L kg−1 average (range) | Analysis method | Method detection limit (MDL) in ng L−1 |
|---|---|---|---|---|---|---|
| a pKa values adopted from https://pubchem.ncbi.nlm.nih.gov/. b K d values are adopted from ref. 35 and the references therein. NA – not available. | ||||||
| Trimethoprim (TMP) | Antibiotic | C14H18N4O3 | 7.12 | 192 (25–330) | Pharmaceuticals and personal care products (PPCP) | 4.05 |
| Sulfamethoxazole (SULF) | C10H11N3O3S | 1.6, 5.7 | 120 (11–269) | 8.3 | ||
| Fluoxetine (FLX) | Antidepressant | C17H18F3NO | 10.1 | 2694 (880–6000) | 1.08 | |
| Norfluoxetine (NFLX) | C16H16F3NO | NA | 0.67 | |||
| Ibuprofen (IBU) | Nonsteroidal anti-inflammatory drugs | C13H18O2 | 4.9 | 90 (6–453) | 2.4 | |
| Naproxen (NPX) | C14H14O3 | 4.15 | 64 (10–217) | 1.22 | ||
| Diclofenac (DCF) | C14H11Cl2NO2 | 4.15 | 46 (2–151) | 9.02 | ||
| Acetaminophen (ACE) | Analgesic | C8H9NO2 | 9.38 | 84 | NA | |
| Gemfibrozil (GFZ) | Lipid regulators | C15H22O3 | 4.5 | 63 (19–129) | 0.43 | |
| Atorvastatin (ATOR) | C33H35FN2O5 | 4.3, 14.9 | 93 | 3.08 | ||
| o-Hydroxy atorvastatin (o-ATOR) | Human metabolites of ATOR | C33H35FN2O6 | NA | 4.96 | ||
| p-Hydroxy atorvastatin (p-ATOR) | C33H35FN2O6 | NA | 7.63 | |||
| Carbamazepine (CBZ) | Anticonvulsant | C15H12N2O | 13.9 | 38 (1–135) | 4.38 | |
| Triclosan (TCS) | Antimicrobial | C12H7Cl3O2 | 7.9 | 3890 | 1.75 | |
| Triclocarban (TCCB) | C13H9Cl3N2O | 12.7 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 703 703 |
0.44 | ||
| Caffeine (CAFF) | Stimulant | C8H10N4O2 | 14 | 190 (30–350) | 5.07 | |
| Estrone (E1) | Natural estrogen | C18H22O2 | 10.91 | 364 (100–645) | Endocrine disrupting chemicals (EDC) method | 4 |
| Estradiol (E2) | C18H24O2 | 10.46 | 270 (13–399) | 3.6 | ||
| Estriol (E3) | C18H24O3 | NA | 127 (51–360) | 3 | ||
| Bisphenol A (BPA) | Plasticizer | C15H16O2 | 9.6 | 547 (181–1060) | 8.81 | |
2.3 OMP analysis
The target OMPs were analyzed in filtered samples using different isotope dilution procedures for the PPCPs36 and endocrine disrupting chemicals (EDCs),37 respectively. The analytical procedure started with spiking 100 μg L−1 of deuterated standards in 100 mL of the sample. The analytes were then extracted and concentrated following two different solid phase extraction methods for PPCPs and EDCs as summarized in Table S2 in SI. Extracted samples were stored in a −20 °C freezer until instrumental analysis. Both PPCP and EDC analyses were performed with an Agilent 1260 HPLC coupled with an Agilent 6460 triple quadrupole mass spectrometer with electrospray ionization. The chromatographic separation of the PPCPs was accomplished using an Agilent Eclipse XDB-C18 column (4.6 mm × 150 mm × 5 μm) at 40 °C and a binary gradient of 5 mM ammonium acetate in MilliQ water and methanol. The EDC method used an Agilent ZORBAX Eclipse Plus C18 column (2.1 mm × 50 mm × 1.8 μm) at 35 °C with a binary mobile phase of 5 mM ammonium fluoride in MilliQ water and acetonitrile. The method detection limits for the analytes can be found in Table 1.2.4 Quality control and quality assurance
The quality control/quality assurance (QA/QC) procedure employed a procedural blank as a negative control to identify any extraneous contamination in each extraction batch. The procedural blanks consisted of a 100 mL MilliQ water sample spiked with 100 μL of 100 μg L−1 deuterated internal standard mix. The procedural blanks almost always reported concentrations less than the detection limit. The QA/QC procedure also included duplicate matrix spike samples, per batch, to adjust for target compound recovery. The matrix spike samples consisted of 100 mL MilliQ water with 100 μL of 100 μg L−1 regular and deuterated standards. A summary of the QA/QC results is provided in Table S3 in SI. The average matrix spike recovery for the target OMPs varied between 96% and 104% indicating satisfactory extraction efficiencies.2.5 Contaminant mass balance and removal efficiency calculations
The measured OMP concentrations were used to assemble OMP mass balances to evaluate the overall plantwide removal and elucidate the contributions of biotransformation in the MABR and aeration tanks and sorption to the suspended biomass to OMP removal. Each sample was analyzed in triplicate, and the resulting concentrations were averaged to obtain a representative concentration at the sample location on the sampling day. The removal and mass balance calculations were performed for each sampling day and then composited during data analysis. Since the analytical method measured only the soluble fraction of the OMPs in the samples, the OMPs associated with the solids fraction in the plant influent and MABR effluent samples were estimated based on sorption–distribution coefficients (Kd) obtained from the literature (Table 1). For two target OMPs, which were human metabolites of atorvastatin, Kd values were not available in the literature. Hence, the values of the parent compounds were adopted to approximate the metabolite values. In the subsequently discussed mass balances, the mass flow of OMPs associated with the solids in the final plant effluent was assumed to be negligible due to the relatively low tendency of the target OMPs to partition to solids and the low suspended solids concentrations in this stream (<25 mg TSS per L). The equations developed for plant-wide mass balances (Fig. S1) are described by eqn (1)–(5).| MInf = QInf × CInf × (1 + KdXInf) | (1) |
| MWAS = QWAS × CEff × (1 + Kd × XWAS) | (2) |
| MEff = QEff × CEff | (3) |
| Mbio = MInf − MEff − MWAS | (4) |
 | (5) |
M Inf = OMP mass flow in influent (g d−1)
M WAS = OMP mass flow in solids wasting stream (g d−1)
M Eff = OMP mass flow in plant effluent (g d−1)
M bio = OMP mass flow biotransformed (g d−1)
Q Inf = Daily average influent flow (m3 d−1)
C Inf = OMP concentration in plant influent (ng L−1)
X Inf = TSS concentration in plant influent (mg L−1)
Q WAS = Daily average WAS flow (m3 d−1)
C Eff = OMP concentration in WAS stream (ng L−1)
X WAS = TSS concentration in WAS stream (mg L−1)
In the pre-upgrade configuration, all biotransformation (Mbio) in the plant was attributed to the activity of the organisms in the aeration tank. However, post MABR upgrade, Mbio included biotransformations in both the MABR and aeration tanks. Additional mass balances were assembled for the post-upgrade configuration to divide the biotransformation into the individual tanks (Fig. S1b). As seen in Fig. S1b, the MABR effluent included both the plant influent and RAS flows. A new system boundary was drawn to calculate the mass flows in the blended flow as shown in eqn (6) to (9). Biotransformations in the MABR and aeration tank (AT) were estimated separately using eqn (10) and (11). The mass balance calculations assumed that sorption was instantaneous and that Kd values were the same for all streams.
| MABRin = QInfCInf (1 + KdXInf) + QRAS,1 × CEff × (1 + Kd × XRAS) | (6) |
| MABRout = (QInf + QRAS,1) × CMABR,Eff × (1 + Kd × XMLSS,1) | (7) |
| ATin = MABRout + (QRAS,2 × CEff × (1 + KdXRAS)) | (8) |
| ATout = (QInf + QRAS,1 + QRAS,2) × CEff × (1 + KdXMLSS,2) | (9) |
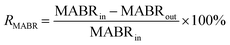 | (10) |
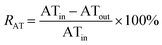 | (11) |
MABRin = OMP mass flow in MABR influent (g d−1)
MABRout = OMP mass flow in MABR influent (g d−1)
Q RAS,1 = Flow of RAS stream 1 (m3 d−1)
Q RAS,2 = Flow of RAS stream 2 (m3 d−1)
C MABR,Eff = OMP concentration in MABR effluent (ng L−1)
X MLSS,1 = MLSS concentration in MABR tank (mg L−1)
X MLSS,2 = MLSS concentration in aeration tank (mg L−1)
X RAS = TSS concentration in the RAS stream (mg L−1)
R MABR = Removal through biotransformation in MABR (%)
R AT = Removal through biotransformation in aeration tank (%)
2.6 Statistical analysis
The measured OMP concentrations and calculated removal efficiencies for different sampling days were categorized into four groups on the basis of plant configuration (before and after upgrade) and seasonality (cold and warm) to analyze the plant performance as a function of the two variables. An initial Shapiro–Wilk test (α = 0.05) indicated that the concentration and percentage removal data were not normally distributed. Hence, the non-parametric Kruskal–Wallis Test was used to ascertain whether configuration and seasonality impacted the concentration and removal values. A Dunn's post hoc test was used for multiple comparisons of the concentration and removal datasets between different configurations and seasonality scenarios. The P value was adjusted using a Bonferroni correction (α/number of comparisons (2 × 2)) from the standard 0.05 value to P* = 0.0084 (adjusted P-value) to minimize the risk of type I error when making multiple comparisons.38 In cases where there was a statistically significant difference in the concentration or the removal values, the magnitude of the difference between the averages was determined using relative percentage difference (eqn (12)).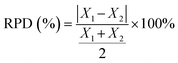 | (12) |
3. Results and discussion
The overall objective of the study was to evaluate the full-scale OMP removal performance as a function of MABR upgrade and seasonality. Hence, the larger dataset collected over three years was clustered into four categories, namely, pre-upgrade warm conditions (21 sampling days), pre-upgrade cold conditions (15 sampling days), post-upgrade warm conditions (24 sampling days), and post-upgrade cold conditions (15 sampling days) to facilitate multiple comparisons between the various scenarios. The impact of MABR addition was examined by comparing the pre-upgrade warm and post-upgrade warm scenarios. The warm condition was chosen because under this condition, the MABR operations were maintained steady, and the effluent NH4 and NO2 + NO3 concentrations were below 1 and 10 mg N per L, respectively.29 The impact of seasonality was assessed for both the pre- and post-upgrade scenarios by comparing the warm weather (T > 15 °C) and cold weather (T < 15 °C) data under the two plant configurations. The comparisons were made using the effluent OMP concentrations first, and this was followed by an analysis of removal efficiencies.3.1 Impact of upgrade and seasonality on OMP concentrations
The OMP concentrations in the plant effluent are toxicologically relevant indicators for the overall quality of the treated wastewater discharging into the receiving water bodies. Hence, the OMP concentrations in the plant effluent before and after the upgrade (Fig. 2) were compared to assess the impact of MABR addition on the effluent quality. The statistical significance in difference between the concentration as determined by the post hoc Dunn's test is shown in Table S4 in SI. After the MABR upgrade, the plant effluent concentrations decreased significantly (P* < 0.008) for 12 of the 20 compounds. The magnitude of change in concentration varied depending on the OMPs. Ibuprofen (P* = 0.004), naproxen (P* < 0.001), caffeine (P* < 0.001), triclosan (P* < 0.001), and BPA had RPD more than 110% indicating a large difference in the plant effluent concentrations after upgrade. Whereas atorvastatin, ortho- and para-hydroxy atorvastatin, sulfamethoxazole, trimethoprim, carbamazepine, and gemfibrozil had moderate differences with RPD values ranging between 15% and 68%. The average concentrations of diclofenac, fluoxetine, and norfluoxetine were the same before and after the upgrade. Overall, the average concentrations of a majority of the targeted OMPs were reduced with the MABR upgrade.The average effluent OMP concentrations were compared between warm and cold weather conditions under the two configurations to study the impact of seasonality on effluent quality. The average wastewater temperatures were 13.1 ± 1.2 °C and 19.3 ± 1.5 °C during the cold and warm weather sampling months, revealing an average temperature differential of 6 °C between the two scenarios. Before the upgrade, only norfluoxetine concentrations were significantly different (P* = 0.002) between the warm and cold weather conditions. While there were some interesting differences such as higher warm concentration for triclosan before upgrade, these trends were not statistically significant. However, after the upgrade, about eight target OMPs had a significant difference between the warm and cold weather concentrations (Table S4). E1 reported the highest RPD of 110% with a significant decrease (P* > 0.0001) in average plant effluent concentration from 25 ng L−1 to 7 ng L−1 (Fig. 2a). Similarly, triclosan, ibuprofen, naproxen, atorvastatin, o-hydroxy atorvastatin, p-hydroxy atorvastatin, trimethoprim, norfluoxetine, and gemfibrozil reported significantly lower concentrations during warm weather conditions with RPD ranging between 80–45%. Overall, after MABR addition, there was a significant impact of wastewater temperature on the concentrations of many of the monitored OMPs in the plant effluent.
Viewed collectively, the two levels of comparisons indicate that the addition of MABR resulted in a general improvement in the overall effluent quality of the plant with respect to the measured target compounds, and the extent of the impact was influenced by the wastewater temperature. However, the observed impacts could also have been due to changes in the influent OMP concentrations over time that might result from seasonal usage of some of the pharmaceuticals.39 The influent concentrations are shown in Fig. S2 and their variability within each operating condition is expressed as the co-efficient of variation in Table S5 in SI. The influent concentration varied from 3% (E3 post upgrade warm conditions) to 158% (BPA pre upgrade cold conditions) over the full monitoring period (Table S3). Hence, plantwide OMP removal efficiencies calculated based on contaminant mass flows were assessed to study plant performance as a function of the upgrade and seasonality.
3.2 Plantwide OMP removal before MABR upgrade
The plantwide removal efficiencies of the OMPs were estimated for the CAS-only configuration to establish a baseline against which the upgraded plant performance could be compared. An initial review of the removal efficiencies revealed that the target OMPs exhibited a wide range of removals. Hence, to facilitate a structured discussion, the target compounds were clustered into three groups based on the magnitudes of removal efficiency as high (>75%), moderate (75 to 25%), and low removal (<25%). Using this structure, Fig. 3(a–c) shows the plantwide removal efficiencies and the contributions of the calculated fate pathways to removal during warm and cold weather conditions.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 703 L kg−1, respectively), reported 10–15% and 20–25% of the influent contaminant load removed through sorption, with the remainder of the influent mass loading (75%) removed by biotransformation. Thus, biotransformation was likely the predominant removal mechanism for highly removed compounds that represented OMPs with a broad range of sorption affinities.
703 L kg−1, respectively), reported 10–15% and 20–25% of the influent contaminant load removed through sorption, with the remainder of the influent mass loading (75%) removed by biotransformation. Thus, biotransformation was likely the predominant removal mechanism for highly removed compounds that represented OMPs with a broad range of sorption affinities.
BPA had an average removal of −45% during cold weather conditions (Fig. 3b) indicating a higher mass flow exiting the process when compared to the influent load. The overall negative removal was accompanied by a negative biotransformation fraction (−25%) and considerable sorption due to a moderately high Kd value of 547 L kg−1. The apparent increase in mass of BPA has been observed in other studies as well with removals as low as −147% due to transformation from glucuronide conjugates40–42 and desorption or release from the sludge or influent solids.43 Temperature sensitive removal of BPA has also been previously reported with high winter removal requiring longer SRTs (>17 days) and higher mixed liquor concentrations (>5300 mg L−1)44,45 both of which were not observed at the study site. Thus, the net increase in BPA across the process can be attributed to a combination of reduced winter biodegradation capacity and the re-mobilization or reconversion of previously sorbed or conjugated forms.
The contribution of adsorption to the overall removal of highly sorptive compounds (Kd < 1000) was investigated to understand its significance relative to biotransformation. For fluoxetine, a compound with a moderately high Kd value (2694 L kg−1), about half of the influent mass load was removed by sorption. The fraction of fluoxetine removed through sorption was much higher than that of triclosan and triclocarban (Fig. 3a), which had higher Kd values. This was due to the differences in biodegradability of the compounds. For triclosan and triclocarban, the aqueous phase concentrations were maintained low by biodegradation, and this reduced the corresponding sorbed concentrations. On the contrary, fluoxetine was less amenable to biotransformation, and higher aqueous concentrations led to greater concentration in solids. Thus, the extent of removal by sorption was influenced by sorption affinity and the degree of biotransformation of the compounds.
The study site, in its CAS configuration, was only capable of seasonal nitrification with very low ammonia removal during cold weather conditions.29 Hence, the plantwide removal efficiencies calculated before the upgrade were compared between cold and warm weather conditions to assess whether OMP removal was consistent with changes in the extent of nitrification. Analysis of variance between the two datasets revealed that there was no statistical significance (P* > 0.008) between the average cold and warm weather removal efficiencies of all 20 OMPs. The similar overall removal efficiencies suggest that the autotrophic microorganisms involved in nitrification may not be solely responsible for OMP transformation. Heterotrophic microbes are capable of co-metabolic transformation of OMPs through reactions such as hydroxylation, hydrolysis, and demethylation, catalyzed by commonly available non-specific enzymes.46 Transformation of OMPs such as sulfamethoxazole, trimethoprim, ibuprofen, naproxen, diclofenac, and triclosan has been observed in aerobic heterotrophic reactors.21,47–49 Thus, the similar removals of OMPs during both the seasonal conditions underpin the significant role played by heterotrophs in the CAS process.
Viewed collectively, more than half of the targeted OMPs were removed moderately or poorly by the CAS system. Assessment of the individual fate processes showed that biotransformation was the major removal mechanism for all of the compounds and removal through sorption was minimal for most compounds. Moreover, there was no significant difference in OMP removal between cold and warm weather conditions before the upgrade.
3.3 Plantwide OMP removal after MABR upgrade
The plantwide removal efficiencies from before and after the upgrade were compared to assess the impact of MABR addition during warm conditions. Warm conditions were selected because the post-upgrade plant operational conditions were maintained steady and the MABR showed optimal performance during this time period.29 The post-upgrade removal efficiencies are shown in Fig. 3(d–f) for the previously defined groups based on the magnitude of removal efficiencies. Some of the OMPs changed groups as their removal efficiency changed after the upgrade. The observed trends within each grouping are discussed subsequently.The relative roles of biotransformation and sorption of highly removed OMPs were compared before and after the upgrade to assess if there were any changes in the contribution of the two fate mechanisms. Biotransformation dominated the overall removal for all the highly removed compounds similar to the pre upgrade trends (Fig. 3d). However, for triclosan and triclocarban, there were subtle changes in the relative contribution of sorption vs. biotransformation after the upgrade. As discussed previously, 10–25% of triclosan and triclocarban were removed through sorption before the upgrade (Fig. 3a). However, after the upgrade, less than 10% of the triclosan and triclocarban were removed through sorption. Correspondingly, the contribution of biotransformation increased after the upgrade for these compounds. Thus, incorporation of the MABR decreased the overall contaminant load in both the wastewater effluent and solids wasting stream for these highly sorptive and biodegradable compounds.
Examining the impact of seasonality on removal after upgrade, the removal efficiencies of ibuprofen, naproxen, triclosan, BPA, and E1 under warm weather conditions were significantly higher (P < 0.008) than cold weather months (Fig. 3d). However, the RPD between the averages was less than 15% for all three compounds, suggesting a very small variation due to seasonality. For other compounds such as gemfibrozil, atorvastatin, and p-hydroxy atorvastatin there was a significantly (P* > 0.008) higher removal during warm weather conditions with a considerable improvement of RPD values (65%, 75%, and 23%, respectively). These compounds had not exhibited significant seasonal difference pre-upgrade when they were poorly removed. The higher sensitivity of select OMPs to seasonality post-upgrade suggests that changes in MABR operational conditions (organic loading, process airflow) that resulted in increased biofilm thickness during the cold weather could have contributed to the variability in removal.29
The moderately removed OMPs after the upgrade had a range of Kd values. Hence, they were assessed further to elucidate the roles of sorption and biotransformation in their overall removal. Biotransformation contributed to almost all of the removal for trimethoprim and diclofenac (Fig. 3e). The highly sorptive behavior of fluoxetine was consistent with the trend seen in the pre-upgrade scenario. However, the average contribution of biotransformation to the removal of fluoxetine also increased from almost nil to nearly 20%. Thus, the improvement in biotransformation post MABR upgrade reduced the removal through adsorption for OMPs with high sorption affinity.
Overall, post-upgrade OMP removal improved substantially, with several compounds shifting to higher removal categories. Some compounds showing enhanced removal after MABR addition were also affected by seasonality, with better performance in warm conditions. Biotransformation was the dominant fate process for all compounds exhibiting improved removal. While these findings provide insights to plant-wide changes following the MABR upgrade, it is also important to assess biotransformation within the MABR and aeration tanks individually to understand their respective contributions.
3.4 Biotransformation in MABR and aeration tank
The post-upgrade results were further analyzed to separate biodegradation occurring in the MABR and the aeration tank (eqn (6)–(12)). An initial analysis of the mass balance results revealed that the OMPs exhibited a variety of removal patterns in the MABR. Hence, select compounds were chosen (Fig. 4) to represent the different removal groups and capture the removal behaviors observed in the MABR. The results for the remaining compounds are shown in the SI (Fig. S3).It was hypothesized that readily degraded compounds (acetaminophen and naproxen) would biodegrade in the MABR because of its upstream location that receives the wastewater before the aeration tank. Biotransformation in the MABR removed almost all the acetaminophen under warm temperatures (Fig. 4). Under cold temperatures, the contribution of the MABR to the overall removal remained high (average = 93%) while the aeration tank contribution increased relative to its contribution under warm conditions. These trends are to be expected as acetaminophen is well known to biotransform under aerobic and anoxic environments that supported nitrification and denitrification54–56 and the MABR at the study site reported active simultaneous nitrification and denitrification activities.29 On the contrary, the aeration tank contributed a greater fraction of the removal of naproxen when compared to the MABR. Although, when comparing the two seasonal conditions, the MABR had a relatively higher contribution of 30% under warm conditions compared to the 7% in the cold weather, similar to acetaminophen (Fig. 4). These findings are consistent with literature where naproxen was favored to biotransform aerobically under fully nitrifying and nitrite oxidizing conditions.47 While naproxen has been observed to be reduced under methanogenic and sulfur reducing conditions,57,58 it is limited under nitrate reducing conditions11 which explains the limited transformation in the largely anoxic MABR tank. Overall, biotransformation in the MABR contributed either wholly or partially towards the removal of readily biodegradable compounds but its contribution was highly compound-specific and varied seasonally.
The fate of sulfamethoxazole was examined to assess the contribution of the MABR to removal of a moderately degradable compound. The mass balance analysis indicated that sulfamethoxazole had opposing responses in the MABR and aeration tank. In the MABR negative removals were apparent while the aeration tank resulted in its removal (Fig. 4). The apparent formation of sulfamethoxazole may have resulted from the deconjugation of unmeasured sulfamethoxazole human metabolites that were present in the raw wastewater. Conjugated products of sulfamethoxazole such as N-acetyl sulfamethoxazole and sulfamethoxazole glucuronide have been reported to occur at comparable or even higher concentrations than the parent compound in raw wastewaters.59,60 In the biological treatment process, the conjugated groups are cleaved releasing the parent compound leading to an apparent negative removal.61,62 The upstream location of the MABR with its anoxic bulk phase environment appeared to result in a condition where formation from deconjugation exceeded biodegradation of sulfamethoxazole that has been reported to degrade in aerobic, anoxic, and anaerobic environments.63,64 Thus, the MABR process resulted in a net formation of sulfamethoxazole although the subsequent aeration tank was able to remove a large fraction of the sulfamethoxazole leaving the MABR.
Examining the seasonality of sulfamethoxazole transformations in the individual tanks, the formation in the MABR was relatively higher during cold weather (−51%) as compared to the warm weather (−37%) conditions (Fig. 4a). Lower apparent formation during warm conditions was consistent with an increase in sulfamethoxazole degradation in the process. Enhanced removal of sulfamethoxazole could also be seen when comparing the percentage removal in aeration tank. Converse to MABR, the average percentage removal of sulfamethoxazole was slightly higher in the warm weather conditions (92%) compared to cold weather (86%). Overall, even though sulfamethoxazole had an apparent overall negative removal in the MABR, seasonal variations in the extent of apparent formation suggests that both formation and degradation reactions were active in the process.
Carbamazepine was examined as a compound that was representative of the poorly degradable category and had apparent negative removals in both tanks (Fig. 4). Carbamazepine is known to transform in the human body to form a range of excreted metabolites including hydroxylated products, N-glucuronide conjugates, and oxidation products such as carbamazepine epoxides that have been detected in wastewaters.65–67 The human metabolites such as the glucuronide conjugates could be deconjugated through enzymatic reactions and transform into the parent compound during wastewater treatment resulting in apparent negative removals of carbamazepine.68,69 Thus, the apparent negative removals for carbamazepine seen in the MABR were likely due to extensive release of the target compound from the unmeasured conjugated human metabolites.
The cold conditions had a greater apparent formation of carbamazepine with an overall average of 124% when compared to 98% in warm weather conditions (Fig. 4). The difference in magnitude of apparent formation indicates that there was a seasonal dependence in the degradation of parent compound removal as seen previously for sulfamethoxazole. The lower magnitude of apparent formation could be because of a relatively higher degree of carbamazepine removal occurring in parallel during warm weather conditions. Although carbamazepine is widely known to be resistant to biotransformation, some studies have shown 20–60% removal in MBRs operating at very long SRTs (88 days) and alternating oxic–anoxic redox conditions.64,70 Overall, the seasonal comparison revealed that carbamazepine could undergo some level of biodegradation in both the MABR and aeration tank. However, the decrease in mass was overshadowed by extensive release from precursors.
Viewed collectively, the mass balance analyses on the individual treatment tanks showed that the MABR could significantly impact on OMPs but its contribution to overall removals was highly compound specific. Of particular interest was that, as the first process in the biological treatment sequence, its contribution to either positive or apparent negative removals was frequently greater than that of the downstream aeration tanks. Further, in cases where removal in the MABR performance was reduced (low temperatures or apparent formation from precursors) the aeration tank was often able to polish the MABR effluent.
3.5 Comparison of removal in MABR–CAS configuration with other secondary treatment processes
The trends in improved OMP removal after the MABR upgrade were compared with reports on other advanced treatment configurations to explore if the responses observed in this study are typical of other configurations with similar design features. BNR configurations with alternating redox zones have shown better OMP removal with reduced effluent toxicity.1,14,71,72 Anoxic and anaerobic redox zones in the BNR configurations select for nitrate-reducing and sulfate-reducing bacteria that can perform complementary reductive reactions such as demethylation and dehalogenation reactions, which are crucial for the breakdown of pharmaceuticals and hormones that persist in conventional aerobic treatment processes.73–75 Prior studies have shown anoxic and anaerobic biotransformation of compounds such as BPA,76 diclofenac,77 and sulfamethoxazole78 that in the current study had improved removal when the MABR was operating. Microbial community analysis at the study site during this monitoring period showed the presence of communities with sulfate reduction in the MABR biofilms and nitrate reduction functions in the MABR biofilms and the suspended sludge.79,80 Thus, the anoxic tank and the varying redox zones in the MABR biofilms appeared to have broadened the range of metabolic pathways for OMP biotransformation, resulting in their enhanced removal.The presence of biofilms can also enhance OMP biotransformation because of increases in microbial diversity from the local redox gradients and retention of slow-growing microorganisms. In this study, the removals of diclofenac and gemfibrozil increased by 2.5 times (31 to 78%) and 4.5 times (13% to 54%), respectively, after the MABR upgrade (Fig. 3c and e). Similar increases in the removal of diclofenac and gemfibrozil have been observed in MBBR biofilms when compared to suspended sludge biomass.18,21 Similarly, Abtahi et al. (2018)81 found that the biofilm biomass contributed to a higher degree of biotransformation of diclofenac, naproxen, and 17β-estradiol when to compared to suspended sludge. The improved biotransformation potential of the biofilms in hybrid systems has been attributed to its ability to select for a proportionally larger and a more diverse core microbial community when compared to suspended sludge.82 The microbial community analysis for this site showed that the MABR biofilms housed the highest number of unique core amplicon sequence variants (ASVs) (128 ASVs) compared to only 23 core ASVs unique to the suspended sludge before upgrade.80 Moreover, the MABR addition also shaped the downstream aeration tank microbial community to be more diverse as indicated by a significant increase in the combined evenness, Shannon index, and the core ASVs unique to suspended sludge (109 ASVs).80 These changes to the microbial community assembly of the plant could be the main driver for the observed improvement in OMP removal.
The biofilm thickness could also play a significant role in the OMP biotransformation. Torresi et al. (2016)83 characterized the biotransformation rate and microbial community of MBBR carriers with controlled biofilm thicknesses of 50, 200, and 500 μm and observed that thicker biofilms exhibited the highest biotransformation rate for a majority of the OMPs studied and the highest microbial diversity. They concluded that while thin biofilms could support some OMP removal, thicker biofilms may be preferred for a wider range of OMPs.83 Similarly, increasing thickness in MABR biofilms has been shown to increase cell density, microbial diversity, and rates of OMP biotransformation due to both nitrifying and heterotrophic activities.26,27 These similarities suggest that the benefits of OMP removal observed in typical biofilm-based configurations such as the MBBRs are applicable to newer processes such as the MABR as well. However, there could be nuanced differences in the biotransformation potential between MBBR and MABR biofilms due to unique microbial niches arising from the co- and counter-diffusion of substrates, respectively. Future controlled experiments are needed to confirm this hypothesis.
Several compounds that had an improvement in removal with the MABR upgrade have also been shown to have higher removal by MBRs when compared to CAS process. Superior removal of EDCs such as BPA, estrone, estradiol, and pharmaceuticals such as gemfibrozil, diclofenac has been reported in the literature.17,51,84,85 MBRs are characterized by long SRT and high MLSS concentrations that can select for slow-growing microorganisms and offer a higher biotransformation rate.86 Viewed collectively, the enhanced removal of some OMPs post MABR addition is consistent with other configurations characterized by alternating redox conditions, biofilm design, and longer biomass residence time. These process features shaped the microbial community of the post upgrade plant to include diverse functionalities that could drive OMP transformation.80 While it is challenging to attribute specific process changes to improvement in the individual OMP removal, the overall results suggest the combination of the process changes resulting from MABR addition can enhance OMP removal.
While the mass balance provided some insights into the contaminant biotransformation in the individual tanks, the approach also has some limitations. Since this study only measured the aqueous phase OMP concentrations, the total concentrations were estimated using Kd values adopted from literature. The uncertainty with using the same Kd value for both influent and mixed liquor solids, and the values themselves could be potential sources of error that could influence the contaminant mass loads in the mixed liquor streams in and out of the bioreactors. Future full-scale campaigns should measure the total OMP concentration (dissolved, sorbed, and conjugated) in the mixed liquor samples for removal calculations in the individual tanks. Moreover, the removal efficiencies discussed here only reflect the fate of the parent compound. Future studies should investigate the fate of transformation products and potential reduction in the biological effects with the improved effluent quality.
4. Conclusion
Hybrid MABR processes include alternating redox conditions and biofilms that extend biomass retention times that can enhance the removal of OMPs from wastewater. This study monitored the removal of a wide range of commonly occurring OMPs in a full-scale WRRF in CAS and hybrid MABR/CAS configurations over three years that spanned warm and cold weather conditions. The results of the study showed that:• Of the 20 target compounds, 13 OMPs had a decreased concentration after the MABR upgrade demonstrating its presence can improve effluent quality.
• A comparison of removal efficiency before and after the upgrade under warm weather conditions, showed that readily treatable compounds such as acetaminophen, naproxen, ibuprofen, and E3, and recalcitrant compounds such as carbamazepine were not impacted by MABR addition.
• The MABR upgrade resulted in a marked improvement in the removal of about half of the target OMPs such as bisphenol A E1, E2, gemfibrozil, sulfamethoxazole, trimethoprim, diclofenac, atorvastatin, and o-hydroxy and p-hydroxy atorvastatin.
• Seasonality influenced removals after the MABR upgrade with some compounds such as gemfibrozil, and atorvastatin and its para hydroxy metabolite showing a significantly higher removal during warm weather conditions.
• The role of the MABR process in overall removal was compound specific and included complete biotransformation, partial biotransformation and apparent formation of target compounds from unmeasured precursors.
Overall, this study demonstrated that upgrading CAS facilities with a hybrid MABR can enhance the removal of several organic micropollutants. Viewed holistically with this study and its well-known conventional performance, MABR is a suitable retrofit technology for WRRFs discharging into ecologically sensitive receiving watersheds. While this study advances understanding of OMP removal in MABR systems, there are still unknowns with respect to characterizing transformation products and assessing residual biological activity. Future research should more comprehensively investigate transformation pathways and products, as well as evaluate treatment performance in terms of minimizing biological effects.
Author contributions
N. L. led the conceptualization and methodology design, coordinated sampling campaigns, performed mass balance analysis, curated and visualized data, and prepared the original draft of the manuscript, including subsequent revisions. S. J. contributed to investigation by supporting OMP analysis and data processing and participated in manuscript review and editing. L. B. oversaw sampling campaigns, supervised OMP analysis and data processing, and contributed to manuscript editing. M. S. provided overall supervision, supported formal data analysis, and assisted in reviewing and editing the manuscript. W. P. secured project funding, provided overall supervision, supported data analysis, and contributed to manuscript revision.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Tables S1 presents the key plant operational data. Table S2 shows the QC/QC performance of the analytical method. The raw concentrations for all the samples in this study are available in the Federated Research Access Depository at: https://doi.org/10.20383/103.01578.Supplementary information is available. See DOI: https://doi.org/10.1039/d5ew01208b.
Acknowledgements
This research was supported by the Natural Sciences and Engineering Research Council (NSERC) Alliance Grant (558427-2020) in partnership with the Regional Municipality of Waterloo, Ontario Clean Water Agency, Veolia Water Technologies, Stantec, and inCtrl Solutions. Funding also came from NSERC Discovery (#RGPIN2017-03816), Global Water Futures (GWF, #418473) and GWF-Observatories (#42687). The authors would like to acknowledge the contributions of Sandra Salic and Rashne Vakharia in the OMP analysis and data processing. The authors would also like to thank the operational team at the study site for their assistance and cooperation with this research.References
- T. Alvarino, J. Lema, F. Omil and S. Suárez, Rev. Environ. Sci. Biotechnol., 2018, 17, 447–469, DOI:10.1007/s11157-018-9472-3.
- A. B. A. Boxall, M. A. Rudd, B. W. Brooks, D. J. Caldwell, K. Choi, S. Hickmann, E. Innes, K. Ostapyk, J. P. Staveley, T. Verslycke, G. T. Ankley, K. F. Beazley, S. E. Belanger, J. P. Berninger, P. Carriquiriborde, A. Coors, P. C. DeLeo, S. D. Dyer and J. F. Ericson, et al. , Environ. Health Perspect., 2012, 120, 1221–1229, DOI:10.1289/ehp.1104477.
- R. P. Schwarzenbach, B. I. Escher, K. Fenner, T. B. Hofstetter, C. A. Johnson, U. von Gunten and B. Wehrli, Science, 2006, 313, 1072–1077, DOI:10.1126/science.1127291.
- Y. Shao, Z. Chen, H. Hollert, S. Zhou, B. Deutschmann and T.-B. Seiler, Sci. Total Environ., 2019, 666, 1273–1282, DOI:10.1016/j.scitotenv.2019.02.047.
- C. Stamm, K. Räsänen, F. J. Burdon, F. Altermatt, J. Jokela, A. Joss, M. Ackermann and R. I. L. Eggen, in Advances in Ecological Research, ed. A. J. Dumbrell, R. L. Kordas and G. Woodward, Academic Press, 2016, vol. 55, pp. 183–223, DOI:10.1016/bs.aecr.2016.07.002.
- B. Petrie, R. Barden and B. Kasprzyk-Hordern, Water Res., 2015, 72, 3–27, DOI:10.1016/j.watres.2014.08.053.
- T. A. Ternes, Water Res., 1998, 32, 3245–3260, DOI:10.1016/S0043-1354(98)00099-2.
- R. L. Oulton, T. Kohn and D. M. Cwiertny, J. Environ. Monit., 2010, 12, 1956–1978, 10.1039/C0EM00068J.
- K. A. Hicks, M. L. M. Fuzzen, E. K. McCann, M. J. Arlos, L. M. Bragg, S. Kleywegt, G. R. Tetreault, M. E. McMaster and M. R. Servos, Environ. Sci. Technol., 2017, 51, 1811–1819, DOI:10.1021/acs.est.6b05370.
- K. E. Nikel, G. R. Tetreault, P. Marjan, K. A. Hicks, M. L. M. Fuzzen, N. Srikanthan, E. K. McCann, H. Dhiyebi, L. M. Bragg, P. Law, D. Celmer-Repin, S. Kleywegt, J. Cunningham, T. Clark, M. E. McMaster and M. R. Servos, Aquat. Toxicol., 2023, 255, 106375, DOI:10.1016/j.aquatox.2022.106375.
- M. Inyang, R. Flowers, D. McAvoy and E. Dickenson, Bioresour. Technol., 2016, 216, 778–784, DOI:10.1016/j.biortech.2016.05.124.
- N. Lakshminarasimman, O. Quiñones, B. J. Vanderford, P. Campo-Moreno, E. V. Dickenson and D. C. McAvoy, Sci. Total Environ., 2018, 640–641, 62–72, DOI:10.1016/j.scitotenv.2018.05.145.
- S. Suarez, J. M. Lema and F. Omil, Water Res., 2010, 44, 3214–3224, DOI:10.1016/j.watres.2010.02.040.
- W. J. Parker, V. Pileggi, P. Seto, X. Chen, M. Ogunlaja, G. Van Der Kraak and J. Parrott, Sci. Total Environ., 2014, 490, 360–369, DOI:10.1016/j.scitotenv.2014.05.023.
- O. O. Ogunlaja and W. J. Parker, Sci. Total Environ., 2015, 514, 202–210, DOI:10.1016/j.scitotenv.2015.01.100.
- S. K. Maeng, B. G. Choi, K. T. Lee and K. G. Song, Water Res., 2013, 47, 3151–3162, DOI:10.1016/j.watres.2013.03.014.
- J. Radjenović, M. Petrović and D. Barceló, Water Res., 2009, 43, 831–841, DOI:10.1016/j.watres.2008.11.043.
- P. Falas, P. Longrée, J. la Cour Jansen, H. Siegrist, J. Hollender and A. Joss, Water Res., 2013, 47, 4498–4506, DOI:10.1016/j.watres.2013.05.010.
- A. A. Mazioti, A. S. Stasinakis, Y. Pantazi and H. R. Andersen, Bioresour. Technol., 2015, 192, 627–635, DOI:10.1016/j.biortech.2015.06.035.
- K. J. Murray, W. J. Parker, L. M. Bragg and M. R. Servos, Water Sci. Technol., 2017, 75, 2680–2691, DOI:10.2166/wst.2017.100.
- P. Falas, H. R. Andersen, A. Ledin and J. la Cour Jansen, Environ. Technol., 2012, 33, 865–872, DOI:10.1080/09593330.2011.601764.
- T. Li, J. Liu, R. Bai and F. S. Wong, Environ. Sci. Technol., 2008, 42, 2099–2104, DOI:10.1021/es702150f.
- X. Mei, Y. Wang, Y. Yang, L. Xu, Y. Wang, Z. Guo, W. Shen, Z. Zhang, M. Ma, Y. Ding, Y. Xiao, X. Yang, C. Yin, W. Guo, K. Xu and C. Wang, Sep. Purif. Technol., 2020, 248, 117078, DOI:10.1016/j.seppur.2020.117078.
- A. Çelik, E. Casey and H. Hasar, J. Hazard. Mater., 2018, 356, 26–33, DOI:10.1016/j.jhazmat.2018.05.040.
- B. Taşkan, E. Casey and H. Hasar, Sci. Total Environ., 2019, 682, 553–560, DOI:10.1016/j.scitotenv.2019.05.111.
- C. Sanchez-Huerta, L. Fortunato, T. Leiknes and P.-Y. Hong, J. Hazard. Mater., 2022, 432, 128698, DOI:10.1016/j.jhazmat.2022.128698.
- C. Sanchez-Huerta, J. S. Medina, C. Wang, L. Fortunato and P.-Y. Hong, Water Res., 2023, 236, 119935, DOI:10.1016/j.watres.2023.119935.
- T. Elad, M. P. Hally, C. Domingo-Félez, O. Knoop, J. E. Drewes, B. Valverde-Pérez and B. F. Smets, Sci. Total Environ., 2023, 891, 164329, DOI:10.1016/j.scitotenv.2023.164329.
- N. Lakshminarasimman and W. J. Parker, J. Water Process Eng., 2025, 70, 106945, DOI:10.1016/j.jwpe.2025.106945.
- M. J. Arlos, L. M. Bragg, W. J. Parker and M. R. Servos, Water Res., 2015, 72, 40–50, DOI:10.1016/j.watres.2014.11.008.
- P. Verlicchi, M. Al Aukidy and E. Zambello, Sci. Total Environ., 2012, 429, 123–155, DOI:10.1016/j.scitotenv.2012.04.028.
- L. Cizmas, V. K. Sharma, C. M. Gray and T. J. McDonald, Environ. Chem. Lett., 2015, 13, 381–394, DOI:10.1007/s10311-015-0524-4.
- A. J. Ebele, M. Abou-Elwafa Abdallah and S. Harrad, Emerging Contam., 2017, 3, 1–16, DOI:10.1016/j.emcon.2016.12.004.
- M. Patel, R. Kumar, K. Kishor, T. Mlsna, C. U. Pittman and D. Mohan, Chem. Rev., 2019, 119, 3510–3673, DOI:10.1021/acs.chemrev.8b00299.
- M. Pomiès, J.-M. Choubert, C. Wisniewski and M. Coquery, Sci. Total Environ., 2013, 443, 733–748, DOI:10.1016/j.scitotenv.2012.11.037.
- H. Mehdi, Master's thesis, University of Waterloo, 2017, Available at: http://hdl.handle.net/10012/12207 Search PubMed.
- N. Srikanthan, Master's thesis, University of Waterloo, 2019, Available at: http://hdl.handle.net/10012/15167 Search PubMed.
- R. A. Armstrong, Ophthalmic Physiol. Opt., 2014, 34, 502–508, DOI:10.1111/opo.12131.
- N. M. Vieno, T. Tuhkanen and L. Kronberg, Environ. Sci. Technol., 2005, 39, 8220–8226, DOI:10.1021/es051124k.
- Y.-J. He, W. Chen, X.-Y. Zheng, X.-N. Wang and X. Huang, Sci. Total Environ., 2013, 447, 248–254, DOI:10.1016/j.scitotenv.2013.01.009.
- M. Z. Lacroix, S. Puel, S. H. Collet, T. Corbel, N. Picard-Hagen, P. L. Toutain, C. Viguié and V. Gayrard, Talanta, 2011, 85, 2053–2059, DOI:10.1016/j.talanta.2011.07.040.
- J. Xue and K. Kannan, Sci. Total Environ., 2019, 648, 442–449, DOI:10.1016/j.scitotenv.2018.08.047.
- R. H. Lindberg, P. Wennberg, M. I. Johansson, M. Tysklind and B. A. V. Andersson, Environ. Sci. Technol., 2005, 39, 3421–3429, DOI:10.1021/es048143z.
- P. Guerra, M. Kim, S. Teslic, M. Alaee and S. A. Smyth, J. Environ. Manage., 2015, 151, 22–30, DOI:10.1016/j.jenvman.2015.01.044.
- H. Wang, Z. Liu, J. Zhang, R. Huang, H. Yin, Z. Dang and Y. Liu, Sci. Total Environ., 2019, 692, 107–116, DOI:10.1016/j.scitotenv.2019.07.134.
- D. M. Kennes-Veiga, B. Vogler, K. Fenner, M. Carballa and J. M. Lema, Sci. Total Environ., 2021, 776, 146564, DOI:10.1016/j.scitotenv.2021.146564.
- E. Fernandez-Fontaina, I. B. Gomes, D. S. Aga, F. Omil, J. M. Lema and M. Carballa, Sci. Total Environ., 2016, 541, 1439–1447, DOI:10.1016/j.scitotenv.2015.10.010.
- W. O. Khunjar, S. A. Mackintosh, J. Skotnicka-Pitak, S. Baik, D. S. Aga and N. G. Love, Environ. Sci. Technol., 2011, 45, 3605–3612, DOI:10.1021/es1037035.
- N. H. Tran, T. Urase and O. Kusakabe, J. Hazard. Mater., 2009, 171, 1051–1057, DOI:10.1016/j.jhazmat.2009.06.114.
- M. Clara, B. Strenn and N. Kreuzinger, Water Res., 2004, 38, 947–954, DOI:10.1016/j.watres.2003.10.058.
- M. Clara, B. Strenn, O. Gans, E. Martinez, N. Kreuzinger and H. Kroiss, Water Res., 2005, 39, 4797–4807, DOI:10.1016/j.watres.2005.09.015.
- A. Joss, E. Keller, A. C. Alder, A. Göbel, C. S. McArdell, T. Ternes and H. Siegrist, Water Res., 2005, 39, 3139–3152, DOI:10.1016/j.watres.2005.05.031.
- Y. Zhang, S.-U. Geißen and C. Gal, Chemosphere, 2008, 73, 1151–1161, DOI:10.1016/j.chemosphere.2008.07.086.
- S. Chiron, E. Gomez and H. Fenet, Environ. Sci. Technol., 2010, 44, 284–289, DOI:10.1021/es902129c.
- D. Saidulu, A. Bhatnagar and A. K. Gupta, J. Environ. Chem. Eng., 2025, 13, 116106, DOI:10.1016/j.jece.2025.116106.
- N. Wang, Y. Xu, L. Peng, C. Liang, S. Song and M. Quintana, Sci. Total Environ., 2025, 958, 177836, DOI:10.1016/j.scitotenv.2024.177836.
- S. J. Wolfson, A. W. Porter, J. K. Campbell and L. Y. Young, Microb. Ecol., 2018, 76, 362–371, DOI:10.1007/s00248-017-1136-2.
- S. J. Wolfson, A. W. Porter, T. S. Villani, J. E. Simon and L. Y. Young, Environ. Toxicol. Chem., 2019, 38, 1585–1593, DOI:10.1002/etc.4406.
- A. K. Brown and C. S. Wong, Water Res., 2018, 144, 774–783, DOI:10.1016/j.watres.2018.08.034.
- A. Göbel, A. Thomsen, C. S. McArdell, A. Joss and W. Giger, Environ. Sci. Technol., 2005, 39, 3981–3989, DOI:10.1021/es048550a.
- S. Achermann, V. Bianco, C. B. Mansfeldt, B. Vogler, B. A. Kolvenbach, P. F. X. Corvini and K. Fenner, Environ. Sci. Technol., 2018, 52, 6265–6274, DOI:10.1021/acs.est.7b06716.
- A. K. Brown, J. Ackerman, N. Cicek and C. S. Wong, Environ. Pollut., 2020, 265, 114852, DOI:10.1016/j.envpol.2020.114852.
- T. Alvarino, P. Nastold, S. Suarez, F. Omil, P. F. X. Corvini and H. Bouju, Sci. Total Environ., 2016, 542, 706–715, DOI:10.1016/j.scitotenv.2015.10.140.
- F. I. Hai, X. Li, W. E. Price and L. D. Nghiem, Bioresour. Technol., 2011, 102, 10386–10390, DOI:10.1016/j.biortech.2011.09.019.
- E. Kaiser, C. Prasse, M. Wagner, K. Bröder and T. A. Ternes, Environ. Sci. Technol., 2014, 48, 10208–10216, DOI:10.1021/es5024493.
- X.-S. Miao, J.-J. Yang and C. D. Metcalfe, Environ. Sci. Technol., 2005, 39, 7469–7475, DOI:10.1021/es050261e.
- Y. G. Puranik, A. K. Birnbaum, S. E. Marino, G. Ahmed, J. C. Cloyd, R. P. Remmel, I. E. Leppik and J. K. Lamba, Pharmacogenomics, 2013, 14, 35–45, DOI:10.2217/pgs.12.180.
- N. Vieno, T. Tuhkanen and L. Kronberg, Water Res., 2007, 41, 1001–1012, DOI:10.1016/j.watres.2006.12.017.
- Y. Zhang, S.-U. Geißen and C. Gal, Chemosphere, 2008, 73, 1151–1161, DOI:10.1016/j.chemosphere.2008.07.086.
- K. C. Wijekoon, F. I. Hai, J. Kang, W. E. Price, W. Guo, H. H. Ngo and L. D. Nghiem, Bioresour. Technol., 2013, 144, 247–254, DOI:10.1016/j.biortech.2013.06.097.
- M. Dubey, A. Rajpal, B. P. Vellanki and A. A. Kazmi, Sci. Total Environ., 2022, 808, 152131, DOI:10.1016/j.scitotenv.2021.152131.
- W. Xue, C. Wu, K. Xiao, X. Huang, H. Zhou, H. Tsuno and H. Tanaka, Water Res., 2010, 44, 5999–6010, DOI:10.1016/j.watres.2010.07.052.
- A.-K. Ghattas, F. Fischer, A. Wick and T. A. Ternes, Water Res., 2017, 116, 268–295, DOI:10.1016/j.watres.2017.02.001.
- M. Martins, S. Sanches and I. A. C. Pereira, J. Hazard. Mater., 2018, 357, 289–297, DOI:10.1016/j.jhazmat.2018.06.001.
- D. Wolff, D. Krah, A. Dötsch, A.-K. Ghattas, A. Wick and T. A. Ternes, Water Res., 2018, 143, 313–324, DOI:10.1016/j.watres.2018.06.033.
- M. Martínez-Quintela, S. Balboa, J. R. Coves, F. Omil and S. Suárez, J. Hazard. Mater., 2023, 442, 129983, DOI:10.1016/j.jhazmat.2022.129983.
- C. Zwiener and F. H. Frimmel, Sci. Total Environ., 2003, 309, 201–211, DOI:10.1016/S0048-9697(03)00002-0.
- D. Saidulu, S. Agrawal, A. Bhatnagar and A. K. Gupta, Bioresour. Technol., 2024, 392, 129998, DOI:10.1016/j.biortech.2023.129998.
- N. Lakshminarasimman, M. M. McKnight, J. D. Neufeld and W. Parker, Bioresour. Technol., 2025, 423, 132207, DOI:10.1016/j.biortech.2025.132207.
- M. M. McKnight, PhD thesis, University of Waterloo, 2025, Available at: https://hdl.handle.net/10012/21381 Search PubMed.
- S. M. Abtahi, M. Petermann, A. Juppeau Flambard, S. Beaufort, F. Terrisse, T. Trotouin, C. Joannis Cassan and C. Albasi, Sci. Total Environ., 2018, 643, 1464–1480, DOI:10.1016/j.scitotenv.2018.06.303.
- D. Wolff, L. Helmholz, S. Castronovo, A.-K. Ghattas, T. A. Ternes and A. Wick, Water Res., 2021, 202, 117441, DOI:10.1016/j.watres.2021.117441.
- E. Torresi, S. J. Fowler, F. Polesel, K. Bester, H. R. Andersen, B. F. Smets, B. Gy. Plósz and M. Christensson, Environ. Sci. Technol., 2016, 50, 9279–9288, DOI:10.1021/acs.est.6b02007.
- T. Leiviskä and S. Risteelä, Environ. Pollut., 2022, 292, 118433, DOI:10.1016/j.envpol.2021.118433.
- B. Lesjean, R. Gnirss, H. Buisson, S. Keller, A. Tazi-Pain and F. Luck, Water Sci. Technol., 2005, 52, 453–460, DOI:10.2166/wst.2005.0723.
- R. Reif, F. Omil and J. M. Lema, in Comprehensive Analytical Chemistry, ed. M. Petrovic, D. Barcelo and S. Pérez, Elsevier, 2013, vol. 62, pp. 287–317, DOI:10.1016/B978-0-444-62657-8.00009-4.
Footnote |
| † Current Address: Stantec, Toronto, ON, Canada. |
| This journal is © The Royal Society of Chemistry 2026 |




