Open Access Article
Open Access ArticleAmmonia oxidizer adaptation to low dissolved oxygen concentrations for biological nutrient removal – a review on oxygen affinity, dual-substrate limitation, and decay
Kester
McCullough
 *ab,
Charles B.
Bott
*ab,
Charles B.
Bott
 b and
Peter A.
Vanrolleghem
b and
Peter A.
Vanrolleghem
 a
a
aModelEAU, Département de génie civil et de génie des eaux, Université Laval, Québec, QC G1V 0A6, Canada. E-mail: kester.mccullough.1@ulaval.ca
bHampton Roads Sanitation District, 1434 Air Rail Avenue, Virginia Beach, VA 23455, USA
First published on 5th March 2026
Abstract
Low dissolved oxygen (DO) operation in suspended growth biological nutrient removal (BNR) systems has become increasingly important for energy-efficient and intensified treatment processes. Novel strategies like continuous low DO, ammonia-based aeration control (ABAC), partial-denitrification anammox (PdNA), and partial-nitritation anammox (PNA) are becoming more commonplace and can drive processes to operate in DO regimes far below conventional norms. Yet nitrification, particularly the first step (ammonia oxidation), must be maintained. This review provides the first comprehensive overview of experimental data on ammonia oxidizer adaptation to low DO, along with analysis on how and why ammonia oxidizer oxygen kinetics respond to these low DO environments. This review examines the available evidence that stable ammonia oxidation at low DO is possible and usually corresponds to a reduction in the estimated ammonia oxidation Monod oxygen half-saturation coefficient (KO2). These changes can arise from intrinsic shifts in microbial community structure (particularly increased comammox Nitrospira (CMX) abundance), from extant factors like floc and microcolony size, or both. Despite evidence for these shifts, the drivers, limits, and durability of kinetic adaptation remain open questions. Also reviewed is the potential interaction of oxygen and substrate affinity, which is poorly understood and infrequently studied, especially under the non-starvation ammonia conditions common in advanced processes. This review also highlights a need to better study ammonia oxidizer decay and decay rate adaptation to low DO. Despite being critical to determining ammonia oxidizer population and thus nitrification capacity, decay kinetics under low DO remain largely unmeasured, mischaracterized, or assumed constant. Existing methods for determining decay are not well suited to capture or characterize the impacts of low DO operation. These findings suggest a necessary shift in practice: ammonia oxidation kinetic parameters, especially KO2, should not be treated as fixed parameters. SRT, DO, substrate concentration, and community structure all interact to drive nitrification performance. This review seeks to provide a comprehensive and systematic review of existing experimental results within each of these domains, with perspective on future research needs and implications for design and operation.
Water impactReduction in WWRF operating DO (i.e. low-DO treatment) allows for major energy savings and novel treatment approaches. However, low-DO processes design is still an open question, especially with respect to nitrification. This critical review examines the evidence for stable nitrification at low DO, analyzes the drivers for nitrifier kinetic change in response to low DO, and highlights future research needs. |
1 Introduction
Low dissolved oxygen (DO) biological nutrient removal (BNR) processes are the subject of increasing research interest as a tool for process intensification. Operating an activated sludge process at lower DO requires less air supply thus significantly reducing energy. Additionally, novel nitrogen removal approaches like ammonia based aeration control (ABAC), or shortcut nitrogen removal strategies like partial-denitrification anammox (PdNA) and partial-nitritation anammox (PNA) which often rely on maintaining a specific ratio of unoxidized ammonia and nitrite/nitrate (i.e. ammonia-versus-NOx (AvN) control), often lead to lower bulk-liquid DO concentrations.1–3 In all of these scenarios, maintaining aerobic ammonia oxidation capacity is critical to maintaining high quality effluent.To avoid losing nitrification capacity when a BNR process is transitioned to low DO, the ammonia oxidizing organisms must adapt or their population must increase. While ammonia oxidizing populations are complex and contain diverse species, broadly speaking the ammonia oxidizers, or some fraction thereof, must attain a higher affinity for oxygen to operate at similar rates in a reduced oxygen environment and thus allow the treatment process to maintain nitrogen removal capacity.
Early research into nitrification processes in wastewater treatment noted the importance of dissolved oxygen,4–6 and the relationship between DO concentration and ammonia oxidation activity was often conceptualized as a Monod function.7,8 An example set of Monod curves for two different nitrifying populations with different maximum rates and Monod oxygen half saturation coefficients (KO2) values are shown in Fig. 1 to illustrate how these kinetics impact rates. In this example only the single Monod function of oxygen is shown, thus ammonia is assumed to be non-limiting (further sections of this paper go into detail on methods for treating multiple limitations).
 | ||
| Fig. 1 Two theoretical Monod functions showing ammonia oxidation rate as a function of oxygen with different maximum rates (μ1 and μ2) and KO2 (KO2,1 and KO2,2). | ||
Stenstrom and Poduska as late as 1980, noted that “…the reported effects of DO concentration on maximum growth rate and process dynamics are varied, and not well understood”.9 They claimed based on surveying the preceding 20 years of work from pure cultures, soil organisms, and activated sludge processes that nitrifiers were limited in their ammonia oxidation rate below DO concentrations of 1.0–2.0 mgO2 per L, but concluded that with long enough solids retention time (SRT) nitrification can be achieved with a DO concentration of 0.5–1.0 mgO2 per L, and that 0.3 mgO2 per L is the lowest DO concentration at which nitrification can be considered possible. From these and other early findings, official design guidance for nitrification within BNR processes suggested operating at a DO of 2.0 mgO2 per L to maximize nitrification efficiency without aerating unnecessarily.10 This principle is generally recommended to this day11 and even codified in design documents such as the Recommended Standards for Wastewater Facilities12 and the EPA Manual on Nitrogen Control.13
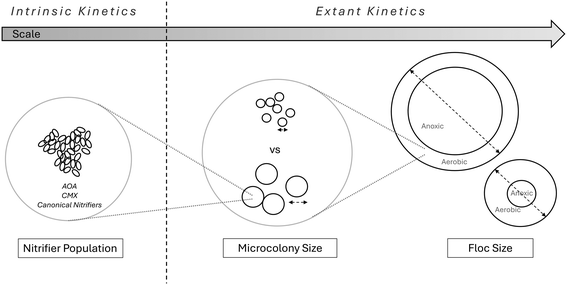
While this early literature mentions variance in nitrifier oxygen kinetics and contains studies of systems that operated at what would today be considered low DO, research was also conducted on explicitly operating nitrification at lower DO to better understand how this could be reliably achieved and understood. This research can be understood in the broader framework of variability in microbial kinetics as it is treated in wastewater processes. It has been noted by multiple researchers that kinetic parameters can vary between systems and over time, and in 1996 Grady et al.14 proposed terminology to characterize the sources of parameter change. Kinetics were conceived of as “intrinsic” or “extant”. Intrinsic kinetics were proposed to mean those to which the commonly used kinetic parameter symbols referred, but more particularly the kinetics of the underlying organism of interest, i.e. “If the kinetics truly reflect unrestricted growth they are representative only of the nature of the organisms and the substrate”.14 Extant kinetic parameters then are the microbial kinetics as they are observed in the system, or expressed by the organism given factors like the existing or recent conditions of substrate concentrations, mixing and advection, floc or biofilm structure and size, etc.Fig. 2 provides a graphical summary of the extant and intrinsic factors covered in this review, namely the ammonia oxidizing community makeup (intrinsic), the microcolony size (extant), and the floc size (extant).
Indeed numerous factors impact the observed or estimated (or apparent) KO2 for a given biomass on a given substrate within a given system, such as advection, diffusion, floc size and density, temperature, inhibition, substrate and enzyme concentrations, and more,15 as well as the dependence of the half-saturation coefficient on the maximum growth rate.16 Many of these phenomena are difficult to measure, however, and this study focuses on the intrinsic and extant drivers for which data are available in the literature. As a consequence of this, the challenge of transferability must also be considered in any discussion on kinetic parameter estimation. Due to the number of factors that influence observed kinetics, and the differing conditions between batch-scale tests and full-scale processes, kinetics estimated ex situ may not perfectly capture in situ kinetics.15,17 Better tools should be developed for in situ parameter estimation to better understand the topics covered herein.
For the sake of brevity and focus, NOB oxygen kinetics and their potential for adaptation are not within the scope of this paper, and studies exist comparing the observed differential response to operating DO of AOB vs. NOB.18–20 The focus of the kinetic portions of this work is explicitly on the first step of nitrification, ammonia oxidation (although comammox are capable of both steps). Similarly, N2O emission dynamics are not covered in this review. Although early studies suggested that low DO operation may increase N2O, particularly via autotrophic denitrification,21–23 recent studies appear promising (e.g. Jimenez et al.24 found that low DO adapted biomass produced more than two-fold lower N2O emissions than high DO adapted biomass exposed to low DO in a batch test). Further investigation is warranted on the impacts of long-term, stable, low DO operation on N2O production.
This critical analysis of the existing literature on low DO ammonia oxidation covers.
1) A review of studies wherein low DO ammonia oxidation is observed but ammonia oxidation kinetic adaptation is not explained.
2) A review of low DO nitrification studies wherein intrinsic kinetic changes are documented (for example, a different ammonia oxidizing species or strain is present).
3) A review of low DO nitrification studies wherein extant kinetic changes are documented.
4) A review of literature and concepts on the impact of single and multiple substrate limitations with respect to ammonia and oxygen for ammonia oxidation.
5) A review of literature on the decay rate of nitrifiers and how the many unknowns about decay may impact low DO nitrification.
2 Observations of ammonia oxidation adaptation to low DO
The possibility of ammonia oxidizer adaptation to low DO conditions has been observed by researchers in the past. This section provides an overview of observations of this phenomenon, demonstrating its existence, irrespective of whether the authors of those studies attempted to justify or explain it. Subsequent sections discuss the potential intrinsic and extant causes of this phenomenon, and existing limitations in the research.An early and influential study, Hanaki et al.25 conducted experiments using lab-scale reactors and synthetic wastewater with and without organic carbon operating at a 0.5 mgO2 per L DO and non-limiting DO. They estimated KO2 by comparing activities between these two batches, thus assuming the ammonia oxidizers in both reactors had the same oxygen kinetics, and found a value on the lower end of what may be considered typical. They reported that the ammonia oxidizers in the nitrification culture that grew at low DO conditions apparently had a much higher yield, which was used to explain how the apparent rates in both batches differed little despite operating at high/low DO setpoints and having the same KO2. It seems likely in this experiment that potential variations in ammonia oxidizer kinetics may have been incorporated in the assumptions and resultant calculations for other kinetic parameters, and it should be noted the yield values and biomass values could not be calculated directly but only relative to the rate.26 operated chemostats with nitrifiers derived from soil samples and determined that reduction in oxygen concentrations led to decreased KO2 for both ammonia oxidizing bacteria (AOB) and nitrite oxidizing bacteria (NOB), but that reducing DO too far led to severely reduced nitrification rates. Park et al.27 modified one of eight parallel trains at a full-scale UCT process to low DO operation (under 0.5 mgO2 per L) and found that the nitrification rate in the train did not decrease and it maintained complete nitrification. Of interest in this experiment was that all of the activated sludge was recombined in the clarifiers, meaning all of the nitrifiers were continuously exposed to low and high DO conditions. Daebel et al.28 operated two activated sludge pilot processes and determined that the KO2 value of nitrifiers were time-dependent, providing evidence that these kinetics should not be treated as constants.
Liu and Wang29 ran a series of lab-scale enriched nitrifier reactors at 10, 20, and 40 day SRTs across a range of DO concentrations from 0.1 to 5.0 mgO2 per L. They fit their process data across these operating conditions to a model that included a single value for KO2 for nitrifier growth (meaning adaptation per se was not modelled) and a separate KO2 value for nitrifier decay. It was hypothesized that the KO2 for decay was high enough to offset (through increased nitrifying biomass) the decrease in rate that low DO caused, as minimal loss of nitrification was seen at the low DOs.
Fan et al.30 and Wang et al.31 both operated lab-scale reactors at high and low DO and found lower KO2 for AOB at low DO conditions, the latter also identifying an increase in AOB yield coefficient. Microbial community analysis was performed and an increase in nitrifier abundance at low DO was noted, although the population composition of the genera tested did not appear to change appreciably at the two DO conditions.
Fig. 3 summarizes the findings of studies that specifically reported ammonia oxidizer KO2 values at both high and low operating DO concentrations. Operating DO and ammonia oxidizer KO2 values from each study are shown along with relative ammonia oxidation capacity contours (see the Fig. 3 caption for more detailed explanation). From these results, it is clear some researchers have found that ammonia oxidation capacity has been maintained or even increased as DO was reduced, but others have found a loss of relative ammonia oxidation capacity as a consequence of operating at low DO (due to insufficient adaptation). Despite varied experimental conditions in the studies shown in this figure, ammonia oxidation KO2 adaptation occurred, establishing the possibility of such adaptation. The rest of this manuscript attempts to detail the possible drivers of this adaptation; differences in adaptation may have been due to changes in floc morphology, microcolony structure, or nitrifier population (although not all of the studies included in this figure report on each of these metrics).
3 Ammonia oxidizer adaptation to low DO due to change in intrinsic kinetics
Changes to intrinsic ammonia oxidizer kinetics have been investigated primarily by looking at microbial community composition and attempting to identify shifts in the strains or species that make up the ammonia oxidizing community at high and low DO concentrations. Early work noted overall shifts in ammonia oxidizing populations that included canonical ammonia oxidizers and explored the sublineages present. Ammonia oxidizing archaea (AOA) were also the subject of investigation as they were found in multiple low DO systems. More recently, comammox were discovered and garnered significant attention due to their heretofore unknown but widespread presence in wastewater treatment processes; multiple researchers finding significant presence of these nitrifiers in low DO systems. Each of these types of population shifts (and the attendant changes in intrinsic kinetics) have been observed at low DO, but the understanding of how, why, and to what extent these population shifts will occur in any given low DO process is still an area of active research.3.1. Canonical ammonia oxidizing bacteria
Ammonia oxidizing bacteria (AOB) are a broad set of chemolithotrophic bacteria capable of oxidizing ammonia to nitrite; known for over a century.32 Numerous researchers have investigated their potential to perform ammonia oxidation at low DO concentrations. Park and Noguera33 ran two chemostats with enriched nitrifier cultures (10 day SRT) at high and low DO concentrations. It was determined that the reactors grew nitrifiers that were phylogenetically distinguishable, but both belonged to the Nitrosomonas europaea sublineage. The researchers claim that the modelled KO2 values for the two enrichments differed, but the differences appeared quite small and the confidence intervals of the estimates overlapped. Interestingly, the determined maximum specific growth rate for the low DO culture was significantly higher than for the high DO, but this is not commented on in the work. This work also documents a full-scale experiment in which two parallel activated sludge treatment trains were maintained, one with high DO and one with low DO for 6 months. At the end of the experiment, the AOB identified in the low DO process mostly belonged to the Nitrosomonas europaea sublineage, while the high DO train AOB were primarily associated with N. oligotropha. Park and Noguera34 also found that by isolating and kinetically testing two strains of AOB found in low DO nitrifying reactors that one had a high affinity for oxygen and a low affinity for ammonia (0.24 mgO2 per L and 1.62 mgN per L respectively) and the other had a low affinity for oxygen and a high affinity for ammonia (1.22 mgO2 per L and 0.48 mgN per L respectively). Bellucci et al.35 ran two sets of high and low (0.5 mgO2 per L) DO reactors on synthetic wastewater with the same SRT to demonstrate the ability of nitrifiers to grow and fully nitrify in low DO conditions. The ammonia oxidizers that grew in the low DO reactor were capable of complete ammonia removal and were estimated to have an unusually high yield. The yield was inferred from calculations where the AOB population (XAOB) was determined using a combination of biomass measures assuming a specific cell size and density and cell counts from qPCR. Metagenomic techniques were also employed to determine that the ammonia oxidizing community in the low DO reactor was a subset of and contained fewer species than the ammonia oxidizing community in the high DO reactor.3.2. Ammonia oxidizing archaea
Numerous researchers identified the potential for AOA to play a role in low DO nitrification. AOA have been noted to be successful ammonia oxidizers at extremely low DO concentrations (<0.1 mgO2 per L).36,37 Measurements of intrinsic AOA KO2 have been performed by multiple researchers; reported values range from 0.038–0.125 mgO2 per L (see Table 1). These KO2 values are lower than those typically estimated for canonical or mixed nitrifier communities, which range from 0.25 to >1.0 mgO2 per L under typical operating DO.20 Researchers have also found AOA present in full-scale activated sludge plants.38–40| Study | Reported KO2 (mgO2 per L) | Organism | Enrichment |
|---|---|---|---|
| Martens-Habbena et al.41 | 0.125 (0.018) | Marine AO (Ca. Nitrosopumilus maritimus strain SCM1) | Pure culture |
| Park et al.42 | 0.064 (0.014) | Marine sediment AOA (strain AR) | Enrichment culture |
| Qin et al.43 | 0.109 (0.00042) | Marine AO (Ca. Nitrosopumilus maritimus strain SCM1) | Pure culture |
| Qin et al.43 | 0.038 (0.00048) | Marine AOA (strain PS0) | Pure culture |
| Straka et al.44 | 0.093 (0.016) | Freshwater AOA (strain DW and AC2) | Enrichment culture |
| Straka et al.44 | 0.090 (0.045) | Soil AOA (Nitrososphaera viennensis) | Pure culture |
| Xie et al.45 | 0.131 (0.08) | Activated sludge + soil AOA (Nitrosocosmicus sp.) | Enrichment culture |
Park et al.46 used PCR to measure the presence of archaeal amoA in sludge samples from five water resource recovery facilities (WRRFs) operating at long SRTs and low DO. Their results provided evidence that AOA may be common in low DO nitrifying reactors. Wells et al.47 analyzed the abundance of different organisms in a full-scale WRRF and found the abundance of AOB amoA genes were strongly negatively correlated with operating DO and no correlation was found between DO and AOA amoA gene abundance. However, the plant was only operated at high DO concentrations. This was proposed as evidence that specific AOB may adapt to low DO conditions better than others (in this case Nitrosospira), and others have identified this possibility as well.48 Giraldo et al.49 observed that in a full-scale membrane bioreactor operated at low DO, the biomass had a very low KO2 value and using qPCR identified AOA as the dominant nitrifier. Liu et al.50 operated a 40 day SRT lab-scale membrane activated sludge reactor fed with synthetic wastewater at high DO (2.0 mgO2 per L) and low DO (0.4 mgO2 per L) and found that AOB abundance decreased while AOA abundance increased dramatically with lowering DO. The reported ratio of AOA/AOB amoA in the low DO condition was >19×. Jimenez et al.24 conducted a survey of 13 full-scale low DO facilities and found that for plants operated below 0.5 mgO2 per L, AOA abundance was 20% on average. Data were also provided for a single plant that transitioned from an operating DO of 2.0 mgO2 per L to 0.3 mgO2 per L, and AOA abundance was found to increase significantly from 9% (±6.2%) to 31% (±8.4%).
Since the development of methods to detect AOA, not all researchers experimenting with low DO nitrification have found them. Liu and Wang51 operated two bench-scale activated sludge reactors fed synthetic wastewater at 10 day and 40 day SRTs. Both reactors were operated in periods with high and low DO and no AOA were detected in either phase. Canonical AOB remained the dominant ammonia oxidizer and it was hypothesized this was due to the greatly reduced nitrifier decay rate at low DO which allowed the total population size of the nitrifiers to increase, not due to a change in KO2, yield, or growth rate. Arnaldos et al.52 ran two enriched culture sequencing-batch-reactors (SBRs) at high and low DO. AOA were not detected in either reactor but the same lineage of AOB was predominant in both; the low DO ammonia oxidizers had a lower KO2. Interestingly, the low DO nitrifying community developed enhanced expression of a heme protein within the cells, which was hypothesized to decrease the intracellular DO concentrations thus increasing the diffusive force of oxygen into the cell from the bulk liquid. Fitzgerald et al.53 ran two lab-scale, enriched culture reactors at 10 day SRTs at DO concentrations of 0.3 mgO2 per L or less. One of the reactors was seeded with sludge from a WRRF operating at high DO, the other from a WRRF operating at low DO. Almost no AOA were detected in either seed sludge or at the end of the experiment, demonstrating that AOA were not necessary for successful low DO nitrifier adaptation. The reactor seeded with low DO acclimated sludge began nitrifying at low DO quickly (<25 days) while the reactor seeded with high DO acclimated sludge took over 80 days to adapt and nitrify fully.
The state of the current literature on AOA as major factors in low DO nitrification is not clear, although there is no doubt remaining that AOA are capable of ammonia oxidation at very low DO concentrations and are present in some wastewater treatment plants. The earliest reports on the phenomena found both the presence46 and absence35 of AOA in low DO nitrifying reactors. Over a decade later, researchers of low DO nitrification still find conflicting results.50,53 The literature exploring this issue comes primarily from bench-scale reactors fed with synthetic wastewater at conditions that may differ from full-scale processes or from comparisons between samples taken from different wastewater treatment plants. At the very least, it would appear unwise from an engineering design perspective to rely upon the growth of AOA for low DO nitrification, and caution would be advised in reading too much into the measured kinetic parameters of different AOA strains as indicative of what would be expected in a full-scale WRRF. The exact conditions that cause the presence or absence of AOA in low DO processes are still not fully understood, and additional kinetic factors such as ammonia affinity and differential growth rates between and within species undoubtedly play a role.
3.3. Comammox
In 2015, a Nitrospira species was discovered that was capable of complete nitrification of ammonia to nitrate.54,55 This organism is referred to as comammox (complete ammonia oxidation) and has been of interest to researchers given its potential favorable kinetics for low DO, lower N2O emissions,56 and prevalence within nitrification processes despite being previously unrecognized. Different comammox (CMX) strains and clades have been identified, and they have been found in a diversity of activated sludge processes57–62 and even drinking water systems63 around the world. It has been suggested that CMX have low maximum ammonia rate and high growth yield.64 This is particularly interesting because for the past 30 years some researchers reported that nitrifiers had high yield when adapted to low DO (see Section 3.1). Recently attempts have been made to characterize the intrinsic KO2 of these organisms (Table 2); both studies found CMX to have extremely high oxygen affinity (KO2 < 0.02 mgO2 per L).Camejo et al.58 operated an 80 day SRT SBR fed synthetic wastewater and seeded with biomass from a full-scale process. Oxygen concentrations were kept low (<0.5 mgO2 per L) and significant CMX abundance was noted (very little AOA were identified). During a second phase of the experiment, still at low DO, CMX abundance dropped to near zero but full nitrification was maintained; no other known AOB or AOA were detected in this second phase and the authors speculated the presence of still-unrecognized ammonia oxidizing organisms. Roots et al.67 operated an intermittently aerated SBR using real wastewater for influent. DO setpoints were kept at 1.0 mgO2 per L or lower and complete nitrification was achieved; the process performed better than the full-scale high DO nitrification reactor at the facility from which the raw influent was taken, which was used as a control. After significant time operating at reduced DO, CMX composed >90% of ammonia oxidizers (measured by amoA) in the reactor. It should be noted the reactor was run with a very long SRT (99 days). Beach and Noguera68 pointed out some potential issues with qPCR primers in use at the time with detecting CMX and designed three new primers for some candidate CMX species; they found CMX present in the majority of samples analyzed from low DO systems, none in the high DO systems, and noted that the largest abundance was found in systems with long SRTs. How et al.60 operated a lab-scale nitrifying SBR fed real wastewater at a DO concentration of 1.7 mgO2 per L and then 0.9 mgO2 per L with a 20 day SRT and consistently achieved full nitrification. CMX were found to be the dominant nitrifiers throughout the experiment. Wen et al.20 reported on a full-scale membrane bioreactor (MBR) process that transitioned from relatively low (0.8 mgO2 per L) to even lower (0.5 mgO2 per L) operation at an 18 day SRT. Complete nitrification was maintained at both DO setpoints, and the already relatively low determined KO2 of AOB (0.23 mgO2 per L) decreased further to 0.19 mgO2 per L (although the error of these parameter estimates is not reported in the study, and it is possible these two values are not significantly different). Nitrospira were detected as the main nitrifying organism during the study, and it was speculated that these could be comammox or another unknown AOB. Li et al.69 operated two bench-scale MBRs at high and low DO, 2.0 mgO2 per L and 0.5 mgO2 per L respectively, and found that while both were capable of full nitrification, the high DO reactor ammonia removal was more consistent. CMX was found to be the dominant nitrifier in both reactors, particularly in the low DO reactor (the CMX/AOB amoA ratio was >17). Jimenez et al.24 found in their low DO survey that for full-scale plants operated below 0.5 mgO2 per L, CMX abundance was 66% on average. In the same study, a single plant was monitored before and after transition from an operating DO of 2.0 mgO2 per L to 0.3 mgO2 per L; CMX abundance was found to increase significantly from 11% (±4.7%) to 60.5% (±10.9%). Two recent studies have attempted comammox enrichment for the purpose of kinetic estimation. Hou et al.65 attempted to enrich Candidatus Nitrospira nitrosa under low DO conditions and succeed in increasing its prevalence 26.5 fold; the results of their subsequent kinetic measurements are shown in Table 2. Zhu et al.66 comparatively enriched Nitrospira-dominant (comammox) and Nitrosomonas-dominant floccular sludges and also reported a lower apparent oxygen affinity constant for the Nitrospira-dominant sludge, which are reported in Table 2.
Two studies have discussed the potential for only certain clades of CMX to be preferentially selected by low DO. Zheng et al.70 cultivated activated sludge samples in lab-scale reactors fed only ammonia with different DO concentrations ranging from 0.5–17 mgO2 per L. CMX were found to be the majority nitrifiers in each reactor and in the lowest oxygen reactor were enriched significantly beyond the predominance found in the seed sludge. AOB and AOA were also detected, but at much lower levels (making up 10% or less of the nitrifying population as measured by 16 s rRNA gene abundance). The majority of CMX in the low DO reactors was made up of a CMX strain from clade A, although they noted a different clade A strain appeared to be more prevalent at higher DO. Sabba et al.62 noted increased comammox abundance in a low DO process (10–12 day SRT) and a high DO process (12–14 day SRT), and found no comammox at another high DO facility (8.5 day SRT). The low DO facility had a very low estimated KO2 for nitrifiers (0.05 mgO2 per L) but this estimate was made using DO measurements that only went down to 0.25 mgO2 per L and CMX clade A and clade B were present. Both high DO facilities had higher estimated KO2 for nitrifiers (0.5 mgO2 per L at both) and one had CMX clade A present. Thus, it was suggested that the ability of CMX to grow at low DO and provide complete nitrification may not be a feature of the species, but only of specific strains.
Two early studies on CMX in nitrification processes operated reactors at quite long SRTs,58,67 and other researchers have noted that CMX may be more prevalent or require long SRT systems.59 However, more recent full-scale low DO studies have found CMX in abundance in relatively shorter (<15 day) SRT systems,62 including its consistent presence in over a dozen full-scale low DO facilities with SRTs ranging from 4.5–18.2 days.24 It appears, then, that long SRT is not a requirement for low DO CMX. Much is also left to learn about potential differences within CMX strains, and why some experiments appear to show CMX predominance at both low and high DO concentrations. As CMX may not be particular to low DO, the understanding of their role and presence in engineered systems is currently not enough to design around.
It does not appear that any low DO activated sludge studies, since the discovery of CMX and development of primers to detect it, have failed to detect CMX (although Liu et al.50 found it to be only a very small fraction); this may in fact be evidence against its particular significance to engineering design. If the organism is so ubiquitous, especially at low DO, then no special steps may be necessary to grow it. If it can be shown to have measurably different in-process kinetics, this still may not necessitate a focus on the organism itself for design (unlike the case of other novel organisms like anammox), only that those different kinetics are used for design purposes.
To provide a clear picture of the current low DO knowledge landscape, studies where both high and low DO operation were documented, and estimated ammonia oxidizer KO2 values were provided for both, are shown in Table 3. The nitrifier population information, if provided, is also summarized therein.
| Study | Processes | Operating DO (mgO2 per L) | Estimated KO2 (mgO2 per L) | Same biomassa | Comment | ||
|---|---|---|---|---|---|---|---|
| High | Low | High | Low | ||||
| a “Same biomass” indicates whether a study compared the behavior of a single biomass in a process at different operating conditions (Y), or two different biomasses from two different processes (N), where operating conditions, process configurations, and influent characteristics may differ. | |||||||
| Sabba et al.62 | Full-scale BNR | 5.00 | 0.71 | 0.50 | 0.05 | N | No AOA detected. CMX clade A found at high DO, CMX clade A + clade B found at low DO |
| Wang et al.31 | Lab-scale BNR | 2.00 | 0.50 | 0.39 ± 0.05 | 0.29 ± 0.04 | Y | Slightly higher canonical AOB and Nitrospira abundance at low DO |
| Wen et al.20 | Full-scale MBR | 0.80 | 0.30 | 0.23 | 0.19 | Y | No AOA detected. Nitrospira was the only known nitrifier detected |
| Keene et al.71 | Full-scale BNR + pilot BNR | 1.80 | 0.33 | 1.38 ± 0.23 | 0.30 ± 0.14 | N | No AOA detected. Overall nitrifier population diverged between high and low DO processes |
| Fan et al.30 | Lab-scale BNR | 1.00 | 0.50 | 0.54 ± 0.09 | 0.14 ± 0.02 | Y | — |
| Liu and Wang29 | Lab-scale BNR | 4.2 | 0.16 | 0.71 | 0.39 | Y | — |
| Jimenez et al.24 | Full-scale BNR (A/O) | 2.0 | 0.3 | 0.5 | 0.2 | Y | Increase in AOA and CMX abundance, details provided in text |
4 Ammonia oxidizer adaptation to low DO due to change in extant kinetics
The second category of changes to the kinetics relevant to ammonia oxidizer low DO adaptation is change in the extant, or observed, kinetics. Extant kinetics may include anything not considered in the intrinsic kinetics of the organisms; for nitrifiers this has often been advection and diffusion limiting mechanisms, such as the floc size (in a suspended growth system) and more recently the microcolony size and number (within the floc). Both of these factors have been found relevant to nitrifier oxygen kinetics, and are thus of interest when considering adaptation to low DO.4.1. Floc size
Based on a general understanding of advection and diffusion, floc size (and the related field of biofilm formation/thickness) have long been of interest to wastewater process researchers. Early on Beccari et al.72 demonstrated an empirical relationship between KNH and measured floc size. Beccari et al.73 then developed an early model to demonstrate that floc size significantly impacted nitrifier oxygen kinetics, and validated this model with experimental data. Their steady-state model assumed uniform biomass distribution with fixed intrinsic kinetic parameters in a spherical floc with negligible external diffusional resistance (essentially no boundary layer). Experimental data to validate the model came from batch tests run with non-limiting ammonia concentrations and low TSS concentrations (very high F/M) wherein DO was increased to non-limiting conditions and modelled as it dropped to zero; floc sizes were measured with a particle size analyzer. They found that at particle size diameters ≤40 μm the internal diffusion resistances can be neglected, but at larger diameters (common in wastewater treatment) and especially at low DO the impact of floc size becomes pronounced: larger internal diffusion resistance will lead to higher apparent measurements of KO2.Chu et al.74 conducted measurements of heterotrophic KO2 in a respirometer at various mixing energies (mean energy dissipation rate). They found that decreased mixing energies led to larger floc diameters and much higher KO2 value below a minimal threshold. It should be noted, however, that mixing energy can affect both floc size and advection, and this study does not attempt to differentiate these impacts separately. Manser et al.75 operated an MBR and a conventional activated sludge (CAS) pilot plant to develop and measure the oxygen kinetics of small (80 μm) and large (300–500 μm) floc systems respectively (both at 20 day SRT with non-limiting DO concentrations). Floc size was measured using a laser light scattering device. A model was developed similar to Beccari et al.73 except that a boundary layer was included and the floc biomass was heterogenous – nitrifiers were assumed to be concentrated in the interior of the floc and heterotrophs were distributed uniformly throughout. The experimental and modelling results demonstrated that AOB, NOB, and OHO KO2 values were estimated to be 2–3 times higher in the process with larger floc sizes, and this difference could be reasonably well explained with their floc model. Daigger et al.76 used DO microprobes to actually measure the DO concentration within individual flocs, providing direct evidence of what had previously been speculated and modelled to occur. Wu et al.77 used sonication to break apart flocs and achieve different floc sizes. They found that the measured KO2 of AOB increased from ∼0.20 mgO2 per L at 50 μm average floc diameter to ∼1.25 mgO2 per L at 450 μm average floc diameter. However, the NOB KO2 was found to be less sensitive to floc diameter. The lower NOB KO2 observed in this study was explained by their 1-D diffusion model, but the authors speculate microcolony structure (discussed in the next section) plays an important role as well. Keene et al.71 demonstrated that their low DO pilot process (<0.33 mgO2 per L) had smaller floc sizes compared with the control high DO full-scale process (152 μm vs. 117 μm, measured via image analysis). Ammonia oxidizers in the low DO pilot had a lower measured KO2 as well (0.30 mgO2 per L vs. 1.38 mgO2 per L). Based on the previously published models and experimental data, Keene et al.71 finding such a large difference in KO2 with such a small difference mean floc diameter may well be evidence against floc size necessarily being a major driver of KO2 in these systems. Yu et al.78 explored AOB/NOB kinetic competition but in the process of doing so reported estimated AOB KO2 values at varying floc sizes. Data from these studies, as well as those on the impact of microcolony size on nitrifier KO2, are summarized in Fig. 4. This figure shows the overall positive correlation, and even similar slopes found amongst some researchers, between floc size and nitrifier KO2 across a wide range of particle sizes.
 | ||
| Fig. 4 Reported ammonia oxidizer KO2 values for differing floc sizes and microcolony sizes as reported in the literature. | ||
Floc size may significantly influence ammonia oxidizer KO2, and is itself influenced by process operational conditions. SRT in particular has been observed to play a role in floc size. Hocaoglu et al.79 found a reduction in floc size at reduced SRT (60 days to 20 days) in an MBR along with lower model-estimated KO2 values. Li and Stenstrom80 also documented a positive relationship between SRT and floc size in a lab-scale MLE, lab-scale IFAS, as well as with full-scale data.
4.2. Microcolonies
In addition to floc size, what occurs within the floc may determine observed KO2 just as much, if not more. While this field of research is much newer, there has been significant research interest in the impacts of nitrifier microcolonies, or cell clusters, within flocs. Manser et al.81 ran an MBR and CAS pilot in parallel and found using metagenomic techniques no significant difference in nitrifying population. They found that the MBR system not only lead to smaller floc sizes, but to smaller AOB microcolony sizes as well. Picioreanu et al.82 constructed a 3D model of AOB and NOB microcolonies within flocs at different sizes and used this to analyze the data previously collected by Manser et al.81 They demonstrated with this model that larger microcolonies, like larger flocs, lead to higher observed KO2 values. They claim the impact of colony size is more significant than the impact of floc size, but this significance is itself contingent upon numerous additional variables (biomass concentration, number of colonies, uniformity of colony size, etc.). Picioreanu et al.82 also claimed that in general longer SRT could lead to larger microcolonies, mirroring the relationship with floc size. Thus, longer SRTs may lead to higher extant KO2 values, if microcolony size is in-fact a dominant driving force in the phenomena.Keene et al.71 observed, along with the reduction in floc size mentioned previously, a reduction in AOB colony size at low DO. This reduction (40%) was larger than the noted reduction in floc size (20%). Either or both of these reductions are posited to have contributed to the lower observed KO2 of nitrifiers at low DO. Law et al.19 in a study on AOB vs. NOB kinetic selection via DO concentration changes, demonstrated that AOB and NOB expressed very different microcolony structures. They suggest, among other things, that apparent half-saturation values may be a function of microcolony shape and porosity, not merely microcolony diameter, adding an additional layer of complexity to the claims of Picioreanu et al.82
5 Single and multiple substrate limitations in the context of ammonia and oxygen limitation of ammonia oxidation
The majority of work on low DO nitrification processes has been done on systems that achieve, or seek to achieve, complete ammonia oxidation. However, novel processes such as ammonia based aeration control (ABAC), partial-denitrification anammox (PdNA), and partial-nitritation anammox (PNA) and their various implementations are designed to perform incomplete aerobic nitrification, leaving an appreciable ammonia concentration throughout the nitrification process. For the anammox processes, ammonia is not oxidized completely in the aerobic process so that anaerobic ammonia oxidation may occur. These processes have been an area of intense research for the past few years, and full-scale implementations have been achieved.83,84 The drivers for PdNA and PNA are reduced external carbon demand for denitrification, reduced oxygen demand for aerobic ammonia oxidation, and reduced supplemental alkalinity design. These processes can also realize significant capacity savings, as maximum available aerobic SRT for nitrification generally determines the capacity of a BNR process, and anammox allows shifting the burden of ammonia oxidation from aerobic to anaerobic (often biofilm) processes.85Little-to-no work has been done to analyze how this residual ammonia concentration (non-starvation conditions) intersects with low DO operations. As documented by Arnaldos et al.15 with respect to other organisms, differing substrate concentrations and conditions may lead to differing substrate degradation pathways. In the numerous experiments discussed so far, some processes had no measurable effluent ammonia, meaning some portion of the process must be substrate limited (starvation), while others had 1–2 mgNHx–N per L or more; little discussion or attention is given to this by study authors. Of course, residual ammonia concentration is also dependent on the SRT of the system9 so the two factors, substrate limitation and SRT, cannot be easily disentangled.
The possibility of multiple limiting substrates (in this case, oxygen and ammonia) has long been discussed in the activated sludge literature. The earliest assumptions were that only a single substrate could be limiting.9 If there were potential for multiple limiting substrate it was taken to be non-interactive, meaning only one substrate could be limiting at a time (the most limiting one). Double Monod (interactive) functions, where two Monod limitation functions are multiplied together, were suggested86 but also dismissed by other researchers.87 Doubts were expressed both ways, whether it would be right to dismiss the complexity of an engineered process with many substrates or how it could make sense to multiply together ever-more limiting mathematical terms.9,88,89 Numerically, the multiplicative Monod functions are simpler to solve than the non-interactive single limiting substrate functions.
Additional interacting models of varying complexity were introduced over time. It is not clear exactly if or how this issue was resolved, but most activated sludge modelling software available today appears to rely primarily, but not solely, on multiplicative Monod functions. Stewart et al.90 provide a thorough overview and analytical comparison between some single and multiple Monod models. There does not appear to be a simple answer to how to apply these models to oxygen and ammonia limitations for nitrifier rates, but most of the research already surveyed herein appears to use the multiplicative model.
Beyond the question of how to model the potential interaction of multiple Monod terms, there appear open questions about what occurs at the biological level with multiple limiting substrates. Some research suggests that different ammonia concentrations can select for different types of AOB through different substrate affinity, and a similar phenomenon applies to oxygen as well.91 Park and Noguera34 documented two strains of AOB which had either high affinity for DO and low affinity for ammonia, or vice versa. However, low DO operation has been shown to shift NOB communities towards populations that have low half-saturation values for both oxygen and ammonia.92 Microcolony or cluster density has also been suggested as a cause for the large range of KNH and KO2 observed in AOB, even within the same lineages,91 but this is unfortunately one of the more limited areas of the literature currently. Very low ammonia concentrations may drive AOA growth, as they have been reported to have KNH values for ammonia far below those of AOB.93,94 Liu et al.50 hypothesized this kinetic difference was responsible for the shift of nitrifier population to AOA at low DO in their experiment. CMX have similarly been reported to have significantly higher affinity for ammonia.61
6 Nitrifier decay rate and low DO
Multiple studies of microbial cultures have shown that AOB survive starvation well, and have a good ability to generate energy as soon as substrates are available again.91 It has been well documented that nitrifier decay rates are much higher, an order of magnitude larger or more, in aerobic conditions versus anoxic conditions.95,96 Low DO conditions could therefore impact not only growth conditions, but decay conditions as well, and there is very limited evidence that ammonia concentrations could play a role in decay also. Little literature exists comparing oxygen or ammonia limitation impacts on nitrifier decay, but some pure culture work shows that ammonia starvation drives a 5-fold higher decay rate than oxygen starvation,97 though these results have been met with skepticism and have not been replicated.91 The role of ammonia starvation on decay is not reflected in any current models, perhaps with good reason as ammonia is available throughout the decay process (thus also available for regrowth) from the death and lysis of other organisms (namely heterotrophs), as will be discussed further on.Most of the low DO work herein surveyed has paid little attention to decay rates: pure and enriched culture studies rarely measure it distinctly and the original ASM models do not include any kinetic modifiers to nitrifier decay but instead treat it as constant.98 Munz et al.99 provides a good overview of how other researchers have proposed implementing decay dynamics in ASM models as well as proposing their own implementation. The authors recognize that aerobic, anoxic, and anaerobic decay rates of nitrifiers differ and various kinetic switches between these rates are discussed and analyzed. The kinetics can become quite complex and difficult to measure, depending on whether the KO2 values for decay and growth are the same or distinct (both approaches are used in the literature). If independent values for KO2 for decay are used, they appear to typically be drawn from decay rate measurements at various high DO concentrations99 or extrapolation between highly aerobic and anoxic decay rates.95,96,100
Two studies (Liu and Wang29,51) focused on decay as a driver for low DO nitrification specifically and determined the decreased decay rate at low DO conditions was the primary contributor to why nitrification rates did not decrease at low DO. Liu and Wang51 used metagenomic evidence to bolster this claim. Liu and Wang29 modelled steady state operation across a range of DO setpoints and fit a model to the reactors' effluent NHxvs. DO. This model assumed two separate, but constant, KO2 for growth rate and decay rate (each a single Monod function). This model form, in disallowing for adaptation (different KO2 at different DO setpoint), forces the variability across the DO operating conditions to be a function of decay difference, and the model fits are very dependent on very small differences in effluent ammonia. Further, this work also suggested that the nitrifiers do adapt and different KO2 values were measured using batch-tests from the highest and lowest DO reactors. However, the authors do not attempt to reconcile these two methods, and no attempt is made to separately batch-test measure the decay rate or KO2 for the decay rate.
Mehrani et al.101 proposed a lower value for comammox decay rate than conventional nitrifiers but did not provide a literature source for this value. As far as can be determined, no explicit considerations for decay rate have been proposed for AOA.
Methods used to measure decay generally follow the same format: a reactor of sludge is aerated for many days and samples are taken from this reactor at intervals and the maximum nitrification rate is measured.102 The decline in this maximum nitrification rate over time is an indicator of the decay rate. It should be noted, then, that this method to measure decay rate is subject to the problems of transferability as well, and here to a large degree as the conditions in the decay reactor will differ widely from those the organisms ever experience in the treatment process. There are no commonly accepted alternatives to measure this parameter. There are shorter-duration, but still ex situ, methods for measuring maximum specific growth rate and decay simultaneously, but these require assumptions about one or the other value and do not get around the transferability problem.
Melcer et al.103 performed a comprehensive evaluation of many kinetic estimation methods including nitrifier decay rate. They note that estimated decay rates in the literature have not only varied widely, but varied with time as different methods for measuring decay were popular during different decades. Experimental methods for determining maximum specific growth rate will be more or less sensitive to decay rates depending on the SRT of the experiment. A new modelling approach to estimate decay was developed by this group, taking into account the ammonia released by heterotrophic death–lysis–hydrolysis in the decay reactor. This method requires regularly measuring the nitrate concentration in the decay reactor but can impact the calculated nitrifier decay value by almost 50%. If this is the case, this would call into question many of the nitrifier decay rate estimates published before this paper, as this method was novel when introduced. Pruden et al.104 provide the most comprehensive recent analysis of nitrifier decay rate measurements in which decay rate measurements were performed using typical methods and DNA measurement methods. Using the method of Melcer et al.103 they found similar values of nitrifier decay rates. However, it was noted in their work that considering the ammonia production in the decay reactor from heterotrophic decay made almost no difference to the estimated decay rate, in stark contrast with Melcer et al.103 They also noted considered variability in nitrifier activity measurements (more so AOB than NOB) across hundreds of tests at various times and temperatures, noting that many outlier values had to be discarded (and different decay values were estimated, depending on which values were taken to be outliers). Importantly, the AOB decay rate estimated using DNA measures was 50% less than the AOB decay rate estimated by loss of nitrification activity.
The potential for nitrifier dormancy rather than true decay should also be considered. Because decay is typically inferred from a loss of measured nitrification activity, it can be difficult to distinguish inactivity from biomass loss. Dormancy is generally viewed as reversible, but the timescale for reactivation is not well defined. Comparative measurements of dormancy versus decay are also limited. For example, AOB can maintain abundance while showing significantly reduced amoA expression during cold-temperature nitrification failure.105 Johnston et al.106 found that AOB and AOA could be revived after two months of starvation, long after AOB amoA dropped below detection limits. Pruden et al.104 note that molecular detection does not confirm viability, while culture-based counts can underestimate viable cells (e.g., sublethal injury can yield nonculturable but still viable cells). Friedrich et al.107 also suggested that differential DNA marker versus activity decay rates may be due to dormancy.
For consideration in design, it is likely that decay rates and their kinetics play a role in low DO nitrification and adaptation, but to what extent is not easily demonstrated. Decay rate is a critical kinetic as it determines the population (XNITO) of nitrifiers in a process at a given load and SRT, and the nitrifier population and maximum specific growth rate together determine the maximum ammonia uptake rate of the process, which governs capacity. It is also well established that nitrifier decay rates depend on oxygen concentrations (at the very least, they are quite different between anoxic and aerobic conditions). Given a model structure where decay rates vary as a function of DO it would be difficult to determine whether nitrifiers at low DO have lower decay rates per se (intrinsic kinetics) or they are merely kinetically limited due to operating DO (extant kinetics) and would be harder still to determine if this decay kinetic factor is itself altered by operating DO concentration (KO2-decay is itself adapting). A discussion of extant and intrinsic decay kinetics and decay kinetic adaptation with respect to nitrifiers and oxygen appears to be absent from the literature.
Current decay rate values, and their associated oxygen kinetics, despite being one of the most important factors affecting nitrifier population and therefore nitrification capacity, should be met with some skepticism as:
1) It is not clear whether decay oxygen kinetics should share the same KO2 value as growth rate, and if not, how it should be measured.
3) No attempt has been made to understand decay adaptation to low DO and what proportion is extant or intrinsic.
3) No explicit measures of decay rates at low DO exist, only extrapolations from high DO decay or anoxic decay.
4) Decay rates in the existing literature may have been measured without taking into account ammonia release from heterotroph decay, but reports vary significantly on whether this matters.
5) AOB decay rate is much more variable than NOB decay rate even when measured using the best methods available, but it is not clear why.
6) AOB decay does not align with death (measured via DNA destruction) implying some kind of dormancy may occur, and measured decay rates may only reflect an immediate effect on nitrification capacity.
7) Current measures of decay are ex situ methods that differ drastically from extant, in-process conditions.
7 Conclusions
This critical review has surveyed the literature on low dissolved oxygen ammonia oxidation with regard to understanding the mechanisms by which ammonia oxidizer communities adapt to low dissolved oxygen conditions. The evidence demonstrates that stable ammonia oxidation at low DO is achievable in activated sludge systems and consistently correlates with reduced apparent ammonia oxidizer KO2 values. Studies spanning laboratory reactors to full-scale wastewater treatment plants have documented successful ammonia oxidation at DO concentrations well below the conventional 2.0 mgO2 per L threshold.Although early studies noted the possibility for ammonia oxidizer adaptation to low DO conditions, in the intervening decades enough data has been gathered to lay out a more concrete understanding of this phenomenon. The mechanisms underlying this adaptation can be characterized as belonging to two different pathways, although they lie along a spectrum: intrinsic and extant changes; the available data on these mechanisms has been presented in this study.
Intrinsic kinetic adaptation involves shifts in the ammonia-oxidizing community toward organisms with higher oxygen affinity. Both AOA and CMX have been shown to have very high oxygen affinity, and both have been documented in low DO systems. In fact, every low DO nitrification study that has looked for CMX has found them. Work on improving CMX detection method accuracy is ongoing; further information on CMX primer development and specificity for detecting clade A and clade B can be found in.108–110 The predominance of these organisms with lower intrinsic KO2 would suggest lower observed KO2 in these systems, which the published research demonstrates.
Extant kinetic adaptation encompasses a broad category of physical and structural changes that affect the apparent oxygen affinity of the biomass without necessarily changing the underlying microbial community. Significant data with respect to ammonia oxidation and operating DO has been gathered for two of these factors: floc size and microcolony size. Multiple studies have demonstrated a link between floc size and apparent KO2 as well as microcolony size and KO2. Increased floc size leads to higher apparent KO2, most likely through an increase of diffusional resistance against DO entering the floc.
It remains unclear how intrinsic (community) shifts and extant (mass-transfer/structure) effects interact in situ, largely because almost no studies reporting reduced apparent KO2 concurrently quantified floc or microcolony structure. The limited available evidence (one study) suggests that intrinsic shifts can be the dominant driver. In the only study that paired oxygen-kinetics shifts with detailed community characterization, a substantial decrease in apparent KO2 coincided with a marked nitrifier community shift (including increased CMX) while floc size changed little, implying that extant changes were not necessary to explain the observed kinetic improvement.71 More broadly, even if unmeasured floc or microcolony changes occurred in other studies, the consistent magnitude and direction of apparent KO2 reductions across systems indicates that any concurrent extant effects were not sufficient to overwhelm the net shift toward higher apparent oxygen affinity observed under sustained low-DO operation.
The pathway to low DO ammonia oxidation, and thus increased potential for process intensification is then: changing operating conditions (lower DO) places enough selective pressure on ammonia oxidizers to generate a community shift over time, which eventually lowers the apparent KO2 and allows for maintaining a substantial fraction of ammonia removal capacity at the reduced operating DO. The timeline for implementing this is not well established; Jimenez et al.,24 surveying over a dozen full-scale low DO facilities, reported that approximately 20 days was needed for full acclimation. It was also reported that the facilities in the study also did not need to increase their SRT to accommodate reduced ammonia removal rates, meaning capacity was maintained as well.
This critical review also surveyed the literature on additional important considerations for ammonia oxidation at low DO. 1) Higher available substrate (ammonia) concentration, a feature in advanced processes like ABAC or those with downstream anammox like PNA or PdNA, may have a significant impact on apparent ammonia oxidizer kinetics, but little research has addressed this explicitly. 2) Significant open questions remain for the impact of oxygen on ammonia oxidizer decay rates, which are critical for process design and kinetic estimation or modelling. 3) Ammonia oxidizer decay rates are likely impacted by operation at low DO and are critical for predicting nitrification capacity, but they are difficult to measure and kinetically parameterize due to problems of methodological accuracy and transferability; numerous open questions remain.
For designers, owners, and operators, process intensification is an important and attainable goal. But these novel processes typically demand operation at DOs outside the typical ranges, and the limits of current knowledge and process design are tested. This is compounded as these operating conditions significantly impact and interact with each other, but the state-of-knowledge must be advanced, as these processes need to be designed, built, and operated. Kinetic adaptation is real, although questions remain about the durability of these shifts, but it is not sufficient to assume static ammonia oxidation kinetics, especially with respect to oxygen. Delineating the intrinsic and extant nature of these shifts provides some evidence about this, but significant work remains to be done. Future work must also resolve outstanding questions around decay kinetics and substrate interactions, especially in systems with residual ammonia concentrations, to better predict system capacity and resilience under low DO conditions.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported in-part by the NSERC Discovery Grant RGPIN-2021-04347 ‘Towards digital twin-based control of water resource recovery facilities – Methods supporting the use of adaptive hybrid digital twins’.References
- P. Regmi, B. Sturm, D. Hiripitiyage, N. Keller, S. Murthy and J. Jimenez, Combining continuous flow aerobic granulation using an external selector and carbon-efficient nutrient removal with AvN control in a full-scale simultaneous nitrification-denitrification process, Water Res., 2022, 210, 117991 CrossRef CAS PubMed.
- P. Regmi, M. W. Miller, B. Holgate, R. Bunce, H. Park, K. Chandran, B. Wett, S. Murthy and C. B. Bott, Control of aeration, aerobic SRT and COD input for mainstream nitritation/denitritation, Water Res., 2014, 57, 162–171 CrossRef CAS PubMed.
- L. Rieger, I. Takács and H. Siegrist, Improving nutrient removal while reducing energy use at three swiss WWTPs using advanced control, Water Environ. Res., 2012, 84, 170–188 CrossRef CAS PubMed.
- E. Ardern and W. T. Lockett, Experiments on the oxidation of sewage without the aid of filters, J. Soc. Chem. Ind., London, 1914, 33, 523–539 Search PubMed.
- S. Rideal, The aeration test for sewage effluents, Analyst, 1901, 26, 197–202 Search PubMed.
- S. Rideal, The purification of sewage by bacteria, J. R. Soc. Arts, 1897, 46, 81–90 Search PubMed.
- G. Knowles, A. L. Downing and M. J. Barrett, Determination of kinetic constants for nitrifying bacteria in mixed culture, with the aid of an electronic computer, Microbiology, 1965, 38, 263–278 CrossRef CAS PubMed.
- B. A. Southgate, Water Pollution Research, Water Pollution Research Board, H. M. Stationery Office, London, UK, 1963 Search PubMed.
- M. K. Stenstrom and R. A. Poduska, The effect of dissolved oxygen concentration on nitrification, Water Res., 1980, 14, 643–649 CrossRef CAS.
- D. S. Parker, Process Design Manual for Nitrogen Control, U.S. Environmental Protection Agency, Technology Transfer, Washington, DC, USA, 1975 Search PubMed.
- C. P. L. Grady, G. T. Daigger and N. G. Love, Biological Wastewater Treatment, IWA Publishing, London, UK, 2011 Search PubMed.
- Recommended standards for wastewater facilities, Albany, N.Y, 2014 Search PubMed.
- O. K. Scheible, M. Mulbarger, P. Sutton, T. Simpkin, G. Daigger, J. Heidman, M. Yoder, D. Schwinn and D. Storrier, Process design manual: Nitrogen control, U.S. Environmental Protection Agency, Office of Research and Development, Center for Environmental Research Information, Risk Reduction Engineering Laboratory; Office of Water, Office of Wastewater Enforcement and Compliance, Washington, DC, USA, EPA/625/R-93/010, 1993 Search PubMed.
- C. P. L. Grady, B. F. Smets and D. S. Barbeau, Variability in kinetic parameter estimates: A review of possible causes and a proposed terminology, Water Res., 1996, 30, 742–748 CrossRef CAS.
- M. Arnaldos, Y. Amerlinck, U. Rehman, T. Maere, S. Van Hoey, W. Naessens and I. Nopens, From the affinity constant to the half-saturation index: Understanding conventional modeling concepts in novel wastewater treatment processes, Water Res., 2015, 70, 458–470 CrossRef CAS PubMed.
- A. Shaw, I. Takács, K. R. Pagilla and S. Murthy, A new approach to assess the dependency of extant half-saturation coefficients on maximum process rates and estimate intrinsic coefficients, Water Res., 2013, 47, 5986–5994 CrossRef CAS PubMed.
- I. B. Petersen, PhD Thesis, Ghent University, Belgium, 2000 Search PubMed.
- R. Blackburne, Z. Yuan and J. Keller, Partial nitrification to nitrite using low dissolved oxygen concentration as the main selection factor, Biodegradation, 2008, 19, 303–312 CrossRef CAS PubMed.
- Y. Law, A. Matysik, X. Chen, S. Swa Thi, T. Q. Ngoc Nguyen, G. Qiu, G. Natarajan, R. B. H. Williams, B.-J. Ni, T. W. Seviour and S. Wuertz, High dissolved oxygen selection against Nitrospira sublineage in full-scale activated sludge, Environ. Sci. Technol., 2019, 53, 8157–8166 CrossRef CAS PubMed.
- J. Wen, M. W. LeChevallier and W. Tao, Nitrification kinetics and microbial communities of activated sludge as a full-scale membrane bioreactor plant transitioned to low dissolved oxygen operation, J. Cleaner Prod., 2020, 252, 119872 CrossRef CAS.
- T. J. Goreau, W. A. Kaplan, S. C. Wofsy, M. B. McElroy, F. W. Valois and S. W. Watson, Production of NO2- and N2O by nitrifying bacteria at reduced concentrations of oxygen, Appl. Environ. Microbiol., 1980, 40, 526–532 CrossRef CAS PubMed.
- R. A. Kester, W. De Boer and H. J. Laanbroek, Production of NO and N2O by pure cultures of nitrifying and denitrifying bacteria during changes in aeration, Appl. Environ. Microbiol., 1997, 63, 3872–3877 CrossRef CAS PubMed.
- G. Tallec, J. Garnier, G. Billen and M. Gousailles, Nitrous oxide emissions from denitrifying activated sludge of urban wastewater treatment plants, under anoxia and low oxygenation, Bioresour. Technol., 2008, 99, 2200–2209 CrossRef CAS PubMed.
- J. Jimenez, K. Bauhs, M. Miller, P. Dold, A. Al-Omari, M. Garrido, D. Hiripitiyage, M. Wittman, G. Burger, K. Chandran and B. Sturm, Low dissolved oxygen nitrification through kinetic selection, Water Res., 2025, 124642 Search PubMed.
- K. Hanaki, C. Wantawin and S. Ohgaki, Nitrification at low levels of dissolved oxygen with and without organic loading in a suspended-growth reactor, Water Res., 1990, 24, 297–302 CrossRef CAS.
- H. J. Laanbroek and S. Gerards, Competition for limiting amounts of oxygen between Nitrosomonas europaea and Nitrobacter winogradskyi grown in mixed continuous cultures, Arch. Microbiol., 1993, 159, 453–459 CrossRef CAS.
- H.-D. Park, L.-M. Whang, S. R. Reusser and D. R. Noguera, Taking advantage of aerated-anoxic operation in a full-scale University of Cape Town process, Water Environ. Res., 2006, 78, 637–642 CrossRef CAS PubMed.
- H. Daebel, R. Manser and W. Gujer, Exploring temporal variations of oxygen saturation constants of nitrifying bacteria, Water Res., 2007, 41, 1094–1102 CrossRef CAS PubMed.
- G. Liu and J. Wang, Quantifying the chronic effect of low DO on the nitrification process, Chemosphere, 2015, 141, 19–25 CrossRef CAS PubMed.
- H. Fan, L. Qi, G. Liu, Y. Zhang, Q. Fan and H. Wang, Aeration optimization through operation at low dissolved oxygen concentrations: Evaluation of oxygen mass transfer dynamics in different activated sludge systems, J. Environ. Sci., 2017, 55, 224–235 Search PubMed.
- L. Wang, B. Li, Y. Li and J. Wang, Enhanced biological nitrogen removal under low dissolved oxygen in an anaerobic-anoxic-oxic system: Kinetics, stoichiometry and microbial community, Chemosphere, 2021, 263, 128184 CrossRef CAS PubMed.
- S. Winogradsky, Recherches sur les organismes de la nitrification, Ann. Inst. Pasteur, 1890, 4, 213–331 Search PubMed.
- H.-D. Park and D. R. Noguera, Evaluating the effect of dissolved oxygen on ammonia-oxidizing bacterial communities in activated sludge, Water Res., 2004, 38, 3275–3286 Search PubMed.
- H.-D. Park and D. R. Noguera, Characterization of two ammonia-oxidizing bacteria isolated from reactors operated with low dissolved oxygen concentrations, J. Appl. Microbiol., 2007, 102, 1401–1417 Search PubMed.
- M. Bellucci, I. D. Ofiţeru, D. W. Graham, I. M. Head and T. P. Curtis, Low-dissolved-oxygen nitrifying systems exploit ammonia-oxidizing bacteria with unusually high yields, Appl. Environ. Microbiol., 2011, 77, 7787–7796 CrossRef CAS PubMed.
- M. J. L. Coolen, B. Abbas, J. van Bleijswijk, E. C. Hopmans, M. M. M. Kuypers, S. G. Wakeham and J. S. Sinninghe Damsté, Putative ammonia-oxidizing Crenarchaeota in suboxic waters of the Black Sea: A basin-wide ecological study using 16S ribosomal and functional genes and membrane lipids, Environ. Microbiol., 2007, 9, 1001–1016 CrossRef CAS PubMed.
- C. A. Francis, K. J. Roberts, J. M. Beman, A. E. Santoro and B. B. Oakley, Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean, Proc. Natl. Acad. Sci. U. S. A., 2005, 102, 14683–14688 CrossRef CAS PubMed.
- T. Limpiyakorn, Y. Shinohara, F. Kurisu and O. Yagi, Communities of ammonia-oxidizing bacteria in activated sludge of various sewage treatment plants in Tokyo, FEMS Microbiol. Ecol., 2005, 54, 205–217 CrossRef CAS PubMed.
- T. Zhang, T. Jin, Q. Yan, M. Shao, G. Wells, C. Criddle and H. H. P. Fang, Occurrence of ammonia-oxidizing Archaea in activated sludges of a laboratory scale reactor and two wastewater treatment plants, J. Appl. Microbiol., 2009, 107, 970–977 CrossRef CAS PubMed.
- N. Sinthusith, A. Terada, M. Hahn, P. (Lek) Noophan, J. Munakata-Marr and L. A. Figueroa, Identification and quantification of bacteria and archaea responsible for ammonia oxidation in different activated sludge of full-scale wastewater treatment plants, J. Environ. Sci. Health, Part A: Toxic/Hazard. Subst. Environ. Eng., 2015, 50, 169–175 CrossRef CAS PubMed.
- W. Martens-Habbena, P. M. Berube, H. Urakawa, J. R. de la Torre and D. A. Stahl, Ammonia oxidation kinetics determine niche separation of nitrifying Archaea and Bacteria, Nature, 2009, 461, 976–979 CrossRef CAS PubMed.
- B.-J. Park, S.-J. Park, D.-N. Yoon, S. Schouten, J. S. S. Damsté and S.-K. Rhee, Cultivation of autotrophic ammonia-oxidizing archaea from marine sediments in coculture with sulfur-oxidizing bacteria, Appl. Environ. Microbiol., 2010, 76, 7575–7587 CrossRef CAS PubMed.
- W. Qin, K. A. Meinhardt, J. W. Moffett, A. H. Devol, E. V. Armbrust, A. E. Ingalls and D. A. Stahl, Influence of oxygen availability on the activities of ammonia-oxidizing archaea, Environ. Microbiol. Rep., 2017, 9, 250–256 CrossRef CAS PubMed.
- L. L. Straka, K. A. Meinhardt, A. Bollmann, D. A. Stahl and M.-K. H. Winkler, Affinity informs environmental cooperation between ammonia-oxidizing archaea (AOA) and anaerobic ammonia-oxidizing (Anammox) bacteria, ISME J., 2019, 13, 1997–2004 CrossRef CAS PubMed.
- J. Xie, J. Yan, H. He, D. Lin, Y. Chen, Y. Li, X. Huang and H. Zhang, Evaluation of the key factors to dominate aerobic ammonia-oxidizing archaea in wastewater treatment plant, Int. Biodeterior. Biodegrad., 2021, 164, 105289 CrossRef CAS.
- H.-D. Park, G. F. Wells, H. Bae, C. S. Criddle and C. A. Francis, Occurrence of ammonia-oxidizing archaea in wastewater treatment plant bioreactors, Appl. Environ. Microbiol., 2006, 72, 5643–5647 CrossRef CAS PubMed.
- G. F. Wells, H.-D. Park, C.-H. Yeung, B. Eggleston, C. A. Francis and C. S. Criddle, Ammonia-oxidizing communities in a highly aerated full-scale activated sludge bioreactor: betaproteobacterial dynamics and low relative abundance of Crenarchaea, Environ. Microbiol., 2009, 11, 2310–2328 CrossRef CAS PubMed.
- H.-D. Park, J. M. Regan and D. R. Noguera, Molecular analysis of ammonia-oxidizing bacterial populations in aerated-anoxic orbal processes, Water Sci. Technol., 2002, 46, 273–280 CrossRef CAS PubMed.
- E. Giraldo, P. Jjemba, Y. Liu and S. Muthukrishnan, Ammonia oxidizing archaea, AOA, population and kinetic changes in a full scale simultaneous nitrogen and phosphorous removal mbr, Proc. Water Environ. Fed., 2011, 2011, 3156–3168 CrossRef.
- G. Liu, X. Wu, D. Li, L. Jiang, J. Huang and L. Zhuang, Long-term low dissolved oxygen operation decreases N2O emissions in the activated sludge process, Environ. Sci. Technol., 2021, 55, 6975–6983 CrossRef CAS PubMed.
- G. Liu and J. Wang, Long-term low DO enriches and shifts nitrifier community in activated sludge, Environ. Sci. Technol., 2013, 47, 5109–5117 CrossRef CAS PubMed.
- M. Arnaldos, S. A. Kunkel, B. C. Stark and K. R. Pagilla, Enhanced heme protein expression by ammonia-oxidizing communities acclimated to low dissolved oxygen conditions, Appl. Microbiol. Biotechnol., 2013, 97, 10211–10221 CrossRef CAS PubMed.
- C. M. Fitzgerald, P. Camejo, J. Z. Oshlag and D. R. Noguera, Ammonia-oxidizing microbial communities in reactors with efficient nitrification at low-dissolved oxygen, Water Res., 2015, 70, 38–51 CrossRef CAS PubMed.
- H. Daims, E. V. Lebedeva, P. Pjevac, P. Han, C. Herbold, M. Albertsen, N. Jehmlich, M. Palatinszky, J. Vierheilig, A. Bulaev, R. H. Kirkegaard, M. von Bergen, T. Rattei, B. Bendinger, P. H. Nielsen and M. Wagner, Complete nitrification by Nitrospira bacteria, Nature, 2015, 528, 504–509 CrossRef CAS PubMed.
- M. A. H. J. van Kessel, D. R. Speth, M. Albertsen, P. H. Nielsen, H. J. M. Op den Camp, B. Kartal, M. S. M. Jetten and S. Lücker, Complete nitrification by a single microorganism, Nature, 2015, 528, 555–559 CrossRef CAS PubMed.
- F. Sabba, A. Terada, G. Wells, B. F. Smets and R. Nerenberg, Nitrous oxide emissions from biofilm processes for wastewater treatment, Appl. Microbiol. Biotechnol., 2018, 102, 9815–9829 CrossRef CAS PubMed.
- M. K. Annavajhala, V. Kapoor, J. Santo-Domingo and K. Chandran, Comammox functionality identified in diverse engineered biological wastewater treatment systems, Environ. Sci. Technol. Lett., 2018, 5, 110–116 CrossRef CAS PubMed.
- P. Y. Camejo, J. S. Domingo, K. D. McMahon and D. R. Noguera, Genome-enabled insights into the ecophysiology of the comammox bacterium “Candidatus Nitrospira nitrosa”, mSystems, 2017, e00059-17 CrossRef PubMed.
- I. Cotto, Z. Dai, L. Huo, C. L. Anderson, K. J. Vilardi, U. Ijaz, W. Khunjar, C. Wilson, H. De Clippeleir, K. Gilmore, E. Bailey and A. J. Pinto, Long solids retention times and attached growth phase favor prevalence of comammox bacteria in nitrogen removal systems, Water Res., 2020, 169, 115268 CrossRef CAS PubMed.
- S. W. How, A. S. M. Chua, G. C. Ngoh, T. Nittami and T. P. Curtis, Enhanced nitrogen removal in an anoxic-oxic-anoxic process treating low COD/N tropical wastewater: Low-dissolved oxygen nitrification and utilization of slowly-biodegradable COD for denitrification, Sci. Total Environ., 2019, 693, 133526 Search PubMed.
- N. R. Maddela, Z. Gan, Y. Meng, F. Fan and F. Meng, Occurrence and roles of comammox bacteria in water and wastewater treatment systems: A critical review, Engineering, 2022, 17, 196–206 CrossRef CAS.
- F. Sabba, E. Redmond, C. Ruff, M. Ramirez, M. Young, M. J. Song, S. Yoon and L. Downing, Exploring community and kinetic shifts in nitrifying microbial communities in low dissolved oxygen activated sludge facilities for energy-efficient biological nitrogen removal, ACS ES&T Water, 2024, 4, 707–718 Search PubMed.
- A. J. Pinto, D. N. Marcus, U. Z. Ijaz, Q. M. Bautista-de lose Santos, G. J. Dick and L. Raskin, Metagenomic evidence for the presence of comammox Nitrospira-like bacteria in a drinking water system, mSphere, 2015, e00054-15 Search PubMed.
- K. D. Kits, C. J. Sedlacek, E. V. Lebedeva, P. Han, A. Bulaev, P. Pjevac, A. Daebeler, S. Romano, M. Albertsen, L. Y. Stein, H. Daims and M. Wagner, Kinetic analysis of a complete nitrifier reveals an oligotrophic lifestyle, Nature, 2017, 549, 269–272 CrossRef CAS PubMed.
- J. Hou, Y. Zhu, Y. Shi, L. Lin, F. Meng, M. Xu, L. Yang, B.-J. Ni and X. Chen, Enrichment and Kinetic Profiling of Candidatus Nitrospira nitrosa Culture Reveal Mechanisms Underlying Its Prevalence in Wastewater Treatment Systems, Environ. Sci. Technol., 2025, 59, 15272–15281 CrossRef CAS PubMed.
- Y. Zhu, J. Hou, F. Meng, M. Xu, L. Lin, L. Yang and X. Chen, Comparative enrichment of complete ammonium oxidation bacteria in floccular sludge reactors: Sequencing batch reactor vs. continuous stirred tank reactor, Water Res.:X, 2025, 27, 100305 Search PubMed.
- P. Roots, Y. Wang, A. F. Rosenthal, J. S. Griffin, F. Sabba, M. Petrovich, F. Yang, J. A. Kozak, H. Zhang and G. F. Wells, Comammox Nitrospira are the dominant ammonia oxidizers in a mainstream low dissolved oxygen nitrification reactor, Water Res., 2019, 157, 396–405 CrossRef CAS PubMed.
- N. K. Beach and D. R. Noguera, Design and assessment of species-level qPCR primers targeting comammox, Front. Microbiol., 2019, 10, 36 Search PubMed.
- D. Li, F. Fang and G. Liu, Efficient nitrification and low-level N2O emission in a weakly acidic bioreactor at low dissolved-oxygen levels are due to comammox, Appl. Environ. Microbiol., 2021, 87, e00154-21 CrossRef PubMed.
- M. Zheng, G. Mu, A. Zhang, J. Wang, F. Chang, J. Niu, X. Wang, T. Gao and Z. Zhao, Predominance of comammox bacteria among ammonia oxidizers under low dissolved oxygen condition, Chemosphere, 2022, 308, 136436 CrossRef CAS PubMed.
- N. A. Keene, S. R. Reusser, M. J. Scarborough, A. L. Grooms, M. Seib, J. Santo Domingo and D. R. Noguera, Pilot plant demonstration of stable and efficient high rate biological nutrient removal with low dissolved oxygen conditions, Water Res., 2017, 121, 72–85 Search PubMed.
- M. Beccari, M. Franzini and R. Ramadori, in Proc. Int. Workshop on Advanced Biological Processes, National Research Council Rome, Italy, 1985, pp. 28–30 Search PubMed.
- M. Beccari, A. C. D. Pinto, R. Ramadori and M. C. Tomei, Effects of dissolved oxygen and diffusion resistances on nitrification kinetics, Water Res., 1992, 26, 1099–1104 CrossRef CAS.
- K. H. Chu, H. M. van Veldhuizen and M. C. M. van Loosdrecht, Respirometric measurement of kinetic parameters: Effect of activated sludge floc size, Water Sci. Technol., 2003, 48, 61–68 CrossRef CAS PubMed.
- R. Manser, W. Gujer and H. Siegrist, Consequences of mass transfer effects on the kinetics of nitrifiers, Water Res., 2005, 39, 4633–4642 CrossRef CAS PubMed.
- G. T. Daigger, C. D. Adams and H. K. Steller, Diffusion of oxygen through activated sludge flocs: Experimental measurement, modeling, and implications for simultaneous nitrification and denitrification, Water Environ. Res., 2007, 79, 375–387 CrossRef CAS PubMed.
- J. Wu, Y. Li and M. Zhang, Activated sludge floc morphology and nitrifier enrichment can explain the conflicting reports on the oxygen half-saturation index for ammonium oxidizing bacteria (AOB) and nitrite oxidizing bacteria (NOB), J. Chem. Technol. Biotechnol., 2017, 92, 2673–2682 Search PubMed.
- L. Yu, S. Chen, W. Chen and J. Wu, Experimental investigation and mathematical modeling of the competition among the fast-growing “r-strategists” and the slow-growing “K-strategists” ammonium-oxidizing bacteria and nitrite-oxidizing bacteria in nitrification, Sci. Total Environ., 2020, 702, 135049 Search PubMed.
- S. M. Hocaoglu, G. Insel, E. U. Cokgor and D. Orhon, Effect of low dissolved oxygen on simultaneous nitrification and denitrification in a membrane bioreactor treating black water, Bioresour. Technol., 2011, 102, 4333–4340 CrossRef CAS PubMed.
- Z. Li and M. K. Stenstrom, Impacts of SRT on particle size distribution and reactor performance in activated sludge processes, Water Environ. Res., 2018, 90, 48–56 CrossRef CAS PubMed.
- R. Manser, W. Gujer and H. Siegrist, Membrane bioreactor versus conventional activated sludge system: Population dynamics of nitrifiers, Water Sci. Technol., 2005, 52, 417–425 CrossRef CAS PubMed.
- C. Picioreanu, J. Pérez and M. C. M. van Loosdrecht, Impact of cell cluster size on apparent half-saturation coefficients for oxygen in nitrifying sludge and biofilms, Water Res., 2016, 106, 371–382 CrossRef CAS PubMed.
- M. Bachmann, C. Lawrence, N. Wieczorek, T. Scott, E. Shelton, B. Elliott, M. Parsons, S. Klaus and C. Bott, Full-scale implementation of partial denitrification-anammox in IFAS processes: Cost savings and operational strategies, Water Environ. Res., 2025, 97, e70093 Search PubMed.
- R. Fofana, M. Parsons, C. Long, K. Chandran, K. Jones, S. Klaus, B. Trovato, C. Wilson, H. De Clippeleir and C. Bott, Full-scale transition from denitrification to partial denitrification–anammox (PdNA) in deep-bed filters: Operational strategies for and benefits of PdNA implementation, Water Environ. Res., 2022, 94, e10727 Search PubMed.
- K. McCullough, S. Klaus, M. Parsons, C. Wilson and C. B. Bott, Advancing the understanding of mainstream shortcut nitrogen removal: Resource efficiency, carbon redirection, and plant capacity, Environ. Sci.: Water Res. Technol., 2022, 8, 2398–2410 RSC.
- R. D. Megee, J. F. Drake, A. G. Fredrickson and H. M. Tsuchiya, Studies in intermicrobial symbiosis. Saccharomyces cerevisiae and Lactobacillus casei, Can. J. Microbiol., 1972, 18, 1733–1742 CrossRef PubMed.
- F. G. Bader, Analysis of double-substrate limited growth, Biotechnol. Bioeng., 1978, 20, 183–202 CrossRef CAS PubMed.
- H. R. Bungay, Growth rate expressions for two substrates one of which is inhibitory, J. Biotechnol., 1994, 34, 97–100 CrossRef CAS.
- T. Mankad and H. R. Bungay, Model for microbial growth with more than one limiting nutrient, J. Biotechnol., 1988, 7, 161–166 CrossRef CAS.
- H. A. Stewart, A. Al-Omari, C. Bott, H. De Clippeleir, C. Su, I. Takacs, B. Wett, A. Massoudieh and S. Murthy, Dual substrate limitation modeling and implications for mainstream deammonification, Water Res., 2017, 116, 95–105 CrossRef CAS PubMed.
- J. Geets, N. Boon and W. Verstraete, Strategies of aerobic ammonia-oxidizing bacteria for coping with nutrient and oxygen fluctuations, FEMS Microbiol. Ecol., 2006, 58, 1–13 CrossRef CAS PubMed.
- J. H. Ahn, R. Yu and K. Chandran, Distinctive microbial ecology and biokinetics of autotrophic ammonia and nitrite oxidation in a partial nitrification bioreactor, Biotechnol. Bioeng., 2008, 100, 1078–1087 CrossRef CAS.
- T. Limpiyakorn, M. Fürhacker, R. Haberl, T. Chodanon, P. Srithep and P. Sonthiphand, amoA-encoding archaea in wastewater treatment plants: A review, Appl. Microbiol. Biotechnol., 2013, 97, 1425–1439 CrossRef CAS PubMed.
- T. Limpiyakorn, P. Sonthiphand, C. Rongsayamanont and C. Polprasert, Abundance of amoA genes of ammonia-oxidizing archaea and bacteria in activated sludge of full-scale wastewater treatment plants, Bioresour. Technol., 2011, 102, 3694–3701 Search PubMed.
- R. Manser, W. Gujer and H. Siegrist, Decay processes of nitrifying bacteria in biological wastewater treatment systems, Water Res., 2006, 40, 2416–2426 CrossRef CAS PubMed.
- H. Siegrist, I. Brunner, G. Koch and L. C. Phan, Reduction of biomass decay rate under anoxic and anaerobic conditions, Water Sci. Technol., 1999, 39, 129–137 CrossRef CAS.
- E. J. T. M. Leenen, A. A. Boogert, A. A. M. van Lammeren, J. Tramper and R. H. Wijffels, Dynamics of artificially immobilized Nitrosomonas europaea: Effect of biomass decay, Biotechnol. Bioeng., 1997, 55, 630–641 CrossRef CAS.
- M. Henze, W. Gujer, T. Mino and M. C. M. van Loosdrecht, Activated Sludge Models ASM1, ASM2, ASM2d and ASM3, IWA Publishing, London, UK, 2000 Search PubMed.
- G. Munz, C. Lubello and J. A. Oleszkiewicz, Modeling the decay of ammonium oxidizing bacteria, Water Res., 2011, 45, 557–564 CrossRef CAS PubMed.
- O. Nowak, K. Svardal and P. Schweighofer, The dynamic behaviour of nitrifying activated sludge systems influenced by inhibiting wastewater compounds, Water Sci. Technol., 1995, 31, 115–124 CrossRef CAS.
- M.-J. Mehrani, X. Lu, P. Kowal, D. Sobotka and J. Mąkinia, Incorporation of the complete ammonia oxidation (comammox) process for modeling nitrification in suspended growth wastewater treatment systems, J. Environ. Manage., 2021, 297, 113223 CrossRef CAS PubMed.
- B. Petersen, K. V. Gernaey, M. Henze and P. A. Vanrolleghem, in Biotechnology for the Environment: Wastewater Treatment and Modeling, Waste Gas Handling, ed. S. N. Agathos and W. Reineke, Kluwer Academic Publishers, Dordrecht, The Netherlands, 2003, pp. 101–186 Search PubMed.
- Methods for wastewater characterization in activated sludge modeling, ed. H. Melcer, P. Dold, R. M. Jones, C. M. Bye, I. Takacs, H. D. Stensel, A. W. Wilson, P. Sun and S. Bury, Water Environment Federation; IWA Publishing, Alexandria, VA: London, U.K., 2003 Search PubMed.
- A. Pruden, J. H. Miller, I. Takacs and M. Higgins, Next generation tools for assessing death and decay of critical wastewater bacteria, Water Intell. Online, 2016, 15, 9781780408194 CrossRef.
- E. French and A. Bollmann, Freshwater ammonia-oxidizing archaea retain amoA mRNA and 16S rRNA during ammonia starvation, Life, 2015, 5, 1396–1404 Search PubMed.
- J. Johnston, Z. Du and S. Behrens, Ammonia-oxidizing bacteria maintain abundance but lower amoA-gene expression during cold temperature nitrification failure in a full-scale municipal wastewater treatment plant, Microbiol. Spectrum, 2023, 11, e02571-22 Search PubMed.
- M. Friedrich, J. Jimenez, A. Pruden, J. H. Miller, J. Metch and I. Takács, Rethinking growth and decay kinetics in activated sludge – towards a new adaptive kinetics approach, Water Sci. Technol., 2016, 75, 501–506 CrossRef PubMed.
- P. Blom, G. J. Smith, M. A. H. J. Van Kessel, H. Koch and S. Lücker, Comprehensive evaluation of primer pairs targeting the ammonia monooxygenase subunit A gene of complete ammonia-oxidizing Nitrospira, Microbiol. Spectrum, 2024, 12, e00516–e00524 Search PubMed.
- R. Jiang, J. Wang, T. Zhu, B. Zou, D. Wang, S. Rhee, D. An, Z. Ji and Z. Quan, Use of newly designed primers for quantification of complete ammonia-oxidizing (comammox) bacterial clades and strict nitrite oxidizers in the genus nitrospira, Appl. Environ. Microbiol., 2020, 86, e01775-20 Search PubMed.
- F. Xia, J. Wang, T. Zhu, B. Zou, S. Rhee and Z. Quan, Ubiquity and diversity of complete ammonia oxidizers (comammox), Appl. Environ. Microbiol., 2018, 84, e01390-18 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |