 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Antibiotic toxicity screening on seedling emergence: beyond traditional species
R. Silvana
Cortés-Lagunes
 a,
Andrea-Lorena
Garduño-Jiménez
a,
Andrea-Lorena
Garduño-Jiménez
 b,
Wenshi
Bao
c,
Juan Carlos
Durán-Álvarez
b,
Wenshi
Bao
c,
Juan Carlos
Durán-Álvarez
 a,
Felicity C. T.
Elder
a,
Felicity C. T.
Elder
 b,
Joshua
Greenwood
b,
John H.
Nightingale
b,
Fan
Zhang
c and
Laura J.
Carter
b,
Joshua
Greenwood
b,
John H.
Nightingale
b,
Fan
Zhang
c and
Laura J.
Carter
 *b
*b
aInstituto de Ciencias Aplicadas y Tecnología, Universidad Nacional Autónoma de México, Circuito Exterior S/N, Ciudad Universitaria, 04510 Mexico City, Mexico
bSchool of Geography and Water@Leeds, Faculty of Environment, University of Leeds, LS2 9JT, UK
cCollege of Science, Nanjing Agricultural University, Nanjing 210085, China
First published on 2nd March 2026
Abstract
Antibiotics are prevalent environmental pollutants with documented plant uptake and effects. Germination and seedling emergence are critical stages of plant development, making toxicity tests valuable for assessing the terrestrial risk. This study aims to address the need to understand species and antibiotic-dependent effects of 10 antibiotics across 23 plant species, including non-cultivated and geographically diverse species, through a Tier I phytotoxicity screening in which all antibiotics were tested at a single nominal concentration of 1 mg L−1, an upper-bound environmentally relevant scenario. Results revealed that antibiotic toxicity is highly species and antibiotic-dependent, highlighting the need to evaluate effects on under-studied species. Some global trends were observed; fluoroquinolones (ciprofloxacin, enrofloxacin, ofloxacin) stimulated germination and root growth in legumes and grasses, while black knapweed (Centaurea nigra) consistently exhibits germination reductions (30–53%) and root growth inhibition under all antibiotic exposures. Florfenicol exposure decreased root length and biomass in Chinese cabbage (Brassica rapa subs. Pekinensis) by nearly 100%, contrasting with stimulation observed in rice (Oryza sativa). The importance of assessing sublethal effects, as root and biomass changes, in addition to germination, for a more comprehensive phytotoxicity assessment is demonstrated. Standardized test conditions may overlook species requiring specific germination conditions. Sorption of antibiotics to filter paper affected exposure concentrations, emphasizing the importance of chemical quantification before and after tests. This study highlights the need for adaptive phytotoxicity protocols, careful experimental design to obtain statistically significant results, and inclusion of non-cultivated species as bioindicators to better assess antibiotic risks to terrestrial plants.
Environmental significanceAntibiotics are increasingly recognized as emerging environmental pollutants due to their widespread occurrence and persistence in agricultural systems. This study provides a comprehensive cross-country Tier I screening, evaluating the phytotoxic effects of ten antibiotics across 23 cultivated and non-cultivated plant species from China, the UK, and Mexico. The results revealed that antibiotic toxicity is both species- and antibiotic-dependent, showing variable effects on germination, root elongation, and biomass. This study identifies, for the first time, sensitive non-cultivated species as potential bioindicators for environmental monitoring, like black knapweed. By highlighting species-specific responses and limitations of current standard testing protocols, these findings contribute to improving environmental risk assessment frameworks and promoting more representative and ecologically relevant methods for evaluating impacts of antibiotics on terrestrial ecosystems. |
Introduction
Antibiotics are extensively used worldwide in human and veterinary medicine1 and are routinely applied in agricultural systems for disease prevention and growth promotion.2,3 As these pharmaceuticals are not fully absorbed by humans or animals, a substantial fraction of the consumed dose, and their intermediates, are excreted in faeces or urine,4 leading to their introduction into soils via wastewater irrigation and the land application of wastewater-derived biosolids, animal manures, and slurries.5 Antibiotic concentrations in agricultural soils have been reported to range from the nanogram to microgram per gram range, with higher levels typically associated with soils receiving livestock-derived amendments.6,7 Sulfonamides and tetracyclines are among the most frequently detected classes at elevated concentrations,6,7 although marked regional variability has been observed depending on soil properties and amendment practices. For example, soils amended with chicken manure in China contained higher fluoroquinolone concentrations than sulfonamides or tetracyclines, with ciprofloxacin levels reaching up to 18.42 µg kg−1,8 whereas soils irrigated with raw wastewater in Cameroon presented substantially lower ciprofloxacin concentrations (0.44–2.33 ng g−1), along with sulfamethoxazole, trimethoprim, enrofloxacin, and oxytetracycline at similar or lower levels.9 Information remains comparatively scarce for other regions, including Latin America. In the Tula Valley (Mexico), where raw wastewater has been used for agricultural irrigation for over a century,10 antibiotic concentrations in irrigation water have been reported between 337.9 ng L−1 (oxytetracycline) and 9059.8 ng L−1 (erythromycin),11 while corresponding soil measurements are limited, with triclosan reported at 7.7 ng g−1 (ref. 12).Once present in soils, antibiotics can be taken up by plants to varying extents depending on the exposure scenario.5,13,14 Studies have reported antimicrobial residues in irrigation water, soils, and crops, although accumulation in plant tissues varies widely. For example, antimicrobials were detected in 85% of water and soil samples but at lower frequencies and concentrations in crops irrigated with reclaimed water in Israel,15 whereas sulfamethoxazole concentrations exceeding 30 mg kg−1 were reported in tomato (Solanum lycopersicum L.) following irrigation with treated wastewater in Spanish glasshouses.16 Similarly, a meta-analysis documented plant accumulation of tetracyclines and sulfonamides above 50 mg kg−1 in studies from China, Germany, and Spain following exposure to pig slurry and manure.17
Following plant uptake, antibiotics have been shown to exert phytotoxic effects, including alterations in germination, growth, and photosynthesis. These effects can arise through multiple pathways, such as disruption of electron transport, reductions in vascular bundle size and stomatal conductance, and disturbances to photosynthetic pigments and chloroplast structure.18 Three out of the four antibiotic biomolecular targets, intended for animals, are shared by plants, therefore antibiotics may easily interfere with plant biomolecular pathways.13 Phytotoxic responses have been reported to be antibiotic class-dependent. For instance, amoxicillin did not affect germination of lettuce (Lactuca sativa), alfalfa (Medicago sativa), or carrot (Daucus carota), whereas other antibiotics, such as sulfamethoxazole, induced effects under comparable conditions.19 Despite growing evidence of antibiotic occurrence in soils, plant uptake, and adverse effects, toxicity studies have predominantly focused on a limited number of fast-growing cultivated species, including spinach (Spinacia oleracea), lettuce (L. sativa), radish (Raphanus sativus), and thale cress (Arabidopsis thaliana),17,20,21 as well as major agricultural crops such as wheat (Triticum aestivum),21–23 rapeseed (Brassica campestris), maize (Zea mays)24 and rice (Oryza sativa L.).21,25 In contrast, far less is known about antibiotic effects on non-cultivated species and on agriculturally relevant species from understudied regions.13,18,26 Addressing this gap is critical, as antibiotic exposure in agricultural systems extends beyond cultivated crops, and effects on non-cultivated species may have cascading consequences for ecosystem functioning.5,13 Furthermore, current risk assessments typically consider a limited range of antibiotic classes17 and rarely evaluate comparable endpoints across multiple compounds, constraining our understanding of differential phytotoxic responses. Given that antibiotic use has increased globally since their introduction in the 1940 decade and is projected to continue rising in many regions,2,27,28 the generation of globally representative toxicity data across species and antibiotic classes remains essential for robust environmental risk assessment.
This study presents, for the first time, an international Tier I hazard-screening of antibiotic phytotoxicity during seed germination, designed to address key gaps in terrestrial plant risk assessment across species and regions. The screening focuses on non-cultivated plant species from the UK, selected based on Annex 3 of OECD Guideline 208 for non-crop terrestrial plant testing,29 alongside agriculturally and culturally relevant species from Mexico and China. Experiments were conducted in three laboratories using standardized protocols, enabling cross-country comparability within a collaborative international framework. In line with a Tier I screening objective, all antibiotics were tested individually at a single nominal concentration of 1 mg L−1,30,31 selected as an upper-bound environmentally relevant exposure scenario. Antibiotic concentrations in pore water of wastewater-irrigated or manure-amended soils have been reported in the high µg L−1 to mg L−1 range,32–34 supporting the relevance of this concentration for conservative screening. Tier I assays intentionally use a single, elevated concentration to identify potential species–antibiotic sensitivities and to prioritize compounds and taxa for further investigation, rather than to derive dose–response relationships, consistent with established screening frameworks, such as EPA Protocol 600/3-88/029A. Hydroponic filter-paper assay was used to expose germinating seeds directly to the dissolved, bioavailable fraction of each antibiotic, providing a functional analogue of pore-water exposure while minimizing confounding effects associated with soil sorption.30,35–37 In addition to biological endpoints, antibiotic concentrations were quantified before and after exposure using liquid chromatography-mass spectrometry, an approach not typically included in seedling toxicity assays, but critical for evaluating exposure dynamics and informing interpretation of screening results. Overall, this study provides a comparative, cross-country assessment of early-stage plant responses to prioritize antibiotics and species that warrant further investigation. In addition, it provides an assessment of existing toxicity guidelines, by applying them in a cross-country, multi-laboratory screening framework. Accordingly, the outcomes of this Tier I screening are intended to identify sensitive species–antibiotic combinations and to inform the design of subsequent Tier II studies, where full dose–response experiments will be required to characterize concentration–effect relationships and derive effect thresholds.
Experimental
Chemicals
For the Mexico experiments, the chemicals used were HPLC grade Methanol and Acetonitrile (≥99.9%, Sigma-Aldrich), ammonium acetate and formic acid (≥97.8 and ≥98% respectively, both from Sigma-Aldrich), and sterile water was from ultrapure water (18.6 MΩ cm, Elix 5 tandem with Synergy® UV High Flow from Millipore®), then filtered through 0.22 µm nitrocellulose membrane. In the UK, Methanol (99.9%, Fisher) and sterile water (ultrapure water 18.6 MΩ cm, Sartorius arium® comfort, EDI UV), which was then autoclaved in (LTE Touchclave-R autoclave at 126 °C for 15 min), were used. In China, methanol (≥99.5%, Aladdin) and sterile water (ultrapure water 18.6 MΩ cm, Shanghai Hitech Instruments Co., Ltd Hitech), which was then autoclaved in ZEALWAY G-85D), were used. In the SI, the details of the antibiotics used, the supplier, and the purity are provided (Table S1). Table 1 contains the supplier and species details of the seeds used for the germination tests. The filter papers used were 100 mm Camlab grade 122P [114V in the UK, 110 mm, Labshark Qualitative filter paper no. 130209069 in China, and 150 mm, Whatman Cat no 1002-147 in Mexico.| Family | Genus | Common name | Species (Latin name) | Use | Seed supplier |
|---|---|---|---|---|---|
| China | |||||
| Fabaceae | Glycine | Soybean | Glycine max | Food crop | Shandong Xuhong Seed Technology Co, Ltd, China |
| Poaceae | Triticum | Wheat | Triticum aestivum | Food crop | Zhoukou Academy of Agricultural Sciences, China |
| Fabaceae | Vigna | Mung bean | Vigna radiata | Food crop | Gengniu Seeds Business Dept., Xinhe Town, Shuyang, China |
| Asteraceae | Lactuca | Lettuce | Lactuca sativa | Vegetable crop | Shouhe Seed Co, Ltd, China |
| Poaceae | Oryza | Rice | Oryza sativa | Food crop | Jiangsu Academy of Agricultural Sciences, China |
| Brassicaceae | Brassica | Chinese cabbage | Brassica rapa subsp.Pekinensis | Vegetable crop | Guangzhou Yunong Seedling High-Tech Co, Ltd China |
| Asteraceae | Cichorium | Chicory | Cichorium intybus | Forage/medicinal | Suqian Zeshun Landscaping Co, Ltd, China |
| Asteraceae | Leucanthemum | Shasta daisy | Leucanthemum maximum | Ornamental | Shouguang Firefly Agricultural Technology Co, Ltd, China |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| UK | |||||
| Fabaceae | Phaseolus | French bean | Phaseolus vulgaris | Food crop | Suttons |
| Fabaceae | Pisum | Pea | Pisum sativum | Food crop | Suttons |
| Fabaceae | Trifolium | Wild white clover | Trifolium repens | Forage | Yellow Flag Wildflowers |
| Fabaceae | Trifolium | Red clover | Trifolium pratense | Forage | Yellow Flag Wildflowers |
| Poaceae | Avena | Oats | Avena sativa | Food/forage crop | — |
| Poaceae | Triticum | Wheat | Triticum aestivum | Food crop | — |
| Poaceae | Hordeum | Barley | Hordeum vulgare | Food/forage crop | KWS Feeris |
| Poaceae | Festuca | Sheep's fescue | Festuca ovina | Forage/turfgrass | Yellow Flag Wildflowers |
| Poaceae | Festuca | Red fescue | Festuca rubra | Forage/turfgrass | Yellow Flag Wildflowers |
| Poaceae | Briza | Quaking grass | Briza media | Ornamental/wild | Yellow Flag Wildflowers |
| Asteraceae | Lactuca | Lettuce | Lactuca sativa | Vegetable crop | Suttons |
| Asteraceae | Leucanthemum | Oxeye daisy | Leucanthemum vulgare | Ornamental/wild | Yellow Flag Wildflowers |
| Asteraceae | Centaurea | Black knapweed | Centaurea nigra | Wildflower | Yellow Flag Wildflowers |
| Asteraceae | Centaurea | Cornflower | Centaurea cyanus | Ornamental/wild | Yellow Flag Wildflowers |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| Mexico | |||||
| Solanaceae | Solanum | Tomato | Solanum lycopersicum | Food crop | Hortaflor |
| Asteraceae | Tagetes | Mexican marigold (cempasúchil) | Tagetes erecta | Ornamental/cultural | Hortaflor |
| Caryophyllaceae | Dianthus | Carnation | Dianthus caryophyllus | Ornamental | Hortaflor |
| Fabaceae | Medicago | Alfalfa | Medicago sativa | Forage crop | Hortaflor |
Seedling germination
Antibiotics were tested individually at a single nominal concentration of 1 mg L−1. In the UK and China, 5 mL of the working solution (0.1% solvent volume) was added per Petri dish, while in Mexico, 40 mL were used to accommodate larger dish formats. The same nominal concentration was applied across all antibiotics and species to enable direct comparative screening. Reported environmental concentrations of the studied antibiotics in irrigation water and soil pore-water, together with the concentration used in this study, are summarized in Table S3 to provide environmental context and illustrate that the selected test concentration represents a conservative upper-bound dissolved-phase exposure relevant for Tier I hazard screening. Treatment details by country are provided in Table S2, and species-specific information is given in Table 1. Control dishes were prepared identically, replacing antibiotics with the equivalent volume of methanol. An additional control without seeds was included in the Mexico experiments to assess potential antibiotic degradation and sorption onto filter paper.
Petri dishes were sealed with parafilm and incubated in the dark for 7 days at temperatures selected to maximise germination (UK: 18 °C; Mexico and China: 22 °C). Dark conditions were used to simulate below-ground germination environments and to minimise confounding light-dependent developmental responses. This approach allows germination and early seedling emergence to be assessed independently of photomorphogenic processes. Seed germination was monitored every weekday, and it was noted when they had germinated. Seeds were considered germinated when the length of plumules (shoots) and radicles (roots) reached at least 2 mm, which is a commonly used criterion in seed germination studies.38
Seedling dry biomass was measured at the end of the seven-day germination assay, in Mexico, considering each Petri dish as an individual experimental unit, and in China and the UK, considering the total of 30 seeds. The relative inhibition index (RCI, defined in eqn (1) is commonly used to quantify the degree of inhibition on plant growth based on dry biomass measurements, and was calculated as follows:39
 | (1) |
| ΔBiomassi = Yfinal,i − Yinitial,i | (2) |
Chromatographic separation was achieved on a Zorbax SB-C18 column (250 × 4.6 mm, 5 µm) maintained at 20 °C, using an isocratic mobile phase consisting of acetonitrile (20%), methanol (40%), and aqueous 0.1% formic acid with 10 mM ammonium acetate (40%) at a flow rate of 0.4 mL min−1. The injection volume was 10 µL. Electrospray ionization was operated in positive mode, with a drying gas temperature of 300 °C and flow rate of 11 L min−1, a nebulizer pressure of 15 psi, and a capillary voltage of 3000 V.
Oxytetracycline, trimethoprim, azithromycin, and ciprofloxacin were identified and quantified using the multiple reaction monitoring (MRM) mode (Table S4). Method validation details are provided in the SI (Table S5). The concentration of the antibiotic solution of each Petri dish was determined, including the controls without seeds. Changes in antibiotic concentration (ΔC%, eqn (3) and seed removal rates eqn (4) were calculated based on measured concentrations before and after exposure.
 | (3) |
 | (4) |
 | (5) |
To describe antibiotic partitioning between water and filter paper in control Petri dishes without seeds, a linear partitioning model was applied eqn (6).
| q = kdCeq | (6) |
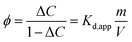 | (7) |
Results and discussion
Germination
Following exposure to antibiotics at 1 mg L−1, germination responses were evaluated for 10 antibiotics across 7 species in China, 10 antibiotics across 14 species in the UK, and 4 antibiotics across 4 species in Mexico (Table S6). Reported environmental concentrations of the target antibiotics in irrigation water and soil pore-water, together with the concentration applied in this study, are summarized in Table S3 to provide environmental context for this screening-level assessment. Replicate-level variability could only be quantified for the Mexico dataset (n = 3), where within-treatment standard deviations ranged from 5 to 20% (Fig. 1; Mexico study). Due to experimental constraints, replicate variability could not be assessed for the UK and China datasets.Given the Tier I hazard-screening nature of this study, a ± 20% difference in germination relative to the control was used as a descriptive indicator of biologically relevant change, rather than as a criterion for statistical significance, to facilitate comparative interpretation across datasets.43 This threshold was informed by the observed variability in the Mexico experiments and by reported coefficients of variation for seed germination in the literature, typically below 20%. For example, values in the range of 6.7 to 11.4% have been reported for M. sativa and L. sativa, respectively,19 while an average of 17.66% was reported across 20 Fabaceae species.44 Formal statistical inference was conducted exclusively for the Mexico dataset using Kruskal–Wallis tests followed by Dwass–Steel–Critchlow–Fligner (DSCF) post hoc comparisons, and these results are presented separately in Table 2
| Kruskal Wallis | DSCF Test | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| g. l. | χ 2 | p-Value | AZI | CPX | OTC | TRM | ||||||
| W | p-Value | W | p-Value | W | p-Value | W | p-Value | |||||
| Tomate (S. lycopersicum) | % Germination | 4 | 2.14 | 0.710 | 0.000 | 1.000 | 0.000 | 1.000 | 1.898 | 0.665 | 0.380 | 0.999 |
| Root length (mm) | 4 | 3.52 | 0.475 | −0.617 | 0.993 | 2.278 | 0.491 | 2.362 | 0.453 | 0.514 | 0.996 | |
| Cempasuchil (T. erecta) | % Germination | 4 | 7.05 | 0.133 | 1.89 | 0.669 | 1.311 | 0.887 | 1.554 | 0.807 | −2.070 | 0.586 |
| Root length (mm) | 4 | 13.9 | 0.007 | −0.199 | 1.000 | −3.486 | 0.099 | 1.792 | 0.712 | 1.705 | 0.748 | |
| Carnation (D. caryophyllus) | % Germination | 4 | 8.78 | 0.067 | 1.671 | 0.762 | 2.06 | 0.591 | −2.611 | 0.347 | 1.671 | 0.762 |
| Root length (mm) | 4 | 5.53 | 0.237 | 0.287 | 0.637 | 0.49 | 0.345 | −1.959 | 1.000 | −2.617 | 0.997 | |
| Alfalfa (M. sativa) | % Germination | 4 | 12.7 | 0.013 | 1.964 | 0.635 | −0.200 | 1.000 | −3.184 | 0.161 | −3.198 | 0.158 |
| Root length (mm) | 4 | 18.4 | 0.001 | 1.294 | 0.891 | 3.635 | 0.076 | −5.132 | 0.003 | −4.100 | 0.031 | |
For germination, Kruskal–Wallis tests indicated a significant overall treatment effect for alfalfa (Medicago sativa; p = 0.013; Table 2). However, DSCF post hoc comparisons did not identify statistically significant differences between individual antibiotic treatments and the control. This discrepancy reflects the limited resolution of pairwise comparisons under low replication (n = 3), a known constraint of non-parametric post hoc testing in screening-level studies. Accordingly, while global distributional differences were detected, individual treatment effects could not be resolved statistically, and germination responses were interpreted descriptively within the Tier I screening framework.
Using the ±20% descriptive threshold, antibiotic exposure was associated with increased germination relative to controls for several species–antibiotic combinations. Across datasets, germination stimulation was more frequently observed in legumes and grasses exposed to fluoroquinolones (ciprofloxacin, enrofloxacin, and ofloxacin) (Table 2 and Fig. 2). These responses were identified descriptively within the Tier I screening framework and highlight species–compound combinations that merit further investigation. Previous studies have reported similar stimulatory responses in crops exposed to low concentrations of fluoroquinolones, including maize, rice, and alfalfa, where enhanced germination and root elongation have been discussed in the context of hormetic effects.45–48 Such responses have been linked to stress-related biochemical pathways, including reactive oxygen species (ROS) signalling and the activation of antioxidant defence mechanisms.35,49,50 However, it is important to note that the mechanistic relationship between fluoroquinolone modes of action and germination-associated biochemical pathways remains poorly understood, and direct causal links have not been established.
Additional species–antibiotic combinations exhibiting > 20% increases in germination included carnation (D. caryophyllus) under trimethoprim exposure, which did not reach statistical significance in post-hoc testing but is interpreted here as a descriptive trend, and in French bean (Phaseolus vulgaris), where 33.4% and 53.4% more germination was observed under ciprofloxacin and enrofloxacin exposure, respectively, compared to the control (for which only 3.3% germination was observed). Similarly, pea (P. sativum), germination increased under ciprofloxacin, enrofloxacin, florfenicol, and sulfonamide exposure, ranging between 23 and 50%. Oats (A. sativa), showed up to 40% increase under florfenicol exposure, and red clover (T. pratense) exhibited 20% increases under azithromycin, sulfonamide, and trimethoprim exposure, suggesting hormetic effects, consistent with previous findings in crops, such as maize and alfalfa.45,46,48 These phenomena may involve biochemical pathways related to ROS signalling and antioxidant responses, warranting further investigation to assess ecological impacts and antibiotic–plant interactions.
For soybean (G. max; Fig. 2a and Table S6), a possible increase in germination exceeding 20% was observed under oxytetracycline exposure, compared to the control (90.0 vs. 66.7%). In contrast, for black knapweed (C. nigra), near-complete inhibition of germination was detected following oxytetracycline exposure (6.7 vs. 53.3% in the control; Fig. 2g), and for alfalfa (M. sativa), no statistically significant effect on germination (p = 0.161) was observed between the control and oxytetracycline treatment (Fig. 1d). Previous studies report no significant effects of oxytetracycline on wheat (Triticum aestivum) germination in soil at concentrations of 1, 10, and 50 mg kg−1,51 whereas significant inhibition has been observed under hydroponic conditions for the same species at higher concentrations (50 and 150 mg L−1).52 These discrepancies between studies and with the current results likely reflect differences in experimental setup (soil versus hydroponic exposure), species-specific sensitivity, and inherent challenges associated with accurately assessing germination responses.
Careful interpretation of germination outcomes is therefore required to avoid both Type I (false positive) and Type II (false negative) errors.43 Statistical power analysis, informed by the variance observed in the Mexico dataset, was conducted to estimate the number of replicates required to minimise these errors. Accordingly, this study is considered a screening-level investigation intended to identify species–antibiotic combinations that warrant further targeted experimentation. It should also be acknowledged that antibiotics were evaluated individually. Under environmental conditions, antibiotics frequently co-occur as complex mixtures, and interactions may be additive, antagonistic, or synergistic. While the single-compound approach adopted here enables controlled comparison of compound-specific effects during early germination, it may underestimate mixture-related responses. The evaluation of antibiotic mixtures therefore represents an important priority for subsequent Tier II studies under environmentally exposure scenarios.
The most pronounced germination reduction observed across all datasets was for black knapweed (C. nigra and Fig. 2g, Table S6), where exposure to all antibiotics resulted in reduced germination relative to the control (53.3%). Reductions ranged from 30% under lincomycin exposure to complete inhibition under ofloxacin. These findings suggest that non-cultivated species, such as C. nigra, may represent sensitive bioindicators for antibiotic-related phytotoxicity. Notably, literature reports indicate that even closely related species can respond differently; for example, brown knapweed (Centaurea jacea) has exhibited a slight but statistically significant increase in germination under tetracycline exposure at 1 mg L−1.44 This highlights substantial interspecific variability in antibiotic responses, even within the same plant genus and antibiotic class.
Another notable reduction in germination was observed for Chinese cabbage (Brassica rapa subsp.pekinensis; Fig. 2f), where a 70% decrease occurred under florfenicol exposure. Similarly, germination reductions were recorded for cornflower (Centaurea cyanus) under sulfamethazine exposure (−30%; Fig. 2i) and for red fescue (Festuca rubra) under chloramphenicol exposure (−20%; Fig. 2h). To date, no studies have evaluated the effects of florfenicol, sulfamethazine, or chloramphenicol on germination in these species. It is hypothesised that the inhibitory responses observed here may relate to interference with protein synthesis and oxidative stress pathways, mechanisms previously reported for other antibiotics in plants.35,53 Moreover, as responses can vary widely even among closely related species,54 these results highlight the importance of species-specific assessments when evaluating antibiotic phytotoxicity.
Azithromycin emerged as the antibiotic exhibiting the most variable effects across species, producing both inhibitory and stimulatory germination responses. In the Mexico dataset, the only observed germination reduction was for cempasúchil (Tagetes erecta), where azithromycin exposure resulted in a 40% decrease relative to the control. In contrast, germination stimulation exceeding 20% was observed in legumes and grasses from the UK, including Avena sativa and Trifolium pratense. In the China dataset, azithromycin exposure resulted in a 27% reduction in germination for soybean (G. max; Fig. 2). These contrasting responses indicate that azithromycin warrants further investigation, as its phytotoxic effects appear to be highly species-dependent.
Root elongation and biomass
Root elongation results are summarised in Table 3, which reports mean, median, and interquartile range (IQR) values for root length, alongside dry biomass differences between treatments and controls and relative change index (RCI) values. Positive RCI values indicate growth stimulation under antibiotic exposure, whereas negative values indicate growth inhibition.| Specie | Treatment | Mean (mm) | Median (mm) | IQR (mm) | Min – max (mm) | Δ mass (mg) (mFDry – mi) | RCI (%) |
|---|---|---|---|---|---|---|---|
| a NG: Not germinated, NA: Not applicable. b No data were collected for chicory seeds, as no germination occurred under any treatment, including the control. | |||||||
| China | |||||||
| Soybean (G. max) | AZI | 114.10 | 89.17 | 101.53 | 42.77–229.22 | −4698.0 | 47.6 |
| CPX | 160.34 | 148.13 | 143.4 | 51.02–255.10 | −3528.1 | 28.0 | |
| CHL | 163.27 | 181.88 | 71.96 | 32.26–239.53 | −3461.9 | 9.1 | |
| ERX | 144.25 | 137.61 | 129.59 | 33.58–251.36 | −3065.2 | 5.8 | |
| FLOR | 166.6 | 171.29 | 91.17 | 16.09–318.39 | −3078.4 | −12.1 | |
| LIN | 160.32 | 139.88 | 179.55 | 24.70–296.49 | −2633.4 | −17.8 | |
| OFX | 152.19 | 150.51 | 136.38 | 15.53–231.90 | −4027.2 | −1.5 | |
| OTC | 115.76 | 122.35 | 168.88 | 7.49–283.35 | −263.1 | −78.0 | |
| SFD | 205.00 | 218.93 | 65.95 | 70.89–269.60 | −2480.5 | −13.0 | |
| TRM | 175.83 | 171.73 | 121.42 | 58.75–247.35 | −4518.0 | 35.8 | |
| Control | 147.92 | 160.98 | 61.79 | 22.76–296.65 | −2825.0 | 0.0 | |
| Wheat (T. aestivum) | AZI | 102.40 | 98.93 | 56.86 | 24.89–148.24 | 536 | −10.2 |
| CPX | 169.24 | 187.58 | 47.97 | 30.08–236.45 | 541.9 | −23.7 | |
| CHL | 108.09 | 127.57 | 94.70 | 9.02–181.28 | 545.7 | −12.4 | |
| ERX | 163.91 | 183.73 | 50.75 | 24.80–245.92 | 273.9 | −1.6 | |
| FLOR | 128.00 | 135.70 | 86.14 | 14.72–226.82 | 240.5 | 1.0 | |
| LIN | 144.91 | 159.42 | 76.37 | 11.00–242.44 | 496.7 | −15.5 | |
| OFX | 178.99 | 183.00 | 58.43 | 79.51–250.16 | 75.7 | 13.4 | |
| OTC | 150.93 | 156.84 | 72.41 | 13.73–217.86 | 186.4 | 7.1 | |
| SFD | 162.46 | 181.49 | 42.21 | 7.30–238.19 | 252.8 | 3.5 | |
| TRM | 131.42 | 159.83 | 117.6 | 14.39–218.82 | 721.5 | −26.5 | |
| Control | 155.10 | 158.11 | 73.82 | 13.61–225.28 | 332.5 | 0.0 | |
| Mung bean (V. radiata) | AZI | 219.38 | 203.13 | 100.44 | 71.04–346.36 | −777.5 | 0.7 |
| CPX | 208.10 | 192.47 | 118.92 | 41.28–315.07 | −304.0 | −19.6 | |
| CHL | 163.00 | 141.07 | 104.475 | 25.10–261.09 | 307.0 | −59.0 | |
| ERX | 170.04 | 159.31 | 110.84 | 22.19–298.91 | −693.9 | 10.8 | |
| FLOR | 158.66 | 145.56 | 116.07 | 13.05–290.92 | −404.3 | −12.1 | |
| LIN | 201.41 | 180.66 | 129.50 | 90.18–320.25 | −130.8 | −40.4 | |
| OFX | 200.59 | 186.47 | 120.63 | 21.78–365.35 | −807.1 | 3.9 | |
| OTC | 155.15 | 176.39 | 152.93 | 12.06–296.20 | −660.3 | 1.6 | |
| SFD | 191.64 | 221.2 | 142.72 | 22.31–329.39 | −401.7 | −16.5 | |
| TRM | 205.85 | 193.8 | 99.57 | 23.43–340.28 | −479.9 | −4.4 | |
| Control | 172.44 | 188.45 | 133.99 | 7.97–293.13 | −747.9 | 0.0 | |
| Lettuce (L. sativa) | AZI | 77.92 | 81.82 | 26.33 | 4.89–118.04 | −12.7 | 38.2 |
| CPX | 68.95 | 68.72 | 45.20 | 9.25–116.13 | 5.6 | 9.3 | |
| CHL | 72.85 | 78.01 | 40.53 | 8.20–138.19 | 11.1 | −14.1 | |
| ERX | 63.01 | 74.64 | 56.89 | 3.16–118.54 | −14.8 | 37.7 | |
| FLOR | 83.98 | 87.75 | 34.76 | 17.04–131.10 | 16.7 | −21.5 | |
| LIN | 81.69 | 83.22 | 14.73 | 51.56–101.72 | 45.5 | −91.2 | |
| OFX | 67.04 | 75.53 | 30.99 | 7.24–106.61 | −2.8 | 8.2 | |
| OTC | 77.28 | 85.78 | 17.78 | 16.69–105.66 | −0.4 | 9.3 | |
| SFD | 81.90 | 85.58 | 34.63 | 18.10–123.64 | 0.5 | 15.4 | |
| TRM | 79.20 | 83.15 | 40.34 | 11.16–119.10 | −16.5 | 23.1 | |
| Control | 77.84 | 82.60 | 33.35 | 16.68–119.38 | 14.9 | 0.0 | |
| Rice (O. sativa) | AZI | 66.68 | 63.21 | 35.86 | 22.34–106.97 | 112.7 | −3.1 |
| CPX | 69.36 | 74.57 | 25.71 | 12.03–97.95 | 143.8 | −9.5 | |
| CHL | 51.35 | 46.27 | 26.45 | 6.79–98.61 | 74.6 | 3.4 | |
| ERX | 53.13 | 58.69 | 31.75 | 9.22–85.47 | 267.8 | −24.6 | |
| FLOR | 65.83 | 67.62 | 20.37 | 21.56–99.34 | 197.5 | −14.3 | |
| LIN | 54.85 | 57.51 | 14.31 | 17.71–82.20 | 79.6 | 0.5 | |
| OFX | 62.26 | 65.14 | 25.22 | 2.66–92.56 | 124.2 | −3.5 | |
| OTC | 60.64 | 63.20 | 30.40 | 9.70–93.53 | 186.8 | −12.7 | |
| SFD | 59.41 | 64.13 | 30.265 | 21.19–88.08 | 178.9 | −16.2 | |
| TRM | 64.35 | 67.23 | 17.0075 | 16.30–95.35 | 68.9 | 4.3 | |
| Control | 43.43 | 46.65 | 35.89 | 2.74–77.86 | 56.9 | 0.0 | |
| Chinese cabbage (B. rapa subsp.Pekinensis) | AZI | 35.52 | 28.36 | 33.035 | 5.99–147.40 | −25.3 | 10.3 |
| CPX | 30.87 | 30.8 | 19.765 | 9.37–97.20 | 10.1 | −45.2 | |
| CHL | 27.4 | 21.2 | 22.115 | 3.33–106.46 | −0.3 | −86.4 | |
| ERX | 29.47 | 25.36 | 27.65 | 4.58–69.42 | −12.2 | −16.5 | |
| FLOR | 13.12 | 12.43 | 4.39 | 9.25–14.33 | −58.5 | 100.0 | |
| LIN | 12.25 | 11.75 | 9.16 | 3.34–24.46 | −12.3 | −5.9 | |
| OFX | 33.8 | 31.32 | 24.76 | 5.63–93.65 | −10 | −5.4 | |
| OTC | 34.77 | 31.64 | 40.885 | 3.27–89.50 | −14.2 | −39.3 | |
| SFD | 32.72 | 24.73 | 31.78 | 5.80–92.67 | −7.3 | −57.9 | |
| TRM | 19.65 | 13.93 | 12.975 | 3.26–43.75 | −9.3 | 0.0 | |
| Control | 24.4 | 17.61 | 26.89 | 2.15–82.88 | −25.2 | 0.0 | |
| Shasta daisy (Leucanthemum maximum) | AZI | 19.57 | 19.63 | 17.15 | 9.4–30.22 | −1.3 | −14.4 |
| CPX | 22.98 | 20.74 | 19.62 | 7.72–29.20 | −6.8 | 11.7 | |
| CHL | 23.16 | 20.71 | 22.58 | 7.18–41.41 | 1.8 | −34.2 | |
| ERX | 17.86 | 9.89 | 17.39 | 7.31–42.97 | −3.5 | 4.5 | |
| FLOR | 11.7 | 7.49 | 4.36 | 6.24–10.60 | −4.6 | 8.1 | |
| LIN | 13.81 | 10.9 | 13.69 | 4.04–23.60 | −2.3 | 9.9 | |
| OFX | 21.64 | 21.15 | 21.08 | 7.61–35.76 | 0.5 | −46.8 | |
| OTC | 19.13 | 11.94 | 24.97 | 3.51–28.48 | 1.8 | −28.8 | |
| SFD | 26.48 | 34.89 | 26.83 | 10.60–37.43 | −1.4 | −5.4 | |
| TRM | 19.67 | 13.42 | 24.57 | 6.71–41.89 | −2.9 | 5.4 | |
| Control | 24.56 | 24.79 | 32.93 | 9.26–43.52 | −2.6 | 0.0 | |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| UK | |||||||
| French bean (P. vulgaris) | AZI | NG | NG | NG | NG | NG | NG |
| CPX | 24.13 | 18.09 | 25.39 | 6.19–48.02 | −1.2048 | −4.1 | |
| CHL | NG | NG | NG | NG | NG | NG | |
| ERX | 37.57 | 29.58 | 31.96 | 11.07–79.33 | −1.3605 | −3.0 | |
| FLOR | 30.7 | 30.7 | NA | 30.7 | −0.9461 | −2.7 | |
| LIN | NG | NG | NG | NG | NG | NG | |
| OFX | NG | NG | NG | NG | NG | NG | |
| OTC | NG | NG | NG | NG | NG | NG | |
| SFZ | 21.83 | 29.45 | NA | 14.2–29.45 | −1.4452 | 5.2 | |
| TRM | NG | NG | NG | NG | NG | NG | |
| Control | 22.7 | 22.7 | NA | 22.7 | −1.1845 | 0.0 | |
| Pea (P. sativum) | AZI | 17.08 | 14.99 | 10.24 | 4.78–48.53 | −0.9375 | 1.5 |
| CPX | 29.46 | 25.99 | 24.96 | 7.789–66.98 | −0.4959 | −14.4 | |
| CHL | 18.87 | 18.47 | 9.96 | 4.02–36.78 | −1.0434 | 12.7 | |
| ERX | 27.94 | 24.43 | 14.14 | 10.84–59.08 | −0.7206 | 6.3 | |
| FLOR | 22.47 | 24.18 | 27.23 | 2.78–40.63 | −0.8992 | 0.9 | |
| LIN | 24.27 | 22.27 | 12.53 | 8.31–50.23 | −0.932 | 27.3 | |
| OFX | 16.73 | 15.62 | 17.79 | 4.15–36.37 | −0.9229 | 4.3 | |
| OTC | 16.55 | 15.41 | 9.55 | 7.48–26.39 | −1.0244 | 2.6 | |
| SFZ | 14.59 | 12.36 | 6.53 | 6.59–39.05 | −1.1888 | 4.1 | |
| TRM | 14.35 | 14.31 | 6.73 | 5.24–21.64 | −1.3664 | 28.1 | |
| Control | 24.47 | 25.34 | 13.82 | 9.65–34.87 | −1.3129 | 0.0 | |
| White clover (T. repens) | AZI | 36.01 | 37.58 | 17.00 | 3.30–57.38 | −0.0038 | −14.3 |
| CPX | 44.72 | 49.45 | 12.68 | 13.55–57.93 | −0.0124 | −76.0 | |
| CHL | 27.46 | 29.25 | 10.06 | 7.56–43.67 | −0.0055 | −4.5 | |
| ERX | 39.06 | 38.56 | 12.34 | 27.61–50.09 | −0.0055 | 1.9 | |
| FLOR | 38.17 | 37.86 | 10.36 | 10.39–56.93 | −0.0063 | 1.3 | |
| LIN | 33.93 | 34.76 | 13.03 | 1.16–48.84 | −0.004 | −3.2 | |
| OFX | 31.77 | 31.88 | 12.34 | 5.25–49.56 | −0.0051 | 8.4 | |
| OTC | 33.11 | 33.30 | 11.36 | 16.51–49.02 | −0.0054 | −8.4 | |
| SFZ | 32.24 | 34.52 | 13.95 | 8.65–52.11 | −0.0055 | −6.5 | |
| TRM | 30.37 | 33.73 | 15.30 | 7.35–45.42 | −0.0016 | −24.0 | |
| Control | 35.31 | 36.82 | 14.15 | 6.10–47.84 | −0.0059 | 0.0 | |
| Red clover (T. pratense) | AZI | 47.05 | 51.01 | 29.52 | 7.97–74.96 | −0.0100 | −6.3 |
| CPX | 33.52 | 31.40 | 12.29 | 13.19–51.86 | −0.0075 | 63.8 | |
| CHL | 33.88 | 33.64 | 27.86 | 8.72–67.21 | −0.0101 | 8.8 | |
| ERX | 37.83 | 37.27 | 23.63 | 6.77–55.54 | −0.0092 | 17.5 | |
| FLOR | 45.61 | 51.64 | 32.69 | 9.65–67.46 | −0.0086 | −1.3 | |
| LIN | 39.69 | 41.31 | 36.11 | 6.48–70.25 | −0.0101 | −5.8 | |
| OFX | 46.13 | 49.24 | 16.03 | 4.11–70.18 | −0.0129 | 12.7 | |
| OTC | 31.58 | 32.99 | 20.68 | 6.22–63.08 | −0.0142 | 2.2 | |
| SFZ | 43.15 | 42.69 | 23.85 | 3.07–72.04 | −0.0101 | −1.0 | |
| TRM | 38.07 | 43.34 | 35.95 | 2.96–62.77 | −0.0099 | 0.5 | |
| Control | 39.90 | 39.32 | 42.43 | 9.68–67.91 | −0.0123 | 0.0 | |
| Oats (A. sativa) | AZI | 29.62 | 25.32 | 39.35 | 4.15–65.62 | −0.0539 | 3.0 |
| CPX | 32.14 | 29.20 | 37.34 | 9.70–61.47 | −0.1212 | 2.0 | |
| CHL | 60.20 | 72.69 | 45.34 | 3.43–91.32 | −0.1404 | 18.0 | |
| ERX | 29.96 | 30.59 | 43.99 | 5.82–52.82 | −0.0936 | 19.8 | |
| FLOR | 48.34 | 54.04 | 32.34 | 4.82–72.42 | −0.1676 | −2.1 | |
| OFX | 35.77 | 34.40 | 33.84 | 7.68–70.40 | −0.1314 | 5.5 | |
| OTC | 14.59 | 12.95 | 16.86 | 5.62–26.85 | −0.1179 | 9.6 | |
| LIN | 20.52 | 14.18 | 31.11 | 5.59–49.01 | −0.1209 | 8.6 | |
| SFZ | 23.34 | 26.02 | 33.22 | 5.40–38.62 | −0.1329 | 3.4 | |
| TRM | 27.89 | 22.25 | 25.48 | 6.25–67.16 | −0.1277 | 14.1 | |
| Control | 22.57 | 23.81 | 25.09 | 3.41–43.66 | −0.1385 | 0.0 | |
| Wheat (T. aestivum) | AZI | 65.04 | 67.02 | 10.25 | 34.08–76.98 | −0.1586 | 1.4 |
| CPX | 58.06 | 64.68 | 22.54 | 13.41–79.69 | −0.1708 | 21.5 | |
| CHL | 77.80 | 81.66 | 20.81 | 13.70–98.67 | −0.2325 | 9.6 | |
| ERX | 63.66 | 67.47 | 12.94 | 22.35–80.46 | −0.2044 | 9.8 | |
| FLOR | 61.29 | 66.14 | 17.69 | 14.52–82.21 | −0.1954 | −2.8 | |
| LIN | 62.29 | 66.83 | 13.22 | 18.81–81.74 | −0.1657 | 17.8 | |
| OFX | 57.45 | 58.30 | 17.92 | 5.64–79.03 | −0.2160 | −9.9 | |
| OTC | 62.65 | 68.63 | 16.61 | 25.45–78.31 | −0.2359 | 3.9 | |
| SFZ | 59.23 | 65.18 | 18.39 | 10.41–78.32 | −0.2200 | 3.1 | |
| TRM | 56.01 | 58.99 | 19.69 | 6.29–83.23 | −0.2346 | 15.5 | |
| Control | 54.63 | 55.03 | 16.96 | 13.32–81.49 | −0.2214 | 0.0 | |
| Barley (H. vulgare) | AZI | 76.04 | 73.76 | 13.79 | 59.49–96.90 | −0.1651 | −4.3 |
| CPX | 63.55 | 69.21 | 21.95 | 13.37–89.77 | −0.2104 | 4.3 | |
| CHL | 92.99 | 94.60 | 14.13 | 72.83–109.23 | −0.282 | 11.1 | |
| ERX | 74.58 | 76.41 | 15.23 | 55.32–92.60 | −0.2388 | 8.1 | |
| FLOR | 79.13 | 78.62 | 13.82 | 52.07–107.12 | −0.229 | 11.3 | |
| OFX | 67.63 | 69.41 | 17.43 | 1.09–91.72 | −0.2273 | 3.5 | |
| OTC | 72.09 | 72.11 | 14.29 | 49.32–89.87 | −0.2444 | 11.9 | |
| LIN | 78.91 | 78.10 | 11.06 | 65.82–98.38 | −0.2271 | 6.9 | |
| SFZ | 71.71 | 73.46 | 12.90 | 53.47–85.08 | −0.2212 | 3.8 | |
| TRM | 78.04 | 76.85 | 12.25 | 61.24–96.28 | −0.2756 | 8.3 | |
| Control | 67.00 | 67.46 | 22.64 | 34.37–92.22 | −0.236 | 0.0 | |
| Sheeps fescue (F. ovina) | AZI | NG | NG | NG | NG | NG | NG |
| CPX | 37.64 | 37.64 | NA | 5.80–69.47 | NA | NA | |
| CHL | 8.00 | 8.00 | NA | 8.00 | NA | NA | |
| ERX | 3.08 | 3.08 | NA | 3.08 | NA | NA | |
| FLOR | 8.35 | 8.35 | NA | 8.35 | NA | NA | |
| LIN | NG | NG | NG | NG | NG | NG | |
| OFX | NG | NG | NG | NG | NG | NG | |
| OTC | NG | NG | NG | NG | NG | NG | |
| SFZ | NG | NG | NG | NG | NG | NG | |
| TRM | NG | NG | NG | NG | NG | NG | |
| Control | NG | NG | NG | NG | NG | NG | |
| Red fescue (Festuca rubra) | AZI | 11.16 | 10.73 | 7.47 | 3.22–22.28 | −0.0028 | −12.5 |
| CPX | 19.48 | 20.67 | 6.5 | 4.71–32.57 | −0.0072 | 14.4 | |
| CHL | 11.98 | 12.65 | 7.78 | 2.57–22.28 | −0.0031 | 22.1 | |
| ERX | 17.63 | 17.98 | 7.39 | 4.08–27.24 | −0.0053 | 11.1 | |
| FLOR | 12.99 | 12.27 | 7.92 | 4.75–45.10 | −0.0033 | 4.8 | |
| LIN | 13.26 | 12.77 | 8.08 | 3.67–.20.61 | −0.0035 | 2.4 | |
| OFX | 14.32 | 15.46 | 4.3 | 3.14–21.86 | −0.0039 | 0.0 | |
| OTC | 11.68 | 10.91 | 8.11 | 1.58–23.55 | −0.0041 | 1.4 | |
| SFZ | 14.49 | 15.84 | 5.71 | 2.81–20.87 | −0.0048 | 0.5 | |
| TRM | 11.38 | 10.82 | 8.6 | 2.84–19.67 | −0.0037 | −3.9 | |
| Control | 14.68 | 15.23 | 6.94 | 5.43–28.06 | −0.0041 | 0.0 | |
| Quaking grass (Briza media) | AZI | NG | NG | NG | NG | NG | NG |
| CPX | NG | NG | NG | NG | NG | NG | |
| CHL | 2.24 | 2.24 | NA | 1.53–2.95 | −0.0016 | 32.5 | |
| ERX | 6.52 | 6.52 | NA | 6.52 | −0.0469 | 45.9 | |
| FLOR | 0.66 | 0.66 | NA | 0.66 | −0.0017 | 24.7 | |
| LIN | 4.03 | 4.03 | NA | 4.03 | −0.0021 | 17.3 | |
| OFX | 1.51 | 1.51 | 0.09 | 1.46–1.55 | −0.0018 | 10.8 | |
| OTC | NG | NG | NG | NG | NG | NG | |
| SFZ | NG | NG | NG | NG | NG | NG | |
| TRM | NG | NG | NG | NG | NG | NG | |
| Control | 1.68 | 1.68 | NA | 1.40–1.96 | −0.0031 | 0 | |
| Cos lettuce (L. sativa) | AZI | 60.81 | 68.36 | 23.77 | 16.94–85.66 | −0.0009 | −10.2 |
| CPX | 48.44 | 50.47 | 37.70 | 15.87–89.84 | −0.0053 | 3.6 | |
| CHL | 60.27 | 63.52 | 19.52 | 6.07–85.21 | −0.0043 | 12.7 | |
| ERX | 58.14 | 59.10 | 25.09 | 32.54–81.36 | −0.0058 | 10.9 | |
| FLOR | 56.94 | 61.39 | 16.10 | 9.61–79.68 | −0.0039 | 6.9 | |
| LIN | 52.10 | 53.71 | 33.24 | 7.76–91.22 | −0.0033 | 0.4 | |
| OFX | 62.82 | 65.49 | 23.08 | 26.26–83.88 | −0.0047 | 2.2 | |
| OTC | 43.40 | 46.23 | 27.19 | 5.41–79.29 | −0.0037 | 7.3 | |
| SFZ | 43.72 | 44.29 | 22.04 | 16.92–64.59 | −0.0044 | 5.8 | |
| TRM | 49.48 | 49.98 | 21.14 | 13.93–71.87 | −0.0035 | 8.0 | |
| Control | 55.40 | 64.35 | 39.46 | 4.05–88.70 | −0.0041 | 0.0 | |
| Oxeye Daisy (L. vulgare) | AZI | 8.80 | 9.49 | 9.94 | 2.47–13.72 | −0.0011 | 2.1 |
| CPX | 13.45 | 12.32 | 14.31 | 5.83–23.48 | −0.0021 | 5.6 | |
| CHL | 7.16 | 7.55 | 2.62 | 5.28–9.16 | −0.0015 | −0.7 | |
| ERX | 10.41 | 7.36 | 14.52 | 3.79–23.12 | −0.0013 | 7.8 | |
| FLOR | 6.57 | 6.57 | NA | 4.55–8.59 | −0.0012 | 9.2 | |
| LIN | 16.46 | 20.16 | 15.20 | 7.01–22.21 | −0.002 | 7.0 | |
| OFX | 12.49 | 14.69 | 8.11 | 7.33–15.45 | −0.0019 | −9.9 | |
| OTC | 6.91 | 6.89 | 6.90 | 2.65–11.22 | −0.0027 | 21.1 | |
| SFZ | 4.81 | 3.77 | 6.94 | 1.66–10.04 | −0.0019 | 11.3 | |
| TRM | 0.46 | 0.46 | NA | 0.46 | −0.0011 | 1.4 | |
| Control | 3.40 | 3.40 | NA | 3.4 | −0.0017 | 0.0 | |
| Black Knapweed (C. nigra) | AZI | 25.53 | 27.37 | 22.75 | 4.10–37.99 | −0.002 | −4.2 |
| CPX | 18.89 | 18.89 | NA | 18.89 | −0.005 | −15.8 | |
| CHL | 8.04 | 7.15 | 3.81 | 6.59–10.39 | −0.0045 | −14.5 | |
| ERX | 10.83 | 10.83 | NA | 6.89–14.77 | −0.0053 | −33.7 | |
| FLOR | 12.04 | 12.04 | NA | 7.69–16.38 | −0.0055 | −26.8 | |
| LIN | 30.49 | 31.13 | 10.13 | 20.22–42.23 | −0.0044 | −12.3 | |
| OFX | NG | NG | NG | NG | NG | −23.2 | |
| OTC | 13.81 | 13.81 | NA | 5.05–22.58 | −0.0063 | −24.8 | |
| SFZ | 6.17 | 6.17 | NA | 6.2 | −0.0108 | −17.6 | |
| TRM | 14.53 | 14.53 | NA | 3.24–25.83 | −0.0057 | −23.9 | |
| Control | 29.84 | 29.49 | 18.42 | 12.17–49.95 | −0.0041 | 0.0 | |
| Cornflower (C. cyanus) | AZI | 85.70 | 92.66 | 36.83 | 5.34–159.35 | 0.1479 | 3.1 |
| CPX | 92.37 | 93.61 | 40.63 | 16.19–157.37 | −0.0055 | 2.1 | |
| CHL | 97.78 | 103.20 | 48.38 | 14.38–142.28 | −0.0092 | 14.4 | |
| ERX | 90.49 | 91.50 | 27.43 | 39.05–165.10 | −0.0052 | 2.9 | |
| FLOR | 95.85 | 96.54 | 41.24 | 20.97–158.50 | −0.0060 | 4.1 | |
| LIN | 72.83 | 75.47 | 13.06 | 32.13–91.61 | −0.0032 | 4.2 | |
| OFX | 86.75 | 89.25 | 28.08 | 27.05–124.10 | −0.0116 | 10.5 | |
| OTC | 75.97 | 71.41 | 41.57 | 34.13–152.55 | −0.0100 | 9.8 | |
| SFZ | 75.92 | 79.75 | 36.93 | 21.90–128.37 | −0.0085 | 6.7 | |
| TRM | 100.53 | 102.75 | 49.75 | 31.01–150.23 | −0.0090 | 4.8 | |
| Control | 82.28 | 78.45 | 57.84 | 14.34–160.36 | −0.0072 | 0.0 | |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Mexico | |||||||
| Tomate (S. lycopersicum) | AZI | 5.97 ± 4.41 | 3.19 | 10.06 | 1.61–11.67 | −1.30 ± 0.44 | 1.8 ± 4.8 |
| CPX | 5.10 ± 2.68 | 4.19 | 5.62 | 2.23–11.41 | −4.90 ± 0.98 | 14.6 ± 6.2 | |
| OTC | 5.25 ± 0.84 | 4.85 | 5.18 | 1.66–11.02 | −1.33 ± 0.21 | −2.7 ± 2.5 | |
| TRM | 3.91 ± 2.92 | 3.285 | 5.28 | 1.64–12.48 | −1.43 ± 0.67 | −5.2 ± 5.3 | |
| Control | 4.04 ± 1.06 | 3.18 | 4.35 | 1.03–8.28 | −1.57 ± 0.42 | 0.0 | |
| Cempasuchil (T. erecta) | AZI | 59.75 ± 7.94 | 54.18 | 51.94 | 4.41–144.59 | −3.57 ± 2.00 | −3.1 ± 11.1 |
| CPX | 38.17 ± 7.52 | 33.01 | 32.20 | 5.02–89.77 | −2.77 ± 2.55 | −2.9 ± 9.8 | |
| OTC | 73.34 ± 21.83 | 81.61 | 55.33 | 4.09–159.56 | −7.47 ± 1.21 | −16.6 ± 7.7 | |
| TRM | 76.52 ± 19.13 | 69.04 | 73.85 | 5.14–161.05 | −4.67 ± 0.29 | −10.3 ± 3.9 | |
| Control | 61.20 ± 14.96 | 57.18 | 53.79 | 6.32–171.69 | −4.72 ± 2.59 | 0.0 | |
| Carnation (D. caryophyllus) | AZI | 4.77 ± 0.93 | 4.06 | 3.62 | 0.99–11.02 | −2.57 ± 0.57 | −2.9 ± 4.7 |
| CPX | 4.99 ± 2.39 | 3.98 | 2.49 | 1.53–23.74 | −1.50 ± 0.95 | −10.6 ± 6.8 | |
| OTC | 5.85 ± 0.89 | 4.92 | 6.41 | 1.51–12.88 | −1.00 ± 0.70 | −19.4 ± 21.7 | |
| TRM | 8.48 ± 2.58 | 6.02 | 10.09 | 1.07–27.96 | −2.70 ± 2.93 | 2.3 ± 11.0 | |
| Control | 5.02 ± 0.56 | 3.6 | 3.865 | 1.17–19.73 | −2.57 ± 1.28 | 0.0 | |
| Alfalfa (M. sativa) | AZI | 23.79 ± 11.22 | 12.27 | 32.2 | 6.38–87.62 | −8.23 ± 2.67 | 3.8 ± 4.8 |
| CPX | 35.61 ± 6.20 | 18.8 | 55.57 | 3.28–85.58 | −8.10 ± 1.13 | −9.5 ± 8.9 | |
| OTC | 39.30 ± 2.49 | 38.045 | 37.24 | 5.91–77.31 | −6.87 ± 0.60 | −20.4 ± 2.6 | |
| TRM | 36.84 ± 5.88 | 30.64 | 50.63 | 6.1–93.61 | −7.17 ± 0.55 | −15.8 ± 6.1 | |
| Control | 19.91 ± 9.58 | 10.07 | 11.02 | 4.41–108.92 | −8.82 ± 1.71 | 0.0 | |
High variability in root length responses was observed across species and antibiotic treatments (Table 3; Fig. 3–5), indicating heterogeneous seedling responses under exposure conditions. Such variability is consistent with species-specific differences in root sensitivity and antibiotic interactions, which have been associated in previous studies with root electrochemical properties and oxidative stress responses.35,55 These observations highlight the need for increased replication and targeted follow-up experiments to confirm patterns identified in this Tier I screening.
For the Mexico dataset, Kruskal–Wallis tests indicated significant overall treatment effects on root elongation for cempasúchil (Tagetes erecta; p = 0.007) and alfalfa (Medicago sativa; p = 0.001), while post-hoc (DSCF) tests identified a statistically significant pairwise effect only for alfalfa under oxytetracycline exposure (p = 0.003; Table 2). In this case, oxytetracycline exposure resulted in a marked increase in median root length from 10.0 mm in the control to 38.0 mm (Table 3; Fig. 3d), indicating substantial stimulation of root elongation. In contrast, while oxytetracycline exposure was associated with increased germination in soybean (Glycine max; Fig. 2a), it resulted in pronounced inhibition of root elongation (122.35 mm compared to 160.98 mm in the control), corresponding to an RCI value of −78%. This demonstrates that enhanced germination does not necessarily translate into increased early root growth. Notably, oxytetracycline exhibited divergent effects in alfalfa and soybean, promoting both germination and root elongation in the former while inhibiting root elongation in the latter. These contrasting responses highlight the importance of assessing sub-lethal endpoints beyond germination alone when evaluating early seedling establishment and potential impacts on plant development.
Following the germination stimulation observed under fluoroquinolone exposure in grasses, similar trends were observed for root elongation. In wheat (Triticum aestivum), median root length increased following exposure to fluoroquinolones (Fig. 4b). Likewise, rice (Oryza sativa) exhibited increased median root length under ciprofloxacin exposure (74.57 mm compared to 46.65 mm in the control; Fig. 4c), accompanied by a negative relative change index (RCI = −9.55; Table 3), indicating increased biomass. Comparable responses were observed in pea (Pisum sativum; Fig. 5a), where ciprofloxacin exposure resulted in a 50% increase in germination together with a negative RCI value of −14.4%, consistent with growth stimulation. These trends align with previous reports describing hormetic responses in plants exposed to low concentrations of fluoroquinolones and other antibiotics, where enhanced root growth has been associated with stress-related signalling pathways, including increased ROS production and activation of antioxidant defence mechanisms.25 However, exceptions to this general trend were also observed. In red clover (Trifolium pratense; Fig. 5c), ciprofloxacin exposure resulted in increased germination, while the RCI value was positive (63.8%), indicating reduced biomass accumulation rather than growth stimulation. Similar response patterns have been reported in previous studies, where ciprofloxacin and related antibiotics promoted seed germination but inhibited subsequent biomass accumulation in plants.35,49 These findings suggest that early seed activation does not necessarily translate into enhanced seedling growth and may reflect differential sensitivities of metabolic processes and stress responses during early development. In contrast, ciprofloxacin exposure in white clover (Trifolium repens; Fig. 5b) resulted in stimulation of both biomass accumulation (RCI = −76%) and median root length (increase of 12.7 mm). The divergent responses observed between closely related species, such as red and white clover, highlight pronounced species-specific physiological differences that influence antibiotic sensitivity and downstream growth outcomes.
Contrasting responses to the same antibiotic were also observed for florfenicol. In Chinese cabbage (Brassica rapa subsp. pekinensis), germination was reduced by 70% under florfenicol exposure (Table 2), and this inhibitory effect persisted across additional endpoints, including root elongation (median root length of 12 mm compared to 17.61 mm in the control) and biomass accumulation, with an RCI value of 99.97%, indicating near-complete growth inhibition (Fig. 4d). In contrast, red clover (Trifolium pratense) exhibited increased median root length under florfenicol (51.64 mm) and azithromycin (51.01 mm) exposure compared to the control (39.32 mm; Fig. 5c), with corresponding RCI values of −1.3% and −6.3%, respectively. Similarly, oats (Avena sativa) displayed a marked increase in median root elongation under florfenicol exposure (54.40 mm compared to 23.81 mm in the control; RCI = −2.1%), although this response was less pronounced than that observed under chloramphenicol exposure (72.69 mm; RCI = 18%). Across these cases, increases in root elongation were not accompanied by proportional increases in biomass accumulation, as indicated by RCI values. This pattern suggests that, under antibiotic exposure, seedlings may preferentially allocate resources towards root elongation during early establishment, rather than overall biomass production. Similar response patterns have been discussed previously in the context of stress adaptation and resource allocation under chemical exposure.35,54,56
Consistent with the germination results, black knapweed (Centaurea nigra) exhibited inhibitory responses in root elongation under all antibiotics tested, with median root lengths markedly reduced relative to the control value of 29.49 mm (Fig. 5h). The most pronounced reductions were observed under sulfamethazine (6.17 mm), chloramphenicol (7.15 mm), and enrofloxacin (10.83 mm). These results further support the high sensitivity of C. nigra to antibiotic exposure across multiple early growth endpoints. Pea (Pisum sativum) was also identified as sensitive to antibiotic exposure. This species exhibited a median root length of 25.3 mm in the control, which decreased under exposure to several antibiotics, accompanied by reductions in biomass accumulation. The most notable effect was observed under trimethoprim exposure, where median root length decreased to 14.3 mm with a corresponding RCI value of 28.1%. Root length reductions and biomass inhibition were also recorded under lincomycin, azithromycin, oxytetracycline, and sulfamethazine exposure (Table 3 and Fig. 5a). In contrast, enrofloxacin exposure in P. sativum resulted in germination stimulation (36.6%), while the RCI value remained positive (6.3%), indicating that increased germination was not associated with enhanced biomass accumulation. This further illustrates that stimulation of early germination does not necessarily translate into improved seedling growth or establishment.
Chloramphenicol also exhibited marked effects on root elongation across cereal species. In wheat (Triticum aestivum), median root length increased substantially from 55.0 mm in the control to 81.7 mm under chloramphenicol exposure (Table 3 and Fig. 5e). Similarly, in barley (Hordeum vulgare; Fig. 5f), median root length increased from 67.5 mm in the control to 94.6 mm following chloramphenicol exposure, representing a markedly greater response than those observed under florfenicol (78.6 mm) and lincomycin (78.1 mm). The consistent stimulation of root elongation observed across cereal species under chloramphenicol exposure suggests recurring response patterns that merit further investigation using higher-powered and mechanistically focused experimental designs.
Standard toxicity test evaluation
| Statistical power | Experiment number | Replicates per treatment | |
|---|---|---|---|
| Tomate (S. lycopersicum) | 70% | 19 | 4 |
| 80% | 23 | 5 | |
| 90% | 27 | 6 | |
| Cempasuchil (T. erecta) | 70% | 25 | 5 |
| 80% | 30 | 6 | |
| 90% | 36 | 8 | |
| Carnation (D. caryophyllus) | 70% | 35 | 7 |
| 80% | 45 | 9 | |
| 90% | 48 | 12 | |
| Alfalfa (M. sativa) | 70% | 11 | 3 |
| 80% | 11 | 3 | |
| 90% | 12 | 4 |
Underpowered studies may therefore underestimate subtle or variable phytotoxic or stimulatory effects, particularly when assessing complex stressors such as antibiotics. These considerations highlight the practical and financial challenges associated with evaluating seedling toxicity across a broad range of species and chemical stressors.43,59 When applied to the UK and China datasets, the replication and experimental effort estimated in this analysis remains directly relevant. In cases where no statistically significant effects were observed (e.g., lettuce or oxeye daisy), the absence of significance may reflect limitations in experimental power rather than a true absence of biological effects. To robustly distinguish between these possibilities, future studies should plan replication levels that meet or exceed those recommended in Table 4 for each species. One practical strategy to address these constraints is the use of screening-level assays, such as the approach adopted in this study, where broad patterns and potentially sensitive species–compound combinations can be identified initially and subsequently investigated using higher replication to achieve the statistical power required for confirmatory testing. This tiered approach minimises the risk of overlooking real effects while maintaining experimental feasibility.
| Tomato | Cempasuchil | Carnation | Alfalfa | No seeds | |||||
|---|---|---|---|---|---|---|---|---|---|
| ΔC% | Seeds removal (%) | ΔC% | Seeds removal (%) | ΔC% | Seeds removal (%) | ΔC% | Seeds removal (%) | ΔC% | |
| AZI | 84.67 ± 4.19 | 38.03 ± 2.86 | 84.66 ± 2.86 | 38.02 ± 3.44 | 56.17 ± 9.08 | 9.53 ± 6.05 | 57.44 ± 5.96 | 10.8 ± 4.57 | 46.64 ± 7.38 |
| CPX | 75.52 ± 4.65 | 17.12 ± 4.33 | 79.94 ± 7.80 | 21.54 ± 5.64 | 58.74 ± 12.30 | 0.34 ± 7.87 | 53.73 ± 12.37 | −4.67 ± 7.91 | 58.40 ± 8.33 |
| OTC | 30.32 ± 14.05 | −1.19 ± 8.92 | 17.89 ± 7.85 | −13.62 ± 5.86 | 32.70 ± 2.16 | 1.19 ± 3.91 | 20.82 ± 7.74 | −10.69 ± 5.81 | 31.51 ± 9.09 |
| TRM | 5.26 ± 3.16 | 7.4 ± 4.25 | 22.66 ± 5.40 | 10.00 ± 4.95 | 14.47 ± 6.68 | −17.04 ± 5.44 | 13.35 ± 1.81 | 0.69 ± 3.98 | 12.66 ± 9.41 |
Across the antibiotics tested, the concentrations decreased during the exposure period, ranging from 12.66 ± 9.41% for trimethoprim to 58.40 ± 8.33% for ciprofloxacin. These reductions are most plausibly attributed to sorption onto the filter paper. Consistent with this interpretation, the apparent partition coefficients (Kd,app) calculated after 7 days followed the order: ciprofloxacin (≈1.40) > azithromycin (≈0.87) > oxytetracycline (≈0.46) > trimethoprim (≈0.15). This ranking mirrors the magnitude of concentration losses observed in the aqueous phase. These Kd,app values are specific to the experimental water–filter paper system and are used here solely for comparative purposes. Because the mass-to-volume ratio (m V−1) remained constant across experiments, differences in sorption behaviour primarily reflect intrinsic physicochemical properties of the antibiotics, including acid-base speciation, hydrogen bonding capacity, and electrostatic interactions with cellulose. Understanding these partitioning dynamics is critical for estimating the fraction of antibiotics that remained freely available for seed exposure (Table 5). Sorption to the filter paper effectively reduces dissolved antibiotic concentrations, which may lead to underestimation of phytotoxic effects and complicates direct comparison with environmental scenarios, where sorption dynamics depend strongly on soil properties and organic matter content.
Separately, and beyond sorption processes, the potential role of root exudates should also be considered. For example, tomato root exudates are known to contain organic acids, amino acids, sugars, and phenolic compounds that might modify local chemical conditions and enhance the desorption and bioavailability of certain pollutants.60 In the absence of supporting water-quality measurements (e.g., pH, electrical conductivity, total organic carbon), however, this interpretation remains speculative and is therefore presented as a plausible mechanism rather than a demonstrated process. This hypothesis is consistent with the observed behaviour of azithromycin and ciprofloxacin, which exhibit a higher cationic or neutral fraction at near-neutral pH, compared to oxytetracycline and trimethoprim, which are more likely to occur as zwitterionic, anionic, or neutral species with lower affinity for cellulose-based filter paper. Such differences in speciation may influence both sorption and desorption dynamics within the experimental system. This framework also provides a potential explanation for the occurrence of negative seed-removal values, defined as the net difference in antibiotic loss between dishes with seeds and control dishes without seeds. Rather than reflecting direct uptake by seeds, these negative values suggest that seed-associated processes, including the release of root exudates or other abiotic interactions, may have promoted desorption of antibiotics from the filter paper, resulting in higher aqueous concentrations after initial sorption.61 Consequently, the seed-removal metric does not exclusively represent plant uptake but instead integrates all seed-associated processes that may either decrease or increase antibiotic availability in solution.
In summary, sorption to filter paper can reduce effective antibiotic exposure concentrations, potentially leading to underestimation of phytotoxic effects in controlled assays. Conversely, seed- and root-associated processes, such as exudate-mediated interactions, may locally modify pollutant bioavailability and influence early seedling development. Direct assessment of exudate-mediated desorption would require additional water-quality measurements and should be addressed in future hypothesis-driven studies. These interactions highlight the complex relationship between antibiotic chemistry and processes at the root surface environment, underlining the importance of considering both physicochemical fate and biological responses in phytotoxicological assessments.61
Short-term assays (<7 days) conducted under fixed temperature, light, and hydroponic conditions may fail to capture germination responses in species that require longer germination periods or specific environmental cues, such as soil-based substrates, distinct temperature ranges, or light exposure. The three species showing no germination in this study are terrestrial plants for which germination is typically recommended in moist, well-drained soils over extended periods. Consequently, the observed low germination likely reflects a mismatch between their ecological requirements and the uniform hydroponic conditions applied here, rather than an absence of viability. For example, optimal germination of sheep's fescue (F. ovina) has been reported at approximately 15 °C, with reduced germination occurring at temperatures between 20 and 30 °C.62 Similarly, quaking grass (B. media) exhibits specific requirements for light exposure and an optimal germination temperature of 28.9 °C; its germination is also influenced by sowing depth, with higher rates reported at shallow depths of 0.5–2 mm.63 These ecological requirements may explain the low germination observed under the fixed temperature (22 °C) and hydroponic conditions used in this study. Rather than indicating an inappropriate experimental design, the exclusion of these species underscores the need for adaptable ecotoxicological testing protocols that account for intra- and inter-species variability. Incorporating species-specific germination requirements is particularly important when assessing non-cultivated or regionally relevant species, whose responses may be overlooked under standardised conditions.
Standard phytotoxicity guidelines often require a minimum of 70% germination under fixed hydroponic, temperature, and light conditions. While this criterion was met for several species tested in Mexico, the UK, and China (including French bean, pea, oats, barley, oxeye daisy, black knapweed, chicory, Shasta daisy, and tomato), such requirements may inadvertently restrict the inclusion of lesser-studied or ecologically important species. Expanding phytotoxicity testing frameworks to allow customised, species-specific germination conditions would enhance both the inclusiveness and ecological relevance of future terrestrial risk assessments.
Conclusions
This study presents a broad Tier I screening of germination and early seedling phytotoxicity for ten antibiotics across 23 plant species, identifying both sensitive species and compounds that warrant further, hypothesis-driven investigation. In addition, this work provides practical guidance on experimental design requirements needed to achieve adequate statistical power in future phytotoxicity studies. A central finding is that antibiotic effects on germination and early growth are highly species- and compound-specific. While fluoroquinolones generally stimulated germination and root elongation in grasses and legumes, notable exceptions highlight the complexity of plant–antibiotic interactions and caution against generalisation across taxa or antibiotic classes. Importantly, the identification of non-cultivated species, such as black knapweed (Centaurea nigra), as particularly sensitive to antibiotic exposure underscores the need to extend phytotoxicity assessments beyond agriculturally or commercially relevant crops. Such species represent promising bioindicators for environmental monitoring of antibiotic contamination. This study also demonstrates that sublethal endpoints, including changes in root elongation and biomass, provide critical information beyond simple germination inhibition or stimulation. Incorporating these endpoints alongside a geographically diverse selection of cultivated and non-cultivated species is essential for a more comprehensive understanding of antibiotic impacts on terrestrial plant communities. Lastly, the results highlight limitations of standard phytotoxicity testing frameworks, as many environmentally relevant species fail to germinate under fixed light, temperature, and hydroponic conditions. More flexible, species-adapted protocols are therefore required to improve ecological relevance. Accurate quantification of antibiotic concentrations throughout experiments is equally critical, as processes such as sorption, degradation, and seed-associated interactions directly influence effective exposure levels and the interpretation of phytotoxic effects. Together, these findings support the need for adaptive, tiered phytotoxicity assessments to better capture the environmental risks posed by antibiotics in terrestrial ecosystems.Author contributions
RSCL: conceptualization, data curation, formal analysis, investigation, methodology, software validation, visualization, writing–original draft. AGJ: conceptualization, data curation, methodology, writing–original draft, project administration, supervision. JCDA: supervision, writing–review & editing, resources, funding acquisition. FE: conceptualization, methodology, writing–review & editing. WSB: conceptualization, investigation, formal analysis, data curation, resources. JG: investigation, software, formal analysis, data curation, writing-review & editing. JN: investigation, writing–review & editing. FZ: conceptualization, investigation, formal analysis, data curation, resources, funding acquisition. LC: conceptualization, funding acquisition, methodology, project administration, resources, writing–review & editingConflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study, including all statistical summaries and results presented in the main text and figures, are included within the article and its supplementary information (SI). The complete raw measurements for root length and seedling growth are available as part of the supplementary information. Supplementary information: Tables (antibiotic details, seed sterilisation, environmental concentrations reported in irrigation water and soil pore-water compared with the nominal concentration used in this study, MRM quantification parameters, and method validation parameters) and Figures (germination and root length data for China and UK species; detailed protocols, and raw data). See DOI: https://doi.org/10.1039/d6em00053c.Acknowledgements
RSCL (CVU 1101000) would like to acknowledge the support from Secretaría de Ciencia, Humanidades, Tecnología e Innovación (Secihti), for her PhD scholarship. AGJ, FE, JG, JN, and LC would like to thank UK Research and Innovation (UKRI) for the funding from a Future Leaders Fellowship (grant no. MR/S032126/1). WSB and FZ would like to thank the support from Fundamental Research Funds for the Central Universities (KYCYXT2023001), China.Notes and references
- J. Wu, J. Wang, Z. Li, S. Guo, K. Li and P. Xu, et al., Antibiotics and antibiotic resistance genes in agricultural soils: A systematic analysis, Crit. Rev. Environ. Sci. Technol., 2023, 53(7), 847–864 Search PubMed.
- L. B. Price, B. J. Koch and B. A. Hungate, Ominous projections for global antibiotic use in food-animal production, Proc. Natl. Acad. Sci., 2015, 112(18), 5554–5555 Search PubMed.
- M. Gonzalez Ronquillo and E. Vargas-Bello-Pérez, Editorial: The use of growth promoters and their alternatives in livestock production, Front. Vet. Sci., 2022, 9, 945308 Search PubMed.
- S. I. Polianciuc, A. E. Gurzău, B. Kiss, M. G. Ștefan and F. Loghin, Antibiotics in the environment: causes and consequences, Med. Pharm. Rep., 2020, 93(3), 231–240 CrossRef PubMed.
- L. J. Carter, B. Adams, T. Berman, N. Cohen, E. Cytryn and F. C. T. Elder, et al., Co-contaminant risks in water reuse and biosolids application for agriculture, Environ. Pollut., 2025, 375, 126219 CrossRef CAS PubMed.
- M. Cycoń, A. Mrozik and Z. Piotrowska-Seget, Antibiotics in the Soil Environment—Degradation and Their Impact on Microbial Activity and Diversity, Front. Microbiol., 2019, 10, 1–45 Search PubMed.
- W. L. Jia, C. Song, L. Y. He, B. Wang, F. Z. Gao and M. Zhang, et al., Antibiotics in soil and water: Occurrence, fate, and risk, Curr. Opin. Environ. Sci. Health, 2023, 32, 100437 CrossRef.
- Y. Zha, Q. Li, H. Liu, Y. Ge, Y. Wei and H. Wang, et al., Occurrence and ecological risk assessment of antibiotics in manure and the surrounding soil from typical chicken farms in Hangzhou, China, Front. Environ. Sci., 2023, 11, 1241405 CrossRef.
- B. P. Bougnom, S. Thiele-Bruhn, V. Ricci, C. Zongo and L. J. V. Piddock, Raw wastewater irrigation for urban agriculture in three African cities increases the abundance of transferable antibiotic resistance genes in soil, including those encoding extended spectrum β-lactamases (ESBLs), Sci. Total Environ., 2020, 698, 134201 CrossRef CAS PubMed.
- A. C. Chávez-Mejía, I. Navarro-González, R. Magaña-López, D. Uscanga-Roldán, P. I. Zaragoza-Sánchez and B. E. Jiménez-Cisneros, Presence and natural treatment of organic micropollutants and their risks after 100 years of incidentalwater reuse in agricultural irrigation, Water, 2019, 11(10), 1–17 CrossRef.
- A. L. Garduño-Jiménez, J. C. Durán-Álvarez, C. A. Ortori, S. Abdelrazig, D. A. Barrett and R. L. Gomes, Delivering on sustainable development goals in wastewater reuse for agriculture: Initial prioritization of emerging pollutants in the Tula Valley, Mexico, Water Res., 2023, 238, 119903 CrossRef PubMed.
- J. C. Durán-Álvarez, B. Prado, D. González, Y. Sánchez and B. Jiménez-Cisneros, Environmental fate of naproxen, carbamazepine and triclosan in wastewater, surface water and wastewater irrigated soil — Results of laboratory scale experiments, Sci. Total Environ., 2015, 538, 350–362 CrossRef PubMed.
- A. L. Garduño-Jiménez and L. J. Carter, Insights into mode of action mediated responses following pharmaceutical uptake and accumulation in plants, Front. Agron., 2024, 5, 1293555 CrossRef.
- I. Nybom, T. D. Bucheli and G. Garland, Antibiotics Uptake from Soil and Translocation in the Plants – Meta-analysis, Chimia, 2024, 78(4), 209–214 CrossRef CAS PubMed.
- M. E. Ben, V. Mordehay, J. Tarchitzky and B. Chefetz, Pharmaceuticals in edible crops irrigated with reclaimed wastewater: Evidence from a large survey in Israel, J. Hazard. Mater., 2021, 416, 126184 Search PubMed.
- R. Camacho-Arévalo, C. García-Delgado, B. Mayans, R. Antón-Herrero, J. Cuevas and M. L. Segura, et al., Sulfonamides in tomato from commercial greenhouses irrigated with reclaimed wastewater: Uptake, translocation and food safety, Agronomy, 2021, 11(5), 1–17 Search PubMed.
- L. Frey, B. Tanunchai and B. Glaser, Antibiotics residues in pig slurry and manure and its environmental contamination potential. A meta-analysis, Agron. Sustain. Dev., 2022, 42(2), 31 CrossRef CAS.
- S. Mansilla, J. Portugal, J. M. Bayona, V. Matamoros, A. M. Leiva and G. Vidal, et al., Compounds of emerging concern as new plant stressors linked to water reuse and biosolid application in agriculture, J. Environ. Chem. Eng., 2021, 9(3), 1–9 Search PubMed.
- D. G. Hillis, J. Fletcher, K. R. Solomon and P. K. Sibley, Effects of Ten Antibiotics on Seed Germination and Root Elongation in Three Plant Species, Arch. Environ. Contam. Toxicol., 2011, 60(2), 220–232 CrossRef CAS PubMed.
- L. Aristilde, A. Melis and G. Sposito, Inhibition of Photosynthesis by a Fluoroquinolone Antibiotic, Environ. Sci. Technol., 2010, 44(4), 1444–1450 CrossRef CAS PubMed.
- M. S. Cheong, K. H. Seo, H. Chohra, Y. E. Yoon, H. Choe and V. Kantharaj, et al., Influence of Sulfonamide Contamination Derived from Veterinary Antibiotics on Plant Growth and Development, Antibiotics, 2020, 9(8), 456 CrossRef CAS PubMed.
- O. Opriş, F. Copaciu, M. Loredana Soran, D. Ristoiu, Ü. Niinemets and L. Copolovici, Influence of nine antibiotics on key secondary metabolites and physiological characteristics in Triticum aestivum: Leaf volatiles as a promising new tool to assess toxicity, Ecotoxicol. Environ. Saf., 2013, 87, 70–79 CrossRef PubMed.
- X. Liu, Y. Lv, K. Xu, X. Xiao, B. Xi and S. Lu, Response of ginger growth to a tetracycline-contaminated environment and residues of antibiotic and antibiotic resistance genes, Chemosphere, 2018, 201, 137–143 CrossRef CAS PubMed.
- M. P. Gomes, D. C. Rocha, J. C. Moreira De Brito, D. S. Tavares, R. Z. Marques and P. Soffiatti, et al., Emerging contaminants in water used for maize irrigation: Economic and food safety losses associated with ciprofloxacin and glyphosate, Ecotoxicol. Environ. Saf., 2020, 196, 110549 CrossRef CAS PubMed.
- J. Jin, L. Xu, S. Zhang, M. Jin, P. Zhang and L. Shen, et al., Oxidative response of rice (Oryza sativa L.) seedlings to quinolone antibiotics and its correlation with phyllosphere microbes and antibiotic resistance genes, Sci. Total Environ., 2023, 867, 161544 CrossRef CAS PubMed.
- P. McKenna, N. Cannon, J. Conway and J. Dooley, The use of red clover (Trifolium pratense) in soil fertility-building: A Review, Field Crops Res., 2018, 221, 38–49 CrossRef.
- A. J. Browne, M. G. Chipeta, G. Haines-Woodhouse, E. P. A. Kumaran, B. H. K. Hamadani and S. Zaraa, et al., Global antibiotic consumption and usage in humans, 2000–18: a spatial modelling study, Lancet Planet. Health, 2021, 5(12), e893–e904 CrossRef PubMed.
- E. Y. Klein, I. Impalli, S. Poleon, P. Denoel, M. Cipriano and T. P. Van Boeckel, et al., Global trends in antibiotic consumption during 2016–2023 and future projections through 2030, Proc. Natl. Acad. Sci., 2024, 121(49), e2411919121 CrossRef CAS PubMed.
- OECD. OECD Test Guideline 208: Terrestrial Plant Test - Seedling Emergence and Seedling Growth Test. Guidel Test Chem Terr Plant Test Seedl Emergence Seedl Growth Test. 2006227(September):1–21.
- USEPA. Protocols for short- term toxicity screening of hazardous waste sites, EPA/600/3–88/029. 1988.
- M. D. Piwoni, J. W. Keeley. Basic concepts of contaminant sorption at hazardous waste sites. Ground Water Issue -EPA5404-90053. 19901–25.
- K. Stando, E. Korzeniewska, E. Felis, M. Harnisz, M. Buta-Hubeny and S. Bajkacz, Determination of antimicrobial agents and their transformation products in an agricultural water-soil system modified with manure, Sci. Rep., 2022, 12(1), 17529 Search PubMed.
- Y. Chen, K. Cui, Q. Huang, Z. Guo, Y. Huang and K. Yu, et al., Comprehensive insights into the occurrence, distribution, risk assessment and indicator screening of antibiotics in a large drinking reservoir system, Sci. Total Environ., 2020, 716, 137060 Search PubMed.
- Q. L. Zhong, Z. Chen, Q. Shen and J. Q. Xiong, Occurrence of antibiotics in reclaimed water, and their uptake dynamics, phytotoxicity, and metabolic fate in Lolium perenne L, Sci. Total Environ., 2023, 904, 166975 CrossRef CAS PubMed.
- V. Minden, A. Deloy, A. M. Volkert, S. D. Leonhardt and G. Pufal, Antibiotics impact plant traits, even at small concentrations, AoB Plants, 2017, 9(2), 1–19 CrossRef PubMed.
- E. Ranucci, S. Treccani, P. Ferruti and J. Alongi, The Seed Germination Test as a Valuable Tool for the Short-Term Phytotoxicity Screening of Water-Soluble Polyamidoamines, Polymers, 2024, 16(12), 1744 CrossRef CAS PubMed.
- F. Liu, G. G. Ying, R. Tao, J. L. Zhao, J. F. Yang and L. F. Zhao, Effects of six selected antibiotics on plant growth and soil microbial and enzymatic activities, Environ. Pollut., 2009, 157(5), 1636–1642 Search PubMed.
- R. Wang, Y. Yang, X. Wang, J. Li, Y. Gao and H. Huang, et al., Response of seed germination and seedling growth of perennial ryegrass (Lolium perenne L.) to drought, salinity, and pH in Karst regions, Sci. Rep., 2025, 15(1), 16874 Search PubMed.
- Q. Javed, J. Sun, A. Azeem, K. Jabran and D. Du, Competitive ability and plasticity of Wedelia trilobata (L.) under wetland hydrological variations, Sci. Rep., 2020, 10(1), 9431 CrossRef CAS PubMed.
- R. Díaz-Sierra, M. Verwijmeren, M. Rietkerk, V. R. De Dios and M. Baudena, A new family of standardized and symmetric indices for measuring the intensity and importance of plant neighbour effects, Methods Ecol. Evol., 2017, 8(5), 580–591 CrossRef.
- F. R. Hay, S. Rezaei and J. Buitink, Seed Moisture Isotherms, Sorption Models, and Longevity, Front. Plant Sci., 2022, 13, 891913 CrossRef PubMed.
- S. Rezaei, J. Buitink and F. R. Hay, Contrasting seed moisture sorption behaviour between two species and the implication for seed longevity, Seed Sci. Res., 2023, 33(2), 126–134 CrossRef.
- G. W. Sileshi, A critique of current trends in the statistical analysis of seed germination and viability data, Seed Sci. Res., 2012, 22(3), 145–159 CrossRef.
- V. J. Pereira and D. G. Santana, Coefficient of variation of normal seedlings obtained from the validation of methods for the seed germination testing of 20 species belonging to the family Fabaceae, J. Seed Sci., 2013, 35(2), 161–170 CrossRef.
- E. Małkowski, K. Sitko, M. Szopiński, Ż. Gieroń, M. Pogrzeba and H. M. Kalaji, et al., Hormesis in Plants: The Role of Oxidative Stress, Auxins and Photosynthesis in Corn Treated with Cd or Pb, Int. J. Mol. Sci., 2020, 21(6), 2099 CrossRef PubMed.
- A. Jalal, J. C. D. Oliveira Junior, J. S. Ribeiro, G. C. Fernandes, G. G. Mariano and V. D. R. Trindade, et al., Hormesis in plants: Physiological and biochemical responses, Ecotoxicol. Environ. Saf., 2021, 207, 111225 CrossRef CAS PubMed.
- M. Salinitro, G. Mattarello, G. Guardigli, M. Odajiu and A. Tassoni, Induction of hormesis in plants by urban trace metal pollution, Sci. Rep., 2021, 11(1), 20329 CrossRef CAS.
- E. A. Erofeeva, Plant hormesis: The energy aspect of low and high-dose stresses, Plant Stress, 2024, 14, 100628 CrossRef CAS.
- M. P. Gomes, V. S. Richardi, E. M. Bicalho, D. C. Da Rocha, M. A. Navarro-Silva and P. Soffiatti, et al., Effects of Ciprofloxacin and Roundup on seed germination and root development of maize, Sci. Total Environ., 2019, 651, 2671–2678 CrossRef CAS PubMed.
- E. Agathokleous, M. Kitao and E. J. Calabrese, Human and veterinary antibiotics induce hormesis in plants: Scientific and regulatory issues and an environmental perspective, Environ. Int., 2018, 120, 489–495 CrossRef CAS PubMed.
- X. Liu, J. Zhu, J. Liang, F. Li, R. Meng and X. Ma, et al., Ecotoxicological effects of individual and combined treatments of chlortetracycline and oxytetracycline on seed germination and seedling growth of wheat (Triticum aestivum L.), J Environ QualSept, 2025, 54(5), 962–977 CrossRef CAS.
- Y. Bao, C. Pan, D. Li, A. Guo and F. Dai, Stress response to oxytetracycline and microplastic-polyethylene in wheat (Triticum aestivum L.) during seed germination and seedling growth stages, Sci. Total Environ., 2022, 806, 150553 CrossRef CAS PubMed.
- F. Liu, G. G. Ying, R. Tao, J. L. Zhao, J. F. Yang and L. F. Zhao, Effects of six selected antibiotics on plant growth and soil microbial and enzymatic activities, Environ. Pollut., 2009, 157(5), 1636–1642 Search PubMed.
- M. Carballo, A. Rodríguez and A. De La Torre, Phytotoxic Effects of Antibiotics on Terrestrial Crop Plants and Wild Plants: A Systematic Review, Arch. Environ. Contam. Toxicol., 2022, 82(1), 48–61 CrossRef CAS PubMed.
- Y. Liu, Z. Tao, H. Lu, S. Li, C. Hu and Z. Li, Electrochemical properties of roots determine antibiotic adsorption on roots, Front. Plant Sci., 2023, 14, 930632 CrossRef CAS PubMed.
- E. Richter, S. Berkner, I. Ebert, B. Förster, N. Graf and M. Herrchen, et al., Results of extended plant tests using more realistic exposure scenarios for improving environmental risk assessment of veterinary pharmaceuticals, Environ. Sci. Eur., 2016, 28(1), 22 CrossRef PubMed.
- C. C. Serdar, M. Cihan, D. Yücel and M. A. Serdar, Sample size, power and effect size revisited: simplified and practical approaches in pre-clinical, clinical and laboratory studies, Biochem. Med., 2021, 31(1), 27–53 CrossRef PubMed.
- D. López-González, D. Ledo, L. Cabeiras-Freijanes, M. Verdeguer, M. Reigosa and A. Sánchez-Moreiras, Phytotoxic Activity of the Natural Compound Norharmane on Crops, Weeds and Model Plants, Plants, 2020, 9(10), 1328 CrossRef PubMed.
- B. J. Burgess, M. C. Jackson and D. J. Murrell, Are experiment sample sizes adequate to detect biologically important interactions between multiple stressors?, Evol. Ecol., 2022, 12(9), e9289 CrossRef PubMed.
- H. Zhang, D. Zheng, C. Hu, W. Cheng, P. Lei and H. Xu, et al., Certain Tomato Root Exudates Induced by Pseudomonas stutzeri NRCB010 Enhance Its Rhizosphere Colonization Capability, Metabolites, 2023, 13(5), 664 CrossRef CAS PubMed.
- G. H. LeFevre, R. M. Hozalski and P. J. Novak, Root Exudate Enhanced Contaminant Desorption: An Abiotic Contribution to the Rhizosphere Effect, Environ. Sci. Technol., 2013, 47(20), 11545–11553 CrossRef CAS PubMed.
- P. Vivanco, J. A. Oliveira and I. Martín, Optimal germination conditions for monitoring seed viability in wild populations of fescues, Span. J. Agric. Res., 2021, 19(3), e0804 CrossRef.
- F. Vahabinia, H. Pirdashti and E. Bakhshandeh, Environmental factors' effect on seed germination and seedling growth of chicory (Cichorium intybus L.) as an important medicinal plant, Acta Physiol. Plant., 2019, 41(2), 27 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |





