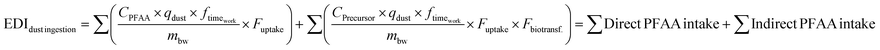Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Per- and polyfluoroalkyl substances in e-waste dust†
Chunjie Xiaa,
William A. Stubbings b,
Linh V. Nguyenc,
Kevin Romanaka,
Liisa Jantunend,
Lisa Melymuk
b,
Linh V. Nguyenc,
Kevin Romanaka,
Liisa Jantunend,
Lisa Melymuk eg,
Victoria Arrandalefc,
Miriam L. Diamond
eg,
Victoria Arrandalefc,
Miriam L. Diamond gh and
Marta Venier
gh and
Marta Venier *a
*a
aO'Neill School of Public and Environmental Affairs, Indiana University, Bloomington, IN, USA. E-mail: mvenier@iu.edu
bGeography, Earth and Environmental Sciences, University of Birmingham, Birmingham, UK
cOccupational Cancer Research Centre, Ontario Health, Ontario, Canada
dAir Quality Processes Research Section, Environment and Climate Change Canada, Egbert, Ontario, Canada
eRECETOX, Faculty of Science, Masaryk University, Brno, Czech Republic
fDalla Lana School of Public Health, University of Toronto, Ontario, Canada
gDepartment of Earth Sciences, University of Toronto, Toronto, Ontario, Canada
hSchool of the Environment, University of Toronto, Toronto, Ontario, Canada
First published on 13th May 2026
Abstract
Per- and polyfluoroalkyl substances (PFAS) have been used in many applications, including electronic products, but little is known about the presence of PFAS in e-waste facilities, especially in North America. In this study, we investigated 87 legacy and novel PFAS in indoor dust samples (n = 19) from two Canadian e-waste dismantling facilities. PFAS were detected in all samples in the range of 364–6090 ng g−1 for Σ64PFAS with a median of 1150 ng g−1. Polyfluoroalkyl phosphate esters (PAPs) were generally the most abundant group of compounds detected and ultrashort PFAS were detected in dust for the first time. After hydrolysis, the concentrations of 3 fluorotelomer alcohols (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH, 8
2 FTOH, 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH and 10
2 FTOH and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH) increased ∼100 times, and those of 3 perfluorooctane sulfonamido ethanol (FASEs; MEFBSE, MeFOSE and EtFOSE) by 20, 20 and 5 times, respectively. After the direct total oxidizable precursor (dTOP) assay, used here in dust for the first time, the levels of perfluoroalkyl carboxylic acids (PFCAs) increased by up to 290 times. These results suggest that there are significant amounts of perfluoroalkyl acid (PFAA) precursors in the e-waste dust samples. Estimated worker exposure via dust ingestion exceeded the US EPA chronic reference dosage for perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) in the intermediate and high-risk scenarios. The presence of PFAS in dust from e-waste electrical and electronic dismantling facilities is a serious concern in terms of both occupational exposure and the risks associated with environmental release from recycling facilities.
2 FTOH) increased ∼100 times, and those of 3 perfluorooctane sulfonamido ethanol (FASEs; MEFBSE, MeFOSE and EtFOSE) by 20, 20 and 5 times, respectively. After the direct total oxidizable precursor (dTOP) assay, used here in dust for the first time, the levels of perfluoroalkyl carboxylic acids (PFCAs) increased by up to 290 times. These results suggest that there are significant amounts of perfluoroalkyl acid (PFAA) precursors in the e-waste dust samples. Estimated worker exposure via dust ingestion exceeded the US EPA chronic reference dosage for perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) in the intermediate and high-risk scenarios. The presence of PFAS in dust from e-waste electrical and electronic dismantling facilities is a serious concern in terms of both occupational exposure and the risks associated with environmental release from recycling facilities.
Environmental significanceElectronic waste recycling is a rapidly expanding industrial activity, yet its role as a source of per- and polyfluoroalkyl substances (PFAS) exposure remains poorly understood. This gap is critical because PFAS are persistent, mobile, and toxic, and e-waste facilities represent a direct exposure pathway for workers and a potential source of environmental release. By measuring 87 legacy and emerging PFAS in dust from e-waste dismantling facilities and applying hydrolysis and the dTOP assay, this study reveals large, previously unrecognized reservoirs of PFAS precursors that can transform into regulated perfluoroalkyl acids. These findings indicate that e-waste recycling is an underappreciated contributor to PFAS exposure in a globally growing waste stream. |
Introduction
E-waste is electric and electronic waste that is discarded after its use has ended. The definition of e-waste is broad, encompassing any device that relies on electrical power or batteries for operation. Examples vary widely across sectors, including household appliances, information technology (IT) and telecommunications equipment, consumer electronics, medical devices, and control systems. These products contain numerous intentionally added chemicals designed to impart specific functional properties such as flame resistance, thermal stability, electrical insulation, lubrication, and surface performance. Consequently, handling and recycling e-waste introduces potential occupational and environmental exposure to complex chemical mixtures.Characterizing chemicals directly in intact e-waste materials presents major challenges. Individual devices are highly heterogeneous, consisting of multiple polymers, coatings, circuit boards, adhesives, and composite materials. The composition varies across manufacturers, product generations, and components even within a single item. Bulk sampling therefore does not represent realistic exposure and cannot easily be standardized across studies.
Moreover, workers are typically not exposed to intact products but to materials released during dismantling and processing. During recycling operations, devices are manually or mechanically disassembled into components such as cables, casings, microchips, and screens. This process releases particles originating both from accumulated internal debris and from abrasion and fragmentation of materials. The resulting e-waste dust becomes a mixed matrix integrating emissions from multiple components and products. Because this dust is airborne, settles on surfaces, and can be ingested or inhaled by workers, it represents the primary exposure pathway during recycling activities. Even in controlled facilities with ventilation and personal protective equipment, exposure to e-waste dust can be substantial;1,2 in informal recycling environments, with no protective equipment for workers or the environment, exposures can be significantly higher.3
One of the inherent risks of handling e-waste is the presence of numerous chemicals added to various components to impart specific properties. Other risks are not chemical and include injuries and safety hazards. Several studies showed that workers in e-waste recycling facilities are exposed to toxic chemicals that could lead to significant health issues, including neurological damage, endocrine disruption, and an increased risk of cancer.4–7 Additionally, these chemicals pose environmental risks, contributing to environmental contamination.3
The widespread presence of flame retardants (FRs) in e-waste has been documented before.1,8–10 We have previously reported the presence in e-waste dust of 100+ compounds across different categories including brominated and organophosphate ester flame retardants, plasticizers and novel compounds, including one alternative FR – 2,4,6-tris(2,4,6-tribromophenoxy)-1,3,5-triazine- and one antioxidant – tri(2,4-di-t-butylphenyl) phosphate.1,9,11,12
However, little information is available on the presence of per- and polyfluoroalkyl substances (PFAS) in e-waste dust. PFAS are used in the manufacturing process in two ways: they are used in factory infrastructure such as refrigerants, lubricants, and structural materials, and in equipment, such as chemical containers that handle chemicals, ensuring stability and preventing leaks or contamination. During production, they are used during the photolithographic phase of incising the silicon base of microchips with the relevant geometrical shapes and for etching to remove unnecessary parts of the silicon wafer after photolithography. They are also used as release agents in the production of molded plastic parts, which can leave residues on the plastic. Environmental contamination from PFAS during microchip production is well documented,13 for example in Taiwan14 and in the US.15 What happens to the products once they reach their end-of-life and the environmental impact of their recycling are less understood or documented.
Only three studies have reported PFAS in e-waste dust in South China16,17 and one review paper summarized the sources, occurrence and health risks of PFAS arising from the manufacture and disposal of electric and electronic products.18 Zhang et al. found that PFAS exposure is associated with adverse health outcomes in the elderly population living around e-waste dismantling sites.19
This lack of information on the presence of PFAS at the end-of-life stage is troubling, considering that e-waste is the fastest-growing solid waste stream in the world.3 In 2022, a record 62 billion kg of e-waste was produced globally, up 82% from 2010.3 Of this, about 7.2 million tonnes were produced in the US alone, corresponding to about 20 kg per person per year. In the US, no federal legislation currently exists as e-waste is regulated at the state level, which has led to a lack of regulatory homogeneity.20 In Canada, e-waste is similarly regulated at the provincial level according to the Extended Producer Responsibility (EPR) model.3 In the EU, the Waste Electrical and Electronic Equipment (WEEE) Directive requires the separate collection and proper treatment of WEEE and sets targets for its collection, recovery and recycling. In 2011 China, which suspended the import of e-waste in 2000, implemented a licensing system for the disposal of e-waste that also requires producers and importers to pay into a fund for its collection and disposal.
The presence of PFAS in e-waste also needs to be considered in light of the increased demand for electric and electronic materials and recent economic initiatives. Currently, 75% of semiconductor manufacturing and research occurs in East Asia. However, manufacturing is poised to expand in the US and the EU as they are making significant investments to boost the semiconductor industry with the goal of bringing up to 20% of the production to their respective regional markets. For example, in 2022, the US government launched the CHIPS and Science Act with significant investments in semiconductor manufacturing and research.21 Thus, a better understanding of the environmental impact of such a shift is warranted.
The purpose of this study was to assess the levels and profiles of PFAS in e-waste dust in North America for the first time. For this purpose, opportunistic samples from two facilities in Canada were analyzed for PFAS. In addition to direct targeted analysis, samples were also subjected to the direct total oxidizable precursors assay (dTOP), in which oxidation reagents are applied directly to the sample instead of to the extracts after drying,22,23 and to hydrolysis, which can free chemically bound fluorotelomer alcohols (FTOHs) and other neutral PFAS, to quantify precursors and compounds that are not captured using solvent extraction. Occupational exposure to PFAS from e-waste dust ingestion was also estimated for three exposure scenarios.
Materials and methods
Sample collection
Dust samples (n = 19) were collected from two facilities near Toronto, Canada.9,24 Details about these facilities were reported previously9,24 and only a brief summary is included here (Table S1). In the first facility, dust samples were collected in pre-cleaned nylon socks attached to the vacuum cleaner from the workshop floor (n = 9), work benches (n = 4), and WEEE sorting bins (n = 5) from the main dismantling hall. Dust from the bins was collected directly from the surface of the WEEE contained in the bin. The WEEE sorting bins from which we collected dust samples were mostly made of cardboard. Some bins used for storage after sorting were made of black plastic material, although the bins themselves were not vacuumed directly. In the second facility, one composite sample of dust from floor sweepings was collected.24 All samples were wrapped in clean aluminum foil, sealed, labelled, and subsequently stored at −20 °C until analysis. Before extraction, dust samples sieved through a 500 µm sieve to remove coarse materials, i.e. fibers and debris, were less likely to be ingested and inhaled by workers.Sample extraction
Detailed extraction and cleanup procedures for dust samples have been reported previously.25,26 Briefly, 0.1 g dust samples were weighed into a pre-cleaned 15 mL polypropylene (PP) tube, spiked with surrogate standards (SS) and extracted twice with 3 mL of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 hexane/isopropyl alcohol, followed by 3 mL of 1
1 hexane/isopropyl alcohol, followed by 3 mL of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 methanol/acetonitrile. For each extraction cycle, the sample was sonicated for 30 min and then centrifuged at 3000 g for 5 min. The supernatants were combined, reduced in volume to ∼5 mL, and cleaned up with ∼100 mg Envi-Carb activated carbon by vortexing for 1 min and centrifuging at 3000 g for 5 min. The resulting sample was concentrated to ∼500 µL under nitrogen and filtered using a centrifuge filter (VWR, modified Nylon, 0.2 µm, 500 µL, part No. 82031-358; see the SI for information on filter testing). The filtrate was then transferred into a 1-mL polypropylene vial and spiked with internal standards (IS) with a final sample volume of 1 mL. The samples were then stored at −20 °C until analysis – see Tables S2 and S3 for a list of IS and SS.
1 methanol/acetonitrile. For each extraction cycle, the sample was sonicated for 30 min and then centrifuged at 3000 g for 5 min. The supernatants were combined, reduced in volume to ∼5 mL, and cleaned up with ∼100 mg Envi-Carb activated carbon by vortexing for 1 min and centrifuging at 3000 g for 5 min. The resulting sample was concentrated to ∼500 µL under nitrogen and filtered using a centrifuge filter (VWR, modified Nylon, 0.2 µm, 500 µL, part No. 82031-358; see the SI for information on filter testing). The filtrate was then transferred into a 1-mL polypropylene vial and spiked with internal standards (IS) with a final sample volume of 1 mL. The samples were then stored at −20 °C until analysis – see Tables S2 and S3 for a list of IS and SS.
Direct TOP assays and hydrolysis
The dTOP assay and hydrolysis followed a protocol previously reported with some modifications.22,23,27,28 For the dTOP assay, 50 mL of a freshly prepared oxidation solution made with 200 mM K2S2O8 and 500 mM NaOH in water was added to the dust sample (∼30 mg) directly, spiked with labeled perfluoroalkyl acids (PFAAs) as surrogate standards (50 ng each, Tables S2 and S3) and incubated at 85–90 °C for 7 h. The sample was then cooled to room temperature, adjusted to pH = 4.0 with formic acid, and cleaned up on an Oasis WAX cartridge following the same protocol reported above and described by Wu et al. (2021).29 The efficiency of the dTOP oxidation was verified by testing the efficiency of removal of M2-8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS, an isotopically labeled standard spiked in the samples at the beginning of the process. For the test, 10 µl of a 10 µg L−1 solution of M2-8
2 FTS, an isotopically labeled standard spiked in the samples at the beginning of the process. For the test, 10 µl of a 10 µg L−1 solution of M2-8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 fluorotelomer sulfonate (M2-8
2 fluorotelomer sulfonate (M2-8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS) were spiked into the vials (n = 3), and the dTOP assay procedure was applied. No M2-8
2 FTS) were spiked into the vials (n = 3), and the dTOP assay procedure was applied. No M2-8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS was detected in the samples, indicating that the analyte was fully oxidized. Additionally, the molar yields of the generated PFCAs were comparable to those reported for 8
2 FTS was detected in the samples, indicating that the analyte was fully oxidized. Additionally, the molar yields of the generated PFCAs were comparable to those reported for 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS by Tsou et al. (2023) – see Table S4.30
2 FTS by Tsou et al. (2023) – see Table S4.30
For the hydrolysis treatment, 0.5 mL of 1 M NaOH solution in methanol/water (90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) was added to a 15 mL glass vial containing ∼30 mg of dust and spiked with 40 ng each of the surrogate standards (see Table S2). The vial was vortexed for 1 min and the sample was then placed in an oven at 60 °C for 16 h. After the vial was cooled to room temperature, the solution was transferred to a 15 mL pre-cleaned PP vial, and 0.6 mL of a 1
10) was added to a 15 mL glass vial containing ∼30 mg of dust and spiked with 40 ng each of the surrogate standards (see Table S2). The vial was vortexed for 1 min and the sample was then placed in an oven at 60 °C for 16 h. After the vial was cooled to room temperature, the solution was transferred to a 15 mL pre-cleaned PP vial, and 0.6 mL of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of methyl tert-butyl ether/n-hexane and 2 mL of LC-MS grade water was added. The samples were shaken for 30 min, and the bottom aqueous layer was removed with a glass pipette. Baked anhydrous Na2SO4 was added to remove the water in the sample until the organic layer became clear. Finally, the extracts were transferred into a 1 mL PP vial, spiked with 100 ng internal standards (see Tables S2 and S3 for a complete list) and then stored at −20 °C until GC-MS analysis. Note that three samples (one collected from the bench and two from bins) were used up after the original extraction described above, so no material was left for hydrolysis and dTOP assay.
1 mixture of methyl tert-butyl ether/n-hexane and 2 mL of LC-MS grade water was added. The samples were shaken for 30 min, and the bottom aqueous layer was removed with a glass pipette. Baked anhydrous Na2SO4 was added to remove the water in the sample until the organic layer became clear. Finally, the extracts were transferred into a 1 mL PP vial, spiked with 100 ng internal standards (see Tables S2 and S3 for a complete list) and then stored at −20 °C until GC-MS analysis. Note that three samples (one collected from the bench and two from bins) were used up after the original extraction described above, so no material was left for hydrolysis and dTOP assay.
Instrumental analyses
Ionic PFAS, i.e., perfluorocarboxylic acids (PFCAs), perfluorosulfonic acids (PFSAs), fluorotelomer sulfonic acids (FTSAs), fluorotelomer carboxylic acids (FTCAs), polyfluoroalkyl phosphate esters (PAPs), perfluoroalkane sulfonamides (FASAs), perfluoroalkane sulfonamido acetic acids (FASAAs), perfluoroalkyl phosphinic acids (PFPiAs), were analyzed by ultrahigh performance liquid chromatography (LC-MS/MS) coupled to a triple quadrupole mass spectrometer (Agilent 1290 Infinity II UPLC 6470 QQQ-MS), operated in negative electrospray ionization mode (ESI-). Ionic PFAS and PAPs were quantitated using an Acquity BEH C18 1.7 µm, 2.1 id × 50 mm column (manufacturer), while ultra-short chain PFAS (trifluoroacetic acid (TFA), perfluoropropanoic acid (PFPrA), trifluoromethanesulfonic acid (TFMS), pentafluoroethanesulfonic acid (PFEtS)) were analyzed on a Raptor Polar X 2.7 µm, 50 × 2.1 mm column (Restek, Bellefonte, PA) – see Table S5 for instrumental details. Gas chromatography mass spectrometry (GC-MS) operating in positive chemical ionization (PCI) mode (Agilent 7890 GC-5977 B PCI-MS) was used to quantify parachlorobenzotrifluoride (PCBTF), perfluoroalkyl acetates (FTOAcs), perfluoroalkane sulfonamido ethanols (FASEs), fluorotelomer alcohols (FTOHs), fluorotelomer acrylates (FTAcs), fluorotelomer methacrylates (FTMAcs) – see Table S6. Detailed instrumental parameters are listed in Tables S5–S8.QA/QC
For the LC-MS/MS and GC-MS analyses, four procedural blanks and four matrix spikes consisting of 5 ng for each targeted PFAS were processed to evaluate background contamination from laboratory operations (see Table S9) and the performance of our methods. The recoveries of surrogate standards ranged between 70% and 114% (see Table S10). The recoveries of matrix spikes were in the range 67% to 126% for the original extraction and 58% to 131% for the dTOP assay after SS correction. The only exceptions were perfluoropropanesulfonic acid (PFPrS) for the dTOP (29.8%) and monoPAPs, which were then excluded from this study. The MDLs (ng g−1) were defined as 3 times the standard deviation of the blank level or the amount of chemical generating a signal-to-noise ratio of 5 if the compound was not detected in procedural blanks divided by the average sample weight (Table S9). All data were blank corrected, but only results for dTOP and hydrolysis analysis were surrogate corrected.As part of the QA/QC procedures, we also verified that none of the analytes were adhering to the filters. For this test, each filter (n = 3) was spiked with SS and matrix spike compounds and eluted with 0.5 mL methanol; the filtrate was transferred into a pre-cleaned 1-mL LC PP vial, the filter was washed with 0.3 mL of methanol and then transferred to an LC vial, spiked with IS and analyzed. The recoveries of analytes in this test ranged from 70% to 115% (see Table S11).
Dust ingestion estimates
We estimated the daily PFAA intake via dust ingestion by workers in the WEEE facilities for both direct and indirect intake (via biotransformation of PFAA precursors) using our dust PFAS data and the parameters described in Table S12.25,31 No other exposure pathways, including inhalation were calculated due to lack of available data. Dust ingestion was calculated for three exposure scenarios (low, intermediate and high using 5th, median, and 95th percentile concentrations) using the following equation:where CPFAA is the concentration of compounds with DF > 50% in dust (ng g−1; the 5th percentile was used for the low scenario, the median for the intermediate scenario, and the 95th percentile for the high scenario), qdust is the dust ingestion rate (mg per day; 20 mg per day, 40 mg per day, and 60 mg per day were used for low, intermediate and high scenarios, respectively),32 mbw is the body weight (kg; 70 kg), ftimework is the fraction of time spent working in the facility (unitless; 0.238), Fuptake is the gastrointestinal uptake fraction (unitless; 0.66, 0.8, and 0.91 were used for low, intermediate and high scenarios, respectively), and Fbiotransf is the biotransformation factor of precursor compounds to PFAAs (unitless), which were estimated from numerous in vivo and in vitro studies, where FTCAs were the metabolites of precursors, e.g. FTOHs.31,33
Detailed information on the parameters used for individual PFAS for estimated daily intakes (EDIs) is provided in Table S12. Compounds in Table S12 with a value of Fbiotransf were considered as precursors. For example, the assumed biotransformation fractions for FTOHs and diPAPs for the three scenarios were 0.0006/0.0012 (low), 0.003/0.006 (intermediate), and 0.01/0.02 (high), respectively.34Fbiotransf for all other precursors are listed in Table S12. Only compounds with DF > 50% were included for the EDI calculation.
Results and discussion
PFAS levels in e-waste dust
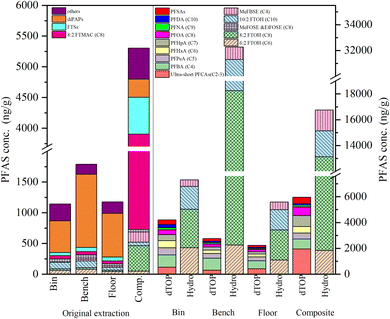
PFAS levels in e-waste dust samples are reported in Table S13 and in Fig. 1, together with the contributions of the different sub-categories of PFAS. Sixty-four of the 87 targeted PFAS compounds were detected in at least one dust sample. Total PFAS (Σ64PFAS) concentrations ranged from 364–6090 ng g−1 with a median of 1150 ng g−1, similar to levels in residential indoor dust samples,25,26,35–39 especially those reported by Zhang et al., who compared e-waste and residential dust.16 In these e-waste dust samples, Σ64PFAS levels were about 2–3 orders of magnitude lower than those of other pollutants including polybrominated diphenyl ethers (PBDEs), which had a median of 120![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng g−1, organophosphate esters (OPEs) with a median of 110
000 ng g−1, organophosphate esters (OPEs) with a median of 110![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng g−1, and non-brominated flame retardants (NBFRs) with a median of 62
000 ng g−1, and non-brominated flame retardants (NBFRs) with a median of 62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng g−1 in the same samples.9 For reference, PBDEs, OPEs, and other FRs were 1–2 orders of magnitude higher than in dust from Toronto residences.9 The lower levels of PFAS compared to FRs and the differences with residential levels might be due to different sources (electronics in e-waste facilities vs. a multitude of products containing PFAS in homes),16 higher amounts of FRs than PFAS used in products, and the presence of precursors or PFAS compounds not accounted for.
000 ng g−1 in the same samples.9 For reference, PBDEs, OPEs, and other FRs were 1–2 orders of magnitude higher than in dust from Toronto residences.9 The lower levels of PFAS compared to FRs and the differences with residential levels might be due to different sources (electronics in e-waste facilities vs. a multitude of products containing PFAS in homes),16 higher amounts of FRs than PFAS used in products, and the presence of precursors or PFAS compounds not accounted for.
PAPs, particularly polyfluoroalkyl phosphoric acid diesters (diPAPs) including 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 diPAP, 6
2 diPAP, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2/8
2/8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 diPAP, 8
2 diPAP, 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 diPAP, and 10
2 diPAP, and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 diPAP, were generally the dominant group of PFAS in e-waste dust samples, contributing 15 to 80% of Σ64PFAS concentrations, except for one composite sample from a second facility (5%), which was similar to what has been found for Canadian household dust.35,38 Sodium bis[2-(N-ethylperfluorooctane-1-sulfonamido)ethyl] phosphate (diSAmPAP), discontinued by 3M in 2000,40 was also detected in all samples, albeit with a low median of 3.9 ng g−1. FTOHs were generally the next dominant group after PAPs, contributing 3.0 to 17.8% of Σ64PFAS concentrations with a median of 122 ng g−1, comparable to the level (152 ng g−1) in the dust samples from homes in Vancouver, Canada.39 In a composite e-waste dust sample from a second facility, 8
2 diPAP, were generally the dominant group of PFAS in e-waste dust samples, contributing 15 to 80% of Σ64PFAS concentrations, except for one composite sample from a second facility (5%), which was similar to what has been found for Canadian household dust.35,38 Sodium bis[2-(N-ethylperfluorooctane-1-sulfonamido)ethyl] phosphate (diSAmPAP), discontinued by 3M in 2000,40 was also detected in all samples, albeit with a low median of 3.9 ng g−1. FTOHs were generally the next dominant group after PAPs, contributing 3.0 to 17.8% of Σ64PFAS concentrations with a median of 122 ng g−1, comparable to the level (152 ng g−1) in the dust samples from homes in Vancouver, Canada.39 In a composite e-waste dust sample from a second facility, 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTMAc contributed 52% of Σ64PFAS concentration, followed by FTSs (10%) and FTOHs (8.5%). In one bench sample, PFBS was the second dominant analyte contributing 20% to the total PFAS, exceeded only by 6
2 FTMAc contributed 52% of Σ64PFAS concentration, followed by FTSs (10%) and FTOHs (8.5%). In one bench sample, PFBS was the second dominant analyte contributing 20% to the total PFAS, exceeded only by 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 diPAP, which accounted for 55% of the total PFAS. Although both facilities received mixed e-waste, this difference might reflect different types of electronic waste handled at each facility: PFBS is a short-chain alternative to long-chain fluorinated compounds, e.g., PFOS, mainly used in chromium plating, aqueous film-forming foams (AFFFs), fluoropolymer processing, and surface treatment,41 while 8
2 diPAP, which accounted for 55% of the total PFAS. Although both facilities received mixed e-waste, this difference might reflect different types of electronic waste handled at each facility: PFBS is a short-chain alternative to long-chain fluorinated compounds, e.g., PFOS, mainly used in chromium plating, aqueous film-forming foams (AFFFs), fluoropolymer processing, and surface treatment,41 while 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTMAc is used as a reactive methacrylate monomer in the production of side-chain fluorinated polymers.
2 FTMAc is used as a reactive methacrylate monomer in the production of side-chain fluorinated polymers.
Ultra-short chain PFAA concentrations, including TFA, PFPrA, TFMS, and PFPrS, reported in dust samples for the first time here, represented 1 to 56% of total PFAS and 6 to 92% of PFAAs, likely generated from degradation of precursors such as neutral PFAS. TFA is an environmentally persistent end-product of fluorinated refrigerants, pesticides, and other PFAS and has recently been detected at high concentrations in human and dust samples.42–44 Because TFA has historically not been included in the targeted PFAS list in most previous studies, its large contribution here indicates that indoor PFAS exposure may have been underestimated in studies excluding ultra-short chain PFAAs.
Hydrolysis
Hydrolysis treatment can free chemically bound FTOHs and other neutral PFAS from impurities or other precursors, e.g., side-chain fluorinated polymers used in electronics production.45 For these dust samples, hydrolysis treatment resulted in a substantial increase in the concentrations of six neutral PFAS (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH, 8
2 FTOH, 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH, 10
2 FTOH, 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH, MeFBSE, MeFOSE, and EtFOSE), with post-hydrolysis concentrations ranging from 1220 to 75
2 FTOH, MeFBSE, MeFOSE, and EtFOSE), with post-hydrolysis concentrations ranging from 1220 to 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 ng g−1. In contrast, the combined concentrations of these compounds in the original extracts ranged from 63.1 to 726 ng g−1, indicating significantly lower levels prior to hydrolysis (p < 0.001; see Fig. 1, Tables S13 and S14). FTOHs, especially 8
800 ng g−1. In contrast, the combined concentrations of these compounds in the original extracts ranged from 63.1 to 726 ng g−1, indicating significantly lower levels prior to hydrolysis (p < 0.001; see Fig. 1, Tables S13 and S14). FTOHs, especially 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH, increased by up to ∼100 times, and MeFOSE, MeFBSE, and EtFOSE increased by ∼20, 20 and 5 times, respectively, similarly to what we found in school uniforms27 and fast-food packaging materials that contained PFAS.28
2 FTOH, increased by up to ∼100 times, and MeFOSE, MeFBSE, and EtFOSE increased by ∼20, 20 and 5 times, respectively, similarly to what we found in school uniforms27 and fast-food packaging materials that contained PFAS.28
Conversely, FT(M)Acs were not detected in any samples after hydrolysis, despite the fact that they were present in the original extraction. We speculate that they contributed to the increase in FTOHs since it has previously been shown that FT(M)Acs could be converted to FTOHs.27,29 PCBTF, an aromatic PFAS previously reported in bridge painting46 and silicone wristbands,47 was detected in one floor dust sample after hydrolysis at 4392 ng g−1; this is the first time this compound has been reported in dust samples and we speculate that its source was paint from the building itself.
PAPs can also be converted to FTOHs at the high temperature of the GC inlet (200 °C), albeit at low yields (5.4–14% for 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 diPAP to 6
2 diPAP to 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH; 7.7–9.2% for 8
2 FTOH; 7.7–9.2% for 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 diPAP to 8
2 diPAP to 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTOH).48 Even assuming that precursors like PAPs and FT(M)Acs were transformed to FTOHs at a 100% conversion rate, more than 73% of the increase in neutral PFAS was contributed by unknown precursors, e.g., side-chain fluorinated polymers.27–29
2 FTOH).48 Even assuming that precursors like PAPs and FT(M)Acs were transformed to FTOHs at a 100% conversion rate, more than 73% of the increase in neutral PFAS was contributed by unknown precursors, e.g., side-chain fluorinated polymers.27–29
Direct TOP assay
The dTOP assay can provide useful information on unknown PFAA precursors, as has been shown in different types of matrices, including soil49,50 and fish.22,23 To the authors' knowledge, this assay has never been applied to dust. After the dTOP assay, total PFAAs, comprising PFCAs and PFSAs, increased from 141–1000 ng g−1 (median: 351 ng g−1) to 674–10800 ng g−1 (median: 1240 ng g−1) for all samples (see Fig. 1 and Tables S13–S15). PFCA concentrations increased by an average of approximately 50-fold; however, substantially larger increases were observed for perfluorobutanoic acid (PFBA), perfluoropentanoic acid (PFPeA), perfluorohexanoic acid (PFHxA), with concentrations rising by as much as 290-fold—a trend previously observed in both biota and consumer products.22,27 Notably, none of the neutral PFAS—except perfluorobutane sulfonamide (FBSA), perfluorooctane sulfonamide (FOSA), 2H-perfluoro-2-octenoic acid (FHUEA)—were detected after the dTOP assay. These compounds were likely converted to PFAAs, as they are known precursors of PFAAs. When expressed as moles of fluorine, approximately 50% of the PFCA mass was, on average, attributable to known (targeted) precursors, while the remaining ∼50% was contributed by unidentified precursors. It should be noted that no standardized protocol exists for this assay, and despite the strong QA/QC performance, it is likely that not all precursors were captured, leading to a potential underestimation of their true abundance in dust samples. For example, engineered fluids and electronic coating products may contribute to the fraction attributed to unknown precursors.PFAA exposure via dust ingestion
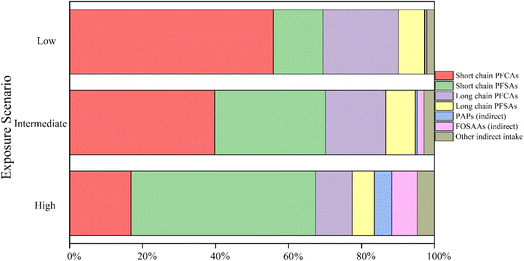
The total estimated PFAA intakes via dust ingestion for workers were 3.90, 36.3, and 280 pg per kg bw per day for the low-, intermediate-, and high-exposure scenarios, respectively (Table 1). These values are comparable to those reported by Gustafsson et al. (2022) (85 and 260 pg per kg bw per day for median and worst-case scenarios)51 and slightly lower than those reported by Chang et al. (2025) (77, 190, and 420 pg per kg bw per day for the low-, intermediate-, and high-exposure scenarios, respectively), likely reflecting differences in the targeted analyte list and products handled at specific locations.37 Direct intake of PFCAs and PFSAs contributed 97%, 94%, 83% to the total PFAA intake for the low-, intermediate-, and high-exposure scenarios, respectively (Table 1 and Fig. 2). In particular, PFPrA, perfluorobutanesulfonic acid (PFBS), perfluorooctane sulfonate (PFOS), and perfluorooctanoic acid (PFOA) were the major contributors to the direct intake, similar to findings from our previous study on indoor dust.25 If one considers only PFOA and PFOS, for which reference exposure doses exist, the EDIs via dust ingestion were 0.55, 3.72 and 19.0 pg per kg bw per day for PFOA and 0.29, 3.86, and 41.0 pg per kg bw per day for PFOS for the low, intermediate and high exposure scenarios (Table 1). EDIs for the intermediate- and high-exposure scenarios exceeded the U.S. EPA chronic reference dose for PFOA (1.5 pg per kg bw per day), whereas for PFOS, only the high-exposure scenario EDIs exceeded the U.S. EPA chronic reference dose (7.9 pg per kg bw per day).52| Intake (pg per kg bw per day) | Exposure scenario | ||
|---|---|---|---|
| Low | Intermediate | High | |
| Direct PFAA intake | 3.79 | 34.4 | 234 |
| Indirect PFAA intake | 0.11 (3%) | 1.92 (5.3%) | 46.3 (17%) |
| Total EDI (Σ38PFAS) | 3.90 | 36.3 | 280 |
| EDIPFOA | 0.55 | 3.72 | 19.0 |
| EDIPFOS | 0.29 | 3.86 | 41.0 |
While direct intake refers to the intake of stable PFAAs (e.g., PFCAs and PFSAs), indirect intake refers to the intake of PFAA precursors, which can then biotransform into PFAAs in vivo. For example, PAPs can be biotransformed to PFCAs in vivo, as shown for rats and fish.54–57 Previous studies also showed that FOSA and N-EtFOSE can be converted to PFOS both in vitro and in vivo.58–60 The calculated PFOS indirect intake resulting from precursors (FOSA, FOSEs & FOSAAs) biotransformation represented 36%, 43%, and 71% of total EDI for the low, intermediate, and high exposure scenarios, respectively, consistent with earlier studies reporting 69% for an intermediate scenario (Winkens et al., 2018)34 and a range of 41–68% (Vestergren et al., 2008).53 Similar contributions were also found for indirect total PFAA intake, with FOSAAs being the major contributors, especially in the high-exposure scenario at 7.0%, followed by PAPs and FOSEs at 4.8% and 3.3%, respectively (Fig. 2). This finding is similar to the result from a previous study.33
Implications and limitations
This study confirmed that e-waste dust contains significant amounts of target PFAS and in particular PAPs. Additionally, the hydrolysis and dTOP assays revealed the presence of a large amount of unknown precursors that could be transformed into PFAA (e.g. side-chain fluorinated polymers), including 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2FTOH, which in turn can be converted to PFOA, and other long-chain PFCAs that are restricted under Canadian regulations. Hence, these estimated daily intake values likely underestimate the potential PFAS exposure from dust for workers at e-waste recycling facilities. Although the use of personal protective equipment (PPE) and good hand hygiene can substantially reduce exposure, the magnitude of this reduction is difficult to quantify, as it depends on user compliance. The estimated daily intakes are based solely on dust ingestion, likely underestimating total occupational PFAS exposure since dermal absorption or inhalation was not included. In particular, inhalation exposure was not evaluated because PFAS concentrations in the indoor air were not measured.
2FTOH, which in turn can be converted to PFOA, and other long-chain PFCAs that are restricted under Canadian regulations. Hence, these estimated daily intake values likely underestimate the potential PFAS exposure from dust for workers at e-waste recycling facilities. Although the use of personal protective equipment (PPE) and good hand hygiene can substantially reduce exposure, the magnitude of this reduction is difficult to quantify, as it depends on user compliance. The estimated daily intakes are based solely on dust ingestion, likely underestimating total occupational PFAS exposure since dermal absorption or inhalation was not included. In particular, inhalation exposure was not evaluated because PFAS concentrations in the indoor air were not measured.
PFAS are the latest group of compounds found in e-waste dust after FRs, plasticizers, and antioxidants. Considering that the production of e-waste is on the rise, the presence of many toxic chemicals is troubling. This is a concern on many levels: for workers in e-waste recycling facilities, including formal and informal facilities1 and for the environment, as improperly disposed e-waste (i.e. in open air landfills) can release these toxic pollutants for decades, increasing the pollution burden, particularly in low income countries where a large percentage of e-waste is shipped.3
We recognize that this dataset is limited in size and provenance, somewhat limiting the ability to draw global conclusions. Nevertheless, given the tight interconnections between the USA, Canada, and Mexico and the global market, we maintain that the results should provide valuable insights into the presence of PFAS in e-waste facilities in North America. Further studies should explore more in depth the relationship between products being dismantled and PFAS levels in dust, which was not possible here due to the opportunistic nature of this work, and the relationship between processed e-waste and the resulting dust. The settled dust analyzed here represents time-integrated accumulation from heterogeneous dismantling activities rather than a quantifiable fraction generated per unit mass of e-waste and therefore cannot be used to derive emission factors or facility-scale PFAS releases.
With the increasingly fast turnround of electric and electronic products and their pervasiveness in our daily lives, the generation of e-waste will rise dramatically over the next decades. The yearly annual growth rate of the e-waste industry is about 8%, with an expected increase from 53.66 billion in 2024 to $58.1 billion in 2025.61 Additionally, the anticipated increase in the production of electronic components in North America and Europe will inevitably lead to an increased risk of PFAS releases to the environment during manufacturing, underscoring the urgent need to phase out PFAS and adopt safer alternatives.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are included as part of the supplementary information (SI). Supplementary information: sample details; targeted PFAS analytes, surrogate and internal standards; compound specific instrument parameters; instrument parameters for LC-MS/MS and GC/MS; blanks, matrix spikes and MDLs; surrogate recoveries; data for original extraction, dTOP assay and hydrolysis. See DOI: https://doi.org/10.1039/d5em01075f.Acknowledgements
We thank the Ontario Ministry of Labor Research Opportunity Program. LM acknowledges support from the RECETOX Research Infrastructure (No. LM2023069) financed by the Ministry of Education, Youth and Sports for supportive background, and the European Union’s Horizon 2020 research and innovation program under grant agreement No. 857560 (CETOCOEN Excellence). This publication reflects only the author's view, and the European Commission is not responsible for any use that may be made of the information it contains.References
- L. V. Nguyen, M. L. Diamond, M. Venier, W. A. Stubbings, K. Romanak, L. Bajard, L. Melymuk, L. M. Jantunen and V. H. Arrandale, Environ. Int., 2019, 129, 95–104 CrossRef CAS PubMed.
- S. Gravel, J. Lavoue, B. Bakhiyi, M. L. Diamond, L. M. Jantunen, J. Lavoie, B. Roberge, M. A. Verner, J. Zayed and F. Labreche, Environ. Int., 2019, 128, 244–253 CrossRef CAS PubMed.
- C. P. Baldé, R. Kuehr, T. Yamamoto, R. McDonald, E. D'Angelo, S. Althaf, G. Bel, O. Deubzer, E. Fernandez-Cubillo, V. Forti, V. Gray, S. Herat, S. Honda, G. Iattoni, D. S. Khetriwal, V. L. d. Cortemiglia, Y. Lobuntsova, I. Nnorom, N. Pralat and M. Wagner, International Telecommunication Union (ITU) and United Nations Institute for Training and Research (UNITAR), 2024 Search PubMed.
- J. Caravanos, E. Clark, R. Fuller and C. Lambertson, J. Health Pollut., 2011, 1, 16–25 CrossRef.
- K. Grant, F. C. Goldizen, P. D. Sly, M. N. Brune, M. Neira, M. van den Berg and R. E. Norman, Lancet Glob. Health, 2013, 1, e350–e361 CrossRef PubMed.
- S. Lin, M. U. Ali, C. Zheng, Z. Cai and M. H. Wong, J. Hazard. Mater., 2022, 426, 127792 CrossRef CAS PubMed.
- B. Tansel, J. Environ. Manage., 2022, 316, 115291 CrossRef CAS PubMed.
- J. Guo, W. A. Stubbings, K. Romanak, L. V. Nguyen, L. Jantunen, L. Melymuk, V. Arrandale, M. L. Diamond and M. Venier, Environ. Sci. Technol., 2018, 52, 3599–3607 CrossRef CAS PubMed.
- W. A. Stubbings, L. V. Nguyen, K. Romanak, L. Jantunen, L. Melymuk, V. Arrandale, M. L. Diamond and M. Venier, Sci. Total Environ., 2019, 675, 594–603 CrossRef CAS PubMed.
- M. Venier, W. A. Stubbings, J. Guo, K. Romanak, L. V. Nguyen, L. Jantunen, L. Melymuk, V. Arrandale, M. L. Diamond and R. A. Hites, Environ. Sci. Technol., 2018, 52, 12997–13003 CrossRef CAS PubMed.
- J. H. Guo, W. A. Stubbings, K. Romanak, L. V. Nguyen, L. Jantunen, L. Melymuk, V. Arrandale, M. L. Diamond and M. Venier, Environ. Sci. Technol., 2018, 52, 3599–3607 CrossRef CAS PubMed.
- M. Venier, W. A. Stubbings, J. H. Guo, K. Romanak, L. V. Nguyen, L. Jantunen, L. Melymuk, V. Arrandale, M. L. Diamond and R. A. Hites, Environ. Sci. Technol., 2018, 52, 12997–13003 CrossRef CAS PubMed.
- J. Gluge, M. Scheringer, I. T. Cousins, J. C. DeWitt, G. Goldenman, D. Herzke, R. Lohmann, C. A. Ng, X. Trier and Z. Wang, Environ. Sci. Process Impacts, 2020, 22, 2345–2373 Search PubMed.
- A. Y. Lin, S. C. Panchangam and C. C. Lo, Environ. Pollut., 2009, 157, 1365–1372 CrossRef CAS PubMed.
- P. Jacob, K. A. Barzen-Hanson and D. E. Helbling, Environ. Sci. Technol., 2021, 55, 2346–2356 CrossRef CAS PubMed.
- B. Zhang, Y. He, Y. Huang, D. Hong, Y. Yao, L. Wang, W. Sun, B. Yang, X. Huang, S. Song, X. Bai, Y. Guo, T. Zhang and H. Sun, Environ. Pollut., 2020, 263, 114461 CrossRef CAS PubMed.
- L. Zhang, X. Liu, W. Huang, Y. Yang and D. Chen, Environ. Sci. Technol. Lett., 2024, 11, 1384–1390 CrossRef CAS.
- S. Garg, P. Kumar, V. Mishra, R. Guijt, P. Singh, L. F. Dumée and R. S. Sharma, J. Water Process Eng., 2020, 38, 101683 CrossRef.
- T. Zhang, B. Zhang, X. Bai, Y. Yao, L. Wang, Y. Shu, K. Kannan, X. Huang and H. Sun, Environ. Sci. Technol. Lett., 2019, 6, 133–140 Search PubMed.
- State Legislation on E-Waste, 2024, http://www.e-takeback.org/docs%20open/Toolkit_Legislators/state%20legislation/state_leg_main.htm, accessed October 10, 2024.
- T. W. House, FACT SHEET: CHIPS and Science Act Will Lower Costs, Create Jobs, Strengthen Supply Chains, and Counter China, 2022 Search PubMed.
- S. L. Capozzi, C. Xia, M. Shuwal, G. Zaharias Miller, J. Gearhart, E. Bloom, L. Gehrenkemper and M. Venier, Chemosphere, 2023, 345, 140454 Search PubMed.
- B. Gockener, A. Fliedner, H. Rudel, I. Fettig and J. Koschorreck, Sci. Total Environ., 2021, 782, 146825 CrossRef PubMed.
- M. Ubukata, K. J. Jobst, E. J. Reiner, S. E. Reichenbach, Q. Tao, J. Hang, Z. Wu, A. J. Dane and R. B. Cody, J. Chromatogr. A, 2015, 1395, 152–159 CrossRef CAS PubMed.
- Y. Wu, K. Romanak, T. Bruton, A. Blum and M. Venier, Chemosphere, 2020, 251, 126771 CrossRef CAS PubMed.
- G. Zheng, B. E. Boor, E. Schreder and A. Salamova, Environ. Pollut., 2020, 258, 113714 Search PubMed.
- C. Xia, M. L. Diamond, G. F. Peaslee, H. Peng, A. Blum, Z. Wang, A. Shalin, H. D. Whitehead, M. Green, H. Schwartz-Narbonne, D. Yang and M. Venier, Environ. Sci. Technol., 2022, 56, 13845–13857 Search PubMed.
- H. Schwartz-Narbonne, C. Xia, A. Shalin, H. D. Whitehead, D. Yang, G. F. Peaslee, Z. Wang, Y. Wu, H. Peng, A. Blum, M. Venier and M. L. Diamond, Environ. Sci. Technol. Lett., 2023, 10, 343–349 CrossRef CAS PubMed.
- Y. Wu, G. Z. Miller, J. Gearhart, G. Peaslee and M. Venier, Environ. Pollut., 2021, 268, 115477 CrossRef CAS PubMed.
- K. Tsou, E. Antell, Y. Duan, C. I. Olivares, S. Yi, L. Alvarez-Cohen and D. L. Sedlak, ACS ES&T Water, 2023, 3, 2996–3003 Search PubMed.
- W. A. Gebbink, U. Berger and I. T. Cousins, Environ. Int., 2015, 74, 160–169 CrossRef CAS PubMed.
- USEPA, Exposure Factors Handbook Chapter 5 (Update 2017), 2017, vol. 7 Search PubMed.
- R. Vestergren, I. T. Cousins, D. Trudel, M. Wormuth and M. Scheringer, Chemosphere, 2008, 73, 1617–1624 CrossRef CAS PubMed.
- K. Winkens, G. Giovanoulis, J. Koponen, R. Vestergren, U. Berger, A. M. Karvonen, J. Pekkanen, H. Kiviranta and I. T. Cousins, Environ. Int., 2018, 119, 493–502 CrossRef CAS PubMed.
- U. Eriksson and A. Karrman, Environ. Sci. Technol., 2015, 49, 14503–14511 CrossRef CAS PubMed.
- Y. Zhi, X. Lu, G. Munoz, L. W. Y. Yeung, A. O. De Silva, S. Hao, H. He, Y. Jia, C. P. Higgins and C. Zhang, Environ. Sci. Technol., 2024, 58, 21393–21410 CrossRef CAS PubMed.
- N. Y. Chang, C. M. A. Eichler, E. A. Cohen Hubal, J. D. Surratt, G. C. Morrison and B. J. Turpin, Environ. Sci.:Processes Impacts, 2025, 27, 1654–1670 RSC.
- A. O. De Silva, C. N. Allard, C. Spencer, G. M. Webster and M. Shoeib, Environ. Sci. Technol., 2012, 46, 12575–12582 CrossRef CAS PubMed.
- M. Shoeib, T. Harner, M. W. G and S. C. Lee, Environ. Sci. Technol., 2011, 45, 7999–8005 CrossRef CAS PubMed.
- J. Ao, W. Tang, X. Liu, Y. Ao, Q. Zhang and J. Zhang, J. Hazard. Mater., 2024, 464, 133018 CrossRef CAS PubMed.
- A. Bogdan and AECOM, Perfluorobutane Sulfonic Acid (PFBS) Chemistry, Production, Uses and Environmental Fate in Michigan, Project No. 60560354, 2019 Search PubMed.
- D. C. G. Muir, F. Freeling, S. Nilsson, B. Bugsel, K. Bowles, P. Hobson, L.-M. Toms and J. F. Mueller, Environ. Sci. Technol. Lett., 2025, 12, 1411–1417 CrossRef CAS.
- B. Wang, Y. Yao, Y. Wang, H. Chen and H. Sun, Environ. Sci. Technol., 2021, 56, 10 Search PubMed.
- G. Zheng, S. M. Eick and A. Salamova, Environ. Sci. Technol., 2023, 57, 15782–15793 CrossRef CAS PubMed.
- P. Jacob and D. E. Helbling, Environ. Sci. Technol., 2023, 57, 12819–12828 CrossRef CAS PubMed.
- M. L. Kim-Fu, A. R. Moll, E. E. Hernandez, B. Droz, T. N. J. Fouquet and J. Field, Environ. Sci.:Processes Impacts, 2024, 26, 2158–2165 RSC.
- T. Hoxie, S. Zhang, N. J. Herkert, R. A. Bauer, Y. Guo, A. Bhattacharya, C. C. Carignan, K. Hoffman, C. P. Higgins and H. M. Stapleton, Environ. Sci. Technol., 2024, 58, 16316–16326 CrossRef CAS PubMed.
- L. Cahuas, D. J. Muensterman, M. L. Kim-Fu, P. N. Reardon, I. A. Titaley and J. A. Field, Environ. Sci. Technol., 2022, 56, 17070–17079 CrossRef CAS PubMed.
- K. Rohler, B. Susset and P. Grathwohl, Sci. Total Environ., 2023, 902, 166555 CrossRef PubMed.
- J. Zweigle, B. Bugsel, K. Rohler, A. A. Haluska and C. Zwiener, Environ. Sci. Technol., 2023, 57, 6647–6655 CrossRef CAS PubMed.
- A. Gustafsson, A. Bergman and J. M. Weiss, Chemosphere, 2022, 303, 135061 CrossRef CAS PubMed.
- U.S.E.P.A, Technical Fact Sheet: Drinking Water Health Advisories for Four PFAS (PFOA, PFOS, GenX Chemicals, and PFBS), accessed June 21 2024 Search PubMed.
- R. Vestergren, I. T. Cousins, D. Trudel, M. Wormuth and M. Scheringer, Chemosphere, 2008, 73, 1617–1624 CrossRef CAS PubMed.
- M. Chen, T. Guo, K. He, L. Zhu, H. Jin, Q. Wang, M. Liu and L. Yang, Sci. Total Environ., 2019, 656, 201–208 CrossRef CAS PubMed.
- J. C. D'Eon and S. A. Mabury, Environ. Sci. Technol., 2007, 41, 4799–4805 CrossRef PubMed.
- I. Zabaleta, E. Bizkarguenaga, U. Izagirre, N. Negreira, A. Covaci, J. P. Benskin, A. Prieto and O. Zuloaga, Sci. Total Environ., 2017, 609, 1085–1092 CrossRef CAS PubMed.
- S. T. Peskett and A. A. Rand, Environ. Sci.:Processes Impacts, 2022, 24, 1758–1768 RSC.
- M. Chen, L. Qiang, X. Pan, S. Fang, Y. Han and L. Zhu, Environ. Sci. Technol., 2015, 49, 13817–13824 CrossRef CAS PubMed.
- W. Xie, Q. Wu, I. Kania-Korwel, J. C. Tharappel, S. Telu, M. C. Coleman, H. P. Glauert, K. Kannan, S. V. Mariappan, D. R. Spitz, J. Weydert and H. J. Lehmler, Arch. Toxicol., 2009, 83, 909–924 CrossRef CAS PubMed.
- L. Xu, D. M. Krenitsky, A. M. Seacat, J. L. Butenhoff and M. W. Anders, Chem. Res. Toxicol., 2004, 17, 767–775 Search PubMed.
- The Business Research Company, Electronic Waste global Market report 2025: Market size, trends, and global forecast 2025–2034., https://www.thebusinessresearchcompany.com/report/electronic-waste-global-market-report, 2025 Search PubMed.
Footnote |
| † The authors dedicate this paper to the memory of Liisa Jantunen. |
| This journal is © The Royal Society of Chemistry 2026 |