Open Access Article
Open Access ArticleTobacco-specific alkaloid (TSA) formation in aged e-cigarette juices: mechanistic insights into hydroxyl radical-initiated nicotine oxidation
Xinyang Guo a,
Bradley H. Isenor
a,
Bradley H. Isenor b,
Kimberly Wong
b,
Kimberly Wong a,
James Davisa,
Arthur Chan
a,
James Davisa,
Arthur Chan c and
Ran Zhao
c and
Ran Zhao *a
*a
aDepartment of Chemistry, University of Alberta, Edmonton, Alberta T6G 2G2, Canada. E-mail: rz@ualberta.ca
bDepartment of Chemistry, University of Toronto, Toronto, Ontario M5S 3H6, Canada
cDepartment of Chemical Engineering and Applied Chemistry, University of Toronto, Toronto, Ontario M5S 1A1, Canada
First published on 24th March 2026
Abstract
Vaping e-cigarette juice (e-juice) is popular among young consumers. Although e-juice compositions are well characterized, chemical transformations during indoor storage are often overlooked. Particularly, the fundamentals of nicotine oxidation in aged e-juices remain poorly understood, despite rising consumer concerns. This work utilizes p-toluenesulfonyl chloride (TsCl) derivatization, coupled with analysis by liquid chromatography-mass spectrometry (LC-MS) and electron paramagnetic resonance (EPR) spectroscopy, to study nicotine oxidation in a realistic storage environment. We report that tobacco-specific alkaloids (TSAs), including nornicotine (NN) and anabasine (ANA), can accumulate in aged e-juice. These compounds are immediate precursors of carcinogens. Within nine days of storage, NN concentration can reach up to 250 µmol L−1 and can remain elevated during prolonged storage. This concentration is 6 times higher than that of fresh e-juices and more than 270 times higher than that of typical human saliva. A hydroxyl radical oxidation (OH-oxidation) experiment indicated that the indoor OH-oxidation of nicotine yields TSAs. Lastly, we propose a radical-driven mechanism supported by the detection of nitroxide radicals in the EPR spectra. These radicals demonstrated strong correlations with nicotine (R2 = 0.94–0.98). This work reveals the transformation of nicotine in popular consumable products and highlights the importance of encouraging proper e-juice storage practices.
Environmental significanceThe aging of e-cigarette juice (e-juice) during storage is a potential health concern for smokers and bystanders. When exposed to indoor air, e-juices can accumulate at least nine tobacco-specific alkaloids (TSAs), which are immediate precursors of carcinogenic tobacco specific N-nitrosamines (TSNAs). Moreover, the aging of e-juice can be significant after one week of air exposure, with the TSA concentrations reaching 6-times higher than their original values. Nicotine oxidation driven by indoor hydroxyl radicals is a plausible TSA formation route, and it can follow an abiotic radical propagation mechanism. Overall, this study highlights a previously overlooked in situ chemical transformation in e-juice products. Its implications emphasize that proper storage practices are necessary to minimize potential high TSA exposure in consumers. |
Introduction
Vaping has gained tremendous popularity among global communities, particularly with a notable spread among younger consumers in North American high schools.1–4 Unlike conventional tobacco smoking, vaping e-cigarettes is occasionally seen in low-ventilation indoor environments, such as homes or cars.5 Therefore, health concerns associated with vaping, including reduced indoor air quality, excessive glycol exposure, and e-cigarette or vaping associated lung injury (EVALI) are emerging topics that require prioritized research to protect young and vulnerable populations.3,6,7E-juice formulations primarily contain glycols, nicotine, and flavoring additives.8 In addition to the chemical complexity, vaping e-juice induces various chemical reactions, including oxidation, pyrolysis, and nitrosation.9–11 Although the chemical composition of e-juice and its aerosols has been frequently studied,12 the chemical transformations of e-juice before vaping are often overlooked. Consumers often share their observation of e-juice changing appearance on social media platforms and express their concerns about unrecognized chemicals. However, only a limited number of studies have been conducted to investigate the cause of this phenomenon and the corresponding health implications.13,14 Existing studies have already demonstrated that the glycol solvent can be oxidized when stored in indoor environments, producing various harmful contaminants.15,16 Similarly, nicotine may undergo a comparable process during storage due to its relatively high concentration. For instance, e-juices in the Canadian market can contain nicotine levels of up to 20 mg mL−1.17 While the oxidation of nicotine in e-juices has not received adequate attention, Korsarac et al. reported a decline in nicotine concentration during the storage of e-juice, suggesting a probable chemical transformation of nicotine before vaping.18 Hoa et al. provided theoretical insights that nicotine may react with atmospheric oxidants, such as hydroxyl (OH) radicals.19 Although the oxidation of nicotine has been preliminarily studied in atmospheric environments,20 it remains unclear in condensed matrices, such as consumable e-juices.
Tobacco-specific alkaloids (TSAs) are common e-juice impurities and potential nicotine oxidation products.12,21 Their presence may modify the toxicological parameters of the e-juice. For example, the toxicities of nornicotine (NN, LD50 = 18.8 mg kg−1) and anabasine (ANA, LD50 = 16 mg kg−1) are slightly lower than that of nicotine (LD50 = 10 mg kg−1) in mice.22,23 Despite having lower direct toxicity values, TSAs are immediate precursors of highly toxic and carcinogenic tobacco-specific nitrosamines (TSNAs) in nitrite-containing e-juices and human saliva.11,24,25 Although TSAs are minor natural nicotine metabolites,25,26 consuming aged e-juices may introduce additional TSAs on top of the metabolic background, subsequently enhancing the endogenous accumulation of TSNAs. Therefore, assessing the presence and accumulation of TSAs in e-juices helps in understanding carcinogen exposure from vaping and provides chemical insights into vaping-related health issues.
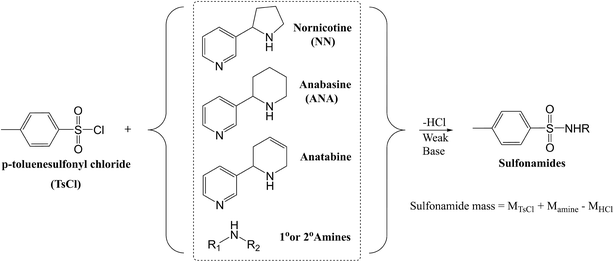
Liquid chromatography coupled with mass spectrometry (LC-MS) is a common approach to analyze trace concentrations of TSAs.27,28 Simple LC-MS analysis mandates calibration with standards and presents challenges in identifying unknown TSAs. Derivatization offers additional selectivity by utilizing the common features of compounds. This study incorporates derivatization with p-toluenesulfonyl chloride (TsCl), which is a popular protecting agent for amines in synthetic chemistry.29–31 In Fig. 1, we illustrate the TsCl reaction scheme with common TSAs.
This study details a chemical analysis of commercial e-juices over a short-term storage period, during which the samples are stored under common improper storage conditions. To explore the formation mechanism of TSAs, we conducted an OH-oxidation experiment and an analysis using continuous-wave electron paramagnetic resonance (EPR) spectroscopy to investigate radical intermediates. The results from this study reveal the transformation of nicotine in consumer-relevant storage scenarios, emphasize the importance of encouraging proper storage protocols for e-juices, and highlight the need to understand the impact of indoor environmental oxidants on consumable products.
Experimental
Chemicals and materials
Ultrapure water was generated using a Thermo-Fisher Scientific Barnstead™ E-Pure™ ultrapure water purification system. Nicotine (99%), nornicotine (NN, 98%), anabasine standard (ANA, 1 mg mL−1 in methanol), p-toluenesulfonyl chloride (TsCl, 99%), boric acid (99.5%), hydrogen peroxide (H2O2, 30% in water), HPLC grade acetonitrile, formic acid (98–100%), caffeine (99%), L-ascorbic acid (vitamin C, 99%), glycerol (VG, 99%), KH2PO4 (99%) and K2HPO4 (99%) were purchased from Sigma-Aldrich. Propylene glycol (PG, 99%) and sodium hydroxide pellets were purchased from Fisher Chemical. 5,5-Dimethyl-1-pyrroline N-oxide (DMPO) was purchased from Cayman Chemical Co. Flavorless commercial e-juice products (0 mg mL−1, 3 mg mL−1, and 20 mg mL−1 nicotine) were purchased from local vape shops in Edmonton, Canada. The 3 mg per mL e-juice is made from free-base nicotine; the 20 mg per mL e-juice is made from nicotine salt.Aging of e-juice
A summary of the experimental workflow is illustrated in Fig. S1 (Section S1). This experiment monitors the aging of e-juice. Flavorless e-juices with two nicotine concentrations (3 mg mL−1 and 20 mg mL−1) were divided into triplicate 10 mL aliquots in 20 mL glass vials. A separate 0 mg per mL triplicate was prepared as the nicotine control. An air exposure control was prepared by sealing 3 mg per mL e-juice in the same container. Two long-term references were prepared by air-exposing 0 mg mL−1 and 3 mg per mL e-juices for one year. All vials were wrapped with aluminum foil in a temperature-controlled lab room (21 ± 1 °C). Air-exposed samples were loosely capped to mimic improper e-juice storage practices. We assumed negligible solvent evaporation due to the low vapor pressure values of glycols (1.68 × 10−4 mmHg for VG and 0.13 mmHg for PG, respectively, at room temperature).32 We acknowledge that it is not practical to mimic all possible storage conditions, but our storage protocol represents the upper-bound scenario of oxidant exposure in e-juices.OH-oxidation of nicotine
This experiment validates whether the OH-oxidation of nicotine produces the TSAs observed in aged e-juices. It is adapted and modified from our previous work.33 Reactions were conducted in duplicate in a photochemical reactor (Rayonet, RPR-200) equipped with two UVB lamps (wavelength 280–320 nm, approximate UV output of 500 µW cm−2 at the working distance). A mixture containing 0.01% (V/V%) aqueous nicotine and 15 mmol per L H2O2 was stirred under UV irradiation. The nicotine concentration is more than 10 times lower than that of typical e-juices (up to 20 mg mL−1 or 2%). It was decided to prevent overloading the LC while preserving a composition similar to that of actual e-juices. H2O2 is the source of OH radicals. It is 24 times more concentrated than nicotine to ensure that further evolution of TSAs can be observed.For the negative control, separate photolysis reactions were conducted without adding H2O2. Thus, only aqueous nicotine was exposed to UV irradiation. Aliquots of both experimental and control mixtures were sampled at time intervals and injected into autosampler vials. These vials were pre-filled with a 25 mmol per L L-ascorbic acid solution to prevent uncontrolled reactions caused by excessive H2O2 and OH radicals.
Derivatization and LC-MS analysis
The derivatization method was introduced and validated in our previous study.34 The working derivatization solution was a 52 mmol per L TsCl solution in acetonitrile. A 0.25 mol per L sodium borate buffer (pH = 9.0) was made from boric acid and sodium chloride. This pH buffer was added only in post-collection sample analysis; it is not involved during the storage of e-juice nor the OH-oxidation of nicotine. An aliquot of caffeine solution (10 mmol L−1 in 50% acetonitrile) was added as the internal standard to account for potentially varying retention times in e-juices from different manufacturers. The derivatization was performed in 2 mL LC glass autosampler vials. Derivatization mixtures were 1-to-25 dilutions of e-juice, containing 20.8 mmol per L TsCl and 200 µmol per L caffeine internal standard. The mixtures were then incubated in a 50 °C water bath for one hour prior to LC-MS injection.Concentrations of TSAs were determined using standard addition. Briefly, a gradient of alkaloid standard concentrations was spiked into derivatization matrices and derivatized together with e-juice samples. This is because commercial standards of TsCl-derivatized TSAs are not readily available. Although the lack of commercial standards makes it challenging to determine key analytical parameters, such as recovery rates and reaction yields, standard addition produces robust quantitative data by normalizing matrix effects. All TSA concentrations reported in this work were determined using a five-point standard addition method.
The LC-MS analysis was conducted using positive electro-spray ionization mode (LC-ESI(+)-MS) on a Thermo-Fisher LTQ-XL mass spectrometer and a Thermo-Fisher Exactive Orbitrap mass spectrometer (resolution = 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000). Samples were separated on a Phenomenex Luna Omega polar C-18 column (150 mm × 2.1 mm × 3 µm) installed on an Agilent 1100 HPLC system. All necessary blank samples, including buffer blanks, TsCl blanks, and e-juice blanks, were analyzed prior to sample injection. Detailed instrumental parameters are tabulated in Tables S1 and S2 in Section S2.
000). Samples were separated on a Phenomenex Luna Omega polar C-18 column (150 mm × 2.1 mm × 3 µm) installed on an Agilent 1100 HPLC system. All necessary blank samples, including buffer blanks, TsCl blanks, and e-juice blanks, were analyzed prior to sample injection. Detailed instrumental parameters are tabulated in Tables S1 and S2 in Section S2.
EPR analysis
This experiment verifies the radical involvement in nicotine oxidation. The EPR analysis was performed using a Bruker ECS-EMXplus EPR spectrometer at the University of Toronto, equipped with an ER 4119 HS resonator. The EPR operates in continuous wave and X-band frequency modes. All solutions were made with ultrapure water. DMPO was used as a spin trap for radical speciation. The DMPO stock solution is a 200 mmol per L solution prepared in a phosphate buffer solution (PBS, 100 mmol L−1, pH = 7.4). It was prepared fresh monthly by adding 100 mmol per L KH2PO4 to 100 mmol per L K2HPO4, and then treated with Chelex® 100 (Sigma Aldrich, sodium form) to remove any heavy metals that may bias the radical generation. The PBS is a commonly used near-neutral condition to ensure a reproducible signal of DMPO-adducts.35 However, the actual pH values of EPR mixtures were measured between 9.1 and 9.5 because nicotine is a weak base. Therefore, the buffering effect of PBS was not active. The mildly basic condition may accelerate the decomposition of DMPO-adducts according to Marriott et al., but it has a limited effect on the spectral pattern.36EPR samples were prepared at their desired concentrations in volumes of 500 µL in 1.5 mL micro-centrifuge tubes by adding chemicals in the following order: PBS, nicotine, DMPO (final concentration 100 mmol L−1), and H2O2 (final concentration 1.25 mol L−1). Depending on the experiment, the samples contained varying concentrations of nicotine (0–1.25 mol L−1). For quality control, all nicotine-containing samples were measured in triplicate, and blank samples were prepared accordingly (presence or absence of nicotine, H2O2, DMPO, and UV light). A glycol-based sample was also prepared by replacing all the water with a glycol solvent (50% PG + 50% VG) in the mixture. Samples were irradiated with UV-B light for 5 minutes and stored in an insulated cooler with ice packs prior to EPR measurement. Additional sample preparation details are described in Section S3.
Results and discussion
Overview of aged e-juices
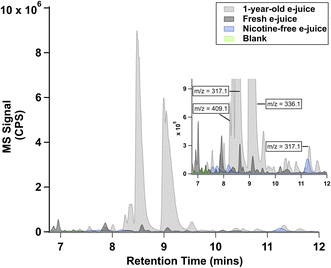
Fig. 2 shows the base-peak chromatogram (BPC) of three commercial e-juice samples: one-year-old 0 mg mL−1, one-year-old 3 mg mL−1, and fresh 3 mg mL−1. The aged 3 mg mL−1 sample has strong chromatographic peaks between 8 and 10 min, with a few smaller but significant peaks shown between 10 and 12 min. The dominating mass-to-charge ratios (m/z) contained in the mass spectra of these peaks have been labeled in the sub-window of Fig. 2. Compared to the aged sample, the same retention time window in the fresh 3 mg mL−1 sample contains fewer and weaker signals. For the aged 0 mg mL−1 sample, the chromatogram does not exhibit significant peaks, even though it is aged. The discrepancy between the three samples suggests that (1) long-term storage could enhance the concentration of minor TSAs in the e-juice; and (2) the presence of nicotine is responsible for the formation of these compounds in both fresh and aged e-juice.To explore the identity of TSAs in aged e-juice, we selected and focused on nine elemental compositions in the aged 3 mg per mL e-juice. These nine compositions are verified TsCl-derivatives according to the validation method described in Section S4 (Fig. S2). In addition, their peak intensities in the aged e-juice are more than 5 times stronger than those in the fresh e-juice. Nine TSAs were identified in the aged 3 mg per mL e-juice. Extracted ion chromatograms (EICs) of TSAs are shown in Fig. 3, which also demonstrates their predicted elemental composition according to high-resolution LC-MS analysis. We assigned peaks of interest in each EIC using Roman letters because each elemental composition contains multiple structural isomers. By referring to commercial standards, NN (I, 8.6 min) and ANA (IV, 10.8 min) were confirmed.
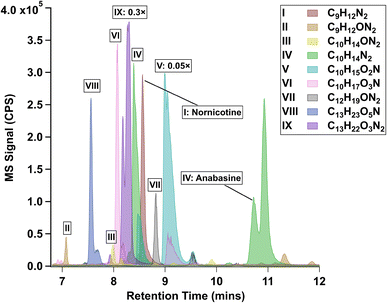 | ||
| Fig. 3 Extracted ion chromatogram (EIC) of nine elemental compositions extrapolated from the aged e-juice. The EICs of V and IX are scaled by factors of 0.05× and 0.3× for visual purposes. | ||
Formation of nornicotine during e-juice storage
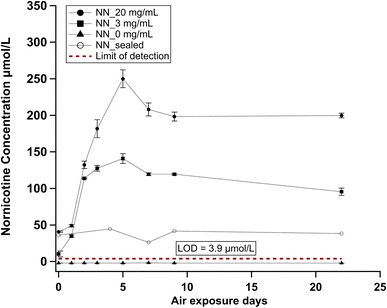
We used the concentration of NN as the marker to track the aging of e-juices during storage. Data points shown in Fig. 4 were determined from standard addition and reflect the back calculated concentration in actual e-juice samples. In the control group (nicotine-free e-juice), the NN concentration was below the detection limit (3.9 µmol L−1) throughout the storage period. In sealed e-juice samples, it remained at a stable level of approximately 36 µmol L−1, suggesting that TSA formation can be controlled by sealing the e-juice in air-tight environments. For the air-exposed 3 mg per mL e-juices, NN started accumulating from 10 ± 4 µmol L−1, peaked at 141 ± 2 µmol L−1 on day 5, and reached a plateau of 119 ± 1 µmol L−1 after the peak. This concentration was reduced to 95 ± 5 µmol L−1 until day 22, suggesting the potential formation of secondary decomposition products. For the air-exposed 20 mg per mL e-juices, the NN concentration is consistently higher than that in 3 mg mL−1: the initial concentration was 41 ± 1 µmol L−1, the peak concentration was 250 ± 12 µmol L−1, and the plateau concentration was 198 ± 6 µmol L−1, which remained relatively unchanged (200 ± 3 µmol L−1) until day 22. Our observed NN concentrations are consistent with the literature-reported range. NN in the 20 mg mL−1 sample is between 41 and 250 µmol L−1, a range equivalent to 6.1–37.0 µg mL−1, or 0.031–0.19% of the stated nicotine concentration. This range overlaps with the 5–21 µg mL−1 range reported by Palazzolo et al.37 and is similar to the 0.2% ratio reported by Flora et al.38The mouth is the gateway for e-cigarette aerosols entering the human body, and saliva is the first matrix that the inhaled aerosols encounter. Numerous studies have reported that TSNAs are produced by the endogenous nitrosation of TSA by nitrite in human saliva.24,25,39 Therefore, an increased TSA concentration in saliva could promote the formation rate of TSNAs, which may subsequently contribute to tobacco-related cancer development.40 NN concentrations in aged e-juice are more than two orders of magnitude higher than saliva concentrations (3.3–136.9 ng mL−1) measured by Bustamante et al.25 For example, the peak concentration of the 20 mg mL−1 e-juice (37 µg mL−1) is 270 times higher than the maximum 136.9 ng mL−1 in human saliva. Although it is challenging to determine the typical delivery efficiencies of TSAs from e-juice to the human body, this efficiency was found to be significant for nicotine (93.8 ± 14.5%).41 Therefore, despite metabolism being conventionally considered the main source of TSAs, the high TSA concentration in e-juice and its aerosol could potentially deliver TSAs into the saliva, increasing their concentration beyond the existing metabolic background and enhancing the endogenous TSNA formation rates.25 In addition, Jin et al. reported the accumulation of TSNAs in nitrite-containing e-juices, suggesting in situ formation of carcinogens from TSAs in aged e-juices.11 According to the average weekly e-juice consumption (44 mL) data reported by Tillery et al.,42 a typical e-cigarette consumer would receive a weekly NN exposure of up to 1.3 mg from the e-juice that is only one-week-old, assuming 100% transport efficiency from e-juice to the consumer.
OH-oxidation of nicotine
TSAs are conventionally reported as by-products of tobacco harvesting due to microbial activities.43–46 However, the accumulation of NN during short-term storage supports the notion that TSA formation may not require a microorganism-rich environment. According to theoretical calculations conducted by Hoa et al., nicotine is prone to oxidation when exposed to atmospheric oxidants.19 Ozone and OH radicals are the two most common indoor oxidants.47 Based on the detection of TSAs, the oxidation route we observed primarily occurred on the saturated pyrrolidine section of the nicotine molecule.48 Given that ozone-driven oxidation prefers unsaturated sites like alkenes, its reaction on the saturated pyrrolidine would be less preferred. Therefore, the oxidation by OH radicals would be a more significant pathway to transform nicotine into TSAs. This section describes an OH-oxidation experiment that simulates the transformation of nicotine when exposed to atmospheric oxidants.This OH-oxidation experiment was conducted in duplicate by reacting nicotine with H2O2 in a photochemical reactor, where H2O2 served as the source of OH radicals. Although typical indoor storage conditions do not involve UV light, it is an established and practical method for generating OH radicals in the aqueous environment.33 Fig. 5 summarizes changes in background-normalized peak height/area ratios of nicotine and TSAs during OH-oxidation. Temporal profiles of nicotine and TSAs in the direct photolysis reaction did not undergo significant changes, indicating that oxidants are required to decompose nicotine. When H2O2 was involved, nicotine exhibited a first-order decay with an estimated rate constant of 1.5 × 10−2/s. Peaks of five TSAs (I, II, IV, VI, and VII in Fig. 3) intensified upon UV irradiation. Temporal profiles of NN and II exhibit a peak shape, suggesting their subsequent decomposition in an oxidative environment. This degradation of NN is more significant compared to that observed in Fig. 4, likely because the oxidant concentration during the OH-oxidation experiment is higher. In contrast, the temporal profiles of ANA, VI, and VII exhibited a plateau shape. This plateau is likely because nicotine was mostly consumed within 5 minutes. The results from this experiment support the notion that OH-oxidation can transform nicotine into NN, ANA, and various TSAs found during the short-term storage of e-juices.
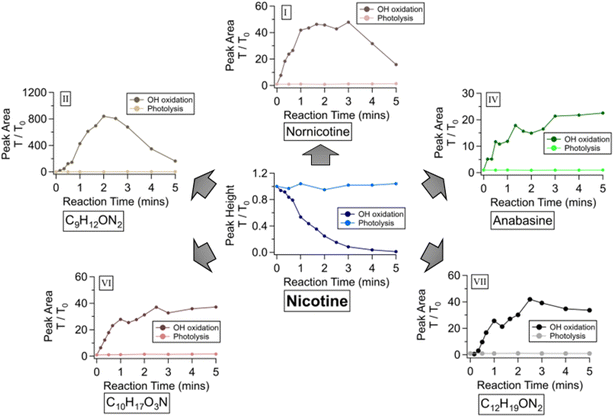 | ||
| Fig. 5 Changes in oxidation products during the photooxidation experiment. The Roman letter in each sub-figure corresponds to compound numbers in Fig. 2. The sub-figures contain temporal profiles of peak area ratios (T/T0) obtained for each compound with and without H2O2. The peak area ratio represents the normalization of the peak area at any given time against the background peak area. | ||
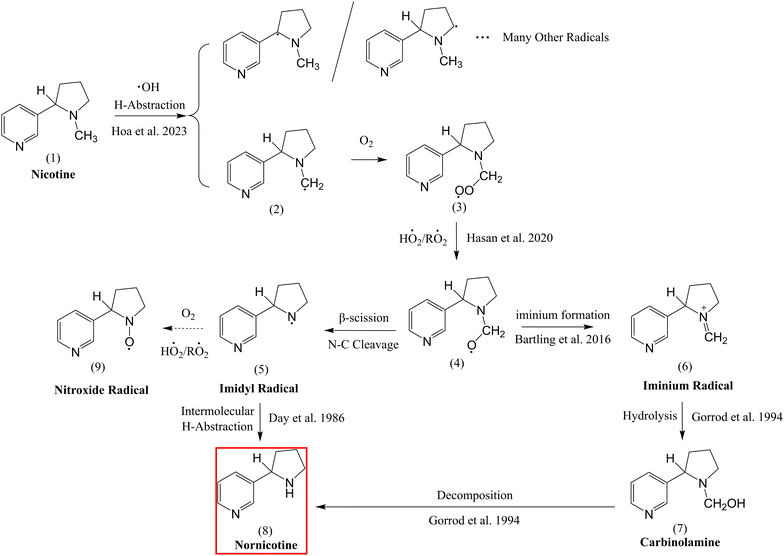
Dynamic temporal profiles of nicotine and TSAs under UV irradiation indicate that nicotine can be readily oxidized by OH radicals. To explore the possible oxidation route, we proposed a mechanism shown in Fig. 6 based on the established OH-oxidation mechanism.49,50 For a tertiary amine like nicotine (1), protons attached to alkyl groups adjacent to the nitrogen can be abstracted by the OH radical due to the nitrogen's electron-donating effect.51,52 The alkyl radical (2) is one of the probable H-abstraction products according to Hoa et al.19 After the addition of oxygen, radical (2) becomes the peroxyl radical (3) and can undergo a cross reaction with another peroxyl radical or hydroperoxyl radical in the system,53 subsequently becoming an alkoxy radical (4). The alkoxy radical is unstable and can further propagate to an imidyl radical (5) or an iminium radical (6). For the imidyl radical (5), it can either propagate to a nitroxide radical (9) or initiate intermolecular H-abstraction from another molecule according to Day et al.,54 forming nornicotine. This is because the imidyl radical is an electrophilic H-atom acceptor. For the iminium radical (6), it can be formed according to schemes proposed by Bartling et al.,55 and can further undergo hydrolysis to form a carbinolamine molecule, which will subsequently decompose to nornicotine.56 This iminium radical route is comparable to the metabolic mechanism of nicotine,56 but the OH-oxidation could activate this transformation in a metabolite-free environment.
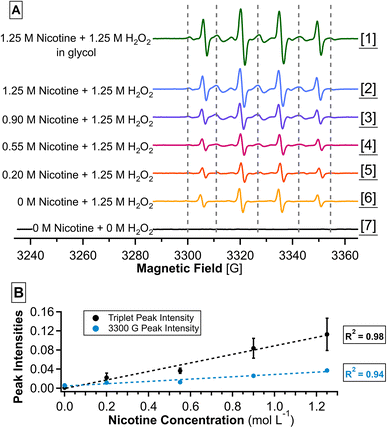
EPR analysis of tobacco-specific radicals
The detection of radical intermediates is an indicator of the proposed OH-oxidation mechanism; however, unstable radicals require the use of sensitive and time-resolved techniques.57 Unfortunately, our instrument (ESI-MS) is not suitable for conducting such tasks. Alternatively, we utilized EPR to conduct free radical analysis. This reaction was conducted at pH 9.1 to 9.5 according to the different nicotine concentrations. This pH range is comparable to that of typical e-juices, as reported by Lisko et al. (up to 9.1).58 Fig. 7a presents the averaged EPR spectra of seven samples ([1]–[7]), each subjected to UV-B irradiation (280–320 nm). Samples [2]–[7] were aqueous samples containing a gradient of nicotine concentrations to discover radicals correlated with nicotine. Sample [1] was prepared in a glycol solvent to mimic the actual e-juice matrix. To improve the level of confidence, we analyzed all necessary control samples (with or without nicotine, UV, and H2O2), as shown in Fig. S3–S8 (Section S5.2).In samples containing H2O2 ([1]–[6]), four strong peaks were observed with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 intensity ratio, which is characteristic of DMPO-OH. The glycol sample [1] displayed no additional peaks compared to the aqueous sample [2], suggesting that the glycol solvent has a limited effect on the formation of radicals. In samples containing nicotine ([1]–[5]), a set of 1
1 intensity ratio, which is characteristic of DMPO-OH. The glycol sample [1] displayed no additional peaks compared to the aqueous sample [2], suggesting that the glycol solvent has a limited effect on the formation of radicals. In samples containing nicotine ([1]–[5]), a set of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 triplet peaks (approximate magnetic field values of 3310, 3326, and 3342 G) was observed in addition to the existing DMPO-OH peaks. A set of small peaks was also observed at 3300 and 3354 G that may be partially masked by the other peaks in the EPR spectrum.
1 triplet peaks (approximate magnetic field values of 3310, 3326, and 3342 G) was observed in addition to the existing DMPO-OH peaks. A set of small peaks was also observed at 3300 and 3354 G that may be partially masked by the other peaks in the EPR spectrum.
The 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 peak shape is considered unusual for DMPO adducts, suggesting that the nitrogen atom neighboring the oxygen-centered radical in DMPO may contribute to its hyperfine splitting. It is also possible that the triplet is part of a larger set of peaks, but other peaks are masked by strong DMPO-OH peaks. To further confirm the possible identity of the triplet peak, we compared it with a set of simulated EPR spectra (Fig. S9–S12) in Section S5.3, generated by SpinFit in Bruker's Xenon software. Briefly, the triplet peak is consistent with nitroxide radicals, and our observation is consistent with a simulated spectrum that contains both nitroxide radicals and an unknown DMPO adduct (DMPO-Y). As a result, the nitroxide radical (9) in Fig. 6 is likely contributing to the EPR signal and supporting the existence of the intermediate iminyl radical (5). In other words, the EPR result supports that the pyrrolidine nitrogen of nicotine is actively involved in the oxidation mechanism of nicotine.
1 peak shape is considered unusual for DMPO adducts, suggesting that the nitrogen atom neighboring the oxygen-centered radical in DMPO may contribute to its hyperfine splitting. It is also possible that the triplet is part of a larger set of peaks, but other peaks are masked by strong DMPO-OH peaks. To further confirm the possible identity of the triplet peak, we compared it with a set of simulated EPR spectra (Fig. S9–S12) in Section S5.3, generated by SpinFit in Bruker's Xenon software. Briefly, the triplet peak is consistent with nitroxide radicals, and our observation is consistent with a simulated spectrum that contains both nitroxide radicals and an unknown DMPO adduct (DMPO-Y). As a result, the nitroxide radical (9) in Fig. 6 is likely contributing to the EPR signal and supporting the existence of the intermediate iminyl radical (5). In other words, the EPR result supports that the pyrrolidine nitrogen of nicotine is actively involved in the oxidation mechanism of nicotine.
To further investigate the relationship between the suspected nitroxide radical and nicotine, we plotted its EPR peak intensity against the changing nicotine concentration, as shown in Fig. 7b. Both the triplet peaks and the two small peaks demonstrated strong correlations with the gradient of nicotine: R2 values are 0.98 and 0.94, respectively. Therefore, we confirmed that the presence of DMPO, nicotine, and UV irradiation is a necessary factor in generating these radicals. While EPR suggests preliminary evidence of nitroxide radicals, their structural characterization will require additional comprehensive studies in the future.
Conclusion
This work reveals the chemical transformation of nicotine in aged e-cigarette juices (e-juices) under typical indoor storage conditions. Oxidation is found to be a significant transformation pathway for nicotine in consumer settings. It can be driven by radical processes involving hydroxyl (OH) radicals and nitrogen-centered radicals, facilitating the accumulation of various tobacco-specific alkaloids (TSAs). Although our OH-oxidation experiment represents an extreme outcome of nicotine oxidation, it highlights the upper-bound consequence when the e-juice is not perfectly sealed in an air-tight environment. Aged e-juices in this study can contain nornicotine (NN) concentrations more than 270 times higher than those in typical human saliva. Vaping aged e-juice can potentially increase the concentration of NN and many other TSAs in saliva, subsequently enhancing the endogenous formation rate of carcinogenic tobacco-specific N-nitrosamines (TSNAs). The implications of this work indicate that the stated shelf life of commercial e-juices may not accurately represent their chemical stability, as notable compositional changes can occur upon exposure to room air. Future vaping studies could consider e-juice aging during storage and handling to better reflect realistic use conditions.Author contributions
Dr Xinyang Guo: designed and oversaw the entire project, developed the analytical method, performed the photooxidation experiment, and wrote the manuscript. Bradley H. Isenor: performed an EPR analysis of nicotine and drafted the methods and discussion associated with the EPR analysis. Kimberly Wong: performed the aging experiment on nicotine, provided critical insights and support for photooxidation experiments, and helped build calibration for alkaloid compounds. James Davis: helped to calibrate alkaloid compounds. Dr Arthur Chan: oversaw the EPR analysis and provided critical resources and connections to the EPR instrument. Dr Ran Zhao: the PI initiated and oversaw the entire project and proofread the manuscript.Conflicts of interest
No conflict of interest was declared.Data availability
The data supporting this article have been included in the supplementary information (SI). Supplementary information: an overview of the experimental design, detailed LC-MS instrumental parameters and settings, detailed EPR sample preparation procedures, TsCl derivative identification procedures, and all supporting EPR analysis spectra. See DOI: https://doi.org/10.1039/d5em01030f.Acknowledgements
The authors thank Dr Randy Whittal at the University of Alberta's Mass Spectrometry facility for technical help and access to an HPLC system; Dr Darcy Bruns from the University of Toronto's ‘Centre for the Spectroscopic Investigation of Complex Organic Molecules and Polymers’ for technical help and access to the EPR. Kimberly Wong and James Davis thank the Undergraduate Research Initiative and Canada Summer Jobs for funding. This research project was supported by the Canada Foundation for Innovation (Project Number 38334) and an NSERC Discovery Grant (RGPIN2018-03814).References
- M. C. Fadus, T. T. Smith and L. M. Squeglia, The rise of e-cigarettes, pod mod devices, and JUUL among youth: Factors influencing use, health implications, and downstream effects, Drug Alcohol Depend., 2019, 201, 85–93 CrossRef PubMed.
- J. Kim, S. Lee and J. S. Chun, An international systematic review of prevalence, risk, and protective factors associated with young people’s e-cigarette use, Int. J. Environ. Res. Pub. Health, 2022, 19(18), 11570 CrossRef PubMed.
- C. Lau, R. Zhao and D. Vethanayagam, Chemistry review of vaping products and respiratory injury, Spectrum, 2020,(6), 92 Search PubMed.
- S. E. McCabe, C. J. Boyd, R. J. Evans-Polce, V. V. McCabe and P. T. Veliz, School-level prevalence and predictors of e-cigarette use in 8th, 10th, and 12th grade U.S. Youth: Results from a national survey (2015–2016), J. Adolesc. Health, 2020, 67, 531–541 CrossRef PubMed.
- H. Tattan-Birch, S. E. Jackson, L. Shahab and J. Brown, Are people more likely to vape or smoke indoors? A population survey of adults in England, Nicot. Tobacco Res., 2024, 26, 1404–1411 CrossRef CAS PubMed.
- K. Stratton, in Public Health Consequences of E-Cigarettes, ed. Stratton, K., Kwan, L. Y. and Eaton, D. L., National Academies Press, 2018 Search PubMed.
- M. E. Rebuli, J. J. Rose, A. Noël, D. P. Croft, N. L. Benowitz, A. H. Cohen, M. L. Goniewicz, B. T. Larsen, N. Leigh, M. D. McGraw, A. C. Melzer, A. L. Penn, I. Rahman, D. Upson, L. E. Crotty Alexander, G. Ewart, I. Jaspers, S. E. Jordt, S. Kligerman, C. E. Loughlin, R. McConnell, E. R. Neptune, T. B. Nguyen, K. E. Pinkerton and T. J. Witek, The E-cigarette or vaping product use–associated lung injury epidemic: Pathogenesis, management, and future directions: An official american thoracic society workshop report, Ann. Am. Thorac. Soc., 2023, 20, 1–17 CrossRef PubMed.
- R. Chen, A. Aherrera, C. Isichei, P. Olmedo, S. Jarmul, J. E. Cohen, A. Navas-Acien and A. M. Rule, Assessment of indoor air quality at an electronic cigarette (Vaping) convention, J. Expos. Sci. Environ. Epidemiol., 2018, 28, 522–529 CrossRef CAS PubMed.
- H. Jiang, C. M. Ahmed, T. J. Martin, A. Canchola, I. W. Oswald, J. A. Garcia, J. Y. Chen, K. A. Koby, A. J. Buchanan, Z. Zhao, H. Zhang, K. Chen and Y. H. Lin, Chemical and toxicological characterization of vaping emission products from commonly used vape juice diluents, Chem. Res. Toxicol., 2020, 33, 2157–2163 Search PubMed.
- K. S. Hopstock, V. Perraud, A. B. Dalton, B. Barletta, S. Meinardi, R. M. Weltman, M. A. Mirkhanian, K. J. Rakosi, D. R. Blake, R. D. Edwards and S. A. Nizkorodov, Chemical analysis of exhaled vape emissions: Unraveling the complexities of humectant fragmentation in a human trial study, Chem. Res. Toxicol., 2024, 37, 1000–1010 Search PubMed.
- X. C. Jin, K. A. Wagner, M. S. Melvin, D. C. Smith, Y. B. Pithawalla, W. P. Gardner, K. C. Avery and G. D. Karles, Influence of nitrite on formation of tobacco-specific nitrosamines in electronic cigarette liquids and aerosols, Chem. Res. Toxicol., 2022, 35, 782–791 Search PubMed.
- E. Eshraghian and W. Al-Delaimy, A review of constituents identified in e-cigarette liquids and aerosols, Tobacco Prevent Cessat, 2021, 7, 1–15 Search PubMed.
- M. K. Page, A. D. Merzianu, N. J. Leigh and M. L. Goniewicz, Stability of flavoring chemicals in e-cigarette liquids: A naturalistic product aging study over 24 months, ACS Omega, 2025, 10, 15706–15715 Search PubMed.
- R. M. Strongin, E. Sharma, H. C. Erythropel, N. O. F. Kassem, A. Noël, D. H. Peyton and I. Rahman, Chemical and physiological interactions between e-liquid constituents: Cause for concern?, Tobacco Control, 2024, 34, 393–396 CrossRef PubMed.
- X. Guo, Y. C. Chan, T. Gautam and R. Zhao, Autoxidation of glycols used in inhalable daily products: Implications for the use of artificial fogs and e-cigarettes, Environ. Sci.: Processes Impacts, 2023, 25, 1657–1669 RSC.
- S. Wu, E. Kim and R. Zhao, Acetal formation of flavoring agents with propylene glycol in e-cigarettes: Impacts on indoor partitioning and thirdhand exposure, Environ. Sci. Technol., 2023, 57, 21284–21294 CrossRef CAS PubMed.
- Health Canada, Vaping Products—New limits on nicotine concentration and consultation on flavour restrictions, https://www.canada.ca/en/health-canada/news, 2021, accessed on 2025/09/04.
- I. Kosarac, G. P. Katuri, C. Kubwabo, S. Siddque and T. K. Mischki, Quantitation and Stability of Nicotine in Canadian Vaping Liquids, Toxics, 2023, 11(4), 378 CrossRef CAS PubMed , 37112605.
- N. T. Hoa, L. T. Ngoc Van and Q. V. Vo, Reactions of nicotine and the hydroxyl radical in the environment: Theoretical insights into the mechanism, kinetics and products, Chemosphere, 2023, 314, 137682 CrossRef CAS PubMed.
- B. Yang, S. Wang and L. Wang, Rapid gas-phase autoxidation of nicotine in the atmosphere, J. Phys. Chem. A, 2022, 126(37), 6495–6501 CrossRef CAS PubMed.
- S. M. Reilly, T. Cheng, C. Feng and M. J. Walters, Harmful and potentially harmful constituents in e-liquids and aerosols from electronic nicotine delivery systems (ENDS), Chem. Res. Toxicol., 2024, 37, 1155–1170 Search PubMed.
- A. H. REES and H. SCHNIEDEN, Effect of nicotine, nornicotine and allylnornicotine on the diuretic response to water in rats, Brit. J. Pharmacol. Chemother., 1962, 18, 299–301 Search PubMed.
- S. T. Lee, K. Wildeboer, K. E. Panter, W. R. Kem, D. R. Gardner, R. J. Molyneux, C.-W. T. Chang, F. Soti and J. A. Pfister, Relative toxicities and neuromuscular nicotinic receptor agonistic potencies of anabasine enantiomers and anabaseine, Neurotoxicol. Teratol., 2006, 28, 220–228 CrossRef CAS PubMed.
- M. Siddiqi, R. Kumar, D. Kaul, B. Spiegelhalder and R. Preussmann, Salivary nitrate and nitrite concentrations from a sample population of children and adults in high risk area for esophageal and gastric cancers in Kashmir, India, Cancer Lett., 1992, 64, 133–136 CrossRef CAS PubMed.
- G. Bustamante, B. Ma, G. Yakovlev, K. Yershova, C. Le, J. Jensen, D. K. Hatsukami and I. Stepanov, Presence of the CarcinogenN-Nitrosonornicotine in saliva of E-cigarette users, Chem. Res. Toxicol., 2018, 31, 731–738 Search PubMed.
- R. Sobkowiak and A. Lesicki, Absorption, metabolism and excretion of nicotine in humans, Postepy Biochem., 2013, 59, 33–44 Search PubMed.
- L. B. Von Weymarn, N. M. Thomson, E. C. Donny, D. K. Hatsukami and S. E. Murphy, Quantitation of the minor tobacco alkaloids nornicotine, anatabine, and anabasine in smokers' urine by high throughput liquid chromatography–mass spectrometry, Chem. Res. Toxicol., 2016, 29, 390–397 Search PubMed.
- J. G. Lisko, S. B. Stanfill, B. W. Duncan and C. H. Watson, Application of GC-MS/MS for the analysis of tobacco alkaloids in cigarette filler and various tobacco species, Anal. Chem., 2013, 85, 3380–3384 CrossRef CAS PubMed.
- R. Ding, Y. He, X. Wang, J. Xu, Y. Chen, M. Feng and C. Qi, Treatment of alcohols with tosyl chloride does not always lead to the formation of tosylates, Molecules, 2011, 16, 5665–5673 CrossRef CAS PubMed.
- M. A. Pasha, R. U. R. Khan and N. Shrivatsa, N-Sulfonylation of amines, imides, amides and anilides using p-TsCl in presence of atomized sodium in EtOH-THF under sonic condition, Ultrason. Sonochem., 2015, 26, 15–21 CrossRef CAS PubMed.
- P. Spieß, A. Sirvent, I. Tiefenbrunner, J. Sargueil, A. J. Fernandes, A. Arroyo-Bondía, R. Meyrelles, D. Just, A. Prado-Roller, S. Shaaban, D. Kaiser and N. Maulide, Nms-amides: An amine protecting group with unique stability and selectivity, Chem.–Eur. J., 2023, 29(41), e202301312 CrossRef PubMed.
- T. Daubert and R. Danner, Physical and Thermodynamic Properties of Pure Chemicals: Data Compilation, Choice Reviews Online, 1990, vol. 27, ch. 6, pp. 27–3319 Search PubMed.
- J. V. Amorim, X. Guo, T. Gautam, R. Fang, C. Fotang, F. J. Williams and R. Zhao, Photo-oxidation of pinic acid in the aqueous phase: a mechanistic investigation under acidic and basic pH conditions, Environ. Sci.: Atmos., 2021, 1, 276–287 CAS.
- X. Guo, R. Afroz, S. Wu, K. Wong, V. Carney, M. J. Zuidhof, J. Saharchuk, H. Osthoff and R. Zhao, Characterizing amino compounds in indoor poultry farms: air quality and its impact on workers and chickens in Canadian egg farms, Environ. Sci.:Processes Impacts, 2025, 27(4), 960–973 RSC.
- A. Fadda, A. Barberis and D. Sanna, Influence of pH, buffers and role of quinolinic acid, a novel iron chelating agent, in the determination of hydroxyl radical scavenging activity of plant extracts by electron paramagnetic resonance (EPR), Food Chem., 2018, 240, 174–182 CrossRef CAS PubMed.
- P. R. Marriott, M. J. Perkins and D. Griller, Spin trapping for hydroxyl in water: a kinetic evaluation of two popular traps, Can. J. Chem., 1980, 58, 803–807 CrossRef CAS.
- D. Palazzolo, J. M. Nelson and Z. Hudson, The use of HPLC-PDA in determining nicotine and nicotine-related alkaloids from E-liquids: A comparison of five E-liquid brands purchased locally, Int. J. Environ. Res. Pub. Health, 2019, 16(17), 3015 CrossRef CAS PubMed.
- J. W. Flora, N. Meruva, C. B. Huang, C. T. Wilkinson, R. Ballentine, D. C. Smith, M. S. Werley and W. J. McKinney, Characterization of potential impurities and degradation products in electronic cigarette formulations and aerosols, Regulat. Toxicol. Pharmacol., 2016, 74, 1–11 CrossRef CAS PubMed.
- A. Knezevich, J. Muzic, D. K. Hatsukami, S. S. Hecht and I. Stepanov, Nornicotine nitrosation in saliva and its relation to endogenous synthesis of N’-nitrosonornicotine in humans, Nicot. Tobacco Res., 2013, 15, 591–595 CrossRef CAS PubMed.
- S. Preston-Martin, Evaluation of the evidence that tobacco-specific nitrosamines (TSNA) cause cancer in humans, Crit. Rev. Toxicol., 1991, 21, 295–298 CrossRef CAS PubMed.
- G. St.Helen, C. Havel, D. A. Dempsey, P. Jacob and N. L. Benowitz, Nicotine delivery, retention and pharmacokinetics from various electronic cigarettes, Addiction, 2015, 111, 535–544 CrossRef PubMed.
- A. Tillery, A. Aherrera, R. Chen, J. J. Y. Lin, M. Tehrani, D. Moustafa, J. Mihalic, A. Navas-Acien and A. M. Rule, Characterization of e-cigarette users according to device type, use behaviors, and self-reported health outcomes: Findings from the EMIT study, Tobacco Induced Dis., 2023, 21, 1–11 CrossRef PubMed.
- B. Siminszky, L. Gavilano, S. W. Bowen and R. E. Dewey, Conversion of nicotine to nornicotine in Nicotiana tabacum is mediated by CYP82E4, a cytochrome P450 monooxygenase, Proc. Natl. Acad. Sci. U. S. A., 2005, 102, 14919–14924 CrossRef CAS PubMed.
- W. Stepka and L. J. Dewey, Conversion of nicotine to nornicotine in harvested tobacco: Fate of the methyl group, Plant Physiol., 1961, 36, 592–597 CrossRef CAS PubMed.
- N. Moghbel, B. Ryu, A. Ratsch and K. J. Steadman, Nicotine alkaloid levels, and nicotine to nornicotine conversion, in Australian Nicotiana species used as chewing tobacco, Heliyon, 2017, 3, e00469 CrossRef PubMed.
- B. Cai, B. Siminszky, J. Chappell, R. E. Dewey and L. P. Bush, Enantioselective demethylation of nicotine as a mechanism for variable nornicotine composition in tobacco leaf, J. Biol. Chem., 2012, 287, 42804–42811 CrossRef CAS PubMed.
- H. Destaillats, M. M. Lunden, B. C. Singer, B. K. Coleman, A. T. Hodgson, C. J. Weschler and W. W. Nazaroff, Indoor secondary pollutants from household product emissions in the presence of ozone: A bench-scale chamber study, Environ. Sci. Technol., 2006, 40, 4421–4428 CrossRef CAS PubMed.
- Y. Gong, Z. Chen and H. Li, The oxidation regime and SOA composition in limonene ozonolysis: Roles of different double bonds, radicals, and water, Atmos. Chem. Phys., 2018, 18, 15105–15123 CrossRef CAS.
- E. Praske, R. V. Otkjær, J. D. Crounse, J. C. Hethcox, B. M. Stoltz, H. G. Kjaergaard and P. O. Wennberg, Atmospheric autoxidation is increasingly important in urban and suburban North America, Proc. Natl. Acad. Sci. U. S. A., 2018, 115, 64–69 CrossRef CAS PubMed.
- J. D. Crounse, L. B. Nielsen, S. Jørgensen, H. G. Kjaergaard and P. O. Wennberg, Autoxidation of organic compounds in the atmosphere, J. Phys. Chem. Lett., 2013, 4, 3513–3520 CrossRef CAS.
- L. Onel, L. Thonger, M. A. Blitz, P. W. Seakins, A. J. Bunkan, M. Solimannejad and C. J. Nielsen, Gas-phase reactions of OH with methyl amines in the presence or absence of molecular oxygen. An experimental and theoretical study, J. Phys. Chem. A, 2013, 117, 10736–10745 CrossRef CAS PubMed.
- C. J. Nielsen, H. Herrmann and C. Weller, Atmospheric chemistry and environmental impact of the use of amines in carbon capture and storage (CCS), Chem. Soc. Rev., 2012, 41, 6684–6704 RSC.
- G. Hasan, V.-T. Salo, R. R. Valiev, J. Kubečka and T. Kurtén, Comparing reaction routes for 3(RO⋯ OR) intermediates formed in peroxy radical self- and cross-reactions, J. Phys. Chem. A, 2020, 124, 8305–8320 CrossRef CAS PubMed.
- J. C. Day, Imidyl radicals. The chemistries of 1,8-Naphthalenedicarboximidyl and phthalimidyl radicals, J. Org. Chem., 1986, 51(25), 4959–4963 CrossRef CAS.
- H. Bartling, A. Eisenhofer, B. König and R. M. Gschwind, The photocatalyzed Aza-Henry reaction of N-Aryltetrahydroisoquinolines: Comprehensive mechanism, H•- versus H+-abstraction, and background reactions, J. Am. Chem. Soc., 2016, 138, 11860–11871 CrossRef CAS PubMed.
- J. W. Gorrod and G. Aislaitner, The metabolism of alicyclic amines to reactive iminium ion intermediates, Eur. J. Drug Metab. Pharmacokinet., 1994, 19, 209–217 CrossRef CAS PubMed.
- B. Nozière and D. R. Hanson, Speciated monitoring of gas-phase organic peroxy radicals by chemical ionization mass spectrometry: Cross-reactions between CH3O2, CH3(CO)O2, (CH3)3CO2, and c-C6H11O2, J. Phys. Chem. A, 2017, 121, 8453–8464 CrossRef PubMed.
- J. G. Lisko, H. Tran, S. B. Stanfill, B. C. Blount and C. H. Watson, Chemical composition and evaluation of nicotine, tobacco alkaloids, pH, and selected flavors in E-cigarette cartridges and refill solutions, Nicot Tobacco Res., 2015, 17, 1270–1278 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |