 Open Access Article
Open Access ArticleHydroxide-promoted transformation of fluorotelomer carboxylic acids at ambient temperature
Liliya Chernysheva ,
Elizabeth G. Curtis
,
Elizabeth G. Curtis and
Kyle Doudrick
and
Kyle Doudrick *
*
Department of Civil and Environmental Engineering and Earth Sciences, University of Notre Dame, Notre Dame, Indiana 46556, USA. E-mail: kdoudrick@nd.edu
First published on 24th April 2026
Abstract
Fluorotelomer carboxylic acids (FTCAs) are key intermediates in the environmental transformation of fluorotelomer-based precursors, yet their abiotic degradation pathways remain poorly understood. This study investigates the hydroxide-promoted transformation of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCAs at ambient temperature and under controlled laboratory conditions to elucidate reaction mechanisms and kinetics. Equilibrium experiments were performed across sodium hydroxide (NaOH) concentrations ranging from 1 × 10−5 to 1 M, revealing transformation onset between 1 × 10−4 and 2 × 10−4 M. Both 6
2 FTCAs at ambient temperature and under controlled laboratory conditions to elucidate reaction mechanisms and kinetics. Equilibrium experiments were performed across sodium hydroxide (NaOH) concentrations ranging from 1 × 10−5 to 1 M, revealing transformation onset between 1 × 10−4 and 2 × 10−4 M. Both 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 8
2 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA were fully converted to the corresponding unsaturated products (FTUCAs) at ≥5 × 10−4 M NaOH, followed by secondary loss of FTUCA at higher base concentrations (>2 × 10−3 M) to undetected nontarget products. Minor yields (<7%) of perfluorocarboxylic acids (PFCAs) were detected at ≥0.1 M NaOH. Kinetic experiments at 1 × 10−2 M NaOH showed first-order transformation of 6
2 FTCA were fully converted to the corresponding unsaturated products (FTUCAs) at ≥5 × 10−4 M NaOH, followed by secondary loss of FTUCA at higher base concentrations (>2 × 10−3 M) to undetected nontarget products. Minor yields (<7%) of perfluorocarboxylic acids (PFCAs) were detected at ≥0.1 M NaOH. Kinetic experiments at 1 × 10−2 M NaOH showed first-order transformation of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 8
2 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, with observed rate constants (kobs) of 0.09 and 0.48 h−1, respectively. When repeated with 0.3% ammonia (NH3; [OH−] ∼ 1.74 × 10−3 M) and 6
2 FTCA, with observed rate constants (kobs) of 0.09 and 0.48 h−1, respectively. When repeated with 0.3% ammonia (NH3; [OH−] ∼ 1.74 × 10−3 M) and 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, the kobs decreased sixfold (0.015 h−1) relative to NaOH, consistent with the approximately sixfold lower [OH−]. Because 0.3% NH3 is commonly used in PFAS extraction methods, these results suggest that prolonged extractions may lead to underestimation of FTCAs and potentially other precursors. Experiments with a base-containing consumer cleaning product confirmed that this hydroxide-promoted transformation of 6
2 FTCA, the kobs decreased sixfold (0.015 h−1) relative to NaOH, consistent with the approximately sixfold lower [OH−]. Because 0.3% NH3 is commonly used in PFAS extraction methods, these results suggest that prolonged extractions may lead to underestimation of FTCAs and potentially other precursors. Experiments with a base-containing consumer cleaning product confirmed that this hydroxide-promoted transformation of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA to 6
2 FTCA to 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA also occurs in more complex matrices. Mechanistic analysis supports a reversible E1cb pathway in which deprotonation of the α-hydrogen generates a stabilized carbanion intermediate, followed by unimolecular C–F bond cleavage. This study provides the first evidence of abiotic FTCA transformation via an E1cb mechanism and highlights the potential for mild alkaline environments, including those in analytical and household contexts, to promote PFAS precursor transformation.
2 FTUCA also occurs in more complex matrices. Mechanistic analysis supports a reversible E1cb pathway in which deprotonation of the α-hydrogen generates a stabilized carbanion intermediate, followed by unimolecular C–F bond cleavage. This study provides the first evidence of abiotic FTCA transformation via an E1cb mechanism and highlights the potential for mild alkaline environments, including those in analytical and household contexts, to promote PFAS precursor transformation.
Environmental significanceFluorotelomer carboxylic acids (FTCAs) are key intermediates in the environmental transformation of fluorotelomer-based PFAS, yet their abiotic reactivity has remained unclear. This study demonstrates, for the first time, that FTCAs undergo hydroxide-promoted transformation via an E1cb elimination mechanism even at ambient temperature. The results reveal that common mildly alkaline conditions, such as those encountered in alkaline pH environments, during laboratory extractions, or even household cleaning, can drive precursor conversion to more stable PFAS. These findings expand current understanding of PFAS abiotic transformation pathways and highlight the potential for routine environmental and analytical processes to alter PFAS speciation and mass balance assessments. |
1. Introduction
Fluorotelomer carboxylic acids (FTCAs) are a class of polyfluorinated alkyl acids that are found in the environment and primarily from both biotic and abiotic degradation pathways of precursor compounds.1–7 Phillips et al. reported that FTCAs exhibit greater toxicity than their perfluorinated analogs.6,8 Although abiotic transformation of FTCAs has been studied only sparingly, biotic transformation has been investigated extensively. Hagen et al. were among the first to demonstrate that fluorotelomer alcohols (FTOHs) degrade to form FTCAs and ultimately perfluorooctanoic acid (PFOA) in rats.9 Subsequent studies have confirmed similar biotic transformation pathways across a range of matrices, including human blood,10,11 rats and mice,12,13 fish,5,14,15 plants,6 fungi,16 landfills,17 wastewater treatment plants (WWTPs),18–24 and soil.25,26 Frequently detected transformation products include even and odd-chain FTCAs, fluorotelomer unsaturated carboxylic acids (FTUCAs), and several perfluorocarboxylic acids (PFCAs).13,27 Transformation of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA is consistently accompanied by the formation of the corresponding n
2 FTCA is consistently accompanied by the formation of the corresponding n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA as a major product.6,28,29
2 FTUCA as a major product.6,28,29
Abiotic transformation of fluorotelomer precursors has received less attention than biotic pathways, with most studies focused on engineered systems such as ultraviolet photolysis, advanced oxidation processes, or alkaline thermal reactions.30–35 For FTCAs in particular, no studies have explicitly examined their abiotic transformation mechanisms. Loewen et al.29 observed that n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA converts to n
2 FTCA converts to n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA in methanol but not in Optima-grade water, with yields increasing at higher temperatures (4 and 20 °C). The reaction was relatively slow, producing only picomole-scale amounts of product over 10 days. The removal of the α-hydrogen and the β-fluorine (i.e., the leaving group) to form the unsaturated product is indicative of an elimination reaction. Because the C–F is a poor leaving group and C–H typically has a high pKa (>50), we initially hypothesized the reaction proceeds via an E2 mechanism. However, given that the experimental conditions were only mildly basic (i.e., methoxide ∼ 10−8.5 M) and near ambient temperature, an E2 pathway, while possible, would be kinetically unfavorable.
2 FTUCA in methanol but not in Optima-grade water, with yields increasing at higher temperatures (4 and 20 °C). The reaction was relatively slow, producing only picomole-scale amounts of product over 10 days. The removal of the α-hydrogen and the β-fluorine (i.e., the leaving group) to form the unsaturated product is indicative of an elimination reaction. Because the C–F is a poor leaving group and C–H typically has a high pKa (>50), we initially hypothesized the reaction proceeds via an E2 mechanism. However, given that the experimental conditions were only mildly basic (i.e., methoxide ∼ 10−8.5 M) and near ambient temperature, an E2 pathway, while possible, would be kinetically unfavorable.
The objective of this study was to investigate the hydroxide-promoted elimination of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA under controlled alkaline conditions. Equilibrium experiments were conducted across a range of sodium hydroxide (NaOH) concentrations to quantify the formation of the unsaturated product, n
2 FTCA under controlled alkaline conditions. Equilibrium experiments were conducted across a range of sodium hydroxide (NaOH) concentrations to quantify the formation of the unsaturated product, n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA, and its disappearance. Reaction kinetics were then evaluated using both NaOH and ammonia (NH3) to assess the influence of base strength and identity. Finally, transformation was demonstrated by spiking n
2 FTUCA, and its disappearance. Reaction kinetics were then evaluated using both NaOH and ammonia (NH3) to assess the influence of base strength and identity. Finally, transformation was demonstrated by spiking n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA into a commercially available base-containing cleaning agent, and the combined results were used to propose a mechanistic framework for FTCA elimination.
2 FTCA into a commercially available base-containing cleaning agent, and the combined results were used to propose a mechanistic framework for FTCA elimination.
2. Methods
2.1. Transformation reaction experiments
Single-compound stock solutions of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA (97%, SynQuest), 6
2 FTCA (97%, SynQuest), 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA (95%, SynQuest), 7
2 FTUCA (95%, SynQuest), 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCA (97%, SynQuest), and 8
3 FTCA (97%, SynQuest), and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA (97%, SynQuest) were prepared in 18.2 MΩ cm ultrapure water (UPW) from pure compound. All sample containers were first cleaned with Optima methanol (MeOH; ≥99.8% LC-MS grade; VWR). Each stock was serially diluted in UPW within 15 mL polypropylene centrifuge vials (VWR) to yield working solutions at a target substrate concentration of 0.2 mg L−1. An aqueous sodium hydroxide (NaOH; 98% pellets; Acros) solution was then added to reach a final volume of 10 mL, leaving sufficient headspace for mixing. Sodium hydroxide solutions were prepared in UPW from anhydrous solid.
2 FTCA (97%, SynQuest) were prepared in 18.2 MΩ cm ultrapure water (UPW) from pure compound. All sample containers were first cleaned with Optima methanol (MeOH; ≥99.8% LC-MS grade; VWR). Each stock was serially diluted in UPW within 15 mL polypropylene centrifuge vials (VWR) to yield working solutions at a target substrate concentration of 0.2 mg L−1. An aqueous sodium hydroxide (NaOH; 98% pellets; Acros) solution was then added to reach a final volume of 10 mL, leaving sufficient headspace for mixing. Sodium hydroxide solutions were prepared in UPW from anhydrous solid.
Final concentrations in the equilibrium experiments ranged from 1 × 10−5 to 1 M. Equilibrium samples were capped, sealed with Parafilm, and placed on a rotator for passive mixing over 30 days. Samples were taken at 14 days and 30 days to confirm equilibrium was reached (<5% change in measured concentration). Initial concentration (C0) samples of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA with no added NaOH were prepared concurrently with NaOH-containing samples and aged under identical conditions. Prior to liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis, samples were diluted ten-fold to an 80
2 FTCA with no added NaOH were prepared concurrently with NaOH-containing samples and aged under identical conditions. Prior to liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis, samples were diluted ten-fold to an 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 (v/v) methanol
20 (v/v) methanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water solvent ratio without neutralization. All equilibrium experiments were conducted in triplicate, unless otherwise noted. Samples were stored at 4 °C until further analysis.
water solvent ratio without neutralization. All equilibrium experiments were conducted in triplicate, unless otherwise noted. Samples were stored at 4 °C until further analysis.
Kinetic experiments followed a similar protocol but were designed to minimize external mixing. Samples were not vortexed or rotated to avoid enhancing reaction rates via mechanical agitation. Sodium hydroxide kinetic experiments were performed at a fixed base concentration of 1 × 10−2 M NaOH in each sample. For ammonium hydroxide (NH4OH; 28–30% as NH3; VWR) experiments, a 30% stock solution (∼15.88 M NH3) was diluted in UPW to achieve a final concentration of 0.30%, matching that of the US EPA Method 1633.36 Kinetic experiments were conducted at discrete time points (i.e., one sample per time point). At the predefined time points (5 s, 15 s, 30 s, 45 s, 1 min, 5 min, 15 min, 30 min, 1 h, 2 h, 4 h, 8 h, 18 h, and 24 h), samples were neutralized with 0.10 mL of 20% (v/v) acetic acid (HAc; glacial; Fisher) to stop the reaction. The resulting pH was verified using a pH probe (Orion 9103BNWP Semi-Micro Combination pH), and it consistently reached pH ∼ 4.75 regardless of the substrate or base concentration. For kinetics studies, two C0 samples were obtained: one matching the equilibrium C0 samples (substrate in UPW) and another containing substrate and HAc with no additional hydroxide (i.e., experimental control). No substantial (>10%) change in FTCA concentration was observed in the control. All NaOH kinetics experiments were conducted in triplicate, and NH3 experiments once.
Equilibrium transformation of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA with a consumer-grade cleaning agent (unnamed) followed a similar setup to equilibrium tests with NaOH. Product selection was driven by analytical feasibility: many commercial formulations contain complex mixtures that interfere with LC-MS/MS analysis and required extensive cleanup beyond simple dilution. The selected product was a multi-purpose concentrate marketed for use on countertops, wood, tile, linoleum, and bathroom surfaces, and intended to be diluted prior to use. Varying water diluted concentrations of the cleaning agent, ranging from 0.1–50% v/v, were spiked with 0.2 mg L−1 of 6
2 FTCA with a consumer-grade cleaning agent (unnamed) followed a similar setup to equilibrium tests with NaOH. Product selection was driven by analytical feasibility: many commercial formulations contain complex mixtures that interfere with LC-MS/MS analysis and required extensive cleanup beyond simple dilution. The selected product was a multi-purpose concentrate marketed for use on countertops, wood, tile, linoleum, and bathroom surfaces, and intended to be diluted prior to use. Varying water diluted concentrations of the cleaning agent, ranging from 0.1–50% v/v, were spiked with 0.2 mg L−1 of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA. Concentrations between 0.1–5% reflected typical consumer use, while 10–50% served as an additional comparison. Experiments were conducted in 20 mL glass vials at a volume of 10 mL for 7 days. Samples were processed through a weak anion exchange (WAX) solid-phase extraction (SPE) column prior to LC-MS/MS analysis, following EPA Method 1633. An extraction internal standard (EIS, 13C5-PFHxA, Wellington) was added before the SPE step to track losses from sample processing. SPE was followed by elution with 5 mL MeOH and acidification with 25 µL of 20% HAc. Final extracts were diluted 10-fold to an 80
2 FTCA. Concentrations between 0.1–5% reflected typical consumer use, while 10–50% served as an additional comparison. Experiments were conducted in 20 mL glass vials at a volume of 10 mL for 7 days. Samples were processed through a weak anion exchange (WAX) solid-phase extraction (SPE) column prior to LC-MS/MS analysis, following EPA Method 1633. An extraction internal standard (EIS, 13C5-PFHxA, Wellington) was added before the SPE step to track losses from sample processing. SPE was followed by elution with 5 mL MeOH and acidification with 25 µL of 20% HAc. Final extracts were diluted 10-fold to an 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 MeOH
20 MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O matrix, and a non-extraction internal standard (NIS; MPFAC-HIF-IS, Wellington) was added directly to the LC-MS/MS sample vial to monitor instrument performance. A C0 sample containing 6
H2O matrix, and a non-extraction internal standard (NIS; MPFAC-HIF-IS, Wellington) was added directly to the LC-MS/MS sample vial to monitor instrument performance. A C0 sample containing 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, EIS, and NIS without the cleaning agent served as a baseline for evaluating transformation and recovery. The selection of 13C5-PFHxA as EIS was based on the US EPA Method 1633, which designates 13C5-PFHxA as the EIS for 5
2 FTCA, EIS, and NIS without the cleaning agent served as a baseline for evaluating transformation and recovery. The selection of 13C5-PFHxA as EIS was based on the US EPA Method 1633, which designates 13C5-PFHxA as the EIS for 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCA37 – the most structurally similar target analyte to 6
3 FTCA37 – the most structurally similar target analyte to 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA. EIS recoveries were 99%, 90%, and 89% for samples containing 0% (i.e., C0), 0.1%, and 2% cleaning agent. At 5% the recovery was only 22% and it got progressively worse with increasing concentration of cleaning agent.
2 FTCA. EIS recoveries were 99%, 90%, and 89% for samples containing 0% (i.e., C0), 0.1%, and 2% cleaning agent. At 5% the recovery was only 22% and it got progressively worse with increasing concentration of cleaning agent.
2.2. Analytics
All PFAS analysis was conducted on an ultra-performance liquid chromatography (UPLC) system (Agilent, 1290 Infinity II LC) coupled to a triple quadrupole mass spectrometer (MS/MS) (Agilent, MS/MS G6470B) operated in the negative ion mode (Agilent, electrospray ion source G1958-65638). PFAS were separated on a ZORBAX RRHD Eclipse Plus column (Agilent, 959758-902; C18, 95 Å, 2.1 × 100 mm, 1.8 µm, 1200 bar). The column was kept at 40 °C with the gradient elution flow rate of 0.4 mL min−1 for 17 min. A PFAS Delay Column (Agilent, 5062-8100; 4.6 × 30 mm, 1200 bar) that sits between the solvent mixer and the injector was used to remove impurities coming from solvents. A PFAS-free LC Conversion Kit was used to replace the original PTFE-containing fittings on the UPLC (Agilent, 5004-0006). Samples were prepared in 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 MeOH/H2O in 300 µL polypropylene vials (9532S-MS-K; Microsolv) with polypropylene screw-top caps (9502S-09-PE; Microsolv). The sample injection volume was 10 µL. The gradient elution started with a solvent mixture of 90% UPW and 10% MeOH for 1 min. After 1 min, the MeOH concentration was increased to 40% and reached 90% linearly for 25 min. After 25 min, the MeOH concentration was decreased to 10%.
20 MeOH/H2O in 300 µL polypropylene vials (9532S-MS-K; Microsolv) with polypropylene screw-top caps (9502S-09-PE; Microsolv). The sample injection volume was 10 µL. The gradient elution started with a solvent mixture of 90% UPW and 10% MeOH for 1 min. After 1 min, the MeOH concentration was increased to 40% and reached 90% linearly for 25 min. After 25 min, the MeOH concentration was decreased to 10%.
Targeted analysis identified 44 potential compounds, including 40 from the US EPA Method 1633 analyte list (including, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCA, 5
3 FTCA, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCA, 7
3 FTCA, 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCA and 8
3 FTCA and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS) and four additional compounds: 6
2 FTS) and four additional compounds: 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, 6
2 FTCA, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA, 8
2 FTUCA, 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, and 8
2 FTCA, and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA. US EPA Method 1633 analytical standards were used because they were available from concurrent experiments rather than specifically purchased for this study. These standards were analyzed as a single mix of 40 PFAS, prepared from five separate native standard mixes (PFAC–FMXF, PFAC–FMXG, PFAC–FMCH, PFAC–FMXI, PFAC–FMXJ, FHEA, FHUEA, FOEA, and FOUEA; Wellington). The four additional precursors were split into two separate mixes: one containing 6
2 FTUCA. US EPA Method 1633 analytical standards were used because they were available from concurrent experiments rather than specifically purchased for this study. These standards were analyzed as a single mix of 40 PFAS, prepared from five separate native standard mixes (PFAC–FMXF, PFAC–FMXG, PFAC–FMCH, PFAC–FMXI, PFAC–FMXJ, FHEA, FHUEA, FOEA, and FOUEA; Wellington). The four additional precursors were split into two separate mixes: one containing 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 8
2 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCAs, and the other containing 6
2 FTCAs, and the other containing 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 8
2 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCAs.
2 FTUCAs.
The freshness of the n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA analytical standards was critical, as n
2 FTCA analytical standards was critical, as n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA gradually transformed into n
2 FTCA gradually transformed into n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA when stored in 80
2 FTUCA when stored in 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 MeOH/H2O at 4 °C. Notably, n
20 MeOH/H2O at 4 °C. Notably, n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA was already present in the n
2 FTUCA was already present in the n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA standards, and its relative abundance increased over the analysis period, consistent with prior observations by Loewen et al. (2005) in 100% MeOH at 4 °C and 20 °C. To distinguish between transformation-derived and native n
2 FTCA standards, and its relative abundance increased over the analysis period, consistent with prior observations by Loewen et al. (2005) in 100% MeOH at 4 °C and 20 °C. To distinguish between transformation-derived and native n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA, the n
2 FTUCA, the n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and n
2 FTCA and n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA compounds were prepared in separate standard mixes. Analytical standards were refrigerated for ∼1 month prior to use, though exact synthesis and packaging dates were unknown, potentially contributing to untracked degradation. LC-MS/MS data were corrected using daily quality control responses, and replicate samples were analyzed on the same day to minimize intraday variability.
2 FTUCA compounds were prepared in separate standard mixes. Analytical standards were refrigerated for ∼1 month prior to use, though exact synthesis and packaging dates were unknown, potentially contributing to untracked degradation. LC-MS/MS data were corrected using daily quality control responses, and replicate samples were analyzed on the same day to minimize intraday variability.
Potential analytical interference on LC-MS/MS from the Na+ ion in NaOH and HAc introduced during the neutralization of kinetic experiments was assessed. 1 × 10−1 M sodium chloride (NaCl; VWR) was used to simulate Na+ ion levels, reflecting the ten-fold dilution of the highest NaOH concentration (1 M). Similarly, HAc interference was evaluated at 0.1% (v/v), corresponding to 1.7 × 10−2 M. Across all tests, recoveries for 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 8
2 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and FTUCA ranged from 92–104% for NaCl and 94–106% for HAc, indicating minimal analytical interference. As a result, interference from Na+ and HAc was considered negligible, and no adjustments were made to the data to account for these effects.
2 FTCA and FTUCA ranged from 92–104% for NaCl and 94–106% for HAc, indicating minimal analytical interference. As a result, interference from Na+ and HAc was considered negligible, and no adjustments were made to the data to account for these effects.
Combustion ion chromatography (CIC) was conducted to assess total F concentrations (Metrohm Profiler F). Samples were combusted at 1050 °C. Oxygen was injected at a flow rate of 300 mL min−1 and argon was used as a carrier gas at a 100 mL min−1 rate to direct the off gas into the absorption liquid.
3. Results and discussion
3.1. Transformation of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 2 FTCA at equilibrium
2 FTCA at equilibrium
The concentration of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and its unsaturated transformation product, n
2 FTCA and its unsaturated transformation product, n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA, were measured as a function of the NaOH concentration (1 × 10−5 to 1 M) at equilibrium (Fig. 1). For both 6
2 FTUCA, were measured as a function of the NaOH concentration (1 × 10−5 to 1 M) at equilibrium (Fig. 1). For both 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and 8
2 FTCA and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, the transformation is initiated between an NaOH concentration of 1 × 10−4 and 2 × 10−4 M. At 5 × 10−4 M, all n
2 FTCA, the transformation is initiated between an NaOH concentration of 1 × 10−4 and 2 × 10−4 M. At 5 × 10−4 M, all n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA had been transformed, at least partially, into n
2 FTCA had been transformed, at least partially, into n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA. With further increases in base concentration (>2 × 10−3 M NaOH), n
2 FTUCA. With further increases in base concentration (>2 × 10−3 M NaOH), n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA concentrations declined until the compound was no longer detectable, suggesting secondary transformation to one or more non-target products not captured by the analytical method used. Total F analysis for the 6
2 FTUCA concentrations declined until the compound was no longer detectable, suggesting secondary transformation to one or more non-target products not captured by the analytical method used. Total F analysis for the 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA experiment revealed that the F mass balance was well maintained up to 1 × 10−1 M (Fig. S1), at which point FTCA and FTUCA was not detectable despite approximately 80% of the total F still being accounted for. This suggests that the final transformation products of 6
2 FTCA experiment revealed that the F mass balance was well maintained up to 1 × 10−1 M (Fig. S1), at which point FTCA and FTUCA was not detectable despite approximately 80% of the total F still being accounted for. This suggests that the final transformation products of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA are a mixture of soluble and (semi-)volatile compounds.
2 FTUCA are a mixture of soluble and (semi-)volatile compounds.
Overall, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and 8
2 FTCA and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA exhibited nearly identical behavior. As NaOH concentration increased, the extent of n
2 FTCA exhibited nearly identical behavior. As NaOH concentration increased, the extent of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA loss and n
2 FTCA loss and n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA formation increased proportionally. Between 1 × 10−5 M and 2 × 10−4 M NaOH, n
2 FTUCA formation increased proportionally. Between 1 × 10−5 M and 2 × 10−4 M NaOH, n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA was still the dominant species in solution based on the molar mass balance. Around 2 × 10−4 M NaOH, the two species were approximately equivalent in abundance, beyond which n
2 FTCA was still the dominant species in solution based on the molar mass balance. Around 2 × 10−4 M NaOH, the two species were approximately equivalent in abundance, beyond which n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA predominated while n
2 FTUCA predominated while n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA continued to decline. At 5 × 10−4 M NaOH, n
2 FTCA continued to decline. At 5 × 10−4 M NaOH, n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA was below the detection limit and n
2 FTCA was below the detection limit and n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA was the only species identified in solution. Between 5 × 10−4 M and 5 × 10−3 M NaOH, n
2 FTUCA was the only species identified in solution. Between 5 × 10−4 M and 5 × 10−3 M NaOH, n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA concentrations plateaued, with the highest yields observed at 1 × 10−3 M and 2 × 10−3 M NaOH. At 1 × 10−2 M NaOH, n
2 FTUCA concentrations plateaued, with the highest yields observed at 1 × 10−3 M and 2 × 10−3 M NaOH. At 1 × 10−2 M NaOH, n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA began to decline and was undetectable at 1 × 10−1 M and 1 M NaOH. No detectable levels of 3
2 FTUCA began to decline and was undetectable at 1 × 10−1 M and 1 M NaOH. No detectable levels of 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 5
3, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, or 7
3, or 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCA were observed in any samples. The potential presence of the corresponding unsaturated homologues (3
3 FTCA were observed in any samples. The potential presence of the corresponding unsaturated homologues (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 5
3, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, or 7
3, or 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTUCA) cannot be ruled out because these were not included in the analytical method.12
3 FTUCA) cannot be ruled out because these were not included in the analytical method.12
Possible PFCA products were also examined, given that prior studies have reported PFCAs as end-products in n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA degradation pathways.3,38,39 Analyses focused on the samples where n
2 FTCA degradation pathways.3,38,39 Analyses focused on the samples where n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and n
2 FTCA and n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA were largely degraded, specifically those treated with 1 × 10−1 M and 1 M NaOH. For 6
2 FTUCA were largely degraded, specifically those treated with 1 × 10−1 M and 1 M NaOH. For 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, perfluorohexanoic acid (PFHxA) was undetectable in the C0 sample, detected at approximately 0.9% of the starting molar mass (<LOQ) at 1 × 10−1 M NaOH, and quantified at ∼6.6% at 1 M NaOH. A similar pattern was observed for 6
2 FTCA, perfluorohexanoic acid (PFHxA) was undetectable in the C0 sample, detected at approximately 0.9% of the starting molar mass (<LOQ) at 1 × 10−1 M NaOH, and quantified at ∼6.6% at 1 M NaOH. A similar pattern was observed for 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA, where PFHxA accounted for ∼0.6% (<LOQ) at 1 × 10−1 M NaOH and ∼4.1% at 1 M NaOH. Perfluoroheptanoic acid (PFHpA) was present in the C0 samples of both 6
2 FTUCA, where PFHxA accounted for ∼0.6% (<LOQ) at 1 × 10−1 M NaOH and ∼4.1% at 1 M NaOH. Perfluoroheptanoic acid (PFHpA) was present in the C0 samples of both 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and 6
2 FTCA and 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA and remained unchanged with NaOH treatment, indicating a contaminant source rather than a transformation product. For 8
2 FTUCA and remained unchanged with NaOH treatment, indicating a contaminant source rather than a transformation product. For 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, perfluorooctanoic acid (PFOA) was absent in the C0 sample, detected at ∼0.2% of the initial molar mass (<LOQ) at 1 × 10−1 M NaOH, and reached ∼1.6% at 1 M NaOH. No other PFCAs were detected in any sample. These results indicate that PFCA formation occurs only to a minor extent under the tested conditions.
2 FTCA, perfluorooctanoic acid (PFOA) was absent in the C0 sample, detected at ∼0.2% of the initial molar mass (<LOQ) at 1 × 10−1 M NaOH, and reached ∼1.6% at 1 M NaOH. No other PFCAs were detected in any sample. These results indicate that PFCA formation occurs only to a minor extent under the tested conditions.
Although n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCAs (e.g., 3
3 FTCAs (e.g., 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 5
3, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and 7
3, and 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) have been reported as byproducts in microbial biotransformation of fluorotelomer acids and related precursors,39 none of these odd-chain FTCAs that we targeted (3
3) have been reported as byproducts in microbial biotransformation of fluorotelomer acids and related precursors,39 none of these odd-chain FTCAs that we targeted (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 5
3, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and 7
3, and 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCA) were detected in our experiments at 0.1 and 1 M NaOH where transformation of FTUCA was most pronounced. Formation of n
3 FTCA) were detected in our experiments at 0.1 and 1 M NaOH where transformation of FTUCA was most pronounced. Formation of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 FTCAs in biological systems is generally attributed to enzyme-mediated pathways capable of C–C bond cleavage, oxidation, and backbone rearrangement, such as β-oxidation like processes and other catalytic transformations. These mechanisms differ fundamentally from hydroxide-promoted reactions, which are limited to acid–base and elimination chemistry under the conditions investigated here. In the absence of enzymatic catalysis, strong oxidants, photochemical activation, or elevated temperatures, C–C bond scission within the fluorotelomer backbone is not expected to occur readily under ambient alkaline conditions. Consequently, while odd-chain FTCAs are plausible products in biotic systems, their formation is not expected under the conditions tested herein.
3 FTCAs in biological systems is generally attributed to enzyme-mediated pathways capable of C–C bond cleavage, oxidation, and backbone rearrangement, such as β-oxidation like processes and other catalytic transformations. These mechanisms differ fundamentally from hydroxide-promoted reactions, which are limited to acid–base and elimination chemistry under the conditions investigated here. In the absence of enzymatic catalysis, strong oxidants, photochemical activation, or elevated temperatures, C–C bond scission within the fluorotelomer backbone is not expected to occur readily under ambient alkaline conditions. Consequently, while odd-chain FTCAs are plausible products in biotic systems, their formation is not expected under the conditions tested herein.
3.2. Transformation kinetics of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 2 FTCA with NaOH
2 FTCA with NaOH
After confirming n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA transformation under equilibrium conditions, reaction kinetics were examined to determine transformation rates and apparent reaction order. Results for 0.2 mg per L n
2 FTCA transformation under equilibrium conditions, reaction kinetics were examined to determine transformation rates and apparent reaction order. Results for 0.2 mg per L n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA reacted with 1 × 10−2 M NaOH are shown in Fig. 2. Both 6
2 FTCA reacted with 1 × 10−2 M NaOH are shown in Fig. 2. Both 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and 8
2 FTCA and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA displayed similar temporal trends and followed apparent first-order kinetics with respect to FTCA loss. The observed rate constants (kobs) were 0.09 h−1 for 6
2 FTCA displayed similar temporal trends and followed apparent first-order kinetics with respect to FTCA loss. The observed rate constants (kobs) were 0.09 h−1 for 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and 0.48 h−1 for 8
2 FTCA and 0.48 h−1 for 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, indicating that 8
2 FTCA, indicating that 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA degraded approximately five times faster than 6
2 FTCA degraded approximately five times faster than 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA.
2 FTCA.
 | ||
Fig. 2 Transformation kinetics of 0.2 mg per L n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA to n 2 FTCA to n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA in the presence of 1 × 10−2 M NaOH during the first 24 hours for (A) 6 2 FTUCA in the presence of 1 × 10−2 M NaOH during the first 24 hours for (A) 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and (B) 8 2 FTCA and (B) 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA. 2 FTCA. | ||
The faster transformation kinetics observed for 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA relative to 6
2 FTCA relative to 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA indicate that chain length exerts a strong control on the likelihood and rate of HF elimination under alkaline conditions. This trend is consistent with recent observations from microbial transformation studies,39 which also report enhanced reactivity for longer-chain fluorotelomer acids. Collectively, these results suggest that chain length governs both abiotic and biotic transformation propensity, with longer-chain fluorotelomer acids being more susceptible to HF elimination than their shorter-chain analogues.
2 FTCA indicate that chain length exerts a strong control on the likelihood and rate of HF elimination under alkaline conditions. This trend is consistent with recent observations from microbial transformation studies,39 which also report enhanced reactivity for longer-chain fluorotelomer acids. Collectively, these results suggest that chain length governs both abiotic and biotic transformation propensity, with longer-chain fluorotelomer acids being more susceptible to HF elimination than their shorter-chain analogues.
Concentrations of the corresponding unsaturated products (n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA) exhibited transient rise profiles that did not fit simple first- or second-order models, consistent with their simultaneous formation and subsequent transformation observed under equilibrium conditions (Fig. 1). The initial rate of 8
2 FTUCA) exhibited transient rise profiles that did not fit simple first- or second-order models, consistent with their simultaneous formation and subsequent transformation observed under equilibrium conditions (Fig. 1). The initial rate of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA formation (∼48 nM h−1) was slower than that of 6
2 FTUCA formation (∼48 nM h−1) was slower than that of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA (∼86 nM h−1), suggesting that 8
2 FTUCA (∼86 nM h−1), suggesting that 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA undergoes faster secondary reactions to nontarget by-products, resulting in lower steady-state accumulation.
2 FTUCA undergoes faster secondary reactions to nontarget by-products, resulting in lower steady-state accumulation.
3.3. Transformation kinetics of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 2 FTCA with NH3
2 FTCA with NH3
Kinetic experiments were also performed with 0.3% NH3 (corresponding to approximately 1.74 × 10−3 M OH−, 1.74 × 10−3 M NH4+, 1.56 × 10−1 M NH3, and 6.34 × 10−12 M H+) (Fig. 3). The observed first-order rate constant for NH3 (kobs = 0.015 h−1) was sixfold lower than that measured for NaOH at 1 × 10−2 M (kobs = 0.09 h−1). This difference corresponds directly to the approximately sixfold lower [OH−], indicating that the transformation rate scales linearly with [OH−]. The conjugate-acid pKa of NH3 (9.25) is nearly identical to the apparent α-C–H pKa of FTCA determined from equilibrium experiments (∼9–10). This parity implies that NH3 is only marginally strong enough to deprotonate the α-carbon, so deprotonation proceeds almost entirely via the small amount of hydroxide present at equilibrium, resulting in a rate directly proportional to [OH−]. Such proportional behavior indicates that hydroxide concentration, rather than base identity, governs the reaction rate under these conditions.
The 0.3% NH3 concentration is commonly employed in extraction procedures, such as those described in U.S. EPA Method 1633.37 In this method, environmental matrices such as soils and biosolids are extracted three times with 0.3% NH3 in methanol, each extraction lasting 20–40 min, resulting in approximately 2 h of direct contact between PFAS and NH3. Subsequent purification, acidification, and solvent evaporation under heated nitrogen can extend this exposure by an additional 1–3 h. Thus, the conditions used in Method 1633 can promote limited abiotic transformation of certain PFAS precursors. While the conversion of n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA to n
2 FTCA to n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA under these conditions appears to remain below 20%, well within analytical uncertainty for typical extraction durations, the possibility of transformation of more reactive precursors such as n
2 FTUCA under these conditions appears to remain below 20%, well within analytical uncertainty for typical extraction durations, the possibility of transformation of more reactive precursors such as n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 fluorotelomer alcohols (FTOHs) remains uncertain. Because FTOHs are known to be more susceptible to base-promoted reactions than FTCA or FTUCA intermediates, unrecognized conversions during alkaline extraction or evaporation steps could contribute to variability in reported PFAS concentrations.40
2 fluorotelomer alcohols (FTOHs) remains uncertain. Because FTOHs are known to be more susceptible to base-promoted reactions than FTCA or FTUCA intermediates, unrecognized conversions during alkaline extraction or evaporation steps could contribute to variability in reported PFAS concentrations.40
3.4. Transformation of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 2 FTCA with a cleaning agent
2 FTCA with a cleaning agent
To evaluate the potential for n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA transformation under realistic indoor conditions, reactions were conducted using a consumer-grade cleaning product that lists a strong base as one of its primary active ingredients (Fig. 4). The C0 had a 6
2 FTCA transformation under realistic indoor conditions, reactions were conducted using a consumer-grade cleaning product that lists a strong base as one of its primary active ingredients (Fig. 4). The C0 had a 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA concentration of 145 nM and it also contained 4.6 nM of background 6
2 FTCA concentration of 145 nM and it also contained 4.6 nM of background 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA. In the presence of 0.1%, 2%, and 5% cleaner, the 6
2 FTUCA. In the presence of 0.1%, 2%, and 5% cleaner, the 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA concentration decreased to 101 nM (C/C0 = 70%), 53 nM (C/C0 = 37%), and 31 nM (C/C0 = 22%), respectively, while 6
2 FTCA concentration decreased to 101 nM (C/C0 = 70%), 53 nM (C/C0 = 37%), and 31 nM (C/C0 = 22%), respectively, while 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA increased to 34 nM, 66 nM, and decreased to 40 nM. Increasing the concentration of the cleaning agent up to 50% decreased both 6
2 FTUCA increased to 34 nM, 66 nM, and decreased to 40 nM. Increasing the concentration of the cleaning agent up to 50% decreased both 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and 6
2 FTCA and 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA, but these values are not reported quantitatively due to poor EIS recoveries. The decrease in 6
2 FTUCA, but these values are not reported quantitatively due to poor EIS recoveries. The decrease in 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA concentration was not equimolar to the increase in 6
2 FTCA concentration was not equimolar to the increase in 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTUCA, suggesting either incomplete extraction, matrix suppression, or transformation of the FTUCA to additional products not captured by the analytical method. The latter explanation agrees with the equilibrium outcomes in water (Fig. 1). Overall, these findings demonstrate that 6
2 FTUCA, suggesting either incomplete extraction, matrix suppression, or transformation of the FTUCA to additional products not captured by the analytical method. The latter explanation agrees with the equilibrium outcomes in water (Fig. 1). Overall, these findings demonstrate that 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, and by extension, n
2 FTCA, and by extension, n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, can undergo transformation in alkaline consumer cleaning formulations under conditions relevant to indoor use.
2 FTCA, can undergo transformation in alkaline consumer cleaning formulations under conditions relevant to indoor use.
Although cleaner concentrations were chosen to reflect consumer use, the 7 day exposure time does not represent typical contact durations, which are much shorter in real-world cleaning. As a result, the experiment should be interpreted as a proof-of-concept demonstration rather than a kinetic study. The findings nonetheless indicate that under sufficiently basic and prolonged conditions, indoor-use products containing bases could promote abiotic transformation of fluorotelomer-based compounds.
Cleaning products are not the only potential indoor matrices capable of inducing n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA transformation. Zhou et al. (2022) reported substantial PFAS emissions and potential transformation during floor waxing, noting that many acrylic floor polishes contain fluorosurfactants (0.01–0.05% by mass).41 In that study, high airborne PFAS levels were observed during wax stripping, a process that typically employs alkaline wash solutions containing bases such as NaOH, potassium hydroxide (KOH), or ammonium hydroxide (NH4OH). Such conditions may promote hydrolysis or elimination reactions, yielding additional fluorinated transformation products. Future research could examine n
2 FTCA transformation. Zhou et al. (2022) reported substantial PFAS emissions and potential transformation during floor waxing, noting that many acrylic floor polishes contain fluorosurfactants (0.01–0.05% by mass).41 In that study, high airborne PFAS levels were observed during wax stripping, a process that typically employs alkaline wash solutions containing bases such as NaOH, potassium hydroxide (KOH), or ammonium hydroxide (NH4OH). Such conditions may promote hydrolysis or elimination reactions, yielding additional fluorinated transformation products. Future research could examine n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA and other PFAS interactions with wax strippers under controlled laboratory conditions, using air capture or real-time gas-phase analysis to identify volatile intermediates and end-products. However, due to the high complexity and surfactant content of such matrices, extensive purification and cleanup would be necessary for quantitative analysis.
2 FTCA and other PFAS interactions with wax strippers under controlled laboratory conditions, using air capture or real-time gas-phase analysis to identify volatile intermediates and end-products. However, due to the high complexity and surfactant content of such matrices, extensive purification and cleanup would be necessary for quantitative analysis.
3.5. Proposed reaction mechanism
The formation of an unsaturated byproduct indicates that the transformation proceeds via an elimination pathway. Three classical mechanisms are possible: E1, E2, and E1cb. An E1 reaction involves initial cleavage of the C–F bond to form a carbocation intermediate; however, this step has a prohibitively high energy barrier because fluorine is a poor leaving group. Thus, an E1 mechanism can be excluded.An E1cb or E2 mechanism remains plausible. In the E1cb pathway, deprotonation of the α-hydrogen occurs first, forming a carbanion intermediate, whereas in an E2 process, C–H and C–F bond breaking occur concertedly. For ordinary alkanes, α-hydrogen deprotonation is highly unfavorable (pKa > 50), so the unimolecular E2 mechanism tends to dominate. PFAS, however, differ substantially from typical alkanes because the perfluoroalkyl chain exerts a strong electron-withdrawing effect that markedly acidifies the α-hydrogen.
Equilibrium data (Fig. 1) support the substantial acidification of the α-hydrogen, with an onset of transformation between 10−5 and 10−4 M NaOH, corresponding to an apparent pKa of ∼9–10. Under these conditions, several observations collectively support an E1cb mechanism rather than an E2 pathway. First, E2 eliminations are disfavored when the leaving group is poor, as fluorine is. Second, the reaction proceeds in anhydrous methanol, a solvent that is only weakly basic (∼10−8.5 M methoxide), implying that the elimination is driven by intrinsic thermodynamics rather than by a strong external base. Finally, while pseudo-first-order conditions mean that both E1cb and E2 would yield apparent first-order kinetics in FTCA, the NH3 experiments demonstrate that kobs decreased in direct proportion to the reduction in [OH−] with no measurable contribution from the more abundant NH3, consistent with specific-base catalysis in which only OH− participates in the deprotonation step.
The first step of an E1cb reaction is deprotonation of the α-hydrogen, so its pKa becomes the key thermodynamic parameter governing whether a given telomer can undergo transformation under mildly basic conditions. To evaluate this dependence, we conducted an equilibrium experiment with 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS, which is expected to possess a significantly less acidified α-hydrogen (pKa > 14). At 1 × 10−4 M [OH−], no detectable transformation of 8
2 FTS, which is expected to possess a significantly less acidified α-hydrogen (pKa > 14). At 1 × 10−4 M [OH−], no detectable transformation of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS was observed. This result supports the requirement for sufficient α-hydrogen acidity to enable the initial deprotonation step, thus the transformation is controlled by acid–base thermodynamics rather than simply by hydroxide presence.
2 FTS was observed. This result supports the requirement for sufficient α-hydrogen acidity to enable the initial deprotonation step, thus the transformation is controlled by acid–base thermodynamics rather than simply by hydroxide presence.
Additional kinetics evidence further supports this interpretation. The reaction rate of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA was greater than 6
2 FTCA was greater than 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA. Increasing chain length amplifies the cumulative inductive electron-withdrawing effect of –CF2– units, which further acidifies the α-hydrogen adjacent to the carboxylate group and stabilizes the resulting carbanion intermediate. Both effects favor the pre-equilibrium deprotonation step required for an E1cb elimination mechanism and lower the energetic barrier for the subsequent C–F bond cleavage. The larger reaction rate constant of 8
2 FTCA. Increasing chain length amplifies the cumulative inductive electron-withdrawing effect of –CF2– units, which further acidifies the α-hydrogen adjacent to the carboxylate group and stabilizes the resulting carbanion intermediate. Both effects favor the pre-equilibrium deprotonation step required for an E1cb elimination mechanism and lower the energetic barrier for the subsequent C–F bond cleavage. The larger reaction rate constant of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA demonstrates that these inductive effects are kinetically relevant even under mild alkaline conditions. When NaOH was replaced with NH3 (a system in which the majority of base exists as NH3 with only small amounts of OH− at equilibrium), the observed rate constant decreased sixfold (from 0.09 to 0.015 h−1), matching the sixfold reduction in [OH−] between the two experiments. This 1
2 FTCA demonstrates that these inductive effects are kinetically relevant even under mild alkaline conditions. When NaOH was replaced with NH3 (a system in which the majority of base exists as NH3 with only small amounts of OH− at equilibrium), the observed rate constant decreased sixfold (from 0.09 to 0.015 h−1), matching the sixfold reduction in [OH−] between the two experiments. This 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 proportionality between kobs and [OH−] confirms that hydroxide concentration directly controls the transformation rate. Such linear dependence on [OH−] is characteristic of specific-base behavior, consistent with a mechanism in which hydroxide participates only in the deprotonation pre-equilibrium.
1 proportionality between kobs and [OH−] confirms that hydroxide concentration directly controls the transformation rate. Such linear dependence on [OH−] is characteristic of specific-base behavior, consistent with a mechanism in which hydroxide participates only in the deprotonation pre-equilibrium.
Together, these findings indicate that n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA transforms via a reversible E1cb mechanism. The two main challenges to this mechanism are the low intrinsic acidity of the α-hydrogen and the potential instability of the resulting carbanion. The first issue is mitigated by the strong electron-withdrawing effect of the fluorinated carbon chain, which substantially lowers the α-hydrogen pKa and allows deprotonation under the mildly basic conditions tested. The second issue concerns the stability of the carbanion intermediate, which might be compromised by the proximity of the negatively charged carboxylate head group. Although the pKa n
2 FTCA transforms via a reversible E1cb mechanism. The two main challenges to this mechanism are the low intrinsic acidity of the α-hydrogen and the potential instability of the resulting carbanion. The first issue is mitigated by the strong electron-withdrawing effect of the fluorinated carbon chain, which substantially lowers the α-hydrogen pKa and allows deprotonation under the mildly basic conditions tested. The second issue concerns the stability of the carbanion intermediate, which might be compromised by the proximity of the negatively charged carboxylate head group. Although the pKa n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA is not known experimentally or theoretically, the basic conditions used in this study ensure that the head group is likely ionized. Despite this, two effects can stabilize the carbanion: (1) inductive charge delocalization along the fluorinated chain, which is the dominant stabilizing effect given the strong electron-withdrawing character of the perfluoroalkyl chain; and (2) hydrogen-bonding interactions with surrounding solvent molecules, which can partially solvate the carbanion and reduce its effective charge density. While resonance stabilization by the carboxylate group would be significant in the protonated form, under the alkaline conditions of this study the carboxylate is likely ionized, and direct resonance delocalization onto an adjacent carbanion would be electrostatically disfavored. Nonetheless, the cumulative inductive stabilization provided by the fluorinated chain is considered sufficient to support the carbanion intermediate required for E1cb elimination. This solvent effect may explain why the reaction proceeds in pure methanol but not in water at [OH−] < 10−5 M. Altogether, these considerations suggest that the carbanion intermediate is sufficiently stabilized for the reaction to proceed via an E1cb mechanism.
2 FTCA is not known experimentally or theoretically, the basic conditions used in this study ensure that the head group is likely ionized. Despite this, two effects can stabilize the carbanion: (1) inductive charge delocalization along the fluorinated chain, which is the dominant stabilizing effect given the strong electron-withdrawing character of the perfluoroalkyl chain; and (2) hydrogen-bonding interactions with surrounding solvent molecules, which can partially solvate the carbanion and reduce its effective charge density. While resonance stabilization by the carboxylate group would be significant in the protonated form, under the alkaline conditions of this study the carboxylate is likely ionized, and direct resonance delocalization onto an adjacent carbanion would be electrostatically disfavored. Nonetheless, the cumulative inductive stabilization provided by the fluorinated chain is considered sufficient to support the carbanion intermediate required for E1cb elimination. This solvent effect may explain why the reaction proceeds in pure methanol but not in water at [OH−] < 10−5 M. Altogether, these considerations suggest that the carbanion intermediate is sufficiently stabilized for the reaction to proceed via an E1cb mechanism.
We therefore propose the following reaction mechanism and kinetic sequence. Hydroxide participates in a pre-equilibrium deprotonation step (eqn (1)), which is the defining feature of the E1cb mechanism. In this step, deprotonation of the α-hydrogen forms a carbanion intermediate (FTCA2−) stabilized by resonance and inductive effects. Increasing [OH−] shifts this equilibrium toward the carbanion intermediate, leading to higher FTUCA yields. Because the carbanion intermediate has a finite lifetime, the system can undergo conformational rotation to achieve a favorable orbital overlap between the carbanion p-orbital and the σ* orbital of the β-C–F bond prior to elimination. This conformational flexibility, which distinguishes E1cb from the strict geometric requirements of a concerted E2 pathway, facilitates electron transfer to form the α–β π-bond and release fluoride (F−). This subsequent fluorine-elimination step is rate-determining (eqn (2)). The equilibrium expression (eqn (3)) is substituted into the rate law (eqn (4) and (5)) to obtain an observed first-order rate constant (eqn (6)) that is proportional to [OH−]. At sufficiently high [OH−], FTCA becomes fully deprotonated and the rate becomes independent of base concentration (eqn (7)). This kinetic transition from base-dependent to base-independent behavior confirms that the rate-determining step is the unimolecular C–F bond cleavage, consistent with a reversible E1cb mechanism (Fig. 5). The mechanistic sequence is as follows:
Step 1: pre-equilibrium deprotonation
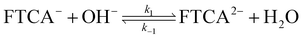 | (1) |
Step 2: rate-determining fluoride elimination
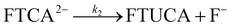 | (2) |
Equilibrium relationship
 | (3) |
Substitution into rate expression
| [FTCA2−] = Keq[FTCA−][OH−] | (4) |
| r = k2[FTCA2−] = k2Keq[OH−][FTCA−] | (5) |
Observed first-order rate law
Because [OH−] ≫ [FTCA], the observed first-order rate constant can be defined as
| kobs = k2Keq[OH−] ⇒ r = kobs[FTCA−] | (6) |
High-base limit
At sufficiently high [OH−], complete deprotonation of FTCA occurs, and the rate becomes independent of [OH−]:
| [FTCA2−] ≈ [FTCA0−] ⇒ r ≈ k2[FTCA0−] | (7) |
4. Conclusions
This study provides the first experimental evidence of abiotic transformation of FTCAs under basic conditions, establishing a reversible E1cb elimination mechanism. Both 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 8
2 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA underwent hydroxide-promoted conversion to their corresponding unsaturated products (FTUCAs) at ambient temperature, with reaction onset between 1 × 10−4 and 2 × 10−4 M [OH−]. Kinetic results confirmed first-order transformation with rates proportional to [OH−], consistent with a specific-base catalysis reaction. Minor yields of perfluorocarboxylic acids were detected only at relatively high base concentrations (≥0.1 M).
2 FTCA underwent hydroxide-promoted conversion to their corresponding unsaturated products (FTUCAs) at ambient temperature, with reaction onset between 1 × 10−4 and 2 × 10−4 M [OH−]. Kinetic results confirmed first-order transformation with rates proportional to [OH−], consistent with a specific-base catalysis reaction. Minor yields of perfluorocarboxylic acids were detected only at relatively high base concentrations (≥0.1 M).
Experiments using NH3 and a base-containing consumer cleaning product demonstrated that these transformations can also occur under conditions relevant to laboratory and household environments. Collectively, these findings reveal that mild alkaline conditions are sufficient to initiate FTCA transformation and that such reactions may proceed inadvertently during PFAS extractions or product use. The identification of an E1cb elimination mechanism provides a new foundation for understanding abiotic precursor chemistry and underscores the need to consider hydroxide-promoted reactions when assessing precursors fate, transformation, and analytical recovery.
Conflicts of interest
The authors have no conflicts of interest to declare. This research was conducted independently, with no financial or personal relationships that could have influenced the results or interpretation of the findings.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5em00821b.Acknowledgements
We express our gratitude to the Sloan Foundation for generously funding this project and the Air & Waste Management Association (A&WMA) for their scholarship awards on solid and hazardous waste research. We thank Dr Paul Westerhoff and Zunhui Lin at Arizona State University (ASU) for use of their CIC and assistance with sample analysis. Special appreciation goes to the University of Notre Dame's Mass Spectrometry and Proteomics Facility (MSPF), with thanks to Bill Boggess, Heather Whitehead, and Alyssa Wicks for their expertise related to LC-MS/MS. We also acknowledge the Center for Environmental Science and Technology (CEST), particularly John Loftus and Mike Brueseke, for their support throughout this study. Lastly, we thank Prof. Dan McCurry (EPFL) for fruitful discussions on elimination reactions.References
- M. P. S. Andersen, O. J. Nielsen, M. D. Hurley, J. C. Ball, T. J. Wallington, D. A. Ellis, J. W. Martin and S. A. Mabury, Atmospheric Chemistry of 4:2 Fluorotelomer Alcohol (n-C4F9CH2CH2OH): Products and Mechanism of Cl Atom Initiated Oxidation in the Presence of NOx, J. Phys. Chem. A, 2005, 109, 1849–1856, DOI:10.1021/jp810585c.
- B. F. Scott, C. Spencer, S. A. Mabury and D. C. G. Muir, Poly and Perfluorinated Carboxylates in North American Precipitation, Environ. Sci. Technol., 2006, 40(23), 7167–7174, DOI:10.1021/es061403n.
- D. A. Ellis, J. W. Martin, A. O. De Silva, S. A. Mabury, M. D. Hurley, M. P. Sulbaek Andersen and T. J. Wallington, Degradation of Fluorotelomer Alcohols: A Likely Atmospheric Source of Perfluorinated Carboxylic Acids, Environ. Sci. Technol., 2004, 38(12), 3316–3321, DOI:10.1021/es049860w.
- C. Ma, H. Peng, H. Chen, W. Shang, X. Zheng, M. Yang and Y. Zhang, Long-Term Trends of Fluorotelomer Alcohols in a Wastewater Treatment Plant Impacted by Textile Manufacturing Industry, Chemosphere, 2022, 299, 134442, DOI:10.1016/j.chemosphere.2022.134442.
- C. M. Butt, D. C. G. Muir and S. A. Mabury, Elucidating the Pathways of Poly- and Perfluorinated Acid Formation in Rainbow Trout, Environ. Sci. Technol., 2010, 44(13), 4973–4980, DOI:10.1021/es100702a.
- J. Zhao, L. Yang, X. Yang, X. Zhao, M. Li, S. Zhao, L. Zhu and J. Zhan, Degradation of 8:2 Fluorotelomer Carboxylic Acid (8:2 FTCA) by Plants and Their Co-Existing Microorganisms, J. Hazard. Mater., 2023, 451, 131129, DOI:10.1016/j.jhazmat.2023.131129.
- J. D. Boettger, N. M. DeLuca, M. A. Zurek-Ost, K. E. Miller, C. Fuller, K. D. Bradham, P. Ashley, W. Friedman, E. A. Pinzer, D. C. Cox, G. Dewalt, K. K. Isaacs, E. A. Cohen Hubal and J. P. McCord, Emerging Per- and Polyfluoroalkyl Substances in Tap Water from the American Healthy Homes Survey II, Environ. Sci. Technol., 2025, 59(5), 2686–2698, DOI:10.1021/acs.est.4c08037.
- M. M. Phillips, M. J. A. Dinglasan-Panlilio, S. A. Mabury, K. R. Solomon and P. K. Sibley, Fluorotelomer Acids Are More Toxic than Perfluorinated Acids, Environ. Sci. Technol., 2007, 41(20), 7159–7163, DOI:10.1021/es070734c.
- D. F. Hagen, J. Belisle, J. D. Johnson and P. Venkateswarlu, Characterization of Fluorinated Metabolites by a Gas Chromatographic-Helium Microwave Plasma Detector-The Biotransformation of 1H,1H,2H,2H-Perfluorodecanol to Perfluorooctanoate, Anal. Biochem., 1981, 118(2), 336–343, DOI:10.1016/0003-2697(81)90591-1.
- H. Nilsson, A. Kärrman, A. Rotander, B. Van Bavel, G. Lindström and H. Westberg, Professional Ski Waxers' Exposure to PFAS and Aerosol Concentrations in Gas Phase and Different Particle Size Fractions, Environ. Sci.: Process. Impacts, 2013, 15(4), 814–822, 10.1039/c3em30739e.
- H. Nilsson, A. Kärrman, A. Rotander, B. van Bavel, G. Lindström and H. Westberg, Biotransformation of Fluorotelomer Compound to Perfluorocarboxylates in Humans, Environ. Int., 2013, 51(2013), 8–12, DOI:10.1016/j.envint.2012.09.001.
- J. C. D'Eon and S. A. Mabury, Production of Perfluorinated Carboxylic Acids (PFCAs) from the Biotransformation of Polyfluoroalkyl Phosphate Surfactants (PAPS): Exploring Routes of Human Contamination, Environ. Sci. Technol., 2007, 41(13), 4799–4805, DOI:10.1021/es070126x.
- C. M. Butt, D. C. G. Muir and S. A. Mabury, Biotransformation pathways of fluorotelomer-based polyfluoroalkyl substances: a review, Environ. Toxicol. Chem., 2014, 33(2), 243–267, DOI:10.1002/etc.2407.
- J. Han, W. Gu, H. Barrett, D. Yang, S. Tang, J. Sun, J. Liu, H. M. Krause, K. A. Houck and H. A. Peng, Roadmap to the Structure-Related Metabolism Pathways of per- and Polyfluoroalkyl Substances in the Early Life Stages of Zebrafish (Danio Rerio), Environ. Health Perspect., 2021, 129(7), 1–15, DOI:10.1289/EHP7169.
- C. M. Butt, D. C. G. Muir and S. A. Mabury, Biotransformation of the 8:2 Fluorotelomer Acrylate in Rainbow Trout. 1. In Vivo Dietary Exposure, Environ. Toxicol. Chem., 2010, 29(12), 2726–2735, DOI:10.1002/etc.349.
- N. Merino, M. Wang, R. Ambrocio, K. Mak, E. O'Connor, A. Gao, E. L. Hawley, R. A. Deeb, L. Y. Tseng and S. Mahendra, Fungal Biotransformation of 6:2 Fluorotelomer Alcohol, Remediat. J. Environ. Cleanup Costs Technol. Tech., 2018, 28(2), 59–70, DOI:10.1002/rem.21550.
- H. Hamid, L. Y. Li and J. R. Grace, Review of the Fate and Transformation of Per- and Polyfluoroalkyl Substances (PFASs) in Landfills, Environ. Pollut., 2018, 74–84, DOI:10.1016/j.envpol.2017.12.030.
- T. L. Coggan, D. Moodie, A. Kolobaric, D. Szabo, J. Shimeta, N. D. Crosbie, E. Lee, M. Fernandes and B. O. Clarke, An Investigation into Per- and Polyfluoroalkyl Substances (PFAS) in Nineteen Australian Wastewater Treatment Plants (WWTPs), Heliyon, 2019, 5(8), e02316, DOI:10.1016/j.heliyon.2019.e02316.
- F. Xiao, T. R. Halbach, M. F. Simcik and J. S. Gulliver, Input Characterization of Perfluoroalkyl Substances in Wastewater Treatment Plants: Source Discrimination by Exploratory Data Analysis, Water Res., 2012, 46(9), 3101–3109, DOI:10.1016/j.watres.2012.03.027.
- W. Zhang, Y. Zhang, S. Taniyasu, L. W. Y. Yeung, P. K. S. Lam, J. Wang, X. Li, N. Yamashita and J. Dai, Distribution and Fate of Perfluoroalkyl Substances in Municipal Wastewater Treatment Plants in Economically Developed Areas of China, Environ. Pollut., 2013, 176, 10–17, DOI:10.1016/j.envpol.2012.12.019.
- H. Chen, H. Peng, M. Yang, J. Hu and Y. Zhang, Detection, Occurrence, and Fate of Fluorotelomer Alcohols in Municipal Wastewater Treatment Plants, Environ. Sci. Technol., 2017, 51(16), 8953–8961, DOI:10.1021/acs.est.7b00315.
- A. Arakaki, Y. Ishii, T. Tokuhisa, S. Murata, K. Sato, T. Sonoi, H. Tatsu and T. Matsunaga, Microbial Biodegradation of a Novel Fluorotelomer Alcohol, 1H,1H,2H,2H,8H,8H-Perfluorododecanol, Yields Short Fluorinated Acids, Appl. Microbiol. Biotechnol., 2010, 88(5), 1193–1203, DOI:10.1007/s00253-010-2815-9.
- Y. Jeong, K. M. Da Silva, E. Iturrospe, Y. Fuiji, T. Boogaerts, A. L. N. van Nuijs, J. Koelmel and A. Covaci, Occurrence and Contamination Profile of Legacy and Emerging Per- and Polyfluoroalkyl Substances (PFAS) in Belgian Wastewater Using Target, Suspect and Non-Target Screening Approaches, J. Hazard. Mater., 2022, 437, 129378, DOI:10.1016/j.jhazmat.2022.129378.
- T. Zhou, X. Li, H. Liu, S. Dong, Z. Zhang, Z. Wang, J. Li, L. D. Nghiem, S. J. Khan and Q. Wang, Occurrence, Fate, and Remediation for per-and Polyfluoroalkyl Substances (PFAS) in Sewage Sludge: A Comprehensive Review, J. Hazard. Mater., 2024, 466, 133637, DOI:10.1016/j.jhazmat.2024.133637.
- J. Liu, L. S. Lee, L. F. Nies, C. H. Nakatsu and R. F. Turco, Biotransformation of 8:2 Fluorotelomer Alcohol in Soil and by Soil Bacteria Isolates, Environ. Sci. Technol., 2007, 41(23), 8024–8030, DOI:10.1021/es0708722.
- A. L. Myers and S. A. Mabury, Fate of Fluorotelomer Acids in a Soil–Water Microcosm, Environ. Toxicol. Chem., 2010, 29(8), 1689–1695, DOI:10.1002/ETC.211.
- J. Liu and S. Mejia Avendaño, Microbial Degradation of Polyfluoroalkyl Chemicals in the Environment: A Review, Environ. Int., 2013, 61, 98–114, DOI:10.1016/j.envint.2013.08.022.
- M. J. A. Dinglasan, Y. Ye, E. A. Edwards and S. A. Mabury, Fluorotelomer Alcohol Biodegradation Yields Poly- and Perfluorinated Acids, Environ. Sci. Technol., 2004, 38(10), 2857–2864, DOI:10.1021/es0350177.
- M. Loewen, T. Halldorson, F. Wang and G. Tomy, Fluorotelomer Carboxylic Acids and PFOS in Rainwater from an Urban Center in Canada, Environ. Sci. Technol., 2005, 39(9), 2944–2951, DOI:10.1021/es048635b.
- T. Anumol, S. Dagnino, D. R. Vandervort and S. A. Snyder, Transformation of Polyfluorinated Compounds in Natural Waters by Advanced Oxidation Processes, Chemosphere, 2016, 144, 1780–1787, DOI:10.1016/j.chemosphere.2015.10.070.
- P. M. Dombrowski, P. Kakarla, W. Caldicott, Y. Chin, V. Sadeghi, D. Bogdan, F. Barajas-Rodriguez and S. Y. D. Chiang, Technology Review and Evaluation of Different Chemical Oxidation Conditions on Treatability of PFAS, Remediation, 2018, 28(2), 135–150, DOI:10.1002/rem.21555.
- X. Fan, Y. Jiang, X. Guan, Y. Bao, M. Gu, M. Mumtaz, J. Huang and G. Yu, Determination of Total Reducible Organofluorine in PFAS-Impacted Aqueous Samples Based on Hydrated Electron Defluorination, Sci. Total Environ., 2022, 829, 154548, DOI:10.1016/j.scitotenv.2022.154548.
- R. Tenorio, J. Liu, X. Xiao, A. Maizel, C. P. Higgins, C. E. Schaefer and T. J. Strathmann, Destruction of Per-and Polyfluoroalkyl Substances (PFASs) in Aqueous Film-Forming Foam (AFFF) with UV-Sulfite Photoreductive Treatment, Environ. Sci. Technol., 2020, 54(11), 6957–6967, DOI:10.1021/acs.est.0c00961.
- H. F. Schröder and R. J. W. Meesters, Stability of Fluorinated Surfactants in Advanced Oxidation Processes - A Follow up of Degradation Products Using Flow Injection-Mass Spectrometry, Liquid Chromatography-Mass Spectrometry and Liquid Chromatography-Multiple Stage Mass Spectrometry, J. Chromatogr. A, 2005, 1082(1 spec. iss.), 110–119, DOI:10.1016/j.chroma.2005.02.070.
- M. H. Plumlee, K. Mcneill and M. Reinhard, Indirect Photolysis of Perfluorochemicals: Hydroxyl Radical-Initiated Oxidation of N-Ethyl Perfluorooctane Sulfonamido Acetate (N-EtFOSAA) and Other Perfluoroalkanesulfonamides, Environ. Sci. Technol., 2009, 43(10), 3662–3668, DOI:10.1021/es803411w.
- US EPA, Analysis of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous, Solid, Biosolids, and Tissue Samples by LC-MS/MS, 2021, pp. 1–65 Search PubMed.
- United States Environmental Protection Agency, 4th Draft Method 1633 - Analysis of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous, Solid, Biosolids, and Tissue Samples by LC-MS/MS, 2023 Search PubMed.
- M. Al Amin, Y. Luo, A. Nolan, F. Robinson, J. Niu, S. Warner, Y. Liu, R. Dharmarajan, M. Mallavarapu, R. Naidu and C. Fang, Total Oxidisable Precursor Assay towards Selective Detection of PFAS in AFFF, J. Cleaner Prod., 2021, 328, 129568, DOI:10.1016/j.jclepro.2021.129568.
- F. Geng and D. E. Helbling, Cascading Pathways Regulate the Biotransformations of Eight Fluorotelomer Acids Performed by Wastewater Microbial Communities, Environ. Sci. Technol., 2024, 58(52), 23201–23211, DOI:10.1021/acs.est.4c09534.
- J. W. Martin, S. A. Mabury and P. J. O'Brien, Metabolic Products and Pathways of Fluorotelomer Alcohols in Isolated Rat Hepatocytes, Chem.-Biol. Interact., 2005, 155(3), 165–180, DOI:10.1016/j.cbi.2005.06.007.
- J. Zhou, K. Baumann, N. Chang, G. Morrison, W. Bodnar, Z. Zhang, J. M. Atkin, J. D. Surratt and B. J. Turpin, Per- and Polyfluoroalkyl Substances (PFASs) in Airborne Particulate Matter (PM2.0) Emitted during Floor Waxing: A Pilot Study, Atmos. Environ., 2022, 268 DOI:10.1016/j.atmosenv.2021.118845.
| This journal is © The Royal Society of Chemistry 2026 |




