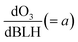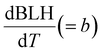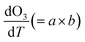Open Access Article
Open Access ArticleAccelerated O3 formation triggered by summer heatwaves in megacity Seoul
Junsu
Gil
 ab,
Meehye
Lee
*ac,
Moon-soo
Park
ab,
Meehye
Lee
*ac,
Moon-soo
Park
 d,
Cheol-Hee
Kim
d,
Cheol-Hee
Kim
 e,
Ja-ho
Koo
e,
Ja-ho
Koo
 f,
Hyojung
Lee
e,
Sunggu
Kang
f,
Hyojung
Lee
e,
Sunggu
Kang
 a and
Hojoon
Rhee
ag
a and
Hojoon
Rhee
ag
aDepartment of Earth and Environmental Sciences, Korea University, Seoul, South Korea
bInstitute of Basic Sciences, Korea University, Seoul, South Korea
cGraduate School of Energy and Environment, Korea University, Seoul, South Korea
dDepartment of Climate and Environment, Sejong University, Seoul, South Korea
eDepartment of Atmospheric Sciences, Pusan National University, Pusan, South Korea
fDepartment of Atmospheric Sciences, Yonsei University, Seoul, South Korea
gSeoul Metropolitan Government Research Institute of Public Health and Environment, Seoul, South Korea
First published on 6th February 2026
Abstract
As compelling evidence of global warming, heatwaves are expected to elevate O3 mixing ratios in highly polluted urban areas. In summer 2018, Seoul, an Asian megacity, experienced elevated O3 levels in conjunction with a temperature surge (maximum of 38.9 °C). This study quantitatively estimates the O3-climate penalty through measurements of nitrogen oxide species, volatile organic compounds (VOCs), and the boundary-layer height, as well as model simulations. The results highlight an acceleration of O3 concentration increment with increased temperature and elevated ozone production efficiency in NOx-saturated conditions. Furthermore, it emphasizes the importance of dynamic boundary-layer processes and increased VOC concentrations resulting from fugitive emissions during the heatwave.
Environmental significanceWith global warming, surface ozone levels are expected to increase with more frequent heatwaves, but the detailed process of interaction between ozone and heatwaves is still unclear. In summer 2018, we observed an ozone enhancement in conjunction with an extreme heatwave, quantified its temperature dependency, and elucidated its driving processes. We found that reinforced vertical mixing of air mass with the additional fugitive emission of volatile organic compounds in the reactive nitrogen oxide saturated megacity plays a key role in boosting ozone production efficiency increase, and it accelerates the daytime ozone enhancement. This implies that more frequent heatwaves due to climate change could induce faster and greater ozone increases in megacities than expected. |
1. Introduction
Numerous spatiotemporal observations and modeling studies indicate an ongoing and projected continuous increase in global surface temperature.1 Under the worst-case scenario of the Shared Socioeconomic Pathways (SSPs), the rise in climate change pollutants could cause a significant increase in global temperature. For example, climate simulation models project a rise in average temperature up to 7.0 °C in Northeast Asia by 2100.2–4 This temperature surge is anticipated to elevate O3 mixing ratios, particularly in urban areas with high emissions of NOx and volatile organic compounds (VOCs) under extreme meteorological conditions. Notably, O3 concentrations spiked during the 2018 heatwaves in European and Asian cities.5–10 This drew attention to O3 as both a short-lived climate pollutant (SLCP) contributing to global warming11–13 and a key air pollutant deteriorating air quality.14To recognize the positive feedback between surface O3 and global warming, a comprehensive understanding of the interplay between chemical ozone production mechanisms and climate variables is imperative. In recent years, increases in surface O3 due to climate change have been labeled the “O3-climate penalty”, underscoring this gas' adverse impacts on the climate system and the associated costs of climate change mitigation and damage repair measures.15,16 Recent studies have suggested methods for quantifying the O3-climate penalty as an “O3-climate penalty factor” (CPFO3) using statistical fitting17–19 or discretization methods combined with model simulations.20–22 Despite the various factors that influence O3 increase across different regions, the CPFO3 calculated from long-term measurement data is quite similar in most areas, particularly at background sites (0.1–3.2 ppbv °C−1).16,21,23
As global warming intensifies, megacities around the world are experiencing increased O3 levels during heatwaves.24 These extreme events are clear indicators of global warming and highlight the urgent need for research to explore the mechanisms and key factors contributing to elevated O3 levels during heatwaves. In summer, the chemical reactivity linked to precursor emissions is significantly increased. Therefore, in this season, the chemical processes driving O3 formation tend to outweigh the physical processes associated with its long-range transport, which can lead to a pronounced increase in CPFO3 in urban areas.25
Various studies have shown a correlation between O3 formation and temperature in polluted regions during short-term heatwaves,26–28 suggesting that O3 formation reinforced over time. Given the significant impact of O3 on human health, its accelerated formation due to temperature rise can lead to dangerously high levels, posing serious risks to residents of megacities.
Seoul, a megacity in South Korea, experienced an extreme heatwave in summer 2018, which saw an unprecedented daily maximum temperature exceeding 38 °C accompanied by a significant increase in O3. Since 2000, Seoul has witnessed a continuous annual increase in the 99th percentile temperature of 0.09 °C per year and in the annual average O3 concentration of 1.28 ppbv per year (Fig. 1). These variations of 99%ile O3 concentration and temperature value represent the changes in summer season, when the O3 level is strongly determined by the photochemical reaction. The 2018 heatwave caused the rate of increase in the annual average temperature and O3 to rise to 0.15 °C per year and 1.73 ppbv per year, respectively,29 approximately 67% and 54% larger than before the heatwave. The exceptionally high temperatures recorded during the 2018 heatwave exemplify the effects of climate change30–32 and resulted in surface O3 concentrations exceeding South Korean air quality standards.33 These observations suggest that, in the future, the rise in extreme temperatures due to global warming may cause critical O3-related challenges in megacities, similar to those observed during the 2018 heatwaves.
This study aimed to estimate the CPFO3 during a short heatwave in a highly polluted urban area in Seoul and explore the crucial factors determining this parameter using observational data and model results obtained in the summer of 2018. The findings provide vital information for policies to abate O3 pollution and for addressing emerging challenges in megacities during heatwaves, potentially intensified by future warming.
2. Materials and methods
2.1. Field measurements
All measurements were conducted between 13 July and 20 August 2018 in urban Seoul, South Korea. Trace gases and aerosol composition were measured at the Korea University campus (KU, 37.59° N, 127.03° E, 27 m a.g.l.). Additionally, 55 VOC species and vertical winds were measured in the districts of Jongro-gu (37.57° N, 127.01° E) and Jungnang-gu (37.59° N, 127.08° E, 22 m a.g.l.), located near KU (Fig. S1).O3, NO, and NO2 were measured using commercial instruments—respectively, a 49i, Thermo Scientific; a 42i, Thermo Scientific; and a T500U, Teledyne API. The detection limits of these instruments were 1, 0.4, and 0.04 ppbv, respectively.
The mixing ratios of 55 VOCs were measured in real-time using an automatic gas chromatography-flame ionization detector (GC-FID; Clarus 500, PerkinElmer) with a Thermal Desorption (TD; TurboMatrix 300, PerkinElmer) unit. Ambient air samples (600 mL) were collected using a small pump for 40 min at a flow rate of 15 mL min−1. The pump was controlled by a mass flow controller. A Nafion dryer was used to remove water from each sample, and the VOCs in the moisture-depleted sample were concentrated at −30 °C in a cold trap using a thermal desorption unit. Then, the temperature of the trap was rapidly increased to >300 °C to induce desorption, and the desorbed sample was transferred to the high-resolution capillary column of the GC-FID through a transfer line with high-purity nitrogen carrier gas. Each desorbed sample was separated through two columns, one polar (plot column) and one non-polar (bp-1 column), before the VOCs were detected by the FID.
Peroxyacetyl nitrate (PAN, CH3COO(O)NO2) was measured using gas chromatography-luminol chemiluminescence detection (GC-LCD).34,35 A 10 m capillary column (DB-1, Agilent, USA) was used and a photomultiplier tube (HC135-01, Hamamatsu, USA) was connected to the reaction cell where the luminol solution and PAN were mixed. The height of the PAN peak was quantified using a Molybdenum-NOx instrument (T500U, Thermo Scientific). The PAN standard was formed by reacting with vaporized liquid acetone with standard NO2 gas under UV light in a chamber. Data was ensembled into one-hour data every two minutes.
The HONO concentration was measured using a parallel-plate diffusion scrubber coupled with an ion chromatography (PPDS-IC) system. Ambient air was sampled slowly (0.8 L min−1) through a 1 m long 1/4″ PTFE tube and was solubilized in the collected solution (deionized water, HPLC grade, J. T. Baker) by penetrating a membrane filter (0.2 µm pore size, Osmoncis) in the PPDS. The collected solution was injected into the analysis column (AS14, Dionex IonPac™) with an eluent solution (3.5 mM Na2CO3, NaHCO3 mixed solution). The time resolution of the HONO and HNO3 measurements was 15 min, and hourly average data were used for the analysis. The detection limit was 20 pptv. Details of the measurement and calibration methods are provided in previous paper.36
A ceilometer (CL51, Vaisala) was used to obtain vertical profiles of backscattering coefficients at intervals of 10 m to 15.4 km using a laser (InGaAs diode laser) with a wavelength of 910 nm.37 A wind lidar (Windcube-200, Leosphere) was used to measure the vertical profile of wind speed and direction using a Doppler beam swinging scanner with a 1532 nm wavelength laser.38 A microwave radiometer (MWR) was used to observe the atmospheric attenuation and brightness temperature based on electromagnetic radiation emitted from the atmosphere using 14 channels (seven channels between 22 and 31 GHz for water vapor, seven channels between 51 and 58 GHz for temperature).39 This instrument measures two types of temperature profiles: zenith scanning for the entire troposphere (0–10 km) and elevation scanning for the boundary layer (0–2 km). The atmospheric boundary-layer height was estimated with the Integrated System for Atmospheric Boundary Layer height Estimation (ISABLE) algorithm using the vertical profile of the backscatter coefficient obtained from a ceilometer and the vertical temperature profile obtained by a MWR.40
2.2. Model simulations
This study employed an open-source box model (For 0-D Atmospheric Modelling, F0AM) to conduct a sensitivity test of O3 precursors in response to temperature changes using measurement data. The F0AM, developed by Wolfe et al. (2016), is suitable for modeling changes in atmospheric pollutants under biogenic influence.41 By applying the Master Chemical Mechanism (MCMv3.3.1), the F0AM model effectively estimates the variation in O3 levels in response to changes in temperature and concentrations of precursor gases such as NOx and VOCs.To facilitate the calculations, the OH reactivity of all VOCs (Table S1) was assessed and the top 10 VOCs were selected (Table S2), which accounted for 70% of the total OH reactivity. In the model simulations, the average diurnal O3 variation profile was calculated using the measurement data, and the modeled value was optimized using a daily maximum O3 concentration as the criterion. Since m,p-xylene could not be distinguished, it was treated as m-xylene in the simulation. Following the model optimization process, the bias of the results was under 1.1 ppbv, with the uncertainty attributed to m,p-xylene being estimated at approximately 0.4 ppbv.
For testing the changes in temperature, NOx, and VOCs to the daily maximum O3 concentration, we controlled each parameter following under conditions.
M T : other parameters in the model were fixed and only the temperature was changed. Two SSPs were used (SSP1 and SSP5), conditions in which the temperature increased every hour following the SSP results (SSP1: +2.6 °C and SSP5: +7.0 °C) of previous studies.2,3 The MA–MS scenario was simulated based on these temperature change condition.
M A: increasing the VOCs (excluding isoprene) from anthropogenic sources (“AVOCs”). Incremental ratio of AVOCs is set to 1.3% °C−1,42 considering the emission characteristics in this region that vehicle sources are dominant.43
M B: isoprene (C5H8) was selected as a representative VOC from biogenic sources (“BVOC”). Its increase ratio was calculated using the temperature response factor (Table S6), which is discussed elsewhere.44,45
M N: the temperature dependency of NOx is not well understood; therefore, NOx was considered to have originated from anthropogenic vehicle emissions, and the increase ratio was set to 1.3% °C−1 (i.e., the same as that used for AVOCs).27,46
M S: all variables (AVOCs, isoprene, and NOx) were changed.
Additionally, the Weather Research and Forecasting model coupled with Chemistry (WRF-Chem) v3.9.1 was used to simulate the variation in O3 and VOC concentrations during both heatwave and non-heatwave periods. The WRF-Chem (version 3.9.1) model47,48 was used to simulate O3 and other atmospheric pollutants, including PM2.5, SO2, and NO2, during both heatwave and non-heatwave periods. The model domain encompassed Northeast Asia (105–145° E, 25–50° N) and was configured with a grid spacing of 27 km and 28 vertical layers extending from the surface to 50 hPa. Meteorological initial and boundary conditions were sourced from the National Center for Environmental Prediction Final (NCEP/FNL) Operational Global Analysis data,49 which have a horizontal resolution of 1° × 1°. The Regional Atmospheric Chemistry Mechanism (RACM)50 was used for the gas-phase chemical scheme and the Modal Aerosol Dynamics Model for Europe/Volatility Basis Set (MADE/VBS)51 for the aerosol scheme. Biogenic emissions were derived from the Model of Emissions of Gases and Aerosols from Nature (MEGAN) version 2.04,45 while anthropogenic emissions were obtained from the KORUSv5 emission inventory, which was developed by a joint research group from Konkuk University and the National Institute of Environmental Research for the 2016 KORea-United States Air Quality (KORUS-AQ) field campaign.52 The physical and chemical model options used in this study are summarized in Table S3.
2.3. O3-climate penalty and CPFO3
Multiannual observation studies have found that O3 levels increase with rising temperature. While the sensitivity of O3 to temperature change is consistent in long-term observations, variability has been reported in short-term measurements.26,53,54 The relationship between O3 and temperature change is often expressed using linear regression or quadratic equations to capture changing dynamics. Based on the quadratic regression curve relating O3 and temperature ([O3]max = b0 + b1[T]max + b2[T]max2), the CPFO3 can be defined by the slope of the quadratic curve (2b2), and the O3–temperature dependency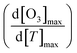 can be calculated using the following equation (eqn (1)):
can be calculated using the following equation (eqn (1)):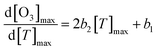 | (1) |
However, accurately estimating the O3 variation caused by increasing temperature is challenging due to multiple influencing factors, including physicochemical changes and anthropogenic–biogenic emissions related to temperature increase. The response of O3 to temperature can be assessed through (i) direct methods that examine the relationship between the observed O3 concentration and temperature, and (ii) indirect methods that analyze how temperature impacts various O3-influencing factors.55–57 Definitions of the O3-climate penalty vary across studies, with some using the slope of O3 concentration versus temperature16 and others deriving it from the results of chemical transport models related to increases in anthropogenic emissions.15 Rasmussen et al. (2013) proposed the following equation (eqn (2)) to express the indirect relationship between O3 concentration and temperature:
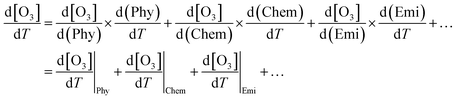 | (2) |
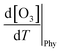 ,
, 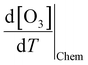 , and
, and 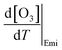 were estimated by considering the change in atmospheric chemical conditions and synoptic meteorology, allowing the sensitivity of O3 to temperature changes during heatwaves in a megacity to be determined.
were estimated by considering the change in atmospheric chemical conditions and synoptic meteorology, allowing the sensitivity of O3 to temperature changes during heatwaves in a megacity to be determined.
3. Results and discussions
3.1. Estimation of the O3-climate penalty factor
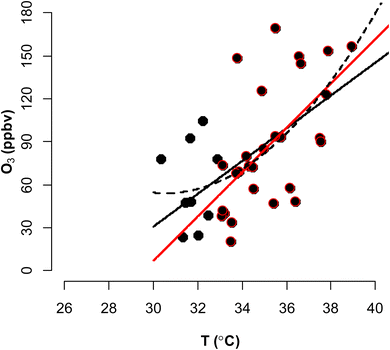
During the measurement period, there were 29 days of heatwaves according to the South Korean criteria, which defines a heatwave as a daily maximum temperature exceeding 33 °C. During this time, an extremely high temperature of 38.9 °C was recorded under blocking conditions over the Korean Peninsula (Fig. S2).30–32 On eight days during this high-temperature period, high-O3 episodes occurred which exceeded the national ambient air quality standard (hourly average O3 concentration of >100 ppbv). Throughout the heatwave period, the average O3 concentration was 36.6 ppbv, with a daily maximum of up to 169.4 ppbv. Additionally, the average and daily maximum O3 levels were higher than in the non-heatwave period by approximately 9 and 54 ppbv, respectively. Ozone levels consistently increased with rising temperatures throughout the measurement period, with the most significant increase observed during the heatwave period, as is clear from the daily maximum values (Fig. 2).The O3-climate penalty factor (CPFO3) was calculated based on the linear relationship (CPFO3_L) between the daily maximum O3 concentration and the daily maximum temperature. The value was 11.5 ppbv °C−1 for the entire measurement period and 15.5 ppbv °C−1 during the heatwave (Table 1). The inclusion of extreme heatwave cases resulted in a higher CPFO3_L for the entire measurement period compared to previous studies (∼3 ppbv °C−1).16,17,19,23 Further the maximum daily 8 h average (MDA8) O3 based CPFO3_L also shows high value (8.1 ppbv °C−1) during the whole periods, and it indicates that increase in O3 concentration during the heatwave period may be appeared with the increase of temperature.
| Period | b 0 | b 1 | b 2 | CPFO3 |
|---|---|---|---|---|
| Heatwave (linear) | −458.0 | 15.5 | 15.5 | |
| Total (linear) | −313.2 | 11.5 | 11.5 | |
| Total (quadratic) | 1371.1 | −86.2 | 1.4 | 2.8T − 86.2 |
The acceleration of the O3 increase at higher temperatures also can be attributed to the quadratic relationship between O3 concentration and temperature. By applying a quadratic regression curve to the relationship between the daily maximum O3 level and the daily maximum temperature, the coefficient of the quadratic function was calculated to represent the slope at each temperature point (eqn (1)). The derivative of CPFO3_Q provides the CPFO3_L as a function of temperature. Based on the measurement data, the following values were obtained: b0 = 1371.1; b1i = −86.2; b2 = 1.4. Thus, the O3–temperature dependency of the quadratic curve can be expressed by the following equation (eqn (3)):
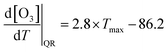 | (3) |
The calculated coefficient of CPFO3_Q (2.8) exceeded the upper boundary of the CPFO3_Q range observed in previous studies (0.1–1.3).26,27
The CPFO3_Q, obtained through quadratic regression, varied widely from 1 to 23 ppbv °C−1 above the observed inflection temperatures of over 30 °C (Tables S4 and S5). Interestingly, in previous studies in Seoul, temperatures exceeding 30 °C were identified as a prerequisite for elevated O3 concentrations exceeding national air quality standards.59,60 The CPFO3_L for the entire measurement period and the heatwave period aligned with the CPFO3_Q at temperatures of 35 and 36 °C, respectively. At the highest daily maximum temperature of 38.9 °C, the CPFO3 was 22.8 ppbv °C−1. These results highlight an accelerated O3 increase during a heatwave in a megacity, which is appropriately represented by different values of O3-temperature dependency at each temperature. This raises the following question: Why does the CPFO3 be different with temperature?
The increase in O3 resulting from a temperature increase can be elucidated through the discrete mechanisms governing O3 enhancement. The CPFO3 is attributed to variations in chemical reactions leading to O3 production, emissions linked to biological activities or evaporation, and physical processes such as advection, diffusion, and vertical mixing, as represented by 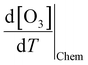 ,
, 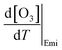 , and
, and 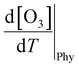 , respectively.20
, respectively.20
The chemical term 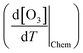 and emission term
and emission term 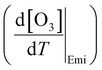 were initially computed together. Using the F0AM model, the change in O3 levels in response to temperature and emission variations was simulated based on the several cases (MT–MS) described in Section 2.2. Using the measurement data and F0AM, the CPFO3 attributable to chemistry and emissions was approximately 1.7–1.8 ppbv °C−1 (Fig. 3 and Table S7). When considering the uncertainties of temperature dependent emission factors from various VOCs emission sources that can be increased up to 6.7% °C−1, it makes the variation in simulated
were initially computed together. Using the F0AM model, the change in O3 levels in response to temperature and emission variations was simulated based on the several cases (MT–MS) described in Section 2.2. Using the measurement data and F0AM, the CPFO3 attributable to chemistry and emissions was approximately 1.7–1.8 ppbv °C−1 (Fig. 3 and Table S7). When considering the uncertainties of temperature dependent emission factors from various VOCs emission sources that can be increased up to 6.7% °C−1, it makes the variation in simulated 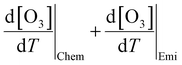 value as 0.2 ppbv °C−1 (Table S7).61 This upper bound of
value as 0.2 ppbv °C−1 (Table S7).61 This upper bound of 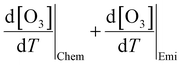 (∼2 ppbv °C−1) is near to the previous studies on the CPFO3_L (∼3 ppbv °C−1);21 however, it is lower than the CPFO3_L for the whole measurement period.
(∼2 ppbv °C−1) is near to the previous studies on the CPFO3_L (∼3 ppbv °C−1);21 however, it is lower than the CPFO3_L for the whole measurement period.
The physical term 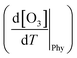 of eqn (2) primarily pertains to dynamic processes such as reinforced or decelerated long-range O3 transport caused by the temperature increase, and consequently influenced surface O3 levels. The transport of O3 and stagnant weather conditions facilitate the formation and accumulation of O3.62,63
of eqn (2) primarily pertains to dynamic processes such as reinforced or decelerated long-range O3 transport caused by the temperature increase, and consequently influenced surface O3 levels. The transport of O3 and stagnant weather conditions facilitate the formation and accumulation of O3.62,63
In this study, during the prolonged heatwave, blocking conditions played a crucial role in occurrence of heatwave over the Korean Peninsula. In Seoul, blocking conditions are commonly associated with synoptic atmospheric circulation, resulting in persistent anticyclonic or stagnant conditions.30 This scenario was observed during the KORea-US Air Quality (KORUS-AQ) campaign accompanied by a significant elevation of O3.64 Similar to this previous study, in the present study, surface O3 concentrations were significantly elevated under stagnant atmospheric conditions during heatwave periods in urban areas.
The boundary layer typically expands greatly under a persistent high-pressure system due to radiative heating. The daily evolution of this layer is thought to promote the photochemical production of O3 through vertical turbulent mixing with the low NOx concentration in the upper atmosphere above urban areas and also affects the surface O3.65–67 During the measurement period, the boundary layer height (BLH) expanded by 1500 m during the daytime, reaching its maximum between 14:00 and 17:00 local standard time (LST) before collapsing to nearly 500 m at nighttime, with its minimum occurring between 00:00 and 05:00 LST. Similarly, the maximum O3 was observed when BLH was maximum and the minimum was observed when BLH was minimum. While BLH generally increased with temperature, the ratio of the daily range of BLH to that of temperature (ΔBLH/ΔT) remained relatively consistent during both the heatwave and non-heatwave periods (Fig. S5). In comparison, the ratio of the daily range of O3 to that of BLH (ΔO3/ΔBLH) was greater during heatwave periods than non-heatwave periods. As a result, the rate of change of O3 with increasing temperature 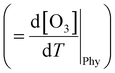 was calculated to be 21.8% higher during the heatwave (10.6 ppbv °C−1) than the non-heatwave period (8.7 ppbv °C−1) (Table 2). Notably, both values of
was calculated to be 21.8% higher during the heatwave (10.6 ppbv °C−1) than the non-heatwave period (8.7 ppbv °C−1) (Table 2). Notably, both values of 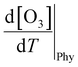 were significantly larger than those estimated for emissions and chemical processes (<2 ppbv °C−1). For the heatwave period, the discretization based on various measurement data resulted in a CPFO3
were significantly larger than those estimated for emissions and chemical processes (<2 ppbv °C−1). For the heatwave period, the discretization based on various measurement data resulted in a CPFO3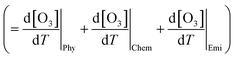 of 12.3–12.4 ppbv °C−1, which is similar to the CPFO3_L in heatwave periods of 15.5 ppbv °C−1. During the heatwave,
of 12.3–12.4 ppbv °C−1, which is similar to the CPFO3_L in heatwave periods of 15.5 ppbv °C−1. During the heatwave, 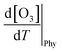 was about five times higher than
was about five times higher than 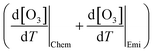 , indicating that physical processes were the primary drivers of the accelerated O3 enhancement during the heatwave.
, indicating that physical processes were the primary drivers of the accelerated O3 enhancement during the heatwave.
3.2. Vertical mixing as a physical driver of accelerated surface O3 enhancement
In NOx-saturated regions, model calculations demonstrate that surface O3 is titrated by NOx in the early morning and accumulates in the afternoon.68 Recent studies conducted in tall towers have demonstrated that free convection and vertical mixing facilitate efficient O3 formation, leading to elevated O3 and PAN concentrations at higher altitudes.69,70 This is consistent with our results that, in discretization methods, physical processes exert the most significant influence on CPFO3.The degree of atmospheric vertical mixing within the boundary layer can be expressed by the turbulent diffusion coefficient (kz),71 which is calculated using the following equations for z < L (eqn (4)):72
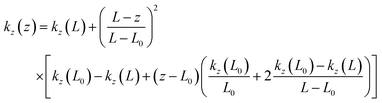 | (4) |
 | ||
| Fig. 4 Turbulent transfer coefficient (kz) based on the wind speed at the top of surface layer during (a) non-heatwave period and (b) heatwave period. | ||
Similar to the variation of kz, the diurnal variation of O3 simulated using the WRF-Chem model also showed large differences between the heatwave and non-heatwave periods (Fig. 5). The diurnal evolution of O3 was strongly correlated with changes in BLH, with O3 levels remaining low (below 10 ppbv) under a stable boundary layer until early morning and then rapidly increasing with the rise in BLH. The two cases exhibited sharply contrast in change of O3, that the morning upward and afternoon downward gradient in O3 concentration was more pronounced during the heatwave. These highlight the influence of physical terms in the CPFO3 equation, which were estimated from the measurements of reactive gases including O3, nitrogen oxide species, and VOCs, and BLH based on discretization methods.
 | ||
| Fig. 5 Diurnal-vertical variations in O3 concentrations during the (a) non-heatwave and (b) heatwave periods in Seoul simulated by WRF-Chem v3.9.1. | ||
3.3. Ozone production efficiency
In a megacity such as Seoul, NO2 levels typically reach their minimum in the afternoon, coinciding with the O3 peak.64 However, it was not observed in this study. Instead, NO2 concentration remained high in the afternoon, especially when the boundary layer was most expanded (Fig. 6(a) and (b)), indicating a rapid chemical cycling of nitrogen oxides. Based on the O3 production (=P(O3)) and loss of NOx (=L(NOx)) chemistry, the ozone production efficiency (OPE) was obtained using the level of Ox (=O3 + NO2) and NOz, where [NO2] + [NOz] ≪ [O3]73,74 (eqn (5)–(7)).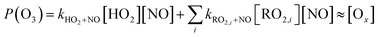 | (5) |
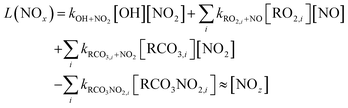 | (6) |
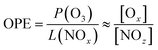 | (7) |
Using measurements of the nitrogen oxide species HONO, PAN, and HNO3, the OPE, represented by the ratio of Ox (O3 + NO2) to NOz (=PAN + HONO + HNO3), was compared between the heatwave and non-heatwave periods. The OPE during the heatwave was 18.9, while it was 16.4 during the non-heatwave period (Fig. 7), with both values being higher than the range of 3–8 reported in previous studies that heatwave had not been occurred.73,74 Hence, the OPE likely played a pivotal role in enhancing O3 levels in the summer heatwave periods.24,67
 | ||
| Fig. 7 The ozone production efficiency (OPE) as the ratio of Ox to NOz during the midday (10–17 LST) for heatwave (red) and non-heatwave (blue) periods. | ||
The formation of O3 was found to be sensitive to the concentrations of VOCs under NOx-saturated conditions. During the heatwave, anthropogenic VOCs (AVOC) and isoprene mixing ratios were larger than in the non-heatwave period, correlated with an increase in daytime temperature (Fig. 6(c) and (d)). Particularly, in the morning, AVOC concentrations were consistently elevated during the heatwave period compared to the non-heatwave period (2.7% °C−1), surpassing the typical temperature dependency of VOCs observed in previous studies (1.3% °C−1).42 This observation aligns with VOC patterns previously observed in Seoul, highlighting the fugitive emissions of AVOCs at elevated temperatures.43
During the heatwave, the mean isoprene concentration increased by approximately four times (10.3% °C−1), more than the increase in AVOCs. However, isoprene emissions are constrained at temperatures above the threshold of plant growth.75 On the other hand, AVOCs from fugitive emissions are likely to continue to increase with temperature. But these increase in VOCs only can contribute to the 4–5 ppbv °C−1 of CPFO3, and it is hard to fully explain the increase of CPFO3 during the heatwave period through only the increase in VOC concentration. Consequently, the heightened OPE, together with a change of boundary-layer related processes and increased VOC levels, was found to be a crucial physical factor in the significantly elevated O3 concentrations during the heatwave. The results show that prolonged heatwaves likely promote deep vertical mixing, thereby increasing O3 production by enhanced OPE due to increased fugitive emissions of VOCs. These findings underscore the importance of focusing on AVOCs to mitigate the accelerated O3 increase in urban areas during heatwaves.
4. Conclusions
This study revealed a significant O3-climate penalty in urban Seoul during short-term heatwave periods in summer 2018, surpassing the values reported in previous long-term observations, with the rate of change of the penalty (penalty factor) increasing with temperature. Measurements and model simulations underscored the significance of dynamic boundary-layer processes as a key pathway for O3 enhancement. However, the effect of the intricate interplay between dynamic processes and chemical reactions on change of O3-climate penalty factor remains incompletely understood.These findings suggest that the increasing frequency of heatwaves due to global warming could lead to rapid O3 increases in megacities, characterized by high levels of NOx and VOCs due to vehicle emissions. Efficient O3 production during heatwaves, facilitated by vertical mixing and fueled by increased fugitive emissions of VOCs, emphasizes the need to reduce AVOCs in megacity and its surrounding areas to mitigate the accelerating increase in O3 observed during heatwaves. These dynamic processes combined with the change of chemical change can lead to the non-linear increase of O3 climate penalty not only in the megacity, but further expanded to the metropolitan region especially during the warm season, which was reported as a critical problem in global ozone pollution.76 Therefore, this study establishes a framework for analyzing O3 enhancement mechanisms and estimating CPFO3 during future heatwaves, providing insights for effective pollutant reduction policies under climate change.
Author contributions
JG and ML conceptualize and design this research. JG, MP, SK, and HR contributed to the data collection. JG, CK, JK, and HL conducted the modelling and contributed to the analysis. All authors contributed to the interpretation of the results and manuscript writing.Conflicts of interest
The authors declare no competing interests.Data availability
Data for this article, including the observation data, are available at the repository (https://zenodo.org/records/11045153).77Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5em00629e.
Acknowledgements
This research was supported by National Research Foundation of Republic of Korea (2020R1A2C3014592) and Seoul Metropolitan Government (2019030A3C700). This study was also supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (RS-2025-00573406).References
- IPCC, in Climate Change 2023: Synthesis Report. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, 2023 Search PubMed.
- NIMS, Global Climate Change Prospect Report, National Institute of Meteorological Sciences, 2020 Search PubMed.
- B. C. O'Neill, C. Tebaldi, D. P. van Vuuren, V. Eyring, P. Friedlingstein, G. Hurtt, R. Knutti, E. Kriegler, J.-F. Lamarque and J. Lowe, The scenario model intercomparison project (ScenarioMIP) for CMIP6, Geosci. Model Dev., 2016, 9, 3461–3482 Search PubMed.
- B. C. O'Neill, E. Kriegler, K. Riahi, K. L. Ebi, S. Hallegatte, T. R. Carter, R. Mathur and D. P. van Vuuren, A new scenario framework for climate change research: the concept of shared socioeconomic pathways, Clim. Change, 2014, 122, 387–400 CrossRef.
- P. A. Dirmeyer, G. Balsamo, E. M. Blyth, R. Morrison and H. M. Cooper, Land-atmosphere interactions exacerbated the drought and heatwave over northern Europe during summer 2018, AGU Adv., 2021, 2, e2020AV000283 CrossRef.
- S. Undorf, K. Allen, J. Hagg, S. Li, F. Lott, M. J. Metzger, S. Sparrow and S. Tett, Learning from the 2018 heatwave in the context of climate change: Are high-temperature extremes important for adaptation in Scotland?, Environ. Res. Lett., 2020, 15, 034051 CrossRef.
- Y. Qian, H. Murakami, P.-c. Hsu and S. Kapnick, Effects of anthropogenic forcing and natural variability on the 2018 heatwave in Northeast Asia, Bull. Am. Meteorol. Soc., 2020, 101, S77–S82 Search PubMed.
- J. Zhang, Y. Gao, K. Luo, L. R. Leung, Y. Zhang, K. Wang and J. Fan, Impacts of compound extreme weather events on ozone in the present and future, Atmos. Chem. Phys., 2018, 18, 9861–9877 CrossRef CAS.
- A. Pyrgou, P. Hadjinicolaou and M. Santamouris, Enhanced near-surface ozone under heatwave conditions in a Mediterranean island, Sci. Rep., 2018, 8, 9191 CrossRef PubMed.
- X. Pu, T. Wang, X. Huang, D. Melas, P. Zanis, D. Papanastasiou and A. Poupkou, Enhanced surface ozone during the heat wave of 2013 in Yangtze River Delta region, China, Sci. Total Environ., 2017, 603, 807–816 CrossRef PubMed.
- D. T. Shindell, J.-F. Lamarque, M. Schulz, M. Flanner, C. Jiao, M. Chin, P. Young, Y. H. Lee, L. Rotstayn and N. Mahowald, Radiative forcing in the ACCMIP historical and future climate simulations, Atmos. Chem. Phys., 2013, 13, 2939–2974 CrossRef CAS.
- D. Stevenson, P. Young, V. Naik, J.-F. Lamarque, D. T. Shindell, A. Voulgarakis, R. B. Skeie, S. B. Dalsoren, G. Myhre and T. K. Berntsen, Tropospheric ozone changes, radiative forcing and attribution to emissions in the Atmospheric Chemistry and Climate Model Intercomparison Project (ACCMIP), Atmos. Chem. Phys., 2013, 13, 3063–3085 CrossRef.
- G. Myhre, W. Aas, R. Cherian, W. Collins, G. Faluvegi, M. Flanner, P. Forster, Ø. Hodnebrog, Z. Klimont and M. T. Lund, Multi-model simulations of aerosol and ozone radiative forcing due to anthropogenic emission changes during the period 1990–2015, Atmos. Chem. Phys., 2017, 17, 2709–2720 CrossRef CAS.
- P. S. Monks, A. Archibald, A. Colette, O. Cooper, M. Coyle, R. Derwent, D. Fowler, C. Granier, K. S. Law and G. Mills, Tropospheric ozone and its precursors from the urban to the global scale from air quality to short-lived climate forcer, Atmos. Chem. Phys., 2015, 15, 8889–8973 CrossRef CAS.
- S. Wu, L. J. Mickley, E. M. Leibensperger, D. J. Jacob, D. Rind and D. G. Streets, Effects of 2000–2050 global change on ozone air quality in the United States, J. Geophys. Res.: Atmos., 2008, 113, D06302 Search PubMed.
- B. J. Bloomer, J. W. Stehr, C. A. Piety, R. J. Salawitch and R. R. Dickerson, Observed relationships of ozone air pollution with temperature and emissions, Geophys. Res. Lett., 2009, 36, L09803 CrossRef.
- N. Otero, J. Sillmann, K. A. Mar, H. W. Rust, S. Solberg, C. Andersson, M. Engardt, R. Bergström, B. Bessagnet and A. Colette, A multi-model comparison of meteorological drivers of surface ozone over Europe, Atmos. Chem. Phys., 2018, 18, 12269–12288 CrossRef CAS.
- C. G. Nolte, T. L. Spero, J. H. Bowden, M. C. Sarofim, J. Martinich and M. S. Mallard, Regional temperature-ozone relationships across the US under multiple climate and emissions scenarios, J. Air Waste Manage. Assoc., 2021, 71, 1251–1264 CrossRef CAS PubMed.
- C. Gong, Y. Wang, H. Liao, P. Wang, J. Jin and Z. Han, Future Co-Occurrences of Hot Days and Ozone-Polluted Days Over China Under Scenarios of Shared Socioeconomic Pathways Predicted Through a Machine-Learning Approach, Earth's Future, 2022, 10, e2022EF002671 CrossRef CAS.
- D. Rasmussen, J. Hu, A. Mahmud and M. J. Kleeman, The ozone–climate penalty: past, present, and future, Environ. Sci. Technol., 2013, 47, 14258–14266 CrossRef CAS PubMed.
- S. Zhao, A. J. Pappin, S. Morteza Mesbah, J. Joyce Zhang, N. L. MacDonald and A. Hakami, Adjoint estimation of ozone climate penalties, Geophys. Res. Lett., 2013, 40, 5559–5563 CrossRef CAS.
- W. C. Porter and C. L. Heald, The mechanisms and meteorological drivers of the summertime ozone–temperature relationship, Atmos. Chem. Phys., 2019, 19, 13367–13381 CrossRef CAS.
- J. M. Johansson, Å. K. Watne, P. E. Karlsson, G. P. Karlsson, H. Danielsson, C. Andersson and H. Pleijel, The European heat wave of 2018 and its promotion of the ozone climate penalty in southwest Sweden, Boreal Environ. Res., 2020, 25, 1 Search PubMed.
- W. Zhao, G. Tang, H. Yu, Y. Yang, Y. Wang, L. Wang, J. An, W. Gao, B. Hu and M. Cheng, Evolution of boundary layer ozone in Shijiazhuang, a suburban site on the North China Plain, J. Environ. Sci., 2019, 83, 152–160 CrossRef CAS PubMed.
- S. E. Pusede, A. L. Steiner and R. C. Cohen, Temperature and recent trends in the chemistry of continental surface ozone, Chem. Rev., 2015, 115, 3898–3918 CrossRef CAS PubMed.
- P. Jing, Z. Lu and A. L. Steiner, The ozone-climate penalty in the Midwestern US, Atmos. Environ., 2017, 170, 130–142 CrossRef CAS.
- Y. Gu, K. Li, J. Xu, H. Liao and G. Zhou, Observed dependence of surface ozone on increasing temperature in Shanghai, China, Atmos. Environ., 2020, 221, 117108 CrossRef CAS.
- X. Zhou, M. Li, X. Huang, T. Liu, H. Zhang, X. Qi, Z. Wang, Y. Qin, G. Geng and J. Wang, Urban meteorology–chemistry coupling in compound heat–ozone extremes, Nat. Cities, 2025, 1–10 Search PubMed.
- Korea Meteorological Administration (KMA), 2018 Meteorological Yearbook, 2019, pp. 1–431 Search PubMed.
- K.-J. Ha, J.-H. Yeo, Y.-W. Seo, E.-S. Chung, J.-Y. Moon, X. Feng, Y.-W. Lee and C.-H. Ho, What caused the extraordinarily hot 2018 summer in Korea?, J. Meteorol. Soc. Jpn. Ser. II, 2020, 153–167 CrossRef.
- D. Yoon, D.-H. Cha, M.-I. Lee, K.-H. Min, J. Kim, S.-Y. Jun and Y. Choi, Recent changes in heatwave characteristics over Korea, Clim. Dyn., 2020, 55, 1685–1696 CrossRef.
- H.-D. Lee, K.-H. Min, J.-H. Bae and D.-H. Cha, Characteristics and comparison of 2016 and 2018 heat wave in Korea, Atmosphere, 2020, 30, 1–15 Search PubMed.
- Seoul Metropolitan Government Research Institute of Public Health and Environment (SMGRIPHE), 2018 Seoul Air Quality Assessment Report, 2019, pp. 1–102 Search PubMed.
- G. Lee, Y. Jang, H. Lee, J.-S. Han, K.-R. Kim and M. Lee, Characteristic behavior of peroxyacetyl nitrate (PAN) in Seoul megacity, Korea, Chemosphere, 2008, 73, 619–628 CrossRef CAS PubMed.
- J. Gaffney, R. Bornick, Y.-H. Chen and N. Marley, Capillary gas chromatographic analysis of nitrogen dioxide and PANs with luminol chemiluminescent detection, Atmos. Environ., 1998, 32, 1445–1454 CrossRef CAS.
- J. Gil, J. Son, S. Kang, J. Park, M. Lee, E. Jeon and M. Shim, HONO measurement in Seoul during Summer 2018 and its Impact on Photochemistry, J. Korean Soc. Atmos. Environ., 2020, 36, 579–588 Search PubMed.
- Vaisala, User's Guide, Vaisala Ceilometer CL51, Helsinki, Finland, 2010 Search PubMed.
- C. Werner, Doppler Wind Lidar, Lidar: Range-Resolved Optical Remote Sensing of the Atmosphere, 2005 Search PubMed.
- RPG, RPG: Instrument Operation and Software Guide, Operational Principles and Software Description for RPG Standard Single Polarization Radiometer (G4 Series), 2015, p. 170 Search PubMed.
- J.-S. Min, M.-S. Park, J.-H. Chae and M. Kang, Integrated System for Atmospheric Boundary Layer Height Estimation (ISABLE) using a ceilometer and microwave radiometer, Atmos. Meas. Tech., 2020, 13, 6965–6987 CrossRef.
- G. M. Wolfe, M. R. Marvin, S. J. Roberts, K. R. Travis and J. Liao, The framework for 0-D atmospheric modeling (F0AM) v3.1, Geosci. Model Dev., 2016, 9, 3309 CrossRef.
- J. I. Rubin, A. J. Kean, R. A. Harley, D. B. Millet and A. H. Goldstein, Temperature dependence of volatile organic compound evaporative emissions from motor vehicles, J. Geophys. Res.: Atmos., 2006, 111, D03305 CrossRef.
- S. Kang, J.-A. Kim, M. Lee, J. Park, E. Jeon, M. Shim and Y. Shin, An analysis of the temporal variability in volatile organic compounds (VOCs) within megacity Seoul and an identification of their sources, Atmos. Pollut. Res., 2022, 13, 101338 CrossRef CAS.
- L. K. Emmons, S. Walters, P. G. Hess, J.-F. Lamarque, G. G. Pfister, D. Fillmore, C. Granier, A. Guenther, D. Kinnison and T. Laepple, Description and evaluation of the Model for Ozone and Related chemical Tracers, version 4 (MOZART-4), Geosci. Model Dev., 2010, 3, 43–67 CrossRef.
- A. Guenther, T. Karl, P. Harley, C. Wiedinmyer, P. Palmer and C. Geron, Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature), Atmos. Chem. Phys., 2006, 6, 3181–3210 CrossRef CAS.
- A. L. Steiner, A. J. Davis, S. Sillman, R. C. Owen, A. M. Michalak and A. M. Fiore, Observed suppression of ozone formation at extremely high temperatures due to chemical and biophysical feedbacks, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 19685–19690 CrossRef CAS PubMed.
- W. C. Skamarock and J. B. Klemp, A time-split nonhydrostatic atmospheric model for weather research and forecasting applications, J. Comput. Phys., 2008, 227, 3465–3485 Search PubMed.
- G. A. Grell, S. E. Peckham, R. Schmitz, S. A. McKeen, G. Frost, W. C. Skamarock and B. Eder, Fully coupled “online” chemistry within the WRF model, Atmos. Environ., 2005, 39, 6957–6975 CrossRef CAS.
- NCEP, NOAA, and USDC, NCEP FNL Operational Model Global Tropospheric Analyses, Continuing from July 1999, Research Data Archive at the National Center for Atmospheric Research, Computational and Information Systems Laboratory, updated daily, 2000, DOI:10.5065/D6M043C6.
- W. R. Stockwell, F. Kirchner, M. Kuhn and S. Seefeld, A new mechanism for regional atmospheric chemistry modeling, J. Geophys. Res.: Atmos., 1997, 102, 25847–25879 CrossRef CAS.
- R. Ahmadov, S. McKeen, A. Robinson, R. Bahreini, A. Middlebrook, J. De Gouw, J. Meagher, E. Y. Hsie, E. Edgerton and S. Shaw, A volatility basis set model for summertime secondary organic aerosols over the eastern United States in 2006, J. Geophys. Res.: Atmos., 2012, 117, D06301 CrossRef.
- R. J. Park, Y. J. Oak, L. K. Emmons, C.-H. Kim, G. G. Pfister, G. R. Carmichael, P. E. Saide, S.-Y. Cho, S. Kim and J.-H. Woo, Multi-model intercomparisons of air quality simulations for the KORUS-AQ campaign, Elementa, 2021, 9, 00139 Search PubMed.
- P. Bloomfield, J. A. Royle, L. J. Steinberg and Q. Yang, Accounting for meteorological effects in measuring urban ozone levels and trends, Atmos. Environ., 1996, 30, 3067–3077 CrossRef CAS.
- L. Carro-Calvo, C. Ordóñez, R. García-Herrera and J. L. Schnell, Spatial clustering and meteorological drivers of summer ozone in Europe, Atmos. Environ., 2017, 167, 496–510 Search PubMed.
- T.-M. Fu and H. Tian, Climate Change Penalty to Ozone Air Quality: Review of Current Understandings and Knowledge Gaps, Curr. Pollut. Rep., 2019, 5, 159–171 CrossRef CAS.
- Y. F. Lam, H. M. Cheung and C. C. Ying, Impact of tropical cyclone track change on regional air quality, Sci. Total Environ., 2018, 610, 1347–1355 CrossRef PubMed.
- A. Ito, S. Sillman and J. E. Penner, Global chemical transport model study of ozone response to changes in chemical kinetics and biogenic volatile organic compounds emissions due to increasing temperatures: Sensitivities to isoprene nitrate chemistry and grid resolution, J. Geophys. Res.: Atmos., 2009, 114, D09301 Search PubMed.
- L. Camalier, W. Cox and P. Dolwick, The effects of meteorology on ozone in urban areas and their use in assessing ozone trends, Atmos. Environ., 2007, 41, 7127–7137 Search PubMed.
- J. Han, H. Kim, M. Lee, S. Kim and S. Kim, Photochemical air pollution of Seoul in the last three decades, J. Korean Soc. Atmos. Environ., 2013, 29, 390–406 Search PubMed.
- H. C. Kim, D. Lee, F. Ngan, B.-U. Kim, S. Kim, C. Bae and J.-H. Yoon, Synoptic weather and surface ozone concentration in South Korea, Atmos. Environ., 2021, 244, 117985 Search PubMed.
- M. Qin, Y. She, M. Wang, H. Wang, Y. Chang, Z. Tan, J. An, J. Huang, Z. Yuan and J. Lu, Increased urban ozone in heatwaves due to temperature-induced emissions of anthropogenic volatile organic compounds, Nat. Geosci., 2025, 18, 50–56 CrossRef PubMed.
- J. Seo, D.-S. R. Park, J. Y. Kim, D. Youn, Y. B. Lim and Y. Kim, Effects of meteorology and emissions on urban air quality: a quantitative statistical approach to long-term records (1999–2016) in Seoul, South Korea, Atmos. Chem. Phys., 2018, 18, 16121–16137 CrossRef CAS.
- E. M. Oswald, L.-A. Dupigny-Giroux, E. M. Leibensperger, R. Poirot and J. Merrell, Climate controls on air quality in the Northeastern US: An examination of summertime ozone statistics during 1993–2012, Atmos. Environ., 2015, 112, 278–288 CrossRef CAS.
- H. Kim, J. Gil, M. Lee, J. Jung, A. Whitehill, J. Szykman, G. Lee, D.-S. Kim, S. Cho and J.-Y. Ahn, Factors controlling surface ozone in the Seoul Metropolitan Area during the KORUS-AQ campaign, Elementa, 2020, 8, 46 Search PubMed.
- X.-B. Li, B. Yuan, S. Wang, C. Wang, J. Lan, Z. Liu, Y. Song, X. He, Y. Huangfu and C. Pei, Variations and sources of volatile organic compounds (VOCs) in urban region: insights from measurements on a tall tower, Atmos. Chem. Phys., 2022, 22, 10567–10587 CrossRef CAS.
- N. Otero, H. W. Rust and T. Butler, Temperature dependence of tropospheric ozone under NOx reductions over Germany, Atmos. Environ., 2021, 253, 118334 CrossRef CAS.
- G. Tang, Y. Liu, X. Huang, Y. Wang, B. Hu, Y. Zhang, T. Song, L. Xiaolan, S. Wu and Q. Li, Aggravated ozone pollution in the strong free convection boundary layer, Sci. Total Environ., 2021, 147740 CrossRef CAS PubMed.
- J. H. Crawford, J.-Y. Ahn, J. Al-Saadi, L. Chang, L. K. Emmons, J. Kim, G. Lee, J.-H. Park, R. J. Park and J. H. Woo, The Korea–United States air quality (KORUS-AQ) field study, Elementa, 2021, 9, 00163 Search PubMed.
- Q. Yao, Z. Ma, J. Liu, Y. Qiu, T. Hao, L. Yao, J. Ding, Y. Tang, Z. Cai and S. Han, Effects of Meteorological Factors on the Vertical Distribution of Peroxyacetyl Nitrate in Autumn in Tianjin, Aerosol Air Qual. Res., 2022, 22, 220226 CrossRef CAS.
- Y. Qiu, W. Lin, K. Li, L. Chen, Q. Yao, Y. Tang and Z. Ma, Vertical characteristics of peroxyacetyl nitrate (PAN) from a 250-m tower in northern China during September 2018, Atmos. Environ., 2019, 213, 55–63 CrossRef CAS.
- R. A. Pielke and Y. Mahrer, Representation of the heated planetary boundary layer in mesoscale models with coarse vertical resolution, J. Atmos. Sci., 1975, 32, 2288–2308 Search PubMed.
- M. Herrmann, L. Cao, H. Sihler, U. Platt and E. Gutheil, On the contribution of chemical oscillations to ozone depletion events in the polar spring, Atmos. Chem. Phys., 2019, 19, 10161–10190 Search PubMed.
- W. Zhou, D. Cohan and B. Henderson, Slower ozone production in Houston, Texas following emission reductions: evidence from Texas Air Quality Studies in 2000 and 2006, Atmos. Chem. Phys., 2014, 14, 2777–2788 Search PubMed.
- C. C. K. Chou, C. Y. Tsai, C. J. Shiu, S. C. Liu and T. Zhu, Measurement of NOy during campaign of air quality research in Beijing 2006 (CAREBeijing-2006): Implications for the ozone production efficiency of NOx, J. Geophys. Res.: Atmos., 2009, 114, D00G01 Search PubMed.
- B. Rasulov, K. Hüve, I. Bichele, A. Laisk and Ü. Niinemets, Temperature response of isoprene emission in vivo reflects a combined effect of substrate limitations and isoprene synthase activity: a kinetic analysis, Plant Physiol., 2010, 154, 1558–1570 CrossRef CAS PubMed.
- X. Lyu, K. Li, H. Guo, L. Morawska, B. Zhou, Y. Zeren, F. Jiang, C. Chen, A. H. Goldstein and X. Xu, A synergistic ozone-climate control to address emerging ozone pollution challenges, One Earth, 2023, 6, 964–977 CrossRef.
- J. Gil, Seoul ground observation data in 2018 summer, Dataset, 2024, DOI:10.5281/zenodo.11045153.
| This journal is © The Royal Society of Chemistry 2026 |