Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Dual interphase design stabilized Mg-metal batteries
Wenwen Wang†
a,
Chengyi Zhang†b,
Xinquan Ma†a,
Weixiao Wang†a,
Qin Sua,
Juncai Long c,
Dongyao Zhua,
Jinghao Li
c,
Dongyao Zhua,
Jinghao Li a,
Ao Xua,
Rui Fangd,
Yuhang Dai
a,
Ao Xua,
Rui Fangd,
Yuhang Dai *e,
Guanbin Gao
*e,
Guanbin Gao *a and
Qinyou An
*a and
Qinyou An *a
*a
aState Key Laboratory of Advanced Technology for Materials Synthesis and Processing, Wuhan University of Technology, Wuhan 430070, PR China. E-mail: gbgao@whut.edu.cn; anqinyou86@whut.edu.cn
bSchool of Chemical Sciences, The University of Auckland, Auckland 1010, New Zealand
cDepartment of Industrial and Systems Engineering, The Hong Kong Polytechnic University, Hung Hom, Hong Kong 999077, PR China
dDepartment of Chemical and Environmental Engineering, University of Cincinnati, Cincinnati, OH 45220, USA
eInstitute for Integrated Cell-Material Sciences, Kyoto University, Yoshida, Sakyo-ku, Kyoto 606-8501, Japan. E-mail: dai.yuhang.72z@st.kyoto-u.ac.jp
First published on 24th April 2026
Abstract
Rechargeable magnesium metal batteries (RMBs) promise high volumetric energy density and resource sustainability, yet interphase instability remains a central barrier to practical full cells. Here, we show that single-electrode interphase stabilization often does not translate into durable full-cell operation, as instability can still emerge at the counter electrode during cycling. We therefore propose an energy-level-guided electrolyte design using a dual-additive strategy that simultaneously programs the formation of both the anode solid–electrolyte interphase (SEI) and the cathode–electrolyte interphase (CEI). By aligning the electrolyte's highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels with the cathode and anode potentials, the electrolyte undergoes spatially selective decomposition, enabling a continuous and uniform SEI/CEI pair. This programmed SEI/CEI pair expands the usable voltage window and supports deeper Mg2+ insertion/extraction, delivering 127.3 mAh g−1 (close to the theoretical capacity of Mo6S8, 128.8 mAh g−1) in Mg‖Mo6S8 full cells, with cycling durability competitive with state-of-the-art RMB reports. These findings establish paired-interphase programming as a general design paradigm to break the capacity-lifetime trade-off in RMBs.
Broader contextRechargeable magnesium metal batteries could offer a safer and more abundant alternative to lithium-based systems, with the potential for high volumetric energy density. In practice, performance is often limited not by the bulk materials but by the thin layers (interphases) that form where the electrolyte meets the anode and the cathode. When these interphases are nonuniform or fracture during cycling, side reactions accelerate, and much of the theoretical charge-storage capacity becomes difficult to access. A key challenge is that cathodic and anodic interphases in full cells are coupled, so improving one can unintentionally worsen reactions at the other. This work shows that both interfaces must be controlled together to build continuous and uniform interphases on each electrode. An energy-level-guided electrolyte design steers electrolyte breakdown to electrode surfaces, enabling concurrent formation of protective interphases on both the anode and cathode. This stabilizes higher-capacity charge storage in Mg‖Mo6S8 full cells, achieving state-of-the-art performance. More broadly, paired interphase control provides a general strategy for multivalent metal batteries, where long-term stability depends on coordinating the anode, cathode, and electrolyte as a single system. |
Introduction
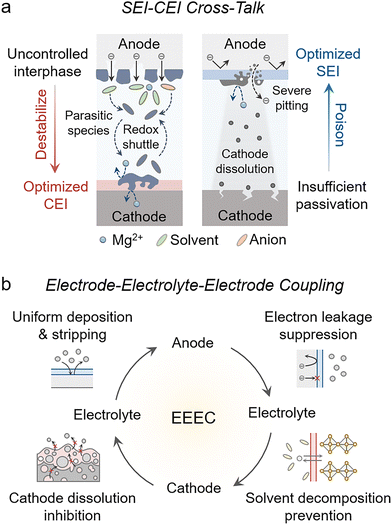
Rechargeable magnesium metal batteries (RMBs) promise a step change in safety and resource sustainability, yet progress remains limited by persistently uncontrolled interfacial evolution that is often misattributed.1–7 Most interphase studies still adopt a single-electrode optimization paradigm, focusing either on engineering a robust anode solid–electrolyte interphase (SEI)8–12 or on stabilizing a cathode–electrolyte interphase (CEI).13,14 In a practical full-cell, however, interfacial evolution is not a simple combination of two independently optimized halves. Rather, it is governed by strong electrode–electrolyte–electrode coupling (EEEC), where inadequate passivation at one electrode continuously generates mobile decomposition intermediates, reshapes electrolyte composition, and alters local charge-transfer behaviors, which in turn destabilizes the counter-electrode interphase (Fig. 1a). Consequently, optimizing only one interface may not deliver the anticipated improvement. It can even be counterproductive, because parasitic reactions at the unprotected electrode can dominate electrolyte–electrode chemistry and ultimately negate, or even outweigh, the benefits gained at the engineered interface.15–18 These considerations recast interfacial stability in RMBs from a local materials-engineering objective into a system-level coupled problem. On this basis, we highlight a critical yet underappreciated requirement: stable RMBs operation demands the simultaneous programming of paired interfaces, namely a CEI and a SEI that are co-designed to function cooperatively under EEEC (Fig. 1b).Several technical challenges remain to be addressed to realize durable battery operation. A functional interphase, particularly the SEI, must satisfy simultaneous morphological and compositional requirements.19 Morphologically, the interphase should be continuous and laterally uniform. Non-uniform thickness concentrates ionic flux and reaction current and enables electron tunneling through thin spots, accelerating side reactions and non-uniform Mg deposition. Cracks or discontinuities expose fresh surface and promote interphase fragmentation under deep cycling, increasing polarization and rapidly eroding stability.20 Compositionally, accumulated evidence suggests that a high fraction of fluorinated species is often beneficial for high ionic conductivity and low interfacial impedance,21,22 while an organic–inorganic composite architecture can provide the fracture resistance needed to accommodate interfacial stress and suppress cracking.23
Rather than relying on laborious trial-and-error screening, we return to a classical yet powerful design tool, aligning the frontier orbital energies of electrolyte components with the electrochemical potentials of both electrodes to program interphase formation. Specifically, by leveraging the matching between the highest occupied molecular orbital (HOMO)/lowest unoccupied molecular orbital (LUMO) levels of electrolyte and the cathode/anode energy landscape, oxidative and reductive decomposition can be made spatially selective.24 On the anode side, components with lower LUMO levels are preferentially reduced in a controlled manner, enabling the targeted construction of an electronically insulating yet Mg2+-conducting SEI. On the cathode side, components with higher HOMO levels are preferentially oxidized to form a stable CEI. In this framework, the effective “electrochemical stability window” is no longer dictated solely by the intrinsic thermodynamic limits of the bulk electrolyte, but is instead co-defined by the electronic structure and morphology of the interphases that emerge at each electrode.25 Such space-programmed interphase formation can decouple interfacial electron transport from ion transport, thereby suppressing parasitic reactions and markedly improving full-cell interfacial stability.
In this work, guided by the energy-level design principle described above, we formulate a dual-additive electrolyte that enables the concurrent formation of both the SEI and the CEI. This is achieved by pairing a salt component that is preferentially reduced at the anode with one that is preferentially oxidized at the cathode. Control electrolytes containing a single salt provide a mechanistic baseline, revealing that anode-targeted formulations produce a defective SEI, whereas cathode-targeted formulations yield an uneven CEI with ion-blocking phases. In contrast, the cooperative dual-additive chemistry generates a continuous, compact, and mutually matched SEI/CEI pair, motivating a system-level electrolyte strategy for paired interphase co-regulation in RMBs.
Results
Thermodynamics of electrolytes
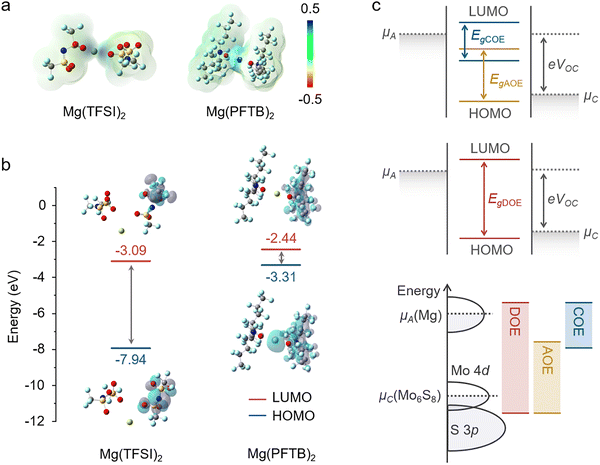
The narrow potential gap between Mg plating/stripping and cathode operation compresses both reductive and oxidative reactions into a confined energy range. In this landscape, we sought to disentangle how specific electrolytes govern interphase formation and stability. To this end, we focused on two model salts with contrasting oxidation/redox stabilities, Mg(TFSI)226–28 and Mg(PFTB)2.29,30 Density functional theory (DFT) calculations of electrostatic potential (ESP) were performed on Mg(TFSI)2 and Mg(PFTB)2 (Fig. 2a). The negative charge of TFSI− is largely delocalized across the sulfonyl framework. Multiple oxygen atoms provide multidentate coordination to Mg2+, strengthening local cation–anion electrostatic interactions and inducing a more pronounced polarization of the electron density. By contrast, PFTB− shows a weaker affinity toward Mg2+, leading to less stable coordination and a more labile cation–anion association. Accordingly, TFSI− is more likely to migrate together with Mg2+ and undergo preferential reductive decomposition at the low-potential anode, thereby contributing to SEI formation. In contrast, PFTB− exhibits a weaker affinity toward Mg2+, resulting in a higher fraction of free anions in solution. It is therefore more prone to participate in oxidative decomposition and interphase-forming reactions at the high-potential cathode, promoting the growth of a CEI. The calculated energy-level alignment reveals that TFSI− possesses a deep-lying LUMO of −3.09 eV, favoring electron acceptance, whereas PFTB− exhibits an elevated HOMO of −3.31 eV, rendering it more prone to oxidative activation (Fig. 2b). To rationalize the asymmetric interphase behavior of the two salts, we mapped their HOMO–LUMO energy levels onto the Mg metal Fermi level and the operating potentials of the Mo6S8 cathode (Fig. 2c). Using a single-salt electrolyte leads to an intrinsically unbalanced interphase. Mg(TFSI)2 tends to form a SEI on the anode, whereas Mg(PFTB)2 preferentially decomposes at high potentials and yields a CEI on the cathode. Crucially, the unprotected counter electrode becomes a continuous source of soluble/reactive decomposition products, which migrate across the bulk-electrolyte and undermine the interphase on the opposite electrode. As a result, apparent passivation on one side cannot be sustained unless both CEI and SEI are simultaneously stabilized. Building on these insights, we selected a magnesium aluminum chloride complex (MACC) electrolyte as the baseline system because of its well-established compatibility with Mg metal. Guided by the HOMO–LUMO energy alignment of the two additional salts, we created three electrolyte variants (Fig. S1) for systematic comparison. Adding Mg(TFSI)2 to the baseline produces an anode-oriented electrolyte (AOE). Incorporating Mg(PFTB)2 yields a cathode-oriented electrolyte (COE). When both salts are introduced simultaneously, the resulting mixed-salt formulation does not simply broaden the intrinsic thermodynamic stability window; rather, it promotes rapid dual-interphase formation on both electrodes, which kinetically suppresses further parasitic decomposition and gives rise to a wider effective operational stability window. This mixed-salt formulation is therefore designated as the dual-electrode-oriented electrolyte (DOE).Electrolyte-controlled interphase formation
To clarify how electrolyte chemistry governs interphase formation, transmission electron microscopy (TEM) was first employed to visualize the morphology of the CEI and SEI after 20 cycles in the three electrolytes (Fig. 3a–f). In DOE, both electrodes are uniformly passivated by continuous and conformal interphases. The Mo6S8 cathode is covered by a smooth and homogeneous CEI (Fig. 3a), while the Mg anode develops a compact and morphologically uniform SEI (Fig. 3d). In AOE, the absence of a discernible CEI leaves the Mo6S8 surface exposed (Fig. 3b). Although an ultrathin SEI forms on Mg, its discontinuity, together with local cracking and interfacial voids, indicates insufficient electronic passivation (Fig. 3e). In COE, both electrodes develop highly heterogeneous interphases. The CEI shows pronounced thickness variations with locally embedded crystalline MgO domains (Fig. 3c and Fig. S2).31,32 The Mg surface layer is likewise nonuniform (Fig. 3f). The coupled nonuniformity across both electrodes aligns with extensive interfacial side product formation and redistribution in COE.X-ray photoelectron spectroscopy (XPS) was then used to resolve the interphase compositions on the cathode and anode (Fig. S3–S6). In the cathode C 1s spectra (Fig. S3a), COE shows an increased contribution from oxygenated carbon species (C–O/–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), indicating an organic-rich surface layer.33 This enrichment is consistent with enhanced solvent decomposition under insufficient anode passivation, leading to the accumulation of by-products on the cathode surface. On the Mg anode, XPS spectra in the Mo 3d region show a distinct S 2s feature in AOE (Fig. S3b),34 consistent with the dissolution of cathode species and their subsequent deposition on the Mg surface in the absence of effective cathode-side protection. In addition, an extra low-binding-energy Mo 3d component assignable to Mo2+ is observed in both AOE and COE relative to DOE. The presence of this reduced Mo component is consistent with a more reductive anode interfacial environment arising from insufficient passivation.
O), indicating an organic-rich surface layer.33 This enrichment is consistent with enhanced solvent decomposition under insufficient anode passivation, leading to the accumulation of by-products on the cathode surface. On the Mg anode, XPS spectra in the Mo 3d region show a distinct S 2s feature in AOE (Fig. S3b),34 consistent with the dissolution of cathode species and their subsequent deposition on the Mg surface in the absence of effective cathode-side protection. In addition, an extra low-binding-energy Mo 3d component assignable to Mo2+ is observed in both AOE and COE relative to DOE. The presence of this reduced Mo component is consistent with a more reductive anode interfacial environment arising from insufficient passivation.
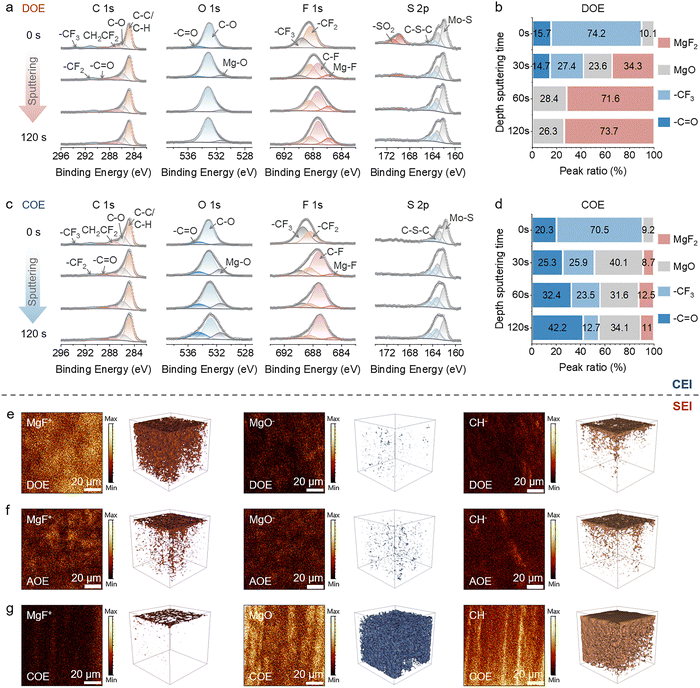
To further probe the depth-resolved chemistry of the interphases on both electrodes, we combined XPS sputter profiling of the cathode CEI with Time-of-flight secondary ion mass spectrometry (TOF-SIMS) analysis of the anode SEI (Fig. 4a–g).
For the cathode, XPS sputter profiling was performed for DOE and COE to track the evolution of C 1s, O 1s, F 1s, and S 2p signals over 0 to 120 s of sputtering (Fig. 4a and c). The DOE-derived CEI shows a stratified architecture, with an MgF2-rich inner layer and an adsorbed organic overlayer (Fig. 4b and Fig. S7a). By contrast, the COE-derived CEI is dominated by MgO and organic species throughout the probed depth (Fig. 4d and Fig. S7b). Together with the morphology and surface spectroscopy results above, these observations delineate three electrolyte-dependent interphase-formation regimes, as schematically summarized in Fig. S8. In DOE, the cathode interphase is stratified, comprising an inner MgF2-rich inorganic framework that provides a continuous Mg2+-conducting backbone, capped by an organic-rich outer layer that enhances mechanical compliance while limiting electron leakage (Fig. S8a). In AOE, the absence of a cathode-side interphase leaves Mo6S8 exposed and is accompanied by substantial cathode dissolution (Fig. S8b). In COE, insufficient anode-side passivation leads to pervasive electron leakage and solvent decomposition, and the resulting by-products deposit at the cathode to form an MgO- and organic-rich CEI that intrinsically constrains Mg2+ transport (Fig. S8c). The contrast between DOE and COE further indicates that the superior CEI uniformity in DOE arises not solely from cathode-directed oxidative interphase formation, but also from the coupled stabilization of both interfaces, as improved anode passivation suppresses cross-talk and by-product deposition during CEI growth.
TOF-SIMS was further used to probe the SEI composition and its depth-dependent distribution on the Mg anodes (Fig. 4e–g and Fig. S9). In DOE, MgF+ fragments are abundant and nearly uniform with depth, whereas MgO− signal is weak. CH− fragments that represent organic components are mainly concentrated at the near-surface region, indicating a layered SEI that mirrors the CEI (Fig. 4e). This trend is consistent with DFT-calculated adsorption energetics of representative salt-derived fragments on Mg, where strong F− binding favors rapid interfacial fluorination and formation of an MgF2-rich backbone, while more weakly bound fragments preferentially reside toward the outer region (Fig. S10). In COE, MgF+ is confined to a narrow near-surface region, while MgO− and CH− signals remain intense throughout the SEI, indicating an MgO and organic-dominated interphase, in line with the CEI composition in COE (Fig. 4g). The SEI formed in AOE exhibits intermediate characteristics, with a stronger MgF2 signature and reduced organic contribution compared with COE (Fig. 4f).
Electrochemical performance
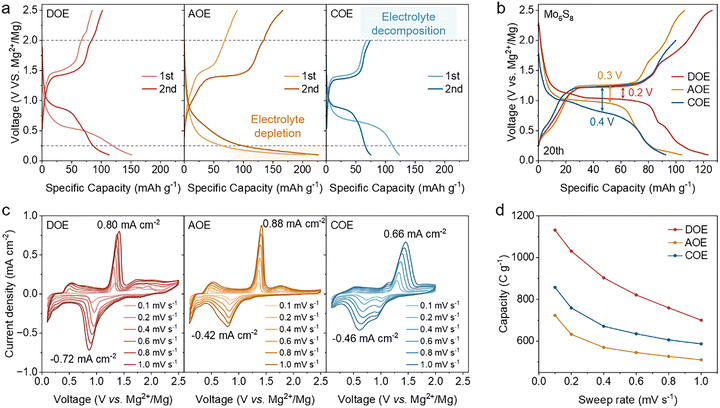
To connect interphase chemistries with cell-level behavior, we assembled Mg‖cathode cells and assessed how concurrent stabilization of both electrodes regulates the usable voltage window, interfacial kinetics, and electrochemical performance.We first compared the voltage profiles of Mg‖Mo6S8 cells over the first two cycles (Fig. 5a). AOE displays pronounced low-voltage tails near the discharge cutoff, while COE does not reach the upper cutoff of 2.5 V during charging (Note S1). In contrast, DOE delivers clean discharge and charge traces without obvious overdischarge or overcharge features, indicating a broader usable voltage window enabled by dual-interphase stabilization. After the electrochemical response stabilizes at the 20th cycle, DOE maintains a more sustained voltage plateau and the smallest charge–discharge hysteresis among the three electrolytes (Fig. 5b), consistent with reduced polarization under dual-interphase regulation. The kinetic signatures from cyclic voltammetry (CV) reinforce this trend. Across scan rates, DOE consistently shows the strongest redox features and the smallest peak separation, whereas AOE and COE exhibit stronger peak broadening and larger peak separation as the scan rate increases (Fig. 5c), consistent with progressively sluggish kinetics. Integrating the CV response to obtain the transferred charge (Q) reveals that DOE sustains the highest capacity at all scan rates (Fig. 5d).35,36
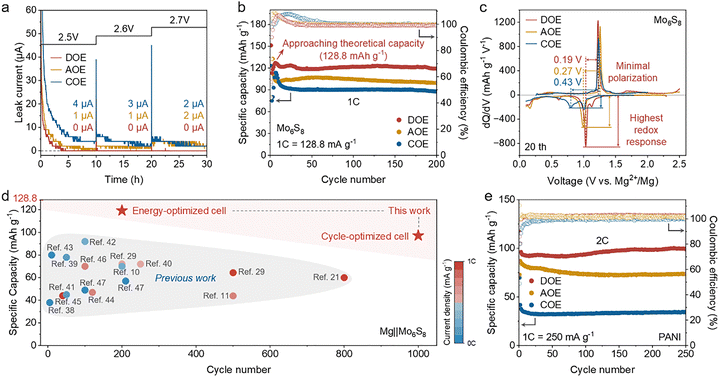
Oxidative stability beyond the nominal operating window was next quantified by potentiostatic hold tests in Mg‖Mo6S8 cells after 20 conditioning cycles. The cathode potential was stepped from 2.5 to 2.7 V (vs. Mg2+/Mg) in 0.1 V increments (Fig. 6a). DOE shows a nearly negligible and stable leakage current across the entire window, consistent with effective electronic passivation, whereas AOE and COE exhibit markedly higher leakage currents.37
Under 1C (128.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mA
mA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) g−1, based on the theoretical capacity of Mo6S8), Mg‖Mo6S8 cells using DOE deliver the highest specific capacity of 127.3 mAh g−1 (close to the theoretical value of Mo6S8), and show improved capacity retention relative to AOE and COE (Fig. 6b). Mg plating morphology after prolonged cycling supports this trend (Fig. S11). The dQ/dV profiles at the stabilized state (20th cycle) further show the sharpest redox features and the smallest peak separation in DOE (Fig. 6c), indicating reduced polarization during operation. When benchmarked against representative reports on Mg‖Mo6S8 cells, DOE-based cells fall within the high-performance regime in both specific capacity and cycling stability10,11,21,29,38–47 (Fig. 6d, Fig. S12, and Table S1). The same electrolyte dependence extends to an organic cathode. In Mg‖PANI cells, DOE provides the largest reversible capacity and a higher average discharge voltage at 1C (250
g−1, based on the theoretical capacity of Mo6S8), Mg‖Mo6S8 cells using DOE deliver the highest specific capacity of 127.3 mAh g−1 (close to the theoretical value of Mo6S8), and show improved capacity retention relative to AOE and COE (Fig. 6b). Mg plating morphology after prolonged cycling supports this trend (Fig. S11). The dQ/dV profiles at the stabilized state (20th cycle) further show the sharpest redox features and the smallest peak separation in DOE (Fig. 6c), indicating reduced polarization during operation. When benchmarked against representative reports on Mg‖Mo6S8 cells, DOE-based cells fall within the high-performance regime in both specific capacity and cycling stability10,11,21,29,38–47 (Fig. 6d, Fig. S12, and Table S1). The same electrolyte dependence extends to an organic cathode. In Mg‖PANI cells, DOE provides the largest reversible capacity and a higher average discharge voltage at 1C (250![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mA
mA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) g−1) (Fig. S13), and it maintains the highest capacity with the most stable retention during cycling at 2C (Fig. 6e and Fig. S14).
g−1) (Fig. S13), and it maintains the highest capacity with the most stable retention during cycling at 2C (Fig. 6e and Fig. S14).
The intrinsic stability of Mg plating and stripping in DOE is further supported by Mg‖Mg symmetric cells, which sustain lower and more stable polarization and markedly longer lifetimes than AOE and COE under both moderate and high throughput cycling conditions (Fig. S15 and S16). Consistently, rate tests and kinetic analysis in Mg‖Mg cells yield the highest exchange current density for DOE (Fig. S17), indicating accelerated interfacial charge transfer. In Mg‖Mo asymmetric cells, DOE likewise shows faster Mg plating and stripping kinetics with improved reversibility relative to AOE and COE (Fig. S18 and S19).
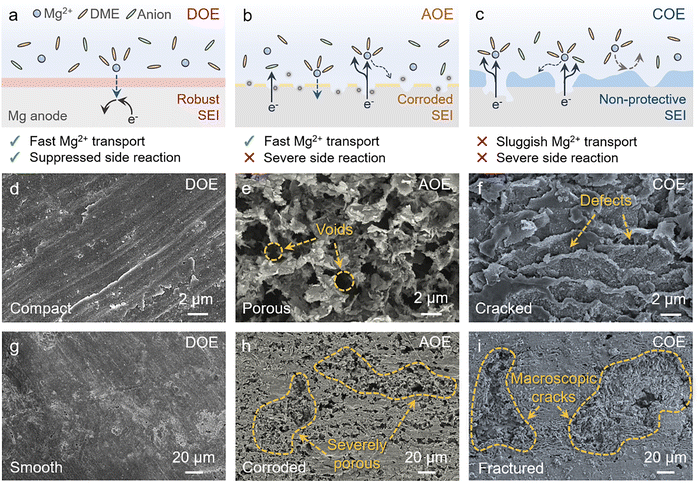
Anode stabilization in different electrolytes
The stability gap among the three electrolytes becomes most evident on the Mg surface after extended cycling, where the cumulative consequences of electrolyte decomposition, cathode-anode cross-talk, and the resulting interphase evolution are directly reflected in the post-cycling morphology (Fig. 7d–i). The schematics in Fig. 7a–c summarize the corresponding interphase scenarios in DOE, AOE, and COE, highlighting how these coupled processes shape Mg deposition and stripping.In DOE, the Mg surface is regulated by a spatially uniform SEI comprising an MgF2-rich inorganic framework and an organic overlayer. This interphase supports Mg2+ transport while maintaining electronic passivation, thereby limiting sustained electrolyte reduction and suppressing interfacial heterogeneity during cycling (Fig. 7a). The resulting uniform Mg plating and stripping preserve a smooth and dense Mg surface after cycling at both low and high magnifications (Fig. 7d and g).
In AOE, the absence of an effective CEI on the cathode side permits progressive dissolution of Mo6S8 and the generation of soluble cathode-derived species during cycling. Although an SEI may initially form on Mg, these soluble species can migrate to the anode and progressively perturb its subsequent evolution through chemical exchange and surface deposition, thereby compromising interphase integrity and triggering localized attack (Fig. 7b). The post-cycling Mg surface therefore becomes densely pitted, with abundant voids and corrosion-like features (Fig. 7e and h).
In COE, insufficient passivation on the anode side leaves the initially formed interphase electronically permeable, allowing persistent electron leakage during cycling. The resulting parasitic reduction sustains electrolyte decomposition and progressively perturbs subsequent interphase evolution through ongoing growth and restructuring, thereby generating pronounced spatial heterogeneity in interphase composition and thickness (Fig. 7c). The associated local variations in Mg2+ transport resistance and interfacial reactivity promote nonuniform Mg nucleation and subsequent plating/stripping, leading to terraced and step-like topographies after cycling (Fig. 7f and i).
Together, these post-cycling Mg morphologies provide an integrated readout of long-term interfacial evolution. By simultaneously suppressing cathode dissolution-driven cross-talk and anode electron-leakage-driven electrolyte decomposition, DOE preserves interfacial homogeneity and sustains uniform Mg plating and stripping during prolonged cycling, whereas single-sided interphase regulation in AOE or COE leads to self-reinforcing interfacial degradation and progressively nonuniform Mg plating and stripping.
Conclusion
In summary, we demonstrate that a dual-additive electrolyte enables deterministic interphase engineering at both electrodes in RMBs. Specifically, the Mg(TFSI)2–Mg(PFTB)2 formulation spatially programs reductive and oxidative decomposition to form interphases that are electronically passivating while allowing Mg2+ diffusion. TEM reveals the formation of a continuous and laterally uniform SEI/CEI pair. XPS depth profiling combined with TOF-SIMS confirms that the SEI/CEI is stratified, featuring an inner MgF2-rich inorganic framework, overlaid by an organic-rich outer layer. These conformal interphases suppress parasitic electrolyte reactions and electrode degradation, thereby extending the voltage window and kinetic stability. This is evidenced by a low leakage current observed during potentiostatic hold tests of Mg‖Mo6S8 cells. Consistently, cycling measurements reveal reduced polarization, while simultaneously delivering the highest specific capacity of 127.3 mAh g−1 at 1C (128.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mA
mA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) g−1, based on the theoretical capacity of Mo6S8), with improved capacity retention. The interphase-enabled benefits extend beyond Mg‖Mo6S8, translating directly to Mg‖PANI full-cells and underscoring the generality of this strategy. More broadly, our results establish programmable interphase chemistry as a design principle for stabilizing multivalent metal batteries.
g−1, based on the theoretical capacity of Mo6S8), with improved capacity retention. The interphase-enabled benefits extend beyond Mg‖Mo6S8, translating directly to Mg‖PANI full-cells and underscoring the generality of this strategy. More broadly, our results establish programmable interphase chemistry as a design principle for stabilizing multivalent metal batteries.
Methods
Preparation of electrolyte
All electrolyte preparations were performed in an argon-filled glovebox. Dry 1,2-dimethoxyethane (DME, Dodo chem, 5.0 mL) was used as the solvent. For the DOE electrolyte, AlCl3 (2.0 mmol) was first dissolved in DME, after which Mg(PFTB)2 (1.0 mmol; synthesized following the reported procedure29) was added. The mixture was magnetically stirred at room temperature for 30 min to give a clear solution. MgCl2 (1.0 mmol) was then added, and the solution was stirred at room temperature for 12 h until it became clear and colorless. Finally, Mg(TFSI)2 (1.25 mmol) was added, and the mixture was stirred for an additional 5 min to obtain the DOE electrolyte. The AOE and COE electrolytes were prepared using the same protocol, except that Mg(TFSI)2 was omitted for AOE, and Mg(PFTB)2 was omitted for COE. All electrolytes were used as-prepared without further purification.Preparation of Mo6S8 and PANI cathode
Mo6S8 and polyaniline (PANI) were used as the cathode active materials. Mo6S8 powders were synthesized according to previous reports.48 Commercial PANI (Alfa Aesar) was used as received. Mo6S8 cathodes were prepared by mixing Mo6S8, acetylene black and PVDF at a mass ratio of 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, whereas PANI cathodes were prepared by mixing PANI, Ketjen black and PTFE at a mass ratio of 6
1, whereas PANI cathodes were prepared by mixing PANI, Ketjen black and PTFE at a mass ratio of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. For both electrodes, the active material and conductive carbon were ground with isopropanol for at least 30 min, followed by addition of the binder to obtain a processable mixture. The Mo6S8 mixture was cast onto carbon paper and vacuum-dried at 110 °C for 12 h before punching discs (10 mm in diameter). For PANI cathodes, the mixture was first kneaded into a dough, then roll-pressed using a fixed roll gap to form a uniform film, and finally cut into square electrodes. The active-material loading was 1.5 ± 0.5 mg cm−2, and all electrodes were stored under vacuum at 60 °C until use.
1. For both electrodes, the active material and conductive carbon were ground with isopropanol for at least 30 min, followed by addition of the binder to obtain a processable mixture. The Mo6S8 mixture was cast onto carbon paper and vacuum-dried at 110 °C for 12 h before punching discs (10 mm in diameter). For PANI cathodes, the mixture was first kneaded into a dough, then roll-pressed using a fixed roll gap to form a uniform film, and finally cut into square electrodes. The active-material loading was 1.5 ± 0.5 mg cm−2, and all electrodes were stored under vacuum at 60 °C until use.
Electrochemical measurement
CR2016 coin cells were assembled in an argon-filled glovebox (H2O and O2 < 0.01 ppm). Prior to assembly, Mg disks were polished to remove the oxide layer and subsequently wiped with ethyl alcohol until the surface was visibly clean. The prepared electrodes and polished Mg discs were used as the working and counter electrodes, respectively. Glass fiber separators (Whatman GF/D, thickness 680 µm, diameter 17 mm) were placed between the electrodes. Each cell was filled with 80 µL of electrolyte. Additionally, a Mo foil (50 µm thickness, 18 mm diameter) was placed on the outer side of the cathode to mitigate Cl−-induced corrosion of the stainless-steel cell casing on the cathode side.The electrochemical tests of the batteries were carried out on a multi-channel battery test system (LAND CT2001A). Electrolyte polarization behavior was examined in symmetric Mg‖Mg cells. Cyclic voltammetry (CV), and linear sweep voltammetry (LSV), were performed on a BioLogic VMP3 multichannel electrochemical workstation. CV and LSV were conducted in asymmetric Mg‖Mo cells using Mo foil as the working electrode and Mg foil as both the counter and reference electrode, with scan rates of 20 and 10 mV s−1, respectively. The exchange current density (i0) for Mg plating was determined using eqn (1),49 where i denotes the current density, η is the total overpotential, F is the Faraday constant, R is the gas constant, and T is the absolute temperature.
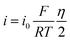 | (1) |
Characterization
To analyze electrolyte speciation and composition, Raman spectra were collected on a LabRAM HR Evolution spectrometer (HORIBA Scientific) using a 532 nm excitation laser, FTIR spectra were acquired on a Nicolet iS50 FTIR spectrometer (Thermo Scientific) in diffuse reflectance mode, and 19F NMR spectra were recorded on a Bruker 400 MHz spectrometer using DMSO-d6 as the solvent. The solid electrolyte interphase (SEI) and cathode–electrolyte interphase (CEI) formed in cells cycled with the three electrolytes were characterized by TEM, XPS, and TOF-SIMS. For TEM and XPS analysis of both the CEI and SEI, the cathode and anode were harvested from the same Mg‖Mo6S8 cells after 20 cycles. The morphology and structure of the interphase layers were examined by TEM on a JEM-F200 microscope operated at 200 kV. XPS measurements were performed on a K-Alpha spectrometer (Thermo Fisher Scientific) using a monochromatic Al Kα source. Ar-ion sputtering was carried out at 4 kV and 140 µA over a 3 mm × 3 mm area; the sputtering rate was calibrated on Ta2O5 to be 10 nm min−1. Binding energies were calibrated against the C 1s peak at 284.8 eV. To probe SEI components under more stringent conditions, TOF-SIMS surface analysis and depth profiling were conducted on a NanoTOF 3 instrument (ULVAC-PHI) using Mg electrodes harvested from Mg‖Mg symmetric cells after 20 cycles. X-ray diffraction (XRD) patterns were collected on a D2 Advance diffractometer (Bruker) using Cu Kα radiation to examine the structures of both cathode and anode electrodes harvested from Mg||Mo6S8 cells after the first cycle. The surface morphology of Mg anodes after prolonged cycling was examined by field-emission scanning electron microscopy (FESEM4000X, CIQTEK) after 200 cycles in Mg‖Mo6S8 cells. Before characterization, all cycled cells were disassembled in an Ar-filled glovebox. The electrodes were rinsed three times with DME to remove residual electrolyte and then dried at 60 °C for 2 h in the glovebox to remove residual solvent. All measurements and sample transfers were performed under Ar to avoid exposure to air.Computational details
Molecular electronic structures were calculated using density functional theory (DFT) as implemented in Gaussian.50 Geometry optimizations and electronic structure calculations were performed using the B3LYP hybrid exchange–correlation functional together with the 6-31G(d) basis set.51–54 All molecular geometries were fully optimized at this level of theory. Harmonic frequency calculations were performed at the same level to confirm that all optimized structures correspond to true local minima, with no imaginary frequencies. The energies of the frontier molecular orbitals (HOMO and LUMO) were extracted directly from the final SCF solutions and used as electronic structure descriptors.All DFT calculations were performed using a periodic slab model with the Vienna Ab initio Simulation Package (VASP).55,56 The generalized gradient approximation was used with Perdew–Burke–Ernzerhof exchange–correlation functional.57 The projector-augmented wave method was utilized to describe the electron–ion interactions, and the plane-wave basis expansion cut-off was set to 450![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) eV.58 All the adsorption geometries were optimized using a force-based conjugate gradient algorithm. Long-range dispersion interactions were included using the DFT-D3 empirical dispersion correction.59 The Mg surface was modeled using a periodic four-layer model, with the two lower layers fixed and the two upper layers relaxed. A p(3
eV.58 All the adsorption geometries were optimized using a force-based conjugate gradient algorithm. Long-range dispersion interactions were included using the DFT-D3 empirical dispersion correction.59 The Mg surface was modeled using a periodic four-layer model, with the two lower layers fixed and the two upper layers relaxed. A p(3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ×
× ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) supercell was chosen with 3
3) supercell was chosen with 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ×
× ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ×
× ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Monkhorst–Pack k-point mesh sampling for Brillouin-zone integration.60
1 Monkhorst–Pack k-point mesh sampling for Brillouin-zone integration.60
Author contributions
Yuhang Dai, Guanbin Gao, Qinyou An, Supervision and Conceptualization. Qinyou An, Funding acquisition. Wenwen Wang, Xinquan Ma, Writing – original draft. Wenwen Wang, Xinquan Ma, Yuhang Dai, Writing – review & editing. Chengyi Zhang, Data curation. Wenwen Wang, Weixiao Wang, Qin Su, Juncai Long, Dongyao Zhu, Jinghao Li, Ao Xu, Rui Fang, Investigation and Validation.Conflicts of interest
The authors declare no conflict of interest.Data availability
All relevant data that support the findings of this study are presented in the article and its supplementary information (SI). Supplementary information: Fig. S1–S38, supplementary Note 1–8, Table S1. See DOI: https://doi.org/10.1039/d6ee01732k.Acknowledgements
This work was supported by the National Key Research and Development Program of China (2023YFB3809500), the National Natural Science Foundation of China (52472246).References
- R. Mohtadi, O. Tutusaus, T. S. Arthur, Z. Zhao-Karger and M. Fichtner, The metamorphosis of rechargeable magnesium batteries, Joule, 2021, 5, 581–617, DOI:10.1016/j.joule.2020.12.021.
- D. Aurbach, Z. Lu, A. Schechter, Y. Gofer, H. Gizbar, R. Turgeman, Y. Cohen, M. Moshkovich and E. Levi, Prototype systems for rechargeable magnesium batteries, Nature, 2000, 407, 724–727, DOI:10.1038/35037553.
- Y. Li, S. Kumar, G. Yang, J. Lu, Y. Yao, K. Kang and Z. W. Seh, The contrast between monovalent and multivalent metal battery anodes, Science, 2025, 389, eadl5482, DOI:10.1126/science.adl5482.
- S. Hou, X. Ji, K. Gaskell, P. Wang, L. Wang, J. Xu, R. Sun, O. Borodin and C. Wang, Solvation sheath reorganization enables divalent metal batteries with fast interfacial charge transfer kinetics, Science, 2021, 374, 172–178, DOI:10.1126/science.abg3954.
- H. Dong, O. Tutusaus, Y. Liang, Y. Zhang, Z. Lebens-Higgins, W. Yang, R. Mohtadi and Y. Yao, High-power Mg batteries enabled by heterogeneous enolization redox chemistry and weakly coordinating electrolytes, Nat. Energy, 2020, 5, 1043–1050, DOI:10.1038/s41560-020-00734-0.
- S. Li, J. Zhang, S. Zhang, Q. Liu, H. Cheng, L. Fan, W. Zhang, X. Wang, Q. Wu and Y. Lu, Cation replacement method enables high-performance electrolytes for multivalent metal batteries, Nat. Energy, 2024, 9, 285–297, DOI:10.1038/s41560-023-01439-w.
- S.-B. Son, T. Gao, S. P. Harvey, K. X. Steirer, A. Stokes, A. Norman, C. Wang, A. Cresce, K. Xu and C. Ban, An artificial interphase enables reversible magnesium chemistry in carbonate electrolytes, Nat. Chem., 2018, 10, 532–539, DOI:10.1038/s41557-018-0019-6.
- S. Chen, H. Ma, Y. Du, W. Zhang and H. Y. Yang, Challenges and recent progress on anodes and their interfacial optimization towards high-performance rechargeable magnesium batteries, Mater. Today, 2024, 72, 282–300, DOI:10.1016/j.mattod.2023.12.002.
- R. Attias, M. Salama, B. Hirsch, Y. Goffer and D. Aurbach, Anode-Electrolyte Interfaces in Secondary Magnesium Batteries, Joule, 2019, 3, 27–52, DOI:10.1016/j.joule.2018.10.028.
- C. Li, A. Shyamsunder, B. Key, Z. Yu and L. F. Nazar, Stabilizing magnesium plating by a low-cost inorganic surface membrane for high-voltage and high-power Mg batteries, Joule, 2023, 7, 2798–2813, DOI:10.1016/j.joule.2023.10.012.
- X. Song, J. Sun, W. Ren, L. Wang, B. Yang, H. Ning, P. Zhang, Z. Caixiang, Z. Tie and X. Zhang, et al., Iodine Boosted Fluoro-Organic Borate Electrolytes Enabling Fluent Ion-Conductive Solid Electrolyte Interphase for High-Performance Magnesium Metal Batteries, Angew. Chem., Int. Ed., 2025, 64, e202417450, DOI:10.1002/anie.202417450.
- Z. Zhao-Karger, R. Liu, W. Dai, Z. Li, T. Diemant, B. P. Vinayan, C. Bonatto Minella, X. Yu, A. Manthiram and R. J. Behm, et al., Toward Highly Reversible Magnesium–Sulfur Batteries with Efficient and Practical Mg[B(hfip)4]2 Electrolyte, ACS Energy Lett., 2018, 3, 2005–2013, DOI:10.1021/acsenergylett.8b01061.
- D. Wang, X. Du, G. Chen, F. Song, J. Du, J. Zhao, Y. Ma, J. Wang, A. Du and Z. Cui, et al., Cathode Electrolyte Interphase (CEI) Endows Mo6 S8 with Fast Interfacial Magnesium-Ion Transfer Kinetics, Angew. Chem., Int. Ed., 2023, 62, e202217709, DOI:10.1002/anie.202217709.
- H. Shi, G. Wang, Z. Wang, L. Yang, S. Zhang, S. Dong, B. Qu, A. Du, Z. Li and X. Zhou, et al., Understanding the Cathode-Electrolyte Interfacial Chemistry in Rechargeable Magnesium Batteries, Adv. Sci., 2024, 11, 2401536, DOI:10.1002/advs.202401536.
- M. Bai, X. Tang, M. Zhang, H. Wang, Z. Wang, A. Shao and Y. Ma, An in-situ polymerization strategy for gel polymer electrolyte Si‖Ni-rich lithium-ion batteries, Nat. Commun., 2024, 15, 5375, DOI:10.1038/s41467-024-49713-z.
- C. Zhan, T. Wu, J. Lu and K. Amine, Dissolution, migration, and deposition of transition metal ions in Li-ion batteries exemplified by Mn-based cathodes – a critical review, Energy Environ. Sci., 2018, 11, 243–257, 10.1039/C7EE03122J.
- C. Zhao, H. Jeong, I. Hwang, T. Li, Y. Wang, J. Bai, L. Li, S. Zhou, C. C. Su and W. Xu, et al., Polysulfide-incompatible additive suppresses spatial reaction heterogeneity of Li-S batteries, Joule, 2024, 8, 3397–3411, DOI:10.1016/j.joule.2024.09.004.
- A. Arifiadi, F. Demelash, N. M. Abke, T. Brake, M. Vahnstiege, L. Alsheimer, S. Wiemers-Meyer, M. Winter and J. Kasnatscheew, Assessment of “Inverse” Cross-Talk (Anode to Cathode) in High-Voltage Li/Mn-Rich Layered Oxide ‖ Li Cells, Adv. Funct. Mater., 2025, 35, 2413958, DOI:10.1002/adfm.202413958.
- Y. Liang, H. Dong, D. Aurbach and Y. Yao, Current status and future directions of multivalent metal-ion batteries, Nat. Energy, 2020, 5, 646–656, DOI:10.1038/s41560-020-0655-0.
- Y. Chen, M. Li, Y. Jie, Y. Liu, Z. Zhang, P. Yu, W. Li, Y. Liu, X. Li and Z. Lei, et al., Dynamic evolution of cathode-electrolyte interphase in lithium metal batteries with ether electrolytes, Joule, 2025, 9, 101885, DOI:10.1016/j.joule.2025.101885.
- J. Xiao, X. Zhang, H. Fan, Y. Zhao, Y. Su, H. Liu, X. Li, Y. Su, H. Yuan and T. Pan, et al., Stable Solid Electrolyte Interphase In Situ Formed on Magnesium-Metal Anode by using a Perfluorinated Alkoxide-Based All-Magnesium Salt Electrolyte, Adv. Mater., 2022, 34, 2203783, DOI:10.1002/adma.202203783.
- B. Li, R. Masse, C. Liu, Y. Hu, W. Li, G. Zhang and G. Cao, Kinetic surface control for improved magnesium-electrolyte interfaces for magnesium ion batteries, Energy Storage Mater., 2019, 22, 96–104, DOI:10.1016/j.ensm.2019.06.035.
- Y. Lu, Q. Cao, W. Zhang, T. Zeng, Y. Ou, S. Yan, H. Liu, X. Song, H. Zhou and W. Hou, et al., Breaking the molecular symmetricity of sulfonimide anions for high-performance lithium metal batteries under extreme cycling conditions, Nat. Energy, 2024, 10, 191–204, DOI:10.1038/s41560-024-01679-4.
- H. Huang, Y. Hu, Y. Hou, X. Wang, Q. Dong, Z. Zhao, M. Ji, W. Zhang, J. Li and J. Xie, et al., Delocalized electrolyte design enables 600 Wh kg−1 lithium metal pouch cells, Nature, 2025, 644, 660–667, DOI:10.1038/s41586-025-09382-4.
- J. B. Goodenough and K.-S. Park, The Li-Ion Rechargeable Battery: A Perspective, J. Am. Chem. Soc., 2013, 135, 1167–1176, DOI:10.1021/ja3091438.
- W. Zhao, Z. Pan, Y. Zhang, Y. Liu, H. Dou, Y. Shi, Z. Zuo, B. Zhang, J. Chen and X. Zhao, et al., Tailoring Coordination in Conventional Ether-Based Electrolytes for Reversible Magnesium-Metal Anodes, Angew. Chem., Int. Ed., 2022, 61, e202205187, DOI:10.1002/anie.202205187.
- S. Wang, K. Wang, Y. Zhang, Y. Jie, X. Li, Y. Pan, X. Gao, Q. Nian, R. Cao and Q. Li, et al., High-entropy Electrolyte Enables High Reversibility and Long Lifespan for Magnesium Metal Anodes, Angew. Chem., Int. Ed., 2023, 62, e202304411, DOI:10.1002/anie.202304411.
- A. Morag, X. Chu, M. Marczewski, J. Kunigkeit, C. Neumann, D. Sabaghi, G. Z. Żukowska, J. Du, X. Li and A. Turchanin, et al., Unlocking Four-electron Conversion in Tellurium Cathodes for Advanced Magnesium-based Dual-ion Batteries, Angew. Chem., Int. Ed., 2024, 63, e202401818, DOI:10.1002/anie.202401818.
- J. Long, S. Tan, J. Wang, F. Xiong, L. Cui, Q. An and L. Mai, Revealing the Interfacial Chemistry of Fluoride Alkyl Magnesium Salts in Magnesium Metal Batteries, Angew. Chem., Int. Ed., 2023, 62, e202301934, DOI:10.1002/anie.202301934.
- J. Long, Z. He, G. Zhang, Y. Ding, J. Zhang, P. Liu, H. Xu, R. Wang, S. Tan and Q. An, et al., Solvation Chemistry of Nonaqueous Electrolytes for Rechargeable Magnesium Batteries, Adv. Mater., 2025, e10488, DOI:10.1002/adma.202510488.
- D. Gu, Y. Yuan, X. Peng, D. Li, L. Wu, G. Huang, J. Wang and F. Pan, Realizing high-stability anodes for rechargeable magnesium batteries via in situ -formed nanoporous Bi and nanosized Sn, J. Mater. Chem. A, 2024, 12, 26890–26901, 10.1039/D4TA04998E.
- Y. Li, X. Feng, G. Yang, W. Y. Lieu, L. Fu, C. Zhang, Z. Xing, M.-F. Ng, Q. Zhang and W. Liu, et al., Toward waterproof magnesium metal anodes by uncovering water-induced passivation and drawing water-tolerant interphases, Nat. Commun., 2024, 15, 9364, DOI:10.1038/s41467-024-53796-z.
- W. Xue, M. Huang, Y. Li, Y. G. Zhu, R. Gao, X. Xiao, W. Zhang, S. Li, G. Xu and Y. Yu, et al., Ultra-high-voltage Ni-rich layered cathodes in practical Li metal batteries enabled by a sulfonamide-based electrolyte, Nat. Energy, 2021, 6, 495–505, DOI:10.1038/s41560-021-00792-y.
- J. Tan, L. Feng, J. Shao, W. Zhang, H. Qin, H. Liu, Y. Shu, L. Yang, Y. Meng and Y. Tang, et al., In Situ Li+ Intercalation into Nanosized Chevrel Phase Mo6S8 toward Efficient Electrochemical Nitroarene Reduction, J. Am. Chem. Soc., 2025, 147, 10118–10128, DOI:10.1021/jacs.4c14111.
- Y. Dai, X. Liao, R. Yu, J. Li, J. Li, S. Tan, P. He, Q. An, Q. Wei and L. Chen, et al., Quicker and More Zn2+ Storage Predominantly from the Interface, Adv. Mater., 2021, 33, 2100359, DOI:10.1002/adma.202100359.
- V. Augustyn, J. Come, M. A. Lowe, J. W. Kim, P.-L. Taberna, S. H. Tolbert, H. D. Abruña, P. Simon and B. Dunn, High-rate electrochemical energy storage through Li+ intercalation pseudocapacitance, Nat. Mater., 2013, 12, 518–522, DOI:10.1038/nmat3601.
- G.-X. Li, R. Kou, A. Nguyen, K. Wang, Y.-S. Li, J. Lee, S. H. Kim and D. Wang, Long-cycling lithium-metal batteries via an integrated solid–electrolyte interphase promoted by a progressive dual-passivation coating, Nat. Energy, 2025, 10, 941–950, DOI:10.1038/s41560-025-01803-y.
- E. N. Keyzer, H. F. J. Glass, Z. Liu, P. M. Bayley, S. E. Dutton, C. P. Grey and D. S. Wright, Mg(PF6)2 -Based Electrolyte Systems: Understanding Electrolyte–Electrode Interactions for the Development of Mg-Ion Batteries, J. Am. Chem. Soc., 2016, 138, 8682–8685, DOI:10.1021/jacs.6b04319.
- Y. Guo, F. Zhang, J. Yang, F. Wang, Y. NuLi and S. Hirano, Boron-based electrolyte solutions with wide electrochemical windows for rechargeable magnesium batteries, Energy Environ. Sci., 2012, 5, 9100, 10.1039/c2ee22509c.
- A. Du, H. Zhang, Z. Zhang, J. Zhao, Z. Cui, Y. Zhao, S. Dong, L. Wang, X. Zhou and G. Cui, A Crosslinked Polytetrahydrofuran-Borate-Based Polymer Electrolyte Enabling Wide-Working-Temperature-Range Rechargeable Magnesium Batteries, Adv. Mater., 2019, 31, 1805930, DOI:10.1002/adma.201805930.
- W. Zhao, Z. Pan, Y. Zhang, Y. Liu, H. Dou, Y. Shi, Z. Zuo, B. Zhang, J. Chen and X. Zhao, et al., Tailoring Coordination in Conventional Ether-Based Electrolytes for Reversible Magnesium-Metal Anodes, Angew. Chem., Int. Ed., 2022, 61, e202205187, DOI:10.1002/anie.202205187.
- Y. He, Q. Li, L. Yang, C. Yang and D. Xu, Electrochemical-Conditioning-Free and Water-Resistant Hybrid AlCl3/MgCl2/Mg(TFSI)2 Electrolytes for Rechargeable Magnesium Batteries, Angew. Chem., Int. Ed., 2019, 58, 7615–7619, DOI:10.1002/anie.201812824.
- O. Tutusaus, R. Mohtadi, T. S. Arthur, F. Mizuno, E. G. Nelson and Y. V. Sevryugina, An Efficient Halogen-Free Electrolyte for Use in Rechargeable Magnesium Batteries, Angew. Chem., Int. Ed., 2015, 54, 7900–7904, DOI:10.1002/anie.201412202.
- T. J. Carter, R. Mohtadi, T. S. Arthur, F. Mizuno, R. Zhang, S. Shirai and J. W. Kampf, Boron Clusters as Highly Stable Magnesium-Battery Electrolytes, Angew. Chem., Int. Ed., 2014, 53, 3173–3177, DOI:10.1002/anie.201310317.
- R. Mohtadi, M. Matsui, T. S. Arthur and S. Hwang, Magnesium Borohydride: From Hydrogen Storage to Magnesium Battery, Angew. Chem., Int. Ed., 2012, 51, 9780–9783, DOI:10.1002/anie.201204913.
- F. Wang, H. Hua, D. Wu, J. Li, Y. Xu, X. Nie, Y. Zhuang, J. Zeng and J. Zhao, Solvent Molecule Design Enables Excellent Charge Transfer Kinetics for a Magnesium Metal Anode, ACS Energy Lett., 2023, 8, 780–789, DOI:10.1021/acsenergylett.2c02525.
- W. Ren, D. Wu, Y. NuLi, D. Zhang, Y. Yang, Y. Wang, J. Yang and J. Wang, An Efficient Bulky Mg[B(Otfe)4 ]2 Electrolyte and Its Derivatively General Design Strategy for Rechargeable Magnesium Batteries, ACS Energy Lett., 2021, 6, 3212–3220, DOI:10.1021/acsenergylett.1c01411.
- E. Lancry, E. Levi, A. Mitelman, S. Malovany and D. Aurbach, Molten salt synthesis (MSS) of Cu2Mo6S8—New way for large-scale production of Chevrel phases, J. Solid State Chem., 2006, 179, 1879–1882, DOI:10.1016/j.jssc.2006.02.032.
- Y. Guo, P. Niu, Y. Liu, Y. Ouyang, D. Li, T. Zhai, H. Li and Y. Cui, An Autotransferable g-C3 N4 Li+ -Modulating Layer toward Stable Lithium Anodes, Adv. Mater., 2019, 31, 1900342, DOI:10.1002/adma.201900342.
- M. Frisch, G. Trucks, H. Schlegel et al., GAUSSIAN 16, Gaussian Inc., Wallingford, CT, 2016 Search PubMed.
- A. D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys., 1993, 98, 5648–5652, DOI:10.1063/1.464913.
- W. J. Hehre, R. Ditchfield and J. A. Pople, Self—Consistent Molecular Orbital Methods. XII. Further Extensions of Gaussian—Type Basis Sets for Use in Molecular Orbital Studies of Organic Molecules, J. Chem. Phys., 1972, 56, 2257–2261, DOI:10.1063/1.1677527.
- C. Lee, W. Yang and R. G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B:Condens. Matter Mater. Phys., 1988, 37, 785–789, DOI:10.1103/PhysRevB.37.785.
- P. C. Hariharan and J. A. Pople, The influence of polarization functions on molecular orbital hydrogenation energies, Theor. Chim. Acta, 1973, 28, 213–222, DOI:10.1007/BF00533485.
- G. Kresse and J. Furthmüller, Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 11169–11186, DOI:10.1103/PhysRevB.54.11169.
- G. Kresse and J. Hafner, Ab initio molecular dynamics for liquid metals, Phys. Rev. B:Condens. Matter Mater. Phys., 1993, 47, 558–561, DOI:10.1103/PhysRevB.47.558.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized Gradient Approximation Made Simple, Phys. Rev. Lett., 1996, 77, 3865–3868, DOI:10.1103/PhysRevLett.77.3865.
- G. Kresse and D. Joubert, From ultrasoft pseudopotentials to the projector augmented-wave method, Phys. Rev. B:Condens. Matter Mater. Phys., 1999, 59, 1758–1775, DOI:10.1103/PhysRevB.59.1758.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu, J. Chem. Phys., 2010, 132, 154104, DOI:10.1063/1.3382344.
- H. J. Monkhorst and J. D. Pack, Special points for Brillouin-zone integrations, Phys. Rev. B: Condens. Matter Mater. Phys., 1976, 13, 5188–5192, DOI:10.1103/PhysRevB.13.5188.
Footnote |
| † These authors contributed equally: Wenwen Wang, Chengyi Zhang, Xinquan Ma, and Weixiao Wang. |
| This journal is © The Royal Society of Chemistry 2026 |