Open Access Article
Open Access ArticleBeyond rapid nucleation: unveiling the role of solvent–precursor interactions in antisolvent-free perovskite fabrication
Yunfan
Wang†
a,
Weiren
Zhao†
 b,
Juncheng
Wang†
c,
Zhuoqiong
Zhang†
b,
Juncheng
Wang†
c,
Zhuoqiong
Zhang†
 *d,
Ziyao
Yue
a,
Yuxuan
Zhang
a,
Yiting
Jiang
e,
Zixin
Zeng
*d,
Ziyao
Yue
a,
Yuxuan
Zhang
a,
Yiting
Jiang
e,
Zixin
Zeng
 a,
Jie
Zeng
a,
Hainam
Do
a,
Jie
Zeng
a,
Hainam
Do
 f,
Hong
Lu
fg,
Yuanhang
Cheng
*h,
Johnny C.
Ho
f,
Hong
Lu
fg,
Yuanhang
Cheng
*h,
Johnny C.
Ho
 a,
Tom
Wu
a,
Tom
Wu
 d,
Hin Lap
Yip
d,
Hin Lap
Yip
 ae,
Dewei
Zhao
ae,
Dewei
Zhao
 *c and
Sai-Wing
Tsang
*c and
Sai-Wing
Tsang
 *ai
*ai
aDepartment of Materials Science and Engineering, City University of Hong Kong, Hong Kong SAR 999077, P. R. China. E-mail: saitsang@cityu.edu.hk
bBioinformatics Center, Institute for Chemical Research, Kyoto University, Kyoto, Japan
cCollege of Materials Science and Engineering & Institute of New Energy and Low-Carbon Technology & Engineering Research Center of Alternative Energy Materials and Devices, Ministry of Education, Sichuan University, Chengdu, China. E-mail: dewei_zhao@hotmail.com
dDepartment of Applied Physics, The Hong Kong Polytechnic University, Hong Kong SAR 999077, P. R. China. E-mail: 18481965@life.hkbu.edu.hk
eSchool of Energy and Environment, City University of Hong Kong, Hong Kong SAR 999077, P. R. China
fDepartment of Chemical and Environmental Engineering, University of Nottingham Ningbo China, Ningbo, 315100, China
gPerovskite Research Institute, Yangzhou Precision Systems Inc., Yangzhou, 225000, China
hSchool of New Energy, Nanjing University of Science and Technology, Jiangyin, Jiangsu 21443, China. E-mail: yhcheng@njust.edu.cn
iCenter of Super-Diamond and Advanced Films (COSDAF), and Hong Kong Institute for Clean Energy, City University of Hong Kong, Hong Kong SAR 999077, P. R. China
First published on 8th April 2026
Abstract
Solution-processed antisolvent-free perovskites (AFPs) are promising candidates for scalable photovoltaic (PV) production. However, achieving high-quality AFP films typically requires stringent processing conditions, limiting reliability in large-scale manufacturing. Here, we employ machine learning (ML) to identify solvent additives that modulate the ambient temperature (TA) processing window. This approach successfully reveals an additive that enables a record-wide TA window, from 16 °C to 28 °C, with constantly high power conversion efficiencies (PCEs) exceeding 24%. Mechanistically, in contrast to the previously reported solvent-lead iodide (PbI2) interaction model, we demonstrate that anchoring formamidinium (FA) cations with the additive to form stable adducts is essential. This interaction effectively suppresses crystallization kinetics, facilitating uniform precursor distribution and high-quality film formation. Importantly, these results challenge the conventional crystallization paradigm for solution-processed perovskites, which emphasizes rapid and complete nucleation during the initial stage of film deposition. Instead, we find that a uniform distribution of precursor ions, even without any nucleation, is sufficient to achieve high-quality perovskite thin films. This work not only demonstrates an effective ML-guided solvent selection strategy but also provides fundamental insight into the primary crystallization requirements for scalable production of high-quality perovskite PV thin films.
Broader contextRecently, solution-processed antisolvent-free perovskites (AFPs) have become promising candidates for scalable photovoltaic manufacturing. However, high-quality AFP films are still suffering from the stringent control of ambient temperature (TA), typically constrained below 20 °C during solution deposition, which hinders large-scale reproducibility. Here, we employ a machine learning-guided strategy that replaces conventional trial-and-error approaches to identify solvents that significantly broaden the TA processing window, from 16 °C to 28 °C, while achieving champion power conversion efficiencies (PCEs) exceeding 24% and maintaining at least 90% of this PCE across the entire TA range. In addition, our findings reveal that the uniform precursor distribution enabled by solvent-based adducts is more critical for AFP fabrication than the traditionally emphasized rapid and complete perovskite nucleation. More importantly, a key prerequisite for forming such stable adducts has often been previously overlooked. In addition to stabilizing metal–halide–solvent adducts, it is also essential to stabilize the cations, thereby preventing competing reactions that can lead to non-uniform crystal growth. |
Introduction
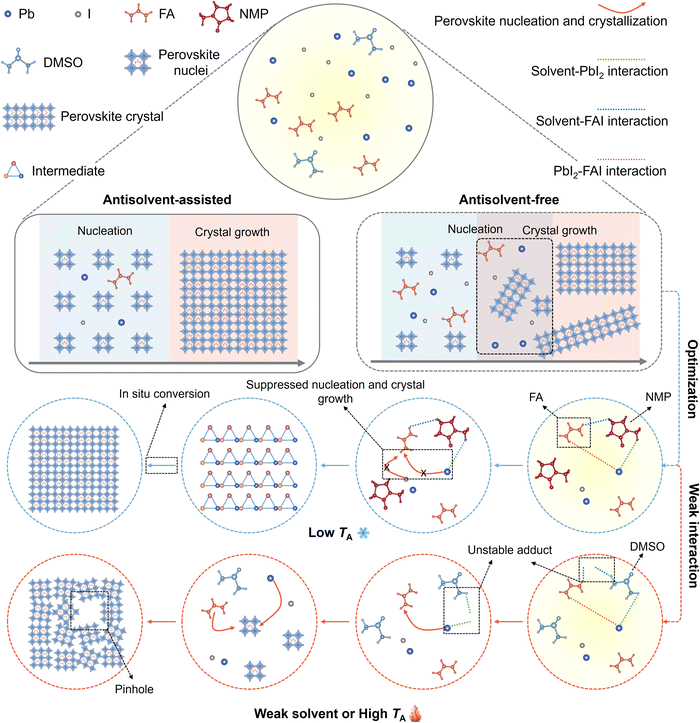
Due to the remarkable optoelectronic properties of perovskites, PSCs have achieved a rapid increase in power conversion efficiency (PCE) from 3.8% to 27.0% and are regarded as a leading candidate for next-generation photovoltaics.1–11 Among various fabrication approaches, solution-processed methods have garnered significant attention due to their simplicity, low cost, and scalability.12,13 In particular, the antisolvent-assisted one-step method has been a popular technique for fabricating high-efficiency PSCs in laboratory settings, as it can effectively accelerate nucleation to improve film morphology.2,14,15 However, this method is typically applied to small-area samples. For larger-area samples (>100 cm2), variations in solvent extraction efficiency caused by differing degrees of antisolvent contact in different regions can result in non-uniform nucleation rates across the film, compromising film quality and hindering large-scale manufacturing.9,16–18 Consequently, the development of antisolvent-free fabrication strategies has become an urgent priority.In antisolvent-free fabrication, the absence of antisolvent extraction significantly retards nucleation, leaving a non-uniform distribution of some perovskite nuclei and a large amount of precursor ions after film deposition. While the ions diffuse toward existing nuclei to promote crystal growth, certain regions become solute-depleted, which in turn suppresses additional nucleation and leads to incomplete film coverage and pinholes.19 This inherent competition between nucleation and crystal growth presents challenges in achieving uniform and high-quality perovskite films. To address this, Bu et al. introduced a solvent engineering strategy by adding N-methyl-2-pyrrolidone (NMP) into the formamidinium (FA)–cesium (Cs) lead triiodide precursor to form a PbI2–NMP adduct in the deposited film.20 This intermediate suppressed nucleation and crystal growth by preventing PbI2 from directly reacting with FAI. The perovskite nucleation and crystal growth are initiated during the subsequent thermal annealing process. Different from the antisolvent approach, the authors demonstrated that a high-quality antisolvent-free perovskite (AFP) film can be achieved by a “two-step” method: a solvent-based intermediate is first formed in the deposited film, which is then followed by its in situ conversion to the perovskite phase through reaction with FA during the annealing process. Using this approach, the authors achieved high-efficiency AFP solar cells (AFPSCs) with a small-area PCE of 23.35% and a 17.1 cm2 solar module with a PCE of 20.42%. Consequently, several attempts have been demonstrated using a similar approach to enhance the quality of AFP films.21–23 Despite the success in producing high-quality AFP films and promising photovoltaic devices, it is questionable whether the formation of PbI2–NMP adducts alone, as proposed by Bu et al., is sufficient to suppress the crystallization kinetics. It is because FA+ cations generally have a stronger interaction with the solvent and other precursor materials; the addition of FA+ in the precursor solution will accelerate the nucleation.
Additionally, a significant challenge remains in antisolvent-free PSCs (AFPSCs) due to their high sensitivity to ambient temperature (TA). The reported high-efficiency AFPSCs are typically fabricated at a TA below 18 °C to ensure optimal crystal and film quality.21,24 However, for industrial-scale applications, the cooling process increases production costs and reduces competitiveness in the photovoltaic market. Moreover, restricting the fabrication process to a narrow TA range complicates production control and negatively impacts the product yield in scalable manufacturing. Previous studies have shown that TA significantly influences crystallization dynamics, and therefore the film quality, in antisolvent-assisted perovskites. Although crystallization can be regulated via solvent engineering to control the ion coordination,25–28 a conventional trial-and-error solvent selection approach is time-consuming and inefficient, necessitating new methodologies for systematic optimization. Recently, machine learning (ML) has offered a promising solution to this challenge. With its ability to analyze large datasets and uncover complex correlations, ML has become a powerful tool in materials science, enabling efficient screening of candidates and minimizing experimental efforts.29,30 Recently, several attempts to employ ML in perovskites have demonstrated success in identifying new passivation molecules and interlayer materials to enhance the PSC lifetime and PCE.31,32 These promising results underscore the utility of ML in addressing persistent challenges in perovskite photovoltaics, offering a pathway to accelerate the commercialization of the technology.
In this work, we report an ML-guided solvent engineering strategy to regulate the nucleation kinetics in FA-based AFPs (FA0.92Cs0.08PbI3), enabling the fabrication of high-performance AFPSCs over a wide TA processing window. A database of several hundred solvents was preprocessed and clustered into groups based on a set of physicochemical descriptors. A cluster containing solvents that have previously been employed in AFP was selected for experimental validation and model training. Using this ML-guided strategy, we successfully achieved consistently high-efficiency AFPSCs with a PCE over 24% in a broad TA processing window from 16 °C to room temperature (24 °C). The developed recipe also demonstrated promising scalability, and a slot-die-coated AFP module with a large aperture area of 693 cm2 achieved a PCE of 17.6%. In addition, density functional theory (DFT) calculations and in situ characterization studies were conducted to further elucidate the mechanism underlying the performance enhancement of the devices induced by the ML-selected solvents. Our findings reveal that the uniform precursor distribution enabled by solvent-based adducts is more critical for AFP fabrication than the traditionally emphasized rapid and complete perovskite nucleation. More importantly, a key prerequisite for forming such stable adducts has often been previously overlooked. In addition to stabilizing metal–halide–solvent adducts, it is also essential to stabilize the cations, thereby preventing competing reactions that can lead to non-uniform crystal growth. Overall, the ML-assisted strategy bridges computational exploration with hypothesis-driven experiments, offering a synergistic pathway that accelerates solvent discovery while deepening the understanding of solvent–solute interactions during perovskite crystallization.
Results and discussion
Widening the TA window based on an ML-guided solvent engineering strategy
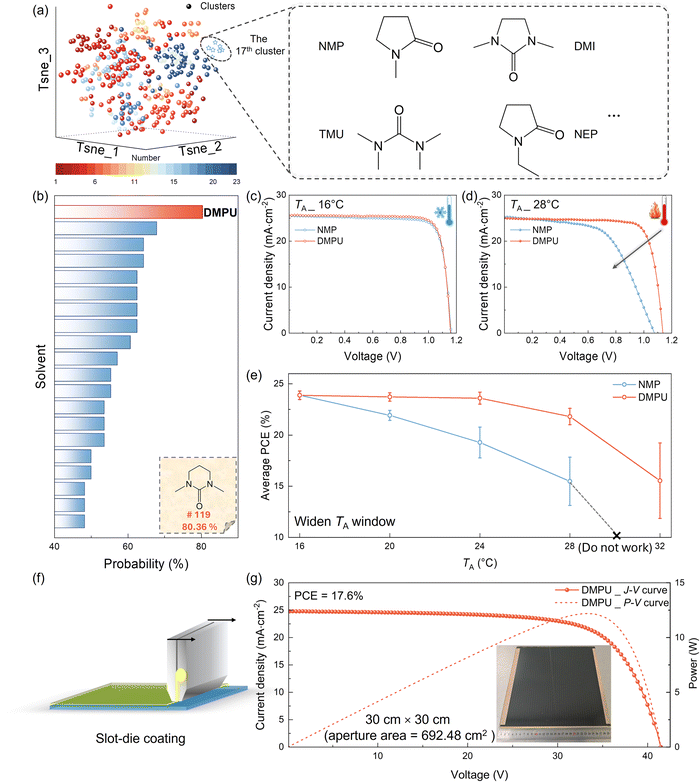
Solvent engineering is a critical strategy for regulating the crystallization process of perovskite films to improve their quality. However, traditional solvent selection often relies on experience and trial-and-error methods, which are time-consuming and inefficient. In this work, we proposed an integrated workflow (Fig. S1, SI) that combines ML with experimental validation to accelerate the identification of optimal solvents within a defined chemical space. Firstly, we constructed a dataset of 348 solvents characterized by 168 physicochemical descriptors (Fig. S2, SI). Using an algorithmic clustering approach, the dataset was organized into 23 clusters based on physicochemical properties (Fig. S3–S6, SI). For example, NMP (#13), a commonly used solvent additive in AFP fabrication, is assigned to the 17th cluster, as shown in Fig. 1a. Since solvents within the same cluster are expected to exhibit similar structural and physicochemical properties, we hypothesized that other solvents in this cluster could also serve as promising candidates. Guided by this classification, we can experimentally validate these solvents to assess their suitability. In contrast, dimethyl sulfoxide (DMSO _ #57), a widely used solvent additive in the antisolvent-assisted perovskite approach, is grouped into a different cluster. To verify the reliability of this grouping, we also performed experimental validation on DMSO. The corresponding morphological and optoelectronic properties demonstrate that the DMSO-treated film is entirely unsuitable for AFP fabrication due to poor surface coverage and uneven grain distribution (SI, Fig. S8). In contrast, most solvents in the 17th cluster are well-suited for AFP fabrication, effectively suppressing pinhole formation even at higher TA, such as tetramethylurea (TMU _ #2) and 1,3-dimethyl-2-imidazolidinone (DMI _ #120) (SI, Fig. S9–S11). However, variations in solvent performance are still observed within the same cluster. For instance, TMU-treated films exhibit superior morphology even at a high processing TA of 28 °C, whereas films treated with N-ethylpyrrolidone (NEP) still display numerous pinholes even when fabricated at a lower TA of 16 °C (SI, Fig. S11). These results indicate that clustering based solely on physicochemical descriptors is insufficient for accurately identifying optimal solvents. To enhance prediction accuracy, experimentally validated solvent data were used as labels to train supervised learning classification models (SI, Fig. S12 and S13). Following the training and validation phases, the ensemble model identified the top 20 solvents as promising candidates based on aggregated predictions, with solvent #119 ranking first due to its high probabilities (80.4%), as shown in Fig. 1b. This solvent, 1,3-dimethyl-3,4,5,6-tetrahydro-2(1H)-pyrimidinone (DMPU _ #119), was selected for further experimental validation. As shown in Fig. S14 (SI), all DMPU-treated films fabricated at a TA ranging from 16 to 28 °C exhibit a smooth, dense morphology with excellent coverage, confirming that DMPU effectively broadens the processing TA window for AFP fabrication. These results also validate the predictive accuracy of the optimized ML model.To systematically evaluate the impact of ML-selected solvents on broadening the perovskite processing TA window, the performances of the corresponding photovoltaic devices were characterized. In this study, AFPSCs with the structure of ITO/Me-2PACz/FAPbI3/PCBM/BCP/Ag were fabricated under various TA conditions. At a TA of 16 °C, both devices treated with different solvents exhibited comparable performance, with PCEs exceeding 24% (Fig. 1c and Table S1, SI). However, at 28 °C, the PCE of the commonly used NMP-treated AFPSCs dropped significantly from 24.05% to 15.60%, with a large reduction in the fill factor (FF) from 81.20% to 57.72% and the open-circuit voltage (VOC) from 1.16 V to 1.07 V under the reverse current–voltage sweep (Fig. 1d; forward scan data are provided in Fig. S16 and Table S2, SI). The VOC reduction is attributed to increased non-radiative recombination, which is consistent with the above PL mapping results (Fig. S10, SI). Pinholes on the NMP-treated film likely introduced defect states, increasing carrier recombination and lowering VOC. Additionally, these pinholes may have caused direct contact between the electron and hole transport layers, resulting in leakage currents and reduced FF. In contrast, devices treated with DMPU demonstrated improved performance, particularly in FF, due to the reduced number of pinholes in the perovskite films. Notably, DMPU-treated devices showed comparable performance at both 16 °C and 28 °C, indicating a superior temperature tolerance. In addition, as shown in Fig. S17 (SI), the external quantum efficiency (EQE)-integrated current density closely matched with the short-circuit current density (JSC) derived from the J–V curves. Regarding the reproducibility of the devices under various TA values, Fig. 1e shows the statistical results of over 20 NMP-treated and DMPU-treated devices fabricated from 16 °C to 32 °C. It is clearly shown that the PCE of NMP-treated devices dropped significantly while TA is higher than 20 °C, and became totally failed over 28 °C. The corresponding performance distribution increases from ±7.7% at 24 °C to ±15.2% at 28 °C. On the other hand, DMPU-treated devices demonstrated stable performance when fabricated from 16 °C to 24 °C, with a performance distribution of less than ±2.5%. Even at 28 °C, the performance distribution of DMPU-treated devices is still less than ±4.0%. The superior reproducibility of the performance of DMPU-treated devices directly correlates with their stability and scalability. More importantly, using the same approach with slot-die coating, 30 cm × 30 cm DMPU-treated AFP solar modules demonstrate impressive PCEs of 17.6% with an aperture area of 693 cm2, indicating their great potential for large-scale production (Fig. 1f and g). It is worth noting that, although the above results demonstrate the effectiveness of ML as a reliable and time-efficient tool for precise solvent selection, selection-algorithm-based ML approaches do not provide mechanistic insight into the origins of the improved performance, in particular: (i) the critical solvent properties influencing AFP film formation, and (ii) their impact on the crystallization process across different TA values.
Mechanism investigation
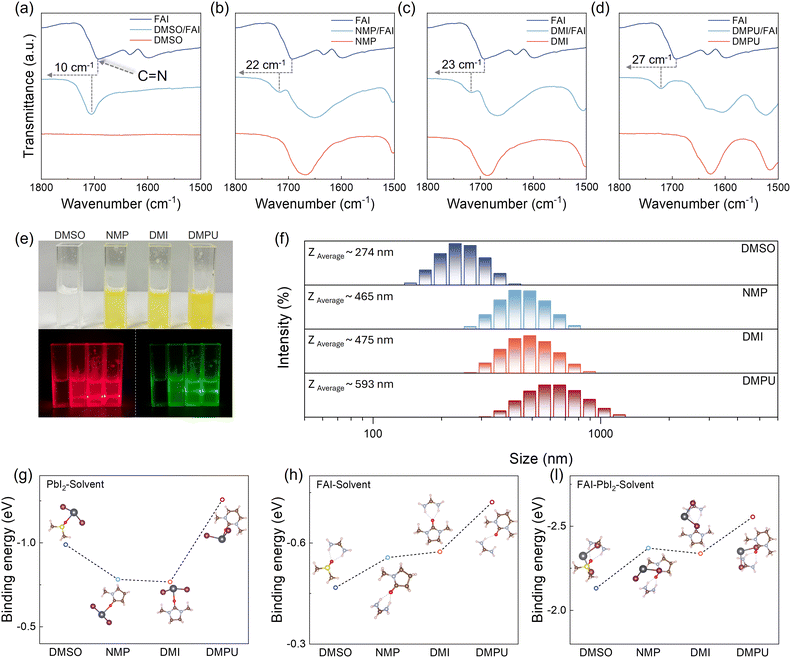
As aforementioned, the key to fabricating AFP films lies in mitigating the competition between nucleation and crystal growth. To achieve this, Bu et al. employed NMP to form a PbI2–NMP adduct to prevent PbI2 from directly reacting with FAI, thus resulting in a retarded crystal growth. This approach enables the formation of dense and uniform films. However, similar to NMP, DMSO has also been demonstrated to interact strongly with PbI2 to form a stable adduct,33 but the DMSO-treated films exhibit poor coverage. Therefore, it is questionable if any crucial interacting process during film formation has been overlooked.Generally, due to the rapid nucleation with the presence of FA cations, FA-based perovskites are often adopted in the development of AFP.21,23 Therefore, besides PbI2, we postulated that FA should play a role in the formation of solvent adducts. To explore the interaction between FA cations and solvents, Fourier-transform infrared (FTIR) spectroscopy was performed. It has been reported that FA cations would interact with Lewis base solvents through hydrogen bonding between the amine functional groups of FAI and the Lewis base, strengthening the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond in FAI.33,34 To analyze this effect, we compared the C
N bond in FAI.33,34 To analyze this effect, we compared the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching vibration peak of FAI in DMSO and three other ML-selected solvents. As shown in Fig. 2a–d, the C
N stretching vibration peak of FAI in DMSO and three other ML-selected solvents. As shown in Fig. 2a–d, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching vibration peak shifts to a higher wavenumber upon interaction with all the solvents. For FAI in DMSO, the C
N stretching vibration peak shifts to a higher wavenumber upon interaction with all the solvents. For FAI in DMSO, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N peak shows a 10 cm−1 shift from 1693 cm−1 (bare FAI) to 1700 cm−1. In contrast, the C
N peak shows a 10 cm−1 shift from 1693 cm−1 (bare FAI) to 1700 cm−1. In contrast, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N peak shows more than a two-fold shift from 22 cm−1 to 27 cm−1 in the other three ML-selected solvents, indicating stronger interactions between FAI and NMP, DMI, and DMPU compared to DMSO. Intuitively, the stronger interaction is attributed to the presence of C–N and C
N peak shows more than a two-fold shift from 22 cm−1 to 27 cm−1 in the other three ML-selected solvents, indicating stronger interactions between FAI and NMP, DMI, and DMPU compared to DMSO. Intuitively, the stronger interaction is attributed to the presence of C–N and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds in the ML-selected solvent molecules, where the lone pair electrons on nitrogen and oxygen atoms serve as hydrogen bond acceptors, promoting stronger hydrogen bonding with FAI, which is supported by the nuclear magnetic resonance (1H NMR) spectra results (Fig. S20, SI).33 Such an interaction can also be inferred from the appearance of a yellowish mixture (Fig. 2e) after mixing the colorless FAI with colorless NMP, DMI, and DMPU. In comparison, no significant color change is observed when FAI is mixed with DMSO. The presence of the yellowish solvent adducts is further confirmed by the Tyndall effect observed in the three ML-selected solvent/FAI solutions by dynamic light scattering (DLS) measurement (Fig. 2f).35,36 The average colloidal particle size (ZAverage) in the DMSO/FAI solution is only 274 nm, whereas the ZAverage values in NMP/FAI and DMI/FAI are almost two times larger, and it is even reaching 593 nm in DMPU/FAI. In addition, the increased absorption at ∼375 nm in UV-vis spectra of the FAI precursors suggests the presence of more dissociated I− ions in the solution, indicating stronger interactions between FA and the solvents, as shown in Fig. S21 (SI).
O bonds in the ML-selected solvent molecules, where the lone pair electrons on nitrogen and oxygen atoms serve as hydrogen bond acceptors, promoting stronger hydrogen bonding with FAI, which is supported by the nuclear magnetic resonance (1H NMR) spectra results (Fig. S20, SI).33 Such an interaction can also be inferred from the appearance of a yellowish mixture (Fig. 2e) after mixing the colorless FAI with colorless NMP, DMI, and DMPU. In comparison, no significant color change is observed when FAI is mixed with DMSO. The presence of the yellowish solvent adducts is further confirmed by the Tyndall effect observed in the three ML-selected solvent/FAI solutions by dynamic light scattering (DLS) measurement (Fig. 2f).35,36 The average colloidal particle size (ZAverage) in the DMSO/FAI solution is only 274 nm, whereas the ZAverage values in NMP/FAI and DMI/FAI are almost two times larger, and it is even reaching 593 nm in DMPU/FAI. In addition, the increased absorption at ∼375 nm in UV-vis spectra of the FAI precursors suggests the presence of more dissociated I− ions in the solution, indicating stronger interactions between FA and the solvents, as shown in Fig. S21 (SI).
To further evaluate the impact of solvent–FAI interactions on the stability of intermediate adducts, the binding energies of various solvent adducts were calculated using DFT, as shown in Fig. 2g–i. For solvent–PbI2 adducts, the binding energy of DMSO–PbI2 (−0.99 eV) is lower than that of NMP and DMI-coordinated adducts (−0.78 eV and −0.77 eV, respectively), which can be attributed to the higher donor number of DMSO.37,38 This result confirms that DMSO can form a more stable adduct with PbI2 (SI, Fig. S22). However, the binding energy between the FA cation and DMSO is only −0.47 eV, significantly higher than that of the other three ML-selected solvents. Similarly, the binding energy of the FAI–PbI2–DMSO adduct is also higher compared to the adducts formed with the other solvents. Particularly, only the binding energy of the FAI–PbI2–DMSO adduct is higher than that of FAPbI3 (−2.19 eV), suggesting that DMSO-based adducts exhibit reduced stability in an FA-rich environment. Consequently, the stronger hydrogen bonding interactions between FA and the ML-selected solvents can hinder the interaction between FAI and PbI2, thereby suppressing the early crystallization and increasing the solvent adduct stability.
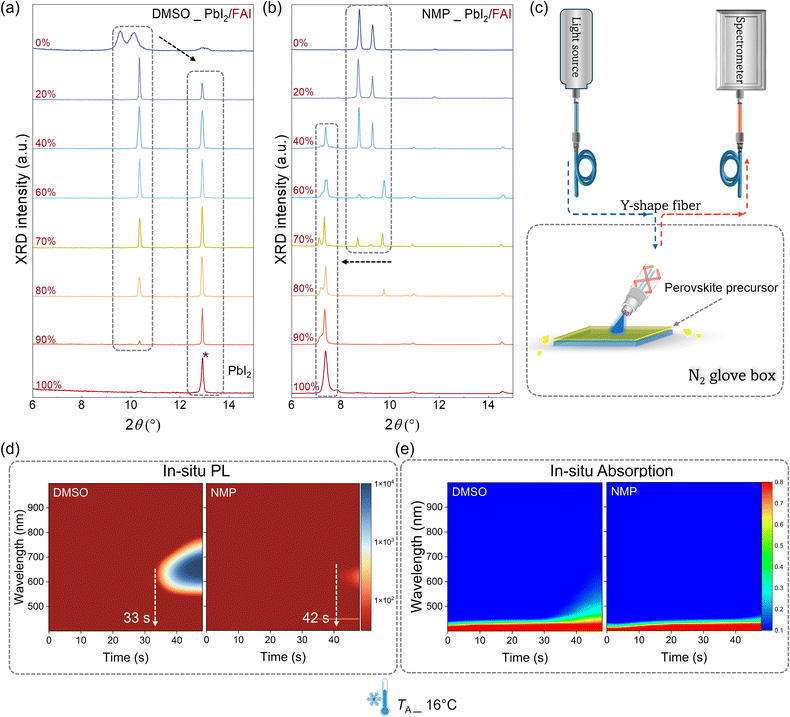
The effect of FA on solvent-based adduct formation was further investigated by X-ray diffraction (XRD) measurements on adduct films prepared from precursors with various FA contents, as shown in Fig. 3a and b. In this experiment, NMP, as a prototypical solvent used in AFP, was selected as a representative ML-identified solvent and compared with DMSO. Both DMSO and NMP were used as single solvents (without dimethylformamide (DMF)) to prepare the corresponding solvent-based adducts, with PbI2 and FAI as solutes. In the case of DMSO, the intensity of the DMSO-based adduct peak (around 10°) decreases as the FA content (FAI/(FAI + PbI2) ratio) increases from 0% to 100%.39 Simultaneously, the intensity of the PbI2 peak (12.9°) gradually increases with higher FA content.40 Notably, when the molar ratio of FAI in the precursor reaches 100%, no solvent adduct phase is detected, and only the PbI2 peak is observed. Combined with the previously discussed binding energy results, the FA cation appears to disrupt the DMSO–PbI2 adduct structure, thereby facilitating the formation of FAPbI3. However, FAPbI3 is unstable in humid air and readily degrades back to PbI2, which explains the observed precipitation of PbI2 during XRD testing. In contrast, the NMP–PbI2 adduct remains stable after adding 20% FA. With further increases in FA content, a new peak at 7° appears, corresponding to the gradual formation of the NMP–PbI2–FAI adduct.20,21 Notably, no PbI2 or δ-phase is detected, highlighting the superior stability of the NMP-based adduct even in an FA-rich environment. Time-resolved XRD measurements further confirm that the solvent adducts are the primary product formed during the spin-coating (Fig. S23, SI), and the phase conversion can only be observed in the DMSO–PbI2 adduct, while the NMP–PbI2–FAI adduct is relatively stable.
We sought to investigate perovskite nucleation and crystal growth dynamics with different solvents and examine their corresponding effects under varying ambient temperatures through an in situ optical probing method. Fig. 3c illustrates the custom-built in situ optical setup for PL and absorption measurements, housed within a temperature-controlled and nitrogen-filled glovebox (detailed information is given in Methods of supplementary information). Fig. 3d presents 2D contour plots of in situ PL results recorded during spin-coating at 16 °C, where “0 s” on the x-axis marks the onset of spin-coating. For the DMSO-treated sample, gradual solvent evaporation leads to an increase in solute concentration, initiating nucleation at 33 s. A noticeable PL signal was detected at this point, followed by a rapid increase in PL intensity over the next 20 s, indicating the formation of a large number of luminous nuclei—a hallmark of fast nucleation (detailed intensity and peak position changes are given in Fig. S24, SI).25,41,42 Concurrently, the PL peak shifted from 600 nm to 700 nm, which is attributed to grain growth reducing the quantum confinement effect.43–45 These observations suggest that the DMSO-treated film undergoes rapid nucleation and crystal growth simultaneously, with competition between these processes potentially causing pinhole formation. In contrast, the onset of the PL signal in the NMP-treated sample was delayed until 42 s. The slower rise in PL intensity indicates fewer luminous nuclei and thus slower nucleation, it is attributed to the strong bonding between NMP and FA cations, as noted earlier. In addition, in the NMP-treated sample, the constant PL peak wavelength at 620 nm throughout the process suggests retarded growth of the formed nuclei. Consistently, the in situ absorption results (Fig. 3e and Fig. S25, SI) also revealed crystal growth in the DMSO-treated sample, whereas the NMP-treated film also showed absorption from intermediate adducts.46 For the DMSO-treated sample, the absorption edge continuously redshifts toward longer wavelengths, reaching approximately 600 nm. In contrast, the absorption edge of the NMP-treated film remains within the characteristic range of the solvent-based adduct phase (400–450 nm), indicating the persistence of the intermediate adduct phase throughout the entire spin-coating process. As shown in Fig. S26 (SI), compared to NMP, no obvious perovskite nucleation was observed in samples using DMI and DMPU as solvents. This observation is consistent with the computational results, which indicate the formation of strong solvent-based adducts.
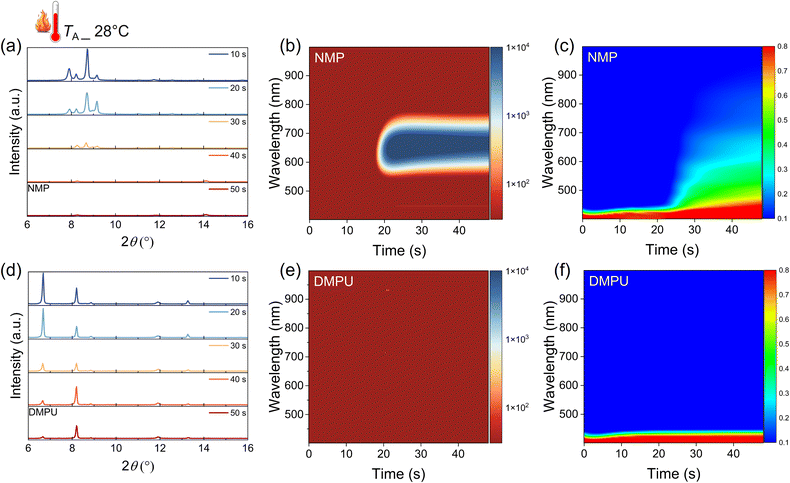
More importantly, such stable adduct formation is believed to be critical for expanding the TA processing window. Indeed, an examination of the crystalline structure of the DMPU-treated film, as shown in Fig. S27 (SI), shows that the DMPU-based adduct exhibits greater thermal stability. Such a stable intermediate will strongly affect the crystallization process. As shown in Fig. 4a and d, the time-resolved XRD spectra indicate that the NMP-based adduct signal decreases dramatically after spin-coating for 30 s at a TA of 28 °C. In contrast, strong adduct diffraction is detected in the whole spin-coating process in the DMPU-treated film, indicating better stability. This is consistent with the previous DFT results, where DMPU has stronger binding to the precursor ions. The effect of stable intermediates on the crystallization is further investigated by in situ optical measurements. As shown in Fig. 4b, when the TA is increased to 28 °C, the PL signal in the NMP-treated film appears earlier than in the film fabricated at 16 °C (at 18 s and 42 s, respectively). Furthermore, the PL intensity increases more rapidly, accompanied by a noticeable redshift in the peak position (detailed intensity and peak position changes are given in Fig. S28, SI). This suggests a fast crystallization process in the NMP-treated film at higher temperatures, leading to the formation of pinholes on the film surface. In contrast, no detectable PL signal is observed during spin-coating of the DMPU-treated film at 28 °C, indicating retarded perovskite nucleation (Fig. 4e). This is comparable to the performance of the NMP-treated film at a lower temperature. The delayed nucleation is attributed to the strong interaction between DMPU and FA cations, which suppresses the reaction between FAI and PbI2 to form perovskite nuclei. As a result, the DMPU-treated AFP film fabricated at 28 °C still forms a dense, pinhole-free surface. Consistently, no sign of crystallization can be observed in the DMPU-treated film in the in situ absorption results, in sharp contrast to the NMP-treated films (Fig. 4c and f). Notably, due to the strong binding affinity of DMPU, high concentration of DMPU will lead to a perovskite film with poor morphology, as shown in Fig. S29 (SI). Therefore, a mixed DMPU:NMP is necessary to optimize the film morphology. On the other hand, an interesting phenomenon during spin-coating was observed: no PL or absorption signals in high-quality AFP films emerged, regardless of low or high TA (Fig. 3d, e and 4e, f), indicating the absence of perovskite nuclei formation. This observation challenges the long-standing belief that rapid nucleation is essential for achieving high-quality films by ensuring uniform nuclei distribution and controlled grain growth.47,48 However, in the AFP system, rapid nucleation is inherently challenging and often uncontrollable. Instead, the formation of an intermediate phase emerges as a viable alternative, facilitating a uniform distribution of precursor ions and circumventing the limitations of rapid nucleation.
Overall, nucleation and crystal growth are critical factors that determine the quality of perovskite thin films in solution-processed fabrication. According to the above results, the differences in crystallization dynamics between antisolvent-assisted perovskite and AFP fabrication processes are illustrated in Fig. 5. In antisolvent-assisted fabrication, the rapid extraction of the antisolvent promotes fast and complete nucleation, ensuring a uniform distribution of perovskite nuclei and precursor ions before crystal growth, resulting in dense perovskite films. However, the crystallization rate will be further increased at a higher TA, which may lead to the nuclei collision and aggregation, finally resulting in the inhomogeneous growth (cracks) due to solute depletion. In contrast, AFP fabrication generally exhibits a slower nucleation rate. In this process, unconsumed precursor ions are simultaneously used for nucleation and diffusion toward existing nuclei, which suppresses further nucleation and promotes crystal growth, inhibiting the uniform distribution of nuclei and ions. This leads to poor film coverage and the formation of pinholes. To address this issue, solvent engineering strategies have been employed to enable uniform precursor ion distribution by forming solvent–PbI2 adducts in the as-deposited film prior to crystal growth during thermal annealing. However, previous studies have primarily focused on the impact of solvent adducts on AFP fabrication while overlooking their formation mechanism. This oversight has led to the unexpected observation that DMSO, a commonly used solvent additive in other solution-processed methods, is unsuitable for AFP fabrication, a phenomenon that remains poorly understood. Here, we found that the interaction between FA and the solvent plays a crucial role in solvent adduct formation. FA disrupts the solvent–PbI2 adduct by interacting with PbI2, whereas solvents containing nitrogen and oxygen atoms can form hydrogen bonds with FA cations (also may be related to the rotatable bond count, as shown in Fig. S30, SI). These bonds effectively prevent the reaction between PbI2 and FAI, stabilizing the solvent adduct and slowing perovskite crystallization. Importantly, if the interaction between FA and the solvent (e.g., DMSO) is insufficiently strong, the solvent adduct cannot be maintained under FA-rich conditions, resulting in poor film morphology. Additionally, this interaction between FA and the solvent can be disrupted at a higher TA, which limits the processing TA of AFPSCs to below 18 °C. Therefore, selecting a solvent capable of strongly interacting with FA cations to form a stable solvent adduct under FA-rich conditions is critical for achieving high-quality films and expanding the processing TA window in AFP fabrication.
 | ||
| Fig. 5 Schematic diagram of possible phase evolution in the crystallization of solution-processed perovskites. | ||
Conclusion
We demonstrate an ML-guided strategy for identifying solvent additives to widen the TA processing window for FA-based AFP fabrication. Using this approach, high-efficiency AFPSCs with PCE exceeding 24% are achieved over a broad TA range from 16 °C to room temperature (24 °C). Mechanistically, we demonstrate that achieving a uniform distribution of precursor ions prior to crystal growth is a critical requirement for forming high-quality AFP films, thereby challenging the prevailing emphasis on rapid and complete perovskite nucleation. This uniform ion distribution arises from the simultaneous formation of stable solvent adducts with both precursor anions and cations. Consequently, in addition to the strong solvent–PbI2 coordination alone, our findings reveal that forming stable adducts between the solvent and FA+ is essential to suppress the nucleation. Overall, this study presents a systematic ML-guided strategy for solvent selection and provides new insights into the role of perovskite intermediates in governing crystallization pathways, offering practical guidance for the scalable fabrication of high-performance AFP thin films.Author contributions
Y. W., W. Z., and Z. Z. conceived and designed the research. Y. W., Z. Y., and Z. Z. performed in situ measurements and data analysis. Z. Z., J. W., and H. L. carried out the device fabrication. J. W. and H. D. carried out the device characterization studies. W. Z. performed machine learning analysis. Y. Z. conducted the DFT calculation. Y. J. and J. Z. carried out film characterization. Y. C., J. H., T. W., and H. L. Y. helped to optimize the perovskite solar cells. Y. W. and W. Z. wrote the first draft of the paper. Z. Z., Y. C., D. Z., and S. W. T. supervised the project and contributed to the manuscript draft. All co-authors discussed the results and commented on the paper.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI), which includes experimental methods, machine learning details, 31 figures, and 2 tables. See DOI: https://doi.org/10.1039/d6ee00170j.Acknowledgements
We acknowledge the General Research Fund (CityU 11304420 and CityU 11317422) from the Research Grants Council of Hong Kong SAR, China.References
- A. Kojima, K. Teshima, Y. Shirai and T. Miyasaka, J. Am. Chem. Soc., 2009, 131, 6050–6051 CrossRef CAS PubMed.
- N. J. Jeon, J. H. Noh, Y. C. Kim, W. S. Yang, S. Ryu and S. I. Seok, Nat. Mater., 2014, 13, 897–903 CrossRef CAS PubMed.
- H. S. Jung and N. G. Park, Small, 2015, 11, 10–25 CrossRef CAS.
- H. Chen, C. Liu, J. Xu, A. Maxwell, W. Zhou, Y. Yang, Q. Zhou, A. S. Bati, H. Wan and Z. Wang, Science, 2024, 384, 189–193 CrossRef CAS.
- M. A. Green, E. D. Dunlop, M. Yoshita, N. Kopidakis, K. Bothe, G. Siefer, D. Hinken, M. Rauer, J. Hohl-Ebinger and X. Hao, Prog. Photovoltaics, 2024, 32, 425–441 Search PubMed.
- Y. Wang, P. Xiang, A. Ren, H. Lai, Z. Zhang, Z. Xuan, Z. Wan, J. Zhang, X. Hao and L. Wu, ACS Appl. Mater. Interfaces, 2020, 12, 53973–53983 CrossRef CAS.
- M. Jung, S.-G. Ji, G. Kim and S. I. Seok, Chem. Soc. Rev., 2019, 48, 2011–2038 RSC.
- J.-W. Lee, S. Tan, S. I. Seok, Y. Yang and N.-G. Park, Science, 2022, 375, eabj1186 CrossRef CAS PubMed.
- Z. Li, T. R. Klein, D. H. Kim, M. Yang, J. J. Berry, M. F. Van Hest and K. Zhu, Nat. Rev. Mater., 2018, 3, 1–20 CAS.
- National Renewable Energy Laboratory. (2025 February 13). Best Research-Cell Efficiency Chart. https://www2.nrel.gov/pv/cell-efficiency, (accessed 08 16, 2023).
- Z. Zhang, Y. Wang, W. Wang, Y. Huang, S. Ouyang, Y. Sun, F. Wang, X. Zhou, G. Xing and S. K. So, Adv. Mater., 2026, e19793 CrossRef CAS.
- J. Prakash, A. Singh, G. Sathiyan, R. Ranjan, A. Singh, A. Garg and R. K. Gupta, Mater. Today Energy, 2018, 9, 440–486 CrossRef.
- L. Yan, H. Huang, P. Cui, S. Du, Z. Lan, Y. Yang, S. Qu, X. Wang, Q. Zhang and B. Liu, Nat. Energy, 2023, 8, 1158–1167 CrossRef CAS.
- M. Yang, Z. Li, M. O. Reese, O. G. Reid, D. H. Kim, S. Siol, T. R. Klein, Y. Yan, J. J. Berry and M. F. Van Hest, Nat. Energy, 2017, 2, 1–9 Search PubMed.
- H.-H. Huang, Q.-H. Liu, H. Tsai, S. Shrestha, L.-Y. Su, P.-T. Chen, Y.-T. Chen, T.-A. Yang, H. Lu and C.-H. Chuang, Joule, 2021, 5, 958–974 CrossRef CAS.
- C. Liu, Y.-B. Cheng and Z. Ge, Chem. Soc. Rev., 2020, 49, 1653–1687 RSC.
- L. Chao, T. Niu, W. Gao, C. Ran, L. Song, Y. Chen and W. Huang, Adv. Mater., 2021, 33, 2005410 CrossRef CAS.
- Z. Saki, M. M. Byranvand, N. Taghavinia, M. Kedia and M. Saliba, Energy Environ. Sci., 2021, 14, 5690–5722 RSC.
- A. Ng, Z. Ren, H. Hu, P. W. Fong, Q. Shen, S. H. Cheung, P. Qin, J. W. Lee, A. B. Djurišić and S. K. So, Adv. Mater., 2018, 30, 1804402 CrossRef PubMed.
- T. Bu, J. Li, H. Li, C. Tian, J. Su, G. Tong, L. K. Ono, C. Wang, Z. Lin and N. Chai, Science, 2021, 372, 1327–1332 CrossRef CAS.
- T. Bu, L. K. Ono, J. Li, J. Su, G. Tong, W. Zhang, Y. Liu, J. Zhang, J. Chang and S. Kazaoui, Nat. Energy, 2022, 7, 528–536 CrossRef CAS.
- Q. Zhang, G. Ma, K. A. Green, K. Gollinger, J. Moore, T. Demeritte, P. C. Ray, G. A. Hill Jr, X. Gu and S. E. Morgan, ACS Appl. Energy Mater., 2022, 5, 1487–1495 CrossRef CAS.
- D. Wang, J. Chen, P. Zhu, Y. Qiao, H. Hu, J. Zeng, J. Zhang, G. Qu, Y. Wang and X. Wang, Adv. Energy Mater., 2023, 13, 2203649 CrossRef CAS.
- J. Li, C. Jin, R. Jiang, J. Su, T. Tian, C. Yin, J. Meng, Z. Kou, S. Bai and P. Müller-Buschbaum, Nat. Energy, 2024, 1–11 Search PubMed.
- Y. Wang, Z. Zeng, Y. Zhang, Z. Zhang, L. Bi, A. He, Y. Cheng, A. K. Y. Jen, J. C. Ho and S. W. Tsang, Adv. Mater., 2024, 36, 2307635 CrossRef CAS.
- Y. Wang, Z. Zhang, Z. Zeng, Y. Zhang, T. Hu, L. Bi, Y. Yang, S. Ouyang, X. Hao and C. Xu, ACS Energy Lett., 2025, 10, 647–657 CrossRef CAS.
- N. Li, X. Niu, L. Li, H. Wang, Z. Huang, Y. Zhang, Y. Chen, X. Zhang, C. Zhu and H. Zai, Science, 2021, 373, 561–567 CrossRef CAS PubMed.
- M. Saliba, J.-P. Correa-Baena, C. M. Wolff, M. Stolterfoht, N. Phung, S. Albrecht, D. Neher and A. Abate, Chem. Mater., 2018, 30, 4193–4201 CrossRef CAS.
- B. Jo, W. Chen and H. S. Jung, J. Energy Chem., 2025, 101, 298–323 CrossRef CAS.
- Q. Zhang, H. Wang, Q. Zhao, A. Ullah, X. Zhong, Y. Wei, C. Zhang, R. Xu, S. De Wolf and K. Wang, ACS Energy Lett., 2024, 9, 5924–5934 CrossRef CAS.
- C. Zhi, S. Wang, S. Sun, C. Li, Z. Li, Z. Wan, H. Wang, Z. Li and Z. Liu, ACS Energy Lett., 2023, 8, 1424–1433 CrossRef CAS.
- J. Wu, L. Torresi, M. Hu, P. Reiser, J. Zhang, J. S. Rocha-Ortiz, L. Wang, Z. Xie, K. Zhang and B.-W. Park, Science, 2024, 386, 1256–1264 CrossRef CAS PubMed.
- J.-W. Lee, Z. Dai, C. Lee, H. M. Lee, T.-H. Han, N. De Marco, O. Lin, C. S. Choi, B. Dunn and J. Koh, J. Am. Chem. Soc., 2018, 140, 6317–6324 CrossRef CAS PubMed.
- M. Li, R. Sun, J. Chang, J. Dong, Q. Tian, H. Wang, Z. Li, P. Yang, H. Shi and C. Yang, Nat. Commun., 2023, 14, 573 CrossRef CAS PubMed.
- X. Yu, Z. Fang, S. Lin, S. Wu, M. Fang, H. Xie, D. Kong and C. Zhou, Small, 2024, 20, 2306101 CrossRef CAS PubMed.
- X. Zhao, R. Luo, C. Yu, X. Xu, W. Zhu, Y. Min and N. Cai, Adv. Funct. Mater., 2024, 34, 2307568 CrossRef CAS.
- Y. Zou, W. Yu, H. Guo, Q. Li, X. Li, L. Li, Y. Liu, H. Wang, Z. Tang and S. Yang, Science, 2024, 385, 161–167 CrossRef CAS PubMed.
- J. C. Hamill Jr, J. Schwartz and Y.-L. Loo, ACS Energy Lett., 2017, 3, 92–97 CrossRef.
- M. Lu, C. Shi, C. Ma, N. Li, L. Li and G. Xiao, J. Mater. Sci.: Mater. Electron., 2017, 28, 5603–5608 CrossRef CAS.
- A. Alberti, C. Bongiorno, E. Smecca, I. Deretzis, A. La Magna and C. Spinella, Nat. Commun., 2019, 10, 2196 CrossRef PubMed.
- A. He, Y. Wang, P. Tang, Z. Zhang, Z. Zeng, Y. Zhang, X. Hao, L. Wu, S. K. So and S. W. Tsang, Adv. Energy Mater., 2023, 13, 2300957 CrossRef CAS.
- L. Bi, Q. Fu, Z. Zeng, Y. Wang, F. R. Lin, Y. Cheng, H.-L. Yip, S. W. Tsang and A. K.-Y. Jen, J. Am. Chem. Soc., 2023, 145, 5920–5929 CrossRef CAS PubMed.
- V. D’Innocenzo, A. R. Srimath Kandada, M. De Bastiani, M. Gandini and A. Petrozza, J. Am. Chem. Soc., 2014, 136, 17730–17733 CrossRef.
- M. Shirayama, H. Kadowaki, T. Miyadera, T. Sugita, M. Tamakoshi, M. Kato, T. Fujiseki, D. Murata, S. Hara and T. N. Murakami, Phys. Rev. Appl., 2016, 5, 014012 CrossRef.
- Z. Zeng, Y. Wang, S. Ding, Y. Li, C. Xiang, C. S. Lee, Y. Cheng and S. W. Tsang, Adv. Funct. Mater., 2024, 2404255 Search PubMed.
- L. Yin, W. Huang, J. Fang, Z. Ding, C. Jin, Y. Du, L. Lang, T. Yang, S. Wang and W. Cai, Adv. Mater., 2023, 35, 2303384 CrossRef CAS PubMed.
- D. Bi, C. Yi, J. Luo, J.-D. Décoppet, F. Zhang, S. M. Zakeeruddin, X. Li, A. Hagfeldt and M. Grätzel, Nat. Energy, 2016, 1, 1–5 Search PubMed.
- K. L. Wang, Z. H. Su, Y. H. Lou, Q. Lv, J. Chen, Y. R. Shi, C. H. Chen, Y. H. Zhou, X. Y. Gao and Z. K. Wang, Adv. Energy Mater., 2022, 12, 2201274 CrossRef CAS.
Footnote |
| † These authors contributed equally: Yunfan Wang, Weiren Zhao, Juncheng Wang, and Zhuoqiong Zhang. |
| This journal is © The Royal Society of Chemistry 2026 |