An ‘ice-like’ water film for corrosion-proof seawater electrolysis
Zihui
Yuan
a,
Yingxia
Zhao
*d,
Yue
Sun
a,
Yingying
Xu
a,
Yuanhong
Zhong
 a,
Shaomin
Peng
a,
Shaomin
Peng
 a,
Ming
Sun
a,
Ming
Sun
 ace,
Ning
Yan
f,
Youwen
Liu
ace,
Ning
Yan
f,
Youwen
Liu
 *bg and
Lin
Yu
*bg and
Lin
Yu
 *ac
*ac
aGuangDong Engineering Technology Research Center of Modern Fine Chemical Engineering, School of Chemical Engineering and Light Industry, Guangdong University of Technology, Guangzhou, 510006, China. E-mail: gych@gdut.edu.cn
bState Key Laboratory of Materials Processing and Die & Mould Technology, and School of Materials Science and Engineering, Huazhong University of Science and Technology, Wuhan, Hubei 430074, China. E-mail: ywliu@hust.edu.cn
cJieyang Branch of Chemistry and Chemical Engineering Guangdong Laboratory (Rongjiang Laboratory), Jieyang, 515200, China
dAnalysis and Test Center Guangdong University of Technology, Guangdong University of Technology, Guangzhou, 510006, China. E-mail: yxzhao@gdut.edu.cn
eGuangdong Yuntao Hydrogen Energy Technology Co., Ltd, Guangzhou, 551040, China
fKey Laboratory of Artificial Micro, and Nano-structures of Ministry of Education, School of Physics and Technology, Wuhan University, Wuhan 430072, China
gResearch Institute of Huazhong University of Science and Technology in Shenzhen, Shenzhen, 518057, P. R. China
First published on 5th March 2026
Abstract
Renewable-powered seawater electrolysis for green hydrogen is hindered by chloride corrosion. We introduce a protective interfacial water film strategy, where a highly ordered hydrogen-bonded network in the outer Helmholtz plane (OHP) leverages strong covalent O–H bonds and dense ice-like ordering to block Cl− ingress. This engineered barrier within the electric double layer prevents chloride access to the inner Helmholtz plane (IHP), eliminating corrosive complexation. The film is realized via dynamic phosphorus migration during the electrochemical reconfiguration of a phosphate-doped cobalt–nickel–iron layered hydroxide (CNFPO), enriching phosphate species that electrostatically template interfacial water molecules. Combined in situ Raman spectroscopy (using D2O) and molecular dynamics reveal the enrichment of tetrahedral hydrogen-bonded water in the OHP, forming the Cl−-repelling barrier while the participation of free water in the reaction pathway is further verified via H2O/D2O kinetic isotope effect (KIE) experiments. Protected by this film, CNFPO achieves an overpotential of 24 mV at 10 mA cm−2, >1200 h stability, and 76-fold current density enhancement in saturated saline. An anion-exchange membrane electrolyzer operates for >1000 h at 500 mA cm−2 and 60 °C, validating practical viability.
Broader contextSeawater electrolysis is critical to scalable green hydrogen production, yet its development has been severely hindered by chloride-induced corrosion. Here, we introduce a fundamentally new protection strategy by constructing an ordered, ‘ice-like’ water film at the electrode interface. Structured via in situ phosphate enrichment, this film leverages a dense hydrogen-bonded network to physically block chloride ions from reaching and corroding the cathode catalyst. This approach directly circumvents the thermodynamic driving force for chloride–metal complexation that limits conventional passivation methods. As a result, it enables an exceptional combination of high hydrogen evolution activity, long-term stability (>1000 h) in alkaline real seawater, and industrially relevant performance – offering a practical pathway toward durable, corrosion-free hydrogen production directly from seawater. |
Introduction
Renewable energy-powered water electrolysis is hailed as a cornerstone approach for realizing profound decarbonization within the industrial domain.1–3 However, the widespread implementation of this technology faces significant obstacles due to the geographical discordance between freshwater reserves and renewable energy sources like offshore wind farms and coastal photovoltaic systems.4–6 This pronounced disparity renders direct seawater electrolysis an indispensable and inevitable pathway for achieving scalable and sustainable green hydrogen production.7 Pertaining to seawater electrolysis, chloride ions (Cl−) at high concentrations (ionic radius ∼0.181 nm),8 leveraging their elevated diffusion coefficient (D0 ≈ 2.03 × 10−9 m2 s−1 at 25 °C), exhibit a pronounced ability to permeate the catalyst's passivation layer (typically an amorphous metal oxide 5–20 nm in depth), an infiltration chiefly governed by mechanisms such as grain boundary diffusion and migration through defect channels.9,10 The inherent strength of metal–Cl− coordination bonds facilitates the reaction of Cl− with constituent metal cations (e.g., Fe2+ and Ni3+) within the passivation film, yielding soluble chloride complexes such as [FeCl4]2− and [NiCl6]3−. This process, in turn, instigates local lattice distortions and promotes the selective dissolution of the said protective layer.9,11 Subsequently, the emergence of pitting with dimensions ranging from micron to submicron at the metal/electrolyte interface markedly intensifies corrosive degradation and critically impairs electrolytic performance.Extensive research has been undertaken to elucidate the potent corrosive mechanisms of Cl−. Prominent theoretical constructs, encompassing point defect models, void models, and adsorption mechanisms, consistently attribute this corrosive action to the structural degradation of the passivation film.12–14 Illustratively, Hoar15et al. posited that while the passivation film effectively shields the underlying metal from the corrosive environment, Cl− ions, by virtue of their pronounced coordination affinity, can displace oxygen within this protective layer. Such displacement perturbs the native metal–oxygen bonding framework, precipitating the formation of soluble chloride complexes and thereby compromising the film's protective integrity. Conventional approaches to mitigate Cl−-induced corrosion, primarily by forming robust passivation films,16–18 have demonstrated limited efficacy, merely retarding the corrosion kinetics without addressing the core thermodynamic challenge: the facile, energetically favorable (ΔG < 0) complexation of Cl− with metal cations, which supersedes oxygen binding.19–21 Therefore, there is an urgent need for innovative strategies that break through the thermodynamic limitations, unlocking significant improvements in the stability of seawater electrolysis.
Herein, we introduce a strategy replacing traditional passivation with a structured interfacial water layer. Leveraging strong covalent O–H bonds (∼460 kJ mol−1) and ice-like ordering to block Cl− penetration, we engineer a hydrogen-bonded water film within the outer Helmholtz plane (OHP) as a robust Cl− barrier. We implement this using an amorphous phosphate-doped cobalt–nickel–iron layered hydroxide, where in situ electrochemical reconfiguration drives phosphate migration. These high-charge-density species order interfacial water, forming the protective hydrogen-bonded film, confirmed by in situ Raman (using D2O) and electrochemical analysis. This film effectively prevents Cl− ingress into the inner Helmholtz plane (IHP), mitigating corrosion. Furthermore, the participation of free water in the reaction pathway was further verified via H2O/D2O kinetic isotope effect (KIE) experiments. Consequently, the catalyst achieves exceptional hydrogen evolution reaction (HER) performance: low overpotentials of 243, 24, 22, and 11 mV at 10 mA cm−2 in neutral real seawater, alkaline real seawater, artificial seawater, and 1 M KOH, respectively. It demonstrates >1200 h stability at 50 mA cm−2 in simulated seawater. Integrated into an anion exchange membrane (AEM) electrolyzer, stable hydrogen production is maintained for >1000 h under industrially relevant conditions (500 mA cm−2, 60 °C), showcasing significant potential for practical seawater electrolysis.
Results and discussion
Mitigating cathodic corrosion in seawater electrolysis: a chloride-repelling hydrogen-bonded water film strategy
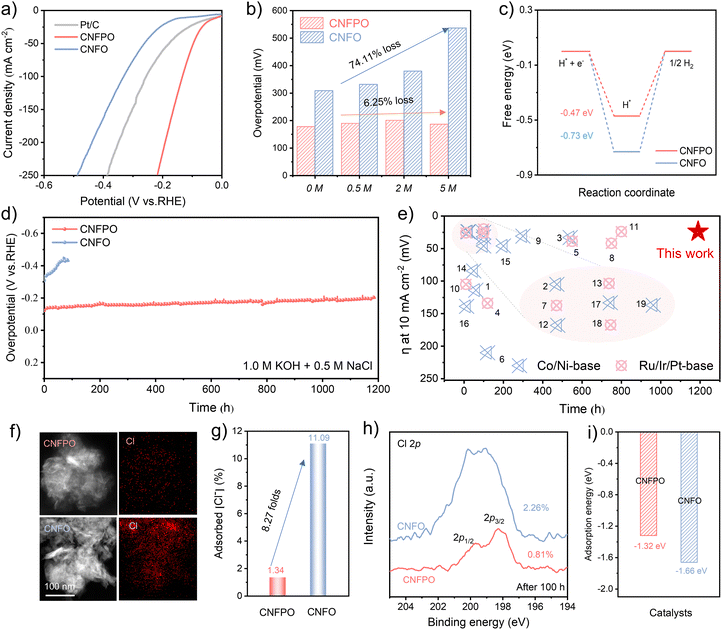
During seawater electrolysis, pitting corrosion is conventionally regarded as predominantly affecting the anode, particularly metal-based anodes.22,23 However, the detrimental effects of Cl− on the cathode remain a significantly under-explored research area.3,24 To systematically investigate this issue, we performed 1000 cycles of cyclic voltammetry (CV) tests on Ni-based Ni(OH)2 electrodes in simulated seawater. The results clearly revealed a significant current density degradation of 63.2% under Cl− exposure (Fig. 1a and Fig. S1), while post-test scanning electron microscopy (SEM) images showed characteristic corrosion pit formation (Fig. 1b and Fig. S2). In stark contrast, the current degradation in a Cl−-free electrolyte was substantially smaller, demonstrating the excellent intrinsic stability of the cathode material in the absence of chloride ions. This evidence compellingly demonstrates that Cl− readily adsorbs onto the cathode surface, leading to both corrosion and catalyst deactivation. Consequently, the Cl−-induced degradation of cathode performance is a critical factor that cannot be neglected during seawater electrolysis. Theoretical investigations spanning decades have established mechanistic models of Cl−-mediated pitting corrosion, emphasizing its characteristic of localized vertical penetration.25 Driven by their intrinsic high mobility and strong concentration-gradient diffusion,6 Cl− first breaches the interfacial electric double-layer capacitor (EDLC) to adsorb onto the Ni(OH)2 surface. This adsorption initiates localized chemical reactions, ultimately enabling the ions to overcome diffusion barriers, infiltrate the crystal lattice, and propagate corrosion deep into the material, thereby carving out deep pores (Fig. 1c). Hence, any viable corrosion protection strategy must not only inhibit surface-level Cl− adsorption but must also disrupt its penetration pathway into the active catalyst lattice. To combat this dual challenge, we have engineered an ordered structure of interfacial water molecules within the EDL. Unlike passive oxide layers vulnerable to Cl− coordination, the water-based barrier leverages two synergistic attributes: the formidable strength of covalent O–H bonds and the entropic stability conferred by dense, ice-like hydrogen-bond ordering. This engineered water layer strategically modulates the local hydrogen-bonded network, creating a robust barrier that effectively suppresses Cl−-induced corrosion.26,27 As illustrated in Fig. 1d, the EDL comprises a diffuse layer and a compact layer (CL), with the CL further subdivided into the outer Helmholtz plane (OHP) and the inner Helmholtz plane (IHP).18 In HER systems, steric constraints naturally confine free H2O molecules to the IHP, while solvated Cl− preferentially accumulate in the OHP.28 Consequently, the crux of achieving corrosion resistance lies in impeding the migration of Cl− from the OHP to the IHP. Our strategy addresses this by constructing a highly ordered, hydrogen-bonded water film within the OHP. This film resists the nucleophilic attack or substitution by Cl− through the strength of covalent O–H bonds and forms a physical barrier via the dense packing of ice-like structured water, effectively blocking the migration path of Cl− to the IHP and thus preventing its corrosive adsorption at the source. To computationally validate our proposed strategy, we conducted classical molecular dynamics (MD) simulations. These simulations were designed to probe the correlation between interfacial H2O and Cl− adsorption by systematically varying the water coverage on a Ni(OH)2 model catalyst and analyzing the resultant surface distributions of Cl− and H2O.29 As depicted in Fig. 1e and f, upon increasing the water concentration from 1 M to 10 M, the concentration of Cl− within 10 Å of the Ni(OH)2 surface (IHP) exhibits a pronounced reduction. This phenomenon is attributed to the enrichment of interfacial water on the catalyst surface. The increased interfacial water content enhances the long-range interactions among water molecules, which in turn attenuate the electron density around protons and ultimately promote the transformation of isolated water molecules into an interconnected, more extensive water network.30 By diminishing the adsorption affinity of Cl− on the oxide surface, the reinforced hydrogen-bonded network in the OHP constructs an effective barrier to ion migration, which prevents Cl− penetration through the EDL and thus endows the catalyst with remarkable resistance to chlorine corrosion in alkaline seawater.Chlorine corrosion-resistance of the CNFPO catalyst
Guided by the insights from our MD simulations, we rationally designed and synthesized a trimetallic Co/Ni/Fe-based catalyst system. Specifically, the phosphate-doped cobalt–nickel–iron layered oxide/hydroxide (denoted as CNFPO) was rapidly fabricated via an in situ electrochemical deposition method (Fig. S3), while the corresponding cobalt–nickel–iron layered oxide/hydroxide (denoted as CNFO) was prepared for comparison using a similar approach. SEM images and transmission electron microscopy (TEM) observations reveal that both catalysts exhibit a distinctive nanosheet morphology (Fig. S4). Through comprehensive characterization, including high-resolution transmission electron microscopy (HRTEM) images, elemental mapping, X-ray photoelectron spectroscopy (XPS), Raman spectroscopy, and inductively coupled plasma-optical emission spectrometry (ICP-OES) (Fig. S5–S9 and Table S1), we have successfully validated the incorporation of phosphorus. This doping process has induced significant electron transfer and redistribution within the catalyst,31–34 laying a crucial foundation for subsequent performance optimization. In electrochemical tests, CNFPO exhibits outstanding hydrogen evolution performance in alkaline simulated seawater, natural seawater, and neutral natural seawater environments (Fig. 2a and Fig. S10–S14), with remarkably low overpotentials of 11, 22, 24, and 243 mV at 10 mA cm−2, further confirming the catalyst's potential in direct seawater electrolysis. Density functional theory (DFT) further confirms its intrinsic activity by revealing a strong H2O adsorption energy (−2.08 eV) and an optimal Gibbs free energy (ΔGH* = −0.47 eV), corroborating rapid reaction kinetics for water dissociation in alkaline seawater (Fig. 2c and Fig. S15 and S17).35,36 Given that Cl− accumulation during seawater electrolysis can trigger catastrophic catalyst degradation through irreversible corrosion,37 a definitive assessment of CNFPO's robustness against high salinity is imperative. We began this evaluation by systematically charting the catalyst's electrochemical behavior in an electrolytic system with NaCl concentrations escalating from 0 M to saturation. The electrochemical tests showed (Fig. S18) that the overpotential of CNFPO increased by only about 5 mV when the NaCl concentration was elevated from 0 M to 5 M (Fig. 2b), exhibiting only 1% decay, underscoring its exceptional salinity tolerance. In stark contrast, the undoped CNFO catalyst suffered significant performance decay under the same conditions, highlighting the detrimental effect of Cl− on conventional catalytic systems.11,38 Long-term durability was confirmed by a 1200 h chronoamperometry test at 50 mA cm−2 (Fig. 2d) and stepwise current testing at 100, 500, and 1000 mA cm−2 over 250 h (Fig. S19). The CNFPO catalyst demonstrated remarkable operational stability, preserving both its nanosheet morphology and high performance without any observable Cl−-induced pitting. This stands in stark contrast to the severe degradation exhibited by the CNFO control sample (Fig. S20), unequivocally validating CNFPO's exceptional resistance to chlorine corrosion. Compared to recently reported seawater HER catalysts (Fig. 2e and Tables S2 and S3), the CNFPO catalyst exhibits both exceptional activity and corrosion resistance. Post-test analyses provided deeper insights into this corrosion resistance. For instance, the elemental mapping images revealed that the amount of Cl− adsorbed on the CNFPO electrode (1.34%) was significantly less than that on the CNFO electrode (11.09%) after testing (Fig. 2f and g). The XPS Cl 2p spectra further support this conclusion, indicating much lower Cl− adsorption on CNFPO even after immersion in the electrolyte (Fig. 2h). Complementing this, DFT calculations yielded a Cl− adsorption energy of −1.32 eV for CNFPO, underscoring its superior ability to inhibit Cl− adsorption (Fig. 2i).39 Collectively, these results affirm the feasibility of our protective interfacial water film strategy. As evidenced by the results, the CNFPO catalyst possesses outstanding chlorine corrosion-resistance and long-term operational stability.Corrosion-resistance mechanism insight: dynamic evolution of interfacial water molecules
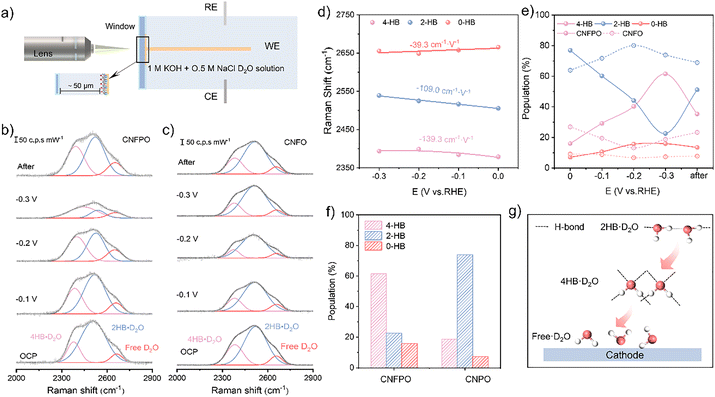
To elucidate the molecular-level mechanism underpinning the catalyst's chlorine corrosion-resistance, we employed operando Raman spectroscopy to investigate the structure of the interfacial water network, utilizing D2O as an isotopic probe in the electrolyte (Fig. 3a). As illustrated in Fig. 3b and c, a broad band appears between 2200 and 2700 cm−1, attributed to the O–D stretching mode of D2O. This band can be deconvoluted into three Gaussian peaks, each representing a distinct type of O–D stretching vibration.40–42 At open-circuit potential (OCP), the intensity of all three D2O-related peaks on CNFPO was markedly higher than those on CNFO, indicating a more pronounced enrichment of D2O at the interface. This observation substantiates the presence of an interfacial water layer on CNFPO. Through a meticulous analysis of the relative contributions of the three D2O species within this interfacial water, we have gained profound insights into the chlorine resistance of the interface water layer. Under HER circumstances (Fig. 3e and f), the percentage of 4-coordinated hydrogen-bonded D2O (4HB·D2O) on CNFPO significantly increased from 18.3% to 67%, signifying a more ordered arrangement of interfacial water molecules. This transformation indicates the formation of a densely packed, ice-like hydrogen-bonded network, which mimics the crystalline lattice of hexagonal ice and serves as an effective physical barrier against chloride ion intrusion. This dense ice-like ordering hydrogen-bonded water film is facilitated by enhanced intermolecular hydrogen bonding and is known in corrosion science to exhibit strong repulsion against Cl− ions.43,44 This was further corroborated by electrochemical corrosion tests. Unlike the symmetric 4HB·D2O, weakly hydrogen-bonded water molecules reside closer to the electrode surface,45 with the percentage of free D2O increasing to over 15.7%. This structure ensures an ample supply of free H2O within the IHP, which is crucial for continuously feeding reactants to the catalytic active sites (Fig. 3g). The presence of free water in the IHP was confirmed through Tafel plots and H2O/D2O isotope substitution experiments (Fig. S21 and S22), showing that CNFPO exhibits a pronounced KIE and enhanced HER activity in D2O, whereas CNFO demonstrates a weaker isotope effect; this directly confirms our conclusion that “an accumulation of free water within the IHP and governs enhanced HER kinetics of CNFPO.” In contrast, the interfacial water structure at the CNFO interface evolves oppositely. Moreover, due to the electrochemical Stark effect,42,46 the vibrational frequency of 4HB·D2O varies with the applied potential. The steeper Stark slope (Fig. 3d) indicates that the 4HB·D2O on the CNFPO catalyst surface is more sensitive to the applied potential.47 This suggests that manipulating the local electric field on the CNFPO surface facilitates the formation of 4HB·D2O, creating a highly ordered hydrogen-bonded water film that shields the catalyst from corrosion during seawater electrolysis. Notably, after the removal of the applied potential, the D2O peak intensity and species distribution on CNFPO rapidly recovered, further demonstrating that the formation of the hydrogen-bonded water film is dependent on the electric field. Interestingly, unlike CNFO, the interfacial water structure on CNFPO did not completely revert to its initial state after potential removal. This implies that, in addition to the external electric field, phosphate also participates in shaping the Cl−-resistant hydrogen-bonded water film during the HER process. To rigorously validate the reproducibility of this key finding, we performed control experiments using pristine Ni foam as a reference and conducted repeated in situ Raman spectroscopy measurements in D2O on two independently prepared CNFPO samples (Fig. S23–S25). The results were highly consistent: all samples exhibited the same spectral evolution trend under polarization. Through detailed in situ Raman spectroscopy (employing D2O) exploration experiments, spectroscopic observation unveils that the presence of 4HB·D2O within interfacial water molecules is crucial in conferring resistance to chlorine corrosion.Operational phosphate migration in CNFPO facilitates the formation of chloride-repellent hydrogen-bonded water films
While the chlorine resistance of the hydrogen-bonded water film is evident, a crucial question arises: how does the phosphate within the CNFPO dynamically shape this interface during high-current seawater electrolysis? Initial insights into this process emerged from post-HER EDS mapping (Fig. S26 and Tables S4 and S5). The analysis revealed a significant depletion of phosphorus from the bulk, coupled with its re-emergence as a surface-concentrated species. This leads us to hypothesize a dynamic leaching/readsorption mechanism, whereby phosphorus first escapes the catalyst matrix and is then readsorbed on the resultant metal hydroxide surface as phosphate. In the in situ Raman spectra, as shown in Fig. 4a and Fig. S27 and S28, the migration of anions and the reduction of metal cations occur simultaneously.48–50 As the applied potential increased positively, a broad phosphate peak emerged at 900–1100 cm−1 in Raman spectra, confirming the migration behaviour of the phosphate group.51 To further investigate the surface chemistry post-reaction, time-of-flight secondary ion mass spectrometry (TOF-SIMS) was employed (Fig. S29). The analysis revealed prominent signals corresponding to OH− (m/z = 17) and various PO43− fragments (m/z = 47, 63, 79, and 95) on the catalyst surface (Fig. 4b and Fig. S30). This finding was corroborated by post-reaction XPS spectra, where the P 2p spectra showed a significant attenuation of M–P bonds concurrent with the emergence of a distinct PO43− peak (Fig. 4c and Fig. S31). Collectively, these results indicate the dynamic migration of phosphate species and their subsequent readsorption onto the surface of the resultant metal hydroxides. Consequently, these surface-readsorbed phosphate groups, possessing a high negative charge density, are poised to generate a strong local electric field within the aqueous electrolyte.52,53 The differential charge density distribution based on DFT calculations shows the charge accumulation of readsorbed phosphate radicals on oxygen atoms, suggesting that the phosphate groups act as charged bodies on the catalyst surface to induce an envisaged electric field (Fig. 4d and Fig. S32).54,55 This view is further supported by the reduced surface work function observed in the UPS spectra (Fig. 4e, and f and Fig. S33). We then used MD simulations to demonstrate how this field reconstructs the interface (Fig. 4g). Simulating the ionic distribution of the two sets of catalysts in 0.5 M NaCl + 1 M KOH solution, the spontaneous structural optimization by MD simulations resulted in a peak concentration of phosphate groups within 5 Å on the surface of the CNFPO catalyst (Fig. S34), whose oxygen atoms acted as strong hydrogen-bonding acceptors to form a high-density hydrogen-bonded network with water molecules (per PO43− average 2.8 hydrogen-bonds), contributing to the maximum ordering of the interfacial water film at 8 Å (Fig. 4h). This highly ordered hydrogen-bonded water film effectively repels Cl− to the diffusion layer 10 Å away. Conversely, the CNFO catalyst surfaces displayed disordered ion distributions without structured water layers, enabling competitive Cl− adsorption at active sites. Theoretical calculations validated our initial hypothesis, these findings provide a clear, molecular-level picture: the in situ formation of a surface phosphate layer enhances both local and long-range hydrogen bonding among water molecules during electrolysis by simultaneously acting as a hydrogen-bonded acceptor and providing electrostatic field guidance, thereby promoting the formation of an extensive, highly ordered hydrogen-bonded water film, which is the cornerstone of the exceptional chloride resistance in seawater electrolysis.To ensure that this strategy can be applied for long-term stable hydrogen production from seawater under practical operating conditions, we further verified that the protective effect of the hydrogen-bonded water film is not transient but sustained. The long-term stability of the CNFPO catalyst is governed by a continuous and dynamic interfacial reconstruction, which perpetually maintains its protective water layer. The Raman spectra of samples after reactions at multiple time points provide evidence for this. The 1000–1100 cm−1 (PO43−) characteristic peak in CNFPO remains stable after prolonged reactions (Fig. S35). ICP-MS quantitative results (Fig. S36) show that the phosphate content remains stable throughout the reaction, indicating that a dynamic equilibrium between dissolution and re-adsorption is maintained, ensuring the continuous presence of interfacial phosphate. and critically, the catalyst fully retains its ability to dynamically restructure interfacial water under an applied potential (Fig. 4i). The proportions of structured (4HB·H2O, surging to 67%) and free H2O (>15.7%) evolved in a near-perfect echo of the pristine sample's behavior (Fig. 4j). The ability of the aged catalyst to perfectly replicate the dynamic evolution of its interfacial water confirms that phosphate migration is a sustained, self-regulating process. It is this perpetual modulation of the interface that underpins the catalyst's exceptional resilience to Cl−-induced corrosion.
Operational validation of chloride resistance in an AEM electrolyzer
We propose a hydrogen-bonded water film to inhibit Cl− corrosion, which greatly enhanced the seawater stability of the catalysts by introducing a local force field modulated by dynamically migrating phosphate groups into the hydrogen-bonded water film, thereby preventing the strong complexation of Cl−with the active metal that instigates corrosion (Fig. 5a). It is noteworthy that simply adding phosphate or sulfate ions to the electrolyte does not replicate this effect (Fig. S37 and S38). This strongly suggests that only through interface reconstruction and electric-field-driven dynamic phosphate migration can the interfacial water network be continuously regulated during the reaction, thereby stably forming the ice-like water film structure. To further validate the industrial applicability of this strategy, we assembled a flow-type AEM electrolyzer with a 9 cm2 geometric area using CNFPO as the cathode catalyst (Fig. 5b) and CoFe-LDH as the anode. The anion exchange electronegativity (AEM) electrolyzer was operated at 25 °C, circulating an alkaline simulated seawater electrolyte and natural seawater via a peristaltic pump. As shown in Fig. 5c and Fig. S40, the CNFPO-based cell achieved an industrially relevant current density of 2 A cm−2 at a cell voltage of only 2.01 V, outperforming the benchmark Pt/C cathode under identical conditions. The overall electrolyzer exhibited an average efficiency of 67% across various current densities with low energy consumption (Fig. 5e). At 1 A cm−2, the cathodic energy efficiency reached 61.4%, requiring only 4.84 kWh of electricity to produce one cubic meter of hydrogen. This corresponds to a hydrogen production cost of just $2.23 per kg, which is competitive with grey hydrogen and aligns with the 2030 global target range of $2.0–2.5 per kg of H2 (Fig. S39 and Table S6).56 Furthermore, long-term durability was assessed under industrially relevant conditions (0.5 A cm−2 at 60 °C). The CNFPO‖CoFe-LDH cell maintained stable operation over 1000 hours without a noticeable voltage increase (Fig. 5d). Even after 1000 hours of continuous membrane electrode operation, the nickel foam substrate maintains its good morphology (Fig. S41), with only a small amount of chloride ions adhering and no significant corrosion observed. In contrast, the Pt/C cathode exhibited significant degradation, with a decay rate of 2.16 mV h−1. Compared to other cathodes reported for seawater electrolysis, CNFPO demonstrates superior performance metrics (Table S7). Collectively, these findings underscore the practical potential of CNFPO catalysts for future large-scale seawater hydrogen production. It paves the way for the large-scale, economically sound production of green hydrogen directly from the ocean.Conclusions
In this work, we propose a hydrogen-bonded water film that inhibits Cl− corrosion, in which the chloride adsorption corrosion is successfully prevented by introducing a local force-field-regulated hydrogen-bonded water film through the dynamic migration of phosphate groups into the CNFPO catalyst. In situ Raman spectroscopy (employing D2O) combined with classical MD simulations revealed the dominant role of an interfacial hydrogen-bonded water film in the seawater HER against Cl−. A highly ordered hydrogen-bonded network in the OHP leverages strong covalent O–H bonds and dense ice-like ordering to achieve the preferential adsorption of water molecules as well as repulsion of Cl−. Furthermore, the participation of free water in the reaction pathway was further verified via H2O/D2O kinetic isotope effect experiments. The CNFPO catalyst delivered exceptional activity and stability under industrially relevant high-current-density seawater electrolysis, with the AEM electrolyzer assembled from this catalyst operating stably for more than 1000 hours at 500 mA cm−2, and 60 °C. This strategy provides a new microscopic perspective for the design of the catalytic field for hydrogen production from seawater under working conditions, and substantially improves the long-term stability of the HER with high-current seawater. Beyond introducing a high-performance alkaline seawater HER electrocatalyst, this work fundamentally combines the dynamic migration of catalyst anions with the dynamic evolution of interfacial water molecules and seawater corrosion protection mechanism, providing critical insights for practical seawater electrolysis cathode development.Author contributions
Y. W. L. and L. Y. conceived and directed the project. Y. W. L., L. Y., X. Y. Z., and H. Z. Y. designed the experiments. H. Z. Y. and Y. S. carried out the experiments. Y. Y. X., X. Y. Z., M. S. P., and M. S. helped with the anion exchange membrane test. X. Y. Z, N. Y., Y. L., and L. Y. revised the paper. All authors discussed the results and assisted during manuscript preparation.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting this study's findings are available from the corresponding author upon reasonable request.The data supporting this article have been included as part of the supplementary information (SI). The SI contains additional electrochemical test data, material characterization, and supporting figures. See DOI: https://doi.org/10.1039/d6ee00098c.
Acknowledgements
This work was financially supported by the Foundation of Basic and Applied Basic Research of Guangdong Province (2023B1515120043), the Natural Science Foundation of Guangdong Province (2025B1515020079), the Smart Medical Innovation Technology Center, GDUT (Project Number: ZYZX24-031), and the Science and Technology Program of Guangzhou (Grant No: 2025A04J3491). The authors acknowledge the technical support from the Analysis and Test Center of the Guangdong University of Technology.Notes and references
- S. Chu and A. Majumdar, Nature, 2012, 488, 294–303 CrossRef CAS PubMed.
- D. Guan, B. Wang, J. Zhang, R. Shi, K. Jiao, L. Li, Y. Wang, B. Xie, Q. Zhang, J. Yu, Y. Zhu, Z. Shao and M. Ni, Energy Environ. Sci., 2023, 16, 4926–4943 RSC.
- Q. Sha, S. Wang, L. Yan, Y. Feng, Z. Zhang, S. Li, X. Guo, T. Li, H. Li, Z. Zhuang, D. Zhou, B. Liu and X. Sun, Nature, 2025, 639, 360–367 CrossRef CAS PubMed.
- H. Xie, Z. Zhao, T. Liu, Y. Wu, C. Lan, W. Jiang, L. Zhu, Y. Wang, D. Yang and Z. Shao, Nature, 2022, 612, 673–678 CrossRef CAS PubMed.
- Y. Gu, N. Nie, J. Liu, Y. Yang, L. Zhao, Z. Lv, Q. Zhang and J. Lai, EcoEnergy, 2023, 1, 405–413 CrossRef.
- J. Guo, Y. Zheng, Z. Hu, C. Zheng, J. Mao, K. Du, M. Jaroniec, S.-Z. Qiao and T. Ling, Nat. Energy, 2023, 8, 264–272 CAS.
- M. A. Khan, T. Al-Attas, S. Roy, M. M. Rahman, N. Ghaffour, V. Thangadurai, S. Larter, J. Hu, P. M. Ajayan and M. G. Kibria, Energy Environ. Sci., 2021, 14, 4831–4839 RSC.
- W. Tong, M. Forster, F. Dionigi, S. Dresp, R. Sadeghi Erami, P. Strasser, A. J. Cowan and P. Farràs, Nat. Energy, 2020, 5, 367–377 CrossRef CAS.
- M. J. Graham, J. A. Bardwell, G. I. Sproule, D. F. Mitchell and B. R. MacDougall, Corros. Sci., 1993, 35, 13–18 CrossRef CAS.
- Y. Yang, M. Zha, D. Wang, T. Wang, Y. Wang, C. Wang, Y. Yuan and H.-Y. Wang, Adv. Mater., 2024, 36, 2409877 CrossRef CAS PubMed.
- W. Xu, Z. Wang, P. Liu, X. Tang, S. Zhang, H. Chen, Q. Yang, X. Chen, Z. Tian, S. Dai, L. Chen and Z. Lu, Adv. Mater., 2024, 36, 2306062 CrossRef CAS PubMed.
- E. Doná, M. Cordin, C. Deisl, E. Bertel, C. Franchini, R. Zucca and J. Redinger, J. Am. Chem. Soc., 2009, 131, 2827–2829 CrossRef PubMed.
- S. Zhang, Y. Wang, S. Li, Z. Wang, H. Chen, L. Yi, X. Chen, Q. Yang, W. Xu, A. Wang and Z. Lu, Nat. Commun., 2023, 14, 4822 CrossRef CAS PubMed.
- P. Li, S. Wang, I. A. Samo, X. Zhang, Z. Wang, C. Wang, Y. Li, Y. Du, Y. Zhong, C. Cheng, W. Xu, X. Liu, Y. Kuang, Z. Lu and X. Sun, Research, 2020, 2020, 2872141 CAS.
- T. P. Hoar, D. C. Mears and G. P. Rothwell, Corros. Sci., 1965, 5, 279–289 CrossRef CAS.
- K. W. Leong, W. Pan, Y. Wang, S. Luo, X. Zhao and D. Y. C. Leung, ACS Energy Lett., 2022, 7, 2657–2666 CrossRef CAS.
- K. Yu, S. Feng, C. Ding, M. Gu, P. Yu and M. Huang, Mater. Today, 2023, 70, 8–16 CrossRef CAS.
- L. Suo, O. Borodin, T. Gao, M. Olguin, J. Ho, X. Fan, C. Luo, C. Wang and K. Xu, Science, 2015, 350, 938–943 CrossRef CAS PubMed.
- C. Cai, K. Liu, L. Zhang, F. Li, Y. Tan, P. Li, Y. Wang, M. Wang, Z. Feng, D. Motta Meira, W. Qu, A. Stefancu, W. Li, H. Li, J. Fu, H. Wang, D. Zhang, E. Cortés and M. Liu, Angew. Chem., Int. Ed., 2023, 62, e202300873 CrossRef CAS PubMed.
- R. J. Gomes, R. Kumar, H. Fejzić, B. Sarkar, I. Roy and C. V. Amanchukwu, Nat. Catal., 2024, 7, 689–701 CrossRef CAS.
- X. Xu, Y. Lu, J. Shi, X. Hao, Z. Ma, K. Yang, T. Zhang, C. Li, D. Zhang, X. Huang and Y. He, Nat. Commun., 2023, 14, 7708 CrossRef CAS PubMed.
- H. Zou, X. Meng, X. Zhao and J. Qiu, Adv. Mater., 2023, 35, 2207262 CrossRef CAS PubMed.
- J. Huang, B. Hu, J. Meng, T. Meng, W. Liu, Y. Guan, L. Jin and X. Zhang, Energy Environ. Sci., 2024, 17, 1007–1045 RSC.
- M. Chen, N. Kitiphatpiboon, C. Feng, A. Abudula, Y. Ma and G. Guan, eScience, 2023, 3, 100111 CrossRef.
- C. Zhang, G. Yang, X. Gao, Z. Li, Y. Li, S. Wang, J. Wang, X. Zhao, K. Zhang, Y. Kang and Y. Cui, Appl. Catal., B, 2025, 377, 125478 CrossRef CAS.
- Z. Zhao, H. Liu, W. Gao, W. Xue, Z. Liu, J. Huang, X. Pan and Y. Huang, J. Am. Chem. Soc., 2018, 140, 9046–9050 CrossRef CAS PubMed.
- X. Yuan, D. Cheng, B. Liu, K. Liang, K. Liang, J. Yu, M. Mecklenburg, P. Sautet and Y. Li, Joule, 2024, 8, 3038–3053 CrossRef CAS.
- X. Chen, L. Chen, C. Chen, D. Shi, J. Song, Y. Qin, X. Wang, M. M. Amjad, D. Sun, B. Sun and K. Zhang, Adv. Mater., 2024, 36, 2401110 CrossRef CAS PubMed.
- S. Li, L. Wu, Q. Liu, M. Zhu, Z. Li, C. Wang, X. Jiang and J. Li, J. Am. Chem. Soc., 2023, 145, 26711–26719 CrossRef CAS PubMed.
- L. Cao, Q. Luo, W. Liu, Y. Lin, X. Liu, Y. Cao, W. Zhang, Y. Wu, J. Yang, T. Yao and S. Wei, Nat. Catal., 2019, 2, 134–141 CrossRef CAS.
- S. Qian, F. Xu, Y. Fan, N. Cheng, H. Xue, Y. Yuan, R. Gautier, T. Jiang and J. Tian, Nat. Commun., 2024, 15, 2774 CrossRef CAS PubMed.
- Y. Zhou, R. Lu, X. Tao, Z. Qiu, G. Chen, J. Yang, Y. Zhao, X. Feng and K. Müllen, J. Am. Chem. Soc., 2023, 145, 3647–3655 CrossRef CAS PubMed.
- L. Peng, N. Yang, Y. Yang, Q. Wang, X. Xie, D. Sun-Waterhouse, L. Shang, T. Zhang and G. I. N. Waterhouse, Angew. Chem., Int. Ed., 2021, 60, 24612–24619 CrossRef CAS PubMed.
- H. Jin, J. Xu, H. Liu, H. Sheng, H. Yu, M. Jaroniec, Z. Yao and S.-Z. Qiao, Sci. Adv., 2023, 9, eadi7755 CrossRef CAS PubMed.
- L. Guo, Z. Zhang, Z. Mu, P. Da, L. An, W. Shen, Y. Hou, P. Xi and C.-H. Yan, Adv. Mater., 2024, 36, 2406682 CrossRef CAS PubMed.
- X. Duan, Q. Sha, P. Li, T. Li, G. Yang, W. Liu, E. Yu, D. Zhou, J. Fang, W. Chen, Y. Chen, L. Zheng, J. Liao, Z. Wang, Y. Li, H. Yang, G. Zhang, Z. Zhuang, S.-F. Hung, C. Jing, J. Luo, L. Bai, J. Dong, H. Xiao, W. Liu, Y. Kuang, B. Liu and X. Sun, Nat. Commun., 2024, 15, 1973 CrossRef CAS PubMed.
- J. Chang and S. Lu, Matter, 2024, 7, 2640–2642 CrossRef CAS.
- Q. Wen, J. Duan, W. Wang, D. Huang, Y. Liu, Y. Shi, J. Fang, A. Nie, H. Li and T. Zhai, Angew. Chem., Int. Ed., 2022, 61, e202206077 CrossRef CAS PubMed.
- Y.-H. Wang, S. Zheng, W.-M. Yang, R.-Y. Zhou, Q.-F. He, P. Radjenovic, J.-C. Dong, S. Li, J. Zheng, Z.-L. Yang, G. Attard, F. Pan, Z.-Q. Tian and J.-F. Li, Nature, 2021, 600, 81–85 CrossRef CAS PubMed.
- A. Groß and S. Sakong, Chem. Rev., 2022, 122, 10746–10776 CrossRef PubMed.
- R. Subbaraman, D. Tripkovic, D. Strmcnik, K.-C. Chang, M. Uchimura, A. P. Paulikas, V. Stamenkovic and N. M. Markovic, Science, 2011, 334, 1256–1260 CrossRef CAS PubMed.
- F. Long, X. Zhang, X. Li, F. Sun, T. Zhou and L. Liu, ACS Appl. Mater. Interfaces, 2024, 16, 1416–1427 CrossRef CAS PubMed.
- H. Ze, Z.-L. Yang, M.-L. Li, X.-G. Zhang, Y.-L. A, Q.-N. Zheng, Y.-H. Wang, J.-H. Tian, Y.-J. Zhang and J.-F. Li, J. Am. Chem. Soc., 2024, 146, 12538–12546 CrossRef CAS PubMed.
- C. Zheng, Y. Mao, T. E. Markland and S. G. Boxer, J. Am. Chem. Soc., 2025, 147, 6227–6235 CrossRef CAS PubMed.
- L. Zhang, H. Hu, C. Sun, D. Xiao, H.-T. Wang, Y. Xiao, S. Zhao, K. H. Chen, W.-X. Lin, Y.-C. Shao, X. Wang, C.-W. Pao and L. Han, Nat. Commun., 2024, 15, 7179 CrossRef CAS PubMed.
- Y. Gao, S. Qian, H. Wang, W. Yuan, Y. Fan, N. Cheng, H. Xue, T. Jiang and J. Tian, Appl. Catal., B, 2023, 320, 122014 CrossRef CAS.
- B. Mao, P. Sun, Y. Jiang, T. Meng, D. Guo, J. Qin and M. Cao, Angew. Chem., Int. Ed., 2020, 59, 15232–15237 CrossRef CAS PubMed.
- A. H. Shah, Z. Zhang, Z. Huang, S. Wang, G. Zhong, C. Wan, A. N. Alexandrova, Y. Huang and X. Duan, Nat. Catal., 2022, 5, 923–933 CrossRef CAS.
- L. Guo, J. Chi, T. Cui, J. Zhu, Y. Xia, H. Guo, J. Lai and L. Wang, Adv. Energy Mater., 2024, 14, 2400975 CrossRef CAS.
- Q. Wen, Y. Lin, Y. Yang, R. Gao, N. Ouyang, D. Ding, Y. Liu and T. Zhai, ACS Nano, 2022, 16, 9572–9582 CrossRef CAS PubMed.
- X. Wang, H. Hu, J. Song, J. Ma, H. Du, J. J. Wang, M. Wang, W. Chen, Y. Zhou, J. Wang, M. Yang and L. Zhang, Adv. Energy Mater., 2025, 15, 2402883 CrossRef CAS.
- Q. Sun, N. J. Oliveira, S. Kwon, S. Tyukhtenko, J. J. Guo, N. Myrthil, S. A. Lopez, I. Kendrick, S. Mukerjee, L. Ma, S. N. Ehrlich, J. Li, W. A. Goddard, Y. Yan and Q. Jia, Nat. Energy, 2023, 8, 859–869 CrossRef CAS.
- H. Hu, X. Wang, Z. Zhang, J. Liu, X. Yan, X. Wang, J. Wang, J. P. Attfield and M. Yang, Adv. Mater., 2025, 37, 2415421 CrossRef CAS PubMed.
- Y.-X. Xiao, J. Ying, J.-B. Chen, X. Yang, G. Tian, J.-H. Li, C. Janiak and X.-Y. Yang, Adv. Funct. Mater., 2025, 35, 2418264 CrossRef CAS.
- Z.-J. Wu, J.-H. Li, J. Ying and C. Janiak, Small, 2025, 21, 2504302 CrossRef CAS PubMed.
- J.-B. Chen, H. Wang, Y.-X. Xiao and J. Ying, Chem. Mater., 2025, 37, 2145–2154 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |