Interlayer chemical confinement enables highly reversible and durable lithium–chlorine batteries
Baoquan
Liu
,
Yanzeng
Ge
,
Changfeng
Lin
,
Haizhen
Jiang
,
Tianyu
Qiu
,
Jing
Li
,
Hui
Zhang
,
Jinlin
Yang
* and
Xinlong
Tian
 *
*
State Key Laboratory of Tropic Ocean Engineering Materials and Materials Evaluation, Hainan Provincial Key Lab of Fine Chem, Hainan University, Haikou, 570228, China. E-mail: tianxl@hainanu.edu.cn; yangjl@hainanu.edu.cn
First published on 10th March 2026
Abstract
Rechargeable lithium–chlorine (Li–Cl2) batteries represent a promising high-energy-density technology for use in large-scale applications. However, conventional cathodes suffer from weak interactions with Cl2 and uneven LiCl deposition, resulting in poor Cl2/LiCl conversion kinetics and limited cycle life. In this study, an interlayer chemical confinement strategy is proposed to enable highly reversible Cl2/LiCl conversion in Ti3C2Sx MXenes. Specifically, the confined space in the MXene interlayer effectively suppresses Cl2 escape while the sulfur surface terminations form covalent interactions with Cl to further immobilize Cl2 and lower the LiCl nucleation barrier to achieve uniform deposition. In situ characterization combined with theoretical calculations reveals that the reversible Cl2/LiCl redox reaction with a low energy barrier occurs within the confined interlayer space. This confinement strategy imparts the Li–Cl2 battery with a high specific capacity of up to 2200 mAh g−1, in conjunction with long cycle lives of 1300 and 1200 cycles when operating at 25 and −60 °C, respectively. This study provides new insights into the nanoconfinement effect and opens a promising avenue for the development of high-performance metal–Cl2 batteries.
Broader contextRechargeable lithium–chlorine (Li–Cl2) batteries are promising next-generation energy storage systems with high energy density and excellent wide-temperature operation capability. However, the escape of Cl2 and uneven deposition of LiCl reduce the reversibility of the Cl2/LiCl conversion, resulting in unsatisfactory electrochemical performance of the Li–Cl2 batteries. Although some porous materials have been explored as a cathode to trap Cl2 through physical confinement, their practical effectiveness has been limited, resulting in a short cycle life of ≤300 cycles. To address the aforementioned issues, herein, we design a sulfur-functionalized MXene with two-dimensional interlayer channels as a cathode to simultaneously immobilize Cl2 and lower the LiCl nucleation barrier. The proposed interlayer chemical confinement strategy significantly improves the Cl2/LiCl conversion kinetics, thus enabling the Li–Cl2 battery to achieve high specific capacity and record-breaking long-term cycling stability at room and subzero temperatures. |
Introduction
The development of high-energy-density secondary batteries is imperative to meet the growing energy storage demands of modern society.1–4 In this context, rechargeable lithium–chlorine (Li–Cl2) batteries, derived from commercial lithium–thionyl chloride (Li–SOCl2) primary batteries, provide a high energy density of >700 Wh kg−1 and a high output potential of ∼3.5 V, rendering them promising candidates for next-generation energy storage systems.5–8 During the initial discharge of a typical Li–Cl2 battery, the SOCl2-based electrolyte undergoes decomposition and combines with Li+ ions from the Li anode to generate LiCl deposits on the cathode host material. During subsequent cycles, these LiCl deposits undergo a reversible reaction (2LiCl ↔ 2Li+ + Cl2 + 2e−) to achieve charge storage.5 Consequently, the reversibility of the conversion reaction between LiCl and Cl2 determines the capacity and stability of the Li–Cl2 battery.To achieve improved Cl2/LiCl conversion kinetics, two key issues must be addressed, namely the efficient enrichment of Cl2 during charging and the uniform nucleation and growth of LiCl during discharging. In the first case, the weak physical or chemical adsorption of Cl2 onto the cathode host material leads to inevitable Cl2 spillage, ultimately resulting in a loss of chlorine active species and triggering potential side reactions.9,10 In the second case, previous studies have revealed that the uneven deposition of insulating chlorides is the primary cause of increased polarization and subsequent failure in alkali metal–Cl2 batteries.11,12 Therefore, reducing the energy barrier for LiCl nucleation is essential to achieve uniform deposition. Although several cathode materials, including carbon-based and porous organic framework materials,6,9,13–19 have been employed in Li–Cl2 batteries owing to their high specific surface areas and structural diversity, these studies typically fail to simultaneously address two key issues, leading to unsatisfactory electrochemical performance, particularly short cycle life (≤300 cycles).
Two-dimensional (2D) transition metal carbides or/and nitrides, collectively known as MXenes,20,21 offer transformative potential as cathode materials for Li–Cl2 batteries. Their relatively large interlayer spacing and weak interlayer interactions enable the intercalation of various foreign species, including ions, atoms, and molecules.22–24 Consequently, the interlayer-confined space can function as a nanoreactor that confines active Cl2 molecules and LiCl deposits. In addition, the diverse and tunable surface chemistry of MXenes provides opportunities to enhance their interactions with Cl2 and promote the uniform deposition of LiCl,25–27 thereby facilitating efficient redox reactions. Therefore, MXene cathodes present a promising strategy for addressing key challenges in Cl2/LiCl redox chemistry and for enhancing the electrochemical performance of Li–Cl2 batteries.
In this study, interlayer chemical confinement engineering is applied to MXenes to accelerate the Cl2/LiCl conversion kinetics in Li–Cl2 batteries (Fig. 1). Following theoretical predictions, a MXene exhibiting S termination, namely Ti3C2Sx, is selected as a model system. Mechanistic studies reveal that the interlayer confinement effect of Ti3C2Sx facilitates the enrichment of Cl2 and induces LiCl deposition along the interlayer. Meanwhile, the S surface terminations further immobilize Cl2via covalent interactions, while also reducing the nucleation barrier for LiCl. Therefore, the Li–Cl2 battery incorporating a Ti3C2Sx cathode delivers a high specific capacity of 2200 mAh g−1 (based on the mass of Ti3C2Sx) and a prolonged lifespan of 1300 cycles at room temperature. Even at an extremely low temperature of −60 °C, the Li–Cl2 battery can still maintain 1200 cycles. The reported cycling performances outperform state-of-the-art counterparts based on other cathode materials. Detailed characterization and computational analyses are performed to reveal the mechanism of the highly reversible Cl2/LiCl conversion reaction.
 | ||
| Fig. 1 Schematic representation of the proposed interlayer chemical confinement strategy for improving the Cl2/LiCl conversion kinetics in Li–Cl2 batteries. | ||
Results and discussion
Theoretical screening and structural characterization of the MXene
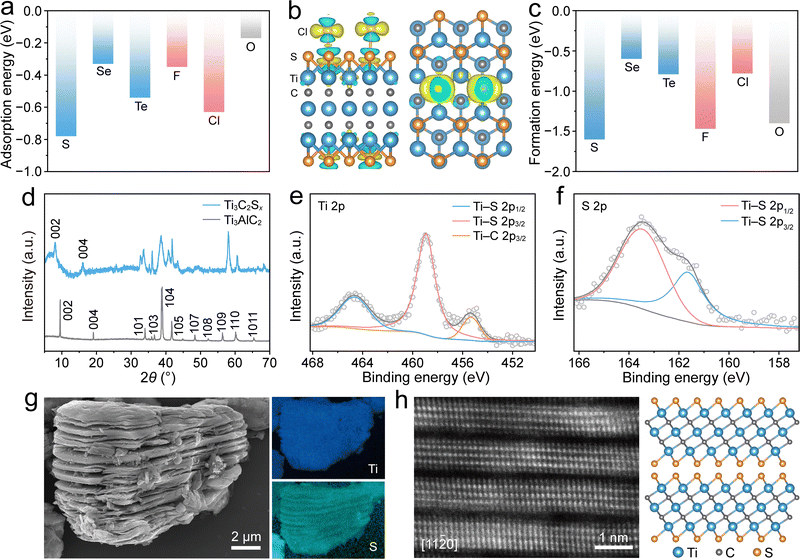
The chemical etching of MXenes and the introduction of specific surface terminations are key to achieving interlayer chemical confinement regulation. Density functional theory (DFT) calculations were performed to analyze the interactions between the Cl2/LiCl and MXene hosts. The Ti3C2Tx MXene (where Tx represents the surface termination groups) was selected as the model system. As shown in Fig. 2a, the adsorption energies of a Cl atom on the O, halogen (F, Cl), and chalcogen (S, Se, Te)-terminated Ti3C2Tx MXenes were calculated to be −0.17, −0.35, −0.63, −0.78, −0.33, and −0.54 eV, respectively. The adsorption energies of the Cl2 molecule on different Ti3C2Tx exhibited a consistent trend, with Ti3C2Sx displaying the highest adsorption energy, indicating its strongest chemical interaction with Cl2 (Fig. S1). The electron density difference map shown in Fig. 2b further supports this strong interaction, indicating a pronounced electron-rich charge density distribution at the Ti3C2Sx–Cl interface, wherein the surface S terminations serve as the adsorption sites. Furthermore, the formation energies of LiCl on different Ti3C2Tx surfaces were calculated (Fig. 2c). The results demonstrated that Ti3C2Sx exhibits a lower LiCl formation energy (−1.62 eV) than the other surface-terminated Ti3C2Tx, suggesting that S termination thermodynamically favors LiCl nucleation and growth. These computational results indicate that Ti3C2Sx possesses the strongest ability to trap Cl2 and promote uniform LiCl deposition, thereby enhancing the Cl2/LiCl conversion kinetics.Guided by the DFT calculations, a chemical scissor-mediated structural editing strategy based on Lewis acidic molten salt (LAMS) etching was employed to synthesize the target S-terminated Ti3C2Sx MXene.28,29 This strategy employs Lewis acidic cations as chemical scissors to create space between the MX sublayers, enabling anions to intercalate into the interlayer and form surface terminations. Such modifications render it possible to obtain specific surface terminations that cannot be achieved via traditional HF etching, such as chalcogen terminations (-S, -Se, -Te).30 The X-ray diffraction (XRD) patterns shown in Fig. 2d indicate that the diffraction peaks corresponding to the Ti3AlC2 precursor weaken or disappear after LAMS etching. In addition, the interlayer gap-dependent (00l) stacking reflections of Ti3C2Sx shifted toward lower angles compared with those of the corresponding Ti3AlC2 specimen, indicating an expanded interlayer distance after etching owing to the removal of the Al layers and the formation of coordination interactions between Ti3C2 and the S anions.31 The interlayer distance for Ti3C2Sx was calculated to be ∼1.1 nm, which is significantly larger than the molecular diameter of Cl2 (0.15 nm), thereby providing sufficient interlayer space to trap Cl2. X-ray photoelectron spectroscopy (XPS) confirmed the coordination between S and Ti, with signals corresponding to Ti–S bonds being observed in the high-resolution Ti 2p and S 2p spectra (Fig. 2e and f).32 Scanning electron microscopy (SEM) imaging revealed a typical accordion-like morphology for Ti3C2Sx (Fig. 2g and Fig. S2), distinct from the bulk morphology of its Ti3AlC2 precursor (Fig. S3). Moreover, energy-dispersive X-ray spectroscopy (EDS) revealed a uniform distribution of surface S terminations (Fig. 2g).
To achieve direct atomic-scale visualization of Ti3C2Sx, a thin lamella was prepared employing the focused ion beam lift-out technique (Fig. S4). Atomic-resolution high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) was utilized to directly observe the atomic configuration of Ti3C2Sx. As shown in Fig. 2h, the STEM image captured along the [11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0] zone axis clearly reveals five atomic layers within each Ti3C2Sx monolayer. The three bright central layers correspond to Ti atoms, whereas the outer layers correspond to surface-terminated S atoms. From the STEM image, the interlayer distance was determined to be ∼1.1 nm, consistent with the XRD results. This interlayer space allows Li+ and Cl− ions to pass freely during discharge, thereby promoting uniform LiCl deposition along the interlayer and preventing excessive crystal growth. However, during the charging process, Cl2 is confined within the channels, effectively preventing its escape. Additionally, the in-plane STEM image captured along the [0001] direction and its corresponding fast Fourier transform patterns revealed that the high crystallinity of Ti3C2Sx was maintained after LAMS etching (Fig. S5).
0] zone axis clearly reveals five atomic layers within each Ti3C2Sx monolayer. The three bright central layers correspond to Ti atoms, whereas the outer layers correspond to surface-terminated S atoms. From the STEM image, the interlayer distance was determined to be ∼1.1 nm, consistent with the XRD results. This interlayer space allows Li+ and Cl− ions to pass freely during discharge, thereby promoting uniform LiCl deposition along the interlayer and preventing excessive crystal growth. However, during the charging process, Cl2 is confined within the channels, effectively preventing its escape. Additionally, the in-plane STEM image captured along the [0001] direction and its corresponding fast Fourier transform patterns revealed that the high crystallinity of Ti3C2Sx was maintained after LAMS etching (Fig. S5).
Electrochemical performance of the Li–Cl2@Ti3C2Sx battery at room temperature
To investigate the electrochemical reversibility of the Cl2/Cl− redox reaction in the Li–Cl2 battery employing Ti3C2Sx as the cathode material (denoted as the Li–Cl2@Ti3C2Sx battery), a two-electrode Swagelok cell was assembled to systematically investigate the electrochemical performance. The Li–Cl2@Ti3C2Sx battery was initially discharged to a cutoff voltage of 2 V at a current density of 0.1 A g−1, which provided an initial discharge capacity of 6860 mAh g−1 and a discharge plateau at ∼3.51 V (Fig. S6). During this process, SOCl2 was reduced to form S, SO2, and LiCl (4Li+ + 2SOCl2 + 4e− → S + SO2 + 4LiCl), which subsequently deposited on the Ti3C2Sx electrode.5 The produced LiCl then acted as the active chlorine species to maintain the reversible LiCl/Cl2 conversion reaction (2LiCl ↔ 2Li+ + Cl2 + 2e−). The LiCl/Cl2 redox couple provides a high theoretical specific capacity of 756 mAh g−1. The electroreduction of SOCl2 was also confirmed by cyclic voltammetry (CV), wherein a distinct reduction peak was observed during the initial cycle (Fig. S7). Subsequently, the Li–Cl2@Ti3C2Sx battery was cycled at 0.5 A g−1 to evaluate the maximum reversible capacity of Ti3C2Sx. As shown in Fig. 3a, upon increasing the cut-off charge capacity from 800 to 2200 mAh g−1, the battery maintained stable discharge capacity with a coulombic efficiency (CE) of ∼99.5%. Additionally, the corresponding voltage profiles recorded under different capacities exhibited low overpotentials of <0.4 V (Fig. S8). The galvanostatic intermittent titration technique (GITT) curve obtained for the Li–Cl2@Ti3C2Sx battery at 1.0 A g−1 further confirmed the low polarization (Fig. S9), exhibiting a small IR drop of only 0.22 V, which is significantly lower than those of previously reported Li–Cl2 batteries based on carbon cathodes.12 Upon further increasing the charging capacity to 2400 mAh g−1 (Fig. S10), fluctuations occurred during the cycling process, allowing the maximum storage capacity of Ti3C2Sx to be defined as 2200 mAh g−1. The rate capability of the Li–Cl2@Ti3C2Sx battery was also investigated (Fig. 3b). Specifically, at a cut-off charge capacity of 1200 mAh g−1, the battery maintained a discharge capacity of >1180 mAh g−1 upon increasing the current density from 0.5 to 6 A g−1. Notably, the discharge voltage plateau exhibited only a marginal decrease from 3.5 to 3.2 V (Fig. 3c), further confirming the fast Cl2/LiCl kinetics facilitated by Ti3C2Sx.Subsequently, the cycling performance of the Li–Cl2@Ti3C2Sx battery was evaluated at a fixed capacity of 1200 mAh g−1 using different current densities. The battery initially underwent a low-current activation process, and the CE rapidly approached 100% within the first five cycles (Fig. S11). As expected, the battery exhibited long-term cycling stabilities of >1000, 1300, and 400 cycles at current densities of 2, 3, and 5 A g−1, respectively (Fig. 3d and Fig. S12), in conjunction with an average CE of >99.2% throughout each cycling process. Upon increasing the charge capacity to 2000 mAh g−1, the Li–Cl2@Ti3C2Sx battery retained ∼98.5% CE over 200 cycles (Fig. S13). Notably, the current Li–Cl2@Ti3C2Sx battery outperformed previously reported Li–Cl2 batteries incorporating carbon-based and porous organic molecular materials in terms of its current density, cycle lifetime, and accumulated capacity (Fig. 3e and Table S1). This excellent long-term stability can be attributed to the interlayer chemical confinement provided by Ti3C2Sx, which traps Cl2 and LiCl within the interlayer, thereby preventing the loss of active species. XRD patterns and HAADF-STEM image demonstrated that the layered structure of Ti3C2Sx with a spacing of 1.1 nm was well preserved after long-term cycling (Fig. S14). Furthermore, Ti3C2Tx materials exhibiting other chalcogen surface terminations (i.e., Se and Te) were synthesized using the same LAMS etching method, and their electrochemical performances were compared with those of Ti3C2Sx (Fig. S15–S19). In contrast, the Li–Cl2@Ti3C2Sex and Li–Cl2@Ti3C2Tex batteries exhibited short cycle lives of 280 and 340 cycles at 1 A g−1, respectively, indicating their limited Cl2/LiCl conversion capability owing to the weak interactions between Cl2 and the Se and Te terminations, in conjunction with the high energy associated with LiCl formation (Fig. 2a and c, and Fig. S20).
Incorporating the fast Cl2/LiCl conversion kinetics in the Li–Cl2@Ti3C2Sx battery, its electrochemical performance was subsequently evaluated at high mass loadings (Fig. S21). Notably, the Li–Cl2@Ti3C2Sx battery with an ultra-high mass loading of 10 mg cm−2 achieved a specific capacity of 1200 mAh g−1 at 0.5 A g−1, and maintained stable operation over 150 cycles. This result indicates that the exceptional electrochemical performance of the Li–Cl2@Ti3C2Sx battery could be also preserved at industrial-level loadings. To validate the practical viability of this Li–Cl2@Ti3C2Sx battery, a pouch-type Li–Cl2@Ti3C2Sx cell (5 cm × 7 cm) was assembled (Fig. 3f). When tested at a current density of 0.5 A g−1, the pouch cell achieved a high initial capacity of 34 mAh (1.7 mAh cm−2) and maintained 98% capacity retention after 50 cycles (Fig. 3g). The E/C ratio (electrolyte weight to cathode capacity ratio) and N/P ratio (negative-to-positive electrode capacity ratio) were calculated to be 41.8 g Ah−1 and 6, respectively. Optical photographs demonstrate that no swelling was observed in the pouch cell after cycling, indicating that the interlayer confinement effect of Ti3C2Sx effectively immobilizes Cl2 (Fig. S22). Furthermore, the voltage profile of the pouch cell exhibited a discharge plateau at ∼3.52 V, similar to the results observed in a corresponding Swagelok cell (Fig. 3h). The excellent electrochemical stability and rechargeability of the Li–Cl2@Ti3C2Sx battery were also confirmed via a retention capability test, which showed negligible performance degradation over a total cycling duration of >19 days (Fig. S23).
Electrochemical performance of the Li–Cl2@Ti3C2Sx battery at subzero temperature
The development of high-energy-density rechargeable batteries capable of stable operation under low temperature conditions remains challenging owing to insufficient charge transfer kinetics and reaction reversibility.33,34 Owing to the excellent Cl2/LiCl conversion reaction kinetics provided by Ti3C2Sx and the low freezing point of SOCl2 (−105 °C), the Li–Cl2@Ti3C2Sx battery exhibited favorable electrochemical performances at extremely low temperatures. Specifically, although the Li–Cl2@Ti3C2Sx battery delivered a reduced initial discharge capacity (3887 mAh g−1) at −20 °C compared with that recorded at room temperature (Fig. S24), the generated LiCl deposits were sufficient to support subsequent reversible reactions. As shown in Fig. 4a, at a high current density of 0.5 A g−1 and a cut-off charge capacity of 1200 mAh g−1, the battery exhibited an impressive cycle stability of >1200 cycles, with an average CE of >99.5%. The voltage profiles remained stable throughout cycling (Fig. 4b and Fig. S25), and the discharge plateau of the battery remained constant at 3.3 V, which is essentially consistent with the room temperature result. Even upon reducing the temperature to −60 °C, an ultra-long cycle life of 1200 cycles was achieved (Fig. 4c), demonstrating the viability of the Li–Cl2@Ti3C2Sx battery for operation in extremely low-temperature environments. Therefore, compared with the low-temperature cycling performance observed for previously reported Li–Cl2 batteries and other Li metal batteries, the current Li–Cl2@Ti3C2Sx battery demonstrates a significant advantage (Fig. 4d and Table S2). The calculated density of states revealed that Ti3C2Sx is metallic in nature, with a finite density of states at the Fermi level mainly arising from the Ti d-orbitals (Fig. S26), thereby facilitating efficient charge transfer and enhancing its low-temperature performance.As a proof of concept, a pouch-type Li–Cl2@Ti3C2Sx battery was fabricated and utilized to power a digital clock for >2 days at −60 °C (Fig. 4e), demonstrating its potential for application under harsh environmental conditions, such as those encountered in space, deep oceans, and polar regions (Fig. 4c inset). Furthermore, the battery retention performance was investigated at −60 °C under fully charged conditions (Fig. 4f). After an overall retention time of 46 days, the battery retained ∼100% of its initial capacity, showing long-term reliability even at extremely low temperatures.
Reaction evolution of the chlorine species
To elucidate the dynamic evolution of redox intermediates and the reversibility of the LiCl/Cl2 conversion reaction in the Li–Cl2@Ti3C2Sx battery, comprehensive characterization was conducted during electrochemical cycling. Specifically, in situ Raman spectroscopy was deployed to capture the real-time reaction intermediates of SOCl2. After the initial discharge (Fig. 5a), the Raman peaks attributed to SOCl2 weakened or disappeared, in conjunction with the appearance of sulfur Raman peaks located at 150, 216, and 471 cm−1. These peak changes were caused by the electroreduction of SOCl2 (4Li+ + 2SOCl2 + 4e− → S + SO2 + 4LiCl), which occurs during discharge. Over subsequent cycles, the Raman peaks ascribed to sulfur underwent reversible changes (Fig. 5b). These peaks disappeared upon charging to the cut-off capacity and gradually reappeared during the discharge process. This was attributed to the reversible reactions between S, SCl2, and S2Cl2, where S reacts with the Cl2 produced by the electrooxidation of LiCl during charging to form SCl2 and S2Cl2, which are trapped in the Ti3C2Sx interlayer; SCl2 and S2Cl2 can then be reversibly reduced back to S and LiCl during discharge.Given that the deposition and dissolution of LiCl are crucial processes in reversible electrochemical reactions, in situ XRD was employed to monitor the evolution of this species during the charge/discharge processes. As shown in Fig. 5c, the diffraction peaks ascribed to the (111), (200), and (220) planes of LiCl (i.e., at 30.2°, 34.9°, and 50.2°, respectively),15 progressively intensified during discharge, indicating the formation of crystalline LiCl (2Li + Cl2 + 2e− → 2LiCl). In contrast, the intensity of these peaks decreased until returning to their initial state during the charge process, consistent with the oxidation of deposited LiCl crystallites to generate Cl2 (2LiCl → 2Li + Cl2 + 2e−). SEM imaging revealed the uniform deposition and reversible stripping of LiCl crystallites on the Ti3C2Sx electrode during the charge and discharge processes (Fig. S27), indicating that LiCl deposition/stripping was readily reversible in the current system. Furthermore, electrochemical impedance spectroscopy (EIS) was performed at different discharge/charge states to confirm the generation of LiCl (Fig. 5d and Fig. S28). During discharge, the deposition of insulated LiCl on the surface of the Ti3C2Sx electrode increases the charge transfer resistance (Rct), as evidenced by the enlarged semicircle diameter in the high-frequency region. Upon charging to a cut-off capacity of 1200 mAh g−1, Rct gradually decreased owing to the stripping of LiCl from the electrode surface.
Subsequently, time-of-flight secondary-ion mass spectrometry (TOF-SIMS) was performed to investigate the spatial distribution of chemical species in the discharged Ti3C2Sx electrode. The TOF-SIMS depth-profiling demonstrated that the signals corresponding to the electroreduction products of SOCl2, namely LiCl−, S−, and SO2−, gradually intensified and stabilized as the ion sputtering time/depth was increased (Fig. 5e). A three-dimensional (3D) view of the secondary ion signals constructed from their depth profiles showed a uniform distribution of discharge products inside the Ti3C2Sx electrode (Fig. 5f). High-resolution chemical maps indicated that the LiCl, S, and SO2 species remained stable within the electrode (Fig. S29), thereby confirming that these three primary species were predominantly confined within the interlayer space of the Ti3C2Sx electrode, rather than on the surface. Furthermore, the oxidation of LiCl to Cl2 and its confinement within the MXene interlayer were verified by mass spectrometry. Compared with the discharged electrode, the charged electrode exhibited a more pronounced Cl2 signal under heating (Fig. S30), indicating that the oxidation of LiCl generated Cl2 during charging. To further demonstrate the interlayer confinement effect of MXenes on immobilized Cl2, few-layer Ti3C2Sx nanosheets were synthesized via the liquid-phase exfoliation method (Fig. S31). Although the exfoliated samples (69.9 m2 g−1) exhibited a higher specific surface area compared to unexfoliated multilayer samples (15.2 m2 g−1, Fig. S32), the former demonstrate inferior performance, giving a maximum capacity and lifespan of 1600 mAh g−1 and 200 cycles, respectively (Fig. S33). This result indicated that the interlayer channels formed by the multilayer MXene significantly suppressed Cl2 release. Similar results were also observed in previous studies, where a multilayer MXene exhibited enhanced H2 storage capacity owing to the “nanopump effect” in the interlayer space.23
XPS analyses were conducted to investigate the interactions between the S surface terminations and the Cl2 molecules. The high-resolution S 2p XPS spectrum exhibited a shift to a higher energy upon charging (Fig. 5g), which can be attributed to electron transfer from the S terminations to Cl2, reflecting the strong electron-withdrawing effect of Cl2 on the S atoms in the Ti3C2Sx host. In addition, the high-resolution Cl 2p XPS spectrum showed that the binding energy of the Cl 2p component shifted toward a higher energy when transitioning from the discharged state to the charged state (Fig. S34). This positive binding energy shift indicates an increase in the oxidation state of chlorine within the host material and provides direct evidence of strong chemical adsorption between the interlayer chlorine species and the surface S terminations.17
Theoretical investigation of the LiCl/Cl2 conversion reaction
COMSOL Multiphysics simulations were conducted to visualize the LiCl concentration distribution on the Ti3C2Sx host (Fig. 6a). Notably, LiCl was uniformly distributed on the interlayer surfaces of Ti3C2Sx with a minimal concentration gradient, which can be attributed to the interlayer confinement effect and the strong binding affinity of the S terminations. This behavior was further confirmed via electrostatic potential (ESP) mapping (Fig. 6b), which showed a strong negative surface electrostatic potential near the S terminations, indicating a significant accumulation of electrons that facilitates the chemical adsorption of Cl2 molecules. The 2D charge distribution map of the electron localization function (ELF) indicated that a polar covalent bond interaction (ELF = 0.5) was formed between the S terminations of the upper/lower layers and the interlayer-adsorbed Cl2 molecules (Fig. 6c and Fig. S35), which promoted the dissociation of Cl2 molecules between the Ti3C2Sx layers. This interlayer decoupling interaction was also validated employing crystal orbital Hamilton population (COHP) analysis, which revealed substantial bonding interactions between the S and Cl orbitals at the Fermi level. Additionally, the integrated COHP (ICOHP) reached −0.38 eV (Fig. 6d), indicating the formation of covalent interactions between the S and Cl atoms. These results suggest that the Cl2 molecules can be effectively restricted within the Ti3C2Sx structure, thereby promoting their adsorption and dissociation.To reveal the mechanism by which Ti3C2Sx catalyzes the Cl2/LiCl conversion, the Gibbs free energy change (ΔG) during the discharge and charge processes was further evaluated. As shown in Fig. 6e and Fig. S36, the adsorption and dissociation of Cl2 (M + Cl2 → *Cl2) show the highest ΔG among all sub-steps, rendering them rate-limiting steps in the discharge reduction process. It was also observed that, among the differently terminated MXenes, the S-terminated Ti3C2Sx demonstrates the lowest ΔG of 0.96 eV, indicating that this material effectively reduces the energy for the adsorption and dissociation of Cl2, facilitating its efficient conversion to LiCl. During charging, Ti3C2Sx also demonstrates a thermodynamic advantage in the oxidation of LiCl to Cl2 (Fig. 6f and Fig. S37), giving the lowest ΔG of 1.36 eV. Overall, these theoretical results confirm that Ti3C2Sx can effectively trap Cl2 and reduce the energy for the LiCl/Cl2 conversion, thereby endowing Li–Cl2 batteries with exceptional stability and reversibility.
Conclusion
In summary, an interlayer chemical confinement strategy was developed to achieve highly reversible Cl2/LiCl conversion for Li–Cl2 batteries. This concept was implemented using Ti3C2Sx MXene as a model electrode material. Detailed characterization and calculation analyses revealed that the interlayer confinement effect of the MXene and its unique S termination successfully enriched Cl2 and induced uniform LiCl deposition, enabling reversible conversion within the confined interlayer space while maintaining a low energy barrier. Surprisingly, the Li–Cl2 battery based on the Ti3C2Sx cathode exhibited outstanding durability over 1300 and 1200 cycles at temperatures of 25 and −60 °C, respectively. Moreover, the potential of a pouch-type Li–Cl2 battery based on the Ti3C2Sx cathode was clearly demonstrated in the context of practical applications. These findings open a new avenue for achieving reversible chlorine redox chemistry in rechargeable metal–Cl2 batteries.Author contributions
B. L., J. Y., and X. T. proposed the concept and designed the experiments. J. Y., and X. T. co-supervised the project. B. L. and Y. G. carried out the material characterization and electrochemical measurements. H. J. assisted with electrochemical measurements. T. Q. and C. L. carried out the theoretical simulations. J. L. and H. Z. helped to discuss and analyze the data. B. L., J. Y., and X. T. co-wrote the manuscript. All authors discussed the data and commented on the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI): detailed experimental section, additional characterization of Ti3C2Sx MXene, electrochemical performance of Li–Cl2@Ti3C2Sx batteries, theoretical calculations, and comparison table of battery performance. See DOI: https://doi.org/10.1039/d5ee07649h.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (52304326, 52274297, 52164028, 22305055, 22505051, and 52563019), the Hainan Province Science and Technology Special Fund (ZDYF2024SHFZ072, ZDYF2025SHFZ024), the Start-up Research Foundation of Hainan University (KYQD(ZR)-23073, 20008), the Specific Research Fund of The Innovation Platform for Academicians of Hainan Province (YSPTZX202315), and the Collaborative Innovation Center of Marine Science and Technology, Hainan University (XTCX2022HYC18, XTCX2022HYC05). The authors acknowledge the support for comprehensive characterization by the Pico Electron Microscopy Center of Hainan University. Computations were performed at the Hefei Advanced Computing Center.References
- J. Kim, N. Kim, Z. Ju, Y. Hong, K. Kang, J. Pang, S. Lee, S. Chae, M. Park, J. Kim, G. Yu and S. Lee, Nat. Energy, 2025, 10, 295–307 CrossRef.
- T. Jiang, D. Shen, Z. Zhang, H. Liu, G. Zhao, Y. Wang, S. Tan, R. Luo and W. Chen, Nat. Rev. Clean Technol., 2025, 1, 474–492 CrossRef.
- Z. Zhu, T. Jiang, M. Ali, Y. Meng, Y. Jin, Y. Cui and W. Chen, Chem. Rev., 2022, 122, 16610–16751 CrossRef PubMed.
- J. W. Choi and D. Aurbach, Nat. Rev. Mater., 2016, 1, 16013 Search PubMed.
- Z. Xie, L. Sun, M. Sajid, Y. Feng, Z. Lv and W. Chen, Chem. Soc. Rev., 2024, 53, 8424–8456 Search PubMed.
- G. Zhu, X. Tian, H. Tai, Y. Li, J. Li, H. Sun, P. Liang, M. Angell, C. Huang, C. Ku, W. Hung, S. Jiang, Y. Meng, H. Chen, M. Lin, B. Hwang and H. Dai, Nature, 2021, 596, 525–530 Search PubMed.
- H. Li, D. Zhang, H. Qian, R. Chen, Y. Cao, X. Ai and J. Wu, J. Energy Chem., 2026, 113, 365–401 CrossRef.
- P. Li, X. Li, Y. Guo, A. Chen, R. Zhang, Y. Hou, Q. Xiong, Y. Wang, Z. Chen, J. Zhu, M. Zhu and C. Zhi, Chem, 2024, 10, 352–364 Search PubMed.
- Y. Xu, L. Jiao, J. Ma, P. Zhang, Y. Tang, L. Liu, Y. Liu, H. Ding, J. Sun, M. Wang, Z. Li, H. Jiang and W. Chen, Joule, 2023, 7, 515–528 CrossRef.
- J. Han, X. Wei, G. Ma, H. Wang, Y. Sun, Z. Liu, L. Zhao, C. Ma, Q. Liu, W. Feng, D. Kong, W. Lv, Q. Yang and L. Zhi, Energy Environ. Sci., 2025, 18, 8052–8065 Search PubMed.
- W. Feng, X. Wei, J. Yang, C. Ma, Y. Sun, J. Han, D. Kong and L. Zhi, Nat. Commun., 2024, 15, 6904 CrossRef PubMed.
- X. Wei, D. Kong, W. Feng, F. Cao, C. Ma, J. Yang, J. Han, L. Wang, G. Ma, H. Wang, Y. Sun, Z. Liu, L. Wei and L. Zhi, Adv. Energy Mater., 2025, 15, 2501333 Search PubMed.
- G. Zhu, P. Liang, C. Huang, C. Huang, Y. Li, S. Wu, J. Li, F. Wang, X. Tian, W. Huang, S. Jiang, W. Huang, H. Chen, M. Lin, B. Hwang and H. Dai, J. Am. Chem. Soc., 2022, 144, 22505–22513 CrossRef CAS.
- P. Liang, G. Zhu, W. Wang, C. Huang, S. Wu, J. Zhou, X. Zhou, Y. Wu, S. Wang, M. Wang, L. Zhang, C. Ming, J. Li, F. Wang, M. Sun, Y. Li, B. Hwang and H. Dai, J. Am. Chem. Soc., 2025, 147, 18541–18549 Search PubMed.
- Y. Xu, S. Zhang, M. Wang, Y. Meng, Z. Xie, L. Sun, C. Huang and W. Chen, J. Am. Chem. Soc., 2023, 145, 27877–27885 Search PubMed.
- Y. Xu, M. Wang, M. Sajid, Y. Meng, Z. Xie, L. Sun, J. Jin, W. Chen and S. Zhang, Angew Chem., Int. Ed., 2024, 63, e202315931 CrossRef CAS.
- P. Li, C. Ma, Y. Wang, S. Zhai, G. Ma, D. Kong and Z. Li, Adv. Mater., 2025, 37, 2418990 Search PubMed.
- G. Chen, W. Li, X. Du, C. Wang, X. Qu, X. Gao, S. Dong, G. Cui and L. Chen, J. Am. Chem. Soc., 2023, 145, 22158–22167 Search PubMed.
- B. Yuan, L. Wu, S. Geng, Q. Xu, X. Zhao, Y. Wang, M. Liao, L. Ye, Z. Qu, X. Zhang, S. Wang, Z. Ouyang, S. Tang, H. Peng and H. Sun, Angew. Chem. Int. Ed., 2023, 62, e202306789 Search PubMed.
- A. VahidMohammadi, J. Rosen and Y. Gogotsi, Science, 2021, 372, eabf1581 CrossRef CAS PubMed.
- B. Anasori, M. R. Lukatskaya and Y. Gogotsi, Nat. Rev. Mater., 2017, 2, 16098 Search PubMed.
- M. R. Lukatskaya, O. Mashtalir, C. E. Ren, Y. Dall Agnese, P. Rozier, P. L. Taberna, M. Naguib, P. Simon, M. W. Barsoum and Y. Gogotsi, Science, 2013, 341, 1502–1505 CrossRef CAS PubMed.
- S. Liu, J. Liu, X. Liu, J. Shang, L. Xu, R. Yu and J. Shui, Nat. Nanotechnol., 2021, 16, 331–336 CrossRef CAS PubMed.
- H. Chen, H. Ma and C. Li, ACS Nano, 2021, 15, 15502–15537 CrossRef CAS PubMed.
- M. Jiang, D. Wang, Y. Kim, C. Duan, D. V. Talapin and C. Zhou, Angew Chem., Int. Ed., 2024, 63, e202409480 CrossRef CAS PubMed.
- J. Yang, Y. Zhang, Y. Song, Y. Ge, S. Tang, J. Li, H. Zhang, D. Wu and X. Tian, J. Am. Chem. Soc., 2024, 146, 25680–25688 CrossRef CAS PubMed.
- H. H. S. Thangavelu, C. Huang, F. Chabanais, J. Palisaitis and P. O. Å. Persson, Adv. Funct. Mater., 2025, e15604 Search PubMed.
- H. Ding, Y. Li, M. Li, K. Chen, K. Liang, G. Chen, J. Lu, J. Palisaitis, P. O. Å. Persson, P. Eklund, L. Hultman, S. Du, Z. Chai, Y. Gogotsi and Q. Huang, Science, 2023, 379, 1130–1135 CrossRef CAS PubMed.
- H. Ding, Y. Li, M. Li, Z. Chai and Q. Huang, Acc. Mater. Res., 2025, 6, 28–39 CrossRef CAS.
- Y. Wei, P. Zhang, R. A. Soomro, Q. Zhu and B. Xu, Adv. Mater., 2021, 33, 2103148 CrossRef CAS PubMed.
- M. Alhabeb, K. Maleski, B. Anasori, P. Lelyukh, L. Clark, S. Sin and Y. Gogotsi, Chem. Mater., 2017, 29, 7633–7644 CrossRef CAS.
- V. Kamysbayev, A. S. Filatov, H. Hu, X. Rui, F. Lagunas, D. Wang, R. F. Klie and D. V. Talapin, Science, 2020, 369, 979–983 CrossRef CAS PubMed.
- Z. Li, Y. Yao, S. Sun, C. Jin, N. Yao, C. Yan and Q. Zhang, Angew Chem., Int. Ed., 2023, 62, e202303888 Search PubMed.
- F. Zhang, B. He, Y. Xin, T. Zhu, Y. Zhang, S. Wang, W. Li, Y. Yang and H. Tian, Chem. Rev., 2024, 124, 4778–4821 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |